Fundamental Insights into Crystallization and Microphase Separation of Conjugated Block CopolymersClick to copy article linkArticle link copied!
- Juan Peng*Juan Peng*E-mail: [email protected]State Key Laboratory of Molecular Engineering of Polymers, Department of Macromolecular Science, Fudan University, Shanghai 200438, ChinaMore by Juan Peng
- Yanchun Han*Yanchun Han*E-mail: [email protected]State Key Laboratory of Polymer Physics and Chemistry, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, 5625 Renmin Street, Changchun 130022, ChinaMore by Yanchun Han
Abstract
Conjugated polymer-based block copolymers (BCPs) are important materials as they incorporate the optoelectronic properties of conjugated polymers and unique microphase-separated properties of BCPs. The ability to tailor the two basic phase behaviors (crystallization and microphase separation) within conjugated BCPs is highly desirable to not only control their physical properties, but also strengthen the fundamental understanding of rod-like BCPs. However, the crystallization of semirigid conjugated polymers has not been well elucidated compared to traditional flexible polymers. Moreover, the phase behaviors of conjugated BCPs are much less understood than those of classical coil–coil BCPs from both theoretical and experimental aspects. In this Viewpoint we begin with a brief introduction of chain stiffness and the crystallization of conjugated polymers. After introducing the phase behaviors of BCPs including coil–coil, rod–coil, and rod–rod types, we discuss recent advances in the interplay and competition between crystallization and microphase separation within conjugated BCPs, as well as their applications in organic electronics. Finally, ongoing challenges and future perspectives will be discussed.
This publication is licensed for personal use by The American Chemical Society.
1. Introduction
Figure 1
Figure 1. Schematic of the main content of this Viewpoint.
2. Crystallization of Conjugated Polymers
Chain Stiffness of Conjugated Polymers
Figure 2
Figure 2. (a) Chain conformation for coil-like, semirigid, and rod-like polymers. Chain rigidity increases with an increased ratio of the chain persistence length (lp) to the contour length (Lc). Reproduced with permission from ref (28) and modified. Copyright 2022 American Chemical Society. (b) Depiction of hindered rotation model (ri: backbone displacement vectors, θi: deflection angles, ϕi: dihedral angles). Reproduced with permission from ref (25). Copyright 2014 American Chemical Society. (c) The correlation between the end-to-end distance squared normalized by the persistence length squared (⟨h2⟩/lp2) and the persistence length per chain (L/lp) in the freely rotating worm-like chain model. Reproduced with permission from ref (31). Copyright 2017 Royal Society of Chemistry.
Crystallization of Conjugated Polymers
Figure 3
Figure 3. (a) Diagram of free energy change with increased nucleus radius based on classical nucleation theory (CNT). Reproduced with permission from ref (60) and modified. Copyright 2019 American Chemical Society. (b) Schematic of the P3HT chain conformation from extended to folded with increased molecular weight. Reproduced with permission from ref (66) and modified. Copyright 2009 American Chemical Society. (c, d) Schematic of (c) a 1D crystal prism model and (d) the possible chain packing in a P3HT lamella. Reproduced with permission from ref (70) and modified. Copyright 2010 Elsevier. (e) Schematic of coexisting ordered crystalline regions and disordered amorphous regions in conjugated polymer thin films. Reproduced with permission from ref (12) and modified. Copyright 2013 Nature Publishing Group.
3. Phase Behaviors of Different Types of BCPs
Figure 4
Figure 4. (a) Phase diagram of AB coil–coil BCPs in the melt state, showing the stability regions of different microphase-separated morphologies. Reproduced with permission from ref (86). Copyright 2012 American Chemical Society. (b) Schematic of representative morphologies formed by AB coil–coil BCPs due to microphase separation. (c) Phase diagrams of wormlike AB BCPs with different chain rigidities (L/a = 10, 4, 2, 1) and (d) schematic of morphologies formed in AB BCPs containing semiflexible chains. Reproduced with permission from ref (104). Copyright 2013 American Physical Society.
4. Tailoring Crystallization and Microphase Separation of Conjugated BCPs
Figure 5
Figure 5. Representative chemical structures of conjugated BCPs in this Viewpoint.
Effect of Molecular Weight
Figure 6
Figure 6. (a) TEM images and the schematic of the annealed P3DDT-b-P3DDS thin films with the increased polymerization degree N. Reproduced with permission from ref (17). Copyright 2019 American Chemical Society. (b) 2D-GIWAXS images and (c) corresponding chain packing in drop-cast P3HT-b-PBTTT thin films with various molecular weights. Reproduced with permission from ref (112). Copyright 2024 American Chemical Society.
Effect of Regioregularity
Figure 7
Figure 7. (a) Schematic and TEM images of P3HT-b-P2VP in the solution influenced by the RR of P3HT, demonstrating the morphology transformation from nanowires to micelles with the decreased RR. Reproduced with permission from ref (116). Copyright 2018 American Chemical Society. (b) TEM images of P3HT-b-P2VP with different contents of P3HT and RR values in the solid state. Reproduced with permission from ref (118). Copyright 2017 American Chemical Society. (c) Different crystalline orientations of P3HT-b-P3MEGT with high and low RR of P3HT in the solid state. Reproduced with permission from ref (120). Copyright 2022 American Chemical Society.
Effect of Block Ratio
Figure 8
Figure 8. (a) TEM images of P3HT-b-PEG with the increased P3HT length when produced from an anisole solution. The schematic describes the chain packing influenced by the P3HT length or solvent. Reproduced with permission from ref (121). Copyright 2012 John Wiley and Sons. (b) TEM image of the P3BT-b-P3OT (50:50) in 1,2-dichlorobenzene and WAXD spectra of P3BT-b-P3OT (50:50 and 76:24). Reproduced with permission from ref (122) and modified. Copyright 2009 American Chemical Society. (c) Schematic of PPP-b-P3HT in various block ratios when produced from chlorobenzene and anisole, illustrating the preferential crystallization and microphase separation, respectively. Reproduced with permission from ref (123) and modified. Copyright 2016 Elsevier. (d) TEM images of PPP-b-P3HT (34:66 and 62:38) films annealed at 280 °C for 1 h, showing fibrillar morphology and microphase-separated nanoribbon morphology, respectively. Reproduced with permission from ref (13). Copyright 2012 American Chemical Society. (e) Three different epitaxy crystallization modes achieved in PPP-b-P3HT films during thermal annealing at different temperatures. Reproduced with permission from ref (124) and modified. Copyright 2015 American Chemical Society.
Effect of the Main Backbone
Figure 9
Figure 9. (a) AFM images of P3HT-b-Mal7 and P3HT-b-AcMal7 thin films after thermal annealing and their SAXS profiles in the as-cast state (black line) and after thermal annealing (red line). Reproduced with permission from ref (125). Copyright 2017 American Chemical Society. (b) TEM image of P3HT-b-AcMal7 thin film after annealing at 220 °C, with lamellar structure and the schematic of tilted P3HT chains with respect to the lamellar plane. Reproduced with permission from ref (126). Copyright 2020 American Chemical Society. (c) Schematic of the crystallization and microphase separation in P3HT-b-PFTBT films without (left) and with (right) inserting a certain content of the random copolymer, demonstrating dominated crystallization and microphase separation, respectively. Reproduced with permission from ref (127). Copyright 2018 American Chemical Society. (d) AFM images of P3HT-b-DPP films in the as-cast state and after thermal annealing at different temperatures. Reproduced with permission from ref (129). Copyright 2012 American Chemical Society. (e) Lorentz corrected SAXS images of P3HT-b-PPerAcr with different block ratios (left) and the corresponding TEM images (right), displaying lamellar and cylindrical morphology in bulk. Reproduced with permission from ref (130). Copyright 2013 American Chemical Society.
Effect of Block Sequence
Figure 10
Figure 10. (a) Schematic of the solution aggregation behavior of P3AT-based triblock copolymers dependent on the block sequence. Reproduced with permission from ref (132). Copyright 2018 American Chemical Society. (b) 2D-GIWAXS images of P3BT-b-P3AT-b-P3HS triblock copolymer thin films after two-stage heating process, and the schematic of chain packing in cocrystals and microphase-separated structures represented by P3BT-b-P3OT-b-P3HS. Reproduced with permission from ref (134). Copyright 2022 Elsevier. (c) AFM images and the corresponding schematic of ABA-type, AB-type, and BAB-type (block A: P3HT, block B: POO) in the annealed thin films. Reproduced with permission from ref (135). Copyright 2019 Royal Society of Chemistry.
Effect of Alkyl Side Chain
Figure 11
Figure 11. (a) TEM images of cylindrical microdomains in P3EHT-b-PMA with different fractions of P3EHT by melt pressing at 150 °C, and the corresponding chain packing confined in cylinders. Reproduced with permission from ref (139). Copyright 2017 American Chemical Society. (b) AFM images of P3HT-b-P3EHT (83:17) thin films after thermal annealing and the corresponding schematic of chain packing. Reproduced with permission from ref (141). Copyright 2009 American Chemical Society.
Effect of Side Chain Functionalization
Figure 12
Figure 12. (a) TEM images of P3HT-b-P3PHT (1:1 and 3:1) in anisole. Reproduced with permission from ref (152). Copyright 2016 Royal Society of Chemistry. (b) AFM image and the schematic of P3HT-b-P3HHT helical nanofibers produced from the pyridine solution. Reproduced with permission from ref (150). Copyright 2018 Royal Society of Chemistry. (c) AFM images of P3HT-b-P3THA thin films (78:22 and 60:40) and the schematic of chain packing. Reproduced with permission from ref (149). Copyright 2012 American Chemical Society.
5. Applications in Organic Electronics
Figure 13
Figure 13. (a) Schematic of a photovoltaic device based on the PBDT2T-b-N2200 BCP film as the active layer. Reproduced with permission from ref (161). Copyright 2020 John Wiley and Sons. (b) The PCE based on the P3HT-b-PFTBT active layer as a function of thermal annealing time. Reproduced with permission from ref (162). Copyright 2015 American Chemical Society. (c) Optical microscopy images of P3HT and UV cross-linked P3HT-azide BCP immersed in chlorobenzene, and the P3HT/fullerene blended films containing 0% and 15% P3HT-azide BCP after thermal annealing. Reproduced with permission from ref (164). Copyright 2012 American Chemical Society. (d) The degree of cross-linking, crack onset strains, and degree of crystallinity of P3HHT and P3HT-b-P3HHT BCP as a function of heating time. Reproduced with permission from ref (165). Copyright 2016 American Chemical Society. (e) Schematic of the OFET device based on the P3HT-b-PBA as the active layer and their transfer curves with or without strain. Reproduced with permission from ref (167). Copyright 2017 American Chemical Society. (f) Schematic of the electret free phototransistor memory and working mechanism. Reproduced with permission from ref (159). Copyright 2022 John Wiley and Sons. (g) Schematic of the ammonia sensor based on OFETs of helical nanofibril P3HT-b-PPI. Reproduced with permission from ref (169). Copyright 2018 American Chemical Society.
6. Conclusions
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (22573020, 22173023 and 52433009) and STCSM and Shanghai Pilot Program for Basic Research-Fudan University 21TQ1400100 (22TQ002). The authors acknowledge the support from Shanghai Synchrotron Radiation Facility of China for using the BL14B1 and BL02U2 beamlines.
References
This article references 170 other publications.
- 1Shirakawa, H.; Louis, E. J.; MacDiarmid, A. G.; Chiang, C. K.; Heeger, A. J. Synthesis of Electrically Conducting Organic Polymers: Halogen Derivatives of Polyacetylene, (CH)X. J. Chem. Soc., Chem. Commun. 1977, 578– 580, DOI: 10.1039/c39770000578Google ScholarThere is no corresponding record for this reference.
- 2Sirringhaus, H. 25th Anniversary Article: Organic Field-Effect Transistors: The Path Beyond Amorphous Silicon. Adv. Mater. 2014, 26, 1319– 1335, DOI: 10.1002/adma.201304346Google ScholarThere is no corresponding record for this reference.
- 3Ding, L.; Yu, Z.-D.; Wang, X.-Y.; Yao, Z.-F.; Lu, Y.; Yang, C.-Y.; Wang, J.-Y.; Pei, J. Polymer Semiconductors: Synthesis, Processing, and Applications. Chem. Rev. 2023, 123, 7421– 7497, DOI: 10.1021/acs.chemrev.2c00696Google ScholarThere is no corresponding record for this reference.
- 4Gu, K. C.; Loo, Y.-L. The Polymer Physics of Multiscale Charge Transport in Conjugated Systems. J. Polym. Sci., Part B: Polym. Phys. 2019, 57, 1559– 1571, DOI: 10.1002/polb.24873Google ScholarThere is no corresponding record for this reference.
- 5Podzorov, V.; Menard, E.; Rogers, J. A.; Gershenson, M. E. Hall Effect in the Accumulation Layers on the Surface of Organic Semiconductors. Phys. Rev. Lett. 2005, 95, 226601 DOI: 10.1103/PhysRevLett.95.226601Google ScholarThere is no corresponding record for this reference.
- 6Xiong, H. G.; Lin, Q. J.; Lu, Y.; Zheng, D.; Li, Y. W.; Wang, S.; Xie, W. B.; Li, C. Q.; Zhang, X.; Lin, Y. Z.; Wang, Z.-X.; Shi, Q. Q.; Marks, T. J.; Huang, H. General Room-Temperature Suzuki–Miyaura Polymerization for Organic Electronics. Nat. Mater. 2024, 23, 695– 702, DOI: 10.1038/s41563-023-01794-9Google ScholarThere is no corresponding record for this reference.
- 7Carsten, B.; He, F.; Son, H. J.; Xu, T.; Yu, L. P. Stille Polycondensation for Synthesis of Functional Materials. Chem. Rev. 2011, 111, 1493– 1528, DOI: 10.1021/cr100320wGoogle ScholarThere is no corresponding record for this reference.
- 8Martin, R.; Buchwald, S. L. Palladium-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands. Acc. Chem. Res. 2008, 41, 1461– 1473, DOI: 10.1021/ar800036sGoogle ScholarThere is no corresponding record for this reference.
- 9Mei, J. G.; Diao, Y.; Appleton, A. L.; Fang, L.; Bao, Z. N. Integrated Materials Design of Organic Semiconductors for Field-Effect Transistors. J. Am. Chem. Soc. 2013, 135, 6724– 6746, DOI: 10.1021/ja400881nGoogle ScholarThere is no corresponding record for this reference.
- 10Paterson, A. F.; Singh, S.; Fallon, K. J.; Hodsden, T.; Han, Y.; Schroeder, B. C.; Bronstein, H.; Heeney, M.; McCulloch, I.; Anthopoulos, T. D. Recent Progress in High-Mobility Organic Transistors: A Reality Check. Adv. Mater. 2018, 30, 1801079 DOI: 10.1002/adma.201801079Google ScholarThere is no corresponding record for this reference.
- 11Luo, C.; Kyaw, A. K. K.; Perez, L. A.; Patel, S.; Wang, M.; Grimm, B.; Bazan, G. C.; Kramer, E. J.; Heeger, A. J. General Strategy for Self-Assembly of Highly Oriented Nanocrystalline Semiconducting Polymers with High Mobility. Nano Lett. 2014, 14, 2764– 2771, DOI: 10.1021/nl500758wGoogle ScholarThere is no corresponding record for this reference.
- 12Noriega, R.; Rivnay, J.; Vandewal, K.; Koch, F. P. V.; Stingelin, N.; Smith, P.; Toney, M. F.; Salleo, A. A General Relationship between Disorder, Aggregation and Charge Transport in Conjugated Polymers. Nat. Mater. 2013, 12, 1038– 1044, DOI: 10.1038/nmat3722Google ScholarThere is no corresponding record for this reference.
- 13Yu, X. H.; Yang, H.; Wu, S. P.; Geng, Y. H.; Han, Y. C. Microphase Separation and Crystallization of All-Conjugated Phenylene–Thiophene Diblock Copolymers. Macromolecules 2012, 45, 266– 274, DOI: 10.1021/ma201024zGoogle ScholarThere is no corresponding record for this reference.
- 14Hicks, G. E. J.; Li, S.; Obhi, N. K.; Jarrett-Wilkins, C. N.; Seferos, D. S. Programmable Assembly of π-Conjugated Polymers. Adv. Mater. 2021, 33, 2006287, DOI: 10.1002/adma.202006287Google ScholarThere is no corresponding record for this reference.
- 15Peng, J.; Han, Y. C. Recent Advances in Conjugated Polythiophene-Based Rod–Rod Block Copolymers: From Morphology Control to Optoelectronic Applications. Giant 2020, 4, 100039 DOI: 10.1016/j.giant.2020.100039Google ScholarThere is no corresponding record for this reference.
- 16Lee, Y.; Gomez, E. D. Challenges and Opportunities in the Development of Conjugated Block Copolymers for Photovoltaics. Macromolecules 2015, 48, 7385– 7395, DOI: 10.1021/acs.macromol.5b00112Google ScholarThere is no corresponding record for this reference.
- 17Kynaston, E. L.; Winchell, K. J.; Yee, P. Y.; Manion, J. G.; Hendsbee, A. D.; Li, Y. N.; Huettner, S.; Tolbert, S. H.; Seferos, D. S. Poly(3-alkylthiophene)-block-poly(3-alkylselenophene)s: Conjugated Diblock Co-polymers with Atypical Self-Assembly Behavior. ACS Appl. Mater. Interfaces 2019, 11, 7174– 7183, DOI: 10.1021/acsami.8b18795Google ScholarThere is no corresponding record for this reference.
- 18Jin, S.-M.; Hwang, J. H.; Lee, E. Crystallization-Driven Solution-State Assembly of Conjugated Block Copolymers in Materials Science. Macromolecules 2023, 56, 3474– 3496, DOI: 10.1021/acs.macromol.3c00374Google ScholarThere is no corresponding record for this reference.
- 19Yassar, A.; Miozzo, L.; Gironda, R.; Horowitz, G. Rod–Coil and All-Conjugated Block Copolymers for Photovoltaic Applications. Prog. Polym. Sci. 2013, 38, 791– 844, DOI: 10.1016/j.progpolymsci.2012.10.001Google ScholarThere is no corresponding record for this reference.
- 20Mitchell, V. D.; Jones, D. J. Advances toward the Effective Use of Block Copolymers as Organic Photovoltaic Active Layers. Polym. Chem. 2018, 9, 795– 814, DOI: 10.1039/C7PY01878AGoogle ScholarThere is no corresponding record for this reference.
- 21Bhardwaj, A.; Kaur, J.; Wuest, M.; Wuest, F. In Situ Click Chemistry Generation of Cyclooxygenase-2 Inhibitors. Nat. Commun. 2017, 8, 1– 14, DOI: 10.1038/s41467-016-0009-6Google ScholarThere is no corresponding record for this reference.
- 22Zhu, M. J.; Pan, S.; Wang, Y.; Tang, P.; Qiu, F.; Lin, Z. Q.; Peng, J. Unravelling the Correlation between Charge Mobility and Cocrystallization in Rod–Rod Block Copolymers for High-Performance Field-Effect Transistors. Angew. Chem., Int. Ed. 2018, 57, 8644– 8648, DOI: 10.1002/anie.201804585Google ScholarThere is no corresponding record for this reference.
