Transition-Metal-Doped Halide Perovskites for Near-Infrared Emissions: Beyond OxidesClick to copy article linkArticle link copied!
- Sajid Saikia*Sajid Saikia*[email protected]Department of Chemistry, Indian Institute of Science Education and Research (IISER), Pune 411008, IndiaMore by Sajid Saikia
- Animesh GhoshAnimesh GhoshDepartment of Chemistry, Indian Institute of Science Education and Research (IISER), Pune 411008, IndiaMore by Animesh Ghosh
- Angshuman Nag*Angshuman Nag*[email protected]Department of Chemistry, Indian Institute of Science Education and Research (IISER), Pune 411008, IndiaMore by Angshuman Nag
Abstract
Transition-metal-doped oxide-based phosphors led to near-infrared (NIR) light-emitting diodes and lasers. Mostly, the first transition series (3d) metal ions, like Cr3+, are doped. Interestingly, heavier transition-metal ions, like Mo3+, Re4+, and Os4+, in their low oxidation states, can be doped in halide perovskites, unlike the oxide hosts that stabilize the higher oxidation state. Consequently, doping heavier transition metal ions in halide hosts opens up new avenues to tailor NIR-I/II emissions for both narrow and broad-band d–d transitions. Furthermore, characteristics such as (i) tunability of crystal field parameters by halide ions, (ii) host emissions, and (iii) colloidal nanocrystal synthesis add new NIR functionalities to transition-metal-doped halide perovskites compared to oxides. Here we provide insights on recent progresses on the NIR emitting transition-metal-doped halides, connecting synthetic materials chemistry, spectroscopy and device fabrication. The present challenges and future opportunities are discussed.
This publication is licensed for personal use by The American Chemical Society.
Special Issue
Published as part of ACS Energy Letters special issue “The Evolving Landscape of Energy Research: Insights from Leading Researchers”.
Figure 1
Figure 1. Schematic highlights the scope of transition metal doping in halide perovskites (and related structures) for NIR emission, which is typically beyond the scope of oxide-based NIR phosphors.
Low Oxidation States of Dopants and Spin-Flip Transition
Mo3+ Doping
Figure 2
Figure 2. (a) Schematic representation of Mo3+-doped Cs2NaInCl6 double perovskite cubic lattice with the space group Fm3m. (b) Description of the Cl environment around Mo (red open symbols) and In (blue open symbols) for Mo3+-doped Cs2NaInCl6 with different dopant concentrations: coordination number (CN, top panel), bond length (R, middle panel), and local order (σ2, bottom panel). (c) d–d vs f–f transition-based NIR-II PL spectra from Ln3+- (Ln = Yb, Er) and Mo3+-doped Cs2NaInCl6. (d) Left panel: Tanabe-Sugano diagram for a d3 ion in an octahedral coordination; middle panel: schematic diagram comparing the orbital interaction between the metal (M) dxy ligand (L) px when the metal ion (d3: Cr3+, Mo3+, Re4+) is in the ground state (GS: 4A2g) and in an excited state (ES: 2Eg); right panel: schematic potential energy surface diagram, indicating that the ground state 4A2g, and excited states 2Eg/2T1g and 2T2g have the same t2g3eg0 electronic configuration and, therefore, have very similar M–L bond lengths. Reproduced with permission from ref (20). Copyright 2025 Wiley.
Figure 3
Figure 3. (a) Temperature-dependent (7–300 K) PL spectra of 3.8% Mo3+-doped Cs2NaInCl6 in the visible to NIR region under 438 nm excitation. Reproduced with permission from ref (20). Copyright 2025 Wiley. (b) PL and PL excitation spectra of Cs2HfCl6:Re4+ at room temperature. Reproduced with permission from ref (26). Copyright 2025 Elsevier. (c) PL spectrum of Cs2HfCl6:Os4+ crystals at 2 K. Since the spectrum was collected in a broad range from visible to NIR-II (∼580–2100 nm), the relative detector response is also shown. Reproduced with permission from ref (14). Copyright 1971 American Physical Society.
Re4+, Os4+, and V3+ Doping
Debate on [MX6]2– Octahedra vs [MOX5]2– Distorted C4v Centers (M: Mo, W, V, etc.)
Figure 4
Figure 4. (a) Crystal structure of Cs2MoCl6 containing a [MoCl6]2– unit with an octahedral symmetry (top) followed by the Tanabe-Sugano diagram as reference for showing possible electronic transitions of Mo4+ (4d2) in an octahedral local symmetry (bottom). The crystal structure was obtained using a CIF file reported in ref (42). (b) Crystal structure of Cs2MoOxCl6–x containing the [MoOCl6]2– unit with reduced C4v symmetry (top) followed by possible electronic transitions of Mo5+ (4d1) (bottom). The crystal structure was obtained using the CIF file reported in ref (35).
Ultrabroad NIR Emission Combining Host and Dopant Emission
Figure 5
Figure 5. (a) PL spectra of undoped, 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6 under the excitation of 350 nm, showing unprecedented spectral widths of 434 and 468 nm, respectively. The spectra were collected using two detectors and then combined at 720 nm. (b) fwhm of various reported broadband NIR emitters is compared with that of 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6. The composition of various NIR emitters is mentioned in the figure with serial numbers that correspond to a table reported in the Supporting Information of ref (46). The position of the tringles indicates the PL peak position(s) of the broadband NIR PL, while the solid and dashed lines show the emission wavelength range coverage with continuity and discontinuity, respectively. The colors of the triangle indicate various dopants, as stated at the top of the figure. Different PL peak for a single dopant due to occupation of two different lattice sites is indicated by tringle of same color with dashed and solid edges. (c) Schematics summarizing the excitation and emission pathways for ultrabroadband NIR emissions from W-doped and Mo-doped Cs2Na0.95Ag0.05BiCl6. (d) PL and PL excitation of 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6 at 7 K. Reproduced with permission from ref (49). Copyright 2025 Wiley.
Halide Ion Dependent Tunable NIR Emission
Figure 6
Figure 6. (a) NIR PL of Ga1.98–x(Al0.68In0.32)xO3:2% Cr3+ (x = 0–0.8) at room temperature. Reproduced with permission from ref (57). Copyright 2022 American Chemical Society. (b) Normalized absorption (violet), PL emission (orange), and PL excitation (blue) spectra of Cs2AgInCl6:1.3% Cr3+ nanocrystals. Reproduced with permission from ref (58). Copyright 2022 American Chemical Society. (c) Low-temperature (4 K) PL spectra of CrX3 (X = Cl, Br, I) showing broad PL due to 4T2g → 4A2g, and corresponding Yb3+-doped CrX3 showing sharp PL due to 2F5/2 → 2F7/2 transition of Yb3+. The absence of lattice PL in the latter indicates efficient ET from Cr3+ to Yb3+. Reproduced with permission from ref (59). Copyright 2023 American Chemical Society. (d, e) NIR to visible upconversion (UC) photoluminescence of Re4+-doped Cs2NaYCl6 and Cs2NaYBr6 at 15 K respectively. Reproduced with permission from ref (17). Copyright 1998 American Chemical Society.
Colloidal Nanocrystals
Figure 7
Figure 7. (a) Hot-injection synthesis setup for colloidal nanocrystals. (b) Precursors and reaction conditions used to prepare (top panel) Cr3+-doped Cs2NaxAg1–xInCl6 (x = 0–1) and (bottom panel) Cs2MoCl6 nanocrystals. (c, d) TEM images of Cr3+-doped Cs2NaInCl6 nanocrystals. Inset images of (c) and (d) show high resolution TEM and size distribution of nanocrystals. (e) Room temperature PL of Cr3+-doped Cs2NaxAg1–xInCl6 (x = 0 −1) colloidal nanocrystals under different excitations. (c)–(e) were reproduced with permission from ref (58). Copyright 2022 American Chemical Society. (f–g) TEM and high-resolution TEM images of Cs2MoCl6 nanocrystals. Reproduced with permission from ref (77). Copyright 2024 American Chemical Society.
Potential Applications
Phosphor-Converted LED (PC-LED) and NIR Imaging
Figure 8
Figure 8. (a) Output power for NIR emission with a peak at ∼980 nm as a function of applied current for a Cs2MCl6 (M = Mo/W) based pc-LED panel. The phosphor was coated with UV chips. Reproduced with permission from ref (34). Copyright 2022 Wiley. (b) Luminescence spectra as a function of biased voltages for a pc-LED fabricated by coating Cs2MoCl6 phosphor composite on a far-red (730 nm) LED chip. Reproduced with permission from ref (42). Copyright 2024 American Chemical Society. (c) Luminescence spectra of 2.5% W-doped Cs2Na0.95Ag0.4BiCl6 NIR pc-LEDs at different driving currents. (d) The composite 2.5% W-doped Cs2Na0.95Ag0.4BiCl6 and poly(lactic acid) forming a 3D printed structure over the UV-LED panel. The 3D printed phosphor composites yield better thermal stability for the prolonged operation of the NIR-pc-LED. (c, d) Reproduced with permission from ref (49). Copyright 2025 Wiley. (e) Luminescence spectra of the pc-LED fabricated by coating a polymer composite of 3.8% Mo3+-doped Cs2NaInCl6 on a blue LED chip. The schematics of fabrication and digital photographs of the devices are shown in the insets. Reproduced with permission from ref (20). Copyright 2025 Wiley. (f) pc-LED panel and NIR imaging for food freshness detection using 2% Cr3+-0.5%Bi3+-codoped Cs2Ag0.6Na0.4InCl6. Images of a pair of apples were collected by a visible camera and NIR camera under illumination from pc-LED. The NIR image could detect the early rotting stage of an apple, as shown with a red circle. Reproduced with permission from ref (79). Copyright 2023 Wiley.
| composition | PL peak positions (nm) | fwhm (nm) | PLQY (%) | pc-LED performance and application | ref |
|---|---|---|---|---|---|
| Mo3+-doped Cs2NaInCl6 | 700, 1095 | 9 | P = 49 mW at 300 mA; ηE–O = 7.5% and ηO–O = 13% at 10 mA | (20) | |
| Cs2MoCl6 | ∼1000 | 200 | 26 | P = 12 W at 350 mA; ηE–O ∼ 13% | (34) |
| deep penetration NIR imaging | |||||
| Cs2MCl6 (M = Mo, W) | 986, 965 | 200, 170 | P = 2 mW at 160 mA | (42) | |
| Mo/W-doped Cs2(AgNa)BiCl6 | 700, 950 | 265, 200/169 | 40 | P = 6.2 mW at 475 mA | (49) |
| 3D printed pc-LED for NIR Imaging | |||||
| Mo-doped Cs2MCl6 (M = Zr, Te) | 950 | 200 | 29 | pc-LED | (37) |
| NIR Imaging | |||||
| Mo-doped Cs2ZnCl4 | 960 | 206 | P = 132 mW at 350 mA | (81) | |
| IQE = 78.7% | |||||
| NIR imaging for food freshness detection | |||||
| W-doped Cs2SnCl6 | 965 | 160 | P = 10 mW at 400 mA | (78) | |
| Mo/Er-codoped Cs2NaBiCl6 | 970, 1540 | 211, 20 | 69 | P = 80 mW at 300 mA | (51) |
| ηE–O ∼ 13% | |||||
| anticounterfeiting; nondestructive inspection | |||||
| Re4+-doped Cs2MCl6 (M = Hf, Te) | 726, 1342, 1600 | 16, 80, 100 | 92 | IQE = 91.9% | (26,27) |
| deep penetration NIR imaging | |||||
| Os4+-doped Cs2HfCl6 | 591, 714, 833, 1000, 1316, 1818 | ∼100–50 (at 2 K) | (14) | ||
| Cs2MoCl6 nanocrystals | 965 | 190 | (77) |
The NIR emission arises from d–d transitions of 4dn and 5dn (n = 1–3) outermost electronic configuration of Mo3+/5+, W5+, and Re4+ ions, which are stable in ambient conditions. P = output power; ηE–O and ηO–O electrical to optical and optical to optical power conversion efficiency, respectively; IQE = internal quantum efficiency.
NIR Electroluminescence LED?
Lasing Applications?
