Overcoming Synthetic Challenges and Evaluating in Vivo Efficacy of the MOG-Fc-Bifunctional Peptide Inhibitor for EAE in MiceClick to copy article linkArticle link copied!
- Rucha MahadikRucha MahadikDepartment of Pharmaceutical Chemistry, The University of Kansas, 2093 Constant Ave., Lawrence, Kansas 66047, United StatesMore by Rucha Mahadik
- Andrea L. Villela-NavaAndrea L. Villela-NavaDepartment of Pharmaceutical Chemistry, The University of Kansas, 2093 Constant Ave., Lawrence, Kansas 66047, United StatesMore by Andrea L. Villela-Nava
- Lun XinLun XinDepartment of Pharmaceutical Chemistry, The University of Kansas, 2093 Constant Ave., Lawrence, Kansas 66047, United StatesBioDev Department, WuXi Biologics USA, Cranbury, New Jersey 08512, United StatesMore by Lun Xin
- Teruna J. Siahaan*Teruna J. Siahaan*Email: [email protected]. Tel.: +1 785-864-7327.Department of Pharmaceutical Chemistry, The University of Kansas, 2093 Constant Ave., Lawrence, Kansas 66047, United StatesMore by Teruna J. Siahaan
- Thomas Tolbert*Thomas Tolbert*Email: [email protected]. Tel.: +1 785-864-1898.Department of Pharmaceutical Chemistry, The University of Kansas, 2093 Constant Ave., Lawrence, Kansas 66047, United StatesMore by Thomas Tolbert
Abstract
Multiple sclerosis (MS) is an autoimmune disease that causes neural degeneration as a result of the immune system launching an attack on the myelin sheath surrounding neurons. MS has multiple disease states; each one has been associated with a different onset pathway and requires a separate treatment. Primary progressive MS (PPMS) is a rare form of MS that affects 10–15% of MS patients, and Ocrelizumab is currently the only FDA-approved treatment on the market. While it can be effective in managing PPMS, Ocrelizumab can only delay the onset of the disease. In this study, MOG-Fc-BPI was designed as a potential therapeutic agent to suppress experimental autoimmune encephalomyelitis (EAE) in an antigen-specific manner, altering immune cells from an inflammatory to a regulatory phenotype. Here, MOG-Fc-BPI was successfully synthesized by conjugating the MOG-R5 peptide using sortase A enzyme to the C-terminus of the Fc-domain with LABL peptide at the N-terminus. Purified MOG-Fc-BPI was formulated to reach a concentration of 15 mg/mL for the in vivo study. MOG-stimulated EAE in C57BL/6 mice (a model for PPMS) that were treated with MOG-Fc-BPI on days 4 and 7 at 35 nmol/dose showed complete disease suppression on day 19 (score = 0; without symptoms) compared to PBS. The MOG-Fc-BPI-treated mice showed increased body weights throughout the study, while PBS-treated mice lost around 10% bodyweight during the peak of the disease without recovery up to the end of the study. Overall, this study provided a proof-of-concept that MOG-Fc-BPI has the potential to suppress PPMS.
This publication is licensed for personal use by The American Chemical Society.
Introduction
Figure 1
Figure 1. Structure of MOG-Fc-BPI consists of an Fc-domain with the LABL peptide at the N-terminus and the MOG-R5 peptide at the C-terminus.
Materials and Methods
Expression and Purification of LABL-Fc-ST
Figure 2
Figure 2. MS spectra of the purified LABL-Fc-ST protein. (A) Intact-MS spectrum of the reduced and deglycosylated LABL-Fc-ST protein with [M + H]: 27,024 amu. (B) The MS spectrum of fully glycosylated LABL-Fc-ST with the Man8 glycan (expected [M + Na]: 28,749 amu; observed mass: 28,749 amu) as well as the Man9 glycan (expected [M + Na]: 28,910 amu; observed mass: 28,911 amu).
Mass Spectrometry Analysis
Synthesis and Purification of the MOG Peptide
Figure 3
Figure 3. MS spectrum of G3MOGR5 peptide showed peaks for [M + 5H] (512.18 m/z), [M + 4H] (639.98 m/z), and [M + 3H] (853.09 m/z) with an overall observed mass of 2555.9 amu, which was similar to the expected mass of 2555.94 amu.
Sortase-Mediated Ligation with LABL-Fc-ST and MOG Peptide
Efficacy of MOG-Fc-BPI in EAE Mice
Results
Synthesis of MOG-Fc-BPI
Sortase-Mediated Ligation
Figure 4
Figure 4. Synthetic scheme for the production of MOG-Fc-BPI. LABL-Fc-ST contains the C-terminal sortase recognition tag, LPETGGG. The G3-MOG-R5 peptide was conjugated to the C-terminus of LABL-Fc-ST using sortase A, which recognizes the LPETGGG sequence and cleaves between the threonine and glycine residues to form a thioester with threonine while releasing the C-terminal triglycine peptide. The N-terminal triglycine residues of G3-MOG-R5 act as a nucleophile in the sortase A reaction to attack the thioester and produce the desired product, MOG-Fc-BPI.
Figure 5
Figure 5. MS spectra of MOG-Fc-BPI. (A) The intact-MS spectrum of the reduced and deglycosylated MOG-Fc-BPI protein with [M + H] = 29,391 amu. (B) MS spectrum of purified glycosylated MOG-Fc-BPI with major species such as (a) Man8 glycoform of MOG-Fc-BPI (expected = 31,093.8 amu, observed = 31,092.8 amu); (b) MOG-Fc-BPI with no glycan (expected = 29,372.3 amu, observed = 29,392.1 amu); and (c) LABL-Fc-ST (expected = 28,726.9 amu, observed = 28,726.7 amu).
