From Waste to Reuse: The Valorization of Almond Shells (Prunus dulcis) as a Sustainable Biosorbent for the Removal and Recovery of Phenolic Compounds in Winery WastewaterClick to copy article linkArticle link copied!
- Elyse DoriaElyse DoriaDepartment of Food Science and Technology, University of California, Davis, One Shields Avenue, Davis, California 95616, United StatesMore by Elyse Doria
- Robert ColemanRobert ColemanDepartment of Viticulture and Enology, Washington State University, Richland, Washington 99162, United StatesMore by Robert Coleman
- Larry LernoLarry LernoDepartment of Viticulture and Enology, University of California, Davis, One Shields Avenue, Davis, California 95616, United StatesMore by Larry Lerno
- Poll ZhangPoll ZhangDepartment of Food Science and Technology, University of California, Davis, One Shields Avenue, Davis, California 95616, United StatesMore by Poll Zhang
- Lingchuan HaoLingchuan HaoDepartment of Food Science and Technology, University of California, Davis, One Shields Avenue, Davis, California 95616, United StatesMore by Lingchuan Hao
- Alyson Mitchell*Alyson Mitchell*Email: [email protected]Department of Food Science and Technology, University of California, Davis, One Shields Avenue, Davis, California 95616, United StatesMore by Alyson Mitchell
Abstract
Almond shell powder (ASP) derived from Nonpareil (soft-shell) and Peerless (hard-shell) almond varieties was evaluated as a natural bioadsorbent for removing phenolic compounds from model and industrial winery wastewater. Binding kinetics, particle size, mass-to-liquid ratio (m/v), and recovery efficiency were assessed. Nonpareil and Peerless adsorption fit an Elovich kinetic model (R2 = 0.986 and 0.982, respectively). Optimal adsorption occurred at a 0.2–0.5 mm particle size and a 1:10 ASP-to-wastewater ratio (m/v), yielding 79.05 ± 0.79% (0.84 ± 0.01 mg g–1) total phenolic content. Phenolic recoveries were 70.26 ± 0.18% (0.51 ± 0.01 mg g–1) for Nonpareil and 52.36 ± 0.30% (0.44 ± 0.00 mg g–1) for Peerless using optimized conditions: 50:50 ethanol/water (v/v) at a 1:25 ASP-to-solvent ratio (m/v). These findings demonstrate that ASP is a viable, low-cost, and sustainable material for phenolic removal from winery wastewater with the added benefit of enabling phenolic compound recovery.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Introduction
Methods and Materials
Reagents and Standards
Stock Solutions
Almond Shell Samples
Winery Wastewater Samples
Water Holding Capacity (WHC)
Evaluation of the Particle Size
Evaluation of the ASP-to-Model Winery Wastewater Ratio
Evaluation of the pH on Phenolic Adsorption
Evaluation of the Initial Phenolic Concentration on Adsorption
Adsorption Kinetics
Recovery of Phenolic Compounds
Reusability
Scanning Electron Microscopy (SEM) Imaging
Analytical Methods
Statistical Analysis
Results and Discussion
Figure 1
Figure 1. SEM local imaging of Nonpareil (soft shell) and Peerless (hard shell) almond shells at 0.2–0.5, 0.5–1, and 1–2 mm particle sizes at 600× magnification.
WHC
| shell type | particle range | g of water/g of dry ASP | % WHC |
|---|---|---|---|
| Nonpareil (soft-shell) | 0.2–0.5 mm | 4.0 ± 0.6a | 398.2 ± 62.1a |
| 0.5–1.0 mm | 1.7 ± 0.4c | 168.9 ± 40.3c | |
| 1.0–2.0 mm | 1.1 ± 0.2d | 110.6 ± 15.9d | |
| Peerless (hard-shell) | 0.2–0.5 mm | 2.4 ± 0.4b | 237.2 ± 41.2b |
| 0.5–1.0 mm | 0.9 ± 0.1d | 89.6 ± 7.5d | |
| 1.0–2.0 mm | 0.7 ± 0.0d | 69.0 ± 4.7d |
Different letters indicate a statistically significant difference (p < 0.05) within the column based on Tukey’s HSD pairwise comparisons.
Model Winery Wastewater
Particle Size Effect
Figure 2
Figure 2. Percent phenolic adsorption onto Nonpareil (soft-shell) and Peerless (hard-shell) ASP exposed to model winery wastewater at three particle size ranges: 0.2–0.5, 0.5–1.0, and 1.0–2.0 mm. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Mass-to-Volume Ratio Effect
Figure 3
Figure 3. Percent phenolic adsorption for Nonpariel and Peerless ASPs of 0.2–0.5 mm exposed to model winery wastewater at 1:10, 1:25, and 1:50 (m/v) ratio. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Effect of the pH on Adsorption
Effect of the Initial Concentration on Adsorption
Adsorption Kinetics
| Nonpareil (soft-shell) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| PFO | PSO | Elovich | |||||||
| compound | k1 min–1 | qe mg/g | R2 | k2 mg/g·min | qe mg/g | R2 | Α mg/g·min | Β mg/g·min | R2 |
| gallic acid | 0.110 | 0.597 | 0.848 | 0.232 | 0.650 | 0.910 | 0.345 | 9.588 | 0.976 |
| (+)-catechin | 0.250 | 0.613 | 0.773 | 0.485 | 0.669 | 0.872 | 1.844 | 11.570 | 0.974 |
| caffeic acid | 0.339 | 0.703 | 0.821 | 0.588 | 0.761 | 0.905 | 5.349 | 11.401 | 0.986 |
| rutin | 0.667 | 0.445 | 0.881 | 1.933 | 0.474 | 0.937 | 87.391 | 25.411 | 0.990 |
| malvidin-3-O-glucoside | 0.335 | 0.136 | 0.887 | 3.511 | 0.145 | 0.956 | 2.572 | 67.355 | 0.994 |
| total phenolics | 0.266 | 2.475 | 0.819 | 0.136 | 2.678 | 0.904 | 10.496 | 3.031 | 0.986 |
| Peerless (hard-shell) | |||||||||
| PFO | PSO | Elovich | |||||||
| compound | k1 min–1 | qe mg/g | R2 | k2 mg/g·min | qe mg/g | R2 | Α mg/g·min | Β mg/g·min | R2 |
| gallic acid | 0.053 | 0.661 | 0.863 | 0.101 | 0.727 | 0.878 | 0.190 | 8.293 | 0.912 |
| (+)-catechin | 0.105 | 0.638 | 0.834 | 0.219 | 0.690 | 0.900 | 0.416 | 9.310 | 0.979 |
| caffeic acid | 0.122 | 0.758 | 0.861 | 0.220 | 0.816 | 0.927 | 0.680 | 8.233 | 0.992 |
| rutin | 0.310 | 0.299 | 0.882 | 1.404 | 0.320 | 0.951 | 3.128 | 28.563 | 0.996 |
| malvidin-3-O-glucoside | 0.189 | 0.095 | 0.896 | 2.742 | 0.102 | 0.959 | 0.231 | 75.221 | 0.999 |
| total phenolics | 0.105 | 2.444 | 0.849 | 0.062 | 2.613 | 0.909 | 1.987 | 2.561 | 0.982 |
Figure 4
Figure 4. Mean total phenolic adsorption of phenolics onto Nonpareil (a) and Peerless ASP (b) and their fit to pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich kinetic models. Bars indicate standard deviation across replicates.
