
Emergent Properties When Molecules Meet the Electromagnetic Vacuum FieldClick to copy article linkArticle link copied!
- Subha BiswasSubha BiswasDepartment of Inorganic and Physical Chemistry, Indian Institute of Science, Bengaluru 560012, IndiaMore by Subha Biswas
- Anoop Thomas*Anoop Thomas*Email: [email protected]Department of Inorganic and Physical Chemistry, Indian Institute of Science, Bengaluru 560012, IndiaMore by Anoop Thomas
Abstract
The strong coupling of molecular transition dipoles to the electromagnetic vacuum field unlocks unique possibilities in chemistry and materials science. The half-light, half-matter characteristics of the polaritonic states have been effective in promoting transport, modifying molecular photophysics, driving chemical reactions, and assembling molecules. However, the impact of electronic and vibrational strong couplings on the intermolecular interactions is rarely discussed. Here, we briefly overview energy- and charge-transport properties modified under electronic and vibrational strong coupling conditions. We discuss how strong coupling can induce changes to molecular assemblies and affect the photophysics of molecular systems. Further, we provide a perspective on the cavity-altered molecular properties by connecting them to cavity-modified molecular interactions.
This publication is licensed for personal use by The American Chemical Society.
1. Introduction
2. Formation of Polaritonic States under Light–Matter Strong Coupling
Figure 1
Figure 1. (A) Two-level atomic system with quantized energy levels emitting a photon into a continuum of vacuum states. (B) Schematic illustration of a Fabry–Pérot cavity with its resonances. (C) FT-IR transmission spectrum showing the resonances of a Fabry–Perot cavity of 8 μm path length filled with air. Schematic representation of (D) the energy exchange between a lossless cavity and a molecule resulting in the Rabi oscillation and (E) the resulting strong coupling and formation of the light–matter hybrid states in a cavity. (F) Angle-dependent dispersion of the polaritonic states with respect to the in-plane wave vector (black diamonds) overlaid on the simulated dispersion contour of the uncoupled cavity mode (ℏωc = 698 cm–1) and the vibrational transition of PS (ℏωm = 698 cm–1). Adapted from ref (34). Copyright 2024 American Chemical Society.
3. Energy Transport under Strong Coupling
Figure 2
Figure 2. (A) Schematic representation of strong coupling of the electronic transitions of the donor and acceptor, together with a cavity resonance, resulting in three polaritonic states: the upper polariton (UP), middle polariton (MP), and lower polariton (LP). Reproduced from ref (30). Copyright 2016 John Wiley and Sons. (B) Excited-state dynamics of MP and LP. Adapted from ref (30). Copyright 2016 John Wiley and Sons. (C) Schematic representation of a cavity containing spatially separated donors and acceptors. The spatial separation corresponds to the thickness (h) of the polymer layer. Reproduced from ref (31). Copyright 2017 John Wiley and Sons. (D) Lifetime of MP as a function of the spacer thickness in the strong coupling conditions. Adapted from ref (31). Copyright 2017 John Wiley and Sons. (E) Momentum-resolved ultrafast imaging of pure exciton transport (top panel) and polariton transport (bottom panel). Reproduced from ref (48). Available under a CC-BY 4.0 license. Copyright 2023 Xu et al. (F) Time-resolved dynamics of the variance of polariton cloud expansion measured at different photonic fractions showing the ballistic and diffusive energy transport mediated by polaritons. Adapted from ref (47). Copyright 2023 Springer Nature.
4. Charge Transport under Strong Coupling
Figure 3
Figure 3. (A) Schematic representation of the ionic conductivity under VSC of the O–H stretching vibrational mode of water. Adapted from ref (50). Copyright 2022 American Chemical Society. (B) Emission intensity ratio IRO–/IROH, as a function of the different cavity tunings. The blue spheres indicate the emission ratio in different cavities with resonances at the wavenumbers noted on the x axis. The purple dotted curve shows absorption of the PVA −OH stretching mode. The black dashed line indicates the average IRO–/IROH observed in noncavities. Adapted from ref (43). Copyright 2025 John Wiley and Sons. (C) I–V plot of the PDI2EH-CN2 deposited on hexagonal hole arrays with different periods of holes. Reproduced from ref (10). Copyright 2015 Springer Nature. (D) Schematic representation of a mirrorless MOSFET Fabry–Pérot cavity. The blue curve shows the cavity mode interacting with the active layer. (E) Electron mobility as a function of the cavity tuning. The first- and second-mode cavities are prepared by tuning the thickness of the organic semiconductor. The bottom panel shows the cavity mode position with respect to the electronic transition of the semiconductor (D and E) Adapted from ref (53). Copyright 2021 John Wiley and Sons.
Figure 4
Figure 4. (A) FT-IR transmission spectra of cavities strongly coupled to the δ(Ar. CH) modes of PS when mixed with different weight percents of PS-d8. (B) Schematic illustration of VSC resulting in polaritonic states and the electrical conductance measurement setup. (C) Variation in conductance as a function of the strong coupling strength. (D) Action spectra of the electrical conductivity of aromatic polymers (PS and PS-d8) and nonaromatic polymer (PMMA) inside the cavity. The cavity modes are tuned with respect to the different vibrations of the polymers. The red, blue, and pink spheres correspond to the current densities of PS, PS-d8, and PMMA, respectively. Adapted from ref (34). Copyright 2024 American Chemical Society.
5. Ground-State Intermolecular Interactions under Strong Coupling
Figure 5
Figure 5. (A) Schematic representation of a Fabry–Pérot cavity containing PPE dissolved in dichlorobenzene. The cooperative VSC transforms the morphology of the polymer from fiber to sheets. Adapted from ref (58). Copyright 2021 John Wiley and Sons. (B) Supramolecular polymerization of chiral zinc porphyrin under on- and off-resonance conditions probed through electronic circular dichroism (ECD). The ECD signals of these cavities are plotted with respect to different temperatures as a function of the concentration. The elongation temperature of S–Zn is denoted by the yellow dashed line. Adapted from ref (59). Available under a CC-BY 4.0 license. Copyright 2025 Joseph et al. (C) Schematic illustration showing transformation of the naphthalenediimide supramolecular polymer in response to the click reaction under VSC and noncavity situations and their atomic force microscopy images. Adapted from ref (60). Available under a CC-BY 4.0 license. Copyright 2025 Imai et al.
Figure 6
Figure 6. (A–C) Transmittance, emission (λex = 450 nm), and excitation (for λem = 770 nm) spectra of cavities ranging from weak (purple curves) to strong (red curves) coupling of the Ce6T Q band. (D and E) Transmittance, emission, and excitation (for λem = 770 nm) spectra of the Ce6T thin film and cavity strongly coupled to the Soret band. (F) Schematic illustration of the excimer-like exciton formation between two interacting Ce6T molecules in a noncavity film upon spin coating (top panel). The bottom panel shows the Ce6T molecules with a suppressed intermolecular interaction under ESC. Reproduced from ref (42). Available under a CC BY-NC-ND 4.0 license. Copyright 2025 Biswas et al.
6. Conclusions and Outlook
Acknowledgments
S.B. thanks DST-INSPIRE and the Prime Minister’s Research Fellowship for a Ph.D. fellowship. A.T. is thankful for a IISc start-up grant and a SERB-CRG grant (CRG/2021/002396) for funding. We thank Harsh Baliyan and Kavya S. Mony for their discussion and inputs.
References
This article references 70 other publications.
- 1Fox, M. Quantum Optics: An Introduction; Oxford University Press: Oxford, NY, 2006.Google ScholarThere is no corresponding record for this reference.
- 2Haroche, S.; Kleppner, D. Cavity Quantum Electrodynamics. Phys. Today 1989, 42 (1), 24– 30, DOI: 10.1063/1.881201Google ScholarThere is no corresponding record for this reference.
- 3Ebbesen, T. W. Hybrid Light–Matter States in a Molecular and Material Science Perspective. Acc. Chem. Res. 2016, 49 (11), 2403– 2412, DOI: 10.1021/acs.accounts.6b00295Google ScholarThere is no corresponding record for this reference.
- 4Garcia-Vidal, F. J.; Ciuti, C.; Ebbesen, T. W. Manipulating Matter by Strong Coupling to Vacuum Fields. Science 2021, 373, eabd0336 DOI: 10.1126/science.abd0336Google ScholarThere is no corresponding record for this reference.