Biographies
Sajid Saikia
Sajid Saikia received his B.Sc. degree in chemistry from Tezpur University. He completed his M.S. and Ph.D. dual degree from the Indian Institute of Science Education and Research (IISER) Pune (2025). His research interests focus on the design of halide perovskites for near-infrared luminescence, photophysical investigations and upconversion luminescence studies.
Animesh Ghosh
Animesh Ghosh completed his M.Sc. from The University of Burdwan. Currently, he is pursuing his Ph.D. at IISER Pune, India. His research focuses on transition metal-based perovskites and their derivatives for near-infrared luminescence and potential application in devices.
Angshuman Nag
Angshuman Nag is a Professor of Chemistry at IISER Pune. He earned his Ph.D. from Indian Institute of Science Bengaluru (2009) and completed two postdoctoral terms. Subsequently, he joined IISER Pune in 2012. His recent research focuses on halide perovskite semiconductors and colloidal quantum dots for optoelectronic applications.
Acknowledgments
We thank our collaborators including past and present members of research group, particularly, Dr. Barnali Mondal, Mr. Vijay Mani Tripathi, Dr. Srijita Banerjee and Dr. Habibul Arfin, for their research contributions and insightful discussion that helped enhance our understanding. S.S. thanks Prime Minister Research Fellowship (PMRF) and A.G. thanks University Grants Commission (UGC) for fellowships. A.N. acknowledges Anusandhan National Research Foundation (ANRF) (Swarnajayanti Fellowship, SB/SJF/2020-21/02), India.
References
This article references 84 other publications.
- 1Shionoya, S.; Yen, W. M.; Yamamoto, H. Phosphor Handbook; CRC Press, 2018.Google ScholarThere is no corresponding record for this reference.
- 2Geusic, J. E.; Marcos, H. M.; Van Uitert, L. G. Laser Oscillations in Nd-doped Yttrium Aluminium, Yttrium Gallium and Gadolanium Garnets. Appl. Phys. Lett. 1964, 4 (10), 182– 184, DOI: 10.1063/1.1753928Google ScholarThere is no corresponding record for this reference.
- 3Maiman, T. H. Stimulated Optical Radiation in Ruby. Nature 1960, 187 (4736), 493– 494, DOI: 10.1038/187493a0Google ScholarThere is no corresponding record for this reference.
- 4Mir, W. J.; Sheikh, T.; Arfin, H.; Xia, Z.; Nag, A. Lanthanide doping in metal halide perovskite nanocrystals: spectral shifting, quantum cutting and optoelectronic applications. NPG Asia Mater. 2020, 12 (1), 9, DOI: 10.1038/s41427-019-0192-0Google ScholarThere is no corresponding record for this reference.
- 5Wang, N.; Zhuang, B.; Pang, T.; Zeng, L.; Chen, D. Recent Advances in Lanthanide-Doped Perovskite Nanocrystals. Laser Photonics Rev. 2025, 19 (18), e00905 DOI: 10.1002/lpor.202500905Google ScholarThere is no corresponding record for this reference.
- 6Nag, A.; Sapra, S.; Gupta, S. S.; Prakash, A.; Ghangrekar, A.; Periasamy, N.; Sarma, D. D. Luminescence in Mn-doped CdS nanocrystals. Bull. Mater. Sci. 2008, 31 (3), 561– 568, DOI: 10.1007/s12034-008-0087-0Google ScholarThere is no corresponding record for this reference.
- 7Santra, P. K.; Kamat, P. V. Mn-Doped Quantum Dot Sensitized Solar Cells: A Strategy to Boost Efficiency over 5%. J. Am. Chem. Soc. 2012, 134 (5), 2508– 2511, DOI: 10.1021/ja211224sGoogle ScholarThere is no corresponding record for this reference.
- 8Wei, Y.; Dang, P.; Dai, Z.; Li, G.; Lin, J. Advances in Near-Infrared Luminescent Materials without Cr3+: Crystal Structure Design, Luminescence Properties, and Applications. Chem. Mater. 2021, 33 (14), 5496– 5526, DOI: 10.1021/acs.chemmater.1c01325Google ScholarThere is no corresponding record for this reference.
- 9Dang, P.; Wei, Y.; Liu, D.; Li, G.; Lin, J. Recent Advances in Chromium-Doped Near-Infrared Luminescent Materials: Fundamentals, Optimization Strategies, and Applications. Adv. Opt. Mater. 2023, 11 (3), 2201739, DOI: 10.1002/adom.202201739Google ScholarThere is no corresponding record for this reference.
- 10Chen, C.; Chang, J.; Chen, R.; Gao, R.; Wang, Y.; Zhu, K.; Xiang, J.; Guo, C. Strategies for broadening the emission spectra of Cr3+-doped near-infrared emitting phosphors. Mater. Chem. Front. 2025, 9 (12), 1821– 1838, DOI: 10.1039/D4QM01109KGoogle ScholarThere is no corresponding record for this reference.
- 11Guria, A. K.; Dutta, S. K.; Adhikari, S. D.; Pradhan, N. Doping Mn2+ in Lead Halide Perovskite Nanocrystals: Successes and Challenges. ACS Energy Lett. 2017, 2 (5), 1014– 1021, DOI: 10.1021/acsenergylett.7b00177Google ScholarThere is no corresponding record for this reference.
- 12Senden, T.; van Dijk-Moes, R. J. A.; Meijerink, A. Quenching of the red Mn4+ luminescence in Mn4+-doped fluoride LED phosphors. Light Sci. Appl. 2018, 7 (1), 8, DOI: 10.1038/s41377-018-0013-1Google ScholarThere is no corresponding record for this reference.
- 13Huang, W.; Li, C.; Zeng, L.; Zhang, J.; Kurochkin, M. A.; Kolesnikov, I. E.; Umar, Z.; Zhang, J.; Liu, W.; Kukovecz, Á.; Kurboniyon, M. S.; Zhang, X. One Shoot Two Birds: Stabilization of Mn5+ in Ba2(Si,Ge)O4 as an Efficient NIR Thermometer and Intense Turquoise Pigment. Inorg. Chem. 2025, 64 (24), 12100– 12111, DOI: 10.1021/acs.inorgchem.5c01428Google ScholarThere is no corresponding record for this reference.
- 14Reinberg, A. R. Sharp-Line Luminescence from Os4+ and Mo3+ in Cs2HfCl6. Phys. Rev. B 1971, 3 (1), 41– 46, DOI: 10.1103/PhysRevB.3.41Google ScholarThere is no corresponding record for this reference.
- 15Flint, C. D.; Paulusz, A. G. Infrared and visible luminescence spectra of MoCl63- and MoBr63- in cubic elpasolite crystals. Mol. Phys. 1981, 44 (4), 925– 938, DOI: 10.1080/00268978100102891Google ScholarThere is no corresponding record for this reference.
- 16Stranger, R.; Moran, G. M.; Krausz, E.; Güdel, H. U.; Furer, N. Octahedral monomeric molybdenum(III). A magneto-optical study of Mo3+ doped in Cs2NaYCl6. Mol. Phys. 1990, 69, 11– 31, DOI: 10.1080/00268979000100021Google ScholarThere is no corresponding record for this reference.
- 17Gamelin, D. R.; Güdel, H. U. Two-Photon Spectroscopy of d3 Transition Metals: Near-IR-to-Visible Upconversion Luminescence by Re4+ and Mo3+. J. Am. Chem. Soc. 1998, 120 (46), 12143– 12144, DOI: 10.1021/ja982742mGoogle ScholarThere is no corresponding record for this reference.
- 18Gamelin, D. R.; Güdel, H. U. Excited-State Dynamics and Sequential Two-Photon Upconversion Excitation of Mo3+-Doped Chloro- and Bromo-elpasolites. J. Phys. Chem. B 2000, 104 (44), 10222– 10234, DOI: 10.1021/jp002132kGoogle ScholarThere is no corresponding record for this reference.
- 19Wenger, O. S.; Güdel, H. U. Broadband near-Infrared Sensitization of Visible Upconversion Luminescence in V3+ and Mo3+ Co-Doped Cs2NaYCl6. J. Phys. Chem. B 2002, 106 (39), 10011– 10019, DOI: 10.1021/jp021076fGoogle ScholarThere is no corresponding record for this reference.
- 20Ghosh, A.; Saikia, S.; Mukherjee, S.; Johannesson, E.; Rensmo, H.; Nag, A. Intra-Configurational Spin-Flip d→d Transition of Mo(III) Doped Perovskite for Ultra-Narrow Near Infrared-II Emission in Ambient Conditions. Angew. Chem., Int. Ed. 2025, 64 (52), e19144 DOI: 10.1002/anie.202519144Google ScholarThere is no corresponding record for this reference.
- 21Binwal, D. C.; Mudoi, P. P.; Panda, D. P.; Vishnoi, P. Molybdenum chloride double perovskites: dimensionality control of optical and magnetic properties. Chem. Sci. 2023, 14 (15), 3982– 3989, DOI: 10.1039/D3SC00132FGoogle ScholarThere is no corresponding record for this reference.
- 22Binwal, D. C.; Anand, K.; Sharma, M.; Saurav, K. V.; Pati, S. K.; Vishnoi, P. Tailorable Magnetic Exchange and Optical Absorption in 1-D Double Perovskite-like Halides (CH3NH3)2NaMoCl(6–x)Brx. Chem. Mater. 2024, 36 (16), 7719– 7730, DOI: 10.1021/acs.chemmater.4c00735Google ScholarThere is no corresponding record for this reference.
- 23Kitzmann, W. R.; Moll, J.; Heinze, K. Spin-flip luminescence. Photochem. Photobiol. Sci. 2022, 21 (7), 1309– 1331, DOI: 10.1007/s43630-022-00186-3Google ScholarThere is no corresponding record for this reference.
- 24Kitzmann, W. R.; Heinze, K. Charge-Transfer and Spin-Flip States: Thriving as Complements. Angew. Chem., Int. Ed. 2023, 62 (15), e202213207 DOI: 10.1002/anie.202213207Google ScholarThere is no corresponding record for this reference.
- 25Gamelin, D. R.; Güdel, H. U. Spectroscopy and Dynamics of Re4+ Near-IR-to-Visible Luminescence Upconversion. Inorg. Chem. 1999, 38 (22), 5154– 5164, DOI: 10.1021/ic990556rGoogle ScholarThere is no corresponding record for this reference.
- 26Fan, Z.; Liu, Y.; Gao, Y.; Xie, J.; Zhao, I. S.; Wang, Y. A novel efficient lead-free near-infrared emission Re4+-doped double perovskite for dental caries medical imaging. Chem. Eng. J. 2025, 524, 169762, DOI: 10.1016/j.cej.2025.169762Google ScholarThere is no corresponding record for this reference.
- 27Cai, P.; Li, B.; Zhan, Y.; Feng, X.; Pu, X.; Wang, L.; Chen, Z.; Liu, Z.; Bi, S. Re4+/Te4+ Co-doped Cs2ZrCl6 double perovskite microcrystals: Broadening excitation range and boosting luminescent performance for near-infrared lighting and non-destructive quality inspection. Mater. Today Chem. 2025, 48, 102954, DOI: 10.1016/j.mtchem.2025.102954Google ScholarThere is no corresponding record for this reference.
- 28Reber, C.; Güdel, H. U. Near-infraded luminescence spectroscopy and relaxation behaviour of V3+ doped in Cs2NaYCl6-mBrm (m = 0, 0.3, 3, 6). J. Lumin. 1988, 42 (1), 1– 13, DOI: 10.1016/0022-2313(88)90059-2Google ScholarThere is no corresponding record for this reference.
- 29Gray, H. B.; Hare, C. R. The electronic structures and spectra of chromyl and molybdenyl ions. Inorg. Chem. 1962, 1 (2), 363– 368, DOI: 10.1021/ic50002a034Google ScholarThere is no corresponding record for this reference.
- 30Winkler, J. R.; Gray, H. B. Emission spectroscopic properties of dioxorhenium(V) complexes in crystals and solutions. J. Am. Chem. Soc. 1983, 105 (5), 1373– 1374, DOI: 10.1021/ja00343a052Google ScholarThere is no corresponding record for this reference.
