Computed Tomography Imaging and Characteristics of In Situ Forming Implants with Different PLGA EndcapsClick to copy article linkArticle link copied!
- Xinhao LinXinhao LinDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Xinhao Lin
- Zixuan ZhenZixuan ZhenDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Zixuan Zhen
- Seyyed Majid EslamiSeyyed Majid EslamiDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Seyyed Majid Eslami
- Nour Al ZouabiNour Al ZouabiDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Nour Al Zouabi
- Lauren Elizabeth WardLauren Elizabeth WardDepartment of Radiology, Biomedical Research Imaging Center, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, United StatesMore by Lauren Elizabeth Ward
- Mittal DarjiMittal DarjiDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Mittal Darji
- Sheyda RanjbarSheyda RanjbarDepartment of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Sheyda Ranjbar
- Francis K. MaseseFrancis K. MaseseDepartment of Chemistry, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Francis K. Masese
- André O’Reilly BeringhsAndré O’Reilly BeringhsOffice of Research and Standards, Office of Generic Drugs, Center for Drug Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland 20903-1058, United StatesMore by André O’Reilly Beringhs
- Rajeswari M. KasiRajeswari M. KasiDepartment of Chemistry, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Rajeswari M. Kasi
- Qiangnan ZhangQiangnan ZhangOffice of Research and Standards, Office of Generic Drugs, Center for Drug Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland 20903-1058, United StatesMore by Qiangnan Zhang
- Qi LiQi LiOffice of Research and Standards, Office of Generic Drugs, Center for Drug Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland 20903-1058, United StatesMore by Qi Li
- Qin BinQin BinOffice of Research and Standards, Office of Generic Drugs, Center for Drug Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland 20903-1058, United StatesMore by Qin Bin
- Yan WangYan WangOffice of Research and Standards, Office of Generic Drugs, Center for Drug Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland 20903-1058, United StatesMore by Yan Wang
- Hong YuanHong YuanDepartment of Radiology, Biomedical Research Imaging Center, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, United StatesMore by Hong Yuan
- Xiuling Lu*Xiuling Lu*Email: [email protected]Department of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut 06269, United StatesMore by Xiuling Lu
Abstract
A poly(lactic-co-glycolic acid) (PLGA)-based in situ-forming implant (ISFI) is a long-acting injectable composed of active ingredients, a biodegradable polymer and a biocompatible solvent. Understanding the role of PLGA end-caps is crucial for designing ISFI formulations due to their impact on drug delivery performance. In this study, using leuprolide acetate as a model drug, we characterized the ISFIs using computed tomography (CT) imaging to explore the influence of PLGA end-cap on the implant’s formation and drug-release behavior. CT imaging enabled a detailed characterization of implant morphology and internal structure, providing new insights into the relationship between phase inversion, implant formation, and in vitro release performance. Acid-ended PLGA showed a higher initial burst release with 100% of the solvent released at 9 days and a faster release duration of the drug in the in vitro release profile. In contrast, ester-ended PLGA showed a more prolonged release profile, with the plateau phase not reached until approximately day 50. Morphological analysis from CT images of the in vitro implants revealed that acid-ended PLGA formed spherical implants with a dense outer layer, whereas ester-ended PLGA resulted in irregular shapes with a larger size expansion. In vivo CT imaging confirmed these trends, although implant evolution occurred more rapidly in biological environments. Overall, this research highlights the impact of PLGA end-caps on the performance of ISFI, providing a scientific basis for formulation development, evaluation, and optimization of ISFIs.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of ISFI Formulations
2.3. Nuclear Magnetic Resonance
2.4. High-Performance Liquid Chromatography
2.5. X-ray CT Imaging of In Vitro-Formed Implants
2.6. In Vitro-Formed Implant Volume and Intensity Measurement
2.7. In Vitro Release Study
2.8. Scanning Electron Microscopy of In Vitro-Formed Implants
2.9. Porosity Measurement
2.10. Gel Permeation Chromatography
2.11. X-ray CT Imaging of In Vivo-Formed Implants
2.12. Statistical Analysis
3. Results and Discussion
3.1. Characterization of PLGA
3.1.1. End-Cap
Figure 1
Figure 1. Comparison of 13C NMR spectra of the PLGA with different end-caps. The peak appearing at 14 ppm was used as the criterion to determine the end-cap of the PLGA.
