Development of a Nanoscaled Ion Source for High-Sensitivity Photoionization Mass SpectrometryClick to copy article linkArticle link copied!
- Laura TenhumbergLaura TenhumbergMax-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, 45470 Mülheim a. d. Ruhr, GermanyMore by Laura Tenhumberg
- Wolfgang SchraderWolfgang SchraderMax-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, 45470 Mülheim a. d. Ruhr, GermanyMore by Wolfgang Schrader
- Alessandro Vetere*Alessandro Vetere*Email: [email protected]Max-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, 45470 Mülheim a. d. Ruhr, GermanyMore by Alessandro Vetere
Abstract
Atmospheric Pressure Photoionization (APPI) has emerged as a versatile ionization method in mass spectrometry, able to ionize compounds of comparably low ionization potential (typically <10.6 eV), irrespective of their functionalities. Aromatic analytes are particularly well suited for photoionization because their delocalized π-electron systems result in comparatively low ionization potentials, enabling both direct single-photon ionization and ionization by charge transfer from dopant ions. While highly effective for a broad range of analytes, APPI faces issues of high sample consumption and elevated background noise, which can limit its effectiveness in trace analysis. Prior advances in miniaturizing flow rates, as seen with the transition from conventional Electrospray to nano-Electrospray, have shown that reducing flow rates not only conserves sample but can also enhances sensitivity by increasing the signal-to-noise ratio. Applying similar miniaturization strategies to APPI holds promise for overcoming current limitations and improving its analytical performance. This study investigates the impact of lowered analyte concentrations and reduced flow rates on the sensitivity of APPI for ultratrace analysis. We introduce a prototype ion source for APPI applications and systematically explore the effects of flow rates below 1 μL min–1 on APPI performance, evaluating the signal-to-noise ratios and detection limits achieved. Our findings indicate that reduced flow rates significantly improve sensitivity, demonstrating the potential to detect ultratrace levels of environmental pollutants with higher efficiency and lower background interference.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Introduction
| 1) | Excitation and/or photodissociation of compounds like water, methanol or oxygen leads to the presence of reactive species (atomic oxygen, OH•, CH3•, CH3O•) that promote oxidation of analyte molecules/ions and (15) | ||||
| 2) | the absorption of photons leads to an exponential decay of light intensity with growing distance from the light source, i.e., the availability of photons for direct photoionization of low abundant analytes is generally poor. | ||||
Experimental Section
Ion Source Design
Figure 1
Figure 1. Photo of the nAPPI source coupled to a TSQ triple quadrupole MS (left) and schematics (right).
Mass Spectrometry
Results and Discussion
Addressing High Background Noises in Photoionization
Figure 2
Figure 2. Pure toluene blanks for APPI and nAPPI at different flow rates. *Plasticizer impurities.
Figure 3
Figure 3. Measurements of phenanthrene in toluene at different concentrations with APPI at a flow rate of 50 μL min–1 and nAPPI at a flow rate of only 1 μL min–1. All spectra were measured on a TSQ triple quadrupole MS. *Plasticizer impurities.
Figure 4
Figure 4. PAH/PAXH, 10 mg L–1 each in toluene, measured with APPI and nAPPI at different flow rates (measured on a TSQ triple quadrupole MS). *Plasticizer impurities.
Improvement in Signal-to-Noise Ratio
Figure 5
Figure 5. S/N calculations for nAPPI (flow rate of 1 μL min–1) and conventional APPI (flow rate of 50 μL min–1).
Figure 6
Figure 6. Mass traces of loop injections of phenanthrene in toluene at low concentrations for nAPPI and APPI.
The Limit of Detection
Alternative nAPPI Analytes
Figure 7
Figure 7. Spectra of different photoionizable analytes spanning a broader polarity range measured with APPI and nAPPI on a TSQ triple quadrupole MS. β-Estradiol and paracetamol were dissolved from neat compounds, and ibuprofen was measured from a pill. *Plasticizer impurities.
Figure 8
Figure 8. Two larger photoionizable analytes, reserpine (left) and gramicidin S (right) measured with APPI and nAPPI.
nAPPI Performance with Real-World Samples
Figure 9
Figure 9. On the left: A complex sample of a heavy crude oil in toluene measured with conventional APPI and nAPPI. On the right: Assigned compositions of the heavy crude oil. While less oxygen-containing compounds are observed, there is an increased tendency for the formation of radical cations in nAPPI than in APPI.
Figure 10
Figure 10. Kendrick plots of the S and S [H] class of the heavy crude for APPI and nAPPI.
Conclusion
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.analchem.5c06912.
