Convergent Synthesis of Tetradentate Aminopyridine C–H Oxidation CatalystsClick to copy article linkArticle link copied!
- Yiheng LuYiheng LuDepartment of Chemistry, University of Basel, Mattenstrasse 22, 4058 Basel, SwitzerlandMore by Yiheng Lu
- Kanstantsin AnisovichKanstantsin AnisovichDepartment of Chemistry, University of Basel, Mattenstrasse 22, 4058 Basel, SwitzerlandMore by Kanstantsin Anisovich
- Konrad Tiefenbacher*Konrad Tiefenbacher*Email: [email protected]; [email protected]Department of Chemistry, University of Basel, Mattenstrasse 22, 4058 Basel, SwitzerlandDepartment of Biosystems Science and Engineering, ETH Zürich, Klingelbergstrasse 48, 4056 Basel, SwitzerlandMore by Konrad Tiefenbacher
Abstract
Controlling the oxidation of unactivated C(sp3)–H bonds through supramolecular catalyst–substrate interactions has recently enabled site-selective transformations previously considered inaccessible. However, progress has been hampered by lengthy linear syntheses of supramolecular catalysts. Here we present a convergent strategy that directly cross-couples preformed tetradentate aminopyridine ligands with recognition motifs, streamlining access to functional catalysts. Using this approach, we prepared four catalysts, two featuring resorcin[4]arene (RS) and two bearing calix[4]arene (CX) units, for substrate recognition using the solvophobic effect in 2,2,2-trifluoroethanol. This convergent synthesis reduces the longest linear sequence from 5 steps to 2 steps (starting from the recognition motif) by shifting from a linear, catalyst-specific synthesis to a modular platform that allows rapid assembly of catalyst libraries using premade, interchangeable building blocks.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Significant progress has recently been achieved in controlling the oxidation of unactivated C(sp3)–H bonds through supramolecular catalyst–substrate interactions. Covalent attachment of recognition motifs to state-of-the-art tetradentate iron and manganese aminopyridine complexes (1−4) has enabled site-selective oxidation of unactivated and even deactivated C–H bonds in alkyl- and steroidal-ammonium substrates using hydrogen peroxide as a benign terminal oxidant. (5−8) More recently, the solvophobic effect in highly polar fluorinated alcohols, a solvent of choice for C–H oxidations, (9,10) has been harnessed as a driving force for substrate binding, (11) a modality hitherto overlooked in catalyst-directed transformations. This strategy enabled the selective oxidation of alkanes at medial positions, (12) a breakthrough given that, even within the broader field of C–H functionalization, (13−15) site-selective derivatization of alkanes at medial sites has remained an unsolved challenge. (16−20) Supramolecular catalyst-directed oxidation therefore represents a powerful tool to expand the site-selectivity landscape of C–H oxidation, overcoming the inherent constraints of traditional catalysts, which typically address only a narrow subset of reactive sites. (1,21−24)
The concept of recognition-driven site-selectivity in C–H oxidation was pioneered by Breslow and co-workers, who employed porphyrin catalysts together with iodosobenzene, a less practical oxidant than hydrogen peroxide. (25,26) Its broader applicability, however, remained limited, as the substrates required at least two functional groups, which furthermore had to be covalently modified before the oxidation. Later studies by Crabtree and Brudvig (27,28) and by Bach, (29−31) succeeded in utilizing hydrogen bonds between substrate and catalyst for the regioselective and even enantioselective C–H oxidation but were restricted to activated benzylic C–H bonds.
Advances in the field have been impeded by the lengthy linear synthetic routes required to access nonheme C–H oxidation catalysts with defined supramolecular recognition motifs, which substantially limit systematic studies and broader applicability. The synthesis of supramolecular catalysts such as 1 (Figure 1a), featuring the tetradentate iron and manganese aminopyridine complexes that have emerged as superior catalysts for the oxidation of nonactivated C–H bonds, (1−4) relied exclusively on a catalyst-specific linear strategy. The tetradentate aminopyridine catalyst is constructed over four to five linear steps starting from the recognition motif 2, making the overall process labor-intensive and time-consuming. (5,7,12,32) In an ideal scenario, compound 2 would be directly cross-coupled to the preformed tetradentate ligand 3 (Figure 1b), enabling a more convergent synthetic strategy that allows rapid assembly of catalyst libraries from premade, interchangeable building blocks. Surprisingly, this approach has not been previously reported. Interestingly, during the preparation of this manuscript, Wang, Nam, and colleagues disclosed that anthracene appendages can be attached via such a strategy. (33) However, the reported conditions were inadequate for the installation of supramolecular recognition units such as resorcin[4]arene (RS) and calix[4]arene (CX) for the recognition of alkyl substrates. We surmise that this strategy was previously disregarded because tetradentate ligand 3 was expected to impede palladium-catalyzed coupling reactions through undesired coordination. Nevertheless, the approach proved viable, and we report its successful implementation.
Figure 1
Figure 1. (a) Traditional linear route to nonheme C–H oxidation catalysts bearing supramolecular recognition motifs. (b) This work: rapid, convergent synthesis of supramolecular C–H oxidation catalysts. Resorcin[4]arene (RS), calix[4]arene (CX).
