
Chemically Recyclable Thermoplastic Elastomers: Preparation, Properties, and On-Demand DepolymerizationClick to copy article linkArticle link copied!
- Yingying LiuYingying LiuState Key Laboratory of Advanced Optical Polymer and Manufacturing Technology, College of Chemical Engineering, Qingdao University of Science and Technology, Qingdao 266042, ChinaMore by Yingying Liu
- Yong Shen*Yong Shen*Email: [email protected]State Key Laboratory of Advanced Optical Polymer and Manufacturing Technology, College of Chemical Engineering, Qingdao University of Science and Technology, Qingdao 266042, ChinaMore by Yong Shen
Abstract
Great advancements have been achieved in the past decade for chemically recyclable polymers that can be selectively depolymerized back to their monomers to address the end-of-life issue of plastics. Nevertheless, the technologically important thermoplastic elastomers (TPEs) with on-demand depolymerization property have received less attention largely due to their intrinsically complicated multiblock architectures and challenges involved in the selective chemical recycling of different monomers. This review highlights the recent achievements of chemically recyclable TPEs by focusing on their preparation, mechanical properties, and on-demand depolymerization and recycling. A variety of TPEs are introduced according to their backbone, including copolyesters, polyurethanes, and polyolefins. The dynamically cross-linked elastomers with chemical recyclability are also briefly discussed. Finally, the current challenges and possible future directions for chemically recyclable TPEs are also addressed.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of Precision Chemistry special issue “Precision Chemistry for Polymer Recycling”.
1. Introduction
| TPE | Mna (kg/mol) | fhard (%) | Tgd (°C) | Tmd (°C) | E (MPa)e | σb (MPa)e | εb (%)e | elastic recovery (%)f | refs |
|---|---|---|---|---|---|---|---|---|---|
| PLLA-b-PδCL-b-PLLA | 90.9 | 0.33 | –35, 57 | 159 | 15.8 ± 3.1 | 20.4 ± 1.0 | 956 ± 105 | 91.8 | (33,34) |
| PδVL-b-PαMeVL-b-PδVL | 104.6 | 0.41 | –51 | 60 | 14.2 ± 0.9 | 14.9 ± 0.5 | 1061 ± 11 | 96 | (35) |
| PδVL-b-PβMVL-b-PδVL | 92.4 | 0.56 | –55 | 42 | n.r.i | 39.4 ± 0.4 | 1986 ± 36 | 96.3 | (36) |
| PPDO-b-PβMVL-b-PPDO | 90.0 | 0.24 | –50.3, −17.6 | 80.4 | n.r. | 21.1 ± 0.5 | 1054 ± 45 | 93.5 | (37) |
| P(5C-TMC)-b-P(MePr-TMC) -b-P(5C-TMC) | 53.8 | 0.32 | n.r. | n.r. | n.r. | 11.7 ± 1.3 | 472 ± 11 | 87.3 | (38) |
| PLLA-b-P(THF-co-CHO)-b-PLLA | 109 | 0.58 | –78, 64 | 178 | 0.21 ± 0.01 | 16.0 ± 3.0 | 277 ± 36 | n.r. | (39) |
| P(CL2000-grad-SHPL500) | 121 | n.r. | n.r. | 49,265 | 157 ± 2.8 | 58.8 ± 4.0 | 1959 ± 53 | 92 | (40) |
| P[CL48-stat-(4Ph-BL)52] | 383 | n.r. | n.r. | n.r. | 31.3 ± 1.7 | 34.5 ± 3.6 | 812 ± 42 | 99.3g | (41) |
| PβMVL-TPU | 102.2 | 0.40b | –33 | n.r. | 7.2 ± 0.2 | 37 ± 3 | 1000 ± 20 | 98.8h | (42) |
| PδCL-TPU | 99.4 | 0.32b | 9.8 | n.r. | 6.0 ± 0.3 | 60.4 ± 2.6 | 899 ± 6 | 97 | (43,44) |
| PγBL-TPU | 70.1 | 0.15c | –23 | n.r. | n.r. | 18.3 ± 1.5 | 2298 ± 75 | 95.9 | (45) |
| PE40 | 82.4 | 0.4c | –50.6 | 109 | 15 ± 1 | 12.0 ± 0.7 | 1030 ± 30 | n.r. | (46) |
| rOBC7.7 (7.7 mol % of 1-octene) | 129.4 | n.r. | –44.7 | 95.1 | n.r. | 24.8 ± 1.7 | 1950 ± 83 | n.r. | (47) |
| polyolefin-like elastomer P4 | 158.3 | n.r. | –21 | 41 | 1.9 ± 0.2 | 31.5 ± 3.1 | 1495 ± 286 | 98 | (48) |
| PE(v)-b-PTMC-b-PE(v) | 185 | 0.13 | –14, 115 | n.r. | 7.1 ± 0.7 | 25.9 ± 1.1 | 822 ± 42 | 96.9 | (49) |
The Mn values of TPEs were measured by size exclusion chromatography (SEC) except for PPDO-b-PβMVL-b-PPDO, which was measured by 1H NMR.
Weight percentage of the urethane-rich hard segments in the polyurethane.
Molar percentage of the urethane-rich hard segments in the polyurethane.
Tg and Tm were measured by differential scanning calorimetry (DSC).
E, σb, and εb represents Young’s modulus, ultimate tensile strength, and elongation at break, respectively.
Calculated from 10 cycles at 100% strain unless otherwise noted.
Calculated from 20 cycles at 100% strain.
Calculated from 20 cycles at 50% strain.
These data were not reported in the references, n.r. = not reported.
2. Chemically Recyclable Block Copolyester-Based TPEs
Figure 1
Figure 1. Illustration of preparation and chemical recycling of PLLA-b-PδCL-b-PLLA triblock elastomer. (34)
Figure 2
Figure 2. Illustration of the sequential ROP of βMVL and δVL toward PδVL-b-PβMVL-b-PδVL triblock copolymers and their chemical recycling. Adapted from ref (36). Copyright 2025 Wiley-VCH Verlag.
Figure 3
Figure 3. Solvent-dependent sequence-controlled copolymerization of 4Ph-BL with ε-CL to produce block or statistical copolyesters and their chemical recycling. (41)
3. Chemically Recyclable Thermoplastic Polyurethane Elastomers
Figure 4
Figure 4. Illustration of the preparation of chemically recyclable polyurethanes from biosourced alkyl-δ-lactones as well as their chemical recycling to recover pristine monomers. Reproduced from ref (44). Copyright 2025 Elsevier.
4. Chemically Recyclable Polyolefin-Like Elastomers
Figure 5
Figure 5. (a) Synthesis of hard and soft building blocks. (b) Preparation and closed-loop recycling of polyolefin-like multiblock elastomers. (46)
Figure 6
Figure 6. (a) Synthesis of homopolymer-based polyolefin-like elastomers and (b) their chemical recycling through three cycles. (48)
5. Chemically Recyclable Dynamically Cross-Linked Elastomers
Figure 7
Figure 7. (a) Preparation and (b) chemical recycling of PE(v)-b-PTMC-b-PE(v) and P3Zn triblock elastomers. (49)
6. Conclusion and Perspective
| (1) | It is attractive to continuously develop TPEs with intrinsic chemical recyclability to create an ideal closed-loop life cycle. Most of the above-reported chemically recyclable TPEs only achieve the recovery of partial components, especially for the thermoplastic polyurethane elastomers. Even for the cases of fully recyclable block copolyester-based TPEs, tedious, costly, and time-consuming separation and purification of monomers by column chromatography are required to close the “material–monomer–material” loop. Utilizing monomers with big differences in solubility or boiling point may facilitate their separation by extraction, recrystallization, or distillation. Another promising strategy is to achieve the selective recovery of different monomers by the selective depolymerization of soft and hard blocks. As an alternative promising strategy, switchable polymerization presents an opportunity to prepare TPEs directly from the monomer mixture so that monomer separation is not necessary. | ||||
| (2) | More efforts should be devoted to developing a more efficient chemical recycling strategy for polyolefin-like elastomers and dynamically cross-linked elastomers. Although polyolefin-like elastomers exhibit excellent thermal stability and mechanical properties comparable to those of commercial POE or OBC elastomers, the current chemical recycling strategy to recover polyolefin oligomers suffers from a large excess use of solvent, harmful reagents (such as strong acids or bases), or high-pressure hydrogen. The same problems exist for the chemical recycling of dynamically cross-linked elastomers. There is an urgent need to develop a milder, greener, and solvent-free chemical recycling strategy to promote their practical applications. | ||||
| (3) | It is crucial to investigate the chemical recycling of these TPEs in the real world. The current studies in the laboratory use single or pure TPEs for their chemical recycling, which is not consistent with reality. Future research on the chemical recycling of these materials in a mixed plastics stream is necessary to evaluate their economic and ecological accessibility. | ||||
| (4) | Last but perhaps the most important thing to keep in mind, it is necessary to prepare TPEs with comparable properties to their commercial analogues before considering their recycling. Otherwise, “wastes” rather than useful materials are produced at the beginning. In addition to the frequently investigated thermal stability, mechanical strength, and elastic recovery, fatigue resistance, high- and low-temperature resistance, chemical stability, creep and stress relaxation, weather resistance, processability, and production cost should be evaluated. The comprehensive evaluation and comparison provide a data foundation for the potential application of chemical recyclable TPEs in the future. | ||||
Acknowledgments
The authors appreciate the financial support by the National Natural Science Foundation of China (Nos. 52322304 and 22475117), the Natural Science Foundation of Qingdao (24-4-4-zrjj-17-jch), and the Postdoctoral Innovation Project of Shandong Province (SDCX-ZG-202400254).
| Chemical recycling | A process that polymer is depolymerized to recover pristine monomers or converted to valuable chemical raw materials. |
| Thermoplastic elastomer (TPE) | A type of polymer material that combines the physical properties of traditional cross-linked vulcanized rubber with the processing properties of a thermoplastic. |
| Closed-loop recyclable TPE | A type of TPE that can be depolymerized back to their pristine monomers under controlled conditions at the end of their service life. The recovered monomers can be repolymerized to produce virgin-quality TPE, thus establishing a “polymer–monomer–polymer” closed-loop life cycle. |
| Dynamic cross-linked elastomers | A type of elastomer material that is cross-linked by dynamic covalent bonds or supramolecular interactions. The dynamic cross-linked elastomers combine the chemical resistance and mechanical strength of thermosets with the reprocessability of thermoplastics. |
| Volume fraction of hard block (fhard) | A critical parameter describing the composition of a block copolymer (such as TPE), which can be calculated according to fhard = ρsoftblock/[ρhardblock (1/ωhardblock – 1) + ρsoftblock], where ω is the weight percent of hard block and ρ represents the density at room temperature. |
References
This article references 83 other publications.
