
Bi(benzimidazole)-Based Super-Electron-Donors in Redox Polymers as Battery Electrode MaterialsClick to copy article linkArticle link copied!
- Philipp PenertPhilipp PenertInstitute of Organic Chemistry II and Advanced Materials, Ulm University, Albert-Einstein-Allee 11, 89081 Ulm, GermanyCELEST Green Energy Lab Ulm, Ulm University, Lise-Meitner-Str. 16, 89081 Ulm, GermanyMore by Philipp Penert
- Bernd SchulzBernd SchulzInstitute of Organic Chemistry II and Advanced Materials, Ulm University, Albert-Einstein-Allee 11, 89081 Ulm, GermanyCELEST Green Energy Lab Ulm, Ulm University, Lise-Meitner-Str. 16, 89081 Ulm, GermanyMore by Bernd Schulz
- Axel FlorentAxel FlorentNantes Université, CNRS, Institut des Matériaux de Nantes Jean Rouxel, IMN, Nantes F-44000, FranceMore by Axel Florent
- Philippe PoizotPhilippe PoizotNantes Université, CNRS, Institut des Matériaux de Nantes Jean Rouxel, IMN, Nantes F-44000, FranceMore by Philippe Poizot
- Birgit Esser*Birgit Esser*Email: [email protected]. Web: https://www.esserlab.com.Institute of Organic Chemistry II and Advanced Materials, Ulm University, Albert-Einstein-Allee 11, 89081 Ulm, GermanyCELEST Green Energy Lab Ulm, Ulm University, Lise-Meitner-Str. 16, 89081 Ulm, GermanyMore by Birgit Esser
Abstract
Organic electrode-active materials (OAMs) represent an alternative to (transition-) metal-based materials used in conventional battery cells. Reversibly oxidizable p-type OAMs allow realization of full-organic battery cells operating in an anion-rocking-chair mechanism. In the search for p-type materials with a low redox potential, so-called super-electron-donors (SEDs) are a promising class of molecules. Herein, we incorporate a bi(benzimidazole) (BBI)-based SED into three polymers, PBBI, a conjugated homopolymer, PSBBI as a styrene-based side-chain polymer, and X-PSBBI as its cross-linked counterpart. Their properties as potential OAMs in lithium–organic half-cells were investigated, and PBBI was found to be electrochemically inactive. The side-chain polymers showed reversible cycling behavior in binder-free powder electrodes in LiBF4-based electrolytes with a low charge/discharge potential of 2.1 V vs Li+/0, even though the accessible capacity quickly faded. As a possible degradation mechanism, we propose decomposition via a dicarbene species as a plausible, reactive key species. This study showcases bi(benzimidazole)s as redox-active groups in OAMs with a low redox potential and provides insight into challenges associated with obtaining reversible cycling behavior in battery electrodes.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Introduction
Figure 1
Figure 1. P-Type RAGs and their redox potentials vs Li+/0. DHP and PT with high redox potentials and DMABP, Vio, and 1 with comparably low redox potentials. 1 is highlighted by an orange box as the herein investigated moiety.
Results and Discussion
Synthesis and Characterization of BBI-Polymers
Scheme 1
aReaction conditions: (a) 6-Bromo-1-methyl-1H-benzo[d]imidazole (2.5 equiv), MeCN, rflx., 15 h, 88%; (b) (i) KHMDS (0.5 m in toluene, 2.0 equiv), DMF/toluene (1:2), rt, 1 h; (ii) C2Cl6 (2.0 equiv), Et2O, rt, 15 min; and (iii) NaPF6 (2.8 equiv), MeOH/H2O (1:1), rt, 1 h, 58%; and (c) (i) [Ni(COD)2] (1.3 equiv), 2,2′-bipyridine (1.3 equiv), COD (1.0 equiv), DMF, 60 °C, 40 h; (ii) HClaq (1 M), rt, 2 h, 75%.
Scheme 2
aReaction conditions: (a) 1-Methyl-1H-benzo[d]imidazole, MeCN, rflx., 4 h, 56%; (b) 5, MeCN, rflx., 36 h, 87%; (c) (i) KHMDS, DMF/toluene, rt, 1 h; (ii) C2Cl6, toluene, rt, 30 min; and (iii) NaPF6, MeOH/H2O, rt, 1 h, 53%; (d) 5, MeCN, rflx., 4 d, 82%; and (e) (i) KHMDS, DMF/toluene, rt, 1 h; (ii) C2Cl6, toluene, rt, 30 min; and (iii) NaPF6, MeOH/H2O, rt, 1 h, 52%.
Scheme 3
Influence of Arylation of the BBI Core on the Electrochemical Properties
Figure 2
Figure 2. (a) Cyclic voltammograms of the salt of 1(PF6)2 (1 mM in DMF, top) and of monomer 8 (1 mM in DMSO, bottom) with 0.1 M n-Bu4NPF6; working electrode: glassy carbon (⌀ = 2 mm). (b) Expected redox reactions of SED 1 to its dication 12+ via radical cation 1•+. (c) Electrostatic potential maps of dicationic and neutral BBI 1 and a tolyl-substituted BBI (B3LYP/6–311+G(d), isovalue 0.02).
Electrochemical Investigations in Li Half-Cells
Figure 3
Figure 3. Electrochemical properties of PSBBI and X-PSBBI-based coated composite electrodes with 1M LiPF6 in EC/DMC 1:1 (v/v) as electrolyte. (a) Charge/discharge curves at a rate of 0.1C of PSBBI electrodes (PSBBI/Super C65/KB600/PVDF (50:20:20:10), active material mass loading: 0.621 mg). (b) Charge/discharge curves at a rate of 0.1C of X-PSBBI electrodes (X-PSBBI/Super C65/PVDF (30:60:10), active material mass loading: 0.291 mg) (all: coated on carbon-coated Al-foil, CE = RE = Li, separator: GF/D).
Figure 4
Figure 4. Electrochemical properties of PSBBI and X-PSBBI-based powder composite electrodes with 1 M LiBF4 in EC/DEC 1:1 (v/v) as electrolyte. (a) Charge/discharge curves and (b) the corresponding differential capacity plot of PSBBI electrodes (PSBBI:KB600 (2:1), active material mass loading: 5.54 mg). (c) Charge/discharge curves and (d) the corresponding differential capacity plot of X-PSBBI electrodes (X-PSBBI:KB600 (2:1), active material mass loading: 5.92 mg) (all: separator: GF/D; plotted cycles: 1–5, then every fifth).
Figure 5
Figure 5. (a) Excerpt of the 1H NMR spectra of (I) 12+, (II) the reduced and reoxidized 12+, and (III) the sample where 12+ was reduced to 1, more 12+ was added and the mixture was then reoxidized. Spectra recorded in DMSO-d6 (300 MHz, 300 K for (I); 400 MHz, 298 K for (II, III)). (b) Simplified reaction schemes for the decomposition experiment. (c) Potential formation of the bicarbene 1bc from the reduced SED 1, according to the Wanzlick equilibrium. (d) Galvanostatic cycling of a PSBBI powder composite electrode to high potential (1.8–3.5 V vs Li+/0) (PSBBI/KB600 (2:1), separator: GF/D, electrolyte: 1 M LiBF4 in EC/DEC 1:1 (v/v), active material mass loading: 5.81 mg); plotted cycles: 1–5, then every fifth.
