
Atomic-Site Coordination Tuning for Precise CO2 ElectroconversionClick to copy article linkArticle link copied!
- Tianshang ShanTianshang ShanSchool of Materials Science & Engineering, Beijing Institute of Technology, Beijing 100081, ChinaZhongguancun Academy, Beijing 100094, ChinaMore by Tianshang Shan
- Gengxian ZhouGengxian ZhouSchool of Computer Science (National Pilot Software Engineering School), Beijing University of Posts and Telecommunications, Beijing 100876, ChinaZhongguancun Academy, Beijing 100094, ChinaMore by Gengxian Zhou
- Hongpan Rong*Hongpan Rong*E-mail: [email protected]School of Materials Science & Engineering, Beijing Institute of Technology, Beijing 100081, ChinaMore by Hongpan Rong
- Jiatao Zhang*Jiatao Zhang*E-mail: [email protected]School of Chemistry and Chemical Engineering, Beijing Key Laboratory of Intelligent Molecular Materials and High-throughput Manufacturing, MIIT Key Laboratory of Medical Molecule Science and Pharmaceutical Engineering, MOE Key Laboratory of Cluster Science, Beijing Institute of Technology, Beijing 100081, ChinaBeijing Institute of Technology, Zhuhai 519088, ChinaMore by Jiatao Zhang
Abstract
Electrochemical carbon dioxide reduction (ECR) presents a promising avenue for achieving carbon neutrality by converting greenhouse gases into high-value fuels and chemicals. However, the advancement of ECR hinges on the precise design and synthesis of catalysts that exhibit high activity, selectivity, and stability. Single-atom site catalysts (SASCs), benefiting from their maximum atom-utilization efficiency, uniform and tunable active sites, and unique electronic properties arising from strong metal–support interactions, have emerged as a powerful platform for investigating the structure–activity relationship of ECR. By tuning coordination environments (e.g., coordination types and numbers) of isolated metal atoms, the electronic structure of metal active sites can be precisely modified for governing the selective synthesis of ECR products. Herein, this review systematically summarizes the precise synthesis strategies, advanced characterization techniques, and structure–activity relationships of SASCs in various coordination environments. Furthermore, the limitations and necessary precautions associated with the current characterization techniques are also discussed. Finally, we outline the future challenges and potential research directions of SASCs in the field of ECR.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of Precision Chemistry special issue “Precision Chemistry for Single-Atom Catalysis”.
1. Introduction
2. Coordination Types of SASCs
2.1. M1-Nx
Figure 1
Figure 1. (a) Synthesis scheme of the Ni–N–C catalysts by using a N-doped carbon host to absorb Ni ions followed by thermal activation at different temperatures to tune the Ni–N bond structures and establish structure–property correlations. Reproduced from ref (131). Copyright 2022, Royal Society of Chemistry. (b) Schematic illustration for the formation of the Ni-NC@Ni catalyst. In the green circle: the possible atomic structure of the Ni–N species. Reproduced from ref (132). Copyright 2020, Elsevier. (c) Schematic illustration of the preparation strategy for PSB-CuN3 and PS-CuN4. Reproduced from ref (133). Copyright 2023, Springer Nature. (d) Schematic illustration for the preparation of Ni–PxNy. Reproduced from ref (91). Copyright 2023, Springer Nature. (e) The formation process of atomically dispersed SnN3O1 active sites. Reproduced from ref (107). Copyright 2021, Wiley-VCH. (f) Schematic illustration of the proposed formation mechanisms. Reproduced from ref (118). Copyright 2021, Springer Nature.
2.2. M1-M’x
Figure 2
Figure 2. (a) Schematic synthesis process of the Sn1Cu-SAA. Reproduced from ref (77). Copyright 2025, Wiley-VCH. (b) Schematic representation of the preparation of the Ru1@Bi and Run@Bi catalysts. Reproduced from ref (143). Copyright 2024, Wiley-VCH. (c) Schematic diagram for the preparation of Mo1Cu catalysts. Reproduced from ref (144). Copyright 2025, Wiley-VCH. (d) Schematic diagram of the NiZn0.03/C bimetallic catalyst synthesis process. Reproduced from ref (145). Copyright 2023, Wiley-VCH.
2.3. M1-Ox
Figure 3
Figure 3. (a) Schematic illustration of preparing Bi1–xVO4–Cu. Reproduced from ref (160). Copyright 2024, Wiley-VCH. (b) Step-by-step preparation of the carbon-supported Cu SA catalyst using an amalgamated Cu–Li method. Reproduced from ref (158). Copyright 2020, Springer Nature. (c) Schematic illustration of the support crystal phase engineering and reaction mechanisms on the surface of Cu1O3-tZrO2 and Cu1O4-mZrO2. Reproduced from ref (83). Copyright 2025, American Chemical Society. (d) Illustration of the synthesis of Zr-BTB-In. Reproduced from ref (154). Copyright 2025, Wiley-VCH. (e) Schematic illustration of the fabrication of SA-Cu-MXene via selective etching of quaternary MAX-Ti3(Al1–xCux)C2. Gray, blue, red, yellow, brown, and green balls represent Al, Ti, Cu, O, C, and Cl atoms, respectively. Reproduced from ref (156). Copyright 2021, American Chemical Society.
3. Coordination Detection of SASCs
3.1. AC-HAADF-STEM
Figure 4
Figure 4. (a) General schematic of an electron microscope in STEM modes with a condenser lens, objective lens, aberration correctors, projector system, retractable detectors, EDS (energy dispersive X-ray spectroscopy), and EELS (electron energy loss spectroscopy) acquisition. p.s. BF (bright-field), ABF (annular bright-field). Reproduced from ref (171). Copyright 2021, Wiley-VCH. (b) Pt single atoms (white circles) are seen to be uniformly dispersed on the iron oxide (FeOx) support (left) and occupy exactly the positions of the Fe atoms (right). Reproduced from ref (44). Copyright 2011, Springer Nature. (c) Schematic diagram of XAFS measurements with fluorescence yield experiment setup. Reproduced from ref (74). Copyright 2024, Springer Nature. (d) Schematic diagram of Pd–O4/BWO and Pd–O3/BWO. (e) Fourier transforms of the EXAFS spectra of the Pd K-edge spectra. Reproduced from ref (172). Copyright 2025, Springer Nature. (f) Schematic diagram of operando Mössbauer spectroscopy for tracking the metastable states of atomically dispersed tin (Sn) in copper oxide (CuO) for selective CO2 electroreduction. Reproduced from ref (162). Copyright 2023, American Chemical Society.
3.2. XAFS
3.3. Mössbauer Spectroscopy
3.4. Intrinsic Limits
Figure 5
Figure 5. (a) Elastic scattering of electrons from an atomic nucleus, shown schematically (particle model) for a large-angle collision (A) and a 180° collision (B). Sputtering of atoms from the beam-exit surface (C) and the beam-entry surface (D). Reproduced from ref (186). Copyright 2010, Elsevier. (b) Energy-band diagram of a metal (left) and a semiconductor or insulator (right), electron energy being plotted vertically upward. CB and VB represent the conduction and valence bands, f is the work function, Evac, Ef and Eg are the vacuum energy, Fermi energy, and CB-VB energy gap. Upward arrows represent single-electron excitations, while downward arrows are de-excitation processes (filling of a VB or K-shell hole). K-shell excitation in an organic material is shown on the right, with Ekin being the typical kinetic energy of an excited K-shell electron. This process also results in the emission of Auger electrons from the VB, with an energy of about 270 eV. Reproduced from ref (187). Copyright 2013, Elsevier. (c) Schematic diagram of heterogeneous Pt/CeO2 SASCs using EXAFS. (d) Scattering paths for SASCs and small clusters. Schematic depiction of scattering paths present in oxide-supported metal catalysts with a heterogeneity of the metal-site structure including isolated metal sites (middle) as well as metal (left) or oxidized (right) nanoclusters. Bonds are for representation only. Color code: metal (gray), oxygen (red), and support metal cation (brown). (e) R-factor of the 5-path SAC fit to data sets with varying contributions from the Pt/CeO2 SASCs site and metallic Pt13 cluster. The inset shows a zoomed-in region with 70–100% SAC sites (30–0% Pt13 cluster sites). Data are taken over Δk = 3–18 Å–1. (f) Modeled FT EXAFS data for mixtures of the Pt/CeO2 SASCs site and metallic Pt13 cluster. Reproduced from ref (188). Copyright 2023, American Chemical Society.
4. Structure–Activity Relationship of SASCs
Figure 6
Figure 6. Overview of reaction pathways for ECR toward different products. Black spheres, carbon; red spheres, oxygen; white spheres, hydrogen; blue spheres, (metal) catalyst. The arrows indicate whether proton, electron or PCETs take place. Reproduced from ref (39). Copyright 2019, Springer Nature.
| Half-cell reaction | Standard potentials(V vs SHE) |
|---|---|
| 2H+ + 2e– → H2 | –0.42 |
| CO2 + e– → *CO2– | –1.9 |
| CO2 + 2H+ + 2e– → CO + H2O | –0.52 |
| CO2 + 2H+ + 2e– → HCOOH | –0.61 |
| CO2 + 4H+ + 4e– → HCHO + H2O | –0.51 |
| CO2 + 6H+ + 6e– → CH3OH + H2O | –0.38 |
| CO2 + 8H+ + 8e– → CH4 + 2H2O | –0.24 |
| 2CO2 + 12H+ + 12e– → C2H4 + 4H2O | –0.34 |
| 2CO2 + 12H+ + 12e– → C2H5OH + 3H2O | –0.33 |
4.1. CO2 to C1 Products
4.1.1. CO
Figure 7
Figure 7. (a) Schematic illustration of CO2RR over a chemical unit of the TM-Pcs. (b) Free energy diagram of TM-PCs for CO2RR at V = 0 V. Reproduced from ref (116). Copyright 2022, American Chemical Society. (c) Stability test of Ni–N3S–C in flow cell with alkaline, acidic, and neutral electrolyte. The electrolytes are 1 M KOH (alkaline, pH = 14), 0.05 M H2SO4 + 1 M KCl (acidic, pH = 1), and 1 M KHCO3 (neutral, pH = 8.2). Reproduced from ref (102). Copyright 2025, Wiley-VCH. (d) Top-left, top-right: Top view and side view optimized adsorption configuration on simulated FeN4 and O–Fe–N-C (Fe, O, N, and C atoms are represented in purple, red, blue, and gray, respectively). Down-left, down-right: Free energy profiles for the CO2RR to CO at 0 V (vs RHE) and at – 0.48 V (vs RHE) on simulated FeN4 and O–Fe–N-C. Reproduced from ref (208). Copyright 2022, Wiley-VCH. (e) Scheme of ECR-to-CO activity for Ni-SAC@NFC. Reproduced from ref (130). Copyright 2024, American Chemical Society. (f) Scheme of ECR-to-CO activity for Ni–N4/C-NH2. Reproduced from ref (209). Copyright 2021, Royal Society Chemistry. (g) Optimized structures of Ni–N4, V1–Ni-N4, V2–Ni-N4, and V3–Ni-N4. (h) Free energy diagram for the ECR-to-CO conversion. Reproduced from ref (81). Copyright 2025, American Chemical Society. (i) Schematic diagram of the orbital matching mechanism, which originated from the characteristic d-d and d-p orbital interactions between the substrate and the dispersed metal atoms. The ″volcano-type″ scaling relationship between the number of valence electrons (nve) of dispersed elements and (j) single atom (k) cluster formation energies. Reproduced from ref (78). Copyright 2025, Wiley-VCH. (l) Different spin states of Co, Fe, and Mn atoms in TM-TCSACs (TM = Co, Fe, and Mn) and CO*. Reproduced from ref (71). Copyright 2025, American Chemical Society.
4.1.2. CH4
Figure 8
Figure 8. (a) Schematic of the synthesis of CuNC and ssCuNC via support engineering. DCD: Dicyandiamide, PNP-Cu: Cu-chelated polydopamine nanoparticles, ss-PNF-Cu: Cu-bound polydopamine nanoparticles and films coated on ss. (b) AC-HAADF-STEM images of (i) ssCuNC40, (ii) ssCuNC80, (iii) ssCuNC100, and (iv) ssCuNC160. Reproduced from ref (113). Copyright 2025, Springer Nature. (c) FE and partial current densities for CH4 of BNC-Cu and NC-Cu as a function of cathodic potentials. Red curve for partial current density and black curve for methane FE. The error bars of FECH4 and jCH4 are calculated based on three independent measurements. Reproduced from ref (105). Copyright 2023, Springer Nature. (d) Conceptual schematic of the self-healing coordination reconstruction process. Reproduced from ref (212). Copyright 2025, Springer Nature. (e) Scheme of ECR-to-CH4 activity for SA-Zn/MNC. Reproduced from ref (213). Copyright 2020, American Chemical Society. (f) Left: Schematic diagram of CO adjacent-adsorption on the Al2O3–CuSAC surface. Right: CO adsorption energy of Al2O3–CuSAC, CeO2–CuSAC, and TiO2–CuSAC. Reproduced from ref (72). Copyright 2025, Springer Nature. (g) Scheme of ECR-toCH4 activity for SA-Zn/MNC. Reproduced from ref (214). Copyright 2022, American Chemical Society. (h) Diagrams of the electroreduction processes over Bi1–xVO4, Bi1–xVO4–Cu, and BiVO4–Cu. Reproduced from ref (160). Copyright 2023, Wiley-VCH. (i) Atomic elemental mapping using the EELS. (j) Comparison of reaction products for pristine Cu and Cu-FeSA. Error bars represent 1 standard deviation on the basis of three independent samples. Reproduced from ref (215). Copyright 2022, Springer Nature.
4.1.3. HCOOH
Figure 9
Figure 9. (a) Scheme of ECR-to-HCOOH activity for In Sas/NC. Reproduced from ref (224). Copyright 2020, Wiley-VCH. (b) Schematic diagrams of band shifts and hybridization for the planar CuN4 moiety with local D4h symmetry and the defective CuN3 moiety with lower local C2v symmetry. a.u. arbitrary units. Reproduced from ref (133). Copyright 2023, Springer Nature. (c) EDD plots of FeN4 and FeN2S2. (d) Distribution of ECR products over different Fe-based catalysts at −0.5 V. Reproduced from ref (89). Copyright 2025, American Chemical Society. (e) Schematic illustration for the preparation of SnPc/CNT–OH. Reproduced from ref (128). Copyright 2023, American Chemical Society. (f) Atomic-resolution AC-HAADF-STEM image and intensity profiles of Ru1@Bi. (g) Gibbs free energy landscapes for ECR-to-HCOOH at 0 V versus RHE. The RDS is labeled with a black text. Reproduced from ref (143). Copyright 2024, Wiley-VCH. (h) Comparison of FECO (orange color) and FEHCOO– (green color) on Cu, Cu99In1, Cu95In5, Cu92In8, Cu1In99 and In catalysts. Reproduced from ref (79). Copyright 2025, Wiley-VCH. (i) Electron localization function maps of Op-Ag1In. (j) Charge density difference map of CO2 on Op-Ag1In. Reproduced from ref (225). Copyright 2024, Wiley-VCH. (k) In-situ ATR-FTIR spectra on CuHHTP/Cu(OH)2 at −1.7 V for 60 min in a CO2-saturated 0.1 M KHCO3–KCl electrode. (l) The charge density difference analysis on CuHHTP/Cu(OH)2 and Cu(OH)2. Reproduced from ref (226). Copyright 2025, Wiley-VCH.
4.2. CO2 to C2+ Products
Figure 10
Figure 10. (a) Low magnification EC-STEM images of pristine-SAC and Cu nanograins after the CO2RR for 120 s in liquids. Reproduced from ref (80). Copyright 2025, American Chemical Society. (b) Linear combination fitting of Cu oxidation states for CuPc-F. (c) Cu composition ratio and FEs of various products at multiple potentials for CuPc-F. Reproduced from ref (82). Copyright 2025, American Chemical Society. (d) F, O-codrived local coordination environment optimized for CuFONC favors the CO2RR toward C2+. (e) The reaction pathways of CO2 to *COCHO on CuN2O1–FC, CuN2O1–C, and CuN3–C. Reproduced from ref (121). Copyright 2024, Wiley-VCH. (f) Schematic illustration of the modular design of the Cu/Ni-NAC hybrid catalyst for tandem catalysis. In-situ SEIRAS spectra of (g) the Cu/Ni-NAC and (h) the Cu NW catalysts under ECR conditions. Reproduced from ref (95). Copyright 2022, American Chemical Society. (i) A proposed reaction mechanism for the CO2RR to C2H4 and C2H5OH. Reproduced from ref (144). Copyright 2025, Wiley-VCH. (j) Simulated geometry and charge density difference of M1/Cu(111). Reproduced from ref (68). Copyright 2025, Springer Nature. FEs and the product distributions under different polarization potentials over (k) Sn/C-0.12 and (l) Sn/C-1.2 (stacked bar chart) and the geometric partial current densities, J, toward (k) acetate and (l) ethanol (blue line-stars). All FEs are calculated from chronoamperometry measurements. Reproduced from ref (161). Copyright 2024, American Chemical Society. (m) A schematic illustration showing the cascade reaction during CO2 reduction to ethanol over SnS2/Sn1–O3G (gray: S, red: O, yellow: H and purple: Sn). Reproduced from ref (157). Copyright 2023, Springer Nature.
5. Conclusions and Outlook
Acknowledgments
This work was supported by the National Natural Science Foundation of China (52272186).
References
This article references 233 other publications.
- 1Wang, Q.; Luo, T.; Cao, X.; Gong, Y.; Liu, Y.; Xiao, Y.; Li, H.; Gröbmeyer, F.; Lu, Y.-R.; Chan, T.-S.; Ma, C.; Liu, K.; Fu, J.; Zhang, S.; Liu, C.; Lin, Z.; Chai, L.; Cortes, E.; Liu, M. Lanthanide Single-Atom Catalysts for Efficient CO2-to-CO Electroreduction. Nat. Commun. 2025, 16 (1), 2985, DOI: 10.1038/s41467-025-57464-8Google ScholarThere is no corresponding record for this reference.
- 2Wei, C.; Wang, S.; Zhao, X. Spatial-Temporal Variation and Coupling Relationship between Primary Energy Consumption and Economic Growth: A Global Assessment. Energy 2025, 323, 135873, DOI: 10.1016/j.energy.2025.135873Google ScholarThere is no corresponding record for this reference.
- 3Raihan, A. Carbon Capture, Utilization, and Storage (CCUS) in the Context of Earth Energy Systems: A Multidisciplinary Review. Geosci. Front. 2025, 16 (6), 102177, DOI: 10.1016/j.gsf.2025.102177Google ScholarThere is no corresponding record for this reference.
- 4Wu, Q.; Ji, S.; Chen, J.; Tan, X.-Q.; Ong, W.-J.; Du, R.; Wang, P.; Wang, H.; Qiu, Y.; Yan, K.; Zhao, Y.; Zhao, W.-W.; Peng, K.-S.; Chen, Y.-Y.; Hung, S.-F.; Zhou, L.; Wang, X.; Qiu, G.; Chen, G. Realizing the Practical Application of CO2 Electroreduction for Urban Wastewater Denitrification. Nat. Water. 2025, 3 (11), 1291– 1302, DOI: 10.1038/s44221-025-00516-6Google ScholarThere is no corresponding record for this reference.
- 5Bai, W.; Zeng, H.; Chen, F.; Wu, S.; Wang, S.; Du, Y.; Liu, S.; Wang, D.; Dai, Z. Emerging Atomistic Modeling Catalysts for C-N Electrocatalysis. Adv. Mater. 2026, 38, e10907 DOI: 10.1002/adma.202510907Google ScholarThere is no corresponding record for this reference.
- 6Zhao, D.; Yu, K.; Song, P.; Feng, W.; Hu, B.; Cheong, W.-C.; Zhuang, Z.; Liu, S.; Sun, K.; Zhang, J.; Chen, C. Atomic-Level Engineering Fe1N2O2 Interfacial Structure Derived from Oxygen-Abundant Metal-Organic Frameworks to Promote Electrochemical CO2 Reduction. Energy Environ. Sci. 2022, 15 (9), 3795– 3804, DOI: 10.1039/D2EE00878EGoogle ScholarThere is no corresponding record for this reference.
- 7Song, P.; Hu, B.; Zhao, D.; Fu, J.; Su, X.; Feng, W.; Yu, K.; Liu, S.; Zhang, J.; Chen, C. Modulating the Asymmetric Atomic Interface of Copper Single Atoms for Efficient CO2 Electroreduction. ACS Nano 2023, 17 (5), 4619– 4628, DOI: 10.1021/acsnano.2c10701Google ScholarThere is no corresponding record for this reference.
- 8Vos, R. E.; Sun, P.; Schauermann, D.; Javed, H.; Hanselman, S. R.; Fu, G.; Koper, M. T. M. CO2 Electroreduction on Cu Operates via an Alternative Chain Growth Mechanism to Form C-C Bonds at Elevated Temperature and Pressure. Nat. Catal. 2025, 8 (12), 1338– 1347, DOI: 10.1038/s41929-025-01451-1Google ScholarThere is no corresponding record for this reference.
- 9Bi, X.; Yan, Y.; Wang, H.; Zhao, Y.; Zhang, J.; Wu, M. Electroreduction of CO2 to C2H4 Regulated by Spacing Effect: Mechanistic Insights from DFT Studies. Energy Mater. Adv. 2023, 4, 0037, DOI: 10.34133/energymatadv.0037Google ScholarThere is no corresponding record for this reference.
- 10Wang, S.; Li, F.; Zhao, J.; Zeng, Y.; Li, Y.; Lin, Z.-Y.; Lee, T.-J.; Liu, S.; Ren, X.; Wang, W.; Chen, Y.; Hung, S.-F.; Lu, Y.-R.; Cui, Y.; Yang, X.; Li, X.; Huang, Y.; Liu, B. Manipulating C-C Coupling Pathway in Electrochemical CO2 Reduction for Selective Ethylene and Ethanol Production over Single-Atom Alloy Catalyst. Nat. Commun. 2024, 15 (1), 10247, DOI: 10.1038/s41467-024-54636-wGoogle ScholarThere is no corresponding record for this reference.
- 11Zhang, W.; Zhou, Z.; Tan, X.; Tan, T. Mastering Crystalline Phase in Bismuth-Based Electrocatalysts for Efficient CO2-to-Formate Conversion. Energy Mater. Adv. 2025, 0, DOI: 10.34133/energymatadv.0242Google ScholarThere is no corresponding record for this reference.
- 12Fu, J.; Zhang, H.; Du, H.; Liu, X.; Lyu, Z.-H.; Jiang, Z.; Chen, F.; Ding, L.; Tang, T.; Zhu, W.; Su, D.; Ling, C.; Wang, J.; Hu, J.-S. Unveiling the Interfacial Species Synergy in Promoting CO2 Tandem Electrocatalysis in Near-Neutral Electrolyte. J. Am. Chem. Soc. 2024, 146 (33), 23625– 23632, DOI: 10.1021/jacs.4c08844Google ScholarThere is no corresponding record for this reference.
- 13Kim, K.-M.; Mun, J.; Yun, G.-N.; You, Y.-W.; Park, J. H.; Lee, J. H.; So, J.; Shin, H.; Kwon, J.; Kim, S.; Kang, S.; Kwon, Y. K.; Kwon, T.-H.; Bae, Y.-S.; Lee, G.; Kim, S.-J.; Kim, Y. J.; Kim, H.-T. Rational Synthesis of Dual-Atom Catalysts for Optimized Thermochemical CO2 Reduction. Nat. Commun. 2025, DOI: 10.1038/s41467-025-66608-9Google ScholarThere is no corresponding record for this reference.
- 14Li, S.; Ma, J.; Wang, N.; Liu, X.; Cui, W.; Lyu, Y.; Perathoner, S.; Centi, G.; Liu, Y. Engineering Dual-Active Sites on CeOx-Modified Defective MgO for Efficient CO2 Thermocatalytic Reduction. J. Energy Chem. 2025, 110, 486– 496, DOI: 10.1016/j.jechem.2025.06.073Google ScholarThere is no corresponding record for this reference.
- 15Hu, X.; Liu, X.; Hu, X.; Zhao, C.; Guan, Q.; Li, W. Hybrid Catalyst Coupling Zn Single Atoms and CuNx Clusters for Synergetic Catalytic Reduction of CO2. Adv. Funct. Mater. 2023, 33 (16), 2214215, DOI: 10.1002/adfm.202214215Google ScholarThere is no corresponding record for this reference.
- 16Hao, J.; Li, Y.; Li, Z.; Yin, Z.; Xu, L.; Wang, Y.; Gao, S.; Liu, Y.; Qiu, J.; Yang, Z.; Song, Z. Dual-Mechanism Regulation in Photocatalytic CO2 Reduction through Er Single-Atom Engineering: Enhancing Charge Dynamics and Molecular Activation. ACS Catal. 2025, 15 (17), 15172– 15182, DOI: 10.1021/acscatal.5c03633Google ScholarThere is no corresponding record for this reference.
- 17Xiao, Y.; Ding, G.; Tao, J.; Wang, Z.; Chen, Z.; Chen, L.; Shuai, L.; Liao, G. Selective Conversion of CO2 to C2H6 in Pure Water Photocatalyzed by Fluorobenzene-Linked Perylene Diimide. Nat. Commun. 2025, 16 (1), 7476, DOI: 10.1038/s41467-025-62369-7Google ScholarThere is no corresponding record for this reference.
- 18Wang, Z.; Hao, M.; Li, C.; Gao, X.; Yu, S.; Zhao, L.; Zhang, H.; Li, G.; Li, Y. Electron-Rich Nanorelay Enhances Ligand-to-Metal Charge Transfer in Lanthanide Metal-Organic Frameworks during Photocatalytic CO2 Conversion. Adv. Mater. 2026, 38, e12763 DOI: 10.1002/adma.202512763Google ScholarThere is no corresponding record for this reference.
- 19Ding, G.; Wang, Z.; Chen, Z.; Xiao, Y.; Liu, X.; Shuai, L.; Chen, L.; Huang, H.; Liao, G. Unsaturated Coordination-Regulated High-Spin Nickel Sites for Selective Solar-Driven Carbon Dioxide Conversion in Pure Water. Energy Environ. Sci. 2026, 19, 486, DOI: 10.1039/D5EE04331JGoogle ScholarThere is no corresponding record for this reference.
- 20Cui, Y.; Wang, Y.; Ma, Y.; Su, X.; Tai, R.; Jia, M.; Chen, J.; Liu, A.; Yu, L.; Tian, C.; Zhang, W.; Han, L.; Che, S.; Fang, Y. A Chiral Mesostructured Photocatalyst for Efficient Solar-Driven CO2 Reduction to Ethanol. Nat. Synth. 2026, 5, 117, DOI: 10.1038/s44160-025-00908-2Google ScholarThere is no corresponding record for this reference.
- 21Kuang, M.; Li, B.; Zhou, L.; Huang, Z.; Wu, J.; Wang, S.; Yang, J. Carbon Dioxide Upgrading to Biodegradable Plastics through Photo/Electro-Synthetic Biohybrid Systems. Angew. Chem., Int. Ed. 2025, 64 (5), e202422357 DOI: 10.1002/anie.202422357Google ScholarThere is no corresponding record for this reference.
- 22Li, Y.; Zhang, B.; Wu, Y.; Tang, T.; Guan, A.; Xu, Q.; Wang, L.; Liu, J.; Sun, L.; Zeng, A.-P. Ultrafast Conversion of CO2 into C3-C4 Diols in a Synergistic Electrochemical and AI-Assisted Biosynthesis System. J. Am. Chem. Soc. 2025, 147 (43), 39827– 39837, DOI: 10.1021/jacs.5c14039Google ScholarThere is no corresponding record for this reference.
- 23Pu, Y.; Wang, Y.; Wu, G.; Wu, X.; Lu, Y.; Yu, Y.; Chu, N.; He, X.; Li, D.; Zeng, R. J.; Jiang, Y. Tandem Acidic CO2 Electrolysis Coupled with Syngas Fermentation: A Two-Stage Process for Producing Medium-Chain Fatty Acids. Environ. Sci. Technol. 2024, 58 (17), 7445– 7456, DOI: 10.1021/acs.est.3c09291Google ScholarThere is no corresponding record for this reference.
- 24Zhang, W.; Hu, Y.; Ma, L.; Zhu, G.; Wang, Y.; Xue, X.; Chen, R.; Yang, S.; Jin, Z. Progress and Perspective of Electrocatalytic CO2 Reduction for Renewable Carbonaceous Fuels and Chemicals. Adv. Sci. 2018, 5 (1), 1700275, DOI: 10.1002/advs.201700275Google ScholarThere is no corresponding record for this reference.
- 25Feng, Y.-X.; Hao, J.; Zhu, S.; Zhang, B.-W.; Chen, Y. Copper Oxide Foam with High Specific Surface Area for Electrocatalytic Reduction of CO2 to C2+ Products. cMat 2024, 1 (1), e19 DOI: 10.1002/cmt2.19Google ScholarThere is no corresponding record for this reference.
- 26Shi, R.; Guo, J.; Zhang, X.; Waterhouse, G. I. N.; Han, Z.; Zhao, Y.; Shang, L.; Zhou, C.; Jiang, L.; Zhang, T. Efficient Wettability-Controlled Electroreduction of CO2 to CO at Au/C Interfaces. Nat. Commun. 2020, 11 (1), 3028, DOI: 10.1038/s41467-020-16847-9Google ScholarThere is no corresponding record for this reference.
- 27Yang, H.; Shang, L.; Zhang, Q.; Shi, R.; Waterhouse, G. I. N.; Gu, L.; Zhang, T. A Universal Ligand Mediated Method for Large Scale Synthesis of Transition Metal Single Atom Catalysts. Nat. Commun. 2019, 10 (1), 4585, DOI: 10.1038/s41467-019-12510-0Google ScholarThere is no corresponding record for this reference.
- 28Yan, T.; Chen, X.; Kumari, L.; Lin, J.; Li, M.; Fan, Q.; Chi, H.; Meyer, T. J.; Zhang, S.; Ma, X. Multiscale CO2 Electrocatalysis to C2+ Products: Reaction Mechanisms, Catalyst Design, and Device Fabrication. Chem. Rev. 2023, 123 (17), 10530– 10583, DOI: 10.1021/acs.chemrev.2c00514Google ScholarThere is no corresponding record for this reference.
