Reduced-Resistances Model for Enhanced Drug Permeation via a Solubilizing Receiver Medium: A Mechanistic Study with Hollow Fiber MembranesClick to copy article linkArticle link copied!
- Jack D. MurrayJack D. MurraySchool of Pharmacy, University College Cork, Cork T12 K8AF, IrelandMore by Jack D. Murray
- Roshni P. PatelRoshni P. PatelSchool of Pharmacy, University of Maryland, Baltimore, Maryland 21201, United StatesMore by Roshni P. Patel
- Harriet Bennett-LenaneHarriet Bennett-LenaneSchool of Pharmacy, University College Cork, Cork T12 K8AF, IrelandMore by Harriet Bennett-Lenane
- Patrick J. O’DwyerPatrick J. O’DwyerSchool of Pharmacy, University College Cork, Cork T12 K8AF, IrelandMore by Patrick J. O’Dwyer
- Brendan T. GriffinBrendan T. GriffinSchool of Pharmacy, University College Cork, Cork T12 K8AF, IrelandMore by Brendan T. Griffin
- James E. Polli*James E. Polli*E-mail: [email protected]School of Pharmacy, University of Maryland, Baltimore, Maryland 21201, United StatesMore by James E. Polli
Abstract
A solubilizing receiver medium has been documented to increase drug flux in vitro, but the mechanisms underlying this effect remain poorly understood. This study investigated these mechanisms and established a mathematical model to describe the increase in apparent permeability. Flow rate experiments were performed to quantify the individual boundary layer and membrane resistances associated with diffusion. The impact of nine solubilizing receiver additives, including surfactants, cyclodextrins, and bovine serum albumin, on the flux of griseofulvin was investigated. The increase in apparent permeability followed the rank-order, though not the magnitude, of the solubility enhancement in the receiver (Spearman’s ρ = 0.93, p < 0.001, n = 20). The mechanistic model, termed the reduced-resistances model, demonstrates that a solubilizing receiver reduces diffusional resistance in the membrane and in the receiver-side boundary layer. At high ratios of receiver to donor solubility, a hyperbolic relationship was observed where diffusion through the donor-side boundary layer becomes rate-limiting. Additional drug cocktail permeability studies with antipyrine, phenytoin, and meloxicam confirmed the broader applicability of this model. These findings provide a framework for informed receiver selection in permeability assays and underscore the importance of considering the receiver medium when comparing results across experiments.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Introduction
Theoretical
Figure 1
Figure 1. Overview of modeling workflow.
Figure 2
Figure 2. Diagram of the HFM setup showing the volumetric flow rate (Q), concentrations (C), and volumes (V) associated with the donor (blue) and receiver (orange) circuits. The donor circuit is recycled, while the receiver circuit is single-pass to provide a constant physical sink.
Materials and Methods
Materials
General Procedure for HFM Studies
Figure 3
Figure 3. Structures of all drugs (blue) and receiver media additives (orange) used in the present study. The higher and lower strength of each receiver media additive is shown below each structure, in % w/v. An additional strength of SLS, 0.01%, which is below the critical micelle concentration, was studied. The visualization of BSA was generated using the Mol* viewer based on coordinates from the Protein Data Bank (PDB ID: 3V03). (27−29)
Preparation of Donor and Receiver Media
Quantification of Drugs
Equilibrium Solubility Measurements
HFM Setup
Flow Rate Experiments
Screening of Receiver Media
HFM Studies with Receiver Media Additives
Dynamic Light Scattering
Assessment of Back-Diffusion
Simulation of Effect of SLS Concentration on Papp
Results and Discussion
GRS Flow Rate Experiments
Figure 4
Figure 4. Top row: Wilson plot showing the scaling of overall diffusional resistance, R, with either donor-side (A) or receiver-side axial velocity (B) to the power of −n. Error bars represent SEM (n = 3), propagated through the reciprocal transformation. Bottom row: Variations in Papp with donor-side (C) and receiver-side (D) volumetric flow rate, along with the theoretical plateaus derived from the intercepts of plots A and B. Error bars represent SEM (n = 3). The shaded region of the dotted line represents the standard error of the y-intercepts. All plots were derived using concentrations sampled from the receiver vessel.
Characterization of Receiver Media Additives
Solubility
Figure 5
Figure 5. (A) Bar plot comparing the thermodynamic equilibrium solubility of GRS in various receiver media at low and high concentrations. The concentrations are available in Figure 3. The horizontal dashed line represents the buffer solubility (10.27 ± 0.14 mg L–1). B. Bar plot comparing Papp values obtained from GRS permeation studies, illustrating the influence of a solubilizing receiver. The horizontal dashed line represents the buffer Papp (3.56 ± 0.22 × 10–5 cm s–1). Error bars and shaded regions represent SEM in both cases (n = 3). Both plots were derived using data sampled from the receiver vessel.
Figure 6
Figure 6. Scatter plot showing the variation in Papp with solubility for MLX and PHT. Note that the equilibrium buffer solubility of MLX (272.68 ± 3.87 mg L–1) was higher than that of PHT (29.33 ± 0.61 mg L–1) which is relevant for the calculation of H. Only variations in Papp are shown for APY as solubility exceeded 1 g mL–1 in all media. Error bars represent SEM (n = 3)
DLS
| Additive | % w/v | Diameter (nm) (mean ± SEM, n = 3) |
|---|---|---|
| SLS | 1 | 4.82 (±0.31) |
| 2 | 3.87 (±0.30) | |
| CTAB | 1 | 6.48 (±0.02) |
| 2 | 6.13 (±0.10) | |
| PS80 | 1 | 12.05 (±0.17) |
| 2 | 12.06 (±0.21) | |
| TPGS | 0.1 | 13.08 (±0.16) |
| 0.2 | 12.41 (±0.35) | |
| P10LE | 1 | 7.97 (±0.30) |
| 2 | 8.90 (±0.45) | |
| P23LE | 1 | 10.07 (±0.17) |
| 2 | 9.03 (±0.12) |
Permeability Testing
Figure 7
Figure 7. Plot of Papp ratio versus solubility ratio for GRS. Error bars represent SEM (n = 3), with error being propagated to account for the calculation of a ratio. The shaded region on the two theoretical curves reflect the standard error of the y-intercept on the relevant Wilson plots.
