
Toxicity, Chemistry, and Public Health Relevance of Emerging Nicotine Analog Vapes, Pods, and PouchesClick to copy article linkArticle link copied!
- Rhea RaghuRhea RaghuDepartment of Environmental Medicine, University of Rochester Medical Center, Rochester, New York 14624, United StatesMore by Rhea Raghu
- Mohana SenguptaMohana SenguptaDepartment of Chemistry, Portland State University, Portland, Oregon 97201, United StatesMore by Mohana Sengupta
- Karen LinKaren LinDepartment of Environmental Medicine, University of Rochester Medical Center, Rochester, New York 14624, United StatesMore by Karen Lin
- Felix EffahFelix EffahDepartment of Environmental Medicine, University of Rochester Medical Center, Rochester, New York 14624, United StatesMore by Felix Effah
- Robert M. StronginRobert M. StronginDepartment of Chemistry, Portland State University, Portland, Oregon 97201, United StatesMore by Robert M. Strongin
- Irfan Rahman*Irfan Rahman*Email: [email protected]. Tel: 1 585 275 6911.Department of Environmental Medicine, University of Rochester Medical Center, Rochester, New York 14624, United StatesMore by Irfan Rahman
Abstract
Electronic nicotine delivery system manufacturers, such as Charlie’s Holdings Inc., ECBlend, Outlaw, and NicRiver, have recently introduced nicotine analogs, such as 6-methyl nicotine, 6-MN (“Metatine”), and nicotinamide, NA (“Nixamide,” “Nixodine,” or “Nixotin-free base and salt”) in products to circumvent the U.S. FDA’s premarket tobacco product application (PMTA) requirements. Marketed as “tobacco-free,” “PMTA-exempt,” or “FDA-approved,” these compounds now appear in oral nicotine pouches and disposable bars/vapes from brands such as Outlaw Dip, Kumi-Six, SBX, Katchmi, and Spree Bar under proprietary labels including “NoNic6,” “Metatine,” or “NIC-SAFE.” These products often mimic the appeal of conventional nicotine delivery systems, with extensive use of fruit, menthol, and candy-inspired flavorings. Independent testing, however, has revealed inconsistencies between labeled and actual concentrations of 6-MN, alongside the presence of undisclosed chemical additives such as “coolants” and numerous other examples. Additionally, emerging toxicological evidence indicates that 6-MN is more potent than nicotine to cause oxidative, inflammatory, and toxic responses. This includes the activation of NF−κΒ, causes epithelial permeability, and lung remodeling due to extracellular matrix (ECM) modifications. Misleading claims by industry sources include erroneous statements that imply nicotinamide interacts with nicotinic acetylcholine receptors (e.g., methylated nicotine analogs-interaction with α4β2 nicotinic acetylcholine receptors -nAChR interactions). Despite health risks, regulatory frameworks remain ambiguous, enabling companies to circumvent oversight by exploiting loopholes around synthetic analogs. There is a need for rigorous chemical and toxicological studies to evaluate the health effects of nicotine analogs, such as 6-MN and NA, and their interactions with flavorings. This review summarizes current knowledge of chemistry, pharmacology, toxicity, product landscape, flavoring profiles, and labeling practices of 6-MN- and nicotinamide-containing and nicotinamide products, highlighting the urgent need for regulatory clarity, transparent labeling, and further chemico-toxicological assessment.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Introduction
Chemical Structure and Thermal Degradation of 6-Methyl Nicotine and Nicotinamide
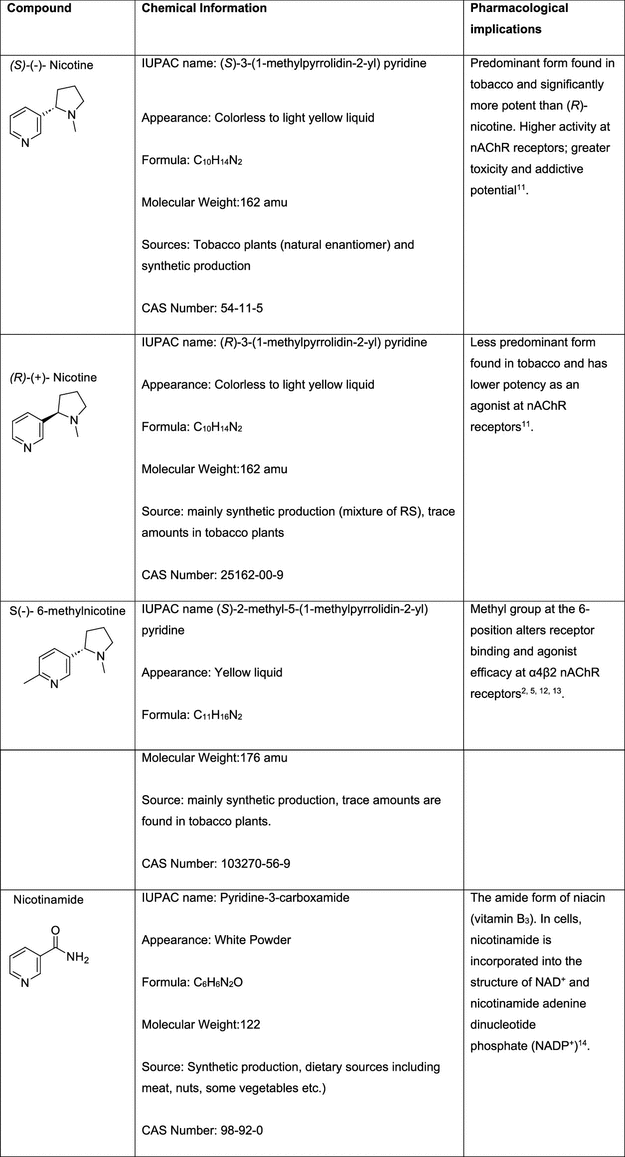
Thermal Degradation Pathways of Nicotine, 6-MN and NA
Figure 1
Figure 1. Different mechanistic pathways of nicotine thermal degradation at high (cigarette) and low (e.g., e-cigarette) temperatures. Left: pyrolysis products. Right: Hydroxyl radical hydrogen-abstraction intermediates Figure 1a–c. The left figure is adapted from Asensio et al. (16) The right figure is adapted from Hoa et al. (17)
Secondary Reactions and Radical-Mediated Chemical Pathways in E-Cigarettes
Potential Thermal Degradation Pathway of 6-Methylnicotine (6-MN)
Figure 2
Figure 2. Proposed radical intermediates from the preferred hydrogen atom abstraction sites of 6-MN at vaping-relevant temperatures.
Thermal Degradation Pathways of Nicotinamide
Figure 3
Figure 3. Temperature-dependent formation of 3-CP from nicotinamide. The 3-CP yield increases with temperature, showing progressive growth from 250 to 335 °C. Reprinted with permission from ref (20). Copyright 2025 by the American Chemical Society.
Figure 4
Figure 4. LC10 dose response (DR) curves and chemical interaction model of 3-cyanopyridine (3CP) and nicotinamide (NA) tested on BEAS-2B cells. (A) DR of 3CP graphed on the log-transformed x-axis of concentrations tested up to 1000 ppm. (B) DR of NA tested at concentrations up to 10,000 ppm. (C) The combined DR curves of 3CP and NA are plotted on the same concentration scale. The dose–response curves are identical to those shown in panels A and C, but on a comparable scale. The 3CP curve above 1000 ppm is a model prediction based on the curve produced at lower concentrations, rather than on tested values. (D) Isobologram of 3CP:NA, indicating synergistic interaction. Reprinted with permission from ref (20). Copyright 2025 by the American Chemical Society.
Solvent and Flavorant Chemistry in E-Liquid Aerosol Formation
Figure 5
Figure 5. Selected relatively common chemical components of e-liquids addition to nicotine and varying effects on the formation of carbonyl toxicants. (28).
Figure 6
Figure 6. Chemical structures of coolants WS-23 and WS-3.
Influence of Nicotine Analogs and Their Salts on Flavorant Acetal Formation in E-Liquids
Figure 7
Figure 7. Effects on acetal formation in e-liquids in the presence of nicotine in free-base versus salt forms. Acetals form in e-liquids when aldehyde flavorants, such as benzaldehyde, vanillin, and trans-cinnamaldehyde. (33)
Toxicology and Pharmacology of 6-MN and NA
6-MN and Nixamide/Nixodine/Nicotinamide Commercial Availability
Figure 8
Figure 8. Series of pie-charts displaying the distribution of flavor categories for nicotine-analog containing products distributed by (A) Spree Bar, (B) Kumi-Six, (C) Katchmi, (D) SBX, and (E) Outlaw Dip.
