The Many Lives of [Ru(bpy)3]2+: A Historical PerspectiveClick to copy article linkArticle link copied!
- Giorgio Scattolini*Giorgio Scattolini*Email: [email protected]Department of Chemistry − Ångström Laboratory, Uppsala University, Box 523, 751 20 Uppsala, SwedenMore by Giorgio Scattolini
- Andrea RosichiniAndrea RosichiniDepartment of Chemistry − Ångström Laboratory, Uppsala University, Box 523, 751 20 Uppsala, SwedenMore by Andrea Rosichini
- Nidhi KaulNidhi KaulDepartment of Chemistry − Ångström Laboratory, Uppsala University, Box 523, 751 20 Uppsala, SwedenMore by Nidhi Kaul
Abstract
[Ru(bpy)3]2+, tris(bipyridine)ruthenium(II), is a popular transition metal complex whose favorable photophysical properties have afforded it a central place in inorganic photochemistry and various related fields. In this perspective, in contrast to the large number of extant technical reviews, we instead note critical developments from a historical context. Of particular note are relatively lesser-known investigations in the field of analytical chemistry that predate the complex’s rise to prominence as a photosensitizer. Recent studies that revisit the complex’s own fundamental photophysics are also highlighted. Thus, in addition to serving as a proverbial almanac for the complex’s rich history, this condensed perspective portends yet more fruitful lives for research into [Ru(bpy)3]2+, despite the many already lived.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Synopsis
In addition to serving as a proverbial almanac for the complex’s rich history, this condensed perspective portends yet more fruitful lives for research into [Ru(bpy)3]2+, despite the many already explored.
Introduction
Figure 1
Figure 1. (a) Chemical structure of [Ru(bpy)3]2+. (b) Web of Science output for “Tris(bipyridine)ruthenium” or “Tris(dipyridine)ruthenium” or “Ru(bpy)3” or “ruthenium trisbipyridine” or “Ru(dipy)3” or “Rubpy3” or “Rudipy3” or “Tris(2,2′-bipyridine) Ruthenium”, i.e., various monikers for [Ru(bpy)3]2+ over the past several decades. (c) Structures of the two enantiomers of [Ru(bpy)3]2+ reported by Burstall in 1936. Reproduced with permission from ref (2). Copyright 1936 Royal Society of Chemistry.
Figure 2
Figure 2. Timeline of the key publications in the history of [Ru(bpy)3]2+ (CL: chemiluminescence; ECL: electrochemiluminescence; TA: transient absorption; DSSC: dye-sensitized solar cell).
Inorganic Chemistry
Analytical Chemistry
Figure 3
Figure 3. (a) First transmission spectrum of [Ru(bpy)3]2+ reported by Brandt et al. in 1949. Reproduced from ref (51). Copyright 1949 American Chemical Society. (b) First emission spectrum (dashed line) reported by Paris et al. in 1959. Reproduced from ref (52). Copyright 1959 American Chemical Society.
Photophysics and Photochemistry
Figure 4
Figure 4. First crystal structure of [Ru(bpy)3]2+ reported by Rillema et al. in 1979. Reproduced with permission from ref (89). Copyright 1979 Royal Society of Chemistry.
Photoelectrochemistry
Figure 5
Figure 5. (a) First cyclic voltammogram of [Ru(bpy)3]2+ reported by Tokel and Bard in 1972. Reproduced from ref (144). Copyright 1972 American Chemical Society. (b) Low temperature cyclic voltammogram of (I) [Ru(bpy)3]2+ and (II) [Ru(4,4′-(CO2Et)2bpy)3]2+ reported by Ohsawa, DeArmond, Hanck et al. in 1983. Reproduced from ref (146). Copyright 1983 American Chemical Society.
Photoredox Catalysis
Figure 6
Figure 6. One of the earliest reported schemes for a photoredox reaction. In this process, [Ru(bpy)3]2+ is first excited, and then it is reductively quenched by 1-benzyl-l,4-dihydronicotinamide (BNAH). Finally, [Ru(bpy)3]+ reacts with benzyl bromide yielding 1,2-diphenylethane, the desired reaction product, and regenerating [Ru(bpy)3]2+. Reproduced with permission from ref (186). Copyright 1984 Royal Society of Chemistry.
Biochemical Studies
Figure 7
Figure 7. (a) Tunneling timetable for intraprotein electron transfer in Ru-modified proteins. Reproduced with permission from ref (233). Copyright 2005 National Academy of Sciences, U.S.A. (b) Model for the interaction of Δ-[Ru(phen)3]2+ (left) and Λ-[Ru(phen)3]2+ (right) with the DNA double helix. Reproduced with permission from ref (238). Copyright 1988 American Association for the Advancement of Science.
Conclusions
Figure 8
Figure 8. License plate of Prof. Thomas Meyer (picture kindly provided by Tyler Meyer).
Acknowledgments
This review is dedicated to Professor Leif Hammarström on the occasion of his 61st birthday. The authors thank Dr. Starla Glover for the feedback on the manuscript, Helena Wagner for the translations from German, and Shabnam Chandel for drawing the TOC graphics. The authors are also thankful to Prof. Jillian Dempsey for confirming the statements on [Ru(bpy)3]2+ at UNC and to Tyler Meyer for providing the pictures of Prof. Thomas Meyer’s license plate. G.S. acknowledges financial support from his supervisors, Michał Maj and Leif Hammarström, in the form of a Swedish Society for Medical Research grant (grant no. S20-0156) and a Swedish Research Council grant (grant no. 2024-04372). A.R. acknowledges financial support from his supervisor, Leif Hammarström, in the form of a Swedish Research Council grant (grants no. 2020-05246). N.K. acknowledges financial support from the Swedish Research Council (grant no. 2024-06490).
References
This article references 263 other publications.
- 1Arias-Rotondo, D. M. The fruit fly of photophysics. Nat. Chem. 2022, 14, 716– 716, DOI: 10.1038/s41557-022-00955-8Google ScholarThere is no corresponding record for this reference.
- 2Burstall, F. H. 34. Optical activity dependent on co-ordinated bivalent ruthenium. Journal of the Chemical Society 1936, 173– 175, DOI: 10.1039/jr9360000173Google ScholarThere is no corresponding record for this reference.
- 3Sutin, N. Light-induced electron transfer reactions. J. Photochem. 1979, 10, 19– 40, DOI: 10.1016/0047-2670(79)80035-0Google ScholarThere is no corresponding record for this reference.
- 4Kalyanasundaram, K. Photophysics, photochemistry and solar energy conversion with tris(bipyridyl)ruthenium(II) and its analogues. Coord. Chem. Rev. 1982, 46, 159– 244, DOI: 10.1016/0010-8545(82)85003-0Google ScholarThere is no corresponding record for this reference.
- 5De Armond, M. K.; Myrick, M. L. The life and times of [Ru(bpy)3]2+: localized orbitals and other strange occurrences. Acc. Chem. Res. 1989, 22, 364– 370, DOI: 10.1021/ar00166a004Google ScholarThere is no corresponding record for this reference.
- 6Dongare, P.; Myron, B. D.; Wang, L.; Thompson, D. W.; Meyer, T. J. [Ru(bpy)3]2+ revisited. Is it localized or delocalized? How does it decay?. Coord. Chem. Rev. 2017, 345, 86– 107, DOI: 10.1016/j.ccr.2017.03.009Google ScholarThere is no corresponding record for this reference.
- 7Bell, J. D.; Murphy, J. A. Recent advances in visible light-activated radical coupling reactions triggered by (i) ruthenium, (ii) iridium and (iii) organic photoredox agents. Chem. Soc. Rev. 2021, 50, 9540– 9685, DOI: 10.1039/D1CS00311AGoogle ScholarThere is no corresponding record for this reference.
- 8Wu, C. Ruthenium-based complex dyes for dye-sensitized solar cells. Journal of the Chinese Chemical Society 2022, 69, 1242– 1252, DOI: 10.1002/jccs.202200225Google ScholarThere is no corresponding record for this reference.
- 9Ankathatti Munegowda, M.; Manalac, A.; Weersink, M.; McFarland, S. A.; Lilge, L. Ru(II) containing photosensitizers for photodynamic therapy: A critique on reporting and an attempt to compare efficacy. Coord. Chem. Rev. 2022, 470, 214712 DOI: 10.1016/j.ccr.2022.214712Google ScholarThere is no corresponding record for this reference.
- 10Juris, A.; Balzani, V.; Barigelletti, F.; Campagna, S.; Belser, P.; von Zelewsky, A. Ru(II) polypyridine complexes: photophysics, photochemistry, eletrochemistry, and chemiluminescence. Coord. Chem. Rev. 1988, 84, 85– 277, DOI: 10.1016/0010-8545(88)80032-8Google ScholarThere is no corresponding record for this reference.
- 11Constable, E. C.; Housecroft, C. E. The Early Years of 2, 2′-Bipyridine─A Ligand in Its Own Lifetime. Molecules 2019, 24, 3951, DOI: 10.3390/molecules24213951Google ScholarThere is no corresponding record for this reference.
- 12Blau, F. Die Destillation pyridinmonocarbonsaurer Salze. Berichte der deutschen chemischen Gesellschaft 1888, 21, 1077– 1078, DOI: 10.1002/cber.188802101201Google ScholarThere is no corresponding record for this reference.
- 13Blau, F. Uber neue organische Metallverbindungen. Monatshefte für Chemie 1898, 19, 647– 689, DOI: 10.1007/BF01517438Google ScholarThere is no corresponding record for this reference.
- 14Miller, R. R.; Brandt, W. W.; Puke, M.; Puke, O. Metal-Amine Coördination Compounds. V. The Ruthenium-2, 2′-Bipyridine System. J. Am. Chem. Soc. 1955, 77, 3178– 3180, DOI: 10.1021/ja01617a003Google ScholarThere is no corresponding record for this reference.
- 15Coulson, E. A. Dr. F.H. Burstall. Nature 1956, 177, 210– 211, DOI: 10.1038/177210a0Google ScholarThere is no corresponding record for this reference.
- 16Gilbert Thomas Morgan, 1872–1940. Obituary Notices of Fellows of the Royal Society ; 1941; pp 354– 362.Google ScholarThere is no corresponding record for this reference.
- 17Morgan, G. T.; Drew, H. D. K. CLXII.─Researches on residual affinity and co-ordination. Part II. Acetylacetones of selenium and tellurium. Journal of the Chemical Society, Transactions 1920, 117, 1456– 1465, DOI: 10.1039/CT9201701456Google ScholarThere is no corresponding record for this reference.
- 18Morgan, G. T.; Burstall, F. H. 3. Dehydrogenation of pyridine by anhydrous ferric chloride. J. Chem. Soc. 1932, 1456– 1465, DOI: 10.1039/jr9320000020Google ScholarThere is no corresponding record for this reference.
- 19Morgan, G.; Burstall, F. H. 347. Researches on residual affinity and co-ordination. Part XXXVII. Complex metallic salts containing 2:6-di-2′-pyridylpyridine (2:2′: 2″-tripyridyl). J. Chem. Soc. 1937, 1649– 1655, DOI: 10.1039/JR9370001649Google ScholarThere is no corresponding record for this reference.
- 20Burstall, F. H. 312. Researches on the polypyridyls. Journal of the Chemical Society 1938, 1662, DOI: 10.1039/jr9380001662Google ScholarThere is no corresponding record for this reference.
- 21Morgan, G. T.; Burstall, F. H. CCCXLIV.─Researches on residual affinity and co-ordination. Part XXXII. Complex salts of bivalent silver. Journal of the Chemical Society 1930, 0, 2594– 2598, DOI: 10.1039/JR9300002594Google ScholarThere is no corresponding record for this reference.
- 22Morgan, G. T.; Burstall, F. H. CCCII.─Researches on residual affinity and co-ordination. Part XXXIII. Optical activity dependent on co-ordinated nickel. Journal of the Chemical Society 1931, 0, 2213– 2218, DOI: 10.1039/JR9310002213Google ScholarThere is no corresponding record for this reference.
- 23Morgan, G. T.; Burstall, F. H. 201. Researches on residual affinity and co-ordination. Part XXXIV. 2:2′-Dipyridyl platinum salts. Journal of the Chemical Society 1934, 0, 965– 971, DOI: 10.1039/JR9340000965Google ScholarThere is no corresponding record for this reference.
- 24Morgan, G. T.; Burstall, F. H. 323. Researches on residual affinity and co-ordination. Part XXXV. 2:2′: 2″-Tripyridylplatinum salts. Journal of the Chemical Society 1934, 0, 1498– 1500, DOI: 10.1039/JR9340001498Google ScholarThere is no corresponding record for this reference.
- 25Werner, A. Über Spiegelbild-Isomerie bei Eisenverbindungen. Berichte der deutschen chemischen Gesellschaft 1912, 45, 433– 436, DOI: 10.1002/cber.19120450165Google ScholarThere is no corresponding record for this reference.
- 26https://www.nobelprize.org/prizes/chemistry/1913/werner/facts/.Google ScholarThere is no corresponding record for this reference.
- 27Baker, A. A. A History of Indicators. Chymia 1964, 9, 147– 167, DOI: 10.2307/27757238Google ScholarThere is no corresponding record for this reference.
- 28Mellon, M. G. Fisher Award Address: A Century of Colorimetry. Anal. Chem. 1952, 24, 924– 931, DOI: 10.1021/ac60066a002Google ScholarThere is no corresponding record for this reference.
- 29Snell, F. T. Colorimetric Analysis; D. Van Nostrand Company: 1921.Google ScholarThere is no corresponding record for this reference.
- 30Johansson, A. The development of the titration methods. Anal. Chim. Acta 1988, 206, 97– 109, DOI: 10.1016/S0003-2670(00)80834-XGoogle ScholarThere is no corresponding record for this reference.
- 31Szabadvary, F. Indicators: A historical perspective. J. Chem. Educ. 1964, 41, 285– 287, DOI: 10.1021/ed041p285Google ScholarThere is no corresponding record for this reference.
- 32Irving, H. M. N. H. Centenary lecture. One hundred years of development in Analytical Chemistry. Analyst 1974, 99, 787– 801, DOI: 10.1039/an9749900787Google ScholarThere is no corresponding record for this reference.
- 33
Inventor of the isoelectric precipitation method for the improvement of insulin production.259
There is no corresponding record for this reference. - 34Walden, G. H.; Hammett, L. P.; Chapman, R. P. A reversible oxidation indicator of high potential especially adapted to oxidimetric titrations. J. Am. Chem. Soc. 1931, 53, 3908– 3908, DOI: 10.1021/ja01361a508Google ScholarThere is no corresponding record for this reference.
- 35Brandt, W. W.; Dwyer, F. P.; Gyarfas, E. D. Chelate Complexes of 1, 10-Phenanthroline and Related Compounds. Chem. Rev. 1954, 54, 959– 1017, DOI: 10.1021/cr60172a003Google ScholarThere is no corresponding record for this reference.
- 36Steigman, J.; Birnbaum, N.; Edmonds, S. Ruthenium Dipyridyl-New Oxidimetric Indicator. Industrial and Engineering Chemistry Analytical Edition 1942, 14, 30, DOI: 10.1021/i560101a010Google ScholarThere is no corresponding record for this reference.
- 37Dwyer, F. P.; Nyholm, R. S. The instability constant of the tris-orthophenanthroline ferrous ion. Journal and Proceedings of the Royal Society of New South Wales 1947, 80, 28– 32, DOI: 10.5962/p.360430Google ScholarThere is no corresponding record for this reference.
- 38Dwyer, F. P. The Chemistry of Ruthenium. Part III. The Redox Potentials of the Ruthenium II Complexes with Substituted Derivatives of 2:2′Dipyridyl and o-Phenanthroline. Journal and Proceedings of the Royal Society of New South Wales 1950, 83, 134– 137, DOI: 10.5962/p.360547Google ScholarThere is no corresponding record for this reference.
- 39Baker, A. T.; Livingstone, S. E. Historical Sketch: The early history of coordination chemistry in Australia. Polyhedron 1985, 4, 1337– 1351, DOI: 10.1016/S0277-5387(00)86962-4Google ScholarThere is no corresponding record for this reference.
- 40Minerals yearbook 1941–1959; United States Department of Interior, Bureau of Mines: 1941–1959.Google ScholarThere is no corresponding record for this reference.
- 41
For reference, the Ru/Fe price ratio in 2024 is 110,000 and it was even lower in the 1990s.
There is no corresponding record for this reference. - 42Dwyer, F. P.; Humpoletz, J. E.; Nyholm, R. S. The Chemistry of Ruthenium. Part I. The Redox Potential of the Tris-Orthophenanthroline Ruthenous Ion. Journal and Proceedings of the Royal Society of New South Wales 1947, 80, 212– 216, DOI: 10.5962/p.360444Google ScholarThere is no corresponding record for this reference.
- 43Backhouse, J.; Dwyer, F. P. The Chemistry of Ruthenium. Part VI. The Existence of the Tris-o- Phenanthroline Ruthenium III Ions in Entantiomorphous Forms. J. Proc. R. Soc. New South Wales 1949, 83, 170– 173, DOI: 10.5962/p.360552Google ScholarThere is no corresponding record for this reference.
- 44Dwyer, F. P.; Gyarfas, E. C. The Chemistry of Ruthenium. Part VII. The Oxidation of D and L Tris 2:2′Dipyridyl Ruthenium II Iodide. Journal and Proceedings of the Royal Society of New South Wales 1950, 83, 174– 176, DOI: 10.5962/p.360553Google ScholarThere is no corresponding record for this reference.
- 45Breckenridge, J. G.; Singer, S. A. G. 5-Hydroxyquinoline-8-carboxylic acid─A colorimetric reagent for ruthenium. Canadian Journal of Research 1947, 25b, 49– 55, DOI: 10.1139/cjr47b-006Google ScholarThere is no corresponding record for this reference.
- 46Deford, D. The Chemistry of Ruthenium; Elsevier: 1948, DOI: 10.2172/4435343 .Google ScholarThere is no corresponding record for this reference.
- 47Ayres, G. H.; Young, F. Spectrophotometric Study of Ruthenium-Thiourea Complex. Anal. Chem. 1950, 22, 1277– 1280, DOI: 10.1021/ac60046a014Google ScholarThere is no corresponding record for this reference.
- 48Sheard, C.; States, M. N. A Concave Grating Photoelectric Spectrophotometer. J. Opt. Soc. Am. 1941, 31, 64, DOI: 10.1364/JOSA.31.000064Google ScholarThere is no corresponding record for this reference.
- 49Thomas, N. C. The early history of spectroscopy. J. Chem. Educ. 1991, 68, 631, DOI: 10.1021/ed068p631Google ScholarThere is no corresponding record for this reference.
- 50Karayannis, M. I.; Efstathiou, C. E. Significant steps in the evolution of analytical chemistry─Is the today’s analytical chemistry only chemistry?. Talanta 2012, 102, 7– 15, DOI: 10.1016/j.talanta.2012.06.003Google ScholarThere is no corresponding record for this reference.
- 51Brandt, W. W.; Smith, G. F. Polysubstituted 1,10-Phenanthrolines and Bipyridines as Multiple Range Redox Indicators. Anal. Chem. 1949, 21, 1313– 1319, DOI: 10.1021/ac60035a003Google ScholarThere is no corresponding record for this reference.
- 52Paris, J. P.; Brandt, W. W. Charge transfer luminescence of a ruthenium(II) chelate. J. Am. Chem. Soc. 1959, 81, 5001– 5002, DOI: 10.1021/ja01527a064Google ScholarThere is no corresponding record for this reference.
- 53
We note that Burstall too remained active, and together with Dwyer reported the absorption spectrum of [Os(bpy)3]2+, the first for an osmium compound.260
There is no corresponding record for this reference. - 54Yamasaki, K. Absorptionsspektren von Metallkomplexsalzen des 2, 2′-Dipyridyls. I-IV. Bull. Chem. Soc. Jpn. 1937, 390– 394, DOI: 10.1246/bcsj.12.390Google ScholarThere is no corresponding record for this reference.
- 55O’Haver, T. C. Development of luminescence spectrometry as an analytical tool. J. Chem. Educ. 1978, 55, 423, DOI: 10.1021/ed055p423Google ScholarThere is no corresponding record for this reference.
- 56Valeur, B.; Berberan-Santos, M. N. A Brief History of Fluorescence and Phosphorescence before the Emergence of Quantum Theory. J. Chem. Educ. 2011, 88, 731– 738, DOI: 10.1021/ed100182hGoogle ScholarThere is no corresponding record for this reference.
