Fluorine-Functionalized Pore-Space-Partitioned Metal–Organic Frameworks for One-Step Methane PurificationClick to copy article linkArticle link copied!
- Jia-Yao LiuJia-Yao LiuKey Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Jia-Yao Liu
- Li-Qiu YangLi-Qiu YangKey Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Li-Qiu Yang
- Yan-Fei LiYan-Fei LiKey Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Yan-Fei Li
- Ying WangYing WangKey Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Ying Wang
- Wen-Yu YuanWen-Yu YuanKey Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Wen-Yu Yuan
- Quan-Guo Zhai*Quan-Guo Zhai*Email: [email protected]Key Laboratory of Applied Surface and Colloid Chemistry (MOE), Key Laboratory of Macromolecular Science of Shaanxi Province, School of Chemistry & Chemical Engineering, Shaanxi Normal University, Xi’an 710062, ChinaMore by Quan-Guo Zhai
Abstract
The efficient removal of ethane (C2H6) and propane (C3H8) from natural gas is vital for purification. A synergistic pore engineering integrating pore space partition and fluorine functionalization in metal–organic frameworks (MOFs), which may effectively promote the C–H···π and C–H···F interactions for effective methane separation. This strategy was validated using two fluorine-functionalized pore-space-partitioned MOFs (SNNU-707/-708) constructed by introducing varying numbers of −CF3 groups on the pore surface. Single-component adsorption isotherms show high adsorption of SNNU-707/-708 for C2H6 and C3H8 were 94.9/63.6 cm3 g–1 and 96.4/68.9 cm3 g–1, significantly exceeding that of CH4 (18.9/13.4 cm3 g–1). Ideal adsorbed solution theory (IAST) indicated high selectivity values of 85.2/116.6 for C3H8/CH4 (50/50) and 16.7/17.0 for C2H6/CH4 (50/50). Notably, the actual breakthrough interval times of SNNU-707 for C3H8/CH4 (5/95) and C2H6/CH4 (10/90) can reach 502 and 78 min·g–1 and yield high-purity CH4 (>99.5%) at 5.89 mmol g–1 from ternary mixtures. Grand Canonical Monte Carlo (GCMC) simulations attribute this performance to synergistic weak interactions (C–H···π, C–H···F, C–H···O/N) between MOF and alkane. Specially, thanks to the fluorine-functionalized pore environments, both MOFs maintain structural integrity and separation performance under harsh conditions up to 98% relative humidity, which is crucial for practical wet natural gas separation.
This publication is licensed for personal use by The American Chemical Society.
Introduction
Experimental Section
Synthesis of [Co2V(OH)(BDC–CF3)3TPP] (SNNU-707)
Synthesis of [Co2V(OH)(BDC-(CF3)2)3TPP] (SNNU-708)
Results and Discussion
Structure Description
Figure 1
Figure 1. Structural features of SNNU-707/-708: (a) metal clusters and organic linkers. (b, c) porous frameworks viewed along the c-axis direction. (d, e) porous frameworks viewed along the a-axis direction. (f, g) side views of octahedral cages (o-cages) and trigonal-bipyramidal cages (t-cages).
Purity and Stability
Figure 2
Figure 2. Gas adsorption performance of SNNU-707/-708: (a) PXRD patterns of SNNU-708 immersed in pH-1–11 solution for 3 days. (b) adsorption isotherms of C3H8 on SNNU-708 at 298 K after immersing in pH-1–11 solution for 3 days. (c) N2 at 77 K with pore size distributions. (d–f) C3H8, C2H6 and CH4 at 273, 283, and 298 K. (g) summary and comparison of C3H8 and C2H6 adsorption capacities for selected reported MOFs adsorbents are presented. (h) the Qst values for C3H8, C2H6 and CH4 at 298 K. (i) the IAST selectivity for 50/50 C3H8/CH4 and C2H6/CH4.
Gas Adsorption Properties
Figure 3
Figure 3. Dynamic breakthrough performance of SNNU-707/-708: (a) for 50/50 C2H6/CH4 and C3H8/CH4 mixtures. (b, c) for 10/90 C2H6/CH4 mixtures and 5/95 C3H8/CH4 mixtures. (d, e) for 5/10/85 C3H8/C2H6/CH4 mixtures. (f, g) for 5/10/85 C3H8/C2H6/CH4 mixtures in the moist and dry environments. (h, i) cycling test results.
In Situ Infrared Spectroscopy
Figure 4
Figure 4. In situ FT-IR spectra of SNNU-707 and SNNU-708: (a–c) loading C3H8, C2H6 and CH4 in SNNU-707 at different pressures. (d–f) loading C3H8, C2H6 and CH4 in SNNU-708 at different pressures. (g, h) comparison of MOF samples before and after loading paraffin with 760 mmHg.
GCMC Simulation
Figure 5
Figure 5. GCMC simulated adsorption binding sites for C3H8, C2H6 and CH4 in cages of SNNU-707/-708: (a–c) adsorption binding sites for C3H8, C2H6 and CH4 in o-cages of SNNU-707. (d–f) adsorption binding sites for C3H8, C2H6 and CH4 in t-cages of SNNU-707. (g–i) adsorption binding sites for C3H8, C2H6 and CH4 in o-cages of SNNU-708. (j–l) adsorption binding sites for C3H8, C2H6 and CH4 in t-cages of SNNU-708.
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.inorgchem.6c00337.
Additional experimental details, tables of crystal data, additional crystal structure pictures, PXRD, TGA, virial fitting, and IAST selectivity results for SNNU-707/-708 (PDF)
Deposition Numbers 2522027–2522028 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This work is financially supported by the National Natural Science Foundation of China (224711490), and the Youth Innovation Team of Shaanxi Universities (2023 and 24JP038).
References
This article references 54 other publications.
- 1Zhou, J.; Ke, T.; Steinke, F.; Stock, N.; Zhang, Z.; Bao, Z.; He, X.; Ren, Q.; Yang, Q. Tunable Confined Aliphatic Pore Environment in Robust Metal-Organic Frameworks for Efficient Separation of Gases with a Similar Structure. J. Am. Chem. Soc. 2022, 144 (31), 14322– 14329, DOI: 10.1021/jacs.2c05448Google ScholarThere is no corresponding record for this reference.
- 2Hui, Y.-T.; Wei, Q.-F.; Wang, S.-Q.; Yang, Z.-R.; Ding, S.; Gong, Y.-J.; Yang, G.-S.; Tang, Z.; Zhang, X. A thiophene-rich microporous metal-organic framework for CH4 purification and C3H8 enrichment from simulated natural gas. Dalton Trans. 2025, 54 (46), 17336– 17342, DOI: 10.1039/D5DT01791BGoogle ScholarThere is no corresponding record for this reference.
