Molecular-Scale Insights into the Interactions between Perfluoroalkyl Substances and PolyethyleneClick to copy article linkArticle link copied!
- Dandara Freitas ThomazDandara Freitas ThomazChemical Engineering Graduate Program, Rio de Janeiro State University, Rio de Janeiro, RJ 20550-900, BrazilMore by Dandara Freitas Thomaz
- Eduardo Rocha de Almeida LimaEduardo Rocha de Almeida LimaChemical Engineering Graduate Program, Rio de Janeiro State University, Rio de Janeiro, RJ 20550-900, BrazilMore by Eduardo Rocha de Almeida Lima
- Nathalia Salles Vernin*Nathalia Salles Vernin*Email: [email protected]Chemical Engineering Graduate Program, Rio de Janeiro State University, Rio de Janeiro, RJ 20550-900, BrazilMore by Nathalia Salles Vernin
Abstract
Microplastics (MPs) and per- and polyfluoroalkyl substances (PFAS) are two classes of highly persistent contaminants that frequently co-occur in the environment, raising concern about potential synergistic effects. To better understand their interactions, we investigated the adsorption of perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) on polyethylene (PE) through molecular dynamics (MD) simulations. The potential of mean force (PMF) at infinite dilution was calculated for both the semicrystalline and crystalline PE models. For semicrystalline PE systems, the PMF minima were −26.5 ± 4.8 kJ mol–1 for PFOA and −43.9 ± 4.3 kJ mol–1 for PFOS, whereas, for crystalline PE, the values were −26.6 ± 5.2 and −42.0 ± 7.7 kJ mol–1, respectively. These results indicate that, within statistical uncertainty, no significant differences are observed between the two PE morphologies for either PFAS when considering the depth of the free-energy minimum. Moreover, PFOS exhibited stronger interactions with PE than PFOA. This behavior reflects not only differences in fluoroalkyl chain length but also the distinct chemical nature of the functional groups, with the larger and more hydrophobic sulfonate headgroup of PFOS compared to the carboxylate group of PFOA. In addition to adsorption strength, molecular orientation at the PE–water interface was characterized. PFAS tails showed a general tendency to align parallel to PE chains within the polymer slab, but this alignment was disrupted upon the transition into water. Notably, PFOS interacting with semicrystalline PE exhibited orientation changes with transitions between parallel and perpendicular alignment associated with local PMF barriers. These orientation-dependent interactions highlight the importance of both chain packing and functional group chemistry in driving PFAS–polymer affinity. Taken together, these findings provide molecular-scale evidence that microplastics can act as reservoirs for PFAS, potentially enhancing their environmental persistence and transport.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of The Journal of Physical Chemistry B special issue “Physical Chemistry of Microplastics and Nanoplastics”.
Introduction
Methodology
General Remarks on Force Field
System Modeling
Figure 1
Figure 1. Insertion of water into the (A) semicrystalline polyethylene simulation box and (B) crystalline polyethylene simulation box, followed by equilibration of the systems. The molecular coordinates were wrapped within the central simulation cell while preserving intramolecular connectivity, thereby preventing atoms belonging to the same molecule from being artificially separated across periodic boundaries. Consequently, the apparent “voids” observed within the PE slab are not genuine empty regions. Rather, they correspond to PE atoms whose molecules extend across the boundaries of the central box and are thus represented as adjacent periodic replicas.
Computational Analysis
Umbrella Sampling (US) and PMF
Density Profiles
Order Parameter and Angle of Interaction between PE and PFAS
Figure 2
Figure 2. Schematic representation of vector calculations, highlighting (A) a representative vector formed between carbon atoms of PFAS molecules (PFOA or PFOS), (B) a representative resultant vector formed between carbon atoms of PE chains, and (C) the cylindrical sampling region defined within the PE slab. For an improved visualization of the cylinder, molecular coordinates were fully wrapped within the central simulation box.
Root-Mean-Square Deviation (RMSD)
Results and Discussion
Figure 3
Figure 3. Potential of mean force profiles obtained for PFOA and PFOS interacting with (A) semicrystalline PE and (B) crystalline PE. The dashed line indicates the Gibbs dividing surface, while the gray shaded area represents the interfacial region where PE and water molecules coexist, and the red and blue shaded areas represent the uncertainties of the respective simulation result.
| polyethylene | PFAS | minimum value (kJ mol–1) |
|---|---|---|
| semicrystalline PE | PFOA | –26.5 ± 4.8 |
| semicrystalline PE | PFOS | –43.9 ± 4.3 |
| crystalline PE | PFOA | –26.6 ± 5.2 |
| crystalline PE | PFOS | –42.0 ± 7.7 |
Figure 4
Figure 4. Order parameter for the interaction between (A) PFOA and semicrystalline PE, (B) PFOS and semicrystalline PE, (C) PFOA and crystalline PE, and (D) PFOS and crystalline PE as a function of the reaction coordinate. The dashed line marks the Gibbs dividing surface, while the gray shaded area denotes the interfacial region where PE and water molecules coexist. The purple shaded area represents the 95% confidence interval (p = 0.05).
Figure 5
Figure 5. Snapshots obtained during the PMF simulations at different separation distances between PFAS and the PE–water interface: (A) PFOA in the semicrystalline PE box, (B) PFOS in the semicrystalline PE box, (C) PFOA in the crystalline PE box, and (D) PFOS in the crystalline PE box.
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06774.
Force field tables, density profiles, umbrella sampling details, angle of interaction between PFAS and PE, and root-mean-square deviation of atomic positions (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We gratefully acknowledge the computational resources provided by the São Paulo National Center for High Performance Processing (CENAPAD-SP). Financial support was provided by the Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (FAPERJ, grants no. E-26/204.308/2025, SEI-260003/004552/2025, SEI-260003/015556/2021, and E-26/010.002523/2019). D.F.T. also acknowledges the scholarship awarded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil.
References
This article references 71 other publications.
- 1European Chemical Agency Microplastics, 2023. https://echa.europa.eu/hot-topics/microplastics (accessed Dec 20, 2023).Google ScholarThere is no corresponding record for this reference.
- 2Fang, C.; Sobhani, Z.; Zhang, X.; McCourt, L.; Routley, B.; Gibson, C. T.; Naidu, R. Identification and visualisation of microplastics/nanoplastics by Raman imaging (iii): algorithm to cross-check multi-images. Water Res. 2021, 194, 116913 DOI: 10.1016/j.watres.2021.116913Google ScholarThere is no corresponding record for this reference.
- 3Xu, S.; Ma, J.; Ji, R.; Pan, K.; Miao, A.-J. Microplastics in aquatic environments: Occurrence, accumulation, and biological effects. Sci. Total Environ. 2020, 703, 134699 DOI: 10.1016/j.scitotenv.2019.134699Google ScholarThere is no corresponding record for this reference.
- 4Thomaz, D. F.; Vernin, N. S.; de Almeida, R. Handbook of Microplastic Pollution in the Environment; CRC Press: Boca Raton, 2025; pp 710– 728.Google ScholarThere is no corresponding record for this reference.
- 5Glüge, J.; Scheringer, M.; Cousins, I. T.; Dewitt, J. C.; Goldenman, G.; Herzke, D.; Lohmann, R.; Ng, C. A.; Trier, X.; Wang, Z. An overview of the uses of per- and polyfluoroalkyl substances (PFAS). Environ. Sci.: Processes Impacts 2020, 22, 2345– 2373, DOI: 10.1039/D0EM00291GGoogle ScholarThere is no corresponding record for this reference.