- 29Tang, Y.-F.; Liu, L.-B.; Yu, M.; Liu, S.; Sui, P.-F.; Sun, W.; Fu, X.-Z.; Luo, J.-L.; Liu, S. Strong Effect-Correlated Electrochemical CO2 Reduction. Chem. Soc. Rev. 2024, 53 (18), 9344– 9377, DOI: 10.1039/D4CS00229FGoogle ScholarThere is no corresponding record for this reference.
- 30Gao, G.; Khiarak, B. N.; Liu, H.; Trȃ̀n-Phú, T.; Obasanjo, C. A.; Crane, J.; Lai, H. D. T.; da Silva, G. T. S. T.; Golovanova, V.; Li, J.; Ze, H.; Weiss, J.; Zhang, Z.; Lee, S.; Hocking, R. K.; García de Arquer, F. P.; Sargent, E. H.; Dinh, C.-T. Recoverable Operation Strategy for Selective and Stable Electrochemical Carbon Dioxide Reduction to Methane. Nat. Energy 2025, 10 (11), 1360– 1370, DOI: 10.1038/s41560-025-01883-wGoogle ScholarThere is no corresponding record for this reference.
- 31Choi, M.; Bae, S.; Kim, Y.; Lee, Y.; Cho, M.; Kang, S.; Lee, J. Selective Formaldehyde Condensation on Phosphorus-Rich Copper Catalyst to Produce Liquid C3+ Chemicals in Electrocatalytic CO2 Reduction. Nat. Catal. 2025, 8 (5), 476– 486, DOI: 10.1038/s41929-025-01341-6Google ScholarThere is no corresponding record for this reference.
- 32Liu, Y.-C.; Huang, J.-R.; Zhu, H.-L.; Qiu, X.-F.; Yu, C.; Chen, X.-M.; Liao, P.-Q. Electrosynthesis of Pure Urea from Pretreated Flue Gas in a Proton-Limited Environment Established in a Porous Solid-State Electrolyte Electrolyser. Nat. Nanotechnol. 2025, 20 (7), 907– 913, DOI: 10.1038/s41565-025-01914-3Google ScholarThere is no corresponding record for this reference.
- 33Huang, D.-S.; Wang, Y.; Tang, Y.; Huang, J.-R.; Li, P.-X.; Liang, C.-P.; Zhao, Z.-H.; Liao, P.-Q.; Chen, X.-M. Embedding a Self-Supporting MOF-Based Molecular Sieve Membrane into an Electrolyzer for Boosting Electroreduction of CO2 in Air and Flue Gas to HCOOH. Natl. Sci. Rev. 2025, 12 (10), nwaf329, DOI: 10.1093/nsr/nwaf329Google ScholarThere is no corresponding record for this reference.
- 34Zhao, Z.-H.; Huang, J.-R.; Huang, D.-S.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Efficient Capture and Electroreduction of Dilute CO2 into Highly Pure and Concentrated Formic Acid Aqueous Solution. J. Am. Chem. Soc. 2024, 146 (20), 14349– 14356, DOI: 10.1021/jacs.4c04841Google ScholarThere is no corresponding record for this reference.
- 35Liang, C.-P.; Huang, J.-R.; Zhu, H.-L.; Zhao, Z.-H.; Yu, C.; Liao, P.-Q.; Chen, X.-M. Precisely Tailoring the First Coordination Shell of Metal Centers in Porous Nitrogen-Doped Carbon Promoting Electroreduction of CO2 Under Neutral Condition. CCS Chem. 2024, 6 (8), 1978– 1986, DOI: 10.31635/ccschem.023.202303333Google ScholarThere is no corresponding record for this reference.
- 36Huang, D.-S.; Qiu, X.-F.; Huang, J.-R.; Mao, M.; Liu, L.; Han, Y.; Zhao, Z.-H.; Liao, P.-Q.; Chen, X.-M. Electrosynthesis of Urea by Using Fe2O3 Nanoparticles Encapsulated in a Conductive Metal-Organic Framework. Nat. Synth. 2024, 3 (11), 1404– 1413, DOI: 10.1038/s44160-024-00603-8Google ScholarThere is no corresponding record for this reference.
- 37Liu, Y.-Y.; Huang, J.-R.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Simultaneous Capture of CO2 Boosting Its Electroreduction in the Micropores of a Metal-Organic Framework. Angew. Chem., Int. Ed. 2023, 62 (52), e202311265 DOI: 10.1002/anie.202311265Google ScholarThere is no corresponding record for this reference.
- 38Guan, R.; Sheng, L.; Li, C.; Gu, J.; Seo, J.-M.; Jang, B.-J.; Kim, S.-H.; Kim, J.; Lim, H.; Li, Q.; Baek, J.-B. Mechanochemical Carbon Dioxide Capture and Conversion. Nat. Nanotechnol. 2025, 20 (9), 1247– 1253, DOI: 10.1038/s41565-025-01949-6Google ScholarThere is no corresponding record for this reference.
- 39Birdja, Y. Y.; Pérez-Gallent, E.; Figueiredo, M. C.; Göttle, A. J.; Calle-Vallejo, F.; Koper, M. T. M. Advances and Challenges in Understanding the Electrocatalytic Conversion of Carbon Dioxide to Fuels. Nat. Energy 2019, 4 (9), 732– 745, DOI: 10.1038/s41560-019-0450-yGoogle ScholarThere is no corresponding record for this reference.
- 40Song, Y.; Musgrave, C. B.; Su, J.; Huang, L.; Guo, W.; Liu, Y.; Li, G.; Xin, Y.; Zhang, Q.; Feng, X.; Liao, C.; Liu, S.; Kwok, R. T. K.; Lam, J. W. Y.; He, M.; Choong, K. S.; Feng, Z.; Tang, B. Z.; Goddard, W. A.; Ye, R. Efficient CO2-to-Methanol Electrocatalysis in Acidic Media via Microenvironment-Tuned Cobalt Phthalocyanine. Nat. Nanotechnol. 2026, 21, 78, DOI: 10.1038/s41565-025-02059-zGoogle ScholarThere is no corresponding record for this reference.
- 41Guan, Y.; Li, Y.; Li, Z.; Hou, Y.; Lei, L.; Yang, B. Promotion of C-C Coupling in the CO2 Electrochemical Reduction to Valuable C2+ Products: From Micro-Foundation to Macro-Application. Adv. Mater. 2025, 37 (23), 2417567, DOI: 10.1002/adma.202417567Google ScholarThere is no corresponding record for this reference.
- 42Liang, X.; Fu, N.; Yao, S.; Li, Z.; Li, Y. The Progress and Outlook of Metal Single-Atom-Site Catalysis. J. Am. Chem. Soc. 2022, 144 (40), 18155– 18174, DOI: 10.1021/jacs.1c12642Google ScholarThere is no corresponding record for this reference.
- 43Taylor, H. S. A Theory of the Catalytic Surface. Proc. A 1925, 108 (745), 105– 111, DOI: 10.1098/rspa.1925.0061Google ScholarThere is no corresponding record for this reference.
- 44Qiao, B.; Wang, A.; Yang, X.; Allard, L. F.; Jiang, Z.; Cui, Y.; Liu, J.; Li, J.; Zhang, T. Single-Atom Catalysis of CO Oxidation Using Pt1/FeOx. Nat. Chem. 2011, 3 (8), 634– 641, DOI: 10.1038/nchem.1095Google ScholarThere is no corresponding record for this reference.
- 45Liu, Y.; Li, Z.; Yu, Q.; Chen, Y.; Chai, Z.; Zhao, G.; Liu, S.; Cheong, W.-C.; Pan, Y.; Zhang, Q.; Gu, L.; Zheng, L.; Wang, Y.; Lu, Y.; Wang, D.; Chen, C.; Peng, Q.; Liu, Y.; Liu, L.; Chen, J.; Li, Y. A General Strategy for Fabricating Isolated Single Metal Atomic Site Catalysts in Y Zeolite. J. Am. Chem. Soc. 2019, 141 (23), 9305– 9311, DOI: 10.1021/jacs.9b02936Google ScholarThere is no corresponding record for this reference.
- 46Xiong, Y.; Dong, J.; Huang, Z.-Q.; Xin, P.; Chen, W.; Wang, Y.; Li, Z.; Jin, Z.; Xing, W.; Zhuang, Z.; Ye, J.; Wei, X.; Cao, R.; Gu, L.; Sun, S.; Zhuang, L.; Chen, X.; Yang, H.; Chen, C.; Peng, Q.; Chang, C.-R.; Wang, D.; Li, Y. Single-Atom Rh/N-Doped Carbon Electrocatalyst for Formic Acid Oxidation. Nat. Nanotechnol. 2020, 15 (5), 390– 397, DOI: 10.1038/s41565-020-0665-xGoogle ScholarThere is no corresponding record for this reference.
- 47Li, Z.; Chen, Y.; Ji, S.; Tang, Y.; Chen, W.; Li, A.; Zhao, J.; Xiong, Y.; Wu, Y.; Gong, Y.; Yao, T.; Liu, W.; Zheng, L.; Dong, J.; Wang, Y.; Zhuang, Z.; Xing, W.; He, C.-T.; Peng, C.; Cheong, W.-C.; Li, Q.; Zhang, M.; Chen, Z.; Fu, N.; Gao, X.; Zhu, W.; Wan, J.; Zhang, J.; Gu, L.; Wei, S.; Hu, P.; Luo, J.; Li, J.; Chen, C.; Peng, Q.; Duan, X.; Huang, Y.; Chen, X.-M.; Wang, D.; Li, Y. Iridium Single-Atom Catalyst on Nitrogen-Doped Carbon for Formic Acid Oxidation Synthesized Using a General Host-Guest Strategy. Nat. Chem. 2020, 12 (8), 764– 772, DOI: 10.1038/s41557-020-0473-9Google ScholarThere is no corresponding record for this reference.
- 48Xiong, Y.; Sun, W.; Xin, P.; Chen, W.; Zheng, X.; Yan, W.; Zheng, L.; Dong, J.; Zhang, J.; Wang, D.; Li, Y. Gram-Scale Synthesis of High-Loading Single-Atomic-Site Fe Catalysts for Effective Epoxidation of Styrene. Adv. Mater. 2020, 32 (34), 2000896, DOI: 10.1002/adma.202000896Google ScholarThere is no corresponding record for this reference.
- 49Xiong, Y.; Sun, W.; Han, Y.; Xin, P.; Zheng, X.; Yan, W.; Dong, J.; Zhang, J.; Wang, D.; Li, Y. Cobalt Single Atom Site Catalysts with Ultrahigh Metal Loading for Enhanced Aerobic Oxidation of Ethylbenzene. Nano Res. 2021, 14 (7), 2418– 2423, DOI: 10.1007/s12274-020-3244-4Google ScholarThere is no corresponding record for this reference.
- 50Wei, S.; Li, A.; Liu, J.-C.; Li, Z.; Chen, W.; Gong, Y.; Zhang, Q.; Cheong, W.-C.; Wang, Y.; Zheng, L.; Xiao, H.; Chen, C.; Wang, D.; Peng, Q.; Gu, L.; Han, X.; Li, J.; Li, Y. Direct Observation of Noble Metal Nanoparticles Transforming to Thermally Stable Single Atoms. Nat. Nanotechnol. 2018, 13 (9), 856– 861, DOI: 10.1038/s41565-018-0197-9Google ScholarThere is no corresponding record for this reference.
- 51Hou, Z.; Lu, Y.; Liu, Y.; Liu, N.; Hu, J.; Wei, L.; Li, Z.; Tian, X.; Gao, R.; Yu, X.; Feng, Y.; Wu, L.; Deng, J.; Wang, D.; Sui, M.; Dai, H.; Li, Y. A General Dual-Metal Nanocrystal Dissociation Strategy to Generate Robust High-Temperature-Stable Alumina-Supported Single-Atom Catalysts. J. Am. Chem. Soc. 2023, 145 (29), 15869– 15878, DOI: 10.1021/jacs.3c02909Google ScholarThere is no corresponding record for this reference.
- 52Liang, X.; Yao, S.; Li, Z.; Li, Y. Challenge and Chance of Single Atom Catalysis: The Development and Application of the Single Atom Site Catalysts Toolbox. Acc. Chem. Res. 2025, 58 (10), 1607– 1619, DOI: 10.1021/acs.accounts.4c00857Google ScholarThere is no corresponding record for this reference.
- 53Li, Y.; Xu, C.-Q.; Chen, C.; Zhang, Y.; Liu, S.; Zhuang, Z.; Zhang, Y.; Zhang, Q.; Li, Z.; Chen, Z.; Zheng, L.; Cheong, W.-C.; Wu, K.; Jiang, G.; Xiao, H.; Lian, C.; Wang, D.; Peng, Q.; Li, J.; Li, Y. Carbon-Boosted and Nitrogen-Stabilized Isolated Single-Atom Sites for Direct Dehydrogenation of Lower Alkanes. J. Am. Chem. Soc. 2024, 146 (30), 20668– 20677, DOI: 10.1021/jacs.4c03048Google ScholarThere is no corresponding record for this reference.
- 54Wang, X.; Fu, N.; Liu, J.-C.; Yu, K.; Li, Z.; Xu, Z.; Liang, X.; Zhu, P.; Ye, C.; Zhou, A.; Li, A.; Zheng, L.; Liu, L.-M.; Chen, C.; Wang, D.; Peng, Q.; Li, Y. Atomic Replacement of PtNi Nanoalloys within Zn-ZIF-8 for the Fabrication of a Multisite CO2 Reduction Electrocatalyst. J. Am. Chem. Soc. 2022, 144 (50), 23223– 23229, DOI: 10.1021/jacs.2c11497Google ScholarThere is no corresponding record for this reference.
- 55Dutta, N.; Giri, B.; Riyaz, M.; Midya, S.; Singh, A.; Bagchi, D.; Mondal, S.; Kediya, S.; Singh, A. K.; Chakraborty, S.; Singh, A. K.; Peter, S. C. Confining Reaction Intermediates in Oxide-Derived Hollow Cu-Zn Bimetallic Catalyst Facilitates Selective Formation of C2+ Alcohols from Electrochemical Carbon Dioxide Reduction. Angew. Chem., Int. Ed. 2026, 65, e23150 DOI: 10.1002/anie.202523150Google ScholarThere is no corresponding record for this reference.
- 56Guo, W.; Wang, X.; Ma, Y.; Song, Y.; Chang, L.; Wu, H.; Li, G.; Li, Z.; Xin, Y.; He, M.; Zhang, J.; Yang, T.; Zhu, M.; Shen, H.; Xi, S.; Wang, X.; Gan, L.; Jiang, Q.; Xia, C.; Zhao, S.; Guo, Z.; Wang, Z.; Tang, B. Z.; Ye, R. Local Coordination-Dependent CO2 Reduction Activity of Bimetallic Cu-Al Catalysts for Selective Ethylene/Ethanol Electrosynthesis. Angew. Chem., Int. Ed. 2026, 65, e20291 DOI: 10.1002/anie.202520291Google ScholarThere is no corresponding record for this reference.
- 57Cao, X.; Liu, Y.; Yu, R.; Yang, Y.; Li, A.; Li, X.; Mao, J. Breaking Ampere-Level Current Density Limitations in Multi-Carbon Products through Crystal Facet Engineering. Chem. 2025, 0 (0), 102824, DOI: 10.1016/j.chempr.2025.102824Google ScholarThere is no corresponding record for this reference.
- 58Yan, Z.; Gao, P.; Li, Z.; Zhang, Y.; Hu, C.; Cao, D.; Cheng, D. Rational Design and Validation of Cu2O/Cu Heterogeneous Interface Catalyst for CO2 Electroreduction to C2+ Products. Small 2025, 21 (25), 2500950, DOI: 10.1002/smll.202500950Google ScholarThere is no corresponding record for this reference.
- 59Wang, H.; Yang, H. X.; Xu, Y. N.; Fu, H. Q.; Li, X. Y.; He, J. J.; Niu, Q.; Wu, J. C.; Yuan, H. Y.; Liu, P. F.; Yang, H. G. Retained Oxygen Regulation in Oxide-Derived Copper for Promoted CO2 Electroreduction Toward Multicarbon Products. Angew. Chem., Int. Ed. 2025, 64 (25), e202423889 DOI: 10.1002/anie.202423889Google ScholarThere is no corresponding record for this reference.
- 60Arán-Ais, R. M.; Scholten, F.; Kunze, S.; Rizo, R.; Roldan Cuenya, B. The Role of in Situ Generated Morphological Motifs and Cu(i) Species in C2+ Product Selectivity during CO2 Pulsed Electroreduction. Nat. Energy 2020, 5 (4), 317– 325, DOI: 10.1038/s41560-020-0594-9Google ScholarThere is no corresponding record for this reference.
- 61Li, L.; Yao, S.; Feng, Y.; Gao, Z.; Wang, J.; Yin, P.; Liu, H.; Zhang, Z. Grain Boundary-Induced Stabilization of Bi3+/Bi0 Coexistence in β-Bi2O3 for Highly Efficient Electrochemical CO2 Reduction to Formate. Appl. Catal. B: Environ. 2025, 372, 125302, DOI: 10.1016/j.apcatb.2025.125302Google ScholarThere is no corresponding record for this reference.
- 62Fu, X.; Guo, X.; Shi, P.; Frauenheim, T.; Novoselov, K. S.; Yuan, M.; Wang, M. Control of CO2 Electrocatalysis via Modularly Customizable Graphdiyne. J. Am. Chem. Soc. 2025, 147 (46), 42394– 42405, DOI: 10.1021/jacs.5c11774Google ScholarThere is no corresponding record for this reference.
- 63Wang, C.; Zhang, G.; Luo, R.; Wang, Y.; Ma, X.; Zhang, M.; Chang, X.; Zhao, Z.-J.; Wang, T.; Gong, J. Selective CO2 Reduction to Acetate via Controlled sp2/sp3 Carbon Hybridization. Nat. Commun. 2025, 16 (1), 10506, DOI: 10.1038/s41467-025-65504-6Google ScholarThere is no corresponding record for this reference.
- 64Wang, L.; Wang, D.; Li, Y. Single-Atom Catalysis for Carbon Neutrality. Carbon Energy 2022, 4 (6), 1021– 1079, DOI: 10.1002/cey2.194Google ScholarThere is no corresponding record for this reference.
- 65Li, H.; Fang, L.; Wang, T.; Bai, R.; Zhang, J.; Li, T.; Duan, Z.; Chen, K.-J.; Pan, F. In Situ Modulated Nickel Single Atoms on Bicontinuous Porous Carbon Fibers and Sheets Networks for Acidic CO2 Reduction. Adv. Mater. 2025, 37 (7), 2416337, DOI: 10.1002/adma.202416337Google ScholarThere is no corresponding record for this reference.
- 66Ma, Y.; Xiao, T.; Zhu, K.; Zhang, W.; Yin, Z.; Dong, A.; Sun, Z.; Zhao, D.; Li, W. Industry-Level Electrocatalytic CO2 to CO Enabled by 2D Mesoporous Ni Single Atom Catalysts. Angew. Chem., Int. Ed. 2025, 64 (5), e202416629 DOI: 10.1002/anie.202416629Google ScholarThere is no corresponding record for this reference.
- 67Li, P.; Mao, Y.; Shin, H.; Yang, Q.; Cheng, X.; Li, Y.; Li, K.; Yu, H.; Mulder, R.; Pang, W. K.; Jin, H.; Zhao, Y.; Zheng, Z.; Finch, E.; Hearn, K.; Jia, B.; Waterhouse, G. I. N.; Wang, Z.; Ma, T. Tandem Amine Scrubbing and CO2 Electrolysis via Direct Piperazine Carbamate Reduction. Nat. Energy 2025, 10 (10), 1262– 1273, DOI: 10.1038/s41560-025-01869-8Google ScholarThere is no corresponding record for this reference.
- 68Huang, L.; Gao, G.; Zhao, J.; Roberts, W. L.; Lu, X. Electrocatalytic Upcycling of High-Pressure Captured CO2 to Ethylene. Nat. Catal. 2025, 8 (9), 968– 976, DOI: 10.1038/s41929-025-01411-9Google ScholarThere is no corresponding record for this reference.
- 69Wang, M.; Fang, M.; Liu, Y.; Chen, C.; Zhang, Y.; Jia, S.; Wu, H.; He, M.; Han, B. Enhanced Intermediates Inter-Migration on Ag Single-Atom Alloys for Boosting Multicarbon Product Selectivity in CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (19), 16450– 16458, DOI: 10.1021/jacs.5c03057Google ScholarThere is no corresponding record for this reference.
- 70Zhao, Y.; Wang, Y.; Yu, Z.; Song, C.; Wang, J.; Huang, H.; Meng, L.; Liu, M.; Liu, L. Gold Single Atom Doped Defective Nanoporous Copper Octahedrons for Electrocatalytic Reduction of Carbon Dioxide to Ethylene. ACS Nano 2025, 19 (4), 4505– 4514, DOI: 10.1021/acsnano.4c13961Google ScholarThere is no corresponding record for this reference.
- 71Liu, Y.; Ren, X.; Wang, J.; Wang, H.; Yin, Z.; Wang, Y.; Huang, W.; Hu, X.; Xu, Z. J.; Deng, Y. Spin-State Effect of Tetrahedron-Coordinated Single-Atom Catalysts on CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (24), 20318– 20328, DOI: 10.1021/jacs.4c18550Google ScholarThere is no corresponding record for this reference.
- 72Zhang, Y.; Chen, F.; Yang, X.; Guo, Y.; Zhang, X.; Dong, H.; Wang, W.; Lu, F.; Lu, Z.; Liu, H.; Liu, H.; Xiao, Y.; Cheng, Y. Electronic Metal-Support Interaction Modulates Cu Electronic Structures for CO2 Electroreduction to Desired Products. Nat. Commun. 2025, 16 (1), 1956, DOI: 10.1038/s41467-025-57307-6Google ScholarThere is no corresponding record for this reference.
- 73Jeon, S.; Nedzbala, H. S.; Huffman, B. L.; Pearce, A. J.; Donley, C. L.; Jia, X.; Bein, G. P.; Choi, J.; Durand, N.; Atallah, H.; Castellano, F. N.; Dempsey, J. L.; Mayer, J. M.; Hazari, N.; Stach, E. A. Statistical Analysis of HAADF-STEM Images to Determine the Surface Coverage and Distribution of Immobilized Molecular Complexes. Matter 2025, 8 (2), 101919, DOI: 10.1016/j.matt.2024.11.013Google ScholarThere is no corresponding record for this reference.
- 74Chantler, C. T.; Bunker, G.; D’Angelo, P.; Diaz-Moreno, S. X-Ray Absorption Spectroscopy. Nat. Rev. Methods Primers 2024, 4 (1), 89, DOI: 10.1038/s43586-024-00366-8Google ScholarThere is no corresponding record for this reference.
- 75Greenwood, N. N. Mössbauer Spectra and Chemical Structure. Nature 1965, 205 (4967), 119– 119, DOI: 10.1038/205119a0Google ScholarThere is no corresponding record for this reference.
- 76Beniya, A.; Higashi, S. Towards Dense Single-Atom Catalysts for Future Automotive Applications. Nat. Catal. 2019, 2 (7), 590– 602, DOI: 10.1038/s41929-019-0282-yGoogle ScholarThere is no corresponding record for this reference.
- 77Liu, Y.; Yue, Z.; Jin, C.; Zheng, L.; Shi, J.; Li, D.; Wang, Y.; Bai, J.; Leng, K.; Wang, W.; Qu, Y.; Li, Q. Isolated Tin Enhanced CO Coverage-Regulation on Sn1Cu Alloy for Selective CO2 Electroreduction to C2+ Products. Small 2025, 21 (8), 2409259, DOI: 10.1002/smll.202409259Google ScholarThere is no corresponding record for this reference.
- 78Xu, Y. N.; Li, J.; Wu, J. C.; Li, W.; Yang, Y.; Wu, H.; Fu, H. Q.; Zhu, M.; Wang, X. L.; Dai, S.; Lian, C.; Liu, P. F.; Yang, H. G. Orbital Matching Mechanism-Guided Synthesis of Cu-Based Single Atom Alloys for Acidic CO2 Electroreduction. Adv. Mater. 2025, 37 (18), 2500343, DOI: 10.1002/adma.202500343Google ScholarThere is no corresponding record for this reference.
- 79Du, J.-H.; Liu, Z.; Sheng, T.; Liu, J.; Ye, J.; Li, Y.; Chen, S.; Zhang, Y.; Gu, X. Host-Guest Metal Interaction in Cu-In Single Atom Alloy Switching Electrocatalytic CO2 Reduction Pathway. Angew. Chem., Int. Ed. 2025, 64 (43), e202512970 DOI: 10.1002/anie.202512970Google ScholarThere is no corresponding record for this reference.
- 80Choi, J.; Kim, S.; Choi, J. Y.; Park, S.; Je, K.; Liu, S.; Jiang, J.; Yang, S.; Pollock, C. J.; Guzman-Soriano, R.; Bairley, K.; Ahmad, B. I. Z.; Milner, P. J.; Thiede, E. H.; Hwang, Y. J.; Park, J.; Yang, Y. Dynamic Evolution from Single-Atom Catalysts to Active Nanograins for CO2 Reduction. J. Am. Chem. Soc. 2025, 147 (41), 37808– 37818, DOI: 10.1021/jacs.5c14123Google ScholarThere is no corresponding record for this reference.
- 81Mei, Z.; He, Y.; Liu, K.; Luo, W.; Tan, Y.; Chen, Q.; Wang, H.; Guo, X.; Wu, Q.; Ma, C.; Fu, J.; Lin, Z.; Liu, M. Enhanced *COOH Adsorption over Edge-Rich Ni-N4 Sites for Efficient Acidic CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (37), 34101– 34108, DOI: 10.1021/jacs.5c12583Google ScholarThere is no corresponding record for this reference.
- 82Lu, T.; Shi, G.; Liu, Y.; Luo, X.; Shen, Y.; Chang, M.; Wu, Y.; Gao, X.; Wu, J.; Li, Y.; Wang, Y.; Zhang, L. Local Coordination Environment-Driven Structural Dynamics of Single-Atom Copper and the CO2 Electroreduction Pathway. J. Am. Chem. Soc. 2025, 147 (30), 26425– 26436, DOI: 10.1021/jacs.5c05984Google ScholarThere is no corresponding record for this reference.
- 83Yang, Z.; Zhang, J.; Tan, X.; Zhu, H.; Xu, L.; Wu, X.; Xu, Z.; Guo, J.; Jia, D.; Chen, C. Manipulating the Coordination Environment of Cu Single Atoms via Regulating ZrO2 Support Crystal Phases for Enhanced CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (39), 35985– 35994, DOI: 10.1021/jacs.5c13661Google ScholarThere is no corresponding record for this reference.
- 84Daiyan, R.; Zhu, X.; Tong, Z.; Gong, L.; Razmjou, A.; Liu, R.-S.; Xia, Z.; Lu, X.; Dai, L.; Amal, R. Transforming Active Sites in Nickel-Nitrogen-Carbon Catalysts for Efficient Electrochemical CO2 Reduction to CO. Nano Energy 2020, 78, 105213, DOI: 10.1016/j.nanoen.2020.105213Google ScholarThere is no corresponding record for this reference.
- 85Hao, Q.; Tang, Q.; Zheng, L.; Liu, K.; Wu, J.; Lu, J. Specific Construction of Asymmetric Carbon-Nickel-Chlorine Single-Atom Sites via Carbon Vacancy Engineering for Efficient CO2 Electroreduction. Nat. Commun. 2025, 16 (1), 8365, DOI: 10.1038/s41467-025-62287-8Google ScholarThere is no corresponding record for this reference.
- 86Zou, H.; Zhao, G.; Dai, H.; Dong, H.; Luo, W.; Wang, L.; Lu, Z.; Luo, Y.; Zhang, G.; Duan, L. Electronic Perturbation of Copper Single-Atom CO2 Reduction Catalysts in a Molecular Way. Angew. Chem., Int. Ed. 2023, 62 (6), e202217220 DOI: 10.1002/anie.202217220Google ScholarThere is no corresponding record for this reference.
- 87Chen, R.; Zhao, J.; Zhang, X.; Zhao, Q.; Li, Y.; Cui, Y.; Zhong, M.; Wang, J.; Li, X.; Huang, Y.; Liu, B. Visualizing Catalytic Dynamics of Single-Cu-Atom-Modified SnS2 in CO2 Electroreduction via Rapid Freeze-Quench Mössbauer Spectroscopy. J. Am. Chem. Soc. 2024, 146 (35), 24368– 24376, DOI: 10.1021/jacs.4c05813Google ScholarThere is no corresponding record for this reference.
- 88Liao, L.; Xia, G.; Yu, F.; Liu, X.; Shu, M.; Zhang, G.; Zeng, X.; Wang, H. Saturated Coordination Lu-N6 Defect Sites for Highly Efficient Electroreduction of CO2. Small 2023, 19 (37), 2300926, DOI: 10.1002/smll.202300926Google ScholarThere is no corresponding record for this reference.
- 89Wang, H.; Sui, H.; Ding, Y.; Yang, Y.; Su, Y.; Li, H. Tailoring CO2 Adsorption Configuration with Spatial Confinement Switches Electroreduction Product from Formate to Acetate. J. Am. Chem. Soc. 2025, 147 (7), 6095– 6107, DOI: 10.1021/jacs.4c17295Google ScholarThere is no corresponding record for this reference.
- 90Song, J.; Lei, X.; Mu, J.; Li, J.; Song, X.; Yan, L.; Ding, Y. Chlorine-Coordinated Unsaturated Ni-N2 Sites for Efficient Electrochemical Carbon Dioxide Reduction. Small 2023, 19 (52), 2304423, DOI: 10.1002/smll.202304423Google ScholarThere is no corresponding record for this reference.