- 23Zhan, H.; Zheng, H.; Dong, Y. J.; Zhao, Q. Q.; Luo, X. B.; Peng, J. Unraveling the Phase Behavior of All-Conjugated Donor–Acceptor Block Copolymers through Molecular Engineering. Macromolecules 2024, 57, 6379– 6389, DOI: 10.1021/acs.macromol.4c00113Google ScholarThere is no corresponding record for this reference.
- 24Cao, Z. Q.; Leng, M. W.; Cao, Y. R.; Gu, X. D.; Fang, L. How Rigid are Conjugated Non-Ladder and Ladder Polymers?. J. Polym. Sci. 2022, 60, 298– 310, DOI: 10.1002/pol.20210550Google ScholarThere is no corresponding record for this reference.
- 25Zhang, W. L.; Gomez, E. D.; Milner, S. T. Predicting Chain Dimensions of Semiflexible Polymers from Dihedral Potentials. Macromolecules 2014, 47, 6453– 6461, DOI: 10.1021/ma500923rGoogle ScholarThere is no corresponding record for this reference.
- 26Matsen, M. M.; Bates, F. S. Unifying Weak- and Strong-Segregation Block Copolymer Theories. Macromolecules 1996, 29, 1091– 1098, DOI: 10.1021/ma951138iGoogle ScholarThere is no corresponding record for this reference.
- 27Hu, W. B. The Physics of Polymer Chain-Folding. Phys. Rep. 2018, 747, 1– 50, DOI: 10.1016/j.physrep.2018.04.004Google ScholarThere is no corresponding record for this reference.
- 28Danielsen, S. P. O.; Bridges, C. R.; Segalman, R. A. Chain Stiffness of Donor–Acceptor Conjugated Polymers in Solution. Macromolecules 2022, 55, 437– 449, DOI: 10.1021/acs.macromol.1c02229Google ScholarThere is no corresponding record for this reference.
- 29Cao, X. X.; Zhao, K. F.; Chen, L.; Liu, J. G.; Han, Y. C. Conjugated Polymer Single Crystals and Nanowires. Polym. Crystallization 2019, 2, 10064– 10081, DOI: 10.1002/pcr2.10064Google ScholarThere is no corresponding record for this reference.
- 30Xiao, M. F.; Carey, R. L.; Chen, H.; Jiao, X. C.; Lemaur, V.; Schott, S.; Nikolka, M.; Jellett, C.; Sadhanala, A.; Rogers, S.; Senanayak, S. P.; Onwubiko, A.; Han, S. Y.; Zhang, Z. L.; Abdi-Jalebi, M.; Zhang, Y. C.; Thomas, T. H.; Mahmoudi, N.; Lai, L. L.; Selezneva, E.; Ren, X. L.; Nguyen, M.; Wang, Q. J.; Jacobs, I.; Yue, W.; McNeill, C. R.; Liu, G. M.; Beljonne, D.; McCulloch, I.; Sirringhaus, H. Charge Transport Physics of a Unique Class of Rigid-Rod Conjugated Polymers with Fused-Ring Conjugated Units Linked by Double Carbon-Carbon Bonds. Sci. Adv. 2021, 7, 5280– 5292, DOI: 10.1126/sciadv.abe5280Google ScholarThere is no corresponding record for this reference.
- 31Kuei, B.; Gomez, E. D. Chain Conformations and Phase Behavior of Conjugated Polymers. Soft Matter 2017, 13, 49– 67, DOI: 10.1039/C6SM00979DGoogle ScholarThere is no corresponding record for this reference.
- 32Hu, Y. B.; Cao, X. X.; Fan, H. Crystallization of D-A Conjugated Polymers: A Review of Recent Research. Polymers 2022, 14, 4612– 4640, DOI: 10.3390/polym14214612Google ScholarThere is no corresponding record for this reference.
- 33Dai, X. B.; Wan, H.-X.; Zhang, X. Y.; Wei, W. J.; Chen, W. L.; Zhang, L. G.; Li, J.; Yan, L.-T. Role of Conformational Entropy in Complex Macromolecular Systems. Chem. Res. Chin. Univ. 2023, 39, 709– 718, DOI: 10.1007/s40242-023-3174-2Google ScholarThere is no corresponding record for this reference.
- 34McCulloch, B.; Ho, V.; Hoarfrost, M.; Stanley, C.; Do, C.; Heller, W. T.; Segalman, R. A. Polymer Chain Shape of Poly(3-alkylthiophenes) in Solution Using Small-Angle Neutron Scattering. Macromolecules 2013, 46, 1899– 1907, DOI: 10.1021/ma302463dGoogle ScholarThere is no corresponding record for this reference.
- 35Podgornik, R. Book Review: Polymer Physics. M. Rubenshtein and R. H. Colby, Oxford University Press, 2003. J. Stat. Phys. 2004, 115, 1757– 1761, DOI: 10.1023/B:JOSS.0000028242.25527.8bGoogle ScholarThere is no corresponding record for this reference.
- 36Gettinger, C. L.; Heeger, A. J.; Drake, J. M.; Pine, D. J. A Photoluminescence Study of Poly(phenylene vinylene) Derivatives: The Effect of Intrinsic Persistence Length. J. Chem. Phys. 1994, 101, 1673– 1678, DOI: 10.1063/1.468438Google ScholarThere is no corresponding record for this reference.
- 37Gettinger, C. L.; Heeger, A. J.; Drake, J. M.; Pine, D. J. The Effect of Intrinsic Rigidity on the Optical Properties of PPV Derivatives. Molecular Crystals and Liquid Crystals Science and Technology. Section A. Mol. Cryst. Liq. Cryst. 1994, 256, 507– 512, DOI: 10.1080/10587259408039283Google ScholarThere is no corresponding record for this reference.
- 38Fytas, G.; Nothofer, H. G.; Scherf, U.; Vlassopoulos, D.; Meier, G. Structure and Dynamics of Nondilute Polyfluorene Solutions. Macromolecules 2002, 35, 481– 488, DOI: 10.1021/ma011416fGoogle ScholarThere is no corresponding record for this reference.
- 39Meier, H.; Stalmach, U.; Kolshorn, H. Effective Conjugation Length and UV/vis Spectra of Oligomers. Acta Polym. 1997, 48, 379– 384, DOI: 10.1002/actp.1997.010480905Google ScholarThere is no corresponding record for this reference.
- 40Socci, E. P.; Farmer, B. L.; Adams, W. W. Molecular Dynamics Simulations of a Poly(p-phenylene). Oligomer. J. Polym. Sci. Part B: Polym. Phys. 1993, 31, 1975– 1982, DOI: 10.1002/polb.1993.090311309Google ScholarThere is no corresponding record for this reference.
- 41Petekidis, G.; Vlassopoulos, D.; Galda, P.; Rehahn, M.; Ballauff, M. Determination of Chain Conformation of Stiff Polymers by Depolarized Rayleigh Scattering in Solution. Macromolecules 1996, 29, 8948– 8953, DOI: 10.1021/ma961128xGoogle ScholarThere is no corresponding record for this reference.
- 42Liu, C.; Hu, W. X.; Jiang, H.; Liu, G.; Han, C. C.; Sirringhaus, H.; Boué, F.; Wang, D. Chain Conformation and Aggregation Structure Formation of a High Charge Mobility DPP-Based Donor–Acceptor Conjugated Polymer. Macromolecules 2020, 53, 8255– 8266, DOI: 10.1021/acs.macromol.0c01646Google ScholarThere is no corresponding record for this reference.
- 43Deng, J. Y.; Zheng, L.; Ding, C. M.; Guo, Y. F.; Xie, Y. F.; Wang, J. W.; Ke, Y. B.; Li, M. M.; Li, L.; Janssen, R. A. J. Determinant Role of Solution-State Supramolecular Assembly in Molecular Orientation of Conjugated Polymer Films. Adv. Funct. Mater. 2023, 33, 2209195– 2209205, DOI: 10.1002/adfm.202209195Google ScholarThere is no corresponding record for this reference.
- 44Gao, M. Y.; Wu, J. J.; Yuan, X. Y.; He, C. Y.; Jiang, H. Q.; Zhao, W. C.; Duan, C. H.; Chen, Y.; Ke, Y. B.; Yao, H. F.; Ye, L. Tuning the Solution Aggregation and Molecular Order for Efficient and Thermally Stable Polymer Solar Cells. Energy Environ. Sci. 2023, 16, 5822– 5831, DOI: 10.1039/D3EE02354KGoogle ScholarThere is no corresponding record for this reference.
- 45Bi, Z. Z.; Naveed, H. B.; Mao, Y. M.; Yan, H. P.; Ma, W. Importance of Nucleation during Morphology Evolution of the Blade-Cast PffBT4T-2OD-Based Organic Solar Cells. Macromolecules 2018, 51, 6682– 6691, DOI: 10.1021/acs.macromol.8b01156Google ScholarThere is no corresponding record for this reference.
- 46Wang, Z. L.; Gao, M. Y.; He, C. Y.; Shi, W. C.; Deng, Y. F.; Han, Y.; Ye, L.; Geng, Y. H. Unraveling the Molar Mass Dependence of Shearing-Induced Aggregation Structure of a High-Mobility Polymer Semiconductor. Adv. Mater. 2022, 34, 2108255 DOI: 10.1002/adma.202108255Google ScholarThere is no corresponding record for this reference.
- 47Tan, W. L.; Tang, L.; Matsidik, R.; Bryant, G.; Martin, T. B.; Sommer, M.; Huang, D. M.; McNeill, C. R. Small-Angle Neutron Scattering of P(NDI2OD-T2) Solutions: Importance of Network Structure for Data Interpretation and Film Morphology. Macromolecules 2024, 57, 691– 706, DOI: 10.1021/acs.macromol.3c01435Google ScholarThere is no corresponding record for this reference.
- 48Zhao, L. H.; Png, R. Q.; Zhuo, J. M.; Wong, L. Y.; Tang, J. C.; Su, Y. S.; Chua, L. L. Role of Borderline Solvents to Induce Pronounced Extended-Chain Lamellar Order in π-Stackable Polymers. Macromolecules 2011, 44, 9692– 9702, DOI: 10.1021/ma201165yGoogle ScholarThere is no corresponding record for this reference.
- 49Heffner, G. W.; Pearson, D. S. Molecular Characterization of Poly(3-hexylthiophene). Macromolecules 1991, 24, 6295– 6299, DOI: 10.1021/ma00023a035Google ScholarThere is no corresponding record for this reference.
- 50Cao, Z. Q.; Tolba, S. A.; Li, Z. F.; Mason, G. T.; Wang, Y.; Do, C. W.; Rondeau-Gagné, S.; Xia, W. J.; Gu, X. D. Molecular Structure and Conformational Design of Donor-Acceptor Conjugated Polymers to Enable Predictable Optoelectronic Property. Adv. Mater. 2023, 35, 2302178– 2302188, DOI: 10.1002/adma.202302178Google ScholarThere is no corresponding record for this reference.
- 51Cao, Z.; Ma, G.; Leng, M.; Zhang, S.; Chen, J.; Do, C.; Hong, K.; Fang, L.; Gu, X. Variable-Temperature Scattering and Spectroscopy Characterizations for Temperature-Dependent Solution Assembly of PffBT4T-Based Conjugated Polymers. ACS Appl. Polym. Mater. 2022, 4, 3023– 3033, DOI: 10.1021/acsapm.1c01511Google ScholarThere is no corresponding record for this reference.
- 52Xu, Y. C.; Ding, L.; Yao, Z. F.; Shao, Y.; Wang, J. Y.; Zhang, W. B.; Pei, J. Conjugated Polymers in Solution: A Physical Perspective. J. Phys. Chem. Lett. 2023, 14, 927– 939, DOI: 10.1021/acs.jpclett.2c03600Google ScholarThere is no corresponding record for this reference.
- 53Lotz, B.; Miyoshi, T.; Cheng, S. Z. D. 50th Anniversary Perspective: Polymer Crystals and Crystallization: Personal Journeys in a Challenging Research Field. Macromolecules 2017, 50, 5995– 6025, DOI: 10.1021/acs.macromol.7b00907Google ScholarThere is no corresponding record for this reference.
- 54Park, K. S.; Kwok, J. J.; Kafle, P.; Diao, Y. When Assembly Meets Processing: Tuning Multiscale Morphology of Printed Conjugated Polymers for Controlled Charge Transport. Chem. Mater. 2021, 33, 469– 498, DOI: 10.1021/acs.chemmater.0c04152Google ScholarThere is no corresponding record for this reference.
- 55Qu, T. F.; Nan, G. M.; Ouyang, Y.; Bieketuerxun, B.; Yan, X. L.; Qi, Y. P.; Zhang, Y. Structure–Property Relationship, Glass Transition, and Crystallization Behaviors of Conjugated Polymers. Polymers 2023, 15, 4268– 4295, DOI: 10.3390/polym15214268Google ScholarThere is no corresponding record for this reference.
- 56Luo, Y. Q.; Santos, F. A.; Wagner, T. W.; Tsoi, E.; Zhang, S. J. Dynamic Interactions Between Poly(3-hexylthiophene) and Single-Walled Carbon Nanotubes in Marginal Solvent. J. Phys. Chem. B 2014, 118, 6038– 6046, DOI: 10.1021/jp503128vGoogle ScholarThere is no corresponding record for this reference.
- 57Li, J. J.; Deepak, F. L. In Situ Kinetic Observations on Crystal Nucleation and Growth. Chem. Rev. 2022, 122, 16911– 16982, DOI: 10.1021/acs.chemrev.1c01067Google ScholarThere is no corresponding record for this reference.
- 58Xu, J.; Reiter, G.; Alamo, R. G. Concepts of Nucleation in Polymer Crystallization. Crystals 2021, 11, 304– 322, DOI: 10.3390/cryst11030304Google ScholarThere is no corresponding record for this reference.
- 59Karthika, S.; Radhakrishnan, T. K.; Kalaichelvi, P. A Review of Classical and Nonclassical Nucleation Theories. Cryst. Growth Des. 2016, 16, 6663– 6681, DOI: 10.1021/acs.cgd.6b00794Google ScholarThere is no corresponding record for this reference.
- 60Tang, X. L.; Chen, W.; Li, L. B. The Tough Journey of Polymer Crystallization: Battling with Chain Flexibility and Connectivity. Macromolecules 2019, 52, 3575– 3591, DOI: 10.1021/acs.macromol.8b02725Google ScholarThere is no corresponding record for this reference.
- 61Turnbull, D.; Fisher, J. C. Rate of Nucleation in Condensed Systems. J. Chem. Phys. 1949, 17, 71– 73, DOI: 10.1063/1.1747055Google ScholarThere is no corresponding record for this reference.
- 62Dingler, C.; Dirnberger, K.; Ludwigs, S. Semiconducting Polymer Spherulites–From Fundamentals to Polymer Electronics. Macromol. Rapid Commun. 2019, 40, 1800601 DOI: 10.1002/marc.201800601Google ScholarThere is no corresponding record for this reference.
- 63Patel, B. B.; Diao, Y. Multiscale Assembly of Solution-Processed Organic Electronics: The Critical Roles of Confinement, Fluid Flow, and Interfaces. Nanotechnology 2018, 29, 044004 DOI: 10.1088/1361-6528/aa9d7cGoogle ScholarThere is no corresponding record for this reference.
- 64Malik, S.; Nandi, A. K. Crystallization Mechanism of Regioregular Poly(3-alkyl thiophene)s. J. Polym. Sci., Part B: Polym. Phys. 2002, 40, 2073– 2085, DOI: 10.1002/polb.10272Google ScholarThere is no corresponding record for this reference.
- 65Mena-Osteritz, E.; Meyer, A.; Langeveld-Voss, B. M. W.; Janssen, R. A. J.; Meijer, E. W.; Bäuerle, P. Two-Dimensional Crystals of Poly(3-Alkyl-thiophene)s: Direct Visualization of Polymer Folds in Submolecular Resolution. Angew. Chem., Int. Ed. 2000, 39, 2679– 2684, DOI: 10.1002/1521-3773(20000804)39:15<2679::AID-ANIE2679>3.0.CO;2-2Google ScholarThere is no corresponding record for this reference.
- 66Liu, J. H.; Arif, M.; Zou, J. H.; Khondaker, S. I.; Zhai, L. Controlling Poly(3-hexylthiophene) Crystal Dimension: Nanowhiskers and Nanoribbons. Macromolecules 2009, 42, 9390– 9393, DOI: 10.1021/ma901955cGoogle ScholarThere is no corresponding record for this reference.
- 67Brun, M.; Demadrille, R.; Rannou, P.; Pron, A.; Travers, J.-P.; Grévin, B. Multiscale Scanning Tunneling Microscopy Study of Self-Assembly Phenomena in Two-Dimensional Polycrystals of π-Conjugated Polymers: The Case of Regioregular Poly(dioctylbithiophene-alt-fluorenone). Adv. Mater. 2004, 16, 2087– 2092, DOI: 10.1002/adma.200400088Google ScholarThere is no corresponding record for this reference.
- 68Yi, S.; Zheng, H.; Peng, J. Synergistic Backbone and Side Chain Engineering on Crystalline Orientation and Charge Transport of Conjugated Polymers in Field-effect Transistors. Polymer 2025, 327, 128391 DOI: 10.1016/j.polymer.2025.128391Google ScholarThere is no corresponding record for this reference.
- 69Yu, Z. D.; Lu, Y.; Yao, Z. F.; Wu, H. T.; Wang, Z. Y.; Pan, C. K.; Wang, J. Y.; Pei, J. Buffer Chain Model for Understanding Crystallization Competition in Conjugated Polymers. Angew. Chem., Int. Ed. 2024, 63, e202405139 DOI: 10.1002/anie.202405139Google ScholarThere is no corresponding record for this reference.
- 70Lim, J. A.; Liu, F.; Ferdous, S.; Muthukumar, M.; Briseno, A. L. Polymer Semiconductor Crystals. Mater. Today 2010, 13, 14– 24, DOI: 10.1016/S1369-7021(10)70080-8Google ScholarThere is no corresponding record for this reference.
- 71Qian, Z. Y.; Cao, Z. Q.; Galuska, L.; Zhang, S.; Xu, J.; Gu, X. D. Glass Transition Phenomenon for Conjugated Polymers. Macromol. Chem. Phys. 2019, 220, 1900062 DOI: 10.1002/macp.201900062Google ScholarThere is no corresponding record for this reference.
- 72Samitsu, S.; Shimomura, T.; Heike, S.; Hashizume, T.; Ito, K. Effective Production of Poly(3-alkylthiophene) Nanofibers by Means of Whisker Method Using Anisole Solvent: Structural, Optical, and Electrical Properties. Macromolecules 2008, 41, 8000– 8010, DOI: 10.1021/ma801128vGoogle ScholarThere is no corresponding record for this reference.
- 73Chu, P. H.; Kleinhenz, N.; Persson, N.; McBride, M.; Hernandez, J. L.; Fu, B. Y.; Zhang, G. Y.; Reichmanis, E. Toward Precision Control of Nanofiber Orientation in Conjugated Polymer Thin Films: Impact on Charge Transport. Chem. Mater. 2016, 28, 9099– 9109, DOI: 10.1021/acs.chemmater.6b04202Google ScholarThere is no corresponding record for this reference.
- 74Cao, X. X.; Du, Z. H.; Chen, L.; Zhao, K. F.; Li, H. X.; Liu, J. G.; Han, Y. C. Long Diketopyrrolopyrrole-Based Polymer Nanowires Prepared by Decreasing the Aggregate Speed of the Polymer in Solution. Polymer 2017, 118, 135– 142, DOI: 10.1016/j.polymer.2017.04.076Google ScholarThere is no corresponding record for this reference.
