Structure-Activity Mechanism of Iron Oxide Nanozymes
- Ruofei ZhangRuofei ZhangCAS Engineering Laboratory for Nanozyme, Key Laboratory of Protein and Peptide Pharmaceutical, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, ChinaR.Z. and H.Z. contributed equally to this work.More by Ruofei Zhang
- ,
- Hanqing ZhaoHanqing ZhaoCAS Engineering Laboratory for Nanozyme, Key Laboratory of Protein and Peptide Pharmaceutical, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, ChinaUniversity of Chinese Academy of Sciences, Chinese Academy of Sciences, Beijing 100408, ChinaR.Z. and H.Z. contributed equally to this work.More by Hanqing Zhao
- , and
- Kelong Fan*Kelong Fan*Email: [email protected]CAS Engineering Laboratory for Nanozyme, Key Laboratory of Protein and Peptide Pharmaceutical, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, ChinaUniversity of Chinese Academy of Sciences, Chinese Academy of Sciences, Beijing 100408, ChinaNanozyme Medical Center, School of Basic Medical Sciences, Zhengzhou University, Zhengzhou 450052, ChinaMore by Kelong Fan
Abstract
The discovery of highly stable and low-cost nanozymes, nanomaterials with enzyme-like activity, opens a new portal for breaking through the application limitations of delicate natural enzymes. Iron oxide nanozymes, as one of the most representative nanozymes, were first discovered to exhibit enzyme-like activity, and were systematically and in-depth studied. Researches have found some structural factors that have an important impact on the catalytic activity. In this chapter, we introduce the enzyme-like activities of iron oxide nanozymes and systematically review the elucidated structure-activity relationships to gain insight into their enzyme-like catalytic mechanisms.
This publication is licensed for personal use by The American Chemical Society.
Introduction
Scheme 1
Enzyme-Like Activities of Iron Oxide Nanozymes
Peroxidase-Like Activity
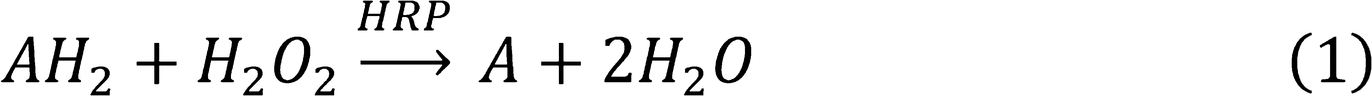
Figure 1
Figure 2
Catalase-Like Activity
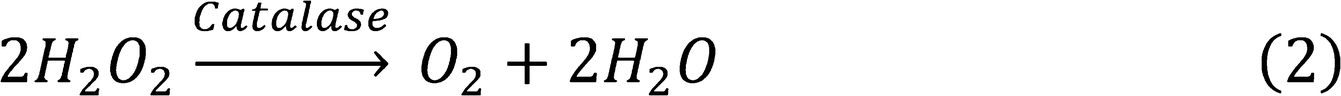
Figure 3
Figure 4
Enzymatic Features of Iron Oxide Nanozymes
High Catalytic Activity Following Enzymatic Kinetics


Figure 5
Figure 6
| Nanozymes | Activity | Substrate | KM (mM) | Vmax (M·s-1) | kcat (s-1) | Ref. |
|---|---|---|---|---|---|---|
| Fe3O4 MNPs | Peroxidase | TMB | 0.098 | 3.44×10-8 | 3.02×104 | ( 11) |
| H2O2 | 154 | 9.78×10-8 | 8.58×104 | |||
| Fe3O4 nanotubes | Peroxidase | TMB | 4.147 | 5.2×10−6 | 9.81 × 105 | ( 48) |
| CoFe2O4@ graphene | Peroxidase | TMB | 0.046 | 1.121×10-8 | / | ( 49) |
| H2O2 | 14.72 | 21.71×10-8 | / | |||
| Fe3O4 | Peroxidase | TMB | 0.150~0.493 | 0.3×10-6~3.4×10-6 | 3.3~56.1 | ( 50) |
| ABTS | 0.093~0.360 | 0.4×10-6~6.5×10-6 | 6.6~108.3 | |||
| GOD-Fe3O4 NPs | Peroxidase | H2O2 | 10.93 | 4.22×10-8 | / | ( 51) |
| Fe3O4@MoS2-Ag | Peroxidase | TMB | 0.19 | 1.12×10-7 | / | ( 52) |
| H2O2 | 1 | 1.82×10-7 | / | |||
| Fe3O4\~NPs | Peroxidase | TMB | 0.27 | / | ( 53) | |
| MWNTs@MNPs | Peroxidase | H2O2 | 0.55 | 23×10-8 | / | ( 54) |
| CoFe2O4 MNP | Peroxidase | H2O2 | 0.0752 | / | / | ( 55) |
| α-Fe2O3 | Peroxidase | TMB | 0.257 | 1.65×10-8 | 5.5 | ( 56) |
| Fe3O4@Pt | Peroxidase | TMB | 0.147 | 0.711×10-7 | 84.112 | ( 57) |
| H2O2 | 702.6 | 7.136×10-7 | 84.420 | |||
| Fe3O4-Au | Peroxidase | TMB | 0.0106 | / | / | ( 58) |
| H2O2 | 0.0344 | / | ||||
| GO-Fe2O3 hybrids | Peroxidase | TMB | 0.118 | 5.38×10-8 | / | ( 59) |
| H2O2 | 305 | 1.01×10-7 | / | |||
| ABTS | 0.153 | 2.50×10-8 | / | |||
| H2O2 | 13.8 | 2.43×10-8 | / | |||
| Iron oxide/Pd NPs | Peroxidase | TMB | 0.11 | / | / | ( 60) |
| His-Fe3O4 | Peroxidase | H2O2 | 37.99 | 5.28×10-9 | 5.39×105 | ( 31) |
| Fe3O4@3D GN | Peroxidase | TMB | 0.103 | 11.6×10-8 | / | ( 61) |
| H2O2 | 1.39 | 10.1×10-8 | / | |||
| Fe3O4 | Peroxidase | TMB | 0.374 | 2.6×10-8 | / | ( 62) |
| H2O2 | 54.6 | 1.8×10-8 | / | |||
| Cu(HBTC)-1/Fe3O4 -AuNPs | Peroxidase | TMB | 0.22 | 15.05×10-7 | / | ( 63) |
| H2O2 | 0.03 | 12.13×10-7 | / | |||
| CoFe2O4 | Peroxidase | TMB | 0.387 | 2.90×10-8 | / | ( 64) |
| H2O2 | 8.89 | 1.93×10-8 | / | |||
| PRGI/Pt nanohybrid | Peroxidase | TMB | 2.37 | 97.1×10-8 | / | ( 65) |
| H2O2 | 3.56 | 36.6×10-8 | / | |||
| 3D GF/ Fe3O4 | Peroxidase | TMB | 0.059 | 4.87×10-7 | / | ( 66) |
| H2O2 | 0.65 | 9.43×10-7 | / | |||
| Fe2O3 Mesoporous | Peroxidase | TMB | 0.298 | 7.36×10-8 | / | ( 67) |
| H2O2 | 146.7 | 6.37×10-8 | / | |||
| Fe2O3 nanocubes | Peroxidase | TMB | 0.214 | 2.21×10-8 | / | ( 68) |
| H2O2 | 115 | 2.89×10-8 | / | |||
| CM-PtNP@m− Fe3O4 | Peroxidase | TMB | 0.257 | 19.5×10-8 | / | ( 69) |
| H2O2 | 0.036 | 31.5×10-8 | / | |||
| Fe3O4@LNPs | Peroxidase | TMB | 0.51 | 1.03×10-8 | / | ( 70) |
| H2O2 | 5.30 | 0.96×10-8 | / | |||
| Fe3O4 | Peroxidase | TMB | 0.155~0.490 | 0.2×10-6~3.22×10-6 | 3.1~49.2 | ( 71) |
| ABTS | 0.101~0.395 | 0.5×10-6~6.4×10-6 | 7.5~97.6 | |||
| IONF | Peroxidase | TMB | 0.24~0.44 | 1.3×10-8~3.07×10-8 | / | ( 72) |
| H2O2 | 150.47~216.08 | 1.12×10-8~3.12×10-8 | ||||
| Co@ Fe3O4 | Peroxidase | TMB | 1.17 | 37.9×10-8 | / | ( 73) |
| H2O2 | 0.19 | 71.5×10-8 | / | |||
| PtFe@ Fe3O4 NRs | Peroxidase | TMB | 0.213 | 5.477×10-8 | / | ( 74) |
| H2O2 | 53.55 | 1.078×10-8 | / | |||
| Cu/Fe3O4@FeOOH | Peroxidase | TMB | 1.369 | 5.61×10-8 | / | ( 75) |
| H2O2 | 1.087 | 5.66×10-8 | / | |||
| Fe3O4–MnO2 | Peroxidase | TMB | 0.101 | 0.57×10-8 | / | ( 76) |
| H2O2 | 0.041 | 2.94×10-8 | / | |||
| or-ZnFe2O4/rGO | Peroxidase | TMB | 0.117 | 4.15×10-8 | / | ( 77) |
| H2O2 | 0.06 | 2.61×10-8 | / | |||
| Mesoporous γ-Fe2O3 | Peroxidase | TMB | 0.0997 | 5.20×10-8 | / | ( 78) |
| H2O2 | 144.30 | 1.84×10-8 | / | |||
| Mesoporous α- Fe2O3 | TMB | 0.5304 | 5.43×10-8 | / | ||
| H2O2 | 127.92 | 3.77×10-8 | / | |||
| Fe3O4 with DA | Peroxidase | TMB | 0.2445 | 0.8255×10-8 | 0.579×105 | ( 79) |
| H2O2 | 54.04 | 1.66×10-8 | 1.165×105 | |||
| TiO2@MoS2/Co Fe2O4 | Peroxidase | TMB | 0.123 | / | / | ( 80) |
| H2O2 | 3.898 | / | / | |||
| Fe3O4 NPs | Peroxidase | TMB | 0.46 | 11×10-8 | / | ( 81) |
| H2O2 | 58.9 | 8.6×10-8 | / | |||
| VB2-IONzymes | Peroxidase | TMB | 0.4369~0.9781 | 84.4×10-9~126.7×10-9 | / | ( 82) |
| H2O2 | 261.2~304.2 | 79.07×10-9~261.7×10-9 | / | |||
| Catalase | H2O2 | 319.2~ 1558 | 1.737×10-9~2.494×10-9 | / | ||
| Fe3O4@SiO2-NH2-Au@Pd0.30NPs | Peroxidase | TMB | 0.090 | 11.2×10-8 | / | ( 83) |
| H2O2 | 3.5 | 6.76×10-8 | / | |||
| PTCA-ZnFe2O4 | Peroxidase | TMB | 0.2217 | 7.801×10-8 | / | ( 84) |
| H2O2 | 0.8343 | 1.144×10-8 | / | |||
| AgNPs@ Fe3O4 | Peroxidase | OPD | 2.91 | / | / | ( 85) |
| H2O2 | 28 | / | / | |||
| IONzymes | Peroxidase | TMB | 0.148~ 0.267 | 2.70×10-7~6.12×10-8 | 6.43×104~1.46×104 | ( 86) |
| H2O2 | 1165~ 2200 | 2.07×10-7~8.52×10-8 | 4.93×104~1.90×104 | |||
| MnFe2O4/g-C3N4 | Peroxidase | TMB | 0.0106 | 28.40×10-8 | / | ( 87) |
| H2O2 | 0.0396 | 43.76×10-8 | / | |||
| FDGs | Peroxidase | TMB | 0.242 | 3.9×10-8 | / | ( 88) |
| H2O2 | 0.089 | 5.55×10-8 | / | |||
| MnFe2O4 | Peroxidase | MBTH | 0.01359 | 5.25×107 | ( 89) | |
| IONPs | Peroxidase | TMB | 3.34×10-5~16.13×10-5 | 1.62×10-6~7.47×10-6 | / | ( 90) |
| H2O2 | 0.596~2.699 | 2.12×10-3~16.99×10-3 | / | |||
| Pt/ZnFe2O4/C | Peroxidase | TMB | 0.041 | 7.58×10-8 | / | ( 91) |
| H2O2 | 0.66 | 13.42×10-8 | / | |||
| CTAB@ ZnFe2O4 | Peroxidase | OPD | 1.01 | 2.55×10-8 | / | ( 92) |
| CuFe2O4@PPy | Peroxidase | TMB | 0.0654 | 19.63×10-8 | / | ( 93) |
| H2O2 | 3.89 | 0.323×10-8 | / | |||
| Fe3O4@C@MnO2 | Peroxidase | TMB | 0.113 | 19.6×10-8 | / | ( 94) |
| Fe3O4 | Peroxidase | TMB | 0.24-0.71 | 0.42×10-7~2.40×10-7 | 0.2×104~1.14×104 | ( 95) |
| ABTS | 0.12-0.73 | 0.52×10-7~6.10×10-7 | 0.25×104~2.9×104 | |||
| CoFe2O4 | Peroxidase | TMB | 0.03551~0.22769 | 4.3×10-4~8.3×10-3 | / | ( 96) |
| H2O2 | 0.00645~0.03476 | 2.64×10-4~1.376×10-3 | / | |||
| MgFe2O4MNPs | Peroxidase | H2O2 | 4.61 | 13.46×10-8 | / | ( 97) |
| TMB | 0.67 | 2.09×10-8 | / | |||
| ABTS | 0.14 | 12.54×10-8 | / | |||
| NiFe2O4 MNPs | H2O2 | 2.60 | 14.11×10-8 | / | ||
| TMB | 0.55 | 4.57×10-8 | / | |||
| ABTS | 0.46 | 13.48×10-8 | / | |||
| 3D graphene/ Fe3O4–AuNPs | Peroxidase | TMB | 0.2 | 4.6×10-7 | / | ( 98) |
| H2O2 | 0.2 | 1.5×10-7 | / | |||
| Fe3O4@C YSNs | Peroxidase | TMB | 0.27 | 12.0×10-8 | / | ( 99) |
| H2O2 | 0.035 | 3.34×10-8 | / | |||
| Fe3O4@CeO2 NCs | Peroxidase | TMB | 0.15 | 0.64×10-8 | / | ( 100) |
| H2O2 | 1.13 | 12.5×10-8 | / | |||
| γ-Fe2O3/CeO2-PDI | Peroxidase | TMB | 0.097 | 5.13×10-8 | / | ( 101) |
| H2O2 | 0.310 | 1.68×10-8 | / | |||
| Ferrihydrite | Catalase | H2O2 | 40.6 | 3.04×10-6 | / | ( 46) |
| NPPLGA | Catalase | H2O2 | 0.76 | 22.32×10-3 | / | ( 102) |
| PEGylated CoFe2O4 nanoflowers | Catalase | H2O2 | 34.58 | 2.33×10-6 | 54.67 | ( 103) |
| Co-Fe3O4 | Catalase | H2O2 | 13.44 | 16.05×10-6 | / | ( 104) |
Regulatory Mechanism
Figure 7
Figure 8
Controllable Multi-Enzyme Activity
Recyclable and Versatility
Figure 9
Rational Design and Synthesis
Figure 10
Structure-Activity Regulation of Iron Oxide Nanozymes
Size
Figure 11
Morphology and Crystal Planes
Figure 12
Surface Modification
Figure 13
Composition
Surface Valence
Other Factors
Conclusion and Perspective
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 82122037, 31900981), National Key Research and Development Program of China (No. 2021YFC2102900), CAS Interdisciplinary Innovation Team (JCTD-2020-08), Youth Innovation Promotion Association of Chinese Academy of Sciences (2019093) and China Postdoctoral Science Foundation (2021M703420).
References
This chapter references 140 other publications.
- 1Schwertmann, U.; Cornell, R. M. Iron Oxides in the Laboratory: Preparation and Characterization; John Wiley & Sons, 2008.Google ScholarThere is no corresponding record for this reference.
- 2Cornell, R. M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences, and Uses; Wiley-VCH Weinheim, 2003; Vol. 2.Google ScholarThere is no corresponding record for this reference.
- 3Sivula K. Le Formal F. Grätzel M. Solar water splitting: progress using hematite (α‐Fe2O3) photoelectrodes ChemSusChem 2011 4 4 432 449Google Scholar3Solar Water Splitting: Progress Using Hematite (α-Fe2O3) PhotoelectrodesSivula, Kevin; Le Formal, Florian; Graetzel, MichaelChemSusChem (2011), 4 (4), 432-449CODEN: CHEMIZ; ISSN:1864-5631. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. Photoelectrochem. cells offer the ability to convert electromagnetic energy from our largest renewable source, the Sun, to stored chem. energy through the splitting of water into mol. oxygen and hydrogen. Hematite (α-Fe2O3) has emerged as a promising photoelectrode material due to its significant light absorption, chem. stability in aq. environments, and ample abundance. However, its performance as a water-oxidizing photoanode has been crucially limited by poor optoelectronic properties that lead to both low light harvesting efficiencies and a large requisite overpotential for photoassisted water oxidn. Recently, the application of nanostructuring techniques and advanced interfacial engineering has afforded landmark improvements in the performance of hematite photoanodes. In this review, new insights into the basic material properties, the attractive aspects, and the challenges in using hematite for photoelectrochem. water splitting are first examd. Next, recent progress enhancing the photocurrent by precise morphol. control and reducing the overpotential with surface treatments are critically detailed and compared. The latest efforts using advanced characterization techniques, particularly electrochem. impedance spectroscopy, are finally presented. These methods help to define the obstacles that remain to be surmounted in order to fully exploit the potential of this promising material for solar energy conversion.
- 4Reddy M. Yu T. Sow C.-H. Shen Z. X. Lim C. T. Subba Rao G. Chowdari B. α‐Fe2O3 nanoflakes as an anode material for Li‐ion batteries Adv. Funct. Mater. 2007 17 15 2792 2799Google Scholar4α-Fe2O3 nanoflakes as an anode material for Li-ion batteriesReddy, M. V.; Yu, Ting; Sow, Chorng-Haur; Shen, Ze Xiang; Lim, Chwee Teck; Rao, G. V. Subba; Chowdari, B. V. R.Advanced Functional Materials (2007), 17 (15), 2792-2799CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)Nanoflakes of α-Fe2O3 were prepd. on Cu foil by using a thermal treatment method. The nanoflakes were characterized by X-ray diffraction, SEM, high-resoln. transmission electron microscopy, and Raman spectroscopy. The reversible Li-cycling properties of the α-Fe2O3 nanoflakes have been evaluated by cyclic voltammery, galvanostatic discharge-charge cycling, and impedance spectral measurements on cells with Li metal as the counter and ref. electrodes, at ambient temp. Results show that Fe2O3 nanoflakes exhibit a stable capacity of (680 ± 20) mA h g-1, corresponding to (4.05 ± 0.05) moles of Li per mol of Fe2O3 with no noticeable capacity fading up to 80 cycles when cycled in the voltage range 0.005-3.0 V at 65 mA g-1 (0.1 C rate), and with a coulombic efficiency of >98% during cycling (after the 15th cycle). The av. discharge and charge voltages are 1.2 and 2.1 V, resp. The obsd. cyclic voltammograms and impedance spectra have been analyzed and interpreted in terms of the 'conversion reaction' involving nanophase Fe0-Li2O. The superior performance of Fe2O3 nanoflakes is clearly established by a comparison of the results with those for Fe2O3 nanoparticles and nanotubes reported in the literature.
- 5Chen J. Xu L. Li W. Gou X. α‐Fe2O3 nanotubes in gas sensor and lithium‐ion battery applications Adv. Mater. 2005 17 5 582 586Google Scholar5α-Fe2O3 nanotubes in gas sensor and lithium-ion battery applicationsChen, Jun; Xu, Lina; Li, Weiyang; Gou, XinglongAdvanced Materials (Weinheim, Germany) (2005), 17 (5), 582-586CODEN: ADVMEW; ISSN:0935-9648. (Wiley-VCH Verlag GmbH & Co. KGaA)Templated arrays of α-Fe2O3 nanotubes, produced using an alumina membrane, exhibit enhanced sensitivity to ethanol and hydrogen as well as enhanced electrochem. activity in lithium-ion batteries. The nanotubes, characterized by uniform size and shape (see Figure), possess a high sp. surface area, leading to electrochem. activities much higher than that of bulk or nanoparticulate α-Fe2O3.
- 6Gupta A. K. Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications Biomaterials 2005 26 18 3995 4021Google Scholar6Synthesis and surface engineering of iron oxide nanoparticles for biomedical applicationsGupta, Ajay Kumar; Gupta, MonaBiomaterials (2005), 26 (18), 3995-4021CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)A review. Superparamagnetic iron oxide nanoparticles (SPION) with appropriate surface chem. have been widely used exptl. for numerous in vivo applications such as magnetic resonance imaging contrast enhancement, tissue repair, immunoassay, detoxification of biol. fluids, hyperthermia, drug delivery and in cell sepn., etc. All these biomedical and bioengineering applications require that these nanoparticles have high magnetization values and size smaller than 100 nm with overall narrow particle size distribution, so that the particles have uniform phys. and chem. properties. In addn., these applications need special surface coating of the magnetic particles, which has to be not only non-toxic and biocompatible but also allow a targetable delivery with particle localization in a specific area. To this end, most work in this field has been done in improving the biocompatibility of the materials, but only a few scientific investigations and developments have been carried out in improving the quality of magnetic particles, their size distribution, their shape and surface in addn. to characterizing them to get a protocol for the quality control of these particles. Nature of surface coatings and their subsequent geometric arrangement on the nanoparticles det. not only the overall size of the colloid but also play a significant role in biokinetics and biodistribution of nanoparticles in the body. The types of specific coating, or derivatization, for these nanoparticles depend on the end application and should be chosen by keeping a particular application in mind, whether it be aimed at inflammation response or anti-cancer agents. Magnetic nanoparticles can bind to drugs, proteins, enzymes, antibodies, or nucleotides and can be directed to an organ, tissue, or tumor using an external magnetic field or can be heated in alternating magnetic fields for use in hyperthermia. This review discusses the synthetic chem., fluid stabilization and surface modification of superparamagnetic iron oxide nanoparticles, as well as their use for above biomedical applications.
- 7Weissleder R. Stark D. D. Engelstad B. L. Bacon B. R. Compton C. C. White D. L. Jacobs P. Lewis J. Superparamagnetic iron oxide: pharmacokinetics and toxicity Am. J. Roentgenol. 1989 152 1 167 173Google Scholar7Superparamagnetic iron oxide: pharmacokinetics and toxicityWeissleder R; Stark D D; Engelstad B L; Bacon B R; Compton C C; White D L; Jacobs P; Lewis JAJR. American journal of roentgenology (1989), 152 (1), 167-73 ISSN:0361-803X.The pharmacokinetics (distribution, metabolism, bioavailability, excretion) and toxicity (acute and subacute toxicity, mutagenicity) of a superparamagnetic iron oxide preparation (AMI-25), currently used in clinical trials, were evaluated by 59Fe radiotracer studies, measurements of relaxation times, the ability to reverse iron deficiency anemia, histologic examination, and laboratory parameters. One hour after administration of AMI-25 to rats (18 mumol Fe/kg; 1 mg Fe/kg), 82.6 +/- 0.3% of the administered dose was sequestered in the liver and 6.2 +/- 7.6% in the spleen. Peak concentrations of 59Fe were found in liver after 2 hr and in the spleen after 4 hr. 59Fe slowly cleared from liver (half-life, 3 days) and spleen (half-life, 4 days) and was incorporated into hemoglobin of erythrocytes in a time-dependent fashion. The half-time of the T2 effect on liver and spleen (24-48 hr) was shorter than the 59Fe clearance, indicating metabolism of AMI-25 into other forms of iron. IV administration of AMI-25 (30 mg Fe/kg) corrected iron-deficiency anemia and showed bioavailability similar to that of commercially available IV iron preparations within 7 days. No acute or subacute toxic effects were detected by histologic or serologic studies in rats or beagle dogs who received a total of 3000 mumol Fe/kg, 150 times the dose proposed for MR imaging of the liver. Our results indicate that AMI-25 is a fully biocompatible potential contrast agent for MR.
- 8Weinstein J. S. Varallyay C. G. Dosa E. Gahramanov S. Hamilton B. Rooney W. D. Muldoon L. L. Neuwelt E. A. Superparamagnetic iron oxide nanoparticles: diagnostic magnetic resonance imaging and potential therapeutic applications in neurooncology and central nervous system inflammatory pathologies, a review J. Cereb. Blood Flow Metab. 2010 30 1 15 35Google Scholar8Superparamagnetic iron oxide nanoparticles: diagnostic magnetic resonance imaging and potential therapeutic applications in neurooncology and central nervous system inflammatory pathologies, a reviewWeinstein, Jason S.; Varallyay, Csanad G.; Dosa, Edit; Gahramanov, Seymur; Hamilton, Bronwyn; Rooney, William D.; Muldoon, Leslie L.; Neuwelt, Edward A.Journal of Cerebral Blood Flow & Metabolism (2010), 30 (1), 15-35CODEN: JCBMDN; ISSN:0271-678X. (Nature Publishing Group)A review. Superparamagnetic iron oxide nanoparticles have diverse diagnostic and potential therapeutic applications in the central nervous system (CNS). They are useful as magnetic resonance imaging (MRI) contrast agents to evaluate: areas of blood-brain barrier (BBB) dysfunction related to tumors and other neuroinflammatory pathologies, the cerebrovasculature using perfusion-weighted MRI sequences, and in vivo cellular tracking in CNS disease or injury. Novel, targeted, nanoparticle synthesis strategies will allow for a rapidly expanding range of applications in patients with brain tumors, cerebral ischemia or stroke, carotid atherosclerosis, multiple sclerosis, traumatic brain injury, and epilepsy. These strategies may ultimately improve disease detection, therapeutic monitoring, and treatment efficacy esp. in the context of antiangiogenic chemotherapy and antiinflammatory medications. The purpose of this review is to outline the current status of superparamagnetic iron oxide nanoparticles in the context of biomedical nanotechnol. as they apply to diagnostic MRI and potential therapeutic applications in neurooncol. and other CNS inflammatory conditions.
- 9Dulińska-Litewka J. Łazarczyk A. Hałubiec P. Szafrański O. Karnas K. Karewicz A. Superparamagnetic iron oxide nanoparticles—Current and prospective medical applications Materials 2019 12 4 617Google Scholar9Superparamagnetic iron oxide nanoparticles- current and prospective medical applicationsDulinska-Litewka, Joanna; Lazarczyk, Agnieszka; Halubiec, Przemyslaw; Szafranski, Oskar; Karnas, Karolina; Karewicz, AnnaMaterials (2019), 12 (4), 617CODEN: MATEG9; ISSN:1996-1944. (MDPI AG)The recent, fast development of nanotechnol. is reflected in the medical sciences. Superparamagnetic Iron Oxide Nanoparticles (SPIONs) are an excellent example. Thanks to their superparamagnetic properties, SPIONs have found application in Magnetic Resonance Imaging (MRI) and magnetic hyperthermia. Unlike bulk iron, SPIONs do not have remnant magnetization in the absence of the external magnetic field; therefore, a precise remote control over their action is possible. This makes them also useful as a component of the advanced drug delivery systems. Due to their easy synthesis, biocompatibility, multifunctionality, and possibility of further surface modification with various chem. agents, SPIONs could support many fields of medicine. SPIONs have also some disadvantages, such as their high uptake by macrophages. Nevertheless, based on the ongoing studies, they seem to be very promising in oncol. therapy (esp. in the brain, breast, prostate, and pancreatic tumors). The main goal of our paper is, therefore, to present the basic properties of SPIONs, to discuss their current role in medicine, and to review their applications in order to inspire future developments of new, improved SPION systems.
- 10Min Y. Caster J. M. Eblan M. J. Wang A. Z. Clinical translation of nanomedicine Chem. Rev. 2015 115 19 11147 11190Google Scholar10Clinical Translation of NanomedicineMin, Yuanzeng; Caster, Joseph M.; Eblan, Michael J.; Wang, Andrew Z.Chemical Reviews (Washington, DC, United States) (2015), 115 (19), 11147-11190CODEN: CHREAY; ISSN:0009-2665. (American Chemical Society)A review discussing the clin. translation of nanomedicine, including liposome and lipid-based nanomedicine, protein nanoparticles, polymer-drug conjugates, polymeric micelles and nanoparticles, inorg. nanoparticles, and summary and outlook.
- 11Gao L. Zhuang J. Nie L. Zhang J. Zhang Y. Gu N. Wang T. Feng J. Yang D. Perrett S. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles Nat. Nanotechnol. 2007 2 9 577 583Google Scholar11Intrinsic peroxidase-like activity of ferromagnetic nanoparticlesGao, Lizeng; Zhuang, Jie; Nie, Leng; Zhang, Jinbin; Zhang, Yu; Gu, Ning; Wang, Taihong; Feng, Jing; Yang, Dongling; Perrett, Sarah; Yan, XiyunNature Nanotechnology (2007), 2 (9), 577-583CODEN: NNAABX; ISSN:1748-3387. (Nature Publishing Group)Nanoparticles contg. magnetic materials, such as magnetite (Fe3O4), are particularly useful for imaging and sepn. techniques. As these nanoparticles are generally considered to be biol. and chem. inert, they are typically coated with metal catalysts, antibodies or enzymes to increase their functionality as sepn. agents. Here, we report that magnetite nanoparticles in fact possess an intrinsic enzyme mimetic activity similar to that found in natural peroxidases, which are widely used to oxidize org. substrates in the treatment of wastewater or as detection tools. Based on this finding, we have developed a novel immunoassay in which antibody-modified magnetite nanoparticles provide three functions: capture, sepn. and detection. The stability, ease of prodn. and versatility of these nanoparticles makes them a powerful tool for a wide range of potential applications in medicine, biotechnol. and environmental chem.
- 12Wei H. Wang E. Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymes Chem. Soc. Rev. 2013 42 14 6060 93Google Scholar12Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymesWei, Hui; Wang, ErkangChemical Society Reviews (2013), 42 (14), 6060-6093CODEN: CSRVBR; ISSN:0306-0012. (Royal Society of Chemistry)A review. Over the past few decades, researchers have established artificial enzymes as highly stable and low-cost alternatives to natural enzymes in a wide range of applications. A variety of materials including cyclodextrins, metal complexes, porphyrins, polymers, dendrimers and biomols. have been extensively explored to mimic the structures and functions of naturally occurring enzymes. Recently, some nanomaterials have been found to exhibit unexpected enzyme-like activities, and great advances have been made in this area due to the tremendous progress in nano-research and the unique characteristics of nanomaterials. To highlight the progress in the field of nanomaterial-based artificial enzymes (nanozymes), this review discusses various nanomaterials that have been explored to mimic different kinds of enzymes. We cover their kinetics, mechanisms and applications in numerous fields, from biosensing and immunoassays, to stem cell growth and pollutant removal. We also summarize several approaches to tune the activities of nanozymes. Finally, we make comparisons between nanozymes and other catalytic materials (other artificial enzymes, natural enzymes, org. catalysts and nanomaterial-based catalysts) and address the current challenges and future directions (302 refs.).
- 13Lin Y. Ren J. Qu X. Catalytically active nanomaterials: a promising candidate for artificial enzymes Acc. Chem. Res. 2014 47 4 1097 105Google Scholar13Catalytically active nanomaterials: A promising candidate for artificial enzymesLin, Youhui; Ren, Jinsong; Qu, XiaogangAccounts of Chemical Research (2014), 47 (4), 1097-1105CODEN: ACHRE4; ISSN:0001-4842. (American Chemical Society)A review. Natural enzymes, exquisite biocatalysts mediating every biol. process in living organisms, are able to accelerate the rate of chem. reactions up to 1019-fold for specific substrates and reactions. However, the practical application of enzymes is often hampered by their intrinsic drawbacks, such as low operational stability, sensitivity of catalytic activity to environmental conditions, and high costs in prepn. and purifn. Therefore, the discovery and development of artificial enzymes is highly desired. Recently, the merging of nanotechnol. with biol. has ignited extensive research efforts for designing functional nanomaterials that exhibit various properties intrinsic to enzymes. As a promising candidate for artificial enzymes, catalytically active nanomaterials (nanozymes) show several advantages over natural enzymes, such as controlled synthesis in low cost, tunability in catalytic activities, as well as high stability against stringent conditions. Here, the authors focus on their recent progress in exploring and constructing such nanoparticulate artificial enzymes, including graphene oxide, graphene-hemin nanocomposites, carbon nanotubes, carbon nanodots, mesoporous silica-encapsulated gold nanoparticles, gold nanoclusters, and nanoceria. According to their structural characteristics, these enzyme mimics have been categorized into 3 classes: carbon-based, metal-based, and metal-oxide-based nanomaterials. The authors highlight the important role of catalytic nanomaterials in the fields of biomimetics. First, the authors provide a practical introduction to the identification of these nanozymes, the source of the enzyme-like activities, and the enhancement of activities via rational design and engineering. Then, the authors briefly describe new or enhanced applications of certain nanozymes in biomedical diagnosis, environmental monitoring, and therapeutics. For instance, the authors have successfully used these biomimetic catalysts as colorimetric probes for the detection of cancer cells, nucleic acids, proteins, metal ions, and other small mols. In addn., the authors also introduce 3 exciting advances in the use of efficient modulators on artificial enzyme systems to improve the catalytic performance of existing nanozymes. For example, the authors report that graphene oxide could serve as a modulator to greatly improve the catalytic activity of lysozyme-stabilized gold nanoclusters at neutral pH, which will have great potential for applications in biol. systems. The authors show that, through the incorporation of modulators into artificial enzymes, they can offer a facile but highly effective way to improve their overall catalytic performance or realize catalytic reactions that were not possible in the past. The authors expect that nanozymes with unique properties and functions will attract increasing research interest and lead to new opportunities in various fields of research.
- 14Gao L. Yan X. Nanozymes: an emerging field bridging nanotechnology and biology Sci. China: Life Sci. 2016 59 4 400 2Google Scholar14Nanozymes: an emerging field bridging nanotechnology and biologyGao Lizeng; Yan Xiyun; Gao LizengScience China. Life sciences (2016), 59 (4), 400-2 ISSN:.There is no expanded citation for this reference.
- 15Ragg R. Tahir M. N. Tremel W. Solids Go Bio: Inorganic Nanoparticles as Enzyme Mimics Eur. J. Inorg. Chem. 2016 2016 13-14 1906 1915Google Scholar15Solids Go Bio: Inorganic Nanoparticles as Enzyme MimicsRagg, Ruben; Tahir, Muhammad N.; Tremel, WolfgangEuropean Journal of Inorganic Chemistry (2016), 2016 (13-14), 1906-1915CODEN: EJICFO; ISSN:1434-1948. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. A longstanding goal of biomimetic chem. is the design and synthesis of functional enzyme mimics. The past three decades have seen a wide variety of materials, including metal complexes, polymers and other biomols., that mimic the structures and functions of naturally occurring enzymes. Among these, inorg. nanoparticles offer huge potential, because they are more stable than their natural counterparts, while having large surface areas and sizes comparable to those of natural enzymes. Therefore, a considerable no. of "artificial enzymes" derived from inorg. nanomaterials have been reported. This microreview highlights the recent progress in the field of enzymically active inorg. nanomaterials, including mimics of peroxidases, haloperoxidases, superoxide dismutases and sulfite oxidases, along with selected biotechnol. applications and their future prospects.
- 16Wang X. Hu Y. Wei H. Nanozymes in bionanotechnology: from sensing to therapeutics and beyond Inorg. Chem. Front. 2016 3 1 41 60Google Scholar16Nanozymes in bionanotechnology: from sensing to therapeutics and beyondWang, Xiaoyu; Hu, Yihui; Wei, HuiInorganic Chemistry Frontiers (2016), 3 (1), 41-60CODEN: ICFNAW; ISSN:2052-1553. (Royal Society of Chemistry)In the past few decades, researchers have developed lots of artificial enzymes with various materials to mimic the structures and functions of natural enzymes. Recently, nanozymes, nanomaterials with enzyme-like characteristics, are emerging as novel artificial enzymes, and attracting researchers' enormous interest. Remarkable advances have been made in the area of nanozymes due to their unique properties compared with natural enzymes and classic artificial enzymes. Until now, lots of nanomaterials have been studied to mimic various natural enzymes for wide applications. To highlight the recent progress of nanozymes (esp. in bionanotechnol.), here we discuss the diverse applications of nanozymes, which range from sensing, imaging, and therapeutics, to logic gates, pollutant removal, water treatment, etc. Finally, we address the current challenges facing nanozyme research as well as possible directions to fulfill their great potential in future.
- 17Tang G. He J. Liu J. Yan X. Fan K. Nanozyme for tumor therapy: Surface modification matters Exploration 2021 1 1 75 89Google ScholarThere is no corresponding record for this reference.
- 18Lee J. Liao H. Wang Q. Han J. Han J.-H. Shin H. E. Ge M. Park W. Li F. Exploration of nanozymes in viral diagnosis and therapy Exploration 2022 2 1 20210086Google ScholarThere is no corresponding record for this reference.
- 19Yan, X.; Gao, L. Nanozymology; Springer, 2020.Google ScholarThere is no corresponding record for this reference.
- 20Wang Z. Zhang R. Yan X. Fan K. Structure and activity of nanozymes: Inspirations for de novo design of nanozymes Mater. Today 2020 41 81 119Google Scholar20Structure and activity of nanozymes: Inspirations for de novo design of nanozymesWang, Zhuoran; Zhang, Ruofei; Yan, Xiyun; Fan, KelongMaterials Today (Oxford, United Kingdom) (2020), 41 (), 81-119CODEN: MTOUAN; ISSN:1369-7021. (Elsevier Ltd.)A review on structure and activity of nanozymes, Inspirations for de novo design of nanozymes. Nanozymes, nanomaterials with enzyme-like activities, are becoming powerful competitors and potential substitutes for natural enzymes because of their excellent performance, including design from scratch, controllable activity, and environmental resistance. In recent years, various nanozymes have been discovered or designed, and gradually applied to mol. detection, biomedical treatment and environmental management. Nevertheless, nanozymes are often regarded as fascinating and confusing black boxes as their catalytic mechanisms remain largely indistinct. Interestingly, recent researches have shed light into these black boxes. It appears that the enzymic activities of nanozymes are closely related to their size, surface lattice, surface modification and compn., etc. Some regular structure-activity relationships have been elucidated in recent reports. In this review, we systematically summarized the studies on the structure-activity relationship of nanozymes in recent years, aiming to illustrate the catalytic mechanism of nanozymes and clarify the key factors regulating their behavior, so as to provide ideas and inspiration for the de novo design of nanozymes.
- 21Jiang B. Duan D. Gao L. Zhou M. Fan K. Tang Y. Xi J. Bi Y. Tong Z. Gao G. F. Standardized assays for determining the catalytic activity and kinetics of peroxidase-like nanozymes Nat. Protoc. 2018 13 7 1506 1520Google Scholar21Standardized assays for determining the catalytic activity and kinetics of peroxidase-like nanozymesJiang, Bing; Duan, Demin; Gao, Lizeng; Zhou, Mengjie; Fan, Kelong; Tang, Yan; Xi, Juqun; Bi, Yuhai; Tong, Zhou; Gao, George Fu; Xie, Ni; Tang, Aifa; Nie, Guohui; Liang, Minmin; Yan, XiyunNature Protocols (2018), 13 (7), 1506-1520CODEN: NPARDW; ISSN:1750-2799. (Nature Research)Nanozymes are nanomaterials exhibiting intrinsic enzyme-like characteristics that have increasingly attracted attention, owing to their high catalytic activity, low cost and high stability. This combination of properties has enabled a broad spectrum of applications, ranging from biol. detection assays to disease diagnosis and biomedicine development. Since the intrinsic peroxidase activity of Fe3O4 nanoparticles (NPs) was first reported in 2007, >40 types of nanozymes have been reported that possess peroxidase-, oxidase-, haloperoxidase- or superoxide dismutase-like catalytic activities. Given the complex interdependence of the physicochem. properties and catalytic characteristics of nanozymes, it is important to establish a std. by which the catalytic activities and kinetics of various nanozymes can be quant. compared and that will benefit the development of nanozyme-based detection and diagnostic technologies. Here, we first present a protocol for measuring and defining the catalytic activity units and kinetics for peroxidase nanozymes, the most widely used type of nanozyme. In addn., we describe the detailed exptl. procedures for a typical nanozyme strip-based biol. detection test and demonstrate that nanozyme-based detection is repeatable and reliable when guided by the presented nanozyme catalytic std. The catalytic activity and kinetics assays for a nanozyme can be performed within 4 h.
- 22Gao L. Fan K. Yan X. Iron Oxide Nanozyme: A Multifunctional Enzyme Mimetic for Biomedical Applications Theranostics 2017 7 13 3207 3227Google Scholar22Iron oxide nanozyme: a multifunctional enzyme mimetic for biomedical applicationsGao, Lizeng; Fan, Kelong; Yan, XiyunTheranostics (2017), 7 (13), 3207-3227CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Iron oxide nanoparticles have been widely used in many important fields due to their excellent nanoscale phys. properties, such as magnetism/superparamagnetism. They are usually assumed to be biol. inert in biomedical applications. However, iron oxide nanoparticles were recently found to also possess intrinsic enzyme-like activities, and are now regarded as novel enzyme mimetics. A special term, "Nanozyme", has thus been coined to highlight the intrinsic enzymic properties of such nanomaterials. Since then, iron oxide nanoparticles have been used as nanozymes to facilitate biomedical applications. In this review, we will introduce the enzymic features of iron oxide nanozyme (IONzyme), and summarize its novel applications in biomedicine.
- 23Chen Z. Yin J.-J. Zhou Y.-T. Zhang Y. Song L. Song M. Hu S. Gu N. Dual enzyme-like activities of iron oxide nanoparticles and their implication for diminishing cytotoxicity ACS Nano 2012 6 5 4001 4012Google Scholar23Dual Enzyme-like Activities of Iron Oxide Nanoparticles and Their Implication for Diminishing CytotoxicityChen, Zhongwen; Yin, Jun-Jie; Zhou, Yu-Ting; Zhang, Yu; Song, Lina; Song, Mengjie; Hu, Sunling; Gu, NingACS Nano (2012), 6 (5), 4001-4012CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)Iron oxide nanoparticles (IONPs) are frequently used in biomedical applications, yet their toxic potential is still a major concern. While most studies of biosafety focus on cellular responses after exposure to nanomaterials, little is reported to analyze reactions on the surface of nanoparticles as a source of cytotoxicity. Here we report that different intracellular microenvironment in which IONPs are located leads to contradictive outcomes in their abilities to produce free radicals. We first verified pH-dependent peroxidase-like and catalase-like activities of IONPs and investigated how they interact with hydrogen peroxide (H2O2) within cells. Results showed that IONPs had a concn.-dependent cytotoxicity on human glioma U251 cells, and they could enhance H2O2-induced cell damage dramatically. By conducting ESR spectroscopy expts., we showed that both Fe3O4 and γ-Fe2O3 nanoparticles could catalyze H2O2 to produce hydroxyl radicals in acidic lysosome mimic conditions, with relative potency Fe3O4 > γ-Fe2O3, which was consistent with their peroxidase-like activities. However, no hydroxyl radicals were obsd. in neutral cytosol mimic conditions with both nanoparticles. Instead, they decompd. H2O2 into H2O and O2 directly in this condition through catalase-like activities. Transmission electron micrographs revealed that IONPs located in lysosomes in cells, the acidic environment of which may contribute to hydroxyl radical prodn. This is the first study regarding cytotoxicity based on their enzyme-like activities. Since H2O2 is continuously produced in cells, our data indicate that lysosome-escaped strategy for IONP delivery would be an efficient way to diminish long-term toxic potential.
- 24Dolphin D. Forman A. Borg D. Fajer J. Felton R. Compounds I of catalase and horse radish peroxidase: π-cation radicals Proc. Natl. Acad. Sci. 1971 68 3 614 618Google Scholar24Compounds I of catalase and horse radish peroxidase: πcation radicalsDolphin, David; Forman, A.; Borg, Douglas C.; Fajer, J.; Felton, Ronald H.Proceedings of the National Academy of Sciences of the United States of America (1971), 68 (3), 614-18CODEN: PNASA6; ISSN:0027-8424.Two-electron oxidn. of cobaltous octaethylporphyrin [Co(II)(Et)8P] yields a stable π-cation radical [Co(III)(Et)8P]2+, the optical spectrum of which exhibits spectral changes dependent upon the nature of the counterion. Comparison of these spectra with those of Compds. I of horseradish peroxidase and catalase suggests that these Compds. I contain a π-cation radical of the heme prosthetic group. This proposal explains the oxidn. level, optical spectra, and stability of the primary compds. without recourse to properties such as stoichiometric mixts. of special porphyrins, stable Fe(V) porphyrins, or unique conformers of heme porphyrins. Explanations are advanced to account for the missing ESR signal of Compd. I of horseradish peroxidase.
- 25Veitch N. C. Horseradish peroxidase: a modern view of a classic enzyme Phytochemistry 2004 65 3 249 259Google Scholar25Horseradish peroxidase: a modern view of a classic enzymeVeitch, Nigel C.Phytochemistry (Elsevier) (2004), 65 (3), 249-259CODEN: PYTCAS; ISSN:0031-9422. (Elsevier Science B.V.)A review. Horseradish peroxidase (HRP) is an important heme-contg. enzyme that has been studied for more than a century. In recent years, new information has become available on the 3-dimensional structure of the enzyme and its catalytic intermediates, mechanisms of catalysis, and the function of specific amino acid residues. Site-directed mutagenesis and directed evolution techniques are now used routinely to investigate the structure and function of HRP and offer the opportunity to develop engineered enzymes for practical applications in natural product and fine chems. synthesis, medical diagnostics, and bioremediation. A combination of HRP and indole-3-acetic acid (IAA) or its derivs. is currently being evaluated as an agent for use in targeted cancer therapies. Physiol. roles traditionally assocd. with the enzyme that include IAA metab., crosslinking of biol. polymers, and lignification are becoming better understood at the mol. level, but the involvement of specific HRP isoenzymes in these processes is not yet clearly defined. Progress in this area should result from the identification of the entire peroxidase gene family of Arabidopsis thaliana, which has now been completed.
- 26Rodríguez-López J. N. Lowe D. J. Hernández-Ruiz J. Hiner A. N. García-Cánovas F. Thorneley R. N. Mechanism of reaction of hydrogen peroxide with horseradish peroxidase: identification of intermediates in the catalytic cycle J. Am. Chem. Soc. 2001 123 48 11838 11847Google Scholar26Mechanism of reaction of hydrogen peroxide with horseradish peroxidase: Identification of intermediates in the catalytic cycleRodriguez-Lopez, Jose Neptuno; Lowe, David J.; Hernandez-Ruiz, Josefa; Hiner, Alexander N. P.; Garcia-Canovas, Francisco; Thorneley, Roger N. F.Journal of the American Chemical Society (2001), 123 (48), 11838-11847CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)The mechanism of the reaction of horseradish peroxidase isoenzyme C (HRPC) with H2O2 to form reactive enzyme intermediate compd. I was studied using electronic absorbance, rapid-scan stopped-flow, and ESR spectroscopies at both acid and basic pH. The roles of active site residues His-42 and Arg-38 in controlling heterolytic cleavage of the H2O2 O-O bond were probed with site-directed mutant enzymes H42L, R38L, and R38G. The biphasic reaction kinetics of H42L with H2O2 suggested the presence of an intermediate species and, at acid pH, a reversible 2nd step, probably due to a neutral enzyme-H2O2 complex and the ferric-peroxoanion-contg. compd. 0. ESR also indicated the formation of a protein radical situated more than ∼10 Å from the heme Fe. The stoichiometry of the reaction of the H42L/H2O2 reaction product and 2,2'-azinobis(3-ethylbenzothiazolinesulfonic acid) (ABTS) was concn.-dependent and fell from a value of 2 to 1 above 0.7 mM ABTS. These data could be explained if H2O2 underwent homolytic cleavage in H42L. The apparent rate of compd. I formation by H42L, while low, was pH-independent in contrast to wild-type HRPC where the rate fell at acid pH, indicating the involvement of an ionizable group with pKa of ∼4. In R38L and R38G, the apparent pKa was shifted to ∼8, but there was no evidence that homolytic cleavage of H2O2 occurred. These data suggest that His-42 acts initially as a proton acceptor (base catalyst) and then as a donor (acid catalyst) at neutral pH and predict the obsd. slower rate and lower efficiency of heterolytic cleavage obsd. at acid pH. Arg-38 was influential in lowering the pKa of His-42 and addnl. in aligning H2O2 in the active site, but it did not play a direct role in proton transfer.
- 27Berglund G. I. Carlsson G. H. Smith A. T. Szöke H. Henriksen A. Hajdu J. The catalytic pathway of horseradish peroxidase at high resolution Nature 2002 417 6887 463 468Google Scholar27The catalytic pathway of horseradish peroxidase at high resolutionBerglund, Gunnar I.; Carlsson, Gunilla H.; Smith, Andrew T.; Szoeke, Hanna; Henriksen, Anette; Hajdu, JanosNature (London, United Kingdom) (2002), 417 (6887), 463-468CODEN: NATUAS; ISSN:0028-0836. (Nature Publishing Group)A mol. description of O2 and H2O2 activation in biol. systems is difficult, because electrons liberated during x-ray data collection reduce the active centers of redox enzymes catalyzing these reactions. Here, the authors describe an effective strategy to obtain crystal structures for high-valency redox intermediates and present a 3-dimensional movie of the x-ray-driven catalytic redn. of a bound O2 species in horseradish peroxidase (HRP). The authors also describe sep. expts. in which high-resoln. structures could be obtained for all 5 oxidn. states of HRP, showing such structures with preserved redox states for the 1st time.
- 28Yuan B. Chou H. L. Peng Y. K. Disclosing the Origin of Transition Metal Oxides as Peroxidase (and Catalase) Mimetics ACS Appl. Mater. Interfaces 2021 1 9Google ScholarThere is no corresponding record for this reference.
- 29Wang N. Zhu L. Wang D. Wang M. Lin Z. Tang H. Sono-assisted preparation of highly-efficient peroxidase-like Fe3O4 magnetic nanoparticles for catalytic removal of organic pollutants with H2O2 Ultrason. Sonochem. 2010 17 3 526 533Google Scholar29Sono-assisted preparation of highly-efficient peroxidase-like Fe3O4 magnetic nanoparticles for catalytic removal of organic pollutants with H2O2Wang, Nan; Zhu, Li-Hua; Wang, Da-Li; Wang, Ming-Qiong; Lin, Zhi-Fen; Tang, He-QingUltrasonics Sonochemistry (2010), 17 (3), 526-533CODEN: ULSOER; ISSN:1350-4177. (Elsevier B.V.)Fe3O4 magnetic nanoparticles (Fe3O4 MNPs) with much improved peroxidase-like activity were successfully prepd. through an advanced reverse co-pptn. method under the assistance of ultrasound irradn. The characterizations with XRD, BET and SEM indicated that the ultrasound irradn. in the prepn. induced the prodn. of Fe3O4 MNPs possessing smaller particle sizes (16.5 nm), greater BET surface area (82.5 m2 g-1) and much higher dispersibility in water. The particle sizes, BET surface area, chem. compn. and then catalytic property of the Fe3O4 MNPs could be tailored by adjusting the initial concn. of ammonia water and the molar ratio of Fe2+/Fe3+ during the prepn. process. The H2O2-activating ability of Fe3O4 MNPs was evaluated by using Rhodamine B (RhB) as a model compd. of org. pollutants to be degraded. At pH 5.4 and temp. 40 °C, the sonochem. synthesized Fe3O4 MNPs were obsd. to be able to activate H2O2 and remove ca. 90% of RhB (0.02 mmol L-1) in 60 min with a apparent rate const. of 0.034 min-1 for the RhB degrdn., being 12.6 folds of that (0.0027 min-1) over the Fe3O4 MNPs prepd. via a conventional reverse co-pptn. method. The mechanisms of the peroxidase-like catalysis with Fe3O4 MNPs were discussed to develop more efficient novel catalysts.
- 30Zhang J. Zhuang J. Gao L. Zhang Y. Gu N. Feng J. Yang D. Zhu J. Yan X. Decomposing phenol by the hidden talent of ferromagnetic nanoparticles Chemosphere 2008 73 9 1524 1528Google Scholar30Decomposing phenol by the hidden talent of ferromagnetic nanoparticlesZhang, Jinbin; Zhuang, Jie; Gao, Lizeng; Zhang, Yu; Gu, Ning; Feng, Jing; Yang, Dongling; Zhu, Jingdong; Yan, XiyunChemosphere (2008), 73 (9), 1524-1528CODEN: CMSHAF; ISSN:0045-6535. (Elsevier Ltd.)Researches on modified Fenton reactions applied in phenol degrdn. have been focused on reducing secondary pollution and enhancing catalytic efficiency. Newly developed methods utilizing carriers, such as Resin and Nafion, to immobilize Fe2+ could avoid iron ion leakage. However, the requirement of high temp. and the limited reaction efficiency still restrained them from broad application. Based on a recently discovered "hidden talent" of ferromagnetic nanoparticles (MNPs), we established a MNP-catalyzed phenol removal assay, which could overcome these limitations. Our results showed that the MNPs removed over 85% phenol from aq. soln. within 3 h even at 16°. The catalytic condition was extensively optimized among a range of pH, temp. as well as initial concn. of phenol and H2O2. TOC and GC/MS anal. revealed that about 30% phenol was mineralized while the rest became small mol. org. acids. Moreover the MNPs were thermo-stable and could be regenerated for at least five rounds. Thus, our findings open up a wide spectrum of environmental friendly applications of MNPs showing several attractive features, such as easy prepn., low cost, thermo-stability and reusability.
- 31Fan K. Wang H. Xi J. Liu Q. Meng X. Duan D. Gao L. Yan X. Optimization of Fe3O4 nanozyme activity via single amino acid modification mimicking an enzyme active site Chem. Commun. 2017 53 2 424 427Google Scholar31Optimization of Fe3O4 nanozyme activity via single amino acid modification mimicking an enzyme active siteFan, Kelong; Wang, Hui; Xi, Juqun; Liu, Qi; Meng, Xiangqin; Duan, Demin; Gao, Lizeng; Yan, XiyunChemical Communications (Cambridge, United Kingdom) (2017), 53 (2), 424-427CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)The Fe3O4 nanozyme was the 1st reported nanoparticle with intrinsic peroxidase-like activity and has been widely used in biomedicine. Here, to optimize its catalytic activity, the authors introduced His residues onto the Fe3O4 nanoparticle surface in order to mimic the enzymic microenvironment of natural peroxidases. The results showed that modification with a single amino acid could improve the apparent affinity (Km) of the Fe3O4 nanozyme by >10-fold for the substrate, H2O2, and enhance its catalytic efficiency (kcat/Km) by ≤20-fold. Thus, the authors not only optimized the activity of the Fe3O4 nanozyme, but also provided a new rationale for improving the efficiency of nanomaterial-based catalysts by utilizing strategies obsd. in Nature.
- 32Cheon H. J. Nguyen Q. H. Kim M. I. Highly Sensitive Fluorescent Detection of Acetylcholine Based on the Enhanced Peroxidase-Like Activity of Histidine Coated Magnetic Nanoparticles Nanomaterials 2021 11 5 1207Google Scholar32Highly sensitive fluorescent detection of acetylcholine based on the enhanced peroxidase-like activity of histidine coated magnetic nanoparticlesCheon, Hong Jae; Nguyen, Quynh Huong; Kim, Moon IlNanomaterials (2021), 11 (5), 1207CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Inspired by the active site structure of natural horseradish peroxidase having iron as a pivotal element with coordinated histidine residues, we have developed histidine coated magnetic nanoparticles (His@MNPs) with relatively uniform and small sizes (less than 10 nm) through one-pot heat treatment. In comparison to pristine MNPs and other amino acid coated MNPs, His@MNPs exhibited a considerably enhanced peroxidase-imitating activity, approaching 10-fold higher in catalytic reactions. With the high activity, His@MNPs then were exploited to detect the important neurotransmitter acetylcholine. By coupling choline oxidase and acetylcholine esterase with His@MNPs as peroxidase mimics, target choline and acetylcholine were successfully detected via fluorescent mode with high specificity and sensitivity with the limits of detection down to 200 and 100 nM, resp. The diagnostic capability of the method is demonstrated by analyzing acetylcholine in human blood serum. This study thus demonstrates the potential of utilizing His@MNPs as peroxidase-mimicking nanozymes for detecting important biol. and clin. targets with high sensitivity and reliability.
- 33Duan D. Fan K. Zhang D. Tan S. Liang M. Liu Y. Zhang J. Zhang P. Liu W. Qiu X. Nanozyme-strip for rapid local diagnosis of Ebola Biosens. Bioelectron. 2015 74 134 141Google Scholar33Nanozyme-strip for rapid local diagnosis of EbolaDuan, Demin; Fan, Kelong; Zhang, Dexi; Tan, Shuguang; Liang, Mifang; Liu, Yang; Zhang, Jianlin; Zhang, Panhe; Liu, Wei; Qiu, Xiangguo; Kobinger, Gary P.; Gao, George Fu; Yan, XiyunBiosensors & Bioelectronics (2015), 74 (), 134-141CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)Ebola continues to rage in West Africa. In the absence of an approved vaccine or treatment, the priority in controlling this epidemic is to promptly identify and isolate infected individuals. To this end, a rapid, highly sensitive, and easy-to-use test for Ebola diagnosis is urgently needed. Here, by using Fe3O4 magnetic nanoparticle (MNP) as a nanozyme probe, we developed a MNP-based immunochromatog. strip (Nanozyme-strip), which detects the glycoprotein of Ebola virus (EBOV) as low as 1 ng/mL, which is 100-fold more sensitive than the std. strip method. The sensitivity of the Nanozyme-strip for EBOV detection and diagnostic accuracy for New Bunyavirus clin. samples is comparable with ELISA, but is much faster (within 30 min) and simpler (without need of specialist facilities). The results demonstrate that the Nanozyme-strip test can rapidly and sensitively detect EBOV, providing a valuable simple screening tool for diagnosis of infection in Ebola-stricken areas.