3.1.2. MW, L/G Ratio, and Blockiness
| MW (kDa) | L/G ratio | ||||
|---|---|---|---|---|---|
| endcap | reported | tested | reported | tested | blockiness (Rc) |
| acid | 25.8 | 32.71 ± 0.16 | 50:50 | 48:52 | 1.527 |
| ester | - | 30.59 ± 1.14 | 53:47 | 51:49 | 1.263 |
Figure 2
Figure 2. 1H and 13C NMR spectra of the PLGA with different end-caps. 1H NMR spectra of PLGA with acid end-caps (a) and ester end-caps (b) were used to determine the L/G ratio of the PLGA. The peaks appearing at 166.5 ppm on 13C NMR spectra of PLGA with acid end-caps (c) and ester end-caps (d) were used to determine the blockiness of the PLGA.
3.2. In Vitro Release Tests
Figure 3
Figure 3. In vitro release test of ISFIs. No drug: placebo formulation, LA: formulation with leuprolide acetate. LA and iohexol: formulation with both leuprolide acetate and iohexol. Cumulative release of NMP from the formulations with no drug (a), leuprolide acetate (b), and both leuprolide acetate and iohexol (c). Cumulative release of leuprolide acetate from the formulations with leuprolide acetate (d) and both leuprolide acetate and iohexol (e). Cumulative release of iohexol from the formulation with both leuprolide acetate and iohexol (f). All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
3.3. CT Imaging of In Vitro-Formed Implants
Figure 4
Figure 4. CT images of in vitro-formed implants. Representative transaxial sections of the 3D CT images collected for each implant at each time point (n = 3 per formulation). CT images of in vitro-formed implants from the formulations with no drug (a), only leuprolide acetate (b), and both leuprolide acetate and iohexol (c), PLGA, solvent (NMP), and water (d).
3.4. Sizes and Densities of the In Vitro-Formed Implants
Figure 5
Figure 5. Volumes and weights of in vitro-formed implants. Volumes and weights of in vitro-formed implants with no drug (a), only leuprolide acetate (b), and both leuprolide acetate and iohexol (c). All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
3.5. Polymer Degradation of In Vitro-Formed Implants
Figure 6
Figure 6. MWs change of the PLGA from in vitro-formed implants. Acid and ester represent the end-cap of PLGA. No drug: placebo formulation, LA: formulation with only leuprolide acetate. Iohexol and LA: formulation with both iohexol and leuprolide acetate. All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
3.6. Microstructure of In Vitro-Formed Implants
Figure 7
Figure 7. SEM images of in vitro-formed implants. Representative SEM images of the core and the shell of in vitro-formed implants in PBS after 7, 15, and 21 days of incubation. Representative images are shown, the regions of core and edge of each implant were scanned, three different implants were analyzed for each time point and formulation. (a) SEM images of the core of the polymer matrix. (b) SEM images of the edge of the polymer matrix. The scale bar represents 100 μm.
Figure 8
Figure 8. Porosity of the microstructure of in vitro-formed implants. Porosity of the microstructure was quantified by ImageJ. Porosity of the core and the edge of in vitro-formed implants after 7 (a), 15 (b), and 21 days (c) are shown in the figure (“*” means P < 0.05).
3.7. CT Images of In Vivo-Formed Implants
Figure 9
Figure 9. CT images of in vivo-formed implants. Representative coronal sections of the 3D CT images were collected for each implant at each time point (n = 5 per formulation). Iohexol: formulation with only iohexol, iohexol and LA: formulation with both iohexol and leuprolide acetate.
4. Conclusions
Data Availability
The data that support the findings of this study are available upon request.
Acknowledgments
The authors acknowledge the U.S. Food and Drug Administration for financial support of this research (contract number: 75F40120C00136).
References
This article references 26 other publications.
- 1O’Brien, M. N.; Jiang, W.; Wang, Y.; Loffredo, D. M. Challenges and opportunities in the development of complex generic long-acting injectable drug products. J. Controlled Release 2021, 336, 144– 158, DOI: 10.1016/j.jconrel.2021.06.017Google ScholarThere is no corresponding record for this reference.