- 6Torres, F.; Guida, Y.; Weber, R.; Torres, J. P. M. Brazilian overview of per- and polyfluoroalkyl substances listed as persistent organic pollutants in the stockholm convention. Chemosphere 2022, 291, 132674 DOI: 10.1016/j.chemosphere.2021.132674Google ScholarThere is no corresponding record for this reference.
- 7Gagliano, E.; Sgroi, M.; Falciglia, P. P.; Vagliasindi, F. G.; Roccaro, P. Removal of poly- and perfluoroalkyl substances (PFAS) from water by adsorption: Role of PFAS chain length, effect of organic matter and challenges in adsorbent regeneration. Water Res. 2020, 171, 115381 DOI: 10.1016/j.watres.2019.115381Google ScholarThere is no corresponding record for this reference.
- 8Santhanam, S. D.; Ramamurthy, K.; Priya, P. S.; Sudhakaran, G.; Guru, A.; Arockiaraj, J. A combinational threat of micro- and nano-plastics (MNPs) as potential emerging vectors for per- and polyfluoroalkyl substances (PFAS) to human health. Environ. Monit. Assess. 2024, 196, 1182 DOI: 10.1007/s10661-024-13292-9Google ScholarThere is no corresponding record for this reference.
- 9Wu, W.; Li, R.; Zhang, Z.; Liu, G.; Sun, Y.; Wang, C. The exploration of chronic combined toxic mechanisms of environmental PFOA and polyethylene micro/nanoplastics on adult zebrafish (Danio rerio), using aquatic microcosm systems. Aquat. Toxicol. 2025, 287, 107534 DOI: 10.1016/j.aquatox.2025.107534Google ScholarThere is no corresponding record for this reference.
- 10Fu, L.; Li, J.; Wang, G.; Luan, Y.; Dai, W. Adsorption behavior of organic pollutants on microplastics. Ecotoxicol. Environ. Saf. 2021, 217, 112207 DOI: 10.1016/j.ecoenv.2021.112207Google ScholarThere is no corresponding record for this reference.
- 11Ning, Z.; Zhou, S.; Yang, Y.; Li, P.; Zhao, Z.; Zhang, W.; Lu, L.; Ren, N. Adsorption behaviors of perfluorooctanoic acid on aged microplastics. Water Environ. Res. 2024, 96, e11080 DOI: 10.1002/wer.11080Google ScholarThere is no corresponding record for this reference.
- 12Bhagwat, G.; Tran, T. K. A.; Lamb, D.; Senathirajah, K.; Grainge, I.; O’Connor, W.; Juhasz, A.; Palanisami, T. Biofilms Enhance the Adsorption of Toxic Contaminants on Plastic Microfibers under Environmentally Relevant Conditions. Environ. Sci. Technol. 2021, 55, 8877– 8887, DOI: 10.1021/acs.est.1c02012Google ScholarThere is no corresponding record for this reference.
- 13Cormier, B.; Borchet, F.; Kärrman, A.; Szot, M.; Yeung, L. W.; Keiter, S. H. Sorption and desorption kinetics of PFOS to pristine microplastic. Environ. Sci. Pollut. Res. 2022, 29, 4497– 4507, DOI: 10.1007/s11356-021-15923-xGoogle ScholarThere is no corresponding record for this reference.
- 14Wang, F.; Shih, K. M.; Li, X. Y. The partition behavior of perfluorooctanesulfonate (PFOS) and perfluorooctanesulfonamide (FOSA) on microplastics. Chemosphere 2015, 119, 841– 847, DOI: 10.1016/j.chemosphere.2014.08.047Google ScholarThere is no corresponding record for this reference.
- 15Dogra, K.; Kumar, M.; Singh, S.; Bahukhandi, K. D. Decoding the interactions between microplastics, polyfluoroalkyl substances, and endocrine disruptors: sorption kinetics and toxicity. Curr. Opin. Chem. Eng. 2025, 48, 101126 DOI: 10.1016/j.coche.2025.101126Google ScholarThere is no corresponding record for this reference.
- 16Tang, K. H. D. Environmental Co-existence of Microplastics and Perfluorochemicals: A Review of Their Interactions. Biointerface Res. Appl. Chem. 2023, 13, 587 DOI: 10.33263/BRIAC136.587Google ScholarThere is no corresponding record for this reference.
- 17Freilinger, J.; Kappacher, C.; Huter, K.; Hofer, T. S.; Back, J. O.; Huck, C. W.; Bakry, R. Interactions between perfluorinated alkyl substances (PFAS) and microplastics (MPs): Findings from an extensive investigation. J. Hazard. Mater. Adv. 2025, 18, 100740 DOI: 10.1016/j.hazadv.2025.100740Google ScholarThere is no corresponding record for this reference.
- 18Singam, E. R. A.; Durkin, K. A.; La Merrill, M. A.; Furlow, J. D.; Wang, J.-C.; Smith, M. T. The vitamin D receptor as a potential target for the toxic effects of per- and polyfluoroalkyl substances (PFASs): An in-silico study. Environ. Res. 2023, 217, 114832 DOI: 10.1016/j.envres.2022.114832Google ScholarThere is no corresponding record for this reference.
- 19Ma, D.; Lu, Y.; Liang, Y.; Ruan, T.; Li, J.; Zhao, C.; Wang, Y.; Jiang, G. A Critical Review on Transplacental Transfer of Per- and Polyfluoroalkyl Substances: Prenatal Exposure Levels, Characteristics, and Mechanisms. Environ. Sci. Technol. 2022, 56, 6014– 6026, DOI: 10.1021/acs.est.1c01057Google ScholarThere is no corresponding record for this reference.
- 20Kutralam-Muniasamy, G.; Shruti, V.; Pérez-Guevara, F.; Roy, P. D. Microplastic diagnostics in humans: “The 3Ps” Progress, problems, and prospects. Sci. Total Environ. 2023, 856, 159164 DOI: 10.1016/j.scitotenv.2022.159164Google ScholarThere is no corresponding record for this reference.
- 21Amato-Lourenço, L. F.; Carvalho-Oliveira, R.; Júnior, G. R.; dos Santos Galvão, L.; Ando, R. A.; Mauad, T. Presence of airborne microplastics in human lung tissue. J. Hazard. Mater. 2021, 416, 126124 DOI: 10.1016/j.jhazmat.2021.126124Google ScholarThere is no corresponding record for this reference.
- 22Zhou, Y.; Cun, D.; Wang, Y.; Wang, Y.; Li, Y.; Jeppesen, E.; Chang, J. Perfluorooctanoic acid and concomitant microplastics pollution impact nitrogen elimination processes and increase N2O emission in wetlands through regulation of the functional microbiome. Water Res. 2025, 283, 123822 DOI: 10.1016/j.watres.2025.123822Google ScholarThere is no corresponding record for this reference.
- 23Parashar, N.; Mahanty, B.; Hait, S. Microplastics as carriers of per- and polyfluoroalkyl substances (PFAS) in aquatic environment: interactions and ecotoxicological effects. Water Emerging Contam. Nanoplast. 2023, 3, 15 DOI: 10.20517/wecn.2023.25Google ScholarThere is no corresponding record for this reference.
