Designing Anode-Electrolyte Interfaces for Low-Temperature Lithium- and Sodium-Ion Batteries: Challenges and StrategiesClick to copy article linkArticle link copied!
- Jeong-A LeeJeong-A LeeDepartment of Chemical and Biomolecular Engineering, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Republic of KoreaMore by Jeong-A Lee
- Chaeeun SongChaeeun SongDepartment of Chemical and Biomolecular Engineering, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Republic of KoreaMore by Chaeeun Song
- Seung Hee HanSeung Hee HanDepartment of Chemical and Biomolecular Engineering, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Republic of KoreaMore by Seung Hee Han
- Boguen KimBoguen KimDepartment of Chemical and Biomolecular Engineering, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Republic of KoreaMore by Boguen Kim
- Nam-Soon Choi*Nam-Soon Choi*E-mail: [email protected]Department of Chemical and Biomolecular Engineering, Korea Advanced Institute of Science and Technology (KAIST), 291 Daehak-ro, Yuseong-gu, Daejeon 34141, Republic of KoreaMore by Nam-Soon Choi
Abstract
Lithium-ion and sodium-ion batteries are gaining prominence as energy storage platforms for extreme environments, particularly at low temperatures. However, the prevailing assumption of electrochemical similarity between Li+ and Na+ in the conventional design paradigm has prevented exploration of their distinct low-temperature degradation pathways, which are intensified by hindered interfacial ion transport at the anode. Herein, we present the differences in solvation structures, desolvation kinetics, and ion-transport mechanisms across the solid-electrolyte interphase (SEI) between Li+ and Na+ at low temperatures. While lithium-ion systems are constrained by sluggish desolvation kinetics, sodium-ion systems face severe interfacial resistance arising from inhomogeneous SEI compositions and weakened interactions between Na+ ions and SEI species. Recognizing these ion-specific interfacial bottlenecks, we propose electrolyte design strategies that enhance ion transport and interfacial stability at the anode. These insights provide rational frameworks for developing next-generation alkali-ion batteries capable of overcoming the distinct challenges posed by low-temperature operation.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of ACS Energy Letters special issue “The Evolving Landscape of Energy Research: Insights from Leading Researchers”.
Figure 1
Figure 1. Schematic illustration of challenges encountered in (a) Li-ion and (b) Na-ion batteries under low-temperature conditions.
Interfacial Challenges Underlying Anode Instability at Low Temperatures
Figure 2
Figure 2. (a) Correlation between the absolute |ESPmin| values of solvent species and the desolvation energy of Li+ ion. Reproduced with permission from ref (23). Copyright 2013 The Electrochemical Society. (b) Comparative desolvation energies of different cation species. (c) Correlation between the coordination number of cations and the solvation power depending on the functional groups. (d) Effect of operating temperature on the solvation environment and SEI structure. Reproduced with permission from ref (35). Copyright 2024 Wiley-VCH. (e) Temperature-dependent bond-breaking modes and corresponding changes in SEI composition at low temperatures. Reproduced with permission from ref (37). Copyright 2023 Springer Nature.
Electrolyte Design Strategies to Overcome Low-Temperature Performance Limits
Figure 3
Figure 3. (a) Schematics of internal limitations and strategies for regulation of solvation structures for low-temperature LIBs. (b) Solvent diagram of DN versus dielectric constant. Solvents located in the blue-colored zone are denoted as soft solvents characterized by a lower DN and moderate dielectric constant, which reduce the Li+-solvent interactions without compromising ionic conductivity, where ε indicates the dielectric constant of solvents. (c) Electrolyte screening via the maximum and minimum values of the ESP surfaces. Reproduced with permission from ref (44). Copyright 2024 American Chemical Society. (d) Selection of deshielding anions based on the relative 7Li chemical shift and compatibility with graphite anodes. Reproduced with permission from ref (35). Copyright 2024 Wiley-VCH. (e) Transport barriers and ionic conductivities of SEI components at room temperature. (36,47−49) (f) Ionic conductivity of SEI as a function of grain size, where η and σ indicate viscosity and ionic conductivity, respectively. Reproduced with permission from ref (51). Copyright 2025 Wiley-VCH.
Figure 4
Figure 4. (a) Schematic of conditions required for low-temperature operation with strongly solvating electrolyte, weakly solvating electrolyte, and hybrid solvating electrolyte. (b) Solvent coordination number and charge-transfer activation energy with strongly solvating solvent, weakly solvating solvent, and hybrid solvating solvent. (c) Solvent-derived SEI formation and slow Na+ transport through inhomogeneous organic-rich SEI. (d) Anion-derived SEI formation and fast Na+ transport through uniform inorganic-rich SEI.
Summary and Outlook
Figure 5
Figure 5. Challenges for achieving suitable low-temperature battery performance and electrolyte design strategies. HFGA: hexafluoroglutaric anhydride, TMSP: tris(trimethylsilyl) phosphate, TMSB: tris(trimethylsilyl) borate, DTD: 1,3,2-dioxathiolane 2,2-dioxide.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c02918.
Coordination number of electrolytes, chemical structures, and full names of solvents and anions (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Biographies
Jeong-A Lee
Jeong-A Lee is a Ph.D. candidate in the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST), South Korea, under the supervision of Prof. Nam-Soon Choi. Her research focuses on the development of functional electrolyte additives for lithium–metal batteries and anode-free lithium–metal batteries.
Chaeeun Song
Chaeeun Song is a Ph.D. candidate at the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST), South Korea, under the supervision of Prof. Nam-Soon Choi. Her research focuses on the development of functional electrolytes for lithium-ion and lithium–metal batteries.
Seung Hee Han
Seung Hee Han is a Ph.D. candidate at the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST), South Korea, under the supervision of Prof. Nam-Soon Choi. Her research focuses on the development of functional electrolytes for lithium-ion batteries and Zn–Br batteries.
Boguen Kim
Boguen Kim is a Ph.D. candidate in the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST), South Korea, under the supervision of Prof. Nam-Soon Choi. His research focuses on the development of electrolytes for lithium–metal batteries.
Nam-Soon Choi
Nam-Soon Choi is a Professor in the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST) in South Korea. Her research focuses on the mechanistic studies of electrode–electrolyte interfaces and the design of tailored electrolyte systems for next-generation rechargeable batteries (https://surfchem.kaist.ac.kr/).
Acknowledgments
This research was supported by the Global TOP Strategic Research Center Project through the Ministry of Science and ICT (MSIT), the National Research Council of Science and Technology (NST) (GTL24012-000), and a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. RS-2024-00427700).
References
This article references 84 other publications.
- 1Chen, S.; Hu, T.; Yu, T.; Luo, X.; Zhang, L.; Li, F. Structural Feature Design for Carbon Materials toward Sodium Storage: Insights and Prospects. ACS Energy Lett. 2025, 10 (4), 1931– 1952, DOI: 10.1021/acsenergylett.5c00231Google ScholarThere is no corresponding record for this reference.
- 2Shi, Y.; Hu, E.; Sumboja, A.; Anggraningrum, I. T.; Syahrial, A. Z.; Yan, Q. Layered 3d Transition Metal-Based Oxides for Sodium-Ion and Lithium-Ion Batteries: Differences, Links and Beyond. Adv. Funct. Mater. 2025, 35 (2), 2413078, DOI: 10.1002/adfm.202413078Google ScholarThere is no corresponding record for this reference.
- 3Nekahi, A.; Dorri, M.; Rezaei, M.; Bouguern, M. D.; Madikere Raghunatha Reddy, A. K.; Li, X.; Deng, S.; Zaghib, K. Comparative Issues of Metal-Ion Batteries toward Sustainable Energy Storage: Lithium vs. Sodium. Batteries 2024, 10 (8), 279, DOI: 10.3390/batteries10080279Google ScholarThere is no corresponding record for this reference.
- 4Tan, S.; Shadike, Z.; Cai, X.; Lin, R.; Kludze, A.; Borodin, O.; Lucht, B. L.; Wang, C.; Hu, E.; Xu, K.; Yang, X.-Q. Review on Low-Temperature Electrolytes for Lithium-Ion and Lithium Metal Batteries. Electrochem. Energy Rev. 2023, 6 (1), 35, DOI: 10.1007/s41918-023-00199-1Google ScholarThere is no corresponding record for this reference.
- 5Goktas, M.; Bolli, C.; Berg, E. J.; Novák, P.; Pollok, K.; Langenhorst, F.; Roeder, M. v.; Lenchuk, O.; Mollenhauer, D.; Adelhelm, P. Graphite as Cointercalation Electrode for Sodium-Ion Batteries: Electrode Dynamics and The Missing Solid Electrolyte Interphase (SEI). Adv. Energy Mater. 2018, 8 (16), 1702724, DOI: 10.1002/aenm.201702724Google ScholarThere is no corresponding record for this reference.
- 6Sun, N.; Qiu, J.; Xu, B. Understanding of Sodium Storage Mechanism in Hard Carbons: Ongoing Development Under Debate. Adv. Energy Mater. 2022, 12 (27), 2200715, DOI: 10.1002/aenm.202200715Google ScholarThere is no corresponding record for this reference.
- 7Irisarri, E.; Ponrouch, A.; Palacin, M. Hard Carbon Negative Electrode Materials for Sodium-Ion Batteries. J. Electrochem. Soc. 2015, 162 (14), A2476, DOI: 10.1149/2.0091514jesGoogle ScholarThere is no corresponding record for this reference.
- 8Xia, J. L.; Yan, D.; Guo, L. P.; Dong, X. L.; Li, W. C.; Lu, A. H. Hard Carbon Nanosheets with Uniform Ultramicropores and Accessible Functional Groups Showing High Realistic Capacity and Superior Rate Performance for Sodium-Ion Storage. Adv. Mater. 2020, 32 (21), 2000447, DOI: 10.1002/adma.202000447Google ScholarThere is no corresponding record for this reference.