- 2Thakur, R. R.; McMillan, H. L.; Jones, D. S. Solvent induced phase inversion-based in situ forming controlled release drug delivery implants. J. Controlled Release 2014, 176, 8– 23, DOI: 10.1016/j.jconrel.2013.12.020Google ScholarThere is no corresponding record for this reference.
- 3Benhabbour, S. R. Ultra-long-acting tunable biodegradable and removable controlled release implants for drug delivery. Nat. Commun. 2019, 10, 4324, DOI: 10.1038/s41467-019-12141-5Google ScholarThere is no corresponding record for this reference.
- 4Wang, X.; Burgess, D. J. Drug release from in situ forming implants and advances in release testing. Adv. Drug Delivery Rev. 2021, 178, 113912, DOI: 10.1016/j.addr.2021.113912Google ScholarThere is no corresponding record for this reference.
- 5Park, K. PLGA-based long-acting injectable (LAI) formulations. J. Controlled Release 2025, 382, 113758, DOI: 10.1016/j.jconrel.2025.113758Google ScholarThere is no corresponding record for this reference.
- 6Ibrahim, T. M.; El-Megrab, N. A.; El-Nahas, H. M. An overview of PLGA in-situ forming implants based on solvent exchange technique: effect of formulation components and characterization. Pharm. Dev. Technol. 2021, 26, 709– 728, DOI: 10.1080/10837450.2021.1944207Google ScholarThere is no corresponding record for this reference.
- 7Parent, M. PLGA in situ implants formed by phase inversion: critical physicochemical parameters to modulate drug release. J. Controlled Release 2013, 172, 292– 304, DOI: 10.1016/j.jconrel.2013.08.024Google ScholarThere is no corresponding record for this reference.
- 8Kempe, S.; Mader, K. In situ forming implants - an attractive formulation principle for parenteral depot formulations. J. Controlled Release 2012, 161, 668– 679, DOI: 10.1016/j.jconrel.2012.04.016Google ScholarThere is no corresponding record for this reference.
- 9Suh, M. S. Effect of implant formation on drug release kinetics of in situ forming implants. Int. J. Pharm. 2021, 592, 120105, DOI: 10.1016/j.ijpharm.2020.120105Google ScholarThere is no corresponding record for this reference.
- 10Sophocleous, A. M. The nature of peptide interactions with acid end-group PLGAs and facile aqueous-based microencapsulation of therapeutic peptides. J. Controlled Release 2013, 172, 662– 670, DOI: 10.1016/j.jconrel.2013.08.295Google ScholarThere is no corresponding record for this reference.
- 11Rapier, C. E.; Shea, K. J.; Lee, A. P. Investigating PLGA microparticle swelling behavior reveals an interplay of expansive intermolecular forces. Sci. Rep. 2021, 11, 14512, DOI: 10.1038/s41598-021-93785-6Google ScholarThere is no corresponding record for this reference.
- 12Sun, J.; Walker, J.; Beck-Broichsitter, M.; Schwendeman, S. P. Characterization of commercial PLGAs by NMR spectroscopy. Drug Delivery Transl. Res. 2022, 12, 720– 729, DOI: 10.1007/s13346-021-01023-3Google ScholarThere is no corresponding record for this reference.
- 13Wang, X. In situ forming risperidone implants: Effect of PLGA attributes on product performance. J. Controlled Release 2023, 361, 777– 791, DOI: 10.1016/j.jconrel.2023.08.029Google ScholarThere is no corresponding record for this reference.
- 14Wan, B.; Bao, Q.; Wang, R.; Burgess, D. J. Polymer source affects in vitro-in vivo correlation of leuprolide acetate PLGA microspheres. Int. J. Pharm. 2022, 625, 122032, DOI: 10.1016/j.ijpharm.2022.122032Google ScholarThere is no corresponding record for this reference.
- 15Wang, X.; Wang, R.; Roy, M.; Kwok, O.; Burgess, D. J. Long-acting injectable in situ forming implants: Impact of polymer attributes and API. Int. J. Pharm. 2025, 670, 125080, DOI: 10.1016/j.ijpharm.2024.125080Google ScholarThere is no corresponding record for this reference.