- 24Frenkel, D.; Smit, B. Understanding Molecular Simulation; Academic Press: San Diego, 2002.Google ScholarThere is no corresponding record for this reference.
- 25Oliveira, Y. M.; Vernin, N. S.; Bila, D. M.; Marques, M.; Tavares, F. W. Pollution caused by nanoplastics: adverse effects and mechanisms of interaction via molecular simulation. PeerJ 2022, 10, e13618 DOI: 10.7717/peerj.13618Google ScholarThere is no corresponding record for this reference.
- 26Leng, Y.; Wang, W.; Cai, H.; Chang, F.; Xiong, W.; Wang, J. Sorption kinetics, isotherms and molecular dynamics simulation of 17β-estradiol onto microplastics. Sci. Total Environ. 2023, 858, 159803 DOI: 10.1016/j.scitotenv.2022.159803Google ScholarThere is no corresponding record for this reference.
- 27Chen, Y.; Li, J.; Wang, F.; Yang, H.; Liu, L. Adsorption of tetracyclines onto polyethylene microplastics: A combined study of experiment and molecular dynamics simulation. Chemosphere 2021, 265, 129133 DOI: 10.1016/j.chemosphere.2020.129133Google ScholarThere is no corresponding record for this reference.
- 28Liu, Y.; Yang, Z.; Ju, X.; Cui, B.; Wang, J.; Wang, D.; Chen, Z.; Zhou, A. Molecular simulation of the slurrying mechanism in microplastic semi-coke water slurry. J. Mol. Model. 2024, 30, 298 DOI: 10.1007/s00894-024-06100-1Google ScholarThere is no corresponding record for this reference.
- 29Su, L.; Wang, Z.; Xiao, Z.; Xia, D.; Wang, Y.; Chen, J. Rapidly Predicting Aqueous Adsorption Constants of Organic Pollutants onto Polyethylene Microplastics by Combining Molecular Dynamics Simulations and Machine Learning. ACS ES&T Water 2024, 4, 4184– 4192, DOI: 10.1021/acsestwater.4c00463Google ScholarThere is no corresponding record for this reference.
- 30Sahnoune, M.; Tokhadzé, N.; Devémy, J.; Dequidt, A.; Goujon, F.; Chennell, P.; Sautou, V.; Malfreyt, P. Understanding and Characterizing the Drug Sorption to PVC and PE Materials. ACS Appl. Mater. Interfaces 2021, 13, 18594– 18603, DOI: 10.1021/acsami.1c03284Google ScholarThere is no corresponding record for this reference.
- 31Wang, Q.; Xu, R.; Zha, F.; Xu, L.; Kang, B.; Han, H. Molecular insights into the adsorption behavior of PFAS on montmorillonite, polyethylene, and polypropylene: A molecular dynamics study. Comput. Geotech. 2025, 187, 107461 DOI: 10.1016/j.compgeo.2025.107461Google ScholarThere is no corresponding record for this reference.
- 32Enyoh, C. E.; Qingyue, W.; Ovuoraye, P. E.; Lu, S.; Egbosiuba, T. C. Perfluorooctanesulfonic acid Sorption onto Polyethylene Microplastics: A Simulation-Driven Response Surface Optimization via Central Composite Design. J. Eng. Technol. Sci. 2025, 57, 11– 26, DOI: 10.5614/j.eng.technol.sci.2025.57.1.2Google ScholarThere is no corresponding record for this reference.
- 33Enyoh, C. E.; Wang, Q.; Wang, W.; Chowdhury, T.; Rabin, M. H.; Islam, R.; Yue, G.; Yichun, L.; Xiao, K. Sorption of Per- and Polyfluoroalkyl Substances (PFAS) using Polyethylene (PE) microplastics as adsorbent: Grand Canonical Monte Carlo and Molecular Dynamics (GCMC-MD) studies. Int. J. Environ. Anal. Chem. 2024, 104, 2719– 2735, DOI: 10.1080/03067319.2022.2070016Google ScholarThere is no corresponding record for this reference.
- 34Berendsen, H. J. C.; Grigera, J. R.; Straatsma, T. P. The Missing Term in Effective Pair Potentials. J. Phys. Chem. A 1987, 91, 6269– 6271, DOI: 10.1021/j100308a038Google ScholarThere is no corresponding record for this reference.
- 35Jorgensen, W. L.; Maxwell, D. S.; Tirado-Rives, J. Development and Testing of the OLPS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 1996, 118, 11225– 11236, DOI: 10.1021/ja9621760Google ScholarThere is no corresponding record for this reference.
- 36Watkins, E. K.; Jorgensen, W. L. Perfluoroalkanes: Conformational Analysis and Liquid-State Properties from ab Initio and Monte Carlo Calculations. J. Phys. Chem. A 2001, 105, 4118– 4125, DOI: 10.1021/jp004071wGoogle ScholarThere is no corresponding record for this reference.
- 37Yabe, M.; Mori, K.; Ueda, K.; Takeda, M. Development of PolyParGen Software to Facilitate the Determination of Molecular Dynamics Simulation Parameters for Polymers. J. Comput. Chem. Jpn. Int. Ed. 2019, 5, 2018-0034 DOI: 10.2477/jccjie.2018-0034Google ScholarThere is no corresponding record for this reference.
- 38Jorgensen, W. L.; Tirado-Rives, J. Potential energy functions for atomic-level simulations of water and organic and biomolecular systems. Proc. Natl. Acad. Sci. U.S.A. 2005, 102, 6665– 6670, DOI: 10.1073/pnas.0408037102Google ScholarThere is no corresponding record for this reference.
- 39Dodda, L. S.; Vilseck, J. Z.; Tirado-Rives, J.; Jorgensen, W. L. 1.14*CM1A-LBCC: Localized Bond-Charge Corrected CM1A Charges for Condensed-Phase Simulations. J. Phys. Chem. B 2017, 121, 3864– 3870, DOI: 10.1021/acs.jpcb.7b00272Google ScholarThere is no corresponding record for this reference.
- 40Dodda, L. S.; Vaca, I. C. D.; Tirado-Rives, J.; Jorgensen, W. L. LigParGen web server: An automatic OPLS-AA parameter generator for organic ligands. Nucleic Acids Res. 2017, 45, W331– W336, DOI: 10.1093/nar/gkx312Google ScholarThere is no corresponding record for this reference.
- 41Jorgensen, W. L. BOSS, Version 5.1. Biochemical and Organic Simulation System User’s Manual for UNIX, Linux, and Window , 2024.Google ScholarThere is no corresponding record for this reference.
- 42Di Battista, V.; Rowe, R. K.; Patch, D.; Weber, K. PFOA and PFOS diffusion through LLDPE and LLDPE coextruded with EVOH at 22 °C, 35 °C, and 50 °C. Waste Manage. 2020, 117, 93– 103, DOI: 10.1016/j.wasman.2020.07.036Google ScholarThere is no corresponding record for this reference.
- 43Kutsuna, S.; Hori, H. Experimental determination of Henry’s law constant of perfluorooctanoic acid (PFOA) at 298 K by means of an inert-gas stripping method with a helical plate. Atmos. Environ. 2008, 42, 8883– 8892, DOI: 10.1016/j.atmosenv.2008.09.008Google ScholarThere is no corresponding record for this reference.