- 5Kaluzny, Y.; Goy, P.; Gross, M.; Raimond, J. M.; Haroche, S. Observation of Self-Induced Rabi Oscillations in Two-Level Atoms Excited Inside a Resonant Cavity: The Ringing Regime of Superradiance. Phys. Rev. Lett. 1983, 51 (13), 1175– 1178, DOI: 10.1103/PhysRevLett.51.1175Google ScholarThere is no corresponding record for this reference.
- 6Weisbuch, C.; Nishioka, M.; Ishikawa, A.; Arakawa, Y. Observation of the Coupled Exciton-Photon Mode Splitting in a Semiconductor Quantum Microcavity. Phys. Rev. Lett. 1992, 69 (23), 3314– 3317, DOI: 10.1103/PhysRevLett.69.3314Google ScholarThere is no corresponding record for this reference.
- 7Pockrand, I.; Brillante, A.; Möbius, D. Exciton–Surface Plasmon Coupling: An Experimental Investigation. J. Chem. Phys. 1982, 77 (12), 6289– 6295, DOI: 10.1063/1.443834Google ScholarThere is no corresponding record for this reference.
- 8Lidzey, D. G.; Bradley, D. D. C.; Skolnick, M. S.; Virgili, T.; Walker, S.; Whittaker, D. M. Strong Exciton–Photon Coupling in an Organic Semiconductor Microcavity. Nature 1998, 395 (6697), 53– 55, DOI: 10.1038/25692Google ScholarThere is no corresponding record for this reference.
- 9Bellessa, J.; Bonnand, C.; Plenet, J. C.; Mugnier, J. Strong Coupling between Surface Plasmons and Excitons in an Organic Semiconductor. Phys. Rev. Lett. 2004, 93 (3), 036404 DOI: 10.1103/PhysRevLett.93.036404Google ScholarThere is no corresponding record for this reference.
- 10Orgiu, E.; George, J.; Hutchison, J. A.; Devaux, E.; Dayen, J. F.; Doudin, B.; Stellacci, F.; Genet, C.; Schachenmayer, J.; Genes, C.; Pupillo, G.; Samorì, P.; Ebbesen, T. W. Conductivity in Organic Semiconductors Hybridized with the Vacuum Field. Nat. Mater. 2015, 14 (11), 1123– 1129, DOI: 10.1038/nmat4392Google ScholarThere is no corresponding record for this reference.
- 11Hutchison, J. A.; Schwartz, T.; Genet, C.; Devaux, E.; Ebbesen, T. W. Modifying Chemical Landscapes by Coupling to Vacuum Fields. Angew. Chem., Int. Ed. 2012, 51 (7), 1592– 1596, DOI: 10.1002/anie.201107033Google ScholarThere is no corresponding record for this reference.
- 12Nagarajan, K.; Thomas, A.; Ebbesen, T. W. Chemistry under Vibrational Strong Coupling. J. Am. Chem. Soc. 2021, 143 (41), 16877– 16889, DOI: 10.1021/jacs.1c07420Google ScholarThere is no corresponding record for this reference.
- 13Hirai, K.; Hutchison, J. A.; Uji-i, H. Molecular Chemistry in Cavity Strong Coupling. Chem. Rev. 2023, 123, 8099– 8126, DOI: 10.1021/acs.chemrev.2c00748Google ScholarThere is no corresponding record for this reference.
- 14Mony, J.; Climent, C.; Petersen, A. U.; Moth-Poulsen, K.; Feist, J.; Börjesson, K. Photoisomerization Efficiency of a Solar Thermal Fuel in the Strong Coupling Regime. Adv. Funct Mater. 2021, 31 (21), 2010737 DOI: 10.1002/adfm.202010737Google ScholarThere is no corresponding record for this reference.
- 15Zeng, H.; Pérez-Sánchez, J. B.; Eckdahl, C. T.; Liu, P.; Chang, W. J.; Weiss, E. A.; Kalow, J. A.; Yuen-Zhou, J.; Stern, N. P. Control of Photoswitching Kinetics with Strong Light–Matter Coupling in a Cavity. J. Am. Chem. Soc. 2023, 145 (36), 19655– 19661, DOI: 10.1021/jacs.3c04254Google ScholarThere is no corresponding record for this reference.
- 16Lee, I.; Melton, S. R.; Xu, D.; Delor, M. Controlling Molecular Photoisomerization in Photonic Cavities through Polariton Funneling. J. Am. Chem. Soc. 2024, 146 (14), 9544– 9553, DOI: 10.1021/jacs.3c11292Google ScholarThere is no corresponding record for this reference.
- 17Thomas, P. A.; Tan, W. J.; Kravets, V. G.; Grigorenko, A. N.; Barnes, W. L. Non-Polaritonic Effects in Cavity-Modified Photochemistry. Adv. Mater. 2024, 36 (7), 2309393 DOI: 10.1002/adma.202309393Google ScholarThere is no corresponding record for this reference.
- 18Schwartz, T.; Hutchison, J. A. Comment on “Non-Polaritonic Effects in Cavity-Modified Photochemistry”: On the Importance of Experimental Details. Adv. Mater. 2025, 37 (24), 2404602 DOI: 10.1002/adma.202404602Google ScholarThere is no corresponding record for this reference.
- 19Shalabney, A.; George, J.; Hutchison, J.; Pupillo, G.; Genet, C.; Ebbesen, T. W. Coherent Coupling of Molecular Resonators with a Microcavity Mode. Nat. Commun. 2015, 6, 5981, DOI: 10.1038/ncomms6981Google ScholarThere is no corresponding record for this reference.
- 20Thomas, A.; George, J.; Shalabney, A.; Dryzhakov, M.; Varma, S. J.; Moran, J.; Chervy, T.; Zhong, X.; Devaux, E.; Genet, C.; Hutchison, J. A.; Ebbesen, T. W. Ground-State Chemical Reactivity under Vibrational Coupling to the Vacuum Electromagnetic Field. Angew. Chem., Int. Ed. 2016, 55 (38), 11462– 11466, DOI: 10.1002/anie.201605504Google ScholarThere is no corresponding record for this reference.
- 21Thomas, A.; Lethuillier-Karl, L.; Nagarajan, K.; Vergauwe, R. M. A.; George, J.; Chervy, T.; Shalabney, A.; Devaux, E.; Genet, C.; Moran, J.; Ebbesen, T. W. Tilting a Ground-State Reactivity Landscape by Vibrational Strong Coupling. Science 2019, 363 (6427), 615– 619, DOI: 10.1126/science.aau7742Google ScholarThere is no corresponding record for this reference.
- 22Imperatore, M. V.; Asbury, J. B.; Giebink, N. C. Reproducibility of Cavity-Enhanced Chemical Reaction Rates in the Vibrational Strong Coupling Regime. J. Chem. Phys. 2021, 154, 191103 DOI: 10.1063/5.0046307Google ScholarThere is no corresponding record for this reference.
- 23Wiesehan, G. D.; Xiong, W. Negligible rate enhancement from reported cooperative vibrational strong coupling catalysis. J. Chem. Phys. 2021, 155, 241103 DOI: 10.1063/5.0077549Google ScholarThere is no corresponding record for this reference.
- 24Lather, J.; Bhatt, P.; Thomas, A.; Ebbesen, T. W.; George, J. Cavity Catalysis by Cooperative Vibrational Strong Coupling of Reactant and Solvent Molecules. Angew. Chem., Int. Ed. 2019, 58 (31), 10635– 10638, DOI: 10.1002/anie.201905407Google ScholarThere is no corresponding record for this reference.
- 25Hiura, H.; Shalabney, A. Vacuum-Field Catalysis: Accelerated Reactions by Vibrational Ultra Strong Coupling. ChemRxiv 2021.Google ScholarThere is no corresponding record for this reference.
- 26Verdelli, F.; Wei, Y.-C.; Joseph, K.; Abdelkhalik, M. S.; Goudarzi, M.; Askes, S. H. C.; Baldi, A.; Meijer, E. W.; Gomez Rivas, J. Polaritonic Chemistry Enabled by Non-Local Metasurfaces. Angew. Chem., Int. Ed. 2024, 63 (48), e202409528 DOI: 10.1002/anie.202409528Google ScholarThere is no corresponding record for this reference.
