Synthetic Applications of Hydroxamic Acids and Their Derivatives in Organic ChemistryClick to copy article linkArticle link copied!
- Rui Wang
- Wenbo H. Liu*Wenbo H. Liu*E-mail: [email protected]School of Chemistry, Sun Yat-Sen University, Guangzhou 510006, P. R. ChinaMore by Wenbo H. Liu
Abstract
Hydroxamic acids and their derivatives are a versatile class of organic compounds with broad utility in synthetic chemistry. This review highlights key synthetic transformations involving these molecules, covering important reactions such as their use in Weinreb amide chemistry, rearrangement processes, C–H activation directed by hydroxamate groups, their role as precursors to N-centered radicals and aza-oxyallyl cations, reduction reactions, and umpolung transformations initiated by the N–O bond cleavage.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
Scheme 1
2. Chemistry of the Weinreb Amides
Scheme 2
3. Rearrangement
3.1. Lossen Rearrangement
Scheme 3
3.2. HERON Rearrangement
Scheme 4
Scheme 5
4. C–H Bond Activation
Scheme 6
Scheme 7
Scheme 8
Scheme 9
Scheme 10
Scheme 11
Scheme 12
Scheme 13
Scheme 14
Scheme 15
Scheme 16
Scheme 17
5. N-Centered Radical Precursor Reagents
Scheme 18
Scheme 19
Scheme 20
Scheme 21
Scheme 22
Scheme 23
Scheme 24
6. Aza-Oxyallyl Cationic Precursor Reagents
6.1. Aza-Oxyallyl Cations as 1,3-Dipoles in Cycloaddition
Scheme 25
6.2. Aza-Oxyallyl Cations as Alkylating Agents
Scheme 26
Scheme 27
Scheme 28
7. Reduction Reaction
Scheme 29
Scheme 30
8. Umpolung Reaction
Scheme 31
Scheme 32
Scheme 33
Scheme 34
Scheme 35
Scheme 36
Scheme 37
Scheme 38
Scheme 39
Scheme 40
9. Other Reaction Modes
9.1. N-Centered Functionalization of Hydroxamates
Scheme 41
Scheme 42
Scheme 43
9.2. α-C–H Functionalization of Nitrogen in Hydroxamates
Scheme 44
Scheme 45
Scheme 46
10. Summary and Outlook
Data Availability
The data underlying this study are available in the published article.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (22471297), and the Guangzhou Municipal Science and Technology Bureau (2025A04J2067).
References
This article references 135 other publications.
- 1(a) Lossen, H. Ueber die Oxalohydroxamsäure. Justus Liebigs Ann. Chem. 1869, 150 (3), 314– 322, DOI: 10.1002/jlac.18691500304Google ScholarThere is no corresponding record for this reference.(b) Bauer, L.; Exner, O. The Chemistry of Hydroxamic Acids and N-Hydroxyimides. Angew. Chem., Int. Ed. 1974, 13 (6), 376– 384, DOI: 10.1002/anie.197403761Google ScholarThere is no corresponding record for this reference.
- 2Keth, J.; Johann, T.; Frey, H. Hydroxamic Acid: An Underrated Moiety? Marrying Bioinorganic Chemistry and Polymer Science. Biomacromolecules 2020, 21 (7), 2546– 2556, DOI: 10.1021/acs.biomac.0c00449Google ScholarThere is no corresponding record for this reference.
- 3Millar, A. W.; Brown, P. D.; Moore, J.; Galloway, W. A.; Cornish, A. G.; Lenehan, T. J.; Lynch, K. P. Results of Single and Repeat Dose Studies of the Oral Matrix Metalloproteinase Inhibitor Marimastat in Healthy Male Volunteers. Br. J. Clin. Pharmacol. 1998, 45 (1), 21– 26, DOI: 10.1046/j.1365-2125.1998.00639.xGoogle ScholarThere is no corresponding record for this reference.
- 4Zhao, C.; Dong, H.; Xu, Q.; Zhang, Y. Histone Deacetylase (HDAC) Inhibitors in Cancer: a Patent Review (2017-present). Expert Opin. Ther. Pat. 2020, 30 (4), 263– 274, DOI: 10.1080/13543776.2020.1725470Google ScholarThere is no corresponding record for this reference.
- 5(a) Lin, Z.; Decker, E. A.; Goddard, J. M. Preparation of Metal Chelating Active Packaging Materials by Laminated Photografting. J. Coat. Technol. Res. 2016, 13 (2), 395– 404, DOI: 10.1007/s11998-015-9767-zGoogle ScholarThere is no corresponding record for this reference.(b) Hamilton, J. L.; Kizhakkedathu, J. N. Polymeric Nanocarriers for the Treatment of Systemic Iron Overload. Mol. Cell. Ther. 2015, 3 (1), 3 DOI: 10.1186/s40591-015-0039-1Google ScholarThere is no corresponding record for this reference.
- 6(a) Nahm, S.; Weinreb, S. M. N-Methoxy-N-methylamides as Effective Acylating Agents. Tetrahedron Lett. 1981, 22 (39), 3815– 3818, DOI: 10.1016/S0040-4039(01)91316-4Google ScholarThere is no corresponding record for this reference.(b) Sibi, M. P. Chemistry of N-Methoxy-N-Methylamides. Applications in Synthesis. A Review. Org. Prep. Proced. Int. 1993, 25 (1), 15– 40, DOI: 10.1080/00304949309457931Google ScholarThere is no corresponding record for this reference.(c) Mentzel, M.; Hoffmann, H. M. R. N-Methoxy-N-Methylamides (Weinreb Amides) in Modern Organic Synthesis. J. Prakt. Chem. 1997, 339 (1), 517– 524, DOI: 10.1002/prac.19973390194Google ScholarThere is no corresponding record for this reference.(d) Balasubramaniam, S.; Aidhen, I. S. The Growing Synthetic Utility of the Weinreb Amide. Synthesis 2008, 2008 (23), 3707– 3738, DOI: 10.1055/s-0028-1083226Google ScholarThere is no corresponding record for this reference.
- 7(a) Molina, P.; Tárraga, A.; Arques, A. 5.26 - Functions with at Least One Oxygen, YCO. In Comprehensive Organic Functional Group Transformations II; Katritzky, A. R.; Taylor, R. J. K., Eds.; Elsevier, 2005; pp 949– 973.Google ScholarThere is no corresponding record for this reference.(b) Wang, R.; Liu, W. H. Amide Synthesis from Decarboxylative Coupling of Isocyanates and Carboxylic Acids. ChemBioChem 2025, 26 (2), e202400770 DOI: 10.1002/cbic.202400770Google ScholarThere is no corresponding record for this reference.
- 8Podlaha, J.; Cíísařová, I.; Soukupová, L.; Schraml, J.; Exner, O. Structure of Alkali Metal Hydroxamates and their Lossen Rearrangement. J. Chem. Res. 1998, (9), 520– 521, DOI: 10.1039/a802942cGoogle ScholarThere is no corresponding record for this reference.
- 9Ducháčková, L.; Roithová, J. The Interaction of Zinc(II) and Hydroxamic Acids and a Metal-Triggered Lossen Rearrangement. Chem. - Eur. J. 2009, 15 (48), 13399– 13405, DOI: 10.1002/chem.200901645Google ScholarThere is no corresponding record for this reference.
- 10Jašíková, L.; Hanikýřová, E.; Škríba, A.; Jašík, J.; Roithová, J. Metal-assisted Lossen Rearrangement. J. Org. Chem. 2012, 77 (6), 2829– 2836, DOI: 10.1021/jo300031fGoogle ScholarThere is no corresponding record for this reference.
- 11Hoshino, Y.; Okuno, M.; Kawamura, E.; Honda, K.; Inoue, S. Base-mediated Rearrangement of Free Aromatichydroxamic Acids (ArCO–NHOH) to Anilines. Chem. Commun. 2009, (17), 2281– 2283, DOI: 10.1039/b822806jGoogle ScholarThere is no corresponding record for this reference.
- 12(a) Hoshino, Y.; Shimbo, Y.; Ohtsuka, N.; Honda, K. Self-propagated Lossen Rearrangement Induced by a Catalytic Amount of Activating Agents under Mild Conditions. Tetrahedron Lett. 2015, 56 (5), 710– 712, DOI: 10.1016/j.tetlet.2014.12.084Google ScholarThere is no corresponding record for this reference.(b) Ohtsuka, N.; Okuno, M.; Hoshino, Y.; Honda, K. A Base-mediated Self-propagative Lossen Rearrangement of Hydroxamic Acids for the Efficient and Facile Synthesis of Aromatic and Aliphatic Primary Amines. Org. Biomol. Chem. 2016, 14 (38), 9046– 9054, DOI: 10.1039/C6OB01178KGoogle ScholarThere is no corresponding record for this reference.(c) Strotman, N. A.; Ortiz, A.; Savage, S. A.; Wilbert, C. R.; Ayers, S.; Kiau, S. Revisiting a Classic Transformation: A Lossen Rearrangement Initiated by Nitriles and “Pseudo-Catalytic” in Isocyanate. J. Org. Chem. 2017, 82 (8), 4044– 4049, DOI: 10.1021/acs.joc.7b00450Google ScholarThere is no corresponding record for this reference.
- 13Ortiz, A.; Soumeillant, M.; Savage, S. A.; Strotman, N. A.; Haley, M.; Benkovics, T.; Nye, J.; Xu, Z.; Tan, Y.; Ayers, S. Synthesis of HIV-Maturation Inhibitor BMS-955176 from Betulin by an Enabling Oxidation Strategy. J. Org. Chem. 2017, 82 (9), 4958– 4963, DOI: 10.1021/acs.joc.7b00438Google ScholarThere is no corresponding record for this reference.
- 14Ghosh, A. K.; Brindisi, M. Organic Carbamates in Drug Design and Medicinal Chemistry. J. Med. Chem. 2015, 58 (7), 2895– 2940, DOI: 10.1021/jm501371sGoogle ScholarThere is no corresponding record for this reference.
- 15Maisonneuve, L.; Lamarzelle, O.; Rix, E.; Grau, E.; Cramail, H. Isocyanate-Free Routes to Polyurethanes and Poly(hydroxy Urethane)s. Chem. Rev. 2015, 115 (22), 12407– 12439, DOI: 10.1021/acs.chemrev.5b00355Google ScholarThere is no corresponding record for this reference.
- 16(a) Glover, S. A.; Rauk, A.; Buccigross, J. M.; Campbell, J. J.; Hammond, G. P.; Mo, G.; Andrews, L. E.; Gillson, A.-M. E. The HERON Reaction, Origin, Theoretical Background, and Prevalence. Can. J. Chem. 2005, 83 (9), 1492– 1509, DOI: 10.1139/v05-150Google ScholarThere is no corresponding record for this reference.(b) Glover, S. A. Development of the HERON Reaction: A Historical Account. Aust. J. Chem. 2017, 70 (4), 344– 361, DOI: 10.1071/CH16683Google ScholarThere is no corresponding record for this reference.
- 17De Almeida, M. V.; Barton, D. H. R.; Bytheway, I.; Ferreira, J. A.; Hall, M. B.; Liu, W.; Taylor, D. K.; Thomson, L. Preparation and Thermal Decomposition of N,N′-Diacyl-N,N′-Dialkoxyhydrazines: Synthetic Applications and Mechanistic Insights. J. Am. Chem. Soc. 1995, 117 (17), 4870– 4874, DOI: 10.1021/ja00122a018Google ScholarThere is no corresponding record for this reference.
- 18(a) Crawford, R. J.; Raap, R. The Synthesis and Reactions of N,N′-Dicarbalkoxy-N,N′-dialkoxyhydrazines and Some Observations on Carbalkoxylium Ions. J. Org. Chem. 1963, 28 (9), 2419– 2424, DOI: 10.1021/jo01044a066Google ScholarThere is no corresponding record for this reference.(b) Cooley, J. H.; Mosher, M. W.; Khan, M. A. Preparation and Reactions of N,N′-diacyl-N,N′-dialkoxyhydrazines. J. Am. Chem. Soc. 1968, 90 (7), 1867– 1871, DOI: 10.1021/ja01009a032Google ScholarThere is no corresponding record for this reference.(c) Glover, S. A.; Mo, G.; Rauk, A. HERON Rearrangement of N,N′-diacyl-N,N′-dialkoxyhydrazines─a Theoretical and Experimental Study. Tetrahedron 1999, 55 (11), 3413– 3426, DOI: 10.1016/S0040-4020(98)01151-XGoogle ScholarThere is no corresponding record for this reference.
- 19Glover, S. A.; Mo, G. Hindered Ester Formation by SN2 Azidation of N-acetoxy-N-alkoxyamides and N-alkoxy-N-chloroamides─Novel Application of HERON Rearrangements. J. Chem. Soc., Perkin Trans. 2 2002, (10), 1728– 1739, DOI: 10.1039/B111250NGoogle ScholarThere is no corresponding record for this reference.
- 20Zhang, N.; Yang, R.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. Direct Conversion of N-Alkoxyamides to Carboxylic Esters through Tandem NBS-Mediated Oxidative Homocoupling and Thermal Denitrogenation. J. Org. Chem. 2013, 78 (17), 8705– 8711, DOI: 10.1021/jo401435vGoogle ScholarThere is no corresponding record for this reference.
- 21Duan, X.; Duan, X.; Yang, K.; Yang, K.; Wang, Z.; Wang, Z.; Zhang, L.; Zhang, L.; Tong, Y.; Tong, Y.; Liu, H.; Liu, H.; Du, K.; Du, K.; Tang, R. PhI(OAc)2/I2-Mediated Oxidative Homocoupling of N-Alkoxyamides: An Approach to Esters. Chin. J. Org. Chem. 2015, 35 (12), 2552– 2558, DOI: 10.6023/cjoc201506024Google ScholarThere is no corresponding record for this reference.
- 22Ghosh, S.; Banerjee, J.; Ghosh, R.; Chattopadhyay, S. K. A Metal-free Iodine-mediated Conversion of Hydroxamates to Esters. Synth. Commun. 2020, 50 (9), 1353– 1360, DOI: 10.1080/00397911.2020.1737130Google ScholarThere is no corresponding record for this reference.
- 23Engle, K. M.; Mei, T.-S.; Wasa, M.; Yu, J.-Q. Weak Coordination as a Powerful Means for Developing Broadly Useful C–H Functionalization Reactions. Acc. Chem. Res. 2012, 45 (6), 788– 802, DOI: 10.1021/ar200185gGoogle ScholarThere is no corresponding record for this reference.
- 24(a) Wang, D.-H.; Engle, K. M.; Shi, B.-F.; Yu, J.-Q. Ligand-Enabled Reactivity and Selectivity in a Synthetically Versatile Aryl C–H Olefination. Science 2010, 327 (5963), 315– 319, DOI: 10.1126/science.1182512Google ScholarThere is no corresponding record for this reference.(b) Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Ligand-Accelerated C–H Activation Reactions: Evidence for a Switch of Mechanism. J. Am. Chem. Soc. 2010, 132 (40), 14137– 14151, DOI: 10.1021/ja105044sGoogle ScholarThere is no corresponding record for this reference.(c) Wasa, M.; Chan, K. S. L.; Zhang, X.-G.; He, J.; Miura, M.; Yu, J.-Q. Ligand-Enabled Methylene C(sp3)–H Bond Activation with a Pd(II) Catalyst. J. Am. Chem. Soc. 2012, 134 (45), 18570– 18572, DOI: 10.1021/ja309325eGoogle ScholarThere is no corresponding record for this reference.(d) Zhu, R.-Y.; Farmer, M. E.; Chen, Y.-Q.; Yu, J.-Q. A Simple and Versatile Amide Directing Group for C–H Functionalizations. Angew. Chem., Int. Ed. 2016, 55 (36), 10578– 10599, DOI: 10.1002/anie.201600791Google ScholarThere is no corresponding record for this reference.
- 25He, J.; Li, S.; Deng, Y.; Fu, H.; Laforteza, B. N.; Spangler, J. E.; Homs, A.; Yu, J.-Q. Ligand-Controlled C(sp3)–H Arylation and Olefination in Synthesis of Unnatural Chiral Amino Acids. Science 2014, 343 (6176), 1216– 1220, DOI: 10.1126/science.1249198Google ScholarThere is no corresponding record for this reference.
- 26Chen, G.; Shigenari, T.; Jain, P.; Zhang, Z.; Jin, Z.; He, J.; Li, S.; Mapelli, C.; Miller, M. M.; Poss, M. A. Ligand-Enabled β-C–H Arylation of α-Amino Acids Using a Simple and Practical Auxiliary. J. Am. Chem. Soc. 2015, 137 (9), 3338– 3351, DOI: 10.1021/ja512690xGoogle ScholarThere is no corresponding record for this reference.
- 27Yus, M.; Radivoy, G.; Alonso, F. Lithium/DTBB-Induced Reduction of N-Alkoxyamides and Acyl Azides. Synthesis 2001, 2001, 0914– 0918, DOI: 10.1055/s-2001-13415Google ScholarThere is no corresponding record for this reference.
- 28Wang, D.-H.; Wasa, M.; Giri, R.; Yu, J.-Q. Pd(II)-Catalyzed Cross-Coupling of sp3 C–H Bonds with sp2 and sp3 Boronic Acids Using Air as the Oxidant. J. Am. Chem. Soc. 2008, 130 (23), 7190– 7191, DOI: 10.1021/ja801355sGoogle ScholarThere is no corresponding record for this reference.
- 29Wang, G.-W.; Yuan, T.-T. Palladium-Catalyzed Alkoxylation of N-Methoxybenzamides via Direct sp2 C–H Bond Activation. J. Org. Chem. 2010, 75 (2), 476– 479, DOI: 10.1021/jo902139bGoogle ScholarThere is no corresponding record for this reference.
- 30Wrigglesworth, J. W.; Cox, B.; Lloyd-Jones, G. C.; Booker-Milburn, K. I. New Heteroannulation Reactions of N-Alkoxybenzamides by Pd(II) Catalyzed C–H Activation. Org. Lett. 2011, 13 (19), 5326– 5329, DOI: 10.1021/ol202187hGoogle ScholarThere is no corresponding record for this reference.
- 31Liu, Y.-J.; Xu, H.; Kong, W.-J.; Shang, M.; Dai, H.-X.; Yu, J.-Q. Overcoming the Limitations of Directed C–H Functionalizations of Heterocycles. Nature 2014, 515 (7527), 389– 393, DOI: 10.1038/nature13885Google ScholarThere is no corresponding record for this reference.
- 32Kong, W.-J.; Liu, Y.-J.; Xu, H.; Chen, Y.-Q.; Dai, H.-X.; Yu, J.-Q. Pd-Catalyzed α-Selective C–H Functionalization of Olefins: En Route to 4-Imino-β-Lactams. J. Am. Chem. Soc. 2016, 138 (7), 2146– 2149, DOI: 10.1021/jacs.5b13353Google ScholarThere is no corresponding record for this reference.
- 33Guimond, N.; Gouliaras, C.; Fagnou, K. Rhodium(III)-Catalyzed Isoquinolone Synthesis: The N–O Bond as a Handle for C–N Bond Formation and Catalyst Turnover. J. Am. Chem. Soc. 2010, 132 (20), 6908– 6909, DOI: 10.1021/ja102571bGoogle ScholarThere is no corresponding record for this reference.
- 34Guimond, N.; Gouliaras, C.; Fagnou, K. Correction to Rhodium(III)-Catalyzed Isoquinolone Synthesis: The N–O Bond as a Handle for C–N Bond Formation and Catalyst Turnover. J. Am. Chem. Soc. 2012, 134 (43), 18148, DOI: 10.1021/ja3096625Google ScholarThere is no corresponding record for this reference.
- 35Shi, Z.; Grohmann, C.; Glorius, F. Mild Rhodium(III)-Catalyzed Cyclization of Amides with α,β-Unsaturated Aldehydes and Ketones to Azepinones: Application to the Synthesis of the Homoprotoberberine Framework. Angew. Chem., Int. Ed. 2013, 52 (20), 5393– 5397, DOI: 10.1002/anie.201301426Google ScholarThere is no corresponding record for this reference.
- 36Zeng, R.; Fu, C.; Ma, S. Highly Selective Mild Stepwise Allylation of N-Methoxybenzamides with Allenes. J. Am. Chem. Soc. 2012, 134 (23), 9597– 9600, DOI: 10.1021/ja303790sGoogle ScholarThere is no corresponding record for this reference.
