Atomistic Insights into Structure and Properties of ε-Caprolactone OligomersClick to copy article linkArticle link copied!
- Mai AhmedMai AhmedInstitute of Organic Chemistry and Macromolecular Chemistry (IOMC), Friedrich Schiller University Jena, Humboldtstraße 10, 07743 Jena, GermanyJena Center for Soft Matter (JCSM), Friedrich Schiller University Jena, Philosophenweg 7, 07743 Jena, GermanyMore by Mai Ahmed
- Deniz YilmazDeniz YilmazInstitute of Organic Chemistry and Macromolecular Chemistry (IOMC), Friedrich Schiller University Jena, Humboldtstraße 10, 07743 Jena, GermanyJena Center for Soft Matter (JCSM), Friedrich Schiller University Jena, Philosophenweg 7, 07743 Jena, GermanyMore by Deniz Yilmaz
- Purushottam PoudelPurushottam PoudelHIPOLE Jena (Helmholtz Institute for Polymers in Energy Applications Jena), Lessingstrasse 12-14, 07743 Jena, GermanyMore by Purushottam Poudel
- Felix H. SchacherFelix H. SchacherInstitute of Organic Chemistry and Macromolecular Chemistry (IOMC), Friedrich Schiller University Jena, Humboldtstraße 10, 07743 Jena, GermanyJena Center for Soft Matter (JCSM), Friedrich Schiller University Jena, Philosophenweg 7, 07743 Jena, GermanyHIPOLE Jena (Helmholtz Institute for Polymers in Energy Applications Jena), Lessingstrasse 12-14, 07743 Jena, GermanyMore by Felix H. Schacher
- Eva Perlt*Eva Perlt*Email: [email protected]Otto Schott Institute of Materials Research (OSIM), Faculty of Physics and Astronomy, Friedrich Schiller University Jena, Löbdergraben 32, 07743 Jena, GermanyMore by Eva Perlt
Abstract
The design of functional and sustainable materials requires a detailed understanding of the material properties and degradation mechanisms. In particular, the design of fully biodegradable polymers could allow a quick and controlled decomposition of materials before they accumulate in the environment and break down to micro- and nanoplastics. An important degradation pathway proceeds via the hydrolysis of polyesters. To obtain the best performing material candidates, a multiscale-level understanding that takes into account electronic structure combined with multiple configurations at the macroscopic scale is necessary. In this contribution, we present the extension of the multiscale Quantum Cluster Equilibrium method to oligomer materials. We showcase the first application of this methodology to oligomer systems, in particular oligo(ε-Caprolactone). The ε-Caprolactone oligomers were synthesized and characterized comprehensively by means of NMR, SEC, DSC, and TGA. Experimentally, two melting temperatures were observed, which were predicted by theoretical calculations and are in convincing agreement.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of The Journal of Physical Chemistry B special issue “Physical Chemistry of Microplastics and Nanoplastics”.
Introduction
Experimental Methods
Materials
Instrumentation
Syntheses of the εCL Oligomer (OεCL)
Computational Details
Figure 1
Figure 1. Example structures, as used in the QCE study. Crystalline motif (OεCL-c, left) as well as one exemplary amorphous structure (OεCL-a3, right).
| cluster label | ΔE [kJ mol–1] |
|---|---|
| OεCL-c | –106.01 |
| OεCL-a1 | –18.29 |
| OεCL-a2 | –83.45 |
| OεCL-a3 | –60.23 |
| OεCL-a4 | –27.30 |
| OεCL-a5 | –67.43 |
| OεCL-a6 | 0.00 |
| OεCL-a7 | –61.54 |
Results and Discussion
Oligomer Synthesis and Characterization
Scheme 1
Figure 2
Figure 2. Characterization of the OεCL1 oligomer. Top, SEC chromatogram; middle, DSC heating and cooling scans; bottom, TGA curve.
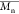 Values, and Dispersity of the Oligomer
Values, and Dispersity of the Oligomer| oligomer | εCL/BnOH (mmol/mmol) | compositiona | 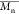 a,b (kg mol–1) a,b (kg mol–1) | 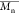 c (kg mol–1) c (kg mol–1) | dispersity Đ |
|---|---|---|---|---|---|
| OεCL1 | 5/0.5 | O(εCL12) | 1.5 | 2.7 | 1.24 |
Determined by 1H NMR spectroscopy (300 MHz, CDCl3).
Determined by 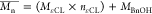 .
.
Determined by SEC measurements (THF, PS calibration).