- 57Randall, J. T. Luminescence of Liquids and Solids. Nature 1944, 154, 625– 625, DOI: 10.1038/154625a0Google ScholarThere is no corresponding record for this reference.
- 58Bowman, R. L.; Caulfield, P. A.; Udenfriend, S. Spectrophotofluorometric Assay in the Visible and Ultraviolet. Science 1955, 122, 32– 33, DOI: 10.1126/science.122.3157.32Google ScholarThere is no corresponding record for this reference.
- 59Slavin, W.; Mooney, R. W.; Palumbo, D. T. Energy-Recording Spectrofluorimeter. J. Opt. Soc. Am. 1961, 51, 93, DOI: 10.1364/JOSA.51.000093Google ScholarThere is no corresponding record for this reference.
- 60Turner, G. K. An Absolute Spectrofluorometer: A new instrument has been developed for simple determinations of absolute fluorescence spectra. Science 1964, 146, 183– 189, DOI: 10.1126/science.146.3641.183Google ScholarThere is no corresponding record for this reference.
- 61Veening, H.; Brandt, W. W. Fluorescimetric Determination of Ruthenium. Anal. Chem. 1960, 32, 1426– 1428, DOI: 10.1021/ac60167a015Google ScholarThere is no corresponding record for this reference.
- 62Paris, J. P. Studies on molecular electronic transitions, Ph.D. thesis; Purdue University: 1960.Google ScholarThere is no corresponding record for this reference.
- 63Stieglitz, J. A Theory of Color Production: I. Proc. Natl. Acad. Sci. U. S. A. 1923, 9, 303– 308, DOI: 10.1073/pnas.9.9.303Google ScholarThere is no corresponding record for this reference.
- 64Stieglitz, J. A Theory of Color Production: II, Inorganic Compounds. Proc. Natl. Acad. Sci. U. S. A. 1923, 9, 308– 312, DOI: 10.1073/pnas.9.9.308Google ScholarThere is no corresponding record for this reference.
- 65Mulliken, R. S. Low Electronic States of Simple Heteropolar Diatomic Molecules: III. Hydrogen and Univalent Metal Halides. Phys. Rev. 1937, 51, 310– 332, DOI: 10.1103/PhysRev.51.310Google ScholarThere is no corresponding record for this reference.
- 66Mulliken, R. S. Intensities of Electronic Transitions in Molecular Spectra II. Charge-Transfer Spectra. J. Chem. Phys. 1939, 7, 20– 34, DOI: 10.1063/1.1750319Google ScholarThere is no corresponding record for this reference.
- 67Rabinowitch, E. Electron Transfer Spectra and Their Photochemical Effects. Rev. Mod. Phys. 1942, 14, 112– 131, DOI: 10.1103/RevModPhys.14.112Google ScholarThere is no corresponding record for this reference.
- 68McClure, D. S. Solid State Physics; Elsevier: 1959; pp 399– 525.Google ScholarThere is no corresponding record for this reference.
- 69Porter, G. B.; Schläfer, H. L. Zusammenfassender Bericht. Zur Frage der Lumineszenz bei Übergangsmetallverbindungen. Berichte der Bunsengesellschaft für physikalische Chemie 1964, 68, 316– 331, DOI: 10.1002/bbpc.19640680404Google ScholarThere is no corresponding record for this reference.
- 70Scheibe, G. Die Absorption der Halogenionen in wässeriger Lösung im beginnenden Schumann-Ultraviolett. Zeitschrift für Physikalische Chemie 1929, 5BB, 355– 364, DOI: 10.1515/zpch-1929-0528Google ScholarThere is no corresponding record for this reference.
- 71Kondo, Y. Electron Transfer Spectra in the Crystals of Hexamminecobalt(III) Salts. Bull. Chem. Soc. Jpn. 1955, 28, 497– 504, DOI: 10.1246/bcsj.28.497Google ScholarThere is no corresponding record for this reference.
- 72Jo̷rgensen, C. K. Complexes of the 4d- and 5d-Groups. III. Absorption Spectra of Marcel Delépine’s Rhodium(III) and Iridium(III) Complexes. Acta Chem. Scand. 1957, 11, 151– 165, DOI: 10.3891/acta.chem.scand.11-0151Google ScholarThere is no corresponding record for this reference.
- 73Jo̷rgensen, C. K. Complexes of the 4d- and 5d-Groups. IV. Electron Transfer Bands with Special Application to M. Delépine’s Complexes and a Transition from Iridium(III) to Pyridine, with some Remarks about Intermediate Coupling in Halide Complexes and the Uranyl Ion. Acta Chem. Scand. 1957, 11, 166– 178, DOI: 10.3891/acta.chem.scand.11-0166Google ScholarThere is no corresponding record for this reference.
- 74Crosby, G. A.; Perkins, W. G.; Klassen, D. M. Luminescence from Transition-Metal Complexes: Tris(2, 2′-bipyridine)- and Tris(1, 10-phenanthroline)Ruthenium(II). J. Chem. Phys. 1965, 43, 1498– 1503, DOI: 10.1063/1.1696960Google ScholarThere is no corresponding record for this reference.
- 75Demas, J.; Crosby, G. On the multiplicity of the emitting state of ruthenium(II) complexes. J. Mol. Spectrosc. 1968, 26, 72– 77, DOI: 10.1016/0022-2852(68)90144-6Google ScholarThere is no corresponding record for this reference.
- 76Fujita, I.; Kobayashi, H. Electronic Excitation Energy Transfer between Complex Ions. J. Chem. Phys. 1970, 52, 4904– 4905, DOI: 10.1063/1.1673730Google ScholarThere is no corresponding record for this reference.
- 77Adamson, A. W.; Demas, J. N. New photosensitizer. Tris(2, 2′-bipyridine)ruthenium(II) chloride. J. Am. Chem. Soc. 1971, 93, 1800– 1801, DOI: 10.1021/ja00736a049Google ScholarThere is no corresponding record for this reference.
- 78
Adamson had previously observed energy transfer and photoinduced electron transfer from organic chromophores to cobalt and chromium complexes.261,262
There is no corresponding record for this reference. - 79Gafney, H. D.; Adamson, A. W. Excited state Ru(bipyr)32+ as an electron-transfer reductant. J. Am. Chem. Soc. 1972, 94, 8238– 8239, DOI: 10.1021/ja00778a054Google ScholarThere is no corresponding record for this reference.
- 80Balzani, V.; Juris, A. Photochemistry and photophysics of Ru(II) polypyridine complexes in the Bologna group. From early studies to recent developments. Coord. Chem. Rev. 2001, 211, 97– 115, DOI: 10.1016/S0010-8545(00)00274-5Google ScholarThere is no corresponding record for this reference.
- 81YouTube, uploaded by American Chemical Society, 1st August 2011. https://www.youtube.com/watch?v=NCQWEUiwyGo.Google ScholarThere is no corresponding record for this reference.
- 82Demas, J. N.; Adamson, A. W. Tris (2, 2′-bipyridine)ruthenium(II) sensitized reactions of some oxalato complexes. J. Am. Chem. Soc. 1973, 95, 5159– 5168, DOI: 10.1021/ja00797a011Google ScholarThere is no corresponding record for this reference.
- 83Sabbatini, N.; Balzani, V. Photosensitized aquation of the hexacyanochromate(III) ion. Evidence against the doublet mechanism. J. Am. Chem. Soc. 1972, 94, 7587– 7589, DOI: 10.1021/ja00776a057Google ScholarThere is no corresponding record for this reference.
- 84Natarajan, P.; Endicott, J. F. Photochemistry of ethylenediaminetetraacetate complexes. Excited state reaction pathways in cobalt(III) complexes. J. Am. Chem. Soc. 1972, 94, 3635– 3636, DOI: 10.1021/ja00765a067Google ScholarThere is no corresponding record for this reference.
- 85Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37– 38, DOI: 10.1038/238037a0Google ScholarThere is no corresponding record for this reference.
- 86Concepcion, J. J.; House, R. L.; Papanikolas, J. M.; Meyer, T. J. Chemical approaches to artificial photosynthesis. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 15560– 15564, DOI: 10.1073/pnas.1212254109Google ScholarThere is no corresponding record for this reference.
- 87Design of Advanced Photocatalytic Materials for Energy and Environmental Applications; Springer: London, 2013.Google ScholarThere is no corresponding record for this reference.
- 88Ciamician, G. The Photochemistry of the Future. Science 1912, 36, 385– 394, DOI: 10.1126/science.36.926.385Google ScholarThere is no corresponding record for this reference.
- 89Rillema, D. P.; Jones, D. S. Structure of tris(2, 2′-bipyridyl)ruthenium(II) hexafluorophosphate, [Ru(bipy)3][PF6]2; X-ray crystallographic determination. J. Chem. Soc., Chem. Commun. 1979, 849– 851, DOI: 10.1039/C39790000849Google ScholarThere is no corresponding record for this reference.
- 90Bolletta, F.; Maestri, M.; Moggi, L.; Balzani, V. Ligand and geometry effects in the quenching of the tris(2, 2′-dipyridyl)ruthenium(II) phosphorescence by some chromium(III) complexes. J. Am. Chem. Soc. 1973, 95, 7864– 7866, DOI: 10.1021/ja00804a052Google ScholarThere is no corresponding record for this reference.
- 91Wrighton, M.; Markham, J. Quenching of the luminescent state of tris(2, 2′-bipyridine)ruthenium(II) by electronic energy transfer. J. Phys. Chem. 1973, 77, 3042– 3044, DOI: 10.1021/j100644a002Google ScholarThere is no corresponding record for this reference.
- 92Navon, G.; Sutin, N. Mechanism of the quenching of the phosphorescence of tris(2, 2′-bipyridine)ruthenium(II) by some cobalt(III) and ruthenium(III) complexes. Inorg. Chem. 1974, 13, 2159– 2164, DOI: 10.1021/ic50139a021Google ScholarThere is no corresponding record for this reference.
- 93Lin, C. T.; Boettcher, W.; Chou, M.; Creutz, C.; Sutin, N. Mechanism of the quenching of the emission of substituted polypyridineruthenium(II) complexes by iron(III), chromium(III), and europium(III) ions. J. Am. Chem. Soc. 1976, 98, 6536– 6544, DOI: 10.1021/ja00437a020Google ScholarThere is no corresponding record for this reference.
- 94Creutz, C.; Sutin, N. Electron-transfer reactions of excited states: direct evidence for reduction of the charge-transfer excited state of tris(2, 2′-bipyridine)ruthenium(II). J. Am. Chem. Soc. 1976, 98, 6384– 6385, DOI: 10.1021/ja00436a054Google ScholarThere is no corresponding record for this reference.
- 95Juris, A.; Gandolfi, M. T.; Manfrin, M. F.; Balzani, V. Electron and energy transfer mechanisms in the quenching of the tris(2, 2′-bipyridine)ruthenium(II) luminescence by cyanide complexes. J. Am. Chem. Soc. 1976, 98, 1047– 1048, DOI: 10.1021/ja00420a046Google ScholarThere is no corresponding record for this reference.
- 96Maestri, M.; Grätzel, M. 530 nm-Laser Photolysis Studies of the Photo Reduction of Tris (2, 2-bipyridine)-ruthenium(II) by Organic Donors. Ber. Bunsengesell. Phys. Chem. 1977, 81, 504– 507, DOI: 10.1002/bbpc.19770810510Google ScholarThere is no corresponding record for this reference.
- 97Bock, C. R.; Meyer, T. J.; Whitten, D. G. Electron transfer quenching of the luminescent excited state of tris(2, 2′-bipyridine)ruthenium(II). Flash photolysis relaxation technique for measuring the rates of very rapid electron transfer reactions. J. Am. Chem. Soc. 1974, 96, 4710– 4712, DOI: 10.1021/ja00821a078Google ScholarThere is no corresponding record for this reference.
- 98Schlicke, B.; Belser, P.; De Cola, L.; Sabbioni, E.; Balzani, V. Photonic Wires of Nanometric Dimensions. Electronic Energy Transfer in Rigid Rodlike Ru(bpy)32+-(ph)n-Os(bpy)32+ Compounds (ph = 1, 4-Phenylene;n= 3, 5, 7). J. Am. Chem. Soc. 1999, 121, 4207– 4214, DOI: 10.1021/ja990044bGoogle ScholarThere is no corresponding record for this reference.
- 99Welter, S.; Salluce, N.; Belser, P.; Groeneveld, M.; De Cola, L. Photoinduced electronic energy transfer in modular, conjugated, dinuclear Ru(II)/Os(II) complexes. Coord. Chem. Rev. 2005, 249, 1360– 1371, DOI: 10.1016/j.ccr.2004.11.008Google ScholarThere is no corresponding record for this reference.
- 100Balzani, V.; Campagna, S.; Denti, G.; Juris, A.; Serroni, S.; Venturi, M. Designing Dendrimers Based on Transition-Metal Complexes. Light-Harvesting Properties and Predetermined Redox Patterns. Acc. Chem. Res. 1998, 31, 26– 34, DOI: 10.1021/ar950202dGoogle ScholarThere is no corresponding record for this reference.
- 101Campagna, S.; Denti, G.; Serroni, S.; Ciano, M.; Juris, A.; Balzani, V. A tridecanuclear ruthenium(II)-polypyridine supramolecular species: synthesis, absorption and luminescence properties and electrochemical oxidation. Inorg. Chem. 1992, 31, 2982– 2984, DOI: 10.1021/ic00039a056Google ScholarThere is no corresponding record for this reference.
- 102Balzani, V.; Ceroni, P.; Credi, A.; Venturi, M. Ruthenium tris(bipyridine) complexes: Interchange between photons and electrons in molecular-scale devices and machines. Coord. Chem. Rev. 2021, 433, 213758 DOI: 10.1016/j.ccr.2020.213758Google ScholarThere is no corresponding record for this reference.
- 103Kirk, A. D.; Hoggard, P. E.; Porter, G. B.; Rockley, M. G.; Windsor, M. W. Picosecond flash photolysis and spectroscopy: Transition metal coordination compounds. Chem. Phys. Lett. 1976, 37, 199– 203, DOI: 10.1016/0009-2614(76)80198-4Google ScholarThere is no corresponding record for this reference.
- 104Bensasson, R.; Salet, C.; Balzani, V. Laser flash spectroscopy of tris(2, 2′-bipyridine)ruthenium(II) in solution. J. Am. Chem. Soc. 1976, 98, 3722– 3724, DOI: 10.1021/ja00428a064Google ScholarThere is no corresponding record for this reference.
- 105Watts, R. J. Ruthenium polypyridyls: A case study. J. Chem. Educ. 1983, 60, 834, DOI: 10.1021/ed060p834Google ScholarThere is no corresponding record for this reference.
- 106Fujita, I.; Kobayashi, H. Luminescence polarization of the tris(2, 2′-bipyridine)ruthenium(II) complex. Inorg. Chem. 1973, 12, 2758– 2762, DOI: 10.1021/ic50130a004Google ScholarThere is no corresponding record for this reference.
- 107Carroll, P. J.; Brus, L. E. Picosecond Raman scattering study of electron localization in the charge transfer excited state of tris(bipyridine)ruthenium(II). J. Am. Chem. Soc. 1987, 109, 7613– 7616, DOI: 10.1021/ja00259a005Google ScholarThere is no corresponding record for this reference.
- 108Hager, G. D.; Crosby, G. A. Charge-transfer exited states of ruthenium(II) complexes. I. Quantum yield and decay measurements. J. Am. Chem. Soc. 1975, 97, 7031– 7037, DOI: 10.1021/ja00857a013Google ScholarThere is no corresponding record for this reference.
- 109Baker, D.; Crosby, G. Spectroscopic and magnetic evidence for multiple-state emission from tris(2, 2′-bipyridine) ruthenium (II) sulfate. Chem. Phys. 1974, 4, 428– 433, DOI: 10.1016/0301-0104(74)85009-3Google ScholarThere is no corresponding record for this reference.
- 110Gallhuber, E.; Hensler, G.; Yersin, H. Low-temperature emission spectra of crystalline [Ru(bpy)3](ClO4)2. Chem. Phys. Lett. 1985, 120, 445– 449, DOI: 10.1016/0009-2614(85)85638-4Google ScholarThere is no corresponding record for this reference.
- 111Kober, E. M.; Meyer, T. J. An electronic structural model for the emitting MLCT excited states of Ru(bpy)32+ and Os(bpy)32+. Inorg. Chem. 1984, 23, 3877– 3886, DOI: 10.1021/ic00192a009Google ScholarThere is no corresponding record for this reference.
- 112Myrick, M.; Blakley, R.; Dearmond, M. The number and spacing of levels in the emitting manifold of [Ru(bpy)3]2+ low temperature measurements. Chem. Phys. Lett. 1989, 157, 73– 77, DOI: 10.1016/0009-2614(89)87210-0Google ScholarThere is no corresponding record for this reference.
- 113Yamauchi, S.; Komada, Y.; Hirota, N. Observation of surprisingly small zero-field splittings in the lowest emitting state of [Ru(bpy)3]2+. Chem. Phys. Lett. 1986, 129, 197– 200, DOI: 10.1016/0009-2614(86)80196-8Google ScholarThere is no corresponding record for this reference.
- 114Yersin, H.; Strasser, J. Triplets in metal–organic compounds. Chemical tunability of relaxation dynamics. Coord. Chem. Rev. 2000, 208, 331– 364, DOI: 10.1016/S0010-8545(00)00326-XGoogle ScholarThere is no corresponding record for this reference.
- 115Sibbett, W.; Lagatsky, A. A.; Brown, C. T. A. The development and application of femtosecond laser systems. Opt. Express 2012, 20, 6989, DOI: 10.1364/OE.20.006989Google ScholarThere is no corresponding record for this reference.
- 116Zewail, A. H. Laser Femtochemistry. Science 1988, 242, 1645– 1653, DOI: 10.1126/science.242.4886.1645Google ScholarThere is no corresponding record for this reference.
- 117Damrauer, N. H.; Cerullo, G.; Yeh, A.; Boussie, T. R.; Shank, C. V.; McCusker, J. K. Femtosecond Dynamics of Excited-State Evolution in [Ru(bpy)3]2+. Science 1997, 275, 54– 57, DOI: 10.1126/science.275.5296.54Google ScholarThere is no corresponding record for this reference.
- 118Atkins, A. J.; González, L. Trajectory Surface-Hopping Dynamics Including Intersystem Crossing in [Ru(bpy)3]2+. J. Phys. Chem. Lett. 2017, 8, 3840– 3845, DOI: 10.1021/acs.jpclett.7b01479Google ScholarThere is no corresponding record for this reference.
- 119Bhasikuttan, A. C.; Suzuki, M.; Nakashima, S.; Okada, T. Ultrafast Fluorescence Detection in Tris(2, 2′-bipyridine)ruthenium(II) Complex in Solution: Relaxation Dynamics Involving Higher Excited States. J. Am. Chem. Soc. 2002, 124, 8398– 8405, DOI: 10.1021/ja026135hGoogle ScholarThere is no corresponding record for this reference.
- 120Cannizzo, A.; van Mourik, F.; Gawelda, W.; Zgrablic, G.; Bressler, C.; Chergui, M. Broadband Femtosecond Fluorescence Spectroscopy of [Ru(bpy)3]2+. Angew. Chem., Int. Ed. 2006, 45, 3174– 3176, DOI: 10.1002/anie.200600125Google ScholarThere is no corresponding record for this reference.
- 121Oh, D. H.; Boxer, S. G. Stark effect spectra of Ru(diimine)32+ complexes. J. Am. Chem. Soc. 1989, 111, 1130– 1131, DOI: 10.1021/ja00185a054Google ScholarThere is no corresponding record for this reference.
- 122Yeh, A. T.; Shank, C. V.; McCusker, J. K. Ultrafast Electron Localization Dynamics Following Photo-Induced Charge Transfer. Science 2000, 289, 935– 938, DOI: 10.1126/science.289.5481.935Google ScholarThere is no corresponding record for this reference.
- 123Malone, R. A.; Kelley, D. F. Interligand electron transfer and transition state dynamics in Ru(II)trisbipyridine. J. Chem. Phys. 1991, 95, 8970– 8976, DOI: 10.1063/1.461228Google ScholarThere is no corresponding record for this reference.