- 37Wang, H.; Glorius, F. Mild Rhodium(III)-Catalyzed C–H Activation and Intermolecular Annulation with Allenes. Angew. Chem., Int. Ed. 2012, 51 (29), 7318– 7322, DOI: 10.1002/anie.201201273Google ScholarThere is no corresponding record for this reference.
- 38Zhang, S.-S.; Wu, J.-Q.; Liu, X.; Wang, H. Tandem Catalysis: Rh(III)-Catalyzed C–H Allylation/Pd(II)-Catalyzed N-Allylation Toward the Synthesis of Vinyl-Substituted N-Heterocycles. ACS Catal. 2015, 5 (1), 210– 214, DOI: 10.1021/cs501601cGoogle ScholarThere is no corresponding record for this reference.
- 39Wu, J.-Q.; Zhang, S.-S.; Gao, H.; Qi, Z.; Zhou, C.-J.; Ji, W.-W.; Liu, Y.; Chen, Y.; Li, Q.; Li, X.; Wang, H. Experimental and Theoretical Studies on Rhodium-Catalyzed Coupling of Benzamides with 2,2-Difluorovinyl Tosylate: Diverse Synthesis of Fluorinated Heterocycles. J. Am. Chem. Soc. 2017, 139 (9), 3537– 3545, DOI: 10.1021/jacs.7b00118Google ScholarThere is no corresponding record for this reference.
- 40(a) Chen, X.; Li, J.-J.; Hao, X.-S.; Goodhue, C. E.; Yu, J.-Q. Palladium-Catalyzed Alkylation of Aryl C–H Bonds with sp3 Organotin Reagents Using Benzoquinone as a Crucial Promoter. J. Am. Chem. Soc. 2006, 128 (1), 78– 79, DOI: 10.1021/ja0570943Google ScholarThere is no corresponding record for this reference.(b) Chen, X.; Goodhue, C. E.; Yu, J.-Q. Palladium-Catalyzed Alkylation of sp2 and sp3 C–H Bonds with Methylboroxine and Alkylboronic Acids: Two Distinct C–H Activation Pathways. J. Am. Chem. Soc. 2006, 128 (39), 12634– 12635, DOI: 10.1021/ja0646747Google ScholarThere is no corresponding record for this reference.(c) Giri, R.; Maugel, N.; Li, J.-J.; Wang, D.-H.; Breazzano, S. P.; Saunders, L. B.; Yu, J.-Q. Palladium-Catalyzed Methylation and Arylation of sp2 and sp3 C–H Bonds in Simple Carboxylic Acids. J. Am. Chem. Soc. 2007, 129 (12), 3510– 3511, DOI: 10.1021/ja0701614Google ScholarThere is no corresponding record for this reference.(d) Wang, D.-H.; Mei, T.-S.; Yu, J.-Q. Versatile Pd(II)-Catalyzed C–H Activation/Aryl–Aryl Coupling of Benzoic and Phenyl Acetic Acids. J. Am. Chem. Soc. 2008, 130 (52), 17676– 17677, DOI: 10.1021/ja806681zGoogle ScholarThere is no corresponding record for this reference.
- 41Karthikeyan, J.; Haridharan, R.; Cheng, C.-H. Rhodium(III)-Catalyzed Oxidative C–H Coupling of N-Methoxybenzamides with Aryl Boronic Acids: One-Pot Synthesis of Phenanthridinones. Angew. Chem., Int. Ed. 2012, 51 (49), 12343– 12347, DOI: 10.1002/anie.201206890Google ScholarThere is no corresponding record for this reference.
- 42Senthilkumar, N.; Parthasarathy, K.; Gandeepan, P.; Cheng, C.-H. Synthesis of Phenanthridinones from N-Methoxybenzamides and Aryltriethoxysilanes through Rh(III)-Catalyzed C–H and N–H Bond Activation. Chem. - Asian J. 2013, 8 (9), 2175– 2181, DOI: 10.1002/asia.201300356Google ScholarThere is no corresponding record for this reference.
- 43Prakash, S.; Muralirajan, K.; Cheng, C.-H. Rh-catalyzed Oxidizing Group-directed Ortho C–H Vinylation of Arenes by Vinylstannanes. Chem. Commun. 2015, 51 (69), 13362– 13364, DOI: 10.1039/C5CC04211AGoogle ScholarThere is no corresponding record for this reference.
- 44Li, B.; Feng, H.; Xu, S.; Wang, B. Ruthenium-Catalyzed Isoquinolone Synthesis through C–H Activation Using an Oxidizing Directing Group. Chem. - Eur. J. 2011, 17 (45), 12573– 12577, DOI: 10.1002/chem.201102445Google ScholarThere is no corresponding record for this reference.
- 45Ackermann, L.; Fenner, S. Ruthenium-Catalyzed C–H/N–O Bond Functionalization: Green Isoquinolone Syntheses in Water. Org. Lett. 2011, 13 (24), 6548– 6551, DOI: 10.1021/ol202861kGoogle ScholarThere is no corresponding record for this reference.
- 46(a) Kärkäs, M. D. Photochemical Generation of Nitrogen-Centered Amidyl, Hydrazonyl, and Imidyl Radicals: Methodology Developments and Catalytic Applications. ACS Catal. 2017, 7 (8), 4999– 5022, DOI: 10.1021/acscatal.7b01385Google ScholarThere is no corresponding record for this reference.(b) Jiang, H.; Studer, A. Chemistry With N-Centered Radicals Generated by Single-Electron Transfer-Oxidation Using Photoredox Catalysis. CCS Chem. 2019, 1 (1), 38– 49, DOI: 10.31635/ccschem.019.20180026Google ScholarThere is no corresponding record for this reference.
- 47(a) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Visible Light Photoredox-controlled Reactions of N-radicals and Radical Ions. Chem. Soc. Rev. 2016, 45 (8), 2044– 2056, DOI: 10.1039/C5CS00655DGoogle ScholarThere is no corresponding record for this reference.(b) Zard, S. Z. Recent Progress in the Generation and Use of Nitrogen-Centred Radicals. Chem. Soc. Rev. 2008, 37 (8), 1603– 1618, DOI: 10.1039/b613443mGoogle ScholarThere is no corresponding record for this reference.
- 48Boivin, J.; Callier-Dublanchet, A.-C.; Quiclet-Sire, B.; Schiano, A.-M.; Zard, S. Z. Iminyl, Amidyl, and Carbamyl Radicals from O-Benzoyl Oximes and O-Benzoyl Hydroxamic Acid Derivatives. Tetrahedron 1995, 51 (23), 6517– 6528, DOI: 10.1016/0040-4020(95)00319-4Google ScholarThere is no corresponding record for this reference.
- 49Lin, X.; Artman, G. D.; Stien, D.; Weinreb, S. M. Development of Efficient New Methodology for Generation, Cyclization and Functional Trapping of Iminyl and Amidyl radicals. Tetrahedron 2001, 57 (42), 8779– 8791, DOI: 10.1016/S0040-4020(01)00878-XGoogle ScholarThere is no corresponding record for this reference.
- 50Artman Iii, G. D.; Waldman, J. H.; Weinreb, S. M. β-Tosylethylhydroxylamine: Preparation and Use as a Hydroxylamine Equivalent in Amidyl Radical-Olefin Cyclizations. Synthesis 2002, (14), 2057– 2063, DOI: 10.1055/s-2002-34371Google ScholarThere is no corresponding record for this reference.
- 51Davies, J.; Svejstrup, T. D.; Reina, D. F.; Sheikh, N. S.; Leonori, D. Visible-Light-Mediated Synthesis of Amidyl Radicals: Transition-Metal-Free Hydroamination and N-Arylation Reactions. J. Am. Chem. Soc. 2016, 138 (26), 8092– 8095, DOI: 10.1021/jacs.6b04920Google ScholarThere is no corresponding record for this reference.
- 52Reina, D. F.; Dauncey, E. M.; Morcillo, S. P.; Svejstrup, T. D.; Popescu, M. V.; Douglas, J. J.; Sheikh, N. S.; Leonori, D. Visible-Light-Mediated 5-exo-dig Cyclizations of Amidyl Radicals. Eur. J. Org. Chem. 2017, 2017 (15), 2108– 2111, DOI: 10.1002/ejoc.201601607Google ScholarThere is no corresponding record for this reference.
- 53Jung, M. E.; Piizzi, G. gem-Disubstituent Effect: Theoretical Basis and Synthetic Applications. Chem. Rev. 2005, 105 (5), 1735– 1766, DOI: 10.1021/cr940337hGoogle ScholarThere is no corresponding record for this reference.
- 54Wu, K.; Du, Y.; Wang, T. Visible-Light-Mediated Construction of Pyrroloindolines via an Amidyl Radical Cyclization/Carbon Radical Addition Cascade: Rapid Synthesis of (±)-Flustramide B. Org. Lett. 2017, 19 (20), 5669– 5672, DOI: 10.1021/acs.orglett.7b02837Google ScholarThere is no corresponding record for this reference.
- 55Wu, K.; Du, Y.; Wei, Z.; Wang, T. Synthesis of Functionalized Pyrroloindolines via a Visible-Light-Induced Radical Cascade Reaction: Rapid Synthesis of (±)-Flustraminol B. Chem. Commun. 2018, 54 (54), 7443– 7446, DOI: 10.1039/C8CC03575JGoogle ScholarThere is no corresponding record for this reference.
- 56(a) Vitaku, E.; Smith, D. T.; Njardarson, J. T. Analysis of the Structural Diversity, Substitution Patterns, and Frequency of Nitrogen Heterocycles among U.S. FDA Approved Pharmaceuticals. J. Med. Chem. 2014, 57 (24), 10257– 10274, DOI: 10.1021/jm501100bGoogle ScholarThere is no corresponding record for this reference.(b) Shi, Q.; Huang, X.; Yang, R.; Liu, W. H. Unified Ionic and Radical C-4 Alkylation and Arylation of Pyridines. Chem. Sci. 2024, 15 (31), 12442– 12450, DOI: 10.1039/D4SC03739AGoogle ScholarThere is no corresponding record for this reference.(c) Huang, X.; Shi, Q.; Lai, G.; Liu, W. H. 1,4-Dearomatization of Pyridines and Quinolines Enabled by the Carbodication Lewis Acid. Angew. Chem., Int. Ed. 2025, 64 (37), e202510385 DOI: 10.1002/anie.202510385Google ScholarThere is no corresponding record for this reference.
- 57Bull, J. A.; Mousseau, J. J.; Pelletier, G.; Charette, A. B. Synthesis of Pyridine and Dihydropyridine Derivatives by Regio- and Stereoselective Addition to N-Activated Pyridines. Chem. Rev. 2012, 112 (5), 2642– 2713, DOI: 10.1021/cr200251dGoogle ScholarThere is no corresponding record for this reference.
- 58(a) Yu, X.-Y.; Zhao, Q.-Q.; Chen, J.; Xiao, W.-J.; Chen, J.-R. When Light Meets Nitrogen-Centered Radicals: From Reagents to Catalysts. Acc. Chem. Res. 2020, 53 (5), 1066– 1083, DOI: 10.1021/acs.accounts.0c00090Google ScholarThere is no corresponding record for this reference.(b) Davies, J.; Morcillo, S. P.; Douglas, J. J.; Leonori, D. Hydroxylamine Derivatives as Nitrogen-Radical Precursors in Visible-Light Photochemistry. Chem. - Eur. J. 2018, 24 (47), 12154– 12163, DOI: 10.1002/chem.201801655Google ScholarThere is no corresponding record for this reference.
- 59Feng, M.; Zhang, H.; Maulide, N. Challenges and Breakthroughs in Selective Amide Activation. Angew. Chem., Int. Ed. 2022, 61 (49), e202212213 DOI: 10.1002/anie.202212213Google ScholarThere is no corresponding record for this reference.
- 60Kweon, B.; Kim, C.; Kim, S.; Hong, S. Remote C–H Pyridylation of Hydroxamates through Direct Photoexcitation of O-Aryl Oxime Pyridinium Intermediates. Angew. Chem., Int. Ed. 2021, 60 (51), 26813– 26821, DOI: 10.1002/anie.202112364Google ScholarThere is no corresponding record for this reference.
- 61(a) Murphy, J. A. Discovery and Development of Organic Super-Electron-Donors. J. Org. Chem. 2014, 79 (9), 3731– 3746, DOI: 10.1021/jo500071uGoogle ScholarThere is no corresponding record for this reference.(b) Barham, J. P.; Coulthard, G.; Emery, K. J.; Doni, E.; Cumine, F.; Nocera, G.; John, M. P.; Berlouis, L. E. A.; McGuire, T.; Tuttle, T.; Murphy, J. A. KOtBu: A Privileged Reagent for Electron Transfer Reactions?. J. Am. Chem. Soc. 2016, 138 (23), 7402– 7410, DOI: 10.1021/jacs.6b03282Google ScholarThere is no corresponding record for this reference.(c) Barham, J. P.; Dalton, S. E.; Allison, M.; Nocera, G.; Young, A.; John, M. P.; McGuire, T.; Campos, S.; Tuttle, T.; Murphy, J. A. Dual Roles for Potassium Hydride in Haloarene Reduction: CSNAr and Single Electron Transfer Reduction via Organic Electron Donors Formed in Benzene. J. Am. Chem. Soc. 2018, 140 (36), 11510– 11518, DOI: 10.1021/jacs.8b07632Google ScholarThere is no corresponding record for this reference.
- 62(a) Li, M.; Berritt, S.; Matuszewski, L.; Deng, G.; Pascual-Escudero, A.; Panetti, G. B.; Poznik, M.; Yang, X.; Chruma, J. J.; Walsh, P. J. Transition-Metal-Free Radical C(sp3)–C(sp2) and C(sp3)–C(sp3) Coupling Enabled by 2-Azaallyls as Super-Electron-Donors and Coupling-Partners. J. Am. Chem. Soc. 2017, 139 (45), 16327– 16333, DOI: 10.1021/jacs.7b09394Google ScholarThere is no corresponding record for this reference.(b) Li, M.; Gutierrez, O.; Berritt, S.; Pascual-Escudero, A.; Yeşilçimen, A.; Yang, X.; Adrio, J.; Huang, G.; Nakamaru-Ogiso, E.; Kozlowski, M. C.; Walsh, P. J. Transition-metal-free Chemo- and Regioselective Vinylation of Azaallyls. Nat. Chem. 2017, 9 (10), 997– 1004, DOI: 10.1038/nchem.2760Google ScholarThere is no corresponding record for this reference.(c) Zhang, L.; Liu, Z.; Tian, X.; Zi, Y.; Duan, S.; Fang, Y.; Chen, W.; Jing, H.; Yang, L.; Yang, X. Transition-Metal-Free C(sp3)–H Coupling of Cycloalkanes Enabled by Single-Electron Transfer and Hydrogen Atom Transfer. Org. Lett. 2021, 23 (5), 1714– 1719, DOI: 10.1021/acs.orglett.1c00135Google ScholarThere is no corresponding record for this reference.(d) Liu, Z.; Li, M.; Deng, G.; Wei, W.; Feng, P.; Zi, Q.; Li, T.; Zhang, H.; Yang, X.; Walsh, P. J. Transition-metal-free C(sp3)–H/C(sp3)–H Dehydrogenative Coupling of Saturated Heterocycles with N-benzyl Imines. Chem. Sci. 2020, 11 (29), 7619– 7625, DOI: 10.1039/D0SC00031KGoogle ScholarThere is no corresponding record for this reference.
- 63(a) Lei, Y.; Yang, J.; Qi, R.; Wang, S.; Wang, R.; Xu, Z. Arylation of Benzyl Amines with Aromatic Nitriles. Chem. Commun. 2018, 54 (84), 11881– 11884, DOI: 10.1039/C8CC06408CGoogle ScholarThere is no corresponding record for this reference.(b) Tang, S.; Zhang, X.; Sun, J.; Niu, D.; Chruma, J. J. 2-Azaallyl Anions, 2-Azaallyl Cations, 2-Azaallyl Radicals, and Azomethine Ylides. Chem. Rev. 2018, 118 (20), 10393– 10457, DOI: 10.1021/acs.chemrev.8b00349Google ScholarThere is no corresponding record for this reference.(c) Wang, Q.; Poznik, M.; Li, M.; Walsh, P. J.; Chruma, J. J. 2-Azaallyl Anions as Light-Tunable Super-Electron-Donors: Coupling with Aryl Fluorides, Chlorides, and Bromides. Adv. Synth. Catal. 2018, 360 (15), 2854– 2868, DOI: 10.1002/adsc.201800396Google ScholarThere is no corresponding record for this reference.
- 64Jiang, Y.; Liu, D.; Rotella, M. E.; Deng, G.; Liu, Z.; Chen, W.; Zhang, H.; Kozlowski, M. C.; Walsh, P. J.; Yang, X. Net-1,2-Hydrogen Atom Transfer of Amidyl Radicals: Toward the Synthesis of 1,2-Diamine Derivatives. J. Am. Chem. Soc. 2023, 145 (29), 16045– 16057, DOI: 10.1021/jacs.3c04376Google ScholarThere is no corresponding record for this reference.
- 65Ye, Y.; Cao, J.; Oblinsky, D. G.; Verma, D.; Prier, C. K.; Scholes, G. D.; Hyster, T. K. Using Enzymes to Tame Nitrogen-Centred Radicals for Enantioselective Hydroamination. Nat. Chem. 2023, 15 (2), 206– 212, DOI: 10.1038/s41557-022-01083-zGoogle ScholarThere is no corresponding record for this reference.
- 66Marshall, C. M.; Federice, J. G.; Bell, C. N.; Cox, P. B.; Njardarson, J. T. An Update on the Nitrogen Heterocycle Compositions and Properties of U.S. FDA-Approved Pharmaceuticals (2013–2023). J. Med. Chem. 2024, 67 (14), 11622– 11655, DOI: 10.1021/acs.jmedchem.4c01122Google ScholarThere is no corresponding record for this reference.
- 67(a) Nesterov, V.; Reiter, D.; Bag, P.; Frisch, P.; Holzner, R.; Porzelt, A.; Inoue, S. NHCs in Main Group Chemistry. Chem. Rev. 2018, 118 (19), 9678– 9842, DOI: 10.1021/acs.chemrev.8b00079Google ScholarThere is no corresponding record for this reference.(b) Peris, E. Smart N-Heterocyclic Carbene Ligands in Catalysis. Chem. Rev. 2018, 118 (19), 9988– 10031, DOI: 10.1021/acs.chemrev.6b00695Google ScholarThere is no corresponding record for this reference.
- 68Ye, Q.; Neo, W. T.; Lin, T.; Song, J.; Yan, H.; Zhou, H.; Shah, K. W.; Chua, S. J.; Xu, J. Pyrrolophthalazine Dione (PPD)-Based Donor–Acceptor Polymers as High Performance Electrochromic Materials. Polym. Chem. 2015, 6 (9), 1487– 1494, DOI: 10.1039/C4PY01608DGoogle ScholarThere is no corresponding record for this reference.
- 69(a) Deiters, A.; Martin, S. F. Synthesis of Oxygen- and Nitrogen-Containing Heterocycles by Ring-Closing Metathesis. Chem. Rev. 2004, 104 (5), 2199– 2238, DOI: 10.1021/cr0200872Google ScholarThere is no corresponding record for this reference.(b) Patil, N. T.; Yamamoto, Y. Coinage Metal-Assisted Synthesis of Heterocycles. Chem. Rev. 2008, 108 (8), 3395– 3442, DOI: 10.1021/cr050041jGoogle ScholarThere is no corresponding record for this reference.(c) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Formal [4 + 1] Annulation Reactions in the Synthesis of Carbocyclic and Heterocyclic Systems. Chem. Rev. 2015, 115 (11), 5301– 5365, DOI: 10.1021/cr5006974Google ScholarThere is no corresponding record for this reference.
- 70Deeksha; Singh, R. Aza-oxyallyl Cations and Their Applications in (3 m) Cycloaddition Reactions. Eur. J. Org. Chem. 2022, 2022 (47), e202201043 DOI: 10.1002/ejoc.202201043Google ScholarThere is no corresponding record for this reference.
- 71Lengyel, I.; Sheehan, J. C. α-Lactams (Aziridinones). Angew. Chem., Int. Ed. 1968, 7 (1), 25– 36, DOI: 10.1002/anie.196800251Google ScholarThere is no corresponding record for this reference.