- 91Qu, M.; Chen, Z.; Sun, Z.; Zhou, D.; Xu, W.; Tang, H.; Gu, H.; Liang, T.; Hu, P.; Li, G.; Wang, Y.; Chen, Z.; Wang, T.; Jia, B. Rational Design of Asymmetric Atomic Ni-P1N3 Active Sites for Promoting Electrochemical CO2 Reduction. Nano Res. 2023, 16 (2), 2170– 2176, DOI: 10.1007/s12274-022-4969-zGoogle ScholarThere is no corresponding record for this reference.
- 92Song, W.; Xiao, C.; Ding, J.; Huang, Z.; Yang, X.; Zhang, T.; Mitlin, D.; Hu, W. Review of Carbon Support Coordination Environments for Single Metal Atom Electrocatalysts (SACS). Adv. Mater. 2024, 36 (1), 2301477, DOI: 10.1002/adma.202301477Google ScholarThere is no corresponding record for this reference.
- 93Bhawnani, R. R.; Sartape, R.; Gande, V. V.; Barsoum, M. L.; Kallon, E. M.; dos Reis, R.; Dravid, V. P.; Singh, M. R. Non-Aqueous Electrochemical CO2 Reduction to Multivariate C2-Products Over Single Atom Catalyst at Current Density up to 100 mA cm–2. Small 2025, 21 (18), 2408010, DOI: 10.1002/smll.202408010Google ScholarThere is no corresponding record for this reference.
- 94Ma, G.; Chen, Y.; Li, H.; Sun, Y.; Fan, H.; Jiang, S.; Cui, X.; Ma, T. Hierarchically Porous N-Doped Carbon Confined Single-Atom Fe Catalyst for Efficient Electrochemical CO2 Reduction. Nano Res. 2025, 18 (4), 94907297, DOI: 10.26599/NR.2025.94907297Google ScholarThere is no corresponding record for this reference.
- 95Yin, Z.; Yu, J.; Xie, Z.; Yu, S.-W.; Zhang, L.; Akauola, T.; Chen, J. G.; Huang, W.; Qi, L.; Zhang, S. Hybrid Catalyst Coupling Single-Atom Ni and Nanoscale Cu for Efficient CO2 Electroreduction to Ethylene. J. Am. Chem. Soc. 2022, 144 (45), 20931– 20938, DOI: 10.1021/jacs.2c09773Google ScholarThere is no corresponding record for this reference.
- 96Chen, S.; Wang, B.; Zhu, J.; Wang, L.; Ou, H.; Zhang, Z.; Liang, X.; Zheng, L.; Zhou, L.; Su, Y.-Q.; Wang, D.; Li, Y. Lewis Acid Site-Promoted Single-Atomic Cu Catalyzes Electrochemical CO2 Methanation. Nano Lett. 2021, 21 (17), 7325– 7331, DOI: 10.1021/acs.nanolett.1c02502Google ScholarThere is no corresponding record for this reference.
- 97Liu, K.; Sun, Z.; Chen, W.; Lang, X.; Gao, X.; Chen, P. Ultra-Fast Pulsed Discharge Preparation of Coordinatively Unsaturated Asymmetric Copper Single-Atom Catalysts for CO2 Reduction. Adv. Funct. Mater. 2024, 34 (16), 2312589, DOI: 10.1002/adfm.202312589Google ScholarThere is no corresponding record for this reference.
- 98Zheng, J.; Xu, S.; Sun, L.; Pan, X.; Xie, Q.; Zhao, G. Boosting the Reduction of CO2 and Dimethylamine for C-N Bonding to Synthesize DMF via Modulating the Electronic Structures of Indium Single Atoms. Energy Environ. Sci. 2025, 18 (8), 3614– 3622, DOI: 10.1039/D4EE05681GGoogle ScholarThere is no corresponding record for this reference.
- 99Ping, D.; Huang, S.; Wu, S.; Zhang, Y.; Wang, S.; Yang, X.; Han, L.; Tian, J.; Guo, D.; Qiu, H.-J.; Fang, S. Confinement Effect and 3D Design Endow Unsaturated Single Ni Atoms with Ultrahigh Stability and Selectivity toward CO2 Electroreduction. Small 2024, 20 (14), 2309014, DOI: 10.1002/smll.202309014Google ScholarThere is no corresponding record for this reference.
- 100Xia, W.; Xie, Y.; Jia, S.; Han, S.; Qi, R.; Chen, T.; Xing, X.; Yao, T.; Zhou, D.; Dong, X.; Zhai, J.; Li, J.; He, J.; Jiang, D.; Yamauchi, Y.; He, M.; Wu, H.; Han, B. Adjacent Copper Single Atoms Promote C-C Coupling in Electrochemical CO2 Reduction for the Efficient Conversion of Ethanol. J. Am. Chem. Soc. 2023, 145 (31), 17253– 17264, DOI: 10.1021/jacs.3c04612Google ScholarThere is no corresponding record for this reference.
- 101Chen, Z.; Wang, C.; Zhong, X.; Lei, H.; Li, J.; Ji, Y.; Liu, C.; Ding, M.; Dai, Y.; Li, X.; Zheng, T.; Jiang, Q.; Peng, H.-J.; Xia, C. Achieving Efficient CO2 Electrolysis to CO by Local Coordination Manipulation of Nickel Single-Atom Catalysts. Nano Lett. 2023, 23 (15), 7046– 7053, DOI: 10.1021/acs.nanolett.3c01808Google ScholarThere is no corresponding record for this reference.
- 102Wang, Z.; Chen, Z.; Du, X.; Zhang, Y.; Liu, Z.; Zang, S.-Q. Modulating the Electronic Properties of Single Ni Atom Catalyst via First-Shell Coordination Engineering to Boost Electrocatalytic Flue Gas CO2 Reduction. Adv. Funct. Mater. 2025, 35 (20), 2420994, DOI: 10.1002/adfm.202420994Google ScholarThere is no corresponding record for this reference.
- 103Zang, Y.; Liu, Y.; Lu, R.; Yang, Q.; Wang, B.; Zhang, M.; Mao, Y.; Wang, Z.; Lum, Y. Tuning Transition Metal 3d Spin State on Single-Atom Catalysts for Selective Electrochemical CO2 Reduction. Adv. Mater. 2025, 37 (16), 2417034, DOI: 10.1002/adma.202417034Google ScholarThere is no corresponding record for this reference.
- 104Chen, Y.; Pan, X.; Li, L.; Chen, M.; Cao, H.; Zhao, Y.; Wang, X.; Lin, J. Modulating Electronic Density of Single-Atom Ni Center by Heteroatoms for Efficient CO2 Electroreduction. Small 2025, 21 (9), 2411249, DOI: 10.1002/smll.202411249Google ScholarThere is no corresponding record for this reference.
- 105Dai, Y.; Li, H.; Wang, C.; Xue, W.; Zhang, M.; Zhao, D.; Xue, J.; Li, J.; Luo, L.; Liu, C.; Li, X.; Cui, P.; Jiang, Q.; Zheng, T.; Gu, S.; Zhang, Y.; Xiao, J.; Xia, C.; Zeng, J. Manipulating Local Coordination of Copper Single Atom Catalyst Enables Efficient CO2-to-CH4 Conversion. Nat. Commun. 2023, 14 (1), 3382, DOI: 10.1038/s41467-023-39048-6Google ScholarThere is no corresponding record for this reference.
- 106Song, J.; Lei, X.; Mu, J.; Li, J.; Song, X.; Yan, L.; Ding, Y. Boron-Doped Nickel-Nitrogen-Carbon Single-Atom Catalyst for Boosting Electrochemical CO2 Reduction. Small 2023, 19 (52), 2305666, DOI: 10.1002/smll.202305666Google ScholarThere is no corresponding record for this reference.
- 107Guo, J.; Zhang, W.; Zhang, L.-H.; Chen, D.; Zhan, J.; Wang, X.; Shiju, N. R.; Yu, F. Control over Electrochemical CO2 Reduction Selectivity by Coordination Engineering of Tin Single-Atom Catalysts. Adv. Sci. 2021, 8 (23), 2102884, DOI: 10.1002/advs.202102884Google ScholarThere is no corresponding record for this reference.
- 108Wang, Q.; Dai, M.; Li, H.; Lu, Y.-R.; Chan, T.-S.; Ma, C.; Liu, K.; Fu, J.; Liao, W.; Chen, S.; Pensa, E.; Wang, Y.; Zhang, S.; Sun, Y.; Cortés, E.; Liu, M. Asymmetric Coordination Induces Electron Localization at Ca Sites for Robust CO2 Electroreduction to CO. Adv. Mater. 2023, 35 (21), 2300695, DOI: 10.1002/adma.202300695Google ScholarThere is no corresponding record for this reference.
- 109Wei, S.; Zhu, J.; Chen, X.; Yang, R.; Gu, K.; Li, L.; Chiang, C.-Y.; Mai, L.; Chen, S. Planar Chlorination Engineering Induced Symmetry-Broken Single-Atom Site Catalyst for Enhanced CO2 Electroreduction. Nat. Commun. 2025, 16 (1), 1652, DOI: 10.1038/s41467-025-56271-5Google ScholarThere is no corresponding record for this reference.
- 110Peng, J.-X.; Yang, W.; Jia, Z.; Jiao, L.; Jiang, H.-L. Axial Coordination Regulation of MOF-Based Single-Atom Ni Catalysts by Halogen Atoms for Enhanced CO2 Electroreduction. Nano Res. 2022, 15 (12), 10063– 10069, DOI: 10.1007/s12274-022-4467-3Google ScholarThere is no corresponding record for this reference.
- 111Zhao, J.; Chen, Y.; Liu, D.; Fai Ip, W.; Lin, J.; Wang, X.; Lin, S.; Fu, X.; Zhang, T. “Outside-in” Design of Single-Atom Catalysts: Linking Specific Peripheral Geometry to Defined CO2 Reduction Performance. Angew. Chem., Int. Ed. 2025, 64 (34), e202511184 DOI: 10.1002/anie.202511184Google ScholarThere is no corresponding record for this reference.
- 112Ni, W.; He, X.; Chen, H.; Dai, M.; Zhang, W.; Zhang, Y.; Wang, S.; Zhang, S. 93% Single-Atom Utilization in Base-Resistant Metal-Organic Framework Quantum Dots for Ampere-Level CO2 Electroreduction. Energy Environ. Sci. 2024, 17 (9), 3191– 3201, DOI: 10.1039/D4EE00720DGoogle ScholarThere is no corresponding record for this reference.
- 113Li, L.; Lei, X.; Zheng, Z.; Dong, Y.; Chen, H.; Chen, J.; Zhong, Y.; Zheng, Y.; Tang, Y.; Zhang, X.; Cheng, H.-M. Tuning Binding Strength between Single Metal Atoms and Supports Enhances Electrochemical CO2 Methanation. Nat. Commun. 2025, 16 (1), 8641, DOI: 10.1038/s41467-025-63781-9Google ScholarThere is no corresponding record for this reference.
- 114Leverett, J.; Baghestani, G.; Tran-Phu, T.; Yuwono, J. A.; Kumar, P.; Johannessen, B.; Simondson, D.; Wen, H.; Chang, S. L. Y.; Tricoli, A.; Simonov, A. N.; Dai, L.; Amal, R.; Daiyan, R.; Hocking, R. K. Direct Observation of Electron Donation onto the Reactants and a Transient Poisoning Mechanism During CO2 Electroreduction on Ni Single Atom Catalysts. Angew. Chem., Int. Ed. 2025, 64 (18), e202424087 DOI: 10.1002/anie.202424087Google ScholarThere is no corresponding record for this reference.
- 115Wang, C.; Chen, B.; Wang, Q.; Hu, H.; Xia, R.; Zhu, J.; Sun, Q.; Wu, Z.; Miao, R. K.; Guan, Q.; Liu, Y.; Liu, M.; Li, W. Dynamic Activation of Edge-Hosted Co-N4 Sites for Energy-Efficient Electrochemical CO2 Reduction at Industry-Level Current Density. ACS Catal. 2025, 15 (10), 8540– 8550, DOI: 10.1021/acscatal.5c01598Google ScholarThere is no corresponding record for this reference.
- 116Chang, Q.; Liu, Y.; Lee, J.-H.; Ologunagba, D.; Hwang, S.; Xie, Z.; Kattel, S.; Lee, J. H.; Chen, J. G. Metal-Coordinated Phthalocyanines as Platform Molecules for Understanding Isolated Metal Sites in the Electrochemical Reduction of CO2. J. Am. Chem. Soc. 2022, 144 (35), 16131– 16138, DOI: 10.1021/jacs.2c06953Google ScholarThere is no corresponding record for this reference.
- 117Liu, T.; Zhang, D.; Hirai, Y.; Ito, K.; Ishibashi, K.; Todoroki, N.; Matsuo, Y.; Yoshida, J.; Ono, S.; Li, H.; Yabu, H. Surface Charge Transfer Enhanced Cobalt-Phthalocyanine Crystals for Efficient CO2-to-CO Electroreduction with Large Current Density Exceeding 1000 mA cm–2. Adv. Sci. 2025, 12 (23), 2501459, DOI: 10.1002/advs.202501459Google ScholarThere is no corresponding record for this reference.
- 118Wang, Z.; Wang, C.; Hu, Y.; Yang, S.; Yang, J.; Chen, W.; Zhou, H.; Zhou, F.; Wang, L.; Du, J.; Li, Y.; Wu, Y. Simultaneous Diffusion of Cation and Anion to Access N, S Co-Coordinated Bi-Sites for Enhanced CO2 Electroreduction. Nano Res. 2021, 14 (8), 2790– 2796, DOI: 10.1007/s12274-021-3287-1Google ScholarThere is no corresponding record for this reference.
- 119Lu, Y.; Wang, H.; Yu, P.; Yuan, Y.; Shahbazian-Yassar, R.; Sheng, Y.; Wu, S.; Tu, W.; Liu, G.; Kraft, M.; Xu, R. Isolated Ni Single Atoms in Nitrogen Doped Ultrathin Porous Carbon Templated from Porous g-C3N4 for High-Performance CO2 Reduction. Nano Energy 2020, 77, 105158, DOI: 10.1016/j.nanoen.2020.105158Google ScholarThere is no corresponding record for this reference.
- 120Sui, R.; Wang, B.; Wang, Y.; Pei, J.; Zhu, W.; Chen, W.; Li, C.; Sun, A.; Zhuang, Z. A General and Facile Calcination Method to Synthesize Single-Site Catalysts for Highly Efficient Electrochemical CO2 Reduction. Nano Res. 2024, 17 (5), 3895– 3901, DOI: 10.1007/s12274-023-6378-3Google ScholarThere is no corresponding record for this reference.
- 121Lv, Z.; Wang, C.; Liu, Y.; Liu, R.; Zhang, F.; Feng, X.; Yang, W.; Wang, B. Improving CO2-to-C2 Conversion of Atomic CuFONC Electrocatalysts through F, O-Codrived Optimization of Local Coordination Environment. Adv. Energy Mater. 2024, 14 (21), 2400057, DOI: 10.1002/aenm.202400057Google ScholarThere is no corresponding record for this reference.
- 122Wang, Z.; Qian, J.; Cao, P.; Shou, H.; Wu, C.; Xu, X.; Wu, X.; He, Q.; Song, L. Identification of Synergies in Fe, Co-Coordinated Polyphthalocyanines Scaffolds for Electrochemical CO2 Reduction Reaction. Nano Lett. 2024, 24 (10), 3249– 3256, DOI: 10.1021/acs.nanolett.4c00306Google ScholarThere is no corresponding record for this reference.
- 123Liu, M.; Xie, Y.; Liu, F.; Dong, X.; Xia, J.; Wang, H.; Zou, R.; Peng, Y.; Zhang, C.; Xu, Q.; Hou, C.-C. Edge-Rich Graphene Nanomesh Thermally Self-Exfoliated From Metal-Organic Frameworks for Boosting CO2 Electroreduction. Adv. Funct. Mater. 2025, 35 (44), 2509429, DOI: 10.1002/adfm.202509429Google ScholarThere is no corresponding record for this reference.
- 124Miao, K.; Qin, J.; Lai, S.; Luo, M.; Kuchkaev, A.; Yakhvarov, D.; Kang, X. Spin Regulation of Nickel Single Atom Catalyst via Axial Phosphor-Coordination Achieves Near Unity CO Selectivity in Electrochemical CO2 Reduction. Adv. Funct. Mater. 2025, 35 (14), 2419989, DOI: 10.1002/adfm.202419989Google ScholarThere is no corresponding record for this reference.
- 125Guo, C.; Wang, F.; Abdukayum, A.; Chen, Q.; Chang, F.; Li, H.; An, X.; Hu, G.; Ma, Y. Axial and Asymmetric Coordination Coupling Adjust the Electronic Structure of Single-Atom Zinc Sites for Efficient Electroreduction of Carbon Dioxide. Adv. Sci. 2025, 12 (42), e09698 DOI: 10.1002/advs.202509698Google ScholarThere is no corresponding record for this reference.
- 126Wang, R.; Wang, Y.; Chen, D.; Gao, C.; Chen, Z.; Cheng, L.; Deng, Y.; Peng, Y.; Li, J. Improving CO2 Reduction Performance towards C2 Products on a Bimetallic Zn1Cux/NC Electrocatalyst. Nano Res. 2025, 18 (11), 94907659, DOI: 10.26599/NR.2025.94907659Google ScholarThere is no corresponding record for this reference.
- 127Liu, M.; Wang, Q.; Luo, T.; Herran, M.; Cao, X.; Liao, W.; Zhu, L.; Li, H.; Stefancu, A.; Lu, Y.-R.; Chan, T.-S.; Pensa, E.; Ma, C.; Zhang, S.; Xiao, R.; Cortés, E. Potential Alignment in Tandem Catalysts Enhances CO2-to-C2H4 Conversion Efficiencies. J. Am. Chem. Soc. 2024, 146 (1), 468– 475, DOI: 10.1021/jacs.3c09632Google ScholarThere is no corresponding record for this reference.
- 128Deng, Y.; Zhao, J.; Wang, S.; Chen, R.; Ding, J.; Tsai, H.-J.; Zeng, W.-J.; Hung, S.-F.; Xu, W.; Wang, J.; Jaouen, F.; Li, X.; Huang, Y.; Liu, B. Operando Spectroscopic Analysis of Axial Oxygen-Coordinated Single-Sn-Atom Sites for Electrochemical CO2 Reduction. J. Am. Chem. Soc. 2023, 145 (13), 7242– 7251, DOI: 10.1021/jacs.2c12952Google ScholarThere is no corresponding record for this reference.
- 129Yuan, L.; Li, X.; Li, G.; Peng, K.; Zhang, H.; Zeng, S.; Sun, X.; Zhang, X. Gram-Scale Preparation of Tri-Coordinated Single-Atom Catalysts for CO2 Electrolysis in Large-Scale Membrane Electrode Assembly. Adv. Sci. 2025, 12 (18), 2500368, DOI: 10.1002/advs.202500368Google ScholarThere is no corresponding record for this reference.
- 130Wang, Y.; Zhu, P.; Wang, R.; Matthews, K. C.; Xie, M.; Wang, M.; Qiu, C.; Liu, Y.; Zhou, H.; Warner, J. H.; Liu, Y.; Wang, H.; Yu, G. Fluorine-Tuned Carbon-Based Nickel Single-Atom Catalysts for Scalable and Highly Efficient CO2 Electrocatalytic Reduction. ACS Nano 2024, 18 (39), 26751– 26758, DOI: 10.1021/acsnano.4c06923Google ScholarThere is no corresponding record for this reference.
- 131Li, Y.; Adli, N. M.; Shan, W.; Wang, M.; Zachman, M. J.; Hwang, S.; Tabassum, H.; Karakalos, S.; Feng, Z.; Wang, G.; Li, Y. C.; Wu, G. Atomically Dispersed Single Ni Site Catalysts for High-Efficiency CO2 Electroreduction at Industrial-Level Current Densities. Energy Environ. Sci. 2022, 15 (5), 2108– 2119, DOI: 10.1039/D2EE00318JGoogle ScholarThere is no corresponding record for this reference.
- 132He, Y.; Li, Y.; Zhang, J.; Wang, S.; Huang, D.; Yang, G.; Yi, X.; Lin, H.; Han, X.; Hu, W.; Deng, Y.; Ye, J. Low-Temperature Strategy toward Ni-NC@Ni Core-Shell Nanostructure with Single-Ni Sites for Efficient CO2 Electroreduction. Nano Energy 2020, 77, 105010, DOI: 10.1016/j.nanoen.2020.105010Google ScholarThere is no corresponding record for this reference.
- 133Dong, J.; Liu, Y.; Pei, J.; Li, H.; Ji, S.; Shi, L.; Zhang, Y.; Li, C.; Tang, C.; Liao, J.; Xu, S.; Zhang, H.; Li, Q.; Zhao, S. Continuous Electroproduction of Formate via CO2 Reduction on Local Symmetry-Broken Single-Atom Catalysts. Nat. Commun. 2023, 14 (1), 6849, DOI: 10.1038/s41467-023-42539-1Google ScholarThere is no corresponding record for this reference.
- 134Huang, Y.; Hu, H.; Wang, C.; Yuan, X.; Wu, X.; Qiao, Z.; Li, Y.; Wang, C. Oxygen-Bearing Defects in Graphene-Supported M-N-C Single-Atom Catalysts: A Hidden Determinant of Catalytic Performance. ACS Catal. 2025, 15 (17), 15358– 15371, DOI: 10.1021/acscatal.5c03974Google ScholarThere is no corresponding record for this reference.
- 135Yang, L.; Pang, H.; Ren, W.; Wang, X.; Wei, Y.; Lu, P.; Chen, J.; Tian, W.; Huang, M.; Wang, H. Joule Heating Driven Graphitization Regulation and Ni Single-Atom Modification in Hard Carbon for Low-Voltage and High-Rate Potassium-Ion Storage. Adv. Funct. Mater. 2026, 36 (6), e16237 DOI: 10.1002/adfm.202516237Google ScholarThere is no corresponding record for this reference.
- 136Wu, Y.; Zhuang, Z.; Chen, C.; Li, J.; Xiao, F.; Chen, C. Atomic-Level Regulation Strategies of Single-Atom Catalysts: Nonmetal Heteroatom Doping and Polymetallic Active Site Construction. Chem. Catalysis 2023, 3 (7), 100586, DOI: 10.1016/j.checat.2023.100586Google ScholarThere is no corresponding record for this reference.
- 137Ruan, Y.; Guo, W.; Lu, Y.; Zhou, H.; Wu, Y. Recent Advances in ″Top-down″ Synthesis Strategies for Single-Atom Catalysts. Sci. China Chem. 2025, 68, 6248– 6271, DOI: 10.1007/s11426-025-3120-4Google ScholarThere is no corresponding record for this reference.
- 138Cai, R.; Zhu, H.; Yang, F.; Ju, M.; Huang, X.; Wang, J.; Gu, M. D.; Gao, J.; Yang, S. The Proximal Protonation Source in Cu-NHx-C Single Atom Catalysts Selectively Boosts CO2 to Methane Electroreduction. Angew. Chem., Int. Ed. 2025, 64 (15), e202424098 DOI: 10.1002/anie.202424098Google ScholarThere is no corresponding record for this reference.
- 139Wu, X.; Zhao, J. Y.; Sun, J. W.; Li, W. J.; Yuan, H. Y.; Liu, P. F.; Dai, S.; Yang, H. G. Isolation of Highly Reactive Cobalt Phthalocyanine via Electrochemical Activation for Enhanced CO2 Reduction Reaction. Small 2023, 19 (23), 2207037, DOI: 10.1002/smll.202207037Google ScholarThere is no corresponding record for this reference.
- 140He, C.; Gong, Y.; Li, S.; Wu, J.; Lu, Z.; Li, Q.; Wang, L.; Wu, S.; Zhang, J. Single-Atom Alloys Materials for CO2 and CH4 Catalytic Conversion. Adv. Mater. 2024, 36 (16), 2311628, DOI: 10.1002/adma.202311628Google ScholarThere is no corresponding record for this reference.
- 141Ji, S.; Chen, Y.; Wang, X.; Zhang, Z.; Wang, D.; Li, Y. Chemical Synthesis of Single Atomic Site Catalysts. Chem. Rev. 2020, 120 (21), 11900– 11955, DOI: 10.1021/acs.chemrev.9b00818Google ScholarThere is no corresponding record for this reference.
- 142Chhetri, M.; Wan, M.; Jin, Z.; Yeager, J.; Sandor, C.; Rapp, C.; Wang, H.; Lee, S.; Bodenschatz, C. J.; Zachman, M. J.; Che, F.; Yang, M. Dual-Site Catalysts Featuring Platinum-Group-Metal Atoms on Copper Shapes Boost Hydrocarbon Formations in Electrocatalytic CO2 Reduction. Nat. Commun. 2023, 14 (1), 3075, DOI: 10.1038/s41467-023-38777-yGoogle ScholarThere is no corresponding record for this reference.
- 143Liu, X.; Zhen, C.; Wu, J.; You, X.; Wu, Y.; Hao, Q.; Yu, G.; Gu, M. D.; Liu, K.; Lu, J. Modulating the Hydrogenation Mechanism of Electrochemical CO2 Reduction Using Ruthenium Atomic Species on Bismuth. Adv. Funct. Mater. 2024, 34 (44), 2405835, DOI: 10.1002/adfm.202405835Google ScholarThere is no corresponding record for this reference.
- 144Jin, C.; Lin, Y.; Wang, Y.; Shi, J.; Li, R.; Liu, Y.; Yue, Z.; Leng, K.; Zhao, Y.; Wang, Y.; Han, X.; Qu, Y.; Bai, J. Engineering Atom-Scale Cascade Catalysis via Multi-Active Site Collaboration for Ampere-Level CO2 Electroreduction to C2+ Products. Adv. Mater. 2025, 37 (8), 2412658, DOI: 10.1002/adma.202412658Google ScholarThere is no corresponding record for this reference.
- 145Tuo, Y.; Lu, Q.; Liu, W.; Wang, M.; Zhou, Y.; Feng, X.; Wu, M.; Chen, D.; Zhang, J. Atomic Zn-Doping Induced Sabatier Optimum in NiZn0.03 Catalyst for CO2 Electroreduction at Industrial-Level Current Densities. Small 2024, 20 (9), 2306945, DOI: 10.1002/smll.202306945Google ScholarThere is no corresponding record for this reference.
- 146Chen, D.; Chu, B.; Li, F.; Zheng, Y.-T.; Lu, Y.; Shao, B.; Li, L.; Huang, N.-Y.; Xu, Q. Synergistic Catalysis by Cu Single Atoms and Atomically Cu-Doped Au Nanoparticles in a Metal-Organic Framework for Photocatalytic CO2 Reduction to C2H6. J. Am. Chem. Soc. 2025, 147 (26), 22705– 22713, DOI: 10.1021/jacs.5c04364Google ScholarThere is no corresponding record for this reference.
- 147Yuan, H.; Jiang, D.; Li, Z.; Liu, X.; Tang, Z.; Zhang, X.; Zhao, L.; Huang, M.; Liu, H.; Song, K.; Zhou, W. Laser Synthesis of PtMo Single-Atom Alloy Electrode for Ultralow Voltage Hydrogen Generation. Adv. Mater. 2024, 36 (5), 2305375, DOI: 10.1002/adma.202305375Google ScholarThere is no corresponding record for this reference.
- 148Wang, H.; Li, J.; Huang, M.; Cui, J.; Cheng, Z.; Yu, R.; Zhu, H. Single-Atom Alloys Prepared by Two-Step Thermal Evaporation. Nano Res. 2024, 17 (4), 2808– 2813, DOI: 10.1007/s12274-023-6146-4Google ScholarThere is no corresponding record for this reference.
- 149Yang, J.; Li, W.; Wang, D.; Li, Y. Electronic Metal-Support Interaction of Single-Atom Catalysts and Applications in Electrocatalysis. Adv. Mater. 2020, 32 (49), 2003300, DOI: 10.1002/adma.202003300Google ScholarThere is no corresponding record for this reference.
- 150Xia, C.; Wang, X.; He, C.; Qi, R.; Zhu, D.; Lu, R.; Li, F.-M.; Chen, Y.; Chen, S.; You, B.; Yao, T.; Guo, W.; Song, F.; Wang, Z.; Xia, B. Y. Highly Selective Electrocatalytic CO2 Conversion to Tailored Products through Precise Regulation of Hydrogenation and C-C Coupling. J. Am. Chem. Soc. 2024, 146 (29), 20530– 20538, DOI: 10.1021/jacs.4c07502Google ScholarThere is no corresponding record for this reference.
- 151Ding, G.; Yu, Y.; Yan, D.; Zhang, J.; Shuai, L.; Chen, L.; Liao, G. Pt-Activated Surface Nucleophilicity on High-Entropy Oxides Enables Superior Biomass Upgrading. Adv. Funct. Mater. 2025, e19698 DOI: 10.1002/adfm.202519698Google ScholarThere is no corresponding record for this reference.
- 152Hu, F.; He, B.; Chen, K.; Ma, W.; Huang, Y.; Zhao, S.; Chen, Y.; Zhao, L. Single-Atom Anchored on Perovskite With Strong Metal-Oxide Interaction for Efficient High Temperature CO2 Electrolysis. Adv. Mater. 2026, 38, e12310 DOI: 10.1002/adma.202512310Google ScholarThere is no corresponding record for this reference.
- 153Wu, B.; Li, X.; Hou, T.; Chen, X.; Wang, J.; Li, S.; Chen, A.; Zhu, C.; Zhang, X.; Ou, H.; Wang, Y.; Wang, D.; Zhang, J. Oxygen-Bridged Cu and V Dual Metal Sites for Enhanced Photo-Oxidative Coupling of Benzylamine. J. Am. Chem. Soc. 2025, 147 (51), 46997– 47007, DOI: 10.1021/jacs.5c11918Google ScholarThere is no corresponding record for this reference.
- 154Li, J.-Y.; Huang, J.-R.; Zhao, Z.-H.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Low-Coordination Indium Single-Atom Sites Anchored on a Metal-Organic Framework Single-Layer Boosts Electroreduction of CO2 Into Formic Acid. Angew. Chem., Int. Ed. 2025, 64 (36), e202511132 DOI: 10.1002/anie.202511132Google ScholarThere is no corresponding record for this reference.