MOG-Fc-BPI Formulation
Animal Studies
Figure 6
Figure 6. Activity of MOG-Fc-BPI to suppress EAE was determined by comparing (A) daily clinical scores, (B) AUC of clinical scores, (C) daily body weights, and (D) AUC of body weights to those of PBS control. (A) The MOG-Fc-BPI-treated mice received 2 doses of 35 nmol/dose injections on days 4 and 7 and showed lower clinical scores than those of PBS-treated mice. (B) AUC of clinical scores for PBS-treated mice was significantly higher than that of MOG-Fc-BPI-treated mice over 25 days (p < 0.001). (C) The differences in the increase body weights over 25 day period for MOG-Fc-BPI- and PBS-treated mice. There is inhibition in the increase of body weights on days 14–25 in PBS-treated mice but not MOG-Fc-BPI-treated mice. (D) A significantly higher ACU of body weights of MOG-Fc-BPI-treated mice compared to PBS-treated mice (p = 0.0274).
Discussion
Figure 7
Figure 7. Proposed mechanisms of action for MOG-Fc-BPI suppression of EAE disease involving iDC and mDC. (A) MOG-Fc-BPI binds simultaneously to ICAM-1 and MHC-II using LABL and MOG peptides, respectively, on iDC to block Signal-2 in the presence of Signal-1. and this also prevents immunological synapse (IS) formation. The presence of Signal-1 in the absence of Signal-2 causes naïve T cells to become Treg cells. (B) MOG-Fc-BPI binds to ICAM-1 and MHC-II on the surface of mDC. The TCRs on naïve T cells recognize the MOG/MHC-II complexes (Signal-1) with blocked Signal-2; as a result, this prevents the differentiation of naïve T cells to inflammatory Th17 or Th1 cells.
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.bioconjchem.5c00518.
Procedure to express and purify the LABL-Fc-ST protein as a starting material; experimental method to produce sortase A enzyme; WES capillary cartridge analysis of the MOG-Fc-BPI molecule in reduced and intact forms (Figure S1) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We acknowledge the support for RM from the National Institutes of Health (NIH) Graduate Training at the Biology-Chemistry Interface Grant T32 GM132061 from the National Institutes of General Medical Sciences. We also acknowledge grant supports to T.J.S. and T.T. from the National Institutes of Health, including R01-AG082273 (NIA), R01-AG071682 (NIA), and P20-GM113117 (Pilot Grant, COBRE Chemical Biology Infectious Disease, NIGMS).
| CNS | central nervous system |
| EAE | experimental autoimmune encephalomyelitis |
| MS | multiple sclerosis (MS) |
| MBP | myelin basic protein |
| MOG | myelin oligodendrocyte glycoprotein |
| PLP | proteolipid protein (PLP) |
| PPMS | primary progressive MS |
| PRMS | progressive relapsing MS |
| RRMS | relapse-remitting MS |
| SPMS | secondary progressive MS |
References
This article references 45 other publications.
- 1Brandstadter, R.; Katz Sand, I. The use of natalizumab for multiple sclerosis. Neuropsychiatr Dis Treat 2017, 13, 1691– 1702, DOI: 10.2147/NDT.S114636Google ScholarThere is no corresponding record for this reference.
- 2Mahadik, R.; Kiptoo, P.; Tolbert, T.; Siahaan, T. J. Immune Modulation by Antigenic Peptides and Antigenic Peptide Conjugates for Treatment of Multiple Sclerosis. Med. Res. Arch. 2022, 10 (5), 2804, DOI: 10.18103/mra.v10i5.2804Google ScholarThere is no corresponding record for this reference.
- 3Ghasemi, N.; Razavi, S.; Nikzad, E. Multiple Sclerosis: Pathogenesis, Symptoms, Diagnoses and Cell-Based Therapy. Cell J. 2017, 19 (1), 1– 10, DOI: 10.22074/cellj.2016.4867Google ScholarThere is no corresponding record for this reference.
- 4Kammona, O.; Kiparissides, C. Recent Advances in Antigen-Specific Immunotherapies for the Treatment of Multiple Sclerosis. Brain Sci. 2020, 10 (6), 333, DOI: 10.3390/brainsci10060333Google ScholarThere is no corresponding record for this reference.
- 5Manikwar, P.; Kiptoo, P.; Badawi, A. H.; Buyuktimkin, B.; Siahaan, T. J. Antigen-specific blocking of CD4-specific immunological synapse formation using BPI and current therapies for autoimmune diseases. Med. Res. Rev. 2012, 32 (4), 727– 764, DOI: 10.1002/med.20243Google ScholarThere is no corresponding record for this reference.
- 6Badawi, A. H.; Siahaan, T. J. Immune modulating peptides for the treatment and suppression of multiple sclerosis. Clin Immunol 2012, 144 (2), 127– 138, DOI: 10.1016/j.clim.2012.05.010Google ScholarThere is no corresponding record for this reference.
- 7Moral, M. E. G.; Siahaan, T. J. Conjugates of Cell Adhesion Peptides for Therapeutics and Diagnostics Against Cancer and Autoimmune Diseases. Curr. Top Med. Chem. 2018, 17 (32), 3425– 3443, DOI: 10.2174/1568026618666180118154514Google ScholarThere is no corresponding record for this reference.
- 8Kobayashi, N.; Kiptoo, P.; Kobayashi, H.; Ridwan, R.; Brocke, S.; Siahaan, T. J. Prophylactic and therapeutic suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. Clin Immunol 2008, 129 (1), 69– 79, DOI: 10.1016/j.clim.2008.06.002Google ScholarThere is no corresponding record for this reference.
- 9Kobayashi, N.; Kobayashi, H.; Gu, L.; Malefyt, T.; Siahaan, T. J. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. J. Pharmacol Exp Ther 2007, 322 (2), 879– 886, DOI: 10.1124/jpet.107.123257Google ScholarThere is no corresponding record for this reference.
- 10Badawi, A. H.; Siahaan, T. J. Suppression of MOG- and PLP-induced experimental autoimmune encephalomyelitis using a novel multivalent bifunctional peptide inhibitor. J. Neuroimmunol 2013, 263 (1–2), 20– 27, DOI: 10.1016/j.jneuroim.2013.07.009Google ScholarThere is no corresponding record for this reference.
