
About the Cover:
Center Stage

This Bioengineer Transforms Food Waste into Haute Cuisine
Kristel Tjandra
This publication is Open Access under the license indicated. Learn More
The Hub

Dino Diets Revealed by Isotopes
Rachel Brazil
This publication is Open Access under the license indicated. Learn More
First Reactions
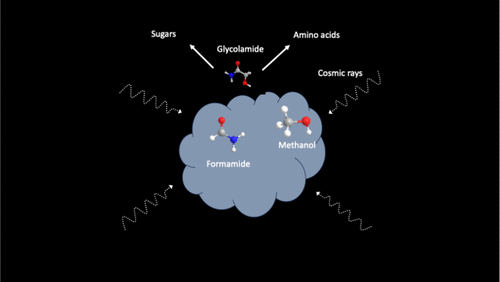
The Formation of Glycolamide on Interstellar Ices
Serena Viti
This publication is Open Access under the license indicated. Learn More
Outlooks

From Prompt to Drug: Toward Pharmaceutical Superintelligence
Alex Zhavoronkov *- ,
David Gennert - , and
Jiye Shi *
This publication is Open Access under the license indicated. Learn More
The convergence of generative artificial intelligence (AI) platforms and automated laboratory systems is ushering in a new era of drug discovery, in which a plain-language prompt can initiate a fully autonomous, end-to-end drug development program. This article explores the recent evolution of AI technologies and presents a “prompt-to-drug” pipeline, where AI not only generates novel hypotheses and designs optimized drug candidates but also orchestrates synthesis, validation, and clinical planning in a closed-loop system. By highlighting key breakthroughs, case studies, and the technological infrastructure required for this paradigm shift, we outline a vision for scalable, efficient, and unbiased drug discovery.

Asymmetric Catalysis: Recent Advances toward a More Sustainable Synthesis
Tanguy Saget - ,
Mohamed Mellah - ,
Philippe Dauban - , and
Emmanuelle Schulz *
This publication is Open Access under the license indicated. Learn More
Asymmetric catalysis is the most sustainable means of achieving efficient production of enantioenriched compounds, which are valuable to the pharmaceutical industry and advanced functionalized materials. The field has undergone considerable development since its inception about 60 years ago. The development of new chiral ligands or organocatalysts or the discovery of new reactions all led to spectacular progress in terms of activity and enantioselectivity. In this context, this Outlook article aims to highlight major advances that demonstrate the central role of asymmetric catalysis in sustainability, with the examples discussed representing a selection shaped by the authors’ scientific perspective and sensitivity. This includes recent innovations in catalyst recycling, the use of less precious and less toxic metals in asymmetric organometallic catalysis, as well as the control of the positioning of catalytic sites, to further improve their efficiency. The development of asymmetric electrocatalysis and photocatalysis as well as of mechanochemistry and continuous flow chemistry is also discussed. Finally, the complexity and step-economy issues are raised with the presentation of relevant examples of asymmetric multicatalytic processes and new challenging transformations that include late-stage functionalization reactions, molecular edition or reactions targeting the creation of axial, planar or helicoidal chirality.

