Two-Dimensional Metal Phosphorus Trichalcogenide Nanostructure for Sustainable Energy Conversion
- Fengmei Wang *Fengmei Wang*Email: [email protected]State Key Laboratory of Chemical Resource Engineering, Beijing Advanced Innovation Center for Soft Matter Science and Engineering, Beijing University of Chemical Technology, Beijing 100029, ChinaCAS Key Laboratory of Nanosystem and Hierarchical Fabrication, National Center for Nanoscience and Technology, Beijing 100190, P. R. ChinaMore by Fengmei Wang
- and
- Marshet Getaye SendekuMarshet Getaye SendekuCAS Key Laboratory of Nanosystem and Hierarchical Fabrication, National Center for Nanoscience and Technology, Beijing 100190, P. R. ChinaResearch Institute of Tsinghua University in Shenzhen, Tsinghua University, Shenzhen 518057, P. R. ChinaMore by Marshet Getaye Sendeku
Abstract
The rational design of novel two-dimensional (2D) materials has long been sought, with a particular objective in catalysis, energy storage, and conversion. The discovery of 2D metal phosphorus trichalcogenides (MPX3) opens lots of success and breakthroughs in diverse fields such as photocatalysis, electrocatalysis, battery storage, and membrane technologies. In this chapter, a comprehensive overview of the synthesis strategies and applications involving 2D MPX3-based nanomaterials is presented. The recent development in bottom-up and top-down approaches for obtaining low-dimensional nanostructures and their limitations are briefly discussed. Their common [P2X6]4- unit, which exhibits the advantage of monitoring the adsorption-desorption chemistry in several reactions along with the rich toolbox to entertain various metal elements in their structure, makes them act as a suitable candidate in catalysis and energy storage device. Thus, the present chapter highlights the advances of 2D MPX3 nanomaterials and promising potential applications in sustainable energy conversion.
This publication is licensed for personal use by The American Chemical Society.
1 Introduction
Figure 1
2 Strategies for MPX3 Synthesis
2.1 Bottom-up Method
2.1.1 Chemical Vapor Transport
Figure 2
2.1.2 Chemical Vapor Conversion (Deposition)
Figure 3
2.2 Top-down Method
2.2.1 Mechanical Exfoliation
2.2.2 Solution and Intercalation Methods
2.2.3 Electrochemical Exfoliation Method
3 Nanostructured MPX3-Based Materials for Sustainable Energy Conversion
3.1 Photocatalysis
Figure 4
3.1.1 Water Photocatalysis for Hydrogen Gas
3.1.2 CO2 Photocatalysis for Fuels
| Photocatalyst | Morphology | Reaction conditions and light source | H2 production rate (μmol g-1 h-1) | Stability (h) | Ref. |
|---|---|---|---|---|---|
| MnPS3 | Nanosheets | 0.35 M Na2S & 0.25 M Na2SO3 aqueous solution, AM 1.5 solar light illumination | 21.2 | - | ( 27) |
| Pure water, AM 1.5 solar light illumination | 3.1 | 18 | |||
| MnPSe3 | Nanosheets | 0.35 M Na2S & 0.25 M Na2SO3 aqueous solution, AM 1.5 solar light illumination | 43.5 | - | |
| Pure water, AM 1.5 solar light illumination | 6.5 | 18 | |||
| NiPS3 | Nanosheets | Pure water, AM 1.5 solar light illumination | 6.46 | - | ( 23) |
| 0.35 M Na2S & 0.25 M Na2SO3 aqueous solution, 300 W Xe lamp illumination | 74.67 | - | |||
| Pure water, 300 W Xe lamp illumination | 26.42 | 15 | |||
| FePS3 | Bulk | 10 vol % TEOA aqueous solution, 300 W Xe lamp illumination | 94 | - | ( 35) |
| Pure water, 300 W Xe lamp illumination | 36.1 | - | |||
| Quantum sheets | 10 vol % TEOA aqueous solution, 300 W Xe lamp illumination | 290 | 40 | ||
| Pure water, 300 W Xe lamp illumination | 89.7 | - | |||
| CuInP2S6 | Nanosheets | 0.1 M Na2S & Na2SO3 aqueous solution, 300 W Xe lamp illumination with 400 mW/cm2 | 804 | 30 | ( 44) |
| FePS3 | Flakes | 0.1 M Na2S & Na2SO3 aqueous solution Pure water, 300 W Xe lamp illumination | 402.4 | 6 | ( 48) |
| Sn2P2S6 | Nanosheets | Pure water, AM 1.5 solar light illumination with 100 mW/cm2 | 202.06 | 40 | ( 26) |
| 0.35 M Na2S & 0.25 M Na2SO3 aqueous solution, AM 1.5 solar light illumination with 100 mW/cm2 | 665.5 | - |
Figure 5
3.2 Electrocatalysis
3.2.1 Electrocatalytic Water Reduction and Oxidation
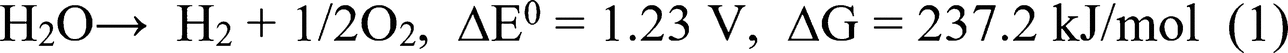
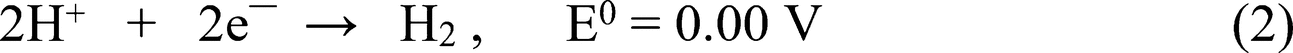
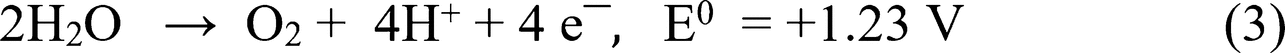
Figure 6
3.2.2 Electrocatalytic CO2 Reduction Reaction
| Half-reaction | E0/V (vs. SHE) |
|---|---|
| CO2 + e- → *CO2- | -1.90 |
| CO2 + 2H+ + 2e- → HCOOH (aq) | -0.61 |
| CO2 + 2H+ + 2e- → CO (g) + H2O | -0.53 |
| CO2 + 6H+ + 6e- → CH3OH (aq) + H2O | -0.38 |
| CO2 + 8H+ + 8e- → CH4 (g) + 2H2O | -0.24 |
| 2CO2 + 8H+ + 8e- → CH3COOH (aq) + 2H2O | -0.30 |
| 2CO2 + 10H+ + 10e- → CH3CHO (aq) + 3H2O | -0.35 |
| 2CO2 + 12H+ + 12e- → C2H5OH (aq) +3H2O | -0.33 |
| 2CO2 + 12H+ + 12e- → C2H4 (g) + 4H2O | -0.34 |
| 2CO2 + 14H+ + 14e- → C2H6 (g) + 4H2O | -0.27 |
| 3CO2 + 18H+ + 18e- → C3H7OH (aq) + 5H2O | -0.32 |
Figure 7
3.3 Battery
Figure 8
Figure 9
3.4 Ion Exchange Membrane
Figure 10
4 Conclusion
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 22179029 and 21805057). The authors also gratefully acknowledge the support of Youth Innovation Promotion Association CAS, and contribution of Mr. Ning Gao on this book chapter writing.
References
This chapter references 79 other publications.
- 1Chittari B. L. Park Y. Lee D. Han M. MacDonald A. H. Hwang E. Jung J. Electronic and Magnetic Properties of Single-Layer MPX3 Metal Phosphorous Trichalcogenides Phys. Rev. B 2016 94 18 184428Google Scholar1Electronic and magnetic properties of single-layer MPX3 metal phosphorous trichalcogenidesChittari, Bheema Lingam; Park, Youngju; Lee, Dongkyu; Han, Moonsup; MacDonald, Allan H.; Hwang, Euyheon; Jung, JeilPhysical Review B (2016), 94 (18), 184428/1-184428/16CODEN: PRBHB7; ISSN:2469-9950. (American Physical Society)We survey the electronic structure and magnetic properties of two-dimensional (2D) MPX3 (M=V,Cr,Mn,Fe,Co,Ni,Cu,Zn, and X=S,Se,Te) transition-metal chalcogenophosphates to shed light on their potential role as single-layer van der Waals materials that possess magnetic order. Our ab initio calcns. predict that most of these single-layer materials are antiferromagnetic semiconductors. The band gaps of the antiferromagnetic states decrease as the at. no. of the chalcogen atom increases (from S to Se to Te), leading in some cases to half-metallic ferromagnetic states or to nonmagnetic metallic states. We find that the competition between antiferromagnetic and ferromagnetic states can be substantially influenced by gating and by strain engineering. The sensitive interdependence we find between magnetic, structural, and electronic properties establishes the potential of this 2D materials class for applications in spintronics.
- 2Susner M. A. Chyasnavichyus M. McGuire M. A. Ganesh P. Maksymovych P. Metal Thio- and Selenophosphates as Multifunctional van der Waals Layered Materials Adv. Mater. 2017 29 38 1602852Google ScholarThere is no corresponding record for this reference.
- 3Samal R. Sanyal G. Chakraborty B. Rout C. S. Two-Dimensional Transition Metal Phosphorous Trichalcogenides (MPX3): A Review on Emerging Trends, Current State and Future Perspectives J. Mater. Chem. A 2021 9 5 2560 2591Google Scholar3Two-dimensional transition metal phosphorous trichalcogenides (MPX3): a review on emerging trends, current state and future perspectivesSamal, Rutuparna; Sanyal, Gopal; Chakraborty, Brahmananda; Rout, Chandra SekharJournal of Materials Chemistry A: Materials for Energy and Sustainability (2021), 9 (5), 2560-2591CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)A review. Profound scientific attention has been devoted to the expansive family of functional 2D materials soon after the isolation of graphene and the discovery of Dirac physics. Of particular note, the re-emergence of layer architecture metal thio(seleno) phosphate materials (MPX3, X = S, Se) materials with appreciable tunability in dimension and proximity on correlated ground states opened novel potential avenues for the ultimate theor. (structural, magnetic) and realistic research. The wide spanned bandgap of 1.2-3.5 eV harnessed multifarious fundamental magnetic, electronic, ferroelec., optical, intercalation properties and their prominent implementation in a plethora of applications, such as Li-ion batteries, field-effect transistors, visible and UV photodetectors, photo-electrochem. reactions and hydrogen (H2) storage. This review examines many of the scarcely touched aspects, such as multiple Dirac cones, vibrational properties, and responses in the presence of strain of MPX3 materials. In addn., the latest advancement in the emergent applications, physiochem. properties and assorted growth methodologies considering the palpable vol. of research has been explored in detail in order to bring across a more complete discussion and outlook. We strongly believe that this review will be very handy for researchers working in various branches of science and engineering, such as physics, chem., materials science, and chem. engineering.
- 4Wang F. Shifa T. A. Yu P. He P. Liu Y. Wang F. Wang Z. Zhan X. Lou X. Xia F. He J. New Frontiers on van der Waals Layered Metal Phosphorous Trichalcogenides Adv. Funct. Mater. 2018 28 37 1802151Google ScholarThere is no corresponding record for this reference.
- 5Friedel M. C. Soufre et ses composés—sur une nouvelle série de sulfophosphures, les thiohypophosphates CR l’Academie. Sci. Ser 1894 3 119 260Google ScholarThere is no corresponding record for this reference.
- 6Ouvrard G. Brec R. Rouxel J. Structural determination of some MPS3 layered phases (M = Mn, Fe, Co, Ni and Cd) Mater. Res. Bull. 1985 20 1181 1189Google Scholar6Structural determination of some MPS3 layered phases (M = Mn, Fe, Co, Ni and Cd)Ouvrard, G.; Brec, R.; Rouxel, J.Materials Research Bulletin (1985), 20 (10), 1181-9CODEN: MRBUAC; ISSN:0025-5408.The structures of most of the MPS3 phases (M = Mn, Fe, Co, Ni, Cd) were detd. from single-crystal anal. They show the expected CdCl2 structural type, some weak disorder on the cationic sites being detected on NiPS3 and CoPS3. Fair stoichiometry is inferred for the phases since no interslab cation could be seen in the Van der Waals' gap of the structures. Polytypism may occur in NiPS3. At. parameters are given.
- 7Hwangbo K. Zhang Q. Jiang Q. Wang Y. Fonseca J. Wang C. Diederich G. M. Gamelin D. R. Xiao D. Chu J. H. Yao W. Xu X. Highly Anisotropic Excitons and Multiple Phonon Bound States in a van der Waals Antiferromagnetic Insulator Nat. Nanotechnol. 2021 16 6 655 660Google Scholar7Highly anisotropic excitons and multiple phonon bound states in a van der Waals antiferromagnetic insulatorHwangbo, Kyle; Zhang, Qi; Jiang, Qianni; Wang, Yong; Fonseca, Jordan; Wang, Chong; Diederich, Geoffrey M.; Gamelin, Daniel R.; Xiao, Di; Chu, Jiun-Haw; Yao, Wang; Xu, XiaodongNature Nanotechnology (2021), 16 (6), 655-660CODEN: NNAABX; ISSN:1748-3387. (Nature Portfolio)Two-dimensional (2D) semiconductors enable the investigation of light-matter interactions in low dimensions1,2. Yet, the study of elementary photoexcitations in 2D semiconductors with intrinsic magnetic order remains a challenge due to the lack of suitable materials3,4. Here, we report the observation of excitons coupled to zigzag antiferromagnetic order in the layered antiferromagnetic insulator NiPS3. The exciton exhibits a narrow photoluminescence linewidth of roughly 350μeV with near-unity linear polarization. When we reduce the sample thickness from five to two layers, the photoluminescence is suppressed and eventually vanishes for the monolayer. This suppression is consistent with the calcd. bandgap of NiPS3, which is highly indirect for both the bilayer and the monolayer5. Furthermore, we observe strong linear dichroism (LD) over a broad spectral range. The optical anisotropy axes of LD and of photoluminescence are locked to the zigzag direction. Furthermore, their temp. dependence is reminiscent of the in-plane magnetic susceptibility anisotropy. Hence, our results indicate that LD and photoluminescence could probe the symmetry breaking magnetic order parameter of 2D magnetic materials. In addn., we observe over ten exciton-A1g-phonon bound states on the high-energy side of the exciton resonance, which we interpret as signs of a strong modulation of the ligand-to-metal charge-transfer energy by electron-lattice interactions. Our work establishes NiPS3 as a 2D platform for exploring magneto-exciton physics with strong correlations.
- 8Kang S. Kim K. Kim B. H. Kim J. Sim K. I. Lee J. U. Lee S. Park K. Yun S. Kim T. Nag A. Walters A. Garcia-Fernandez M. Li J. Chapon L. Zhou K. J. Son Y. W. Kim J. H. Cheong H. Park J. G. Coherent Many-Body Exciton in van der Waals Antiferromagnet NiPS3 Nature 2020 583 7818 785 789Google Scholar8Coherent many-body exciton in van der Waals antiferromagnet NiPS3Kang, Soonmin; Kim, Kangwon; Kim, Beom Hyun; Kim, Jonghyeon; Sim, Kyung Ik; Lee, Jae-Ung; Lee, Sungmin; Park, Kisoo; Yun, Seokhwan; Kim, Taehun; Nag, Abhishek; Walters, Andrew; Garcia-Fernandez, Mirian; Li, Jiemin; Chapon, Laurent; Zhou, Ke-Jin; Son, Young-Woo; Kim, Jae Hoon; Cheong, Hyeonsik; Park, Je-GeunNature (London, United Kingdom) (2020), 583 (7818), 785-789CODEN: NATUAS; ISSN:0028-0836. (Nature Research)Abstr.: An exciton is the bosonic quasiparticle of electron-hole pairs bound by the Coulomb interaction. Bose-Einstein condensation of this exciton state has long been the subject of speculation in various model systems2,3, and examples have been found more recently in optical lattices and two-dimensional materials4-9. Unlike these conventional excitons formed from extended Bloch states4-9, excitonic bound states from intrinsically many-body localized states are rare. Here we show that a spin-orbit-entangled exciton state appears below the Neel temp. of 150 K in NiPS3, an antiferromagnetic van der Waals material. It arises intrinsically from the archetypal many-body states of the Zhang-Rice singlet10,11, and reaches a coherent state assisted by the antiferromagnetic order. Using configuration-interaction theory, we det. the origin of the coherent excitonic excitation to be a transition from a Zhang-Rice triplet to a Zhang-Rice singlet. We combine three spectroscopic tools-resonant inelastic X-ray scattering, photoluminescence and optical absorption-to characterize the exciton and to demonstrate an extremely narrow excitonic linewidth below 50 K. The discovery of the spin-orbit-entangled exciton in antiferromagnetic NiPS3 introduces van der Waals magnets as a platform to study coherent many-body excitons.
- 9Joy P. A. Vasudevan S. Magnetism in the Layered Transition-Metal Thiophosphates MPS3 (M=Mn, Fe, and Ni) Phys. Rev. B 1992 46 9 5425 5433Google Scholar9Magnetism in the layered transition-metal thiophosphates MPS3 (M = manganese, iron, and nickel)Joy, P. A.; Vasudevan, S.Physical Review B: Condensed Matter and Materials Physics (1992), 46 (9), 5425-33CODEN: PRBMDO; ISSN:0163-1829.Anisotropic magnetic susceptibilities of single crystals of the layered transition-metal thiophosphates MnPS3, FePS3, and NiPS3 have been measured as a function of temp. The materials order antiferromagnetically at low temps., the Neel temps. being 78, 123, and 155 K, resp. In the ordered state, the magnetization axis lies perpendicular to the layers of MnPS3 and FePS3, while for NiPS3 it lies in the layer. In the paramagnetic regime, the anisotropies of these compds. are different; while the susceptibility for MnPS3 is isotropic and that for NiPS3 shows only a weak anisotropy, FePS3 exhibits highly anisotropic susceptibility. The anisotropic susceptibilities have been analyzed to obtain information on the state of the magnetic ions and the nature of magnetic interactions between them. The results show that MnPS3, FePS3, and NiPS3 form a unique class of compds. Although all three compds. are isostructural with the magnetic lattice being the two-dimensional honeycomb, the spin dimensionalities for the three are different. While MnPS3 is best described by the isotropic Heisenberg Hamiltonian, FePS3 is most effectively treated by the Ising model and NiPS3 by the anisotropic Heisenberg Hamiltonian. The origin of the anisotropy in these compds. has been discussed, and it is shown how it arises from a combination of spin-orbit coupling and the trigonal distortion of the MS6 octahedra. The magnetic exchange const., J and the zero-field splitting energies of the ground state of the transition-metal ion have been evaluated from the anisotropic paramagnetic susceptibilities.
- 10Wildes A. R. Simonet V. Ressouche E. McIntyre G. J. Avdeev M. Suard E. Kimber S. A. J. Lançon D. Pepe G. Moubaraki B. Hicks T. J. Magnetic Structure of the Quasi-Two-Dimensional Antiferromagnet NiPS3 Phys. Rev. B 2015 92 22 224408Google Scholar10Magnetic structure of the quasi-two-dimensional antiferromagnet NiPS3Wildes, A. R.; Simonet, V.; Ressouche, E.; McIntyre, G. J.; Avdeev, M.; Suard, E.; Kimber, S. A. J.; Lancon, D.; Pepe, G.; Moubaraki, B.; Hicks, T. J.Physical Review B: Condensed Matter and Materials Physics (2015), 92 (22), 224408/1-224408/11CODEN: PRBMDO; ISSN:1098-0121. (American Physical Society)The magnetic structure of the quasi-two-dimensional antiferromagnet NiPS3 has been detd. by magnetometry and a variety of neutron diffraction techniques. The expts. show that the samples must be carefully handled, as gluing influences the magnetometry measurements while preferred orientation complicates the interpretation of powder diffraction measurements. Our global set of consistent measurements show numerous departures from previously published results. We show that the compd. adopts a k = [010] antiferromagnetic structure with the moment directions mostly along the a axis, and that the paramagnetic susceptibility is isotropic. The crit. behavior was also investigated through the temp. dependence of the magnetic Bragg peaks below the N´eel temp.
- 11Wang F. Mathur N. Janes A. N. Sheng H. He P. Zheng X. Yu P. DeRuiter A. J. Schmidt J. R. He J. Jin S. Defect-Mediated Ferromagnetism in Correlated Two-Dimensional Transition Metal Phosphorus Trisulfides Sci. Adv. 2021 7 43 eabj4086Google ScholarThere is no corresponding record for this reference.
- 12Wang X. Cao J. Lu Z. Cohen A. Kitadai H. Li T. Tan Q. Wilson M. Lui C. H. Smirnov D. Sharifzadeh S. Ling X. Spin-Induced Linear Polarization of Photoluminescence in Antiferromagnetic van der Waals Crystals Nat. Mater. 2021 20 7 964 970Google Scholar12Spin-induced linear polarization of photoluminescence in antiferromagnetic van der Waals crystalsWang, Xingzhi; Cao, Jun; Lu, Zhengguang; Cohen, Arielle; Kitadai, Hikari; Li, Tianshu; Tan, Qishuo; Wilson, Matthew; Lui, Chun Hung; Smirnov, Dmitry; Sharifzadeh, Sahar; Ling, XiNature Materials (2021), 20 (7), 964-970CODEN: NMAACR; ISSN:1476-1122. (Nature Portfolio)Antiferromagnets are promising components for spintronics due to their terahertz resonance, multilevel states and absence of stray fields. However, the zero net magnetic moment of antiferromagnets makes the detection of the antiferromagnetic order and the investigation of fundamental spin properties notoriously difficult. Here, we report an optical detection of Neel vector orientation through an ultra-sharp photoluminescence in the van der Waals antiferromagnet NiPS3 from bulk to atomically thin flakes. The strong correlation between spin flipping and elec. dipole oscillator results in a linear polarization of the sharp emission, which aligns perpendicular to the spin orientation in the crystal. By applying an in-plane magnetic field, we achieve manipulation of the photoluminescence polarization. This correlation between emitted photons and spins in layered magnets provides routes for investigating magneto-optics in two-dimensional materials, and hence opens a path for developing opto-spintronic devices and antiferromagnet-based quantum information technologies.
- 13Kim K. Lim S. Y. Lee J. U. Lee S. Kim T. Y. Park K. Jeon G. S. Park C. H. Park J. G. Cheong H. Suppression of Magnetic Ordering in XXZ-Type Antiferromagnetic Monolayer NiPS3 Nat. Commun. 2019 10 1 345Google Scholar13Suppression of magnetic ordering in XXZ-type antiferromagnetic monolayer NiPS3Kim Kangwon; Lim Soo Yeon; Lee Jae-Ung; Cheong Hyeonsik; Lee Sungmin; Park Kisoo; Park Je-Geun; Lee Sungmin; Kim Tae Yun; Park Kisoo; Park Cheol-Hwan; Park Je-Geun; Kim Tae Yun; Park Cheol-Hwan; Jeon Gun SangNature communications (2019), 10 (1), 345 ISSN:.How a certain ground state of complex physical systems emerges, especially in two-dimensional materials, is a fundamental question in condensed-matter physics. A particularly interesting case is systems belonging to the class of XY Hamiltonian where the magnetic order parameter of conventional nature is unstable in two-dimensional materials leading to a Berezinskii-Kosterlitz-Thouless transition. Here, we report how the XXZ-type antiferromagnetic order of a magnetic van der Waals material, NiPS3, behaves upon reducing the thickness and ultimately becomes unstable in the monolayer limit. Our experimental data are consistent with the findings based on renormalization-group theory that at low temperatures a two-dimensional XXZ system behaves like a two-dimensional XY one, which cannot have a long-range order at finite temperatures. This work provides the experimental examination of the XY magnetism in the atomically thin limit and opens opportunities of exploiting these fundamental theorems of magnetism using magnetic van der Waals materials.
- 14Belianinov A. He Q. Dziaugys A. Maksymovych P. Eliseev E. Borisevich A. Morozovska A. Banys J. Vysochanskii Y. Kalinin S. V. CuInP2S6 Room Temperature Layered Ferroelectric Nano Lett. 2015 15 6 3808 3814Google Scholar14CuInP2S6 Room Temperature Layered FerroelectricBelianinov, A.; He, Q.; Dziaugys, A.; Maksymovych, P.; Eliseev, E.; Borisevich, A.; Morozovska, A.; Banys, J.; Vysochanskii, Y.; Kalinin, S. V.Nano Letters (2015), 15 (6), 3808-3814CODEN: NALEFD; ISSN:1530-6984. (American Chemical Society)The authors explore ferroelec. properties of cleaved 2-dimensional flakes of copper indium thiophosphate, CuInP2S6 (CITP), and probe size effects along with limits of ferroelec. phase stability, by ambient and ultra high vacuum scanning probe microscopy. CITP belongs to the only material family known to display ferroelec. polarization in a van der Waals, layered crystal at room temp. and above. The authors' measurements directly reveal stable, ferroelec. polarization as evidenced by domain structures, switchable polarization, and hysteresis loops. At room temp. the domain structure of flakes thicker than 100 nm is similar to the cleaved bulk surfaces, whereas <50 nm polarization disappears. The authors ascribe this behavior to a known instability of polarization due to depolarization field. Also, polarization switching at high bias is also assocd. with ionic mobility, as evidenced both by macroscopic measurements and by formation of surface damage under the tip at a bias of 4 V-likely due to copper redn. Mobile Cu ions may therefore also contribute to internal screening mechanisms. The existence of stable polarization in a van-der-Waals crystal naturally points toward new strategies for ultimate scaling of polar materials, quasi-2D, and single-layer materials with advanced and nonlinear dielec. properties that are presently not found in any members of the growing graphene family.
- 15Dziaugys A. Banys J. Macutkevic J. Sobiestianskas R. Vysochanskii Y. Dipolar Glass Phase in Ferrielectrics: CuInP2S6 and Ag0.1Cu0.9InP2S6 Crystals Phys. Status Solidi A 2010 207 8 1960 1967Google Scholar15Dipolar glass phase in ferrielectrics: CuInP2S6 and Ag0.1Cu0.9InP2S6 crystalsDziaugys, Andrius; Banys, Juras; Macutkevic, Jan; Sobiestianskas, Ricardas; Vysochanskii, YulianPhysica Status Solidi A: Applications and Materials Science (2010), 207 (8), 1960-1967CODEN: PSSABA; ISSN:1862-6300. (Wiley-VCH Verlag GmbH & Co. KGaA)The dielec. properties of CuInP2S6 and Ag0.1Cu0.9InP2S6 crystals were investigated in wide frequency (20 Hz-3 GHz) and temp. ranges (25-360 K). The low frequencies dielec. spectra at high temps. are highly influenced by the high ionic cond. The temp. dependences of the static dielec. permittivity and the mean relaxation time reveal the first order order-disorder ferrielec. phase transition at Tc = 288 K in Ag0.1Cu0.9InP2S6. The cond. and its activation energy exhibit only a weak change close to the ferrielec. phase transition temp. in both investigated crystals. The freezing phenomena in CuInP2S6 and Ag0.1Cu0.9InP2S6 revealed the complex dielec. permittivity behavior characteristic for transition into dipolar glass state.
- 16Silipigni L. Schiro L. Scolaro L. M. De Luca G. Salvato G. Lithium Ions Conduction in Li2xMn1-xPS3 Films Philos. Mag. 2014 94 35 4026 4036Google Scholar16Lithium ions conduction in Li2xMn1-xPS3 filmsSilipigni, L.; Schiro, L.; Monsu Scolaro, L.; De Luca, G.; Salvato, G.Philosophical Magazine (2014), 94 (35), 4026-4036CODEN: PMHABF; ISSN:1478-6435. (Taylor & Francis Ltd.)Li2xMn1-xPS3 films have been synthesized by exfoliating MnPS3 through the successive intercalations of K+ and Li+ ions. Their dielec. response has been measured from 80 to 350 K in the frequency range of (102-106) Hz. The obtained data have been analyzed in terms of both complex permittivity ε* and the ac cond. σac. The frequency dependence of σac has been interpreted in terms of the Jonscher's law, whose exponent n decreases by increasing temp. The n values lie between 0.479 and 0.501 and are typical of materials in which the ac cond. is due to multiple hops of carriers. By analyzing the σdc temp. dependence, the obsd. dielec. response has been attributed to the intercalated lithium ions, and the Li2xMn1-xPS3 films have been classified as hopping charge carrier systems.