- 27Lian, J.; Song, Y.; Si, Q.; Zhao, X.; Chen, L.; Xu, R.; Zhang, F. Continuous-Flow Fabry-Pérot Cavity for Enhanced Catalysis via Cooperative Vibrational Strong Coupling. ACS Photonics 2025, 12 (7), 3557– 3564, DOI: 10.1021/acsphotonics.5c00417Google ScholarThere is no corresponding record for this reference.
- 28Wang, Y.; Rosenmann, D.; Muntean, J. V.; Ma, X. Polaritonic Bright and Dark States Collectively Affect the Reactivity of a Hydrolysis Reaction. ACS Photonics 2025, 12 (1), 263– 270, DOI: 10.1021/acsphotonics.4c01630Google ScholarThere is no corresponding record for this reference.
- 29Chikkaraddy, R.; de Nijs, B.; Benz, F.; Barrow, S. J.; Scherman, O. A.; Rosta, E.; Demetriadou, A.; Fox, P.; Hess, O.; Baumberg, J. J. Single-Molecule Strong Coupling at Room Temperature in Plasmonic Nanocavities. Nature 2016, 535 (7610), 127– 130, DOI: 10.1038/nature17974Google ScholarThere is no corresponding record for this reference.
- 30Zhong, X.; Chervy, T.; Wang, S.; George, J.; Thomas, A.; Hutchison, J. A.; Devaux, E.; Genet, C.; Ebbesen, T. W. Non-Radiative Energy Transfer Mediated by Hybrid Light-Matter States. Angew. Chem., Int. Ed. 2016, 55 (21), 6202– 6206, DOI: 10.1002/anie.201600428Google ScholarThere is no corresponding record for this reference.
- 31Zhong, X.; Chervy, T.; Zhang, L.; Thomas, A.; George, J.; Genet, C.; Hutchison, J. A.; Ebbesen, T. W. Energy Transfer between Spatially Separated Entangled Molecules. Angew. Chem., Int. Ed. 2017, 56 (31), 9034– 9038, DOI: 10.1002/anie.201703539Google ScholarThere is no corresponding record for this reference.
- 32Xiang, B.; Ribeiro, R. F.; Du, M.; Chen, L.; Yang, Z.; Wang, J.; Yuen-Zhou, J.; Xiong, W. Intermolecular vibrational energy transfer enabled by microcavity strong light-matter coupling. Science 2020, 368, 665– 667, DOI: 10.1126/science.aba3544Google ScholarThere is no corresponding record for this reference.
- 33Pang, Y.; Thomas, A.; Nagarajan, K.; Vergauwe, R. M. A.; Joseph, K.; Patrahau, B.; Wang, K.; Genet, C.; Ebbesen, T. W. On the Role of Symmetry in Vibrational Strong Coupling: The Case of Charge-Transfer Complexation. Angew. Chem., Int. Ed. 2020, 59 (26), 10436– 10440, DOI: 10.1002/anie.202002527Google ScholarThere is no corresponding record for this reference.
- 34Kumar, S.; Biswas, S.; Rashid, U.; Mony, K. S.; Chandrasekharan, G.; Mattiotti, F.; Vergauwe, R. M. A.; Hagenmuller, D.; Kaliginedi, V.; Thomas, A. Extraordinary Electrical Conductance through Amorphous Nonconducting Polymers under Vibrational Strong Coupling. J. Am. Chem. Soc. 2024, 146 (28), 18999– 19008, DOI: 10.1021/jacs.4c03016Google ScholarThere is no corresponding record for this reference.
- 35Jayachandran, A.; Patrahau, B.; Ricca, J. G.; Mahato, M.; Pang, Y.; Nagarajan, K.; Thomas, A.; Genet, C.; Ebbesen, T. W. A Phenomenological Symmetry Rule for Chemical Reactivity under Vibrational Strong Coupling. Angew. Chem., Int. Ed. 2025, 137 (35), e202503915 DOI: 10.1002/ange.202503915Google ScholarThere is no corresponding record for this reference.
- 36Flick, J.; Ruggenthaler, M.; Appel, H.; Rubio, A. Atoms and Molecules in Cavities, from Weak to Strong Coupling in Quantum-Electrodynamics (QED) Chemistry. Proc. Natl. Acad. Sci. U. S. A. 2017, 114, 3026– 3034, DOI: 10.1073/pnas.1615509114Google ScholarThere is no corresponding record for this reference.
- 37Ribeiro, R. F.; Martínez-Martínez, L. A.; Du, M.; Campos-Gonzalez-Angulo, J.; Yuen-Zhou, J. Polariton Chemistry: Controlling Molecular Dynamics with Optical Cavities. Chem. Sci. 2018, 9, 6325– 6339, DOI: 10.1039/C8SC01043AGoogle ScholarThere is no corresponding record for this reference.
- 38Mandal, A.; Taylor, M. A. D.; Weight, B. M.; Koessler, E. R.; Li, X.; Huo, P. Theoretical Advances in Polariton Chemistry and Molecular Cavity Quantum Electrodynamics. Chem. Rev. 2023, 123 (16), 9786– 9879, DOI: 10.1021/acs.chemrev.2c00855Google ScholarThere is no corresponding record for this reference.
- 39Mondal, S.; Keshavamurthy, S. Cavity Induced Modulation of Intramolecular Vibrational Energy Flow Pathways. J. Chem. Phys. 2024, 161 (19), 194302 DOI: 10.1063/5.0236437Google ScholarThere is no corresponding record for this reference.
- 40Jaynes, E. T.; Cummings, F. W. Comparison of Quantum and Semiclassical Radiation Theories with Application to the Beam Maser. Proc. IEEE 1963, 51 (1), 89– 109, DOI: 10.1109/PROC.1963.1664Google ScholarThere is no corresponding record for this reference.
- 41Tavis, M.; Cummings, F. W. Exact Solution for an N-Molecule-Radiation-Field Hamiltonian. Phys. Rev. 1968, 170 (2), 379– 384, DOI: 10.1103/PhysRev.170.379Google ScholarThere is no corresponding record for this reference.
- 42Biswas, S.; Mondal, M.; Chandrasekharan, G.; Mony, K. S.; Singh, A.; Thomas, A. Electronic Strong Coupling Modifies the Ground-State Intermolecular Interactions in Self-Assembled Chlorin Molecules. Nat. Commun. 2025, 16 (1), 5115, DOI: 10.1038/s41467-025-60025-8Google ScholarThere is no corresponding record for this reference.
- 43Mahato, M. K.; Mony, K. S.; Baliyan, H.; Biswas, S.; Thomas, A. Exploring Excited State Proton Transfer in Thin Films Under Vibrational Strong Coupling. Angew. Chem. Int. Ed. 2025, 64, e202424247 DOI: 10.1002/ange.202424247Google ScholarThere is no corresponding record for this reference.
- 44Mirkovic, T.; Ostroumov, E. E.; Anna, J. M.; van Grondelle, R.; Govindjee; Scholes, G. D. Light Absorption and Energy Transfer in the Antenna Complexes of Photosynthetic Organisms. Chem. Rev. 2017, 117 (2), 249– 293, DOI: 10.1021/acs.chemrev.6b00002Google ScholarThere is no corresponding record for this reference.
- 45Sáez-Blázquez, R.; Feist, J.; Fernández-Domínguez, A. I.; García-Vidal, F. J. Organic Polaritons Enable Local Vibrations to Drive Long-Range Energy Transfer. Phys. Rev. B 2018, 97 (24), 241407 DOI: 10.1103/PhysRevB.97.241407Google ScholarThere is no corresponding record for this reference.
- 46Aberra Guebrou, S.; Symonds, C.; Homeyer, E.; Plenet, J. C.; Gartstein, Yu. N.; Agranovich, V. M.; Bellessa, J. Coherent Emission from a Disordered Organic Semiconductor Induced by Strong Coupling with Surface Plasmons. Phys. Rev. Lett. 2012, 108 (6), 066401 DOI: 10.1103/PhysRevLett.108.066401Google ScholarThere is no corresponding record for this reference.
- 47Balasubrahmaniyam, M.; Simkhovich, A.; Golombek, A.; Sandik, G.; Ankonina, G.; Schwartz, T. From Enhanced Diffusion to Ultrafast Ballistic Motion of Hybrid Light–Matter Excitations. Nat. Mater. 2023, 22 (3), 338– 344, DOI: 10.1038/s41563-022-01463-3Google ScholarThere is no corresponding record for this reference.