- 1Seymour, R. B.; Kauffman, G. B. Elastomers: III. Thermoplastic elastomers. J. Chem. Educ. 1992, 69 (12), 967 DOI: 10.1021/ed069p967Google ScholarThere is no corresponding record for this reference.
- 2Wang, W.; Lu, W.; Goodwin, A.; Wang, H.; Yin, P.; Kang, N.; Hong, K.; Mays, J. W. Recent advances in thermoplastic elastomers from living polymerizations: Macromolecular architectures and supramolecular chemistry. Prog. Polym. Sci. 2019, 95, 1– 31, DOI: 10.1016/j.progpolymsci.2019.04.002Google ScholarThere is no corresponding record for this reference.
- 3Guo, Z.; Lu, X.; Wang, X.; Li, X.; Li, J.; Sun, J. Engineering of Chain Rigidity and Hydrogen Bond Cross-Linking toward Ultra-Strong, Healable, Recyclable, and Water-Resistant Elastomers. Adv. Mater. 2023, 35 (21), 2300286 DOI: 10.1002/adma.202300286Google ScholarThere is no corresponding record for this reference.
- 4Zhu, L. Q.; X, Y. K.; Wang, W. J.; Liu, P. W.; Wang, Q. Y.; Jie, S. Y.; Hu, J. J.; Yao, Z.; Li, B. G. State-of-the-art and research progress of block-type thermoplastic elastomers. Acta Polym. Sin 2025, 56 (9), 1463– 1479Google ScholarThere is no corresponding record for this reference.
- 5Thermoplastic Elastomer Market Size, Share & COVID-19 Impact Analysis, By Type, By Application, and Regional Forecast, 2024–2032. Fortune Business Insights. https://www.fortunebusinessinsights.com/zh/thermoplastic-elastomer-tpe-market-104515. (accessed Oct. 6, 2025).Google ScholarThere is no corresponding record for this reference.
- 6Bates, F. S.; Hillmyer, M. A.; Lodge, T. P.; Bates, C. M.; Delaney, K. T.; Fredrickson, G. H. Multiblock Polymers: Panacea or Pandora’s Box?. Science 2012, 336 (6080), 434– 440, DOI: 10.1126/science.1215368Google ScholarThere is no corresponding record for this reference.
- 7Sinturel, C.; Bates, F. S.; Hillmyer, M. A. High χ–Low N Block Polymers: How Far Can We Go?. ACS Macro Lett. 2015, 4 (9), 1044– 1050, DOI: 10.1021/acsmacrolett.5b00472Google ScholarThere is no corresponding record for this reference.
- 8He, W.-N.; Xu, J.-T. Crystallization assisted self-assembly of semicrystalline block copolymers. Prog. Polym. Sci. 2012, 37 (10), 1350– 1400, DOI: 10.1016/j.progpolymsci.2012.05.002Google ScholarThere is no corresponding record for this reference.
- 9Lynd, N. A.; Meuler, A. J.; Hillmyer, M. A. Polydispersity and block copolymer self-assembly. Prog. Polym. Sci. 2008, 33 (9), 875– 893, DOI: 10.1016/j.progpolymsci.2008.07.003Google ScholarThere is no corresponding record for this reference.
- 10Tang, J.; Gong, S.; Zhao, B.; Chen, K.; Deng, J.; Pan, K. Biobased Thermoplastic Polyamide Elastomers: Preparation, Properties, and Applications. Ind. Eng. Chem. Res. 2024, 63 (8), 3465– 3482, DOI: 10.1021/acs.iecr.3c04554Google ScholarThere is no corresponding record for this reference.
- 11Wang, L.; Guo, L.; Zhang, K.; Xia, Y.; Hao, J.; Wang, X. Development of Tough Thermoplastic Elastomers by Leveraging Rigid–Flexible Supramolecular Segment Interplays. Angew. Chem., Int. Ed. 2023, 62 (29), e202301762 DOI: 10.1002/anie.202301762Google ScholarThere is no corresponding record for this reference.
- 12Gonçalves, R.; Serra, J.; Reizabal, A.; Correia, D. M.; Fernandes, L. C.; Brito-Pereira, R.; Lizundia, E.; Costa, C. M.; Lanceros-Méndez, S. Biobased polymers for advanced applications: Towards a sustainable future. Prog. Polym. Sci. 2025, 162, 101934 DOI: 10.1016/j.progpolymsci.2025.101934Google ScholarThere is no corresponding record for this reference.
- 13Wang, Z.; Yuan, L.; Tang, C. Sustainable Elastomers from Renewable Biomass. Acc. Chem. Res. 2017, 50 (7), 1762– 1773, DOI: 10.1021/acs.accounts.7b00209Google ScholarThere is no corresponding record for this reference.
- 14Liu, Q.; Lou, P.; Sun, Z.; Li, D.; Ji, H.; Xu, Z.; Li, L.; Xue, J.; Wang, R.; Wang, Z.; Zhang, L. Bio-Based Elastomers: Design, Properties, and Biomedical Applications. Adv. Mater. 2025, 37 (22), 2417193 DOI: 10.1002/adma.202417193Google ScholarThere is no corresponding record for this reference.
- 15Zhang, Q.; Song, M.; Xu, Y.; Wang, W.; Wang, Z.; Zhang, L. Bio-based polyesters: Recent progress and future prospects. Prog. Polym. Sci. 2021, 120, 101430 DOI: 10.1016/j.progpolymsci.2021.101430Google ScholarThere is no corresponding record for this reference.
- 16Liu, Y.; Zhang, M.; Shen, Y.; Li, Z. Biodegradable and Chemically Recyclable Thermoplastic Elastomers Prepared by Ring-Opening Polymerization of Cyclic Monomers. Polym. Sci. Technol. 2025, 1 (6), 529– 540, DOI: 10.1021/polymscitech.5c00050Google ScholarThere is no corresponding record for this reference.
- 17Jeon, J. H.; Jung, J. H.; Choi, C. Toward a greener future: Exploring sustainable thermoplastic elastomers. J. Polym. Sci. 2024, 62 (4), 662– 678, DOI: 10.1002/pol.20230293Google ScholarThere is no corresponding record for this reference.
- 18Sha, Y.; Sun, W.; Ding, S.; Ye, P.; Zhang, Y.; Jia, P. Performance-Guided Design of Chemically Recyclable Polymeric Materials: A Case Study on Thermoplastic Elastomers. ACS Polym. Au 2025, 5 (5), 432– 444, DOI: 10.1021/acspolymersau.5c00033Google ScholarThere is no corresponding record for this reference.
- 19Zhou, D.-L.; Lin, X.; Guo, Q.-Y.; Han, D.; Fu, Q. Mechanically Accelerated Chemical Deconstruction for Nondeteriorative Recycling of Industrial Unsaturated Thermosets and Rubbers. Macromolecules 2025, 58 (20), 11361– 11369, DOI: 10.1021/acs.macromol.5c01682Google ScholarThere is no corresponding record for this reference.
- 20Plummer, C. M.; Li, L.; Chen, Y. Ring-Opening Polymerization for the Goal of Chemically Recyclable Polymers. Macromolecules 2023, 56 (3), 731– 750, DOI: 10.1021/acs.macromol.2c01694Google ScholarThere is no corresponding record for this reference.
- 21Shi, C.; Quinn, E. C.; Diment, W. T.; Chen, E. Y. X. Recyclable and (Bio)degradable Polyesters in a Circular Plastics Economy. Chem. Rev. 2024, 124 (7), 4393– 4478, DOI: 10.1021/acs.chemrev.3c00848Google ScholarThere is no corresponding record for this reference.
- 22Tang, X.; Chen, E. Y. X. Toward Infinitely Recyclable Plastics Derived from Renewable Cyclic Esters. Chem 2019, 5 (2), 284– 312, DOI: 10.1016/j.chempr.2018.10.011Google ScholarThere is no corresponding record for this reference.
- 23Li, Z.; Shen, Y.; Li, Z. Ring-Opening Polymerization of Lactones to Prepare Closed-Loop Recyclable Polyesters. Macromolecules 2024, 57 (5), 1919– 1940, DOI: 10.1021/acs.macromol.3c01912Google ScholarThere is no corresponding record for this reference.
- 24Ibrahim, T.; Ritacco, A.; Nalley, D.; Emon, O. F.; Liang, Y.; Sun, H. Chemical recycling of polyolefins via ring-closing metathesis depolymerization. Chem. Commun. 2024, 60 (11), 1361– 1371, DOI: 10.1039/D3CC05612KGoogle ScholarThere is no corresponding record for this reference.