- 31Isovitsch, R. A.; Beadle, A. S.; Fronczek, F. R.; Maverick, A. W. Electronic Absorption Spectra and Phosphorescence of Oxygen-Containing Molybdenum(IV) Complexes. Inorg. Chem. 1998, 37 (17), 4258– 4264, DOI: 10.1021/ic971186eGoogle ScholarThere is no corresponding record for this reference.
- 32Wermuth, M.; Güdel, H. U. Photon Avalanche in Cs2ZrBr6:Os4+. J. Am. Chem. Soc. 1999, 121 (43), 10102– 10111, DOI: 10.1021/ja990847zGoogle ScholarThere is no corresponding record for this reference.
- 33Ghosh, A.; Saikia, S.; Molokeev, M. S.; Nag, A. Synthesis and Optical Properties of Organic-Inorganic Hybrid [(18-Crown-6)K][MoOCl4(H2O)]. Chem.–Asian J. 2025, 20 (1), e202401052 DOI: 10.1002/asia.202401052Google ScholarThere is no corresponding record for this reference.
- 34Liu, Z.; Qin, X.; Chen, Q.; Chen, Q.; Jing, Y.; Zhou, Z.; Zhao, Y. S.; Chen, J.; Liu, X. Highly Stable Lead-Free Perovskite Single Crystals with NIR Emission Beyond 1100 nm. Adv. Opt. Mater. 2022, 10 (21), 2201254, DOI: 10.1002/adom.202201254Google ScholarThere is no corresponding record for this reference.
- 35Morgan, E. E.; Brumberg, A.; Panuganti, S.; Kent, G. T.; Zohar, A.; Mikhailovsky, A. A.; Kanatzidis, M. G.; Schaller, R. D.; Chabinyc, M. L.; Cheetham, A. K.; Seshadri, R. Molecular Origins of Near-Infrared Luminescence in Molybdenum and Tungsten Oxyhalide Perovskites. Chem. Mater. 2024, 36 (16), 7754– 7763, DOI: 10.1021/acs.chemmater.4c00856Google ScholarThere is no corresponding record for this reference.
- 36Siddique, H.; Xu, Z.; Li, X.; Saeed, S.; Liang, W.; Wang, X.; Gao, C.; Dai, R.; Wang, Z.; Zhang, Z. Anomalous Octahedron Distortion of Bi-Alloyed Cs2AgInCl6 Crystal via XRD, Raman, Huang-Rhys Factor, and Photoluminescence. J. Phys. Chem. Lett. 2020, 11 (22), 9572– 9578, DOI: 10.1021/acs.jpclett.0c02852Google ScholarThere is no corresponding record for this reference.
- 37Wang, C.; Wang, J.; Wang, D.; He, L. Efficient near-infrared emission and imaging of lead-free double perovskite Cs2TeCl6 by Mo4+ doping. J. Lumin. 2025, 286, 121381, DOI: 10.1016/j.jlumin.2025.121381Google ScholarThere is no corresponding record for this reference.
- 38Zhang, G.; Li, L.; Mao, Y.; Dang, P.; Zhao, Y.; Suo, H.; Wang, Z.; Li, P.; Cheng, Z.; Lian, H.; Lin, J. Broad-Band Near-Infrared Emission with High External Quantum Efficiency from Molybdenum-Doped Vacancy-Ordered Double Perovskites for Spectroscopic Analysis. ACS Mater. Lett. 2025, 7 (6), 2190– 2198, DOI: 10.1021/acsmaterialslett.5c00334Google ScholarThere is no corresponding record for this reference.
- 39Kumar, S.; Lamba, R. S.; Monga, S.; Jha, V.; Singh, R.; Saha, S.; Bhattacharya, S.; Sapra, S. Broad Dual Emission from Cs2Zr1-xMoxCl6: Enhancing the NIR Emission in Lead-free Vacancy Ordered Double Perovskites. Chem. Mater. 2024, 36 (9), 4561– 4570, DOI: 10.1021/acs.chemmater.4c00231Google ScholarThere is no corresponding record for this reference.
- 40Li, Z.; Xu, M.; Liu, W.; Wang, X.; Li, Y.; Zhou, X.; Fang, Z.; Ning, L. Broadband Near-Infrared Luminescence from Mo4+ in Zero-Dimensional Perovskite Cs2Zr(Cl,Br)6 with an Exceptionally High Quantum Efficiency and Thermal Stability. Chem. Mater. 2024, 36 (2), 901– 910, DOI: 10.1021/acs.chemmater.3c02805Google ScholarThere is no corresponding record for this reference.
- 41Kumar, S.; Lamba, R. S.; Singh, R.; Jha, V.; Yadav, J.; Sapra, S. Efficient Near-Infrared Luminescence with Broad-Band Sensitization in Mo4+-Er3+ Co-Doped Cs2ZrCl6 Vacancy-Ordered Double Perovskites. Small 2025, 21 (46), e07964 DOI: 10.1002/smll.202507964Google ScholarThere is no corresponding record for this reference.
- 42Mondal, B.; Shinde, A.; Rajput, P. K.; Arfin, H.; Tanwar, R.; Ghosh, P.; Nag, A. Vibronically Coupled Near-Infrared Emission and Excitation from d–d Transitions of Cs2MX6 (M = Mo/W, X = Cl/Br). ACS Energy Lett. 2024, 9 (3), 819– 828, DOI: 10.1021/acsenergylett.3c02613Google ScholarThere is no corresponding record for this reference.
- 43Han, Y.; Cheng, X.; Cui, B. B. Factors influencing self-trapped exciton emission of low-dimensional metal halides. Mater. Adv. 2023, 4 (2), 355– 373, DOI: 10.1039/D2MA00676FGoogle ScholarThere is no corresponding record for this reference.
- 44Zhang, G.; Wang, D.; Lou, B.; Ma, C. G.; Meijerink, A.; Wang, Y. Efficient Broadband Near-Infrared Emission from Lead-Free Halide Double Perovskite Single Crystal. Angew. Chem., Int. Ed. 2022, 61 (33), e202207454 DOI: 10.1002/anie.202207454Google ScholarThere is no corresponding record for this reference.
- 45Cheng, X.; Xie, Z.; Zheng, W.; Li, R.; Deng, Z.; Tu, D.; Shang, X.; Xu, J.; Gong, Z.; Li, X.; Chen, X. Boosting the Self-Trapped Exciton Emission in Alloyed Cs2(Ag/Na)InCl6 Double Perovskite via Cu+ Doping. Adv. Sci. 2022, 9 (7), 2103724, DOI: 10.1002/advs.202103724Google ScholarThere is no corresponding record for this reference.
- 46Liu, Y.; Tu, D.; Yang, M.; Li, H.; Li, C.; Xie, Z.; Zhou, S.; Yu, S.; Xu, J.; Chen, X. Near-Infrared and Visible Dual-Band Self-Trapped Exciton Emissions from Li+-Doped Cs2NaScCl6 Double Perovskites. ACS Energy Lett. 2025, 10 (5), 2150– 2159, DOI: 10.1021/acsenergylett.5c00182Google ScholarThere is no corresponding record for this reference.
- 47Grandhi, G. K.; Dhama, R.; Viswanath, N. S. M.; Lisitsyna, E. S.; Al-Anesi, B.; Dana, J.; Sugathan, V.; Caglayan, H.; Vivo, P. Role of Self-Trapped Excitons in the Broadband Emission of Lead-Free Perovskite-Inspired Cu2AgBiI6. J. Phys. Chem. Lett. 2023, 14 (18), 4192– 4199, DOI: 10.1021/acs.jpclett.3c00439Google ScholarThere is no corresponding record for this reference.
- 48Ma, Z.; Lv, P.; He, X.; Wang, F.; Li, Y.; Xiao, G.; Zou, B. Self-Trapped Excitons or Bi3+ Ions for Broad Emission in a Lead-Free Double Perovskite? Hearing What Pressure Says. Nano Lett. 2025, 25 (23), 9345– 9352, DOI: 10.1021/acs.nanolett.5c01709Google ScholarThere is no corresponding record for this reference.
- 49Saikia, S.; Gopal, A.; Rathod, R.; Joshi, A.; Priolkar, K. R.; Saha, S.; Santra, P. K.; Shanmuganathan, K.; Nag, A. Ultrabroad Near Infrared Emitting Perovskites. Angew. Chem., Int. Ed. 2025, 64 (2), e202415003 DOI: 10.1002/anie.202415003Google ScholarThere is no corresponding record for this reference.
- 50Wang, Y.; Dang, P.; Zeng, Z.; Liu, D.; Zhang, G.; Tian, L.; Li, K.; Ma, P. a.; Wei, Y.; Lian, H.; Hou, Z.; Li, G.; Lin, J. Sensitizing effect of lanthanide luminescence by Mo4+/Ag+ in double perovskites: great enhancement of near-infrared emission via wide range of excitation (250–850 nm). Light Sci. Appl. 2026, 15 (1), 87, DOI: 10.1038/s41377-025-02159-4Google ScholarThere is no corresponding record for this reference.
- 51Wang, Z.; Xu, S.; Gu, Y.; Li, Y.; Gao, Y.; Zhu, G.; Wang, Y.; Yu, H.; Chen, B. Enhancement of Blue-Light-Excited NIR-II Emission of Er3+ in Double Perovskites via Mo4+ Sensitization. Laser Photonics Rev. 2025, e02353 DOI: 10.1002/lpor.202502353Google ScholarThere is no corresponding record for this reference.
- 52Rajendran, V.; Fang, M. H.; Guzman, G. N. D.; Lesniewski, T.; Mahlik, S.; Grinberg, M.; Leniec, G.; Kaczmarek, S. M.; Lin, Y. S.; Lu, K. M.; Lin, C. M.; Chang, H.; Hu, S. F.; Liu, R. S. Super Broadband Near-Infrared Phosphors with High Radiant Flux as Future Light Sources for Spectroscopy Applications. ACS Energy Lett. 2018, 3 (11), 2679– 2684, DOI: 10.1021/acsenergylett.8b01643Google ScholarThere is no corresponding record for this reference.
- 53Fang, M. H.; Chen, K. C.; Majewska, N.; Leśniewski, T.; Mahlik, S.; Leniec, G.; Kaczmarek, S. M.; Yang, C. W.; Lu, K. M.; Sheu, H. S.; Liu, R. S. Hidden Structural Evolution and Bond Valence Control in Near-Infrared Phosphors for Light-Emitting Diodes. ACS Energy Lett. 2021, 6 (1), 109– 114, DOI: 10.1021/acsenergylett.0c02373Google ScholarThere is no corresponding record for this reference.
- 54Rajendran, V.; Chen, K. C.; Huang, W. T.; Kamiński, M.; Grzegorczyk, M.; Mahlik, S.; Leniec, G.; Lu, K. M.; Wei, D. H.; Chang, H.; Liu, R. S. Unraveling Luminescent Energy Transfer Pathways: Futuristic Approach of Miniature Shortwave Infrared Light-Emitting Diode Design. ACS Energy Lett. 2023, 8 (5), 2395– 2400, DOI: 10.1021/acsenergylett.3c00680Google ScholarThere is no corresponding record for this reference.
- 55Chen, K. C.; Hsiao, Y. H.; Chien, C. L.; Huang, W. T.; Majewska, N.; Mazurek, M. R.; Leśniewski, T.; Mahlik, S.; Leniec, G.; Cherng, D. H.; Lu, K. M.; Huang, S. L.; Liu, R. S. Bifunctional Energy Efficient (Ga,Ge)2O3:Cr3+,Ni2+ Phosphor for Shortwave Infrared Optical Applications. ACS Energy Lett. 2025, 10 (7), 3050– 3057, DOI: 10.1021/acsenergylett.5c01251Google ScholarThere is no corresponding record for this reference.
- 56Fang, M. H.; Yang, T. H.; Lesniewski, T.; Lee, C.; Mahlik, S.; Grinberg, M.; Peterson, V. K.; Didier, C.; Pang, W. K.; Su, C.; Liu, R. S. Hydrogen-Containing Na3HTi1-xMnxF8 Narrow-Band Phosphor for Light-Emitting Diodes. ACS Energy Lett. 2019, 4 (2), 527– 533, DOI: 10.1021/acsenergylett.8b02307Google ScholarThere is no corresponding record for this reference.