- 17Kuzminskii Y. V. Voronin B. M. Redin N. N. Iron and Nickel Phosphorus Trisulfides as Electroactive Materials for Primary Lithium Batteries J. Power Sources 1995 55 2 133 141Google Scholar17Iron and nickel phosphorus trisulfides as electroactive materials for primary lithium batteriesKuzminskii, Y. V.; Voronin, B. M.; Redin, N. N.Journal of Power Sources (1995), 55 (2), 133-41CODEN: JPSODZ; ISSN:0378-7753. (Elsevier)The theor. specific capacities of a family of layered compds. MPX3 (M = Fe, Co, Ni; X = S, Se) for current-producing reactions involving 1.5, 2, 6 and 9 lithium atoms per MPX3 mol. have been estd. These data show that FePS3 and NiPS3 are good electroactive materials for primary lithium cells. Conditions which allow one to shorten substantially the synthesis time of iron and nickel phosphorus trisulfides in ampules are presented. According to cond. measurements in the 293-673 K temp. range, NiPS3 is an intrinsic and FePS3 an extrinsic semiconductor. The discharge characteristics of Li/MPS3 (M = Fe, Ni) cells with an org. electrolyte have been studied; they confirmed that the participation of nine electrons in the redox process is possible. For a primary button cell of the std. size 2325, with a FePS3 cathode, the specific capacity and specific energy values obtained in a discharge 1.8-1.2 V range at load resistances of 3.0 to 30 kΩ were 500-1160 Ah/kg and 700-1770 Wh/kg (on a pure FePS3 basis), resp.
- 18Qian X. Chen L. Yin L. Liu Z. Pei S. Li F. Hou G. Chen S. Song L. Thebo K. H. Cheng H. M. Ren W. CdPS3 Nanosheets-Based Membrane with High Proton Conductivity Enabled by Cd Vacancies Science 2020 370 6516 596 600Google Scholar18CdPS3 nanosheets-based membrane with high proton conductivity enabled by Cd vacanciesQian, Xitang; Chen, Long; Yin, Lichang; Liu, Zhibo; Pei, Songfeng; Li, Fan; Hou, Guangjin; Chen, Shuangming; Song, Li; Thebo, Khalid Hussain; Cheng, Hui-Ming; Ren, WencaiScience (Washington, DC, United States) (2020), 370 (6516), 596-600CODEN: SCIEAS; ISSN:1095-9203. (American Association for the Advancement of Science)P transport in nanochannels under humid conditions is crucial for the application in energy storage and conversion. However, existing materials, including Nafion, suffer from limited cond. of up to 0.2 S per cm. The authors report a class of membranes assembled with 2-dimensional transition-metal P trichalcogenide nanosheets, in which the transition-metal vacancies enable exceptionally high ion cond. A Cd0.85PS3Li0.15H0.15 membrane exhibits a p conduction dominant cond. of ∼0.95 S per cm at 90°elsius and 98% relative humidity. This performance mainly originates from the abundant p donor centers, easy p desorption, and excellent hydration of the membranes induced by Cd vacancies. The authors also obsd. superhigh Li ion cond. in Cd0.85PS3Li0.3 and Mn0.77PS3Li0.46 membranes.
- 19Du K. Z. Wang X. Z. Liu Y. Hu P. Utama M. I. Gan C. K. Xiong Q. Kloc C. Weak Van der Waals Stacking, Wide-Range Band Gap, and Raman Study on Ultrathin Layers of Metal Phosphorus Trichalcogenides ACS Nano 2016 10 2 1738 1743Google Scholar19Weak Van der Waals Stacking, Wide-Range Band Gap, and Raman Study on Ultrathin Layers of Metal Phosphorus TrichalcogenidesDu, Ke-zhao; Wang, Xing-zhi; Liu, Yang; Hu, Peng; Utama, M. Iqbal Bakti; Gan, Chee Kwan; Xiong, Qihua; Kloc, ChristianACS Nano (2016), 10 (2), 1738-1743CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)2D semiconducting metal phosphorus trichalcogenides, particularly the bulk crystals of MPS3 (M = Fe, Mn, Ni, Cd and Zn) sulfides and MPSe3 (M = Fe and Mn) selenides, have been synthesized, crystd. and exfoliated into monolayers. The Raman spectra of monolayer FePS3 and 3-layer FePSe3 show the strong intralayer vibrations and structural stability of the atomically thin layers under ambient condition. The band gaps can be adjusted by element choices in the range of 1.3-3.5 eV. The wide-range band gaps suggest their optoelectronic applications in a broad wavelength range. The calcd. cleavage energies of MPS3 are smaller than that of graphite. Therefore, the monolayers used for building of heterostructures by van der Waals stacking could be considered as the candidates for artificial 2D materials with unusual ferroelec. and magnetic properties.
- 20Nitsche R. Wild P. Crystal Growth of Metal-Phosphorus-Sulfur Compounds by Vapor Transport Mater. Res. Bull. 1970 5 6 419 423Google Scholar20Crystal growth of metal-phosphorus-sulfur compounds by vapor transportNitsche, Rudolf; Wild, PetrMaterials Research Bulletin (1970), 5 (6), 419-23CODEN: MRBUAC; ISSN:0025-5408.Single crystals of the thiophosphates: Cu3PS4, InPS4, GaPS4, BiPS4 and the thiohypophosphates Sn2P2S6, Cd2P2S6, Fe2P2S6, and Mn2P2S6 were grown in sizes up to 10 mm by vapor transport with I. Lattice parameters and space groups are given.
- 21Gusmao R. Sofer Z. Pumera M. Metal Phosphorous Trichalcogenides (MPCh3 ): From Synthesis to Contemporary Energy Challenges Angew. Chem. Int. Ed. Engl. 2019 58 28 9326 9337Google Scholar21Metal Phosphorous Trichalcogenides (MPCh3 ): From Synthesis to Contemporary Energy ChallengesGusmao Rui; Sofer Zdenek; Pumera MartinAngewandte Chemie (International ed. in English) (2019), 58 (28), 9326-9337 ISSN:.Owing to their unique physical and chemical properties, layered two-dimensional (2D) materials have been established as the most significant topic in materials science for the current decade. This includes layers comprising mono-element (graphene, phosphorene), di-element (metal dichalcogenides), and even multi-element. A distinctive class of 2D layered materials is the metal phosphorous trichalcogenides (MPCh3 , Ch=S, Se), first synthesized in the late 1800s. Having an unusual intercalation behavior, MPCh3 were intensively studied in the 1970s for their magnetic properties and as secondary electrodes in lithium batteries, but fell from scrutiny until very recently, being 2D nanomaterials. Based on their synthesis and most significant properties, the present surge of reports related to water-splitting catalysis and energy storage are discussed in detail. This Minireview is intended as a baseline for the anticipated new wave of researchers who aim to explore these 2D layered materials for their electrochemical energy applications.
- 22Gave M. A. Bilc D. Mahanti S. D. Breshears J. D. Kanatzidis M. G. On the Lamellar Compounds CuBiP2Se6, AgBiP2Se6 and AgBiP2S6. Antiferroelectric Phase Transitions due to Cooperative Cu+ and Bi3+ Ion Motion Inorg. Chem. 2005 44 15 5293 5303Google Scholar22On the Lamellar Compounds CuBiP2Se6, AgBiP2Se6 and AgBiP2S6. Antiferroelectric Phase Transitions Due to Cooperative Cu+ and Bi3+ Ion MotionGave, Matthew A.; Bilc, Daniel; Mahanti, S. D.; Breshears, Jean D.; Kanatzidis, Mercouri G.Inorganic Chemistry (2005), 44 (15), 5293-5303CODEN: INOCAJ; ISSN:0020-1669. (American Chemical Society)CuBiP2Se6, AgBiP2Se6, and AgBiP2S6 were prepd. from the corresponding elements. CuBiP2Se6 and AgBiP2Se6 crystallize in the space group R‾3 with a 6.5532(16) and c 39.762(13) Å for CuBiP2Se6 and a 6.6524(13) and c 39.615(15) Å for AgBiP2Se6. AgBiP2S6 crystallizes in the triclinic space group P‾1 with a 6.3833(13), b 7.1439(14), c 9.5366(19) Å, α 91.89(3), β 91.45(3), γ 94.05(3)°. CuBiP2Se6 was found to exhibit a temp.-dependent antiferroelec. ordering of the Cu+ and Bi3+ ions in the lattice. An intermediate and a fully ordered structure were refined at 173 and 97 K, resp. Electronic band and total energy calcns. at the DFT level clearly suggest that the antiferroelec. model is energetically favored over the paraelec. and hypothetical ferrielec. models. This phase transition can be classified as a 2nd-order Jahn-Teller distortion. The antiferroelec. state of CuBiP2Se6 is an indirect gap semiconductor. The compds. were characterized with DTA and solid-state UV/visible diffuse reflectance spectroscopy. Generalized implications regarding the expected ferroelec. behavior of compds. in the CuMP2Se6 system (M = trivalent metal) are discussed.
- 23Wang F. Shifa T. A. He P. Cheng Z. Chu J. Liu Y. Wang Z. Wang F. Wen Y. Liang L. He J. Two-Dimensional Metal Phosphorus Trisulfide Nanosheet with Solar Hydrogen-Evolving Activity Nano Energy 2017 40 673 680Google Scholar23Two-dimensional metal phosphorus trisulfide nanosheet with solar hydrogen-evolving activityWang, Fengmei; Shifa, Tofik Ahmed; He, Peng; Cheng, Zhongzhou; Chu, Junwei; Liu, Yang; Wang, Zhenxing; Wang, Feng; Wen, Yao; Liang, Lirong; He, JunNano Energy (2017), 40 (), 673-680CODEN: NEANCA; ISSN:2211-2855. (Elsevier Ltd.)The development and utilization of photocatalysts to realize water-splitting without any external bias or sacrificial agents has received the limelight. As a novel two-dimensional layered material, metal phosphorus trichalcogenides (MPTs) cause wide research interest, presently. However, the growth of ultrathin two-dimensional MPT crystals is a great challenge to hinder their application. Here, we initially grow few-at. layered nickel phosphorus trisulfide (NiPS3) as promising photocatalyst for hydrogen evolution. The as-prepd. NiPS3 hexagonal nanosheet, as thin as few at. layers (≤ 3.5 nm), has lateral size of larger than 15 μm. These ultrathin NiPS3 crystals can directly generate hydrogen gas from pure water without any sacrificial agents under sunlight. With UV photoelectron spectrometer and electrochem. impedance spectroscopy, we show that the attractive photocatalytic activity of the ultrathin NiPS3 crystals arise from their appropriate positions of the band edges. This discovery is expected to make a contribution to develop next generation solar-fuel conversion catalysts for H2 prodn.
- 24Yu P. Meng J. Wang F. Sendeku M. G. Wu B. Sui X. Gao N. Zhan X. Lou X. Wang Z. He J. Carbonate-Ion-Mediated Photogenerated Hole Transfer to Boost Hydrogen Production J. Phys. Chem. C 2022 126 25 10367 10377Google Scholar24Carbonate-Ion-Mediated Photogenerated Hole Transfer to Boost Hydrogen ProductionYu, Peng; Meng, Jun; Wang, Fengmei; Sendeku, Marshet Getaye; Wu, Binglan; Sui, Xinyu; Gao, Ning; Zhan, Xueying; Lou, Xiaoding; Wang, Zhenxing; He, JunJournal of Physical Chemistry C (2022), 126 (25), 10367-10377CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Sustainable and scalable H2 evolution through water photocatalysis is an attractive path for carbon-neutral energy supply; however, it is severely limited by sluggish charge sepn. and photocorrosion of semiconductor photocatalysts. Here, we demonstrate that earth-abundant carbonate ions, widely existing in daily-life water, serve as a hole mediator to redirect the photogenerated hole transfer pathway and then promote the hole-transfer kinetics. The accelerated hole transfer could efficiently reduce the recombination of electron-hole pairs for continuous H2 prodn. with improved photostability of catalysts, including layered indium phosphorus sulfide (In4/3P2S6) and cadmium sulfide. A sustainable H2 evolution rate of 5.1 mmol g-1 h-1 within 60 h or more operation is achieved in the presence of CO32- anions. In situ ESR (ESR) spectroscopy studies and transient absorption (TA) measurements reveal that the CO32-/CO3- redox couple could rapidly shuttle the photogenerated holes from OH radicals anchored on the catalyst surface, effectively eliminating the recombination of electron-hole pairs and catalyst oxidn. for boosted H2 generation. The carbonate-ion-mediated hole-transfer strategy provides a new paradigm for designing a cost-effective and advanced photosynthetic system in practical applications.
- 25Zhou J. Zhu C. Zhou Y. Dong J. Li P. Zhang Z. Wang Z. Lin Y. C. Shi J. Zhang R. Zheng Y. Yu H. Tang B. Liu F. Wang L. Liu L. Liu G. B. Hu W. Gao Y. Yang H. Gao W. Lu L. Wang Y. Suenaga K. Liu G. Ding F. Yao Y. Liu Z. Composition and Phase Engineering of Metal Chalcogenides and Phosphorous Chalcogenides Nat. Mater. 2022Google ScholarThere is no corresponding record for this reference.https://doi.org/10.1038/s41563-022-01291-5.Google ScholarThere is no corresponding record for this reference.
- 26Sendeku M. G. Wang F. Cheng Z. Yu P. Gao N. Zhan X. Wang Z. He J. Nonlayered Tin Thiohypodiphosphate Nanosheets: Controllable Growth and Solar-Light-Driven Water Splitting ACS Appl. Mater. Interfaces 2021 13 11 13392 13399Google Scholar26Nonlayered Tin Thiohypodiphosphate Nanosheets: Controllable Growth and Solar-Light-Driven Water SplittingSendeku, Marshet G.; Wang, Fengmei; Cheng, Zhongzhou; Yu, Peng; Gao, Ning; Zhan, Xueying; Wang, Zhenxing; He, JunACS Applied Materials & Interfaces (2021), 13 (11), 13392-13399CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)As a promising candidate in various fields, including energy conversion and electronics, layered van der Waals metal phosphorus trichalcogenides (MPX3) have been widely explored. In addn. to the layered structures, MPX3 comprising post-transition metals (i.e., Sn and Pb) are known to form a unique 3D framework with nonlayered structure. However, the nonlayered two-dimensional (2D) crystals of this family have remained unexplored until now. Herein, we successfully synthesized 2D nonlayered tin thiohypodiphosphate (Sn2P2S6) nanosheets, having an indirect bandgap of 2.25 eV and a thickness down to ~ 10 nm. The as-obtained nanosheets demonstrate promising photocatalytic water splitting activity to generate H2 in pure water under simulated solar light (AM 1.5G). Moreover, the ultrathin Sn2P2S6 catalyst shows auspicious performance and stability with a continuous operation of 40 h. This work is not only an expansion of the MPX3 family, but it is also a major milestone in the search for new materials for future energy conversion.
- 27Shifa T. A. Wang F. Cheng Z. He P. Liu Y. Jiang C. Wang Z. He J. High Crystal Quality 2D Manganese Phosphorus Trichalcogenide Nanosheets and Their Photocatalytic Activity Adv. Funct. Mater. 2018 28 18 1800548Google ScholarThere is no corresponding record for this reference.
- 28Zhu W. Gan W. Muhammad Z. Wang C. Wu C. Liu H. Liu D. Zhang K. He Q. Jiang H. Zheng X. Sun Z. Chen S. Song L. Exfoliation of Ultrathin FePS3 Layers as a Promising Electrocatalyst for the Oxygen Evolution Reaction Chem. Commun. 2018 54 35 4481 4484Google Scholar28Exfoliation of ultrathin FePS3 layers as a promising electrocatalyst for the oxygen evolution reactionZhu, Wen; Gan, Wei; Muhammad, Zahir; Wang, Changda; Wu, Chuanqiang; Liu, Hengjie; Liu, Daobin; Zhang, Ke; He, Qun; Jiang, Hongliang; Zheng, Xusheng; Sun, Zhe; Chen, Shuangming; Song, LiChemical Communications (Cambridge, United Kingdom) (2018), 54 (35), 4481-4484CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)Few-layer ternary FePS3 nanosheets, prepd. via chem. vapor transport synthesis and ball-milling exfoliation, exhibit excellent electrocatalytic performance for the oxygen evolution reaction in an alk. medium. Combined with first principles calcns., our X-ray spectroscopy and HRTEM results clearly reveal that the introduction of in-plane defects in FePS3 layers after exfoliation and formation of a FePS3-FeOOH heterostructure during the OER process largely contribute to the catalytic activity enhancement.
- 29Sang Y. Wang L. Cao X. Ding G. Ding Y. Hao Y. Xu N. Yu H. Li L. Peng S. Emerging 2D-Layered MnPS3/rGO Composite as a Superior Anode for Sodium-Ion Batteries J. Alloys Compd. 2020 831 154775Google Scholar29Emerging 2D-Layered MnPS3/rGO composite as a superior anode for sodium-ion batteriesSang, Yan; Wang, Lvxuan; Cao, Xi; Ding, Gaofei; Ding, Yonghao; Hao, Yanan; Xu, Na; Yu, Hanzhi; Li, Linlin; Peng, ShengjieJournal of Alloys and Compounds (2020), 831 (), 154775CODEN: JALCEU; ISSN:0925-8388. (Elsevier B.V.)Two-dimensional (2D) layered materials were widely studied due to their unique Na storage properties and rapid ion transport rates. Herein, the authors synthesized a 2-dimensional layered transition metal P sulfide MnPS3 by one step high-temp. solid-phase synthesis. After combining with graphene by high-energy ball milling followed by high-temp. Ar calcination, the novel 2D/2D heterojunction of extra-thin MnPS3/rGO was successfully prepd. The resultant MnPS3/rGO hybrid can strengthen the cond. of the material and ameliorate the vol. change during the insertion/extn. process of the Na-ion storage, compared to the pristine MnPS3. As a result, the 2D/2D heterojunction of ultra-thin MnPS3/rGO composite exhibits high cycling performance (290 mA-h g-1 after 150 cycles at 0.2 A g-1), capacity retention rates up to 92%. The superior performance is ascribed to the combination of extra-thin MnPS3 nanosheets with graphene, which effectively enhances the interface contact area and the electronic transmission rate, greatly improving the adaptability of vol. change and interfacial charge transfer abilities. This work provides a novel and promising MnPS3/rGO anode for Na-ion batteries.
- 30Liu J. Li X. B. Wang D. Lau W.-M. Peng P. Liu L.-M. Diverse and Tunable Electronic Structures of Single-layer Metal Phosphorus Trichalcogenides for Photocatalytic Water Splitting J. Chem. Phys. 2014 140 5 054707Google Scholar30Diverse and tunable electronic structures of single-layer metal phosphorus trichalcogenides for photocatalytic water splittingLiu, Jian; Li, Xi-Bo; Wang, Da; Lau, Woon-Ming; Peng, Ping; Liu, Li-MinJournal of Chemical Physics (2014), 140 (5), 054707/1-054707/7CODEN: JCPSA6; ISSN:0021-9606. (American Institute of Physics)The family of bulk metal phosphorus trichalcogenides (APX3, A = MII, MI0.5MIII0.5; X = S, Se; MI, MII, and MIII represent Group-I, Group-II, and Group-III metals, resp.) has attracted great attentions because such materials not only own magnetic and ferroelec. properties, but also exhibit excellent properties in hydrogen storage and lithium battery because of the layered structures. Many layered materials have been exfoliated into two-dimensional (2D) materials, and they show distinct electronic properties compared with their bulks. Here the authors present a systematical study of single-layer metal phosphorus trichalcogenides by d. functional theory calcns. The results show that the single layer metal phosphorus trichalcogenides have very low formation energies, which indicates that the exfoliation of single layer APX3 should not be difficult. The family of single layer metal phosphorus trichalcogenides exhibits a large range of band gaps from 1.77 to 3.94 eV, and the electronic structures are greatly affected by the metal or the chalcogenide atoms. The calcd. band edges of metal phosphorus trichalcogenides further reveal that single-layer ZnPSe3, CdPSe3, Ag0.5Sc0.5PSe3, and Ag0.5In0.5PX3 (X = S and Se) have both suitable band gaps for visible-light driving and sufficient over-potentials for water splitting. More fascinatingly, single-layer Ag0.5Sc0.5PSe3 is a direct band gap semiconductor, and the calcd. optical absorption further convinces that such materials own outstanding properties for light absorption. Such results demonstrate that the single layer metal phosphorus trichalcogenides own high stability, versatile electronic properties, and high optical absorption, thus such materials have great chances to be high efficient photocatalysts for water-splitting. (c) 2014 American Institute of Physics.
- 31Mukherjee D. Austeria M. P. Sampath S. Few-Layer Iron Selenophosphate, FePSe3: Efficient Electrocatalyst toward Water Splitting and Oxygen Reduction Reactions ACS Appl. Energy Mater. 2018 1 1 220 231Google Scholar31Few-Layer Iron Selenophosphate, FePSe3: Efficient Electrocatalyst toward Water Splitting and Oxygen Reduction ReactionsMukherjee, Debdyuti; Muthu, Austeria P.; Sampath, S.ACS Applied Energy Materials (2018), 1 (1), 220-231CODEN: AAEMCQ; ISSN:2574-0962. (American Chemical Society)There has been a spurt of activity in using layered MPX3 (M = transition metal, X = chalcogen, S/Se/Te) compds. in various studies including catalysis and devices. In the present study, low band gap, ternary iron selenophosphate (FePSe3) is introduced as an excellent and highly stable trifunctional electrocatalyst for hydrogen evolution, oxygen evolution, and oxygen redn. reactions. It is obsd. that the present catalyst is useful in evolving hydrogen over a wide pH range including seawater environment. D. functional theory calcns. reveal various parameters that help improve the electrocatalytic activity of the layered material. Covalency of the Fe-Se bond, distortion in the crystal structure, and adsorption properties are shown to be responsible for the obsd. high catalytic activity.
- 32Mukherjee D. Austeria P. M. Sampath S. Two-Dimensional, Few-Layer Phosphochalcogenide, FePS3: A New Catalyst for Electrochemical Hydrogen Evolution over Wide pH Range ACS Energy Lett. 2016 1 2 367 372Google Scholar32Two-Dimensional, Few-Layer Phosphochalcogenide, FePS3: A New Catalyst for Electrochemical Hydrogen Evolution over Wide pH RangeMukherjee, Debdyuti; Austeria, P. Muthu; Sampath, S.ACS Energy Letters (2016), 1 (2), 367-372CODEN: AELCCP; ISSN:2380-8195. (American Chemical Society)A layered MPS3-type compd., FePS3, is introduced as an electrocatalyst for hydrogen evolution reaction (HER). The non-noble metal-based FePS3 is a semiconductor that could be solvent exfoliated into few-layer, two-dimensional (2D) nanosheets. The 2-dimensional thin sheets exhibit very good catalytic activity and stability toward HER over a wide pH range of acidic, alk., phosphate buffer, and 3.5% aq. NaCl solns. The Tafel slope and exchange c.d. in acidic medium are ∼(45-50) mV/dec and 1±0.2 × 10-3 A/cm2, resp. The stability of the catalyst is very good. D. functional theory calcns. reveal P and S as favorable hydrogen adsorption sites. This material opens up a new class of ternary, layered semiconductors for various electrocatalytic studies and might also become important from a device physics point of view.
- 33Jenjeti R. N. Kumar R. Sampath S. Two-Dimensional, Few-layer NiPS3 for Flexible Humidity Sensor with High Selectivity J. Mater. Chem. A 2019 7 24 14545 14551Google Scholar33Two-dimensional, few-layer NiPS3 for flexible humidity sensor with high selectivityJenjeti, Ramesh Naidu; Kumar, Rajat; Sampath, S.Journal of Materials Chemistry A: Materials for Energy and Sustainability (2019), 7 (24), 14545-14551CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Chem. and elec. sensitive two-dimensional (2D) nanomaterials are of immense interest as probing electrodes for wearable electronic devices. A new family of two dimensional (2D) layered materials, namely metal phosphochalcogenides (MPX3), are potential candidates towards the development of sensors for various analytes. Herein, we demonstrate the ability of few-layer NiPS3 nanosheets for humidity sensing by fabricating a cost-effective, flexible sensor device. The results indicate that the NiPS3 nanosheet-based humidity sensors possess high sensitivity with a response of ∼106, superior selectivity, and most importantly, rapid response, recovery times and good reproducibility. Response times of ∼1-2 s at low humidity levels and ∼3 s at high humidity levels with recovery times of ∼2-3 s are obsd. The device was tested in both flat and bent states, causing no prominent changes in the response; hence, the sensor is an excellent candidate for use in flexible devices. The characteristics of the NiPS3-based sensor were further investigated using complex impedance studies and in situ Raman spectroscopy to understand the sensing mechanism. The fast response and recovery assocd. with the NiPS3-based humidity sensors allow real time monitoring of human respiration and water evapn. on skin. These humidity sensors can also be utilized for non-contact anal. and hence will be an attractive candidate for health and environmental monitoring.
- 34Song B. Li K. Yin Y. Wu T. Dang L. Caban-Acevedo M. Han J. Gao T. Wang X. Zhang Z. Schmidt J. R. Xu P. Jin S. Tuning Mixed Nickel Iron Phosphosulfide Nanosheet Electrocatalysts for Enhanced Hydrogen and Oxygen Evolution ACS Catal. 2017 7 12 8549 8557Google Scholar34Tuning Mixed Nickel Iron Phosphosulfide Nanosheet Electrocatalysts for Enhanced Hydrogen and Oxygen EvolutionSong, Bo; Li, Kai; Yin, Ying; Wu, Tao; Dang, Lianna; Caban-Acevedo, Miguel; Han, Jiecai; Gao, Tangling; Wang, Xianjie; Zhang, Zhihua; Schmidt, J. R.; Xu, Ping; Jin, SongACS Catalysis (2017), 7 (12), 8549-8557CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)Highly efficient earth-abundant electrocatalysts for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) are of great importance for renewable energy conversion systems. Herein, guided by theor. calcns., we demonstrate highly efficient water splitting in alk. soln. using quaternary mixed nickel iron phosphosulfide (Ni1-xFexPS3) nanosheets (NSs), even though neither NiPS3 nor FePS3 is a good HER (or OER) electrocatalyst. With tuned electronic structure and improved elec. cond. induced by mixing appropriate amt. of Fe into NiPS3, Ni0.9Fe0.1PS3 NSs display excellent HER activity (an overpotential of 72 mV vs reversible hydrogen electrode (RHE) at a geometric catalytic c.d. of -10 mA cm-2 and a Tafel slope of 73 mV dec-1), which is among the best HER catalysts under alk. conditions. Ni0.9Fe0.1PS3 NSs also show a good apparent OER activity (an overpotential of 329 mV vs RHE at a catalytic c.d. of 20 mA cm-2 and a Tafel slope of 69 mV dec-1), although structural investigation indicates the formation of Ni(Fe)OOH and Ni(Fe)(OH)2 layers on the catalyst surface after OER reactions as likely the real active species. These mixed nickel iron phosphosulfide non-precious-metal electrocatalysts with enhanced intrinsic activity and long-term stability and durability should have great potential in overall water-splitting applications.
- 35Cheng Z. Shifa T. A. Wang F. Gao Y. He P. Zhang K. Jiang C. Liu Q. He J. High-Yield Production of Monolayer FePS3 Quantum Sheets via Chemical Exfoliation for Efficient Photocatalytic Hydrogen Evolution Adv. Mater. 2018 30 26 1707433Google ScholarThere is no corresponding record for this reference.
- 36Dinh Hoa L. Thanh Luan P. Ghimire G. Dinh Loc D. Lee Y. H. Revealing Antiferromagnetic Transition of van der Waals MnPS3 via Vertical Tunneling Electrical Resistance Measurement APL Mater. 2019 7 8 081102Google ScholarThere is no corresponding record for this reference.