Back-Diffusion
| Additive | Low Strength | High Strength |
|---|---|---|
| Surfactants | ||
| SLS | Not detected | Not detected |
| CTAB | Not detected | Detected |
| PS80 | Not detected | Not detected |
| TPGS | Not detected | Detected |
| P10LE | Not detected | Detected |
| P23LE | Not detected | Detected |
| Cyclodextrins | ||
| HPβCD | 8.24 ± 1.18% | 7.27 ± 0.07% |
| γCD | 13.01 ± 2.32% | 10.96 ± 1.24% |
| BSA | ||
| BSA | Below LOD | Below LOD |
The presence of surfactant micelles in the donor media was detected by DLS. Cyclodextrins were quantified via a phenolphthalein inclusion complexation assay. BSA was quantified by the Bradford assay.
Simulation of the Effect of SLS Concentration on Papp
Figure 8
Figure 8. Simulated effect of increasing receiver-side concentrations of SLS on Papp at four levels of HSLS 2%. Additionally shown are experimentally measured Papp values for GRS, APY, PHT, and MLX at various SLS concentrations.
Limitations
Impact on Permeability Testing
Future Directions
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.5c01771.
Phase solubility profiles for GRS in the presence of γCD and HPβCD (Figure S1); permeability and recovery data for GRS at different flow rate combinations using both donor and receiver-side data (Table S1–S2); Papp, solubility, and recovery of GRS in different media (Table S3); bar plot comparing Papp values obtained from GRS permeation studies based on donor-side sampling (Figure S2); Papp and solubility for APY, PHT, and MLX in various media (Table S4); description of SEM propagation (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
J.D.M. is funded by the Taighde Éireann – Research Ireland Government of Ireland Postgraduate Scholarship Programme Grant GOIPG/2022/1580. J.D.M. received a scholarship from the Fulbright Commission in Ireland to support research conducted at the University of Maryland School of Pharmacy.
References
This article references 58 other publications.
- 1O’Shea, J. P.; Augustijns, P.; Brandl, M.; Brayden, D. J.; Brouwers, J.; Griffin, B. T.; Holm, R.; Jacobsen, A.-C.; Lennernäs, H.; Vinarov, Z.; O’Driscoll, C. M. Best practices in current models mimicking drug permeability in the gastrointestinal tract - An UNGAP review. European Journal of Pharmaceutical Sciences 2022, 170, 106098 DOI: 10.1016/j.ejps.2021.106098Google ScholarThere is no corresponding record for this reference.
- 2Haering, B.; Seyferth, S.; Schiffter, H. A. The tangential flow absorption model (TFAM) – A novel dissolution method for evaluating the performance of amorphous solid dispersions of poorly water-soluble actives. Eur. J. Pharm. Biopharm. 2020, 154, 74– 88, DOI: 10.1016/j.ejpb.2020.06.013Google ScholarThere is no corresponding record for this reference.
- 3Repligen Corporation. Hollow Fiber Modules for Fast and Gentle TFF. 2023. https://www.repligen.com/Products/Hollow%20Fibers/DS_0144_0164_Hollow_Fiber_Datasheet_05Jan2024.pdf (accessed 2026-01-15).Google ScholarThere is no corresponding record for this reference.
- 4Dahuron, L. Designing Liquid-Liquid Extractions in Hollow Fiber Modules. Ph.D. Thesis, University of Minnesota, Minneapolis, 1987.Google ScholarThere is no corresponding record for this reference.
- 5Hate, S. S.; Reutzel-Edens, S. M.; Taylor, L. S. Absorptive Dissolution Testing of Supersaturating Systems: Impact of Absorptive Sink Conditions on Solution Phase Behavior and Mass Transport. Mol. Pharmaceutics 2017, 14, 4052– 4063, DOI: 10.1021/acs.molpharmaceut.7b00740Google ScholarThere is no corresponding record for this reference.
- 6Hate, S. S.; Reutzel-Edens, S. M.; Taylor, L. S. Insight into Amorphous Solid Dispersion Performance by Coupled Dissolution and Membrane Mass Transfer Measurements. Mol. Pharmaceutics 2019, 16, 448– 461, DOI: 10.1021/acs.molpharmaceut.8b01117Google ScholarThere is no corresponding record for this reference.
- 7Adhikari, A.; Seo, P. R.; Polli, J. E. Dissolution-Hollow Fiber Membrane (D-HFM) System to Anticipate Biopharmaceutics Risk of Tablets and Capsules. J. Pharm. Sci. 2023, 112, 751– 759, DOI: 10.1016/j.xphs.2022.09.030Google ScholarThere is no corresponding record for this reference.
- 8Patel, R. P.; Taylor, L. S.; Polli, J. E. Impact of drug incorporation into micelle on reduced griseofulvin and meloxicam permeation across a hollow fiber membrane. J. Pharm. Sci. 2025, 114, 402– 415, DOI: 10.1016/j.xphs.2024.10.017Google ScholarThere is no corresponding record for this reference.
- 9Avdeef, A. Absorption and Drug Development: Solubility, Permeability, and Charge State, 1st ed.; Wiley, 2012.Google ScholarThere is no corresponding record for this reference.
- 10Buckley, S. T.; Fischer, S. M.; Fricker, G.; Brandl, M. In vitro models to evaluate the permeability of poorly soluble drug entities: Challenges and perspectives. European Journal of Pharmaceutical Sciences 2012, 45, 235– 250, DOI: 10.1016/j.ejps.2011.12.007Google ScholarThere is no corresponding record for this reference.
- 11Koplin, S.; Kumpugdee-Vollrath, M.; Bauer-Brandl, A.; Brandl, M. Surfactants enhance recovery of poorly soluble drugs during microdialysis sampling: Implications for in vitro dissolution-/permeation-studies. J. Pharm. Biomed. Anal. 2017, 145, 586– 592, DOI: 10.1016/j.jpba.2017.07.022Google ScholarThere is no corresponding record for this reference.
- 12Hate, S. S.; Mosquera-Giraldo, L. I.; Taylor, L. S. A Mechanistic Study of Drug Mass Transport from Supersaturated Solutions Across PAMPA Membranes. J. Pharm. Sci. 2022, 111, 102– 115, DOI: 10.1016/j.xphs.2021.07.003Google ScholarThere is no corresponding record for this reference.
- 13Jacobsen, A.-C.; Krupa, A.; Brandl, M.; Bauer-Brandl, A. High-Throughput Dissolution/Permeation Screening─A 96-Well Two-Compartment Microplate Approach. Pharmaceutics 2019, 11, 227, DOI: 10.3390/pharmaceutics11050227Google ScholarThere is no corresponding record for this reference.