- 75Um, H. A.; Lee, D. H.; Heo, D. U.; Yang, D. S.; Shin, J.; Baik, H.; Cho, M. J.; Choi, D. H. High Aspect Ratio Conjugated Polymer Nanowires for High Performance Field-Effect Transistors and Phototransistors. ACS Nano 2015, 9, 5264– 5274, DOI: 10.1021/acsnano.5b01982Google ScholarThere is no corresponding record for this reference.
- 76Cao, X.; Fan, H. Formation of D-A Conjugated Polymer Crystals: Diffusion and Conformational Transition Theory. Polymer 2022, 243, 124606 DOI: 10.1016/j.polymer.2022.124606Google ScholarThere is no corresponding record for this reference.
- 77Yao, Z. F.; Wang, J. Y.; Pei, J. Controlling Morphology and Microstructure of Conjugated Polymers via Solution-State Aggregation. Prog. Polym. Sci. 2023, 136, 101626 DOI: 10.1016/j.progpolymsci.2022.101626Google ScholarThere is no corresponding record for this reference.
- 78Luo, X. B.; Zhao, Q. Q.; Li, L.; Zheng, H.; Guo, Y. N.; Peng, J. Concentration Effect on the Solution-state Aggregation and Solid-state Crystalline Structures of Poly(3-butylthiophene). Acta Polym. Sin. 2024, 55, 910– 920, DOI: 10.11777/j.issn1000-3304.2023.23302Google ScholarThere is no corresponding record for this reference.
- 79Lan, Y. K.; Huang, C. I. Charge Mobility and Transport Behavior in the Ordered and Disordered States of the Regioregular Poly(3-hexylthiophene). J. Phys. Chem. B 2009, 113, 14555– 14564, DOI: 10.1021/jp904841jGoogle ScholarThere is no corresponding record for this reference.
- 80Liang, Q. J.; Duan, M. Z.; Geng, Z. Q.; Zhang, M.; Xu, W. Z.; Geng, H. J.; He, Z. M.; Liu, J. G. Regulation of Molecular Orientation in Organic Solar Cells. Chem. Eng. J. 2024, 488, 150783 DOI: 10.1016/j.cej.2024.150783Google ScholarThere is no corresponding record for this reference.
- 81Zhu, S.; Peng, J. Meniscus-Assisted Solution Printing Enables Cocrystallization in Poly(3-alkylthiophene)-based Blends for Field-Effect Transistors. Chin. J. Polym. Sci. 2023, 41, 1269– 1276, DOI: 10.1007/s10118-023-2916-4Google ScholarThere is no corresponding record for this reference.
- 82Qian, Z. Y.; Galuska, L. A.; Ma, G. R.; McNutt, W. W.; Zhang, S.; Mei, J. G.; Gu, X. D. Backbone Flexibility on Conjugated Polymer’s Crystallization Behavior and Thin Film Mechanical Stability. J. Polym. Sci. 2022, 60, 548– 558, DOI: 10.1002/pol.20210462Google ScholarThere is no corresponding record for this reference.
- 83Yu, L.; Davidson, E.; Sharma, A.; Andersson, M. R.; Segalman, R.; Müller, C. Isothermal Crystallization Kinetics and Time-Temperature-Transformation of the Conjugated Polymer: Poly(3-(2′-ethyl)hexylthiophene). Chem. Mater. 2017, 29, 5654– 5662, DOI: 10.1021/acs.chemmater.7b01393Google ScholarThere is no corresponding record for this reference.
- 84Qian, Z. Y.; Luo, S. C.; Qu, T. F.; Galuska, L. A.; Zhang, S.; Cao, Z. Q.; Dhakal, S.; He, Y. J.; Hong, K. L.; Zhou, D. S.; Gu, X. D. Influence of Side-Chain Isomerization on the Isothermal Crystallization Kinetics of Poly(3-alkylthiophenes). J. Mater. Res. 2021, 36, 191– 202, DOI: 10.1557/s43578-020-00062-9Google ScholarThere is no corresponding record for this reference.
- 85Palacios, J. K.; Michell, R. M.; Müller, A. J. Crystallization, Morphology and Self-Assembly of Double, Triple and Tetra Crystalline Block Polymers. Polym. Test. 2023, 121, 107995– 108026, DOI: 10.1016/j.polymertesting.2023.107995Google ScholarThere is no corresponding record for this reference.
- 86Matsen, M. W. Effect of Architecture on the Phase Behavior of AB-Type Block Copolymer Melts. Macromolecules 2012, 45, 2161– 2165, DOI: 10.1021/ma202782sGoogle ScholarThere is no corresponding record for this reference.
- 87Ji, S. X.; Wan, L.; Liu, C.-C.; Nealey, P. F. Directed Self-Assembly of Block Copolymers on Chemical Patterns: A Platform for Nanofabrication. Prog. Polym. Sci. 2016, 54–55, 76– 127, DOI: 10.1016/j.progpolymsci.2015.10.006Google ScholarThere is no corresponding record for this reference.
- 88Gao, Y.; Deng, H. L.; Li, W. H.; Qiu, F.; Shi, A. C. Formation of Nonclassical Ordered Phases of AB-Type Multiarm Block Copolymers. Phys. Rev. Lett. 2016, 116, 068304 DOI: 10.1103/PhysRevLett.116.068304Google ScholarThere is no corresponding record for this reference.
- 89Bates, F. S.; Fredrickson, G. H. Block Copolymer Thermodynamics: Theory and Experiment. Annu. Rev. Phys. Chem. 1990, 41, 525– 557, DOI: 10.1146/annurev.pc.41.100190.002521Google ScholarThere is no corresponding record for this reference.
- 90Kim, J. K.; Yang, S. Y.; Lee, Y.; Kim, Y. Functional Nanomaterials Based on Block Copolymer Self-Assembly. Prog. Polym. Sci. 2010, 35, 1325– 1349, DOI: 10.1016/j.progpolymsci.2010.06.002Google ScholarThere is no corresponding record for this reference.
- 91Suh, H. S.; Kim, D. H.; Moni, P.; Xiong, S. S.; Ocola, L. E.; Zaluzec, N. J.; Gleason, K. K.; Nealey, P. F. Sub-10-nm Patterning via Directed Self-Assembly of Block Copolymer Films with a Vapour-Phase Deposited Topcoat. Nat. Nanotechnol. 2017, 12, 575– 583, DOI: 10.1038/nnano.2017.34Google ScholarThere is no corresponding record for this reference.
- 92Hu, X.-H.; Xiong, S. S. Fabrication of Nanodevices Through Block Copolymer Self-Assembly. Front. Nanotechnol. 2022, 4, 762996 DOI: 10.3389/fnano.2022.762996Google ScholarThere is no corresponding record for this reference.
- 93Sakurai, S.; Mori, K.; Okawara, A.; Kimishima, K.; Hashimoto, T. Evaluation of Segmental Interaction by Small-Angle X-ray Scattering Based on the Random-Phase Approximation for Asymmetric, Polydisperse Triblock Copolymers. Macromolecules 1992, 25, 2679– 2691, DOI: 10.1021/ma00036a019Google ScholarThere is no corresponding record for this reference.
- 94Feldman, K. E.; Kade, M. J.; Meijer, E. W.; Hawker, C. J.; Kramer, E. J. Phase Behavior of Complementary Multiply Hydrogen Bonded End-Functional Polymer Blends. Macromolecules 2010, 43, 5121– 5127, DOI: 10.1021/ma1003776Google ScholarThere is no corresponding record for this reference.
- 95Scheiwiller, S. C.; Mata, J. P.; Pozzo, L. D. Morphology of Poly-3-Hexyl-Thiophene Blends with Styrene-Isoprene-Styrene Block-Copolymer Elastomers from X-ray and Neutron Scattering. Soft Matter 2024, 20, 6247– 6265, DOI: 10.1039/D4SM00495GGoogle ScholarThere is no corresponding record for this reference.
- 96Olsen, B. D.; Shah, M.; Ganesan, V.; Segalman, R. A. Universalization of the Phase Diagram for a Model Rod-Coil Diblock Copolymer. Macromolecules 2008, 41, 6809– 6817, DOI: 10.1021/ma800978cGoogle ScholarThere is no corresponding record for this reference.
- 97Kim, W. N.; Burns, C. M. Blends of Polycarbonate and Poly(methyl methacrylate) and the Determination of the Polymer-Polymer Interaction Parameter of the Two Polymers. Macromolecules 1987, 20, 1876– 1882, DOI: 10.1021/ma00174a030Google ScholarThere is no corresponding record for this reference.
- 98Kipp, D.; Mok, J.; Strzalka, J.; Darling, S. B.; Ganesan, V.; Verduzco, R. Rational Design of Thermally Stable, Bicontinuous Donor/Acceptor Morphologies with Conjugated Block Copolymer Additives. ACS Macro Lett. 2015, 4, 867– 871, DOI: 10.1021/acsmacrolett.5b00413Google ScholarThere is no corresponding record for this reference.
- 99Leibler, L. Theory of Microphase Separation in Block Copolymers. Macromolecules 1980, 13, 1602– 1617, DOI: 10.1021/ma60078a047Google ScholarThere is no corresponding record for this reference.
- 100Porter, J. D. The Solubility of Nonelectrolytes. 3rd ed. Joel H. Hildebrand and Robert L. Scott. New York: Reinhold, 1950.. Science 1951, 113, 450– 451, DOI: 10.1126/science.113.2938.450-bGoogle ScholarThere is no corresponding record for this reference.
- 101Miquelard-Garnier, G.; Roland, S. Beware of the Flory Parameter to Characterize Polymer-Polymer Interactions: A Critical Reexamination of the Experimental Literature. Eur. Polym. J. 2016, 84, 111– 124, DOI: 10.1016/j.eurpolymj.2016.09.009Google ScholarThere is no corresponding record for this reference.
- 102Liu, C. L.; Lin, C. H.; Kuo, C. C.; Lin, S. T.; Chen, W. C. Conjugated Rod–Coil Block Copolymers: Synthesis, Morphology, Photophysical Properties, and Stimuli-Responsive Applications. Prog. Polym. Sci. 2011, 36, 603– 637, DOI: 10.1016/j.progpolymsci.2010.07.008Google ScholarThere is no corresponding record for this reference.
- 103Park, H.; Ma, B. S.; Kim, J. S.; Kim, Y.; Kim, H. J.; Kim, D.; Yun, H.; Han, J.; Kim, F. S.; Kim, T.-S.; Kim, B. J. Regioregular-block-Regiorandom Poly(3-hexylthiophene) Copolymers for Mechanically Robust and High-Performance Thin-Film Transistors. Macromolecules 2019, 52, 7721– 7730, DOI: 10.1021/acs.macromol.9b01540Google ScholarThere is no corresponding record for this reference.
- 104Jiang, Y.; Chen, J. Z. Y. Influence of Chain Rigidity on the Phase Behavior of Wormlike Diblock Copolymers. Phys. Rev. Lett. 2013, 110, 138305 DOI: 10.1103/PhysRevLett.110.138305Google ScholarThere is no corresponding record for this reference.
- 105He, W. N.; Xu, J. T. Crystallization Assisted Self-Assembly of Semicrystalline Block Copolymers. Prog. Polym. Sci. 2012, 37, 1350– 1400, DOI: 10.1016/j.progpolymsci.2012.05.002Google ScholarThere is no corresponding record for this reference.
- 106Loo, Y.-L.; Register, R. A.; Ryan, A. J. Modes of Crystallization in Block Copolymer Microdomains: Breakout, Templated, and Confined. Macromolecules 2002, 35, 2365– 2374, DOI: 10.1021/ma011824jGoogle ScholarThere is no corresponding record for this reference.
- 107Xiao, L. L.; Zhou, X.; Yue, K.; Guo, Z. H. Synthesis and Self-Assembly of Conjugated Block Copolymers. Polymers 2021, 13, 110– 129, DOI: 10.3390/polym13010110Google ScholarThere is no corresponding record for this reference.
- 108Zhang, L.; Li, H. X.; Zhao, K. F.; Zhang, T.; Liu, D.; Wang, S. C.; Wu, F.; Zhang, Q.; Han, Y. C. Achieving the High Charge Mobility of Conjugated Polymers under Cyclic Stretching by Changing the Interaction Parameter between Solvent and Sidechain. Polymer 2023, 275, 125912 DOI: 10.1016/j.polymer.2023.125912Google ScholarThere is no corresponding record for this reference.
- 109Chen, L.; Zhao, K. F.; Cao, X. X.; Liu, J. G.; Yu, X. H.; Han, Y. H. Nanowires of Conjugated Polymer Prepared by Tuning the Interaction between the Solvent and Polymer. Polymer 2018, 149, 23– 29, DOI: 10.1016/j.polymer.2018.06.068Google ScholarThere is no corresponding record for this reference.
- 110Kynaston, E. L.; Fang, Y.; Manion, J. G.; Obhi, N. K.; Howe, J. Y.; Perepichka, D. F.; Seferos, D. S. Patchy Nanofibers from the Thin Film Self-Assembly of A Conjugated Diblock Copolymer. Angew. Chem., Int. Ed. 2017, 56, 6152– 6156, DOI: 10.1002/anie.201700134Google ScholarThere is no corresponding record for this reference.
- 111Chen, S. W.; Zheng, H.; Liu, X. F.; Peng, J. Tailoring Co-crystallization over Microphase Separation in Conjugated Block Copolymers via Rational Film Processing for Field-Effect Transistors. Macromolecules 2022, 55, 10405– 10414, DOI: 10.1021/acs.macromol.2c02048Google ScholarThere is no corresponding record for this reference.
- 112Zhao, Q. Q.; Zheng, H.; Li, D. K.; Peng, J. Interrogating Cocrystallization and Microphase Segregation in Meticulously Engineered Rod–Rod Poly(thieno)thiophene-Based Block Copolymers for Organic Field-Effect Transistors. Macromolecules 2024, 57, 4782– 4792, DOI: 10.1021/acs.macromol.4c00632Google ScholarThere is no corresponding record for this reference.
- 113Kline, R. J.; McGehee, M. D.; Kadnikova, E. N.; Liu, J. S.; Fréchet, J. M. J. Controlling the Field-Effect Mobility of Regioregular Polythiophene by Changing the Molecular Weight. Adv. Mater. 2003, 15, 1519– 1522, DOI: 10.1002/adma.200305275Google ScholarThere is no corresponding record for this reference.
- 114Chen, S. W.; Li, L. X.; Zhai, D. L.; Yin, Y.; Shang, X.; Ni, B. J.; Peng, J. Cocrystallization-Promoted Charge Mobility in All-Conjugated Diblock Copolymers for High-Performance Field-Effect Transistors. ACS Appl. Mater. Interfaces 2020, 12, 58094– 58104, DOI: 10.1021/acsami.0c17671Google ScholarThere is no corresponding record for this reference.
- 115Ge, J.; He, M.; Qiu, F.; Yang, Y. L. Synthesis, Cocrystallization, and Microphase Separation of All-Conjugated Diblock Copoly(3-alkylthiophene)s. Macromolecules 2010, 43, 6422– 6428, DOI: 10.1021/ma1010167Google ScholarThere is no corresponding record for this reference.
- 116Kim, Y.; Kim, H. J.; Kim, J.-S.; Yun, H.; Park, H.; Han, J.; Kim, B. J. Modulating Regioregularity of Poly(3-hexylthiophene)-based Amphiphilic Block Copolymers To Control Solution Assembly from Nanowires to Micelles. Chem. Mater. 2018, 30, 7912– 7921, DOI: 10.1021/acs.chemmater.8b03510Google ScholarThere is no corresponding record for this reference.
- 117Kim, Y.-J.; Cho, C.-H.; Paek, K.; Jo, M.; Park, M.-k.; Lee, N.-E.; Kim, Y.-j.; Kim, B. J.; Lee, E. Precise Control of Quantum Dot Location within the P3HT-b-P2VP/QD Nanowires Formed by Crystallization-Driven 1D Growth of Hybrid Dimeric Seeds. J. Am. Chem. Soc. 2014, 136, 2767– 2774, DOI: 10.1021/ja410165fGoogle ScholarThere is no corresponding record for this reference.
- 118Kim, J. S.; Kim, Y.; Kim, H. J.; Kim, H. J.; Yang, H.; Jung, Y. S.; Stein, G. E.; Kim, B. J. Regioregularity-Driven Morphological Transition of Poly(3-hexylthiophene)-Based Block Copolymers. Macromolecules 2017, 50, 1902– 1908, DOI: 10.1021/acs.macromol.7b00128Google ScholarThere is no corresponding record for this reference.
- 119Coote, J. P.; Kim, J. S.; Lee, B.; Han, J.; Kim, B. J.; Stein, G. E. Crystallization Modes of Poly(3-dodecylthiophene)-Based Block Copolymers Depend on Regioregularity and Morphology. Macromolecules 2018, 51, 9276– 9283, DOI: 10.1021/acs.macromol.8b01985Google ScholarThere is no corresponding record for this reference.
- 120Kim, P.; Kim, E. Y.; Han, S. G.; Lee, J.; Kang, S.; Park, C.; Cho, K.; Moon, H. C.; Kim, J. K. Enhanced Vertical Hole Mobility Through End-on Chain Orientation of Poly(3-hexylthiophene)-Based Diblock Copolymers by Microphase Separation. Macromolecules 2022, 55, 6160– 6166, DOI: 10.1021/acs.macromol.2c00791Google ScholarThere is no corresponding record for this reference.
- 121Yang, H.; Xia, H.; Wang, G. W.; Peng, J.; Qiu, F. Insights into Poly(3-hexylthiophene)-b-Poly(ethylene oxide) Block Copolymer: Synthesis and Solvent-Induced Structure Formation in Thin Films. J. Polym. Sci., Polym. Chem. 2012, 50, 5060– 5067, DOI: 10.1002/pola.26353Google ScholarThere is no corresponding record for this reference.
- 122Wu, P. T.; Ren, G. Q.; Li, C. X.; Mezzenga, R.; Jenekhe, S. A. Crystalline Diblock Conjugated Copolymers: Synthesis, Self-Assembly, and Microphase Separation of Poly(3-butylthiophene)-b-poly(3-octylthiophene). Macromolecules 2009, 42, 2317– 2320, DOI: 10.1021/ma802874vGoogle ScholarThere is no corresponding record for this reference.
- 123Yang, H.; Zhang, R.; Wang, L.; Zhang, J. D.; Yu, X. H.; Geng, Y. H.; Han, Y. C. Crystallization Assisted Microphase Separation in All-Conjugated Phenylene-Thiophene Diblock Copolymers. Polymer 2016, 97, 238– 246, DOI: 10.1016/j.polymer.2016.05.037Google ScholarThere is no corresponding record for this reference.
- 124Yang, H.; Zhang, R.; Wang, L.; Zhang, J. D.; Yu, X. H.; Liu, J. G.; Xing, R. B.; Geng, Y. H.; Han, Y. C. Face-On and Edge-On Orientation Transition and Self-Epitaxial Crystallization of All-Conjugated Diblock Copolymer. Macromolecules 2015, 48, 7557– 7566, DOI: 10.1021/acs.macromol.5b01804Google ScholarThere is no corresponding record for this reference.