- 57Chang, C. Y.; Majewska, N.; Chen, K. C.; Huang, W. T.; Leśniewski, T.; Leniec, G.; Kaczmarek, S. M.; Pang, W. K.; Peterson, V. K.; Cherng, D. H.; Lu, K. M.; Mahlik, S.; Liu, R. S. Broadening Phosphor-Converted Light-Emitting Diode Emission: Controlling Disorder. Chem. Mater. 2022, 34 (22), 10190– 10199, DOI: 10.1021/acs.chemmater.2c03045Google ScholarThere is no corresponding record for this reference.
- 58Zhang, A.; Liu, Y.; Liu, G.; Xia, Z. Dopant and Compositional Modulation Triggered Broadband and Tunable Near-Infrared Emission in Cs2Ag1-xNaxInCl6:Cr3+ Nanocrystals. Chem. Mater. 2022, 34 (7), 3006– 3012, DOI: 10.1021/acs.chemmater.1c03878Google ScholarThere is no corresponding record for this reference.
- 59Snoeren, T. J.; Pressler, K.; Kluherz, K. T.; Walsh, K. M.; De Yoreo, J. J.; Gamelin, D. R. Luminescence and Covalency in Ytterbium-Doped CrX3 (X = Cl, Br, I) van der Waals Compounds. J. Am. Chem. Soc. 2023, 145 (31), 17427– 17434, DOI: 10.1021/jacs.3c05989Google ScholarThere is no corresponding record for this reference.
- 60Lyu, K.; Liu, G.; Molokeev, M. S.; Xia, Z. Double-Site Occupation Triggered Broadband and Tunable NIR-I and NIR-II Luminescence in AlNbO4:Cr3+ Phosphors. Adv. Phys. Res. 2023, 2 (4), 2200056, DOI: 10.1002/apxr.202200056Google ScholarThere is no corresponding record for this reference.
- 61Dou, C.; Liang, T.; Zhao, M.; Song, Z.; Ning, L.; Peng, D.; Liu, Q. Self-Recoverable and NIR-I to NIR-II Tunable Broadband Mechanoluminescence of Cr3+-Doped Antimonate Double Perovskites. Adv. Funct. Mater. 2025, 35 (14), 2419716, DOI: 10.1002/adfm.202419716Google ScholarThere is no corresponding record for this reference.
- 62Gamelin, D. R.; Güdel, H. U. Design of Luminescent Inorganic Materials: New Photophysical Processes Studied by Optical Spectroscopy. Acc. Chem. Res. 2000, 33 (4), 235– 242, DOI: 10.1021/ar990102yGoogle ScholarThere is no corresponding record for this reference.
- 63Aebischer, A.; Wenger, O. S.; Güdel, H. U. Near-infrared to visible photon up-conversion in V3+, Re4+ co-doped Cs2NaYCl6. J. Lumin. 2003, 102–103, 48– 53, DOI: 10.1016/S0022-2313(02)00528-8Google ScholarThere is no corresponding record for this reference.
- 64Ravi, V. K.; Scheidt, R. A.; Nag, A.; Kuno, M.; Kamat, P. V. To Exchange or Not to Exchange. Suppressing Anion Exchange in Cesium Lead Halide Perovskites with PbSO4-Oleate Capping. ACS Energy Lett. 2018, 3 (4), 1049– 1055, DOI: 10.1021/acsenergylett.8b00380Google ScholarThere is no corresponding record for this reference.
- 65Zhu, D.; Zito, J.; Pinchetti, V.; Dang, Z.; Olivati, A.; Pasquale, L.; Tang, A.; Zaffalon, M. L.; Meinardi, F.; Infante, I.; De Trizio, L.; Manna, L.; Brovelli, S. Compositional Tuning of Carrier Dynamics in Cs2Na1-xAgxBiCl6 Double-Perovskite Nanocrystals. ACS Energy Lett. 2020, 5 (6), 1840– 1847, DOI: 10.1021/acsenergylett.0c00914Google ScholarThere is no corresponding record for this reference.
- 66Liang, W.; Li, Y.; Xiang, D.; Han, Y.; Jiang, Q.; Zhang, W.; Wu, K. Efficient Optical Orientation and Slow Spin Relaxation in Lead-Free CsSnBr3 Perovskite Nanocrystals. ACS Energy Lett. 2021, 6 (5), 1670– 1676, DOI: 10.1021/acsenergylett.1c00413Google ScholarThere is no corresponding record for this reference.
- 67Dey, A.; Ye, J.; De, A.; Debroye, E.; Ha, S. K.; Bladt, E.; Kshirsagar, A. S.; Wang, Z.; Yin, J.; Wang, Y.; Quan, L. N.; Yan, F.; Gao, M.; Li, X.; Shamsi, J.; Debnath, T.; Cao, M.; Scheel, M. A.; Kumar, S.; Steele, J. A.; Gerhard, M.; Chouhan, L.; Xu, K.; Wu, X. g.; Li, Y.; Zhang, Y.; Dutta, A.; Han, C.; Vincon, I.; Rogach, A. L.; Nag, A.; Samanta, A.; Korgel, B. A.; Shih, C. J.; Gamelin, D. R.; Son, D. H.; Zeng, H.; Zhong, H.; Sun, H.; Demir, H. V.; Scheblykin, I. G.; Mora-Seró, I.; Stolarczyk, J. K.; Zhang, J. Z.; Feldmann, J.; Hofkens, J.; Luther, J. M.; Pérez-Prieto, J.; Li, L.; Manna, L.; Bodnarchuk, M. I.; Kovalenko, M. V.; Roeffaers, M. B. J.; Pradhan, N.; Mohammed, O. F.; Bakr, O. M.; Yang, P.; Müller-Buschbaum, P.; Kamat, P. V.; Bao, Q.; Zhang, Q.; Krahne, R.; Galian, R. E.; Stranks, S. D.; Bals, S.; Biju, V.; Tisdale, W. A.; Yan, Y.; Hoye, R. L. Z.; Polavarapu, L. State of the Art and Prospects for Halide Perovskite Nanocrystals. ACS Nano 2021, 15 (7), 10775– 10981, DOI: 10.1021/acsnano.0c08903Google ScholarThere is no corresponding record for this reference.
- 68Chen, Z.; Hoye, R. L. Z.; Yip, H. L.; Fiuza-Maneiro, N.; López-Fernández, I.; Otero-Martínez, C.; Polavarapu, L.; Mondal, N.; Mirabelli, A.; Anaya, M.; Stranks, S. D.; Liu, H.; Shi, G.; Xiao, Z.; Kim, N.; Kim, Y.; Shin, B.; Shi, J.; Liu, M.; Zhang, Q.; Fan, Z.; Loy, J. C.; Zhao, L.; Rand, B. P.; Arfin, H.; Saikia, S.; Nag, A.; Zou, C.; Lin, L. Y.; Xiang, H.; Zeng, H.; Liu, D.; Su, S. J.; Wang, C.; Zhong, H.; Xuan, T. T.; Xie, R. J.; Bao, C.; Gao, F.; Gao, X.; Qin, C.; Kim, Y. H.; Beard, M. C. Roadmap on perovskite light-emitting diodes. J. Phys. Photonics 2024, 6 (3), 032501, DOI: 10.1088/2515-7647/ad46a6Google ScholarThere is no corresponding record for this reference.
- 69Mukherjee, P.; Das, R.; Pariari, D.; Das, K.; Mahadevan, P.; Sarma, D. D. Energy-Transfer Pathways That Sensitize Mn2+ Emissions Using Mn@CsPbCl3 Nanocrystals as a Generic Platform. ACS Energy Lett. 2025, 10 (12), 6381– 6389, DOI: 10.1021/acsenergylett.5c02937Google ScholarThere is no corresponding record for this reference.
- 70Shukla, A.; Kaur, G.; Justice Babu, K.; Bhatt, H.; Kumar, V.; Ghosh, H. N. The Retarded Hot Carrier Relaxation and Augmented Photoconductivity in Eu3+-Doped CsPbBr3 Nanocrystals. ACS Photonics 2025, 12 (8), 4224– 4233, DOI: 10.1021/acsphotonics.5c00396Google ScholarThere is no corresponding record for this reference.
- 71Ji, F.; Klarbring, J.; Zhang, B.; Wang, F.; Wang, L.; Miao, X.; Ning, W.; Zhang, M.; Cai, X.; Bakhit, B.; Magnuson, M.; Ren, X.; Sun, L.; Fahlman, M.; Buyanova, I. A.; Chen, W. M.; Simak, S. I.; Abrikosov, I. A.; Gao, F. Remarkable Thermochromism in the Double Perovskite Cs2NaFeCl6. Adv. Opt. Mater. 2024, 12 (8), 2301102, DOI: 10.1002/adom.202301102Google ScholarThere is no corresponding record for this reference.
- 72Boyer, J. C.; Cuccia, L. A.; Capobianco, J. A. Synthesis of Colloidal Upconverting NaYF4: Er3+/Yb3+ and Tm3+/Yb3+ Monodisperse Nanocrystals. Nano Lett. 2007, 7 (3), 847– 852, DOI: 10.1021/nl070235+Google ScholarThere is no corresponding record for this reference.
- 73Ghosh, P.; Kar, A.; Patra, A. Structural and photoluminescence properties of doped and core-shell LaPO4:Eu3+ nanocrystals. J. Appl. Phys. 2010, 108 (11), na, DOI: 10.1063/1.3514137Google ScholarThere is no corresponding record for this reference.
- 74Tzanetopoulos, E.; Schwartz, J.; Gamelin, D. R. HF-Free synthesis of colloidal Cs2ZrF6 and (NH4)2ZrF6 nanocrystals. Chem. Commun. 2023, 59 (36), 5451– 5454, DOI: 10.1039/D3CC00374DGoogle ScholarThere is no corresponding record for this reference.
- 75Imran, M.; Caligiuri, V.; Wang, M.; Goldoni, L.; Prato, M.; Krahne, R.; De Trizio, L.; Manna, L. Benzoyl Halides as Alternative Precursors for the Colloidal Synthesis of Lead-Based Halide Perovskite Nanocrystals. J. Am. Chem. Soc. 2018, 140 (7), 2656– 2664, DOI: 10.1021/jacs.7b13477Google ScholarThere is no corresponding record for this reference.
- 76Ming, J.; Chen, Y.; Miao, H.; Fan, Y.; Wang, S.; Chen, Z.; Guo, Z.; Guo, Z.; Qi, L.; Wang, X.; Yun, B.; Pei, P.; He, H.; Zhang, H.; Tang, Y.; Zhao, D.; Wong, G. K.-L.; Bünzli, J.-C. G.; Zhang, F. High-brightness transition metal-sensitized lanthanide near-infrared luminescent nanoparticles. Nat. Photonics 2024, 18 (12), 1254– 1262, DOI: 10.1038/s41566-024-01517-9Google ScholarThere is no corresponding record for this reference.
- 77Kong, Q.; Jiang, S.; Wang, Z.; Xu, X.; Zhang, R.; Zhu, G.; Yang, J.; Han, P.; Liu, R.; Hong, F.; Luo, N.; Chen, J.; Yang, B. Highly Stable Cesium Molybdenum Chloride Perovskite Nanocrystals for Photothermal Semihydrogenation Applications. ACS Appl. Mater. Interfaces 2024, 16 (27), 35752– 35760, DOI: 10.1021/acsami.4c05157Google ScholarThere is no corresponding record for this reference.
- 78Mondal, B.; Mehta, S.; Mondal, A.; Nag, A. W-Doped Cs2SnCl6 for Near-Infrared Emission. J. Phys. Chem. Lett. 2025, 16 (20), 4937– 4943, DOI: 10.1021/acs.jpclett.5c00822Google ScholarThere is no corresponding record for this reference.
- 79Saikia, S.; Ghosh, A.; Nag, A. Broad Dual Emission by Codoping Cr3+ (d→d) and Bi3+ (s→p) in Cs2Ag0.6Na0.4InCl6 Double Perovskite. Angew. Chem. Inter. Ed. 2023, 62 (33), e202307689 DOI: 10.1002/anie.202307689Google ScholarThere is no corresponding record for this reference.
- 80Tripathi, V. M.; Saikia, S.; Nag, A. Li(Sc0.70Ga0.24Cr0.06)Ge2O6 in Polylactic Acid for Robust Near-Infrared Phosphor-Converted Light-Emitting Diodes. Chem. Mater. 2025, 37 (18), 7462– 7470, DOI: 10.1021/acs.chemmater.5c01897Google ScholarThere is no corresponding record for this reference.