List of reference compounds and additional graphs highlighting the characteristics of the source (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We thank the Bundesministerium für Wirtschaft und Klimaschutz (BMWK) for funding through the ZIM Project KK5171402BR3 in cooperation with MasCom Technologies GmbH. We also thank Sebastian Plankert and Dirk Ullner from the department of precision mechanics at the Max-Planck-Institut für Kohlenforschung for turning our ideas into a fully functional device.
References
This article references 37 other publications.
- 1Gaspar, A.; Zellermann, E.; Lababidi, S.; Reece, J.; Schrader, W. Impact of different ionization methods on the molecular assignments of asphaltenes by FT-ICR mass spectrometry. Anal. Chem. 2012, 84 (12), 5257– 5267, DOI: 10.1021/ac300133pGoogle ScholarThere is no corresponding record for this reference.
- 2Cai, S.-S.; Syage, J. A. Comparison of atmospheric pressure photoionization, atmospheric pressure chemical ionization, and electrospray ionization mass spectrometry for analysis of lipids. Anal. Chem. 2006, 78 (4), 1191– 1199, DOI: 10.1021/ac0515834Google ScholarThere is no corresponding record for this reference.
- 3Huba, A. K.; Huba, K.; Gardinali, P. R. Understanding the atmospheric pressure ionization of petroleum components: The effects of size, structure, and presence of heteroatoms. Sci. Total Environ. 2016, 568, 1018– 1025, DOI: 10.1016/j.scitotenv.2016.06.044Google ScholarThere is no corresponding record for this reference.
- 4Kauppila, T.; Nikkola, T.; Ketola, R. A.; Kostiainen, R. Atmospheric pressure photoionization-mass spectrometry and atmospheric pressure chemical ionization-mass spectrometry of neurotransmitters. J. Mass Spectrom. 2006, 41 (6), 781– 789, DOI: 10.1002/jms.1034Google ScholarThere is no corresponding record for this reference.
- 5Gowda, G. N.; Djukovic, D. Overview of mass spectrometry-based metabolomics: opportunities and challenges. Methods Mol. Biol. (Clifton, N.J.) 1198 2014, 1198, 3– 12, DOI: 10.1007/978-1-4939-1258-2_1Google ScholarThere is no corresponding record for this reference.
- 6Nadler, W. M.; Waidelich, D.; Kerner, A.; Hanke, S.; Berg, R.; Trumpp, A.; Rösli, C. MALDI versus ESI: the impact of the ion source on peptide identification. J. Proteome Res. 2017, 16 (3), 1207– 1215, DOI: 10.1021/acs.jproteome.6b00805Google ScholarThere is no corresponding record for this reference.
- 7Jia, S.; Marques Dos Santos, M.; Li, C.; Snyder, S. A. Recent advances in mass spectrometry analytical techniques for per-and polyfluoroalkyl substances (PFAS). Anal. Bioanal. Chem. 2022, 414 (9), 2795– 2807, DOI: 10.1007/s00216-022-03905-yGoogle ScholarThere is no corresponding record for this reference.
- 8Robb, D. B.; Covey, T. R.; Bruins, A. P. Atmospheric Pressure Photoionization: An Ionization Method for Liquid Chromatography-Mass Spectrometry. Anal. Chem. 2000, 72 (15), 3653– 3659, DOI: 10.1021/ac0001636Google ScholarThere is no corresponding record for this reference.
- 9Syage, J.; Evans, M. D.; Hanold, K. A. Photoionization mass spectrometry. Am. Lab. 2000, 32, 24– 29Google ScholarThere is no corresponding record for this reference.
- 10Gaskell, S. J. Electrospray: principles and practice. J. Mass Spectrom. 1997, 32 (7), 677– 688, DOI: 10.1002/(SICI)1096-9888(199707)32:7<677::AID-JMS536>3.0.CO;2-GGoogle ScholarThere is no corresponding record for this reference.
- 11Kauppila, T. J.; Kersten, H.; Benter, T. Ionization of EPA contaminants in direct and dopant-assisted atmospheric pressure photoionization and atmospheric pressure laser ionization. J. Am. Soc. Mass Spectrom. 2015, 26 (6), 1036– 1045, DOI: 10.1007/s13361-015-1092-3Google ScholarThere is no corresponding record for this reference.
- 12Neumann, A.; Tiemann, O.; Hansen, H. J.; Rüger, C. P.; Zimmermann, R. Detailed comparison of Xenon APPI (9.6/8.4 eV), Krypton APPI (10.6/10.0 eV), APCI, and APLI (266 nm) for gas chromatography high resolution mass spectrometry of standards and complex mixtures. J. Am. Soc. Mass Spectrom. 2023, 34 (8), 1632– 1646, DOI: 10.1021/jasms.3c00085Google ScholarThere is no corresponding record for this reference.