- 44Moody, C. A.; Field, J. A. Perfluorinated Surfactants and the Environmental Implications of Their Use in Fire-Fighting Foams. Environ. Sci. Technol. 2000, 34, 3864– 3870, DOI: 10.1021/es991359uGoogle ScholarThere is no corresponding record for this reference.
- 45Andersson, M. P.; Olsson, M. H.; Stipp, S. L. Predicting the pKa and stability of organic acids and bases at an oil-water interface. Langmuir 2014, 30, 6437– 6445, DOI: 10.1021/la5008318Google ScholarThere is no corresponding record for this reference.
- 46Sit, I.; Fashina, B. T.; Baldo, A. P.; Leung, K.; Grassian, V. H.; Ilgen, A. G. Formic and acetic acid pKa values increase under nanoconfinement. RSC Adv. 2023, 13, 23147– 23157, DOI: 10.1039/D2RA07944EGoogle ScholarThere is no corresponding record for this reference.
- 47Bogusz, S.; Cheatham, T. E.; Brooks, B. R. Removal of pressure and free energy artifacts in charged periodic systems via net charge corrections to the Ewald potential. J. Chem. Phys. 1998, 108, 7070– 7084, DOI: 10.1063/1.476320Google ScholarThere is no corresponding record for this reference.
- 48Hockney, R. W.; Eastwood, J. W. Computer Simulation Using Particles, 1st ed.; CRC Press: Boca Raton, 1988.Google ScholarThere is no corresponding record for this reference.
- 49Abreu, C. R. A.; Segtovich, I.; Silveira, G. M. PLAYMOL: Applied Thermodynamics and Molecular Simulation Group; Federal University of Rio de Janeiro, 2021. https://github.com/atomsufrj/playmol (accessed March 5, 2023).Google ScholarThere is no corresponding record for this reference.
- 50Martínez, L.; Andrade, R.; Birgin, E. G.; Martínez, J. M. PACKMOL: A package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 2009, 30, 2157– 2164, DOI: 10.1002/jcc.21224Google ScholarThere is no corresponding record for this reference.
- 51Thompson, A. P.; Aktulga, H. M.; Berger, R.; Bolintineanu, D. S.; Brown, W. M.; Crozier, P. S.; in ’t Veld, P. J.; Kohlmeyer, A.; Moore, S. G.; Nguyen, T. D.; Shan, R.; Stevens, M. J.; Tranchida, J.; Trott, C.; Plimpton, S. J. LAMMPS - a flexible simulation tool for particle-based materials modeling at the atomic, meso, and continuum scales. Comput. Phys. Commun. 2022, 271, 108171 DOI: 10.1016/j.cpc.2021.108171Google ScholarThere is no corresponding record for this reference.
- 52Plimpton, S. Fast Parallel Algorithms for Short-Range Molecular Dynamics. J. Comput. Phys. 1995, 117, 1– 19, DOI: 10.1006/jcph.1995.1039Google ScholarThere is no corresponding record for this reference.
- 53Leach, A. Molecular Modelling: Principles and Applications, 2nd ed.; Prentice Hall: Harlow, 2001.Google ScholarThere is no corresponding record for this reference.
- 54Tuckerman, M. E. Statistical Mechanics: Theory and Molecular Simulation; Oxford University Press: New York, 2010.Google ScholarThere is no corresponding record for this reference.
- 55Torrie, G. M.; Valleau, J. P. Monte Carlo study of a phase-separating liquid mixture by umbrella sampling. J. Chem. Phys. 1977, 66, 1402– 1408, DOI: 10.1063/1.434125Google ScholarThere is no corresponding record for this reference.
- 56Kumar, S.; Rosenberg, J. M.; Bouzida, D.; Swendsen, R. H.; Kollman, P. A. The weighted histogram analysis method for free-energy calculations on biomolecules. I. The method. J. Comput. Chem. 1992, 13, 1011– 1021, DOI: 10.1002/jcc.540130812Google ScholarThere is no corresponding record for this reference.
- 57Roux, B. The calculation of the potential of mean force using computer simulations. Comput. Phys. Commun. 1995, 91, 275– 282, DOI: 10.1016/0010-4655(95)00053-IGoogle ScholarThere is no corresponding record for this reference.
- 58Jia, J.; Fan, C.; Li, J.; Peng, B.; Liang, Y.; Tsuji, T. Evaluation of the interfacial elasticity of surfactant monolayer at the CO2-water interface by molecular dynamics simulation: Screening surfactants to enhance the CO2 foam stability. Fuel 2024, 360, 130593 DOI: 10.1016/j.fuel.2023.130593Google ScholarThere is no corresponding record for this reference.
- 59Humphrey, W.; Dalke, A.; Schulten, K. VMD - Visual Molecular Dynamics. J. Mol. Graphics 1996, 14, 33– 38, DOI: 10.1016/0263-7855(96)00018-5Google ScholarThere is no corresponding record for this reference.
- 60Li, D.; Zhou, L.; Wang, X.; He, L.; Yang, X. Effect of Crystallinity of Polyethylene with Different Densities on Breakdown Strength and Conductance Property. Materials 2019, 12, 1746 DOI: 10.3390/ma12111746Google ScholarThere is no corresponding record for this reference.
- 61Yeh, I. C.; Andzelm, J. W.; Rutledge, G. C. Mechanical and Structural Characterization of Semicrystalline Polyethylene under Tensile Deformation by Molecular Dynamics Simulations. Macromolecules 2015, 48, 4228– 4239, DOI: 10.1021/acs.macromol.5b00697Google ScholarThere is no corresponding record for this reference.
- 62Zheng, S.; Sarker, P.; Gursoy, D.; Wei, T.; Hsiao, B. S. Molecular Mechanisms of Perfluoroalkyl Substances Integration into Phospholipid Membranes. Langmuir 2025, 41, 9369– 9376, DOI: 10.1021/acs.langmuir.5c00124Google ScholarThere is no corresponding record for this reference.
- 63Bao, Y.; Niu, J.; Xu, Z.; Gao, D.; Shi, J.; Sun, X.; Huang, Q. Removal of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) from water by coagulation: Mechanisms and influencing factors. J. Colloid Interface Sci. 2014, 434, 59– 64, DOI: 10.1016/j.jcis.2014.07.041Google ScholarThere is no corresponding record for this reference.
- 64Scott, J. W.; Gunderson, K. G.; Green, L. A.; Rediske, R. R.; Steinman, A. D. Perfluoroalkylated Substances (PFAS) Associated with Microplastics in a Lake Environment. Toxics 2021, 9, 106 DOI: 10.3390/toxics9050106Google ScholarThere is no corresponding record for this reference.
- 65Ke, Z.-W.; Wei, S.-J.; Shen, P.; Chen, Y.-M.; Li, Y.-C. Mechanism for the adsorption of per- and polyfluoroalkyl substances on kaolinite: Molecular dynamics modeling. Appl. Clay Sci. 2023, 232, 106804 DOI: 10.1016/j.clay.2022.106804Google ScholarThere is no corresponding record for this reference.