- 9Hong, S. Y.; Kim, Y.; Park, Y.; Choi, A.; Choi, N.-S.; Lee, K. T. Charge Carriers in Rechargeable Batteries: Na Ions vs. Li Ions. Energy Environ. Sci. 2013, 6 (7), 2067– 2081, DOI: 10.1039/c3ee40811fGoogle ScholarThere is no corresponding record for this reference.
- 10Jache, B.; Binder, J. O.; Abe, T.; Adelhelm, P. A Comparative Study on The Impact of Different Glymes and Their Derivatives as Electrolyte Solvents for Graphite Co-Intercalation Electrodes in Lithium-Ion and Sodium-Ion Batteries. Phys. Chem. Chem. Phys. 2016, 18 (21), 14299– 14316, DOI: 10.1039/C6CP00651EGoogle ScholarThere is no corresponding record for this reference.
- 11Cresce, A. V.; Russell, S. M.; Borodin, O.; Allen, J. A.; Schroeder, M. A.; Dai, M.; Peng, J.; Gobet, M. P.; Greenbaum, S. G.; Rogers, R. E.; Xu, K. Solvation Behavior of Carbonate-Based Electrolytes in Sodium Ion Batteries. Phys. Chem. Chem. Phys. 2017, 19 (1), 574– 586, DOI: 10.1039/C6CP07215AGoogle ScholarThere is no corresponding record for this reference.
- 12Huang, Z.; Xiao, Z.; Jin, R.; Li, Z.; Shu, C.; Shi, R.; Wang, X.; Tang, Z.; Tang, W.; Wu, Y. A Comprehensive Review on Liquid Electrolyte Design for Low-Temperature Lithium/Sodium Metal Batteries. Energy Environ. Sci. 2024, 17 (15), 5365– 5386, DOI: 10.1039/D4EE02060JGoogle ScholarThere is no corresponding record for this reference.
- 13Shanbedi, M.; Shahali, H.; Polycarpou, A. A.; Amiri, A. Advances and Future Prospects of Low-Temperature Electrolytes for Lithium-Ion Batteries. EES Batteries. 2025, 1, 385– 426, DOI: 10.1039/D5EB00013KGoogle ScholarThere is no corresponding record for this reference.
- 14Jeong, D.; Tackett, B. M.; Pol, V. G. Tailored Li-Ion Battery Electrodes and Electrolytes for Extreme Condition Operation. Commun. Chem. 2025, 8, 170, DOI: 10.1038/s42004-025-01564-5Google ScholarThere is no corresponding record for this reference.
- 15Su, Q.; Hu, C.; Li, S.; Huang, W.; Dong, J.; Song, R.; Xu, L.; Fang, G. Sodium-Ion Batteries at Low Temperature: Storage Mechanism and Modification Strategies. Chin. Chem. Lett. 2025, 111267, DOI: 10.1016/j.cclet.2025.111267Google ScholarThere is no corresponding record for this reference.
- 16Li, M.; Zhuo, H.; Jing, Q.; Gu, Y.; Liao, Z.; Wang, K.; Hu, J.; Geng, D.; Sun, X.; Xiao, B. Low-Temperature Performance of Na-Ion Batteries. Carbon Energy 2024, 6 (10), e546 DOI: 10.1002/cey2.546Google ScholarThere is no corresponding record for this reference.
- 17Bai, Z.; Yao, Q.; Wang, M.; Meng, W.; Dou, S.; Liu, H. K.; Wang, N. Low-Temperature Sodium-Ion Batteries: Challenges and Progress. Adv. Energy Mater. 2024, 14, 2303788, DOI: 10.1002/aenm.202303788Google ScholarThere is no corresponding record for this reference.
- 18Qiu, X.; Chen, Y.; Sun, Y.; Wang, Y.; Liang, Z.; Zhou, G.; Xue, Y.; Shi, L.; Jiang, J.; Kong, X.; Zhuang, Q.; Ju, Z. Research on Low-Temperature Sodium-Ion Batteries: Challenges, Strategies and Prospect. Energy Storage Mater. 2024, 72, 103760, DOI: 10.1016/j.ensm.2024.103760Google ScholarThere is no corresponding record for this reference.
- 19Yang, S.; Cheng, K.; Cao, Z. Status and Strategies of Electrolyte Engineering for Low-Temperature Sodium-Ion Batteries. J. Mater. Chem. A 2024, 12 (22), 13059– 13080, DOI: 10.1039/D4TA01400FGoogle ScholarThere is no corresponding record for this reference.
- 20Zhang, S.; Xu, K.; Jow, T. The Low Temperature Performance of Li-Ion Batteries. J. Power Sources 2003, 115 (1), 137– 140, DOI: 10.1016/S0378-7753(02)00618-3Google ScholarThere is no corresponding record for this reference.
- 21Abe, T.; Fukuda, H.; Iriyama, Y.; Ogumi, Z. Solvated Li-Ion Transfer at Interface between Graphite and Electrolyte. J. Electrochem. Soc. 2004, 151 (8), A1120, DOI: 10.1149/1.1763141Google ScholarThere is no corresponding record for this reference.
- 22Xu, K.; von Cresce, A.; Lee, U. Differentiating Contributions to “Ion Transfer” Barrier from Interphasial Resistance and Li+ Desolvation at Electrolyte/Graphite Interface. Langmuir 2010, 26 (13), 11538– 11543, DOI: 10.1021/la1009994Google ScholarThere is no corresponding record for this reference.
- 23Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical Analysis on De-Solvation of Lithium, Sodium, and Magnesium Cations to Organic Electrolyte Solvents. J. Electrochem. Soc. 2013, 160 (11), A2160, DOI: 10.1149/2.074311jesGoogle ScholarThere is no corresponding record for this reference.
- 24Pham, T. A.; Kweon, K. E.; Samanta, A.; Lordi, V.; Pask, J. E. Solvation and Dynamics of Sodium and Potassium in Ethylene Carbonate from Ab initio Molecular Dynamics Simulations. J. Phys. Chem. C 2017, 121 (40), 21913– 21920, DOI: 10.1021/acs.jpcc.7b06457Google ScholarThere is no corresponding record for this reference.
- 25Song, Z.; Xing, Z.; Yang, J.; Chen, J.; Hu, W.; Li, P.; Feng, W.; Eshetu, G. G.; Figgemeier, E.; Passerini, S. Electrolyte Chemistry Development for Sodium-Based Batteries: A Blueprint from Lithium or a Step Toward Originality?. Angew. Chem., Int. Ed. 2025, 64 (23), e202424543 DOI: 10.1002/anie.202424543Google ScholarThere is no corresponding record for this reference.
- 26Wang, Y.; Lan, H.; Dong, S.; Zhu, Q.; Cheng, L.; Wang, H.; Wang, J.; Wang, S.; Tang, M.; Shodievich, K. M. A High-Power Rechargeable Sodium-Ion Full Battery Operating at −40 °C. Adv. Funct. Mater. 2024, 34 (26), 2315498, DOI: 10.1002/adfm.202315498Google ScholarThere is no corresponding record for this reference.
- 27Kondo, Y.; Fukutsuka, T.; Yokoyama, Y.; Miyahara, Y.; Miyazaki, K.; Abe, T. Sodium/Lithium-Ion Transfer Reaction at the Interface between Low-Crystallized Carbon Nanosphere Electrodes and Organic Electrolytes. ACS omega 2021, 6 (29), 18737– 18744, DOI: 10.1021/acsomega.1c01751Google ScholarThere is no corresponding record for this reference.
- 28Soto, F. A.; Marzouk, A.; El-Mellouhi, F.; Balbuena, P. B. Understanding Ionic Diffusion through SEI Components for Lithium-Ion and Sodium-Ion Batteries: Insights from First-Principles Calculations. Chem. Mater. 2018, 30 (10), 3315– 3322, DOI: 10.1021/acs.chemmater.8b00635Google ScholarThere is no corresponding record for this reference.
- 29Shinde, S. S.; Wagh, N. K.; Kim, S. H.; Lee, J. H. Li, Na, K, Mg, Zn, Al, and Ca Anode Interface Chemistries Developed by Solid-State Electrolytes. Adv. Sci. 2023, 10 (32), 2304235, DOI: 10.1002/advs.202304235Google ScholarThere is no corresponding record for this reference.
- 30Loaiza, L. C.; Monconduit, L.; Seznec, V. Si and Ge-Based Anode Materials for Li-, Na-, and K-ion Batteries: A Perspective from Structure to Electrochemical Mechanism. Small 2020, 16 (5), 1905260, DOI: 10.1002/smll.201905260Google ScholarThere is no corresponding record for this reference.
- 31Yildirim, H.; Kinaci, A.; Chan, M. K.; Greeley, J. P. First-Principles Analysis of Defect Thermodynamics and Ion Transport in Inorganic SEI Compounds: LiF and NaF. ACS Appl. Mater. Interfaces 2015, 7 (34), 18985– 18996, DOI: 10.1021/acsami.5b02904Google ScholarThere is no corresponding record for this reference.
- 32Wu, Y.; Hu, Q.; Liang, H.; Wang, A.; Xu, H.; Wang, L.; He, X. Electrostatic Potential as Solvent Descriptor to Enable Rational Electrolyte Design for Lithium Batteries. Adv. Energy Mater. 2023, 13 (22), 2300259, DOI: 10.1002/aenm.202300259Google ScholarThere is no corresponding record for this reference.
- 33Chae, Y.; Lim, C.; Jeon, J.; Kim, M.; Lee, K.-K.; Kwak, K.; Cho, M. Lithium-Ion Solvation Structure in Organic Carbonate Electrolytes at Low Temperatures. J. Phys. Chem. Lett. 2022, 13 (33), 7881– 7888, DOI: 10.1021/acs.jpclett.2c02106Google ScholarThere is no corresponding record for this reference.