In this study, the convergent syntheses of four tetradentate aminopyridine-based supramolecular C–H oxidation catalysts, the novel Mn(S,S-pdp)-RS2 (4, S,S-pdp = 2-({(S)-2-[(S)-1-(pyridin-2-ylmethyl)pyrrolidin-2-yl]pyrrolidin-1-yl}methyl)pyridine), Mn(S,S-pdp)-CX2 (5), Mn(S,S-mcp)-CX2 (6, S,S-mcp = N,N′-dimethyl-N,N′-bis(2-pyridylmethyl)-cyclohexane-1S,2S-diamine), and the previously disclosed Mn(S,S-mcp)-RS2 (7), (12) were developed (Scheme 1). The two-step synthesis of the common dibromo-backbone 8 and 9 commenced from commercially available (5-bromo-2-pyridinyl)methanol (10), which was first converted to the benzylic bromide 11 followed by alkylation of the commercially available diamine 12 or 13, respectively. The subsequent Suzuki cross-coupling had to be carefully optimized (see details below). Under optimized conditions, coupling with the recognition motifs BPin-RS 14 or BPin-CX 15 afforded the corresponding diRS- and diCX-functionalized aminopyridine ligands (step 1a: (S,S-pdp)-RS2 (16) and (S,S-pdp)-CX2 (17), step 1b: (S,S-mcp)-CX2 (18) and (S,S-mcp)-RS2 (19)). The supramolecular C–H oxidation catalysts 4–7 were obtained after coordination of the Mn(II)-metal center (steps 2a–d).
Scheme 1
The reaction conditions of the crucial cross-coupling step were first screened with the backbone (S,S-mcp)-diBr (9), and the findings are summarized in Table 1. Conditions analogous to those employed in our previous work for the linear installation of the pyridine moiety onto the RS recognition motif served as the starting point of this investigation (entry 1, NMR yield (Y) = 32% of diarylated product, determined by NMR with tetraethylsilane as an internal standard). (12) In the solvent mixture of dimethylformamide/water (9/1 v/v%) neither higher catalyst loading (entry 2) nor variation of the Pd catalyst (entry 3) led to an increase in yield of the desired diarylated product 19. The first notable hit was observed upon switching the solvent to a 9:1 mixture of 1,4-dioxane/water and replacing potassium carbonate with cesium carbonate as the base (entry 4, NMR Y = 59 ± 9.8% of 19, results from three experiments). Raising the temperature proved unfavorable, as it increased the formation of the defunctionalized side product SI-1 (see SI, Bpin of 14 → H, entries 5 and 6). Attempts to reduce the catalyst loading likewise resulted in lower yields of desired ligand 19 (entries 7 and 8). Applying the conditions of entry 4 to a 3-fold scale-up, the diRS-functionalized ligand 19 was isolated in 43% yield (NMR Y = 54%). The successful but challenging separation of 19 from triphenylphosphine oxide (TPPO) is described in the Supporting Information.
| Entry | Pd cat. (mol %) | Scale of 9 (mg) | Base (4.5 equiv) | Solvents (v/v %) | T (°C) | Diaryl. (NMR Y, %) | Monoaryl.b (NMR Y, %) | Isolated Y (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | Pd(PPh3)4 (17) | 5 | K2CO3 | DMF | 100 | 32 | 0 | - |
| 2 | Pd(PPh3)4 (20) | 5 | K2CO3 | DMF/H2O (9/1) | 100 | 34 | 0 | - |
| 3 | Pd(dppf)Cl2 (20) | 5 | K2CO3 | DMF/H2O (9/1) | 100 | 35 | 13 | - |
| 4 | Pd(PPh3)4 (20) | 5 | Cs2CO3 | dioxane/H2O (9/1) | 100 | 59 ± 9.8c | 0 | - |
| 5 | Pd(PPh3)4 (20) | 5 | Cs2CO3 | dioxane/H2O (9/1) | 105 | 31 | 13 | - |
| 6 | Pd(dppf)Cl2 (20) | 5 | Cs2CO3 | dioxane/H2O (9/1) | 105 | 40 | 0 | - |
| 7 | Pd(PPh3)4 (10) | 5 | Cs2CO3 | dioxane/H2O (9/1) | 100 | 5 | 18 | - |
| 8 | Pd(PPh3)4 (15) | 5 | Cs2CO3 | dioxane/H2O (9/1) | 100 | 23 | 24 | - |
| 9 | Pd(PPh3)4 (20) | 15 | Cs2CO3 | dioxane/H2O (9/1) | 100 | 54 | 0 | 43 |
3 day reaction time. Arylated (aryl.).
Monoaryl. species = (S,S-mcp)-RSBr (a RS unit appended on one pyridine moiety and a bromide remained on the other). Yield (Y).
Average of three experiments.
The optimized Suzuki cross-coupling conditions used to access ligand (S,S-mcp)-RS2 (19, Table 1, entry 4) were directly applied to the synthesis of (S,S-pdp)-RS2 (16) and to the installation of the calix[4]arene recognition motif 15, yielding ligands 17 and 18 in 43–60% yield (Scheme 1) without further condition optimization, highlighting the broad applicability of this convergent ligand-construction strategy.