- 66Oliver, D. P.; Li, Y.; Orr, R.; Nelson, P.; Barnes, M.; McLaughlin, M. J.; Kookana, R. S. Sorption behaviour of per- and polyfluoroalkyl substances (PFASs) in tropical soils. Environ. Pollut. 2020, 258, 113726 DOI: 10.1016/j.envpol.2019.113726Google ScholarThere is no corresponding record for this reference.
- 67Xiang, X.; Bouazza, A.; Mikhael, E.; Scheirs, J. Perfluoroalkyl substances (PFAS) partitioning into a high-density polyethylene geomembrane. Geosynth. Int. 2025, 1– 9, DOI: 10.1680/jgein.24.00139Google ScholarThere is no corresponding record for this reference.
- 68Oliveira, Y. M.; Vernin, N. S.; Zhang, Y.; Maginn, E.; Tavares, F. W. Interaction Between Endocrine Disruptors and Polyethylene Nanoplastic by Molecular Dynamics Simulations. J. Phys. Chem. B 2024, 128, 2045– 2052, DOI: 10.1021/acs.jpcb.3c07966Google ScholarThere is no corresponding record for this reference.
- 69Dixon-Anderson, E.; Lohmann, R. Field-testing polyethylene passive samplers for the detection of neutral polyfluorinated alkyl substances in air and water. Environ. Toxicol. Chem. 2018, 37, 3002– 3010, DOI: 10.1002/etc.4264Google ScholarThere is no corresponding record for this reference.
- 70Klevan, C.; Caines, S.; Gomes, A.; Pennell, K. D. Accurate Determination of Perfluorooctanoate Aqueous Solubility, Critical Micelle Concentration, and Acid Dissociation Constant. Environ. Sci. Technol. Lett. 2024, 11, 1398– 1405, DOI: 10.1021/acs.estlett.4c00858Google ScholarThere is no corresponding record for this reference.
- 71Lei, X.; Lian, Q.; Zhang, X.; Karsili, T. K.; Holmes, W.; Chen, Y.; Zappi, M. E.; Gang, D. D. A review of PFAS adsorption from aqueous solutions: Current approaches, engineering applications, challenges, and opportunities. Environ. Pollut. 2023, 321, 121138 DOI: 10.1016/j.envpol.2023.121138Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
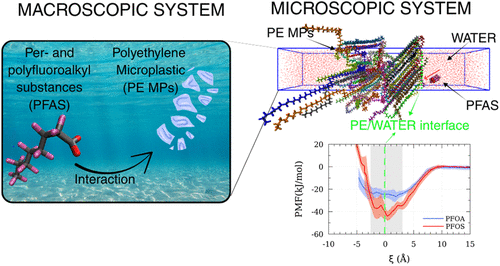
Figure 1

Figure 1. Insertion of water into the (A) semicrystalline polyethylene simulation box and (B) crystalline polyethylene simulation box, followed by equilibration of the systems. The molecular coordinates were wrapped within the central simulation cell while preserving intramolecular connectivity, thereby preventing atoms belonging to the same molecule from being artificially separated across periodic boundaries. Consequently, the apparent “voids” observed within the PE slab are not genuine empty regions. Rather, they correspond to PE atoms whose molecules extend across the boundaries of the central box and are thus represented as adjacent periodic replicas.
Figure 2

Figure 2. Schematic representation of vector calculations, highlighting (A) a representative vector formed between carbon atoms of PFAS molecules (PFOA or PFOS), (B) a representative resultant vector formed between carbon atoms of PE chains, and (C) the cylindrical sampling region defined within the PE slab. For an improved visualization of the cylinder, molecular coordinates were fully wrapped within the central simulation box.
Figure 3
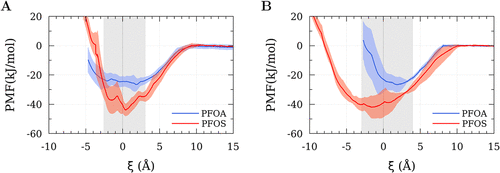
Figure 3. Potential of mean force profiles obtained for PFOA and PFOS interacting with (A) semicrystalline PE and (B) crystalline PE. The dashed line indicates the Gibbs dividing surface, while the gray shaded area represents the interfacial region where PE and water molecules coexist, and the red and blue shaded areas represent the uncertainties of the respective simulation result.
Figure 4

Figure 4. Order parameter for the interaction between (A) PFOA and semicrystalline PE, (B) PFOS and semicrystalline PE, (C) PFOA and crystalline PE, and (D) PFOS and crystalline PE as a function of the reaction coordinate. The dashed line marks the Gibbs dividing surface, while the gray shaded area denotes the interfacial region where PE and water molecules coexist. The purple shaded area represents the 95% confidence interval (p = 0.05).
Figure 5

Figure 5. Snapshots obtained during the PMF simulations at different separation distances between PFAS and the PE–water interface: (A) PFOA in the semicrystalline PE box, (B) PFOS in the semicrystalline PE box, (C) PFOA in the crystalline PE box, and (D) PFOS in the crystalline PE box.
References
This article references 71 other publications.
- 1European Chemical Agency Microplastics, 2023. https://echa.europa.eu/hot-topics/microplastics (accessed Dec 20, 2023).There is no corresponding record for this reference.
- 2Fang, C.; Sobhani, Z.; Zhang, X.; McCourt, L.; Routley, B.; Gibson, C. T.; Naidu, R. Identification and visualisation of microplastics/nanoplastics by Raman imaging (iii): algorithm to cross-check multi-images. Water Res. 2021, 194, 116913 DOI: 10.1016/j.watres.2021.116913There is no corresponding record for this reference.
- 3Xu, S.; Ma, J.; Ji, R.; Pan, K.; Miao, A.-J. Microplastics in aquatic environments: Occurrence, accumulation, and biological effects. Sci. Total Environ. 2020, 703, 134699 DOI: 10.1016/j.scitotenv.2019.134699There is no corresponding record for this reference.
- 4Thomaz, D. F.; Vernin, N. S.; de Almeida, R. Handbook of Microplastic Pollution in the Environment; CRC Press: Boca Raton, 2025; pp 710– 728.There is no corresponding record for this reference.
- 5Glüge, J.; Scheringer, M.; Cousins, I. T.; Dewitt, J. C.; Goldenman, G.; Herzke, D.; Lohmann, R.; Ng, C. A.; Trier, X.; Wang, Z. An overview of the uses of per- and polyfluoroalkyl substances (PFAS). Environ. Sci.: Processes Impacts 2020, 22, 2345– 2373, DOI: 10.1039/D0EM00291GThere is no corresponding record for this reference.
- 6Torres, F.; Guida, Y.; Weber, R.; Torres, J. P. M. Brazilian overview of per- and polyfluoroalkyl substances listed as persistent organic pollutants in the stockholm convention. Chemosphere 2022, 291, 132674 DOI: 10.1016/j.chemosphere.2021.132674There is no corresponding record for this reference.
- 7Gagliano, E.; Sgroi, M.; Falciglia, P. P.; Vagliasindi, F. G.; Roccaro, P. Removal of poly- and perfluoroalkyl substances (PFAS) from water by adsorption: Role of PFAS chain length, effect of organic matter and challenges in adsorbent regeneration. Water Res. 2020, 171, 115381 DOI: 10.1016/j.watres.2019.115381There is no corresponding record for this reference.