- 34Mo, Y.; Liu, G.; Chen, J.; Zhu, X.; Peng, Y.; Wang, Y.; Wang, C.; Dong, X.; Xia, Y. Unraveling the Temperature-Responsive Solvation Structure and Interfacial Chemistry for Graphite Anodes. Energy Environ. Sci. 2024, 17 (1), 227– 237, DOI: 10.1039/D3EE03176DGoogle ScholarThere is no corresponding record for this reference.
- 35Yuan, S.; Cao, S.; Chen, X.; Wei, J.; Lv, Z.; Xia, H.; Li, J.; Zhang, H.; Liu, L.; Tian, C. Deshielding Anions Enable Solvation Chemistry Control of LiPF6-Based Electrolyte toward Low-Temperature Lithium-Ion Batteries. Adv. Mater. 2024, 36 (16), 2311327, DOI: 10.1002/adma.202311327Google ScholarThere is no corresponding record for this reference.
- 36Park, S.; Kim, S.; Lee, J.-A.; Ue, M.; Choi, N.-S. Liquid Electrolyte Chemistries for Solid Electrolyte Interphase Construction on Silicon and Lithium-Metal Anodes. Chem. Sci. 2023, 14 (37), 9996– 10024, DOI: 10.1039/D3SC03514JGoogle ScholarThere is no corresponding record for this reference.
- 37Weng, S.; Zhang, X.; Yang, G.; Zhang, S.; Ma, B.; Liu, Q.; Liu, Y.; Peng, C.; Chen, H.; Yu, H. Temperature-Dependent Interphase Formation and Li+ Transport in Lithium Metal Batteries. Nat. Commun. 2023, 14 (1), 4474, DOI: 10.1038/s41467-023-40221-0Google ScholarThere is no corresponding record for this reference.
- 38Wang, Z.; Han, R.; Huang, D.; Wei, Y.; Song, H.; Liu, Y.; Xue, J.; Zhang, H.; Zhang, F.; Liu, L. Co-Intercalation-Free Ether-Based Weakly Solvating Electrolytes Enable Fast-Charging and Wide-Temperature Lithium-Ion Batteries. ACS Nano 2023, 17 (18), 18103– 18113, DOI: 10.1021/acsnano.3c04907Google ScholarThere is no corresponding record for this reference.
- 39Mo, Y.; Liu, G.; Yin, Y.; Tao, M.; Chen, J.; Peng, Y.; Wang, Y.; Yang, Y.; Wang, C.; Dong, X.; Xia, Y. Fluorinated Solvent Molecule Tuning Enables Fast-Charging and Low-Temperature Lithium-Ion Batteries. Adv. Energy Mater. 2023, 13 (32), 2301285, DOI: 10.1002/aenm.202301285Google ScholarThere is no corresponding record for this reference.
- 40Xu, J.; Zhang, J.; Pollard, T. P.; Li, Q.; Tan, S.; Hou, S.; Wan, H.; Chen, F.; He, H.; Hu, E. Electrolyte Design for Li-Ion Batteries under Extreme Operating Conditions. Nature 2023, 614 (7949), 694– 700, DOI: 10.1038/s41586-022-05627-8Google ScholarThere is no corresponding record for this reference.
- 41Qin, M.; Liu, M.; Zeng, Z.; Wu, Q.; Wu, Y.; Zhang, H.; Lei, S.; Cheng, S.; Xie, J. Rejuvenating Propylene Carbonate-Based Electrolyte through Nonsolvating Interactions for Wide-Temperature Li-Ions Batteries. Adv. Energy Mater. 2022, 12 (48), 2201801, DOI: 10.1002/aenm.202201801Google ScholarThere is no corresponding record for this reference.
- 42Liu, C.; Li, Z.; Jiang, L.; Zhu, H.; Wang, F.; Sheng, L. Dipole-Dipole Interactions in Electrolyte to Facilitate Li-Ion Desolvation for Low-Temperature Li-Ion Batteries. J. Energy Chem. 2025, 104, 678– 686, DOI: 10.1016/j.jechem.2025.01.017Google ScholarThere is no corresponding record for this reference.
- 43Gao, X.; Piao, N.; Yan, Y.; Wang, J.; Zou, H.; Guan, S.; Zeng, L.; Sun, Z.; Hu, G.; Li, F. Synergistic Fluorinated and Non-Fluorinated Solvents for Electrolytes of Lithium-Ion Batteries at Low Temperatures. Chin. Chem. Lett. 2024, 110591, DOI: 10.1016/j.cclet.2024.110591Google ScholarThere is no corresponding record for this reference.
- 44Cui, Z.; Wang, D.; Guo, J.; Nian, Q.; Ruan, D.; Fan, J.; Ma, J.; Li, L.; Dong, Q.; Luo, X. Push-Pull Electrolyte Design Strategy Enables High-Voltage Low-Temperature Lithium Metal Batteries. J. Am. Chem. Soc. 2024, 146 (40), 27644– 27654, DOI: 10.1021/jacs.4c09027Google ScholarThere is no corresponding record for this reference.
- 45Choi, N.-S.; Kim, S.-S.; Yew, K.-H.; Lee, K.-Y. Lithium Secondary Battery. U.S. Patent No. 482,512, Feb 15, 2007.Google ScholarThere is no corresponding record for this reference.
- 46Choi, N.-S.; Lee, Y. M.; Seol, W.; Lee, J. A.; Park, J.-K. Protective Coating of Lithium Metal Electrode for Interfacial Enhancement with Gel Polymer Electrolyte. Solid State Ionics 2004, 172 (1–4), 19– 24, DOI: 10.1016/j.ssi.2004.05.008Google ScholarThere is no corresponding record for this reference.
- 47Luo, Z.; Li, S.; Yang, L.; Tian, Y.; Xu, L.; Zou, G.; Hou, H.; Wei, W.; Chen, L.; Ji, X. Interfacially Redistributed Charge for Robust Lithium Metal Anode. Nano Energy 2021, 87, 106212, DOI: 10.1016/j.nanoen.2021.106212Google ScholarThere is no corresponding record for this reference.
- 48Li, W.; Wu, G.; Araújo, C. M.; Scheicher, R. H.; Blomqvist, A.; Ahuja, R.; Xiong, Z.; Feng, Y.; Chen, P. Li+ Ion Conductivity and Diffusion Mechanism in α-Li3N and β-Li3N. Energy Environ. Sci. 2010, 3 (10), 1524– 1530, DOI: 10.1039/c0ee00052cGoogle ScholarThere is no corresponding record for this reference.
- 49Lacivita, V.; Artrith, N.; Ceder, G. Structural and Compositional Factors that Control The Li-Ion Conductivity in LiPON Electrolytes. Chem. Mater. 2018, 30 (20), 7077– 7090, DOI: 10.1021/acs.chemmater.8b02812Google ScholarThere is no corresponding record for this reference.
- 50Lee, J. A.; Kim, S.; Cho, Y.; Kweon, S. H.; Kang, H.; Byun, J. H.; Kwon, E.; Seo, S.; Kim, W.; Ryu, K. H. Compositionally Sequenced Interfacial Layers for High-Energy Li-Metal Batteries. Adv. Sci. 2024, 11 (17), 2310094, DOI: 10.1002/advs.202310094Google ScholarThere is no corresponding record for this reference.
- 51Song, C.; Han, S. H.; Choi, Y.; Shin, H. R.; Kim, M. K.; Gong, C.; Chen, D.; Lee, J. W.; Hong, S.; Choi, N. S. Geometric Design of Interface Structures and Electrolyte Solvation Chemistry for Fast Charging Lithium-Ion Batteries. Adv. Mater. 2025, 37 (19), 2418773, DOI: 10.1002/adma.202418773Google ScholarThere is no corresponding record for this reference.
- 52Bommier, C.; Ji, X. Electrolytes, SEI Formation, and Binders: A Review of Nonelectrode Factors for Sodium-Ion Battery Anodes. Small 2018, 14 (16), 1703576, DOI: 10.1002/smll.201703576Google ScholarThere is no corresponding record for this reference.
- 53Besenhard, J.; Winter, M.; Yang, J.; Biberacher, W. Filming Mechanism of Lithium-Carbon Anodes in Organic and Inorganic Electrolytes. J. Power Sources 1995, 54 (2), 228– 231, DOI: 10.1016/0378-7753(94)02073-CGoogle ScholarThere is no corresponding record for this reference.
- 54Shi, P.; Lin, M.; Zheng, H.; He, X.; Xue, Z.; Xiang, H.; Chen, C. Effect of Propylene Carbonate-Li+ Solvation Structures on Graphite Exfoliation and Its Application in Li-Ion Batteries. Electrochim. Acta 2017, 247, 12– 18, DOI: 10.1016/j.electacta.2017.06.174Google ScholarThere is no corresponding record for this reference.
- 55Kim, H.; Yoon, G.; Lim, K.; Kang, K. A Comparative Study of Graphite Electrodes using The Co-Intercalation Phenomenon for Rechargeable Li, Na and K Batteries. Chem. Commun. 2016, 52 (85), 12618– 12621, DOI: 10.1039/C6CC05362AGoogle ScholarThere is no corresponding record for this reference.
- 56Kim, H.; Lim, K.; Yoon, G.; Park, J. H.; Ku, K.; Lim, H. D.; Sung, Y. E.; Kang, K. Exploiting Lithium-Ether Co-Intercalation in Graphite for High-Power Lithium-Ion Batteries. Adv. Energy Mater. 2017, 7 (19), 1700418, DOI: 10.1002/aenm.201700418Google ScholarThere is no corresponding record for this reference.
- 57Lai, P.; Zhang, Y.; Liu, J.; Zhang, Z.; Xie, H.; Li, X.; Deng, X.; Huang, B.; Zhang, P.; Zhao, J. Solvation-Structure-Preserved Electrolyte Breaks the Low Temperature Barrier for Sodium Metal Battery. J. Energy Chem. 2025, 107, 852– 860, DOI: 10.1016/j.jechem.2025.05.010Google ScholarThere is no corresponding record for this reference.