Conclusions
Materials and Methods
Chemicals
Inert Working Procedures
Chromatography
Nuclear Magnetic Resonance Spectroscopy (NMR)
Elemental Analysis
Polymerization Reaction Yields
Cyclic Voltammetry in Solution
Electrochemical Measurements on Coated Composite Electrodes
Electrochemical Measurements on Binder-Free Powder Composite Electrodes
Electrolytes
Synthesis of the Polymers
Poly(2,12-(14,15-dimethyl-7,8,14,15-tetrahydro-6H-benzo[4,5]imidazo[1,2-a]benzo[4,5]-imidazo[2,1-c][1,4]diazepine-5,9-diium) Hexafluorophosphate(V)) (PBBI)
Poly(styryl-bisbenzimidazolium Hexafluorophosphate(V)) (PSBBI)
Poly(styryl-bisbenzimidazolium-stat-bisstyryl-bibenzimidazolium Hexafluorophosphate(V)) (X-PSBBI)
Data Availability
The data that support the findings of this study can be accessed on Zenodo (10.5281/zenodo.18399627).
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsapm.6c00515.
Additional Materials and Methods, including the calculation of the theoretical specific capacities and C-rates, synthetic procedures and characterization data of 1(PF6)2, 3, 4, 5, 6, 7, 8, 9, and 10, details on degradation experiments, NMR spectra, IR spectra, additional lithium half-cell measurements, and details on DFT calculations (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This work was realized in the framework of the ANR-DFG project ORGANION (N° ANR-20-CE92-0046-01) and was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Project IDs 446026621 (DFG-ANR project ORGANION), 390874152 (POLiS Cluster of Excellence, EXC 2154), 441236036 (SPP 2248, Polymer-based batteries), 445471097, and 445471845. It contributes to the research performed at CELEST (Center for Electrochemical Energy Storage Ulm-Karlsruhe). The authors acknowledge support by the state of Baden-Württemberg through bwHPC and the German Research Foundation (DFG) through grant no INST 40/575-1 FUGG (JUSTUS 2 cluster).
References
This article references 58 other publications.
- 1Esser, B.; Ehrenberg, H.; Fichtner, M.; Groß, A.; Janek, J. Post-Lithium Storage─Shaping the Future. Adv. Energy Mater. 2025, 15, 2402824 DOI: 10.1002/aenm.202402824Google ScholarThere is no corresponding record for this reference.
- 2Dai, H.; Guan, L.; Mao, M.; Wang, C. Evaluating the Present and Future of Organic Batteries. Nat. Rev. Clean Technol. 2025, 1 (7), 493– 510, DOI: 10.1038/s44359-025-00079-5Google ScholarThere is no corresponding record for this reference.
- 3Bitenc, J.; Pirnat, K.; Lužanin, O.; Dominko, R. Organic Cathodes, a Path toward Future Sustainable Batteries: Mirage or Realistic Future?. Chem. Mater. 2024, 36 (3), 1025– 1040, DOI: 10.1021/acs.chemmater.3c02408Google ScholarThere is no corresponding record for this reference.
- 4Lu, Y.; Chen, J. Prospects of Organic Electrode Materials for Practical Lithium Batteries. Nat. Rev. Chem. 2020, 4 (3), 127– 142, DOI: 10.1038/s41570-020-0160-9Google ScholarThere is no corresponding record for this reference.
- 5Xiao, J.; Huang, Y.; Ma, Y.; Li, C.; Fu, L.; Zeng, W.; Wang, X.; Li, X.; Wang, M.; Guo, B.; Lin, Y.; Cao, H. Organic Active Materials in Rechargeable Batteries: Recent Advances and Prospects. Energy Storage Mater. 2023, 63, 103046 DOI: 10.1016/j.ensm.2023.103046Google ScholarThere is no corresponding record for this reference.
- 6Poizot, P.; Gaubicher, J.; Renault, S.; Dubois, L.; Liang, Y.; Yao, Y. Opportunities and Challenges for Organic Electrodes in Electrochemical Energy Storage. Chem. Rev. 2020, 120, 6490– 6557, DOI: 10.1021/acs.chemrev.9b00482Google ScholarThere is no corresponding record for this reference.
- 7Poizot, P.; Dolhem, F. Clean Energy New Deal for a Sustainable World: From Non-CO2 Generating Energy Sources to Greener Electrochemical Storage Devices. Energy Environ. Sci. 2011, 4 (6), 2003– 2019, DOI: 10.1039/c0ee00731eGoogle ScholarThere is no corresponding record for this reference.
- 8Esser, B. Redox Polymers as Electrode-Active Materials for Batteries. Org. Mater 2019, 01 (1), 63– 70, DOI: 10.1055/s-0039-3401016Google ScholarThere is no corresponding record for this reference.
- 9Deuchert, K.; Hünig, S. Multistage Organic Redox Systems─A General Structural Principle. Angew. Chem., Int. Ed. 1978, 17 (12), 875– 886, DOI: 10.1002/anie.197808753Google ScholarThere is no corresponding record for this reference.
- 10Poizot, P.; Dolhem, F.; Gaubicher, J. Progress in All-Organic Rechargeable Batteries Using Cationic and Anionic Configurations: Toward Low-Cost and Greener Storage Solutions?. Curr. Opin. Electrochem. 2018, 9, 70– 80, DOI: 10.1016/j.coelec.2018.04.003Google ScholarThere is no corresponding record for this reference.
- 11Kim, J.; Kim, Y.; Yoo, J.; Kwon, G.; Ko, Y.; Kang, K. Organic Batteries for a Greener Rechargeable World. Nat. Rev. Mater. 2023, 8 (1), 54– 70, DOI: 10.1038/s41578-022-00478-1Google ScholarThere is no corresponding record for this reference.
- 12Yao, M.; Sano, H.; Ando, H.; Kiyobayashi, T. Molecular Ion Battery: A Rechargeable System without Using Any Elemental Ions as a Charge Carrier. Sci. Rep. 2015, 5 (1), 10962 DOI: 10.1038/srep10962Google ScholarThere is no corresponding record for this reference.
- 13Kye, H.; Kang, Y.; Jang, D.; Kwon, J. E.; Kim, B.-G. P-Type Redox-Active Organic Electrode Materials for Next-Generation Rechargeable Batteries. Adv. Energy Sustainability Res. 2022, 3 (8), 2200030 DOI: 10.1002/aesr.202200030Google ScholarThere is no corresponding record for this reference.
- 14Esser, B.; Morhenn, I. H.; Keis, M. Phenothiazine Polymers as Versatile Electrode Materials for Next-Generation Batteries. Acc. Mater. Res. 2025, 6 (6), 754– 764, DOI: 10.1021/accountsmr.5c00053Google ScholarThere is no corresponding record for this reference.
- 15Otteny, F.; Desmaizieres, G.; Esser, B. Phenothiazine-Based Redox Polymers for Energy Storage. In Redox Polymers for Energy and Nanomedicine, Polymer Chemistry Series; Royal Society of Chemistry, Chapter 5, 2020; pp 166– 197 DOI: 10.1039/9781788019743-00166 .Google ScholarThere is no corresponding record for this reference.
- 16Nishide, H.; Koshika, K.; Oyaizu, K. Environmentally benign batteries based on organic radical polymers. Pure Appl. Chem. 2009, 81 (11), 1961– 1970, DOI: 10.1351/PAC-CON-08-12-03Google ScholarThere is no corresponding record for this reference.