- 11Murray, J. S.; Oney, S.; Page, J. E.; Kratochvil-Stava, A.; Hu, Y.; Makagiansar, I. T.; Brown, J. C.; Kobayashi, N.; Siahaan, T. J. Suppression of type 1 diabetes in NOD mice by bifunctional peptide inhibitor: modulation of the immunological synapse formation. Chem. Biol. Drug Des 2007, 70 (3), 227– 236, DOI: 10.1111/j.1747-0285.2007.00552.xGoogle ScholarThere is no corresponding record for this reference.
- 12Buyuktimkin, B.; Kiptoo, P.; Siahaan, T. J. Bifunctional Peptide Inhibitors Suppress Interleukin-6 Proliferation and Ameliorates Murine Collagen-Induced Arthritis. J. Clin. Cell. Immunol. 2014, 5 (6), 1000273 DOI: 10.4172/2155-9899.1000273Google ScholarThere is no corresponding record for this reference.
- 13Ridwan, R.; Kiptoo, P.; Kobayashi, N.; Weir, S.; Hughes, M.; Williams, T.; Soegianto, R.; Siahaan, T. J. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor: structure optimization and pharmacokinetics. J. Pharmacol Exp Ther 2010, 332 (3), 1136– 1145, DOI: 10.1124/jpet.109.161109Google ScholarThere is no corresponding record for this reference.
- 14White, D. R.; Khedri, Z.; Kiptoo, P.; Siahaan, T. J.; Tolbert, T. J. Synthesis of a Bifunctional Peptide Inhibitor-IgG1 Fc Fusion That Suppresses Experimental Autoimmune Encephalomyelitis. Bioconjug Chem. 2017, 28 (7), 1867– 1877, DOI: 10.1021/acs.bioconjchem.7b00175Google ScholarThere is no corresponding record for this reference.
- 15Xiao, J.; Chen, R.; Pawlicki, M. A.; Tolbert, T. J. Targeting a homogeneously glycosylated antibody Fc to bind cancer cells using a synthetic receptor ligand. J. Am. Chem. Soc. 2009, 131 (38), 13616– 13618, DOI: 10.1021/ja9045179Google ScholarThere is no corresponding record for this reference.
- 16Zou, Z.; Ji, Y.; Schwaneberg, U. Empowering Site-Specific Bioconjugations In Vitro and In Vivo: Advances in Sortase Engineering and Sortase-Mediated Ligation. Angew. Chem., Int. Ed. Engl. 2024, 63 (12), e202310910 DOI: 10.1002/anie.202310910Google ScholarThere is no corresponding record for this reference.
- 17Antos, J. M.; Truttmann, M. C.; Ploegh, H. L. Recent advances in sortase-catalyzed ligation methodology. Curr. Opin Struct Biol. 2016, 38, 111– 118, DOI: 10.1016/j.sbi.2016.05.021Google ScholarThere is no corresponding record for this reference.
- 18Heistinger, L.; Gasser, B.; Mattanovich, D. Microbe Profile: Komagataella phaffii: a methanol devouring biotech yeast formerly known as Pichia pastoris. Microbiology (Reading) 2020, 166 (7), 614– 616, DOI: 10.1099/mic.0.000958Google ScholarThere is no corresponding record for this reference.
- 19Miller, D. H.; Leary, S. M. Primary-progressive multiple sclerosis. Lancet Neurol 2007, 6 (10), 903– 912, DOI: 10.1016/S1474-4422(07)70243-0Google ScholarThere is no corresponding record for this reference.
- 20Lamb, Y. N. Ocrelizumab: A Review in Multiple Sclerosis. Drugs 2022, 82 (3), 323– 334, DOI: 10.1007/s40265-022-01672-9Google ScholarThere is no corresponding record for this reference.
- 21Hauser, S. L.; Kappos, L.; Montalban, X.; Craveiro, L.; Chognot, C.; Hughes, R.; Koendgen, H.; Pasquarelli, N.; Pradhan, A.; Prajapati, K. Safety of Ocrelizumab in Patients With Relapsing and Primary Progressive Multiple Sclerosis. Neurology 2021, 97 (16), e1546– e1559, DOI: 10.1212/WNL.0000000000012700Google ScholarThere is no corresponding record for this reference.
- 22Kiptoo, P.; Buyuktimkin, B.; Badawi, A. H.; Stewart, J.; Ridwan, R.; Siahaan, T. J. Controlling immune response and demyelination using highly potent bifunctional peptide inhibitors in the suppression of experimental autoimmune encephalomyelitis. Clin. Exp. Immunol. 2013, 172 (1), 23– 36, DOI: 10.1111/cei.12029Google ScholarThere is no corresponding record for this reference.
- 23Badawi, A. H.; Kiptoo, P.; Wang, W. T.; Choi, I. Y.; Lee, P.; Vines, C. M.; Siahaan, T. J. Suppression of EAE and prevention of blood-brain barrier breakdown after vaccination with novel bifunctional peptide inhibitor. Neuropharmacology 2012, 62 (4), 1874– 1881, DOI: 10.1016/j.neuropharm.2011.12.013Google ScholarThere is no corresponding record for this reference.
- 24Littman, D. R.; Rudensky, A. Y. Th17 and regulatory T cells in mediating and restraining inflammation. Cell 2010, 140 (6), 845– 858, DOI: 10.1016/j.cell.2010.02.021Google ScholarThere is no corresponding record for this reference.
- 25Moorman, C. D.; Bastian, A. G.; DeOca, K. B.; Mannie, M. D. A GM-CSF-neuroantigen tolerogenic vaccine elicits inefficient antigen recognition events below the CD40L triggering threshold to expand CD4(+) CD25(+) FOXP3(+) Tregs that inhibit experimental autoimmune encephalomyelitis (EAE). J. Neuroinflammation 2020, 17 (1), 180, DOI: 10.1186/s12974-020-01856-8Google ScholarThere is no corresponding record for this reference.
- 26Gagliani, N.; Amezcua Vesely, M. C.; Iseppon, A.; Brockmann, L.; Xu, H.; Palm, N. W.; de Zoete, M. R.; Licona-Limon, P.; Paiva, R. S.; Ching, T. Th17 cells transdifferentiate into regulatory T cells during resolution of inflammation. Nature 2015, 523 (7559), 221– 225, DOI: 10.1038/nature14452Google ScholarThere is no corresponding record for this reference.