- 37Wang J. Li X. Wei B. Sun R. Yu W. Hoh H. Y. Xu H. Li J. Ge X. Chen Z. Su C. Wang Z. Activating Basal Planes of NiPS3 for Hydrogen Evolution by Nonmetal Heteroatom Doping Adv. Funct. Mater. 2020 30 12 1908708Google Scholar37Activating Basal Planes of NiPS3 for Hydrogen Evolution by Nonmetal Heteroatom DopingWang, Jun; Li, Xinzhe; Wei, Bin; Sun, Rong; Yu, Wei; Hoh, Hui Ying; Xu, Haomin; Li, Jing; Ge, Xingbo; Chen, Zuxin; Su, Chenliang; Wang, ZhongchangAdvanced Functional Materials (2020), 30 (12), 1908708CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)NiPS3, one of the most promising catalysts among transition metal trichalcogenidophosphates (MTPs) in hydrogen evolution reaction (HER) electrocatalysis, is still inhibited by its unsatisfactory activity originating from its semiconducting nature and inert basal plane. Here, it is proposed, for the first time, to engineer the basal surface activity of NiPS3 by nonmetal heteroatom doping, and predict that the degree to which the valance band of NiPS3 is filled dominates not only the elec. cond. of the catalyst, but also the strength of hydrogen adsorption at its surface. Direct exptl. evidence is offered that in all the single nonmetal doping samples, C-doped NiPS3 exhibits the optimum activity owing to its moderate filled state of valance band and that C, N codoping even shows Pt-like activity with an ultralow overpotential of 53.2 mV to afford 10 mA cm-2 c.d. and a high exchange c.d. of 0.7 mA cm-2 in 1 M KOH. The findings that less valance electrons of dopants than substitutional atoms are of pivotal importance for improving HER activity of NiPS3 catalyst pave the way for readily designing novel MTPs of ever high performance to replace the incumbent Pt-based catalysts.
- 38Li X. Fang Y. Wang J. Wei B. Qi K. Hoh H. Y. Hao Q. Sun T. Wang Z. Yin Z. Zhang Y. Lu J. Bao Q. Su C. High-Yield Electrochemical Production of Large-Sized and Thinly Layered NiPS3 Flakes for Overall Water Splitting Small 2019 15 30 1902427Google ScholarThere is no corresponding record for this reference.
- 39Sekine T. Jouanne M. Julien C. Balkanski M. Raman Scattering in the Antiferromagnet FePS3 Intercalated with Lithium Mater. Sci. Eng. B Solid State Mater. Adv. Technol. 1989 3 1−2 91 95Google ScholarThere is no corresponding record for this reference.
- 40Fujishima A. Honda K. Electrochemical Photolysis of Water at a Semiconductor Electrode Nature 1972 238 5358 37 38Google Scholar40Electrochemical photolysis of water at a semiconductor electrodeFujishima, Akira; Honda, KenichiNature (London, United Kingdom) (1972), 238 (5358), 37-8CODEN: NATUAS; ISSN:0028-0836.For electrochem. decompn. of H2O, a p.d. of >1.23 V is necessary between the anode and the cathode. This p.d. is equal to the energy of radiation with wavelength of ∼1000 nm. This energy, in the form of visible light, can be used effectively in an electrochem. system to decomp. H2O. A novel type of electrochem. cell was developed, in which a TiO2 electrode was connected with a Pt-black electrode through an external load. The direction of current revealed that oxidn. occurred at the TiO2 electrode and redn. at the Pt-black electrode. To increase the efficiency of the process, more reducible species, such as dissolved O2 or Fe3+, must be added in the Pt electrode compartment. The use of a p-type semiconductor electrode instead of Pt leads to more effective electrochem. photolysis of H2O.
- 41Nakata K. Fujishima A. TiO2 Photocatalysis: Design and Applications J. Photochem. Photobiol. C 2012 13 3 169 189Google Scholar41TiO2 photocatalysis: Design and applicationsNakata, Kazuya; Fujishima, AkiraJournal of Photochemistry and Photobiology, C: Photochemistry Reviews (2012), 13 (3), 169-189CODEN: JPPCAF; ISSN:1389-5567. (Elsevier B.V.)In this review, recent developments in the area of TiO2 photocatalysis research, in terms of new materials from a structural design perspective, have been summarized. The dimensionality assocd. with the structure of a TiO2 material can affect its properties and functions, including its photocatalytic performance, and also more specifically its surface area, adsorption, reflectance, adhesion, and carrier transportation properties. The authors provide a brief introduction to the current situation in TiO2 photocatalysis, and describe structurally controlled TiO2 photocatalysts which can be classified into zero-, one-, two-, and three-dimensional structures. Furthermore, novel applications of TiO2 surfaces for the fabrication of wettability patterns and for printing are discussed.
- 42Chiesa P. Consonni S. Kreutz T. Williams R. Co-Production of Hydrogen, Electricity and CO2 from Coal with Commercially Ready Technology. PartA: Performance and Emissions Int. J. Hydrogen Energy 2005 30 7 747 767Google Scholar42Co-production of hydrogen, electricity and CO2 from coal with commercially ready technology. Part A: Performance and emissionsChiesa, Paolo; Consonni, Stefano; Kreutz, Thomas; Williams, RobertInternational Journal of Hydrogen Energy (2005), 30 (7), 747-767CODEN: IJHEDX; ISSN:0360-3199. (Elsevier Ltd.)This two-part paper investigates performances, costs and prospects of using com. ready technol. to convert coal to H2 and electricity, with CO2 capture and storage. Part A focuses on plant configuration and the evaluation of performances and CO2 emissions. Part B focuses on economics, establishing benchmarks for the assessment of novel technologies and guidelines for technol. development. In the co-prodn. plants considered in the paper, coal is gasified to synthesis gas in an entrained flow gasifier. The syngas is cooled, cleaned of particulate matter, and shifted (to primarily H2 and CO2) in sour water-gas shift reactors. After further cooling, H2S is removed from the syngas using a phys. solvent (Selexol); CO2 is then removed from the syngas, again using Selexol; after being stripped from the solvent, the CO2 is dried and compressed to 150 bar for pipeline transport and underground storage. High purity H2 (99.999%) is extd. from the H2-rich syngas via a pressure swing adsorption (PSA) unit and delivered at 60 bar. The PSA purge gas is compressed and burned in a conventional gas turbine combined cycle, generating co-product electricity. The H2/electricity ratio can be varied by lowering the steam-to-carbon ratio in the syngas or by letting part of the de-carbonized syngas bypass the PSA unit. Performances and emissions of H2/electricity co-prodn. with CO2 capture are compared with those of a system that vents the CO2. We examine different methods of syngas heat recovery (quench vs. radiant cooling) and explore the effects of changing the electricity/H2 ratio, gasifier pressure and hydrogen purity. Results show that state-of-the-art com. technol. allows transferring to de-carbonized hydrogen 57-58% of coal LHV, while exporting to the grid decarbonized electricity amounting to 2-6% of coal LHV. In contrast to decarbonizing coal IGCC electricity, which entails a loss of 6-8 percentage points of electricity conversion when capturing CO2 as an alternative to venting it, CO2 capture for H2 prodn. gives a minor energy penalty (∼ 2 percentage points of export electricity). For H2 prodn., the efficiency gain achievable by hot syngas cooling vs. quench is a modest 2 percentage point increase in electricity for export, compared to 2-4 percentage points in the electricity case. Reducing H2 purity or increasing gasification pressure has minor effects on performance.
- 43Zhang X. Zhao X. Wu D. Jing Y. Zhou Z. MnPSe3 Monolayer: A Promising 2D Visible-Light Photohydrolytic Catalyst with High Carrier Mobility Adv. Sci. 2016 3 10 1600062Google Scholar43MnPSe3 Monolayer: A Promising 2D Visible-Light Photohydrolytic Catalyst with High Carrier MobilityZhang Xu; Zhao Xudong; Wu Dihua; Jing Yu; Zhou ZhenAdvanced science (Weinheim, Baden-Wurttemberg, Germany) (2016), 3 (10), 1600062 ISSN:2198-3844.The 2D material single-layer MnPSe3 would be a promising photocatalyst for water splitting, as indicated by the proper positions of band edges, strong absorption in visible-light spectrum, broad applicability (pH = 0 - 7), and high carrier mobility.
- 44Yu P. Wang F. Meng J. Shifa T. A. Sendeku M. G. Fang J. Li S. Cheng Z. Lou X. He J. Few-Layered CuInP2S6 Nanosheet with Sulfur Vacancy Boosting Photocatalytic Hydrogen Evolution CrystEngComm 2021 591 598Google Scholar44Few-layered CuInP2S6 nanosheet with sulfur vacancy boosting photocatalytic hydrogen evolutionYu, Peng; Wang, Fengmei; Meng, Jun; Shifa, Tofik Ahmed; Sendeku, Marshet Getaye; Fang, Ju; Li, Shuxian; Cheng, Zhongzhou; Lou, Xiaoding; He, JunCrystEngComm (2021), 23 (3), 591-598CODEN: CRECF4; ISSN:1466-8033. (Royal Society of Chemistry)Photochem. water splitting offers an economic and sustainable approach for solar energy conversion into hydrogen fuel to mitigate the problem of greenhouse gas emissions. To this end, exploring novel semiconductor photocatalysts, which have efficient light absorption and thermodynamically favorable band alignment for water splitting, is crucial. Here, we rationally develop a new photocatalyst of CuInP2S6 nanosheets to generate hydrogen gas under light illumination. The CuInP2S6 nanosheet (with a thickness of around 4-7 nm) photocatalyst exhibits a high hydrogen prodn. rate of 804μmol g-1 h-1, eight times higher than that of the microsheet counterpart, due to the introduced abundant sulfur vacancies. Exptl. characterization and theor. calcns. verify that the prolonged carrier lifetime and optimized band alignment in ultrathin CuInP2S6 nanosheets boost photocatalytic hydrogen evolution. This work opens a new avenue for photocatalysis via using novel layered binary metal phosphorous trichalcogenides.
- 45Navarro R. M. Sánchez-Sánchez M. C. Alvarez-Galvan M. C. Valle F. d. Fierro J. L. G. Hydrogen Production from Renewable Sources: Biomass and Photocatalytic Opportunities Energy Environ. Sci. 2009 2 1 35 54Google Scholar45Hydrogen production from renewable sources: biomass and photocatalytic opportunitiesNavarro, R. M.; Sanchez-Sanchez, M. C.; Alvarez-Galvan, M. C.; del Valle, F.; Fierro, J. L. G.Energy & Environmental Science (2009), 2 (1), 35-54CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)A review. The demand for hydrogen over the coming decade is expected to grow for both traditional uses (ammonia, methanol, refinery) and running fuel cells. At least in the near future, this thirst for hydrogen will be quenched primarily through the reforming of fossil fuels. However, reforming fossil fuels emits huge amts. of carbon dioxide. One approach to reduce carbon dioxide emissions, which is considered first in this review, is to apply reforming methods to alternative renewable materials. Such materials might be derived from plant crops, agricultural residues, woody biomass, etc. Clean biomass is a proven source of renewable energy that is already used for generating heat, electricity, and liq. transportation fuels. Clean biomass and biomass-derived precursors such as ethanol and sugars are appropriate precursors for producing hydrogen through different conversion strategies. Virtually no net greenhouse gas emissions result because a natural cycle is maintained, in which carbon is extd. from the atm. during plant growth and released during hydrogen prodn. The second option explored here is hydrogen prodn. from water splitting by means of the photons in the visible spectrum. The sun provides silent and precious energy that is distributed fairly evenly all over the earth. However, its tremendous potential as a clean, safe and economical energy source cannot be exploited unless it is accumulated or converted into more useful forms of energy. Finally, this review discusses the use of semiconductors, more specifically CdS and CdS-based semiconductors, which are able to absorb photons in the visible region of the spectrum. The energy stored within a semiconductor as electronic energy (electrons and holes) can be used to split water mols. by simultaneous reactions into H2 and O2. This conversion of solar energy into a clean fuel (H2) is perhaps the greatest challenge for scientists in the 21st century.
- 46Shimura K. Yoshida H. Heterogeneous Photocatalytic Hydrogen Production from Water and Biomass Derivatives Energy Environ. Sci. 2011 4 7 2467 2481Google Scholar46Heterogeneous photocatalytic hydrogen production from water and biomass derivativesShimura, Katsuya; Yoshida, HisaoEnergy & Environmental Science (2011), 4 (7), 2467-2481CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)A review. Hydrogen, a clean energy carrier, should be produced from not the irreproducible fossil fuels but renewable resources. Currently hydrogen is mainly produced by the reforming of petroleum and natural gas at high temp., which consumes huge energy. Thus, development of an alternative hydrogen prodn. method is absolutely imperative to mitigate the environmental and energy issues. Photocatalytic hydrogen prodn. from water and renewable resources is one of the attractive systems, since it can utilize the solar energy. In this short review, we remind you that the systems of the photocatalytic hydrogen prodn. from water and biomass derivs., such as ethanol, glycerol, sugars and methane, should be considered as available candidates for the hydrogen prodn. method from renewable resources and solar energy, and these systems have been improved by developments of new photocatalysts with some cocatalysts, modifications of photocatalysts, optimization of reaction conditions, and so on.
- 47Zhang Y. Zhao Y. Bao C. Xiao Y. Xiang Y. Song M. Huang W. Ma L. Hou H. Chen X. Facile synthesis of cadmium phosphorus trisulfide nanosheets for highly efficient photocatalytic performance J. Alloys Compd. 2022 909 164731Google Scholar47Facile synthesis of cadmium phosphorus trisulfide nanosheets for highly efficient photocatalytic performanceZhang, Yue; Zhao, Yadi; Bao, Cunyou; Xiao, Yue; Xiang, Yuesong; Song, Mengting; Huang, Wenjuan; Ma, Liang; Hou, Huayi; Chen, XiangbaiJournal of Alloys and Compounds (2022), 909 (), 164731CODEN: JALCEU; ISSN:0925-8388. (Elsevier B.V.)With the rise of two-dimensional (2D) materials, layered metal thiophosphates (MPS3), as an important branch, have attracted much attention because of their excellent phys. and chem. properties. Cadmium phosphorus trisulfide (CdPS3), as a promising photocatalyst, can be used for hydrogen evolution and org. degrdn. Herein, high-quality 2D CdPS3 few layers are obtained by exfoliating bulk CdPS3 crystal. The optical properties of the as-obtained CdPS3 few layers show good light-harvesting ability, endowing it with excellent photocatalytic hydrogen evolution performance (10.88 mmol g-1 h-1) and high org. degrdn. performance (more than 92% within 30 min) under Xe lamp irradn. This research provides opportunities for the prepn. and application of other 2D MPS3 type materials in the field of photocatalysis.
- 48Cheng Z. Sendeku M. G. Liu Q. Layered Metal Phosphorous Trichalcogenides Nanosheets: Facile Synthesis and Photocatalytic Hydrogen Evolution Nanotechnology 2020 31 13 135405Google Scholar48Layered metal phosphorous trichalcogenides nanosheets: Facile synthesis and photocatalytic hydrogen evolutionCheng, Zhongzhou; Sendeku, Marshet Getaye; Liu, QuanlinNanotechnology (2020), 31 (13), 135405CODEN: NNOTER; ISSN:1361-6528. (IOP Publishing Ltd.)Layered transition metal phosphorous trichalcogenide (MPX3) materials have attracted immense attention due to their excellent optical and elec. properties. However, the controllable synthesis of ultrathin MPX3 nanosheets is still challenging. Here, we present a facile phosphosulfurization scheme to prep. high-quality layered FePS3 nanosheets, with ~ 20 nm in thickness. The optical properties of the as-obtained FePS3 show good light-harvesting ability, which endows it with excellent photocatalytic hydrogen evolution performance (402.4μmol g-1 h-1) under the simulated solar illumination. We further show that other MPX3 family members such as In2/3PS3 and CdPS3 can be also synthesized by the same approach. Our finding can offer a feasible approach for rationally designing other MPX3 materials for various applications.
- 49Chang X. Wang T. Gong J. CO2 Photo-reduction: Insights into CO2 Activation and Reaction on Surfaces of Photocatalysts Energy Environ. Sci. 2016 9 7 2177 2196Google Scholar49CO2 photo-reduction: insights into CO2 activation and reaction on surfaces of photocatalystsChang, Xiaoxia; Wang, Tuo; Gong, JinlongEnergy & Environmental Science (2016), 9 (7), 2177-2196CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)Large amts. of anthropogenic CO2 emissions assocd. with increased fossil fuel consumption have led to global warming and an energy crisis. The photocatalytic redn. of CO2 into solar fuels such as methane or methanol is believed to be one of the best methods to address these two problems. In addn. to light harvesting and charge sepn., the adsorption/activation and redn. of CO2 on the surface of heterogeneous catalysts remain a scientifically crit. challenge, which greatly limits the overall photoconversion efficiency and selectivity of CO2 redn. This review describes recent advances in the fundamental understanding of CO2 photoredn. on the surface of heterogeneous catalysts and particularly provides an overview of enhancing the adsorption/activation of CO2 mols. The reaction mechanism and pathways of CO2 redn. as well as their dependent factors are also analyzed and discussed, which is expected to enable an increase in the overall efficiency of CO2 redn. through minimizing the reaction barriers and controlling the selectivity towards the desired products. The challenges and perspectives of CO2 photoredn. over heterogeneous catalysts are presented as well.
- 50Gao W. Li S. He H. Li X. Cheng Z. Yang Y. Wang J. Shen Q. Wang X. Xiong Y. Zhou Y. Zou Z. Vacancy-Defect Modulated Pathway of Photoreduction of CO2 on Single Atomically Thin AgInP2S6 Sheets into Olefiant Gas Nat. Commun. 2021 12 1 4747Google Scholar50Vacancy-defect modulated pathway of photoreduction of CO2 on single atomically thin AgInP2S6 sheets into olefiant gasGao, Wa; Li, Shi; He, Huichao; Li, Xiaoning; Cheng, Zhenxiang; Yang, Yong; Wang, Jinlan; Shen, Qing; Wang, Xiaoyong; Xiong, Yujie; Zhou, Yong; Zou, ZhigangNature Communications (2021), 12 (1), 4747CODEN: NCAOBW; ISSN:2041-1723. (Nature Research)Abstr.: Artificial photosynthesis, light-driving CO2 conversion into hydrocarbon fuels, is a promising strategy to synchronously overcome global warming and energy-supply issues. The quaternary AgInP2S6 at. layer with the thickness of ∼ 0.70 nm were successfully synthesized through facile ultrasonic exfoliation of the corresponding bulk crystal. The sulfur defect engineering on this at. layer through a H2O2 etching treatment can excitingly change the CO2 photoredn. reaction pathway to steer dominant generation of ethene with the yield-based selectivity reaching ∼73% and the electron-based selectivity as high as ∼89%. Both DFT calcn. and in-situ FTIR spectra demonstrate that as the introduction of S vacancies in AgInP2S6 causes the charge accumulation on the Ag atoms near the S vacancies, the exposed Ag sites can thus effectively capture the forming *CO mols. It makes the catalyst surface enrich with key reaction intermediates to lower the C-C binding coupling barrier, which facilitates the prodn. of ethene.
- 51Fan Y. Song X. Ai H. Li W. Zhao M. Highly Efficient Photocatalytic CO2 Reduction in Two-Dimensional Ferroelectric CuInP2S6 Bilayers ACS Appl. Mater. Interfaces 2021 13 29 34486 34494Google Scholar51Highly efficient photocatalytic CO2 reduction in two-dimensional ferroelectric CuInP2S6 bilayersFan, Yingcai; Song, Xiaohan; Ai, Haoqiang; Li, Weifeng; Zhao, MingwenACS Applied Materials & Interfaces (2021), 13 (29), 34486-34494CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Photocatalytic CO2 conversion into reproducible chem. fuels (e.g., CO, CH3OH, or CH4) provides a promising scheme to solve the increasing environmental problems and energy demands simultaneously. However, the efficiency is severely restricted by the high overpotential of the CO2 redn. reaction (CO2RR) and rapid recombination of photoexcited carriers. Here, we propose that a novel type-II photocatalytic mechanism based on two-dimensional (2D) ferroelec. multilayers would be ideal for addressing these issues. Using d.-functional theory and nonadiabatic mol. dynamics calcns., we find that the ferroelec. CuInP2S6 bilayers exhibit a staggered band structure induced by the vertical intrinsic elec. fields. Different from the traditional type-II band alignment, the unique structure of the CuInP2S6 bilayer not only effectively suppresses the recombination of photogenerated electron-hole (e-h) pairs but also produces a sufficient photovoltage to drive the CO2RR. The predicted recombination time of photogenerated e-h pairs, 1.03 ns, is much longer than the transferring times of photoinduced electrons and holes, 5.45 and 0.27 ps, resp. Moreover, the overpotential of the CO2RR will decrease by substituting an S atom with a Cu atom, making the redox reaction proceed spontaneously under solar radiation. The solar-to-fuel efficiency with an upper limit of 8.40% is achieved in the CuInP2S6 bilayer and can be further improved to 32.57% for the CuInP2S6 five-layer. Our results indicate that this novel type-II photocatalytic mechanism would be a promising way to achieve highly efficient photocatalytic CO2 conversion based on the 2D ferroelec. multilayers.
- 52De Battisti, A.; Martínez-Huitle, C. A. , Electrocatalysis in Wastewater Treatment. In Electrochemical Water and Wastewater Treatment; Martínez-Huitle, C. A., Rodrigo, M. A., Scialdone, O. , Eds.; Butterworth-Heinemann: 2018; pp 119− 131.Google ScholarThere is no corresponding record for this reference.
- 53de Freitas Araujo K. C. da Silva D. R. dos Santos E. V. Varela H. Martinez-Huitle C. A. Investigation of Persulfate Production on BDD Anode by Understanding the Impact of Water Concentration J. Electroanal. Chem. 2020 860 113927Google ScholarThere is no corresponding record for this reference.
- 54Huang H. Li F. Xue Q. Zhang Y. Yin S. Chen Y. Salt-Templated Construction of Ultrathin Cobalt Doped Iron Thiophosphite Nanosheets toward Electrochemical Ammonia Synthesis Small 2019 15 51 1903500Google Scholar54Salt-Templated Construction of Ultrathin Cobalt Doped Iron Thiophosphite Nanosheets toward Electrochemical Ammonia SynthesisHuang, Hao; Li, Fumin; Xue, Qi; Zhang, Ying; Yin, Shiwei; Chen, YuSmall (2019), 15 (51), 1903500CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)Exploiting efficient electrocatalysts for electrochem. N redn. (NRR) is highly desired and deeply meaningful for realizing sustainable NH3 (NH3) prodn. under ambient conditions. The Fe protein contains one [Fe4S4] cluster and P cluster, which play an important role for transfer electron during the N fixing of nitrogenases. Based on the understanding of nitrogenase, the rising-star 2-dimensional Fe thiophosphite (FePS3) nanomaterials may be highly active electrocatalysts toward NRR due to the ideal elemental compn. 2D FePS3 nanosheets are successfully synthesized by a facile salt-templated method. The FePS3 nanosheets show better electrocatalytic NH3 yield and faradaic efficiency (FE) than Fe2S3, which demonstrates that the P element indeed improves the NRR activity of Fe-S. Theor., Co incorporation not only effectively prompts the cond. of FePS3, but also enhances the catalytic activities of Fe-edge sites. Exptl., Co-doped FePS3 (Co-FePS3) nanosheets exhibit a remarkable electrocatalytic performance toward NRR, such as high NH3 yield rate of 90.6μg h-1 mgcat-1, high FE of 3.38%, and an excellent long-term stability. Being the 1st theor. and exptl. report regarding FePS3-based electrocatalyst toward NRR, this work represents an important beginning to the family of metal thiophosphite as advanced electrocatalysts toward NRR.
- 55Mayorga-Martinez C. C. Sofer Z. Sedmidubsky D. Huber S. Eng A. Y. Pumera M. Layered Metal Thiophosphite Materials: Magnetic, Electrochemical, and Electronic Properties ACS Appl. Mater. Interfaces 2017 9 14 12563 12573Google Scholar55Layered Metal Thiophosphite Materials: Magnetic, Electrochemical, and Electronic PropertiesMayorga-Martinez, Carmen C.; Sofer, Zdenek; Sedmidubsky, David; Huber, Stepan; Eng, Alex Yong Sheng; Pumera, MartinACS Applied Materials & Interfaces (2017), 9 (14), 12563-12573CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Beyond graphene, transitional metal dichalcogenides, and black phosphorus, there are other layered materials called metal thiophosphites (MPSx), which are recently attracting the attention of scientists. Here we present the synthesis, structural and morphol. characterization, magnetic properties, electrochem. performance, and the calcd. d. of states of different layered metal thiophosphite materials with a general formula MPSx, and as a result of varying the metal component, we obtain CrPS4, MnPS3, FePS3, CoPS3, NiPS3, ZnPS3, CdPS3, GaPS4, SnPS3, and BiPS4. SnPS3, ZnPS3, CdPS3, GaPS4, and BiPS4 exhibit only diamagnetic behavior due to core electrons. By contrast, trisulfides with M = Mn, Fe, Co, and Ni, as well as CrPS4, are paramagnetic at high temps. and undergo a transition to antiferromagnetic state on cooling. Within the trisulfides series the Neel temp. characterizing the transition from paramagnetic to antiferromagnetic phase increases with the increasing at. no. and the orbital component enhancing the total effective magnetic moment. Interestingly, in terms of catalysis NiPS3, CoPS3, and BiPS4 show the highest efficiency for hydrogen evolution reaction (HER), while for the oxygen evolution reaction (OER) the highest performance is obsd. for CoPS3. Finally, MnPS3 presents the highest oxygen redn. reaction (ORR) activity compared to the other MPSx studied here. This great catalytic performance reported for these MPSx demonstrates their promising capabilities in energy applications.
- 56Yu Z. Peng J. Liu Y. Liu W. Liu H. Guo Y. Amine-Assisted Exfoliation and Electrical Conductivity Modulation Toward Few-layer FePS3 Nanosheets for Efficient Hydrogen Evolution J. Mater. Chem. A 2019 7 23 13928 13934Google Scholar56Amine-assisted exfoliation and electrical conductivity modulation toward few-layer FePS3 nanosheets for efficient hydrogen evolutionYu, Zhi; Peng, Jing; Liu, Yuhua; Liu, Wenxiu; Liu, Haifeng; Guo, YuqiaoJournal of Materials Chemistry A: Materials for Energy and Sustainability (2019), 7 (23), 13928-13934CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Layered metal phosphorus trichalcogenides (MPX3, M = transition metal, X = S or Se) have been established as new efficient catalysts for water splitting, with FePS3 as one representative example. Here, a novel amine-assisted exfoliation strategy is presented to obtain few-layer FePS3 nanosheets, whose intrinsic elec. cond. can be easily regulated by adjusting the reaction temp. The improved elec. cond. originates from the increase of the Fe3+/Fe2+ ratio, while matrix crystallinity also plays a vital role in this aspect. The combined effect of mixed valence and crystallinity results in superior HER performance with a decrease of the overpotential of 147 mV and a drop of the Tafel slope from 139-94 mV dec-1.
- 57Lian Q. Zhong L. Du C. Luo Y. Zhao J. Zheng Y. Xu J. Ma J. Liu C. Li S. Yan Q. Interfacing Epitaxial Dinickel Phosphide to 2D Nickel Thiophosphate Nanosheets for Boosting Electrocatalytic Water Splitting ACS Nano 2019 13 7 7975 7984Google ScholarThere is no corresponding record for this reference.