- 3Chu, Z.; Guo, K.; Chen, F.; Hai, G.; Yang, Q.; Zhang, Z.; Ren, Q.; Bao, Z. Enhanced natural gas purification using Cyclodextrin-Based Metal-Organic frameworks with polar Nanotraps. Sep. Purif. Technol. 2025, 358, 130467 DOI: 10.1016/j.seppur.2024.130467Google ScholarThere is no corresponding record for this reference.
- 4Zhang, Y.; Yang, L.; Wang, L.; Duttwyler, S.; Xing, H. A Microporous Metal-Organic Framework Supramolecularly Assembled from a CuII Dodecaborate Cluster Complex for Selective Gas Separation. Angew. Chem., Int. Ed. 2019, 58 (24), 8145– 8150, DOI: 10.1002/anie.201903600Google ScholarThere is no corresponding record for this reference.
- 5Kundu, T.; Wahiduzzaman, M.; Shah, B. B.; Maurin, G.; Zhao, D. Solvent-Induced Control over Breathing Behavior in Flexible Metal-Organic Frameworks for Natural-Gas Delivery. Angew. Chem., Int. Ed. 2019, 58 (24), 8073– 8077, DOI: 10.1002/anie.201902738Google ScholarThere is no corresponding record for this reference.
- 6Nandanwar, S. U.; Corbin, D. R.; Shiflett, M. B. A Review of Porous Adsorbents for the Separation of Nitrogen from Natural Gas. Ind. Eng. Chem. Res. 2020, 59 (30), 13355– 13369, DOI: 10.1021/acs.iecr.0c02730Google ScholarThere is no corresponding record for this reference.
- 7Shen, J.; Dailly, A.; Beckner, M. Natural gas sorption evaluation on microporous materials. Microporous Mesoporous Mater. 2016, 235, 170– 177, DOI: 10.1016/j.micromeso.2016.08.013Google ScholarThere is no corresponding record for this reference.
- 8Wang, L.; Zhang, W.; Ding, J.; Gong, L.; Krishna, R.; Ran, Y.; Chen, L.; Luo, F. Th-MOF showing six-fold imide-sealed pockets for middle-size-separation of propane from natural gas. Nano Res. 2023, 16 (2), 3287– 3293, DOI: 10.1007/s12274-022-4915-0Google ScholarThere is no corresponding record for this reference.
- 9Zeng, Z.; Wang, W.; Xiong, X.; Zhu, N.; Xiong, Y.; Wei, Z.; Jiang, J.-J. Flexible Microporous Copper(II) Metal-Organic Framework toward the Storage and Separation of C1-C3 Hydrocarbons in Natural Gas. Inorg. Chem. 2021, 60 (12), 8456– 8460, DOI: 10.1021/acs.inorgchem.1c01045Google ScholarThere is no corresponding record for this reference.
- 10Li, J.; Luo, X.; Zhao, N.; Zhang, L.; Huo, Q.; Liu, Y. Two Finite Binuclear [M2(μ2-OH)(COO)2] (M = Co, Ni) Based Highly Porous Metal-Organic Frameworks with High Performance for Gas Sorption and Separation. Inorg. Chem. 2017, 56 (7), 4141– 4147, DOI: 10.1021/acs.inorgchem.7b00156Google ScholarThere is no corresponding record for this reference.
- 11Eldridge, R. B. Olefin/paraffin separation technology: a review. Ind. Eng. Chem. Res. 1993, 32 (10), 2208– 2212, DOI: 10.1021/ie00022a002Google ScholarThere is no corresponding record for this reference.
- 12Chen, J.; Eldridge, R. B.; Rosen, E. L.; Bielawski, C. W. A study of Cu(I)-ethylene complexation for olefin-paraffin separation. AlChE J. 2011, 57 (3), 630– 644, DOI: 10.1002/aic.12286Google ScholarThere is no corresponding record for this reference.
- 13Chuah, C. Y.; Lee, H.; Bae, T.-H. Recent advances of nanoporous adsorbents for light hydrocarbon (C1-C3) separation. Chem. Eng. J. 2022, 430, 132654 DOI: 10.1016/j.cej.2021.132654Google ScholarThere is no corresponding record for this reference.
- 14Ding, M.; Flaig, R. W.; Jiang, H.-L.; Yaghi, O. M. Carbon capture and conversion using metal-organic frameworks and MOF-based materials. Chem. Soc. Rev. 2019, 48 (10), 2783– 2828, DOI: 10.1039/C8CS00829AGoogle ScholarThere is no corresponding record for this reference.
- 15Gong, W.; Xie, Y.; Wang, X.; Kirlikovali, K. O.; Idrees, K. B.; Sha, F.; Xie, H.; Liu, Y.; Chen, B.; Cui, Y.; Fraha, O. K. Programmed Polarizability Engineering in a Cyclen-Based Cubic Zr(IV) Metal-Organic Framework to Boost Xe/Kr Separation. J. Am. Chem. Soc. 2023, 145 (4), 2679– 2689, DOI: 10.1021/jacs.2c13171Google ScholarThere is no corresponding record for this reference.
- 16Chen, C.-X.; Pham, T.; Tan, K.; Krishna, R.; Lan, P. C.; Wang, L.; Chen, S.; Al-Enizi, A. M.; Nafady, A.; Forrest, K. A.; Wang, H.-P.; Wang, S.-C.; Chuan, S.; Lei, Z.; Cheng, Y.-S.; Ma, S.-Q. Regulating C2H2/CO2 adsorption selectivity by electronic-state manipulation of iron in metal-organic frameworks. Cell Rep. Phys. Sci. 2022, 3 (8), 100977 DOI: 10.1016/j.xcrp.2022.100977Google ScholarThere is no corresponding record for this reference.
- 17Yu, L.; Ullah, S.; Zhou, K.; Xia, Q.; Wang, H.; Tu, S.; Huang, J.; Xia, H.-L.; Liu, X.-Y.; Thonhauser, T.; Jing, L. A Microporous Metal-Organic Framework Incorporating Both Primary and Secondary Building Units for Splitting Alkane Isomers. J. Am. Chem. Soc. 2022, 144 (9), 3766– 3770, DOI: 10.1021/jacs.1c12068Google ScholarThere is no corresponding record for this reference.
- 18Zhou, H.-C.; Long, J. R.; Yaghi, O. M. Introduction to Metal-Organic Frameworks. Chem. Rev. 2012, 112 (2), 673– 674, DOI: 10.1021/cr300014xGoogle ScholarThere is no corresponding record for this reference.
- 19Li, B.; Wen, H.-M.; Zhou, W.; Xu, J. Q.; Chen, B. Porous Metal-Organic Frameworks: Promising Materials for Methane Storage. Chem. 2016, 1 (4), 557– 580, DOI: 10.1016/j.chempr.2016.09.009Google ScholarThere is no corresponding record for this reference.
