
Molecular Steric Engineering Enables High-Strength and Self-Healing Polyurethane Elastomer for Flexible and Sensitive NanosensorsClick to copy article linkArticle link copied!
- Shudiao WeiShudiao WeiEngineering Research Center for Energy Conversion and Storage Technology of Guizhou, School of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Shudiao Wei
- Weile GuoWeile GuoEngineering Research Center for Energy Conversion and Storage Technology of Guizhou, School of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Weile Guo
- Aiqin LiAiqin LiEngineering Research Center for Energy Conversion and Storage Technology of Guizhou, School of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Aiqin Li
- Menglan Lv*Menglan Lv*E-mail: [email protected]Engineering Research Center for Energy Conversion and Storage Technology of Guizhou, School of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Menglan Lv
- Lingli KongLingli KongSchool of Chemistry and Chemical Engineering, Guangxi University, Nanning 530004, ChinaMore by Lingli Kong
- Chuanhui XuChuanhui XuSchool of Chemistry and Chemical Engineering, Guangxi University, Nanning 530004, ChinaMore by Chuanhui Xu
- Ting GuTing GuSchool of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Ting Gu
- Bin Zhang*Bin Zhang*E-mail: [email protected]Engineering Research Center for Energy Conversion and Storage Technology of Guizhou, School of Chemistry and Chemical Engineering, Guizhou University, Guiyang 550025, ChinaMore by Bin Zhang
Abstract
Acquiring a versatile polyurethane (PU) elastomer with both high mechanical properties and rapid low-temperature healing for flexible nanosensors represents a significant challenge. Inspired by the synergistic effects of dynamic bonds, we introduce the dynamic sextuple hydrogen bonds (H-bonds) from adipic dihydrazide (AD) and flexible dynamic disulfide bonds (S–S bonds) from the chain extender 3,3′-dithiobis(2-butanol) (DS) into PU main chains to fabricate a high-performance elastomer (PU-3) with remarkable mechanical robustness and rapid self-healing capability. Specifically, AD imparts PU-3 with an exceptional tensile strength of 40.5 MPa and a toughness of 287.8 MJ m–3. A 1.1 g sample (50 mm × 10 mm × 1.3 mm) supports loads up to 11,000 times its own weight. In addition, the excellent DS unit, featuring four branched methyl groups with a substantially larger molecular volume, renders the PU-3 more flexible with an outstanding elongation at break of 1445.2%. Moreover, PU-3 exhibits excellent resilience, self-healing efficiency, and recyclability. As a result, a flexible polymer/carbon nanotube composite nanosensor is constructed from this elastomer matrix and multiwalled carboxylated carbon nanotubes (MWCNTs-COOH) as the conductive filler, exhibiting outstanding sensitivity and rapid response and recovery capabilities, thereby highlighting its potential for applications in health monitoring and intelligent wearable electronics.
This publication is licensed for personal use by The American Chemical Society.
1. Introduction
2. Results and Discussion
2.1. Preparation and Characterization of the Samples
Figure 1
Figure 1. (a) Molecular structure of the PU-3 elastomer. (b) Schematic of the network structure and dynamic bond interactions. (c) Proposed network structure of the PU-3 elastomer.
Figure 2
Figure 2. (a) FTIR spectra of PU-1, PU-2, and PU-3 elastomers. (b) Raman spectra. (c) XRD patterns. (d–f) Elemental mapping images of PU-1, PU-2, and PU-3 obtained from EDS. (g) Storage modulus (E′). (h) Loss modulus (E″). (i) Loss factor (tan δ).
2.2. Mechanical Properties and Fatigue Resistance of the Samples
Figure 3
Figure 3. (a) Stress–strain curves of the PU samples. (b) Toughness bar chart for different samples. (c, e) Cyclic tensile tests conducted at 500% strain for five consecutive loading–unloading cycles. (f) Performance comparison. (g) Fracture energy of PU-3. (h) Photograph of a notched PU-3 sample stretched to 800% strain.
2.3. Self-Healability of the PU Elastomers
Figure 4
Figure 4. (a, b) Stress–strain curves and bar chart of self-healing at various times at 60 °C for PU-1, (c, d) PU-2, and (e, f) PU-3, respectively. (g) Optical images of the self-healing process of PU-3. (h) Photograph of PU-3 after healing lifting a 5 kg weight.
2.4. Recyclability of the PU Elastomers
Figure 5
Figure 5. (a) Schematic illustration of the recycling process of the PU-3 elastomer. (b) FTIR spectra of the original and recycled PU-3 sample. (c) XRD patterns of the original and recycled PU-3 sample. (d) Stress–strain curves of PU-3 after two recycling cycles. (e) Demonstration of the recycled PU-3 elastomer lifting a 12 kg weight.
2.5. Preparation and Potential Applications for Flexible and Sensitive Nanosensors
Figure 6
Figure 6. (a) Schematic illustration of the fabrication process of the flexible and sensitive polymer/carbon nanotube composite nanosensor. (b) Relative resistance variation (ΔR/R0) of the sensor under tensile strains ranging from 3% to 300%. (c) GF of the nanosensor, where GF = ΔR/(R0·ε) and ε denotes strain. (d) Response and recovery times of the sensor at 100% strain. (e) Real-time signal output of the sensor during mouth breathing. (f) Schematic representation of the potential applications in human motion monitoring. (g) Signal response of the sensor during throat articulation of the word “drink”. (h) Electrical response of the sensor during finger bending and stretching at angles of 30°, 45°, 60°, and 90°. (i) Signal output during wrist bending and (j) signal response during walking and stair climbing.
3. Conclusions
4. Experimental Section
4.1. Materials
4.2. Synthesis of DS
4.3. Synthesis of PU Elastomers
4.4. Fabrication of Flexible and Sensitive Polymer/Carbon Nanotube Composite Nanosensors
4.5. Instrumentation and Characterization
4.6. Mechanical Properties Testing
| (1) | The specimens were prepared in a standard dumbbell geometry with an effective testing region of 20.0 mm × 4.0 mm × 0.5 mm. All tensile measurements were conducted at a constant crosshead speed of 50 mm min–1, and each formulation was tested at least three times to ensure reproducibility of the results. The tensile stress (σ) and elongation at break (ε) were obtained from the corresponding stress–strain profiles and calculated using the following equations: (2) | ||||
| (2) | The toughness (τ) of the specimen can be acquired by computing the area enclosed by the stress (σ)–strain (ε) curve, in accordance with the following formula: (3) | ||||
| (3) | In the cyclic testing procedure, the loading/unloading rate was determined at a constant pace with a strain rate of 50 mm min–1 under room temperature. The specimen was elongated to 500% elongation and subsequently underwent continuous loading and unloading cycles five times without interruption. | ||||
| (4) | For the fracture energy evaluation, the Greensmith approach was adopted to test both notched specimens (crack length: 1 mm) and their unnotched counterparts at a loading rate of 3 mm min–1. The samples used in the measurements had a gauge length of 10.0 mm, a width of 5.0 mm, and a thickness of 0.5 mm. The fracture energy Gc was determined according to the following expression: (4) | ||||
| (5) | For the self-healing experiments, the samples were first bisected, and the freshly cut surfaces were immediately brought into contact. The specimens were then subjected to healing at various temperatures. The self-healing efficiency was subsequently calculated using the following definition: (5) | ||||
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsapm.5c04892.
