Recent Advances in Disposable Electrochemical Sensors
- Manjushree S. G. Manjushree S. G.Department of Chemistry, Siddaganga Institute of Technology, Tumkur–572103, Karnataka, IndiaMore by Manjushree S. G.
- and
- Prashanth S. Adarakatti*Prashanth S. Adarakatti*Email: [email protected]Department of Chemistry, SVM Arts, Science and Commerce College, Ilkal–587125, India, affiliated to Rani Channamma University, Belagavi, Karnataka, IndiaMore by Prashanth S. Adarakatti
Abstract
Disposable sensors are becoming increasingly used in the healthcare industry for quick and affordable biomarker screening and monitoring. The electrochemical detection technique makes it simple to scale up and miniaturize inexpensive disposable sensors, can be employed for accurate biomarker measurement at or close to the patient site. The paper-based analytic tools make it easier to sample in situ, search for biomarkers, and detect them multiplexedly. This chapter describes the high-throughput fabrication methods used to create disposable electrodes, such as paper based electrodes, screen-printing, ink jet printing, and laser induced printing, and goes into detail on how they can be utilized for various enzymatic, nonenzymatic, and aptamer-based sensing. The effectiveness of nanostructured materials in enhancing electrochemical sensor performance is outlined. Additionally, current innovations in hybrid wearable devices capable of sensing various analytes.
This publication is licensed for personal use by The American Chemical Society.
1 Introduction
2 Types of Electrodes
2.1 Glassy Carbon Electrode
2.2 Carbon Paste Electrodes
2.3 Basal and Edge Plane Pyrolytic Graphite Electrodes
Figure 1
2.4 Disposable Electrodes
2.4.1 Paper-Based Disposable Electrochemical Sensors (PES) for Point-of-Care Testing
2.4.2 Screen-Printed Disposable Sensors
3 Indirect Electrochemical Immunoassays
3.1 Direct Electrochemical Immunoassays
3.2 Ink-Jet Printed Disposable Sensors
3.3 Construction of Inkjet-Printed Sensors
Figure 2
3.4 Laser-Induced Graphene (LIG) Based Disposable Sensors
3.5 Fabrication and Engineering of LIG
Figure 3
| Paper Substrate | Target | Types of Electrochemical Analysis | Ref. |
|---|---|---|---|
| Whatman RC60 generated membrane filter | Bromide, Iodide, Chloride | Cyclic voltammetry | ( 73) |
| Nitrocellulose membrane | Acetomenophen | Cyclic voltammetry and differential pulse voltammetry | ( 74) |
| Filter papers (qualitative, 102, 15 mm) | Glucose | Cyclic voltammetry and chronopotentiometry | ( 75) |
| Whatman chromatography paper 3mm | Staphylococc aureusus | Differential pulse voltammetry | ( 76) |
| Analyte | Modifier | Detection | Linear range | Detection limit | Ref. |
|---|---|---|---|---|---|
| L or D Amino acids | rhodinized carbon | Amperometric | 0.1–1.0 mM | 0.15–0.47 mM | ( 77) |
| Secreted Placental alkaline phosphate (SPAP) | 2-Naphthol Osteryoung | Square wave voltammetry | 10 pfu/cell Herpes simplex virus or 20 mM | ( 78) | |
| Cu2+ | Cation exchanger, Dowex 50W-X8 (H+ form, 20–50 mesh) | DPASV | 0–110 ng/mL | 0.5 ng/m | ( 79) |
| Pb2+, Cd2+ | Hg thin film | DPASV | 20–100 ng/mL | Cd2+ 50 ng/l, Pb2+ 30 ng/l | ( 80) |
| Dopamine | GO/Fe3O4@SiO2 | DPV | 0.1–600.0 μM | 8.9×10−8 M | ( 81) |