- 48Xu, D.; Mandal, A.; Baxter, J. M.; Cheng, S.-W.; Lee, I.; Su, H.; Liu, S.; Reichman, D. R.; Delor, M. Ultrafast Imaging of Polariton Propagation and Interactions. Nat. Commun. 2023, 14 (1), 3881, DOI: 10.1038/s41467-023-39550-xGoogle ScholarThere is no corresponding record for this reference.
- 49Scholes, G. D. Emergence of Collective Coherent States from Strong-Light Coupling of Disordered Systems. J. Phys. Chem. A 2021, 125 (31), 6739– 6750, DOI: 10.1021/acs.jpca.1c05400Google ScholarThere is no corresponding record for this reference.
- 50Fukushima, T.; Yoshimitsu, S.; Murakoshi, K. Inherent Promotion of Ionic Conductivity via Collective Vibrational Strong Coupling of Water with the Vacuum Electromagnetic Field. J. Am. Chem. Soc. 2022, 144 (27), 12177– 12183, DOI: 10.1021/jacs.2c02991Google ScholarThere is no corresponding record for this reference.
- 51Fukushima, T.; Yoshimitsu, S.; Murakoshi, K. Unlimiting Ionic Conduction: Manipulating Hydration Dynamics through Vibrational Strong Coupling of Water. Chem. Sci. 2023, 14 (41), 11441– 11446, DOI: 10.1039/D3SC03364CGoogle ScholarThere is no corresponding record for this reference.
- 52Jones, B. A.; Facchetti, A.; Wasielewski, M. R.; Marks, T. J. Tuning Orbital Energetics in Arylene Diimide Semiconductors. Materials Design for Ambient Stability of n-Type Charge Transport. J. Am. Chem. Soc. 2007, 129 (49), 15259– 15278, DOI: 10.1021/ja075242eGoogle ScholarThere is no corresponding record for this reference.
- 53Kaur, K.; Johns, B.; Bhatt, P.; George, J. Controlling Electron Mobility of Strongly Coupled Organic Semiconductors in Mirrorless Cavities. Adv. Funct. Mater. 2023, 33 (47), 2306058 DOI: 10.1002/adfm.202306058Google ScholarThere is no corresponding record for this reference.
- 54Hagenmüller, D.; Schachenmayer, J.; Schütz, S.; Genes, C.; Pupillo, G. Cavity-Enhanced Transport of Charge. Phys. Rev. Lett. 2017, 119 (22), 223601 DOI: 10.1103/PhysRevLett.119.223601Google ScholarThere is no corresponding record for this reference.
- 55Nagarajan, K.; George, J.; Thomas, A.; Devaux, E.; Chervy, T.; Azzini, S.; Joseph, K.; Jouaiti, A.; Hosseini, M. W.; Kumar, A.; Genet, C.; Bartolo, N.; Ciuti, C.; Ebbesen, T. W. Conductivity and Photoconductivity of a P-Type Organic Semiconductor under Ultrastrong Coupling. ACS Nano 2020, 14 (8), 10219– 10225, DOI: 10.1021/acsnano.0c03496Google ScholarThere is no corresponding record for this reference.
- 56Lehn, J.-M. Supramolecular Chemistry─Scope and Perspectives Molecules, Supermolecules, and Molecular Devices (Nobel Lecture). Angew. Chem., Int. Ed. 1988, 27 (1), 89– 112, DOI: 10.1002/anie.198800891Google ScholarThere is no corresponding record for this reference.
- 57Hirai, K.; Ishikawa, H.; Chervy, T.; Hutchison, J. A.; Uji-i, H. Selective Crystallization via Vibrational Strong Coupling. Chem. Sci. 2021, 12 (36), 11986– 11994, DOI: 10.1039/D1SC03706DGoogle ScholarThere is no corresponding record for this reference.
- 58Joseph, K.; Kushida, S.; Smarsly, E.; Ihiawakrim, D.; Thomas, A.; Paravicini-Bagliani, G. L.; Nagarajan, K.; Vergauwe, R.; Devaux, E.; Ersen, O.; Bunz, U. H. F.; Ebbesen, T. W. Supramolecular Assembly of Conjugated Polymers under Vibrational Strong Coupling. Angew. Chem., Int. Ed. 2021, 60 (36), 19665– 19670, DOI: 10.1002/anie.202105840Google ScholarThere is no corresponding record for this reference.
- 59Joseph, K.; de Waal, B.; Jansen, S. A. H.; van der Tol, J. J. B.; Vantomme, G.; Meijer, E. W. Consequences of Vibrational Strong Coupling on Supramolecular Polymerization of Porphyrins. J. Am. Chem. Soc. 2024, 146 (17), 12130– 12137, DOI: 10.1021/jacs.4c02267Google ScholarThere is no corresponding record for this reference.
- 60Imai, S.; Hamada, T.; Nozaki, M.; Fujita, T.; Takahashi, M.; Fujita, Y.; Harano, K.; Uji-i, H.; Takai, A.; Hirai, K. Accessing a Hidden Pathway to Supramolecular Toroid through Vibrational Strong Coupling. J. Am. Chem. Soc. 2025, 147 (27), 23528– 23535, DOI: 10.1021/jacs.5c02960Google ScholarThere is no corresponding record for this reference.
- 61Patrahau, B.; Piejko, M.; Mayer, R. J.; Antheaume, C.; Sangchai, T.; Ragazzon, G.; Jayachandran, A.; Devaux, E.; Genet, C.; Moran, J.; Ebbesen, T. W. Direct Observation of Polaritonic Chemistry by Nuclear Magnetic Resonance Spectroscopy. Angew. Chem., Int. Ed. 2024, 63 (23), e202401368 DOI: 10.1002/anie.202401368Google ScholarThere is no corresponding record for this reference.
- 62Canaguier-Durand, A.; Devaux, E.; George, J.; Pang, Y.; Hutchison, J. A.; Schwartz, T.; Genet, C.; Wilhelms, N.; Lehn, J.-M.; Ebbesen, T. W. Thermodynamics of Molecules Strongly Coupled to the Vacuum Field. Angew. Chem., Int. Ed. 2013, 52 (40), 10533– 10536, DOI: 10.1002/anie.201301861Google ScholarThere is no corresponding record for this reference.
- 63Hutchison, J. A.; Liscio, A.; Schwartz, T.; Canaguier-Durand, A.; Genet, C.; Palermo, V.; Samorì, P.; Ebbesen, T. W. Tuning the Work-Function Via Strong Coupling. Adv. Mater. 2013, 25 (17), 2481– 2485, DOI: 10.1002/adma.201203682Google ScholarThere is no corresponding record for this reference.
- 64Castagnola, M.; Haugland, T. S.; Ronca, E.; Koch, H.; Schäfer, C. Collective Strong Coupling Modifies Aggregation and Solvation. J. Phys. Chem. Lett. 2024, 15 (5), 1428– 1434, DOI: 10.1021/acs.jpclett.3c03506Google ScholarThere is no corresponding record for this reference.
- 65Haugland, T. S.; Schäfer, C.; Ronca, E.; Rubio, A.; Koch, H. Intermolecular Interactions in Optical Cavities: An Ab Initio QED Study. J. Chem. Phys. 2021, 154 (9), 094113 DOI: 10.1063/5.0039256Google ScholarThere is no corresponding record for this reference.
- 66Scholes, G. D.; DelPo, C. A.; Kudisch, B. Entropy Reorders Polariton States. J. Phys. Chem. Lett. 2020, 11 (15), 6389– 6395, DOI: 10.1021/acs.jpclett.0c02000Google ScholarThere is no corresponding record for this reference.
- 67Zhou, Q.; Kukier, G.; Gordiy, I.; Hoffmann, R.; Seeman, J. I.; Houk, K. N. A 21st Century View of Allowed and Forbidden Electrocyclic Reactions. J. Org. Chem. 2024, 89 (2), 1018– 1034, DOI: 10.1021/acs.joc.3c02103Google ScholarThere is no corresponding record for this reference.
- 68Mondal, S.; Keshavamurthy, S. Phase Space Perspective on a Model for Isomerization in an Optical Cavity. J. Chem. Phys. 2023, 159 (7), 074106 DOI: 10.1063/5.0160586Google ScholarThere is no corresponding record for this reference.