Synthetic routes and characterization data; 1H NMR spectra, 13C NMR spectra, and mass spectrum of monomer; 1H NMR spectra of PUs; SEC test results of PUs; SEM characterization results of PUs; TGA and DSC data of PUs; cyclic tensile test results of PUs; signal output diagrams of throat vibration during the utterance of the Chinese word “apple” collected by the sensor; signal output diagrams of elbow flexion states collected by the sensor (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (52373175), the Guizhou Provincial Basic Research Program (General Program) (MS[2025]603), the High-Level Innovative Talents Foundation of Guizhou Province (QKHPTRC-GCC[2023]-024), the Natural Science Foundation of Guizhou Province (QKHPTRC-CXTD[2023]005), and the Science and Technology Innovation Team of the Higher Education Department of Guizhou Province (QJJ[2023]053).
References
This article references 46 other publications.
- 1Alqaderi, A. J.; Ramakrishnan, N. Carbon-Based Flexible Strain Sensors: Recent Advances and Performance Insights in Human Motion Detection. Chem. Eng. J. 2025, 513, 162609 DOI: 10.1016/j.cej.2025.162609Google ScholarThere is no corresponding record for this reference.
- 2Lin, M. Z.; Zheng, Z. J.; Yang, L.; Luo, M. S.; Fu, L. H.; Lin, B. F.; Xu, C. H. A High-Performance, Sensitive, Wearable Multifunctional Sensor Based on Rubber/CNT for Human Motion and Skin Temperature Detection. Adv. Mater. 2022, 34 (1), 2107309 DOI: 10.1002/adma.202107309Google ScholarThere is no corresponding record for this reference.
- 3Tang, Y.; Fu, H.; Xu, B. G. Advanced Design of Triboelectric Nanogenerators for Future Eco-Smart Cities. Adv. Compos. Hybrid Mater. 2024, 7 (3), 102, DOI: 10.1007/s42114-024-00909-3Google ScholarThere is no corresponding record for this reference.
- 4Wu, C. J.; Gao, X. Q.; Zhu, H.; Wang, X.; Wang, S. L.; Fu, Y.; Han, Y. T.; Yuan, W. N.; Tian, Y. L.; Jiang, X. C.; Lai, W. Y. Multiscale Stress Transmission Regulation toward Flexible Strain Sensors with High Stability. Adv. Funct. Mater. 2026, 36, 10852, DOI: 10.1002/adfm.202510852Google ScholarThere is no corresponding record for this reference.
- 5Yadav, N.; Zhang, Q. Q.; Qi, D. F.; Yadav, A.; Zheng, H. Y. AI-Integrated Wearable Strain Sensors: Advances in E-Skin, Robotics, and Personalized Health Monitoring. Nanoscale Adv. 2025, 7 (16), 4803– 4819, DOI: 10.1039/D5NA00574DGoogle ScholarThere is no corresponding record for this reference.
- 6Lei, T. D.; Wang, Y. H.; Zhang, Q. S.; Wang, H. X.; Duan, X. R.; Yan, J.; Xia, Z. P.; Wang, R.; Shou, W.; Li, X. P.; Fan, J. Ultra-Stretchable and Anti-Freezing Ionic Conductive Hydrogels as High-Performance Strain Sensors and Flexible Triboelectric Nanogenerators in Extreme Environments. Nano Energy 2024, 126, 109633 DOI: 10.1016/j.nanoen.2024.109633Google ScholarThere is no corresponding record for this reference.
- 7Maji, A.; Kuila, C.; Murmu, N. C.; Kuila, T. Stretch, Sense, and Innovate: Advances in Next-Generation Strain Sensors. Compos. Part B Eng. 2025, 306, 112749 DOI: 10.1016/j.compositesb.2025.112749Google ScholarThere is no corresponding record for this reference.
- 8Padhan, A. K.; Sharma, D.; Thomas, T. S.; Sinha, A. P.; Mallick, A. N.; Mandal, D. Rapid Self-Healing and Superior Toughness in Ionically Crosslinked Polymer Ionogels and Strain Sensing Applications. J. Mater. Chem. A 2024, 12 (16), 9508– 9517, DOI: 10.1039/D3TA07277KGoogle ScholarThere is no corresponding record for this reference.
- 9Qu, J. T.; Cui, G. M.; Li, Z. K.; Fang, S. T.; Zhang, X. R.; Liu, A.; Han, M. Y.; Liu, H. D.; Wang, X. Q.; Wang, X. H. Advanced Flexible Sensing Technologies for Soft Robots. Adv. Funct. Mater. 2024, 34 (29), 2401311 DOI: 10.1002/adfm.202401311Google ScholarThere is no corresponding record for this reference.
- 10Yu, S. J.; Ye, Q. Q.; Yang, B.; Liu, X. J.; Zhou, H.; Hu, L.; Lu, C. X. Ultrasensitive, Highly Stretchable, and Multifunctional Strain Sensors Based on Scorpion-Leg-Inspired Gradient Crack Arrays. Chem. Eng. J. 2024, 497, 154952 DOI: 10.1016/j.cej.2024.154952Google ScholarThere is no corresponding record for this reference.
- 11Zhang, H. Y.; Yang, Q. N.; Xu, L. J.; Li, N.; Tan, H. H.; Du, J. J.; Yu, M. L.; Xu, J. X. Triboelectric Nanogenerators Based on Hydrated Lithium Ions Incorporated Double-Network Hydrogels for Biomechanical Sensing and Energy Harvesting at Low Temperature. Nano Energy 2024, 125, 109521 DOI: 10.1016/j.nanoen.2024.109521Google ScholarThere is no corresponding record for this reference.
- 12Dai, X. Y.; Huang, L. B.; Du, Y. Z.; Han, J. C.; Zheng, Q. Q.; Kong, J.; Hao, J. H. Self-Healing, Flexible, and Tailorable Triboelectric Nanogenerators for Self-Powered Sensors Based on Thermal Effect of Infrared Radiation. Adv. Funct. Mater. 2020, 30 (16), 1910723 DOI: 10.1002/adfm.201910723Google ScholarThere is no corresponding record for this reference.