- 8Santhanam, S. D.; Ramamurthy, K.; Priya, P. S.; Sudhakaran, G.; Guru, A.; Arockiaraj, J. A combinational threat of micro- and nano-plastics (MNPs) as potential emerging vectors for per- and polyfluoroalkyl substances (PFAS) to human health. Environ. Monit. Assess. 2024, 196, 1182 DOI: 10.1007/s10661-024-13292-9There is no corresponding record for this reference.
- 9Wu, W.; Li, R.; Zhang, Z.; Liu, G.; Sun, Y.; Wang, C. The exploration of chronic combined toxic mechanisms of environmental PFOA and polyethylene micro/nanoplastics on adult zebrafish (Danio rerio), using aquatic microcosm systems. Aquat. Toxicol. 2025, 287, 107534 DOI: 10.1016/j.aquatox.2025.107534There is no corresponding record for this reference.
- 10Fu, L.; Li, J.; Wang, G.; Luan, Y.; Dai, W. Adsorption behavior of organic pollutants on microplastics. Ecotoxicol. Environ. Saf. 2021, 217, 112207 DOI: 10.1016/j.ecoenv.2021.112207There is no corresponding record for this reference.
- 11Ning, Z.; Zhou, S.; Yang, Y.; Li, P.; Zhao, Z.; Zhang, W.; Lu, L.; Ren, N. Adsorption behaviors of perfluorooctanoic acid on aged microplastics. Water Environ. Res. 2024, 96, e11080 DOI: 10.1002/wer.11080There is no corresponding record for this reference.
- 12Bhagwat, G.; Tran, T. K. A.; Lamb, D.; Senathirajah, K.; Grainge, I.; O’Connor, W.; Juhasz, A.; Palanisami, T. Biofilms Enhance the Adsorption of Toxic Contaminants on Plastic Microfibers under Environmentally Relevant Conditions. Environ. Sci. Technol. 2021, 55, 8877– 8887, DOI: 10.1021/acs.est.1c02012There is no corresponding record for this reference.
- 13Cormier, B.; Borchet, F.; Kärrman, A.; Szot, M.; Yeung, L. W.; Keiter, S. H. Sorption and desorption kinetics of PFOS to pristine microplastic. Environ. Sci. Pollut. Res. 2022, 29, 4497– 4507, DOI: 10.1007/s11356-021-15923-xThere is no corresponding record for this reference.
- 14Wang, F.; Shih, K. M.; Li, X. Y. The partition behavior of perfluorooctanesulfonate (PFOS) and perfluorooctanesulfonamide (FOSA) on microplastics. Chemosphere 2015, 119, 841– 847, DOI: 10.1016/j.chemosphere.2014.08.047There is no corresponding record for this reference.
- 15Dogra, K.; Kumar, M.; Singh, S.; Bahukhandi, K. D. Decoding the interactions between microplastics, polyfluoroalkyl substances, and endocrine disruptors: sorption kinetics and toxicity. Curr. Opin. Chem. Eng. 2025, 48, 101126 DOI: 10.1016/j.coche.2025.101126There is no corresponding record for this reference.
- 16Tang, K. H. D. Environmental Co-existence of Microplastics and Perfluorochemicals: A Review of Their Interactions. Biointerface Res. Appl. Chem. 2023, 13, 587 DOI: 10.33263/BRIAC136.587There is no corresponding record for this reference.
- 17Freilinger, J.; Kappacher, C.; Huter, K.; Hofer, T. S.; Back, J. O.; Huck, C. W.; Bakry, R. Interactions between perfluorinated alkyl substances (PFAS) and microplastics (MPs): Findings from an extensive investigation. J. Hazard. Mater. Adv. 2025, 18, 100740 DOI: 10.1016/j.hazadv.2025.100740There is no corresponding record for this reference.
- 18Singam, E. R. A.; Durkin, K. A.; La Merrill, M. A.; Furlow, J. D.; Wang, J.-C.; Smith, M. T. The vitamin D receptor as a potential target for the toxic effects of per- and polyfluoroalkyl substances (PFASs): An in-silico study. Environ. Res. 2023, 217, 114832 DOI: 10.1016/j.envres.2022.114832There is no corresponding record for this reference.
- 19Ma, D.; Lu, Y.; Liang, Y.; Ruan, T.; Li, J.; Zhao, C.; Wang, Y.; Jiang, G. A Critical Review on Transplacental Transfer of Per- and Polyfluoroalkyl Substances: Prenatal Exposure Levels, Characteristics, and Mechanisms. Environ. Sci. Technol. 2022, 56, 6014– 6026, DOI: 10.1021/acs.est.1c01057There is no corresponding record for this reference.
- 20Kutralam-Muniasamy, G.; Shruti, V.; Pérez-Guevara, F.; Roy, P. D. Microplastic diagnostics in humans: “The 3Ps” Progress, problems, and prospects. Sci. Total Environ. 2023, 856, 159164 DOI: 10.1016/j.scitotenv.2022.159164There is no corresponding record for this reference.
- 21Amato-Lourenço, L. F.; Carvalho-Oliveira, R.; Júnior, G. R.; dos Santos Galvão, L.; Ando, R. A.; Mauad, T. Presence of airborne microplastics in human lung tissue. J. Hazard. Mater. 2021, 416, 126124 DOI: 10.1016/j.jhazmat.2021.126124There is no corresponding record for this reference.
- 22Zhou, Y.; Cun, D.; Wang, Y.; Wang, Y.; Li, Y.; Jeppesen, E.; Chang, J. Perfluorooctanoic acid and concomitant microplastics pollution impact nitrogen elimination processes and increase N2O emission in wetlands through regulation of the functional microbiome. Water Res. 2025, 283, 123822 DOI: 10.1016/j.watres.2025.123822There is no corresponding record for this reference.
- 23Parashar, N.; Mahanty, B.; Hait, S. Microplastics as carriers of per- and polyfluoroalkyl substances (PFAS) in aquatic environment: interactions and ecotoxicological effects. Water Emerging Contam. Nanoplast. 2023, 3, 15 DOI: 10.20517/wecn.2023.25There is no corresponding record for this reference.
- 24Frenkel, D.; Smit, B. Understanding Molecular Simulation; Academic Press: San Diego, 2002.There is no corresponding record for this reference.
- 25Oliveira, Y. M.; Vernin, N. S.; Bila, D. M.; Marques, M.; Tavares, F. W. Pollution caused by nanoplastics: adverse effects and mechanisms of interaction via molecular simulation. PeerJ 2022, 10, e13618 DOI: 10.7717/peerj.13618There is no corresponding record for this reference.
- 26Leng, Y.; Wang, W.; Cai, H.; Chang, F.; Xiong, W.; Wang, J. Sorption kinetics, isotherms and molecular dynamics simulation of 17β-estradiol onto microplastics. Sci. Total Environ. 2023, 858, 159803 DOI: 10.1016/j.scitotenv.2022.159803There is no corresponding record for this reference.
- 27Chen, Y.; Li, J.; Wang, F.; Yang, H.; Liu, L. Adsorption of tetracyclines onto polyethylene microplastics: A combined study of experiment and molecular dynamics simulation. Chemosphere 2021, 265, 129133 DOI: 10.1016/j.chemosphere.2020.129133There is no corresponding record for this reference.