- 27Mari, E. R.; Rasouli, J.; Ciric, B.; Moore, J. N.; Conejo-Garcia, J. R.; Rajasagi, N.; Zhang, G. X.; Rabinovich, G. A.; Rostami, A. Galectin-1 is essential for the induction of MOG35–55 -based intravenous tolerance in experimental autoimmune encephalomyelitis. Eur. J. Immunol. 2016, 46 (7), 1783– 1796, DOI: 10.1002/eji.201546212Google ScholarThere is no corresponding record for this reference.
- 28Leno-Duran, E.; Ng, S. L.; Strominger, J. L. Regulation of EAE by spontaneously generated IL-10-secreting regulatory T cells in HLA-DR15/TCR.Ob1A12 double transgenic mice. Immunology 2021, 163 (3), 338– 343, DOI: 10.1111/imm.13321Google ScholarThere is no corresponding record for this reference.
- 29Raverdeau, M.; Christofi, M.; Malara, A.; Wilk, M. M.; Misiak, A.; Kuffova, L.; Yu, T.; McGinley, A. M.; Quinn, S. M.; Massilamany, C. Retinoic acid-induced autoantigen-specific type 1 regulatory T cells suppress autoimmunity. EMBO Rep. 2019, 20 (5), e47121 DOI: 10.15252/embr.201847121Google ScholarThere is no corresponding record for this reference.
- 30Loos, J.; Schmaul, S.; Noll, T. M.; Paterka, M.; Schillner, M.; Loffel, J. T.; Zipp, F.; Bittner, S. Functional characteristics of Th1, Th17, and ex-Th17 cells in EAE revealed by intravital two-photon microscopy. J. Neuroinflammation 2020, 17 (1), 357, DOI: 10.1186/s12974-020-02021-xGoogle ScholarThere is no corresponding record for this reference.
- 31Xu, H.; Agalioti, T.; Zhao, J.; Steglich, B.; Wahib, R.; Vesely, M. C. A.; Bielecki, P.; Bailis, W.; Jackson, R.; Perez, D. The induction and function of the anti-inflammatory fate of T(H)17 cells. Nat. Commun. 2020, 11 (1), 3334, DOI: 10.1038/s41467-020-17097-5Google ScholarThere is no corresponding record for this reference.
- 32Worzner, K.; Zimmermann, J.; Buhl, R.; Desoi, A.; Christensen, D.; Dietrich, J.; Nguyen, N.; Lindenstrom, T.; Woodworth, J. S.; Alhakeem, R. S. Repeated immunization with ATRA-containing liposomal adjuvant transdifferentiates Th17 cells to a Tr1-like phenotype. J. Autoimmun 2024, 144, 103174 DOI: 10.1016/j.jaut.2024.103174Google ScholarThere is no corresponding record for this reference.
- 33Vieira, P. L.; Christensen, J. R.; Minaee, S.; O’Neill, E. J.; Barrat, F. J.; Boonstra, A.; Barthlott, T.; Stockinger, B.; Wraith, D. C.; O’Garra, A. IL-10-secreting regulatory T cells do not express Foxp3 but have comparable regulatory function to naturally occurring CD4+CD25+ regulatory T cells. J. Immunol 2004, 172 (10), 5986– 5993, DOI: 10.4049/jimmunol.172.10.5986Google ScholarThere is no corresponding record for this reference.
- 34Maimone, D.; Guazzi, G. C.; Annunziata, P. IL-6 detection in multiple sclerosis brain. J. Neurol Sci. 1997, 146 (1), 59– 65, DOI: 10.1016/S0022-510X(96)00283-3Google ScholarThere is no corresponding record for this reference.Kebir, H.; Ifergan, I.; Alvarez, J. I.; Bernard, M.; Poirier, J.; Arbour, N.; Duquette, P.; Prat, A. Preferential recruitment of interferon-gamma-expressing TH17 cells in multiple sclerosis. Ann. Neurol. 2009, 66 (3), 390– 402, DOI: 10.1002/ana.21748Google ScholarThere is no corresponding record for this reference.
- 35Prendergast, C. T.; Anderton, S. M. Immune cell entry to central nervous system--current understanding and prospective therapeutic targets. Endocr Metab Immune Disord Drug Targets 2009, 9 (4), 315– 327, DOI: 10.2174/187153009789839219Google ScholarThere is no corresponding record for this reference.
- 36Lin, W.; Kemper, A.; Dupree, J. L.; Harding, H. P.; Ron, D.; Popko, B. Interferon-gamma inhibits central nervous system remyelination through a process modulated by endoplasmic reticulum stress. Brain 2006, 129 (Pt 5), 1306– 1318, DOI: 10.1093/brain/awl044Google ScholarThere is no corresponding record for this reference.
- 37Buyuktimkin, B.; Manikwar, P.; Kiptoo, P. K.; Badawi, A. H.; Stewart, J. M., Jr; Siahaan, T. J. Vaccinelike and prophylactic treatments of EAE with novel I-domain antigen conjugates (IDAC): targeting multiple antigenic peptides to APC. Mol. Pharm. 2013, 10 (1), 297– 306, DOI: 10.1021/mp300440xGoogle ScholarThere is no corresponding record for this reference.
- 38Martin, T. R.; Galli, S. J.; Katona, I. M.; Drazen, J. M. Role of mast cells in anaphylaxis. Evidence for the importance of mast cells in the cardiopulmonary alterations and death induced by anti-IgE in mice. J. Clin Invest 1989, 83 (4), 1375– 1383, DOI: 10.1172/JCI114025Google ScholarThere is no corresponding record for this reference.
- 39Smith, C. E.; Eagar, T. N.; Strominger, J. L.; Miller, S. D. Differential induction of IgE-mediated anaphylaxis after soluble vs. cell-bound tolerogenic peptide therapy of autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. U. S. A. 2005, 102 (27), 9595– 9600, DOI: 10.1073/pnas.0504131102Google ScholarThere is no corresponding record for this reference.