- 13Lan, M. H.; Guan, X. X.; Zhu, D. Y.; Chen, Z. P.; Liu, T. S.; Tang, Z. H. Highly Elastic, Self-Healing, Recyclable Interlocking Double-Network Liquid-Free Ionic Conductive Elastomers via Facile Fabrication for Wearable Strain Sensors. ACS Appl. Mater. Interfaces 2023, 15 (15), 19447– 19458, DOI: 10.1021/acsami.3c01585Google ScholarThere is no corresponding record for this reference.
- 14Li, C. P.; Shi, Y. T.; Su, H. X.; Yang, Y. F.; Li, W.; Zhang, T.; Chen, W. Y.; Lin, R. J.; Li, Y. Z.; Liao, L. S. Mechanically Robust and Recyclable Siloxane Elastomers Enabled by Adjustable Dynamic Polymer Networks for Electronic Skin. Eur. Polym. J. 2023, 189, 111984 DOI: 10.1016/j.eurpolymj.2023.111984Google ScholarThere is no corresponding record for this reference.
- 15Liu, R. Y.; Lai, Y.; Li, S. X.; Wu, F.; Shao, J. M.; Liu, D.; Dong, X.; Wang, J.; Wang, Z. L. Ultrathin, Transparent, and Robust Self-Healing Electronic Skins for Tactile and Non-Contact Sensing. Nano Energy 2022, 95, 107056 DOI: 10.1016/j.nanoen.2022.107056Google ScholarThere is no corresponding record for this reference.
- 16Ma, J. X.; Sa, Z.; Zhang, H.; Feng, J. Y.; Wen, J. Y.; Wang, S.; Tian, Y. H. Microconfined Assembly of High-Resolution and Mechanically Robust EGaIn Liquid Metal Stretchable Electrodes for Wearable Electronic Systems. Adv. Sci. 2024, 11 (34), 2402818 DOI: 10.1002/advs.202402818Google ScholarThere is no corresponding record for this reference.
- 17Tian, H.; Lu, W. T.; Wang, C. Y.; Wang, R. H.; Zhou, P. L.; Fei, F.; Xu, M. Y.; Wang, J. C. Development of Highly Robust Polyurethane Elastomers Possessing Self-Healing Capabilities for Flexible Sensors. Mater. Horiz. 2025, 12 (11), 3949– 3964, DOI: 10.1039/D5MH00022JGoogle ScholarThere is no corresponding record for this reference.
- 18Wang, Y. M.; Wang, Y.; Yang, Y. Graphene–Polymer Nanocomposite-Based Redox-Induced Electricity for Flexible Self-Powered Strain Sensors. Adv. Energy Mater. 2018, 8 (22), 1800961 DOI: 10.1002/aenm.201800961Google ScholarThere is no corresponding record for this reference.
- 19Zhu, X. B.; Zhang, W. J.; Lu, G. M.; Zhao, H. C.; Wang, L. P. Ultrahigh Mechanical Strength and Robust Room-Temperature Self-Healing Properties of a Polyurethane–Graphene Oxide Network Resulting from Multiple Dynamic Bonds. ACS Nano 2022, 16 (10), 16724– 16735, DOI: 10.1021/acsnano.2c06264Google ScholarThere is no corresponding record for this reference.
- 20Bustero, I.; Gaztelumendi, I.; Obieta, I.; Mendizabal, M. A.; Zurutuza, A.; Ortega, A.; Alonso, B. Free-Standing Graphene Films Embedded in Epoxy Resin with Enhanced Thermal Properties. Adv. Compos. Hybrid Mater. 2020, 3 (1), 31– 40, DOI: 10.1007/s42114-020-00136-6Google ScholarThere is no corresponding record for this reference.
- 21Huang, L. Q.; Huang, H. X.; Yu, N.; Chen, C. Z.; Liu, Y.; Hu, G. H.; Du, J.; Zhao, H. High-Strength and Excellent Self-Healing Polyurethane Elastomer Based on Rigid Chain Segment Reinforcement. Macromolecules 2025, 58 (3), 1425– 1434, DOI: 10.1021/acs.macromol.5c00013Google ScholarThere is no corresponding record for this reference.
- 22Xu, Y. W.; Zhou, S.; Wu, Z. H.; Yang, X. Y.; Li, N.; Qin, Z. H.; Jiao, T. F. Room-Temperature Self-Healing and Recyclable Polyurethane Elastomers with High Strength and Superior Robustness Based on Dynamic Double-Crosslinked Structure. Chem. Eng. J. 2023, 466, 143179 DOI: 10.1016/j.cej.2023.143179Google ScholarThere is no corresponding record for this reference.
- 23Ekeocha, J.; Ellingford, C.; Pan, M.; Wemyss, A. M.; Bowen, C.; Wan, C. Y. Challenges and Opportunities of Self-Healing Polymers and Devices for Extreme and Hostile Environments. Adv. Mater. 2021, 33 (33), 2008052 DOI: 10.1002/adma.202008052Google ScholarThere is no corresponding record for this reference.
- 24He, C. L.; Liang, F. C.; Veeramuthu, L.; Cho, C. J.; Benas, J. S.; Tzeng, Y. R.; Tseng, Y. L.; Chen, W. C.; Rwei, A.; Kuo, C. C. Super Tough and Spontaneous Water-Assisted Autonomous Self-Healing Elastomer for Underwater Wearable Electronics. Adv. Sci. 2021, 8 (21), 2102275 DOI: 10.1002/advs.202102275Google ScholarThere is no corresponding record for this reference.
- 25Rodríguez-Albelo, L. M.; López-Maya, E.; Hamad, S.; Ruiz-Salvador, A. R.; Calero, S.; Navarro, J. A. R. Selective Sulfur Dioxide Adsorption on Crystal Defect Sites on an Isoreticular Metal–Organic Framework Series. Nat. Commun. 2017, 8, 14457, DOI: 10.1038/ncomms14457Google ScholarThere is no corresponding record for this reference.
- 26Tian, Y. S.; Wei, Y.; Wang, M.; Wang, J. D.; Li, X. F.; Qin, X.; Zhang, L. Q. Ultra-Stretchable, Tough, and Self-Healing Polyurethane with Tunable Microphase Separation for Flexible Wearable Electronics. Nano Energy 2025, 139, 110908 DOI: 10.1016/j.nanoen.2025.110908Google ScholarThere is no corresponding record for this reference.
- 27Li, M. M.; Lyu, Q.; Peng, B. L.; Chen, X. D.; Zhang, L. B.; Zhu, J. T. Bioinspired Colloidal Photonic Composites: Fabrication and Emerging Applications. Adv. Mater. 2022, 34 (52), 2110488 DOI: 10.1002/adma.202110488Google ScholarThere is no corresponding record for this reference.