- 69Mellini, F.; Vendrell, O. Competition between Coherent Ultrafast Energy Redistribution and Photochemistry in the Collective Strong Coupling Regime: The Role of Static Disorder. J. Phys. Chem. Lett. 2025, 16 (24), 6155– 6162, DOI: 10.1021/acs.jpclett.5c01117Google ScholarThere is no corresponding record for this reference.
- 70Sidler, D.; Ruggenthaler, M.; Rubio, A. Collectively-Modified Inter-Molecular Electron Correlations: The Connection of Polaritonic Chemistry and Spin Glass Physics. arXiv 2025.Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1
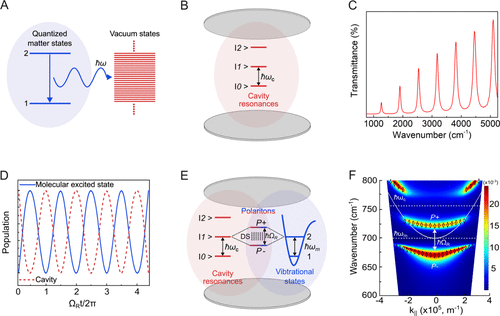
Figure 1. (A) Two-level atomic system with quantized energy levels emitting a photon into a continuum of vacuum states. (B) Schematic illustration of a Fabry–Pérot cavity with its resonances. (C) FT-IR transmission spectrum showing the resonances of a Fabry–Perot cavity of 8 μm path length filled with air. Schematic representation of (D) the energy exchange between a lossless cavity and a molecule resulting in the Rabi oscillation and (E) the resulting strong coupling and formation of the light–matter hybrid states in a cavity. (F) Angle-dependent dispersion of the polaritonic states with respect to the in-plane wave vector (black diamonds) overlaid on the simulated dispersion contour of the uncoupled cavity mode (ℏωc = 698 cm–1) and the vibrational transition of PS (ℏωm = 698 cm–1). Adapted from ref (34). Copyright 2024 American Chemical Society.
Figure 2
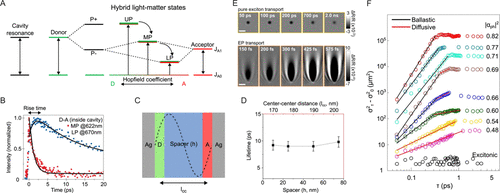
Figure 2. (A) Schematic representation of strong coupling of the electronic transitions of the donor and acceptor, together with a cavity resonance, resulting in three polaritonic states: the upper polariton (UP), middle polariton (MP), and lower polariton (LP). Reproduced from ref (30). Copyright 2016 John Wiley and Sons. (B) Excited-state dynamics of MP and LP. Adapted from ref (30). Copyright 2016 John Wiley and Sons. (C) Schematic representation of a cavity containing spatially separated donors and acceptors. The spatial separation corresponds to the thickness (h) of the polymer layer. Reproduced from ref (31). Copyright 2017 John Wiley and Sons. (D) Lifetime of MP as a function of the spacer thickness in the strong coupling conditions. Adapted from ref (31). Copyright 2017 John Wiley and Sons. (E) Momentum-resolved ultrafast imaging of pure exciton transport (top panel) and polariton transport (bottom panel). Reproduced from ref (48). Available under a CC-BY 4.0 license. Copyright 2023 Xu et al. (F) Time-resolved dynamics of the variance of polariton cloud expansion measured at different photonic fractions showing the ballistic and diffusive energy transport mediated by polaritons. Adapted from ref (47). Copyright 2023 Springer Nature.
Figure 3

Figure 3. (A) Schematic representation of the ionic conductivity under VSC of the O–H stretching vibrational mode of water. Adapted from ref (50). Copyright 2022 American Chemical Society. (B) Emission intensity ratio IRO–/IROH, as a function of the different cavity tunings. The blue spheres indicate the emission ratio in different cavities with resonances at the wavenumbers noted on the x axis. The purple dotted curve shows absorption of the PVA −OH stretching mode. The black dashed line indicates the average IRO–/IROH observed in noncavities. Adapted from ref (43). Copyright 2025 John Wiley and Sons. (C) I–V plot of the PDI2EH-CN2 deposited on hexagonal hole arrays with different periods of holes. Reproduced from ref (10). Copyright 2015 Springer Nature. (D) Schematic representation of a mirrorless MOSFET Fabry–Pérot cavity. The blue curve shows the cavity mode interacting with the active layer. (E) Electron mobility as a function of the cavity tuning. The first- and second-mode cavities are prepared by tuning the thickness of the organic semiconductor. The bottom panel shows the cavity mode position with respect to the electronic transition of the semiconductor (D and E) Adapted from ref (53). Copyright 2021 John Wiley and Sons.
Figure 4
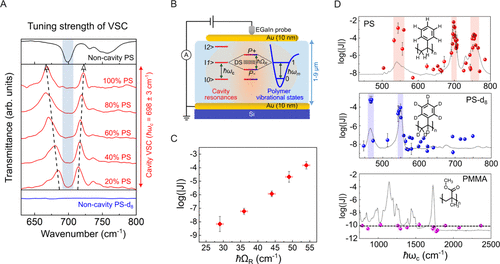
Figure 4. (A) FT-IR transmission spectra of cavities strongly coupled to the δ(Ar. CH) modes of PS when mixed with different weight percents of PS-d8. (B) Schematic illustration of VSC resulting in polaritonic states and the electrical conductance measurement setup. (C) Variation in conductance as a function of the strong coupling strength. (D) Action spectra of the electrical conductivity of aromatic polymers (PS and PS-d8) and nonaromatic polymer (PMMA) inside the cavity. The cavity modes are tuned with respect to the different vibrations of the polymers. The red, blue, and pink spheres correspond to the current densities of PS, PS-d8, and PMMA, respectively. Adapted from ref (34). Copyright 2024 American Chemical Society.
Figure 5

Figure 5. (A) Schematic representation of a Fabry–Pérot cavity containing PPE dissolved in dichlorobenzene. The cooperative VSC transforms the morphology of the polymer from fiber to sheets. Adapted from ref (58). Copyright 2021 John Wiley and Sons. (B) Supramolecular polymerization of chiral zinc porphyrin under on- and off-resonance conditions probed through electronic circular dichroism (ECD). The ECD signals of these cavities are plotted with respect to different temperatures as a function of the concentration. The elongation temperature of S–Zn is denoted by the yellow dashed line. Adapted from ref (59). Available under a CC-BY 4.0 license. Copyright 2025 Joseph et al. (C) Schematic illustration showing transformation of the naphthalenediimide supramolecular polymer in response to the click reaction under VSC and noncavity situations and their atomic force microscopy images. Adapted from ref (60). Available under a CC-BY 4.0 license. Copyright 2025 Imai et al.
Figure 6
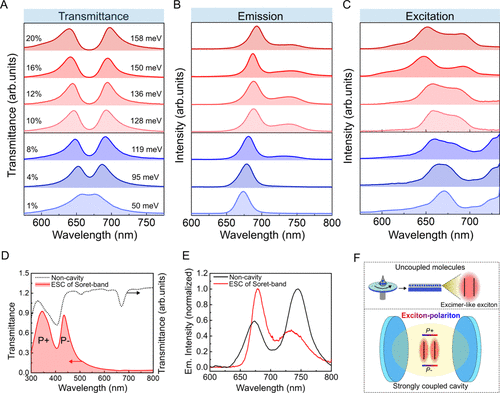
Figure 6. (A–C) Transmittance, emission (λex = 450 nm), and excitation (for λem = 770 nm) spectra of cavities ranging from weak (purple curves) to strong (red curves) coupling of the Ce6T Q band. (D and E) Transmittance, emission, and excitation (for λem = 770 nm) spectra of the Ce6T thin film and cavity strongly coupled to the Soret band. (F) Schematic illustration of the excimer-like exciton formation between two interacting Ce6T molecules in a noncavity film upon spin coating (top panel). The bottom panel shows the Ce6T molecules with a suppressed intermolecular interaction under ESC. Reproduced from ref (42). Available under a CC BY-NC-ND 4.0 license. Copyright 2025 Biswas et al.
References
This article references 70 other publications.
- 1Fox, M. Quantum Optics: An Introduction; Oxford University Press: Oxford, NY, 2006.There is no corresponding record for this reference.
- 2Haroche, S.; Kleppner, D. Cavity Quantum Electrodynamics. Phys. Today 1989, 42 (1), 24– 30, DOI: 10.1063/1.881201There is no corresponding record for this reference.