| flavoring | brand | category |
|---|---|---|
| Strawberry Fanta | Kumi-Six | fruit |
| Blue Razz Ice | Kumi-Six | fruit-ice |
| Watermelon Ice | Kumi-Six | fruit-ice |
| Banana Ice | Kumi-Six | fruit-ice |
| Peach Ice | Kumi-Six | fruit-ice |
| Cool Mint | Kumi-Six | menthol |
| Sour Blue Gummy | Kumi-Six | candy |
| Tobacco | Kumi-Six | tobacco |
| Strawberry Watermelon | Kumi-Six | fruit |
| Triple Berry Ice | Kumi-Six | fruit-ice |
| Blue Razz | Kumi-Six | fruit |
| Menthol | Kumi-Six | menthol |
| Blue Kiwi Ice | Kumi-Six - Kurve | fruit-ice |
| B-Pop | Kumi-Six - Kurve | unknown/not listed |
| Pineapple Peach | Kumi-Six - Kurve | fruit |
| Baja Blue | Kumi-Six - Kurve | unknown/not listed |
| Sour Gush | Kumi-Six - Kurve | candy |
| Grape Rancher | Kumi-Six - Kurve | fruit |
| Strawberry Cake | Kumi-Six - Kurve | dessert |
| Sour Watermelon Gummy | Kumi-Six - Kurve | candy |
| Strawberry Watermelon | Kumi-Six - Scenic | fruit |
| Buns | Kumi-Six - Scenic | dessert |
| Grape Rancher | Kumi-Six - Scenic | fruit |
| Loopy Jammy Blue | Kumi-Six - Scenic | candy |
| Sour Rocket Pop | Kumi-Six - Scenic | candy |
| Cool Mint | Kumi-Six - Scenic | menthol |
| Baja Blue | Kumi-Six - Scenic | unknown/not listed |
| Vanilla Cola | Kumi-Six - Scenic | beverage |
| Sour Blue Gummy | Kumi-Six - Scenic | candy |
| Rainbow Fruit | Spree Bar | fruit |
| Blue Razz Ice | Spree Bar | fruit-ice |
| Strawberry Apple Melon | Spree Bar | fruit |
| Blood Orange Peach | Spree Bar | fruit |
| Watermelon Grapefruit | Spree Bar | fruit |
| Strawberry Mango | Spree Bar | fruit |
| Creamy Melon | Spree Bar | fruit |
| Sweet Spearmint | Spree Bar | menthol |
| Pineapple Coconut | Spree Bar | fruit |
| White Gummy Dream | Katchmi | candy |
| Acid Tears | Katchmi | candy |
| Sour Blue Cherry | Katchmi | candy |
| Miami Mint | Katchmi | menthol |
| Strazz Watermelon | Katchmi | fruit |
| Arizona Blast | Katchmi | beverage |
| Blue Razz Ice | Katchmi | fruit-ice |
| Watermelon Ice | Katchmi | fruit-ice |
| Blueberry Burst | SBX | fruit |
| Blue Razz Ice | SBX | fruit-ice |
| Clear | SBX | unknown/not listed |
| Confetti | SBX | unknown/not listed |
| Georgia Peach | SBX | fruit |
| Grape Ice | SBX | fruit-ice |
| Lemon Apple Ice | SBX | fruit-ice |
| Miami Mint | SBX | menthol |
| Mystic Ice | SBX | unknown-ice |
| Ripe Apple | SBX | fruit |
| Strawberry Kiwi | SBX | fruit |
| Sweet Spearmint | SBX | menthol |
| Summer Strawberry | SBX | fruit |
| Virginia Tobacco | SBX | tobacco |
| Wintergreen | Outlaw Dip - Fat Cut | menthol |
| Dark Wintergreen | Outlaw Dip - Fat Cut | menthol |
| Southern Sweet Tea | Outlaw Dip - Fat Cut | beverage |
| Killer Vaniller | Outlaw Dip - Fat Cut | candy |
| Ramblin’ Root Beer | Outlaw Dip - Fat Cut | beverage |
| Georgia Peach | Outlaw Dip - Fat Cut | fruit |
| Backwoods Blueberry | Outlaw Dip - Fat Cut | fruit |
| Wild Watermelon | Outlaw Dip - Fat Cut | fruit |
| Orange Creamsicle | Outlaw Dip - Fat Cut | fruit |
| Straight | Outlaw Dip - Fat Cut | tobacco |
| Gold | Outlaw Dip - Fat Cut | tobacco |
| Original | Outlaw Dip - Fat Cut | tobacco |
| Mint | Outlaw Dip - Fat Cut | menthol |
| Winterberry | Outlaw Dip - Pouches | fruit |
| Wintergreen | Outlaw Dip - Pouches | menthol |
| Apple Danish | Outlaw Dip - Pouches | dessert |
| Kraken Kola | Outlaw Dip - Pouches | beverage |
| Melon Ice | Outlaw Dip - Pouches | fruit |
| Red Deer | Outlaw Dip - Pouches | beverage |
| Sweden’s Fish | Outlaw Dip - Pouches | candy |
| Eagles Blood | Outlaw Dip - Pouches | beverage |
| Mead & Honey | Outlaw Dip - Pouches | beverage |
| Vanilla | Outlaw Dip - Pouches | candy |
| Mint | Outlaw Dip - Pouches | menthol |
Emerging Nicotine Product Content and Labeling Disclosures
Public Health Implications: Second-Hand and Third-Hand Exposures: Implications on Children and Adolescents
Regulation of Emerging Nicotine Analog Products
Conclusion
Biographies
Rhea Raghu
Rhea Raghu is an undergraduate student at the University of Rochester majoring in Health Policy and is a member of Rochester’s REMS BS/MD program. She intends to pursue a career as a physician, with a particular interest in the intersection of clinical practice and healthcare policy. She conducts research in Dr. Irfan Rahman’s laboratory, where her work focuses on toxicology, mechanisms of inflammation, and the regulatory implications of environmental exposures. Her academic interests center on translating scientific research into evidence-based policy.
Mohana Sengupta
Mohana Sengupta is a doctoral student in Professor Robert Strongin’s lab at Portland State University. She is investigating the levels, identities and origins of toxicant emissions from electronic cigarettes. Her work is part of broader collaborative efforts to inform the understanding of the health effects of vaping. Mohana has synthesized valuable chemical standards, has designed chemical sensors for specific aerosol toxicants and discovered unique aerosol chemical products of nicotine, cannabinoids and flavorants. She is continuously developing sample processing and analytical methods to more effectively characterize highly complex e-cigarette aerosols.
Karen Lin
Karen Lin is an undergraduate student at the University of Rochester majoring in microbiology and minoring in psychology. She is currently a research assistant in Dr. Irfan Rahman’s Laboratory in the Department of Environmental Medicine. Her research interests include toxicology, oxidative stress, and inflammation. Karen aspires to pursue medical school and graduate studies in immunology.
Felix Effah
Felix Effah, Ph.D., is a toxicologist, conducting research on the effects of e-cigarette vapor on pulmonary and neurochemical parameters and organ toxicity. Dr Effah has won several awards from the British Pharmacological Society, British toxicology society and UK Health Agency. Dr. Effah has contributed to the UK Health Security Agency as a research scientist, where he explored toxicological assessments of e-cigarette flavors using 3D lung models. Currently, he serves as a Postdoctoral Fellow at the University of Rochester Medical Center, specializing in toxicology, exposure science, inhalation toxicology, and 3D lung modeling.
Robert M. Strongin
Robert M. Strongin, Ph.D., is a Professor of Organic Chemistry at Portland State University. He has studied the chemistry of vaping since 2013. He has been interested in determining how flavorings, including terpenes, as well as other vape constituents, react and degrade when heated and aerosolized. Dr. Strongin runs an internationally recognized research program, is a founder of two biotech startup companies and is known for his contributions to STEM education. In addition to tobacco control and cannabis science, he is an expert in the field of biosensors and targeted molecular probes for imaging cancer tissue.
Irfan Rahman
Irfan Rahman, Ph.D., is a Dean’s Professor of Environmental Medicine, Medicine (Pulmonary), and Public Health Sciences at the University of Rochester Medical Center, NY and Director of Flavoring Inhalation Toxicology Center. His research interests include oxidative stress, inflammation, molecular clock, mitochondrial dysfunction, epigenetics, and cellular senescence by tobacco smoke/tobacco products (cigarette smoke, e-cigarettes, waterpipe/hookah, and cigars). Dr. Rahman is an author of over three hundred (300) publications in peer-reviewed journals with an ‘H impact factor of 123. He is the author/editor of a book on “Inflammation, Aging, Diet and Nutrition”, and awarded as Highly Cited Researchers by Thomson Reuters.
| 6-MN | 6-methyl nicotine |
| NA | nicotinamide |
| ENDS | electronic nicotine delivery system |
| e-cig | electronic cigarettes |
| CCR2 | CC chemokine receptor 2 |
| CCL2 | CC chemokine ligand 2 |
| CCR5 | CC chemokine receptor 5 |
| TLC | thin layer chromatography |
| PMTA | premarket tobacco product application |
| COPD | chronic obstructive pulmonary disease |
| FDA | Food and Drug Administration |
| TCA | Tobacco Control Act |
| GC/MS | gas chromatography and mass spectrometry |
| HRLC/MS/MS | high-resolution liquid chromatography coupled to tandem mass spectrometry |
| ROS | reactive oxygen species |
| nAChRs | α4β2 nicotinic acetylcholine receptors |
| 3-CP | 3-cyanopyridine |
| HCN | hydrogen cyanide |
| NH3 | ammonia |
| PG | propylene glycol |
| GL | glycerol |
| SFN | substitute for nicotine |
| ALI | air–liquid interface |
| OSHA | Occupational Safety and Health Administration |
| WS-23 | 2 isopropyl N,2,3 trimethylbutanamide |
| WS-3 | N-ethyl-2-isopropyl-5-methylcyclohexanecarboxamide |
| TRPM8 | transient receptor potential melastatin member 8 |
| IL-1ß | interleukin-1 beta |
| IL-10 | interleukin-10 |
| CYCL1 | C-Y-C motif chemokine ligand 1 |
| CXCL2 | C-X-C motif chemokine ligand 2 |
| CXCL10 | C-X-C motif chemokine ligand 10 |
| NK | natural killer |
| GRAS | generally recognized as safe |
| TFN | tobacco-free nicotine |
| ECM | extracellular matrix |
References
This article references 68 other publications.
- 1Vanhee, C.; Dill, M.; Canfyn, M.; Tuenter, E.; Barhdadi, S. The Emergence of a Novel Synthetic Nicotine Analog 6-Methyl Nicotine (6-MN) in Proclaimed Tobacco- and Nicotine-Free Pouches Available in Europe. Drug Test. Anal. 2025, 17 (8), 1368– 1379, DOI: 10.1002/dta.3841Google ScholarThere is no corresponding record for this reference.