- 58Huang, Z.; Xiao, Z.; Zhang, H.; Zhang, Q.; Cui, J.; Luo, J.; Tang, W.; Wu, Y. Temperature-Robust Solvation Enabled by Solvent Interactions for Low-Temperature Sodium Metal Batteries. J. Am. Chem. Soc. 2025, 147 (6), 5162– 5171, DOI: 10.1021/jacs.4c15478Google ScholarThere is no corresponding record for this reference.
- 59Shen, Y.; Xu, J.; Li, Y.; Che, H.; Zhao, S.; Ishaq, M.; Jabeen, M.; Zhang, Y.; Wu, J.; Li, J. Anion-Cation Synergistic Interactions for Low-Temperature and Fast-Charging Performance in Sodium Batteries. J. Mater. Chem. A 2025, 13 (25), 19631– 19643, DOI: 10.1039/D5TA02276BGoogle ScholarThere is no corresponding record for this reference.
- 60Zhou, X.; Huang, Y.; Wen, B.; Yang, Z.; Hao, Z.; Li, L.; Chou, S.-L.; Li, F. Regulation of Anion-Na+ Coordination Chemistry in Electrolyte Solvates for Low-Temperature Sodium-Ion Batteries. Proc. Natl. Acad. Sci. U. S. A. 2024, 121 (5), e2316914121 DOI: 10.1073/pnas.2316914121Google ScholarThere is no corresponding record for this reference.
- 61Kim, S. C.; Wang, J.; Xu, R.; Zhang, P.; Chen, Y.; Huang, Z.; Yang, Y.; Yu, Z.; Oyakhire, S. T.; Zhang, W. High-Entropy Electrolytes for Practical Lithium Metal Batteries. Nat. Energy 2023, 8 (8), 814– 826, DOI: 10.1038/s41560-023-01280-1Google ScholarThere is no corresponding record for this reference.
- 62Yang, C.; Liu, X.; Lin, Y.; Yin, L.; Lu, J.; You, Y. Entropy-Driven Solvation toward Low-Temperature Sodium-Ion Batteries with Temperature-Adaptive Feature. Adv. Mater. 2023, 35 (28), 2301817, DOI: 10.1002/adma.202301817Google ScholarThere is no corresponding record for this reference.
- 63Zheng, C.; Yao, Q.; Sun, Y.; Lv, H.; Bai, Z.; Zhang, G.; Wang, N.; Yang, J. Enhancing Low-Temperature Performance of Sodium-Ion Batteries via Anion-Solvent Interactions. Adv. Funct. Mater. 2025, 2501303, DOI: 10.1002/adfm.202501303Google ScholarThere is no corresponding record for this reference.
- 64Fang, H.; Huang, Y.; Hu, W.; Song, Z.; Wei, X.; Geng, J.; Jiang, Z.; Qu, H.; Chen, J.; Li, F. Regulating Ion-Dipole Interactions in Weakly Solvating Electrolyte towards Ultra-Low Temperature Sodium-Ion Batteries. Angew. Chem. 2024, 136 (15), e202400539 DOI: 10.1002/ange.202400539Google ScholarThere is no corresponding record for this reference.
- 65Cui, Y.; Ni, Y.; Wang, Y.; Wang, L.; Yang, W.; Wu, S.; Xie, W.; Zhang, K.; Yan, Z.; Chen, J. A Temperature-Adapted Ultraweakly Solvating Electrolyte for Cold-Resistant Sodium-Ion Batteries. Adv. Energy Mater. 2025, 15 (21), 2405363, DOI: 10.1002/aenm.202405363Google ScholarThere is no corresponding record for this reference.
- 66Cui, K.; Hou, R.; Zhou, H.; Guo, S. Electrolyte Engineering of Hard Carbon for Sodium-Ion Batteries: From Mechanism Analysis to Design Strategies. Adv. Funct. Mater. 2025, 35 (16), 2419275, DOI: 10.1002/adfm.202419275Google ScholarThere is no corresponding record for this reference.
- 67Ma, X.-X.; Shen, X.; Chen, X.; Fu, Z.-H.; Yao, N.; Zhang, R.; Zhang, Q. The Origin of Fast Lithium-Ion Transport in The Inorganic Solid Electrolyte Interphase on Lithium Metal Anodes. Small Struct. 2022, 3 (8), 2200071, DOI: 10.1002/sstr.202200071Google ScholarThere is no corresponding record for this reference.
- 68Wang, Y.; Hou, H.; Tantratian, K.; Goncharova, L. V.; Fu, B.; Jin, E.; Pirayesh, P.; Abdolvand, H.; Pang, X.; Chen, L. Insight into the Interface Design for Li Metal Anode: Organic-Rich or Inorganic-Rich. Adv. Funct. Mater. 2024, 34 (46), 2406426, DOI: 10.1002/adfm.202406426Google ScholarThere is no corresponding record for this reference.
- 69Dong, R.; Zheng, L.; Bai, Y.; Ni, Q.; Li, Y.; Wu, F.; Ren, H.; Wu, C. Elucidating The Mechanism of Fast Na Storage Kinetics in Ether Electrolytes for Hard Carbon Anodes. Adv. Mater. 2021, 33 (36), 2008810, DOI: 10.1002/adma.202008810Google ScholarThere is no corresponding record for this reference.
- 70Ma, M.; Cai, H.; Xu, C.; Huang, R.; Wang, S.; Pan, H.; Hu, Y. S. Engineering Solid Electrolyte Interface at Nano-Scale for High-Performance Hard Carbon in Sodium-Ion Batteries. Adv. Funct. Mater. 2021, 31 (25), 2100278, DOI: 10.1002/adfm.202100278Google ScholarThere is no corresponding record for this reference.
- 71Kwon, H.; Kim, S.; Hyun, J.; Lee, H. E.; Kim, S. S.; Kim, Y.; Kim, I. J.; Shin, K.; Kim, S.; Park, C. Covariance of Interphasic Properties and Fast Chargeability of Energy-Dense Lithium Metal Batteries. Nat. Energy 2025, 10, 1132, DOI: 10.1038/s41560-025-01838-1Google ScholarThere is no corresponding record for this reference.
- 72Hirsh, H. S.; Sayahpour, B.; Shen, A.; Li, W.; Lu, B.; Zhao, E.; Zhang, M.; Meng, Y. S. Role of Electrolyte in Stabilizing Hard Carbon as An Anode for Rechargeable Sodium-Ion Batteries with Long Cycle Life. Energy Storage Mater. 2021, 42, 78– 87, DOI: 10.1016/j.ensm.2021.07.021Google ScholarThere is no corresponding record for this reference.
- 73Ji, Y.; Qiu, J.; Zhao, W.; Liu, T.; Dong, Z.; Yang, K.; Zheng, G.; Qian, G.; Yang, M.; Chen, Q. In Situ Probing The Origin of Interfacial Instability of Na Metal Anode. Chem. 2023, 9 (10), 2943– 2955, DOI: 10.1016/j.chempr.2023.06.002Google ScholarThere is no corresponding record for this reference.
- 74Bhattacharya, S.; Alpas, A. T. Micromechanisms of Solid Electrolyte Interphase Formation on Electrochemically Cycled Graphite Electrodes in Lithium-Ion Cells. Carbon 2012, 50 (15), 5359– 5371, DOI: 10.1016/j.carbon.2012.07.009Google ScholarThere is no corresponding record for this reference.
- 75Wang, Q.; Zhao, C.; Hu, X.; Wang, J.; Ganapathy, S.; Eustace, S.; Bai, X.; Li, B.; Li, H.; Aurbach, D. Grain-Boundary-Rich Interphases for Rechargeable Batteries. J. Am. Chem. Soc. 2024, 146 (46), 31778– 31787, DOI: 10.1021/jacs.4c10650Google ScholarThere is no corresponding record for this reference.
- 76Tian, J.; Chen, N.; Feng, M.; Sun, L.; Yang, B.; Zhang, F.; Wu, F.; Chen, R. Cofactor-Assisted Weakly Solvated Electrolytes for Low-Temperature Charging of Graphite Anode. Adv. Funct. Mater. 2025, 35, 2504252, DOI: 10.1002/adfm.202504252Google ScholarThere is no corresponding record for this reference.
- 77Perez-Beltran, S.; Kuai, D.; Balbuena, P. B. SEI Formation and Lithium-Ion Electrodeposition Dynamics in Lithium Metal Batteries via First-Principles Kinetic Monte Carlo Modeling. ACS Energy Lett. 2024, 9 (11), 5268– 5278, DOI: 10.1021/acsenergylett.4c02019Google ScholarThere is no corresponding record for this reference.
- 78Mogensen, R.; Brandell, D.; Younesi, R. Solubility of the Solid Electrolyte Interphase (SEI) in Sodium Ion Batteries. ACS Energy Lett. 2016, 1 (6), 1173– 1178, DOI: 10.1021/acsenergylett.6b00491Google ScholarThere is no corresponding record for this reference.
- 79Liu, J.; Hao, W.; Fang, M.; Chen, X.; Dong, Y.; Chen, Y.; Wang, Z.; Yue, X.; Liang, Z. Screening of F-containing Electrolyte Additives and Clarifying Their Decomposition Routes for Stable Li Metal Anodes. Nat. Commun. 2024, 15 (1), 9356, DOI: 10.1038/s41467-024-53807-zGoogle ScholarThere is no corresponding record for this reference.
- 80Cai, J.; Wang, A.; Li, S.; Wu, Z.; Jiang, T.; Wang, W.; Fan, W.; Kang, T.; Xiao, X.; Nan, J. A Hard-Soft Synergy Strategy Enables Drastic Temperature Performance of Pouch Sodium-Ion Batteries over Wide-Temperature Range of −30 to 60 °C. Energy Storage Materials 2025, 75, 103997, DOI: 10.1016/j.ensm.2024.103997Google ScholarThere is no corresponding record for this reference.