- 3Ebbesen, T. W. Hybrid Light–Matter States in a Molecular and Material Science Perspective. Acc. Chem. Res. 2016, 49 (11), 2403– 2412, DOI: 10.1021/acs.accounts.6b00295There is no corresponding record for this reference.
- 4Garcia-Vidal, F. J.; Ciuti, C.; Ebbesen, T. W. Manipulating Matter by Strong Coupling to Vacuum Fields. Science 2021, 373, eabd0336 DOI: 10.1126/science.abd0336There is no corresponding record for this reference.
- 5Kaluzny, Y.; Goy, P.; Gross, M.; Raimond, J. M.; Haroche, S. Observation of Self-Induced Rabi Oscillations in Two-Level Atoms Excited Inside a Resonant Cavity: The Ringing Regime of Superradiance. Phys. Rev. Lett. 1983, 51 (13), 1175– 1178, DOI: 10.1103/PhysRevLett.51.1175There is no corresponding record for this reference.
- 6Weisbuch, C.; Nishioka, M.; Ishikawa, A.; Arakawa, Y. Observation of the Coupled Exciton-Photon Mode Splitting in a Semiconductor Quantum Microcavity. Phys. Rev. Lett. 1992, 69 (23), 3314– 3317, DOI: 10.1103/PhysRevLett.69.3314There is no corresponding record for this reference.
- 7Pockrand, I.; Brillante, A.; Möbius, D. Exciton–Surface Plasmon Coupling: An Experimental Investigation. J. Chem. Phys. 1982, 77 (12), 6289– 6295, DOI: 10.1063/1.443834There is no corresponding record for this reference.
- 8Lidzey, D. G.; Bradley, D. D. C.; Skolnick, M. S.; Virgili, T.; Walker, S.; Whittaker, D. M. Strong Exciton–Photon Coupling in an Organic Semiconductor Microcavity. Nature 1998, 395 (6697), 53– 55, DOI: 10.1038/25692There is no corresponding record for this reference.
- 9Bellessa, J.; Bonnand, C.; Plenet, J. C.; Mugnier, J. Strong Coupling between Surface Plasmons and Excitons in an Organic Semiconductor. Phys. Rev. Lett. 2004, 93 (3), 036404 DOI: 10.1103/PhysRevLett.93.036404There is no corresponding record for this reference.
- 10Orgiu, E.; George, J.; Hutchison, J. A.; Devaux, E.; Dayen, J. F.; Doudin, B.; Stellacci, F.; Genet, C.; Schachenmayer, J.; Genes, C.; Pupillo, G.; Samorì, P.; Ebbesen, T. W. Conductivity in Organic Semiconductors Hybridized with the Vacuum Field. Nat. Mater. 2015, 14 (11), 1123– 1129, DOI: 10.1038/nmat4392There is no corresponding record for this reference.
- 11Hutchison, J. A.; Schwartz, T.; Genet, C.; Devaux, E.; Ebbesen, T. W. Modifying Chemical Landscapes by Coupling to Vacuum Fields. Angew. Chem., Int. Ed. 2012, 51 (7), 1592– 1596, DOI: 10.1002/anie.201107033There is no corresponding record for this reference.
- 12Nagarajan, K.; Thomas, A.; Ebbesen, T. W. Chemistry under Vibrational Strong Coupling. J. Am. Chem. Soc. 2021, 143 (41), 16877– 16889, DOI: 10.1021/jacs.1c07420There is no corresponding record for this reference.
- 13Hirai, K.; Hutchison, J. A.; Uji-i, H. Molecular Chemistry in Cavity Strong Coupling. Chem. Rev. 2023, 123, 8099– 8126, DOI: 10.1021/acs.chemrev.2c00748There is no corresponding record for this reference.
- 14Mony, J.; Climent, C.; Petersen, A. U.; Moth-Poulsen, K.; Feist, J.; Börjesson, K. Photoisomerization Efficiency of a Solar Thermal Fuel in the Strong Coupling Regime. Adv. Funct Mater. 2021, 31 (21), 2010737 DOI: 10.1002/adfm.202010737There is no corresponding record for this reference.
- 15Zeng, H.; Pérez-Sánchez, J. B.; Eckdahl, C. T.; Liu, P.; Chang, W. J.; Weiss, E. A.; Kalow, J. A.; Yuen-Zhou, J.; Stern, N. P. Control of Photoswitching Kinetics with Strong Light–Matter Coupling in a Cavity. J. Am. Chem. Soc. 2023, 145 (36), 19655– 19661, DOI: 10.1021/jacs.3c04254There is no corresponding record for this reference.
- 16Lee, I.; Melton, S. R.; Xu, D.; Delor, M. Controlling Molecular Photoisomerization in Photonic Cavities through Polariton Funneling. J. Am. Chem. Soc. 2024, 146 (14), 9544– 9553, DOI: 10.1021/jacs.3c11292There is no corresponding record for this reference.
- 17Thomas, P. A.; Tan, W. J.; Kravets, V. G.; Grigorenko, A. N.; Barnes, W. L. Non-Polaritonic Effects in Cavity-Modified Photochemistry. Adv. Mater. 2024, 36 (7), 2309393 DOI: 10.1002/adma.202309393There is no corresponding record for this reference.
- 18Schwartz, T.; Hutchison, J. A. Comment on “Non-Polaritonic Effects in Cavity-Modified Photochemistry”: On the Importance of Experimental Details. Adv. Mater. 2025, 37 (24), 2404602 DOI: 10.1002/adma.202404602There is no corresponding record for this reference.
- 19Shalabney, A.; George, J.; Hutchison, J.; Pupillo, G.; Genet, C.; Ebbesen, T. W. Coherent Coupling of Molecular Resonators with a Microcavity Mode. Nat. Commun. 2015, 6, 5981, DOI: 10.1038/ncomms6981There is no corresponding record for this reference.
- 20Thomas, A.; George, J.; Shalabney, A.; Dryzhakov, M.; Varma, S. J.; Moran, J.; Chervy, T.; Zhong, X.; Devaux, E.; Genet, C.; Hutchison, J. A.; Ebbesen, T. W. Ground-State Chemical Reactivity under Vibrational Coupling to the Vacuum Electromagnetic Field. Angew. Chem., Int. Ed. 2016, 55 (38), 11462– 11466, DOI: 10.1002/anie.201605504There is no corresponding record for this reference.
- 21Thomas, A.; Lethuillier-Karl, L.; Nagarajan, K.; Vergauwe, R. M. A.; George, J.; Chervy, T.; Shalabney, A.; Devaux, E.; Genet, C.; Moran, J.; Ebbesen, T. W. Tilting a Ground-State Reactivity Landscape by Vibrational Strong Coupling. Science 2019, 363 (6427), 615– 619, DOI: 10.1126/science.aau7742There is no corresponding record for this reference.
- 22Imperatore, M. V.; Asbury, J. B.; Giebink, N. C. Reproducibility of Cavity-Enhanced Chemical Reaction Rates in the Vibrational Strong Coupling Regime. J. Chem. Phys. 2021, 154, 191103 DOI: 10.1063/5.0046307There is no corresponding record for this reference.
- 23Wiesehan, G. D.; Xiong, W. Negligible rate enhancement from reported cooperative vibrational strong coupling catalysis. J. Chem. Phys. 2021, 155, 241103 DOI: 10.1063/5.0077549There is no corresponding record for this reference.
- 24Lather, J.; Bhatt, P.; Thomas, A.; Ebbesen, T. W.; George, J. Cavity Catalysis by Cooperative Vibrational Strong Coupling of Reactant and Solvent Molecules. Angew. Chem., Int. Ed. 2019, 58 (31), 10635– 10638, DOI: 10.1002/anie.201905407There is no corresponding record for this reference.
- 25Hiura, H.; Shalabney, A. Vacuum-Field Catalysis: Accelerated Reactions by Vibrational Ultra Strong Coupling. ChemRxiv 2021.There is no corresponding record for this reference.
- 26Verdelli, F.; Wei, Y.-C.; Joseph, K.; Abdelkhalik, M. S.; Goudarzi, M.; Askes, S. H. C.; Baldi, A.; Meijer, E. W.; Gomez Rivas, J. Polaritonic Chemistry Enabled by Non-Local Metasurfaces. Angew. Chem., Int. Ed. 2024, 63 (48), e202409528 DOI: 10.1002/anie.202409528There is no corresponding record for this reference.