- 13Raffaelli, A.; Saba, A. Atmospheric pressure photoionization mass spectrometry. Mass Spectrom. Rev. 2003, 22 (5), 318– 331, DOI: 10.1002/mas.10060Google ScholarThere is no corresponding record for this reference.
- 14Short, L. C.; Cai, S.-S.; Syage, J. A. APPI-MS: Effects of Mobile Phases and VUV Lamps on the Detection of PAH Compounds. J. Am. Soc. Mass Spectrom. 2007, 18 (4), 589– 599, DOI: 10.1016/j.jasms.2006.11.004Google ScholarThere is no corresponding record for this reference.
- 15Panda, S. K.; Brockmann, K. J.; Benter, T.; Schrader, W. Atmospheric pressure laser ionization (APLI) coupled with Fourier transform ion cyclotron resonance mass spectrometry applied to petroleum samples analysis: comparison with electrospray ionization and atmospheric pressure photoionization methods. Rapid Commun. Mass Spectrom. 2011, 25 (16), 2317– 2326, DOI: 10.1002/rcm.5122Google ScholarThere is no corresponding record for this reference.
- 16Kauppila, T. J.; Kuuranne, T.; Meurer, E. C.; Eberlin, M. N.; Kotiaho, T.; Kostiainen, R. Atmospheric pressure photoionization mass spectrometry. Ionization mechanism and the effect of solvent on the ionization of naphthalenes. Anal. Chem. 2002, 74 (21), 5470– 5479, DOI: 10.1021/ac025659xGoogle ScholarThere is no corresponding record for this reference.
- 17Ahmed, A.; Choi, C. H.; Choi, M. C.; Kim, S. Mechanisms Behind the Generation of Protonated Ions for Polyaromatic Hydrocarbons by Atmospheric Pressure Photoionization. Anal. Chem. 2012, 84 (2), 1146– 1151, DOI: 10.1021/ac202858kGoogle ScholarThere is no corresponding record for this reference.
- 18Klee, S.; Albrecht, S.; Derpmann, V.; Kersten, H.; Benter, T. Generation of ion-bound solvent clusters as reactant ions in dopant-assisted APPI and APLI. Anal. Bioanal. Chem. 2013, 405 (22), 6933– 6951, DOI: 10.1007/s00216-013-7114-8Google ScholarThere is no corresponding record for this reference.
- 19Robb, D. B.; Blades, M. W. Effects of solvent flow, dopant flow, and lamp current on dopant-assisted atmospheric pressure photoionization (DA-APPI) for LC-MS. Ionizationvia proton transfer. J. Am. Soc. Mass Spectrom. 2005, 16 (8), 1275– 1290, DOI: 10.1016/j.jasms.2005.03.017Google ScholarThere is no corresponding record for this reference.
- 20Kauppila, T. J.; Bruins, A. P.; Kostiainen, R. Effect of the solvent flow rate on the ionization efficiency in atmospheric pressure photoionization-mass spectrometry. J. Am. Soc. Mass Spectrom. 2005, 16 (8), 1399– 1407, DOI: 10.1016/j.jasms.2005.03.051Google ScholarThere is no corresponding record for this reference.
- 21McCulloch, R. D.; Robb, D.; Blades, M. Development of a next-generation field-free atmospheric pressure photoionization source for liquid chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2016, 30 (2), 333– 339, DOI: 10.1002/rcm.7444Google ScholarThere is no corresponding record for this reference.
- 22Kauppila, T. J.; Östman, P.; Marttila, S.; Ketola, R. A.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Atmospheric pressure photoionization-mass spectrometry with a microchip heated nebulizer. Anal. Chem. 2004, 76 (22), 6797– 6801, DOI: 10.1021/ac049058cGoogle ScholarThere is no corresponding record for this reference.
- 23Haapala, M.; Purcell, J. M.; Saarela, V.; Franssila, S.; Rodgers, R. P.; Hendrickson, C. L.; Kotiaho, T.; Marshall, A. G.; Kostiainen, R. Microchip atmospheric pressure photoionization for analysis of petroleum by Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 2009, 81 (7), 2799– 2803, DOI: 10.1021/ac802427mGoogle ScholarThere is no corresponding record for this reference.