- 25Chen, H.; Shi, Z.; Hsu, T.-G.; Wang, J. Overcoming the Low Driving Force in Forming Depolymerizable Polymers through Monomer Isomerization. Angew. Chem., Int. Ed. 2021, 60 (48), 25493– 25498, DOI: 10.1002/anie.202111181Google ScholarThere is no corresponding record for this reference.
- 26Sathe, D.; Zhou, J.; Chen, H.; Su, H.-W.; Xie, W.; Hsu, T.-G.; Schrage, B. R.; Smith, T.; Ziegler, C. J.; Wang, J. Olefin metathesis-based chemically recyclable polymers enabled by fused-ring monomers. Nat. Chem. 2021, 13 (8), 743– 750, DOI: 10.1038/s41557-021-00748-5Google ScholarThere is no corresponding record for this reference.
- 27Cederholm, L.; Olsén, P.; Hakkarainen, M.; Odelius, K. Design for Recycling: Polyester- and Polycarbonate-Based A–B–A Block Copolymers and Their Recyclability Back to Monomers. Macromolecules 2023, 56 (10), 3641– 3649, DOI: 10.1021/acs.macromol.3c00274Google ScholarThere is no corresponding record for this reference.
- 28Niu, H.; Wang, L.; Zhang, Z.; Liu, Y.; Shen, Y.; Li, Z. Ring-Opening Copolymerization of Biorenewable δ-Caprolactone with -Hexahydro-(4,5)-benzofuranone toward Closed-Loop Recyclable Copolyesters and Their Application as Pressure-Sensitive Adhesives. Chin. J. Chem. 2024, 42 (17), 2035– 2042, DOI: 10.1002/cjoc.202400244Google ScholarThere is no corresponding record for this reference.
- 29Zhong, Q.-Y.; O-Y, X.-P.; Li, F.; Chen, S.-C.; Wu, G.; Wang, Y.-Z. Synthesis of Chemically Recyclable Poly(glycolic acid)-based Triblock Copolymers with Adjustable Performance. Chin. J. Polym. Sci. 2025, 43 (12), 2310– 2324, DOI: 10.1007/s10118-025-3454-zGoogle ScholarThere is no corresponding record for this reference.
- 30Sathe, D.; Yoon, S.; Wang, Z.; Chen, H.; Wang, J. Deconstruction of Polymers through Olefin Metathesis. Chem. Rev. 2024, 124 (11), 7007– 7044, DOI: 10.1021/acs.chemrev.3c00748Google ScholarThere is no corresponding record for this reference.
- 31Luo, J.; Demchuk, Z.; Zhao, X.; Saito, T.; Tian, M.; Sokolov, A. P.; Cao, P.-F. Elastic vitrimers: Beyond thermoplastic and thermoset elastomers. Matter 2022, 5 (5), 1391– 1422, DOI: 10.1016/j.matt.2022.04.007Google ScholarThere is no corresponding record for this reference.
- 32Yu, S.; Wu, S.; Fang, S.; Tang, Z.; Zhang, L.; Guo, B. Skeletal Network Enabling New-Generation Thermoplastic Vulcanizates. Adv. Mater. 2023, 35 (24), 2300856 DOI: 10.1002/adma.202300856Google ScholarThere is no corresponding record for this reference.
- 33Wang, L.; Shen, Y.; Li, Z. Properties Study of Thermoplastic Elastomers Prepared by Sequential Ring-opening Copolymerization of Bio-based δ-Caprolactone and L-lactide. Acta Polym. Sin. 2024, 55 (5), 582– 593Google ScholarThere is no corresponding record for this reference.
- 34Li, C.; Wang, L.; Yan, Q.; Liu, F.; Shen, Y.; Li, Z. Rapid and Controlled Polymerization of Bio-sourced δ-Caprolactone toward Fully Recyclable Polyesters and Thermoplastic Elastomers. Angew. Chem., Int. Ed. 2022, 61 (16), e202201407 DOI: 10.1002/anie.202201407Google ScholarThere is no corresponding record for this reference.
- 35Ma, K.; An, H.; Nam, J.; Reilly, L. T.; Zhang, Y.; Chen, E. Y. X.; Xu, T. Fully recyclable and tough thermoplastic elastomers from simple bio-sourced δ-valerolactones. Nat. Commun. 2024, 15 (1), 7904 DOI: 10.1038/s41467-024-52229-1Google ScholarThere is no corresponding record for this reference.
- 36Yu, X.; Wang, L.; Zhang, M.; Dong, X.; Shen, Y.; Li, Z. Tough and Closed-Loop Recyclable Thermoplastic Elastomers From Sequential Ring-Opening Copolymerization of Bio-Renewable β-Methyl-δ-Valerolactone and δ-Valerolactone. Adv. Funct. Mater. 2025, 2422779 DOI: 10.1002/adfm.202422779Google ScholarThere is no corresponding record for this reference.
- 37Wang, L.; Niu, H.; Liu, D.; Zhang, L.; Yu, X.; Chang, H.; Shen, Y.; Li, Z. High-Performance and Closed-Loop Recyclable Thermoplastic Elastomers from Sequential Ring-Opening Copolymerization of Biobased β-Methyl-δ-valerolactone and p-Dioxanone ACS Sustainable Chem. Eng. 2026 DOI: 10.1021/acssuschemeng.5c12503 .Google ScholarThere is no corresponding record for this reference.
- 38Liu, W.-N.; Wang, M.; Ding, Z.; Li, Y.; Wang, B. Modular Access to Aliphatic Polycarbonates with Tunable Properties and Dual Closed-Loop Recyclability by Polycondensation–Depolymerization–Repolymerization Strategy. Angew. Chem., Int. Ed. 2025, 64 (27), e202505333 DOI: 10.1002/anie.202505333Google ScholarThere is no corresponding record for this reference.
- 39Liu, J.; Blosch, S. E.; Volokhova, A. S.; Crater, E. R.; Gallin, C. F.; Moore, R. B.; Matson, J. B.; Byers, J. A. Using Redox-Switchable Polymerization Catalysis to Synthesize a Chemically Recyclable Thermoplastic Elastomer. Angew. Chem., Int. Ed. 2024, 63 (6), e202317699 DOI: 10.1002/anie.202317699Google ScholarThere is no corresponding record for this reference.
- 40Huang, H.-Y.; Xie, M.; Wang, S.-Q.; Huang, Y.-T.; Luo, Y.-H.; Yu, D.-G.; Cai, Z.; Zhu, J.-B. Ultratough Thermoplastic Elastomers Based on Chemically Recyclable Cycloalkyl-Substituted Polyhydroxyalkanoates. J. Am. Chem. Soc. 2025, 147 (9), 7788– 7798, DOI: 10.1021/jacs.4c17703Google ScholarThere is no corresponding record for this reference.
- 41Weng, C.; Li, X.; Tang, X. Solvent-Dependent Sequence-Controlled Copolymerization of Lactones: Tailoring Material Properties from Robust Plastics to Tough Elastomers. Angew. Chem., Int. Ed. 2025, 64 (3), e202415388 DOI: 10.1002/anie.202415388Google ScholarThere is no corresponding record for this reference.
- 42Schneiderman, D. K.; Vanderlaan, M. E.; Mannion, A. M.; Panthani, T. R.; Batiste, D. C.; Wang, J. Z.; Bates, F. S.; Macosko, C. W.; Hillmyer, M. A. Chemically Recyclable Biobased Polyurethanes. ACS Macro Lett. 2016, 5 (4), 515– 518, DOI: 10.1021/acsmacrolett.6b00193Google ScholarThere is no corresponding record for this reference.
- 43Yan, Q.; Li, C.; Yan, T.; Shen, Y.; Li, Z. Chemically Recyclable Thermoplastic Polyurethane Elastomers via a Cascade Ring-Opening and Step-Growth Polymerization Strategy from Bio-renewable δ-Caprolactone. Macromolecules 2022, 55 (10), 3860– 3868, DOI: 10.1021/acs.macromol.2c00439Google ScholarThere is no corresponding record for this reference.
- 44Zhang, M.; Yan, Q.; Yu, X.; Shen, Y.; Li, Z. High performance polyurethanes with extraordinary hydrolytic resistance prepared from bio-renewable alkyl-δ-lactones: Synthesis, properties and chemical recycling. Polym. Degrad. Stab. 2025, 237, 111322 DOI: 10.1016/j.polymdegradstab.2025.111322Google ScholarThere is no corresponding record for this reference.
- 45Yuan, L.; Zhou, W.; Shen, Y.; Li, Z. Chemically recyclable polyurethanes based on bio-renewable γ-butyrolactone: From thermoplastics to elastomers. Polym. Degrad. Stab. 2022, 204, 110116 DOI: 10.1016/j.polymdegradstab.2022.110116Google ScholarThere is no corresponding record for this reference.
- 46Zhao, Y.; Rettner, E. M.; Harry, K. L.; Hu, Z.; Miscall, J.; Rorrer, N. A.; Miyake, G. M. Chemically recyclable polyolefin-like multiblock polymers. Science 2023, 382 (6668), 310– 314, DOI: 10.1126/science.adh3353Google ScholarThere is no corresponding record for this reference.
- 47Han, X.-W.; Zhang, X.; Zhou, Y.; Maimaitiming, A.; Sun, X.-L.; Gao, Y.; Li, P.; Zhu, B.; Chen, E. Y. X.; Kuang, X.; Tang, Y. Circular olefin copolymers made de novo from ethylene and α-olefins. Nat. Commun. 2024, 15 (1), 1462 DOI: 10.1038/s41467-024-45219-wGoogle ScholarThere is no corresponding record for this reference.