| Ascorbic acid | Phenylenediamine | SWV | 0.45–409.10 μM | 0.11 μM | ( 82) |
| Analyte | Sensor feature | Printer | Ink | Substrate | Ref. |
|---|---|---|---|---|---|
| Dissolved oxygen | Fully ink get Range 0–8 mgL-1 Detection limit- 0.11 mgL-1 | Dimatix DMP (piezo) | Gold, Silver and dielectric (commercial) | PEN | ( 83) |
| H2O2 | Clark-type electrode Range 0–200 mgL-1 Detection limit- 0.05 mgL-1 | Dimatix DMP (piezo) | Graphene | Paper | ( 84) |
| Thyroid stimulating hormone (TSH) and atrazine (ATR) | Fully-inkjet Detection limit: ATR: 0.01 μg×L−1. TSH: 0.5μIU×L−1. | X-series Cera Printer (Piezo) | Carbon nanotubes, silver and dielectric (commercial) | PET | ( 85) |
| Glucose | Inkjet on printed circuit board. Range: 0−40 mg×dL−1. Detection Limit 0.3 mgdL−1 | Homemade printer (Piezo) | Graphene (Commercial) | Polyimide | ( 86) |
| Iron ion (Fe2+) and dopamine (DA) | Fully-inkjet. Range: 10–200 μM (Fe2+). 10 μM - 100 μM (DA). | HP deskjet (Thermal) | Dielectric commercial and carbon nanotube | Paper | ( 87) |
| Cholesterol | Screen printing+inkjet. Range: 0−15 mM. Sensitivity: 2.1 μA×mM−1 cm−2. Detection limit: 0.02 mM | Dimatix DMP (piezo) | Prussian blue nanoparticles | Polyester | ( 88) |
| Analyte | Sensor configuration | Modifier | Linear range | LOD | Ref. |
|---|---|---|---|---|---|
| Glucose, lactate, alcohol | The enzyme/Ti3C2Tx/PB/NLSG | Enzyme | ‘10 μM - 5.3 mM’, ‘0–20 mM’, ‘0–50 mM’ | ‘0.3 μM’, ‘0.5 μM’ - | ( 89) |
| AA, UA, DA | Pt/LSG | Electrodeposition | AA: 10–680 DA: 0.5–32.5 μM UA: 0.5–19.5 μM | AA:6.1 DA:0.07 UA:0.22 | ( 90) |
| Salmonella typhimurium | LIG-based sensor | Antibody | 25–105 CFU/mL | 13±7 CFU/ mL | ( 91) |
| Cd+2 and Pd+2 | PANI/N-LEG/GCE | Polymerization and laser scribing | Cd2+ 4.4×10−2−3.38 Pb+2: 2.4×10−3−1. | Cd2+: 9.6×10−3 Pb+2: 7.7×10−4 | ( 92) |
| Chloramphenicol | EBT MIP/Amine/ PEDOT/LIG | Electropolymerization | 10–3 μM –10000 μM | 6.2×10−4 μM | ( 93) |
| Nitrite | MWNT/AuNP/LSG | Drop casting | 10–90 μM | 6.75 μM | ( 94) |
Abbreviations: pfu- plaque forming units per volume; DPASV- Differential pulse anodic stripping voltammetry; DPV- Differential pulse voltammetry; GO/Fe3O4@SiO2-Graphene oxide/Iron oxide/ Silicon oxide; DMP- Dimatix material printer; HP- Hewlett-Packard; Ti3C2Tx/PB/NLSG- MXene/Prussian Blue/ nitrogen-doped laser-scribed graphene; Pt/LSG- Platinum/ Laser Scribed Graphene; PANI/N-LEG/GCE- Polyaniline Nitrogen doped Laser Engraved Graphene; LIG-Laser Induced Graphene; EBT MIP/Amine/ PEDOT/LIG-Eriochrome Black-T Molecularly Imprinted Polymer Poly(3,4-ethylenedioxythiophene)/ Laser Induced Graphene; MWNT/AuNP/LSG- Multi walled Carbon Nano Tubes/Gold nanoparticles/Laser Scribed Graphene; AA, UA, DA- Ascorbic acid, Uric acid, Dopamine; CFU- Colony Forming Unit.
4 Conclusion and Future Perspectives
Acknowledgments
Manjushree SG is grateful for the financial assistance provided by the Department of Science and Technology through the Women Scientists Scheme-A (WOS-A) SR/WOS-A/CS-153/2018. Prashanth SA expresses his gratitude to SVM Arts, Science and Commerce College, ILKAL for their unwavering support and encouragement as well as to Karnataka Science and Technology Academy (KSTA) for their financial assistance.
References
This chapter references 94 other publications.