- 34Fan K. Cao C. Pan Y. Lu D. Yang D. Feng J. Song L. Liang M. Yan X. Magnetoferritin nanoparticles for targeting and visualizing tumour tissues Nat. Nanotechnol. 2012 7 7 459 464Google Scholar34Magnetoferritin nanoparticles for targeting and visualizing tumour tissuesFan, Kelong; Cao, Changqian; Pan, Yongxin; Lu, Di; Yang, Dongling; Feng, Jing; Song, Lina; Liang, Minmin; Yan, XiyunNature Nanotechnology (2012), 7 (7), 459-464CODEN: NNAABX; ISSN:1748-3387. (Nature Publishing Group)Engineered nanoparticles have been used to provide diagnostic, therapeutic and prognostic information about the status of disease. Nanoparticles developed for these purposes are typically modified with targeting ligands (such as antibodies, peptides or small mols.) or contrast agents using complicated processes and expensive reagents. Moreover, this approach can lead to an excess of ligands on the nanoparticle surface, and this causes non-specific binding and aggregation of nanoparticles, which decreases detection sensitivity. Here, we show that magnetoferritin nanoparticles (M-HFn) can be used to target and visualize tumor tissues without the use of any targeting ligands or contrast agents. Iron oxide nanoparticles are encapsulated inside a recombinant human heavy-chain ferritin (HFn) protein shell, which binds to tumor cells that overexpress transferrin receptor 1 (TfR1). The iron oxide core catalyzes the oxidn. of peroxidase substrates in the presence of hydrogen peroxide to produce a color reaction that is used to visualize tumor tissues. We examd. 474 clin. specimens from patients with nine types of cancer and verified that these nanoparticles can distinguish cancerous cells from normal cells with a sensitivity of 98% and specificity of 95%.
- 35Liu D. Ju C. Han C. Shi R. Chen X. Duan D. Yan J. Yan X. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen Biosens. Bioelectron. 2021 173 112817Google Scholar35Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigenLiu, Dan; Ju, Chenhui; Han, Chao; Shi, Rui; Chen, Xuehui; Duan, Demin; Yan, Jinghua; Yan, XiyunBiosensors & Bioelectronics (2021), 173 (), 112817CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)COVID-19 has evolved into a global pandemic. Early and rapid detection is crucial to control of the SARS-CoV-2 transmission. While representing the gold std. for early diagnosis, nucleic acid tests for SARS-CoV-2 are often complicated and time-consuming. Serol. rapid antibody tests are characterized by high rates of false-neg. diagnoses, esp. during early infection. Here, we developed a novel nanozyme-based chemiluminescence paper assay for rapid and sensitive detection of SARS-CoV-2 spike antigen, which integrates nanozyme and enzymic chemiluminescence immunoassay with the lateral flow strip. The core of our paper test is a robust Co-Fe@hemin-peroxidase nanozyme that catalyzes chemiluminescence comparable with natural peroxidase HRP and thus amplifies immune reaction signal. The detection limit for recombinant spike antigen of SARS-CoV-2 was 0.1 ng/mL, with a linear range of 0.2-100 ng/mL. Moreover, the sensitivity of test for pseudovirus could reach 360 TCID50/mL, which was comparable with ELISA method. The strip recognized SARS-CoV-2 antigen specifically, and there was no cross reaction with other coronaviruses or influenza A subtypes. This testing can be completed within 16 min, much shorter compared to the usual 1-2 h required for currently used nucleic acid tests. Furthermore, signal detection is feasible using the camera of a std. smartphone. Ingredients for nanozyme synthesis are simple and readily available, considerably lowering the overall cost. In conclusion, our paper test provides a high-sensitive point-of-care testing (POCT) approach for SARS-CoV-2 antigen detection, which should greatly facilitate early screening of SARS-CoV-2 infections, and considerably lower the financial burden on national healthcare resources.
- 36Mansur A. A. Mansur H. S. Carvalho S. M. Engineered hybrid nanozyme catalyst cascade based on polysaccharide-enzyme-magnetic iron oxide nanostructures for potential application in cancer therapy Catal. Today 2020 187 198Google ScholarThere is no corresponding record for this reference.
- 37Vallabani N. S. Vinu A. Singh S. Karakoti A. Tuning the ATP-triggered pro-oxidant activity of iron oxide-based nanozyme towards an efficient antibacterial strategy J. Colloid Interface Sci. 2020 567 154 164Google Scholar37Tuning the ATP-triggered pro-oxidant activity of iron oxide-based nanozyme towards an efficient antibacterial strategyVallabani, N. V. Srikanth; Vinu, Ajayan; Singh, Sanjay; Karakoti, AjayJournal of Colloid and Interface Science (2020), 567 (), 154-164CODEN: JCISA5; ISSN:0021-9797. (Elsevier B.V.)An alarming increase in bacterial resistance towards various types of antibiotics makes it imperative to design alternate or combinational therapies to treat stubborn bacterial infections. In this perspective, emerging tools like nanozymes, nanomaterials with biol. enzyme like characteristics, are being utilized to control infections caused by bacterial pathogens. Among several nanozymes used for antibacterial applications, Fe3O4 nanoparticles (NP) received great attention due to their effective peroxidase like activity. The pH dependent peroxidase activity of Fe3O4 NP results in generation of ·OH radical via the unique Fenton chem. of iron. However, their pH dependent activity is restricted to acidic environment and dramatic loss in antibacterial activity is obsd. at near neutral pH. Here we describe a novel strategy to overcome the pH lacunas of citrate coated Fe3O4 NP by utilizing ATP disodium salt (ATP) as a synergistic agent to accelerate the ·OH radical prodn. and restore its antibacterial activity over a wide range of pH. This synergistic combination (30μg/mL Fe3O4 NP and 2.5 mM ATP) shows a high bactericidal activity against both gram pos. (B. subtilis) and gram neg. (E. coli) bacterial strains, in presence of H2O2, at neutral pH. The synergistic effect (Fe3O4 NP + ATP) is detd. from the viability assessment and membrane damage studies and is further confirmed by comparing the concn. of generated ·OH radicals. Over all, this study illustrates ATP assisted and ·OH-mediated bactericidal activity of Fe3O4 nanozyme at near neutral pH.
- 38Shi S. Wu S. Shen Y. Zhang S. Xiao Y. He X. Gong J. Farnell Y. Tang Y. Huang Y. Iron oxide nanozyme suppresses intracellular Salmonella Enteritidis growth and alleviates infection in vivo Theranostics 2018 8 22 6149Google Scholar38Iron oxide nanozyme suppresses intracellular Salmonella Enteritidis growth and alleviates infection in vivoShi, Shourong; Wu, Shu; Shen, Yiru; Zhang, Shan; Xiao, Yunqi; He, Xi; Gong, Jiansen; Farnell, Yuhua; Tang, Yan; Huang, Yixin; Gao, LizengTheranostics (2018), 8 (22), 6149-6162CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Salmonella Enteritidis S.Enteritidis is a globally significant zoonotic foodborne pathogen which has led to large nos. of deaths in humans and caused economic losses in animal husbandry. S.Enteritidis invades host cells and survives within the cells, causing resistance to antibiotic treatment. Effective methods of elimination and eradication of intracellular S.Enteritidis are still very limited. Here we evaluated whether a new intracellular antibacterial strategy using iron oxide nanozymes (IONzymes) exerted highly antibacterial efficacy via its intrinsic peroxidase-like activity in vitro and in vivo. The antibacterial activities of IONzymes against planktonic S.Enteritidis, intracellular S.Enteritidis in Leghorn Male Hepatoma-derived cells (LMH), and liver from specific pathogen free (SPF) chicks were investigated by spread-plate colony count method and cell viability assay. Changes in levels of microtubule-assocd. protein light chain 3 (LC3), a widely used marker for autophagosomes, were analyzed by immunoblotting, immunofluorescence, and electron microscopy. Reactive oxygen species (ROS) prodn. was also assessed in vitro. High-throughput RNA sequencing was used to investigate the effects of IONzymes on liver transcriptome of S.Enteritidis-infected chicks. We demonstrated that IONzymes had high biocompatibility with cultured LMH cells and chickens, which significantly inhibited intracellular S.Enteritidis survival in vitro and in vivo. In addn., co-localization of IONzymes with S.Enteritidis were obsd. in autophagic vacuoles of LMH cells and liver of chickens infected by S.Enteritidis, indicating that IONzymes mediated antibacterial reaction of S.Enteritidis with autophagic pathway. We found ROS level was significantly increased in infected LMH cells treated with IONzymes, which might enhance the autophagic elimination of intracellular S.Enteritidis. Moreover, orally administered IONzymes decreased S.Enteritidis organ invasion of the liver and prevented pathol. lesions in a chicken-infection model. Non-target transcriptomic profiling also discovered IONzymes could change hepatic oxidn.-redn. and autophagy related gene expressions in the S.Enteritidis infected chickens. These data suggest that IONzymes can increase ROS levels to promote the antibacterial effects of acid autophagic vacuoles, and thus suppress the establishment and survival of invading intracellular S.Enteritidis. As a result, IONzymes may be a novel alternative to current antibiotics for the control of intractable S.Enteritidis infections.
- 39Guo S. Guo L. Unraveling the Multi-Enzyme-Like Activities of Iron Oxide Nanozyme via a First-Principles Microkinetic Study J. Phys. Chem. C 2019 123 50 30318 30334Google Scholar39Unraveling the Multi-Enzyme-Like Activities of Iron Oxide Nanozyme via a First-Principles Microkinetic StudyGuo, Sibei; Guo, LingJournal of Physical Chemistry C (2019), 123 (50), 30318-30334CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)The design and construction of efficient artificial enzyme-mimicking nanomaterials, nanozymes, is highly desirable because of their high stability and low cost. Recent studies have demonstrated iron oxide nanomaterials as multifunctional nanozymes. However, the catalytic mechanism remains unclear. Herein we have combined d. functional theory calcns. with microkinetic modeling to demonstrate (Fe3O4)n (n = 1 to 2) exhibiting the intrinsic activity of mimicking enzymes of catalases (CATs), superoxide dismutases (SODs), and peroxidases (PODs). Their catalytic activities are facilitated by the close proximity of undercoordinated, tunable Fe/O pairs on the (Fe3O4)n surfaces. The (Fe3O4)n (n = 1 to 2) with different morphologies and sizes exhibited different catalytic activities on the order of Fe3O4 > (Fe3O4)2. Three possible reaction mechanisms of CAT-like activity (i.e., base-like dissociative mechanism, acid-like dissociative mechanism, and bihydrogen peroxide associative mechanism) and two possible reaction mechanisms of SOD-like activity (i.e., Langmuir-Hinshelwood mechanism and Eley-Rideal mechanism) are systematically explored based on min. energy path calcns. It is identified that the acid-like dissociative mechanism and the Langmuir-Hinshelwood mechanism are the energetically most favorable pathways, which is proved by the anal. of the rate-detg. step, the energetic span model, and the rate const. The degree of turnover frequency control (XTOF) of the species in the mechanism is calcd. and identifies the rate-controlling intermediates and transition states (i.e., those with the highest XTOF), which are used as descriptors to modify and improve the (Fe3O4)n catalysts. This study should not only aid our understanding of Fe3O4 artificial enzymes from at. level but also facilitate the design and construction of other types of target-specific artificial enzymes.
- 40Shen X. Wang Z. Gao X. Zhao Y. Density Functional Theory-Based Method to Predict the Activities of Nanomaterials as Peroxidase Mimics ACS Catal. 2020 10 21 12657 12665Google Scholar40Density Functional Theory-Based Method to Predict the Activities of Nanomaterials as Peroxidase MimicsShen, Xiaomei; Wang, Zhenzhen; Gao, Xingfa; Zhao, YuliangACS Catalysis (2020), 10 (21), 12657-12665CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)A wide variety of nanomaterials possess peroxidase-like catalytic activities and show promise as cost-effective and versatile replacements for natural peroxidases. However, a universal tool for predicting the activities of these materials is still lacking, thus hindering the efficient discovery of nanomaterials as peroxidase mimics. Here, we use d. functional theory calcns. to reveal the peroxidase-mimetic mechanisms for a series of iron-oxide nanosurfaces, and we derive a volcano-shaped plot of catalytic activity as a function of simple energy-based descriptors. The activity curves and the descriptors can be used to predict peroxidase-like activities for not only iron oxides but also other nanomaterials that share similar catalytic mechanisms. The results demonstrate that the method developed herein can systematically predict the peroxidase-like activities of nanomaterials and thus is expected to be of use for computer-aided design of nanomaterial-based peroxidase mimics.
- 41Chance B. Oshino N. Kinetics and mechanisms of catalase in peroxisomes of the mitochondrial fraction Biochem. J. 1971 122 2 225 233Google Scholar41Kinetics and mechanisma of catalase in peroxisomes of the mitochondrial fractionChance, Britton; Oshino, N.Biochemical Journal (1971), 122 (2), 225-33CODEN: BIJOAK; ISSN:0264-6021.The primary intermediate of catalase and H2O2 was identified and investigated in peroxisome-rich mitochondrial fractions of rat liver. On the basis of kinetic consts. detd. in vitro, it is possible to calc. with reasonable precision the mol. statistics of catalase action in the peroxisomes. The endogenous H2O2 generation is adequate to sustain a concn. of the catalase intermediate of 60-70% of the H2O2 satn. value. Total amt. of catalase corresponds to 0.12-0.15 nmole of heme Fe/mg protein. In State 1 the rate of H2O2 generation corresponds to 0.9 nmole/min/mg protein or 5% of the mitochondrial respiratory rate in State 4. Partial satn. of the catalase intermediate with H2O2 in the mitochondrial fraction suggests its significant peroxidatic activity towards its endogenous H donor. A variation of this value from 0.3 in State 4 to 0 under anaerobic conditions is obsd. For a particular prepn. the H2O2 generation rate in the substrate-supplemented State 4 corresponds to 0.17/sec, the H2O2 concn. to 2.5mM and the H-donor concn. (in terms of EtOH) to 0.12mM. The reaction is 70% peroxidatic and 30% catalatic. A coordinated production of both oxidizing and reducing substrates for catalase in the mitochondrial fraction is suggested by a 2.2-fold increase of H2O2 generation and a 3-fold increase in H-donor generation in the State 1 to State 4 transition.
- 42Hwang I. Uddin M. J. Pak E. S. Kang H. Jin E.-J. Jo S. Kang D. Lee H. Ha H. The impaired redox balance in peroxisomes of catalase knockout mice accelerates nonalcoholic fatty liver disease through endoplasmic reticulum stress Free Radical Biol. Med. 2020 148 22 32Google Scholar42The impaired redox balance in peroxisomes of catalase knockout mice accelerates nonalcoholic fatty liver disease through endoplasmic reticulum stressHwang, Inah; Uddin, Md. Jamal; Pak, Eun Seon; Kang, Hyeji; Jin, Eun-Jung; Jo, Suin; Kang, Dongmin; Lee, Hyukjin; Ha, HunjooFree Radical Biology & Medicine (2020), 148 (), 22-32CODEN: FRBMEH; ISSN:0891-5849. (Elsevier B.V.)Peroxisomes are essential organelles for maintaining the homeostasis of lipids and reactive oxygen species (ROS). While oxidative stress-induced endoplasmic reticulum (ER) stress plays an important role in nonalcoholic fatty liver disease (NAFLD), the role of peroxisomes in ROS-mediated ER stress in the development of NAFLD remains elusive. We investigated whether an impaired peroxisomal redox state accelerates NAFLD by activating ER stress by inhibiting catalase, an antioxidant expressed exclusively in peroxisomes. Wild-type (WT) and catalase knockout (CKO) mice were fed either a normal diet or a high-fat diet (HFD) for 11 wk. HFD-induced phenotype changes and liver injury accompanied by ER stress and peroxisomal dysfunction were accelerated in CKO mice compared to WT mice. Interestingly, these changes were also significantly increased in CKO mice fed a normal diet. Inhibition of catalase by 3-aminotriazole in hepatocytes resulted in the following effects: (i) increased peroxisomal H2O2 levels as measured by a peroxisome-targeted H2O2 probe (HyPer-P); (ii) elevated intracellular ROS; (iii) decreased peroxisomal biogenesis; (iv) activated ER stress; (v) induced lipogenic genes and neutral lipid accumulation; and (vi) suppressed insulin signaling cascade assocd. with JNK activation. N-acetylcysteine or 4-phenylbutyric acid effectively prevented those alterations. These results suggest that a redox imbalance in peroxisomes perturbs cellular metab. through the activation of ER stress in the liver.
- 43Ighodaro O. Akinloye O. First line defence antioxidants-superoxide dismutase (SOD) catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid Alexandria J.Med. 2018 54 4 287 293Google ScholarThere is no corresponding record for this reference.
- 44Fita I. Rossmann M. G. The active center of catalase J. Mol. Biol. 1985 185 1 21 37Google Scholar44The active center of catalaseFita, Ignacio; Rossmann, Michael G.Journal of Molecular Biology (1985), 185 (1), 21-37CODEN: JMOBAK; ISSN:0022-2836.The refined structure of beef liver catalase is examd. with regard to possible catalytic mechanisms. The distal side of the deeply buried heme pocket is connected with the surface of the mol. by 1 (or possibly 2) channel(s). The electron d. representing the heme group, in each of the 2 crystallog. independent subunits, is consistent with degrdn. of the porphyrin rings. The heme group appears to be buckled, reflecting the high content of bile pigment in liver catalase. The spatial organization on the proximal side (where the 5th ligand of the Fe is located) shows an elaborate network of interactions; the distal side contains the substrate pocket. The limited space in this region severely constrains possible substrate positions and orientations. The Nδ atom of essential histidine-74 H bonds with Oγ of serine-13, which in turn H bonds to an H2O mol. assocd. with the propionic carbonylic group of pyrrole III. These interactions are also visible in the refined structure of Penicillium vitale catalase. Model building suggests a pathway for a catalase mechanism (compd. I formation, as well as catalase and peroxidase reactions). There are some similarities in compd. I formation of catalase and cytochrome c peroxidase.
- 45Obinger C. Maj M. Nicholls P. Loewen P. Activity, Peroxide Compound Formation, and Heme d Synthesis in Escherichia coli HPII Catalase Arch. Biochem. Biophys. 1997 342 1 58 67Google Scholar45Activity, peroxide compound formation, and heme d synthesis in Escherichia coli HPII catalaseObinger, C.; Maj, M.; Nicholls, P.; Loewen, P.Archives of Biochemistry and Biophysics (1997), 342 (1), 58-67CODEN: ABBIA4; ISSN:0003-9861. (Academic)Wild-type Escherichia coli HPII catalase (heme d contg.) has 15% the activity of beef liver enzyme per heme. The rate const. for compd. I formation with H2O2 is 1.3×106 M-1 s-1. HPII compd. I reacts with H2O2 to form O2 with a rate const. of 1.8×106 M-1 s-1. Forty percent of HPII hemes are in the compd. I state during turnover. Compd. I is reduced by ethanol and formate at rates of 5 and 13 M-1 s-1 (pH 7.0), resp. Incubation of HPII compd. I with ferrocyanide and ascorbate does not form a compd. II species. Mutation of His128 to alanine or asparagine gives inactive protoheme proteins. Mutation of Asn-201 gives partially active heme d forms. Asn201Ala has 24%, Asn201Asp 10%, and Asn201Gln 0.4% of wild-type activity. Asn201His contains protoheme when isolated and converts this via protoheme compd. I to a heme d species. Both distal heme cavity residues His128 and Asn201 are implicated in catalytic activity, compd. I formation, and in situ heme d biosynthesis. HPII Asn201, like the corresponding residue in protoheme catalases, may promote H+ transfer to His128 imidazole, facilitating (i) peroxide anion binding to heme and (ii) stabilization of a transition state for heterolytic cleavage of the O-O bond.
- 46Zhang R. Chen L. Liang Q. Xi J. Zhao H. Jin Y. Gao X. Yan X. Gao L. Fan K. Unveiling the active sites on ferrihydrite with apparent catalase-like activity for potentiating radiotherapy Nano Today 2021 41 101317Google Scholar46Unveiling the active sites on ferrihydrite with apparent catalase-like activity for potentiating radiotherapyZhang, Ruofei; Chen, Lei; Liang, Qian; Xi, Juqun; Zhao, Hanqing; Jin, Yiliang; Gao, Xingfa; Yan, Xiyun; Gao, Lizeng; Fan, KelongNano Today (2021), 41 (), 101317CODEN: NTAOCG; ISSN:1748-0132. (Elsevier Ltd.)The use of catalase-like nanozymes in relieving tumor hypoxia has been a promising strategy for adjuvant radiotherapy, yet their catalytic mechanism and toxic potential remain elusive. While most studies on catalase-like mechanisms focus on the substrates and products in the catalytic process, little is reported to analyze the dominating surface structure for catalytic activity of nanozymes. Here, we reported the catalase-like activity of ferrihydrite and revealed its structure-activity relationship. Among the ten main forms of iron oxide nanomaterials, ferrihydrite, esp. 2-line ferrihydrite, exhibited the highest catalase-like activity. Importantly, the structure-activity fitting and d. functional anal. revealed that the abundant surface iron-assocd. hydroxyl groups dominantly affect the catalase-like activity of ferrihydrites. In addn., the ferrihydrite exhibited no peroxidase-like and superoxide dismutase-like activities, and constantly catalyzed the decompn. of H2O2 to release O2 in the pH range of 4.0-8.7. With the help of steady and single catalase-like activity in the physiol. environment, ferrihydrite safely and effectively catalyzed the H2O2 in tumor microenvironment to in-situ produce O2 and significantly enhanced the therapeutic effect of radiotherapy. Moreover, ferrihydrite exhibited excellent biosafety and biodegradability. These findings demonstrate the great potential of ferrihydrite as a biocompatible catalase-like nanozyme for relieving hypoxia in cancer therapy.
- 47Srinivasan B. A guide to the Michaelis–Menten equation: steady state and beyond FEBS J. 2021 1 13Google ScholarThere is no corresponding record for this reference.
- 48Fan H. M. Yi J. B. Yang Y. Kho K. W. Tan H. R. Shen Z. X. Ding J. Sun X. W. Olivo M. C. Feng Y. P. Single-Crystalline MFe2O4 Nanotubes/Nanorings Synthesized by Thermal Transformation Process for Biological Applications ACS Nano 2009 3 9 2798 2808Google Scholar48Single-Crystalline MFe2O4 Nanotubes/Nanorings Synthesized by Thermal Transformation Process for Biological ApplicationsFan, Hai-Ming; Yi, Jia-Bao; Yang, Yi; Kho, Kiang-Wei; Tan, Hui-Ru; Shen, Ze-Xiang; Ding, Jun; Sun, Xiao-Wei; Olivo, Malini Carolene; Feng, Yuan-PingACS Nano (2009), 3 (9), 2798-2808CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)The authors report a general thermal transformation approach to synthesize single-cryst. magnetic transition metal oxides nanotubes/nanorings including magnetite Fe3O4, maghemite γ-Fe2O3, and ferrites MFe2O4 (M = Co, Mn, Ni, Cu) using hematite α-Fe2O3 nanotubes/nanorings template. While the straightforward redn. or redn.-oxides process was employed to produce Fe3O4 and γ-Fe2O3, the α-Fe2O3/M(OH)2 core/shell nanostructure was used as precursor to prep. MFe2O4 nanotubes via MFe2O4-x (0 < x < 1) intermediate. The transformed ferrites nanocrystals retain the hollow structure and single-cryst. nature of the original templates. However, the crystallog. orientation-relationships of cubic spinel ferrites and trigonal hematite show strong correlation with their morphologies. The hollow-structured MFe2O4 nanocrystals with tunable size, shape, and compn. have exhibited unique magnetic properties. Moreover, they have been demonstrated as a highly effective peroxidase mimic catalysts for lab. immunoassays or as a universal nanocapsules hybridized with luminescent QDs for magnetic sepn. and optical probe of lung cancer cells, suggesting that these biocompatible magnetic nanotubes/nanorings have great potential in biomedicine and biomagnetic applications.
- 49Hao J. H. Zhang Z. Yang W. S. Lu B. P. Ke X. Zhang B. L. Tang J. L. In situ controllable growth of CoFe2O4 ferrite nanocubes on graphene for colorimetric detection of hydrogen peroxide J. Mater. Chem. A 2013 1 13 4352 4357Google Scholar49In situ controllable growth of CoFe2O4 ferrite nanocubes on graphene for colorimetric detection of hydrogen peroxideHao, Jinhui; Zhang, Zhe; Yang, Wenshu; Lu, Baoping; Ke, Xi; Zhang, Bailin; Tang, JilinJournal of Materials Chemistry A: Materials for Energy and Sustainability (2013), 1 (13), 4352-4357CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)A facile method is proposed for the synthesis of cubic CoFe2O4 ferrite-reduced graphene oxide nanocomposite sheets (rGO-CFs), using poly(N-vinyl-2-pyrrolidone) as the reductant and stabilizer. The rGO-CFs functioned as efficient peroxidase mimetics and were successfully applied for colorimetric assay. The morphol. and compn. of the rGO-CFs were characterized by TEM, XPS, and x-ray diffraction. Significantly, the peroxidase-like activity of the rGO-CFs followed typical Michaelis-Menten kinetics and showed a good affinity to 3,3',5,5'-tetra-Me benzidine. Under optimum conditions, the colorimetric assay showed a lower detection limit (S/N = 3) of 0.3 μM when compared with that of other nanoparticle based colorimetric assays. Furthermore, the cubic nanostructured rGO-CFs exhibited better stability than horseradish peroxidase when they were exposed to solns. with different solvents and temps. These excellent properties made the cubic nanostructured rGO-CFs an ideal candidate for a wide range of potential applications as peroxidase mimetics.
- 50Zhang Z. J. Zhang X. H. Liu B. W. Liu J. W. Molecular Imprinting on Inorganic Nanozymes for Hundred-fold Enzyme Specificity J. Am. Chem. Soc. 2017 139 15 5412 5419Google Scholar50Molecular Imprinting on Inorganic Nanozymes for Hundred-fold Enzyme SpecificityZhang, Zijie; Zhang, Xiaohan; Liu, Biwu; Liu, JuewenJournal of the American Chemical Society (2017), 139 (15), 5412-5419CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)Enzyme-mimicking nanomaterials (nanozymes) are more cost-effective and robust than protein enzymes, but they lack specificity. Herein, molecularly imprinted polymers (MIPs) were grown on Fe3O4 nanozymes with peroxidase-like activity to create substrate binding pockets. Electron microscopy confirmed a shell of nanogel. By imprinting with an adsorbed substrate, moderate specificity was achieved with neutral monomers. Further introducing charged monomers led to nearly 100-fold specificity for the imprinted substrate over the non-imprinted compared to that of bare Fe3O4. Selective substrate binding was further confirmed by isothermal titrn. calorimetry. The same method was also successfully applied for imprinting on gold nanoparticles (peroxidase mimics) and nanoceria (oxidase mimics). Mol. imprinting furthers the functional enzyme mimicking aspect of nanozymes, and such hybrid materials will find applications in biosensor development, sepn., environmental remediation, and drug delivery.
- 51Huo M. F. Wang L. Y. Chen Y. Shi J. L. Tumor-selective catalytic nanomedicine by nanocatalyst delivery Nat. Commun. 2017 8 357Google Scholar51Tumor-selective catalytic nanomedicine by nanocatalyst deliveryHuo Minfeng; Wang Liying; Chen Yu; Shi Jianlin; Huo Minfeng; Wang Liying; Wang LiyingNature communications (2017), 8 (1), 357 ISSN:.Tumor cells metabolize in distinct pathways compared with most normal tissue cells. The resulting tumor microenvironment would provide characteristic physiochemical conditions for selective tumor modalities. Here we introduce a concept of sequential catalytic nanomedicine for efficient tumor therapy by designing and delivering biocompatible nanocatalysts into tumor sites. Natural glucose oxidase (GOD, enzyme catalyst) and ultrasmall Fe3O4 nanoparticles (inorganic nanozyme, Fenton reaction catalyst) have been integrated into the large pore-sized and biodegradable dendritic silica nanoparticles to fabricate the sequential nanocatalyst. GOD in sequential nanocatalyst could effectively deplete glucose in tumor cells, and meanwhile produce a considerable amount of H2O2 for subsequent Fenton-like reaction catalyzed by Fe3O4 nanoparticles in response to mild acidic tumor microenvironment. Highly toxic hydroxyl radicals are generated through these sequential catalytic reactions to trigger the apoptosis and death of tumor cells. The current work manifests a proof of concept of catalytic nanomedicine by approaching selectivity and efficiency concurrently for tumor therapeutics.The specific metabolism of cancer cells may allow for selective tumor therapeutics. Here, the authors show that a suitable combination of an enzyme and iron nanoparticles loaded on dendritic silica induces apoptosis of cancer cells in response to the glucose-reliant and mild acidic microenvironment.
- 52Wei F. Cui X. Y. Wang Z. Dong C. C. Li J. D. Han X. J. Recoverable peroxidase-like Fe3O4@MoS2-Ag nanozyme with enhanced antibacterial ability Chem. Eng. J. 2021 408 127240Google Scholar52Recoverable peroxidase-like Fe3O4@MoS2-Ag nanozyme with enhanced antibacterial abilityWei, Feng; Cui, Xinyu; Wang, Zhao; Dong, Changchang; Li, Jiadong; Han, XiaojunChemical Engineering Journal (Amsterdam, Netherlands) (2021), 408 (), 127240CODEN: CMEJAJ; ISSN:1385-8947. (Elsevier B.V.)Antibacterial agents with enzyme-like properties and bacteria-binding ability have provided an alternative method to efficiently disinfect drug-resistance microorganism. Herein, a Fe3O4@MoS2-Ag nanozyme with defect-rich rough surface was constructed by a simple hydrothermal method and in-situ photodeposition of Ag nanoparticles. The nanozyme exhibited good antibacterial performance against E. coli (∼69.4%) by the generated ROS and released Ag+, while the nanozyme could further achieve an excellent synergistic disinfection (∼100%) by combining with the near-IR photothermal property of Fe3O4@MoS2-Ag. The antibacterial mechanism study showed that the antibacterial process was detd. by the collaborative work of peroxidase-like activity, photothermal effect and leakage of Ag+. The defect-rich rough surface of MoS2 layers facilitated the capture of bacteria, which enhanced the accurate and rapid attack of ·OH and Ag+ to the membrane of E. coli with the assistance of local hyperthermia. This method showed broad-spectrum antibacterial performance against Gram-neg. bacteria, Gram-pos. bacteria, drug-resistant bacteria and fungal bacteria. Meanwhile, the magnetism of Fe3O4 was used to recycle the nanozyme. This work showed great potential of engineered nanozymes for efficient disinfection treatment.
- 53Peng F. F. Zhang Y. Gu N. Size-dependent peroxidase-like catalytic activity of Fe3O4 nanoparticles Chin. Chem. Lett. 2008 19 6 730 733Google Scholar53Size-dependent peroxidase-like catalytic activity of Fe3O4 nanoparticlesPeng, Fang Fang; Zhang, Yu; Gu, NingChinese Chemical Letters (2008), 19 (6), 730-733CODEN: CCLEE7; ISSN:1001-8417. (Elsevier B.V.)Peroxidase-like catalytic properties of Fe3O4 nanoparticles (NPs) with three different sizes, synthesized by chem. copptn. and sol-gel methods, were investigated by UV-vis spectrum anal. By comparing Fe3O4 NPs with av. diams. of 11, 20, and 150 nm, we found that the catalytic activity increases with the reduced nanoparticle size. The electrochem. method to characterize the catalytic activity of Fe3O4 NPs using the response currents of the reaction product and substrate was also developed.
- 54Zuo X. L. Peng C. Huang Q. Song S. P. Wang L. H. Li D. Fan C. H. Design of a Carbon Nanotube/Magnetic Nanoparticle-Based Peroxidase-Like Nanocomplex and Its Application for Highly Efficient Catalytic Oxidation of Phenols Nano Res. 2009 2 8 617 623Google Scholar54Design of a carbon nanotube/magnetic nanoparticle-based peroxidase-like nanocomplex and its application for highly efficient catalytic oxidation of phenolsZuo, Xiaolei; Peng, Cheng; Huang, Qing; Song, Shiping; Wang, Lihua; Li, Di; Fan, ChunhaiNano Research (2009), 2 (8), 617-623CODEN: NRAEB5; ISSN:1998-0124. (Springer)We report a novel nanotechnol.-based approach for the highly efficient catalytic oxidn. of phenols and their removal from wastewater. We use a nanocomplex made of multi-walled carbon nanotubes (MWNTs) and magnetic nanoparticles (MNPs). This nanocomplex retains the magnetic properties of individual MNPs and can be effectively sepd. under an external magnetic field. More importantly, the formation of the nanocomplex enhances the intrinsic peroxidase-like activity of the MNPs that can catalyze the redn. of hydrogen peroxide (H2O2). Significantly, in the presence of H2O2, this nanocomplex catalyzes the oxidn. of phenols with high efficiency, generating insol. polyarom. products that can be readily sepd. from water.
- 55Shi W. B. Zhang X. D. He S. H. Huang Y. M. CoFe2O4 magnetic nanoparticles as a peroxidase mimic mediated chemiluminescence for hydrogen peroxide and glucose Chem. Commun. 2011 47 38 10785 10787Google Scholar55CoFe2O4 magnetic nanoparticles as a peroxidase mimic mediated chemiluminescence for hydrogen peroxide and glucoseShi, Wenbing; Zhang, Xiaodan; He, Shaohui; Huang, YumingChemical Communications (Cambridge, United Kingdom) (2011), 47 (38), 10785-10787CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)This communication presents a new peroxidase mimic of CoFe2O4 nanoparticles evaluated by the luminol-based chemiluminescent (CL) reaction. This offers a new method for evaluation and screening of the nanoparticles-based enzyme mimetics.
- 56Chaudhari K. N. Chaudhari N. K. Yu J.-S. Peroxidase mimic activity of hematite iron oxides (α-Fe2O3) with different nanostructures Catal. Sci. Technol. 2012 2 1 119 124Google Scholar56Peroxidase mimic activity of hematite iron oxides (α-Fe2O3) with different nanostructuresChaudhari, Kiran N.; Chaudhari, Nitin K.; Yu, Jong-SungCatalysis Science & Technology (2012), 2 (1), 119-124CODEN: CSTAGD; ISSN:2044-4753. (Royal Society of Chemistry)Enzyme mimics have garnered considerable attention as they can overcome some serious disadvantages assocd. with the natural enzymes. In recently developed sphere and rod shaped iron oxide peroxidase mimic nanoparticles, the influence of phys. parameters such as shape, size and surface area on the catalytic performance was not clearly demonstrated. In order to better understand the influence of phys. parameters on the enzyme mimic activity of iron oxide nanoparticles, the present study was initiated using three different shaped hematite α-Fe2O3 nanostructures, particularly hexagonal prism, cube-like and rods as model systems. A comparative account of kinetic parameters (Km, Vmax and Kcat) of the peroxidase mimic activity by the various α-Fe2O3 nanostructures indicated that the enzymic potential of these nanoparticles increased from hexagonal prism to rods, via cube-like, suggesting that one-dimensional particles act as a more efficient enzyme mimic system compared to their multi-dimensional counterparts. Surface area is likely to be a key phys. aspect responsible for the enzyme mimic activity. Interestingly, however, particles with lower surface area showed better catalytic performance in the case of one-dimensional rod structure. Upon further anal. of the one-dimensional rods, addnl. phys. properties such as porosity and pore shape also seem to have a significant contribution to their catalytic activity.
- 57Ma M. Xie J. Zhang Y. Chen Z. P. Gu N. Fe3O4@Pt nanoparticles with enhanced peroxidase-like catalytic activity Mater. Lett. 2013 105 36 39Google Scholar57Fe3O4@Pt nanoparticles with enhanced peroxidase-like catalytic activityMa, Ming; Xie, Jun; Zhang, Yu; Chen, Zhongping; Gu, NingMaterials Letters (2013), 105 (), 36-39CODEN: MLETDJ; ISSN:0167-577X. (Elsevier B.V.)Pt modified Fe3O4 magnetic nanoparticles (Fe3O4@Pt NPs) were synthesized by a simple method. The morphol. and crystal structure of the as-prepd. nanocomposite were characterized by TEM, XRD and EDS. The peroxidase-like catalytic activity of the synthesized nanoparticles was also investigated. Catalysis was found to follow Michaelis-Menten kinetics. The calcd. kinetic parameters of the Fe3O4@Pt NPs show that they exhibited strong affinity with substrates and enhanced catalytic activity than that of Fe3O4 nanoparticles, suggesting a higher peroxidase-like activity.
- 58Sun H. Y. Jiao X. L. Han Y. Y. Jiang Z. Chen D. R. Synthesis of Fe3O4-Au Nanocomposites with Enhanced Peroxidase-Like Activity Eur. J. Inorg. Chem. 2013 1 109 114Google Scholar58Synthesis of Fe3O4-Au Nanocomposites with Enhanced Peroxidase-Like ActivitySun, Haiyan; Jiao, Xiuling; Han, Yanyang; Jiang, Zhen; Chen, DairongEuropean Journal of Inorganic Chemistry (2013), 2013 (1), 109-114CODEN: EJICFO; ISSN:1434-1948. (Wiley-VCH Verlag GmbH & Co. KGaA)Sphere-like Fe3O4 aggregates were solvothermally prepd. with ethylene glycol, NaOAc and FeCl3·6H2O as raw materials. The sphere-like Fe3O4 aggregates provided heterogeneous growth sites for Au nanoparticles. These were obtained by redn. of HAuCl4 by Na citrate under mild reaction conditions and the Fe3O4-Au nanocomposites were subsequently formed. The peroxidase-like activity of nanocomposites was studied with H2O2 and 3,3',5,5'-tetramethylbenzidine as substrates. Fe3O4-Au nanocomposites exhibited better catalytic activity than pure Fe3O4 aggregates, mainly resulting from the special electronic structure at the interfaces between the sphere-like Fe3O4 aggregates and the Au nanoparticles.
- 59Song L. N. Huang C. Zhang W. Ma M. Chen Z. W. Gu N. Zhang Y. Graphene oxide-based Fe2O3 hybrid enzyme mimetic with enhanced peroxidase and catalase-like activities Colloids Surf., A 2016 506 747 755Google Scholar59Graphene oxide-based Fe2O3 hybrid enzyme mimetic with enhanced peroxidase and catalase-like activitiesSong, Lina; Huang, Chao; Zhang, Wei; Ma, Ming; Chen, Zhongwen; Gu, Ning; Zhang, YuColloids and Surfaces, A: Physicochemical and Engineering Aspects (2016), 506 (), 747-755CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)Graphene based materials are widely used in energy conversation and catalytic reaction due to their rapid electron transfer capacity and large surface area. Herein, graphene Fe2O3 (GO-Fe2O3) hybrids with enhanced peroxidase-like activity were fabricated. Enhanced peroxidase-like activity strongly depending on pH, temp. and hybrid concn. was obsd. and the peroxidase-like behavior fits well the Michaelis-Menten kinetic model. Free radicals, such as ·OH and O-2·, as intermediates in the hybrid-H2O2 reaction system were directly demonstrated by ESR (ESR) technique and the selective radical inhibition expts. Furthermore, it was found that the addn. of TMB rapidly consumed ·OH and O-2·, and subsequently leaded to the formation of blue TMB radical confirmed by the ESR and absorption spectroscopy. Besides acting as mimetic peroxidase in acidic buffer, the GO-Fe2O3 hybrids also displayed the enhanced catalase-like activity in neutral and alk. buffers compared with individual Fe2O3 nanoparticles and GO sheet, resp. The H2O2 decompn. catalyzed by the hybrids was demonstrated by ESR and the generated O2 was measured using the dissolved oxygen electrode. All the results above demonstrate that the GO-Fe2O3 hybrids are a kind of effective pH-dependent mimetic dual-enzyme.
- 60Li J. Cai J. H. Jia H. M. Zhang L. X. Lei Y. He W. W. Yin J. J. Formation of iron oxide/Pd hybrid nanostructures with enhanced peroxidase-like activity and catalytic reduction of 4-nitrophenol J. Environ. Sci. Health, Part C: Environ. Carcinog. Rev. 2017 35 3 159 172Google Scholar60Formation of iron oxide/Pd hybrid nanostructures with enhanced peroxidase-like activity and catalytic reduction of 4-nitrophenolLi, Jing; Cai, Junhui; Jia, Huimin; Zhang, Lixia; Lei, Yan; He, Weiwei; Yin, Jun-JieJournal of Environmental Science and Health, Part C: Environmental Carcinogenesis & Ecotoxicology Reviews (2017), 35 (3), 159-172CODEN: JESHA2; ISSN:1532-4095. (Taylor & Francis, Inc.)Iron oxide/Pd hybrid nanostructures with controllable Pd loading from 0.05 to 1.0 (calcd. as Pd/Fe molar ratio) have been synthesized by chem. redn. of Pd2+ on iron oxide particles. The combination of iron oxide and Pd exhibits enhanced peroxidase-like activity and catalytic activity toward redn. of 4-nitrophenol. The catalytic enhancements were found to be dependent on the Pd loading amt. as well as the synergistic effect between iron oxide and Pd. These results suggest that iron oxide with unique surface chem. state can be an active supporter and suggest an effective way to design superior hybrid nanostructures for catalytic applications.
- 61Wang Q. Q. Zhang X. P. Huang L. Zhang Z. Q. Dong S. J. One-Pot Synthesis of Fe3O4 Nanoparticle Loaded 3D Porous Graphene Nanocomposites with Enhanced Nanozyme Activity for Glucose Detection ACS Appl. Mater. Interfaces 2017 9 8 7465 7471Google Scholar61One-Pot Synthesis of Fe3O4 Nanoparticle Loaded 3D Porous Graphene Nanocomposites with Enhanced Nanozyme Activity for Glucose DetectionWang, Qingqing; Zhang, Xueping; Huang, Liang; Zhang, Zhiquan; Dong, ShaojunACS Applied Materials & Interfaces (2017), 9 (8), 7465-7471CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)A novel one-pot strategy is proposed to fabricate 3D porous graphene (3D GN) decorated with Fe3O4 nanoparticles (Fe3O4 NPs) by using hemin as the iron source. During the process, graphene oxide was simultaneously reduced and self-assembled to form 3D graphene hydrogel, while Fe3O4 NPs synthesized from hemin distributed uniformly on 3D GN. The prepn. process is simple, facile, economical and green. The obtained freeze-dried product (3D GH-5) exhibits outstanding peroxidase-like activity. Compared to the traditional 2D graphene-based nanocomposites, the introduced 3D porous structure dramatically improved the catalytic activity, as well as the catalysis velocity and its affinity for substrate. The high catalytic activity could be ascribed to the forming Fe3O4 NPs and 3D porous graphene structures. Based on its peroxidase-like activity, 3D GH-5 was used for colorimetric detn. of glucose with a low detection limit of 0.8 μM.
- 62Vallabani N. V. S. Karakoti A. S. Singh S. ATP-mediated intrinsic peroxidase-like activity of Fe3O4-based nanozyme: One step detection of blood glucose at physiological pH Colloids Surf., B 2017 153 52 60Google Scholar62ATP-mediated intrinsic peroxidase-like activity of Fe3O4-based nanozyme: One step detection of blood glucose at physiological pHVallabani, N. V. Srikanth; Karakoti, Ajay S.; Singh, SanjayColloids and Surfaces, B: Biointerfaces (2017), 153 (), 52-60CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)Fe3O4 nanoparticles (Fe3O4 NPs), demonstrating peroxidase-like activity has garnered attention in the detection of several biomols., therefore, emerged as an excellent nano-biosensing agent. The intrinsic peroxidase-like activity of Fe3O4 NPs at acidic pH is the fundamental action driving the oxidn. of substrates like TMB, resulting in a colorimetric product formation used in the detection of biomols. Hence, the detection sensitivity essentially depends on the ability of oxidn. by Fe3O4 NPs in presence of H2O2. However, the limited sensitivity and pH condition constraint have been identified as the major drawbacks in the detection of biomols. at physiol. pH. Herein, we report overwhelming of the fundamental limitation of acidic pH and tuning the peroxidase-like activity of Fe3O4 NPs at physiol. pH by using ATP. In presence of ATP, Fe3O4 NPs exhibited enhanced peroxidase-like activity over a wide range of pH and temps. Mechanistically, it was found that the ability of ATP to participate in single electron transfer reaction, through complexation with Fe3O4 NPs, results in the generation of hydroxyl radicals We utilized this ATP-mediated enhanced peroxidase-like activity of Fe3O4 NPs for single step detection of glucose with a colorimetric detection limit of 50μM.
- 63Peng J. Yang Q. Li W. Tan L. Xiao Y. Chen L. Hao Y. Qian Z. Erythrocyte-Membrane-Coated Prussian Blue/Manganese Dioxide Nanoparticles as H2O2-Responsive Oxygen Generators To Enhance Cancer Chemotherapy/Photothermal Therapy ACS Appl. Mater. Interfaces 2017 9 51 44410 44422Google Scholar63Erythrocyte-Membrane-Coated Prussian Blue/Manganese Dioxide Nanoparticles as H2O2-Responsive Oxygen Generators To Enhance Cancer Chemotherapy/Photothermal TherapyPeng, Jinrong; Yang, Qian; Li, Wenting; Tan, Liwei; Xiao, Yao; Chen, Lijuan; Hao, Ying; Qian, ZhiyongACS Applied Materials & Interfaces (2017), 9 (51), 44410-44422CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Because of the nontargeting release of anticancer drugs, conventional chemotherapy results in serious side effects and poor therapeutic outcomes. In addn., hypoxia situation in the tumor microenvironment also promotes the growth and metastasis of tumors. Multifunctional nanocarriers with stimuli-activation and hypoxia-relieving properties can help overcome some of these limitations. In this study, we have constructed a nanocarrier which is named PBMn-DOX@RBC. A Prussian blue/manganese dioxide (PBMn) nanoparticle is used as an oxygen precursor or catalyzer for H2O2 activation, and a red blood cell (RBC) membrane is used to increase the loading capacity of doxorubicin (DOX) and prolong the circulation time in vivo. H2O2 is overproduced in tumor tissues and tumor cells. It can be used as a stimulus to activate drug release. In the presence of H2O2, the hypoxia inside the tumors is relieved by the administration of PBMn-DOX@RBC. The generated oxygen disrupts the RBC coated on the surface of PBMn, which accelerates the release of DOX. RBCs also prolong the circulation time of the nanometer system in vivo. By combining the photothermal therapy (PTT) and chemotherapy, the tumor growth inhibition mediated by PBMn-DOX@RBC is further enhanced. PBMn-DOX@RBC fulfills the demands to relieve tumor hypoxia and enhance cancer chemotherapy/PTT.
- 64Wu L. H. Wan G. P. Hu N. He Z. Y. Shi S. H. Suo Y. R. Wang K. Xu X. F. Tang Y. L. Wang G. Z. Synthesis of Porous CoFe2O4 and Its Application as a Peroxidase Mimetic for Colorimetric Detection of H2O2 and Organic Pollutant Degradation Nanomaterials 2018 8 7 451Google Scholar64Synthesis of porous CoFe2O4 and its application as a peroxidase mimetic for colorimetric detection of H2O2 and organic pollutant degradationWu, Lihong; Wan, Gengping; Hu, Na; He, Zhengyi; Shi, Shaohua; Suo, Yourui; Wang, Kan; Xu, Xuefei; Tang, Yulin; Wang, GuizhenNanomaterials (2018), 8 (7), 451/1-451/16CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Porous CoFe2O4 was prepd. via a simple and controllable method to develop a low-cost, high-efficiency, and good-stability nanozyme. The morphol. and microstructure of the obtained CoFe2O4 was investigated by X-ray diffraction (XRD), XPS, transmission electron microscopy (TEM), high-resoln. TEM (HRTEM), sp. surface area and pore anal., and Raman spectroscopy. The results show that the annealing temp. has an important effect on the crystallinity, grain size, and sp. surface area of CoFe2O4. CoFe2O4 obtained at 300° C (CF300) exhibits the largest surface area (up to 204.1 m2 g-1) and the smallest grain size. The peroxidase-like activity of CoFe2O4 was further verified based on the oxidn. of peroxidase substrate 3,30',5,5'tetramethylbenzidine (TMB) in the presence of H2O2. The best peroxidase-like activity for CF300 should be ascribed to its largest surface area and smallest grain size. On this basis, an effective method of colorimetric detection H2O2 was established. In addn., the porous CoFe2O4 was also used for the catalytic oxidn. of methylene blue (MB), indicating potential applications in pollutant removal and water treatment.
- 65Liu L. Du B. J. Shang C. S. Wang J. Wang E. K. Construction of surface charge-controlled reduced graphene oxide-loaded Fe3O4 and Pt nanohybrid for peroxidase mimic with enhanced catalytic activity Anal. Chim. Acta 2018 1014 77 84Google Scholar65Construction of surface charge-controlled reduced graphene oxide-loaded Fe3O4 and Pt nanohybrid for peroxidase mimic with enhanced catalytic activityLiu, Lan; Du, Baoji; Shang, Changshuai; Wang, Jin; Wang, ErkangAnalytica Chimica Acta (2018), 1014 (), 77-84CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)Hybrid nanomaterials with synergistic effect are highly potential for developing advanced nanozymes. Herein, the authors designed a nanozyme assembled by polyethylenimine (PEI)-protected reduced graphene oxide anchoring Fe oxide (PRGI) and Pt nanoparticle using electrostatic interaction, PRGI/Pt nanohybrid. The different ratio of PRGI nanocomposite and Pt nanoparticle could control PRGI/Pt nanohybrid's surface charge and stability, which detd. PRGI/Pt nanohybrid's catalytic activity. At the mass ratio of 0.8, the as-obtained PRGI/Pt nanohybrid showed the highest catalytic ability, and was better than Pt nanoparticle at different pH and temp., although the PRGI/Pt nanohybrid showed lower affinity for TMB than Pt nanoparticle, which maybe attributed to the fact that PRGI/Pt nanohybrid possessed better product desorption ability or larger contact area. Also, PRGI/Pt nanohybrid showed much higher catalytic activity than the sum of PRGI nanocomposite and Pt nanoparticle, indicating the strong cooperation between PRGI nanocomposite and Pt nanoparticle. This study also provided a new way to conveniently construct nanozyme based on hybrid nanomaterials.
- 66Qiu N. Liu Y. Xiang M. Lu X. M. Yang Q. Guo R. A facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 nanohybrid for highly sensitive and selective detection of p-nitrophenol Sens. Actuators, B 2018 266 86 94Google Scholar66A facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 nanohybrid for highly sensitive and selective detection of p-nitrophenolQiu, Na; Liu, Yan; Xiang, Min; Lu, Ximing; Yang, Qian; Guo, RongSensors and Actuators, B: Chemical (2018), 266 (), 86-94CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)To date, fabricating high sensitive, stable and facile sensing strategy for detection of nitrophenol still remains a challenge in practical applications. This article demonstrates for the first time a facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 (3D GF/m-Fe3O4) heterogeneous nanozyme. Using a simple hydrothermal procedure, mesoporous Fe3O4 nanoparticles were in situ synthesized on 3D GF. The 3D GF/m-Fe3O4 nanohybrid possesses high peroxidase mimicking activity due to the synergistic effect of 3D GF and m-Fe3O4. Based on the peroxidase-like activity and p-nitrophenol-mediated inhibition controllability, the nanohybrid was successfully adopted in the sensitive and selective sensing detections of p-nitrophenol (PNP) with a low detection limit of 45nM. Moreover, 3D GF/m-Fe3O4 nanohybrid shows to be extremely robust, as it still retains its original response to PNP after being reused for 10 times. The method was applied to the detn. of PNP in spiked lake water and gave good recoveries. With the merits of high sensitivity and selectivity, simplification, recyclability, and excellent stability, this sensing platform not only expands the application of 3D GF and Fe3O4, but also provides an alternative technique to construct robust nanozyme with multiple functionalities for various biol. and environmental applications.
- 67Bhattacharjee R. Tanaka S. Moriam S. Masud M. K. Lin J. J. Alshehri S. M. Ahamad T. Salunkhe R. R. Nguyen N. T. Yamauchi Y. Hossain M. S. A. Shiddiky M. J. A. Porous nanozymes: the peroxidase-mimetic activity of mesoporous iron oxide for the colorimetric and electrochemical detection of global DNA methylation J. Mater. Chem. B 2018 6 29 4783 4791Google Scholar67Porous nanozymes: the peroxidase-mimetic activity of mesoporous iron oxide for the colorimetric and electrochemical detection of global DNA methylationBhattacharjee, Ripon; Tanaka, Shunsuke; Moriam, Sofia; Masud, Mostafa Kamal; Lin, Jianjian; Alshehri, Saad M.; Ahamad, Tansir; Salunkhe, Rahul R.; Nguyen, Nam-Trung; Yamauchi, Yusuke; Hossain, Md. Shahriar A.; Shiddiky, Muhammad J. A.Journal of Materials Chemistry B: Materials for Biology and Medicine (2018), 6 (29), 4783-4791CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Nanomaterials (nanozymes) with peroxidase-mimetic activity have been widely used in biosensing platforms as low-cost, relatively stable and prevailing alternatives to natural enzymes. Herein, the authors report on the synthesis and application of the peroxidase-mimetic activity of mesoporous iron oxide (MIO) for the detection of global DNA methylation in colorectal cancer cell lines. The target DNA was extd. and denatured to get ssDNA followed by direct adsorption onto the surface of a bare screen-printed gold electrode (SPGE). A 5-methylcytosine antibody (5mC) functionalized nanomaterial (MIO-5mC) was then used to recognize the methylcytosine groups present on the SPGE. The MIO-5mC conjugates catalyze the TMB soln. in the presence of hydrogen peroxide to give the colorimetric (i.e., naked-eye observation) and electrochem. detection of DNA methylation. The assay could successfully detect as low as 10% difference in the global DNA methylation level in synthetic samples and cell lines with good reproducibility and specificity (%RSD = <5%, for n = 3). This strategy avoids the use of natural enzyme horseradish peroxidase (HRP), traditional PCR based amplification and bisulfite treatment steps that are generally used in many conventional DNA methylation assays. The authors envisage that the assay could be a low-cost platform with great potential for genome-wide DNA methylation anal. in point-of-care applications.
- 68Yang Y. Zhu W. Feng L. Chao Y. Yi X. Dong Z. Yang K. Tan W. Liu Z. Chen M. G-Quadruplex-Based Nanoscale Coordination Polymers to Modulate Tumor Hypoxia and Achieve Nuclear-Targeted Drug Delivery for Enhanced Photodynamic Therapy Nano Lett. 2018 18 11 6867 6875Google Scholar68G-Quadruplex-Based Nanoscale Coordination Polymers to Modulate Tumor Hypoxia and Achieve Nuclear-Targeted Drug Delivery for Enhanced Photodynamic TherapyYang, Yu; Zhu, Wenjun; Feng, Liangzhu; Chao, Yu; Yi, Xuan; Dong, Ziliang; Yang, Kai; Tan, Weihong; Liu, Zhuang; Chen, MeiwanNano Letters (2018), 18 (11), 6867-6875CODEN: NALEFD; ISSN:1530-6984. (American Chemical Society)Photodynamic therapy (PDT) is a light-triggered therapy used to kill cancer cells by producing reactive oxygen species (ROS). Herein, a new kind of DNA nanostructure based on the coordination between calcium ions (Ca2+) and AS1411 DNA G quadruplexes to form nanoscale coordination polymers (NCPs) is developed via a simple method. Both chlorine e6 (Ce6), a photosensitizer, and hemin, an iron-contg. porphyrin, can be inserted into the G-quadruplex structure in the obtained NCPs. With further polyethylene glycol (PEG) modification, we obtain Ca-AS1411/Ce6/hemin@pHis-PEG (CACH-PEG) NCP nanostructure that enables the intranuclear transport of photosensitizer Ce6 to generate ROS inside cell nuclei that are the most vulnerable to ROS. Meanwhile, the inhibition of antiapoptotic protein B-cell lymphoma 2 (Bcl-2) expression by AS1411 allows for greatly improved PDT-induced cell apoptosis. Furthermore, the catalase-mimicking DNAzyme function of G-quadruplexes and hemin in those NCPs could decomp. tumor endogenous H2O2 to in situ generate oxygen so as to further enhance PDT by overcoming the hypoxia-assocd. resistance. This work develops a simple yet general method with which to fabricate DNA-based NCPs and presents an interesting concept of a nanoscale drug-delivery system that could achieve the intranuclear delivery of photosensitizers, the down-regulation of anti-apoptotic proteins, and the modulation of the unfavorable tumor microenvironment simultaneously for improved cancer therapy.
- 69Liu Y. Qin Y. L. Zheng Y. L. Qin Y. Cheng M. J. Guo R. A one-pot and modular self-assembly strategy for high-performance organized enzyme cascade bioplatforms based on dual-functionalized protein-PtNP@mesoporous iron oxide hybrid J. Mater. Chem. B 2019 7 1 43 52Google Scholar69A one-pot and modular self-assembly strategy for high-performance organized enzyme cascade bioplatforms based on dual-functionalized protein-PtNP@mesoporous iron oxide hybridLiu, Yan; Qin, Yuling; Zheng, Yuanlin; Qin, Yong; Cheng, Mengjun; Guo, RongJournal of Materials Chemistry B: Materials for Biology and Medicine (2019), 7 (1), 43-52CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Inspired by the delicate structure and prominent efficiency of natural multiple-enzyme systems, combining nanotechnologies such as nanomaterials, self-assemblies, and enzyme mimics is fascinating for the development of next-generation high-performance organized enzyme cascade bioplatforms. In our facile and convenient design, a dual-functionalized β-casein-Pt nanoparticles@mesoporous-Fe3O4 (CM-PtNP@m-Fe3O4) hybrid acts as both a nanozyme with outstanding peroxidase-like activity and a scaffold to immobilize and stabilize a natural oxidase, resulting in a high-performance organized enzyme cascade bioplatform for a one-pot assembly procedure. Owing to special physicochem. surface properties, the multipoint attachment of various interactions between natural enzymes and protein/inorg. hybrids leads to efficient immobilization of the enzyme with retained activity. The proposed cascade bioplatform provides superior cholesterol sensing, including simplicity (one-step detection), reusable enzymes (peroxidase mimic and oxidase), and excellent sensitivity (detection limit, 0.05 μM). To our knowledge, the bioplatform presented in this work shows the highest sensitivity for cholesterol detection among all reported colorimetric methods based on nanozymes. Therefore, the highly rationally designed protein/inorg. hybrid and dual-functional strategy used in this study will provide a facile one-pot and effective high-performance organized enzyme cascade bioplatform with potential applications in biosensing, biotransformation, decontamination, and biofuel.