- 2Effah, F.; Sun, Y.; Lin, K.; Rahman, I. A Comparative Toxicological Evaluation of Emerging Nicotine Analogs 6-Methyl Nicotine and Nicotinamide: A Scoping Review. Arch. Toxicol. 2025, 99 (4), 1333– 1340, DOI: 10.1007/s00204-025-03960-1Google ScholarThere is no corresponding record for this reference.
- 3Jabba, S. V.; Jordt, S. E. Marketing of Nicotinamide as Nicotine Replacement in Electronic Cigarettes and Smokeless Tobacco. Tob. Prev. Cessat. 2024, 10 (August), 1– 5, DOI: 10.18332/tpc/187767Google ScholarThere is no corresponding record for this reference.
- 4Pankow, J. F.; Luo, W.; McWhirter, K. J.; Sengupta, M.; Strongin, R. M. Levels of the Nicotine Analog 6-Methyl Nicotine as a Naturally Formed Tobacco Alkaloid in Tobacco and Tobacco Products. Sci. Rep. 2025, 15 (1), 17945 DOI: 10.1038/s41598-025-01392-6Google ScholarThere is no corresponding record for this reference.
- 5Jordt, S. E.; Jabba, S. V. Introduction of Nicotine Analogue-Containing Oral Pouch Products in the United States. Tob. Prev. Cessat. 2024, 10, 61 DOI: 10.18332/tpc/195621Google ScholarThere is no corresponding record for this reference.
- 6Effah, F.; Sun, Y.; Friedman, A.; Rahman, I. Emerging Nicotine Analog 6-Methyl Nicotine Increases Reactive Oxygen Species in Aerosols and Cytotoxicity in Human Bronchial Epithelial Cells. Toxicol. Lett. 2025, 405, 9– 15, DOI: 10.1016/j.toxlet.2025.01.007Google ScholarThere is no corresponding record for this reference.
- 7Qi, H.; Chang, X.; Wang, K.; Xu, Q.; Liu, M.; Han, B. Comparative Analyses of Transcriptome Sequencing and Carcinogenic Exposure Toxicity of Nicotine and 6-Methyl Nicotine in Human Bronchial Epithelial Cells. Toxicol. In Vitro 2023, 93, 105661 DOI: 10.1016/j.tiv.2023.105661Google ScholarThere is no corresponding record for this reference.
- 8Effah, F.; Shaikh, S. B.; Chalupa, D.; Faizan, M. I.; Elder, A.; Rahman, I. High-Resistance Coils in E-Cigarettes Increase Heavy Metals Leaching into Aerosols to Cause Oxidants Generation in Human Bronchial Epithelial Cells at Air-Liquid Interface: A Unique Non-Animal Methodological Approach on Vaping Studies. NAM J. 2025, 1, 100060 DOI: 10.1016/j.namjnl.2025.100060Google ScholarThere is no corresponding record for this reference.
- 9Sanchez, L. M.; Bae, D.; Cho, J.; Harlow, A. F.; Kirkpatrick, M. G.; Schuler, L. A.; Miech, R. A.; Dai, H. D.; Sussman, S. Y.; Han, D.-H.; Meza, L.; Leventhal, A. M. Awareness and Use of Vaping Products With a Nicotine Analogue Among Adolescents and Young Adults. Pediatrics 2025, 156 (2), e2024070484 DOI: 10.1542/peds.2024-070484Google ScholarThere is no corresponding record for this reference.
- 10Jordt, S. E.; Jabba, S. V.; Zettler, P. J.; Berman, M. L. Spree Bar, a Vaping System Delivering a Synthetic Nicotine Analogue, Marketed in the USA as ‘PMTA Exempt.’. Tob. Control 2025, 34 (3), 414– 418, DOI: 10.1136/tc-2023-058469Google ScholarThere is no corresponding record for this reference.
- 11Salam, S.; El-Hajj Moussa, F.; El-Hage, R.; El-Hellani, A.; Aoun Saliba, N. A Systematic Review of Analytical Methods for the Separation of Nicotine Enantiomers and Evaluation of Nicotine Sources. Chem. Res. Toxicol. 2023, 36 (3), 334– 341, DOI: 10.1021/acs.chemrestox.2c00310Google ScholarThere is no corresponding record for this reference.
- 12Xing, H.; Andrud, K. W.; Soti, F.; Rouchaud, A.; Jahn, S. C.; Lu, Z.; Cho, Y.-H.; Habibi, S.; Corsino, P.; Slavov, S.; Rocca, J. R.; Lindstrom, J. M.; Lukas, R. J.; Kem, W. R. A Methyl Scan of the Pyrrolidinium Ring of Nicotine Reveals Significant Differences in Its Interactions with Α7 and Α4β2 Nicotinic Acetylcholine Receptors. Mol. Pharmacol. 2020, 98 (2), 168– 180, DOI: 10.1124/mol.119.118786Google ScholarThere is no corresponding record for this reference.
- 13Pourchez, J.; Mercier, C.; Forest, V. The Rise of Nicotine Analogues in Vaping Products. Lancet Respir. Med. 2025, 13 (11), e57– e58, DOI: 10.1016/S2213-2600(25)00296-6Google ScholarThere is no corresponding record for this reference.
- 14Camillo, L.; Zavattaro, E.; Savoia, P. Nicotinamide: A Multifaceted Molecule in Skin Health and Beyond. Medicina 2025, 61, 254, DOI: 10.3390/medicina61020254Google ScholarThere is no corresponding record for this reference.
- 15Chavarrio Cañas, J. E.; Monge-Palacios, M.; Grajales-González, E.; Sarathy, S. M. Early Chemistry of Nicotine Degradation in Heat-Not-Burn Smoking Devices and Conventional Cigarettes: Implications for Users and Second- and Third-Hand Smokers. J. Phys. Chem. A 2021, 125 (15), 3177– 3188, DOI: 10.1021/acs.jpca.1c01650Google ScholarThere is no corresponding record for this reference.
- 16Asensio, J.; Berenguer, D.; Marcilla, A.; Beltran, M. I. Nicotine Fast Pyrolysis under Inert and Air Environments. Effect of Catalysts. J. Anal. Appl. Pyrolysis 2023, 170, 105899 DOI: 10.1016/j.jaap.2023.105899Google ScholarThere is no corresponding record for this reference.
- 17Hoa, N. T.; Ngoc Van, L. T.; Vo, Q. V. Reactions of Nicotine and the Hydroxyl Radical in the Environment: Theoretical Insights into the Mechanism, Kinetics and Products. Chemosphere 2023, 314, 137682 DOI: 10.1016/j.chemosphere.2022.137682Google ScholarThere is no corresponding record for this reference.
- 18Strongin, R. M.; Sharma, E.; Erythropel, H. C.; Kassem, N. O. F.; Noël, A.; Peyton, D. H.; Rahman, I. Chemical and Physiological Interactions between E-Liquid Constituents: Cause for Concern?. Tob. Control 2025, 34 (3), 393– 396, DOI: 10.1136/tc-2023-058546Google ScholarThere is no corresponding record for this reference.
- 19Bitzer, Z. T.; Goel, R.; Reilly, S. M.; Elias, R. J.; Silakov, A.; Foulds, J.; Muscat, J.; Richie, J. P. Effect of Flavoring Chemicals on Free Radical Formation in Electronic Cigarette Aerosols. Free Radical Biol. Med. 2018, 120, 72– 79, DOI: 10.1016/j.freeradbiomed.2018.03.020Google ScholarThere is no corresponding record for this reference.
- 20Jefferis, T.; Sengupta, M.; Bui, A.; Dawkins, C.; Griffin, K.; Strongin, R. M.; Sayes, C. M. Production of 3-Cyanopyridine upon Vaping Nicotinamide and Evaluation of the Toxicological Effects of Individual and Combined Exposures via Isobologram-Based Analysis. Chem. Res. Toxicol. 2025, 38 (11), 2010– 2018, DOI: 10.1021/acs.chemrestox.5c00332Google ScholarThere is no corresponding record for this reference.
- 21Tanaka, A.; Iijima, M.; Kikuchi, Y.; Hoshino, J.; Nose, N. Gas Chromatographic Determination of Nicotinamide in Meats and Meat Products as 3-Cyanopyridine. J. Chromatogr. A 1989, 466, 307– 317, DOI: 10.1016/S0021-9673(01)84626-4Google ScholarThere is no corresponding record for this reference.
- 22Li, B.; Su, J.; Tao, J. Enzyme and Process Development for Production of Nicotinamide. Org. Process Res. Dev. 2011, 15 (1), 291– 293, DOI: 10.1021/op100309gGoogle ScholarThere is no corresponding record for this reference.
- 23Liu, J.; Fan, X.; Zhao, W.; Yang, S.; Xie, W.; Hu, B.; Lu, Q. A Theoretical Investigation on the Thermal Decomposition of Pyridine and the Effect of H2O on the Formation of NOx Precursors. Front. Chem. Sci. Eng. 2021, 15 (5), 1217– 1228, DOI: 10.1007/s11705-020-2024-8Google ScholarThere is no corresponding record for this reference.
- 24Heine, M.; Fink, L.; Schmidt, M. U. 3-Cyanopyridine as a Bridging and Terminal Ligand in Coordination Polymers. CrystEngComm 2018, 20 (46), 7556– 7566, DOI: 10.1039/C8CE01568FGoogle ScholarThere is no corresponding record for this reference.
- 25Gulcin, İ.; Alwasel, S. H. Metal Ions, Metal Chelators and Metal Chelating Assay as Antioxidant Method. Processes 2022, 10 (1), 132, DOI: 10.3390/pr10010132Google ScholarThere is no corresponding record for this reference.