- 124Wallin, S.; Davidsson, J.; Modin, J.; Hammarström, L. Femtosecond Transient Absorption Anisotropy Study on [Ru(bpy)3]2+ and [Ru(bpy)(py)4]2+. Ultrafast Interligand Randomization of the MLCT State. J. Phys. Chem. A 2005, 109, 4697– 4704, DOI: 10.1021/jp0509212Google ScholarThere is no corresponding record for this reference.
- 125Kawamoto, K.; Tamiya, Y.; Storr, T.; Cogdell, R. J.; Kinoshita, I.; Hashimoto, H. Disentangling the 1MLCT transition of [Ru(bpy)3]2+ by Stark absorption spectroscopy. J. Photochem. Photobiol., A 2018, 353, 618– 624, DOI: 10.1016/j.jphotochem.2017.08.025Google ScholarThere is no corresponding record for this reference.
- 126Stark, C. W.; Schreier, W. J.; Lucon, J.; Edwards, E.; Douglas, T.; Kohler, B. Interligand Electron Transfer in Heteroleptic Ruthenium(II) Complexes Occurs on Multiple Time Scales. J. Phys. Chem. A 2015, 119, 4813– 4824, DOI: 10.1021/acs.jpca.5b01770Google ScholarThere is no corresponding record for this reference.
- 127Moret, M.; Tavernelli, I.; Chergui, M.; Rothlisberger, U. Electron Localization Dynamics in the Triplet Excited State of [Ru(bpy)3]2+ in Aqueous Solution. Chem. – Eur. J. 2010, 16, 5889– 5894, DOI: 10.1002/chem.201000184Google ScholarThere is no corresponding record for this reference.
- 128Tavernelli, I.; Curchod, B. F.; Rothlisberger, U. Nonadiabatic molecular dynamics with solvent effects: A LR-TDDFT QM/MM study of ruthenium (II) tris (bipyridine) in water. Chem. Phys. 2011, 391, 101– 109, DOI: 10.1016/j.chemphys.2011.03.021Google ScholarThere is no corresponding record for this reference.
- 129Alcover-Fortuny, G.; Wu, J.; Caballol, R.; de Graaf, C. Quantum Chemical Study of the Interligand Electron Transfer in Ru Polypyridyl Complexes. J. Phys. Chem. A 2018, 122, 1114– 1123, DOI: 10.1021/acs.jpca.7b11422Google ScholarThere is no corresponding record for this reference.
- 130Zeng, C.; Li, Y.; Zheng, H.; Ren, M.; Wu, W.; Chen, Z. Nature of ultrafast dynamics in the lowest-lying singlet excited state of [Ru(bpy)3]2+. Phys. Chem. Chem. Phys. 2024, 26, 6524– 6531, DOI: 10.1039/D3CP03806HGoogle ScholarThere is no corresponding record for this reference.
- 131Stockett, M. H.; Bro̷ndsted Nielsen, S. Communication: Does a single CH3CN molecule attached to Ru(bipy)32+ affect its absorption spectrum?. J. Chem. Phys. 2015, 142, 171102, DOI: 10.1063/1.4919781Google ScholarThere is no corresponding record for this reference.
- 132Stark, C. W.; Trummal, A.; Uudsemaa, M.; Pahapill, J.; Rammo, M.; Petritsenko, K.; Sildoja, M.-M.; Rebane, A. Solute-solvent electronic interaction is responsible for initial charge separation in ruthenium complexes [Ru(bpy)3]2+ and [Ru(phen)3]2+. Commun. Chem. 2019, 2, 108, DOI: 10.1038/s42004-019-0213-5Google ScholarThere is no corresponding record for this reference.
- 133Pelczarski, D.; Korolevych, O.; Gierczyk, B.; Zalas, M.; Makowska-Janusik, M.; Stampor, W. Electronic States of Tris(bipyridine) Ruthenium(II) Complexes in Neat Solid Films Investigated by Electroabsorption Spectroscopy. Materials 2022, 15, 2278, DOI: 10.3390/ma15062278Google ScholarThere is no corresponding record for this reference.
- 134Yersin, H.; Humbs, W.; Strasser, J. Low-lying electronic states of [Rh(bpy)3]3+, [Pt(bpy)2]2+, and [Ru(bpy)3]2+. A comparative study based on highly resolved and time-resolved spectra. Coord. Chem. Rev. 1997, 159, 325– 358, DOI: 10.1016/S0010-8545(96)01318-5Google ScholarThere is no corresponding record for this reference.
- 135Visco, R. E.; Chandross, E. A. Electroluminescence in Solutions of Aromatic Hydrocarbons. J. Am. Chem. Soc. 1964, 86, 5350– 5351, DOI: 10.1021/ja01077a073Google ScholarThere is no corresponding record for this reference.
- 136Hercules, D. M. Chemiluminescence Resulting from Electrochemically Generated Species. Science 1964, 145, 808– 809, DOI: 10.1126/science.145.3634.808Google ScholarThere is no corresponding record for this reference.
- 137Santhanam, K. S. V.; Bard, A. J. Chemiluminescence of Electrogenerated 9, 10-Diphenylanthracene Anion Radical1. J. Am. Chem. Soc. 1965, 87, 139– 140, DOI: 10.1021/ja01079a039Google ScholarThere is no corresponding record for this reference.
- 138Lemon, H. B. Luminosity of Rectifier Electrode. Science 1918, 47, 170, DOI: 10.1126/science.47.1207.170-bGoogle ScholarThere is no corresponding record for this reference.
- 139Dufford, R. T.; Nightingale, D.; Gaddum, L. W. Luminescence of Grignard compounds in electric and magnetic fields, and related electrical phenomena. J. Am. Chem. Soc. 1927, 49, 1858– 1864, DOI: 10.1021/ja01407a002Google ScholarThere is no corresponding record for this reference.
- 140Dufford, R. T. Luminescence Associated with Electrolysis. J. Opt. Soc. Am. 1929, 18, 17, DOI: 10.1364/JOSA.18.000017Google ScholarThere is no corresponding record for this reference.
- 141Harvey, N. Luminescence during Electrolysis. J. Phys. Chem. 1929, 33, 1456– 1459, DOI: 10.1021/j150304a002Google ScholarThere is no corresponding record for this reference.
- 142Hercules, D. M.; Lytle, F. E. Chemiluminescence from Reduction Reactions. J. Am. Chem. Soc. 1966, 88, 4745– 4746, DOI: 10.1021/ja00972a052Google ScholarThere is no corresponding record for this reference.
- 143
Indeed, the same year which saw first publication of photoinduced electron transfer studies in [Ru(bpy)3]2+, as well as the landmark paper on water-splitting on TiO2 by Fujishima and Honda.
There is no corresponding record for this reference. - 144Tokel, N. E.; Bard, A. J. Electrogenerated chemiluminescence. IX. Electrochemistry and emission from systems containing tris(2, 2′-bipyridine)ruthenium(II) dichloride. J. Am. Chem. Soc. 1972, 94, 2862– 2863, DOI: 10.1021/ja00763a056Google ScholarThere is no corresponding record for this reference.
- 145Tokel-Takvoryan, N. E.; Hemingway, R. E.; Bard, A. J. Electrogenerated chemiluminescence. XIII. Electrochemical and electrogenerated chemiluminescence studies of ruthenium chelates. J. Am. Chem. Soc. 1973, 95, 6582– 6589, DOI: 10.1021/ja00801a011Google ScholarThere is no corresponding record for this reference.
- 146Ohsawa, Y.; DeArmond, M. K.; Hanck, K. W.; Morris, D. E.; Whitten, D. G.; Neveux, P. E. Spatially isolated redox orbitals: evidence from low-temperature voltammetry. J. Am. Chem. Soc. 1983, 105, 6522– 6524, DOI: 10.1021/ja00359a045Google ScholarThere is no corresponding record for this reference.
- 147Vlcek, A. Ligand based redox series. Coord. Chem. Rev. 1982, 43, 39– 62, DOI: 10.1016/S0010-8545(00)82091-3Google ScholarThere is no corresponding record for this reference.
- 148Gorman, B. A.; Francis, P. S.; Barnett, N. W. Tris(2, 2′-bipyridyl)ruthenium(ii) chemiluminescence. Analyst 2006, 131, 616– 639, DOI: 10.1039/B518454AGoogle ScholarThere is no corresponding record for this reference.
- 149Villani, E.; Sakanoue, K.; Einaga, Y.; Inagi, S.; Fiorani, A. Photophysics and electrochemistry of ruthenium complexes for electrogenerated chemiluminescence. J. Electroanal. Chem. 2022, 921, 116677 DOI: 10.1016/j.jelechem.2022.116677Google ScholarThere is no corresponding record for this reference.
- 150Deaver, D. R. A new non-isotopic detection system for immunoassays. Nature 1995, 377, 758– 760, DOI: 10.1038/377758a0Google ScholarThere is no corresponding record for this reference.
- 151Fracassa, A.; Ferrari, G.; Balli, M. V.; Rimoldi, I.; Facchetti, G.; Arnal, L.; Marconi, A.; Calvaresi, M.; Prodi, L.; De Cola, L.; Valenti, G. Stimuli-Responsive Luminophore Drives Mechanism Switch for Highly Efficient Electrochemiluminescence Immunosensing. J. Am. Chem. Soc. 2025, 147, 35501, DOI: 10.1021/jacs.5c10211Google ScholarThere is no corresponding record for this reference.
- 152Ashton, P. R.; Ballardini, R.; Balzani, V.; Constable, E. C.; Credi, A.; Kocian, O.; Langford, S. J.; Preece, J. A.; Prodi, L.; Schofield, E. R.; Spencer, N.; Stoddart, J. F.; Wenger, S. RuII-Polypyridine Complexes Covalently Linked to Electron Acceptors as Wires for Light-Driven Pseudorotaxane-Type Molecular Machines. Chem. – Eur. J. 1998, 4, 2413– 2422, DOI: 10.1002/(SICI)1521-3765(19981204)4:12<2413::AID-CHEM2413>3.0.CO;2-AGoogle ScholarThere is no corresponding record for this reference.
- 153Ballardini, R.; Balzani, V.; Credi, A.; Gandolfi, M. T.; Venturi, M. Artificial molecular-level machines with [Ru(bpy)3]2+ as a “light-fueled motor”. International Journal of Photoenergy 2001, 3, 63– 77, DOI: 10.1155/S1110662X01000083Google ScholarThere is no corresponding record for this reference.
- 154Ceroni, P.; Bergamini, G.; Balzani, V. Old Molecules, New Concepts: [Ru(bpy)3]2+ as a Molecular Encoder–Decoder. Angew. Chem., Int. Ed. 2009, 48, 8516– 8518, DOI: 10.1002/anie.200904764Google ScholarThere is no corresponding record for this reference.
- 155Bolletta, F.; Balzani, V. Oscillating chemiluminescence from the reduction of bromate by malonic acid catalyzed by tris(2, 2′-bipyridine)ruthenium(II). J. Am. Chem. Soc. 1982, 104, 4250– 4251, DOI: 10.1021/ja00379a036Google ScholarThere is no corresponding record for this reference.
- 156Gerischer, H. Electrochemical photo and solar cells principles and some experiments. Journal of Electroanalytical Chemistry and Interfacial Electrochemistry 1975, 58, 263– 274, DOI: 10.1016/S0022-0728(75)80359-7Google ScholarThere is no corresponding record for this reference.
- 157Gleria, M.; Memming, R. Photoelectrochemical Studies of tris [2, 2′-bipyridyl]ruthenium(II) at Semiconductor Electrodes. Zeitschrift für Physikalische Chemie 1975, 98, 303– 316, DOI: 10.1524/zpch.1975.98.1-6.303Google ScholarThere is no corresponding record for this reference.
- 158Akimov, I. A. Optical sensibilization of the photoeffect in silver bromide and thallium bromide by organic dyes. Dokl. Akad. Nauk SSSR 1958, 121, 311– 314Google ScholarThere is no corresponding record for this reference.
- 159Dudkowski, S. J.; Grossweiner, L. I. Sensitization of Photoconduction in a Zinc Oxide Film by Eosin. J. Opt. Soc. Am. 1964, 54, 486, DOI: 10.1364/JOSA.54.000486Google ScholarThere is no corresponding record for this reference.
- 160Inoue, E.; Kokado, H.; Yamaguchi, T. Hypersensitization of Photoconduction in Microcrystalline Zinc Oxide1. J. Phys. Chem. 1965, 69, 767– 774, DOI: 10.1021/j100887a009Google ScholarThere is no corresponding record for this reference.
- 161Gerischer, H.; Michel-Beyerle, M.; Rebentrost, F.; Tributsch, H. Sensitization of charge injection into semiconductors with large band gap. Electrochim. Acta 1968, 13, 1509– 1515, DOI: 10.1016/0013-4686(68)80076-3Google ScholarThere is no corresponding record for this reference.
- 162Tributsch, H.; Gerischer, H. The use of semiconductor electrodes in the study of photochemical reactions. Berichte der Bunsengesellschaft f ü r physikalische Chemie 1969, 73, 850– 854, DOI: 10.1002/bbpc.19690730821Google ScholarThere is no corresponding record for this reference.
- 163Tributsch, H.; Calvin, M. Electrochemistry of excited molecules: photo-electrochemical reactions of chlorophylls. Photochem. Photobiol. 1971, 14, 95– 112, DOI: 10.1111/j.1751-1097.1971.tb06156.xGoogle ScholarThere is no corresponding record for this reference.
- 164Terenin, A.; Putzeiko, E. Sensibilisation optique des semi-conducteurs par la chlorophylle et pigments apparentés. Journal de Chimie Physique 1958, 55, 681– 687, DOI: 10.1051/jcp/1958550681Google ScholarThere is no corresponding record for this reference.
- 165
It is worth mentioning here that Gleria and Memming also further investigated [Ru(bpy)3]2+ ECL with a focus on the semiconductor electrodes used.263
There is no corresponding record for this reference. - 166Osa, T.; Fujihira, M. Photocell using covalently-bound dyes on semiconductor surfaces. Nature 1976, 264, 349– 350, DOI: 10.1038/264349a0Google ScholarThere is no corresponding record for this reference.
- 167Wrighton, M. S. Photoelectrochemical conversion of optical energy to electricity and fuels. Acc. Chem. Res. 1979, 12, 303– 310, DOI: 10.1021/ar50141a001Google ScholarThere is no corresponding record for this reference.
- 168Clark, W. D. K.; Sutin, N. Spectral sensitization of n-type titanium dioxide electrodes by polypyridineruthenium(II) complexes. J. Am. Chem. Soc. 1977, 99, 4676– 4682, DOI: 10.1021/ja00456a025Google ScholarThere is no corresponding record for this reference.
- 169Hamnett, A.; Dare-Edwards, M. P.; Wright, R. D.; Seddon, K. R.; Goodenough, J. B. Photosensitization of titanium(IV) oxide with tris(2, 2′-bipyridine)ruthenium(II) chloride. Surface states of titanium(IV) oxide. J. Phys. Chem. 1979, 83, 3280– 3290, DOI: 10.1021/j100488a020Google ScholarThere is no corresponding record for this reference.
- 170Anderson, S.; Constable, E. C.; Dare-Edwards, M. P.; Goodenough, J. B.; Hamnett, A.; Seddon, K. R.; Wright, R. D. Chemical modification of a titanium (IV) oxide electrode to give stable dye sensitisation without a supersensitiser. Nature 1979, 280, 571– 573, DOI: 10.1038/280571a0Google ScholarThere is no corresponding record for this reference.
- 171Dare-Edwards, M. P.; Goodenough, J. B.; Hamnett, A.; Seddon, K. R.; Wright, R. D. Sensitisation of semiconducting electrodes with ruthenium-based dyes. Faraday Discuss. Chem. Soc. 1980, 70, 285, DOI: 10.1039/dc9807000285Google ScholarThere is no corresponding record for this reference.
- 172Memming, R.; Schröppel, F. Electron transfer reactions of excited ruthenium(II) complexes in monolayer assemblies at the SnO2-water interface. Chem. Phys. Lett. 1979, 62, 207– 210, DOI: 10.1016/0009-2614(79)80160-8Google ScholarThere is no corresponding record for this reference.
- 173Desilvestro, J.; Graetzel, M.; Kavan, L.; Moser, J.; Augustynski, J. Highly efficient sensitization of titanium dioxide. J. Am. Chem. Soc. 1985, 107, 2988– 2990, DOI: 10.1021/ja00296a035Google ScholarThere is no corresponding record for this reference.
- 174Vlachopoulos, N.; Liska, P.; Augustynski, J.; Graetzel, M. Very efficient visible light energy harvesting and conversion by spectral sensitization of high surface area polycrystalline titanium dioxide films. J. Am. Chem. Soc. 1988, 110, 1216– 1220, DOI: 10.1021/ja00212a033Google ScholarThere is no corresponding record for this reference.
- 175Nazeeruddin, M. K.; Liska, P.; Moser, J.; Vlachopoulos, N.; Grätzel, M. Conversion of Light into Electricity with Trinuclear Ruthenium Complexes Adsorbed on Textured TiO2 Films. Helv. Chim. Acta 1990, 73, 1788– 1803, DOI: 10.1002/hlca.19900730624Google ScholarThere is no corresponding record for this reference.
- 176O’Regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737– 740, DOI: 10.1038/353737a0Google ScholarThere is no corresponding record for this reference.
- 177Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-Sensitized Solar Cells. Chem. Rev. 2010, 110, 6595– 6663, DOI: 10.1021/cr900356pGoogle ScholarThere is no corresponding record for this reference.
- 178Muñoz-García, A. B.; Benesperi, I.; Boschloo, G.; Concepcion, J. J.; Delcamp, J. H.; Gibson, E. A.; Meyer, G. J.; Pavone, M.; Pettersson, H.; Hagfeldt, A.; Freitag, M. Dye-sensitized solar cells strike back. Chem. Soc. Rev. 2021, 50, 12450– 12550, DOI: 10.1039/D0CS01336FGoogle ScholarThere is no corresponding record for this reference.
- 179Vougioukalakis, G. C.; Philippopoulos, A. I.; Stergiopoulos, T.; Falaras, P. Contributions to the development of ruthenium-based sensitizers for dye-sensitized solar cells. Coord. Chem. Rev. 2011, 255, 2602– 2621, DOI: 10.1016/j.ccr.2010.11.006Google ScholarThere is no corresponding record for this reference.
- 180Basheer, B.; Mathew, D.; George, B. K.; Reghunadhan Nair, C. An overview on the spectrum of sensitizers: The heart of Dye Sensitized Solar Cells. Sol. Energy 2014, 108, 479– 507, DOI: 10.1016/j.solener.2014.08.002Google ScholarThere is no corresponding record for this reference.
- 181Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. Light induced and dye accelerated reductions of phenacyl onium salts by 1, 4-dihydropyridines. Tetrahedron Lett. 1978, 19, 1255– 1258, DOI: 10.1016/S0040-4039(01)94515-0Google ScholarThere is no corresponding record for this reference.
- 182Van Bergen, T. J.; Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. Chemistry of dihydropyridines. 9. Hydride transfer from 1, 4-dihydropyridines to sp3-hybridized carbon in sulfonium salts and activated halides. Studies with NAD(P)H models. Journal of Organic Chemistry 1979, 44, 4953– 4962, DOI: 10.1021/jo00394a044Google ScholarThere is no corresponding record for this reference.
- 183DeLaive, P. J.; Giannotti, C.; Whitten, D. G. Applications of photoinduced electron-transfer reactions: visible light mediated redox reactions of substrates using transition metal complexes as catalysts. J. Am. Chem. Soc. 1978, 100, 7413– 7415, DOI: 10.1021/ja00491a049Google ScholarThere is no corresponding record for this reference.
- 184DeLaive, P. J.; Foreman, T. K.; Giannotti, C.; Whitten, D. G. Photoinduced electron transfer reactions of transition-metal complexes with amines. Mechanistic studies of alternate pathways to back electron transfer. J. Am. Chem. Soc. 1980, 102, 5627– 5631, DOI: 10.1021/ja00537a037Google ScholarThere is no corresponding record for this reference.
- 185Cano-Yelo, H.; Deronzier, A. Photo-oxidation of tris(2, 2′-bipyridine)ruthenium(II) by para-substituted benzene diazonium salts in acetonitrile. Two-compartment photoelectrochemical cell applications. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases 1984, 80, 3011, DOI: 10.1039/f19848003011Google ScholarThere is no corresponding record for this reference.
- 186Hironaka, K.; Fukuzumi, S.; Tanaka, T. Tris(bipyridyl)ruthenium(II)-photosensitized reaction of 1-benzyl-1, 4-dihydronicotinamide with benzyl bromide. J. Chem. Soc., Perkin Trans. 1984, 2, 1705, DOI: 10.1039/p29840001705Google ScholarThere is no corresponding record for this reference.