- 81Zhang, Y.; Wu, Z.; Wang, Y.; Chen, G.; Yang, K.; Li, L.; Liu, X.; Ma, Z.; Chen, L. Revisiting Interfacial Enhancement Effects of Fluorinated Cyclophosphazene in Practical Sodium-Ion Batteries. ACS Appl. Mater. & Interfaces 2025, 17 (21), 31553– 31560, DOI: 10.1021/acsami.5c02243Google ScholarThere is no corresponding record for this reference.
- 82Liang, H.-J.; Qian, W.-Y.; Liu, H.-H.; Wang, X.-T.; Gu, Z.-Y.; Dong, F.; Deng, Y.; Tang, Y.-Z.; Zhang, J.; Zhao, J. Sulfite-Based Electrolyte Chemistry with Ion-Dipole Interactions and Robust Interphase Achieves Wide-Temperature Sodium-Ion Batteries. J. Am. Chem. Soc. 2025, 147 (21), 17860– 17870, DOI: 10.1021/jacs.5c01864Google ScholarThere is no corresponding record for this reference.
- 83Li, S.; Liu, H.; Qiu, H.; He, X.; Fan, W.; Wang, W.; Nan, J. An Electrolyte Design Strategy for Commercial All-Climate Pouch Sodium-Ion Batteries. Chem. Eng. J. 2025, 505, 159102, DOI: 10.1016/j.cej.2024.159102Google ScholarThere is no corresponding record for this reference.
- 84Hu, R.; Yang, L.; Zhang, C.; Chen, B.; Hong, H.; Shen, H.; Mao, C.; Yang, Z.; Zhang, S.; Huang, Q. Bifunctional Sodium Tetrakis [3,5-bis(trifluoromethyl)phenyl] Borate Additive for Long-Lifespan Sodium-Ion Batteries with NaNi0.33Fe0.33Mn0.33O2 Cathode. Chem. Eng. J. 2025, 512, 162144, DOI: 10.1016/j.cej.2025.162144Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 2 publications.
- Chao Li, Yiming Yu, Jiaqin Huang, Guanglan Zhuo, Jizhang Chen, Qinghua Tian. Boosting the performance of irregular Si particles for lithium storage by simultaneously integrating with Sn and carbon. Journal of Physics and Chemistry of Solids 2026, 213 , 113592. https://doi.org/10.1016/j.jpcs.2026.113592
- Ling Liang, Shijian Zhu, Lei Gao, Liping Wang, Bingkai Zhang, Jing Liu, Jinlong Zhu, Songbai Han. 3D printing and in-situ polymerization enabled Interface-free double-layered polymer gel electrolyte for highly stable lithium metal batteries. Chemical Engineering Journal 2026, 530 , 173399. https://doi.org/10.1016/j.cej.2026.173399
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
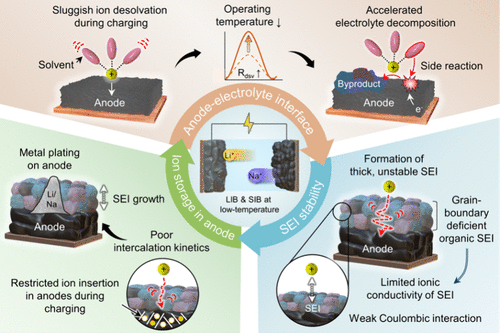
Figure 1
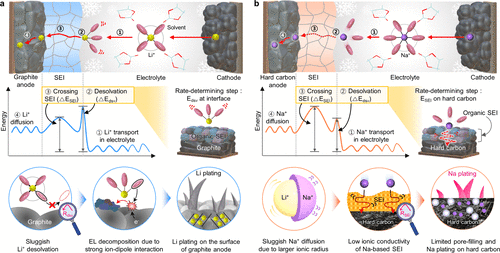
Figure 1. Schematic illustration of challenges encountered in (a) Li-ion and (b) Na-ion batteries under low-temperature conditions.
Figure 2

Figure 2. (a) Correlation between the absolute |ESPmin| values of solvent species and the desolvation energy of Li+ ion. Reproduced with permission from ref (23). Copyright 2013 The Electrochemical Society. (b) Comparative desolvation energies of different cation species. (c) Correlation between the coordination number of cations and the solvation power depending on the functional groups. (d) Effect of operating temperature on the solvation environment and SEI structure. Reproduced with permission from ref (35). Copyright 2024 Wiley-VCH. (e) Temperature-dependent bond-breaking modes and corresponding changes in SEI composition at low temperatures. Reproduced with permission from ref (37). Copyright 2023 Springer Nature.
Figure 3

Figure 3. (a) Schematics of internal limitations and strategies for regulation of solvation structures for low-temperature LIBs. (b) Solvent diagram of DN versus dielectric constant. Solvents located in the blue-colored zone are denoted as soft solvents characterized by a lower DN and moderate dielectric constant, which reduce the Li+-solvent interactions without compromising ionic conductivity, where ε indicates the dielectric constant of solvents. (c) Electrolyte screening via the maximum and minimum values of the ESP surfaces. Reproduced with permission from ref (44). Copyright 2024 American Chemical Society. (d) Selection of deshielding anions based on the relative 7Li chemical shift and compatibility with graphite anodes. Reproduced with permission from ref (35). Copyright 2024 Wiley-VCH. (e) Transport barriers and ionic conductivities of SEI components at room temperature. (36,47−49) (f) Ionic conductivity of SEI as a function of grain size, where η and σ indicate viscosity and ionic conductivity, respectively. Reproduced with permission from ref (51). Copyright 2025 Wiley-VCH.
Figure 4

Figure 4. (a) Schematic of conditions required for low-temperature operation with strongly solvating electrolyte, weakly solvating electrolyte, and hybrid solvating electrolyte. (b) Solvent coordination number and charge-transfer activation energy with strongly solvating solvent, weakly solvating solvent, and hybrid solvating solvent. (c) Solvent-derived SEI formation and slow Na+ transport through inhomogeneous organic-rich SEI. (d) Anion-derived SEI formation and fast Na+ transport through uniform inorganic-rich SEI.
Figure 5

Figure 5. Challenges for achieving suitable low-temperature battery performance and electrolyte design strategies. HFGA: hexafluoroglutaric anhydride, TMSP: tris(trimethylsilyl) phosphate, TMSB: tris(trimethylsilyl) borate, DTD: 1,3,2-dioxathiolane 2,2-dioxide.
References
This article references 84 other publications.
- 1Chen, S.; Hu, T.; Yu, T.; Luo, X.; Zhang, L.; Li, F. Structural Feature Design for Carbon Materials toward Sodium Storage: Insights and Prospects. ACS Energy Lett. 2025, 10 (4), 1931– 1952, DOI: 10.1021/acsenergylett.5c00231There is no corresponding record for this reference.
- 2Shi, Y.; Hu, E.; Sumboja, A.; Anggraningrum, I. T.; Syahrial, A. Z.; Yan, Q. Layered 3d Transition Metal-Based Oxides for Sodium-Ion and Lithium-Ion Batteries: Differences, Links and Beyond. Adv. Funct. Mater. 2025, 35 (2), 2413078, DOI: 10.1002/adfm.202413078There is no corresponding record for this reference.
- 3Nekahi, A.; Dorri, M.; Rezaei, M.; Bouguern, M. D.; Madikere Raghunatha Reddy, A. K.; Li, X.; Deng, S.; Zaghib, K. Comparative Issues of Metal-Ion Batteries toward Sustainable Energy Storage: Lithium vs. Sodium. Batteries 2024, 10 (8), 279, DOI: 10.3390/batteries10080279There is no corresponding record for this reference.
- 4Tan, S.; Shadike, Z.; Cai, X.; Lin, R.; Kludze, A.; Borodin, O.; Lucht, B. L.; Wang, C.; Hu, E.; Xu, K.; Yang, X.-Q. Review on Low-Temperature Electrolytes for Lithium-Ion and Lithium Metal Batteries. Electrochem. Energy Rev. 2023, 6 (1), 35, DOI: 10.1007/s41918-023-00199-1There is no corresponding record for this reference.
- 5Goktas, M.; Bolli, C.; Berg, E. J.; Novák, P.; Pollok, K.; Langenhorst, F.; Roeder, M. v.; Lenchuk, O.; Mollenhauer, D.; Adelhelm, P. Graphite as Cointercalation Electrode for Sodium-Ion Batteries: Electrode Dynamics and The Missing Solid Electrolyte Interphase (SEI). Adv. Energy Mater. 2018, 8 (16), 1702724, DOI: 10.1002/aenm.201702724There is no corresponding record for this reference.
- 6Sun, N.; Qiu, J.; Xu, B. Understanding of Sodium Storage Mechanism in Hard Carbons: Ongoing Development Under Debate. Adv. Energy Mater. 2022, 12 (27), 2200715, DOI: 10.1002/aenm.202200715There is no corresponding record for this reference.
- 7Irisarri, E.; Ponrouch, A.; Palacin, M. Hard Carbon Negative Electrode Materials for Sodium-Ion Batteries. J. Electrochem. Soc. 2015, 162 (14), A2476, DOI: 10.1149/2.0091514jesThere is no corresponding record for this reference.
- 8Xia, J. L.; Yan, D.; Guo, L. P.; Dong, X. L.; Li, W. C.; Lu, A. H. Hard Carbon Nanosheets with Uniform Ultramicropores and Accessible Functional Groups Showing High Realistic Capacity and Superior Rate Performance for Sodium-Ion Storage. Adv. Mater. 2020, 32 (21), 2000447, DOI: 10.1002/adma.202000447There is no corresponding record for this reference.
- 9Hong, S. Y.; Kim, Y.; Park, Y.; Choi, A.; Choi, N.-S.; Lee, K. T. Charge Carriers in Rechargeable Batteries: Na Ions vs. Li Ions. Energy Environ. Sci. 2013, 6 (7), 2067– 2081, DOI: 10.1039/c3ee40811fThere is no corresponding record for this reference.
