
Lithium Recovery from Brines: A Comprehensive Review of Advanced Separation TechnologiesClick to copy article linkArticle link copied!
- Hemant Mittal*Hemant Mittal*E-mail: [email protected] (Hemant Mittal).DEWA R&D Center, Dubai Electricity & Water Authority (DEWA), P.O. Box 564, Dubai, United Arab EmiratesMore by Hemant Mittal
- Syed Nasir Shah*Syed Nasir Shah*Email: [email protected], [email protected] (Syed Nasir Shah).DEWA R&D Center, Dubai Electricity & Water Authority (DEWA), P.O. Box 564, Dubai, United Arab EmiratesMore by Syed Nasir Shah
- Maryam AlsuwaidiMaryam AlsuwaidiDEWA R&D Center, Dubai Electricity & Water Authority (DEWA), P.O. Box 564, Dubai, United Arab EmiratesMore by Maryam Alsuwaidi
- Akram AlfantaziAkram AlfantaziDepartment of Chemical and Petroleum Engineering, Khalifa University of Science and Technology, P.O. Box 127788, Abu Dhabi, United Arab EmiratesEmirates Nuclear Technology Center (ENTC), Khalifa University of Science and Technology, P.O. Box 127788, Abu Dhabi, United Arab EmiratesMore by Akram Alfantazi
Abstract
The increasing lithium (Li) demand, attributable to its essential function in energy storage applications, has necessitated the advancement in Li extraction and recovery methods. Brine-based Li extraction presents a cost-effective and scalable solution; however, challenges such as high magnesium-to-Li (Mg/Li) ratios and environmental concerns necessitate innovative approaches. This review comprehensively examines various Li recovery techniques from brine, focusing on precipitation, adsorption and ion exchange, membrane separation, electrochemical separation, solvent extraction, and hybrid technologies. Precipitation methods, including carbonate and aluminate precipitation, offer straightforward processing but face selectivity challenges in high Mg/Li ratio brines. Adsorption and ion exchange techniques, particularly Li-ion sieves and layered double hydroxides, have demonstrated high selectivity and efficiency. Electrochemical methods present promising low-energy alternatives for Li separation. Furthermore, solvent extraction and advanced membrane-based techniques provide additional pathways for Li recovery, enhancing the yield and purity. This Perspective highlights recent advancements, process optimizations, and emerging strategies aimed at improving Li recovery while reducing environmental impact. Comparative assessment of these techniques is provided, considering factors such as the Mg/Li ratio, operational feasibility, energy consumption, and scalability. Special emphasis is placed on emerging strategies such as engineered sorbents, electrochemical separation, and advanced membrane filtration, which offer sustainable alternatives to traditional methods. Additionally, this review bridges the gap between laboratory-scale research and industrial applications, outlining key challenges and potential solutions for Li extraction. This Review provides a roadmap for developing more efficient, sustainable, and scalable Li extraction processes. The insights presented here aid researchers, industry professionals, and policymakers in advancing Li recovery technologies to meet growing global demand.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
Figure 1
Figure 1. Publication counts retrieved from the Scopus database using the search query: TITLE-ABS-KEY (lithium AND extraction) AND PUBYEAR > 1999 AND PUBYEAR < 2026.
2. Different Methods of Li-Ion Recovery
Figure 2
Figure 2. Different methods or techniques for Li extraction from brine.
2.1. Precipitation
Figure 3
Figure 3. Schematic illustration of lithium recovery via chemical precipitation from high-Li brines, highlighting the process steps, recovered lithium products, and key advantages and limitations of the method.
Figure 4
Figure 4. (a) Flowsheet for the crystallization-precipitation method adapted with permission from ref (80). Copyright 2020, Elsevier. (b, c) Effects of the reaction time on (b) percentage recovery and (c) precipitation rates; (d, e) effects of stirring rate on (d) percentage recovery and (e) precipitation rates of Li and Mg-ion recovery adapted with permission from ref (81). Copyright 2019, Elsevier.
| reagents/precipitant | brine source | Li recovery rate (%) | temperature (°C) | year | reference |
|---|---|---|---|---|---|
| Sodium carbonite, calcium hydroxide | Uyuni Salar brine | More than 90% | 90 °C | 2012 | (68) |
| Na2CO3, EDTA-Li, mineral acid | Salt Lake brine from Damxungcuo saline lake | 99.6% | 60 °C | 2014 | (71) |
| Al–Ca alloy | Salt Lake brine | 94.6% | 70 °C | 2015 | (73) |
| Na3PO4 | Salt Lake brine | 91.94% | 90 °C | 2018 | (79) |
| KCl and Na2HPO4 | Salt Lake brine | 93.2% | - | 2020 | (80) |
| Sodium metasilicate nonahydrate | Simulated Salt Lake brine | 86.73% | 25 ± 2 | 2019 | (81) |
| TBP in methyl isobutyl ketone | Salt Lake brine | 98.9% | 23–27 | 2016 | (82) |
| Facet engineered Li3PO4 | Salt Lake brine from Dangxiong Co Salt Lake in China | 51.62% | 30 °C | 2021 or 2022 | (83) |
| Na2CO3 | The brine source used in the study was from the Qaidam Basin. | 93.3–97.2% | 60 °C | 2023 | (84) |
| Na2CO3, Li3PO4 | α-spodumene from China | 93.3% | 65 °C | 2019 | (85) |
| Trisodium phosphate | Sea water brine | 40% | 40 °C | 2020 | (86) |
| Trisodium phosphate | Environmental brine from the Dead Sea | 40% | 40 °C | 2021 | (87) |
| NaOH and CO2 | Synthetic simulated brine | 80% | 80 °C | 2022 | (88) |
| AlCl3·6H2O and NaOH | Artificial Uyuni brine | 95% | - | 2025 | (89) |
| (NH4)2SO4 | Simulated Salt-Lake brine | 74.8% | 25 °C | 2024 | (90) |
2.2. Electrochemical Extraction
Figure 5
Figure 5. Overview of lithium recovery pathways from brines: (a) conventional evaporation–precipitation processes characterized by high energy and chemical consumption; (b) electrochemical lithium extraction methods offering improved energy efficiency and selectivity; (c) lithium insertion (charging) into lithium-selective electrodes under an applied potential; (d) lithium extraction (discharging) through controlled release of Li+ ions into a recovery solution; and (e) overall advantages of insertion–extraction systems operating under mild conditions with high lithium selectivity and reduced energy and chemical requirements.
2.2.1. Electrochemical Deintercalation/Intercalation Method (EDIM)
Figure 6
Figure 6. (a–c) Schematic illustration of reactions occurring at the anode and cathode sides Li extraction device; (d, e) comparison of the anode and cathode electrode potential with pH variations; (f) analysis of Mn dissolution losses at the anode and ion intercalation capacity at the cathode adapted with permission from ref (95). Copyright 2025, Elsevier. (g) Structure of the electrolytic cell adapted with permission from ref (96). Copyright 2021, Elsevier. (h) Schematic diagram of electrodes preparation adapted with permission from ref (97). Copyright 2024, Elsevier. (i, j) Mg/Li ratio, Li+ concentration and current density for each cycle adapted with permission from ref (98). Copyright 2021, Elsevier.
2.2.2. Electrodialysis
Figure 7
Figure 7. Schematic of (a) traditional electrodialysis, (b) bipolar membrane electrodialysis, and (c) selective electrodialysis adapted with permission from ref (102). Copyright 2024, Elsevier. (d) Changes in the behavior of partially dehydrated ions and membrane pore structure at varying temperatures adapted with permission from ref (103). Copyright 2020, Elsevier.
Figure 8
Figure 8. (a) Rate of Li removal and (b) results of Li extraction adapted with permission from ref (104). Copyright 2022, Elsevier. (c) Schematic representation of the multistage integrated membrane process, which includes a single-stage NF, followed by a first-stage and second-stage S-ED adapted with permission from ref (105). Copyright 2023, Elsevier. (d, e) Illustration of schematic of Li-Mg migration in (d) batch S-ED; (e) feed and bleed mode adapted with permission from ref (106). Copyright 2021, Elsevier. (f) Complete scheme of the fabrication of LATP membrane and the Mg/Li separation adapted with permission from ref (107). Copyright 2024, Elsevier.
2.2.3. Electrochemical Ion Pumping
Figure 9
Figure 9. (a) Design of ESIP stack for continuous Li-ions separation; (b) the mechanism illustrating Li-ions separation; (c) the diffusion pathway and (d) energy barrier analysis; (e) mean square displacement of Li and Mg-ions, with diffusion coefficient shown in the inset adapted with permission from ref (108). Copyright 2024, Elsevier.
2.2.4. Capacitive Deionization (CDI)
Figure 10
Figure 10. (a) The adsorption capacity in single-salt solutions (0.05 mol/L); (b) separation factor and adsorption capacity across different Mg/Li ratios adapted with permission from ref (109). Copyright 2024, Elsevier. (c) Synthesis process of electrodes; (d) representation of HCDI device adapted with permission from ref (110). Copyright 2024, Elsevier.
| electrochemical technique | electrode material | brine type | adsorption capacity | Li recovery (%) | Mg/Li reduction | energy requirement | ref |
|---|---|---|---|---|---|---|---|
| Intercalation/Deintercalation | LiFePO4/FePO4 (olivine type) | West Taijinar natural brine | ∼32 mg/g (initial), 28 mg/g after 120 cycles | 85.3% | 54.27 to 1.65 | - | (98) |
| Spinel LiMn2O4/Li1–xMn2O4 | Da Qaidam original brine | 15 mg/g | 50–75% (single-pass); up to 83.3% after four cycles | 147.8 to 0.37 | 16 Wh/mol | (96) | |
| LiNi0.05Mn1.95O4 | Simulated brine | 25.87 mg/g | 88.2% | 60 to 2.3 | 13.42 Wh/mol | (111) | |
| LiMn2O4 thin film on Ti foil | Simulated brine | 34.3 mg/g | 83.4% (after 20 cycles) | Li/Mg selectivity ≈ 73.6 | 18.6 Wh/mol | (112) | |
| LiMn1.8Cr0.2O4 | Jieze Caka brine | 21.85 mg/g | 98.93% (100 cycles), 86.05% (500 cycles) | Improved selectivity vs Mg-ions | 2.16 Wh/mol | (113) | |
| TiO2-coated FePO4 | Authentic seawater | - | Li/(Li+Na) ≈ 50% (10 cycles) | Li/Na improved from 5 × 10–5 → 1:1 (Li selectivity 1.8 × 104) | Not stated (pulsed C/5) | (114) | |
| Porous carbon-supported LiFePO4 | Synthetic brine | 5.13 mg/cm3 ([Li-ions] = 100 mg/L) | 96% | Selective against Mg and N (Mg/Li < 0.5) | 33.52 Wh/mol | (115) | |
| LiMn2O4/λ-MnO2 | Baqiancuo Salt-Lake brine (Li = 0.33 g/L) | - | 95.0 (analytical recovery) | - | - | (116) | |
| Capacitive Deionization (CDI) | Cr-doped LiMn2O4 | Original brine | 15 mg/g | - | - | - | (113) |
| Graphene supported LiMn2O4 | Simulated Atacama brine | 4.34 mmol/g | - | Separation factor = 71.32 (Mg/Li) | - | (109) | |
| GO-encapsulated La-doped LiMn2O4 | Simulated salt-lake brine | 1.33 mmol/g | 80.4 | Separation factor = 126 (Mg/Li) | - | (117) | |
| PVDF-ethylenediamine with modified electrodes | LiCl solution | 30 mg/g | 96 | Over 0.9 current efficiency | (118) | ||
| AC/PB-20% | LiCl | 24.42 mg/g | ∼95.1% (retention after 50 cycles) | - | (119) | ||
| PM, NG | LiCl with other cations | - | 59–95.9% (Li-ions purity) | Li/Mg = 268.1 Li/Na = 44.25 | - | (120) | |
| Carbon-buffered Cr-doped LiMn1.9Cr0.1O4 | Original brine from Jiezechaka | 21 mg/g | 95.7% after 500 cycles | Increased from 1.67 to 158 | 13 Wh/mol | (110) | |
| (C-LMO) | Simulated brine | 18.1 mg/g | - | - | - | (121) | |
| Electrodialysis (ED) | M-GA/PEI | Simulated brine | - | Not explicitly stated; but high purity achieved | High Mg/Li to low (not numerically specified) | First-stage: 29–39 Wh/mol; second-stage: 11–14 Wh/mol | (105) |
| Acid-treated Li0.29La0.57TiO3 (LA-LLT) | Mixed alkaline solution (LiOH, NaOH, KOH); LIB leachate | 40 ppm of Li in 72 h | ∼100% after 72 h | Perfect selectivity vs Na, K; No Mg reported | - | (104) | |
| Selemion CSO/ASV ion-exchange membranes | High Mg/Li brine from Llullaillaco Salt Lake, Argentina | - | 90% | 9.85 to 0.57 | - | (122) | |
| LLTO ceramic ion-selective membrane | Artificial brine (LiCl, NaCl, KCl) | ∼1.07 Wh/g of Li recovered | ∼100% | Excellent Li/Na/K selectivity | - | (106) | |
| LiFePO4-based membrane | Simulated brine | ∼25.3 (from Li content in μmol/g) | ∼94% | 60 to <1 | ∼3.1 kWh/kg Li | (93) | |
| Composite IEM | Simulated brine | 1.2 mmol/g | - | High Mg/Li selectivity | - | (123) | |
| LATP-Al2O3 composite ceramic membrane, NASICON | Simulated brine | - | 77.15% | 40 to 2.1 | 47.6 kWh/kg Li2CO3 (89.6 Wh/mol of Li) | (107) | |
| C4mim][TFSI] + TBP (RTIL system as Li carrier) | Simulated brine | - | - | Mg/Li 50 to 0.5 | 16 Wh/g Li (111 Wh/mol of Li) | (124) | |
| [C4mim][TFSI] + TBP | Natural brine (West Taijinair) | - | - | 53 to 0.26 | - | (124) | |
| SPEEK/Mg–Li–MnO | Simulated brine | 15.2 mg/g | 64% (pure Li-ions); 84% (in mix ions) | Mg/Li reduced, SF = 4.82 | - | (125) | |
| Commercial monovalent selective cation-exchange membrane | Synthetic brine | - | >85% | Mg/Li SF = 2.7 (optimized) | 60–180 Wh/mol | (126) |
2.3. Adsorption/Ion Exchange
Figure 11
Figure 11. Overview of adsorption and ion-exchange–based direct lithium extraction (DLE) from brines, highlighting selective Li-ion capture, regeneration, and sustainable lithium recovery.
2.3.1. Li-Ion Sieves (LIS)
Figure 12
Figure 12. (a) The full process of the formation of ion-sieves adapted with permission from ref (137). Copyright 2020, Elsevier. (b) Process of the selective Li-ion adsorption adapted with permission from ref (22). Copyright 2024, Elsevier. (c) Plot of the adsorption kinetics; (d, e) plots of pseudo-first-order kinetics and pseudo-second-order kinetics (d) HFTO and (e) HTO. Adapted with permission from ref (143). Copyright 2024, Elsevier. (f) The method of the preparation of HMO-Ti ionic sieves and its adsorption–desorption mechanism; adapted with permission from ref (144). Copyright 2025, Elsevier.
2.3.2. Li-Aluminum Layered Double Hydroxides (LiAl-LDHs)
Figure 13
Figure 13. (a) The steering interlayer interaction strategy in LiAl-LDHs beads; (b) effect of PAN concentration on Li extraction; (c) adsorption kinetics; (d) intraparticle diffusion and (e) desorption of Li from BLDH-P and BLDH-Cl adapted with permission from ref (150). Copyright 2024, Elsevier. (f) The full scheme of binder less LiAl-LDH@NiAl-F preparation adapted with permission from ref (151). Copyright 2025, Elsevier.
2.3.3. Polymers Based Adsorbents
Figure 14
Figure 14. (a) The adsorption isotherm and kinetics plots of Li-ions adsorption onto MOF-808-12C4E adapted with permission from ref (153). Copyright 2025, Elsevier. (b) The full scheme of the synthesis of BCEEM and further copolymerization with NIPAM adapted with permission from ref (154). Copyright 2025, Elsevier. (c) Complete scheme of the synthesis of PPy@HNTs/PVA hydrogel composite adapted with permission from ref (32). Copyright 2024, Elsevier. (d) The plots of adsorption isotherm, kinetics, and multicycle use of PPy@HNTs/PVA for Li-ions extraction adapted with permission from ref (32). Copyright 2024, Elsevier.
| adsorbent | Li source | adsorption parameters | adsorption capacity (mg/g) | adsorption isotherm | adsorption kinetics | ref |
|---|---|---|---|---|---|---|
| Manganese based ion exchange nanoparticles | Synthetic brine | Dose: 2.0 g/L; temp: 70 °C | 18.0 | - | - | (128) |
| High layer charge layered double hydroxides | Synthetic brine | Temp: 30 °C | 5.45 | Sips | PSO | (130) |
| Ion-exchange resin: (K2629) | Synthetic brine | Dose: 1.0 g; temp: 25 °C | 1.84 | Langmuir | PSO | (134) |
| Ion-exchange resin: (TP207) | Synthetic brine | Dose: 1.0 g; temp: 25 °C | 2.54 | Langmuir | PSO | |
| Ion-exchange resin: (TP208) | Synthetic brine | Dose: 1.0 g; temp: 25 °C | 1.23 | Langmuir | PSO | |
| PVB- H2TiO3 | Simulated brine | pH: 9.2; adsorption time: 3 h | 12 | - | - | (139) |
| P(BCEEM-st-NiPAAm) | Simulated brine | Temp: 17 °C; pH: 10; adsorption time: 24 h | 0.075 mmol/g | - | - | (155) |
| Fe-doped H2TiO3 | Simulated brine | Dose: 0.10g; temp: 25 °C; pH: 12.0 | 34.27 | Langmuir | PSO | (143) |
| HMO-Ti-6 | Simulated brine | Dose: 0.05g; temp: 25 °C; pH: 12.0 | 47.71 | Langmuir | PSO | (144) |
| 3D-LIS-600 | LiOH | Dose: 0.5 g; temp: 25 °C; pH: 12.1 | 12.3 | - | - | (156) |
| M–T– LIS | Simulated brine | Temp: 55 °C; pH: 12.5 | 31.80 | Langmuir | PSO | (157) |
| Li/Al-LDHs | Simulated brine | Temp: 30 °C | 7.26 | Langmuir | PSO | (148) |
| BLDH-P | Simulated brine | Dose: 20g/L; temp: 30 °C | 5.26 | - | - | (150) |
| MOF-808–12C4E | Simulated brine | Dose: 5 mg | 30.4 | Langmuir | PSO, PFO | (153) |
| PEF-HTO | Geothermal brine | Dose: 0.1 g; temp: 60 °C; pH: 12 | 25.78 | Langmuir | PSO | (158) |
| PVA/HTO | Simulated brine | Dose: 20 mg; pH: 11 | 12.0 | Langmuir | PSO | (159) |
| CTS/LMO | Simulated brine | Temp: 35 °C | 11.4 | - | PSO | (160) |
| PVA/CAM-HMO | Simulated brine | Dose: 200 mg; pH: 11 | 23.26 | - | PSO | (161) |
| Cellulose/HMO | Simulated brine | Dose: 0.1 g; temp: 25 °C; pH: 10.5 | 21.64 | Langmuir | PSO | (162) |
| Porous HTO | Simulated brine | Temp: 60 °C; pH: 12 | 12.29 | Langmuir | PSO | (163) |
| PPy@HNTs-hydrogels | Simulated brine | Dose: 0.5 g; temp: 25 °C | 168.3 | Freundlich | PFO | (32) |
| HMO/PAN | Simulated brine | Temp: 25 °C; pH: 11 | 10.3 | Langmuir | - | (155) |
| PAM-MnO2 ion-sieve | Simulated brine | Dose: 0.2 g; Temp: 20 °C | 2.64 mmol/g | Langmuir | - | (164) |
| HTO-PVA/PAAm | Simulated brine | Dose: 100 mg; temp: 25 °C; pH: 12 | 31.31 | Langmuir | PSO | (165) |
| λ-MnO2-PPy/PVA | Simulated brine | Dose: 100 mg; temp: 25 °C; pH: 5 | 27.93 | Langmuir | PSO | (166) |
| Fluorine-rich supramolecular hydrogel (FCH) | Simulated brine | Dose: 15 mg; temp: 25 °C; pH: 12 | 122.3 | Langmuir | Intraparticle diffusion | (167) |
| LIIP@N-CMS/GA imprinted film | Simulated brine | Temp: 25 °C; pH: 9.0 | 41.05 | Langmuir | PSO | (168) |
| Granulated Li/Al-LDHs | Salt Lake brine | Dose: 0.5 g; Temp: 25 °C | 14.5 | Langmuir | PSO | (169) |
| Fe/Ti-0.15(H) | Simulated brine | Dose: 1.5 g/L; temp: 25 °C; pH: 12.0; adsorption time: 12 h | 53.3 | Langmuir | PSO | (170) |
| HTO@BCA | LiOH | Dose: 0.5 g; adsorption time: 12 h | 39.8 | Langmuir | PSO | (171) |
| LDH/chitosan composite hydrogel | Simulated brine | Dose: 0.3 g/10 mL;; temp: 30 °C pH: 6.5 | 12.5 | Langmuir, Sips | PSO | (48) |
| Magnetic LDHs | Salt Lake brine | Dose: 1 g/30 mL; temp: 25 °C; adsorption time: 10 h | 6.0 | Langmuir | PSO | (47) |
| Granulated Li/Al-LDHs | Salt Lake brine | Dose: 4.0 g/150 mL; temp: 30 °C; adsorption time: 72 h | 4.82 | Sips | PSO | (172) |
2.4. Solvent Extraction
Figure 15
Figure 15. (a) Liquid–liquid equilibrium data for Li solution with TBP and cosolvents with FeCl3 as a coextracting agent adapted with permission from ref (175). Copyright 2011, American Chemical Society. (b) Relationship between the partition coefficient of Li and the extractability of iron at different Fe/Li ratios adapted with permission from ref (176). Copyright 2012, American Chemical Society. (c) Calculation of the theoretical stages of extraction using a McCabe–Thiele diagram adapted with permission from ref (178). Copyright 2016, Elsevier. (d) Simulation flow diagram for a three stage counter-current extraction process at an organic/aqueous ratio of 2/1 with initial Fe/Li = 1.3 for 10 min; adapted with permission from ref (178). Copyright 2016, Elsevier.
Figure 16
Figure 16. (a) Li extraction and stripping mechanism using the ternary solvent approach TBP-FeCl3-P507; (b) proposed structures as a result of the presence of P507 (2-ethylhexyl phosphonic acid mono-2-ethylhexyl ester); adapted with permission from ref (179). Copyright 2020, American Chemical Society. (c) tThe effect of dosage of IL in TBP as a function of extraction efficiency adapted with permission from ref (181). Copyright 2014, Elsevier. (d) Extraction efficiency as a function of ratio of molar ratio of ClO4– and Li-ions adapted with permission from ref (182). Copyright 2015, Elsevier. (e) Dosage of ILs as a function of extraction efficiency and distribution ratio from ref (183). Copyright 2016, Elsevier. (f) Extraction efficiency of synthesized ILs and [DEHP], as a function of HCl concentration in the aqueous phase adapted with permission from ref (184). Copyright 2017, Elsevier.