- 72Kikugawa, Y.; Shimada, M.; Kato, M.; Sakamoto, T. A New Synthesis of N-Alkoxy-2-ethoxyarylacetamides from N-Alkoxy-N-chloroarylacetamides with Triethylamine in Ethanol. Chem. Pharm. Bull. 1993, 41 (12), 2192– 2194, DOI: 10.1248/cpb.41.2192Google ScholarThere is no corresponding record for this reference.
- 73Jeffrey, C. S.; Barnes, K. L.; Eickhoff, J. A.; Carson, C. R. Generation and Reactivity of Aza-Oxyallyl Cationic Intermediates: Aza-[4 + 3] Cycloaddition Reactions for Heterocycle Synthesis. J. Am. Chem. Soc. 2011, 133 (20), 7688– 7691, DOI: 10.1021/ja201901dGoogle ScholarThere is no corresponding record for this reference.
- 74Acharya, A.; Eickhoff, J. A.; Jeffrey, C. S. Intramolecular Aza-[4 + 3] Cycloaddition Reactions of α-Halohydroxamates. Synthesis 2013, 45 (13), 1825– 1836, DOI: 10.1055/s-0033-1338883Google ScholarThere is no corresponding record for this reference.
- 75(a) Xuan, J.; Cao, X.; Cheng, X. Advances in Heterocycle Synthesis via [3+m]-Cycloaddition Reactions Involving an Azaoxyallyl Cation as the key Intermediate. Chem. Commun. 2018, 54 (41), 5154– 5163, DOI: 10.1039/C8CC00787JGoogle ScholarThere is no corresponding record for this reference.(b) Fantinati, A.; Zanirato, V.; Marchetti, P.; Trapella, C. The Fascinating Chemistry of α-Haloamides. ChemistryOpen 2020, 9 (2), 100– 170, DOI: 10.1002/open.201900220Google ScholarThere is no corresponding record for this reference.(c) El Bouakher, A.; Martel, A.; Comesse, S. α-Halogenoacetamides: Versatile and Efficient Tools for the Synthesis of Complex Aza-heterocycles. Org. Biomol. Chem. 2019, 17 (37), 8467– 8485, DOI: 10.1039/C9OB01683JGoogle ScholarThere is no corresponding record for this reference.
- 76Ansari, A. J.; Yadav, A.; Mukherjee, A.; Sathish, E.; Nagesh, K.; Singh, R. Metal Free Amination of Congested and Functionalized Alkyl Bromides at Room Temperature. Chem. Commun. 2020, 56 (35), 4804– 4807, DOI: 10.1039/D0CC00826EGoogle ScholarThere is no corresponding record for this reference.
- 77Kwon, Y.; Choi, S.; Jang, H. S.; Kim, S.-G. Rapid Access to Hindered α-Amino Acid Derivatives and Benzodiazepin-3-ones from Aza-Oxyallyl Cations. Org. Lett. 2020, 22 (4), 1420– 1425, DOI: 10.1021/acs.orglett.0c00023Google ScholarThere is no corresponding record for this reference.
- 78Lee, C. Y.; Kim, S.-G. Metal-free Nucleophilic α-Azidation of α-Halohydroxamates with Azidotrimethylsilane. Asian J. Org. Chem. 2021, 10 (12), 3257– 3260, DOI: 10.1002/ajoc.202100556Google ScholarThere is no corresponding record for this reference.
- 79Chae Son, E.; Kim, S.-G. Metal-free Nucleophilic Alkoxylation of in Situ-Generated Azaoxyallyl Cations: Synthesis of Hindered Dialkyl Ether Derivatives. Asian J. Org. Chem. 2020, 9 (6), 914– 917, DOI: 10.1002/ajoc.202000173Google ScholarThere is no corresponding record for this reference.
- 80Yoon Lee, C.; Kim, S.-G. Highly Efficient DMSO-Promoted α-Hydrolysis of α-Halohydroxamates under Mild Conditions. Eur. J. Org. Chem. 2021, 2021 (10), 1607– 1614, DOI: 10.1002/ejoc.202100088Google ScholarThere is no corresponding record for this reference.
- 81Taily, I. M.; Saha, D.; Banerjee, P. Aza-Oxyallyl Cation Driven 3-Amido Oxetane Rearrangement to 2-Oxazolines: Access to Oxazoline Amide Ethers. J. Org. Chem. 2022, 87 (5), 2155– 2166, DOI: 10.1021/acs.joc.1c03108Google ScholarThere is no corresponding record for this reference.
- 82Bera, T.; Singh, B.; Jana, M.; Saha, J. Access to 3,3′-Disubstituted Peroxyoxindole Derivatives and α-Peroxyamides via Azaoxyallyl Cation-guided Addition of Hydroperoxides. Chem. Commun. 2022, 58 (54), 7538– 7541, DOI: 10.1039/D2CC02378DGoogle ScholarThere is no corresponding record for this reference.
- 83Deeksha; Sathish, E.; Kiran; Singh, R. Access to Sterically Hindered Thioethers (α-Thioamides) Under Mild Conditions Using α-Halohydroxamates: Application toward 1,4-Benzothiazinones and 4,1-Benzothiazepinones. J. Org. Chem. 2023, 88 (2), 901– 908, DOI: 10.1021/acs.joc.2c02274Google ScholarThere is no corresponding record for this reference.
- 84Gao, J.; Hu, M.; Wang, Y.; Luo, S.; Mi, X. Transition-Metal-Free Direct α-Arylation of Weinreb-type Amides with Arylboronic Acids through Aza-oxyallyl Cation Intermediates. J. Org. Chem. 2024, 89 (18), 13730– 13734, DOI: 10.1021/acs.joc.4c01391Google ScholarThere is no corresponding record for this reference.
- 85Graham, S. L.; Scholz, T. H. A New Mode of Reactivity of N-methoxy-N-methylamides with Strongly Basic Reagents. Tetrahedron Lett. 1990, 31 (44), 6269– 6272, DOI: 10.1016/S0040-4039(00)97039-4Google ScholarThere is no corresponding record for this reference.
- 86Keck, G. E.; McHardy, S. F.; Murry, J. A. Some Unusual Reactions of Weinreb Amides. Tetrahedron Lett. 1993, 34 (39), 6215– 6218, DOI: 10.1016/S0040-4039(00)73713-0Google ScholarThere is no corresponding record for this reference.
- 87Mattingly, P. G.; Miller, M. J. Titanium Trichloride Reduction of Substituted N-hydroxy-2-azetidinones and Other Hydroxamic Acids. J. Org. Chem. 1980, 45 (3), 410– 415, DOI: 10.1021/jo01291a007Google ScholarThere is no corresponding record for this reference.
- 88Ritter, A. R.; Miller, M. J. Amino Acid-Derived Chiral Acyl Nitroso Compounds: Diastereoselectivity in Intermolecular Hetero Diels-Alder Reactions. J. Org. Chem. 1994, 59 (16), 4602– 4611, DOI: 10.1021/jo00095a040Google ScholarThere is no corresponding record for this reference.
- 89Keck, G. E.; Wager, T. T.; McHardy, S. F. Reductive Cleavage of N–O Bonds in Hydroxylamines and Hydroxamic acid Derivatives Using Samarium Diiodide. Tetrahedron 1999, 55 (40), 11755– 11772, DOI: 10.1016/S0040-4020(99)00486-XGoogle ScholarThere is no corresponding record for this reference.
- 90Fukuzawa, H.; Ura, Y.; Kataoka, Y. Ruthenium-Catalyzed Reduction of N-alkoxy- and N-hydroxyamides. J. Organomet. Chem. 2011, 696 (23), 3643– 3648, DOI: 10.1016/j.jorganchem.2011.08.026Google ScholarThere is no corresponding record for this reference.
- 91Wang, S.; Zhao, X.; Zhang-Negrerie, D.; Du, Y. Reductive Cleavage of the N–O Bond: Elemental Sulfur-Mediated Conversion of N-alkoxyamides to Amides. Org. Chem. Front. 2019, 6 (3), 347– 351, DOI: 10.1039/C8QO01052HGoogle ScholarThere is no corresponding record for this reference.
- 92Cutulic, S. P. Y.; Murphy, J. A.; Farwaha, H.; Zhou, S.-Z.; Chrystal, E. Metal-Free Reductive Cleavage of N–O Bonds in Weinreb Amides by an Organic Neutral Super-Electron Donor. Synlett 2008, 2008 (14), 2132– 2136, DOI: 10.1055/s-2008-1078240Google ScholarThere is no corresponding record for this reference.
- 93(a) Shi, Q.; Liu, W. H. Reactivity Umpolung of Tertiary Amide Enabled by Catalytic Reductive Stannylation. Angew. Chem., Int. Ed. 2023, 62 (37), e202309567 DOI: 10.1002/anie.202309567Google ScholarThere is no corresponding record for this reference.(b) Shi, Q.; Liu, W. H. Reactivity Umpolung of Amides in Organic Synthesis. Asian J. Org. Chem. 2023, 12 (12), e202300473 DOI: 10.1002/ajoc.202300473Google ScholarThere is no corresponding record for this reference.(c) Seebach, D.; Corey, E. J. Generation and Synthetic Applications of 2-Lithio-1,3-dithianes. J. Org. Chem. 1975, 40 (2), 231– 237, DOI: 10.1021/jo00890a018Google ScholarThere is no corresponding record for this reference.
- 94(a) Wu, Y.; Hu, L.; Li, Z.; Deng, L. Catalytic Asymmetric Umpolung Reactions of Imines. Nature 2015, 523 (7561), 445– 450, DOI: 10.1038/nature14617Google ScholarThere is no corresponding record for this reference.(b) Miyoshi, T.; Miyakawa, T.; Ueda, M.; Miyata, O. Nucleophilic α-Arylation and α-Alkylation of Ketones by Polarity Inversion of N-Alkoxyenamines: Entry to the Umpolung Reaction at the α-Carbon Position of Carbonyl Compounds. Angew. Chem., Int. Ed. 2011, 50 (4), 928– 931, DOI: 10.1002/anie.201004374Google ScholarThere is no corresponding record for this reference.(c) Quiclet-Sire, B.; Tölle, N.; Zafar, S. N.; Zard, S. Z. Oxime Derivatives as α-Electrophiles. From α-Tetralone Oximes to Tetracyclic Frameworks. Org. Lett. 2011, 13 (12), 3266– 3269, DOI: 10.1021/ol2012204Google ScholarThere is no corresponding record for this reference.(d) Sukhorukov, A. Y. Umpolung of Enamines: An Overview on Strategies and Synthons. Synlett 2020, 31 (05), 439– 449, DOI: 10.1055/s-0037-1610741Google ScholarThere is no corresponding record for this reference.
- 95Dai, X.-J.; Li, C.-C.; Li, C.-J. Carbonyl Umpolung as an Organometallic Reagent Surrogate. Chem. Soc. Rev. 2021, 50 (19), 10733– 10742, DOI: 10.1039/D1CS00418BGoogle ScholarThere is no corresponding record for this reference.
- 96(a) Trost, B. M.; Li, C.-J. Novel “Umpolung” in C–C Bond Formation Catalyzed by Triphenylphosphine. J. Am. Chem. Soc. 1994, 116 (7), 3167– 3168, DOI: 10.1021/ja00086a074Google ScholarThere is no corresponding record for this reference.(b) Guo, H.; Fan, Y. C.; Sun, Z.; Wu, Y.; Kwon, O. Phosphine Organocatalysis. Chem. Rev. 2018, 118 (20), 10049– 10293, DOI: 10.1021/acs.chemrev.8b00081Google ScholarThere is no corresponding record for this reference.
- 97Bugaut, X.; Glorius, F. Organocatalytic Umpolung: N-heterocyclic Carbenes and Beyond. Chem. Soc. Rev. 2012, 41 (9), 3511– 3522, DOI: 10.1039/c2cs15333eGoogle ScholarThere is no corresponding record for this reference.
- 98Hari, D. P.; Caramenti, P.; Waser, J. Cyclic Hypervalent Iodine Reagents: Enabling Tools for Bond Disconnection via Reactivity Umpolung. Acc. Chem. Res. 2018, 51 (12), 3212– 3225, DOI: 10.1021/acs.accounts.8b00468Google ScholarThere is no corresponding record for this reference.
- 99(a) Masson, G.; Py, S.; Vallée, Y. Samarium Diiodide-Induced Reductive Cross-Coupling of Nitrones with Aldehydes and Ketones. Angew. Chem., Int. Ed. 2002, 41 (10), 1772– 1775, DOI: 10.1002/1521-3773(20020517)41:10<1772::AID-ANIE1772>3.0.CO;2-QGoogle ScholarThere is no corresponding record for this reference.(b) Masson, G.; Cividino, P.; Py, S.; Vallée, Y. SmI2-Induced Umpolung of the C–N Bond: First Reductive Conjugate Addition of Nitrones to α,β-Unsaturated Esters. Angew. Chem., Int. Ed. 2003, 42 (20), 2265– 2268, DOI: 10.1002/anie.200250480Google ScholarThere is no corresponding record for this reference.(c) Riber, D.; Skrydstrup, T. SmI2-Promoted Radical Addition of Nitrones to α,β-Unsaturated Amides and Esters: Synthesis of γ-Amino Acids via a Nitrogen Equivalent to the Ketyl Radical. Org. Lett. 2003, 5 (2), 229– 231, DOI: 10.1021/ol027386yGoogle ScholarThere is no corresponding record for this reference.
- 100(a) Kaiser, D.; Bauer, A.; Lemmerer, M.; Maulide, N. Amide Activation: an Emerging Tool for Chemoselective Synthesis. Chem. Soc. Rev. 2018, 47 (21), 7899– 7925, DOI: 10.1039/C8CS00335AGoogle ScholarThere is no corresponding record for this reference.(b) Spieß, P.; Shaaban, S.; Kaiser, D.; Maulide, N. New Strategies for the Functionalization of Carbonyl Derivatives via α-Umpolung: From Enolates to Enolonium Ions. Acc. Chem. Res. 2023, 56 (12), 1634– 1644, DOI: 10.1021/acs.accounts.3c00171Google ScholarThere is no corresponding record for this reference.
- 101Bladon, C. M.; Kirby, G. W. Formation of Aziridinones (α-lactams) from Hydroxamic Derivatives. J. Chem. Soc., Chem. Commun. 1982, (24), 1402– 1404, DOI: 10.1039/C39820001402Google ScholarThere is no corresponding record for this reference.
- 102(a) Hoffman, R. V.; Nayyar, N. K.; Klinekole, B. W. Efficient Conversion of O-sulfonylated Arylacetohydroxamic Acids to 2-Substituted Secondary Amides. J. Am. Chem. Soc. 1992, 114 (15), 6262– 6263, DOI: 10.1021/ja00041a064Google ScholarThere is no corresponding record for this reference.(b) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Base-promoted Reaction of O-sulfonylated Hydroxamic Acids with Nucleophiles. A New Method for the Synthesis of alpha-Substituted Amides. J. Org. Chem. 1992, 57 (21), 5700– 5707, DOI: 10.1021/jo00047a024Google ScholarThere is no corresponding record for this reference.(c) Hoffman, R. V.; Nayyar, N. K. Preparation of alpha-Alkoxy Amides and alpha-Hydrazino Amides by Base-Promoted Reactions of N-Sulfonyloxy Amides. J. Org. Chem. 1995, 60 (21), 7043– 7046, DOI: 10.1021/jo00126a072Google ScholarThere is no corresponding record for this reference.(d) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Synthesis of alpha-Azido and alpha-Amino Amides from N-[(methylsulfonyl)oxy]amides. J. Org. Chem. 1993, 58 (9), 2355– 2359, DOI: 10.1021/jo00061a004Google ScholarThere is no corresponding record for this reference.(e) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Origins of Regioselectivity in the Reactions of alpha-Lactams with Nucleophiles. J. Org. Chem. 1995, 60 (13), 4121– 4125, DOI: 10.1021/jo00118a032Google ScholarThere is no corresponding record for this reference.(f) Hoffman, R. V.; Nayyar, N. K. Reaction of Hydrazines with alpha-Lactams for the Preparation of 1,2,4-Triazine-3,6-diones and Aza-Urea Peptide Mimetics. J. Org. Chem. 1995, 60 (18), 5992– 5994, DOI: 10.1021/jo00123a048Google ScholarThere is no corresponding record for this reference.
- 103Hoffman, R. V.; Zhao, Z.; Costales, A.; Clarke, D. Origins of Regioselectivity in the Reactions of α-Lactams with Nucleophiles. Two Distinct Acid-Catalyzed Pathways Involving O- and N-Protonation. J. Org. Chem. 2002, 67 (15), 5284– 5294, DOI: 10.1021/jo020246hGoogle ScholarThere is no corresponding record for this reference.
- 104Han, S.; Guo, Y.; Zhang, W.; Chen, J.; Wang, Z.; Zeng, Y.-F. Regioselective Heterofunctionalization of alpha-Aryl Amides with Heteroatom Nucleophiles via Electrophilic Activation. Org. Chem. Front. 2023, 10 (21), 5512– 5518, DOI: 10.1039/D3QO01174GGoogle ScholarThere is no corresponding record for this reference.
- 105Zhang, W.; Han, S.; Zeng, Y.-F.; Han, F. Metal-Free α-(Hetero)Arylation of Amides Enabled Access to Diaryl Acetamides via Umpolung. Eur. J. Org. Chem. 2024, 27 (47), e202400905 DOI: 10.1002/ejoc.202400905Google ScholarThere is no corresponding record for this reference.
- 106Wang, R.; An, S.; Xin, Y.-X.; Jiang, Y.-Y.; Liu, W. H. Redox-Neutral Umpolung Synthesis of α-Functionalized Amides. JACS Au 2024, 4 (11), 4435– 4444, DOI: 10.1021/jacsau.4c00767Google ScholarThere is no corresponding record for this reference.
- 107An, S.; Lai, G.; Liu, W. H. Catalytic Dehydrogenative Synthesis of α,β-Unsaturated Secondary Amides without External Oxidants. Chem. Sci. 2024, 15 (37), 15385– 15390, DOI: 10.1039/D4SC04419CGoogle ScholarThere is no corresponding record for this reference.
- 108Wang, R.; Jiang, Q.; Jiang, L.; Liu, W. H. Nucleophilic α- and β-Additions Enable Redox-Neutral Aziridination of Conjugated Hydroxamates. J. Am. Chem. Soc. 2025, 147 (30), 26298– 26306, DOI: 10.1021/jacs.5c04286Google ScholarThere is no corresponding record for this reference.
- 109Zhang, W.; Han, S.; Zhang, S.-Y.; Jiang, D.-X.; Chen, J.; Wang, Z.; Zeng, Y.-F. Synthesis of Indolyl Pyrroloindolines via a Cascade Arylation/Cyclization of Indole Acetamides with 3-Substituted Indoles. Org. Chem. Front. 2024, 11 (18), 5167– 5171, DOI: 10.1039/D4QO01118JGoogle ScholarThere is no corresponding record for this reference.
- 110Tang, X.; Zhang, W.; Jiang, D.-X.; Chen, Z.; Shen, L.; Tian, Y.; Chen, J.; Wang, Z.; Zeng, Y.-F. Access to Divergent Indolyl Pyrrolones via [3 + 2] Annulation of Indolyl Hydroxamates with Phosphonium Ylides. Org. Lett. 2025, 27 (43), 12137– 12141, DOI: 10.1021/acs.orglett.5c03964Google ScholarThere is no corresponding record for this reference.
- 111Hirner, S.; Panknin, O.; Edefuhr, M.; Somfai, P. Synthesis of Aryl Glycines by the α Arylation of Weinreb Amides. Angew. Chem., Int. Ed. 2008, 47 (10), 1907– 1909, DOI: 10.1002/anie.200704689Google ScholarThere is no corresponding record for this reference.
- 112Hirner, S.; Kirchner, D. K.; Somfai, P. Synthesis of α-Amino Acids by Umpolung of Weinreb Amide Enolates. Eur. J. Org. Chem. 2008, 2008 (33), 5583– 5589, DOI: 10.1002/ejoc.200800683Google ScholarThere is no corresponding record for this reference.
- 113Takeda, N.; Futaki, E.; Kobori, Y.; Ueda, M.; Miyata, O. Nucleophilic Arylation of N,O-Ketene Acetals with Triaryl Aluminum Reagents: Access to α-Aryl Amides through an Umpolung Process. Angew. Chem., Int. Ed. 2017, 56 (51), 16342– 16346, DOI: 10.1002/anie.201708665Google ScholarThere is no corresponding record for this reference.