- 1Killard A. J. Disposable sensors Current Opinion in Electrochemistry 2017 3 57 62Google Scholar1Disposable sensorsKillard, Anthony J.Current Opinion in Electrochemistry (2017), 3 (1), 57-62CODEN: COEUCY; ISSN:2451-9111. (Elsevier B.V.)A review. Device disposability is a crit. feature of electrochem. sensor prodn., allowing complex measurements to be carried out in a low cost and efficient manner. Disposable sensors are increasing rapidly in their level of sophistication, supported by developments in materials and fabrication processing. This is, in turn, leading to greater levels of device integration and functionality and has the potential to significantly expand the no. of people who can use them, where they can be deployed and how they can be used. As well as becoming more complex, there is also significant emphasis in making such disposable devices more environmentally sustainable. Current themes in the development of disposable sensors are explored here.
- 2Beebe D. J. Denton D. D. Radwin R. G. Webster J. G. A silicon-based tactile sensor for finger-mounted applications IEEE Transactions on Biomedical Engineering 1998 45 151 159Google Scholar2A silicon-based tactile sensor for finger-mounted applicationsBeebe D J; Denton D D; Radwin R G; Webster J GIEEE transactions on bio-medical engineering (1998), 45 (2), 151-9 ISSN:0018-9294.This paper presents a silicon-based force sensor packaged in a flexible package and describes the sensors performance on human subjects. The sensing element consists of a circular silicon diaphragm (200-micron thick with a 2-mm radius) over a 10-micron sealed cavity with a solid Torlon dome providing force-to-pressure transduction to the diaphragm. Two dome heights (0.5 and 1.5 mm) were compared. The sensor with the taller dome showed improved sensitivity. Dynamic calibration and tracking experiments are performed with the sensor mounted on the dominant thumb of five human subjects. Both force and loading direction are statistically significant (P < 0.05). Subject variability accounted for 8.7% of the variance, while loading direction accounted for 1.9% of the variance. Average errors for the tracking experiment range from-2.8 to 1.0 N and are subject dependent. Three out of four subjects showed increasing negative error with increasing load.
- 3Han T. Nag A. Afsarimanesh N. Mukhopadhyay S. C. Kundu S. Xu Y. Laser-assisted printed flexible sensors: a review Sensors 2019 19 1462Google ScholarThere is no corresponding record for this reference.
- 4Jung H.-C. Moon J.-H. Baek D.-H. Lee J.-H. Choi Y.-Y. Hong J.-S. Lee S.-H. CNT/PDMS composite flexible dry electrodesfor long-term ECG monitoring IEEE Transactions on Biomedical Engineering 2012 59 1472 1479Google Scholar4CNT/PDMS composite flexible dry electrodes for long-term ECG monitoringJung Ha-Chul; Moon Jin-Hee; Baek Dong-Hyun; Lee Jae-Hee; Choi Yoon-Young; Hong Joung-Sook; Lee Sang-HoonIEEE transactions on bio-medical engineering (2012), 59 (5), 1472-9 ISSN:.We fabricated a carbon nanotube (CNT)/ polydimethylsiloxane (PDMS) composite-based dry ECG electrode that can be readily connected to conventional ECG devices, and showed its long-term wearable monitoring capability and robustness to motion and sweat. While the dispersion of CNTs in PDMS is challenging, we optimized the process to disperse untreated CNTs within PDMS by mechanical force only. The electrical and mechanical characteristics of the CNT/PDMS electrode were tested according to the concentration of CNTs and its thickness. The performances of ECG electrodes were evaluated by using 36 types of electrodes which were fabricated with different concentrations of CNTs, and with a differing diameter and thickness. The ECG signals were obtained by using electrodes of diverse sizes to observe the effects of motion and sweat, and the proposed electrode was shown to be robust to both factors. The CNT concentration and diameter of the electrodes were critical parameters in obtaining high-quality ECG signals. The electrode was shown to be biocompatible from the cytotoxicity test. A seven-day continuous wearability test showed that the quality of the ECG signal did not degrade over time, and skin reactions such as itching or erythema were not observed. This electrode could be used for the long-term measurement of other electrical biosignals for ubiquitous health monitoring including EMG, EEG, and ERG.