- 24Haapala, M.; Luosujärvi, L.; Saarela, V.; Kotiaho, T.; Ketola, R. A.; Franssila, S.; Kostiainen, R. Microchip for combining gas chromatography or capillary liquid chromatography with atmospheric pressure photoionization-mass spectrometry. Anal. Chem. 2007, 79 (13), 4994– 4999, DOI: 10.1021/ac070157aGoogle ScholarThere is no corresponding record for this reference.
- 25Östman, P.; Jäntti, S.; Grigoras, K.; Saarela, V.; Ketola, R. A.; Franssila, S.; Kotiaho, T.; Kostiainen, R. Capillary liquid chromatography-microchip atmospheric pressure chemical ionization-mass spectrometry. J. Lab. on a Chip 2006, 6 (7), 948– 953, DOI: 10.1039/B601290FGoogle ScholarThere is no corresponding record for this reference.
- 26Ahonen, L. L.; Haapala, M.; Saarela, V.; Franssila, S.; Kotiaho, T.; Kostiainen, R. Feasibility of capillary liquid chromatography/microchip atmospheric pressure photoionization mass spectrometry in analyzing anabolic steroids in urine samples. Rapid Commun. Mass Spectrom. 2010, 24 (7), 958– 964, DOI: 10.1002/rcm.4468Google ScholarThere is no corresponding record for this reference.
- 27Haapala, M.; Saarela, V.; Pól, J.; Kolari, K.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Integrated liquid chromatography-heated nebulizer microchip for mass spectrometry. Anal. Chim. Acta 2010, 662 (2), 163– 169, DOI: 10.1016/j.aca.2010.01.005Google ScholarThere is no corresponding record for this reference.
- 28Kruve, A.; Haapala, M.; Saarela, V.; Franssila, S.; Kostiainen, R.; Kotiaho, T.; Ketola, R. A. Feasibility of capillary liquid chromatography-microchip-atmospheric pressure photoionization-mass spectrometry for pesticide analysis in tomato. Anal. Chim. Acta 2011, 696 (1–2), 77– 83, DOI: 10.1016/j.aca.2011.04.006Google ScholarThere is no corresponding record for this reference.
- 29Vrkoslav, V.; Rumlová, B.; Strmeň, T.; Nekvasilová, P.; Šulc, M.; Cvačka, J. Applicability of low-flow atmospheric pressure chemical ionization and photoionization mass spectrometry with a microfabricated nebulizer for neutral lipids. Rapid Commun. Mass Spectrom. 2018, 32 (8), 639– 648, DOI: 10.1002/rcm.8086Google ScholarThere is no corresponding record for this reference.
- 30Saarela, V.; Haapala, M.; Kostiainen, R.; Kotiaho, T.; Franssila, S. Glass microfabricated nebulizer chip for mass spectrometry. J. Lab. on a Chip 2007, 7 (5), 644– 646, DOI: 10.1039/b700101kGoogle ScholarThere is no corresponding record for this reference.
- 31Östman, P.; Luosujärvi, L.; Haapala, M.; Grigoras, K.; Ketola, R. A.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Gas chromatography-microchip atmospheric pressure chemical ionization-mass spectrometry. Anal. Chem. 2006, 78 (9), 3027– 3031, DOI: 10.1021/ac052260aGoogle ScholarThere is no corresponding record for this reference.
- 32Tubaro, M.; Marotta, E.; Seraglia, R.; Traldi, P. Atmospheric pressure photoionization mechanisms. 2. The case of benzene and toluene. Rapid Commun. Mass Spectrom. 2003, 17 (21), 2423– 2429, DOI: 10.1002/rcm.1208Google ScholarThere is no corresponding record for this reference.
- 33Takahata, Y.; Vendrame, R. Ionization energies, electron affinities and excitation energies of some steroid hormones calculated with the semiempirical HAM/3 method. J. Braz. Chem. Soc. 2001, 12, 165– 172, DOI: 10.1590/S0103-50532001000200007Google ScholarThere is no corresponding record for this reference.
- 34Zayed, M. A.; Hawash, M.; Fahmey, M.; El-Gizouli, A. M. Investigation of ibuprofen drug using mass spectrometry, thermal analyses, and semi-empirical molecular orbital calculation. J. Therm. Anal. Calorim. 2012, 108 (1), 315– 322, DOI: 10.1007/s10973-011-1876-zGoogle ScholarThere is no corresponding record for this reference.
- 35Ostrowski, W.; Karczewska, K.; Frański, R. Oxidation of paracetamol by Cu2+-formation of the paracetamol radical cation. Rapid Commun. Mass Spectrom. 2013, 27 (13), 1579– 1584, DOI: 10.1002/rcm.6583Google ScholarThere is no corresponding record for this reference.