2.5. Membrane-Based Technologies
Figure 17
Figure 17. (a) Effect of pressure on rejection rate of ions; (b) effect of pH on rejection rate of ions; (c) effect of pH on separation factor and permeate amount of pure water adapted with permission from ref (190). Copyright 2015, Elsevier. (d) Flux and rejection for different salts by the positively charged composite hollow fiber as a function DAPP concentration adapted with permission from ref (191). Copyright 2015, Elsevier. Panels (e) and (f) permeate flux and rejection rate performance as a function of time for two simulated brine mixtures adapted with permission from ref (192). Copyright 2017, Elsevier.
Figure 18
Figure 18. (a–d) Performance parameters of membrane at different concentrations of ILs: (a) flux; (b) percent salt rejection; (c) ion rejection; and (d) selectivity (conditions: 0.6 MPa, pH = 6.4) adapted with permission from ref (193). Copyright 2020, Elsevier. (e) Effect of time on the (Mg/Li)P ratio of the permeate stream at varying feeds ratios of Mg/Li; adapted with permission from ref (196). Copyright 2017, Elsevier. (f, g) Effect of time on the (f) migration rate and (g) recovery of Li-ion at different ratios of K/L adapted with permission from ref (197). Copyright 2018, Elsevier.
2.6. Hybrid Methods
3. Challenges and Drawbacks of Li Extraction Techniques
Figure 19
Figure 19. Overview of conventional lithium recovery techniques, including solvent extraction, precipitation, electrochemical extraction, and adsorption-based methods, highlighting their key limitations.
4. Conclusions and Future Prospects
| technique | key parameters | advantages | challenges | research outlook/next steps |
|---|---|---|---|---|
| Adsorption/ion exchange | Li-ion selectivity (esp. over Mg-ions), adsorption capacity (mg/g), pH range, regeneration efficiency | High selectivity, reusable, applicable to low Li concentrations | Slow kinetics, fouling, limited regeneration cycles | Design of low-cost, highly selective, regenerable sorbents, fast kinetics |
| Solvent extraction | Solvent selectivity, phase ratio, distribution coefficient | High Li purity, continuous processing possible | Organic solvent loss, toxicity, emulsification | Development of greener extractants and stable systems |
| Precipitation | pH, precipitation agent type, Mg/Li interference | Simple operation, low-cost reagents | Poor selectivity, coprecipitation with Mg/Ca | Selective coprecipitants; integration with upstream Mg removal |
| Electrochemical separation | Electrode selectivity, current density, membrane type | Low energy consumption, precise ion control | Electrode fouling, membrane degradation | Durable electrode/membrane materials; selective transport layers |
| Membrane separation | Membrane selectivity (Li vs Mg), flux rate, fouling resistance | Energy-efficient, modular, scalable | Limited Li selectivity, fouling, high Mg-ion interference | Design of highly Li-ions selective membranes; antifouling coatings |
| Hybrid methods | Coupled processes (e.g., CDI or ED coupled with adsorption) | Synergistic performance, process intensification | Operational complexity, cost, integration challenges | Modular hybrid system design; pilot-scale validation |
References
This article references 212 other publications.
- 1Hensel, N. D. Economic challenges in the clean energy supply chain: The market for rare earth minerals and other critical inputs. Bus. Econ. 2011, 46 (3), 171– 184, DOI: 10.1057/be.2011.17Google ScholarThere is no corresponding record for this reference.
- 2Diouf, B.; Pode, R. Potential of lithium-ion batteries in renewable energy. Renew. Energy 2015, 76, 375– 380, DOI: 10.1016/j.renene.2014.11.058Google ScholarThere is no corresponding record for this reference.
- 3Dorn, F. M.; Peyré, F. R. Lithium as a strategic resource: Geopolitics, industrialization, and mining in Argentina. J. Lat. Am. Geogr. 2020, 19 (1), 68– 90, DOI: 10.1353/lag.2020.0101Google ScholarThere is no corresponding record for this reference.
- 4Role of Critical Minerals in Clean Energy Transitions; IEA: Paris, 2021. https://www.iea.org/reports/the-role-of-critical-minerals-in-clean-energy-transitions (accessed June 17, 2025).Google ScholarThere is no corresponding record for this reference.
- 5Kanagasundaram, T.; Murphy, O.; Haji, M. N.; Wilson, J. J. The recovery and separation of lithium using solvent extraction methods. Coord. Chem. Rev. 2024, 509, 215727, DOI: 10.1016/j.ccr.2024.215727Google ScholarThere is no corresponding record for this reference.
- 6Choubey, P. K.; Kim, M. S.; Srivastava, R. R.; Lee, J. C.; Lee, J. Y. Exploitation of the prominent energy-storage element lithium. Part I: From mineral and brine resources. Miner. Eng. 2016, 89, 119– 137, DOI: 10.1016/j.mineng.2016.01.010Google ScholarThere is no corresponding record for this reference.
- 7Swain, B. Recovery and recycling of lithium: A review. Sep. Purif. Technol. 2017, 172, 388– 403, DOI: 10.1016/j.seppur.2016.08.031Google ScholarThere is no corresponding record for this reference.
- 8Wang, J.; Yue, X.; Wang, P.; Yu, T.; Du, X.; Hao, X.; Abudula, A.; Guan, G. Electrochemical technologies for lithium recovery from liquid resources: A review. Renew. Sustain. Energy Rev. 2022, 154, 111813, DOI: 10.1016/j.rser.2021.111813Google ScholarThere is no corresponding record for this reference.
- 9Epstein, J. A.; Feist, E. M.; Zmora, J.; Marcus, Y. Extraction of lithium from the Dead Sea. Hydrometallurgy 1981, 6 (3), 269– 275, DOI: 10.1016/0304-386X(81)90044-XGoogle ScholarThere is no corresponding record for this reference.
- 10Martin, G.; Rentsch, L.; Höck, M.; Bertau, M. Lithium market research: Global supply, future demand, and price development. Energy Storage Mater. 2017, 6, 171– 179, DOI: 10.1016/j.ensm.2016.11.004Google ScholarThere is no corresponding record for this reference.
- 11Balaram, V.; Santosh, M.; Satyanarayanan, M.; Srinivas, N.; Gupta, H. Lithium: A review of applications, occurrence, exploration, extraction, recycling, analysis, and environmental impact. Geosci. Front. 2024, 15, 101868, DOI: 10.1016/j.gsf.2024.101868Google ScholarThere is no corresponding record for this reference.
- 12Sajna, M. S.; Elmakki, T.; Zavahir, S.; Tariq, H.; Abdulhameed, A.; Park, H.; Shon, H. K.; Han, D. S. Enhanced lithium separation from brines using nanofiltration (NF) technology: A review. Desalination 2024, 592, 118148, DOI: 10.1016/j.desal.2024.118148Google ScholarThere is no corresponding record for this reference.
- 13Gao, Y.; Sun, Y.; Chen, L.; Tan, Y.; Fan, R.; Hai, C.; Zhang, H. Progress of research in titanium-based lithium-ion sieves for the extraction of lithium from salt lake brine. ACS Omega 2025, 10, 11643, DOI: 10.1021/acsomega.4c08886Google ScholarThere is no corresponding record for this reference.
- 14Ramesh, M.; Tamil Selvan, M.; Rama Chandran, P.; Hariprasad, P.; Arunkumar, A.; Ram Kumar, C.; Prem Kumar, R. Experimental investigation on the effect of lithium and alkali-resistant glass fiber on aluminum AA5383 fabricated by the stir-casting process. Mater. Today Proc. 2023, DOI: 10.1016/j.matpr.2023.05.152Google ScholarThere is no corresponding record for this reference.
- 15Zhang, K.; Tang, H.; Shi, X.; Xue, Y.; Huang, Q. Effect of Ti3C2 MXene additive on the tribological properties of lithium grease at different temperatures. Wear 2023, 526–527, 204953, DOI: 10.1016/j.wear.2023.204953Google ScholarThere is no corresponding record for this reference.
- 16Gu, Y.; Zhong, K.; Cao, R.; Yang, Z. Aqueous lithium chloride solution as a non-toxic bactericidal and fungicidal disinfectant for air-conditioning systems: Efficacy and mechanism. Environ. Res. 2022, 212, 113112, DOI: 10.1016/j.envres.2022.113112Google ScholarThere is no corresponding record for this reference.
- 17Gu, J.; Liang, B.; Luo, X.; Zhang, X.; Yuan, W.; Xiao, B.; Tang, X. Recent advances and future prospects of lithium recovery from low-grade lithium resources: A review. Inorganics 2025, 13 (1), 4, DOI: 10.3390/inorganics13010004Google ScholarThere is no corresponding record for this reference.
- 18Rahighi, R.; Hosseini-Hosseinabad, S. M.; Zeraati, A. S.; Suwaileh, W.; Norouzi, A.; Panahi, M.; Gholipour, S.; Karaman, C.; Akhavan, O.; Khollari, M. A. R.; Vinu, A.; Rashidi, A.; Abdala, A.; Karimi-Maleh, H.; Orooji, Y. Two-dimensional materials in enhancement of membrane-based lithium recovery from metallic-ion-rich wastewaters: A review. Desalination 2022, 543, 116096, DOI: 10.1016/j.desal.2022.116096Google ScholarThere is no corresponding record for this reference.
- 19Park, J.; Lee, J.; Shim, I. T.; Kim, E.; Nam, S. H.; Koo, J. W.; Hwang, T. M. Electrochemical direct lithium extraction: A review of electrodialysis and capacitive deionization technologies. Resources 2025, 14, 27, DOI: 10.3390/resources14020027Google ScholarThere is no corresponding record for this reference.
- 20Marthi, R.; Smith, Y. R. Selective recovery of lithium from the Great Salt Lake using lithium manganese oxide-diatomaceous earth composite. Hydrometallurgy 2019, 186, 115– 125, DOI: 10.1016/j.hydromet.2019.03.011Google ScholarThere is no corresponding record for this reference.
- 21Rivard, E.; Trudeau, M.; Zaghib, K. Hydrogen storage for mobility: A review. Materials 2019, 12, 1973, DOI: 10.3390/ma12121973Google ScholarThere is no corresponding record for this reference.
- 22Zhang, L.; Zhang, T.; Lv, S.; Song, S.; Galván, H. J. O.; Quintana, M.; Zhao, Y. Adsorbents for lithium extraction from salt lake brine with high magnesium/lithium ratio: From structure-performance relationship to industrial applications. Desalination 2024, 579, 117480, DOI: 10.1016/j.desal.2024.117480Google ScholarThere is no corresponding record for this reference.
- 23Mojid, M. R.; Lee, K. J.; You, J. Advances in direct lithium extraction from continental brines: Ion-sieve adsorption and electrochemical methods for varied Mg/Li ratios. Sustainable Mater. Technol. 2024, 40, e00923 DOI: 10.1016/j.susmat.2024.e00923Google ScholarThere is no corresponding record for this reference.
- 24Battistel, A.; Palagonia, M. S.; Brogioli, D.; La Mantia, F.; Trócoli, R. Electrochemical methods for lithium recovery: A comprehensive and critical review. Adv. Mater. 2020, 32, 1905440, DOI: 10.1002/adma.201905440Google ScholarThere is no corresponding record for this reference.
- 25Tabelin, C. B.; Dallas, J.; Casanova, S.; Pelech, T.; Bournival, G.; Saydam, S.; Canbulat, I. Towards a low-carbon society: Lithium resource availability, challenges, and innovations in mining, extraction, and recycling. Miner. Eng. 2021, 163, 106743, DOI: 10.1016/j.mineng.2020.106743Google ScholarThere is no corresponding record for this reference.
- 26Wu, L.; Zhang, C.; Kim, S.; Hatton, T. A.; Mo, H.; Waite, T. D. Lithium recovery using electrochemical technologies: Advances and challenges. Water Res. 2022, 221, 118822, DOI: 10.1016/j.watres.2022.118822Google ScholarThere is no corresponding record for this reference.
- 27Vera, M. L.; Torres, W. R.; Galli, C. I.; Chagnes, A.; Flexer, V. Environmental impact of direct lithium extraction from brines. Nat. Rev. Earth Environ. 2023, 4, 149– 165, DOI: 10.1038/s43017-022-00387-5Google ScholarThere is no corresponding record for this reference.
- 28Ni, C.; Liu, C.; Wang, J.; Liang, Y.; Xie, W.; Zhong, H.; He, Z. Advances and promotion strategies of processes for extracting lithium from mineral resources. J. Ind. Eng. Chem. 2024, 140, 47– 64, DOI: 10.1016/j.jiec.2024.05.052Google ScholarThere is no corresponding record for this reference.
- 29Tansel, B. Significance of thermodynamic and physical characteristics on permeation of ions during membrane separation: Hydrated radius, hydration free energy, and viscous effects. Sep. Purif. Technol. 2012, 86, 119– 126, DOI: 10.1016/j.seppur.2011.10.033Google ScholarThere is no corresponding record for this reference.
- 30Orooji, Y.; Nezafat, Z.; Nasrollahzadeh, M.; Shafiei, N.; Afsari, M.; Pakzad, K.; Razmjou, A. Recent advances in nanomaterial development for lithium ion-sieving technologies. Desalination 2022, 529, 115624, DOI: 10.1016/j.desal.2022.115624Google ScholarThere is no corresponding record for this reference.
- 31Zhao, Z.; Si, X.; Liu, X.; He, L.; Liang, X. Lithium extraction from high Mg/Li ratio brine using LiFePO4/FePO4 electrode materials. Hydrometallurgy 2013, 133, 75– 83, DOI: 10.1016/j.hydromet.2012.11.013Google ScholarThere is no corresponding record for this reference.
- 32Zhou, J.; Li, X.; Xie, W.; Chen, Q.; Jiang, Y.; Wu, X.; Yang, L. Efficient solar evaporation and rapid lithium sorption by halloysite nanotube-based hydrogel. J. Clean. Prod. 2024, 437, 140523, DOI: 10.1016/j.jclepro.2023.140523Google ScholarThere is no corresponding record for this reference.
- 33Gulied, M.; Zavahir, S.; Elmakki, T.; Park, H.; Gago, G. H.; Shon, H. K.; Han, D. S. Efficient lithium recovery from simulated brine using a hybrid DCMD-ESIX system. Desalination 2024, 572, 117127, DOI: 10.1016/j.desal.2023.117127Google ScholarThere is no corresponding record for this reference.
- 34Wen, T.; Zhao, Y.; Wang, X.; Chen, L.; Gao, R.; Wang, S.; Zhang, T. Ultrafast separation of Li+ and Mg2+ by porous two-dimensional perm-selective montmorillonite membrane. Chem. Eng. J. 2023, 475, 146101, DOI: 10.1016/j.cej.2023.146101Google ScholarThere is no corresponding record for this reference.
- 35Paredes, C.; Rodríguez de San Miguel, E. Selective lithium extraction from diluted alkaline aqueous media using polymer inclusion membranes. Desalination 2020, 487, 114500, DOI: 10.1016/j.desal.2020.114500Google ScholarThere is no corresponding record for this reference.
- 36Zhu, Z.; Li, G. Polyaniline-covered lithium manganese oxide for high-performance capacitive deionization lithium extraction. ECS Meet. Abstr. 2025, MA2025-01, 1455 DOI: 10.1149/MA2025-01261455MTGABSGoogle ScholarThere is no corresponding record for this reference.
- 37Yoo, H.; Jang, H. J.; Kim, C.; Song, J.; Senthamaraikannan, T. G.; Lim, D. H.; Hong, H. J. Antimony-doped layered hydrogen titanium oxide as an advanced lithium-ion sieve. Desalination 2025, 614, 119175, DOI: 10.1016/j.desal.2025.119175Google ScholarThere is no corresponding record for this reference.
- 38Zia, S.; Kashif, M.; Ge, H.; Zhao, K.; Guo, Y.; Deng, T. Advanced PDMS@Li/ZnAl-LDH microspheres for lithium recovery from Qarhan Salt Lake brine. Desalination 2025, 615, 119286, DOI: 10.1016/j.desal.2025.119286Google ScholarThere is no corresponding record for this reference.
- 39Dai, Y.; Zhang, X.; Sun, J.; Wu, S.; Zhao, Z.; Xu, W.; Ren, G.; Li, Y.; Liu, D. Porous manganese-based ion sieve for sustainable lithium extraction from brine. Desalination 2025, 614, 119168, DOI: 10.1016/j.desal.2025.119168Google ScholarThere is no corresponding record for this reference.
- 40Deng, G.; Jiang, R.; Tang, F.; Hou, Z.; Feng, Y. Porous hydrophilic ion sieves for lithium recovery from alkaline brines: Batch and continuous operation. Desalination 2025, 614, 119159, DOI: 10.1016/j.desal.2025.119159Google ScholarThere is no corresponding record for this reference.
- 41Evans, B. R.; Jayanthi, K.; Popovs, I.; Moyer, B. A.; Paranthaman, M. P. New directions and principles for solvent extraction for recovery of lithium from aqueous brines and mineral leachates: A brief review. Sustainable Mater. Technol. 2025, 46, e01724 DOI: 10.1016/j.susmat.2025.e01724Google ScholarThere is no corresponding record for this reference.
- 42González, A.; Choque, G.; Grágeda, M.; Ushak, S. Membrane-assisted electrochemical removal of Mg2+ and Ca2+ from lithium brines: Effects of temperature and current density through a zeta potential approach. Membranes 2025, 15, 250, DOI: 10.3390/membranes15090250Google ScholarThere is no corresponding record for this reference.
- 43Wang, R.; Sheng, M.; Liu, X.; Wang, Q.; Wang, Z.; Wang, L. Electrosorption theoretically outperforms nanofiltration and electrodialysis for direct lithium extraction from brines. Environ. Sci. Technol. 2026, 60, 1577– 1595, DOI: 10.1021/acs.est.5c13571Google ScholarThere is no corresponding record for this reference.
- 44Li, C.; Wang, X.; Xie, Y.; Li, J.; Zhang, J. Extraction of lithium from salt-lake brine by functional ionic solid porous polymer adsorption material. J. Environ. Chem. Eng. 2025, 13, 117344, DOI: 10.1016/j.jece.2025.117344Google ScholarThere is no corresponding record for this reference.
- 45Yin, Z.; Zhao, W.; Sun, Y.; Wang, G.; Ma, T.; Sun, J.; Ji, P. Application of fly ash-based iron-doped lithium aluminum layered double hydroxide in brine-type lithium resources extraction: Adsorption mechanism analysis and life cycle assessment. Chem. Eng. J. 2025, 520, 165828, DOI: 10.1016/j.cej.2025.165828Google ScholarThere is no corresponding record for this reference.
- 46Abujayyab, M.; Hasan, S. W.; Arafat, H. A.; Banat, F. High-efficiency lithium recovery from desalination brine via MnAl layered double hydroxide. Desalination 2025, 616, 119376, DOI: 10.1016/j.desal.2025.119376Google ScholarThere is no corresponding record for this reference.
- 47Chen, J.; Lin, S.; Yu, J. High-selective cyclic adsorption and magnetic recovery performance of magnetic lithium-aluminum layered double hydroxides (MLDHs) in extracting Li+ from ultrahigh Mg/Li ratio brines. Sep. Purif. Technol. 2021, 255, 117710, DOI: 10.1016/j.seppur.2020.117710Google ScholarThere is no corresponding record for this reference.
- 48Zhao, L.; Zhang, X.; Liu, K.; Liu, X.; Gong, Y.; Peng, H.; Qi, W. Convenient synthesis of granulated Li/Al-layered double hydroxides/chitosan composite adsorbents for lithium extraction from simulated brine with a high Mg2+/Li+ ratio. Chem. Eng. J. 2024, 501, 157780, DOI: 10.1016/j.cej.2024.157780Google ScholarThere is no corresponding record for this reference.
- 49Zhang, J.; Ji, K.; Wang, Z.; Shi, W.; Zhao, Z.; Du, X.; Hao, X. Strongly cross-linked three-dimensional Li1-xMn2O4/C/SA-c-PEO@GF electrode for lithium recovery from high Mg2+/Li+ ratio salt lake brine. Sep. Purif. Technol. 2025, 376, 134102, DOI: 10.1016/j.seppur.2025.134102Google ScholarThere is no corresponding record for this reference.
- 50Nadzri, N.; Lim, Y. J.; Liao, X.; Liang, Y.; Goh, K.; Liao, Y.; Goto, A.; Wang, R. Lithium recovery from seawater desalination brines using ion-sieve electrospun nanofibrous membranes: The role of nanofiber design. Chem. Eng. J. 2025, 513, 162886, DOI: 10.1016/j.cej.2025.162886Google ScholarThere is no corresponding record for this reference.
- 51Yin, X.; Xu, P.; Wang, H. Applications of crown ether-based materials for enhancing lithium recovery from brines. Sep. Purif. Technol. 2025, 371, 133093, DOI: 10.1016/j.seppur.2025.133093Google ScholarThere is no corresponding record for this reference.
- 52Dach, E.; Marston, J.; Abu-Obaid, S.; Peng, A.; Yip, N. Y. A novel approach for direct lithium extraction from alkali metal cations in brine mixtures using thermally switchable solvents. Joule 2026, 10, 102265, DOI: 10.1016/j.joule.2025.102265Google ScholarThere is no corresponding record for this reference.
- 53Su, H.; Li, Z.; Zhang, J.; Zhu, Z.; Wang, L.; Qi, T. Recovery of lithium from salt lake brine using a mixed ternary solvent extraction system consisting of TBP, FeCl3, and P507. Hydrometallurgy 2020, 197, 105487, DOI: 10.1016/j.hydromet.2020.105487Google ScholarThere is no corresponding record for this reference.
- 54Zhang, L.; Li, J.; Liu, R.; Zhou, Y.; Zhang, Y.; Ji, L.; Li, L. Recovery of lithium from salt lake brine with high Na/Li ratio using solvent extraction. J. Mol. Liq. 2022, 362, 119667, DOI: 10.1016/j.molliq.2022.119667Google ScholarThere is no corresponding record for this reference.
- 55Wang, L.; Zhang, J.; Meng, S.; Wu, H.; Zhao, Y.; Meng, Z.; Feng, L.; Wang, H.; Cheng, Y.; Yang, L.; Liu, H. Recovery of lithium from high Mg2+/Li+ ratio brine using a novel solvent extraction system TOP-FeCl3-MIBK. Desalination 2025, 599, 118445, DOI: 10.1016/j.desal.2024.118445Google ScholarThere is no corresponding record for this reference.
- 56Bao, Y.; Ji, Z. Y.; Zhou, H.; Zhang, C.; Song, S.; Jia, F.; Li, J.; Quintana, M. Li-ion pre-intercalation strategy of manganese oxides for capacitive deionization-based selective lithium extraction from low-grade brine. Small 2024, 2406951, DOI: 10.1002/smll.202406951Google ScholarThere is no corresponding record for this reference.
- 57Dhanushkotti, R.; Mohammed, A. K.; Ranjeesh, K. C.; Krishnegowda, H. M.; Aldaqqa, N. M.; Shetty, D. Inherited nitrogen distribution control in covalent organic framework cathodes for efficient electrochemical lithium recovery via capacitive deionization. Adv. Sci. 2025, 12, 2417140, DOI: 10.1002/advs.202417140Google ScholarThere is no corresponding record for this reference.
- 58Liao, G.; Yu, L.; Xia, Y.; Wang, Z.; Lu, Z.; Mei, J.; Liu, H.; Liu, C. High selectivity, capacity, and stability for electrochemical lithium extraction on boron-doped H1.6Mn1.6O4 by tailoring lattice constant and intercalation energy. Water Res. 2025, 274, 123131, DOI: 10.1016/j.watres.2025.123131Google ScholarThere is no corresponding record for this reference.