- 28Nellepalli, P.; Patel, T.; Oh, J. K. Dynamic Covalent Polyurethane Network Materials: Synthesis and Self-Healability. Macromol. Rapid Commun. 2021, 42 (20), 2100391 DOI: 10.1002/marc.202100391Google ScholarThere is no corresponding record for this reference.
- 29Ren, Y. T.; Dong, X. Dynamic Polymeric Materials via Hydrogen-Bond Cross-Linking: Effect of Multiple Network Topologies. Prog. Polym. Sci. 2024, 158, 101890 DOI: 10.1016/j.progpolymsci.2024.101890Google ScholarThere is no corresponding record for this reference.
- 30Wang, Z. H.; Liu, B. W.; Zeng, F. R.; Lin, X. C.; Zhang, J. Y.; Wang, X. L.; Wang, Y. Z.; Zhao, H. B. Fully Recyclable Multifunctional Adhesive with High Durability, Transparency, Flame Retardancy, and Harsh-Environment Resistance. Sci. Adv. 2022, 8 (50), eadd8527 DOI: 10.1126/sciadv.add8527Google ScholarThere is no corresponding record for this reference.
- 31Xiang, Z. T.; Zhang, Y. A.; Lu, X. A Self-Healing Transparent Waterborne Polyurethane Film with High Strength and Toughness Based on Cation−π Interactions. ACS Appl. Mater. Interfaces 2024, 16 (51), 70948– 70962, DOI: 10.1021/acsami.4c18429Google ScholarThere is no corresponding record for this reference.
- 32Yang, J. W.; Bai, R. B.; Chen, B. H.; Suo, Z. G. Hydrogel Adhesion: A Supramolecular Synergy of Chemistry, Topology, and Mechanics. Adv. Funct. Mater. 2020, 30 (2), 1901693 DOI: 10.1002/adfm.201901693Google ScholarThere is no corresponding record for this reference.
- 33Yang, W. J.; Zhu, Y. L.; Liu, T. X.; Puglia, D.; Kenny, J. M.; Xu, P. W.; Zhang, R.; Ma, P. M. Multiple Structure Reconstruction by Dual Dynamic Crosslinking Strategy Inducing Self-Reinforcing and Toughening the Polyurethane/Nanocellulose Elastomers. Adv. Funct. Mater. 2023, 33 (12), 2213294 DOI: 10.1002/adfm.202213294Google ScholarThere is no corresponding record for this reference.
- 34Zhang, J. Y.; Yan, L. J.; Zhao, Y.; Su, Y. T.; Sun, J. Z.; Jiang, H. M.; Ma, T. L. A Multifunctional Supramolecular Polymer Binder with Hard/Soft Phase Interaction for Si-Based Lithium-Ion Batteries. Nano Energy 2024, 125, 109573 DOI: 10.1016/j.nanoen.2024.109573Google ScholarThere is no corresponding record for this reference.
- 35Chen, Y. L.; Kushner, A. M.; Williams, G. A.; Guan, Z. B. Multiphase Design of Autonomic Self-Healing Thermoplastic Elastomers. Nat. Chem. 2012, 4 (6), 467– 472, DOI: 10.1038/nchem.1314Google ScholarThere is no corresponding record for this reference.
- 36Yan, X. Z.; Liu, Z. Y.; Zhang, Q. H.; Lopez, J.; Wang, H.; Wu, H. C.; Niu, S. M.; Yan, H. P.; Wang, S. H.; Lei, T.; Li, J. H.; Qi, D. P.; Huang, P. G.; Huang, J. P.; Zhang, Y.; Wang, Y. Y.; Li, G. L.; Tok, J. B. H.; Chen, X. D.; Bao, Z. A. Quadruple H-Bonding Cross-Linked Supramolecular Polymeric Materials as Substrates for Stretchable, Antitearing, and Self-Healable Thin-Film Electrodes. J. Am. Chem. Soc. 2018, 140 (15), 5280– 5289, DOI: 10.1021/jacs.8b01682Google ScholarThere is no corresponding record for this reference.
- 37Jiang, H.; Yan, T.; Cheng, M.; Zhao, Z. H.; He, T. L.; Wang, Z. K.; Li, C. L.; Sun, S. Q.; Hu, S. Q. Autonomous Self-Healing and Superior Tough Polyurethane Elastomers Enabled by Strong and Highly Dynamic Hard Domains. Mater. Horiz. 2025, 12 (2), 599– 607, DOI: 10.1039/D4MH01129EGoogle ScholarThere is no corresponding record for this reference.
- 38Li, J. L.; Song, Y. H.; Song, G. J.; Li, Z. W.; Li, Z. J.; Yang, X. R.; Li, X. R. Room-Temperature Self-Healing Polyurethane with Superior Mechanical Properties Based on Supramolecular Structure for Flame-Retardant Application. Chem. Eng. J. 2024, 498, 155628 DOI: 10.1016/j.cej.2024.155628Google ScholarThere is no corresponding record for this reference.
- 39Hu, J.; Mo, R.; Jiang, X.; Sheng, X. X.; Zhang, X. Y. Toward Mechanical Robust yet Self-Healing Polyurethane Elastomers via Combination of Dynamic Main Chain and Dangling Quadruple Hydrogen Bonds. Polymer 2019, 183, 121912 DOI: 10.1016/j.polymer.2019.121912Google ScholarThere is no corresponding record for this reference.
- 40Wang, H.; Huang, J.; Liu, W.; Huang, J.; Yang, D.; Qiu, X.; Zhang, J. Tough and Fast Light-Controlled Healable Lignin-Containing Polyurethane Elastomers. Macromolecules 2022, 55 (19), 8629– 8641, DOI: 10.1021/acs.macromol.2c01401Google ScholarThere is no corresponding record for this reference.
- 41Li, Z. Q.; Feng, D. M.; Li, D. Z.; Zhou, L. X.; Ge, C. H.; Zhang, X. D. Synergistic Hydrogen Bonding Topology Enables Ultra-Robust Recyclable Polyurethane Elastomers for Multifunctional Strain Sensors. Small 2025, 21 (32), 2504828 DOI: 10.1002/smll.202504828Google ScholarThere is no corresponding record for this reference.
- 42Xu, X. B.; Ma, X. Z.; Wang, X. L.; Zhu, J.; Wang, J. G.; Yan, N.; Chen, J. Catalyst-Free Synthesis of Betulin-Based Polyurethane Elastomers with Outstanding Mechanical Properties and Solvent Resistance. ACS Appl. Polym. Mater. 2023, 5 (10), 8260– 8269, DOI: 10.1021/acsapm.3c01431Google ScholarThere is no corresponding record for this reference.