Canonical Variate Analysis
Figure 5
Figure 5. Canonical variate analysis (CVA) of compound adsorption behavior on ASP. CVA showing a) score plot and b)loadings of adsorption parameters and compound characteristics, including α and β rates from the Elovich model, and adsorption at 240 min (mol g–1). Arrows indicate the direction and strength of each variable’s contribution to the canonical variates. CVA scores plot displaying individual compounds projected along the canonical variates.
Optimization of Phenolic Recovery
Figure 6
Figure 6. Percent phenolic recovery for Nonpareil and Peerless ASP of 0.2–0.5 mm exposed to ethanol:water solutions at 0:100, 20:80, 50:50, and 70:30 (v/v). Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Figure 7
Figure 7. Recovery of phenolic compounds from Nonpareil and Peerless ASP using 50:50 ethanol/water (v/v) and varying the ratio of ASP/winery wastewater at 1:10, 1:20, and 1:25 m/v. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Reusability of ASPs
Applications to Industrial Winery Wastewater
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsfoodscitech.5c00652.
Phenolic adsorption and recoveries for Nonpareil and Peerless ASP at various pH values, initial concentrations, and reusability (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
The authors thank Blue Diamond Growers and Treasury Wine Estates. We also extend our appreciation to Charlene Hui for her assistance with sample preparation and data acquisition and to Patrick Gravesen for his invaluable technical support.
| ANOVA | analysis of variance |
| ASP | almond shell powder |
| BOD | oxygen demand |
| COD | chemical oxygen demand |
| CVA | canonical variate analysis |
| DAD | diode-array detector |
| DI | deionized |
| DPR | direct potable reuse |
| HPLC | high-performance liquid chromatography |
| MANOVA | multivariate analysis of variance |
| m/v | mass-to-volume |
| PFO | pseudo-first-order |
| PSO | pseudo-second-order |
| SEM | scanning electron microscopy |
| WHC | water holding capacity |
References
This article references 65 other publications.
- 1California & US Wine Production. Wine Institute. https://wineinstitute.org/our-industry/statistics/california-us-wine-production/ (accessed 2024-09-30).Google ScholarThere is no corresponding record for this reference.
- 2Jorge, N.; Teixeira, A. R.; Gomes, A.; Peres, J. A.; Lucas, M. S. Winery wastewater: challenges and perspectives. Eng. Proc. 2023, 56 (267). DOI: 10.3390/ASEC2023-15968 .Google ScholarThere is no corresponding record for this reference.
- 3Welz, P. J.; Holtman, G.; Haldenwang, R.; Le Roes-Hill, M. Characterisation of winery wastewater from continuous flow settling basins and waste stabilisation ponds over the course of 1 year: Implications for biological wastewater treatment and land application. Water Sci. Technol. 2016, 74 (9), 2036– 2050, DOI: 10.2166/wst.2016.226Google ScholarThere is no corresponding record for this reference.
- 4Vlyssides, A. G.; Barampouti, E. M.; Mai, S. Wastewater characteristics from greek wineries and distilleries. Water Sci. Technol. 2005, 51 (1), 53– 60, DOI: 10.2166/wst.2005.0007Google ScholarThere is no corresponding record for this reference.
- 5Bories, A.; Sire, Y. Impacts of winemaking methods on wastewaters and their treatment. S. Afr. J. Enol. Vitic. 2016, 31 (1), 38– 44, DOI: 10.21548/31-1-1398Google ScholarThere is no corresponding record for this reference.
- 6Fiorentino, A.; Gentili, A.; Isidori, M.; Monaco, P.; Nardelli, A.; Parrella, A.; Temussi, F. Environmental effects caused by olive mill wastewaters: toxicity comparison of low-molecular-eeight phenol components. J. Agric. Food Chem. 2003, 51 (4), 1005– 1009, DOI: 10.1021/jf020887dGoogle ScholarThere is no corresponding record for this reference.
- 7Stom, D. I.; Roth, R. Some effects of polyphenols on aquatic plants: I. toxicity of phenols in aquatic plants. Bull. Environ. Contam. Toxicol. 1981, 27 (3), 332– 337, DOI: 10.1007/BF01611028Google ScholarThere is no corresponding record for this reference.
- 8Siqueira, J. O.; Nair, M. G.; Hammerschmidt, R.; Safir, G. R.; Putnam, A. R. Significance of phenolic compounds in plant-soil-microbial systems. CRC Crit. Rev. Plant Sci. 1991, 10 (1), 63– 121, DOI: 10.1080/07352689109382307Google ScholarThere is no corresponding record for this reference.
- 9Cueva, C.; Moreno-Arribas, M. V.; Martín-Alvarez, P. J.; Bills, G.; Vicente, M. F.; Basilio, A.; Rivas, C. L.; Requena, T.; Rodríguez, J. M.; Bartolomé, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161 (5), 372– 382, DOI: 10.1016/j.resmic.2010.04.006Google ScholarThere is no corresponding record for this reference.
- 10Lofrano, G.; Meric, S. A comprehensive approach to winery wastewater treatment: A review of the state-of the-art. Desalin. Water Treat. 2016, 57 (7), 3011– 3028, DOI: 10.1080/19443994.2014.982196Google ScholarThere is no corresponding record for this reference.
- 11Water Resources Control Board. State Water Resources Control Board Order WQ XXXX-XXXX-DWQ General waste discharge requirements for winery process water public draft ; 2020.Google ScholarThere is no corresponding record for this reference.
- 12Mosse, K. P. M.; Patti, A. F.; Christen, E. W.; Cavagnaro, T. R. Review: Winery wastewater quality and treatment options in Australia. Aust. J. Grape Wine Res. 2011, 17 (2), 111– 122, DOI: 10.1111/j.1755-0238.2011.00132.xGoogle ScholarThere is no corresponding record for this reference.
- 13Mohamad Said, K. A.; Ismail, A. F.; Abdul Karim, Z.; Abdullah, M. S.; Hafeez, A. A review of technologies for the phenolic compounds recovery and phenol removal from wastewater. Process Saf. Environ. Prot. 2021, 151, 257– 289, DOI: 10.1016/j.psep.2021.05.015Google ScholarThere is no corresponding record for this reference.
- 14Bories, A.; Guillot, J.-M.; Sire, Y.; Couderc, M.; Lemaire, S.-A.; Kreim, V.; Roux, J.-C. Prevention of volatile fatty acids production and limitation of odours from winery wastewaters by denitrification. Water Res. 2007, 41 (13), 2987– 2995, DOI: 10.1016/j.watres.2007.03.022Google ScholarThere is no corresponding record for this reference.