- 17Janoschka, T.; Hager, M. D.; Schubert, U. S. Powering up the Future: Radical Polymers for Battery Applications. Adv. Mater. 2012, 24 (48), 6397– 6409, DOI: 10.1002/adma.201203119Google ScholarThere is no corresponding record for this reference.
- 18Deunf, É.; Jiménez, P.; Guyomard, D.; Dolhem, F.; Poizot, P. A Dual–Ion Battery Using Diamino–Rubicene as Anion–Inserting Positive Electrode Material. Electrochem. Commun. 2016, 72, 64– 68, DOI: 10.1016/j.elecom.2016.09.002Google ScholarThere is no corresponding record for this reference.
- 19Wessling, R.; Koger, H.; Otteny, F.; Schmidt, M.; Semmelmaier, A.; Esser, B. Unlocking Twofold Oxidation in Phenothiazine Polymers for Application in Symmetric All-Organic Anionic Batteries. ACS Appl. Polym. Mater. 2024, 6 (14), 7956– 7968, DOI: 10.1021/acsapm.4c00500Google ScholarThere is no corresponding record for this reference.
- 20Wu, T.; Liu, Y.; Ye, J.; Chen, Y.; Dai, G.; Zhang, X.; Zhao, Y. High Performance Phenothiazine-Based Battery Materials Achieved by Synergistic Steric Blocking and Extended Conjugation. J. Power Sources 2024, 612, 234779 DOI: 10.1016/j.jpowsour.2024.234779Google ScholarThere is no corresponding record for this reference.
- 21Zhang, S.; Cai, J.; Li, H.; Xing, F.; Chen, L.; Wang, X.; He, X. Two-Electron Phenothiazine Based Cathode Achieved by Raising HOMO Energy Level for High Performance Lithium Organic Battery Adv. Energy Mater. 2025, 15 2403029 DOI: 10.1002/aenm.202403029 .Google ScholarThere is no corresponding record for this reference.
- 22Kowalski, J. A.; Casselman, M. D.; Kaur, A. P.; Milshtein, J. D.; Elliott, C. F.; Modekrutti, S.; Attanayake, N. H.; Zhang, N.; Parkin, S. R.; Risko, C.; Brushett, F. R.; Odom, S. A. A Stable Two-Electron-Donating Phenothiazine for Application in Nonaqueous Redox Flow Batteries. J. Mater. Chem. A 2017, 5 (46), 24371– 24379, DOI: 10.1039/C7TA05883GGoogle ScholarThere is no corresponding record for this reference.
- 23Acker, P.; Speer, M. E.; Wössner, J. S.; Esser, B. Azine-Based Polymers with a Two-Electron Redox Process as Cathode Materials for Organic Batteries. J. Mater. Chem. A 2020, 8 (22), 11195– 11201, DOI: 10.1039/D0TA04083EGoogle ScholarThere is no corresponding record for this reference.
- 24Fataj, X.; Achazi, A. J.; Stolze, C.; Muench, S.; Burges, R.; Anufriev, I.; Mignon, M.; Mollenhauer, D.; Nischang, I.; Hager, M. D.; Schubert, U. S. Pyridinium-Benzoxazole-Based Anode Material for Sustainable All-Organic Polymer-Based Batteries. ACS Appl. Energy Mater. 2025, 8, 4220– 4230, DOI: 10.1021/acsaem.4c03127Google ScholarThere is no corresponding record for this reference.
- 25Gao, G.; Wang, X.; Chen, L.; Baumgartner, T.; He, X. Conjugated Polymers with Benzoyl-N-Methylpyridinium Units: An Effective Design Strategy for High-Performance Lithium-Ion Batteries. Chem. Mater. 2021, 33 (12), 4596– 4605, DOI: 10.1021/acs.chemmater.1c01072Google ScholarThere is no corresponding record for this reference.
- 26He, X.; Chen, L.; Baumgartner, T. Modified Viologen- and Carbonylpyridinium-Based Electrodes for Organic Batteries. ACS Appl. Mater. Interfaces 2024, 16, 48689– 48705, DOI: 10.1021/acsami.3c09856Google ScholarThere is no corresponding record for this reference.
- 27Striepe, L.; Baumgartner, T. Viologens and Their Application as Functional Materials. Chem. – Eur. J. 2017, 23 (67), 16924– 16940, DOI: 10.1002/chem.201703348Google ScholarThere is no corresponding record for this reference.
- 28Wang, X.; Xue, W.; Gao, G.; Chen, L.; Baumgartner, T.; He, X. Bio-Derived 4-Electron-Accepting Carbonyl-N-Methylpyridinium Species for High-Performance Lithium-Organic Batteries. Cell Rep. Phys. Sci. 2022, 3 (7), 100951 DOI: 10.1016/j.xcrp.2022.100951Google ScholarThere is no corresponding record for this reference.
- 29Sevov, C. S.; Hendriks, K. H.; Sanford, M. S. Low-Potential Pyridinium Anolyte for Aqueous Redox Flow Batteries. J. Phys. Chem. C 2017, 121 (44), 24376– 24380, DOI: 10.1021/acs.jpcc.7b06247Google ScholarThere is no corresponding record for this reference.
- 30Sevov, C. S.; Hickey, D. P.; Cook, M. E.; Robinson, S. G.; Barnett, S.; Minteer, S. D.; Sigman, M. S.; Sanford, M. S. Physical Organic Approach to Persistent, Cyclable, Low-Potential Electrolytes for Flow Battery Applications. J. Am. Chem. Soc. 2017, 139 (8), 2924– 2927, DOI: 10.1021/jacs.7b00147Google ScholarThere is no corresponding record for this reference.
- 31Li, H.; Chen, L.; Xing, F.; Miao, H.; Zeng, J.; Zhang, S.; He, X. Cross-Linking Enhances the Performance of Four-Electron Carbonylpyridinium Based Polymers for Lithium Organic Batteries. Chem. Sci. 2024, 15 (35), 14399– 14405, DOI: 10.1039/D4SC04179HGoogle ScholarThere is no corresponding record for this reference.
- 32Bhosale, M.; Schmidt, C.; Penert, P.; Studer, G.; Esser, B. Anion-Rocking Chair Batteries with Tuneable Voltage Using Viologen- and Phenothiazine Polymer-Based Electrodes. ChemSusChem 2024, 17 (5), e202301143 DOI: 10.1002/cssc.202301143Google ScholarThere is no corresponding record for this reference.
- 33Murphy, J. A. Discovery and Development of Organic Super-Electron-Donors. J. Org. Chem. 2014, 79 (9), 3731– 3746, DOI: 10.1021/jo500071uGoogle ScholarThere is no corresponding record for this reference.
- 34Murphy, J. A.; Khan, T. A.; Zhou, S.; Thomson, D. W.; Mahesh, M. Highly Efficient Reduction of Unactivated Aryl and Alkyl Iodides by a Ground-State Neutral Organic Electron Donor. Angew. Chem., Int. Ed. 2005, 44 (9), 1356– 1360, DOI: 10.1002/anie.200462038Google ScholarThere is no corresponding record for this reference.
- 35Zhou, S.; Anderson, G. M.; Mondal, B.; Doni, E.; Ironmonger, V.; Kranz, M.; Tuttle, T.; Murphy, J. A. Organic Super-Electron-Donors: Initiators in Transition Metal-Free Haloarene–Arene Coupling. Chem. Sci. 2014, 5 (2), 476– 482, DOI: 10.1039/C3SC52315BGoogle ScholarThere is no corresponding record for this reference.