- 14Narula, A.; Sabra, R.; Li, N. Mechanisms and Extent of Enhanced Passive Permeation by Colloidal Drug Particles. Mol. Pharmaceutics 2022, 19, 3085– 3099, DOI: 10.1021/acs.molpharmaceut.2c00124Google ScholarThere is no corresponding record for this reference.
- 15Sitovs, A.; Mohylyuk, V. Ex vivo permeability study of poorly soluble drugs across gastrointestinal membranes: acceptor compartment media composition. Drug Discovery Today 2024, 29, 104214 DOI: 10.1016/j.drudis.2024.104214Google ScholarThere is no corresponding record for this reference.
- 16Dahan, A.; Beig, A.; Lindley, D.; Miller, J. M. The solubility–permeability interplay and oral drug formulation design: Two heads are better than one. Adv. Drug Delivery Rev. 2016, 101, 99– 107, DOI: 10.1016/j.addr.2016.04.018Google ScholarThere is no corresponding record for this reference.
- 17Higuchi, T. Pro-drug, molecular structure and percutaneous delivery. In Design of Biopharmaceutical properties through Prodrugs and Analogs: APhA: Washington DC, 1977; pp 409– 21.Google ScholarThere is no corresponding record for this reference.
- 18Yang, D. H.; Najafian, S.; Chaudhuri, B.; Li, N. The Particle Drifting Effect: A Combined Function of Colloidal and Drug Properties. Mol. Pharmaceutics 2024, 21, 5510– 5528, DOI: 10.1021/acs.molpharmaceut.4c00751Google ScholarThere is no corresponding record for this reference.
- 19Makino, K.; Ohshima, H.; Kondo, T. Kinetic model for membrane transport. III. Solute transport through an asymmetrical membrane. Biophys. Chem. 1992, 43, 89– 105, DOI: 10.1016/0301-4622(92)80045-7Google ScholarThere is no corresponding record for this reference.
- 20Yoon, K. A.; Burgess, D. J. Mathematical modelling of drug transport in emulsion systems. J. Pharm. Pharmacol. 1998, 50, 601– 610, DOI: 10.1111/j.2042-7158.1998.tb06893.xGoogle ScholarThere is no corresponding record for this reference.
- 21Borbás, E.; Sinkó, B.; Tsinman, O.; Tsinman, K.; Kiserdei, É.; Démuth, B.; Balogh, A.; Bodák, B.; Domokos, A.; Dargó, G.; Balogh, G. T.; Nagy, Z. K. Investigation and Mathematical Description of the Real Driving Force of Passive Transport of Drug Molecules from Supersaturated Solutions. Mol. Pharmaceutics 2016, 13, 3816– 3826, DOI: 10.1021/acs.molpharmaceut.6b00613Google ScholarThere is no corresponding record for this reference.
- 22Holzem, F. L.; Parrott, N.; Petrig Schaffland, J.; Brandl, M.; Bauer-Brandl, A.; Stillhart, C. Oral Absorption from Surfactant-Based Drug Formulations: The Impact of Molecularly Dissolved Drug on Bioavailability. J. Pharm. Sci. 2024, 113, 3054– 3064, DOI: 10.1016/j.xphs.2024.07.017Google ScholarThere is no corresponding record for this reference.
- 23Sugano, K. Possible reduction of effective thickness of intestinal unstirred water layer by particle drifting effect. Int. J. Pharm. 2010, 387, 103– 109, DOI: 10.1016/j.ijpharm.2009.12.014Google ScholarThere is no corresponding record for this reference.
- 24Kedem, O.; Katchalsky, A. Thermodynamic analysis of the permeability of biological membranes to non-electrolytes. Biochim. Biophys. Acta 1958, 27, 229– 246, DOI: 10.1016/0006-3002(58)90330-5Google ScholarThere is no corresponding record for this reference.
- 25Adhikari, A.; Seo, P. R.; Polli, J. E. Characterization of Dissolution-Permeation System using Hollow Fiber Membrane Module and Utility to Predict in Vivo Drug Permeation Across BCS Classes. J. Pharm. Sci. 2022, 111, 3075– 3087, DOI: 10.1016/j.xphs.2022.07.002Google ScholarThere is no corresponding record for this reference.
- 26Benet, L. Z.; Amidon, G. L.; Barends, D. M.; Lennernäs, H.; Polli, J. E.; Shah, V. P.; Stavchansky, S. A.; Yu, L. X. The Use of BDDCS in Classifying the Permeability of Marketed Drugs. Pharm. Res. 2008, 25, 483– 488, DOI: 10.1007/s11095-007-9523-xGoogle ScholarThere is no corresponding record for this reference.
- 27Majorek, K. A.; Porebski, P. J.; Dayal, A.; Zimmerman, M. D.; Jablonska, K.; Stewart, A. J.; Chruszcz, M.; Minor, W. Structural and Immunologic Characterization of Bovine, Horse, and Rabbit Serum Albumins. Mol. Immunol. 2012, 52 (3−4), 174– 182, DOI: 10.1016/j.molimm.2012.05.011Google ScholarThere is no corresponding record for this reference.
- 28Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodová, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S. K.; Koča, J.; Rose, A. S. Mol* Viewer: Modern Web App for 3D Visualization and Analysis of Large Biomolecular Structures. Nucleic Acids Res. 2021, 49 (W1), W431– W437, DOI: 10.1093/nar/gkab314Google ScholarThere is no corresponding record for this reference.
- 29Berman, H. M. The Protein Data Bank. Nucleic Acids Res. 2000, 28 (1), 235– 242, DOI: 10.1093/nar/28.1.235Google ScholarThere is no corresponding record for this reference.
- 30Goel, A.; Nene, S. N. Modifications in the Phenolphthalein Method for Spectrophotometric Estimation of Beta Cyclodextrin. Starch - St ä rke 1995, 47, 399– 400, DOI: 10.1002/star.19950471006Google ScholarThere is no corresponding record for this reference.
- 31Mäkelä, M.; Korpela, T.; Laakso, S. Colorimetric determination of β-cyclodextrin: two assay modifications based on molecular complexation of phenolphatalein. Journal of Biochemical and Biophysical Methods 1987, 14, 85– 92, DOI: 10.1016/0165-022X(87)90043-1Google ScholarThere is no corresponding record for this reference.
- 32Fuguet, E.; Ràfols, C.; Rosés, M.; Bosch, E. Critical micelle concentration of surfactants in aqueous buffered and unbuffered systems. Anal. Chim. Acta 2005, 548, 95– 100, DOI: 10.1016/j.aca.2005.05.069Google ScholarThere is no corresponding record for this reference.