- 15Ngwenya, N.; Gaszynski, C.; Ikumi, D. A review of winery wastewater treatment: A focus on UASB biotechnology optimization and recovery strategies. J. Environ. Chem. Eng. 2022, 10 (4), 108172, DOI: 10.1016/j.jece.2022.108172Google ScholarThere is no corresponding record for this reference.
- 16Lahnsteiner, J.; van Rensburg, P.; Esterhuizen, J. Direct potable reuse - a feasible water management option. J. Water Reuse Desalin. 2018, 8 (1), 14– 28, DOI: 10.2166/wrd.2017.172Google ScholarThere is no corresponding record for this reference.
- 17Khan, S. J.; Anderson, R. Potable reuse: Experiences in Australia. Curr. Opin. Environ. Sci. Health 2018, 2, 55– 60, DOI: 10.1016/j.coesh.2018.02.002Google ScholarThere is no corresponding record for this reference.
- 18California Almond Board. 2023 Almond Almanac. https://www.almonds.com/sites/default/files/2023-12/Almanac_2023.pdf (accessed 2025-05-26).Google ScholarThere is no corresponding record for this reference.
- 19Kaur, M.; Kumar, M.; Sachdeva, S.; Puri, S. K. An efficient multiphase bioprocess for enhancing the renewable energy production from almond shells. Energy Convers. Manage. 2020, 203, 112235, DOI: 10.1016/j.enconman.2019.112235Google ScholarThere is no corresponding record for this reference.
- 20Prgomet, I.; Gonçalves, B.; Domínguez-Perles, R.; Pascual-Seva, N.; Barros, A. I. R. N. A. Valorization challenges to almond residues: Phytochemical composition and functional application. Molecules. 2017, 22 (10), 1774, DOI: 10.3390/molecules22101774Google ScholarThere is no corresponding record for this reference.
- 21Almond Board of California. 2022 Almond Almanac ; 2022. https://www.almonds.org/sites/default/files/2023-04/2022_Almanac.pdf (accessed 2025-10-06).Google ScholarThere is no corresponding record for this reference.
- 22Chiou, B.-S.; Valenzuela-Medina, D.; Bilbao-Sainz, C.; Klamczynski, A. P.; Avena-Bustillos, R. J.; Milczarek, R. R.; Du, W.-X.; Glenn, G. M.; Orts, W. J. Torrefaction of almond shells: Effects of torrefaction conditions on properties of solid and condensate products. Ind. Crops Prod. 2016, 86, 40– 48, DOI: 10.1016/j.indcrop.2016.03.030Google ScholarThere is no corresponding record for this reference.
- 23Li, X.; Liu, Y.; Hao, J.; Wang, W. Study of almond shell characteristics. Materials. 2018, 11 (9), 1782, DOI: 10.3390/ma11091782Google ScholarThere is no corresponding record for this reference.
- 24Tomishima, H.; Luo, K.; Mitchell, A. E. The almond (Prunus dulcis): chemical properties, utilization, and valorization of coproducts. Annu. Rev. Food Sci. Technol. 2022, 13 (1), 145– 166, DOI: 10.1146/annurev-food-052720-111942Google ScholarThere is no corresponding record for this reference.
- 25Ledbetter, C. A. Shell cracking strength in almond (Prunus dulcis [Mill.] D.A. Webb.) and its implication in uses as a value-added product. Bioresour. Technol. 2008, 99 (13), 5567– 5573, DOI: 10.1016/j.biortech.2007.10.059Google ScholarThere is no corresponding record for this reference.
- 26Zakzeski, J.; Bruijnincx, P. C. A.; Jongerius, A. L.; Weckhuysen, B. M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110 (6), 3552– 3599, DOI: 10.1021/cr900354uGoogle ScholarThere is no corresponding record for this reference.
- 27Azadi, P.; Inderwildi, O. R.; Farnood, R.; King, D. A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renew. Sustain. Energy Rev. 2013, 21, 506– 523, DOI: 10.1016/j.rser.2012.12.022Google ScholarThere is no corresponding record for this reference.
- 28Castro-Muñoz, R.; Yáñez-Fernández, J.; Fíla, V. Phenolic compounds recovered from agro-food by-products using membrane technologies: An overview. Food Chem. 2016, 213, 753– 762, DOI: 10.1016/j.foodchem.2016.07.030Google ScholarThere is no corresponding record for this reference.
- 29Soto, M. L.; Moure, A.; Domínguez, H.; Parajó, J. C. Batch and fixed bed column studies on phenolic adsorption from wine vinasses by polymeric resins. J. Food Eng. 2017, 209, 52– 60, DOI: 10.1016/j.jfoodeng.2017.04.008Google ScholarThere is no corresponding record for this reference.
- 30Thiel, A.; Tippkötter, N.; Suck, K.; Sohling, U.; Ruf, F.; Ulber, R. New zeolite adsorbents for downstream processing of polyphenols from renewable resources. Eng. Life Sci. 2013, 13 (3), 239– 246, DOI: 10.1002/elsc.201200188Google ScholarThere is no corresponding record for this reference.
- 31Kammerer, J.; Kammerer, D. R.; Jensen, U.; Carle, R. Interaction of apple polyphenols in a multi-compound system upon adsorption onto a food-grade resin. J. Food Eng. 2010, 96 (4), 544– 554, DOI: 10.1016/j.jfoodeng.2009.08.038Google ScholarThere is no corresponding record for this reference.
- 32Frascari, D.; Rubertelli, G.; Arous, F.; Ragini, A.; Bresciani, L.; Arzu, A.; Pinelli, D. Valorisation of olive mill wastewater by phenolic compounds adsorption: development and application of a procedure for adsorbent selection. Chem. Eng. J. 2019, 360, 124– 138, DOI: 10.1016/j.cej.2018.11.188Google ScholarThere is no corresponding record for this reference.
- 33Wang, Z.; Wang, C.; Yuan, J.; Zhang, C. Adsorption characteristics of adsorbent resins and antioxidant capacity for enrichment of phenolics from two-phase olive waste. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2017, 1040, 38– 46, DOI: 10.1016/j.jchromb.2016.11.023Google ScholarThere is no corresponding record for this reference.
- 34Ben Arfi, R.; Karoui, S.; Mougin, K.; Ghorbal, A. Adsorptive removal of cationic and anionic dyes from aqueous solution by utilizing almond shell as bioadsorbent. Euro–Mediterr. J. Environ. Integr. 2017, 2 (1). DOI: 10.1007/s41207-017-0032-y .Google ScholarThere is no corresponding record for this reference.