- 155Chen, H.; Huang, Q.; Yang, K.; Chen, S.; Feng, H.; Jia, C.; Li, Q.; Zhao, C.; Duan, J. Tunable Ag-Ox Coordination for Industrial-Level Carbon-Negative CO2 Electrolysis. Nano Energy 2024, 131, 110265, DOI: 10.1016/j.nanoen.2024.110265Google ScholarThere is no corresponding record for this reference.
- 156Zhao, Q.; Zhang, C.; Hu, R.; Du, Z.; Gu, J.; Cui, Y.; Chen, X.; Xu, W.; Cheng, Z.; Li, S.; Li, B.; Liu, Y.; Chen, W.; Liu, C.; Shang, J.; Song, L.; Yang, S. Selective Etching Quaternary MAX Phase toward Single Atom Copper Immobilized MXene (Ti3C2Clx) for Efficient CO2 Electroreduction to Methanol. ACS Nano 2021, 15 (3), 4927– 4936, DOI: 10.1021/acsnano.0c09755Google ScholarThere is no corresponding record for this reference.
- 157Ding, J.; Bin Yang, H.; Ma, X.-L.; Liu, S.; Liu, W.; Mao, Q.; Huang, Y.; Li, J.; Zhang, T.; Liu, B. A Tin-Based Tandem Electrocatalyst for CO2 Reduction to Ethanol with 80% Selectivity. Nat. Energy 2023, 8 (12), 1386– 1394, DOI: 10.1038/s41560-023-01389-3Google ScholarThere is no corresponding record for this reference.
- 158Xu, H.; Rebollar, D.; He, H.; Chong, L.; Liu, Y.; Liu, C.; Sun, C.-J.; Li, T.; Muntean, J. V.; Winans, R. E.; Liu, D.-J.; Xu, T. Highly Selective Electrocatalytic CO2 Reduction to Ethanol by Metallic Clusters Dynamically Formed from Atomically Dispersed Copper. Nat. Energy 2020, 5 (8), 623– 632, DOI: 10.1038/s41560-020-0666-xGoogle ScholarThere is no corresponding record for this reference.
- 159Jiang, Y.; Mao, K.; Li, J.; Duan, D.; Li, J.; Wang, X.; Zhong, Y.; Zhang, C.; Liu, H.; Gong, W.; Long, R.; Xiong, Y. Pushing the Performance Limit of Cu/CeO2 Catalyst in CO2 Electroreduction: A Cluster Model Study for Loading Single Atoms. ACS Nano 2023, 17 (3), 2620– 2628, DOI: 10.1021/acsnano.2c10534Google ScholarThere is no corresponding record for this reference.
- 160Guo, Z.; Zhou, P.; Jiang, L.; Liu, S.; Yang, Y.; Li, Z.; Wu, P.; Zhang, Z.; Li, H. Electron Localization-Triggered Proton Pumping Toward Cu Single Atoms for Electrochemical CO2 Methanation of Unprecedented Selectivity. Adv. Mater. 2024, 36 (14), 2311149, DOI: 10.1002/adma.202311149Google ScholarThere is no corresponding record for this reference.
- 161Xu, H.; Wang, J.; He, H.; Hwang, I.; Liu, Y.; Sun, C.; Zhang, H.; Li, T.; Muntean, J. V.; Xu, T.; Liu, D.-J. Modulating CO2 Electrocatalytic Conversion to the Organics Pathway by the Catalytic Site Dimension. J. Am. Chem. Soc. 2024, 146 (15), 10357– 10366, DOI: 10.1021/jacs.3c12722Google ScholarThere is no corresponding record for this reference.
- 162Chen, R.; Zhao, J.; Li, Y.; Cui, Y.; Lu, Y.-R.; Hung, S.-F.; Wang, S.; Wang, W.; Huo, G.; Zhao, Y.; Liu, W.; Wang, J.; Xiao, H.; Li, X.; Huang, Y.; Liu, B. Operando Mössbauer Spectroscopic Tracking the Metastable State of Atomically Dispersed Tin in Copper Oxide for Selective CO2 Electroreduction. J. Am. Chem. Soc. 2023, 145 (37), 20683– 20691, DOI: 10.1021/jacs.3c06738Google ScholarThere is no corresponding record for this reference.
- 163Xu, T.; Lin, C.; Wang, C.; Brewe, D. L.; Ito, Y.; Lu, J. Synthesis of Supported Platinum Nanoparticles from Li-Pt Solid Solution. J. Am. Chem. Soc. 2010, 132 (7), 2151– 2153, DOI: 10.1021/ja909442cGoogle ScholarThere is no corresponding record for this reference.
- 164Xu, H.; Zhang, Z.; Liu, J.; Do-Thanh, C.-L.; Chen, H.; Xu, S.; Lin, Q.; Jiao, Y.; Wang, J.; Wang, Y.; Chen, Y.; Dai, S. Entropy-Stabilized Single-Atom Pd Catalysts via High-Entropy Fluorite Oxide Supports. Nat. Commun. 2020, 11 (1), 3908, DOI: 10.1038/s41467-020-17738-9Google ScholarThere is no corresponding record for this reference.
- 165Fonseca, J.; Lu, J. Single-Atom Catalysts Designed and Prepared by the Atomic Layer Deposition Technique. ACS Catal. 2021, 11 (12), 7018– 7059, DOI: 10.1021/acscatal.1c01200Google ScholarThere is no corresponding record for this reference.
- 166Knoll, M.; Ruska, E. Das Elektronenmikroskop. Z. Physik 1932, 78 (5), 318– 339, DOI: 10.1007/BF01342199Google ScholarThere is no corresponding record for this reference.
- 167von Ardenne, M. Das Elektronen-Rastermikroskop. Z. Physik 1938, 109 (9), 553– 572, DOI: 10.1007/BF01341584Google ScholarThere is no corresponding record for this reference.
- 168Scherzer, O. Über einige Fehler von Elektronenlinsen. Z. Physik 1936, 101 (9), 593– 603, DOI: 10.1007/BF01349606Google ScholarThere is no corresponding record for this reference.
- 169Crewe, A. V.; Wall, J.; Langmore, J. Visibility of Single Atoms. Science 1970, 168 (3937), 1338– 1340, DOI: 10.1126/science.168.3937.1338Google ScholarThere is no corresponding record for this reference.
- 170Pennycook, S. J.; Jesson, D. E. High-Resolution Z-Contrast Imaging of Crystals. Ultramicroscopy 1991, 37 (1), 14– 38, DOI: 10.1016/0304-3991(91)90004-PGoogle ScholarThere is no corresponding record for this reference.
- 171Tieu, P.; Yan, X.; Xu, M.; Christopher, P.; Pan, X. Directly Probing the Local Coordination, Charge State, and Stability of Single Atom Catalysts by Advanced Electron Microscopy: A Review. Small 2021, 17 (16), 2006482, DOI: 10.1002/smll.202006482Google ScholarThere is no corresponding record for this reference.
- 172Sun, R.; Cao, X.; Ma, J.; Chen, H.-C.; Chen, C.; Peng, Q.; Li, Y. Regulation of Pd Single-Atom Coordination for Enhanced Photocatalytic Oxidation of Toluene to Benzaldehyde. Nat. Synth. 2025, 4 (8), 965– 975, DOI: 10.1038/s44160-025-00782-yGoogle ScholarThere is no corresponding record for this reference.
- 173Haider, M.; Uhlemann, S.; Schwan, E.; Rose, H.; Kabius, B.; Urban, K. Electron Microscopy Image Enhanced. Nature 1998, 392 (6678), 768– 769, DOI: 10.1038/33823Google ScholarThere is no corresponding record for this reference.
- 174Batson, P. E.; Dellby, N.; Krivanek, O. L. Sub-Ångstrom Resolution Using Aberration Corrected Electron Optics. Nature 2002, 418 (6898), 617– 620, DOI: 10.1038/nature00972Google ScholarThere is no corresponding record for this reference.
- 175Fricke, H. The K-Characteristic Absorption Frequencies for the Chemical Elements Magnesium to Chromium. Phys. Rev. 1920, 16 (3), 202– 215, DOI: 10.1103/PhysRev.16.202Google ScholarThere is no corresponding record for this reference.
- 176Kronig, R. de L. Zur Theorie der Feinstruktur in den Röntgenabsorptionsspektren. Z. Physik 1931, 70 (5), 317– 323, DOI: 10.1007/BF01339581Google ScholarThere is no corresponding record for this reference.
- 177Sayers, D. E. New Technique for Investigating Noncrystalline Structures: Fourier Analysis of the Extended X-Ray─Absorption Fine Structure. Phys. Rev. Lett. 1971, 27 (18), 1204– 1207, DOI: 10.1103/PhysRevLett.27.1204Google ScholarThere is no corresponding record for this reference.
- 178Winick, H.; Bienenstock, A. Synchrotron Radiation Research. Annu. Rev. Nucl. Part. Sci. 1978, 28 (28), 33– 113, DOI: 10.1146/annurev.ns.28.120178.000341Google ScholarThere is no corresponding record for this reference.
- 179Rehr, J. J.; Albers, R. C. Theoretical Approaches to X-Ray Absorption Fine Structure. Rev. Mod. Phys. 2000, 72 (3), 621– 654, DOI: 10.1103/RevModPhys.72.621Google ScholarThere is no corresponding record for this reference.
- 180Bordiga, S.; Groppo, E.; Agostini, G.; van Bokhoven, J. A.; Lamberti, C. Reactivity of Surface Species in Heterogeneous Catalysts Probed by In Situ X-Ray Absorption Techniques. Chem. Rev. 2013, 113 (3), 1736– 1850, DOI: 10.1021/cr2000898Google ScholarThere is no corresponding record for this reference.
- 181Mössbauer, R. L. Kernresonanzfluoreszenz von Gammastrahlung in Ir191. Z. Physik 1958, 151 (2), 124– 143, DOI: 10.1007/BF01344210Google ScholarThere is no corresponding record for this reference.
- 182Greenwood, N. N.; Gibb, T. C. Mössbauer Spectroscopy. Springer Netherlands 1971, DOI: 10.1007/978-94-009-5697-1Google ScholarThere is no corresponding record for this reference.
- 183Pound, R. V.; Rebka, G. A. Resonant Absorption of the 14.4-keV γ Ray from 0.10-Μsec Fe57. Phys. Rev. Lett. 1959, 3 (12), 554– 556, DOI: 10.1103/PhysRevLett.3.554Google ScholarThere is no corresponding record for this reference.
- 184Zeng, Y. In Situ/Operando Mössbauer Spectroscopy for Probing Heterogeneous Catalysis. Chem. Catal. 2021, 1 (6), 1215– 1233, DOI: 10.1016/j.checat.2021.08.013Google ScholarThere is no corresponding record for this reference.
- 185Li, J.; Sougrati, M. T.; Zitolo, A.; Ablett, J. M.; Oğuz, I. C.; Mineva, T.; Matanovic, I.; Atanassov, P.; Huang, Y.; Zenyuk, I.; Di Cicco, A.; Kumar, K.; Dubau, L.; Maillard, F.; Dražić, G.; Jaouen, F. Identification of Durable and Non-Durable FeNx Sites in Fe-N-C Materials for Proton Exchange Membrane Fuel Cells. Nat. Catal. 2021, 4 (1), 10– 19, DOI: 10.1038/s41929-020-00545-2Google ScholarThere is no corresponding record for this reference.
- 186Egerton, R. F.; McLeod, R.; Wang, F.; Malac, M. Basic Questions Related to Electron-Induced Sputtering in the TEM. Ultramicroscopy 2010, 110 (8), 991– 997, DOI: 10.1016/j.ultramic.2009.11.003Google ScholarThere is no corresponding record for this reference.
- 187Egerton, R. F. Control of Radiation Damage in the TEM. Ultramicroscopy 2013, 127, 100– 108, DOI: 10.1016/j.ultramic.2012.07.006Google ScholarThere is no corresponding record for this reference.
- 188Finzel, J.; Sanroman Gutierrez, K. M.; Hoffman, A. S.; Resasco, J.; Christopher, P.; Bare, S. R. Limits of Detection for EXAFS Characterization of Heterogeneous Single-Atom Catalysts. ACS Catal. 2023, 13 (9), 6462– 6473, DOI: 10.1021/acscatal.3c01116Google ScholarThere is no corresponding record for this reference.
- 189Egerton, R. f. Mechanisms of Radiation Damage in Beam-Sensitive Specimens, for TEM Accelerating Voltages between 10 and 300 kV. Micro. Res. Technol. 2012, 75 (11), 1550– 1556, DOI: 10.1002/jemt.22099Google ScholarThere is no corresponding record for this reference.
- 190He, Q.; Li, A.; Yao, B.; Zhou, W.; Kiely, C. J. Springer Handbook of Advanced Catalyst Characterization. Springer Cham 2023, 409, DOI: 10.1007/978-3-031-07125-6_20Google ScholarThere is no corresponding record for this reference.
- 191Egerton, R. F. Radiation Damage to Organic and Inorganic Specimens in the TEM. Micron 2019, 119, 72– 87, DOI: 10.1016/j.micron.2019.01.005Google ScholarThere is no corresponding record for this reference.
- 192Zhang, T.; Chen, Z.; Walsh, A. G.; Li, Y.; Zhang, P. Single-Atom Catalysts Supported by Crystalline Porous Materials: Views from the Inside. Adv. Mater. 2020, 32 (44), 2002910, DOI: 10.1002/adma.202002910Google ScholarThere is no corresponding record for this reference.
- 193Wang, A.; Li, J.; Zhang, T. Heterogeneous Single-Atom Catalysis. Nat. Rev. Chem. 2018, 2 (6), 65– 81, DOI: 10.1038/s41570-018-0010-1Google ScholarThere is no corresponding record for this reference.
- 194Ogino, I. X-Ray Absorption Spectroscopy for Single-Atom Catalysts: Critical Importance and Persistent Challenges. Chin. J. Catal. 2017, 38 (9), 1481– 1488, DOI: 10.1016/S1872-2067(17)62880-8Google ScholarThere is no corresponding record for this reference.
- 195Feng, K.; Zhang, H.; Gao, J.; Xu, J.; Dong, Y.; Kang, Z.; Zhong, J. Single Atoms or Not? The Limitation of EXAFS. Appl. Phys. Lett. 2020, 116 (19), 191903, DOI: 10.1063/5.0008748Google ScholarThere is no corresponding record for this reference.
- 196Bazin, D. C.; Sayers, D. A.; Rehr, J. J. Comparison between X-Ray Absorption Spectroscopy, Anomalous Wide Angle X-Ray Scattering, Anomalous Small Angle X-Ray Scattering, and Diffraction Anomalous Fine Structure Techniques Applied to Nanometer-Scale Metallic Clusters. J. Phys. Chem. B 1997, 101 (51), 11040– 11050, DOI: 10.1021/jp9721311Google ScholarThere is no corresponding record for this reference.
- 197Liu, L.; Corma, A. Identification of the Active Sites in Supported Subnanometric Metal Catalysts. Nat. Catal. 2021, 4 (6), 453– 456, DOI: 10.1038/s41929-021-00632-yGoogle ScholarThere is no corresponding record for this reference.
- 198Liu, A.; Liu, L.; Cao, Y.; Wang, J.; Si, R.; Gao, F.; Dong, L. Controlling Dynamic Structural Transformation of Atomically Dispersed CuOx Species and Influence on Their Catalytic Performances. ACS Catal. 2019, 9 (11), 9840– 9851, DOI: 10.1021/acscatal.9b02773Google ScholarThere is no corresponding record for this reference.
- 199Gütlich, P.; Bill, E.; Trautwein, A. X. Mössbauer Spectroscopy and Transition Metal Chemistry. Springer Berlin 2011, DOI: 10.1007/978-3-540-88428-6Google ScholarThere is no corresponding record for this reference.
- 200Chen, C.; Li, J.; Tan, X.; Zhang, Y.; Li, Y.; He, C.; Xu, Z.; Zhang, C.; Chen, C. Harnessing Single-Atom Catalysts for CO2 Electroreduction: A Review of Recent Advances. EES Catal. 2024, 2 (1), 71– 93, DOI: 10.1039/D3EY00150DGoogle ScholarThere is no corresponding record for this reference.
- 201Ren, C.; Cui, Y.; Li, Q.; Ling, C.; Wang, J. Single-Atom Saturation: A Fundamental Principle for Single-Atom-Site Catalyst Design. J. Am. Chem. Soc. 2025, 147 (16), 13610– 13617, DOI: 10.1021/jacs.5c00643Google ScholarThere is no corresponding record for this reference.
- 202Liu, X.; Ding, G.; Guo, X.; Wang, X.; Wang, Z.; Chen, Z.; Xiao, Y.; Shuai, L.; Liao, G. Synergizing Donor-Acceptor Ni-MOF with Lignin Proton Relay for Efficient and Selective CO2-to-CO Photoreduction. Adv. Mater. 2026, 38, e20384 DOI: 10.1002/adma.202520384Google ScholarThere is no corresponding record for this reference.
- 203Vijay, S.; Ju, W.; Brückner, S.; Tsang, S.-C.; Strasser, P.; Chan, K. Unified Mechanistic Understanding of CO2 Reduction to CO on Transition Metal and Single Atom Catalysts. Nat. Catal. 2021, 4 (12), 1024– 1031, DOI: 10.1038/s41929-021-00705-yGoogle ScholarThere is no corresponding record for this reference.
- 204Kortlever, R.; Shen, J.; Schouten, K. J. P.; Calle-Vallejo, F.; Koper, M. T. M. Catalysts and Reaction Pathways for the Electrochemical Reduction of Carbon Dioxide. J. Phys. Chem. Lett. 2015, 6 (20), 4073– 4082, DOI: 10.1021/acs.jpclett.5b01559Google ScholarThere is no corresponding record for this reference.
- 205Göttle, A. J.; Koper, M. T. M. Determinant Role of Electrogenerated Reactive Nucleophilic Species on Selectivity during Reduction of CO2 Catalyzed by Metalloporphyrins. J. Am. Chem. Soc. 2018, 140 (14), 4826– 4834, DOI: 10.1021/jacs.7b11267Google ScholarThere is no corresponding record for this reference.
- 206Fan, L.; Li, F.; Liu, T.; Huang, J. E.; Miao, R. K.; Yan, Y.; Feng, S.; Tai, C.-W.; Hung, S.-F.; Tsai, H.-J.; Chen, M.-C.; Bai, Y.; Kim, D.; Park, S.; Papangelakis, P.; Wu, C.; Shayesteh Zeraati, A.; Dorakhan, R.; Sun, L.; Sinton, D.; Sargent, E. Atomic-Level Cu Active Sites Enable Energy-Efficient CO2 Electroreduction to Multicarbon Products in Strong Acid. Nat. Synth. 2025, 4 (2), 262– 270, DOI: 10.1038/s44160-024-00689-0Google ScholarThere is no corresponding record for this reference.
- 207Zhao, Y.; Shi, Z.; Li, F.; Jia, C.; Sun, Q.; Su, Z.; Zhao, C. Deciphering Mesopore-Augmented CO2 Electroreduction over Atomically Dispersed Fe-N-Doped Carbon Catalysts. ACS Catal. 2024, 14 (6), 3926– 3932, DOI: 10.1021/acscatal.3c05144Google ScholarThere is no corresponding record for this reference.
- 208Zhang, T.; Han, X.; Liu, H.; Biset-Peiró, M.; Li, J.; Zhang, X.; Tang, P.; Yang, B.; Zheng, L.; Morante, J. R.; Arbiol, J. Site-Specific Axial Oxygen Coordinated FeN4 Active Sites for Highly Selective Electroreduction of Carbon Dioxide. Adv. Funct. Mater. 2022, 32 (18), 2111446, DOI: 10.1002/adfm.202111446Google ScholarThere is no corresponding record for this reference.
- 209Chen, Z.; Zhang, X.; Liu, W.; Jiao, M.; Mou, K.; Zhang, X.; Liu, L. Amination Strategy to Boost the CO2 Electroreduction Current Density of M-N/C Single-Atom Catalysts to the Industrial Application Level. Energy Environ. Sci. 2021, 14 (4), 2349– 2356, DOI: 10.1039/D0EE04052EGoogle ScholarThere is no corresponding record for this reference.
- 210Tierney, H. L.; Baber, A. E.; Kitchin, J. R.; Sykes, E. C. H. Hydrogen Dissociation and Spillover on Individual Isolated Palladium Atoms. Phys. Rev. Lett. 2009, 103 (24), 246102, DOI: 10.1103/PhysRevLett.103.246102Google ScholarThere is no corresponding record for this reference.
- 211Sun, L.; Wang, Y.; Guan, N.; Li, L. Methane Activation and Utilization: Current Status and Future Challenges. Energy Technol. 2020, 8 (8), 1900826, DOI: 10.1002/ente.201900826Google ScholarThere is no corresponding record for this reference.
- 212Shen, W.; Gao, X.; Liu, Q.; Li, P.; Huang, R.; Tan, Y.; Wang, Z.; Zhang, Y.; Zhao, F.; Wang, X.; Ji, S.; Zheng, X.; Zhang, Y.; Wu, Y. Self-Healing Cu Single-Atom Catalyst for High-Performance Electrocatalytic CO2 Methanation. Nat. Commun. 2025, 16 (1), 7943, DOI: 10.1038/s41467-025-63274-9Google ScholarThere is no corresponding record for this reference.
- 213Han, L.; Song, S.; Liu, M.; Yao, S.; Liang, Z.; Cheng, H.; Ren, Z.; Liu, W.; Lin, R.; Qi, G.; Liu, X.; Wu, Q.; Luo, J.; Xin, H. L. Stable and Efficient Single-Atom Zn Catalyst for CO2 Reduction to CH4. J. Am. Chem. Soc. 2020, 142 (29), 12563– 12567, DOI: 10.1021/jacs.9b12111Google ScholarThere is no corresponding record for this reference.
- 214Chen, S.; Zhang, Z.; Jiang, W.; Zhang, S.; Zhu, J.; Wang, L.; Ou, H.; Zaman, S.; Tan, L.; Zhu, P.; Zhang, E.; Jiang, P.; Su, Y.; Wang, D.; Li, Y. Engineering Water Molecules Activation Center on Multisite Electrocatalysts for Enhanced CO2 Methanation. J. Am. Chem. Soc. 2022, 144 (28), 12807– 12815, DOI: 10.1021/jacs.2c03875Google ScholarThere is no corresponding record for this reference.
- 215Hung, S.-F.; Xu, A.; Wang, X.; Li, F.; Hsu, S.-H.; Li, Y.; Wicks, J.; Cervantes, E. G.; Rasouli, A. S.; Li, Y. C.; Luo, M.; Nam, D.-H.; Wang, N.; Peng, T.; Yan, Y.; Lee, G.; Sargent, E. H. A Metal-Supported Single-Atom Catalytic Site Enables Carbon Dioxide Hydrogenation. Nat. Commun. 2022, 13 (1), 819, DOI: 10.1038/s41467-022-28456-9Google ScholarThere is no corresponding record for this reference.
- 216Osella, S.; Goddard, W. A., III CO2 Reduction to Methane and Ethylene on a Single-Atom Catalyst: A Grand Canonical Quantum Mechanics Study. J. Am. Chem. Soc. 2023, 145 (39), 21319– 21329, DOI: 10.1021/jacs.3c05650Google ScholarThere is no corresponding record for this reference.
- 217He, H.; Lee, J.; Zong, Z.; Lim, K. M.; Ryu, J.; Oh, J.; Kim, J.; Sessler, J. L.; Ke, X.-S. Precise Internal Postsynthetic Oxygen-Doping of Metallonanographenes. Prec. Chem. 2025, 3 (8), 456– 462, DOI: 10.1021/prechem.5c00035Google ScholarThere is no corresponding record for this reference.
- 218Xie, C.; Chen, W.; Wang, Y.; Yang, Y.; Wang, S. Dynamic Evolution Processes in Electrocatalysis: Structure Evolution, Characterization and Regulation. Chem. Soc. Rev. 2024, 53 (22), 10852– 10877, DOI: 10.1039/D3CS00756AGoogle ScholarThere is no corresponding record for this reference.
- 219He, H.; Jagvaral, Y. Electrochemical Reduction of CO2 on Graphene Supported Transition Metals-towards Single Atom Catalysts. Phys. Chem. Chem. Phys. 2017, 19 (18), 11436– 11446, DOI: 10.1039/C7CP00915AGoogle ScholarThere is no corresponding record for this reference.
- 220Zhang, D.; Ren, P.; Liu, W.; Li, Y.; Salli, S.; Han, F.; Qiao, W.; Liu, Y.; Fan, Y.; Cui, Y.; Shen, Y.; Richards, E.; Wen, X.; Rummeli, M. H.; Li, Y.; Besenbacher, F.; Niemantsverdriet, H.; Lim, T.; Su, R. Photocatalytic Abstraction of Hydrogen Atoms from Water Using Hydroxylated Graphitic Carbon Nitride for Hydrogenative Coupling Reactions. Angew. Chem., Int. Ed. 2022, 61 (24), e202204256 DOI: 10.1002/anie.202204256Google ScholarThere is no corresponding record for this reference.
- 221Yang, T.; Yang, F.; Wang, T.; Zhang, W.; Deng, F.; Zhong, C.; Peng, Q.; Liu, C.; Chen, X.; Zhang, Z.; Li, Y.; Feng, Y.; Li, X.; Wang, K.; Zhang, C.; Zhou, Y.; Lu, X.; Zhu, W.; Wei, Y.; Xie, L.; Huang, W. Gridized Nanopolymer Catalysis with Atomically Dispersed Iron Achieves the Nearly 100% Selective Electrosynthesis of Methanol From CO2. Adv. Mater. 2026, 38, e11233 DOI: 10.1002/adma.202511233Google ScholarThere is no corresponding record for this reference.
- 222Ren, X.; Zhao, J.; Li, X.; Shao, J.; Pan, B.; Salamé, A.; Boutin, E.; Groizard, T.; Wang, S.; Ding, J.; Zhang, X.; Huang, W.-Y.; Zeng, W.-J.; Liu, C.; Li, Y.; Hung, S.-F.; Huang, Y.; Robert, M.; Liu, B. In-Situ Spectroscopic Probe of the Intrinsic Structure Feature of Single-Atom Center in Electrochemical CO/CO2 Reduction to Methanol. Nat. Commun. 2023, 14 (1), 3401, DOI: 10.1038/s41467-023-39153-6Google ScholarThere is no corresponding record for this reference.
- 223Chatterjee, S.; Dutta, I.; Lum, Y.; Lai, Z.; Huang, K.-W. Enabling Storage and Utilization of Low-Carbon Electricity: Power to Formic Acid. Energy Environ. Sci. 2021, 14 (3), 1194– 1246, DOI: 10.1039/D0EE03011BGoogle ScholarThere is no corresponding record for this reference.
- 224Shang, H.; Wang, T.; Pei, J.; Jiang, Z.; Zhou, D.; Wang, Y.; Li, H.; Dong, J.; Zhuang, Z.; Chen, W.; Wang, D.; Zhang, J.; Li, Y. Design of a Single-Atom Indiumδ+-N4 Interface for Efficient Electroreduction of CO2 to Formate. Angew. Chem., Int. Ed. 2020, 59 (50), 22465– 22469, DOI: 10.1002/anie.202010903Google ScholarThere is no corresponding record for this reference.
- 225Fang, C.; Huang, L.; Gao, W.; Jiang, X.; Liu, H.; Hu, R.; Li, X.; Yu, J.; Zhou, W. Oxygen-Pinned Ag1In Single-Atom Alloy for Efficient Electroreduction CO2 to Formate. Adv. Energy Mater. 2024, 14 (27), 2400813, DOI: 10.1002/aenm.202400813Google ScholarThere is no corresponding record for this reference.
- 226Shao, L.-Y.; Wang, Y.; Meng, J.; Yue, J.-N.; Zhang, A.-M.; Yuan, W.; Li, S.-N.; Zhai, Q.-G. Exclusive Bridged *OCHO Pathway Activated by Phenol-Quinone Conversion on the Surface of Cu(OH)2 for Highly Selective Formate Production in CO2 Electroreduction. Adv. Funct. Mater. 2026, 36, e14222 DOI: 10.1002/adfm.202514222Google ScholarThere is no corresponding record for this reference.
- 227Jiang, Z.; Liu, X.; Liu, X.-Z.; Huang, S.; Liu, Y.; Yao, Z.-C.; Zhang, Y.; Zhang, Q.-H.; Gu, L.; Zheng, L.-R.; Li, L.; Zhang, J.; Fan, Y.; Tang, T.; Zhuang, Z.; Hu, J.-S. Interfacial Assembly of Binary Atomic Metal-Nx Sites for High-Performance Energy Devices. Nat. Commun. 2023, 14 (1), 1822, DOI: 10.1038/s41467-023-37529-2Google ScholarThere is no corresponding record for this reference.
- 228Cui, Y.; Ren, C.; Li, Q.; Ling, C.; Wang, J. Hybridization State Transition under Working Conditions: Activity Origin of Single-Atom Catalysts. J. Am. Chem. Soc. 2024, 146 (22), 15640– 15647, DOI: 10.1021/jacs.4c05630Google ScholarThere is no corresponding record for this reference.
- 229Wang, F.; Su, Q.; Zhang, X.; Chen, A. Spin-State Regulation of Single-Atom Catalysts: From Quantum Mechanisms to Multidimensional Dynamic Regulation Strategies. Adv. Funct. Mater. 2026, 36, e18600 DOI: 10.1002/adfm.202518600Google ScholarThere is no corresponding record for this reference.
- 230Zeng, Y.; Zhao, J.; Wang, S.; Wang, W.; Lu, Y.-R.; Xi, S.; Xu, W.; Yoda, Y.; Masuda, R.; Li, X.; Huang, Y.; Liu, B.; Zhang, T. Unraveling the Dynamic Low-Spin State Evolution of Single-Fe-Atom Sites for Efficient CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (46), 42539– 42548, DOI: 10.1021/jacs.5c13466Google ScholarThere is no corresponding record for this reference.