- 28Liu, Y.; Yang, Z.; Ju, X.; Cui, B.; Wang, J.; Wang, D.; Chen, Z.; Zhou, A. Molecular simulation of the slurrying mechanism in microplastic semi-coke water slurry. J. Mol. Model. 2024, 30, 298 DOI: 10.1007/s00894-024-06100-1There is no corresponding record for this reference.
- 29Su, L.; Wang, Z.; Xiao, Z.; Xia, D.; Wang, Y.; Chen, J. Rapidly Predicting Aqueous Adsorption Constants of Organic Pollutants onto Polyethylene Microplastics by Combining Molecular Dynamics Simulations and Machine Learning. ACS ES&T Water 2024, 4, 4184– 4192, DOI: 10.1021/acsestwater.4c00463There is no corresponding record for this reference.
- 30Sahnoune, M.; Tokhadzé, N.; Devémy, J.; Dequidt, A.; Goujon, F.; Chennell, P.; Sautou, V.; Malfreyt, P. Understanding and Characterizing the Drug Sorption to PVC and PE Materials. ACS Appl. Mater. Interfaces 2021, 13, 18594– 18603, DOI: 10.1021/acsami.1c03284There is no corresponding record for this reference.
- 31Wang, Q.; Xu, R.; Zha, F.; Xu, L.; Kang, B.; Han, H. Molecular insights into the adsorption behavior of PFAS on montmorillonite, polyethylene, and polypropylene: A molecular dynamics study. Comput. Geotech. 2025, 187, 107461 DOI: 10.1016/j.compgeo.2025.107461There is no corresponding record for this reference.
- 32Enyoh, C. E.; Qingyue, W.; Ovuoraye, P. E.; Lu, S.; Egbosiuba, T. C. Perfluorooctanesulfonic acid Sorption onto Polyethylene Microplastics: A Simulation-Driven Response Surface Optimization via Central Composite Design. J. Eng. Technol. Sci. 2025, 57, 11– 26, DOI: 10.5614/j.eng.technol.sci.2025.57.1.2There is no corresponding record for this reference.
- 33Enyoh, C. E.; Wang, Q.; Wang, W.; Chowdhury, T.; Rabin, M. H.; Islam, R.; Yue, G.; Yichun, L.; Xiao, K. Sorption of Per- and Polyfluoroalkyl Substances (PFAS) using Polyethylene (PE) microplastics as adsorbent: Grand Canonical Monte Carlo and Molecular Dynamics (GCMC-MD) studies. Int. J. Environ. Anal. Chem. 2024, 104, 2719– 2735, DOI: 10.1080/03067319.2022.2070016There is no corresponding record for this reference.
- 34Berendsen, H. J. C.; Grigera, J. R.; Straatsma, T. P. The Missing Term in Effective Pair Potentials. J. Phys. Chem. A 1987, 91, 6269– 6271, DOI: 10.1021/j100308a038There is no corresponding record for this reference.
- 35Jorgensen, W. L.; Maxwell, D. S.; Tirado-Rives, J. Development and Testing of the OLPS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 1996, 118, 11225– 11236, DOI: 10.1021/ja9621760There is no corresponding record for this reference.
- 36Watkins, E. K.; Jorgensen, W. L. Perfluoroalkanes: Conformational Analysis and Liquid-State Properties from ab Initio and Monte Carlo Calculations. J. Phys. Chem. A 2001, 105, 4118– 4125, DOI: 10.1021/jp004071wThere is no corresponding record for this reference.
- 37Yabe, M.; Mori, K.; Ueda, K.; Takeda, M. Development of PolyParGen Software to Facilitate the Determination of Molecular Dynamics Simulation Parameters for Polymers. J. Comput. Chem. Jpn. Int. Ed. 2019, 5, 2018-0034 DOI: 10.2477/jccjie.2018-0034There is no corresponding record for this reference.
- 38Jorgensen, W. L.; Tirado-Rives, J. Potential energy functions for atomic-level simulations of water and organic and biomolecular systems. Proc. Natl. Acad. Sci. U.S.A. 2005, 102, 6665– 6670, DOI: 10.1073/pnas.0408037102There is no corresponding record for this reference.
- 39Dodda, L. S.; Vilseck, J. Z.; Tirado-Rives, J.; Jorgensen, W. L. 1.14*CM1A-LBCC: Localized Bond-Charge Corrected CM1A Charges for Condensed-Phase Simulations. J. Phys. Chem. B 2017, 121, 3864– 3870, DOI: 10.1021/acs.jpcb.7b00272There is no corresponding record for this reference.
- 40Dodda, L. S.; Vaca, I. C. D.; Tirado-Rives, J.; Jorgensen, W. L. LigParGen web server: An automatic OPLS-AA parameter generator for organic ligands. Nucleic Acids Res. 2017, 45, W331– W336, DOI: 10.1093/nar/gkx312There is no corresponding record for this reference.
- 41Jorgensen, W. L. BOSS, Version 5.1. Biochemical and Organic Simulation System User’s Manual for UNIX, Linux, and Window , 2024.There is no corresponding record for this reference.
- 42Di Battista, V.; Rowe, R. K.; Patch, D.; Weber, K. PFOA and PFOS diffusion through LLDPE and LLDPE coextruded with EVOH at 22 °C, 35 °C, and 50 °C. Waste Manage. 2020, 117, 93– 103, DOI: 10.1016/j.wasman.2020.07.036There is no corresponding record for this reference.
- 43Kutsuna, S.; Hori, H. Experimental determination of Henry’s law constant of perfluorooctanoic acid (PFOA) at 298 K by means of an inert-gas stripping method with a helical plate. Atmos. Environ. 2008, 42, 8883– 8892, DOI: 10.1016/j.atmosenv.2008.09.008There is no corresponding record for this reference.
- 44Moody, C. A.; Field, J. A. Perfluorinated Surfactants and the Environmental Implications of Their Use in Fire-Fighting Foams. Environ. Sci. Technol. 2000, 34, 3864– 3870, DOI: 10.1021/es991359uThere is no corresponding record for this reference.
- 45Andersson, M. P.; Olsson, M. H.; Stipp, S. L. Predicting the pKa and stability of organic acids and bases at an oil-water interface. Langmuir 2014, 30, 6437– 6445, DOI: 10.1021/la5008318There is no corresponding record for this reference.
- 46Sit, I.; Fashina, B. T.; Baldo, A. P.; Leung, K.; Grassian, V. H.; Ilgen, A. G. Formic and acetic acid pKa values increase under nanoconfinement. RSC Adv. 2023, 13, 23147– 23157, DOI: 10.1039/D2RA07944EThere is no corresponding record for this reference.
- 47Bogusz, S.; Cheatham, T. E.; Brooks, B. R. Removal of pressure and free energy artifacts in charged periodic systems via net charge corrections to the Ewald potential. J. Chem. Phys. 1998, 108, 7070– 7084, DOI: 10.1063/1.476320There is no corresponding record for this reference.
- 48Hockney, R. W.; Eastwood, J. W. Computer Simulation Using Particles, 1st ed.; CRC Press: Boca Raton, 1988.There is no corresponding record for this reference.
- 49Abreu, C. R. A.; Segtovich, I.; Silveira, G. M. PLAYMOL: Applied Thermodynamics and Molecular Simulation Group; Federal University of Rio de Janeiro, 2021. https://github.com/atomsufrj/playmol (accessed March 5, 2023).There is no corresponding record for this reference.