- 26Chen, J. Y.; Canchola, A.; Lin, Y.-H. Carbonyl Composition and Electrophilicity in Vaping Emissions of Flavored and Unflavored E-Liquids. Toxics 2021, 9 (12), 345, DOI: 10.3390/toxics9120345Google ScholarThere is no corresponding record for this reference.
- 27Jensen, R. P.; Strongin, R. M.; Peyton, D. H. Solvent Chemistry in the Electronic Cigarette Reaction Vessel. Sci. Rep. 2017, 7 (1), 42549 DOI: 10.1038/srep42549Google ScholarThere is no corresponding record for this reference.
- 28Kerber, P. J.; Duell, A. K.; Powers, M.; Strongin, R. M.; Peyton, D. H. Effects of Common E-Liquid Flavorants and Added Nicotine on Toxicant Formation during Vaping Analyzed by1 H NMR Spectroscopy. Chem. Res. Toxicol. 2022, 35 (7), 1267– 1276, DOI: 10.1021/acs.chemrestox.2c00110Google ScholarThere is no corresponding record for this reference.
- 29Ferger, B.; Spratt, C.; Earl, C. D.; Teismann, P.; Oertel, W. H.; Kuschinsky, K. Effects of Monoamine Oxidase Inhibition on MPTP-Induced Dopaminergic Neurotoxicity in Mice Naunyn-Schmiedeberg’s Arch. Pharmacol., 358 351 359.Google ScholarThere is no corresponding record for this reference.
- 30Yu, C.; Li, Y.-L.; Liang, M.; Dai, S.-Y.; Ma, L.; Li, W.-G.; Lai, F.; Liu, X.-M. Characteristics and Hazards of the Cinnamaldehyde Oxidation Process. RSC Adv. 2020, 10 (32), 19124– 19133, DOI: 10.1039/C9RA10820CGoogle ScholarThere is no corresponding record for this reference.
- 31Barnes, I.; Solignac, G.; Mellouki, A.; Becker, K. H. Aspects of the Atmospheric Chemistry of Amides. ChemPhysChem 2010, 11 (18), 3844– 3857, DOI: 10.1002/cphc.201000374Google ScholarThere is no corresponding record for this reference.
- 32Erythropel, H. C.; Jabba, S. V.; DeWinter, T. M.; Mendizabal, M.; Anastas, P. T.; Jordt, S. E.; Zimmerman, J. B. Formation of Flavorant–Propylene Glycol Adducts With Novel Toxicological Properties in Chemically Unstable E-Cigarette Liquids. Nicotine Tob. Res. 2019, 21 (9), 1248– 1258, DOI: 10.1093/ntr/nty192Google ScholarThere is no corresponding record for this reference.
- 33Kerber, P. J.; Peyton, D. H. Kinetics of Aldehyde Flavorant-Acetal Formation in E-Liquids with Different E-Cigarette Solvents and Common Additives Studied by1 H NMR Spectroscopy. Chem. Res. Toxicol. 2022, 35 (8), 1410– 1417, DOI: 10.1021/acs.chemrestox.2c00159Google ScholarThere is no corresponding record for this reference.
- 34Mazzaferro, S.; Bermudez, I.; Sine, S. M. Α4β2 Nicotinic Acetylcholine Receptors. J. Biol. Chem. 2017, 292 (7), 2729– 2740, DOI: 10.1074/jbc.M116.764183Google ScholarThere is no corresponding record for this reference.
- 35Taffe, M. A.; Coons, T. R.; Doran, T. A.; Grant, Y.; Vandewater, S. A. Effects of 6-Methyl Nicotine in Middle Aged Female Rats with a History of Nicotine Vapor Self-Administration. Pharmacol. Toxicol. 2025, DOI: 10.1101/2025.09.15.676338Google ScholarThere is no corresponding record for this reference.
- 36Erythropel, H. C.; Jabba, S. V.; Silinski, P.; Anastas, P. T.; Krishnan-Sarin, S.; Zimmerman, J. B.; Jordt, S. E. Variability in Constituents of E-Cigarette Products Containing Nicotine Analogues. JAMA 2024, 332 (9), 753, DOI: 10.1001/jama.2024.12408Google ScholarThere is no corresponding record for this reference.
- 37Effah, F.; Sengupta, M.; Sun, Y.; Faizan, M. I.; Kaur, G.; Raghu, R.; Elzein, A.; Marczylo, T.; Strongin, R. M.; Rahman, I. A Comparative Toxicological Evaluation of Nicotine and Its Analog 6-Methyl Nicotine in E-Cigarette Aerosol Utilizing a 3D in Vitro Human Respiratory Model. Toxicology 2026, 522, 154421 DOI: 10.1016/j.tox.2026.154421Google ScholarThere is no corresponding record for this reference.
- 38Leigh, N. J.; Lawton, R. I.; Hershberger, P. A.; Goniewicz, M. L. Flavourings Significantly Affect Inhalation Toxicity of Aerosol Generated from Electronic Nicotine Delivery Systems (ENDS). Tob. Control 2016, 25 (Suppl 2), ii81– ii87, DOI: 10.1136/tobaccocontrol-2016-053205Google ScholarThere is no corresponding record for this reference.
- 39Leslie, L. J.; Vasanthi Bathrinarayanan, P.; Jackson, P.; Mabiala Ma Muanda, J. A.; Pallett, R.; Stillman, C. J. P.; Marshall, L. J. A Comparative Study of Electronic Cigarette Vapor Extracts on Airway-Related Cell Lines in Vitro. Inhal. Toxicol. 2017, 29 (3), 126– 136, DOI: 10.1080/08958378.2017.1318193Google ScholarThere is no corresponding record for this reference.
- 40Hua, M.; Omaiye, E. E.; Luo, W.; McWhirter, K. J.; Pankow, J. F.; Talbot, P. Identification of Cytotoxic Flavor Chemicals in Top-Selling Electronic Cigarette Refill Fluids. Sci. Rep. 2019, 9 (1), 2782 DOI: 10.1038/s41598-019-38978-wGoogle ScholarThere is no corresponding record for this reference.
- 41Ganguly, K.; Nordström, A.; Thimraj, T. A.; Rahman, M.; Ramström, M.; Sompa, S. I.; Lin, E. Z.; O’Brien, F.; Koelmel, J.; Ernstgård, L.; Johanson, G.; Pollitt, K. J. G.; Palmberg, L.; Upadhyay, S. Addressing the Challenges of E-Cigarette Safety Profiling by Assessment of Pulmonary Toxicological Response in Bronchial and Alveolar Mucosa Models. Sci. Rep. 2020, 10 (1), 20460 DOI: 10.1038/s41598-020-77452-wGoogle ScholarThere is no corresponding record for this reference.
- 42Czekala, L.; Simms, L.; Stevenson, M.; Trelles-Sticken, E.; Walker, P.; Walele, T. High Content Screening in NHBE Cells Shows Significantly Reduced Biological Activity of Flavoured E-Liquids, When Compared to Cigarette Smoke Condensate. Toxicol. In Vitro 2019, 58, 86– 96, DOI: 10.1016/j.tiv.2019.03.018Google ScholarThere is no corresponding record for this reference.
- 43Tierney, P. A.; Karpinski, C. D.; Brown, J. E.; Luo, W.; Pankow, J. F. Flavour Chemicals in Electronic Cigarette Fluids. Tob. Control 2016, 25 (e1), e10– e15, DOI: 10.1136/tobaccocontrol-2014-052175Google ScholarThere is no corresponding record for this reference.
- 44Effah, F.; Taiwo, B.; Baines, D.; Bailey, A.; Marczylo, T. Pulmonary Effects of E-Liquid Flavors: A Systematic Review. J. Toxicol. Environ. Health, Part B 2022, 25 (7), 343– 371, DOI: 10.1080/10937404.2022.2124563Google ScholarThere is no corresponding record for this reference.
- 45Lerner, C. A.; Sundar, I. K.; Yao, H.; Gerloff, J.; Ossip, D. J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors Produced by Electronic Cigarettes and E-Juices with Flavorings Induce Toxicity, Oxidative Stress, and Inflammatory Response in Lung Epithelial Cells and in Mouse Lung. PLoS One 2015, 10 (2), e0116732 DOI: 10.1371/journal.pone.0116732Google ScholarThere is no corresponding record for this reference.
- 46Raduka, A.; Gao, N.; Chatburn, R. L.; Rezaee, F. Electronic Cigarette Exposure Disrupts Airway Epithelial Barrier Function and Exacerbates Viral Infection. Am. J. Physiol. 2023, 325 (5), L580– L593, DOI: 10.1152/ajplung.00135.2023Google ScholarThere is no corresponding record for this reference.
- 47Herbert, J.; Kelty, J. S.; Laskin, J. D.; Laskin, D. L.; Gow, A. J. Menthol Flavoring in E-Cigarette Condensate Causes Pulmonary Dysfunction and Cytotoxicity in Precision Cut Lung Slices. Am. J. Physiol. 2023, 324 (3), L345– L357, DOI: 10.1152/ajplung.00222.2022Google ScholarThere is no corresponding record for this reference.
- 48Kaur, G.; Muthumalage, T.; Rahman, I. Mechanisms of toxicity and biomarkers of flavoring and flavor enhancing chemicals in emerging tobacco and non-tobacco products. Toxicol. Lett. 2018, 288, 143– 155, DOI: 10.1016/j.toxlet.2018.02.025Google ScholarThere is no corresponding record for this reference.
- 49Flavor and Extract Manufacturers Association of the United States. Respiratory Health and Safety in the Flavor Manufacturing Workplace; The Flavor and Extract Manufacturers Association of the United States: Washington, DC, 2004.Google ScholarThere is no corresponding record for this reference.