- 36Vaid, T. P.; Sanford, M. S. An Organic Super-Electron-Donor as a High Energy Density Negative Electrolyte for Nonaqueous Flow Batteries. Chem. Commun. 2019, 55 (74), 11037– 11040, DOI: 10.1039/C9CC06080DGoogle ScholarThere is no corresponding record for this reference.
- 37Alkhayri, F.; Dyker, C. A. A Two-Electron Bispyridinylidene Anolyte for Non-Aqueous Organic Redox Flow Batteries. J. Electrochem. Soc. 2020, 167 (16), 160548 DOI: 10.1149/1945-7111/abd492Google ScholarThere is no corresponding record for this reference.
- 38Rzesny, L.; Sajeevan, A. S.; Schmidt, C.; Lahr, J.; Lorenz, C.; Hermann, M.; Renault, S.; Poizot, P.; Esser, B. Super-Electron-Donor Polymers: From Monomer Design to Redox Functionality ChemRxiv 2025, DOI: 10.26434/chemrxiv-2025-h1fjg/v2 .Google ScholarThere is no corresponding record for this reference.
- 39Ames, J. R.; Houghtaling, M. A.; Terrian, D. L.; Mitchell, T. P. Annulated Derivatives of 2,2′-Biimidazole, 2-(2′-Imidazolyl)Benzimidazole, and 2,2′-Bibenzimidazole. Can. J. Chem. 1997, 75 (1), 28– 36, DOI: 10.1139/v97-004Google ScholarThere is no corresponding record for this reference.
- 40Murphy, J. A.; Zhou, S.; Thomson, D. W.; Schoenebeck, F.; Mahesh, M.; Park, S. R.; Tuttle, T.; Berlouis, L. E. A. The Generation of Aryl Anions by Double Electron Transfer to Aryl Iodides from a Neutral Ground-State Organic Super-Electron Donor. Angew. Chem., Int. Ed. 2007, 46 (27), 5178– 5183, DOI: 10.1002/anie.200700554Google ScholarThere is no corresponding record for this reference.
- 41Sword, R.; Baldwin, L. A.; Murphy, J. A. Fragmentations Observed in the Reactions of α-Methoxy-γ-Alkoxyalkyl Iodide Substrates with Super-Electron-Donors Derived from 4-DMAP and N-Methylbenzimidazole. Org. Biomol. Chem. 2011, 9 (9), 3560– 3570, DOI: 10.1039/c0ob01282cGoogle ScholarThere is no corresponding record for this reference.
- 42Acker, P.; Wössner, J. S.; Desmaizieres, G.; Esser, B. Conjugated Copolymer Design in Phenothiazine-Based Battery Materials Enables High Mass Loading Electrodes. ACS Sustainable Chem. Eng. 2022, 10 (10), 3236– 3244, DOI: 10.1021/acssuschemeng.1c07564Google ScholarThere is no corresponding record for this reference.
- 43Kolek, M.; Otteny, F.; Schmidt, P.; Mück-Lichtenfeld, C.; Einholz, C.; Becking, J.; Schleicher, E.; Winter, M.; Bieker, P.; Esser, B. Ultra-High Cycling Stability of Poly(Vinylphenothiazine) as a Battery Cathode Material Resulting from π–π Interactions. Energy Environ. Sci. 2017, 10 (11), 2334– 2341, DOI: 10.1039/C7EE01473BGoogle ScholarThere is no corresponding record for this reference.
- 44Otteny, F.; Perner, V.; Einholz, C.; Desmaizieres, G.; Schleicher, E.; Kolek, M.; Bieker, P.; Winter, M.; Esser, B. Bridging the Gap between Small Molecular π-Interactions and Their Effect on Phenothiazine-Based Redox Polymers in Organic Batteries. ACS Appl. Energy Mater. 2021, 4 (8), 7622– 7631, DOI: 10.1021/acsaem.1c00917Google ScholarThere is no corresponding record for this reference.
- 45Laoire, C. O.; Plichta, E.; Hendrickson, M.; Mukerjee, S.; Abraham, K. M. Electrochemical Studies of Ferrocene in a Lithium Ion Conducting Organic Carbonate Electrolyte. Electrochim. Acta 2009, 54 (26), 6560– 6564, DOI: 10.1016/j.electacta.2009.06.041Google ScholarThere is no corresponding record for this reference.
- 46Tengen, B.; Winkelmann, T.; Ortlieb, N.; Perner, V.; Studer, G.; Winter, M.; Esser, B.; Fischer, A.; Bieker, P. Immobilizing Poly(Vinylphenothiazine) in Ketjenblack-Based Electrodes to Access Its Full Specific Capacity as Battery Electrode Material. Adv. Funct. Mater. 2023, 33 (9), 2210512 DOI: 10.1002/adfm.202210512Google ScholarThere is no corresponding record for this reference.
- 47Adil, M.; Schmidt, M.; Vogt, J.; Diemant, T.; Oschatz, M.; Esser, B. Mitigating Dissolution to Enhance the Performance of Pillar[5]Quinone in Sodium Batteries. Batteries & Supercaps 2024, 7 (12), e202400312 DOI: 10.1002/batt.202400312Google ScholarThere is no corresponding record for this reference.
- 48Sudant, G.; Baudrin, E.; Dunn, B.; Tarascon, J.-M. Synthesis and Electrochemical Properties of Vanadium Oxide Aerogels Prepared by a Freeze-Drying Process. J. Electrochem. Soc. 2004, 151 (5), A666 DOI: 10.1149/1.1687427Google ScholarThere is no corresponding record for this reference.
- 49Larcher, D.; Masquelier, C.; Bonnin, D.; Chabre, Y.; Masson, V.; Leriche, J.-B.; Tarascon, J.-M. Effect of Particle Size on Lithium Intercalation into α-Fe2O3. J. Electrochem. Soc. 2003, 150 (1), A133 DOI: 10.1149/1.1528941Google ScholarThere is no corresponding record for this reference.
- 50Ue, M. Ionic Radius of (CF3SO2)3C─and Applicability of Stokes Law to Its Propylene Carbonate Solution. J. Electrochem. Soc. 1996, 143 (11), L270 DOI: 10.1149/1.1837231Google ScholarThere is no corresponding record for this reference.
- 51Merschel, A.; Rottschäfer, D.; Neumann, B.; Stammler, H.-G.; Ringenberg, M.; van Gastel, M.; Demirer, T. I.; Andrada, D. M.; Ghadwal, R. S. Crystalline Anions Based on Classical N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2023, 62 (5), e202215244 DOI: 10.1002/anie.202215244Google ScholarThere is no corresponding record for this reference.
- 52Rottschäfer, D.; Neumann, B.; Stammler, H.-G.; van Gastel, M.; Andrada, D. M.; Ghadwal, R. S. Crystalline Radicals Derived from Classical N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2018, 57 (17), 4765– 4768, DOI: 10.1002/anie.201801596Google ScholarThere is no corresponding record for this reference.
- 53Liu, Y.; Lindner, P. E.; Lemal, D. M. Thermodynamics of a Diaminocarbene–Tetraaminoethylene Equilibrium. J. Am. Chem. Soc. 1999, 121 (45), 10626– 10627, DOI: 10.1021/ja9922678Google ScholarThere is no corresponding record for this reference.