- 114Gassman, P. G. Nitrenium ions. Acc. Chem. Res. 1970, 3 (1), 26– 33, DOI: 10.1021/ar50025a004Google ScholarThere is no corresponding record for this reference.
- 115Novak, M.; Xu, L.; Wolf, R. A. Nitrenium Ions from Food-Derived Heterocyclic Arylamine Mutagens. J. Am. Chem. Soc. 1998, 120 (7), 1643– 1644, DOI: 10.1021/ja974222hGoogle ScholarThere is no corresponding record for this reference.
- 116O’Neil, L. G.; Bower, J. F. Electrophilic Aminating Agents in Total Synthesis. Angew. Chem., Int. Ed. 2021, 60 (49), 25640– 25666, DOI: 10.1002/anie.202102864Google ScholarThere is no corresponding record for this reference.
- 117(a) Kawase, M.; Kitamura, T.; Kikugawa, Y. Electrophilic Aromatic Substitution with N-Methoxy-N-acylnitrenium Ions Generated from N-Chloro-N-methoxy Amides: Syntheses of Nitrogen Heterocyclic Compounds Bearing an N-Methoxy Amide Group. J. Org. Chem. 1989, 54 (14), 3394– 3403, DOI: 10.1021/jo00275a027Google ScholarThere is no corresponding record for this reference.(b) Glover, S. A.; Goosen, A.; McClei, C. V.; Schoonraad, J. L. N-Alkoxy-N-acylnitrenium Ions in Intramolecular Aromatic Addition Reactions. Tetrahedron 1987, 43 (11), 2577– 2592, DOI: 10.1016/S0040-4020(01)81665-3Google ScholarThere is no corresponding record for this reference.
- 118Sakamoto, R.; Inada, T.; Selvakumar, S. Efficient Photolytic C–H Bond Functionalization of Alkylbenzene with Hypervalent Iodine(iii) Reagent. Chem. Commun. 2016, 52 (19), 3758– 3761, DOI: 10.1039/C5CC07647AGoogle ScholarThere is no corresponding record for this reference.
- 119Maiti, S.; Alam, M. T.; Bal, A.; Mal, P. Nitrenium Ions from Amine-Iodine(III) Combinations. Adv. Synth. Catal. 2019, 361 (19), 4401– 4425, DOI: 10.1002/adsc.201900441Google ScholarThere is no corresponding record for this reference.
- 120Glover, S. A.; Scott, A. P. MNDO Properties of Heteroatom and Phenyl Substituted Nitrenium Ions. Tetrahedron 1989, 45 (6), 1763– 1776, DOI: 10.1016/S0040-4020(01)80041-7Google ScholarThere is no corresponding record for this reference.
- 121Kikugawa, Y.; Kawase, M. An Electrophilic Aromatic Substitution by N-Methoxyamides via Hypervalent Iodine Intermediates. Chem. Lett. 1990, 19 (4), 581– 582, DOI: 10.1246/cl.1990.581Google ScholarThere is no corresponding record for this reference.
- 122Miyazawa, E.; Sakamoto, T.; Kikugawa, Y. Synthesis of Spiro-fused Nitrogen Heterocyclic Compounds via N-Methoxy-N-acylnitrenium Ions Using Phenyliodine (III) bis (trifluoroacetate) in Trifluoroethanol. Heterocycles 2003, 59 (1), 149– 160, DOI: 10.3987/COM-02-S7Google ScholarThere is no corresponding record for this reference.
- 123Zhu, C.; Liang, Y.; Hong, X. Iodoarene-Catalyzed Stereospecific Intramolecular sp3 C–H Amination: Reaction Development and Mechanistic Insights. J. Am. Chem. Soc. 2015, 137 (24), 7564– 7567, DOI: 10.1021/jacs.5b03488Google ScholarThere is no corresponding record for this reference.
- 124Wang, Y.; Yang, M.; Sun, Y.-Y. An Efficient Approach for 3,3-Disubstituted Oxindoles Synthesis: Aryl Iodine Catalyzed Intramolecular C–N Bond Oxidative Cross-Coupling. Org. Lett. 2021, 23 (22), 8750– 8754, DOI: 10.1021/acs.orglett.1c03224Google ScholarThere is no corresponding record for this reference.
- 125Das, T. K.; Ghosh, A.; Grimm, Z. Organocatalytic Electrophilic Arene Amination: Rapid Synthesis of 2-Quinolones. Chem. - Eur. J. 2025, 31 (6), e202403524 DOI: 10.1002/chem.202403524Google ScholarThere is no corresponding record for this reference.
- 126Sasa, H.; Mori, K.; Kikushima, K. μ-Oxo-Hypervalent-Iodine-Catalyzed Oxidative C–H Amination for Synthesis of Benzolactam Derivatives. Chem. Pharm. Bull. 2022, 70 (2), 106– 110, DOI: 10.1248/cpb.c21-00980Google ScholarThere is no corresponding record for this reference.
- 127Deeksha; Singh, R. A Modular Approach to Access 1,4-Benzoxazinones, Spirooxazolidinones, and 4,1-Benzoxazepinones via Oxidative Aryl C–N Bond Formation Using Catalytic Hypervalent Iodine. Org. Lett. 2025, 27 (50), 14112– 14117, DOI: 10.1021/acs.orglett.5c04757Google ScholarThere is no corresponding record for this reference.
- 128Parveen, N.; Aziz, M. N.; Gout, D.; Lovely, C. J. Dearomatization Reactions of N-OMe Ureas. Org. Lett. 2025, 27 (40), 11307– 11312, DOI: 10.1021/acs.orglett.5c03607Google ScholarThere is no corresponding record for this reference.
- 129Ortiz, G. X., Jr.; Hemric, B. N.; Wang, Q. Direct and Selective 3-Amidation of Indoles Using Electrophilic N-[(Benzenesulfonyl)oxy]amides. Org. Lett. 2017, 19 (6), 1314– 1317, DOI: 10.1021/acs.orglett.7b00358Google ScholarThere is no corresponding record for this reference.
- 130Barbor, J. P.; Nair, V. N.; Sharp, K. R. Development of a Nickel-Catalyzed N–N Coupling for the Synthesis of Hydrazides. J. Am. Chem. Soc. 2023, 145 (28), 15071– 15077, DOI: 10.1021/jacs.3c04834Google ScholarThere is no corresponding record for this reference.
- 131Fang, W.; Luo, Z.-W.; Wang, Y.-C. SN2 Reaction at the Amide Nitrogen Center Enables Hydrazide Synthesis. Angew. Chem., Int. Ed. 2024, 63 (14), e202317570 DOI: 10.1002/anie.202317570Google ScholarThere is no corresponding record for this reference.
- 132Yang, H.; Zhang, X.; Cao, F.-Y. Reverse Polarity of Amide Nitrogen Enables Expedient Access to N-Cyano Amides. Nat. Commun. 2025, 16 (1), 7706 DOI: 10.1038/s41467-025-63052-7Google ScholarThere is no corresponding record for this reference.
- 133Malik, M.; Senatore, R.; Langer, T. Base-Mediated Homologative Rearrangement of Nitrogen–oxygen Bonds of N-Methyl-N-oxyamides. Chem. Sci. 2023, 14 (37), 10140– 10146, DOI: 10.1039/D3SC03216GGoogle ScholarThere is no corresponding record for this reference.
- 134Li, G.; Lübken, T.; Plietker, B. A Ru-Catalyzed Desulfonylative C-Arylation of O-Tosyl-hydroxamates. Chem. Commun. 2024, 60 (7), 851– 853, DOI: 10.1039/D3CC05825EGoogle ScholarThere is no corresponding record for this reference.
- 135Wu, J.-C.; Yang, H.; Luo, Z.-W. Direct N-Methyl C–H Esterification of O-Tosyl hydroxamates to Enable Facile Synthesis of Hemiaminal Esters. Chem. Commun. 2025, 61 (57), 10542– 10545, DOI: 10.1039/D5CC02570BGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Scheme 1
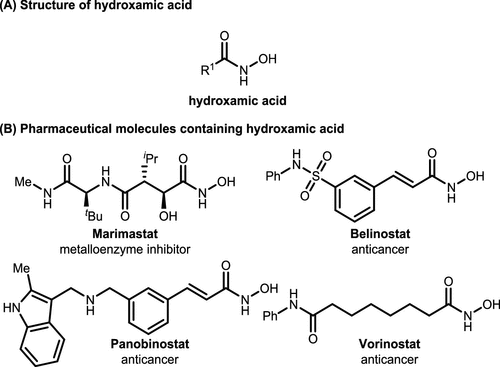 Scheme 1. Hydroxamic Acid and Related Pharmaceuticals
Scheme 1. Hydroxamic Acid and Related PharmaceuticalsScheme 2
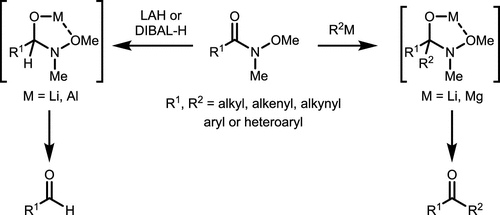 Scheme 2. Addition Reaction of Weinreb Amides with Organometallic Reagents
Scheme 2. Addition Reaction of Weinreb Amides with Organometallic ReagentsScheme 3
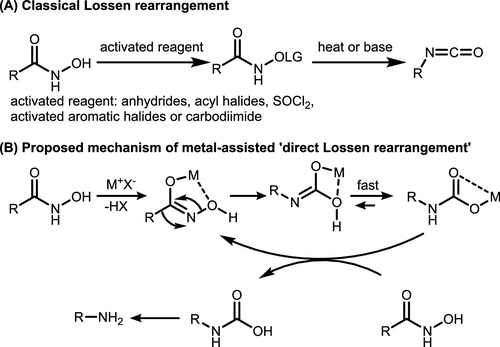 Scheme 3. Classical Lossen Rearrangement and the “Direct Lossen Rearrangement”
Scheme 3. Classical Lossen Rearrangement and the “Direct Lossen Rearrangement”Scheme 4
 Scheme 4. HERON Rearrangement
Scheme 4. HERON RearrangementScheme 5
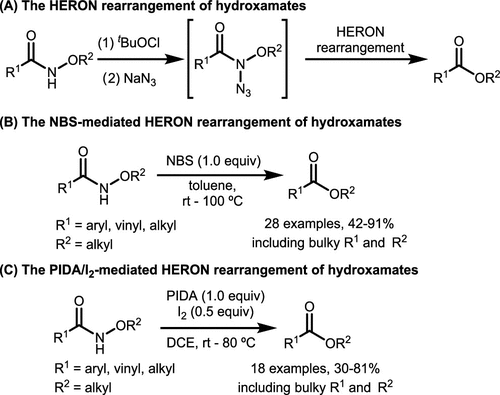 Scheme 5. Oxidative HERON Rearrangement
Scheme 5. Oxidative HERON RearrangementScheme 6
 Scheme 6. Pd-Catalyzed Cross-Coupling Reaction of Hydroxamates with Boronic Acid
Scheme 6. Pd-Catalyzed Cross-Coupling Reaction of Hydroxamates with Boronic AcidScheme 7
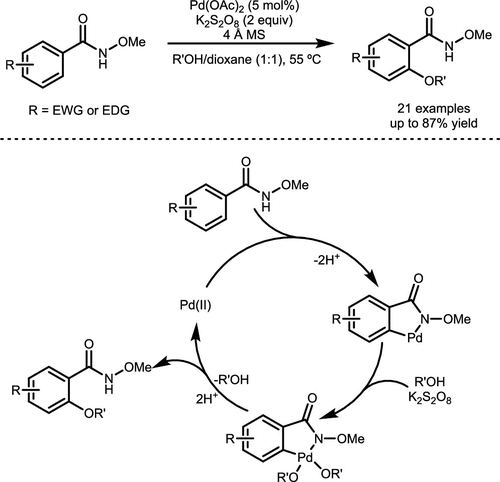 Scheme 7. Pd-Catalyzed Alkoxylation of N-Methoxybenzamides
Scheme 7. Pd-Catalyzed Alkoxylation of N-MethoxybenzamidesScheme 8
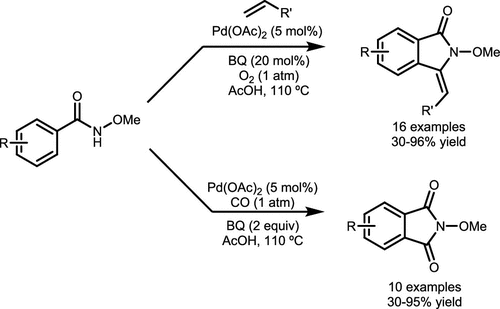 Scheme 8. PdII-Catalyzed Synthesis of Alkylidene Isoindolinones and Phthalimide
Scheme 8. PdII-Catalyzed Synthesis of Alkylidene Isoindolinones and PhthalimideScheme 9
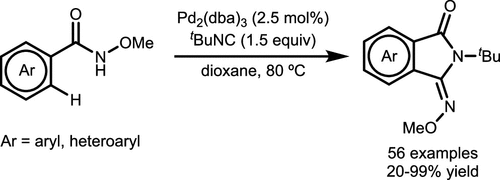 Scheme 9. Pd0-Catalyzed Directed C–H Functionalization of Heterocycles
Scheme 9. Pd0-Catalyzed Directed C–H Functionalization of HeterocyclesScheme 10
 Scheme 10. Pd0-Catalyzed Directed C–H Functionalization of Alkenes
Scheme 10. Pd0-Catalyzed Directed C–H Functionalization of AlkenesScheme 11
 Scheme 11. Ligand-Enabled β-C(sp3)-H Arylation of α-Amino Acids
Scheme 11. Ligand-Enabled β-C(sp3)-H Arylation of α-Amino AcidsScheme 12
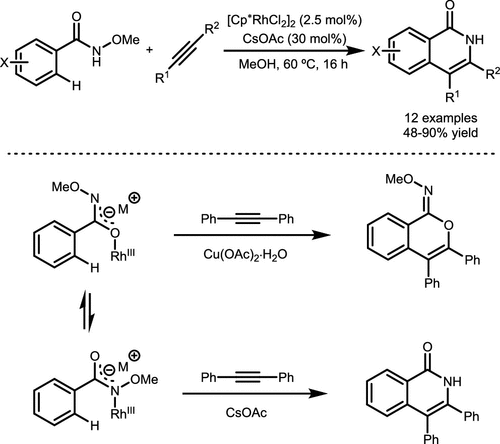 Scheme 12. Rh-Catalyzed Isoquinolone Synthesis from N-methoxybenzamide and Alkyne
Scheme 12. Rh-Catalyzed Isoquinolone Synthesis from N-methoxybenzamide and AlkyneScheme 13
 Scheme 13. Rh-Catalyzed Azepinone Synthesis from N-methoxybenzamide and α, β-Unsaturated Aldehydes or Ketones
Scheme 13. Rh-Catalyzed Azepinone Synthesis from N-methoxybenzamide and α, β-Unsaturated Aldehydes or KetonesScheme 14
