
About the Cover:
Review
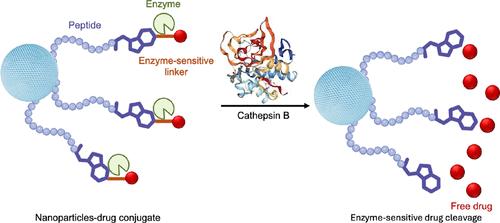
Cathepsin B-Responsive Nanocarriers: Silica and Liposomes for Precision Cancer Therapy
Sumiya Iqbal - ,
Niklas Schlensog - ,
Valerie Tan Shu Li - ,
Shaista Ilyas - , and
Sanjay Mathur *
Enzyme-responsive nanocarriers hold great promises for precision delivery of therapeutics. Silica nanocarriers and liposomes functionalized with protease-cleavable peptide linkers (e.g., cathepsin B-cleavable peptide) have emerged as powerful platforms for selectively releasing therapeutic payloads within tumor microenvironments. This review presents recent advances in cathepsin B-responsive systems, emphasizing design principles such as peptide-linker selection, nanoparticle engineering, and incorporation into antibody-drug conjugates. We also address persisting challenges in clinical translation, including enzyme expression variability and linker stability in order to discuss strategies to overcome these barriers through advanced bioengineering approaches.

Plasmonic Nanotheranostics: Merging Imaging and Therapy on a Unified Platform for Precision Oncology
Yuan-Fong Chou Chau *
Plasmonic nanoplatforms have emerged as a powerful and versatile class of cancer theranostic agents, enabling the seamless integration of diagnostic imaging and therapeutic intervention within a single nanoscale system. By exploiting localized surface plasmon resonance (LSPR), these nanostructures concentrate electromagnetic energy into nanoscale volumes, thereby enhancing optical contrast, improving photothermal conversion efficiency, and enabling plasmon-mediated photochemical processes. This review provides a comprehensive and critical overview of recent advances in plasmonic cancer theranostics, with particular emphasis on the mechanistic foundations, nanostructure design strategies, and system-level integration that enable imaging-guided and feedback-controlled therapy. We discuss how nanoparticle geometry, composition, and surface engineering govern key plasmonic functions, including surface-enhanced Raman scattering (SERS), photoacoustic imaging, computed tomography contrast, and nonlinear optical responses, as well as therapeutic modalities such as photothermal therapy (PTT), photodynamic therapy (PDT), and triggered drug delivery. Special attention is given to synergistic combination therapies—PTT−PDT, PTT−chemotherapy, and plasmonic phototherapy−immunotherapy—in which plasmonic nanoplatforms serve as central integrators that amplify therapeutic efficacy while minimizing systemic toxicity. We further highlight intelligent and tumor-microenvironment-responsive designs that enable real-time monitoring and adaptive treatment within closed-loop theranostic frameworks. Finally, we analyze key translational challenges, including biosafety, biodistribution, scalable manufacturing, regulatory pathways, and clinical integration, and outline emerging opportunities in biodegradable nanoplatforms, AI-assisted design, and personalized oncology. Collectively, this review underscores the transformative potential of plasmonic nanoplatforms to advance precision, image-guided cancer therapy from laboratory innovation toward clinical reality.
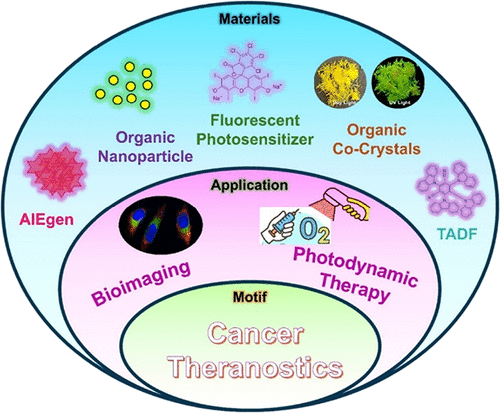
Design and Photophysical Engineering of Functional Organic Luminogens for Precision Cancer Theranostics
Priyam Ghosh - and
Parameswar Krishnan Iyer *
Cancer remains a significant global health challenge, necessitating innovative therapeutic strategies. Functional organic luminogens have emerged as a versatile class of biomaterials for cancer theranostics, enabling the integration of diagnostic imaging and therapeutic intervention within a single molecular or supramolecular platform. In this perspective, recent advances in the rational design of these luminogens as next-generation cancer theranostics were discussed. Particular emphasis is placed on emerging organic luminogen systems, including aggregation-induced emission (AIE) small molecules, thermally activated delayed fluorescence (TADF)-based probes, polymer-based nanostructures, organic co-crystals, and charge-transfer (CT) assemblies, with an emphasis on their structure−property−function relationships. Unlike conventional nanoparticle systems, small-molecule luminogens have defined structures, improved biocompatibility, and faster clearance rates, enabling deeper tumor penetration and reduced long-term toxicity. Key molecular design strategies that regulate excited-state dynamics, aggregation behavior, charge transfer, and microenvironment responsiveness are discussed in the context of near-infrared (NIR) and NIR-II imaging, photodynamic and photothermal therapy, and synergistic multimodal treatments. Finally, challenges related to specificity, biosafety, and translational implementation are outlined, while emerging opportunities like data driven molecular discovery and artificial intelligence-assisted discovery are highlighted as future directions for the development of organic luminogens-based biomaterials in precision cancer theranostics.
Article

Injectable Montmorillonite Nanoclay-Reinforced Hydrogel for Rapid Hemostasis in Noncompressible Torso Hemorrhage
My-An Tran Le - ,
Khoi Minh Le - ,
Thy Nguyen Ngoc Khanh - ,
Thong Vu Lam - ,
Tin Anh Tran - ,
Du Khanh Tran - ,
An Huyen Lieu - ,
Hoan Ngoc Doan - , and
Thi-Hiep Nguyen *
Noncompressible torso hemorrhage (NCTH) presents a significant challenge in trauma care, necessitating innovative hemostatic approaches. This study reports the development of an injectable composite hydrogel incorporating montmorillonite nanoclay, carboxymethyl cellulose, and oxidized xanthan gum cross-linked via acyl hydrazone linkages. The hydrogel demonstrates exceptionally rapid gelation, solidifying within 5 s at the wound site, thereby enabling immediate hemostatic action. Integration of montmorillonite nanoclay significantly enhances both the hemostatic efficacy and mechanical stability of the hydrogel, with a compressive strength of 0.12 MPa. In rat liver injury models, application of this hydrogel reduced hemostasis time by over 80% and blood loss by more than 85% compared to conventional treatments. Furthermore, biocompatibility and biodegradability assessments indicated minimal inflammatory response and effective support for tissue regeneration. The hydrogel’s ability to rapidly form a stable, conformal seal at injury sites highlights its potential as a minimally invasive therapeutic agent for NCTH and other severe bleeding scenarios, as well as a promising scaffold for tissue repair post-hemostasis.
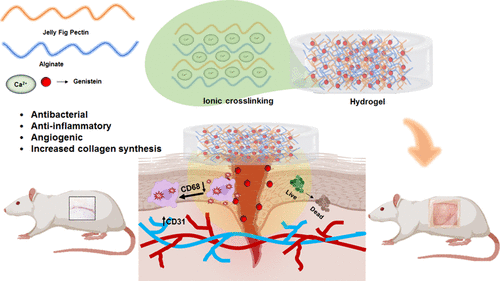
Biomimetic Multifunctional Hydrogels from Jelly Fig Polysaccharide (Ficus awkeotsang Makino), Alginate, and Genistein for Enhanced Diabetic Wound Healing Applications
Ponrasu Thangavel *- ,
Iniyan Saravanakumar - ,
Manoj Kumar Sundaram - ,
Balamurugan Rathinam - , and
Vignesh Muthuvijayan *
The main causes of delayed wound healing in diabetes are bacterial infections, inadequate vasculature, tissue inflammation, and impaired collagen deposition. Here, hydrogels based on jelly fig polysaccharide (JFP) and alginate (Alg) were created using calcium ion crosslinking, which may aid in treating diabetic wounds. Genistein (Gen) was loaded as a bioactive ingredient due to its wound healing, anti-inflammatory, and antioxidant capabilities. The physicochemical characterization of the hydrogels revealed that the hydrogels exhibited significant swelling and biodegradation. The JFP + Alg + Gen 0.05% hydrogel presented significantly greater antioxidant activity (71 ± 3.5%) compared to other hydrogels. The antibacterial studies showed that the JFP + Alg + Gen 0.05% hydrogel exhibited significantly higher inhibition against Staphylococcus aureus (S. aureus). In vitro cell culture studies revealed that the JFP + Alg + Gen hydrogel supported adequate cell viability and proliferation. In addition, in vivo animal experiments were carried out using Wistar albino rats to determine the efficacy of these hydrogels in diabetic wound healing applications. The JFP + Alg + Gen 0.05% hydrogel-treated group displayed faster wound contraction and shorter re-epithelialization time (21 ± 1 days) compared to the control (30 ± 2 days), JFP, and Alg hydrogels (24 ± 2 days). Furthermore, the JFP + Alg + Gen 0.05% hydrogel-treated wounds showed greater vasculature, collagen synthesis, and reduced inflammation compared to the control. Overall, the JFP + Alg + Gen 0.05% hydrogel showed 30% more therapeutic potential against diabetic wounds than the control.
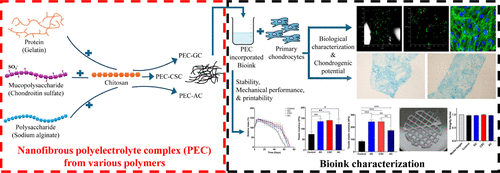
Tailoring Bioink Properties via Nanofibrous Polyelectrolyte Complexes of Distinct Polymeric Classes for Cartilage Tissue Engineering
Shreya Chrungoo - ,
Tanmay Bharadwaj - ,
Aishwarya Swain - , and
Devendra Verma *
Cartilage is a specialized connective tissue with limited self-regenerative ability, and current cartilage-related trauma treatments often fail to achieve consistent healing. Developing bioinks that closely mimic the native physical, mechanical, and biological microenvironment of cartilage is a critical challenge in tissue engineering. Here, we present a systematic study on the development of composite bioinks reinforced with nanofibrous polyelectrolyte complexes (PECs) derived from various categories of biopolymers: proteins (gelatin), mucopolysaccharides (chondroitin sulfate), and polysaccharides (alginate) in combination with chitosan. By integrating PECs of distinct biochemical origin into a BSA–alginate matrix, we demonstrate precise modulation of key bioink properties relevant for cartilage tissue engineering. Our findings reveal that incorporating PECs significantly enhances crosslinking density, in vitro stability, and mechanical strength (>120 kPa compressive modulus and >170 kPa tensile modulus). All of the bioinks exhibit excellent printability, shape fidelity, shear-thinning behavior, and cytocompatibility. Specifically, bioinks incorporated with gelatin–chitosan and chondroitin sulfate–chitosan PECs demonstrated proliferation of encapsulated primary chondrocytes and production of cartilage-specific glycosaminoglycans within the bioprinted constructs after 14 days of in vitro culture. This work highlights the pivotal role of tailored PEC compositions in tuning the physicochemical, mechanical, and biological performance of natural polymer-based bioinks. Our approach provides a versatile platform for engineering next-generation cartilage-biomimetic bioinks, offering significant promise for advancing cartilage tissue engineering.