- 40Miller, S. D.; Turley, D. M.; Podojil, J. R. Antigen-specific tolerance strategies for the prevention and treatment of autoimmune disease. Nat. Rev. Immunol 2007, 7 (9), 665– 677, DOI: 10.1038/nri2153Google ScholarThere is no corresponding record for this reference.
- 41Sabatos-Peyton, C. A.; Verhagen, J.; Wraith, D. C. Antigen-specific immunotherapy of autoimmune and allergic diseases. Curr. Opin Immunol 2010, 22 (5), 609– 615, DOI: 10.1016/j.coi.2010.08.006Google ScholarThere is no corresponding record for this reference.
- 42Pirzer, T.; Becher, K. S.; Rieker, M.; Meckel, T.; Mootz, H. D.; Kolmar, H. Generation of Potent Anti-HER1/2 Immunotoxins by Protein Ligation Using Split Inteins. ACS Chem. Biol. 2018, 13 (8), 2058– 2066, DOI: 10.1021/acschembio.8b00222Google ScholarThere is no corresponding record for this reference.
- 43Ludwig, C.; Pfeiff, M.; Linne, U.; Mootz, H. D. Ligation of a synthetic peptide to the N terminus of a recombinant protein using semisynthetic protein trans-splicing. Angew. Chem., Int. Ed. Engl. 2006, 45 (31), 5218– 5221, DOI: 10.1002/anie.200600570Google ScholarThere is no corresponding record for this reference.
- 44Manikwar, P.; Tejo, B. A.; Shinogle, H.; Moore, D. S.; Zimmerman, T.; Blanco, F.; Siahaan, T. J. Utilization of I-domain of LFA-1 to Target Drug and Marker Molecules to Leukocytes. Theranostics 2011, 1, 277– 289, DOI: 10.7150/thno/v01p0277Google ScholarThere is no corresponding record for this reference.
- 45Manikwar, P.; Buyuktimkin, B.; Kiptoo, P.; Badawi, A. H.; Galeva, N. A.; Williams, T. D.; Siahaan, T. J. I-domain-antigen conjugate (IDAC) for delivering antigenic peptides to APC: synthesis, characterization, and in vivo EAE suppression. Bioconjug Chem. 2012, 23 (3), 509– 517, DOI: 10.1021/bc200580jGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1

Figure 1. Structure of MOG-Fc-BPI consists of an Fc-domain with the LABL peptide at the N-terminus and the MOG-R5 peptide at the C-terminus.
Figure 2
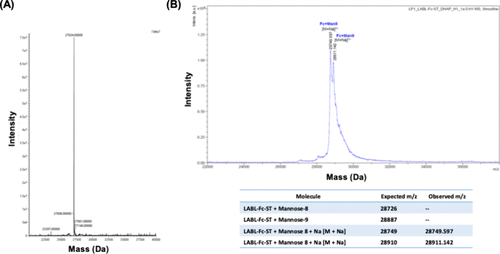
Figure 2. MS spectra of the purified LABL-Fc-ST protein. (A) Intact-MS spectrum of the reduced and deglycosylated LABL-Fc-ST protein with [M + H]: 27,024 amu. (B) The MS spectrum of fully glycosylated LABL-Fc-ST with the Man8 glycan (expected [M + Na]: 28,749 amu; observed mass: 28,749 amu) as well as the Man9 glycan (expected [M + Na]: 28,910 amu; observed mass: 28,911 amu).
Figure 3

Figure 3. MS spectrum of G3MOGR5 peptide showed peaks for [M + 5H] (512.18 m/z), [M + 4H] (639.98 m/z), and [M + 3H] (853.09 m/z) with an overall observed mass of 2555.9 amu, which was similar to the expected mass of 2555.94 amu.
Figure 4

Figure 4. Synthetic scheme for the production of MOG-Fc-BPI. LABL-Fc-ST contains the C-terminal sortase recognition tag, LPETGGG. The G3-MOG-R5 peptide was conjugated to the C-terminus of LABL-Fc-ST using sortase A, which recognizes the LPETGGG sequence and cleaves between the threonine and glycine residues to form a thioester with threonine while releasing the C-terminal triglycine peptide. The N-terminal triglycine residues of G3-MOG-R5 act as a nucleophile in the sortase A reaction to attack the thioester and produce the desired product, MOG-Fc-BPI.
Figure 5
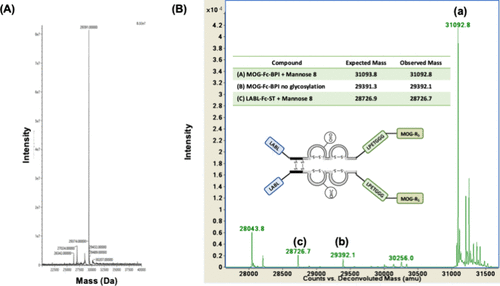
Figure 5. MS spectra of MOG-Fc-BPI. (A) The intact-MS spectrum of the reduced and deglycosylated MOG-Fc-BPI protein with [M + H] = 29,391 amu. (B) MS spectrum of purified glycosylated MOG-Fc-BPI with major species such as (a) Man8 glycoform of MOG-Fc-BPI (expected = 31,093.8 amu, observed = 31,092.8 amu); (b) MOG-Fc-BPI with no glycan (expected = 29,372.3 amu, observed = 29,392.1 amu); and (c) LABL-Fc-ST (expected = 28,726.9 amu, observed = 28,726.7 amu).
Figure 6

Figure 6. Activity of MOG-Fc-BPI to suppress EAE was determined by comparing (A) daily clinical scores, (B) AUC of clinical scores, (C) daily body weights, and (D) AUC of body weights to those of PBS control. (A) The MOG-Fc-BPI-treated mice received 2 doses of 35 nmol/dose injections on days 4 and 7 and showed lower clinical scores than those of PBS-treated mice. (B) AUC of clinical scores for PBS-treated mice was significantly higher than that of MOG-Fc-BPI-treated mice over 25 days (p < 0.001). (C) The differences in the increase body weights over 25 day period for MOG-Fc-BPI- and PBS-treated mice. There is inhibition in the increase of body weights on days 14–25 in PBS-treated mice but not MOG-Fc-BPI-treated mice. (D) A significantly higher ACU of body weights of MOG-Fc-BPI-treated mice compared to PBS-treated mice (p = 0.0274).