- 20Sahoo, R.; Das, M. C. C2s/C1 hydrocarbon separation: The major step towards natural gas purification by metal-organic frameworks (MOFs). Coord. Chem. Rev. 2021, 442, 213998 DOI: 10.1016/j.ccr.2021.213998Google ScholarThere is no corresponding record for this reference.
- 21Han, X.; Yang, S. Molecular Mechanisms behind Acetylene Adsorption and Selectivity in Functional Porous Materials. Angew. Chem., Int. Ed. 2023, 62 (20), e202218274 DOI: 10.1002/anie.202218274Google ScholarThere is no corresponding record for this reference.
- 22Qazvini, O. T.; Babarao, R.; Shi, Z.-L.; Zhang, Y.-B.; Telfer, S. G. A Robust Ethane-Trapping Metal-Organic Framework with a High Capacity for Ethylene Purification. J. Am. Chem. Soc. 2019, 141 (12), 5014– 5020, DOI: 10.1021/jacs.9b00913Google ScholarThere is no corresponding record for this reference.
- 23Zhang, Y.; Zou, Z.; Liu, Q.; Qiao, Y.; Jiang, C. Dual-functions of the carbon-confined oxygen on the capacitance and cycle stability enhancements of Zn-ion capacitors. J. Mater. Sci. Technol. 2025, 221, 278– 288, DOI: 10.1016/j.jmst.2024.10.003Google ScholarThere is no corresponding record for this reference.
- 24Ding, M.; Cai, X.; Jiang, H.-L. Improving MOF stability: approaches and applications. Chem. Sci. 2019, 10 (44), 10209– 10230, DOI: 10.1039/C9SC03916CGoogle ScholarThere is no corresponding record for this reference.
- 25Yuan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, J.; Yang, X.; Zhang, P.; Wang, Q.; Zou, L.-F.; Zhang, Y.-M.; Zhang, L.-L.; Fang, Y.; Li, J.-L.; Zhou, H.-C. Stable Metal-Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 30 (37), 1704303 DOI: 10.1002/adma.201704303Google ScholarThere is no corresponding record for this reference.
- 26Burtch, N. C.; Jasuja, H.; Walton, K. S. Water Stability and Adsorption in Metal-Organic Frameworks. Chem. Rev. 2014, 114 (20), 10575– 10612, DOI: 10.1021/cr5002589Google ScholarThere is no corresponding record for this reference.
- 27Wang, Y.; Li, T.; Li, L.; Lin, R. B.; Jia, X.; Chang, Z.; Wen, H. M.; Chen, X. M.; Li, J. Construction of Fluorinated Propane-Trap in Metal-Organic Frameworks for Record Polymer-Grade Propylene Production under High Humidity Conditions. Adv. Mater. 2023, 35 (14), 2207955 DOI: 10.1002/adma.202207955Google ScholarThere is no corresponding record for this reference.
- 28Chen, T.-H.; Popov, I.; Zenasni, O.; Daugulis, O.; Miljanić, O. Š. Superhydrophobic perfluorinated metal–organic frameworks. Chem. Commun. 2013, 49 (61), 6846– 6848, DOI: 10.1039/c3cc41564cGoogle ScholarThere is no corresponding record for this reference.
- 29Sun, Q.; Aguila, B.; Verma, G.; Liu, X.; Dai, Z.; Deng, F.; Meng, X.; Xiao, F.-S.; Ma, S. Superhydrophobicity: Constructing Homogeneous Catalysts into Superhydrophobic Porous Frameworks to Protect Them from Hydrolytic Degradation. Chem 2016, 1 (4), 628– 639, DOI: 10.1016/j.chempr.2016.09.008Google ScholarThere is no corresponding record for this reference.
- 30Yang, C.; Kaipa, U.; Mather, Q. Z.; Wang, X.; Nesterov, V.; Venero, A. F.; Omary, M. A. Fluorous Metal-Organic Frameworks with Superior Adsorption and Hydrophobic Properties toward Oil Spill Cleanup and Hydrocarbon Storage. J. Am. Chem. Soc. 2011, 133 (45), 18094– 18097, DOI: 10.1021/ja208408nGoogle ScholarThere is no corresponding record for this reference.
- 31Yang, H.; Wang, Y.; Krishna, R.; Jia, X.; Wang, Y.; Hong, A. N.; Dang, C.; Castillo, H. E.; Bu, X.; Feng, P. Pore-Space-Partition-Enabled Exceptional Ethane Uptake and Ethane-Selective Ethane-Ethylene Separation. J. Am. Chem. Soc. 2020, 142 (5), 2222– 2227, DOI: 10.1021/jacs.9b12924Google ScholarThere is no corresponding record for this reference.
- 32Li, S.-Y.; Fan, S.-C.; Zhang, P.; Yuan, W.-Y.; Wang, Y.; Zhai, Q.-G. Metal-cluster-powered ultramicropore alliance in pore-space-partitioned metal-organic frameworks for benchmark one-step ethylene purification. Chem 2024, 10 (9), 2761– 2775, DOI: 10.1016/j.chempr.2024.04.010Google ScholarThere is no corresponding record for this reference.
- 33Zhong, Y.; Fang, P.; Zhai, M.; Low, Z.; Chen, Z. Complexity in Order: High-Porosity Multicomponent Metal-Organic Frameworks for Clean Energy Gas Storage. J. Am. Chem. Soc. 2025, 147 (49), 45377– 45385, DOI: 10.1021/jacs.5c15567Google ScholarThere is no corresponding record for this reference.
- 34Xue, Y.-Y.; Li, S.-N.; Jiang, Y.-C.; Hu, M.-C.; Zhai, Q.-G. Quest for 9-connected robust metal-organic framework platforms based on [M3(O/OH)(COO)6(pyridine)3] clusters as excellent gas separation and asymmetric supercapacitor materials. J. Mater. Chem. A 2019, 7 (9), 4640– 4650, DOI: 10.1039/C8TA09080GGoogle ScholarThere is no corresponding record for this reference.
- 35Xue, Y. Y.; Bai, X. Y.; Zhang, J.; Wang, Y.; Li, S. N.; Jiang, Y. C.; Hu, M. C.; Zhai, Q. G. Precise Pore Space Partitions Combined with High-Density Hydrogen-Bonding Acceptors within Metal-Organic Frameworks for Highly Efficient Acetylene Storage and Separation. Angew. Chem., Int. Ed. 2021, 60 (18), 10122– 10128, DOI: 10.1002/anie.202015861Google ScholarThere is no corresponding record for this reference.