- 81Li, X.; Li, J.; Wang, B.; Pan, D.; Gu, L.; Zhang, J.; Zhang, L.; Kong, Y.; Huang, J. Near-Infrared Luminescence in Mo4+-Activated Metal Halide for Advanced Optoelectronics. Inorg. Chem. 2025, 64 (45), 22384– 22392, DOI: 10.1021/acs.inorgchem.5c03606Google ScholarThere is no corresponding record for this reference.
- 82Li, J.; Du, Z.; Li, X.; Zhang, L.; Gu, L.; Pan, D.; Zhang, J.; Wang, B. Mo4+-Doped Cs4ZnBi2Cl12 Lead-Free Double Perovskite for Near-Infrared Luminescence. Luminescence 2025, 40 (11), e70352 DOI: 10.1002/bio.70352Google ScholarThere is no corresponding record for this reference.
- 83Pan, J. L.; Yu, Y. J.; Wang, Y. K.; Liao, L. S. Lanthanide Ion-Doped Perovskite Nanocrystals in Electroluminescent Device. Adv. Funct. Mater. 2024, 34 (36), 2401327, DOI: 10.1002/adfm.202401327Google ScholarThere is no corresponding record for this reference.
- 84Wang, H.; Treglia, A.; Albaqami, M. D.; Gao, F.; Petrozza, A. Tin-Halide Perovskites for Near-Infrared Light-Emitting Diodes. ACS Energy Lett. 2024, 9 (6), 2500– 2507, DOI: 10.1021/acsenergylett.4c00470Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 2 publications.
- Sajid Saikia, Animesh Ghosh, Diogo Alves Gálico, Claudia Manuela Santos Calado, Muralee Murugesu, Angshuman Nag. Spin‐Flip Upconversion Luminescence and Tunable Downshifting Near‐Infrared Emissions via 4
d
‐4
f
Interaction in Doped Halide Perovskite. Angewandte Chemie 2026, 11 https://doi.org/10.1002/ange.7420770
- Sajid Saikia, Animesh Ghosh, Diogo Alves Gálico, Claudia Manuela Santos Calado, Muralee Murugesu, Angshuman Nag. Spin‐Flip Upconversion Luminescence and Tunable Downshifting Near‐Infrared Emissions via 4
d
‐4
f
Interaction in Doped Halide Perovskite. Angewandte Chemie International Edition 2026, 11 https://doi.org/10.1002/anie.7420770
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
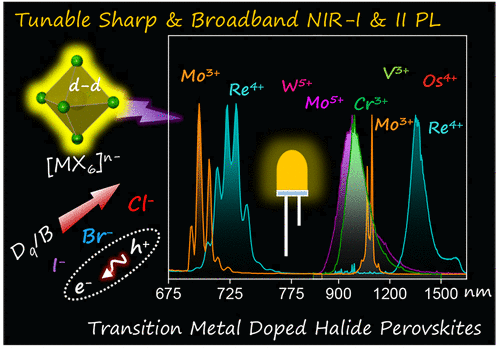
Figure 1

Figure 1. Schematic highlights the scope of transition metal doping in halide perovskites (and related structures) for NIR emission, which is typically beyond the scope of oxide-based NIR phosphors.
Figure 2
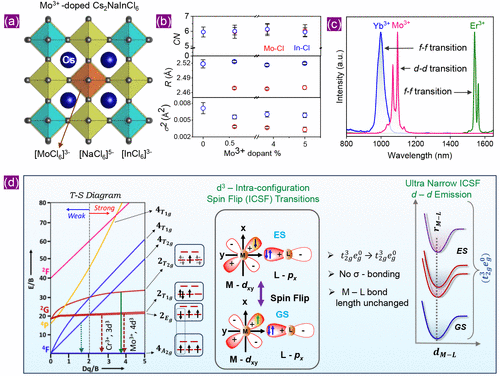
Figure 2. (a) Schematic representation of Mo3+-doped Cs2NaInCl6 double perovskite cubic lattice with the space group Fm3m. (b) Description of the Cl environment around Mo (red open symbols) and In (blue open symbols) for Mo3+-doped Cs2NaInCl6 with different dopant concentrations: coordination number (CN, top panel), bond length (R, middle panel), and local order (σ2, bottom panel). (c) d–d vs f–f transition-based NIR-II PL spectra from Ln3+- (Ln = Yb, Er) and Mo3+-doped Cs2NaInCl6. (d) Left panel: Tanabe-Sugano diagram for a d3 ion in an octahedral coordination; middle panel: schematic diagram comparing the orbital interaction between the metal (M) dxy ligand (L) px when the metal ion (d3: Cr3+, Mo3+, Re4+) is in the ground state (GS: 4A2g) and in an excited state (ES: 2Eg); right panel: schematic potential energy surface diagram, indicating that the ground state 4A2g, and excited states 2Eg/2T1g and 2T2g have the same t2g3eg0 electronic configuration and, therefore, have very similar M–L bond lengths. Reproduced with permission from ref (20). Copyright 2025 Wiley.
Figure 3
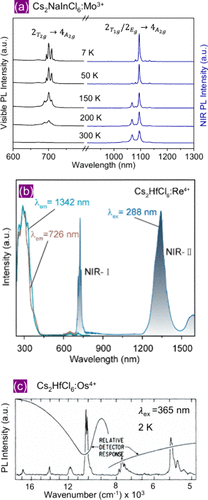
Figure 3. (a) Temperature-dependent (7–300 K) PL spectra of 3.8% Mo3+-doped Cs2NaInCl6 in the visible to NIR region under 438 nm excitation. Reproduced with permission from ref (20). Copyright 2025 Wiley. (b) PL and PL excitation spectra of Cs2HfCl6:Re4+ at room temperature. Reproduced with permission from ref (26). Copyright 2025 Elsevier. (c) PL spectrum of Cs2HfCl6:Os4+ crystals at 2 K. Since the spectrum was collected in a broad range from visible to NIR-II (∼580–2100 nm), the relative detector response is also shown. Reproduced with permission from ref (14). Copyright 1971 American Physical Society.
Figure 4

Figure 4. (a) Crystal structure of Cs2MoCl6 containing a [MoCl6]2– unit with an octahedral symmetry (top) followed by the Tanabe-Sugano diagram as reference for showing possible electronic transitions of Mo4+ (4d2) in an octahedral local symmetry (bottom). The crystal structure was obtained using a CIF file reported in ref (42). (b) Crystal structure of Cs2MoOxCl6–x containing the [MoOCl6]2– unit with reduced C4v symmetry (top) followed by possible electronic transitions of Mo5+ (4d1) (bottom). The crystal structure was obtained using the CIF file reported in ref (35).
Figure 5
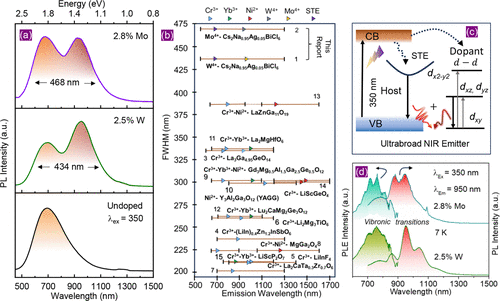
Figure 5. (a) PL spectra of undoped, 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6 under the excitation of 350 nm, showing unprecedented spectral widths of 434 and 468 nm, respectively. The spectra were collected using two detectors and then combined at 720 nm. (b) fwhm of various reported broadband NIR emitters is compared with that of 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6. The composition of various NIR emitters is mentioned in the figure with serial numbers that correspond to a table reported in the Supporting Information of ref (46). The position of the tringles indicates the PL peak position(s) of the broadband NIR PL, while the solid and dashed lines show the emission wavelength range coverage with continuity and discontinuity, respectively. The colors of the triangle indicate various dopants, as stated at the top of the figure. Different PL peak for a single dopant due to occupation of two different lattice sites is indicated by tringle of same color with dashed and solid edges. (c) Schematics summarizing the excitation and emission pathways for ultrabroadband NIR emissions from W-doped and Mo-doped Cs2Na0.95Ag0.05BiCl6. (d) PL and PL excitation of 2.5% W-doped and 2.8% Mo-doped Cs2Na0.95Ag0.05BiCl6 at 7 K. Reproduced with permission from ref (49). Copyright 2025 Wiley.
Figure 6
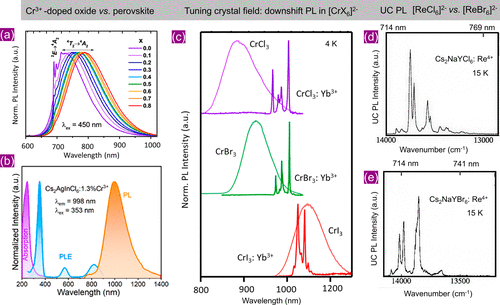
Figure 6. (a) NIR PL of Ga1.98–x(Al0.68In0.32)xO3:2% Cr3+ (x = 0–0.8) at room temperature. Reproduced with permission from ref (57). Copyright 2022 American Chemical Society. (b) Normalized absorption (violet), PL emission (orange), and PL excitation (blue) spectra of Cs2AgInCl6:1.3% Cr3+ nanocrystals. Reproduced with permission from ref (58). Copyright 2022 American Chemical Society. (c) Low-temperature (4 K) PL spectra of CrX3 (X = Cl, Br, I) showing broad PL due to 4T2g → 4A2g, and corresponding Yb3+-doped CrX3 showing sharp PL due to 2F5/2 → 2F7/2 transition of Yb3+. The absence of lattice PL in the latter indicates efficient ET from Cr3+ to Yb3+. Reproduced with permission from ref (59). Copyright 2023 American Chemical Society. (d, e) NIR to visible upconversion (UC) photoluminescence of Re4+-doped Cs2NaYCl6 and Cs2NaYBr6 at 15 K respectively. Reproduced with permission from ref (17). Copyright 1998 American Chemical Society.
Figure 7
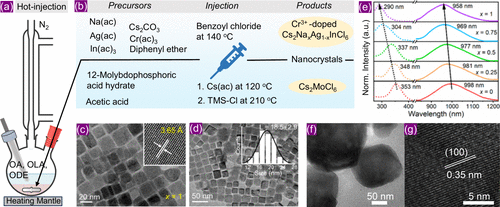
Figure 7. (a) Hot-injection synthesis setup for colloidal nanocrystals. (b) Precursors and reaction conditions used to prepare (top panel) Cr3+-doped Cs2NaxAg1–xInCl6 (x = 0–1) and (bottom panel) Cs2MoCl6 nanocrystals. (c, d) TEM images of Cr3+-doped Cs2NaInCl6 nanocrystals. Inset images of (c) and (d) show high resolution TEM and size distribution of nanocrystals. (e) Room temperature PL of Cr3+-doped Cs2NaxAg1–xInCl6 (x = 0 −1) colloidal nanocrystals under different excitations. (c)–(e) were reproduced with permission from ref (58). Copyright 2022 American Chemical Society. (f–g) TEM and high-resolution TEM images of Cs2MoCl6 nanocrystals. Reproduced with permission from ref (77). Copyright 2024 American Chemical Society.