 Scheme 14. Rh-Catalyzed C(sp2)-H Activation Reactions Involving 1,1-Disubstituted Allenes and Trisubstituted Allenes
Scheme 14. Rh-Catalyzed C(sp2)-H Activation Reactions Involving 1,1-Disubstituted Allenes and Trisubstituted AllenesScheme 15
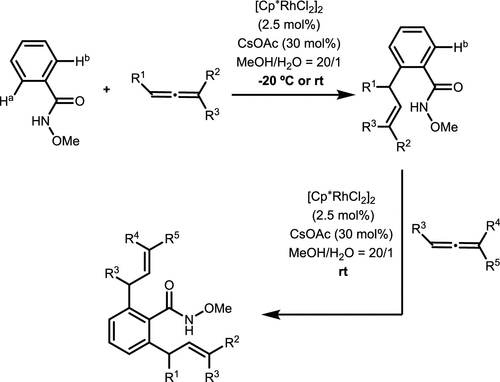
 Scheme 15. Rh-Catalyzed C(sp2)-H Activation Reactions
Scheme 15. Rh-Catalyzed C(sp2)-H Activation ReactionsScheme 16
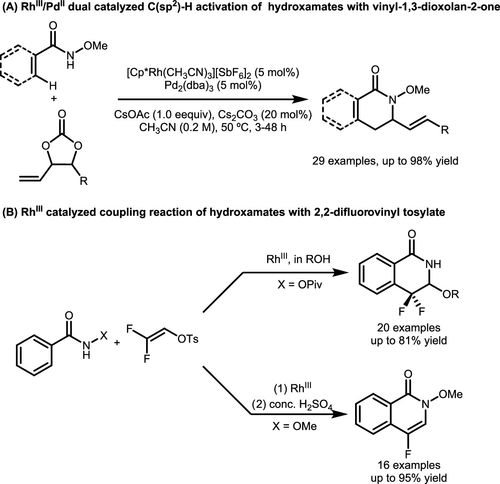
 Scheme 16. Rh-Catalyzed C(sp2)-H Activation Reactions Involving N-methoxyamides and Organometallic Reagents
Scheme 16. Rh-Catalyzed C(sp2)-H Activation Reactions Involving N-methoxyamides and Organometallic ReagentsScheme 17
 Scheme 17. Ru-Catalyzed C(sp2)-H Activation Reactions Involving N-Methoxyamide and Alkynes
Scheme 17. Ru-Catalyzed C(sp2)-H Activation Reactions Involving N-Methoxyamide and AlkynesScheme 18
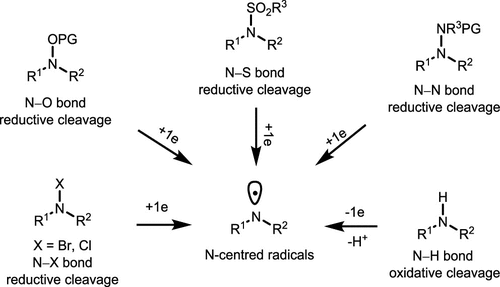 Scheme 18. Methods for the Generation of N-Centered Radicals
Scheme 18. Methods for the Generation of N-Centered RadicalsScheme 19
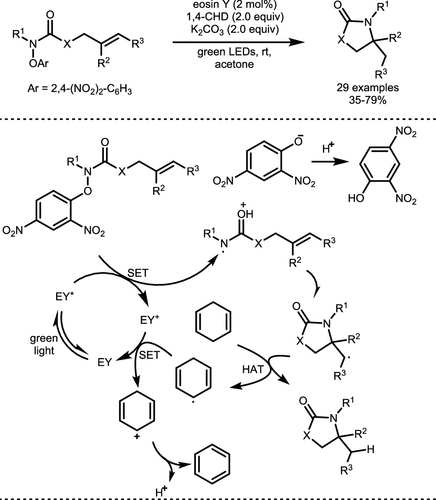 Scheme 19. Hydroamination of Amidyl Radicals Derived from Hydroxamates
Scheme 19. Hydroamination of Amidyl Radicals Derived from HydroxamatesScheme 20
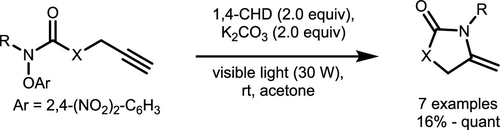 Scheme 20. Intramolecular 5-exo-dig Radical Cyclization of Amidyl Radicals and Alkynes
Scheme 20. Intramolecular 5-exo-dig Radical Cyclization of Amidyl Radicals and AlkynesScheme 21
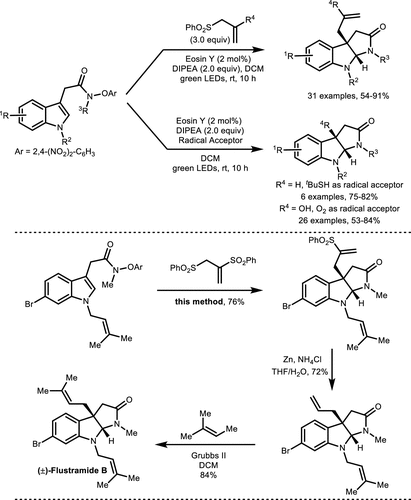 Scheme 21. Synthesis of Pyrroloindoline via an Amidyl Radical Cyclization/Carbon Radical Addition Cascade
Scheme 21. Synthesis of Pyrroloindoline via an Amidyl Radical Cyclization/Carbon Radical Addition CascadeScheme 22
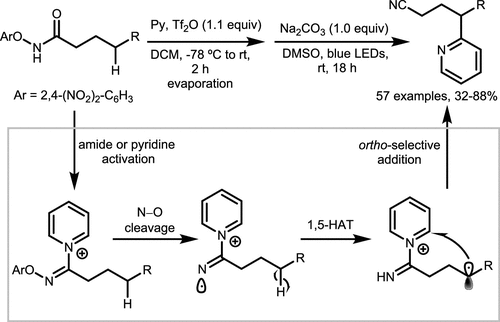 Scheme 22. γ-Selective Pyridylation of Hydroxamate
Scheme 22. γ-Selective Pyridylation of HydroxamateScheme 23
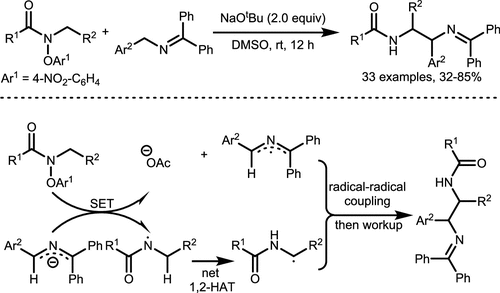 Scheme 23. Amidyl Radicals α-C(sp3)–H Coupling via Net-1,2-HAT
Scheme 23. Amidyl Radicals α-C(sp3)–H Coupling via Net-1,2-HATScheme 24
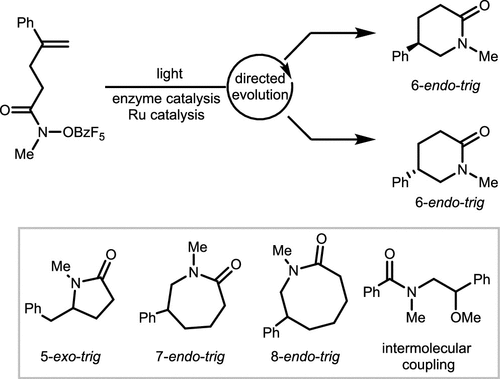 Scheme 24. Photo-Enzyme Co-Catalyzed Hydroamination of Alkenes and Hydroxamates
Scheme 24. Photo-Enzyme Co-Catalyzed Hydroamination of Alkenes and HydroxamatesScheme 25
 Scheme 25. [3 + 4] Cycloaddition of Aza-Oxyallyl Cationic Intermediates
Scheme 25. [3 + 4] Cycloaddition of Aza-Oxyallyl Cationic IntermediatesScheme 26
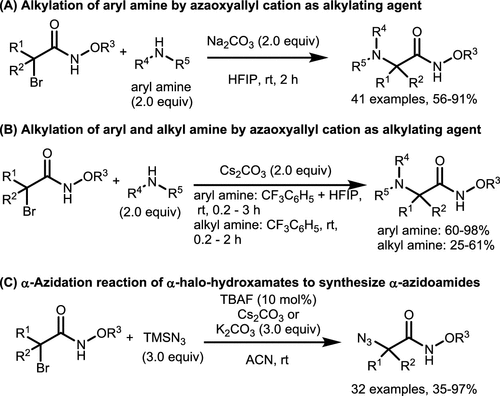 Scheme 26. Aza-Oxyallyl Cations as Alkylating Reagents for Amines
Scheme 26. Aza-Oxyallyl Cations as Alkylating Reagents for AminesScheme 27
 Scheme 27. Other Alkylation of Nucleophiles
Scheme 27. Other Alkylation of NucleophilesScheme 28
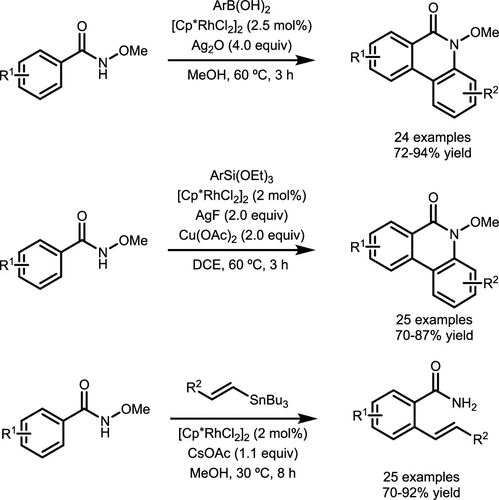
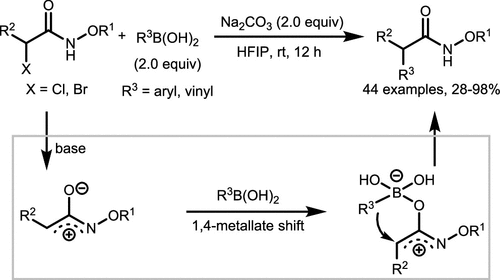 Scheme 28. Coupling of Boronic Acid and Hydroxamates via 1,4-Metallate Shift
Scheme 28. Coupling of Boronic Acid and Hydroxamates via 1,4-Metallate ShiftScheme 29
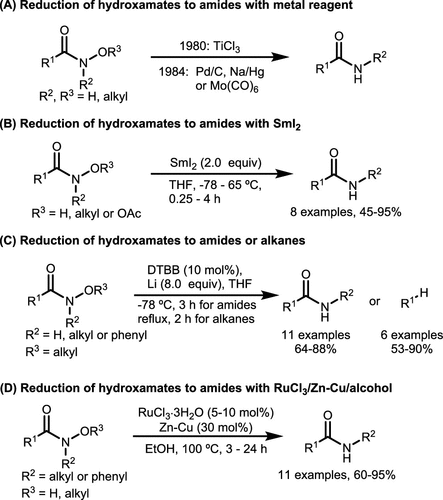 Scheme 29. Reduction Reactions of Hydroxamates via Metal Reagents
Scheme 29. Reduction Reactions of Hydroxamates via Metal ReagentsScheme 30
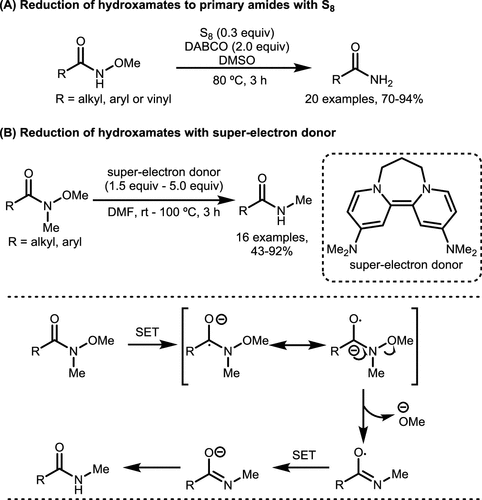 Scheme 30. Metal-Free Reduction Reactions of Hydroxamates
Scheme 30. Metal-Free Reduction Reactions of HydroxamatesScheme 31
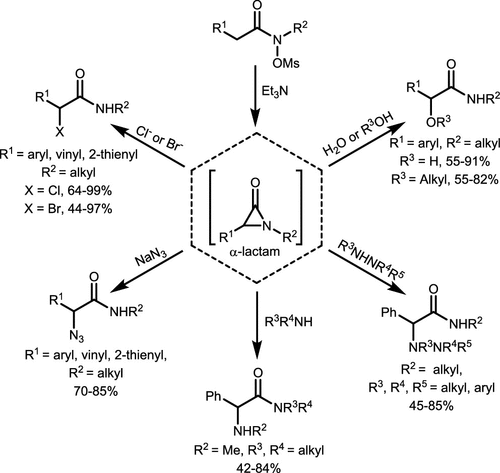 Scheme 31. α-Functionalized Amide Synthesis from Hydroxamates
Scheme 31. α-Functionalized Amide Synthesis from HydroxamatesScheme 32
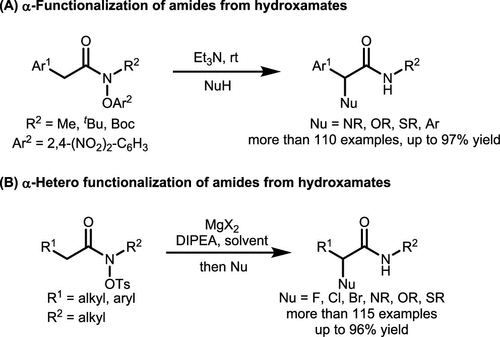 Scheme 32. α-Functionalization of Amides via α-Lactam Umpolung
Scheme 32. α-Functionalization of Amides via α-Lactam UmpolungScheme 33
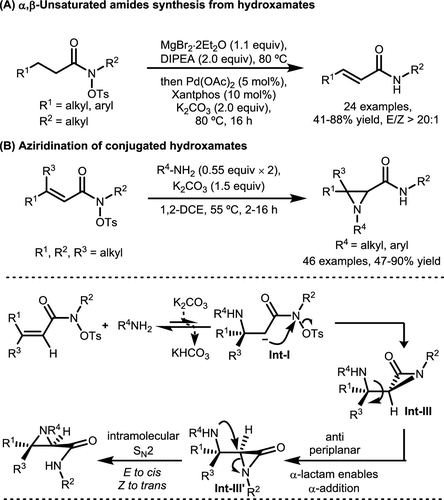 Scheme 33. α, β-Unsaturated Secondary Amides and Aziridination-Containing Amides’ Synthesis via α-Lactam Umpolung
Scheme 33. α, β-Unsaturated Secondary Amides and Aziridination-Containing Amides’ Synthesis via α-Lactam UmpolungScheme 34
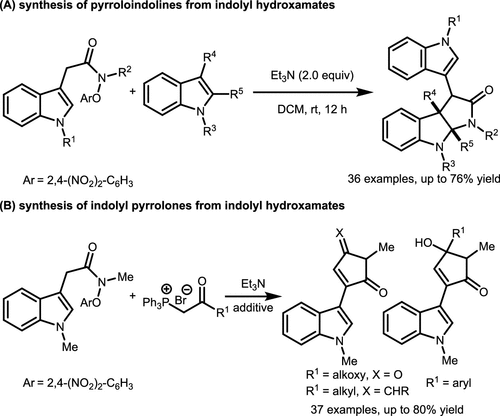 Scheme 34. Cycloaddition of Hydroxamates via α-Lactam Umpolung
Scheme 34. Cycloaddition of Hydroxamates via α-Lactam UmpolungScheme 35
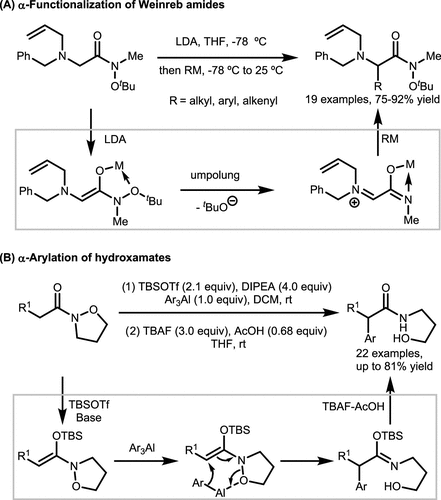 Scheme 35. α-Functionalization of Hydroxamates via Enolate Umpolung
Scheme 35. α-Functionalization of Hydroxamates via Enolate UmpolungScheme 36
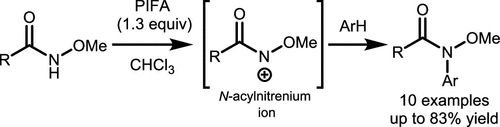 Scheme 36. Electrophilic Aromatic Amination Reaction of N-methoxyamides
Scheme 36. Electrophilic Aromatic Amination Reaction of N-methoxyamidesScheme 37
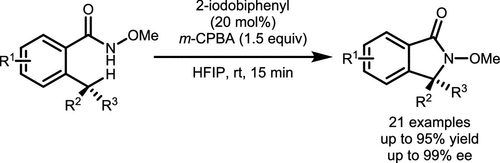 Scheme 37. γ-Lactam Synthesis via Intramolecular C(sp3)-H Amination of N-Methoxyamides
Scheme 37. γ-Lactam Synthesis via Intramolecular C(sp3)-H Amination of N-MethoxyamidesScheme 38
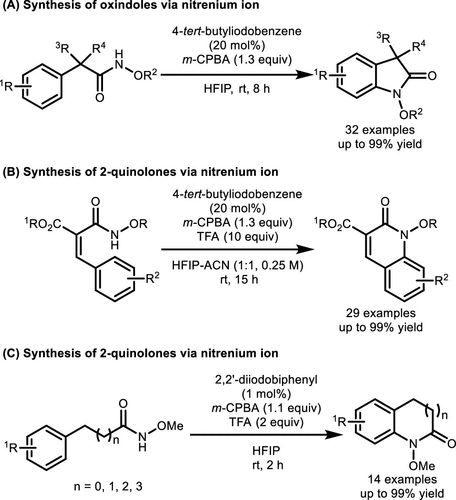 Scheme 38. Intramolecular C(sp2)–H Amination of Hydroxamates Catalyzed by Aryl Iodide
Scheme 38. Intramolecular C(sp2)–H Amination of Hydroxamates Catalyzed by Aryl IodideScheme 39
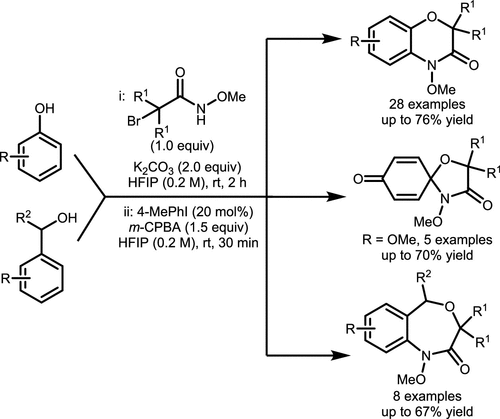 Scheme 39. Synthesis of 1,4-Benzoxazinones and 4,1-Benzoxazepinones via Aryl C(sp2)–H Amination
Scheme 39. Synthesis of 1,4-Benzoxazinones and 4,1-Benzoxazepinones via Aryl C(sp2)–H AminationScheme 40
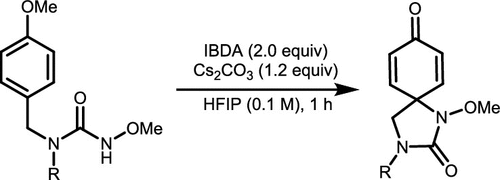 Scheme 40. Dearomatizative Spirocyclization of p-Methoxy-Substituted Phenols
Scheme 40. Dearomatizative Spirocyclization of p-Methoxy-Substituted PhenolsScheme 41
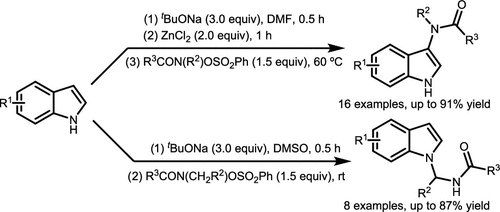 Scheme 41. N-Centered Functionalization Reactions of Hydroxamates
Scheme 41. N-Centered Functionalization Reactions of HydroxamatesScheme 42
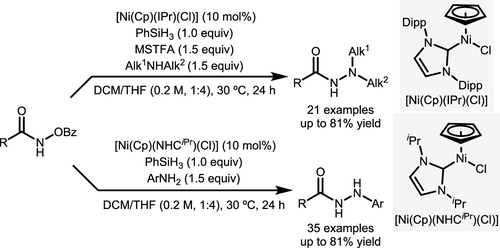 Scheme 42. Ni-Catalyzed N–N Coupling Reaction of Hydroxamates and Amines to Synthesize Hydrazides
Scheme 42. Ni-Catalyzed N–N Coupling Reaction of Hydroxamates and Amines to Synthesize HydrazidesScheme 43
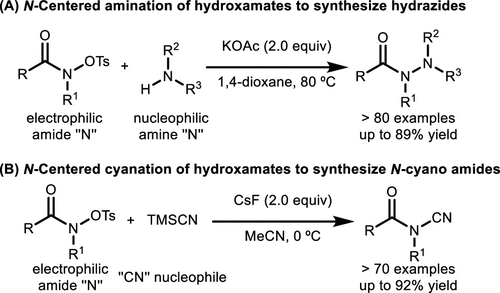 Scheme 43. Synthesis of Hydrazides and N-Cyanoamides via N-Amination and Cyanation of Hydroxamates
Scheme 43. Synthesis of Hydrazides and N-Cyanoamides via N-Amination and Cyanation of HydroxamatesScheme 44
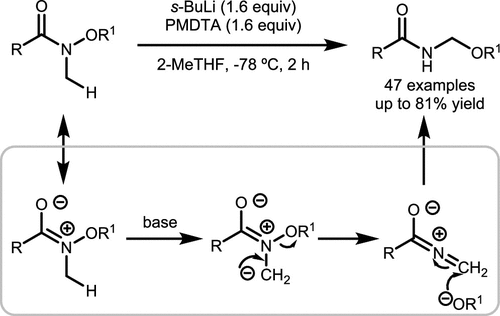 Scheme 44. Base-Mediated Homologation Rearrangement of N-Methyl-N-Alkoxyamides
Scheme 44. Base-Mediated Homologation Rearrangement of N-Methyl-N-AlkoxyamidesScheme 45
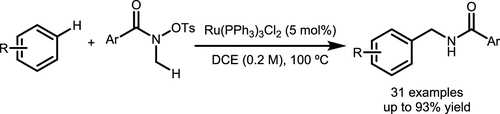 Scheme 45. Ru-Catalyzed α-C–H Arylation Reaction of the Nitrogen Atom in Hydroxamates
Scheme 45. Ru-Catalyzed α-C–H Arylation Reaction of the Nitrogen Atom in HydroxamatesScheme 46
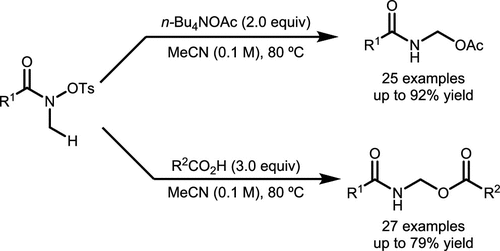 Scheme 46. α-C–H Esterification of the Nitrogen Atom in Hydroxamates
Scheme 46. α-C–H Esterification of the Nitrogen Atom in HydroxamatesReferences
This article references 135 other publications.
- 1(a) Lossen, H. Ueber die Oxalohydroxamsäure. Justus Liebigs Ann. Chem. 1869, 150 (3), 314– 322, DOI: 10.1002/jlac.18691500304There is no corresponding record for this reference.(b) Bauer, L.; Exner, O. The Chemistry of Hydroxamic Acids and N-Hydroxyimides. Angew. Chem., Int. Ed. 1974, 13 (6), 376– 384, DOI: 10.1002/anie.197403761There is no corresponding record for this reference.
- 2Keth, J.; Johann, T.; Frey, H. Hydroxamic Acid: An Underrated Moiety? Marrying Bioinorganic Chemistry and Polymer Science. Biomacromolecules 2020, 21 (7), 2546– 2556, DOI: 10.1021/acs.biomac.0c00449There is no corresponding record for this reference.
- 3Millar, A. W.; Brown, P. D.; Moore, J.; Galloway, W. A.; Cornish, A. G.; Lenehan, T. J.; Lynch, K. P. Results of Single and Repeat Dose Studies of the Oral Matrix Metalloproteinase Inhibitor Marimastat in Healthy Male Volunteers. Br. J. Clin. Pharmacol. 1998, 45 (1), 21– 26, DOI: 10.1046/j.1365-2125.1998.00639.xThere is no corresponding record for this reference.
- 4Zhao, C.; Dong, H.; Xu, Q.; Zhang, Y. Histone Deacetylase (HDAC) Inhibitors in Cancer: a Patent Review (2017-present). Expert Opin. Ther. Pat. 2020, 30 (4), 263– 274, DOI: 10.1080/13543776.2020.1725470There is no corresponding record for this reference.
- 5(a) Lin, Z.; Decker, E. A.; Goddard, J. M. Preparation of Metal Chelating Active Packaging Materials by Laminated Photografting. J. Coat. Technol. Res. 2016, 13 (2), 395– 404, DOI: 10.1007/s11998-015-9767-zThere is no corresponding record for this reference.(b) Hamilton, J. L.; Kizhakkedathu, J. N. Polymeric Nanocarriers for the Treatment of Systemic Iron Overload. Mol. Cell. Ther. 2015, 3 (1), 3 DOI: 10.1186/s40591-015-0039-1There is no corresponding record for this reference.