Figure 7

Figure 7. Proposed mechanisms of action for MOG-Fc-BPI suppression of EAE disease involving iDC and mDC. (A) MOG-Fc-BPI binds simultaneously to ICAM-1 and MHC-II using LABL and MOG peptides, respectively, on iDC to block Signal-2 in the presence of Signal-1. and this also prevents immunological synapse (IS) formation. The presence of Signal-1 in the absence of Signal-2 causes naïve T cells to become Treg cells. (B) MOG-Fc-BPI binds to ICAM-1 and MHC-II on the surface of mDC. The TCRs on naïve T cells recognize the MOG/MHC-II complexes (Signal-1) with blocked Signal-2; as a result, this prevents the differentiation of naïve T cells to inflammatory Th17 or Th1 cells.
References
This article references 45 other publications.
- 1Brandstadter, R.; Katz Sand, I. The use of natalizumab for multiple sclerosis. Neuropsychiatr Dis Treat 2017, 13, 1691– 1702, DOI: 10.2147/NDT.S114636There is no corresponding record for this reference.
- 2Mahadik, R.; Kiptoo, P.; Tolbert, T.; Siahaan, T. J. Immune Modulation by Antigenic Peptides and Antigenic Peptide Conjugates for Treatment of Multiple Sclerosis. Med. Res. Arch. 2022, 10 (5), 2804, DOI: 10.18103/mra.v10i5.2804There is no corresponding record for this reference.
- 3Ghasemi, N.; Razavi, S.; Nikzad, E. Multiple Sclerosis: Pathogenesis, Symptoms, Diagnoses and Cell-Based Therapy. Cell J. 2017, 19 (1), 1– 10, DOI: 10.22074/cellj.2016.4867There is no corresponding record for this reference.
- 4Kammona, O.; Kiparissides, C. Recent Advances in Antigen-Specific Immunotherapies for the Treatment of Multiple Sclerosis. Brain Sci. 2020, 10 (6), 333, DOI: 10.3390/brainsci10060333There is no corresponding record for this reference.
- 5Manikwar, P.; Kiptoo, P.; Badawi, A. H.; Buyuktimkin, B.; Siahaan, T. J. Antigen-specific blocking of CD4-specific immunological synapse formation using BPI and current therapies for autoimmune diseases. Med. Res. Rev. 2012, 32 (4), 727– 764, DOI: 10.1002/med.20243There is no corresponding record for this reference.
- 6Badawi, A. H.; Siahaan, T. J. Immune modulating peptides for the treatment and suppression of multiple sclerosis. Clin Immunol 2012, 144 (2), 127– 138, DOI: 10.1016/j.clim.2012.05.010There is no corresponding record for this reference.
- 7Moral, M. E. G.; Siahaan, T. J. Conjugates of Cell Adhesion Peptides for Therapeutics and Diagnostics Against Cancer and Autoimmune Diseases. Curr. Top Med. Chem. 2018, 17 (32), 3425– 3443, DOI: 10.2174/1568026618666180118154514There is no corresponding record for this reference.
- 8Kobayashi, N.; Kiptoo, P.; Kobayashi, H.; Ridwan, R.; Brocke, S.; Siahaan, T. J. Prophylactic and therapeutic suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. Clin Immunol 2008, 129 (1), 69– 79, DOI: 10.1016/j.clim.2008.06.002There is no corresponding record for this reference.
- 9Kobayashi, N.; Kobayashi, H.; Gu, L.; Malefyt, T.; Siahaan, T. J. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. J. Pharmacol Exp Ther 2007, 322 (2), 879– 886, DOI: 10.1124/jpet.107.123257There is no corresponding record for this reference.
- 10Badawi, A. H.; Siahaan, T. J. Suppression of MOG- and PLP-induced experimental autoimmune encephalomyelitis using a novel multivalent bifunctional peptide inhibitor. J. Neuroimmunol 2013, 263 (1–2), 20– 27, DOI: 10.1016/j.jneuroim.2013.07.009There is no corresponding record for this reference.
- 11Murray, J. S.; Oney, S.; Page, J. E.; Kratochvil-Stava, A.; Hu, Y.; Makagiansar, I. T.; Brown, J. C.; Kobayashi, N.; Siahaan, T. J. Suppression of type 1 diabetes in NOD mice by bifunctional peptide inhibitor: modulation of the immunological synapse formation. Chem. Biol. Drug Des 2007, 70 (3), 227– 236, DOI: 10.1111/j.1747-0285.2007.00552.xThere is no corresponding record for this reference.
- 12Buyuktimkin, B.; Kiptoo, P.; Siahaan, T. J. Bifunctional Peptide Inhibitors Suppress Interleukin-6 Proliferation and Ameliorates Murine Collagen-Induced Arthritis. J. Clin. Cell. Immunol. 2014, 5 (6), 1000273 DOI: 10.4172/2155-9899.1000273There is no corresponding record for this reference.
- 13Ridwan, R.; Kiptoo, P.; Kobayashi, N.; Weir, S.; Hughes, M.; Williams, T.; Soegianto, R.; Siahaan, T. J. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor: structure optimization and pharmacokinetics. J. Pharmacol Exp Ther 2010, 332 (3), 1136– 1145, DOI: 10.1124/jpet.109.161109There is no corresponding record for this reference.
- 14White, D. R.; Khedri, Z.; Kiptoo, P.; Siahaan, T. J.; Tolbert, T. J. Synthesis of a Bifunctional Peptide Inhibitor-IgG1 Fc Fusion That Suppresses Experimental Autoimmune Encephalomyelitis. Bioconjug Chem. 2017, 28 (7), 1867– 1877, DOI: 10.1021/acs.bioconjchem.7b00175There is no corresponding record for this reference.
- 15Xiao, J.; Chen, R.; Pawlicki, M. A.; Tolbert, T. J. Targeting a homogeneously glycosylated antibody Fc to bind cancer cells using a synthetic receptor ligand. J. Am. Chem. Soc. 2009, 131 (38), 13616– 13618, DOI: 10.1021/ja9045179There is no corresponding record for this reference.