- 43Luo, Y. L.; Chen, J. L.; Situ, G.; Li, C. C.; Zhang, C. R.; Li, F. Z.; Li, C. H.; Luo, Z. Y.; Zhang, X. Aromatic Disulfide-Induced Self-Reinforcing Polyurethane Elastomer with Self-Healability. Chem. Eng. J. 2023, 469, 143958, DOI: 10.1016/j.cej.2023.143958Google ScholarThere is no corresponding record for this reference.
- 44Tayebi, P.; Asefnejad, A.; Khonakdar, H. A. Water-Based Polyurethane/Functionalized Chitosan/Zinc Oxide Nanoparticle Nanocomposites: Physical, Mechanical, and Biocompatibility Properties. Polym.-Plast. Technol. Mater. 2021, 60 (13), 1474– 1489, DOI: 10.1080/25740881.2021.1921206Google ScholarThere is no corresponding record for this reference.
- 45Nie, R. P.; Huang, H. D.; Yan, D. X.; Jia, L. C.; Lei, J.; Li, Z. M. Boosting the Actuation Performance of a Dynamic Supramolecular Polyurethane–Urea Elastomer via Kinetic Control. ACS Appl. Mater. Interfaces 2025, 17 (2), 3982– 3994, DOI: 10.1021/acsami.4c19128Google ScholarThere is no corresponding record for this reference.
- 46Zhang, B.; Pan, Z. S.; Li, W. M.; Zhao, Y. S.; Qin, X. L.; Li, A. Q.; Lv, M. L.; Qin, X. F.; Guo, W. L.; He, Z. C.; Wang, E. G. Natural Dextran as an Efficient Interfacial Passivator for ZnO-Based Electron-Transport Layers in Inverted Organic Solar Cells. Adv. Energy Mater. 2026, 16 (3), 2404297 DOI: 10.1002/aenm.202404297Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
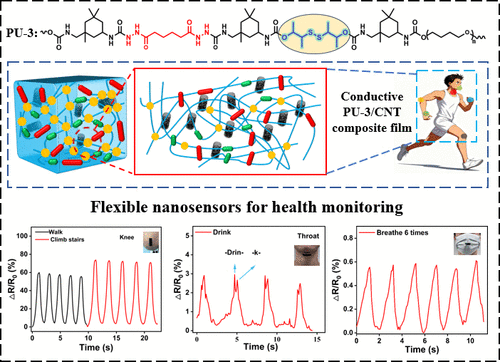
Figure 1

Figure 1. (a) Molecular structure of the PU-3 elastomer. (b) Schematic of the network structure and dynamic bond interactions. (c) Proposed network structure of the PU-3 elastomer.
Figure 2
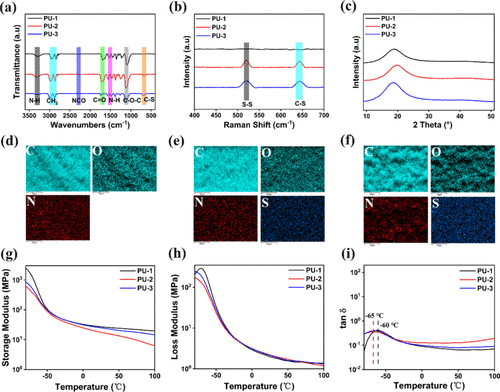
Figure 2. (a) FTIR spectra of PU-1, PU-2, and PU-3 elastomers. (b) Raman spectra. (c) XRD patterns. (d–f) Elemental mapping images of PU-1, PU-2, and PU-3 obtained from EDS. (g) Storage modulus (E′). (h) Loss modulus (E″). (i) Loss factor (tan δ).
Figure 3

Figure 3. (a) Stress–strain curves of the PU samples. (b) Toughness bar chart for different samples. (c, e) Cyclic tensile tests conducted at 500% strain for five consecutive loading–unloading cycles. (f) Performance comparison. (g) Fracture energy of PU-3. (h) Photograph of a notched PU-3 sample stretched to 800% strain.
Figure 4

Figure 4. (a, b) Stress–strain curves and bar chart of self-healing at various times at 60 °C for PU-1, (c, d) PU-2, and (e, f) PU-3, respectively. (g) Optical images of the self-healing process of PU-3. (h) Photograph of PU-3 after healing lifting a 5 kg weight.
Figure 5
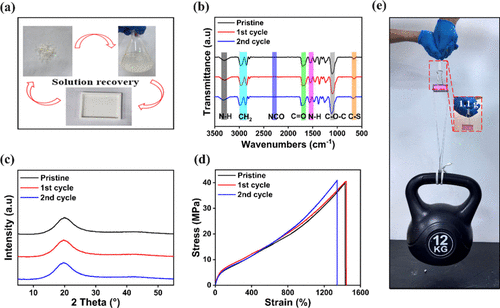
Figure 5. (a) Schematic illustration of the recycling process of the PU-3 elastomer. (b) FTIR spectra of the original and recycled PU-3 sample. (c) XRD patterns of the original and recycled PU-3 sample. (d) Stress–strain curves of PU-3 after two recycling cycles. (e) Demonstration of the recycled PU-3 elastomer lifting a 12 kg weight.
Figure 6
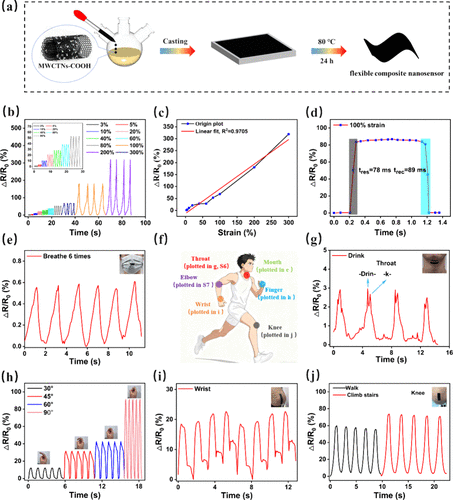
Figure 6. (a) Schematic illustration of the fabrication process of the flexible and sensitive polymer/carbon nanotube composite nanosensor. (b) Relative resistance variation (ΔR/R0) of the sensor under tensile strains ranging from 3% to 300%. (c) GF of the nanosensor, where GF = ΔR/(R0·ε) and ε denotes strain. (d) Response and recovery times of the sensor at 100% strain. (e) Real-time signal output of the sensor during mouth breathing. (f) Schematic representation of the potential applications in human motion monitoring. (g) Signal response of the sensor during throat articulation of the word “drink”. (h) Electrical response of the sensor during finger bending and stretching at angles of 30°, 45°, 60°, and 90°. (i) Signal output during wrist bending and (j) signal response during walking and stair climbing.
References
This article references 46 other publications.