- 6(a) Nahm, S.; Weinreb, S. M. N-Methoxy-N-methylamides as Effective Acylating Agents. Tetrahedron Lett. 1981, 22 (39), 3815– 3818, DOI: 10.1016/S0040-4039(01)91316-4There is no corresponding record for this reference.(b) Sibi, M. P. Chemistry of N-Methoxy-N-Methylamides. Applications in Synthesis. A Review. Org. Prep. Proced. Int. 1993, 25 (1), 15– 40, DOI: 10.1080/00304949309457931There is no corresponding record for this reference.(c) Mentzel, M.; Hoffmann, H. M. R. N-Methoxy-N-Methylamides (Weinreb Amides) in Modern Organic Synthesis. J. Prakt. Chem. 1997, 339 (1), 517– 524, DOI: 10.1002/prac.19973390194There is no corresponding record for this reference.(d) Balasubramaniam, S.; Aidhen, I. S. The Growing Synthetic Utility of the Weinreb Amide. Synthesis 2008, 2008 (23), 3707– 3738, DOI: 10.1055/s-0028-1083226There is no corresponding record for this reference.
- 7(a) Molina, P.; Tárraga, A.; Arques, A. 5.26 - Functions with at Least One Oxygen, YCO. In Comprehensive Organic Functional Group Transformations II; Katritzky, A. R.; Taylor, R. J. K., Eds.; Elsevier, 2005; pp 949– 973.There is no corresponding record for this reference.(b) Wang, R.; Liu, W. H. Amide Synthesis from Decarboxylative Coupling of Isocyanates and Carboxylic Acids. ChemBioChem 2025, 26 (2), e202400770 DOI: 10.1002/cbic.202400770There is no corresponding record for this reference.
- 8Podlaha, J.; Cíísařová, I.; Soukupová, L.; Schraml, J.; Exner, O. Structure of Alkali Metal Hydroxamates and their Lossen Rearrangement. J. Chem. Res. 1998, (9), 520– 521, DOI: 10.1039/a802942cThere is no corresponding record for this reference.
- 9Ducháčková, L.; Roithová, J. The Interaction of Zinc(II) and Hydroxamic Acids and a Metal-Triggered Lossen Rearrangement. Chem. - Eur. J. 2009, 15 (48), 13399– 13405, DOI: 10.1002/chem.200901645There is no corresponding record for this reference.
- 10Jašíková, L.; Hanikýřová, E.; Škríba, A.; Jašík, J.; Roithová, J. Metal-assisted Lossen Rearrangement. J. Org. Chem. 2012, 77 (6), 2829– 2836, DOI: 10.1021/jo300031fThere is no corresponding record for this reference.
- 11Hoshino, Y.; Okuno, M.; Kawamura, E.; Honda, K.; Inoue, S. Base-mediated Rearrangement of Free Aromatichydroxamic Acids (ArCO–NHOH) to Anilines. Chem. Commun. 2009, (17), 2281– 2283, DOI: 10.1039/b822806jThere is no corresponding record for this reference.
- 12(a) Hoshino, Y.; Shimbo, Y.; Ohtsuka, N.; Honda, K. Self-propagated Lossen Rearrangement Induced by a Catalytic Amount of Activating Agents under Mild Conditions. Tetrahedron Lett. 2015, 56 (5), 710– 712, DOI: 10.1016/j.tetlet.2014.12.084There is no corresponding record for this reference.(b) Ohtsuka, N.; Okuno, M.; Hoshino, Y.; Honda, K. A Base-mediated Self-propagative Lossen Rearrangement of Hydroxamic Acids for the Efficient and Facile Synthesis of Aromatic and Aliphatic Primary Amines. Org. Biomol. Chem. 2016, 14 (38), 9046– 9054, DOI: 10.1039/C6OB01178KThere is no corresponding record for this reference.(c) Strotman, N. A.; Ortiz, A.; Savage, S. A.; Wilbert, C. R.; Ayers, S.; Kiau, S. Revisiting a Classic Transformation: A Lossen Rearrangement Initiated by Nitriles and “Pseudo-Catalytic” in Isocyanate. J. Org. Chem. 2017, 82 (8), 4044– 4049, DOI: 10.1021/acs.joc.7b00450There is no corresponding record for this reference.
- 13Ortiz, A.; Soumeillant, M.; Savage, S. A.; Strotman, N. A.; Haley, M.; Benkovics, T.; Nye, J.; Xu, Z.; Tan, Y.; Ayers, S. Synthesis of HIV-Maturation Inhibitor BMS-955176 from Betulin by an Enabling Oxidation Strategy. J. Org. Chem. 2017, 82 (9), 4958– 4963, DOI: 10.1021/acs.joc.7b00438There is no corresponding record for this reference.
- 14Ghosh, A. K.; Brindisi, M. Organic Carbamates in Drug Design and Medicinal Chemistry. J. Med. Chem. 2015, 58 (7), 2895– 2940, DOI: 10.1021/jm501371sThere is no corresponding record for this reference.
- 15Maisonneuve, L.; Lamarzelle, O.; Rix, E.; Grau, E.; Cramail, H. Isocyanate-Free Routes to Polyurethanes and Poly(hydroxy Urethane)s. Chem. Rev. 2015, 115 (22), 12407– 12439, DOI: 10.1021/acs.chemrev.5b00355There is no corresponding record for this reference.
- 16(a) Glover, S. A.; Rauk, A.; Buccigross, J. M.; Campbell, J. J.; Hammond, G. P.; Mo, G.; Andrews, L. E.; Gillson, A.-M. E. The HERON Reaction, Origin, Theoretical Background, and Prevalence. Can. J. Chem. 2005, 83 (9), 1492– 1509, DOI: 10.1139/v05-150There is no corresponding record for this reference.(b) Glover, S. A. Development of the HERON Reaction: A Historical Account. Aust. J. Chem. 2017, 70 (4), 344– 361, DOI: 10.1071/CH16683There is no corresponding record for this reference.
- 17De Almeida, M. V.; Barton, D. H. R.; Bytheway, I.; Ferreira, J. A.; Hall, M. B.; Liu, W.; Taylor, D. K.; Thomson, L. Preparation and Thermal Decomposition of N,N′-Diacyl-N,N′-Dialkoxyhydrazines: Synthetic Applications and Mechanistic Insights. J. Am. Chem. Soc. 1995, 117 (17), 4870– 4874, DOI: 10.1021/ja00122a018There is no corresponding record for this reference.
- 18(a) Crawford, R. J.; Raap, R. The Synthesis and Reactions of N,N′-Dicarbalkoxy-N,N′-dialkoxyhydrazines and Some Observations on Carbalkoxylium Ions. J. Org. Chem. 1963, 28 (9), 2419– 2424, DOI: 10.1021/jo01044a066There is no corresponding record for this reference.(b) Cooley, J. H.; Mosher, M. W.; Khan, M. A. Preparation and Reactions of N,N′-diacyl-N,N′-dialkoxyhydrazines. J. Am. Chem. Soc. 1968, 90 (7), 1867– 1871, DOI: 10.1021/ja01009a032There is no corresponding record for this reference.(c) Glover, S. A.; Mo, G.; Rauk, A. HERON Rearrangement of N,N′-diacyl-N,N′-dialkoxyhydrazines─a Theoretical and Experimental Study. Tetrahedron 1999, 55 (11), 3413– 3426, DOI: 10.1016/S0040-4020(98)01151-XThere is no corresponding record for this reference.
- 19Glover, S. A.; Mo, G. Hindered Ester Formation by SN2 Azidation of N-acetoxy-N-alkoxyamides and N-alkoxy-N-chloroamides─Novel Application of HERON Rearrangements. J. Chem. Soc., Perkin Trans. 2 2002, (10), 1728– 1739, DOI: 10.1039/B111250NThere is no corresponding record for this reference.
- 20Zhang, N.; Yang, R.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. Direct Conversion of N-Alkoxyamides to Carboxylic Esters through Tandem NBS-Mediated Oxidative Homocoupling and Thermal Denitrogenation. J. Org. Chem. 2013, 78 (17), 8705– 8711, DOI: 10.1021/jo401435vThere is no corresponding record for this reference.
- 21Duan, X.; Duan, X.; Yang, K.; Yang, K.; Wang, Z.; Wang, Z.; Zhang, L.; Zhang, L.; Tong, Y.; Tong, Y.; Liu, H.; Liu, H.; Du, K.; Du, K.; Tang, R. PhI(OAc)2/I2-Mediated Oxidative Homocoupling of N-Alkoxyamides: An Approach to Esters. Chin. J. Org. Chem. 2015, 35 (12), 2552– 2558, DOI: 10.6023/cjoc201506024There is no corresponding record for this reference.
- 22Ghosh, S.; Banerjee, J.; Ghosh, R.; Chattopadhyay, S. K. A Metal-free Iodine-mediated Conversion of Hydroxamates to Esters. Synth. Commun. 2020, 50 (9), 1353– 1360, DOI: 10.1080/00397911.2020.1737130There is no corresponding record for this reference.
- 23Engle, K. M.; Mei, T.-S.; Wasa, M.; Yu, J.-Q. Weak Coordination as a Powerful Means for Developing Broadly Useful C–H Functionalization Reactions. Acc. Chem. Res. 2012, 45 (6), 788– 802, DOI: 10.1021/ar200185gThere is no corresponding record for this reference.
- 24(a) Wang, D.-H.; Engle, K. M.; Shi, B.-F.; Yu, J.-Q. Ligand-Enabled Reactivity and Selectivity in a Synthetically Versatile Aryl C–H Olefination. Science 2010, 327 (5963), 315– 319, DOI: 10.1126/science.1182512There is no corresponding record for this reference.(b) Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Ligand-Accelerated C–H Activation Reactions: Evidence for a Switch of Mechanism. J. Am. Chem. Soc. 2010, 132 (40), 14137– 14151, DOI: 10.1021/ja105044sThere is no corresponding record for this reference.(c) Wasa, M.; Chan, K. S. L.; Zhang, X.-G.; He, J.; Miura, M.; Yu, J.-Q. Ligand-Enabled Methylene C(sp3)–H Bond Activation with a Pd(II) Catalyst. J. Am. Chem. Soc. 2012, 134 (45), 18570– 18572, DOI: 10.1021/ja309325eThere is no corresponding record for this reference.(d) Zhu, R.-Y.; Farmer, M. E.; Chen, Y.-Q.; Yu, J.-Q. A Simple and Versatile Amide Directing Group for C–H Functionalizations. Angew. Chem., Int. Ed. 2016, 55 (36), 10578– 10599, DOI: 10.1002/anie.201600791There is no corresponding record for this reference.
- 25He, J.; Li, S.; Deng, Y.; Fu, H.; Laforteza, B. N.; Spangler, J. E.; Homs, A.; Yu, J.-Q. Ligand-Controlled C(sp3)–H Arylation and Olefination in Synthesis of Unnatural Chiral Amino Acids. Science 2014, 343 (6176), 1216– 1220, DOI: 10.1126/science.1249198There is no corresponding record for this reference.
- 26Chen, G.; Shigenari, T.; Jain, P.; Zhang, Z.; Jin, Z.; He, J.; Li, S.; Mapelli, C.; Miller, M. M.; Poss, M. A. Ligand-Enabled β-C–H Arylation of α-Amino Acids Using a Simple and Practical Auxiliary. J. Am. Chem. Soc. 2015, 137 (9), 3338– 3351, DOI: 10.1021/ja512690xThere is no corresponding record for this reference.
- 27Yus, M.; Radivoy, G.; Alonso, F. Lithium/DTBB-Induced Reduction of N-Alkoxyamides and Acyl Azides. Synthesis 2001, 2001, 0914– 0918, DOI: 10.1055/s-2001-13415There is no corresponding record for this reference.
- 28Wang, D.-H.; Wasa, M.; Giri, R.; Yu, J.-Q. Pd(II)-Catalyzed Cross-Coupling of sp3 C–H Bonds with sp2 and sp3 Boronic Acids Using Air as the Oxidant. J. Am. Chem. Soc. 2008, 130 (23), 7190– 7191, DOI: 10.1021/ja801355sThere is no corresponding record for this reference.
- 29Wang, G.-W.; Yuan, T.-T. Palladium-Catalyzed Alkoxylation of N-Methoxybenzamides via Direct sp2 C–H Bond Activation. J. Org. Chem. 2010, 75 (2), 476– 479, DOI: 10.1021/jo902139bThere is no corresponding record for this reference.
- 30Wrigglesworth, J. W.; Cox, B.; Lloyd-Jones, G. C.; Booker-Milburn, K. I. New Heteroannulation Reactions of N-Alkoxybenzamides by Pd(II) Catalyzed C–H Activation. Org. Lett. 2011, 13 (19), 5326– 5329, DOI: 10.1021/ol202187hThere is no corresponding record for this reference.
- 31Liu, Y.-J.; Xu, H.; Kong, W.-J.; Shang, M.; Dai, H.-X.; Yu, J.-Q. Overcoming the Limitations of Directed C–H Functionalizations of Heterocycles. Nature 2014, 515 (7527), 389– 393, DOI: 10.1038/nature13885There is no corresponding record for this reference.
- 32Kong, W.-J.; Liu, Y.-J.; Xu, H.; Chen, Y.-Q.; Dai, H.-X.; Yu, J.-Q. Pd-Catalyzed α-Selective C–H Functionalization of Olefins: En Route to 4-Imino-β-Lactams. J. Am. Chem. Soc. 2016, 138 (7), 2146– 2149, DOI: 10.1021/jacs.5b13353There is no corresponding record for this reference.
- 33Guimond, N.; Gouliaras, C.; Fagnou, K. Rhodium(III)-Catalyzed Isoquinolone Synthesis: The N–O Bond as a Handle for C–N Bond Formation and Catalyst Turnover. J. Am. Chem. Soc. 2010, 132 (20), 6908– 6909, DOI: 10.1021/ja102571bThere is no corresponding record for this reference.
- 34Guimond, N.; Gouliaras, C.; Fagnou, K. Correction to Rhodium(III)-Catalyzed Isoquinolone Synthesis: The N–O Bond as a Handle for C–N Bond Formation and Catalyst Turnover. J. Am. Chem. Soc. 2012, 134 (43), 18148, DOI: 10.1021/ja3096625There is no corresponding record for this reference.
- 35Shi, Z.; Grohmann, C.; Glorius, F. Mild Rhodium(III)-Catalyzed Cyclization of Amides with α,β-Unsaturated Aldehydes and Ketones to Azepinones: Application to the Synthesis of the Homoprotoberberine Framework. Angew. Chem., Int. Ed. 2013, 52 (20), 5393– 5397, DOI: 10.1002/anie.201301426There is no corresponding record for this reference.
- 36Zeng, R.; Fu, C.; Ma, S. Highly Selective Mild Stepwise Allylation of N-Methoxybenzamides with Allenes. J. Am. Chem. Soc. 2012, 134 (23), 9597– 9600, DOI: 10.1021/ja303790sThere is no corresponding record for this reference.
- 37Wang, H.; Glorius, F. Mild Rhodium(III)-Catalyzed C–H Activation and Intermolecular Annulation with Allenes. Angew. Chem., Int. Ed. 2012, 51 (29), 7318– 7322, DOI: 10.1002/anie.201201273There is no corresponding record for this reference.
- 38Zhang, S.-S.; Wu, J.-Q.; Liu, X.; Wang, H. Tandem Catalysis: Rh(III)-Catalyzed C–H Allylation/Pd(II)-Catalyzed N-Allylation Toward the Synthesis of Vinyl-Substituted N-Heterocycles. ACS Catal. 2015, 5 (1), 210– 214, DOI: 10.1021/cs501601cThere is no corresponding record for this reference.
- 39Wu, J.-Q.; Zhang, S.-S.; Gao, H.; Qi, Z.; Zhou, C.-J.; Ji, W.-W.; Liu, Y.; Chen, Y.; Li, Q.; Li, X.; Wang, H. Experimental and Theoretical Studies on Rhodium-Catalyzed Coupling of Benzamides with 2,2-Difluorovinyl Tosylate: Diverse Synthesis of Fluorinated Heterocycles. J. Am. Chem. Soc. 2017, 139 (9), 3537– 3545, DOI: 10.1021/jacs.7b00118There is no corresponding record for this reference.
- 40(a) Chen, X.; Li, J.-J.; Hao, X.-S.; Goodhue, C. E.; Yu, J.-Q. Palladium-Catalyzed Alkylation of Aryl C–H Bonds with sp3 Organotin Reagents Using Benzoquinone as a Crucial Promoter. J. Am. Chem. Soc. 2006, 128 (1), 78– 79, DOI: 10.1021/ja0570943There is no corresponding record for this reference.(b) Chen, X.; Goodhue, C. E.; Yu, J.-Q. Palladium-Catalyzed Alkylation of sp2 and sp3 C–H Bonds with Methylboroxine and Alkylboronic Acids: Two Distinct C–H Activation Pathways. J. Am. Chem. Soc. 2006, 128 (39), 12634– 12635, DOI: 10.1021/ja0646747There is no corresponding record for this reference.(c) Giri, R.; Maugel, N.; Li, J.-J.; Wang, D.-H.; Breazzano, S. P.; Saunders, L. B.; Yu, J.-Q. Palladium-Catalyzed Methylation and Arylation of sp2 and sp3 C–H Bonds in Simple Carboxylic Acids. J. Am. Chem. Soc. 2007, 129 (12), 3510– 3511, DOI: 10.1021/ja0701614There is no corresponding record for this reference.(d) Wang, D.-H.; Mei, T.-S.; Yu, J.-Q. Versatile Pd(II)-Catalyzed C–H Activation/Aryl–Aryl Coupling of Benzoic and Phenyl Acetic Acids. J. Am. Chem. Soc. 2008, 130 (52), 17676– 17677, DOI: 10.1021/ja806681zThere is no corresponding record for this reference.
- 41Karthikeyan, J.; Haridharan, R.; Cheng, C.-H. Rhodium(III)-Catalyzed Oxidative C–H Coupling of N-Methoxybenzamides with Aryl Boronic Acids: One-Pot Synthesis of Phenanthridinones. Angew. Chem., Int. Ed. 2012, 51 (49), 12343– 12347, DOI: 10.1002/anie.201206890There is no corresponding record for this reference.
- 42Senthilkumar, N.; Parthasarathy, K.; Gandeepan, P.; Cheng, C.-H. Synthesis of Phenanthridinones from N-Methoxybenzamides and Aryltriethoxysilanes through Rh(III)-Catalyzed C–H and N–H Bond Activation. Chem. - Asian J. 2013, 8 (9), 2175– 2181, DOI: 10.1002/asia.201300356There is no corresponding record for this reference.
- 43Prakash, S.; Muralirajan, K.; Cheng, C.-H. Rh-catalyzed Oxidizing Group-directed Ortho C–H Vinylation of Arenes by Vinylstannanes. Chem. Commun. 2015, 51 (69), 13362– 13364, DOI: 10.1039/C5CC04211AThere is no corresponding record for this reference.
- 44Li, B.; Feng, H.; Xu, S.; Wang, B. Ruthenium-Catalyzed Isoquinolone Synthesis through C–H Activation Using an Oxidizing Directing Group. Chem. - Eur. J. 2011, 17 (45), 12573– 12577, DOI: 10.1002/chem.201102445There is no corresponding record for this reference.
- 45Ackermann, L.; Fenner, S. Ruthenium-Catalyzed C–H/N–O Bond Functionalization: Green Isoquinolone Syntheses in Water. Org. Lett. 2011, 13 (24), 6548– 6551, DOI: 10.1021/ol202861kThere is no corresponding record for this reference.
- 46(a) Kärkäs, M. D. Photochemical Generation of Nitrogen-Centered Amidyl, Hydrazonyl, and Imidyl Radicals: Methodology Developments and Catalytic Applications. ACS Catal. 2017, 7 (8), 4999– 5022, DOI: 10.1021/acscatal.7b01385There is no corresponding record for this reference.(b) Jiang, H.; Studer, A. Chemistry With N-Centered Radicals Generated by Single-Electron Transfer-Oxidation Using Photoredox Catalysis. CCS Chem. 2019, 1 (1), 38– 49, DOI: 10.31635/ccschem.019.20180026There is no corresponding record for this reference.
- 47(a) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Visible Light Photoredox-controlled Reactions of N-radicals and Radical Ions. Chem. Soc. Rev. 2016, 45 (8), 2044– 2056, DOI: 10.1039/C5CS00655DThere is no corresponding record for this reference.(b) Zard, S. Z. Recent Progress in the Generation and Use of Nitrogen-Centred Radicals. Chem. Soc. Rev. 2008, 37 (8), 1603– 1618, DOI: 10.1039/b613443mThere is no corresponding record for this reference.