- 50Martínez, L.; Andrade, R.; Birgin, E. G.; Martínez, J. M. PACKMOL: A package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 2009, 30, 2157– 2164, DOI: 10.1002/jcc.21224There is no corresponding record for this reference.
- 51Thompson, A. P.; Aktulga, H. M.; Berger, R.; Bolintineanu, D. S.; Brown, W. M.; Crozier, P. S.; in ’t Veld, P. J.; Kohlmeyer, A.; Moore, S. G.; Nguyen, T. D.; Shan, R.; Stevens, M. J.; Tranchida, J.; Trott, C.; Plimpton, S. J. LAMMPS - a flexible simulation tool for particle-based materials modeling at the atomic, meso, and continuum scales. Comput. Phys. Commun. 2022, 271, 108171 DOI: 10.1016/j.cpc.2021.108171There is no corresponding record for this reference.
- 52Plimpton, S. Fast Parallel Algorithms for Short-Range Molecular Dynamics. J. Comput. Phys. 1995, 117, 1– 19, DOI: 10.1006/jcph.1995.1039There is no corresponding record for this reference.
- 53Leach, A. Molecular Modelling: Principles and Applications, 2nd ed.; Prentice Hall: Harlow, 2001.There is no corresponding record for this reference.
- 54Tuckerman, M. E. Statistical Mechanics: Theory and Molecular Simulation; Oxford University Press: New York, 2010.There is no corresponding record for this reference.
- 55Torrie, G. M.; Valleau, J. P. Monte Carlo study of a phase-separating liquid mixture by umbrella sampling. J. Chem. Phys. 1977, 66, 1402– 1408, DOI: 10.1063/1.434125There is no corresponding record for this reference.
- 56Kumar, S.; Rosenberg, J. M.; Bouzida, D.; Swendsen, R. H.; Kollman, P. A. The weighted histogram analysis method for free-energy calculations on biomolecules. I. The method. J. Comput. Chem. 1992, 13, 1011– 1021, DOI: 10.1002/jcc.540130812There is no corresponding record for this reference.
- 57Roux, B. The calculation of the potential of mean force using computer simulations. Comput. Phys. Commun. 1995, 91, 275– 282, DOI: 10.1016/0010-4655(95)00053-IThere is no corresponding record for this reference.
- 58Jia, J.; Fan, C.; Li, J.; Peng, B.; Liang, Y.; Tsuji, T. Evaluation of the interfacial elasticity of surfactant monolayer at the CO2-water interface by molecular dynamics simulation: Screening surfactants to enhance the CO2 foam stability. Fuel 2024, 360, 130593 DOI: 10.1016/j.fuel.2023.130593There is no corresponding record for this reference.
- 59Humphrey, W.; Dalke, A.; Schulten, K. VMD - Visual Molecular Dynamics. J. Mol. Graphics 1996, 14, 33– 38, DOI: 10.1016/0263-7855(96)00018-5There is no corresponding record for this reference.
- 60Li, D.; Zhou, L.; Wang, X.; He, L.; Yang, X. Effect of Crystallinity of Polyethylene with Different Densities on Breakdown Strength and Conductance Property. Materials 2019, 12, 1746 DOI: 10.3390/ma12111746There is no corresponding record for this reference.
- 61Yeh, I. C.; Andzelm, J. W.; Rutledge, G. C. Mechanical and Structural Characterization of Semicrystalline Polyethylene under Tensile Deformation by Molecular Dynamics Simulations. Macromolecules 2015, 48, 4228– 4239, DOI: 10.1021/acs.macromol.5b00697There is no corresponding record for this reference.
- 62Zheng, S.; Sarker, P.; Gursoy, D.; Wei, T.; Hsiao, B. S. Molecular Mechanisms of Perfluoroalkyl Substances Integration into Phospholipid Membranes. Langmuir 2025, 41, 9369– 9376, DOI: 10.1021/acs.langmuir.5c00124There is no corresponding record for this reference.
- 63Bao, Y.; Niu, J.; Xu, Z.; Gao, D.; Shi, J.; Sun, X.; Huang, Q. Removal of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) from water by coagulation: Mechanisms and influencing factors. J. Colloid Interface Sci. 2014, 434, 59– 64, DOI: 10.1016/j.jcis.2014.07.041There is no corresponding record for this reference.
- 64Scott, J. W.; Gunderson, K. G.; Green, L. A.; Rediske, R. R.; Steinman, A. D. Perfluoroalkylated Substances (PFAS) Associated with Microplastics in a Lake Environment. Toxics 2021, 9, 106 DOI: 10.3390/toxics9050106There is no corresponding record for this reference.
- 65Ke, Z.-W.; Wei, S.-J.; Shen, P.; Chen, Y.-M.; Li, Y.-C. Mechanism for the adsorption of per- and polyfluoroalkyl substances on kaolinite: Molecular dynamics modeling. Appl. Clay Sci. 2023, 232, 106804 DOI: 10.1016/j.clay.2022.106804There is no corresponding record for this reference.
- 66Oliver, D. P.; Li, Y.; Orr, R.; Nelson, P.; Barnes, M.; McLaughlin, M. J.; Kookana, R. S. Sorption behaviour of per- and polyfluoroalkyl substances (PFASs) in tropical soils. Environ. Pollut. 2020, 258, 113726 DOI: 10.1016/j.envpol.2019.113726There is no corresponding record for this reference.
- 67Xiang, X.; Bouazza, A.; Mikhael, E.; Scheirs, J. Perfluoroalkyl substances (PFAS) partitioning into a high-density polyethylene geomembrane. Geosynth. Int. 2025, 1– 9, DOI: 10.1680/jgein.24.00139There is no corresponding record for this reference.
- 68Oliveira, Y. M.; Vernin, N. S.; Zhang, Y.; Maginn, E.; Tavares, F. W. Interaction Between Endocrine Disruptors and Polyethylene Nanoplastic by Molecular Dynamics Simulations. J. Phys. Chem. B 2024, 128, 2045– 2052, DOI: 10.1021/acs.jpcb.3c07966There is no corresponding record for this reference.
- 69Dixon-Anderson, E.; Lohmann, R. Field-testing polyethylene passive samplers for the detection of neutral polyfluorinated alkyl substances in air and water. Environ. Toxicol. Chem. 2018, 37, 3002– 3010, DOI: 10.1002/etc.4264There is no corresponding record for this reference.
- 70Klevan, C.; Caines, S.; Gomes, A.; Pennell, K. D. Accurate Determination of Perfluorooctanoate Aqueous Solubility, Critical Micelle Concentration, and Acid Dissociation Constant. Environ. Sci. Technol. Lett. 2024, 11, 1398– 1405, DOI: 10.1021/acs.estlett.4c00858There is no corresponding record for this reference.
- 71Lei, X.; Lian, Q.; Zhang, X.; Karsili, T. K.; Holmes, W.; Chen, Y.; Zappi, M. E.; Gang, D. D. A review of PFAS adsorption from aqueous solutions: Current approaches, engineering applications, challenges, and opportunities. Environ. Pollut. 2023, 321, 121138 DOI: 10.1016/j.envpol.2023.121138There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06774.
Force field tables, density profiles, umbrella sampling details, angle of interaction between PFAS and PE, and root-mean-square deviation of atomic positions (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