- 36Slifkin, M.; Allison, A. Measurement of ionization potentials from contact charge transfer spectra. Nature 1967, 215 (5104), 949– 950, DOI: 10.1038/215949a0Google ScholarThere is no corresponding record for this reference.
- 37Gross, D. S.; Williams, E. R. Structure of gramicidin S (M+ H+ X) 2+ ions (X= Li, Na, K) probed by proton transfer reactions. J. Am. Chem. Soc. 1996, 118 (1), 202– 204, DOI: 10.1021/ja952426xGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
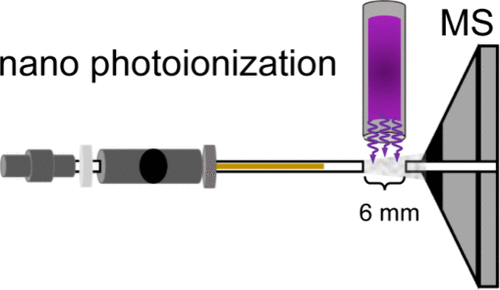
Figure 1

Figure 1. Photo of the nAPPI source coupled to a TSQ triple quadrupole MS (left) and schematics (right).
Figure 2
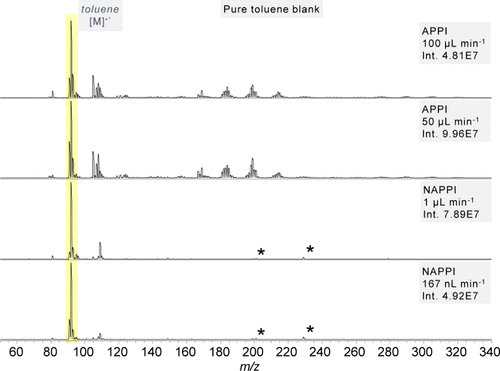
Figure 2. Pure toluene blanks for APPI and nAPPI at different flow rates. *Plasticizer impurities.
Figure 3

Figure 3. Measurements of phenanthrene in toluene at different concentrations with APPI at a flow rate of 50 μL min–1 and nAPPI at a flow rate of only 1 μL min–1. All spectra were measured on a TSQ triple quadrupole MS. *Plasticizer impurities.
Figure 4
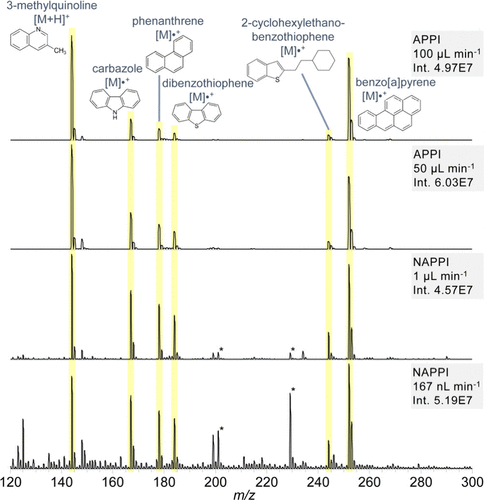
Figure 4. PAH/PAXH, 10 mg L–1 each in toluene, measured with APPI and nAPPI at different flow rates (measured on a TSQ triple quadrupole MS). *Plasticizer impurities.
Figure 5

Figure 5. S/N calculations for nAPPI (flow rate of 1 μL min–1) and conventional APPI (flow rate of 50 μL min–1).
Figure 6

Figure 6. Mass traces of loop injections of phenanthrene in toluene at low concentrations for nAPPI and APPI.
Figure 7

Figure 7. Spectra of different photoionizable analytes spanning a broader polarity range measured with APPI and nAPPI on a TSQ triple quadrupole MS. β-Estradiol and paracetamol were dissolved from neat compounds, and ibuprofen was measured from a pill. *Plasticizer impurities.
Figure 8

Figure 8. Two larger photoionizable analytes, reserpine (left) and gramicidin S (right) measured with APPI and nAPPI.
Figure 9

Figure 9. On the left: A complex sample of a heavy crude oil in toluene measured with conventional APPI and nAPPI. On the right: Assigned compositions of the heavy crude oil. While less oxygen-containing compounds are observed, there is an increased tendency for the formation of radical cations in nAPPI than in APPI.