- 54Wanzlick, H. W. Aspects of Nucleophilic Carbene Chemistry. Angew. Chem. Int. Ed. 1962, 1 (2), 75– 80, DOI: 10.1002/anie.196200751Google ScholarThere is no corresponding record for this reference.
- 55Böhm, V. P. W.; Herrmann, W. A. The “Wanzlick Equilibrium”. Angew. Chem. Int. Ed. 2000, 39 (22), 4036– 4038, DOI: 10.1002/1521-3773(20001117)39:22%253C4036:AID-ANIE4036%253E3.0.CO;2-LGoogle ScholarThere is no corresponding record for this reference.
- 56Messelberger, J.; Kumar, M.; Goodner, S. J.; Munz, D. Wanzlick’s Equilibrium in Tri- and Tetraaminoolefins. Org. Chem. Front. 2021, 8 (23), 6663– 6669, DOI: 10.1039/D1QO01320CGoogle ScholarThere is no corresponding record for this reference.
- 57Gorodetsky, B.; Ramnial, T.; Branda, N. R.; Clyburne, J. A. C. Electrochemical Reduction of an Imidazolium Cation: A Convenient Preparation of Imidazol-2-Ylidenes and Their Observation in an Ionic Liquid. Chem. Commun. 2004, 1972– 1973, DOI: 10.1039/B407386JGoogle ScholarThere is no corresponding record for this reference.
- 58Ramnial, T.; McKenzie, I.; Gorodetsky, B.; Tsang, E. M. W.; Clyburne, J. A. C. Reactions of N-Heterocyclic Carbenes (NHCs) with One-Electron Oxidants: Possible Formation of a Carbene Cation Radical. Chem. Commun. 2004, 1054– 1055, DOI: 10.1039/B314110AGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. P-Type RAGs and their redox potentials vs Li+/0. DHP and PT with high redox potentials and DMABP, Vio, and 1 with comparably low redox potentials. 1 is highlighted by an orange box as the herein investigated moiety.
Scheme 1
 Scheme 1. Synthesis of the Homopolymer Poly(bi(benzimidazole)) (PBBI) Starting from 1,3-Diiodopropanea
Scheme 1. Synthesis of the Homopolymer Poly(bi(benzimidazole)) (PBBI) Starting from 1,3-DiiodopropaneaaReaction conditions: (a) 6-Bromo-1-methyl-1H-benzo[d]imidazole (2.5 equiv), MeCN, rflx., 15 h, 88%; (b) (i) KHMDS (0.5 m in toluene, 2.0 equiv), DMF/toluene (1:2), rt, 1 h; (ii) C2Cl6 (2.0 equiv), Et2O, rt, 15 min; and (iii) NaPF6 (2.8 equiv), MeOH/H2O (1:1), rt, 1 h, 58%; and (c) (i) [Ni(COD)2] (1.3 equiv), 2,2′-bipyridine (1.3 equiv), COD (1.0 equiv), DMF, 60 °C, 40 h; (ii) HClaq (1 M), rt, 2 h, 75%.
Scheme 2
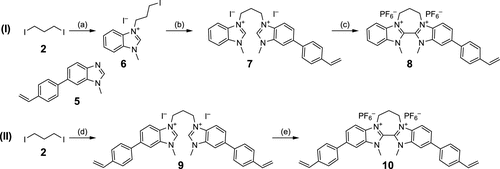 Scheme 2. Synthesis of the Styryl-BBI Monomer (I) and Cross-linker (II)a
Scheme 2. Synthesis of the Styryl-BBI Monomer (I) and Cross-linker (II)aaReaction conditions: (a) 1-Methyl-1H-benzo[d]imidazole, MeCN, rflx., 4 h, 56%; (b) 5, MeCN, rflx., 36 h, 87%; (c) (i) KHMDS, DMF/toluene, rt, 1 h; (ii) C2Cl6, toluene, rt, 30 min; and (iii) NaPF6, MeOH/H2O, rt, 1 h, 53%; (d) 5, MeCN, rflx., 4 d, 82%; and (e) (i) KHMDS, DMF/toluene, rt, 1 h; (ii) C2Cl6, toluene, rt, 30 min; and (iii) NaPF6, MeOH/H2O, rt, 1 h, 52%.
Scheme 3
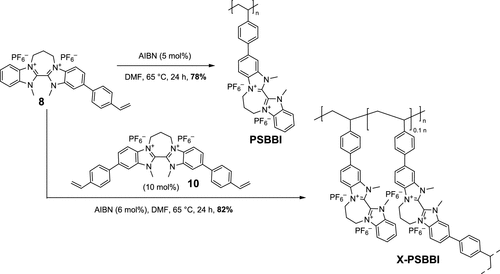 Scheme 3. Synthesis of the Linear and Cross-linked Polymers PSBBI and X-PSBBI
Scheme 3. Synthesis of the Linear and Cross-linked Polymers PSBBI and X-PSBBIFigure 2
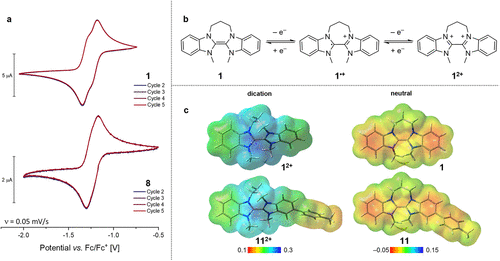
Figure 2. (a) Cyclic voltammograms of the salt of 1(PF6)2 (1 mM in DMF, top) and of monomer 8 (1 mM in DMSO, bottom) with 0.1 M n-Bu4NPF6; working electrode: glassy carbon (⌀ = 2 mm). (b) Expected redox reactions of SED 1 to its dication 12+ via radical cation 1•+. (c) Electrostatic potential maps of dicationic and neutral BBI 1 and a tolyl-substituted BBI (B3LYP/6–311+G(d), isovalue 0.02).
Figure 3
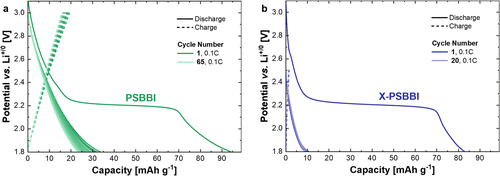
Figure 3. Electrochemical properties of PSBBI and X-PSBBI-based coated composite electrodes with 1M LiPF6 in EC/DMC 1:1 (v/v) as electrolyte. (a) Charge/discharge curves at a rate of 0.1C of PSBBI electrodes (PSBBI/Super C65/KB600/PVDF (50:20:20:10), active material mass loading: 0.621 mg). (b) Charge/discharge curves at a rate of 0.1C of X-PSBBI electrodes (X-PSBBI/Super C65/PVDF (30:60:10), active material mass loading: 0.291 mg) (all: coated on carbon-coated Al-foil, CE = RE = Li, separator: GF/D).
Figure 4

Figure 4. Electrochemical properties of PSBBI and X-PSBBI-based powder composite electrodes with 1 M LiBF4 in EC/DEC 1:1 (v/v) as electrolyte. (a) Charge/discharge curves and (b) the corresponding differential capacity plot of PSBBI electrodes (PSBBI:KB600 (2:1), active material mass loading: 5.54 mg). (c) Charge/discharge curves and (d) the corresponding differential capacity plot of X-PSBBI electrodes (X-PSBBI:KB600 (2:1), active material mass loading: 5.92 mg) (all: separator: GF/D; plotted cycles: 1–5, then every fifth).