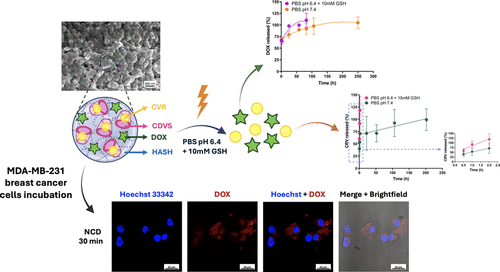
Codelivery of Doxorubicin and Carvacrol via a β-Cyclodextrin−Hyaluronic Acid Nanogel System for Targeted and Stimuli-Responsive Anticancer Therapy
Lakshmi Sathi Devi - ,
Maria Beatrice Morelli - ,
Consuelo Amantini - ,
Maria Rosa Gigliobianco - ,
Qisan Ma - ,
Piera Di Martino - ,
Cristina Casadidio *- , and
Roberta Censi
This publication is Open Access under the license indicated. Learn More
Nanogel systems offer transformative potential in overcoming critical challenges of traditional therapies, such as poor solubility, low bioavailability, and systemic toxicity. In this study, nanogels were developed using thiolated hyaluronic acid (HASH) and vinyl sulfonated β-cyclodextrin (CDVS) via a water-in-oil emulsion method. The resulting HASH/CDVS nanogels uniquely integrate HA CD44-targeting capability with CD host−guest chemistry to encapsulate both hydrophilic doxorubicin hydrochloride (DOX) and hydrophobic carvacrol (CRV), enabling effective codelivery of chemically distinct drugs. The HASH/CDVS nanogels displayed a uniform spherical morphology with an average diameter of 200 ± 25 nm and a PDI of 0.21 ± 0.13. Stability studies confirmed structural integrity under physiological (150 mM PBS pH 7.4) and tumor-like (178 mM PBS pH 6.4 + 10 mM glutathione) conditions, with selective degradation triggered by hyaluronidase (150 U/mL), highlighting their enzyme-responsive behavior. In vitro release experiments demonstrated a sustained, stimuli-responsive release profile, with accelerated drug release in acidic and reductive environments mimicking the tumor microenvironment. Cellular uptake and cytotoxicity assays in triple-negative breast cancer cells confirmed efficient internalization and significantly enhanced therapeutic efficacy of the nanogel system. Overall, these HASH/CDVS-based nanogel represent the first nano dual-delivery system for DOX and CRV, offering a synergistic and tumor-responsive platform for advanced combination cancer therapy.

Triple-Synergistic Therapy with Cobalt-Pheophytin Coordination Micelles for Overcoming Drug Resistance in Pancreatic Cancers
Zhen Jiang - ,
Yue Pan - ,
Gengqi Liu - ,
He Ren - , and
Yumiao Zhang *
Pancreatic ductal adenocarcinoma (PDAC) represents one of the deadliest malignancies as KRAS (Kirsten rat sarcoma viral oncogene homolog) mutations continuously activate multiple downstream signaling pathways, leading to drug resistance. To overcome this issue, herein, we developed a cobalt-pheophytin (CoPheo) coordination micelle chelating two chemotherapeutic agents including ONC201 and Palbociclib (Pal), yielding CoPheo-ONC201-Pal-F127. Compared with other metals, cobalt exhibits stronger coordination ability and higher drug-loading efficiency. The resulting ligand bonds are both acid- and GSH-responsive so that the cargos could be responsively released in the tumor microenvironment. To overcome drug resistance in KRAS-mutant cancers, CoPheo-ONC201-Pal-F127 is able to regulate three independent cell death pathways including ferroptosis, disulfidptosis, and cellular senescence. In particular, the cobalt-mediated Fenton-like effect synergistically enhances ONC201-induced serine−threonine protein kinase AKT (AKT) inhibition to amplify ferroptosis. Also, cobalt-mediated NADPH oxidation disrupts redox homeostasis in synergy with Pal-induced NADPH depletion, triggering disulfidptosis. In addition, ONC201-mediated mitogen-activated protein kinase kinase (MEK) inhibition, combined with Pal-induced CDK4/6 blockade, promotes cellular senescence and remodels the tumor microenvironment. These three independent mechanisms collectively establish a mutually enhanced therapeutic strategy capable of overcoming the complex drug resistance driven by multiple downstream signaling pathways in KRAS-mutant pancreatic cancer.
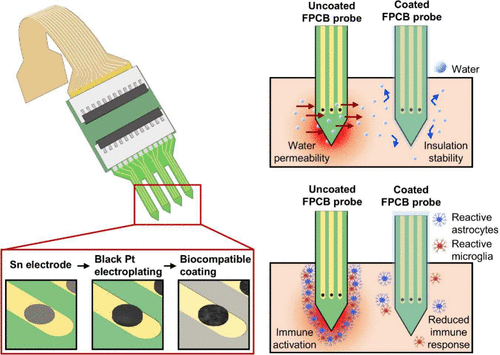
Biocompatible Lubricant-Coated Flexible Neural Probes with Enhanced Long-Term Recording Stability
Haeyun Lee - ,
Seungjun Lee - ,
Kyeong Seob Hwang - ,
Giheon Kim - ,
Yeonghwa Hong - ,
Minseok Kim - ,
Jonghee Eun - ,
Hong Nam Kim - ,
Namsun Chou *- , and
Hyogeun Shin *
Implantable neural probes enable high-resolution, multi-unit recordings and are essential tools for studying neurological disorders and developing brain−machine interface (BMI) technologies. However, conventional metal- or silicon-based probes exhibit significant mechanical mismatch with brain tissue, both of which elicit inflammatory responses and compromise long-term recording stability. Here, we introduce a flexible neural probe fabricated through a commercial flexible printed circuit board (FPCB) process and functionalized with a biocompatible lubricant coating to overcome these challenges. The inherent flexibility of the FPCB minimizes mechanical mismatch with brain tissue, while the coating enhances surface hydrophobicity and reduces insertion friction, thereby minimizing tissue damage during implantation. Its resistance to water ingress contributes to maintaining the probe’s electrical insulation stability, supporting stable long-term performance. In chronic mouse hippocampal implants, lubricant-coated probes maintained consistent neural signal quality for several weeks, while immunohistochemical analysis revealed markedly reduced astrocytic and microglial activation (GFAP/Iba1) compared with uncoated controls, indicating effective mitigation of neuroinflammation. In vitro cell viability assays further confirmed the high biocompatibility of the coated devices. Importantly, because this approach leverages scalable and cost-effective FPCB manufacturing, it enables the production of flexible neural interfaces that combine long-term electrical and biological stability with manufacturing practicality. This work establishes a broadly applicable strategy for next-generation neural probes, offering durable, minimally invasive, and scalable solutions for chronic recordings in BMI systems, deep brain stimulation, and neurological disease models.
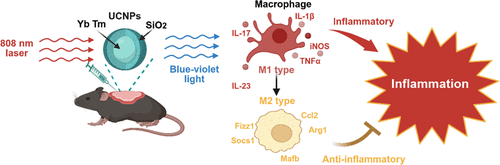
Upconversion-Mediated Phototherapy for Psoriasis Treatment
Xiaoyu Ji - ,
Zhenjia Lin - ,
Ni Ding - ,
Weijia Huang - ,
Jiaming Ji - ,
Chaojin Chen - ,
Sufang Chen - ,
Du Cheng - ,
Ziqing Hei *- ,
Weifeng Yao *- , and
Zhen Zhang *
Psoriasis is a chronic, relapsing, immune-mediated systemic inflammatory disease that affects 2–3% of the global population. It affects the skin and leads to complications in multiple systems, severely impacting patients' quality of life. Although traditional topical therapies, systemic treatments, phototherapy, and emerging biological treatments have progressed in treating psoriatic dermatitis, they still cannot cure psoriasis and are associated with multiple side effects, including infections and phototoxicity. Building upon conventional narrowband ultraviolet B (NB-UVB) phototherapy, our research utilized upconversion nanoparticles (UCNPs) for in vivo phototherapy in the treatment of psoriasis. When taken up by macrophages, under 808 nm near-infrared (NIR) light with better tissue penetration, the UV light at 365 nm and blue light at 475 nm emitted by UCNPs could potentially modulate opsin 3, inhibiting pro-inflammatory (M1) macrophage (marked by IL-1β, iNOS, TNF-α, IL-23, IL-17A) and promoting anti-inflammatory (M2) macrophage (marked by Fizz 1 and Arginase 1), indicating that UCNPs-mediated phototherapy could switch psoriasis macrophages from M1 to M2. Transcriptomic profiling further revealed that this phototherapy reverses the IL‑23−induced psoriatic phenotype in RAW264.7 macrophages by upregulating M2‑related gene expression while downregulating key inflammatory effectors, which significantly improves psoriasis-like dermatitis without causing phototoxicity, highlighting the potential of phototherapy for psoriasis.