- 35Melhaoui, R.; Miyah, Y.; Kodad, S.; Houmy, N.; Addi, M.; Abid, M.; Mihamou, A.; Serghini-Caid, H.; Lairini, S.; Tijani, N.; Hano, C.; Elamrani, A. On the suitability of almond shells for the manufacture of a natural low-cost bioadsorbent to remove brilliant green: Kinetics and equilibrium isotherms study. Sci. World J. 2021, 2021. 1 DOI: 10.1155/2021/6659902 .Google ScholarThere is no corresponding record for this reference.
- 36Duran, C.; Ozdes, D.; Gundogdu, A.; Senturk, H. B. Kinetics and isotherm analysis of basic dyes adsorption onto almond shell (Prunus dulcis) as a low cost adsorbent. J. Chem. Eng. Data 2011, 56 (5), 2136– 2147, DOI: 10.1021/je101204jGoogle ScholarThere is no corresponding record for this reference.
- 37Ziarati, P.; Kermanshah, A.; Moslehishad, M. Adsorption heavy metal from contaminated water by modified shell of wild endemic almonds: Amygdalus lycioides and amygdalus wendelboi. Biosci. Biotechnol. Res. Asia. 2015, 12 (3), 2451– 2457, DOI: 10.13005/bbra/1923Google ScholarThere is no corresponding record for this reference.
- 38Mirmohammadmakki, F.; Gharachorloo, M.; Ghavami, M.; Abdossi, V.; Azizinezhad, R. The efficiency of almond shell (Amygdalus communis L.) bio-sorption in reduction of heavy metals (lead, cadmium, arsenic, and nickel) from parsley (Petroselinum crispum). Biomass Convers. Biorefin. 2023, 13 (2), 16149– 16160, DOI: 10.1007/s13399-022-02635-6Google ScholarThere is no corresponding record for this reference.
- 39Taha, A. A.; Moustafa, A. H. E.; Abdel-Rahman, H. H.; Abd El-Hameed, M. M. A. Comparative biosorption study of Hg (II) using raw and chemically activated almond shell. Adsorp. Sci. Technol. 2018, 36 (1–2), 521– 548, DOI: 10.1177/0263617417705473Google ScholarThere is no corresponding record for this reference.
- 40McCaffrey, Z.; Torres, L. F.; Chiou, B.-S.; Hart-Cooper, W.; McMahan, C.; Orts, W. J. Almond and walnut shell activated carbon for methylene blue adsorption. ACS Sustainable Resour. Manage. 2024, 1 (7), 1421– 1431, DOI: 10.1021/acssusresmgt.4c00080Google ScholarThere is no corresponding record for this reference.
- 41Omri, A.; Benzina, M.; Ammar, N. Preparation, modification and industrial application of activated carbon from almond shell. J. Ind. Eng. Chem. 2013, 19 (6), 2092– 2099, DOI: 10.1016/j.jiec.2013.03.025Google ScholarThere is no corresponding record for this reference.
- 42Loulidi, I.; Boukhlifi, F.; Ouchabi, M.; Amar, A.; Jabri, M.; Kali, A.; Chraibi, S.; Hadey, C.; Aziz, F. Adsorption of crystal violet onto an agricultural waste residue: Kinetics, isotherm, thermodynamics, and mechanism of adsorption. Sci. World J. 2020, 2020, 5873521, DOI: 10.1155/2020/5873521Google ScholarThere is no corresponding record for this reference.
- 43Liao, M.; Kelley, S.; Yao, Y. Generating energy and greenhouse gas inventory data of activated carbon production using machine learning and kinetic based process simulation. ACS Sustain. Chem. Eng. 2020, 8 (2), 1252– 1261, DOI: 10.1021/acssuschemeng.9b06522Google ScholarThere is no corresponding record for this reference.
- 44Ardia, D.; Boudt, K.; Carl, P.; Mullen, K. M.; Peterson, B. G. Differential evolution with DEoptim: An application to non-convex portfolio optimization. R J. 2011, 3 (1), 27– 34, DOI: 10.32614/RJ-2011-005Google ScholarThere is no corresponding record for this reference.
- 45R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2025. https://www.R-project.org.Google ScholarThere is no corresponding record for this reference.
- 46Posit team. RStudio: Integrated Development Environment for R. Posit Software, PBC: Boston, MA, 2025. http://www.posit.co/.Google ScholarThere is no corresponding record for this reference.
- 47Pinton, S.; Furlan Goncalves Dias, F.; Lerno, L. A.; Barile, D.; Leite Nobrega de Moura Bell, J. M. Revitalizing unfermented cabernet sauvignon pomace using an eco-friendly, two-stage countercurrent process: role of pH on the extractability of bioactive phenolics. Processes. 2022, 10 (10), 2093, DOI: 10.3390/pr10102093Google ScholarThere is no corresponding record for this reference.
- 48Harbertson, J. F.; Mireles, M.; Yu, Y. Improvement of BSA tannin precipitation assay by reformulation of resuspension buffer. Am. J. Enol. Vitic. 2015, 66 (1), 95– 99, DOI: 10.5344/ajev.2014.14082Google ScholarThere is no corresponding record for this reference.
- 49Harbertson, J. F.; DeBeer, D.; Waterhouse, A. L.; Adams, D. O. Use of ferric chloride for the measurement of total phenolics in red and white wines. Am. J. Enol. Vitic. 2004, 55, 295AGoogle ScholarThere is no corresponding record for this reference.
- 50Waterhouse, L.; Wrolstad, R. E. Determination of total phenolics. Current Protocols in Food Analytical Chemistry; John Wiley & Sons: New York, 2002. DOI: 10.1002/0471142913.fai0101s06 .Google ScholarThere is no corresponding record for this reference.
- 51Singleton, V. L.; Orthofer, R.; Lamuela-Raventós, R. M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152– 178, DOI: 10.1016/S0076-6879(99)99017-1Google ScholarThere is no corresponding record for this reference.
- 52Friendly, M.; Fox, J. Candisc: Visualizing generalized canonical discriminant and canonical correlation analysis. 2024. https://CRAN.R-project.org/package=heplots.Google ScholarThere is no corresponding record for this reference.
- 53Showing all polyphenols found in Wine [White]; Phenol-Explorer. http://phenol-explorer.eu/contents/food/136 (accessed 2025-06-04).Google ScholarThere is no corresponding record for this reference.
- 54Showing all polyphenols found in Wine [Red]; Phenol-Explorer. http://phenol-explorer.eu/contents/food/135 (accessed 2025-06-04).Google ScholarThere is no corresponding record for this reference.
- 55Sharma, P.; Kaur, R.; Baskar, C.; Chung, W.-J. Removal of methylene blue from aqueous waste using rice husk and rice husk ash. Desalination. 2010, 259 (1–3), 249– 257, DOI: 10.1016/j.desal.2010.03.044Google ScholarThere is no corresponding record for this reference.
- 56Das, A.; Banerjee, M.; Bar, N.; Das, S. K. Adsorptive removal of Cr(VI) from aqueous solution: kinetic, isotherm, thermodynamics, toxicity, scale-up design, and GA modeling. SN Appl. Sci. 2019, 1 (7), 1– 19, DOI: 10.1007/s42452-019-0813-9Google ScholarThere is no corresponding record for this reference.