- 16Zou, Z.; Ji, Y.; Schwaneberg, U. Empowering Site-Specific Bioconjugations In Vitro and In Vivo: Advances in Sortase Engineering and Sortase-Mediated Ligation. Angew. Chem., Int. Ed. Engl. 2024, 63 (12), e202310910 DOI: 10.1002/anie.202310910There is no corresponding record for this reference.
- 17Antos, J. M.; Truttmann, M. C.; Ploegh, H. L. Recent advances in sortase-catalyzed ligation methodology. Curr. Opin Struct Biol. 2016, 38, 111– 118, DOI: 10.1016/j.sbi.2016.05.021There is no corresponding record for this reference.
- 18Heistinger, L.; Gasser, B.; Mattanovich, D. Microbe Profile: Komagataella phaffii: a methanol devouring biotech yeast formerly known as Pichia pastoris. Microbiology (Reading) 2020, 166 (7), 614– 616, DOI: 10.1099/mic.0.000958There is no corresponding record for this reference.
- 19Miller, D. H.; Leary, S. M. Primary-progressive multiple sclerosis. Lancet Neurol 2007, 6 (10), 903– 912, DOI: 10.1016/S1474-4422(07)70243-0There is no corresponding record for this reference.
- 20Lamb, Y. N. Ocrelizumab: A Review in Multiple Sclerosis. Drugs 2022, 82 (3), 323– 334, DOI: 10.1007/s40265-022-01672-9There is no corresponding record for this reference.
- 21Hauser, S. L.; Kappos, L.; Montalban, X.; Craveiro, L.; Chognot, C.; Hughes, R.; Koendgen, H.; Pasquarelli, N.; Pradhan, A.; Prajapati, K. Safety of Ocrelizumab in Patients With Relapsing and Primary Progressive Multiple Sclerosis. Neurology 2021, 97 (16), e1546– e1559, DOI: 10.1212/WNL.0000000000012700There is no corresponding record for this reference.
- 22Kiptoo, P.; Buyuktimkin, B.; Badawi, A. H.; Stewart, J.; Ridwan, R.; Siahaan, T. J. Controlling immune response and demyelination using highly potent bifunctional peptide inhibitors in the suppression of experimental autoimmune encephalomyelitis. Clin. Exp. Immunol. 2013, 172 (1), 23– 36, DOI: 10.1111/cei.12029There is no corresponding record for this reference.
- 23Badawi, A. H.; Kiptoo, P.; Wang, W. T.; Choi, I. Y.; Lee, P.; Vines, C. M.; Siahaan, T. J. Suppression of EAE and prevention of blood-brain barrier breakdown after vaccination with novel bifunctional peptide inhibitor. Neuropharmacology 2012, 62 (4), 1874– 1881, DOI: 10.1016/j.neuropharm.2011.12.013There is no corresponding record for this reference.
- 24Littman, D. R.; Rudensky, A. Y. Th17 and regulatory T cells in mediating and restraining inflammation. Cell 2010, 140 (6), 845– 858, DOI: 10.1016/j.cell.2010.02.021There is no corresponding record for this reference.
- 25Moorman, C. D.; Bastian, A. G.; DeOca, K. B.; Mannie, M. D. A GM-CSF-neuroantigen tolerogenic vaccine elicits inefficient antigen recognition events below the CD40L triggering threshold to expand CD4(+) CD25(+) FOXP3(+) Tregs that inhibit experimental autoimmune encephalomyelitis (EAE). J. Neuroinflammation 2020, 17 (1), 180, DOI: 10.1186/s12974-020-01856-8There is no corresponding record for this reference.
- 26Gagliani, N.; Amezcua Vesely, M. C.; Iseppon, A.; Brockmann, L.; Xu, H.; Palm, N. W.; de Zoete, M. R.; Licona-Limon, P.; Paiva, R. S.; Ching, T. Th17 cells transdifferentiate into regulatory T cells during resolution of inflammation. Nature 2015, 523 (7559), 221– 225, DOI: 10.1038/nature14452There is no corresponding record for this reference.
- 27Mari, E. R.; Rasouli, J.; Ciric, B.; Moore, J. N.; Conejo-Garcia, J. R.; Rajasagi, N.; Zhang, G. X.; Rabinovich, G. A.; Rostami, A. Galectin-1 is essential for the induction of MOG35–55 -based intravenous tolerance in experimental autoimmune encephalomyelitis. Eur. J. Immunol. 2016, 46 (7), 1783– 1796, DOI: 10.1002/eji.201546212There is no corresponding record for this reference.
- 28Leno-Duran, E.; Ng, S. L.; Strominger, J. L. Regulation of EAE by spontaneously generated IL-10-secreting regulatory T cells in HLA-DR15/TCR.Ob1A12 double transgenic mice. Immunology 2021, 163 (3), 338– 343, DOI: 10.1111/imm.13321There is no corresponding record for this reference.
- 29Raverdeau, M.; Christofi, M.; Malara, A.; Wilk, M. M.; Misiak, A.; Kuffova, L.; Yu, T.; McGinley, A. M.; Quinn, S. M.; Massilamany, C. Retinoic acid-induced autoantigen-specific type 1 regulatory T cells suppress autoimmunity. EMBO Rep. 2019, 20 (5), e47121 DOI: 10.15252/embr.201847121There is no corresponding record for this reference.
- 30Loos, J.; Schmaul, S.; Noll, T. M.; Paterka, M.; Schillner, M.; Loffel, J. T.; Zipp, F.; Bittner, S. Functional characteristics of Th1, Th17, and ex-Th17 cells in EAE revealed by intravital two-photon microscopy. J. Neuroinflammation 2020, 17 (1), 357, DOI: 10.1186/s12974-020-02021-xThere is no corresponding record for this reference.
- 31Xu, H.; Agalioti, T.; Zhao, J.; Steglich, B.; Wahib, R.; Vesely, M. C. A.; Bielecki, P.; Bailis, W.; Jackson, R.; Perez, D. The induction and function of the anti-inflammatory fate of T(H)17 cells. Nat. Commun. 2020, 11 (1), 3334, DOI: 10.1038/s41467-020-17097-5There is no corresponding record for this reference.