- 125Sakai-Otsuka, Y.; Zaioncz, S.; Otsuka, I.; Halila, S.; Rannou, P.; Borsali, R. Self-Assembly of Carbohydrate-block-Poly(3-hexylthiophene) Diblock Copolymers into Sub-10 nm Scale Lamellar Structures. Macromolecules 2017, 50, 3365– 3376, DOI: 10.1021/acs.macromol.7b00118Google ScholarThere is no corresponding record for this reference.
- 126Sakai-Otsuka, Y.; Nishiyama, Y.; Putaux, J. L.; Brinkmann, M.; Satoh, T.; Chen, W.-C.; Borsali, R. Competing Molecular Packing of Blocks in a Lamella-Forming Carbohydrate-block-poly(3-hexylthiophene) Copolymer. Macromolecules 2020, 53, 9054– 9064, DOI: 10.1021/acs.macromol.0c01801Google ScholarThere is no corresponding record for this reference.
- 127Lee, Y.; Aplan, M. P.; Seibers, Z. D.; Xie, R. X.; Culp, T. E.; Wang, C.; Hexemer, A.; Kilbey, S. M.; Wang, Q.; Gomez, E. D. Random Copolymers Allow Control of Crystallization and Microphase Separation in Fully Conjugated Block Copolymers. Macromolecules 2018, 51, 8844– 8852, DOI: 10.1021/acs.macromol.8b01859Google ScholarThere is no corresponding record for this reference.
- 128Brady, M. A.; Ku, S. Y.; Perez, L. A.; Cochran, J. E.; Schmidt, K.; Weiss, T. M.; Toney, M. F.; Ade, H.; Hexemer, A.; Wang, C.; Hawker, C. J.; Kramer, E. J.; Chabinyc, M. L. Role of Solution Structure in Self-Assembly of Conjugated Block Copolymer Thin Films. Macromolecules 2016, 49, 8187– 8197, DOI: 10.1021/acs.macromol.6b01686Google ScholarThere is no corresponding record for this reference.
- 129Ku, S. Y.; Brady, M. A.; Treat, N. D.; Cochran, J. E.; Robb, M. J.; Kramer, E. J.; Chabinyc, M. L.; Hawker, C. J. A Modular Strategy for Fully Conjugated Donor-Acceptor Block Copolymers. J. Am. Chem. Soc. 2012, 134, 16040– 16046, DOI: 10.1021/ja307431kGoogle ScholarThere is no corresponding record for this reference.
- 130Lohwasser, R. H.; Gupta, G.; Kohn, P.; Sommer, M.; Lang, A. S.; Thurn-Albrecht, T.; Thelakkat, M. Phase Separation in the Melt and Confined Crystallization as the Key to Well-Ordered Microphase Separated Donor–Acceptor Block Copolymers. Macromolecules 2013, 46, 4403– 4410, DOI: 10.1021/ma3021147Google ScholarThere is no corresponding record for this reference.
- 131Verswyvel, M.; Steverlynck, J.; Hadj Mohamed, S.; Trabelsi, M.; Champagne, B.; Koeckelberghs, G. All-Conjugated ABC-block-copolymer Formation with a Varying Sequence via an Unassociated Catalyst. Macromolecules 2014, 47, 4668– 4675, DOI: 10.1021/ma500610pGoogle ScholarThere is no corresponding record for this reference.
- 132Verheyen, L.; Timmermans, B.; Koeckelberghs, G. Influence of the Sequence in Conjugated Triblock Copolymers on Their Aggregation Behavior. Macromolecules 2018, 51, 6421– 6429, DOI: 10.1021/acs.macromol.8b01302Google ScholarThere is no corresponding record for this reference.
- 133Zhu, S. Y.; Li, D. K.; Zhan, H.; Chen, S. W.; Peng, J. Synergistic Manipulation of Phase Transition between Cocrystallization and Microphase Segregation in Conjugated Triblock Copolymers for Organic Field-Effect Transistors. Macromolecules 2024, 57, 2067– 2077, DOI: 10.1021/acs.macromol.3c02275Google ScholarThere is no corresponding record for this reference.
- 134Li, L. X.; Zhao, Q. Q.; Chen, S. W.; Lin, Z. Q.; Peng, J. Unfolding the Cocrystallization–Charge Transport Correlation in All-Conjugated Triblock Copolymers via Meticulous Molecular Engineering for Organic Field-Effect Transistors. Nano Energy 2022, 100, 107489– 107497, DOI: 10.1016/j.nanoen.2022.107489Google ScholarThere is no corresponding record for this reference.
- 135Chiang, Y. C.; Kobayashi, S.; Isono, T.; Shih, C. C.; Shingu, T.; Hung, C.-C.; Hsieh, H.-C.; Tung, S.-H.; Satoh, T.; Chen, W.-C. Effect of A Conjugated/Elastic Block Sequence on the Morphology and Electronic Properties of Polythiophene Based Stretchable Block Copolymers. Polym. Chem. 2019, 10, 5452– 5464, DOI: 10.1039/C9PY01216HGoogle ScholarThere is no corresponding record for this reference.
- 136Zhang, Y.; Tajima, K.; Hirota, K.; Hashimoto, K. Synthesis of All-Conjugated Diblock Copolymers by Quasi-Living Polymerization and Observation of Their Microphase Separation. J. Am. Chem. Soc. 2008, 130, 7812– 7813, DOI: 10.1021/ja8023516Google ScholarThere is no corresponding record for this reference.
- 137Ho, V.; Boudouris, B. W.; McCulloch, B. L.; Shuttle, C. G.; Burkhardt, M.; Chabinyc, M. L.; Segalman, R. A. Poly(3-alkylthiophene) Diblock Copolymers with Ordered Microstructures and Continuous Semiconducting Pathways. J. Am. Chem. Soc. 2011, 133, 9270– 9273, DOI: 10.1021/ja2035317Google ScholarThere is no corresponding record for this reference.
- 138Lee, I. H.; Amaladass, P.; Yoon, K. Y.; Shin, S.; Kim, Y.-J.; Kim, I.; Lee, E.; Choi, T.-L. Nanostar and Nanonetwork Crystals Fabricated by In Situ Nanoparticlization of Fully Conjugated Polythiophene Diblock Copolymers. J. Am. Chem. Soc. 2013, 135, 17695– 17698, DOI: 10.1021/ja409206pGoogle ScholarThere is no corresponding record for this reference.
- 139Davidson, E. C.; Segalman, R. A. Confined Crystallization within Cylindrical P3EHT Block Copolymer Microdomains. Macromolecules 2017, 50, 6128– 6136, DOI: 10.1021/acs.macromol.7b01323Google ScholarThere is no corresponding record for this reference.
- 140Davidson, E. C.; Segalman, R. A. Thermal Control of Confined Crystallization within P3EHT Block Copolymer Microdomains. Macromolecules 2017, 50, 8097– 8105, DOI: 10.1021/acs.macromol.7b01616Google ScholarThere is no corresponding record for this reference.
- 141Zhang, Y.; Tajima, K.; Hashimoto, K. Nanostructure Formation in Poly(3-hexylthiophene-block-3-(2-ethylhexyl)thiophene)s. Macromolecules 2009, 42, 7008– 7015, DOI: 10.1021/ma9013065Google ScholarThere is no corresponding record for this reference.
- 142Lee, Y. H.; Chen, W. C.; Yang, Y. L.; Chiang, C. J.; Yokozawa, T.; Dai, C.-A. Co-Crystallization Phase Transformations in All Pi-Conjugated Block Copolymers With Different Main-Chain Moieties. Nanoscale 2014, 6, 5208– 5216, DOI: 10.1039/c3nr06037cGoogle ScholarThere is no corresponding record for this reference.
- 143Moon, H. C.; Bae, D.; Kim, J. K. Self-Assembly of Poly(3-dodecylthiophene)-block-Poly(methyl methacrylate) Copolymers Driven by Competition between Microphase Separation and Crystallization. Macromolecules 2012, 45, 5201– 5207, DOI: 10.1021/ma300902nGoogle ScholarThere is no corresponding record for this reference.
- 144Lin, S. H.; Wu, S. J.; Ho, C. C.; Su, W. F. Rational Design of Versatile Self-Assembly Morphology of Rod-Coil Block Copolymer. Macromolecules 2013, 46, 2725– 2732, DOI: 10.1021/ma302220bGoogle ScholarThere is no corresponding record for this reference.
- 145Boudouris, B. W.; Frisbie, C. D.; Hillmyer, M. A. Nanoporous Poly(3-alkylthiophene) Thin Films Generated from Block Copolymer Templates. Macromolecules 2008, 41, 67– 75, DOI: 10.1021/ma071626dGoogle ScholarThere is no corresponding record for this reference.
- 146Chen, Y.; Cui, H. J.; Li, L. S.; Tian, Z. Y.; Tang, Z. Y. Controlling Micro-Phase Separation in Semi-Crystalline/Amorphous Conjugated Block Copolymers. Polym. Chem. 2014, 5, 4400– 4404, DOI: 10.1039/C4PY00498AGoogle ScholarThere is no corresponding record for this reference.
- 147Li, W.; He, Y. G.; Shi, S. Y.; Liu, N.; Zhu, Y. Y.; Ding, Y. S.; Yin, J.; Wu, Z. Q. Fabrication of A Multi-Charge Generable Poly(phenyl Isocyanide)-block-Poly(3-hexylthiophene) Rod–Rod Conjugated Copolymer. Polym. Chem. 2015, 6, 2348– 2355, DOI: 10.1039/C4PY01624FGoogle ScholarThere is no corresponding record for this reference.
- 148Kim, H. J.; Skinner, M.; Yu, H.; Oh, J. H.; Briseno, A. L.; Emrick, T.; Kim, B. J.; Hayward, R. C. Water Processable Polythiophene Nanowires by Photo-Cross-Linking and Click-Functionalization. Nano Lett. 2015, 15, 5689– 5695, DOI: 10.1021/acs.nanolett.5b01185Google ScholarThere is no corresponding record for this reference.
- 149Ho, C. C.; Liu, Y. C.; Lin, S. H.; Su, W. F. Synthesis, Morphology, and Optical and Electrochemical Properties of Poly(3-hexylthiophene)-b-poly(3-thiophene hexylacetate). Macromolecules 2012, 45, 813– 820, DOI: 10.1021/ma202164dGoogle ScholarThere is no corresponding record for this reference.
- 150Cui, H. N.; Chen, X. S.; Wang, Y.; Wei, D. C.; Qiu, F.; Peng, J. Hydrogen-Bonding-Directed Helical Nanofibers in A Polythiophene-Based All-Conjugated Diblock Copolymer. Soft Matter 2018, 14, 5906– 5912, DOI: 10.1039/C8SM01130CGoogle ScholarThere is no corresponding record for this reference.
- 151Shi, Y. Q.; Tan, L. C.; Chen, L.; Chen, Y. W. In Situ Fabricating One-Dimensional Donor–Acceptor Core-Shell Hybrid Nanobeams Network Driven by Self-Assembly of Diblock Copolythiophenes. Macromolecules 2014, 47, 1757– 1767, DOI: 10.1021/ma402154gGoogle ScholarThere is no corresponding record for this reference.
- 152Zhu, M. J.; Kim, H.; Jang, Y. J.; Park, S.; Ryu, D. Y.; Kim, K.; Tang, P.; Qiu, F.; Kim, D. H.; Peng, J. Toward High Efficiency Organic Photovoltaic Devices with Enhanced Thermal Stability Utilizing P3HT-b-P3PHT Block Copolymer Additives. J. Mater. Chem. A 2016, 4, 18432– 18443, DOI: 10.1039/C6TA08181AGoogle ScholarThere is no corresponding record for this reference.
- 153Xu, L.; Xu, X.-H.; Liu, N.; Zou, H.; Wu, Z.-Q. A Facile Synthetic Route to Multifunctional Poly(3-hexylthiophene)-b-poly(phenyl isocyanide) Copolymers: From Aggregation-Induced Emission to Controlled Helicity. Macromolecules 2018, 51, 7546– 7555, DOI: 10.1021/acs.macromol.8b01478Google ScholarThere is no corresponding record for this reference.
- 154Timmermans, B.; Koeckelberghs, G. Chiral expression of co-crystallizing poly(thiophene)-block-poly(selenophene) copolymers. Polym. Chem. 2020, 11, 2715– 2723, DOI: 10.1039/C9PY01775EGoogle ScholarThere is no corresponding record for this reference.
- 155Lombeck, F.; Komber, H.; Sepe, A.; Friend, R. H.; Sommer, M. Enhancing Phase Separation and Photovoltaic Performance of All-Conjugated Donor–Acceptor Block Copolymers with Semifluorinated Alkyl Side Chains. Macromolecules 2015, 48, 7851– 7860, DOI: 10.1021/acs.macromol.5b01845Google ScholarThere is no corresponding record for this reference.
- 156Mitchell, V. D.; Gann, E.; Huettner, S.; Singh, C. R.; Subbiah, J.; Thomsen, L.; McNeill, C. R.; Thelakkat, M.; Jones, D. J. Morphological and Device Evaluation of an Amphiphilic Block Copolymer for Organic Photovoltaic Applications. Macromolecules 2017, 50, 4942– 4951, DOI: 10.1021/acs.macromol.7b00377Google ScholarThere is no corresponding record for this reference.
- 157Gao, D.; Hollinger, J.; Seferos, D. S. Selenophene-Thiophene Block Copolymer Solar Cells with Thermostable Nanostructures. ACS Nano 2012, 6, 7114– 7121, DOI: 10.1021/nn3021844Google ScholarThere is no corresponding record for this reference.
- 158Yin, Y.; Zhai, D.; Chen, S.; Shang, X.; Li, L.; Peng, J. Controlling the Condensed Structure of Polythiophene and Polyselenophene-based All-conjugated Block Copolymers. Acta Polym. Sin. 2020, 51, 434– 447Google ScholarThere is no corresponding record for this reference.
- 159Yang, W. C.; Chen, Y. W.; Yu, Y. Y.; Lin, Y. C.; Higashihara, T.; Chen, W. C. Enhancing the Performance of Electret-Free Phototransistor Memory by Using All-Conjugated Block Copolymer. Macromol. Rapid Commun. 2023, 44, 2200756 DOI: 10.1002/marc.202200756Google ScholarThere is no corresponding record for this reference.
- 160Jung, Y. S.; Jung, W.; Tuller, H. L.; Ross, C. A. Nanowire Conductive Polymer Gas Sensor Patterned Using Self-Assembled Block Copolymer Lithography. Nano Lett. 2008, 8, 3776– 3780, DOI: 10.1021/nl802099kGoogle ScholarThere is no corresponding record for this reference.
- 161Park, S. H.; Kim, Y.; Kwon, N. Y.; Lee, Y. W.; Woo, H. Y.; Chae, W. S.; Park, S.; Cho, M. J.; Choi, D. H. Significantly Improved Morphology and Efficiency of Nonhalogenated Solvent-Processed Solar Cells Derived from a Conjugated Donor-Acceptor Block Copolymer. Adv. Sci. 2020, 7, 1902470 DOI: 10.1002/advs.201902470Google ScholarThere is no corresponding record for this reference.
- 162Smith, K. A.; Lin, Y.-H.; Mok, J. W.; Yager, K. G.; Strzalka, J.; Nie, W.; Mohite, A. D.; Verduzco, R. Molecular Origin of Photovoltaic Performance in Donor-block-Acceptor All-Conjugated Block Copolymers. Macromolecules 2015, 48, 8346– 8353, DOI: 10.1021/acs.macromol.5b01383Google ScholarThere is no corresponding record for this reference.
- 163Park, S. H.; Kwon, N. Y.; Jung, S. H.; Harit, A. K.; Woo, H. Y.; Cho, M. J.; Choi, D. H. Enhanced Efficiency and Stability of Novel Pseudo-ternary Polymer Solar Cells Enabled by a Conjugated Donor Block Copolymer. ACS Appl. Mater. Interfaces. 2023, 15, 20266– 20277, DOI: 10.1021/acsami.3c00815Google ScholarThere is no corresponding record for this reference.
- 164Kim, H. J.; Han, A.-R.; Cho, C.-H.; Kang, H.; Cho, H.-H.; Lee, M. Y.; Fréchet, J. M. J.; Oh, J. H.; Kim, B. J. Solvent-Resistant Organic Transistors and Thermally Stable Organic Photovoltaics Based on Cross-linkable Conjugated Polymers. Chem. Mater. 2012, 24, 215– 221, DOI: 10.1021/cm203058pGoogle ScholarThere is no corresponding record for this reference.
- 165Yang, X. B.; Ge, J.; He, M.; Ye, Z.; Liu, X. F.; Peng, J.; Qiu, F. Crystallization and Microphase Morphology of Side-Chain Cross-Linkable Poly(3-hexylthiophene)-block-poly[3-(6-hydroxy) hexylthiophene] Diblock Copolymers. Macromolecules 2016, 49, 287– 297, DOI: 10.1021/acs.macromol.5b02001Google ScholarThere is no corresponding record for this reference.
- 166Li, X. Y.; Wolanin, P. J.; MacFarlane, L. R.; Harniman, R. L.; Qian, J. S.; Gould, O. E. C.; Dane, T. G.; Rudin, J.; Cryan, M. J.; Schmaltz, T.; Frauenrath, H.; Winnik, M. A.; Faul, C. F. J.; Manners, I. Uniform Electroactive Fibre-Like Micelle Nanowires for Organic Electronics. Nat. Commun. 2017, 8, 15909– 15917, DOI: 10.1038/ncomms15909Google ScholarThere is no corresponding record for this reference.
- 167Wang, J.-T.; Takshima, S.; Wu, H.-C.; Shih, C.-C.; Isono, T.; Kakuchi, T.; Satoh, T.; Chen, W.-C. Stretchable Conjugated Rod–Coil Poly(3-hexylthiophene)-block-poly(butyl acrylate) Thin Films for Field Effect Transistor Applications. Macromolecules 2017, 50, 1442– 1452, DOI: 10.1021/acs.macromol.6b02722Google ScholarThere is no corresponding record for this reference.
- 168Xu, L.; Wang, C.; Li, Y. X.; Xu, X. H.; Zhou, L.; Liu, N.; Wu, Z. Q. Crystallization-Driven Asymmetric Helical Assembly of Conjugated Block Copolymers and the Aggregation Induced White-light Emission and Circularly Polarized Luminescence. Angew. Chem., Int. Ed. 2020, 59, 16675– 16682, DOI: 10.1002/anie.202006561Google ScholarThere is no corresponding record for this reference.
- 169Wei, S.; Tian, F.; Ge, F.; Wang, X. H.; Zhang, G. B.; Lu, H. B.; Yin, J.; Wu, Z. Q.; Qiu, L. Z. Helical Nanofibrils of Block Copolymer for High-Performance Ammonia Sensors. ACS Appl. Mater. Interfaces 2018, 10, 22504– 22512, DOI: 10.1021/acsami.8b06458Google ScholarThere is no corresponding record for this reference.
- 170Pei, J. Multi-level Aggregation of Conjugated Polymers in Solution: Advances and Challenges. Chin. J. Polym. Sci. 2023, 41, 1498– 1500, DOI: 10.1007/s10118-023-2938-yGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
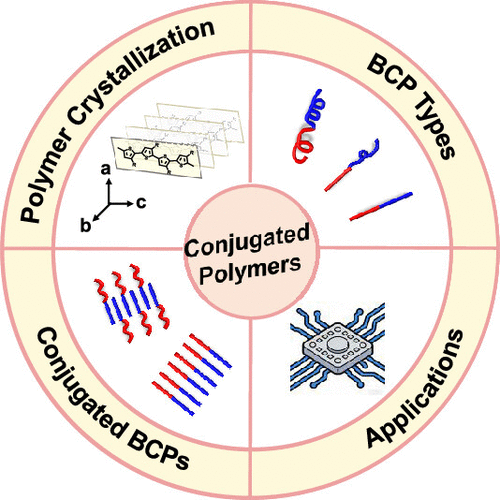
Figure 1

Figure 1. Schematic of the main content of this Viewpoint.