- 59Xu, Y.; Wu, W.; Yi, Q.; Zhang, H.; Lu, Q.; Zhang, K.; Pang, T.; Song, J.; Zhang, D. LiMn2O4 nanoparticles in situ embedded in carbon networks for lithium extraction from brine via hybrid capacitive deionization. ACS Appl. Mater. Interfaces 2025, 17, 4821– 4831, DOI: 10.1021/acsami.4c16382Google ScholarThere is no corresponding record for this reference.
- 60Li, P.; Chong, X.; Guo, Y.; Wu, G.; Wang, S.; Liu, R.; Ma, J.; Wang, Y.; Chen, W.; Cai, Y.; Wang, Y.; Yuan, Q.; Zhao, J. Heterostructure-engineered conductive MOFs coupled with MoSe2@MXene framework for efficient and selective lithium extraction from salt lake brine via capacitive deionization. Adv. Sci. 2026, 13, e21830 DOI: 10.1002/advs.202521830Google ScholarThere is no corresponding record for this reference.
- 61Zhang, L.; Huang, Z.; Chen, Y.; Li, F.; Li, G.; Zhang, F. Highly selective lithium-ion separation by regulating ion transport energy barriers of vermiculite membranes. ACS Nano 2026, 20, 1345– 1356, DOI: 10.1021/acsnano.5c17718Google ScholarThere is no corresponding record for this reference.
- 62Li, K.; Wei, X.; Wang, W.; Zhang, Q.; Ding, Y.; Yang, Q. Optimization of ion migration during lithium-ion recovery through electrodialysis by numerical simulations. Asia-Pac. J. Chem. Eng. 2026, 21, e70125 DOI: 10.1002/apj.70125Google ScholarThere is no corresponding record for this reference.
- 63Oladipo, A. A.; Ahmad, M. Energy-efficient ion recovery from water using electro-driven membranes: A comprehensive critical review. Water 2025, 17, 2456, DOI: 10.3390/w17162456Google ScholarThere is no corresponding record for this reference.
- 64Zhao, Z.; Yang, J.; Kong, D.; Peng, Y.; Jin, D. Effects of operational parameters on Mg2+/Li+ separation performance in electrodialysis system. Membranes 2025, 15, 260, DOI: 10.3390/membranes15090260Google ScholarThere is no corresponding record for this reference.
- 65González, A.; Choque, G.; Grágeda, M.; Ushak, S. Membrane-assisted electrochemical removal of Mg2+ and Ca2+ from lithium brines: Effects of temperature and current density through a zeta potential approach. Membranes 2025, 15, 250, DOI: 10.3390/membranes15090250Google ScholarThere is no corresponding record for this reference.
- 66Aljarrah, S.; Alsabbagh, A.; Almahasneh, M. Selective recovery of lithium from Dead Sea end brines using UBK10 ion exchange resin. Can. J. Chem. Eng. 2023, 101, 1185– 1194, DOI: 10.1002/cjce.24559Google ScholarThere is no corresponding record for this reference.
- 67Khalil, A.; Mohammed, S.; Hashaikeh, R.; Hilal, N. Lithium recovery from brine: Recent developments and challenges. Desalination 2022, 528, 115611, DOI: 10.1016/j.desal.2022.115611Google ScholarThere is no corresponding record for this reference.
- 68An, J. W.; Kang, D. J.; Tran, K. T.; Kim, M. J.; Lim, T.; Tran, T. Recovery of lithium from Uyuni salar brine. Hydrometallurgy 2012, 117–118, 64– 70, DOI: 10.1016/j.hydromet.2012.02.008Google ScholarThere is no corresponding record for this reference.
- 69Dave, R. H.; Ghosh, P. K. Enrichment of bromine in sea-bittern with recovery of other marine chemicals. Ind. Eng. Chem. Res. 2005, 44, 2903– 2907, DOI: 10.1021/ie049130xGoogle ScholarThere is no corresponding record for this reference.
- 70Bertau, M.; Martin, G.; Bertau, M. Integrated direct carbonation process for lithium recovery from primary and secondary resources. Mater. Sci. Forum 2019, 959, 69– 73, DOI: 10.4028/www.scientific.net/MSF.959.69Google ScholarThere is no corresponding record for this reference.
- 71Xu, Z.; Zhang, H.; Wang, R.; Gui, W.; Liu, G.; Yang, Y. Systemic and direct production of battery-grade lithium carbonate from a saline lake. Ind. Eng. Chem. Res. 2014, 53, 16502– 16507, DOI: 10.1021/ie502749nGoogle ScholarThere is no corresponding record for this reference.
- 72Shi, L.; Peng, Z.; Ning, P.; Sun, X.; Li, K.; Zhang, H.; Qu, T. Clean and efficient recovery of lithium from Al-Li alloys via vacuum fractional condensation. Separations 2023, 10, 374, DOI: 10.3390/separations10070374Google ScholarThere is no corresponding record for this reference.
- 73Li, Y. H.; Zhao, Z. W.; Liu, X. H.; Chen, X. Y.; Zhong, M. L. Extraction of lithium from salt lake brine by aluminum-based alloys. Trans. Nonferrous Met. Soc. China 2015, 25, 3484– 3489, DOI: 10.1016/S1003-6326(15)64032-8Google ScholarThere is no corresponding record for this reference.
- 74Zhang, L.; Zhang, T.; Zhao, Y.; Dong, G.; Lv, S.; Ma, S.; Song, S.; Quintana, M. Doping engineering of lithium-aluminum layered double hydroxides for high-efficiency lithium extraction from salt lake brines. Nano Res. 2024, 17, 1646– 1654, DOI: 10.1007/s12274-023-5950-1Google ScholarThere is no corresponding record for this reference.
- 75Yong, M.; Tang, M.; Sun, L.; Xiong, F.; Xie, L.; Zeng, G.; Ren, X.; Wang, K.; Cheng, Y.; Li, Z.; Li, E.; Zhang, X.; Wang, H. Sustainable lithium extraction and magnesium hydroxide co-production from salt-lake brines. Nat. Sustain. 2024, 7, 1662– 1671, DOI: 10.1038/s41893-024-01435-2Google ScholarThere is no corresponding record for this reference.
- 76Jiang, H.; Zhang, S.; Yang, Y.; Yu, J. Synergic and competitive adsorption of Li-Na-MgCl2 onto lithium-aluminum hydroxides. Adsorption 2020, 26, 1039– 1049, DOI: 10.1007/s10450-020-00208-5Google ScholarThere is no corresponding record for this reference.
- 77Hamzaoui, A. H.; M’nif, A.; Hammi, H.; Rokbani, R. Contribution to the lithium recovery from brine. Desalination 2003, 158, 221– 224, DOI: 10.1016/S0011-9164(03)00455-7Google ScholarThere is no corresponding record for this reference.
- 78Tran, K. T.; Van Luong, T.; An, J. W.; Kang, D. J.; Kim, M. J.; Tran, T. Recovery of magnesium from Uyuni salar brine as high-purity magnesium oxalate. Hydrometallurgy 2013, 138, 93– 99, DOI: 10.1016/j.hydromet.2013.05.013Google ScholarThere is no corresponding record for this reference.
- 79Wang, H.; Zhong, Y.; Du, B.; Zhao, Y.; Wang, M. Recovery of both magnesium and lithium from high Mg/Li ratio brines using a novel process. Hydrometallurgy 2018, 175, 102– 108, DOI: 10.1016/j.hydromet.2017.10.017Google ScholarThere is no corresponding record for this reference.
- 80Lai, X.; Xiong, P.; Zhong, H. Extraction of lithium from brines with high Mg/Li ratio by the crystallization-precipitation method. Hydrometallurgy 2020, 192, 105252, DOI: 10.1016/j.hydromet.2020.105252Google ScholarThere is no corresponding record for this reference.
- 81Zhang, Y.; Hu, Y.; Sun, N.; Khoso, S. A.; Wang, L.; Sun, W. A novel precipitant for separating lithium from magnesium in high Mg/Li ratio brine. Hydrometallurgy 2019, 187, 125– 133, DOI: 10.1016/j.hydromet.2019.05.019Google ScholarThere is no corresponding record for this reference.
- 82Xiang, W.; Liang, S.; Zhou, Z.; Qin, W.; Fei, W. Extraction of lithium from salt lake brine containing borate anion and high concentration of magnesium. Hydrometallurgy 2016, 166, 9– 15, DOI: 10.1016/j.hydromet.2016.08.005Google ScholarThere is no corresponding record for this reference.
- 83Liu, D.; Li, Z.; He, L.; Zhao, Z. Facet-engineered Li3PO4 for lithium recovery from brines. Desalination 2021, 514, 115186, DOI: 10.1016/j.desal.2021.115186Google ScholarThere is no corresponding record for this reference.
- 84Zhang, H.; Zhang, S. S.; Zhang, W.; Ma, W. C.; Li, Y. P.; Chen, L.; Zhu, L.; Pan, Y. Recovering phosphorus and lithium separately from wastewater and brine using a novel coupled biofilm-precipitation system. J. Water Process Eng. 2023, 55, 104097, DOI: 10.1016/j.jwpe.2023.104097Google ScholarThere is no corresponding record for this reference.
- 85Song, Y.; Zhao, T.; He, L.; Zhao, Z.; Liu, X. A promising approach for directly extracting lithium from α-spodumene by alkaline digestion and precipitation as phosphate. Hydrometallurgy 2019, 189, 105141, DOI: 10.1016/j.hydromet.2019.105141Google ScholarThere is no corresponding record for this reference.
- 86Quintero, C.; Dahlkamp, J. M.; Fierro, F.; Thennis, T.; Zhang, Y.; Videla, Á.; Rojas, R. Development of a co-precipitation process for the preparation of magnesium hydroxide containing lithium carbonate from Li-enriched brines. Hydrometallurgy 2020, 198, 105515, DOI: 10.1016/j.hydromet.2020.105515Google ScholarThere is no corresponding record for this reference.
- 87Alsabbagh, A.; Aljarrah, S.; Almahasneh, M. Lithium enrichment optimization from Dead Sea end brine by chemical precipitation technique. Miner. Eng. 2021, 170, 107038, DOI: 10.1016/j.mineng.2021.107038Google ScholarThere is no corresponding record for this reference.
- 88Cipollina, A.; Winter, D.; Battaglia, G.; Berkemeyer, L.; Cortina, J. L.; de Labastida, M. F.; Rodriguez, J. L. Recovery of lithium carbonate from dilute Li-rich brine via homogeneous and heterogeneous precipitation. Ind. Eng. Chem. Res. 2022, 61, 13589– 13602, DOI: 10.1021/acs.iecr.2c01397Google ScholarThere is no corresponding record for this reference.
- 89Kim, H. S.; Suh, Y. J.; Kim, D. W.; Yoo, H.; Hong, H. J.; Park, I. S. Direct aluminate precipitation technique with a phase transformation strategy for selective lithium extraction from Uyuni brine. Chem. Eng. J. 2025, 505, 159220, DOI: 10.1016/j.cej.2025.159220Google ScholarThere is no corresponding record for this reference.
- 90Ma, C.; Zhang, J.; Liu, Y.; Sun, L.; Liu, Q. Integrated separation of precipitation and three-liquid-phase extraction for boron, lithium, and magnesium from salt lake brine with high magnesium-lithium ratio. Sep. Purif. Technol. 2024, 347, 127541, DOI: 10.1016/j.seppur.2024.127541Google ScholarThere is no corresponding record for this reference.
- 91Zhao, X.; Li, G.; Feng, M.; Wang, Y. Semi-continuous electrochemical extraction of lithium from brine using CF-NMMO/AC asymmetric hybrid capacitors. Electrochim. Acta 2020, 331, 135285, DOI: 10.1016/j.electacta.2019.135285Google ScholarThere is no corresponding record for this reference.
- 92Song, Y.; Zhao, Z. Recovery of lithium from spent lithium-ion batteries using precipitation and electrodialysis techniques. Sep. Purif. Technol. 2018, 206, 335– 342, DOI: 10.1016/j.seppur.2018.06.022Google ScholarThere is no corresponding record for this reference.
- 93Guo, Z. Y.; Ji, Z. Y.; Chen, Q. B.; Liu, J.; Zhao, Y. Y.; Li, F.; Liu, Z. Y.; Yuan, J. S. Prefractionation of LiCl from concentrated seawater/salt lake brines by electrodialysis with monovalent selective ion-exchange membranes. J. Clean. Prod. 2018, 193, 338– 350, DOI: 10.1016/j.jclepro.2018.05.077Google ScholarThere is no corresponding record for this reference.
- 94Zhao, X.; Yang, H.; Wang, Y.; Sha, Z. Review on the electrochemical extraction of lithium from seawater/brine. J. Electroanal. Chem. 2019, 850, 113389, DOI: 10.1016/j.jelechem.2019.113389Google ScholarThere is no corresponding record for this reference.
- 95Yin, R.; Zhao, Z.; Xu, W.; He, L. LiMn2O4 submicronization: Shortened Li+ diffusion pathways for enhanced electrochemical lithium extraction and cycle performance. Sep. Purif. Technol. 2025, 359, 130394, DOI: 10.1016/j.seppur.2024.130394Google ScholarThere is no corresponding record for this reference.
- 96Xu, W.; He, L.; Zhao, Z. Lithium extraction from high Mg/Li brine via electrochemical intercalation/de-intercalation using LiMn2O4 materials. Desalination 2021, 503, 114935, DOI: 10.1016/j.desal.2021.114935Google ScholarThere is no corresponding record for this reference.
- 97Zhu, W.; Xu, W.; Liu, D.; He, L.; Liu, X.; Zhao, Z. Ionic transport kinetics of selective electrochemical lithium extraction from brines. Electrochim. Acta 2024, 475, 143519, DOI: 10.1016/j.electacta.2023.143519Google ScholarThere is no corresponding record for this reference.
- 98Xiong, J.; He, L.; Liu, D.; Xu, W.; Zhao, Z. Olivine-FePO4 preparation for lithium extraction from brines via electrochemical de-intercalation/intercalation. Desalination 2021, 520, 115326, DOI: 10.1016/j.desal.2021.115326Google ScholarThere is no corresponding record for this reference.
- 99Parsa, N.; Moheb, A.; Mehrabani-Zeinabad, M. A.; Masigol, M. A. Recovery of lithium ions from sodium-contaminated lithium bromide solution using electrodialysis. Chem. Eng. Res. Des. 2015, 98, 81– 88, DOI: 10.1016/j.cherd.2015.03.025Google ScholarThere is no corresponding record for this reference.
- 100Strathmann, H. Electrodialysis, a mature technology with a multitude of new applications. Desalination 2010, 264, 268– 288, DOI: 10.1016/j.desal.2010.04.069Google ScholarThere is no corresponding record for this reference.
- 101Ji, Z. Y.; Chen, Q. B.; Yuan, J. S.; Liu, J.; Zhao, Y. Y.; Feng, W. X. Preliminary study on recovering lithium from high Mg2+/Li+ ratio brines by electrodialysis. Sep. Purif. Technol. 2017, 172, 168– 177, DOI: 10.1016/j.seppur.2016.08.006Google ScholarThere is no corresponding record for this reference.
- 102Jiang, D.; Xu, R.; Bai, L.; Wu, W.; Luo, D.; Li, Z.; Asahi, T.; Mai, Y.; Liu, Z.; Yamauchi, Y.; Xu, X. Insights into electrochemical paradigms for lithium extraction: Electrodialysis versus capacitive deionization. Coord. Chem. Rev. 2024, 516, 215923, DOI: 10.1016/j.ccr.2024.215923Google ScholarThere is no corresponding record for this reference.
- 103Ying, J.; Luo, M.; Jin, Y.; Yu, J. Selective separation of lithium from high Mg/Li ratio brine using single-stage and multi-stage selective electrodialysis. Desalination 2020, 492, 114621, DOI: 10.1016/j.desal.2020.114621Google ScholarThere is no corresponding record for this reference.
- 104Morita, K.; Matsumoto, T.; Hoshino, T. Efficient lithium extraction via electrodialysis using acid-processed lithium-adsorbing lithium lanthanum titanate. Desalination 2022, 543, 116117, DOI: 10.1016/j.desal.2022.116117Google ScholarThere is no corresponding record for this reference.
- 105Wang, W.; Zhang, Y.; Li, F.; Chen, Y.; Mojallali Rostami, S. M.; Hosseini, S. S.; Shao, L. Designing an energy-efficient multi-stage selective electrodialysis process based on high-performance materials for lithium extraction. J. Membr. Sci. 2023, 675, 121534, DOI: 10.1016/j.memsci.2023.121534Google ScholarThere is no corresponding record for this reference.
- 106Zhang, X. C.; Wang, J.; Ji, Z. Y.; Ji, P. Y.; Liu, J.; Zhao, Y. Y.; Li, F.; Yuan, J. S. Preparation of Li2CO3 from high Mg2+/Li+ brines based on selective electrodialysis with feed-and-bleed mode. J. Environ. Chem. Eng. 2021, 9, 106635, DOI: 10.1016/j.jece.2021.106635Google ScholarThere is no corresponding record for this reference.
- 107Han, J.; Wang, K.; Chen, S.; Lu, H.; Zhang, Z.; Li, Z.; Zhou, L.; Du, Y.; Meng, Q.; Lewis, A.; Huang, Y. Lithium aluminum titanium phosphate-based ceramic composite membrane for selective lithium-ion separation via scalable electrodialysis. Desalination 2024, 591, 118010, DOI: 10.1016/j.desal.2024.118010Google ScholarThere is no corresponding record for this reference.
- 108Ma, W.; Han, G.; Li, J.; An, X.; Gao, F.; Du, X.; Li, J.; Liu, Z.; Guan, G.; Hao, X. Hierarchical electroactive ion-permselective membrane with electrochemical switched ion-pump effect for continuous lithium recovery. J. Membr. Sci. 2024, 700, 122719, DOI: 10.1016/j.memsci.2024.122719Google ScholarThere is no corresponding record for this reference.
- 109Zhang, H.; Huang, Z.; Zhao, L.; Guo, Z.; Wang, J.; Liu, J.; Zhao, Y.; Li, F.; Zhang, P.; Ji, Z. Y. Fast and stable lithium extraction enabled by less-defective graphene-supported LiMn2O4 networks in hybrid capacitive deionization. Chem. Eng. J. 2024, 482, 148802, DOI: 10.1016/j.cej.2024.148802Google ScholarThere is no corresponding record for this reference.
- 110Tian, G.; Gao, J.; Yan, S.; Wang, H.; Li, R.; Xiang, J.; Du, W.; Zhang, L.; Tang, N. Long-acting electrochemical lithium recovery from original brine using super-stable spinel lithium manganate oxide. Desalination 2024, 592, 118103, DOI: 10.1016/j.desal.2024.118103Google ScholarThere is no corresponding record for this reference.
- 111Meng, X.; Jing, Y.; Li, J.; Sun, Z.; Wu, Z. Electrochemical recovery of lithium from brine by highly stable truncated octahedral LiNi0.05Mn1.95O4. Chem. Eng. Sci. 2024, 283, 119400, DOI: 10.1016/j.ces.2023.119400Google ScholarThere is no corresponding record for this reference.
- 112Gu, J.; Zhou, G.; Chen, L.; Li, X.; Luo, G.; Fan, L.; Chao, Y.; Ji, H.; Zhu, W. Particle size control and electrochemical lithium extraction performance of LiMn2O4. J. Electroanal. Chem. 2023, 940, 117487, DOI: 10.1016/j.jelechem.2023.117487Google ScholarThere is no corresponding record for this reference.
- 113Tian, G.; Gao, J.; Wang, M.; Wen, X.; Liu, Y.; Xiang, J.; Zhang, L.; Cheng, P.; Zhang, J.; Tang, N. Structural stabilization of Cr-doped spinel LiMn2O4 for long-term cyclability in electrochemical lithium recovery. Electrochim. Acta 2024, 475, 143361, DOI: 10.1016/j.electacta.2023.143361Google ScholarThere is no corresponding record for this reference.
- 114Liu, C.; Li, Y.; Lin, D.; Hsu, P. C.; Liu, B.; Yan, G.; Wu, T.; Cui, Y.; Chu, S. Lithium extraction from seawater through pulsed electrochemical intercalation. Joule 2020, 4, 1459– 1469, DOI: 10.1016/j.joule.2020.05.017Google ScholarThere is no corresponding record for this reference.
- 115Zhang, J.; Su, W.; Yi, B.; Guo, Y.; Deng, T.; Yu, X. A new strategy for preparing highly stable and high-capacity electrodes for green electrochemical lithium extraction. Chem. Eng. J. 2023, 454, 140416, DOI: 10.1016/j.cej.2022.140416Google ScholarThere is no corresponding record for this reference.
- 116Fu, H.; Zhu, W.; Xu, W.; Zhao, Z. Understanding ion intercalation of LiMn2O4 in brine for electrochemical detection of Li+. J. Electroanal. Chem. 2025, 983, 119033, DOI: 10.1016/j.jelechem.2025.119033Google ScholarThere is no corresponding record for this reference.
- 117Hu, B.; Wang, Y.; Zhang, B.; Song, X.; Jiang, H.; Ma, J.; Liu, J. GO-encapsulated La-doped lithium manganese oxide assemblies for enhanced lithium extraction in capacitive deionization. Sep. Purif. Technol. 2024, 348, 127693, DOI: 10.1016/j.seppur.2024.127693Google ScholarThere is no corresponding record for this reference.
- 118Siekierka, A.; Bryjak, M. Novel anion exchange membrane for lithium salt concentration in hybrid capacitive deionization. Desalination 2019, 452, 279– 289, DOI: 10.1016/j.desal.2018.10.009Google ScholarThere is no corresponding record for this reference.
- 119Rethinasabapathy, M.; Bhaskaran, G.; Hwang, S. K.; Ryu, T.; Huh, Y. S. Efficient lithium extraction using redox-active Prussian blue nanoparticle-anchored activated carbon electrodes via membrane capacitive deionization. Chemosphere 2023, 336, 139256, DOI: 10.1016/j.chemosphere.2023.139256Google ScholarThere is no corresponding record for this reference.
- 120Faheem, M.; Alam, R.; Alhajaj, A.; Zou, L. Recovery of lithium by pseudocapacitive electrodes in capacitive deionization. Electrochim. Acta 2024, 489, 144267, DOI: 10.1016/j.electacta.2024.144267Google ScholarThere is no corresponding record for this reference.
- 121Siekierka, A. Lithium iron manganese oxide as an adsorbent for capturing lithium ions in hybrid capacitive deionization under different electrical modes. Sep. Purif. Technol. 2020, 236, 116234, DOI: 10.1016/j.seppur.2019.116234Google ScholarThere is no corresponding record for this reference.
- 122Ying, J.; Luo, M.; Jin, Y.; Yu, J. Selective separation of lithium from high Mg/Li ratio brine using single-stage and multi-stage selective electrodialysis processes. Desalination 2020, 492, 114621, DOI: 10.1016/j.desal.2020.114621Google ScholarThere is no corresponding record for this reference.
- 123Zhao, Y.; Wu, M.; Shen, P.; Uytterhoeven, C.; Mamrol, N.; Shen, J.; Gao, C.; Van der Bruggen, B. Composite anti-scaling membrane with interpenetrating nanofiber networks for selective lithium separation. J. Membr. Sci. 2021, 618, 118668, DOI: 10.1016/j.memsci.2020.118668Google ScholarThere is no corresponding record for this reference.