- 58Gusmão R. Sofer Z. Sedmidubský D. Huber Š. Pumera M. The Role of the Metal Element in Layered Metal Phosphorus Triselenides upon Their Electrochemical Sensing and Energy Applications ACS Catal. 2017 7 12 8159 8170Google Scholar58The Role of the Metal Element in Layered Metal Phosphorus Triselenides upon Their Electrochemical Sensing and Energy ApplicationsGusmao, Rui; Sofer, Zdenek; Sedmidubsky, David; Huber, Stepan; Pumera, MartinACS Catalysis (2017), 7 (12), 8159-8170CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)The no. of layered materials seems to be ever-growing, from mono- to multielement, with affiliates and applications being tested continuously. Chalcogenophosphites, also designated as metal phosphorus chalcogenides (MPXn), have attracted great interest because of not only their magnetic properties but also promising capabilities in energy applications. Herein, bulk crystals of different layered metal triselenophosphites, with a general formula MPSe3 (M = Cd, Cr, Fe, Mn, Sn, Zn), were synthesized. Structural and morphol. characterization was performed prior to testing their electrochem. performance. From the set of ternary layered materials, FePSe3, followed by MnPSe3, yielded the highest efficiency for the hydrogen evolution reaction (HER) both in acidic and alk. media with good stability after 100 cycles. MnPSe3 also holds the lowest oxidn. potential for cysteine, although this is due to the presence of MnO2 in the structure as detected by XPS. For the oxygen evolution reaction, the best performance was obsd. for FePSe3, although the stability of the material was not as good as in the case of HER. These findings have profound implications in the application of these layered ternary compds. in energy-related fields.
- 59Hao Y. Huang A. Han S. Huang H. Song J. Sun X. Wang Z. Li L. Hu F. Xue J. Peng S. Plasma-Treated Ultrathin Ternary FePSe3 Nanosheets as a Bifunctional Electrocatalyst for Efficient Zinc–Air Batteries ACS Appl. Mater. Interfaces 2020 12 26 29393 29403Google Scholar59Plasma-Treated Ultrathin Ternary FePSe3 Nanosheets as a Bifunctional Electrocatalyst for Efficient Zinc-Air BatteriesHao, Yanan; Huang, Aijian; Han, Silin; Huang, Hongjiao; Song, Junnan; Sun, Xiaoli; Wang, Zhiguo; Li, Linlin; Hu, Feng; Xue, Jianjun; Peng, ShengjieACS Applied Materials & Interfaces (2020), 12 (26), 29393-29403CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Developing novel bifunctional electrocatalysts with advanced oxygen electrocatalytic activity is pivotal for next-generation energy-storage devices. Herein, we present ultrathin oxygen-doped FePSe3 (FePSe3-O) nanosheets by Ar/O2 plasma treatment, with remarkable surface atom reorganization. Such surface atom reorganization generates multiple cryst.-amorphous interfaces that benefit the kinetics of oxygen evolution reaction, achieving a low overpotential of only 261 mV at 10 mA cm-2 with a small Tafel slope of 41.13 mV dec-1. D. functional theory calcn. indicates that oxygen doping can also modulate the elec. states at the Fermi level with a decreased band gap responsible for the enhanced electrocatalytic performance. Such unique FePSe3-O nanosheets can be further fabricated as the air cathode in rechargeable liq. zinc-air batteries (ZABs), which deliver a high open circuit potential of 1.47 V, a small charge-discharge voltage gap of 0.80 V, and good cycling stability for more than 800 circles. As a proof of concept, the flexible solid-state ZABs assembled with FePSe3-O nanosheets as cathode also display a favorable charge-discharge performance, durable stability, and good bendability. This work sheds new insights into the rational design of defect-rich ternary thiophosphate nanosheets by plasma treatment toward enhanced oxygen electrocatalysts in metal-air batteries.
- 60Konkena B. Masa J. Botz A. J. R. Sinev I. Xia W. Koßmann J. Drautz R. Muhler M. Schuhmann W. Metallic NiPS3@NiOOH Core–Shell Heterostructures as Highly Efficient and Stable Electrocatalyst for the Oxygen Evolution Reaction ACS Catal. 2017 7 1 229 237Google Scholar60Metallic NiPS3@NiOOH Core-Shell Heterostructures as Highly Efficient and Stable Electrocatalyst for the Oxygen Evolution ReactionKonkena, Bharathi; Masa, Justus; Botz, Alexander J. R.; Sinev, Ilya; Xia, Wei; Kossmann, Joerg; Drautz, Ralf; Muhler, Martin; Schuhmann, WolfgangACS Catalysis (2017), 7 (1), 229-237CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)We report metallic NiPS3@NiOOH core-shell heterostructures as an efficient and durable electrocatalyst for the oxygen evolution reaction, exhibiting a low onset potential of 1.48 V (vs. RHE) and stable performance for over 160 h. The atomically thin NiPS3 nanosheets are obtained by exfoliation of bulk NiPS3 in the presence of an ionic surfactant. The OER mechanism was studied by a combination of SECM, in situ Raman spectroscopy, SEM, and XPS measurements, which enabled direct observation of the formation of a NiPS3@NiOOH core-shell heterostructure at the electrode interface. Hence, the active form of the catalyst is represented as NiPS3@NiOOH core-shell structure. Moreover, DFT calcns. indicate an intrinsic metallic character of the NiPS3 nanosheets with densities of states (DOS) similar to the bulk material. The high OER activity of the NiPS3 nanosheets is attributed to a high d. of accessible active metallic-edge and defect sites due to structural disorder, a unique NiPS3@NiOOH core-shell heterostructure, where the presence of P and S modulates the surface electronic structure of Ni in NiPS3, thus providing excellent conductive pathway for efficient electron-transport to the NiOOH shell. These findings suggest that good size control during liq. exfoliation may be advantageously used for the formation of elec. conductive NiPS3@NiOOH core-shell electrode materials for the electrochem. water oxidn.
- 61Bushuyev O. S. De Luna P. Dinh C. T. Tao L. Saur G. van de Lagemaat J. Kelley S. O. Sargent E. H. What Should We Make with CO2 and How Can We Make It ? Joule 2018 2 5 825 832Google Scholar61What Should We Make with CO2 and How Can We Make It?Bushuyev, Oleksandr S.; De Luna, Phil; Dinh, Cao Thang; Tao, Ling; Saur, Genevieve; van de Lagemaat, Jao; Kelley, Shana O.; Sargent, Edward H.Joule (2018), 2 (5), 825-832CODEN: JOULBR; ISSN:2542-4351. (Cell Press)In this forward-looking Perspective, we discuss the current state of technol. and the economics of electrocatalytic transformation of CO2 into various chem. fuels. Our anal. finds that short-chain simple building-block mols. currently present the most economically compelling targets. Making an optimistic prediction of technol. advancement in the future, we propose the gradual rise of photocatalytic, CO2 polymn., biohybrid, and mol. machine technologies to augment and enhance already practical electrocatalytic CO2 conversion methods.
- 62Nitopi S. Bertheussen E. Scott S. B. Liu X. Y. Engstfeld A. K. Horch S. Seger B. Stephens I. E. L. Chan K. Hahn C. Norskov J. K. Jaramillo T. F. Chorkendorff I. Progress and Perspectives of Electrochemical CO2 Reduction on Copper in Aqueous Electrolyte Chem. Rev. 2019 119 12 7610 7672Google Scholar62Progress and Perspectives of Electrochemical CO2 Reduction on Copper in Aqueous ElectrolyteNitopi, Stephanie; Bertheussen, Erlend; Scott, Soren B.; Liu, Xinyan; Engstfeld, Albert K.; Horch, Sebastian; Seger, Brian; Stephens, Ifan E. L.; Chan, Karen; Hahn, Christopher; Noerskov, Jens K.; Jaramillo, Thomas F.; Chorkendorff, IbChemical Reviews (Washington, DC, United States) (2019), 119 (12), 7610-7672CODEN: CHREAY; ISSN:0009-2665. (American Chemical Society)A review. To date, copper is the only heterogeneous catalyst that has shown a propensity to produce valuable hydrocarbons and alcs., such as ethylene and ethanol, from electrochem. CO2 redn. (CO2R). There are variety of factors that impact CO2R activity and selectivity, including the catalyst surface structure, morphol., compn., the choice of electrolyte ions and pH, the electrochem. cell design, etc. Many of these factors are often intertwined, which can complicate catalyst discovery and design efforts. Here we take a broad and historical view of these different aspects and their complex interplay in CO2R catalysis on Cu, with the purpose of providing new insights, crit. evaluations, and guidance to the field with regards to research directions and best practices. First, we describe the various exptl. probes and complementary theor. methods that have been used to discern the mechanisms by which products are formed, and next we present our current understanding of the complex reaction networks for CO2R on Cu. Then we analyze two key methods that have been used in attempts to alter the activity and selectivity of Cu: nanostructuring and the formation of bimetallic electrodes. Finally, we offer some perspectives on the future outlook for electrochem. CO2R.
- 63Zhao K. Quan X. Carbon-Based Materials for Electrochemical Reduction of CO2 to C2+ Oxygenates: Recent Progress and Remaining Challenges ACS Catal. 2021 11 4 2076 2097Google Scholar63Carbon-Based Materials for Electrochemical Reduction of CO2 to C2+ Oxygenates: Recent Progress and Remaining ChallengesZhao, Kun; Quan, XieACS Catalysis (2021), 11 (4), 2076-2097CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)A review. Electrochem. redn. of CO2 to multicarbon (C2+) products is desirable because of the higher energy d. and economic value of C2+ products and the significant scientific issue for coupling of multicarbons. However, efficient conversion of CO2 into C2+ products remains challenging because of the difficulty in C-C coupling. Recently, numerous papers have reported carbon-based materials for C2+ products prodn. from CO2 electrochem. redn. Because of the unique properties of carbon-based materials in C2+ prodn., carbon-based materials can be used as a potential alternative for the electrocatalytic conversion of CO2. This Review summarized recent progresses in the formation of C2+ oxygenates from CO2 redn. on carbon-based materials. In this Review, we highlighted the strategies available for achieving C-C coupling on carbon-based electrocatalysts and revealed the relationships between intermediate adsorption energy and the selectivity of oxygenate prodn. from CO2 redn. Moreover, we provided the understandings for fabricating active sites of CO2 redn. on carbon-based materials and related mechanisms of C2+ oxygenate generation. The remaining challenges and opportunities for the electrochem. conversion of CO2 into C2+ oxygenates were discussed.
- 64Birdja Y. Y. Pérez-Gallent E. Figueiredo M. C. Göttle A. J. Calle-Vallejo F. Koper M. T. M. Advances and Challenges in Understanding the Electrocatalytic Conversion of Carbon Dioxide to Fuels Nat. Energy 2019 4 9 732 745Google Scholar64Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuelsBirdja, Yuvraj Y.; Perez-Gallent, Elena; Figueiredo, Marta C.; Gottle, Adrien J.; Calle-Vallejo, Federico; Koper, Marc T. M.Nature Energy (2019), 4 (9), 732-745CODEN: NEANFD; ISSN:2058-7546. (Nature Research)A review. The electrocatalytic redn. of carbon dioxide is a promising approach for storing (excess) renewable electricity as chem. energy in fuels. Here, we review recent advances and challenges in the understanding of electrochem. CO2 redn. We discuss existing models for the initial activation of CO2 on the electrocatalyst and their importance for understanding selectivity. Carbon-carbon bond formation is also a key mechanistic step in CO2 electroredn. to high-d. and high-value fuels. We show that both the initial CO2 activation and C-C bond formation are influenced by an intricate interplay between surface structure (both on the nano- and on the mesoscale), electrolyte effects (pH, buffer strength, ion effects) and mass transport conditions. This complex interplay is currently still far from being completely understood. In addn., we discuss recent progress in in situ spectroscopic techniques and computational techniques for mechanistic work. Finally, we identify some challenges in furthering our understanding of these themes.
- 65Ji L. Chang L. Zhang Y. Mou S. Wang T. Luo Y. Wang Z. Sun X. Electrocatalytic CO2 Reduction to Alcohols with High Selectivity over a Two-Dimensional Fe2P2S6 Nanosheet ACS Catal. 2019 9 11 9721 9725Google Scholar65Electrocatalytic CO2 Reduction to Alcohols with High Selectivity over a Two-Dimensional Fe2P2S6 NanosheetJi, Lei; Chang, Le; Zhang, Ya; Mou, Shiyong; Wang, Ting; Luo, Yonglan; Wang, Zhiming; Sun, XupingACS Catalysis (2019), 9 (11), 9721-9725CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)Electrochem. conversion of CO2 into alcs. provides an attractive path toward achieving a carbon-neutral cycle, while its efficiency is challenged by identifying active electrocatalysts for the CO2 redn. reaction (CO2RR). In this work, we report the Fe2P2S6 nanosheet acts as an efficient CO2RR electrocatalyst toward highly selective hydrogenation of CO2 to alcs. This catalyst is capable of achieving a high total Faradaic efficiency (FEmethanol+ethanol) of 88.3%, with a FEmethanol up to 65.2% at -0.20 V vs the reversible hydrogen electrode in 0.5 M KHCO3 soln., much higher than most reported CO2RR electrocatalysts. D. functional theory calcns. indicate that Fe atom on the Fe2P2S6 surface can be regared as the active site for alc. formation.
- 66Thompson A. H. Whittingham M. S. Transition Metal Phosphorus Trisulfides as Battery Cathodes Mater. Res. Bull. 1977 12 7 741 744Google Scholar66Transition metal phosphorus trisulfides as battery cathodesThompson, A. H.; Whittingham, M. S.Materials Research Bulletin (1977), 12 (7), 741-4CODEN: MRBUAC; ISSN:0025-5408.The layered, transition-metal P trisulfides exhibit electrochem. activity. NiPS3 reacts with >4 Li resulting in a cell with a theor. energy double that of TiS2.
- 67Brec R. Review on structural and chemical properties of transition metal phosphorous trisulfides MPS3 Solid State Ion. 1986 22 1 3 30Google ScholarThere is no corresponding record for this reference.
- 68Foot P. J. S. Katz T. Patel S. N. Nevett B. A. Pieecy A. R. Balchin A. A. The Structures and Conduction Mechanisms of Lithium-intercalated and Lithium-Substituted Nickel Phosphorus Trisulphide (NiPS3), and the Use of the Material as a Secondary Battery Electrode Phys. Status Solidi A 1987 100 1 11 29Google Scholar68The structures and conduction mechanisms of lithium-intercalated and lithium-substituted nickel phosphorus trisulfide (NiPS3), and the use of the material as a secondary battery electrodeFoot, P. J. S.; Katz, T.; Patel, S. N.; Nevett, B. A.; Piercy, A. R.; Balchin, A. A.Physica Status Solidi A: Applied Research (1987), 100 (1), 11-29CODEN: PSSABA; ISSN:0031-8965.A review with 35 refs. The crystal structure and conduction of the title compds. are discussed and the possible use of these NiPS3 compds. as battery electrodes is suggested. An app. is given for the electrochem. lithiation of NiPS3 to form LixNiPS3.
- 69Rouxel J. Brec R. Low-Dimensional Chalcogenides as Secondary Cathodic Materials: Some Geometric and Electronic Aspects Annu. Rev. Mater. Sci. 1986 16 1 137 162Google Scholar69Low-dimensional chalcogenides as secondary cathodic materials: some geometric and electronic aspectsRouxel, J.; Brec, R.Annual Review of Materials Science (1986), 16 (), 137-62CODEN: ARMSCX; ISSN:0084-6600.A review with 45 refs. on electronic and ionic cond. and reversibility of the title cathodic materials for batteries. The geometric aspects assocd. with intercalation (parameter expansion, long distance, and local structures), electronic transfer, and phase transitions are analyzed.
- 70Li X. Wu X. Yang J. Half-Metallicity in MnPSe3 Exfoliated Nanosheet with Carrier Doping J. Am. Chem. Soc. 2014 136 31 11065 11069Google Scholar70Half-Metallicity in MnPSe3 Exfoliated Nanosheet with Carrier DopingLi, Xingxing; Wu, Xiaojun; Yang, JinlongJournal of the American Chemical Society (2014), 136 (31), 11065-11069CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)Searching two-dimensional (2D) half-metallic crystals that are feasible in expt. is essential to develop next-generation nanospintronic devices. Here, a 2-dimensional exfoliated MnPSe3 nanosheet with novel magnetism is 1st proposed based on 1st-principles calcns. In particular, the evaluated low cleavage energy and high in-plane stiffness indicate that the free-standing MnPSe3 nanosheet can be exfoliated from its bulk structure in expt. The MnPSe3 nanosheet is an antiferromagnetic semiconductor at its ground state, whereas both electron and hole doping induce its transition from antiferromagnetic semiconductor to ferromagnetic half-metal. Also, the spin-polarization directions of 2-dimensional half-metallic MnPSe3 are opposite for electron and hole doping, which can be controlled by applying an external voltage gate. The Monte Carlo simulation based on the Ising model suggests the Curie temp. of the doped 2-dimensional MnPSe3 crystal is up to 206 K. These advantages render the 2-dimensional MnPSe3 crystal with great potentials for application in elec.-field controlled spintronic devices.
- 71Cabria I. El-Meligi A. A. DFT Simulation of Hydrogen Storage on Manganese Phosphorous Trisulphide (MnPS3) Int. J. Hydrogen Energy 2018 43 11 5903 5912Google Scholar71DFT simulation of hydrogen storage on manganese phosphorous trisulfide (MnPS3)Cabria, I.; El-Meligi, A. A.International Journal of Hydrogen Energy (2018), 43 (11), 5903-5912CODEN: IJHEDX; ISSN:0360-3199. (Elsevier Ltd.)Manganese phosphorous trisulfide, MnPS3, is a solid layered material. The H2 gravimetric storage capacity of powd. MnPS3 at 80.15, 173.15, and 298.15° K and at moderate pressures was recently exptl. measured. The origin of storage capacity of this material is not well understood. The main hypothesis is that H2 is stored in powd. MnPS3 pores. Pores were modeled as two parallel MnPS3 layers sepd. by a certain distance. D. functional theory (DFT) simulations of the H2 interaction with the MnPS3 layer surface were performed to test this hypothesis. Simulations indicated H2 adsorption on the MnPS3 layer surface was energetically favorable, but only by a physisorption mechanism. Gravimetric capacity calcns. of powd. MnPS3 pores were also performed and were in reasonable agreement with exptl. results. A comparison of calcd. and exptl. gravimetric capacity showed that H2 storage on powd. MnPS3 was mainly due to in-pore compression; the contribution of the physisorption process to storage was very small.
- 72Kuzminskii Y. V. Voronin B. M. Redin N. N. Iron and Nickel Phosphorus Trisulfides as Electroactive Materials for Primary Lithium Batteries J. Power Sources 1995 55 2 133 141Google Scholar72Iron and nickel phosphorus trisulfides as electroactive materials for primary lithium batteriesKuzminskii, Y. V.; Voronin, B. M.; Redin, N. N.Journal of Power Sources (1995), 55 (2), 133-41CODEN: JPSODZ; ISSN:0378-7753. (Elsevier)The theor. specific capacities of a family of layered compds. MPX3 (M = Fe, Co, Ni; X = S, Se) for current-producing reactions involving 1.5, 2, 6 and 9 lithium atoms per MPX3 mol. have been estd. These data show that FePS3 and NiPS3 are good electroactive materials for primary lithium cells. Conditions which allow one to shorten substantially the synthesis time of iron and nickel phosphorus trisulfides in ampules are presented. According to cond. measurements in the 293-673 K temp. range, NiPS3 is an intrinsic and FePS3 an extrinsic semiconductor. The discharge characteristics of Li/MPS3 (M = Fe, Ni) cells with an org. electrolyte have been studied; they confirmed that the participation of nine electrons in the redox process is possible. For a primary button cell of the std. size 2325, with a FePS3 cathode, the specific capacity and specific energy values obtained in a discharge 1.8-1.2 V range at load resistances of 3.0 to 30 kΩ were 500-1160 Ah/kg and 700-1770 Wh/kg (on a pure FePS3 basis), resp.
- 73Kuzminskii Y. V. Voronin B. M. Petrushina I. M. Redin N. N. Prikhodko G. P. Nickel Phosphorus Trisulfide: An Electroactive Material for Medium-Temperature Lithium Batteries J. Power Sources 1995 55 1 1 6Google ScholarThere is no corresponding record for this reference.
- 74Liang Q. Zheng Y. Du C. Luo Y. Zhang J. Li B. Zong Y. Yan Q. General and Scalable Solid-State Synthesis of 2D MPS3 (M = Fe, Co, Ni) Nanosheets and Tuning Their Li/Na Storage Properties Small Methods 2017 1 12 1700304Google ScholarThere is no corresponding record for this reference.
- 75Fujii Y. Miura A. Rosero-Navarro N. C. Higuchi M. Tadanaga K. FePS3 Electrodes in All-Solid-State Lithium Secondary Batteries Using Sulfide-Based Solid Electrolytes Electrochim. Acta 2017 241 370 374Google Scholar75FePS3 electrodes in all-solid-state lithium secondary batteries using sulfide-based solid electrolytesFujii, Yuta; Miura, Akira; Rosero-Navarro, Nataly Carolina; Higuchi, Mikio; Tadanaga, KiyoharuElectrochimica Acta (2017), 241 (), 370-374CODEN: ELCAAV; ISSN:0013-4686. (Elsevier Ltd.)In bulk-type all-solid-state Li batteries with sulfide-based solid electrolytes, composite electrodes, in which an active material, a solid electrolyte, and a conductive additive are mixed, were used to enhance Li-ion diffusion and electronic cond. However, the addn. of electrolytes and electron-conductive additives to the composite electrodes decreases the amt. of active materials in the batteries. FePS3 was employed as the electrode in an all-solid-state Li secondary battery, without mixing the solid electrolytes and conductive additives. The all-solid-state cell using the FePS3 electrode exhibited reversible charge-discharge behavior for >30 cycles under a const. c.d. of 0.13 mA cm-2 at room temp. The discharge capacity of the cell was 107 mAh g-1 at the 30th cycle. This behavior was comparable to that of a cell with an electrode including a solid electrolyte and/or a conductive additive, indicating that the FePS3 electrode had sufficient paths of Li ions and of electron conduction. Probably FePS3 is an attractive Fe-based electrode for an all-solid-state battery using a sulfide-based solid electrolyte.
- 76Liang Q. Zheng Y. Du C. Luo Y. Zhao J. Ren H. Xu J. Yan Q. Asymmetric-Layered Tin Thiophosphate: An Emerging 2D Ternary Anode for High-Performance Sodium Ion Full Cell ACS Nano 2018 12 12 12902 12911Google Scholar76Asymmetric-Layered Tin Thiophosphate: An Emerging 2D Ternary Anode for High-Performance Sodium Ion Full CellLiang, Qinghua; Zheng, Yun; Du, Chengfeng; Luo, Yubo; Zhao, Jin; Ren, Hao; Xu, Jianwei; Yan, QingyuACS Nano (2018), 12 (12), 12902-12911CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)The emerging Na ion batteries (SIBs) are believed to be prospective substitutes for Li ion batteries (LIBs) because of the wide distribution of Na resources. However, to compensate for the sluggish reaction kinetics and higher intrinsic potential of Na+ compared to Li+, cost-effective, reliable, and sustainable electrode materials must be explored for practical applications. Here, 2D ternary tin thiophosphate (SnP2S6) nanosheets (∼10 nm thickness) grown on graphene (denoted as SPS/G hybrid) are demonstrated as intriguing anodes for SIBs. The asym.-layered structure and ternary compn. enable the SPS/G hybrid with a high reversible capacity (1230 mAh/g at 50 mA/g), superior rate capability (200 mAh/g at 15 A/g), and an exceptional capacity retention of 76% after 1000 cycles at 2.0 A/g. A prototype Na-ion full cell constructed by pairing with the Na3V2O2(PO4)3F cathode affords a high capacity of 470 mAh/g at 30 mA/g (on the basis of anode wt.) and good cyclic capacity of 360 mAh/g at 150 mA/g. Such 2D ternary chalcogenides with low-cost elements are promising materials for superior SIBs.
- 77Brec, R. , Review on Structural and Chemical Properties of Transition Metal Phosphorus Trisulfides MPS3. In Intercalation in Layered Materials; Dresselhaus, M. S. , Ed.; Springer US: Boston, MA, 1986; pp 93− 124.Google ScholarThere is no corresponding record for this reference.
- 78Clement R. P. Garnier O. Jegoudez J. Coordination Chemistry of the Lamellar MPS3 Materials: Metal-Ligand Cleavage as the Source of an Unusual Cation-Transfer Intercalation Process Inorg. Chem. 1986 25 1404 1409Google Scholar78Coordination chemistry of the lamellar MPS3 materials: metal-ligand cleavage as the source of an unusual "cation-transfer" intercalation processClement, Rene; Garnier, Odile; Jegoudez, JocelyneInorganic Chemistry (1986), 25 (9), 1404-9CODEN: INOCAJ; ISSN:0020-1669.Several MPS3 lamellar materials exhibit a very unusual intercalation chem. based on a cation-transfer process between the solid and a solvent. This contrasts with the electron-transfer processes that governs the intercalation chem. of the layered dichalcogenides MX2. Although the MPS3 layers are made up of infinite arrays of P2S64- units coordinated to the M2+ cations, the metal-ligand (M-S) bonding still exhibits enough lability to allow the M2+ cations to jump, under very mild conditions, from intra- toward interlamellar sites (or the opposite) and even to be removed from the material, provided a suitable cation is available to intercalate the lattice and maintain elec. neutrality. Depending on the metal, the solvent, and the guest species, the reaction may take place spontaneously or require an assist, usually by complexing the leaving M2+ cations (EDTA). Small inserted ions (Na+, K+, etc.) are strongly solvated and mobile, and they can in turn be exchanged. To rationalize this behavior, it is suggested that the intercalation chem. of the MPS3 involves a chem. transformation of the whole of the material, rather than a diffusional process of the guest species within the host lattice. The MPS3 layered materials are considered as polynuclear complexes that can undergo heterogeneous equil. with their constitutive species M2+ and P2S64- and other species present in soln. In support of this mechanism, MPS3 materials were successfully prepd. by mixing aq. solns. of Na4P2S6 and transition-metal salts. Some implications of this mechanism are discussed.
- 79Wang F. He J. Speeding Protons with Metal Vacancies Science 2020 370 6516 525 526Google Scholar79Speeding protons with metal vacanciesWang, Fengmei; He, JunScience (Washington, DC, United States) (2020), 370 (6516), 525-526CODEN: SCIEAS; ISSN:1095-9203. (American Association for the Advancement of Science)Proton exchange membranes (PEMs) find applications not only in fuel cells and sensors but as chem. filters and in biol. systems. In particular, the Nafion (Chemours Company) PEM, which consists of sulfonated tetrafluoroethylene-based fluoropolymer-copolymer, is widely used in practical electrochem. processes. It has a high proton cond. up to 0.2 S/cm below 80°C and high relative humidity (RH) (>93%), but its cond. drops severely at higher temps. or at low RH (below 50%). On page 596 of this issue, Qian et al. report a new class of proton membranes assembled from two-dimensional (2D) layered transition-metal phosphorus trichalcogenide (TMPTC) nanosheets in which metal vacancies boost ion cond. These membranes exhibit a proton cond. of ∼0.95 S/cm at 90°C and 98% RH but still have a cond. of 0.26 S/cm even at 60% RH.
Figure 1
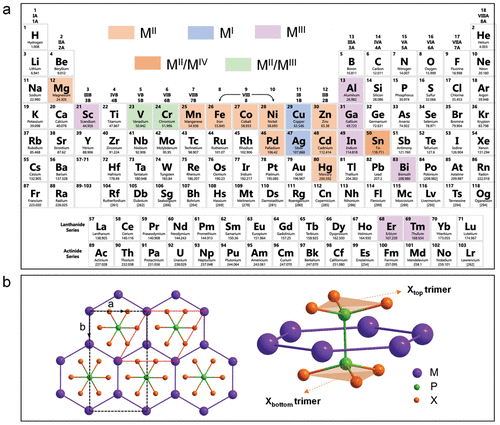 Figure 1. (a) The metal atoms and their valence states constituted in MPX3 crystals across the periodic table of elements. Filled blocks represent elements for which the layered MPX3 structure has been reported. (b) Schematic structure of MPX3 with [P2X6]4- bipyramids enclosing the metal atoms. The X layers are distinguished through Xtop and Xbottom.