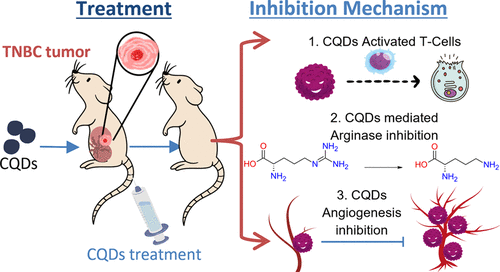
Anti-Breast Cancer Effects of Carbon Quantum Dots: In Vivo Angiogenesis and Arginase Co-Inhibition
Gurmeet Singh - ,
Somedutta Maity - ,
Desh Deepak Yadav - ,
Taniya Das - ,
Kirti Wasnik - ,
Rajalaxmi Pradhan - ,
Aman Srikant Kudada - ,
Divya Pareek - ,
Sukanya Patra - ,
Anjali Mourya - ,
Subrata Panda - ,
Tarun Batra - , and
Pradip Paik *
Breast cancer evades the immune system through immune-regulating enzymes. One of them is arginase, primarily produced in tumor-resident cells, especially by tumor-associated macrophages and myeloid-derived suppressor cells. This helps malignant tumors thrive and escape immune surveillance. However, targeting arginase is challenging because of its crucial role in the urea cycle. Pharmacological inhibitors of arginases have been linked to severe side effects in nitrogen metabolism, particularly hyperammonemia. Herein, therefore, we report on the arginase-inhibition-based in vitro and in vivo treatment of BC-bearing mice with carbon quantum dots (CQDs) derived from Azadirachta indica. These CQDs inhibit the arginase enzyme, thereby exhibiting anti-angiogenic and anti-proliferative properties. These CQDs contribute to 68 ± 5% remission of malignant tumors and 58 ± 5% inhibition of cell growth through arginase inhibition. This is particularly difficult to achieve with contemporary anti-cancer drugs. Protein upregulation of IFN-γ, IL-6, IL-8, IL-21, TLR4, LAT-1, T-cell marker, and IgG and downregulation of TNFα and IL-1β corroborate an increase in the anti-tumor immune response. Furthermore, histological results show an increase in the inhibition of cancer cells and an increase in immune cells after CQD treatment. Overall, the CQDs reported herein are paramount for the treatment of aggressive BC, which can further be used for the treatment of other cancers. The results further demonstrate that these CQDs can potentially be delivered to terminally ill patients who are not responding to contemporary therapies.

A Non-Peptidic Fluorescent Probe for In Vitro Detection of Chymotrypsin
Ramakrishnan AbhijnaKrishna - ,
Yu-Tung Fu - ,
Shu-Pao Wu - , and
Sivan Velmathi *
The work presents the development of a non-peptide fluorescent probe, NQ , for in vitro detection of chymotrypsin (Chy). Unlike traditional peptide-based probes that suffer from instability and enzymatic degradation, NQ incorporates hydrocinnamoyl chloride as a recognition group, offering enhanced stability under physiological conditions. Upon enzymatic cleavage by Chy, the probe releases a fluorogenic product (NQOH), generating a turn-on fluorescence signal. NQ exhibits a rapid initial response, with a distinguishable fluorescence signal generated within 10 min, and a low detection limit of 7.64 ng/mL. Although the enzymatic reaction reaches saturation over a period of 30−60 min, the rapid initial turn-on allows for rapid qualitative screening. A notable feature is the probe's large Stokes shift (177nm) combined with an efficient non-peptidic recognition group. This synergistic design significantly improves detection accuracy by minimizing background interference and resisting non-specific degradation in complex biological environments. The probe showed effective performance in HepG2 cell models, confirming its suitability for biological applications. This study underscores the value of non-peptide probe designs for fast and precise enzyme detection in complex biological environments.

Natural and Engineered Halloysite Clay Interact with Bacteria in a Double-Edged Manner
Simona Filice *- ,
Annalisa Pinsino - ,
Viviana Scuderi - ,
Mauro Biondo - ,
Salvatore Walter Papasergi - ,
Mario Scuderi - ,
Maria Laura Amoruso - ,
Roberta Farina - ,
Sebania Libertino - , and
Silvia Scalese *
This publication is Open Access under the license indicated. Learn More
In this paper, we investigated the behavior of a natural, low-cost, and biocompatible clay, focusing on its potential use in biomedical applications, with an eye on its ability as a material that inhibits or promotes bacterial growth. The interaction of raw and acid-treated halloysite (HT) with Gram-positive and Gram-negative bacteria representative of different environments, such as the human body, food, air, soil, water, and marine environments, was explored. Environmental strains of Escherichia coli, Acinetobacter baumannii, Lactococcus lactis, and Staphylococcus aureus were isolated and examined for their responses to HT and its derivatives after acid treatment, including acidic HT (HT (H+)), precipitate (P), and supernatant (S). HT before and after acid treatment did not have any effect on the growth of this subset of opportunistic bacteria that mainly inhabit air and water. Bacteria of marine origin (Vibrio spp and Halomonas spp) were isolated from the body lesions of a spotted diseased sea urchin, Paracentrotus lividus. These species were highly sensitive to the material tested, showing an opposite survival response under treatment with the raw or the acidic HT forms. Materials were fully characterized by scanning and transmission electron microscopy (SEM and TEM) and X-ray photoelectron spectroscopy (XPS). The responses of marine bacteria exposed to HT and its derivatives were dependent on their structural and physicochemical properties, as elucidated here.
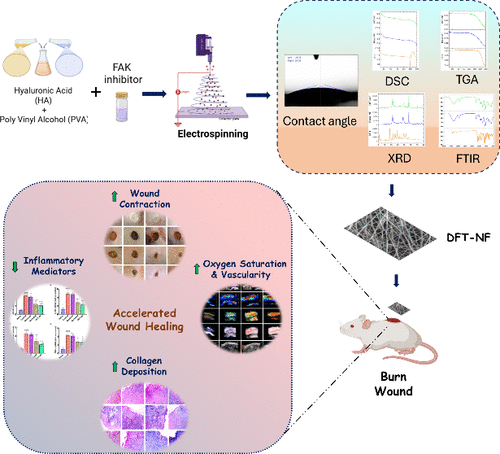
Electrospun Hyaluronic Acid/Polyvinyl Alcohol Nanofibers Encapsulating Defactinib as Bioactive Dressings for Burn Wound Therapy
Deepak Chouhan - ,
Darshita Jain - ,
Anurag Kumar Singh - ,
Abhishek Jha - ,
Rohan Dattatray Londhe - ,
Brahmeshwar Mishra - , and
Vinod Tiwari *
Chronic burn wounds remain a significant clinical challenge due to prolonged inflammation, delayed tissue regeneration, and limited effectiveness of current topical therapies. In this study, electrospun hyaluronic acid/poly(vinyl alcohol) (HA/PVA) nanofiber scaffolds loaded with Defactinib, a focal adhesion kinase inhibitor, were developed to promote burn wound healing through localized drug delivery. Optimized electrospinning conditions produced uniform, bead-free nanofibers with stable amorphous drug dispersion, as confirmed by SEM, TEM, XRD, and DSC analyses. The Defactinib-loaded nanofibers exhibited (DFT-NF) high drug entrapment efficiency, good hydrophilicity, and a biphasic release profile characterized by an initial burst followed by sustained release over 24 h. In a rat burn wound model, treatment with Defactinib-loaded nanofibers significantly accelerated wound closure compared with untreated and standard-treated groups. Non-invasive photoacoustic imaging enabled real-time monitoring of wound healing, revealing increased vascularity and improved tissue oxygen saturation in treated wounds by Day 21, indicative of enhanced vascular recovery. Histological evaluation further confirmed improved re-epithelialization, reduced inflammatory infiltration, and well-organized collagen deposition. Consistent with these findings, RT-PCR analysis showed marked suppression of key pro-inflammatory mediators (NF-κB, IL-1β, TNF-α, and IL-6) in the DFT-NF group, highlighting its potent anti-inflammatory activity. Overall, this study demonstrates that Defactinib-loaded HA/PVA nanofiber scaffolds, combined with imaging-based functional assessment, represent a promising and clinically relevant platform for advanced burn wound management.

Single-Sided Dual-Functional MPC-HEMA Coating for DMEK Grafts to Achieve Fluid-Barrier/Anti-Fouling Performance and Native Matrix Preservation
Keyi Sun - ,
Yumeng Xue - ,
Danyu Yao - ,
Huanyun Niu - ,
Shiwei Wang - ,
Jin Zhou *- ,
Feng Ze *- , and
Lizhen Wang *
Corneal endothelial dysfunction is a leading cause of global blindness. Descemet's membrane endothelial keratoplasty (DMEK) is an advanced treatment, offering minimal invasiveness, rapid visual recovery, and low risk of immune rejection. However, its clinical application remains limited because the donor graft is extremely thin (about 15 μm), which makes it vulnerable to scrolling and aqueous humor leakage during surgery. To address these issues, this study developed a unilaterally functionalized coating for donor grafts using hydroxyethyl methacrylate (HEMA) and 2-methacryloyloxyethyl phosphorylcholine (MPC). The coating is designed with dual functionality: the MPC‑modified side forms a hydration layer that blocks non‑specific protein adsorption and aqueous penetration through steric hindrance, while the HEMA component provides mechanical stability to reduce graft scrolling and displacement during surgery. The opposite side of the graft retains its natural matrix structure to support human corneal endothelial cell (HCEC) adhesion and function. Two photo-crosslinking strategies were compared: bulk copolymerization of MPC with HEMA (p(MPC-co-HEMA)) and surface grafting of MPC onto p(HEMA) (p(MPC-graft-HEMA)). Their physicochemical properties, antifouling performance, and clinical operability were evaluated to select the optimal coating method. The developed materials demonstrated stable mechanical properties, good biocompatibility, and strong resistance to protein and bacterial adhesion. In summary, this unilaterally functionalized coating offers a practical solution to the key challenges of DMEK surgery, showing significant potential to improve surgical success and promote broader clinical application.
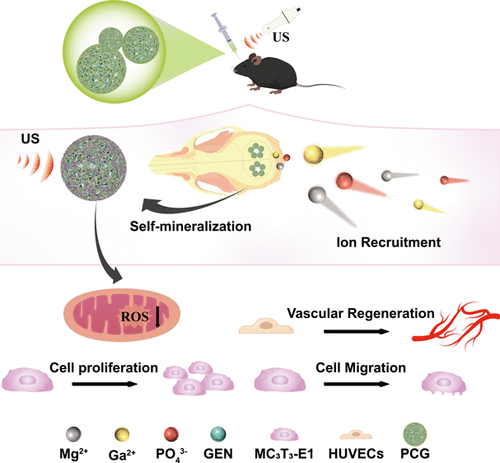
Four-Synergy Piezoelectric Microspheres Based on Bone Self-Mineralization for Enhanced Bone Regeneration
Jiawei Yao - ,
Xiaoyu Zheng - ,
Jingxu Wang - ,
Zhaowei Xing - ,
Houjuan Qi *- , and
Zhanhua Huang *
The clinical treatment of irregular bone defects resulting from trauma, tumor resection, and infection remains a significant challenge. To address this issue while enhancing material applicability and bioactivity, this study pioneers the development of injectable piezoelectric bioactive poly(l-lactic acid) (PLLA) microspheres with time-controlled release functionality. These microspheres possess a unique internal three-dimensional network structure, are surface-modified with carboxymethyl cellulose (CMC), and are loaded with geniposide (GEN), achieving a quadruple synergistic effect encompassing structural support, biomimetic mineralization, electrical stimulation, and drug release. Specifically, the negatively charged CMC coating chelates Mg2+/Ca2+ ions in body fluids, significantly enhancing osseointegration through a biomimetic mineralization process. Following CMC modification, the zeta potential of the microspheres shifted from −9.9 mV to −32.27 mV, and the water contact angle decreased from 116° to 56°, markedly improving their stability and hydrophilicity. The 3D network scaffold generates rhythmic electrical stimulation via the piezoelectric effect, effectively promoting directed migration and differentiation of osteoblasts. Under ultrasonic activation, the peak cumulative drug release rate increased from 68.17% to 73.12%, while the 36 h closure rate in a cell scratch assay rose from 84.9% to 98.8%. The PLLA microspheres employ a piezoelectric-chemically coupled bioactive regulation model, offering an innovative solution to treating traumatic bone injuries.