- 33Kunikata, S.; Fukuda, M.; Yamamoto, K.-i; Yagi, Y.; Matsuda, M.; Sakai, K. Technical Characterization of Dialysis Fluid Flow of Newly Developed Dialyzers Using Mass Transfer Correlation Equations. ASAIO Journal 2009, 55, 231– 235, DOI: 10.1097/MAT.0b013e318198d870Google ScholarThere is no corresponding record for this reference.
- 34Yang, M.; Cussler, E. L. Designing hollow-fiber contactors. AIChE J. 1986, 32, 1910– 1916, DOI: 10.1002/aic.690321117Google ScholarThere is no corresponding record for this reference.
- 35Wickramasinghe, S.; Semmens, M. J.; Cussler, E. Mass transfer in various hollow fiber geometries. J. Membr. Sci. 1992, 69, 235– 250, DOI: 10.1016/0376-7388(92)80042-IGoogle ScholarThere is no corresponding record for this reference.
- 36D’elia, N. A.; Dahuron, L.; Cussler, E. Liquid-liquid extractions with microporous hollow fibers. J. Membr. Sci. 1986, 29, 309– 319, DOI: 10.1016/S0376-7388(00)81269-7Google ScholarThere is no corresponding record for this reference.
- 37Dahuron, L.; Cussler, E. L. Protein extractions with hollow fibers. AIChE J. 1988, 34, 130– 136, DOI: 10.1002/aic.690340115Google ScholarThere is no corresponding record for this reference.
- 38De Smidt, J. H.; Offringa, J. C.; Crommelin, D. J. Dissolution Kinetics of Griseofulvin in Sodium Dodecylsulphate Solutions. J. Pharm. Sci. 1987, 76, 711– 714, DOI: 10.1002/jps.2600760909Google ScholarThere is no corresponding record for this reference.
- 39Ng, S.-F.; Rouse, J.; Sanderson, D.; Eccleston, G. A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells. Pharmaceutics 2010, 2, 209– 223, DOI: 10.3390/pharmaceutics2020209Google ScholarThere is no corresponding record for this reference.
- 40Mohajerani, F.; Clark, W. R.; Ronco, C.; Narsimhan, V. Mass Transport in High-Flux Hemodialysis: Application of Engineering Principles to Clinical Prescription. Clinical Journal of the American Society of Nephrology 2022, 17, 749– 756, DOI: 10.2215/CJN.09410721Google ScholarThere is no corresponding record for this reference.
- 41Veiga, M. D.; Díaz, P. J.; Ahsan, F. Interactions of Griseofulvin with Cyclodextrins in Solid Binary Systems. J. Pharm. Sci. 1998, 87, 891– 900, DOI: 10.1021/js970233xGoogle ScholarThere is no corresponding record for this reference.
- 42Ding, Y.; Cui, W.; Zhang, Z.; Ma, Y.; Ding, C.; Lin, Y.; Xu, Z. Solubility and Pharmacokinetic Profile Improvement of Griseofulvin through Supercritical Carbon Dioxide-Assisted Complexation with HP-γ-Cyclodextrin. Molecules 2023, 28, 7360, DOI: 10.3390/molecules28217360Google ScholarThere is no corresponding record for this reference.
- 43Nunes, P. D.; Pinto, J. F.; Bauer-Brandl, A.; Brandl, M.; Henriques, J.; Paiva, A. M. In vitro dissolution/permeation tools for amorphous solid dispersions bioavailability forecasting I: Experimental design for PermeaLoopTM. European Journal of Pharmaceutical Sciences 2023, 188, 106512 DOI: 10.1016/j.ejps.2023.106512Google ScholarThere is no corresponding record for this reference.
- 44Metry, M.; Polli, J. E. Evaluation of Excipient Risk in BCS Class I and III Biowaivers. AAPS Journal 2022, 24, 20, DOI: 10.1208/s12248-021-00670-1Google ScholarThere is no corresponding record for this reference.
- 45Yaqoob, T.; Ahsan, M.; Hussain, A.; Ahmad, I. Computational Fluid Dynamics (CFD) Modeling and Simulation of Flow Regulatory Mechanism in Artificial Kidney Using Finite Element Method. Membranes 2020, 10, 139, DOI: 10.3390/membranes10070139Google ScholarThere is no corresponding record for this reference.
- 46Pharmacopoeia, E. Recommendations on Methods for Dosage Forms Testing, European Pharmacopoeia; Druckerei CH Beck: Nördlingen, Germany, 2010; pp 665– 667.Google ScholarThere is no corresponding record for this reference.
- 47United States Pharmacopeia Convention. USP General Chapter ⟨1092⟩: The Dissolution Procedure: Development and Validation , 2023.Google ScholarThere is no corresponding record for this reference.
- 48Gao, Z. In Vitro Dissolution Testing with Flow-Through Method: A Technical Note. AAPS PharmSciTech 2009, 10, 1401, DOI: 10.1208/s12249-009-9339-6Google ScholarThere is no corresponding record for this reference.
- 49Avdeef, A.; Kansy, M. Trends in PhysChem Properties of Newly Approved Drugs over the Last Six Years; Predicting Solubility of Drugs Approved in 2021. J. Solution Chem. 2022, 51, 1455– 1481, DOI: 10.1007/s10953-022-01199-3Google ScholarThere is no corresponding record for this reference.
- 50Stegemann, S.; Moreton, C.; Svanbäck, S.; Box, K.; Motte, G.; Paudel, A. Trends in oral small-molecule drug discovery and product development based on product launches before and after the Rule of Five. Drug Discovery Today 2023, 28, 103344 DOI: 10.1016/j.drudis.2022.103344Google ScholarThere is no corresponding record for this reference.
- 51Kataoka, M.; Masaoka, Y.; Yamazaki, Y.; Sakane, T.; Sezaki, H.; Yamashita, S. In Vitro System to Evaluate Oral Absorption of Poorly Water-Soluble Drugs: Simultaneous Analysis on Dissolution and Permeation of Drugs. Pharm. Res. 2003, 20, 1674– 1680, DOI: 10.1023/A:1026107906191Google ScholarThere is no corresponding record for this reference.
- 52Avdeef, A.; Artursson, P.; Neuhoff, S.; Lazorova, L.; Gråsjö, J.; Tavelin, S. Caco-2 permeability of weakly basic drugs predicted with the Double-Sink PAMPA method. European Journal of Pharmaceutical Sciences 2005, 24, 333– 349, DOI: 10.1016/j.ejps.2004.11.011Google ScholarThere is no corresponding record for this reference.