- 16Beig, A. Physical-Chemical Characterization of Octreotide Encapsulated in Commercial Glucose-Star PLGA Microspheres. Mol. Pharmaceutics 2020, 17, 4141– 4151, DOI: 10.1021/acs.molpharmaceut.0c00619Google ScholarThere is no corresponding record for this reference.
- 17Wang, J.; Helder, L.; Shao, J.; Jansen, J. A.; Yang, M.; Yang, F. Encapsulation and release of doxycycline from electrospray-generated PLGA microspheres: Effect of polymer end groups. Int. J. Pharm. 2019, 564, 1– 9, DOI: 10.1016/j.ijpharm.2019.04.023Google ScholarThere is no corresponding record for this reference.
- 18Black, C. pH-Dependent Protein Chemical Degradation as a Representation of Effective pH Around Proteins Within Polymer-Based Sustained Release Formulations. J. Pharm. Sci. 2025, 114, 103963, DOI: 10.1016/j.xphs.2025.103963Google ScholarThere is no corresponding record for this reference.
- 19Lin, X.; O’Reilly Beringhs, A.; Lu, X. Applications of Nanoparticle-Antibody Conjugates in Immunoassays and Tumor Imaging. AAPS J. 2021, 23, 43, DOI: 10.1208/s12248-021-00561-5Google ScholarThere is no corresponding record for this reference.
- 20Lin, E.; Alessio, A. What are the basic concepts of temporal, contrast, and spatial resolution in cardiac CT?. J. Cardiovasc. Comput. Tomogr. 2009, 3, 403– 408, DOI: 10.1016/j.jcct.2009.07.003Google ScholarThere is no corresponding record for this reference.
- 21Lin, X. Implant dynamics, inner structure, and their impact on drug release of in situ forming implants uncovered through CT imaging. J. Controlled Release 2024, 375, 802– 811, DOI: 10.1016/j.jconrel.2024.09.045Google ScholarThere is no corresponding record for this reference.
- 22Saha, S. Evaluation of the impact of the polymer end groups and molecular weight on in vitro and in vivo performances of PLGA based in situ forming implants for ketoprofen. J. Pharm. Sci. 2025, 114, 424, DOI: 10.1016/j.xphs.2024.10.019Google ScholarThere is no corresponding record for this reference.
- 23Li, J.; Nemes, P.; Guo, J. Mapping intermediate degradation products of poly(lactic-co-glycolic acid) in vitro. J. Biomed. Mater. Res., Part B 2018, 106, 1129– 1137, DOI: 10.1002/jbm.b.33920Google ScholarThere is no corresponding record for this reference.
- 24Saraf, I. Influence of PLGA End Groups on the Release Profile of Dexamethasone from Ocular Implants. Mol. Pharmaceutics 2023, 20, 1307– 1322, DOI: 10.1021/acs.molpharmaceut.2c00945Google ScholarThere is no corresponding record for this reference.
- 25Wu, X. S.; Wang, N. Synthesis, characterization, biodegradation, and drug delivery application of biodegradable lactic/glycolic acid polymers. Part II: biodegradation. J. Biomater. Sci., Polym. Ed. 2001, 12, 21– 34, DOI: 10.1163/156856201744425Google ScholarThere is no corresponding record for this reference.
- 26Fredenberg, S.; Wahlgren, M.; Reslow, M.; Axelsson, A. The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems─A review. Int. J. Pharm. 2011, 415, 34– 52, DOI: 10.1016/j.ijpharm.2011.05.049Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
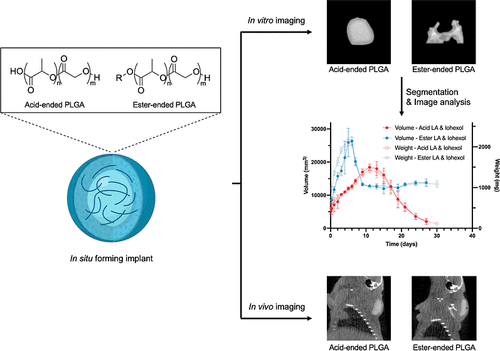
Figure 1
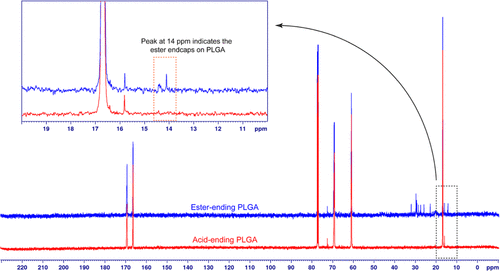
Figure 1. Comparison of 13C NMR spectra of the PLGA with different end-caps. The peak appearing at 14 ppm was used as the criterion to determine the end-cap of the PLGA.