Figure 1. (a) The metal atoms and their valence states constituted in MPX3 crystals across the periodic table of elements. Filled blocks represent elements for which the layered MPX3 structure has been reported. (b) Schematic structure of MPX3 with [P2X6]4- bipyramids enclosing the metal atoms. The X layers are distinguished through Xtop and Xbottom.Figure 2
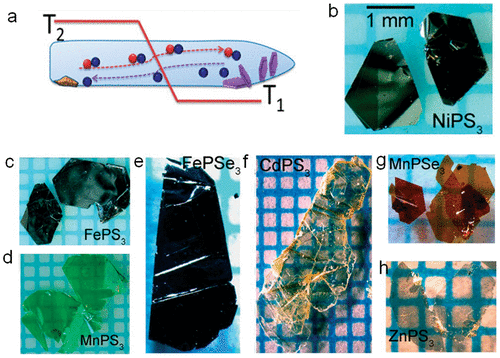 Figure 2. (a) Schematic illustration of the CVT approach. (b-h) the digital images for the resulting MPX3 crystal.Reproduced with permission from ref ( 19). Copyright 2016, American Chemical Society.
Figure 2. (a) Schematic illustration of the CVT approach. (b-h) the digital images for the resulting MPX3 crystal.Reproduced with permission from ref ( 19). Copyright 2016, American Chemical Society.Figure 3
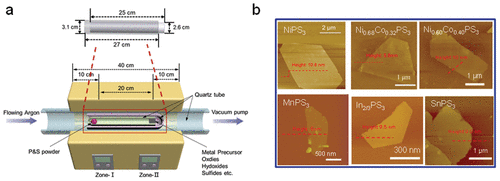
Figure 4
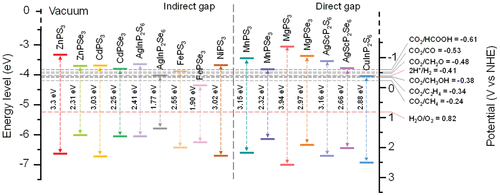
Figure 5
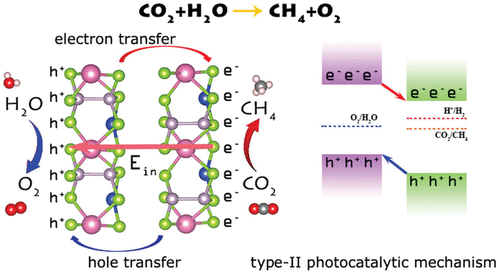 Figure 5. Photocatalytic CO2 reduction on bilayer CuInP2S6 with schematic type-II band diagram.Reproduced with permission from ref ( 51). Copyright 2021, American Chemical Society.
Figure 5. Photocatalytic CO2 reduction on bilayer CuInP2S6 with schematic type-II band diagram.Reproduced with permission from ref ( 51). Copyright 2021, American Chemical Society.Figure 6
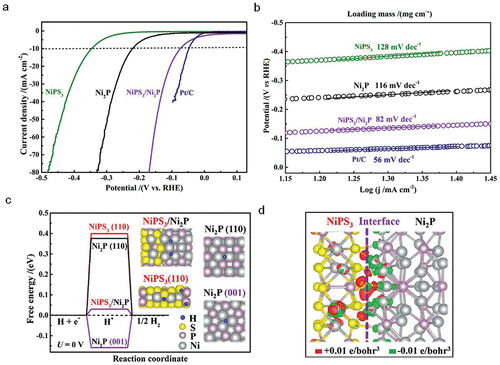 Figure 6. Electrocatalytic HER activity on NiPS3/Ni2P electrode. a-b) Comparison of the linear sweep voltammetry profiles (a) and the corresponding Tafel plots (b) for the as-grown NiPS3, Ni2P, NiPS3/Ni2P electrodes. c) the Gibbs energy profile for H adsorption (ΔGH*) obtained at the equilibrium potential (U = 0 V) for the as-grown NiPS3/Ni2P, Ni2P (001), Ni2P (110), and NiPS3 (110). d) Charge density difference at the heterointerfaces between NiPS3 and Ni2P. The electron accumulation and depletion are marked with red and green, respectively. The grey, yellow, purple, and blue colors represent Ni, S, P, and H atoms, which are marked in, respectively.Reproduced with permission from ref ( 57). Copyright 2019, American Chemical Society.
Figure 6. Electrocatalytic HER activity on NiPS3/Ni2P electrode. a-b) Comparison of the linear sweep voltammetry profiles (a) and the corresponding Tafel plots (b) for the as-grown NiPS3, Ni2P, NiPS3/Ni2P electrodes. c) the Gibbs energy profile for H adsorption (ΔGH*) obtained at the equilibrium potential (U = 0 V) for the as-grown NiPS3/Ni2P, Ni2P (001), Ni2P (110), and NiPS3 (110). d) Charge density difference at the heterointerfaces between NiPS3 and Ni2P. The electron accumulation and depletion are marked with red and green, respectively. The grey, yellow, purple, and blue colors represent Ni, S, P, and H atoms, which are marked in, respectively.Reproduced with permission from ref ( 57). Copyright 2019, American Chemical Society.Figure 7
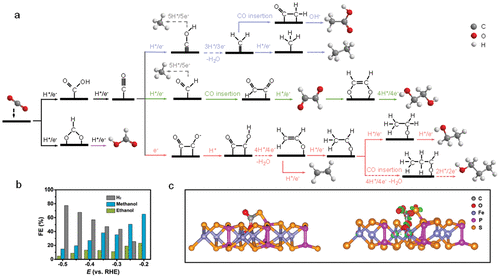 Figure 7. (a) Overview of the primary pathways for CO2RR towards different products. b-c) Electrocatalytic CO2RR for different products at different potentials over Fe2P2S6 nanosheets (b), and optimized adsorption of CO2 molecular over Fe2P2S6 surface (c), in which red and blue regions represent positive and negative charges, respectively.Reproduced with permission from ref ( 65). Copyright 2019, American Chemical Society.
Figure 7. (a) Overview of the primary pathways for CO2RR towards different products. b-c) Electrocatalytic CO2RR for different products at different potentials over Fe2P2S6 nanosheets (b), and optimized adsorption of CO2 molecular over Fe2P2S6 surface (c), in which red and blue regions represent positive and negative charges, respectively.Reproduced with permission from ref ( 65). Copyright 2019, American Chemical Society.Figure 8
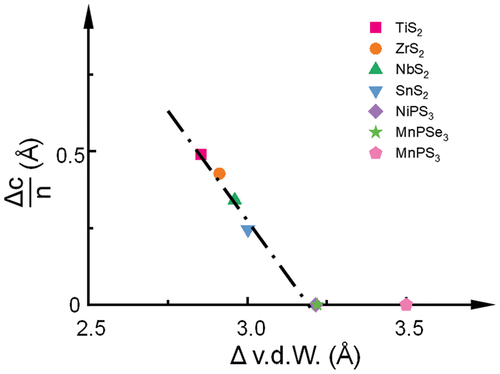
Figure 9
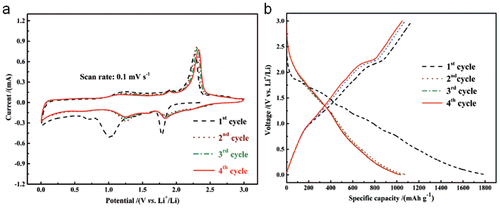 Figure 9. Electrochemical performance of NiPS3 nanosheet electrode used for Li-ion battery. (a) Cyclic voltammetric curves for the first four cycles at 0.1 mV/s. (b) The representative galvanostatic charge-discharge profiles for the four cycles at 0.05 A/g.Reproduced with permission from ref ( 74). Copyright 2017, Wiley-VCH.
Figure 9. Electrochemical performance of NiPS3 nanosheet electrode used for Li-ion battery. (a) Cyclic voltammetric curves for the first four cycles at 0.1 mV/s. (b) The representative galvanostatic charge-discharge profiles for the four cycles at 0.05 A/g.Reproduced with permission from ref ( 74). Copyright 2017, Wiley-VCH.Figure 10
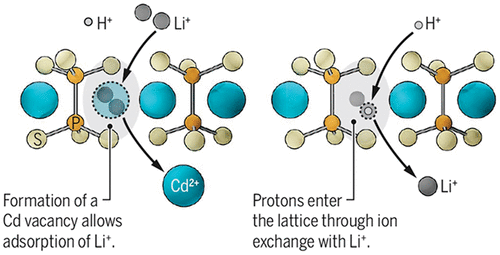 Figure 10. Ion-exchange property in layered CdPS3 material as proton exchange membrane.Reproduced with permission from ref ( 79). Copyright 2020, Science.
Figure 10. Ion-exchange property in layered CdPS3 material as proton exchange membrane.Reproduced with permission from ref ( 79). Copyright 2020, Science.References
CHAPTER SECTIONSThis chapter references 79 other publications.
- 1Chittari B. L. Park Y. Lee D. Han M. MacDonald A. H. Hwang E. Jung J. Electronic and Magnetic Properties of Single-Layer MPX3 Metal Phosphorous Trichalcogenides Phys. Rev. B 2016 94 18 1844281Electronic and magnetic properties of single-layer MPX3 metal phosphorous trichalcogenidesChittari, Bheema Lingam; Park, Youngju; Lee, Dongkyu; Han, Moonsup; MacDonald, Allan H.; Hwang, Euyheon; Jung, JeilPhysical Review B (2016), 94 (18), 184428/1-184428/16CODEN: PRBHB7; ISSN:2469-9950. (American Physical Society)We survey the electronic structure and magnetic properties of two-dimensional (2D) MPX3 (M=V,Cr,Mn,Fe,Co,Ni,Cu,Zn, and X=S,Se,Te) transition-metal chalcogenophosphates to shed light on their potential role as single-layer van der Waals materials that possess magnetic order. Our ab initio calcns. predict that most of these single-layer materials are antiferromagnetic semiconductors. The band gaps of the antiferromagnetic states decrease as the at. no. of the chalcogen atom increases (from S to Se to Te), leading in some cases to half-metallic ferromagnetic states or to nonmagnetic metallic states. We find that the competition between antiferromagnetic and ferromagnetic states can be substantially influenced by gating and by strain engineering. The sensitive interdependence we find between magnetic, structural, and electronic properties establishes the potential of this 2D materials class for applications in spintronics.
- 2Susner M. A. Chyasnavichyus M. McGuire M. A. Ganesh P. Maksymovych P. Metal Thio- and Selenophosphates as Multifunctional van der Waals Layered Materials Adv. Mater. 2017 29 38 1602852There is no corresponding record for this reference.
- 3Samal R. Sanyal G. Chakraborty B. Rout C. S. Two-Dimensional Transition Metal Phosphorous Trichalcogenides (MPX3): A Review on Emerging Trends, Current State and Future Perspectives J. Mater. Chem. A 2021 9 5 2560 25913Two-dimensional transition metal phosphorous trichalcogenides (MPX3): a review on emerging trends, current state and future perspectivesSamal, Rutuparna; Sanyal, Gopal; Chakraborty, Brahmananda; Rout, Chandra SekharJournal of Materials Chemistry A: Materials for Energy and Sustainability (2021), 9 (5), 2560-2591CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)A review. Profound scientific attention has been devoted to the expansive family of functional 2D materials soon after the isolation of graphene and the discovery of Dirac physics. Of particular note, the re-emergence of layer architecture metal thio(seleno) phosphate materials (MPX3, X = S, Se) materials with appreciable tunability in dimension and proximity on correlated ground states opened novel potential avenues for the ultimate theor. (structural, magnetic) and realistic research. The wide spanned bandgap of 1.2-3.5 eV harnessed multifarious fundamental magnetic, electronic, ferroelec., optical, intercalation properties and their prominent implementation in a plethora of applications, such as Li-ion batteries, field-effect transistors, visible and UV photodetectors, photo-electrochem. reactions and hydrogen (H2) storage. This review examines many of the scarcely touched aspects, such as multiple Dirac cones, vibrational properties, and responses in the presence of strain of MPX3 materials. In addn., the latest advancement in the emergent applications, physiochem. properties and assorted growth methodologies considering the palpable vol. of research has been explored in detail in order to bring across a more complete discussion and outlook. We strongly believe that this review will be very handy for researchers working in various branches of science and engineering, such as physics, chem., materials science, and chem. engineering.
- 4Wang F. Shifa T. A. Yu P. He P. Liu Y. Wang F. Wang Z. Zhan X. Lou X. Xia F. He J. New Frontiers on van der Waals Layered Metal Phosphorous Trichalcogenides Adv. Funct. Mater. 2018 28 37 1802151There is no corresponding record for this reference.
- 5Friedel M. C. Soufre et ses composés—sur une nouvelle série de sulfophosphures, les thiohypophosphates CR l’Academie. Sci. Ser 1894 3 119 260There is no corresponding record for this reference.
- 6Ouvrard G. Brec R. Rouxel J. Structural determination of some MPS3 layered phases (M = Mn, Fe, Co, Ni and Cd) Mater. Res. Bull. 1985 20 1181 11896Structural determination of some MPS3 layered phases (M = Mn, Fe, Co, Ni and Cd)Ouvrard, G.; Brec, R.; Rouxel, J.Materials Research Bulletin (1985), 20 (10), 1181-9CODEN: MRBUAC; ISSN:0025-5408.The structures of most of the MPS3 phases (M = Mn, Fe, Co, Ni, Cd) were detd. from single-crystal anal. They show the expected CdCl2 structural type, some weak disorder on the cationic sites being detected on NiPS3 and CoPS3. Fair stoichiometry is inferred for the phases since no interslab cation could be seen in the Van der Waals' gap of the structures. Polytypism may occur in NiPS3. At. parameters are given.
- 7Hwangbo K. Zhang Q. Jiang Q. Wang Y. Fonseca J. Wang C. Diederich G. M. Gamelin D. R. Xiao D. Chu J. H. Yao W. Xu X. Highly Anisotropic Excitons and Multiple Phonon Bound States in a van der Waals Antiferromagnetic Insulator Nat. Nanotechnol. 2021 16 6 655 6607Highly anisotropic excitons and multiple phonon bound states in a van der Waals antiferromagnetic insulatorHwangbo, Kyle; Zhang, Qi; Jiang, Qianni; Wang, Yong; Fonseca, Jordan; Wang, Chong; Diederich, Geoffrey M.; Gamelin, Daniel R.; Xiao, Di; Chu, Jiun-Haw; Yao, Wang; Xu, XiaodongNature Nanotechnology (2021), 16 (6), 655-660CODEN: NNAABX; ISSN:1748-3387. (Nature Portfolio)Two-dimensional (2D) semiconductors enable the investigation of light-matter interactions in low dimensions1,2. Yet, the study of elementary photoexcitations in 2D semiconductors with intrinsic magnetic order remains a challenge due to the lack of suitable materials3,4. Here, we report the observation of excitons coupled to zigzag antiferromagnetic order in the layered antiferromagnetic insulator NiPS3. The exciton exhibits a narrow photoluminescence linewidth of roughly 350μeV with near-unity linear polarization. When we reduce the sample thickness from five to two layers, the photoluminescence is suppressed and eventually vanishes for the monolayer. This suppression is consistent with the calcd. bandgap of NiPS3, which is highly indirect for both the bilayer and the monolayer5. Furthermore, we observe strong linear dichroism (LD) over a broad spectral range. The optical anisotropy axes of LD and of photoluminescence are locked to the zigzag direction. Furthermore, their temp. dependence is reminiscent of the in-plane magnetic susceptibility anisotropy. Hence, our results indicate that LD and photoluminescence could probe the symmetry breaking magnetic order parameter of 2D magnetic materials. In addn., we observe over ten exciton-A1g-phonon bound states on the high-energy side of the exciton resonance, which we interpret as signs of a strong modulation of the ligand-to-metal charge-transfer energy by electron-lattice interactions. Our work establishes NiPS3 as a 2D platform for exploring magneto-exciton physics with strong correlations.
- 8Kang S. Kim K. Kim B. H. Kim J. Sim K. I. Lee J. U. Lee S. Park K. Yun S. Kim T. Nag A. Walters A. Garcia-Fernandez M. Li J. Chapon L. Zhou K. J. Son Y. W. Kim J. H. Cheong H. Park J. G. Coherent Many-Body Exciton in van der Waals Antiferromagnet NiPS3 Nature 2020 583 7818 785 7898Coherent many-body exciton in van der Waals antiferromagnet NiPS3Kang, Soonmin; Kim, Kangwon; Kim, Beom Hyun; Kim, Jonghyeon; Sim, Kyung Ik; Lee, Jae-Ung; Lee, Sungmin; Park, Kisoo; Yun, Seokhwan; Kim, Taehun; Nag, Abhishek; Walters, Andrew; Garcia-Fernandez, Mirian; Li, Jiemin; Chapon, Laurent; Zhou, Ke-Jin; Son, Young-Woo; Kim, Jae Hoon; Cheong, Hyeonsik; Park, Je-GeunNature (London, United Kingdom) (2020), 583 (7818), 785-789CODEN: NATUAS; ISSN:0028-0836. (Nature Research)Abstr.: An exciton is the bosonic quasiparticle of electron-hole pairs bound by the Coulomb interaction. Bose-Einstein condensation of this exciton state has long been the subject of speculation in various model systems2,3, and examples have been found more recently in optical lattices and two-dimensional materials4-9. Unlike these conventional excitons formed from extended Bloch states4-9, excitonic bound states from intrinsically many-body localized states are rare. Here we show that a spin-orbit-entangled exciton state appears below the Neel temp. of 150 K in NiPS3, an antiferromagnetic van der Waals material. It arises intrinsically from the archetypal many-body states of the Zhang-Rice singlet10,11, and reaches a coherent state assisted by the antiferromagnetic order. Using configuration-interaction theory, we det. the origin of the coherent excitonic excitation to be a transition from a Zhang-Rice triplet to a Zhang-Rice singlet. We combine three spectroscopic tools-resonant inelastic X-ray scattering, photoluminescence and optical absorption-to characterize the exciton and to demonstrate an extremely narrow excitonic linewidth below 50 K. The discovery of the spin-orbit-entangled exciton in antiferromagnetic NiPS3 introduces van der Waals magnets as a platform to study coherent many-body excitons.
- 9Joy P. A. Vasudevan S. Magnetism in the Layered Transition-Metal Thiophosphates MPS3 (M=Mn, Fe, and Ni) Phys. Rev. B 1992 46 9 5425 54339Magnetism in the layered transition-metal thiophosphates MPS3 (M = manganese, iron, and nickel)Joy, P. A.; Vasudevan, S.Physical Review B: Condensed Matter and Materials Physics (1992), 46 (9), 5425-33CODEN: PRBMDO; ISSN:0163-1829.Anisotropic magnetic susceptibilities of single crystals of the layered transition-metal thiophosphates MnPS3, FePS3, and NiPS3 have been measured as a function of temp. The materials order antiferromagnetically at low temps., the Neel temps. being 78, 123, and 155 K, resp. In the ordered state, the magnetization axis lies perpendicular to the layers of MnPS3 and FePS3, while for NiPS3 it lies in the layer. In the paramagnetic regime, the anisotropies of these compds. are different; while the susceptibility for MnPS3 is isotropic and that for NiPS3 shows only a weak anisotropy, FePS3 exhibits highly anisotropic susceptibility. The anisotropic susceptibilities have been analyzed to obtain information on the state of the magnetic ions and the nature of magnetic interactions between them. The results show that MnPS3, FePS3, and NiPS3 form a unique class of compds. Although all three compds. are isostructural with the magnetic lattice being the two-dimensional honeycomb, the spin dimensionalities for the three are different. While MnPS3 is best described by the isotropic Heisenberg Hamiltonian, FePS3 is most effectively treated by the Ising model and NiPS3 by the anisotropic Heisenberg Hamiltonian. The origin of the anisotropy in these compds. has been discussed, and it is shown how it arises from a combination of spin-orbit coupling and the trigonal distortion of the MS6 octahedra. The magnetic exchange const., J and the zero-field splitting energies of the ground state of the transition-metal ion have been evaluated from the anisotropic paramagnetic susceptibilities.
- 10Wildes A. R. Simonet V. Ressouche E. McIntyre G. J. Avdeev M. Suard E. Kimber S. A. J. Lançon D. Pepe G. Moubaraki B. Hicks T. J. Magnetic Structure of the Quasi-Two-Dimensional Antiferromagnet NiPS3 Phys. Rev. B 2015 92 22 22440810Magnetic structure of the quasi-two-dimensional antiferromagnet NiPS3Wildes, A. R.; Simonet, V.; Ressouche, E.; McIntyre, G. J.; Avdeev, M.; Suard, E.; Kimber, S. A. J.; Lancon, D.; Pepe, G.; Moubaraki, B.; Hicks, T. J.Physical Review B: Condensed Matter and Materials Physics (2015), 92 (22), 224408/1-224408/11CODEN: PRBMDO; ISSN:1098-0121. (American Physical Society)The magnetic structure of the quasi-two-dimensional antiferromagnet NiPS3 has been detd. by magnetometry and a variety of neutron diffraction techniques. The expts. show that the samples must be carefully handled, as gluing influences the magnetometry measurements while preferred orientation complicates the interpretation of powder diffraction measurements. Our global set of consistent measurements show numerous departures from previously published results. We show that the compd. adopts a k = [010] antiferromagnetic structure with the moment directions mostly along the a axis, and that the paramagnetic susceptibility is isotropic. The crit. behavior was also investigated through the temp. dependence of the magnetic Bragg peaks below the N´eel temp.
- 11Wang F. Mathur N. Janes A. N. Sheng H. He P. Zheng X. Yu P. DeRuiter A. J. Schmidt J. R. He J. Jin S. Defect-Mediated Ferromagnetism in Correlated Two-Dimensional Transition Metal Phosphorus Trisulfides Sci. Adv. 2021 7 43 eabj4086There is no corresponding record for this reference.
- 12Wang X. Cao J. Lu Z. Cohen A. Kitadai H. Li T. Tan Q. Wilson M. Lui C. H. Smirnov D. Sharifzadeh S. Ling X. Spin-Induced Linear Polarization of Photoluminescence in Antiferromagnetic van der Waals Crystals Nat. Mater. 2021 20 7 964 97012Spin-induced linear polarization of photoluminescence in antiferromagnetic van der Waals crystalsWang, Xingzhi; Cao, Jun; Lu, Zhengguang; Cohen, Arielle; Kitadai, Hikari; Li, Tianshu; Tan, Qishuo; Wilson, Matthew; Lui, Chun Hung; Smirnov, Dmitry; Sharifzadeh, Sahar; Ling, XiNature Materials (2021), 20 (7), 964-970CODEN: NMAACR; ISSN:1476-1122. (Nature Portfolio)Antiferromagnets are promising components for spintronics due to their terahertz resonance, multilevel states and absence of stray fields. However, the zero net magnetic moment of antiferromagnets makes the detection of the antiferromagnetic order and the investigation of fundamental spin properties notoriously difficult. Here, we report an optical detection of Neel vector orientation through an ultra-sharp photoluminescence in the van der Waals antiferromagnet NiPS3 from bulk to atomically thin flakes. The strong correlation between spin flipping and elec. dipole oscillator results in a linear polarization of the sharp emission, which aligns perpendicular to the spin orientation in the crystal. By applying an in-plane magnetic field, we achieve manipulation of the photoluminescence polarization. This correlation between emitted photons and spins in layered magnets provides routes for investigating magneto-optics in two-dimensional materials, and hence opens a path for developing opto-spintronic devices and antiferromagnet-based quantum information technologies.
- 13Kim K. Lim S. Y. Lee J. U. Lee S. Kim T. Y. Park K. Jeon G. S. Park C. H. Park J. G. Cheong H. Suppression of Magnetic Ordering in XXZ-Type Antiferromagnetic Monolayer NiPS3 Nat. Commun. 2019 10 1 34513Suppression of magnetic ordering in XXZ-type antiferromagnetic monolayer NiPS3Kim Kangwon; Lim Soo Yeon; Lee Jae-Ung; Cheong Hyeonsik; Lee Sungmin; Park Kisoo; Park Je-Geun; Lee Sungmin; Kim Tae Yun; Park Kisoo; Park Cheol-Hwan; Park Je-Geun; Kim Tae Yun; Park Cheol-Hwan; Jeon Gun SangNature communications (2019), 10 (1), 345 ISSN:.How a certain ground state of complex physical systems emerges, especially in two-dimensional materials, is a fundamental question in condensed-matter physics. A particularly interesting case is systems belonging to the class of XY Hamiltonian where the magnetic order parameter of conventional nature is unstable in two-dimensional materials leading to a Berezinskii-Kosterlitz-Thouless transition. Here, we report how the XXZ-type antiferromagnetic order of a magnetic van der Waals material, NiPS3, behaves upon reducing the thickness and ultimately becomes unstable in the monolayer limit. Our experimental data are consistent with the findings based on renormalization-group theory that at low temperatures a two-dimensional XXZ system behaves like a two-dimensional XY one, which cannot have a long-range order at finite temperatures. This work provides the experimental examination of the XY magnetism in the atomically thin limit and opens opportunities of exploiting these fundamental theorems of magnetism using magnetic van der Waals materials.
- 14Belianinov A. He Q. Dziaugys A. Maksymovych P. Eliseev E. Borisevich A. Morozovska A. Banys J. Vysochanskii Y. Kalinin S. V. CuInP2S6 Room Temperature Layered Ferroelectric Nano Lett. 2015 15 6 3808 381414CuInP2S6 Room Temperature Layered FerroelectricBelianinov, A.; He, Q.; Dziaugys, A.; Maksymovych, P.; Eliseev, E.; Borisevich, A.; Morozovska, A.; Banys, J.; Vysochanskii, Y.; Kalinin, S. V.Nano Letters (2015), 15 (6), 3808-3814CODEN: NALEFD; ISSN:1530-6984. (American Chemical Society)The authors explore ferroelec. properties of cleaved 2-dimensional flakes of copper indium thiophosphate, CuInP2S6 (CITP), and probe size effects along with limits of ferroelec. phase stability, by ambient and ultra high vacuum scanning probe microscopy. CITP belongs to the only material family known to display ferroelec. polarization in a van der Waals, layered crystal at room temp. and above. The authors' measurements directly reveal stable, ferroelec. polarization as evidenced by domain structures, switchable polarization, and hysteresis loops. At room temp. the domain structure of flakes thicker than 100 nm is similar to the cleaved bulk surfaces, whereas <50 nm polarization disappears. The authors ascribe this behavior to a known instability of polarization due to depolarization field. Also, polarization switching at high bias is also assocd. with ionic mobility, as evidenced both by macroscopic measurements and by formation of surface damage under the tip at a bias of 4 V-likely due to copper redn. Mobile Cu ions may therefore also contribute to internal screening mechanisms. The existence of stable polarization in a van-der-Waals crystal naturally points toward new strategies for ultimate scaling of polar materials, quasi-2D, and single-layer materials with advanced and nonlinear dielec. properties that are presently not found in any members of the growing graphene family.
- 15Dziaugys A. Banys J. Macutkevic J. Sobiestianskas R. Vysochanskii Y. Dipolar Glass Phase in Ferrielectrics: CuInP2S6 and Ag0.1Cu0.9InP2S6 Crystals Phys. Status Solidi A 2010 207 8 1960 196715Dipolar glass phase in ferrielectrics: CuInP2S6 and Ag0.1Cu0.9InP2S6 crystalsDziaugys, Andrius; Banys, Juras; Macutkevic, Jan; Sobiestianskas, Ricardas; Vysochanskii, YulianPhysica Status Solidi A: Applications and Materials Science (2010), 207 (8), 1960-1967CODEN: PSSABA; ISSN:1862-6300. (Wiley-VCH Verlag GmbH & Co. KGaA)The dielec. properties of CuInP2S6 and Ag0.1Cu0.9InP2S6 crystals were investigated in wide frequency (20 Hz-3 GHz) and temp. ranges (25-360 K). The low frequencies dielec. spectra at high temps. are highly influenced by the high ionic cond. The temp. dependences of the static dielec. permittivity and the mean relaxation time reveal the first order order-disorder ferrielec. phase transition at Tc = 288 K in Ag0.1Cu0.9InP2S6. The cond. and its activation energy exhibit only a weak change close to the ferrielec. phase transition temp. in both investigated crystals. The freezing phenomena in CuInP2S6 and Ag0.1Cu0.9InP2S6 revealed the complex dielec. permittivity behavior characteristic for transition into dipolar glass state.