- 53Youdim, K. A.; Avdeef, A.; Abbott, N. In vitro trans-monolayer permeability calculations: often forgotten assumptions. Drug Discovery Today 2003, 8, 997– 1003, DOI: 10.1016/S1359-6446(03)02873-3Google ScholarThere is no corresponding record for this reference.
- 54Saha, P.; Kou, J. H. Effect of bovine serum albumin on drug permeability estimation across Caco-2 monolayers. Eur. J. Pharm. Biopharm. 2002, 54, 319– 324, DOI: 10.1016/S0939-6411(02)00089-9Google ScholarThere is no corresponding record for this reference.
- 55Macheras, P.; Chryssafidis, P. Revising Pharmacokinetics of Oral Drug Absorption: I Models Based on Biopharmaceutical/Physiological and Finite Absorption Time Concepts. Pharm. Res. 2020, 37, 187, DOI: 10.1007/s11095-020-02935-4Google ScholarThere is no corresponding record for this reference.
- 56Guo, Y.; Sun, C. C. Pharmaceutical Lauryl Sulfate Salts: Prevalence, Formation Rules, and Formulation Implications. Mol. Pharmaceutics 2022, 19, 432– 439, DOI: 10.1021/acs.molpharmaceut.1c00690Google ScholarThere is no corresponding record for this reference.
- 57Rege, B. D.; Yu, L. X.; Hussain, A. S.; Polli, J. E. Effect of Common Excipients on Caco-2 Transport of Low-Permeability Drugs. J. Pharm. Sci. 2001, 90, 1776– 1786, DOI: 10.1002/jps.1127Google ScholarThere is no corresponding record for this reference.
- 58Raines, K. Drug Dissolution in Oral Drug Absorption: Workshop Report. AAPS Journal 2023, 25, 103, DOI: 10.1208/s12248-023-00865-8Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
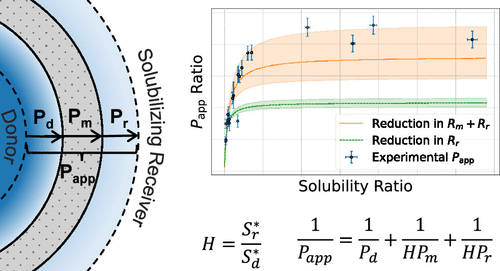
Figure 1

Figure 1. Overview of modeling workflow.
Figure 2
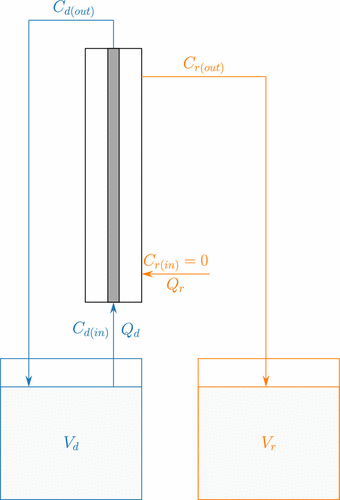
Figure 2. Diagram of the HFM setup showing the volumetric flow rate (Q), concentrations (C), and volumes (V) associated with the donor (blue) and receiver (orange) circuits. The donor circuit is recycled, while the receiver circuit is single-pass to provide a constant physical sink.
Figure 3

Figure 3. Structures of all drugs (blue) and receiver media additives (orange) used in the present study. The higher and lower strength of each receiver media additive is shown below each structure, in % w/v. An additional strength of SLS, 0.01%, which is below the critical micelle concentration, was studied. The visualization of BSA was generated using the Mol* viewer based on coordinates from the Protein Data Bank (PDB ID: 3V03). (27−29)
Figure 4

Figure 4. Top row: Wilson plot showing the scaling of overall diffusional resistance, R, with either donor-side (A) or receiver-side axial velocity (B) to the power of −n. Error bars represent SEM (n = 3), propagated through the reciprocal transformation. Bottom row: Variations in Papp with donor-side (C) and receiver-side (D) volumetric flow rate, along with the theoretical plateaus derived from the intercepts of plots A and B. Error bars represent SEM (n = 3). The shaded region of the dotted line represents the standard error of the y-intercepts. All plots were derived using concentrations sampled from the receiver vessel.
Figure 5
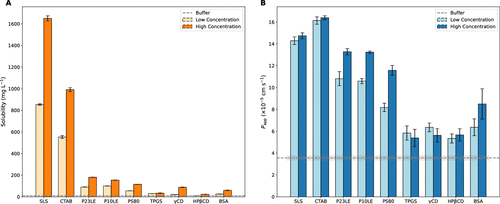
Figure 5. (A) Bar plot comparing the thermodynamic equilibrium solubility of GRS in various receiver media at low and high concentrations. The concentrations are available in Figure 3. The horizontal dashed line represents the buffer solubility (10.27 ± 0.14 mg L–1). B. Bar plot comparing Papp values obtained from GRS permeation studies, illustrating the influence of a solubilizing receiver. The horizontal dashed line represents the buffer Papp (3.56 ± 0.22 × 10–5 cm s–1). Error bars and shaded regions represent SEM in both cases (n = 3). Both plots were derived using data sampled from the receiver vessel.
Figure 6
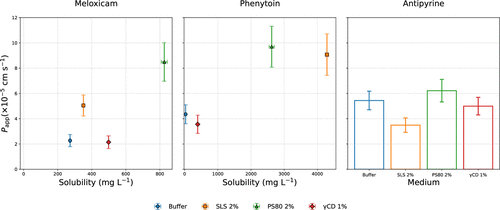
Figure 6. Scatter plot showing the variation in Papp with solubility for MLX and PHT. Note that the equilibrium buffer solubility of MLX (272.68 ± 3.87 mg L–1) was higher than that of PHT (29.33 ± 0.61 mg L–1) which is relevant for the calculation of H. Only variations in Papp are shown for APY as solubility exceeded 1 g mL–1 in all media. Error bars represent SEM (n = 3)
Figure 7
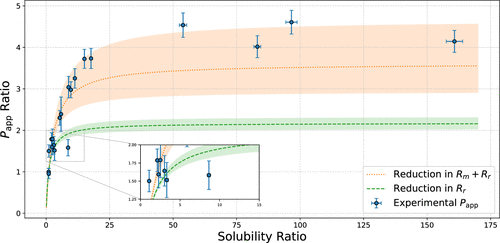
Figure 7. Plot of Papp ratio versus solubility ratio for GRS. Error bars represent SEM (n = 3), with error being propagated to account for the calculation of a ratio. The shaded region on the two theoretical curves reflect the standard error of the y-intercept on the relevant Wilson plots.