- 70Zhang Q. T. Li M. F. Guo C. Y. Jia Z. Wan G. C. Wang S. F. Min D. Y. Fe3O4 Nanoparticles Loaded on Lignin Nanoparticles Applied as a Peroxidase Mimic for the Sensitively Colorimetric Detection of H2O2 Nanomaterials 2019 9 2 210Google Scholar70Fe3O4 nanoparticles loaded on lignin nanoparticles applied as a peroxidase mimic for the sensitively Colorimetric Detection of H2O2Zhang, Qingtong; Li, Mingfu; Guo, Chenyan; Jia, Zhuan; Wan, Guangcong; Wang, Shuangfei; Min, DouyongNanomaterials (2019), 9 (2), 210CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Lignin is the second largest naturally renewable resource and is primarily a byproduct of the pulp and paper industry; however, its inefficient use presents a challenge. In this work, Fe3O4 nanoparticles loaded on lignin nanoparticles (Fe3O4@LNPs) were prepd. by the self-assembly method and it possessed an enhanced peroxidase-like activity. Fe3O4@LNPs catalyzed the oxidn. of 3,30,5,50-tetramethylbenzidine (TMB) in the presence of H2O2 to generate a blue color, was observable by the naked eye. Under the optimal conditions, Fe3O4@LNPs showed the ability of sensitive colorimetric detection of H2O2within a range of 5-100μM and the limit of detection was 2μM. The high catalytic activity of Fe3O4@LNPs allows its prospective use in a wide variety of applications, including clin. diagnosis, food safety, and environmental monitoring.
- 71Zhang Z. J. Li Y. Q. Zhang X. H. Liu J. W. Molecularly imprinted nanozymes with faster catalytic activity and better specificity Nanoscale 2019 11 11 4854 4863Google Scholar71Molecularly imprinted nanozymes with faster catalytic activity and better specificityZhang, Zijie; Li, Yuqing; Zhang, Xiaohan; Liu, JuewenNanoscale (2019), 11 (11), 4854-4863CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Nanozymes are nanomaterials mimicking the activity of natural enzymes, while most nanozymes lack substrate specificity. Mol. imprinting on nanozymes provides a simple soln. to this problem, and the catalytic activity is also enhanced. To understand enhanced activity, a surface science approach is taken by dissecting the nanozyme reaction into adsorption of substrates, reaction, and product release. Each step is individually studied using reaction kinetics measurement, dynamic light scattering, UV-vis spectrometry. Enrichment of local substrate concn. due to imprinting is around 8-fold, and increased substrate concn. could contribute to increased activity. Diffusion of the substrate across the imprinted gel layer is studied by a pre-incubation expt., also highlighting the difference between imprinted and non-imprinted gel layers. The activation energy is measured and a substrate-imprinted sample had the lowest activation energy of 13.8 kJ mol-1. Product release is also improved after imprinting as indicated by isothermal titrn. calorimetry using samples resp. imprinted with the substrate and the product. This study has rationalized improved activity and specificity of molecularly imprinted nanozymes and may guide further rational design of such materials.
- 72Tanaka S. Masud M. K. Kaneti Y. V. Shiddiky M. J. A. Fatehmulla A. Aldhafiri A. M. Farooq W. A. Bando Y. Hossain M. S. A. Yamauchi Y. Enhanced Peroxidase Mimetic Activity of Porous Iron Oxide Nanoflakes ChemNanoMat 2019 5 4 506 513Google Scholar72Enhanced Peroxidase Mimetic Activity of Porous Iron Oxide NanoflakesTanaka, Shunsuke; Masud, Mostafa Kamal; Kaneti, Yusuf Valentino; Shiddiky, Muhammad J. A.; Fatehmulla, Amanullah; Aldhafiri, Abdullah M.; Farooq, W. Aslam; Bando, Yoshio; Hossain, Md. Shahriar A.; Yamauchi, YusukeChemNanoMat (2019), 5 (4), 506-513CODEN: CHEMSB; ISSN:2199-692X. (Wiley-VCH Verlag GmbH & Co. KGaA)Porous nanomaterials with superior peroxidase mimetic activity (nanozyme) at room temp. have gained increasing attention as potential alternatives to natural peroxidase enzymes. Herein, we report the application of porous iron oxide nanoflakes (IONFs), synthesized using the combination of solvothermal method and high-temp. calcination as peroxidase nanozyme for the oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The four IONF catalysts possess porous structures with a wide pore size distribution between 2-30 nm and high sp. surface areas around to 200 m2 g-1. The increase of calcination temp. of the IONFs from 250°C to 400°C resulted in a gradual decrease in their sp. surface area and Michaelis-Menten const. (Km) for TMB oxidn. The optimum IONF sample showed a much lower Km at 0.24 mM (towards TMB) compared to natural enzyme horseradish peroxidase (HRP) at 0.434 mM, revealing the promising potential of the as-prepd. IONFs as alternatives to HRP for biosensing applications.
- 73Wang Y. Li H. Guo L. Jiang Q. Liu F. A cobalt-doped iron oxide nanozyme as a highly active peroxidase for renal tumor catalytic therapy RSC Adv. 2019 9 33 18815 18822Google Scholar73A cobalt-doped iron oxide nanozyme as a highly active peroxidase for renal tumor catalytic therapyWang, Yixuan; Li, Hongjun; Guo, Lihua; Jiang, Qi; Liu, FengRSC Advances (2019), 9 (33), 18815-18822CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)The Fe3O4 nanozyme, the first reported nanozyme with intrinsic peroxidase-like activity, has been successfully employed for various diagnostic applications. However, only a few studies have been reported on the therapeutic applications of the Fe3O4 nanozyme partly due to its low affinity to the substrate H2O2. Herein, we report a new strategy for improving the peroxidase-like activity and affinity of the Fe3O4 nanozyme to H2O2 to generate reactive oxygen species (ROS) for kidney tumor catalytic therapy. We showed that cobalt-doped Fe3O4 (Co@Fe3O4) nanozymes possessed stronger peroxidase activity and a 100-fold higher affinity to H2O2 than the Fe3O4 nanozymes. The lysosome localization properties of Co@Fe3O4 enable Co@Fe3O4 to catalyze the decompn. of H2O2 at ultralow doses for the generation of ROS bursts to effectively kill human renal tumor cells both in vitro and in vivo. Moreover, our study provides the first evidence that the Co@Fe3O4 nanozyme is a powerful nanozyme for the generation of ROS bursts upon the addn. of H2O2 at ultralow doses, presenting a potential novel avenue for tumor nanozyme catalytic therapy.
- 74Li S. S. Shang L. Xu B. L. Wang S. H. Gu K. Wu Q. Y. Sun Y. Zhang Q. H. Yang H. L. Zhang F. R. Gu L. Zhang T. R. Liu H. Y. A Nanozyme with Photo-Enhanced Dual Enzyme-Like Activities for Deep Pancreatic Cancer Therapy Angew. Chem., Int. Ed. 2019 58 36 12624 12631Google Scholar74A nanozyme with photo-enhanced dual enzyme-like activities for deep pancreatic cancer therapyLi, Shanshan; Shang, Lu; Xu, Bolong; Wang, Shunhao; Gu, Kai; Wu, Qingyuan; Sun, Yun; Zhang, Qinghua; Yang, Hailong; Zhang, Fengrong; Gu, Lin; Zhang, Tierui; Liu, HuiyuAngewandte Chemie, International Edition (2019), 58 (36), 12624-12631CODEN: ACIEF5; ISSN:1433-7851. (Wiley-VCH Verlag GmbH & Co. KGaA)Nanozymes have attracted extensive interest owing to their high stability, low cost and easy prepn., esp. in the field of cancer therapy. However, the relatively low catalytic activity of nanozymes in the tumor microenvironment (TME) has limited their applications. Herein, we report a novel nanozyme (PtFe@Fe3O4) with dual enzyme-like activities for highly efficient tumor catalytic therapy. PtFe@Fe3O4 shows the intrinsic photothermal effect as well as photo-enhanced peroxidase-like and catalase-like activities in the acidic TME, thereby effectively killing tumor cells and overcoming the tumor hypoxia. Importantly, a possible photo-enhanced synergistic catalytic mechanism of PtFe@Fe3O4 was first disclosed. We believe that this work will advance the development of nanozymes in tumor catalytic therapy.
- 75Huang Y. L. Liang G. Z. Lin T. R. Hou L. Ye F. G. Zhao S. L. Magnetic Cu/Fe3O4@FeOOH with intrinsic HRP-like activity at nearly neutral pH for one-step biosensing Anal. Bioanal. Chem. 2019 411 17 3801 3810Google Scholar75Magnetic Cu/Fe3O4@FeOOH with intrinsic HRP-like activity at nearly neutral pH for one-step biosensingHuang, Yuanlin; Liang, Guangzhao; Lin, Tianran; Hou, Li; Ye, Fanggui; Zhao, ShulinAnalytical and Bioanalytical Chemistry (2019), 411 (17), 3801-3810CODEN: ABCNBP; ISSN:1618-2642. (Springer)The convenience of colorimetric sensors is useful for practical applications. In this work, the authors constructed a novel colorimetric sensor with magnetic sepn. ability that can be operated in nearly neutral conditions and achieve one-step detection of metabolites. Magnetic Cu doped Fe3O4@FeOOH magnetic nanocomposite (Cu/Fe3O4@FeOOH) with an oxygen vacancy was prepd. by a one-step self-assembly hydrothermal method, and fully characterized by different methods. The oxygen vacancy generated by the incorporation of Cu2+ cations into the Fe3O4@FeOOH structure was confirmed to be a vital reactive site for enhancing the catalytic activity, which opens up a new way of designing highly efficient enzyme mimics. Benefiting from its inherent horseradish-peroxidase-like activity, a simple and selective enzyme-based colorimetric sensor was developed for one-step detection of H2O2 and cholesterol, and 3,3',5,5'-tetramethylbenzidine was catalyzed by H2O2 to generate a colored product of oxidized 3,3',5,5'-tetramethylbenzidine for signaling. H2O2 and cholesterol can be linearly detected in the same range from 0.01 to 0.4 mmol L-1 with detection limits of 0.0075 mmol L-1 and 0.0082 mmol L-1, resp. The proposed colorimetric sensor has satisfactory reusability, accuracy, and practicability in human serum samples, indicating its potential application for the detection of different metabolites in the fields of life science and anal. science.
- 76Wang J. Huang F. Wang X. Wan Y. Xue Y. Cai N. Chen W. Yu F. Hierarchically structured Fe3O4-doped MnO2 microspheres as an enhanced peroxidase-like catalyst for low limit of detection Process Biochem. 2019 83 35 43Google Scholar76Hierarchically structured Fe3O4-doped MnO2 microspheres as an enhanced peroxidase-like catalyst for low limit of detectionWang, Jianzhi; Huang, Fei; Wang, Xianming; Wan, Yinjia; Xue, Yanan; Cai, Ning; Chen, Weimin; Yu, FaquanProcess Biochemistry (Oxford, United Kingdom) (2019), 83 (), 35-43CODEN: PBCHE5; ISSN:1359-5113. (Elsevier Ltd.)In order to lower the limit of detection of glucose, a peroxidase-like artificial enzyme with elevated catalysis capacity has been achieved. Fe3O4-doped MnO2 microspheres were fabricated through a two-step hydrothermal method for this purpose. TEM revealed that down-sized Fe3O4 nanoparticles were dispersed throughout the urchin-like MnO2 burrs. An addnl. XRD peak beyond Fe3O4 and MnO2 nanoparticles was obsd., indicating a dislocation structure was formed. The defect in structure as well as the synergistic effect would allow extra enzyme ability. Based on the steady-state kinetic analyses and Michaelis-Menten model, the Michaelis-Menten consts. (Km and vmax) were figured out. The results showed that the Fe3O4-MnO2 composite has elevated affinity toward the substrate TMB and H2O2. The limit of detection for glucose was estd. to be 0.75μmol L-1 based on the Fe3O4-MnO2 composite artificial enzyme. The superparamagnetic properties endowed the material easy sepn. The composite of this structure will provide a highly sensitive candidate method for accurate detection of glucose.
- 77Bian B. Liu Q. Y. Yu S. T. Peroxidase mimetic activity of porphyrin modified ZnFe2O4/reduced graphene oxide and its application for colorimetric detection of H2O2 and glutathione Colloids Surf., B 2019 181 567 575Google Scholar77Peroxidase mimetic activity of porphyrin modified ZnFe2O4/reduced graphene oxide and its application for colorimetric detection of H2O2 and glutathioneBian, Bing; Liu, Qingyun; Yu, ShitaoColloids and Surfaces, B: Biointerfaces (2019), 181 (), 567-575CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)Artificial nanoenzymes which can overcome some drawbacks of natural enzymes is a challenging topic in the biosensor field. Herein, we demonstrated 5,10,15,20 tetrakis (4-carboxylpheyl)-porphyrin modified magnetic ZnFe2O4 nanoparticles loaded on the surface of reduced graphene oxide (Por-ZnFe2O4/rGO), which exhibited intrinsic peroxidase-like activity and rapidly oxidized the peroxidase substrate 3, 3', 5, 5'-tetramethylbenzidine (TMB) into a blue product (OxTMB) distinguished by naked eyes. Interestingly, by comparative study of different nanomaterials ZnFe2O4 nanoparticles, ZnFe2O4/rGO and Por-ZnFe2O4, Por-ZnFe2O4/rGO was proved to possess the highest peroxidase-like activity. ESR (ESR) verified the catalytic activity of Por-ZnFe2O4/rGO for H2O2 was due to hydroxyl radical from decompn. of H2O2. Temp. and pH strongly affected the peroxidase-like activity of Por-ZnFe2O4/rGO nanocomposites. Under optimal conditions (pH = 4, 40°C), the constructed sensor based on the catalytic activity of the Por-ZnFe2O4/rGO could be conveniently used for colorimetric detection of H2O2 in the range of 0.7-30μM with the detection limit of 0.54μM. Moreover, the colorimetric sensor based on Por-ZnFe2O4/rGO exhibited a good linear response to glutathione (GHS) in the range of 2-40μM with a low detection limit of 0.76μM. The detection of GHS can be easily realized through the obvious color change by naked eyes without any complicated instrumentation.
- 78Masud M. K. Kim J. Billah M. M. Wood K. Shiddiky M. J. A. Nguyen N. T. Parsapur R. K. Kaneti Y. V. Alshehri A. A. Alghamidi Y. G. Alzahrani K. A. Adharvanachari M. Selvam P. Hossain M. S. A. Yamauchi Y. Nanoarchitectured peroxidase-mimetic nanozymes: mesoporous nanocrystalline alpha- or gamma-iron oxide? J. Mater. Chem. B 2019 7 35 5412 5422Google Scholar78Nanoarchitectured peroxidase-mimetic nanozymes: mesoporous nanocrystalline α- or γ-iron oxide?Masud, Mostafa Kamal; Kim, Jeonghun; Billah, Md. Motasim; Wood, Kathleen; Shiddiky, Mohammad J. A.; Nguyen, Nam-Trung; Parsapur, Rajesh Kumar; Kaneti, Yusuf Valentino; Alshehri, Abdulmohsen Ali; Alghamidi, Yousef Gamaan; Alzahrani, Khalid Ahmed; Adharvanachari, Murugulla; Selvam, Parasuraman; Hossain, Md. Shahriar A.; Yamauchi, YusukeJournal of Materials Chemistry B: Materials for Biology and Medicine (2019), 7 (35), 5412-5422CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Nanozymes (nanoparticles with enzyme-like properties) have attracted considerable attention in recent years owing to their intrinsic enzyme-like properties and broad application in the fields of ELISA based immunoassay and biosensing. Herein, the authors systematically study the influence of crystal phases (γ-Fe2O3 and α-Fe2O3) of mesoporous iron oxide (IO) on their peroxidase mimetic activity. In addn., the authors also demonstrated the applicability of these mesoporous IOs as nanozymes for detecting the glucose biomarker with a limit of detection (LOD) of 0.9 μM. Mesoporous γ-Fe2O3 shows high nanozyme activities (and magnetism) toward the catalytic oxidn. of chromogenic substances, such as 3,3',5,5'-tetramethylbenzidine (TMB) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)-ABTS, as well as for the colorimetric detection of glucose, compared to that of α-Fe2O3. The authors believe that this in-depth study of crystal structure based nanozyme activity will guide designing highly effective nanozymes based on iron oxide nanostructures for chem. sensing, biosensing and environmental remediation.
- 79Yin M. Y. Li S. Wan Y. Q. Feng L. P. Zhao X. T. Zhang S. Liu S. H. Cao P. Wang H. A selective colorimetric strategy for probing dopamine and levodopa through the mussel-inspired enhancement of Fe3O4 catalysis Chem. Commun. 2019 55 80 12008 12011Google Scholar79A selective colorimetric strategy for probing dopamine and levodopa through the mussel-inspired enhancement of Fe3O4 catalysisYin, Mengyuan; Li, Shuai; Wan, Yuqi; Feng, Luping; Zhao, Xiaoting; Zhang, Sheng; Liu, Shuhui; Cao, Peng; Wang, HuaChemical Communications (Cambridge, United Kingdom) (2019), 55 (80), 12008-12011CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)Mussel-inspired enhancement of Fe3O4 catalysis was discovered towards a highly selective and sensitive colorimetric strategy for the magnetic sepn.-based evaluation of dopamine and/or levodopa in urine, in which the specific interaction of bis-catechol-contg. analytes and mesoporous Fe3O4 NPs would form highly stable complexes of bis-catechol-Fe coordination.
- 80Zhu W. Li M. Chen S. Wang C. Lu X. Interfacial engineering regulating the peroxidase-like property of ternary composite nanofibers and their sensing applications Appl. Surf. Sci. 2019 491 138 146Google Scholar80Interfacial engineering regulating the peroxidase-like property of ternary composite nanofibers and their sensing applicationsZhu, Wendong; Li, Meixuan; Chen, Sihui; Wang, Ce; Lu, XiaofengApplied Surface Science (2019), 491 (), 138-146CODEN: ASUSEE; ISSN:0169-4332. (Elsevier B.V.)Interfacial engineering is an efficient and versatile strategy to accelerate the catalytic performance of functional nanomaterials. Herein, the authors have constructed ternary titanium dioxide (TiO2)@molybdenum disulfide (MoS2)/cobalt ferrite (CoFe2O4) composite nanofibers via a two-step hydrothermal reaction. Firstly, MoS2 nanosheets are grown on TiO2 nanofibers, then acting as an interfacial barrier to load ultrafine CoFe2O4 nanoparticles. Thanks to the synergistic effects among the interfacial interactions between MoS2 and TiO2 as well as CoFe2O4 component, the prepd. ternary TiO2@MoS2/CoFe2O4 composite nanofibers displayed much better catalytic activity than any single and bicomponent composite nanofibers for peroxidase mimicking. Due to the superior catalytic efficiency of the ternary TiO2@MoS2/CoFe2O4 composite nanofibers, a highly sensitive way for L-cysteine sensing was developed. The authors' work offers an advanced surface engineering soln. to construct a peroxidase-like catalyst to detect L-cysteine, with bright future in environmental science and biol. technol.
- 81Vallabani N. S. Singh S. Karakoti A. S. Investigating the role of ATP towards amplified peroxidase activity of Iron oxide nanoparticles in different biologically relevant buffers Appl. Surf. Sci. 2019 492 337 348Google Scholar81Investigating the role of ATP towards amplified peroxidase activity of Iron oxide nanoparticles in different biologically relevant buffersVallabani, N. V. Srikanth; Singh, Sanjay; Karakoti, Ajay S.Applied Surface Science (2019), 492 (), 337-348CODEN: ASUSEE; ISSN:0169-4332. (Elsevier B.V.)Evaluating the colloidal stability and enzyme mimetic nature of nanozymes at different buffer conditions is necessary to develop novel biosensing, biomedical and environmental applications. It is established that Fe3O4 nanoparticles show optimum peroxidase activity at pH 4 and it has also been shown that nucleotides like ATP can exhibit synergistic effect to enhance peroxidase activity at neutral pH. In this study we show the effect of buffers, pH and presence of ATP on peroxidase activity of Fe3O4 NPs and elucidate the mechanism involved in enhanced activity at neutral pH. Fe3O4 NPs colloidal stability was assessed over a period of 72 h from acidic to neutral pH. It was explained that OH radical generation from the synergistic combination of nucleotides and Fe3O4 NPs resulted in peroxidase activity at neutral pH and found that buffer concn. has a major effect on this activity. These findings challenge the existing theory of peroxidase activity demonstrated by Fe3O4 NPs at acidic pH. Moreover, the posibility can overcome the pH lacuna in designing novel biosensors and can also extend the heterogeneous Fenton reaction of Fe3O4 NPs over a wide range of pH.
- 82Gu Y. Huang Y. Qiu Z. Xu Z. Li D. Chen L. Jiang J. Gao L. Vitamin B2 functionalized iron oxide nanozymes for mouth ulcer healing Sci. China: Life Sci. 2020 63 1 68 79Google Scholar82Vitamin B2 functionalized iron oxide nanozymes for mouth ulcer healingGu, Yunhao; Huang, Yixin; Qiu, Zhiyue; Xu, Zhuobin; Li, Dandan; Chen, Lei; Jiang, Jing; Gao, LizengScience China: Life Sciences (2020), 63 (1), 68-79CODEN: SCLSCJ; ISSN:1674-7305. (Science China Press)Mouth ulcer is assocd. with inflammation and high risk of bacterial infection, which aggravates the patient's condition. Currently, there is no effective treatment for mouth ulcer. Herein, we report that vitamin-modified iron oxide nanoparticles improve the healing of mouth ulcer through anti-inflammation and antibacterial activities. We discovered that vitamin B2 (VB2) modified iron oxide nanoparticles performed enhanced peroxidase-like, catalase-like, and superoxide dismutase (SOD)-like activities, acting as typical iron oxide nanozymes (IONzymes) with triad activities. In particular, VB2 modification significantly improved the SOD-like activity, thus providing a reactive oxygen species (ROS)-scavenging ability. Cellular antioxidant expts. showed that vitamin B2 modified IONzymes (VB2-IONzymes) protect human oral keratinocytes (HOK) and BALB/3T3 cells from hydrogen peroxide (H2O2), and these cells have high biocompatibility to eukaryotic cells. In addn., VB2-IONzymes exerted an antibacterial activity against Streptococcus mutans, Staphylococcus aureus, and Escherichia coli. Importantly, VB2-IONzymes accelerated the recovery of mouth ulcer and reduced the local secretion of inflammatory factors in mouse ulcer model via ROS scavenging and antibacterial activity. Taken together, our work demonstrates that vitamin B2 modification endows iron oxide nanoparticles with enhanced enzyme-like activities and VB2-IONzymes may be a promising reagent in the treatment of mouth ulcer because of their intrinsic anti-inflammation and antibacterial capabilities.
- 83Adeniyi O. Sicwetsha S. Mashazi P. Nanomagnet-Silica Nanoparticles Decorated with Au@Pd for Enhanced Peroxidase-Like Activity and Colorimetric Glucose Sensing ACS Appl. Mater. Interfaces 2020 12 2 1973 1987Google Scholar83Nanomagnet-Silica Nanoparticles Decorated with Au@Pd for Enhanced Peroxidase-Like Activity and Colorimetric Glucose SensingAdeniyi, Omotayo; Sicwetsha, Simbongile; Mashazi, PhilaniACS Applied Materials & Interfaces (2020), 12 (2), 1973-1987CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Nanomagnet-silica shell (Fe3O4@SiO2) decorated with Au@Pd nanoparticles (NPs) were synthesized successfully. The characterization of Fe3O4@SiO2-NH2-Au@PdNPs was achieved using several spectroscopic and microscopic techniques. The quant. surface anal. was confirmed using XPS. The Fe3O4@[email protected] exhibited excellent peroxidase-like activity by effectively catalyzing the oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The absorption peaks at 370 and 652 nm confirmed the peroxidase-like activity of the Fe3O4@[email protected]. The Michaelis-Menten const. (Km) of 0.350 and 0.090 mM showed strong affinity toward H2O2 and TMB at Fe3O4@[email protected]. The mechanism of the peroxidase-like activity proceeds via an electron transfer process. A simple colorimetric sensor based on glucose oxidase and Fe3O4@[email protected] showed excellent selectivity and sensitivity towards the detection of glucose. The fabricated glucose biosensor exhibited a wide linear response toward glucose from 0.010 to 60.0 μM with an limit of detection of 60.0 nM and limit of quantification of 200 nM. The colorimetric biosensor based on Fe3O4@[email protected] as a peroxidase mimic was also successfully applied for the detn. of glucose concns. in serum samples. The synthesized Fe3O4@[email protected] nanozymes exhibited excellent potential as an alternative to horseradish peroxidase for low-cost glucose monitoring.
- 84Lyu H. Y. Zhao X. Yao X. X. Chen W. Liu Z. X. Gao L. N. Fan G. C. Zhu X. X. Liu Q. Y. Zhang X. X. Zhang X. 3,4:9,10-perylene tetracarboxylic acid-modified zinc ferrite with the enhanced peroxidase activity for sensing of ascorbic acid Colloids Surf., A 2020 586 124250Google Scholar843,4:9,10-perylene tetracarboxylic acid-modified zinc ferrite with the enhanced peroxidase activity for sensing of ascorbic acidLyu, Haoyuan; Zhao, Xin; Yao, Xiuxiu; Chen, Wei; Liu, Zhenxue; Gao, Linna; Fan, Gaochao; Zhu, Xixi; Liu, Qingyun; Zhang, Xianxi; Zhang, XiaoColloids and Surfaces, A: Physicochemical and Engineering Aspects (2020), 586 (), 124250CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)3,4:9,10-Perylene tetracarboxylic acid (PTCA) modified litchi-like zinc ferrite (ZnFe2O4) was prepd. by a simple hydrothermal method and was found to possess the enhanced peroxidase-like activity. Under the catalysis of PTCA-ZnFe2O4, the colorless colorimetric substrate 3,3',5,5'-tetramethylbenzidine (TMB) was rapidly oxidized by H2O2 and fast turned into blue color. Comparatively, the peroxidase-like activity of PTCA-ZnFe2O4 nanocomposites is higher than that of pure ZnFe2O4. In addn., it is hydroxyl radical that plays the key role in the process of catalysis reaction, which verified by fluorescent probe. On the basis of the higher peroxidase-like activity of PTCA-ZnFe2O4, a simple colorimetric biosensor platform has been proposed for detection of ascorbic acid (AA) in the linear range of 1-10μM with the detection limit of 0.834μM.
- 85Mazhani M. Alula M. T. Murape D. Development of a cysteine sensor based on the peroxidase-like activity of AgNPs@Fe3O4 core-shell nanostructures Anal. Chim. Acta 2020 1107 193 202Google Scholar85Development of a cysteine sensor based on the peroxidase-like activity of AgNPs@ Fe3O4 core-shell nanostructuresMazhani, Micode; Alula, Melisew Tadele; Murape, DavisonAnalytica Chimica Acta (2020), 1107 (), 193-202CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)A facile one step solvothermal procedure has been employed in generating magnetite-silver core-shell nanocomposites (AgNPs@ Fe3O4) with superior peroxidase-like catalytic property than bare magnetic nanoparticles (Fe3O4). The composites were characterized using different techniques such as TEM, SEM, XRD, and surface-enhanced IR absorption spectroscopy (SEIRA). In the presence of hydrogen peroxide, the synthesized composites were able to oxidize the colorless o-phenylenediamine (OPD) to a yellow color 2,3-diaminophenazine (DAP) with a better peroxidase-like activity than Fe3O4 alone. The obtained Km value of AgNPs@ Fe3O4 with H2O2 and OPD substrates are 28.0 mM and 2.91 mM, resp. These are substantially lower than previously reported values and indicate the strong binding affinity of the substrates towards AgNPs@ Fe3O4 nanocomposites. Based on the obstruction activity of cysteine on the peroxidase-like catalytic property of the nanocomposites, a sensor was developed for detection of cystein with a limit of detection as low as 87 nM and a wider range of linearity. The sensor also exhibited excellent selectivity against potentially interfering mols.
- 86Mo W. Yu J. Gao L. Liu Y. Wei Y. He R. Reversible Inhibition of Iron Oxide Nanozyme by Guanidine Chloride Front. Chem. 2020 8 491Google Scholar86Reversible inhibition of iron oxide nanozyme by guanidine chlorideMo, Wei-chuan; Yu, Jia; Gao, Li-zeng; Liu, Ying; Wei, Yan; He, Rong-qiaoFrontiers in Chemistry (Lausanne, Switzerland) (2020), 8 (), 00491CODEN: FCLSAA; ISSN:2296-2646. (Frontiers Media S.A.)Nanozymes have been widely applied in bio-assays in the field of biotechnol. and biomedicines. However, the physicochem. basis of nanozyme catalytic activity remains elusive. To test whether nanozymes exhibit an inactivation effect similar to that of natural enzymes, we used guanidine chloride (GuHCl) to disturb the iron oxide nanozyme (IONzyme) and obsd. that GuHCl induced IONzyme aggregation and that the peroxidase-like activity of IONzyme significantly decreased in the presence of GuHCl. However, the aggregation appeared to be unrelated to the quick process of inactivation, as GuHCl acted as a reversible inhibitor of IONzyme instead of a solo denaturant. Inhibition kinetic anal. showed that GuHCl binds to IONzyme competitively with H2O2 but non-competitively with tetramethylbenzidine. In addn., ESR spectroscopy showed that increasing GuHCl level of GuHCl induced a correlated pattern of changes in the activity and the state of the unpaired electrons of the IONzymes. This result indicates that GuHCl probably directly interacts with the iron atoms of IONzyme and affects the electron d. of iron, which may then induce IONzyme inactivation. These findings not only contribute to understanding the essence of nanozyme catalytic activity but also suggest a practically feasible method to regulate the catalytic activity of IONzyme.
- 87Ngo Y. L. T. Nguyen P. L. Choi W. M. Chung J. S. Hur S. H. Highly sensitive smartphone-integrated colorimetric glucose sensor based on MnFe2O4 - graphitic carbon nitride hybrid nanostructure Mater. Res. Bull. 2020 129 110910Google Scholar87Highly sensitive smartphone-integrated colorimetric glucose sensor based on MnFe2O4 - graphitic carbon nitride hybrid nanostructureNgo, Yen-Linh Thi; Nguyen, Phi Luan; Choi, Won Mook; Chung, Jin Suk; Hur, Seung HyunMaterials Research Bulletin (2020), 129 (), 110910CODEN: MRBUAC; ISSN:0025-5408. (Elsevier Ltd.)An extremely sensitive colorimetric glucose sensor was fabricated using a novel hybrid nanostructure comprised of manganese ferrite oxide- graphitic carbon nitride (MnFe2O4/g-C3N4). A neural network-based glucose anal. smartphone application can be used to easily monitor the glucose concn. with a high accuracy by only taking images with the smartphone camera. The as-synthesized MnFe2O4/g-C3N4 exhibited a very high affinity toward 3,3',5,5'-tetramethylbenzidine (TMB) and H2O2, as calcd. via Michaelis-Menten kinetics. The glucose sensors using as-synthesized MnFe2O4/g-C3N4 exhibit a wide linear range and very low detection limits of 20.5 nM for H2O2 and 17.3 nM for glucose, which are one of the lowest values ever reported. The instrumental anal. with XPS, XRD, and TEM revealed that MnFe2O4/g-C3N4 had a highly cryst. structure of MnFe2O4 and g-C3N4 with abundant oxygen vacancies and well-developed hybrid structures, which might result in excellent catalytic activity and glucose sensitivity.
- 88Boruah P. K. Darabdhara G. Das M. R. Polydopamine functionalized graphene sheets decorated with magnetic metal oxide nanoparticles as efficient nanozyme for the detection and degradation of harmful triazine pesticides Chemosphere 2021 268 129328Google Scholar88Polydopamine functionalized graphene sheets decorated with magnetic metal oxide nanoparticles as efficient nanozyme for the detection and degradation of harmful triazine pesticidesBoruah, Purna K.; Darabdhara, Gitashree; Das, Manash R.Chemosphere (2021), 268 (), 129328CODEN: CMSHAF; ISSN:0045-6535. (Elsevier Ltd.)A facile and an eco-friendly redn. and functionalization of reduced graphene oxide (rGO) sheets is carried out using dopamine and decorated with magnetic Fe3O4 nanoparticles with an av. size of 12 nm by a simple co-pptn. method which is established as an artificial nanozyme. Here, functionalization of graphene using dopamine has introduced several advantages and insights into this study. The Fe3O4 nanoparticles decorated functionalized rGO sheets (FDGs) nanozymes are characterized by X-ray diffraction (XRD), XPS, high-resoln. transmission electron microscopy (HRTEM), Raman spectroscopy, at. force microscopy (AFM), thermogravimetric (TGA) and vibrating sample magnetometer (VSM) anal. FDGs nanozymes exhibits dual characteristics towards detection and degrdn. of harmful simazine pesticide. The hydrogen bonding interactions between pesticide mols. and 3,3',5,5'-tetramethylbenzidine (TMB) causes inhibition of the catalytic activity of the FDGs towards oxidn. of TMB mol. Based on that, the presence of simazine pesticide in an aq. medium can be easily detd. and a certain value (2.24μM) of detection limit was achieved. The photocatalytic degrdn. of simazine is also executed and excellent photocatalytic activity was obsd. under irradn. of direct natural sunlight. The FDGs nanozyme is also reusable up to several times with insignificant loss in its catalytic activity towards simazine degrdn.
- 89Hermosilla E. Seabra A. B. Lourenco I. M. Ferreira F. F. Tortella G. Rubilar O. Highly sensitive oxidation of MBTH/DMAB by MnFe2O4 nanoparticles as a promising method for nanozyme-based sensor development Colloids Surf., A 2021 621 126585Google Scholar89Highly sensitive oxidation of MBTH/DMAB by MnFe2O4 nanoparticles as a promising method for nanozyme-based sensor developmentHermosilla, Edward; Seabra, Amedea B.; Lourenco, Isabella M.; Furlan, Fabio F.; Tortella, Gonzalo; Rubilar, OlgaColloids and Surfaces, A: Physicochemical and Engineering Aspects (2021), 621 (), 126585CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)Nanozymes are human-made nanomaterials with an intrinsic enzyme-like activity that offer a versatile alternative to natural enzymes for disease treatment, environmental remediation, and sensing. In this work, we proposed a novel spectrophotometric assay for detg. the oxidase-like activity of manganese ferrite nanoparticles (MnFe2O4 NPs) based on the oxidative coupling of 3-methyl-2-benzothiazolinone-hydrazone (MBTH) and 3-(dimethylamino) benzoic acid (DMAB). The effects of pH (2-8), temp. (20-50 °C), and kinetics parameters as the effect of substrate and nanoparticle concn. on the oxidase-like activity were evaluated. The MnFe2O4 NPs (∼3.19 ± 0.62 nm size) were synthesized by a co-pptn. method and characterized by TEM, SEM/EDS, VSM-SQUID, PXRD, and FTIR-ATR analyses. The optimum condition for MBTH/DMAB oxidn. catalyzed by MnFe2O4 NPs was obsd. at pH = 3.9 and 30 °C. The MBTH/DMAB oxidn. was adjusted to the Michaelis Menten kinetics model (R2 = 0.96) and showed a Km = 13.59 μM MBTH, kcat = 5.25 x 107 s-1 and kcat/Km = 3.86 x 1012 M-1 s-1. These high kcat and kcat/Km values indicate that the MBTH/DMAB couple is more sensitive than previously reported oxidase substrates by about three orders of magnitude. Hence, our findings open up a wide range of new potential applications on the development of more sensitive sensors based on the MBTH/DMAB reaction catalyzed by MnFe2O4 NPs for detecting several analytes (including alk. phosphatase, heavy metals, dissolved oxygen in water, among others) in the medical and environmental field.
- 90You S. M. Park J. S. Luo K. Jeong K. B. Adra H. J. Kim Y. R. Modulation of the peroxidase-like activity of iron oxide nanoparticles by surface functionalization with polysaccharides and its application for the detection of glutathione Carbohydr. Polym. 2021 267 118164Google Scholar90Modulation of the peroxidase-like activity of iron oxide nanoparticles by surface functionalization with polysaccharides and its application for the detection of glutathioneYou, Sang-Mook; Park, Jin-Sung; Luo, Ke; Jeong, Ki-Baek; Adra, Hazzel Joy; Kim, Young-RokCarbohydrate Polymers (2021), 267 (), 118164CODEN: CAPOD8; ISSN:0144-8617. (Elsevier Ltd.)Here, we employed three polysaccharides, such as dextran, hyaluronic acid, and chitosan, for surface modification of iron oxide nanoparticles (IONPs) and carried out in-depth investigation to elucidate the effect of surface functionalities on the peroxidase (POD) like activity of IONPs. The affinity of substrates to the catalytic site of IONPs was found to be detd. by the surface functional groups and hydration layer of polysaccharide coating on the surface of IONPs. The role of hydration layer was further confirmed by the results that the POD-like activity of IONPs coated with a certain polysaccharide having higher water holding capacity was significantly enhanced by salting-out reagent, such as ammonium chloride that is known to reduce the thickness of hydration layer. Moreover, the excellent catalytic activity of dextran-coated IONPs was successfully applied to develop a highly sensitive sensing system for the detection of glutathione (GSH) with a limit of detection of 2.3 nM.
- 91Bian B. Zhu X. X. Wu Q. Liu Y. Liu S. W. Liu Q. Y. Yu S. T. Pt and ZnFe2O4 Nanoparticles Immobilized on Carbon for the Detection of Glutathione ACS Appl. Nano Mater. 2021 4 9 9479 9488Google Scholar91Pt and ZnFe2O4 Nanoparticles Immobilized on Carbon for the Detection of GlutathioneBian, Bing; Zhu, Xixi; Wu, Qiong; Liu, Yue; Liu, Shiwei; Liu, Qingyun; Yu, ShitaoACS Applied Nano Materials (2021), 4 (9), 9479-9488CODEN: AANMF6; ISSN:2574-0970. (American Chemical Society)The catalytic activity of nanozymes has attracted much attention in biochem. analyses and the antibiosis and environmental fields. However, easy aggregation and difficulty in recycling greatly result in a decrease of their catalytic activity and further restrict their practical application. Herein, the authors immobilized Pt and ZnFe2O4 nanoparticles on poplar-wood-derived carbon (ZnFe2O4/C), which exhibited excellent peroxidase-like activity and high affinity toward H2O2 and 3,3',5,5'-tetramethylbenzidine (TMB). Moreover, it could be easily sepd. by an external magnetic force from the reaction system. A sensitive and selective colorimetric sensor for glutathione (GSH) was established based on Pt/ZnFe2O4/C nanoperoxidase. The linear range for GSH was 1-30 μM, and the limit of detection for GSH was calcd. as 0.38 μM. The colorimetric method was applied to det. GSH in real samples for injection.
- 92Singh E. Kaur M. Sharma S. Structural tuning of CTAB@MgFe2O4 nanocomposite as peroxidase mimic for H2O2 and glucose sensing Mater. Chem. Phys. 2021 271 124851Google Scholar92Structural tuning of CTAB@MgFe2O4 nanocomposite as peroxidase mimic for H2O2 and glucose sensingSingh, Ekjot; Kaur, Manpreet; Sharma, SuchetaMaterials Chemistry and Physics (2021), 271 (), 124851CODEN: MCHPDR; ISSN:0254-0584. (Elsevier B.V.)Natural enzymes have been used in food industry, biosensing and pharmaceuticals due to their high catalytic activity. Their catalytic potential is governed by the environmental conditions leading to their denaturation, thus increasing operational cost. Nanoparticles (NPs) as enzyme mimic is a thrust area of research. Magnesium ferrite nanoparticles (MgFe2O4 NPs) have gained focus of researchers due to good catalytic activity, stability and magnetic properties. Their catalytic activity can be enhanced by surface coating with suitable surfactant. In this work, CTAB (cetyl tri-Me ammonium bromide) coated MgFe2O4 NPs were prepd. and evaluated as peroxidase mimics. The XRD pattern of MgFe2O4 NPs confirmed single-phase formation of spinel structure with cubic geometry. FT-IR studies confirmed the presence of CTAB along with MgFe2O4 NPs. Surface area, Particle size and magnetic character of the NPs were evaluated by Brunauer Emmett Teller anal., Transmission Electron Microscope and Vibrating Sample Magnetometer resp. CTAB coated ferrite NPs showed increased surface area and decreased particle size as compared to pristine NPs. CTAB@MgFe2O4 in wt./wt. ratio (1:1) showed best peroxidase like activity towards o-dianisidine dihydrochloride and was influenced by pH, temp. and substrate concn. in soln. The synthesized CTAB@MgFe2O4 (1:1) based system could be used for colorimetric detection of H2O2 in the linear range of 20-300μM with a detection limit of 10μM and also for the detection for glucose in the range of 10-1000μM with a detection limit of 5μM.
- 93Zhu W. D. Cheng Y. Wang C. Lu X. F. Fiber-in-Tube Design of a CuFe2O4@Conducting Polymer with Synergistically Enhanced Peroxidase-like Activity for Total Antioxidant Capacity Assays ACS Sustainable Chem. Eng. 2021 9 44 14811 14820Google Scholar93Fiber-in-Tube Design of a CuFe2O4@Conducting Polymer with Synergistically Enhanced Peroxidase-like Activity for Total Antioxidant Capacity AssaysZhu, Wendong; Cheng, Ya; Wang, Ce; Lu, XiaofengACS Sustainable Chemistry & Engineering (2021), 9 (44), 14811-14820CODEN: ASCECG; ISSN:2168-0485. (American Chemical Society)The exploration of novel nanozymes with a high catalytic efficiency is crucial for their biosensing applications. Intricate hollow nanostructures possess distinct hierarchical open and porous features, usually presenting fascinating physicochem. properties. In this work, we have demonstrated the fabrication of a hierarchical fiber-in-tube CuFe2O4@conducting polypyrrole (PPy) nanostructure for peroxidase mimicking. Owing to the unique confined structure and synergistic effect, an exceptional peroxidase-like performance of CuFe2O4@PPy is achieved compared with the pristine CuFe2O4 nanofibers and PPy nanotubes alone. The prepd. catalyst displays a desirable long-term and cycling stability. The exptl. results demonstrate the key role of the formation of hydroxyl radicals for peroxidase mimicking. Based on the outstanding peroxidase-like activity, a high-efficiency ascorbic acid (AA) colorimetric sensor is developed, also displaying a satisfactory selectivity. Furthermore, according to the high sensitivity of the CuFe2O4@PPy nanotubes to detect AA, a total antioxidant capacity (TAC) sensor for a series of real samples has been developed. This work provides a new approach to construct efficient nanozymes with high efficiencies and develop a simple TAC assay with further vision in the fields of biosensing and food technol.
- 94Xing Y. Y. Chen M. L. Zhao Y. K. Xu J. B. Hou X. H. Triple-enzyme mimetic activity of Fe3O4@C@MnO2 composites derived from metal-organic frameworks and their application to colorimetric biosensing of dopamine Microchim. Acta 2022 189 1 1 10Google ScholarThere is no corresponding record for this reference.
- 95Yu F. Q. Huang Y. Z. Cole A. J. Yang V. C. The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detection Biomaterials 2009 30 27 4716 4722Google Scholar95The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detectionYu, Faquan; Huang, Yongzhuo; Cole, Adam J.; Yang, Victor C.Biomaterials (2009), 30 (27), 4716-4722CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)Aside from their superparamagnetic properties exploited in clin. magnetic resonance imaging (MRI), it was recently discovered that magnetic, iron oxide nanoparticles could function as an artificial, inorg. peroxidase. In this paper, the authors studied the impact of coating on the peroxidase activity of these nanoparticles. Nanoparticles with six different coating structures were synthesized and characterized by FTIR, TGA, TEM, size, zeta potential, and SQUID; and evaluated for peroxidase activity. Catalysis was found to follow Michaelis-Menten kinetics and peroxidase activity varied with respect to electrostatic affinity between nanoparticles and substrates, evidenced by differences in detd. kinetic parameters. Glucose detection was selected as a model system because glucose could be indirectly measured from the release of hydrogen peroxide after its oxidn. Nanoparticles with high peroxidase activity exhibited higher sensitivity toward glucose, showing a larger linear slope when compared with those of low activity. A significantly improved linear correlation and detection limit of measured glucose could be readily obtained by manipulating the nanoparticle coating. The authors' findings suggest that iron oxide nanoparticles can be tailor-made to possess improved peroxidase-like activity. Such enhancements could further widen nanoparticle scope in glucose detection and extend its peroxidase functionality to other biomedical applications.
- 96Zhang K. Zuo W. Wang Z. Y. Liu J. Li T. R. Wang B. D. Yang Z. Y. A simple route to CoFe2O4 nanoparticles with shape and size control and their tunable peroxidase-like activity RSC Adv. 2015 5 14 10632 10640Google Scholar96A simple route to CoFe2O4 nanoparticles with shape and size control and their tunable peroxidase-like activityZhang, Ke; Zuo, Wei; Wang, Zhiyi; Liu, Jian; Li, Tianrong; Wang, Baodui; Yang, ZhengyinRSC Advances (2015), 5 (14), 10632-10640CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)Recent studies have suggested that the phys. and chem. properties of nanoparticles (NPs) strongly depend on local chem. compn., size, and shape. Here, we report a new precursor-mediated growth of monodisperse magnetic cobalt ferrite (CoFe2O4) NPs with controlled size and shape. CoFe2O4 NPs with near corner-grown cubic, near cubic and polyhedron shape can be successfully prepd. by simply tuning the amt. of iron and cobalt acetylacetonates in oleic acid. Interestingly, the product shape varies from near corner-grown cubic to starlike by only changing the reaction temp. from 320 °C to 330 °C. These CoFe2O4 NPs exhibit size and shape-dependent peroxidase-like activity towards 3,3',5,5'-tetramethylbenzdine (TMB) in the presence of H2O2, and thus exhibited different levels of peroxidase-like activities, in the order of spherical > near corner-grown cubic > starlike > near cubic > polyhedron; this order was closely related to their particle size and crystal morphol. CoFe2O4NPs exhibited high stability in HAc-NaAc buffer (pH = 4.0) and high activity over a broad pH (2.5-6.0). Furthermore, the Michaelis consts. Km value for the CoFe2O4 NPs (0.006 mM) with TMB as the substrate was lower than HRP (0.062 mM) and Fe3O4 NPs (0.010 mM). After further surface functionalization with folic acid (FA), the folate-conjugated CoFe2O4 nanoparticles allow discrimination of HeLa cells (folate receptor overexpression) from NIH-3T3 cells (without folate receptor expression). Such investigation is of great significance for peroxidase nanomimetics with enhanced activity and utilization.
- 97Su L. Qin W. Zhang H. Rahman Z. U. Ren C. Ma S. Chen X. The peroxidase/catalase-like activities of MFe2O4 (M= Mg, Ni, Cu) MNPs and their application in colorimetric biosensing of glucose Biosens. Bioelectron. 2015 63 384 391Google Scholar97The peroxidase/catalase-like activities of MFe2O4 (M=Mg, Ni, Cu) MNPs and their application in colorimetric biosensing of glucoseSu, Li; Qin, Wenjie; Zhang, Huige; Rahman, Zia Ur; Ren, Cuiling; Ma, Sudai; Chen, XingguoBiosensors & Bioelectronics (2015), 63 (), 384-391CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)MFe2O4 (M=Mg, Ni, Cu) magnetic nanoparticles (MNPs) were found to have catalytic activities similar to those of biol. enzymes such as catalase and peroxidase. These nanomaterials, as bifunctional catalase/peroxidases (KatGs), not only could catalyze H2O2 to produce hydroxyl radicals, which oxidized peroxidase substrate to produce color, but also could catalyze the decompn. reaction of H2O2 into water and oxygen directly in the same condition through the catalase-like activity. And it was also found that the amt. of generated hydroxyl radicals and oxygen was related to the concn. of MFe2O4 (M=Mg, Ni, Cu) MNPs. The peroxidase-like catalytic behavior of MFe2O4 MNPs was analyzed in detail. Under the optimized conditions, NiFe2O4 MNPs were used as a colorimetric biosensor for the detection of 9.4×10-7-2.5×10-5 mol L-1 glucose with a limit of detection (LOD) of 4.5×10-7 mol L-1. The sensor was successfully applied to glucose detection in urine sample.
- 98Yuan F. Zhao H. Zang H. Ye F. Quan X. Three-dimensional graphene supported bimetallic nanocomposites with DNA regulated-flexibly switchable peroxidase-like activity ACS Appl. Mater. Interfaces 2016 8 15 9855 9864Google Scholar98Three-Dimensional Graphene Supported Bimetallic Nanocomposites with DNA Regulated-Flexibly Switchable Peroxidase-Like ActivityYuan, Fang; Zhao, Huimin; Zang, Hongmei; Ye, Fei; Quan, XieACS Applied Materials & Interfaces (2016), 8 (15), 9855-9864CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)A synergistic bimetallic enzyme mimetic catalyst, three-dimensional (3D) graphene/Fe3O4-AuNPs, was successfully fabricated which exhibited flexibly switchable peroxidase-like activity. Compared to the traditional 2D graphene-based monometallic composite, the introduced 3D structure, which was induced by the addn. of glutamic acid, and bimetallic anchoring approach dramatically improved the catalytic activity, as well as the catalysis velocity and its affinity for substrate. Herein, Fe3O4NPs acted as supporters for AuNPs, which contributed to enhance the efficiency of electron transfer. On the basis of the measurement of Mott-Schottky plots of graphene and metal anchored hybrids, the catalysis mechanism was elucidated by the decrease of Fermi level resulted from the chem. doping behavior. Notably, the catalytic activity was able to be regulated by the adsorption and desorption of single-stranded DNA mols., which laid a basis for its utilization in the construction of single-stranded DNA-based colorimetric biosensors. This strategy not only simplified the operation process including labeling, modification, and imprinting, but also protected the intrinsic affinity between the target and biol. probe. Accordingly, based on the peroxidase-like activity and its controllability, our prepd. nanohybrids was successfully adopted in the visualized and label-free sensing detections of glucose, sequence-specific DNA, mismatched nucleotides, and oxytetracycline.
- 99Lu N. Zhang M. Ding L. Zheng J. Zeng C. Wen Y. Liu G. Aldalbahi A. Shi J. Song S. Yolk–shell nanostructured Fe3O4@C magnetic nanoparticles with enhanced peroxidase-like activity for label-free colorimetric detection of H2O2 and glucose Nanoscale 2017 9 13 4508 4515Google Scholar99Yolk-shell nanostructured Fe3O4@C magnetic nanoparticles with enhanced peroxidase-like activity for label-free colorimetric detection of H2O2 and glucoseLu, Na; Zhang, Min; Ding, Lei; Zheng, Jing; Zeng, Caixia; Wen, Yanli; Liu, Gang; Aldalbahi, Ali; Shi, Jiye; Song, Shiping; Zuo, Xiaolei; Wang, LihuaNanoscale (2017), 9 (13), 4508-4515CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Herein, we have developed a simple and facile method to synthesize yolk-shell nanostructured Fe3O4@C nanoparticles (NPs) as a multifunctional biosensing platform for the label-free colorimetric detection of H2O2 and glucose. It was demonstrated that Fe3O4@C yolk-shell nanostructures (YSNs) retained the magnetic properties that can be used for sepn. and concn. Also importantly, the Fe3O4@C YSNs exhibited an intrinsic peroxidase-like activity that could quickly catalyze the enzyme substrate in the presence of H2O2 and produce a blue color. Compared to other similar ferric oxide-based NPs with different structures, Fe3O4@C YSNs exhibited greatly enhanced catalytic activities due to their unique structural features. Moreover, steady-state kinetics indicated the catalytic behaviors in agreement with the classic Michaelis-Menten models. Taking advantage of the high catalytic activity, Fe3O4@C YSNs were employed as novel peroxidase mimetics for label-free, rapid, sensitive, and specific colorimetric sensing of H2O2 and glucose, suggesting that Fe3O4@C YSNs have the potential for construction of portable sensors in the application of point-of-care (POC) diagnosis and on-site tests.
- 100Huang F. Wang J. Z. Chen W. M. Wan Y. J. Wang X. M. Cai N. Liu J. Yu F. Q. Synergistic peroxidase-like activity of CeO2-coated hollow Fe3O4 nanocomposites as an enzymatic mimic for low detection limit of glucose J. Taiwan Inst. Chem. Eng. 2018 83 40 49Google Scholar100Synergistic peroxidase-like activity of CeO2-coated hollow Fe3O4 nanocomposites as an enzymatic mimic for low detection limit of glucoseHuang, Fei; Wang, Jianzhi; Chen, Weimin; Wan, Yinjia; Wang, Xianming; Cai, Ning; Liu, Jie; Yu, FaquanJournal of the Taiwan Institute of Chemical Engineers (2018), 83 (), 40-49CODEN: JTICA8; ISSN:1876-1070. (Elsevier B.V.)Fe3O4@CeO2 yolk-shell nanocomposites (NCs) were synthesized by growing CeO2 onto Fe3O4 hollow nanoparticles (NPs) as an artificial peroxidase for the purpose of elevated activity. The resultant NCs exhibited strong magnetic response with narrow size distribution and high surface area. Owing to the individual peroxidase-like catalytic activity of CeO2 onto Fe3O4 nanoparticles (NPs), Fe3O4@CeO2 NCs was investigated to achieve synergistic catalytic activity. As a result, the as-prepd. Fe3O4@CeO2 NCs exhibited impressive peroxidase-like catalysis activity for 3, 3', 5, 5'-tetramethylbenzidine (TMB). Moreover, comparing with natural enzymes, hollow Fe3O4 NPs and CeO2 NPs, the Fe3O4@CeO2 NCs displayed a lower Michaelis const. (Km) value or stronger affinity to H2O2 and TMB. A limit of detection of 8.5μmol/L for H2O2 and 21μmol/L for glucose was attained with acceptable stability and reproducibility. The activation mechanism was also studies using competitive radical tests and ESR (ESR) anal.
- 101Lian J. J. Liu P. Li X. C. Gao L. N. Luo X. L. Zhang X. Shi Z. Q. Liu Q. Y. Perylene diimide-modified magnetic gamma-Fe2O3/CeO2 nanoparticles as peroxidase mimics for highly sensitive colorimetric detection of Vitamin C Appl. Organomet. Chem. 2019 33 5 e4884Google ScholarThere is no corresponding record for this reference.
- 102Cui M. Zhang H. R. Ouyang F. Guo Y. Q. Li R. F. Duan S. F. Xiong T. D. Wang Y. L. Wang X. Q. Dual Enzyme-Like Performances of PLGA Grafted Maghemite Nanocrystals and Their Synergistic Chemo/Chemodynamic Treatment for Human Lung Adenocarcinoma A549 Cells J. Biomed. Nanotechnol. 2021 17 6 1007 1019Google Scholar102Dual enzyme-like performances of PLGA grafted maghemite nanocrystals and their synergistic chemo/chemodynamic treatment for human lung adenocarcinoma A549 cellsCui, Miao; Zhang, Hui-Ru; Ouyang, Fan; Guo, Yu-Qi; Li, Rui-Fang; Duan, Shao-Feng; Xiong, Tian-Di; Wang, Yun-Long; Wang, Xue-QinJournal of Biomedical Nanotechnology (2021), 17 (6), 1007-1019CODEN: JBNOAB; ISSN:1550-7033. (American Scientific Publishers)In recent years, the emergence of non-toxic but catalytically active inorg. nanoparticles has attracted great attention for cancer treatment, but the therapeutic effect has been affected by the limited reactive oxygen species in tumors. Therefore, the combination of chemotherapy and chemodynamic therapy is regarded as a promising therapeutic strategy. In this paper, we reported the prepn. and bioactivity evaluation of poly(lactic acid-co-glycolic acid) (PLGA) grafted-γ-Fe2O3 nanoparticles with dual response of endogenous peroxidase and catalase like activities. Our hypothesis is that PLGA-grafted γ-Fe2O3 nanoparticles could be used as a drug delivery system for the anti-tumor drug doxorubicin to inhibit the growth of lung adenocarcinoma A549 cells; meanwhile, based on its mimic enzyme properties, this kind of nanoparticles could be combined with doxorubicin in the treatment of A549 cells. Our exptl. results showed that the PLGA-grafted γ-Fe2O3 nanoparticles could simulate the activity of catalase and decomp. hydrogen peroxide into H2O and oxygen in neutral tumor microenvironment, thus reducing the oxidative damage caused by hydrogenperoxide to lung adenocarcinoma A549 cells. In acidic microenvironment, PLGA grafted γ-Fe2O3 nanoparticles could simulate the activity of peroxidase and effectively catalyze the decompn. of hydrogen peroxide to generate highly toxic hydroxyl radicals, which could cause the death of A549 cells. Furthermore, the synergistic effect of peroxidase-like activity of PLGA-grafted γ-Fe2O3 nanoparticles and doxorubicin could accelerate the apoptosisand destruction of A549 cells, thus enhancing the antitumor effect of doxorubicin-loaded PLGA-grafted γ-Fe2O3 nanoparticles. Therefore, this study provides an effective nanoplatform based on dual inorg. biomimetic nanozymes for the treatment of lung cancer.