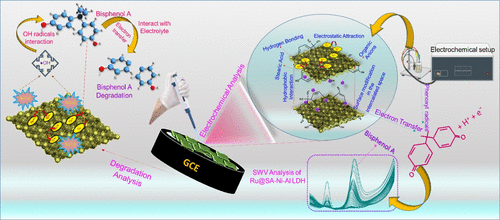
Biomodified NiAl LDH for High-Performance Electrochemical Sensing and Degradation of Bisphenol A
Madasamy Madhu Malar - ,
Anandhakumar Muthaiah - , and
Jeyaraj Wilson *
Bisphenol A (BPA) is a chemical of increasing concern because of its widespread harmful impacts on the environment and aquatic systems. To address this issue, we synthesized a stearic-acid-modified nickel–aluminum layered double hydroxide with rutin intercalation (Ru@SA-NiAl LDH) for the electrochemical detection of BPA. The excellent reduction in band gap lowers the energy required for electron excitation, thereby improving conductivity and charge transfer at the electrode surface. In the same way, increased roughness in the composite indicates successful Ru loading and provides more active sites, improved surface heterogeneity, and enhanced interaction capability for BPA detection. The [Fe(CN)6]3–/4– redox probe validated the electrochemical activity of Ru@SA–NiAl LDH and its suitability for BPA sensing by demonstrating efficient interfacial charge transport, high stability, successful surface modification, enhanced electroactive surface area, and rapid electron-transfer kinetics. In the SWV analysis, rutin (Ru) acts as the electron mediator to shuttle electrons to BPA and hence improves the sensitivity of the Ru@SA-NiAl LDH with a linear range of 10 nM–600 μM. Its LOD is 1.29 nM and serves as a reliable platform of BPA in real-time environmental monitoring. Interestingly, we also performed a degradation study of BPA with different metal ions such as Fe, Cu, and Cr interacting with •OH radicals, oxidatively breaking BPA into phenoxyl radicals, and reducing the amount of electroactive BPA available for oxidation at the electrode. Also, the analysis of the degradation ability with different buffers demonstrated that BPA is deprotonated by phenolate ions, which are nucleophilic and have more reactivity to degrade BPA in NaOH buffer. Overall, Ru@SA-NiAl LDH exhibited outstanding electrochemical sensing and degradation ability.
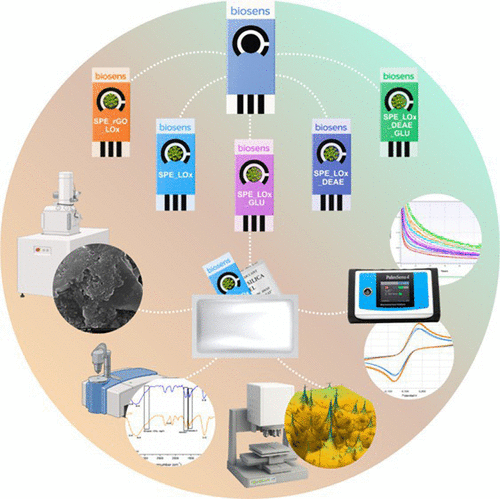
Eliminating Complex Matrices: A Minimalist Approach to Enzyme-Based Biosensors Using Screen-Printed Electrodes with a Packaging Strategy to Enhance Durability for Lactate Detection
Milleny Germann Souza - ,
Marcele Arais Hocevar - ,
Duane da Silva Moraes - ,
Gabriela Victória de Mello Jantzch - ,
Felipe Longaray Kadel - ,
Thuany Maraschin - ,
Willyan Hasenkamp - ,
Julia Konzen Moreira *- , and
Jacqueline Ferreira Leite Santos *
This publication is Open Access under the license indicated. Learn More
Enzyme-based biosensors commonly rely on complex immobilization matrices to ensure sensitivity and stability, increasing fabrication cost and limiting scalability. Here, we demonstrate a simple and straightforward packing strategy to allow using a minimalist lactate biosensor that eliminates these matrices by directly immobilizing lactate oxidase onto screen-printed electrodes. This matrix-free configuration achieved the highest sensitivity (4.22 μA dL mg−1) and a low detection limit (3.1 mg dL−1), outperforming electrodes incorporating nanomaterials or chemical stabilizers. Although crosslinking and polymeric additives slightly reduced sensitivity, they preserved concentration discrimination without offering clear performance advantages. When tested in human plasma samples containing potential interferents, the biosensor exhibited comparable behavior, achieving a sensitivity of 4.89 μA dL mg−1. Long-term durability tests showed stable enzymatic activity and consistent current responses over six months when electrodes were stored using a simple aluminum packaging and desiccant strategy, demonstrating that appropriate post-fabrication preservation can replace complex immobilization approaches. In the absence of appropriate packaging, the biosensors exhibited significant performance degradation, leading to underestimation of lactate concentrations after two days of storage under ambient temperature and humidity conditions. The biosensor reliably detects lactate across clinically relevant concentrations and establishes that simplifying design can enhance performance, durability, and translational potential, supporting more accessible point-of-care lactate monitoring.
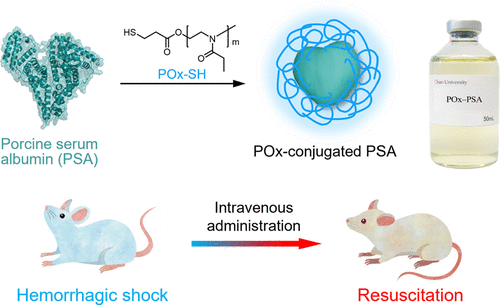
Poly(2-ethyl-2-oxazoline)-Conjugated Porcine Serum Albumin as a Veterinary Albumin Therapeutic
Junya Fujisawa - ,
Ryota Hattori - ,
Kazuki Saito - ,
Taiga Yamada - ,
Masatoshi Yamaguchi - ,
Ryo Hashimoto - ,
Mitsutomo Kohno - ,
Ryota Tochinai - , and
Teruyuki Komatsu *
Advances in veterinary medicine have greatly improved the health and quality of life of companion animals. However, no reliable supply system exists for blood-derived products. Poly(2-ethyl-2-oxazoline)-conjugated porcine serum albumin (POx−PSA) was engineered as a veterinary albumin therapeutic to maintain circulating blood volume in domestic cats and dogs. This study details the synthesis, structural characteristics, and physicochemical properties of POxM-n−PSA conjugates bearing varying average numbers (n) of POxM chains (where M denotes polymer molecular weight). Their preclinical safety, efficacy, and non-toxicity were further assessed in vivo. The conjugation number n was precisely controlled within the range of 1–17. Four variants—POx5k-6−PSA, POx5k-10−PSA, POx10k-6−PSA, and POx10k-10−PSA—were prepared, and their solution properties were comprehensively analyzed. Particle size increased proportionally with POx chain length. At equivalent PSA concentrations, both viscosity and colloid osmotic pressure increased with higher POx molecular weight and greater degrees of conjugation. Among the prepared conjugates, POx5k-6−PSA was identified as the most suitable structure for veterinary albumin therapeutic. In a rat hemorrhagic shock model, POx5k-6−PSA rapidly restored mean arterial pressure and the heart rate to pre-shock levels. No abnormalities were observed in serum biochemical parameters or histopathological examinations at 2 h or 7 days post-resuscitation. Moreover, rats receiving a high dose corresponding to 2.4-fold the anticipated maximum clinical dose exhibited no adverse clinical signs or organ alternations over a 14-day monitoring period. These findings demonstrate that POx5k-6−PSA effectively replenishes circulating blood volume and exhibits a favorable safety and efficacy as a veterinary albumin therapeutic.

Multitargeted Anticancer Strategy through DNA Damage and Mitochondrial Collapse by a Ferrocene-Benzimidazolium Salt
Nitisha Beniwal - ,
Bikash Lahkar - ,
Girbide Amitkumar Ramakant - ,
Chandra Lekha Putta - ,
Aravind Kumar Rengan *- , and
Ganesan Prabusankar *
The development of targeted and selective organometallic anticancer drugs is a primary emphasis in contemporary chemotherapeutic research. We provide the synthesis and thorough biological assessment of a new benzimidazolium-derived ferrocenyl compound, N-ferrocenylmethyl-N′-(2-pyridylmethyl) benzimidazolium iodide (FBP). The compound demonstrates significant cytotoxic efficacy against HeLa cervical cancer cells, exhibiting a substantially reduced IC₅₀ value and an elevated selectivity index relative to Doxorubicin and Ferrocene. Biocompatibility evaluations in standard fibroblast cell lines (NIH3/T3 and L929) demonstrated no off-target toxicity within physiologically pertinent dosage ranges. Cellular uptake investigations utilizing Hoechst and MitoTracker Green labeling demonstrated effective internalization and primary mitochondrial location of FBP in HeLa cells. Mechanistic studies, encompassing Live/Dead viability assays, DCFDA-based quantification of reactive oxygen species, singlet oxygen detection through SOSG, and JC-1 analysis of mitochondrial membrane potential, collectively indicate that FBP induces oxidative stress and mitochondrial dysfunction, resulting in apoptotic cell death. Subsequent assessments related to apoptosis revealed distinct nuclear condensation and fragmentation through DAPI staining, along with notable DNA double-strand break production indicated by γ-H2AX expression. Furthermore, 3D multicellular spheroid experiments validated the penetrating ability and prolonged anticancer effectiveness of FBP in a tumor-mimicking environment.