Figure 2
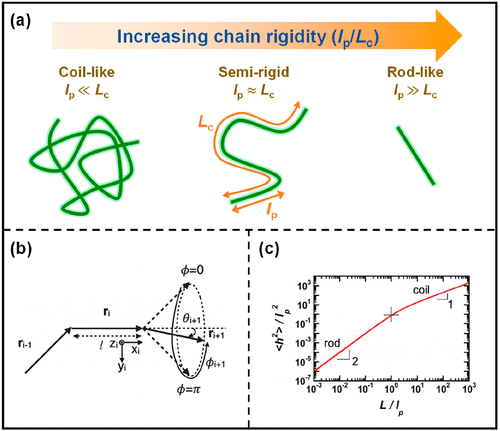
Figure 2. (a) Chain conformation for coil-like, semirigid, and rod-like polymers. Chain rigidity increases with an increased ratio of the chain persistence length (lp) to the contour length (Lc). Reproduced with permission from ref (28) and modified. Copyright 2022 American Chemical Society. (b) Depiction of hindered rotation model (ri: backbone displacement vectors, θi: deflection angles, ϕi: dihedral angles). Reproduced with permission from ref (25). Copyright 2014 American Chemical Society. (c) The correlation between the end-to-end distance squared normalized by the persistence length squared (⟨h2⟩/lp2) and the persistence length per chain (L/lp) in the freely rotating worm-like chain model. Reproduced with permission from ref (31). Copyright 2017 Royal Society of Chemistry.
Figure 3
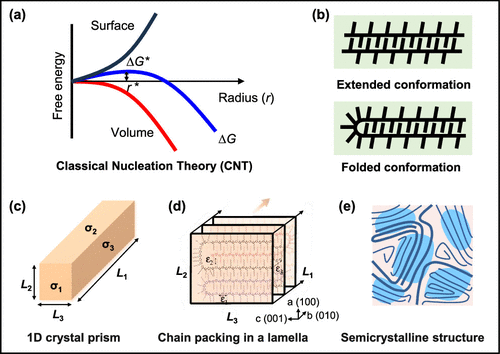
Figure 3. (a) Diagram of free energy change with increased nucleus radius based on classical nucleation theory (CNT). Reproduced with permission from ref (60) and modified. Copyright 2019 American Chemical Society. (b) Schematic of the P3HT chain conformation from extended to folded with increased molecular weight. Reproduced with permission from ref (66) and modified. Copyright 2009 American Chemical Society. (c, d) Schematic of (c) a 1D crystal prism model and (d) the possible chain packing in a P3HT lamella. Reproduced with permission from ref (70) and modified. Copyright 2010 Elsevier. (e) Schematic of coexisting ordered crystalline regions and disordered amorphous regions in conjugated polymer thin films. Reproduced with permission from ref (12) and modified. Copyright 2013 Nature Publishing Group.
Figure 4

Figure 4. (a) Phase diagram of AB coil–coil BCPs in the melt state, showing the stability regions of different microphase-separated morphologies. Reproduced with permission from ref (86). Copyright 2012 American Chemical Society. (b) Schematic of representative morphologies formed by AB coil–coil BCPs due to microphase separation. (c) Phase diagrams of wormlike AB BCPs with different chain rigidities (L/a = 10, 4, 2, 1) and (d) schematic of morphologies formed in AB BCPs containing semiflexible chains. Reproduced with permission from ref (104). Copyright 2013 American Physical Society.
Figure 5
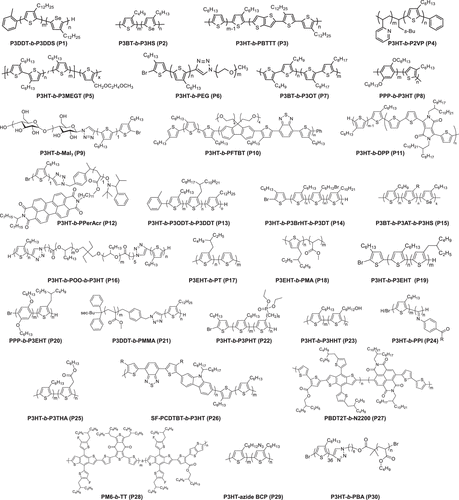
Figure 5. Representative chemical structures of conjugated BCPs in this Viewpoint.
Figure 6
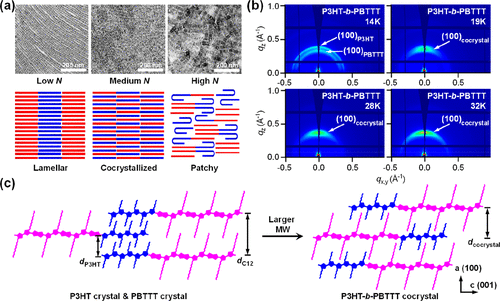
Figure 6. (a) TEM images and the schematic of the annealed P3DDT-b-P3DDS thin films with the increased polymerization degree N. Reproduced with permission from ref (17). Copyright 2019 American Chemical Society. (b) 2D-GIWAXS images and (c) corresponding chain packing in drop-cast P3HT-b-PBTTT thin films with various molecular weights. Reproduced with permission from ref (112). Copyright 2024 American Chemical Society.
Figure 7
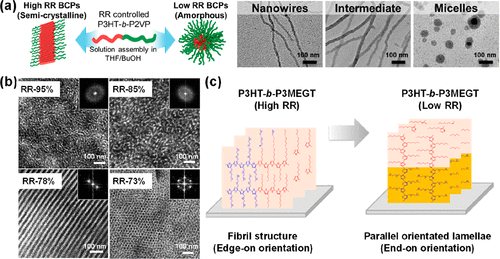
Figure 7. (a) Schematic and TEM images of P3HT-b-P2VP in the solution influenced by the RR of P3HT, demonstrating the morphology transformation from nanowires to micelles with the decreased RR. Reproduced with permission from ref (116). Copyright 2018 American Chemical Society. (b) TEM images of P3HT-b-P2VP with different contents of P3HT and RR values in the solid state. Reproduced with permission from ref (118). Copyright 2017 American Chemical Society. (c) Different crystalline orientations of P3HT-b-P3MEGT with high and low RR of P3HT in the solid state. Reproduced with permission from ref (120). Copyright 2022 American Chemical Society.
Figure 8
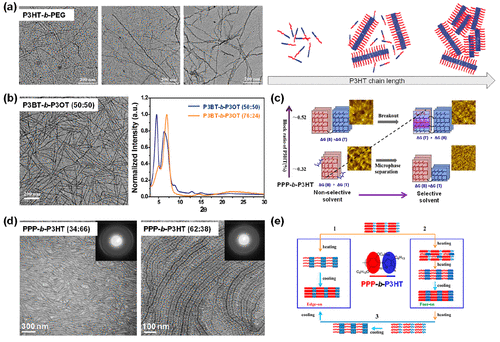
Figure 8. (a) TEM images of P3HT-b-PEG with the increased P3HT length when produced from an anisole solution. The schematic describes the chain packing influenced by the P3HT length or solvent. Reproduced with permission from ref (121). Copyright 2012 John Wiley and Sons. (b) TEM image of the P3BT-b-P3OT (50:50) in 1,2-dichlorobenzene and WAXD spectra of P3BT-b-P3OT (50:50 and 76:24). Reproduced with permission from ref (122) and modified. Copyright 2009 American Chemical Society. (c) Schematic of PPP-b-P3HT in various block ratios when produced from chlorobenzene and anisole, illustrating the preferential crystallization and microphase separation, respectively. Reproduced with permission from ref (123) and modified. Copyright 2016 Elsevier. (d) TEM images of PPP-b-P3HT (34:66 and 62:38) films annealed at 280 °C for 1 h, showing fibrillar morphology and microphase-separated nanoribbon morphology, respectively. Reproduced with permission from ref (13). Copyright 2012 American Chemical Society. (e) Three different epitaxy crystallization modes achieved in PPP-b-P3HT films during thermal annealing at different temperatures. Reproduced with permission from ref (124) and modified. Copyright 2015 American Chemical Society.
Figure 9

Figure 9. (a) AFM images of P3HT-b-Mal7 and P3HT-b-AcMal7 thin films after thermal annealing and their SAXS profiles in the as-cast state (black line) and after thermal annealing (red line). Reproduced with permission from ref (125). Copyright 2017 American Chemical Society. (b) TEM image of P3HT-b-AcMal7 thin film after annealing at 220 °C, with lamellar structure and the schematic of tilted P3HT chains with respect to the lamellar plane. Reproduced with permission from ref (126). Copyright 2020 American Chemical Society. (c) Schematic of the crystallization and microphase separation in P3HT-b-PFTBT films without (left) and with (right) inserting a certain content of the random copolymer, demonstrating dominated crystallization and microphase separation, respectively. Reproduced with permission from ref (127). Copyright 2018 American Chemical Society. (d) AFM images of P3HT-b-DPP films in the as-cast state and after thermal annealing at different temperatures. Reproduced with permission from ref (129). Copyright 2012 American Chemical Society. (e) Lorentz corrected SAXS images of P3HT-b-PPerAcr with different block ratios (left) and the corresponding TEM images (right), displaying lamellar and cylindrical morphology in bulk. Reproduced with permission from ref (130). Copyright 2013 American Chemical Society.
Figure 10
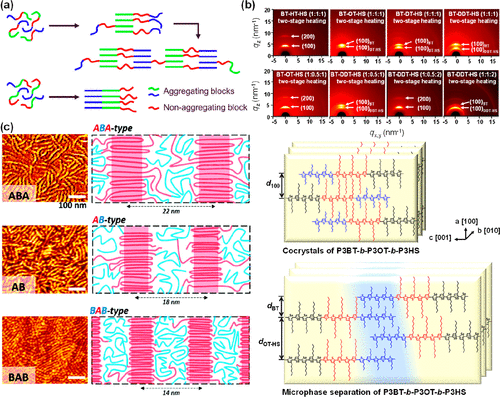
Figure 10. (a) Schematic of the solution aggregation behavior of P3AT-based triblock copolymers dependent on the block sequence. Reproduced with permission from ref (132). Copyright 2018 American Chemical Society. (b) 2D-GIWAXS images of P3BT-b-P3AT-b-P3HS triblock copolymer thin films after two-stage heating process, and the schematic of chain packing in cocrystals and microphase-separated structures represented by P3BT-b-P3OT-b-P3HS. Reproduced with permission from ref (134). Copyright 2022 Elsevier. (c) AFM images and the corresponding schematic of ABA-type, AB-type, and BAB-type (block A: P3HT, block B: POO) in the annealed thin films. Reproduced with permission from ref (135). Copyright 2019 Royal Society of Chemistry.
Figure 11
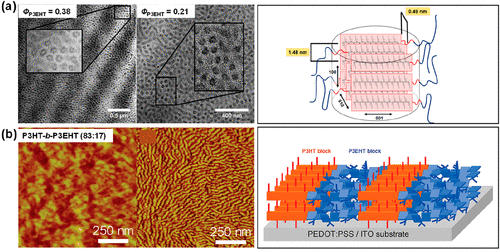
Figure 11. (a) TEM images of cylindrical microdomains in P3EHT-b-PMA with different fractions of P3EHT by melt pressing at 150 °C, and the corresponding chain packing confined in cylinders. Reproduced with permission from ref (139). Copyright 2017 American Chemical Society. (b) AFM images of P3HT-b-P3EHT (83:17) thin films after thermal annealing and the corresponding schematic of chain packing. Reproduced with permission from ref (141). Copyright 2009 American Chemical Society.
Figure 12

Figure 12. (a) TEM images of P3HT-b-P3PHT (1:1 and 3:1) in anisole. Reproduced with permission from ref (152). Copyright 2016 Royal Society of Chemistry. (b) AFM image and the schematic of P3HT-b-P3HHT helical nanofibers produced from the pyridine solution. Reproduced with permission from ref (150). Copyright 2018 Royal Society of Chemistry. (c) AFM images of P3HT-b-P3THA thin films (78:22 and 60:40) and the schematic of chain packing. Reproduced with permission from ref (149). Copyright 2012 American Chemical Society.
Figure 13
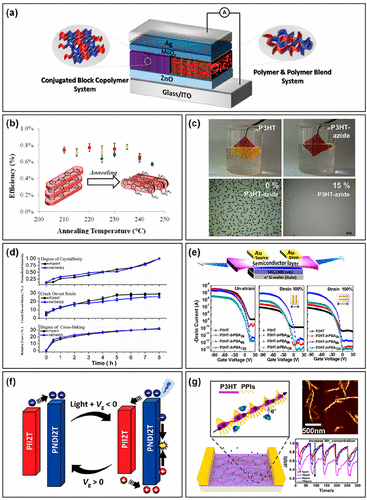
Figure 13. (a) Schematic of a photovoltaic device based on the PBDT2T-b-N2200 BCP film as the active layer. Reproduced with permission from ref (161). Copyright 2020 John Wiley and Sons. (b) The PCE based on the P3HT-b-PFTBT active layer as a function of thermal annealing time. Reproduced with permission from ref (162). Copyright 2015 American Chemical Society. (c) Optical microscopy images of P3HT and UV cross-linked P3HT-azide BCP immersed in chlorobenzene, and the P3HT/fullerene blended films containing 0% and 15% P3HT-azide BCP after thermal annealing. Reproduced with permission from ref (164). Copyright 2012 American Chemical Society. (d) The degree of cross-linking, crack onset strains, and degree of crystallinity of P3HHT and P3HT-b-P3HHT BCP as a function of heating time. Reproduced with permission from ref (165). Copyright 2016 American Chemical Society. (e) Schematic of the OFET device based on the P3HT-b-PBA as the active layer and their transfer curves with or without strain. Reproduced with permission from ref (167). Copyright 2017 American Chemical Society. (f) Schematic of the electret free phototransistor memory and working mechanism. Reproduced with permission from ref (159). Copyright 2022 John Wiley and Sons. (g) Schematic of the ammonia sensor based on OFETs of helical nanofibril P3HT-b-PPI. Reproduced with permission from ref (169). Copyright 2018 American Chemical Society.
References
This article references 170 other publications.
- 1Shirakawa, H.; Louis, E. J.; MacDiarmid, A. G.; Chiang, C. K.; Heeger, A. J. Synthesis of Electrically Conducting Organic Polymers: Halogen Derivatives of Polyacetylene, (CH)X. J. Chem. Soc., Chem. Commun. 1977, 578– 580, DOI: 10.1039/c39770000578There is no corresponding record for this reference.
- 2Sirringhaus, H. 25th Anniversary Article: Organic Field-Effect Transistors: The Path Beyond Amorphous Silicon. Adv. Mater. 2014, 26, 1319– 1335, DOI: 10.1002/adma.201304346There is no corresponding record for this reference.
- 3Ding, L.; Yu, Z.-D.; Wang, X.-Y.; Yao, Z.-F.; Lu, Y.; Yang, C.-Y.; Wang, J.-Y.; Pei, J. Polymer Semiconductors: Synthesis, Processing, and Applications. Chem. Rev. 2023, 123, 7421– 7497, DOI: 10.1021/acs.chemrev.2c00696There is no corresponding record for this reference.
- 4Gu, K. C.; Loo, Y.-L. The Polymer Physics of Multiscale Charge Transport in Conjugated Systems. J. Polym. Sci., Part B: Polym. Phys. 2019, 57, 1559– 1571, DOI: 10.1002/polb.24873There is no corresponding record for this reference.
- 5Podzorov, V.; Menard, E.; Rogers, J. A.; Gershenson, M. E. Hall Effect in the Accumulation Layers on the Surface of Organic Semiconductors. Phys. Rev. Lett. 2005, 95, 226601 DOI: 10.1103/PhysRevLett.95.226601There is no corresponding record for this reference.
- 6Xiong, H. G.; Lin, Q. J.; Lu, Y.; Zheng, D.; Li, Y. W.; Wang, S.; Xie, W. B.; Li, C. Q.; Zhang, X.; Lin, Y. Z.; Wang, Z.-X.; Shi, Q. Q.; Marks, T. J.; Huang, H. General Room-Temperature Suzuki–Miyaura Polymerization for Organic Electronics. Nat. Mater. 2024, 23, 695– 702, DOI: 10.1038/s41563-023-01794-9There is no corresponding record for this reference.
- 7Carsten, B.; He, F.; Son, H. J.; Xu, T.; Yu, L. P. Stille Polycondensation for Synthesis of Functional Materials. Chem. Rev. 2011, 111, 1493– 1528, DOI: 10.1021/cr100320wThere is no corresponding record for this reference.
- 8Martin, R.; Buchwald, S. L. Palladium-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands. Acc. Chem. Res. 2008, 41, 1461– 1473, DOI: 10.1021/ar800036sThere is no corresponding record for this reference.
- 9Mei, J. G.; Diao, Y.; Appleton, A. L.; Fang, L.; Bao, Z. N. Integrated Materials Design of Organic Semiconductors for Field-Effect Transistors. J. Am. Chem. Soc. 2013, 135, 6724– 6746, DOI: 10.1021/ja400881nThere is no corresponding record for this reference.
- 10Paterson, A. F.; Singh, S.; Fallon, K. J.; Hodsden, T.; Han, Y.; Schroeder, B. C.; Bronstein, H.; Heeney, M.; McCulloch, I.; Anthopoulos, T. D. Recent Progress in High-Mobility Organic Transistors: A Reality Check. Adv. Mater. 2018, 30, 1801079 DOI: 10.1002/adma.201801079There is no corresponding record for this reference.
- 11Luo, C.; Kyaw, A. K. K.; Perez, L. A.; Patel, S.; Wang, M.; Grimm, B.; Bazan, G. C.; Kramer, E. J.; Heeger, A. J. General Strategy for Self-Assembly of Highly Oriented Nanocrystalline Semiconducting Polymers with High Mobility. Nano Lett. 2014, 14, 2764– 2771, DOI: 10.1021/nl500758wThere is no corresponding record for this reference.
- 12Noriega, R.; Rivnay, J.; Vandewal, K.; Koch, F. P. V.; Stingelin, N.; Smith, P.; Toney, M. F.; Salleo, A. A General Relationship between Disorder, Aggregation and Charge Transport in Conjugated Polymers. Nat. Mater. 2013, 12, 1038– 1044, DOI: 10.1038/nmat3722There is no corresponding record for this reference.
- 13Yu, X. H.; Yang, H.; Wu, S. P.; Geng, Y. H.; Han, Y. C. Microphase Separation and Crystallization of All-Conjugated Phenylene–Thiophene Diblock Copolymers. Macromolecules 2012, 45, 266– 274, DOI: 10.1021/ma201024zThere is no corresponding record for this reference.
- 14Hicks, G. E. J.; Li, S.; Obhi, N. K.; Jarrett-Wilkins, C. N.; Seferos, D. S. Programmable Assembly of π-Conjugated Polymers. Adv. Mater. 2021, 33, 2006287, DOI: 10.1002/adma.202006287There is no corresponding record for this reference.