- 231Vaz, C. a. F.; van der Laan, G.; Cavill, S. A.; Dürr, H. A.; Fraile Rodríguez, A.; Kronast, F.; Kuch, W.; Sainctavit, P.; Schütz, G.; Wende, H.; Weschke, E.; Wilhelm, F. X-Ray Magnetic Circular Dichroism. Nat. Rev. Methods Primers 2025, 5 (1), 27, DOI: 10.1038/s43586-025-00397-9Google ScholarThere is no corresponding record for this reference.
- 232Bai, R.; Yao, Y.; Lin, Q.; Wu, L.; Li, Z.; Wang, H.; Ma, M.; Mu, D.; Hu, L.; Yang, H.; Li, W.; Zhu, S.; Wu, X.; Rui, X.; Yu, Y. Preferable Single-Atom Catalysts Enabled by Natural Language Processing for High Energy Density Na-S Batteries. Nat. Commun. 2025, 16 (1), 5827, DOI: 10.1038/s41467-025-60931-xGoogle ScholarThere is no corresponding record for this reference.
- 233Szymanski, N. J.; Rendy, B.; Fei, Y.; Kumar, R. E.; He, T.; Milsted, D.; McDermott, M. J.; Gallant, M.; Cubuk, E. D.; Merchant, A.; Kim, H.; Jain, A.; Bartel, C. J.; Persson, K.; Zeng, Y.; Ceder, G. An Autonomous Laboratory for the Accelerated Synthesis of Novel Materials. Nature 2023, 624 (7990), 86– 91, DOI: 10.1038/s41586-023-06734-wGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. (a) Synthesis scheme of the Ni–N–C catalysts by using a N-doped carbon host to absorb Ni ions followed by thermal activation at different temperatures to tune the Ni–N bond structures and establish structure–property correlations. Reproduced from ref (131). Copyright 2022, Royal Society of Chemistry. (b) Schematic illustration for the formation of the Ni-NC@Ni catalyst. In the green circle: the possible atomic structure of the Ni–N species. Reproduced from ref (132). Copyright 2020, Elsevier. (c) Schematic illustration of the preparation strategy for PSB-CuN3 and PS-CuN4. Reproduced from ref (133). Copyright 2023, Springer Nature. (d) Schematic illustration for the preparation of Ni–PxNy. Reproduced from ref (91). Copyright 2023, Springer Nature. (e) The formation process of atomically dispersed SnN3O1 active sites. Reproduced from ref (107). Copyright 2021, Wiley-VCH. (f) Schematic illustration of the proposed formation mechanisms. Reproduced from ref (118). Copyright 2021, Springer Nature.
Figure 2

Figure 2. (a) Schematic synthesis process of the Sn1Cu-SAA. Reproduced from ref (77). Copyright 2025, Wiley-VCH. (b) Schematic representation of the preparation of the Ru1@Bi and Run@Bi catalysts. Reproduced from ref (143). Copyright 2024, Wiley-VCH. (c) Schematic diagram for the preparation of Mo1Cu catalysts. Reproduced from ref (144). Copyright 2025, Wiley-VCH. (d) Schematic diagram of the NiZn0.03/C bimetallic catalyst synthesis process. Reproduced from ref (145). Copyright 2023, Wiley-VCH.
Figure 3
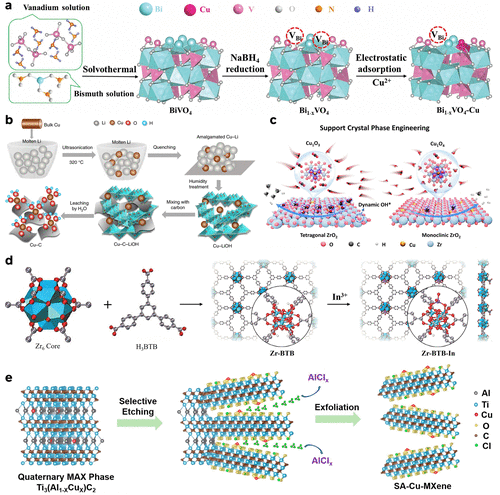
Figure 3. (a) Schematic illustration of preparing Bi1–xVO4–Cu. Reproduced from ref (160). Copyright 2024, Wiley-VCH. (b) Step-by-step preparation of the carbon-supported Cu SA catalyst using an amalgamated Cu–Li method. Reproduced from ref (158). Copyright 2020, Springer Nature. (c) Schematic illustration of the support crystal phase engineering and reaction mechanisms on the surface of Cu1O3-tZrO2 and Cu1O4-mZrO2. Reproduced from ref (83). Copyright 2025, American Chemical Society. (d) Illustration of the synthesis of Zr-BTB-In. Reproduced from ref (154). Copyright 2025, Wiley-VCH. (e) Schematic illustration of the fabrication of SA-Cu-MXene via selective etching of quaternary MAX-Ti3(Al1–xCux)C2. Gray, blue, red, yellow, brown, and green balls represent Al, Ti, Cu, O, C, and Cl atoms, respectively. Reproduced from ref (156). Copyright 2021, American Chemical Society.
Figure 4

Figure 4. (a) General schematic of an electron microscope in STEM modes with a condenser lens, objective lens, aberration correctors, projector system, retractable detectors, EDS (energy dispersive X-ray spectroscopy), and EELS (electron energy loss spectroscopy) acquisition. p.s. BF (bright-field), ABF (annular bright-field). Reproduced from ref (171). Copyright 2021, Wiley-VCH. (b) Pt single atoms (white circles) are seen to be uniformly dispersed on the iron oxide (FeOx) support (left) and occupy exactly the positions of the Fe atoms (right). Reproduced from ref (44). Copyright 2011, Springer Nature. (c) Schematic diagram of XAFS measurements with fluorescence yield experiment setup. Reproduced from ref (74). Copyright 2024, Springer Nature. (d) Schematic diagram of Pd–O4/BWO and Pd–O3/BWO. (e) Fourier transforms of the EXAFS spectra of the Pd K-edge spectra. Reproduced from ref (172). Copyright 2025, Springer Nature. (f) Schematic diagram of operando Mössbauer spectroscopy for tracking the metastable states of atomically dispersed tin (Sn) in copper oxide (CuO) for selective CO2 electroreduction. Reproduced from ref (162). Copyright 2023, American Chemical Society.
Figure 5
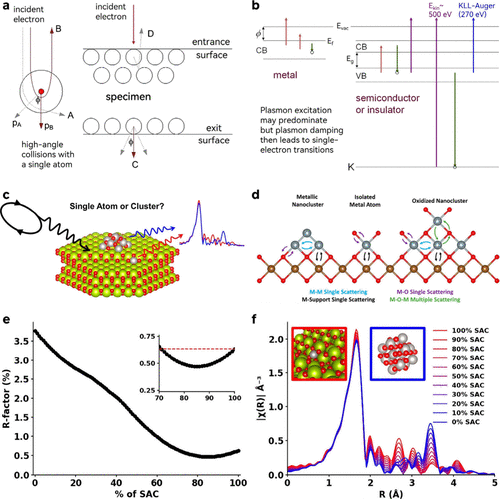
Figure 5. (a) Elastic scattering of electrons from an atomic nucleus, shown schematically (particle model) for a large-angle collision (A) and a 180° collision (B). Sputtering of atoms from the beam-exit surface (C) and the beam-entry surface (D). Reproduced from ref (186). Copyright 2010, Elsevier. (b) Energy-band diagram of a metal (left) and a semiconductor or insulator (right), electron energy being plotted vertically upward. CB and VB represent the conduction and valence bands, f is the work function, Evac, Ef and Eg are the vacuum energy, Fermi energy, and CB-VB energy gap. Upward arrows represent single-electron excitations, while downward arrows are de-excitation processes (filling of a VB or K-shell hole). K-shell excitation in an organic material is shown on the right, with Ekin being the typical kinetic energy of an excited K-shell electron. This process also results in the emission of Auger electrons from the VB, with an energy of about 270 eV. Reproduced from ref (187). Copyright 2013, Elsevier. (c) Schematic diagram of heterogeneous Pt/CeO2 SASCs using EXAFS. (d) Scattering paths for SASCs and small clusters. Schematic depiction of scattering paths present in oxide-supported metal catalysts with a heterogeneity of the metal-site structure including isolated metal sites (middle) as well as metal (left) or oxidized (right) nanoclusters. Bonds are for representation only. Color code: metal (gray), oxygen (red), and support metal cation (brown). (e) R-factor of the 5-path SAC fit to data sets with varying contributions from the Pt/CeO2 SASCs site and metallic Pt13 cluster. The inset shows a zoomed-in region with 70–100% SAC sites (30–0% Pt13 cluster sites). Data are taken over Δk = 3–18 Å–1. (f) Modeled FT EXAFS data for mixtures of the Pt/CeO2 SASCs site and metallic Pt13 cluster. Reproduced from ref (188). Copyright 2023, American Chemical Society.
Figure 6

Figure 6. Overview of reaction pathways for ECR toward different products. Black spheres, carbon; red spheres, oxygen; white spheres, hydrogen; blue spheres, (metal) catalyst. The arrows indicate whether proton, electron or PCETs take place. Reproduced from ref (39). Copyright 2019, Springer Nature.
Figure 7
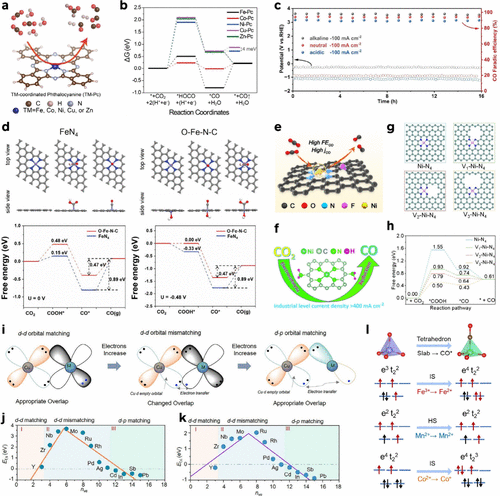
Figure 7. (a) Schematic illustration of CO2RR over a chemical unit of the TM-Pcs. (b) Free energy diagram of TM-PCs for CO2RR at V = 0 V. Reproduced from ref (116). Copyright 2022, American Chemical Society. (c) Stability test of Ni–N3S–C in flow cell with alkaline, acidic, and neutral electrolyte. The electrolytes are 1 M KOH (alkaline, pH = 14), 0.05 M H2SO4 + 1 M KCl (acidic, pH = 1), and 1 M KHCO3 (neutral, pH = 8.2). Reproduced from ref (102). Copyright 2025, Wiley-VCH. (d) Top-left, top-right: Top view and side view optimized adsorption configuration on simulated FeN4 and O–Fe–N-C (Fe, O, N, and C atoms are represented in purple, red, blue, and gray, respectively). Down-left, down-right: Free energy profiles for the CO2RR to CO at 0 V (vs RHE) and at – 0.48 V (vs RHE) on simulated FeN4 and O–Fe–N-C. Reproduced from ref (208). Copyright 2022, Wiley-VCH. (e) Scheme of ECR-to-CO activity for Ni-SAC@NFC. Reproduced from ref (130). Copyright 2024, American Chemical Society. (f) Scheme of ECR-to-CO activity for Ni–N4/C-NH2. Reproduced from ref (209). Copyright 2021, Royal Society Chemistry. (g) Optimized structures of Ni–N4, V1–Ni-N4, V2–Ni-N4, and V3–Ni-N4. (h) Free energy diagram for the ECR-to-CO conversion. Reproduced from ref (81). Copyright 2025, American Chemical Society. (i) Schematic diagram of the orbital matching mechanism, which originated from the characteristic d-d and d-p orbital interactions between the substrate and the dispersed metal atoms. The ″volcano-type″ scaling relationship between the number of valence electrons (nve) of dispersed elements and (j) single atom (k) cluster formation energies. Reproduced from ref (78). Copyright 2025, Wiley-VCH. (l) Different spin states of Co, Fe, and Mn atoms in TM-TCSACs (TM = Co, Fe, and Mn) and CO*. Reproduced from ref (71). Copyright 2025, American Chemical Society.
Figure 8
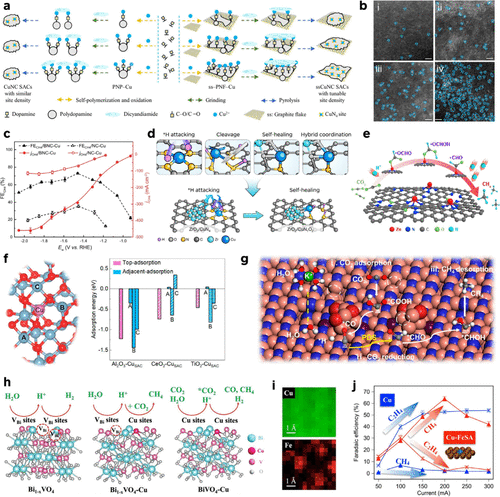
Figure 8. (a) Schematic of the synthesis of CuNC and ssCuNC via support engineering. DCD: Dicyandiamide, PNP-Cu: Cu-chelated polydopamine nanoparticles, ss-PNF-Cu: Cu-bound polydopamine nanoparticles and films coated on ss. (b) AC-HAADF-STEM images of (i) ssCuNC40, (ii) ssCuNC80, (iii) ssCuNC100, and (iv) ssCuNC160. Reproduced from ref (113). Copyright 2025, Springer Nature. (c) FE and partial current densities for CH4 of BNC-Cu and NC-Cu as a function of cathodic potentials. Red curve for partial current density and black curve for methane FE. The error bars of FECH4 and jCH4 are calculated based on three independent measurements. Reproduced from ref (105). Copyright 2023, Springer Nature. (d) Conceptual schematic of the self-healing coordination reconstruction process. Reproduced from ref (212). Copyright 2025, Springer Nature. (e) Scheme of ECR-to-CH4 activity for SA-Zn/MNC. Reproduced from ref (213). Copyright 2020, American Chemical Society. (f) Left: Schematic diagram of CO adjacent-adsorption on the Al2O3–CuSAC surface. Right: CO adsorption energy of Al2O3–CuSAC, CeO2–CuSAC, and TiO2–CuSAC. Reproduced from ref (72). Copyright 2025, Springer Nature. (g) Scheme of ECR-toCH4 activity for SA-Zn/MNC. Reproduced from ref (214). Copyright 2022, American Chemical Society. (h) Diagrams of the electroreduction processes over Bi1–xVO4, Bi1–xVO4–Cu, and BiVO4–Cu. Reproduced from ref (160). Copyright 2023, Wiley-VCH. (i) Atomic elemental mapping using the EELS. (j) Comparison of reaction products for pristine Cu and Cu-FeSA. Error bars represent 1 standard deviation on the basis of three independent samples. Reproduced from ref (215). Copyright 2022, Springer Nature.
Figure 9
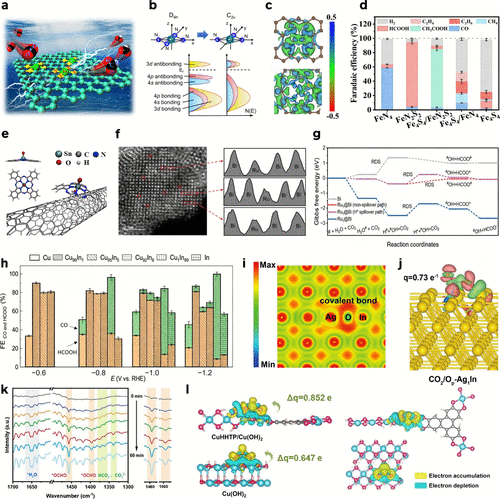
Figure 9. (a) Scheme of ECR-to-HCOOH activity for In Sas/NC. Reproduced from ref (224). Copyright 2020, Wiley-VCH. (b) Schematic diagrams of band shifts and hybridization for the planar CuN4 moiety with local D4h symmetry and the defective CuN3 moiety with lower local C2v symmetry. a.u. arbitrary units. Reproduced from ref (133). Copyright 2023, Springer Nature. (c) EDD plots of FeN4 and FeN2S2. (d) Distribution of ECR products over different Fe-based catalysts at −0.5 V. Reproduced from ref (89). Copyright 2025, American Chemical Society. (e) Schematic illustration for the preparation of SnPc/CNT–OH. Reproduced from ref (128). Copyright 2023, American Chemical Society. (f) Atomic-resolution AC-HAADF-STEM image and intensity profiles of Ru1@Bi. (g) Gibbs free energy landscapes for ECR-to-HCOOH at 0 V versus RHE. The RDS is labeled with a black text. Reproduced from ref (143). Copyright 2024, Wiley-VCH. (h) Comparison of FECO (orange color) and FEHCOO– (green color) on Cu, Cu99In1, Cu95In5, Cu92In8, Cu1In99 and In catalysts. Reproduced from ref (79). Copyright 2025, Wiley-VCH. (i) Electron localization function maps of Op-Ag1In. (j) Charge density difference map of CO2 on Op-Ag1In. Reproduced from ref (225). Copyright 2024, Wiley-VCH. (k) In-situ ATR-FTIR spectra on CuHHTP/Cu(OH)2 at −1.7 V for 60 min in a CO2-saturated 0.1 M KHCO3–KCl electrode. (l) The charge density difference analysis on CuHHTP/Cu(OH)2 and Cu(OH)2. Reproduced from ref (226). Copyright 2025, Wiley-VCH.
Figure 10
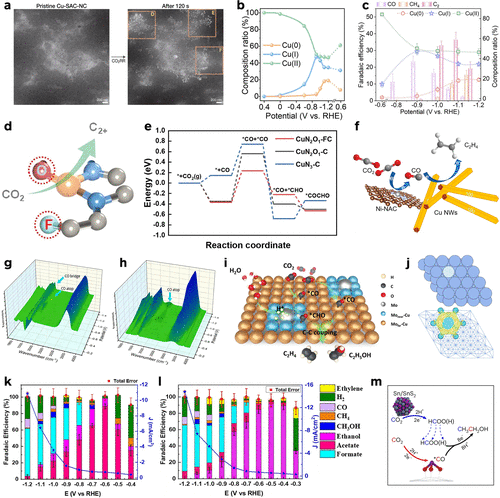
Figure 10. (a) Low magnification EC-STEM images of pristine-SAC and Cu nanograins after the CO2RR for 120 s in liquids. Reproduced from ref (80). Copyright 2025, American Chemical Society. (b) Linear combination fitting of Cu oxidation states for CuPc-F. (c) Cu composition ratio and FEs of various products at multiple potentials for CuPc-F. Reproduced from ref (82). Copyright 2025, American Chemical Society. (d) F, O-codrived local coordination environment optimized for CuFONC favors the CO2RR toward C2+. (e) The reaction pathways of CO2 to *COCHO on CuN2O1–FC, CuN2O1–C, and CuN3–C. Reproduced from ref (121). Copyright 2024, Wiley-VCH. (f) Schematic illustration of the modular design of the Cu/Ni-NAC hybrid catalyst for tandem catalysis. In-situ SEIRAS spectra of (g) the Cu/Ni-NAC and (h) the Cu NW catalysts under ECR conditions. Reproduced from ref (95). Copyright 2022, American Chemical Society. (i) A proposed reaction mechanism for the CO2RR to C2H4 and C2H5OH. Reproduced from ref (144). Copyright 2025, Wiley-VCH. (j) Simulated geometry and charge density difference of M1/Cu(111). Reproduced from ref (68). Copyright 2025, Springer Nature. FEs and the product distributions under different polarization potentials over (k) Sn/C-0.12 and (l) Sn/C-1.2 (stacked bar chart) and the geometric partial current densities, J, toward (k) acetate and (l) ethanol (blue line-stars). All FEs are calculated from chronoamperometry measurements. Reproduced from ref (161). Copyright 2024, American Chemical Society. (m) A schematic illustration showing the cascade reaction during CO2 reduction to ethanol over SnS2/Sn1–O3G (gray: S, red: O, yellow: H and purple: Sn). Reproduced from ref (157). Copyright 2023, Springer Nature.
References
This article references 233 other publications.
- 1Wang, Q.; Luo, T.; Cao, X.; Gong, Y.; Liu, Y.; Xiao, Y.; Li, H.; Gröbmeyer, F.; Lu, Y.-R.; Chan, T.-S.; Ma, C.; Liu, K.; Fu, J.; Zhang, S.; Liu, C.; Lin, Z.; Chai, L.; Cortes, E.; Liu, M. Lanthanide Single-Atom Catalysts for Efficient CO2-to-CO Electroreduction. Nat. Commun. 2025, 16 (1), 2985, DOI: 10.1038/s41467-025-57464-8There is no corresponding record for this reference.
- 2Wei, C.; Wang, S.; Zhao, X. Spatial-Temporal Variation and Coupling Relationship between Primary Energy Consumption and Economic Growth: A Global Assessment. Energy 2025, 323, 135873, DOI: 10.1016/j.energy.2025.135873There is no corresponding record for this reference.
- 3Raihan, A. Carbon Capture, Utilization, and Storage (CCUS) in the Context of Earth Energy Systems: A Multidisciplinary Review. Geosci. Front. 2025, 16 (6), 102177, DOI: 10.1016/j.gsf.2025.102177There is no corresponding record for this reference.
- 4Wu, Q.; Ji, S.; Chen, J.; Tan, X.-Q.; Ong, W.-J.; Du, R.; Wang, P.; Wang, H.; Qiu, Y.; Yan, K.; Zhao, Y.; Zhao, W.-W.; Peng, K.-S.; Chen, Y.-Y.; Hung, S.-F.; Zhou, L.; Wang, X.; Qiu, G.; Chen, G. Realizing the Practical Application of CO2 Electroreduction for Urban Wastewater Denitrification. Nat. Water. 2025, 3 (11), 1291– 1302, DOI: 10.1038/s44221-025-00516-6There is no corresponding record for this reference.
- 5Bai, W.; Zeng, H.; Chen, F.; Wu, S.; Wang, S.; Du, Y.; Liu, S.; Wang, D.; Dai, Z. Emerging Atomistic Modeling Catalysts for C-N Electrocatalysis. Adv. Mater. 2026, 38, e10907 DOI: 10.1002/adma.202510907There is no corresponding record for this reference.
- 6Zhao, D.; Yu, K.; Song, P.; Feng, W.; Hu, B.; Cheong, W.-C.; Zhuang, Z.; Liu, S.; Sun, K.; Zhang, J.; Chen, C. Atomic-Level Engineering Fe1N2O2 Interfacial Structure Derived from Oxygen-Abundant Metal-Organic Frameworks to Promote Electrochemical CO2 Reduction. Energy Environ. Sci. 2022, 15 (9), 3795– 3804, DOI: 10.1039/D2EE00878EThere is no corresponding record for this reference.
- 7Song, P.; Hu, B.; Zhao, D.; Fu, J.; Su, X.; Feng, W.; Yu, K.; Liu, S.; Zhang, J.; Chen, C. Modulating the Asymmetric Atomic Interface of Copper Single Atoms for Efficient CO2 Electroreduction. ACS Nano 2023, 17 (5), 4619– 4628, DOI: 10.1021/acsnano.2c10701There is no corresponding record for this reference.
- 8Vos, R. E.; Sun, P.; Schauermann, D.; Javed, H.; Hanselman, S. R.; Fu, G.; Koper, M. T. M. CO2 Electroreduction on Cu Operates via an Alternative Chain Growth Mechanism to Form C-C Bonds at Elevated Temperature and Pressure. Nat. Catal. 2025, 8 (12), 1338– 1347, DOI: 10.1038/s41929-025-01451-1There is no corresponding record for this reference.
- 9Bi, X.; Yan, Y.; Wang, H.; Zhao, Y.; Zhang, J.; Wu, M. Electroreduction of CO2 to C2H4 Regulated by Spacing Effect: Mechanistic Insights from DFT Studies. Energy Mater. Adv. 2023, 4, 0037, DOI: 10.34133/energymatadv.0037There is no corresponding record for this reference.
- 10Wang, S.; Li, F.; Zhao, J.; Zeng, Y.; Li, Y.; Lin, Z.-Y.; Lee, T.-J.; Liu, S.; Ren, X.; Wang, W.; Chen, Y.; Hung, S.-F.; Lu, Y.-R.; Cui, Y.; Yang, X.; Li, X.; Huang, Y.; Liu, B. Manipulating C-C Coupling Pathway in Electrochemical CO2 Reduction for Selective Ethylene and Ethanol Production over Single-Atom Alloy Catalyst. Nat. Commun. 2024, 15 (1), 10247, DOI: 10.1038/s41467-024-54636-wThere is no corresponding record for this reference.
- 11Zhang, W.; Zhou, Z.; Tan, X.; Tan, T. Mastering Crystalline Phase in Bismuth-Based Electrocatalysts for Efficient CO2-to-Formate Conversion. Energy Mater. Adv. 2025, 0, DOI: 10.34133/energymatadv.0242There is no corresponding record for this reference.
- 12Fu, J.; Zhang, H.; Du, H.; Liu, X.; Lyu, Z.-H.; Jiang, Z.; Chen, F.; Ding, L.; Tang, T.; Zhu, W.; Su, D.; Ling, C.; Wang, J.; Hu, J.-S. Unveiling the Interfacial Species Synergy in Promoting CO2 Tandem Electrocatalysis in Near-Neutral Electrolyte. J. Am. Chem. Soc. 2024, 146 (33), 23625– 23632, DOI: 10.1021/jacs.4c08844There is no corresponding record for this reference.
- 13Kim, K.-M.; Mun, J.; Yun, G.-N.; You, Y.-W.; Park, J. H.; Lee, J. H.; So, J.; Shin, H.; Kwon, J.; Kim, S.; Kang, S.; Kwon, Y. K.; Kwon, T.-H.; Bae, Y.-S.; Lee, G.; Kim, S.-J.; Kim, Y. J.; Kim, H.-T. Rational Synthesis of Dual-Atom Catalysts for Optimized Thermochemical CO2 Reduction. Nat. Commun. 2025, DOI: 10.1038/s41467-025-66608-9There is no corresponding record for this reference.
- 14Li, S.; Ma, J.; Wang, N.; Liu, X.; Cui, W.; Lyu, Y.; Perathoner, S.; Centi, G.; Liu, Y. Engineering Dual-Active Sites on CeOx-Modified Defective MgO for Efficient CO2 Thermocatalytic Reduction. J. Energy Chem. 2025, 110, 486– 496, DOI: 10.1016/j.jechem.2025.06.073There is no corresponding record for this reference.
- 15Hu, X.; Liu, X.; Hu, X.; Zhao, C.; Guan, Q.; Li, W. Hybrid Catalyst Coupling Zn Single Atoms and CuNx Clusters for Synergetic Catalytic Reduction of CO2. Adv. Funct. Mater. 2023, 33 (16), 2214215, DOI: 10.1002/adfm.202214215There is no corresponding record for this reference.
- 16Hao, J.; Li, Y.; Li, Z.; Yin, Z.; Xu, L.; Wang, Y.; Gao, S.; Liu, Y.; Qiu, J.; Yang, Z.; Song, Z. Dual-Mechanism Regulation in Photocatalytic CO2 Reduction through Er Single-Atom Engineering: Enhancing Charge Dynamics and Molecular Activation. ACS Catal. 2025, 15 (17), 15172– 15182, DOI: 10.1021/acscatal.5c03633There is no corresponding record for this reference.
- 17Xiao, Y.; Ding, G.; Tao, J.; Wang, Z.; Chen, Z.; Chen, L.; Shuai, L.; Liao, G. Selective Conversion of CO2 to C2H6 in Pure Water Photocatalyzed by Fluorobenzene-Linked Perylene Diimide. Nat. Commun. 2025, 16 (1), 7476, DOI: 10.1038/s41467-025-62369-7There is no corresponding record for this reference.
- 18Wang, Z.; Hao, M.; Li, C.; Gao, X.; Yu, S.; Zhao, L.; Zhang, H.; Li, G.; Li, Y. Electron-Rich Nanorelay Enhances Ligand-to-Metal Charge Transfer in Lanthanide Metal-Organic Frameworks during Photocatalytic CO2 Conversion. Adv. Mater. 2026, 38, e12763 DOI: 10.1002/adma.202512763There is no corresponding record for this reference.
- 19Ding, G.; Wang, Z.; Chen, Z.; Xiao, Y.; Liu, X.; Shuai, L.; Chen, L.; Huang, H.; Liao, G. Unsaturated Coordination-Regulated High-Spin Nickel Sites for Selective Solar-Driven Carbon Dioxide Conversion in Pure Water. Energy Environ. Sci. 2026, 19, 486, DOI: 10.1039/D5EE04331JThere is no corresponding record for this reference.
- 20Cui, Y.; Wang, Y.; Ma, Y.; Su, X.; Tai, R.; Jia, M.; Chen, J.; Liu, A.; Yu, L.; Tian, C.; Zhang, W.; Han, L.; Che, S.; Fang, Y. A Chiral Mesostructured Photocatalyst for Efficient Solar-Driven CO2 Reduction to Ethanol. Nat. Synth. 2026, 5, 117, DOI: 10.1038/s44160-025-00908-2There is no corresponding record for this reference.
- 21Kuang, M.; Li, B.; Zhou, L.; Huang, Z.; Wu, J.; Wang, S.; Yang, J. Carbon Dioxide Upgrading to Biodegradable Plastics through Photo/Electro-Synthetic Biohybrid Systems. Angew. Chem., Int. Ed. 2025, 64 (5), e202422357 DOI: 10.1002/anie.202422357There is no corresponding record for this reference.
- 22Li, Y.; Zhang, B.; Wu, Y.; Tang, T.; Guan, A.; Xu, Q.; Wang, L.; Liu, J.; Sun, L.; Zeng, A.-P. Ultrafast Conversion of CO2 into C3-C4 Diols in a Synergistic Electrochemical and AI-Assisted Biosynthesis System. J. Am. Chem. Soc. 2025, 147 (43), 39827– 39837, DOI: 10.1021/jacs.5c14039There is no corresponding record for this reference.
- 23Pu, Y.; Wang, Y.; Wu, G.; Wu, X.; Lu, Y.; Yu, Y.; Chu, N.; He, X.; Li, D.; Zeng, R. J.; Jiang, Y. Tandem Acidic CO2 Electrolysis Coupled with Syngas Fermentation: A Two-Stage Process for Producing Medium-Chain Fatty Acids. Environ. Sci. Technol. 2024, 58 (17), 7445– 7456, DOI: 10.1021/acs.est.3c09291There is no corresponding record for this reference.