Figure 3
Figure 3. 1D integrated (a) SAXS patterns of OεCL1 corresponding to q and (b) WAXS patterns of OεCL1 corresponding to 2θ.
| Dhkl (nm) | |||
|---|---|---|---|
| d (nm) | D100 | D200 | Xc (%) |
| 10.98 | 7.95 ± 0.83 | 7.20 ± 0.76 | 68.90 |
QCE Calculations
Figure 4
Figure 4. Temperature-dependent cluster populations showing only significantly populated clusters (>1%). The gray line indicates the interception of both curves.
Figure 5
Figure 5. Phase volume of 1 mol OεCL. At 314 K, a sharp phase transition in the volume is observed.
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06385.
Snapshots of all cluster structures considered in the QCE calculation (Figures S1–S8) and the 1H NMR spectrum of OεCL1 (Figure S9) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
The SAXS setup was funded by the Free State of Thuringia and REACT-EU (2021 FGI 0036─SAXS/WAXS). E.P., F.H.S., and P.P. were further supported by funding from the Carl Zeiss Foundation (“Durchbrüche”─Intelligente Substrate). Funding provided to M.A. within the ProChance program of FSU Jena is gratefully acknowledged.
References
This article references 47 other publications.
- 1Gupta, V.; Biswas, D.; Roy, S. Materials 2022, 15 (17), 5899 DOI: 10.3390/ma15175899Google ScholarThere is no corresponding record for this reference.
- 2Tajeddin, B.; Arabkhedri, M. Polymer Science and Innovative Applications; AlMaadeed, M. A. A.; Ponnamma, D.; Carignano, M. A., Eds.; Elsevier, 2020; pp 525– 543.Google ScholarThere is no corresponding record for this reference.
- 3Hu, J.; Chen, S. J. Mater. Chem. 2010, 20, 3346– 3355, DOI: 10.1039/b922872aGoogle ScholarThere is no corresponding record for this reference.
- 4Moghadasi, K.; Mohd Isa, M. S.; Ariffin, M. A.; Mohd jamil, M. Z.; Raja, S.; Wu, B.; Yamani, M.; Bin Muhamad, M. R.; Yusof, F.; Jamaludin, M. F.; bin Ab Karim, M. S.; binti Abdul Razak, B.; bin Yusoff, N. J. Mater. Res. Technol. 2022, 17, 1054– 1121Google ScholarThere is no corresponding record for this reference.
- 5Chen, W.-H.; Chen, Q.-W.; Chen, Q.; Cui, C.; Duan, S.; Kang, Y.; Liu, Y.; Liu, Y.; Muhammad, W.; Shao, S.; Tang, C.; Wang, J.; Wang, L.; Xiong, M.-H.; Yin, L.; Zhang, K.; Zhang, Z.; Zhen, X.; Feng, J.; Chen, X. Sci. China: Chem. 2022, 65, 1010– 1075, DOI: 10.1007/s11426-022-1243-5Google ScholarThere is no corresponding record for this reference.
- 6Ding, L.; Yu, Z.-D.; Wang, X.-Y.; Yao, Z.-F.; Lu, Y.; Yang, C.-Y.; Wang, J.-Y.; Pei, J. Chem. Rev. 2023, 123, 7421– 7497, DOI: 10.1021/acs.chemrev.2c00696Google ScholarThere is no corresponding record for this reference.
- 7Li, L.; Han, L.; Hu, H.; Zhang, R. Mater. Adv. 2023, 4, 726– 746, DOI: 10.1039/D2MA00940DGoogle ScholarThere is no corresponding record for this reference.
- 8Campbell, I. R.; Lin, M.-Y.; Iyer, H.; Parker, M.; Fredricks, J. L.; Liao, K.; Jimenez, A. M.; Grandgeorge, P.; Roumeli, E. Annu. Rev. Mater. Res. 2023, 53, 81– 104, DOI: 10.1146/annurev-matsci-080921-083655Google ScholarThere is no corresponding record for this reference.
- 9Jayakumar, A.; Radoor, S.; Siengchin, S.; Shin, G. H.; Kim, J. T. Sci. Total Environ. 2023, 878, 163156 DOI: 10.1016/j.scitotenv.2023.163156Google ScholarThere is no corresponding record for this reference.
- 10Sánchez, C. Biotechnol. Adv. 2020, 40, 107501 DOI: 10.1016/j.biotechadv.2019.107501Google ScholarThere is no corresponding record for this reference.
- 11EL-Ghoul, Y.; Alminderej, F. M.; Alsubaie, F. M.; Alrasheed, R.; Almousa, N. H. Polymers 2021, 13 (24), 4327 DOI: 10.3390/polym13244327Google ScholarThere is no corresponding record for this reference.