- 187Okada, K.; Okamoto, K.; Oda, M. A new and practical method of decarboxylation: photosensitized decarboxylation of N-acyloxyphthalimides via electron-transfer mechanism. J. Am. Chem. Soc. 1988, 110, 8736– 8738, DOI: 10.1021/ja00234a047Google ScholarThere is no corresponding record for this reference.
- 188Ischay, M. A.; Anzovino, M. E.; Du, J.; Yoon, T. P. Efficient Visible Light Photocatalysis of [2 + 2] Enone Cycloadditions. J. Am. Chem. Soc. 2008, 130, 12886– 12887, DOI: 10.1021/ja805387fGoogle ScholarThere is no corresponding record for this reference.
- 189Nicewicz, D. A.; MacMillan, D. W. C. Merging Photoredox Catalysis with Organocatalysis: The Direct Asymmetric Alkylation of Aldehydes. Science 2008, 322, 77– 80, DOI: 10.1126/science.1161976Google ScholarThere is no corresponding record for this reference.
- 190Narayanam, J. M. R.; Tucker, J. W.; Stephenson, C. R. J. Electron-Transfer Photoredox Catalysis: Development of a Tin-Free Reductive Dehalogenation Reaction. J. Am. Chem. Soc. 2009, 131, 8756– 8757, DOI: 10.1021/ja9033582Google ScholarThere is no corresponding record for this reference.
- 191Merck The brilliant history of photoredox catalysis. Nat. Portfolio.Google ScholarThere is no corresponding record for this reference.
- 192Shaw, M. H.; Twilton, J.; MacMillan, D. W. C. Photoredox Catalysis in Organic Chemistry. Journal of Organic Chemistry 2016, 81, 6898– 6926, DOI: 10.1021/acs.joc.6b01449Google ScholarThere is no corresponding record for this reference.
- 193Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. I. General toxicity. Boll. - Soc. Ital. Biol. Sper. 1938, 13, 6– 8Google ScholarThere is no corresponding record for this reference.
- 194Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. II. Distribution in the organism. Boll. - Soc. Ital. Biol. Sper. 1938, 13, 8– 11Google ScholarThere is no corresponding record for this reference.
- 195Beccari, E. Pharmacological studies on the ferrous tri-α,α’-bipyridyl complex. III. Further experiments on distribution in the tissues. Boll. - Soc. Ital. Biol. Sper. 1941, 16, 214– 216Google ScholarThere is no corresponding record for this reference.
- 196Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. IV. Relation between concentration in the blood and action on the central nervous system. Boll. - Soc. Ital. Biol. Sper. 1941, 16, 216– 218Google ScholarThere is no corresponding record for this reference.
- 197Dwyer et al. do not reference the works by Beccari in their first paper on the biological activity of complex ions, but they do reference his work in later publications.Google ScholarThere is no corresponding record for this reference.
- 198Dwyer, F. P.; Gyarfas, E. C.; O’Dwyer, M. F. Configurational Activity. Nature 1951, 167, 1036– 1036, DOI: 10.1038/1671036a0Google ScholarThere is no corresponding record for this reference.
- 199Dwyer, F. P.; Gyarfas, E. C.; Rogers, W. P.; Koch, J. H. Biological Activity of Complex Ions. Nature 1952, 170, 190– 191, DOI: 10.1038/170190a0Google ScholarThere is no corresponding record for this reference.
- 200Lindoy, L. F. Celebration of inorganic lives: Interview with Alan M. Sargeson. Coordination Chemistry Reviews 2005, 249, 2731– 2739, DOI: 10.1016/j.ccr.2005.04.015Google ScholarThere is no corresponding record for this reference.
- 201Meggers, E. Exploring biologically relevant chemical space with metal complexes. Curr. Opin. Chem. Biol. 2007, 11, 287– 292, DOI: 10.1016/j.cbpa.2007.05.013Google ScholarThere is no corresponding record for this reference.
- 202Levina, A.; Mitra, A.; Lay, P. A. Recent developments in ruthenium anticancer drugs. Metallomics 2009, 1, 458, DOI: 10.1039/b904071dGoogle ScholarThere is no corresponding record for this reference.
- 203Kilah, N. L.; Meggers, E. Sixty Years Young: The Diverse Biological Activities of Metal Polypyridyl Complexes Pioneered by Francis P. Dwyer. Aust. J. Chem. 2012, 65, 1325, DOI: 10.1071/CH12275Google ScholarThere is no corresponding record for this reference.
- 204Lee, S. Y.; Kim, C. Y.; Nam, T.-G. Ruthenium Complexes as Anticancer Agents: A Brief History and Perspectives. Drug Design, Development and Therapy 2020, 14, 5375– 5392, DOI: 10.2147/DDDT.S275007Google ScholarThere is no corresponding record for this reference.
- 205Koch, J. H.; Gyarfas, E. C.; Dwyer, F. Biological Activity of Complex Ions Mechanism of Inhibition of Acetylcholinesterase. Aust. J. Biol. Sci. 1956, 9, 371, DOI: 10.1071/BI9560371Google ScholarThere is no corresponding record for this reference.
- 206Koch, J. H.; Rogers, W.; Dwyer, F.; Gyarfas, E. C. The Metabolic Fate of Tris-1, 10-Phenanthroline 106Ruthenium (II) Perchlorate, a Compound With Anticholinesterase and Curare-Like Activity. Aust. J. Biol. Sci. 1957, 10, 342, DOI: 10.1071/BI9570342Google ScholarThere is no corresponding record for this reference.
- 207Dwyer, F. P.; Gyarfas, E. C.; Wright, R. D.; Shulman, A. Effect of Inorganic Complex Ions on Transmission at a Neuromuscular Junction. Nature 1957, 179, 425– 426, DOI: 10.1038/179425a0Google ScholarThere is no corresponding record for this reference.
- 208Dwyer, F.; Reid, I.; Shulman, A.; Laycock, G. M.; Dixson, S. The biological actions of 1, 10-phenanthroline and 2, 2'-bipyridine hydrochlorides, quaternary salts and metal chelates and related compounds: 1. Bacteriostatic action on selected Gram-positive, Gram-negative and acid-fast bacteria. Australian Journal of Experimental Biology and Medical Science 1969, 47, 203– 218, DOI: 10.1038/icb.1969.21Google ScholarThere is no corresponding record for this reference.
- 209White, D. O.; Harris, A. W.; Dwyer, F. P.; Shulman, A. Actions of metal chelates of substituted 1, 10-phenanthrolines on viruses and cells: I. Inactivation of viruses. Australian Journal of Experimental Biology and Medical Science 1963, 41, 517– 526, DOI: 10.1038/icb.1963.42Google ScholarThere is no corresponding record for this reference.
- 210Rosenberg, B.; Van Camp, L.; Krigas, T. Inhibition of Cell Division in Escherichia coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698– 699, DOI: 10.1038/205698a0Google ScholarThere is no corresponding record for this reference.
- 211Alderden, R. A.; Hall, M. D.; Hambley, T. W. The Discovery and Development of Cisplatin. J. Chem. Educ. 2006, 83, 728, DOI: 10.1021/ed083p728Google ScholarThere is no corresponding record for this reference.
- 212Diamond, I.; Mcdonagh, A.; Wilson, C.; Granelli, S.; Nielsen, S.; Jaenicke, R. Photodynamic therapy of malignant tumors. Lancet 1972, 300, 1175– 1177, DOI: 10.1016/S0140-6736(72)92596-2Google ScholarThere is no corresponding record for this reference.
- 213Kessel, D. Photodynamic Therapy: A Brief History. Journal of Clinical Medicine 2019, 8, 1581, DOI: 10.3390/jcm8101581Google ScholarThere is no corresponding record for this reference.
- 214Knoll, J. D.; Turro, C. Control and utilization of ruthenium and rhodium metal complex excited states for photoactivated cancer therapy. Coord. Chem. Rev. 2015, 282–283, 110– 126, DOI: 10.1016/j.ccr.2014.05.018Google ScholarThere is no corresponding record for this reference.
- 215Heinemann, F.; Karges, J.; Gasser, G. Critical Overview of the Use of Ru(II) Polypyridyl Complexes as Photosensitizers in One-Photon and Two-Photon Photodynamic Therapy. Acc. Chem. Res. 2017, 50, 2727– 2736, DOI: 10.1021/acs.accounts.7b00180Google ScholarThere is no corresponding record for this reference.
- 216Gandosio, A.; Purkait, K.; Gasser, G. Recent Approaches towards the Development of Ru(II) Polypyridyl Complexes for Anticancer Photodynamic Therapy. CHIMIA 2021, 75, 845, DOI: 10.2533/chimia.2021.845Google ScholarThere is no corresponding record for this reference.
- 217Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. I. The Journal of Chemical Physics 1956, 24, 966– 978, DOI: 10.1063/1.1742723Google ScholarThere is no corresponding record for this reference.
- 218Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. II. Applications to Data on the Rates of Isotopic Exchange Reactions. J. Chem. Phys. 1957, 26, 867– 871, DOI: 10.1063/1.1743423Google ScholarThere is no corresponding record for this reference.
- 219Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. III. Applications to Data on the Rates of Organic Redox Reactions. J. Chem. Phys. 1957, 26, 872– 877, DOI: 10.1063/1.1743424Google ScholarThere is no corresponding record for this reference.
- 220Chance, B.; Nishimura, M. On the mechanism of chlorophyll-cytochrome interaction: the temperature insensitivity of light-induced cytochrome oxidation in chromatium. Proc. Natl. Acad. Sci. U. S. A. 1960, 46, 19– 24, DOI: 10.1073/pnas.46.1.19Google ScholarThere is no corresponding record for this reference.
- 221Mitchell, P. Coupling of Phosphorylation to Electron and Hydrogen Transfer by a Chemi-Osmotic type of Mechanism. Nature 1961, 191, 144– 148, DOI: 10.1038/191144a0Google ScholarThere is no corresponding record for this reference.
- 222Vredenberg, W.; Duysens, L. Light-induced oxidation of cytochromes in photosynthetic bacteria between 20 and −170°. Biochim. Biophys. Acta, Spec. Sect. Biophys. Subj. 1964, 79, 456– 463, DOI: 10.1016/0926-6577(64)90211-6Google ScholarThere is no corresponding record for this reference.
- 223De Vault, D.; Chance, B. Studies of Photosynthesis Using a Pulsed Laser. Biophys. J. 1966, 6, 825– 847, DOI: 10.1016/S0006-3495(66)86698-5Google ScholarThere is no corresponding record for this reference.
- 224Sutin, N. Bioinorganic Chemistry─II; American Chemical Society: 1977; pp 156– 172.Google ScholarThere is no corresponding record for this reference.
- 225English, A. M.; DeLaive, P. J.; Gray, H. B.; Lum, V. R. Metalloprotein electron-transfer mechanisms. Quenching of electronically excited tris(2, 2′-bipyridine)ruthenium(II) by reduced blue copper proteins. J. Am. Chem. Soc. 1982, 104, 870– 871, DOI: 10.1021/ja00367a047Google ScholarThere is no corresponding record for this reference.
- 226Dawson, J. W.; Gray, H. B.; Holwerda, R. A.; Westhead, E. W. Kinetics of the Reduction of Metalloproteins by Chromous Ion. Proc. Natl. Acad. Sci. U. S. A. 1972, 69, 30– 33, DOI: 10.1073/pnas.69.1.30Google ScholarThere is no corresponding record for this reference.
- 227Hodges, H. L.; Holwerda, R. A.; Gray, H. B. Kinetic studies of the reduction of ferricytochrome c by ethylenediaminetetraacetatoiron(II). J. Am. Chem. Soc. 1974, 96, 3132– 3137, DOI: 10.1021/ja00817a019Google ScholarThere is no corresponding record for this reference.
- 228Cummins, D.; Gray, H. B. Electron-transfer protein reactivities. Kinetic studies of the oxidation of horse heart cytochrome c, Chromatium vinosum high potential iron-sulfur protein, Pseudomonas aeruginosa azurin, bean plastocyanin, and Rhus vernicifera stellacyanin by pentaammminepyridineruthenium(III). J. Am. Chem. Soc. 1977, 99, 5158– 5167, DOI: 10.1021/ja00457a042Google ScholarThere is no corresponding record for this reference.
- 229Winkler, J. R.; Nocera, D. G.; Yocom, K. M.; Bordignon, E.; Gray, H. B. Electron-transfer kinetics of pentaammineruthenium(III)(histidine-33)-ferricytochrome c. Measurement of the rate of intramolecular electron transfer between redox centers separated by 15.ANG. in a protein. J. Am. Chem. Soc. 1982, 104, 5798– 5800, DOI: 10.1021/ja00385a047Google ScholarThere is no corresponding record for this reference.
- 230Winkler, J. R.; Gray, H. B. Electron transfer in ruthenium-modified proteins. Chem. Rev. 1992, 92, 369– 379, DOI: 10.1021/cr00011a001Google ScholarThere is no corresponding record for this reference.
- 231Marcus, R. A. Electron Transfer Reactions in Chemistry: Theory and Experiment (Nobel Lecture). Angewandte Chemie International Edition in English 1993, 32, 1111– 1121, DOI: 10.1002/anie.199311113Google ScholarThere is no corresponding record for this reference.
- 232Chang, I. J.; Gray, H. B.; Winkler, J. R. High-driving-force electron transfer in metalloproteins: intramolecular oxidation of ferrocytochrome c by Ru(2, 2′-bpy)2(im)(His-33)3+. J. Am. Chem. Soc. 1991, 113, 7056– 7057, DOI: 10.1021/ja00018a064Google ScholarThere is no corresponding record for this reference.
- 233Gray, H. B.; Winkler, J. R. Long-range electron transfer. Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 3534– 3539, DOI: 10.1073/pnas.0408029102Google ScholarThere is no corresponding record for this reference.
- 234Moser, C. C.; Keske, J. M.; Warncke, K.; Farid, R. S.; Dutton, P. L. Nature of biological electron transfer. Nature 1992, 355, 796– 802, DOI: 10.1038/355796a0Google ScholarThere is no corresponding record for this reference.
- 235Page, C.; Moser, C. C.; Dutton, P. L. Mechanism for electron transfer within and between proteins. Curr. Opin. Chem. Biol. 2003, 7, 551– 556, DOI: 10.1016/j.cbpa.2003.08.005Google ScholarThere is no corresponding record for this reference.
- 236Dempsey, J. L.; Winkler, J. R.; Gray, H. B. Proton-Coupled Electron Flow in Protein Redox Machines. Chem. Rev. 2010, 110, 7024– 7039, DOI: 10.1021/cr100182bGoogle ScholarThere is no corresponding record for this reference.
- 237Utterback, J. K.; Ruzicka, J. L.; Keller, H. R.; Pellows, L. M.; Dukovic, G. Electron Transfer from Semiconductor Nanocrystals to Redox Enzymes. Annu. Rev. Phys. Chem. 2020, 71, 335– 359, DOI: 10.1146/annurev-physchem-050317-014232Google ScholarThere is no corresponding record for this reference.
- 238Purugganan, M. D.; Kumar, C. V.; Turro, N. J.; Barton, J. K. Accelerated Electron Transfer Between Metal Complexes Mediated by DNA. Science 1988, 241, 1645– 1649, DOI: 10.1126/science.241.4873.1645Google ScholarThere is no corresponding record for this reference.
- 239Lippard, S. J. Platinum complexes: probes of polynucleotide structure and antitumor drugs. Acc. Chem. Res. 1978, 11, 211– 217, DOI: 10.1021/ar50125a006Google ScholarThere is no corresponding record for this reference.
- 240Barton, J. K.; Danishefsky, A.; Goldberg, J. Tris(phenanthroline)ruthenium(II): stereoselectivity in binding to DNA. J. Am. Chem. Soc. 1984, 106, 2172– 2176, DOI: 10.1021/ja00319a043Google ScholarThere is no corresponding record for this reference.
- 241Friedman, A. E.; Chambron, J. C.; Sauvage, J. P.; Turro, N. J.; Barton, J. K. A molecular light switch for DNA: Ru(bpy)2(dppz)2+. J. Am. Chem. Soc. 1990, 112, 4960– 4962, DOI: 10.1021/ja00168a052Google ScholarThere is no corresponding record for this reference.
- 242Li, G.; Sun, L.; Ji, L.; Chao, H. Ruthenium(ii) complexes with dppz: from molecular photoswitch to biological applications. Dalton Transactions 2016, 45, 13261– 13276, DOI: 10.1039/C6DT01624CGoogle ScholarThere is no corresponding record for this reference.
- 243Giese, B. Long-Distance Electron Transfer Through DNA. Annu. Rev. Biochem. 2002, 71, 51– 70, DOI: 10.1146/annurev.biochem.71.083101.134037Google ScholarThere is no corresponding record for this reference.
- 244Barton, J. K.; Kumar, C. V.; Turro, N. J. DNA-mediated photoelectron transfer reactions. J. Am. Chem. Soc. 1986, 108, 6391– 6393, DOI: 10.1021/ja00280a047Google ScholarThere is no corresponding record for this reference.
- 245Barton, J. K.; Olmon, E. D.; Sontz, P. A. Metal complexes for DNA-mediated charge transport. Coord. Chem. Rev. 2011, 255, 619– 634, DOI: 10.1016/j.ccr.2010.09.002Google ScholarThere is no corresponding record for this reference.
- 246Taniguchi, M.; Kawai, T. DNA electronics. Physica E: Low-dimensional Systems and Nanostructures 2006, 33, 1– 12, DOI: 10.1016/j.physe.2006.01.005Google ScholarThere is no corresponding record for this reference.
- 247Fancy, D. A.; Kodadek, T. Chemistry for the analysis of protein–protein interactions: Rapid and efficient cross-linking triggered by long wavelength light. Proc. Natl. Acad. Sci. U. S. A. 1999, 96, 6020– 6024, DOI: 10.1073/pnas.96.11.6020Google ScholarThere is no corresponding record for this reference.
- 248Preston, G. W.; Wilson, A. J. Photo-induced covalent cross-linking for the analysis of biomolecular interactions. Chem. Soc. Rev. 2013, 42, 3289, DOI: 10.1039/c3cs35459hGoogle ScholarThere is no corresponding record for this reference.
- 249https://www.science.org/content/article/rubipy-toast-eminent-caltech-personality.Google ScholarThere is no corresponding record for this reference.
- 250
This statement has originated at the University of North Carolina at Chapel Hill, as confirmed by Professor Jillian Dempsey (UNC) via email on the 11th of February 2025.
There is no corresponding record for this reference. - 251Büldt, L. A.; Wenger, O. S. Chromium(0), Molybdenum(0), and Tungsten(0) Isocyanide Complexes as Luminophores and Photosensitizers with Long-Lived Excited States. Angew. Chem., Int. Ed. 2017, 56, 5676– 5682, DOI: 10.1002/anie.201701210Google ScholarThere is no corresponding record for this reference.
- 252Kjær, K. S. Luminescence and reactivity of a charge-transfer excited iron complex with nanosecond lifetime. Science 2019, 363, 249– 253, DOI: 10.1126/science.aau7160Google ScholarThere is no corresponding record for this reference.
- 253Reichenauer, F.; Wang, C.; Förster, C.; Boden, P.; Ugur, N.; Báez-Cruz, R.; Kalmbach, J.; Carrella, L. M.; Rentschler, E.; Ramanan, C.; Niedner-Schatteburg, G.; Gerhards, M.; Seitz, M.; Resch-Genger, U.; Heinze, K. Strongly Red-Emissive Molecular Ruby [Cr(bpmp)2]3+ Surpasses [Ru(bpy)3]2+. J. Am. Chem. Soc. 2021, 143, 11843– 11855, DOI: 10.1021/jacs.1c05971Google ScholarThere is no corresponding record for this reference.
- 254Brouwer, A. M. Standards for photoluminescence quantum yield measurements in solution (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 2213– 2228, DOI: 10.1351/PAC-REP-10-09-31Google ScholarThere is no corresponding record for this reference.
- 255Ripak, A.; De Kreijger, S.; Elias, B.; Troian-Gautier, L. A protocol for determining cage-escape yields using nanosecond transient absorption spectroscopy. STAR Protocols 2023, 4, 102312 DOI: 10.1016/j.xpro.2023.102312Google ScholarThere is no corresponding record for this reference.