- 48Sha, Y.; Chen, X.; Sun, W.; Zhou, J.; He, Y.; Xu, E.; Luo, Z.; Zhou, Y.; Jia, P. Biorenewable and circular polyolefin thermoplastic elastomers. Nat. Commun. 2024, 15 (1), 8480 DOI: 10.1038/s41467-024-52850-0Google ScholarThere is no corresponding record for this reference.
- 49Gregory, G. L.; Sulley, G. S.; Kimpel, J.; Łagodzińska, M.; Häfele, L.; Carrodeguas, L. P.; Williams, C. K. Block Poly(carbonate-ester) Ionomers as High-Performance and Recyclable Thermoplastic Elastomers. Angew. Chem., Int. Ed. 2022, 61 (47), e202210748 DOI: 10.1002/anie.202210748Google ScholarThere is no corresponding record for this reference.
- 50Schneiderman, D. K.; Hillmyer, M. A. Aliphatic Polyester Block Polymer Design. Macromolecules 2016, 49 (7), 2419– 2428, DOI: 10.1021/acs.macromol.6b00211Google ScholarThere is no corresponding record for this reference.
- 51Martello, M. T.; Hillmyer, M. A. Polylactide–Poly(6-methyl-ε-caprolactone)–Polylactide Thermoplastic Elastomers. Macromolecules 2011, 44 (21), 8537– 8545, DOI: 10.1021/ma201063tGoogle ScholarThere is no corresponding record for this reference.
- 52Olsén, P.; Borke, T.; Odelius, K.; Albertsson, A.-C. ε-Decalactone: A Thermoresilient and Toughening Comonomer to Poly(l-lactide). Biomacromolecules 2013, 14 (8), 2883– 2890, DOI: 10.1021/bm400733eGoogle ScholarThere is no corresponding record for this reference.
- 53Watts, A.; Kurokawa, N.; Hillmyer, M. A. Strong, Resilient, and Sustainable Aliphatic Polyester Thermoplastic Elastomers. Biomacromolecules 2017, 18 (6), 1845– 1854, DOI: 10.1021/acs.biomac.7b00283Google ScholarThere is no corresponding record for this reference.
- 54Diment, W. T.; Gowda, R. R.; Chen, E. Y. X. Unraveling the Mechanism of Catalyzed Melt-Phase Polyester Depolymerization via Studies of Kinetics and Model Reactions. J. Am. Chem. Soc. 2024, 146 (37), 25745– 25754, DOI: 10.1021/jacs.4c08127Google ScholarThere is no corresponding record for this reference.
- 55Zhao, W.; Guo, Z.; He, J.; Zhang, Y. Solvent-Free Chemical Recycling of Polyesters and Polycarbonates by Magnesium-Based Lewis Acid Catalyst. Angew. Chem., Int. Ed. 2025, 64 (9), e202420688 DOI: 10.1002/anie.202420688Google ScholarThere is no corresponding record for this reference.
- 56Gallin, C. F.; Lee, W.-W.; Byers, J. A. A Simple, Selective, and General Catalyst for Ring Closing Depolymerization of Polyesters and Polycarbonates for Chemical Recycling. Angew. Chem., Int. Ed. 2023, 62 (25), e202303762 DOI: 10.1002/anie.202303762Google ScholarThere is no corresponding record for this reference.
- 57Pereira, C. S. M.; Silva, V. M. T. M.; Rodrigues, A. E. Ethyl lactate as a solvent: Properties, applications and production processes─a review. Green Chem. 2011, 13 (10), 2658– 2671, DOI: 10.1039/c1gc15523gGoogle ScholarThere is no corresponding record for this reference.
- 58Wang, M.; Ding, Z.; Xiao, J.; Liao, Y.; Ma, Z.; Li, Y.; Wang, B. Chemically Recyclable Alternating Poly(thioether-alt-ester)s with Tunable Properties Enabled by Substitution Effects and Stereochemistry. Angew. Chem., Int. Ed. 2025, 64 (34), e202509468 DOI: 10.1002/anie.202509468Google ScholarThere is no corresponding record for this reference.
- 59Deacy, A. C.; Gregory, G. L.; Sulley, G. S.; Chen, T. T. D.; Williams, C. K. Sequence Control from Mixtures: Switchable Polymerization Catalysis and Future Materials Applications. J. Am. Chem. Soc. 2021, 143 (27), 10021– 10040, DOI: 10.1021/jacs.1c03250Google ScholarThere is no corresponding record for this reference.
- 60Teator, A. J.; Lastovickova, D. N.; Bielawski, C. W. Switchable Polymerization Catalysts. Chem. Rev. 2016, 116 (4), 1969– 1992, DOI: 10.1021/acs.chemrev.5b00426Google ScholarThere is no corresponding record for this reference.
- 61Zhu, Y.; Romain, C.; Williams, C. K. Selective Polymerization Catalysis: Controlling the Metal Chain End Group to Prepare Block Copolyesters. J. Am. Chem. Soc. 2015, 137 (38), 12179– 12182, DOI: 10.1021/jacs.5b04541Google ScholarThere is no corresponding record for this reference.
- 62Belleghem, L. V.; Dirix, R.; de Oliveira Silva, R.; Wéry, J.; Sakellariou, D.; Velthoven, N. V.; Vos, D. D. Recovery of Polyol and Aromatic Amines from Rigid Polyurethane Foams via Ammonolysis. JACS Au 2025, 5 (7), 3444– 3452, DOI: 10.1021/jacsau.5c00496Google ScholarThere is no corresponding record for this reference.
- 63Gama, N.; Godinho, B.; Marques, G.; Silva, R.; Barros-Timmons, A.; Ferreira, A. Recycling of polyurethane scraps via acidolysis. Chem. Eng. J. 2020, 395, 125102 DOI: 10.1016/j.cej.2020.125102Google ScholarThere is no corresponding record for this reference.
- 64Ko, J.; Zarei, M.; Lee, S. G.; Cho, K. Single-Phase Recycling of Flexible Polyurethane Foam by Glycolysis and Oxyalkylation: Large-Scale Industrial Evaluation. ACS Sustainable Chem. Eng. 2023, 11 (27), 10074– 10082, DOI: 10.1021/acssuschemeng.3c01927Google ScholarThere is no corresponding record for this reference.
- 65Borda, J.; Pásztor, G.; Zsuga, M. Glycolysis of polyurethane foams and elastomers. Polym. Degrad. Stab. 2000, 68 (3), 419– 422, DOI: 10.1016/S0141-3910(00)00030-6Google ScholarThere is no corresponding record for this reference.
- 66Xiong, M.; Schneiderman, D. K.; Bates, F. S.; Hillmyer, M. A.; Zhang, K. Scalable production of mechanically tunable block polymers from sugar. Proc. Natl. Acad. Sci. U.S.A. 2014, 111 (23), 8357– 8362, DOI: 10.1073/pnas.1404596111Google ScholarThere is no corresponding record for this reference.
- 67Meyersohn, M. S.; Block, A.; Bates, F. S.; Hillmyer, M. A. Tackling the Thermodynamic Stability of Low-Ceiling Temperature Polymers for the Preparation of Tough and Chemically Recyclable Thermoplastic Polyurethane-Urea Elastomers. Macromolecules 2024, 57 (19), 9230– 9240, DOI: 10.1021/acs.macromol.4c01431Google ScholarThere is no corresponding record for this reference.
- 68Bai, J.; Wang, Y.; You, W. Ring-opening metathesis polymerization of cyclopropene derivatives towards polyolefin elastomer analogues. Sci. China Chem. 2022, 65 (11), 2182– 2187, DOI: 10.1007/s11426-022-1395-0Google ScholarThere is no corresponding record for this reference.
- 69Zintl, M.; Rieger, B. Novel Olefin Block Copolymers through Chain-Shuttling Polymerization. Angew. Chem., Int. Ed. 2007, 46 (3), 333– 335, DOI: 10.1002/anie.200602889Google ScholarThere is no corresponding record for this reference.
- 70Sun, M.; Xiao, Y.; Liu, K.; Yang, X.; Liu, P.; Jie, S.; Hu, J.; Shi, S.; Wang, Q.; Lim, K. H. Synthesis and characterization of polyolefin thermoplastic elastomers: A review. Can. J. Chem. Eng. 2023, 101 (9), 4886– 4906, DOI: 10.1002/cjce.24825Google ScholarThere is no corresponding record for this reference.
- 71Ohtaki, H.; Deplace, F.; Vo, G. D.; LaPointe, A. M.; Shimizu, F.; Sugano, T.; Kramer, E. J.; Fredrickson, G. H.; Coates, G. W. Allyl-Terminated Polypropylene Macromonomers: A Route to Polyolefin Elastomers with Excellent Elastic Behavior. Macromolecules 2015, 48 (20), 7489– 7494, DOI: 10.1021/acs.macromol.5b01975Google ScholarThere is no corresponding record for this reference.
- 72Arriola, D. J.; Carnahan, E. M.; Hustad, P. D.; Kuhlman, R. L.; Wenzel, T. T. Catalytic Production of Olefin Block Copolymers via Chain Shuttling Polymerization. Science 2006, 312 (5774), 714– 719, DOI: 10.1126/science.1125268Google ScholarThere is no corresponding record for this reference.
- 73Si, G.; Wang, Z.; Zou, C.; Chen, C. Tandem Olefin Metathesis Polymerization to Access Degradable and Recyclable Thermosets, Thermoplastics, and Elastomers. CCS Chem. 2025, 7, 883– 892, DOI: 10.31635/ccschem.024.202404170Google ScholarThere is no corresponding record for this reference.