- 24Zhang, W.; Hu, Y.; Ma, L.; Zhu, G.; Wang, Y.; Xue, X.; Chen, R.; Yang, S.; Jin, Z. Progress and Perspective of Electrocatalytic CO2 Reduction for Renewable Carbonaceous Fuels and Chemicals. Adv. Sci. 2018, 5 (1), 1700275, DOI: 10.1002/advs.201700275There is no corresponding record for this reference.
- 25Feng, Y.-X.; Hao, J.; Zhu, S.; Zhang, B.-W.; Chen, Y. Copper Oxide Foam with High Specific Surface Area for Electrocatalytic Reduction of CO2 to C2+ Products. cMat 2024, 1 (1), e19 DOI: 10.1002/cmt2.19There is no corresponding record for this reference.
- 26Shi, R.; Guo, J.; Zhang, X.; Waterhouse, G. I. N.; Han, Z.; Zhao, Y.; Shang, L.; Zhou, C.; Jiang, L.; Zhang, T. Efficient Wettability-Controlled Electroreduction of CO2 to CO at Au/C Interfaces. Nat. Commun. 2020, 11 (1), 3028, DOI: 10.1038/s41467-020-16847-9There is no corresponding record for this reference.
- 27Yang, H.; Shang, L.; Zhang, Q.; Shi, R.; Waterhouse, G. I. N.; Gu, L.; Zhang, T. A Universal Ligand Mediated Method for Large Scale Synthesis of Transition Metal Single Atom Catalysts. Nat. Commun. 2019, 10 (1), 4585, DOI: 10.1038/s41467-019-12510-0There is no corresponding record for this reference.
- 28Yan, T.; Chen, X.; Kumari, L.; Lin, J.; Li, M.; Fan, Q.; Chi, H.; Meyer, T. J.; Zhang, S.; Ma, X. Multiscale CO2 Electrocatalysis to C2+ Products: Reaction Mechanisms, Catalyst Design, and Device Fabrication. Chem. Rev. 2023, 123 (17), 10530– 10583, DOI: 10.1021/acs.chemrev.2c00514There is no corresponding record for this reference.
- 29Tang, Y.-F.; Liu, L.-B.; Yu, M.; Liu, S.; Sui, P.-F.; Sun, W.; Fu, X.-Z.; Luo, J.-L.; Liu, S. Strong Effect-Correlated Electrochemical CO2 Reduction. Chem. Soc. Rev. 2024, 53 (18), 9344– 9377, DOI: 10.1039/D4CS00229FThere is no corresponding record for this reference.
- 30Gao, G.; Khiarak, B. N.; Liu, H.; Trȃ̀n-Phú, T.; Obasanjo, C. A.; Crane, J.; Lai, H. D. T.; da Silva, G. T. S. T.; Golovanova, V.; Li, J.; Ze, H.; Weiss, J.; Zhang, Z.; Lee, S.; Hocking, R. K.; García de Arquer, F. P.; Sargent, E. H.; Dinh, C.-T. Recoverable Operation Strategy for Selective and Stable Electrochemical Carbon Dioxide Reduction to Methane. Nat. Energy 2025, 10 (11), 1360– 1370, DOI: 10.1038/s41560-025-01883-wThere is no corresponding record for this reference.
- 31Choi, M.; Bae, S.; Kim, Y.; Lee, Y.; Cho, M.; Kang, S.; Lee, J. Selective Formaldehyde Condensation on Phosphorus-Rich Copper Catalyst to Produce Liquid C3+ Chemicals in Electrocatalytic CO2 Reduction. Nat. Catal. 2025, 8 (5), 476– 486, DOI: 10.1038/s41929-025-01341-6There is no corresponding record for this reference.
- 32Liu, Y.-C.; Huang, J.-R.; Zhu, H.-L.; Qiu, X.-F.; Yu, C.; Chen, X.-M.; Liao, P.-Q. Electrosynthesis of Pure Urea from Pretreated Flue Gas in a Proton-Limited Environment Established in a Porous Solid-State Electrolyte Electrolyser. Nat. Nanotechnol. 2025, 20 (7), 907– 913, DOI: 10.1038/s41565-025-01914-3There is no corresponding record for this reference.
- 33Huang, D.-S.; Wang, Y.; Tang, Y.; Huang, J.-R.; Li, P.-X.; Liang, C.-P.; Zhao, Z.-H.; Liao, P.-Q.; Chen, X.-M. Embedding a Self-Supporting MOF-Based Molecular Sieve Membrane into an Electrolyzer for Boosting Electroreduction of CO2 in Air and Flue Gas to HCOOH. Natl. Sci. Rev. 2025, 12 (10), nwaf329, DOI: 10.1093/nsr/nwaf329There is no corresponding record for this reference.
- 34Zhao, Z.-H.; Huang, J.-R.; Huang, D.-S.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Efficient Capture and Electroreduction of Dilute CO2 into Highly Pure and Concentrated Formic Acid Aqueous Solution. J. Am. Chem. Soc. 2024, 146 (20), 14349– 14356, DOI: 10.1021/jacs.4c04841There is no corresponding record for this reference.
- 35Liang, C.-P.; Huang, J.-R.; Zhu, H.-L.; Zhao, Z.-H.; Yu, C.; Liao, P.-Q.; Chen, X.-M. Precisely Tailoring the First Coordination Shell of Metal Centers in Porous Nitrogen-Doped Carbon Promoting Electroreduction of CO2 Under Neutral Condition. CCS Chem. 2024, 6 (8), 1978– 1986, DOI: 10.31635/ccschem.023.202303333There is no corresponding record for this reference.
- 36Huang, D.-S.; Qiu, X.-F.; Huang, J.-R.; Mao, M.; Liu, L.; Han, Y.; Zhao, Z.-H.; Liao, P.-Q.; Chen, X.-M. Electrosynthesis of Urea by Using Fe2O3 Nanoparticles Encapsulated in a Conductive Metal-Organic Framework. Nat. Synth. 2024, 3 (11), 1404– 1413, DOI: 10.1038/s44160-024-00603-8There is no corresponding record for this reference.
- 37Liu, Y.-Y.; Huang, J.-R.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Simultaneous Capture of CO2 Boosting Its Electroreduction in the Micropores of a Metal-Organic Framework. Angew. Chem., Int. Ed. 2023, 62 (52), e202311265 DOI: 10.1002/anie.202311265There is no corresponding record for this reference.
- 38Guan, R.; Sheng, L.; Li, C.; Gu, J.; Seo, J.-M.; Jang, B.-J.; Kim, S.-H.; Kim, J.; Lim, H.; Li, Q.; Baek, J.-B. Mechanochemical Carbon Dioxide Capture and Conversion. Nat. Nanotechnol. 2025, 20 (9), 1247– 1253, DOI: 10.1038/s41565-025-01949-6There is no corresponding record for this reference.
- 39Birdja, Y. Y.; Pérez-Gallent, E.; Figueiredo, M. C.; Göttle, A. J.; Calle-Vallejo, F.; Koper, M. T. M. Advances and Challenges in Understanding the Electrocatalytic Conversion of Carbon Dioxide to Fuels. Nat. Energy 2019, 4 (9), 732– 745, DOI: 10.1038/s41560-019-0450-yThere is no corresponding record for this reference.
- 40Song, Y.; Musgrave, C. B.; Su, J.; Huang, L.; Guo, W.; Liu, Y.; Li, G.; Xin, Y.; Zhang, Q.; Feng, X.; Liao, C.; Liu, S.; Kwok, R. T. K.; Lam, J. W. Y.; He, M.; Choong, K. S.; Feng, Z.; Tang, B. Z.; Goddard, W. A.; Ye, R. Efficient CO2-to-Methanol Electrocatalysis in Acidic Media via Microenvironment-Tuned Cobalt Phthalocyanine. Nat. Nanotechnol. 2026, 21, 78, DOI: 10.1038/s41565-025-02059-zThere is no corresponding record for this reference.
- 41Guan, Y.; Li, Y.; Li, Z.; Hou, Y.; Lei, L.; Yang, B. Promotion of C-C Coupling in the CO2 Electrochemical Reduction to Valuable C2+ Products: From Micro-Foundation to Macro-Application. Adv. Mater. 2025, 37 (23), 2417567, DOI: 10.1002/adma.202417567There is no corresponding record for this reference.
- 42Liang, X.; Fu, N.; Yao, S.; Li, Z.; Li, Y. The Progress and Outlook of Metal Single-Atom-Site Catalysis. J. Am. Chem. Soc. 2022, 144 (40), 18155– 18174, DOI: 10.1021/jacs.1c12642There is no corresponding record for this reference.
- 43Taylor, H. S. A Theory of the Catalytic Surface. Proc. A 1925, 108 (745), 105– 111, DOI: 10.1098/rspa.1925.0061There is no corresponding record for this reference.
- 44Qiao, B.; Wang, A.; Yang, X.; Allard, L. F.; Jiang, Z.; Cui, Y.; Liu, J.; Li, J.; Zhang, T. Single-Atom Catalysis of CO Oxidation Using Pt1/FeOx. Nat. Chem. 2011, 3 (8), 634– 641, DOI: 10.1038/nchem.1095There is no corresponding record for this reference.
- 45Liu, Y.; Li, Z.; Yu, Q.; Chen, Y.; Chai, Z.; Zhao, G.; Liu, S.; Cheong, W.-C.; Pan, Y.; Zhang, Q.; Gu, L.; Zheng, L.; Wang, Y.; Lu, Y.; Wang, D.; Chen, C.; Peng, Q.; Liu, Y.; Liu, L.; Chen, J.; Li, Y. A General Strategy for Fabricating Isolated Single Metal Atomic Site Catalysts in Y Zeolite. J. Am. Chem. Soc. 2019, 141 (23), 9305– 9311, DOI: 10.1021/jacs.9b02936There is no corresponding record for this reference.
- 46Xiong, Y.; Dong, J.; Huang, Z.-Q.; Xin, P.; Chen, W.; Wang, Y.; Li, Z.; Jin, Z.; Xing, W.; Zhuang, Z.; Ye, J.; Wei, X.; Cao, R.; Gu, L.; Sun, S.; Zhuang, L.; Chen, X.; Yang, H.; Chen, C.; Peng, Q.; Chang, C.-R.; Wang, D.; Li, Y. Single-Atom Rh/N-Doped Carbon Electrocatalyst for Formic Acid Oxidation. Nat. Nanotechnol. 2020, 15 (5), 390– 397, DOI: 10.1038/s41565-020-0665-xThere is no corresponding record for this reference.
- 47Li, Z.; Chen, Y.; Ji, S.; Tang, Y.; Chen, W.; Li, A.; Zhao, J.; Xiong, Y.; Wu, Y.; Gong, Y.; Yao, T.; Liu, W.; Zheng, L.; Dong, J.; Wang, Y.; Zhuang, Z.; Xing, W.; He, C.-T.; Peng, C.; Cheong, W.-C.; Li, Q.; Zhang, M.; Chen, Z.; Fu, N.; Gao, X.; Zhu, W.; Wan, J.; Zhang, J.; Gu, L.; Wei, S.; Hu, P.; Luo, J.; Li, J.; Chen, C.; Peng, Q.; Duan, X.; Huang, Y.; Chen, X.-M.; Wang, D.; Li, Y. Iridium Single-Atom Catalyst on Nitrogen-Doped Carbon for Formic Acid Oxidation Synthesized Using a General Host-Guest Strategy. Nat. Chem. 2020, 12 (8), 764– 772, DOI: 10.1038/s41557-020-0473-9There is no corresponding record for this reference.
- 48Xiong, Y.; Sun, W.; Xin, P.; Chen, W.; Zheng, X.; Yan, W.; Zheng, L.; Dong, J.; Zhang, J.; Wang, D.; Li, Y. Gram-Scale Synthesis of High-Loading Single-Atomic-Site Fe Catalysts for Effective Epoxidation of Styrene. Adv. Mater. 2020, 32 (34), 2000896, DOI: 10.1002/adma.202000896There is no corresponding record for this reference.
- 49Xiong, Y.; Sun, W.; Han, Y.; Xin, P.; Zheng, X.; Yan, W.; Dong, J.; Zhang, J.; Wang, D.; Li, Y. Cobalt Single Atom Site Catalysts with Ultrahigh Metal Loading for Enhanced Aerobic Oxidation of Ethylbenzene. Nano Res. 2021, 14 (7), 2418– 2423, DOI: 10.1007/s12274-020-3244-4There is no corresponding record for this reference.
- 50Wei, S.; Li, A.; Liu, J.-C.; Li, Z.; Chen, W.; Gong, Y.; Zhang, Q.; Cheong, W.-C.; Wang, Y.; Zheng, L.; Xiao, H.; Chen, C.; Wang, D.; Peng, Q.; Gu, L.; Han, X.; Li, J.; Li, Y. Direct Observation of Noble Metal Nanoparticles Transforming to Thermally Stable Single Atoms. Nat. Nanotechnol. 2018, 13 (9), 856– 861, DOI: 10.1038/s41565-018-0197-9There is no corresponding record for this reference.
- 51Hou, Z.; Lu, Y.; Liu, Y.; Liu, N.; Hu, J.; Wei, L.; Li, Z.; Tian, X.; Gao, R.; Yu, X.; Feng, Y.; Wu, L.; Deng, J.; Wang, D.; Sui, M.; Dai, H.; Li, Y. A General Dual-Metal Nanocrystal Dissociation Strategy to Generate Robust High-Temperature-Stable Alumina-Supported Single-Atom Catalysts. J. Am. Chem. Soc. 2023, 145 (29), 15869– 15878, DOI: 10.1021/jacs.3c02909There is no corresponding record for this reference.
- 52Liang, X.; Yao, S.; Li, Z.; Li, Y. Challenge and Chance of Single Atom Catalysis: The Development and Application of the Single Atom Site Catalysts Toolbox. Acc. Chem. Res. 2025, 58 (10), 1607– 1619, DOI: 10.1021/acs.accounts.4c00857There is no corresponding record for this reference.
- 53Li, Y.; Xu, C.-Q.; Chen, C.; Zhang, Y.; Liu, S.; Zhuang, Z.; Zhang, Y.; Zhang, Q.; Li, Z.; Chen, Z.; Zheng, L.; Cheong, W.-C.; Wu, K.; Jiang, G.; Xiao, H.; Lian, C.; Wang, D.; Peng, Q.; Li, J.; Li, Y. Carbon-Boosted and Nitrogen-Stabilized Isolated Single-Atom Sites for Direct Dehydrogenation of Lower Alkanes. J. Am. Chem. Soc. 2024, 146 (30), 20668– 20677, DOI: 10.1021/jacs.4c03048There is no corresponding record for this reference.
- 54Wang, X.; Fu, N.; Liu, J.-C.; Yu, K.; Li, Z.; Xu, Z.; Liang, X.; Zhu, P.; Ye, C.; Zhou, A.; Li, A.; Zheng, L.; Liu, L.-M.; Chen, C.; Wang, D.; Peng, Q.; Li, Y. Atomic Replacement of PtNi Nanoalloys within Zn-ZIF-8 for the Fabrication of a Multisite CO2 Reduction Electrocatalyst. J. Am. Chem. Soc. 2022, 144 (50), 23223– 23229, DOI: 10.1021/jacs.2c11497There is no corresponding record for this reference.
- 55Dutta, N.; Giri, B.; Riyaz, M.; Midya, S.; Singh, A.; Bagchi, D.; Mondal, S.; Kediya, S.; Singh, A. K.; Chakraborty, S.; Singh, A. K.; Peter, S. C. Confining Reaction Intermediates in Oxide-Derived Hollow Cu-Zn Bimetallic Catalyst Facilitates Selective Formation of C2+ Alcohols from Electrochemical Carbon Dioxide Reduction. Angew. Chem., Int. Ed. 2026, 65, e23150 DOI: 10.1002/anie.202523150There is no corresponding record for this reference.
- 56Guo, W.; Wang, X.; Ma, Y.; Song, Y.; Chang, L.; Wu, H.; Li, G.; Li, Z.; Xin, Y.; He, M.; Zhang, J.; Yang, T.; Zhu, M.; Shen, H.; Xi, S.; Wang, X.; Gan, L.; Jiang, Q.; Xia, C.; Zhao, S.; Guo, Z.; Wang, Z.; Tang, B. Z.; Ye, R. Local Coordination-Dependent CO2 Reduction Activity of Bimetallic Cu-Al Catalysts for Selective Ethylene/Ethanol Electrosynthesis. Angew. Chem., Int. Ed. 2026, 65, e20291 DOI: 10.1002/anie.202520291There is no corresponding record for this reference.
- 57Cao, X.; Liu, Y.; Yu, R.; Yang, Y.; Li, A.; Li, X.; Mao, J. Breaking Ampere-Level Current Density Limitations in Multi-Carbon Products through Crystal Facet Engineering. Chem. 2025, 0 (0), 102824, DOI: 10.1016/j.chempr.2025.102824There is no corresponding record for this reference.
- 58Yan, Z.; Gao, P.; Li, Z.; Zhang, Y.; Hu, C.; Cao, D.; Cheng, D. Rational Design and Validation of Cu2O/Cu Heterogeneous Interface Catalyst for CO2 Electroreduction to C2+ Products. Small 2025, 21 (25), 2500950, DOI: 10.1002/smll.202500950There is no corresponding record for this reference.
- 59Wang, H.; Yang, H. X.; Xu, Y. N.; Fu, H. Q.; Li, X. Y.; He, J. J.; Niu, Q.; Wu, J. C.; Yuan, H. Y.; Liu, P. F.; Yang, H. G. Retained Oxygen Regulation in Oxide-Derived Copper for Promoted CO2 Electroreduction Toward Multicarbon Products. Angew. Chem., Int. Ed. 2025, 64 (25), e202423889 DOI: 10.1002/anie.202423889There is no corresponding record for this reference.
- 60Arán-Ais, R. M.; Scholten, F.; Kunze, S.; Rizo, R.; Roldan Cuenya, B. The Role of in Situ Generated Morphological Motifs and Cu(i) Species in C2+ Product Selectivity during CO2 Pulsed Electroreduction. Nat. Energy 2020, 5 (4), 317– 325, DOI: 10.1038/s41560-020-0594-9There is no corresponding record for this reference.
- 61Li, L.; Yao, S.; Feng, Y.; Gao, Z.; Wang, J.; Yin, P.; Liu, H.; Zhang, Z. Grain Boundary-Induced Stabilization of Bi3+/Bi0 Coexistence in β-Bi2O3 for Highly Efficient Electrochemical CO2 Reduction to Formate. Appl. Catal. B: Environ. 2025, 372, 125302, DOI: 10.1016/j.apcatb.2025.125302There is no corresponding record for this reference.
- 62Fu, X.; Guo, X.; Shi, P.; Frauenheim, T.; Novoselov, K. S.; Yuan, M.; Wang, M. Control of CO2 Electrocatalysis via Modularly Customizable Graphdiyne. J. Am. Chem. Soc. 2025, 147 (46), 42394– 42405, DOI: 10.1021/jacs.5c11774There is no corresponding record for this reference.
- 63Wang, C.; Zhang, G.; Luo, R.; Wang, Y.; Ma, X.; Zhang, M.; Chang, X.; Zhao, Z.-J.; Wang, T.; Gong, J. Selective CO2 Reduction to Acetate via Controlled sp2/sp3 Carbon Hybridization. Nat. Commun. 2025, 16 (1), 10506, DOI: 10.1038/s41467-025-65504-6There is no corresponding record for this reference.
- 64Wang, L.; Wang, D.; Li, Y. Single-Atom Catalysis for Carbon Neutrality. Carbon Energy 2022, 4 (6), 1021– 1079, DOI: 10.1002/cey2.194There is no corresponding record for this reference.
- 65Li, H.; Fang, L.; Wang, T.; Bai, R.; Zhang, J.; Li, T.; Duan, Z.; Chen, K.-J.; Pan, F. In Situ Modulated Nickel Single Atoms on Bicontinuous Porous Carbon Fibers and Sheets Networks for Acidic CO2 Reduction. Adv. Mater. 2025, 37 (7), 2416337, DOI: 10.1002/adma.202416337There is no corresponding record for this reference.
- 66Ma, Y.; Xiao, T.; Zhu, K.; Zhang, W.; Yin, Z.; Dong, A.; Sun, Z.; Zhao, D.; Li, W. Industry-Level Electrocatalytic CO2 to CO Enabled by 2D Mesoporous Ni Single Atom Catalysts. Angew. Chem., Int. Ed. 2025, 64 (5), e202416629 DOI: 10.1002/anie.202416629There is no corresponding record for this reference.
- 67Li, P.; Mao, Y.; Shin, H.; Yang, Q.; Cheng, X.; Li, Y.; Li, K.; Yu, H.; Mulder, R.; Pang, W. K.; Jin, H.; Zhao, Y.; Zheng, Z.; Finch, E.; Hearn, K.; Jia, B.; Waterhouse, G. I. N.; Wang, Z.; Ma, T. Tandem Amine Scrubbing and CO2 Electrolysis via Direct Piperazine Carbamate Reduction. Nat. Energy 2025, 10 (10), 1262– 1273, DOI: 10.1038/s41560-025-01869-8There is no corresponding record for this reference.
- 68Huang, L.; Gao, G.; Zhao, J.; Roberts, W. L.; Lu, X. Electrocatalytic Upcycling of High-Pressure Captured CO2 to Ethylene. Nat. Catal. 2025, 8 (9), 968– 976, DOI: 10.1038/s41929-025-01411-9There is no corresponding record for this reference.
- 69Wang, M.; Fang, M.; Liu, Y.; Chen, C.; Zhang, Y.; Jia, S.; Wu, H.; He, M.; Han, B. Enhanced Intermediates Inter-Migration on Ag Single-Atom Alloys for Boosting Multicarbon Product Selectivity in CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (19), 16450– 16458, DOI: 10.1021/jacs.5c03057There is no corresponding record for this reference.
- 70Zhao, Y.; Wang, Y.; Yu, Z.; Song, C.; Wang, J.; Huang, H.; Meng, L.; Liu, M.; Liu, L. Gold Single Atom Doped Defective Nanoporous Copper Octahedrons for Electrocatalytic Reduction of Carbon Dioxide to Ethylene. ACS Nano 2025, 19 (4), 4505– 4514, DOI: 10.1021/acsnano.4c13961There is no corresponding record for this reference.
- 71Liu, Y.; Ren, X.; Wang, J.; Wang, H.; Yin, Z.; Wang, Y.; Huang, W.; Hu, X.; Xu, Z. J.; Deng, Y. Spin-State Effect of Tetrahedron-Coordinated Single-Atom Catalysts on CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (24), 20318– 20328, DOI: 10.1021/jacs.4c18550There is no corresponding record for this reference.
- 72Zhang, Y.; Chen, F.; Yang, X.; Guo, Y.; Zhang, X.; Dong, H.; Wang, W.; Lu, F.; Lu, Z.; Liu, H.; Liu, H.; Xiao, Y.; Cheng, Y. Electronic Metal-Support Interaction Modulates Cu Electronic Structures for CO2 Electroreduction to Desired Products. Nat. Commun. 2025, 16 (1), 1956, DOI: 10.1038/s41467-025-57307-6There is no corresponding record for this reference.
- 73Jeon, S.; Nedzbala, H. S.; Huffman, B. L.; Pearce, A. J.; Donley, C. L.; Jia, X.; Bein, G. P.; Choi, J.; Durand, N.; Atallah, H.; Castellano, F. N.; Dempsey, J. L.; Mayer, J. M.; Hazari, N.; Stach, E. A. Statistical Analysis of HAADF-STEM Images to Determine the Surface Coverage and Distribution of Immobilized Molecular Complexes. Matter 2025, 8 (2), 101919, DOI: 10.1016/j.matt.2024.11.013There is no corresponding record for this reference.
- 74Chantler, C. T.; Bunker, G.; D’Angelo, P.; Diaz-Moreno, S. X-Ray Absorption Spectroscopy. Nat. Rev. Methods Primers 2024, 4 (1), 89, DOI: 10.1038/s43586-024-00366-8There is no corresponding record for this reference.
- 75Greenwood, N. N. Mössbauer Spectra and Chemical Structure. Nature 1965, 205 (4967), 119– 119, DOI: 10.1038/205119a0There is no corresponding record for this reference.
- 76Beniya, A.; Higashi, S. Towards Dense Single-Atom Catalysts for Future Automotive Applications. Nat. Catal. 2019, 2 (7), 590– 602, DOI: 10.1038/s41929-019-0282-yThere is no corresponding record for this reference.
- 77Liu, Y.; Yue, Z.; Jin, C.; Zheng, L.; Shi, J.; Li, D.; Wang, Y.; Bai, J.; Leng, K.; Wang, W.; Qu, Y.; Li, Q. Isolated Tin Enhanced CO Coverage-Regulation on Sn1Cu Alloy for Selective CO2 Electroreduction to C2+ Products. Small 2025, 21 (8), 2409259, DOI: 10.1002/smll.202409259There is no corresponding record for this reference.
- 78Xu, Y. N.; Li, J.; Wu, J. C.; Li, W.; Yang, Y.; Wu, H.; Fu, H. Q.; Zhu, M.; Wang, X. L.; Dai, S.; Lian, C.; Liu, P. F.; Yang, H. G. Orbital Matching Mechanism-Guided Synthesis of Cu-Based Single Atom Alloys for Acidic CO2 Electroreduction. Adv. Mater. 2025, 37 (18), 2500343, DOI: 10.1002/adma.202500343There is no corresponding record for this reference.
- 79Du, J.-H.; Liu, Z.; Sheng, T.; Liu, J.; Ye, J.; Li, Y.; Chen, S.; Zhang, Y.; Gu, X. Host-Guest Metal Interaction in Cu-In Single Atom Alloy Switching Electrocatalytic CO2 Reduction Pathway. Angew. Chem., Int. Ed. 2025, 64 (43), e202512970 DOI: 10.1002/anie.202512970There is no corresponding record for this reference.
- 80Choi, J.; Kim, S.; Choi, J. Y.; Park, S.; Je, K.; Liu, S.; Jiang, J.; Yang, S.; Pollock, C. J.; Guzman-Soriano, R.; Bairley, K.; Ahmad, B. I. Z.; Milner, P. J.; Thiede, E. H.; Hwang, Y. J.; Park, J.; Yang, Y. Dynamic Evolution from Single-Atom Catalysts to Active Nanograins for CO2 Reduction. J. Am. Chem. Soc. 2025, 147 (41), 37808– 37818, DOI: 10.1021/jacs.5c14123There is no corresponding record for this reference.
- 81Mei, Z.; He, Y.; Liu, K.; Luo, W.; Tan, Y.; Chen, Q.; Wang, H.; Guo, X.; Wu, Q.; Ma, C.; Fu, J.; Lin, Z.; Liu, M. Enhanced *COOH Adsorption over Edge-Rich Ni-N4 Sites for Efficient Acidic CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (37), 34101– 34108, DOI: 10.1021/jacs.5c12583There is no corresponding record for this reference.
- 82Lu, T.; Shi, G.; Liu, Y.; Luo, X.; Shen, Y.; Chang, M.; Wu, Y.; Gao, X.; Wu, J.; Li, Y.; Wang, Y.; Zhang, L. Local Coordination Environment-Driven Structural Dynamics of Single-Atom Copper and the CO2 Electroreduction Pathway. J. Am. Chem. Soc. 2025, 147 (30), 26425– 26436, DOI: 10.1021/jacs.5c05984There is no corresponding record for this reference.
- 83Yang, Z.; Zhang, J.; Tan, X.; Zhu, H.; Xu, L.; Wu, X.; Xu, Z.; Guo, J.; Jia, D.; Chen, C. Manipulating the Coordination Environment of Cu Single Atoms via Regulating ZrO2 Support Crystal Phases for Enhanced CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (39), 35985– 35994, DOI: 10.1021/jacs.5c13661There is no corresponding record for this reference.
- 84Daiyan, R.; Zhu, X.; Tong, Z.; Gong, L.; Razmjou, A.; Liu, R.-S.; Xia, Z.; Lu, X.; Dai, L.; Amal, R. Transforming Active Sites in Nickel-Nitrogen-Carbon Catalysts for Efficient Electrochemical CO2 Reduction to CO. Nano Energy 2020, 78, 105213, DOI: 10.1016/j.nanoen.2020.105213There is no corresponding record for this reference.
- 85Hao, Q.; Tang, Q.; Zheng, L.; Liu, K.; Wu, J.; Lu, J. Specific Construction of Asymmetric Carbon-Nickel-Chlorine Single-Atom Sites via Carbon Vacancy Engineering for Efficient CO2 Electroreduction. Nat. Commun. 2025, 16 (1), 8365, DOI: 10.1038/s41467-025-62287-8There is no corresponding record for this reference.
- 86Zou, H.; Zhao, G.; Dai, H.; Dong, H.; Luo, W.; Wang, L.; Lu, Z.; Luo, Y.; Zhang, G.; Duan, L. Electronic Perturbation of Copper Single-Atom CO2 Reduction Catalysts in a Molecular Way. Angew. Chem., Int. Ed. 2023, 62 (6), e202217220 DOI: 10.1002/anie.202217220There is no corresponding record for this reference.
- 87Chen, R.; Zhao, J.; Zhang, X.; Zhao, Q.; Li, Y.; Cui, Y.; Zhong, M.; Wang, J.; Li, X.; Huang, Y.; Liu, B. Visualizing Catalytic Dynamics of Single-Cu-Atom-Modified SnS2 in CO2 Electroreduction via Rapid Freeze-Quench Mössbauer Spectroscopy. J. Am. Chem. Soc. 2024, 146 (35), 24368– 24376, DOI: 10.1021/jacs.4c05813There is no corresponding record for this reference.
- 88Liao, L.; Xia, G.; Yu, F.; Liu, X.; Shu, M.; Zhang, G.; Zeng, X.; Wang, H. Saturated Coordination Lu-N6 Defect Sites for Highly Efficient Electroreduction of CO2. Small 2023, 19 (37), 2300926, DOI: 10.1002/smll.202300926There is no corresponding record for this reference.