- 16Silipigni L. Schiro L. Scolaro L. M. De Luca G. Salvato G. Lithium Ions Conduction in Li2xMn1-xPS3 Films Philos. Mag. 2014 94 35 4026 403616Lithium ions conduction in Li2xMn1-xPS3 filmsSilipigni, L.; Schiro, L.; Monsu Scolaro, L.; De Luca, G.; Salvato, G.Philosophical Magazine (2014), 94 (35), 4026-4036CODEN: PMHABF; ISSN:1478-6435. (Taylor & Francis Ltd.)Li2xMn1-xPS3 films have been synthesized by exfoliating MnPS3 through the successive intercalations of K+ and Li+ ions. Their dielec. response has been measured from 80 to 350 K in the frequency range of (102-106) Hz. The obtained data have been analyzed in terms of both complex permittivity ε* and the ac cond. σac. The frequency dependence of σac has been interpreted in terms of the Jonscher's law, whose exponent n decreases by increasing temp. The n values lie between 0.479 and 0.501 and are typical of materials in which the ac cond. is due to multiple hops of carriers. By analyzing the σdc temp. dependence, the obsd. dielec. response has been attributed to the intercalated lithium ions, and the Li2xMn1-xPS3 films have been classified as hopping charge carrier systems.
- 17Kuzminskii Y. V. Voronin B. M. Redin N. N. Iron and Nickel Phosphorus Trisulfides as Electroactive Materials for Primary Lithium Batteries J. Power Sources 1995 55 2 133 14117Iron and nickel phosphorus trisulfides as electroactive materials for primary lithium batteriesKuzminskii, Y. V.; Voronin, B. M.; Redin, N. N.Journal of Power Sources (1995), 55 (2), 133-41CODEN: JPSODZ; ISSN:0378-7753. (Elsevier)The theor. specific capacities of a family of layered compds. MPX3 (M = Fe, Co, Ni; X = S, Se) for current-producing reactions involving 1.5, 2, 6 and 9 lithium atoms per MPX3 mol. have been estd. These data show that FePS3 and NiPS3 are good electroactive materials for primary lithium cells. Conditions which allow one to shorten substantially the synthesis time of iron and nickel phosphorus trisulfides in ampules are presented. According to cond. measurements in the 293-673 K temp. range, NiPS3 is an intrinsic and FePS3 an extrinsic semiconductor. The discharge characteristics of Li/MPS3 (M = Fe, Ni) cells with an org. electrolyte have been studied; they confirmed that the participation of nine electrons in the redox process is possible. For a primary button cell of the std. size 2325, with a FePS3 cathode, the specific capacity and specific energy values obtained in a discharge 1.8-1.2 V range at load resistances of 3.0 to 30 kΩ were 500-1160 Ah/kg and 700-1770 Wh/kg (on a pure FePS3 basis), resp.
- 18Qian X. Chen L. Yin L. Liu Z. Pei S. Li F. Hou G. Chen S. Song L. Thebo K. H. Cheng H. M. Ren W. CdPS3 Nanosheets-Based Membrane with High Proton Conductivity Enabled by Cd Vacancies Science 2020 370 6516 596 60018CdPS3 nanosheets-based membrane with high proton conductivity enabled by Cd vacanciesQian, Xitang; Chen, Long; Yin, Lichang; Liu, Zhibo; Pei, Songfeng; Li, Fan; Hou, Guangjin; Chen, Shuangming; Song, Li; Thebo, Khalid Hussain; Cheng, Hui-Ming; Ren, WencaiScience (Washington, DC, United States) (2020), 370 (6516), 596-600CODEN: SCIEAS; ISSN:1095-9203. (American Association for the Advancement of Science)P transport in nanochannels under humid conditions is crucial for the application in energy storage and conversion. However, existing materials, including Nafion, suffer from limited cond. of up to 0.2 S per cm. The authors report a class of membranes assembled with 2-dimensional transition-metal P trichalcogenide nanosheets, in which the transition-metal vacancies enable exceptionally high ion cond. A Cd0.85PS3Li0.15H0.15 membrane exhibits a p conduction dominant cond. of ∼0.95 S per cm at 90°elsius and 98% relative humidity. This performance mainly originates from the abundant p donor centers, easy p desorption, and excellent hydration of the membranes induced by Cd vacancies. The authors also obsd. superhigh Li ion cond. in Cd0.85PS3Li0.3 and Mn0.77PS3Li0.46 membranes.
- 19Du K. Z. Wang X. Z. Liu Y. Hu P. Utama M. I. Gan C. K. Xiong Q. Kloc C. Weak Van der Waals Stacking, Wide-Range Band Gap, and Raman Study on Ultrathin Layers of Metal Phosphorus Trichalcogenides ACS Nano 2016 10 2 1738 174319Weak Van der Waals Stacking, Wide-Range Band Gap, and Raman Study on Ultrathin Layers of Metal Phosphorus TrichalcogenidesDu, Ke-zhao; Wang, Xing-zhi; Liu, Yang; Hu, Peng; Utama, M. Iqbal Bakti; Gan, Chee Kwan; Xiong, Qihua; Kloc, ChristianACS Nano (2016), 10 (2), 1738-1743CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)2D semiconducting metal phosphorus trichalcogenides, particularly the bulk crystals of MPS3 (M = Fe, Mn, Ni, Cd and Zn) sulfides and MPSe3 (M = Fe and Mn) selenides, have been synthesized, crystd. and exfoliated into monolayers. The Raman spectra of monolayer FePS3 and 3-layer FePSe3 show the strong intralayer vibrations and structural stability of the atomically thin layers under ambient condition. The band gaps can be adjusted by element choices in the range of 1.3-3.5 eV. The wide-range band gaps suggest their optoelectronic applications in a broad wavelength range. The calcd. cleavage energies of MPS3 are smaller than that of graphite. Therefore, the monolayers used for building of heterostructures by van der Waals stacking could be considered as the candidates for artificial 2D materials with unusual ferroelec. and magnetic properties.
- 20Nitsche R. Wild P. Crystal Growth of Metal-Phosphorus-Sulfur Compounds by Vapor Transport Mater. Res. Bull. 1970 5 6 419 42320Crystal growth of metal-phosphorus-sulfur compounds by vapor transportNitsche, Rudolf; Wild, PetrMaterials Research Bulletin (1970), 5 (6), 419-23CODEN: MRBUAC; ISSN:0025-5408.Single crystals of the thiophosphates: Cu3PS4, InPS4, GaPS4, BiPS4 and the thiohypophosphates Sn2P2S6, Cd2P2S6, Fe2P2S6, and Mn2P2S6 were grown in sizes up to 10 mm by vapor transport with I. Lattice parameters and space groups are given.
- 21Gusmao R. Sofer Z. Pumera M. Metal Phosphorous Trichalcogenides (MPCh3 ): From Synthesis to Contemporary Energy Challenges Angew. Chem. Int. Ed. Engl. 2019 58 28 9326 933721Metal Phosphorous Trichalcogenides (MPCh3 ): From Synthesis to Contemporary Energy ChallengesGusmao Rui; Sofer Zdenek; Pumera MartinAngewandte Chemie (International ed. in English) (2019), 58 (28), 9326-9337 ISSN:.Owing to their unique physical and chemical properties, layered two-dimensional (2D) materials have been established as the most significant topic in materials science for the current decade. This includes layers comprising mono-element (graphene, phosphorene), di-element (metal dichalcogenides), and even multi-element. A distinctive class of 2D layered materials is the metal phosphorous trichalcogenides (MPCh3 , Ch=S, Se), first synthesized in the late 1800s. Having an unusual intercalation behavior, MPCh3 were intensively studied in the 1970s for their magnetic properties and as secondary electrodes in lithium batteries, but fell from scrutiny until very recently, being 2D nanomaterials. Based on their synthesis and most significant properties, the present surge of reports related to water-splitting catalysis and energy storage are discussed in detail. This Minireview is intended as a baseline for the anticipated new wave of researchers who aim to explore these 2D layered materials for their electrochemical energy applications.
- 22Gave M. A. Bilc D. Mahanti S. D. Breshears J. D. Kanatzidis M. G. On the Lamellar Compounds CuBiP2Se6, AgBiP2Se6 and AgBiP2S6. Antiferroelectric Phase Transitions due to Cooperative Cu+ and Bi3+ Ion Motion Inorg. Chem. 2005 44 15 5293 530322On the Lamellar Compounds CuBiP2Se6, AgBiP2Se6 and AgBiP2S6. Antiferroelectric Phase Transitions Due to Cooperative Cu+ and Bi3+ Ion MotionGave, Matthew A.; Bilc, Daniel; Mahanti, S. D.; Breshears, Jean D.; Kanatzidis, Mercouri G.Inorganic Chemistry (2005), 44 (15), 5293-5303CODEN: INOCAJ; ISSN:0020-1669. (American Chemical Society)CuBiP2Se6, AgBiP2Se6, and AgBiP2S6 were prepd. from the corresponding elements. CuBiP2Se6 and AgBiP2Se6 crystallize in the space group R‾3 with a 6.5532(16) and c 39.762(13) Å for CuBiP2Se6 and a 6.6524(13) and c 39.615(15) Å for AgBiP2Se6. AgBiP2S6 crystallizes in the triclinic space group P‾1 with a 6.3833(13), b 7.1439(14), c 9.5366(19) Å, α 91.89(3), β 91.45(3), γ 94.05(3)°. CuBiP2Se6 was found to exhibit a temp.-dependent antiferroelec. ordering of the Cu+ and Bi3+ ions in the lattice. An intermediate and a fully ordered structure were refined at 173 and 97 K, resp. Electronic band and total energy calcns. at the DFT level clearly suggest that the antiferroelec. model is energetically favored over the paraelec. and hypothetical ferrielec. models. This phase transition can be classified as a 2nd-order Jahn-Teller distortion. The antiferroelec. state of CuBiP2Se6 is an indirect gap semiconductor. The compds. were characterized with DTA and solid-state UV/visible diffuse reflectance spectroscopy. Generalized implications regarding the expected ferroelec. behavior of compds. in the CuMP2Se6 system (M = trivalent metal) are discussed.
- 23Wang F. Shifa T. A. He P. Cheng Z. Chu J. Liu Y. Wang Z. Wang F. Wen Y. Liang L. He J. Two-Dimensional Metal Phosphorus Trisulfide Nanosheet with Solar Hydrogen-Evolving Activity Nano Energy 2017 40 673 68023Two-dimensional metal phosphorus trisulfide nanosheet with solar hydrogen-evolving activityWang, Fengmei; Shifa, Tofik Ahmed; He, Peng; Cheng, Zhongzhou; Chu, Junwei; Liu, Yang; Wang, Zhenxing; Wang, Feng; Wen, Yao; Liang, Lirong; He, JunNano Energy (2017), 40 (), 673-680CODEN: NEANCA; ISSN:2211-2855. (Elsevier Ltd.)The development and utilization of photocatalysts to realize water-splitting without any external bias or sacrificial agents has received the limelight. As a novel two-dimensional layered material, metal phosphorus trichalcogenides (MPTs) cause wide research interest, presently. However, the growth of ultrathin two-dimensional MPT crystals is a great challenge to hinder their application. Here, we initially grow few-at. layered nickel phosphorus trisulfide (NiPS3) as promising photocatalyst for hydrogen evolution. The as-prepd. NiPS3 hexagonal nanosheet, as thin as few at. layers (≤ 3.5 nm), has lateral size of larger than 15 μm. These ultrathin NiPS3 crystals can directly generate hydrogen gas from pure water without any sacrificial agents under sunlight. With UV photoelectron spectrometer and electrochem. impedance spectroscopy, we show that the attractive photocatalytic activity of the ultrathin NiPS3 crystals arise from their appropriate positions of the band edges. This discovery is expected to make a contribution to develop next generation solar-fuel conversion catalysts for H2 prodn.
- 24Yu P. Meng J. Wang F. Sendeku M. G. Wu B. Sui X. Gao N. Zhan X. Lou X. Wang Z. He J. Carbonate-Ion-Mediated Photogenerated Hole Transfer to Boost Hydrogen Production J. Phys. Chem. C 2022 126 25 10367 1037724Carbonate-Ion-Mediated Photogenerated Hole Transfer to Boost Hydrogen ProductionYu, Peng; Meng, Jun; Wang, Fengmei; Sendeku, Marshet Getaye; Wu, Binglan; Sui, Xinyu; Gao, Ning; Zhan, Xueying; Lou, Xiaoding; Wang, Zhenxing; He, JunJournal of Physical Chemistry C (2022), 126 (25), 10367-10377CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Sustainable and scalable H2 evolution through water photocatalysis is an attractive path for carbon-neutral energy supply; however, it is severely limited by sluggish charge sepn. and photocorrosion of semiconductor photocatalysts. Here, we demonstrate that earth-abundant carbonate ions, widely existing in daily-life water, serve as a hole mediator to redirect the photogenerated hole transfer pathway and then promote the hole-transfer kinetics. The accelerated hole transfer could efficiently reduce the recombination of electron-hole pairs for continuous H2 prodn. with improved photostability of catalysts, including layered indium phosphorus sulfide (In4/3P2S6) and cadmium sulfide. A sustainable H2 evolution rate of 5.1 mmol g-1 h-1 within 60 h or more operation is achieved in the presence of CO32- anions. In situ ESR (ESR) spectroscopy studies and transient absorption (TA) measurements reveal that the CO32-/CO3- redox couple could rapidly shuttle the photogenerated holes from OH radicals anchored on the catalyst surface, effectively eliminating the recombination of electron-hole pairs and catalyst oxidn. for boosted H2 generation. The carbonate-ion-mediated hole-transfer strategy provides a new paradigm for designing a cost-effective and advanced photosynthetic system in practical applications.
- 25Zhou J. Zhu C. Zhou Y. Dong J. Li P. Zhang Z. Wang Z. Lin Y. C. Shi J. Zhang R. Zheng Y. Yu H. Tang B. Liu F. Wang L. Liu L. Liu G. B. Hu W. Gao Y. Yang H. Gao W. Lu L. Wang Y. Suenaga K. Liu G. Ding F. Yao Y. Liu Z. Composition and Phase Engineering of Metal Chalcogenides and Phosphorous Chalcogenides Nat. Mater. 2022There is no corresponding record for this reference.https://doi.org/10.1038/s41563-022-01291-5.There is no corresponding record for this reference.
- 26Sendeku M. G. Wang F. Cheng Z. Yu P. Gao N. Zhan X. Wang Z. He J. Nonlayered Tin Thiohypodiphosphate Nanosheets: Controllable Growth and Solar-Light-Driven Water Splitting ACS Appl. Mater. Interfaces 2021 13 11 13392 1339926Nonlayered Tin Thiohypodiphosphate Nanosheets: Controllable Growth and Solar-Light-Driven Water SplittingSendeku, Marshet G.; Wang, Fengmei; Cheng, Zhongzhou; Yu, Peng; Gao, Ning; Zhan, Xueying; Wang, Zhenxing; He, JunACS Applied Materials & Interfaces (2021), 13 (11), 13392-13399CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)As a promising candidate in various fields, including energy conversion and electronics, layered van der Waals metal phosphorus trichalcogenides (MPX3) have been widely explored. In addn. to the layered structures, MPX3 comprising post-transition metals (i.e., Sn and Pb) are known to form a unique 3D framework with nonlayered structure. However, the nonlayered two-dimensional (2D) crystals of this family have remained unexplored until now. Herein, we successfully synthesized 2D nonlayered tin thiohypodiphosphate (Sn2P2S6) nanosheets, having an indirect bandgap of 2.25 eV and a thickness down to ~ 10 nm. The as-obtained nanosheets demonstrate promising photocatalytic water splitting activity to generate H2 in pure water under simulated solar light (AM 1.5G). Moreover, the ultrathin Sn2P2S6 catalyst shows auspicious performance and stability with a continuous operation of 40 h. This work is not only an expansion of the MPX3 family, but it is also a major milestone in the search for new materials for future energy conversion.
- 27Shifa T. A. Wang F. Cheng Z. He P. Liu Y. Jiang C. Wang Z. He J. High Crystal Quality 2D Manganese Phosphorus Trichalcogenide Nanosheets and Their Photocatalytic Activity Adv. Funct. Mater. 2018 28 18 1800548There is no corresponding record for this reference.
- 28Zhu W. Gan W. Muhammad Z. Wang C. Wu C. Liu H. Liu D. Zhang K. He Q. Jiang H. Zheng X. Sun Z. Chen S. Song L. Exfoliation of Ultrathin FePS3 Layers as a Promising Electrocatalyst for the Oxygen Evolution Reaction Chem. Commun. 2018 54 35 4481 448428Exfoliation of ultrathin FePS3 layers as a promising electrocatalyst for the oxygen evolution reactionZhu, Wen; Gan, Wei; Muhammad, Zahir; Wang, Changda; Wu, Chuanqiang; Liu, Hengjie; Liu, Daobin; Zhang, Ke; He, Qun; Jiang, Hongliang; Zheng, Xusheng; Sun, Zhe; Chen, Shuangming; Song, LiChemical Communications (Cambridge, United Kingdom) (2018), 54 (35), 4481-4484CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)Few-layer ternary FePS3 nanosheets, prepd. via chem. vapor transport synthesis and ball-milling exfoliation, exhibit excellent electrocatalytic performance for the oxygen evolution reaction in an alk. medium. Combined with first principles calcns., our X-ray spectroscopy and HRTEM results clearly reveal that the introduction of in-plane defects in FePS3 layers after exfoliation and formation of a FePS3-FeOOH heterostructure during the OER process largely contribute to the catalytic activity enhancement.
- 29Sang Y. Wang L. Cao X. Ding G. Ding Y. Hao Y. Xu N. Yu H. Li L. Peng S. Emerging 2D-Layered MnPS3/rGO Composite as a Superior Anode for Sodium-Ion Batteries J. Alloys Compd. 2020 831 15477529Emerging 2D-Layered MnPS3/rGO composite as a superior anode for sodium-ion batteriesSang, Yan; Wang, Lvxuan; Cao, Xi; Ding, Gaofei; Ding, Yonghao; Hao, Yanan; Xu, Na; Yu, Hanzhi; Li, Linlin; Peng, ShengjieJournal of Alloys and Compounds (2020), 831 (), 154775CODEN: JALCEU; ISSN:0925-8388. (Elsevier B.V.)Two-dimensional (2D) layered materials were widely studied due to their unique Na storage properties and rapid ion transport rates. Herein, the authors synthesized a 2-dimensional layered transition metal P sulfide MnPS3 by one step high-temp. solid-phase synthesis. After combining with graphene by high-energy ball milling followed by high-temp. Ar calcination, the novel 2D/2D heterojunction of extra-thin MnPS3/rGO was successfully prepd. The resultant MnPS3/rGO hybrid can strengthen the cond. of the material and ameliorate the vol. change during the insertion/extn. process of the Na-ion storage, compared to the pristine MnPS3. As a result, the 2D/2D heterojunction of ultra-thin MnPS3/rGO composite exhibits high cycling performance (290 mA-h g-1 after 150 cycles at 0.2 A g-1), capacity retention rates up to 92%. The superior performance is ascribed to the combination of extra-thin MnPS3 nanosheets with graphene, which effectively enhances the interface contact area and the electronic transmission rate, greatly improving the adaptability of vol. change and interfacial charge transfer abilities. This work provides a novel and promising MnPS3/rGO anode for Na-ion batteries.
- 30Liu J. Li X. B. Wang D. Lau W.-M. Peng P. Liu L.-M. Diverse and Tunable Electronic Structures of Single-layer Metal Phosphorus Trichalcogenides for Photocatalytic Water Splitting J. Chem. Phys. 2014 140 5 05470730Diverse and tunable electronic structures of single-layer metal phosphorus trichalcogenides for photocatalytic water splittingLiu, Jian; Li, Xi-Bo; Wang, Da; Lau, Woon-Ming; Peng, Ping; Liu, Li-MinJournal of Chemical Physics (2014), 140 (5), 054707/1-054707/7CODEN: JCPSA6; ISSN:0021-9606. (American Institute of Physics)The family of bulk metal phosphorus trichalcogenides (APX3, A = MII, MI0.5MIII0.5; X = S, Se; MI, MII, and MIII represent Group-I, Group-II, and Group-III metals, resp.) has attracted great attentions because such materials not only own magnetic and ferroelec. properties, but also exhibit excellent properties in hydrogen storage and lithium battery because of the layered structures. Many layered materials have been exfoliated into two-dimensional (2D) materials, and they show distinct electronic properties compared with their bulks. Here the authors present a systematical study of single-layer metal phosphorus trichalcogenides by d. functional theory calcns. The results show that the single layer metal phosphorus trichalcogenides have very low formation energies, which indicates that the exfoliation of single layer APX3 should not be difficult. The family of single layer metal phosphorus trichalcogenides exhibits a large range of band gaps from 1.77 to 3.94 eV, and the electronic structures are greatly affected by the metal or the chalcogenide atoms. The calcd. band edges of metal phosphorus trichalcogenides further reveal that single-layer ZnPSe3, CdPSe3, Ag0.5Sc0.5PSe3, and Ag0.5In0.5PX3 (X = S and Se) have both suitable band gaps for visible-light driving and sufficient over-potentials for water splitting. More fascinatingly, single-layer Ag0.5Sc0.5PSe3 is a direct band gap semiconductor, and the calcd. optical absorption further convinces that such materials own outstanding properties for light absorption. Such results demonstrate that the single layer metal phosphorus trichalcogenides own high stability, versatile electronic properties, and high optical absorption, thus such materials have great chances to be high efficient photocatalysts for water-splitting. (c) 2014 American Institute of Physics.
- 31Mukherjee D. Austeria M. P. Sampath S. Few-Layer Iron Selenophosphate, FePSe3: Efficient Electrocatalyst toward Water Splitting and Oxygen Reduction Reactions ACS Appl. Energy Mater. 2018 1 1 220 23131Few-Layer Iron Selenophosphate, FePSe3: Efficient Electrocatalyst toward Water Splitting and Oxygen Reduction ReactionsMukherjee, Debdyuti; Muthu, Austeria P.; Sampath, S.ACS Applied Energy Materials (2018), 1 (1), 220-231CODEN: AAEMCQ; ISSN:2574-0962. (American Chemical Society)There has been a spurt of activity in using layered MPX3 (M = transition metal, X = chalcogen, S/Se/Te) compds. in various studies including catalysis and devices. In the present study, low band gap, ternary iron selenophosphate (FePSe3) is introduced as an excellent and highly stable trifunctional electrocatalyst for hydrogen evolution, oxygen evolution, and oxygen redn. reactions. It is obsd. that the present catalyst is useful in evolving hydrogen over a wide pH range including seawater environment. D. functional theory calcns. reveal various parameters that help improve the electrocatalytic activity of the layered material. Covalency of the Fe-Se bond, distortion in the crystal structure, and adsorption properties are shown to be responsible for the obsd. high catalytic activity.
- 32Mukherjee D. Austeria P. M. Sampath S. Two-Dimensional, Few-Layer Phosphochalcogenide, FePS3: A New Catalyst for Electrochemical Hydrogen Evolution over Wide pH Range ACS Energy Lett. 2016 1 2 367 37232Two-Dimensional, Few-Layer Phosphochalcogenide, FePS3: A New Catalyst for Electrochemical Hydrogen Evolution over Wide pH RangeMukherjee, Debdyuti; Austeria, P. Muthu; Sampath, S.ACS Energy Letters (2016), 1 (2), 367-372CODEN: AELCCP; ISSN:2380-8195. (American Chemical Society)A layered MPS3-type compd., FePS3, is introduced as an electrocatalyst for hydrogen evolution reaction (HER). The non-noble metal-based FePS3 is a semiconductor that could be solvent exfoliated into few-layer, two-dimensional (2D) nanosheets. The 2-dimensional thin sheets exhibit very good catalytic activity and stability toward HER over a wide pH range of acidic, alk., phosphate buffer, and 3.5% aq. NaCl solns. The Tafel slope and exchange c.d. in acidic medium are ∼(45-50) mV/dec and 1±0.2 × 10-3 A/cm2, resp. The stability of the catalyst is very good. D. functional theory calcns. reveal P and S as favorable hydrogen adsorption sites. This material opens up a new class of ternary, layered semiconductors for various electrocatalytic studies and might also become important from a device physics point of view.
- 33Jenjeti R. N. Kumar R. Sampath S. Two-Dimensional, Few-layer NiPS3 for Flexible Humidity Sensor with High Selectivity J. Mater. Chem. A 2019 7 24 14545 1455133Two-dimensional, few-layer NiPS3 for flexible humidity sensor with high selectivityJenjeti, Ramesh Naidu; Kumar, Rajat; Sampath, S.Journal of Materials Chemistry A: Materials for Energy and Sustainability (2019), 7 (24), 14545-14551CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Chem. and elec. sensitive two-dimensional (2D) nanomaterials are of immense interest as probing electrodes for wearable electronic devices. A new family of two dimensional (2D) layered materials, namely metal phosphochalcogenides (MPX3), are potential candidates towards the development of sensors for various analytes. Herein, we demonstrate the ability of few-layer NiPS3 nanosheets for humidity sensing by fabricating a cost-effective, flexible sensor device. The results indicate that the NiPS3 nanosheet-based humidity sensors possess high sensitivity with a response of ∼106, superior selectivity, and most importantly, rapid response, recovery times and good reproducibility. Response times of ∼1-2 s at low humidity levels and ∼3 s at high humidity levels with recovery times of ∼2-3 s are obsd. The device was tested in both flat and bent states, causing no prominent changes in the response; hence, the sensor is an excellent candidate for use in flexible devices. The characteristics of the NiPS3-based sensor were further investigated using complex impedance studies and in situ Raman spectroscopy to understand the sensing mechanism. The fast response and recovery assocd. with the NiPS3-based humidity sensors allow real time monitoring of human respiration and water evapn. on skin. These humidity sensors can also be utilized for non-contact anal. and hence will be an attractive candidate for health and environmental monitoring.
- 34Song B. Li K. Yin Y. Wu T. Dang L. Caban-Acevedo M. Han J. Gao T. Wang X. Zhang Z. Schmidt J. R. Xu P. Jin S. Tuning Mixed Nickel Iron Phosphosulfide Nanosheet Electrocatalysts for Enhanced Hydrogen and Oxygen Evolution ACS Catal. 2017 7 12 8549 855734Tuning Mixed Nickel Iron Phosphosulfide Nanosheet Electrocatalysts for Enhanced Hydrogen and Oxygen EvolutionSong, Bo; Li, Kai; Yin, Ying; Wu, Tao; Dang, Lianna; Caban-Acevedo, Miguel; Han, Jiecai; Gao, Tangling; Wang, Xianjie; Zhang, Zhihua; Schmidt, J. R.; Xu, Ping; Jin, SongACS Catalysis (2017), 7 (12), 8549-8557CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)Highly efficient earth-abundant electrocatalysts for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) are of great importance for renewable energy conversion systems. Herein, guided by theor. calcns., we demonstrate highly efficient water splitting in alk. soln. using quaternary mixed nickel iron phosphosulfide (Ni1-xFexPS3) nanosheets (NSs), even though neither NiPS3 nor FePS3 is a good HER (or OER) electrocatalyst. With tuned electronic structure and improved elec. cond. induced by mixing appropriate amt. of Fe into NiPS3, Ni0.9Fe0.1PS3 NSs display excellent HER activity (an overpotential of 72 mV vs reversible hydrogen electrode (RHE) at a geometric catalytic c.d. of -10 mA cm-2 and a Tafel slope of 73 mV dec-1), which is among the best HER catalysts under alk. conditions. Ni0.9Fe0.1PS3 NSs also show a good apparent OER activity (an overpotential of 329 mV vs RHE at a catalytic c.d. of 20 mA cm-2 and a Tafel slope of 69 mV dec-1), although structural investigation indicates the formation of Ni(Fe)OOH and Ni(Fe)(OH)2 layers on the catalyst surface after OER reactions as likely the real active species. These mixed nickel iron phosphosulfide non-precious-metal electrocatalysts with enhanced intrinsic activity and long-term stability and durability should have great potential in overall water-splitting applications.