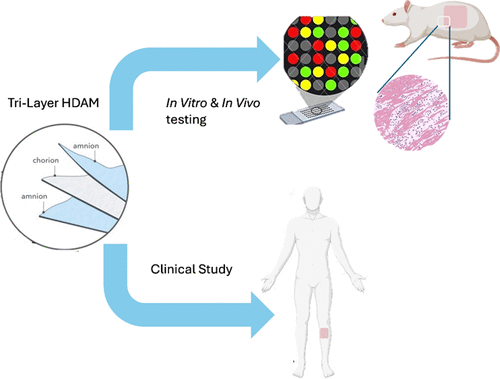
A Triple-Layer Amniotic Membrane Dressing Drives Robust Wound Healing: In-Depth Protein Profiling and In Vivo Validation in Rat and Human Subjects
Mohammadali Safavieh *- ,
H. Frank Burrows - ,
Olivia Logan - ,
Sadhana S. Joshi - ,
Laura K. Rivera-Tarazona - , and
Mora C. Melican *
This publication is Open Access under the license indicated. Learn More
In chronic ulcers, early intervention to obtain wound closure is a critical requirement for optimal reparative success. Human dried amniotic membrane (HDAM) has been widely used as a wound covering due to three characteristics: its content of highly enriched extracellular matrix (ECM) and bioactive cytokines, its long shelf life, and its suitable biomechanical properties adequate for handling in the dried state. The ECM composition can be enhanced by the creation of a sandwich composed of three distinct layers of the amniotic membrane without creating any detrimental manufacturing shortcomings or clinical procedural complications. In this study, we investigate the mechanisms of action that contribute to wound healing using the histological analysis of various proteins, which exist in tri-layer HDAM. In addition, extensive proteomic analysis of 80 cytokines and growth factors is compartmentalized and quantified based on their functions in wound healing. More importantly, the reciprocal interactions between ECM components and keratinocyte proliferation are investigated. Using an animal wound model, in vivo efficacy of the tri-layer HDAM is assessed based on wound closure, tissue granulation, and re-epithelialization rates and is compared to a no-graft group. Lastly, the therapeutic effects of tri-layer HDAM and possible adverse effects on patients are carefully recorded through conducting a clinical study with a 37-patient population. The collective results suggest that the tri-layer HDAM positively affects various critical pathways in chronic wound healing and shows no adverse effects in human clinical studies.
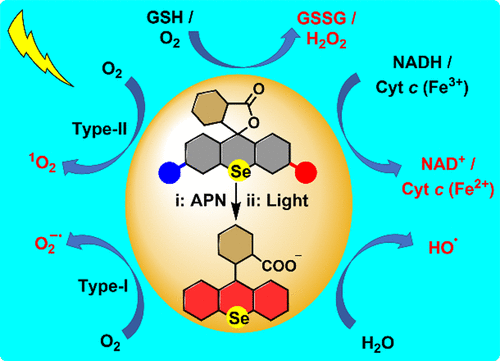
Photocatalytic Depletion of GSH/NADH and O2-Adaptive Pathway Switching in Producing ROS: Overcoming Treatment Resistances of Cancer Cells to Photodynamic Therapy and Inducing Ferroptotic Cell Death
Peiwen Fan - ,
Jianzhi Chen - ,
Jing Liu *- ,
Hongxing Zhang - ,
Yuanqiang Sun *- , and
Wei Guo *
Photosensitizers (PSs), capable of overcoming the treatment resistances caused by the elevated levels of glutathione (GSH) and nicotinamide adenine dinucleotide (NADH) within cancer cells as well as the hypoxic microenvironment of solid tumors, are highly desirable for photodynamic therapy (PDT) of tumors. Herein, leveraging the redox activity and heavy atom effect of selenium (Se), we develop a cancer-cell-targetable, enzyme/light dual-activatable PS based on a Se-rhodamine platform. Upon encountering cancer cells, the dual-activatable PS can be enzymatically cleaved by aminopeptidase N (APN, overexpressed on the outer membrane of cancer cells) to release a cytomembrane-permeable prodrug PS, which, after being internalized by cancer cells, can be further activated by light to produce an active PS. The resulting active PS features not only dual photocatalytic activities in depleting GSH and NADH but also O2-adaptive type-II (under normoxia) → type-I (under hypoxia) pathway switching in producing reactive oxygen species (ROS), thereby effectively potentiating the sensitivity of cancer cells to PDT and overcoming the treatment resistances. Due to interrupting the GSH/NADH-dependent antioxidant systems, the active PS almost exclusively induces cancer cell ferroptosis. The study provides an all-in-one strategy in overcoming the treatment resistances of tumors to PDT, paving the way for developing high-performance PSs in the future.
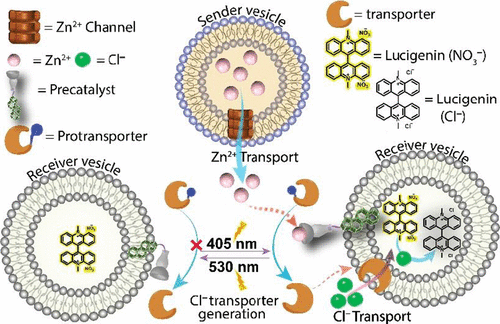
Photoregulated Sequential Chemical Relay via Artificial Nanochannels
Mrinal Kanti Kar - ,
Soumya Srimayee - ,
Biswa Mohan Prusty - ,
Rahul Kumar - , and
Debasis Manna *
Development of synthetic ion transporters and in situ chemical reaction-mediated communication systems that mimic cellular signaling processes is crucial for preparing protocells to understand complex cellular signaling and related functions. Although there have been advances in synthetic ion transporters, the role of this synthetic ion transporter-mediated inter-vesicular signal transduction remains largely unexplored. Herein, we describe a photoregulated inter-vesicular molecular communication system that employs synthetic ion transporters and a photoresponsive precatalyst. This artificial communication system regulates the transformation of one chemical signal (Zn2+) from the sender vesicle into another chemical signal (Cl−) to the receiver vesicle through a photoregulated chemical reaction in the extravesicular environment. Fluorescence-based ion transport and electrophysiological studies showed that the potent salicylaldehyde-based imine derivatives of 9-alkyl-9H-carbazole-3,6-diamine self-assemble within the lipid bilayer to form supramolecular nanochannels that selectively efflux Zn2+ from the sender vesicles. The use of a photoresponsive precatalyst anchored to the outer membrane of the receiver vesicle and the protransporter plays a crucial role in transforming the Zn2+-based chemical signal from the sender vesicles to a Cl−-based chemical signal into the receiver vesicle in a controlled manner. The Zn2+-bound catalyst-driven hydrolysis facilitates the release of an active transporter, a salicylanilide derivative, from its protransporter to transport Cl− to a larger population of receiver vesicles, resulting in inter-vesicle signal transduction.

Engineered Dual pH/GSH-Responsive Lignin-Based Nanocarriers for Enhanced Tumor Growth Inhibition
Jin Wang - ,
Zhuang Wang - ,
Suyang Dai - ,
Meijun Zhao - ,
Yuqiang Li *- ,
Qingsong Yu - ,
Danhua Zhou *- ,
Zhihua Gan - , and
Zhenbo Ning *
In this study, we designed a lignin-based nanocarrier system with dual-stimuli responsiveness to the acidic pH and elevated glutathione (GSH) levels in the tumor microenvironment, and doxorubicin (DOX) was loaded to develop a nanomedicine delivery platform for antitumor treatment. Under the optimized conditions, uniform lignin-based nanoparticles with a diameter of approximately 150 nm and well-controlled physicochemical properties were successfully prepared. In vitro release studies confirmed the selective drug release under simulated tumor conditions (pH 6.5, 10 mM GSH). Biological evaluation revealed that the drug-loaded nanoparticles exhibited potent cytotoxicity against 4T1 breast cancer cells, while maintaining over 85% viability in normal cells, indicating good biocompatibility. In 4T1 tumor-bearing mice, the engineered nanocarriers demonstrated significantly enhanced therapeutic efficacy, achieving 71.84% tumor growth inhibition compared to that of 58.78% with free DOX, along with reduced systemic toxicity. In summary, this study demonstrates the successful transformation of sustainable lignin into an efficient therapeutic platform via precise chemical modification, effectively combining biopolymer advantages with stimulus-triggered drug release for more promising and biodegradable nanocarriers for cancer therapy.

A Biomimetic Nanoplatform for Near-Infrared-Assisted Heat-Mediated Synergistic Therapy for Glioblastoma
Buse Sancakli - ,
Sezen Atasoy - ,
Tugba Gencoglu Katmerlikaya - ,
Zeynep Cagman - , and
Aydan Dag *
This publication is Open Access under the license indicated. Learn More
Cell membrane (CM) camouflage has emerged as a powerful biomimetic strategy for enhancing the precision of targeted drug delivery, offering an effective solution to the major limitations of phototheranostic nanomaterials, which often suffer from reduced therapeutic efficacy and imaging performance due to tumor heterogeneity and microenvironmental delivery barriers. Here, we report a biomimetic phototheranostic nanoplatform engineered for the synergistic treatment of glioblastoma multiforme (GBM) via near-infrared (NIR)-assisted, heat-enhanced chemotherapy. Magnetic nanoparticles (M) with improved stability and biocompatibility were synthesized to enable efficient magnetic hyperthermia. These nanoparticles were further functionalized with cancer CM and temozolomide (TMZ)-conjugated, cell-penetrating peptide-based copolymers (P), providing homotypic targeting capability and enhanced intratumoral accumulation. This multifunctional coating significantly improved selectivity toward GBM cells and enabled combined photothermal and chemotherapeutic activity. Cellular studies under both illuminated and dark conditions revealed pronounced synergistic cytotoxicity arising from the integration of NIR-mediated magnetic hyperthermia with TMZ delivery. Compared to nonpolymer-functionalized controls, the proposed system more effectively overcame tumor heterogeneity and microenvironmental barriers, yielding superior therapeutic performance. These findings highlight the potential of this biomimetic platform for targeted and multimodal GBM theranostics. Overall, this work introduces a versatile biomimetic nanocamouflage strategy that leverages natural cellular functionalities and nanotechnology to advance targeted GBM therapy, providing a promising avenue for more precise and effective treatment of highly malignant brain tumors.