- 103Fu S. Y. Yang R. H. Ren J. J. Liu J. H. Zhang L. Xu Z. G. Kang Y. J. Xue P. Catalytically Active CoFe2O4 Nanoflowers for Augmented Sonodynamic and Chemodynamic Combination Therapy with Elicitation of Robust Immune Response ACS Nano 2021 15 7 11953 11969Google Scholar103Catalytically Active CoFe2O4 Nanoflowers for Augmented Sonodynamic and Chemodynamic Combination Therapy with Elicitation of Robust Immune ResponseFu, Shiyan; Yang, Ruihao; Ren, Junjie; Liu, Jiahui; Zhang, Lei; Xu, Zhigang; Kang, Yuejun; Xue, PengACS Nano (2021), 15 (7), 11953-11969CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)A hypoxic and acidic tumor microenvironment (TME) plays a significant role in cancer development through complex cellular signaling networks, and it is thus challenging to completely eradicate tumors via monotherapy. Here, PEGylated CoFe2O4 nanoflowers (CFP) with multiple enzymic activities, serving as bioreactors responsive to TME cues, were synthesized via a typical solvothermal method for augmented sonodynamic therapy (SDT) and chemodynamic therapy (CDT) with elicitation of robust immune response. The CFP occupying multivalent elements (Co2+/3+, Fe2+/3+) exhibited strong Fenton-like and catalase-like activity. In another aspect, CFP itself is a brand-new sonosensitizer for high-performance SDT based on ultrasound-triggered electron (e-)/hole (h+) pair sepn. from the energy band with promptness and high efficiency. With efficient enrichment in tumorous tissue as revealed by magnetic resonance imaging, CPF could generate •OH for CDT relying on Fenton-like reactions. Moreover, catalase-mimicking CFP could react with endogenous H2O2 to generate mol. oxygen, and high O2 level may promote the prodn. of 1O2 for SDT. What's more, the reactive oxygen species obtained from combined SDT/CDT could efficiently trigger immunogenic cell death through a synergistic therapy based on the elicitation of antitumor immunity with the aid of an immune checkpoint blockade for the sake of suppressing primary and distant tumors as well as lung metastasis. Taken together, this paradigm delivers useful insights for developing in-coming nanocomposites based on cobalt ferrite for cancer theranostics.
- 104Liu Y. S. Wang X. J. Li X. Z. Qiao S. S. Huang G. D. Hermann D. M. Doeppner T. R. Zeng M. L. Liu W. Xu G. L. Ren L. J. Zhang Y. Liu W. L. Casals E. Li W. P. Wang Y. C. A Co-Doped Fe3O4 Nanozyme Shows Enhanced Reactive Oxygen and Nitrogen Species Scavenging Activity and Ameliorates the Deleterious Effects of Ischemic Stroke ACS Appl. Mater. Interfaces 2021 13 39 46213 46224Google Scholar104A Co-Doped Fe3O4 Nanozyme Shows Enhanced Reactive Oxygen and Nitrogen Species Scavenging Activity and Ameliorates the Deleterious Effects of Ischemic StrokeLiu, Yunsheng; Wang, Xiaojun; Li, Xiangzhu; Qiao, Shanshan; Huang, Guodong; Hermann, Dirk Matthias; Doeppner, Thorsten Roland; Zeng, Muling; Liu, Wei; Xu, Gelin; Ren, Lijie; Zhang, Yuan; Liu, Wenlan; Casals, Eudald; Li, Weiping; Wang, Ya-ChaoACS Applied Materials & Interfaces (2021), 13 (39), 46213-46224CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Acute ischemic stroke has become the major cause of mortality and disability worldwide. Following ischemic stroke, the reperfusion injury is mainly mediated by the burst of reactive oxygen and nitrogen species (RONS). Therefore, blocking the excessive prodn. or removing RONS holds great promise as a potential therapeutic strategy. Herein, we developed a Co-doped Fe3O4 nanozyme that is capable of scavenging H2O2, O2·-, ·NO, and ONOO- in vitro and in vivo and provides neuroprotection against ischemic stroke. In vitro expts. showed that pre-incubation with the Co-Fe3O4 nanozyme could prevent neurotoxicity and neuroinflammation induced by H2O2 or lipopolysaccharide, resp., in HT22 cells. After i.v. administration, the Co-Fe3O4 nanozyme showed no signs of toxicity in peripheral organs of C57BL/6J mice, even after prolonged delivery for 4 wk. In permanent photothrombotic stroke model and transient middle cerebral artery occlusion stroke model, the Co-Fe3O4 nanozyme specifically accumulated in the infarct rim at 72 h post-stroke and was endocytosed by neurons, astrocytes, microglia, and endothelial cells. Importantly, the Co-Fe3O4 nanozyme delivery reduced the infarct vol. in both stroke models. The observation that the Co-Fe3O4 nanozyme was efficacious in two well-characterized ischemic stroke models provides strong evidence that it represents a powerful tool for targeting oxidative and nitrosative stress in the ischemic brain.
- 105Raineri M. Winkler E. L. Torres T. E. Vasquez Mansilla M. Nadal M. S. Zysler R. D. Lima E. Effects of biological buffer solutions on the peroxidase-like catalytic activity of Fe3O4 nanoparticles Nanoscale 2019 11 39 18393 18406Google Scholar105Effects of biological buffer solutions on the peroxidase-like catalytic activity of Fe3O4 nanoparticlesRaineri, Mariana; Winkler, Elin L.; Torres, Teobaldo E.; Vasquez Mansilla, Marcelo; Nadal, Marcela S.; Zysler, Roberto D.; Lima, EnioNanoscale (2019), 11 (39), 18393-18406CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)In our study, IONP activity was evaluated in different buffering solns. under different exptl. conditions and predominant free radical species were measured by ESR using the spin-trap 5,5-dimethyl-1-pyrroline N-oxide (DMPO). The formation kinetics of the reactive oxygen species were studied by UV-visible spectroscopy with TMB and DAB peroxidase substrates. We found that the highest catalytic oxidn. of peroxidase substrates and free radical generation were achieved in acetate buffer, while phosphate buffer inhibited the peroxidase-like activity of IONPs in a concn. dependent manner. When emulating the physiol. conditions, a lower catalytic activity was obsd. at pH 7.4. Also, in phosphate buffered saline (PBS), we obsd. an enhancement in the peroxidase substrate oxidn. rate that was not accompanied by an increase in DMPO/adduct formation which could be related to a non-specific oxidn. catalyzed by the chloride ion. Similar observations were found after the addn. of a bicarbonate to HEPES buffer. TMB oxidn. did not occur when the reaction was conducted with free iron ions from metal salts with the same concn. of the IONPs (0.33 Fe2+ and 0.66 Fe3+). These results indicate that biol. buffering solns. need to be carefully considered when evaluating IONP catalytic activity and their potential toxicol. effects since under physiol. conditions of pH, salinity and buffering species, the peroxidase-like activity of IONPs is dramatically reduced.
- 106Gao L. Yan X. Discovery and current application of nanozyme Prog. Biochem. Biophys. 2013 40 10 892 902Google ScholarThere is no corresponding record for this reference.
- 107Qin T. Ma R. Yin Y. Miao X. Chen S. Fan K. Xi J. Liu Q. Gu Y. Yin Y. Catalytic inactivation of influenza virus by iron oxide nanozyme Theranostics 2019 9 23 6920Google Scholar107Catalytic inactivation of influenza virus by iron oxide nanozymeQin, Tao; Ma, Ruonan; Yin, Yinyan; Miao, Xinyu; Chen, Sujuan; Fan, Kelong; Xi, Juqun; Liu, Qi; Gu, Yunhao; Yin, Yuncong; Hu, Jiao; Liu, Xiufan; Peng, Daxin; Gao, LizengTheranostics (2019), 9 (23), 6920-6935CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Influenza poses a severe threat to human health in the world. However, developing a universal anti-viral strategy has remained challenging due to the presence of diverse subtypes as well as its high mutation rate, resulting in antigenic shift and drift. Here we developed an antiviral strategy using iron oxide nanozymes (IONzymes) to target the lipid envelope of the influenza virus. Methods: We evaluated the antiviral activities of our IONzymes using a hemagglutination assay, together with a 50% tissue culture infectious doses (TCID50) method. Lipid peroxidn. of the viral envelope was analyzed using a maleic dialdehyde (MDA) assay and transmission electron microscopy (TEM). The neighboring viral proteins were detected by western blotting. Results: We show that IONzymes induce envelope lipid peroxidn. and destroy the integrity of neighboring proteins, including hemagglutinin, neuraminidase, and matrix protein 1, causing the inactivation of influenza A viruses (IAVs). Furthermore, we show that our IONzymes possess a broad-spectrum antiviral activity on 12 subtypes of IAVs (H1~ H12). Lastly, we demonstrate that applying IONzymes to a facemask improves the ability of virus protection against 3 important subtypes that pose a threat to human, including H1N1, H5N1, and H7N9 subtype. Conclusion: Together, our results clearly demonstrate that IONzymes can catalyze lipid peroxidn. of the viral lipid envelope to inactivate enveloped viruses and provide protection from viral transmission and infection.
- 108Hong C. Meng X. He J. Fan K. Yan X. Nanozyme: A promising tool from clinical diagnosis and environmental monitoring to wastewater treatment Particuology 2022 71 90 107Google Scholar108Nanozyme: A promising tool from clinical diagnosis and environmental monitoring to wastewater treatmentHong, Chaoyi; Meng, Xiangqin; He, Jiuyang; Fan, Kelong; Yan, XiyunParticuology (2022), 71 (), 90-107CODEN: PARTC5; ISSN:1674-2001. (Elsevier B.V.)A review. Natural enzymes, owing to their outstanding catalytic efficiency and substrate specificity, have been used in a variety of applications including clin. diagnosis, environmental monitoring and wastewater treatment. However, they face inevitable problems such as relatively high cost and lack of stability, dramatically hindering their practical applications in the industry. Recently, a class of nanomaterial that possesses intrinsic enzyme-like properties, nanozyme, has emerged exhibiting numerous advantages over its natural counterpart and has been used as a viable enzyme alternative. In the past decade there are many reviews on nanozyme. The previous discussions tend to view nanozyme as a type of nanomaterial rather than an enzyme. However, it is the enzyme-like activity of nanozymes that provides foundation for their application and nanozymes with the same enzymic activity usually have some regularity in application. Herein, in this review, we attempt to classify nanozymes by their enzyme-like activity to explain the application principle and relevant cases of nanozymes in clin. diagnosis, environmental monitoring and wastewater treatment, expecting to promote deeper thinking of nanozymes as enzyme mimics and provide useful guidance for future research.
- 109Chen M. Deng G. He Y. Li X. Liu W. Wang W. Zhou Z. Yang H. Yang S. Ultrasound-enhanced generation of reactive oxygen species for MRI-guided tumor therapy by the Fe@Fe3O4-Based peroxidase-mimicking nanozyme ACS Appl. Bio Mater. 2019 3 1 639 647Google ScholarThere is no corresponding record for this reference.
- 110Shi C. Li Y. Gu N. Iron-Based Nanozymes in Disease Diagnosis and Treatment ChemBioChem 2020 21 19 2722 2732Google Scholar110Iron-Based Nanozymes in Disease Diagnosis and TreatmentShi, Chu; Li, Yan; Gu, NingChemBioChem (2020), 21 (19), 2722-2732CODEN: CBCHFX; ISSN:1439-4227. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. Iron-based nanozymes are currently one of the few clin. inorg. nanoparticles for disease diagnosis and treatment. Overcoming the shortcomings of natural enzymes, such as easy inactivation and low yield, combined with their special nanometer properties and magnetic functions, iron-based nanozymes have broad prospects in biomedicine. This minireview summarizes their prepn., biol. activity, catalytic mechanism, and applications in diagnosis and treatment of diseases. Finally, challenges to their future development and the trends of iron-based nanozymes are discussed. The purpose of this minireview is to better understand and reasonably speculate on the rational design of iron-based nanozymes as an increasingly important new paradigm for diagnostics.
- 111Ji S. Jiang B. Hao H. Chen Y. Dong J. Mao Y. Zhang Z. Gao R. Chen W. Zhang R. Matching the kinetics of natural enzymes with a single-atom iron nanozyme Nat. Catal. 2021 4 5 407 417Google Scholar111Matching the kinetics of natural enzymes with a single-atom iron nanozymeJi, Shufang; Jiang, Bing; Hao, Haigang; Chen, Yuanjun; Dong, Juncai; Mao, Yu; Zhang, Zedong; Gao, Rui; Chen, Wenxing; Zhang, Ruofei; Liang, Qian; Li, Haijing; Liu, Shuhu; Wang, Yu; Zhang, Qinghua; Gu, Lin; Duan, Demin; Liang, Minmin; Wang, Dingsheng; Yan, Xiyun; Li, YadongNature Catalysis (2021), 4 (5), 407-417CODEN: NCAACP; ISSN:2520-1158. (Nature Portfolio)Abstr.: Developing artificial enzymes with the excellent catalytic performance of natural enzymes has been a long-standing goal for chemists. Single-atom catalysts with well-defined at. structure and electronic coordination environments can effectively mimic natural enzymes. Here, we report an engineered FeN3P-centered single-atom nanozyme (FeN3P-SAzyme) that exhibits comparable peroxidase-like catalytic activity and kinetics to natural enzymes, by controlling the electronic structure of the single-atom iron active center through the precise coordination of phosphorus and nitrogen. In particular, the engineered FeN3P-SAzyme, with well-defined geometric and electronic structures, displays catalytic performance that is consistent with Michaelis-Menten kinetics. We rationalize the origin of the high enzyme-like activity using d. functional theory calcns. Finally, we demonstrate that the developed FeN3P-SAzyme with superior peroxidase-like activity can be used as an effective therapeutic strategy for inhibiting tumor cell growth in vitro and in vivo. Therefore, SAzymes show promising potential for developing artificial enzymes that have the catalytic kinetics of natural enzymes. [graphic not available: see fulltext].
- 112Xi J. Zhang R. Wang L. Xu W. Liang Q. Li J. Jiang J. Yang Y. Yan X. Fan K. A nanozyme‐based artificial peroxisome ameliorates hyperuricemia and ischemic stroke Adv. Funct. Mater. 2021 31 9 2007130Google Scholar112A Nanozyme-Based Artificial Peroxisome Ameliorates Hyperuricemia and Ischemic StrokeXi, Juqun; Zhang, Ruofei; Wang, Liming; Xu, Wei; Liang, Qian; Li, Jingyun; Jiang, Jian; Yang, Yili; Yan, Xiyun; Fan, Kelong; Gao, LizengAdvanced Functional Materials (2021), 31 (9), 2007130CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)Artificial peroxisome has drawn a lot of attentions for its usefulness in fabricating protocell system and great potential in treating diseases. However, it is still a significant challenge to prep. a practicable artificial peroxisome to complement multiple and stable functions under physiol. condition. Herein, a novel strategy is reported to design an artificial peroxisome using a nanozyme to accommodate multiple enzyme-like activities that mimics those enzymes in natural peroxisome. The enzymic active sites are introduced into graphitized moieties on the shell of a hollow carbon nanozyme by doping iron and nitrogen to form Fe-N4 coordination and at. Fe cluster. With Fe clusters as reversible cofactors and Fe-N4 as prosthetic group, the resulted carbon nanozyme exhibits stable and multiple peroxisomal-like activities, including catalase, uricase, superoxide dismutase, peroxidase, and oxidase, which is referred as nanozyme-based artificial peroxisome (pero-nanozysome). This pero-nanozysome shows therapeutic effect for treating hyperuricemia and protecting neurons from free radicals generated during ischemic stroke by employing the tandem activities of uricase-catalase and superoxide-dismutase-catalase, resp. This study indicates that the pero-nanozysome is a promising candidate to design artificial peroxisome performing in vivo functions.
- 113Zhang R. Fan K. Yan X. Nanozymes: created by learning from nature Sci. China: Life Sci. 2020 63 8 1183 1200Google Scholar113Nanozymes: created by learning from natureZhang Ruofei; Fan Kelong; Yan Xiyun; Fan Kelong; Yan XiyunScience China. Life sciences (2020), 63 (8), 1183-1200 ISSN:.Nanozymes, a type of nanomaterials with enzyme-like activity, have shown great potential to replace natural enzymes in many fields such as biochemical detection, environmental management and disease treatment. However, the catalytic efficiency and substrate specificity of nanozymes still need improvement. To further optimize the enzymatic properties of nanozymes, recent studies have introduced the structural characteristics of natural enzymes into the rational design of nanozymes, either by employing small molecules to mimic the cofactors of natural enzymes to boost nanozymes' catalytic potential, or by simulating the active center of natural enzymes to construct the nanostructure of nanozymes. This review introduces the commonly used bio-inspired strategies to create nanozymes, aiming at clarifying the current progress and bottlenecks. Advances and challenges focusing on the research of bio-inspired nanozymes are outlined to provide ideas for the de novo design of ideal nanozymes.
- 114Zhang R. Yan X. Fan K. Nanozymes inspired by natural enzymes Acc. Mater. Res. 2021 2 7 534 547Google Scholar114Nanozymes Inspired by Natural EnzymesZhang, Ruofei; Yan, Xiyun; Fan, KelongAccounts of Materials Research (2021), 2 (7), 534-547CODEN: AMRCDA; ISSN:2643-6728. (American Chemical Society)A review. Nanozymes, nanomaterials with enzyme-like activities with high structural stability, adjustable catalytic activity, functional diversity, recyclability, and feasibility in large-scale prepn., have become a hot spot in the field of artificial enzymes in recent years and are expected to become potential surrogates and competitors for natural enzymes in practical applications. With the development of in-depth research and a wide range of application requirements, creating nanozymes with catalytic performance comparable to or even surpassing that of natural enzymes has been the key research topic in this field. Most of the nanozymes reported in the past were obtained based on random synthesis and screening, for which the catalytic efficiency is far inferior to that of natural enzymes. Natural enzymes that have evolved over hundreds of millions of years have developed a lot of high-efficiency catalysis know-how hidden in their structural features. To create highly active nanozymes, we assumed that there is a general structure-activity relationship between nanozymes and natural enzymes and proposed the nanozyme optimization strategy by grafting the catalytic principles of natural enzymes into the rational design of nanozymes. On the basis of this bioinspired strategy, a series of nanozymes that exhibit similar catalytic activities that are closer to or even beyond those of natural enzymes have been successfully synthesized. By now, rationally designed high-activity bioinspired nanozymes have become a hot topic in the current research on nanozymes. In this Account, we focus on recent representative research progress in the systemic design and construction of bioinspired nanozymes and are devoted to introducing strategic concepts in the bioinspired optimization of nanozymes. We show that the de novo design of nanozymes by simulating the amino acid microenvironment and using metal-free architecture and the coordination structure of metal active sites in natural enzymes is an effective strategy for significantly improving the catalytic performance of nanozymes. A future perspective of the challenges and countermeasures of bioinspired nanozymes is proposed on the basis of these achievements. We hope that the biol. inspired perception will arouse widespread interest in fundamental research and practical applications as well as provide inspiration for the rational design of nanozymes.
- 115Liu S. Lu F. Xing R. Zhu J. J. Structural effects of Fe3O4 nanocrystals on peroxidase‐like activity Chem. - Eur. J. 2011 17 2 620 625Google Scholar115Structural Effects of Fe3O4 Nanocrystals on Peroxidase-Like ActivityLiu, Shanhu; Lu, Feng; Xing, Ruimin; Zhu, Jun-JieChemistry - A European Journal (2011), 17 (2), 620-625, S620/1-S620/4CODEN: CEUJED; ISSN:0947-6539. (Wiley-VCH Verlag GmbH & Co. KGaA)The catalytic activity of nanocrystal catalysts depends strongly on their structures. Herein, we report three distinct structures of Fe3O4 nanocrystals, cluster spheres, octahedra, and triangular plates, prepd. by a similar hydrothermal procedure. Addnl., the three Fe3O4 nanostructures were used as peroxidase nanomimetics and the correlation between the catalytic activities and the structures was first explored by using 3,3',5,5'-tetramethylbenzidine and H2O2 as peroxidase substrates. The results showed that the peroxidase-like activities of the Fe3O4 nanocrystals were structure dependent and followed the order cluster spheres>triangular plates>octahedra; this order was closely related to their preferential exposure of catalytically active iron atoms or crystal planes. Such investigation is of great significance for peroxidase nanomimetics with enhanced activity and utilization.
- 116Puvvada N. Panigrahi P. K. Mandal D. Pathak A. Shape dependent peroxidase mimetic activity towards oxidation of pyrogallol by H2O2 RSC Adv. 2012 2 8 3270 3273Google Scholar116Shape dependent peroxidase mimetic activity towards oxidation of pyrogallol by H2O2Puvvada, Nagaprasad; Panigrahi, Pravas Kumar; Mandal, Dhritabrata; Pathak, AmitaRSC Advances (2012), 2 (8), 3270-3273CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)Truncated octahedron shaped magnetite nanoparticles have been prepd. via a chem. method and subsequently their shape dependent peroxidase mimetic activity has been verified using pyrogallol substrate. Their peroxidase mimetic activity has been found to be superior to that of spherical-shaped nanoparticles.
- 117Cheng X.-L. Jiang J.-S. Jiang D.-M. Zhao Z.-J. Synthesis of rhombic dodecahedral Fe3O4 nanocrystals with exposed high-energy {110} facets and their peroxidase-like activity and lithium storage properties J. Phys. Chem. C 2014 118 24 12588 12598Google Scholar117Synthesis of Rhombic Dodecahedral Fe3O4 Nanocrystals with Exposed High-Energy {110} Facets and Their Peroxidase-like Activity and Lithium Storage PropertiesCheng, Xun-Liang; Jiang, Ji-Sen; Jiang, Dong-Mei; Zhao, Zhen-JieJournal of Physical Chemistry C (2014), 118 (24), 12588-12598CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Large quantities of monodispersed single-crystal rhombic dodecahedral (RD) Fe3O4 nanocrystals (NCs) bounded by high-energy {110} facets were prepd. by using a 1-pot toluene-H2O 2-phase interfacial reaction method. By adjusting the Fe(acac)3 concn. and the vol. of hydrazine hydrate, their sizes could be controlled at 60-100 nm. The formation of these RD Fe3O4 NCs with exposed high-energy {110} surfaces might be the result of selective stabilization of such unstable facets by the C17H33COO- based on FTIR anal. The magnetic measurement revealed that the satn. magnetization (Ms) of the RD Fe3O4 NCs was 85 emu/g at room temp. When used as peroxidase mimetics, the prepd. RD Fe3O4 NCs showed excellent peroxidase-like catalytic activity toward oxidn. of the substrate 3,3',5,5'-tetramethylbenzidine and degrdn. of the aniline compd. methylene blue dye in the presence of H2O2. When tested as anode materials for Li-ion batteries, these as-obtained RD Fe3O4 NCs showed a high initial discharge capacity of 1147 mA-h g-1 at the c.d. of 0.2 C, a good cycle performance (362 mA-h g-1 at 0.2 C after 100 cycles and 191 mA-h g-1 at 1 C up to 130 cycles), and good rate capability at high current rates of 1-4 C.
- 118Zhao J. Zhang S. Wang D. Jia C. Yang P. Synthesis of Rhombic Dodecahedral Fe3O4 Single Crystals Towards Their High Peroxidase-Like Activity J. Nanosci. Nanotechnol. 2016 16 8 8846 8853Google Scholar118Synthesis of rhombic dodecahedral Fe3O4 single crystals towards their high peroxidase-like activityZhao, Jie; Zhang, Sha; Wang, Dan; Jia, Changchao; Yang, PingJournal of Nanoscience and Nanotechnology (2016), 16 (8), 8846-8853CODEN: JNNOAR; ISSN:1533-4880. (American Scientific Publishers)A solvothermal method has been developed to synthesize rhombic dodecahedral (RD) Fe3O4 single crystal structures bound by high-energy active {110} facets from inorg. Fe salts and N2H2 in N,N-dimethylformamide soln. The growth process and formation mechanism of the RD Fe3O4 structures was investigated in detail by changing different parameters. The as-synthesized products exhibit high magnetic properties with satn. magnetization value of 80 emu/g and coercive force value of 57 Oe. The RD Fe3O4 structures possessing active basal facets showed excellent peroxidase-like catalytic activity towards oxidn. of methylene blue (MB) dye in the presence of H2O2. After deposition of Au nanoparticles on the RD Fe3O4 structures, the Fe3O4/Au composites exhibit enhanced catalytic activity towards MB in the absence of H2O2. The Fe3O4 and Fe3O4/Au composites can be easily recycled from the treated water using a magnet. There was no obvious decrease of the peroxidase-like catalytic activity of the RD Fe3O4 structures after being used even for six cycles.
- 119Wan D. Li W. Wang G. Wei X. Shape-Controllable Synthesis of Peroxidase-Like Fe3O4 Nanoparticles for Catalytic Removal of Organic Pollutants J. Mater. Eng. Perform. 2016 25 10 4333 4340Google Scholar119Shape-Controllable Synthesis of Peroxidase-Like Fe3O4 Nanoparticles for Catalytic Removal of Organic PollutantsWan, Dong; Li, Wenbing; Wang, Guanghua; Wei, XiaobiJournal of Materials Engineering and Performance (2016), 25 (10), 4333-4340CODEN: JMEPEG; ISSN:1059-9495. (Springer)The shape of Fe3O4 nanoparticles is controlled using a simple oxidn.-pptn. method without any surfactant. The morphol. and structure of the obtained Fe3O4 nanoparticles were characterized by using x-ray diffraction, SEM, XPS, N2 physisorption and vibrating sample magnetometer. As-prepd. Fe3O4 samples showed octahedron, cube, hexagonal plate and sphere morphologies. Peroxidase-like activity of the four nanostructures was evaluated for catalytic removal of org. pollutants in the presence of H2O2, using rhodamine B as a model compd. The results showed that the H2O2-activating ability of the Fe3O4 nanocrystals was structure dependent and followed the order sphere > cube > octahedron > hexagonal plate, which was closely related to their surface FeII/FeIII ratios or crystal planes. The reusability of Fe3O4 spheres was also investigated after five successive runs, which demonstrated the promising application of the catalyst in the degrdn. of org. pollutants. This investigation is of great significance for the heterogeneous catalysts with enhanced activity and practical application.
- 120Gao Y. Wei Z. Li F. Yang Z. M. Chen Y. M. Zrinyi M. Osada Y. Synthesis of a morphology controllable Fe3O4 nanoparticle/hydrogel magnetic nanocomposite inspired by magnetotactic bacteria and its application in H2O2 detection Green Chem. 2014 16 3 1255 1261Google Scholar120Synthesis of a morphology controllable Fe3O4 nanoparticle/hydrogel magnetic nanocomposite inspired by magnetotactic bacteria and its application in H2O2 detectionGao, Yang; Wei, Zhao; Li, Fei; Yang, Zhi Mao; Chen, Yong Mei; Zrinyi, Miklos; Osada, YoshihitoGreen Chemistry (2014), 16 (3), 1255-1261CODEN: GRCHFJ; ISSN:1463-9262. (Royal Society of Chemistry)Owing to the shape-dependent catalytic activity of Fe3O4 nanoparticles, controlling their morphol. is of great significance. In this work, we propose a simple, nontoxic, water-based strategy for the fabrication of magnetic nanoparticle/hydrogel nanocomposites in which highly cryst. Fe3O4 nanooctahedra can be fabricated in situ within a neg. charged hydrogel matrix. The morphol. of the Fe3O4 nanocrystals can be easily tuned by adjusting the crosslinking concn. of the hydrogel. Furthermore, the catalytic activities of the magnetic nanocomposites were studied, and the magnetic nanocomposite loaded with Fe3O4 nanooctahedra exhibited excellent catalytic activity and provided a sensitive response for H2O2 detection. This scalable approach for the fabrication of magnetic nanocomposites, loaded with morphol. controllable Fe3O4 nanoparticles, potentially promotes their applications in biotechnol. and environmental chem.
- 121Peng Y. Wang Z. Liu W. Zhang H. Zuo W. Tang H. Chen F. Wang B. Size-and shape-dependent peroxidase-like catalytic activity of MnFe2O4 Nanoparticles and their applications in highly efficient colorimetric detection of target cancer cells Dalton Trans. 2015 44 28 12871 12877Google Scholar121Size- and shape-dependent peroxidase-like catalytic activity of MnFe2O4 Nanoparticles and their applications in highly efficient colorimetric detection of target cancer cellsPeng, Yunhua; Wang, Zhiyi; Liu, Weisheng; Zhang, Haoli; Zuo, Wei; Tang, Huiang; Chen, Fengjuan; Wang, BaoduiDalton Transactions (2015), 44 (28), 12871-12877CODEN: DTARAF; ISSN:1477-9226. (Royal Society of Chemistry)The catalytic activity of nanocrystal catalysts depends strongly on their chem. compn., size, and shape. Herein, the authors report four different sizes and shapes of MnFe2O4 nanoparticles (NPs) prepd. by a hydrothermal procedure. In addn., the size- and shape-dependent peroxidase-like activity of these NPs was first explored using 3,3',5,5'-tetramethyl-benzidine and H2O2 as peroxidase substrates. The peroxidase-like activities of the MnFe2O4 NPs were size- and shape-dependent and followed the order of 4 nm (spherical) > 18 nm (plate-like) > 27 nm (near-cubic) > 16 nm (spherical); this order was closely related to their surface-to-vol. ratio and atom arrangements. Such a study is of great significance for peroxidase nanomimetics with enhanced activity and use. Furthermore, folic acid (FA)-conjugated MnFe2O4 NPs allow the detection of folate receptor-rich cancer cells. Such study can be widely used for the identification of important target mols.
- 122Mao J. Hou X. Wang X. Hu S. Xiang L. The cubic aggregated CoFe2O4 nanoparticle anode material for lithium ion battery with good performance Mater. Lett. 2015 161 652 655Google Scholar122The cubic aggregated CoFe2O4 nanoparticle anode material for lithium ion battery with good performanceMao, Junwei; Hou, Xianhua; Wang, Xinyu; Hu, Shejun; Xiang, LiangzhongMaterials Letters (2015), 161 (), 652-655CODEN: MLETDJ; ISSN:0167-577X. (Elsevier B.V.)Novel cubic aggregated CoFe2O4 nanoparticles are successfully synthesized by hydrothermal method. Electrochem. results show that the CoFe2O4 nanoparticles exhibit good cycling performance and excellent rate capability. The 1st discharge and charge capacities of these nanoparticles are 1672.8 mA h g-1 and 1309.1 mA h g-1, resp., with an initial coulombic efficiency of 78.3%. The electrode can retain a high capacity of 1133.5 mA h g-1 after 120 cycles at a c.d. of 100 mA g-1. Also, it could still maintain a reversible capacity of 679 mA h g-1 even at a high c.d. of 3.2 A g-1. The improved electrochem. performance can be ascribed to the hierarchical cubic aggregated structural with large sp. surface area and lots of interspaces between the particles, which not only can effectively increase the active surface area, but also can make better penetration of the electrolyte and accommodate the vol. expansion.
- 123Tian T. Ai L. Liu X. Li L. Li J. Jiang J. Synthesis of hierarchical FeWO4 architectures with {100}-faceted nanosheet assemblies as a robust biomimetic catalyst Ind. Eng. Chem. Res. 2015 54 4 1171 1178Google Scholar123Synthesis of Hierarchical FeWO4 Architectures with {100}-Faceted Nanosheet Assemblies as a Robust Biomimetic CatalystTian, Tian; Ai, Lunhong; Liu, Xiaomin; Li, Lili; Li, Jie; Jiang, JingIndustrial & Engineering Chemistry Research (2015), 54 (4), 1171-1178CODEN: IECRED; ISSN:0888-5885. (American Chemical Society)Surface structure is of special significance to the heterogeneous catalysis, because catalysis is primarily a surface phenomenon. Therefore, a comprehensive understanding of the relationship between the catalytic activity and surface structure is of great importance. Herein, hierarchical FeWO4 architectures assembled by {100}-faceted nanosheets have been successfully synthesized by a facile one-pot solvothermal method and employed as a novel biomimetic catalyst mimicking enzyme for the first time. Due to the large d. of terminal iron atoms per unit surface area on the {100} facet, the FeWO4 architectures exhibited excellent peroxidase-like activity for the catalytic oxidn. of peroxidase substrate 3,3',5,5'-tetramethylbenzidine (TMB) to produce a blue color reaction in the presence of H2O2. Using the FeWO4 architectures as peroxidase mimetics, a colorimetric sensor for H2O2 was developed, which provided good response toward H2O2 concn. over a range from 0.95 to 66.7 μM with a detection limit of 0.28 μM. The proposed colorimetric method can be further extended for the sensitive detection of glucose by coupling with glucose oxidase (GOx). The limit of detection for glucose was detd. to be as low as 0.67 μM and the linear range was from 4 to 60 μM. It is believed that these findings are useful in understanding the peroxidase-like activity of catalysts with facet exposure and suggest a new strategy to pursue for highly active peroxidase-like catalysts.
- 124Liu B. Liu J. Surface modification of nanozymes Nano Res. 2017 10 4 1125 1148Google Scholar124Surface modification of nanozymesLiu, Biwu; Liu, JuewenNano Research (2017), 10 (4), 1125-1148CODEN: NRAEB5; ISSN:1998-0000. (Springer GmbH)Nanoparticles and proteins are similar in a no. of aspects, and using nanoparticles to mimic the catalytic function of enzymes is an interesting yet challenging task. Impressive developments have been made over the past two decades on this front. The term nanozyme was coined to refer to nanoparticlebased enzyme mimics. To date, many different types of nanozymes have been reported to catalyze a broad range of reactions for chem., anal., and biomedical applications. Since chem. reactions happen mainly on the surface of nanozymes, an interesting aspect for investigation is surface modification. In this review, we summarize three types of nanozyme materials catalyzing various reactions with a focus on their surface chem. For metal oxides, cerium oxide and iron oxide are discussed as they are the most extensively studied. Then, gold nanoparticles and graphene oxide are reviewed to represent metallic and carbon nanomaterials, resp. Types of modifications include ions, small mols., and polymers mainly by physisorption, while in a few cases, covalent modifications were also employed. The functional aspect of such modification is to improve catalytic activity, substrate specificity, and stability. Future perspectives of this field are speculated at the end of this review. [Figure not available: see fulltext.].
- 125Bruce I. J. Taylor J. Todd M. Davies M. J. Borioni E. Sangregorio C. Sen T. Synthesis, characterisation and application of silica-magnetite nanocomposites J. Magn. Magn. Mater. 2004 284 145 160Google Scholar125Synthesis, characterization and application of silica-magnetite nanocompositesBruce, Ian J.; Taylor, James; Todd, Michael; Davies, Martin J.; Borioni, Enrico; Sangregorio, Claudio; Sen, TapasJournal of Magnetism and Magnetic Materials (2004), 284 (), 145-160CODEN: JMMMDC; ISSN:0304-8853. (Elsevier B.V.)Silica-magnetite composites were prepd. for eventual applications in biomol. sepns. (nucleic acids). Their prodn. on large scale has been optimized and they have been extensively characterized in a phys. and chem. context. They perform at least as well, if not better than a com. available equiv. at adsorbing and eluting DNA. Several methods for the prepn. of magnetite were compared in order to select one, which produced particles, possessing high magnetic susceptibility, low rate of sedimentation and good chem. stability. Of the main methods studied: (i) oxidative hydrolysis of iron(II) sulfate in alk. media, (ii) alk. hydrolysis of iron(II) and iron(III) chloride solns., and (iii) pptn. from iron(II) and iron(III) chloride solns. by hydrolysis of urea, method (i) produced the best' magnetite particles. Silica-magnetite composites were prepd. using the best' magnetite, and, for comparison, two methods for depositing silica were used to coat the silica onto magnetite nanoparticles, from silicic acid at pH 10 and by acid hydrolysis of tetraethoxysilane (TEOS) at 90°. The best method for yielding silica-magnetite composites that worked well in DNA adsorption and elution proved to be that involving silicic acid and this material could be made in 20 g batch sizes. Silica-magnetite composites from the two methods proved to have distinct and different phys. and chem. properties. All magnetite and silica-magnetite samples were fully characterized for their relative chem. compn. using Fourier-transform IR, XRF and thermo-gravimetric anal. Their phys. characteristics were detd. using SEM and N2 adsorption and Mossbauer spectroscopy was used to confirm the identity of the iron oxides produced. Selected samples were comparatively tested for their ability to adsorb, and subsequently elute, 2-deoxyguanosine-5-monophosphate (GMP) and its non-phosphorylated analog 2-deoxyguanosine (G) and a range of sequence defined oligonucleotides (NAs) and sheared salmon sperm DNA. It was found that magnetite readily adsorbed GMP via the GMP phosphate anion in water, whereas silica did not, due to electrostatic repulsion between the neg. charged surface of silica and the GMP. Both magnetite and silica magnetite were further tested in adsorption studies of G and GMP in different chaotropic media, 4 M sodium chloride or 4 M ammonium sulfate. The high salt conditions aided binding of GMP silica magnetite but inhibited adsorption to magnetite presumably due to competition for binding sites on the magnetite's surface by the chaotrope anions. Interestingly, the results from NAs binding studies indicated that sequence appeared to play an important role in adsorption of the different species to silica-magnetite composites. This may indicate a contribution by hydrophobic interactions to the binding mechanism. Multiple depositions of silica onto magnetite performed by deposition from silicic acid at pH 10 did not appear to greatly increase the composite percentage represented by silica while composite produced by the acid hydrolysis of TEOS at 90° did. However, it appeared that the silica deposited by the first method represented a complete coating of the magnetite core while the second method yielded a porous or incomplete coating. In comparison with com. available silica-magnetite composite in DNA adsorption and elution, the material was obsd. to perform approx. 10% more efficiently. These findings indicate that it is possible to produce a consistent and cheap silica-magnetite nanoparticle on relatively large scale (greater than 20 g batch size) which is at least as good as, if not better than, a com. available alternative.
- 126Sun Y. Duan L. Guo Z. DuanMu Y. Ma M. Xu L. Zhang Y. Gu N. An improved way to prepare superparamagnetic magnetite-silica core-shell nanoparticles for possible biological application J. Magn. Magn. Mater. 2005 285 1-2 65 70Google ScholarThere is no corresponding record for this reference.
- 127Zhang X.-Q. Gong S.-W. Zhang Y. Yang T. Wang C.-Y. Gu N. Prussian blue modified iron oxide magnetic nanoparticles and their high peroxidase-like activity J. Mater. Chem. 2010 20 24 5110 5116Google Scholar127Prussian blue modified iron oxide magnetic nanoparticles and their high peroxidase-like activityZhang, Xiao-Qing; Gong, Shang-Wenyan; Zhang, Yu; Yang, Ting; Wang, Chun-Yu; Gu, NingJournal of Materials Chemistry (2010), 20 (24), 5110-5116CODEN: JMACEP; ISSN:0959-9428. (Royal Society of Chemistry)Prussian blue (PB) modified γ-Fe2O3 magnetic nanoparticles (MNPs) featuring varying PB proportions were synthesized and characterized by TEM, FTIR, UV-vis, EDS, XRD and XPS. The magnetic properties and peroxidase-like catalytic activity of the synthesized nanoparticles were investigated. With increasing PB content, the magnetism could still maintain a high level. Peroxidase-like activity was enhanced as the PB proportion increased. Catalysis was found to follow Michaelis-Menten kinetics. The calcd. kinetic parameters exhibited strong affinity with substrates and high catalytic activity, which are three orders of magnitudes larger than that for magnetite nanoparticles of similar size. Based on the high activity, an enzyme immunoassay model was established: staphylococcal protein A (SPA) was conjugated onto the surface of the nanoparticles to construct a new nanoprobe which was employed to detect IgG immobilized to 96-well plates. The results presented a linear absorbance enhancement with concn. of IgG, suggesting that PBMNPs serve as an inexpensive horseradish peroxidase (HRP) mimic enzyme with potential applications in bio-detection.
- 128Garg D. Kaur M. Sharma S. Verma V. Effect of CTAB coating on structural, magnetic and peroxidase mimic activity of ferric oxide nanoparticles Bull. Mater. Sci. 2018 41 5 1 9Google Scholar128Effect of CTAB coating on structural, magnetic and peroxidase mimic activity of ferric oxide nanoparticlesGarg, Dikshit; Kaur, Manpreet; Sharma, Sucheta; Verma, VibhaBulletin of Materials Science (2018), 41 (5), 1-9CODEN: BUMSDW; ISSN:0250-4707. (Indian Academy of Sciences)In the present work, pristine and cetyl tri-Me ammonium bromide (CTAB)-coated ferric oxide nanoparticles (CTAB@Fe2O3NPs) were synthesized and studied as enzyme mimics. The wt./wt. ratio of Fe2O3 to CTAB was varied as 1:1 and 1:2. Transmission electron microscopic anal. revealed that pristine NPs had an av. size of 50 nm, whereas the presence of CTAB resulted in the formation of nanorods with length of 130 nm. BET studies confirmed enhancement of surface area on CTAB coating, which was max. for wt./wt. ratio 1:1. The synthesized pristine NPs and CTAB-coated NPs were evaluated for their peroxidase mimic activity using o-dianisidine dihydrochloride as substrate. Optimum pH, temp., substrate and NPs concn. for the reaction were 1, 25oC, 0.16mgml-1 and 1mgml-1, resp. Peroxidase mimic activity of CTAB@Fe2O3NPs (wt./wt. 1:1) was higher than that of pristine NPs. However, further increase in CTAB coating (wt./wt. 1:2) resulted in lowering of peroxidase mimic activity. Kinetic anal. was carried out at optimized conditions; max. velocity (Vmax) and Michaelis const. (Km) value of CTAB@Fe2O3NPs at 1:1 wt./wt. ratio were 7.69 mM and 1.12μmols-1, resp.
- 129Fan Y. Shi W. Zhang X. Huang Y. Mesoporous material-based manipulation of the enzyme-like activity of CoFe2O4 nanoparticles J. Mater. Chem. A 2014 2 8 2482 2486Google Scholar129Mesoporous material-based manipulation of the enzyme-like activity of CoFe2O4 nanoparticlesFan, Yingwei; Shi, Wenbing; Zhang, Xiaodan; Huang, YumingJournal of Materials Chemistry A: Materials for Energy and Sustainability (2014), 2 (8), 2482-2486CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Mesoporous material supported CoFe2O4 magnetic nanoparticles possess unique peroxidase/oxidase-like activity, and react with luminol to yield a novel chemiluminescence without the need of H2O2. Their oxidase-like activity shows pH and support dependence, and could be reversibly controlled by their pH. This offers a new method for manipulating the enzyme-like activity of nanoparticles.
- 130Liu C.-H. Yu C.-J. Tseng W.-L. Fluorescence assay of catecholamines based on the inhibition of peroxidase-like activity of magnetite nanoparticles Anal. Chim. Acta 2012 745 143 148Google Scholar130Fluorescence assay of catecholamines based on the inhibition of peroxidase-like activity of magnetite nanoparticlesLiu, Cheng-Hao; Yu, Cheng-Ju; Tseng, Wei-LungAnalytica Chimica Acta (2012), 745 (), 143-148CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)We report a fluorescence approach for the highly selective and sensitive detection of catecholamines using magnetite nanoparticles (Fe3O4 NPs) in the presence of Amplex UltraRed (AUR) and H2O2. Fe3O4 NPs catalyze H2O2-mediated oxidn. of AUR. The resulting product fluoresces (excitation/emission maxima, ca. 568/587 nm) more strongly, relative to AUR. When catecholamines bind to Fe3O4, the complexes that are formed induce decreased activity of Fe3O4 NPs, mediated through the coordination between Fe3+ on the NP surface and the catechol moiety of catecholamines. As a result, Fe3O4 NPs-catalyzed H2O2-mediated oxidn. of AUR is inhibited by catecholamines. The limits of detection for dopamine (DA), L-DOPA, norepinephrine, and epinephrine were 3 nM, 3 nM, 3 nM, and 6 nM, resp. The Fe3O4 NPs-H2O2-AUR probe exhibited high selectivity (>1000-fold) toward catecholamines over other tested biomols. that commonly exist in urine. Four catecholamines had similar sensitivity because the inhibition of the Fe3O4 NPs activity relies on the presence of the catechol moiety. This approach also allowed the detn. of tyrosinase activity because tyrosinase catalyzes the conversion of L-tyrosine to L-DOPA. We validated the practicality of the use of the Fe3O4 NPs-H2O2-AUR probe for the detn. of the concns. of DA in urine samples.
- 131Chen C. Lu L. Zheng Y. Zhao D. Yang F. Yang X. A new colorimetric protocol for selective detection of phosphate based on the inhibition of peroxidase-like activity of magnetite nanoparticles Anal. Methods 2015 7 1 161 167Google Scholar131A new colorimetric protocol for selective detection of phosphate based on the inhibition of peroxidase-like activity of magnetite nanoparticlesChen, Chuanxia; Lu, Lixia; Zheng, Yu; Zhao, Dan; Yang, Fan; Yang, XiurongAnalytical Methods (2015), 7 (1), 161-167CODEN: AMNEGX; ISSN:1759-9679. (Royal Society of Chemistry)A simple colorimetric assay for phosphate ion (Pi) has been established based on analyte-induced inhibition of the magnetite nanoparticles (MNPs)-catalyzed oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The Fe3O4 MNPs can catalyze the H2O2-mediated oxidn. of TMB and yields a blue oxidized product, which exhibits a max. absorption at 652 nm. Pi could be absorbed on the surface of the Fe3O4 MNPs through coordinating with Fe3+, inducing a reduced colorimetric signal. The colorimetric signal change (ΔA652) in this process was proportional to the concn. of Pi, ranging from 0.2 μM to 200 μM. The limit of detection (S/N = 3) was as low as 0.11 μM. The as-proposed Fe3O4 MNPs-TMB-H2O2 probe exhibited a high selectivity toward Pi over other relevant ions that commonly exist in water and has been applied to Pi detection in drinking water, ground water and lake water samples with satisfactory results.
- 132Park K. S. Kim M. I. Cho D. Y. Park H. G. Label‐free colorimetric detection of nucleic acids based on target‐induced shielding against the peroxidase‐mimicking activity of magnetic nanoparticles Small 2011 7 11 1521 1525Google Scholar132Label-Free Colorimetric Detection of Nucleic Acids Based on Target-Induced Shielding Against the Peroxidase-Mimicking Activity of Magnetic NanoparticlesPark, Ki Soo; Kim, Moon Il; Cho, Dae-Yeon; Park, Hyun GyuSmall (2011), 7 (11), 1521-1525CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)A new and simple colorimetric methodol. for the detection of nucleic acids has been developed, which relies on target-DNA-induced inhibition of the peroxidase-mimicking activity of MNPs. This is the first example in which the peroxidase activity of MNPs serves as the basis for colorimetric detection of nucleic acids. Significant features of the new assay system are its simplicity and cost-effectiveness. Importantly, assays can be accomplished using naked-eye detection within a 30 min period. Furthermore, the new assay system does not require surface modification of either the MNPs or DNA, and a salt-induced aggregation step is not needed: these are time-consuming and costly manipulations that are generally required in AuNP-based DNA detection procedures. An application of this simple yet efficient colorimetric detection system was successfully demonstrated by its use in diagnosing C. trachomatis using a human urine sample.
- 133Li X. Wen F. Creran B. Jeong Y. Zhang X. Rotello V. M. Colorimetric protein sensing using catalytically amplified sensor arrays Small 2012 8 23 3589 3592Google Scholar133Colorimetric protein sensing using catalytically amplified sensor arraysLi, Xiaoning; Wen, Fang; Creran, Brian; Jeong, Youngdo; Zhang, Xinrong; Rotello, Vincent M.Small (2012), 8 (23), 3589-3592CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)Dopamine- or trimethylammonium- functionalized Fe3O4 nanoparticles were fabricated and used for array-based sensing of ten proteins.
- 134Le T. N. Tran T. D. Kim M. I. A Convenient Colorimetric Bacteria Detection Method Utilizing Chitosan-Coated Magnetic Nanoparticles Nanomaterials 2020 10 1 92Google Scholar134A convenient colorimetric bacteria detection method utilizing chitosan-coated magnetic nanoparticlesLe, Thao Nguyen; Tran, Tai Duc; Kim, Moon IlNanomaterials (2020), 10 (1), 92CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)An effective novel strategy to detect bacteria is promising because it may improve human health by allowing early diagnosis and timely treatment of bacterial infections. Here, we report a simple, reliable, and economical colorimetric assay using the peroxidase-like activity of chitosan-coated iron oxide magnetic nanoparticles (CS-MNPs). When CS-MNPs are incubated with a sample contg. bacterial cells such as the gram-neg. Escherichia coli or the gram-pos. Staphylococcus aureus, the neg.-charged bacterial membrane interacts with pos.-charged chitosan on the surface of CS-MNPs, thus resulting in significant redn. of their peroxidase-like activity presumably by a hindrance in the accessibility of the neg. charged substrate, 2-2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) to the pos.-charged CS-MNPs. This simple colorimetric strategy allowed the rapid detection of bacterial cells down to 104 CFU mL-1 by the naked eye and 102 CFU mL-1 by spectrophotometry within 10 min. Based on the results, we anticipate that the CS-MNPs-based assay has great potential for the on-site diagnosis of bacterial infections in facility-limited or point-of-care testing (POCT) environments.
- 135Huang X. Xu C. Ma J. Chen F. Ionothermal synthesis of Cu-doped Fe3O4 magnetic nanoparticles with enhanced peroxidase-like activity for organic wastewater treatment Adv. Powder Technol. 2018 29 3 796 803Google Scholar135Ionothermal synthesis of Cu-doped Fe3O4 magnetic nanoparticles with enhanced peroxidase-like activity for organic wastewater treatmentHuang, Xuanlin; Xu, Cong; Ma, Jiping; Chen, FengxiAdvanced Powder Technology (2018), 29 (3), 796-803CODEN: APTEEE; ISSN:0921-8831. (Elsevier B.V.)The intrinsic peroxidase-like activity of magnetite magnetic nanoparticles (Fe3O4 MNPs) has to be improved to activate H2O2 under mild conditions for practical applications. Herein copper-doped Fe3O4 (Fe3-xCuxO4, x: 0.06-0.23) MNPs were successfully prepd. by oxidative pptn.-combined ionothermal synthesis and characterized by XRD, VSM, XPS, BET, etc. The Cu2+ dopants are mainly substituted for Fe2+ ions at octahedral sites and significantly surface-enriched, which expedite the Fe3+/Fe2+ redox cycling and enhance the H2O2-activation ability of Fe3-xCuxO4 MNPs. Kinetic study showed that the decompn. of H2O2 on Fe2.88Cu0.12O4 was much faster than that on the undoped Fe3O4 (0.584 vs. 0.153 h-1 at 25 °C) due to the lower activation energy of the former (55.3 vs. 62.1 kJ/mol). The enhanced H2O2-activation ability upon copper doping was exploited to efficiently degrade recalcitrant org. pollutants (e.g., rhodamine B) with H2O2 at pH ∼ 7 and 25 °C on Fe2.88Cu0.12O4 with good stability and reusability (16 h tested in eight cycles).
- 136Moreno Maldonado A. C. Winkler E. L. Raineri M. Toro Córdova A. Rodríguez L. M. Troiani H. E. Mojica Pisciotti M. L. Mansilla M. V. Tobia D. Nadal M. S. Torres T. E. De Biasi E. Ramos C. A. Goya G. F. Zysler R. D. Lima E. Free-Radical Formation by the Peroxidase-Like Catalytic Activity of MFe2O4 (M = Fe, Ni, and Mn) Nanoparticles J. Phys. Chem. C 2019 123 33 20617 20627Google Scholar136Free-Radical Formation by the Peroxidase-Like Catalytic Activity of MFe2O4 (M = Fe, Ni, and Mn) NanoparticlesMoreno Maldonado, Ana Carolina; Winkler, Elin L.; Raineri, Mariana; Toro Cordova, Alfonso; Rodriguez, Luis M.; Troiani, Horacio E.; Mojica Pisciotti, Mary Luz; Mansilla, Marcelo Vasquez; Tobia, Dina; Nadal, Marcela S.; Torres, Teobaldo E.; De Biasi, Emilio; Ramos, Carlos A.; Goya, Gerardo F.; Zysler, Roberto D.; Lima, EnioJournal of Physical Chemistry C (2019), 123 (33), 20617-20627CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Ferrite magnetic nanoparticles (MNPs) have peroxidase-like activity and thus catalyze the decompn. of H2O2-producing reactive oxygen species (ROS). Increasingly important applications of these ferrite MNPs in biol. and medicine require that their morphol., physicochem., and magnetic properties need to be strictly controlled. Usually, the tuning of their magnetic properties is achieved by the replacement of Fe by other 3d metals, such as Mn or Ni. Here, we studied the catalytic activity of ferrite MNPs (MFe2O4, M = Fe2+/Fe3+, Ni, and Mn) with the mean diam. ranging from 10 to 12 nm. Peroxidase-like activity was studied by ESR (EPR) using the spin-trap 5,5-dimethyl-1-pyrroline N-oxide at different pHs (4.8 and 7.4) and temps. (25 and 40 °C). We identified an enhanced amt. of hydroxyl (•OH) and perhydroxyl (•OOH) radicals for all samples, compared to a blank soln. Quant. studies show that [•OH] is the dominant radical formed for Fe3O4, which is strongly reduced with the concomitant oxidn. of Fe2+ or its substitution (Ni or Mn). A comparative anal. of the EPR data against in vitro prodn. of ROS in microglial BV2 cell culture provided addnl. insights regarding the catalytic activity of ferrite MNPs, which should be considered if biomedical uses are intended. Our results contribute to a better understanding of the role played by different divalent ions in the catalytic activity of ferrite nanoparticles, which is very important because of their use in biomedical applications.
- 137Verma V. Kaur M. Sharma S. Superoxide dismutase mimic activity of spinel ferrite MFe2O4 (M= Mn, Co and Cu) nanoparticles Bull. Mater. Sci. 2019 42 3 1 6Google Scholar137Effect of vanadyl doping on relative intensities of factor group split Raman bands in potassium oxalate monohydratePant, Navin; Verma, A. L.; Pandey, S. D.Bulletin of Materials Science (2019), 42 (4), 1-7CODEN: BUMSDW; ISSN:0250-4707. (Indian Academy of Sciences)We report Raman spectral studies of pure potassium oxalate monohydrate, K2C2O4·H2O (POM) and its doped varieties with 1 and 5% vanadyl concns. at room temp. Apart from many other interesting changes in the Raman spectra on doping with VO2+, one of the most intriguing observations is the change in relative intensities of factor group split components of several Raman bands including some bands in the lattice mode region. We have explained the relative intensity changes of factor group split components in terms of locking of adjacent C2O4 - H2O - C2O4 - H2O chains due to vanadyl doping in the POM lattice. The redn. in the intensities of water bands on vanadyl doping also supports this view.
- 138Zhang C. Wang X. Dong X. Mei L. Wu X. Gu Z. Zhao Y. X-ray-facilitated redox cycling of nanozyme possessing peroxidase-mimicking activity for reactive oxygen species-enhanced cancer therapy Biomaterials 2021 276 121023Google Scholar138X-ray-facilitated redox cycling of nanozyme possessing peroxidase-mimicking activity for reactive oxygen species-enhanced cancer therapyZhang, Chenyang; Wang, Xin; Dong, Xinghua; Mei, Linqiang; Wu, Xiaochen; Gu, Zhanjun; Zhao, YuliangBiomaterials (2021), 276 (), 121023CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)Nanomaterials with shifting or mixed redox states is one of the most common studied nanozyme with peroxidase-like activity for chemodynamic therapy (CDT), which can decomp. hydrogen peroxide (H2O2) of tumor microenvironment into highly toxic reactive oxygen species (ROS) by a nano-catalytic way. However, most of them exhibit an insufficient catalytic efficiency due to their dependence on catalytic condition. Herein, a potential methodol. is proposed to enhance their enzymic activity by accelerating the redox cycling of these nanomaterials with shifting or mixed redox states in the presence of X-ray. In this study, the nanocomposite consisting of SnS2 nanoplates and Fe3O4 quantum dots with shifting or mixed redox states (Fe2+/Fe3+) is used to explore the strategy. Under external X-ray irradn., SnS2 cofactor as electron donor can be triggered to transfer electrons to Fe3O4, which promotes the regeneration of Fe2+ sites on the surface of the Fe3O4. Consequently, the regenerated Fe2+ sites react with the overexpressed H2O2 to persistently generate ROS for enhanced tumor therapy. The designed nanocomposite displays the synergistic effects of radiotherapy and CDT. The strategy provides a new avenue for the development of artificial nanozymes with shifting or mixed redox states in precise cancer treatments based on X-ray-enhanced enzymic efficacy.
- 139Zhu M. Dai Y. Wu Y. Liu K. Qi X. Sun Y. Bandgap control of α-Fe2O3 nanozymes and their superior visible light promoted peroxidase-like catalytic activity Nanotechnology 2018 29 46 465704Google Scholar139Bandgap control of α-Fe2O3 nanozymes and their superior visible light promoted peroxidase-like catalytic activityZhu, Mingyun; Dai, Yunqian; Wu, Yanan; Liu, Ken; Qi, Xiaomian; Sun, YuemingNanotechnology (2018), 29 (46), 465704/1-465704/10CODEN: NNOTER; ISSN:1361-6528. (IOP Publishing Ltd.)Iron oxide nanoparticles (NPs) possessing peroxidase-like catalytic activity have been widely explored in recent decades, owing to their high stability against harsh conditions, low cost, flexibility in structure design and compn., adjustable activities and excellent biocompatibility in comparison with natural enzymes. Recently, a lot of great achievements have been made in this field of iron oxide nanozymes, however, this research has now reached a bottleneck in that the max. activity enhancement is difficult to achieve via a material design. Hence, in this work, visible light was introduced to improve the peroxidase-like activity of Fe2O3 NPs synthesized via a combination of electrospinning technol. and hydrothermal reaction. Our results showed that with the assistance of visible light, Fe2O3 NPs exhibited at least 2.2-fold higher peroxidase activity than those tested under darkness, confirming the superiorly visible light promoted peroxidase-like catalytic activity of Fe2O3 NPs. Furthermore, the affinity and max. reaction velocity of Fe2O3 nanoflowers (bandgap,,Q=,,Q1.78 eV) towards 3,3',5,5'- tetramethylbenanozymeidine (TMB) were at least over 3.7 and 4.3 times greater than in Fe2O3 nanocubes (bandgap = 2.08 eV), suggesting that the reaction performance of semiconductors could be controlled by proper adjustment of the bandgap. Moreover, the Fe2O3 NPs were also successfully utilized to detect glucose. Herein, we believe that the present work exhibits a fascinating perspective for peroxidase-like catalytic fields.