- 74Yang, F.; Pan, L.; Ma, Z.; Lou, Y.; Li, Y.; Li, Y. Highly elastic, strong, and reprocessable cross-linked polyolefin elastomers enabled by boronic ester bonds. Polym. Chem. 2020, 11 (19), 3285– 3295, DOI: 10.1039/D0PY00235FGoogle ScholarThere is no corresponding record for this reference.
- 75Breuillac, A.; Kassalias, A.; Nicolaÿ, R. Polybutadiene Vitrimers Based on Dioxaborolane Chemistry and Dual Networks with Static and Dynamic Cross-links. Macromolecules 2019, 52 (18), 7102– 7113, DOI: 10.1021/acs.macromol.9b01288Google ScholarThere is no corresponding record for this reference.
- 76Yang, Y.; Du, F.-S.; Li, Z.-C. Highly Stretchable, Self-Healable, and Adhesive Polyurethane Elastomers Based on Boronic Ester Bonds. ACS Appl. Polym. Mater. 2020, 2 (12), 5630– 5640, DOI: 10.1021/acsapm.0c00941Google ScholarThere is no corresponding record for this reference.
- 77Liu, Z.; Cao, M.; Liu, H.; Wu, B.; Miao, J.; Tian, Y.; Ke, Y.; Xia, R.; Qian, J. Reprocessable cross-linked EPDM elastomers with dynamic and tuneable imine network based on electronic effect. Chem. Eng. J. 2025, 522, 167494 DOI: 10.1016/j.cej.2025.167494Google ScholarThere is no corresponding record for this reference.
- 78Zhu, L.; Xu, L.; Jie, S.; Li, B.-G. Preparation of Styrene–Butadiene Rubber Vitrimers with High Strength and Toughness through Imine and Hydrogen Bonds. Ind. Eng. Chem. Res. 2023, 62 (5), 2299– 2308, DOI: 10.1021/acs.iecr.2c03133Google ScholarThere is no corresponding record for this reference.
- 79Chen, Y.; Tang, Z.; Liu, Y.; Wu, S.; Guo, B. Mechanically Robust, Self-Healable, and Reprocessable Elastomers Enabled by Dynamic Dual Cross-Links. Macromolecules 2019, 52 (10), 3805– 3812, DOI: 10.1021/acs.macromol.9b00419Google ScholarThere is no corresponding record for this reference.
- 80Jiang, X.; Zhang, Y.; Li, Z.; Ge, L.; Chen, H.; Li, X.; Chen, Q.; Gao, H.; Yu, J. Mechanically Strong, Chemical Recycling Supramolecular Elastomers via Boron-Based Dynamic Bonds. CCS Chem. 2025, 7 (12), 3757– 3770, DOI: 10.31635/ccschem.025.202506135Google ScholarThere is no corresponding record for this reference.
- 81Zhang, C.; Yang, Z.; Duong, N. T.; Li, X.; Nishiyama, Y.; Wu, Q.; Zhang, R.; Sun, P. Using Dynamic Bonds to Enhance the Mechanical Performance: From Microscopic Molecular Interactions to Macroscopic Properties. Macromolecules 2019, 52 (13), 5014– 5025, DOI: 10.1021/acs.macromol.9b00503Google ScholarThere is no corresponding record for this reference.
- 82Mao, X.; Wang, F.; Pan, L.; Ma, Z.; Li, Y. Ionic crosslinked polypropylene-based thermoplastic elastomers with excellent mechanical properties. Polym. Chem. 2024, 15 (22), 2277– 2286, DOI: 10.1039/D4PY00372AGoogle ScholarThere is no corresponding record for this reference.
- 83Zhao, J.; Zhang, Z.; Cheng, L.; Bai, R.; Zhao, D.; Wang, Y.; Yu, W.; Yan, X. Mechanically Interlocked Vitrimers. J. Am. Chem. Soc. 2022, 144 (2), 872– 882, DOI: 10.1021/jacs.1c10427Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
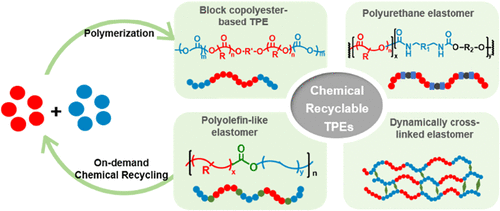
Figure 1
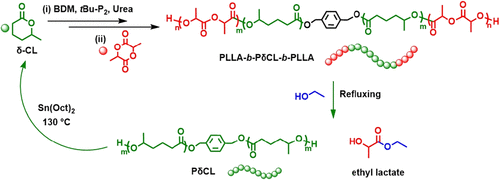
Figure 1. Illustration of preparation and chemical recycling of PLLA-b-PδCL-b-PLLA triblock elastomer. (34)
Figure 2
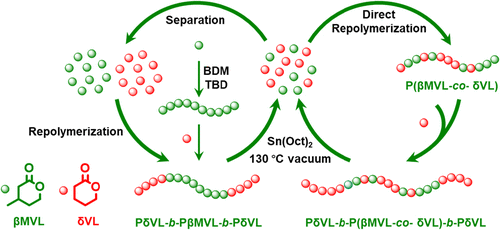
Figure 2. Illustration of the sequential ROP of βMVL and δVL toward PδVL-b-PβMVL-b-PδVL triblock copolymers and their chemical recycling. Adapted from ref (36). Copyright 2025 Wiley-VCH Verlag.
Figure 3
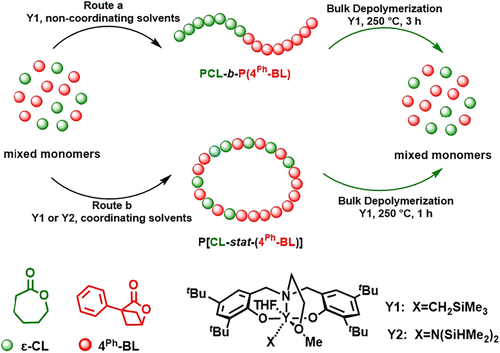
Figure 3. Solvent-dependent sequence-controlled copolymerization of 4Ph-BL with ε-CL to produce block or statistical copolyesters and their chemical recycling. (41)
Figure 4

Figure 4. Illustration of the preparation of chemically recyclable polyurethanes from biosourced alkyl-δ-lactones as well as their chemical recycling to recover pristine monomers. Reproduced from ref (44). Copyright 2025 Elsevier.
Figure 5

Figure 5. (a) Synthesis of hard and soft building blocks. (b) Preparation and closed-loop recycling of polyolefin-like multiblock elastomers. (46)
Figure 6
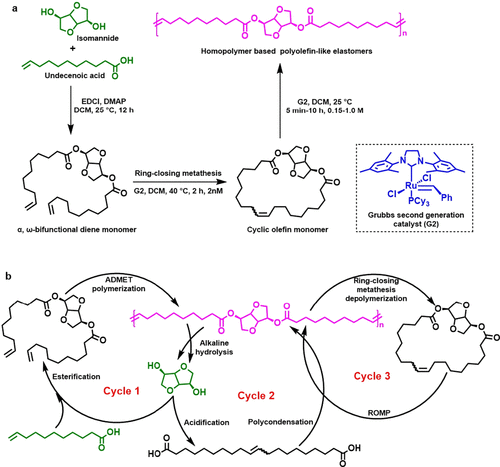
Figure 6. (a) Synthesis of homopolymer-based polyolefin-like elastomers and (b) their chemical recycling through three cycles. (48)
Figure 7
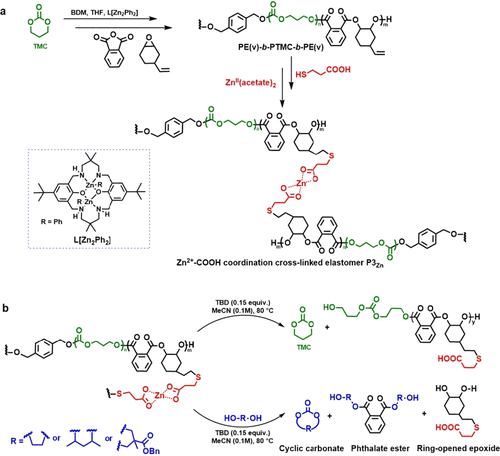
Figure 7. (a) Preparation and (b) chemical recycling of PE(v)-b-PTMC-b-PE(v) and P3Zn triblock elastomers. (49)
References
This article references 83 other publications.
- 1Seymour, R. B.; Kauffman, G. B. Elastomers: III. Thermoplastic elastomers. J. Chem. Educ. 1992, 69 (12), 967 DOI: 10.1021/ed069p967There is no corresponding record for this reference.
- 2Wang, W.; Lu, W.; Goodwin, A.; Wang, H.; Yin, P.; Kang, N.; Hong, K.; Mays, J. W. Recent advances in thermoplastic elastomers from living polymerizations: Macromolecular architectures and supramolecular chemistry. Prog. Polym. Sci. 2019, 95, 1– 31, DOI: 10.1016/j.progpolymsci.2019.04.002There is no corresponding record for this reference.
- 3Guo, Z.; Lu, X.; Wang, X.; Li, X.; Li, J.; Sun, J. Engineering of Chain Rigidity and Hydrogen Bond Cross-Linking toward Ultra-Strong, Healable, Recyclable, and Water-Resistant Elastomers. Adv. Mater. 2023, 35 (21), 2300286 DOI: 10.1002/adma.202300286There is no corresponding record for this reference.
- 4Zhu, L. Q.; X, Y. K.; Wang, W. J.; Liu, P. W.; Wang, Q. Y.; Jie, S. Y.; Hu, J. J.; Yao, Z.; Li, B. G. State-of-the-art and research progress of block-type thermoplastic elastomers. Acta Polym. Sin 2025, 56 (9), 1463– 1479There is no corresponding record for this reference.