With the three novel catalysts in hand, their performance was evaluated relative to that of the unsubstituted core catalysts 20 and 21 using four benchmark substrates (S1–4, Scheme 2). It is important to note that only the GC yields (Y) of the hydroxylated products and the corresponding ketones are given, and only these two products were taken into account in the study on the site-selectivity of the C–H oxidation reaction. Further details on the side products (epoxides and esters) were discussed in our previous work. (12) Using the previously reported conditions with the additive 2,2-dimethylpropanoic acid (2,2-diMe-PA) and the polar solvent 2,2,2-trifluoroethanol (TFE), (12) both RS-based supramolecular catalysts 4 and 7 (highlighted in blue) exhibit comparable conversion and yield and, most importantly, a similar preference for oxidation at the fifth carbon (red sphere), counted from the less hindered terminus of the substrate (gray dot). S3 represents the sole exception, where the more reactive tertiary C(3)–H (red dotted circle, 3 denotes the third position on the carbon framework) lies adjacent to the fifth-position methylene group. The results indicate that the site-selectivity (S), driven by the solvophobic RS recognition motifs, is largely independent of the two distinct cores (mcp and pdp). Both catalysts are capable of (1) differentiating between three chemically similar secondary C(sp3)–H bonds in S1, (2) overriding the intrinsic reactivity of C–H bonds in S2, (3) predominantly oxidizing the electronically deactivated proximal C(3)–H bond over the electron-rich distal C(7)–H on S3, and (4) favoring the sterically congested, tertiary C(4)–H bond in S4 that is hardly oxidized at all by the unsubstituted catalysts 20 and 21.
Scheme 2
a5.0 equiv of 2,2-DiMe-PA used.
b2.5 mol % of trifluoromethanesulfonic acid added.
cOxidation with 2,2-dimethylpropanoic acid (2,2-diMe-PA) in 2,2,2-trifluoroethanol (TFE); 2-iodoxybenzoic acid (IBX) oxidation was performed to facilitate the identification of the oxidation products. Site-selectivity (S) and conversion (conv.) were calculated based on remaining substrate. GC yield (Y) equals the sum of the alcohols or/and ketones.
As for the CX-appended catalysts 5 and 6 (marked in gray), interestingly the observed site-selectivity did not differ significantly from the one shown by the unsubstituted parent catalysts 20 and 21. Screening of additional carboxylic acid additives and more polar solvents likewise resulted in no improvement (SI Section 3.2). Notably, while both CX and RS macrocycles can freely rotate around the pyridine–aryl C–C bond, only the CX catalysts show no change in selectivity. This suggests that the observed behavior arises from inherent structural features of the cavitands rather than conformational flexibility. Specifically, the smaller and less preorganized cavity of the CX motif is expected to bind substrates more weakly than RS. In terms of catalytic activity, both CX catalysts require a second acid additive, trifluoromethanesulfonic acid (TfOH), to be active. This can be attributed to the reported ability of Brønsted acids to assist in the heterolytic cleavage of the O–O bond to form the strongly oxidizing high-valent metal oxo species. (34,35) Nevertheless, in the case of 5, the activity was still substantially lower compared to that of the RS counterpart 4 and to that of the parent 20. By reduction of the equivalents of the carboxylic acid additive from 22 to 5, catalyst 5 achieved comparable activity with substrate S2. However, the same amount of carboxylic acid did not lead to a higher activity in the instance of S3. On the other hand, the addition of TfOH aided catalyst 6 in attaining consistent good catalytic activity in comparison to 7 and 21.
In summary, we have developed a concise convergent strategy that allows rapid assembly of catalyst libraries using premade, interchangeable building blocks. This approach overcomes the limitations of previous linear, catalyst specific routes, streamlining the synthesis of catalysts such as Mn(S,S-pdp)-RS2 (4), Mn(S,S-pdp)-CX2 (5), Mn(S,S-mcp)-CX2 (6), and Mn(S,S-mcp)-RS2 (7). The screening results of the obtained novel catalysts indicated that the backbone (mcp or pdp) of the RS-based catalysts 4 and 7 does not influence the recognition-driven oxidation selectivity for the fifth position on alkyl substrates that greatly differs from the selectivity observed with unfunctionalized catalysts 20 and 21. Furthermore, we demonstrated that closely related catalysts featuring CX recognition motifs do not enable selective C–H oxidation. The convergent route developed in this study provides a broadly applicable platform for building diverse catalyst libraries. We expect this route to facilitate a systematic exploration of site-selective C–H oxidation across a wide range of substrates and catalysts.
Data Availability
The data underlying this study are available in the published article, in its Supporting Information, and openly available in Zenodo:18300467 at https://zenodo.org/records/18300467.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsorginorgau.6c00006.
Experimental procedures, analytical data of the compounds, GC- and GC-MS chromatograms, and NMR spectra (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We thank the National Centre of Competence in Research (NCCR) Molecular Systems Engineering (MSE) for financial support and Dr. Michael Pfeffer for the high-resolution mass spectrometry analysis.
References
This article references 35 other publications.
- 1White, M. C.; Zhao, J. Aliphatic C–H Oxidations for Late-Stage Functionalization. J. Am. Chem. Soc. 2018, 140 (43), 13988– 14009, DOI: 10.1021/jacs.8b05195Google ScholarThere is no corresponding record for this reference.
- 2Olivo, G.; Cussó, O.; Borrell, M.; Costas, M. Oxidation of Alkane and Alkene Moieties with Biologically Inspired Nonheme Iron Catalysts and Hydrogen Peroxide: From Free Radicals to Stereoselective Transformations. J. Biol. Inorg. Chem. 2017, 22 (2), 425– 452, DOI: 10.1007/s00775-016-1434-zGoogle ScholarThere is no corresponding record for this reference.
- 3Vicens, L.; Olivo, G.; Costas, M. Rational Design of Bioinspired Catalysts for Selective Oxidations. ACS Catal. 2020, 10 (15), 8611– 8631, DOI: 10.1021/acscatal.0c02073Google ScholarThere is no corresponding record for this reference.
- 4Talsi, E. P.; Bryliakov, K. P. Chemo- and Stereoselective CH Oxidations and Epoxidations/Cis-Dihydroxylations with H2O2, Catalyzed by Non-Heme Iron and Manganese Complexes. Coord. Chem. Rev. 2012, 256 (13), 1418– 1434, DOI: 10.1016/j.ccr.2012.04.005Google ScholarThere is no corresponding record for this reference.