- 5El-Saftawy A. Elfalaky A. Ragheb M. Zakhary S. Electron beam induced surface modifications of PET film Radiation Physics and Chemistry 2014 102 96 102Google Scholar5Electron beam induced surface modifications of PET filmEl-Saftawy, A. A.; Elfalaky, A.; Ragheb, M. S.; Zakhary, S. G.Radiation Physics and Chemistry (2014), 102 (), 96-102CODEN: RPCHDM; ISSN:0969-806X. (Elsevier B. V.)The semi-cryst. poly (ethylene terephthalate) PET film was treated by low energy electrons with different fluences and the surface wettability modification is investigated. Contact angle technique is employed to prove PET surface wettability enhancement. The contact angle decreases due to alteration of surface chem., morphol. and free energy after electrons irradn. Fourier transform IR spectroscopy (FT-IR) shows the creation of polar groups on the surface which are responsible for surface wettability improvement. Atomic force microscope (AFM) observations reveal that the surface roughness changes after electrons-PET interactions. Surface free energy anal. has been obsd. as well. Low energy electrons irradn. of PET film improves and controls its wettability, adhesion and then biocompatibility.
- 6Yoo K.-P. Lim L.-T. Min N.-K. Lee M. J. Lee C. J. Park C.-W. Novel resistive-type humidity sensor based on multiwall carbon nanotube/polyimide composite films Sensors and Actuators B: Chemical 2010 145 120 125Google Scholar6Novel resistive-type humidity sensor based on multiwall carbon nanotube/polyimide composite filmsYoo, Kum-Pyo; Lim, Lee-Taek; Min, Nam-Ki; Lee, Myung Jin; Lee, Chul Jin; Park, Chan-WonSensors and Actuators, B: Chemical (2010), 145 (1), 120-125CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)A novel resistive-type relative humidity (RH) sensor based on plasma-treated multiwall carbon nanotube/polyimide (p-MWCNT/PI) composite films is reported. Details of the fabrication process and sensing characteristics such as linearity and sensitivity are described. The elec. resistance of the p-MWCNT/PI composite film shows typical concn. percolation behavior with increasing p-MWCNT loading. At loadings beyond the percolation threshold (0.05 wt%), this sensor exhibits very good linearity with a pos. slope over the entire operational RH range. The sensor has a humidity sensitivity of about 0.0047/% RH and a linearity correlation (R2) of 0.9999. A humidity sensing mechanism for the p-MWCNT/PI composite film is proposed on the basis of charge transfer between adsorbed water mols. and the p-MWCNTs.
- 7Karmakar R. S. Lu Y.-J. Fu Y. Wei K.-C. Chan S.-H. Wu M.-C. Lee J.-W. Lin T.-K. Wang J.-C. Cross-talk immunity of PEDOT: PSS pressure sensing arrays with gold nanoparticle incorporation Scientific reports 2017 7 1 10Google Scholar7Bisphenol A Affects on the Functional Properties and Proteome of Testicular Germ Cells and Spermatogonial Stem Cells in vitro Culture ModelKarmakar, Polash Chandra; Kang, Hyun-Gu; Kim, Yong-Hee; Jung, Sang-Eun; Rahman, Md. Saidur; Lee, Hee-Seok; Kim, Young-Hyun; Pang, Myung-Geol; Ryu, Buom-YongScientific Reports (2017), 7 (1), 1-14CODEN: SRCEC3; ISSN:2045-2322. (Nature Research)The endocrine disruptor bisphenol A (BPA) is well known for its adverse effect on male fertility. Growing evidence suggests that BPA may interact with testicular germ cells and cause infertility as a result of its estrogenic activity. Objective of current in vitro study was to investigate the proliferation, survivability and stemness properties of mouse testicular germ cells exposed to BPA, and to evaluate possible expression of cellular proteome. Our results showed that germ cell viability and proliferation were not affected by low concns. (0.01, 0.1, 1, and 10μM) although significant redn. obsd. at 100μM BPA. Germ cell self-renewal and differentiation related marker proteins expression found unchanged at those concns. When BPA-exposed germ cells were transplanted into recipient testes, we obsd. fewer colonies at higher concns. (10 and 100μM). Addnl., a significant frequency of recombination failure during meiosis was obsd. in 10μM BPA-exposed germ cell transplanted recipient. Moreover, expt. on continuous BPA-exposed and 100μM BPA-recovered germ cells suggested that spermatogonial stem cells are more potential to survive in adverse environment. Finally, scrutinizing differentially expressed cellular proteins resulted from our proteomic anal., we conclude that BPA exposure might be assocd. with several health risks and infertility.