- 27Lian, J.; Song, Y.; Si, Q.; Zhao, X.; Chen, L.; Xu, R.; Zhang, F. Continuous-Flow Fabry-Pérot Cavity for Enhanced Catalysis via Cooperative Vibrational Strong Coupling. ACS Photonics 2025, 12 (7), 3557– 3564, DOI: 10.1021/acsphotonics.5c00417There is no corresponding record for this reference.
- 28Wang, Y.; Rosenmann, D.; Muntean, J. V.; Ma, X. Polaritonic Bright and Dark States Collectively Affect the Reactivity of a Hydrolysis Reaction. ACS Photonics 2025, 12 (1), 263– 270, DOI: 10.1021/acsphotonics.4c01630There is no corresponding record for this reference.
- 29Chikkaraddy, R.; de Nijs, B.; Benz, F.; Barrow, S. J.; Scherman, O. A.; Rosta, E.; Demetriadou, A.; Fox, P.; Hess, O.; Baumberg, J. J. Single-Molecule Strong Coupling at Room Temperature in Plasmonic Nanocavities. Nature 2016, 535 (7610), 127– 130, DOI: 10.1038/nature17974There is no corresponding record for this reference.
- 30Zhong, X.; Chervy, T.; Wang, S.; George, J.; Thomas, A.; Hutchison, J. A.; Devaux, E.; Genet, C.; Ebbesen, T. W. Non-Radiative Energy Transfer Mediated by Hybrid Light-Matter States. Angew. Chem., Int. Ed. 2016, 55 (21), 6202– 6206, DOI: 10.1002/anie.201600428There is no corresponding record for this reference.
- 31Zhong, X.; Chervy, T.; Zhang, L.; Thomas, A.; George, J.; Genet, C.; Hutchison, J. A.; Ebbesen, T. W. Energy Transfer between Spatially Separated Entangled Molecules. Angew. Chem., Int. Ed. 2017, 56 (31), 9034– 9038, DOI: 10.1002/anie.201703539There is no corresponding record for this reference.
- 32Xiang, B.; Ribeiro, R. F.; Du, M.; Chen, L.; Yang, Z.; Wang, J.; Yuen-Zhou, J.; Xiong, W. Intermolecular vibrational energy transfer enabled by microcavity strong light-matter coupling. Science 2020, 368, 665– 667, DOI: 10.1126/science.aba3544There is no corresponding record for this reference.
- 33Pang, Y.; Thomas, A.; Nagarajan, K.; Vergauwe, R. M. A.; Joseph, K.; Patrahau, B.; Wang, K.; Genet, C.; Ebbesen, T. W. On the Role of Symmetry in Vibrational Strong Coupling: The Case of Charge-Transfer Complexation. Angew. Chem., Int. Ed. 2020, 59 (26), 10436– 10440, DOI: 10.1002/anie.202002527There is no corresponding record for this reference.
- 34Kumar, S.; Biswas, S.; Rashid, U.; Mony, K. S.; Chandrasekharan, G.; Mattiotti, F.; Vergauwe, R. M. A.; Hagenmuller, D.; Kaliginedi, V.; Thomas, A. Extraordinary Electrical Conductance through Amorphous Nonconducting Polymers under Vibrational Strong Coupling. J. Am. Chem. Soc. 2024, 146 (28), 18999– 19008, DOI: 10.1021/jacs.4c03016There is no corresponding record for this reference.
- 35Jayachandran, A.; Patrahau, B.; Ricca, J. G.; Mahato, M.; Pang, Y.; Nagarajan, K.; Thomas, A.; Genet, C.; Ebbesen, T. W. A Phenomenological Symmetry Rule for Chemical Reactivity under Vibrational Strong Coupling. Angew. Chem., Int. Ed. 2025, 137 (35), e202503915 DOI: 10.1002/ange.202503915There is no corresponding record for this reference.
- 36Flick, J.; Ruggenthaler, M.; Appel, H.; Rubio, A. Atoms and Molecules in Cavities, from Weak to Strong Coupling in Quantum-Electrodynamics (QED) Chemistry. Proc. Natl. Acad. Sci. U. S. A. 2017, 114, 3026– 3034, DOI: 10.1073/pnas.1615509114There is no corresponding record for this reference.
- 37Ribeiro, R. F.; Martínez-Martínez, L. A.; Du, M.; Campos-Gonzalez-Angulo, J.; Yuen-Zhou, J. Polariton Chemistry: Controlling Molecular Dynamics with Optical Cavities. Chem. Sci. 2018, 9, 6325– 6339, DOI: 10.1039/C8SC01043AThere is no corresponding record for this reference.
- 38Mandal, A.; Taylor, M. A. D.; Weight, B. M.; Koessler, E. R.; Li, X.; Huo, P. Theoretical Advances in Polariton Chemistry and Molecular Cavity Quantum Electrodynamics. Chem. Rev. 2023, 123 (16), 9786– 9879, DOI: 10.1021/acs.chemrev.2c00855There is no corresponding record for this reference.
- 39Mondal, S.; Keshavamurthy, S. Cavity Induced Modulation of Intramolecular Vibrational Energy Flow Pathways. J. Chem. Phys. 2024, 161 (19), 194302 DOI: 10.1063/5.0236437There is no corresponding record for this reference.
- 40Jaynes, E. T.; Cummings, F. W. Comparison of Quantum and Semiclassical Radiation Theories with Application to the Beam Maser. Proc. IEEE 1963, 51 (1), 89– 109, DOI: 10.1109/PROC.1963.1664There is no corresponding record for this reference.
- 41Tavis, M.; Cummings, F. W. Exact Solution for an N-Molecule-Radiation-Field Hamiltonian. Phys. Rev. 1968, 170 (2), 379– 384, DOI: 10.1103/PhysRev.170.379There is no corresponding record for this reference.
- 42Biswas, S.; Mondal, M.; Chandrasekharan, G.; Mony, K. S.; Singh, A.; Thomas, A. Electronic Strong Coupling Modifies the Ground-State Intermolecular Interactions in Self-Assembled Chlorin Molecules. Nat. Commun. 2025, 16 (1), 5115, DOI: 10.1038/s41467-025-60025-8There is no corresponding record for this reference.
- 43Mahato, M. K.; Mony, K. S.; Baliyan, H.; Biswas, S.; Thomas, A. Exploring Excited State Proton Transfer in Thin Films Under Vibrational Strong Coupling. Angew. Chem. Int. Ed. 2025, 64, e202424247 DOI: 10.1002/ange.202424247There is no corresponding record for this reference.
- 44Mirkovic, T.; Ostroumov, E. E.; Anna, J. M.; van Grondelle, R.; Govindjee; Scholes, G. D. Light Absorption and Energy Transfer in the Antenna Complexes of Photosynthetic Organisms. Chem. Rev. 2017, 117 (2), 249– 293, DOI: 10.1021/acs.chemrev.6b00002There is no corresponding record for this reference.
- 45Sáez-Blázquez, R.; Feist, J.; Fernández-Domínguez, A. I.; García-Vidal, F. J. Organic Polaritons Enable Local Vibrations to Drive Long-Range Energy Transfer. Phys. Rev. B 2018, 97 (24), 241407 DOI: 10.1103/PhysRevB.97.241407There is no corresponding record for this reference.
- 46Aberra Guebrou, S.; Symonds, C.; Homeyer, E.; Plenet, J. C.; Gartstein, Yu. N.; Agranovich, V. M.; Bellessa, J. Coherent Emission from a Disordered Organic Semiconductor Induced by Strong Coupling with Surface Plasmons. Phys. Rev. Lett. 2012, 108 (6), 066401 DOI: 10.1103/PhysRevLett.108.066401There is no corresponding record for this reference.
- 47Balasubrahmaniyam, M.; Simkhovich, A.; Golombek, A.; Sandik, G.; Ankonina, G.; Schwartz, T. From Enhanced Diffusion to Ultrafast Ballistic Motion of Hybrid Light–Matter Excitations. Nat. Mater. 2023, 22 (3), 338– 344, DOI: 10.1038/s41563-022-01463-3There is no corresponding record for this reference.