Figure 5

Figure 5. (a) Excerpt of the 1H NMR spectra of (I) 12+, (II) the reduced and reoxidized 12+, and (III) the sample where 12+ was reduced to 1, more 12+ was added and the mixture was then reoxidized. Spectra recorded in DMSO-d6 (300 MHz, 300 K for (I); 400 MHz, 298 K for (II, III)). (b) Simplified reaction schemes for the decomposition experiment. (c) Potential formation of the bicarbene 1bc from the reduced SED 1, according to the Wanzlick equilibrium. (d) Galvanostatic cycling of a PSBBI powder composite electrode to high potential (1.8–3.5 V vs Li+/0) (PSBBI/KB600 (2:1), separator: GF/D, electrolyte: 1 M LiBF4 in EC/DEC 1:1 (v/v), active material mass loading: 5.81 mg); plotted cycles: 1–5, then every fifth.
References
This article references 58 other publications.
- 1Esser, B.; Ehrenberg, H.; Fichtner, M.; Groß, A.; Janek, J. Post-Lithium Storage─Shaping the Future. Adv. Energy Mater. 2025, 15, 2402824 DOI: 10.1002/aenm.202402824There is no corresponding record for this reference.
- 2Dai, H.; Guan, L.; Mao, M.; Wang, C. Evaluating the Present and Future of Organic Batteries. Nat. Rev. Clean Technol. 2025, 1 (7), 493– 510, DOI: 10.1038/s44359-025-00079-5There is no corresponding record for this reference.
- 3Bitenc, J.; Pirnat, K.; Lužanin, O.; Dominko, R. Organic Cathodes, a Path toward Future Sustainable Batteries: Mirage or Realistic Future?. Chem. Mater. 2024, 36 (3), 1025– 1040, DOI: 10.1021/acs.chemmater.3c02408There is no corresponding record for this reference.
- 4Lu, Y.; Chen, J. Prospects of Organic Electrode Materials for Practical Lithium Batteries. Nat. Rev. Chem. 2020, 4 (3), 127– 142, DOI: 10.1038/s41570-020-0160-9There is no corresponding record for this reference.
- 5Xiao, J.; Huang, Y.; Ma, Y.; Li, C.; Fu, L.; Zeng, W.; Wang, X.; Li, X.; Wang, M.; Guo, B.; Lin, Y.; Cao, H. Organic Active Materials in Rechargeable Batteries: Recent Advances and Prospects. Energy Storage Mater. 2023, 63, 103046 DOI: 10.1016/j.ensm.2023.103046There is no corresponding record for this reference.
- 6Poizot, P.; Gaubicher, J.; Renault, S.; Dubois, L.; Liang, Y.; Yao, Y. Opportunities and Challenges for Organic Electrodes in Electrochemical Energy Storage. Chem. Rev. 2020, 120, 6490– 6557, DOI: 10.1021/acs.chemrev.9b00482There is no corresponding record for this reference.
- 7Poizot, P.; Dolhem, F. Clean Energy New Deal for a Sustainable World: From Non-CO2 Generating Energy Sources to Greener Electrochemical Storage Devices. Energy Environ. Sci. 2011, 4 (6), 2003– 2019, DOI: 10.1039/c0ee00731eThere is no corresponding record for this reference.
- 8Esser, B. Redox Polymers as Electrode-Active Materials for Batteries. Org. Mater 2019, 01 (1), 63– 70, DOI: 10.1055/s-0039-3401016There is no corresponding record for this reference.
- 9Deuchert, K.; Hünig, S. Multistage Organic Redox Systems─A General Structural Principle. Angew. Chem., Int. Ed. 1978, 17 (12), 875– 886, DOI: 10.1002/anie.197808753There is no corresponding record for this reference.
- 10Poizot, P.; Dolhem, F.; Gaubicher, J. Progress in All-Organic Rechargeable Batteries Using Cationic and Anionic Configurations: Toward Low-Cost and Greener Storage Solutions?. Curr. Opin. Electrochem. 2018, 9, 70– 80, DOI: 10.1016/j.coelec.2018.04.003There is no corresponding record for this reference.
- 11Kim, J.; Kim, Y.; Yoo, J.; Kwon, G.; Ko, Y.; Kang, K. Organic Batteries for a Greener Rechargeable World. Nat. Rev. Mater. 2023, 8 (1), 54– 70, DOI: 10.1038/s41578-022-00478-1There is no corresponding record for this reference.
- 12Yao, M.; Sano, H.; Ando, H.; Kiyobayashi, T. Molecular Ion Battery: A Rechargeable System without Using Any Elemental Ions as a Charge Carrier. Sci. Rep. 2015, 5 (1), 10962 DOI: 10.1038/srep10962There is no corresponding record for this reference.
- 13Kye, H.; Kang, Y.; Jang, D.; Kwon, J. E.; Kim, B.-G. P-Type Redox-Active Organic Electrode Materials for Next-Generation Rechargeable Batteries. Adv. Energy Sustainability Res. 2022, 3 (8), 2200030 DOI: 10.1002/aesr.202200030There is no corresponding record for this reference.
- 14Esser, B.; Morhenn, I. H.; Keis, M. Phenothiazine Polymers as Versatile Electrode Materials for Next-Generation Batteries. Acc. Mater. Res. 2025, 6 (6), 754– 764, DOI: 10.1021/accountsmr.5c00053There is no corresponding record for this reference.
- 15Otteny, F.; Desmaizieres, G.; Esser, B. Phenothiazine-Based Redox Polymers for Energy Storage. In Redox Polymers for Energy and Nanomedicine, Polymer Chemistry Series; Royal Society of Chemistry, Chapter 5, 2020; pp 166– 197 DOI: 10.1039/9781788019743-00166 .There is no corresponding record for this reference.
- 16Nishide, H.; Koshika, K.; Oyaizu, K. Environmentally benign batteries based on organic radical polymers. Pure Appl. Chem. 2009, 81 (11), 1961– 1970, DOI: 10.1351/PAC-CON-08-12-03There is no corresponding record for this reference.
- 17Janoschka, T.; Hager, M. D.; Schubert, U. S. Powering up the Future: Radical Polymers for Battery Applications. Adv. Mater. 2012, 24 (48), 6397– 6409, DOI: 10.1002/adma.201203119There is no corresponding record for this reference.
- 18Deunf, É.; Jiménez, P.; Guyomard, D.; Dolhem, F.; Poizot, P. A Dual–Ion Battery Using Diamino–Rubicene as Anion–Inserting Positive Electrode Material. Electrochem. Commun. 2016, 72, 64– 68, DOI: 10.1016/j.elecom.2016.09.002There is no corresponding record for this reference.
- 19Wessling, R.; Koger, H.; Otteny, F.; Schmidt, M.; Semmelmaier, A.; Esser, B. Unlocking Twofold Oxidation in Phenothiazine Polymers for Application in Symmetric All-Organic Anionic Batteries. ACS Appl. Polym. Mater. 2024, 6 (14), 7956– 7968, DOI: 10.1021/acsapm.4c00500There is no corresponding record for this reference.
- 20Wu, T.; Liu, Y.; Ye, J.; Chen, Y.; Dai, G.; Zhang, X.; Zhao, Y. High Performance Phenothiazine-Based Battery Materials Achieved by Synergistic Steric Blocking and Extended Conjugation. J. Power Sources 2024, 612, 234779 DOI: 10.1016/j.jpowsour.2024.234779There is no corresponding record for this reference.