Figure 2
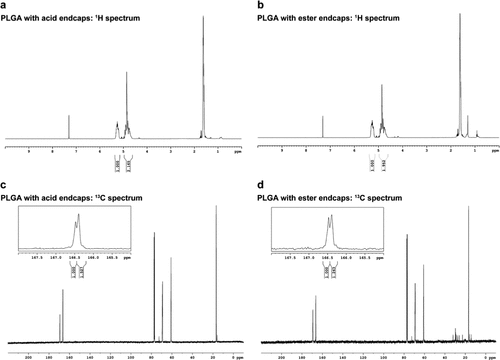
Figure 2. 1H and 13C NMR spectra of the PLGA with different end-caps. 1H NMR spectra of PLGA with acid end-caps (a) and ester end-caps (b) were used to determine the L/G ratio of the PLGA. The peaks appearing at 166.5 ppm on 13C NMR spectra of PLGA with acid end-caps (c) and ester end-caps (d) were used to determine the blockiness of the PLGA.
Figure 3

Figure 3. In vitro release test of ISFIs. No drug: placebo formulation, LA: formulation with leuprolide acetate. LA and iohexol: formulation with both leuprolide acetate and iohexol. Cumulative release of NMP from the formulations with no drug (a), leuprolide acetate (b), and both leuprolide acetate and iohexol (c). Cumulative release of leuprolide acetate from the formulations with leuprolide acetate (d) and both leuprolide acetate and iohexol (e). Cumulative release of iohexol from the formulation with both leuprolide acetate and iohexol (f). All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
Figure 4
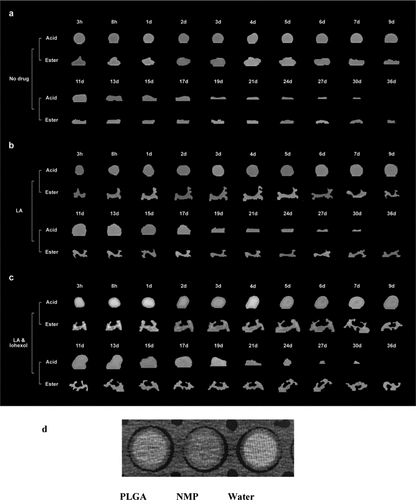
Figure 4. CT images of in vitro-formed implants. Representative transaxial sections of the 3D CT images collected for each implant at each time point (n = 3 per formulation). CT images of in vitro-formed implants from the formulations with no drug (a), only leuprolide acetate (b), and both leuprolide acetate and iohexol (c), PLGA, solvent (NMP), and water (d).
Figure 5

Figure 5. Volumes and weights of in vitro-formed implants. Volumes and weights of in vitro-formed implants with no drug (a), only leuprolide acetate (b), and both leuprolide acetate and iohexol (c). All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
Figure 6

Figure 6. MWs change of the PLGA from in vitro-formed implants. Acid and ester represent the end-cap of PLGA. No drug: placebo formulation, LA: formulation with only leuprolide acetate. Iohexol and LA: formulation with both iohexol and leuprolide acetate. All error bars are equivalent (SD positive and negative values) and represent standard deviation with n = 3.
Figure 7

Figure 7. SEM images of in vitro-formed implants. Representative SEM images of the core and the shell of in vitro-formed implants in PBS after 7, 15, and 21 days of incubation. Representative images are shown, the regions of core and edge of each implant were scanned, three different implants were analyzed for each time point and formulation. (a) SEM images of the core of the polymer matrix. (b) SEM images of the edge of the polymer matrix. The scale bar represents 100 μm.