- 12Delfi, M.; Ghomi, M.; Zarrabi, A.; Mohammadinejad, R.; Taraghdari, Z. B.; Ashrafizadeh, M.; Zare, E. N.; Agarwal, T.; Padil, V. V. T.; Mokhtari, B.; Rossi, F.; Perale, G.; Sillanpaa, M.; Borzacchiello, A.; Kumar Maiti, T.; Makvandi, P. Prosthesis 2020, 2, 117– 139, DOI: 10.3390/prosthesis2020012Google ScholarThere is no corresponding record for this reference.
- 13Nashchekina, Y.; Chabina, A.; Moskalyuk, O.; Voronkina, I.; Evstigneeva, P.; Vaganov, G.; Nashchekin, A.; Yudin, V.; Mikhailova, N. Polymers 2022, 14, 4654 DOI: 10.3390/polym14214654Google ScholarThere is no corresponding record for this reference.
- 14Shen, J.; Yuan, W.; Badv, M.; Moshaverinia, A.; Weiss, P. S. ACS Mater. Au 2023, 3, 540– 547, DOI: 10.1021/acsmaterialsau.3c00027Google ScholarThere is no corresponding record for this reference.
- 15Leroux, A.; Ngoc Nguyen, T.; Rangel, A.; Cacciapuoti, I.; Duprez, D.; Castner, D. G.; Migonney, V. Biointerphases 2020, 15, 061006 DOI: 10.1116/6.0000429Google ScholarThere is no corresponding record for this reference.
- 16Feng, S.; Yue, Y.; Chen, J.; Zhou, J.; Li, Y.; Zhang, Q. Environ. Sci.: Processes Impacts 2020, 22, 2332– 2344, DOI: 10.1039/D0EM00340AGoogle ScholarThere is no corresponding record for this reference.
- 17Barber, V. J.; Borden, M. A.; Alty, J. W.; Tran, L. D.; Koerner, H.; Baldwin, L. A.; Alexanian, E. J.; Leibfarth, F. A. Macromolecules 2023, 56, 3679– 3686, DOI: 10.1021/acs.macromol.3c00125Google ScholarThere is no corresponding record for this reference.
- 18Castilla-Cortázar, I.; Más-Estellés, J.; Meseguer-Dueñas, J.; Escobar Ivirico, J.; Marí, B.; Vidaurre, A. Polym. Degrad. Stab. 2012, 97, 1241– 1248, DOI: 10.1016/j.polymdegradstab.2012.05.038Google ScholarThere is no corresponding record for this reference.
- 19Bhadran, A.; Shah, T.; Babanyinah, G. K.; Polara, H.; Taslimy, S.; Biewer, M. C.; Stefan, M. C. Pharmaceutics 2023, 15, 1977 DOI: 10.3390/pharmaceutics15071977Google ScholarThere is no corresponding record for this reference.
- 20Schmid, F. ACS Polym. Au 2023, 3, 28– 58, DOI: 10.1021/acspolymersau.2c00049Google ScholarThere is no corresponding record for this reference.
- 21Kirchner, B. J. Chem. Phys. 2005, 123, 204116 DOI: 10.1063/1.2126977Google ScholarThere is no corresponding record for this reference.
- 22Kirchner, B. P. Phys. Rep. 2007, 440, 1– 111, DOI: 10.1016/j.physrep.2006.11.005Google ScholarThere is no corresponding record for this reference.
- 23von Domaros, M.; Perlt, E.; Ingenmey, J.; Marchelli, G.; Kirchner, B. SoftwareX 2018, 7, 356– 359, DOI: 10.1016/j.softx.2018.11.002Google ScholarThere is no corresponding record for this reference.
- 24Perlt, E.; Friedrich, J.; von Domaros, M.; Kirchner, B. ChemPhysChem 2011, 12, 3474– 3482, DOI: 10.1002/cphc.201100592Google ScholarThere is no corresponding record for this reference.
- 25Brüssel, M.; Perlt, E.; Lehmann, S. B. C.; von Domaros, M.; Kirchner, B. J. Chem. Phys. 2011, 135, 194113 DOI: 10.1063/1.3662071Google ScholarThere is no corresponding record for this reference.
- 26Ludwig, R.; Weinhold, F. J. Chem. Phys. 1999, 110, 508– 515, DOI: 10.1063/1.478136Google ScholarThere is no corresponding record for this reference.
- 27Perlt, E.; von Domaros, M.; Kirchner, B.; Ludwig, R.; Weinhold, F. Sci. Rep. 2017, 7, 10244Google ScholarThere is no corresponding record for this reference.
- 28Blasius, J.; Ingenmey, J.; Perlt, E.; von Domaros, M.; Hollóczki, O.; Kirchner, B. Angew. Chem., Int. Ed. 2019, 58, 3212– 3216, DOI: 10.1002/anie.201811839Google ScholarThere is no corresponding record for this reference.