- 15Peng, J.; Han, Y. C. Recent Advances in Conjugated Polythiophene-Based Rod–Rod Block Copolymers: From Morphology Control to Optoelectronic Applications. Giant 2020, 4, 100039 DOI: 10.1016/j.giant.2020.100039There is no corresponding record for this reference.
- 16Lee, Y.; Gomez, E. D. Challenges and Opportunities in the Development of Conjugated Block Copolymers for Photovoltaics. Macromolecules 2015, 48, 7385– 7395, DOI: 10.1021/acs.macromol.5b00112There is no corresponding record for this reference.
- 17Kynaston, E. L.; Winchell, K. J.; Yee, P. Y.; Manion, J. G.; Hendsbee, A. D.; Li, Y. N.; Huettner, S.; Tolbert, S. H.; Seferos, D. S. Poly(3-alkylthiophene)-block-poly(3-alkylselenophene)s: Conjugated Diblock Co-polymers with Atypical Self-Assembly Behavior. ACS Appl. Mater. Interfaces 2019, 11, 7174– 7183, DOI: 10.1021/acsami.8b18795There is no corresponding record for this reference.
- 18Jin, S.-M.; Hwang, J. H.; Lee, E. Crystallization-Driven Solution-State Assembly of Conjugated Block Copolymers in Materials Science. Macromolecules 2023, 56, 3474– 3496, DOI: 10.1021/acs.macromol.3c00374There is no corresponding record for this reference.
- 19Yassar, A.; Miozzo, L.; Gironda, R.; Horowitz, G. Rod–Coil and All-Conjugated Block Copolymers for Photovoltaic Applications. Prog. Polym. Sci. 2013, 38, 791– 844, DOI: 10.1016/j.progpolymsci.2012.10.001There is no corresponding record for this reference.
- 20Mitchell, V. D.; Jones, D. J. Advances toward the Effective Use of Block Copolymers as Organic Photovoltaic Active Layers. Polym. Chem. 2018, 9, 795– 814, DOI: 10.1039/C7PY01878AThere is no corresponding record for this reference.
- 21Bhardwaj, A.; Kaur, J.; Wuest, M.; Wuest, F. In Situ Click Chemistry Generation of Cyclooxygenase-2 Inhibitors. Nat. Commun. 2017, 8, 1– 14, DOI: 10.1038/s41467-016-0009-6There is no corresponding record for this reference.
- 22Zhu, M. J.; Pan, S.; Wang, Y.; Tang, P.; Qiu, F.; Lin, Z. Q.; Peng, J. Unravelling the Correlation between Charge Mobility and Cocrystallization in Rod–Rod Block Copolymers for High-Performance Field-Effect Transistors. Angew. Chem., Int. Ed. 2018, 57, 8644– 8648, DOI: 10.1002/anie.201804585There is no corresponding record for this reference.
- 23Zhan, H.; Zheng, H.; Dong, Y. J.; Zhao, Q. Q.; Luo, X. B.; Peng, J. Unraveling the Phase Behavior of All-Conjugated Donor–Acceptor Block Copolymers through Molecular Engineering. Macromolecules 2024, 57, 6379– 6389, DOI: 10.1021/acs.macromol.4c00113There is no corresponding record for this reference.
- 24Cao, Z. Q.; Leng, M. W.; Cao, Y. R.; Gu, X. D.; Fang, L. How Rigid are Conjugated Non-Ladder and Ladder Polymers?. J. Polym. Sci. 2022, 60, 298– 310, DOI: 10.1002/pol.20210550There is no corresponding record for this reference.
- 25Zhang, W. L.; Gomez, E. D.; Milner, S. T. Predicting Chain Dimensions of Semiflexible Polymers from Dihedral Potentials. Macromolecules 2014, 47, 6453– 6461, DOI: 10.1021/ma500923rThere is no corresponding record for this reference.
- 26Matsen, M. M.; Bates, F. S. Unifying Weak- and Strong-Segregation Block Copolymer Theories. Macromolecules 1996, 29, 1091– 1098, DOI: 10.1021/ma951138iThere is no corresponding record for this reference.
- 27Hu, W. B. The Physics of Polymer Chain-Folding. Phys. Rep. 2018, 747, 1– 50, DOI: 10.1016/j.physrep.2018.04.004There is no corresponding record for this reference.
- 28Danielsen, S. P. O.; Bridges, C. R.; Segalman, R. A. Chain Stiffness of Donor–Acceptor Conjugated Polymers in Solution. Macromolecules 2022, 55, 437– 449, DOI: 10.1021/acs.macromol.1c02229There is no corresponding record for this reference.
- 29Cao, X. X.; Zhao, K. F.; Chen, L.; Liu, J. G.; Han, Y. C. Conjugated Polymer Single Crystals and Nanowires. Polym. Crystallization 2019, 2, 10064– 10081, DOI: 10.1002/pcr2.10064There is no corresponding record for this reference.
- 30Xiao, M. F.; Carey, R. L.; Chen, H.; Jiao, X. C.; Lemaur, V.; Schott, S.; Nikolka, M.; Jellett, C.; Sadhanala, A.; Rogers, S.; Senanayak, S. P.; Onwubiko, A.; Han, S. Y.; Zhang, Z. L.; Abdi-Jalebi, M.; Zhang, Y. C.; Thomas, T. H.; Mahmoudi, N.; Lai, L. L.; Selezneva, E.; Ren, X. L.; Nguyen, M.; Wang, Q. J.; Jacobs, I.; Yue, W.; McNeill, C. R.; Liu, G. M.; Beljonne, D.; McCulloch, I.; Sirringhaus, H. Charge Transport Physics of a Unique Class of Rigid-Rod Conjugated Polymers with Fused-Ring Conjugated Units Linked by Double Carbon-Carbon Bonds. Sci. Adv. 2021, 7, 5280– 5292, DOI: 10.1126/sciadv.abe5280There is no corresponding record for this reference.
- 31Kuei, B.; Gomez, E. D. Chain Conformations and Phase Behavior of Conjugated Polymers. Soft Matter 2017, 13, 49– 67, DOI: 10.1039/C6SM00979DThere is no corresponding record for this reference.
- 32Hu, Y. B.; Cao, X. X.; Fan, H. Crystallization of D-A Conjugated Polymers: A Review of Recent Research. Polymers 2022, 14, 4612– 4640, DOI: 10.3390/polym14214612There is no corresponding record for this reference.
- 33Dai, X. B.; Wan, H.-X.; Zhang, X. Y.; Wei, W. J.; Chen, W. L.; Zhang, L. G.; Li, J.; Yan, L.-T. Role of Conformational Entropy in Complex Macromolecular Systems. Chem. Res. Chin. Univ. 2023, 39, 709– 718, DOI: 10.1007/s40242-023-3174-2There is no corresponding record for this reference.
- 34McCulloch, B.; Ho, V.; Hoarfrost, M.; Stanley, C.; Do, C.; Heller, W. T.; Segalman, R. A. Polymer Chain Shape of Poly(3-alkylthiophenes) in Solution Using Small-Angle Neutron Scattering. Macromolecules 2013, 46, 1899– 1907, DOI: 10.1021/ma302463dThere is no corresponding record for this reference.
- 35Podgornik, R. Book Review: Polymer Physics. M. Rubenshtein and R. H. Colby, Oxford University Press, 2003. J. Stat. Phys. 2004, 115, 1757– 1761, DOI: 10.1023/B:JOSS.0000028242.25527.8bThere is no corresponding record for this reference.
- 36Gettinger, C. L.; Heeger, A. J.; Drake, J. M.; Pine, D. J. A Photoluminescence Study of Poly(phenylene vinylene) Derivatives: The Effect of Intrinsic Persistence Length. J. Chem. Phys. 1994, 101, 1673– 1678, DOI: 10.1063/1.468438There is no corresponding record for this reference.
- 37Gettinger, C. L.; Heeger, A. J.; Drake, J. M.; Pine, D. J. The Effect of Intrinsic Rigidity on the Optical Properties of PPV Derivatives. Molecular Crystals and Liquid Crystals Science and Technology. Section A. Mol. Cryst. Liq. Cryst. 1994, 256, 507– 512, DOI: 10.1080/10587259408039283There is no corresponding record for this reference.
- 38Fytas, G.; Nothofer, H. G.; Scherf, U.; Vlassopoulos, D.; Meier, G. Structure and Dynamics of Nondilute Polyfluorene Solutions. Macromolecules 2002, 35, 481– 488, DOI: 10.1021/ma011416fThere is no corresponding record for this reference.
- 39Meier, H.; Stalmach, U.; Kolshorn, H. Effective Conjugation Length and UV/vis Spectra of Oligomers. Acta Polym. 1997, 48, 379– 384, DOI: 10.1002/actp.1997.010480905There is no corresponding record for this reference.
- 40Socci, E. P.; Farmer, B. L.; Adams, W. W. Molecular Dynamics Simulations of a Poly(p-phenylene). Oligomer. J. Polym. Sci. Part B: Polym. Phys. 1993, 31, 1975– 1982, DOI: 10.1002/polb.1993.090311309There is no corresponding record for this reference.
- 41Petekidis, G.; Vlassopoulos, D.; Galda, P.; Rehahn, M.; Ballauff, M. Determination of Chain Conformation of Stiff Polymers by Depolarized Rayleigh Scattering in Solution. Macromolecules 1996, 29, 8948– 8953, DOI: 10.1021/ma961128xThere is no corresponding record for this reference.
- 42Liu, C.; Hu, W. X.; Jiang, H.; Liu, G.; Han, C. C.; Sirringhaus, H.; Boué, F.; Wang, D. Chain Conformation and Aggregation Structure Formation of a High Charge Mobility DPP-Based Donor–Acceptor Conjugated Polymer. Macromolecules 2020, 53, 8255– 8266, DOI: 10.1021/acs.macromol.0c01646There is no corresponding record for this reference.
- 43Deng, J. Y.; Zheng, L.; Ding, C. M.; Guo, Y. F.; Xie, Y. F.; Wang, J. W.; Ke, Y. B.; Li, M. M.; Li, L.; Janssen, R. A. J. Determinant Role of Solution-State Supramolecular Assembly in Molecular Orientation of Conjugated Polymer Films. Adv. Funct. Mater. 2023, 33, 2209195– 2209205, DOI: 10.1002/adfm.202209195There is no corresponding record for this reference.
- 44Gao, M. Y.; Wu, J. J.; Yuan, X. Y.; He, C. Y.; Jiang, H. Q.; Zhao, W. C.; Duan, C. H.; Chen, Y.; Ke, Y. B.; Yao, H. F.; Ye, L. Tuning the Solution Aggregation and Molecular Order for Efficient and Thermally Stable Polymer Solar Cells. Energy Environ. Sci. 2023, 16, 5822– 5831, DOI: 10.1039/D3EE02354KThere is no corresponding record for this reference.
- 45Bi, Z. Z.; Naveed, H. B.; Mao, Y. M.; Yan, H. P.; Ma, W. Importance of Nucleation during Morphology Evolution of the Blade-Cast PffBT4T-2OD-Based Organic Solar Cells. Macromolecules 2018, 51, 6682– 6691, DOI: 10.1021/acs.macromol.8b01156There is no corresponding record for this reference.
- 46Wang, Z. L.; Gao, M. Y.; He, C. Y.; Shi, W. C.; Deng, Y. F.; Han, Y.; Ye, L.; Geng, Y. H. Unraveling the Molar Mass Dependence of Shearing-Induced Aggregation Structure of a High-Mobility Polymer Semiconductor. Adv. Mater. 2022, 34, 2108255 DOI: 10.1002/adma.202108255There is no corresponding record for this reference.
- 47Tan, W. L.; Tang, L.; Matsidik, R.; Bryant, G.; Martin, T. B.; Sommer, M.; Huang, D. M.; McNeill, C. R. Small-Angle Neutron Scattering of P(NDI2OD-T2) Solutions: Importance of Network Structure for Data Interpretation and Film Morphology. Macromolecules 2024, 57, 691– 706, DOI: 10.1021/acs.macromol.3c01435There is no corresponding record for this reference.
- 48Zhao, L. H.; Png, R. Q.; Zhuo, J. M.; Wong, L. Y.; Tang, J. C.; Su, Y. S.; Chua, L. L. Role of Borderline Solvents to Induce Pronounced Extended-Chain Lamellar Order in π-Stackable Polymers. Macromolecules 2011, 44, 9692– 9702, DOI: 10.1021/ma201165yThere is no corresponding record for this reference.
- 49Heffner, G. W.; Pearson, D. S. Molecular Characterization of Poly(3-hexylthiophene). Macromolecules 1991, 24, 6295– 6299, DOI: 10.1021/ma00023a035There is no corresponding record for this reference.
- 50Cao, Z. Q.; Tolba, S. A.; Li, Z. F.; Mason, G. T.; Wang, Y.; Do, C. W.; Rondeau-Gagné, S.; Xia, W. J.; Gu, X. D. Molecular Structure and Conformational Design of Donor-Acceptor Conjugated Polymers to Enable Predictable Optoelectronic Property. Adv. Mater. 2023, 35, 2302178– 2302188, DOI: 10.1002/adma.202302178There is no corresponding record for this reference.
- 51Cao, Z.; Ma, G.; Leng, M.; Zhang, S.; Chen, J.; Do, C.; Hong, K.; Fang, L.; Gu, X. Variable-Temperature Scattering and Spectroscopy Characterizations for Temperature-Dependent Solution Assembly of PffBT4T-Based Conjugated Polymers. ACS Appl. Polym. Mater. 2022, 4, 3023– 3033, DOI: 10.1021/acsapm.1c01511There is no corresponding record for this reference.
- 52Xu, Y. C.; Ding, L.; Yao, Z. F.; Shao, Y.; Wang, J. Y.; Zhang, W. B.; Pei, J. Conjugated Polymers in Solution: A Physical Perspective. J. Phys. Chem. Lett. 2023, 14, 927– 939, DOI: 10.1021/acs.jpclett.2c03600There is no corresponding record for this reference.
- 53Lotz, B.; Miyoshi, T.; Cheng, S. Z. D. 50th Anniversary Perspective: Polymer Crystals and Crystallization: Personal Journeys in a Challenging Research Field. Macromolecules 2017, 50, 5995– 6025, DOI: 10.1021/acs.macromol.7b00907There is no corresponding record for this reference.
- 54Park, K. S.; Kwok, J. J.; Kafle, P.; Diao, Y. When Assembly Meets Processing: Tuning Multiscale Morphology of Printed Conjugated Polymers for Controlled Charge Transport. Chem. Mater. 2021, 33, 469– 498, DOI: 10.1021/acs.chemmater.0c04152There is no corresponding record for this reference.
- 55Qu, T. F.; Nan, G. M.; Ouyang, Y.; Bieketuerxun, B.; Yan, X. L.; Qi, Y. P.; Zhang, Y. Structure–Property Relationship, Glass Transition, and Crystallization Behaviors of Conjugated Polymers. Polymers 2023, 15, 4268– 4295, DOI: 10.3390/polym15214268There is no corresponding record for this reference.
- 56Luo, Y. Q.; Santos, F. A.; Wagner, T. W.; Tsoi, E.; Zhang, S. J. Dynamic Interactions Between Poly(3-hexylthiophene) and Single-Walled Carbon Nanotubes in Marginal Solvent. J. Phys. Chem. B 2014, 118, 6038– 6046, DOI: 10.1021/jp503128vThere is no corresponding record for this reference.
- 57Li, J. J.; Deepak, F. L. In Situ Kinetic Observations on Crystal Nucleation and Growth. Chem. Rev. 2022, 122, 16911– 16982, DOI: 10.1021/acs.chemrev.1c01067There is no corresponding record for this reference.
- 58Xu, J.; Reiter, G.; Alamo, R. G. Concepts of Nucleation in Polymer Crystallization. Crystals 2021, 11, 304– 322, DOI: 10.3390/cryst11030304There is no corresponding record for this reference.
- 59Karthika, S.; Radhakrishnan, T. K.; Kalaichelvi, P. A Review of Classical and Nonclassical Nucleation Theories. Cryst. Growth Des. 2016, 16, 6663– 6681, DOI: 10.1021/acs.cgd.6b00794There is no corresponding record for this reference.
- 60Tang, X. L.; Chen, W.; Li, L. B. The Tough Journey of Polymer Crystallization: Battling with Chain Flexibility and Connectivity. Macromolecules 2019, 52, 3575– 3591, DOI: 10.1021/acs.macromol.8b02725There is no corresponding record for this reference.
- 61Turnbull, D.; Fisher, J. C. Rate of Nucleation in Condensed Systems. J. Chem. Phys. 1949, 17, 71– 73, DOI: 10.1063/1.1747055There is no corresponding record for this reference.
- 62Dingler, C.; Dirnberger, K.; Ludwigs, S. Semiconducting Polymer Spherulites–From Fundamentals to Polymer Electronics. Macromol. Rapid Commun. 2019, 40, 1800601 DOI: 10.1002/marc.201800601There is no corresponding record for this reference.
- 63Patel, B. B.; Diao, Y. Multiscale Assembly of Solution-Processed Organic Electronics: The Critical Roles of Confinement, Fluid Flow, and Interfaces. Nanotechnology 2018, 29, 044004 DOI: 10.1088/1361-6528/aa9d7cThere is no corresponding record for this reference.
- 64Malik, S.; Nandi, A. K. Crystallization Mechanism of Regioregular Poly(3-alkyl thiophene)s. J. Polym. Sci., Part B: Polym. Phys. 2002, 40, 2073– 2085, DOI: 10.1002/polb.10272There is no corresponding record for this reference.
- 65Mena-Osteritz, E.; Meyer, A.; Langeveld-Voss, B. M. W.; Janssen, R. A. J.; Meijer, E. W.; Bäuerle, P. Two-Dimensional Crystals of Poly(3-Alkyl-thiophene)s: Direct Visualization of Polymer Folds in Submolecular Resolution. Angew. Chem., Int. Ed. 2000, 39, 2679– 2684, DOI: 10.1002/1521-3773(20000804)39:15<2679::AID-ANIE2679>3.0.CO;2-2There is no corresponding record for this reference.
- 66Liu, J. H.; Arif, M.; Zou, J. H.; Khondaker, S. I.; Zhai, L. Controlling Poly(3-hexylthiophene) Crystal Dimension: Nanowhiskers and Nanoribbons. Macromolecules 2009, 42, 9390– 9393, DOI: 10.1021/ma901955cThere is no corresponding record for this reference.
- 67Brun, M.; Demadrille, R.; Rannou, P.; Pron, A.; Travers, J.-P.; Grévin, B. Multiscale Scanning Tunneling Microscopy Study of Self-Assembly Phenomena in Two-Dimensional Polycrystals of π-Conjugated Polymers: The Case of Regioregular Poly(dioctylbithiophene-alt-fluorenone). Adv. Mater. 2004, 16, 2087– 2092, DOI: 10.1002/adma.200400088There is no corresponding record for this reference.
- 68Yi, S.; Zheng, H.; Peng, J. Synergistic Backbone and Side Chain Engineering on Crystalline Orientation and Charge Transport of Conjugated Polymers in Field-effect Transistors. Polymer 2025, 327, 128391 DOI: 10.1016/j.polymer.2025.128391There is no corresponding record for this reference.