Figure 8

Figure 8. Simulated effect of increasing receiver-side concentrations of SLS on Papp at four levels of HSLS 2%. Additionally shown are experimentally measured Papp values for GRS, APY, PHT, and MLX at various SLS concentrations.
References
This article references 58 other publications.
- 1O’Shea, J. P.; Augustijns, P.; Brandl, M.; Brayden, D. J.; Brouwers, J.; Griffin, B. T.; Holm, R.; Jacobsen, A.-C.; Lennernäs, H.; Vinarov, Z.; O’Driscoll, C. M. Best practices in current models mimicking drug permeability in the gastrointestinal tract - An UNGAP review. European Journal of Pharmaceutical Sciences 2022, 170, 106098 DOI: 10.1016/j.ejps.2021.106098There is no corresponding record for this reference.
- 2Haering, B.; Seyferth, S.; Schiffter, H. A. The tangential flow absorption model (TFAM) – A novel dissolution method for evaluating the performance of amorphous solid dispersions of poorly water-soluble actives. Eur. J. Pharm. Biopharm. 2020, 154, 74– 88, DOI: 10.1016/j.ejpb.2020.06.013There is no corresponding record for this reference.
- 3Repligen Corporation. Hollow Fiber Modules for Fast and Gentle TFF. 2023. https://www.repligen.com/Products/Hollow%20Fibers/DS_0144_0164_Hollow_Fiber_Datasheet_05Jan2024.pdf (accessed 2026-01-15).There is no corresponding record for this reference.
- 4Dahuron, L. Designing Liquid-Liquid Extractions in Hollow Fiber Modules. Ph.D. Thesis, University of Minnesota, Minneapolis, 1987.There is no corresponding record for this reference.
- 5Hate, S. S.; Reutzel-Edens, S. M.; Taylor, L. S. Absorptive Dissolution Testing of Supersaturating Systems: Impact of Absorptive Sink Conditions on Solution Phase Behavior and Mass Transport. Mol. Pharmaceutics 2017, 14, 4052– 4063, DOI: 10.1021/acs.molpharmaceut.7b00740There is no corresponding record for this reference.
- 6Hate, S. S.; Reutzel-Edens, S. M.; Taylor, L. S. Insight into Amorphous Solid Dispersion Performance by Coupled Dissolution and Membrane Mass Transfer Measurements. Mol. Pharmaceutics 2019, 16, 448– 461, DOI: 10.1021/acs.molpharmaceut.8b01117There is no corresponding record for this reference.
- 7Adhikari, A.; Seo, P. R.; Polli, J. E. Dissolution-Hollow Fiber Membrane (D-HFM) System to Anticipate Biopharmaceutics Risk of Tablets and Capsules. J. Pharm. Sci. 2023, 112, 751– 759, DOI: 10.1016/j.xphs.2022.09.030There is no corresponding record for this reference.
- 8Patel, R. P.; Taylor, L. S.; Polli, J. E. Impact of drug incorporation into micelle on reduced griseofulvin and meloxicam permeation across a hollow fiber membrane. J. Pharm. Sci. 2025, 114, 402– 415, DOI: 10.1016/j.xphs.2024.10.017There is no corresponding record for this reference.
- 9Avdeef, A. Absorption and Drug Development: Solubility, Permeability, and Charge State, 1st ed.; Wiley, 2012.There is no corresponding record for this reference.
- 10Buckley, S. T.; Fischer, S. M.; Fricker, G.; Brandl, M. In vitro models to evaluate the permeability of poorly soluble drug entities: Challenges and perspectives. European Journal of Pharmaceutical Sciences 2012, 45, 235– 250, DOI: 10.1016/j.ejps.2011.12.007There is no corresponding record for this reference.
- 11Koplin, S.; Kumpugdee-Vollrath, M.; Bauer-Brandl, A.; Brandl, M. Surfactants enhance recovery of poorly soluble drugs during microdialysis sampling: Implications for in vitro dissolution-/permeation-studies. J. Pharm. Biomed. Anal. 2017, 145, 586– 592, DOI: 10.1016/j.jpba.2017.07.022There is no corresponding record for this reference.
- 12Hate, S. S.; Mosquera-Giraldo, L. I.; Taylor, L. S. A Mechanistic Study of Drug Mass Transport from Supersaturated Solutions Across PAMPA Membranes. J. Pharm. Sci. 2022, 111, 102– 115, DOI: 10.1016/j.xphs.2021.07.003There is no corresponding record for this reference.
- 13Jacobsen, A.-C.; Krupa, A.; Brandl, M.; Bauer-Brandl, A. High-Throughput Dissolution/Permeation Screening─A 96-Well Two-Compartment Microplate Approach. Pharmaceutics 2019, 11, 227, DOI: 10.3390/pharmaceutics11050227There is no corresponding record for this reference.
- 14Narula, A.; Sabra, R.; Li, N. Mechanisms and Extent of Enhanced Passive Permeation by Colloidal Drug Particles. Mol. Pharmaceutics 2022, 19, 3085– 3099, DOI: 10.1021/acs.molpharmaceut.2c00124There is no corresponding record for this reference.
- 15Sitovs, A.; Mohylyuk, V. Ex vivo permeability study of poorly soluble drugs across gastrointestinal membranes: acceptor compartment media composition. Drug Discovery Today 2024, 29, 104214 DOI: 10.1016/j.drudis.2024.104214There is no corresponding record for this reference.
- 16Dahan, A.; Beig, A.; Lindley, D.; Miller, J. M. The solubility–permeability interplay and oral drug formulation design: Two heads are better than one. Adv. Drug Delivery Rev. 2016, 101, 99– 107, DOI: 10.1016/j.addr.2016.04.018There is no corresponding record for this reference.
- 17Higuchi, T. Pro-drug, molecular structure and percutaneous delivery. In Design of Biopharmaceutical properties through Prodrugs and Analogs: APhA: Washington DC, 1977; pp 409– 21.There is no corresponding record for this reference.
- 18Yang, D. H.; Najafian, S.; Chaudhuri, B.; Li, N. The Particle Drifting Effect: A Combined Function of Colloidal and Drug Properties. Mol. Pharmaceutics 2024, 21, 5510– 5528, DOI: 10.1021/acs.molpharmaceut.4c00751There is no corresponding record for this reference.
- 19Makino, K.; Ohshima, H.; Kondo, T. Kinetic model for membrane transport. III. Solute transport through an asymmetrical membrane. Biophys. Chem. 1992, 43, 89– 105, DOI: 10.1016/0301-4622(92)80045-7There is no corresponding record for this reference.