- 36Yang, H.; Chen, Y.; Dang, C.; Hong, A. N.; Feng, P.; Bu, X. Optimization of Pore-Space-Partitioned Metal-Organic Frameworks Using the Bioisosteric Concept. J. Am. Chem. Soc. 2022, 144 (44), 20221– 20226, DOI: 10.1021/jacs.2c09349Google ScholarThere is no corresponding record for this reference.
- 37Fan, S.-C.; Zhang, Y.-L.; Ni, J.-J.; Li, Y.-P.; Li, S.-N.; Zhai, Q.-G. Substituent Engineering in Pore-Space-Partitioned Metal-Organic Frameworks for CO2 Selective Adsorption and Fixation. Inorg. Chem. 2023, 62 (49), 20279– 20287, DOI: 10.1021/acs.inorgchem.3c03289Google ScholarThere is no corresponding record for this reference.
- 38Wang, W.; Yang, H.; Chen, Y.; Bu, X.; Feng, P. Cyclobutanedicarboxylate Metal-Organic Frameworks as a Platform for Dramatic Amplification of Pore Partition Effect. J. Am. Chem. Soc. 2023, 145 (32), 17551– 17556, DOI: 10.1021/jacs.3c05980Google ScholarThere is no corresponding record for this reference.
- 39Li, Z.-F.; Zhang, Q.; Wang, L.-L.; Li, Y.-L.; Yang, S.-Q.; Hu, T.-L. Aromatic pore engineering in microporous metal-organic frameworks for high-production one-step methane purification from ternary alkanes mixtures. Sep. Purif. Technol. 2025, 355, 129685 DOI: 10.1016/j.seppur.2024.129685Google ScholarThere is no corresponding record for this reference.
- 40Luo, X.; Sun, L.; Zhao, J.; Li, D.-S.; Wang, D.; Li, G.; Huo, Q.; Liu, Y. Three Metal-Organic Frameworks Based on Binodal Inorganic Building Units and Hetero-O, N Donor Ligand: Solvothermal Syntheses, Structures, and Gas Sorption Properties. Cryst. Growth Des. 2015, 15 (10), 4901– 4907, DOI: 10.1021/acs.cgd.5b00791Google ScholarThere is no corresponding record for this reference.
- 41Zhang, L.; Xiong, X.-H.; Meng, L.-L.; Qin, L.-Z.; Chen, C.-X.; Wei, Z.-W.; Su, C.-Y. Engineering pore nanospaces by introducing aromatic effects in UiO-66 for efficient separation of light hydrocarbons. J. Mater. Chem. A 2023, 11 (24), 12902– 12909, DOI: 10.1039/D2TA09338CGoogle ScholarThere is no corresponding record for this reference.
- 42Lu, N.; Lan, L.; Gao, Q.; Li, N.; Bu, X.-H. A Scalable Pillar Layered Metal-Organic Framework for Natural Gas Purification. Chem. Res. Chin. Univ. 2024, 40 (4), 675– 681, DOI: 10.1007/s40242-024-4125-2Google ScholarThere is no corresponding record for this reference.
- 43Lan, L.; Lu, N.; Yin, J.-C.; Gao, Q.; Lang, F.; Zhang, Y.-H.; Nie, H.-X.; Li, N.; Bu, X.-H. Simultaneous extraction of C3H8 and C2H6 from ternary C3H8/C2H6/CH4 mixtures in an ultra-microporous metal-organic framework. Chem. Eng. J. 2023, 476, 146750 DOI: 10.1016/j.cej.2023.146750Google ScholarThere is no corresponding record for this reference.
- 44Zhou, X.; Feng, M.; Zhang, K.; Li, J.; Wang, X.; Wang, D.; Chen, B. Metal-Organic Framework with Polar Pore Surface Designed for Purification of Both Natural Gas and Ethylene. Chem. - Eur. J. 2025, 31 (15), e202404191 DOI: 10.1002/chem.202404191Google ScholarThere is no corresponding record for this reference.
- 45Chen, Y.; Jiang, Y.; Li, J.; Hong, X.; Ni, H.; Wang, L.; Ma, N.; Tong, M.; Krishna, R.; Zhang, Y. Optimizing the cask effect in multicomponent natural gas purification to provide high methane productivity. AlChE J. 2023, 70 (3), e18320 DOI: 10.1002/aic.18320Google ScholarThere is no corresponding record for this reference.
- 46He, Y.; Zhang, Z.; Xiang, S.; Fronczek, F. R.; Krishna, R.; Chen, B. A robust doubly interpenetrated metal-organic framework constructed from a novel aromatic tricarboxylate for highly selective separation of small hydrocarbons. Chem. Commun. 2012, 48 (52), 6493– 6495, DOI: 10.1039/c2cc31792cGoogle ScholarThere is no corresponding record for this reference.
- 47Xue, C.; Peng, H.; Hou, J.; Qu, K. Pore space partition on a sulfonate-rich metal-organic framework for purification of methane from natural gas. Chem. Commun. 2025, 61 (45), 8244– 8247, DOI: 10.1039/D5CC01014DGoogle ScholarThere is no corresponding record for this reference.
- 48Chen, Y.; Qiao, Z.; Lv, D.; Wu, H.; Shi, R.; Xia, Q.; Wang, H.; Zhou, J.; Li, Z. Selective Adsorption of Light Alkanes on a Highly Robust Indium Based Metal-Organic Framework. Ind. Eng. Chem. Res. 2017, 56 (15), 4488– 4495, DOI: 10.1021/acs.iecr.6b05010Google ScholarThere is no corresponding record for this reference.
- 49Jia, J.; Wang, L.; Sun, F.; Jing, X.; Bian, Z.; Gao, L.; Krishna, R.; Zhu, G. The Adsorption and Simulated Separation of Light Hydrocarbons in Isoreticular Metal-Organic Frameworks Based on Dendritic Ligands with Different Aliphatic Side Chains. Chem. - Eur. J. 2014, 20 (29), 9073– 9080, DOI: 10.1002/chem.201304962Google ScholarThere is no corresponding record for this reference.
- 50Fan, W.; Wang, X.; Zhang, X.; Liu, X.; Wang, Y.; Kang, Z.; Dai, F.; Xu, B.; Wang, R.; Sun, D. Fine-Tuning the Pore Environment of the Microporous Cu-MOF for High Propylene Storage and Efficient Separation of Light Hydrocarbons. ACS Cent. Sci. 2019, 5 (7), 1261– 1268, DOI: 10.1021/acscentsci.9b00423Google ScholarThere is no corresponding record for this reference.
- 51He, Y.-P.; Tan, Y.-X.; Zhang, J. Tuning a layer to a pillared-layer metal-organic framework for adsorption and separation of light hydrocarbons. Chem. Commun. 2013, 49 (96), 11323– 11352, DOI: 10.1039/c3cc47286hGoogle ScholarThere is no corresponding record for this reference.