- 140Zhang Y. Wang X. Chu C. Zhou Z. Chen B. Pang X. Lin G. Lin H. Guo Y. Ren E. Lv P. Shi Y. Zheng Q. Yan X. Chen X. Liu G. Genetically engineered magnetic nanocages for cancer magneto-catalytic theranostics Nat. Commun. 2020 11 1 5421Google Scholar140Genetically engineered magnetic nanocages for cancer magneto-catalytic theranosticsZhang Yang; Wang Xiaoyong; Chu Chengchao; Pang Xin; Lin Gan; Lin Huirong; Ren En; Lv Peng; Shi Yesi; Zheng Qingbing; Yan Xiaohui; Liu Gang; Wang Xiaoyong; Zheng Qingbing; Liu Gang; Zhou Zijian; Chen Xiaoyuan; Chen Biaoqi; Guo YuxinNature communications (2020), 11 (1), 5421 ISSN:.The clinical applications of magnetic hyperthermia therapy (MHT) have been largely hindered by the poor magnetic-to-thermal conversion efficiency of MHT agents. Herein, we develop a facile and efficient strategy for engineering encapsulin-produced magnetic iron oxide nanocomposites (eMIONs) via a green biomineralization procedure. We demonstrate that eMIONs have excellent magnetic saturation and remnant magnetization properties, featuring superior magnetic-to-thermal conversion efficiency with an ultrahigh specific absorption rate of 2390 W/g to overcome the critical issues of MHT. We also show that eMIONs act as a nanozyme and have enhanced catalase-like activity in the presence of an alternative magnetic field, leading to tumor angiogenesis inhibition with a corresponding sharp decrease in the expression of HIF-1α. The inherent excellent magnetic-heat capability, coupled with catalysis-triggered tumor suppression, allows eMIONs to provide an MRI-guided magneto-catalytic combination therapy, which may open up a new avenue for bench-to-bed translational research of MHT.
Scheme 1
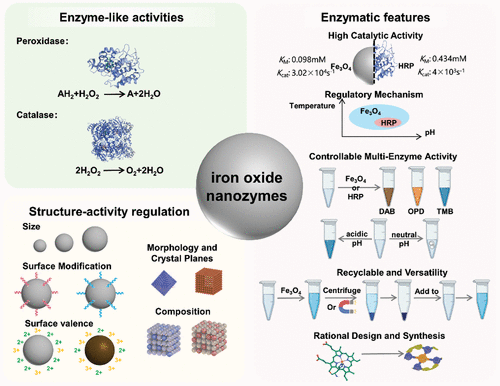 Scheme 1. Graphic abstract of the chapter. In this chapter, the enzymatic activity and catalytic characteristics of iron oxide nanozymes were introduced, and the structure-activity mechanism was reviewed.
Scheme 1. Graphic abstract of the chapter. In this chapter, the enzymatic activity and catalytic characteristics of iron oxide nanozymes were introduced, and the structure-activity mechanism was reviewed.Figure 1
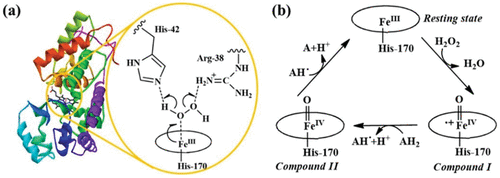 Figure 1. Schematic illustration of (a) the reaction pocket of HRP and (b) the corresponding catalytic cycle (AH2 is a two-electron donor substrate).Reproduced with permission from reference ( 28). Copyright 2021 American Chemical Society.
Figure 1. Schematic illustration of (a) the reaction pocket of HRP and (b) the corresponding catalytic cycle (AH2 is a two-electron donor substrate).Reproduced with permission from reference ( 28). Copyright 2021 American Chemical Society.Figure 2
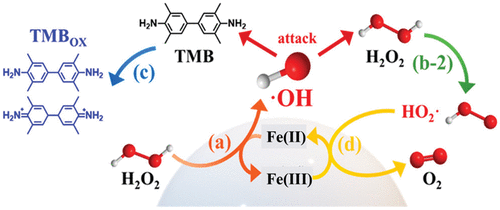 Figure 2. Schematic illustration of the origin of transition iron oxides as peroxidase mimetics.Reproduced with permission from reference ( 28). Copyright 2021 American Chemical Society.
Figure 2. Schematic illustration of the origin of transition iron oxides as peroxidase mimetics.Reproduced with permission from reference ( 28). Copyright 2021 American Chemical Society.Figure 3
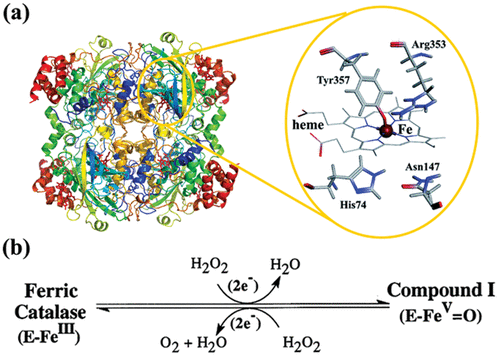 Figure 3. Schematic illustration of (a) the reaction pocket of catalase and (b) the corresponding catalytic cycle.
Figure 3. Schematic illustration of (a) the reaction pocket of catalase and (b) the corresponding catalytic cycle.Figure 4
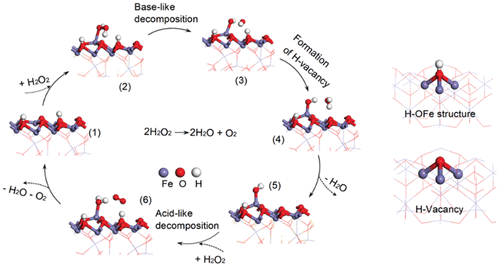 Figure 4. The corresponding catalytic cycle of ferrihydrite catalase-like nanozyme.Reproduced with permission from reference ( 46). Copyright 2021 Elsevier.
Figure 4. The corresponding catalytic cycle of ferrihydrite catalase-like nanozyme.Reproduced with permission from reference ( 46). Copyright 2021 Elsevier.Figure 5
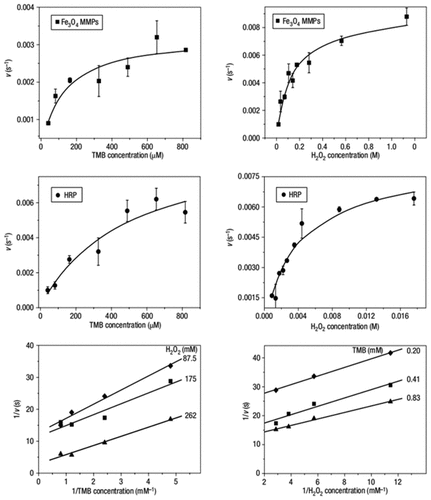 Figure 5. Steady-state kinetic assay and catalytic mechanism of Fe3O4 nanozymes(named as Fe3O4 MNPs) and HRP for different substrates.Reproduced with permission from reference ( 11). Copyright 2007 Springer Nature.
Figure 5. Steady-state kinetic assay and catalytic mechanism of Fe3O4 nanozymes(named as Fe3O4 MNPs) and HRP for different substrates.Reproduced with permission from reference ( 11). Copyright 2007 Springer Nature.Figure 6
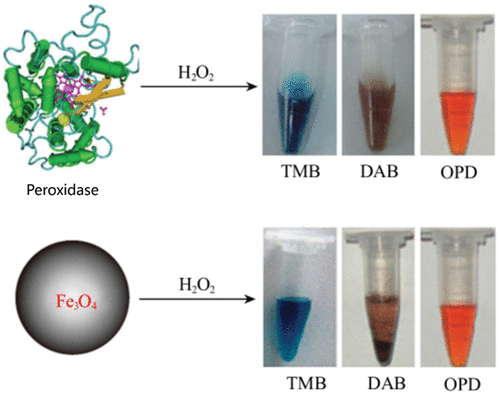 Figure 6. Fe3O4 nanozyme demonstrates peroxidase like activity by catalyzing H2O2 and substrates with color reaction.Adapted with permission from reference ( 11). Copyright 2007 Springer Nature.
Figure 6. Fe3O4 nanozyme demonstrates peroxidase like activity by catalyzing H2O2 and substrates with color reaction.Adapted with permission from reference ( 11). Copyright 2007 Springer Nature.Figure 7
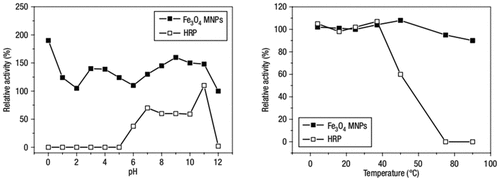 Figure 7. Comparison of the stability of Fe3O4 nanozymes and HRP at a range of values of pH from 0 to 12 and a range of temperatures between 4 and 90 °C.Reproduced with permission from reference ( 11). Copyright 2007 Springer Nature.
Figure 7. Comparison of the stability of Fe3O4 nanozymes and HRP at a range of values of pH from 0 to 12 and a range of temperatures between 4 and 90 °C.Reproduced with permission from reference ( 11). Copyright 2007 Springer Nature.Figure 8
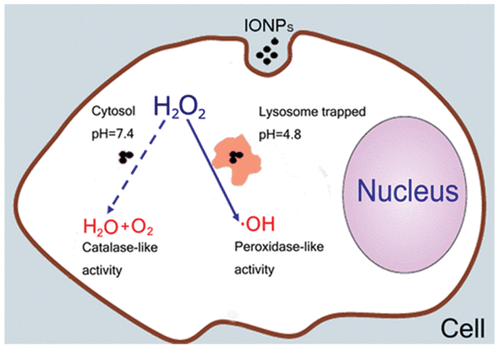 Figure 8. The enzyme activity of iron oxide nanozyme regulated by pH.IONPs: iron oxide nanoparticles.Reproduced with permission from reference ( 23). Copyright 2012 American Chemical Society.
Figure 8. The enzyme activity of iron oxide nanozyme regulated by pH.IONPs: iron oxide nanoparticles.Reproduced with permission from reference ( 23). Copyright 2012 American Chemical Society.Figure 9
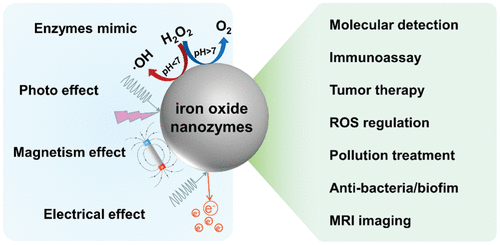 Figure 9. Properties and multi-functions of iron oxide nanozymes.
Figure 9. Properties and multi-functions of iron oxide nanozymes.Figure 10
 Figure 10. Rational design and synthesis of nanozymes.Reproduced with permission from reference ( 20). Copyright 2020 Elsevier.
Figure 10. Rational design and synthesis of nanozymes.Reproduced with permission from reference ( 20). Copyright 2020 Elsevier.Figure 11
 Figure 11. Typical SEM images of the obtained Fe3O4 nanoparticles of various sizes: (a) 30 nm, (b) 150 nm, (c) 300 nm. (d) Under the same conditions, smaller Fe3O4 MNPs show higher peroxidase-like activity.Adapted with permission from reference ( 11). Copyright 2007 Springer Nature.
Figure 11. Typical SEM images of the obtained Fe3O4 nanoparticles of various sizes: (a) 30 nm, (b) 150 nm, (c) 300 nm. (d) Under the same conditions, smaller Fe3O4 MNPs show higher peroxidase-like activity.Adapted with permission from reference ( 11). Copyright 2007 Springer Nature.Figure 12
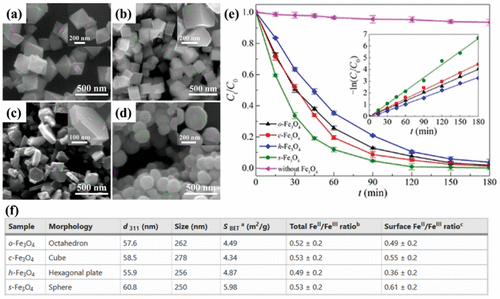 Figure 12. Typical SEM images of the obtained Fe3O4 nanoparticles of various morphologies: (a) octahedron, (b) cube, (c) hexagonal plate, and (d) sphere. (e) Degradation efficiency of RhB by different morphologies of Fe3O4. (f) Comparison of the structural characteristics of the Fe3O4 nanoparticles with various morphologies.Reproduced with permission from reference ( 119). Copyright 2016 Springer Nature.
Figure 12. Typical SEM images of the obtained Fe3O4 nanoparticles of various morphologies: (a) octahedron, (b) cube, (c) hexagonal plate, and (d) sphere. (e) Degradation efficiency of RhB by different morphologies of Fe3O4. (f) Comparison of the structural characteristics of the Fe3O4 nanoparticles with various morphologies.Reproduced with permission from reference ( 119). Copyright 2016 Springer Nature.Figure 13
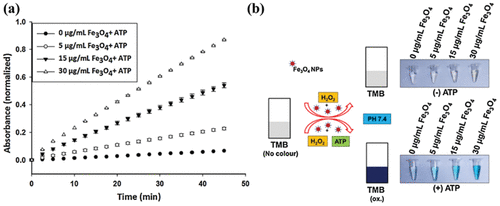 Figure 13. (a) Concentration-dependent kinetic study of the peroxidase-like activity of Fe3O4 nanozymes (5, 15 and 30 μg·mL−1) in presence of ATP followed for 45 min. (b) Schematic diagram showing the comparative peroxidase-like activity of Fe3O4 nanozymes at physiological pH in the absence (upper panel) and presence (lower panel) of ATP. Colour change in tubes indicates the oxidation of TMB (blue) by Fe3O4 nanozyme.Reproduced with permission from reference ( 62). Copyright 2017 Elsevier.
Figure 13. (a) Concentration-dependent kinetic study of the peroxidase-like activity of Fe3O4 nanozymes (5, 15 and 30 μg·mL−1) in presence of ATP followed for 45 min. (b) Schematic diagram showing the comparative peroxidase-like activity of Fe3O4 nanozymes at physiological pH in the absence (upper panel) and presence (lower panel) of ATP. Colour change in tubes indicates the oxidation of TMB (blue) by Fe3O4 nanozyme.Reproduced with permission from reference ( 62). Copyright 2017 Elsevier.References
CHAPTER SECTIONSThis chapter references 140 other publications.
- 1Schwertmann, U.; Cornell, R. M. Iron Oxides in the Laboratory: Preparation and Characterization; John Wiley & Sons, 2008.There is no corresponding record for this reference.
- 2Cornell, R. M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences, and Uses; Wiley-VCH Weinheim, 2003; Vol. 2.There is no corresponding record for this reference.
- 3Sivula K. Le Formal F. Grätzel M. Solar water splitting: progress using hematite (α‐Fe2O3) photoelectrodes ChemSusChem 2011 4 4 432 4493Solar Water Splitting: Progress Using Hematite (α-Fe2O3) PhotoelectrodesSivula, Kevin; Le Formal, Florian; Graetzel, MichaelChemSusChem (2011), 4 (4), 432-449CODEN: CHEMIZ; ISSN:1864-5631. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. Photoelectrochem. cells offer the ability to convert electromagnetic energy from our largest renewable source, the Sun, to stored chem. energy through the splitting of water into mol. oxygen and hydrogen. Hematite (α-Fe2O3) has emerged as a promising photoelectrode material due to its significant light absorption, chem. stability in aq. environments, and ample abundance. However, its performance as a water-oxidizing photoanode has been crucially limited by poor optoelectronic properties that lead to both low light harvesting efficiencies and a large requisite overpotential for photoassisted water oxidn. Recently, the application of nanostructuring techniques and advanced interfacial engineering has afforded landmark improvements in the performance of hematite photoanodes. In this review, new insights into the basic material properties, the attractive aspects, and the challenges in using hematite for photoelectrochem. water splitting are first examd. Next, recent progress enhancing the photocurrent by precise morphol. control and reducing the overpotential with surface treatments are critically detailed and compared. The latest efforts using advanced characterization techniques, particularly electrochem. impedance spectroscopy, are finally presented. These methods help to define the obstacles that remain to be surmounted in order to fully exploit the potential of this promising material for solar energy conversion.
- 4Reddy M. Yu T. Sow C.-H. Shen Z. X. Lim C. T. Subba Rao G. Chowdari B. α‐Fe2O3 nanoflakes as an anode material for Li‐ion batteries Adv. Funct. Mater. 2007 17 15 2792 27994α-Fe2O3 nanoflakes as an anode material for Li-ion batteriesReddy, M. V.; Yu, Ting; Sow, Chorng-Haur; Shen, Ze Xiang; Lim, Chwee Teck; Rao, G. V. Subba; Chowdari, B. V. R.Advanced Functional Materials (2007), 17 (15), 2792-2799CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)Nanoflakes of α-Fe2O3 were prepd. on Cu foil by using a thermal treatment method. The nanoflakes were characterized by X-ray diffraction, SEM, high-resoln. transmission electron microscopy, and Raman spectroscopy. The reversible Li-cycling properties of the α-Fe2O3 nanoflakes have been evaluated by cyclic voltammery, galvanostatic discharge-charge cycling, and impedance spectral measurements on cells with Li metal as the counter and ref. electrodes, at ambient temp. Results show that Fe2O3 nanoflakes exhibit a stable capacity of (680 ± 20) mA h g-1, corresponding to (4.05 ± 0.05) moles of Li per mol of Fe2O3 with no noticeable capacity fading up to 80 cycles when cycled in the voltage range 0.005-3.0 V at 65 mA g-1 (0.1 C rate), and with a coulombic efficiency of >98% during cycling (after the 15th cycle). The av. discharge and charge voltages are 1.2 and 2.1 V, resp. The obsd. cyclic voltammograms and impedance spectra have been analyzed and interpreted in terms of the 'conversion reaction' involving nanophase Fe0-Li2O. The superior performance of Fe2O3 nanoflakes is clearly established by a comparison of the results with those for Fe2O3 nanoparticles and nanotubes reported in the literature.
- 5Chen J. Xu L. Li W. Gou X. α‐Fe2O3 nanotubes in gas sensor and lithium‐ion battery applications Adv. Mater. 2005 17 5 582 5865α-Fe2O3 nanotubes in gas sensor and lithium-ion battery applicationsChen, Jun; Xu, Lina; Li, Weiyang; Gou, XinglongAdvanced Materials (Weinheim, Germany) (2005), 17 (5), 582-586CODEN: ADVMEW; ISSN:0935-9648. (Wiley-VCH Verlag GmbH & Co. KGaA)Templated arrays of α-Fe2O3 nanotubes, produced using an alumina membrane, exhibit enhanced sensitivity to ethanol and hydrogen as well as enhanced electrochem. activity in lithium-ion batteries. The nanotubes, characterized by uniform size and shape (see Figure), possess a high sp. surface area, leading to electrochem. activities much higher than that of bulk or nanoparticulate α-Fe2O3.
- 6Gupta A. K. Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications Biomaterials 2005 26 18 3995 40216Synthesis and surface engineering of iron oxide nanoparticles for biomedical applicationsGupta, Ajay Kumar; Gupta, MonaBiomaterials (2005), 26 (18), 3995-4021CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)A review. Superparamagnetic iron oxide nanoparticles (SPION) with appropriate surface chem. have been widely used exptl. for numerous in vivo applications such as magnetic resonance imaging contrast enhancement, tissue repair, immunoassay, detoxification of biol. fluids, hyperthermia, drug delivery and in cell sepn., etc. All these biomedical and bioengineering applications require that these nanoparticles have high magnetization values and size smaller than 100 nm with overall narrow particle size distribution, so that the particles have uniform phys. and chem. properties. In addn., these applications need special surface coating of the magnetic particles, which has to be not only non-toxic and biocompatible but also allow a targetable delivery with particle localization in a specific area. To this end, most work in this field has been done in improving the biocompatibility of the materials, but only a few scientific investigations and developments have been carried out in improving the quality of magnetic particles, their size distribution, their shape and surface in addn. to characterizing them to get a protocol for the quality control of these particles. Nature of surface coatings and their subsequent geometric arrangement on the nanoparticles det. not only the overall size of the colloid but also play a significant role in biokinetics and biodistribution of nanoparticles in the body. The types of specific coating, or derivatization, for these nanoparticles depend on the end application and should be chosen by keeping a particular application in mind, whether it be aimed at inflammation response or anti-cancer agents. Magnetic nanoparticles can bind to drugs, proteins, enzymes, antibodies, or nucleotides and can be directed to an organ, tissue, or tumor using an external magnetic field or can be heated in alternating magnetic fields for use in hyperthermia. This review discusses the synthetic chem., fluid stabilization and surface modification of superparamagnetic iron oxide nanoparticles, as well as their use for above biomedical applications.
- 7Weissleder R. Stark D. D. Engelstad B. L. Bacon B. R. Compton C. C. White D. L. Jacobs P. Lewis J. Superparamagnetic iron oxide: pharmacokinetics and toxicity Am. J. Roentgenol. 1989 152 1 167 1737Superparamagnetic iron oxide: pharmacokinetics and toxicityWeissleder R; Stark D D; Engelstad B L; Bacon B R; Compton C C; White D L; Jacobs P; Lewis JAJR. American journal of roentgenology (1989), 152 (1), 167-73 ISSN:0361-803X.The pharmacokinetics (distribution, metabolism, bioavailability, excretion) and toxicity (acute and subacute toxicity, mutagenicity) of a superparamagnetic iron oxide preparation (AMI-25), currently used in clinical trials, were evaluated by 59Fe radiotracer studies, measurements of relaxation times, the ability to reverse iron deficiency anemia, histologic examination, and laboratory parameters. One hour after administration of AMI-25 to rats (18 mumol Fe/kg; 1 mg Fe/kg), 82.6 +/- 0.3% of the administered dose was sequestered in the liver and 6.2 +/- 7.6% in the spleen. Peak concentrations of 59Fe were found in liver after 2 hr and in the spleen after 4 hr. 59Fe slowly cleared from liver (half-life, 3 days) and spleen (half-life, 4 days) and was incorporated into hemoglobin of erythrocytes in a time-dependent fashion. The half-time of the T2 effect on liver and spleen (24-48 hr) was shorter than the 59Fe clearance, indicating metabolism of AMI-25 into other forms of iron. IV administration of AMI-25 (30 mg Fe/kg) corrected iron-deficiency anemia and showed bioavailability similar to that of commercially available IV iron preparations within 7 days. No acute or subacute toxic effects were detected by histologic or serologic studies in rats or beagle dogs who received a total of 3000 mumol Fe/kg, 150 times the dose proposed for MR imaging of the liver. Our results indicate that AMI-25 is a fully biocompatible potential contrast agent for MR.
- 8Weinstein J. S. Varallyay C. G. Dosa E. Gahramanov S. Hamilton B. Rooney W. D. Muldoon L. L. Neuwelt E. A. Superparamagnetic iron oxide nanoparticles: diagnostic magnetic resonance imaging and potential therapeutic applications in neurooncology and central nervous system inflammatory pathologies, a review J. Cereb. Blood Flow Metab. 2010 30 1 15 358Superparamagnetic iron oxide nanoparticles: diagnostic magnetic resonance imaging and potential therapeutic applications in neurooncology and central nervous system inflammatory pathologies, a reviewWeinstein, Jason S.; Varallyay, Csanad G.; Dosa, Edit; Gahramanov, Seymur; Hamilton, Bronwyn; Rooney, William D.; Muldoon, Leslie L.; Neuwelt, Edward A.Journal of Cerebral Blood Flow & Metabolism (2010), 30 (1), 15-35CODEN: JCBMDN; ISSN:0271-678X. (Nature Publishing Group)A review. Superparamagnetic iron oxide nanoparticles have diverse diagnostic and potential therapeutic applications in the central nervous system (CNS). They are useful as magnetic resonance imaging (MRI) contrast agents to evaluate: areas of blood-brain barrier (BBB) dysfunction related to tumors and other neuroinflammatory pathologies, the cerebrovasculature using perfusion-weighted MRI sequences, and in vivo cellular tracking in CNS disease or injury. Novel, targeted, nanoparticle synthesis strategies will allow for a rapidly expanding range of applications in patients with brain tumors, cerebral ischemia or stroke, carotid atherosclerosis, multiple sclerosis, traumatic brain injury, and epilepsy. These strategies may ultimately improve disease detection, therapeutic monitoring, and treatment efficacy esp. in the context of antiangiogenic chemotherapy and antiinflammatory medications. The purpose of this review is to outline the current status of superparamagnetic iron oxide nanoparticles in the context of biomedical nanotechnol. as they apply to diagnostic MRI and potential therapeutic applications in neurooncol. and other CNS inflammatory conditions.
- 9Dulińska-Litewka J. Łazarczyk A. Hałubiec P. Szafrański O. Karnas K. Karewicz A. Superparamagnetic iron oxide nanoparticles—Current and prospective medical applications Materials 2019 12 4 6179Superparamagnetic iron oxide nanoparticles- current and prospective medical applicationsDulinska-Litewka, Joanna; Lazarczyk, Agnieszka; Halubiec, Przemyslaw; Szafranski, Oskar; Karnas, Karolina; Karewicz, AnnaMaterials (2019), 12 (4), 617CODEN: MATEG9; ISSN:1996-1944. (MDPI AG)The recent, fast development of nanotechnol. is reflected in the medical sciences. Superparamagnetic Iron Oxide Nanoparticles (SPIONs) are an excellent example. Thanks to their superparamagnetic properties, SPIONs have found application in Magnetic Resonance Imaging (MRI) and magnetic hyperthermia. Unlike bulk iron, SPIONs do not have remnant magnetization in the absence of the external magnetic field; therefore, a precise remote control over their action is possible. This makes them also useful as a component of the advanced drug delivery systems. Due to their easy synthesis, biocompatibility, multifunctionality, and possibility of further surface modification with various chem. agents, SPIONs could support many fields of medicine. SPIONs have also some disadvantages, such as their high uptake by macrophages. Nevertheless, based on the ongoing studies, they seem to be very promising in oncol. therapy (esp. in the brain, breast, prostate, and pancreatic tumors). The main goal of our paper is, therefore, to present the basic properties of SPIONs, to discuss their current role in medicine, and to review their applications in order to inspire future developments of new, improved SPION systems.
- 10Min Y. Caster J. M. Eblan M. J. Wang A. Z. Clinical translation of nanomedicine Chem. Rev. 2015 115 19 11147 1119010Clinical Translation of NanomedicineMin, Yuanzeng; Caster, Joseph M.; Eblan, Michael J.; Wang, Andrew Z.Chemical Reviews (Washington, DC, United States) (2015), 115 (19), 11147-11190CODEN: CHREAY; ISSN:0009-2665. (American Chemical Society)A review discussing the clin. translation of nanomedicine, including liposome and lipid-based nanomedicine, protein nanoparticles, polymer-drug conjugates, polymeric micelles and nanoparticles, inorg. nanoparticles, and summary and outlook.
- 11Gao L. Zhuang J. Nie L. Zhang J. Zhang Y. Gu N. Wang T. Feng J. Yang D. Perrett S. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles Nat. Nanotechnol. 2007 2 9 577 58311Intrinsic peroxidase-like activity of ferromagnetic nanoparticlesGao, Lizeng; Zhuang, Jie; Nie, Leng; Zhang, Jinbin; Zhang, Yu; Gu, Ning; Wang, Taihong; Feng, Jing; Yang, Dongling; Perrett, Sarah; Yan, XiyunNature Nanotechnology (2007), 2 (9), 577-583CODEN: NNAABX; ISSN:1748-3387. (Nature Publishing Group)Nanoparticles contg. magnetic materials, such as magnetite (Fe3O4), are particularly useful for imaging and sepn. techniques. As these nanoparticles are generally considered to be biol. and chem. inert, they are typically coated with metal catalysts, antibodies or enzymes to increase their functionality as sepn. agents. Here, we report that magnetite nanoparticles in fact possess an intrinsic enzyme mimetic activity similar to that found in natural peroxidases, which are widely used to oxidize org. substrates in the treatment of wastewater or as detection tools. Based on this finding, we have developed a novel immunoassay in which antibody-modified magnetite nanoparticles provide three functions: capture, sepn. and detection. The stability, ease of prodn. and versatility of these nanoparticles makes them a powerful tool for a wide range of potential applications in medicine, biotechnol. and environmental chem.
- 12Wei H. Wang E. Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymes Chem. Soc. Rev. 2013 42 14 6060 9312Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymesWei, Hui; Wang, ErkangChemical Society Reviews (2013), 42 (14), 6060-6093CODEN: CSRVBR; ISSN:0306-0012. (Royal Society of Chemistry)A review. Over the past few decades, researchers have established artificial enzymes as highly stable and low-cost alternatives to natural enzymes in a wide range of applications. A variety of materials including cyclodextrins, metal complexes, porphyrins, polymers, dendrimers and biomols. have been extensively explored to mimic the structures and functions of naturally occurring enzymes. Recently, some nanomaterials have been found to exhibit unexpected enzyme-like activities, and great advances have been made in this area due to the tremendous progress in nano-research and the unique characteristics of nanomaterials. To highlight the progress in the field of nanomaterial-based artificial enzymes (nanozymes), this review discusses various nanomaterials that have been explored to mimic different kinds of enzymes. We cover their kinetics, mechanisms and applications in numerous fields, from biosensing and immunoassays, to stem cell growth and pollutant removal. We also summarize several approaches to tune the activities of nanozymes. Finally, we make comparisons between nanozymes and other catalytic materials (other artificial enzymes, natural enzymes, org. catalysts and nanomaterial-based catalysts) and address the current challenges and future directions (302 refs.).
- 13Lin Y. Ren J. Qu X. Catalytically active nanomaterials: a promising candidate for artificial enzymes Acc. Chem. Res. 2014 47 4 1097 10513Catalytically active nanomaterials: A promising candidate for artificial enzymesLin, Youhui; Ren, Jinsong; Qu, XiaogangAccounts of Chemical Research (2014), 47 (4), 1097-1105CODEN: ACHRE4; ISSN:0001-4842. (American Chemical Society)A review. Natural enzymes, exquisite biocatalysts mediating every biol. process in living organisms, are able to accelerate the rate of chem. reactions up to 1019-fold for specific substrates and reactions. However, the practical application of enzymes is often hampered by their intrinsic drawbacks, such as low operational stability, sensitivity of catalytic activity to environmental conditions, and high costs in prepn. and purifn. Therefore, the discovery and development of artificial enzymes is highly desired. Recently, the merging of nanotechnol. with biol. has ignited extensive research efforts for designing functional nanomaterials that exhibit various properties intrinsic to enzymes. As a promising candidate for artificial enzymes, catalytically active nanomaterials (nanozymes) show several advantages over natural enzymes, such as controlled synthesis in low cost, tunability in catalytic activities, as well as high stability against stringent conditions. Here, the authors focus on their recent progress in exploring and constructing such nanoparticulate artificial enzymes, including graphene oxide, graphene-hemin nanocomposites, carbon nanotubes, carbon nanodots, mesoporous silica-encapsulated gold nanoparticles, gold nanoclusters, and nanoceria. According to their structural characteristics, these enzyme mimics have been categorized into 3 classes: carbon-based, metal-based, and metal-oxide-based nanomaterials. The authors highlight the important role of catalytic nanomaterials in the fields of biomimetics. First, the authors provide a practical introduction to the identification of these nanozymes, the source of the enzyme-like activities, and the enhancement of activities via rational design and engineering. Then, the authors briefly describe new or enhanced applications of certain nanozymes in biomedical diagnosis, environmental monitoring, and therapeutics. For instance, the authors have successfully used these biomimetic catalysts as colorimetric probes for the detection of cancer cells, nucleic acids, proteins, metal ions, and other small mols. In addn., the authors also introduce 3 exciting advances in the use of efficient modulators on artificial enzyme systems to improve the catalytic performance of existing nanozymes. For example, the authors report that graphene oxide could serve as a modulator to greatly improve the catalytic activity of lysozyme-stabilized gold nanoclusters at neutral pH, which will have great potential for applications in biol. systems. The authors show that, through the incorporation of modulators into artificial enzymes, they can offer a facile but highly effective way to improve their overall catalytic performance or realize catalytic reactions that were not possible in the past. The authors expect that nanozymes with unique properties and functions will attract increasing research interest and lead to new opportunities in various fields of research.
- 14Gao L. Yan X. Nanozymes: an emerging field bridging nanotechnology and biology Sci. China: Life Sci. 2016 59 4 400 214Nanozymes: an emerging field bridging nanotechnology and biologyGao Lizeng; Yan Xiyun; Gao LizengScience China. Life sciences (2016), 59 (4), 400-2 ISSN:.There is no expanded citation for this reference.
- 15Ragg R. Tahir M. N. Tremel W. Solids Go Bio: Inorganic Nanoparticles as Enzyme Mimics Eur. J. Inorg. Chem. 2016 2016 13-14 1906 191515Solids Go Bio: Inorganic Nanoparticles as Enzyme MimicsRagg, Ruben; Tahir, Muhammad N.; Tremel, WolfgangEuropean Journal of Inorganic Chemistry (2016), 2016 (13-14), 1906-1915CODEN: EJICFO; ISSN:1434-1948. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. A longstanding goal of biomimetic chem. is the design and synthesis of functional enzyme mimics. The past three decades have seen a wide variety of materials, including metal complexes, polymers and other biomols., that mimic the structures and functions of naturally occurring enzymes. Among these, inorg. nanoparticles offer huge potential, because they are more stable than their natural counterparts, while having large surface areas and sizes comparable to those of natural enzymes. Therefore, a considerable no. of "artificial enzymes" derived from inorg. nanomaterials have been reported. This microreview highlights the recent progress in the field of enzymically active inorg. nanomaterials, including mimics of peroxidases, haloperoxidases, superoxide dismutases and sulfite oxidases, along with selected biotechnol. applications and their future prospects.
- 16Wang X. Hu Y. Wei H. Nanozymes in bionanotechnology: from sensing to therapeutics and beyond Inorg. Chem. Front. 2016 3 1 41 6016Nanozymes in bionanotechnology: from sensing to therapeutics and beyondWang, Xiaoyu; Hu, Yihui; Wei, HuiInorganic Chemistry Frontiers (2016), 3 (1), 41-60CODEN: ICFNAW; ISSN:2052-1553. (Royal Society of Chemistry)In the past few decades, researchers have developed lots of artificial enzymes with various materials to mimic the structures and functions of natural enzymes. Recently, nanozymes, nanomaterials with enzyme-like characteristics, are emerging as novel artificial enzymes, and attracting researchers' enormous interest. Remarkable advances have been made in the area of nanozymes due to their unique properties compared with natural enzymes and classic artificial enzymes. Until now, lots of nanomaterials have been studied to mimic various natural enzymes for wide applications. To highlight the recent progress of nanozymes (esp. in bionanotechnol.), here we discuss the diverse applications of nanozymes, which range from sensing, imaging, and therapeutics, to logic gates, pollutant removal, water treatment, etc. Finally, we address the current challenges facing nanozyme research as well as possible directions to fulfill their great potential in future.
- 17Tang G. He J. Liu J. Yan X. Fan K. Nanozyme for tumor therapy: Surface modification matters Exploration 2021 1 1 75 89There is no corresponding record for this reference.
- 18Lee J. Liao H. Wang Q. Han J. Han J.-H. Shin H. E. Ge M. Park W. Li F. Exploration of nanozymes in viral diagnosis and therapy Exploration 2022 2 1 20210086There is no corresponding record for this reference.
- 19Yan, X.; Gao, L. Nanozymology; Springer, 2020.There is no corresponding record for this reference.
- 20Wang Z. Zhang R. Yan X. Fan K. Structure and activity of nanozymes: Inspirations for de novo design of nanozymes Mater. Today 2020 41 81 11920Structure and activity of nanozymes: Inspirations for de novo design of nanozymesWang, Zhuoran; Zhang, Ruofei; Yan, Xiyun; Fan, KelongMaterials Today (Oxford, United Kingdom) (2020), 41 (), 81-119CODEN: MTOUAN; ISSN:1369-7021. (Elsevier Ltd.)A review on structure and activity of nanozymes, Inspirations for de novo design of nanozymes. Nanozymes, nanomaterials with enzyme-like activities, are becoming powerful competitors and potential substitutes for natural enzymes because of their excellent performance, including design from scratch, controllable activity, and environmental resistance. In recent years, various nanozymes have been discovered or designed, and gradually applied to mol. detection, biomedical treatment and environmental management. Nevertheless, nanozymes are often regarded as fascinating and confusing black boxes as their catalytic mechanisms remain largely indistinct. Interestingly, recent researches have shed light into these black boxes. It appears that the enzymic activities of nanozymes are closely related to their size, surface lattice, surface modification and compn., etc. Some regular structure-activity relationships have been elucidated in recent reports. In this review, we systematically summarized the studies on the structure-activity relationship of nanozymes in recent years, aiming to illustrate the catalytic mechanism of nanozymes and clarify the key factors regulating their behavior, so as to provide ideas and inspiration for the de novo design of nanozymes.
- 21Jiang B. Duan D. Gao L. Zhou M. Fan K. Tang Y. Xi J. Bi Y. Tong Z. Gao G. F. Standardized assays for determining the catalytic activity and kinetics of peroxidase-like nanozymes Nat. Protoc. 2018 13 7 1506 152021Standardized assays for determining the catalytic activity and kinetics of peroxidase-like nanozymesJiang, Bing; Duan, Demin; Gao, Lizeng; Zhou, Mengjie; Fan, Kelong; Tang, Yan; Xi, Juqun; Bi, Yuhai; Tong, Zhou; Gao, George Fu; Xie, Ni; Tang, Aifa; Nie, Guohui; Liang, Minmin; Yan, XiyunNature Protocols (2018), 13 (7), 1506-1520CODEN: NPARDW; ISSN:1750-2799. (Nature Research)Nanozymes are nanomaterials exhibiting intrinsic enzyme-like characteristics that have increasingly attracted attention, owing to their high catalytic activity, low cost and high stability. This combination of properties has enabled a broad spectrum of applications, ranging from biol. detection assays to disease diagnosis and biomedicine development. Since the intrinsic peroxidase activity of Fe3O4 nanoparticles (NPs) was first reported in 2007, >40 types of nanozymes have been reported that possess peroxidase-, oxidase-, haloperoxidase- or superoxide dismutase-like catalytic activities. Given the complex interdependence of the physicochem. properties and catalytic characteristics of nanozymes, it is important to establish a std. by which the catalytic activities and kinetics of various nanozymes can be quant. compared and that will benefit the development of nanozyme-based detection and diagnostic technologies. Here, we first present a protocol for measuring and defining the catalytic activity units and kinetics for peroxidase nanozymes, the most widely used type of nanozyme. In addn., we describe the detailed exptl. procedures for a typical nanozyme strip-based biol. detection test and demonstrate that nanozyme-based detection is repeatable and reliable when guided by the presented nanozyme catalytic std. The catalytic activity and kinetics assays for a nanozyme can be performed within 4 h.
- 22Gao L. Fan K. Yan X. Iron Oxide Nanozyme: A Multifunctional Enzyme Mimetic for Biomedical Applications Theranostics 2017 7 13 3207 322722Iron oxide nanozyme: a multifunctional enzyme mimetic for biomedical applicationsGao, Lizeng; Fan, Kelong; Yan, XiyunTheranostics (2017), 7 (13), 3207-3227CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Iron oxide nanoparticles have been widely used in many important fields due to their excellent nanoscale phys. properties, such as magnetism/superparamagnetism. They are usually assumed to be biol. inert in biomedical applications. However, iron oxide nanoparticles were recently found to also possess intrinsic enzyme-like activities, and are now regarded as novel enzyme mimetics. A special term, "Nanozyme", has thus been coined to highlight the intrinsic enzymic properties of such nanomaterials. Since then, iron oxide nanoparticles have been used as nanozymes to facilitate biomedical applications. In this review, we will introduce the enzymic features of iron oxide nanozyme (IONzyme), and summarize its novel applications in biomedicine.
- 23Chen Z. Yin J.-J. Zhou Y.-T. Zhang Y. Song L. Song M. Hu S. Gu N. Dual enzyme-like activities of iron oxide nanoparticles and their implication for diminishing cytotoxicity ACS Nano 2012 6 5 4001 401223Dual Enzyme-like Activities of Iron Oxide Nanoparticles and Their Implication for Diminishing CytotoxicityChen, Zhongwen; Yin, Jun-Jie; Zhou, Yu-Ting; Zhang, Yu; Song, Lina; Song, Mengjie; Hu, Sunling; Gu, NingACS Nano (2012), 6 (5), 4001-4012CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)Iron oxide nanoparticles (IONPs) are frequently used in biomedical applications, yet their toxic potential is still a major concern. While most studies of biosafety focus on cellular responses after exposure to nanomaterials, little is reported to analyze reactions on the surface of nanoparticles as a source of cytotoxicity. Here we report that different intracellular microenvironment in which IONPs are located leads to contradictive outcomes in their abilities to produce free radicals. We first verified pH-dependent peroxidase-like and catalase-like activities of IONPs and investigated how they interact with hydrogen peroxide (H2O2) within cells. Results showed that IONPs had a concn.-dependent cytotoxicity on human glioma U251 cells, and they could enhance H2O2-induced cell damage dramatically. By conducting ESR spectroscopy expts., we showed that both Fe3O4 and γ-Fe2O3 nanoparticles could catalyze H2O2 to produce hydroxyl radicals in acidic lysosome mimic conditions, with relative potency Fe3O4 > γ-Fe2O3, which was consistent with their peroxidase-like activities. However, no hydroxyl radicals were obsd. in neutral cytosol mimic conditions with both nanoparticles. Instead, they decompd. H2O2 into H2O and O2 directly in this condition through catalase-like activities. Transmission electron micrographs revealed that IONPs located in lysosomes in cells, the acidic environment of which may contribute to hydroxyl radical prodn. This is the first study regarding cytotoxicity based on their enzyme-like activities. Since H2O2 is continuously produced in cells, our data indicate that lysosome-escaped strategy for IONP delivery would be an efficient way to diminish long-term toxic potential.
- 24Dolphin D. Forman A. Borg D. Fajer J. Felton R. Compounds I of catalase and horse radish peroxidase: π-cation radicals Proc. Natl. Acad. Sci. 1971 68 3 614 61824Compounds I of catalase and horse radish peroxidase: πcation radicalsDolphin, David; Forman, A.; Borg, Douglas C.; Fajer, J.; Felton, Ronald H.Proceedings of the National Academy of Sciences of the United States of America (1971), 68 (3), 614-18CODEN: PNASA6; ISSN:0027-8424.Two-electron oxidn. of cobaltous octaethylporphyrin [Co(II)(Et)8P] yields a stable π-cation radical [Co(III)(Et)8P]2+, the optical spectrum of which exhibits spectral changes dependent upon the nature of the counterion. Comparison of these spectra with those of Compds. I of horseradish peroxidase and catalase suggests that these Compds. I contain a π-cation radical of the heme prosthetic group. This proposal explains the oxidn. level, optical spectra, and stability of the primary compds. without recourse to properties such as stoichiometric mixts. of special porphyrins, stable Fe(V) porphyrins, or unique conformers of heme porphyrins. Explanations are advanced to account for the missing ESR signal of Compd. I of horseradish peroxidase.
- 25Veitch N. C. Horseradish peroxidase: a modern view of a classic enzyme Phytochemistry 2004 65 3 249 25925Horseradish peroxidase: a modern view of a classic enzymeVeitch, Nigel C.Phytochemistry (Elsevier) (2004), 65 (3), 249-259CODEN: PYTCAS; ISSN:0031-9422. (Elsevier Science B.V.)A review. Horseradish peroxidase (HRP) is an important heme-contg. enzyme that has been studied for more than a century. In recent years, new information has become available on the 3-dimensional structure of the enzyme and its catalytic intermediates, mechanisms of catalysis, and the function of specific amino acid residues. Site-directed mutagenesis and directed evolution techniques are now used routinely to investigate the structure and function of HRP and offer the opportunity to develop engineered enzymes for practical applications in natural product and fine chems. synthesis, medical diagnostics, and bioremediation. A combination of HRP and indole-3-acetic acid (IAA) or its derivs. is currently being evaluated as an agent for use in targeted cancer therapies. Physiol. roles traditionally assocd. with the enzyme that include IAA metab., crosslinking of biol. polymers, and lignification are becoming better understood at the mol. level, but the involvement of specific HRP isoenzymes in these processes is not yet clearly defined. Progress in this area should result from the identification of the entire peroxidase gene family of Arabidopsis thaliana, which has now been completed.
- 26Rodríguez-López J. N. Lowe D. J. Hernández-Ruiz J. Hiner A. N. García-Cánovas F. Thorneley R. N. Mechanism of reaction of hydrogen peroxide with horseradish peroxidase: identification of intermediates in the catalytic cycle J. Am. Chem. Soc. 2001 123 48 11838 1184726Mechanism of reaction of hydrogen peroxide with horseradish peroxidase: Identification of intermediates in the catalytic cycleRodriguez-Lopez, Jose Neptuno; Lowe, David J.; Hernandez-Ruiz, Josefa; Hiner, Alexander N. P.; Garcia-Canovas, Francisco; Thorneley, Roger N. F.Journal of the American Chemical Society (2001), 123 (48), 11838-11847CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)The mechanism of the reaction of horseradish peroxidase isoenzyme C (HRPC) with H2O2 to form reactive enzyme intermediate compd. I was studied using electronic absorbance, rapid-scan stopped-flow, and ESR spectroscopies at both acid and basic pH. The roles of active site residues His-42 and Arg-38 in controlling heterolytic cleavage of the H2O2 O-O bond were probed with site-directed mutant enzymes H42L, R38L, and R38G. The biphasic reaction kinetics of H42L with H2O2 suggested the presence of an intermediate species and, at acid pH, a reversible 2nd step, probably due to a neutral enzyme-H2O2 complex and the ferric-peroxoanion-contg. compd. 0. ESR also indicated the formation of a protein radical situated more than ∼10 Å from the heme Fe. The stoichiometry of the reaction of the H42L/H2O2 reaction product and 2,2'-azinobis(3-ethylbenzothiazolinesulfonic acid) (ABTS) was concn.-dependent and fell from a value of 2 to 1 above 0.7 mM ABTS. These data could be explained if H2O2 underwent homolytic cleavage in H42L. The apparent rate of compd. I formation by H42L, while low, was pH-independent in contrast to wild-type HRPC where the rate fell at acid pH, indicating the involvement of an ionizable group with pKa of ∼4. In R38L and R38G, the apparent pKa was shifted to ∼8, but there was no evidence that homolytic cleavage of H2O2 occurred. These data suggest that His-42 acts initially as a proton acceptor (base catalyst) and then as a donor (acid catalyst) at neutral pH and predict the obsd. slower rate and lower efficiency of heterolytic cleavage obsd. at acid pH. Arg-38 was influential in lowering the pKa of His-42 and addnl. in aligning H2O2 in the active site, but it did not play a direct role in proton transfer.
- 27Berglund G. I. Carlsson G. H. Smith A. T. Szöke H. Henriksen A. Hajdu J. The catalytic pathway of horseradish peroxidase at high resolution Nature 2002 417 6887 463 46827The catalytic pathway of horseradish peroxidase at high resolutionBerglund, Gunnar I.; Carlsson, Gunilla H.; Smith, Andrew T.; Szoeke, Hanna; Henriksen, Anette; Hajdu, JanosNature (London, United Kingdom) (2002), 417 (6887), 463-468CODEN: NATUAS; ISSN:0028-0836. (Nature Publishing Group)A mol. description of O2 and H2O2 activation in biol. systems is difficult, because electrons liberated during x-ray data collection reduce the active centers of redox enzymes catalyzing these reactions. Here, the authors describe an effective strategy to obtain crystal structures for high-valency redox intermediates and present a 3-dimensional movie of the x-ray-driven catalytic redn. of a bound O2 species in horseradish peroxidase (HRP). The authors also describe sep. expts. in which high-resoln. structures could be obtained for all 5 oxidn. states of HRP, showing such structures with preserved redox states for the 1st time.
- 28Yuan B. Chou H. L. Peng Y. K. Disclosing the Origin of Transition Metal Oxides as Peroxidase (and Catalase) Mimetics ACS Appl. Mater. Interfaces 2021 1 9There is no corresponding record for this reference.
- 29Wang N. Zhu L. Wang D. Wang M. Lin Z. Tang H. Sono-assisted preparation of highly-efficient peroxidase-like Fe3O4 magnetic nanoparticles for catalytic removal of organic pollutants with H2O2 Ultrason. Sonochem. 2010 17 3 526 53329Sono-assisted preparation of highly-efficient peroxidase-like Fe3O4 magnetic nanoparticles for catalytic removal of organic pollutants with H2O2Wang, Nan; Zhu, Li-Hua; Wang, Da-Li; Wang, Ming-Qiong; Lin, Zhi-Fen; Tang, He-QingUltrasonics Sonochemistry (2010), 17 (3), 526-533CODEN: ULSOER; ISSN:1350-4177. (Elsevier B.V.)Fe3O4 magnetic nanoparticles (Fe3O4 MNPs) with much improved peroxidase-like activity were successfully prepd. through an advanced reverse co-pptn. method under the assistance of ultrasound irradn. The characterizations with XRD, BET and SEM indicated that the ultrasound irradn. in the prepn. induced the prodn. of Fe3O4 MNPs possessing smaller particle sizes (16.5 nm), greater BET surface area (82.5 m2 g-1) and much higher dispersibility in water. The particle sizes, BET surface area, chem. compn. and then catalytic property of the Fe3O4 MNPs could be tailored by adjusting the initial concn. of ammonia water and the molar ratio of Fe2+/Fe3+ during the prepn. process. The H2O2-activating ability of Fe3O4 MNPs was evaluated by using Rhodamine B (RhB) as a model compd. of org. pollutants to be degraded. At pH 5.4 and temp. 40 °C, the sonochem. synthesized Fe3O4 MNPs were obsd. to be able to activate H2O2 and remove ca. 90% of RhB (0.02 mmol L-1) in 60 min with a apparent rate const. of 0.034 min-1 for the RhB degrdn., being 12.6 folds of that (0.0027 min-1) over the Fe3O4 MNPs prepd. via a conventional reverse co-pptn. method. The mechanisms of the peroxidase-like catalysis with Fe3O4 MNPs were discussed to develop more efficient novel catalysts.
- 30Zhang J. Zhuang J. Gao L. Zhang Y. Gu N. Feng J. Yang D. Zhu J. Yan X. Decomposing phenol by the hidden talent of ferromagnetic nanoparticles Chemosphere 2008 73 9 1524 152830Decomposing phenol by the hidden talent of ferromagnetic nanoparticlesZhang, Jinbin; Zhuang, Jie; Gao, Lizeng; Zhang, Yu; Gu, Ning; Feng, Jing; Yang, Dongling; Zhu, Jingdong; Yan, XiyunChemosphere (2008), 73 (9), 1524-1528CODEN: CMSHAF; ISSN:0045-6535. (Elsevier Ltd.)Researches on modified Fenton reactions applied in phenol degrdn. have been focused on reducing secondary pollution and enhancing catalytic efficiency. Newly developed methods utilizing carriers, such as Resin and Nafion, to immobilize Fe2+ could avoid iron ion leakage. However, the requirement of high temp. and the limited reaction efficiency still restrained them from broad application. Based on a recently discovered "hidden talent" of ferromagnetic nanoparticles (MNPs), we established a MNP-catalyzed phenol removal assay, which could overcome these limitations. Our results showed that the MNPs removed over 85% phenol from aq. soln. within 3 h even at 16°. The catalytic condition was extensively optimized among a range of pH, temp. as well as initial concn. of phenol and H2O2. TOC and GC/MS anal. revealed that about 30% phenol was mineralized while the rest became small mol. org. acids. Moreover the MNPs were thermo-stable and could be regenerated for at least five rounds. Thus, our findings open up a wide spectrum of environmental friendly applications of MNPs showing several attractive features, such as easy prepn., low cost, thermo-stability and reusability.
- 31Fan K. Wang H. Xi J. Liu Q. Meng X. Duan D. Gao L. Yan X. Optimization of Fe3O4 nanozyme activity via single amino acid modification mimicking an enzyme active site Chem. Commun. 2017 53 2 424 42731Optimization of Fe3O4 nanozyme activity via single amino acid modification mimicking an enzyme active siteFan, Kelong; Wang, Hui; Xi, Juqun; Liu, Qi; Meng, Xiangqin; Duan, Demin; Gao, Lizeng; Yan, XiyunChemical Communications (Cambridge, United Kingdom) (2017), 53 (2), 424-427CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)The Fe3O4 nanozyme was the 1st reported nanoparticle with intrinsic peroxidase-like activity and has been widely used in biomedicine. Here, to optimize its catalytic activity, the authors introduced His residues onto the Fe3O4 nanoparticle surface in order to mimic the enzymic microenvironment of natural peroxidases. The results showed that modification with a single amino acid could improve the apparent affinity (Km) of the Fe3O4 nanozyme by >10-fold for the substrate, H2O2, and enhance its catalytic efficiency (kcat/Km) by ≤20-fold. Thus, the authors not only optimized the activity of the Fe3O4 nanozyme, but also provided a new rationale for improving the efficiency of nanomaterial-based catalysts by utilizing strategies obsd. in Nature.
- 32Cheon H. J. Nguyen Q. H. Kim M. I. Highly Sensitive Fluorescent Detection of Acetylcholine Based on the Enhanced Peroxidase-Like Activity of Histidine Coated Magnetic Nanoparticles Nanomaterials 2021 11 5 120732Highly sensitive fluorescent detection of acetylcholine based on the enhanced peroxidase-like activity of histidine coated magnetic nanoparticlesCheon, Hong Jae; Nguyen, Quynh Huong; Kim, Moon IlNanomaterials (2021), 11 (5), 1207CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Inspired by the active site structure of natural horseradish peroxidase having iron as a pivotal element with coordinated histidine residues, we have developed histidine coated magnetic nanoparticles (His@MNPs) with relatively uniform and small sizes (less than 10 nm) through one-pot heat treatment. In comparison to pristine MNPs and other amino acid coated MNPs, His@MNPs exhibited a considerably enhanced peroxidase-imitating activity, approaching 10-fold higher in catalytic reactions. With the high activity, His@MNPs then were exploited to detect the important neurotransmitter acetylcholine. By coupling choline oxidase and acetylcholine esterase with His@MNPs as peroxidase mimics, target choline and acetylcholine were successfully detected via fluorescent mode with high specificity and sensitivity with the limits of detection down to 200 and 100 nM, resp. The diagnostic capability of the method is demonstrated by analyzing acetylcholine in human blood serum. This study thus demonstrates the potential of utilizing His@MNPs as peroxidase-mimicking nanozymes for detecting important biol. and clin. targets with high sensitivity and reliability.
- 33Duan D. Fan K. Zhang D. Tan S. Liang M. Liu Y. Zhang J. Zhang P. Liu W. Qiu X. Nanozyme-strip for rapid local diagnosis of Ebola Biosens. Bioelectron. 2015 74 134 14133Nanozyme-strip for rapid local diagnosis of EbolaDuan, Demin; Fan, Kelong; Zhang, Dexi; Tan, Shuguang; Liang, Mifang; Liu, Yang; Zhang, Jianlin; Zhang, Panhe; Liu, Wei; Qiu, Xiangguo; Kobinger, Gary P.; Gao, George Fu; Yan, XiyunBiosensors & Bioelectronics (2015), 74 (), 134-141CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)Ebola continues to rage in West Africa. In the absence of an approved vaccine or treatment, the priority in controlling this epidemic is to promptly identify and isolate infected individuals. To this end, a rapid, highly sensitive, and easy-to-use test for Ebola diagnosis is urgently needed. Here, by using Fe3O4 magnetic nanoparticle (MNP) as a nanozyme probe, we developed a MNP-based immunochromatog. strip (Nanozyme-strip), which detects the glycoprotein of Ebola virus (EBOV) as low as 1 ng/mL, which is 100-fold more sensitive than the std. strip method. The sensitivity of the Nanozyme-strip for EBOV detection and diagnostic accuracy for New Bunyavirus clin. samples is comparable with ELISA, but is much faster (within 30 min) and simpler (without need of specialist facilities). The results demonstrate that the Nanozyme-strip test can rapidly and sensitively detect EBOV, providing a valuable simple screening tool for diagnosis of infection in Ebola-stricken areas.
- 34Fan K. Cao C. Pan Y. Lu D. Yang D. Feng J. Song L. Liang M. Yan X. Magnetoferritin nanoparticles for targeting and visualizing tumour tissues Nat. Nanotechnol. 2012 7 7 459 46434Magnetoferritin nanoparticles for targeting and visualizing tumour tissuesFan, Kelong; Cao, Changqian; Pan, Yongxin; Lu, Di; Yang, Dongling; Feng, Jing; Song, Lina; Liang, Minmin; Yan, XiyunNature Nanotechnology (2012), 7 (7), 459-464CODEN: NNAABX; ISSN:1748-3387. (Nature Publishing Group)Engineered nanoparticles have been used to provide diagnostic, therapeutic and prognostic information about the status of disease. Nanoparticles developed for these purposes are typically modified with targeting ligands (such as antibodies, peptides or small mols.) or contrast agents using complicated processes and expensive reagents. Moreover, this approach can lead to an excess of ligands on the nanoparticle surface, and this causes non-specific binding and aggregation of nanoparticles, which decreases detection sensitivity. Here, we show that magnetoferritin nanoparticles (M-HFn) can be used to target and visualize tumor tissues without the use of any targeting ligands or contrast agents. Iron oxide nanoparticles are encapsulated inside a recombinant human heavy-chain ferritin (HFn) protein shell, which binds to tumor cells that overexpress transferrin receptor 1 (TfR1). The iron oxide core catalyzes the oxidn. of peroxidase substrates in the presence of hydrogen peroxide to produce a color reaction that is used to visualize tumor tissues. We examd. 474 clin. specimens from patients with nine types of cancer and verified that these nanoparticles can distinguish cancerous cells from normal cells with a sensitivity of 98% and specificity of 95%.