- 256Müller, P.; Brettel, K. [Ru(bpy)3]2+ as a reference in transient absorption spectroscopy: differential absorption coefficients for formation of the long-lived 3MLCT excited state. Photochemical & Photobiological Sciences 2012, 11, 632– 636, DOI: 10.1039/c2pp05333kGoogle ScholarThere is no corresponding record for this reference.
- 257Arias-Rotondo, D. M.; McCusker, J. K. The photophysics of photoredox catalysis: a roadmap for catalyst design. Chem. Soc. Rev. 2016, 45, 5803– 5820, DOI: 10.1039/C6CS00526HGoogle ScholarThere is no corresponding record for this reference.
- 258Gaynor, J. D.; Petrone, A.; Li, X.; Khalil, M. Mapping Vibronic Couplings in a Solar Cell Dye with Polarization-Selective Two-Dimensional Electronic–Vibrational Spectroscopy. J. Phys. Chem. Lett. 2018, 9, 6289– 6295, DOI: 10.1021/acs.jpclett.8b02752Google ScholarThere is no corresponding record for this reference.
- 259Rosenfeld, L. Insulin: discovery and controversy. Clinical chemistry 2002, 48, 2270– 2288, DOI: 10.1093/clinchem/48.12.2270Google ScholarThere is no corresponding record for this reference.
- 260Burstall, F. H.; Dwyer, F. P.; Gyarfas, E. C. 194. Optical activity dependent on a six-covalent bivalent osmium complex. Journal of the Chemical Society 1950, 953, DOI: 10.1039/jr9500000953Google ScholarThere is no corresponding record for this reference.
- 261Vogler, A.; Adamson, A. W. Photosensitized decomposition of some cobalt ammines. J. Am. Chem. Soc. 1968, 90, 5943– 5945, DOI: 10.1021/ja01023a077Google ScholarThere is no corresponding record for this reference.
- 262Adamson, A. W.; Martin, J. E.; Camessei, F. D. Photosensitized aquation of some chromium(III) complexes. J. Am. Chem. Soc. 1969, 91, 7530– 7532, DOI: 10.1021/ja01054a064Google ScholarThere is no corresponding record for this reference.
- 263Gleria, M.; Memming, R. Novel Luminescence Generation by Electron Transfer from Semiconductor Electrodes to Ruthenium-Bipyridil Complexes. Zeitschrift f ü r Physikalische Chemie 1976, 101, 171– 179, DOI: 10.1524/zpch.1976.101.1-6.171Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 1 publications.
- Céline Bourgois, Ludovic Troian-Gautier, Winald R. Kitzmann. [Ru(bpy)3]2+: The Ongoing Story of a Photochemical Icon. Inorganic Chemistry 2026, Article ASAP.
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. (a) Chemical structure of [Ru(bpy)3]2+. (b) Web of Science output for “Tris(bipyridine)ruthenium” or “Tris(dipyridine)ruthenium” or “Ru(bpy)3” or “ruthenium trisbipyridine” or “Ru(dipy)3” or “Rubpy3” or “Rudipy3” or “Tris(2,2′-bipyridine) Ruthenium”, i.e., various monikers for [Ru(bpy)3]2+ over the past several decades. (c) Structures of the two enantiomers of [Ru(bpy)3]2+ reported by Burstall in 1936. Reproduced with permission from ref (2). Copyright 1936 Royal Society of Chemistry.
Figure 2
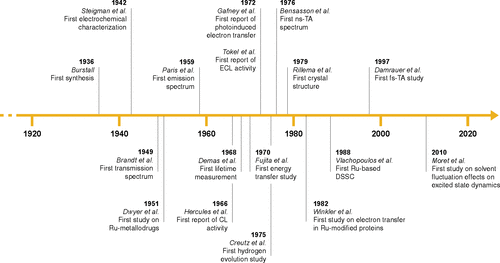
Figure 2. Timeline of the key publications in the history of [Ru(bpy)3]2+ (CL: chemiluminescence; ECL: electrochemiluminescence; TA: transient absorption; DSSC: dye-sensitized solar cell).
Figure 3
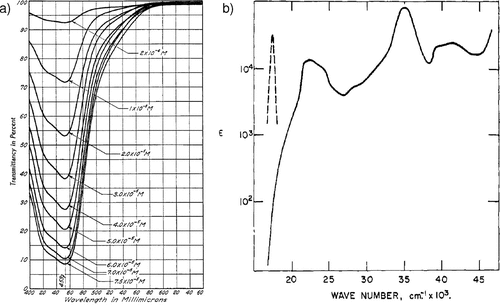
Figure 3. (a) First transmission spectrum of [Ru(bpy)3]2+ reported by Brandt et al. in 1949. Reproduced from ref (51). Copyright 1949 American Chemical Society. (b) First emission spectrum (dashed line) reported by Paris et al. in 1959. Reproduced from ref (52). Copyright 1959 American Chemical Society.
Figure 4

Figure 4. First crystal structure of [Ru(bpy)3]2+ reported by Rillema et al. in 1979. Reproduced with permission from ref (89). Copyright 1979 Royal Society of Chemistry.
Figure 5
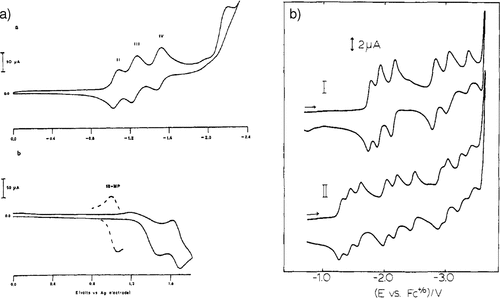
Figure 5. (a) First cyclic voltammogram of [Ru(bpy)3]2+ reported by Tokel and Bard in 1972. Reproduced from ref (144). Copyright 1972 American Chemical Society. (b) Low temperature cyclic voltammogram of (I) [Ru(bpy)3]2+ and (II) [Ru(4,4′-(CO2Et)2bpy)3]2+ reported by Ohsawa, DeArmond, Hanck et al. in 1983. Reproduced from ref (146). Copyright 1983 American Chemical Society.
Figure 6
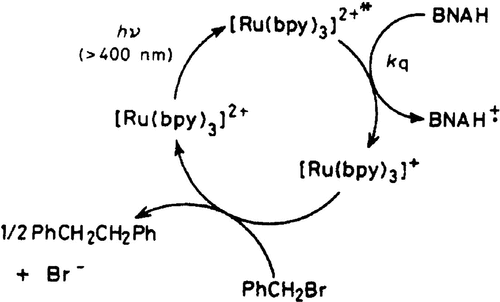
Figure 6. One of the earliest reported schemes for a photoredox reaction. In this process, [Ru(bpy)3]2+ is first excited, and then it is reductively quenched by 1-benzyl-l,4-dihydronicotinamide (BNAH). Finally, [Ru(bpy)3]+ reacts with benzyl bromide yielding 1,2-diphenylethane, the desired reaction product, and regenerating [Ru(bpy)3]2+. Reproduced with permission from ref (186). Copyright 1984 Royal Society of Chemistry.
Figure 7

Figure 7. (a) Tunneling timetable for intraprotein electron transfer in Ru-modified proteins. Reproduced with permission from ref (233). Copyright 2005 National Academy of Sciences, U.S.A. (b) Model for the interaction of Δ-[Ru(phen)3]2+ (left) and Λ-[Ru(phen)3]2+ (right) with the DNA double helix. Reproduced with permission from ref (238). Copyright 1988 American Association for the Advancement of Science.
Figure 8

Figure 8. License plate of Prof. Thomas Meyer (picture kindly provided by Tyler Meyer).
References
This article references 263 other publications.
- 1Arias-Rotondo, D. M. The fruit fly of photophysics. Nat. Chem. 2022, 14, 716– 716, DOI: 10.1038/s41557-022-00955-8There is no corresponding record for this reference.
- 2Burstall, F. H. 34. Optical activity dependent on co-ordinated bivalent ruthenium. Journal of the Chemical Society 1936, 173– 175, DOI: 10.1039/jr9360000173There is no corresponding record for this reference.
- 3Sutin, N. Light-induced electron transfer reactions. J. Photochem. 1979, 10, 19– 40, DOI: 10.1016/0047-2670(79)80035-0There is no corresponding record for this reference.
- 4Kalyanasundaram, K. Photophysics, photochemistry and solar energy conversion with tris(bipyridyl)ruthenium(II) and its analogues. Coord. Chem. Rev. 1982, 46, 159– 244, DOI: 10.1016/0010-8545(82)85003-0There is no corresponding record for this reference.
- 5De Armond, M. K.; Myrick, M. L. The life and times of [Ru(bpy)3]2+: localized orbitals and other strange occurrences. Acc. Chem. Res. 1989, 22, 364– 370, DOI: 10.1021/ar00166a004There is no corresponding record for this reference.
- 6Dongare, P.; Myron, B. D.; Wang, L.; Thompson, D. W.; Meyer, T. J. [Ru(bpy)3]2+ revisited. Is it localized or delocalized? How does it decay?. Coord. Chem. Rev. 2017, 345, 86– 107, DOI: 10.1016/j.ccr.2017.03.009There is no corresponding record for this reference.
- 7Bell, J. D.; Murphy, J. A. Recent advances in visible light-activated radical coupling reactions triggered by (i) ruthenium, (ii) iridium and (iii) organic photoredox agents. Chem. Soc. Rev. 2021, 50, 9540– 9685, DOI: 10.1039/D1CS00311AThere is no corresponding record for this reference.
- 8Wu, C. Ruthenium-based complex dyes for dye-sensitized solar cells. Journal of the Chinese Chemical Society 2022, 69, 1242– 1252, DOI: 10.1002/jccs.202200225There is no corresponding record for this reference.
- 9Ankathatti Munegowda, M.; Manalac, A.; Weersink, M.; McFarland, S. A.; Lilge, L. Ru(II) containing photosensitizers for photodynamic therapy: A critique on reporting and an attempt to compare efficacy. Coord. Chem. Rev. 2022, 470, 214712 DOI: 10.1016/j.ccr.2022.214712There is no corresponding record for this reference.
- 10Juris, A.; Balzani, V.; Barigelletti, F.; Campagna, S.; Belser, P.; von Zelewsky, A. Ru(II) polypyridine complexes: photophysics, photochemistry, eletrochemistry, and chemiluminescence. Coord. Chem. Rev. 1988, 84, 85– 277, DOI: 10.1016/0010-8545(88)80032-8There is no corresponding record for this reference.
- 11Constable, E. C.; Housecroft, C. E. The Early Years of 2, 2′-Bipyridine─A Ligand in Its Own Lifetime. Molecules 2019, 24, 3951, DOI: 10.3390/molecules24213951There is no corresponding record for this reference.
- 12Blau, F. Die Destillation pyridinmonocarbonsaurer Salze. Berichte der deutschen chemischen Gesellschaft 1888, 21, 1077– 1078, DOI: 10.1002/cber.188802101201There is no corresponding record for this reference.
- 13Blau, F. Uber neue organische Metallverbindungen. Monatshefte für Chemie 1898, 19, 647– 689, DOI: 10.1007/BF01517438There is no corresponding record for this reference.
- 14Miller, R. R.; Brandt, W. W.; Puke, M.; Puke, O. Metal-Amine Coördination Compounds. V. The Ruthenium-2, 2′-Bipyridine System. J. Am. Chem. Soc. 1955, 77, 3178– 3180, DOI: 10.1021/ja01617a003There is no corresponding record for this reference.
- 15Coulson, E. A. Dr. F.H. Burstall. Nature 1956, 177, 210– 211, DOI: 10.1038/177210a0There is no corresponding record for this reference.
- 16Gilbert Thomas Morgan, 1872–1940. Obituary Notices of Fellows of the Royal Society ; 1941; pp 354– 362.There is no corresponding record for this reference.
- 17Morgan, G. T.; Drew, H. D. K. CLXII.─Researches on residual affinity and co-ordination. Part II. Acetylacetones of selenium and tellurium. Journal of the Chemical Society, Transactions 1920, 117, 1456– 1465, DOI: 10.1039/CT9201701456There is no corresponding record for this reference.
- 18Morgan, G. T.; Burstall, F. H. 3. Dehydrogenation of pyridine by anhydrous ferric chloride. J. Chem. Soc. 1932, 1456– 1465, DOI: 10.1039/jr9320000020There is no corresponding record for this reference.
- 19Morgan, G.; Burstall, F. H. 347. Researches on residual affinity and co-ordination. Part XXXVII. Complex metallic salts containing 2:6-di-2′-pyridylpyridine (2:2′: 2″-tripyridyl). J. Chem. Soc. 1937, 1649– 1655, DOI: 10.1039/JR9370001649There is no corresponding record for this reference.
- 20Burstall, F. H. 312. Researches on the polypyridyls. Journal of the Chemical Society 1938, 1662, DOI: 10.1039/jr9380001662There is no corresponding record for this reference.
- 21Morgan, G. T.; Burstall, F. H. CCCXLIV.─Researches on residual affinity and co-ordination. Part XXXII. Complex salts of bivalent silver. Journal of the Chemical Society 1930, 0, 2594– 2598, DOI: 10.1039/JR9300002594There is no corresponding record for this reference.
- 22Morgan, G. T.; Burstall, F. H. CCCII.─Researches on residual affinity and co-ordination. Part XXXIII. Optical activity dependent on co-ordinated nickel. Journal of the Chemical Society 1931, 0, 2213– 2218, DOI: 10.1039/JR9310002213There is no corresponding record for this reference.
- 23Morgan, G. T.; Burstall, F. H. 201. Researches on residual affinity and co-ordination. Part XXXIV. 2:2′-Dipyridyl platinum salts. Journal of the Chemical Society 1934, 0, 965– 971, DOI: 10.1039/JR9340000965There is no corresponding record for this reference.
- 24Morgan, G. T.; Burstall, F. H. 323. Researches on residual affinity and co-ordination. Part XXXV. 2:2′: 2″-Tripyridylplatinum salts. Journal of the Chemical Society 1934, 0, 1498– 1500, DOI: 10.1039/JR9340001498There is no corresponding record for this reference.
- 25Werner, A. Über Spiegelbild-Isomerie bei Eisenverbindungen. Berichte der deutschen chemischen Gesellschaft 1912, 45, 433– 436, DOI: 10.1002/cber.19120450165There is no corresponding record for this reference.
- 26https://www.nobelprize.org/prizes/chemistry/1913/werner/facts/.There is no corresponding record for this reference.
- 27Baker, A. A. A History of Indicators. Chymia 1964, 9, 147– 167, DOI: 10.2307/27757238There is no corresponding record for this reference.
- 28Mellon, M. G. Fisher Award Address: A Century of Colorimetry. Anal. Chem. 1952, 24, 924– 931, DOI: 10.1021/ac60066a002There is no corresponding record for this reference.
- 29Snell, F. T. Colorimetric Analysis; D. Van Nostrand Company: 1921.There is no corresponding record for this reference.
- 30Johansson, A. The development of the titration methods. Anal. Chim. Acta 1988, 206, 97– 109, DOI: 10.1016/S0003-2670(00)80834-XThere is no corresponding record for this reference.
- 31Szabadvary, F. Indicators: A historical perspective. J. Chem. Educ. 1964, 41, 285– 287, DOI: 10.1021/ed041p285There is no corresponding record for this reference.
- 32Irving, H. M. N. H. Centenary lecture. One hundred years of development in Analytical Chemistry. Analyst 1974, 99, 787– 801, DOI: 10.1039/an9749900787There is no corresponding record for this reference.
- 33
Inventor of the isoelectric precipitation method for the improvement of insulin production.259
There is no corresponding record for this reference. - 34Walden, G. H.; Hammett, L. P.; Chapman, R. P. A reversible oxidation indicator of high potential especially adapted to oxidimetric titrations. J. Am. Chem. Soc. 1931, 53, 3908– 3908, DOI: 10.1021/ja01361a508There is no corresponding record for this reference.
- 35Brandt, W. W.; Dwyer, F. P.; Gyarfas, E. D. Chelate Complexes of 1, 10-Phenanthroline and Related Compounds. Chem. Rev. 1954, 54, 959– 1017, DOI: 10.1021/cr60172a003There is no corresponding record for this reference.
- 36Steigman, J.; Birnbaum, N.; Edmonds, S. Ruthenium Dipyridyl-New Oxidimetric Indicator. Industrial and Engineering Chemistry Analytical Edition 1942, 14, 30, DOI: 10.1021/i560101a010There is no corresponding record for this reference.
- 37Dwyer, F. P.; Nyholm, R. S. The instability constant of the tris-orthophenanthroline ferrous ion. Journal and Proceedings of the Royal Society of New South Wales 1947, 80, 28– 32, DOI: 10.5962/p.360430There is no corresponding record for this reference.
- 38Dwyer, F. P. The Chemistry of Ruthenium. Part III. The Redox Potentials of the Ruthenium II Complexes with Substituted Derivatives of 2:2′Dipyridyl and o-Phenanthroline. Journal and Proceedings of the Royal Society of New South Wales 1950, 83, 134– 137, DOI: 10.5962/p.360547There is no corresponding record for this reference.
- 39Baker, A. T.; Livingstone, S. E. Historical Sketch: The early history of coordination chemistry in Australia. Polyhedron 1985, 4, 1337– 1351, DOI: 10.1016/S0277-5387(00)86962-4There is no corresponding record for this reference.
- 40Minerals yearbook 1941–1959; United States Department of Interior, Bureau of Mines: 1941–1959.There is no corresponding record for this reference.
- 41
For reference, the Ru/Fe price ratio in 2024 is 110,000 and it was even lower in the 1990s.
There is no corresponding record for this reference. - 42Dwyer, F. P.; Humpoletz, J. E.; Nyholm, R. S. The Chemistry of Ruthenium. Part I. The Redox Potential of the Tris-Orthophenanthroline Ruthenous Ion. Journal and Proceedings of the Royal Society of New South Wales 1947, 80, 212– 216, DOI: 10.5962/p.360444There is no corresponding record for this reference.
- 43Backhouse, J.; Dwyer, F. P. The Chemistry of Ruthenium. Part VI. The Existence of the Tris-o- Phenanthroline Ruthenium III Ions in Entantiomorphous Forms. J. Proc. R. Soc. New South Wales 1949, 83, 170– 173, DOI: 10.5962/p.360552There is no corresponding record for this reference.
- 44Dwyer, F. P.; Gyarfas, E. C. The Chemistry of Ruthenium. Part VII. The Oxidation of D and L Tris 2:2′Dipyridyl Ruthenium II Iodide. Journal and Proceedings of the Royal Society of New South Wales 1950, 83, 174– 176, DOI: 10.5962/p.360553There is no corresponding record for this reference.
- 45Breckenridge, J. G.; Singer, S. A. G. 5-Hydroxyquinoline-8-carboxylic acid─A colorimetric reagent for ruthenium. Canadian Journal of Research 1947, 25b, 49– 55, DOI: 10.1139/cjr47b-006There is no corresponding record for this reference.
- 46Deford, D. The Chemistry of Ruthenium; Elsevier: 1948, DOI: 10.2172/4435343 .There is no corresponding record for this reference.
- 47Ayres, G. H.; Young, F. Spectrophotometric Study of Ruthenium-Thiourea Complex. Anal. Chem. 1950, 22, 1277– 1280, DOI: 10.1021/ac60046a014There is no corresponding record for this reference.
- 48Sheard, C.; States, M. N. A Concave Grating Photoelectric Spectrophotometer. J. Opt. Soc. Am. 1941, 31, 64, DOI: 10.1364/JOSA.31.000064There is no corresponding record for this reference.
- 49Thomas, N. C. The early history of spectroscopy. J. Chem. Educ. 1991, 68, 631, DOI: 10.1021/ed068p631There is no corresponding record for this reference.
- 50Karayannis, M. I.; Efstathiou, C. E. Significant steps in the evolution of analytical chemistry─Is the today’s analytical chemistry only chemistry?. Talanta 2012, 102, 7– 15, DOI: 10.1016/j.talanta.2012.06.003There is no corresponding record for this reference.
- 51Brandt, W. W.; Smith, G. F. Polysubstituted 1,10-Phenanthrolines and Bipyridines as Multiple Range Redox Indicators. Anal. Chem. 1949, 21, 1313– 1319, DOI: 10.1021/ac60035a003There is no corresponding record for this reference.
- 52Paris, J. P.; Brandt, W. W. Charge transfer luminescence of a ruthenium(II) chelate. J. Am. Chem. Soc. 1959, 81, 5001– 5002, DOI: 10.1021/ja01527a064There is no corresponding record for this reference.