Figure 8

Figure 8. Porosity of the microstructure of in vitro-formed implants. Porosity of the microstructure was quantified by ImageJ. Porosity of the core and the edge of in vitro-formed implants after 7 (a), 15 (b), and 21 days (c) are shown in the figure (“*” means P < 0.05).
Figure 9

Figure 9. CT images of in vivo-formed implants. Representative coronal sections of the 3D CT images were collected for each implant at each time point (n = 5 per formulation). Iohexol: formulation with only iohexol, iohexol and LA: formulation with both iohexol and leuprolide acetate.
References
This article references 26 other publications.
- 1O’Brien, M. N.; Jiang, W.; Wang, Y.; Loffredo, D. M. Challenges and opportunities in the development of complex generic long-acting injectable drug products. J. Controlled Release 2021, 336, 144– 158, DOI: 10.1016/j.jconrel.2021.06.017There is no corresponding record for this reference.
- 2Thakur, R. R.; McMillan, H. L.; Jones, D. S. Solvent induced phase inversion-based in situ forming controlled release drug delivery implants. J. Controlled Release 2014, 176, 8– 23, DOI: 10.1016/j.jconrel.2013.12.020There is no corresponding record for this reference.
- 3Benhabbour, S. R. Ultra-long-acting tunable biodegradable and removable controlled release implants for drug delivery. Nat. Commun. 2019, 10, 4324, DOI: 10.1038/s41467-019-12141-5There is no corresponding record for this reference.
- 4Wang, X.; Burgess, D. J. Drug release from in situ forming implants and advances in release testing. Adv. Drug Delivery Rev. 2021, 178, 113912, DOI: 10.1016/j.addr.2021.113912There is no corresponding record for this reference.
- 5Park, K. PLGA-based long-acting injectable (LAI) formulations. J. Controlled Release 2025, 382, 113758, DOI: 10.1016/j.jconrel.2025.113758There is no corresponding record for this reference.
- 6Ibrahim, T. M.; El-Megrab, N. A.; El-Nahas, H. M. An overview of PLGA in-situ forming implants based on solvent exchange technique: effect of formulation components and characterization. Pharm. Dev. Technol. 2021, 26, 709– 728, DOI: 10.1080/10837450.2021.1944207There is no corresponding record for this reference.
- 7Parent, M. PLGA in situ implants formed by phase inversion: critical physicochemical parameters to modulate drug release. J. Controlled Release 2013, 172, 292– 304, DOI: 10.1016/j.jconrel.2013.08.024There is no corresponding record for this reference.
- 8Kempe, S.; Mader, K. In situ forming implants - an attractive formulation principle for parenteral depot formulations. J. Controlled Release 2012, 161, 668– 679, DOI: 10.1016/j.jconrel.2012.04.016There is no corresponding record for this reference.
- 9Suh, M. S. Effect of implant formation on drug release kinetics of in situ forming implants. Int. J. Pharm. 2021, 592, 120105, DOI: 10.1016/j.ijpharm.2020.120105There is no corresponding record for this reference.
- 10Sophocleous, A. M. The nature of peptide interactions with acid end-group PLGAs and facile aqueous-based microencapsulation of therapeutic peptides. J. Controlled Release 2013, 172, 662– 670, DOI: 10.1016/j.jconrel.2013.08.295There is no corresponding record for this reference.
- 11Rapier, C. E.; Shea, K. J.; Lee, A. P. Investigating PLGA microparticle swelling behavior reveals an interplay of expansive intermolecular forces. Sci. Rep. 2021, 11, 14512, DOI: 10.1038/s41598-021-93785-6There is no corresponding record for this reference.
- 12Sun, J.; Walker, J.; Beck-Broichsitter, M.; Schwendeman, S. P. Characterization of commercial PLGAs by NMR spectroscopy. Drug Delivery Transl. Res. 2022, 12, 720– 729, DOI: 10.1007/s13346-021-01023-3There is no corresponding record for this reference.