- 35Liu D. Ju C. Han C. Shi R. Chen X. Duan D. Yan J. Yan X. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen Biosens. Bioelectron. 2021 173 11281735Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigenLiu, Dan; Ju, Chenhui; Han, Chao; Shi, Rui; Chen, Xuehui; Duan, Demin; Yan, Jinghua; Yan, XiyunBiosensors & Bioelectronics (2021), 173 (), 112817CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)COVID-19 has evolved into a global pandemic. Early and rapid detection is crucial to control of the SARS-CoV-2 transmission. While representing the gold std. for early diagnosis, nucleic acid tests for SARS-CoV-2 are often complicated and time-consuming. Serol. rapid antibody tests are characterized by high rates of false-neg. diagnoses, esp. during early infection. Here, we developed a novel nanozyme-based chemiluminescence paper assay for rapid and sensitive detection of SARS-CoV-2 spike antigen, which integrates nanozyme and enzymic chemiluminescence immunoassay with the lateral flow strip. The core of our paper test is a robust Co-Fe@hemin-peroxidase nanozyme that catalyzes chemiluminescence comparable with natural peroxidase HRP and thus amplifies immune reaction signal. The detection limit for recombinant spike antigen of SARS-CoV-2 was 0.1 ng/mL, with a linear range of 0.2-100 ng/mL. Moreover, the sensitivity of test for pseudovirus could reach 360 TCID50/mL, which was comparable with ELISA method. The strip recognized SARS-CoV-2 antigen specifically, and there was no cross reaction with other coronaviruses or influenza A subtypes. This testing can be completed within 16 min, much shorter compared to the usual 1-2 h required for currently used nucleic acid tests. Furthermore, signal detection is feasible using the camera of a std. smartphone. Ingredients for nanozyme synthesis are simple and readily available, considerably lowering the overall cost. In conclusion, our paper test provides a high-sensitive point-of-care testing (POCT) approach for SARS-CoV-2 antigen detection, which should greatly facilitate early screening of SARS-CoV-2 infections, and considerably lower the financial burden on national healthcare resources.
- 36Mansur A. A. Mansur H. S. Carvalho S. M. Engineered hybrid nanozyme catalyst cascade based on polysaccharide-enzyme-magnetic iron oxide nanostructures for potential application in cancer therapy Catal. Today 2020 187 198There is no corresponding record for this reference.
- 37Vallabani N. S. Vinu A. Singh S. Karakoti A. Tuning the ATP-triggered pro-oxidant activity of iron oxide-based nanozyme towards an efficient antibacterial strategy J. Colloid Interface Sci. 2020 567 154 16437Tuning the ATP-triggered pro-oxidant activity of iron oxide-based nanozyme towards an efficient antibacterial strategyVallabani, N. V. Srikanth; Vinu, Ajayan; Singh, Sanjay; Karakoti, AjayJournal of Colloid and Interface Science (2020), 567 (), 154-164CODEN: JCISA5; ISSN:0021-9797. (Elsevier B.V.)An alarming increase in bacterial resistance towards various types of antibiotics makes it imperative to design alternate or combinational therapies to treat stubborn bacterial infections. In this perspective, emerging tools like nanozymes, nanomaterials with biol. enzyme like characteristics, are being utilized to control infections caused by bacterial pathogens. Among several nanozymes used for antibacterial applications, Fe3O4 nanoparticles (NP) received great attention due to their effective peroxidase like activity. The pH dependent peroxidase activity of Fe3O4 NP results in generation of ·OH radical via the unique Fenton chem. of iron. However, their pH dependent activity is restricted to acidic environment and dramatic loss in antibacterial activity is obsd. at near neutral pH. Here we describe a novel strategy to overcome the pH lacunas of citrate coated Fe3O4 NP by utilizing ATP disodium salt (ATP) as a synergistic agent to accelerate the ·OH radical prodn. and restore its antibacterial activity over a wide range of pH. This synergistic combination (30μg/mL Fe3O4 NP and 2.5 mM ATP) shows a high bactericidal activity against both gram pos. (B. subtilis) and gram neg. (E. coli) bacterial strains, in presence of H2O2, at neutral pH. The synergistic effect (Fe3O4 NP + ATP) is detd. from the viability assessment and membrane damage studies and is further confirmed by comparing the concn. of generated ·OH radicals. Over all, this study illustrates ATP assisted and ·OH-mediated bactericidal activity of Fe3O4 nanozyme at near neutral pH.
- 38Shi S. Wu S. Shen Y. Zhang S. Xiao Y. He X. Gong J. Farnell Y. Tang Y. Huang Y. Iron oxide nanozyme suppresses intracellular Salmonella Enteritidis growth and alleviates infection in vivo Theranostics 2018 8 22 614938Iron oxide nanozyme suppresses intracellular Salmonella Enteritidis growth and alleviates infection in vivoShi, Shourong; Wu, Shu; Shen, Yiru; Zhang, Shan; Xiao, Yunqi; He, Xi; Gong, Jiansen; Farnell, Yuhua; Tang, Yan; Huang, Yixin; Gao, LizengTheranostics (2018), 8 (22), 6149-6162CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Salmonella Enteritidis S.Enteritidis is a globally significant zoonotic foodborne pathogen which has led to large nos. of deaths in humans and caused economic losses in animal husbandry. S.Enteritidis invades host cells and survives within the cells, causing resistance to antibiotic treatment. Effective methods of elimination and eradication of intracellular S.Enteritidis are still very limited. Here we evaluated whether a new intracellular antibacterial strategy using iron oxide nanozymes (IONzymes) exerted highly antibacterial efficacy via its intrinsic peroxidase-like activity in vitro and in vivo. The antibacterial activities of IONzymes against planktonic S.Enteritidis, intracellular S.Enteritidis in Leghorn Male Hepatoma-derived cells (LMH), and liver from specific pathogen free (SPF) chicks were investigated by spread-plate colony count method and cell viability assay. Changes in levels of microtubule-assocd. protein light chain 3 (LC3), a widely used marker for autophagosomes, were analyzed by immunoblotting, immunofluorescence, and electron microscopy. Reactive oxygen species (ROS) prodn. was also assessed in vitro. High-throughput RNA sequencing was used to investigate the effects of IONzymes on liver transcriptome of S.Enteritidis-infected chicks. We demonstrated that IONzymes had high biocompatibility with cultured LMH cells and chickens, which significantly inhibited intracellular S.Enteritidis survival in vitro and in vivo. In addn., co-localization of IONzymes with S.Enteritidis were obsd. in autophagic vacuoles of LMH cells and liver of chickens infected by S.Enteritidis, indicating that IONzymes mediated antibacterial reaction of S.Enteritidis with autophagic pathway. We found ROS level was significantly increased in infected LMH cells treated with IONzymes, which might enhance the autophagic elimination of intracellular S.Enteritidis. Moreover, orally administered IONzymes decreased S.Enteritidis organ invasion of the liver and prevented pathol. lesions in a chicken-infection model. Non-target transcriptomic profiling also discovered IONzymes could change hepatic oxidn.-redn. and autophagy related gene expressions in the S.Enteritidis infected chickens. These data suggest that IONzymes can increase ROS levels to promote the antibacterial effects of acid autophagic vacuoles, and thus suppress the establishment and survival of invading intracellular S.Enteritidis. As a result, IONzymes may be a novel alternative to current antibiotics for the control of intractable S.Enteritidis infections.
- 39Guo S. Guo L. Unraveling the Multi-Enzyme-Like Activities of Iron Oxide Nanozyme via a First-Principles Microkinetic Study J. Phys. Chem. C 2019 123 50 30318 3033439Unraveling the Multi-Enzyme-Like Activities of Iron Oxide Nanozyme via a First-Principles Microkinetic StudyGuo, Sibei; Guo, LingJournal of Physical Chemistry C (2019), 123 (50), 30318-30334CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)The design and construction of efficient artificial enzyme-mimicking nanomaterials, nanozymes, is highly desirable because of their high stability and low cost. Recent studies have demonstrated iron oxide nanomaterials as multifunctional nanozymes. However, the catalytic mechanism remains unclear. Herein we have combined d. functional theory calcns. with microkinetic modeling to demonstrate (Fe3O4)n (n = 1 to 2) exhibiting the intrinsic activity of mimicking enzymes of catalases (CATs), superoxide dismutases (SODs), and peroxidases (PODs). Their catalytic activities are facilitated by the close proximity of undercoordinated, tunable Fe/O pairs on the (Fe3O4)n surfaces. The (Fe3O4)n (n = 1 to 2) with different morphologies and sizes exhibited different catalytic activities on the order of Fe3O4 > (Fe3O4)2. Three possible reaction mechanisms of CAT-like activity (i.e., base-like dissociative mechanism, acid-like dissociative mechanism, and bihydrogen peroxide associative mechanism) and two possible reaction mechanisms of SOD-like activity (i.e., Langmuir-Hinshelwood mechanism and Eley-Rideal mechanism) are systematically explored based on min. energy path calcns. It is identified that the acid-like dissociative mechanism and the Langmuir-Hinshelwood mechanism are the energetically most favorable pathways, which is proved by the anal. of the rate-detg. step, the energetic span model, and the rate const. The degree of turnover frequency control (XTOF) of the species in the mechanism is calcd. and identifies the rate-controlling intermediates and transition states (i.e., those with the highest XTOF), which are used as descriptors to modify and improve the (Fe3O4)n catalysts. This study should not only aid our understanding of Fe3O4 artificial enzymes from at. level but also facilitate the design and construction of other types of target-specific artificial enzymes.
- 40Shen X. Wang Z. Gao X. Zhao Y. Density Functional Theory-Based Method to Predict the Activities of Nanomaterials as Peroxidase Mimics ACS Catal. 2020 10 21 12657 1266540Density Functional Theory-Based Method to Predict the Activities of Nanomaterials as Peroxidase MimicsShen, Xiaomei; Wang, Zhenzhen; Gao, Xingfa; Zhao, YuliangACS Catalysis (2020), 10 (21), 12657-12665CODEN: ACCACS; ISSN:2155-5435. (American Chemical Society)A wide variety of nanomaterials possess peroxidase-like catalytic activities and show promise as cost-effective and versatile replacements for natural peroxidases. However, a universal tool for predicting the activities of these materials is still lacking, thus hindering the efficient discovery of nanomaterials as peroxidase mimics. Here, we use d. functional theory calcns. to reveal the peroxidase-mimetic mechanisms for a series of iron-oxide nanosurfaces, and we derive a volcano-shaped plot of catalytic activity as a function of simple energy-based descriptors. The activity curves and the descriptors can be used to predict peroxidase-like activities for not only iron oxides but also other nanomaterials that share similar catalytic mechanisms. The results demonstrate that the method developed herein can systematically predict the peroxidase-like activities of nanomaterials and thus is expected to be of use for computer-aided design of nanomaterial-based peroxidase mimics.
- 41Chance B. Oshino N. Kinetics and mechanisms of catalase in peroxisomes of the mitochondrial fraction Biochem. J. 1971 122 2 225 23341Kinetics and mechanisma of catalase in peroxisomes of the mitochondrial fractionChance, Britton; Oshino, N.Biochemical Journal (1971), 122 (2), 225-33CODEN: BIJOAK; ISSN:0264-6021.The primary intermediate of catalase and H2O2 was identified and investigated in peroxisome-rich mitochondrial fractions of rat liver. On the basis of kinetic consts. detd. in vitro, it is possible to calc. with reasonable precision the mol. statistics of catalase action in the peroxisomes. The endogenous H2O2 generation is adequate to sustain a concn. of the catalase intermediate of 60-70% of the H2O2 satn. value. Total amt. of catalase corresponds to 0.12-0.15 nmole of heme Fe/mg protein. In State 1 the rate of H2O2 generation corresponds to 0.9 nmole/min/mg protein or 5% of the mitochondrial respiratory rate in State 4. Partial satn. of the catalase intermediate with H2O2 in the mitochondrial fraction suggests its significant peroxidatic activity towards its endogenous H donor. A variation of this value from 0.3 in State 4 to 0 under anaerobic conditions is obsd. For a particular prepn. the H2O2 generation rate in the substrate-supplemented State 4 corresponds to 0.17/sec, the H2O2 concn. to 2.5mM and the H-donor concn. (in terms of EtOH) to 0.12mM. The reaction is 70% peroxidatic and 30% catalatic. A coordinated production of both oxidizing and reducing substrates for catalase in the mitochondrial fraction is suggested by a 2.2-fold increase of H2O2 generation and a 3-fold increase in H-donor generation in the State 1 to State 4 transition.
- 42Hwang I. Uddin M. J. Pak E. S. Kang H. Jin E.-J. Jo S. Kang D. Lee H. Ha H. The impaired redox balance in peroxisomes of catalase knockout mice accelerates nonalcoholic fatty liver disease through endoplasmic reticulum stress Free Radical Biol. Med. 2020 148 22 3242The impaired redox balance in peroxisomes of catalase knockout mice accelerates nonalcoholic fatty liver disease through endoplasmic reticulum stressHwang, Inah; Uddin, Md. Jamal; Pak, Eun Seon; Kang, Hyeji; Jin, Eun-Jung; Jo, Suin; Kang, Dongmin; Lee, Hyukjin; Ha, HunjooFree Radical Biology & Medicine (2020), 148 (), 22-32CODEN: FRBMEH; ISSN:0891-5849. (Elsevier B.V.)Peroxisomes are essential organelles for maintaining the homeostasis of lipids and reactive oxygen species (ROS). While oxidative stress-induced endoplasmic reticulum (ER) stress plays an important role in nonalcoholic fatty liver disease (NAFLD), the role of peroxisomes in ROS-mediated ER stress in the development of NAFLD remains elusive. We investigated whether an impaired peroxisomal redox state accelerates NAFLD by activating ER stress by inhibiting catalase, an antioxidant expressed exclusively in peroxisomes. Wild-type (WT) and catalase knockout (CKO) mice were fed either a normal diet or a high-fat diet (HFD) for 11 wk. HFD-induced phenotype changes and liver injury accompanied by ER stress and peroxisomal dysfunction were accelerated in CKO mice compared to WT mice. Interestingly, these changes were also significantly increased in CKO mice fed a normal diet. Inhibition of catalase by 3-aminotriazole in hepatocytes resulted in the following effects: (i) increased peroxisomal H2O2 levels as measured by a peroxisome-targeted H2O2 probe (HyPer-P); (ii) elevated intracellular ROS; (iii) decreased peroxisomal biogenesis; (iv) activated ER stress; (v) induced lipogenic genes and neutral lipid accumulation; and (vi) suppressed insulin signaling cascade assocd. with JNK activation. N-acetylcysteine or 4-phenylbutyric acid effectively prevented those alterations. These results suggest that a redox imbalance in peroxisomes perturbs cellular metab. through the activation of ER stress in the liver.
- 43Ighodaro O. Akinloye O. First line defence antioxidants-superoxide dismutase (SOD) catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid Alexandria J.Med. 2018 54 4 287 293There is no corresponding record for this reference.
- 44Fita I. Rossmann M. G. The active center of catalase J. Mol. Biol. 1985 185 1 21 3744The active center of catalaseFita, Ignacio; Rossmann, Michael G.Journal of Molecular Biology (1985), 185 (1), 21-37CODEN: JMOBAK; ISSN:0022-2836.The refined structure of beef liver catalase is examd. with regard to possible catalytic mechanisms. The distal side of the deeply buried heme pocket is connected with the surface of the mol. by 1 (or possibly 2) channel(s). The electron d. representing the heme group, in each of the 2 crystallog. independent subunits, is consistent with degrdn. of the porphyrin rings. The heme group appears to be buckled, reflecting the high content of bile pigment in liver catalase. The spatial organization on the proximal side (where the 5th ligand of the Fe is located) shows an elaborate network of interactions; the distal side contains the substrate pocket. The limited space in this region severely constrains possible substrate positions and orientations. The Nδ atom of essential histidine-74 H bonds with Oγ of serine-13, which in turn H bonds to an H2O mol. assocd. with the propionic carbonylic group of pyrrole III. These interactions are also visible in the refined structure of Penicillium vitale catalase. Model building suggests a pathway for a catalase mechanism (compd. I formation, as well as catalase and peroxidase reactions). There are some similarities in compd. I formation of catalase and cytochrome c peroxidase.
- 45Obinger C. Maj M. Nicholls P. Loewen P. Activity, Peroxide Compound Formation, and Heme d Synthesis in Escherichia coli HPII Catalase Arch. Biochem. Biophys. 1997 342 1 58 6745Activity, peroxide compound formation, and heme d synthesis in Escherichia coli HPII catalaseObinger, C.; Maj, M.; Nicholls, P.; Loewen, P.Archives of Biochemistry and Biophysics (1997), 342 (1), 58-67CODEN: ABBIA4; ISSN:0003-9861. (Academic)Wild-type Escherichia coli HPII catalase (heme d contg.) has 15% the activity of beef liver enzyme per heme. The rate const. for compd. I formation with H2O2 is 1.3×106 M-1 s-1. HPII compd. I reacts with H2O2 to form O2 with a rate const. of 1.8×106 M-1 s-1. Forty percent of HPII hemes are in the compd. I state during turnover. Compd. I is reduced by ethanol and formate at rates of 5 and 13 M-1 s-1 (pH 7.0), resp. Incubation of HPII compd. I with ferrocyanide and ascorbate does not form a compd. II species. Mutation of His128 to alanine or asparagine gives inactive protoheme proteins. Mutation of Asn-201 gives partially active heme d forms. Asn201Ala has 24%, Asn201Asp 10%, and Asn201Gln 0.4% of wild-type activity. Asn201His contains protoheme when isolated and converts this via protoheme compd. I to a heme d species. Both distal heme cavity residues His128 and Asn201 are implicated in catalytic activity, compd. I formation, and in situ heme d biosynthesis. HPII Asn201, like the corresponding residue in protoheme catalases, may promote H+ transfer to His128 imidazole, facilitating (i) peroxide anion binding to heme and (ii) stabilization of a transition state for heterolytic cleavage of the O-O bond.
- 46Zhang R. Chen L. Liang Q. Xi J. Zhao H. Jin Y. Gao X. Yan X. Gao L. Fan K. Unveiling the active sites on ferrihydrite with apparent catalase-like activity for potentiating radiotherapy Nano Today 2021 41 10131746Unveiling the active sites on ferrihydrite with apparent catalase-like activity for potentiating radiotherapyZhang, Ruofei; Chen, Lei; Liang, Qian; Xi, Juqun; Zhao, Hanqing; Jin, Yiliang; Gao, Xingfa; Yan, Xiyun; Gao, Lizeng; Fan, KelongNano Today (2021), 41 (), 101317CODEN: NTAOCG; ISSN:1748-0132. (Elsevier Ltd.)The use of catalase-like nanozymes in relieving tumor hypoxia has been a promising strategy for adjuvant radiotherapy, yet their catalytic mechanism and toxic potential remain elusive. While most studies on catalase-like mechanisms focus on the substrates and products in the catalytic process, little is reported to analyze the dominating surface structure for catalytic activity of nanozymes. Here, we reported the catalase-like activity of ferrihydrite and revealed its structure-activity relationship. Among the ten main forms of iron oxide nanomaterials, ferrihydrite, esp. 2-line ferrihydrite, exhibited the highest catalase-like activity. Importantly, the structure-activity fitting and d. functional anal. revealed that the abundant surface iron-assocd. hydroxyl groups dominantly affect the catalase-like activity of ferrihydrites. In addn., the ferrihydrite exhibited no peroxidase-like and superoxide dismutase-like activities, and constantly catalyzed the decompn. of H2O2 to release O2 in the pH range of 4.0-8.7. With the help of steady and single catalase-like activity in the physiol. environment, ferrihydrite safely and effectively catalyzed the H2O2 in tumor microenvironment to in-situ produce O2 and significantly enhanced the therapeutic effect of radiotherapy. Moreover, ferrihydrite exhibited excellent biosafety and biodegradability. These findings demonstrate the great potential of ferrihydrite as a biocompatible catalase-like nanozyme for relieving hypoxia in cancer therapy.
- 47Srinivasan B. A guide to the Michaelis–Menten equation: steady state and beyond FEBS J. 2021 1 13There is no corresponding record for this reference.
- 48Fan H. M. Yi J. B. Yang Y. Kho K. W. Tan H. R. Shen Z. X. Ding J. Sun X. W. Olivo M. C. Feng Y. P. Single-Crystalline MFe2O4 Nanotubes/Nanorings Synthesized by Thermal Transformation Process for Biological Applications ACS Nano 2009 3 9 2798 280848Single-Crystalline MFe2O4 Nanotubes/Nanorings Synthesized by Thermal Transformation Process for Biological ApplicationsFan, Hai-Ming; Yi, Jia-Bao; Yang, Yi; Kho, Kiang-Wei; Tan, Hui-Ru; Shen, Ze-Xiang; Ding, Jun; Sun, Xiao-Wei; Olivo, Malini Carolene; Feng, Yuan-PingACS Nano (2009), 3 (9), 2798-2808CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)The authors report a general thermal transformation approach to synthesize single-cryst. magnetic transition metal oxides nanotubes/nanorings including magnetite Fe3O4, maghemite γ-Fe2O3, and ferrites MFe2O4 (M = Co, Mn, Ni, Cu) using hematite α-Fe2O3 nanotubes/nanorings template. While the straightforward redn. or redn.-oxides process was employed to produce Fe3O4 and γ-Fe2O3, the α-Fe2O3/M(OH)2 core/shell nanostructure was used as precursor to prep. MFe2O4 nanotubes via MFe2O4-x (0 < x < 1) intermediate. The transformed ferrites nanocrystals retain the hollow structure and single-cryst. nature of the original templates. However, the crystallog. orientation-relationships of cubic spinel ferrites and trigonal hematite show strong correlation with their morphologies. The hollow-structured MFe2O4 nanocrystals with tunable size, shape, and compn. have exhibited unique magnetic properties. Moreover, they have been demonstrated as a highly effective peroxidase mimic catalysts for lab. immunoassays or as a universal nanocapsules hybridized with luminescent QDs for magnetic sepn. and optical probe of lung cancer cells, suggesting that these biocompatible magnetic nanotubes/nanorings have great potential in biomedicine and biomagnetic applications.
- 49Hao J. H. Zhang Z. Yang W. S. Lu B. P. Ke X. Zhang B. L. Tang J. L. In situ controllable growth of CoFe2O4 ferrite nanocubes on graphene for colorimetric detection of hydrogen peroxide J. Mater. Chem. A 2013 1 13 4352 435749In situ controllable growth of CoFe2O4 ferrite nanocubes on graphene for colorimetric detection of hydrogen peroxideHao, Jinhui; Zhang, Zhe; Yang, Wenshu; Lu, Baoping; Ke, Xi; Zhang, Bailin; Tang, JilinJournal of Materials Chemistry A: Materials for Energy and Sustainability (2013), 1 (13), 4352-4357CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)A facile method is proposed for the synthesis of cubic CoFe2O4 ferrite-reduced graphene oxide nanocomposite sheets (rGO-CFs), using poly(N-vinyl-2-pyrrolidone) as the reductant and stabilizer. The rGO-CFs functioned as efficient peroxidase mimetics and were successfully applied for colorimetric assay. The morphol. and compn. of the rGO-CFs were characterized by TEM, XPS, and x-ray diffraction. Significantly, the peroxidase-like activity of the rGO-CFs followed typical Michaelis-Menten kinetics and showed a good affinity to 3,3',5,5'-tetra-Me benzidine. Under optimum conditions, the colorimetric assay showed a lower detection limit (S/N = 3) of 0.3 μM when compared with that of other nanoparticle based colorimetric assays. Furthermore, the cubic nanostructured rGO-CFs exhibited better stability than horseradish peroxidase when they were exposed to solns. with different solvents and temps. These excellent properties made the cubic nanostructured rGO-CFs an ideal candidate for a wide range of potential applications as peroxidase mimetics.
- 50Zhang Z. J. Zhang X. H. Liu B. W. Liu J. W. Molecular Imprinting on Inorganic Nanozymes for Hundred-fold Enzyme Specificity J. Am. Chem. Soc. 2017 139 15 5412 541950Molecular Imprinting on Inorganic Nanozymes for Hundred-fold Enzyme SpecificityZhang, Zijie; Zhang, Xiaohan; Liu, Biwu; Liu, JuewenJournal of the American Chemical Society (2017), 139 (15), 5412-5419CODEN: JACSAT; ISSN:0002-7863. (American Chemical Society)Enzyme-mimicking nanomaterials (nanozymes) are more cost-effective and robust than protein enzymes, but they lack specificity. Herein, molecularly imprinted polymers (MIPs) were grown on Fe3O4 nanozymes with peroxidase-like activity to create substrate binding pockets. Electron microscopy confirmed a shell of nanogel. By imprinting with an adsorbed substrate, moderate specificity was achieved with neutral monomers. Further introducing charged monomers led to nearly 100-fold specificity for the imprinted substrate over the non-imprinted compared to that of bare Fe3O4. Selective substrate binding was further confirmed by isothermal titrn. calorimetry. The same method was also successfully applied for imprinting on gold nanoparticles (peroxidase mimics) and nanoceria (oxidase mimics). Mol. imprinting furthers the functional enzyme mimicking aspect of nanozymes, and such hybrid materials will find applications in biosensor development, sepn., environmental remediation, and drug delivery.
- 51Huo M. F. Wang L. Y. Chen Y. Shi J. L. Tumor-selective catalytic nanomedicine by nanocatalyst delivery Nat. Commun. 2017 8 35751Tumor-selective catalytic nanomedicine by nanocatalyst deliveryHuo Minfeng; Wang Liying; Chen Yu; Shi Jianlin; Huo Minfeng; Wang Liying; Wang LiyingNature communications (2017), 8 (1), 357 ISSN:.Tumor cells metabolize in distinct pathways compared with most normal tissue cells. The resulting tumor microenvironment would provide characteristic physiochemical conditions for selective tumor modalities. Here we introduce a concept of sequential catalytic nanomedicine for efficient tumor therapy by designing and delivering biocompatible nanocatalysts into tumor sites. Natural glucose oxidase (GOD, enzyme catalyst) and ultrasmall Fe3O4 nanoparticles (inorganic nanozyme, Fenton reaction catalyst) have been integrated into the large pore-sized and biodegradable dendritic silica nanoparticles to fabricate the sequential nanocatalyst. GOD in sequential nanocatalyst could effectively deplete glucose in tumor cells, and meanwhile produce a considerable amount of H2O2 for subsequent Fenton-like reaction catalyzed by Fe3O4 nanoparticles in response to mild acidic tumor microenvironment. Highly toxic hydroxyl radicals are generated through these sequential catalytic reactions to trigger the apoptosis and death of tumor cells. The current work manifests a proof of concept of catalytic nanomedicine by approaching selectivity and efficiency concurrently for tumor therapeutics.The specific metabolism of cancer cells may allow for selective tumor therapeutics. Here, the authors show that a suitable combination of an enzyme and iron nanoparticles loaded on dendritic silica induces apoptosis of cancer cells in response to the glucose-reliant and mild acidic microenvironment.
- 52Wei F. Cui X. Y. Wang Z. Dong C. C. Li J. D. Han X. J. Recoverable peroxidase-like Fe3O4@MoS2-Ag nanozyme with enhanced antibacterial ability Chem. Eng. J. 2021 408 12724052Recoverable peroxidase-like Fe3O4@MoS2-Ag nanozyme with enhanced antibacterial abilityWei, Feng; Cui, Xinyu; Wang, Zhao; Dong, Changchang; Li, Jiadong; Han, XiaojunChemical Engineering Journal (Amsterdam, Netherlands) (2021), 408 (), 127240CODEN: CMEJAJ; ISSN:1385-8947. (Elsevier B.V.)Antibacterial agents with enzyme-like properties and bacteria-binding ability have provided an alternative method to efficiently disinfect drug-resistance microorganism. Herein, a Fe3O4@MoS2-Ag nanozyme with defect-rich rough surface was constructed by a simple hydrothermal method and in-situ photodeposition of Ag nanoparticles. The nanozyme exhibited good antibacterial performance against E. coli (∼69.4%) by the generated ROS and released Ag+, while the nanozyme could further achieve an excellent synergistic disinfection (∼100%) by combining with the near-IR photothermal property of Fe3O4@MoS2-Ag. The antibacterial mechanism study showed that the antibacterial process was detd. by the collaborative work of peroxidase-like activity, photothermal effect and leakage of Ag+. The defect-rich rough surface of MoS2 layers facilitated the capture of bacteria, which enhanced the accurate and rapid attack of ·OH and Ag+ to the membrane of E. coli with the assistance of local hyperthermia. This method showed broad-spectrum antibacterial performance against Gram-neg. bacteria, Gram-pos. bacteria, drug-resistant bacteria and fungal bacteria. Meanwhile, the magnetism of Fe3O4 was used to recycle the nanozyme. This work showed great potential of engineered nanozymes for efficient disinfection treatment.
- 53Peng F. F. Zhang Y. Gu N. Size-dependent peroxidase-like catalytic activity of Fe3O4 nanoparticles Chin. Chem. Lett. 2008 19 6 730 73353Size-dependent peroxidase-like catalytic activity of Fe3O4 nanoparticlesPeng, Fang Fang; Zhang, Yu; Gu, NingChinese Chemical Letters (2008), 19 (6), 730-733CODEN: CCLEE7; ISSN:1001-8417. (Elsevier B.V.)Peroxidase-like catalytic properties of Fe3O4 nanoparticles (NPs) with three different sizes, synthesized by chem. copptn. and sol-gel methods, were investigated by UV-vis spectrum anal. By comparing Fe3O4 NPs with av. diams. of 11, 20, and 150 nm, we found that the catalytic activity increases with the reduced nanoparticle size. The electrochem. method to characterize the catalytic activity of Fe3O4 NPs using the response currents of the reaction product and substrate was also developed.
- 54Zuo X. L. Peng C. Huang Q. Song S. P. Wang L. H. Li D. Fan C. H. Design of a Carbon Nanotube/Magnetic Nanoparticle-Based Peroxidase-Like Nanocomplex and Its Application for Highly Efficient Catalytic Oxidation of Phenols Nano Res. 2009 2 8 617 62354Design of a carbon nanotube/magnetic nanoparticle-based peroxidase-like nanocomplex and its application for highly efficient catalytic oxidation of phenolsZuo, Xiaolei; Peng, Cheng; Huang, Qing; Song, Shiping; Wang, Lihua; Li, Di; Fan, ChunhaiNano Research (2009), 2 (8), 617-623CODEN: NRAEB5; ISSN:1998-0124. (Springer)We report a novel nanotechnol.-based approach for the highly efficient catalytic oxidn. of phenols and their removal from wastewater. We use a nanocomplex made of multi-walled carbon nanotubes (MWNTs) and magnetic nanoparticles (MNPs). This nanocomplex retains the magnetic properties of individual MNPs and can be effectively sepd. under an external magnetic field. More importantly, the formation of the nanocomplex enhances the intrinsic peroxidase-like activity of the MNPs that can catalyze the redn. of hydrogen peroxide (H2O2). Significantly, in the presence of H2O2, this nanocomplex catalyzes the oxidn. of phenols with high efficiency, generating insol. polyarom. products that can be readily sepd. from water.
- 55Shi W. B. Zhang X. D. He S. H. Huang Y. M. CoFe2O4 magnetic nanoparticles as a peroxidase mimic mediated chemiluminescence for hydrogen peroxide and glucose Chem. Commun. 2011 47 38 10785 1078755CoFe2O4 magnetic nanoparticles as a peroxidase mimic mediated chemiluminescence for hydrogen peroxide and glucoseShi, Wenbing; Zhang, Xiaodan; He, Shaohui; Huang, YumingChemical Communications (Cambridge, United Kingdom) (2011), 47 (38), 10785-10787CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)This communication presents a new peroxidase mimic of CoFe2O4 nanoparticles evaluated by the luminol-based chemiluminescent (CL) reaction. This offers a new method for evaluation and screening of the nanoparticles-based enzyme mimetics.
- 56Chaudhari K. N. Chaudhari N. K. Yu J.-S. Peroxidase mimic activity of hematite iron oxides (α-Fe2O3) with different nanostructures Catal. Sci. Technol. 2012 2 1 119 12456Peroxidase mimic activity of hematite iron oxides (α-Fe2O3) with different nanostructuresChaudhari, Kiran N.; Chaudhari, Nitin K.; Yu, Jong-SungCatalysis Science & Technology (2012), 2 (1), 119-124CODEN: CSTAGD; ISSN:2044-4753. (Royal Society of Chemistry)Enzyme mimics have garnered considerable attention as they can overcome some serious disadvantages assocd. with the natural enzymes. In recently developed sphere and rod shaped iron oxide peroxidase mimic nanoparticles, the influence of phys. parameters such as shape, size and surface area on the catalytic performance was not clearly demonstrated. In order to better understand the influence of phys. parameters on the enzyme mimic activity of iron oxide nanoparticles, the present study was initiated using three different shaped hematite α-Fe2O3 nanostructures, particularly hexagonal prism, cube-like and rods as model systems. A comparative account of kinetic parameters (Km, Vmax and Kcat) of the peroxidase mimic activity by the various α-Fe2O3 nanostructures indicated that the enzymic potential of these nanoparticles increased from hexagonal prism to rods, via cube-like, suggesting that one-dimensional particles act as a more efficient enzyme mimic system compared to their multi-dimensional counterparts. Surface area is likely to be a key phys. aspect responsible for the enzyme mimic activity. Interestingly, however, particles with lower surface area showed better catalytic performance in the case of one-dimensional rod structure. Upon further anal. of the one-dimensional rods, addnl. phys. properties such as porosity and pore shape also seem to have a significant contribution to their catalytic activity.
- 57Ma M. Xie J. Zhang Y. Chen Z. P. Gu N. Fe3O4@Pt nanoparticles with enhanced peroxidase-like catalytic activity Mater. Lett. 2013 105 36 3957Fe3O4@Pt nanoparticles with enhanced peroxidase-like catalytic activityMa, Ming; Xie, Jun; Zhang, Yu; Chen, Zhongping; Gu, NingMaterials Letters (2013), 105 (), 36-39CODEN: MLETDJ; ISSN:0167-577X. (Elsevier B.V.)Pt modified Fe3O4 magnetic nanoparticles (Fe3O4@Pt NPs) were synthesized by a simple method. The morphol. and crystal structure of the as-prepd. nanocomposite were characterized by TEM, XRD and EDS. The peroxidase-like catalytic activity of the synthesized nanoparticles was also investigated. Catalysis was found to follow Michaelis-Menten kinetics. The calcd. kinetic parameters of the Fe3O4@Pt NPs show that they exhibited strong affinity with substrates and enhanced catalytic activity than that of Fe3O4 nanoparticles, suggesting a higher peroxidase-like activity.
- 58Sun H. Y. Jiao X. L. Han Y. Y. Jiang Z. Chen D. R. Synthesis of Fe3O4-Au Nanocomposites with Enhanced Peroxidase-Like Activity Eur. J. Inorg. Chem. 2013 1 109 11458Synthesis of Fe3O4-Au Nanocomposites with Enhanced Peroxidase-Like ActivitySun, Haiyan; Jiao, Xiuling; Han, Yanyang; Jiang, Zhen; Chen, DairongEuropean Journal of Inorganic Chemistry (2013), 2013 (1), 109-114CODEN: EJICFO; ISSN:1434-1948. (Wiley-VCH Verlag GmbH & Co. KGaA)Sphere-like Fe3O4 aggregates were solvothermally prepd. with ethylene glycol, NaOAc and FeCl3·6H2O as raw materials. The sphere-like Fe3O4 aggregates provided heterogeneous growth sites for Au nanoparticles. These were obtained by redn. of HAuCl4 by Na citrate under mild reaction conditions and the Fe3O4-Au nanocomposites were subsequently formed. The peroxidase-like activity of nanocomposites was studied with H2O2 and 3,3',5,5'-tetramethylbenzidine as substrates. Fe3O4-Au nanocomposites exhibited better catalytic activity than pure Fe3O4 aggregates, mainly resulting from the special electronic structure at the interfaces between the sphere-like Fe3O4 aggregates and the Au nanoparticles.
- 59Song L. N. Huang C. Zhang W. Ma M. Chen Z. W. Gu N. Zhang Y. Graphene oxide-based Fe2O3 hybrid enzyme mimetic with enhanced peroxidase and catalase-like activities Colloids Surf., A 2016 506 747 75559Graphene oxide-based Fe2O3 hybrid enzyme mimetic with enhanced peroxidase and catalase-like activitiesSong, Lina; Huang, Chao; Zhang, Wei; Ma, Ming; Chen, Zhongwen; Gu, Ning; Zhang, YuColloids and Surfaces, A: Physicochemical and Engineering Aspects (2016), 506 (), 747-755CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)Graphene based materials are widely used in energy conversation and catalytic reaction due to their rapid electron transfer capacity and large surface area. Herein, graphene Fe2O3 (GO-Fe2O3) hybrids with enhanced peroxidase-like activity were fabricated. Enhanced peroxidase-like activity strongly depending on pH, temp. and hybrid concn. was obsd. and the peroxidase-like behavior fits well the Michaelis-Menten kinetic model. Free radicals, such as ·OH and O-2·, as intermediates in the hybrid-H2O2 reaction system were directly demonstrated by ESR (ESR) technique and the selective radical inhibition expts. Furthermore, it was found that the addn. of TMB rapidly consumed ·OH and O-2·, and subsequently leaded to the formation of blue TMB radical confirmed by the ESR and absorption spectroscopy. Besides acting as mimetic peroxidase in acidic buffer, the GO-Fe2O3 hybrids also displayed the enhanced catalase-like activity in neutral and alk. buffers compared with individual Fe2O3 nanoparticles and GO sheet, resp. The H2O2 decompn. catalyzed by the hybrids was demonstrated by ESR and the generated O2 was measured using the dissolved oxygen electrode. All the results above demonstrate that the GO-Fe2O3 hybrids are a kind of effective pH-dependent mimetic dual-enzyme.
- 60Li J. Cai J. H. Jia H. M. Zhang L. X. Lei Y. He W. W. Yin J. J. Formation of iron oxide/Pd hybrid nanostructures with enhanced peroxidase-like activity and catalytic reduction of 4-nitrophenol J. Environ. Sci. Health, Part C: Environ. Carcinog. Rev. 2017 35 3 159 17260Formation of iron oxide/Pd hybrid nanostructures with enhanced peroxidase-like activity and catalytic reduction of 4-nitrophenolLi, Jing; Cai, Junhui; Jia, Huimin; Zhang, Lixia; Lei, Yan; He, Weiwei; Yin, Jun-JieJournal of Environmental Science and Health, Part C: Environmental Carcinogenesis & Ecotoxicology Reviews (2017), 35 (3), 159-172CODEN: JESHA2; ISSN:1532-4095. (Taylor & Francis, Inc.)Iron oxide/Pd hybrid nanostructures with controllable Pd loading from 0.05 to 1.0 (calcd. as Pd/Fe molar ratio) have been synthesized by chem. redn. of Pd2+ on iron oxide particles. The combination of iron oxide and Pd exhibits enhanced peroxidase-like activity and catalytic activity toward redn. of 4-nitrophenol. The catalytic enhancements were found to be dependent on the Pd loading amt. as well as the synergistic effect between iron oxide and Pd. These results suggest that iron oxide with unique surface chem. state can be an active supporter and suggest an effective way to design superior hybrid nanostructures for catalytic applications.
- 61Wang Q. Q. Zhang X. P. Huang L. Zhang Z. Q. Dong S. J. One-Pot Synthesis of Fe3O4 Nanoparticle Loaded 3D Porous Graphene Nanocomposites with Enhanced Nanozyme Activity for Glucose Detection ACS Appl. Mater. Interfaces 2017 9 8 7465 747161One-Pot Synthesis of Fe3O4 Nanoparticle Loaded 3D Porous Graphene Nanocomposites with Enhanced Nanozyme Activity for Glucose DetectionWang, Qingqing; Zhang, Xueping; Huang, Liang; Zhang, Zhiquan; Dong, ShaojunACS Applied Materials & Interfaces (2017), 9 (8), 7465-7471CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)A novel one-pot strategy is proposed to fabricate 3D porous graphene (3D GN) decorated with Fe3O4 nanoparticles (Fe3O4 NPs) by using hemin as the iron source. During the process, graphene oxide was simultaneously reduced and self-assembled to form 3D graphene hydrogel, while Fe3O4 NPs synthesized from hemin distributed uniformly on 3D GN. The prepn. process is simple, facile, economical and green. The obtained freeze-dried product (3D GH-5) exhibits outstanding peroxidase-like activity. Compared to the traditional 2D graphene-based nanocomposites, the introduced 3D porous structure dramatically improved the catalytic activity, as well as the catalysis velocity and its affinity for substrate. The high catalytic activity could be ascribed to the forming Fe3O4 NPs and 3D porous graphene structures. Based on its peroxidase-like activity, 3D GH-5 was used for colorimetric detn. of glucose with a low detection limit of 0.8 μM.
- 62Vallabani N. V. S. Karakoti A. S. Singh S. ATP-mediated intrinsic peroxidase-like activity of Fe3O4-based nanozyme: One step detection of blood glucose at physiological pH Colloids Surf., B 2017 153 52 6062ATP-mediated intrinsic peroxidase-like activity of Fe3O4-based nanozyme: One step detection of blood glucose at physiological pHVallabani, N. V. Srikanth; Karakoti, Ajay S.; Singh, SanjayColloids and Surfaces, B: Biointerfaces (2017), 153 (), 52-60CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)Fe3O4 nanoparticles (Fe3O4 NPs), demonstrating peroxidase-like activity has garnered attention in the detection of several biomols., therefore, emerged as an excellent nano-biosensing agent. The intrinsic peroxidase-like activity of Fe3O4 NPs at acidic pH is the fundamental action driving the oxidn. of substrates like TMB, resulting in a colorimetric product formation used in the detection of biomols. Hence, the detection sensitivity essentially depends on the ability of oxidn. by Fe3O4 NPs in presence of H2O2. However, the limited sensitivity and pH condition constraint have been identified as the major drawbacks in the detection of biomols. at physiol. pH. Herein, we report overwhelming of the fundamental limitation of acidic pH and tuning the peroxidase-like activity of Fe3O4 NPs at physiol. pH by using ATP. In presence of ATP, Fe3O4 NPs exhibited enhanced peroxidase-like activity over a wide range of pH and temps. Mechanistically, it was found that the ability of ATP to participate in single electron transfer reaction, through complexation with Fe3O4 NPs, results in the generation of hydroxyl radicals We utilized this ATP-mediated enhanced peroxidase-like activity of Fe3O4 NPs for single step detection of glucose with a colorimetric detection limit of 50μM.
- 63Peng J. Yang Q. Li W. Tan L. Xiao Y. Chen L. Hao Y. Qian Z. Erythrocyte-Membrane-Coated Prussian Blue/Manganese Dioxide Nanoparticles as H2O2-Responsive Oxygen Generators To Enhance Cancer Chemotherapy/Photothermal Therapy ACS Appl. Mater. Interfaces 2017 9 51 44410 4442263Erythrocyte-Membrane-Coated Prussian Blue/Manganese Dioxide Nanoparticles as H2O2-Responsive Oxygen Generators To Enhance Cancer Chemotherapy/Photothermal TherapyPeng, Jinrong; Yang, Qian; Li, Wenting; Tan, Liwei; Xiao, Yao; Chen, Lijuan; Hao, Ying; Qian, ZhiyongACS Applied Materials & Interfaces (2017), 9 (51), 44410-44422CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Because of the nontargeting release of anticancer drugs, conventional chemotherapy results in serious side effects and poor therapeutic outcomes. In addn., hypoxia situation in the tumor microenvironment also promotes the growth and metastasis of tumors. Multifunctional nanocarriers with stimuli-activation and hypoxia-relieving properties can help overcome some of these limitations. In this study, we have constructed a nanocarrier which is named PBMn-DOX@RBC. A Prussian blue/manganese dioxide (PBMn) nanoparticle is used as an oxygen precursor or catalyzer for H2O2 activation, and a red blood cell (RBC) membrane is used to increase the loading capacity of doxorubicin (DOX) and prolong the circulation time in vivo. H2O2 is overproduced in tumor tissues and tumor cells. It can be used as a stimulus to activate drug release. In the presence of H2O2, the hypoxia inside the tumors is relieved by the administration of PBMn-DOX@RBC. The generated oxygen disrupts the RBC coated on the surface of PBMn, which accelerates the release of DOX. RBCs also prolong the circulation time of the nanometer system in vivo. By combining the photothermal therapy (PTT) and chemotherapy, the tumor growth inhibition mediated by PBMn-DOX@RBC is further enhanced. PBMn-DOX@RBC fulfills the demands to relieve tumor hypoxia and enhance cancer chemotherapy/PTT.
- 64Wu L. H. Wan G. P. Hu N. He Z. Y. Shi S. H. Suo Y. R. Wang K. Xu X. F. Tang Y. L. Wang G. Z. Synthesis of Porous CoFe2O4 and Its Application as a Peroxidase Mimetic for Colorimetric Detection of H2O2 and Organic Pollutant Degradation Nanomaterials 2018 8 7 45164Synthesis of porous CoFe2O4 and its application as a peroxidase mimetic for colorimetric detection of H2O2 and organic pollutant degradationWu, Lihong; Wan, Gengping; Hu, Na; He, Zhengyi; Shi, Shaohua; Suo, Yourui; Wang, Kan; Xu, Xuefei; Tang, Yulin; Wang, GuizhenNanomaterials (2018), 8 (7), 451/1-451/16CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Porous CoFe2O4 was prepd. via a simple and controllable method to develop a low-cost, high-efficiency, and good-stability nanozyme. The morphol. and microstructure of the obtained CoFe2O4 was investigated by X-ray diffraction (XRD), XPS, transmission electron microscopy (TEM), high-resoln. TEM (HRTEM), sp. surface area and pore anal., and Raman spectroscopy. The results show that the annealing temp. has an important effect on the crystallinity, grain size, and sp. surface area of CoFe2O4. CoFe2O4 obtained at 300° C (CF300) exhibits the largest surface area (up to 204.1 m2 g-1) and the smallest grain size. The peroxidase-like activity of CoFe2O4 was further verified based on the oxidn. of peroxidase substrate 3,30',5,5'tetramethylbenzidine (TMB) in the presence of H2O2. The best peroxidase-like activity for CF300 should be ascribed to its largest surface area and smallest grain size. On this basis, an effective method of colorimetric detection H2O2 was established. In addn., the porous CoFe2O4 was also used for the catalytic oxidn. of methylene blue (MB), indicating potential applications in pollutant removal and water treatment.
- 65Liu L. Du B. J. Shang C. S. Wang J. Wang E. K. Construction of surface charge-controlled reduced graphene oxide-loaded Fe3O4 and Pt nanohybrid for peroxidase mimic with enhanced catalytic activity Anal. Chim. Acta 2018 1014 77 8465Construction of surface charge-controlled reduced graphene oxide-loaded Fe3O4 and Pt nanohybrid for peroxidase mimic with enhanced catalytic activityLiu, Lan; Du, Baoji; Shang, Changshuai; Wang, Jin; Wang, ErkangAnalytica Chimica Acta (2018), 1014 (), 77-84CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)Hybrid nanomaterials with synergistic effect are highly potential for developing advanced nanozymes. Herein, the authors designed a nanozyme assembled by polyethylenimine (PEI)-protected reduced graphene oxide anchoring Fe oxide (PRGI) and Pt nanoparticle using electrostatic interaction, PRGI/Pt nanohybrid. The different ratio of PRGI nanocomposite and Pt nanoparticle could control PRGI/Pt nanohybrid's surface charge and stability, which detd. PRGI/Pt nanohybrid's catalytic activity. At the mass ratio of 0.8, the as-obtained PRGI/Pt nanohybrid showed the highest catalytic ability, and was better than Pt nanoparticle at different pH and temp., although the PRGI/Pt nanohybrid showed lower affinity for TMB than Pt nanoparticle, which maybe attributed to the fact that PRGI/Pt nanohybrid possessed better product desorption ability or larger contact area. Also, PRGI/Pt nanohybrid showed much higher catalytic activity than the sum of PRGI nanocomposite and Pt nanoparticle, indicating the strong cooperation between PRGI nanocomposite and Pt nanoparticle. This study also provided a new way to conveniently construct nanozyme based on hybrid nanomaterials.
- 66Qiu N. Liu Y. Xiang M. Lu X. M. Yang Q. Guo R. A facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 nanohybrid for highly sensitive and selective detection of p-nitrophenol Sens. Actuators, B 2018 266 86 9466A facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 nanohybrid for highly sensitive and selective detection of p-nitrophenolQiu, Na; Liu, Yan; Xiang, Min; Lu, Ximing; Yang, Qian; Guo, RongSensors and Actuators, B: Chemical (2018), 266 (), 86-94CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)To date, fabricating high sensitive, stable and facile sensing strategy for detection of nitrophenol still remains a challenge in practical applications. This article demonstrates for the first time a facile and stable colorimetric sensor based on three-dimensional graphene/mesoporous Fe3O4 (3D GF/m-Fe3O4) heterogeneous nanozyme. Using a simple hydrothermal procedure, mesoporous Fe3O4 nanoparticles were in situ synthesized on 3D GF. The 3D GF/m-Fe3O4 nanohybrid possesses high peroxidase mimicking activity due to the synergistic effect of 3D GF and m-Fe3O4. Based on the peroxidase-like activity and p-nitrophenol-mediated inhibition controllability, the nanohybrid was successfully adopted in the sensitive and selective sensing detections of p-nitrophenol (PNP) with a low detection limit of 45nM. Moreover, 3D GF/m-Fe3O4 nanohybrid shows to be extremely robust, as it still retains its original response to PNP after being reused for 10 times. The method was applied to the detn. of PNP in spiked lake water and gave good recoveries. With the merits of high sensitivity and selectivity, simplification, recyclability, and excellent stability, this sensing platform not only expands the application of 3D GF and Fe3O4, but also provides an alternative technique to construct robust nanozyme with multiple functionalities for various biol. and environmental applications.
- 67Bhattacharjee R. Tanaka S. Moriam S. Masud M. K. Lin J. J. Alshehri S. M. Ahamad T. Salunkhe R. R. Nguyen N. T. Yamauchi Y. Hossain M. S. A. Shiddiky M. J. A. Porous nanozymes: the peroxidase-mimetic activity of mesoporous iron oxide for the colorimetric and electrochemical detection of global DNA methylation J. Mater. Chem. B 2018 6 29 4783 479167Porous nanozymes: the peroxidase-mimetic activity of mesoporous iron oxide for the colorimetric and electrochemical detection of global DNA methylationBhattacharjee, Ripon; Tanaka, Shunsuke; Moriam, Sofia; Masud, Mostafa Kamal; Lin, Jianjian; Alshehri, Saad M.; Ahamad, Tansir; Salunkhe, Rahul R.; Nguyen, Nam-Trung; Yamauchi, Yusuke; Hossain, Md. Shahriar A.; Shiddiky, Muhammad J. A.Journal of Materials Chemistry B: Materials for Biology and Medicine (2018), 6 (29), 4783-4791CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Nanomaterials (nanozymes) with peroxidase-mimetic activity have been widely used in biosensing platforms as low-cost, relatively stable and prevailing alternatives to natural enzymes. Herein, the authors report on the synthesis and application of the peroxidase-mimetic activity of mesoporous iron oxide (MIO) for the detection of global DNA methylation in colorectal cancer cell lines. The target DNA was extd. and denatured to get ssDNA followed by direct adsorption onto the surface of a bare screen-printed gold electrode (SPGE). A 5-methylcytosine antibody (5mC) functionalized nanomaterial (MIO-5mC) was then used to recognize the methylcytosine groups present on the SPGE. The MIO-5mC conjugates catalyze the TMB soln. in the presence of hydrogen peroxide to give the colorimetric (i.e., naked-eye observation) and electrochem. detection of DNA methylation. The assay could successfully detect as low as 10% difference in the global DNA methylation level in synthetic samples and cell lines with good reproducibility and specificity (%RSD = <5%, for n = 3). This strategy avoids the use of natural enzyme horseradish peroxidase (HRP), traditional PCR based amplification and bisulfite treatment steps that are generally used in many conventional DNA methylation assays. The authors envisage that the assay could be a low-cost platform with great potential for genome-wide DNA methylation anal. in point-of-care applications.
- 68Yang Y. Zhu W. Feng L. Chao Y. Yi X. Dong Z. Yang K. Tan W. Liu Z. Chen M. G-Quadruplex-Based Nanoscale Coordination Polymers to Modulate Tumor Hypoxia and Achieve Nuclear-Targeted Drug Delivery for Enhanced Photodynamic Therapy Nano Lett. 2018 18 11 6867 687568G-Quadruplex-Based Nanoscale Coordination Polymers to Modulate Tumor Hypoxia and Achieve Nuclear-Targeted Drug Delivery for Enhanced Photodynamic TherapyYang, Yu; Zhu, Wenjun; Feng, Liangzhu; Chao, Yu; Yi, Xuan; Dong, Ziliang; Yang, Kai; Tan, Weihong; Liu, Zhuang; Chen, MeiwanNano Letters (2018), 18 (11), 6867-6875CODEN: NALEFD; ISSN:1530-6984. (American Chemical Society)Photodynamic therapy (PDT) is a light-triggered therapy used to kill cancer cells by producing reactive oxygen species (ROS). Herein, a new kind of DNA nanostructure based on the coordination between calcium ions (Ca2+) and AS1411 DNA G quadruplexes to form nanoscale coordination polymers (NCPs) is developed via a simple method. Both chlorine e6 (Ce6), a photosensitizer, and hemin, an iron-contg. porphyrin, can be inserted into the G-quadruplex structure in the obtained NCPs. With further polyethylene glycol (PEG) modification, we obtain Ca-AS1411/Ce6/hemin@pHis-PEG (CACH-PEG) NCP nanostructure that enables the intranuclear transport of photosensitizer Ce6 to generate ROS inside cell nuclei that are the most vulnerable to ROS. Meanwhile, the inhibition of antiapoptotic protein B-cell lymphoma 2 (Bcl-2) expression by AS1411 allows for greatly improved PDT-induced cell apoptosis. Furthermore, the catalase-mimicking DNAzyme function of G-quadruplexes and hemin in those NCPs could decomp. tumor endogenous H2O2 to in situ generate oxygen so as to further enhance PDT by overcoming the hypoxia-assocd. resistance. This work develops a simple yet general method with which to fabricate DNA-based NCPs and presents an interesting concept of a nanoscale drug-delivery system that could achieve the intranuclear delivery of photosensitizers, the down-regulation of anti-apoptotic proteins, and the modulation of the unfavorable tumor microenvironment simultaneously for improved cancer therapy.
- 69Liu Y. Qin Y. L. Zheng Y. L. Qin Y. Cheng M. J. Guo R. A one-pot and modular self-assembly strategy for high-performance organized enzyme cascade bioplatforms based on dual-functionalized protein-PtNP@mesoporous iron oxide hybrid J. Mater. Chem. B 2019 7 1 43 5269A one-pot and modular self-assembly strategy for high-performance organized enzyme cascade bioplatforms based on dual-functionalized protein-PtNP@mesoporous iron oxide hybridLiu, Yan; Qin, Yuling; Zheng, Yuanlin; Qin, Yong; Cheng, Mengjun; Guo, RongJournal of Materials Chemistry B: Materials for Biology and Medicine (2019), 7 (1), 43-52CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Inspired by the delicate structure and prominent efficiency of natural multiple-enzyme systems, combining nanotechnologies such as nanomaterials, self-assemblies, and enzyme mimics is fascinating for the development of next-generation high-performance organized enzyme cascade bioplatforms. In our facile and convenient design, a dual-functionalized β-casein-Pt nanoparticles@mesoporous-Fe3O4 (CM-PtNP@m-Fe3O4) hybrid acts as both a nanozyme with outstanding peroxidase-like activity and a scaffold to immobilize and stabilize a natural oxidase, resulting in a high-performance organized enzyme cascade bioplatform for a one-pot assembly procedure. Owing to special physicochem. surface properties, the multipoint attachment of various interactions between natural enzymes and protein/inorg. hybrids leads to efficient immobilization of the enzyme with retained activity. The proposed cascade bioplatform provides superior cholesterol sensing, including simplicity (one-step detection), reusable enzymes (peroxidase mimic and oxidase), and excellent sensitivity (detection limit, 0.05 μM). To our knowledge, the bioplatform presented in this work shows the highest sensitivity for cholesterol detection among all reported colorimetric methods based on nanozymes. Therefore, the highly rationally designed protein/inorg. hybrid and dual-functional strategy used in this study will provide a facile one-pot and effective high-performance organized enzyme cascade bioplatform with potential applications in biosensing, biotransformation, decontamination, and biofuel.
- 70Zhang Q. T. Li M. F. Guo C. Y. Jia Z. Wan G. C. Wang S. F. Min D. Y. Fe3O4 Nanoparticles Loaded on Lignin Nanoparticles Applied as a Peroxidase Mimic for the Sensitively Colorimetric Detection of H2O2 Nanomaterials 2019 9 2 21070Fe3O4 nanoparticles loaded on lignin nanoparticles applied as a peroxidase mimic for the sensitively Colorimetric Detection of H2O2Zhang, Qingtong; Li, Mingfu; Guo, Chenyan; Jia, Zhuan; Wan, Guangcong; Wang, Shuangfei; Min, DouyongNanomaterials (2019), 9 (2), 210CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)Lignin is the second largest naturally renewable resource and is primarily a byproduct of the pulp and paper industry; however, its inefficient use presents a challenge. In this work, Fe3O4 nanoparticles loaded on lignin nanoparticles (Fe3O4@LNPs) were prepd. by the self-assembly method and it possessed an enhanced peroxidase-like activity. Fe3O4@LNPs catalyzed the oxidn. of 3,30,5,50-tetramethylbenzidine (TMB) in the presence of H2O2 to generate a blue color, was observable by the naked eye. Under the optimal conditions, Fe3O4@LNPs showed the ability of sensitive colorimetric detection of H2O2within a range of 5-100μM and the limit of detection was 2μM. The high catalytic activity of Fe3O4@LNPs allows its prospective use in a wide variety of applications, including clin. diagnosis, food safety, and environmental monitoring.