- 57Şenol, Z. M.; Messaoudi, N. E.; Fernine, Y.; Keskin, Z. S. Bioremoval of rhodamine B dye from aqueous solution by using agricultural solid waste (almond shell): experimental and DFT modeling studies. Biomass Convers. Biorefin. 2024, 14 (15), 17927– 17940, DOI: 10.1007/s13399-023-03781-1Google ScholarThere is no corresponding record for this reference.
- 58Elovich, S. Yu.; Larionov, O. G. Experimental Verification of the Equation for the Adsorption Isotherm from Solutions. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Scienc. 1962, 2, 198–203. Izv. Akad. Nauk SSSR, Otd. Khim. Nauk. 1961, 2, 216– 222Google ScholarThere is no corresponding record for this reference.
- 59Dias, M.; Pinto, J.; Henriques, B.; Figueira, P.; Fabre, E.; Tavares, D.; Vale, C.; Pereira, E. Nutshells as efficient biosorbents to remove cadmium, lead, and mercury from contaminated solutions. Int. J. Environ. Res. Public Health. 2021, 18 (4), 1580, DOI: 10.3390/ijerph18041580Google ScholarThere is no corresponding record for this reference.
- 60Soto, M. L.; Moure, A.; Domínguez, H.; Parajó, J. C. Recovery, concentration and purification of phenolic compounds by adsorption: A review. J. Food Eng. 2011, 105 (1), 1– 27, DOI: 10.1016/j.jfoodeng.2011.02.010Google ScholarThere is no corresponding record for this reference.
- 61Caldas, T. W.; Mazza, K. E. L.; Teles, A. S. C.; Mattos, G. N.; Brígida, A. I. S.; Conte-Junior, C. A.; Borguini, R. G.; Godoy, R. L. O.; Cabral, L. M. C.; Tonon, R. V. Phenolic compounds recovery from grape skin using conventional and non-conventional extraction methods. Ind. Crops Prod. 2018, 111, 86– 91, DOI: 10.1016/j.indcrop.2017.10.012Google ScholarThere is no corresponding record for this reference.
- 62Mikucka, W.; Zielinska, M.; Bulkowska, K.; Witonska, I. Recovery of polyphenols from distillery stillage by microwave-assisted, ultrasound-assisted and conventional solid-liquid extraction. Sci. Rep. 2022, 12 (1), 3232, DOI: 10.1038/s41598-022-07322-0Google ScholarThere is no corresponding record for this reference.
- 63Cifuentes-Cabezas, M.; Sánchez-Arévalo, C. M.; Mendoza-Roca, J. A.; Vincent-Vela, M. C.; Álvarez-Blanco, S. Recovery of phenolic compounds from olive oil washing wastewater by adsorption/desorption process. Sep. Purif. Technol. 2022, 298 (8), 121562, DOI: 10.2139/ssrn.4103327Google ScholarThere is no corresponding record for this reference.
- 64Lehmann, M. L.; Counce, R. M.; Counce, R. W.; Watson, J. S.; Labbé, N.; Tao, J. Recovery of phenolic compounds from switchgrass extract. ACS Sustain. Chem. Eng. 2018, 6 (1), 374– 379, DOI: 10.1021/acssuschemeng.7b02639Google ScholarThere is no corresponding record for this reference.
- 65Cacace, J. E.; Mazza, G. Mass transfer process during extraction of phenolic compounds from milled berries. J. Food Eng. 2003, 59 (4), 379– 389, DOI: 10.1016/S0260-8774(02)00497-1Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. SEM local imaging of Nonpareil (soft shell) and Peerless (hard shell) almond shells at 0.2–0.5, 0.5–1, and 1–2 mm particle sizes at 600× magnification.
Figure 2

Figure 2. Percent phenolic adsorption onto Nonpareil (soft-shell) and Peerless (hard-shell) ASP exposed to model winery wastewater at three particle size ranges: 0.2–0.5, 0.5–1.0, and 1.0–2.0 mm. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Figure 3
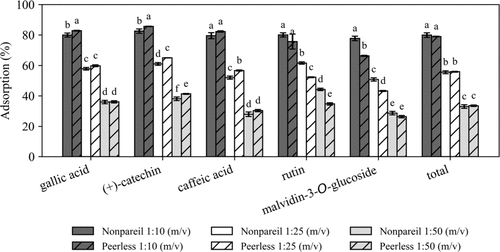
Figure 3. Percent phenolic adsorption for Nonpariel and Peerless ASPs of 0.2–0.5 mm exposed to model winery wastewater at 1:10, 1:25, and 1:50 (m/v) ratio. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Figure 4

Figure 4. Mean total phenolic adsorption of phenolics onto Nonpareil (a) and Peerless ASP (b) and their fit to pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich kinetic models. Bars indicate standard deviation across replicates.
Figure 5
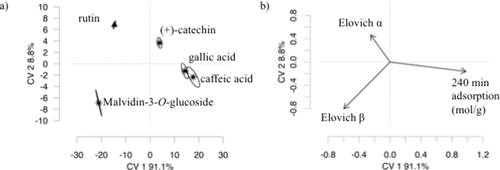
Figure 5. Canonical variate analysis (CVA) of compound adsorption behavior on ASP. CVA showing a) score plot and b)loadings of adsorption parameters and compound characteristics, including α and β rates from the Elovich model, and adsorption at 240 min (mol g–1). Arrows indicate the direction and strength of each variable’s contribution to the canonical variates. CVA scores plot displaying individual compounds projected along the canonical variates.
Figure 6
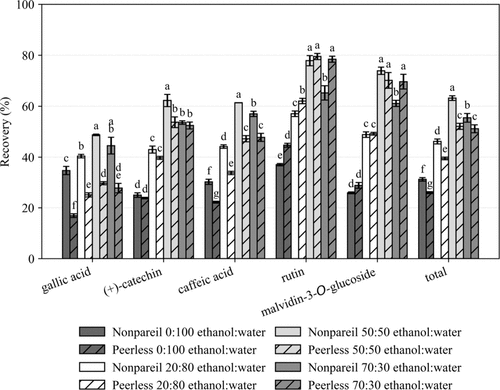
Figure 6. Percent phenolic recovery for Nonpareil and Peerless ASP of 0.2–0.5 mm exposed to ethanol:water solutions at 0:100, 20:80, 50:50, and 70:30 (v/v). Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
Figure 7

Figure 7. Recovery of phenolic compounds from Nonpareil and Peerless ASP using 50:50 ethanol/water (v/v) and varying the ratio of ASP/winery wastewater at 1:10, 1:20, and 1:25 m/v. Different letters indicate statistically significant differences among treatments within each compound (Tukey’s HSD, p < 0.05).
References
This article references 65 other publications.
- 1California & US Wine Production. Wine Institute. https://wineinstitute.org/our-industry/statistics/california-us-wine-production/ (accessed 2024-09-30).There is no corresponding record for this reference.