- 21Zhang, S.; Cai, J.; Li, H.; Xing, F.; Chen, L.; Wang, X.; He, X. Two-Electron Phenothiazine Based Cathode Achieved by Raising HOMO Energy Level for High Performance Lithium Organic Battery Adv. Energy Mater. 2025, 15 2403029 DOI: 10.1002/aenm.202403029 .There is no corresponding record for this reference.
- 22Kowalski, J. A.; Casselman, M. D.; Kaur, A. P.; Milshtein, J. D.; Elliott, C. F.; Modekrutti, S.; Attanayake, N. H.; Zhang, N.; Parkin, S. R.; Risko, C.; Brushett, F. R.; Odom, S. A. A Stable Two-Electron-Donating Phenothiazine for Application in Nonaqueous Redox Flow Batteries. J. Mater. Chem. A 2017, 5 (46), 24371– 24379, DOI: 10.1039/C7TA05883GThere is no corresponding record for this reference.
- 23Acker, P.; Speer, M. E.; Wössner, J. S.; Esser, B. Azine-Based Polymers with a Two-Electron Redox Process as Cathode Materials for Organic Batteries. J. Mater. Chem. A 2020, 8 (22), 11195– 11201, DOI: 10.1039/D0TA04083EThere is no corresponding record for this reference.
- 24Fataj, X.; Achazi, A. J.; Stolze, C.; Muench, S.; Burges, R.; Anufriev, I.; Mignon, M.; Mollenhauer, D.; Nischang, I.; Hager, M. D.; Schubert, U. S. Pyridinium-Benzoxazole-Based Anode Material for Sustainable All-Organic Polymer-Based Batteries. ACS Appl. Energy Mater. 2025, 8, 4220– 4230, DOI: 10.1021/acsaem.4c03127There is no corresponding record for this reference.
- 25Gao, G.; Wang, X.; Chen, L.; Baumgartner, T.; He, X. Conjugated Polymers with Benzoyl-N-Methylpyridinium Units: An Effective Design Strategy for High-Performance Lithium-Ion Batteries. Chem. Mater. 2021, 33 (12), 4596– 4605, DOI: 10.1021/acs.chemmater.1c01072There is no corresponding record for this reference.
- 26He, X.; Chen, L.; Baumgartner, T. Modified Viologen- and Carbonylpyridinium-Based Electrodes for Organic Batteries. ACS Appl. Mater. Interfaces 2024, 16, 48689– 48705, DOI: 10.1021/acsami.3c09856There is no corresponding record for this reference.
- 27Striepe, L.; Baumgartner, T. Viologens and Their Application as Functional Materials. Chem. – Eur. J. 2017, 23 (67), 16924– 16940, DOI: 10.1002/chem.201703348There is no corresponding record for this reference.
- 28Wang, X.; Xue, W.; Gao, G.; Chen, L.; Baumgartner, T.; He, X. Bio-Derived 4-Electron-Accepting Carbonyl-N-Methylpyridinium Species for High-Performance Lithium-Organic Batteries. Cell Rep. Phys. Sci. 2022, 3 (7), 100951 DOI: 10.1016/j.xcrp.2022.100951There is no corresponding record for this reference.
- 29Sevov, C. S.; Hendriks, K. H.; Sanford, M. S. Low-Potential Pyridinium Anolyte for Aqueous Redox Flow Batteries. J. Phys. Chem. C 2017, 121 (44), 24376– 24380, DOI: 10.1021/acs.jpcc.7b06247There is no corresponding record for this reference.
- 30Sevov, C. S.; Hickey, D. P.; Cook, M. E.; Robinson, S. G.; Barnett, S.; Minteer, S. D.; Sigman, M. S.; Sanford, M. S. Physical Organic Approach to Persistent, Cyclable, Low-Potential Electrolytes for Flow Battery Applications. J. Am. Chem. Soc. 2017, 139 (8), 2924– 2927, DOI: 10.1021/jacs.7b00147There is no corresponding record for this reference.
- 31Li, H.; Chen, L.; Xing, F.; Miao, H.; Zeng, J.; Zhang, S.; He, X. Cross-Linking Enhances the Performance of Four-Electron Carbonylpyridinium Based Polymers for Lithium Organic Batteries. Chem. Sci. 2024, 15 (35), 14399– 14405, DOI: 10.1039/D4SC04179HThere is no corresponding record for this reference.
- 32Bhosale, M.; Schmidt, C.; Penert, P.; Studer, G.; Esser, B. Anion-Rocking Chair Batteries with Tuneable Voltage Using Viologen- and Phenothiazine Polymer-Based Electrodes. ChemSusChem 2024, 17 (5), e202301143 DOI: 10.1002/cssc.202301143There is no corresponding record for this reference.
- 33Murphy, J. A. Discovery and Development of Organic Super-Electron-Donors. J. Org. Chem. 2014, 79 (9), 3731– 3746, DOI: 10.1021/jo500071uThere is no corresponding record for this reference.
- 34Murphy, J. A.; Khan, T. A.; Zhou, S.; Thomson, D. W.; Mahesh, M. Highly Efficient Reduction of Unactivated Aryl and Alkyl Iodides by a Ground-State Neutral Organic Electron Donor. Angew. Chem., Int. Ed. 2005, 44 (9), 1356– 1360, DOI: 10.1002/anie.200462038There is no corresponding record for this reference.
- 35Zhou, S.; Anderson, G. M.; Mondal, B.; Doni, E.; Ironmonger, V.; Kranz, M.; Tuttle, T.; Murphy, J. A. Organic Super-Electron-Donors: Initiators in Transition Metal-Free Haloarene–Arene Coupling. Chem. Sci. 2014, 5 (2), 476– 482, DOI: 10.1039/C3SC52315BThere is no corresponding record for this reference.
- 36Vaid, T. P.; Sanford, M. S. An Organic Super-Electron-Donor as a High Energy Density Negative Electrolyte for Nonaqueous Flow Batteries. Chem. Commun. 2019, 55 (74), 11037– 11040, DOI: 10.1039/C9CC06080DThere is no corresponding record for this reference.
- 37Alkhayri, F.; Dyker, C. A. A Two-Electron Bispyridinylidene Anolyte for Non-Aqueous Organic Redox Flow Batteries. J. Electrochem. Soc. 2020, 167 (16), 160548 DOI: 10.1149/1945-7111/abd492There is no corresponding record for this reference.
- 38Rzesny, L.; Sajeevan, A. S.; Schmidt, C.; Lahr, J.; Lorenz, C.; Hermann, M.; Renault, S.; Poizot, P.; Esser, B. Super-Electron-Donor Polymers: From Monomer Design to Redox Functionality ChemRxiv 2025, DOI: 10.26434/chemrxiv-2025-h1fjg/v2 .There is no corresponding record for this reference.
- 39Ames, J. R.; Houghtaling, M. A.; Terrian, D. L.; Mitchell, T. P. Annulated Derivatives of 2,2′-Biimidazole, 2-(2′-Imidazolyl)Benzimidazole, and 2,2′-Bibenzimidazole. Can. J. Chem. 1997, 75 (1), 28– 36, DOI: 10.1139/v97-004There is no corresponding record for this reference.