- 71Zhang Z. J. Li Y. Q. Zhang X. H. Liu J. W. Molecularly imprinted nanozymes with faster catalytic activity and better specificity Nanoscale 2019 11 11 4854 486371Molecularly imprinted nanozymes with faster catalytic activity and better specificityZhang, Zijie; Li, Yuqing; Zhang, Xiaohan; Liu, JuewenNanoscale (2019), 11 (11), 4854-4863CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Nanozymes are nanomaterials mimicking the activity of natural enzymes, while most nanozymes lack substrate specificity. Mol. imprinting on nanozymes provides a simple soln. to this problem, and the catalytic activity is also enhanced. To understand enhanced activity, a surface science approach is taken by dissecting the nanozyme reaction into adsorption of substrates, reaction, and product release. Each step is individually studied using reaction kinetics measurement, dynamic light scattering, UV-vis spectrometry. Enrichment of local substrate concn. due to imprinting is around 8-fold, and increased substrate concn. could contribute to increased activity. Diffusion of the substrate across the imprinted gel layer is studied by a pre-incubation expt., also highlighting the difference between imprinted and non-imprinted gel layers. The activation energy is measured and a substrate-imprinted sample had the lowest activation energy of 13.8 kJ mol-1. Product release is also improved after imprinting as indicated by isothermal titrn. calorimetry using samples resp. imprinted with the substrate and the product. This study has rationalized improved activity and specificity of molecularly imprinted nanozymes and may guide further rational design of such materials.
- 72Tanaka S. Masud M. K. Kaneti Y. V. Shiddiky M. J. A. Fatehmulla A. Aldhafiri A. M. Farooq W. A. Bando Y. Hossain M. S. A. Yamauchi Y. Enhanced Peroxidase Mimetic Activity of Porous Iron Oxide Nanoflakes ChemNanoMat 2019 5 4 506 51372Enhanced Peroxidase Mimetic Activity of Porous Iron Oxide NanoflakesTanaka, Shunsuke; Masud, Mostafa Kamal; Kaneti, Yusuf Valentino; Shiddiky, Muhammad J. A.; Fatehmulla, Amanullah; Aldhafiri, Abdullah M.; Farooq, W. Aslam; Bando, Yoshio; Hossain, Md. Shahriar A.; Yamauchi, YusukeChemNanoMat (2019), 5 (4), 506-513CODEN: CHEMSB; ISSN:2199-692X. (Wiley-VCH Verlag GmbH & Co. KGaA)Porous nanomaterials with superior peroxidase mimetic activity (nanozyme) at room temp. have gained increasing attention as potential alternatives to natural peroxidase enzymes. Herein, we report the application of porous iron oxide nanoflakes (IONFs), synthesized using the combination of solvothermal method and high-temp. calcination as peroxidase nanozyme for the oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The four IONF catalysts possess porous structures with a wide pore size distribution between 2-30 nm and high sp. surface areas around to 200 m2 g-1. The increase of calcination temp. of the IONFs from 250°C to 400°C resulted in a gradual decrease in their sp. surface area and Michaelis-Menten const. (Km) for TMB oxidn. The optimum IONF sample showed a much lower Km at 0.24 mM (towards TMB) compared to natural enzyme horseradish peroxidase (HRP) at 0.434 mM, revealing the promising potential of the as-prepd. IONFs as alternatives to HRP for biosensing applications.
- 73Wang Y. Li H. Guo L. Jiang Q. Liu F. A cobalt-doped iron oxide nanozyme as a highly active peroxidase for renal tumor catalytic therapy RSC Adv. 2019 9 33 18815 1882273A cobalt-doped iron oxide nanozyme as a highly active peroxidase for renal tumor catalytic therapyWang, Yixuan; Li, Hongjun; Guo, Lihua; Jiang, Qi; Liu, FengRSC Advances (2019), 9 (33), 18815-18822CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)The Fe3O4 nanozyme, the first reported nanozyme with intrinsic peroxidase-like activity, has been successfully employed for various diagnostic applications. However, only a few studies have been reported on the therapeutic applications of the Fe3O4 nanozyme partly due to its low affinity to the substrate H2O2. Herein, we report a new strategy for improving the peroxidase-like activity and affinity of the Fe3O4 nanozyme to H2O2 to generate reactive oxygen species (ROS) for kidney tumor catalytic therapy. We showed that cobalt-doped Fe3O4 (Co@Fe3O4) nanozymes possessed stronger peroxidase activity and a 100-fold higher affinity to H2O2 than the Fe3O4 nanozymes. The lysosome localization properties of Co@Fe3O4 enable Co@Fe3O4 to catalyze the decompn. of H2O2 at ultralow doses for the generation of ROS bursts to effectively kill human renal tumor cells both in vitro and in vivo. Moreover, our study provides the first evidence that the Co@Fe3O4 nanozyme is a powerful nanozyme for the generation of ROS bursts upon the addn. of H2O2 at ultralow doses, presenting a potential novel avenue for tumor nanozyme catalytic therapy.
- 74Li S. S. Shang L. Xu B. L. Wang S. H. Gu K. Wu Q. Y. Sun Y. Zhang Q. H. Yang H. L. Zhang F. R. Gu L. Zhang T. R. Liu H. Y. A Nanozyme with Photo-Enhanced Dual Enzyme-Like Activities for Deep Pancreatic Cancer Therapy Angew. Chem., Int. Ed. 2019 58 36 12624 1263174A nanozyme with photo-enhanced dual enzyme-like activities for deep pancreatic cancer therapyLi, Shanshan; Shang, Lu; Xu, Bolong; Wang, Shunhao; Gu, Kai; Wu, Qingyuan; Sun, Yun; Zhang, Qinghua; Yang, Hailong; Zhang, Fengrong; Gu, Lin; Zhang, Tierui; Liu, HuiyuAngewandte Chemie, International Edition (2019), 58 (36), 12624-12631CODEN: ACIEF5; ISSN:1433-7851. (Wiley-VCH Verlag GmbH & Co. KGaA)Nanozymes have attracted extensive interest owing to their high stability, low cost and easy prepn., esp. in the field of cancer therapy. However, the relatively low catalytic activity of nanozymes in the tumor microenvironment (TME) has limited their applications. Herein, we report a novel nanozyme (PtFe@Fe3O4) with dual enzyme-like activities for highly efficient tumor catalytic therapy. PtFe@Fe3O4 shows the intrinsic photothermal effect as well as photo-enhanced peroxidase-like and catalase-like activities in the acidic TME, thereby effectively killing tumor cells and overcoming the tumor hypoxia. Importantly, a possible photo-enhanced synergistic catalytic mechanism of PtFe@Fe3O4 was first disclosed. We believe that this work will advance the development of nanozymes in tumor catalytic therapy.
- 75Huang Y. L. Liang G. Z. Lin T. R. Hou L. Ye F. G. Zhao S. L. Magnetic Cu/Fe3O4@FeOOH with intrinsic HRP-like activity at nearly neutral pH for one-step biosensing Anal. Bioanal. Chem. 2019 411 17 3801 381075Magnetic Cu/Fe3O4@FeOOH with intrinsic HRP-like activity at nearly neutral pH for one-step biosensingHuang, Yuanlin; Liang, Guangzhao; Lin, Tianran; Hou, Li; Ye, Fanggui; Zhao, ShulinAnalytical and Bioanalytical Chemistry (2019), 411 (17), 3801-3810CODEN: ABCNBP; ISSN:1618-2642. (Springer)The convenience of colorimetric sensors is useful for practical applications. In this work, the authors constructed a novel colorimetric sensor with magnetic sepn. ability that can be operated in nearly neutral conditions and achieve one-step detection of metabolites. Magnetic Cu doped Fe3O4@FeOOH magnetic nanocomposite (Cu/Fe3O4@FeOOH) with an oxygen vacancy was prepd. by a one-step self-assembly hydrothermal method, and fully characterized by different methods. The oxygen vacancy generated by the incorporation of Cu2+ cations into the Fe3O4@FeOOH structure was confirmed to be a vital reactive site for enhancing the catalytic activity, which opens up a new way of designing highly efficient enzyme mimics. Benefiting from its inherent horseradish-peroxidase-like activity, a simple and selective enzyme-based colorimetric sensor was developed for one-step detection of H2O2 and cholesterol, and 3,3',5,5'-tetramethylbenzidine was catalyzed by H2O2 to generate a colored product of oxidized 3,3',5,5'-tetramethylbenzidine for signaling. H2O2 and cholesterol can be linearly detected in the same range from 0.01 to 0.4 mmol L-1 with detection limits of 0.0075 mmol L-1 and 0.0082 mmol L-1, resp. The proposed colorimetric sensor has satisfactory reusability, accuracy, and practicability in human serum samples, indicating its potential application for the detection of different metabolites in the fields of life science and anal. science.
- 76Wang J. Huang F. Wang X. Wan Y. Xue Y. Cai N. Chen W. Yu F. Hierarchically structured Fe3O4-doped MnO2 microspheres as an enhanced peroxidase-like catalyst for low limit of detection Process Biochem. 2019 83 35 4376Hierarchically structured Fe3O4-doped MnO2 microspheres as an enhanced peroxidase-like catalyst for low limit of detectionWang, Jianzhi; Huang, Fei; Wang, Xianming; Wan, Yinjia; Xue, Yanan; Cai, Ning; Chen, Weimin; Yu, FaquanProcess Biochemistry (Oxford, United Kingdom) (2019), 83 (), 35-43CODEN: PBCHE5; ISSN:1359-5113. (Elsevier Ltd.)In order to lower the limit of detection of glucose, a peroxidase-like artificial enzyme with elevated catalysis capacity has been achieved. Fe3O4-doped MnO2 microspheres were fabricated through a two-step hydrothermal method for this purpose. TEM revealed that down-sized Fe3O4 nanoparticles were dispersed throughout the urchin-like MnO2 burrs. An addnl. XRD peak beyond Fe3O4 and MnO2 nanoparticles was obsd., indicating a dislocation structure was formed. The defect in structure as well as the synergistic effect would allow extra enzyme ability. Based on the steady-state kinetic analyses and Michaelis-Menten model, the Michaelis-Menten consts. (Km and vmax) were figured out. The results showed that the Fe3O4-MnO2 composite has elevated affinity toward the substrate TMB and H2O2. The limit of detection for glucose was estd. to be 0.75μmol L-1 based on the Fe3O4-MnO2 composite artificial enzyme. The superparamagnetic properties endowed the material easy sepn. The composite of this structure will provide a highly sensitive candidate method for accurate detection of glucose.
- 77Bian B. Liu Q. Y. Yu S. T. Peroxidase mimetic activity of porphyrin modified ZnFe2O4/reduced graphene oxide and its application for colorimetric detection of H2O2 and glutathione Colloids Surf., B 2019 181 567 57577Peroxidase mimetic activity of porphyrin modified ZnFe2O4/reduced graphene oxide and its application for colorimetric detection of H2O2 and glutathioneBian, Bing; Liu, Qingyun; Yu, ShitaoColloids and Surfaces, B: Biointerfaces (2019), 181 (), 567-575CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)Artificial nanoenzymes which can overcome some drawbacks of natural enzymes is a challenging topic in the biosensor field. Herein, we demonstrated 5,10,15,20 tetrakis (4-carboxylpheyl)-porphyrin modified magnetic ZnFe2O4 nanoparticles loaded on the surface of reduced graphene oxide (Por-ZnFe2O4/rGO), which exhibited intrinsic peroxidase-like activity and rapidly oxidized the peroxidase substrate 3, 3', 5, 5'-tetramethylbenzidine (TMB) into a blue product (OxTMB) distinguished by naked eyes. Interestingly, by comparative study of different nanomaterials ZnFe2O4 nanoparticles, ZnFe2O4/rGO and Por-ZnFe2O4, Por-ZnFe2O4/rGO was proved to possess the highest peroxidase-like activity. ESR (ESR) verified the catalytic activity of Por-ZnFe2O4/rGO for H2O2 was due to hydroxyl radical from decompn. of H2O2. Temp. and pH strongly affected the peroxidase-like activity of Por-ZnFe2O4/rGO nanocomposites. Under optimal conditions (pH = 4, 40°C), the constructed sensor based on the catalytic activity of the Por-ZnFe2O4/rGO could be conveniently used for colorimetric detection of H2O2 in the range of 0.7-30μM with the detection limit of 0.54μM. Moreover, the colorimetric sensor based on Por-ZnFe2O4/rGO exhibited a good linear response to glutathione (GHS) in the range of 2-40μM with a low detection limit of 0.76μM. The detection of GHS can be easily realized through the obvious color change by naked eyes without any complicated instrumentation.
- 78Masud M. K. Kim J. Billah M. M. Wood K. Shiddiky M. J. A. Nguyen N. T. Parsapur R. K. Kaneti Y. V. Alshehri A. A. Alghamidi Y. G. Alzahrani K. A. Adharvanachari M. Selvam P. Hossain M. S. A. Yamauchi Y. Nanoarchitectured peroxidase-mimetic nanozymes: mesoporous nanocrystalline alpha- or gamma-iron oxide? J. Mater. Chem. B 2019 7 35 5412 542278Nanoarchitectured peroxidase-mimetic nanozymes: mesoporous nanocrystalline α- or γ-iron oxide?Masud, Mostafa Kamal; Kim, Jeonghun; Billah, Md. Motasim; Wood, Kathleen; Shiddiky, Mohammad J. A.; Nguyen, Nam-Trung; Parsapur, Rajesh Kumar; Kaneti, Yusuf Valentino; Alshehri, Abdulmohsen Ali; Alghamidi, Yousef Gamaan; Alzahrani, Khalid Ahmed; Adharvanachari, Murugulla; Selvam, Parasuraman; Hossain, Md. Shahriar A.; Yamauchi, YusukeJournal of Materials Chemistry B: Materials for Biology and Medicine (2019), 7 (35), 5412-5422CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)Nanozymes (nanoparticles with enzyme-like properties) have attracted considerable attention in recent years owing to their intrinsic enzyme-like properties and broad application in the fields of ELISA based immunoassay and biosensing. Herein, the authors systematically study the influence of crystal phases (γ-Fe2O3 and α-Fe2O3) of mesoporous iron oxide (IO) on their peroxidase mimetic activity. In addn., the authors also demonstrated the applicability of these mesoporous IOs as nanozymes for detecting the glucose biomarker with a limit of detection (LOD) of 0.9 μM. Mesoporous γ-Fe2O3 shows high nanozyme activities (and magnetism) toward the catalytic oxidn. of chromogenic substances, such as 3,3',5,5'-tetramethylbenzidine (TMB) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)-ABTS, as well as for the colorimetric detection of glucose, compared to that of α-Fe2O3. The authors believe that this in-depth study of crystal structure based nanozyme activity will guide designing highly effective nanozymes based on iron oxide nanostructures for chem. sensing, biosensing and environmental remediation.
- 79Yin M. Y. Li S. Wan Y. Q. Feng L. P. Zhao X. T. Zhang S. Liu S. H. Cao P. Wang H. A selective colorimetric strategy for probing dopamine and levodopa through the mussel-inspired enhancement of Fe3O4 catalysis Chem. Commun. 2019 55 80 12008 1201179A selective colorimetric strategy for probing dopamine and levodopa through the mussel-inspired enhancement of Fe3O4 catalysisYin, Mengyuan; Li, Shuai; Wan, Yuqi; Feng, Luping; Zhao, Xiaoting; Zhang, Sheng; Liu, Shuhui; Cao, Peng; Wang, HuaChemical Communications (Cambridge, United Kingdom) (2019), 55 (80), 12008-12011CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)Mussel-inspired enhancement of Fe3O4 catalysis was discovered towards a highly selective and sensitive colorimetric strategy for the magnetic sepn.-based evaluation of dopamine and/or levodopa in urine, in which the specific interaction of bis-catechol-contg. analytes and mesoporous Fe3O4 NPs would form highly stable complexes of bis-catechol-Fe coordination.
- 80Zhu W. Li M. Chen S. Wang C. Lu X. Interfacial engineering regulating the peroxidase-like property of ternary composite nanofibers and their sensing applications Appl. Surf. Sci. 2019 491 138 14680Interfacial engineering regulating the peroxidase-like property of ternary composite nanofibers and their sensing applicationsZhu, Wendong; Li, Meixuan; Chen, Sihui; Wang, Ce; Lu, XiaofengApplied Surface Science (2019), 491 (), 138-146CODEN: ASUSEE; ISSN:0169-4332. (Elsevier B.V.)Interfacial engineering is an efficient and versatile strategy to accelerate the catalytic performance of functional nanomaterials. Herein, the authors have constructed ternary titanium dioxide (TiO2)@molybdenum disulfide (MoS2)/cobalt ferrite (CoFe2O4) composite nanofibers via a two-step hydrothermal reaction. Firstly, MoS2 nanosheets are grown on TiO2 nanofibers, then acting as an interfacial barrier to load ultrafine CoFe2O4 nanoparticles. Thanks to the synergistic effects among the interfacial interactions between MoS2 and TiO2 as well as CoFe2O4 component, the prepd. ternary TiO2@MoS2/CoFe2O4 composite nanofibers displayed much better catalytic activity than any single and bicomponent composite nanofibers for peroxidase mimicking. Due to the superior catalytic efficiency of the ternary TiO2@MoS2/CoFe2O4 composite nanofibers, a highly sensitive way for L-cysteine sensing was developed. The authors' work offers an advanced surface engineering soln. to construct a peroxidase-like catalyst to detect L-cysteine, with bright future in environmental science and biol. technol.
- 81Vallabani N. S. Singh S. Karakoti A. S. Investigating the role of ATP towards amplified peroxidase activity of Iron oxide nanoparticles in different biologically relevant buffers Appl. Surf. Sci. 2019 492 337 34881Investigating the role of ATP towards amplified peroxidase activity of Iron oxide nanoparticles in different biologically relevant buffersVallabani, N. V. Srikanth; Singh, Sanjay; Karakoti, Ajay S.Applied Surface Science (2019), 492 (), 337-348CODEN: ASUSEE; ISSN:0169-4332. (Elsevier B.V.)Evaluating the colloidal stability and enzyme mimetic nature of nanozymes at different buffer conditions is necessary to develop novel biosensing, biomedical and environmental applications. It is established that Fe3O4 nanoparticles show optimum peroxidase activity at pH 4 and it has also been shown that nucleotides like ATP can exhibit synergistic effect to enhance peroxidase activity at neutral pH. In this study we show the effect of buffers, pH and presence of ATP on peroxidase activity of Fe3O4 NPs and elucidate the mechanism involved in enhanced activity at neutral pH. Fe3O4 NPs colloidal stability was assessed over a period of 72 h from acidic to neutral pH. It was explained that OH radical generation from the synergistic combination of nucleotides and Fe3O4 NPs resulted in peroxidase activity at neutral pH and found that buffer concn. has a major effect on this activity. These findings challenge the existing theory of peroxidase activity demonstrated by Fe3O4 NPs at acidic pH. Moreover, the posibility can overcome the pH lacuna in designing novel biosensors and can also extend the heterogeneous Fenton reaction of Fe3O4 NPs over a wide range of pH.
- 82Gu Y. Huang Y. Qiu Z. Xu Z. Li D. Chen L. Jiang J. Gao L. Vitamin B2 functionalized iron oxide nanozymes for mouth ulcer healing Sci. China: Life Sci. 2020 63 1 68 7982Vitamin B2 functionalized iron oxide nanozymes for mouth ulcer healingGu, Yunhao; Huang, Yixin; Qiu, Zhiyue; Xu, Zhuobin; Li, Dandan; Chen, Lei; Jiang, Jing; Gao, LizengScience China: Life Sciences (2020), 63 (1), 68-79CODEN: SCLSCJ; ISSN:1674-7305. (Science China Press)Mouth ulcer is assocd. with inflammation and high risk of bacterial infection, which aggravates the patient's condition. Currently, there is no effective treatment for mouth ulcer. Herein, we report that vitamin-modified iron oxide nanoparticles improve the healing of mouth ulcer through anti-inflammation and antibacterial activities. We discovered that vitamin B2 (VB2) modified iron oxide nanoparticles performed enhanced peroxidase-like, catalase-like, and superoxide dismutase (SOD)-like activities, acting as typical iron oxide nanozymes (IONzymes) with triad activities. In particular, VB2 modification significantly improved the SOD-like activity, thus providing a reactive oxygen species (ROS)-scavenging ability. Cellular antioxidant expts. showed that vitamin B2 modified IONzymes (VB2-IONzymes) protect human oral keratinocytes (HOK) and BALB/3T3 cells from hydrogen peroxide (H2O2), and these cells have high biocompatibility to eukaryotic cells. In addn., VB2-IONzymes exerted an antibacterial activity against Streptococcus mutans, Staphylococcus aureus, and Escherichia coli. Importantly, VB2-IONzymes accelerated the recovery of mouth ulcer and reduced the local secretion of inflammatory factors in mouse ulcer model via ROS scavenging and antibacterial activity. Taken together, our work demonstrates that vitamin B2 modification endows iron oxide nanoparticles with enhanced enzyme-like activities and VB2-IONzymes may be a promising reagent in the treatment of mouth ulcer because of their intrinsic anti-inflammation and antibacterial capabilities.
- 83Adeniyi O. Sicwetsha S. Mashazi P. Nanomagnet-Silica Nanoparticles Decorated with Au@Pd for Enhanced Peroxidase-Like Activity and Colorimetric Glucose Sensing ACS Appl. Mater. Interfaces 2020 12 2 1973 198783Nanomagnet-Silica Nanoparticles Decorated with Au@Pd for Enhanced Peroxidase-Like Activity and Colorimetric Glucose SensingAdeniyi, Omotayo; Sicwetsha, Simbongile; Mashazi, PhilaniACS Applied Materials & Interfaces (2020), 12 (2), 1973-1987CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Nanomagnet-silica shell (Fe3O4@SiO2) decorated with Au@Pd nanoparticles (NPs) were synthesized successfully. The characterization of Fe3O4@SiO2-NH2-Au@PdNPs was achieved using several spectroscopic and microscopic techniques. The quant. surface anal. was confirmed using XPS. The Fe3O4@[email protected] exhibited excellent peroxidase-like activity by effectively catalyzing the oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The absorption peaks at 370 and 652 nm confirmed the peroxidase-like activity of the Fe3O4@[email protected]. The Michaelis-Menten const. (Km) of 0.350 and 0.090 mM showed strong affinity toward H2O2 and TMB at Fe3O4@[email protected]. The mechanism of the peroxidase-like activity proceeds via an electron transfer process. A simple colorimetric sensor based on glucose oxidase and Fe3O4@[email protected] showed excellent selectivity and sensitivity towards the detection of glucose. The fabricated glucose biosensor exhibited a wide linear response toward glucose from 0.010 to 60.0 μM with an limit of detection of 60.0 nM and limit of quantification of 200 nM. The colorimetric biosensor based on Fe3O4@[email protected] as a peroxidase mimic was also successfully applied for the detn. of glucose concns. in serum samples. The synthesized Fe3O4@[email protected] nanozymes exhibited excellent potential as an alternative to horseradish peroxidase for low-cost glucose monitoring.
- 84Lyu H. Y. Zhao X. Yao X. X. Chen W. Liu Z. X. Gao L. N. Fan G. C. Zhu X. X. Liu Q. Y. Zhang X. X. Zhang X. 3,4:9,10-perylene tetracarboxylic acid-modified zinc ferrite with the enhanced peroxidase activity for sensing of ascorbic acid Colloids Surf., A 2020 586 124250843,4:9,10-perylene tetracarboxylic acid-modified zinc ferrite with the enhanced peroxidase activity for sensing of ascorbic acidLyu, Haoyuan; Zhao, Xin; Yao, Xiuxiu; Chen, Wei; Liu, Zhenxue; Gao, Linna; Fan, Gaochao; Zhu, Xixi; Liu, Qingyun; Zhang, Xianxi; Zhang, XiaoColloids and Surfaces, A: Physicochemical and Engineering Aspects (2020), 586 (), 124250CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)3,4:9,10-Perylene tetracarboxylic acid (PTCA) modified litchi-like zinc ferrite (ZnFe2O4) was prepd. by a simple hydrothermal method and was found to possess the enhanced peroxidase-like activity. Under the catalysis of PTCA-ZnFe2O4, the colorless colorimetric substrate 3,3',5,5'-tetramethylbenzidine (TMB) was rapidly oxidized by H2O2 and fast turned into blue color. Comparatively, the peroxidase-like activity of PTCA-ZnFe2O4 nanocomposites is higher than that of pure ZnFe2O4. In addn., it is hydroxyl radical that plays the key role in the process of catalysis reaction, which verified by fluorescent probe. On the basis of the higher peroxidase-like activity of PTCA-ZnFe2O4, a simple colorimetric biosensor platform has been proposed for detection of ascorbic acid (AA) in the linear range of 1-10μM with the detection limit of 0.834μM.
- 85Mazhani M. Alula M. T. Murape D. Development of a cysteine sensor based on the peroxidase-like activity of AgNPs@Fe3O4 core-shell nanostructures Anal. Chim. Acta 2020 1107 193 20285Development of a cysteine sensor based on the peroxidase-like activity of AgNPs@ Fe3O4 core-shell nanostructuresMazhani, Micode; Alula, Melisew Tadele; Murape, DavisonAnalytica Chimica Acta (2020), 1107 (), 193-202CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)A facile one step solvothermal procedure has been employed in generating magnetite-silver core-shell nanocomposites (AgNPs@ Fe3O4) with superior peroxidase-like catalytic property than bare magnetic nanoparticles (Fe3O4). The composites were characterized using different techniques such as TEM, SEM, XRD, and surface-enhanced IR absorption spectroscopy (SEIRA). In the presence of hydrogen peroxide, the synthesized composites were able to oxidize the colorless o-phenylenediamine (OPD) to a yellow color 2,3-diaminophenazine (DAP) with a better peroxidase-like activity than Fe3O4 alone. The obtained Km value of AgNPs@ Fe3O4 with H2O2 and OPD substrates are 28.0 mM and 2.91 mM, resp. These are substantially lower than previously reported values and indicate the strong binding affinity of the substrates towards AgNPs@ Fe3O4 nanocomposites. Based on the obstruction activity of cysteine on the peroxidase-like catalytic property of the nanocomposites, a sensor was developed for detection of cystein with a limit of detection as low as 87 nM and a wider range of linearity. The sensor also exhibited excellent selectivity against potentially interfering mols.
- 86Mo W. Yu J. Gao L. Liu Y. Wei Y. He R. Reversible Inhibition of Iron Oxide Nanozyme by Guanidine Chloride Front. Chem. 2020 8 49186Reversible inhibition of iron oxide nanozyme by guanidine chlorideMo, Wei-chuan; Yu, Jia; Gao, Li-zeng; Liu, Ying; Wei, Yan; He, Rong-qiaoFrontiers in Chemistry (Lausanne, Switzerland) (2020), 8 (), 00491CODEN: FCLSAA; ISSN:2296-2646. (Frontiers Media S.A.)Nanozymes have been widely applied in bio-assays in the field of biotechnol. and biomedicines. However, the physicochem. basis of nanozyme catalytic activity remains elusive. To test whether nanozymes exhibit an inactivation effect similar to that of natural enzymes, we used guanidine chloride (GuHCl) to disturb the iron oxide nanozyme (IONzyme) and obsd. that GuHCl induced IONzyme aggregation and that the peroxidase-like activity of IONzyme significantly decreased in the presence of GuHCl. However, the aggregation appeared to be unrelated to the quick process of inactivation, as GuHCl acted as a reversible inhibitor of IONzyme instead of a solo denaturant. Inhibition kinetic anal. showed that GuHCl binds to IONzyme competitively with H2O2 but non-competitively with tetramethylbenzidine. In addn., ESR spectroscopy showed that increasing GuHCl level of GuHCl induced a correlated pattern of changes in the activity and the state of the unpaired electrons of the IONzymes. This result indicates that GuHCl probably directly interacts with the iron atoms of IONzyme and affects the electron d. of iron, which may then induce IONzyme inactivation. These findings not only contribute to understanding the essence of nanozyme catalytic activity but also suggest a practically feasible method to regulate the catalytic activity of IONzyme.
- 87Ngo Y. L. T. Nguyen P. L. Choi W. M. Chung J. S. Hur S. H. Highly sensitive smartphone-integrated colorimetric glucose sensor based on MnFe2O4 - graphitic carbon nitride hybrid nanostructure Mater. Res. Bull. 2020 129 11091087Highly sensitive smartphone-integrated colorimetric glucose sensor based on MnFe2O4 - graphitic carbon nitride hybrid nanostructureNgo, Yen-Linh Thi; Nguyen, Phi Luan; Choi, Won Mook; Chung, Jin Suk; Hur, Seung HyunMaterials Research Bulletin (2020), 129 (), 110910CODEN: MRBUAC; ISSN:0025-5408. (Elsevier Ltd.)An extremely sensitive colorimetric glucose sensor was fabricated using a novel hybrid nanostructure comprised of manganese ferrite oxide- graphitic carbon nitride (MnFe2O4/g-C3N4). A neural network-based glucose anal. smartphone application can be used to easily monitor the glucose concn. with a high accuracy by only taking images with the smartphone camera. The as-synthesized MnFe2O4/g-C3N4 exhibited a very high affinity toward 3,3',5,5'-tetramethylbenzidine (TMB) and H2O2, as calcd. via Michaelis-Menten kinetics. The glucose sensors using as-synthesized MnFe2O4/g-C3N4 exhibit a wide linear range and very low detection limits of 20.5 nM for H2O2 and 17.3 nM for glucose, which are one of the lowest values ever reported. The instrumental anal. with XPS, XRD, and TEM revealed that MnFe2O4/g-C3N4 had a highly cryst. structure of MnFe2O4 and g-C3N4 with abundant oxygen vacancies and well-developed hybrid structures, which might result in excellent catalytic activity and glucose sensitivity.
- 88Boruah P. K. Darabdhara G. Das M. R. Polydopamine functionalized graphene sheets decorated with magnetic metal oxide nanoparticles as efficient nanozyme for the detection and degradation of harmful triazine pesticides Chemosphere 2021 268 12932888Polydopamine functionalized graphene sheets decorated with magnetic metal oxide nanoparticles as efficient nanozyme for the detection and degradation of harmful triazine pesticidesBoruah, Purna K.; Darabdhara, Gitashree; Das, Manash R.Chemosphere (2021), 268 (), 129328CODEN: CMSHAF; ISSN:0045-6535. (Elsevier Ltd.)A facile and an eco-friendly redn. and functionalization of reduced graphene oxide (rGO) sheets is carried out using dopamine and decorated with magnetic Fe3O4 nanoparticles with an av. size of 12 nm by a simple co-pptn. method which is established as an artificial nanozyme. Here, functionalization of graphene using dopamine has introduced several advantages and insights into this study. The Fe3O4 nanoparticles decorated functionalized rGO sheets (FDGs) nanozymes are characterized by X-ray diffraction (XRD), XPS, high-resoln. transmission electron microscopy (HRTEM), Raman spectroscopy, at. force microscopy (AFM), thermogravimetric (TGA) and vibrating sample magnetometer (VSM) anal. FDGs nanozymes exhibits dual characteristics towards detection and degrdn. of harmful simazine pesticide. The hydrogen bonding interactions between pesticide mols. and 3,3',5,5'-tetramethylbenzidine (TMB) causes inhibition of the catalytic activity of the FDGs towards oxidn. of TMB mol. Based on that, the presence of simazine pesticide in an aq. medium can be easily detd. and a certain value (2.24μM) of detection limit was achieved. The photocatalytic degrdn. of simazine is also executed and excellent photocatalytic activity was obsd. under irradn. of direct natural sunlight. The FDGs nanozyme is also reusable up to several times with insignificant loss in its catalytic activity towards simazine degrdn.
- 89Hermosilla E. Seabra A. B. Lourenco I. M. Ferreira F. F. Tortella G. Rubilar O. Highly sensitive oxidation of MBTH/DMAB by MnFe2O4 nanoparticles as a promising method for nanozyme-based sensor development Colloids Surf., A 2021 621 12658589Highly sensitive oxidation of MBTH/DMAB by MnFe2O4 nanoparticles as a promising method for nanozyme-based sensor developmentHermosilla, Edward; Seabra, Amedea B.; Lourenco, Isabella M.; Furlan, Fabio F.; Tortella, Gonzalo; Rubilar, OlgaColloids and Surfaces, A: Physicochemical and Engineering Aspects (2021), 621 (), 126585CODEN: CPEAEH; ISSN:0927-7757. (Elsevier B.V.)Nanozymes are human-made nanomaterials with an intrinsic enzyme-like activity that offer a versatile alternative to natural enzymes for disease treatment, environmental remediation, and sensing. In this work, we proposed a novel spectrophotometric assay for detg. the oxidase-like activity of manganese ferrite nanoparticles (MnFe2O4 NPs) based on the oxidative coupling of 3-methyl-2-benzothiazolinone-hydrazone (MBTH) and 3-(dimethylamino) benzoic acid (DMAB). The effects of pH (2-8), temp. (20-50 °C), and kinetics parameters as the effect of substrate and nanoparticle concn. on the oxidase-like activity were evaluated. The MnFe2O4 NPs (∼3.19 ± 0.62 nm size) were synthesized by a co-pptn. method and characterized by TEM, SEM/EDS, VSM-SQUID, PXRD, and FTIR-ATR analyses. The optimum condition for MBTH/DMAB oxidn. catalyzed by MnFe2O4 NPs was obsd. at pH = 3.9 and 30 °C. The MBTH/DMAB oxidn. was adjusted to the Michaelis Menten kinetics model (R2 = 0.96) and showed a Km = 13.59 μM MBTH, kcat = 5.25 x 107 s-1 and kcat/Km = 3.86 x 1012 M-1 s-1. These high kcat and kcat/Km values indicate that the MBTH/DMAB couple is more sensitive than previously reported oxidase substrates by about three orders of magnitude. Hence, our findings open up a wide range of new potential applications on the development of more sensitive sensors based on the MBTH/DMAB reaction catalyzed by MnFe2O4 NPs for detecting several analytes (including alk. phosphatase, heavy metals, dissolved oxygen in water, among others) in the medical and environmental field.
- 90You S. M. Park J. S. Luo K. Jeong K. B. Adra H. J. Kim Y. R. Modulation of the peroxidase-like activity of iron oxide nanoparticles by surface functionalization with polysaccharides and its application for the detection of glutathione Carbohydr. Polym. 2021 267 11816490Modulation of the peroxidase-like activity of iron oxide nanoparticles by surface functionalization with polysaccharides and its application for the detection of glutathioneYou, Sang-Mook; Park, Jin-Sung; Luo, Ke; Jeong, Ki-Baek; Adra, Hazzel Joy; Kim, Young-RokCarbohydrate Polymers (2021), 267 (), 118164CODEN: CAPOD8; ISSN:0144-8617. (Elsevier Ltd.)Here, we employed three polysaccharides, such as dextran, hyaluronic acid, and chitosan, for surface modification of iron oxide nanoparticles (IONPs) and carried out in-depth investigation to elucidate the effect of surface functionalities on the peroxidase (POD) like activity of IONPs. The affinity of substrates to the catalytic site of IONPs was found to be detd. by the surface functional groups and hydration layer of polysaccharide coating on the surface of IONPs. The role of hydration layer was further confirmed by the results that the POD-like activity of IONPs coated with a certain polysaccharide having higher water holding capacity was significantly enhanced by salting-out reagent, such as ammonium chloride that is known to reduce the thickness of hydration layer. Moreover, the excellent catalytic activity of dextran-coated IONPs was successfully applied to develop a highly sensitive sensing system for the detection of glutathione (GSH) with a limit of detection of 2.3 nM.
- 91Bian B. Zhu X. X. Wu Q. Liu Y. Liu S. W. Liu Q. Y. Yu S. T. Pt and ZnFe2O4 Nanoparticles Immobilized on Carbon for the Detection of Glutathione ACS Appl. Nano Mater. 2021 4 9 9479 948891Pt and ZnFe2O4 Nanoparticles Immobilized on Carbon for the Detection of GlutathioneBian, Bing; Zhu, Xixi; Wu, Qiong; Liu, Yue; Liu, Shiwei; Liu, Qingyun; Yu, ShitaoACS Applied Nano Materials (2021), 4 (9), 9479-9488CODEN: AANMF6; ISSN:2574-0970. (American Chemical Society)The catalytic activity of nanozymes has attracted much attention in biochem. analyses and the antibiosis and environmental fields. However, easy aggregation and difficulty in recycling greatly result in a decrease of their catalytic activity and further restrict their practical application. Herein, the authors immobilized Pt and ZnFe2O4 nanoparticles on poplar-wood-derived carbon (ZnFe2O4/C), which exhibited excellent peroxidase-like activity and high affinity toward H2O2 and 3,3',5,5'-tetramethylbenzidine (TMB). Moreover, it could be easily sepd. by an external magnetic force from the reaction system. A sensitive and selective colorimetric sensor for glutathione (GSH) was established based on Pt/ZnFe2O4/C nanoperoxidase. The linear range for GSH was 1-30 μM, and the limit of detection for GSH was calcd. as 0.38 μM. The colorimetric method was applied to det. GSH in real samples for injection.
- 92Singh E. Kaur M. Sharma S. Structural tuning of CTAB@MgFe2O4 nanocomposite as peroxidase mimic for H2O2 and glucose sensing Mater. Chem. Phys. 2021 271 12485192Structural tuning of CTAB@MgFe2O4 nanocomposite as peroxidase mimic for H2O2 and glucose sensingSingh, Ekjot; Kaur, Manpreet; Sharma, SuchetaMaterials Chemistry and Physics (2021), 271 (), 124851CODEN: MCHPDR; ISSN:0254-0584. (Elsevier B.V.)Natural enzymes have been used in food industry, biosensing and pharmaceuticals due to their high catalytic activity. Their catalytic potential is governed by the environmental conditions leading to their denaturation, thus increasing operational cost. Nanoparticles (NPs) as enzyme mimic is a thrust area of research. Magnesium ferrite nanoparticles (MgFe2O4 NPs) have gained focus of researchers due to good catalytic activity, stability and magnetic properties. Their catalytic activity can be enhanced by surface coating with suitable surfactant. In this work, CTAB (cetyl tri-Me ammonium bromide) coated MgFe2O4 NPs were prepd. and evaluated as peroxidase mimics. The XRD pattern of MgFe2O4 NPs confirmed single-phase formation of spinel structure with cubic geometry. FT-IR studies confirmed the presence of CTAB along with MgFe2O4 NPs. Surface area, Particle size and magnetic character of the NPs were evaluated by Brunauer Emmett Teller anal., Transmission Electron Microscope and Vibrating Sample Magnetometer resp. CTAB coated ferrite NPs showed increased surface area and decreased particle size as compared to pristine NPs. CTAB@MgFe2O4 in wt./wt. ratio (1:1) showed best peroxidase like activity towards o-dianisidine dihydrochloride and was influenced by pH, temp. and substrate concn. in soln. The synthesized CTAB@MgFe2O4 (1:1) based system could be used for colorimetric detection of H2O2 in the linear range of 20-300μM with a detection limit of 10μM and also for the detection for glucose in the range of 10-1000μM with a detection limit of 5μM.
- 93Zhu W. D. Cheng Y. Wang C. Lu X. F. Fiber-in-Tube Design of a CuFe2O4@Conducting Polymer with Synergistically Enhanced Peroxidase-like Activity for Total Antioxidant Capacity Assays ACS Sustainable Chem. Eng. 2021 9 44 14811 1482093Fiber-in-Tube Design of a CuFe2O4@Conducting Polymer with Synergistically Enhanced Peroxidase-like Activity for Total Antioxidant Capacity AssaysZhu, Wendong; Cheng, Ya; Wang, Ce; Lu, XiaofengACS Sustainable Chemistry & Engineering (2021), 9 (44), 14811-14820CODEN: ASCECG; ISSN:2168-0485. (American Chemical Society)The exploration of novel nanozymes with a high catalytic efficiency is crucial for their biosensing applications. Intricate hollow nanostructures possess distinct hierarchical open and porous features, usually presenting fascinating physicochem. properties. In this work, we have demonstrated the fabrication of a hierarchical fiber-in-tube CuFe2O4@conducting polypyrrole (PPy) nanostructure for peroxidase mimicking. Owing to the unique confined structure and synergistic effect, an exceptional peroxidase-like performance of CuFe2O4@PPy is achieved compared with the pristine CuFe2O4 nanofibers and PPy nanotubes alone. The prepd. catalyst displays a desirable long-term and cycling stability. The exptl. results demonstrate the key role of the formation of hydroxyl radicals for peroxidase mimicking. Based on the outstanding peroxidase-like activity, a high-efficiency ascorbic acid (AA) colorimetric sensor is developed, also displaying a satisfactory selectivity. Furthermore, according to the high sensitivity of the CuFe2O4@PPy nanotubes to detect AA, a total antioxidant capacity (TAC) sensor for a series of real samples has been developed. This work provides a new approach to construct efficient nanozymes with high efficiencies and develop a simple TAC assay with further vision in the fields of biosensing and food technol.
- 94Xing Y. Y. Chen M. L. Zhao Y. K. Xu J. B. Hou X. H. Triple-enzyme mimetic activity of Fe3O4@C@MnO2 composites derived from metal-organic frameworks and their application to colorimetric biosensing of dopamine Microchim. Acta 2022 189 1 1 10There is no corresponding record for this reference.
- 95Yu F. Q. Huang Y. Z. Cole A. J. Yang V. C. The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detection Biomaterials 2009 30 27 4716 472295The artificial peroxidase activity of magnetic iron oxide nanoparticles and its application to glucose detectionYu, Faquan; Huang, Yongzhuo; Cole, Adam J.; Yang, Victor C.Biomaterials (2009), 30 (27), 4716-4722CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)Aside from their superparamagnetic properties exploited in clin. magnetic resonance imaging (MRI), it was recently discovered that magnetic, iron oxide nanoparticles could function as an artificial, inorg. peroxidase. In this paper, the authors studied the impact of coating on the peroxidase activity of these nanoparticles. Nanoparticles with six different coating structures were synthesized and characterized by FTIR, TGA, TEM, size, zeta potential, and SQUID; and evaluated for peroxidase activity. Catalysis was found to follow Michaelis-Menten kinetics and peroxidase activity varied with respect to electrostatic affinity between nanoparticles and substrates, evidenced by differences in detd. kinetic parameters. Glucose detection was selected as a model system because glucose could be indirectly measured from the release of hydrogen peroxide after its oxidn. Nanoparticles with high peroxidase activity exhibited higher sensitivity toward glucose, showing a larger linear slope when compared with those of low activity. A significantly improved linear correlation and detection limit of measured glucose could be readily obtained by manipulating the nanoparticle coating. The authors' findings suggest that iron oxide nanoparticles can be tailor-made to possess improved peroxidase-like activity. Such enhancements could further widen nanoparticle scope in glucose detection and extend its peroxidase functionality to other biomedical applications.
- 96Zhang K. Zuo W. Wang Z. Y. Liu J. Li T. R. Wang B. D. Yang Z. Y. A simple route to CoFe2O4 nanoparticles with shape and size control and their tunable peroxidase-like activity RSC Adv. 2015 5 14 10632 1064096A simple route to CoFe2O4 nanoparticles with shape and size control and their tunable peroxidase-like activityZhang, Ke; Zuo, Wei; Wang, Zhiyi; Liu, Jian; Li, Tianrong; Wang, Baodui; Yang, ZhengyinRSC Advances (2015), 5 (14), 10632-10640CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)Recent studies have suggested that the phys. and chem. properties of nanoparticles (NPs) strongly depend on local chem. compn., size, and shape. Here, we report a new precursor-mediated growth of monodisperse magnetic cobalt ferrite (CoFe2O4) NPs with controlled size and shape. CoFe2O4 NPs with near corner-grown cubic, near cubic and polyhedron shape can be successfully prepd. by simply tuning the amt. of iron and cobalt acetylacetonates in oleic acid. Interestingly, the product shape varies from near corner-grown cubic to starlike by only changing the reaction temp. from 320 °C to 330 °C. These CoFe2O4 NPs exhibit size and shape-dependent peroxidase-like activity towards 3,3',5,5'-tetramethylbenzdine (TMB) in the presence of H2O2, and thus exhibited different levels of peroxidase-like activities, in the order of spherical > near corner-grown cubic > starlike > near cubic > polyhedron; this order was closely related to their particle size and crystal morphol. CoFe2O4NPs exhibited high stability in HAc-NaAc buffer (pH = 4.0) and high activity over a broad pH (2.5-6.0). Furthermore, the Michaelis consts. Km value for the CoFe2O4 NPs (0.006 mM) with TMB as the substrate was lower than HRP (0.062 mM) and Fe3O4 NPs (0.010 mM). After further surface functionalization with folic acid (FA), the folate-conjugated CoFe2O4 nanoparticles allow discrimination of HeLa cells (folate receptor overexpression) from NIH-3T3 cells (without folate receptor expression). Such investigation is of great significance for peroxidase nanomimetics with enhanced activity and utilization.
- 97Su L. Qin W. Zhang H. Rahman Z. U. Ren C. Ma S. Chen X. The peroxidase/catalase-like activities of MFe2O4 (M= Mg, Ni, Cu) MNPs and their application in colorimetric biosensing of glucose Biosens. Bioelectron. 2015 63 384 39197The peroxidase/catalase-like activities of MFe2O4 (M=Mg, Ni, Cu) MNPs and their application in colorimetric biosensing of glucoseSu, Li; Qin, Wenjie; Zhang, Huige; Rahman, Zia Ur; Ren, Cuiling; Ma, Sudai; Chen, XingguoBiosensors & Bioelectronics (2015), 63 (), 384-391CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)MFe2O4 (M=Mg, Ni, Cu) magnetic nanoparticles (MNPs) were found to have catalytic activities similar to those of biol. enzymes such as catalase and peroxidase. These nanomaterials, as bifunctional catalase/peroxidases (KatGs), not only could catalyze H2O2 to produce hydroxyl radicals, which oxidized peroxidase substrate to produce color, but also could catalyze the decompn. reaction of H2O2 into water and oxygen directly in the same condition through the catalase-like activity. And it was also found that the amt. of generated hydroxyl radicals and oxygen was related to the concn. of MFe2O4 (M=Mg, Ni, Cu) MNPs. The peroxidase-like catalytic behavior of MFe2O4 MNPs was analyzed in detail. Under the optimized conditions, NiFe2O4 MNPs were used as a colorimetric biosensor for the detection of 9.4×10-7-2.5×10-5 mol L-1 glucose with a limit of detection (LOD) of 4.5×10-7 mol L-1. The sensor was successfully applied to glucose detection in urine sample.
- 98Yuan F. Zhao H. Zang H. Ye F. Quan X. Three-dimensional graphene supported bimetallic nanocomposites with DNA regulated-flexibly switchable peroxidase-like activity ACS Appl. Mater. Interfaces 2016 8 15 9855 986498Three-Dimensional Graphene Supported Bimetallic Nanocomposites with DNA Regulated-Flexibly Switchable Peroxidase-Like ActivityYuan, Fang; Zhao, Huimin; Zang, Hongmei; Ye, Fei; Quan, XieACS Applied Materials & Interfaces (2016), 8 (15), 9855-9864CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)A synergistic bimetallic enzyme mimetic catalyst, three-dimensional (3D) graphene/Fe3O4-AuNPs, was successfully fabricated which exhibited flexibly switchable peroxidase-like activity. Compared to the traditional 2D graphene-based monometallic composite, the introduced 3D structure, which was induced by the addn. of glutamic acid, and bimetallic anchoring approach dramatically improved the catalytic activity, as well as the catalysis velocity and its affinity for substrate. Herein, Fe3O4NPs acted as supporters for AuNPs, which contributed to enhance the efficiency of electron transfer. On the basis of the measurement of Mott-Schottky plots of graphene and metal anchored hybrids, the catalysis mechanism was elucidated by the decrease of Fermi level resulted from the chem. doping behavior. Notably, the catalytic activity was able to be regulated by the adsorption and desorption of single-stranded DNA mols., which laid a basis for its utilization in the construction of single-stranded DNA-based colorimetric biosensors. This strategy not only simplified the operation process including labeling, modification, and imprinting, but also protected the intrinsic affinity between the target and biol. probe. Accordingly, based on the peroxidase-like activity and its controllability, our prepd. nanohybrids was successfully adopted in the visualized and label-free sensing detections of glucose, sequence-specific DNA, mismatched nucleotides, and oxytetracycline.
- 99Lu N. Zhang M. Ding L. Zheng J. Zeng C. Wen Y. Liu G. Aldalbahi A. Shi J. Song S. Yolk–shell nanostructured Fe3O4@C magnetic nanoparticles with enhanced peroxidase-like activity for label-free colorimetric detection of H2O2 and glucose Nanoscale 2017 9 13 4508 451599Yolk-shell nanostructured Fe3O4@C magnetic nanoparticles with enhanced peroxidase-like activity for label-free colorimetric detection of H2O2 and glucoseLu, Na; Zhang, Min; Ding, Lei; Zheng, Jing; Zeng, Caixia; Wen, Yanli; Liu, Gang; Aldalbahi, Ali; Shi, Jiye; Song, Shiping; Zuo, Xiaolei; Wang, LihuaNanoscale (2017), 9 (13), 4508-4515CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Herein, we have developed a simple and facile method to synthesize yolk-shell nanostructured Fe3O4@C nanoparticles (NPs) as a multifunctional biosensing platform for the label-free colorimetric detection of H2O2 and glucose. It was demonstrated that Fe3O4@C yolk-shell nanostructures (YSNs) retained the magnetic properties that can be used for sepn. and concn. Also importantly, the Fe3O4@C YSNs exhibited an intrinsic peroxidase-like activity that could quickly catalyze the enzyme substrate in the presence of H2O2 and produce a blue color. Compared to other similar ferric oxide-based NPs with different structures, Fe3O4@C YSNs exhibited greatly enhanced catalytic activities due to their unique structural features. Moreover, steady-state kinetics indicated the catalytic behaviors in agreement with the classic Michaelis-Menten models. Taking advantage of the high catalytic activity, Fe3O4@C YSNs were employed as novel peroxidase mimetics for label-free, rapid, sensitive, and specific colorimetric sensing of H2O2 and glucose, suggesting that Fe3O4@C YSNs have the potential for construction of portable sensors in the application of point-of-care (POC) diagnosis and on-site tests.
- 100Huang F. Wang J. Z. Chen W. M. Wan Y. J. Wang X. M. Cai N. Liu J. Yu F. Q. Synergistic peroxidase-like activity of CeO2-coated hollow Fe3O4 nanocomposites as an enzymatic mimic for low detection limit of glucose J. Taiwan Inst. Chem. Eng. 2018 83 40 49100Synergistic peroxidase-like activity of CeO2-coated hollow Fe3O4 nanocomposites as an enzymatic mimic for low detection limit of glucoseHuang, Fei; Wang, Jianzhi; Chen, Weimin; Wan, Yinjia; Wang, Xianming; Cai, Ning; Liu, Jie; Yu, FaquanJournal of the Taiwan Institute of Chemical Engineers (2018), 83 (), 40-49CODEN: JTICA8; ISSN:1876-1070. (Elsevier B.V.)Fe3O4@CeO2 yolk-shell nanocomposites (NCs) were synthesized by growing CeO2 onto Fe3O4 hollow nanoparticles (NPs) as an artificial peroxidase for the purpose of elevated activity. The resultant NCs exhibited strong magnetic response with narrow size distribution and high surface area. Owing to the individual peroxidase-like catalytic activity of CeO2 onto Fe3O4 nanoparticles (NPs), Fe3O4@CeO2 NCs was investigated to achieve synergistic catalytic activity. As a result, the as-prepd. Fe3O4@CeO2 NCs exhibited impressive peroxidase-like catalysis activity for 3, 3', 5, 5'-tetramethylbenzidine (TMB). Moreover, comparing with natural enzymes, hollow Fe3O4 NPs and CeO2 NPs, the Fe3O4@CeO2 NCs displayed a lower Michaelis const. (Km) value or stronger affinity to H2O2 and TMB. A limit of detection of 8.5μmol/L for H2O2 and 21μmol/L for glucose was attained with acceptable stability and reproducibility. The activation mechanism was also studies using competitive radical tests and ESR (ESR) anal.
- 101Lian J. J. Liu P. Li X. C. Gao L. N. Luo X. L. Zhang X. Shi Z. Q. Liu Q. Y. Perylene diimide-modified magnetic gamma-Fe2O3/CeO2 nanoparticles as peroxidase mimics for highly sensitive colorimetric detection of Vitamin C Appl. Organomet. Chem. 2019 33 5 e4884There is no corresponding record for this reference.
- 102Cui M. Zhang H. R. Ouyang F. Guo Y. Q. Li R. F. Duan S. F. Xiong T. D. Wang Y. L. Wang X. Q. Dual Enzyme-Like Performances of PLGA Grafted Maghemite Nanocrystals and Their Synergistic Chemo/Chemodynamic Treatment for Human Lung Adenocarcinoma A549 Cells J. Biomed. Nanotechnol. 2021 17 6 1007 1019102Dual enzyme-like performances of PLGA grafted maghemite nanocrystals and their synergistic chemo/chemodynamic treatment for human lung adenocarcinoma A549 cellsCui, Miao; Zhang, Hui-Ru; Ouyang, Fan; Guo, Yu-Qi; Li, Rui-Fang; Duan, Shao-Feng; Xiong, Tian-Di; Wang, Yun-Long; Wang, Xue-QinJournal of Biomedical Nanotechnology (2021), 17 (6), 1007-1019CODEN: JBNOAB; ISSN:1550-7033. (American Scientific Publishers)In recent years, the emergence of non-toxic but catalytically active inorg. nanoparticles has attracted great attention for cancer treatment, but the therapeutic effect has been affected by the limited reactive oxygen species in tumors. Therefore, the combination of chemotherapy and chemodynamic therapy is regarded as a promising therapeutic strategy. In this paper, we reported the prepn. and bioactivity evaluation of poly(lactic acid-co-glycolic acid) (PLGA) grafted-γ-Fe2O3 nanoparticles with dual response of endogenous peroxidase and catalase like activities. Our hypothesis is that PLGA-grafted γ-Fe2O3 nanoparticles could be used as a drug delivery system for the anti-tumor drug doxorubicin to inhibit the growth of lung adenocarcinoma A549 cells; meanwhile, based on its mimic enzyme properties, this kind of nanoparticles could be combined with doxorubicin in the treatment of A549 cells. Our exptl. results showed that the PLGA-grafted γ-Fe2O3 nanoparticles could simulate the activity of catalase and decomp. hydrogen peroxide into H2O and oxygen in neutral tumor microenvironment, thus reducing the oxidative damage caused by hydrogenperoxide to lung adenocarcinoma A549 cells. In acidic microenvironment, PLGA grafted γ-Fe2O3 nanoparticles could simulate the activity of peroxidase and effectively catalyze the decompn. of hydrogen peroxide to generate highly toxic hydroxyl radicals, which could cause the death of A549 cells. Furthermore, the synergistic effect of peroxidase-like activity of PLGA-grafted γ-Fe2O3 nanoparticles and doxorubicin could accelerate the apoptosisand destruction of A549 cells, thus enhancing the antitumor effect of doxorubicin-loaded PLGA-grafted γ-Fe2O3 nanoparticles. Therefore, this study provides an effective nanoplatform based on dual inorg. biomimetic nanozymes for the treatment of lung cancer.
- 103Fu S. Y. Yang R. H. Ren J. J. Liu J. H. Zhang L. Xu Z. G. Kang Y. J. Xue P. Catalytically Active CoFe2O4 Nanoflowers for Augmented Sonodynamic and Chemodynamic Combination Therapy with Elicitation of Robust Immune Response ACS Nano 2021 15 7 11953 11969103Catalytically Active CoFe2O4 Nanoflowers for Augmented Sonodynamic and Chemodynamic Combination Therapy with Elicitation of Robust Immune ResponseFu, Shiyan; Yang, Ruihao; Ren, Junjie; Liu, Jiahui; Zhang, Lei; Xu, Zhigang; Kang, Yuejun; Xue, PengACS Nano (2021), 15 (7), 11953-11969CODEN: ANCAC3; ISSN:1936-0851. (American Chemical Society)A hypoxic and acidic tumor microenvironment (TME) plays a significant role in cancer development through complex cellular signaling networks, and it is thus challenging to completely eradicate tumors via monotherapy. Here, PEGylated CoFe2O4 nanoflowers (CFP) with multiple enzymic activities, serving as bioreactors responsive to TME cues, were synthesized via a typical solvothermal method for augmented sonodynamic therapy (SDT) and chemodynamic therapy (CDT) with elicitation of robust immune response. The CFP occupying multivalent elements (Co2+/3+, Fe2+/3+) exhibited strong Fenton-like and catalase-like activity. In another aspect, CFP itself is a brand-new sonosensitizer for high-performance SDT based on ultrasound-triggered electron (e-)/hole (h+) pair sepn. from the energy band with promptness and high efficiency. With efficient enrichment in tumorous tissue as revealed by magnetic resonance imaging, CPF could generate •OH for CDT relying on Fenton-like reactions. Moreover, catalase-mimicking CFP could react with endogenous H2O2 to generate mol. oxygen, and high O2 level may promote the prodn. of 1O2 for SDT. What's more, the reactive oxygen species obtained from combined SDT/CDT could efficiently trigger immunogenic cell death through a synergistic therapy based on the elicitation of antitumor immunity with the aid of an immune checkpoint blockade for the sake of suppressing primary and distant tumors as well as lung metastasis. Taken together, this paradigm delivers useful insights for developing in-coming nanocomposites based on cobalt ferrite for cancer theranostics.