- 48Boivin, J.; Callier-Dublanchet, A.-C.; Quiclet-Sire, B.; Schiano, A.-M.; Zard, S. Z. Iminyl, Amidyl, and Carbamyl Radicals from O-Benzoyl Oximes and O-Benzoyl Hydroxamic Acid Derivatives. Tetrahedron 1995, 51 (23), 6517– 6528, DOI: 10.1016/0040-4020(95)00319-4There is no corresponding record for this reference.
- 49Lin, X.; Artman, G. D.; Stien, D.; Weinreb, S. M. Development of Efficient New Methodology for Generation, Cyclization and Functional Trapping of Iminyl and Amidyl radicals. Tetrahedron 2001, 57 (42), 8779– 8791, DOI: 10.1016/S0040-4020(01)00878-XThere is no corresponding record for this reference.
- 50Artman Iii, G. D.; Waldman, J. H.; Weinreb, S. M. β-Tosylethylhydroxylamine: Preparation and Use as a Hydroxylamine Equivalent in Amidyl Radical-Olefin Cyclizations. Synthesis 2002, (14), 2057– 2063, DOI: 10.1055/s-2002-34371There is no corresponding record for this reference.
- 51Davies, J.; Svejstrup, T. D.; Reina, D. F.; Sheikh, N. S.; Leonori, D. Visible-Light-Mediated Synthesis of Amidyl Radicals: Transition-Metal-Free Hydroamination and N-Arylation Reactions. J. Am. Chem. Soc. 2016, 138 (26), 8092– 8095, DOI: 10.1021/jacs.6b04920There is no corresponding record for this reference.
- 52Reina, D. F.; Dauncey, E. M.; Morcillo, S. P.; Svejstrup, T. D.; Popescu, M. V.; Douglas, J. J.; Sheikh, N. S.; Leonori, D. Visible-Light-Mediated 5-exo-dig Cyclizations of Amidyl Radicals. Eur. J. Org. Chem. 2017, 2017 (15), 2108– 2111, DOI: 10.1002/ejoc.201601607There is no corresponding record for this reference.
- 53Jung, M. E.; Piizzi, G. gem-Disubstituent Effect: Theoretical Basis and Synthetic Applications. Chem. Rev. 2005, 105 (5), 1735– 1766, DOI: 10.1021/cr940337hThere is no corresponding record for this reference.
- 54Wu, K.; Du, Y.; Wang, T. Visible-Light-Mediated Construction of Pyrroloindolines via an Amidyl Radical Cyclization/Carbon Radical Addition Cascade: Rapid Synthesis of (±)-Flustramide B. Org. Lett. 2017, 19 (20), 5669– 5672, DOI: 10.1021/acs.orglett.7b02837There is no corresponding record for this reference.
- 55Wu, K.; Du, Y.; Wei, Z.; Wang, T. Synthesis of Functionalized Pyrroloindolines via a Visible-Light-Induced Radical Cascade Reaction: Rapid Synthesis of (±)-Flustraminol B. Chem. Commun. 2018, 54 (54), 7443– 7446, DOI: 10.1039/C8CC03575JThere is no corresponding record for this reference.
- 56(a) Vitaku, E.; Smith, D. T.; Njardarson, J. T. Analysis of the Structural Diversity, Substitution Patterns, and Frequency of Nitrogen Heterocycles among U.S. FDA Approved Pharmaceuticals. J. Med. Chem. 2014, 57 (24), 10257– 10274, DOI: 10.1021/jm501100bThere is no corresponding record for this reference.(b) Shi, Q.; Huang, X.; Yang, R.; Liu, W. H. Unified Ionic and Radical C-4 Alkylation and Arylation of Pyridines. Chem. Sci. 2024, 15 (31), 12442– 12450, DOI: 10.1039/D4SC03739AThere is no corresponding record for this reference.(c) Huang, X.; Shi, Q.; Lai, G.; Liu, W. H. 1,4-Dearomatization of Pyridines and Quinolines Enabled by the Carbodication Lewis Acid. Angew. Chem., Int. Ed. 2025, 64 (37), e202510385 DOI: 10.1002/anie.202510385There is no corresponding record for this reference.
- 57Bull, J. A.; Mousseau, J. J.; Pelletier, G.; Charette, A. B. Synthesis of Pyridine and Dihydropyridine Derivatives by Regio- and Stereoselective Addition to N-Activated Pyridines. Chem. Rev. 2012, 112 (5), 2642– 2713, DOI: 10.1021/cr200251dThere is no corresponding record for this reference.
- 58(a) Yu, X.-Y.; Zhao, Q.-Q.; Chen, J.; Xiao, W.-J.; Chen, J.-R. When Light Meets Nitrogen-Centered Radicals: From Reagents to Catalysts. Acc. Chem. Res. 2020, 53 (5), 1066– 1083, DOI: 10.1021/acs.accounts.0c00090There is no corresponding record for this reference.(b) Davies, J.; Morcillo, S. P.; Douglas, J. J.; Leonori, D. Hydroxylamine Derivatives as Nitrogen-Radical Precursors in Visible-Light Photochemistry. Chem. - Eur. J. 2018, 24 (47), 12154– 12163, DOI: 10.1002/chem.201801655There is no corresponding record for this reference.
- 59Feng, M.; Zhang, H.; Maulide, N. Challenges and Breakthroughs in Selective Amide Activation. Angew. Chem., Int. Ed. 2022, 61 (49), e202212213 DOI: 10.1002/anie.202212213There is no corresponding record for this reference.
- 60Kweon, B.; Kim, C.; Kim, S.; Hong, S. Remote C–H Pyridylation of Hydroxamates through Direct Photoexcitation of O-Aryl Oxime Pyridinium Intermediates. Angew. Chem., Int. Ed. 2021, 60 (51), 26813– 26821, DOI: 10.1002/anie.202112364There is no corresponding record for this reference.
- 61(a) Murphy, J. A. Discovery and Development of Organic Super-Electron-Donors. J. Org. Chem. 2014, 79 (9), 3731– 3746, DOI: 10.1021/jo500071uThere is no corresponding record for this reference.(b) Barham, J. P.; Coulthard, G.; Emery, K. J.; Doni, E.; Cumine, F.; Nocera, G.; John, M. P.; Berlouis, L. E. A.; McGuire, T.; Tuttle, T.; Murphy, J. A. KOtBu: A Privileged Reagent for Electron Transfer Reactions?. J. Am. Chem. Soc. 2016, 138 (23), 7402– 7410, DOI: 10.1021/jacs.6b03282There is no corresponding record for this reference.(c) Barham, J. P.; Dalton, S. E.; Allison, M.; Nocera, G.; Young, A.; John, M. P.; McGuire, T.; Campos, S.; Tuttle, T.; Murphy, J. A. Dual Roles for Potassium Hydride in Haloarene Reduction: CSNAr and Single Electron Transfer Reduction via Organic Electron Donors Formed in Benzene. J. Am. Chem. Soc. 2018, 140 (36), 11510– 11518, DOI: 10.1021/jacs.8b07632There is no corresponding record for this reference.
- 62(a) Li, M.; Berritt, S.; Matuszewski, L.; Deng, G.; Pascual-Escudero, A.; Panetti, G. B.; Poznik, M.; Yang, X.; Chruma, J. J.; Walsh, P. J. Transition-Metal-Free Radical C(sp3)–C(sp2) and C(sp3)–C(sp3) Coupling Enabled by 2-Azaallyls as Super-Electron-Donors and Coupling-Partners. J. Am. Chem. Soc. 2017, 139 (45), 16327– 16333, DOI: 10.1021/jacs.7b09394There is no corresponding record for this reference.(b) Li, M.; Gutierrez, O.; Berritt, S.; Pascual-Escudero, A.; Yeşilçimen, A.; Yang, X.; Adrio, J.; Huang, G.; Nakamaru-Ogiso, E.; Kozlowski, M. C.; Walsh, P. J. Transition-metal-free Chemo- and Regioselective Vinylation of Azaallyls. Nat. Chem. 2017, 9 (10), 997– 1004, DOI: 10.1038/nchem.2760There is no corresponding record for this reference.(c) Zhang, L.; Liu, Z.; Tian, X.; Zi, Y.; Duan, S.; Fang, Y.; Chen, W.; Jing, H.; Yang, L.; Yang, X. Transition-Metal-Free C(sp3)–H Coupling of Cycloalkanes Enabled by Single-Electron Transfer and Hydrogen Atom Transfer. Org. Lett. 2021, 23 (5), 1714– 1719, DOI: 10.1021/acs.orglett.1c00135There is no corresponding record for this reference.(d) Liu, Z.; Li, M.; Deng, G.; Wei, W.; Feng, P.; Zi, Q.; Li, T.; Zhang, H.; Yang, X.; Walsh, P. J. Transition-metal-free C(sp3)–H/C(sp3)–H Dehydrogenative Coupling of Saturated Heterocycles with N-benzyl Imines. Chem. Sci. 2020, 11 (29), 7619– 7625, DOI: 10.1039/D0SC00031KThere is no corresponding record for this reference.
- 63(a) Lei, Y.; Yang, J.; Qi, R.; Wang, S.; Wang, R.; Xu, Z. Arylation of Benzyl Amines with Aromatic Nitriles. Chem. Commun. 2018, 54 (84), 11881– 11884, DOI: 10.1039/C8CC06408CThere is no corresponding record for this reference.(b) Tang, S.; Zhang, X.; Sun, J.; Niu, D.; Chruma, J. J. 2-Azaallyl Anions, 2-Azaallyl Cations, 2-Azaallyl Radicals, and Azomethine Ylides. Chem. Rev. 2018, 118 (20), 10393– 10457, DOI: 10.1021/acs.chemrev.8b00349There is no corresponding record for this reference.(c) Wang, Q.; Poznik, M.; Li, M.; Walsh, P. J.; Chruma, J. J. 2-Azaallyl Anions as Light-Tunable Super-Electron-Donors: Coupling with Aryl Fluorides, Chlorides, and Bromides. Adv. Synth. Catal. 2018, 360 (15), 2854– 2868, DOI: 10.1002/adsc.201800396There is no corresponding record for this reference.
- 64Jiang, Y.; Liu, D.; Rotella, M. E.; Deng, G.; Liu, Z.; Chen, W.; Zhang, H.; Kozlowski, M. C.; Walsh, P. J.; Yang, X. Net-1,2-Hydrogen Atom Transfer of Amidyl Radicals: Toward the Synthesis of 1,2-Diamine Derivatives. J. Am. Chem. Soc. 2023, 145 (29), 16045– 16057, DOI: 10.1021/jacs.3c04376There is no corresponding record for this reference.
- 65Ye, Y.; Cao, J.; Oblinsky, D. G.; Verma, D.; Prier, C. K.; Scholes, G. D.; Hyster, T. K. Using Enzymes to Tame Nitrogen-Centred Radicals for Enantioselective Hydroamination. Nat. Chem. 2023, 15 (2), 206– 212, DOI: 10.1038/s41557-022-01083-zThere is no corresponding record for this reference.
- 66Marshall, C. M.; Federice, J. G.; Bell, C. N.; Cox, P. B.; Njardarson, J. T. An Update on the Nitrogen Heterocycle Compositions and Properties of U.S. FDA-Approved Pharmaceuticals (2013–2023). J. Med. Chem. 2024, 67 (14), 11622– 11655, DOI: 10.1021/acs.jmedchem.4c01122There is no corresponding record for this reference.
- 67(a) Nesterov, V.; Reiter, D.; Bag, P.; Frisch, P.; Holzner, R.; Porzelt, A.; Inoue, S. NHCs in Main Group Chemistry. Chem. Rev. 2018, 118 (19), 9678– 9842, DOI: 10.1021/acs.chemrev.8b00079There is no corresponding record for this reference.(b) Peris, E. Smart N-Heterocyclic Carbene Ligands in Catalysis. Chem. Rev. 2018, 118 (19), 9988– 10031, DOI: 10.1021/acs.chemrev.6b00695There is no corresponding record for this reference.
- 68Ye, Q.; Neo, W. T.; Lin, T.; Song, J.; Yan, H.; Zhou, H.; Shah, K. W.; Chua, S. J.; Xu, J. Pyrrolophthalazine Dione (PPD)-Based Donor–Acceptor Polymers as High Performance Electrochromic Materials. Polym. Chem. 2015, 6 (9), 1487– 1494, DOI: 10.1039/C4PY01608DThere is no corresponding record for this reference.
- 69(a) Deiters, A.; Martin, S. F. Synthesis of Oxygen- and Nitrogen-Containing Heterocycles by Ring-Closing Metathesis. Chem. Rev. 2004, 104 (5), 2199– 2238, DOI: 10.1021/cr0200872There is no corresponding record for this reference.(b) Patil, N. T.; Yamamoto, Y. Coinage Metal-Assisted Synthesis of Heterocycles. Chem. Rev. 2008, 108 (8), 3395– 3442, DOI: 10.1021/cr050041jThere is no corresponding record for this reference.(c) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Formal [4 + 1] Annulation Reactions in the Synthesis of Carbocyclic and Heterocyclic Systems. Chem. Rev. 2015, 115 (11), 5301– 5365, DOI: 10.1021/cr5006974There is no corresponding record for this reference.
- 70Deeksha; Singh, R. Aza-oxyallyl Cations and Their Applications in (3 m) Cycloaddition Reactions. Eur. J. Org. Chem. 2022, 2022 (47), e202201043 DOI: 10.1002/ejoc.202201043There is no corresponding record for this reference.
- 71Lengyel, I.; Sheehan, J. C. α-Lactams (Aziridinones). Angew. Chem., Int. Ed. 1968, 7 (1), 25– 36, DOI: 10.1002/anie.196800251There is no corresponding record for this reference.
- 72Kikugawa, Y.; Shimada, M.; Kato, M.; Sakamoto, T. A New Synthesis of N-Alkoxy-2-ethoxyarylacetamides from N-Alkoxy-N-chloroarylacetamides with Triethylamine in Ethanol. Chem. Pharm. Bull. 1993, 41 (12), 2192– 2194, DOI: 10.1248/cpb.41.2192There is no corresponding record for this reference.
- 73Jeffrey, C. S.; Barnes, K. L.; Eickhoff, J. A.; Carson, C. R. Generation and Reactivity of Aza-Oxyallyl Cationic Intermediates: Aza-[4 + 3] Cycloaddition Reactions for Heterocycle Synthesis. J. Am. Chem. Soc. 2011, 133 (20), 7688– 7691, DOI: 10.1021/ja201901dThere is no corresponding record for this reference.
- 74Acharya, A.; Eickhoff, J. A.; Jeffrey, C. S. Intramolecular Aza-[4 + 3] Cycloaddition Reactions of α-Halohydroxamates. Synthesis 2013, 45 (13), 1825– 1836, DOI: 10.1055/s-0033-1338883There is no corresponding record for this reference.
- 75(a) Xuan, J.; Cao, X.; Cheng, X. Advances in Heterocycle Synthesis via [3+m]-Cycloaddition Reactions Involving an Azaoxyallyl Cation as the key Intermediate. Chem. Commun. 2018, 54 (41), 5154– 5163, DOI: 10.1039/C8CC00787JThere is no corresponding record for this reference.(b) Fantinati, A.; Zanirato, V.; Marchetti, P.; Trapella, C. The Fascinating Chemistry of α-Haloamides. ChemistryOpen 2020, 9 (2), 100– 170, DOI: 10.1002/open.201900220There is no corresponding record for this reference.(c) El Bouakher, A.; Martel, A.; Comesse, S. α-Halogenoacetamides: Versatile and Efficient Tools for the Synthesis of Complex Aza-heterocycles. Org. Biomol. Chem. 2019, 17 (37), 8467– 8485, DOI: 10.1039/C9OB01683JThere is no corresponding record for this reference.
- 76Ansari, A. J.; Yadav, A.; Mukherjee, A.; Sathish, E.; Nagesh, K.; Singh, R. Metal Free Amination of Congested and Functionalized Alkyl Bromides at Room Temperature. Chem. Commun. 2020, 56 (35), 4804– 4807, DOI: 10.1039/D0CC00826EThere is no corresponding record for this reference.
- 77Kwon, Y.; Choi, S.; Jang, H. S.; Kim, S.-G. Rapid Access to Hindered α-Amino Acid Derivatives and Benzodiazepin-3-ones from Aza-Oxyallyl Cations. Org. Lett. 2020, 22 (4), 1420– 1425, DOI: 10.1021/acs.orglett.0c00023There is no corresponding record for this reference.
- 78Lee, C. Y.; Kim, S.-G. Metal-free Nucleophilic α-Azidation of α-Halohydroxamates with Azidotrimethylsilane. Asian J. Org. Chem. 2021, 10 (12), 3257– 3260, DOI: 10.1002/ajoc.202100556There is no corresponding record for this reference.
- 79Chae Son, E.; Kim, S.-G. Metal-free Nucleophilic Alkoxylation of in Situ-Generated Azaoxyallyl Cations: Synthesis of Hindered Dialkyl Ether Derivatives. Asian J. Org. Chem. 2020, 9 (6), 914– 917, DOI: 10.1002/ajoc.202000173There is no corresponding record for this reference.
- 80Yoon Lee, C.; Kim, S.-G. Highly Efficient DMSO-Promoted α-Hydrolysis of α-Halohydroxamates under Mild Conditions. Eur. J. Org. Chem. 2021, 2021 (10), 1607– 1614, DOI: 10.1002/ejoc.202100088There is no corresponding record for this reference.
- 81Taily, I. M.; Saha, D.; Banerjee, P. Aza-Oxyallyl Cation Driven 3-Amido Oxetane Rearrangement to 2-Oxazolines: Access to Oxazoline Amide Ethers. J. Org. Chem. 2022, 87 (5), 2155– 2166, DOI: 10.1021/acs.joc.1c03108There is no corresponding record for this reference.
- 82Bera, T.; Singh, B.; Jana, M.; Saha, J. Access to 3,3′-Disubstituted Peroxyoxindole Derivatives and α-Peroxyamides via Azaoxyallyl Cation-guided Addition of Hydroperoxides. Chem. Commun. 2022, 58 (54), 7538– 7541, DOI: 10.1039/D2CC02378DThere is no corresponding record for this reference.
- 83Deeksha; Sathish, E.; Kiran; Singh, R. Access to Sterically Hindered Thioethers (α-Thioamides) Under Mild Conditions Using α-Halohydroxamates: Application toward 1,4-Benzothiazinones and 4,1-Benzothiazepinones. J. Org. Chem. 2023, 88 (2), 901– 908, DOI: 10.1021/acs.joc.2c02274There is no corresponding record for this reference.
- 84Gao, J.; Hu, M.; Wang, Y.; Luo, S.; Mi, X. Transition-Metal-Free Direct α-Arylation of Weinreb-type Amides with Arylboronic Acids through Aza-oxyallyl Cation Intermediates. J. Org. Chem. 2024, 89 (18), 13730– 13734, DOI: 10.1021/acs.joc.4c01391There is no corresponding record for this reference.
- 85Graham, S. L.; Scholz, T. H. A New Mode of Reactivity of N-methoxy-N-methylamides with Strongly Basic Reagents. Tetrahedron Lett. 1990, 31 (44), 6269– 6272, DOI: 10.1016/S0040-4039(00)97039-4There is no corresponding record for this reference.
- 86Keck, G. E.; McHardy, S. F.; Murry, J. A. Some Unusual Reactions of Weinreb Amides. Tetrahedron Lett. 1993, 34 (39), 6215– 6218, DOI: 10.1016/S0040-4039(00)73713-0There is no corresponding record for this reference.
- 87Mattingly, P. G.; Miller, M. J. Titanium Trichloride Reduction of Substituted N-hydroxy-2-azetidinones and Other Hydroxamic Acids. J. Org. Chem. 1980, 45 (3), 410– 415, DOI: 10.1021/jo01291a007There is no corresponding record for this reference.
- 88Ritter, A. R.; Miller, M. J. Amino Acid-Derived Chiral Acyl Nitroso Compounds: Diastereoselectivity in Intermolecular Hetero Diels-Alder Reactions. J. Org. Chem. 1994, 59 (16), 4602– 4611, DOI: 10.1021/jo00095a040There is no corresponding record for this reference.