- 2Jorge, N.; Teixeira, A. R.; Gomes, A.; Peres, J. A.; Lucas, M. S. Winery wastewater: challenges and perspectives. Eng. Proc. 2023, 56 (267). DOI: 10.3390/ASEC2023-15968 .There is no corresponding record for this reference.
- 3Welz, P. J.; Holtman, G.; Haldenwang, R.; Le Roes-Hill, M. Characterisation of winery wastewater from continuous flow settling basins and waste stabilisation ponds over the course of 1 year: Implications for biological wastewater treatment and land application. Water Sci. Technol. 2016, 74 (9), 2036– 2050, DOI: 10.2166/wst.2016.226There is no corresponding record for this reference.
- 4Vlyssides, A. G.; Barampouti, E. M.; Mai, S. Wastewater characteristics from greek wineries and distilleries. Water Sci. Technol. 2005, 51 (1), 53– 60, DOI: 10.2166/wst.2005.0007There is no corresponding record for this reference.
- 5Bories, A.; Sire, Y. Impacts of winemaking methods on wastewaters and their treatment. S. Afr. J. Enol. Vitic. 2016, 31 (1), 38– 44, DOI: 10.21548/31-1-1398There is no corresponding record for this reference.
- 6Fiorentino, A.; Gentili, A.; Isidori, M.; Monaco, P.; Nardelli, A.; Parrella, A.; Temussi, F. Environmental effects caused by olive mill wastewaters: toxicity comparison of low-molecular-eeight phenol components. J. Agric. Food Chem. 2003, 51 (4), 1005– 1009, DOI: 10.1021/jf020887dThere is no corresponding record for this reference.
- 7Stom, D. I.; Roth, R. Some effects of polyphenols on aquatic plants: I. toxicity of phenols in aquatic plants. Bull. Environ. Contam. Toxicol. 1981, 27 (3), 332– 337, DOI: 10.1007/BF01611028There is no corresponding record for this reference.
- 8Siqueira, J. O.; Nair, M. G.; Hammerschmidt, R.; Safir, G. R.; Putnam, A. R. Significance of phenolic compounds in plant-soil-microbial systems. CRC Crit. Rev. Plant Sci. 1991, 10 (1), 63– 121, DOI: 10.1080/07352689109382307There is no corresponding record for this reference.
- 9Cueva, C.; Moreno-Arribas, M. V.; Martín-Alvarez, P. J.; Bills, G.; Vicente, M. F.; Basilio, A.; Rivas, C. L.; Requena, T.; Rodríguez, J. M.; Bartolomé, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161 (5), 372– 382, DOI: 10.1016/j.resmic.2010.04.006There is no corresponding record for this reference.
- 10Lofrano, G.; Meric, S. A comprehensive approach to winery wastewater treatment: A review of the state-of the-art. Desalin. Water Treat. 2016, 57 (7), 3011– 3028, DOI: 10.1080/19443994.2014.982196There is no corresponding record for this reference.
- 11Water Resources Control Board. State Water Resources Control Board Order WQ XXXX-XXXX-DWQ General waste discharge requirements for winery process water public draft ; 2020.There is no corresponding record for this reference.
- 12Mosse, K. P. M.; Patti, A. F.; Christen, E. W.; Cavagnaro, T. R. Review: Winery wastewater quality and treatment options in Australia. Aust. J. Grape Wine Res. 2011, 17 (2), 111– 122, DOI: 10.1111/j.1755-0238.2011.00132.xThere is no corresponding record for this reference.
- 13Mohamad Said, K. A.; Ismail, A. F.; Abdul Karim, Z.; Abdullah, M. S.; Hafeez, A. A review of technologies for the phenolic compounds recovery and phenol removal from wastewater. Process Saf. Environ. Prot. 2021, 151, 257– 289, DOI: 10.1016/j.psep.2021.05.015There is no corresponding record for this reference.
- 14Bories, A.; Guillot, J.-M.; Sire, Y.; Couderc, M.; Lemaire, S.-A.; Kreim, V.; Roux, J.-C. Prevention of volatile fatty acids production and limitation of odours from winery wastewaters by denitrification. Water Res. 2007, 41 (13), 2987– 2995, DOI: 10.1016/j.watres.2007.03.022There is no corresponding record for this reference.
- 15Ngwenya, N.; Gaszynski, C.; Ikumi, D. A review of winery wastewater treatment: A focus on UASB biotechnology optimization and recovery strategies. J. Environ. Chem. Eng. 2022, 10 (4), 108172, DOI: 10.1016/j.jece.2022.108172There is no corresponding record for this reference.
- 16Lahnsteiner, J.; van Rensburg, P.; Esterhuizen, J. Direct potable reuse - a feasible water management option. J. Water Reuse Desalin. 2018, 8 (1), 14– 28, DOI: 10.2166/wrd.2017.172There is no corresponding record for this reference.
- 17Khan, S. J.; Anderson, R. Potable reuse: Experiences in Australia. Curr. Opin. Environ. Sci. Health 2018, 2, 55– 60, DOI: 10.1016/j.coesh.2018.02.002There is no corresponding record for this reference.
- 18California Almond Board. 2023 Almond Almanac. https://www.almonds.com/sites/default/files/2023-12/Almanac_2023.pdf (accessed 2025-05-26).There is no corresponding record for this reference.
- 19Kaur, M.; Kumar, M.; Sachdeva, S.; Puri, S. K. An efficient multiphase bioprocess for enhancing the renewable energy production from almond shells. Energy Convers. Manage. 2020, 203, 112235, DOI: 10.1016/j.enconman.2019.112235There is no corresponding record for this reference.
- 20Prgomet, I.; Gonçalves, B.; Domínguez-Perles, R.; Pascual-Seva, N.; Barros, A. I. R. N. A. Valorization challenges to almond residues: Phytochemical composition and functional application. Molecules. 2017, 22 (10), 1774, DOI: 10.3390/molecules22101774There is no corresponding record for this reference.
- 21Almond Board of California. 2022 Almond Almanac ; 2022. https://www.almonds.org/sites/default/files/2023-04/2022_Almanac.pdf (accessed 2025-10-06).There is no corresponding record for this reference.
- 22Chiou, B.-S.; Valenzuela-Medina, D.; Bilbao-Sainz, C.; Klamczynski, A. P.; Avena-Bustillos, R. J.; Milczarek, R. R.; Du, W.-X.; Glenn, G. M.; Orts, W. J. Torrefaction of almond shells: Effects of torrefaction conditions on properties of solid and condensate products. Ind. Crops Prod. 2016, 86, 40– 48, DOI: 10.1016/j.indcrop.2016.03.030There is no corresponding record for this reference.
- 23Li, X.; Liu, Y.; Hao, J.; Wang, W. Study of almond shell characteristics. Materials. 2018, 11 (9), 1782, DOI: 10.3390/ma11091782There is no corresponding record for this reference.