- 5Olivo, G.; Farinelli, G.; Barbieri, A.; Lanzalunga, O.; Di Stefano, S.; Costas, M. Supramolecular Recognition Allows Remote, Site-Selective C–H Oxidation of Methylenic Sites in Linear Amines. Angew. Chem., Int. Ed. 2017, 56 (51), 16347– 16351, DOI: 10.1002/anie.201709280Google ScholarThere is no corresponding record for this reference.
- 6Olivo, G.; Capocasa, G.; Ticconi, B.; Lanzalunga, O.; Di Stefano, S.; Costas, M. Predictable Selectivity in Remote C–H Oxidation of Steroids: Analysis of Substrate Binding Mode. Angew. Chem., Int. Ed. 2020, 59 (31), 12703– 12708, DOI: 10.1002/anie.202003078Google ScholarThere is no corresponding record for this reference.
- 7Knezevic, M.; Heilmann, M.; Piccini, G. M.; Tiefenbacher, K. Overriding Intrinsic Reactivity in Aliphatic C–H Oxidation: Preferential C3/C4 Oxidation of Aliphatic Ammonium Substrates. Angew. Chem., Int. Ed. 2020, 59 (30), 12387– 12391, DOI: 10.1002/anie.202004242Google ScholarThere is no corresponding record for this reference.
- 8Knezevic, M.; Tiefenbacher, K. Tweezer-Based C–H Oxidation Catalysts Overriding the Intrinsic Reactivity of Aliphatic Ammonium Substrates. Chem. Eur. J. 2023, 29 (13), e202203480 DOI: 10.1002/chem.202203480Google ScholarThere is no corresponding record for this reference.
- 9Dantignana, V.; Milan, M.; Cussó, O.; Company, A.; Bietti, M.; Costas, M. Chemoselective Aliphatic C–H Bond Oxidation Enabled by Polarity Reversal. ACS Cent. Sci. 2017, 3 (12), 1350– 1358, DOI: 10.1021/acscentsci.7b00532Google ScholarThere is no corresponding record for this reference.
- 10Bietti, M. Activation and Deactivation Strategies Promoted by Medium Effects for Selective Aliphatic C–H Bond Functionalization. Angew. Chem., Int. Ed. 2018, 57 (51), 16618– 16637, DOI: 10.1002/anie.201804929Google ScholarThere is no corresponding record for this reference.
- 11Smithrud, D. B.; Diederich, F. Strength of Molecular Complexation of Apolar Solutes in Water and in Organic Solvents Is Predictable by Linear Free Energy Relationships: A General Model for Solvation Effects on Apolar Binding. J. Am. Chem. Soc. 1990, 112 (1), 339, DOI: 10.1021/ja00157a052Google ScholarThere is no corresponding record for this reference.
- 12Lu, Y.; Knezevic, M.; Prescimone, A.; Goldfuss, B.; Tiefenbacher, K. Site-Selective C(Sp3)–H Oxidation of Alkyl Substrates Devoid of Functional Handles. Chem. 2025, 11 (3), 102442 DOI: 10.1016/j.chempr.2025.102442Google ScholarThere is no corresponding record for this reference.
- 13Yamaguchi, J.; Yamaguchi, A. D.; Itami, K. C-H Bond Functionalization: Emerging Synthetic Tools for Natural Products and Pharmaceuticals. Angew. Chem., Int. Ed. 2012, 51 (36), 8960– 9009, DOI: 10.1002/anie.201201666Google ScholarThere is no corresponding record for this reference.
- 14Davies, H. M. L.; Morton, D. Recent Advances in C–H Functionalization. J. Org. Chem. 2016, 81 (2), 343– 350, DOI: 10.1021/acs.joc.5b02818Google ScholarThere is no corresponding record for this reference.
- 15Rogge, T.; Kaplaneris, N.; Chatani, N.; Kim, J.; Chang, S.; Punji, B.; Schafer, L. L.; Musaev, D. G.; Wencel-Delord, J.; Roberts, C. A.; Sarpong, R.; Wilson, Z. E.; Brimble, M. A.; Johansson, M. J.; Ackermann, L. C–H Activation. Nat. Rev. Methods Primers 2021, 1 (1), 1– 31, DOI: 10.1038/s43586-021-00041-2Google ScholarThere is no corresponding record for this reference.
- 16Tang, X.; Jia, X.; Huang, Z. Challenges and Opportunities for Alkane Functionalisation Using Molecular Catalysts. Chem. Sci. 2018, 9 (2), 288– 299, DOI: 10.1039/C7SC03610HGoogle ScholarThere is no corresponding record for this reference.
- 17Hartwig, J. F. Borylation and Silylation of C–H Bonds: A Platform for Diverse C–H Bond Functionalizations. Acc. Chem. Res. 2012, 45 (6), 864– 873, DOI: 10.1021/ar200206aGoogle ScholarThere is no corresponding record for this reference.
- 18Liao, K.; Negretti, S.; Musaev, D. G.; Bacsa, J.; Davies, H. M. L. Site-Selective and Stereoselective Functionalization of Unactivated C–H Bonds. Nature 2016, 533 (7602), 230– 234, DOI: 10.1038/nature17651Google ScholarThere is no corresponding record for this reference.
