Current Status of Molecular Dynamics Simulations of Membrane Permeabilization by Antimicrobial Peptides and Pore-Forming Proteins: A ReviewClick to copy article linkArticle link copied!
- Sofia CrescaSofia CrescaTheory Department, National Institute of Chemistry, Hajdrihova 19, 1000 Ljubljana, SloveniaDoctoral School, Faculty of Medicine, University of Ljubljana, 1000 Ljubljana, SloveniaMore by Sofia Cresca
- Jure Borišek*Jure Borišek*Email: [email protected]Theory Department, National Institute of Chemistry, Hajdrihova 19, 1000 Ljubljana, SloveniaMore by Jure Borišek
- Alessandra Magistrato*Alessandra Magistrato*Email: [email protected]Consiglio Nazionale delle Ricerche (CNR)-IOM, c/o International School for Advanced Studies (SISSA/ISAS), via Bonomia 265, 34136 Trieste, ItalyMore by Alessandra Magistrato
- Igor Križaj*Igor Križaj*Email: [email protected]Department of Molecular and Biomedical Sciences, Jožef Stefan Institute, Jamova 39, 1000 Ljubljana, SloveniaMore by Igor Križaj
Abstract
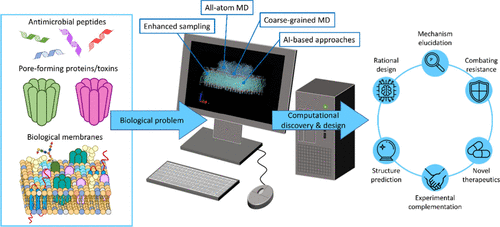
Biological membranes are crucial for cellular integrity and function, but their selective permeability can be compromised by various peptides and proteins, such as antimicrobial peptides (AMPs) and pore-forming proteins/toxins (PFPs/PFTs). These molecules induce membrane permeabilization through diverse mechanisms, ranging from the formation of well-defined pores to more nuanced disruptions of the lipid bilayer. Understanding molecular mechanisms underlying membrane integrity disruption is vital for developing novel tools to be applied in medicine, biotechnology, and agriculture. However, due to their transient and dynamic nature, characterizing membrane-disrupting mechanisms is a significant experimental challenge. In silico methods, particularly all-atom and coarse-grained molecular dynamics (MD) simulations, are an indispensable tool to complement and enrich experimental studies, and can offer detailed insights into peptide/protein–membrane interactions, insertion, oligomerization, and pore formation. This review provides a comprehensive overview of the structural and mechanistic diversity of AMPs and PFPs, highlighting representative case studies and discussing key challenges emerging from MD simulations.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.