- 29Ren, J. M.; Fu, Q.; Blencowe, A.; Qiao, G. G. ACS Macro Lett. 2012, 1, 681– 686, DOI: 10.1021/mz300169mGoogle ScholarThere is no corresponding record for this reference.
- 30El Habnouni, S.; Blanquer, S.; Darcos, V.; Coudane, J. J. Polym. Sci., Part A: Polym. Chem. 2009, 47, 6104– 6115, DOI: 10.1002/pola.23652Google ScholarThere is no corresponding record for this reference.
- 31O’Boyle, N. M.; Banck, M.; James, C. A.; Morley, C.; Vandermeersch, T.; Hutchison, G. R. J. Cheminf. 2011, 3, 33 DOI: 10.1186/1758-2946-3-33Google ScholarThere is no corresponding record for this reference.
- 32Balasubramani, S. G.; Chen, G. P.; Coriani, S.; Diedenhofen, M.; Frank, M. S.; Franzke, Y. J.; Furche, F.; Grotjahn, R.; Harding, M. E.; Hättig, C.; Hellweg, A.; Helmich-Paris, B.; Holzer, C.; Huniar, U.; Kaupp, M.; Marefat Khah, A.; Karbalaei Khani, S.; Müller, T.; Mack, F.; Nguyen, B. D.; Parker, S. M.; Perlt, E.; Rappoport, D.; Reiter, K.; Roy, S.; Rückert, M.; Schmitz, G.; Sierka, M.; Tapavicza, E.; Tew, D. P.; Van Wüllen, C.; Voora, V. K.; Weigend, F.; Wodyński, A.; Yu, J. M. J. Chem. Phys. 2020, 152, 184107 DOI: 10.1063/5.0004635Google ScholarThere is no corresponding record for this reference.
- 33Franzke, Y. J.; Holzer, C.; Andersen, J. H.; Begušić, T.; Bruder, F.; Coriani, S.; Della Sala, F.; Fabiano, E.; Fedotov, D. A.; Fürst, S.; Gillhuber, S.; Grotjahn, R.; Kaupp, M.; Kehry, M.; Krstić, M.; Mack, F.; Majumdar, S.; Nguyen, B. D.; Parker, S. M.; Pauly, F.; Pausch, A.; Perlt, E.; Phun, G. S.; Rajabi, A.; Rappoport, D.; Samal, B.; Schrader, T.; Sharma, M.; Tapavicza, E.; Treß, R. S.; Voora, V.; Wodyński, A.; Yu, J. M.; Zerulla, B.; Furche, F.; Hättig, C.; Sierka, M.; Tew, D. P.; Weigend, F. J. Chem. Theory Comput. 2023, 19, 6859– 6890, DOI: 10.1021/acs.jctc.3c00347Google ScholarThere is no corresponding record for this reference.
- 34Grimme, S.; Brandenburg, J. G.; Bannwarth, C.; Hansen, A. J. Chem. Phys. 2015, 143, 054107 DOI: 10.1063/1.4927476Google ScholarThere is no corresponding record for this reference.
- 35Adamo, C.; Barone, V. J. Chem. Phys. 1999, 110, 6158– 6170, DOI: 10.1063/1.478522Google ScholarThere is no corresponding record for this reference.
- 36Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. J. Chem. Phys. 2010, 132, 154104 DOI: 10.1063/1.3382344Google ScholarThere is no corresponding record for this reference.
- 37Grimme, S.; Ehrlich, S.; Goerigk, L. J. Comput. Chem. 2011, 32, 1456– 1465, DOI: 10.1002/jcc.21759Google ScholarThere is no corresponding record for this reference.
- 38Fernández-Tena, A.; Pérez-Camargo, R. A.; Coulembier, O.; Sangroniz, L.; Aranburu, N.; Guerrica-Echevarria, G.; Liu, G.; Wang, D.; Cavallo, D.; Müller, A. J. Effect of Molecular Weight on the Crystallization and Melt Memory of Poly(ε-caprolactone) (PCL). Macromolecules 2023, 56, 4602– 4620, DOI: 10.1021/acs.macromol.3c00234Google ScholarThere is no corresponding record for this reference.
- 39Ju, Y.; Zhang, M.; Zhao, H. Polym. Chem. 2017, 8, 5415– 5426, DOI: 10.1039/C7PY01012EGoogle ScholarThere is no corresponding record for this reference.
- 40Klonos, P. A.; Bikiaris, N. D.; Christodoulou, E.; Zamboulis, A.; Papageorgiou, G. Z.; Kyritsis, A. Polymer 2022, 242, 124603 DOI: 10.1016/j.polymer.2022.124603Google ScholarThere is no corresponding record for this reference.