- 69Yu, Z. D.; Lu, Y.; Yao, Z. F.; Wu, H. T.; Wang, Z. Y.; Pan, C. K.; Wang, J. Y.; Pei, J. Buffer Chain Model for Understanding Crystallization Competition in Conjugated Polymers. Angew. Chem., Int. Ed. 2024, 63, e202405139 DOI: 10.1002/anie.202405139There is no corresponding record for this reference.
- 70Lim, J. A.; Liu, F.; Ferdous, S.; Muthukumar, M.; Briseno, A. L. Polymer Semiconductor Crystals. Mater. Today 2010, 13, 14– 24, DOI: 10.1016/S1369-7021(10)70080-8There is no corresponding record for this reference.
- 71Qian, Z. Y.; Cao, Z. Q.; Galuska, L.; Zhang, S.; Xu, J.; Gu, X. D. Glass Transition Phenomenon for Conjugated Polymers. Macromol. Chem. Phys. 2019, 220, 1900062 DOI: 10.1002/macp.201900062There is no corresponding record for this reference.
- 72Samitsu, S.; Shimomura, T.; Heike, S.; Hashizume, T.; Ito, K. Effective Production of Poly(3-alkylthiophene) Nanofibers by Means of Whisker Method Using Anisole Solvent: Structural, Optical, and Electrical Properties. Macromolecules 2008, 41, 8000– 8010, DOI: 10.1021/ma801128vThere is no corresponding record for this reference.
- 73Chu, P. H.; Kleinhenz, N.; Persson, N.; McBride, M.; Hernandez, J. L.; Fu, B. Y.; Zhang, G. Y.; Reichmanis, E. Toward Precision Control of Nanofiber Orientation in Conjugated Polymer Thin Films: Impact on Charge Transport. Chem. Mater. 2016, 28, 9099– 9109, DOI: 10.1021/acs.chemmater.6b04202There is no corresponding record for this reference.
- 74Cao, X. X.; Du, Z. H.; Chen, L.; Zhao, K. F.; Li, H. X.; Liu, J. G.; Han, Y. C. Long Diketopyrrolopyrrole-Based Polymer Nanowires Prepared by Decreasing the Aggregate Speed of the Polymer in Solution. Polymer 2017, 118, 135– 142, DOI: 10.1016/j.polymer.2017.04.076There is no corresponding record for this reference.
- 75Um, H. A.; Lee, D. H.; Heo, D. U.; Yang, D. S.; Shin, J.; Baik, H.; Cho, M. J.; Choi, D. H. High Aspect Ratio Conjugated Polymer Nanowires for High Performance Field-Effect Transistors and Phototransistors. ACS Nano 2015, 9, 5264– 5274, DOI: 10.1021/acsnano.5b01982There is no corresponding record for this reference.
- 76Cao, X.; Fan, H. Formation of D-A Conjugated Polymer Crystals: Diffusion and Conformational Transition Theory. Polymer 2022, 243, 124606 DOI: 10.1016/j.polymer.2022.124606There is no corresponding record for this reference.
- 77Yao, Z. F.; Wang, J. Y.; Pei, J. Controlling Morphology and Microstructure of Conjugated Polymers via Solution-State Aggregation. Prog. Polym. Sci. 2023, 136, 101626 DOI: 10.1016/j.progpolymsci.2022.101626There is no corresponding record for this reference.
- 78Luo, X. B.; Zhao, Q. Q.; Li, L.; Zheng, H.; Guo, Y. N.; Peng, J. Concentration Effect on the Solution-state Aggregation and Solid-state Crystalline Structures of Poly(3-butylthiophene). Acta Polym. Sin. 2024, 55, 910– 920, DOI: 10.11777/j.issn1000-3304.2023.23302There is no corresponding record for this reference.
- 79Lan, Y. K.; Huang, C. I. Charge Mobility and Transport Behavior in the Ordered and Disordered States of the Regioregular Poly(3-hexylthiophene). J. Phys. Chem. B 2009, 113, 14555– 14564, DOI: 10.1021/jp904841jThere is no corresponding record for this reference.
- 80Liang, Q. J.; Duan, M. Z.; Geng, Z. Q.; Zhang, M.; Xu, W. Z.; Geng, H. J.; He, Z. M.; Liu, J. G. Regulation of Molecular Orientation in Organic Solar Cells. Chem. Eng. J. 2024, 488, 150783 DOI: 10.1016/j.cej.2024.150783There is no corresponding record for this reference.
- 81Zhu, S.; Peng, J. Meniscus-Assisted Solution Printing Enables Cocrystallization in Poly(3-alkylthiophene)-based Blends for Field-Effect Transistors. Chin. J. Polym. Sci. 2023, 41, 1269– 1276, DOI: 10.1007/s10118-023-2916-4There is no corresponding record for this reference.
- 82Qian, Z. Y.; Galuska, L. A.; Ma, G. R.; McNutt, W. W.; Zhang, S.; Mei, J. G.; Gu, X. D. Backbone Flexibility on Conjugated Polymer’s Crystallization Behavior and Thin Film Mechanical Stability. J. Polym. Sci. 2022, 60, 548– 558, DOI: 10.1002/pol.20210462There is no corresponding record for this reference.
- 83Yu, L.; Davidson, E.; Sharma, A.; Andersson, M. R.; Segalman, R.; Müller, C. Isothermal Crystallization Kinetics and Time-Temperature-Transformation of the Conjugated Polymer: Poly(3-(2′-ethyl)hexylthiophene). Chem. Mater. 2017, 29, 5654– 5662, DOI: 10.1021/acs.chemmater.7b01393There is no corresponding record for this reference.
- 84Qian, Z. Y.; Luo, S. C.; Qu, T. F.; Galuska, L. A.; Zhang, S.; Cao, Z. Q.; Dhakal, S.; He, Y. J.; Hong, K. L.; Zhou, D. S.; Gu, X. D. Influence of Side-Chain Isomerization on the Isothermal Crystallization Kinetics of Poly(3-alkylthiophenes). J. Mater. Res. 2021, 36, 191– 202, DOI: 10.1557/s43578-020-00062-9There is no corresponding record for this reference.
- 85Palacios, J. K.; Michell, R. M.; Müller, A. J. Crystallization, Morphology and Self-Assembly of Double, Triple and Tetra Crystalline Block Polymers. Polym. Test. 2023, 121, 107995– 108026, DOI: 10.1016/j.polymertesting.2023.107995There is no corresponding record for this reference.
- 86Matsen, M. W. Effect of Architecture on the Phase Behavior of AB-Type Block Copolymer Melts. Macromolecules 2012, 45, 2161– 2165, DOI: 10.1021/ma202782sThere is no corresponding record for this reference.
- 87Ji, S. X.; Wan, L.; Liu, C.-C.; Nealey, P. F. Directed Self-Assembly of Block Copolymers on Chemical Patterns: A Platform for Nanofabrication. Prog. Polym. Sci. 2016, 54–55, 76– 127, DOI: 10.1016/j.progpolymsci.2015.10.006There is no corresponding record for this reference.
- 88Gao, Y.; Deng, H. L.; Li, W. H.; Qiu, F.; Shi, A. C. Formation of Nonclassical Ordered Phases of AB-Type Multiarm Block Copolymers. Phys. Rev. Lett. 2016, 116, 068304 DOI: 10.1103/PhysRevLett.116.068304There is no corresponding record for this reference.
- 89Bates, F. S.; Fredrickson, G. H. Block Copolymer Thermodynamics: Theory and Experiment. Annu. Rev. Phys. Chem. 1990, 41, 525– 557, DOI: 10.1146/annurev.pc.41.100190.002521There is no corresponding record for this reference.
- 90Kim, J. K.; Yang, S. Y.; Lee, Y.; Kim, Y. Functional Nanomaterials Based on Block Copolymer Self-Assembly. Prog. Polym. Sci. 2010, 35, 1325– 1349, DOI: 10.1016/j.progpolymsci.2010.06.002There is no corresponding record for this reference.
- 91Suh, H. S.; Kim, D. H.; Moni, P.; Xiong, S. S.; Ocola, L. E.; Zaluzec, N. J.; Gleason, K. K.; Nealey, P. F. Sub-10-nm Patterning via Directed Self-Assembly of Block Copolymer Films with a Vapour-Phase Deposited Topcoat. Nat. Nanotechnol. 2017, 12, 575– 583, DOI: 10.1038/nnano.2017.34There is no corresponding record for this reference.
- 92Hu, X.-H.; Xiong, S. S. Fabrication of Nanodevices Through Block Copolymer Self-Assembly. Front. Nanotechnol. 2022, 4, 762996 DOI: 10.3389/fnano.2022.762996There is no corresponding record for this reference.
- 93Sakurai, S.; Mori, K.; Okawara, A.; Kimishima, K.; Hashimoto, T. Evaluation of Segmental Interaction by Small-Angle X-ray Scattering Based on the Random-Phase Approximation for Asymmetric, Polydisperse Triblock Copolymers. Macromolecules 1992, 25, 2679– 2691, DOI: 10.1021/ma00036a019There is no corresponding record for this reference.
- 94Feldman, K. E.; Kade, M. J.; Meijer, E. W.; Hawker, C. J.; Kramer, E. J. Phase Behavior of Complementary Multiply Hydrogen Bonded End-Functional Polymer Blends. Macromolecules 2010, 43, 5121– 5127, DOI: 10.1021/ma1003776There is no corresponding record for this reference.
- 95Scheiwiller, S. C.; Mata, J. P.; Pozzo, L. D. Morphology of Poly-3-Hexyl-Thiophene Blends with Styrene-Isoprene-Styrene Block-Copolymer Elastomers from X-ray and Neutron Scattering. Soft Matter 2024, 20, 6247– 6265, DOI: 10.1039/D4SM00495GThere is no corresponding record for this reference.
- 96Olsen, B. D.; Shah, M.; Ganesan, V.; Segalman, R. A. Universalization of the Phase Diagram for a Model Rod-Coil Diblock Copolymer. Macromolecules 2008, 41, 6809– 6817, DOI: 10.1021/ma800978cThere is no corresponding record for this reference.
- 97Kim, W. N.; Burns, C. M. Blends of Polycarbonate and Poly(methyl methacrylate) and the Determination of the Polymer-Polymer Interaction Parameter of the Two Polymers. Macromolecules 1987, 20, 1876– 1882, DOI: 10.1021/ma00174a030There is no corresponding record for this reference.
- 98Kipp, D.; Mok, J.; Strzalka, J.; Darling, S. B.; Ganesan, V.; Verduzco, R. Rational Design of Thermally Stable, Bicontinuous Donor/Acceptor Morphologies with Conjugated Block Copolymer Additives. ACS Macro Lett. 2015, 4, 867– 871, DOI: 10.1021/acsmacrolett.5b00413There is no corresponding record for this reference.
- 99Leibler, L. Theory of Microphase Separation in Block Copolymers. Macromolecules 1980, 13, 1602– 1617, DOI: 10.1021/ma60078a047There is no corresponding record for this reference.
- 100Porter, J. D. The Solubility of Nonelectrolytes. 3rd ed. Joel H. Hildebrand and Robert L. Scott. New York: Reinhold, 1950.. Science 1951, 113, 450– 451, DOI: 10.1126/science.113.2938.450-bThere is no corresponding record for this reference.
- 101Miquelard-Garnier, G.; Roland, S. Beware of the Flory Parameter to Characterize Polymer-Polymer Interactions: A Critical Reexamination of the Experimental Literature. Eur. Polym. J. 2016, 84, 111– 124, DOI: 10.1016/j.eurpolymj.2016.09.009There is no corresponding record for this reference.
- 102Liu, C. L.; Lin, C. H.; Kuo, C. C.; Lin, S. T.; Chen, W. C. Conjugated Rod–Coil Block Copolymers: Synthesis, Morphology, Photophysical Properties, and Stimuli-Responsive Applications. Prog. Polym. Sci. 2011, 36, 603– 637, DOI: 10.1016/j.progpolymsci.2010.07.008There is no corresponding record for this reference.
- 103Park, H.; Ma, B. S.; Kim, J. S.; Kim, Y.; Kim, H. J.; Kim, D.; Yun, H.; Han, J.; Kim, F. S.; Kim, T.-S.; Kim, B. J. Regioregular-block-Regiorandom Poly(3-hexylthiophene) Copolymers for Mechanically Robust and High-Performance Thin-Film Transistors. Macromolecules 2019, 52, 7721– 7730, DOI: 10.1021/acs.macromol.9b01540There is no corresponding record for this reference.
- 104Jiang, Y.; Chen, J. Z. Y. Influence of Chain Rigidity on the Phase Behavior of Wormlike Diblock Copolymers. Phys. Rev. Lett. 2013, 110, 138305 DOI: 10.1103/PhysRevLett.110.138305There is no corresponding record for this reference.
- 105He, W. N.; Xu, J. T. Crystallization Assisted Self-Assembly of Semicrystalline Block Copolymers. Prog. Polym. Sci. 2012, 37, 1350– 1400, DOI: 10.1016/j.progpolymsci.2012.05.002There is no corresponding record for this reference.
- 106Loo, Y.-L.; Register, R. A.; Ryan, A. J. Modes of Crystallization in Block Copolymer Microdomains: Breakout, Templated, and Confined. Macromolecules 2002, 35, 2365– 2374, DOI: 10.1021/ma011824jThere is no corresponding record for this reference.
- 107Xiao, L. L.; Zhou, X.; Yue, K.; Guo, Z. H. Synthesis and Self-Assembly of Conjugated Block Copolymers. Polymers 2021, 13, 110– 129, DOI: 10.3390/polym13010110There is no corresponding record for this reference.
- 108Zhang, L.; Li, H. X.; Zhao, K. F.; Zhang, T.; Liu, D.; Wang, S. C.; Wu, F.; Zhang, Q.; Han, Y. C. Achieving the High Charge Mobility of Conjugated Polymers under Cyclic Stretching by Changing the Interaction Parameter between Solvent and Sidechain. Polymer 2023, 275, 125912 DOI: 10.1016/j.polymer.2023.125912There is no corresponding record for this reference.
- 109Chen, L.; Zhao, K. F.; Cao, X. X.; Liu, J. G.; Yu, X. H.; Han, Y. H. Nanowires of Conjugated Polymer Prepared by Tuning the Interaction between the Solvent and Polymer. Polymer 2018, 149, 23– 29, DOI: 10.1016/j.polymer.2018.06.068There is no corresponding record for this reference.
- 110Kynaston, E. L.; Fang, Y.; Manion, J. G.; Obhi, N. K.; Howe, J. Y.; Perepichka, D. F.; Seferos, D. S. Patchy Nanofibers from the Thin Film Self-Assembly of A Conjugated Diblock Copolymer. Angew. Chem., Int. Ed. 2017, 56, 6152– 6156, DOI: 10.1002/anie.201700134There is no corresponding record for this reference.
- 111Chen, S. W.; Zheng, H.; Liu, X. F.; Peng, J. Tailoring Co-crystallization over Microphase Separation in Conjugated Block Copolymers via Rational Film Processing for Field-Effect Transistors. Macromolecules 2022, 55, 10405– 10414, DOI: 10.1021/acs.macromol.2c02048There is no corresponding record for this reference.
- 112Zhao, Q. Q.; Zheng, H.; Li, D. K.; Peng, J. Interrogating Cocrystallization and Microphase Segregation in Meticulously Engineered Rod–Rod Poly(thieno)thiophene-Based Block Copolymers for Organic Field-Effect Transistors. Macromolecules 2024, 57, 4782– 4792, DOI: 10.1021/acs.macromol.4c00632There is no corresponding record for this reference.
- 113Kline, R. J.; McGehee, M. D.; Kadnikova, E. N.; Liu, J. S.; Fréchet, J. M. J. Controlling the Field-Effect Mobility of Regioregular Polythiophene by Changing the Molecular Weight. Adv. Mater. 2003, 15, 1519– 1522, DOI: 10.1002/adma.200305275There is no corresponding record for this reference.
- 114Chen, S. W.; Li, L. X.; Zhai, D. L.; Yin, Y.; Shang, X.; Ni, B. J.; Peng, J. Cocrystallization-Promoted Charge Mobility in All-Conjugated Diblock Copolymers for High-Performance Field-Effect Transistors. ACS Appl. Mater. Interfaces 2020, 12, 58094– 58104, DOI: 10.1021/acsami.0c17671There is no corresponding record for this reference.
- 115Ge, J.; He, M.; Qiu, F.; Yang, Y. L. Synthesis, Cocrystallization, and Microphase Separation of All-Conjugated Diblock Copoly(3-alkylthiophene)s. Macromolecules 2010, 43, 6422– 6428, DOI: 10.1021/ma1010167There is no corresponding record for this reference.
- 116Kim, Y.; Kim, H. J.; Kim, J.-S.; Yun, H.; Park, H.; Han, J.; Kim, B. J. Modulating Regioregularity of Poly(3-hexylthiophene)-based Amphiphilic Block Copolymers To Control Solution Assembly from Nanowires to Micelles. Chem. Mater. 2018, 30, 7912– 7921, DOI: 10.1021/acs.chemmater.8b03510There is no corresponding record for this reference.
- 117Kim, Y.-J.; Cho, C.-H.; Paek, K.; Jo, M.; Park, M.-k.; Lee, N.-E.; Kim, Y.-j.; Kim, B. J.; Lee, E. Precise Control of Quantum Dot Location within the P3HT-b-P2VP/QD Nanowires Formed by Crystallization-Driven 1D Growth of Hybrid Dimeric Seeds. J. Am. Chem. Soc. 2014, 136, 2767– 2774, DOI: 10.1021/ja410165fThere is no corresponding record for this reference.
- 118Kim, J. S.; Kim, Y.; Kim, H. J.; Kim, H. J.; Yang, H.; Jung, Y. S.; Stein, G. E.; Kim, B. J. Regioregularity-Driven Morphological Transition of Poly(3-hexylthiophene)-Based Block Copolymers. Macromolecules 2017, 50, 1902– 1908, DOI: 10.1021/acs.macromol.7b00128There is no corresponding record for this reference.
- 119Coote, J. P.; Kim, J. S.; Lee, B.; Han, J.; Kim, B. J.; Stein, G. E. Crystallization Modes of Poly(3-dodecylthiophene)-Based Block Copolymers Depend on Regioregularity and Morphology. Macromolecules 2018, 51, 9276– 9283, DOI: 10.1021/acs.macromol.8b01985There is no corresponding record for this reference.
- 120Kim, P.; Kim, E. Y.; Han, S. G.; Lee, J.; Kang, S.; Park, C.; Cho, K.; Moon, H. C.; Kim, J. K. Enhanced Vertical Hole Mobility Through End-on Chain Orientation of Poly(3-hexylthiophene)-Based Diblock Copolymers by Microphase Separation. Macromolecules 2022, 55, 6160– 6166, DOI: 10.1021/acs.macromol.2c00791There is no corresponding record for this reference.
- 121Yang, H.; Xia, H.; Wang, G. W.; Peng, J.; Qiu, F. Insights into Poly(3-hexylthiophene)-b-Poly(ethylene oxide) Block Copolymer: Synthesis and Solvent-Induced Structure Formation in Thin Films. J. Polym. Sci., Polym. Chem. 2012, 50, 5060– 5067, DOI: 10.1002/pola.26353There is no corresponding record for this reference.
- 122Wu, P. T.; Ren, G. Q.; Li, C. X.; Mezzenga, R.; Jenekhe, S. A. Crystalline Diblock Conjugated Copolymers: Synthesis, Self-Assembly, and Microphase Separation of Poly(3-butylthiophene)-b-poly(3-octylthiophene). Macromolecules 2009, 42, 2317– 2320, DOI: 10.1021/ma802874vThere is no corresponding record for this reference.