- 5Thermoplastic Elastomer Market Size, Share & COVID-19 Impact Analysis, By Type, By Application, and Regional Forecast, 2024–2032. Fortune Business Insights. https://www.fortunebusinessinsights.com/zh/thermoplastic-elastomer-tpe-market-104515. (accessed Oct. 6, 2025).There is no corresponding record for this reference.
- 6Bates, F. S.; Hillmyer, M. A.; Lodge, T. P.; Bates, C. M.; Delaney, K. T.; Fredrickson, G. H. Multiblock Polymers: Panacea or Pandora’s Box?. Science 2012, 336 (6080), 434– 440, DOI: 10.1126/science.1215368There is no corresponding record for this reference.
- 7Sinturel, C.; Bates, F. S.; Hillmyer, M. A. High χ–Low N Block Polymers: How Far Can We Go?. ACS Macro Lett. 2015, 4 (9), 1044– 1050, DOI: 10.1021/acsmacrolett.5b00472There is no corresponding record for this reference.
- 8He, W.-N.; Xu, J.-T. Crystallization assisted self-assembly of semicrystalline block copolymers. Prog. Polym. Sci. 2012, 37 (10), 1350– 1400, DOI: 10.1016/j.progpolymsci.2012.05.002There is no corresponding record for this reference.
- 9Lynd, N. A.; Meuler, A. J.; Hillmyer, M. A. Polydispersity and block copolymer self-assembly. Prog. Polym. Sci. 2008, 33 (9), 875– 893, DOI: 10.1016/j.progpolymsci.2008.07.003There is no corresponding record for this reference.
- 10Tang, J.; Gong, S.; Zhao, B.; Chen, K.; Deng, J.; Pan, K. Biobased Thermoplastic Polyamide Elastomers: Preparation, Properties, and Applications. Ind. Eng. Chem. Res. 2024, 63 (8), 3465– 3482, DOI: 10.1021/acs.iecr.3c04554There is no corresponding record for this reference.
- 11Wang, L.; Guo, L.; Zhang, K.; Xia, Y.; Hao, J.; Wang, X. Development of Tough Thermoplastic Elastomers by Leveraging Rigid–Flexible Supramolecular Segment Interplays. Angew. Chem., Int. Ed. 2023, 62 (29), e202301762 DOI: 10.1002/anie.202301762There is no corresponding record for this reference.
- 12Gonçalves, R.; Serra, J.; Reizabal, A.; Correia, D. M.; Fernandes, L. C.; Brito-Pereira, R.; Lizundia, E.; Costa, C. M.; Lanceros-Méndez, S. Biobased polymers for advanced applications: Towards a sustainable future. Prog. Polym. Sci. 2025, 162, 101934 DOI: 10.1016/j.progpolymsci.2025.101934There is no corresponding record for this reference.
- 13Wang, Z.; Yuan, L.; Tang, C. Sustainable Elastomers from Renewable Biomass. Acc. Chem. Res. 2017, 50 (7), 1762– 1773, DOI: 10.1021/acs.accounts.7b00209There is no corresponding record for this reference.
- 14Liu, Q.; Lou, P.; Sun, Z.; Li, D.; Ji, H.; Xu, Z.; Li, L.; Xue, J.; Wang, R.; Wang, Z.; Zhang, L. Bio-Based Elastomers: Design, Properties, and Biomedical Applications. Adv. Mater. 2025, 37 (22), 2417193 DOI: 10.1002/adma.202417193There is no corresponding record for this reference.
- 15Zhang, Q.; Song, M.; Xu, Y.; Wang, W.; Wang, Z.; Zhang, L. Bio-based polyesters: Recent progress and future prospects. Prog. Polym. Sci. 2021, 120, 101430 DOI: 10.1016/j.progpolymsci.2021.101430There is no corresponding record for this reference.
- 16Liu, Y.; Zhang, M.; Shen, Y.; Li, Z. Biodegradable and Chemically Recyclable Thermoplastic Elastomers Prepared by Ring-Opening Polymerization of Cyclic Monomers. Polym. Sci. Technol. 2025, 1 (6), 529– 540, DOI: 10.1021/polymscitech.5c00050There is no corresponding record for this reference.
- 17Jeon, J. H.; Jung, J. H.; Choi, C. Toward a greener future: Exploring sustainable thermoplastic elastomers. J. Polym. Sci. 2024, 62 (4), 662– 678, DOI: 10.1002/pol.20230293There is no corresponding record for this reference.
- 18Sha, Y.; Sun, W.; Ding, S.; Ye, P.; Zhang, Y.; Jia, P. Performance-Guided Design of Chemically Recyclable Polymeric Materials: A Case Study on Thermoplastic Elastomers. ACS Polym. Au 2025, 5 (5), 432– 444, DOI: 10.1021/acspolymersau.5c00033There is no corresponding record for this reference.
- 19Zhou, D.-L.; Lin, X.; Guo, Q.-Y.; Han, D.; Fu, Q. Mechanically Accelerated Chemical Deconstruction for Nondeteriorative Recycling of Industrial Unsaturated Thermosets and Rubbers. Macromolecules 2025, 58 (20), 11361– 11369, DOI: 10.1021/acs.macromol.5c01682There is no corresponding record for this reference.
- 20Plummer, C. M.; Li, L.; Chen, Y. Ring-Opening Polymerization for the Goal of Chemically Recyclable Polymers. Macromolecules 2023, 56 (3), 731– 750, DOI: 10.1021/acs.macromol.2c01694There is no corresponding record for this reference.
- 21Shi, C.; Quinn, E. C.; Diment, W. T.; Chen, E. Y. X. Recyclable and (Bio)degradable Polyesters in a Circular Plastics Economy. Chem. Rev. 2024, 124 (7), 4393– 4478, DOI: 10.1021/acs.chemrev.3c00848There is no corresponding record for this reference.
- 22Tang, X.; Chen, E. Y. X. Toward Infinitely Recyclable Plastics Derived from Renewable Cyclic Esters. Chem 2019, 5 (2), 284– 312, DOI: 10.1016/j.chempr.2018.10.011There is no corresponding record for this reference.
- 23Li, Z.; Shen, Y.; Li, Z. Ring-Opening Polymerization of Lactones to Prepare Closed-Loop Recyclable Polyesters. Macromolecules 2024, 57 (5), 1919– 1940, DOI: 10.1021/acs.macromol.3c01912There is no corresponding record for this reference.
- 24Ibrahim, T.; Ritacco, A.; Nalley, D.; Emon, O. F.; Liang, Y.; Sun, H. Chemical recycling of polyolefins via ring-closing metathesis depolymerization. Chem. Commun. 2024, 60 (11), 1361– 1371, DOI: 10.1039/D3CC05612KThere is no corresponding record for this reference.
- 25Chen, H.; Shi, Z.; Hsu, T.-G.; Wang, J. Overcoming the Low Driving Force in Forming Depolymerizable Polymers through Monomer Isomerization. Angew. Chem., Int. Ed. 2021, 60 (48), 25493– 25498, DOI: 10.1002/anie.202111181There is no corresponding record for this reference.
- 26Sathe, D.; Zhou, J.; Chen, H.; Su, H.-W.; Xie, W.; Hsu, T.-G.; Schrage, B. R.; Smith, T.; Ziegler, C. J.; Wang, J. Olefin metathesis-based chemically recyclable polymers enabled by fused-ring monomers. Nat. Chem. 2021, 13 (8), 743– 750, DOI: 10.1038/s41557-021-00748-5There is no corresponding record for this reference.
- 27Cederholm, L.; Olsén, P.; Hakkarainen, M.; Odelius, K. Design for Recycling: Polyester- and Polycarbonate-Based A–B–A Block Copolymers and Their Recyclability Back to Monomers. Macromolecules 2023, 56 (10), 3641– 3649, DOI: 10.1021/acs.macromol.3c00274There is no corresponding record for this reference.
- 28Niu, H.; Wang, L.; Zhang, Z.; Liu, Y.; Shen, Y.; Li, Z. Ring-Opening Copolymerization of Biorenewable δ-Caprolactone with -Hexahydro-(4,5)-benzofuranone toward Closed-Loop Recyclable Copolyesters and Their Application as Pressure-Sensitive Adhesives. Chin. J. Chem. 2024, 42 (17), 2035– 2042, DOI: 10.1002/cjoc.202400244There is no corresponding record for this reference.
- 29Zhong, Q.-Y.; O-Y, X.-P.; Li, F.; Chen, S.-C.; Wu, G.; Wang, Y.-Z. Synthesis of Chemically Recyclable Poly(glycolic acid)-based Triblock Copolymers with Adjustable Performance. Chin. J. Polym. Sci. 2025, 43 (12), 2310– 2324, DOI: 10.1007/s10118-025-3454-zThere is no corresponding record for this reference.
- 30Sathe, D.; Yoon, S.; Wang, Z.; Chen, H.; Wang, J. Deconstruction of Polymers through Olefin Metathesis. Chem. Rev. 2024, 124 (11), 7007– 7044, DOI: 10.1021/acs.chemrev.3c00748There is no corresponding record for this reference.
- 31Luo, J.; Demchuk, Z.; Zhao, X.; Saito, T.; Tian, M.; Sokolov, A. P.; Cao, P.-F. Elastic vitrimers: Beyond thermoplastic and thermoset elastomers. Matter 2022, 5 (5), 1391– 1422, DOI: 10.1016/j.matt.2022.04.007There is no corresponding record for this reference.