- 52Fan, W.; Wang, Y.; Xiao, Z.; Huang, Z.; Dai, F.; Wang, R.; Sun, D. Two-dimensional cobalt metal-organic frameworks for efficient C3H6/CH4 and C3H8/CH4 hydrocarbon separation. Chin. Chem. Lett. 2018, 29 (6), 865– 868, DOI: 10.1016/j.cclet.2017.11.020Google ScholarThere is no corresponding record for this reference.
- 53Wang, G.-D.; Krishna, R.; Li, Y.-Z.; Ma, Y.-Y.; Hou, L.; Wang, Y.-Y.; Zhu, Z. Rational Construction of Ultrahigh Thermal Stable MOF for Efficient Separation of MTO Products and Natural Gas. ACS Mater. Lett. 2023, 5 (4), 1091– 1099, DOI: 10.1021/acsmaterialslett.3c00096Google ScholarThere is no corresponding record for this reference.
- 54Qin, L.-Z.; Xiong, X.-H.; Wang, S.-H.; Zhang, L.; Meng, L.-L.; Yan, L.; Fan, Y.-N.; Yan, T.-A.; Liu, D.-H.; Wei, Z.-W.; Su, C.-Y. MIL-101-Cr/Fe/Fe-NH2 for Efficient Separation of CH4 and C3H8 from Simulated Natural Gas. ACS Appl. Mater. Interfaces 2022, 14 (40), 45444– 45450, DOI: 10.1021/acsami.2c13446Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1

Figure 1. Structural features of SNNU-707/-708: (a) metal clusters and organic linkers. (b, c) porous frameworks viewed along the c-axis direction. (d, e) porous frameworks viewed along the a-axis direction. (f, g) side views of octahedral cages (o-cages) and trigonal-bipyramidal cages (t-cages).
Figure 2

Figure 2. Gas adsorption performance of SNNU-707/-708: (a) PXRD patterns of SNNU-708 immersed in pH-1–11 solution for 3 days. (b) adsorption isotherms of C3H8 on SNNU-708 at 298 K after immersing in pH-1–11 solution for 3 days. (c) N2 at 77 K with pore size distributions. (d–f) C3H8, C2H6 and CH4 at 273, 283, and 298 K. (g) summary and comparison of C3H8 and C2H6 adsorption capacities for selected reported MOFs adsorbents are presented. (h) the Qst values for C3H8, C2H6 and CH4 at 298 K. (i) the IAST selectivity for 50/50 C3H8/CH4 and C2H6/CH4.
Figure 3

Figure 3. Dynamic breakthrough performance of SNNU-707/-708: (a) for 50/50 C2H6/CH4 and C3H8/CH4 mixtures. (b, c) for 10/90 C2H6/CH4 mixtures and 5/95 C3H8/CH4 mixtures. (d, e) for 5/10/85 C3H8/C2H6/CH4 mixtures. (f, g) for 5/10/85 C3H8/C2H6/CH4 mixtures in the moist and dry environments. (h, i) cycling test results.
Figure 4
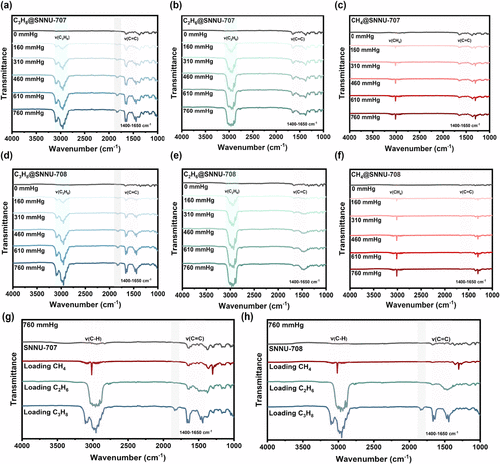
Figure 4. In situ FT-IR spectra of SNNU-707 and SNNU-708: (a–c) loading C3H8, C2H6 and CH4 in SNNU-707 at different pressures. (d–f) loading C3H8, C2H6 and CH4 in SNNU-708 at different pressures. (g, h) comparison of MOF samples before and after loading paraffin with 760 mmHg.
Figure 5

Figure 5. GCMC simulated adsorption binding sites for C3H8, C2H6 and CH4 in cages of SNNU-707/-708: (a–c) adsorption binding sites for C3H8, C2H6 and CH4 in o-cages of SNNU-707. (d–f) adsorption binding sites for C3H8, C2H6 and CH4 in t-cages of SNNU-707. (g–i) adsorption binding sites for C3H8, C2H6 and CH4 in o-cages of SNNU-708. (j–l) adsorption binding sites for C3H8, C2H6 and CH4 in t-cages of SNNU-708.
References
This article references 54 other publications.
- 1Zhou, J.; Ke, T.; Steinke, F.; Stock, N.; Zhang, Z.; Bao, Z.; He, X.; Ren, Q.; Yang, Q. Tunable Confined Aliphatic Pore Environment in Robust Metal-Organic Frameworks for Efficient Separation of Gases with a Similar Structure. J. Am. Chem. Soc. 2022, 144 (31), 14322– 14329, DOI: 10.1021/jacs.2c05448There is no corresponding record for this reference.
- 2Hui, Y.-T.; Wei, Q.-F.; Wang, S.-Q.; Yang, Z.-R.; Ding, S.; Gong, Y.-J.; Yang, G.-S.; Tang, Z.; Zhang, X. A thiophene-rich microporous metal-organic framework for CH4 purification and C3H8 enrichment from simulated natural gas. Dalton Trans. 2025, 54 (46), 17336– 17342, DOI: 10.1039/D5DT01791BThere is no corresponding record for this reference.
- 3Chu, Z.; Guo, K.; Chen, F.; Hai, G.; Yang, Q.; Zhang, Z.; Ren, Q.; Bao, Z. Enhanced natural gas purification using Cyclodextrin-Based Metal-Organic frameworks with polar Nanotraps. Sep. Purif. Technol. 2025, 358, 130467 DOI: 10.1016/j.seppur.2024.130467There is no corresponding record for this reference.
- 4Zhang, Y.; Yang, L.; Wang, L.; Duttwyler, S.; Xing, H. A Microporous Metal-Organic Framework Supramolecularly Assembled from a CuII Dodecaborate Cluster Complex for Selective Gas Separation. Angew. Chem., Int. Ed. 2019, 58 (24), 8145– 8150, DOI: 10.1002/anie.201903600There is no corresponding record for this reference.