- 48Xu, D.; Mandal, A.; Baxter, J. M.; Cheng, S.-W.; Lee, I.; Su, H.; Liu, S.; Reichman, D. R.; Delor, M. Ultrafast Imaging of Polariton Propagation and Interactions. Nat. Commun. 2023, 14 (1), 3881, DOI: 10.1038/s41467-023-39550-xThere is no corresponding record for this reference.
- 49Scholes, G. D. Emergence of Collective Coherent States from Strong-Light Coupling of Disordered Systems. J. Phys. Chem. A 2021, 125 (31), 6739– 6750, DOI: 10.1021/acs.jpca.1c05400There is no corresponding record for this reference.
- 50Fukushima, T.; Yoshimitsu, S.; Murakoshi, K. Inherent Promotion of Ionic Conductivity via Collective Vibrational Strong Coupling of Water with the Vacuum Electromagnetic Field. J. Am. Chem. Soc. 2022, 144 (27), 12177– 12183, DOI: 10.1021/jacs.2c02991There is no corresponding record for this reference.
- 51Fukushima, T.; Yoshimitsu, S.; Murakoshi, K. Unlimiting Ionic Conduction: Manipulating Hydration Dynamics through Vibrational Strong Coupling of Water. Chem. Sci. 2023, 14 (41), 11441– 11446, DOI: 10.1039/D3SC03364CThere is no corresponding record for this reference.
- 52Jones, B. A.; Facchetti, A.; Wasielewski, M. R.; Marks, T. J. Tuning Orbital Energetics in Arylene Diimide Semiconductors. Materials Design for Ambient Stability of n-Type Charge Transport. J. Am. Chem. Soc. 2007, 129 (49), 15259– 15278, DOI: 10.1021/ja075242eThere is no corresponding record for this reference.
- 53Kaur, K.; Johns, B.; Bhatt, P.; George, J. Controlling Electron Mobility of Strongly Coupled Organic Semiconductors in Mirrorless Cavities. Adv. Funct. Mater. 2023, 33 (47), 2306058 DOI: 10.1002/adfm.202306058There is no corresponding record for this reference.
- 54Hagenmüller, D.; Schachenmayer, J.; Schütz, S.; Genes, C.; Pupillo, G. Cavity-Enhanced Transport of Charge. Phys. Rev. Lett. 2017, 119 (22), 223601 DOI: 10.1103/PhysRevLett.119.223601There is no corresponding record for this reference.
- 55Nagarajan, K.; George, J.; Thomas, A.; Devaux, E.; Chervy, T.; Azzini, S.; Joseph, K.; Jouaiti, A.; Hosseini, M. W.; Kumar, A.; Genet, C.; Bartolo, N.; Ciuti, C.; Ebbesen, T. W. Conductivity and Photoconductivity of a P-Type Organic Semiconductor under Ultrastrong Coupling. ACS Nano 2020, 14 (8), 10219– 10225, DOI: 10.1021/acsnano.0c03496There is no corresponding record for this reference.
- 56Lehn, J.-M. Supramolecular Chemistry─Scope and Perspectives Molecules, Supermolecules, and Molecular Devices (Nobel Lecture). Angew. Chem., Int. Ed. 1988, 27 (1), 89– 112, DOI: 10.1002/anie.198800891There is no corresponding record for this reference.
- 57Hirai, K.; Ishikawa, H.; Chervy, T.; Hutchison, J. A.; Uji-i, H. Selective Crystallization via Vibrational Strong Coupling. Chem. Sci. 2021, 12 (36), 11986– 11994, DOI: 10.1039/D1SC03706DThere is no corresponding record for this reference.
- 58Joseph, K.; Kushida, S.; Smarsly, E.; Ihiawakrim, D.; Thomas, A.; Paravicini-Bagliani, G. L.; Nagarajan, K.; Vergauwe, R.; Devaux, E.; Ersen, O.; Bunz, U. H. F.; Ebbesen, T. W. Supramolecular Assembly of Conjugated Polymers under Vibrational Strong Coupling. Angew. Chem., Int. Ed. 2021, 60 (36), 19665– 19670, DOI: 10.1002/anie.202105840There is no corresponding record for this reference.
- 59Joseph, K.; de Waal, B.; Jansen, S. A. H.; van der Tol, J. J. B.; Vantomme, G.; Meijer, E. W. Consequences of Vibrational Strong Coupling on Supramolecular Polymerization of Porphyrins. J. Am. Chem. Soc. 2024, 146 (17), 12130– 12137, DOI: 10.1021/jacs.4c02267There is no corresponding record for this reference.
- 60Imai, S.; Hamada, T.; Nozaki, M.; Fujita, T.; Takahashi, M.; Fujita, Y.; Harano, K.; Uji-i, H.; Takai, A.; Hirai, K. Accessing a Hidden Pathway to Supramolecular Toroid through Vibrational Strong Coupling. J. Am. Chem. Soc. 2025, 147 (27), 23528– 23535, DOI: 10.1021/jacs.5c02960There is no corresponding record for this reference.
- 61Patrahau, B.; Piejko, M.; Mayer, R. J.; Antheaume, C.; Sangchai, T.; Ragazzon, G.; Jayachandran, A.; Devaux, E.; Genet, C.; Moran, J.; Ebbesen, T. W. Direct Observation of Polaritonic Chemistry by Nuclear Magnetic Resonance Spectroscopy. Angew. Chem., Int. Ed. 2024, 63 (23), e202401368 DOI: 10.1002/anie.202401368There is no corresponding record for this reference.
- 62Canaguier-Durand, A.; Devaux, E.; George, J.; Pang, Y.; Hutchison, J. A.; Schwartz, T.; Genet, C.; Wilhelms, N.; Lehn, J.-M.; Ebbesen, T. W. Thermodynamics of Molecules Strongly Coupled to the Vacuum Field. Angew. Chem., Int. Ed. 2013, 52 (40), 10533– 10536, DOI: 10.1002/anie.201301861There is no corresponding record for this reference.
- 63Hutchison, J. A.; Liscio, A.; Schwartz, T.; Canaguier-Durand, A.; Genet, C.; Palermo, V.; Samorì, P.; Ebbesen, T. W. Tuning the Work-Function Via Strong Coupling. Adv. Mater. 2013, 25 (17), 2481– 2485, DOI: 10.1002/adma.201203682There is no corresponding record for this reference.
- 64Castagnola, M.; Haugland, T. S.; Ronca, E.; Koch, H.; Schäfer, C. Collective Strong Coupling Modifies Aggregation and Solvation. J. Phys. Chem. Lett. 2024, 15 (5), 1428– 1434, DOI: 10.1021/acs.jpclett.3c03506There is no corresponding record for this reference.
- 65Haugland, T. S.; Schäfer, C.; Ronca, E.; Rubio, A.; Koch, H. Intermolecular Interactions in Optical Cavities: An Ab Initio QED Study. J. Chem. Phys. 2021, 154 (9), 094113 DOI: 10.1063/5.0039256There is no corresponding record for this reference.
- 66Scholes, G. D.; DelPo, C. A.; Kudisch, B. Entropy Reorders Polariton States. J. Phys. Chem. Lett. 2020, 11 (15), 6389– 6395, DOI: 10.1021/acs.jpclett.0c02000There is no corresponding record for this reference.
- 67Zhou, Q.; Kukier, G.; Gordiy, I.; Hoffmann, R.; Seeman, J. I.; Houk, K. N. A 21st Century View of Allowed and Forbidden Electrocyclic Reactions. J. Org. Chem. 2024, 89 (2), 1018– 1034, DOI: 10.1021/acs.joc.3c02103There is no corresponding record for this reference.
- 68Mondal, S.; Keshavamurthy, S. Phase Space Perspective on a Model for Isomerization in an Optical Cavity. J. Chem. Phys. 2023, 159 (7), 074106 DOI: 10.1063/5.0160586There is no corresponding record for this reference.
- 69Mellini, F.; Vendrell, O. Competition between Coherent Ultrafast Energy Redistribution and Photochemistry in the Collective Strong Coupling Regime: The Role of Static Disorder. J. Phys. Chem. Lett. 2025, 16 (24), 6155– 6162, DOI: 10.1021/acs.jpclett.5c01117There is no corresponding record for this reference.
- 70Sidler, D.; Ruggenthaler, M.; Rubio, A. Collectively-Modified Inter-Molecular Electron Correlations: The Connection of Polaritonic Chemistry and Spin Glass Physics. arXiv 2025.There is no corresponding record for this reference.