- 32Yu, S.; Wu, S.; Fang, S.; Tang, Z.; Zhang, L.; Guo, B. Skeletal Network Enabling New-Generation Thermoplastic Vulcanizates. Adv. Mater. 2023, 35 (24), 2300856 DOI: 10.1002/adma.202300856There is no corresponding record for this reference.
- 33Wang, L.; Shen, Y.; Li, Z. Properties Study of Thermoplastic Elastomers Prepared by Sequential Ring-opening Copolymerization of Bio-based δ-Caprolactone and L-lactide. Acta Polym. Sin. 2024, 55 (5), 582– 593There is no corresponding record for this reference.
- 34Li, C.; Wang, L.; Yan, Q.; Liu, F.; Shen, Y.; Li, Z. Rapid and Controlled Polymerization of Bio-sourced δ-Caprolactone toward Fully Recyclable Polyesters and Thermoplastic Elastomers. Angew. Chem., Int. Ed. 2022, 61 (16), e202201407 DOI: 10.1002/anie.202201407There is no corresponding record for this reference.
- 35Ma, K.; An, H.; Nam, J.; Reilly, L. T.; Zhang, Y.; Chen, E. Y. X.; Xu, T. Fully recyclable and tough thermoplastic elastomers from simple bio-sourced δ-valerolactones. Nat. Commun. 2024, 15 (1), 7904 DOI: 10.1038/s41467-024-52229-1There is no corresponding record for this reference.
- 36Yu, X.; Wang, L.; Zhang, M.; Dong, X.; Shen, Y.; Li, Z. Tough and Closed-Loop Recyclable Thermoplastic Elastomers From Sequential Ring-Opening Copolymerization of Bio-Renewable β-Methyl-δ-Valerolactone and δ-Valerolactone. Adv. Funct. Mater. 2025, 2422779 DOI: 10.1002/adfm.202422779There is no corresponding record for this reference.
- 37Wang, L.; Niu, H.; Liu, D.; Zhang, L.; Yu, X.; Chang, H.; Shen, Y.; Li, Z. High-Performance and Closed-Loop Recyclable Thermoplastic Elastomers from Sequential Ring-Opening Copolymerization of Biobased β-Methyl-δ-valerolactone and p-Dioxanone ACS Sustainable Chem. Eng. 2026 DOI: 10.1021/acssuschemeng.5c12503 .There is no corresponding record for this reference.
- 38Liu, W.-N.; Wang, M.; Ding, Z.; Li, Y.; Wang, B. Modular Access to Aliphatic Polycarbonates with Tunable Properties and Dual Closed-Loop Recyclability by Polycondensation–Depolymerization–Repolymerization Strategy. Angew. Chem., Int. Ed. 2025, 64 (27), e202505333 DOI: 10.1002/anie.202505333There is no corresponding record for this reference.
- 39Liu, J.; Blosch, S. E.; Volokhova, A. S.; Crater, E. R.; Gallin, C. F.; Moore, R. B.; Matson, J. B.; Byers, J. A. Using Redox-Switchable Polymerization Catalysis to Synthesize a Chemically Recyclable Thermoplastic Elastomer. Angew. Chem., Int. Ed. 2024, 63 (6), e202317699 DOI: 10.1002/anie.202317699There is no corresponding record for this reference.
- 40Huang, H.-Y.; Xie, M.; Wang, S.-Q.; Huang, Y.-T.; Luo, Y.-H.; Yu, D.-G.; Cai, Z.; Zhu, J.-B. Ultratough Thermoplastic Elastomers Based on Chemically Recyclable Cycloalkyl-Substituted Polyhydroxyalkanoates. J. Am. Chem. Soc. 2025, 147 (9), 7788– 7798, DOI: 10.1021/jacs.4c17703There is no corresponding record for this reference.
- 41Weng, C.; Li, X.; Tang, X. Solvent-Dependent Sequence-Controlled Copolymerization of Lactones: Tailoring Material Properties from Robust Plastics to Tough Elastomers. Angew. Chem., Int. Ed. 2025, 64 (3), e202415388 DOI: 10.1002/anie.202415388There is no corresponding record for this reference.
- 42Schneiderman, D. K.; Vanderlaan, M. E.; Mannion, A. M.; Panthani, T. R.; Batiste, D. C.; Wang, J. Z.; Bates, F. S.; Macosko, C. W.; Hillmyer, M. A. Chemically Recyclable Biobased Polyurethanes. ACS Macro Lett. 2016, 5 (4), 515– 518, DOI: 10.1021/acsmacrolett.6b00193There is no corresponding record for this reference.
- 43Yan, Q.; Li, C.; Yan, T.; Shen, Y.; Li, Z. Chemically Recyclable Thermoplastic Polyurethane Elastomers via a Cascade Ring-Opening and Step-Growth Polymerization Strategy from Bio-renewable δ-Caprolactone. Macromolecules 2022, 55 (10), 3860– 3868, DOI: 10.1021/acs.macromol.2c00439There is no corresponding record for this reference.
- 44Zhang, M.; Yan, Q.; Yu, X.; Shen, Y.; Li, Z. High performance polyurethanes with extraordinary hydrolytic resistance prepared from bio-renewable alkyl-δ-lactones: Synthesis, properties and chemical recycling. Polym. Degrad. Stab. 2025, 237, 111322 DOI: 10.1016/j.polymdegradstab.2025.111322There is no corresponding record for this reference.
- 45Yuan, L.; Zhou, W.; Shen, Y.; Li, Z. Chemically recyclable polyurethanes based on bio-renewable γ-butyrolactone: From thermoplastics to elastomers. Polym. Degrad. Stab. 2022, 204, 110116 DOI: 10.1016/j.polymdegradstab.2022.110116There is no corresponding record for this reference.
- 46Zhao, Y.; Rettner, E. M.; Harry, K. L.; Hu, Z.; Miscall, J.; Rorrer, N. A.; Miyake, G. M. Chemically recyclable polyolefin-like multiblock polymers. Science 2023, 382 (6668), 310– 314, DOI: 10.1126/science.adh3353There is no corresponding record for this reference.
- 47Han, X.-W.; Zhang, X.; Zhou, Y.; Maimaitiming, A.; Sun, X.-L.; Gao, Y.; Li, P.; Zhu, B.; Chen, E. Y. X.; Kuang, X.; Tang, Y. Circular olefin copolymers made de novo from ethylene and α-olefins. Nat. Commun. 2024, 15 (1), 1462 DOI: 10.1038/s41467-024-45219-wThere is no corresponding record for this reference.
- 48Sha, Y.; Chen, X.; Sun, W.; Zhou, J.; He, Y.; Xu, E.; Luo, Z.; Zhou, Y.; Jia, P. Biorenewable and circular polyolefin thermoplastic elastomers. Nat. Commun. 2024, 15 (1), 8480 DOI: 10.1038/s41467-024-52850-0There is no corresponding record for this reference.
- 49Gregory, G. L.; Sulley, G. S.; Kimpel, J.; Łagodzińska, M.; Häfele, L.; Carrodeguas, L. P.; Williams, C. K. Block Poly(carbonate-ester) Ionomers as High-Performance and Recyclable Thermoplastic Elastomers. Angew. Chem., Int. Ed. 2022, 61 (47), e202210748 DOI: 10.1002/anie.202210748There is no corresponding record for this reference.
- 50Schneiderman, D. K.; Hillmyer, M. A. Aliphatic Polyester Block Polymer Design. Macromolecules 2016, 49 (7), 2419– 2428, DOI: 10.1021/acs.macromol.6b00211There is no corresponding record for this reference.
- 51Martello, M. T.; Hillmyer, M. A. Polylactide–Poly(6-methyl-ε-caprolactone)–Polylactide Thermoplastic Elastomers. Macromolecules 2011, 44 (21), 8537– 8545, DOI: 10.1021/ma201063tThere is no corresponding record for this reference.
- 52Olsén, P.; Borke, T.; Odelius, K.; Albertsson, A.-C. ε-Decalactone: A Thermoresilient and Toughening Comonomer to Poly(l-lactide). Biomacromolecules 2013, 14 (8), 2883– 2890, DOI: 10.1021/bm400733eThere is no corresponding record for this reference.
- 53Watts, A.; Kurokawa, N.; Hillmyer, M. A. Strong, Resilient, and Sustainable Aliphatic Polyester Thermoplastic Elastomers. Biomacromolecules 2017, 18 (6), 1845– 1854, DOI: 10.1021/acs.biomac.7b00283There is no corresponding record for this reference.
- 54Diment, W. T.; Gowda, R. R.; Chen, E. Y. X. Unraveling the Mechanism of Catalyzed Melt-Phase Polyester Depolymerization via Studies of Kinetics and Model Reactions. J. Am. Chem. Soc. 2024, 146 (37), 25745– 25754, DOI: 10.1021/jacs.4c08127There is no corresponding record for this reference.
- 55Zhao, W.; Guo, Z.; He, J.; Zhang, Y. Solvent-Free Chemical Recycling of Polyesters and Polycarbonates by Magnesium-Based Lewis Acid Catalyst. Angew. Chem., Int. Ed. 2025, 64 (9), e202420688 DOI: 10.1002/anie.202420688There is no corresponding record for this reference.
- 56Gallin, C. F.; Lee, W.-W.; Byers, J. A. A Simple, Selective, and General Catalyst for Ring Closing Depolymerization of Polyesters and Polycarbonates for Chemical Recycling. Angew. Chem., Int. Ed. 2023, 62 (25), e202303762 DOI: 10.1002/anie.202303762There is no corresponding record for this reference.
- 57Pereira, C. S. M.; Silva, V. M. T. M.; Rodrigues, A. E. Ethyl lactate as a solvent: Properties, applications and production processes─a review. Green Chem. 2011, 13 (10), 2658– 2671, DOI: 10.1039/c1gc15523gThere is no corresponding record for this reference.