- 10Jache, B.; Binder, J. O.; Abe, T.; Adelhelm, P. A Comparative Study on The Impact of Different Glymes and Their Derivatives as Electrolyte Solvents for Graphite Co-Intercalation Electrodes in Lithium-Ion and Sodium-Ion Batteries. Phys. Chem. Chem. Phys. 2016, 18 (21), 14299– 14316, DOI: 10.1039/C6CP00651EThere is no corresponding record for this reference.
- 11Cresce, A. V.; Russell, S. M.; Borodin, O.; Allen, J. A.; Schroeder, M. A.; Dai, M.; Peng, J.; Gobet, M. P.; Greenbaum, S. G.; Rogers, R. E.; Xu, K. Solvation Behavior of Carbonate-Based Electrolytes in Sodium Ion Batteries. Phys. Chem. Chem. Phys. 2017, 19 (1), 574– 586, DOI: 10.1039/C6CP07215AThere is no corresponding record for this reference.
- 12Huang, Z.; Xiao, Z.; Jin, R.; Li, Z.; Shu, C.; Shi, R.; Wang, X.; Tang, Z.; Tang, W.; Wu, Y. A Comprehensive Review on Liquid Electrolyte Design for Low-Temperature Lithium/Sodium Metal Batteries. Energy Environ. Sci. 2024, 17 (15), 5365– 5386, DOI: 10.1039/D4EE02060JThere is no corresponding record for this reference.
- 13Shanbedi, M.; Shahali, H.; Polycarpou, A. A.; Amiri, A. Advances and Future Prospects of Low-Temperature Electrolytes for Lithium-Ion Batteries. EES Batteries. 2025, 1, 385– 426, DOI: 10.1039/D5EB00013KThere is no corresponding record for this reference.
- 14Jeong, D.; Tackett, B. M.; Pol, V. G. Tailored Li-Ion Battery Electrodes and Electrolytes for Extreme Condition Operation. Commun. Chem. 2025, 8, 170, DOI: 10.1038/s42004-025-01564-5There is no corresponding record for this reference.
- 15Su, Q.; Hu, C.; Li, S.; Huang, W.; Dong, J.; Song, R.; Xu, L.; Fang, G. Sodium-Ion Batteries at Low Temperature: Storage Mechanism and Modification Strategies. Chin. Chem. Lett. 2025, 111267, DOI: 10.1016/j.cclet.2025.111267There is no corresponding record for this reference.
- 16Li, M.; Zhuo, H.; Jing, Q.; Gu, Y.; Liao, Z.; Wang, K.; Hu, J.; Geng, D.; Sun, X.; Xiao, B. Low-Temperature Performance of Na-Ion Batteries. Carbon Energy 2024, 6 (10), e546 DOI: 10.1002/cey2.546There is no corresponding record for this reference.
- 17Bai, Z.; Yao, Q.; Wang, M.; Meng, W.; Dou, S.; Liu, H. K.; Wang, N. Low-Temperature Sodium-Ion Batteries: Challenges and Progress. Adv. Energy Mater. 2024, 14, 2303788, DOI: 10.1002/aenm.202303788There is no corresponding record for this reference.
- 18Qiu, X.; Chen, Y.; Sun, Y.; Wang, Y.; Liang, Z.; Zhou, G.; Xue, Y.; Shi, L.; Jiang, J.; Kong, X.; Zhuang, Q.; Ju, Z. Research on Low-Temperature Sodium-Ion Batteries: Challenges, Strategies and Prospect. Energy Storage Mater. 2024, 72, 103760, DOI: 10.1016/j.ensm.2024.103760There is no corresponding record for this reference.
- 19Yang, S.; Cheng, K.; Cao, Z. Status and Strategies of Electrolyte Engineering for Low-Temperature Sodium-Ion Batteries. J. Mater. Chem. A 2024, 12 (22), 13059– 13080, DOI: 10.1039/D4TA01400FThere is no corresponding record for this reference.
- 20Zhang, S.; Xu, K.; Jow, T. The Low Temperature Performance of Li-Ion Batteries. J. Power Sources 2003, 115 (1), 137– 140, DOI: 10.1016/S0378-7753(02)00618-3There is no corresponding record for this reference.
- 21Abe, T.; Fukuda, H.; Iriyama, Y.; Ogumi, Z. Solvated Li-Ion Transfer at Interface between Graphite and Electrolyte. J. Electrochem. Soc. 2004, 151 (8), A1120, DOI: 10.1149/1.1763141There is no corresponding record for this reference.
- 22Xu, K.; von Cresce, A.; Lee, U. Differentiating Contributions to “Ion Transfer” Barrier from Interphasial Resistance and Li+ Desolvation at Electrolyte/Graphite Interface. Langmuir 2010, 26 (13), 11538– 11543, DOI: 10.1021/la1009994There is no corresponding record for this reference.
- 23Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical Analysis on De-Solvation of Lithium, Sodium, and Magnesium Cations to Organic Electrolyte Solvents. J. Electrochem. Soc. 2013, 160 (11), A2160, DOI: 10.1149/2.074311jesThere is no corresponding record for this reference.
- 24Pham, T. A.; Kweon, K. E.; Samanta, A.; Lordi, V.; Pask, J. E. Solvation and Dynamics of Sodium and Potassium in Ethylene Carbonate from Ab initio Molecular Dynamics Simulations. J. Phys. Chem. C 2017, 121 (40), 21913– 21920, DOI: 10.1021/acs.jpcc.7b06457There is no corresponding record for this reference.
- 25Song, Z.; Xing, Z.; Yang, J.; Chen, J.; Hu, W.; Li, P.; Feng, W.; Eshetu, G. G.; Figgemeier, E.; Passerini, S. Electrolyte Chemistry Development for Sodium-Based Batteries: A Blueprint from Lithium or a Step Toward Originality?. Angew. Chem., Int. Ed. 2025, 64 (23), e202424543 DOI: 10.1002/anie.202424543There is no corresponding record for this reference.
- 26Wang, Y.; Lan, H.; Dong, S.; Zhu, Q.; Cheng, L.; Wang, H.; Wang, J.; Wang, S.; Tang, M.; Shodievich, K. M. A High-Power Rechargeable Sodium-Ion Full Battery Operating at −40 °C. Adv. Funct. Mater. 2024, 34 (26), 2315498, DOI: 10.1002/adfm.202315498There is no corresponding record for this reference.
- 27Kondo, Y.; Fukutsuka, T.; Yokoyama, Y.; Miyahara, Y.; Miyazaki, K.; Abe, T. Sodium/Lithium-Ion Transfer Reaction at the Interface between Low-Crystallized Carbon Nanosphere Electrodes and Organic Electrolytes. ACS omega 2021, 6 (29), 18737– 18744, DOI: 10.1021/acsomega.1c01751There is no corresponding record for this reference.
- 28Soto, F. A.; Marzouk, A.; El-Mellouhi, F.; Balbuena, P. B. Understanding Ionic Diffusion through SEI Components for Lithium-Ion and Sodium-Ion Batteries: Insights from First-Principles Calculations. Chem. Mater. 2018, 30 (10), 3315– 3322, DOI: 10.1021/acs.chemmater.8b00635There is no corresponding record for this reference.
- 29Shinde, S. S.; Wagh, N. K.; Kim, S. H.; Lee, J. H. Li, Na, K, Mg, Zn, Al, and Ca Anode Interface Chemistries Developed by Solid-State Electrolytes. Adv. Sci. 2023, 10 (32), 2304235, DOI: 10.1002/advs.202304235There is no corresponding record for this reference.
- 30Loaiza, L. C.; Monconduit, L.; Seznec, V. Si and Ge-Based Anode Materials for Li-, Na-, and K-ion Batteries: A Perspective from Structure to Electrochemical Mechanism. Small 2020, 16 (5), 1905260, DOI: 10.1002/smll.201905260There is no corresponding record for this reference.
- 31Yildirim, H.; Kinaci, A.; Chan, M. K.; Greeley, J. P. First-Principles Analysis of Defect Thermodynamics and Ion Transport in Inorganic SEI Compounds: LiF and NaF. ACS Appl. Mater. Interfaces 2015, 7 (34), 18985– 18996, DOI: 10.1021/acsami.5b02904There is no corresponding record for this reference.
- 32Wu, Y.; Hu, Q.; Liang, H.; Wang, A.; Xu, H.; Wang, L.; He, X. Electrostatic Potential as Solvent Descriptor to Enable Rational Electrolyte Design for Lithium Batteries. Adv. Energy Mater. 2023, 13 (22), 2300259, DOI: 10.1002/aenm.202300259There is no corresponding record for this reference.
- 33Chae, Y.; Lim, C.; Jeon, J.; Kim, M.; Lee, K.-K.; Kwak, K.; Cho, M. Lithium-Ion Solvation Structure in Organic Carbonate Electrolytes at Low Temperatures. J. Phys. Chem. Lett. 2022, 13 (33), 7881– 7888, DOI: 10.1021/acs.jpclett.2c02106There is no corresponding record for this reference.
- 34Mo, Y.; Liu, G.; Chen, J.; Zhu, X.; Peng, Y.; Wang, Y.; Wang, C.; Dong, X.; Xia, Y. Unraveling the Temperature-Responsive Solvation Structure and Interfacial Chemistry for Graphite Anodes. Energy Environ. Sci. 2024, 17 (1), 227– 237, DOI: 10.1039/D3EE03176DThere is no corresponding record for this reference.
- 35Yuan, S.; Cao, S.; Chen, X.; Wei, J.; Lv, Z.; Xia, H.; Li, J.; Zhang, H.; Liu, L.; Tian, C. Deshielding Anions Enable Solvation Chemistry Control of LiPF6-Based Electrolyte toward Low-Temperature Lithium-Ion Batteries. Adv. Mater. 2024, 36 (16), 2311327, DOI: 10.1002/adma.202311327There is no corresponding record for this reference.
- 36Park, S.; Kim, S.; Lee, J.-A.; Ue, M.; Choi, N.-S. Liquid Electrolyte Chemistries for Solid Electrolyte Interphase Construction on Silicon and Lithium-Metal Anodes. Chem. Sci. 2023, 14 (37), 9996– 10024, DOI: 10.1039/D3SC03514JThere is no corresponding record for this reference.