Accelerated Emergence of Self-Driving Laboratories for Accelerating Materials Discovery
Amanda K. Brown - ,
Abhishek Soni - ,
Daniel Lin - , and
Curtis P. Berlinguette *
This publication is Open Access under the license indicated. Learn More
The rise of self-driving laboratories (SDLs) marks a transformative era in materials science.
By leveraging automation and machine learning, SDLs provide unprecedented opportunities to rapidly explore complex chemical landscapes and accelerate discoveries.
In this Outlook, we explore the growth of the global SDL ecosystem in materials science, describe recent advances, and discuss how SDLs are poised to drive more discoveries and accelerate commercialization.Articles
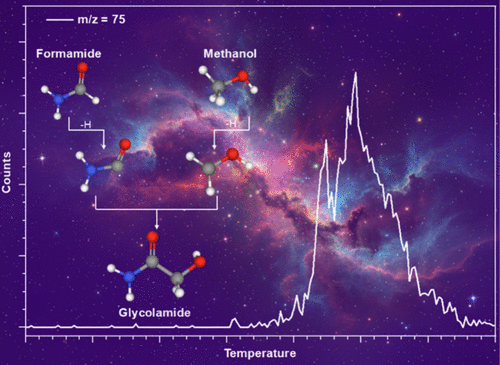
Nonequilibrium Synthesis of Glycolamide (NH2COCH2OH), a Precursor to Amino Acids, on Interstellar Nanoparticles
Alexandre Bergantini - ,
Jia Wang - ,
Ivan Antonov - ,
Evgenia A. Batrakova - ,
Sergey O. Tuchin - , and
Ralf I. Kaiser *
This publication is Open Access under the license indicated. Learn More
Complex organic molecules (COMs) are thought to form in cold interstellar environments, yet the chemical routes to key prebiotic precursors remain poorly understood. Glycine, the simplest amino acid, has not been detected in the interstellar medium so far, prompting interest in its structural isomer glycolamide, recently observed toward Sgr B2. Here, we provide the first experimental evidence that glycolamide forms efficiently at cryogenic temperatures via a barrierless carbon–carbon bond coupling between two carbon-centered carbamoyl (ĊONH2) and hydroxymethyl (ĊH2OH) radicals, generated from formamide and methanol in astrophysical ice analogues exposed to cosmic-ray proxies. This radical–radical pathway proceeds within the ices of ice-coated grains over typical dense cloud lifetimes (∼106 years), thus establishing a nonequilibrium mechanism to glycine isomers of astrobiological relevance. Once formed, glycolamide can act as a versatile precursor to amino acids and sugars thereby contributing to the molecular inventory inherited by nascent exoplanetary systems such as Fomalhaut along with Alpha Lyrae and our own.
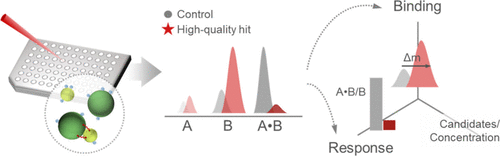
Integrating Functional Response and Target Binding for Mechanism-Centered Drug Screening by High-Mass MALDI-MS
Congrui Tan - ,
Yu Gao - ,
Marcus Buggert *- ,
Yuye Zhou *- , and
Renato Zenobi *
This publication is Open Access under the license indicated. Learn More
Early stage drug discovery is limited by the disjunction of function and binding assays, creating an information gap that leads to the high failure rate in hit advancement. This limitation is particularly pronounced for protein–protein interactions, whose large and shallow interfaces make it difficult to distinguish hits mechanistically. To address this, we developed a cross-linking matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS) platform that integrates biochemical functional response and target binding in a single assay, thereby generating a multidimensional pharmacological profile. Using the SARS-CoV-2 RBD–ACE2 interaction and a set of 17 drug candidates for a proof-of-concept study, the platform revealed a clear difference between two inhibitors that appeared indistinguishable in conventional functional assays: one showed stronger affinity and preferential ACE2 binding, while the other showed weaker and less specific binding. These mechanistic differences were consistent with the results of a cellular antiviral assay, in which only the high-affinity inhibitor improved cell viability. This work presents a mechanism-centered, rapid screening strategy that provides early multiparameter insight, enables rational selection of high-quality leads for challenging drug targets, and is compatible with high-throughput formats.

B-Alkyl-borabicyclo[3.3.1]nonane Reagents Promote Closed-Shell Nickel-Catalyzed Alkylarylation Toward Encoded Cyclooctene Monomers
Anne K. Ravn - ,
Aimee L. Bangerter - ,
Ethan M. Wagner - ,
Shijia Li - ,
Camille Z. Rubel - ,
Steven R. Wisniewski - ,
Peng Liu *- ,
Will. R. Gutekunst *- , and
Keary M. Engle *
This publication is Open Access under the license indicated. Learn More
Access to new tailored monomers is essential to explore unprecedented polymer structures and modulate their properties. In previous work, we disclosed an iteroselective diarylation of 1,5-cyclooctadiene as an attractive platform for preparing diverse cyclooctene monomers; however, it was limited to C(sp2) coupling partners. Established difunctionalization methods with C(sp3) fragments rely on carbon-based radical formation, which is incompatible with 1,5-cyclooctadiene. This work describes a two-electron redox manifold for the nickel-catalyzed alkylarylation of 1,5-cyclooctadiene and discloses B-alkyl-borabicyclo[3.3.1]nonane (alkyl-9-BBN) as an effective transmetalating reagent for maintaining a polar reaction mechanism. The method provides 5,6-alkylarylated cyclooctenes suitable for ring-opening metathesis polymerization to obtain new materials. The properties of these polymers are benchmarked and fine-tuned by variation of coupling partners in the nickel catalysis. Density functional theory calculations revealed that destabilization of the pretransmetalation complex promotes the reactivity of alkyl-9-BBN in transmetalation compared to alkylboronic esters.