Figure 10
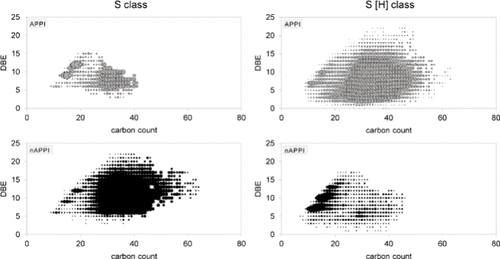
Figure 10. Kendrick plots of the S and S [H] class of the heavy crude for APPI and nAPPI.
References
This article references 37 other publications.
- 1Gaspar, A.; Zellermann, E.; Lababidi, S.; Reece, J.; Schrader, W. Impact of different ionization methods on the molecular assignments of asphaltenes by FT-ICR mass spectrometry. Anal. Chem. 2012, 84 (12), 5257– 5267, DOI: 10.1021/ac300133pThere is no corresponding record for this reference.
- 2Cai, S.-S.; Syage, J. A. Comparison of atmospheric pressure photoionization, atmospheric pressure chemical ionization, and electrospray ionization mass spectrometry for analysis of lipids. Anal. Chem. 2006, 78 (4), 1191– 1199, DOI: 10.1021/ac0515834There is no corresponding record for this reference.
- 3Huba, A. K.; Huba, K.; Gardinali, P. R. Understanding the atmospheric pressure ionization of petroleum components: The effects of size, structure, and presence of heteroatoms. Sci. Total Environ. 2016, 568, 1018– 1025, DOI: 10.1016/j.scitotenv.2016.06.044There is no corresponding record for this reference.
- 4Kauppila, T.; Nikkola, T.; Ketola, R. A.; Kostiainen, R. Atmospheric pressure photoionization-mass spectrometry and atmospheric pressure chemical ionization-mass spectrometry of neurotransmitters. J. Mass Spectrom. 2006, 41 (6), 781– 789, DOI: 10.1002/jms.1034There is no corresponding record for this reference.
- 5Gowda, G. N.; Djukovic, D. Overview of mass spectrometry-based metabolomics: opportunities and challenges. Methods Mol. Biol. (Clifton, N.J.) 1198 2014, 1198, 3– 12, DOI: 10.1007/978-1-4939-1258-2_1There is no corresponding record for this reference.
- 6Nadler, W. M.; Waidelich, D.; Kerner, A.; Hanke, S.; Berg, R.; Trumpp, A.; Rösli, C. MALDI versus ESI: the impact of the ion source on peptide identification. J. Proteome Res. 2017, 16 (3), 1207– 1215, DOI: 10.1021/acs.jproteome.6b00805There is no corresponding record for this reference.
- 7Jia, S.; Marques Dos Santos, M.; Li, C.; Snyder, S. A. Recent advances in mass spectrometry analytical techniques for per-and polyfluoroalkyl substances (PFAS). Anal. Bioanal. Chem. 2022, 414 (9), 2795– 2807, DOI: 10.1007/s00216-022-03905-yThere is no corresponding record for this reference.
- 8Robb, D. B.; Covey, T. R.; Bruins, A. P. Atmospheric Pressure Photoionization: An Ionization Method for Liquid Chromatography-Mass Spectrometry. Anal. Chem. 2000, 72 (15), 3653– 3659, DOI: 10.1021/ac0001636There is no corresponding record for this reference.
- 9Syage, J.; Evans, M. D.; Hanold, K. A. Photoionization mass spectrometry. Am. Lab. 2000, 32, 24– 29There is no corresponding record for this reference.
- 10Gaskell, S. J. Electrospray: principles and practice. J. Mass Spectrom. 1997, 32 (7), 677– 688, DOI: 10.1002/(SICI)1096-9888(199707)32:7<677::AID-JMS536>3.0.CO;2-GThere is no corresponding record for this reference.
- 11Kauppila, T. J.; Kersten, H.; Benter, T. Ionization of EPA contaminants in direct and dopant-assisted atmospheric pressure photoionization and atmospheric pressure laser ionization. J. Am. Soc. Mass Spectrom. 2015, 26 (6), 1036– 1045, DOI: 10.1007/s13361-015-1092-3There is no corresponding record for this reference.
- 12Neumann, A.; Tiemann, O.; Hansen, H. J.; Rüger, C. P.; Zimmermann, R. Detailed comparison of Xenon APPI (9.6/8.4 eV), Krypton APPI (10.6/10.0 eV), APCI, and APLI (266 nm) for gas chromatography high resolution mass spectrometry of standards and complex mixtures. J. Am. Soc. Mass Spectrom. 2023, 34 (8), 1632– 1646, DOI: 10.1021/jasms.3c00085There is no corresponding record for this reference.