- 13Wang, X. In situ forming risperidone implants: Effect of PLGA attributes on product performance. J. Controlled Release 2023, 361, 777– 791, DOI: 10.1016/j.jconrel.2023.08.029There is no corresponding record for this reference.
- 14Wan, B.; Bao, Q.; Wang, R.; Burgess, D. J. Polymer source affects in vitro-in vivo correlation of leuprolide acetate PLGA microspheres. Int. J. Pharm. 2022, 625, 122032, DOI: 10.1016/j.ijpharm.2022.122032There is no corresponding record for this reference.
- 15Wang, X.; Wang, R.; Roy, M.; Kwok, O.; Burgess, D. J. Long-acting injectable in situ forming implants: Impact of polymer attributes and API. Int. J. Pharm. 2025, 670, 125080, DOI: 10.1016/j.ijpharm.2024.125080There is no corresponding record for this reference.
- 16Beig, A. Physical-Chemical Characterization of Octreotide Encapsulated in Commercial Glucose-Star PLGA Microspheres. Mol. Pharmaceutics 2020, 17, 4141– 4151, DOI: 10.1021/acs.molpharmaceut.0c00619There is no corresponding record for this reference.
- 17Wang, J.; Helder, L.; Shao, J.; Jansen, J. A.; Yang, M.; Yang, F. Encapsulation and release of doxycycline from electrospray-generated PLGA microspheres: Effect of polymer end groups. Int. J. Pharm. 2019, 564, 1– 9, DOI: 10.1016/j.ijpharm.2019.04.023There is no corresponding record for this reference.
- 18Black, C. pH-Dependent Protein Chemical Degradation as a Representation of Effective pH Around Proteins Within Polymer-Based Sustained Release Formulations. J. Pharm. Sci. 2025, 114, 103963, DOI: 10.1016/j.xphs.2025.103963There is no corresponding record for this reference.
- 19Lin, X.; O’Reilly Beringhs, A.; Lu, X. Applications of Nanoparticle-Antibody Conjugates in Immunoassays and Tumor Imaging. AAPS J. 2021, 23, 43, DOI: 10.1208/s12248-021-00561-5There is no corresponding record for this reference.
- 20Lin, E.; Alessio, A. What are the basic concepts of temporal, contrast, and spatial resolution in cardiac CT?. J. Cardiovasc. Comput. Tomogr. 2009, 3, 403– 408, DOI: 10.1016/j.jcct.2009.07.003There is no corresponding record for this reference.
- 21Lin, X. Implant dynamics, inner structure, and their impact on drug release of in situ forming implants uncovered through CT imaging. J. Controlled Release 2024, 375, 802– 811, DOI: 10.1016/j.jconrel.2024.09.045There is no corresponding record for this reference.
- 22Saha, S. Evaluation of the impact of the polymer end groups and molecular weight on in vitro and in vivo performances of PLGA based in situ forming implants for ketoprofen. J. Pharm. Sci. 2025, 114, 424, DOI: 10.1016/j.xphs.2024.10.019There is no corresponding record for this reference.
- 23Li, J.; Nemes, P.; Guo, J. Mapping intermediate degradation products of poly(lactic-co-glycolic acid) in vitro. J. Biomed. Mater. Res., Part B 2018, 106, 1129– 1137, DOI: 10.1002/jbm.b.33920There is no corresponding record for this reference.
- 24Saraf, I. Influence of PLGA End Groups on the Release Profile of Dexamethasone from Ocular Implants. Mol. Pharmaceutics 2023, 20, 1307– 1322, DOI: 10.1021/acs.molpharmaceut.2c00945There is no corresponding record for this reference.
- 25Wu, X. S.; Wang, N. Synthesis, characterization, biodegradation, and drug delivery application of biodegradable lactic/glycolic acid polymers. Part II: biodegradation. J. Biomater. Sci., Polym. Ed. 2001, 12, 21– 34, DOI: 10.1163/156856201744425There is no corresponding record for this reference.
- 26Fredenberg, S.; Wahlgren, M.; Reslow, M.; Axelsson, A. The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems─A review. Int. J. Pharm. 2011, 415, 34– 52, DOI: 10.1016/j.ijpharm.2011.05.049There is no corresponding record for this reference.