- 53
We note that Burstall too remained active, and together with Dwyer reported the absorption spectrum of [Os(bpy)3]2+, the first for an osmium compound.260
There is no corresponding record for this reference. - 54Yamasaki, K. Absorptionsspektren von Metallkomplexsalzen des 2, 2′-Dipyridyls. I-IV. Bull. Chem. Soc. Jpn. 1937, 390– 394, DOI: 10.1246/bcsj.12.390There is no corresponding record for this reference.
- 55O’Haver, T. C. Development of luminescence spectrometry as an analytical tool. J. Chem. Educ. 1978, 55, 423, DOI: 10.1021/ed055p423There is no corresponding record for this reference.
- 56Valeur, B.; Berberan-Santos, M. N. A Brief History of Fluorescence and Phosphorescence before the Emergence of Quantum Theory. J. Chem. Educ. 2011, 88, 731– 738, DOI: 10.1021/ed100182hThere is no corresponding record for this reference.
- 57Randall, J. T. Luminescence of Liquids and Solids. Nature 1944, 154, 625– 625, DOI: 10.1038/154625a0There is no corresponding record for this reference.
- 58Bowman, R. L.; Caulfield, P. A.; Udenfriend, S. Spectrophotofluorometric Assay in the Visible and Ultraviolet. Science 1955, 122, 32– 33, DOI: 10.1126/science.122.3157.32There is no corresponding record for this reference.
- 59Slavin, W.; Mooney, R. W.; Palumbo, D. T. Energy-Recording Spectrofluorimeter. J. Opt. Soc. Am. 1961, 51, 93, DOI: 10.1364/JOSA.51.000093There is no corresponding record for this reference.
- 60Turner, G. K. An Absolute Spectrofluorometer: A new instrument has been developed for simple determinations of absolute fluorescence spectra. Science 1964, 146, 183– 189, DOI: 10.1126/science.146.3641.183There is no corresponding record for this reference.
- 61Veening, H.; Brandt, W. W. Fluorescimetric Determination of Ruthenium. Anal. Chem. 1960, 32, 1426– 1428, DOI: 10.1021/ac60167a015There is no corresponding record for this reference.
- 62Paris, J. P. Studies on molecular electronic transitions, Ph.D. thesis; Purdue University: 1960.There is no corresponding record for this reference.
- 63Stieglitz, J. A Theory of Color Production: I. Proc. Natl. Acad. Sci. U. S. A. 1923, 9, 303– 308, DOI: 10.1073/pnas.9.9.303There is no corresponding record for this reference.
- 64Stieglitz, J. A Theory of Color Production: II, Inorganic Compounds. Proc. Natl. Acad. Sci. U. S. A. 1923, 9, 308– 312, DOI: 10.1073/pnas.9.9.308There is no corresponding record for this reference.
- 65Mulliken, R. S. Low Electronic States of Simple Heteropolar Diatomic Molecules: III. Hydrogen and Univalent Metal Halides. Phys. Rev. 1937, 51, 310– 332, DOI: 10.1103/PhysRev.51.310There is no corresponding record for this reference.
- 66Mulliken, R. S. Intensities of Electronic Transitions in Molecular Spectra II. Charge-Transfer Spectra. J. Chem. Phys. 1939, 7, 20– 34, DOI: 10.1063/1.1750319There is no corresponding record for this reference.
- 67Rabinowitch, E. Electron Transfer Spectra and Their Photochemical Effects. Rev. Mod. Phys. 1942, 14, 112– 131, DOI: 10.1103/RevModPhys.14.112There is no corresponding record for this reference.
- 68McClure, D. S. Solid State Physics; Elsevier: 1959; pp 399– 525.There is no corresponding record for this reference.
- 69Porter, G. B.; Schläfer, H. L. Zusammenfassender Bericht. Zur Frage der Lumineszenz bei Übergangsmetallverbindungen. Berichte der Bunsengesellschaft für physikalische Chemie 1964, 68, 316– 331, DOI: 10.1002/bbpc.19640680404There is no corresponding record for this reference.
- 70Scheibe, G. Die Absorption der Halogenionen in wässeriger Lösung im beginnenden Schumann-Ultraviolett. Zeitschrift für Physikalische Chemie 1929, 5BB, 355– 364, DOI: 10.1515/zpch-1929-0528There is no corresponding record for this reference.
- 71Kondo, Y. Electron Transfer Spectra in the Crystals of Hexamminecobalt(III) Salts. Bull. Chem. Soc. Jpn. 1955, 28, 497– 504, DOI: 10.1246/bcsj.28.497There is no corresponding record for this reference.
- 72Jo̷rgensen, C. K. Complexes of the 4d- and 5d-Groups. III. Absorption Spectra of Marcel Delépine’s Rhodium(III) and Iridium(III) Complexes. Acta Chem. Scand. 1957, 11, 151– 165, DOI: 10.3891/acta.chem.scand.11-0151There is no corresponding record for this reference.
- 73Jo̷rgensen, C. K. Complexes of the 4d- and 5d-Groups. IV. Electron Transfer Bands with Special Application to M. Delépine’s Complexes and a Transition from Iridium(III) to Pyridine, with some Remarks about Intermediate Coupling in Halide Complexes and the Uranyl Ion. Acta Chem. Scand. 1957, 11, 166– 178, DOI: 10.3891/acta.chem.scand.11-0166There is no corresponding record for this reference.
- 74Crosby, G. A.; Perkins, W. G.; Klassen, D. M. Luminescence from Transition-Metal Complexes: Tris(2, 2′-bipyridine)- and Tris(1, 10-phenanthroline)Ruthenium(II). J. Chem. Phys. 1965, 43, 1498– 1503, DOI: 10.1063/1.1696960There is no corresponding record for this reference.
- 75Demas, J.; Crosby, G. On the multiplicity of the emitting state of ruthenium(II) complexes. J. Mol. Spectrosc. 1968, 26, 72– 77, DOI: 10.1016/0022-2852(68)90144-6There is no corresponding record for this reference.
- 76Fujita, I.; Kobayashi, H. Electronic Excitation Energy Transfer between Complex Ions. J. Chem. Phys. 1970, 52, 4904– 4905, DOI: 10.1063/1.1673730There is no corresponding record for this reference.
- 77Adamson, A. W.; Demas, J. N. New photosensitizer. Tris(2, 2′-bipyridine)ruthenium(II) chloride. J. Am. Chem. Soc. 1971, 93, 1800– 1801, DOI: 10.1021/ja00736a049There is no corresponding record for this reference.
- 78
Adamson had previously observed energy transfer and photoinduced electron transfer from organic chromophores to cobalt and chromium complexes.261,262
There is no corresponding record for this reference. - 79Gafney, H. D.; Adamson, A. W. Excited state Ru(bipyr)32+ as an electron-transfer reductant. J. Am. Chem. Soc. 1972, 94, 8238– 8239, DOI: 10.1021/ja00778a054There is no corresponding record for this reference.
- 80Balzani, V.; Juris, A. Photochemistry and photophysics of Ru(II) polypyridine complexes in the Bologna group. From early studies to recent developments. Coord. Chem. Rev. 2001, 211, 97– 115, DOI: 10.1016/S0010-8545(00)00274-5There is no corresponding record for this reference.
- 81YouTube, uploaded by American Chemical Society, 1st August 2011. https://www.youtube.com/watch?v=NCQWEUiwyGo.There is no corresponding record for this reference.
- 82Demas, J. N.; Adamson, A. W. Tris (2, 2′-bipyridine)ruthenium(II) sensitized reactions of some oxalato complexes. J. Am. Chem. Soc. 1973, 95, 5159– 5168, DOI: 10.1021/ja00797a011There is no corresponding record for this reference.
- 83Sabbatini, N.; Balzani, V. Photosensitized aquation of the hexacyanochromate(III) ion. Evidence against the doublet mechanism. J. Am. Chem. Soc. 1972, 94, 7587– 7589, DOI: 10.1021/ja00776a057There is no corresponding record for this reference.
- 84Natarajan, P.; Endicott, J. F. Photochemistry of ethylenediaminetetraacetate complexes. Excited state reaction pathways in cobalt(III) complexes. J. Am. Chem. Soc. 1972, 94, 3635– 3636, DOI: 10.1021/ja00765a067There is no corresponding record for this reference.
- 85Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37– 38, DOI: 10.1038/238037a0There is no corresponding record for this reference.
- 86Concepcion, J. J.; House, R. L.; Papanikolas, J. M.; Meyer, T. J. Chemical approaches to artificial photosynthesis. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 15560– 15564, DOI: 10.1073/pnas.1212254109There is no corresponding record for this reference.
- 87Design of Advanced Photocatalytic Materials for Energy and Environmental Applications; Springer: London, 2013.There is no corresponding record for this reference.
- 88Ciamician, G. The Photochemistry of the Future. Science 1912, 36, 385– 394, DOI: 10.1126/science.36.926.385There is no corresponding record for this reference.
- 89Rillema, D. P.; Jones, D. S. Structure of tris(2, 2′-bipyridyl)ruthenium(II) hexafluorophosphate, [Ru(bipy)3][PF6]2; X-ray crystallographic determination. J. Chem. Soc., Chem. Commun. 1979, 849– 851, DOI: 10.1039/C39790000849There is no corresponding record for this reference.
- 90Bolletta, F.; Maestri, M.; Moggi, L.; Balzani, V. Ligand and geometry effects in the quenching of the tris(2, 2′-dipyridyl)ruthenium(II) phosphorescence by some chromium(III) complexes. J. Am. Chem. Soc. 1973, 95, 7864– 7866, DOI: 10.1021/ja00804a052There is no corresponding record for this reference.
- 91Wrighton, M.; Markham, J. Quenching of the luminescent state of tris(2, 2′-bipyridine)ruthenium(II) by electronic energy transfer. J. Phys. Chem. 1973, 77, 3042– 3044, DOI: 10.1021/j100644a002There is no corresponding record for this reference.
- 92Navon, G.; Sutin, N. Mechanism of the quenching of the phosphorescence of tris(2, 2′-bipyridine)ruthenium(II) by some cobalt(III) and ruthenium(III) complexes. Inorg. Chem. 1974, 13, 2159– 2164, DOI: 10.1021/ic50139a021There is no corresponding record for this reference.
- 93Lin, C. T.; Boettcher, W.; Chou, M.; Creutz, C.; Sutin, N. Mechanism of the quenching of the emission of substituted polypyridineruthenium(II) complexes by iron(III), chromium(III), and europium(III) ions. J. Am. Chem. Soc. 1976, 98, 6536– 6544, DOI: 10.1021/ja00437a020There is no corresponding record for this reference.
- 94Creutz, C.; Sutin, N. Electron-transfer reactions of excited states: direct evidence for reduction of the charge-transfer excited state of tris(2, 2′-bipyridine)ruthenium(II). J. Am. Chem. Soc. 1976, 98, 6384– 6385, DOI: 10.1021/ja00436a054There is no corresponding record for this reference.
- 95Juris, A.; Gandolfi, M. T.; Manfrin, M. F.; Balzani, V. Electron and energy transfer mechanisms in the quenching of the tris(2, 2′-bipyridine)ruthenium(II) luminescence by cyanide complexes. J. Am. Chem. Soc. 1976, 98, 1047– 1048, DOI: 10.1021/ja00420a046There is no corresponding record for this reference.
- 96Maestri, M.; Grätzel, M. 530 nm-Laser Photolysis Studies of the Photo Reduction of Tris (2, 2-bipyridine)-ruthenium(II) by Organic Donors. Ber. Bunsengesell. Phys. Chem. 1977, 81, 504– 507, DOI: 10.1002/bbpc.19770810510There is no corresponding record for this reference.
- 97Bock, C. R.; Meyer, T. J.; Whitten, D. G. Electron transfer quenching of the luminescent excited state of tris(2, 2′-bipyridine)ruthenium(II). Flash photolysis relaxation technique for measuring the rates of very rapid electron transfer reactions. J. Am. Chem. Soc. 1974, 96, 4710– 4712, DOI: 10.1021/ja00821a078There is no corresponding record for this reference.
- 98Schlicke, B.; Belser, P.; De Cola, L.; Sabbioni, E.; Balzani, V. Photonic Wires of Nanometric Dimensions. Electronic Energy Transfer in Rigid Rodlike Ru(bpy)32+-(ph)n-Os(bpy)32+ Compounds (ph = 1, 4-Phenylene;n= 3, 5, 7). J. Am. Chem. Soc. 1999, 121, 4207– 4214, DOI: 10.1021/ja990044bThere is no corresponding record for this reference.
- 99Welter, S.; Salluce, N.; Belser, P.; Groeneveld, M.; De Cola, L. Photoinduced electronic energy transfer in modular, conjugated, dinuclear Ru(II)/Os(II) complexes. Coord. Chem. Rev. 2005, 249, 1360– 1371, DOI: 10.1016/j.ccr.2004.11.008There is no corresponding record for this reference.
- 100Balzani, V.; Campagna, S.; Denti, G.; Juris, A.; Serroni, S.; Venturi, M. Designing Dendrimers Based on Transition-Metal Complexes. Light-Harvesting Properties and Predetermined Redox Patterns. Acc. Chem. Res. 1998, 31, 26– 34, DOI: 10.1021/ar950202dThere is no corresponding record for this reference.
- 101Campagna, S.; Denti, G.; Serroni, S.; Ciano, M.; Juris, A.; Balzani, V. A tridecanuclear ruthenium(II)-polypyridine supramolecular species: synthesis, absorption and luminescence properties and electrochemical oxidation. Inorg. Chem. 1992, 31, 2982– 2984, DOI: 10.1021/ic00039a056There is no corresponding record for this reference.
- 102Balzani, V.; Ceroni, P.; Credi, A.; Venturi, M. Ruthenium tris(bipyridine) complexes: Interchange between photons and electrons in molecular-scale devices and machines. Coord. Chem. Rev. 2021, 433, 213758 DOI: 10.1016/j.ccr.2020.213758There is no corresponding record for this reference.
- 103Kirk, A. D.; Hoggard, P. E.; Porter, G. B.; Rockley, M. G.; Windsor, M. W. Picosecond flash photolysis and spectroscopy: Transition metal coordination compounds. Chem. Phys. Lett. 1976, 37, 199– 203, DOI: 10.1016/0009-2614(76)80198-4There is no corresponding record for this reference.
- 104Bensasson, R.; Salet, C.; Balzani, V. Laser flash spectroscopy of tris(2, 2′-bipyridine)ruthenium(II) in solution. J. Am. Chem. Soc. 1976, 98, 3722– 3724, DOI: 10.1021/ja00428a064There is no corresponding record for this reference.
- 105Watts, R. J. Ruthenium polypyridyls: A case study. J. Chem. Educ. 1983, 60, 834, DOI: 10.1021/ed060p834There is no corresponding record for this reference.
- 106Fujita, I.; Kobayashi, H. Luminescence polarization of the tris(2, 2′-bipyridine)ruthenium(II) complex. Inorg. Chem. 1973, 12, 2758– 2762, DOI: 10.1021/ic50130a004There is no corresponding record for this reference.
- 107Carroll, P. J.; Brus, L. E. Picosecond Raman scattering study of electron localization in the charge transfer excited state of tris(bipyridine)ruthenium(II). J. Am. Chem. Soc. 1987, 109, 7613– 7616, DOI: 10.1021/ja00259a005There is no corresponding record for this reference.
- 108Hager, G. D.; Crosby, G. A. Charge-transfer exited states of ruthenium(II) complexes. I. Quantum yield and decay measurements. J. Am. Chem. Soc. 1975, 97, 7031– 7037, DOI: 10.1021/ja00857a013There is no corresponding record for this reference.
- 109Baker, D.; Crosby, G. Spectroscopic and magnetic evidence for multiple-state emission from tris(2, 2′-bipyridine) ruthenium (II) sulfate. Chem. Phys. 1974, 4, 428– 433, DOI: 10.1016/0301-0104(74)85009-3There is no corresponding record for this reference.
- 110Gallhuber, E.; Hensler, G.; Yersin, H. Low-temperature emission spectra of crystalline [Ru(bpy)3](ClO4)2. Chem. Phys. Lett. 1985, 120, 445– 449, DOI: 10.1016/0009-2614(85)85638-4There is no corresponding record for this reference.
- 111Kober, E. M.; Meyer, T. J. An electronic structural model for the emitting MLCT excited states of Ru(bpy)32+ and Os(bpy)32+. Inorg. Chem. 1984, 23, 3877– 3886, DOI: 10.1021/ic00192a009There is no corresponding record for this reference.
- 112Myrick, M.; Blakley, R.; Dearmond, M. The number and spacing of levels in the emitting manifold of [Ru(bpy)3]2+ low temperature measurements. Chem. Phys. Lett. 1989, 157, 73– 77, DOI: 10.1016/0009-2614(89)87210-0There is no corresponding record for this reference.
- 113Yamauchi, S.; Komada, Y.; Hirota, N. Observation of surprisingly small zero-field splittings in the lowest emitting state of [Ru(bpy)3]2+. Chem. Phys. Lett. 1986, 129, 197– 200, DOI: 10.1016/0009-2614(86)80196-8There is no corresponding record for this reference.
- 114Yersin, H.; Strasser, J. Triplets in metal–organic compounds. Chemical tunability of relaxation dynamics. Coord. Chem. Rev. 2000, 208, 331– 364, DOI: 10.1016/S0010-8545(00)00326-XThere is no corresponding record for this reference.
- 115Sibbett, W.; Lagatsky, A. A.; Brown, C. T. A. The development and application of femtosecond laser systems. Opt. Express 2012, 20, 6989, DOI: 10.1364/OE.20.006989There is no corresponding record for this reference.
- 116Zewail, A. H. Laser Femtochemistry. Science 1988, 242, 1645– 1653, DOI: 10.1126/science.242.4886.1645There is no corresponding record for this reference.
- 117Damrauer, N. H.; Cerullo, G.; Yeh, A.; Boussie, T. R.; Shank, C. V.; McCusker, J. K. Femtosecond Dynamics of Excited-State Evolution in [Ru(bpy)3]2+. Science 1997, 275, 54– 57, DOI: 10.1126/science.275.5296.54There is no corresponding record for this reference.
- 118Atkins, A. J.; González, L. Trajectory Surface-Hopping Dynamics Including Intersystem Crossing in [Ru(bpy)3]2+. J. Phys. Chem. Lett. 2017, 8, 3840– 3845, DOI: 10.1021/acs.jpclett.7b01479There is no corresponding record for this reference.
- 119Bhasikuttan, A. C.; Suzuki, M.; Nakashima, S.; Okada, T. Ultrafast Fluorescence Detection in Tris(2, 2′-bipyridine)ruthenium(II) Complex in Solution: Relaxation Dynamics Involving Higher Excited States. J. Am. Chem. Soc. 2002, 124, 8398– 8405, DOI: 10.1021/ja026135hThere is no corresponding record for this reference.
- 120Cannizzo, A.; van Mourik, F.; Gawelda, W.; Zgrablic, G.; Bressler, C.; Chergui, M. Broadband Femtosecond Fluorescence Spectroscopy of [Ru(bpy)3]2+. Angew. Chem., Int. Ed. 2006, 45, 3174– 3176, DOI: 10.1002/anie.200600125There is no corresponding record for this reference.
- 121Oh, D. H.; Boxer, S. G. Stark effect spectra of Ru(diimine)32+ complexes. J. Am. Chem. Soc. 1989, 111, 1130– 1131, DOI: 10.1021/ja00185a054There is no corresponding record for this reference.
- 122Yeh, A. T.; Shank, C. V.; McCusker, J. K. Ultrafast Electron Localization Dynamics Following Photo-Induced Charge Transfer. Science 2000, 289, 935– 938, DOI: 10.1126/science.289.5481.935There is no corresponding record for this reference.
- 123Malone, R. A.; Kelley, D. F. Interligand electron transfer and transition state dynamics in Ru(II)trisbipyridine. J. Chem. Phys. 1991, 95, 8970– 8976, DOI: 10.1063/1.461228There is no corresponding record for this reference.
- 124Wallin, S.; Davidsson, J.; Modin, J.; Hammarström, L. Femtosecond Transient Absorption Anisotropy Study on [Ru(bpy)3]2+ and [Ru(bpy)(py)4]2+. Ultrafast Interligand Randomization of the MLCT State. J. Phys. Chem. A 2005, 109, 4697– 4704, DOI: 10.1021/jp0509212There is no corresponding record for this reference.