- 13Raffaelli, A.; Saba, A. Atmospheric pressure photoionization mass spectrometry. Mass Spectrom. Rev. 2003, 22 (5), 318– 331, DOI: 10.1002/mas.10060There is no corresponding record for this reference.
- 14Short, L. C.; Cai, S.-S.; Syage, J. A. APPI-MS: Effects of Mobile Phases and VUV Lamps on the Detection of PAH Compounds. J. Am. Soc. Mass Spectrom. 2007, 18 (4), 589– 599, DOI: 10.1016/j.jasms.2006.11.004There is no corresponding record for this reference.
- 15Panda, S. K.; Brockmann, K. J.; Benter, T.; Schrader, W. Atmospheric pressure laser ionization (APLI) coupled with Fourier transform ion cyclotron resonance mass spectrometry applied to petroleum samples analysis: comparison with electrospray ionization and atmospheric pressure photoionization methods. Rapid Commun. Mass Spectrom. 2011, 25 (16), 2317– 2326, DOI: 10.1002/rcm.5122There is no corresponding record for this reference.
- 16Kauppila, T. J.; Kuuranne, T.; Meurer, E. C.; Eberlin, M. N.; Kotiaho, T.; Kostiainen, R. Atmospheric pressure photoionization mass spectrometry. Ionization mechanism and the effect of solvent on the ionization of naphthalenes. Anal. Chem. 2002, 74 (21), 5470– 5479, DOI: 10.1021/ac025659xThere is no corresponding record for this reference.
- 17Ahmed, A.; Choi, C. H.; Choi, M. C.; Kim, S. Mechanisms Behind the Generation of Protonated Ions for Polyaromatic Hydrocarbons by Atmospheric Pressure Photoionization. Anal. Chem. 2012, 84 (2), 1146– 1151, DOI: 10.1021/ac202858kThere is no corresponding record for this reference.
- 18Klee, S.; Albrecht, S.; Derpmann, V.; Kersten, H.; Benter, T. Generation of ion-bound solvent clusters as reactant ions in dopant-assisted APPI and APLI. Anal. Bioanal. Chem. 2013, 405 (22), 6933– 6951, DOI: 10.1007/s00216-013-7114-8There is no corresponding record for this reference.
- 19Robb, D. B.; Blades, M. W. Effects of solvent flow, dopant flow, and lamp current on dopant-assisted atmospheric pressure photoionization (DA-APPI) for LC-MS. Ionizationvia proton transfer. J. Am. Soc. Mass Spectrom. 2005, 16 (8), 1275– 1290, DOI: 10.1016/j.jasms.2005.03.017There is no corresponding record for this reference.
- 20Kauppila, T. J.; Bruins, A. P.; Kostiainen, R. Effect of the solvent flow rate on the ionization efficiency in atmospheric pressure photoionization-mass spectrometry. J. Am. Soc. Mass Spectrom. 2005, 16 (8), 1399– 1407, DOI: 10.1016/j.jasms.2005.03.051There is no corresponding record for this reference.
- 21McCulloch, R. D.; Robb, D.; Blades, M. Development of a next-generation field-free atmospheric pressure photoionization source for liquid chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2016, 30 (2), 333– 339, DOI: 10.1002/rcm.7444There is no corresponding record for this reference.
- 22Kauppila, T. J.; Östman, P.; Marttila, S.; Ketola, R. A.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Atmospheric pressure photoionization-mass spectrometry with a microchip heated nebulizer. Anal. Chem. 2004, 76 (22), 6797– 6801, DOI: 10.1021/ac049058cThere is no corresponding record for this reference.
- 23Haapala, M.; Purcell, J. M.; Saarela, V.; Franssila, S.; Rodgers, R. P.; Hendrickson, C. L.; Kotiaho, T.; Marshall, A. G.; Kostiainen, R. Microchip atmospheric pressure photoionization for analysis of petroleum by Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 2009, 81 (7), 2799– 2803, DOI: 10.1021/ac802427mThere is no corresponding record for this reference.
- 24Haapala, M.; Luosujärvi, L.; Saarela, V.; Kotiaho, T.; Ketola, R. A.; Franssila, S.; Kostiainen, R. Microchip for combining gas chromatography or capillary liquid chromatography with atmospheric pressure photoionization-mass spectrometry. Anal. Chem. 2007, 79 (13), 4994– 4999, DOI: 10.1021/ac070157aThere is no corresponding record for this reference.
- 25Östman, P.; Jäntti, S.; Grigoras, K.; Saarela, V.; Ketola, R. A.; Franssila, S.; Kotiaho, T.; Kostiainen, R. Capillary liquid chromatography-microchip atmospheric pressure chemical ionization-mass spectrometry. J. Lab. on a Chip 2006, 6 (7), 948– 953, DOI: 10.1039/B601290FThere is no corresponding record for this reference.