- 19Liao, K.; Yang, Y.-F.; Li, Y.; Sanders, J. N.; Houk, K. N.; Musaev, D. G.; Davies, H. M. L. Design of Catalysts for Site-Selective and Enantioselective Functionalization of Non-Activated Primary C–H Bonds. Nat. Chem. 2018, 10 (10), 1048– 1055, DOI: 10.1038/s41557-018-0087-7Google ScholarThere is no corresponding record for this reference.
- 20Jia, X.; Huang, Z. Conversion of Alkanes to Linear Alkylsilanes Using an Iridium–Iron-Catalysed Tandem Dehydrogenation–Isomerization–Hydrosilylation. Nat. Chem. 2016, 8 (2), 157– 161, DOI: 10.1038/nchem.2417Google ScholarThere is no corresponding record for this reference.
- 21Curci, R.; D’Accolti, L.; Fusco, C. A Novel Approach to the Efficient Oxygenation of Hydrocarbons under Mild Conditions. Superior Oxo Transfer Selectivity Using Dioxiranes. Acc. Chem. Res. 2006, 39 (1), 1– 9, DOI: 10.1021/ar050163yGoogle ScholarThere is no corresponding record for this reference.
- 22Newhouse, T.; Baran, P. S. If C-H Bonds Could Talk: Selective C-H Bond Oxidation. Angew. Chem., Int. Ed. 2011, 50 (15), 3362– 3374, DOI: 10.1002/anie.201006368Google ScholarThere is no corresponding record for this reference.
- 23Gormisky, P. E.; White, M. C. Catalyst-Controlled Aliphatic C–H Oxidations with a Predictive Model for Site-Selectivity. J. Am. Chem. Soc. 2013, 135 (38), 14052– 14055, DOI: 10.1021/ja407388yGoogle ScholarThere is no corresponding record for this reference.
- 24D’Accolti, L.; Annese, C.; Fusco, C. Continued Progress towards Efficient Functionalization of Natural and Non-Natural Targets under Mild Conditions: Oxygenation by C–H Bond Activation with Dioxirane. Chem. Eur. J. 2019, 25 (52), 12003– 12017, DOI: 10.1002/chem.201901687Google ScholarThere is no corresponding record for this reference.
- 25Breslow, R.; Gabriele, B.; Yang, J. Geometrically Directed Selective Steroid Hydroxylation with High Turnover by a Fluorinated Artificial Cytochrome P-450. Tetrahedron Lett. 1998, 39 (19), 2887– 2890, DOI: 10.1016/S0040-4039(98)00425-0Google ScholarThere is no corresponding record for this reference.
- 26Breslow, R.; Dong, S. D. Biomimetic Reactions Catalyzed by Cyclodextrins and Their Derivatives. Chem. Rev. 1998, 98 (5), 1997– 2012, DOI: 10.1021/cr970011jGoogle ScholarThere is no corresponding record for this reference.
- 27Das, S.; Incarvito, C. D.; Crabtree, R. H.; Brudvig, G. W. Molecular Recognition in the Selective Oxygenation of Saturated C-H Bonds by a Dimanganese Catalyst. Science 2006, 312 (5782), 1941– 1943, DOI: 10.1126/science.1127899Google ScholarThere is no corresponding record for this reference.
- 28Das, S.; Brudvig, G. W.; Crabtree, R. H. High Turnover Remote Catalytic Oxygenation of Alkyl Groups: How Steric Exclusion of Unbound Substrate Contributes to High Molecular Recognition Selectivity. J. Am. Chem. Soc. 2008, 130 (5), 1628– 1637, DOI: 10.1021/ja076039mGoogle ScholarThere is no corresponding record for this reference.
- 29Frost, J. R.; Huber, S. M.; Breitenlechner, S.; Bannwarth, C.; Bach, T. Enantiotopos-Selective C-H Oxygenation Catalyzed by a Supramolecular Ruthenium Complex. Angew. Chem., Int. Ed. 2015, 54 (2), 691– 695, DOI: 10.1002/anie.201409224Google ScholarThere is no corresponding record for this reference.
- 30Burg, F.; Gicquel, M.; Breitenlechner, S.; Pöthig, A.; Bach, T. Site- and Enantioselective C–H Oxygenation Catalyzed by a Chiral Manganese Porphyrin Complex with a Remote Binding Site. Angew. Chem., Int. Ed. 2018, 57 (11), 2953– 2957, DOI: 10.1002/anie.201712340Google ScholarThere is no corresponding record for this reference.
- 31Burg, F.; Breitenlechner, S.; Jandl, C.; Bach, T. Enantioselective Oxygenation of Exocyclic Methylene Groups by a Manganese Porphyrin Catalyst with a Chiral Recognition Site. Chem. Sci. 2020, 11 (8), 2121– 2129, DOI: 10.1039/C9SC06089HGoogle ScholarThere is no corresponding record for this reference.
- 32Vidal, D.; Costas, M.; Lledó, A. A Deep Cavitand Receptor Functionalized with Fe(II) and Mn(II) Aminopyridine Complexes for Bioinspired Oxidation Catalysis. ACS Catal. 2018, 8 (4), 3667– 3672, DOI: 10.1021/acscatal.7b04426Google ScholarThere is no corresponding record for this reference.
- 33Wu, C.; Wang, S.; Sun, D.; Chen, J.; Ji, W.; Wang, Y.; Nam, W.; Wang, B. Nonheme Manganese-Catalyzed Oxidative N-Dealkylation of Tertiary Amides: Manganese(IV)-Oxo Aminopyridine Cation Radical Species and Hydride Transfer Mechanism. J. Am. Chem. Soc. 2025, 147 (13), 11432– 11445, DOI: 10.1021/jacs.5c01391Google ScholarThere is no corresponding record for this reference.