- 89Wang, H.; Sui, H.; Ding, Y.; Yang, Y.; Su, Y.; Li, H. Tailoring CO2 Adsorption Configuration with Spatial Confinement Switches Electroreduction Product from Formate to Acetate. J. Am. Chem. Soc. 2025, 147 (7), 6095– 6107, DOI: 10.1021/jacs.4c17295There is no corresponding record for this reference.
- 90Song, J.; Lei, X.; Mu, J.; Li, J.; Song, X.; Yan, L.; Ding, Y. Chlorine-Coordinated Unsaturated Ni-N2 Sites for Efficient Electrochemical Carbon Dioxide Reduction. Small 2023, 19 (52), 2304423, DOI: 10.1002/smll.202304423There is no corresponding record for this reference.
- 91Qu, M.; Chen, Z.; Sun, Z.; Zhou, D.; Xu, W.; Tang, H.; Gu, H.; Liang, T.; Hu, P.; Li, G.; Wang, Y.; Chen, Z.; Wang, T.; Jia, B. Rational Design of Asymmetric Atomic Ni-P1N3 Active Sites for Promoting Electrochemical CO2 Reduction. Nano Res. 2023, 16 (2), 2170– 2176, DOI: 10.1007/s12274-022-4969-zThere is no corresponding record for this reference.
- 92Song, W.; Xiao, C.; Ding, J.; Huang, Z.; Yang, X.; Zhang, T.; Mitlin, D.; Hu, W. Review of Carbon Support Coordination Environments for Single Metal Atom Electrocatalysts (SACS). Adv. Mater. 2024, 36 (1), 2301477, DOI: 10.1002/adma.202301477There is no corresponding record for this reference.
- 93Bhawnani, R. R.; Sartape, R.; Gande, V. V.; Barsoum, M. L.; Kallon, E. M.; dos Reis, R.; Dravid, V. P.; Singh, M. R. Non-Aqueous Electrochemical CO2 Reduction to Multivariate C2-Products Over Single Atom Catalyst at Current Density up to 100 mA cm–2. Small 2025, 21 (18), 2408010, DOI: 10.1002/smll.202408010There is no corresponding record for this reference.
- 94Ma, G.; Chen, Y.; Li, H.; Sun, Y.; Fan, H.; Jiang, S.; Cui, X.; Ma, T. Hierarchically Porous N-Doped Carbon Confined Single-Atom Fe Catalyst for Efficient Electrochemical CO2 Reduction. Nano Res. 2025, 18 (4), 94907297, DOI: 10.26599/NR.2025.94907297There is no corresponding record for this reference.
- 95Yin, Z.; Yu, J.; Xie, Z.; Yu, S.-W.; Zhang, L.; Akauola, T.; Chen, J. G.; Huang, W.; Qi, L.; Zhang, S. Hybrid Catalyst Coupling Single-Atom Ni and Nanoscale Cu for Efficient CO2 Electroreduction to Ethylene. J. Am. Chem. Soc. 2022, 144 (45), 20931– 20938, DOI: 10.1021/jacs.2c09773There is no corresponding record for this reference.
- 96Chen, S.; Wang, B.; Zhu, J.; Wang, L.; Ou, H.; Zhang, Z.; Liang, X.; Zheng, L.; Zhou, L.; Su, Y.-Q.; Wang, D.; Li, Y. Lewis Acid Site-Promoted Single-Atomic Cu Catalyzes Electrochemical CO2 Methanation. Nano Lett. 2021, 21 (17), 7325– 7331, DOI: 10.1021/acs.nanolett.1c02502There is no corresponding record for this reference.
- 97Liu, K.; Sun, Z.; Chen, W.; Lang, X.; Gao, X.; Chen, P. Ultra-Fast Pulsed Discharge Preparation of Coordinatively Unsaturated Asymmetric Copper Single-Atom Catalysts for CO2 Reduction. Adv. Funct. Mater. 2024, 34 (16), 2312589, DOI: 10.1002/adfm.202312589There is no corresponding record for this reference.
- 98Zheng, J.; Xu, S.; Sun, L.; Pan, X.; Xie, Q.; Zhao, G. Boosting the Reduction of CO2 and Dimethylamine for C-N Bonding to Synthesize DMF via Modulating the Electronic Structures of Indium Single Atoms. Energy Environ. Sci. 2025, 18 (8), 3614– 3622, DOI: 10.1039/D4EE05681GThere is no corresponding record for this reference.
- 99Ping, D.; Huang, S.; Wu, S.; Zhang, Y.; Wang, S.; Yang, X.; Han, L.; Tian, J.; Guo, D.; Qiu, H.-J.; Fang, S. Confinement Effect and 3D Design Endow Unsaturated Single Ni Atoms with Ultrahigh Stability and Selectivity toward CO2 Electroreduction. Small 2024, 20 (14), 2309014, DOI: 10.1002/smll.202309014There is no corresponding record for this reference.
- 100Xia, W.; Xie, Y.; Jia, S.; Han, S.; Qi, R.; Chen, T.; Xing, X.; Yao, T.; Zhou, D.; Dong, X.; Zhai, J.; Li, J.; He, J.; Jiang, D.; Yamauchi, Y.; He, M.; Wu, H.; Han, B. Adjacent Copper Single Atoms Promote C-C Coupling in Electrochemical CO2 Reduction for the Efficient Conversion of Ethanol. J. Am. Chem. Soc. 2023, 145 (31), 17253– 17264, DOI: 10.1021/jacs.3c04612There is no corresponding record for this reference.
- 101Chen, Z.; Wang, C.; Zhong, X.; Lei, H.; Li, J.; Ji, Y.; Liu, C.; Ding, M.; Dai, Y.; Li, X.; Zheng, T.; Jiang, Q.; Peng, H.-J.; Xia, C. Achieving Efficient CO2 Electrolysis to CO by Local Coordination Manipulation of Nickel Single-Atom Catalysts. Nano Lett. 2023, 23 (15), 7046– 7053, DOI: 10.1021/acs.nanolett.3c01808There is no corresponding record for this reference.
- 102Wang, Z.; Chen, Z.; Du, X.; Zhang, Y.; Liu, Z.; Zang, S.-Q. Modulating the Electronic Properties of Single Ni Atom Catalyst via First-Shell Coordination Engineering to Boost Electrocatalytic Flue Gas CO2 Reduction. Adv. Funct. Mater. 2025, 35 (20), 2420994, DOI: 10.1002/adfm.202420994There is no corresponding record for this reference.
- 103Zang, Y.; Liu, Y.; Lu, R.; Yang, Q.; Wang, B.; Zhang, M.; Mao, Y.; Wang, Z.; Lum, Y. Tuning Transition Metal 3d Spin State on Single-Atom Catalysts for Selective Electrochemical CO2 Reduction. Adv. Mater. 2025, 37 (16), 2417034, DOI: 10.1002/adma.202417034There is no corresponding record for this reference.
- 104Chen, Y.; Pan, X.; Li, L.; Chen, M.; Cao, H.; Zhao, Y.; Wang, X.; Lin, J. Modulating Electronic Density of Single-Atom Ni Center by Heteroatoms for Efficient CO2 Electroreduction. Small 2025, 21 (9), 2411249, DOI: 10.1002/smll.202411249There is no corresponding record for this reference.
- 105Dai, Y.; Li, H.; Wang, C.; Xue, W.; Zhang, M.; Zhao, D.; Xue, J.; Li, J.; Luo, L.; Liu, C.; Li, X.; Cui, P.; Jiang, Q.; Zheng, T.; Gu, S.; Zhang, Y.; Xiao, J.; Xia, C.; Zeng, J. Manipulating Local Coordination of Copper Single Atom Catalyst Enables Efficient CO2-to-CH4 Conversion. Nat. Commun. 2023, 14 (1), 3382, DOI: 10.1038/s41467-023-39048-6There is no corresponding record for this reference.
- 106Song, J.; Lei, X.; Mu, J.; Li, J.; Song, X.; Yan, L.; Ding, Y. Boron-Doped Nickel-Nitrogen-Carbon Single-Atom Catalyst for Boosting Electrochemical CO2 Reduction. Small 2023, 19 (52), 2305666, DOI: 10.1002/smll.202305666There is no corresponding record for this reference.
- 107Guo, J.; Zhang, W.; Zhang, L.-H.; Chen, D.; Zhan, J.; Wang, X.; Shiju, N. R.; Yu, F. Control over Electrochemical CO2 Reduction Selectivity by Coordination Engineering of Tin Single-Atom Catalysts. Adv. Sci. 2021, 8 (23), 2102884, DOI: 10.1002/advs.202102884There is no corresponding record for this reference.
- 108Wang, Q.; Dai, M.; Li, H.; Lu, Y.-R.; Chan, T.-S.; Ma, C.; Liu, K.; Fu, J.; Liao, W.; Chen, S.; Pensa, E.; Wang, Y.; Zhang, S.; Sun, Y.; Cortés, E.; Liu, M. Asymmetric Coordination Induces Electron Localization at Ca Sites for Robust CO2 Electroreduction to CO. Adv. Mater. 2023, 35 (21), 2300695, DOI: 10.1002/adma.202300695There is no corresponding record for this reference.
- 109Wei, S.; Zhu, J.; Chen, X.; Yang, R.; Gu, K.; Li, L.; Chiang, C.-Y.; Mai, L.; Chen, S. Planar Chlorination Engineering Induced Symmetry-Broken Single-Atom Site Catalyst for Enhanced CO2 Electroreduction. Nat. Commun. 2025, 16 (1), 1652, DOI: 10.1038/s41467-025-56271-5There is no corresponding record for this reference.
- 110Peng, J.-X.; Yang, W.; Jia, Z.; Jiao, L.; Jiang, H.-L. Axial Coordination Regulation of MOF-Based Single-Atom Ni Catalysts by Halogen Atoms for Enhanced CO2 Electroreduction. Nano Res. 2022, 15 (12), 10063– 10069, DOI: 10.1007/s12274-022-4467-3There is no corresponding record for this reference.
- 111Zhao, J.; Chen, Y.; Liu, D.; Fai Ip, W.; Lin, J.; Wang, X.; Lin, S.; Fu, X.; Zhang, T. “Outside-in” Design of Single-Atom Catalysts: Linking Specific Peripheral Geometry to Defined CO2 Reduction Performance. Angew. Chem., Int. Ed. 2025, 64 (34), e202511184 DOI: 10.1002/anie.202511184There is no corresponding record for this reference.
- 112Ni, W.; He, X.; Chen, H.; Dai, M.; Zhang, W.; Zhang, Y.; Wang, S.; Zhang, S. 93% Single-Atom Utilization in Base-Resistant Metal-Organic Framework Quantum Dots for Ampere-Level CO2 Electroreduction. Energy Environ. Sci. 2024, 17 (9), 3191– 3201, DOI: 10.1039/D4EE00720DThere is no corresponding record for this reference.
- 113Li, L.; Lei, X.; Zheng, Z.; Dong, Y.; Chen, H.; Chen, J.; Zhong, Y.; Zheng, Y.; Tang, Y.; Zhang, X.; Cheng, H.-M. Tuning Binding Strength between Single Metal Atoms and Supports Enhances Electrochemical CO2 Methanation. Nat. Commun. 2025, 16 (1), 8641, DOI: 10.1038/s41467-025-63781-9There is no corresponding record for this reference.
- 114Leverett, J.; Baghestani, G.; Tran-Phu, T.; Yuwono, J. A.; Kumar, P.; Johannessen, B.; Simondson, D.; Wen, H.; Chang, S. L. Y.; Tricoli, A.; Simonov, A. N.; Dai, L.; Amal, R.; Daiyan, R.; Hocking, R. K. Direct Observation of Electron Donation onto the Reactants and a Transient Poisoning Mechanism During CO2 Electroreduction on Ni Single Atom Catalysts. Angew. Chem., Int. Ed. 2025, 64 (18), e202424087 DOI: 10.1002/anie.202424087There is no corresponding record for this reference.
- 115Wang, C.; Chen, B.; Wang, Q.; Hu, H.; Xia, R.; Zhu, J.; Sun, Q.; Wu, Z.; Miao, R. K.; Guan, Q.; Liu, Y.; Liu, M.; Li, W. Dynamic Activation of Edge-Hosted Co-N4 Sites for Energy-Efficient Electrochemical CO2 Reduction at Industry-Level Current Density. ACS Catal. 2025, 15 (10), 8540– 8550, DOI: 10.1021/acscatal.5c01598There is no corresponding record for this reference.
- 116Chang, Q.; Liu, Y.; Lee, J.-H.; Ologunagba, D.; Hwang, S.; Xie, Z.; Kattel, S.; Lee, J. H.; Chen, J. G. Metal-Coordinated Phthalocyanines as Platform Molecules for Understanding Isolated Metal Sites in the Electrochemical Reduction of CO2. J. Am. Chem. Soc. 2022, 144 (35), 16131– 16138, DOI: 10.1021/jacs.2c06953There is no corresponding record for this reference.
- 117Liu, T.; Zhang, D.; Hirai, Y.; Ito, K.; Ishibashi, K.; Todoroki, N.; Matsuo, Y.; Yoshida, J.; Ono, S.; Li, H.; Yabu, H. Surface Charge Transfer Enhanced Cobalt-Phthalocyanine Crystals for Efficient CO2-to-CO Electroreduction with Large Current Density Exceeding 1000 mA cm–2. Adv. Sci. 2025, 12 (23), 2501459, DOI: 10.1002/advs.202501459There is no corresponding record for this reference.
- 118Wang, Z.; Wang, C.; Hu, Y.; Yang, S.; Yang, J.; Chen, W.; Zhou, H.; Zhou, F.; Wang, L.; Du, J.; Li, Y.; Wu, Y. Simultaneous Diffusion of Cation and Anion to Access N, S Co-Coordinated Bi-Sites for Enhanced CO2 Electroreduction. Nano Res. 2021, 14 (8), 2790– 2796, DOI: 10.1007/s12274-021-3287-1There is no corresponding record for this reference.
- 119Lu, Y.; Wang, H.; Yu, P.; Yuan, Y.; Shahbazian-Yassar, R.; Sheng, Y.; Wu, S.; Tu, W.; Liu, G.; Kraft, M.; Xu, R. Isolated Ni Single Atoms in Nitrogen Doped Ultrathin Porous Carbon Templated from Porous g-C3N4 for High-Performance CO2 Reduction. Nano Energy 2020, 77, 105158, DOI: 10.1016/j.nanoen.2020.105158There is no corresponding record for this reference.
- 120Sui, R.; Wang, B.; Wang, Y.; Pei, J.; Zhu, W.; Chen, W.; Li, C.; Sun, A.; Zhuang, Z. A General and Facile Calcination Method to Synthesize Single-Site Catalysts for Highly Efficient Electrochemical CO2 Reduction. Nano Res. 2024, 17 (5), 3895– 3901, DOI: 10.1007/s12274-023-6378-3There is no corresponding record for this reference.
- 121Lv, Z.; Wang, C.; Liu, Y.; Liu, R.; Zhang, F.; Feng, X.; Yang, W.; Wang, B. Improving CO2-to-C2 Conversion of Atomic CuFONC Electrocatalysts through F, O-Codrived Optimization of Local Coordination Environment. Adv. Energy Mater. 2024, 14 (21), 2400057, DOI: 10.1002/aenm.202400057There is no corresponding record for this reference.
- 122Wang, Z.; Qian, J.; Cao, P.; Shou, H.; Wu, C.; Xu, X.; Wu, X.; He, Q.; Song, L. Identification of Synergies in Fe, Co-Coordinated Polyphthalocyanines Scaffolds for Electrochemical CO2 Reduction Reaction. Nano Lett. 2024, 24 (10), 3249– 3256, DOI: 10.1021/acs.nanolett.4c00306There is no corresponding record for this reference.
- 123Liu, M.; Xie, Y.; Liu, F.; Dong, X.; Xia, J.; Wang, H.; Zou, R.; Peng, Y.; Zhang, C.; Xu, Q.; Hou, C.-C. Edge-Rich Graphene Nanomesh Thermally Self-Exfoliated From Metal-Organic Frameworks for Boosting CO2 Electroreduction. Adv. Funct. Mater. 2025, 35 (44), 2509429, DOI: 10.1002/adfm.202509429There is no corresponding record for this reference.
- 124Miao, K.; Qin, J.; Lai, S.; Luo, M.; Kuchkaev, A.; Yakhvarov, D.; Kang, X. Spin Regulation of Nickel Single Atom Catalyst via Axial Phosphor-Coordination Achieves Near Unity CO Selectivity in Electrochemical CO2 Reduction. Adv. Funct. Mater. 2025, 35 (14), 2419989, DOI: 10.1002/adfm.202419989There is no corresponding record for this reference.
- 125Guo, C.; Wang, F.; Abdukayum, A.; Chen, Q.; Chang, F.; Li, H.; An, X.; Hu, G.; Ma, Y. Axial and Asymmetric Coordination Coupling Adjust the Electronic Structure of Single-Atom Zinc Sites for Efficient Electroreduction of Carbon Dioxide. Adv. Sci. 2025, 12 (42), e09698 DOI: 10.1002/advs.202509698There is no corresponding record for this reference.
- 126Wang, R.; Wang, Y.; Chen, D.; Gao, C.; Chen, Z.; Cheng, L.; Deng, Y.; Peng, Y.; Li, J. Improving CO2 Reduction Performance towards C2 Products on a Bimetallic Zn1Cux/NC Electrocatalyst. Nano Res. 2025, 18 (11), 94907659, DOI: 10.26599/NR.2025.94907659There is no corresponding record for this reference.
- 127Liu, M.; Wang, Q.; Luo, T.; Herran, M.; Cao, X.; Liao, W.; Zhu, L.; Li, H.; Stefancu, A.; Lu, Y.-R.; Chan, T.-S.; Pensa, E.; Ma, C.; Zhang, S.; Xiao, R.; Cortés, E. Potential Alignment in Tandem Catalysts Enhances CO2-to-C2H4 Conversion Efficiencies. J. Am. Chem. Soc. 2024, 146 (1), 468– 475, DOI: 10.1021/jacs.3c09632There is no corresponding record for this reference.
- 128Deng, Y.; Zhao, J.; Wang, S.; Chen, R.; Ding, J.; Tsai, H.-J.; Zeng, W.-J.; Hung, S.-F.; Xu, W.; Wang, J.; Jaouen, F.; Li, X.; Huang, Y.; Liu, B. Operando Spectroscopic Analysis of Axial Oxygen-Coordinated Single-Sn-Atom Sites for Electrochemical CO2 Reduction. J. Am. Chem. Soc. 2023, 145 (13), 7242– 7251, DOI: 10.1021/jacs.2c12952There is no corresponding record for this reference.
- 129Yuan, L.; Li, X.; Li, G.; Peng, K.; Zhang, H.; Zeng, S.; Sun, X.; Zhang, X. Gram-Scale Preparation of Tri-Coordinated Single-Atom Catalysts for CO2 Electrolysis in Large-Scale Membrane Electrode Assembly. Adv. Sci. 2025, 12 (18), 2500368, DOI: 10.1002/advs.202500368There is no corresponding record for this reference.
- 130Wang, Y.; Zhu, P.; Wang, R.; Matthews, K. C.; Xie, M.; Wang, M.; Qiu, C.; Liu, Y.; Zhou, H.; Warner, J. H.; Liu, Y.; Wang, H.; Yu, G. Fluorine-Tuned Carbon-Based Nickel Single-Atom Catalysts for Scalable and Highly Efficient CO2 Electrocatalytic Reduction. ACS Nano 2024, 18 (39), 26751– 26758, DOI: 10.1021/acsnano.4c06923There is no corresponding record for this reference.
- 131Li, Y.; Adli, N. M.; Shan, W.; Wang, M.; Zachman, M. J.; Hwang, S.; Tabassum, H.; Karakalos, S.; Feng, Z.; Wang, G.; Li, Y. C.; Wu, G. Atomically Dispersed Single Ni Site Catalysts for High-Efficiency CO2 Electroreduction at Industrial-Level Current Densities. Energy Environ. Sci. 2022, 15 (5), 2108– 2119, DOI: 10.1039/D2EE00318JThere is no corresponding record for this reference.
- 132He, Y.; Li, Y.; Zhang, J.; Wang, S.; Huang, D.; Yang, G.; Yi, X.; Lin, H.; Han, X.; Hu, W.; Deng, Y.; Ye, J. Low-Temperature Strategy toward Ni-NC@Ni Core-Shell Nanostructure with Single-Ni Sites for Efficient CO2 Electroreduction. Nano Energy 2020, 77, 105010, DOI: 10.1016/j.nanoen.2020.105010There is no corresponding record for this reference.
- 133Dong, J.; Liu, Y.; Pei, J.; Li, H.; Ji, S.; Shi, L.; Zhang, Y.; Li, C.; Tang, C.; Liao, J.; Xu, S.; Zhang, H.; Li, Q.; Zhao, S. Continuous Electroproduction of Formate via CO2 Reduction on Local Symmetry-Broken Single-Atom Catalysts. Nat. Commun. 2023, 14 (1), 6849, DOI: 10.1038/s41467-023-42539-1There is no corresponding record for this reference.
- 134Huang, Y.; Hu, H.; Wang, C.; Yuan, X.; Wu, X.; Qiao, Z.; Li, Y.; Wang, C. Oxygen-Bearing Defects in Graphene-Supported M-N-C Single-Atom Catalysts: A Hidden Determinant of Catalytic Performance. ACS Catal. 2025, 15 (17), 15358– 15371, DOI: 10.1021/acscatal.5c03974There is no corresponding record for this reference.
- 135Yang, L.; Pang, H.; Ren, W.; Wang, X.; Wei, Y.; Lu, P.; Chen, J.; Tian, W.; Huang, M.; Wang, H. Joule Heating Driven Graphitization Regulation and Ni Single-Atom Modification in Hard Carbon for Low-Voltage and High-Rate Potassium-Ion Storage. Adv. Funct. Mater. 2026, 36 (6), e16237 DOI: 10.1002/adfm.202516237There is no corresponding record for this reference.
- 136Wu, Y.; Zhuang, Z.; Chen, C.; Li, J.; Xiao, F.; Chen, C. Atomic-Level Regulation Strategies of Single-Atom Catalysts: Nonmetal Heteroatom Doping and Polymetallic Active Site Construction. Chem. Catalysis 2023, 3 (7), 100586, DOI: 10.1016/j.checat.2023.100586There is no corresponding record for this reference.
- 137Ruan, Y.; Guo, W.; Lu, Y.; Zhou, H.; Wu, Y. Recent Advances in ″Top-down″ Synthesis Strategies for Single-Atom Catalysts. Sci. China Chem. 2025, 68, 6248– 6271, DOI: 10.1007/s11426-025-3120-4There is no corresponding record for this reference.
- 138Cai, R.; Zhu, H.; Yang, F.; Ju, M.; Huang, X.; Wang, J.; Gu, M. D.; Gao, J.; Yang, S. The Proximal Protonation Source in Cu-NHx-C Single Atom Catalysts Selectively Boosts CO2 to Methane Electroreduction. Angew. Chem., Int. Ed. 2025, 64 (15), e202424098 DOI: 10.1002/anie.202424098There is no corresponding record for this reference.
- 139Wu, X.; Zhao, J. Y.; Sun, J. W.; Li, W. J.; Yuan, H. Y.; Liu, P. F.; Dai, S.; Yang, H. G. Isolation of Highly Reactive Cobalt Phthalocyanine via Electrochemical Activation for Enhanced CO2 Reduction Reaction. Small 2023, 19 (23), 2207037, DOI: 10.1002/smll.202207037There is no corresponding record for this reference.
- 140He, C.; Gong, Y.; Li, S.; Wu, J.; Lu, Z.; Li, Q.; Wang, L.; Wu, S.; Zhang, J. Single-Atom Alloys Materials for CO2 and CH4 Catalytic Conversion. Adv. Mater. 2024, 36 (16), 2311628, DOI: 10.1002/adma.202311628There is no corresponding record for this reference.
- 141Ji, S.; Chen, Y.; Wang, X.; Zhang, Z.; Wang, D.; Li, Y. Chemical Synthesis of Single Atomic Site Catalysts. Chem. Rev. 2020, 120 (21), 11900– 11955, DOI: 10.1021/acs.chemrev.9b00818There is no corresponding record for this reference.
- 142Chhetri, M.; Wan, M.; Jin, Z.; Yeager, J.; Sandor, C.; Rapp, C.; Wang, H.; Lee, S.; Bodenschatz, C. J.; Zachman, M. J.; Che, F.; Yang, M. Dual-Site Catalysts Featuring Platinum-Group-Metal Atoms on Copper Shapes Boost Hydrocarbon Formations in Electrocatalytic CO2 Reduction. Nat. Commun. 2023, 14 (1), 3075, DOI: 10.1038/s41467-023-38777-yThere is no corresponding record for this reference.
- 143Liu, X.; Zhen, C.; Wu, J.; You, X.; Wu, Y.; Hao, Q.; Yu, G.; Gu, M. D.; Liu, K.; Lu, J. Modulating the Hydrogenation Mechanism of Electrochemical CO2 Reduction Using Ruthenium Atomic Species on Bismuth. Adv. Funct. Mater. 2024, 34 (44), 2405835, DOI: 10.1002/adfm.202405835There is no corresponding record for this reference.
- 144Jin, C.; Lin, Y.; Wang, Y.; Shi, J.; Li, R.; Liu, Y.; Yue, Z.; Leng, K.; Zhao, Y.; Wang, Y.; Han, X.; Qu, Y.; Bai, J. Engineering Atom-Scale Cascade Catalysis via Multi-Active Site Collaboration for Ampere-Level CO2 Electroreduction to C2+ Products. Adv. Mater. 2025, 37 (8), 2412658, DOI: 10.1002/adma.202412658There is no corresponding record for this reference.
- 145Tuo, Y.; Lu, Q.; Liu, W.; Wang, M.; Zhou, Y.; Feng, X.; Wu, M.; Chen, D.; Zhang, J. Atomic Zn-Doping Induced Sabatier Optimum in NiZn0.03 Catalyst for CO2 Electroreduction at Industrial-Level Current Densities. Small 2024, 20 (9), 2306945, DOI: 10.1002/smll.202306945There is no corresponding record for this reference.
- 146Chen, D.; Chu, B.; Li, F.; Zheng, Y.-T.; Lu, Y.; Shao, B.; Li, L.; Huang, N.-Y.; Xu, Q. Synergistic Catalysis by Cu Single Atoms and Atomically Cu-Doped Au Nanoparticles in a Metal-Organic Framework for Photocatalytic CO2 Reduction to C2H6. J. Am. Chem. Soc. 2025, 147 (26), 22705– 22713, DOI: 10.1021/jacs.5c04364There is no corresponding record for this reference.
- 147Yuan, H.; Jiang, D.; Li, Z.; Liu, X.; Tang, Z.; Zhang, X.; Zhao, L.; Huang, M.; Liu, H.; Song, K.; Zhou, W. Laser Synthesis of PtMo Single-Atom Alloy Electrode for Ultralow Voltage Hydrogen Generation. Adv. Mater. 2024, 36 (5), 2305375, DOI: 10.1002/adma.202305375There is no corresponding record for this reference.
- 148Wang, H.; Li, J.; Huang, M.; Cui, J.; Cheng, Z.; Yu, R.; Zhu, H. Single-Atom Alloys Prepared by Two-Step Thermal Evaporation. Nano Res. 2024, 17 (4), 2808– 2813, DOI: 10.1007/s12274-023-6146-4There is no corresponding record for this reference.
- 149Yang, J.; Li, W.; Wang, D.; Li, Y. Electronic Metal-Support Interaction of Single-Atom Catalysts and Applications in Electrocatalysis. Adv. Mater. 2020, 32 (49), 2003300, DOI: 10.1002/adma.202003300There is no corresponding record for this reference.
- 150Xia, C.; Wang, X.; He, C.; Qi, R.; Zhu, D.; Lu, R.; Li, F.-M.; Chen, Y.; Chen, S.; You, B.; Yao, T.; Guo, W.; Song, F.; Wang, Z.; Xia, B. Y. Highly Selective Electrocatalytic CO2 Conversion to Tailored Products through Precise Regulation of Hydrogenation and C-C Coupling. J. Am. Chem. Soc. 2024, 146 (29), 20530– 20538, DOI: 10.1021/jacs.4c07502There is no corresponding record for this reference.
- 151Ding, G.; Yu, Y.; Yan, D.; Zhang, J.; Shuai, L.; Chen, L.; Liao, G. Pt-Activated Surface Nucleophilicity on High-Entropy Oxides Enables Superior Biomass Upgrading. Adv. Funct. Mater. 2025, e19698 DOI: 10.1002/adfm.202519698There is no corresponding record for this reference.
- 152Hu, F.; He, B.; Chen, K.; Ma, W.; Huang, Y.; Zhao, S.; Chen, Y.; Zhao, L. Single-Atom Anchored on Perovskite With Strong Metal-Oxide Interaction for Efficient High Temperature CO2 Electrolysis. Adv. Mater. 2026, 38, e12310 DOI: 10.1002/adma.202512310There is no corresponding record for this reference.
- 153Wu, B.; Li, X.; Hou, T.; Chen, X.; Wang, J.; Li, S.; Chen, A.; Zhu, C.; Zhang, X.; Ou, H.; Wang, Y.; Wang, D.; Zhang, J. Oxygen-Bridged Cu and V Dual Metal Sites for Enhanced Photo-Oxidative Coupling of Benzylamine. J. Am. Chem. Soc. 2025, 147 (51), 46997– 47007, DOI: 10.1021/jacs.5c11918There is no corresponding record for this reference.
- 154Li, J.-Y.; Huang, J.-R.; Zhao, Z.-H.; Zhu, H.-L.; Liao, P.-Q.; Chen, X.-M. Low-Coordination Indium Single-Atom Sites Anchored on a Metal-Organic Framework Single-Layer Boosts Electroreduction of CO2 Into Formic Acid. Angew. Chem., Int. Ed. 2025, 64 (36), e202511132 DOI: 10.1002/anie.202511132There is no corresponding record for this reference.