- 26Ahonen, L. L.; Haapala, M.; Saarela, V.; Franssila, S.; Kotiaho, T.; Kostiainen, R. Feasibility of capillary liquid chromatography/microchip atmospheric pressure photoionization mass spectrometry in analyzing anabolic steroids in urine samples. Rapid Commun. Mass Spectrom. 2010, 24 (7), 958– 964, DOI: 10.1002/rcm.4468There is no corresponding record for this reference.
- 27Haapala, M.; Saarela, V.; Pól, J.; Kolari, K.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Integrated liquid chromatography-heated nebulizer microchip for mass spectrometry. Anal. Chim. Acta 2010, 662 (2), 163– 169, DOI: 10.1016/j.aca.2010.01.005There is no corresponding record for this reference.
- 28Kruve, A.; Haapala, M.; Saarela, V.; Franssila, S.; Kostiainen, R.; Kotiaho, T.; Ketola, R. A. Feasibility of capillary liquid chromatography-microchip-atmospheric pressure photoionization-mass spectrometry for pesticide analysis in tomato. Anal. Chim. Acta 2011, 696 (1–2), 77– 83, DOI: 10.1016/j.aca.2011.04.006There is no corresponding record for this reference.
- 29Vrkoslav, V.; Rumlová, B.; Strmeň, T.; Nekvasilová, P.; Šulc, M.; Cvačka, J. Applicability of low-flow atmospheric pressure chemical ionization and photoionization mass spectrometry with a microfabricated nebulizer for neutral lipids. Rapid Commun. Mass Spectrom. 2018, 32 (8), 639– 648, DOI: 10.1002/rcm.8086There is no corresponding record for this reference.
- 30Saarela, V.; Haapala, M.; Kostiainen, R.; Kotiaho, T.; Franssila, S. Glass microfabricated nebulizer chip for mass spectrometry. J. Lab. on a Chip 2007, 7 (5), 644– 646, DOI: 10.1039/b700101kThere is no corresponding record for this reference.
- 31Östman, P.; Luosujärvi, L.; Haapala, M.; Grigoras, K.; Ketola, R. A.; Kotiaho, T.; Franssila, S.; Kostiainen, R. Gas chromatography-microchip atmospheric pressure chemical ionization-mass spectrometry. Anal. Chem. 2006, 78 (9), 3027– 3031, DOI: 10.1021/ac052260aThere is no corresponding record for this reference.
- 32Tubaro, M.; Marotta, E.; Seraglia, R.; Traldi, P. Atmospheric pressure photoionization mechanisms. 2. The case of benzene and toluene. Rapid Commun. Mass Spectrom. 2003, 17 (21), 2423– 2429, DOI: 10.1002/rcm.1208There is no corresponding record for this reference.
- 33Takahata, Y.; Vendrame, R. Ionization energies, electron affinities and excitation energies of some steroid hormones calculated with the semiempirical HAM/3 method. J. Braz. Chem. Soc. 2001, 12, 165– 172, DOI: 10.1590/S0103-50532001000200007There is no corresponding record for this reference.
- 34Zayed, M. A.; Hawash, M.; Fahmey, M.; El-Gizouli, A. M. Investigation of ibuprofen drug using mass spectrometry, thermal analyses, and semi-empirical molecular orbital calculation. J. Therm. Anal. Calorim. 2012, 108 (1), 315– 322, DOI: 10.1007/s10973-011-1876-zThere is no corresponding record for this reference.
- 35Ostrowski, W.; Karczewska, K.; Frański, R. Oxidation of paracetamol by Cu2+-formation of the paracetamol radical cation. Rapid Commun. Mass Spectrom. 2013, 27 (13), 1579– 1584, DOI: 10.1002/rcm.6583There is no corresponding record for this reference.
- 36Slifkin, M.; Allison, A. Measurement of ionization potentials from contact charge transfer spectra. Nature 1967, 215 (5104), 949– 950, DOI: 10.1038/215949a0There is no corresponding record for this reference.
- 37Gross, D. S.; Williams, E. R. Structure of gramicidin S (M+ H+ X) 2+ ions (X= Li, Na, K) probed by proton transfer reactions. J. Am. Chem. Soc. 1996, 118 (1), 202– 204, DOI: 10.1021/ja952426xThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.analchem.5c06912.
List of reference compounds and additional graphs highlighting the characteristics of the source (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