- 104Liu Y. S. Wang X. J. Li X. Z. Qiao S. S. Huang G. D. Hermann D. M. Doeppner T. R. Zeng M. L. Liu W. Xu G. L. Ren L. J. Zhang Y. Liu W. L. Casals E. Li W. P. Wang Y. C. A Co-Doped Fe3O4 Nanozyme Shows Enhanced Reactive Oxygen and Nitrogen Species Scavenging Activity and Ameliorates the Deleterious Effects of Ischemic Stroke ACS Appl. Mater. Interfaces 2021 13 39 46213 46224104A Co-Doped Fe3O4 Nanozyme Shows Enhanced Reactive Oxygen and Nitrogen Species Scavenging Activity and Ameliorates the Deleterious Effects of Ischemic StrokeLiu, Yunsheng; Wang, Xiaojun; Li, Xiangzhu; Qiao, Shanshan; Huang, Guodong; Hermann, Dirk Matthias; Doeppner, Thorsten Roland; Zeng, Muling; Liu, Wei; Xu, Gelin; Ren, Lijie; Zhang, Yuan; Liu, Wenlan; Casals, Eudald; Li, Weiping; Wang, Ya-ChaoACS Applied Materials & Interfaces (2021), 13 (39), 46213-46224CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)Acute ischemic stroke has become the major cause of mortality and disability worldwide. Following ischemic stroke, the reperfusion injury is mainly mediated by the burst of reactive oxygen and nitrogen species (RONS). Therefore, blocking the excessive prodn. or removing RONS holds great promise as a potential therapeutic strategy. Herein, we developed a Co-doped Fe3O4 nanozyme that is capable of scavenging H2O2, O2·-, ·NO, and ONOO- in vitro and in vivo and provides neuroprotection against ischemic stroke. In vitro expts. showed that pre-incubation with the Co-Fe3O4 nanozyme could prevent neurotoxicity and neuroinflammation induced by H2O2 or lipopolysaccharide, resp., in HT22 cells. After i.v. administration, the Co-Fe3O4 nanozyme showed no signs of toxicity in peripheral organs of C57BL/6J mice, even after prolonged delivery for 4 wk. In permanent photothrombotic stroke model and transient middle cerebral artery occlusion stroke model, the Co-Fe3O4 nanozyme specifically accumulated in the infarct rim at 72 h post-stroke and was endocytosed by neurons, astrocytes, microglia, and endothelial cells. Importantly, the Co-Fe3O4 nanozyme delivery reduced the infarct vol. in both stroke models. The observation that the Co-Fe3O4 nanozyme was efficacious in two well-characterized ischemic stroke models provides strong evidence that it represents a powerful tool for targeting oxidative and nitrosative stress in the ischemic brain.
- 105Raineri M. Winkler E. L. Torres T. E. Vasquez Mansilla M. Nadal M. S. Zysler R. D. Lima E. Effects of biological buffer solutions on the peroxidase-like catalytic activity of Fe3O4 nanoparticles Nanoscale 2019 11 39 18393 18406105Effects of biological buffer solutions on the peroxidase-like catalytic activity of Fe3O4 nanoparticlesRaineri, Mariana; Winkler, Elin L.; Torres, Teobaldo E.; Vasquez Mansilla, Marcelo; Nadal, Marcela S.; Zysler, Roberto D.; Lima, EnioNanoscale (2019), 11 (39), 18393-18406CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)In our study, IONP activity was evaluated in different buffering solns. under different exptl. conditions and predominant free radical species were measured by ESR using the spin-trap 5,5-dimethyl-1-pyrroline N-oxide (DMPO). The formation kinetics of the reactive oxygen species were studied by UV-visible spectroscopy with TMB and DAB peroxidase substrates. We found that the highest catalytic oxidn. of peroxidase substrates and free radical generation were achieved in acetate buffer, while phosphate buffer inhibited the peroxidase-like activity of IONPs in a concn. dependent manner. When emulating the physiol. conditions, a lower catalytic activity was obsd. at pH 7.4. Also, in phosphate buffered saline (PBS), we obsd. an enhancement in the peroxidase substrate oxidn. rate that was not accompanied by an increase in DMPO/adduct formation which could be related to a non-specific oxidn. catalyzed by the chloride ion. Similar observations were found after the addn. of a bicarbonate to HEPES buffer. TMB oxidn. did not occur when the reaction was conducted with free iron ions from metal salts with the same concn. of the IONPs (0.33 Fe2+ and 0.66 Fe3+). These results indicate that biol. buffering solns. need to be carefully considered when evaluating IONP catalytic activity and their potential toxicol. effects since under physiol. conditions of pH, salinity and buffering species, the peroxidase-like activity of IONPs is dramatically reduced.
- 106Gao L. Yan X. Discovery and current application of nanozyme Prog. Biochem. Biophys. 2013 40 10 892 902There is no corresponding record for this reference.
- 107Qin T. Ma R. Yin Y. Miao X. Chen S. Fan K. Xi J. Liu Q. Gu Y. Yin Y. Catalytic inactivation of influenza virus by iron oxide nanozyme Theranostics 2019 9 23 6920107Catalytic inactivation of influenza virus by iron oxide nanozymeQin, Tao; Ma, Ruonan; Yin, Yinyan; Miao, Xinyu; Chen, Sujuan; Fan, Kelong; Xi, Juqun; Liu, Qi; Gu, Yunhao; Yin, Yuncong; Hu, Jiao; Liu, Xiufan; Peng, Daxin; Gao, LizengTheranostics (2019), 9 (23), 6920-6935CODEN: THERDS; ISSN:1838-7640. (Ivyspring International Publisher)Influenza poses a severe threat to human health in the world. However, developing a universal anti-viral strategy has remained challenging due to the presence of diverse subtypes as well as its high mutation rate, resulting in antigenic shift and drift. Here we developed an antiviral strategy using iron oxide nanozymes (IONzymes) to target the lipid envelope of the influenza virus. Methods: We evaluated the antiviral activities of our IONzymes using a hemagglutination assay, together with a 50% tissue culture infectious doses (TCID50) method. Lipid peroxidn. of the viral envelope was analyzed using a maleic dialdehyde (MDA) assay and transmission electron microscopy (TEM). The neighboring viral proteins were detected by western blotting. Results: We show that IONzymes induce envelope lipid peroxidn. and destroy the integrity of neighboring proteins, including hemagglutinin, neuraminidase, and matrix protein 1, causing the inactivation of influenza A viruses (IAVs). Furthermore, we show that our IONzymes possess a broad-spectrum antiviral activity on 12 subtypes of IAVs (H1~ H12). Lastly, we demonstrate that applying IONzymes to a facemask improves the ability of virus protection against 3 important subtypes that pose a threat to human, including H1N1, H5N1, and H7N9 subtype. Conclusion: Together, our results clearly demonstrate that IONzymes can catalyze lipid peroxidn. of the viral lipid envelope to inactivate enveloped viruses and provide protection from viral transmission and infection.
- 108Hong C. Meng X. He J. Fan K. Yan X. Nanozyme: A promising tool from clinical diagnosis and environmental monitoring to wastewater treatment Particuology 2022 71 90 107108Nanozyme: A promising tool from clinical diagnosis and environmental monitoring to wastewater treatmentHong, Chaoyi; Meng, Xiangqin; He, Jiuyang; Fan, Kelong; Yan, XiyunParticuology (2022), 71 (), 90-107CODEN: PARTC5; ISSN:1674-2001. (Elsevier B.V.)A review. Natural enzymes, owing to their outstanding catalytic efficiency and substrate specificity, have been used in a variety of applications including clin. diagnosis, environmental monitoring and wastewater treatment. However, they face inevitable problems such as relatively high cost and lack of stability, dramatically hindering their practical applications in the industry. Recently, a class of nanomaterial that possesses intrinsic enzyme-like properties, nanozyme, has emerged exhibiting numerous advantages over its natural counterpart and has been used as a viable enzyme alternative. In the past decade there are many reviews on nanozyme. The previous discussions tend to view nanozyme as a type of nanomaterial rather than an enzyme. However, it is the enzyme-like activity of nanozymes that provides foundation for their application and nanozymes with the same enzymic activity usually have some regularity in application. Herein, in this review, we attempt to classify nanozymes by their enzyme-like activity to explain the application principle and relevant cases of nanozymes in clin. diagnosis, environmental monitoring and wastewater treatment, expecting to promote deeper thinking of nanozymes as enzyme mimics and provide useful guidance for future research.
- 109Chen M. Deng G. He Y. Li X. Liu W. Wang W. Zhou Z. Yang H. Yang S. Ultrasound-enhanced generation of reactive oxygen species for MRI-guided tumor therapy by the Fe@Fe3O4-Based peroxidase-mimicking nanozyme ACS Appl. Bio Mater. 2019 3 1 639 647There is no corresponding record for this reference.
- 110Shi C. Li Y. Gu N. Iron-Based Nanozymes in Disease Diagnosis and Treatment ChemBioChem 2020 21 19 2722 2732110Iron-Based Nanozymes in Disease Diagnosis and TreatmentShi, Chu; Li, Yan; Gu, NingChemBioChem (2020), 21 (19), 2722-2732CODEN: CBCHFX; ISSN:1439-4227. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. Iron-based nanozymes are currently one of the few clin. inorg. nanoparticles for disease diagnosis and treatment. Overcoming the shortcomings of natural enzymes, such as easy inactivation and low yield, combined with their special nanometer properties and magnetic functions, iron-based nanozymes have broad prospects in biomedicine. This minireview summarizes their prepn., biol. activity, catalytic mechanism, and applications in diagnosis and treatment of diseases. Finally, challenges to their future development and the trends of iron-based nanozymes are discussed. The purpose of this minireview is to better understand and reasonably speculate on the rational design of iron-based nanozymes as an increasingly important new paradigm for diagnostics.
- 111Ji S. Jiang B. Hao H. Chen Y. Dong J. Mao Y. Zhang Z. Gao R. Chen W. Zhang R. Matching the kinetics of natural enzymes with a single-atom iron nanozyme Nat. Catal. 2021 4 5 407 417111Matching the kinetics of natural enzymes with a single-atom iron nanozymeJi, Shufang; Jiang, Bing; Hao, Haigang; Chen, Yuanjun; Dong, Juncai; Mao, Yu; Zhang, Zedong; Gao, Rui; Chen, Wenxing; Zhang, Ruofei; Liang, Qian; Li, Haijing; Liu, Shuhu; Wang, Yu; Zhang, Qinghua; Gu, Lin; Duan, Demin; Liang, Minmin; Wang, Dingsheng; Yan, Xiyun; Li, YadongNature Catalysis (2021), 4 (5), 407-417CODEN: NCAACP; ISSN:2520-1158. (Nature Portfolio)Abstr.: Developing artificial enzymes with the excellent catalytic performance of natural enzymes has been a long-standing goal for chemists. Single-atom catalysts with well-defined at. structure and electronic coordination environments can effectively mimic natural enzymes. Here, we report an engineered FeN3P-centered single-atom nanozyme (FeN3P-SAzyme) that exhibits comparable peroxidase-like catalytic activity and kinetics to natural enzymes, by controlling the electronic structure of the single-atom iron active center through the precise coordination of phosphorus and nitrogen. In particular, the engineered FeN3P-SAzyme, with well-defined geometric and electronic structures, displays catalytic performance that is consistent with Michaelis-Menten kinetics. We rationalize the origin of the high enzyme-like activity using d. functional theory calcns. Finally, we demonstrate that the developed FeN3P-SAzyme with superior peroxidase-like activity can be used as an effective therapeutic strategy for inhibiting tumor cell growth in vitro and in vivo. Therefore, SAzymes show promising potential for developing artificial enzymes that have the catalytic kinetics of natural enzymes. [graphic not available: see fulltext].
- 112Xi J. Zhang R. Wang L. Xu W. Liang Q. Li J. Jiang J. Yang Y. Yan X. Fan K. A nanozyme‐based artificial peroxisome ameliorates hyperuricemia and ischemic stroke Adv. Funct. Mater. 2021 31 9 2007130112A Nanozyme-Based Artificial Peroxisome Ameliorates Hyperuricemia and Ischemic StrokeXi, Juqun; Zhang, Ruofei; Wang, Liming; Xu, Wei; Liang, Qian; Li, Jingyun; Jiang, Jian; Yang, Yili; Yan, Xiyun; Fan, Kelong; Gao, LizengAdvanced Functional Materials (2021), 31 (9), 2007130CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)Artificial peroxisome has drawn a lot of attentions for its usefulness in fabricating protocell system and great potential in treating diseases. However, it is still a significant challenge to prep. a practicable artificial peroxisome to complement multiple and stable functions under physiol. condition. Herein, a novel strategy is reported to design an artificial peroxisome using a nanozyme to accommodate multiple enzyme-like activities that mimics those enzymes in natural peroxisome. The enzymic active sites are introduced into graphitized moieties on the shell of a hollow carbon nanozyme by doping iron and nitrogen to form Fe-N4 coordination and at. Fe cluster. With Fe clusters as reversible cofactors and Fe-N4 as prosthetic group, the resulted carbon nanozyme exhibits stable and multiple peroxisomal-like activities, including catalase, uricase, superoxide dismutase, peroxidase, and oxidase, which is referred as nanozyme-based artificial peroxisome (pero-nanozysome). This pero-nanozysome shows therapeutic effect for treating hyperuricemia and protecting neurons from free radicals generated during ischemic stroke by employing the tandem activities of uricase-catalase and superoxide-dismutase-catalase, resp. This study indicates that the pero-nanozysome is a promising candidate to design artificial peroxisome performing in vivo functions.
- 113Zhang R. Fan K. Yan X. Nanozymes: created by learning from nature Sci. China: Life Sci. 2020 63 8 1183 1200113Nanozymes: created by learning from natureZhang Ruofei; Fan Kelong; Yan Xiyun; Fan Kelong; Yan XiyunScience China. Life sciences (2020), 63 (8), 1183-1200 ISSN:.Nanozymes, a type of nanomaterials with enzyme-like activity, have shown great potential to replace natural enzymes in many fields such as biochemical detection, environmental management and disease treatment. However, the catalytic efficiency and substrate specificity of nanozymes still need improvement. To further optimize the enzymatic properties of nanozymes, recent studies have introduced the structural characteristics of natural enzymes into the rational design of nanozymes, either by employing small molecules to mimic the cofactors of natural enzymes to boost nanozymes' catalytic potential, or by simulating the active center of natural enzymes to construct the nanostructure of nanozymes. This review introduces the commonly used bio-inspired strategies to create nanozymes, aiming at clarifying the current progress and bottlenecks. Advances and challenges focusing on the research of bio-inspired nanozymes are outlined to provide ideas for the de novo design of ideal nanozymes.
- 114Zhang R. Yan X. Fan K. Nanozymes inspired by natural enzymes Acc. Mater. Res. 2021 2 7 534 547114Nanozymes Inspired by Natural EnzymesZhang, Ruofei; Yan, Xiyun; Fan, KelongAccounts of Materials Research (2021), 2 (7), 534-547CODEN: AMRCDA; ISSN:2643-6728. (American Chemical Society)A review. Nanozymes, nanomaterials with enzyme-like activities with high structural stability, adjustable catalytic activity, functional diversity, recyclability, and feasibility in large-scale prepn., have become a hot spot in the field of artificial enzymes in recent years and are expected to become potential surrogates and competitors for natural enzymes in practical applications. With the development of in-depth research and a wide range of application requirements, creating nanozymes with catalytic performance comparable to or even surpassing that of natural enzymes has been the key research topic in this field. Most of the nanozymes reported in the past were obtained based on random synthesis and screening, for which the catalytic efficiency is far inferior to that of natural enzymes. Natural enzymes that have evolved over hundreds of millions of years have developed a lot of high-efficiency catalysis know-how hidden in their structural features. To create highly active nanozymes, we assumed that there is a general structure-activity relationship between nanozymes and natural enzymes and proposed the nanozyme optimization strategy by grafting the catalytic principles of natural enzymes into the rational design of nanozymes. On the basis of this bioinspired strategy, a series of nanozymes that exhibit similar catalytic activities that are closer to or even beyond those of natural enzymes have been successfully synthesized. By now, rationally designed high-activity bioinspired nanozymes have become a hot topic in the current research on nanozymes. In this Account, we focus on recent representative research progress in the systemic design and construction of bioinspired nanozymes and are devoted to introducing strategic concepts in the bioinspired optimization of nanozymes. We show that the de novo design of nanozymes by simulating the amino acid microenvironment and using metal-free architecture and the coordination structure of metal active sites in natural enzymes is an effective strategy for significantly improving the catalytic performance of nanozymes. A future perspective of the challenges and countermeasures of bioinspired nanozymes is proposed on the basis of these achievements. We hope that the biol. inspired perception will arouse widespread interest in fundamental research and practical applications as well as provide inspiration for the rational design of nanozymes.
- 115Liu S. Lu F. Xing R. Zhu J. J. Structural effects of Fe3O4 nanocrystals on peroxidase‐like activity Chem. - Eur. J. 2011 17 2 620 625115Structural Effects of Fe3O4 Nanocrystals on Peroxidase-Like ActivityLiu, Shanhu; Lu, Feng; Xing, Ruimin; Zhu, Jun-JieChemistry - A European Journal (2011), 17 (2), 620-625, S620/1-S620/4CODEN: CEUJED; ISSN:0947-6539. (Wiley-VCH Verlag GmbH & Co. KGaA)The catalytic activity of nanocrystal catalysts depends strongly on their structures. Herein, we report three distinct structures of Fe3O4 nanocrystals, cluster spheres, octahedra, and triangular plates, prepd. by a similar hydrothermal procedure. Addnl., the three Fe3O4 nanostructures were used as peroxidase nanomimetics and the correlation between the catalytic activities and the structures was first explored by using 3,3',5,5'-tetramethylbenzidine and H2O2 as peroxidase substrates. The results showed that the peroxidase-like activities of the Fe3O4 nanocrystals were structure dependent and followed the order cluster spheres>triangular plates>octahedra; this order was closely related to their preferential exposure of catalytically active iron atoms or crystal planes. Such investigation is of great significance for peroxidase nanomimetics with enhanced activity and utilization.
- 116Puvvada N. Panigrahi P. K. Mandal D. Pathak A. Shape dependent peroxidase mimetic activity towards oxidation of pyrogallol by H2O2 RSC Adv. 2012 2 8 3270 3273116Shape dependent peroxidase mimetic activity towards oxidation of pyrogallol by H2O2Puvvada, Nagaprasad; Panigrahi, Pravas Kumar; Mandal, Dhritabrata; Pathak, AmitaRSC Advances (2012), 2 (8), 3270-3273CODEN: RSCACL; ISSN:2046-2069. (Royal Society of Chemistry)Truncated octahedron shaped magnetite nanoparticles have been prepd. via a chem. method and subsequently their shape dependent peroxidase mimetic activity has been verified using pyrogallol substrate. Their peroxidase mimetic activity has been found to be superior to that of spherical-shaped nanoparticles.
- 117Cheng X.-L. Jiang J.-S. Jiang D.-M. Zhao Z.-J. Synthesis of rhombic dodecahedral Fe3O4 nanocrystals with exposed high-energy {110} facets and their peroxidase-like activity and lithium storage properties J. Phys. Chem. C 2014 118 24 12588 12598117Synthesis of Rhombic Dodecahedral Fe3O4 Nanocrystals with Exposed High-Energy {110} Facets and Their Peroxidase-like Activity and Lithium Storage PropertiesCheng, Xun-Liang; Jiang, Ji-Sen; Jiang, Dong-Mei; Zhao, Zhen-JieJournal of Physical Chemistry C (2014), 118 (24), 12588-12598CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Large quantities of monodispersed single-crystal rhombic dodecahedral (RD) Fe3O4 nanocrystals (NCs) bounded by high-energy {110} facets were prepd. by using a 1-pot toluene-H2O 2-phase interfacial reaction method. By adjusting the Fe(acac)3 concn. and the vol. of hydrazine hydrate, their sizes could be controlled at 60-100 nm. The formation of these RD Fe3O4 NCs with exposed high-energy {110} surfaces might be the result of selective stabilization of such unstable facets by the C17H33COO- based on FTIR anal. The magnetic measurement revealed that the satn. magnetization (Ms) of the RD Fe3O4 NCs was 85 emu/g at room temp. When used as peroxidase mimetics, the prepd. RD Fe3O4 NCs showed excellent peroxidase-like catalytic activity toward oxidn. of the substrate 3,3',5,5'-tetramethylbenzidine and degrdn. of the aniline compd. methylene blue dye in the presence of H2O2. When tested as anode materials for Li-ion batteries, these as-obtained RD Fe3O4 NCs showed a high initial discharge capacity of 1147 mA-h g-1 at the c.d. of 0.2 C, a good cycle performance (362 mA-h g-1 at 0.2 C after 100 cycles and 191 mA-h g-1 at 1 C up to 130 cycles), and good rate capability at high current rates of 1-4 C.
- 118Zhao J. Zhang S. Wang D. Jia C. Yang P. Synthesis of Rhombic Dodecahedral Fe3O4 Single Crystals Towards Their High Peroxidase-Like Activity J. Nanosci. Nanotechnol. 2016 16 8 8846 8853118Synthesis of rhombic dodecahedral Fe3O4 single crystals towards their high peroxidase-like activityZhao, Jie; Zhang, Sha; Wang, Dan; Jia, Changchao; Yang, PingJournal of Nanoscience and Nanotechnology (2016), 16 (8), 8846-8853CODEN: JNNOAR; ISSN:1533-4880. (American Scientific Publishers)A solvothermal method has been developed to synthesize rhombic dodecahedral (RD) Fe3O4 single crystal structures bound by high-energy active {110} facets from inorg. Fe salts and N2H2 in N,N-dimethylformamide soln. The growth process and formation mechanism of the RD Fe3O4 structures was investigated in detail by changing different parameters. The as-synthesized products exhibit high magnetic properties with satn. magnetization value of 80 emu/g and coercive force value of 57 Oe. The RD Fe3O4 structures possessing active basal facets showed excellent peroxidase-like catalytic activity towards oxidn. of methylene blue (MB) dye in the presence of H2O2. After deposition of Au nanoparticles on the RD Fe3O4 structures, the Fe3O4/Au composites exhibit enhanced catalytic activity towards MB in the absence of H2O2. The Fe3O4 and Fe3O4/Au composites can be easily recycled from the treated water using a magnet. There was no obvious decrease of the peroxidase-like catalytic activity of the RD Fe3O4 structures after being used even for six cycles.
- 119Wan D. Li W. Wang G. Wei X. Shape-Controllable Synthesis of Peroxidase-Like Fe3O4 Nanoparticles for Catalytic Removal of Organic Pollutants J. Mater. Eng. Perform. 2016 25 10 4333 4340119Shape-Controllable Synthesis of Peroxidase-Like Fe3O4 Nanoparticles for Catalytic Removal of Organic PollutantsWan, Dong; Li, Wenbing; Wang, Guanghua; Wei, XiaobiJournal of Materials Engineering and Performance (2016), 25 (10), 4333-4340CODEN: JMEPEG; ISSN:1059-9495. (Springer)The shape of Fe3O4 nanoparticles is controlled using a simple oxidn.-pptn. method without any surfactant. The morphol. and structure of the obtained Fe3O4 nanoparticles were characterized by using x-ray diffraction, SEM, XPS, N2 physisorption and vibrating sample magnetometer. As-prepd. Fe3O4 samples showed octahedron, cube, hexagonal plate and sphere morphologies. Peroxidase-like activity of the four nanostructures was evaluated for catalytic removal of org. pollutants in the presence of H2O2, using rhodamine B as a model compd. The results showed that the H2O2-activating ability of the Fe3O4 nanocrystals was structure dependent and followed the order sphere > cube > octahedron > hexagonal plate, which was closely related to their surface FeII/FeIII ratios or crystal planes. The reusability of Fe3O4 spheres was also investigated after five successive runs, which demonstrated the promising application of the catalyst in the degrdn. of org. pollutants. This investigation is of great significance for the heterogeneous catalysts with enhanced activity and practical application.
- 120Gao Y. Wei Z. Li F. Yang Z. M. Chen Y. M. Zrinyi M. Osada Y. Synthesis of a morphology controllable Fe3O4 nanoparticle/hydrogel magnetic nanocomposite inspired by magnetotactic bacteria and its application in H2O2 detection Green Chem. 2014 16 3 1255 1261120Synthesis of a morphology controllable Fe3O4 nanoparticle/hydrogel magnetic nanocomposite inspired by magnetotactic bacteria and its application in H2O2 detectionGao, Yang; Wei, Zhao; Li, Fei; Yang, Zhi Mao; Chen, Yong Mei; Zrinyi, Miklos; Osada, YoshihitoGreen Chemistry (2014), 16 (3), 1255-1261CODEN: GRCHFJ; ISSN:1463-9262. (Royal Society of Chemistry)Owing to the shape-dependent catalytic activity of Fe3O4 nanoparticles, controlling their morphol. is of great significance. In this work, we propose a simple, nontoxic, water-based strategy for the fabrication of magnetic nanoparticle/hydrogel nanocomposites in which highly cryst. Fe3O4 nanooctahedra can be fabricated in situ within a neg. charged hydrogel matrix. The morphol. of the Fe3O4 nanocrystals can be easily tuned by adjusting the crosslinking concn. of the hydrogel. Furthermore, the catalytic activities of the magnetic nanocomposites were studied, and the magnetic nanocomposite loaded with Fe3O4 nanooctahedra exhibited excellent catalytic activity and provided a sensitive response for H2O2 detection. This scalable approach for the fabrication of magnetic nanocomposites, loaded with morphol. controllable Fe3O4 nanoparticles, potentially promotes their applications in biotechnol. and environmental chem.
- 121Peng Y. Wang Z. Liu W. Zhang H. Zuo W. Tang H. Chen F. Wang B. Size-and shape-dependent peroxidase-like catalytic activity of MnFe2O4 Nanoparticles and their applications in highly efficient colorimetric detection of target cancer cells Dalton Trans. 2015 44 28 12871 12877121Size- and shape-dependent peroxidase-like catalytic activity of MnFe2O4 Nanoparticles and their applications in highly efficient colorimetric detection of target cancer cellsPeng, Yunhua; Wang, Zhiyi; Liu, Weisheng; Zhang, Haoli; Zuo, Wei; Tang, Huiang; Chen, Fengjuan; Wang, BaoduiDalton Transactions (2015), 44 (28), 12871-12877CODEN: DTARAF; ISSN:1477-9226. (Royal Society of Chemistry)The catalytic activity of nanocrystal catalysts depends strongly on their chem. compn., size, and shape. Herein, the authors report four different sizes and shapes of MnFe2O4 nanoparticles (NPs) prepd. by a hydrothermal procedure. In addn., the size- and shape-dependent peroxidase-like activity of these NPs was first explored using 3,3',5,5'-tetramethyl-benzidine and H2O2 as peroxidase substrates. The peroxidase-like activities of the MnFe2O4 NPs were size- and shape-dependent and followed the order of 4 nm (spherical) > 18 nm (plate-like) > 27 nm (near-cubic) > 16 nm (spherical); this order was closely related to their surface-to-vol. ratio and atom arrangements. Such a study is of great significance for peroxidase nanomimetics with enhanced activity and use. Furthermore, folic acid (FA)-conjugated MnFe2O4 NPs allow the detection of folate receptor-rich cancer cells. Such study can be widely used for the identification of important target mols.
- 122Mao J. Hou X. Wang X. Hu S. Xiang L. The cubic aggregated CoFe2O4 nanoparticle anode material for lithium ion battery with good performance Mater. Lett. 2015 161 652 655122The cubic aggregated CoFe2O4 nanoparticle anode material for lithium ion battery with good performanceMao, Junwei; Hou, Xianhua; Wang, Xinyu; Hu, Shejun; Xiang, LiangzhongMaterials Letters (2015), 161 (), 652-655CODEN: MLETDJ; ISSN:0167-577X. (Elsevier B.V.)Novel cubic aggregated CoFe2O4 nanoparticles are successfully synthesized by hydrothermal method. Electrochem. results show that the CoFe2O4 nanoparticles exhibit good cycling performance and excellent rate capability. The 1st discharge and charge capacities of these nanoparticles are 1672.8 mA h g-1 and 1309.1 mA h g-1, resp., with an initial coulombic efficiency of 78.3%. The electrode can retain a high capacity of 1133.5 mA h g-1 after 120 cycles at a c.d. of 100 mA g-1. Also, it could still maintain a reversible capacity of 679 mA h g-1 even at a high c.d. of 3.2 A g-1. The improved electrochem. performance can be ascribed to the hierarchical cubic aggregated structural with large sp. surface area and lots of interspaces between the particles, which not only can effectively increase the active surface area, but also can make better penetration of the electrolyte and accommodate the vol. expansion.
- 123Tian T. Ai L. Liu X. Li L. Li J. Jiang J. Synthesis of hierarchical FeWO4 architectures with {100}-faceted nanosheet assemblies as a robust biomimetic catalyst Ind. Eng. Chem. Res. 2015 54 4 1171 1178123Synthesis of Hierarchical FeWO4 Architectures with {100}-Faceted Nanosheet Assemblies as a Robust Biomimetic CatalystTian, Tian; Ai, Lunhong; Liu, Xiaomin; Li, Lili; Li, Jie; Jiang, JingIndustrial & Engineering Chemistry Research (2015), 54 (4), 1171-1178CODEN: IECRED; ISSN:0888-5885. (American Chemical Society)Surface structure is of special significance to the heterogeneous catalysis, because catalysis is primarily a surface phenomenon. Therefore, a comprehensive understanding of the relationship between the catalytic activity and surface structure is of great importance. Herein, hierarchical FeWO4 architectures assembled by {100}-faceted nanosheets have been successfully synthesized by a facile one-pot solvothermal method and employed as a novel biomimetic catalyst mimicking enzyme for the first time. Due to the large d. of terminal iron atoms per unit surface area on the {100} facet, the FeWO4 architectures exhibited excellent peroxidase-like activity for the catalytic oxidn. of peroxidase substrate 3,3',5,5'-tetramethylbenzidine (TMB) to produce a blue color reaction in the presence of H2O2. Using the FeWO4 architectures as peroxidase mimetics, a colorimetric sensor for H2O2 was developed, which provided good response toward H2O2 concn. over a range from 0.95 to 66.7 μM with a detection limit of 0.28 μM. The proposed colorimetric method can be further extended for the sensitive detection of glucose by coupling with glucose oxidase (GOx). The limit of detection for glucose was detd. to be as low as 0.67 μM and the linear range was from 4 to 60 μM. It is believed that these findings are useful in understanding the peroxidase-like activity of catalysts with facet exposure and suggest a new strategy to pursue for highly active peroxidase-like catalysts.
- 124Liu B. Liu J. Surface modification of nanozymes Nano Res. 2017 10 4 1125 1148124Surface modification of nanozymesLiu, Biwu; Liu, JuewenNano Research (2017), 10 (4), 1125-1148CODEN: NRAEB5; ISSN:1998-0000. (Springer GmbH)Nanoparticles and proteins are similar in a no. of aspects, and using nanoparticles to mimic the catalytic function of enzymes is an interesting yet challenging task. Impressive developments have been made over the past two decades on this front. The term nanozyme was coined to refer to nanoparticlebased enzyme mimics. To date, many different types of nanozymes have been reported to catalyze a broad range of reactions for chem., anal., and biomedical applications. Since chem. reactions happen mainly on the surface of nanozymes, an interesting aspect for investigation is surface modification. In this review, we summarize three types of nanozyme materials catalyzing various reactions with a focus on their surface chem. For metal oxides, cerium oxide and iron oxide are discussed as they are the most extensively studied. Then, gold nanoparticles and graphene oxide are reviewed to represent metallic and carbon nanomaterials, resp. Types of modifications include ions, small mols., and polymers mainly by physisorption, while in a few cases, covalent modifications were also employed. The functional aspect of such modification is to improve catalytic activity, substrate specificity, and stability. Future perspectives of this field are speculated at the end of this review. [Figure not available: see fulltext.].
- 125Bruce I. J. Taylor J. Todd M. Davies M. J. Borioni E. Sangregorio C. Sen T. Synthesis, characterisation and application of silica-magnetite nanocomposites J. Magn. Magn. Mater. 2004 284 145 160125Synthesis, characterization and application of silica-magnetite nanocompositesBruce, Ian J.; Taylor, James; Todd, Michael; Davies, Martin J.; Borioni, Enrico; Sangregorio, Claudio; Sen, TapasJournal of Magnetism and Magnetic Materials (2004), 284 (), 145-160CODEN: JMMMDC; ISSN:0304-8853. (Elsevier B.V.)Silica-magnetite composites were prepd. for eventual applications in biomol. sepns. (nucleic acids). Their prodn. on large scale has been optimized and they have been extensively characterized in a phys. and chem. context. They perform at least as well, if not better than a com. available equiv. at adsorbing and eluting DNA. Several methods for the prepn. of magnetite were compared in order to select one, which produced particles, possessing high magnetic susceptibility, low rate of sedimentation and good chem. stability. Of the main methods studied: (i) oxidative hydrolysis of iron(II) sulfate in alk. media, (ii) alk. hydrolysis of iron(II) and iron(III) chloride solns., and (iii) pptn. from iron(II) and iron(III) chloride solns. by hydrolysis of urea, method (i) produced the best' magnetite particles. Silica-magnetite composites were prepd. using the best' magnetite, and, for comparison, two methods for depositing silica were used to coat the silica onto magnetite nanoparticles, from silicic acid at pH 10 and by acid hydrolysis of tetraethoxysilane (TEOS) at 90°. The best method for yielding silica-magnetite composites that worked well in DNA adsorption and elution proved to be that involving silicic acid and this material could be made in 20 g batch sizes. Silica-magnetite composites from the two methods proved to have distinct and different phys. and chem. properties. All magnetite and silica-magnetite samples were fully characterized for their relative chem. compn. using Fourier-transform IR, XRF and thermo-gravimetric anal. Their phys. characteristics were detd. using SEM and N2 adsorption and Mossbauer spectroscopy was used to confirm the identity of the iron oxides produced. Selected samples were comparatively tested for their ability to adsorb, and subsequently elute, 2-deoxyguanosine-5-monophosphate (GMP) and its non-phosphorylated analog 2-deoxyguanosine (G) and a range of sequence defined oligonucleotides (NAs) and sheared salmon sperm DNA. It was found that magnetite readily adsorbed GMP via the GMP phosphate anion in water, whereas silica did not, due to electrostatic repulsion between the neg. charged surface of silica and the GMP. Both magnetite and silica magnetite were further tested in adsorption studies of G and GMP in different chaotropic media, 4 M sodium chloride or 4 M ammonium sulfate. The high salt conditions aided binding of GMP silica magnetite but inhibited adsorption to magnetite presumably due to competition for binding sites on the magnetite's surface by the chaotrope anions. Interestingly, the results from NAs binding studies indicated that sequence appeared to play an important role in adsorption of the different species to silica-magnetite composites. This may indicate a contribution by hydrophobic interactions to the binding mechanism. Multiple depositions of silica onto magnetite performed by deposition from silicic acid at pH 10 did not appear to greatly increase the composite percentage represented by silica while composite produced by the acid hydrolysis of TEOS at 90° did. However, it appeared that the silica deposited by the first method represented a complete coating of the magnetite core while the second method yielded a porous or incomplete coating. In comparison with com. available silica-magnetite composite in DNA adsorption and elution, the material was obsd. to perform approx. 10% more efficiently. These findings indicate that it is possible to produce a consistent and cheap silica-magnetite nanoparticle on relatively large scale (greater than 20 g batch size) which is at least as good as, if not better than, a com. available alternative.
- 126Sun Y. Duan L. Guo Z. DuanMu Y. Ma M. Xu L. Zhang Y. Gu N. An improved way to prepare superparamagnetic magnetite-silica core-shell nanoparticles for possible biological application J. Magn. Magn. Mater. 2005 285 1-2 65 70There is no corresponding record for this reference.
- 127Zhang X.-Q. Gong S.-W. Zhang Y. Yang T. Wang C.-Y. Gu N. Prussian blue modified iron oxide magnetic nanoparticles and their high peroxidase-like activity J. Mater. Chem. 2010 20 24 5110 5116127Prussian blue modified iron oxide magnetic nanoparticles and their high peroxidase-like activityZhang, Xiao-Qing; Gong, Shang-Wenyan; Zhang, Yu; Yang, Ting; Wang, Chun-Yu; Gu, NingJournal of Materials Chemistry (2010), 20 (24), 5110-5116CODEN: JMACEP; ISSN:0959-9428. (Royal Society of Chemistry)Prussian blue (PB) modified γ-Fe2O3 magnetic nanoparticles (MNPs) featuring varying PB proportions were synthesized and characterized by TEM, FTIR, UV-vis, EDS, XRD and XPS. The magnetic properties and peroxidase-like catalytic activity of the synthesized nanoparticles were investigated. With increasing PB content, the magnetism could still maintain a high level. Peroxidase-like activity was enhanced as the PB proportion increased. Catalysis was found to follow Michaelis-Menten kinetics. The calcd. kinetic parameters exhibited strong affinity with substrates and high catalytic activity, which are three orders of magnitudes larger than that for magnetite nanoparticles of similar size. Based on the high activity, an enzyme immunoassay model was established: staphylococcal protein A (SPA) was conjugated onto the surface of the nanoparticles to construct a new nanoprobe which was employed to detect IgG immobilized to 96-well plates. The results presented a linear absorbance enhancement with concn. of IgG, suggesting that PBMNPs serve as an inexpensive horseradish peroxidase (HRP) mimic enzyme with potential applications in bio-detection.
- 128Garg D. Kaur M. Sharma S. Verma V. Effect of CTAB coating on structural, magnetic and peroxidase mimic activity of ferric oxide nanoparticles Bull. Mater. Sci. 2018 41 5 1 9128Effect of CTAB coating on structural, magnetic and peroxidase mimic activity of ferric oxide nanoparticlesGarg, Dikshit; Kaur, Manpreet; Sharma, Sucheta; Verma, VibhaBulletin of Materials Science (2018), 41 (5), 1-9CODEN: BUMSDW; ISSN:0250-4707. (Indian Academy of Sciences)In the present work, pristine and cetyl tri-Me ammonium bromide (CTAB)-coated ferric oxide nanoparticles (CTAB@Fe2O3NPs) were synthesized and studied as enzyme mimics. The wt./wt. ratio of Fe2O3 to CTAB was varied as 1:1 and 1:2. Transmission electron microscopic anal. revealed that pristine NPs had an av. size of 50 nm, whereas the presence of CTAB resulted in the formation of nanorods with length of 130 nm. BET studies confirmed enhancement of surface area on CTAB coating, which was max. for wt./wt. ratio 1:1. The synthesized pristine NPs and CTAB-coated NPs were evaluated for their peroxidase mimic activity using o-dianisidine dihydrochloride as substrate. Optimum pH, temp., substrate and NPs concn. for the reaction were 1, 25oC, 0.16mgml-1 and 1mgml-1, resp. Peroxidase mimic activity of CTAB@Fe2O3NPs (wt./wt. 1:1) was higher than that of pristine NPs. However, further increase in CTAB coating (wt./wt. 1:2) resulted in lowering of peroxidase mimic activity. Kinetic anal. was carried out at optimized conditions; max. velocity (Vmax) and Michaelis const. (Km) value of CTAB@Fe2O3NPs at 1:1 wt./wt. ratio were 7.69 mM and 1.12μmols-1, resp.
- 129Fan Y. Shi W. Zhang X. Huang Y. Mesoporous material-based manipulation of the enzyme-like activity of CoFe2O4 nanoparticles J. Mater. Chem. A 2014 2 8 2482 2486129Mesoporous material-based manipulation of the enzyme-like activity of CoFe2O4 nanoparticlesFan, Yingwei; Shi, Wenbing; Zhang, Xiaodan; Huang, YumingJournal of Materials Chemistry A: Materials for Energy and Sustainability (2014), 2 (8), 2482-2486CODEN: JMCAET; ISSN:2050-7496. (Royal Society of Chemistry)Mesoporous material supported CoFe2O4 magnetic nanoparticles possess unique peroxidase/oxidase-like activity, and react with luminol to yield a novel chemiluminescence without the need of H2O2. Their oxidase-like activity shows pH and support dependence, and could be reversibly controlled by their pH. This offers a new method for manipulating the enzyme-like activity of nanoparticles.
- 130Liu C.-H. Yu C.-J. Tseng W.-L. Fluorescence assay of catecholamines based on the inhibition of peroxidase-like activity of magnetite nanoparticles Anal. Chim. Acta 2012 745 143 148130Fluorescence assay of catecholamines based on the inhibition of peroxidase-like activity of magnetite nanoparticlesLiu, Cheng-Hao; Yu, Cheng-Ju; Tseng, Wei-LungAnalytica Chimica Acta (2012), 745 (), 143-148CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)We report a fluorescence approach for the highly selective and sensitive detection of catecholamines using magnetite nanoparticles (Fe3O4 NPs) in the presence of Amplex UltraRed (AUR) and H2O2. Fe3O4 NPs catalyze H2O2-mediated oxidn. of AUR. The resulting product fluoresces (excitation/emission maxima, ca. 568/587 nm) more strongly, relative to AUR. When catecholamines bind to Fe3O4, the complexes that are formed induce decreased activity of Fe3O4 NPs, mediated through the coordination between Fe3+ on the NP surface and the catechol moiety of catecholamines. As a result, Fe3O4 NPs-catalyzed H2O2-mediated oxidn. of AUR is inhibited by catecholamines. The limits of detection for dopamine (DA), L-DOPA, norepinephrine, and epinephrine were 3 nM, 3 nM, 3 nM, and 6 nM, resp. The Fe3O4 NPs-H2O2-AUR probe exhibited high selectivity (>1000-fold) toward catecholamines over other tested biomols. that commonly exist in urine. Four catecholamines had similar sensitivity because the inhibition of the Fe3O4 NPs activity relies on the presence of the catechol moiety. This approach also allowed the detn. of tyrosinase activity because tyrosinase catalyzes the conversion of L-tyrosine to L-DOPA. We validated the practicality of the use of the Fe3O4 NPs-H2O2-AUR probe for the detn. of the concns. of DA in urine samples.
- 131Chen C. Lu L. Zheng Y. Zhao D. Yang F. Yang X. A new colorimetric protocol for selective detection of phosphate based on the inhibition of peroxidase-like activity of magnetite nanoparticles Anal. Methods 2015 7 1 161 167131A new colorimetric protocol for selective detection of phosphate based on the inhibition of peroxidase-like activity of magnetite nanoparticlesChen, Chuanxia; Lu, Lixia; Zheng, Yu; Zhao, Dan; Yang, Fan; Yang, XiurongAnalytical Methods (2015), 7 (1), 161-167CODEN: AMNEGX; ISSN:1759-9679. (Royal Society of Chemistry)A simple colorimetric assay for phosphate ion (Pi) has been established based on analyte-induced inhibition of the magnetite nanoparticles (MNPs)-catalyzed oxidn. of 3,3',5,5'-tetramethylbenzidine (TMB) in the presence of H2O2. The Fe3O4 MNPs can catalyze the H2O2-mediated oxidn. of TMB and yields a blue oxidized product, which exhibits a max. absorption at 652 nm. Pi could be absorbed on the surface of the Fe3O4 MNPs through coordinating with Fe3+, inducing a reduced colorimetric signal. The colorimetric signal change (ΔA652) in this process was proportional to the concn. of Pi, ranging from 0.2 μM to 200 μM. The limit of detection (S/N = 3) was as low as 0.11 μM. The as-proposed Fe3O4 MNPs-TMB-H2O2 probe exhibited a high selectivity toward Pi over other relevant ions that commonly exist in water and has been applied to Pi detection in drinking water, ground water and lake water samples with satisfactory results.
- 132Park K. S. Kim M. I. Cho D. Y. Park H. G. Label‐free colorimetric detection of nucleic acids based on target‐induced shielding against the peroxidase‐mimicking activity of magnetic nanoparticles Small 2011 7 11 1521 1525132Label-Free Colorimetric Detection of Nucleic Acids Based on Target-Induced Shielding Against the Peroxidase-Mimicking Activity of Magnetic NanoparticlesPark, Ki Soo; Kim, Moon Il; Cho, Dae-Yeon; Park, Hyun GyuSmall (2011), 7 (11), 1521-1525CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)A new and simple colorimetric methodol. for the detection of nucleic acids has been developed, which relies on target-DNA-induced inhibition of the peroxidase-mimicking activity of MNPs. This is the first example in which the peroxidase activity of MNPs serves as the basis for colorimetric detection of nucleic acids. Significant features of the new assay system are its simplicity and cost-effectiveness. Importantly, assays can be accomplished using naked-eye detection within a 30 min period. Furthermore, the new assay system does not require surface modification of either the MNPs or DNA, and a salt-induced aggregation step is not needed: these are time-consuming and costly manipulations that are generally required in AuNP-based DNA detection procedures. An application of this simple yet efficient colorimetric detection system was successfully demonstrated by its use in diagnosing C. trachomatis using a human urine sample.
- 133Li X. Wen F. Creran B. Jeong Y. Zhang X. Rotello V. M. Colorimetric protein sensing using catalytically amplified sensor arrays Small 2012 8 23 3589 3592133Colorimetric protein sensing using catalytically amplified sensor arraysLi, Xiaoning; Wen, Fang; Creran, Brian; Jeong, Youngdo; Zhang, Xinrong; Rotello, Vincent M.Small (2012), 8 (23), 3589-3592CODEN: SMALBC; ISSN:1613-6810. (Wiley-VCH Verlag GmbH & Co. KGaA)Dopamine- or trimethylammonium- functionalized Fe3O4 nanoparticles were fabricated and used for array-based sensing of ten proteins.
- 134Le T. N. Tran T. D. Kim M. I. A Convenient Colorimetric Bacteria Detection Method Utilizing Chitosan-Coated Magnetic Nanoparticles Nanomaterials 2020 10 1 92134A convenient colorimetric bacteria detection method utilizing chitosan-coated magnetic nanoparticlesLe, Thao Nguyen; Tran, Tai Duc; Kim, Moon IlNanomaterials (2020), 10 (1), 92CODEN: NANOKO; ISSN:2079-4991. (MDPI AG)An effective novel strategy to detect bacteria is promising because it may improve human health by allowing early diagnosis and timely treatment of bacterial infections. Here, we report a simple, reliable, and economical colorimetric assay using the peroxidase-like activity of chitosan-coated iron oxide magnetic nanoparticles (CS-MNPs). When CS-MNPs are incubated with a sample contg. bacterial cells such as the gram-neg. Escherichia coli or the gram-pos. Staphylococcus aureus, the neg.-charged bacterial membrane interacts with pos.-charged chitosan on the surface of CS-MNPs, thus resulting in significant redn. of their peroxidase-like activity presumably by a hindrance in the accessibility of the neg. charged substrate, 2-2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) to the pos.-charged CS-MNPs. This simple colorimetric strategy allowed the rapid detection of bacterial cells down to 104 CFU mL-1 by the naked eye and 102 CFU mL-1 by spectrophotometry within 10 min. Based on the results, we anticipate that the CS-MNPs-based assay has great potential for the on-site diagnosis of bacterial infections in facility-limited or point-of-care testing (POCT) environments.
- 135Huang X. Xu C. Ma J. Chen F. Ionothermal synthesis of Cu-doped Fe3O4 magnetic nanoparticles with enhanced peroxidase-like activity for organic wastewater treatment Adv. Powder Technol. 2018 29 3 796 803135Ionothermal synthesis of Cu-doped Fe3O4 magnetic nanoparticles with enhanced peroxidase-like activity for organic wastewater treatmentHuang, Xuanlin; Xu, Cong; Ma, Jiping; Chen, FengxiAdvanced Powder Technology (2018), 29 (3), 796-803CODEN: APTEEE; ISSN:0921-8831. (Elsevier B.V.)The intrinsic peroxidase-like activity of magnetite magnetic nanoparticles (Fe3O4 MNPs) has to be improved to activate H2O2 under mild conditions for practical applications. Herein copper-doped Fe3O4 (Fe3-xCuxO4, x: 0.06-0.23) MNPs were successfully prepd. by oxidative pptn.-combined ionothermal synthesis and characterized by XRD, VSM, XPS, BET, etc. The Cu2+ dopants are mainly substituted for Fe2+ ions at octahedral sites and significantly surface-enriched, which expedite the Fe3+/Fe2+ redox cycling and enhance the H2O2-activation ability of Fe3-xCuxO4 MNPs. Kinetic study showed that the decompn. of H2O2 on Fe2.88Cu0.12O4 was much faster than that on the undoped Fe3O4 (0.584 vs. 0.153 h-1 at 25 °C) due to the lower activation energy of the former (55.3 vs. 62.1 kJ/mol). The enhanced H2O2-activation ability upon copper doping was exploited to efficiently degrade recalcitrant org. pollutants (e.g., rhodamine B) with H2O2 at pH ∼ 7 and 25 °C on Fe2.88Cu0.12O4 with good stability and reusability (16 h tested in eight cycles).
- 136Moreno Maldonado A. C. Winkler E. L. Raineri M. Toro Córdova A. Rodríguez L. M. Troiani H. E. Mojica Pisciotti M. L. Mansilla M. V. Tobia D. Nadal M. S. Torres T. E. De Biasi E. Ramos C. A. Goya G. F. Zysler R. D. Lima E. Free-Radical Formation by the Peroxidase-Like Catalytic Activity of MFe2O4 (M = Fe, Ni, and Mn) Nanoparticles J. Phys. Chem. C 2019 123 33 20617 20627136Free-Radical Formation by the Peroxidase-Like Catalytic Activity of MFe2O4 (M = Fe, Ni, and Mn) NanoparticlesMoreno Maldonado, Ana Carolina; Winkler, Elin L.; Raineri, Mariana; Toro Cordova, Alfonso; Rodriguez, Luis M.; Troiani, Horacio E.; Mojica Pisciotti, Mary Luz; Mansilla, Marcelo Vasquez; Tobia, Dina; Nadal, Marcela S.; Torres, Teobaldo E.; De Biasi, Emilio; Ramos, Carlos A.; Goya, Gerardo F.; Zysler, Roberto D.; Lima, EnioJournal of Physical Chemistry C (2019), 123 (33), 20617-20627CODEN: JPCCCK; ISSN:1932-7447. (American Chemical Society)Ferrite magnetic nanoparticles (MNPs) have peroxidase-like activity and thus catalyze the decompn. of H2O2-producing reactive oxygen species (ROS). Increasingly important applications of these ferrite MNPs in biol. and medicine require that their morphol., physicochem., and magnetic properties need to be strictly controlled. Usually, the tuning of their magnetic properties is achieved by the replacement of Fe by other 3d metals, such as Mn or Ni. Here, we studied the catalytic activity of ferrite MNPs (MFe2O4, M = Fe2+/Fe3+, Ni, and Mn) with the mean diam. ranging from 10 to 12 nm. Peroxidase-like activity was studied by ESR (EPR) using the spin-trap 5,5-dimethyl-1-pyrroline N-oxide at different pHs (4.8 and 7.4) and temps. (25 and 40 °C). We identified an enhanced amt. of hydroxyl (•OH) and perhydroxyl (•OOH) radicals for all samples, compared to a blank soln. Quant. studies show that [•OH] is the dominant radical formed for Fe3O4, which is strongly reduced with the concomitant oxidn. of Fe2+ or its substitution (Ni or Mn). A comparative anal. of the EPR data against in vitro prodn. of ROS in microglial BV2 cell culture provided addnl. insights regarding the catalytic activity of ferrite MNPs, which should be considered if biomedical uses are intended. Our results contribute to a better understanding of the role played by different divalent ions in the catalytic activity of ferrite nanoparticles, which is very important because of their use in biomedical applications.
- 137Verma V. Kaur M. Sharma S. Superoxide dismutase mimic activity of spinel ferrite MFe2O4 (M= Mn, Co and Cu) nanoparticles Bull. Mater. Sci. 2019 42 3 1 6137Effect of vanadyl doping on relative intensities of factor group split Raman bands in potassium oxalate monohydratePant, Navin; Verma, A. L.; Pandey, S. D.Bulletin of Materials Science (2019), 42 (4), 1-7CODEN: BUMSDW; ISSN:0250-4707. (Indian Academy of Sciences)We report Raman spectral studies of pure potassium oxalate monohydrate, K2C2O4·H2O (POM) and its doped varieties with 1 and 5% vanadyl concns. at room temp. Apart from many other interesting changes in the Raman spectra on doping with VO2+, one of the most intriguing observations is the change in relative intensities of factor group split components of several Raman bands including some bands in the lattice mode region. We have explained the relative intensity changes of factor group split components in terms of locking of adjacent C2O4 - H2O - C2O4 - H2O chains due to vanadyl doping in the POM lattice. The redn. in the intensities of water bands on vanadyl doping also supports this view.
- 138Zhang C. Wang X. Dong X. Mei L. Wu X. Gu Z. Zhao Y. X-ray-facilitated redox cycling of nanozyme possessing peroxidase-mimicking activity for reactive oxygen species-enhanced cancer therapy Biomaterials 2021 276 121023138X-ray-facilitated redox cycling of nanozyme possessing peroxidase-mimicking activity for reactive oxygen species-enhanced cancer therapyZhang, Chenyang; Wang, Xin; Dong, Xinghua; Mei, Linqiang; Wu, Xiaochen; Gu, Zhanjun; Zhao, YuliangBiomaterials (2021), 276 (), 121023CODEN: BIMADU; ISSN:0142-9612. (Elsevier Ltd.)Nanomaterials with shifting or mixed redox states is one of the most common studied nanozyme with peroxidase-like activity for chemodynamic therapy (CDT), which can decomp. hydrogen peroxide (H2O2) of tumor microenvironment into highly toxic reactive oxygen species (ROS) by a nano-catalytic way. However, most of them exhibit an insufficient catalytic efficiency due to their dependence on catalytic condition. Herein, a potential methodol. is proposed to enhance their enzymic activity by accelerating the redox cycling of these nanomaterials with shifting or mixed redox states in the presence of X-ray. In this study, the nanocomposite consisting of SnS2 nanoplates and Fe3O4 quantum dots with shifting or mixed redox states (Fe2+/Fe3+) is used to explore the strategy. Under external X-ray irradn., SnS2 cofactor as electron donor can be triggered to transfer electrons to Fe3O4, which promotes the regeneration of Fe2+ sites on the surface of the Fe3O4. Consequently, the regenerated Fe2+ sites react with the overexpressed H2O2 to persistently generate ROS for enhanced tumor therapy. The designed nanocomposite displays the synergistic effects of radiotherapy and CDT. The strategy provides a new avenue for the development of artificial nanozymes with shifting or mixed redox states in precise cancer treatments based on X-ray-enhanced enzymic efficacy.
- 139Zhu M. Dai Y. Wu Y. Liu K. Qi X. Sun Y. Bandgap control of α-Fe2O3 nanozymes and their superior visible light promoted peroxidase-like catalytic activity Nanotechnology 2018 29 46 465704139Bandgap control of α-Fe2O3 nanozymes and their superior visible light promoted peroxidase-like catalytic activityZhu, Mingyun; Dai, Yunqian; Wu, Yanan; Liu, Ken; Qi, Xiaomian; Sun, YuemingNanotechnology (2018), 29 (46), 465704/1-465704/10CODEN: NNOTER; ISSN:1361-6528. (IOP Publishing Ltd.)Iron oxide nanoparticles (NPs) possessing peroxidase-like catalytic activity have been widely explored in recent decades, owing to their high stability against harsh conditions, low cost, flexibility in structure design and compn., adjustable activities and excellent biocompatibility in comparison with natural enzymes. Recently, a lot of great achievements have been made in this field of iron oxide nanozymes, however, this research has now reached a bottleneck in that the max. activity enhancement is difficult to achieve via a material design. Hence, in this work, visible light was introduced to improve the peroxidase-like activity of Fe2O3 NPs synthesized via a combination of electrospinning technol. and hydrothermal reaction. Our results showed that with the assistance of visible light, Fe2O3 NPs exhibited at least 2.2-fold higher peroxidase activity than those tested under darkness, confirming the superiorly visible light promoted peroxidase-like catalytic activity of Fe2O3 NPs. Furthermore, the affinity and max. reaction velocity of Fe2O3 nanoflowers (bandgap,,Q=,,Q1.78 eV) towards 3,3',5,5'- tetramethylbenanozymeidine (TMB) were at least over 3.7 and 4.3 times greater than in Fe2O3 nanocubes (bandgap = 2.08 eV), suggesting that the reaction performance of semiconductors could be controlled by proper adjustment of the bandgap. Moreover, the Fe2O3 NPs were also successfully utilized to detect glucose. Herein, we believe that the present work exhibits a fascinating perspective for peroxidase-like catalytic fields.
- 140Zhang Y. Wang X. Chu C. Zhou Z. Chen B. Pang X. Lin G. Lin H. Guo Y. Ren E. Lv P. Shi Y. Zheng Q. Yan X. Chen X. Liu G. Genetically engineered magnetic nanocages for cancer magneto-catalytic theranostics Nat. Commun. 2020 11 1 5421140Genetically engineered magnetic nanocages for cancer magneto-catalytic theranosticsZhang Yang; Wang Xiaoyong; Chu Chengchao; Pang Xin; Lin Gan; Lin Huirong; Ren En; Lv Peng; Shi Yesi; Zheng Qingbing; Yan Xiaohui; Liu Gang; Wang Xiaoyong; Zheng Qingbing; Liu Gang; Zhou Zijian; Chen Xiaoyuan; Chen Biaoqi; Guo YuxinNature communications (2020), 11 (1), 5421 ISSN:.The clinical applications of magnetic hyperthermia therapy (MHT) have been largely hindered by the poor magnetic-to-thermal conversion efficiency of MHT agents. Herein, we develop a facile and efficient strategy for engineering encapsulin-produced magnetic iron oxide nanocomposites (eMIONs) via a green biomineralization procedure. We demonstrate that eMIONs have excellent magnetic saturation and remnant magnetization properties, featuring superior magnetic-to-thermal conversion efficiency with an ultrahigh specific absorption rate of 2390 W/g to overcome the critical issues of MHT. We also show that eMIONs act as a nanozyme and have enhanced catalase-like activity in the presence of an alternative magnetic field, leading to tumor angiogenesis inhibition with a corresponding sharp decrease in the expression of HIF-1α. The inherent excellent magnetic-heat capability, coupled with catalysis-triggered tumor suppression, allows eMIONs to provide an MRI-guided magneto-catalytic combination therapy, which may open up a new avenue for bench-to-bed translational research of MHT.