- 125Kawamoto, K.; Tamiya, Y.; Storr, T.; Cogdell, R. J.; Kinoshita, I.; Hashimoto, H. Disentangling the 1MLCT transition of [Ru(bpy)3]2+ by Stark absorption spectroscopy. J. Photochem. Photobiol., A 2018, 353, 618– 624, DOI: 10.1016/j.jphotochem.2017.08.025There is no corresponding record for this reference.
- 126Stark, C. W.; Schreier, W. J.; Lucon, J.; Edwards, E.; Douglas, T.; Kohler, B. Interligand Electron Transfer in Heteroleptic Ruthenium(II) Complexes Occurs on Multiple Time Scales. J. Phys. Chem. A 2015, 119, 4813– 4824, DOI: 10.1021/acs.jpca.5b01770There is no corresponding record for this reference.
- 127Moret, M.; Tavernelli, I.; Chergui, M.; Rothlisberger, U. Electron Localization Dynamics in the Triplet Excited State of [Ru(bpy)3]2+ in Aqueous Solution. Chem. – Eur. J. 2010, 16, 5889– 5894, DOI: 10.1002/chem.201000184There is no corresponding record for this reference.
- 128Tavernelli, I.; Curchod, B. F.; Rothlisberger, U. Nonadiabatic molecular dynamics with solvent effects: A LR-TDDFT QM/MM study of ruthenium (II) tris (bipyridine) in water. Chem. Phys. 2011, 391, 101– 109, DOI: 10.1016/j.chemphys.2011.03.021There is no corresponding record for this reference.
- 129Alcover-Fortuny, G.; Wu, J.; Caballol, R.; de Graaf, C. Quantum Chemical Study of the Interligand Electron Transfer in Ru Polypyridyl Complexes. J. Phys. Chem. A 2018, 122, 1114– 1123, DOI: 10.1021/acs.jpca.7b11422There is no corresponding record for this reference.
- 130Zeng, C.; Li, Y.; Zheng, H.; Ren, M.; Wu, W.; Chen, Z. Nature of ultrafast dynamics in the lowest-lying singlet excited state of [Ru(bpy)3]2+. Phys. Chem. Chem. Phys. 2024, 26, 6524– 6531, DOI: 10.1039/D3CP03806HThere is no corresponding record for this reference.
- 131Stockett, M. H.; Bro̷ndsted Nielsen, S. Communication: Does a single CH3CN molecule attached to Ru(bipy)32+ affect its absorption spectrum?. J. Chem. Phys. 2015, 142, 171102, DOI: 10.1063/1.4919781There is no corresponding record for this reference.
- 132Stark, C. W.; Trummal, A.; Uudsemaa, M.; Pahapill, J.; Rammo, M.; Petritsenko, K.; Sildoja, M.-M.; Rebane, A. Solute-solvent electronic interaction is responsible for initial charge separation in ruthenium complexes [Ru(bpy)3]2+ and [Ru(phen)3]2+. Commun. Chem. 2019, 2, 108, DOI: 10.1038/s42004-019-0213-5There is no corresponding record for this reference.
- 133Pelczarski, D.; Korolevych, O.; Gierczyk, B.; Zalas, M.; Makowska-Janusik, M.; Stampor, W. Electronic States of Tris(bipyridine) Ruthenium(II) Complexes in Neat Solid Films Investigated by Electroabsorption Spectroscopy. Materials 2022, 15, 2278, DOI: 10.3390/ma15062278There is no corresponding record for this reference.
- 134Yersin, H.; Humbs, W.; Strasser, J. Low-lying electronic states of [Rh(bpy)3]3+, [Pt(bpy)2]2+, and [Ru(bpy)3]2+. A comparative study based on highly resolved and time-resolved spectra. Coord. Chem. Rev. 1997, 159, 325– 358, DOI: 10.1016/S0010-8545(96)01318-5There is no corresponding record for this reference.
- 135Visco, R. E.; Chandross, E. A. Electroluminescence in Solutions of Aromatic Hydrocarbons. J. Am. Chem. Soc. 1964, 86, 5350– 5351, DOI: 10.1021/ja01077a073There is no corresponding record for this reference.
- 136Hercules, D. M. Chemiluminescence Resulting from Electrochemically Generated Species. Science 1964, 145, 808– 809, DOI: 10.1126/science.145.3634.808There is no corresponding record for this reference.
- 137Santhanam, K. S. V.; Bard, A. J. Chemiluminescence of Electrogenerated 9, 10-Diphenylanthracene Anion Radical1. J. Am. Chem. Soc. 1965, 87, 139– 140, DOI: 10.1021/ja01079a039There is no corresponding record for this reference.
- 138Lemon, H. B. Luminosity of Rectifier Electrode. Science 1918, 47, 170, DOI: 10.1126/science.47.1207.170-bThere is no corresponding record for this reference.
- 139Dufford, R. T.; Nightingale, D.; Gaddum, L. W. Luminescence of Grignard compounds in electric and magnetic fields, and related electrical phenomena. J. Am. Chem. Soc. 1927, 49, 1858– 1864, DOI: 10.1021/ja01407a002There is no corresponding record for this reference.
- 140Dufford, R. T. Luminescence Associated with Electrolysis. J. Opt. Soc. Am. 1929, 18, 17, DOI: 10.1364/JOSA.18.000017There is no corresponding record for this reference.
- 141Harvey, N. Luminescence during Electrolysis. J. Phys. Chem. 1929, 33, 1456– 1459, DOI: 10.1021/j150304a002There is no corresponding record for this reference.
- 142Hercules, D. M.; Lytle, F. E. Chemiluminescence from Reduction Reactions. J. Am. Chem. Soc. 1966, 88, 4745– 4746, DOI: 10.1021/ja00972a052There is no corresponding record for this reference.
- 143
Indeed, the same year which saw first publication of photoinduced electron transfer studies in [Ru(bpy)3]2+, as well as the landmark paper on water-splitting on TiO2 by Fujishima and Honda.
There is no corresponding record for this reference. - 144Tokel, N. E.; Bard, A. J. Electrogenerated chemiluminescence. IX. Electrochemistry and emission from systems containing tris(2, 2′-bipyridine)ruthenium(II) dichloride. J. Am. Chem. Soc. 1972, 94, 2862– 2863, DOI: 10.1021/ja00763a056There is no corresponding record for this reference.
- 145Tokel-Takvoryan, N. E.; Hemingway, R. E.; Bard, A. J. Electrogenerated chemiluminescence. XIII. Electrochemical and electrogenerated chemiluminescence studies of ruthenium chelates. J. Am. Chem. Soc. 1973, 95, 6582– 6589, DOI: 10.1021/ja00801a011There is no corresponding record for this reference.
- 146Ohsawa, Y.; DeArmond, M. K.; Hanck, K. W.; Morris, D. E.; Whitten, D. G.; Neveux, P. E. Spatially isolated redox orbitals: evidence from low-temperature voltammetry. J. Am. Chem. Soc. 1983, 105, 6522– 6524, DOI: 10.1021/ja00359a045There is no corresponding record for this reference.
- 147Vlcek, A. Ligand based redox series. Coord. Chem. Rev. 1982, 43, 39– 62, DOI: 10.1016/S0010-8545(00)82091-3There is no corresponding record for this reference.
- 148Gorman, B. A.; Francis, P. S.; Barnett, N. W. Tris(2, 2′-bipyridyl)ruthenium(ii) chemiluminescence. Analyst 2006, 131, 616– 639, DOI: 10.1039/B518454AThere is no corresponding record for this reference.
- 149Villani, E.; Sakanoue, K.; Einaga, Y.; Inagi, S.; Fiorani, A. Photophysics and electrochemistry of ruthenium complexes for electrogenerated chemiluminescence. J. Electroanal. Chem. 2022, 921, 116677 DOI: 10.1016/j.jelechem.2022.116677There is no corresponding record for this reference.
- 150Deaver, D. R. A new non-isotopic detection system for immunoassays. Nature 1995, 377, 758– 760, DOI: 10.1038/377758a0There is no corresponding record for this reference.
- 151Fracassa, A.; Ferrari, G.; Balli, M. V.; Rimoldi, I.; Facchetti, G.; Arnal, L.; Marconi, A.; Calvaresi, M.; Prodi, L.; De Cola, L.; Valenti, G. Stimuli-Responsive Luminophore Drives Mechanism Switch for Highly Efficient Electrochemiluminescence Immunosensing. J. Am. Chem. Soc. 2025, 147, 35501, DOI: 10.1021/jacs.5c10211There is no corresponding record for this reference.
- 152Ashton, P. R.; Ballardini, R.; Balzani, V.; Constable, E. C.; Credi, A.; Kocian, O.; Langford, S. J.; Preece, J. A.; Prodi, L.; Schofield, E. R.; Spencer, N.; Stoddart, J. F.; Wenger, S. RuII-Polypyridine Complexes Covalently Linked to Electron Acceptors as Wires for Light-Driven Pseudorotaxane-Type Molecular Machines. Chem. – Eur. J. 1998, 4, 2413– 2422, DOI: 10.1002/(SICI)1521-3765(19981204)4:12<2413::AID-CHEM2413>3.0.CO;2-AThere is no corresponding record for this reference.
- 153Ballardini, R.; Balzani, V.; Credi, A.; Gandolfi, M. T.; Venturi, M. Artificial molecular-level machines with [Ru(bpy)3]2+ as a “light-fueled motor”. International Journal of Photoenergy 2001, 3, 63– 77, DOI: 10.1155/S1110662X01000083There is no corresponding record for this reference.
- 154Ceroni, P.; Bergamini, G.; Balzani, V. Old Molecules, New Concepts: [Ru(bpy)3]2+ as a Molecular Encoder–Decoder. Angew. Chem., Int. Ed. 2009, 48, 8516– 8518, DOI: 10.1002/anie.200904764There is no corresponding record for this reference.
- 155Bolletta, F.; Balzani, V. Oscillating chemiluminescence from the reduction of bromate by malonic acid catalyzed by tris(2, 2′-bipyridine)ruthenium(II). J. Am. Chem. Soc. 1982, 104, 4250– 4251, DOI: 10.1021/ja00379a036There is no corresponding record for this reference.
- 156Gerischer, H. Electrochemical photo and solar cells principles and some experiments. Journal of Electroanalytical Chemistry and Interfacial Electrochemistry 1975, 58, 263– 274, DOI: 10.1016/S0022-0728(75)80359-7There is no corresponding record for this reference.
- 157Gleria, M.; Memming, R. Photoelectrochemical Studies of tris [2, 2′-bipyridyl]ruthenium(II) at Semiconductor Electrodes. Zeitschrift für Physikalische Chemie 1975, 98, 303– 316, DOI: 10.1524/zpch.1975.98.1-6.303There is no corresponding record for this reference.
- 158Akimov, I. A. Optical sensibilization of the photoeffect in silver bromide and thallium bromide by organic dyes. Dokl. Akad. Nauk SSSR 1958, 121, 311– 314There is no corresponding record for this reference.
- 159Dudkowski, S. J.; Grossweiner, L. I. Sensitization of Photoconduction in a Zinc Oxide Film by Eosin. J. Opt. Soc. Am. 1964, 54, 486, DOI: 10.1364/JOSA.54.000486There is no corresponding record for this reference.
- 160Inoue, E.; Kokado, H.; Yamaguchi, T. Hypersensitization of Photoconduction in Microcrystalline Zinc Oxide1. J. Phys. Chem. 1965, 69, 767– 774, DOI: 10.1021/j100887a009There is no corresponding record for this reference.
- 161Gerischer, H.; Michel-Beyerle, M.; Rebentrost, F.; Tributsch, H. Sensitization of charge injection into semiconductors with large band gap. Electrochim. Acta 1968, 13, 1509– 1515, DOI: 10.1016/0013-4686(68)80076-3There is no corresponding record for this reference.
- 162Tributsch, H.; Gerischer, H. The use of semiconductor electrodes in the study of photochemical reactions. Berichte der Bunsengesellschaft f ü r physikalische Chemie 1969, 73, 850– 854, DOI: 10.1002/bbpc.19690730821There is no corresponding record for this reference.
- 163Tributsch, H.; Calvin, M. Electrochemistry of excited molecules: photo-electrochemical reactions of chlorophylls. Photochem. Photobiol. 1971, 14, 95– 112, DOI: 10.1111/j.1751-1097.1971.tb06156.xThere is no corresponding record for this reference.
- 164Terenin, A.; Putzeiko, E. Sensibilisation optique des semi-conducteurs par la chlorophylle et pigments apparentés. Journal de Chimie Physique 1958, 55, 681– 687, DOI: 10.1051/jcp/1958550681There is no corresponding record for this reference.
- 165
It is worth mentioning here that Gleria and Memming also further investigated [Ru(bpy)3]2+ ECL with a focus on the semiconductor electrodes used.263
There is no corresponding record for this reference. - 166Osa, T.; Fujihira, M. Photocell using covalently-bound dyes on semiconductor surfaces. Nature 1976, 264, 349– 350, DOI: 10.1038/264349a0There is no corresponding record for this reference.
- 167Wrighton, M. S. Photoelectrochemical conversion of optical energy to electricity and fuels. Acc. Chem. Res. 1979, 12, 303– 310, DOI: 10.1021/ar50141a001There is no corresponding record for this reference.
- 168Clark, W. D. K.; Sutin, N. Spectral sensitization of n-type titanium dioxide electrodes by polypyridineruthenium(II) complexes. J. Am. Chem. Soc. 1977, 99, 4676– 4682, DOI: 10.1021/ja00456a025There is no corresponding record for this reference.
- 169Hamnett, A.; Dare-Edwards, M. P.; Wright, R. D.; Seddon, K. R.; Goodenough, J. B. Photosensitization of titanium(IV) oxide with tris(2, 2′-bipyridine)ruthenium(II) chloride. Surface states of titanium(IV) oxide. J. Phys. Chem. 1979, 83, 3280– 3290, DOI: 10.1021/j100488a020There is no corresponding record for this reference.
- 170Anderson, S.; Constable, E. C.; Dare-Edwards, M. P.; Goodenough, J. B.; Hamnett, A.; Seddon, K. R.; Wright, R. D. Chemical modification of a titanium (IV) oxide electrode to give stable dye sensitisation without a supersensitiser. Nature 1979, 280, 571– 573, DOI: 10.1038/280571a0There is no corresponding record for this reference.
- 171Dare-Edwards, M. P.; Goodenough, J. B.; Hamnett, A.; Seddon, K. R.; Wright, R. D. Sensitisation of semiconducting electrodes with ruthenium-based dyes. Faraday Discuss. Chem. Soc. 1980, 70, 285, DOI: 10.1039/dc9807000285There is no corresponding record for this reference.
- 172Memming, R.; Schröppel, F. Electron transfer reactions of excited ruthenium(II) complexes in monolayer assemblies at the SnO2-water interface. Chem. Phys. Lett. 1979, 62, 207– 210, DOI: 10.1016/0009-2614(79)80160-8There is no corresponding record for this reference.
- 173Desilvestro, J.; Graetzel, M.; Kavan, L.; Moser, J.; Augustynski, J. Highly efficient sensitization of titanium dioxide. J. Am. Chem. Soc. 1985, 107, 2988– 2990, DOI: 10.1021/ja00296a035There is no corresponding record for this reference.
- 174Vlachopoulos, N.; Liska, P.; Augustynski, J.; Graetzel, M. Very efficient visible light energy harvesting and conversion by spectral sensitization of high surface area polycrystalline titanium dioxide films. J. Am. Chem. Soc. 1988, 110, 1216– 1220, DOI: 10.1021/ja00212a033There is no corresponding record for this reference.
- 175Nazeeruddin, M. K.; Liska, P.; Moser, J.; Vlachopoulos, N.; Grätzel, M. Conversion of Light into Electricity with Trinuclear Ruthenium Complexes Adsorbed on Textured TiO2 Films. Helv. Chim. Acta 1990, 73, 1788– 1803, DOI: 10.1002/hlca.19900730624There is no corresponding record for this reference.
- 176O’Regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737– 740, DOI: 10.1038/353737a0There is no corresponding record for this reference.
- 177Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-Sensitized Solar Cells. Chem. Rev. 2010, 110, 6595– 6663, DOI: 10.1021/cr900356pThere is no corresponding record for this reference.
- 178Muñoz-García, A. B.; Benesperi, I.; Boschloo, G.; Concepcion, J. J.; Delcamp, J. H.; Gibson, E. A.; Meyer, G. J.; Pavone, M.; Pettersson, H.; Hagfeldt, A.; Freitag, M. Dye-sensitized solar cells strike back. Chem. Soc. Rev. 2021, 50, 12450– 12550, DOI: 10.1039/D0CS01336FThere is no corresponding record for this reference.
- 179Vougioukalakis, G. C.; Philippopoulos, A. I.; Stergiopoulos, T.; Falaras, P. Contributions to the development of ruthenium-based sensitizers for dye-sensitized solar cells. Coord. Chem. Rev. 2011, 255, 2602– 2621, DOI: 10.1016/j.ccr.2010.11.006There is no corresponding record for this reference.
- 180Basheer, B.; Mathew, D.; George, B. K.; Reghunadhan Nair, C. An overview on the spectrum of sensitizers: The heart of Dye Sensitized Solar Cells. Sol. Energy 2014, 108, 479– 507, DOI: 10.1016/j.solener.2014.08.002There is no corresponding record for this reference.
- 181Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. Light induced and dye accelerated reductions of phenacyl onium salts by 1, 4-dihydropyridines. Tetrahedron Lett. 1978, 19, 1255– 1258, DOI: 10.1016/S0040-4039(01)94515-0There is no corresponding record for this reference.
- 182Van Bergen, T. J.; Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. Chemistry of dihydropyridines. 9. Hydride transfer from 1, 4-dihydropyridines to sp3-hybridized carbon in sulfonium salts and activated halides. Studies with NAD(P)H models. Journal of Organic Chemistry 1979, 44, 4953– 4962, DOI: 10.1021/jo00394a044There is no corresponding record for this reference.
- 183DeLaive, P. J.; Giannotti, C.; Whitten, D. G. Applications of photoinduced electron-transfer reactions: visible light mediated redox reactions of substrates using transition metal complexes as catalysts. J. Am. Chem. Soc. 1978, 100, 7413– 7415, DOI: 10.1021/ja00491a049There is no corresponding record for this reference.
- 184DeLaive, P. J.; Foreman, T. K.; Giannotti, C.; Whitten, D. G. Photoinduced electron transfer reactions of transition-metal complexes with amines. Mechanistic studies of alternate pathways to back electron transfer. J. Am. Chem. Soc. 1980, 102, 5627– 5631, DOI: 10.1021/ja00537a037There is no corresponding record for this reference.
- 185Cano-Yelo, H.; Deronzier, A. Photo-oxidation of tris(2, 2′-bipyridine)ruthenium(II) by para-substituted benzene diazonium salts in acetonitrile. Two-compartment photoelectrochemical cell applications. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases 1984, 80, 3011, DOI: 10.1039/f19848003011There is no corresponding record for this reference.
- 186Hironaka, K.; Fukuzumi, S.; Tanaka, T. Tris(bipyridyl)ruthenium(II)-photosensitized reaction of 1-benzyl-1, 4-dihydronicotinamide with benzyl bromide. J. Chem. Soc., Perkin Trans. 1984, 2, 1705, DOI: 10.1039/p29840001705There is no corresponding record for this reference.
- 187Okada, K.; Okamoto, K.; Oda, M. A new and practical method of decarboxylation: photosensitized decarboxylation of N-acyloxyphthalimides via electron-transfer mechanism. J. Am. Chem. Soc. 1988, 110, 8736– 8738, DOI: 10.1021/ja00234a047There is no corresponding record for this reference.
- 188Ischay, M. A.; Anzovino, M. E.; Du, J.; Yoon, T. P. Efficient Visible Light Photocatalysis of [2 + 2] Enone Cycloadditions. J. Am. Chem. Soc. 2008, 130, 12886– 12887, DOI: 10.1021/ja805387fThere is no corresponding record for this reference.
- 189Nicewicz, D. A.; MacMillan, D. W. C. Merging Photoredox Catalysis with Organocatalysis: The Direct Asymmetric Alkylation of Aldehydes. Science 2008, 322, 77– 80, DOI: 10.1126/science.1161976There is no corresponding record for this reference.