Figure 8
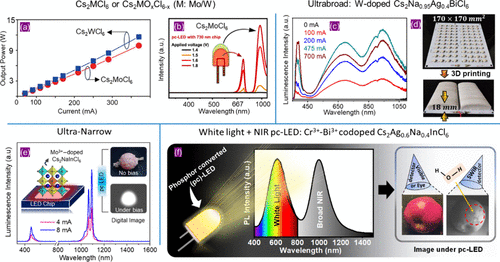
Figure 8. (a) Output power for NIR emission with a peak at ∼980 nm as a function of applied current for a Cs2MCl6 (M = Mo/W) based pc-LED panel. The phosphor was coated with UV chips. Reproduced with permission from ref (34). Copyright 2022 Wiley. (b) Luminescence spectra as a function of biased voltages for a pc-LED fabricated by coating Cs2MoCl6 phosphor composite on a far-red (730 nm) LED chip. Reproduced with permission from ref (42). Copyright 2024 American Chemical Society. (c) Luminescence spectra of 2.5% W-doped Cs2Na0.95Ag0.4BiCl6 NIR pc-LEDs at different driving currents. (d) The composite 2.5% W-doped Cs2Na0.95Ag0.4BiCl6 and poly(lactic acid) forming a 3D printed structure over the UV-LED panel. The 3D printed phosphor composites yield better thermal stability for the prolonged operation of the NIR-pc-LED. (c, d) Reproduced with permission from ref (49). Copyright 2025 Wiley. (e) Luminescence spectra of the pc-LED fabricated by coating a polymer composite of 3.8% Mo3+-doped Cs2NaInCl6 on a blue LED chip. The schematics of fabrication and digital photographs of the devices are shown in the insets. Reproduced with permission from ref (20). Copyright 2025 Wiley. (f) pc-LED panel and NIR imaging for food freshness detection using 2% Cr3+-0.5%Bi3+-codoped Cs2Ag0.6Na0.4InCl6. Images of a pair of apples were collected by a visible camera and NIR camera under illumination from pc-LED. The NIR image could detect the early rotting stage of an apple, as shown with a red circle. Reproduced with permission from ref (79). Copyright 2023 Wiley.
References
This article references 84 other publications.
- 1Shionoya, S.; Yen, W. M.; Yamamoto, H. Phosphor Handbook; CRC Press, 2018.There is no corresponding record for this reference.
- 2Geusic, J. E.; Marcos, H. M.; Van Uitert, L. G. Laser Oscillations in Nd-doped Yttrium Aluminium, Yttrium Gallium and Gadolanium Garnets. Appl. Phys. Lett. 1964, 4 (10), 182– 184, DOI: 10.1063/1.1753928There is no corresponding record for this reference.
- 3Maiman, T. H. Stimulated Optical Radiation in Ruby. Nature 1960, 187 (4736), 493– 494, DOI: 10.1038/187493a0There is no corresponding record for this reference.
- 4Mir, W. J.; Sheikh, T.; Arfin, H.; Xia, Z.; Nag, A. Lanthanide doping in metal halide perovskite nanocrystals: spectral shifting, quantum cutting and optoelectronic applications. NPG Asia Mater. 2020, 12 (1), 9, DOI: 10.1038/s41427-019-0192-0There is no corresponding record for this reference.
- 5Wang, N.; Zhuang, B.; Pang, T.; Zeng, L.; Chen, D. Recent Advances in Lanthanide-Doped Perovskite Nanocrystals. Laser Photonics Rev. 2025, 19 (18), e00905 DOI: 10.1002/lpor.202500905There is no corresponding record for this reference.
- 6Nag, A.; Sapra, S.; Gupta, S. S.; Prakash, A.; Ghangrekar, A.; Periasamy, N.; Sarma, D. D. Luminescence in Mn-doped CdS nanocrystals. Bull. Mater. Sci. 2008, 31 (3), 561– 568, DOI: 10.1007/s12034-008-0087-0There is no corresponding record for this reference.
- 7Santra, P. K.; Kamat, P. V. Mn-Doped Quantum Dot Sensitized Solar Cells: A Strategy to Boost Efficiency over 5%. J. Am. Chem. Soc. 2012, 134 (5), 2508– 2511, DOI: 10.1021/ja211224sThere is no corresponding record for this reference.
- 8Wei, Y.; Dang, P.; Dai, Z.; Li, G.; Lin, J. Advances in Near-Infrared Luminescent Materials without Cr3+: Crystal Structure Design, Luminescence Properties, and Applications. Chem. Mater. 2021, 33 (14), 5496– 5526, DOI: 10.1021/acs.chemmater.1c01325There is no corresponding record for this reference.
- 9Dang, P.; Wei, Y.; Liu, D.; Li, G.; Lin, J. Recent Advances in Chromium-Doped Near-Infrared Luminescent Materials: Fundamentals, Optimization Strategies, and Applications. Adv. Opt. Mater. 2023, 11 (3), 2201739, DOI: 10.1002/adom.202201739There is no corresponding record for this reference.
- 10Chen, C.; Chang, J.; Chen, R.; Gao, R.; Wang, Y.; Zhu, K.; Xiang, J.; Guo, C. Strategies for broadening the emission spectra of Cr3+-doped near-infrared emitting phosphors. Mater. Chem. Front. 2025, 9 (12), 1821– 1838, DOI: 10.1039/D4QM01109KThere is no corresponding record for this reference.
- 11Guria, A. K.; Dutta, S. K.; Adhikari, S. D.; Pradhan, N. Doping Mn2+ in Lead Halide Perovskite Nanocrystals: Successes and Challenges. ACS Energy Lett. 2017, 2 (5), 1014– 1021, DOI: 10.1021/acsenergylett.7b00177There is no corresponding record for this reference.
- 12Senden, T.; van Dijk-Moes, R. J. A.; Meijerink, A. Quenching of the red Mn4+ luminescence in Mn4+-doped fluoride LED phosphors. Light Sci. Appl. 2018, 7 (1), 8, DOI: 10.1038/s41377-018-0013-1There is no corresponding record for this reference.
- 13Huang, W.; Li, C.; Zeng, L.; Zhang, J.; Kurochkin, M. A.; Kolesnikov, I. E.; Umar, Z.; Zhang, J.; Liu, W.; Kukovecz, Á.; Kurboniyon, M. S.; Zhang, X. One Shoot Two Birds: Stabilization of Mn5+ in Ba2(Si,Ge)O4 as an Efficient NIR Thermometer and Intense Turquoise Pigment. Inorg. Chem. 2025, 64 (24), 12100– 12111, DOI: 10.1021/acs.inorgchem.5c01428There is no corresponding record for this reference.
- 14Reinberg, A. R. Sharp-Line Luminescence from Os4+ and Mo3+ in Cs2HfCl6. Phys. Rev. B 1971, 3 (1), 41– 46, DOI: 10.1103/PhysRevB.3.41There is no corresponding record for this reference.
- 15Flint, C. D.; Paulusz, A. G. Infrared and visible luminescence spectra of MoCl63- and MoBr63- in cubic elpasolite crystals. Mol. Phys. 1981, 44 (4), 925– 938, DOI: 10.1080/00268978100102891There is no corresponding record for this reference.
- 16Stranger, R.; Moran, G. M.; Krausz, E.; Güdel, H. U.; Furer, N. Octahedral monomeric molybdenum(III). A magneto-optical study of Mo3+ doped in Cs2NaYCl6. Mol. Phys. 1990, 69, 11– 31, DOI: 10.1080/00268979000100021There is no corresponding record for this reference.
- 17Gamelin, D. R.; Güdel, H. U. Two-Photon Spectroscopy of d3 Transition Metals: Near-IR-to-Visible Upconversion Luminescence by Re4+ and Mo3+. J. Am. Chem. Soc. 1998, 120 (46), 12143– 12144, DOI: 10.1021/ja982742mThere is no corresponding record for this reference.
- 18Gamelin, D. R.; Güdel, H. U. Excited-State Dynamics and Sequential Two-Photon Upconversion Excitation of Mo3+-Doped Chloro- and Bromo-elpasolites. J. Phys. Chem. B 2000, 104 (44), 10222– 10234, DOI: 10.1021/jp002132kThere is no corresponding record for this reference.
- 19Wenger, O. S.; Güdel, H. U. Broadband near-Infrared Sensitization of Visible Upconversion Luminescence in V3+ and Mo3+ Co-Doped Cs2NaYCl6. J. Phys. Chem. B 2002, 106 (39), 10011– 10019, DOI: 10.1021/jp021076fThere is no corresponding record for this reference.
- 20Ghosh, A.; Saikia, S.; Mukherjee, S.; Johannesson, E.; Rensmo, H.; Nag, A. Intra-Configurational Spin-Flip d→d Transition of Mo(III) Doped Perovskite for Ultra-Narrow Near Infrared-II Emission in Ambient Conditions. Angew. Chem., Int. Ed. 2025, 64 (52), e19144 DOI: 10.1002/anie.202519144There is no corresponding record for this reference.
- 21Binwal, D. C.; Mudoi, P. P.; Panda, D. P.; Vishnoi, P. Molybdenum chloride double perovskites: dimensionality control of optical and magnetic properties. Chem. Sci. 2023, 14 (15), 3982– 3989, DOI: 10.1039/D3SC00132FThere is no corresponding record for this reference.
- 22Binwal, D. C.; Anand, K.; Sharma, M.; Saurav, K. V.; Pati, S. K.; Vishnoi, P. Tailorable Magnetic Exchange and Optical Absorption in 1-D Double Perovskite-like Halides (CH3NH3)2NaMoCl(6–x)Brx. Chem. Mater. 2024, 36 (16), 7719– 7730, DOI: 10.1021/acs.chemmater.4c00735There is no corresponding record for this reference.
- 23Kitzmann, W. R.; Moll, J.; Heinze, K. Spin-flip luminescence. Photochem. Photobiol. Sci. 2022, 21 (7), 1309– 1331, DOI: 10.1007/s43630-022-00186-3There is no corresponding record for this reference.
- 24Kitzmann, W. R.; Heinze, K. Charge-Transfer and Spin-Flip States: Thriving as Complements. Angew. Chem., Int. Ed. 2023, 62 (15), e202213207 DOI: 10.1002/anie.202213207There is no corresponding record for this reference.
- 25Gamelin, D. R.; Güdel, H. U. Spectroscopy and Dynamics of Re4+ Near-IR-to-Visible Luminescence Upconversion. Inorg. Chem. 1999, 38 (22), 5154– 5164, DOI: 10.1021/ic990556rThere is no corresponding record for this reference.
- 26Fan, Z.; Liu, Y.; Gao, Y.; Xie, J.; Zhao, I. S.; Wang, Y. A novel efficient lead-free near-infrared emission Re4+-doped double perovskite for dental caries medical imaging. Chem. Eng. J. 2025, 524, 169762, DOI: 10.1016/j.cej.2025.169762There is no corresponding record for this reference.
- 27Cai, P.; Li, B.; Zhan, Y.; Feng, X.; Pu, X.; Wang, L.; Chen, Z.; Liu, Z.; Bi, S. Re4+/Te4+ Co-doped Cs2ZrCl6 double perovskite microcrystals: Broadening excitation range and boosting luminescent performance for near-infrared lighting and non-destructive quality inspection. Mater. Today Chem. 2025, 48, 102954, DOI: 10.1016/j.mtchem.2025.102954There is no corresponding record for this reference.
- 28Reber, C.; Güdel, H. U. Near-infraded luminescence spectroscopy and relaxation behaviour of V3+ doped in Cs2NaYCl6-mBrm (m = 0, 0.3, 3, 6). J. Lumin. 1988, 42 (1), 1– 13, DOI: 10.1016/0022-2313(88)90059-2There is no corresponding record for this reference.
- 29Gray, H. B.; Hare, C. R. The electronic structures and spectra of chromyl and molybdenyl ions. Inorg. Chem. 1962, 1 (2), 363– 368, DOI: 10.1021/ic50002a034There is no corresponding record for this reference.
- 30Winkler, J. R.; Gray, H. B. Emission spectroscopic properties of dioxorhenium(V) complexes in crystals and solutions. J. Am. Chem. Soc. 1983, 105 (5), 1373– 1374, DOI: 10.1021/ja00343a052There is no corresponding record for this reference.
- 31Isovitsch, R. A.; Beadle, A. S.; Fronczek, F. R.; Maverick, A. W. Electronic Absorption Spectra and Phosphorescence of Oxygen-Containing Molybdenum(IV) Complexes. Inorg. Chem. 1998, 37 (17), 4258– 4264, DOI: 10.1021/ic971186eThere is no corresponding record for this reference.
- 32Wermuth, M.; Güdel, H. U. Photon Avalanche in Cs2ZrBr6:Os4+. J. Am. Chem. Soc. 1999, 121 (43), 10102– 10111, DOI: 10.1021/ja990847zThere is no corresponding record for this reference.
- 33Ghosh, A.; Saikia, S.; Molokeev, M. S.; Nag, A. Synthesis and Optical Properties of Organic-Inorganic Hybrid [(18-Crown-6)K][MoOCl4(H2O)]. Chem.–Asian J. 2025, 20 (1), e202401052 DOI: 10.1002/asia.202401052There is no corresponding record for this reference.