- 41Persenaire, O.; Alexandre, M.; Degée, P.; Dubois, P. Biomacromolecules 2001, 2, 288– 294, DOI: 10.1021/bm0056310Google ScholarThere is no corresponding record for this reference.
- 42Tenorio-Alfonso, A.; Ramos, E. V.; Martínez, I.; Ambrosi, M.; Raudino, M. Assessment of the structures contribution (crystalline and mesophases) and mechanical properties of polycaprolactone/pluronic blends. J. Mech. Behav. Biomed. Mater. 2023, 139, 105668 DOI: 10.1016/j.jmbbm.2023.105668Google ScholarThere is no corresponding record for this reference.
- 43Singh, P. S. Membrane Characterization; Hilal, N.; Ismail, A. F.; Matsuura, T.; Oatley-Radcliffe, D., Eds.; Elsevier, 2017; pp 95– 111.Google ScholarThere is no corresponding record for this reference.
- 44Nanaki, S. G.; Papageorgiou, G. Z.; Bikiaris, D. N. J. Therm. Anal. Calorim. 2012, 108, 633– 645, DOI: 10.1007/s10973-011-2155-8Google ScholarThere is no corresponding record for this reference.
- 45Holzwarth, U.; Gibson, N. Nat. Nanotechnol. 2011, 6, 534, DOI: 10.1038/nnano.2011.145Google ScholarThere is no corresponding record for this reference.
- 46Tencé-Girault, S.; Lebreton, S.; Bunau, O.; Dang, P.; Bargain, F. Crystals 2019, 9, 271 DOI: 10.3390/cryst9050271Google ScholarThere is no corresponding record for this reference.
- 47Wang, Z.; He, Y.; Müller, A. J. Polymer 2023, 271, 125783 DOI: 10.1016/j.polymer.2023.125783Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
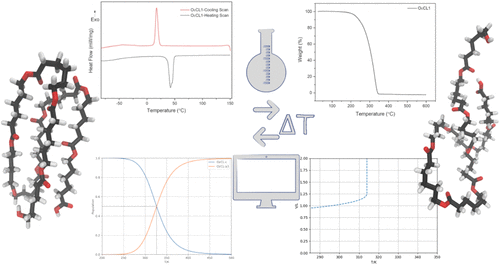
Figure 1

Figure 1. Example structures, as used in the QCE study. Crystalline motif (OεCL-c, left) as well as one exemplary amorphous structure (OεCL-a3, right).
Scheme 1
 Scheme 1. Oligomer Formation of εCL with BnOH/SnOct2 as the Initiator/Catalyst
Scheme 1. Oligomer Formation of εCL with BnOH/SnOct2 as the Initiator/CatalystFigure 2
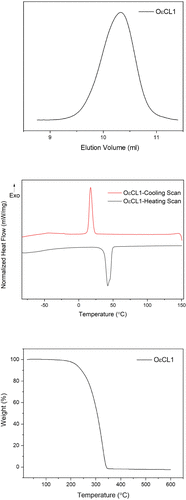
Figure 2. Characterization of the OεCL1 oligomer. Top, SEC chromatogram; middle, DSC heating and cooling scans; bottom, TGA curve.
Figure 3
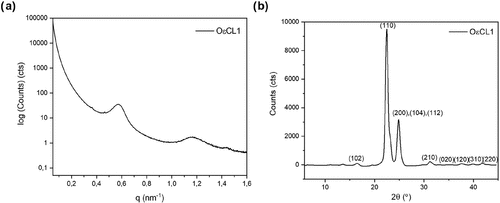
Figure 3. 1D integrated (a) SAXS patterns of OεCL1 corresponding to q and (b) WAXS patterns of OεCL1 corresponding to 2θ.
Figure 4
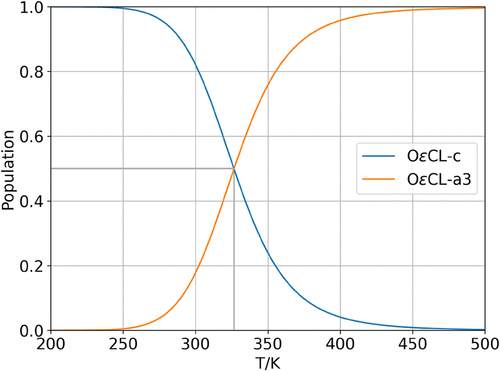
Figure 4. Temperature-dependent cluster populations showing only significantly populated clusters (>1%). The gray line indicates the interception of both curves.
Figure 5

Figure 5. Phase volume of 1 mol OεCL. At 314 K, a sharp phase transition in the volume is observed.
References
This article references 47 other publications.
- 1Gupta, V.; Biswas, D.; Roy, S. Materials 2022, 15 (17), 5899 DOI: 10.3390/ma15175899There is no corresponding record for this reference.