- 35Cheng Z. Shifa T. A. Wang F. Gao Y. He P. Zhang K. Jiang C. Liu Q. He J. High-Yield Production of Monolayer FePS3 Quantum Sheets via Chemical Exfoliation for Efficient Photocatalytic Hydrogen Evolution Adv. Mater. 2018 30 26 1707433There is no corresponding record for this reference.
- 36Dinh Hoa L. Thanh Luan P. Ghimire G. Dinh Loc D. Lee Y. H. Revealing Antiferromagnetic Transition of van der Waals MnPS3 via Vertical Tunneling Electrical Resistance Measurement APL Mater. 2019 7 8 081102There is no corresponding record for this reference.
- 37Wang J. Li X. Wei B. Sun R. Yu W. Hoh H. Y. Xu H. Li J. Ge X. Chen Z. Su C. Wang Z. Activating Basal Planes of NiPS3 for Hydrogen Evolution by Nonmetal Heteroatom Doping Adv. Funct. Mater. 2020 30 12 190870837Activating Basal Planes of NiPS3 for Hydrogen Evolution by Nonmetal Heteroatom DopingWang, Jun; Li, Xinzhe; Wei, Bin; Sun, Rong; Yu, Wei; Hoh, Hui Ying; Xu, Haomin; Li, Jing; Ge, Xingbo; Chen, Zuxin; Su, Chenliang; Wang, ZhongchangAdvanced Functional Materials (2020), 30 (12), 1908708CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)NiPS3, one of the most promising catalysts among transition metal trichalcogenidophosphates (MTPs) in hydrogen evolution reaction (HER) electrocatalysis, is still inhibited by its unsatisfactory activity originating from its semiconducting nature and inert basal plane. Here, it is proposed, for the first time, to engineer the basal surface activity of NiPS3 by nonmetal heteroatom doping, and predict that the degree to which the valance band of NiPS3 is filled dominates not only the elec. cond. of the catalyst, but also the strength of hydrogen adsorption at its surface. Direct exptl. evidence is offered that in all the single nonmetal doping samples, C-doped NiPS3 exhibits the optimum activity owing to its moderate filled state of valance band and that C, N codoping even shows Pt-like activity with an ultralow overpotential of 53.2 mV to afford 10 mA cm-2 c.d. and a high exchange c.d. of 0.7 mA cm-2 in 1 M KOH. The findings that less valance electrons of dopants than substitutional atoms are of pivotal importance for improving HER activity of NiPS3 catalyst pave the way for readily designing novel MTPs of ever high performance to replace the incumbent Pt-based catalysts.
- 38Li X. Fang Y. Wang J. Wei B. Qi K. Hoh H. Y. Hao Q. Sun T. Wang Z. Yin Z. Zhang Y. Lu J. Bao Q. Su C. High-Yield Electrochemical Production of Large-Sized and Thinly Layered NiPS3 Flakes for Overall Water Splitting Small 2019 15 30 1902427There is no corresponding record for this reference.
- 39Sekine T. Jouanne M. Julien C. Balkanski M. Raman Scattering in the Antiferromagnet FePS3 Intercalated with Lithium Mater. Sci. Eng. B Solid State Mater. Adv. Technol. 1989 3 1−2 91 95There is no corresponding record for this reference.
- 40Fujishima A. Honda K. Electrochemical Photolysis of Water at a Semiconductor Electrode Nature 1972 238 5358 37 3840Electrochemical photolysis of water at a semiconductor electrodeFujishima, Akira; Honda, KenichiNature (London, United Kingdom) (1972), 238 (5358), 37-8CODEN: NATUAS; ISSN:0028-0836.For electrochem. decompn. of H2O, a p.d. of >1.23 V is necessary between the anode and the cathode. This p.d. is equal to the energy of radiation with wavelength of ∼1000 nm. This energy, in the form of visible light, can be used effectively in an electrochem. system to decomp. H2O. A novel type of electrochem. cell was developed, in which a TiO2 electrode was connected with a Pt-black electrode through an external load. The direction of current revealed that oxidn. occurred at the TiO2 electrode and redn. at the Pt-black electrode. To increase the efficiency of the process, more reducible species, such as dissolved O2 or Fe3+, must be added in the Pt electrode compartment. The use of a p-type semiconductor electrode instead of Pt leads to more effective electrochem. photolysis of H2O.
- 41Nakata K. Fujishima A. TiO2 Photocatalysis: Design and Applications J. Photochem. Photobiol. C 2012 13 3 169 18941TiO2 photocatalysis: Design and applicationsNakata, Kazuya; Fujishima, AkiraJournal of Photochemistry and Photobiology, C: Photochemistry Reviews (2012), 13 (3), 169-189CODEN: JPPCAF; ISSN:1389-5567. (Elsevier B.V.)In this review, recent developments in the area of TiO2 photocatalysis research, in terms of new materials from a structural design perspective, have been summarized. The dimensionality assocd. with the structure of a TiO2 material can affect its properties and functions, including its photocatalytic performance, and also more specifically its surface area, adsorption, reflectance, adhesion, and carrier transportation properties. The authors provide a brief introduction to the current situation in TiO2 photocatalysis, and describe structurally controlled TiO2 photocatalysts which can be classified into zero-, one-, two-, and three-dimensional structures. Furthermore, novel applications of TiO2 surfaces for the fabrication of wettability patterns and for printing are discussed.
- 42Chiesa P. Consonni S. Kreutz T. Williams R. Co-Production of Hydrogen, Electricity and CO2 from Coal with Commercially Ready Technology. PartA: Performance and Emissions Int. J. Hydrogen Energy 2005 30 7 747 76742Co-production of hydrogen, electricity and CO2 from coal with commercially ready technology. Part A: Performance and emissionsChiesa, Paolo; Consonni, Stefano; Kreutz, Thomas; Williams, RobertInternational Journal of Hydrogen Energy (2005), 30 (7), 747-767CODEN: IJHEDX; ISSN:0360-3199. (Elsevier Ltd.)This two-part paper investigates performances, costs and prospects of using com. ready technol. to convert coal to H2 and electricity, with CO2 capture and storage. Part A focuses on plant configuration and the evaluation of performances and CO2 emissions. Part B focuses on economics, establishing benchmarks for the assessment of novel technologies and guidelines for technol. development. In the co-prodn. plants considered in the paper, coal is gasified to synthesis gas in an entrained flow gasifier. The syngas is cooled, cleaned of particulate matter, and shifted (to primarily H2 and CO2) in sour water-gas shift reactors. After further cooling, H2S is removed from the syngas using a phys. solvent (Selexol); CO2 is then removed from the syngas, again using Selexol; after being stripped from the solvent, the CO2 is dried and compressed to 150 bar for pipeline transport and underground storage. High purity H2 (99.999%) is extd. from the H2-rich syngas via a pressure swing adsorption (PSA) unit and delivered at 60 bar. The PSA purge gas is compressed and burned in a conventional gas turbine combined cycle, generating co-product electricity. The H2/electricity ratio can be varied by lowering the steam-to-carbon ratio in the syngas or by letting part of the de-carbonized syngas bypass the PSA unit. Performances and emissions of H2/electricity co-prodn. with CO2 capture are compared with those of a system that vents the CO2. We examine different methods of syngas heat recovery (quench vs. radiant cooling) and explore the effects of changing the electricity/H2 ratio, gasifier pressure and hydrogen purity. Results show that state-of-the-art com. technol. allows transferring to de-carbonized hydrogen 57-58% of coal LHV, while exporting to the grid decarbonized electricity amounting to 2-6% of coal LHV. In contrast to decarbonizing coal IGCC electricity, which entails a loss of 6-8 percentage points of electricity conversion when capturing CO2 as an alternative to venting it, CO2 capture for H2 prodn. gives a minor energy penalty (∼ 2 percentage points of export electricity). For H2 prodn., the efficiency gain achievable by hot syngas cooling vs. quench is a modest 2 percentage point increase in electricity for export, compared to 2-4 percentage points in the electricity case. Reducing H2 purity or increasing gasification pressure has minor effects on performance.
- 43Zhang X. Zhao X. Wu D. Jing Y. Zhou Z. MnPSe3 Monolayer: A Promising 2D Visible-Light Photohydrolytic Catalyst with High Carrier Mobility Adv. Sci. 2016 3 10 160006243MnPSe3 Monolayer: A Promising 2D Visible-Light Photohydrolytic Catalyst with High Carrier MobilityZhang Xu; Zhao Xudong; Wu Dihua; Jing Yu; Zhou ZhenAdvanced science (Weinheim, Baden-Wurttemberg, Germany) (2016), 3 (10), 1600062 ISSN:2198-3844.The 2D material single-layer MnPSe3 would be a promising photocatalyst for water splitting, as indicated by the proper positions of band edges, strong absorption in visible-light spectrum, broad applicability (pH = 0 - 7), and high carrier mobility.
- 44Yu P. Wang F. Meng J. Shifa T. A. Sendeku M. G. Fang J. Li S. Cheng Z. Lou X. He J. Few-Layered CuInP2S6 Nanosheet with Sulfur Vacancy Boosting Photocatalytic Hydrogen Evolution CrystEngComm 2021 591 59844Few-layered CuInP2S6 nanosheet with sulfur vacancy boosting photocatalytic hydrogen evolutionYu, Peng; Wang, Fengmei; Meng, Jun; Shifa, Tofik Ahmed; Sendeku, Marshet Getaye; Fang, Ju; Li, Shuxian; Cheng, Zhongzhou; Lou, Xiaoding; He, JunCrystEngComm (2021), 23 (3), 591-598CODEN: CRECF4; ISSN:1466-8033. (Royal Society of Chemistry)Photochem. water splitting offers an economic and sustainable approach for solar energy conversion into hydrogen fuel to mitigate the problem of greenhouse gas emissions. To this end, exploring novel semiconductor photocatalysts, which have efficient light absorption and thermodynamically favorable band alignment for water splitting, is crucial. Here, we rationally develop a new photocatalyst of CuInP2S6 nanosheets to generate hydrogen gas under light illumination. The CuInP2S6 nanosheet (with a thickness of around 4-7 nm) photocatalyst exhibits a high hydrogen prodn. rate of 804μmol g-1 h-1, eight times higher than that of the microsheet counterpart, due to the introduced abundant sulfur vacancies. Exptl. characterization and theor. calcns. verify that the prolonged carrier lifetime and optimized band alignment in ultrathin CuInP2S6 nanosheets boost photocatalytic hydrogen evolution. This work opens a new avenue for photocatalysis via using novel layered binary metal phosphorous trichalcogenides.
- 45Navarro R. M. Sánchez-Sánchez M. C. Alvarez-Galvan M. C. Valle F. d. Fierro J. L. G. Hydrogen Production from Renewable Sources: Biomass and Photocatalytic Opportunities Energy Environ. Sci. 2009 2 1 35 5445Hydrogen production from renewable sources: biomass and photocatalytic opportunitiesNavarro, R. M.; Sanchez-Sanchez, M. C.; Alvarez-Galvan, M. C.; del Valle, F.; Fierro, J. L. G.Energy & Environmental Science (2009), 2 (1), 35-54CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)A review. The demand for hydrogen over the coming decade is expected to grow for both traditional uses (ammonia, methanol, refinery) and running fuel cells. At least in the near future, this thirst for hydrogen will be quenched primarily through the reforming of fossil fuels. However, reforming fossil fuels emits huge amts. of carbon dioxide. One approach to reduce carbon dioxide emissions, which is considered first in this review, is to apply reforming methods to alternative renewable materials. Such materials might be derived from plant crops, agricultural residues, woody biomass, etc. Clean biomass is a proven source of renewable energy that is already used for generating heat, electricity, and liq. transportation fuels. Clean biomass and biomass-derived precursors such as ethanol and sugars are appropriate precursors for producing hydrogen through different conversion strategies. Virtually no net greenhouse gas emissions result because a natural cycle is maintained, in which carbon is extd. from the atm. during plant growth and released during hydrogen prodn. The second option explored here is hydrogen prodn. from water splitting by means of the photons in the visible spectrum. The sun provides silent and precious energy that is distributed fairly evenly all over the earth. However, its tremendous potential as a clean, safe and economical energy source cannot be exploited unless it is accumulated or converted into more useful forms of energy. Finally, this review discusses the use of semiconductors, more specifically CdS and CdS-based semiconductors, which are able to absorb photons in the visible region of the spectrum. The energy stored within a semiconductor as electronic energy (electrons and holes) can be used to split water mols. by simultaneous reactions into H2 and O2. This conversion of solar energy into a clean fuel (H2) is perhaps the greatest challenge for scientists in the 21st century.
- 46Shimura K. Yoshida H. Heterogeneous Photocatalytic Hydrogen Production from Water and Biomass Derivatives Energy Environ. Sci. 2011 4 7 2467 248146Heterogeneous photocatalytic hydrogen production from water and biomass derivativesShimura, Katsuya; Yoshida, HisaoEnergy & Environmental Science (2011), 4 (7), 2467-2481CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)A review. Hydrogen, a clean energy carrier, should be produced from not the irreproducible fossil fuels but renewable resources. Currently hydrogen is mainly produced by the reforming of petroleum and natural gas at high temp., which consumes huge energy. Thus, development of an alternative hydrogen prodn. method is absolutely imperative to mitigate the environmental and energy issues. Photocatalytic hydrogen prodn. from water and renewable resources is one of the attractive systems, since it can utilize the solar energy. In this short review, we remind you that the systems of the photocatalytic hydrogen prodn. from water and biomass derivs., such as ethanol, glycerol, sugars and methane, should be considered as available candidates for the hydrogen prodn. method from renewable resources and solar energy, and these systems have been improved by developments of new photocatalysts with some cocatalysts, modifications of photocatalysts, optimization of reaction conditions, and so on.
- 47Zhang Y. Zhao Y. Bao C. Xiao Y. Xiang Y. Song M. Huang W. Ma L. Hou H. Chen X. Facile synthesis of cadmium phosphorus trisulfide nanosheets for highly efficient photocatalytic performance J. Alloys Compd. 2022 909 16473147Facile synthesis of cadmium phosphorus trisulfide nanosheets for highly efficient photocatalytic performanceZhang, Yue; Zhao, Yadi; Bao, Cunyou; Xiao, Yue; Xiang, Yuesong; Song, Mengting; Huang, Wenjuan; Ma, Liang; Hou, Huayi; Chen, XiangbaiJournal of Alloys and Compounds (2022), 909 (), 164731CODEN: JALCEU; ISSN:0925-8388. (Elsevier B.V.)With the rise of two-dimensional (2D) materials, layered metal thiophosphates (MPS3), as an important branch, have attracted much attention because of their excellent phys. and chem. properties. Cadmium phosphorus trisulfide (CdPS3), as a promising photocatalyst, can be used for hydrogen evolution and org. degrdn. Herein, high-quality 2D CdPS3 few layers are obtained by exfoliating bulk CdPS3 crystal. The optical properties of the as-obtained CdPS3 few layers show good light-harvesting ability, endowing it with excellent photocatalytic hydrogen evolution performance (10.88 mmol g-1 h-1) and high org. degrdn. performance (more than 92% within 30 min) under Xe lamp irradn. This research provides opportunities for the prepn. and application of other 2D MPS3 type materials in the field of photocatalysis.
- 48Cheng Z. Sendeku M. G. Liu Q. Layered Metal Phosphorous Trichalcogenides Nanosheets: Facile Synthesis and Photocatalytic Hydrogen Evolution Nanotechnology 2020 31 13 13540548Layered metal phosphorous trichalcogenides nanosheets: Facile synthesis and photocatalytic hydrogen evolutionCheng, Zhongzhou; Sendeku, Marshet Getaye; Liu, QuanlinNanotechnology (2020), 31 (13), 135405CODEN: NNOTER; ISSN:1361-6528. (IOP Publishing Ltd.)Layered transition metal phosphorous trichalcogenide (MPX3) materials have attracted immense attention due to their excellent optical and elec. properties. However, the controllable synthesis of ultrathin MPX3 nanosheets is still challenging. Here, we present a facile phosphosulfurization scheme to prep. high-quality layered FePS3 nanosheets, with ~ 20 nm in thickness. The optical properties of the as-obtained FePS3 show good light-harvesting ability, which endows it with excellent photocatalytic hydrogen evolution performance (402.4μmol g-1 h-1) under the simulated solar illumination. We further show that other MPX3 family members such as In2/3PS3 and CdPS3 can be also synthesized by the same approach. Our finding can offer a feasible approach for rationally designing other MPX3 materials for various applications.
- 49Chang X. Wang T. Gong J. CO2 Photo-reduction: Insights into CO2 Activation and Reaction on Surfaces of Photocatalysts Energy Environ. Sci. 2016 9 7 2177 219649CO2 photo-reduction: insights into CO2 activation and reaction on surfaces of photocatalystsChang, Xiaoxia; Wang, Tuo; Gong, JinlongEnergy & Environmental Science (2016), 9 (7), 2177-2196CODEN: EESNBY; ISSN:1754-5706. (Royal Society of Chemistry)Large amts. of anthropogenic CO2 emissions assocd. with increased fossil fuel consumption have led to global warming and an energy crisis. The photocatalytic redn. of CO2 into solar fuels such as methane or methanol is believed to be one of the best methods to address these two problems. In addn. to light harvesting and charge sepn., the adsorption/activation and redn. of CO2 on the surface of heterogeneous catalysts remain a scientifically crit. challenge, which greatly limits the overall photoconversion efficiency and selectivity of CO2 redn. This review describes recent advances in the fundamental understanding of CO2 photoredn. on the surface of heterogeneous catalysts and particularly provides an overview of enhancing the adsorption/activation of CO2 mols. The reaction mechanism and pathways of CO2 redn. as well as their dependent factors are also analyzed and discussed, which is expected to enable an increase in the overall efficiency of CO2 redn. through minimizing the reaction barriers and controlling the selectivity towards the desired products. The challenges and perspectives of CO2 photoredn. over heterogeneous catalysts are presented as well.
- 50Gao W. Li S. He H. Li X. Cheng Z. Yang Y. Wang J. Shen Q. Wang X. Xiong Y. Zhou Y. Zou Z. Vacancy-Defect Modulated Pathway of Photoreduction of CO2 on Single Atomically Thin AgInP2S6 Sheets into Olefiant Gas Nat. Commun. 2021 12 1 474750Vacancy-defect modulated pathway of photoreduction of CO2 on single atomically thin AgInP2S6 sheets into olefiant gasGao, Wa; Li, Shi; He, Huichao; Li, Xiaoning; Cheng, Zhenxiang; Yang, Yong; Wang, Jinlan; Shen, Qing; Wang, Xiaoyong; Xiong, Yujie; Zhou, Yong; Zou, ZhigangNature Communications (2021), 12 (1), 4747CODEN: NCAOBW; ISSN:2041-1723. (Nature Research)Abstr.: Artificial photosynthesis, light-driving CO2 conversion into hydrocarbon fuels, is a promising strategy to synchronously overcome global warming and energy-supply issues. The quaternary AgInP2S6 at. layer with the thickness of ∼ 0.70 nm were successfully synthesized through facile ultrasonic exfoliation of the corresponding bulk crystal. The sulfur defect engineering on this at. layer through a H2O2 etching treatment can excitingly change the CO2 photoredn. reaction pathway to steer dominant generation of ethene with the yield-based selectivity reaching ∼73% and the electron-based selectivity as high as ∼89%. Both DFT calcn. and in-situ FTIR spectra demonstrate that as the introduction of S vacancies in AgInP2S6 causes the charge accumulation on the Ag atoms near the S vacancies, the exposed Ag sites can thus effectively capture the forming *CO mols. It makes the catalyst surface enrich with key reaction intermediates to lower the C-C binding coupling barrier, which facilitates the prodn. of ethene.
- 51Fan Y. Song X. Ai H. Li W. Zhao M. Highly Efficient Photocatalytic CO2 Reduction in Two-Dimensional Ferroelectric CuInP2S6 Bilayers ACS Appl. Mater. Interfaces 2021 13 29 34486 3449451Highly efficient photocatalytic CO2 reduction in two-dimensional ferroelectric CuInP2S6 bilayersFan, Yingcai; Song, Xiaohan; Ai, Haoqiang; Li, Weifeng; Zhao, MingwenACS Applied Materials & Interfaces (2021), 13 (29), 34486-34494CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Photocatalytic CO2 conversion into reproducible chem. fuels (e.g., CO, CH3OH, or CH4) provides a promising scheme to solve the increasing environmental problems and energy demands simultaneously. However, the efficiency is severely restricted by the high overpotential of the CO2 redn. reaction (CO2RR) and rapid recombination of photoexcited carriers. Here, we propose that a novel type-II photocatalytic mechanism based on two-dimensional (2D) ferroelec. multilayers would be ideal for addressing these issues. Using d.-functional theory and nonadiabatic mol. dynamics calcns., we find that the ferroelec. CuInP2S6 bilayers exhibit a staggered band structure induced by the vertical intrinsic elec. fields. Different from the traditional type-II band alignment, the unique structure of the CuInP2S6 bilayer not only effectively suppresses the recombination of photogenerated electron-hole (e-h) pairs but also produces a sufficient photovoltage to drive the CO2RR. The predicted recombination time of photogenerated e-h pairs, 1.03 ns, is much longer than the transferring times of photoinduced electrons and holes, 5.45 and 0.27 ps, resp. Moreover, the overpotential of the CO2RR will decrease by substituting an S atom with a Cu atom, making the redox reaction proceed spontaneously under solar radiation. The solar-to-fuel efficiency with an upper limit of 8.40% is achieved in the CuInP2S6 bilayer and can be further improved to 32.57% for the CuInP2S6 five-layer. Our results indicate that this novel type-II photocatalytic mechanism would be a promising way to achieve highly efficient photocatalytic CO2 conversion based on the 2D ferroelec. multilayers.
- 52De Battisti, A.; Martínez-Huitle, C. A. , Electrocatalysis in Wastewater Treatment. In Electrochemical Water and Wastewater Treatment; Martínez-Huitle, C. A., Rodrigo, M. A., Scialdone, O. , Eds.; Butterworth-Heinemann: 2018; pp 119− 131.There is no corresponding record for this reference.
- 53de Freitas Araujo K. C. da Silva D. R. dos Santos E. V. Varela H. Martinez-Huitle C. A. Investigation of Persulfate Production on BDD Anode by Understanding the Impact of Water Concentration J. Electroanal. Chem. 2020 860 113927There is no corresponding record for this reference.
- 54Huang H. Li F. Xue Q. Zhang Y. Yin S. Chen Y. Salt-Templated Construction of Ultrathin Cobalt Doped Iron Thiophosphite Nanosheets toward Electrochemical Ammonia Synthesis Small 2019 15 51 190350054Salt-Templated Construction of Ultrathin Cobalt Doped Iron Thiophosphite Nanosheets toward Electrochemical Ammonia SynthesisHuang, Hao; Li, Fumin; Xue, Qi; Zhang, Ying; Yin, Shiwei; Chen, YuSmall (2019), 15 (51), 1903500CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)Exploiting efficient electrocatalysts for electrochem. N redn. (NRR) is highly desired and deeply meaningful for realizing sustainable NH3 (NH3) prodn. under ambient conditions. The Fe protein contains one [Fe4S4] cluster and P cluster, which play an important role for transfer electron during the N fixing of nitrogenases. Based on the understanding of nitrogenase, the rising-star 2-dimensional Fe thiophosphite (FePS3) nanomaterials may be highly active electrocatalysts toward NRR due to the ideal elemental compn. 2D FePS3 nanosheets are successfully synthesized by a facile salt-templated method. The FePS3 nanosheets show better electrocatalytic NH3 yield and faradaic efficiency (FE) than Fe2S3, which demonstrates that the P element indeed improves the NRR activity of Fe-S. Theor., Co incorporation not only effectively prompts the cond. of FePS3, but also enhances the catalytic activities of Fe-edge sites. Exptl., Co-doped FePS3 (Co-FePS3) nanosheets exhibit a remarkable electrocatalytic performance toward NRR, such as high NH3 yield rate of 90.6μg h-1 mgcat-1, high FE of 3.38%, and an excellent long-term stability. Being the 1st theor. and exptl. report regarding FePS3-based electrocatalyst toward NRR, this work represents an important beginning to the family of metal thiophosphite as advanced electrocatalysts toward NRR.
- 55Mayorga-Martinez C. C. Sofer Z. Sedmidubsky D. Huber S. Eng A. Y. Pumera M. Layered Metal Thiophosphite Materials: Magnetic, Electrochemical, and Electronic Properties ACS Appl. Mater. Interfaces 2017 9 14 12563 1257355Layered Metal Thiophosphite Materials: Magnetic, Electrochemical, and Electronic PropertiesMayorga-Martinez, Carmen C.; Sofer, Zdenek; Sedmidubsky, David; Huber, Stepan; Eng, Alex Yong Sheng; Pumera, MartinACS Applied Materials & Interfaces (2017), 9 (14), 12563-12573CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Beyond graphene, transitional metal dichalcogenides, and black phosphorus, there are other layered materials called metal thiophosphites (MPSx), which are recently attracting the attention of scientists. Here we present the synthesis, structural and morphol. characterization, magnetic properties, electrochem. performance, and the calcd. d. of states of different layered metal thiophosphite materials with a general formula MPSx, and as a result of varying the metal component, we obtain CrPS4, MnPS3, FePS3, CoPS3, NiPS3, ZnPS3, CdPS3, GaPS4, SnPS3, and BiPS4. SnPS3, ZnPS3, CdPS3, GaPS4, and BiPS4 exhibit only diamagnetic behavior due to core electrons. By contrast, trisulfides with M = Mn, Fe, Co, and Ni, as well as CrPS4, are paramagnetic at high temps. and undergo a transition to antiferromagnetic state on cooling. Within the trisulfides series the Neel temp. characterizing the transition from paramagnetic to antiferromagnetic phase increases with the increasing at. no. and the orbital component enhancing the total effective magnetic moment. Interestingly, in terms of catalysis NiPS3, CoPS3, and BiPS4 show the highest efficiency for hydrogen evolution reaction (HER), while for the oxygen evolution reaction (OER) the highest performance is obsd. for CoPS3. Finally, MnPS3 presents the highest oxygen redn. reaction (ORR) activity compared to the other MPSx studied here. This great catalytic performance reported for these MPSx demonstrates their promising capabilities in energy applications.
- 56Yu Z. Peng J. Liu Y. Liu W. Liu H. Guo Y. Amine-Assisted Exfoliation and Electrical Conductivity Modulation Toward Few-layer FePS3 Nanosheets for Efficient Hydrogen Evolution J. Mater. Chem. A 2019 7 23 13928 1393456Amine-assisted exfoliation and electrical conductivity modulation toward few-layer FePS3 nanosheets for efficient hydrogen evolutionYu, Zhi; Peng, Jing; Liu, Yuhua; Liu, Wenxiu; Liu, Haifeng; Guo, YuqiaoJournal of Materials Chemistry A: Materials for Energy and Sustainability (2019), 7 (23), 13928-13934CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Layered metal phosphorus trichalcogenides (MPX3, M = transition metal, X = S or Se) have been established as new efficient catalysts for water splitting, with FePS3 as one representative example. Here, a novel amine-assisted exfoliation strategy is presented to obtain few-layer FePS3 nanosheets, whose intrinsic elec. cond. can be easily regulated by adjusting the reaction temp. The improved elec. cond. originates from the increase of the Fe3+/Fe2+ ratio, while matrix crystallinity also plays a vital role in this aspect. The combined effect of mixed valence and crystallinity results in superior HER performance with a decrease of the overpotential of 147 mV and a drop of the Tafel slope from 139-94 mV dec-1.