- 24Tomishima, H.; Luo, K.; Mitchell, A. E. The almond (Prunus dulcis): chemical properties, utilization, and valorization of coproducts. Annu. Rev. Food Sci. Technol. 2022, 13 (1), 145– 166, DOI: 10.1146/annurev-food-052720-111942There is no corresponding record for this reference.
- 25Ledbetter, C. A. Shell cracking strength in almond (Prunus dulcis [Mill.] D.A. Webb.) and its implication in uses as a value-added product. Bioresour. Technol. 2008, 99 (13), 5567– 5573, DOI: 10.1016/j.biortech.2007.10.059There is no corresponding record for this reference.
- 26Zakzeski, J.; Bruijnincx, P. C. A.; Jongerius, A. L.; Weckhuysen, B. M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110 (6), 3552– 3599, DOI: 10.1021/cr900354uThere is no corresponding record for this reference.
- 27Azadi, P.; Inderwildi, O. R.; Farnood, R.; King, D. A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renew. Sustain. Energy Rev. 2013, 21, 506– 523, DOI: 10.1016/j.rser.2012.12.022There is no corresponding record for this reference.
- 28Castro-Muñoz, R.; Yáñez-Fernández, J.; Fíla, V. Phenolic compounds recovered from agro-food by-products using membrane technologies: An overview. Food Chem. 2016, 213, 753– 762, DOI: 10.1016/j.foodchem.2016.07.030There is no corresponding record for this reference.
- 29Soto, M. L.; Moure, A.; Domínguez, H.; Parajó, J. C. Batch and fixed bed column studies on phenolic adsorption from wine vinasses by polymeric resins. J. Food Eng. 2017, 209, 52– 60, DOI: 10.1016/j.jfoodeng.2017.04.008There is no corresponding record for this reference.
- 30Thiel, A.; Tippkötter, N.; Suck, K.; Sohling, U.; Ruf, F.; Ulber, R. New zeolite adsorbents for downstream processing of polyphenols from renewable resources. Eng. Life Sci. 2013, 13 (3), 239– 246, DOI: 10.1002/elsc.201200188There is no corresponding record for this reference.
- 31Kammerer, J.; Kammerer, D. R.; Jensen, U.; Carle, R. Interaction of apple polyphenols in a multi-compound system upon adsorption onto a food-grade resin. J. Food Eng. 2010, 96 (4), 544– 554, DOI: 10.1016/j.jfoodeng.2009.08.038There is no corresponding record for this reference.
- 32Frascari, D.; Rubertelli, G.; Arous, F.; Ragini, A.; Bresciani, L.; Arzu, A.; Pinelli, D. Valorisation of olive mill wastewater by phenolic compounds adsorption: development and application of a procedure for adsorbent selection. Chem. Eng. J. 2019, 360, 124– 138, DOI: 10.1016/j.cej.2018.11.188There is no corresponding record for this reference.
- 33Wang, Z.; Wang, C.; Yuan, J.; Zhang, C. Adsorption characteristics of adsorbent resins and antioxidant capacity for enrichment of phenolics from two-phase olive waste. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2017, 1040, 38– 46, DOI: 10.1016/j.jchromb.2016.11.023There is no corresponding record for this reference.
- 34Ben Arfi, R.; Karoui, S.; Mougin, K.; Ghorbal, A. Adsorptive removal of cationic and anionic dyes from aqueous solution by utilizing almond shell as bioadsorbent. Euro–Mediterr. J. Environ. Integr. 2017, 2 (1). DOI: 10.1007/s41207-017-0032-y .There is no corresponding record for this reference.
- 35Melhaoui, R.; Miyah, Y.; Kodad, S.; Houmy, N.; Addi, M.; Abid, M.; Mihamou, A.; Serghini-Caid, H.; Lairini, S.; Tijani, N.; Hano, C.; Elamrani, A. On the suitability of almond shells for the manufacture of a natural low-cost bioadsorbent to remove brilliant green: Kinetics and equilibrium isotherms study. Sci. World J. 2021, 2021. 1 DOI: 10.1155/2021/6659902 .There is no corresponding record for this reference.
- 36Duran, C.; Ozdes, D.; Gundogdu, A.; Senturk, H. B. Kinetics and isotherm analysis of basic dyes adsorption onto almond shell (Prunus dulcis) as a low cost adsorbent. J. Chem. Eng. Data 2011, 56 (5), 2136– 2147, DOI: 10.1021/je101204jThere is no corresponding record for this reference.
- 37Ziarati, P.; Kermanshah, A.; Moslehishad, M. Adsorption heavy metal from contaminated water by modified shell of wild endemic almonds: Amygdalus lycioides and amygdalus wendelboi. Biosci. Biotechnol. Res. Asia. 2015, 12 (3), 2451– 2457, DOI: 10.13005/bbra/1923There is no corresponding record for this reference.
- 38Mirmohammadmakki, F.; Gharachorloo, M.; Ghavami, M.; Abdossi, V.; Azizinezhad, R. The efficiency of almond shell (Amygdalus communis L.) bio-sorption in reduction of heavy metals (lead, cadmium, arsenic, and nickel) from parsley (Petroselinum crispum). Biomass Convers. Biorefin. 2023, 13 (2), 16149– 16160, DOI: 10.1007/s13399-022-02635-6There is no corresponding record for this reference.
- 39Taha, A. A.; Moustafa, A. H. E.; Abdel-Rahman, H. H.; Abd El-Hameed, M. M. A. Comparative biosorption study of Hg (II) using raw and chemically activated almond shell. Adsorp. Sci. Technol. 2018, 36 (1–2), 521– 548, DOI: 10.1177/0263617417705473There is no corresponding record for this reference.
- 40McCaffrey, Z.; Torres, L. F.; Chiou, B.-S.; Hart-Cooper, W.; McMahan, C.; Orts, W. J. Almond and walnut shell activated carbon for methylene blue adsorption. ACS Sustainable Resour. Manage. 2024, 1 (7), 1421– 1431, DOI: 10.1021/acssusresmgt.4c00080There is no corresponding record for this reference.
- 41Omri, A.; Benzina, M.; Ammar, N. Preparation, modification and industrial application of activated carbon from almond shell. J. Ind. Eng. Chem. 2013, 19 (6), 2092– 2099, DOI: 10.1016/j.jiec.2013.03.025There is no corresponding record for this reference.
- 42Loulidi, I.; Boukhlifi, F.; Ouchabi, M.; Amar, A.; Jabri, M.; Kali, A.; Chraibi, S.; Hadey, C.; Aziz, F. Adsorption of crystal violet onto an agricultural waste residue: Kinetics, isotherm, thermodynamics, and mechanism of adsorption. Sci. World J. 2020, 2020, 5873521, DOI: 10.1155/2020/5873521There is no corresponding record for this reference.
- 43Liao, M.; Kelley, S.; Yao, Y. Generating energy and greenhouse gas inventory data of activated carbon production using machine learning and kinetic based process simulation. ACS Sustain. Chem. Eng. 2020, 8 (2), 1252– 1261, DOI: 10.1021/acssuschemeng.9b06522There is no corresponding record for this reference.