- 124Liu, G.; Zhao, Z.; He, L. Highly selective lithium recovery from high Mg/Li ratio brines. Desalination 2020, 474, 114185, DOI: 10.1016/j.desal.2019.114185Google ScholarThere is no corresponding record for this reference.
- 125Sharma, P. P.; Yadav, V.; Rajput, A.; Gupta, H.; Saravaia, H.; Kulshrestha, V. Sulfonated poly(ether ether ketone) composite cation-exchange membrane for selective lithium recovery by electrodialysis. Desalination 2020, 496, 114755, DOI: 10.1016/j.desal.2020.114755Google ScholarThere is no corresponding record for this reference.
- 126Wang, W.; Zhang, Y.; Li, F.; Chen, Y.; Mojallali Rostami, S. M.; Hosseini, S. S.; Shao, L. Mussel-inspired polyphenol/polyethyleneimine-assembled membranes with highly positive surface charge for unprecedented cation permselectivity. J. Membr. Sci. 2022, 658, 120703, DOI: 10.1016/j.memsci.2022.120703Google ScholarThere is no corresponding record for this reference.
- 127Flexer, V.; Baspineiro, C. F.; Galli, C. I. Lithium recovery from brines: A vital raw material for green energies with potential environmental impacts. Sci. Total Environ. 2018, 639, 1188– 1204, DOI: 10.1016/j.scitotenv.2018.05.223Google ScholarThere is no corresponding record for this reference.
- 128Seip, A.; Safari, S.; Pickup, D. M.; Chadwick, A. V.; Ramos, S.; Velasco, C. A.; Cerrato, J. M.; Alessi, D. S. Lithium recovery from hydraulic fracturing flowback and produced water using selective ion-exchange sorbents. Chem. Eng. J. 2021, 426, 130713, DOI: 10.1016/j.cej.2021.130713Google ScholarThere is no corresponding record for this reference.
- 129Meng, F.; McNeice, J.; Zadeh, S. S.; Ghahreman, A. Review of lithium production and recovery from minerals, brines, and lithium-ion batteries. Miner. Process. Extr. Metall. Rev. 2021, 42, 123– 141, DOI: 10.1080/08827508.2019.1668387Google ScholarThere is no corresponding record for this reference.
- 130Sheng, B.; Su, H.; Yu, J.; Lin, S. Lithium extraction from low-grade Na+/K+ brines using high layer-charge layered double hydroxides. Desalination 2023, 565, 116856, DOI: 10.1016/j.desal.2023.116856Google ScholarThere is no corresponding record for this reference.
- 131Gao, A.; Sun, Z.; Li, S.; Hou, X.; Li, H.; Wang, C.; Wu, W. Self-assembled layered lithium manganese oxide with ultrahigh adsorption capacity and selectivity for lithium. Chem. Eng. J. 2023, 471, 144287, DOI: 10.1016/j.cej.2023.144287Google ScholarThere is no corresponding record for this reference.
- 132Ighalo, J. O.; Amaku, J. F.; Olisah, C.; Adeola, A. O.; Iwuozor, K. G.; Akpomie, K. G.; Conradie, J.; Adegoke, K. A.; Oyedotun, K. O. Utilization of adsorption as a resource recovery technique for lithium in geothermal water. J. Mol. Liq. 2022, 365, 120107, DOI: 10.1016/j.molliq.2022.120107Google ScholarThere is no corresponding record for this reference.
- 133Safari, S.; Lottermoser, B. G.; Alessi, D. S. Metal oxide sorbents for sustainable lithium recovery from unconventional resources. Appl. Mater. Today 2020, 19, 100638, DOI: 10.1016/j.apmt.2020.100638Google ScholarThere is no corresponding record for this reference.
- 134Arroyo, F.; Morillo, J.; Usero, J.; Rosado, D.; El Bakouri, H. Lithium recovery from desalination brines using specific ion-exchange resins. Desalination 2019, 468, 114073, DOI: 10.1016/j.desal.2019.114073Google ScholarThere is no corresponding record for this reference.
- 135Huang, Y.; Li, X.; Chen, L.; Luo, G.; Tao, D.; Sun, J.; Qiu, Z.; Chao, Y.; Zhu, W. Synthesis of magnetic crown-ether ion-imprinted polymer for selective lithium adsorption. New J. Chem. 2023, 47, 3134– 3139, DOI: 10.1039/D2NJ05377BGoogle ScholarThere is no corresponding record for this reference.
- 136Baudino, L.; Santos, C.; Pirri, C. F.; La Mantia, F.; Lamberti, A. Recent advances in lithium recovery from water resources: From passive to electrochemical methods. Adv. Sci. 2022, 9, 2201380, DOI: 10.1002/advs.202201380Google ScholarThere is no corresponding record for this reference.
- 137Weng, D.; Duan, H.; Hou, Y.; Huo, J.; Chen, L.; Zhang, F.; Wang, J. Introduction of manganese-based lithium-ion sieves: A review. Prog. Nat. Sci.: Mater. Int. 2020, 30, 139– 152, DOI: 10.1016/j.pnsc.2020.01.017Google ScholarThere is no corresponding record for this reference.
- 138Yu, H.; Naidu, G.; Zhang, C.; Wang, C.; Razmjou, A.; Han, D. S.; Shon, H. Metal-based adsorbents for lithium recovery from aqueous resources. Desalination 2022, 539, 115951, DOI: 10.1016/j.desal.2022.115951Google ScholarThere is no corresponding record for this reference.
- 139Zhang, Y.; Liu, J.; Yang, Y.; Lin, S.; Li, P. Preparation of granular titanium-type lithium-ion sieves and recyclability assessment for lithium recovery from brines with varying pH. Sep. Purif. Technol. 2021, 267, 118613, DOI: 10.1016/j.seppur.2021.118613Google ScholarThere is no corresponding record for this reference.
- 140Borghols, W. J. H.; Wagemaker, M.; Lafont, U.; Kelder, E. M.; Mulder, F. M. Size effects in the Li4+xTi5O12 spinel. J. Am. Chem. Soc. 2009, 131, 17786– 17792, DOI: 10.1021/ja902423eGoogle ScholarThere is no corresponding record for this reference.
- 141Zhao, B.; Qiao, Y.; Qian, Z.; Wei, W.; Li, J.; Wu, Z.; Liu, Z. Unraveling Li+ desorption behavior and mechanism of Li4Ti5O12 with different facets to enhance lithium extraction. J. Mater. Chem. A 2023, 11, 7094– 7104, DOI: 10.1039/D2TA10016AGoogle ScholarThere is no corresponding record for this reference.
- 142Du, Y. E.; Niu, X.; Li, W.; An, J.; Liu, Y.; Chen, Y.; Wang, P.; Yang, X.; Feng, Q. Microwave-assisted synthesis of high-energy faceted TiO2 nanocrystals derived from exfoliated porous metatitanic acid nanosheets. Materials 2019, 12, 3614, DOI: 10.3390/ma12213614Google ScholarThere is no corresponding record for this reference.
- 143Wang, Q.; Yang, X.; Li, C.; Zhao, C.; Huang, Y.; Qiu, S.; Qin, Y.; Shi, C. Titanium-based lithium-ion sieve H2TiO3 with enhanced Li+ adsorption by magnetic Fe doping. Sep. Purif. Technol. 2024, 346, 127455, DOI: 10.1016/j.seppur.2024.127455Google ScholarThere is no corresponding record for this reference.
- 144Tu, Y.; Zhou, Z.; Wei, W.; Guan, L.; Liu, Y. X.; Xu, Z.; Liu, H.; Liu, Z. Ti surface-doped spinel lithium manganese oxide with porous structure and improved stability for lithium extraction from salt-lake brine. Chem. Eng. J. 2025, 503, 158533, DOI: 10.1016/j.cej.2024.158533Google ScholarThere is no corresponding record for this reference.
- 145Ma, W.; Huang, H.; Zhu, G.; Wang, X.; Kong, Q.; Shi, X. Fabrication of spinel-type H4Ti5O12 ion sieve for lithium recovery from aqueous resources: Adsorption performance and mechanism. Processes 2025, 13, 2981, DOI: 10.3390/pr13092981Google ScholarThere is no corresponding record for this reference.
- 146US4159311A Patent. Recovery of lithium from brines, 1979.Google ScholarThere is no corresponding record for this reference.
- 147Besserguenev, A. V.; Fogg, A. M.; Francis, R. J.; Price, S. J.; O’Hare, D.; Isupov, V. P.; Tolochko, B. P. Synthesis and structure of gibbsite intercalation compounds [LiAl2(OH)6]X (X = Cl, Br, NO3). Chem. Mater. 1997, 9, 241– 247, DOI: 10.1021/cm960316zGoogle ScholarThere is no corresponding record for this reference.
- 148Zhong, J.; Lin, S.; Yu, J. Li+ adsorption performance and mechanism of lithium-aluminum layered double hydroxides in low-grade brines. Desalination 2021, 505, 114983, DOI: 10.1016/j.desal.2021.114983Google ScholarThere is no corresponding record for this reference.
- 149Paranthaman, M. P.; Li, L.; Luo, J.; Hoke, T.; Ucar, H.; Moyer, B. A.; Harrison, S. Recovery of lithium from geothermal brine with lithium-aluminum layered double hydroxide chloride sorbents. Environ. Sci. Technol. 2017, 51, 13481– 13486, DOI: 10.1021/acs.est.7b03464Google ScholarThere is no corresponding record for this reference.
- 150Zhang, L.; Zhang, T.; Lv, S.; Cheng, S.; Dong, G.; Quintana, M.; Song, S.; Zhao, Y. Steering interlayer interactions of lithium-aluminum layered double hydroxide beads for stable lithium extraction from sulfate-type brines. Desalination 2024, 592, 118130, DOI: 10.1016/j.desal.2024.118130Google ScholarThere is no corresponding record for this reference.
- 151Gao, J.; Hong, M.; Zhong, J.; Na, S.; Su, Y.; Li, H.; Hu, M.; Yang, S. Semi-sacrificial fabrication of binder-free lithium-aluminum layered double hydroxide adsorbents for lithium extraction. Desalination 2025, 597, 118409, DOI: 10.1016/j.desal.2024.118409Google ScholarThere is no corresponding record for this reference.
- 152Wu, Y. H.; Guo, W.; Sun, Y. Z.; Sun, S. Y. Resin functionalized with 2-(hydroxymethyl)-12-crown-4 ether for selective adsorption of lithium ions from desilication solution of coal-based solid waste. Sep. Purif. Technol. 2025, 355, 129500, DOI: 10.1016/j.seppur.2024.129500Google ScholarThere is no corresponding record for this reference.
- 153Zhang, X.; Xing, X.; Zhao, X.; Yang, J.; Liu, M.; Liu, B.; Gao, X.; Huang, H. Crown ethers decorated metal-organic framework for selective lithium-ion sieving. Chem. Eng. J. 2025, 503, 158435, DOI: 10.1016/j.cej.2024.158435Google ScholarThere is no corresponding record for this reference.
- 154Liberati, A. F.; Geneste, A.; Prelot, B.; Mericq, J. P.; Faur, C.; Monge, S. Synthesis, characterization, and Li adsorption study of a new thermoresponsive crown-ether-bearing copolymer. Sep. Purif. Technol. 2025, 354, 129123, DOI: 10.1016/j.seppur.2024.129123Google ScholarThere is no corresponding record for this reference.
- 155Park, M. J.; Nisola, G. M.; Beltran, A. B.; Torrejos, R. E. C.; Seo, J. G.; Lee, S. P.; Kim, H.; Chung, W. J. Recyclable composite nanofiber adsorbent for Li+ recovery from seawater desalination retentate. Chem. Eng. J. 2014, 254, 73– 81, DOI: 10.1016/j.cej.2014.05.095Google ScholarThere is no corresponding record for this reference.
- 156Yu, J.; Zhu, J.; Luo, G.; Chen, L.; Li, X.; Cui, P.; Wu, P.; Chao, Y.; Zhu, W.; Liu, Z. 3D-printed titanium-based ionic sieve monolithic adsorbent for selective lithium recovery from salt lakes. Desalination 2023, 560, 116651, DOI: 10.1016/j.desal.2023.116651Google ScholarThere is no corresponding record for this reference.
- 157Ding, Y.; Nhung, N. T. H.; An, J.; Chen, H.; Liao, L.; He, C.; Wang, X.; Fujita, T. Manganese-titanium mixed ion sieves for selective adsorption of lithium ions from artificial salt-lake brine. Materials 2023, 16, 4190, DOI: 10.3390/ma16114190Google ScholarThere is no corresponding record for this reference.
- 158Zhao, K.; Tong, B.; Yu, X.; Guo, Y.; Xie, Y.; Deng, T. Synthesis of porous fiber-supported lithium ion-sieve adsorbent for lithium recovery from geothermal water. Chem. Eng. J. 2022, 430, 131423, DOI: 10.1016/j.cej.2021.131423Google ScholarThere is no corresponding record for this reference.
- 159Limjuco, L. A.; Nisola, G. M.; Lawagon, C. P.; Lee, S. P.; Seo, J. G.; Kim, H.; Chung, W. J. H2TiO3 composite adsorbent foam for efficient and continuous recovery of Li+ from liquid resources. Colloids Surf., A 2016, 504, 267– 279, DOI: 10.1016/j.colsurfa.2016.05.072Google ScholarThere is no corresponding record for this reference.
- 160Hong, H. J.; Park, I. S.; Ryu, T.; Ryu, J.; Kim, B. G.; Chung, K. S. Granulation of Li1.33Mn1.67O4 through cross-linked chitosan for effective recovery of Li+ from seawater. Chem. Eng. J. 2013, 234, 16– 22, DOI: 10.1016/j.cej.2013.08.060Google ScholarThere is no corresponding record for this reference.
- 161Qiu, Z.; Wang, M.; Chen, Y.; Zhang, T.; Yang, D.; Qiu, F. Li4Mn5O12-doped cellulose acetate membrane with low Mn loss and high stability for enhanced lithium extraction from seawater. Desalination 2021, 506, 115003, DOI: 10.1016/j.desal.2021.115003Google ScholarThere is no corresponding record for this reference.
- 162Tang, L.; Huang, S.; Wang, Y.; Liang, D.; Li, Y.; Li, J.; Wang, Y.; Xie, Y.; Wang, W. Highly efficient, stable, and recyclable hydrogen manganese oxide/cellulose film for lithium extraction from seawater. ACS Appl. Mater. Interfaces 2020, 12, 9775– 9781, DOI: 10.1021/acsami.9b21612Google ScholarThere is no corresponding record for this reference.
- 163Chen, S.; Chen, Z.; Wei, Z.; Hu, J.; Guo, Y.; Deng, T. Titanium-based ion sieve with enhanced post-separation ability for high-performance lithium recovery from geothermal water. Chem. Eng. J. 2021, 410, 128320, DOI: 10.1016/j.cej.2020.128320Google ScholarThere is no corresponding record for this reference.
- 164Xiao, J. L.; Sun, S. Y.; Song, X.; Li, P.; Yu, J. G. Lithium-ion recovery from brine using granulated polyacrylamide-MnO2 ion sieve. Chem. Eng. J. 2015, 279, 659– 666, DOI: 10.1016/j.cej.2015.05.075Google ScholarThere is no corresponding record for this reference.
- 165Zhang, X.; Wu, J.; He, Y.; Zhao, C.; An, M.; Li, L. Porous poly(vinyl alcohol)/polyacrylamide hydrogels loaded with HTO lithium-ion sieves for rapid and efficient Li+ extraction. Desalination 2024, 580, 117587, DOI: 10.1016/j.desal.2024.117587Google ScholarThere is no corresponding record for this reference.
- 166Meng, Z.; Wang, M.; Cao, X.; Wang, T.; Wang, Y.; Xu, Y.; Liu, W.; Chen, L.; Huang, Y.; Liu, X. Highly flexible interconnected Li+ ion-sieve porous hydrogels with self-regulating nanonetworks for marine lithium recovery. Chem. Eng. J. 2022, 445, 136780, DOI: 10.1016/j.cej.2022.136780Google ScholarThere is no corresponding record for this reference.
- 167Yang, L.; Tu, Y.; Li, H.; Zhan, W.; Hu, H.; Wei, Y.; Chen, C.; Liu, K.; Shao, P.; Li, M.; Yang, G.; Luo, X. Fluorine-rich supramolecular nanocontainer cross-linked hydrogel for lithium extraction with super-high capacity and selectivity. Angew. Chem., Int. Ed. 2023, 62, e202308702 DOI: 10.1002/anie.202308702Google ScholarThere is no corresponding record for this reference.
- 168Zhang, E.; Liu, W.; Liang, Q.; Liu, X.; Zhao, Z.; Yang, Y. Selective recovery of Li+ in acidic environments using an electroactive Li+-imprinted graphene-based hybrid aerogel. Chem. Eng. J. 2020, 385, 123948, DOI: 10.1016/j.cej.2019.123948Google ScholarThere is no corresponding record for this reference.
- 169Li, J.; Luo, Q.; Dong, M.; Nie, G.; Liu, Z.; Wu, Z. Granulated Li/Al-LDH adsorbents for lithium recovery from synthetic and real salt-lake brines. Hydrometallurgy 2022, 209, 105828, DOI: 10.1016/j.hydromet.2022.105828Google ScholarThere is no corresponding record for this reference.
- 170Wang, S.; Zheng, S.; Wang, Z.; Cui, W.; Zhang, H.; Yang, L.; Zhang, Y.; Li, P. Iron-doped lithium ion-sieves with superior lithium adsorption and magnetic separation behavior. Chem. Eng. J. 2018, 332, 160– 168, DOI: 10.1016/j.cej.2017.09.055Google ScholarThere is no corresponding record for this reference.
- 171Lin, Y.; Li, M.; Wang, S.; Xue, F.; Ju, S. Biomass carbonaceous aerogel-modified ion-sieve composites for lithium adsorption. Mater. Lett. 2024, 363, 136282, DOI: 10.1016/j.matlet.2024.136282Google ScholarThere is no corresponding record for this reference.
- 172Zhong, J.; Lin, S.; Yu, J. Lithium recovery from ultrahigh Mg2+/Li+ ratio brine using granulated Li/Al-LDH adsorbents. Sep. Purif. Technol. 2021, 256, 117780, DOI: 10.1016/j.seppur.2020.117780Google ScholarThere is no corresponding record for this reference.
- 173Zhang, Y.; Ray, A. K. Liquid-liquid extraction. In Coulson and Richardson’s Chemical Engineering; Elsevier: Oxford, 2023; Vol. 2B, pp 257– 312. DOI: 10.1016/B978-0-08-101097-6.00004-3 .Google ScholarThere is no corresponding record for this reference.
- 174Song, J. F.; Nghiem, L. D.; Li, X. M.; He, T. Lithium extraction from Chinese salt-lake brines: Opportunities, challenges, and future outlook. Environ. Sci. (Camb.) 2017, 3, 593– 597, DOI: 10.1039/C7EW00020KGoogle ScholarThere is no corresponding record for this reference.
- 175Zhou, Z.; Qin, W.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in three diluents. J. Chem. Eng. Data 2011, 56, 3518– 3522, DOI: 10.1021/je200246xGoogle ScholarThere is no corresponding record for this reference.
- 176Zhou, Z.; Qin, W.; Liu, Y.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in kerosene and FeCl3. J. Chem. Eng. Data 2012, 57, 82– 86, DOI: 10.1021/je200803hGoogle ScholarThere is no corresponding record for this reference.
- 177Harvianto, G. R.; Kim, S. H.; Ju, C. S. Solvent extraction and stripping of lithium ion from aqueous solution and application to seawater. Rare Met. 2016, 35, 948– 953, DOI: 10.1007/s12598-015-0453-1Google ScholarThere is no corresponding record for this reference.
- 178Ji, L.; Hu, Y.; Li, L.; Shi, D.; Li, J.; Nie, F.; Song, F.; Zeng, Z.; Sun, W.; Liu, Z. Lithium extraction using a synergistic system of dioctyl phthalate and tributyl phosphate in kerosene and FeCl3. Hydrometallurgy 2016, 162, 71– 78, DOI: 10.1016/j.hydromet.2016.02.018Google ScholarThere is no corresponding record for this reference.
- 179Su, H.; Li, Z.; Zhang, J.; Liu, W.; Zhu, Z.; Wang, L.; Qi, T. Regulation of Fe3+ coordination in ternary solvent systems for selective lithium extraction and easy stripping. ACS Sustain. Chem. Eng. 2020, 8, 1971– 1979, DOI: 10.1021/acssuschemeng.9b06432Google ScholarThere is no corresponding record for this reference.
- 180Su, H.; Li, Z.; Zhang, J.; Zhu, Z.; Wang, L.; Qi, T. Recovery of lithium from salt-lake brine using TBP-FeCl3-P507 mixed ternary solvent extraction. Hydrometallurgy 2020, 197, 105487, DOI: 10.1016/j.hydromet.2020.105487Google ScholarThere is no corresponding record for this reference.
- 181Shi, C.; Duan, D.; Jia, Y.; Jing, Y. Highly efficient lithium ion extraction using ionic-liquid-containing tributyl phosphate solvent systems. J. Mol. Liq. 2014, 200, 191– 195, DOI: 10.1016/j.molliq.2014.10.004Google ScholarThere is no corresponding record for this reference.
- 182Shi, C.; Jia, Y.; Zhang, C.; Liu, H.; Jing, Y. Extraction of lithium from salt-lake brine using room-temperature ionic liquids in tributyl phosphate. Fusion Eng. Des. 2015, 90, 1– 6, DOI: 10.1016/j.fusengdes.2014.09.021Google ScholarThere is no corresponding record for this reference.
- 183Shi, C.; Jing, Y.; Jia, Y. Solvent extraction of lithium ions by tributyl phosphate using room-temperature ionic liquids. J. Mol. Liq. 2016, 215, 640– 646, DOI: 10.1016/j.molliq.2016.01.025Google ScholarThere is no corresponding record for this reference.
- 184Shi, C.; Jing, Y.; Xiao, J.; Wang, X.; Yao, Y.; Jia, Y. Solvent extraction of lithium using non-fluorinated functionalized ionic liquids. Sep. Purif. Technol. 2017, 172, 473– 479, DOI: 10.1016/j.seppur.2016.08.034Google ScholarThere is no corresponding record for this reference.
- 185Yang, S.; Liu, G.; Wang, J.; Cui, L.; Chen, Y. Recovery of lithium from alkaline brine by solvent extraction with functionalized ionic liquids. Fluid Phase Equilib. 2019, 493, 129– 136, DOI: 10.1016/j.fluid.2019.04.015Google ScholarThere is no corresponding record for this reference.
- 186Yusuf, A.; Sodiq, A.; Giwa, A.; Eke, J.; Pikuda, O.; De Luca, G.; Di Salvo, J. L.; Chakraborty, S. Emerging trends in membrane science and technology for sustainable water treatment. J. Clean. Prod. 2020, 266, 121867, DOI: 10.1016/j.jclepro.2020.121867Google ScholarThere is no corresponding record for this reference.
- 187Li, X.; Mo, Y.; Qing, W.; Shao, S.; Tang, C. Y.; Li, J. Membrane-based technologies for lithium recovery from aqueous resources: A review. J. Membr. Sci. 2019, 591, 117317, DOI: 10.1016/j.memsci.2019.117317Google ScholarThere is no corresponding record for this reference.
- 188Wen, X.; Ma, P.; Zhu, C.; He, Q.; Deng, X. Preliminary study on recovering lithium chloride from lithium-containing waters by nanofiltration. Sep. Purif. Technol. 2006, 49, 230– 236, DOI: 10.1016/j.seppur.2005.10.004Google ScholarThere is no corresponding record for this reference.