- 34Miao, C.; Wang, B.; Wang, Y.; Xia, C.; Lee, Y.-M.; Nam, W.; Sun, W. Proton-Promoted and Anion-Enhanced Epoxidation of Olefins by Hydrogen Peroxide in the Presence of Nonheme Manganese Catalysts. J. Am. Chem. Soc. 2016, 138 (3), 936– 943, DOI: 10.1021/jacs.5b11579Google ScholarThere is no corresponding record for this reference.
- 35Serrano-Plana, J.; Acuña-Parés, F.; Dantignana, V.; Oloo, W. N.; Castillo, E.; Draksharapu, A.; Whiteoak, C. J.; Martin-Diaconescu, V.; Basallote, M. G.; Luis, J. M.; Que, L., Jr.; Costas, M.; Company, A. Acid-Triggered O–O Bond Heterolysis of a Nonheme FeIII(OOH) Species for the Stereospecific Hydroxylation of Strong C–H Bonds. Chem. Eur. J. 2018, 24 (20), 5331– 5340, DOI: 10.1002/chem.201704851Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
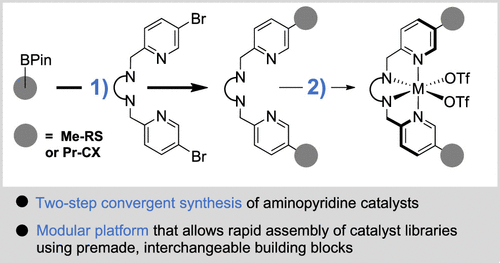
Figure 1
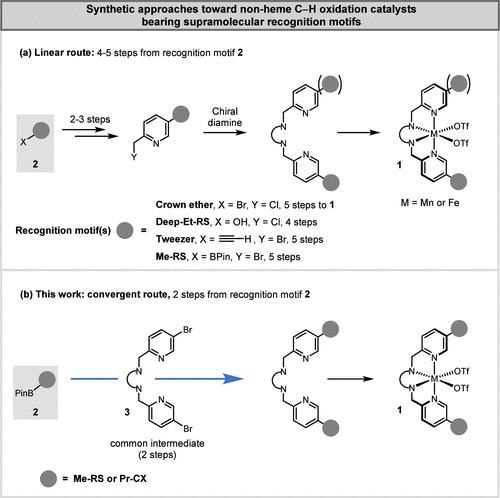
Figure 1. (a) Traditional linear route to nonheme C–H oxidation catalysts bearing supramolecular recognition motifs. (b) This work: rapid, convergent synthesis of supramolecular C–H oxidation catalysts. Resorcin[4]arene (RS), calix[4]arene (CX).
Scheme 1
 Scheme 1. Convergent Synthesis Route to Construct Supramolecular Catalysts 4–7
Scheme 1. Convergent Synthesis Route to Construct Supramolecular Catalysts 4–7Scheme 2
 Scheme 2. Comparison of Catalyst-Directed Site-Selectivity in C–H Oxidation of Selected Substratesc
Scheme 2. Comparison of Catalyst-Directed Site-Selectivity in C–H Oxidation of Selected Substratesca5.0 equiv of 2,2-DiMe-PA used.
b2.5 mol % of trifluoromethanesulfonic acid added.
cOxidation with 2,2-dimethylpropanoic acid (2,2-diMe-PA) in 2,2,2-trifluoroethanol (TFE); 2-iodoxybenzoic acid (IBX) oxidation was performed to facilitate the identification of the oxidation products. Site-selectivity (S) and conversion (conv.) were calculated based on remaining substrate. GC yield (Y) equals the sum of the alcohols or/and ketones.
References
This article references 35 other publications.
- 1White, M. C.; Zhao, J. Aliphatic C–H Oxidations for Late-Stage Functionalization. J. Am. Chem. Soc. 2018, 140 (43), 13988– 14009, DOI: 10.1021/jacs.8b05195There is no corresponding record for this reference.
- 2Olivo, G.; Cussó, O.; Borrell, M.; Costas, M. Oxidation of Alkane and Alkene Moieties with Biologically Inspired Nonheme Iron Catalysts and Hydrogen Peroxide: From Free Radicals to Stereoselective Transformations. J. Biol. Inorg. Chem. 2017, 22 (2), 425– 452, DOI: 10.1007/s00775-016-1434-zThere is no corresponding record for this reference.
- 3Vicens, L.; Olivo, G.; Costas, M. Rational Design of Bioinspired Catalysts for Selective Oxidations. ACS Catal. 2020, 10 (15), 8611– 8631, DOI: 10.1021/acscatal.0c02073There is no corresponding record for this reference.
- 4Talsi, E. P.; Bryliakov, K. P. Chemo- and Stereoselective CH Oxidations and Epoxidations/Cis-Dihydroxylations with H2O2, Catalyzed by Non-Heme Iron and Manganese Complexes. Coord. Chem. Rev. 2012, 256 (13), 1418– 1434, DOI: 10.1016/j.ccr.2012.04.005There is no corresponding record for this reference.
- 5Olivo, G.; Farinelli, G.; Barbieri, A.; Lanzalunga, O.; Di Stefano, S.; Costas, M. Supramolecular Recognition Allows Remote, Site-Selective C–H Oxidation of Methylenic Sites in Linear Amines. Angew. Chem., Int. Ed. 2017, 56 (51), 16347– 16351, DOI: 10.1002/anie.201709280There is no corresponding record for this reference.