- 190Narayanam, J. M. R.; Tucker, J. W.; Stephenson, C. R. J. Electron-Transfer Photoredox Catalysis: Development of a Tin-Free Reductive Dehalogenation Reaction. J. Am. Chem. Soc. 2009, 131, 8756– 8757, DOI: 10.1021/ja9033582There is no corresponding record for this reference.
- 191Merck The brilliant history of photoredox catalysis. Nat. Portfolio.There is no corresponding record for this reference.
- 192Shaw, M. H.; Twilton, J.; MacMillan, D. W. C. Photoredox Catalysis in Organic Chemistry. Journal of Organic Chemistry 2016, 81, 6898– 6926, DOI: 10.1021/acs.joc.6b01449There is no corresponding record for this reference.
- 193Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. I. General toxicity. Boll. - Soc. Ital. Biol. Sper. 1938, 13, 6– 8There is no corresponding record for this reference.
- 194Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. II. Distribution in the organism. Boll. - Soc. Ital. Biol. Sper. 1938, 13, 8– 11There is no corresponding record for this reference.
- 195Beccari, E. Pharmacological studies on the ferrous tri-α,α’-bipyridyl complex. III. Further experiments on distribution in the tissues. Boll. - Soc. Ital. Biol. Sper. 1941, 16, 214– 216There is no corresponding record for this reference.
- 196Beccari, E. Pharmacological studies on the ferrous tri-α,α′-bipyridyl complex. IV. Relation between concentration in the blood and action on the central nervous system. Boll. - Soc. Ital. Biol. Sper. 1941, 16, 216– 218There is no corresponding record for this reference.
- 197Dwyer et al. do not reference the works by Beccari in their first paper on the biological activity of complex ions, but they do reference his work in later publications.There is no corresponding record for this reference.
- 198Dwyer, F. P.; Gyarfas, E. C.; O’Dwyer, M. F. Configurational Activity. Nature 1951, 167, 1036– 1036, DOI: 10.1038/1671036a0There is no corresponding record for this reference.
- 199Dwyer, F. P.; Gyarfas, E. C.; Rogers, W. P.; Koch, J. H. Biological Activity of Complex Ions. Nature 1952, 170, 190– 191, DOI: 10.1038/170190a0There is no corresponding record for this reference.
- 200Lindoy, L. F. Celebration of inorganic lives: Interview with Alan M. Sargeson. Coordination Chemistry Reviews 2005, 249, 2731– 2739, DOI: 10.1016/j.ccr.2005.04.015There is no corresponding record for this reference.
- 201Meggers, E. Exploring biologically relevant chemical space with metal complexes. Curr. Opin. Chem. Biol. 2007, 11, 287– 292, DOI: 10.1016/j.cbpa.2007.05.013There is no corresponding record for this reference.
- 202Levina, A.; Mitra, A.; Lay, P. A. Recent developments in ruthenium anticancer drugs. Metallomics 2009, 1, 458, DOI: 10.1039/b904071dThere is no corresponding record for this reference.
- 203Kilah, N. L.; Meggers, E. Sixty Years Young: The Diverse Biological Activities of Metal Polypyridyl Complexes Pioneered by Francis P. Dwyer. Aust. J. Chem. 2012, 65, 1325, DOI: 10.1071/CH12275There is no corresponding record for this reference.
- 204Lee, S. Y.; Kim, C. Y.; Nam, T.-G. Ruthenium Complexes as Anticancer Agents: A Brief History and Perspectives. Drug Design, Development and Therapy 2020, 14, 5375– 5392, DOI: 10.2147/DDDT.S275007There is no corresponding record for this reference.
- 205Koch, J. H.; Gyarfas, E. C.; Dwyer, F. Biological Activity of Complex Ions Mechanism of Inhibition of Acetylcholinesterase. Aust. J. Biol. Sci. 1956, 9, 371, DOI: 10.1071/BI9560371There is no corresponding record for this reference.
- 206Koch, J. H.; Rogers, W.; Dwyer, F.; Gyarfas, E. C. The Metabolic Fate of Tris-1, 10-Phenanthroline 106Ruthenium (II) Perchlorate, a Compound With Anticholinesterase and Curare-Like Activity. Aust. J. Biol. Sci. 1957, 10, 342, DOI: 10.1071/BI9570342There is no corresponding record for this reference.
- 207Dwyer, F. P.; Gyarfas, E. C.; Wright, R. D.; Shulman, A. Effect of Inorganic Complex Ions on Transmission at a Neuromuscular Junction. Nature 1957, 179, 425– 426, DOI: 10.1038/179425a0There is no corresponding record for this reference.
- 208Dwyer, F.; Reid, I.; Shulman, A.; Laycock, G. M.; Dixson, S. The biological actions of 1, 10-phenanthroline and 2, 2'-bipyridine hydrochlorides, quaternary salts and metal chelates and related compounds: 1. Bacteriostatic action on selected Gram-positive, Gram-negative and acid-fast bacteria. Australian Journal of Experimental Biology and Medical Science 1969, 47, 203– 218, DOI: 10.1038/icb.1969.21There is no corresponding record for this reference.
- 209White, D. O.; Harris, A. W.; Dwyer, F. P.; Shulman, A. Actions of metal chelates of substituted 1, 10-phenanthrolines on viruses and cells: I. Inactivation of viruses. Australian Journal of Experimental Biology and Medical Science 1963, 41, 517– 526, DOI: 10.1038/icb.1963.42There is no corresponding record for this reference.
- 210Rosenberg, B.; Van Camp, L.; Krigas, T. Inhibition of Cell Division in Escherichia coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698– 699, DOI: 10.1038/205698a0There is no corresponding record for this reference.
- 211Alderden, R. A.; Hall, M. D.; Hambley, T. W. The Discovery and Development of Cisplatin. J. Chem. Educ. 2006, 83, 728, DOI: 10.1021/ed083p728There is no corresponding record for this reference.
- 212Diamond, I.; Mcdonagh, A.; Wilson, C.; Granelli, S.; Nielsen, S.; Jaenicke, R. Photodynamic therapy of malignant tumors. Lancet 1972, 300, 1175– 1177, DOI: 10.1016/S0140-6736(72)92596-2There is no corresponding record for this reference.
- 213Kessel, D. Photodynamic Therapy: A Brief History. Journal of Clinical Medicine 2019, 8, 1581, DOI: 10.3390/jcm8101581There is no corresponding record for this reference.
- 214Knoll, J. D.; Turro, C. Control and utilization of ruthenium and rhodium metal complex excited states for photoactivated cancer therapy. Coord. Chem. Rev. 2015, 282–283, 110– 126, DOI: 10.1016/j.ccr.2014.05.018There is no corresponding record for this reference.
- 215Heinemann, F.; Karges, J.; Gasser, G. Critical Overview of the Use of Ru(II) Polypyridyl Complexes as Photosensitizers in One-Photon and Two-Photon Photodynamic Therapy. Acc. Chem. Res. 2017, 50, 2727– 2736, DOI: 10.1021/acs.accounts.7b00180There is no corresponding record for this reference.
- 216Gandosio, A.; Purkait, K.; Gasser, G. Recent Approaches towards the Development of Ru(II) Polypyridyl Complexes for Anticancer Photodynamic Therapy. CHIMIA 2021, 75, 845, DOI: 10.2533/chimia.2021.845There is no corresponding record for this reference.
- 217Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. I. The Journal of Chemical Physics 1956, 24, 966– 978, DOI: 10.1063/1.1742723There is no corresponding record for this reference.
- 218Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. II. Applications to Data on the Rates of Isotopic Exchange Reactions. J. Chem. Phys. 1957, 26, 867– 871, DOI: 10.1063/1.1743423There is no corresponding record for this reference.
- 219Marcus, R. A. On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. III. Applications to Data on the Rates of Organic Redox Reactions. J. Chem. Phys. 1957, 26, 872– 877, DOI: 10.1063/1.1743424There is no corresponding record for this reference.
- 220Chance, B.; Nishimura, M. On the mechanism of chlorophyll-cytochrome interaction: the temperature insensitivity of light-induced cytochrome oxidation in chromatium. Proc. Natl. Acad. Sci. U. S. A. 1960, 46, 19– 24, DOI: 10.1073/pnas.46.1.19There is no corresponding record for this reference.
- 221Mitchell, P. Coupling of Phosphorylation to Electron and Hydrogen Transfer by a Chemi-Osmotic type of Mechanism. Nature 1961, 191, 144– 148, DOI: 10.1038/191144a0There is no corresponding record for this reference.
- 222Vredenberg, W.; Duysens, L. Light-induced oxidation of cytochromes in photosynthetic bacteria between 20 and −170°. Biochim. Biophys. Acta, Spec. Sect. Biophys. Subj. 1964, 79, 456– 463, DOI: 10.1016/0926-6577(64)90211-6There is no corresponding record for this reference.
- 223De Vault, D.; Chance, B. Studies of Photosynthesis Using a Pulsed Laser. Biophys. J. 1966, 6, 825– 847, DOI: 10.1016/S0006-3495(66)86698-5There is no corresponding record for this reference.
- 224Sutin, N. Bioinorganic Chemistry─II; American Chemical Society: 1977; pp 156– 172.There is no corresponding record for this reference.
- 225English, A. M.; DeLaive, P. J.; Gray, H. B.; Lum, V. R. Metalloprotein electron-transfer mechanisms. Quenching of electronically excited tris(2, 2′-bipyridine)ruthenium(II) by reduced blue copper proteins. J. Am. Chem. Soc. 1982, 104, 870– 871, DOI: 10.1021/ja00367a047There is no corresponding record for this reference.
- 226Dawson, J. W.; Gray, H. B.; Holwerda, R. A.; Westhead, E. W. Kinetics of the Reduction of Metalloproteins by Chromous Ion. Proc. Natl. Acad. Sci. U. S. A. 1972, 69, 30– 33, DOI: 10.1073/pnas.69.1.30There is no corresponding record for this reference.
- 227Hodges, H. L.; Holwerda, R. A.; Gray, H. B. Kinetic studies of the reduction of ferricytochrome c by ethylenediaminetetraacetatoiron(II). J. Am. Chem. Soc. 1974, 96, 3132– 3137, DOI: 10.1021/ja00817a019There is no corresponding record for this reference.
- 228Cummins, D.; Gray, H. B. Electron-transfer protein reactivities. Kinetic studies of the oxidation of horse heart cytochrome c, Chromatium vinosum high potential iron-sulfur protein, Pseudomonas aeruginosa azurin, bean plastocyanin, and Rhus vernicifera stellacyanin by pentaammminepyridineruthenium(III). J. Am. Chem. Soc. 1977, 99, 5158– 5167, DOI: 10.1021/ja00457a042There is no corresponding record for this reference.
- 229Winkler, J. R.; Nocera, D. G.; Yocom, K. M.; Bordignon, E.; Gray, H. B. Electron-transfer kinetics of pentaammineruthenium(III)(histidine-33)-ferricytochrome c. Measurement of the rate of intramolecular electron transfer between redox centers separated by 15.ANG. in a protein. J. Am. Chem. Soc. 1982, 104, 5798– 5800, DOI: 10.1021/ja00385a047There is no corresponding record for this reference.
- 230Winkler, J. R.; Gray, H. B. Electron transfer in ruthenium-modified proteins. Chem. Rev. 1992, 92, 369– 379, DOI: 10.1021/cr00011a001There is no corresponding record for this reference.
- 231Marcus, R. A. Electron Transfer Reactions in Chemistry: Theory and Experiment (Nobel Lecture). Angewandte Chemie International Edition in English 1993, 32, 1111– 1121, DOI: 10.1002/anie.199311113There is no corresponding record for this reference.
- 232Chang, I. J.; Gray, H. B.; Winkler, J. R. High-driving-force electron transfer in metalloproteins: intramolecular oxidation of ferrocytochrome c by Ru(2, 2′-bpy)2(im)(His-33)3+. J. Am. Chem. Soc. 1991, 113, 7056– 7057, DOI: 10.1021/ja00018a064There is no corresponding record for this reference.
- 233Gray, H. B.; Winkler, J. R. Long-range electron transfer. Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 3534– 3539, DOI: 10.1073/pnas.0408029102There is no corresponding record for this reference.
- 234Moser, C. C.; Keske, J. M.; Warncke, K.; Farid, R. S.; Dutton, P. L. Nature of biological electron transfer. Nature 1992, 355, 796– 802, DOI: 10.1038/355796a0There is no corresponding record for this reference.
- 235Page, C.; Moser, C. C.; Dutton, P. L. Mechanism for electron transfer within and between proteins. Curr. Opin. Chem. Biol. 2003, 7, 551– 556, DOI: 10.1016/j.cbpa.2003.08.005There is no corresponding record for this reference.
- 236Dempsey, J. L.; Winkler, J. R.; Gray, H. B. Proton-Coupled Electron Flow in Protein Redox Machines. Chem. Rev. 2010, 110, 7024– 7039, DOI: 10.1021/cr100182bThere is no corresponding record for this reference.
- 237Utterback, J. K.; Ruzicka, J. L.; Keller, H. R.; Pellows, L. M.; Dukovic, G. Electron Transfer from Semiconductor Nanocrystals to Redox Enzymes. Annu. Rev. Phys. Chem. 2020, 71, 335– 359, DOI: 10.1146/annurev-physchem-050317-014232There is no corresponding record for this reference.
- 238Purugganan, M. D.; Kumar, C. V.; Turro, N. J.; Barton, J. K. Accelerated Electron Transfer Between Metal Complexes Mediated by DNA. Science 1988, 241, 1645– 1649, DOI: 10.1126/science.241.4873.1645There is no corresponding record for this reference.
- 239Lippard, S. J. Platinum complexes: probes of polynucleotide structure and antitumor drugs. Acc. Chem. Res. 1978, 11, 211– 217, DOI: 10.1021/ar50125a006There is no corresponding record for this reference.
- 240Barton, J. K.; Danishefsky, A.; Goldberg, J. Tris(phenanthroline)ruthenium(II): stereoselectivity in binding to DNA. J. Am. Chem. Soc. 1984, 106, 2172– 2176, DOI: 10.1021/ja00319a043There is no corresponding record for this reference.
- 241Friedman, A. E.; Chambron, J. C.; Sauvage, J. P.; Turro, N. J.; Barton, J. K. A molecular light switch for DNA: Ru(bpy)2(dppz)2+. J. Am. Chem. Soc. 1990, 112, 4960– 4962, DOI: 10.1021/ja00168a052There is no corresponding record for this reference.
- 242Li, G.; Sun, L.; Ji, L.; Chao, H. Ruthenium(ii) complexes with dppz: from molecular photoswitch to biological applications. Dalton Transactions 2016, 45, 13261– 13276, DOI: 10.1039/C6DT01624CThere is no corresponding record for this reference.
- 243Giese, B. Long-Distance Electron Transfer Through DNA. Annu. Rev. Biochem. 2002, 71, 51– 70, DOI: 10.1146/annurev.biochem.71.083101.134037There is no corresponding record for this reference.
- 244Barton, J. K.; Kumar, C. V.; Turro, N. J. DNA-mediated photoelectron transfer reactions. J. Am. Chem. Soc. 1986, 108, 6391– 6393, DOI: 10.1021/ja00280a047There is no corresponding record for this reference.
- 245Barton, J. K.; Olmon, E. D.; Sontz, P. A. Metal complexes for DNA-mediated charge transport. Coord. Chem. Rev. 2011, 255, 619– 634, DOI: 10.1016/j.ccr.2010.09.002There is no corresponding record for this reference.
- 246Taniguchi, M.; Kawai, T. DNA electronics. Physica E: Low-dimensional Systems and Nanostructures 2006, 33, 1– 12, DOI: 10.1016/j.physe.2006.01.005There is no corresponding record for this reference.
- 247Fancy, D. A.; Kodadek, T. Chemistry for the analysis of protein–protein interactions: Rapid and efficient cross-linking triggered by long wavelength light. Proc. Natl. Acad. Sci. U. S. A. 1999, 96, 6020– 6024, DOI: 10.1073/pnas.96.11.6020There is no corresponding record for this reference.
- 248Preston, G. W.; Wilson, A. J. Photo-induced covalent cross-linking for the analysis of biomolecular interactions. Chem. Soc. Rev. 2013, 42, 3289, DOI: 10.1039/c3cs35459hThere is no corresponding record for this reference.
- 249https://www.science.org/content/article/rubipy-toast-eminent-caltech-personality.There is no corresponding record for this reference.
- 250
This statement has originated at the University of North Carolina at Chapel Hill, as confirmed by Professor Jillian Dempsey (UNC) via email on the 11th of February 2025.
There is no corresponding record for this reference. - 251Büldt, L. A.; Wenger, O. S. Chromium(0), Molybdenum(0), and Tungsten(0) Isocyanide Complexes as Luminophores and Photosensitizers with Long-Lived Excited States. Angew. Chem., Int. Ed. 2017, 56, 5676– 5682, DOI: 10.1002/anie.201701210There is no corresponding record for this reference.
- 252Kjær, K. S. Luminescence and reactivity of a charge-transfer excited iron complex with nanosecond lifetime. Science 2019, 363, 249– 253, DOI: 10.1126/science.aau7160There is no corresponding record for this reference.
- 253Reichenauer, F.; Wang, C.; Förster, C.; Boden, P.; Ugur, N.; Báez-Cruz, R.; Kalmbach, J.; Carrella, L. M.; Rentschler, E.; Ramanan, C.; Niedner-Schatteburg, G.; Gerhards, M.; Seitz, M.; Resch-Genger, U.; Heinze, K. Strongly Red-Emissive Molecular Ruby [Cr(bpmp)2]3+ Surpasses [Ru(bpy)3]2+. J. Am. Chem. Soc. 2021, 143, 11843– 11855, DOI: 10.1021/jacs.1c05971There is no corresponding record for this reference.
- 254Brouwer, A. M. Standards for photoluminescence quantum yield measurements in solution (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 2213– 2228, DOI: 10.1351/PAC-REP-10-09-31There is no corresponding record for this reference.
- 255Ripak, A.; De Kreijger, S.; Elias, B.; Troian-Gautier, L. A protocol for determining cage-escape yields using nanosecond transient absorption spectroscopy. STAR Protocols 2023, 4, 102312 DOI: 10.1016/j.xpro.2023.102312There is no corresponding record for this reference.
- 256Müller, P.; Brettel, K. [Ru(bpy)3]2+ as a reference in transient absorption spectroscopy: differential absorption coefficients for formation of the long-lived 3MLCT excited state. Photochemical & Photobiological Sciences 2012, 11, 632– 636, DOI: 10.1039/c2pp05333kThere is no corresponding record for this reference.
- 257Arias-Rotondo, D. M.; McCusker, J. K. The photophysics of photoredox catalysis: a roadmap for catalyst design. Chem. Soc. Rev. 2016, 45, 5803– 5820, DOI: 10.1039/C6CS00526HThere is no corresponding record for this reference.
- 258Gaynor, J. D.; Petrone, A.; Li, X.; Khalil, M. Mapping Vibronic Couplings in a Solar Cell Dye with Polarization-Selective Two-Dimensional Electronic–Vibrational Spectroscopy. J. Phys. Chem. Lett. 2018, 9, 6289– 6295, DOI: 10.1021/acs.jpclett.8b02752There is no corresponding record for this reference.
- 259Rosenfeld, L. Insulin: discovery and controversy. Clinical chemistry 2002, 48, 2270– 2288, DOI: 10.1093/clinchem/48.12.2270There is no corresponding record for this reference.
- 260Burstall, F. H.; Dwyer, F. P.; Gyarfas, E. C. 194. Optical activity dependent on a six-covalent bivalent osmium complex. Journal of the Chemical Society 1950, 953, DOI: 10.1039/jr9500000953There is no corresponding record for this reference.
- 261Vogler, A.; Adamson, A. W. Photosensitized decomposition of some cobalt ammines. J. Am. Chem. Soc. 1968, 90, 5943– 5945, DOI: 10.1021/ja01023a077There is no corresponding record for this reference.
- 262Adamson, A. W.; Martin, J. E.; Camessei, F. D. Photosensitized aquation of some chromium(III) complexes. J. Am. Chem. Soc. 1969, 91, 7530– 7532, DOI: 10.1021/ja01054a064There is no corresponding record for this reference.
- 263Gleria, M.; Memming, R. Novel Luminescence Generation by Electron Transfer from Semiconductor Electrodes to Ruthenium-Bipyridil Complexes. Zeitschrift f ü r Physikalische Chemie 1976, 101, 171– 179, DOI: 10.1524/zpch.1976.101.1-6.171There is no corresponding record for this reference.