- 8Agarwal P. B. Alam B. Sharma D. S. Sharma S. Mandal S. Agarwal A. Flexible NO2 gas sensor based on single-walled carbon nanotubes on polytetrafluoroethylene substrates Flexible and Printed Electronics 2018 3 035001Google Scholar8Flexible NO2 gas sensor based on single-walled carbon nanotubes on polytetrafluoroethylene substratesAgarwal, Pankaj B.; Alam, Bayazeed; Sharma, Daya Shankar; Sharma, Sumit; Mandal, Soumen; Agarwal, AjayFlexible and Printed Electronics (2018), 3 (3), 035001/1-035001/8CODEN: FPELAB; ISSN:2058-8585. (IOP Publishing Ltd.)This paper presents a flexible and reliable chemiresistor-type NO2 gas sensor based on single-walled carbon nanotubes (SWNTs) on polytetrafluoroethylene (PTFE) membrane filter substrates. The sensor is realized by using a cost-effective spray coating in the prepn. of SWNTs thin film, followed by the fabrication of metal contacts using a shadow mask and polyethyleneimine (PEI) noncovalent functionalization of the SWNTs. This showed a high sensitivity to NO2 gas at room temp. in dry air; 21.58% to 167.7% for concns. of 0.75 ppm to 5 ppm, and was almost nonsensitive to ammonia. Gas sensing characterization results, obtained for various substrate bending/wrapping over different cylinders with diams. of 75 mm, 12.5 mm, and 6 mm showed that bending does not significantly affect sensitivity for NO2 concns. of 0.75 ppm to 2 ppm, while in the case of 3 ppm to 5 ppm NO2, the bent samples indicate enhanced sensitivity. This is probably because of the porous nature of PTFE substrates; these sensors were 1.5 to 2.7 times more sensitive than those fabricated over silicon substrate for 1 ppm and 5 ppm, resp. Moreover, the relative humidity of 10% and 30% significantly reduced the sensitivity of the sensors. The presented results could be useful for the future development of flexible electronics/sensors for monitoring outdoor air quality and for the detection of volatile org. compds.
- 9Rahimi R. Ochoa M. Ziaie B. Direct laser writing of porous-carbon/silver nanocomposite for flexible electronics ACS applied materials & interfaces 2016 8 16907 16913Google Scholar9Direct Laser Writing of Porous-Carbon/Silver Nanocomposite for Flexible ElectronicsRahimi, Rahim; Ochoa, Manuel; Ziaie, BabakACS Applied Materials & Interfaces (2016), 8 (26), 16907-16913CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)In this research article, we demonstrate a facile method for the fabrication of porous-carbon/silver nanocomposites using direct laser writing on polymeric substrates. Our technique uses a combination of CO2 laser-induced carbonization and selective silver deposition on a polyimide sheet to create flexible highly conductive traces. The localized laser irradn. selectively converts the polyimide to a highly porous and conductive carbonized film with superhydrophilic wettability. The resulting pattern allows for selective trapping of aq. silver ionic ink solns. into the carbonized regions, which are converted to silver nanoparticle fillers upon an annealing step. Elemental and surface morphol. anal. via XRD and SEM reveals a uniform coating of Ag nanoparticles on the porous carbon. The Ag/C composite lowers the sheet resistance of the original laser carbonized polyimide from 50 to 0.02 Ω/.box.. The resulting patterns are flexible and electromechanically robust with less than 0.6 Ω variation in resistance after >15000 bending flexion cycles at a radius of curvature of 5 mm. Furthermore, using this technique, we demonstrate the fabrication of a wireless resonant pressure sensor capable of detecting pressures ranging from 0 to 97 kPa with an av. sensitivity of -26 kHz/kPa.