- 2Tajeddin, B.; Arabkhedri, M. Polymer Science and Innovative Applications; AlMaadeed, M. A. A.; Ponnamma, D.; Carignano, M. A., Eds.; Elsevier, 2020; pp 525– 543.There is no corresponding record for this reference.
- 3Hu, J.; Chen, S. J. Mater. Chem. 2010, 20, 3346– 3355, DOI: 10.1039/b922872aThere is no corresponding record for this reference.
- 4Moghadasi, K.; Mohd Isa, M. S.; Ariffin, M. A.; Mohd jamil, M. Z.; Raja, S.; Wu, B.; Yamani, M.; Bin Muhamad, M. R.; Yusof, F.; Jamaludin, M. F.; bin Ab Karim, M. S.; binti Abdul Razak, B.; bin Yusoff, N. J. Mater. Res. Technol. 2022, 17, 1054– 1121There is no corresponding record for this reference.
- 5Chen, W.-H.; Chen, Q.-W.; Chen, Q.; Cui, C.; Duan, S.; Kang, Y.; Liu, Y.; Liu, Y.; Muhammad, W.; Shao, S.; Tang, C.; Wang, J.; Wang, L.; Xiong, M.-H.; Yin, L.; Zhang, K.; Zhang, Z.; Zhen, X.; Feng, J.; Chen, X. Sci. China: Chem. 2022, 65, 1010– 1075, DOI: 10.1007/s11426-022-1243-5There is no corresponding record for this reference.
- 6Ding, L.; Yu, Z.-D.; Wang, X.-Y.; Yao, Z.-F.; Lu, Y.; Yang, C.-Y.; Wang, J.-Y.; Pei, J. Chem. Rev. 2023, 123, 7421– 7497, DOI: 10.1021/acs.chemrev.2c00696There is no corresponding record for this reference.
- 7Li, L.; Han, L.; Hu, H.; Zhang, R. Mater. Adv. 2023, 4, 726– 746, DOI: 10.1039/D2MA00940DThere is no corresponding record for this reference.
- 8Campbell, I. R.; Lin, M.-Y.; Iyer, H.; Parker, M.; Fredricks, J. L.; Liao, K.; Jimenez, A. M.; Grandgeorge, P.; Roumeli, E. Annu. Rev. Mater. Res. 2023, 53, 81– 104, DOI: 10.1146/annurev-matsci-080921-083655There is no corresponding record for this reference.
- 9Jayakumar, A.; Radoor, S.; Siengchin, S.; Shin, G. H.; Kim, J. T. Sci. Total Environ. 2023, 878, 163156 DOI: 10.1016/j.scitotenv.2023.163156There is no corresponding record for this reference.
- 10Sánchez, C. Biotechnol. Adv. 2020, 40, 107501 DOI: 10.1016/j.biotechadv.2019.107501There is no corresponding record for this reference.
- 11EL-Ghoul, Y.; Alminderej, F. M.; Alsubaie, F. M.; Alrasheed, R.; Almousa, N. H. Polymers 2021, 13 (24), 4327 DOI: 10.3390/polym13244327There is no corresponding record for this reference.
- 12Delfi, M.; Ghomi, M.; Zarrabi, A.; Mohammadinejad, R.; Taraghdari, Z. B.; Ashrafizadeh, M.; Zare, E. N.; Agarwal, T.; Padil, V. V. T.; Mokhtari, B.; Rossi, F.; Perale, G.; Sillanpaa, M.; Borzacchiello, A.; Kumar Maiti, T.; Makvandi, P. Prosthesis 2020, 2, 117– 139, DOI: 10.3390/prosthesis2020012There is no corresponding record for this reference.
- 13Nashchekina, Y.; Chabina, A.; Moskalyuk, O.; Voronkina, I.; Evstigneeva, P.; Vaganov, G.; Nashchekin, A.; Yudin, V.; Mikhailova, N. Polymers 2022, 14, 4654 DOI: 10.3390/polym14214654There is no corresponding record for this reference.
- 14Shen, J.; Yuan, W.; Badv, M.; Moshaverinia, A.; Weiss, P. S. ACS Mater. Au 2023, 3, 540– 547, DOI: 10.1021/acsmaterialsau.3c00027There is no corresponding record for this reference.
- 15Leroux, A.; Ngoc Nguyen, T.; Rangel, A.; Cacciapuoti, I.; Duprez, D.; Castner, D. G.; Migonney, V. Biointerphases 2020, 15, 061006 DOI: 10.1116/6.0000429There is no corresponding record for this reference.