- 5Kundu, T.; Wahiduzzaman, M.; Shah, B. B.; Maurin, G.; Zhao, D. Solvent-Induced Control over Breathing Behavior in Flexible Metal-Organic Frameworks for Natural-Gas Delivery. Angew. Chem., Int. Ed. 2019, 58 (24), 8073– 8077, DOI: 10.1002/anie.201902738There is no corresponding record for this reference.
- 6Nandanwar, S. U.; Corbin, D. R.; Shiflett, M. B. A Review of Porous Adsorbents for the Separation of Nitrogen from Natural Gas. Ind. Eng. Chem. Res. 2020, 59 (30), 13355– 13369, DOI: 10.1021/acs.iecr.0c02730There is no corresponding record for this reference.
- 7Shen, J.; Dailly, A.; Beckner, M. Natural gas sorption evaluation on microporous materials. Microporous Mesoporous Mater. 2016, 235, 170– 177, DOI: 10.1016/j.micromeso.2016.08.013There is no corresponding record for this reference.
- 8Wang, L.; Zhang, W.; Ding, J.; Gong, L.; Krishna, R.; Ran, Y.; Chen, L.; Luo, F. Th-MOF showing six-fold imide-sealed pockets for middle-size-separation of propane from natural gas. Nano Res. 2023, 16 (2), 3287– 3293, DOI: 10.1007/s12274-022-4915-0There is no corresponding record for this reference.
- 9Zeng, Z.; Wang, W.; Xiong, X.; Zhu, N.; Xiong, Y.; Wei, Z.; Jiang, J.-J. Flexible Microporous Copper(II) Metal-Organic Framework toward the Storage and Separation of C1-C3 Hydrocarbons in Natural Gas. Inorg. Chem. 2021, 60 (12), 8456– 8460, DOI: 10.1021/acs.inorgchem.1c01045There is no corresponding record for this reference.
- 10Li, J.; Luo, X.; Zhao, N.; Zhang, L.; Huo, Q.; Liu, Y. Two Finite Binuclear [M2(μ2-OH)(COO)2] (M = Co, Ni) Based Highly Porous Metal-Organic Frameworks with High Performance for Gas Sorption and Separation. Inorg. Chem. 2017, 56 (7), 4141– 4147, DOI: 10.1021/acs.inorgchem.7b00156There is no corresponding record for this reference.
- 11Eldridge, R. B. Olefin/paraffin separation technology: a review. Ind. Eng. Chem. Res. 1993, 32 (10), 2208– 2212, DOI: 10.1021/ie00022a002There is no corresponding record for this reference.
- 12Chen, J.; Eldridge, R. B.; Rosen, E. L.; Bielawski, C. W. A study of Cu(I)-ethylene complexation for olefin-paraffin separation. AlChE J. 2011, 57 (3), 630– 644, DOI: 10.1002/aic.12286There is no corresponding record for this reference.
- 13Chuah, C. Y.; Lee, H.; Bae, T.-H. Recent advances of nanoporous adsorbents for light hydrocarbon (C1-C3) separation. Chem. Eng. J. 2022, 430, 132654 DOI: 10.1016/j.cej.2021.132654There is no corresponding record for this reference.
- 14Ding, M.; Flaig, R. W.; Jiang, H.-L.; Yaghi, O. M. Carbon capture and conversion using metal-organic frameworks and MOF-based materials. Chem. Soc. Rev. 2019, 48 (10), 2783– 2828, DOI: 10.1039/C8CS00829AThere is no corresponding record for this reference.
- 15Gong, W.; Xie, Y.; Wang, X.; Kirlikovali, K. O.; Idrees, K. B.; Sha, F.; Xie, H.; Liu, Y.; Chen, B.; Cui, Y.; Fraha, O. K. Programmed Polarizability Engineering in a Cyclen-Based Cubic Zr(IV) Metal-Organic Framework to Boost Xe/Kr Separation. J. Am. Chem. Soc. 2023, 145 (4), 2679– 2689, DOI: 10.1021/jacs.2c13171There is no corresponding record for this reference.
- 16Chen, C.-X.; Pham, T.; Tan, K.; Krishna, R.; Lan, P. C.; Wang, L.; Chen, S.; Al-Enizi, A. M.; Nafady, A.; Forrest, K. A.; Wang, H.-P.; Wang, S.-C.; Chuan, S.; Lei, Z.; Cheng, Y.-S.; Ma, S.-Q. Regulating C2H2/CO2 adsorption selectivity by electronic-state manipulation of iron in metal-organic frameworks. Cell Rep. Phys. Sci. 2022, 3 (8), 100977 DOI: 10.1016/j.xcrp.2022.100977There is no corresponding record for this reference.
- 17Yu, L.; Ullah, S.; Zhou, K.; Xia, Q.; Wang, H.; Tu, S.; Huang, J.; Xia, H.-L.; Liu, X.-Y.; Thonhauser, T.; Jing, L. A Microporous Metal-Organic Framework Incorporating Both Primary and Secondary Building Units for Splitting Alkane Isomers. J. Am. Chem. Soc. 2022, 144 (9), 3766– 3770, DOI: 10.1021/jacs.1c12068There is no corresponding record for this reference.
- 18Zhou, H.-C.; Long, J. R.; Yaghi, O. M. Introduction to Metal-Organic Frameworks. Chem. Rev. 2012, 112 (2), 673– 674, DOI: 10.1021/cr300014xThere is no corresponding record for this reference.
- 19Li, B.; Wen, H.-M.; Zhou, W.; Xu, J. Q.; Chen, B. Porous Metal-Organic Frameworks: Promising Materials for Methane Storage. Chem. 2016, 1 (4), 557– 580, DOI: 10.1016/j.chempr.2016.09.009There is no corresponding record for this reference.
- 20Sahoo, R.; Das, M. C. C2s/C1 hydrocarbon separation: The major step towards natural gas purification by metal-organic frameworks (MOFs). Coord. Chem. Rev. 2021, 442, 213998 DOI: 10.1016/j.ccr.2021.213998There is no corresponding record for this reference.
- 21Han, X.; Yang, S. Molecular Mechanisms behind Acetylene Adsorption and Selectivity in Functional Porous Materials. Angew. Chem., Int. Ed. 2023, 62 (20), e202218274 DOI: 10.1002/anie.202218274There is no corresponding record for this reference.
- 22Qazvini, O. T.; Babarao, R.; Shi, Z.-L.; Zhang, Y.-B.; Telfer, S. G. A Robust Ethane-Trapping Metal-Organic Framework with a High Capacity for Ethylene Purification. J. Am. Chem. Soc. 2019, 141 (12), 5014– 5020, DOI: 10.1021/jacs.9b00913There is no corresponding record for this reference.