- 20Yoon, K. A.; Burgess, D. J. Mathematical modelling of drug transport in emulsion systems. J. Pharm. Pharmacol. 1998, 50, 601– 610, DOI: 10.1111/j.2042-7158.1998.tb06893.xThere is no corresponding record for this reference.
- 21Borbás, E.; Sinkó, B.; Tsinman, O.; Tsinman, K.; Kiserdei, É.; Démuth, B.; Balogh, A.; Bodák, B.; Domokos, A.; Dargó, G.; Balogh, G. T.; Nagy, Z. K. Investigation and Mathematical Description of the Real Driving Force of Passive Transport of Drug Molecules from Supersaturated Solutions. Mol. Pharmaceutics 2016, 13, 3816– 3826, DOI: 10.1021/acs.molpharmaceut.6b00613There is no corresponding record for this reference.
- 22Holzem, F. L.; Parrott, N.; Petrig Schaffland, J.; Brandl, M.; Bauer-Brandl, A.; Stillhart, C. Oral Absorption from Surfactant-Based Drug Formulations: The Impact of Molecularly Dissolved Drug on Bioavailability. J. Pharm. Sci. 2024, 113, 3054– 3064, DOI: 10.1016/j.xphs.2024.07.017There is no corresponding record for this reference.
- 23Sugano, K. Possible reduction of effective thickness of intestinal unstirred water layer by particle drifting effect. Int. J. Pharm. 2010, 387, 103– 109, DOI: 10.1016/j.ijpharm.2009.12.014There is no corresponding record for this reference.
- 24Kedem, O.; Katchalsky, A. Thermodynamic analysis of the permeability of biological membranes to non-electrolytes. Biochim. Biophys. Acta 1958, 27, 229– 246, DOI: 10.1016/0006-3002(58)90330-5There is no corresponding record for this reference.
- 25Adhikari, A.; Seo, P. R.; Polli, J. E. Characterization of Dissolution-Permeation System using Hollow Fiber Membrane Module and Utility to Predict in Vivo Drug Permeation Across BCS Classes. J. Pharm. Sci. 2022, 111, 3075– 3087, DOI: 10.1016/j.xphs.2022.07.002There is no corresponding record for this reference.
- 26Benet, L. Z.; Amidon, G. L.; Barends, D. M.; Lennernäs, H.; Polli, J. E.; Shah, V. P.; Stavchansky, S. A.; Yu, L. X. The Use of BDDCS in Classifying the Permeability of Marketed Drugs. Pharm. Res. 2008, 25, 483– 488, DOI: 10.1007/s11095-007-9523-xThere is no corresponding record for this reference.
- 27Majorek, K. A.; Porebski, P. J.; Dayal, A.; Zimmerman, M. D.; Jablonska, K.; Stewart, A. J.; Chruszcz, M.; Minor, W. Structural and Immunologic Characterization of Bovine, Horse, and Rabbit Serum Albumins. Mol. Immunol. 2012, 52 (3−4), 174– 182, DOI: 10.1016/j.molimm.2012.05.011There is no corresponding record for this reference.
- 28Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodová, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S. K.; Koča, J.; Rose, A. S. Mol* Viewer: Modern Web App for 3D Visualization and Analysis of Large Biomolecular Structures. Nucleic Acids Res. 2021, 49 (W1), W431– W437, DOI: 10.1093/nar/gkab314There is no corresponding record for this reference.
- 29Berman, H. M. The Protein Data Bank. Nucleic Acids Res. 2000, 28 (1), 235– 242, DOI: 10.1093/nar/28.1.235There is no corresponding record for this reference.
- 30Goel, A.; Nene, S. N. Modifications in the Phenolphthalein Method for Spectrophotometric Estimation of Beta Cyclodextrin. Starch - St ä rke 1995, 47, 399– 400, DOI: 10.1002/star.19950471006There is no corresponding record for this reference.
- 31Mäkelä, M.; Korpela, T.; Laakso, S. Colorimetric determination of β-cyclodextrin: two assay modifications based on molecular complexation of phenolphatalein. Journal of Biochemical and Biophysical Methods 1987, 14, 85– 92, DOI: 10.1016/0165-022X(87)90043-1There is no corresponding record for this reference.
- 32Fuguet, E.; Ràfols, C.; Rosés, M.; Bosch, E. Critical micelle concentration of surfactants in aqueous buffered and unbuffered systems. Anal. Chim. Acta 2005, 548, 95– 100, DOI: 10.1016/j.aca.2005.05.069There is no corresponding record for this reference.
- 33Kunikata, S.; Fukuda, M.; Yamamoto, K.-i; Yagi, Y.; Matsuda, M.; Sakai, K. Technical Characterization of Dialysis Fluid Flow of Newly Developed Dialyzers Using Mass Transfer Correlation Equations. ASAIO Journal 2009, 55, 231– 235, DOI: 10.1097/MAT.0b013e318198d870There is no corresponding record for this reference.
- 34Yang, M.; Cussler, E. L. Designing hollow-fiber contactors. AIChE J. 1986, 32, 1910– 1916, DOI: 10.1002/aic.690321117There is no corresponding record for this reference.
- 35Wickramasinghe, S.; Semmens, M. J.; Cussler, E. Mass transfer in various hollow fiber geometries. J. Membr. Sci. 1992, 69, 235– 250, DOI: 10.1016/0376-7388(92)80042-IThere is no corresponding record for this reference.
- 36D’elia, N. A.; Dahuron, L.; Cussler, E. Liquid-liquid extractions with microporous hollow fibers. J. Membr. Sci. 1986, 29, 309– 319, DOI: 10.1016/S0376-7388(00)81269-7There is no corresponding record for this reference.
- 37Dahuron, L.; Cussler, E. L. Protein extractions with hollow fibers. AIChE J. 1988, 34, 130– 136, DOI: 10.1002/aic.690340115There is no corresponding record for this reference.
- 38De Smidt, J. H.; Offringa, J. C.; Crommelin, D. J. Dissolution Kinetics of Griseofulvin in Sodium Dodecylsulphate Solutions. J. Pharm. Sci. 1987, 76, 711– 714, DOI: 10.1002/jps.2600760909There is no corresponding record for this reference.
- 39Ng, S.-F.; Rouse, J.; Sanderson, D.; Eccleston, G. A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells. Pharmaceutics 2010, 2, 209– 223, DOI: 10.3390/pharmaceutics2020209There is no corresponding record for this reference.