- 1Alqaderi, A. J.; Ramakrishnan, N. Carbon-Based Flexible Strain Sensors: Recent Advances and Performance Insights in Human Motion Detection. Chem. Eng. J. 2025, 513, 162609 DOI: 10.1016/j.cej.2025.162609There is no corresponding record for this reference.
- 2Lin, M. Z.; Zheng, Z. J.; Yang, L.; Luo, M. S.; Fu, L. H.; Lin, B. F.; Xu, C. H. A High-Performance, Sensitive, Wearable Multifunctional Sensor Based on Rubber/CNT for Human Motion and Skin Temperature Detection. Adv. Mater. 2022, 34 (1), 2107309 DOI: 10.1002/adma.202107309There is no corresponding record for this reference.
- 3Tang, Y.; Fu, H.; Xu, B. G. Advanced Design of Triboelectric Nanogenerators for Future Eco-Smart Cities. Adv. Compos. Hybrid Mater. 2024, 7 (3), 102, DOI: 10.1007/s42114-024-00909-3There is no corresponding record for this reference.
- 4Wu, C. J.; Gao, X. Q.; Zhu, H.; Wang, X.; Wang, S. L.; Fu, Y.; Han, Y. T.; Yuan, W. N.; Tian, Y. L.; Jiang, X. C.; Lai, W. Y. Multiscale Stress Transmission Regulation toward Flexible Strain Sensors with High Stability. Adv. Funct. Mater. 2026, 36, 10852, DOI: 10.1002/adfm.202510852There is no corresponding record for this reference.
- 5Yadav, N.; Zhang, Q. Q.; Qi, D. F.; Yadav, A.; Zheng, H. Y. AI-Integrated Wearable Strain Sensors: Advances in E-Skin, Robotics, and Personalized Health Monitoring. Nanoscale Adv. 2025, 7 (16), 4803– 4819, DOI: 10.1039/D5NA00574DThere is no corresponding record for this reference.
- 6Lei, T. D.; Wang, Y. H.; Zhang, Q. S.; Wang, H. X.; Duan, X. R.; Yan, J.; Xia, Z. P.; Wang, R.; Shou, W.; Li, X. P.; Fan, J. Ultra-Stretchable and Anti-Freezing Ionic Conductive Hydrogels as High-Performance Strain Sensors and Flexible Triboelectric Nanogenerators in Extreme Environments. Nano Energy 2024, 126, 109633 DOI: 10.1016/j.nanoen.2024.109633There is no corresponding record for this reference.
- 7Maji, A.; Kuila, C.; Murmu, N. C.; Kuila, T. Stretch, Sense, and Innovate: Advances in Next-Generation Strain Sensors. Compos. Part B Eng. 2025, 306, 112749 DOI: 10.1016/j.compositesb.2025.112749There is no corresponding record for this reference.
- 8Padhan, A. K.; Sharma, D.; Thomas, T. S.; Sinha, A. P.; Mallick, A. N.; Mandal, D. Rapid Self-Healing and Superior Toughness in Ionically Crosslinked Polymer Ionogels and Strain Sensing Applications. J. Mater. Chem. A 2024, 12 (16), 9508– 9517, DOI: 10.1039/D3TA07277KThere is no corresponding record for this reference.
- 9Qu, J. T.; Cui, G. M.; Li, Z. K.; Fang, S. T.; Zhang, X. R.; Liu, A.; Han, M. Y.; Liu, H. D.; Wang, X. Q.; Wang, X. H. Advanced Flexible Sensing Technologies for Soft Robots. Adv. Funct. Mater. 2024, 34 (29), 2401311 DOI: 10.1002/adfm.202401311There is no corresponding record for this reference.
- 10Yu, S. J.; Ye, Q. Q.; Yang, B.; Liu, X. J.; Zhou, H.; Hu, L.; Lu, C. X. Ultrasensitive, Highly Stretchable, and Multifunctional Strain Sensors Based on Scorpion-Leg-Inspired Gradient Crack Arrays. Chem. Eng. J. 2024, 497, 154952 DOI: 10.1016/j.cej.2024.154952There is no corresponding record for this reference.
- 11Zhang, H. Y.; Yang, Q. N.; Xu, L. J.; Li, N.; Tan, H. H.; Du, J. J.; Yu, M. L.; Xu, J. X. Triboelectric Nanogenerators Based on Hydrated Lithium Ions Incorporated Double-Network Hydrogels for Biomechanical Sensing and Energy Harvesting at Low Temperature. Nano Energy 2024, 125, 109521 DOI: 10.1016/j.nanoen.2024.109521There is no corresponding record for this reference.
- 12Dai, X. Y.; Huang, L. B.; Du, Y. Z.; Han, J. C.; Zheng, Q. Q.; Kong, J.; Hao, J. H. Self-Healing, Flexible, and Tailorable Triboelectric Nanogenerators for Self-Powered Sensors Based on Thermal Effect of Infrared Radiation. Adv. Funct. Mater. 2020, 30 (16), 1910723 DOI: 10.1002/adfm.201910723There is no corresponding record for this reference.
- 13Lan, M. H.; Guan, X. X.; Zhu, D. Y.; Chen, Z. P.; Liu, T. S.; Tang, Z. H. Highly Elastic, Self-Healing, Recyclable Interlocking Double-Network Liquid-Free Ionic Conductive Elastomers via Facile Fabrication for Wearable Strain Sensors. ACS Appl. Mater. Interfaces 2023, 15 (15), 19447– 19458, DOI: 10.1021/acsami.3c01585There is no corresponding record for this reference.
- 14Li, C. P.; Shi, Y. T.; Su, H. X.; Yang, Y. F.; Li, W.; Zhang, T.; Chen, W. Y.; Lin, R. J.; Li, Y. Z.; Liao, L. S. Mechanically Robust and Recyclable Siloxane Elastomers Enabled by Adjustable Dynamic Polymer Networks for Electronic Skin. Eur. Polym. J. 2023, 189, 111984 DOI: 10.1016/j.eurpolymj.2023.111984There is no corresponding record for this reference.
- 15Liu, R. Y.; Lai, Y.; Li, S. X.; Wu, F.; Shao, J. M.; Liu, D.; Dong, X.; Wang, J.; Wang, Z. L. Ultrathin, Transparent, and Robust Self-Healing Electronic Skins for Tactile and Non-Contact Sensing. Nano Energy 2022, 95, 107056 DOI: 10.1016/j.nanoen.2022.107056There is no corresponding record for this reference.
- 16Ma, J. X.; Sa, Z.; Zhang, H.; Feng, J. Y.; Wen, J. Y.; Wang, S.; Tian, Y. H. Microconfined Assembly of High-Resolution and Mechanically Robust EGaIn Liquid Metal Stretchable Electrodes for Wearable Electronic Systems. Adv. Sci. 2024, 11 (34), 2402818 DOI: 10.1002/advs.202402818There is no corresponding record for this reference.