- 34Liu, Z.; Qin, X.; Chen, Q.; Chen, Q.; Jing, Y.; Zhou, Z.; Zhao, Y. S.; Chen, J.; Liu, X. Highly Stable Lead-Free Perovskite Single Crystals with NIR Emission Beyond 1100 nm. Adv. Opt. Mater. 2022, 10 (21), 2201254, DOI: 10.1002/adom.202201254There is no corresponding record for this reference.
- 35Morgan, E. E.; Brumberg, A.; Panuganti, S.; Kent, G. T.; Zohar, A.; Mikhailovsky, A. A.; Kanatzidis, M. G.; Schaller, R. D.; Chabinyc, M. L.; Cheetham, A. K.; Seshadri, R. Molecular Origins of Near-Infrared Luminescence in Molybdenum and Tungsten Oxyhalide Perovskites. Chem. Mater. 2024, 36 (16), 7754– 7763, DOI: 10.1021/acs.chemmater.4c00856There is no corresponding record for this reference.
- 36Siddique, H.; Xu, Z.; Li, X.; Saeed, S.; Liang, W.; Wang, X.; Gao, C.; Dai, R.; Wang, Z.; Zhang, Z. Anomalous Octahedron Distortion of Bi-Alloyed Cs2AgInCl6 Crystal via XRD, Raman, Huang-Rhys Factor, and Photoluminescence. J. Phys. Chem. Lett. 2020, 11 (22), 9572– 9578, DOI: 10.1021/acs.jpclett.0c02852There is no corresponding record for this reference.
- 37Wang, C.; Wang, J.; Wang, D.; He, L. Efficient near-infrared emission and imaging of lead-free double perovskite Cs2TeCl6 by Mo4+ doping. J. Lumin. 2025, 286, 121381, DOI: 10.1016/j.jlumin.2025.121381There is no corresponding record for this reference.
- 38Zhang, G.; Li, L.; Mao, Y.; Dang, P.; Zhao, Y.; Suo, H.; Wang, Z.; Li, P.; Cheng, Z.; Lian, H.; Lin, J. Broad-Band Near-Infrared Emission with High External Quantum Efficiency from Molybdenum-Doped Vacancy-Ordered Double Perovskites for Spectroscopic Analysis. ACS Mater. Lett. 2025, 7 (6), 2190– 2198, DOI: 10.1021/acsmaterialslett.5c00334There is no corresponding record for this reference.
- 39Kumar, S.; Lamba, R. S.; Monga, S.; Jha, V.; Singh, R.; Saha, S.; Bhattacharya, S.; Sapra, S. Broad Dual Emission from Cs2Zr1-xMoxCl6: Enhancing the NIR Emission in Lead-free Vacancy Ordered Double Perovskites. Chem. Mater. 2024, 36 (9), 4561– 4570, DOI: 10.1021/acs.chemmater.4c00231There is no corresponding record for this reference.
- 40Li, Z.; Xu, M.; Liu, W.; Wang, X.; Li, Y.; Zhou, X.; Fang, Z.; Ning, L. Broadband Near-Infrared Luminescence from Mo4+ in Zero-Dimensional Perovskite Cs2Zr(Cl,Br)6 with an Exceptionally High Quantum Efficiency and Thermal Stability. Chem. Mater. 2024, 36 (2), 901– 910, DOI: 10.1021/acs.chemmater.3c02805There is no corresponding record for this reference.
- 41Kumar, S.; Lamba, R. S.; Singh, R.; Jha, V.; Yadav, J.; Sapra, S. Efficient Near-Infrared Luminescence with Broad-Band Sensitization in Mo4+-Er3+ Co-Doped Cs2ZrCl6 Vacancy-Ordered Double Perovskites. Small 2025, 21 (46), e07964 DOI: 10.1002/smll.202507964There is no corresponding record for this reference.
- 42Mondal, B.; Shinde, A.; Rajput, P. K.; Arfin, H.; Tanwar, R.; Ghosh, P.; Nag, A. Vibronically Coupled Near-Infrared Emission and Excitation from d–d Transitions of Cs2MX6 (M = Mo/W, X = Cl/Br). ACS Energy Lett. 2024, 9 (3), 819– 828, DOI: 10.1021/acsenergylett.3c02613There is no corresponding record for this reference.
- 43Han, Y.; Cheng, X.; Cui, B. B. Factors influencing self-trapped exciton emission of low-dimensional metal halides. Mater. Adv. 2023, 4 (2), 355– 373, DOI: 10.1039/D2MA00676FThere is no corresponding record for this reference.
- 44Zhang, G.; Wang, D.; Lou, B.; Ma, C. G.; Meijerink, A.; Wang, Y. Efficient Broadband Near-Infrared Emission from Lead-Free Halide Double Perovskite Single Crystal. Angew. Chem., Int. Ed. 2022, 61 (33), e202207454 DOI: 10.1002/anie.202207454There is no corresponding record for this reference.
- 45Cheng, X.; Xie, Z.; Zheng, W.; Li, R.; Deng, Z.; Tu, D.; Shang, X.; Xu, J.; Gong, Z.; Li, X.; Chen, X. Boosting the Self-Trapped Exciton Emission in Alloyed Cs2(Ag/Na)InCl6 Double Perovskite via Cu+ Doping. Adv. Sci. 2022, 9 (7), 2103724, DOI: 10.1002/advs.202103724There is no corresponding record for this reference.
- 46Liu, Y.; Tu, D.; Yang, M.; Li, H.; Li, C.; Xie, Z.; Zhou, S.; Yu, S.; Xu, J.; Chen, X. Near-Infrared and Visible Dual-Band Self-Trapped Exciton Emissions from Li+-Doped Cs2NaScCl6 Double Perovskites. ACS Energy Lett. 2025, 10 (5), 2150– 2159, DOI: 10.1021/acsenergylett.5c00182There is no corresponding record for this reference.
- 47Grandhi, G. K.; Dhama, R.; Viswanath, N. S. M.; Lisitsyna, E. S.; Al-Anesi, B.; Dana, J.; Sugathan, V.; Caglayan, H.; Vivo, P. Role of Self-Trapped Excitons in the Broadband Emission of Lead-Free Perovskite-Inspired Cu2AgBiI6. J. Phys. Chem. Lett. 2023, 14 (18), 4192– 4199, DOI: 10.1021/acs.jpclett.3c00439There is no corresponding record for this reference.
- 48Ma, Z.; Lv, P.; He, X.; Wang, F.; Li, Y.; Xiao, G.; Zou, B. Self-Trapped Excitons or Bi3+ Ions for Broad Emission in a Lead-Free Double Perovskite? Hearing What Pressure Says. Nano Lett. 2025, 25 (23), 9345– 9352, DOI: 10.1021/acs.nanolett.5c01709There is no corresponding record for this reference.
- 49Saikia, S.; Gopal, A.; Rathod, R.; Joshi, A.; Priolkar, K. R.; Saha, S.; Santra, P. K.; Shanmuganathan, K.; Nag, A. Ultrabroad Near Infrared Emitting Perovskites. Angew. Chem., Int. Ed. 2025, 64 (2), e202415003 DOI: 10.1002/anie.202415003There is no corresponding record for this reference.
- 50Wang, Y.; Dang, P.; Zeng, Z.; Liu, D.; Zhang, G.; Tian, L.; Li, K.; Ma, P. a.; Wei, Y.; Lian, H.; Hou, Z.; Li, G.; Lin, J. Sensitizing effect of lanthanide luminescence by Mo4+/Ag+ in double perovskites: great enhancement of near-infrared emission via wide range of excitation (250–850 nm). Light Sci. Appl. 2026, 15 (1), 87, DOI: 10.1038/s41377-025-02159-4There is no corresponding record for this reference.
- 51Wang, Z.; Xu, S.; Gu, Y.; Li, Y.; Gao, Y.; Zhu, G.; Wang, Y.; Yu, H.; Chen, B. Enhancement of Blue-Light-Excited NIR-II Emission of Er3+ in Double Perovskites via Mo4+ Sensitization. Laser Photonics Rev. 2025, e02353 DOI: 10.1002/lpor.202502353There is no corresponding record for this reference.
- 52Rajendran, V.; Fang, M. H.; Guzman, G. N. D.; Lesniewski, T.; Mahlik, S.; Grinberg, M.; Leniec, G.; Kaczmarek, S. M.; Lin, Y. S.; Lu, K. M.; Lin, C. M.; Chang, H.; Hu, S. F.; Liu, R. S. Super Broadband Near-Infrared Phosphors with High Radiant Flux as Future Light Sources for Spectroscopy Applications. ACS Energy Lett. 2018, 3 (11), 2679– 2684, DOI: 10.1021/acsenergylett.8b01643There is no corresponding record for this reference.
- 53Fang, M. H.; Chen, K. C.; Majewska, N.; Leśniewski, T.; Mahlik, S.; Leniec, G.; Kaczmarek, S. M.; Yang, C. W.; Lu, K. M.; Sheu, H. S.; Liu, R. S. Hidden Structural Evolution and Bond Valence Control in Near-Infrared Phosphors for Light-Emitting Diodes. ACS Energy Lett. 2021, 6 (1), 109– 114, DOI: 10.1021/acsenergylett.0c02373There is no corresponding record for this reference.
- 54Rajendran, V.; Chen, K. C.; Huang, W. T.; Kamiński, M.; Grzegorczyk, M.; Mahlik, S.; Leniec, G.; Lu, K. M.; Wei, D. H.; Chang, H.; Liu, R. S. Unraveling Luminescent Energy Transfer Pathways: Futuristic Approach of Miniature Shortwave Infrared Light-Emitting Diode Design. ACS Energy Lett. 2023, 8 (5), 2395– 2400, DOI: 10.1021/acsenergylett.3c00680There is no corresponding record for this reference.
- 55Chen, K. C.; Hsiao, Y. H.; Chien, C. L.; Huang, W. T.; Majewska, N.; Mazurek, M. R.; Leśniewski, T.; Mahlik, S.; Leniec, G.; Cherng, D. H.; Lu, K. M.; Huang, S. L.; Liu, R. S. Bifunctional Energy Efficient (Ga,Ge)2O3:Cr3+,Ni2+ Phosphor for Shortwave Infrared Optical Applications. ACS Energy Lett. 2025, 10 (7), 3050– 3057, DOI: 10.1021/acsenergylett.5c01251There is no corresponding record for this reference.
- 56Fang, M. H.; Yang, T. H.; Lesniewski, T.; Lee, C.; Mahlik, S.; Grinberg, M.; Peterson, V. K.; Didier, C.; Pang, W. K.; Su, C.; Liu, R. S. Hydrogen-Containing Na3HTi1-xMnxF8 Narrow-Band Phosphor for Light-Emitting Diodes. ACS Energy Lett. 2019, 4 (2), 527– 533, DOI: 10.1021/acsenergylett.8b02307There is no corresponding record for this reference.
- 57Chang, C. Y.; Majewska, N.; Chen, K. C.; Huang, W. T.; Leśniewski, T.; Leniec, G.; Kaczmarek, S. M.; Pang, W. K.; Peterson, V. K.; Cherng, D. H.; Lu, K. M.; Mahlik, S.; Liu, R. S. Broadening Phosphor-Converted Light-Emitting Diode Emission: Controlling Disorder. Chem. Mater. 2022, 34 (22), 10190– 10199, DOI: 10.1021/acs.chemmater.2c03045There is no corresponding record for this reference.
- 58Zhang, A.; Liu, Y.; Liu, G.; Xia, Z. Dopant and Compositional Modulation Triggered Broadband and Tunable Near-Infrared Emission in Cs2Ag1-xNaxInCl6:Cr3+ Nanocrystals. Chem. Mater. 2022, 34 (7), 3006– 3012, DOI: 10.1021/acs.chemmater.1c03878There is no corresponding record for this reference.
- 59Snoeren, T. J.; Pressler, K.; Kluherz, K. T.; Walsh, K. M.; De Yoreo, J. J.; Gamelin, D. R. Luminescence and Covalency in Ytterbium-Doped CrX3 (X = Cl, Br, I) van der Waals Compounds. J. Am. Chem. Soc. 2023, 145 (31), 17427– 17434, DOI: 10.1021/jacs.3c05989There is no corresponding record for this reference.