- 57Lian Q. Zhong L. Du C. Luo Y. Zhao J. Zheng Y. Xu J. Ma J. Liu C. Li S. Yan Q. Interfacing Epitaxial Dinickel Phosphide to 2D Nickel Thiophosphate Nanosheets for Boosting Electrocatalytic Water Splitting ACS Nano 2019 13 7 7975 7984There is no corresponding record for this reference.
- 58Gusmão R. Sofer Z. Sedmidubský D. Huber Š. Pumera M. The Role of the Metal Element in Layered Metal Phosphorus Triselenides upon Their Electrochemical Sensing and Energy Applications ACS Catal. 2017 7 12 8159 817058The Role of the Metal Element in Layered Metal Phosphorus Triselenides upon Their Electrochemical Sensing and Energy ApplicationsGusmao, Rui; Sofer, Zdenek; Sedmidubsky, David; Huber, Stepan; Pumera, MartinACS Catalysis (2017), 7 (12), 8159-8170CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)The no. of layered materials seems to be ever-growing, from mono- to multielement, with affiliates and applications being tested continuously. Chalcogenophosphites, also designated as metal phosphorus chalcogenides (MPXn), have attracted great interest because of not only their magnetic properties but also promising capabilities in energy applications. Herein, bulk crystals of different layered metal triselenophosphites, with a general formula MPSe3 (M = Cd, Cr, Fe, Mn, Sn, Zn), were synthesized. Structural and morphol. characterization was performed prior to testing their electrochem. performance. From the set of ternary layered materials, FePSe3, followed by MnPSe3, yielded the highest efficiency for the hydrogen evolution reaction (HER) both in acidic and alk. media with good stability after 100 cycles. MnPSe3 also holds the lowest oxidn. potential for cysteine, although this is due to the presence of MnO2 in the structure as detected by XPS. For the oxygen evolution reaction, the best performance was obsd. for FePSe3, although the stability of the material was not as good as in the case of HER. These findings have profound implications in the application of these layered ternary compds. in energy-related fields.
- 59Hao Y. Huang A. Han S. Huang H. Song J. Sun X. Wang Z. Li L. Hu F. Xue J. Peng S. Plasma-Treated Ultrathin Ternary FePSe3 Nanosheets as a Bifunctional Electrocatalyst for Efficient Zinc–Air Batteries ACS Appl. Mater. Interfaces 2020 12 26 29393 2940359Plasma-Treated Ultrathin Ternary FePSe3 Nanosheets as a Bifunctional Electrocatalyst for Efficient Zinc-Air BatteriesHao, Yanan; Huang, Aijian; Han, Silin; Huang, Hongjiao; Song, Junnan; Sun, Xiaoli; Wang, Zhiguo; Li, Linlin; Hu, Feng; Xue, Jianjun; Peng, ShengjieACS Applied Materials & Interfaces (2020), 12 (26), 29393-29403CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Developing novel bifunctional electrocatalysts with advanced oxygen electrocatalytic activity is pivotal for next-generation energy-storage devices. Herein, we present ultrathin oxygen-doped FePSe3 (FePSe3-O) nanosheets by Ar/O2 plasma treatment, with remarkable surface atom reorganization. Such surface atom reorganization generates multiple cryst.-amorphous interfaces that benefit the kinetics of oxygen evolution reaction, achieving a low overpotential of only 261 mV at 10 mA cm-2 with a small Tafel slope of 41.13 mV dec-1. D. functional theory calcn. indicates that oxygen doping can also modulate the elec. states at the Fermi level with a decreased band gap responsible for the enhanced electrocatalytic performance. Such unique FePSe3-O nanosheets can be further fabricated as the air cathode in rechargeable liq. zinc-air batteries (ZABs), which deliver a high open circuit potential of 1.47 V, a small charge-discharge voltage gap of 0.80 V, and good cycling stability for more than 800 circles. As a proof of concept, the flexible solid-state ZABs assembled with FePSe3-O nanosheets as cathode also display a favorable charge-discharge performance, durable stability, and good bendability. This work sheds new insights into the rational design of defect-rich ternary thiophosphate nanosheets by plasma treatment toward enhanced oxygen electrocatalysts in metal-air batteries.
- 60Konkena B. Masa J. Botz A. J. R. Sinev I. Xia W. Koßmann J. Drautz R. Muhler M. Schuhmann W. Metallic NiPS3@NiOOH Core–Shell Heterostructures as Highly Efficient and Stable Electrocatalyst for the Oxygen Evolution Reaction ACS Catal. 2017 7 1 229 23760Metallic NiPS3@NiOOH Core-Shell Heterostructures as Highly Efficient and Stable Electrocatalyst for the Oxygen Evolution ReactionKonkena, Bharathi; Masa, Justus; Botz, Alexander J. R.; Sinev, Ilya; Xia, Wei; Kossmann, Joerg; Drautz, Ralf; Muhler, Martin; Schuhmann, WolfgangACS Catalysis (2017), 7 (1), 229-237CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)We report metallic NiPS3@NiOOH core-shell heterostructures as an efficient and durable electrocatalyst for the oxygen evolution reaction, exhibiting a low onset potential of 1.48 V (vs. RHE) and stable performance for over 160 h. The atomically thin NiPS3 nanosheets are obtained by exfoliation of bulk NiPS3 in the presence of an ionic surfactant. The OER mechanism was studied by a combination of SECM, in situ Raman spectroscopy, SEM, and XPS measurements, which enabled direct observation of the formation of a NiPS3@NiOOH core-shell heterostructure at the electrode interface. Hence, the active form of the catalyst is represented as NiPS3@NiOOH core-shell structure. Moreover, DFT calcns. indicate an intrinsic metallic character of the NiPS3 nanosheets with densities of states (DOS) similar to the bulk material. The high OER activity of the NiPS3 nanosheets is attributed to a high d. of accessible active metallic-edge and defect sites due to structural disorder, a unique NiPS3@NiOOH core-shell heterostructure, where the presence of P and S modulates the surface electronic structure of Ni in NiPS3, thus providing excellent conductive pathway for efficient electron-transport to the NiOOH shell. These findings suggest that good size control during liq. exfoliation may be advantageously used for the formation of elec. conductive NiPS3@NiOOH core-shell electrode materials for the electrochem. water oxidn.
- 61Bushuyev O. S. De Luna P. Dinh C. T. Tao L. Saur G. van de Lagemaat J. Kelley S. O. Sargent E. H. What Should We Make with CO2 and How Can We Make It ? Joule 2018 2 5 825 83261What Should We Make with CO2 and How Can We Make It?Bushuyev, Oleksandr S.; De Luna, Phil; Dinh, Cao Thang; Tao, Ling; Saur, Genevieve; van de Lagemaat, Jao; Kelley, Shana O.; Sargent, Edward H.Joule (2018), 2 (5), 825-832CODEN: JOULBR; ISSN:2542-4351. (Cell Press)In this forward-looking Perspective, we discuss the current state of technol. and the economics of electrocatalytic transformation of CO2 into various chem. fuels. Our anal. finds that short-chain simple building-block mols. currently present the most economically compelling targets. Making an optimistic prediction of technol. advancement in the future, we propose the gradual rise of photocatalytic, CO2 polymn., biohybrid, and mol. machine technologies to augment and enhance already practical electrocatalytic CO2 conversion methods.
- 62Nitopi S. Bertheussen E. Scott S. B. Liu X. Y. Engstfeld A. K. Horch S. Seger B. Stephens I. E. L. Chan K. Hahn C. Norskov J. K. Jaramillo T. F. Chorkendorff I. Progress and Perspectives of Electrochemical CO2 Reduction on Copper in Aqueous Electrolyte Chem. Rev. 2019 119 12 7610 767262Progress and Perspectives of Electrochemical CO2 Reduction on Copper in Aqueous ElectrolyteNitopi, Stephanie; Bertheussen, Erlend; Scott, Soren B.; Liu, Xinyan; Engstfeld, Albert K.; Horch, Sebastian; Seger, Brian; Stephens, Ifan E. L.; Chan, Karen; Hahn, Christopher; Noerskov, Jens K.; Jaramillo, Thomas F.; Chorkendorff, IbChemical Reviews (Washington, DC, United States) (2019), 119 (12), 7610-7672CODEN: CHREAY; ISSN:0009-2665. (American Chemical Society)A review. To date, copper is the only heterogeneous catalyst that has shown a propensity to produce valuable hydrocarbons and alcs., such as ethylene and ethanol, from electrochem. CO2 redn. (CO2R). There are variety of factors that impact CO2R activity and selectivity, including the catalyst surface structure, morphol., compn., the choice of electrolyte ions and pH, the electrochem. cell design, etc. Many of these factors are often intertwined, which can complicate catalyst discovery and design efforts. Here we take a broad and historical view of these different aspects and their complex interplay in CO2R catalysis on Cu, with the purpose of providing new insights, crit. evaluations, and guidance to the field with regards to research directions and best practices. First, we describe the various exptl. probes and complementary theor. methods that have been used to discern the mechanisms by which products are formed, and next we present our current understanding of the complex reaction networks for CO2R on Cu. Then we analyze two key methods that have been used in attempts to alter the activity and selectivity of Cu: nanostructuring and the formation of bimetallic electrodes. Finally, we offer some perspectives on the future outlook for electrochem. CO2R.
- 63Zhao K. Quan X. Carbon-Based Materials for Electrochemical Reduction of CO2 to C2+ Oxygenates: Recent Progress and Remaining Challenges ACS Catal. 2021 11 4 2076 209763Carbon-Based Materials for Electrochemical Reduction of CO2 to C2+ Oxygenates: Recent Progress and Remaining ChallengesZhao, Kun; Quan, XieACS Catalysis (2021), 11 (4), 2076-2097CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)A review. Electrochem. redn. of CO2 to multicarbon (C2+) products is desirable because of the higher energy d. and economic value of C2+ products and the significant scientific issue for coupling of multicarbons. However, efficient conversion of CO2 into C2+ products remains challenging because of the difficulty in C-C coupling. Recently, numerous papers have reported carbon-based materials for C2+ products prodn. from CO2 electrochem. redn. Because of the unique properties of carbon-based materials in C2+ prodn., carbon-based materials can be used as a potential alternative for the electrocatalytic conversion of CO2. This Review summarized recent progresses in the formation of C2+ oxygenates from CO2 redn. on carbon-based materials. In this Review, we highlighted the strategies available for achieving C-C coupling on carbon-based electrocatalysts and revealed the relationships between intermediate adsorption energy and the selectivity of oxygenate prodn. from CO2 redn. Moreover, we provided the understandings for fabricating active sites of CO2 redn. on carbon-based materials and related mechanisms of C2+ oxygenate generation. The remaining challenges and opportunities for the electrochem. conversion of CO2 into C2+ oxygenates were discussed.
- 64Birdja Y. Y. Pérez-Gallent E. Figueiredo M. C. Göttle A. J. Calle-Vallejo F. Koper M. T. M. Advances and Challenges in Understanding the Electrocatalytic Conversion of Carbon Dioxide to Fuels Nat. Energy 2019 4 9 732 74564Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuelsBirdja, Yuvraj Y.; Perez-Gallent, Elena; Figueiredo, Marta C.; Gottle, Adrien J.; Calle-Vallejo, Federico; Koper, Marc T. M.Nature Energy (2019), 4 (9), 732-745CODEN: NEANFD; ISSN:2058-7546. (Nature Research)A review. The electrocatalytic redn. of carbon dioxide is a promising approach for storing (excess) renewable electricity as chem. energy in fuels. Here, we review recent advances and challenges in the understanding of electrochem. CO2 redn. We discuss existing models for the initial activation of CO2 on the electrocatalyst and their importance for understanding selectivity. Carbon-carbon bond formation is also a key mechanistic step in CO2 electroredn. to high-d. and high-value fuels. We show that both the initial CO2 activation and C-C bond formation are influenced by an intricate interplay between surface structure (both on the nano- and on the mesoscale), electrolyte effects (pH, buffer strength, ion effects) and mass transport conditions. This complex interplay is currently still far from being completely understood. In addn., we discuss recent progress in in situ spectroscopic techniques and computational techniques for mechanistic work. Finally, we identify some challenges in furthering our understanding of these themes.
- 65Ji L. Chang L. Zhang Y. Mou S. Wang T. Luo Y. Wang Z. Sun X. Electrocatalytic CO2 Reduction to Alcohols with High Selectivity over a Two-Dimensional Fe2P2S6 Nanosheet ACS Catal. 2019 9 11 9721 972565Electrocatalytic CO2 Reduction to Alcohols with High Selectivity over a Two-Dimensional Fe2P2S6 NanosheetJi, Lei; Chang, Le; Zhang, Ya; Mou, Shiyong; Wang, Ting; Luo, Yonglan; Wang, Zhiming; Sun, XupingACS Catalysis (2019), 9 (11), 9721-9725CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)Electrochem. conversion of CO2 into alcs. provides an attractive path toward achieving a carbon-neutral cycle, while its efficiency is challenged by identifying active electrocatalysts for the CO2 redn. reaction (CO2RR). In this work, we report the Fe2P2S6 nanosheet acts as an efficient CO2RR electrocatalyst toward highly selective hydrogenation of CO2 to alcs. This catalyst is capable of achieving a high total Faradaic efficiency (FEmethanol+ethanol) of 88.3%, with a FEmethanol up to 65.2% at -0.20 V vs the reversible hydrogen electrode in 0.5 M KHCO3 soln., much higher than most reported CO2RR electrocatalysts. D. functional theory calcns. indicate that Fe atom on the Fe2P2S6 surface can be regared as the active site for alc. formation.
- 66Thompson A. H. Whittingham M. S. Transition Metal Phosphorus Trisulfides as Battery Cathodes Mater. Res. Bull. 1977 12 7 741 74466Transition metal phosphorus trisulfides as battery cathodesThompson, A. H.; Whittingham, M. S.Materials Research Bulletin (1977), 12 (7), 741-4CODEN: MRBUAC; ISSN:0025-5408.The layered, transition-metal P trisulfides exhibit electrochem. activity. NiPS3 reacts with >4 Li resulting in a cell with a theor. energy double that of TiS2.
- 67Brec R. Review on structural and chemical properties of transition metal phosphorous trisulfides MPS3 Solid State Ion. 1986 22 1 3 30There is no corresponding record for this reference.
- 68Foot P. J. S. Katz T. Patel S. N. Nevett B. A. Pieecy A. R. Balchin A. A. The Structures and Conduction Mechanisms of Lithium-intercalated and Lithium-Substituted Nickel Phosphorus Trisulphide (NiPS3), and the Use of the Material as a Secondary Battery Electrode Phys. Status Solidi A 1987 100 1 11 2968The structures and conduction mechanisms of lithium-intercalated and lithium-substituted nickel phosphorus trisulfide (NiPS3), and the use of the material as a secondary battery electrodeFoot, P. J. S.; Katz, T.; Patel, S. N.; Nevett, B. A.; Piercy, A. R.; Balchin, A. A.Physica Status Solidi A: Applied Research (1987), 100 (1), 11-29CODEN: PSSABA; ISSN:0031-8965.A review with 35 refs. The crystal structure and conduction of the title compds. are discussed and the possible use of these NiPS3 compds. as battery electrodes is suggested. An app. is given for the electrochem. lithiation of NiPS3 to form LixNiPS3.
- 69Rouxel J. Brec R. Low-Dimensional Chalcogenides as Secondary Cathodic Materials: Some Geometric and Electronic Aspects Annu. Rev. Mater. Sci. 1986 16 1 137 16269Low-dimensional chalcogenides as secondary cathodic materials: some geometric and electronic aspectsRouxel, J.; Brec, R.Annual Review of Materials Science (1986), 16 (), 137-62CODEN: ARMSCX; ISSN:0084-6600.A review with 45 refs. on electronic and ionic cond. and reversibility of the title cathodic materials for batteries. The geometric aspects assocd. with intercalation (parameter expansion, long distance, and local structures), electronic transfer, and phase transitions are analyzed.
- 70Li X. Wu X. Yang J. Half-Metallicity in MnPSe3 Exfoliated Nanosheet with Carrier Doping J. Am. Chem. Soc. 2014 136 31 11065 1106970Half-Metallicity in MnPSe3 Exfoliated Nanosheet with Carrier DopingLi, Xingxing; Wu, Xiaojun; Yang, JinlongJournal of the American Chemical Society (2014), 136 (31), 11065-11069CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)Searching two-dimensional (2D) half-metallic crystals that are feasible in expt. is essential to develop next-generation nanospintronic devices. Here, a 2-dimensional exfoliated MnPSe3 nanosheet with novel magnetism is 1st proposed based on 1st-principles calcns. In particular, the evaluated low cleavage energy and high in-plane stiffness indicate that the free-standing MnPSe3 nanosheet can be exfoliated from its bulk structure in expt. The MnPSe3 nanosheet is an antiferromagnetic semiconductor at its ground state, whereas both electron and hole doping induce its transition from antiferromagnetic semiconductor to ferromagnetic half-metal. Also, the spin-polarization directions of 2-dimensional half-metallic MnPSe3 are opposite for electron and hole doping, which can be controlled by applying an external voltage gate. The Monte Carlo simulation based on the Ising model suggests the Curie temp. of the doped 2-dimensional MnPSe3 crystal is up to 206 K. These advantages render the 2-dimensional MnPSe3 crystal with great potentials for application in elec.-field controlled spintronic devices.
- 71Cabria I. El-Meligi A. A. DFT Simulation of Hydrogen Storage on Manganese Phosphorous Trisulphide (MnPS3) Int. J. Hydrogen Energy 2018 43 11 5903 591271DFT simulation of hydrogen storage on manganese phosphorous trisulfide (MnPS3)Cabria, I.; El-Meligi, A. A.International Journal of Hydrogen Energy (2018), 43 (11), 5903-5912CODEN: IJHEDX; ISSN:0360-3199. (Elsevier Ltd.)Manganese phosphorous trisulfide, MnPS3, is a solid layered material. The H2 gravimetric storage capacity of powd. MnPS3 at 80.15, 173.15, and 298.15° K and at moderate pressures was recently exptl. measured. The origin of storage capacity of this material is not well understood. The main hypothesis is that H2 is stored in powd. MnPS3 pores. Pores were modeled as two parallel MnPS3 layers sepd. by a certain distance. D. functional theory (DFT) simulations of the H2 interaction with the MnPS3 layer surface were performed to test this hypothesis. Simulations indicated H2 adsorption on the MnPS3 layer surface was energetically favorable, but only by a physisorption mechanism. Gravimetric capacity calcns. of powd. MnPS3 pores were also performed and were in reasonable agreement with exptl. results. A comparison of calcd. and exptl. gravimetric capacity showed that H2 storage on powd. MnPS3 was mainly due to in-pore compression; the contribution of the physisorption process to storage was very small.
- 72Kuzminskii Y. V. Voronin B. M. Redin N. N. Iron and Nickel Phosphorus Trisulfides as Electroactive Materials for Primary Lithium Batteries J. Power Sources 1995 55 2 133 14172Iron and nickel phosphorus trisulfides as electroactive materials for primary lithium batteriesKuzminskii, Y. V.; Voronin, B. M.; Redin, N. N.Journal of Power Sources (1995), 55 (2), 133-41CODEN: JPSODZ; ISSN:0378-7753. (Elsevier)The theor. specific capacities of a family of layered compds. MPX3 (M = Fe, Co, Ni; X = S, Se) for current-producing reactions involving 1.5, 2, 6 and 9 lithium atoms per MPX3 mol. have been estd. These data show that FePS3 and NiPS3 are good electroactive materials for primary lithium cells. Conditions which allow one to shorten substantially the synthesis time of iron and nickel phosphorus trisulfides in ampules are presented. According to cond. measurements in the 293-673 K temp. range, NiPS3 is an intrinsic and FePS3 an extrinsic semiconductor. The discharge characteristics of Li/MPS3 (M = Fe, Ni) cells with an org. electrolyte have been studied; they confirmed that the participation of nine electrons in the redox process is possible. For a primary button cell of the std. size 2325, with a FePS3 cathode, the specific capacity and specific energy values obtained in a discharge 1.8-1.2 V range at load resistances of 3.0 to 30 kΩ were 500-1160 Ah/kg and 700-1770 Wh/kg (on a pure FePS3 basis), resp.
- 73Kuzminskii Y. V. Voronin B. M. Petrushina I. M. Redin N. N. Prikhodko G. P. Nickel Phosphorus Trisulfide: An Electroactive Material for Medium-Temperature Lithium Batteries J. Power Sources 1995 55 1 1 6There is no corresponding record for this reference.
- 74Liang Q. Zheng Y. Du C. Luo Y. Zhang J. Li B. Zong Y. Yan Q. General and Scalable Solid-State Synthesis of 2D MPS3 (M = Fe, Co, Ni) Nanosheets and Tuning Their Li/Na Storage Properties Small Methods 2017 1 12 1700304There is no corresponding record for this reference.
- 75Fujii Y. Miura A. Rosero-Navarro N. C. Higuchi M. Tadanaga K. FePS3 Electrodes in All-Solid-State Lithium Secondary Batteries Using Sulfide-Based Solid Electrolytes Electrochim. Acta 2017 241 370 37475FePS3 electrodes in all-solid-state lithium secondary batteries using sulfide-based solid electrolytesFujii, Yuta; Miura, Akira; Rosero-Navarro, Nataly Carolina; Higuchi, Mikio; Tadanaga, KiyoharuElectrochimica Acta (2017), 241 (), 370-374CODEN: ELCAAV; ISSN:0013-4686. (Elsevier Ltd.)In bulk-type all-solid-state Li batteries with sulfide-based solid electrolytes, composite electrodes, in which an active material, a solid electrolyte, and a conductive additive are mixed, were used to enhance Li-ion diffusion and electronic cond. However, the addn. of electrolytes and electron-conductive additives to the composite electrodes decreases the amt. of active materials in the batteries. FePS3 was employed as the electrode in an all-solid-state Li secondary battery, without mixing the solid electrolytes and conductive additives. The all-solid-state cell using the FePS3 electrode exhibited reversible charge-discharge behavior for >30 cycles under a const. c.d. of 0.13 mA cm-2 at room temp. The discharge capacity of the cell was 107 mAh g-1 at the 30th cycle. This behavior was comparable to that of a cell with an electrode including a solid electrolyte and/or a conductive additive, indicating that the FePS3 electrode had sufficient paths of Li ions and of electron conduction. Probably FePS3 is an attractive Fe-based electrode for an all-solid-state battery using a sulfide-based solid electrolyte.
- 76Liang Q. Zheng Y. Du C. Luo Y. Zhao J. Ren H. Xu J. Yan Q. Asymmetric-Layered Tin Thiophosphate: An Emerging 2D Ternary Anode for High-Performance Sodium Ion Full Cell ACS Nano 2018 12 12 12902 1291176Asymmetric-Layered Tin Thiophosphate: An Emerging 2D Ternary Anode for High-Performance Sodium Ion Full CellLiang, Qinghua; Zheng, Yun; Du, Chengfeng; Luo, Yubo; Zhao, Jin; Ren, Hao; Xu, Jianwei; Yan, QingyuACS Nano (2018), 12 (12), 12902-12911CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)The emerging Na ion batteries (SIBs) are believed to be prospective substitutes for Li ion batteries (LIBs) because of the wide distribution of Na resources. However, to compensate for the sluggish reaction kinetics and higher intrinsic potential of Na+ compared to Li+, cost-effective, reliable, and sustainable electrode materials must be explored for practical applications. Here, 2D ternary tin thiophosphate (SnP2S6) nanosheets (∼10 nm thickness) grown on graphene (denoted as SPS/G hybrid) are demonstrated as intriguing anodes for SIBs. The asym.-layered structure and ternary compn. enable the SPS/G hybrid with a high reversible capacity (1230 mAh/g at 50 mA/g), superior rate capability (200 mAh/g at 15 A/g), and an exceptional capacity retention of 76% after 1000 cycles at 2.0 A/g. A prototype Na-ion full cell constructed by pairing with the Na3V2O2(PO4)3F cathode affords a high capacity of 470 mAh/g at 30 mA/g (on the basis of anode wt.) and good cyclic capacity of 360 mAh/g at 150 mA/g. Such 2D ternary chalcogenides with low-cost elements are promising materials for superior SIBs.
- 77Brec, R. , Review on Structural and Chemical Properties of Transition Metal Phosphorus Trisulfides MPS3. In Intercalation in Layered Materials; Dresselhaus, M. S. , Ed.; Springer US: Boston, MA, 1986; pp 93− 124.There is no corresponding record for this reference.
- 78Clement R. P. Garnier O. Jegoudez J. Coordination Chemistry of the Lamellar MPS3 Materials: Metal-Ligand Cleavage as the Source of an Unusual Cation-Transfer Intercalation Process Inorg. Chem. 1986 25 1404 140978Coordination chemistry of the lamellar MPS3 materials: metal-ligand cleavage as the source of an unusual "cation-transfer" intercalation processClement, Rene; Garnier, Odile; Jegoudez, JocelyneInorganic Chemistry (1986), 25 (9), 1404-9CODEN: INOCAJ; ISSN:0020-1669.Several MPS3 lamellar materials exhibit a very unusual intercalation chem. based on a cation-transfer process between the solid and a solvent. This contrasts with the electron-transfer processes that governs the intercalation chem. of the layered dichalcogenides MX2. Although the MPS3 layers are made up of infinite arrays of P2S64- units coordinated to the M2+ cations, the metal-ligand (M-S) bonding still exhibits enough lability to allow the M2+ cations to jump, under very mild conditions, from intra- toward interlamellar sites (or the opposite) and even to be removed from the material, provided a suitable cation is available to intercalate the lattice and maintain elec. neutrality. Depending on the metal, the solvent, and the guest species, the reaction may take place spontaneously or require an assist, usually by complexing the leaving M2+ cations (EDTA). Small inserted ions (Na+, K+, etc.) are strongly solvated and mobile, and they can in turn be exchanged. To rationalize this behavior, it is suggested that the intercalation chem. of the MPS3 involves a chem. transformation of the whole of the material, rather than a diffusional process of the guest species within the host lattice. The MPS3 layered materials are considered as polynuclear complexes that can undergo heterogeneous equil. with their constitutive species M2+ and P2S64- and other species present in soln. In support of this mechanism, MPS3 materials were successfully prepd. by mixing aq. solns. of Na4P2S6 and transition-metal salts. Some implications of this mechanism are discussed.
- 79Wang F. He J. Speeding Protons with Metal Vacancies Science 2020 370 6516 525 52679Speeding protons with metal vacanciesWang, Fengmei; He, JunScience (Washington, DC, United States) (2020), 370 (6516), 525-526CODEN: SCIEAS; ISSN:1095-9203. (American Association for the Advancement of Science)Proton exchange membranes (PEMs) find applications not only in fuel cells and sensors but as chem. filters and in biol. systems. In particular, the Nafion (Chemours Company) PEM, which consists of sulfonated tetrafluoroethylene-based fluoropolymer-copolymer, is widely used in practical electrochem. processes. It has a high proton cond. up to 0.2 S/cm below 80°C and high relative humidity (RH) (>93%), but its cond. drops severely at higher temps. or at low RH (below 50%). On page 596 of this issue, Qian et al. report a new class of proton membranes assembled from two-dimensional (2D) layered transition-metal phosphorus trichalcogenide (TMPTC) nanosheets in which metal vacancies boost ion cond. These membranes exhibit a proton cond. of ∼0.95 S/cm at 90°C and 98% RH but still have a cond. of 0.26 S/cm even at 60% RH.