- 6Olivo, G.; Capocasa, G.; Ticconi, B.; Lanzalunga, O.; Di Stefano, S.; Costas, M. Predictable Selectivity in Remote C–H Oxidation of Steroids: Analysis of Substrate Binding Mode. Angew. Chem., Int. Ed. 2020, 59 (31), 12703– 12708, DOI: 10.1002/anie.202003078There is no corresponding record for this reference.
- 7Knezevic, M.; Heilmann, M.; Piccini, G. M.; Tiefenbacher, K. Overriding Intrinsic Reactivity in Aliphatic C–H Oxidation: Preferential C3/C4 Oxidation of Aliphatic Ammonium Substrates. Angew. Chem., Int. Ed. 2020, 59 (30), 12387– 12391, DOI: 10.1002/anie.202004242There is no corresponding record for this reference.
- 8Knezevic, M.; Tiefenbacher, K. Tweezer-Based C–H Oxidation Catalysts Overriding the Intrinsic Reactivity of Aliphatic Ammonium Substrates. Chem. Eur. J. 2023, 29 (13), e202203480 DOI: 10.1002/chem.202203480There is no corresponding record for this reference.
- 9Dantignana, V.; Milan, M.; Cussó, O.; Company, A.; Bietti, M.; Costas, M. Chemoselective Aliphatic C–H Bond Oxidation Enabled by Polarity Reversal. ACS Cent. Sci. 2017, 3 (12), 1350– 1358, DOI: 10.1021/acscentsci.7b00532There is no corresponding record for this reference.
- 10Bietti, M. Activation and Deactivation Strategies Promoted by Medium Effects for Selective Aliphatic C–H Bond Functionalization. Angew. Chem., Int. Ed. 2018, 57 (51), 16618– 16637, DOI: 10.1002/anie.201804929There is no corresponding record for this reference.
- 11Smithrud, D. B.; Diederich, F. Strength of Molecular Complexation of Apolar Solutes in Water and in Organic Solvents Is Predictable by Linear Free Energy Relationships: A General Model for Solvation Effects on Apolar Binding. J. Am. Chem. Soc. 1990, 112 (1), 339, DOI: 10.1021/ja00157a052There is no corresponding record for this reference.
- 12Lu, Y.; Knezevic, M.; Prescimone, A.; Goldfuss, B.; Tiefenbacher, K. Site-Selective C(Sp3)–H Oxidation of Alkyl Substrates Devoid of Functional Handles. Chem. 2025, 11 (3), 102442 DOI: 10.1016/j.chempr.2025.102442There is no corresponding record for this reference.
- 13Yamaguchi, J.; Yamaguchi, A. D.; Itami, K. C-H Bond Functionalization: Emerging Synthetic Tools for Natural Products and Pharmaceuticals. Angew. Chem., Int. Ed. 2012, 51 (36), 8960– 9009, DOI: 10.1002/anie.201201666There is no corresponding record for this reference.
- 14Davies, H. M. L.; Morton, D. Recent Advances in C–H Functionalization. J. Org. Chem. 2016, 81 (2), 343– 350, DOI: 10.1021/acs.joc.5b02818There is no corresponding record for this reference.
- 15Rogge, T.; Kaplaneris, N.; Chatani, N.; Kim, J.; Chang, S.; Punji, B.; Schafer, L. L.; Musaev, D. G.; Wencel-Delord, J.; Roberts, C. A.; Sarpong, R.; Wilson, Z. E.; Brimble, M. A.; Johansson, M. J.; Ackermann, L. C–H Activation. Nat. Rev. Methods Primers 2021, 1 (1), 1– 31, DOI: 10.1038/s43586-021-00041-2There is no corresponding record for this reference.
- 16Tang, X.; Jia, X.; Huang, Z. Challenges and Opportunities for Alkane Functionalisation Using Molecular Catalysts. Chem. Sci. 2018, 9 (2), 288– 299, DOI: 10.1039/C7SC03610HThere is no corresponding record for this reference.
- 17Hartwig, J. F. Borylation and Silylation of C–H Bonds: A Platform for Diverse C–H Bond Functionalizations. Acc. Chem. Res. 2012, 45 (6), 864– 873, DOI: 10.1021/ar200206aThere is no corresponding record for this reference.
- 18Liao, K.; Negretti, S.; Musaev, D. G.; Bacsa, J.; Davies, H. M. L. Site-Selective and Stereoselective Functionalization of Unactivated C–H Bonds. Nature 2016, 533 (7602), 230– 234, DOI: 10.1038/nature17651There is no corresponding record for this reference.
- 19Liao, K.; Yang, Y.-F.; Li, Y.; Sanders, J. N.; Houk, K. N.; Musaev, D. G.; Davies, H. M. L. Design of Catalysts for Site-Selective and Enantioselective Functionalization of Non-Activated Primary C–H Bonds. Nat. Chem. 2018, 10 (10), 1048– 1055, DOI: 10.1038/s41557-018-0087-7There is no corresponding record for this reference.
- 20Jia, X.; Huang, Z. Conversion of Alkanes to Linear Alkylsilanes Using an Iridium–Iron-Catalysed Tandem Dehydrogenation–Isomerization–Hydrosilylation. Nat. Chem. 2016, 8 (2), 157– 161, DOI: 10.1038/nchem.2417There is no corresponding record for this reference.
- 21Curci, R.; D’Accolti, L.; Fusco, C. A Novel Approach to the Efficient Oxygenation of Hydrocarbons under Mild Conditions. Superior Oxo Transfer Selectivity Using Dioxiranes. Acc. Chem. Res. 2006, 39 (1), 1– 9, DOI: 10.1021/ar050163yThere is no corresponding record for this reference.