- 50Jabba, S. V.; Diaz, A. N.; Erythropel, H. C.; Zimmerman, J. B.; Jordt, S.-E. Chemical Adducts of Reactive Flavor Aldehydes Formed in E-Cigarette Liquids Are Cytotoxic and Inhibit Mitochondrial Function in Respiratory Epithelial Cells. Nicotine Tob. Res. 2020, 22 (Supplement_1), S25– S34, DOI: 10.1093/ntr/ntaa185Google ScholarThere is no corresponding record for this reference.
- 51Gerloff, J.; Sundar, I. K.; Freter, R.; Sekera, E. R.; Friedman, A. E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different E-Cigarette Flavoring Chemicals Identified by Gas Chromatography–Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. Vitro Toxicol. 2017, 3 (1), 28– 40, DOI: 10.1089/aivt.2016.0030Google ScholarThere is no corresponding record for this reference.
- 52Clapp, P. W.; Lavrich, K. S.; Van Heusden, C. A.; Lazarowski, E. R.; Carson, J. L.; Jaspers, I. Cinnamaldehyde in Flavored E-Cigarette Liquids Temporarily Suppresses Bronchial Epithelial Cell Ciliary Motility by Dysregulation of Mitochondrial Function. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2019, 316 (3), L470– L486, DOI: 10.1152/ajplung.00304.2018Google ScholarThere is no corresponding record for this reference.
- 53Sherwood, C. L.; Boitano, S. Airway Epithelial Cell Exposure to Distinct E-Cigarette Liquid Flavorings Reveals Toxicity Thresholds and Activation of CFTR by the Chocolate Flavoring 2,5-Dimethypyrazine. Respir. Res. 2016, 17 (1), 57 DOI: 10.1186/s12931-016-0369-9Google ScholarThere is no corresponding record for this reference.
- 54Zaccone, E. J.; Goldsmith, W. T.; Shimko, M. J.; Wells, J. R.; Schwegler-Berry, D.; Willard, P. A.; Case, S. L.; Thompson, J. A.; Fedan, J. S. Diacetyl and 2,3-Pentanedione Exposure of Human Cultured Airway Epithelial Cells: Ion Transport Effects and Metabolism of Butter Flavoring Agents. Toxicol. Appl. Pharmacol. 2015, 289 (3), 542– 549, DOI: 10.1016/j.taap.2015.10.004Google ScholarThere is no corresponding record for this reference.
- 55Krüsemann, E. J. Z.; Havermans, A.; Pennings, J. L. A.; De Graaf, K.; Boesveldt, S.; Talhout, R. Comprehensive Overview of Common E-Liquid Ingredients and How They Can Be Used to Predict an e-Liquid’s Flavour Category. Tob. Control 2021, 30 (2), 185– 191, DOI: 10.1136/tobaccocontrol-2019-055447Google ScholarThere is no corresponding record for this reference.
- 56Yogeswaran, S.; Shaikh, S. Bi.; Manevski, M.; Chand, H. S.; Rahman, I. The Role of Synthetic Coolants, WS-3 and WS-23, in Modulating E-Cigarette-Induced Reactive Oxygen Species (ROS) in Lung Epithelial Cells. Toxicol. Rep. 2022, 9, 1700– 1709, DOI: 10.1016/j.toxrep.2022.08.007Google ScholarThere is no corresponding record for this reference.
- 57Muthumalage, T.; Rahman, I. Pulmonary Immune Response Regulation, Genotoxicity, and Metabolic Reprogramming by Menthol- and Tobacco-Flavored e-Cigarette Exposures in Mice. Toxicol. Sci. 2023, 193 (2), 146– 165, DOI: 10.1093/toxsci/kfad033Google ScholarThere is no corresponding record for this reference.
- 58Goldenson, N. I.; Leventhal, A. M.; Simpson, K. A.; Barrington-Trimis, J. L. A Review of the Use and Appeal of Flavored Electronic Cigarettes. Curr. Addict. Rep. 2019, 6 (2), 98– 113, DOI: 10.1007/s40429-019-00244-4Google ScholarThere is no corresponding record for this reference.
- 59Rutten, J.; Rorije, E.; Geraets, L.; Pauwels, C. Advisory Values for Maximum Emission of Nicotine and 6- Methylnicotine from Nicotine Products without Tobacco for Inhalation; Rijksinstituut voor Volksgezondheid en Milieu RIVM, 2025 DOI: 10.21945/RIVM-2025-0067 .Google ScholarThere is no corresponding record for this reference.
- 60Northrup, T. F.; Stotts, A. L.; Suchting, R.; Khan, A. M.; Green, C.; Klawans, M. R.; Quintana, P. J. E.; Hoh, E.; Hovell, M. F.; Matt, G. E. Thirdhand Smoke Contamination and Infant Nicotine Exposure in a Neonatal Intensive Care Unit: An Observational Study. Nicotine Tob. Res. 2021, 23 (2), 373– 382, DOI: 10.1093/ntr/ntaa167Google ScholarThere is no corresponding record for this reference.
- 61Rodriguez, J.; Liang, D.; Tchen, R.; Yang, I. Saliva and Exhaled Breath Condensate Correlate With Serum in 4–12-Year-Olds Exposed to Secondhand Electronic Cigarette Vapors: A Pilot Study. Biol. Res. Nurs. 2023, 25 (3), 417– 425, DOI: 10.1177/10998004221149959Google ScholarThere is no corresponding record for this reference.
- 62Aramakis, V. B.; Hsieh, C. Y.; Leslie, F. M.; Metherate, R. A Critical Period for Nicotine-Induced Disruption of Synaptic Development in Rat Auditory Cortex. J. Neurosci. 2000, 20 (16), 6106– 6116, DOI: 10.1523/JNEUROSCI.20-16-06106.2000Google ScholarThere is no corresponding record for this reference.
- 63Kowitt, S. D.; Seidenberg, A. B.; Gottfredson O’Shea, N. C.; Ritchie, C.; Galper, E. F.; Sutfin, E. L.; Sheeran, P.; Noar, S. M. Synthetic Nicotine Descriptors: Awareness and Impact on Perceptions of e-Cigarettes among US Youth. Tob. Control 2024, 33 (6), 713– 719, DOI: 10.1136/tc-2023-057928Google ScholarThere is no corresponding record for this reference.
- 64Chang, J. T.; Rostron, B. L. Electronic Nicotine Delivery System (ENDS) Liquid Nicotine Exposure in Young Children Presenting to US Emergency Departments, 2018. Inj. Epidemiol. 2019, 6 (1), 43 DOI: 10.1186/s40621-019-0219-6Google ScholarThere is no corresponding record for this reference.
- 65Tashakkori, N. A.; Rostron, B. L.; Christensen, C. H.; Cullen, K. A. Notes from the Field: E-Cigarette–Associated Cases Reported to Poison Centers ─ United States, April 1, 2022–March 31, 2023. MMWR Morb. Mortal. Wkly. Rep. 2023, 72 (25), 694– 695, DOI: 10.15585/mmwr.mm7225a5Google ScholarThere is no corresponding record for this reference.
- 66Nationwide Children’s. New Study Finds Sharp Increase in Nicotine Pouch Ingestions Among Young Children. https://www.nationwidechildrens.org/newsroom/news-releases/2025/07/nicotine_exposures.Google ScholarThere is no corresponding record for this reference.
- 67U.S. Food and Drug Administration. Regulation and Enforcement of Non-Tobacco Nicotine (NTN) Products , 2023. https://www.fda.gov/tobacco-products/products-ingredients-components/regulation-and-enforcement-non-tobacco-nicotine-ntn-products.Google ScholarThere is no corresponding record for this reference.
- 68Dukat, M.; Fiedler, W.; Dumas, D.; Damaj, I.; Martin, B.; Rosecrans, J.; James, J.; Glennon, R. Pyrrolidine-Modified and 6-Substituted Analogs of Nicotine: A Structure─Affinity Investigation. Eur. J. Med. Chem. 1996, 31 (11), 875– 888, DOI: 10.1016/S0223-5234(97)89850-9Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
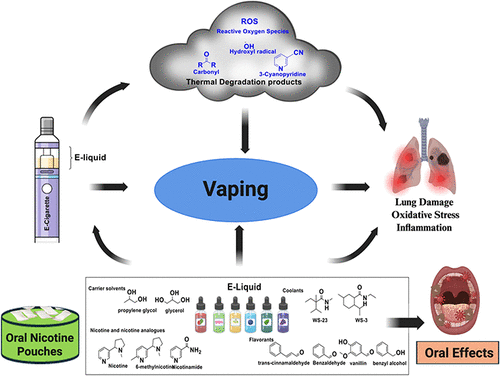
Figure 1
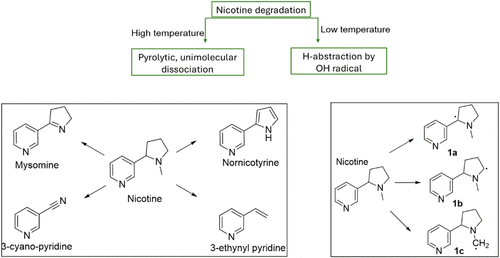
Figure 1. Different mechanistic pathways of nicotine thermal degradation at high (cigarette) and low (e.g., e-cigarette) temperatures. Left: pyrolysis products. Right: Hydroxyl radical hydrogen-abstraction intermediates Figure 1a–c. The left figure is adapted from Asensio et al. (16) The right figure is adapted from Hoa et al. (17)
Figure 2
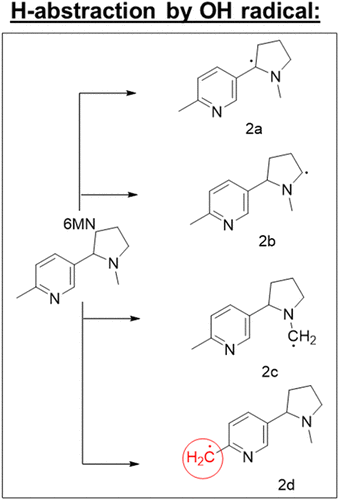
Figure 2. Proposed radical intermediates from the preferred hydrogen atom abstraction sites of 6-MN at vaping-relevant temperatures.