- 89Keck, G. E.; Wager, T. T.; McHardy, S. F. Reductive Cleavage of N–O Bonds in Hydroxylamines and Hydroxamic acid Derivatives Using Samarium Diiodide. Tetrahedron 1999, 55 (40), 11755– 11772, DOI: 10.1016/S0040-4020(99)00486-XThere is no corresponding record for this reference.
- 90Fukuzawa, H.; Ura, Y.; Kataoka, Y. Ruthenium-Catalyzed Reduction of N-alkoxy- and N-hydroxyamides. J. Organomet. Chem. 2011, 696 (23), 3643– 3648, DOI: 10.1016/j.jorganchem.2011.08.026There is no corresponding record for this reference.
- 91Wang, S.; Zhao, X.; Zhang-Negrerie, D.; Du, Y. Reductive Cleavage of the N–O Bond: Elemental Sulfur-Mediated Conversion of N-alkoxyamides to Amides. Org. Chem. Front. 2019, 6 (3), 347– 351, DOI: 10.1039/C8QO01052HThere is no corresponding record for this reference.
- 92Cutulic, S. P. Y.; Murphy, J. A.; Farwaha, H.; Zhou, S.-Z.; Chrystal, E. Metal-Free Reductive Cleavage of N–O Bonds in Weinreb Amides by an Organic Neutral Super-Electron Donor. Synlett 2008, 2008 (14), 2132– 2136, DOI: 10.1055/s-2008-1078240There is no corresponding record for this reference.
- 93(a) Shi, Q.; Liu, W. H. Reactivity Umpolung of Tertiary Amide Enabled by Catalytic Reductive Stannylation. Angew. Chem., Int. Ed. 2023, 62 (37), e202309567 DOI: 10.1002/anie.202309567There is no corresponding record for this reference.(b) Shi, Q.; Liu, W. H. Reactivity Umpolung of Amides in Organic Synthesis. Asian J. Org. Chem. 2023, 12 (12), e202300473 DOI: 10.1002/ajoc.202300473There is no corresponding record for this reference.(c) Seebach, D.; Corey, E. J. Generation and Synthetic Applications of 2-Lithio-1,3-dithianes. J. Org. Chem. 1975, 40 (2), 231– 237, DOI: 10.1021/jo00890a018There is no corresponding record for this reference.
- 94(a) Wu, Y.; Hu, L.; Li, Z.; Deng, L. Catalytic Asymmetric Umpolung Reactions of Imines. Nature 2015, 523 (7561), 445– 450, DOI: 10.1038/nature14617There is no corresponding record for this reference.(b) Miyoshi, T.; Miyakawa, T.; Ueda, M.; Miyata, O. Nucleophilic α-Arylation and α-Alkylation of Ketones by Polarity Inversion of N-Alkoxyenamines: Entry to the Umpolung Reaction at the α-Carbon Position of Carbonyl Compounds. Angew. Chem., Int. Ed. 2011, 50 (4), 928– 931, DOI: 10.1002/anie.201004374There is no corresponding record for this reference.(c) Quiclet-Sire, B.; Tölle, N.; Zafar, S. N.; Zard, S. Z. Oxime Derivatives as α-Electrophiles. From α-Tetralone Oximes to Tetracyclic Frameworks. Org. Lett. 2011, 13 (12), 3266– 3269, DOI: 10.1021/ol2012204There is no corresponding record for this reference.(d) Sukhorukov, A. Y. Umpolung of Enamines: An Overview on Strategies and Synthons. Synlett 2020, 31 (05), 439– 449, DOI: 10.1055/s-0037-1610741There is no corresponding record for this reference.
- 95Dai, X.-J.; Li, C.-C.; Li, C.-J. Carbonyl Umpolung as an Organometallic Reagent Surrogate. Chem. Soc. Rev. 2021, 50 (19), 10733– 10742, DOI: 10.1039/D1CS00418BThere is no corresponding record for this reference.
- 96(a) Trost, B. M.; Li, C.-J. Novel “Umpolung” in C–C Bond Formation Catalyzed by Triphenylphosphine. J. Am. Chem. Soc. 1994, 116 (7), 3167– 3168, DOI: 10.1021/ja00086a074There is no corresponding record for this reference.(b) Guo, H.; Fan, Y. C.; Sun, Z.; Wu, Y.; Kwon, O. Phosphine Organocatalysis. Chem. Rev. 2018, 118 (20), 10049– 10293, DOI: 10.1021/acs.chemrev.8b00081There is no corresponding record for this reference.
- 97Bugaut, X.; Glorius, F. Organocatalytic Umpolung: N-heterocyclic Carbenes and Beyond. Chem. Soc. Rev. 2012, 41 (9), 3511– 3522, DOI: 10.1039/c2cs15333eThere is no corresponding record for this reference.
- 98Hari, D. P.; Caramenti, P.; Waser, J. Cyclic Hypervalent Iodine Reagents: Enabling Tools for Bond Disconnection via Reactivity Umpolung. Acc. Chem. Res. 2018, 51 (12), 3212– 3225, DOI: 10.1021/acs.accounts.8b00468There is no corresponding record for this reference.
- 99(a) Masson, G.; Py, S.; Vallée, Y. Samarium Diiodide-Induced Reductive Cross-Coupling of Nitrones with Aldehydes and Ketones. Angew. Chem., Int. Ed. 2002, 41 (10), 1772– 1775, DOI: 10.1002/1521-3773(20020517)41:10<1772::AID-ANIE1772>3.0.CO;2-QThere is no corresponding record for this reference.(b) Masson, G.; Cividino, P.; Py, S.; Vallée, Y. SmI2-Induced Umpolung of the C–N Bond: First Reductive Conjugate Addition of Nitrones to α,β-Unsaturated Esters. Angew. Chem., Int. Ed. 2003, 42 (20), 2265– 2268, DOI: 10.1002/anie.200250480There is no corresponding record for this reference.(c) Riber, D.; Skrydstrup, T. SmI2-Promoted Radical Addition of Nitrones to α,β-Unsaturated Amides and Esters: Synthesis of γ-Amino Acids via a Nitrogen Equivalent to the Ketyl Radical. Org. Lett. 2003, 5 (2), 229– 231, DOI: 10.1021/ol027386yThere is no corresponding record for this reference.
- 100(a) Kaiser, D.; Bauer, A.; Lemmerer, M.; Maulide, N. Amide Activation: an Emerging Tool for Chemoselective Synthesis. Chem. Soc. Rev. 2018, 47 (21), 7899– 7925, DOI: 10.1039/C8CS00335AThere is no corresponding record for this reference.(b) Spieß, P.; Shaaban, S.; Kaiser, D.; Maulide, N. New Strategies for the Functionalization of Carbonyl Derivatives via α-Umpolung: From Enolates to Enolonium Ions. Acc. Chem. Res. 2023, 56 (12), 1634– 1644, DOI: 10.1021/acs.accounts.3c00171There is no corresponding record for this reference.
- 101Bladon, C. M.; Kirby, G. W. Formation of Aziridinones (α-lactams) from Hydroxamic Derivatives. J. Chem. Soc., Chem. Commun. 1982, (24), 1402– 1404, DOI: 10.1039/C39820001402There is no corresponding record for this reference.
- 102(a) Hoffman, R. V.; Nayyar, N. K.; Klinekole, B. W. Efficient Conversion of O-sulfonylated Arylacetohydroxamic Acids to 2-Substituted Secondary Amides. J. Am. Chem. Soc. 1992, 114 (15), 6262– 6263, DOI: 10.1021/ja00041a064There is no corresponding record for this reference.(b) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Base-promoted Reaction of O-sulfonylated Hydroxamic Acids with Nucleophiles. A New Method for the Synthesis of alpha-Substituted Amides. J. Org. Chem. 1992, 57 (21), 5700– 5707, DOI: 10.1021/jo00047a024There is no corresponding record for this reference.(c) Hoffman, R. V.; Nayyar, N. K. Preparation of alpha-Alkoxy Amides and alpha-Hydrazino Amides by Base-Promoted Reactions of N-Sulfonyloxy Amides. J. Org. Chem. 1995, 60 (21), 7043– 7046, DOI: 10.1021/jo00126a072There is no corresponding record for this reference.(d) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Synthesis of alpha-Azido and alpha-Amino Amides from N-[(methylsulfonyl)oxy]amides. J. Org. Chem. 1993, 58 (9), 2355– 2359, DOI: 10.1021/jo00061a004There is no corresponding record for this reference.(e) Hoffman, R. V.; Nayyar, N. K.; Chen, W. Origins of Regioselectivity in the Reactions of alpha-Lactams with Nucleophiles. J. Org. Chem. 1995, 60 (13), 4121– 4125, DOI: 10.1021/jo00118a032There is no corresponding record for this reference.(f) Hoffman, R. V.; Nayyar, N. K. Reaction of Hydrazines with alpha-Lactams for the Preparation of 1,2,4-Triazine-3,6-diones and Aza-Urea Peptide Mimetics. J. Org. Chem. 1995, 60 (18), 5992– 5994, DOI: 10.1021/jo00123a048There is no corresponding record for this reference.
- 103Hoffman, R. V.; Zhao, Z.; Costales, A.; Clarke, D. Origins of Regioselectivity in the Reactions of α-Lactams with Nucleophiles. Two Distinct Acid-Catalyzed Pathways Involving O- and N-Protonation. J. Org. Chem. 2002, 67 (15), 5284– 5294, DOI: 10.1021/jo020246hThere is no corresponding record for this reference.
- 104Han, S.; Guo, Y.; Zhang, W.; Chen, J.; Wang, Z.; Zeng, Y.-F. Regioselective Heterofunctionalization of alpha-Aryl Amides with Heteroatom Nucleophiles via Electrophilic Activation. Org. Chem. Front. 2023, 10 (21), 5512– 5518, DOI: 10.1039/D3QO01174GThere is no corresponding record for this reference.
- 105Zhang, W.; Han, S.; Zeng, Y.-F.; Han, F. Metal-Free α-(Hetero)Arylation of Amides Enabled Access to Diaryl Acetamides via Umpolung. Eur. J. Org. Chem. 2024, 27 (47), e202400905 DOI: 10.1002/ejoc.202400905There is no corresponding record for this reference.
- 106Wang, R.; An, S.; Xin, Y.-X.; Jiang, Y.-Y.; Liu, W. H. Redox-Neutral Umpolung Synthesis of α-Functionalized Amides. JACS Au 2024, 4 (11), 4435– 4444, DOI: 10.1021/jacsau.4c00767There is no corresponding record for this reference.
- 107An, S.; Lai, G.; Liu, W. H. Catalytic Dehydrogenative Synthesis of α,β-Unsaturated Secondary Amides without External Oxidants. Chem. Sci. 2024, 15 (37), 15385– 15390, DOI: 10.1039/D4SC04419CThere is no corresponding record for this reference.
- 108Wang, R.; Jiang, Q.; Jiang, L.; Liu, W. H. Nucleophilic α- and β-Additions Enable Redox-Neutral Aziridination of Conjugated Hydroxamates. J. Am. Chem. Soc. 2025, 147 (30), 26298– 26306, DOI: 10.1021/jacs.5c04286There is no corresponding record for this reference.
- 109Zhang, W.; Han, S.; Zhang, S.-Y.; Jiang, D.-X.; Chen, J.; Wang, Z.; Zeng, Y.-F. Synthesis of Indolyl Pyrroloindolines via a Cascade Arylation/Cyclization of Indole Acetamides with 3-Substituted Indoles. Org. Chem. Front. 2024, 11 (18), 5167– 5171, DOI: 10.1039/D4QO01118JThere is no corresponding record for this reference.
- 110Tang, X.; Zhang, W.; Jiang, D.-X.; Chen, Z.; Shen, L.; Tian, Y.; Chen, J.; Wang, Z.; Zeng, Y.-F. Access to Divergent Indolyl Pyrrolones via [3 + 2] Annulation of Indolyl Hydroxamates with Phosphonium Ylides. Org. Lett. 2025, 27 (43), 12137– 12141, DOI: 10.1021/acs.orglett.5c03964There is no corresponding record for this reference.
- 111Hirner, S.; Panknin, O.; Edefuhr, M.; Somfai, P. Synthesis of Aryl Glycines by the α Arylation of Weinreb Amides. Angew. Chem., Int. Ed. 2008, 47 (10), 1907– 1909, DOI: 10.1002/anie.200704689There is no corresponding record for this reference.
- 112Hirner, S.; Kirchner, D. K.; Somfai, P. Synthesis of α-Amino Acids by Umpolung of Weinreb Amide Enolates. Eur. J. Org. Chem. 2008, 2008 (33), 5583– 5589, DOI: 10.1002/ejoc.200800683There is no corresponding record for this reference.
- 113Takeda, N.; Futaki, E.; Kobori, Y.; Ueda, M.; Miyata, O. Nucleophilic Arylation of N,O-Ketene Acetals with Triaryl Aluminum Reagents: Access to α-Aryl Amides through an Umpolung Process. Angew. Chem., Int. Ed. 2017, 56 (51), 16342– 16346, DOI: 10.1002/anie.201708665There is no corresponding record for this reference.
- 114Gassman, P. G. Nitrenium ions. Acc. Chem. Res. 1970, 3 (1), 26– 33, DOI: 10.1021/ar50025a004There is no corresponding record for this reference.
- 115Novak, M.; Xu, L.; Wolf, R. A. Nitrenium Ions from Food-Derived Heterocyclic Arylamine Mutagens. J. Am. Chem. Soc. 1998, 120 (7), 1643– 1644, DOI: 10.1021/ja974222hThere is no corresponding record for this reference.
- 116O’Neil, L. G.; Bower, J. F. Electrophilic Aminating Agents in Total Synthesis. Angew. Chem., Int. Ed. 2021, 60 (49), 25640– 25666, DOI: 10.1002/anie.202102864There is no corresponding record for this reference.
- 117(a) Kawase, M.; Kitamura, T.; Kikugawa, Y. Electrophilic Aromatic Substitution with N-Methoxy-N-acylnitrenium Ions Generated from N-Chloro-N-methoxy Amides: Syntheses of Nitrogen Heterocyclic Compounds Bearing an N-Methoxy Amide Group. J. Org. Chem. 1989, 54 (14), 3394– 3403, DOI: 10.1021/jo00275a027There is no corresponding record for this reference.(b) Glover, S. A.; Goosen, A.; McClei, C. V.; Schoonraad, J. L. N-Alkoxy-N-acylnitrenium Ions in Intramolecular Aromatic Addition Reactions. Tetrahedron 1987, 43 (11), 2577– 2592, DOI: 10.1016/S0040-4020(01)81665-3There is no corresponding record for this reference.
- 118Sakamoto, R.; Inada, T.; Selvakumar, S. Efficient Photolytic C–H Bond Functionalization of Alkylbenzene with Hypervalent Iodine(iii) Reagent. Chem. Commun. 2016, 52 (19), 3758– 3761, DOI: 10.1039/C5CC07647AThere is no corresponding record for this reference.
- 119Maiti, S.; Alam, M. T.; Bal, A.; Mal, P. Nitrenium Ions from Amine-Iodine(III) Combinations. Adv. Synth. Catal. 2019, 361 (19), 4401– 4425, DOI: 10.1002/adsc.201900441There is no corresponding record for this reference.
- 120Glover, S. A.; Scott, A. P. MNDO Properties of Heteroatom and Phenyl Substituted Nitrenium Ions. Tetrahedron 1989, 45 (6), 1763– 1776, DOI: 10.1016/S0040-4020(01)80041-7There is no corresponding record for this reference.
- 121Kikugawa, Y.; Kawase, M. An Electrophilic Aromatic Substitution by N-Methoxyamides via Hypervalent Iodine Intermediates. Chem. Lett. 1990, 19 (4), 581– 582, DOI: 10.1246/cl.1990.581There is no corresponding record for this reference.
- 122Miyazawa, E.; Sakamoto, T.; Kikugawa, Y. Synthesis of Spiro-fused Nitrogen Heterocyclic Compounds via N-Methoxy-N-acylnitrenium Ions Using Phenyliodine (III) bis (trifluoroacetate) in Trifluoroethanol. Heterocycles 2003, 59 (1), 149– 160, DOI: 10.3987/COM-02-S7There is no corresponding record for this reference.
- 123Zhu, C.; Liang, Y.; Hong, X. Iodoarene-Catalyzed Stereospecific Intramolecular sp3 C–H Amination: Reaction Development and Mechanistic Insights. J. Am. Chem. Soc. 2015, 137 (24), 7564– 7567, DOI: 10.1021/jacs.5b03488There is no corresponding record for this reference.
- 124Wang, Y.; Yang, M.; Sun, Y.-Y. An Efficient Approach for 3,3-Disubstituted Oxindoles Synthesis: Aryl Iodine Catalyzed Intramolecular C–N Bond Oxidative Cross-Coupling. Org. Lett. 2021, 23 (22), 8750– 8754, DOI: 10.1021/acs.orglett.1c03224There is no corresponding record for this reference.
- 125Das, T. K.; Ghosh, A.; Grimm, Z. Organocatalytic Electrophilic Arene Amination: Rapid Synthesis of 2-Quinolones. Chem. - Eur. J. 2025, 31 (6), e202403524 DOI: 10.1002/chem.202403524There is no corresponding record for this reference.
- 126Sasa, H.; Mori, K.; Kikushima, K. μ-Oxo-Hypervalent-Iodine-Catalyzed Oxidative C–H Amination for Synthesis of Benzolactam Derivatives. Chem. Pharm. Bull. 2022, 70 (2), 106– 110, DOI: 10.1248/cpb.c21-00980There is no corresponding record for this reference.
- 127Deeksha; Singh, R. A Modular Approach to Access 1,4-Benzoxazinones, Spirooxazolidinones, and 4,1-Benzoxazepinones via Oxidative Aryl C–N Bond Formation Using Catalytic Hypervalent Iodine. Org. Lett. 2025, 27 (50), 14112– 14117, DOI: 10.1021/acs.orglett.5c04757There is no corresponding record for this reference.
- 128Parveen, N.; Aziz, M. N.; Gout, D.; Lovely, C. J. Dearomatization Reactions of N-OMe Ureas. Org. Lett. 2025, 27 (40), 11307– 11312, DOI: 10.1021/acs.orglett.5c03607There is no corresponding record for this reference.
- 129Ortiz, G. X., Jr.; Hemric, B. N.; Wang, Q. Direct and Selective 3-Amidation of Indoles Using Electrophilic N-[(Benzenesulfonyl)oxy]amides. Org. Lett. 2017, 19 (6), 1314– 1317, DOI: 10.1021/acs.orglett.7b00358There is no corresponding record for this reference.
- 130Barbor, J. P.; Nair, V. N.; Sharp, K. R. Development of a Nickel-Catalyzed N–N Coupling for the Synthesis of Hydrazides. J. Am. Chem. Soc. 2023, 145 (28), 15071– 15077, DOI: 10.1021/jacs.3c04834There is no corresponding record for this reference.
- 131Fang, W.; Luo, Z.-W.; Wang, Y.-C. SN2 Reaction at the Amide Nitrogen Center Enables Hydrazide Synthesis. Angew. Chem., Int. Ed. 2024, 63 (14), e202317570 DOI: 10.1002/anie.202317570There is no corresponding record for this reference.
- 132Yang, H.; Zhang, X.; Cao, F.-Y. Reverse Polarity of Amide Nitrogen Enables Expedient Access to N-Cyano Amides. Nat. Commun. 2025, 16 (1), 7706 DOI: 10.1038/s41467-025-63052-7There is no corresponding record for this reference.
- 133Malik, M.; Senatore, R.; Langer, T. Base-Mediated Homologative Rearrangement of Nitrogen–oxygen Bonds of N-Methyl-N-oxyamides. Chem. Sci. 2023, 14 (37), 10140– 10146, DOI: 10.1039/D3SC03216GThere is no corresponding record for this reference.
- 134Li, G.; Lübken, T.; Plietker, B. A Ru-Catalyzed Desulfonylative C-Arylation of O-Tosyl-hydroxamates. Chem. Commun. 2024, 60 (7), 851– 853, DOI: 10.1039/D3CC05825EThere is no corresponding record for this reference.
- 135Wu, J.-C.; Yang, H.; Luo, Z.-W. Direct N-Methyl C–H Esterification of O-Tosyl hydroxamates to Enable Facile Synthesis of Hemiaminal Esters. Chem. Commun. 2025, 61 (57), 10542– 10545, DOI: 10.1039/D5CC02570BThere is no corresponding record for this reference.