- 23Zhang, Y.; Zou, Z.; Liu, Q.; Qiao, Y.; Jiang, C. Dual-functions of the carbon-confined oxygen on the capacitance and cycle stability enhancements of Zn-ion capacitors. J. Mater. Sci. Technol. 2025, 221, 278– 288, DOI: 10.1016/j.jmst.2024.10.003There is no corresponding record for this reference.
- 24Ding, M.; Cai, X.; Jiang, H.-L. Improving MOF stability: approaches and applications. Chem. Sci. 2019, 10 (44), 10209– 10230, DOI: 10.1039/C9SC03916CThere is no corresponding record for this reference.
- 25Yuan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, J.; Yang, X.; Zhang, P.; Wang, Q.; Zou, L.-F.; Zhang, Y.-M.; Zhang, L.-L.; Fang, Y.; Li, J.-L.; Zhou, H.-C. Stable Metal-Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 30 (37), 1704303 DOI: 10.1002/adma.201704303There is no corresponding record for this reference.
- 26Burtch, N. C.; Jasuja, H.; Walton, K. S. Water Stability and Adsorption in Metal-Organic Frameworks. Chem. Rev. 2014, 114 (20), 10575– 10612, DOI: 10.1021/cr5002589There is no corresponding record for this reference.
- 27Wang, Y.; Li, T.; Li, L.; Lin, R. B.; Jia, X.; Chang, Z.; Wen, H. M.; Chen, X. M.; Li, J. Construction of Fluorinated Propane-Trap in Metal-Organic Frameworks for Record Polymer-Grade Propylene Production under High Humidity Conditions. Adv. Mater. 2023, 35 (14), 2207955 DOI: 10.1002/adma.202207955There is no corresponding record for this reference.
- 28Chen, T.-H.; Popov, I.; Zenasni, O.; Daugulis, O.; Miljanić, O. Š. Superhydrophobic perfluorinated metal–organic frameworks. Chem. Commun. 2013, 49 (61), 6846– 6848, DOI: 10.1039/c3cc41564cThere is no corresponding record for this reference.
- 29Sun, Q.; Aguila, B.; Verma, G.; Liu, X.; Dai, Z.; Deng, F.; Meng, X.; Xiao, F.-S.; Ma, S. Superhydrophobicity: Constructing Homogeneous Catalysts into Superhydrophobic Porous Frameworks to Protect Them from Hydrolytic Degradation. Chem 2016, 1 (4), 628– 639, DOI: 10.1016/j.chempr.2016.09.008There is no corresponding record for this reference.
- 30Yang, C.; Kaipa, U.; Mather, Q. Z.; Wang, X.; Nesterov, V.; Venero, A. F.; Omary, M. A. Fluorous Metal-Organic Frameworks with Superior Adsorption and Hydrophobic Properties toward Oil Spill Cleanup and Hydrocarbon Storage. J. Am. Chem. Soc. 2011, 133 (45), 18094– 18097, DOI: 10.1021/ja208408nThere is no corresponding record for this reference.
- 31Yang, H.; Wang, Y.; Krishna, R.; Jia, X.; Wang, Y.; Hong, A. N.; Dang, C.; Castillo, H. E.; Bu, X.; Feng, P. Pore-Space-Partition-Enabled Exceptional Ethane Uptake and Ethane-Selective Ethane-Ethylene Separation. J. Am. Chem. Soc. 2020, 142 (5), 2222– 2227, DOI: 10.1021/jacs.9b12924There is no corresponding record for this reference.
- 32Li, S.-Y.; Fan, S.-C.; Zhang, P.; Yuan, W.-Y.; Wang, Y.; Zhai, Q.-G. Metal-cluster-powered ultramicropore alliance in pore-space-partitioned metal-organic frameworks for benchmark one-step ethylene purification. Chem 2024, 10 (9), 2761– 2775, DOI: 10.1016/j.chempr.2024.04.010There is no corresponding record for this reference.
- 33Zhong, Y.; Fang, P.; Zhai, M.; Low, Z.; Chen, Z. Complexity in Order: High-Porosity Multicomponent Metal-Organic Frameworks for Clean Energy Gas Storage. J. Am. Chem. Soc. 2025, 147 (49), 45377– 45385, DOI: 10.1021/jacs.5c15567There is no corresponding record for this reference.
- 34Xue, Y.-Y.; Li, S.-N.; Jiang, Y.-C.; Hu, M.-C.; Zhai, Q.-G. Quest for 9-connected robust metal-organic framework platforms based on [M3(O/OH)(COO)6(pyridine)3] clusters as excellent gas separation and asymmetric supercapacitor materials. J. Mater. Chem. A 2019, 7 (9), 4640– 4650, DOI: 10.1039/C8TA09080GThere is no corresponding record for this reference.
- 35Xue, Y. Y.; Bai, X. Y.; Zhang, J.; Wang, Y.; Li, S. N.; Jiang, Y. C.; Hu, M. C.; Zhai, Q. G. Precise Pore Space Partitions Combined with High-Density Hydrogen-Bonding Acceptors within Metal-Organic Frameworks for Highly Efficient Acetylene Storage and Separation. Angew. Chem., Int. Ed. 2021, 60 (18), 10122– 10128, DOI: 10.1002/anie.202015861There is no corresponding record for this reference.
- 36Yang, H.; Chen, Y.; Dang, C.; Hong, A. N.; Feng, P.; Bu, X. Optimization of Pore-Space-Partitioned Metal-Organic Frameworks Using the Bioisosteric Concept. J. Am. Chem. Soc. 2022, 144 (44), 20221– 20226, DOI: 10.1021/jacs.2c09349There is no corresponding record for this reference.
- 37Fan, S.-C.; Zhang, Y.-L.; Ni, J.-J.; Li, Y.-P.; Li, S.-N.; Zhai, Q.-G. Substituent Engineering in Pore-Space-Partitioned Metal-Organic Frameworks for CO2 Selective Adsorption and Fixation. Inorg. Chem. 2023, 62 (49), 20279– 20287, DOI: 10.1021/acs.inorgchem.3c03289There is no corresponding record for this reference.
- 38Wang, W.; Yang, H.; Chen, Y.; Bu, X.; Feng, P. Cyclobutanedicarboxylate Metal-Organic Frameworks as a Platform for Dramatic Amplification of Pore Partition Effect. J. Am. Chem. Soc. 2023, 145 (32), 17551– 17556, DOI: 10.1021/jacs.3c05980There is no corresponding record for this reference.
- 39Li, Z.-F.; Zhang, Q.; Wang, L.-L.; Li, Y.-L.; Yang, S.-Q.; Hu, T.-L. Aromatic pore engineering in microporous metal-organic frameworks for high-production one-step methane purification from ternary alkanes mixtures. Sep. Purif. Technol. 2025, 355, 129685 DOI: 10.1016/j.seppur.2024.129685There is no corresponding record for this reference.