- 189Sarkar, S.; SenGupta, A. K.; Prakash, P. The Donnan membrane principle: Opportunities for sustainable engineered processes and materials. Environ. Sci. Technol. 2010, 44, 1161– 1166, DOI: 10.1021/es9024029Google ScholarThere is no corresponding record for this reference.
- 190Sun, S. Y.; Cai, L. J.; Nie, X. Y.; Song, X.; Yu, J. G. Separation of magnesium and lithium from brine using desal nanofiltration membranes. J. Water Process Eng. 2015, 7, 210– 217, DOI: 10.1016/j.jwpe.2015.06.012Google ScholarThere is no corresponding record for this reference.
- 191Li, X.; Zhang, C.; Zhang, S.; Li, J.; He, B.; Cui, Z. Positively charged polyamide composite nanofiltration hollow fiber membranes for lithium and magnesium separation. Desalination 2015, 369, 26– 36, DOI: 10.1016/j.desal.2015.04.027Google ScholarThere is no corresponding record for this reference.
- 192Zhang, H. Z.; Xu, Z. L.; Ding, H.; Tang, Y. J. Positively charged capillary nanofiltration membranes with high Mg2+/Ca2+ rejection and Mg2+/Li+ separation. Desalination 2017, 420, 158– 166, DOI: 10.1016/j.desal.2017.07.011Google ScholarThere is no corresponding record for this reference.
- 193Wu, H.; Lin, Y.; Feng, W.; Liu, T.; Wang, L.; Yao, H.; Wang, X. Ionic-liquid-based nanofiltration membranes for lithium extraction from high Mg2+/Li+ brines. J. Membr. Sci. 2020, 603, 117997, DOI: 10.1016/j.memsci.2020.117997Google ScholarThere is no corresponding record for this reference.
- 194Wu, X.; Liu, H.; Wei, Y.; Fei, Y.; Qi, H. Negatively charged organic-inorganic hybrid silica nanofiltration membranes for lithium extraction. Chin. J. Chem. Eng. 2020, 28, 749– 757, DOI: 10.1016/j.cjche.2019.11.004Google ScholarThere is no corresponding record for this reference.
- 195Pramanik, B. K.; Asif, M. B.; Kentish, S.; Nghiem, L. D.; Hai, F. I. Lithium enrichment from simulated salt-lake brine using integrated nanofiltration-membrane distillation. J. Environ. Chem. Eng. 2019, 7, 103395, DOI: 10.1016/j.jece.2019.103395Google ScholarThere is no corresponding record for this reference.
- 196Nie, X. Y.; Sun, S. Y.; Sun, Z.; Song, X.; Yu, J. G. Ion fractionation of lithium and magnesium by electrodialysis using monovalent selective ion-exchange membranes. Desalination 2017, 403, 128– 135, DOI: 10.1016/j.desal.2016.05.010Google ScholarThere is no corresponding record for this reference.
- 197Chen, Q. B.; Ji, Z. Y.; Liu, J.; Zhao, Y. Y.; Wang, S. Z.; Yuan, J. S. Effect of coexisting cations on lithium migration during selective electrodialysis. J. Membr. Sci. 2018, 548, 408– 420, DOI: 10.1016/j.memsci.2017.11.040Google ScholarThere is no corresponding record for this reference.
- 198Jiang, C.; Wang, Y.; Wang, Q.; Feng, H.; Xu, T. Production of lithium hydroxide from lake brines through electro-electrodialysis with bipolar membranes. Ind. Eng. Chem. Res. 2014, 53, 6103– 6112, DOI: 10.1021/ie404334sGoogle ScholarThere is no corresponding record for this reference.
- 199Yang, D.; Yang, Y.; Wong, T.; Iguodala, S.; Wang, A.; Lovell, L.; Foglia, F.; Fouquet, P.; Breakwell, C.; Fan, Z.; Wang, Y.; Britton, M. M.; Williams, D. R.; Shah, N.; Xu, T.; McKeown, N. B.; Titirici, M.-M.; Jelfs, K. E.; Song, Q. Solution-processable polymer membranes with hydrophilic subnanometer pores for sustainable lithium extraction. Nat. Water 2025, 3, 319– 333, DOI: 10.1038/s44221-025-00398-8Google ScholarThere is no corresponding record for this reference.
- 200Martínez-López, J. C.; Santos Rodríguez, M.; Oliver Cuenca, V.; Silva Testa, G.; van Eck, E.; Zhao, E. W.; Lozano, Á. E.; Álvarez, C.; Carretero-González, J. Dibenzodioxin-based polymers of intrinsic microporosity with enhanced lithium-ion transport. Macromolecules 2024, 57, 9442– 9456, DOI: 10.1021/acs.macromol.4c01243Google ScholarThere is no corresponding record for this reference.
- 201Gou, F.; Wang, Q.; Yang, Z.; Chang, W.; Shen, J.; Zeng, H. Artificial lithium channels built from polymers of intrinsic microporosity. Angew. Chem., Int. Ed. 2025, 64, e202418304 DOI: 10.1002/anie.202418304Google ScholarThere is no corresponding record for this reference.
- 202Lei, D.; Zhu, Y.; Lou, L. L.; Liu, Z. Covalent organic framework membranes for lithium extraction: Facilitated ion-transport strategies to enhance selectivity. Mater. Horiz. 2025, 12, 5459– 5472, DOI: 10.1039/D5MH00457HGoogle ScholarThere is no corresponding record for this reference.
- 203Meng, W.; Chen, S.; Wu, M.; Gao, F.; Hou, Y.; Zhan, X.; Hu, W.; Liang, L.; Zhang, Q. Dehydration-enhanced ion recognition of triazine covalent organic frameworks for high-resolution Li+/Mg2+ separation. Angew. Chem., Int. Ed. 2025, 64, e202422423 DOI: 10.1002/anie.202422423Google ScholarThere is no corresponding record for this reference.
- 204Bao, S.; Ma, Z.; Yu, L.; Li, Q.; Xia, J.; Song, S.; Sui, K.; Zhao, Y.; Liu, X.; Gao, J. Randomly oriented covalent organic framework membranes for selective Li+ sieving. Nat. Commun. 2025, 16, 3896, DOI: 10.1038/s41467-025-59188-1Google ScholarThere is no corresponding record for this reference.
- 205Habiba, U.; Siddique, T. A.; Fattah, I. M. R.; Mohammed, S.; Attenborough, E.; Lai, Q.; Wang, Z. Porous alumina-supported lithium aluminum titanium phosphate membrane for lithium extraction via electrodialysis. Sep. Purif. Technol. 2025, 354, 128657, DOI: 10.1016/j.seppur.2024.128657Google ScholarThere is no corresponding record for this reference.
- 206Epp, V.; Ma, Q.; Hammer, E. M.; Tietz, F.; Wilkening, M. Very fast bulk lithium-ion diffusivity in crystalline Li1.5Al0.5Ti1.5(PO4)3. Phys. Chem. Chem. Phys. 2015, 17, 32115– 32121, DOI: 10.1039/C5CP05337DGoogle ScholarThere is no corresponding record for this reference.
- 207Jeong, W.; Liu, L.; Lim, H. S.; Chung, J.; Chen, K.; Ryu, B. K. Improved ion conductivity of lithium aluminum titanium phosphate solid-state electrolytes by divalent cation addition. J. Korean Ceram. Soc. 2025, 62, 83– 89, DOI: 10.1007/s43207-024-00419-4Google ScholarThere is no corresponding record for this reference.
- 208Gulied, M.; Zavahir, S.; Elmakki, T.; Park, H.; Gago, G. H.; Shon, H. K.; Han, D. S. Efficient lithium recovery from simulated brine using a hybrid DCMD-ESIX system. Desalination 2024, 572, 117127, DOI: 10.1016/j.desal.2023.117127Google ScholarThere is no corresponding record for this reference.
- 209Wan, L.; Zhang, T.; Li, H.; Huang, Q.; Hu, J.; Pan, L. In situ construction of rod-shaped Fe3O4/N-doped carbon architectures for superior lithium extraction via hybrid capacitive deionization. Desalination 2025, 601, 118538, DOI: 10.1016/j.desal.2025.118538Google ScholarThere is no corresponding record for this reference.
- 210Zegeye, R. B.; Tufa, R. A.; Santoro, S.; Inzillo, B. M.; Aquino, M.; Politano, G. G.; Embaye, A. S.; Argurio, P.; De Bartolo, L.; Curcio, E. Sustainable lithium recovery from geothermal brine via integrated membrane distillation-selective electrodialysis. Green Chem. 2025, 27, 4205– 4221, DOI: 10.1039/D4GC06530AGoogle ScholarThere is no corresponding record for this reference.
- 211Siekierka, A. Lithium and magnesium separation from brines by hybrid capacitive deionization. Desalination 2022, 527, 115569, DOI: 10.1016/j.desal.2022.115569Google ScholarThere is no corresponding record for this reference.
- 212Baudino, L.; Santos, C.; Pirri, C. F.; La Mantia, F.; Lamberti, A. Recent advances in lithium recovery from water resources: From passive to electrochemical methods. Adv. Sci. 2022, 9, 2201380, DOI: 10.1002/advs.202201380Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
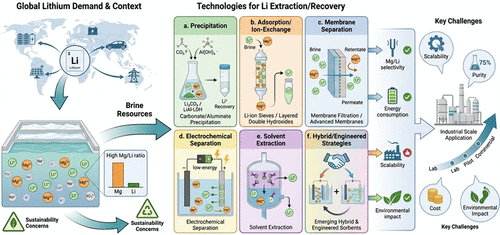
Figure 1
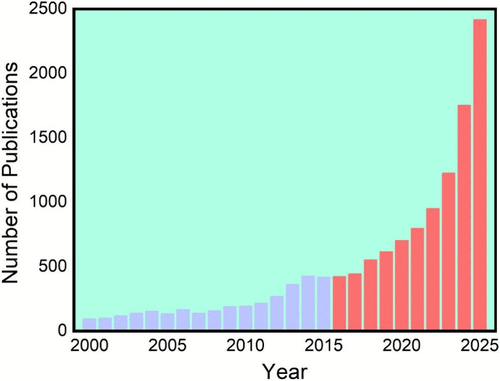
Figure 1. Publication counts retrieved from the Scopus database using the search query: TITLE-ABS-KEY (lithium AND extraction) AND PUBYEAR > 1999 AND PUBYEAR < 2026.
Figure 2

Figure 2. Different methods or techniques for Li extraction from brine.
Figure 3
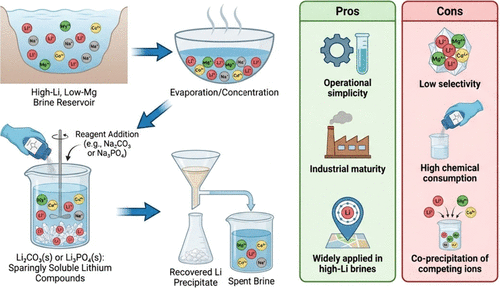
Figure 3. Schematic illustration of lithium recovery via chemical precipitation from high-Li brines, highlighting the process steps, recovered lithium products, and key advantages and limitations of the method.
Figure 4

Figure 4. (a) Flowsheet for the crystallization-precipitation method adapted with permission from ref (80). Copyright 2020, Elsevier. (b, c) Effects of the reaction time on (b) percentage recovery and (c) precipitation rates; (d, e) effects of stirring rate on (d) percentage recovery and (e) precipitation rates of Li and Mg-ion recovery adapted with permission from ref (81). Copyright 2019, Elsevier.
Figure 5

Figure 5. Overview of lithium recovery pathways from brines: (a) conventional evaporation–precipitation processes characterized by high energy and chemical consumption; (b) electrochemical lithium extraction methods offering improved energy efficiency and selectivity; (c) lithium insertion (charging) into lithium-selective electrodes under an applied potential; (d) lithium extraction (discharging) through controlled release of Li+ ions into a recovery solution; and (e) overall advantages of insertion–extraction systems operating under mild conditions with high lithium selectivity and reduced energy and chemical requirements.
Figure 6

Figure 6. (a–c) Schematic illustration of reactions occurring at the anode and cathode sides Li extraction device; (d, e) comparison of the anode and cathode electrode potential with pH variations; (f) analysis of Mn dissolution losses at the anode and ion intercalation capacity at the cathode adapted with permission from ref (95). Copyright 2025, Elsevier. (g) Structure of the electrolytic cell adapted with permission from ref (96). Copyright 2021, Elsevier. (h) Schematic diagram of electrodes preparation adapted with permission from ref (97). Copyright 2024, Elsevier. (i, j) Mg/Li ratio, Li+ concentration and current density for each cycle adapted with permission from ref (98). Copyright 2021, Elsevier.
Figure 7
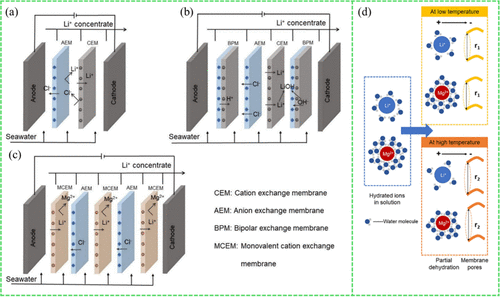
Figure 7. Schematic of (a) traditional electrodialysis, (b) bipolar membrane electrodialysis, and (c) selective electrodialysis adapted with permission from ref (102). Copyright 2024, Elsevier. (d) Changes in the behavior of partially dehydrated ions and membrane pore structure at varying temperatures adapted with permission from ref (103). Copyright 2020, Elsevier.
Figure 8

Figure 8. (a) Rate of Li removal and (b) results of Li extraction adapted with permission from ref (104). Copyright 2022, Elsevier. (c) Schematic representation of the multistage integrated membrane process, which includes a single-stage NF, followed by a first-stage and second-stage S-ED adapted with permission from ref (105). Copyright 2023, Elsevier. (d, e) Illustration of schematic of Li-Mg migration in (d) batch S-ED; (e) feed and bleed mode adapted with permission from ref (106). Copyright 2021, Elsevier. (f) Complete scheme of the fabrication of LATP membrane and the Mg/Li separation adapted with permission from ref (107). Copyright 2024, Elsevier.
Figure 9

Figure 9. (a) Design of ESIP stack for continuous Li-ions separation; (b) the mechanism illustrating Li-ions separation; (c) the diffusion pathway and (d) energy barrier analysis; (e) mean square displacement of Li and Mg-ions, with diffusion coefficient shown in the inset adapted with permission from ref (108). Copyright 2024, Elsevier.
Figure 10
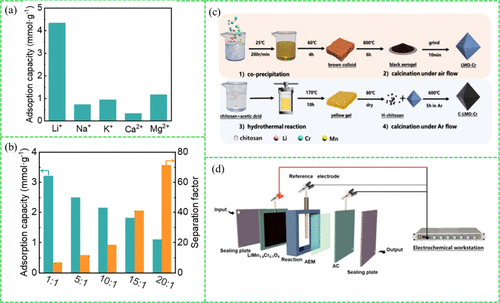
Figure 10. (a) The adsorption capacity in single-salt solutions (0.05 mol/L); (b) separation factor and adsorption capacity across different Mg/Li ratios adapted with permission from ref (109). Copyright 2024, Elsevier. (c) Synthesis process of electrodes; (d) representation of HCDI device adapted with permission from ref (110). Copyright 2024, Elsevier.
Figure 11
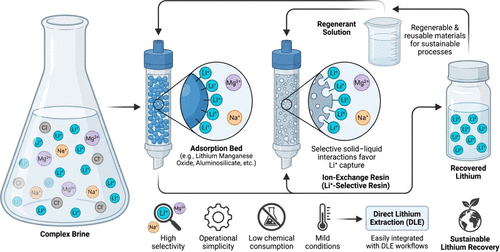
Figure 11. Overview of adsorption and ion-exchange–based direct lithium extraction (DLE) from brines, highlighting selective Li-ion capture, regeneration, and sustainable lithium recovery.
Figure 12

Figure 12. (a) The full process of the formation of ion-sieves adapted with permission from ref (137). Copyright 2020, Elsevier. (b) Process of the selective Li-ion adsorption adapted with permission from ref (22). Copyright 2024, Elsevier. (c) Plot of the adsorption kinetics; (d, e) plots of pseudo-first-order kinetics and pseudo-second-order kinetics (d) HFTO and (e) HTO. Adapted with permission from ref (143). Copyright 2024, Elsevier. (f) The method of the preparation of HMO-Ti ionic sieves and its adsorption–desorption mechanism; adapted with permission from ref (144). Copyright 2025, Elsevier.
Figure 13

Figure 13. (a) The steering interlayer interaction strategy in LiAl-LDHs beads; (b) effect of PAN concentration on Li extraction; (c) adsorption kinetics; (d) intraparticle diffusion and (e) desorption of Li from BLDH-P and BLDH-Cl adapted with permission from ref (150). Copyright 2024, Elsevier. (f) The full scheme of binder less LiAl-LDH@NiAl-F preparation adapted with permission from ref (151). Copyright 2025, Elsevier.
Figure 14

Figure 14. (a) The adsorption isotherm and kinetics plots of Li-ions adsorption onto MOF-808-12C4E adapted with permission from ref (153). Copyright 2025, Elsevier. (b) The full scheme of the synthesis of BCEEM and further copolymerization with NIPAM adapted with permission from ref (154). Copyright 2025, Elsevier. (c) Complete scheme of the synthesis of PPy@HNTs/PVA hydrogel composite adapted with permission from ref (32). Copyright 2024, Elsevier. (d) The plots of adsorption isotherm, kinetics, and multicycle use of PPy@HNTs/PVA for Li-ions extraction adapted with permission from ref (32). Copyright 2024, Elsevier.
Figure 15

Figure 15. (a) Liquid–liquid equilibrium data for Li solution with TBP and cosolvents with FeCl3 as a coextracting agent adapted with permission from ref (175). Copyright 2011, American Chemical Society. (b) Relationship between the partition coefficient of Li and the extractability of iron at different Fe/Li ratios adapted with permission from ref (176). Copyright 2012, American Chemical Society. (c) Calculation of the theoretical stages of extraction using a McCabe–Thiele diagram adapted with permission from ref (178). Copyright 2016, Elsevier. (d) Simulation flow diagram for a three stage counter-current extraction process at an organic/aqueous ratio of 2/1 with initial Fe/Li = 1.3 for 10 min; adapted with permission from ref (178). Copyright 2016, Elsevier.
Figure 16

Figure 16. (a) Li extraction and stripping mechanism using the ternary solvent approach TBP-FeCl3-P507; (b) proposed structures as a result of the presence of P507 (2-ethylhexyl phosphonic acid mono-2-ethylhexyl ester); adapted with permission from ref (179). Copyright 2020, American Chemical Society. (c) tThe effect of dosage of IL in TBP as a function of extraction efficiency adapted with permission from ref (181). Copyright 2014, Elsevier. (d) Extraction efficiency as a function of ratio of molar ratio of ClO4– and Li-ions adapted with permission from ref (182). Copyright 2015, Elsevier. (e) Dosage of ILs as a function of extraction efficiency and distribution ratio from ref (183). Copyright 2016, Elsevier. (f) Extraction efficiency of synthesized ILs and [DEHP], as a function of HCl concentration in the aqueous phase adapted with permission from ref (184). Copyright 2017, Elsevier.
Figure 17

Figure 17. (a) Effect of pressure on rejection rate of ions; (b) effect of pH on rejection rate of ions; (c) effect of pH on separation factor and permeate amount of pure water adapted with permission from ref (190). Copyright 2015, Elsevier. (d) Flux and rejection for different salts by the positively charged composite hollow fiber as a function DAPP concentration adapted with permission from ref (191). Copyright 2015, Elsevier. Panels (e) and (f) permeate flux and rejection rate performance as a function of time for two simulated brine mixtures adapted with permission from ref (192). Copyright 2017, Elsevier.
Figure 18

Figure 18. (a–d) Performance parameters of membrane at different concentrations of ILs: (a) flux; (b) percent salt rejection; (c) ion rejection; and (d) selectivity (conditions: 0.6 MPa, pH = 6.4) adapted with permission from ref (193). Copyright 2020, Elsevier. (e) Effect of time on the (Mg/Li)P ratio of the permeate stream at varying feeds ratios of Mg/Li; adapted with permission from ref (196). Copyright 2017, Elsevier. (f, g) Effect of time on the (f) migration rate and (g) recovery of Li-ion at different ratios of K/L adapted with permission from ref (197). Copyright 2018, Elsevier.
Figure 19

Figure 19. Overview of conventional lithium recovery techniques, including solvent extraction, precipitation, electrochemical extraction, and adsorption-based methods, highlighting their key limitations.
References
This article references 212 other publications.
- 1Hensel, N. D. Economic challenges in the clean energy supply chain: The market for rare earth minerals and other critical inputs. Bus. Econ. 2011, 46 (3), 171– 184, DOI: 10.1057/be.2011.17There is no corresponding record for this reference.
- 2Diouf, B.; Pode, R. Potential of lithium-ion batteries in renewable energy. Renew. Energy 2015, 76, 375– 380, DOI: 10.1016/j.renene.2014.11.058There is no corresponding record for this reference.
- 3Dorn, F. M.; Peyré, F. R. Lithium as a strategic resource: Geopolitics, industrialization, and mining in Argentina. J. Lat. Am. Geogr. 2020, 19 (1), 68– 90, DOI: 10.1353/lag.2020.0101There is no corresponding record for this reference.
- 4Role of Critical Minerals in Clean Energy Transitions; IEA: Paris, 2021. https://www.iea.org/reports/the-role-of-critical-minerals-in-clean-energy-transitions (accessed June 17, 2025).There is no corresponding record for this reference.
- 5Kanagasundaram, T.; Murphy, O.; Haji, M. N.; Wilson, J. J. The recovery and separation of lithium using solvent extraction methods. Coord. Chem. Rev. 2024, 509, 215727, DOI: 10.1016/j.ccr.2024.215727There is no corresponding record for this reference.
- 6Choubey, P. K.; Kim, M. S.; Srivastava, R. R.; Lee, J. C.; Lee, J. Y. Exploitation of the prominent energy-storage element lithium. Part I: From mineral and brine resources. Miner. Eng. 2016, 89, 119– 137, DOI: 10.1016/j.mineng.2016.01.010There is no corresponding record for this reference.
- 7Swain, B. Recovery and recycling of lithium: A review. Sep. Purif. Technol. 2017, 172, 388– 403, DOI: 10.1016/j.seppur.2016.08.031There is no corresponding record for this reference.
- 8Wang, J.; Yue, X.; Wang, P.; Yu, T.; Du, X.; Hao, X.; Abudula, A.; Guan, G. Electrochemical technologies for lithium recovery from liquid resources: A review. Renew. Sustain. Energy Rev. 2022, 154, 111813, DOI: 10.1016/j.rser.2021.111813There is no corresponding record for this reference.
- 9Epstein, J. A.; Feist, E. M.; Zmora, J.; Marcus, Y. Extraction of lithium from the Dead Sea. Hydrometallurgy 1981, 6 (3), 269– 275, DOI: 10.1016/0304-386X(81)90044-XThere is no corresponding record for this reference.
- 10Martin, G.; Rentsch, L.; Höck, M.; Bertau, M. Lithium market research: Global supply, future demand, and price development. Energy Storage Mater. 2017, 6, 171– 179, DOI: 10.1016/j.ensm.2016.11.004There is no corresponding record for this reference.