- 37Weng, S.; Zhang, X.; Yang, G.; Zhang, S.; Ma, B.; Liu, Q.; Liu, Y.; Peng, C.; Chen, H.; Yu, H. Temperature-Dependent Interphase Formation and Li+ Transport in Lithium Metal Batteries. Nat. Commun. 2023, 14 (1), 4474, DOI: 10.1038/s41467-023-40221-0There is no corresponding record for this reference.
- 38Wang, Z.; Han, R.; Huang, D.; Wei, Y.; Song, H.; Liu, Y.; Xue, J.; Zhang, H.; Zhang, F.; Liu, L. Co-Intercalation-Free Ether-Based Weakly Solvating Electrolytes Enable Fast-Charging and Wide-Temperature Lithium-Ion Batteries. ACS Nano 2023, 17 (18), 18103– 18113, DOI: 10.1021/acsnano.3c04907There is no corresponding record for this reference.
- 39Mo, Y.; Liu, G.; Yin, Y.; Tao, M.; Chen, J.; Peng, Y.; Wang, Y.; Yang, Y.; Wang, C.; Dong, X.; Xia, Y. Fluorinated Solvent Molecule Tuning Enables Fast-Charging and Low-Temperature Lithium-Ion Batteries. Adv. Energy Mater. 2023, 13 (32), 2301285, DOI: 10.1002/aenm.202301285There is no corresponding record for this reference.
- 40Xu, J.; Zhang, J.; Pollard, T. P.; Li, Q.; Tan, S.; Hou, S.; Wan, H.; Chen, F.; He, H.; Hu, E. Electrolyte Design for Li-Ion Batteries under Extreme Operating Conditions. Nature 2023, 614 (7949), 694– 700, DOI: 10.1038/s41586-022-05627-8There is no corresponding record for this reference.
- 41Qin, M.; Liu, M.; Zeng, Z.; Wu, Q.; Wu, Y.; Zhang, H.; Lei, S.; Cheng, S.; Xie, J. Rejuvenating Propylene Carbonate-Based Electrolyte through Nonsolvating Interactions for Wide-Temperature Li-Ions Batteries. Adv. Energy Mater. 2022, 12 (48), 2201801, DOI: 10.1002/aenm.202201801There is no corresponding record for this reference.
- 42Liu, C.; Li, Z.; Jiang, L.; Zhu, H.; Wang, F.; Sheng, L. Dipole-Dipole Interactions in Electrolyte to Facilitate Li-Ion Desolvation for Low-Temperature Li-Ion Batteries. J. Energy Chem. 2025, 104, 678– 686, DOI: 10.1016/j.jechem.2025.01.017There is no corresponding record for this reference.
- 43Gao, X.; Piao, N.; Yan, Y.; Wang, J.; Zou, H.; Guan, S.; Zeng, L.; Sun, Z.; Hu, G.; Li, F. Synergistic Fluorinated and Non-Fluorinated Solvents for Electrolytes of Lithium-Ion Batteries at Low Temperatures. Chin. Chem. Lett. 2024, 110591, DOI: 10.1016/j.cclet.2024.110591There is no corresponding record for this reference.
- 44Cui, Z.; Wang, D.; Guo, J.; Nian, Q.; Ruan, D.; Fan, J.; Ma, J.; Li, L.; Dong, Q.; Luo, X. Push-Pull Electrolyte Design Strategy Enables High-Voltage Low-Temperature Lithium Metal Batteries. J. Am. Chem. Soc. 2024, 146 (40), 27644– 27654, DOI: 10.1021/jacs.4c09027There is no corresponding record for this reference.
- 45Choi, N.-S.; Kim, S.-S.; Yew, K.-H.; Lee, K.-Y. Lithium Secondary Battery. U.S. Patent No. 482,512, Feb 15, 2007.There is no corresponding record for this reference.
- 46Choi, N.-S.; Lee, Y. M.; Seol, W.; Lee, J. A.; Park, J.-K. Protective Coating of Lithium Metal Electrode for Interfacial Enhancement with Gel Polymer Electrolyte. Solid State Ionics 2004, 172 (1–4), 19– 24, DOI: 10.1016/j.ssi.2004.05.008There is no corresponding record for this reference.
- 47Luo, Z.; Li, S.; Yang, L.; Tian, Y.; Xu, L.; Zou, G.; Hou, H.; Wei, W.; Chen, L.; Ji, X. Interfacially Redistributed Charge for Robust Lithium Metal Anode. Nano Energy 2021, 87, 106212, DOI: 10.1016/j.nanoen.2021.106212There is no corresponding record for this reference.
- 48Li, W.; Wu, G.; Araújo, C. M.; Scheicher, R. H.; Blomqvist, A.; Ahuja, R.; Xiong, Z.; Feng, Y.; Chen, P. Li+ Ion Conductivity and Diffusion Mechanism in α-Li3N and β-Li3N. Energy Environ. Sci. 2010, 3 (10), 1524– 1530, DOI: 10.1039/c0ee00052cThere is no corresponding record for this reference.
- 49Lacivita, V.; Artrith, N.; Ceder, G. Structural and Compositional Factors that Control The Li-Ion Conductivity in LiPON Electrolytes. Chem. Mater. 2018, 30 (20), 7077– 7090, DOI: 10.1021/acs.chemmater.8b02812There is no corresponding record for this reference.
- 50Lee, J. A.; Kim, S.; Cho, Y.; Kweon, S. H.; Kang, H.; Byun, J. H.; Kwon, E.; Seo, S.; Kim, W.; Ryu, K. H. Compositionally Sequenced Interfacial Layers for High-Energy Li-Metal Batteries. Adv. Sci. 2024, 11 (17), 2310094, DOI: 10.1002/advs.202310094There is no corresponding record for this reference.
- 51Song, C.; Han, S. H.; Choi, Y.; Shin, H. R.; Kim, M. K.; Gong, C.; Chen, D.; Lee, J. W.; Hong, S.; Choi, N. S. Geometric Design of Interface Structures and Electrolyte Solvation Chemistry for Fast Charging Lithium-Ion Batteries. Adv. Mater. 2025, 37 (19), 2418773, DOI: 10.1002/adma.202418773There is no corresponding record for this reference.
- 52Bommier, C.; Ji, X. Electrolytes, SEI Formation, and Binders: A Review of Nonelectrode Factors for Sodium-Ion Battery Anodes. Small 2018, 14 (16), 1703576, DOI: 10.1002/smll.201703576There is no corresponding record for this reference.
- 53Besenhard, J.; Winter, M.; Yang, J.; Biberacher, W. Filming Mechanism of Lithium-Carbon Anodes in Organic and Inorganic Electrolytes. J. Power Sources 1995, 54 (2), 228– 231, DOI: 10.1016/0378-7753(94)02073-CThere is no corresponding record for this reference.
- 54Shi, P.; Lin, M.; Zheng, H.; He, X.; Xue, Z.; Xiang, H.; Chen, C. Effect of Propylene Carbonate-Li+ Solvation Structures on Graphite Exfoliation and Its Application in Li-Ion Batteries. Electrochim. Acta 2017, 247, 12– 18, DOI: 10.1016/j.electacta.2017.06.174There is no corresponding record for this reference.
- 55Kim, H.; Yoon, G.; Lim, K.; Kang, K. A Comparative Study of Graphite Electrodes using The Co-Intercalation Phenomenon for Rechargeable Li, Na and K Batteries. Chem. Commun. 2016, 52 (85), 12618– 12621, DOI: 10.1039/C6CC05362AThere is no corresponding record for this reference.
- 56Kim, H.; Lim, K.; Yoon, G.; Park, J. H.; Ku, K.; Lim, H. D.; Sung, Y. E.; Kang, K. Exploiting Lithium-Ether Co-Intercalation in Graphite for High-Power Lithium-Ion Batteries. Adv. Energy Mater. 2017, 7 (19), 1700418, DOI: 10.1002/aenm.201700418There is no corresponding record for this reference.
- 57Lai, P.; Zhang, Y.; Liu, J.; Zhang, Z.; Xie, H.; Li, X.; Deng, X.; Huang, B.; Zhang, P.; Zhao, J. Solvation-Structure-Preserved Electrolyte Breaks the Low Temperature Barrier for Sodium Metal Battery. J. Energy Chem. 2025, 107, 852– 860, DOI: 10.1016/j.jechem.2025.05.010There is no corresponding record for this reference.
- 58Huang, Z.; Xiao, Z.; Zhang, H.; Zhang, Q.; Cui, J.; Luo, J.; Tang, W.; Wu, Y. Temperature-Robust Solvation Enabled by Solvent Interactions for Low-Temperature Sodium Metal Batteries. J. Am. Chem. Soc. 2025, 147 (6), 5162– 5171, DOI: 10.1021/jacs.4c15478There is no corresponding record for this reference.
- 59Shen, Y.; Xu, J.; Li, Y.; Che, H.; Zhao, S.; Ishaq, M.; Jabeen, M.; Zhang, Y.; Wu, J.; Li, J. Anion-Cation Synergistic Interactions for Low-Temperature and Fast-Charging Performance in Sodium Batteries. J. Mater. Chem. A 2025, 13 (25), 19631– 19643, DOI: 10.1039/D5TA02276BThere is no corresponding record for this reference.
- 60Zhou, X.; Huang, Y.; Wen, B.; Yang, Z.; Hao, Z.; Li, L.; Chou, S.-L.; Li, F. Regulation of Anion-Na+ Coordination Chemistry in Electrolyte Solvates for Low-Temperature Sodium-Ion Batteries. Proc. Natl. Acad. Sci. U. S. A. 2024, 121 (5), e2316914121 DOI: 10.1073/pnas.2316914121There is no corresponding record for this reference.