- 32Worzner, K.; Zimmermann, J.; Buhl, R.; Desoi, A.; Christensen, D.; Dietrich, J.; Nguyen, N.; Lindenstrom, T.; Woodworth, J. S.; Alhakeem, R. S. Repeated immunization with ATRA-containing liposomal adjuvant transdifferentiates Th17 cells to a Tr1-like phenotype. J. Autoimmun 2024, 144, 103174 DOI: 10.1016/j.jaut.2024.103174There is no corresponding record for this reference.
- 33Vieira, P. L.; Christensen, J. R.; Minaee, S.; O’Neill, E. J.; Barrat, F. J.; Boonstra, A.; Barthlott, T.; Stockinger, B.; Wraith, D. C.; O’Garra, A. IL-10-secreting regulatory T cells do not express Foxp3 but have comparable regulatory function to naturally occurring CD4+CD25+ regulatory T cells. J. Immunol 2004, 172 (10), 5986– 5993, DOI: 10.4049/jimmunol.172.10.5986There is no corresponding record for this reference.
- 34Maimone, D.; Guazzi, G. C.; Annunziata, P. IL-6 detection in multiple sclerosis brain. J. Neurol Sci. 1997, 146 (1), 59– 65, DOI: 10.1016/S0022-510X(96)00283-3There is no corresponding record for this reference.Kebir, H.; Ifergan, I.; Alvarez, J. I.; Bernard, M.; Poirier, J.; Arbour, N.; Duquette, P.; Prat, A. Preferential recruitment of interferon-gamma-expressing TH17 cells in multiple sclerosis. Ann. Neurol. 2009, 66 (3), 390– 402, DOI: 10.1002/ana.21748There is no corresponding record for this reference.
- 35Prendergast, C. T.; Anderton, S. M. Immune cell entry to central nervous system--current understanding and prospective therapeutic targets. Endocr Metab Immune Disord Drug Targets 2009, 9 (4), 315– 327, DOI: 10.2174/187153009789839219There is no corresponding record for this reference.
- 36Lin, W.; Kemper, A.; Dupree, J. L.; Harding, H. P.; Ron, D.; Popko, B. Interferon-gamma inhibits central nervous system remyelination through a process modulated by endoplasmic reticulum stress. Brain 2006, 129 (Pt 5), 1306– 1318, DOI: 10.1093/brain/awl044There is no corresponding record for this reference.
- 37Buyuktimkin, B.; Manikwar, P.; Kiptoo, P. K.; Badawi, A. H.; Stewart, J. M., Jr; Siahaan, T. J. Vaccinelike and prophylactic treatments of EAE with novel I-domain antigen conjugates (IDAC): targeting multiple antigenic peptides to APC. Mol. Pharm. 2013, 10 (1), 297– 306, DOI: 10.1021/mp300440xThere is no corresponding record for this reference.
- 38Martin, T. R.; Galli, S. J.; Katona, I. M.; Drazen, J. M. Role of mast cells in anaphylaxis. Evidence for the importance of mast cells in the cardiopulmonary alterations and death induced by anti-IgE in mice. J. Clin Invest 1989, 83 (4), 1375– 1383, DOI: 10.1172/JCI114025There is no corresponding record for this reference.
- 39Smith, C. E.; Eagar, T. N.; Strominger, J. L.; Miller, S. D. Differential induction of IgE-mediated anaphylaxis after soluble vs. cell-bound tolerogenic peptide therapy of autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. U. S. A. 2005, 102 (27), 9595– 9600, DOI: 10.1073/pnas.0504131102There is no corresponding record for this reference.
- 40Miller, S. D.; Turley, D. M.; Podojil, J. R. Antigen-specific tolerance strategies for the prevention and treatment of autoimmune disease. Nat. Rev. Immunol 2007, 7 (9), 665– 677, DOI: 10.1038/nri2153There is no corresponding record for this reference.
- 41Sabatos-Peyton, C. A.; Verhagen, J.; Wraith, D. C. Antigen-specific immunotherapy of autoimmune and allergic diseases. Curr. Opin Immunol 2010, 22 (5), 609– 615, DOI: 10.1016/j.coi.2010.08.006There is no corresponding record for this reference.
- 42Pirzer, T.; Becher, K. S.; Rieker, M.; Meckel, T.; Mootz, H. D.; Kolmar, H. Generation of Potent Anti-HER1/2 Immunotoxins by Protein Ligation Using Split Inteins. ACS Chem. Biol. 2018, 13 (8), 2058– 2066, DOI: 10.1021/acschembio.8b00222There is no corresponding record for this reference.
- 43Ludwig, C.; Pfeiff, M.; Linne, U.; Mootz, H. D. Ligation of a synthetic peptide to the N terminus of a recombinant protein using semisynthetic protein trans-splicing. Angew. Chem., Int. Ed. Engl. 2006, 45 (31), 5218– 5221, DOI: 10.1002/anie.200600570There is no corresponding record for this reference.
- 44Manikwar, P.; Tejo, B. A.; Shinogle, H.; Moore, D. S.; Zimmerman, T.; Blanco, F.; Siahaan, T. J. Utilization of I-domain of LFA-1 to Target Drug and Marker Molecules to Leukocytes. Theranostics 2011, 1, 277– 289, DOI: 10.7150/thno/v01p0277There is no corresponding record for this reference.
- 45Manikwar, P.; Buyuktimkin, B.; Kiptoo, P.; Badawi, A. H.; Galeva, N. A.; Williams, T. D.; Siahaan, T. J. I-domain-antigen conjugate (IDAC) for delivering antigenic peptides to APC: synthesis, characterization, and in vivo EAE suppression. Bioconjug Chem. 2012, 23 (3), 509– 517, DOI: 10.1021/bc200580jThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.bioconjchem.5c00518.
Procedure to express and purify the LABL-Fc-ST protein as a starting material; experimental method to produce sortase A enzyme; WES capillary cartridge analysis of the MOG-Fc-BPI molecule in reduced and intact forms (Figure S1) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