- 22Newhouse, T.; Baran, P. S. If C-H Bonds Could Talk: Selective C-H Bond Oxidation. Angew. Chem., Int. Ed. 2011, 50 (15), 3362– 3374, DOI: 10.1002/anie.201006368There is no corresponding record for this reference.
- 23Gormisky, P. E.; White, M. C. Catalyst-Controlled Aliphatic C–H Oxidations with a Predictive Model for Site-Selectivity. J. Am. Chem. Soc. 2013, 135 (38), 14052– 14055, DOI: 10.1021/ja407388yThere is no corresponding record for this reference.
- 24D’Accolti, L.; Annese, C.; Fusco, C. Continued Progress towards Efficient Functionalization of Natural and Non-Natural Targets under Mild Conditions: Oxygenation by C–H Bond Activation with Dioxirane. Chem. Eur. J. 2019, 25 (52), 12003– 12017, DOI: 10.1002/chem.201901687There is no corresponding record for this reference.
- 25Breslow, R.; Gabriele, B.; Yang, J. Geometrically Directed Selective Steroid Hydroxylation with High Turnover by a Fluorinated Artificial Cytochrome P-450. Tetrahedron Lett. 1998, 39 (19), 2887– 2890, DOI: 10.1016/S0040-4039(98)00425-0There is no corresponding record for this reference.
- 26Breslow, R.; Dong, S. D. Biomimetic Reactions Catalyzed by Cyclodextrins and Their Derivatives. Chem. Rev. 1998, 98 (5), 1997– 2012, DOI: 10.1021/cr970011jThere is no corresponding record for this reference.
- 27Das, S.; Incarvito, C. D.; Crabtree, R. H.; Brudvig, G. W. Molecular Recognition in the Selective Oxygenation of Saturated C-H Bonds by a Dimanganese Catalyst. Science 2006, 312 (5782), 1941– 1943, DOI: 10.1126/science.1127899There is no corresponding record for this reference.
- 28Das, S.; Brudvig, G. W.; Crabtree, R. H. High Turnover Remote Catalytic Oxygenation of Alkyl Groups: How Steric Exclusion of Unbound Substrate Contributes to High Molecular Recognition Selectivity. J. Am. Chem. Soc. 2008, 130 (5), 1628– 1637, DOI: 10.1021/ja076039mThere is no corresponding record for this reference.
- 29Frost, J. R.; Huber, S. M.; Breitenlechner, S.; Bannwarth, C.; Bach, T. Enantiotopos-Selective C-H Oxygenation Catalyzed by a Supramolecular Ruthenium Complex. Angew. Chem., Int. Ed. 2015, 54 (2), 691– 695, DOI: 10.1002/anie.201409224There is no corresponding record for this reference.
- 30Burg, F.; Gicquel, M.; Breitenlechner, S.; Pöthig, A.; Bach, T. Site- and Enantioselective C–H Oxygenation Catalyzed by a Chiral Manganese Porphyrin Complex with a Remote Binding Site. Angew. Chem., Int. Ed. 2018, 57 (11), 2953– 2957, DOI: 10.1002/anie.201712340There is no corresponding record for this reference.
- 31Burg, F.; Breitenlechner, S.; Jandl, C.; Bach, T. Enantioselective Oxygenation of Exocyclic Methylene Groups by a Manganese Porphyrin Catalyst with a Chiral Recognition Site. Chem. Sci. 2020, 11 (8), 2121– 2129, DOI: 10.1039/C9SC06089HThere is no corresponding record for this reference.
- 32Vidal, D.; Costas, M.; Lledó, A. A Deep Cavitand Receptor Functionalized with Fe(II) and Mn(II) Aminopyridine Complexes for Bioinspired Oxidation Catalysis. ACS Catal. 2018, 8 (4), 3667– 3672, DOI: 10.1021/acscatal.7b04426There is no corresponding record for this reference.
- 33Wu, C.; Wang, S.; Sun, D.; Chen, J.; Ji, W.; Wang, Y.; Nam, W.; Wang, B. Nonheme Manganese-Catalyzed Oxidative N-Dealkylation of Tertiary Amides: Manganese(IV)-Oxo Aminopyridine Cation Radical Species and Hydride Transfer Mechanism. J. Am. Chem. Soc. 2025, 147 (13), 11432– 11445, DOI: 10.1021/jacs.5c01391There is no corresponding record for this reference.
- 34Miao, C.; Wang, B.; Wang, Y.; Xia, C.; Lee, Y.-M.; Nam, W.; Sun, W. Proton-Promoted and Anion-Enhanced Epoxidation of Olefins by Hydrogen Peroxide in the Presence of Nonheme Manganese Catalysts. J. Am. Chem. Soc. 2016, 138 (3), 936– 943, DOI: 10.1021/jacs.5b11579There is no corresponding record for this reference.
- 35Serrano-Plana, J.; Acuña-Parés, F.; Dantignana, V.; Oloo, W. N.; Castillo, E.; Draksharapu, A.; Whiteoak, C. J.; Martin-Diaconescu, V.; Basallote, M. G.; Luis, J. M.; Que, L., Jr.; Costas, M.; Company, A. Acid-Triggered O–O Bond Heterolysis of a Nonheme FeIII(OOH) Species for the Stereospecific Hydroxylation of Strong C–H Bonds. Chem. Eur. J. 2018, 24 (20), 5331– 5340, DOI: 10.1002/chem.201704851There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsorginorgau.6c00006.
Experimental procedures, analytical data of the compounds, GC- and GC-MS chromatograms, and NMR spectra (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