- 61Kim, S. C.; Wang, J.; Xu, R.; Zhang, P.; Chen, Y.; Huang, Z.; Yang, Y.; Yu, Z.; Oyakhire, S. T.; Zhang, W. High-Entropy Electrolytes for Practical Lithium Metal Batteries. Nat. Energy 2023, 8 (8), 814– 826, DOI: 10.1038/s41560-023-01280-1There is no corresponding record for this reference.
- 62Yang, C.; Liu, X.; Lin, Y.; Yin, L.; Lu, J.; You, Y. Entropy-Driven Solvation toward Low-Temperature Sodium-Ion Batteries with Temperature-Adaptive Feature. Adv. Mater. 2023, 35 (28), 2301817, DOI: 10.1002/adma.202301817There is no corresponding record for this reference.
- 63Zheng, C.; Yao, Q.; Sun, Y.; Lv, H.; Bai, Z.; Zhang, G.; Wang, N.; Yang, J. Enhancing Low-Temperature Performance of Sodium-Ion Batteries via Anion-Solvent Interactions. Adv. Funct. Mater. 2025, 2501303, DOI: 10.1002/adfm.202501303There is no corresponding record for this reference.
- 64Fang, H.; Huang, Y.; Hu, W.; Song, Z.; Wei, X.; Geng, J.; Jiang, Z.; Qu, H.; Chen, J.; Li, F. Regulating Ion-Dipole Interactions in Weakly Solvating Electrolyte towards Ultra-Low Temperature Sodium-Ion Batteries. Angew. Chem. 2024, 136 (15), e202400539 DOI: 10.1002/ange.202400539There is no corresponding record for this reference.
- 65Cui, Y.; Ni, Y.; Wang, Y.; Wang, L.; Yang, W.; Wu, S.; Xie, W.; Zhang, K.; Yan, Z.; Chen, J. A Temperature-Adapted Ultraweakly Solvating Electrolyte for Cold-Resistant Sodium-Ion Batteries. Adv. Energy Mater. 2025, 15 (21), 2405363, DOI: 10.1002/aenm.202405363There is no corresponding record for this reference.
- 66Cui, K.; Hou, R.; Zhou, H.; Guo, S. Electrolyte Engineering of Hard Carbon for Sodium-Ion Batteries: From Mechanism Analysis to Design Strategies. Adv. Funct. Mater. 2025, 35 (16), 2419275, DOI: 10.1002/adfm.202419275There is no corresponding record for this reference.
- 67Ma, X.-X.; Shen, X.; Chen, X.; Fu, Z.-H.; Yao, N.; Zhang, R.; Zhang, Q. The Origin of Fast Lithium-Ion Transport in The Inorganic Solid Electrolyte Interphase on Lithium Metal Anodes. Small Struct. 2022, 3 (8), 2200071, DOI: 10.1002/sstr.202200071There is no corresponding record for this reference.
- 68Wang, Y.; Hou, H.; Tantratian, K.; Goncharova, L. V.; Fu, B.; Jin, E.; Pirayesh, P.; Abdolvand, H.; Pang, X.; Chen, L. Insight into the Interface Design for Li Metal Anode: Organic-Rich or Inorganic-Rich. Adv. Funct. Mater. 2024, 34 (46), 2406426, DOI: 10.1002/adfm.202406426There is no corresponding record for this reference.
- 69Dong, R.; Zheng, L.; Bai, Y.; Ni, Q.; Li, Y.; Wu, F.; Ren, H.; Wu, C. Elucidating The Mechanism of Fast Na Storage Kinetics in Ether Electrolytes for Hard Carbon Anodes. Adv. Mater. 2021, 33 (36), 2008810, DOI: 10.1002/adma.202008810There is no corresponding record for this reference.
- 70Ma, M.; Cai, H.; Xu, C.; Huang, R.; Wang, S.; Pan, H.; Hu, Y. S. Engineering Solid Electrolyte Interface at Nano-Scale for High-Performance Hard Carbon in Sodium-Ion Batteries. Adv. Funct. Mater. 2021, 31 (25), 2100278, DOI: 10.1002/adfm.202100278There is no corresponding record for this reference.
- 71Kwon, H.; Kim, S.; Hyun, J.; Lee, H. E.; Kim, S. S.; Kim, Y.; Kim, I. J.; Shin, K.; Kim, S.; Park, C. Covariance of Interphasic Properties and Fast Chargeability of Energy-Dense Lithium Metal Batteries. Nat. Energy 2025, 10, 1132, DOI: 10.1038/s41560-025-01838-1There is no corresponding record for this reference.
- 72Hirsh, H. S.; Sayahpour, B.; Shen, A.; Li, W.; Lu, B.; Zhao, E.; Zhang, M.; Meng, Y. S. Role of Electrolyte in Stabilizing Hard Carbon as An Anode for Rechargeable Sodium-Ion Batteries with Long Cycle Life. Energy Storage Mater. 2021, 42, 78– 87, DOI: 10.1016/j.ensm.2021.07.021There is no corresponding record for this reference.
- 73Ji, Y.; Qiu, J.; Zhao, W.; Liu, T.; Dong, Z.; Yang, K.; Zheng, G.; Qian, G.; Yang, M.; Chen, Q. In Situ Probing The Origin of Interfacial Instability of Na Metal Anode. Chem. 2023, 9 (10), 2943– 2955, DOI: 10.1016/j.chempr.2023.06.002There is no corresponding record for this reference.
- 74Bhattacharya, S.; Alpas, A. T. Micromechanisms of Solid Electrolyte Interphase Formation on Electrochemically Cycled Graphite Electrodes in Lithium-Ion Cells. Carbon 2012, 50 (15), 5359– 5371, DOI: 10.1016/j.carbon.2012.07.009There is no corresponding record for this reference.
- 75Wang, Q.; Zhao, C.; Hu, X.; Wang, J.; Ganapathy, S.; Eustace, S.; Bai, X.; Li, B.; Li, H.; Aurbach, D. Grain-Boundary-Rich Interphases for Rechargeable Batteries. J. Am. Chem. Soc. 2024, 146 (46), 31778– 31787, DOI: 10.1021/jacs.4c10650There is no corresponding record for this reference.
- 76Tian, J.; Chen, N.; Feng, M.; Sun, L.; Yang, B.; Zhang, F.; Wu, F.; Chen, R. Cofactor-Assisted Weakly Solvated Electrolytes for Low-Temperature Charging of Graphite Anode. Adv. Funct. Mater. 2025, 35, 2504252, DOI: 10.1002/adfm.202504252There is no corresponding record for this reference.
- 77Perez-Beltran, S.; Kuai, D.; Balbuena, P. B. SEI Formation and Lithium-Ion Electrodeposition Dynamics in Lithium Metal Batteries via First-Principles Kinetic Monte Carlo Modeling. ACS Energy Lett. 2024, 9 (11), 5268– 5278, DOI: 10.1021/acsenergylett.4c02019There is no corresponding record for this reference.
- 78Mogensen, R.; Brandell, D.; Younesi, R. Solubility of the Solid Electrolyte Interphase (SEI) in Sodium Ion Batteries. ACS Energy Lett. 2016, 1 (6), 1173– 1178, DOI: 10.1021/acsenergylett.6b00491There is no corresponding record for this reference.
- 79Liu, J.; Hao, W.; Fang, M.; Chen, X.; Dong, Y.; Chen, Y.; Wang, Z.; Yue, X.; Liang, Z. Screening of F-containing Electrolyte Additives and Clarifying Their Decomposition Routes for Stable Li Metal Anodes. Nat. Commun. 2024, 15 (1), 9356, DOI: 10.1038/s41467-024-53807-zThere is no corresponding record for this reference.
- 80Cai, J.; Wang, A.; Li, S.; Wu, Z.; Jiang, T.; Wang, W.; Fan, W.; Kang, T.; Xiao, X.; Nan, J. A Hard-Soft Synergy Strategy Enables Drastic Temperature Performance of Pouch Sodium-Ion Batteries over Wide-Temperature Range of −30 to 60 °C. Energy Storage Materials 2025, 75, 103997, DOI: 10.1016/j.ensm.2024.103997There is no corresponding record for this reference.
- 81Zhang, Y.; Wu, Z.; Wang, Y.; Chen, G.; Yang, K.; Li, L.; Liu, X.; Ma, Z.; Chen, L. Revisiting Interfacial Enhancement Effects of Fluorinated Cyclophosphazene in Practical Sodium-Ion Batteries. ACS Appl. Mater. & Interfaces 2025, 17 (21), 31553– 31560, DOI: 10.1021/acsami.5c02243There is no corresponding record for this reference.
- 82Liang, H.-J.; Qian, W.-Y.; Liu, H.-H.; Wang, X.-T.; Gu, Z.-Y.; Dong, F.; Deng, Y.; Tang, Y.-Z.; Zhang, J.; Zhao, J. Sulfite-Based Electrolyte Chemistry with Ion-Dipole Interactions and Robust Interphase Achieves Wide-Temperature Sodium-Ion Batteries. J. Am. Chem. Soc. 2025, 147 (21), 17860– 17870, DOI: 10.1021/jacs.5c01864There is no corresponding record for this reference.
- 83Li, S.; Liu, H.; Qiu, H.; He, X.; Fan, W.; Wang, W.; Nan, J. An Electrolyte Design Strategy for Commercial All-Climate Pouch Sodium-Ion Batteries. Chem. Eng. J. 2025, 505, 159102, DOI: 10.1016/j.cej.2024.159102There is no corresponding record for this reference.
- 84Hu, R.; Yang, L.; Zhang, C.; Chen, B.; Hong, H.; Shen, H.; Mao, C.; Yang, Z.; Zhang, S.; Huang, Q. Bifunctional Sodium Tetrakis [3,5-bis(trifluoromethyl)phenyl] Borate Additive for Long-Lifespan Sodium-Ion Batteries with NaNi0.33Fe0.33Mn0.33O2 Cathode. Chem. Eng. J. 2025, 512, 162144, DOI: 10.1016/j.cej.2025.162144There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c02918.
Coordination number of electrolytes, chemical structures, and full names of solvents and anions (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