- 155Chen, H.; Huang, Q.; Yang, K.; Chen, S.; Feng, H.; Jia, C.; Li, Q.; Zhao, C.; Duan, J. Tunable Ag-Ox Coordination for Industrial-Level Carbon-Negative CO2 Electrolysis. Nano Energy 2024, 131, 110265, DOI: 10.1016/j.nanoen.2024.110265There is no corresponding record for this reference.
- 156Zhao, Q.; Zhang, C.; Hu, R.; Du, Z.; Gu, J.; Cui, Y.; Chen, X.; Xu, W.; Cheng, Z.; Li, S.; Li, B.; Liu, Y.; Chen, W.; Liu, C.; Shang, J.; Song, L.; Yang, S. Selective Etching Quaternary MAX Phase toward Single Atom Copper Immobilized MXene (Ti3C2Clx) for Efficient CO2 Electroreduction to Methanol. ACS Nano 2021, 15 (3), 4927– 4936, DOI: 10.1021/acsnano.0c09755There is no corresponding record for this reference.
- 157Ding, J.; Bin Yang, H.; Ma, X.-L.; Liu, S.; Liu, W.; Mao, Q.; Huang, Y.; Li, J.; Zhang, T.; Liu, B. A Tin-Based Tandem Electrocatalyst for CO2 Reduction to Ethanol with 80% Selectivity. Nat. Energy 2023, 8 (12), 1386– 1394, DOI: 10.1038/s41560-023-01389-3There is no corresponding record for this reference.
- 158Xu, H.; Rebollar, D.; He, H.; Chong, L.; Liu, Y.; Liu, C.; Sun, C.-J.; Li, T.; Muntean, J. V.; Winans, R. E.; Liu, D.-J.; Xu, T. Highly Selective Electrocatalytic CO2 Reduction to Ethanol by Metallic Clusters Dynamically Formed from Atomically Dispersed Copper. Nat. Energy 2020, 5 (8), 623– 632, DOI: 10.1038/s41560-020-0666-xThere is no corresponding record for this reference.
- 159Jiang, Y.; Mao, K.; Li, J.; Duan, D.; Li, J.; Wang, X.; Zhong, Y.; Zhang, C.; Liu, H.; Gong, W.; Long, R.; Xiong, Y. Pushing the Performance Limit of Cu/CeO2 Catalyst in CO2 Electroreduction: A Cluster Model Study for Loading Single Atoms. ACS Nano 2023, 17 (3), 2620– 2628, DOI: 10.1021/acsnano.2c10534There is no corresponding record for this reference.
- 160Guo, Z.; Zhou, P.; Jiang, L.; Liu, S.; Yang, Y.; Li, Z.; Wu, P.; Zhang, Z.; Li, H. Electron Localization-Triggered Proton Pumping Toward Cu Single Atoms for Electrochemical CO2 Methanation of Unprecedented Selectivity. Adv. Mater. 2024, 36 (14), 2311149, DOI: 10.1002/adma.202311149There is no corresponding record for this reference.
- 161Xu, H.; Wang, J.; He, H.; Hwang, I.; Liu, Y.; Sun, C.; Zhang, H.; Li, T.; Muntean, J. V.; Xu, T.; Liu, D.-J. Modulating CO2 Electrocatalytic Conversion to the Organics Pathway by the Catalytic Site Dimension. J. Am. Chem. Soc. 2024, 146 (15), 10357– 10366, DOI: 10.1021/jacs.3c12722There is no corresponding record for this reference.
- 162Chen, R.; Zhao, J.; Li, Y.; Cui, Y.; Lu, Y.-R.; Hung, S.-F.; Wang, S.; Wang, W.; Huo, G.; Zhao, Y.; Liu, W.; Wang, J.; Xiao, H.; Li, X.; Huang, Y.; Liu, B. Operando Mössbauer Spectroscopic Tracking the Metastable State of Atomically Dispersed Tin in Copper Oxide for Selective CO2 Electroreduction. J. Am. Chem. Soc. 2023, 145 (37), 20683– 20691, DOI: 10.1021/jacs.3c06738There is no corresponding record for this reference.
- 163Xu, T.; Lin, C.; Wang, C.; Brewe, D. L.; Ito, Y.; Lu, J. Synthesis of Supported Platinum Nanoparticles from Li-Pt Solid Solution. J. Am. Chem. Soc. 2010, 132 (7), 2151– 2153, DOI: 10.1021/ja909442cThere is no corresponding record for this reference.
- 164Xu, H.; Zhang, Z.; Liu, J.; Do-Thanh, C.-L.; Chen, H.; Xu, S.; Lin, Q.; Jiao, Y.; Wang, J.; Wang, Y.; Chen, Y.; Dai, S. Entropy-Stabilized Single-Atom Pd Catalysts via High-Entropy Fluorite Oxide Supports. Nat. Commun. 2020, 11 (1), 3908, DOI: 10.1038/s41467-020-17738-9There is no corresponding record for this reference.
- 165Fonseca, J.; Lu, J. Single-Atom Catalysts Designed and Prepared by the Atomic Layer Deposition Technique. ACS Catal. 2021, 11 (12), 7018– 7059, DOI: 10.1021/acscatal.1c01200There is no corresponding record for this reference.
- 166Knoll, M.; Ruska, E. Das Elektronenmikroskop. Z. Physik 1932, 78 (5), 318– 339, DOI: 10.1007/BF01342199There is no corresponding record for this reference.
- 167von Ardenne, M. Das Elektronen-Rastermikroskop. Z. Physik 1938, 109 (9), 553– 572, DOI: 10.1007/BF01341584There is no corresponding record for this reference.
- 168Scherzer, O. Über einige Fehler von Elektronenlinsen. Z. Physik 1936, 101 (9), 593– 603, DOI: 10.1007/BF01349606There is no corresponding record for this reference.
- 169Crewe, A. V.; Wall, J.; Langmore, J. Visibility of Single Atoms. Science 1970, 168 (3937), 1338– 1340, DOI: 10.1126/science.168.3937.1338There is no corresponding record for this reference.
- 170Pennycook, S. J.; Jesson, D. E. High-Resolution Z-Contrast Imaging of Crystals. Ultramicroscopy 1991, 37 (1), 14– 38, DOI: 10.1016/0304-3991(91)90004-PThere is no corresponding record for this reference.
- 171Tieu, P.; Yan, X.; Xu, M.; Christopher, P.; Pan, X. Directly Probing the Local Coordination, Charge State, and Stability of Single Atom Catalysts by Advanced Electron Microscopy: A Review. Small 2021, 17 (16), 2006482, DOI: 10.1002/smll.202006482There is no corresponding record for this reference.
- 172Sun, R.; Cao, X.; Ma, J.; Chen, H.-C.; Chen, C.; Peng, Q.; Li, Y. Regulation of Pd Single-Atom Coordination for Enhanced Photocatalytic Oxidation of Toluene to Benzaldehyde. Nat. Synth. 2025, 4 (8), 965– 975, DOI: 10.1038/s44160-025-00782-yThere is no corresponding record for this reference.
- 173Haider, M.; Uhlemann, S.; Schwan, E.; Rose, H.; Kabius, B.; Urban, K. Electron Microscopy Image Enhanced. Nature 1998, 392 (6678), 768– 769, DOI: 10.1038/33823There is no corresponding record for this reference.
- 174Batson, P. E.; Dellby, N.; Krivanek, O. L. Sub-Ångstrom Resolution Using Aberration Corrected Electron Optics. Nature 2002, 418 (6898), 617– 620, DOI: 10.1038/nature00972There is no corresponding record for this reference.
- 175Fricke, H. The K-Characteristic Absorption Frequencies for the Chemical Elements Magnesium to Chromium. Phys. Rev. 1920, 16 (3), 202– 215, DOI: 10.1103/PhysRev.16.202There is no corresponding record for this reference.
- 176Kronig, R. de L. Zur Theorie der Feinstruktur in den Röntgenabsorptionsspektren. Z. Physik 1931, 70 (5), 317– 323, DOI: 10.1007/BF01339581There is no corresponding record for this reference.
- 177Sayers, D. E. New Technique for Investigating Noncrystalline Structures: Fourier Analysis of the Extended X-Ray─Absorption Fine Structure. Phys. Rev. Lett. 1971, 27 (18), 1204– 1207, DOI: 10.1103/PhysRevLett.27.1204There is no corresponding record for this reference.
- 178Winick, H.; Bienenstock, A. Synchrotron Radiation Research. Annu. Rev. Nucl. Part. Sci. 1978, 28 (28), 33– 113, DOI: 10.1146/annurev.ns.28.120178.000341There is no corresponding record for this reference.
- 179Rehr, J. J.; Albers, R. C. Theoretical Approaches to X-Ray Absorption Fine Structure. Rev. Mod. Phys. 2000, 72 (3), 621– 654, DOI: 10.1103/RevModPhys.72.621There is no corresponding record for this reference.
- 180Bordiga, S.; Groppo, E.; Agostini, G.; van Bokhoven, J. A.; Lamberti, C. Reactivity of Surface Species in Heterogeneous Catalysts Probed by In Situ X-Ray Absorption Techniques. Chem. Rev. 2013, 113 (3), 1736– 1850, DOI: 10.1021/cr2000898There is no corresponding record for this reference.
- 181Mössbauer, R. L. Kernresonanzfluoreszenz von Gammastrahlung in Ir191. Z. Physik 1958, 151 (2), 124– 143, DOI: 10.1007/BF01344210There is no corresponding record for this reference.
- 182Greenwood, N. N.; Gibb, T. C. Mössbauer Spectroscopy. Springer Netherlands 1971, DOI: 10.1007/978-94-009-5697-1There is no corresponding record for this reference.
- 183Pound, R. V.; Rebka, G. A. Resonant Absorption of the 14.4-keV γ Ray from 0.10-Μsec Fe57. Phys. Rev. Lett. 1959, 3 (12), 554– 556, DOI: 10.1103/PhysRevLett.3.554There is no corresponding record for this reference.
- 184Zeng, Y. In Situ/Operando Mössbauer Spectroscopy for Probing Heterogeneous Catalysis. Chem. Catal. 2021, 1 (6), 1215– 1233, DOI: 10.1016/j.checat.2021.08.013There is no corresponding record for this reference.
- 185Li, J.; Sougrati, M. T.; Zitolo, A.; Ablett, J. M.; Oğuz, I. C.; Mineva, T.; Matanovic, I.; Atanassov, P.; Huang, Y.; Zenyuk, I.; Di Cicco, A.; Kumar, K.; Dubau, L.; Maillard, F.; Dražić, G.; Jaouen, F. Identification of Durable and Non-Durable FeNx Sites in Fe-N-C Materials for Proton Exchange Membrane Fuel Cells. Nat. Catal. 2021, 4 (1), 10– 19, DOI: 10.1038/s41929-020-00545-2There is no corresponding record for this reference.
- 186Egerton, R. F.; McLeod, R.; Wang, F.; Malac, M. Basic Questions Related to Electron-Induced Sputtering in the TEM. Ultramicroscopy 2010, 110 (8), 991– 997, DOI: 10.1016/j.ultramic.2009.11.003There is no corresponding record for this reference.
- 187Egerton, R. F. Control of Radiation Damage in the TEM. Ultramicroscopy 2013, 127, 100– 108, DOI: 10.1016/j.ultramic.2012.07.006There is no corresponding record for this reference.
- 188Finzel, J.; Sanroman Gutierrez, K. M.; Hoffman, A. S.; Resasco, J.; Christopher, P.; Bare, S. R. Limits of Detection for EXAFS Characterization of Heterogeneous Single-Atom Catalysts. ACS Catal. 2023, 13 (9), 6462– 6473, DOI: 10.1021/acscatal.3c01116There is no corresponding record for this reference.
- 189Egerton, R. f. Mechanisms of Radiation Damage in Beam-Sensitive Specimens, for TEM Accelerating Voltages between 10 and 300 kV. Micro. Res. Technol. 2012, 75 (11), 1550– 1556, DOI: 10.1002/jemt.22099There is no corresponding record for this reference.
- 190He, Q.; Li, A.; Yao, B.; Zhou, W.; Kiely, C. J. Springer Handbook of Advanced Catalyst Characterization. Springer Cham 2023, 409, DOI: 10.1007/978-3-031-07125-6_20There is no corresponding record for this reference.
- 191Egerton, R. F. Radiation Damage to Organic and Inorganic Specimens in the TEM. Micron 2019, 119, 72– 87, DOI: 10.1016/j.micron.2019.01.005There is no corresponding record for this reference.
- 192Zhang, T.; Chen, Z.; Walsh, A. G.; Li, Y.; Zhang, P. Single-Atom Catalysts Supported by Crystalline Porous Materials: Views from the Inside. Adv. Mater. 2020, 32 (44), 2002910, DOI: 10.1002/adma.202002910There is no corresponding record for this reference.
- 193Wang, A.; Li, J.; Zhang, T. Heterogeneous Single-Atom Catalysis. Nat. Rev. Chem. 2018, 2 (6), 65– 81, DOI: 10.1038/s41570-018-0010-1There is no corresponding record for this reference.
- 194Ogino, I. X-Ray Absorption Spectroscopy for Single-Atom Catalysts: Critical Importance and Persistent Challenges. Chin. J. Catal. 2017, 38 (9), 1481– 1488, DOI: 10.1016/S1872-2067(17)62880-8There is no corresponding record for this reference.
- 195Feng, K.; Zhang, H.; Gao, J.; Xu, J.; Dong, Y.; Kang, Z.; Zhong, J. Single Atoms or Not? The Limitation of EXAFS. Appl. Phys. Lett. 2020, 116 (19), 191903, DOI: 10.1063/5.0008748There is no corresponding record for this reference.
- 196Bazin, D. C.; Sayers, D. A.; Rehr, J. J. Comparison between X-Ray Absorption Spectroscopy, Anomalous Wide Angle X-Ray Scattering, Anomalous Small Angle X-Ray Scattering, and Diffraction Anomalous Fine Structure Techniques Applied to Nanometer-Scale Metallic Clusters. J. Phys. Chem. B 1997, 101 (51), 11040– 11050, DOI: 10.1021/jp9721311There is no corresponding record for this reference.
- 197Liu, L.; Corma, A. Identification of the Active Sites in Supported Subnanometric Metal Catalysts. Nat. Catal. 2021, 4 (6), 453– 456, DOI: 10.1038/s41929-021-00632-yThere is no corresponding record for this reference.
- 198Liu, A.; Liu, L.; Cao, Y.; Wang, J.; Si, R.; Gao, F.; Dong, L. Controlling Dynamic Structural Transformation of Atomically Dispersed CuOx Species and Influence on Their Catalytic Performances. ACS Catal. 2019, 9 (11), 9840– 9851, DOI: 10.1021/acscatal.9b02773There is no corresponding record for this reference.
- 199Gütlich, P.; Bill, E.; Trautwein, A. X. Mössbauer Spectroscopy and Transition Metal Chemistry. Springer Berlin 2011, DOI: 10.1007/978-3-540-88428-6There is no corresponding record for this reference.
- 200Chen, C.; Li, J.; Tan, X.; Zhang, Y.; Li, Y.; He, C.; Xu, Z.; Zhang, C.; Chen, C. Harnessing Single-Atom Catalysts for CO2 Electroreduction: A Review of Recent Advances. EES Catal. 2024, 2 (1), 71– 93, DOI: 10.1039/D3EY00150DThere is no corresponding record for this reference.
- 201Ren, C.; Cui, Y.; Li, Q.; Ling, C.; Wang, J. Single-Atom Saturation: A Fundamental Principle for Single-Atom-Site Catalyst Design. J. Am. Chem. Soc. 2025, 147 (16), 13610– 13617, DOI: 10.1021/jacs.5c00643There is no corresponding record for this reference.
- 202Liu, X.; Ding, G.; Guo, X.; Wang, X.; Wang, Z.; Chen, Z.; Xiao, Y.; Shuai, L.; Liao, G. Synergizing Donor-Acceptor Ni-MOF with Lignin Proton Relay for Efficient and Selective CO2-to-CO Photoreduction. Adv. Mater. 2026, 38, e20384 DOI: 10.1002/adma.202520384There is no corresponding record for this reference.
- 203Vijay, S.; Ju, W.; Brückner, S.; Tsang, S.-C.; Strasser, P.; Chan, K. Unified Mechanistic Understanding of CO2 Reduction to CO on Transition Metal and Single Atom Catalysts. Nat. Catal. 2021, 4 (12), 1024– 1031, DOI: 10.1038/s41929-021-00705-yThere is no corresponding record for this reference.
- 204Kortlever, R.; Shen, J.; Schouten, K. J. P.; Calle-Vallejo, F.; Koper, M. T. M. Catalysts and Reaction Pathways for the Electrochemical Reduction of Carbon Dioxide. J. Phys. Chem. Lett. 2015, 6 (20), 4073– 4082, DOI: 10.1021/acs.jpclett.5b01559There is no corresponding record for this reference.
- 205Göttle, A. J.; Koper, M. T. M. Determinant Role of Electrogenerated Reactive Nucleophilic Species on Selectivity during Reduction of CO2 Catalyzed by Metalloporphyrins. J. Am. Chem. Soc. 2018, 140 (14), 4826– 4834, DOI: 10.1021/jacs.7b11267There is no corresponding record for this reference.
- 206Fan, L.; Li, F.; Liu, T.; Huang, J. E.; Miao, R. K.; Yan, Y.; Feng, S.; Tai, C.-W.; Hung, S.-F.; Tsai, H.-J.; Chen, M.-C.; Bai, Y.; Kim, D.; Park, S.; Papangelakis, P.; Wu, C.; Shayesteh Zeraati, A.; Dorakhan, R.; Sun, L.; Sinton, D.; Sargent, E. Atomic-Level Cu Active Sites Enable Energy-Efficient CO2 Electroreduction to Multicarbon Products in Strong Acid. Nat. Synth. 2025, 4 (2), 262– 270, DOI: 10.1038/s44160-024-00689-0There is no corresponding record for this reference.
- 207Zhao, Y.; Shi, Z.; Li, F.; Jia, C.; Sun, Q.; Su, Z.; Zhao, C. Deciphering Mesopore-Augmented CO2 Electroreduction over Atomically Dispersed Fe-N-Doped Carbon Catalysts. ACS Catal. 2024, 14 (6), 3926– 3932, DOI: 10.1021/acscatal.3c05144There is no corresponding record for this reference.
- 208Zhang, T.; Han, X.; Liu, H.; Biset-Peiró, M.; Li, J.; Zhang, X.; Tang, P.; Yang, B.; Zheng, L.; Morante, J. R.; Arbiol, J. Site-Specific Axial Oxygen Coordinated FeN4 Active Sites for Highly Selective Electroreduction of Carbon Dioxide. Adv. Funct. Mater. 2022, 32 (18), 2111446, DOI: 10.1002/adfm.202111446There is no corresponding record for this reference.
- 209Chen, Z.; Zhang, X.; Liu, W.; Jiao, M.; Mou, K.; Zhang, X.; Liu, L. Amination Strategy to Boost the CO2 Electroreduction Current Density of M-N/C Single-Atom Catalysts to the Industrial Application Level. Energy Environ. Sci. 2021, 14 (4), 2349– 2356, DOI: 10.1039/D0EE04052EThere is no corresponding record for this reference.
- 210Tierney, H. L.; Baber, A. E.; Kitchin, J. R.; Sykes, E. C. H. Hydrogen Dissociation and Spillover on Individual Isolated Palladium Atoms. Phys. Rev. Lett. 2009, 103 (24), 246102, DOI: 10.1103/PhysRevLett.103.246102There is no corresponding record for this reference.
- 211Sun, L.; Wang, Y.; Guan, N.; Li, L. Methane Activation and Utilization: Current Status and Future Challenges. Energy Technol. 2020, 8 (8), 1900826, DOI: 10.1002/ente.201900826There is no corresponding record for this reference.
- 212Shen, W.; Gao, X.; Liu, Q.; Li, P.; Huang, R.; Tan, Y.; Wang, Z.; Zhang, Y.; Zhao, F.; Wang, X.; Ji, S.; Zheng, X.; Zhang, Y.; Wu, Y. Self-Healing Cu Single-Atom Catalyst for High-Performance Electrocatalytic CO2 Methanation. Nat. Commun. 2025, 16 (1), 7943, DOI: 10.1038/s41467-025-63274-9There is no corresponding record for this reference.
- 213Han, L.; Song, S.; Liu, M.; Yao, S.; Liang, Z.; Cheng, H.; Ren, Z.; Liu, W.; Lin, R.; Qi, G.; Liu, X.; Wu, Q.; Luo, J.; Xin, H. L. Stable and Efficient Single-Atom Zn Catalyst for CO2 Reduction to CH4. J. Am. Chem. Soc. 2020, 142 (29), 12563– 12567, DOI: 10.1021/jacs.9b12111There is no corresponding record for this reference.
- 214Chen, S.; Zhang, Z.; Jiang, W.; Zhang, S.; Zhu, J.; Wang, L.; Ou, H.; Zaman, S.; Tan, L.; Zhu, P.; Zhang, E.; Jiang, P.; Su, Y.; Wang, D.; Li, Y. Engineering Water Molecules Activation Center on Multisite Electrocatalysts for Enhanced CO2 Methanation. J. Am. Chem. Soc. 2022, 144 (28), 12807– 12815, DOI: 10.1021/jacs.2c03875There is no corresponding record for this reference.
- 215Hung, S.-F.; Xu, A.; Wang, X.; Li, F.; Hsu, S.-H.; Li, Y.; Wicks, J.; Cervantes, E. G.; Rasouli, A. S.; Li, Y. C.; Luo, M.; Nam, D.-H.; Wang, N.; Peng, T.; Yan, Y.; Lee, G.; Sargent, E. H. A Metal-Supported Single-Atom Catalytic Site Enables Carbon Dioxide Hydrogenation. Nat. Commun. 2022, 13 (1), 819, DOI: 10.1038/s41467-022-28456-9There is no corresponding record for this reference.
- 216Osella, S.; Goddard, W. A., III CO2 Reduction to Methane and Ethylene on a Single-Atom Catalyst: A Grand Canonical Quantum Mechanics Study. J. Am. Chem. Soc. 2023, 145 (39), 21319– 21329, DOI: 10.1021/jacs.3c05650There is no corresponding record for this reference.
- 217He, H.; Lee, J.; Zong, Z.; Lim, K. M.; Ryu, J.; Oh, J.; Kim, J.; Sessler, J. L.; Ke, X.-S. Precise Internal Postsynthetic Oxygen-Doping of Metallonanographenes. Prec. Chem. 2025, 3 (8), 456– 462, DOI: 10.1021/prechem.5c00035There is no corresponding record for this reference.
- 218Xie, C.; Chen, W.; Wang, Y.; Yang, Y.; Wang, S. Dynamic Evolution Processes in Electrocatalysis: Structure Evolution, Characterization and Regulation. Chem. Soc. Rev. 2024, 53 (22), 10852– 10877, DOI: 10.1039/D3CS00756AThere is no corresponding record for this reference.
- 219He, H.; Jagvaral, Y. Electrochemical Reduction of CO2 on Graphene Supported Transition Metals-towards Single Atom Catalysts. Phys. Chem. Chem. Phys. 2017, 19 (18), 11436– 11446, DOI: 10.1039/C7CP00915AThere is no corresponding record for this reference.
- 220Zhang, D.; Ren, P.; Liu, W.; Li, Y.; Salli, S.; Han, F.; Qiao, W.; Liu, Y.; Fan, Y.; Cui, Y.; Shen, Y.; Richards, E.; Wen, X.; Rummeli, M. H.; Li, Y.; Besenbacher, F.; Niemantsverdriet, H.; Lim, T.; Su, R. Photocatalytic Abstraction of Hydrogen Atoms from Water Using Hydroxylated Graphitic Carbon Nitride for Hydrogenative Coupling Reactions. Angew. Chem., Int. Ed. 2022, 61 (24), e202204256 DOI: 10.1002/anie.202204256There is no corresponding record for this reference.
- 221Yang, T.; Yang, F.; Wang, T.; Zhang, W.; Deng, F.; Zhong, C.; Peng, Q.; Liu, C.; Chen, X.; Zhang, Z.; Li, Y.; Feng, Y.; Li, X.; Wang, K.; Zhang, C.; Zhou, Y.; Lu, X.; Zhu, W.; Wei, Y.; Xie, L.; Huang, W. Gridized Nanopolymer Catalysis with Atomically Dispersed Iron Achieves the Nearly 100% Selective Electrosynthesis of Methanol From CO2. Adv. Mater. 2026, 38, e11233 DOI: 10.1002/adma.202511233There is no corresponding record for this reference.
- 222Ren, X.; Zhao, J.; Li, X.; Shao, J.; Pan, B.; Salamé, A.; Boutin, E.; Groizard, T.; Wang, S.; Ding, J.; Zhang, X.; Huang, W.-Y.; Zeng, W.-J.; Liu, C.; Li, Y.; Hung, S.-F.; Huang, Y.; Robert, M.; Liu, B. In-Situ Spectroscopic Probe of the Intrinsic Structure Feature of Single-Atom Center in Electrochemical CO/CO2 Reduction to Methanol. Nat. Commun. 2023, 14 (1), 3401, DOI: 10.1038/s41467-023-39153-6There is no corresponding record for this reference.
- 223Chatterjee, S.; Dutta, I.; Lum, Y.; Lai, Z.; Huang, K.-W. Enabling Storage and Utilization of Low-Carbon Electricity: Power to Formic Acid. Energy Environ. Sci. 2021, 14 (3), 1194– 1246, DOI: 10.1039/D0EE03011BThere is no corresponding record for this reference.
- 224Shang, H.; Wang, T.; Pei, J.; Jiang, Z.; Zhou, D.; Wang, Y.; Li, H.; Dong, J.; Zhuang, Z.; Chen, W.; Wang, D.; Zhang, J.; Li, Y. Design of a Single-Atom Indiumδ+-N4 Interface for Efficient Electroreduction of CO2 to Formate. Angew. Chem., Int. Ed. 2020, 59 (50), 22465– 22469, DOI: 10.1002/anie.202010903There is no corresponding record for this reference.
- 225Fang, C.; Huang, L.; Gao, W.; Jiang, X.; Liu, H.; Hu, R.; Li, X.; Yu, J.; Zhou, W. Oxygen-Pinned Ag1In Single-Atom Alloy for Efficient Electroreduction CO2 to Formate. Adv. Energy Mater. 2024, 14 (27), 2400813, DOI: 10.1002/aenm.202400813There is no corresponding record for this reference.
- 226Shao, L.-Y.; Wang, Y.; Meng, J.; Yue, J.-N.; Zhang, A.-M.; Yuan, W.; Li, S.-N.; Zhai, Q.-G. Exclusive Bridged *OCHO Pathway Activated by Phenol-Quinone Conversion on the Surface of Cu(OH)2 for Highly Selective Formate Production in CO2 Electroreduction. Adv. Funct. Mater. 2026, 36, e14222 DOI: 10.1002/adfm.202514222There is no corresponding record for this reference.
- 227Jiang, Z.; Liu, X.; Liu, X.-Z.; Huang, S.; Liu, Y.; Yao, Z.-C.; Zhang, Y.; Zhang, Q.-H.; Gu, L.; Zheng, L.-R.; Li, L.; Zhang, J.; Fan, Y.; Tang, T.; Zhuang, Z.; Hu, J.-S. Interfacial Assembly of Binary Atomic Metal-Nx Sites for High-Performance Energy Devices. Nat. Commun. 2023, 14 (1), 1822, DOI: 10.1038/s41467-023-37529-2There is no corresponding record for this reference.
- 228Cui, Y.; Ren, C.; Li, Q.; Ling, C.; Wang, J. Hybridization State Transition under Working Conditions: Activity Origin of Single-Atom Catalysts. J. Am. Chem. Soc. 2024, 146 (22), 15640– 15647, DOI: 10.1021/jacs.4c05630There is no corresponding record for this reference.
- 229Wang, F.; Su, Q.; Zhang, X.; Chen, A. Spin-State Regulation of Single-Atom Catalysts: From Quantum Mechanisms to Multidimensional Dynamic Regulation Strategies. Adv. Funct. Mater. 2026, 36, e18600 DOI: 10.1002/adfm.202518600There is no corresponding record for this reference.
- 230Zeng, Y.; Zhao, J.; Wang, S.; Wang, W.; Lu, Y.-R.; Xi, S.; Xu, W.; Yoda, Y.; Masuda, R.; Li, X.; Huang, Y.; Liu, B.; Zhang, T. Unraveling the Dynamic Low-Spin State Evolution of Single-Fe-Atom Sites for Efficient CO2 Electroreduction. J. Am. Chem. Soc. 2025, 147 (46), 42539– 42548, DOI: 10.1021/jacs.5c13466There is no corresponding record for this reference.
- 231Vaz, C. a. F.; van der Laan, G.; Cavill, S. A.; Dürr, H. A.; Fraile Rodríguez, A.; Kronast, F.; Kuch, W.; Sainctavit, P.; Schütz, G.; Wende, H.; Weschke, E.; Wilhelm, F. X-Ray Magnetic Circular Dichroism. Nat. Rev. Methods Primers 2025, 5 (1), 27, DOI: 10.1038/s43586-025-00397-9There is no corresponding record for this reference.
- 232Bai, R.; Yao, Y.; Lin, Q.; Wu, L.; Li, Z.; Wang, H.; Ma, M.; Mu, D.; Hu, L.; Yang, H.; Li, W.; Zhu, S.; Wu, X.; Rui, X.; Yu, Y. Preferable Single-Atom Catalysts Enabled by Natural Language Processing for High Energy Density Na-S Batteries. Nat. Commun. 2025, 16 (1), 5827, DOI: 10.1038/s41467-025-60931-xThere is no corresponding record for this reference.
- 233Szymanski, N. J.; Rendy, B.; Fei, Y.; Kumar, R. E.; He, T.; Milsted, D.; McDermott, M. J.; Gallant, M.; Cubuk, E. D.; Merchant, A.; Kim, H.; Jain, A.; Bartel, C. J.; Persson, K.; Zeng, Y.; Ceder, G. An Autonomous Laboratory for the Accelerated Synthesis of Novel Materials. Nature 2023, 624 (7990), 86– 91, DOI: 10.1038/s41586-023-06734-wThere is no corresponding record for this reference.