Figure 3

Figure 3. Temperature-dependent formation of 3-CP from nicotinamide. The 3-CP yield increases with temperature, showing progressive growth from 250 to 335 °C. Reprinted with permission from ref (20). Copyright 2025 by the American Chemical Society.
Figure 4

Figure 4. LC10 dose response (DR) curves and chemical interaction model of 3-cyanopyridine (3CP) and nicotinamide (NA) tested on BEAS-2B cells. (A) DR of 3CP graphed on the log-transformed x-axis of concentrations tested up to 1000 ppm. (B) DR of NA tested at concentrations up to 10,000 ppm. (C) The combined DR curves of 3CP and NA are plotted on the same concentration scale. The dose–response curves are identical to those shown in panels A and C, but on a comparable scale. The 3CP curve above 1000 ppm is a model prediction based on the curve produced at lower concentrations, rather than on tested values. (D) Isobologram of 3CP:NA, indicating synergistic interaction. Reprinted with permission from ref (20). Copyright 2025 by the American Chemical Society.
Figure 5
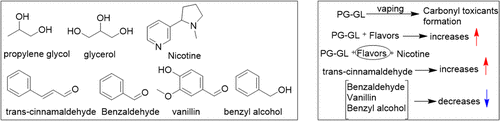
Figure 5. Selected relatively common chemical components of e-liquids addition to nicotine and varying effects on the formation of carbonyl toxicants. (28).
Figure 6
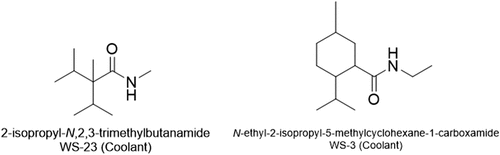
Figure 6. Chemical structures of coolants WS-23 and WS-3.
Figure 7

Figure 7. Effects on acetal formation in e-liquids in the presence of nicotine in free-base versus salt forms. Acetals form in e-liquids when aldehyde flavorants, such as benzaldehyde, vanillin, and trans-cinnamaldehyde. (33)
Figure 8

Figure 8. Series of pie-charts displaying the distribution of flavor categories for nicotine-analog containing products distributed by (A) Spree Bar, (B) Kumi-Six, (C) Katchmi, (D) SBX, and (E) Outlaw Dip.
References
This article references 68 other publications.
- 1Vanhee, C.; Dill, M.; Canfyn, M.; Tuenter, E.; Barhdadi, S. The Emergence of a Novel Synthetic Nicotine Analog 6-Methyl Nicotine (6-MN) in Proclaimed Tobacco- and Nicotine-Free Pouches Available in Europe. Drug Test. Anal. 2025, 17 (8), 1368– 1379, DOI: 10.1002/dta.3841There is no corresponding record for this reference.
- 2Effah, F.; Sun, Y.; Lin, K.; Rahman, I. A Comparative Toxicological Evaluation of Emerging Nicotine Analogs 6-Methyl Nicotine and Nicotinamide: A Scoping Review. Arch. Toxicol. 2025, 99 (4), 1333– 1340, DOI: 10.1007/s00204-025-03960-1There is no corresponding record for this reference.
- 3Jabba, S. V.; Jordt, S. E. Marketing of Nicotinamide as Nicotine Replacement in Electronic Cigarettes and Smokeless Tobacco. Tob. Prev. Cessat. 2024, 10 (August), 1– 5, DOI: 10.18332/tpc/187767There is no corresponding record for this reference.
- 4Pankow, J. F.; Luo, W.; McWhirter, K. J.; Sengupta, M.; Strongin, R. M. Levels of the Nicotine Analog 6-Methyl Nicotine as a Naturally Formed Tobacco Alkaloid in Tobacco and Tobacco Products. Sci. Rep. 2025, 15 (1), 17945 DOI: 10.1038/s41598-025-01392-6There is no corresponding record for this reference.
- 5Jordt, S. E.; Jabba, S. V. Introduction of Nicotine Analogue-Containing Oral Pouch Products in the United States. Tob. Prev. Cessat. 2024, 10, 61 DOI: 10.18332/tpc/195621There is no corresponding record for this reference.
- 6Effah, F.; Sun, Y.; Friedman, A.; Rahman, I. Emerging Nicotine Analog 6-Methyl Nicotine Increases Reactive Oxygen Species in Aerosols and Cytotoxicity in Human Bronchial Epithelial Cells. Toxicol. Lett. 2025, 405, 9– 15, DOI: 10.1016/j.toxlet.2025.01.007There is no corresponding record for this reference.
- 7Qi, H.; Chang, X.; Wang, K.; Xu, Q.; Liu, M.; Han, B. Comparative Analyses of Transcriptome Sequencing and Carcinogenic Exposure Toxicity of Nicotine and 6-Methyl Nicotine in Human Bronchial Epithelial Cells. Toxicol. In Vitro 2023, 93, 105661 DOI: 10.1016/j.tiv.2023.105661There is no corresponding record for this reference.
- 8Effah, F.; Shaikh, S. B.; Chalupa, D.; Faizan, M. I.; Elder, A.; Rahman, I. High-Resistance Coils in E-Cigarettes Increase Heavy Metals Leaching into Aerosols to Cause Oxidants Generation in Human Bronchial Epithelial Cells at Air-Liquid Interface: A Unique Non-Animal Methodological Approach on Vaping Studies. NAM J. 2025, 1, 100060 DOI: 10.1016/j.namjnl.2025.100060There is no corresponding record for this reference.
- 9Sanchez, L. M.; Bae, D.; Cho, J.; Harlow, A. F.; Kirkpatrick, M. G.; Schuler, L. A.; Miech, R. A.; Dai, H. D.; Sussman, S. Y.; Han, D.-H.; Meza, L.; Leventhal, A. M. Awareness and Use of Vaping Products With a Nicotine Analogue Among Adolescents and Young Adults. Pediatrics 2025, 156 (2), e2024070484 DOI: 10.1542/peds.2024-070484There is no corresponding record for this reference.
- 10Jordt, S. E.; Jabba, S. V.; Zettler, P. J.; Berman, M. L. Spree Bar, a Vaping System Delivering a Synthetic Nicotine Analogue, Marketed in the USA as ‘PMTA Exempt.’. Tob. Control 2025, 34 (3), 414– 418, DOI: 10.1136/tc-2023-058469There is no corresponding record for this reference.
- 11Salam, S.; El-Hajj Moussa, F.; El-Hage, R.; El-Hellani, A.; Aoun Saliba, N. A Systematic Review of Analytical Methods for the Separation of Nicotine Enantiomers and Evaluation of Nicotine Sources. Chem. Res. Toxicol. 2023, 36 (3), 334– 341, DOI: 10.1021/acs.chemrestox.2c00310There is no corresponding record for this reference.
- 12Xing, H.; Andrud, K. W.; Soti, F.; Rouchaud, A.; Jahn, S. C.; Lu, Z.; Cho, Y.-H.; Habibi, S.; Corsino, P.; Slavov, S.; Rocca, J. R.; Lindstrom, J. M.; Lukas, R. J.; Kem, W. R. A Methyl Scan of the Pyrrolidinium Ring of Nicotine Reveals Significant Differences in Its Interactions with Α7 and Α4β2 Nicotinic Acetylcholine Receptors. Mol. Pharmacol. 2020, 98 (2), 168– 180, DOI: 10.1124/mol.119.118786There is no corresponding record for this reference.
- 13Pourchez, J.; Mercier, C.; Forest, V. The Rise of Nicotine Analogues in Vaping Products. Lancet Respir. Med. 2025, 13 (11), e57– e58, DOI: 10.1016/S2213-2600(25)00296-6There is no corresponding record for this reference.
- 14Camillo, L.; Zavattaro, E.; Savoia, P. Nicotinamide: A Multifaceted Molecule in Skin Health and Beyond. Medicina 2025, 61, 254, DOI: 10.3390/medicina61020254There is no corresponding record for this reference.
- 15Chavarrio Cañas, J. E.; Monge-Palacios, M.; Grajales-González, E.; Sarathy, S. M. Early Chemistry of Nicotine Degradation in Heat-Not-Burn Smoking Devices and Conventional Cigarettes: Implications for Users and Second- and Third-Hand Smokers. J. Phys. Chem. A 2021, 125 (15), 3177– 3188, DOI: 10.1021/acs.jpca.1c01650There is no corresponding record for this reference.
- 16Asensio, J.; Berenguer, D.; Marcilla, A.; Beltran, M. I. Nicotine Fast Pyrolysis under Inert and Air Environments. Effect of Catalysts. J. Anal. Appl. Pyrolysis 2023, 170, 105899 DOI: 10.1016/j.jaap.2023.105899There is no corresponding record for this reference.
- 17Hoa, N. T.; Ngoc Van, L. T.; Vo, Q. V. Reactions of Nicotine and the Hydroxyl Radical in the Environment: Theoretical Insights into the Mechanism, Kinetics and Products. Chemosphere 2023, 314, 137682 DOI: 10.1016/j.chemosphere.2022.137682There is no corresponding record for this reference.
- 18Strongin, R. M.; Sharma, E.; Erythropel, H. C.; Kassem, N. O. F.; Noël, A.; Peyton, D. H.; Rahman, I. Chemical and Physiological Interactions between E-Liquid Constituents: Cause for Concern?. Tob. Control 2025, 34 (3), 393– 396, DOI: 10.1136/tc-2023-058546There is no corresponding record for this reference.