- 11Balaram, V.; Santosh, M.; Satyanarayanan, M.; Srinivas, N.; Gupta, H. Lithium: A review of applications, occurrence, exploration, extraction, recycling, analysis, and environmental impact. Geosci. Front. 2024, 15, 101868, DOI: 10.1016/j.gsf.2024.101868There is no corresponding record for this reference.
- 12Sajna, M. S.; Elmakki, T.; Zavahir, S.; Tariq, H.; Abdulhameed, A.; Park, H.; Shon, H. K.; Han, D. S. Enhanced lithium separation from brines using nanofiltration (NF) technology: A review. Desalination 2024, 592, 118148, DOI: 10.1016/j.desal.2024.118148There is no corresponding record for this reference.
- 13Gao, Y.; Sun, Y.; Chen, L.; Tan, Y.; Fan, R.; Hai, C.; Zhang, H. Progress of research in titanium-based lithium-ion sieves for the extraction of lithium from salt lake brine. ACS Omega 2025, 10, 11643, DOI: 10.1021/acsomega.4c08886There is no corresponding record for this reference.
- 14Ramesh, M.; Tamil Selvan, M.; Rama Chandran, P.; Hariprasad, P.; Arunkumar, A.; Ram Kumar, C.; Prem Kumar, R. Experimental investigation on the effect of lithium and alkali-resistant glass fiber on aluminum AA5383 fabricated by the stir-casting process. Mater. Today Proc. 2023, DOI: 10.1016/j.matpr.2023.05.152There is no corresponding record for this reference.
- 15Zhang, K.; Tang, H.; Shi, X.; Xue, Y.; Huang, Q. Effect of Ti3C2 MXene additive on the tribological properties of lithium grease at different temperatures. Wear 2023, 526–527, 204953, DOI: 10.1016/j.wear.2023.204953There is no corresponding record for this reference.
- 16Gu, Y.; Zhong, K.; Cao, R.; Yang, Z. Aqueous lithium chloride solution as a non-toxic bactericidal and fungicidal disinfectant for air-conditioning systems: Efficacy and mechanism. Environ. Res. 2022, 212, 113112, DOI: 10.1016/j.envres.2022.113112There is no corresponding record for this reference.
- 17Gu, J.; Liang, B.; Luo, X.; Zhang, X.; Yuan, W.; Xiao, B.; Tang, X. Recent advances and future prospects of lithium recovery from low-grade lithium resources: A review. Inorganics 2025, 13 (1), 4, DOI: 10.3390/inorganics13010004There is no corresponding record for this reference.
- 18Rahighi, R.; Hosseini-Hosseinabad, S. M.; Zeraati, A. S.; Suwaileh, W.; Norouzi, A.; Panahi, M.; Gholipour, S.; Karaman, C.; Akhavan, O.; Khollari, M. A. R.; Vinu, A.; Rashidi, A.; Abdala, A.; Karimi-Maleh, H.; Orooji, Y. Two-dimensional materials in enhancement of membrane-based lithium recovery from metallic-ion-rich wastewaters: A review. Desalination 2022, 543, 116096, DOI: 10.1016/j.desal.2022.116096There is no corresponding record for this reference.
- 19Park, J.; Lee, J.; Shim, I. T.; Kim, E.; Nam, S. H.; Koo, J. W.; Hwang, T. M. Electrochemical direct lithium extraction: A review of electrodialysis and capacitive deionization technologies. Resources 2025, 14, 27, DOI: 10.3390/resources14020027There is no corresponding record for this reference.
- 20Marthi, R.; Smith, Y. R. Selective recovery of lithium from the Great Salt Lake using lithium manganese oxide-diatomaceous earth composite. Hydrometallurgy 2019, 186, 115– 125, DOI: 10.1016/j.hydromet.2019.03.011There is no corresponding record for this reference.
- 21Rivard, E.; Trudeau, M.; Zaghib, K. Hydrogen storage for mobility: A review. Materials 2019, 12, 1973, DOI: 10.3390/ma12121973There is no corresponding record for this reference.
- 22Zhang, L.; Zhang, T.; Lv, S.; Song, S.; Galván, H. J. O.; Quintana, M.; Zhao, Y. Adsorbents for lithium extraction from salt lake brine with high magnesium/lithium ratio: From structure-performance relationship to industrial applications. Desalination 2024, 579, 117480, DOI: 10.1016/j.desal.2024.117480There is no corresponding record for this reference.
- 23Mojid, M. R.; Lee, K. J.; You, J. Advances in direct lithium extraction from continental brines: Ion-sieve adsorption and electrochemical methods for varied Mg/Li ratios. Sustainable Mater. Technol. 2024, 40, e00923 DOI: 10.1016/j.susmat.2024.e00923There is no corresponding record for this reference.
- 24Battistel, A.; Palagonia, M. S.; Brogioli, D.; La Mantia, F.; Trócoli, R. Electrochemical methods for lithium recovery: A comprehensive and critical review. Adv. Mater. 2020, 32, 1905440, DOI: 10.1002/adma.201905440There is no corresponding record for this reference.
- 25Tabelin, C. B.; Dallas, J.; Casanova, S.; Pelech, T.; Bournival, G.; Saydam, S.; Canbulat, I. Towards a low-carbon society: Lithium resource availability, challenges, and innovations in mining, extraction, and recycling. Miner. Eng. 2021, 163, 106743, DOI: 10.1016/j.mineng.2020.106743There is no corresponding record for this reference.
- 26Wu, L.; Zhang, C.; Kim, S.; Hatton, T. A.; Mo, H.; Waite, T. D. Lithium recovery using electrochemical technologies: Advances and challenges. Water Res. 2022, 221, 118822, DOI: 10.1016/j.watres.2022.118822There is no corresponding record for this reference.
- 27Vera, M. L.; Torres, W. R.; Galli, C. I.; Chagnes, A.; Flexer, V. Environmental impact of direct lithium extraction from brines. Nat. Rev. Earth Environ. 2023, 4, 149– 165, DOI: 10.1038/s43017-022-00387-5There is no corresponding record for this reference.
- 28Ni, C.; Liu, C.; Wang, J.; Liang, Y.; Xie, W.; Zhong, H.; He, Z. Advances and promotion strategies of processes for extracting lithium from mineral resources. J. Ind. Eng. Chem. 2024, 140, 47– 64, DOI: 10.1016/j.jiec.2024.05.052There is no corresponding record for this reference.
- 29Tansel, B. Significance of thermodynamic and physical characteristics on permeation of ions during membrane separation: Hydrated radius, hydration free energy, and viscous effects. Sep. Purif. Technol. 2012, 86, 119– 126, DOI: 10.1016/j.seppur.2011.10.033There is no corresponding record for this reference.
- 30Orooji, Y.; Nezafat, Z.; Nasrollahzadeh, M.; Shafiei, N.; Afsari, M.; Pakzad, K.; Razmjou, A. Recent advances in nanomaterial development for lithium ion-sieving technologies. Desalination 2022, 529, 115624, DOI: 10.1016/j.desal.2022.115624There is no corresponding record for this reference.
- 31Zhao, Z.; Si, X.; Liu, X.; He, L.; Liang, X. Lithium extraction from high Mg/Li ratio brine using LiFePO4/FePO4 electrode materials. Hydrometallurgy 2013, 133, 75– 83, DOI: 10.1016/j.hydromet.2012.11.013There is no corresponding record for this reference.
- 32Zhou, J.; Li, X.; Xie, W.; Chen, Q.; Jiang, Y.; Wu, X.; Yang, L. Efficient solar evaporation and rapid lithium sorption by halloysite nanotube-based hydrogel. J. Clean. Prod. 2024, 437, 140523, DOI: 10.1016/j.jclepro.2023.140523There is no corresponding record for this reference.
- 33Gulied, M.; Zavahir, S.; Elmakki, T.; Park, H.; Gago, G. H.; Shon, H. K.; Han, D. S. Efficient lithium recovery from simulated brine using a hybrid DCMD-ESIX system. Desalination 2024, 572, 117127, DOI: 10.1016/j.desal.2023.117127There is no corresponding record for this reference.
- 34Wen, T.; Zhao, Y.; Wang, X.; Chen, L.; Gao, R.; Wang, S.; Zhang, T. Ultrafast separation of Li+ and Mg2+ by porous two-dimensional perm-selective montmorillonite membrane. Chem. Eng. J. 2023, 475, 146101, DOI: 10.1016/j.cej.2023.146101There is no corresponding record for this reference.
- 35Paredes, C.; Rodríguez de San Miguel, E. Selective lithium extraction from diluted alkaline aqueous media using polymer inclusion membranes. Desalination 2020, 487, 114500, DOI: 10.1016/j.desal.2020.114500There is no corresponding record for this reference.
- 36Zhu, Z.; Li, G. Polyaniline-covered lithium manganese oxide for high-performance capacitive deionization lithium extraction. ECS Meet. Abstr. 2025, MA2025-01, 1455 DOI: 10.1149/MA2025-01261455MTGABSThere is no corresponding record for this reference.
- 37Yoo, H.; Jang, H. J.; Kim, C.; Song, J.; Senthamaraikannan, T. G.; Lim, D. H.; Hong, H. J. Antimony-doped layered hydrogen titanium oxide as an advanced lithium-ion sieve. Desalination 2025, 614, 119175, DOI: 10.1016/j.desal.2025.119175There is no corresponding record for this reference.
- 38Zia, S.; Kashif, M.; Ge, H.; Zhao, K.; Guo, Y.; Deng, T. Advanced PDMS@Li/ZnAl-LDH microspheres for lithium recovery from Qarhan Salt Lake brine. Desalination 2025, 615, 119286, DOI: 10.1016/j.desal.2025.119286There is no corresponding record for this reference.
- 39Dai, Y.; Zhang, X.; Sun, J.; Wu, S.; Zhao, Z.; Xu, W.; Ren, G.; Li, Y.; Liu, D. Porous manganese-based ion sieve for sustainable lithium extraction from brine. Desalination 2025, 614, 119168, DOI: 10.1016/j.desal.2025.119168There is no corresponding record for this reference.
- 40Deng, G.; Jiang, R.; Tang, F.; Hou, Z.; Feng, Y. Porous hydrophilic ion sieves for lithium recovery from alkaline brines: Batch and continuous operation. Desalination 2025, 614, 119159, DOI: 10.1016/j.desal.2025.119159There is no corresponding record for this reference.
- 41Evans, B. R.; Jayanthi, K.; Popovs, I.; Moyer, B. A.; Paranthaman, M. P. New directions and principles for solvent extraction for recovery of lithium from aqueous brines and mineral leachates: A brief review. Sustainable Mater. Technol. 2025, 46, e01724 DOI: 10.1016/j.susmat.2025.e01724There is no corresponding record for this reference.
- 42González, A.; Choque, G.; Grágeda, M.; Ushak, S. Membrane-assisted electrochemical removal of Mg2+ and Ca2+ from lithium brines: Effects of temperature and current density through a zeta potential approach. Membranes 2025, 15, 250, DOI: 10.3390/membranes15090250There is no corresponding record for this reference.
- 43Wang, R.; Sheng, M.; Liu, X.; Wang, Q.; Wang, Z.; Wang, L. Electrosorption theoretically outperforms nanofiltration and electrodialysis for direct lithium extraction from brines. Environ. Sci. Technol. 2026, 60, 1577– 1595, DOI: 10.1021/acs.est.5c13571There is no corresponding record for this reference.
- 44Li, C.; Wang, X.; Xie, Y.; Li, J.; Zhang, J. Extraction of lithium from salt-lake brine by functional ionic solid porous polymer adsorption material. J. Environ. Chem. Eng. 2025, 13, 117344, DOI: 10.1016/j.jece.2025.117344There is no corresponding record for this reference.
- 45Yin, Z.; Zhao, W.; Sun, Y.; Wang, G.; Ma, T.; Sun, J.; Ji, P. Application of fly ash-based iron-doped lithium aluminum layered double hydroxide in brine-type lithium resources extraction: Adsorption mechanism analysis and life cycle assessment. Chem. Eng. J. 2025, 520, 165828, DOI: 10.1016/j.cej.2025.165828There is no corresponding record for this reference.
- 46Abujayyab, M.; Hasan, S. W.; Arafat, H. A.; Banat, F. High-efficiency lithium recovery from desalination brine via MnAl layered double hydroxide. Desalination 2025, 616, 119376, DOI: 10.1016/j.desal.2025.119376There is no corresponding record for this reference.
- 47Chen, J.; Lin, S.; Yu, J. High-selective cyclic adsorption and magnetic recovery performance of magnetic lithium-aluminum layered double hydroxides (MLDHs) in extracting Li+ from ultrahigh Mg/Li ratio brines. Sep. Purif. Technol. 2021, 255, 117710, DOI: 10.1016/j.seppur.2020.117710There is no corresponding record for this reference.
- 48Zhao, L.; Zhang, X.; Liu, K.; Liu, X.; Gong, Y.; Peng, H.; Qi, W. Convenient synthesis of granulated Li/Al-layered double hydroxides/chitosan composite adsorbents for lithium extraction from simulated brine with a high Mg2+/Li+ ratio. Chem. Eng. J. 2024, 501, 157780, DOI: 10.1016/j.cej.2024.157780There is no corresponding record for this reference.
- 49Zhang, J.; Ji, K.; Wang, Z.; Shi, W.; Zhao, Z.; Du, X.; Hao, X. Strongly cross-linked three-dimensional Li1-xMn2O4/C/SA-c-PEO@GF electrode for lithium recovery from high Mg2+/Li+ ratio salt lake brine. Sep. Purif. Technol. 2025, 376, 134102, DOI: 10.1016/j.seppur.2025.134102There is no corresponding record for this reference.
- 50Nadzri, N.; Lim, Y. J.; Liao, X.; Liang, Y.; Goh, K.; Liao, Y.; Goto, A.; Wang, R. Lithium recovery from seawater desalination brines using ion-sieve electrospun nanofibrous membranes: The role of nanofiber design. Chem. Eng. J. 2025, 513, 162886, DOI: 10.1016/j.cej.2025.162886There is no corresponding record for this reference.
- 51Yin, X.; Xu, P.; Wang, H. Applications of crown ether-based materials for enhancing lithium recovery from brines. Sep. Purif. Technol. 2025, 371, 133093, DOI: 10.1016/j.seppur.2025.133093There is no corresponding record for this reference.
- 52Dach, E.; Marston, J.; Abu-Obaid, S.; Peng, A.; Yip, N. Y. A novel approach for direct lithium extraction from alkali metal cations in brine mixtures using thermally switchable solvents. Joule 2026, 10, 102265, DOI: 10.1016/j.joule.2025.102265There is no corresponding record for this reference.
- 53Su, H.; Li, Z.; Zhang, J.; Zhu, Z.; Wang, L.; Qi, T. Recovery of lithium from salt lake brine using a mixed ternary solvent extraction system consisting of TBP, FeCl3, and P507. Hydrometallurgy 2020, 197, 105487, DOI: 10.1016/j.hydromet.2020.105487There is no corresponding record for this reference.
- 54Zhang, L.; Li, J.; Liu, R.; Zhou, Y.; Zhang, Y.; Ji, L.; Li, L. Recovery of lithium from salt lake brine with high Na/Li ratio using solvent extraction. J. Mol. Liq. 2022, 362, 119667, DOI: 10.1016/j.molliq.2022.119667There is no corresponding record for this reference.
- 55Wang, L.; Zhang, J.; Meng, S.; Wu, H.; Zhao, Y.; Meng, Z.; Feng, L.; Wang, H.; Cheng, Y.; Yang, L.; Liu, H. Recovery of lithium from high Mg2+/Li+ ratio brine using a novel solvent extraction system TOP-FeCl3-MIBK. Desalination 2025, 599, 118445, DOI: 10.1016/j.desal.2024.118445There is no corresponding record for this reference.
- 56Bao, Y.; Ji, Z. Y.; Zhou, H.; Zhang, C.; Song, S.; Jia, F.; Li, J.; Quintana, M. Li-ion pre-intercalation strategy of manganese oxides for capacitive deionization-based selective lithium extraction from low-grade brine. Small 2024, 2406951, DOI: 10.1002/smll.202406951There is no corresponding record for this reference.
- 57Dhanushkotti, R.; Mohammed, A. K.; Ranjeesh, K. C.; Krishnegowda, H. M.; Aldaqqa, N. M.; Shetty, D. Inherited nitrogen distribution control in covalent organic framework cathodes for efficient electrochemical lithium recovery via capacitive deionization. Adv. Sci. 2025, 12, 2417140, DOI: 10.1002/advs.202417140There is no corresponding record for this reference.
- 58Liao, G.; Yu, L.; Xia, Y.; Wang, Z.; Lu, Z.; Mei, J.; Liu, H.; Liu, C. High selectivity, capacity, and stability for electrochemical lithium extraction on boron-doped H1.6Mn1.6O4 by tailoring lattice constant and intercalation energy. Water Res. 2025, 274, 123131, DOI: 10.1016/j.watres.2025.123131There is no corresponding record for this reference.
- 59Xu, Y.; Wu, W.; Yi, Q.; Zhang, H.; Lu, Q.; Zhang, K.; Pang, T.; Song, J.; Zhang, D. LiMn2O4 nanoparticles in situ embedded in carbon networks for lithium extraction from brine via hybrid capacitive deionization. ACS Appl. Mater. Interfaces 2025, 17, 4821– 4831, DOI: 10.1021/acsami.4c16382There is no corresponding record for this reference.
- 60Li, P.; Chong, X.; Guo, Y.; Wu, G.; Wang, S.; Liu, R.; Ma, J.; Wang, Y.; Chen, W.; Cai, Y.; Wang, Y.; Yuan, Q.; Zhao, J. Heterostructure-engineered conductive MOFs coupled with MoSe2@MXene framework for efficient and selective lithium extraction from salt lake brine via capacitive deionization. Adv. Sci. 2026, 13, e21830 DOI: 10.1002/advs.202521830There is no corresponding record for this reference.
- 61Zhang, L.; Huang, Z.; Chen, Y.; Li, F.; Li, G.; Zhang, F. Highly selective lithium-ion separation by regulating ion transport energy barriers of vermiculite membranes. ACS Nano 2026, 20, 1345– 1356, DOI: 10.1021/acsnano.5c17718There is no corresponding record for this reference.
- 62Li, K.; Wei, X.; Wang, W.; Zhang, Q.; Ding, Y.; Yang, Q. Optimization of ion migration during lithium-ion recovery through electrodialysis by numerical simulations. Asia-Pac. J. Chem. Eng. 2026, 21, e70125 DOI: 10.1002/apj.70125There is no corresponding record for this reference.
- 63Oladipo, A. A.; Ahmad, M. Energy-efficient ion recovery from water using electro-driven membranes: A comprehensive critical review. Water 2025, 17, 2456, DOI: 10.3390/w17162456There is no corresponding record for this reference.
- 64Zhao, Z.; Yang, J.; Kong, D.; Peng, Y.; Jin, D. Effects of operational parameters on Mg2+/Li+ separation performance in electrodialysis system. Membranes 2025, 15, 260, DOI: 10.3390/membranes15090260There is no corresponding record for this reference.
- 65González, A.; Choque, G.; Grágeda, M.; Ushak, S. Membrane-assisted electrochemical removal of Mg2+ and Ca2+ from lithium brines: Effects of temperature and current density through a zeta potential approach. Membranes 2025, 15, 250, DOI: 10.3390/membranes15090250There is no corresponding record for this reference.
- 66Aljarrah, S.; Alsabbagh, A.; Almahasneh, M. Selective recovery of lithium from Dead Sea end brines using UBK10 ion exchange resin. Can. J. Chem. Eng. 2023, 101, 1185– 1194, DOI: 10.1002/cjce.24559There is no corresponding record for this reference.
- 67Khalil, A.; Mohammed, S.; Hashaikeh, R.; Hilal, N. Lithium recovery from brine: Recent developments and challenges. Desalination 2022, 528, 115611, DOI: 10.1016/j.desal.2022.115611There is no corresponding record for this reference.
- 68An, J. W.; Kang, D. J.; Tran, K. T.; Kim, M. J.; Lim, T.; Tran, T. Recovery of lithium from Uyuni salar brine. Hydrometallurgy 2012, 117–118, 64– 70, DOI: 10.1016/j.hydromet.2012.02.008There is no corresponding record for this reference.
- 69Dave, R. H.; Ghosh, P. K. Enrichment of bromine in sea-bittern with recovery of other marine chemicals. Ind. Eng. Chem. Res. 2005, 44, 2903– 2907, DOI: 10.1021/ie049130xThere is no corresponding record for this reference.
- 70Bertau, M.; Martin, G.; Bertau, M. Integrated direct carbonation process for lithium recovery from primary and secondary resources. Mater. Sci. Forum 2019, 959, 69– 73, DOI: 10.4028/www.scientific.net/MSF.959.69There is no corresponding record for this reference.
- 71Xu, Z.; Zhang, H.; Wang, R.; Gui, W.; Liu, G.; Yang, Y. Systemic and direct production of battery-grade lithium carbonate from a saline lake. Ind. Eng. Chem. Res. 2014, 53, 16502– 16507, DOI: 10.1021/ie502749nThere is no corresponding record for this reference.
- 72Shi, L.; Peng, Z.; Ning, P.; Sun, X.; Li, K.; Zhang, H.; Qu, T. Clean and efficient recovery of lithium from Al-Li alloys via vacuum fractional condensation. Separations 2023, 10, 374, DOI: 10.3390/separations10070374There is no corresponding record for this reference.
- 73Li, Y. H.; Zhao, Z. W.; Liu, X. H.; Chen, X. Y.; Zhong, M. L. Extraction of lithium from salt lake brine by aluminum-based alloys. Trans. Nonferrous Met. Soc. China 2015, 25, 3484– 3489, DOI: 10.1016/S1003-6326(15)64032-8There is no corresponding record for this reference.
- 74Zhang, L.; Zhang, T.; Zhao, Y.; Dong, G.; Lv, S.; Ma, S.; Song, S.; Quintana, M. Doping engineering of lithium-aluminum layered double hydroxides for high-efficiency lithium extraction from salt lake brines. Nano Res. 2024, 17, 1646– 1654, DOI: 10.1007/s12274-023-5950-1There is no corresponding record for this reference.
- 75Yong, M.; Tang, M.; Sun, L.; Xiong, F.; Xie, L.; Zeng, G.; Ren, X.; Wang, K.; Cheng, Y.; Li, Z.; Li, E.; Zhang, X.; Wang, H. Sustainable lithium extraction and magnesium hydroxide co-production from salt-lake brines. Nat. Sustain. 2024, 7, 1662– 1671, DOI: 10.1038/s41893-024-01435-2There is no corresponding record for this reference.
- 76Jiang, H.; Zhang, S.; Yang, Y.; Yu, J. Synergic and competitive adsorption of Li-Na-MgCl2 onto lithium-aluminum hydroxides. Adsorption 2020, 26, 1039– 1049, DOI: 10.1007/s10450-020-00208-5There is no corresponding record for this reference.
- 77Hamzaoui, A. H.; M’nif, A.; Hammi, H.; Rokbani, R. Contribution to the lithium recovery from brine. Desalination 2003, 158, 221– 224, DOI: 10.1016/S0011-9164(03)00455-7There is no corresponding record for this reference.
- 78Tran, K. T.; Van Luong, T.; An, J. W.; Kang, D. J.; Kim, M. J.; Tran, T. Recovery of magnesium from Uyuni salar brine as high-purity magnesium oxalate. Hydrometallurgy 2013, 138, 93– 99, DOI: 10.1016/j.hydromet.2013.05.013There is no corresponding record for this reference.