- 60Lyu, K.; Liu, G.; Molokeev, M. S.; Xia, Z. Double-Site Occupation Triggered Broadband and Tunable NIR-I and NIR-II Luminescence in AlNbO4:Cr3+ Phosphors. Adv. Phys. Res. 2023, 2 (4), 2200056, DOI: 10.1002/apxr.202200056There is no corresponding record for this reference.
- 61Dou, C.; Liang, T.; Zhao, M.; Song, Z.; Ning, L.; Peng, D.; Liu, Q. Self-Recoverable and NIR-I to NIR-II Tunable Broadband Mechanoluminescence of Cr3+-Doped Antimonate Double Perovskites. Adv. Funct. Mater. 2025, 35 (14), 2419716, DOI: 10.1002/adfm.202419716There is no corresponding record for this reference.
- 62Gamelin, D. R.; Güdel, H. U. Design of Luminescent Inorganic Materials: New Photophysical Processes Studied by Optical Spectroscopy. Acc. Chem. Res. 2000, 33 (4), 235– 242, DOI: 10.1021/ar990102yThere is no corresponding record for this reference.
- 63Aebischer, A.; Wenger, O. S.; Güdel, H. U. Near-infrared to visible photon up-conversion in V3+, Re4+ co-doped Cs2NaYCl6. J. Lumin. 2003, 102–103, 48– 53, DOI: 10.1016/S0022-2313(02)00528-8There is no corresponding record for this reference.
- 64Ravi, V. K.; Scheidt, R. A.; Nag, A.; Kuno, M.; Kamat, P. V. To Exchange or Not to Exchange. Suppressing Anion Exchange in Cesium Lead Halide Perovskites with PbSO4-Oleate Capping. ACS Energy Lett. 2018, 3 (4), 1049– 1055, DOI: 10.1021/acsenergylett.8b00380There is no corresponding record for this reference.
- 65Zhu, D.; Zito, J.; Pinchetti, V.; Dang, Z.; Olivati, A.; Pasquale, L.; Tang, A.; Zaffalon, M. L.; Meinardi, F.; Infante, I.; De Trizio, L.; Manna, L.; Brovelli, S. Compositional Tuning of Carrier Dynamics in Cs2Na1-xAgxBiCl6 Double-Perovskite Nanocrystals. ACS Energy Lett. 2020, 5 (6), 1840– 1847, DOI: 10.1021/acsenergylett.0c00914There is no corresponding record for this reference.
- 66Liang, W.; Li, Y.; Xiang, D.; Han, Y.; Jiang, Q.; Zhang, W.; Wu, K. Efficient Optical Orientation and Slow Spin Relaxation in Lead-Free CsSnBr3 Perovskite Nanocrystals. ACS Energy Lett. 2021, 6 (5), 1670– 1676, DOI: 10.1021/acsenergylett.1c00413There is no corresponding record for this reference.
- 67Dey, A.; Ye, J.; De, A.; Debroye, E.; Ha, S. K.; Bladt, E.; Kshirsagar, A. S.; Wang, Z.; Yin, J.; Wang, Y.; Quan, L. N.; Yan, F.; Gao, M.; Li, X.; Shamsi, J.; Debnath, T.; Cao, M.; Scheel, M. A.; Kumar, S.; Steele, J. A.; Gerhard, M.; Chouhan, L.; Xu, K.; Wu, X. g.; Li, Y.; Zhang, Y.; Dutta, A.; Han, C.; Vincon, I.; Rogach, A. L.; Nag, A.; Samanta, A.; Korgel, B. A.; Shih, C. J.; Gamelin, D. R.; Son, D. H.; Zeng, H.; Zhong, H.; Sun, H.; Demir, H. V.; Scheblykin, I. G.; Mora-Seró, I.; Stolarczyk, J. K.; Zhang, J. Z.; Feldmann, J.; Hofkens, J.; Luther, J. M.; Pérez-Prieto, J.; Li, L.; Manna, L.; Bodnarchuk, M. I.; Kovalenko, M. V.; Roeffaers, M. B. J.; Pradhan, N.; Mohammed, O. F.; Bakr, O. M.; Yang, P.; Müller-Buschbaum, P.; Kamat, P. V.; Bao, Q.; Zhang, Q.; Krahne, R.; Galian, R. E.; Stranks, S. D.; Bals, S.; Biju, V.; Tisdale, W. A.; Yan, Y.; Hoye, R. L. Z.; Polavarapu, L. State of the Art and Prospects for Halide Perovskite Nanocrystals. ACS Nano 2021, 15 (7), 10775– 10981, DOI: 10.1021/acsnano.0c08903There is no corresponding record for this reference.
- 68Chen, Z.; Hoye, R. L. Z.; Yip, H. L.; Fiuza-Maneiro, N.; López-Fernández, I.; Otero-Martínez, C.; Polavarapu, L.; Mondal, N.; Mirabelli, A.; Anaya, M.; Stranks, S. D.; Liu, H.; Shi, G.; Xiao, Z.; Kim, N.; Kim, Y.; Shin, B.; Shi, J.; Liu, M.; Zhang, Q.; Fan, Z.; Loy, J. C.; Zhao, L.; Rand, B. P.; Arfin, H.; Saikia, S.; Nag, A.; Zou, C.; Lin, L. Y.; Xiang, H.; Zeng, H.; Liu, D.; Su, S. J.; Wang, C.; Zhong, H.; Xuan, T. T.; Xie, R. J.; Bao, C.; Gao, F.; Gao, X.; Qin, C.; Kim, Y. H.; Beard, M. C. Roadmap on perovskite light-emitting diodes. J. Phys. Photonics 2024, 6 (3), 032501, DOI: 10.1088/2515-7647/ad46a6There is no corresponding record for this reference.
- 69Mukherjee, P.; Das, R.; Pariari, D.; Das, K.; Mahadevan, P.; Sarma, D. D. Energy-Transfer Pathways That Sensitize Mn2+ Emissions Using Mn@CsPbCl3 Nanocrystals as a Generic Platform. ACS Energy Lett. 2025, 10 (12), 6381– 6389, DOI: 10.1021/acsenergylett.5c02937There is no corresponding record for this reference.
- 70Shukla, A.; Kaur, G.; Justice Babu, K.; Bhatt, H.; Kumar, V.; Ghosh, H. N. The Retarded Hot Carrier Relaxation and Augmented Photoconductivity in Eu3+-Doped CsPbBr3 Nanocrystals. ACS Photonics 2025, 12 (8), 4224– 4233, DOI: 10.1021/acsphotonics.5c00396There is no corresponding record for this reference.
- 71Ji, F.; Klarbring, J.; Zhang, B.; Wang, F.; Wang, L.; Miao, X.; Ning, W.; Zhang, M.; Cai, X.; Bakhit, B.; Magnuson, M.; Ren, X.; Sun, L.; Fahlman, M.; Buyanova, I. A.; Chen, W. M.; Simak, S. I.; Abrikosov, I. A.; Gao, F. Remarkable Thermochromism in the Double Perovskite Cs2NaFeCl6. Adv. Opt. Mater. 2024, 12 (8), 2301102, DOI: 10.1002/adom.202301102There is no corresponding record for this reference.
- 72Boyer, J. C.; Cuccia, L. A.; Capobianco, J. A. Synthesis of Colloidal Upconverting NaYF4: Er3+/Yb3+ and Tm3+/Yb3+ Monodisperse Nanocrystals. Nano Lett. 2007, 7 (3), 847– 852, DOI: 10.1021/nl070235+There is no corresponding record for this reference.
- 73Ghosh, P.; Kar, A.; Patra, A. Structural and photoluminescence properties of doped and core-shell LaPO4:Eu3+ nanocrystals. J. Appl. Phys. 2010, 108 (11), na, DOI: 10.1063/1.3514137There is no corresponding record for this reference.
- 74Tzanetopoulos, E.; Schwartz, J.; Gamelin, D. R. HF-Free synthesis of colloidal Cs2ZrF6 and (NH4)2ZrF6 nanocrystals. Chem. Commun. 2023, 59 (36), 5451– 5454, DOI: 10.1039/D3CC00374DThere is no corresponding record for this reference.
- 75Imran, M.; Caligiuri, V.; Wang, M.; Goldoni, L.; Prato, M.; Krahne, R.; De Trizio, L.; Manna, L. Benzoyl Halides as Alternative Precursors for the Colloidal Synthesis of Lead-Based Halide Perovskite Nanocrystals. J. Am. Chem. Soc. 2018, 140 (7), 2656– 2664, DOI: 10.1021/jacs.7b13477There is no corresponding record for this reference.
- 76Ming, J.; Chen, Y.; Miao, H.; Fan, Y.; Wang, S.; Chen, Z.; Guo, Z.; Guo, Z.; Qi, L.; Wang, X.; Yun, B.; Pei, P.; He, H.; Zhang, H.; Tang, Y.; Zhao, D.; Wong, G. K.-L.; Bünzli, J.-C. G.; Zhang, F. High-brightness transition metal-sensitized lanthanide near-infrared luminescent nanoparticles. Nat. Photonics 2024, 18 (12), 1254– 1262, DOI: 10.1038/s41566-024-01517-9There is no corresponding record for this reference.
- 77Kong, Q.; Jiang, S.; Wang, Z.; Xu, X.; Zhang, R.; Zhu, G.; Yang, J.; Han, P.; Liu, R.; Hong, F.; Luo, N.; Chen, J.; Yang, B. Highly Stable Cesium Molybdenum Chloride Perovskite Nanocrystals for Photothermal Semihydrogenation Applications. ACS Appl. Mater. Interfaces 2024, 16 (27), 35752– 35760, DOI: 10.1021/acsami.4c05157There is no corresponding record for this reference.
- 78Mondal, B.; Mehta, S.; Mondal, A.; Nag, A. W-Doped Cs2SnCl6 for Near-Infrared Emission. J. Phys. Chem. Lett. 2025, 16 (20), 4937– 4943, DOI: 10.1021/acs.jpclett.5c00822There is no corresponding record for this reference.
- 79Saikia, S.; Ghosh, A.; Nag, A. Broad Dual Emission by Codoping Cr3+ (d→d) and Bi3+ (s→p) in Cs2Ag0.6Na0.4InCl6 Double Perovskite. Angew. Chem. Inter. Ed. 2023, 62 (33), e202307689 DOI: 10.1002/anie.202307689There is no corresponding record for this reference.
- 80Tripathi, V. M.; Saikia, S.; Nag, A. Li(Sc0.70Ga0.24Cr0.06)Ge2O6 in Polylactic Acid for Robust Near-Infrared Phosphor-Converted Light-Emitting Diodes. Chem. Mater. 2025, 37 (18), 7462– 7470, DOI: 10.1021/acs.chemmater.5c01897There is no corresponding record for this reference.
- 81Li, X.; Li, J.; Wang, B.; Pan, D.; Gu, L.; Zhang, J.; Zhang, L.; Kong, Y.; Huang, J. Near-Infrared Luminescence in Mo4+-Activated Metal Halide for Advanced Optoelectronics. Inorg. Chem. 2025, 64 (45), 22384– 22392, DOI: 10.1021/acs.inorgchem.5c03606There is no corresponding record for this reference.
- 82Li, J.; Du, Z.; Li, X.; Zhang, L.; Gu, L.; Pan, D.; Zhang, J.; Wang, B. Mo4+-Doped Cs4ZnBi2Cl12 Lead-Free Double Perovskite for Near-Infrared Luminescence. Luminescence 2025, 40 (11), e70352 DOI: 10.1002/bio.70352There is no corresponding record for this reference.
- 83Pan, J. L.; Yu, Y. J.; Wang, Y. K.; Liao, L. S. Lanthanide Ion-Doped Perovskite Nanocrystals in Electroluminescent Device. Adv. Funct. Mater. 2024, 34 (36), 2401327, DOI: 10.1002/adfm.202401327There is no corresponding record for this reference.
- 84Wang, H.; Treglia, A.; Albaqami, M. D.; Gao, F.; Petrozza, A. Tin-Halide Perovskites for Near-Infrared Light-Emitting Diodes. ACS Energy Lett. 2024, 9 (6), 2500– 2507, DOI: 10.1021/acsenergylett.4c00470There is no corresponding record for this reference.