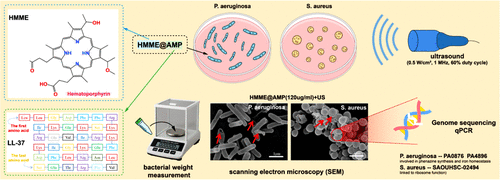
Sonodynamic Therapy with HMME@AMP Conjugate: Efficient Bactericidal Efficacy through Bacterial Structural Damage and Critical Gene Downregulation in Pathogenic Bacteria
Changlong Li - ,
Fengmeng Teng - ,
Fengmin Wu *- ,
Hui Zhang - ,
Chunbing Zhang *- , and
Dong Zhang *
To combat the growing challenge of bacterial infections, sonodynamic antibacterial technology has gained increasing attention. However, current approaches still face limitations, including suboptimal efficacy and a narrow antibacterial spectrum (only targeting a single bacterial species). To overcome these drawbacks, this study designed a composite sonosensitizer, HMME@AMP, by conjugating hematoporphyrin monomethyl ether (HMME) with the antimicrobial peptide LL-37. This composite achieves efficient inhibitory effects against representative Gram-negative and Gram-positive bacteria through sonodynamic therapy (SDT). Using optimized ultrasound parameters (0.5 W/cm2, 1 MHz, 60% duty cycle), we evaluated the antibacterial activity and mechanisms of HMME@AMP through agar culture, flow cytometry, bacterial weight measurement, scanning electron microscopy (SEM), whole-genome sequencing, and quantitative real-time PCR (qPCR). Results demonstrated that HMME@AMP exhibited strong, concentration-dependent antibacterial effects against Pseudomonas aeruginosa (P. aeruginosa) and Staphylococcus aureus (S. aureus) under ultrasound irradiation. At a concentration of 120 μg/mL, bacterial weight measurement results showed that the bacterial weight inhibition rates reach 56.4% (for P. aeruginosa) and 87.3% (for S. aureus), while flow cytometry indicated survival rates below 3 and 7%, respectively, confirming its excellent inhibitory ability against P. aeruginosa and S. aureus. SEM imaging revealed ultrasound-induced nanoscale pores and membrane collapse, indicating combined physical damage and ROS-mediated oxidative stress. Genome sequencing and qPCR further showed that HMME@AMP downregulated critical genes in P. aeruginosa (e.g., PA0876, PA4896, involved in phenazine synthesis and iron homeostasis) and S. aureus (e.g., SAOUHSC-02494, linked to ribosome function), disrupting bacterial metabolism and proliferation. This study demonstrates that HMME@AMP-mediated SDT achieves potent antibacterial effects through dual mechanismsphysical disruption and genetic regulationoffering a promising, antibiotic-free strategy for treating deep-tissue infections.

Tuning Th1 Immunity through a TLR7/8 Agonist HYBRID2-Formalin-Killed Leishmania donovani Antigen Immunomodulatory System in Visceral Leishmaniasis
Sandeep Kaur - ,
Shivani Thakur - ,
Kushvinder Kumar - ,
Sukhbir Kaur *- , and
Deepak B. Salunke *
Visceral leishmaniasis (VL) is a fatal parasitic disease for which safe and effective immunotherapeutic strategies remain limited. Modulation of innate immune signaling using small-molecule Toll-like receptor (TLR) agonists represents a promising approach to enhance host-directed antiparasitic responses. In this study, we investigated the immunomodulatory efficacy of a synthetic TLR7/8 agonist, HYBRID2, formulated with formalin-killed Leishmania donovani antigen (FK), in a BALB/c mouse model of VL. Immunized mice challenged with L. donovani exhibited a marked reduction in splenic parasite burden following FK + HYBRID2 administration. Mechanistic evaluation of splenocytes revealed enhanced redox activity, with significantly elevated reactive oxygen species and nitric oxide production, accompanied by up-regulation of inducible nitric oxide synthase and NF-κB expression. Cytokine analysis demonstrated a pronounced Th1-biased immune response, characterized by increased INF-γ and TNF-α levels. Comparative assessment showed that FK + HYBRID2 elicited superior immunostimulatory and antileishmanial effects relative to FKAlone or FK combined with Resiquimod. Collectively, these findings establish HYBRID2 as a potent innate immune modulator enhancing redox responses and NF-κB activation alongside Th1 polarization, highlighting its potential utility as an immunomodulatory adjuvant platform against intracellular parasitic infections.
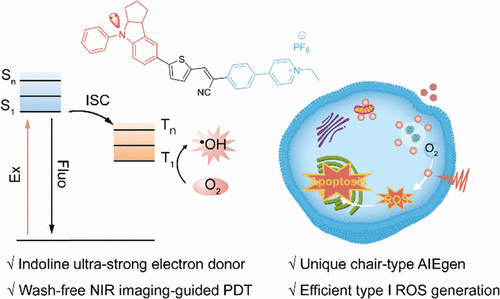
Chair-Shaped Indoline Donor Enabling Near-Infrared Imaging-Guided Type I Photodynamic Therapy
Guofeng Yi - ,
Qiqi Xu - ,
Haodong Liu - ,
Chenggong Ma - ,
Ying Wang *- ,
Weijun Zhao *- , and
Chengyun Wang *
Imaging-guided photodynamic therapy (PDT) represents a promising treatment modality owing to its high precision and minimal invasiveness. However, insufficient oxygen supply within solid tumors and aggregation-caused quenching (ACQ) of photosensitizers (PSs) limit the clinical application of imaging-guided PDT. Herein, we developed a type I reactive oxygen species (ROS) predominant photosensitizer (IDL-CPy) based on a unique chair-shaped ultra-strong electron donor indoline with aggregation-induced emission (AIE) characteristics, enabling imaging-guided type I PDT. The electron-rich indoline structure enhances intramolecular charge transfer, which narrows the singlet-triplet energy gap, thereby facilitating type I ROS generation. Theoretical and experimental analyses demonstrate that IDL-CPy exhibits an ideal intersystem crossing rate and triplet excited state lifetime for generating ROS, contributing to potent anticancer efficacy in vitro (IC50 = 0.34 μM). This research represents the first indoline-based AIE photosensitizers and provides a promising candidate for imaging-guided type I PDT.
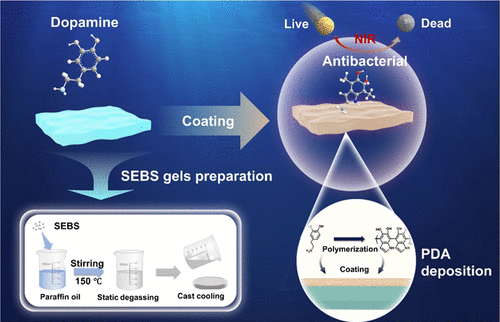
Multidimensional Biological Evaluation of Polydopamine-Modified SEBS Gels for Improved Safety
Xinlin Ma - ,
Zijun Jia - ,
Yihuan Deng - ,
Yuanyuan Xiang - ,
Jichuan Zhang - ,
Tongliang Zhou - ,
Honglin Liu - ,
Hui Wang - ,
Haiyan Gong - ,
Lan Ling - , and
Anchao Feng
While SEBS-based elastomers are utilized in medical devices, the lack of systemic safety protocols in physiological microenvironments hinders their clinical translation. Furthermore, the intrinsic hydrophobicity of SEBS and the inability of traditional 2D/animal models to replicate human tissue responses necessitate more robust evaluation frameworks. Herein, a SEBS/paraffin oil gel was synthesized via a one-pot method and functionalized with polydopamine (PDA) coatings to enhance hydrophilicity and antimicrobial efficacy. A multiscale safety evaluation framework was established, integrating mechanical characterization, 3D human skin models, in vitro cytotoxicity profiling, and in vivo animal assays. Results demonstrate that the SEBS gel exhibits exceptional toughness and fatigue stability, while the PDA coating achieves high cellular viability (>98%). Crucially, 3D organoid models and animal studies confirmed negligible IL-6 upregulation and an absence of skin irritancy, validating the material's safety at both tissue and organism levels. This work provides a translatable paradigm that unifies materials engineering with human-mimetic evaluation, offering a standardized template for the systematic preclinical assessment of advanced medical elastomers.

Synergistically Enhanced Peroxidase-like Activity of FeSe2/rGO Nanohybrids: Kinetic, Mechanistic, and Molecular Docking Studies
Saumya Maurya - ,
Priyanka Jha - ,
Shashank Sharma - ,
Nisha Yadav *- , and
Ashish Chalana *
FeSe2/rGO nanohybrids were prepared via in situ one-pot synthesis using the hydrothermal method and were assessed for their potential for peroxidase-like activity. Structural and morphological analysis using FE-SEM and TEM confirmed the successful in situ growth and uniform distribution of FeSe2 NPs onto the rGO nanosheet matrix. Furthermore, the FeSe2/rGO nanohybrids showed superior and enhanced peroxidase-like activities compared to the bare FeSe2, as demonstrated using two substrates, 3,3′,5,5′-tetramethylbenzidine (TMB) and o-phenylenediamine (OPD). Michaelis–Menten kinetics showed a significantly lower Km of 0.00952 mM for TMB and 7.62 mM for H2O2, along with a high Vmax of 6.48 × 10−8 M s−1 (TMB) and 1.706 × 10−6 Ms−1 (H2O2) for FeSe2/rGO, outperforming bare FeSe2 and several other reported nanozymes. The kinetic parameters showed a strong substrate affinity and improved catalytic efficiency. Mechanistic study using terephthalic acid (fluorescent probe) revealed that •OH is majorly responsible for substrate oxidation, which is significantly enhanced by the synergistic effect of FeSe2 and rGO. Furthermore, in support of our experimental results, molecular docking studies with multiple peroxidase proteins also showed significantly higher binding affinities for FeSe2/rGO (–9.9 to –11.9 kcal mol−1), as compared to FeSe2 (–4.8 to –9.1 kcal mol−1). Overall, this work highlights FeSe2/rGO as a robust peroxidase mimic and efficient nanozyme with strong potential for colorimetric sensing and catalytic performance.