- 40Luo, X.; Sun, L.; Zhao, J.; Li, D.-S.; Wang, D.; Li, G.; Huo, Q.; Liu, Y. Three Metal-Organic Frameworks Based on Binodal Inorganic Building Units and Hetero-O, N Donor Ligand: Solvothermal Syntheses, Structures, and Gas Sorption Properties. Cryst. Growth Des. 2015, 15 (10), 4901– 4907, DOI: 10.1021/acs.cgd.5b00791There is no corresponding record for this reference.
- 41Zhang, L.; Xiong, X.-H.; Meng, L.-L.; Qin, L.-Z.; Chen, C.-X.; Wei, Z.-W.; Su, C.-Y. Engineering pore nanospaces by introducing aromatic effects in UiO-66 for efficient separation of light hydrocarbons. J. Mater. Chem. A 2023, 11 (24), 12902– 12909, DOI: 10.1039/D2TA09338CThere is no corresponding record for this reference.
- 42Lu, N.; Lan, L.; Gao, Q.; Li, N.; Bu, X.-H. A Scalable Pillar Layered Metal-Organic Framework for Natural Gas Purification. Chem. Res. Chin. Univ. 2024, 40 (4), 675– 681, DOI: 10.1007/s40242-024-4125-2There is no corresponding record for this reference.
- 43Lan, L.; Lu, N.; Yin, J.-C.; Gao, Q.; Lang, F.; Zhang, Y.-H.; Nie, H.-X.; Li, N.; Bu, X.-H. Simultaneous extraction of C3H8 and C2H6 from ternary C3H8/C2H6/CH4 mixtures in an ultra-microporous metal-organic framework. Chem. Eng. J. 2023, 476, 146750 DOI: 10.1016/j.cej.2023.146750There is no corresponding record for this reference.
- 44Zhou, X.; Feng, M.; Zhang, K.; Li, J.; Wang, X.; Wang, D.; Chen, B. Metal-Organic Framework with Polar Pore Surface Designed for Purification of Both Natural Gas and Ethylene. Chem. - Eur. J. 2025, 31 (15), e202404191 DOI: 10.1002/chem.202404191There is no corresponding record for this reference.
- 45Chen, Y.; Jiang, Y.; Li, J.; Hong, X.; Ni, H.; Wang, L.; Ma, N.; Tong, M.; Krishna, R.; Zhang, Y. Optimizing the cask effect in multicomponent natural gas purification to provide high methane productivity. AlChE J. 2023, 70 (3), e18320 DOI: 10.1002/aic.18320There is no corresponding record for this reference.
- 46He, Y.; Zhang, Z.; Xiang, S.; Fronczek, F. R.; Krishna, R.; Chen, B. A robust doubly interpenetrated metal-organic framework constructed from a novel aromatic tricarboxylate for highly selective separation of small hydrocarbons. Chem. Commun. 2012, 48 (52), 6493– 6495, DOI: 10.1039/c2cc31792cThere is no corresponding record for this reference.
- 47Xue, C.; Peng, H.; Hou, J.; Qu, K. Pore space partition on a sulfonate-rich metal-organic framework for purification of methane from natural gas. Chem. Commun. 2025, 61 (45), 8244– 8247, DOI: 10.1039/D5CC01014DThere is no corresponding record for this reference.
- 48Chen, Y.; Qiao, Z.; Lv, D.; Wu, H.; Shi, R.; Xia, Q.; Wang, H.; Zhou, J.; Li, Z. Selective Adsorption of Light Alkanes on a Highly Robust Indium Based Metal-Organic Framework. Ind. Eng. Chem. Res. 2017, 56 (15), 4488– 4495, DOI: 10.1021/acs.iecr.6b05010There is no corresponding record for this reference.
- 49Jia, J.; Wang, L.; Sun, F.; Jing, X.; Bian, Z.; Gao, L.; Krishna, R.; Zhu, G. The Adsorption and Simulated Separation of Light Hydrocarbons in Isoreticular Metal-Organic Frameworks Based on Dendritic Ligands with Different Aliphatic Side Chains. Chem. - Eur. J. 2014, 20 (29), 9073– 9080, DOI: 10.1002/chem.201304962There is no corresponding record for this reference.
- 50Fan, W.; Wang, X.; Zhang, X.; Liu, X.; Wang, Y.; Kang, Z.; Dai, F.; Xu, B.; Wang, R.; Sun, D. Fine-Tuning the Pore Environment of the Microporous Cu-MOF for High Propylene Storage and Efficient Separation of Light Hydrocarbons. ACS Cent. Sci. 2019, 5 (7), 1261– 1268, DOI: 10.1021/acscentsci.9b00423There is no corresponding record for this reference.
- 51He, Y.-P.; Tan, Y.-X.; Zhang, J. Tuning a layer to a pillared-layer metal-organic framework for adsorption and separation of light hydrocarbons. Chem. Commun. 2013, 49 (96), 11323– 11352, DOI: 10.1039/c3cc47286hThere is no corresponding record for this reference.
- 52Fan, W.; Wang, Y.; Xiao, Z.; Huang, Z.; Dai, F.; Wang, R.; Sun, D. Two-dimensional cobalt metal-organic frameworks for efficient C3H6/CH4 and C3H8/CH4 hydrocarbon separation. Chin. Chem. Lett. 2018, 29 (6), 865– 868, DOI: 10.1016/j.cclet.2017.11.020There is no corresponding record for this reference.
- 53Wang, G.-D.; Krishna, R.; Li, Y.-Z.; Ma, Y.-Y.; Hou, L.; Wang, Y.-Y.; Zhu, Z. Rational Construction of Ultrahigh Thermal Stable MOF for Efficient Separation of MTO Products and Natural Gas. ACS Mater. Lett. 2023, 5 (4), 1091– 1099, DOI: 10.1021/acsmaterialslett.3c00096There is no corresponding record for this reference.
- 54Qin, L.-Z.; Xiong, X.-H.; Wang, S.-H.; Zhang, L.; Meng, L.-L.; Yan, L.; Fan, Y.-N.; Yan, T.-A.; Liu, D.-H.; Wei, Z.-W.; Su, C.-Y. MIL-101-Cr/Fe/Fe-NH2 for Efficient Separation of CH4 and C3H8 from Simulated Natural Gas. ACS Appl. Mater. Interfaces 2022, 14 (40), 45444– 45450, DOI: 10.1021/acsami.2c13446There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.inorgchem.6c00337.
Additional experimental details, tables of crystal data, additional crystal structure pictures, PXRD, TGA, virial fitting, and IAST selectivity results for SNNU-707/-708 (PDF)
Deposition Numbers 2522027–2522028 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