- 19Bitzer, Z. T.; Goel, R.; Reilly, S. M.; Elias, R. J.; Silakov, A.; Foulds, J.; Muscat, J.; Richie, J. P. Effect of Flavoring Chemicals on Free Radical Formation in Electronic Cigarette Aerosols. Free Radical Biol. Med. 2018, 120, 72– 79, DOI: 10.1016/j.freeradbiomed.2018.03.020There is no corresponding record for this reference.
- 20Jefferis, T.; Sengupta, M.; Bui, A.; Dawkins, C.; Griffin, K.; Strongin, R. M.; Sayes, C. M. Production of 3-Cyanopyridine upon Vaping Nicotinamide and Evaluation of the Toxicological Effects of Individual and Combined Exposures via Isobologram-Based Analysis. Chem. Res. Toxicol. 2025, 38 (11), 2010– 2018, DOI: 10.1021/acs.chemrestox.5c00332There is no corresponding record for this reference.
- 21Tanaka, A.; Iijima, M.; Kikuchi, Y.; Hoshino, J.; Nose, N. Gas Chromatographic Determination of Nicotinamide in Meats and Meat Products as 3-Cyanopyridine. J. Chromatogr. A 1989, 466, 307– 317, DOI: 10.1016/S0021-9673(01)84626-4There is no corresponding record for this reference.
- 22Li, B.; Su, J.; Tao, J. Enzyme and Process Development for Production of Nicotinamide. Org. Process Res. Dev. 2011, 15 (1), 291– 293, DOI: 10.1021/op100309gThere is no corresponding record for this reference.
- 23Liu, J.; Fan, X.; Zhao, W.; Yang, S.; Xie, W.; Hu, B.; Lu, Q. A Theoretical Investigation on the Thermal Decomposition of Pyridine and the Effect of H2O on the Formation of NOx Precursors. Front. Chem. Sci. Eng. 2021, 15 (5), 1217– 1228, DOI: 10.1007/s11705-020-2024-8There is no corresponding record for this reference.
- 24Heine, M.; Fink, L.; Schmidt, M. U. 3-Cyanopyridine as a Bridging and Terminal Ligand in Coordination Polymers. CrystEngComm 2018, 20 (46), 7556– 7566, DOI: 10.1039/C8CE01568FThere is no corresponding record for this reference.
- 25Gulcin, İ.; Alwasel, S. H. Metal Ions, Metal Chelators and Metal Chelating Assay as Antioxidant Method. Processes 2022, 10 (1), 132, DOI: 10.3390/pr10010132There is no corresponding record for this reference.
- 26Chen, J. Y.; Canchola, A.; Lin, Y.-H. Carbonyl Composition and Electrophilicity in Vaping Emissions of Flavored and Unflavored E-Liquids. Toxics 2021, 9 (12), 345, DOI: 10.3390/toxics9120345There is no corresponding record for this reference.
- 27Jensen, R. P.; Strongin, R. M.; Peyton, D. H. Solvent Chemistry in the Electronic Cigarette Reaction Vessel. Sci. Rep. 2017, 7 (1), 42549 DOI: 10.1038/srep42549There is no corresponding record for this reference.
- 28Kerber, P. J.; Duell, A. K.; Powers, M.; Strongin, R. M.; Peyton, D. H. Effects of Common E-Liquid Flavorants and Added Nicotine on Toxicant Formation during Vaping Analyzed by1 H NMR Spectroscopy. Chem. Res. Toxicol. 2022, 35 (7), 1267– 1276, DOI: 10.1021/acs.chemrestox.2c00110There is no corresponding record for this reference.
- 29Ferger, B.; Spratt, C.; Earl, C. D.; Teismann, P.; Oertel, W. H.; Kuschinsky, K. Effects of Monoamine Oxidase Inhibition on MPTP-Induced Dopaminergic Neurotoxicity in Mice Naunyn-Schmiedeberg’s Arch. Pharmacol., 358 351 359.There is no corresponding record for this reference.
- 30Yu, C.; Li, Y.-L.; Liang, M.; Dai, S.-Y.; Ma, L.; Li, W.-G.; Lai, F.; Liu, X.-M. Characteristics and Hazards of the Cinnamaldehyde Oxidation Process. RSC Adv. 2020, 10 (32), 19124– 19133, DOI: 10.1039/C9RA10820CThere is no corresponding record for this reference.
- 31Barnes, I.; Solignac, G.; Mellouki, A.; Becker, K. H. Aspects of the Atmospheric Chemistry of Amides. ChemPhysChem 2010, 11 (18), 3844– 3857, DOI: 10.1002/cphc.201000374There is no corresponding record for this reference.
- 32Erythropel, H. C.; Jabba, S. V.; DeWinter, T. M.; Mendizabal, M.; Anastas, P. T.; Jordt, S. E.; Zimmerman, J. B. Formation of Flavorant–Propylene Glycol Adducts With Novel Toxicological Properties in Chemically Unstable E-Cigarette Liquids. Nicotine Tob. Res. 2019, 21 (9), 1248– 1258, DOI: 10.1093/ntr/nty192There is no corresponding record for this reference.
- 33Kerber, P. J.; Peyton, D. H. Kinetics of Aldehyde Flavorant-Acetal Formation in E-Liquids with Different E-Cigarette Solvents and Common Additives Studied by1 H NMR Spectroscopy. Chem. Res. Toxicol. 2022, 35 (8), 1410– 1417, DOI: 10.1021/acs.chemrestox.2c00159There is no corresponding record for this reference.
- 34Mazzaferro, S.; Bermudez, I.; Sine, S. M. Α4β2 Nicotinic Acetylcholine Receptors. J. Biol. Chem. 2017, 292 (7), 2729– 2740, DOI: 10.1074/jbc.M116.764183There is no corresponding record for this reference.
- 35Taffe, M. A.; Coons, T. R.; Doran, T. A.; Grant, Y.; Vandewater, S. A. Effects of 6-Methyl Nicotine in Middle Aged Female Rats with a History of Nicotine Vapor Self-Administration. Pharmacol. Toxicol. 2025, DOI: 10.1101/2025.09.15.676338There is no corresponding record for this reference.
- 36Erythropel, H. C.; Jabba, S. V.; Silinski, P.; Anastas, P. T.; Krishnan-Sarin, S.; Zimmerman, J. B.; Jordt, S. E. Variability in Constituents of E-Cigarette Products Containing Nicotine Analogues. JAMA 2024, 332 (9), 753, DOI: 10.1001/jama.2024.12408There is no corresponding record for this reference.
- 37Effah, F.; Sengupta, M.; Sun, Y.; Faizan, M. I.; Kaur, G.; Raghu, R.; Elzein, A.; Marczylo, T.; Strongin, R. M.; Rahman, I. A Comparative Toxicological Evaluation of Nicotine and Its Analog 6-Methyl Nicotine in E-Cigarette Aerosol Utilizing a 3D in Vitro Human Respiratory Model. Toxicology 2026, 522, 154421 DOI: 10.1016/j.tox.2026.154421There is no corresponding record for this reference.
- 38Leigh, N. J.; Lawton, R. I.; Hershberger, P. A.; Goniewicz, M. L. Flavourings Significantly Affect Inhalation Toxicity of Aerosol Generated from Electronic Nicotine Delivery Systems (ENDS). Tob. Control 2016, 25 (Suppl 2), ii81– ii87, DOI: 10.1136/tobaccocontrol-2016-053205There is no corresponding record for this reference.
- 39Leslie, L. J.; Vasanthi Bathrinarayanan, P.; Jackson, P.; Mabiala Ma Muanda, J. A.; Pallett, R.; Stillman, C. J. P.; Marshall, L. J. A Comparative Study of Electronic Cigarette Vapor Extracts on Airway-Related Cell Lines in Vitro. Inhal. Toxicol. 2017, 29 (3), 126– 136, DOI: 10.1080/08958378.2017.1318193There is no corresponding record for this reference.
- 40Hua, M.; Omaiye, E. E.; Luo, W.; McWhirter, K. J.; Pankow, J. F.; Talbot, P. Identification of Cytotoxic Flavor Chemicals in Top-Selling Electronic Cigarette Refill Fluids. Sci. Rep. 2019, 9 (1), 2782 DOI: 10.1038/s41598-019-38978-wThere is no corresponding record for this reference.
- 41Ganguly, K.; Nordström, A.; Thimraj, T. A.; Rahman, M.; Ramström, M.; Sompa, S. I.; Lin, E. Z.; O’Brien, F.; Koelmel, J.; Ernstgård, L.; Johanson, G.; Pollitt, K. J. G.; Palmberg, L.; Upadhyay, S. Addressing the Challenges of E-Cigarette Safety Profiling by Assessment of Pulmonary Toxicological Response in Bronchial and Alveolar Mucosa Models. Sci. Rep. 2020, 10 (1), 20460 DOI: 10.1038/s41598-020-77452-wThere is no corresponding record for this reference.
- 42Czekala, L.; Simms, L.; Stevenson, M.; Trelles-Sticken, E.; Walker, P.; Walele, T. High Content Screening in NHBE Cells Shows Significantly Reduced Biological Activity of Flavoured E-Liquids, When Compared to Cigarette Smoke Condensate. Toxicol. In Vitro 2019, 58, 86– 96, DOI: 10.1016/j.tiv.2019.03.018There is no corresponding record for this reference.
- 43Tierney, P. A.; Karpinski, C. D.; Brown, J. E.; Luo, W.; Pankow, J. F. Flavour Chemicals in Electronic Cigarette Fluids. Tob. Control 2016, 25 (e1), e10– e15, DOI: 10.1136/tobaccocontrol-2014-052175There is no corresponding record for this reference.