- 44Ardia, D.; Boudt, K.; Carl, P.; Mullen, K. M.; Peterson, B. G. Differential evolution with DEoptim: An application to non-convex portfolio optimization. R J. 2011, 3 (1), 27– 34, DOI: 10.32614/RJ-2011-005There is no corresponding record for this reference.
- 45R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2025. https://www.R-project.org.There is no corresponding record for this reference.
- 46Posit team. RStudio: Integrated Development Environment for R. Posit Software, PBC: Boston, MA, 2025. http://www.posit.co/.There is no corresponding record for this reference.
- 47Pinton, S.; Furlan Goncalves Dias, F.; Lerno, L. A.; Barile, D.; Leite Nobrega de Moura Bell, J. M. Revitalizing unfermented cabernet sauvignon pomace using an eco-friendly, two-stage countercurrent process: role of pH on the extractability of bioactive phenolics. Processes. 2022, 10 (10), 2093, DOI: 10.3390/pr10102093There is no corresponding record for this reference.
- 48Harbertson, J. F.; Mireles, M.; Yu, Y. Improvement of BSA tannin precipitation assay by reformulation of resuspension buffer. Am. J. Enol. Vitic. 2015, 66 (1), 95– 99, DOI: 10.5344/ajev.2014.14082There is no corresponding record for this reference.
- 49Harbertson, J. F.; DeBeer, D.; Waterhouse, A. L.; Adams, D. O. Use of ferric chloride for the measurement of total phenolics in red and white wines. Am. J. Enol. Vitic. 2004, 55, 295AThere is no corresponding record for this reference.
- 50Waterhouse, L.; Wrolstad, R. E. Determination of total phenolics. Current Protocols in Food Analytical Chemistry; John Wiley & Sons: New York, 2002. DOI: 10.1002/0471142913.fai0101s06 .There is no corresponding record for this reference.
- 51Singleton, V. L.; Orthofer, R.; Lamuela-Raventós, R. M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152– 178, DOI: 10.1016/S0076-6879(99)99017-1There is no corresponding record for this reference.
- 52Friendly, M.; Fox, J. Candisc: Visualizing generalized canonical discriminant and canonical correlation analysis. 2024. https://CRAN.R-project.org/package=heplots.There is no corresponding record for this reference.
- 53Showing all polyphenols found in Wine [White]; Phenol-Explorer. http://phenol-explorer.eu/contents/food/136 (accessed 2025-06-04).There is no corresponding record for this reference.
- 54Showing all polyphenols found in Wine [Red]; Phenol-Explorer. http://phenol-explorer.eu/contents/food/135 (accessed 2025-06-04).There is no corresponding record for this reference.
- 55Sharma, P.; Kaur, R.; Baskar, C.; Chung, W.-J. Removal of methylene blue from aqueous waste using rice husk and rice husk ash. Desalination. 2010, 259 (1–3), 249– 257, DOI: 10.1016/j.desal.2010.03.044There is no corresponding record for this reference.
- 56Das, A.; Banerjee, M.; Bar, N.; Das, S. K. Adsorptive removal of Cr(VI) from aqueous solution: kinetic, isotherm, thermodynamics, toxicity, scale-up design, and GA modeling. SN Appl. Sci. 2019, 1 (7), 1– 19, DOI: 10.1007/s42452-019-0813-9There is no corresponding record for this reference.
- 57Şenol, Z. M.; Messaoudi, N. E.; Fernine, Y.; Keskin, Z. S. Bioremoval of rhodamine B dye from aqueous solution by using agricultural solid waste (almond shell): experimental and DFT modeling studies. Biomass Convers. Biorefin. 2024, 14 (15), 17927– 17940, DOI: 10.1007/s13399-023-03781-1There is no corresponding record for this reference.
- 58Elovich, S. Yu.; Larionov, O. G. Experimental Verification of the Equation for the Adsorption Isotherm from Solutions. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Scienc. 1962, 2, 198–203. Izv. Akad. Nauk SSSR, Otd. Khim. Nauk. 1961, 2, 216– 222There is no corresponding record for this reference.
- 59Dias, M.; Pinto, J.; Henriques, B.; Figueira, P.; Fabre, E.; Tavares, D.; Vale, C.; Pereira, E. Nutshells as efficient biosorbents to remove cadmium, lead, and mercury from contaminated solutions. Int. J. Environ. Res. Public Health. 2021, 18 (4), 1580, DOI: 10.3390/ijerph18041580There is no corresponding record for this reference.
- 60Soto, M. L.; Moure, A.; Domínguez, H.; Parajó, J. C. Recovery, concentration and purification of phenolic compounds by adsorption: A review. J. Food Eng. 2011, 105 (1), 1– 27, DOI: 10.1016/j.jfoodeng.2011.02.010There is no corresponding record for this reference.
- 61Caldas, T. W.; Mazza, K. E. L.; Teles, A. S. C.; Mattos, G. N.; Brígida, A. I. S.; Conte-Junior, C. A.; Borguini, R. G.; Godoy, R. L. O.; Cabral, L. M. C.; Tonon, R. V. Phenolic compounds recovery from grape skin using conventional and non-conventional extraction methods. Ind. Crops Prod. 2018, 111, 86– 91, DOI: 10.1016/j.indcrop.2017.10.012There is no corresponding record for this reference.
- 62Mikucka, W.; Zielinska, M.; Bulkowska, K.; Witonska, I. Recovery of polyphenols from distillery stillage by microwave-assisted, ultrasound-assisted and conventional solid-liquid extraction. Sci. Rep. 2022, 12 (1), 3232, DOI: 10.1038/s41598-022-07322-0There is no corresponding record for this reference.
- 63Cifuentes-Cabezas, M.; Sánchez-Arévalo, C. M.; Mendoza-Roca, J. A.; Vincent-Vela, M. C.; Álvarez-Blanco, S. Recovery of phenolic compounds from olive oil washing wastewater by adsorption/desorption process. Sep. Purif. Technol. 2022, 298 (8), 121562, DOI: 10.2139/ssrn.4103327There is no corresponding record for this reference.
- 64Lehmann, M. L.; Counce, R. M.; Counce, R. W.; Watson, J. S.; Labbé, N.; Tao, J. Recovery of phenolic compounds from switchgrass extract. ACS Sustain. Chem. Eng. 2018, 6 (1), 374– 379, DOI: 10.1021/acssuschemeng.7b02639There is no corresponding record for this reference.
- 65Cacace, J. E.; Mazza, G. Mass transfer process during extraction of phenolic compounds from milled berries. J. Food Eng. 2003, 59 (4), 379– 389, DOI: 10.1016/S0260-8774(02)00497-1There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsfoodscitech.5c00652.
Phenolic adsorption and recoveries for Nonpareil and Peerless ASP at various pH values, initial concentrations, and reusability (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