- 123Yang, H.; Zhang, R.; Wang, L.; Zhang, J. D.; Yu, X. H.; Geng, Y. H.; Han, Y. C. Crystallization Assisted Microphase Separation in All-Conjugated Phenylene-Thiophene Diblock Copolymers. Polymer 2016, 97, 238– 246, DOI: 10.1016/j.polymer.2016.05.037There is no corresponding record for this reference.
- 124Yang, H.; Zhang, R.; Wang, L.; Zhang, J. D.; Yu, X. H.; Liu, J. G.; Xing, R. B.; Geng, Y. H.; Han, Y. C. Face-On and Edge-On Orientation Transition and Self-Epitaxial Crystallization of All-Conjugated Diblock Copolymer. Macromolecules 2015, 48, 7557– 7566, DOI: 10.1021/acs.macromol.5b01804There is no corresponding record for this reference.
- 125Sakai-Otsuka, Y.; Zaioncz, S.; Otsuka, I.; Halila, S.; Rannou, P.; Borsali, R. Self-Assembly of Carbohydrate-block-Poly(3-hexylthiophene) Diblock Copolymers into Sub-10 nm Scale Lamellar Structures. Macromolecules 2017, 50, 3365– 3376, DOI: 10.1021/acs.macromol.7b00118There is no corresponding record for this reference.
- 126Sakai-Otsuka, Y.; Nishiyama, Y.; Putaux, J. L.; Brinkmann, M.; Satoh, T.; Chen, W.-C.; Borsali, R. Competing Molecular Packing of Blocks in a Lamella-Forming Carbohydrate-block-poly(3-hexylthiophene) Copolymer. Macromolecules 2020, 53, 9054– 9064, DOI: 10.1021/acs.macromol.0c01801There is no corresponding record for this reference.
- 127Lee, Y.; Aplan, M. P.; Seibers, Z. D.; Xie, R. X.; Culp, T. E.; Wang, C.; Hexemer, A.; Kilbey, S. M.; Wang, Q.; Gomez, E. D. Random Copolymers Allow Control of Crystallization and Microphase Separation in Fully Conjugated Block Copolymers. Macromolecules 2018, 51, 8844– 8852, DOI: 10.1021/acs.macromol.8b01859There is no corresponding record for this reference.
- 128Brady, M. A.; Ku, S. Y.; Perez, L. A.; Cochran, J. E.; Schmidt, K.; Weiss, T. M.; Toney, M. F.; Ade, H.; Hexemer, A.; Wang, C.; Hawker, C. J.; Kramer, E. J.; Chabinyc, M. L. Role of Solution Structure in Self-Assembly of Conjugated Block Copolymer Thin Films. Macromolecules 2016, 49, 8187– 8197, DOI: 10.1021/acs.macromol.6b01686There is no corresponding record for this reference.
- 129Ku, S. Y.; Brady, M. A.; Treat, N. D.; Cochran, J. E.; Robb, M. J.; Kramer, E. J.; Chabinyc, M. L.; Hawker, C. J. A Modular Strategy for Fully Conjugated Donor-Acceptor Block Copolymers. J. Am. Chem. Soc. 2012, 134, 16040– 16046, DOI: 10.1021/ja307431kThere is no corresponding record for this reference.
- 130Lohwasser, R. H.; Gupta, G.; Kohn, P.; Sommer, M.; Lang, A. S.; Thurn-Albrecht, T.; Thelakkat, M. Phase Separation in the Melt and Confined Crystallization as the Key to Well-Ordered Microphase Separated Donor–Acceptor Block Copolymers. Macromolecules 2013, 46, 4403– 4410, DOI: 10.1021/ma3021147There is no corresponding record for this reference.
- 131Verswyvel, M.; Steverlynck, J.; Hadj Mohamed, S.; Trabelsi, M.; Champagne, B.; Koeckelberghs, G. All-Conjugated ABC-block-copolymer Formation with a Varying Sequence via an Unassociated Catalyst. Macromolecules 2014, 47, 4668– 4675, DOI: 10.1021/ma500610pThere is no corresponding record for this reference.
- 132Verheyen, L.; Timmermans, B.; Koeckelberghs, G. Influence of the Sequence in Conjugated Triblock Copolymers on Their Aggregation Behavior. Macromolecules 2018, 51, 6421– 6429, DOI: 10.1021/acs.macromol.8b01302There is no corresponding record for this reference.
- 133Zhu, S. Y.; Li, D. K.; Zhan, H.; Chen, S. W.; Peng, J. Synergistic Manipulation of Phase Transition between Cocrystallization and Microphase Segregation in Conjugated Triblock Copolymers for Organic Field-Effect Transistors. Macromolecules 2024, 57, 2067– 2077, DOI: 10.1021/acs.macromol.3c02275There is no corresponding record for this reference.
- 134Li, L. X.; Zhao, Q. Q.; Chen, S. W.; Lin, Z. Q.; Peng, J. Unfolding the Cocrystallization–Charge Transport Correlation in All-Conjugated Triblock Copolymers via Meticulous Molecular Engineering for Organic Field-Effect Transistors. Nano Energy 2022, 100, 107489– 107497, DOI: 10.1016/j.nanoen.2022.107489There is no corresponding record for this reference.
- 135Chiang, Y. C.; Kobayashi, S.; Isono, T.; Shih, C. C.; Shingu, T.; Hung, C.-C.; Hsieh, H.-C.; Tung, S.-H.; Satoh, T.; Chen, W.-C. Effect of A Conjugated/Elastic Block Sequence on the Morphology and Electronic Properties of Polythiophene Based Stretchable Block Copolymers. Polym. Chem. 2019, 10, 5452– 5464, DOI: 10.1039/C9PY01216HThere is no corresponding record for this reference.
- 136Zhang, Y.; Tajima, K.; Hirota, K.; Hashimoto, K. Synthesis of All-Conjugated Diblock Copolymers by Quasi-Living Polymerization and Observation of Their Microphase Separation. J. Am. Chem. Soc. 2008, 130, 7812– 7813, DOI: 10.1021/ja8023516There is no corresponding record for this reference.
- 137Ho, V.; Boudouris, B. W.; McCulloch, B. L.; Shuttle, C. G.; Burkhardt, M.; Chabinyc, M. L.; Segalman, R. A. Poly(3-alkylthiophene) Diblock Copolymers with Ordered Microstructures and Continuous Semiconducting Pathways. J. Am. Chem. Soc. 2011, 133, 9270– 9273, DOI: 10.1021/ja2035317There is no corresponding record for this reference.
- 138Lee, I. H.; Amaladass, P.; Yoon, K. Y.; Shin, S.; Kim, Y.-J.; Kim, I.; Lee, E.; Choi, T.-L. Nanostar and Nanonetwork Crystals Fabricated by In Situ Nanoparticlization of Fully Conjugated Polythiophene Diblock Copolymers. J. Am. Chem. Soc. 2013, 135, 17695– 17698, DOI: 10.1021/ja409206pThere is no corresponding record for this reference.
- 139Davidson, E. C.; Segalman, R. A. Confined Crystallization within Cylindrical P3EHT Block Copolymer Microdomains. Macromolecules 2017, 50, 6128– 6136, DOI: 10.1021/acs.macromol.7b01323There is no corresponding record for this reference.
- 140Davidson, E. C.; Segalman, R. A. Thermal Control of Confined Crystallization within P3EHT Block Copolymer Microdomains. Macromolecules 2017, 50, 8097– 8105, DOI: 10.1021/acs.macromol.7b01616There is no corresponding record for this reference.
- 141Zhang, Y.; Tajima, K.; Hashimoto, K. Nanostructure Formation in Poly(3-hexylthiophene-block-3-(2-ethylhexyl)thiophene)s. Macromolecules 2009, 42, 7008– 7015, DOI: 10.1021/ma9013065There is no corresponding record for this reference.
- 142Lee, Y. H.; Chen, W. C.; Yang, Y. L.; Chiang, C. J.; Yokozawa, T.; Dai, C.-A. Co-Crystallization Phase Transformations in All Pi-Conjugated Block Copolymers With Different Main-Chain Moieties. Nanoscale 2014, 6, 5208– 5216, DOI: 10.1039/c3nr06037cThere is no corresponding record for this reference.
- 143Moon, H. C.; Bae, D.; Kim, J. K. Self-Assembly of Poly(3-dodecylthiophene)-block-Poly(methyl methacrylate) Copolymers Driven by Competition between Microphase Separation and Crystallization. Macromolecules 2012, 45, 5201– 5207, DOI: 10.1021/ma300902nThere is no corresponding record for this reference.
- 144Lin, S. H.; Wu, S. J.; Ho, C. C.; Su, W. F. Rational Design of Versatile Self-Assembly Morphology of Rod-Coil Block Copolymer. Macromolecules 2013, 46, 2725– 2732, DOI: 10.1021/ma302220bThere is no corresponding record for this reference.
- 145Boudouris, B. W.; Frisbie, C. D.; Hillmyer, M. A. Nanoporous Poly(3-alkylthiophene) Thin Films Generated from Block Copolymer Templates. Macromolecules 2008, 41, 67– 75, DOI: 10.1021/ma071626dThere is no corresponding record for this reference.
- 146Chen, Y.; Cui, H. J.; Li, L. S.; Tian, Z. Y.; Tang, Z. Y. Controlling Micro-Phase Separation in Semi-Crystalline/Amorphous Conjugated Block Copolymers. Polym. Chem. 2014, 5, 4400– 4404, DOI: 10.1039/C4PY00498AThere is no corresponding record for this reference.
- 147Li, W.; He, Y. G.; Shi, S. Y.; Liu, N.; Zhu, Y. Y.; Ding, Y. S.; Yin, J.; Wu, Z. Q. Fabrication of A Multi-Charge Generable Poly(phenyl Isocyanide)-block-Poly(3-hexylthiophene) Rod–Rod Conjugated Copolymer. Polym. Chem. 2015, 6, 2348– 2355, DOI: 10.1039/C4PY01624FThere is no corresponding record for this reference.
- 148Kim, H. J.; Skinner, M.; Yu, H.; Oh, J. H.; Briseno, A. L.; Emrick, T.; Kim, B. J.; Hayward, R. C. Water Processable Polythiophene Nanowires by Photo-Cross-Linking and Click-Functionalization. Nano Lett. 2015, 15, 5689– 5695, DOI: 10.1021/acs.nanolett.5b01185There is no corresponding record for this reference.
- 149Ho, C. C.; Liu, Y. C.; Lin, S. H.; Su, W. F. Synthesis, Morphology, and Optical and Electrochemical Properties of Poly(3-hexylthiophene)-b-poly(3-thiophene hexylacetate). Macromolecules 2012, 45, 813– 820, DOI: 10.1021/ma202164dThere is no corresponding record for this reference.
- 150Cui, H. N.; Chen, X. S.; Wang, Y.; Wei, D. C.; Qiu, F.; Peng, J. Hydrogen-Bonding-Directed Helical Nanofibers in A Polythiophene-Based All-Conjugated Diblock Copolymer. Soft Matter 2018, 14, 5906– 5912, DOI: 10.1039/C8SM01130CThere is no corresponding record for this reference.
- 151Shi, Y. Q.; Tan, L. C.; Chen, L.; Chen, Y. W. In Situ Fabricating One-Dimensional Donor–Acceptor Core-Shell Hybrid Nanobeams Network Driven by Self-Assembly of Diblock Copolythiophenes. Macromolecules 2014, 47, 1757– 1767, DOI: 10.1021/ma402154gThere is no corresponding record for this reference.
- 152Zhu, M. J.; Kim, H.; Jang, Y. J.; Park, S.; Ryu, D. Y.; Kim, K.; Tang, P.; Qiu, F.; Kim, D. H.; Peng, J. Toward High Efficiency Organic Photovoltaic Devices with Enhanced Thermal Stability Utilizing P3HT-b-P3PHT Block Copolymer Additives. J. Mater. Chem. A 2016, 4, 18432– 18443, DOI: 10.1039/C6TA08181AThere is no corresponding record for this reference.
- 153Xu, L.; Xu, X.-H.; Liu, N.; Zou, H.; Wu, Z.-Q. A Facile Synthetic Route to Multifunctional Poly(3-hexylthiophene)-b-poly(phenyl isocyanide) Copolymers: From Aggregation-Induced Emission to Controlled Helicity. Macromolecules 2018, 51, 7546– 7555, DOI: 10.1021/acs.macromol.8b01478There is no corresponding record for this reference.
- 154Timmermans, B.; Koeckelberghs, G. Chiral expression of co-crystallizing poly(thiophene)-block-poly(selenophene) copolymers. Polym. Chem. 2020, 11, 2715– 2723, DOI: 10.1039/C9PY01775EThere is no corresponding record for this reference.
- 155Lombeck, F.; Komber, H.; Sepe, A.; Friend, R. H.; Sommer, M. Enhancing Phase Separation and Photovoltaic Performance of All-Conjugated Donor–Acceptor Block Copolymers with Semifluorinated Alkyl Side Chains. Macromolecules 2015, 48, 7851– 7860, DOI: 10.1021/acs.macromol.5b01845There is no corresponding record for this reference.
- 156Mitchell, V. D.; Gann, E.; Huettner, S.; Singh, C. R.; Subbiah, J.; Thomsen, L.; McNeill, C. R.; Thelakkat, M.; Jones, D. J. Morphological and Device Evaluation of an Amphiphilic Block Copolymer for Organic Photovoltaic Applications. Macromolecules 2017, 50, 4942– 4951, DOI: 10.1021/acs.macromol.7b00377There is no corresponding record for this reference.
- 157Gao, D.; Hollinger, J.; Seferos, D. S. Selenophene-Thiophene Block Copolymer Solar Cells with Thermostable Nanostructures. ACS Nano 2012, 6, 7114– 7121, DOI: 10.1021/nn3021844There is no corresponding record for this reference.
- 158Yin, Y.; Zhai, D.; Chen, S.; Shang, X.; Li, L.; Peng, J. Controlling the Condensed Structure of Polythiophene and Polyselenophene-based All-conjugated Block Copolymers. Acta Polym. Sin. 2020, 51, 434– 447There is no corresponding record for this reference.
- 159Yang, W. C.; Chen, Y. W.; Yu, Y. Y.; Lin, Y. C.; Higashihara, T.; Chen, W. C. Enhancing the Performance of Electret-Free Phototransistor Memory by Using All-Conjugated Block Copolymer. Macromol. Rapid Commun. 2023, 44, 2200756 DOI: 10.1002/marc.202200756There is no corresponding record for this reference.
- 160Jung, Y. S.; Jung, W.; Tuller, H. L.; Ross, C. A. Nanowire Conductive Polymer Gas Sensor Patterned Using Self-Assembled Block Copolymer Lithography. Nano Lett. 2008, 8, 3776– 3780, DOI: 10.1021/nl802099kThere is no corresponding record for this reference.
- 161Park, S. H.; Kim, Y.; Kwon, N. Y.; Lee, Y. W.; Woo, H. Y.; Chae, W. S.; Park, S.; Cho, M. J.; Choi, D. H. Significantly Improved Morphology and Efficiency of Nonhalogenated Solvent-Processed Solar Cells Derived from a Conjugated Donor-Acceptor Block Copolymer. Adv. Sci. 2020, 7, 1902470 DOI: 10.1002/advs.201902470There is no corresponding record for this reference.
- 162Smith, K. A.; Lin, Y.-H.; Mok, J. W.; Yager, K. G.; Strzalka, J.; Nie, W.; Mohite, A. D.; Verduzco, R. Molecular Origin of Photovoltaic Performance in Donor-block-Acceptor All-Conjugated Block Copolymers. Macromolecules 2015, 48, 8346– 8353, DOI: 10.1021/acs.macromol.5b01383There is no corresponding record for this reference.
- 163Park, S. H.; Kwon, N. Y.; Jung, S. H.; Harit, A. K.; Woo, H. Y.; Cho, M. J.; Choi, D. H. Enhanced Efficiency and Stability of Novel Pseudo-ternary Polymer Solar Cells Enabled by a Conjugated Donor Block Copolymer. ACS Appl. Mater. Interfaces. 2023, 15, 20266– 20277, DOI: 10.1021/acsami.3c00815There is no corresponding record for this reference.
- 164Kim, H. J.; Han, A.-R.; Cho, C.-H.; Kang, H.; Cho, H.-H.; Lee, M. Y.; Fréchet, J. M. J.; Oh, J. H.; Kim, B. J. Solvent-Resistant Organic Transistors and Thermally Stable Organic Photovoltaics Based on Cross-linkable Conjugated Polymers. Chem. Mater. 2012, 24, 215– 221, DOI: 10.1021/cm203058pThere is no corresponding record for this reference.
- 165Yang, X. B.; Ge, J.; He, M.; Ye, Z.; Liu, X. F.; Peng, J.; Qiu, F. Crystallization and Microphase Morphology of Side-Chain Cross-Linkable Poly(3-hexylthiophene)-block-poly[3-(6-hydroxy) hexylthiophene] Diblock Copolymers. Macromolecules 2016, 49, 287– 297, DOI: 10.1021/acs.macromol.5b02001There is no corresponding record for this reference.
- 166Li, X. Y.; Wolanin, P. J.; MacFarlane, L. R.; Harniman, R. L.; Qian, J. S.; Gould, O. E. C.; Dane, T. G.; Rudin, J.; Cryan, M. J.; Schmaltz, T.; Frauenrath, H.; Winnik, M. A.; Faul, C. F. J.; Manners, I. Uniform Electroactive Fibre-Like Micelle Nanowires for Organic Electronics. Nat. Commun. 2017, 8, 15909– 15917, DOI: 10.1038/ncomms15909There is no corresponding record for this reference.
- 167Wang, J.-T.; Takshima, S.; Wu, H.-C.; Shih, C.-C.; Isono, T.; Kakuchi, T.; Satoh, T.; Chen, W.-C. Stretchable Conjugated Rod–Coil Poly(3-hexylthiophene)-block-poly(butyl acrylate) Thin Films for Field Effect Transistor Applications. Macromolecules 2017, 50, 1442– 1452, DOI: 10.1021/acs.macromol.6b02722There is no corresponding record for this reference.
- 168Xu, L.; Wang, C.; Li, Y. X.; Xu, X. H.; Zhou, L.; Liu, N.; Wu, Z. Q. Crystallization-Driven Asymmetric Helical Assembly of Conjugated Block Copolymers and the Aggregation Induced White-light Emission and Circularly Polarized Luminescence. Angew. Chem., Int. Ed. 2020, 59, 16675– 16682, DOI: 10.1002/anie.202006561There is no corresponding record for this reference.
- 169Wei, S.; Tian, F.; Ge, F.; Wang, X. H.; Zhang, G. B.; Lu, H. B.; Yin, J.; Wu, Z. Q.; Qiu, L. Z. Helical Nanofibrils of Block Copolymer for High-Performance Ammonia Sensors. ACS Appl. Mater. Interfaces 2018, 10, 22504– 22512, DOI: 10.1021/acsami.8b06458There is no corresponding record for this reference.
- 170Pei, J. Multi-level Aggregation of Conjugated Polymers in Solution: Advances and Challenges. Chin. J. Polym. Sci. 2023, 41, 1498– 1500, DOI: 10.1007/s10118-023-2938-yThere is no corresponding record for this reference.