Roblonski: A Material-Efficient Robo-Fluidic Toolbox for Rapid Photochemical Characterization
Azka Arshad - ,
Richard B. Canty - ,
Evgeny O. Danilov - ,
Milad Abolhasani *- , and
Felix N. Castellano *
This publication is Open Access under the license indicated. Learn More
Reliable photochemical and photophysical characterization is essential for understanding and optimizing photocatalytic processes; however, traditional, manual spectroscopic methods for determining bimolecular photoreaction quenching constants, molar extinction coefficients, and photoluminescence quantum yields (PLQYs) are time-, cost-, material-, and labor-intensive and generate considerable chemical waste. Herein, we report Roblonski, a compact, material-efficient microfluidic robotic platform that automates these three foundational photochemical assays with high precision, reproducibility, and accuracy. Using Ru(bpy)3(PF6)2 as a model photosensitizer and photocatalyst, we performed Stern–Volmer analyses with 11 excited state electron and triplet energy transfer quenchers, Beer–Lambert studies of five compounds spanning 3 orders of magnitude in their molar extinction coefficients across multiple solvents, and relative PLQY determinations for fluorophores/luminophores with PLQYs ranging over 3 orders of magnitude in efficiency. The machine-generated results matched manual experimental measurements and literature benchmarks across diverse spectral features, solvent environments, and signal intensity regimes. Roblonski reduces sample consumption (20-fold by solution volume, 1000-fold by reagent moles) and accelerates data collection (4-fold) compared to traditional, manual approaches. By integrating these photochemically relevant assays into a single, compact automated platform, Roblonski has the potential to lower experimental barriers, enable data-rich evaluation of photocatalysts and substrates, and augment autonomous photochemical discovery and characterization.