- 79Wang, H.; Zhong, Y.; Du, B.; Zhao, Y.; Wang, M. Recovery of both magnesium and lithium from high Mg/Li ratio brines using a novel process. Hydrometallurgy 2018, 175, 102– 108, DOI: 10.1016/j.hydromet.2017.10.017There is no corresponding record for this reference.
- 80Lai, X.; Xiong, P.; Zhong, H. Extraction of lithium from brines with high Mg/Li ratio by the crystallization-precipitation method. Hydrometallurgy 2020, 192, 105252, DOI: 10.1016/j.hydromet.2020.105252There is no corresponding record for this reference.
- 81Zhang, Y.; Hu, Y.; Sun, N.; Khoso, S. A.; Wang, L.; Sun, W. A novel precipitant for separating lithium from magnesium in high Mg/Li ratio brine. Hydrometallurgy 2019, 187, 125– 133, DOI: 10.1016/j.hydromet.2019.05.019There is no corresponding record for this reference.
- 82Xiang, W.; Liang, S.; Zhou, Z.; Qin, W.; Fei, W. Extraction of lithium from salt lake brine containing borate anion and high concentration of magnesium. Hydrometallurgy 2016, 166, 9– 15, DOI: 10.1016/j.hydromet.2016.08.005There is no corresponding record for this reference.
- 83Liu, D.; Li, Z.; He, L.; Zhao, Z. Facet-engineered Li3PO4 for lithium recovery from brines. Desalination 2021, 514, 115186, DOI: 10.1016/j.desal.2021.115186There is no corresponding record for this reference.
- 84Zhang, H.; Zhang, S. S.; Zhang, W.; Ma, W. C.; Li, Y. P.; Chen, L.; Zhu, L.; Pan, Y. Recovering phosphorus and lithium separately from wastewater and brine using a novel coupled biofilm-precipitation system. J. Water Process Eng. 2023, 55, 104097, DOI: 10.1016/j.jwpe.2023.104097There is no corresponding record for this reference.
- 85Song, Y.; Zhao, T.; He, L.; Zhao, Z.; Liu, X. A promising approach for directly extracting lithium from α-spodumene by alkaline digestion and precipitation as phosphate. Hydrometallurgy 2019, 189, 105141, DOI: 10.1016/j.hydromet.2019.105141There is no corresponding record for this reference.
- 86Quintero, C.; Dahlkamp, J. M.; Fierro, F.; Thennis, T.; Zhang, Y.; Videla, Á.; Rojas, R. Development of a co-precipitation process for the preparation of magnesium hydroxide containing lithium carbonate from Li-enriched brines. Hydrometallurgy 2020, 198, 105515, DOI: 10.1016/j.hydromet.2020.105515There is no corresponding record for this reference.
- 87Alsabbagh, A.; Aljarrah, S.; Almahasneh, M. Lithium enrichment optimization from Dead Sea end brine by chemical precipitation technique. Miner. Eng. 2021, 170, 107038, DOI: 10.1016/j.mineng.2021.107038There is no corresponding record for this reference.
- 88Cipollina, A.; Winter, D.; Battaglia, G.; Berkemeyer, L.; Cortina, J. L.; de Labastida, M. F.; Rodriguez, J. L. Recovery of lithium carbonate from dilute Li-rich brine via homogeneous and heterogeneous precipitation. Ind. Eng. Chem. Res. 2022, 61, 13589– 13602, DOI: 10.1021/acs.iecr.2c01397There is no corresponding record for this reference.
- 89Kim, H. S.; Suh, Y. J.; Kim, D. W.; Yoo, H.; Hong, H. J.; Park, I. S. Direct aluminate precipitation technique with a phase transformation strategy for selective lithium extraction from Uyuni brine. Chem. Eng. J. 2025, 505, 159220, DOI: 10.1016/j.cej.2025.159220There is no corresponding record for this reference.
- 90Ma, C.; Zhang, J.; Liu, Y.; Sun, L.; Liu, Q. Integrated separation of precipitation and three-liquid-phase extraction for boron, lithium, and magnesium from salt lake brine with high magnesium-lithium ratio. Sep. Purif. Technol. 2024, 347, 127541, DOI: 10.1016/j.seppur.2024.127541There is no corresponding record for this reference.
- 91Zhao, X.; Li, G.; Feng, M.; Wang, Y. Semi-continuous electrochemical extraction of lithium from brine using CF-NMMO/AC asymmetric hybrid capacitors. Electrochim. Acta 2020, 331, 135285, DOI: 10.1016/j.electacta.2019.135285There is no corresponding record for this reference.
- 92Song, Y.; Zhao, Z. Recovery of lithium from spent lithium-ion batteries using precipitation and electrodialysis techniques. Sep. Purif. Technol. 2018, 206, 335– 342, DOI: 10.1016/j.seppur.2018.06.022There is no corresponding record for this reference.
- 93Guo, Z. Y.; Ji, Z. Y.; Chen, Q. B.; Liu, J.; Zhao, Y. Y.; Li, F.; Liu, Z. Y.; Yuan, J. S. Prefractionation of LiCl from concentrated seawater/salt lake brines by electrodialysis with monovalent selective ion-exchange membranes. J. Clean. Prod. 2018, 193, 338– 350, DOI: 10.1016/j.jclepro.2018.05.077There is no corresponding record for this reference.
- 94Zhao, X.; Yang, H.; Wang, Y.; Sha, Z. Review on the electrochemical extraction of lithium from seawater/brine. J. Electroanal. Chem. 2019, 850, 113389, DOI: 10.1016/j.jelechem.2019.113389There is no corresponding record for this reference.
- 95Yin, R.; Zhao, Z.; Xu, W.; He, L. LiMn2O4 submicronization: Shortened Li+ diffusion pathways for enhanced electrochemical lithium extraction and cycle performance. Sep. Purif. Technol. 2025, 359, 130394, DOI: 10.1016/j.seppur.2024.130394There is no corresponding record for this reference.
- 96Xu, W.; He, L.; Zhao, Z. Lithium extraction from high Mg/Li brine via electrochemical intercalation/de-intercalation using LiMn2O4 materials. Desalination 2021, 503, 114935, DOI: 10.1016/j.desal.2021.114935There is no corresponding record for this reference.
- 97Zhu, W.; Xu, W.; Liu, D.; He, L.; Liu, X.; Zhao, Z. Ionic transport kinetics of selective electrochemical lithium extraction from brines. Electrochim. Acta 2024, 475, 143519, DOI: 10.1016/j.electacta.2023.143519There is no corresponding record for this reference.
- 98Xiong, J.; He, L.; Liu, D.; Xu, W.; Zhao, Z. Olivine-FePO4 preparation for lithium extraction from brines via electrochemical de-intercalation/intercalation. Desalination 2021, 520, 115326, DOI: 10.1016/j.desal.2021.115326There is no corresponding record for this reference.
- 99Parsa, N.; Moheb, A.; Mehrabani-Zeinabad, M. A.; Masigol, M. A. Recovery of lithium ions from sodium-contaminated lithium bromide solution using electrodialysis. Chem. Eng. Res. Des. 2015, 98, 81– 88, DOI: 10.1016/j.cherd.2015.03.025There is no corresponding record for this reference.
- 100Strathmann, H. Electrodialysis, a mature technology with a multitude of new applications. Desalination 2010, 264, 268– 288, DOI: 10.1016/j.desal.2010.04.069There is no corresponding record for this reference.
- 101Ji, Z. Y.; Chen, Q. B.; Yuan, J. S.; Liu, J.; Zhao, Y. Y.; Feng, W. X. Preliminary study on recovering lithium from high Mg2+/Li+ ratio brines by electrodialysis. Sep. Purif. Technol. 2017, 172, 168– 177, DOI: 10.1016/j.seppur.2016.08.006There is no corresponding record for this reference.
- 102Jiang, D.; Xu, R.; Bai, L.; Wu, W.; Luo, D.; Li, Z.; Asahi, T.; Mai, Y.; Liu, Z.; Yamauchi, Y.; Xu, X. Insights into electrochemical paradigms for lithium extraction: Electrodialysis versus capacitive deionization. Coord. Chem. Rev. 2024, 516, 215923, DOI: 10.1016/j.ccr.2024.215923There is no corresponding record for this reference.
- 103Ying, J.; Luo, M.; Jin, Y.; Yu, J. Selective separation of lithium from high Mg/Li ratio brine using single-stage and multi-stage selective electrodialysis. Desalination 2020, 492, 114621, DOI: 10.1016/j.desal.2020.114621There is no corresponding record for this reference.
- 104Morita, K.; Matsumoto, T.; Hoshino, T. Efficient lithium extraction via electrodialysis using acid-processed lithium-adsorbing lithium lanthanum titanate. Desalination 2022, 543, 116117, DOI: 10.1016/j.desal.2022.116117There is no corresponding record for this reference.
- 105Wang, W.; Zhang, Y.; Li, F.; Chen, Y.; Mojallali Rostami, S. M.; Hosseini, S. S.; Shao, L. Designing an energy-efficient multi-stage selective electrodialysis process based on high-performance materials for lithium extraction. J. Membr. Sci. 2023, 675, 121534, DOI: 10.1016/j.memsci.2023.121534There is no corresponding record for this reference.
- 106Zhang, X. C.; Wang, J.; Ji, Z. Y.; Ji, P. Y.; Liu, J.; Zhao, Y. Y.; Li, F.; Yuan, J. S. Preparation of Li2CO3 from high Mg2+/Li+ brines based on selective electrodialysis with feed-and-bleed mode. J. Environ. Chem. Eng. 2021, 9, 106635, DOI: 10.1016/j.jece.2021.106635There is no corresponding record for this reference.
- 107Han, J.; Wang, K.; Chen, S.; Lu, H.; Zhang, Z.; Li, Z.; Zhou, L.; Du, Y.; Meng, Q.; Lewis, A.; Huang, Y. Lithium aluminum titanium phosphate-based ceramic composite membrane for selective lithium-ion separation via scalable electrodialysis. Desalination 2024, 591, 118010, DOI: 10.1016/j.desal.2024.118010There is no corresponding record for this reference.
- 108Ma, W.; Han, G.; Li, J.; An, X.; Gao, F.; Du, X.; Li, J.; Liu, Z.; Guan, G.; Hao, X. Hierarchical electroactive ion-permselective membrane with electrochemical switched ion-pump effect for continuous lithium recovery. J. Membr. Sci. 2024, 700, 122719, DOI: 10.1016/j.memsci.2024.122719There is no corresponding record for this reference.
- 109Zhang, H.; Huang, Z.; Zhao, L.; Guo, Z.; Wang, J.; Liu, J.; Zhao, Y.; Li, F.; Zhang, P.; Ji, Z. Y. Fast and stable lithium extraction enabled by less-defective graphene-supported LiMn2O4 networks in hybrid capacitive deionization. Chem. Eng. J. 2024, 482, 148802, DOI: 10.1016/j.cej.2024.148802There is no corresponding record for this reference.
- 110Tian, G.; Gao, J.; Yan, S.; Wang, H.; Li, R.; Xiang, J.; Du, W.; Zhang, L.; Tang, N. Long-acting electrochemical lithium recovery from original brine using super-stable spinel lithium manganate oxide. Desalination 2024, 592, 118103, DOI: 10.1016/j.desal.2024.118103There is no corresponding record for this reference.
- 111Meng, X.; Jing, Y.; Li, J.; Sun, Z.; Wu, Z. Electrochemical recovery of lithium from brine by highly stable truncated octahedral LiNi0.05Mn1.95O4. Chem. Eng. Sci. 2024, 283, 119400, DOI: 10.1016/j.ces.2023.119400There is no corresponding record for this reference.
- 112Gu, J.; Zhou, G.; Chen, L.; Li, X.; Luo, G.; Fan, L.; Chao, Y.; Ji, H.; Zhu, W. Particle size control and electrochemical lithium extraction performance of LiMn2O4. J. Electroanal. Chem. 2023, 940, 117487, DOI: 10.1016/j.jelechem.2023.117487There is no corresponding record for this reference.
- 113Tian, G.; Gao, J.; Wang, M.; Wen, X.; Liu, Y.; Xiang, J.; Zhang, L.; Cheng, P.; Zhang, J.; Tang, N. Structural stabilization of Cr-doped spinel LiMn2O4 for long-term cyclability in electrochemical lithium recovery. Electrochim. Acta 2024, 475, 143361, DOI: 10.1016/j.electacta.2023.143361There is no corresponding record for this reference.
- 114Liu, C.; Li, Y.; Lin, D.; Hsu, P. C.; Liu, B.; Yan, G.; Wu, T.; Cui, Y.; Chu, S. Lithium extraction from seawater through pulsed electrochemical intercalation. Joule 2020, 4, 1459– 1469, DOI: 10.1016/j.joule.2020.05.017There is no corresponding record for this reference.
- 115Zhang, J.; Su, W.; Yi, B.; Guo, Y.; Deng, T.; Yu, X. A new strategy for preparing highly stable and high-capacity electrodes for green electrochemical lithium extraction. Chem. Eng. J. 2023, 454, 140416, DOI: 10.1016/j.cej.2022.140416There is no corresponding record for this reference.
- 116Fu, H.; Zhu, W.; Xu, W.; Zhao, Z. Understanding ion intercalation of LiMn2O4 in brine for electrochemical detection of Li+. J. Electroanal. Chem. 2025, 983, 119033, DOI: 10.1016/j.jelechem.2025.119033There is no corresponding record for this reference.
- 117Hu, B.; Wang, Y.; Zhang, B.; Song, X.; Jiang, H.; Ma, J.; Liu, J. GO-encapsulated La-doped lithium manganese oxide assemblies for enhanced lithium extraction in capacitive deionization. Sep. Purif. Technol. 2024, 348, 127693, DOI: 10.1016/j.seppur.2024.127693There is no corresponding record for this reference.
- 118Siekierka, A.; Bryjak, M. Novel anion exchange membrane for lithium salt concentration in hybrid capacitive deionization. Desalination 2019, 452, 279– 289, DOI: 10.1016/j.desal.2018.10.009There is no corresponding record for this reference.
- 119Rethinasabapathy, M.; Bhaskaran, G.; Hwang, S. K.; Ryu, T.; Huh, Y. S. Efficient lithium extraction using redox-active Prussian blue nanoparticle-anchored activated carbon electrodes via membrane capacitive deionization. Chemosphere 2023, 336, 139256, DOI: 10.1016/j.chemosphere.2023.139256There is no corresponding record for this reference.
- 120Faheem, M.; Alam, R.; Alhajaj, A.; Zou, L. Recovery of lithium by pseudocapacitive electrodes in capacitive deionization. Electrochim. Acta 2024, 489, 144267, DOI: 10.1016/j.electacta.2024.144267There is no corresponding record for this reference.
- 121Siekierka, A. Lithium iron manganese oxide as an adsorbent for capturing lithium ions in hybrid capacitive deionization under different electrical modes. Sep. Purif. Technol. 2020, 236, 116234, DOI: 10.1016/j.seppur.2019.116234There is no corresponding record for this reference.
- 122Ying, J.; Luo, M.; Jin, Y.; Yu, J. Selective separation of lithium from high Mg/Li ratio brine using single-stage and multi-stage selective electrodialysis processes. Desalination 2020, 492, 114621, DOI: 10.1016/j.desal.2020.114621There is no corresponding record for this reference.
- 123Zhao, Y.; Wu, M.; Shen, P.; Uytterhoeven, C.; Mamrol, N.; Shen, J.; Gao, C.; Van der Bruggen, B. Composite anti-scaling membrane with interpenetrating nanofiber networks for selective lithium separation. J. Membr. Sci. 2021, 618, 118668, DOI: 10.1016/j.memsci.2020.118668There is no corresponding record for this reference.
- 124Liu, G.; Zhao, Z.; He, L. Highly selective lithium recovery from high Mg/Li ratio brines. Desalination 2020, 474, 114185, DOI: 10.1016/j.desal.2019.114185There is no corresponding record for this reference.
- 125Sharma, P. P.; Yadav, V.; Rajput, A.; Gupta, H.; Saravaia, H.; Kulshrestha, V. Sulfonated poly(ether ether ketone) composite cation-exchange membrane for selective lithium recovery by electrodialysis. Desalination 2020, 496, 114755, DOI: 10.1016/j.desal.2020.114755There is no corresponding record for this reference.
- 126Wang, W.; Zhang, Y.; Li, F.; Chen, Y.; Mojallali Rostami, S. M.; Hosseini, S. S.; Shao, L. Mussel-inspired polyphenol/polyethyleneimine-assembled membranes with highly positive surface charge for unprecedented cation permselectivity. J. Membr. Sci. 2022, 658, 120703, DOI: 10.1016/j.memsci.2022.120703There is no corresponding record for this reference.
- 127Flexer, V.; Baspineiro, C. F.; Galli, C. I. Lithium recovery from brines: A vital raw material for green energies with potential environmental impacts. Sci. Total Environ. 2018, 639, 1188– 1204, DOI: 10.1016/j.scitotenv.2018.05.223There is no corresponding record for this reference.
- 128Seip, A.; Safari, S.; Pickup, D. M.; Chadwick, A. V.; Ramos, S.; Velasco, C. A.; Cerrato, J. M.; Alessi, D. S. Lithium recovery from hydraulic fracturing flowback and produced water using selective ion-exchange sorbents. Chem. Eng. J. 2021, 426, 130713, DOI: 10.1016/j.cej.2021.130713There is no corresponding record for this reference.
- 129Meng, F.; McNeice, J.; Zadeh, S. S.; Ghahreman, A. Review of lithium production and recovery from minerals, brines, and lithium-ion batteries. Miner. Process. Extr. Metall. Rev. 2021, 42, 123– 141, DOI: 10.1080/08827508.2019.1668387There is no corresponding record for this reference.
- 130Sheng, B.; Su, H.; Yu, J.; Lin, S. Lithium extraction from low-grade Na+/K+ brines using high layer-charge layered double hydroxides. Desalination 2023, 565, 116856, DOI: 10.1016/j.desal.2023.116856There is no corresponding record for this reference.
- 131Gao, A.; Sun, Z.; Li, S.; Hou, X.; Li, H.; Wang, C.; Wu, W. Self-assembled layered lithium manganese oxide with ultrahigh adsorption capacity and selectivity for lithium. Chem. Eng. J. 2023, 471, 144287, DOI: 10.1016/j.cej.2023.144287There is no corresponding record for this reference.
- 132Ighalo, J. O.; Amaku, J. F.; Olisah, C.; Adeola, A. O.; Iwuozor, K. G.; Akpomie, K. G.; Conradie, J.; Adegoke, K. A.; Oyedotun, K. O. Utilization of adsorption as a resource recovery technique for lithium in geothermal water. J. Mol. Liq. 2022, 365, 120107, DOI: 10.1016/j.molliq.2022.120107There is no corresponding record for this reference.
- 133Safari, S.; Lottermoser, B. G.; Alessi, D. S. Metal oxide sorbents for sustainable lithium recovery from unconventional resources. Appl. Mater. Today 2020, 19, 100638, DOI: 10.1016/j.apmt.2020.100638There is no corresponding record for this reference.
- 134Arroyo, F.; Morillo, J.; Usero, J.; Rosado, D.; El Bakouri, H. Lithium recovery from desalination brines using specific ion-exchange resins. Desalination 2019, 468, 114073, DOI: 10.1016/j.desal.2019.114073There is no corresponding record for this reference.
- 135Huang, Y.; Li, X.; Chen, L.; Luo, G.; Tao, D.; Sun, J.; Qiu, Z.; Chao, Y.; Zhu, W. Synthesis of magnetic crown-ether ion-imprinted polymer for selective lithium adsorption. New J. Chem. 2023, 47, 3134– 3139, DOI: 10.1039/D2NJ05377BThere is no corresponding record for this reference.
- 136Baudino, L.; Santos, C.; Pirri, C. F.; La Mantia, F.; Lamberti, A. Recent advances in lithium recovery from water resources: From passive to electrochemical methods. Adv. Sci. 2022, 9, 2201380, DOI: 10.1002/advs.202201380There is no corresponding record for this reference.
- 137Weng, D.; Duan, H.; Hou, Y.; Huo, J.; Chen, L.; Zhang, F.; Wang, J. Introduction of manganese-based lithium-ion sieves: A review. Prog. Nat. Sci.: Mater. Int. 2020, 30, 139– 152, DOI: 10.1016/j.pnsc.2020.01.017There is no corresponding record for this reference.
- 138Yu, H.; Naidu, G.; Zhang, C.; Wang, C.; Razmjou, A.; Han, D. S.; Shon, H. Metal-based adsorbents for lithium recovery from aqueous resources. Desalination 2022, 539, 115951, DOI: 10.1016/j.desal.2022.115951There is no corresponding record for this reference.
- 139Zhang, Y.; Liu, J.; Yang, Y.; Lin, S.; Li, P. Preparation of granular titanium-type lithium-ion sieves and recyclability assessment for lithium recovery from brines with varying pH. Sep. Purif. Technol. 2021, 267, 118613, DOI: 10.1016/j.seppur.2021.118613There is no corresponding record for this reference.
- 140Borghols, W. J. H.; Wagemaker, M.; Lafont, U.; Kelder, E. M.; Mulder, F. M. Size effects in the Li4+xTi5O12 spinel. J. Am. Chem. Soc. 2009, 131, 17786– 17792, DOI: 10.1021/ja902423eThere is no corresponding record for this reference.
- 141Zhao, B.; Qiao, Y.; Qian, Z.; Wei, W.; Li, J.; Wu, Z.; Liu, Z. Unraveling Li+ desorption behavior and mechanism of Li4Ti5O12 with different facets to enhance lithium extraction. J. Mater. Chem. A 2023, 11, 7094– 7104, DOI: 10.1039/D2TA10016AThere is no corresponding record for this reference.
- 142Du, Y. E.; Niu, X.; Li, W.; An, J.; Liu, Y.; Chen, Y.; Wang, P.; Yang, X.; Feng, Q. Microwave-assisted synthesis of high-energy faceted TiO2 nanocrystals derived from exfoliated porous metatitanic acid nanosheets. Materials 2019, 12, 3614, DOI: 10.3390/ma12213614There is no corresponding record for this reference.
- 143Wang, Q.; Yang, X.; Li, C.; Zhao, C.; Huang, Y.; Qiu, S.; Qin, Y.; Shi, C. Titanium-based lithium-ion sieve H2TiO3 with enhanced Li+ adsorption by magnetic Fe doping. Sep. Purif. Technol. 2024, 346, 127455, DOI: 10.1016/j.seppur.2024.127455There is no corresponding record for this reference.
- 144Tu, Y.; Zhou, Z.; Wei, W.; Guan, L.; Liu, Y. X.; Xu, Z.; Liu, H.; Liu, Z. Ti surface-doped spinel lithium manganese oxide with porous structure and improved stability for lithium extraction from salt-lake brine. Chem. Eng. J. 2025, 503, 158533, DOI: 10.1016/j.cej.2024.158533There is no corresponding record for this reference.
- 145Ma, W.; Huang, H.; Zhu, G.; Wang, X.; Kong, Q.; Shi, X. Fabrication of spinel-type H4Ti5O12 ion sieve for lithium recovery from aqueous resources: Adsorption performance and mechanism. Processes 2025, 13, 2981, DOI: 10.3390/pr13092981There is no corresponding record for this reference.
- 146US4159311A Patent. Recovery of lithium from brines, 1979.There is no corresponding record for this reference.