- 16Feng, S.; Yue, Y.; Chen, J.; Zhou, J.; Li, Y.; Zhang, Q. Environ. Sci.: Processes Impacts 2020, 22, 2332– 2344, DOI: 10.1039/D0EM00340AThere is no corresponding record for this reference.
- 17Barber, V. J.; Borden, M. A.; Alty, J. W.; Tran, L. D.; Koerner, H.; Baldwin, L. A.; Alexanian, E. J.; Leibfarth, F. A. Macromolecules 2023, 56, 3679– 3686, DOI: 10.1021/acs.macromol.3c00125There is no corresponding record for this reference.
- 18Castilla-Cortázar, I.; Más-Estellés, J.; Meseguer-Dueñas, J.; Escobar Ivirico, J.; Marí, B.; Vidaurre, A. Polym. Degrad. Stab. 2012, 97, 1241– 1248, DOI: 10.1016/j.polymdegradstab.2012.05.038There is no corresponding record for this reference.
- 19Bhadran, A.; Shah, T.; Babanyinah, G. K.; Polara, H.; Taslimy, S.; Biewer, M. C.; Stefan, M. C. Pharmaceutics 2023, 15, 1977 DOI: 10.3390/pharmaceutics15071977There is no corresponding record for this reference.
- 20Schmid, F. ACS Polym. Au 2023, 3, 28– 58, DOI: 10.1021/acspolymersau.2c00049There is no corresponding record for this reference.
- 21Kirchner, B. J. Chem. Phys. 2005, 123, 204116 DOI: 10.1063/1.2126977There is no corresponding record for this reference.
- 22Kirchner, B. P. Phys. Rep. 2007, 440, 1– 111, DOI: 10.1016/j.physrep.2006.11.005There is no corresponding record for this reference.
- 23von Domaros, M.; Perlt, E.; Ingenmey, J.; Marchelli, G.; Kirchner, B. SoftwareX 2018, 7, 356– 359, DOI: 10.1016/j.softx.2018.11.002There is no corresponding record for this reference.
- 24Perlt, E.; Friedrich, J.; von Domaros, M.; Kirchner, B. ChemPhysChem 2011, 12, 3474– 3482, DOI: 10.1002/cphc.201100592There is no corresponding record for this reference.
- 25Brüssel, M.; Perlt, E.; Lehmann, S. B. C.; von Domaros, M.; Kirchner, B. J. Chem. Phys. 2011, 135, 194113 DOI: 10.1063/1.3662071There is no corresponding record for this reference.
- 26Ludwig, R.; Weinhold, F. J. Chem. Phys. 1999, 110, 508– 515, DOI: 10.1063/1.478136There is no corresponding record for this reference.
- 27Perlt, E.; von Domaros, M.; Kirchner, B.; Ludwig, R.; Weinhold, F. Sci. Rep. 2017, 7, 10244There is no corresponding record for this reference.
- 28Blasius, J.; Ingenmey, J.; Perlt, E.; von Domaros, M.; Hollóczki, O.; Kirchner, B. Angew. Chem., Int. Ed. 2019, 58, 3212– 3216, DOI: 10.1002/anie.201811839There is no corresponding record for this reference.
- 29Ren, J. M.; Fu, Q.; Blencowe, A.; Qiao, G. G. ACS Macro Lett. 2012, 1, 681– 686, DOI: 10.1021/mz300169mThere is no corresponding record for this reference.
- 30El Habnouni, S.; Blanquer, S.; Darcos, V.; Coudane, J. J. Polym. Sci., Part A: Polym. Chem. 2009, 47, 6104– 6115, DOI: 10.1002/pola.23652There is no corresponding record for this reference.
- 31O’Boyle, N. M.; Banck, M.; James, C. A.; Morley, C.; Vandermeersch, T.; Hutchison, G. R. J. Cheminf. 2011, 3, 33 DOI: 10.1186/1758-2946-3-33There is no corresponding record for this reference.
- 32Balasubramani, S. G.; Chen, G. P.; Coriani, S.; Diedenhofen, M.; Frank, M. S.; Franzke, Y. J.; Furche, F.; Grotjahn, R.; Harding, M. E.; Hättig, C.; Hellweg, A.; Helmich-Paris, B.; Holzer, C.; Huniar, U.; Kaupp, M.; Marefat Khah, A.; Karbalaei Khani, S.; Müller, T.; Mack, F.; Nguyen, B. D.; Parker, S. M.; Perlt, E.; Rappoport, D.; Reiter, K.; Roy, S.; Rückert, M.; Schmitz, G.; Sierka, M.; Tapavicza, E.; Tew, D. P.; Van Wüllen, C.; Voora, V. K.; Weigend, F.; Wodyński, A.; Yu, J. M. J. Chem. Phys. 2020, 152, 184107 DOI: 10.1063/5.0004635There is no corresponding record for this reference.