Higher-Level Strategies for Computer-Aided Retrosynthesis
Jihye Roh - ,
Joonyoung F. Joung - ,
Kevin Yu - ,
Zhengkai Tu - ,
G. Logan Bartholomew - ,
Omar A. Santiago-Reyes - ,
Mun Hong Fong - ,
Richmond Sarpong - ,
Sarah E. Reisman - , and
Connor W. Coley *
This publication is Open Access under the license indicated. Learn More
Retrosynthesis is a core technique in organic chemistry that simplifies target molecules into more readily available components. Computer-aided synthesis planning (CASP) automates this process by recursively proposing immediate precursors to identify multistep synthetic pathways. However, CASP typically struggles for complex molecules that require longer synthetic pathways and present a greater number of possible disconnections. Here, we introduce a new higher-level framework for computer-aided retrosynthesis. Our approach abstracts detailed substructures in pathway intermediates not appearing in the target product, allowing the algorithm to emphasize higher-level strategies while postponing the consideration of specific functional group choices, thus reducing the effective width and depth of the search space. This framework achieves higher top-k accuracy in single-step retrosynthesis and identifies multistep routes for more targets than the original approach. Through case studies on complex drugs and natural products, we demonstrate how routes proposed by our framework provide a powerful basis for developing full synthesis plans, particularly in challenging cases where the original approach fails, while enabling chemists to leverage their expertise to refine the synthesis design. Ultimately, focusing on higher-level strategies enables an effective and intuitive approach for challenging targets in computer-aided retrosynthesis.
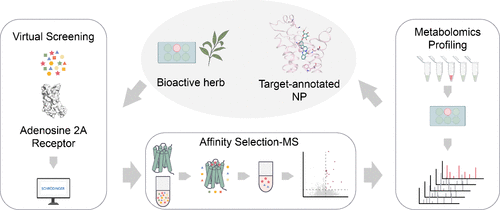
Target-Centric Multiplexed Screening of an Herbal Extract Identifies a Novel Dual A2A/A2B Receptor Antagonist for Cancer Immunotherapy
Hongyue Liu - ,
Xinyu Yang - ,
Jingyi Xu - ,
Yuefei Wang - ,
Zichen Zhao - ,
Ke Quan - ,
Aoqi Gao - ,
Yang Wang - ,
Jingbo Wu - ,
Fei Li - ,
Zhaoyu Zhang - ,
Yuanyuan Ma - ,
Yuan Weng - ,
Ying Chen - ,
Liping Sun - ,
Gaojie Song - ,
Yibing Shan - ,
Xin Chai *- ,
Bingjie Zhang *- ,
Weiqiang Lu *- , and
Wenqing Shui *
This publication is Open Access under the license indicated. Learn More
Medicinal herbs contain natural products (NPs) possessing rich scaffolds valuable for drug discovery, particularly in oncology. While most NP-derived cancer therapeutics directly kill tumor cells, emerging opportunities lie in modulating antitumor immunity. However, target-annotated NPs for cancer immunotherapy remain scarce. Herein we established a multiplexed platform combining virtual screening, affinity selection-mass spectrometry, and metabolomics profiling to identify bioactive NPs targeting the adenosine 2A receptor (A2AR), a master regulator of tumor immunosuppression. Screening the crude extract of a medicinal herb and isolating the active constituent resulted in the discovery of a novel dual antagonist for A2AR/A2BR with preferential activity on A2AR. This compound, ER-15, adopts a unique binding mode as revealed by structural modeling, MD simulations, mutagenesis, and SAR analysis. Functionally, ER-15 reversed adenosine-mediated immunosuppression and augmented the immune checkpoint inhibitor therapy in both the animal model and patient-derived tumor organoids, supporting its therapeutic potential in anti-PD-1-resistant tumors. Therefore, our strategy is expected to overcome traditional NP discovery bottlenecks, enabling efficient identification of target-annotated novel leads for drug development.
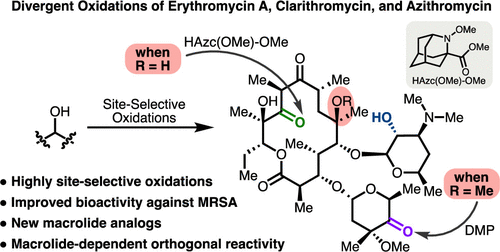
Site-Divergent Oxidations within Venerable Macrolide Antibiotic Scaffolds Unveil Compounds with Broad Spectrum and Anti-MRSA Activities
Olivia C. Langner - ,
Brandon Q. Mercado - ,
Sebastian M. Krajewski - ,
Song Lin - , and
Scott J. Miller *
This publication is Open Access under the license indicated. Learn More
The synthesis of bioactive compounds with differential, and ideally enhanced, activities presents persistent and growing challenges for the field of organic synthesis. By leveraging Nature’s ability to build complex, stereochemically rich, and biologically active molecular scaffolds, site-selective modification of natural products can deliver analogs without the need for lengthy de novo syntheses. Yet, achieving selective reactivity at a single desired position is complicated by the presence of multiple iterations of similar reactive functional groups, thus precluding widespread adoption of catalyst-controlled site-selective modification. Herein we describe the development of complementary systems for the oxidation of secondary alcohols on erythromycin A, clarithromycin, and azithromycin using a newly designed azaadamantyl oxoammonium catalyst, wherein different hydroxyl groups show disparate reactivities under the same conditions. The application of this methodology has enabled the generation of a suite of oxidized macrolide antibiotics and derivatives that take advantage of the newly installed carbonyls. Antimicrobial activity testing revealed that multiple compounds retain activity against a broad range of pathogens consistent with erythromycin A coverage. Additionally, three of the compounds reported herein display antibiotic activity against CA-MRSA and MRSA(mph(C)), for which the clinical analogs erythromycin A, clarithromycin, and azithromycin exhibit no activity at tested concentrations.
Additions and Corrections
Correction to “High-Performance Concurrent Chemo-Immuno-Radiotherapy for the Treatment of Hematologic Cancer through Selective High-Affinity Ligand Antibody Mimic-Functionalized Doxorubicin-Encapsulated Nanoparticles”
Kin Man Au - ,
Rod Balhorn - ,
Monique C. Balhorn - ,
Steven I. Park *- , and
Andrew Z. Wang *
This publication is Open Access under the license indicated. Learn More
Mastheads
Issue Editorial Masthead
This publication is Open Access under the license indicated. Learn More
Issue Publication Information
This publication is Open Access under the license indicated. Learn More