- 40Mohajerani, F.; Clark, W. R.; Ronco, C.; Narsimhan, V. Mass Transport in High-Flux Hemodialysis: Application of Engineering Principles to Clinical Prescription. Clinical Journal of the American Society of Nephrology 2022, 17, 749– 756, DOI: 10.2215/CJN.09410721There is no corresponding record for this reference.
- 41Veiga, M. D.; Díaz, P. J.; Ahsan, F. Interactions of Griseofulvin with Cyclodextrins in Solid Binary Systems. J. Pharm. Sci. 1998, 87, 891– 900, DOI: 10.1021/js970233xThere is no corresponding record for this reference.
- 42Ding, Y.; Cui, W.; Zhang, Z.; Ma, Y.; Ding, C.; Lin, Y.; Xu, Z. Solubility and Pharmacokinetic Profile Improvement of Griseofulvin through Supercritical Carbon Dioxide-Assisted Complexation with HP-γ-Cyclodextrin. Molecules 2023, 28, 7360, DOI: 10.3390/molecules28217360There is no corresponding record for this reference.
- 43Nunes, P. D.; Pinto, J. F.; Bauer-Brandl, A.; Brandl, M.; Henriques, J.; Paiva, A. M. In vitro dissolution/permeation tools for amorphous solid dispersions bioavailability forecasting I: Experimental design for PermeaLoopTM. European Journal of Pharmaceutical Sciences 2023, 188, 106512 DOI: 10.1016/j.ejps.2023.106512There is no corresponding record for this reference.
- 44Metry, M.; Polli, J. E. Evaluation of Excipient Risk in BCS Class I and III Biowaivers. AAPS Journal 2022, 24, 20, DOI: 10.1208/s12248-021-00670-1There is no corresponding record for this reference.
- 45Yaqoob, T.; Ahsan, M.; Hussain, A.; Ahmad, I. Computational Fluid Dynamics (CFD) Modeling and Simulation of Flow Regulatory Mechanism in Artificial Kidney Using Finite Element Method. Membranes 2020, 10, 139, DOI: 10.3390/membranes10070139There is no corresponding record for this reference.
- 46Pharmacopoeia, E. Recommendations on Methods for Dosage Forms Testing, European Pharmacopoeia; Druckerei CH Beck: Nördlingen, Germany, 2010; pp 665– 667.There is no corresponding record for this reference.
- 47United States Pharmacopeia Convention. USP General Chapter ⟨1092⟩: The Dissolution Procedure: Development and Validation , 2023.There is no corresponding record for this reference.
- 48Gao, Z. In Vitro Dissolution Testing with Flow-Through Method: A Technical Note. AAPS PharmSciTech 2009, 10, 1401, DOI: 10.1208/s12249-009-9339-6There is no corresponding record for this reference.
- 49Avdeef, A.; Kansy, M. Trends in PhysChem Properties of Newly Approved Drugs over the Last Six Years; Predicting Solubility of Drugs Approved in 2021. J. Solution Chem. 2022, 51, 1455– 1481, DOI: 10.1007/s10953-022-01199-3There is no corresponding record for this reference.
- 50Stegemann, S.; Moreton, C.; Svanbäck, S.; Box, K.; Motte, G.; Paudel, A. Trends in oral small-molecule drug discovery and product development based on product launches before and after the Rule of Five. Drug Discovery Today 2023, 28, 103344 DOI: 10.1016/j.drudis.2022.103344There is no corresponding record for this reference.
- 51Kataoka, M.; Masaoka, Y.; Yamazaki, Y.; Sakane, T.; Sezaki, H.; Yamashita, S. In Vitro System to Evaluate Oral Absorption of Poorly Water-Soluble Drugs: Simultaneous Analysis on Dissolution and Permeation of Drugs. Pharm. Res. 2003, 20, 1674– 1680, DOI: 10.1023/A:1026107906191There is no corresponding record for this reference.
- 52Avdeef, A.; Artursson, P.; Neuhoff, S.; Lazorova, L.; Gråsjö, J.; Tavelin, S. Caco-2 permeability of weakly basic drugs predicted with the Double-Sink PAMPA method. European Journal of Pharmaceutical Sciences 2005, 24, 333– 349, DOI: 10.1016/j.ejps.2004.11.011There is no corresponding record for this reference.
- 53Youdim, K. A.; Avdeef, A.; Abbott, N. In vitro trans-monolayer permeability calculations: often forgotten assumptions. Drug Discovery Today 2003, 8, 997– 1003, DOI: 10.1016/S1359-6446(03)02873-3There is no corresponding record for this reference.
- 54Saha, P.; Kou, J. H. Effect of bovine serum albumin on drug permeability estimation across Caco-2 monolayers. Eur. J. Pharm. Biopharm. 2002, 54, 319– 324, DOI: 10.1016/S0939-6411(02)00089-9There is no corresponding record for this reference.
- 55Macheras, P.; Chryssafidis, P. Revising Pharmacokinetics of Oral Drug Absorption: I Models Based on Biopharmaceutical/Physiological and Finite Absorption Time Concepts. Pharm. Res. 2020, 37, 187, DOI: 10.1007/s11095-020-02935-4There is no corresponding record for this reference.
- 56Guo, Y.; Sun, C. C. Pharmaceutical Lauryl Sulfate Salts: Prevalence, Formation Rules, and Formulation Implications. Mol. Pharmaceutics 2022, 19, 432– 439, DOI: 10.1021/acs.molpharmaceut.1c00690There is no corresponding record for this reference.
- 57Rege, B. D.; Yu, L. X.; Hussain, A. S.; Polli, J. E. Effect of Common Excipients on Caco-2 Transport of Low-Permeability Drugs. J. Pharm. Sci. 2001, 90, 1776– 1786, DOI: 10.1002/jps.1127There is no corresponding record for this reference.
- 58Raines, K. Drug Dissolution in Oral Drug Absorption: Workshop Report. AAPS Journal 2023, 25, 103, DOI: 10.1208/s12248-023-00865-8There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.molpharmaceut.5c01771.
Phase solubility profiles for GRS in the presence of γCD and HPβCD (Figure S1); permeability and recovery data for GRS at different flow rate combinations using both donor and receiver-side data (Table S1–S2); Papp, solubility, and recovery of GRS in different media (Table S3); bar plot comparing Papp values obtained from GRS permeation studies based on donor-side sampling (Figure S2); Papp and solubility for APY, PHT, and MLX in various media (Table S4); description of SEM propagation (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