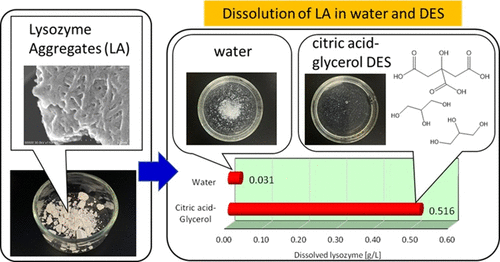
Dissolving Amyloid Fibrils with Natural Deep Eutectic Solvents: Citric Acid−Glycerol Achieves Superior Solubilization and Partial Protein Refolding
Sora Hirano - ,
Tatsuya Oshima *- ,
Asuka Inada - , and
Toshihiro Tsuruda
Amyloid protein aggregates pose a significant challenge due to their extreme insolubility and association with neurodegenerative diseases. To identify effective solvents for dissolving amyloid-forming proteins, a screening of deep eutectic solvents (DESs), including natural DESs (NADES), and related solvents was conducted. Aggregated lysozyme (LA) was reproducibly prepared under acidic and thermal conditions as a model of poorly soluble protein aggregates. Spectroscopic analyses using Thioflavin T fluorescence and circular dichroism (CD), along with morphological observations under polarized light and SEM, confirmed the formation of β-sheet-rich amyloid-like structures. The citric acid−glycerol DES showed the highest solubility for LA, outperforming other DESs and DMSO. Its performance was further enhanced by the addition of ∼5% water. This result also suggests a strong contribution of multivalent organic acids, such as citric and malic acid, to protein solubilization. CD analysis indicated that lysozyme recovered a native-like secondary structure, suggesting partial structural recovery after dissolution in this DES. These findings highlight the potential of DESs as effective media for dissolving protein aggregates and suggest their possible utility in protein refolding and related biotechnological applications, including potential future biomedical applications.
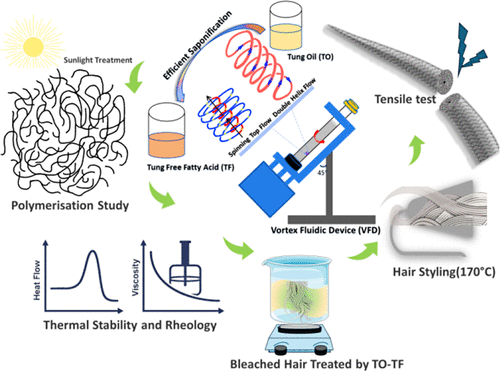
Sustainable Biopolymer Based on Tung Oil and Tung Free Fatty Acids for Hair Tensile Enhancement
Xuejiao Cao - ,
Yanting Yin - ,
Jonathan A. Campbell - ,
Xuan Luo - ,
Youhong Tang - , and
Colin L. Raston *
Tung oil (TO) and its derivatives, characterized by a high content of α-eleostearic acid, exhibit significant potential as renewable feedstocks for the development of sustainable biopolymers, particularly for structural keratin repair applications. In this study, Tung free fatty acids (TF) were generated in a one-step process from TO via a vortex fluidic device (VFD)-assisted saponification process, enabling rapid, solvent-minimal conversion with high efficiency while preserving the reactive conjugated triene system. TO, TF, and a hybrid TO–TF (1:1) copolymer were then subsequently formed under natural solar irradiation. Morphological analysis revealed that the TO–TF copolymer yields a homogeneous, conformal film, attributed to the steric modulating effect of flexible TF chains, which regulates excessive crosslinking and mitigates the brittle network formation characteristic of neat TO polymers. Structural analysis confirmed a chemically saturated surface in the copolymer, suggesting enhanced resistance to oxidative degradation. Differential scanning calorimetry revealed that the copolymer exhibited a higher post-curing transition temperature (165 °C) compared to the TO polymer (142 °C) and TF polymer (134 °C), indicating enhanced thermal network stability. Rheological measurements demonstrated improved viscoelastic performance, with a strain yield approximately seven times greater than that of the TF polymer, confirming a superior balance between flexibility and mechanical resilience. When formulated as an emulsion and applied to bleached hair under thermal styling conditions, the TO–TF (1:1) emulsion treatment produced uniform surface coverage and effective cuticle sealing on the hair surface, leading to significant mechanical recovery. Notably, yield stress increased from 89 to 124 MPa, and Young’s modulus improved by over 40% without compromising fiber elongation at break. These results demonstrated that copolymerizing TO with VFD-based TF yields a structurally optimized, sustainable biopolymer capable of reinforcing damaged hair. The established structure−performance relationship highlights its potential for next-generation eco-friendly hair-repair applications.
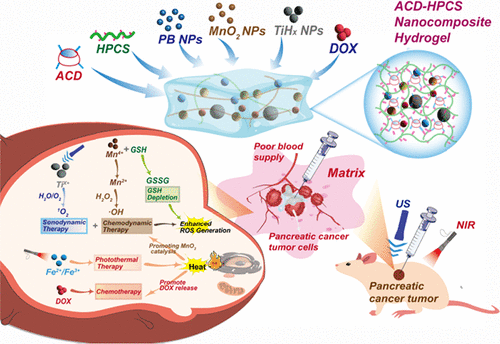
Injectable and Self-Healing Multifunctional Nanocomposite Hydrogel with an All-in-One Strategy for Efficient Synergistic Photothermal−Sonodynamic−Chemodynamic−Chemotherapy of Pancreatic Cancer
Junhang Li - ,
Jiacheng Li - ,
Weizhong Yuan *- , and
Shilong Han *
Traditional pancreatic cancer treatment has long been severely hindered by the dense stromal matrix barrier, which drastically impedes the deep penetration of chemotherapy drugs and thus compromises therapeutic efficacy, leading to unsatisfactory clinical outcomes. An injectable and rapid self-healing hydrogel based on β-cyclodextrin (β-CD) and hydroxypropyl chitosan (HPCS) loading multifunctional nanoparticles was designed for efficient synergistic therapy. Specifically, aldehyde-modified β-CD (ACD) and HPCS were cross-linked via a dynamic Schiff base reaction between aldehyde and amino groups, endowing the resultant ACD-HPCS hydrogel with excellent injectability, rapid self-healing capacity, and good biocompatibility for in situ administration, allowing it to conformably fill the irregular tumor cavities after injection. An all-in-one strategy was employed to integrate Prussian blue nanoparticle (PB NP) photothermal agent, titanium hydride nanoparticle (TiHx NP, x ≈ 2−4) sonosensitizer, manganese dioxide nanoparticle (MnO2 NP) nanozymes that generate ROS and scavenge GSH, and the chemotherapeutic drug doxorubicin (DOX) within an ACD-HPCS hydrogel. The multifunctional NPs provide the combination of near-infrared (NIR)-induced photothermal therapy (PTT), ultrasonic (US)-excited sonodynamic therapy (SDT), and chemodynamic therapy (CDT). In addition, DOX serves as a chemotherapeutic agent to exert the effect of chemotherapy (CT). By directly injecting ACD-HPCS into pancreatic cancer tissues, the multifunctional hydrogel can effectively overcome the stromal matrix barrier and circumvent the limitations of single-modal therapy, boosting drug accumulation in tumors to realize a quadruple-modal synergistic treatment effect combining PTT, SDT, CDT, and CT. This integrated therapeutic system therefore offers a highly promising and efficient strategy for clinical pancreatic cancer treatment.
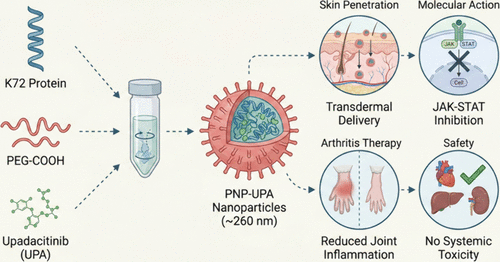
Engineered Protein Nanoparticles Enable Targeted Topical Delivery of Upadacitinib for Enhanced Arthritis Therapy
Wenrui Zhang - ,
Chaonan Jin - ,
Chao Ma - ,
Yiwen Wang - ,
Xiaojian Ji - ,
Yufei Guo - ,
Jiali Yu - ,
Chao Xue - ,
Jiaxin Bai - ,
Kai Liu *- ,
Jianyou Chen *- , and
Jian Zhu *
Rheumatoid arthritis (RA) is a chronic autoimmune disorder driven by persistent synovial inflammation and leading to joint destruction. Although the Janus kinase (JAK) inhibitor upadacitinib (UPA) is clinically effective, its systemic administration is associated with dose-limiting toxicities and poor patient adherence, highlighting an urgent need to minimize systemic exposure. Topical application offers a promising route for targeted joint delivery, but its efficacy is hampered by the formidable skin barrier and the poor retention of conventional formulations. To overcome these challenges, we developed a protein-based nanoparticle system by assembling engineered protein K72 with carboxylate-terminated poly(ethylene glycol) (PEG-COOH) to encapsulate UPA. This design significantly enhanced nanoparticle stability, drug loading capacity, and transdermal penetration efficiency. The engineered nanoparticles demonstrated sustained drug release and superior skin penetration. In a collagen-induced arthritis (CIA) mouse model, topical application of these nanoparticles achieved pronounced therapeutic efficacy against RA progression, with no observable systemic toxicity. This work establishes a safe, convenient, and highly effective topical nanoplatform for the local management of RA.
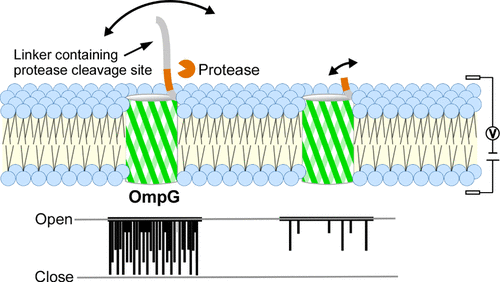
Single OmpG Nanopore Gating Regulated by Linker Length for Protease Detection
Koki Kamiya *- ,
Haruka Suzuki - ,
Kazuha Endo - , and
Toshiyuki Tosaka
Nanopore sensing is a powerful technology for single-molecule analysis. The OmpG protein is particularly suitable for occlusion-based detection owing to its inherent gating properties. However, insertion of long sequences into the internal loops of OmpG can compromise structural stability. In this study, we investigated an approach of fusing functional linkers to the N-terminus of OmpG, a method that preserves structural integrity while allowing for the introduction of specific protease cleavage sites for TEV protease, thrombin, and HRV3C. Using a planar lipid bilayer system, we analyzed the ionic currents of the OmpG variants before and after protease treatment. Our results showed that N-terminal linker length significantly modulated gating dynamics. Variants with a linker exhibited high gating frequencies, characterized by over 3.6 events/s of a close event rate and mean interevent intervals below 300 ms. Protease-mediated cleavage effectively reversed these behaviors, allowing for clear discrimination of the linker presence with accuracies of up to 90.9%. Molecular dynamics simulations corroborated these findings, showing that longer linkers had a higher probability of occluding pore entrances. Furthermore, mismatch experiments confirmed high specificity and analysis of short-duration signals revealed that the mean interevent interval is a reliable index for high-throughput screening. This study suggests that N-terminal linker-fused OmpG platforms can be used for specific detection of proteases.