- 17Tian, H.; Lu, W. T.; Wang, C. Y.; Wang, R. H.; Zhou, P. L.; Fei, F.; Xu, M. Y.; Wang, J. C. Development of Highly Robust Polyurethane Elastomers Possessing Self-Healing Capabilities for Flexible Sensors. Mater. Horiz. 2025, 12 (11), 3949– 3964, DOI: 10.1039/D5MH00022JThere is no corresponding record for this reference.
- 18Wang, Y. M.; Wang, Y.; Yang, Y. Graphene–Polymer Nanocomposite-Based Redox-Induced Electricity for Flexible Self-Powered Strain Sensors. Adv. Energy Mater. 2018, 8 (22), 1800961 DOI: 10.1002/aenm.201800961There is no corresponding record for this reference.
- 19Zhu, X. B.; Zhang, W. J.; Lu, G. M.; Zhao, H. C.; Wang, L. P. Ultrahigh Mechanical Strength and Robust Room-Temperature Self-Healing Properties of a Polyurethane–Graphene Oxide Network Resulting from Multiple Dynamic Bonds. ACS Nano 2022, 16 (10), 16724– 16735, DOI: 10.1021/acsnano.2c06264There is no corresponding record for this reference.
- 20Bustero, I.; Gaztelumendi, I.; Obieta, I.; Mendizabal, M. A.; Zurutuza, A.; Ortega, A.; Alonso, B. Free-Standing Graphene Films Embedded in Epoxy Resin with Enhanced Thermal Properties. Adv. Compos. Hybrid Mater. 2020, 3 (1), 31– 40, DOI: 10.1007/s42114-020-00136-6There is no corresponding record for this reference.
- 21Huang, L. Q.; Huang, H. X.; Yu, N.; Chen, C. Z.; Liu, Y.; Hu, G. H.; Du, J.; Zhao, H. High-Strength and Excellent Self-Healing Polyurethane Elastomer Based on Rigid Chain Segment Reinforcement. Macromolecules 2025, 58 (3), 1425– 1434, DOI: 10.1021/acs.macromol.5c00013There is no corresponding record for this reference.
- 22Xu, Y. W.; Zhou, S.; Wu, Z. H.; Yang, X. Y.; Li, N.; Qin, Z. H.; Jiao, T. F. Room-Temperature Self-Healing and Recyclable Polyurethane Elastomers with High Strength and Superior Robustness Based on Dynamic Double-Crosslinked Structure. Chem. Eng. J. 2023, 466, 143179 DOI: 10.1016/j.cej.2023.143179There is no corresponding record for this reference.
- 23Ekeocha, J.; Ellingford, C.; Pan, M.; Wemyss, A. M.; Bowen, C.; Wan, C. Y. Challenges and Opportunities of Self-Healing Polymers and Devices for Extreme and Hostile Environments. Adv. Mater. 2021, 33 (33), 2008052 DOI: 10.1002/adma.202008052There is no corresponding record for this reference.
- 24He, C. L.; Liang, F. C.; Veeramuthu, L.; Cho, C. J.; Benas, J. S.; Tzeng, Y. R.; Tseng, Y. L.; Chen, W. C.; Rwei, A.; Kuo, C. C. Super Tough and Spontaneous Water-Assisted Autonomous Self-Healing Elastomer for Underwater Wearable Electronics. Adv. Sci. 2021, 8 (21), 2102275 DOI: 10.1002/advs.202102275There is no corresponding record for this reference.
- 25Rodríguez-Albelo, L. M.; López-Maya, E.; Hamad, S.; Ruiz-Salvador, A. R.; Calero, S.; Navarro, J. A. R. Selective Sulfur Dioxide Adsorption on Crystal Defect Sites on an Isoreticular Metal–Organic Framework Series. Nat. Commun. 2017, 8, 14457, DOI: 10.1038/ncomms14457There is no corresponding record for this reference.
- 26Tian, Y. S.; Wei, Y.; Wang, M.; Wang, J. D.; Li, X. F.; Qin, X.; Zhang, L. Q. Ultra-Stretchable, Tough, and Self-Healing Polyurethane with Tunable Microphase Separation for Flexible Wearable Electronics. Nano Energy 2025, 139, 110908 DOI: 10.1016/j.nanoen.2025.110908There is no corresponding record for this reference.
- 27Li, M. M.; Lyu, Q.; Peng, B. L.; Chen, X. D.; Zhang, L. B.; Zhu, J. T. Bioinspired Colloidal Photonic Composites: Fabrication and Emerging Applications. Adv. Mater. 2022, 34 (52), 2110488 DOI: 10.1002/adma.202110488There is no corresponding record for this reference.
- 28Nellepalli, P.; Patel, T.; Oh, J. K. Dynamic Covalent Polyurethane Network Materials: Synthesis and Self-Healability. Macromol. Rapid Commun. 2021, 42 (20), 2100391 DOI: 10.1002/marc.202100391There is no corresponding record for this reference.
- 29Ren, Y. T.; Dong, X. Dynamic Polymeric Materials via Hydrogen-Bond Cross-Linking: Effect of Multiple Network Topologies. Prog. Polym. Sci. 2024, 158, 101890 DOI: 10.1016/j.progpolymsci.2024.101890There is no corresponding record for this reference.
- 30Wang, Z. H.; Liu, B. W.; Zeng, F. R.; Lin, X. C.; Zhang, J. Y.; Wang, X. L.; Wang, Y. Z.; Zhao, H. B. Fully Recyclable Multifunctional Adhesive with High Durability, Transparency, Flame Retardancy, and Harsh-Environment Resistance. Sci. Adv. 2022, 8 (50), eadd8527 DOI: 10.1126/sciadv.add8527There is no corresponding record for this reference.
- 31Xiang, Z. T.; Zhang, Y. A.; Lu, X. A Self-Healing Transparent Waterborne Polyurethane Film with High Strength and Toughness Based on Cation−π Interactions. ACS Appl. Mater. Interfaces 2024, 16 (51), 70948– 70962, DOI: 10.1021/acsami.4c18429There is no corresponding record for this reference.
- 32Yang, J. W.; Bai, R. B.; Chen, B. H.; Suo, Z. G. Hydrogel Adhesion: A Supramolecular Synergy of Chemistry, Topology, and Mechanics. Adv. Funct. Mater. 2020, 30 (2), 1901693 DOI: 10.1002/adfm.201901693There is no corresponding record for this reference.