- 147Besserguenev, A. V.; Fogg, A. M.; Francis, R. J.; Price, S. J.; O’Hare, D.; Isupov, V. P.; Tolochko, B. P. Synthesis and structure of gibbsite intercalation compounds [LiAl2(OH)6]X (X = Cl, Br, NO3). Chem. Mater. 1997, 9, 241– 247, DOI: 10.1021/cm960316zThere is no corresponding record for this reference.
- 148Zhong, J.; Lin, S.; Yu, J. Li+ adsorption performance and mechanism of lithium-aluminum layered double hydroxides in low-grade brines. Desalination 2021, 505, 114983, DOI: 10.1016/j.desal.2021.114983There is no corresponding record for this reference.
- 149Paranthaman, M. P.; Li, L.; Luo, J.; Hoke, T.; Ucar, H.; Moyer, B. A.; Harrison, S. Recovery of lithium from geothermal brine with lithium-aluminum layered double hydroxide chloride sorbents. Environ. Sci. Technol. 2017, 51, 13481– 13486, DOI: 10.1021/acs.est.7b03464There is no corresponding record for this reference.
- 150Zhang, L.; Zhang, T.; Lv, S.; Cheng, S.; Dong, G.; Quintana, M.; Song, S.; Zhao, Y. Steering interlayer interactions of lithium-aluminum layered double hydroxide beads for stable lithium extraction from sulfate-type brines. Desalination 2024, 592, 118130, DOI: 10.1016/j.desal.2024.118130There is no corresponding record for this reference.
- 151Gao, J.; Hong, M.; Zhong, J.; Na, S.; Su, Y.; Li, H.; Hu, M.; Yang, S. Semi-sacrificial fabrication of binder-free lithium-aluminum layered double hydroxide adsorbents for lithium extraction. Desalination 2025, 597, 118409, DOI: 10.1016/j.desal.2024.118409There is no corresponding record for this reference.
- 152Wu, Y. H.; Guo, W.; Sun, Y. Z.; Sun, S. Y. Resin functionalized with 2-(hydroxymethyl)-12-crown-4 ether for selective adsorption of lithium ions from desilication solution of coal-based solid waste. Sep. Purif. Technol. 2025, 355, 129500, DOI: 10.1016/j.seppur.2024.129500There is no corresponding record for this reference.
- 153Zhang, X.; Xing, X.; Zhao, X.; Yang, J.; Liu, M.; Liu, B.; Gao, X.; Huang, H. Crown ethers decorated metal-organic framework for selective lithium-ion sieving. Chem. Eng. J. 2025, 503, 158435, DOI: 10.1016/j.cej.2024.158435There is no corresponding record for this reference.
- 154Liberati, A. F.; Geneste, A.; Prelot, B.; Mericq, J. P.; Faur, C.; Monge, S. Synthesis, characterization, and Li adsorption study of a new thermoresponsive crown-ether-bearing copolymer. Sep. Purif. Technol. 2025, 354, 129123, DOI: 10.1016/j.seppur.2024.129123There is no corresponding record for this reference.
- 155Park, M. J.; Nisola, G. M.; Beltran, A. B.; Torrejos, R. E. C.; Seo, J. G.; Lee, S. P.; Kim, H.; Chung, W. J. Recyclable composite nanofiber adsorbent for Li+ recovery from seawater desalination retentate. Chem. Eng. J. 2014, 254, 73– 81, DOI: 10.1016/j.cej.2014.05.095There is no corresponding record for this reference.
- 156Yu, J.; Zhu, J.; Luo, G.; Chen, L.; Li, X.; Cui, P.; Wu, P.; Chao, Y.; Zhu, W.; Liu, Z. 3D-printed titanium-based ionic sieve monolithic adsorbent for selective lithium recovery from salt lakes. Desalination 2023, 560, 116651, DOI: 10.1016/j.desal.2023.116651There is no corresponding record for this reference.
- 157Ding, Y.; Nhung, N. T. H.; An, J.; Chen, H.; Liao, L.; He, C.; Wang, X.; Fujita, T. Manganese-titanium mixed ion sieves for selective adsorption of lithium ions from artificial salt-lake brine. Materials 2023, 16, 4190, DOI: 10.3390/ma16114190There is no corresponding record for this reference.
- 158Zhao, K.; Tong, B.; Yu, X.; Guo, Y.; Xie, Y.; Deng, T. Synthesis of porous fiber-supported lithium ion-sieve adsorbent for lithium recovery from geothermal water. Chem. Eng. J. 2022, 430, 131423, DOI: 10.1016/j.cej.2021.131423There is no corresponding record for this reference.
- 159Limjuco, L. A.; Nisola, G. M.; Lawagon, C. P.; Lee, S. P.; Seo, J. G.; Kim, H.; Chung, W. J. H2TiO3 composite adsorbent foam for efficient and continuous recovery of Li+ from liquid resources. Colloids Surf., A 2016, 504, 267– 279, DOI: 10.1016/j.colsurfa.2016.05.072There is no corresponding record for this reference.
- 160Hong, H. J.; Park, I. S.; Ryu, T.; Ryu, J.; Kim, B. G.; Chung, K. S. Granulation of Li1.33Mn1.67O4 through cross-linked chitosan for effective recovery of Li+ from seawater. Chem. Eng. J. 2013, 234, 16– 22, DOI: 10.1016/j.cej.2013.08.060There is no corresponding record for this reference.
- 161Qiu, Z.; Wang, M.; Chen, Y.; Zhang, T.; Yang, D.; Qiu, F. Li4Mn5O12-doped cellulose acetate membrane with low Mn loss and high stability for enhanced lithium extraction from seawater. Desalination 2021, 506, 115003, DOI: 10.1016/j.desal.2021.115003There is no corresponding record for this reference.
- 162Tang, L.; Huang, S.; Wang, Y.; Liang, D.; Li, Y.; Li, J.; Wang, Y.; Xie, Y.; Wang, W. Highly efficient, stable, and recyclable hydrogen manganese oxide/cellulose film for lithium extraction from seawater. ACS Appl. Mater. Interfaces 2020, 12, 9775– 9781, DOI: 10.1021/acsami.9b21612There is no corresponding record for this reference.
- 163Chen, S.; Chen, Z.; Wei, Z.; Hu, J.; Guo, Y.; Deng, T. Titanium-based ion sieve with enhanced post-separation ability for high-performance lithium recovery from geothermal water. Chem. Eng. J. 2021, 410, 128320, DOI: 10.1016/j.cej.2020.128320There is no corresponding record for this reference.
- 164Xiao, J. L.; Sun, S. Y.; Song, X.; Li, P.; Yu, J. G. Lithium-ion recovery from brine using granulated polyacrylamide-MnO2 ion sieve. Chem. Eng. J. 2015, 279, 659– 666, DOI: 10.1016/j.cej.2015.05.075There is no corresponding record for this reference.
- 165Zhang, X.; Wu, J.; He, Y.; Zhao, C.; An, M.; Li, L. Porous poly(vinyl alcohol)/polyacrylamide hydrogels loaded with HTO lithium-ion sieves for rapid and efficient Li+ extraction. Desalination 2024, 580, 117587, DOI: 10.1016/j.desal.2024.117587There is no corresponding record for this reference.
- 166Meng, Z.; Wang, M.; Cao, X.; Wang, T.; Wang, Y.; Xu, Y.; Liu, W.; Chen, L.; Huang, Y.; Liu, X. Highly flexible interconnected Li+ ion-sieve porous hydrogels with self-regulating nanonetworks for marine lithium recovery. Chem. Eng. J. 2022, 445, 136780, DOI: 10.1016/j.cej.2022.136780There is no corresponding record for this reference.
- 167Yang, L.; Tu, Y.; Li, H.; Zhan, W.; Hu, H.; Wei, Y.; Chen, C.; Liu, K.; Shao, P.; Li, M.; Yang, G.; Luo, X. Fluorine-rich supramolecular nanocontainer cross-linked hydrogel for lithium extraction with super-high capacity and selectivity. Angew. Chem., Int. Ed. 2023, 62, e202308702 DOI: 10.1002/anie.202308702There is no corresponding record for this reference.
- 168Zhang, E.; Liu, W.; Liang, Q.; Liu, X.; Zhao, Z.; Yang, Y. Selective recovery of Li+ in acidic environments using an electroactive Li+-imprinted graphene-based hybrid aerogel. Chem. Eng. J. 2020, 385, 123948, DOI: 10.1016/j.cej.2019.123948There is no corresponding record for this reference.
- 169Li, J.; Luo, Q.; Dong, M.; Nie, G.; Liu, Z.; Wu, Z. Granulated Li/Al-LDH adsorbents for lithium recovery from synthetic and real salt-lake brines. Hydrometallurgy 2022, 209, 105828, DOI: 10.1016/j.hydromet.2022.105828There is no corresponding record for this reference.
- 170Wang, S.; Zheng, S.; Wang, Z.; Cui, W.; Zhang, H.; Yang, L.; Zhang, Y.; Li, P. Iron-doped lithium ion-sieves with superior lithium adsorption and magnetic separation behavior. Chem. Eng. J. 2018, 332, 160– 168, DOI: 10.1016/j.cej.2017.09.055There is no corresponding record for this reference.
- 171Lin, Y.; Li, M.; Wang, S.; Xue, F.; Ju, S. Biomass carbonaceous aerogel-modified ion-sieve composites for lithium adsorption. Mater. Lett. 2024, 363, 136282, DOI: 10.1016/j.matlet.2024.136282There is no corresponding record for this reference.
- 172Zhong, J.; Lin, S.; Yu, J. Lithium recovery from ultrahigh Mg2+/Li+ ratio brine using granulated Li/Al-LDH adsorbents. Sep. Purif. Technol. 2021, 256, 117780, DOI: 10.1016/j.seppur.2020.117780There is no corresponding record for this reference.
- 173Zhang, Y.; Ray, A. K. Liquid-liquid extraction. In Coulson and Richardson’s Chemical Engineering; Elsevier: Oxford, 2023; Vol. 2B, pp 257– 312. DOI: 10.1016/B978-0-08-101097-6.00004-3 .There is no corresponding record for this reference.
- 174Song, J. F.; Nghiem, L. D.; Li, X. M.; He, T. Lithium extraction from Chinese salt-lake brines: Opportunities, challenges, and future outlook. Environ. Sci. (Camb.) 2017, 3, 593– 597, DOI: 10.1039/C7EW00020KThere is no corresponding record for this reference.
- 175Zhou, Z.; Qin, W.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in three diluents. J. Chem. Eng. Data 2011, 56, 3518– 3522, DOI: 10.1021/je200246xThere is no corresponding record for this reference.
- 176Zhou, Z.; Qin, W.; Liu, Y.; Fei, W. Extraction equilibria of lithium with tributyl phosphate in kerosene and FeCl3. J. Chem. Eng. Data 2012, 57, 82– 86, DOI: 10.1021/je200803hThere is no corresponding record for this reference.
- 177Harvianto, G. R.; Kim, S. H.; Ju, C. S. Solvent extraction and stripping of lithium ion from aqueous solution and application to seawater. Rare Met. 2016, 35, 948– 953, DOI: 10.1007/s12598-015-0453-1There is no corresponding record for this reference.
- 178Ji, L.; Hu, Y.; Li, L.; Shi, D.; Li, J.; Nie, F.; Song, F.; Zeng, Z.; Sun, W.; Liu, Z. Lithium extraction using a synergistic system of dioctyl phthalate and tributyl phosphate in kerosene and FeCl3. Hydrometallurgy 2016, 162, 71– 78, DOI: 10.1016/j.hydromet.2016.02.018There is no corresponding record for this reference.
- 179Su, H.; Li, Z.; Zhang, J.; Liu, W.; Zhu, Z.; Wang, L.; Qi, T. Regulation of Fe3+ coordination in ternary solvent systems for selective lithium extraction and easy stripping. ACS Sustain. Chem. Eng. 2020, 8, 1971– 1979, DOI: 10.1021/acssuschemeng.9b06432There is no corresponding record for this reference.
- 180Su, H.; Li, Z.; Zhang, J.; Zhu, Z.; Wang, L.; Qi, T. Recovery of lithium from salt-lake brine using TBP-FeCl3-P507 mixed ternary solvent extraction. Hydrometallurgy 2020, 197, 105487, DOI: 10.1016/j.hydromet.2020.105487There is no corresponding record for this reference.
- 181Shi, C.; Duan, D.; Jia, Y.; Jing, Y. Highly efficient lithium ion extraction using ionic-liquid-containing tributyl phosphate solvent systems. J. Mol. Liq. 2014, 200, 191– 195, DOI: 10.1016/j.molliq.2014.10.004There is no corresponding record for this reference.
- 182Shi, C.; Jia, Y.; Zhang, C.; Liu, H.; Jing, Y. Extraction of lithium from salt-lake brine using room-temperature ionic liquids in tributyl phosphate. Fusion Eng. Des. 2015, 90, 1– 6, DOI: 10.1016/j.fusengdes.2014.09.021There is no corresponding record for this reference.
- 183Shi, C.; Jing, Y.; Jia, Y. Solvent extraction of lithium ions by tributyl phosphate using room-temperature ionic liquids. J. Mol. Liq. 2016, 215, 640– 646, DOI: 10.1016/j.molliq.2016.01.025There is no corresponding record for this reference.
- 184Shi, C.; Jing, Y.; Xiao, J.; Wang, X.; Yao, Y.; Jia, Y. Solvent extraction of lithium using non-fluorinated functionalized ionic liquids. Sep. Purif. Technol. 2017, 172, 473– 479, DOI: 10.1016/j.seppur.2016.08.034There is no corresponding record for this reference.
- 185Yang, S.; Liu, G.; Wang, J.; Cui, L.; Chen, Y. Recovery of lithium from alkaline brine by solvent extraction with functionalized ionic liquids. Fluid Phase Equilib. 2019, 493, 129– 136, DOI: 10.1016/j.fluid.2019.04.015There is no corresponding record for this reference.
- 186Yusuf, A.; Sodiq, A.; Giwa, A.; Eke, J.; Pikuda, O.; De Luca, G.; Di Salvo, J. L.; Chakraborty, S. Emerging trends in membrane science and technology for sustainable water treatment. J. Clean. Prod. 2020, 266, 121867, DOI: 10.1016/j.jclepro.2020.121867There is no corresponding record for this reference.
- 187Li, X.; Mo, Y.; Qing, W.; Shao, S.; Tang, C. Y.; Li, J. Membrane-based technologies for lithium recovery from aqueous resources: A review. J. Membr. Sci. 2019, 591, 117317, DOI: 10.1016/j.memsci.2019.117317There is no corresponding record for this reference.
- 188Wen, X.; Ma, P.; Zhu, C.; He, Q.; Deng, X. Preliminary study on recovering lithium chloride from lithium-containing waters by nanofiltration. Sep. Purif. Technol. 2006, 49, 230– 236, DOI: 10.1016/j.seppur.2005.10.004There is no corresponding record for this reference.
- 189Sarkar, S.; SenGupta, A. K.; Prakash, P. The Donnan membrane principle: Opportunities for sustainable engineered processes and materials. Environ. Sci. Technol. 2010, 44, 1161– 1166, DOI: 10.1021/es9024029There is no corresponding record for this reference.
- 190Sun, S. Y.; Cai, L. J.; Nie, X. Y.; Song, X.; Yu, J. G. Separation of magnesium and lithium from brine using desal nanofiltration membranes. J. Water Process Eng. 2015, 7, 210– 217, DOI: 10.1016/j.jwpe.2015.06.012There is no corresponding record for this reference.
- 191Li, X.; Zhang, C.; Zhang, S.; Li, J.; He, B.; Cui, Z. Positively charged polyamide composite nanofiltration hollow fiber membranes for lithium and magnesium separation. Desalination 2015, 369, 26– 36, DOI: 10.1016/j.desal.2015.04.027There is no corresponding record for this reference.
- 192Zhang, H. Z.; Xu, Z. L.; Ding, H.; Tang, Y. J. Positively charged capillary nanofiltration membranes with high Mg2+/Ca2+ rejection and Mg2+/Li+ separation. Desalination 2017, 420, 158– 166, DOI: 10.1016/j.desal.2017.07.011There is no corresponding record for this reference.
- 193Wu, H.; Lin, Y.; Feng, W.; Liu, T.; Wang, L.; Yao, H.; Wang, X. Ionic-liquid-based nanofiltration membranes for lithium extraction from high Mg2+/Li+ brines. J. Membr. Sci. 2020, 603, 117997, DOI: 10.1016/j.memsci.2020.117997There is no corresponding record for this reference.
- 194Wu, X.; Liu, H.; Wei, Y.; Fei, Y.; Qi, H. Negatively charged organic-inorganic hybrid silica nanofiltration membranes for lithium extraction. Chin. J. Chem. Eng. 2020, 28, 749– 757, DOI: 10.1016/j.cjche.2019.11.004There is no corresponding record for this reference.
- 195Pramanik, B. K.; Asif, M. B.; Kentish, S.; Nghiem, L. D.; Hai, F. I. Lithium enrichment from simulated salt-lake brine using integrated nanofiltration-membrane distillation. J. Environ. Chem. Eng. 2019, 7, 103395, DOI: 10.1016/j.jece.2019.103395There is no corresponding record for this reference.
- 196Nie, X. Y.; Sun, S. Y.; Sun, Z.; Song, X.; Yu, J. G. Ion fractionation of lithium and magnesium by electrodialysis using monovalent selective ion-exchange membranes. Desalination 2017, 403, 128– 135, DOI: 10.1016/j.desal.2016.05.010There is no corresponding record for this reference.
- 197Chen, Q. B.; Ji, Z. Y.; Liu, J.; Zhao, Y. Y.; Wang, S. Z.; Yuan, J. S. Effect of coexisting cations on lithium migration during selective electrodialysis. J. Membr. Sci. 2018, 548, 408– 420, DOI: 10.1016/j.memsci.2017.11.040There is no corresponding record for this reference.
- 198Jiang, C.; Wang, Y.; Wang, Q.; Feng, H.; Xu, T. Production of lithium hydroxide from lake brines through electro-electrodialysis with bipolar membranes. Ind. Eng. Chem. Res. 2014, 53, 6103– 6112, DOI: 10.1021/ie404334sThere is no corresponding record for this reference.
- 199Yang, D.; Yang, Y.; Wong, T.; Iguodala, S.; Wang, A.; Lovell, L.; Foglia, F.; Fouquet, P.; Breakwell, C.; Fan, Z.; Wang, Y.; Britton, M. M.; Williams, D. R.; Shah, N.; Xu, T.; McKeown, N. B.; Titirici, M.-M.; Jelfs, K. E.; Song, Q. Solution-processable polymer membranes with hydrophilic subnanometer pores for sustainable lithium extraction. Nat. Water 2025, 3, 319– 333, DOI: 10.1038/s44221-025-00398-8There is no corresponding record for this reference.
- 200Martínez-López, J. C.; Santos Rodríguez, M.; Oliver Cuenca, V.; Silva Testa, G.; van Eck, E.; Zhao, E. W.; Lozano, Á. E.; Álvarez, C.; Carretero-González, J. Dibenzodioxin-based polymers of intrinsic microporosity with enhanced lithium-ion transport. Macromolecules 2024, 57, 9442– 9456, DOI: 10.1021/acs.macromol.4c01243There is no corresponding record for this reference.
- 201Gou, F.; Wang, Q.; Yang, Z.; Chang, W.; Shen, J.; Zeng, H. Artificial lithium channels built from polymers of intrinsic microporosity. Angew. Chem., Int. Ed. 2025, 64, e202418304 DOI: 10.1002/anie.202418304There is no corresponding record for this reference.
- 202Lei, D.; Zhu, Y.; Lou, L. L.; Liu, Z. Covalent organic framework membranes for lithium extraction: Facilitated ion-transport strategies to enhance selectivity. Mater. Horiz. 2025, 12, 5459– 5472, DOI: 10.1039/D5MH00457HThere is no corresponding record for this reference.
- 203Meng, W.; Chen, S.; Wu, M.; Gao, F.; Hou, Y.; Zhan, X.; Hu, W.; Liang, L.; Zhang, Q. Dehydration-enhanced ion recognition of triazine covalent organic frameworks for high-resolution Li+/Mg2+ separation. Angew. Chem., Int. Ed. 2025, 64, e202422423 DOI: 10.1002/anie.202422423There is no corresponding record for this reference.
- 204Bao, S.; Ma, Z.; Yu, L.; Li, Q.; Xia, J.; Song, S.; Sui, K.; Zhao, Y.; Liu, X.; Gao, J. Randomly oriented covalent organic framework membranes for selective Li+ sieving. Nat. Commun. 2025, 16, 3896, DOI: 10.1038/s41467-025-59188-1There is no corresponding record for this reference.
- 205Habiba, U.; Siddique, T. A.; Fattah, I. M. R.; Mohammed, S.; Attenborough, E.; Lai, Q.; Wang, Z. Porous alumina-supported lithium aluminum titanium phosphate membrane for lithium extraction via electrodialysis. Sep. Purif. Technol. 2025, 354, 128657, DOI: 10.1016/j.seppur.2024.128657There is no corresponding record for this reference.
- 206Epp, V.; Ma, Q.; Hammer, E. M.; Tietz, F.; Wilkening, M. Very fast bulk lithium-ion diffusivity in crystalline Li1.5Al0.5Ti1.5(PO4)3. Phys. Chem. Chem. Phys. 2015, 17, 32115– 32121, DOI: 10.1039/C5CP05337DThere is no corresponding record for this reference.
- 207Jeong, W.; Liu, L.; Lim, H. S.; Chung, J.; Chen, K.; Ryu, B. K. Improved ion conductivity of lithium aluminum titanium phosphate solid-state electrolytes by divalent cation addition. J. Korean Ceram. Soc. 2025, 62, 83– 89, DOI: 10.1007/s43207-024-00419-4There is no corresponding record for this reference.
- 208Gulied, M.; Zavahir, S.; Elmakki, T.; Park, H.; Gago, G. H.; Shon, H. K.; Han, D. S. Efficient lithium recovery from simulated brine using a hybrid DCMD-ESIX system. Desalination 2024, 572, 117127, DOI: 10.1016/j.desal.2023.117127There is no corresponding record for this reference.
- 209Wan, L.; Zhang, T.; Li, H.; Huang, Q.; Hu, J.; Pan, L. In situ construction of rod-shaped Fe3O4/N-doped carbon architectures for superior lithium extraction via hybrid capacitive deionization. Desalination 2025, 601, 118538, DOI: 10.1016/j.desal.2025.118538There is no corresponding record for this reference.
- 210Zegeye, R. B.; Tufa, R. A.; Santoro, S.; Inzillo, B. M.; Aquino, M.; Politano, G. G.; Embaye, A. S.; Argurio, P.; De Bartolo, L.; Curcio, E. Sustainable lithium recovery from geothermal brine via integrated membrane distillation-selective electrodialysis. Green Chem. 2025, 27, 4205– 4221, DOI: 10.1039/D4GC06530AThere is no corresponding record for this reference.
- 211Siekierka, A. Lithium and magnesium separation from brines by hybrid capacitive deionization. Desalination 2022, 527, 115569, DOI: 10.1016/j.desal.2022.115569There is no corresponding record for this reference.
- 212Baudino, L.; Santos, C.; Pirri, C. F.; La Mantia, F.; Lamberti, A. Recent advances in lithium recovery from water resources: From passive to electrochemical methods. Adv. Sci. 2022, 9, 2201380, DOI: 10.1002/advs.202201380There is no corresponding record for this reference.