- 40Murphy, J. A.; Zhou, S.; Thomson, D. W.; Schoenebeck, F.; Mahesh, M.; Park, S. R.; Tuttle, T.; Berlouis, L. E. A. The Generation of Aryl Anions by Double Electron Transfer to Aryl Iodides from a Neutral Ground-State Organic Super-Electron Donor. Angew. Chem., Int. Ed. 2007, 46 (27), 5178– 5183, DOI: 10.1002/anie.200700554There is no corresponding record for this reference.
- 41Sword, R.; Baldwin, L. A.; Murphy, J. A. Fragmentations Observed in the Reactions of α-Methoxy-γ-Alkoxyalkyl Iodide Substrates with Super-Electron-Donors Derived from 4-DMAP and N-Methylbenzimidazole. Org. Biomol. Chem. 2011, 9 (9), 3560– 3570, DOI: 10.1039/c0ob01282cThere is no corresponding record for this reference.
- 42Acker, P.; Wössner, J. S.; Desmaizieres, G.; Esser, B. Conjugated Copolymer Design in Phenothiazine-Based Battery Materials Enables High Mass Loading Electrodes. ACS Sustainable Chem. Eng. 2022, 10 (10), 3236– 3244, DOI: 10.1021/acssuschemeng.1c07564There is no corresponding record for this reference.
- 43Kolek, M.; Otteny, F.; Schmidt, P.; Mück-Lichtenfeld, C.; Einholz, C.; Becking, J.; Schleicher, E.; Winter, M.; Bieker, P.; Esser, B. Ultra-High Cycling Stability of Poly(Vinylphenothiazine) as a Battery Cathode Material Resulting from π–π Interactions. Energy Environ. Sci. 2017, 10 (11), 2334– 2341, DOI: 10.1039/C7EE01473BThere is no corresponding record for this reference.
- 44Otteny, F.; Perner, V.; Einholz, C.; Desmaizieres, G.; Schleicher, E.; Kolek, M.; Bieker, P.; Winter, M.; Esser, B. Bridging the Gap between Small Molecular π-Interactions and Their Effect on Phenothiazine-Based Redox Polymers in Organic Batteries. ACS Appl. Energy Mater. 2021, 4 (8), 7622– 7631, DOI: 10.1021/acsaem.1c00917There is no corresponding record for this reference.
- 45Laoire, C. O.; Plichta, E.; Hendrickson, M.; Mukerjee, S.; Abraham, K. M. Electrochemical Studies of Ferrocene in a Lithium Ion Conducting Organic Carbonate Electrolyte. Electrochim. Acta 2009, 54 (26), 6560– 6564, DOI: 10.1016/j.electacta.2009.06.041There is no corresponding record for this reference.
- 46Tengen, B.; Winkelmann, T.; Ortlieb, N.; Perner, V.; Studer, G.; Winter, M.; Esser, B.; Fischer, A.; Bieker, P. Immobilizing Poly(Vinylphenothiazine) in Ketjenblack-Based Electrodes to Access Its Full Specific Capacity as Battery Electrode Material. Adv. Funct. Mater. 2023, 33 (9), 2210512 DOI: 10.1002/adfm.202210512There is no corresponding record for this reference.
- 47Adil, M.; Schmidt, M.; Vogt, J.; Diemant, T.; Oschatz, M.; Esser, B. Mitigating Dissolution to Enhance the Performance of Pillar[5]Quinone in Sodium Batteries. Batteries & Supercaps 2024, 7 (12), e202400312 DOI: 10.1002/batt.202400312There is no corresponding record for this reference.
- 48Sudant, G.; Baudrin, E.; Dunn, B.; Tarascon, J.-M. Synthesis and Electrochemical Properties of Vanadium Oxide Aerogels Prepared by a Freeze-Drying Process. J. Electrochem. Soc. 2004, 151 (5), A666 DOI: 10.1149/1.1687427There is no corresponding record for this reference.
- 49Larcher, D.; Masquelier, C.; Bonnin, D.; Chabre, Y.; Masson, V.; Leriche, J.-B.; Tarascon, J.-M. Effect of Particle Size on Lithium Intercalation into α-Fe2O3. J. Electrochem. Soc. 2003, 150 (1), A133 DOI: 10.1149/1.1528941There is no corresponding record for this reference.
- 50Ue, M. Ionic Radius of (CF3SO2)3C─and Applicability of Stokes Law to Its Propylene Carbonate Solution. J. Electrochem. Soc. 1996, 143 (11), L270 DOI: 10.1149/1.1837231There is no corresponding record for this reference.
- 51Merschel, A.; Rottschäfer, D.; Neumann, B.; Stammler, H.-G.; Ringenberg, M.; van Gastel, M.; Demirer, T. I.; Andrada, D. M.; Ghadwal, R. S. Crystalline Anions Based on Classical N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2023, 62 (5), e202215244 DOI: 10.1002/anie.202215244There is no corresponding record for this reference.
- 52Rottschäfer, D.; Neumann, B.; Stammler, H.-G.; van Gastel, M.; Andrada, D. M.; Ghadwal, R. S. Crystalline Radicals Derived from Classical N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2018, 57 (17), 4765– 4768, DOI: 10.1002/anie.201801596There is no corresponding record for this reference.
- 53Liu, Y.; Lindner, P. E.; Lemal, D. M. Thermodynamics of a Diaminocarbene–Tetraaminoethylene Equilibrium. J. Am. Chem. Soc. 1999, 121 (45), 10626– 10627, DOI: 10.1021/ja9922678There is no corresponding record for this reference.
- 54Wanzlick, H. W. Aspects of Nucleophilic Carbene Chemistry. Angew. Chem. Int. Ed. 1962, 1 (2), 75– 80, DOI: 10.1002/anie.196200751There is no corresponding record for this reference.
- 55Böhm, V. P. W.; Herrmann, W. A. The “Wanzlick Equilibrium”. Angew. Chem. Int. Ed. 2000, 39 (22), 4036– 4038, DOI: 10.1002/1521-3773(20001117)39:22%253C4036:AID-ANIE4036%253E3.0.CO;2-LThere is no corresponding record for this reference.
- 56Messelberger, J.; Kumar, M.; Goodner, S. J.; Munz, D. Wanzlick’s Equilibrium in Tri- and Tetraaminoolefins. Org. Chem. Front. 2021, 8 (23), 6663– 6669, DOI: 10.1039/D1QO01320CThere is no corresponding record for this reference.
- 57Gorodetsky, B.; Ramnial, T.; Branda, N. R.; Clyburne, J. A. C. Electrochemical Reduction of an Imidazolium Cation: A Convenient Preparation of Imidazol-2-Ylidenes and Their Observation in an Ionic Liquid. Chem. Commun. 2004, 1972– 1973, DOI: 10.1039/B407386JThere is no corresponding record for this reference.
- 58Ramnial, T.; McKenzie, I.; Gorodetsky, B.; Tsang, E. M. W.; Clyburne, J. A. C. Reactions of N-Heterocyclic Carbenes (NHCs) with One-Electron Oxidants: Possible Formation of a Carbene Cation Radical. Chem. Commun. 2004, 1054– 1055, DOI: 10.1039/B314110AThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsapm.6c00515.
Additional Materials and Methods, including the calculation of the theoretical specific capacities and C-rates, synthetic procedures and characterization data of 1(PF6)2, 3, 4, 5, 6, 7, 8, 9, and 10, details on degradation experiments, NMR spectra, IR spectra, additional lithium half-cell measurements, and details on DFT calculations (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