- 33Yang, W. J.; Zhu, Y. L.; Liu, T. X.; Puglia, D.; Kenny, J. M.; Xu, P. W.; Zhang, R.; Ma, P. M. Multiple Structure Reconstruction by Dual Dynamic Crosslinking Strategy Inducing Self-Reinforcing and Toughening the Polyurethane/Nanocellulose Elastomers. Adv. Funct. Mater. 2023, 33 (12), 2213294 DOI: 10.1002/adfm.202213294There is no corresponding record for this reference.
- 34Zhang, J. Y.; Yan, L. J.; Zhao, Y.; Su, Y. T.; Sun, J. Z.; Jiang, H. M.; Ma, T. L. A Multifunctional Supramolecular Polymer Binder with Hard/Soft Phase Interaction for Si-Based Lithium-Ion Batteries. Nano Energy 2024, 125, 109573 DOI: 10.1016/j.nanoen.2024.109573There is no corresponding record for this reference.
- 35Chen, Y. L.; Kushner, A. M.; Williams, G. A.; Guan, Z. B. Multiphase Design of Autonomic Self-Healing Thermoplastic Elastomers. Nat. Chem. 2012, 4 (6), 467– 472, DOI: 10.1038/nchem.1314There is no corresponding record for this reference.
- 36Yan, X. Z.; Liu, Z. Y.; Zhang, Q. H.; Lopez, J.; Wang, H.; Wu, H. C.; Niu, S. M.; Yan, H. P.; Wang, S. H.; Lei, T.; Li, J. H.; Qi, D. P.; Huang, P. G.; Huang, J. P.; Zhang, Y.; Wang, Y. Y.; Li, G. L.; Tok, J. B. H.; Chen, X. D.; Bao, Z. A. Quadruple H-Bonding Cross-Linked Supramolecular Polymeric Materials as Substrates for Stretchable, Antitearing, and Self-Healable Thin-Film Electrodes. J. Am. Chem. Soc. 2018, 140 (15), 5280– 5289, DOI: 10.1021/jacs.8b01682There is no corresponding record for this reference.
- 37Jiang, H.; Yan, T.; Cheng, M.; Zhao, Z. H.; He, T. L.; Wang, Z. K.; Li, C. L.; Sun, S. Q.; Hu, S. Q. Autonomous Self-Healing and Superior Tough Polyurethane Elastomers Enabled by Strong and Highly Dynamic Hard Domains. Mater. Horiz. 2025, 12 (2), 599– 607, DOI: 10.1039/D4MH01129EThere is no corresponding record for this reference.
- 38Li, J. L.; Song, Y. H.; Song, G. J.; Li, Z. W.; Li, Z. J.; Yang, X. R.; Li, X. R. Room-Temperature Self-Healing Polyurethane with Superior Mechanical Properties Based on Supramolecular Structure for Flame-Retardant Application. Chem. Eng. J. 2024, 498, 155628 DOI: 10.1016/j.cej.2024.155628There is no corresponding record for this reference.
- 39Hu, J.; Mo, R.; Jiang, X.; Sheng, X. X.; Zhang, X. Y. Toward Mechanical Robust yet Self-Healing Polyurethane Elastomers via Combination of Dynamic Main Chain and Dangling Quadruple Hydrogen Bonds. Polymer 2019, 183, 121912 DOI: 10.1016/j.polymer.2019.121912There is no corresponding record for this reference.
- 40Wang, H.; Huang, J.; Liu, W.; Huang, J.; Yang, D.; Qiu, X.; Zhang, J. Tough and Fast Light-Controlled Healable Lignin-Containing Polyurethane Elastomers. Macromolecules 2022, 55 (19), 8629– 8641, DOI: 10.1021/acs.macromol.2c01401There is no corresponding record for this reference.
- 41Li, Z. Q.; Feng, D. M.; Li, D. Z.; Zhou, L. X.; Ge, C. H.; Zhang, X. D. Synergistic Hydrogen Bonding Topology Enables Ultra-Robust Recyclable Polyurethane Elastomers for Multifunctional Strain Sensors. Small 2025, 21 (32), 2504828 DOI: 10.1002/smll.202504828There is no corresponding record for this reference.
- 42Xu, X. B.; Ma, X. Z.; Wang, X. L.; Zhu, J.; Wang, J. G.; Yan, N.; Chen, J. Catalyst-Free Synthesis of Betulin-Based Polyurethane Elastomers with Outstanding Mechanical Properties and Solvent Resistance. ACS Appl. Polym. Mater. 2023, 5 (10), 8260– 8269, DOI: 10.1021/acsapm.3c01431There is no corresponding record for this reference.
- 43Luo, Y. L.; Chen, J. L.; Situ, G.; Li, C. C.; Zhang, C. R.; Li, F. Z.; Li, C. H.; Luo, Z. Y.; Zhang, X. Aromatic Disulfide-Induced Self-Reinforcing Polyurethane Elastomer with Self-Healability. Chem. Eng. J. 2023, 469, 143958, DOI: 10.1016/j.cej.2023.143958There is no corresponding record for this reference.
- 44Tayebi, P.; Asefnejad, A.; Khonakdar, H. A. Water-Based Polyurethane/Functionalized Chitosan/Zinc Oxide Nanoparticle Nanocomposites: Physical, Mechanical, and Biocompatibility Properties. Polym.-Plast. Technol. Mater. 2021, 60 (13), 1474– 1489, DOI: 10.1080/25740881.2021.1921206There is no corresponding record for this reference.
- 45Nie, R. P.; Huang, H. D.; Yan, D. X.; Jia, L. C.; Lei, J.; Li, Z. M. Boosting the Actuation Performance of a Dynamic Supramolecular Polyurethane–Urea Elastomer via Kinetic Control. ACS Appl. Mater. Interfaces 2025, 17 (2), 3982– 3994, DOI: 10.1021/acsami.4c19128There is no corresponding record for this reference.
- 46Zhang, B.; Pan, Z. S.; Li, W. M.; Zhao, Y. S.; Qin, X. L.; Li, A. Q.; Lv, M. L.; Qin, X. F.; Guo, W. L.; He, Z. C.; Wang, E. G. Natural Dextran as an Efficient Interfacial Passivator for ZnO-Based Electron-Transport Layers in Inverted Organic Solar Cells. Adv. Energy Mater. 2026, 16 (3), 2404297 DOI: 10.1002/aenm.202404297There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsapm.5c04892.
Synthetic routes and characterization data; 1H NMR spectra, 13C NMR spectra, and mass spectrum of monomer; 1H NMR spectra of PUs; SEC test results of PUs; SEM characterization results of PUs; TGA and DSC data of PUs; cyclic tensile test results of PUs; signal output diagrams of throat vibration during the utterance of the Chinese word “apple” collected by the sensor; signal output diagrams of elbow flexion states collected by the sensor (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