Glucose-Responsive Antibacterial Nanogel for Synergistic Dual-Mode Therapy of Diabetic Wound Infections via Enhancing Cascade Generation of Hydroxyl Radicals and Hypoxia-Induced Activation of Metronidazole
Yinbang Zheng - ,
Weiqing Hu - ,
Yuze Luo - ,
Weiwei Chen - ,
Cheng Jin - ,
Li Wang - ,
Yifeng Deng - ,
Zefeng Wang - , and
Dinggeng He *
Pathogen colonization causes tissue damage, chronic inflammation, and antimicrobial resistance, leading to nonhealing wounds. Moreover, bacteria-infected diabetic wounds are particularly difficult to heal owing to persistent hyperglycemia. Herein, a glucose (Glu)-responsive antibacterial nanogel (PAMGHM) consisting of polyacrylamide (PAM) loaded with glucose oxidase (GOx), horseradish peroxidase (HRP), and metronidazole (MTZ) is rationally constructed for combating the diabetic wound infections. This nanogel was fabricated via a reverse microemulsion polymerization approach, which enabled the co-encapsulation of GOx and HRP within nanoscale gel microspheres. Within PAMGHM, GOx first catalyzes the oxidation of Glu to produce H2O2, which is subsequently utilized by HRP to generate hydroxyl radicals (•OH). The close integration of cascade enzymes ensures rapid Glu consumption and •OH production, which not only directly kills bacteria but also establishes a localized hypoxic microenvironment through O2 depletion. The induced hypoxia promotes bacterial nitroreductase expression, thereby activating the antibacterial properties of MTZ. In vitro experiments reveal that PAMGHM exhibited a powerful killing effect against both Staphylococcus aureus and Escherichia coli, demonstrating synergistic chemodynamic/chemo dual-mode antibacterial therapy. Further in vivo assays exhibit that the proposed PAMGHM accelerates diabetic wound healing by killing bacteria and alleviating hyperglycemia-induced inflammation. This cascade enzyme-mediated antibiotic activation strategy offers a useful therapeutic approach to meet the requirements of efficient antibacterial therapy, hyperglycemia control, and anti-inflammation, showing great potential in the treatment of chronic diabetic wound infections.
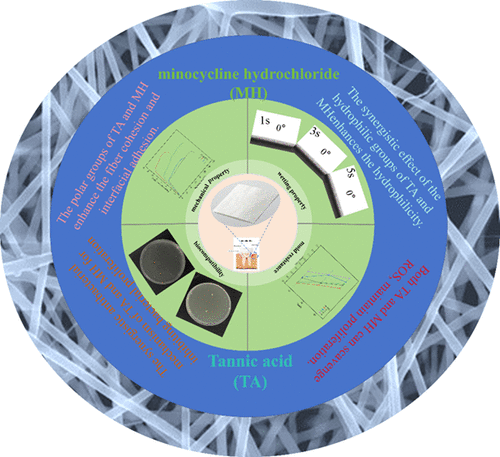
Study on the Synergistic Effect of Tannic Acid-Minocycline Hydrochloride Dual-Loaded Core-Shell Structured Nanofiber Membrane for Periodontal Tissue Repair
Xinjiao Wang - ,
Ping Liu - ,
Xiaohong Chen *- ,
Honglei Zhou *- ,
Tao Jiang - ,
Pengfei Gao - , and
Xin Kuang
This publication is Open Access under the license indicated. Learn More
The biological invasion of periodontal pathogens has brought great difficulties to periodontal tissue repair. This study reported the feasibility of coaxial electrospinning to prepare tannic acid (TA) and minocycline hydrochloride (MH) double-loaded nanofiber membranes as periodontal tissue functional repair dressings. The TA-MH @ CSNF nanofiber membrane has the best mechanical properties and superhydrophilicity, which are 83% and 72.1% higher than the tensile strength and Young's modulus of the matrix, respectively. The improvement of mechanical properties is due to the enhancement of fiber cohesion and interfacial adhesion by TA and MH polar groups, and the synergistic optimization of fiber strength and toughness is realized. Wettability optimization is due to the synergistic effect of TA and MH hydrophilic groups. The TA-MH @ CSNF nanofiber membrane with dual loading of TA and MH has the best antibacterial and cell proliferation properties. Compared with the matrix, the antibacterial rate of TA-MH @ CSNF nanofiber membrane was close to 100%, and cell proliferation was increased by 18%. TA and MH destroy the integrity of the bacterial membrane structure and block the synergistic mechanism of bacterial proliferation, which is the main reason for improving the antibacterial property of the nanofiber membrane. Moreover, TA's mild cell proliferation and MH's efficient targeted antibacterial mechanism provide an ideal environment for cell proliferation.
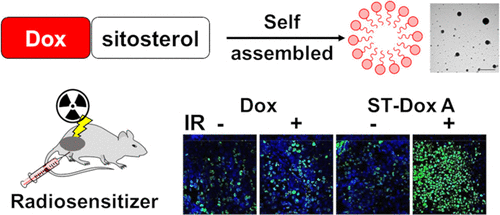
Development of a Doxorubicin−Sitosterol Conjugate as a Radiosensitizer for Cancer Therapy
Jae-Won Park - ,
Kyung-Oh Doh - ,
Sungjin Jeon - ,
Kwang Seok Kim - ,
Jong-Ik Heo - , and
Jong-Soo Choi *
Doxorubicin (Dox) is a widely used anthracycline anticancer drug. However, the clinical application is limited by severe dose-limiting cardiotoxicity and systemic toxicity. To overcome these challenges, recent studies have focused on chemoradiotherapy, reporting that combination nanoparticles with irradiation maximize the therapeutic efficiency. Herein, we developed a radiosensitizing chemotherapeutic agent by conjugating Dox and β-sitosterol (ST-Dox A) to enhance the radiotherapeutic efficacy while minimizing drug dose and toxicity. ST-Dox A self-assembled into 170 nm nanoparticles, which are expected to facilitate passive tumor accumulation via the enhanced permeability and retention (EPR) effect. Moreover, the positively charged nature of ST-Dox A (+22.7 mV) led to improved cytotoxicity and facilitated endosomal escape via the proton sponge effect, compared to the non-charged ST-Dox B. In addition, ST-Dox A showed prolonged intracellular retention, thereby enhancing its availability even at low dosage. In an animal study, a low dose of ST-Dox A (equivalent to 2 mg/kg Dox) combined with a single local irradiation (5 Gy) significantly suppressed tumor growth compared to free Dox or radio combination by increased apoptotic cell death. Consequently, the conjugation of β-sitosterol to Dox would enable passive targeting and sustained exposure of ST-Dox A, offering a promising strategy for a potent radiosensitizer. This approach achieves a significantly enhanced antitumor effect with a low dose of drug and irradiation, minimizing systemic toxicity.

Second-Near-Infrared Light-Induced SnSe Nanosheets for Augmented Photonic Tumor Hyperthermia
Xiuying Chen - ,
Haoqin Zheng - ,
Lile Dong *- , and
Peng Zhang *
Triple-negative breast cancer (TNBC) poses a serious threat to the lives and health of patients, as traditional treatments are limited by psychological burden, systemic toxicity, or drug resistance. Photonic hyperthermia triggered by the second near-infrared (NIR-II) window is a promising minimally invasive strategy. Herein, we synthesized SnSe nanosheets via a simple, scalable method for NIR-II-triggered photonic tumor hyperthermia. SnSe nanosheets exhibited irregular lamellar structures with uniform Sn/Se distribution, favorable biocompatibility, and prominent photothermal performance under NIR-II irradiation. Under NIR-II irradiation, the SnSe+NIR-II group significantly suppressed tumor growth. This work offers a platform based on NIR-II light-induced photonic tumor hyperthermia, providing a minimally invasive strategy for TNBC treatment.

One-Step Pulsed Electrodeposition of ZnO/ZnP Composite Coatings on Titanium Implants for Enhanced Antibacterial Activity and Biocompatibility
Junchong Chen - ,
Jionghong Liang - ,
Haojie Liu - ,
Aiyi Chen - ,
Haixing Feng - ,
Jiangwen Liu *- , and
Guie Xie *
Titanium implants are widely used in orthopedic and dental applications. However, bacterial adhesion and subsequent biofilm formation on Ti surfaces remain critical challenges, often leading to implant-associated infections. Although zinc oxide (ZnO) provides broad‑spectrum antibacterial activity, excessive Zn2+ release may induce cytotoxicity, and many zinc‑containing coatings still rely on complex multistep conversion routes or zinc‑based substrates. In this study, a ZnO/zinc-phosphate (ZnP) composite coating was successfully constructed on Ti substrates via a one-step pulse electrodeposition method. Compared with conventional multistep conversion routes, this method employs only two precursor salts and a single electrolyte bath to achieve rapid (15 min), one-step in situ deposition on Ti. By avoiding post-conversion reactions and repeated post-treatments, the process substantially simplifies the overall fabrication workflow. The composite achieves multifunctionality through a ZnO−ZnP synergy: ZnO delivers rapid antibacterial action, while the ZnP phase seals pores within the ZnO matrix and serves as a barrier, enabling controlled Zn2+ release (25% lower cumulative release at day 14). This densified composite structure delivered the highest corrosion resistance across all samples and promoted bioactivity, evidenced by dense apatite formation after immersion. In addition, ALP activity increased to 34.8 U/L at day 14 and CCK-8 assays showed the highest cell viability, demonstrating improved osteogenic potential and cytocompatibility. Furthermore, the coating showed effective antibacterial activity against Escherichia coli (E. coli) (96.2%) and Staphylococcus aureus (S. aureus) (89.8%), thereby effectively reducing the risk of clinical infections. This work provides a facile and efficient strategy for the one-step construction of multifunctional antibacterial coatings on metallic implants.
Additions and Corrections
Correction to "Nanofibers Embedded with Nanoparticles as Carriers for the Controlled Release of Anticancer Drug: Promoting the Apoptosis of Breast Cancer Cell Line and Growth Inhibition of Microbial Strains"
Edumpan Jaisankar - ,
Raja Sulaiman Azarudeen *- , and
Marimuthu Thirumarimurugan
This publication is free to access through this site. Learn More
Mastheads
Issue Editorial Masthead
This publication is free to access through this site. Learn More
Issue Publication Information
This publication is free to access through this site. Learn More