- 33Franzke, Y. J.; Holzer, C.; Andersen, J. H.; Begušić, T.; Bruder, F.; Coriani, S.; Della Sala, F.; Fabiano, E.; Fedotov, D. A.; Fürst, S.; Gillhuber, S.; Grotjahn, R.; Kaupp, M.; Kehry, M.; Krstić, M.; Mack, F.; Majumdar, S.; Nguyen, B. D.; Parker, S. M.; Pauly, F.; Pausch, A.; Perlt, E.; Phun, G. S.; Rajabi, A.; Rappoport, D.; Samal, B.; Schrader, T.; Sharma, M.; Tapavicza, E.; Treß, R. S.; Voora, V.; Wodyński, A.; Yu, J. M.; Zerulla, B.; Furche, F.; Hättig, C.; Sierka, M.; Tew, D. P.; Weigend, F. J. Chem. Theory Comput. 2023, 19, 6859– 6890, DOI: 10.1021/acs.jctc.3c00347There is no corresponding record for this reference.
- 34Grimme, S.; Brandenburg, J. G.; Bannwarth, C.; Hansen, A. J. Chem. Phys. 2015, 143, 054107 DOI: 10.1063/1.4927476There is no corresponding record for this reference.
- 35Adamo, C.; Barone, V. J. Chem. Phys. 1999, 110, 6158– 6170, DOI: 10.1063/1.478522There is no corresponding record for this reference.
- 36Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. J. Chem. Phys. 2010, 132, 154104 DOI: 10.1063/1.3382344There is no corresponding record for this reference.
- 37Grimme, S.; Ehrlich, S.; Goerigk, L. J. Comput. Chem. 2011, 32, 1456– 1465, DOI: 10.1002/jcc.21759There is no corresponding record for this reference.
- 38Fernández-Tena, A.; Pérez-Camargo, R. A.; Coulembier, O.; Sangroniz, L.; Aranburu, N.; Guerrica-Echevarria, G.; Liu, G.; Wang, D.; Cavallo, D.; Müller, A. J. Effect of Molecular Weight on the Crystallization and Melt Memory of Poly(ε-caprolactone) (PCL). Macromolecules 2023, 56, 4602– 4620, DOI: 10.1021/acs.macromol.3c00234There is no corresponding record for this reference.
- 39Ju, Y.; Zhang, M.; Zhao, H. Polym. Chem. 2017, 8, 5415– 5426, DOI: 10.1039/C7PY01012EThere is no corresponding record for this reference.
- 40Klonos, P. A.; Bikiaris, N. D.; Christodoulou, E.; Zamboulis, A.; Papageorgiou, G. Z.; Kyritsis, A. Polymer 2022, 242, 124603 DOI: 10.1016/j.polymer.2022.124603There is no corresponding record for this reference.
- 41Persenaire, O.; Alexandre, M.; Degée, P.; Dubois, P. Biomacromolecules 2001, 2, 288– 294, DOI: 10.1021/bm0056310There is no corresponding record for this reference.
- 42Tenorio-Alfonso, A.; Ramos, E. V.; Martínez, I.; Ambrosi, M.; Raudino, M. Assessment of the structures contribution (crystalline and mesophases) and mechanical properties of polycaprolactone/pluronic blends. J. Mech. Behav. Biomed. Mater. 2023, 139, 105668 DOI: 10.1016/j.jmbbm.2023.105668There is no corresponding record for this reference.
- 43Singh, P. S. Membrane Characterization; Hilal, N.; Ismail, A. F.; Matsuura, T.; Oatley-Radcliffe, D., Eds.; Elsevier, 2017; pp 95– 111.There is no corresponding record for this reference.
- 44Nanaki, S. G.; Papageorgiou, G. Z.; Bikiaris, D. N. J. Therm. Anal. Calorim. 2012, 108, 633– 645, DOI: 10.1007/s10973-011-2155-8There is no corresponding record for this reference.
- 45Holzwarth, U.; Gibson, N. Nat. Nanotechnol. 2011, 6, 534, DOI: 10.1038/nnano.2011.145There is no corresponding record for this reference.
- 46Tencé-Girault, S.; Lebreton, S.; Bunau, O.; Dang, P.; Bargain, F. Crystals 2019, 9, 271 DOI: 10.3390/cryst9050271There is no corresponding record for this reference.
- 47Wang, Z.; He, Y.; Müller, A. J. Polymer 2023, 271, 125783 DOI: 10.1016/j.polymer.2023.125783There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06385.
Snapshots of all cluster structures considered in the QCE calculation (Figures S1–S8) and the 1H NMR spectrum of OεCL1 (Figure S9) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