- 44Effah, F.; Taiwo, B.; Baines, D.; Bailey, A.; Marczylo, T. Pulmonary Effects of E-Liquid Flavors: A Systematic Review. J. Toxicol. Environ. Health, Part B 2022, 25 (7), 343– 371, DOI: 10.1080/10937404.2022.2124563There is no corresponding record for this reference.
- 45Lerner, C. A.; Sundar, I. K.; Yao, H.; Gerloff, J.; Ossip, D. J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors Produced by Electronic Cigarettes and E-Juices with Flavorings Induce Toxicity, Oxidative Stress, and Inflammatory Response in Lung Epithelial Cells and in Mouse Lung. PLoS One 2015, 10 (2), e0116732 DOI: 10.1371/journal.pone.0116732There is no corresponding record for this reference.
- 46Raduka, A.; Gao, N.; Chatburn, R. L.; Rezaee, F. Electronic Cigarette Exposure Disrupts Airway Epithelial Barrier Function and Exacerbates Viral Infection. Am. J. Physiol. 2023, 325 (5), L580– L593, DOI: 10.1152/ajplung.00135.2023There is no corresponding record for this reference.
- 47Herbert, J.; Kelty, J. S.; Laskin, J. D.; Laskin, D. L.; Gow, A. J. Menthol Flavoring in E-Cigarette Condensate Causes Pulmonary Dysfunction and Cytotoxicity in Precision Cut Lung Slices. Am. J. Physiol. 2023, 324 (3), L345– L357, DOI: 10.1152/ajplung.00222.2022There is no corresponding record for this reference.
- 48Kaur, G.; Muthumalage, T.; Rahman, I. Mechanisms of toxicity and biomarkers of flavoring and flavor enhancing chemicals in emerging tobacco and non-tobacco products. Toxicol. Lett. 2018, 288, 143– 155, DOI: 10.1016/j.toxlet.2018.02.025There is no corresponding record for this reference.
- 49Flavor and Extract Manufacturers Association of the United States. Respiratory Health and Safety in the Flavor Manufacturing Workplace; The Flavor and Extract Manufacturers Association of the United States: Washington, DC, 2004.There is no corresponding record for this reference.
- 50Jabba, S. V.; Diaz, A. N.; Erythropel, H. C.; Zimmerman, J. B.; Jordt, S.-E. Chemical Adducts of Reactive Flavor Aldehydes Formed in E-Cigarette Liquids Are Cytotoxic and Inhibit Mitochondrial Function in Respiratory Epithelial Cells. Nicotine Tob. Res. 2020, 22 (Supplement_1), S25– S34, DOI: 10.1093/ntr/ntaa185There is no corresponding record for this reference.
- 51Gerloff, J.; Sundar, I. K.; Freter, R.; Sekera, E. R.; Friedman, A. E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different E-Cigarette Flavoring Chemicals Identified by Gas Chromatography–Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. Vitro Toxicol. 2017, 3 (1), 28– 40, DOI: 10.1089/aivt.2016.0030There is no corresponding record for this reference.
- 52Clapp, P. W.; Lavrich, K. S.; Van Heusden, C. A.; Lazarowski, E. R.; Carson, J. L.; Jaspers, I. Cinnamaldehyde in Flavored E-Cigarette Liquids Temporarily Suppresses Bronchial Epithelial Cell Ciliary Motility by Dysregulation of Mitochondrial Function. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2019, 316 (3), L470– L486, DOI: 10.1152/ajplung.00304.2018There is no corresponding record for this reference.
- 53Sherwood, C. L.; Boitano, S. Airway Epithelial Cell Exposure to Distinct E-Cigarette Liquid Flavorings Reveals Toxicity Thresholds and Activation of CFTR by the Chocolate Flavoring 2,5-Dimethypyrazine. Respir. Res. 2016, 17 (1), 57 DOI: 10.1186/s12931-016-0369-9There is no corresponding record for this reference.
- 54Zaccone, E. J.; Goldsmith, W. T.; Shimko, M. J.; Wells, J. R.; Schwegler-Berry, D.; Willard, P. A.; Case, S. L.; Thompson, J. A.; Fedan, J. S. Diacetyl and 2,3-Pentanedione Exposure of Human Cultured Airway Epithelial Cells: Ion Transport Effects and Metabolism of Butter Flavoring Agents. Toxicol. Appl. Pharmacol. 2015, 289 (3), 542– 549, DOI: 10.1016/j.taap.2015.10.004There is no corresponding record for this reference.
- 55Krüsemann, E. J. Z.; Havermans, A.; Pennings, J. L. A.; De Graaf, K.; Boesveldt, S.; Talhout, R. Comprehensive Overview of Common E-Liquid Ingredients and How They Can Be Used to Predict an e-Liquid’s Flavour Category. Tob. Control 2021, 30 (2), 185– 191, DOI: 10.1136/tobaccocontrol-2019-055447There is no corresponding record for this reference.
- 56Yogeswaran, S.; Shaikh, S. Bi.; Manevski, M.; Chand, H. S.; Rahman, I. The Role of Synthetic Coolants, WS-3 and WS-23, in Modulating E-Cigarette-Induced Reactive Oxygen Species (ROS) in Lung Epithelial Cells. Toxicol. Rep. 2022, 9, 1700– 1709, DOI: 10.1016/j.toxrep.2022.08.007There is no corresponding record for this reference.
- 57Muthumalage, T.; Rahman, I. Pulmonary Immune Response Regulation, Genotoxicity, and Metabolic Reprogramming by Menthol- and Tobacco-Flavored e-Cigarette Exposures in Mice. Toxicol. Sci. 2023, 193 (2), 146– 165, DOI: 10.1093/toxsci/kfad033There is no corresponding record for this reference.
- 58Goldenson, N. I.; Leventhal, A. M.; Simpson, K. A.; Barrington-Trimis, J. L. A Review of the Use and Appeal of Flavored Electronic Cigarettes. Curr. Addict. Rep. 2019, 6 (2), 98– 113, DOI: 10.1007/s40429-019-00244-4There is no corresponding record for this reference.
- 59Rutten, J.; Rorije, E.; Geraets, L.; Pauwels, C. Advisory Values for Maximum Emission of Nicotine and 6- Methylnicotine from Nicotine Products without Tobacco for Inhalation; Rijksinstituut voor Volksgezondheid en Milieu RIVM, 2025 DOI: 10.21945/RIVM-2025-0067 .There is no corresponding record for this reference.
- 60Northrup, T. F.; Stotts, A. L.; Suchting, R.; Khan, A. M.; Green, C.; Klawans, M. R.; Quintana, P. J. E.; Hoh, E.; Hovell, M. F.; Matt, G. E. Thirdhand Smoke Contamination and Infant Nicotine Exposure in a Neonatal Intensive Care Unit: An Observational Study. Nicotine Tob. Res. 2021, 23 (2), 373– 382, DOI: 10.1093/ntr/ntaa167There is no corresponding record for this reference.
- 61Rodriguez, J.; Liang, D.; Tchen, R.; Yang, I. Saliva and Exhaled Breath Condensate Correlate With Serum in 4–12-Year-Olds Exposed to Secondhand Electronic Cigarette Vapors: A Pilot Study. Biol. Res. Nurs. 2023, 25 (3), 417– 425, DOI: 10.1177/10998004221149959There is no corresponding record for this reference.
- 62Aramakis, V. B.; Hsieh, C. Y.; Leslie, F. M.; Metherate, R. A Critical Period for Nicotine-Induced Disruption of Synaptic Development in Rat Auditory Cortex. J. Neurosci. 2000, 20 (16), 6106– 6116, DOI: 10.1523/JNEUROSCI.20-16-06106.2000There is no corresponding record for this reference.
- 63Kowitt, S. D.; Seidenberg, A. B.; Gottfredson O’Shea, N. C.; Ritchie, C.; Galper, E. F.; Sutfin, E. L.; Sheeran, P.; Noar, S. M. Synthetic Nicotine Descriptors: Awareness and Impact on Perceptions of e-Cigarettes among US Youth. Tob. Control 2024, 33 (6), 713– 719, DOI: 10.1136/tc-2023-057928There is no corresponding record for this reference.
- 64Chang, J. T.; Rostron, B. L. Electronic Nicotine Delivery System (ENDS) Liquid Nicotine Exposure in Young Children Presenting to US Emergency Departments, 2018. Inj. Epidemiol. 2019, 6 (1), 43 DOI: 10.1186/s40621-019-0219-6There is no corresponding record for this reference.
- 65Tashakkori, N. A.; Rostron, B. L.; Christensen, C. H.; Cullen, K. A. Notes from the Field: E-Cigarette–Associated Cases Reported to Poison Centers ─ United States, April 1, 2022–March 31, 2023. MMWR Morb. Mortal. Wkly. Rep. 2023, 72 (25), 694– 695, DOI: 10.15585/mmwr.mm7225a5There is no corresponding record for this reference.
- 66Nationwide Children’s. New Study Finds Sharp Increase in Nicotine Pouch Ingestions Among Young Children. https://www.nationwidechildrens.org/newsroom/news-releases/2025/07/nicotine_exposures.There is no corresponding record for this reference.
- 67U.S. Food and Drug Administration. Regulation and Enforcement of Non-Tobacco Nicotine (NTN) Products , 2023. https://www.fda.gov/tobacco-products/products-ingredients-components/regulation-and-enforcement-non-tobacco-nicotine-ntn-products.There is no corresponding record for this reference.
- 68Dukat, M.; Fiedler, W.; Dumas, D.; Damaj, I.; Martin, B.; Rosecrans, J.; James, J.; Glennon, R. Pyrrolidine-Modified and 6-Substituted Analogs of Nicotine: A Structure─Affinity Investigation. Eur. J. Med. Chem. 1996, 31 (11), 875– 888, DOI: 10.1016/S0223-5234(97)89850-9There is no corresponding record for this reference.