- 58Wang, M.; Ding, Z.; Xiao, J.; Liao, Y.; Ma, Z.; Li, Y.; Wang, B. Chemically Recyclable Alternating Poly(thioether-alt-ester)s with Tunable Properties Enabled by Substitution Effects and Stereochemistry. Angew. Chem., Int. Ed. 2025, 64 (34), e202509468 DOI: 10.1002/anie.202509468There is no corresponding record for this reference.
- 59Deacy, A. C.; Gregory, G. L.; Sulley, G. S.; Chen, T. T. D.; Williams, C. K. Sequence Control from Mixtures: Switchable Polymerization Catalysis and Future Materials Applications. J. Am. Chem. Soc. 2021, 143 (27), 10021– 10040, DOI: 10.1021/jacs.1c03250There is no corresponding record for this reference.
- 60Teator, A. J.; Lastovickova, D. N.; Bielawski, C. W. Switchable Polymerization Catalysts. Chem. Rev. 2016, 116 (4), 1969– 1992, DOI: 10.1021/acs.chemrev.5b00426There is no corresponding record for this reference.
- 61Zhu, Y.; Romain, C.; Williams, C. K. Selective Polymerization Catalysis: Controlling the Metal Chain End Group to Prepare Block Copolyesters. J. Am. Chem. Soc. 2015, 137 (38), 12179– 12182, DOI: 10.1021/jacs.5b04541There is no corresponding record for this reference.
- 62Belleghem, L. V.; Dirix, R.; de Oliveira Silva, R.; Wéry, J.; Sakellariou, D.; Velthoven, N. V.; Vos, D. D. Recovery of Polyol and Aromatic Amines from Rigid Polyurethane Foams via Ammonolysis. JACS Au 2025, 5 (7), 3444– 3452, DOI: 10.1021/jacsau.5c00496There is no corresponding record for this reference.
- 63Gama, N.; Godinho, B.; Marques, G.; Silva, R.; Barros-Timmons, A.; Ferreira, A. Recycling of polyurethane scraps via acidolysis. Chem. Eng. J. 2020, 395, 125102 DOI: 10.1016/j.cej.2020.125102There is no corresponding record for this reference.
- 64Ko, J.; Zarei, M.; Lee, S. G.; Cho, K. Single-Phase Recycling of Flexible Polyurethane Foam by Glycolysis and Oxyalkylation: Large-Scale Industrial Evaluation. ACS Sustainable Chem. Eng. 2023, 11 (27), 10074– 10082, DOI: 10.1021/acssuschemeng.3c01927There is no corresponding record for this reference.
- 65Borda, J.; Pásztor, G.; Zsuga, M. Glycolysis of polyurethane foams and elastomers. Polym. Degrad. Stab. 2000, 68 (3), 419– 422, DOI: 10.1016/S0141-3910(00)00030-6There is no corresponding record for this reference.
- 66Xiong, M.; Schneiderman, D. K.; Bates, F. S.; Hillmyer, M. A.; Zhang, K. Scalable production of mechanically tunable block polymers from sugar. Proc. Natl. Acad. Sci. U.S.A. 2014, 111 (23), 8357– 8362, DOI: 10.1073/pnas.1404596111There is no corresponding record for this reference.
- 67Meyersohn, M. S.; Block, A.; Bates, F. S.; Hillmyer, M. A. Tackling the Thermodynamic Stability of Low-Ceiling Temperature Polymers for the Preparation of Tough and Chemically Recyclable Thermoplastic Polyurethane-Urea Elastomers. Macromolecules 2024, 57 (19), 9230– 9240, DOI: 10.1021/acs.macromol.4c01431There is no corresponding record for this reference.
- 68Bai, J.; Wang, Y.; You, W. Ring-opening metathesis polymerization of cyclopropene derivatives towards polyolefin elastomer analogues. Sci. China Chem. 2022, 65 (11), 2182– 2187, DOI: 10.1007/s11426-022-1395-0There is no corresponding record for this reference.
- 69Zintl, M.; Rieger, B. Novel Olefin Block Copolymers through Chain-Shuttling Polymerization. Angew. Chem., Int. Ed. 2007, 46 (3), 333– 335, DOI: 10.1002/anie.200602889There is no corresponding record for this reference.
- 70Sun, M.; Xiao, Y.; Liu, K.; Yang, X.; Liu, P.; Jie, S.; Hu, J.; Shi, S.; Wang, Q.; Lim, K. H. Synthesis and characterization of polyolefin thermoplastic elastomers: A review. Can. J. Chem. Eng. 2023, 101 (9), 4886– 4906, DOI: 10.1002/cjce.24825There is no corresponding record for this reference.
- 71Ohtaki, H.; Deplace, F.; Vo, G. D.; LaPointe, A. M.; Shimizu, F.; Sugano, T.; Kramer, E. J.; Fredrickson, G. H.; Coates, G. W. Allyl-Terminated Polypropylene Macromonomers: A Route to Polyolefin Elastomers with Excellent Elastic Behavior. Macromolecules 2015, 48 (20), 7489– 7494, DOI: 10.1021/acs.macromol.5b01975There is no corresponding record for this reference.
- 72Arriola, D. J.; Carnahan, E. M.; Hustad, P. D.; Kuhlman, R. L.; Wenzel, T. T. Catalytic Production of Olefin Block Copolymers via Chain Shuttling Polymerization. Science 2006, 312 (5774), 714– 719, DOI: 10.1126/science.1125268There is no corresponding record for this reference.
- 73Si, G.; Wang, Z.; Zou, C.; Chen, C. Tandem Olefin Metathesis Polymerization to Access Degradable and Recyclable Thermosets, Thermoplastics, and Elastomers. CCS Chem. 2025, 7, 883– 892, DOI: 10.31635/ccschem.024.202404170There is no corresponding record for this reference.
- 74Yang, F.; Pan, L.; Ma, Z.; Lou, Y.; Li, Y.; Li, Y. Highly elastic, strong, and reprocessable cross-linked polyolefin elastomers enabled by boronic ester bonds. Polym. Chem. 2020, 11 (19), 3285– 3295, DOI: 10.1039/D0PY00235FThere is no corresponding record for this reference.
- 75Breuillac, A.; Kassalias, A.; Nicolaÿ, R. Polybutadiene Vitrimers Based on Dioxaborolane Chemistry and Dual Networks with Static and Dynamic Cross-links. Macromolecules 2019, 52 (18), 7102– 7113, DOI: 10.1021/acs.macromol.9b01288There is no corresponding record for this reference.
- 76Yang, Y.; Du, F.-S.; Li, Z.-C. Highly Stretchable, Self-Healable, and Adhesive Polyurethane Elastomers Based on Boronic Ester Bonds. ACS Appl. Polym. Mater. 2020, 2 (12), 5630– 5640, DOI: 10.1021/acsapm.0c00941There is no corresponding record for this reference.
- 77Liu, Z.; Cao, M.; Liu, H.; Wu, B.; Miao, J.; Tian, Y.; Ke, Y.; Xia, R.; Qian, J. Reprocessable cross-linked EPDM elastomers with dynamic and tuneable imine network based on electronic effect. Chem. Eng. J. 2025, 522, 167494 DOI: 10.1016/j.cej.2025.167494There is no corresponding record for this reference.
- 78Zhu, L.; Xu, L.; Jie, S.; Li, B.-G. Preparation of Styrene–Butadiene Rubber Vitrimers with High Strength and Toughness through Imine and Hydrogen Bonds. Ind. Eng. Chem. Res. 2023, 62 (5), 2299– 2308, DOI: 10.1021/acs.iecr.2c03133There is no corresponding record for this reference.
- 79Chen, Y.; Tang, Z.; Liu, Y.; Wu, S.; Guo, B. Mechanically Robust, Self-Healable, and Reprocessable Elastomers Enabled by Dynamic Dual Cross-Links. Macromolecules 2019, 52 (10), 3805– 3812, DOI: 10.1021/acs.macromol.9b00419There is no corresponding record for this reference.
- 80Jiang, X.; Zhang, Y.; Li, Z.; Ge, L.; Chen, H.; Li, X.; Chen, Q.; Gao, H.; Yu, J. Mechanically Strong, Chemical Recycling Supramolecular Elastomers via Boron-Based Dynamic Bonds. CCS Chem. 2025, 7 (12), 3757– 3770, DOI: 10.31635/ccschem.025.202506135There is no corresponding record for this reference.
- 81Zhang, C.; Yang, Z.; Duong, N. T.; Li, X.; Nishiyama, Y.; Wu, Q.; Zhang, R.; Sun, P. Using Dynamic Bonds to Enhance the Mechanical Performance: From Microscopic Molecular Interactions to Macroscopic Properties. Macromolecules 2019, 52 (13), 5014– 5025, DOI: 10.1021/acs.macromol.9b00503There is no corresponding record for this reference.
- 82Mao, X.; Wang, F.; Pan, L.; Ma, Z.; Li, Y. Ionic crosslinked polypropylene-based thermoplastic elastomers with excellent mechanical properties. Polym. Chem. 2024, 15 (22), 2277– 2286, DOI: 10.1039/D4PY00372AThere is no corresponding record for this reference.
- 83Zhao, J.; Zhang, Z.; Cheng, L.; Bai, R.; Zhao, D.; Wang, Y.; Yu, W.; Yan, X. Mechanically Interlocked Vitrimers. J. Am. Chem. Soc. 2022, 144 (2), 872– 882, DOI: 10.1021/jacs.1c10427There is no corresponding record for this reference.