- 10Tasaltin C. Basarir F. Preparation of flexible VOC sensor based on carbon nanotubes and gold nanoparticles Sensors and Actuators B: Chemical 2014 194 173 179Google Scholar10Preparation of flexible VOC sensor based on carbon nanotubes and gold nanoparticlesTasaltin, Cihat; Basarir, FevzihanSensors and Actuators, B: Chemical (2014), 194 (), 173-179CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Novel flexible volatile org. compd. (VOC) sensor was prepd. by deposition of gold nanoparticle (AuNP) and amine-modified multi-walled carbon nanotubes (MWCNT-NH2) on polyimide substrate via electrospraying technique. First, interdigitated electrodes (IDE) were fabricated on polyimide (Kapton) substrate by photolithog. Then, the substrates were subjected to oxygen plasma etching, followed by electrospraying of AuNP and MWCNT-NH2 soln. on the substrate. The thin films were characterized by FE-SEM and AFM as well as elec. cond. measurement. Chem. sensing behaviors of the sensors were analyzed against polar (water, propanol and ethanol) and nonpolar (hexane, toluene, trichloroethylene and chloroform) VOCs. In addn., effect of the AuNP/MWCNT-NH2 ratio on the cond. and sensing was investigated. Bending test (100 times) resulted in negligible resistance change, demonstrating the successful prepn. of flexible sensor.
- 11Mayousse C. Celle C. Carella A. Simonato J.-P. Synthesis and purification of long copper nanowires. Application to high performance flexible transparent electrodes with and without PEDOT: PSS Nano Research 2014 7 315 324Google Scholar11Synthesis and purification of long copper nanowires. Application to high performance flexible transparent electrodes with and without PEDOT:PSSMayousse, Celine; Celle, Caroline; Carella, Alexandre; Simonato, Jean-PierreNano Research (2014), 7 (3), 315-324CODEN: NRAEB5; ISSN:1998-0000. (Springer GmbH)We demonstrate the hydrothermal synthesis of long copper nanowires based on a simple protocol. We show that the purifn. of the nanowires is very important and can be achieved easily by wet treatment with glacial acetic acid. Fabrication of random networks of purified copper nanowires leads to flexible transparent electrodes with excellent optoelectronic performances (e.g., 55 Ω/sq. at 94% transparency). The process is carried out at room temp. and no post-treatment is necessary. Hybrid materials with the conductive polymer PEDOT:PSS show similar properties (e.g., 46 Ω/sq. at 93% transparency), with improved mech. properties. Both electrodes were integrated in capacitive touch sensors.
- 12Jin H. Zhou J. Dong S. Feng B. Luo J. Wang D. Milne W. Yang C. Y. Deposition of c-axis orientation aluminum nitride films on flexible polymer substrates by reactive direct-current magnetron sputtering Thin Solid Films 2012 520 4863 4870Google Scholar12Deposition of c-axis orientation aluminum nitride films on flexible polymer substrates by reactive direct-current magnetron sputteringJin, H.; Zhou, J.; Dong, S. R.; Feng, B.; Luo, J. K.; Wang, D. M.; Milne, W. I.; Yang, C. Y.Thin Solid Films (2012), 520 (15), 4863-4870CODEN: THSFAP; ISSN:0040-6090. (Elsevier B.V.)Aluminum nitride (AlN) piezoelec. thin films with c-axis crystal orientation on polymer substrates can potentially be used for development of flexible electronics and lab-on-chip systems. In this study, we investigated the effects of deposition parameters on the crystal structure of AlN thin films on polymer substrates deposited by reactive direct-current magnetron sputtering. The results show that low sputtering pressure as well as optimized N2/Ar flow ratio and sputtering power is beneficial for AlN (002) orientation and can produce a highly (002) oriented columnar structure on polymer substrates. High sputtering power and low N2/Ar flow ratio increase the deposition rate. In addn., the thickness of Al underlayer also has a strong influence on the film crystallog. The optimal deposition parameters in our expts. are: deposition pressure 0.38 Pa, N2/Ar flow ratio 2:3, sputtering power 414 W, and thickness of Al underlayer less than 100 nm.
- 13Nag A. Mitra A. Mukhopadhyay S. C. Graphene and its sensor-based applications: A review Sensors and Actuators A: Physical 2018 270 177 194Google Scholar13Graphene and its sensor-based applications: A reviewNag, Anindya; Mitra, Arkadeep; Mukhopadhyay, Subhas ChandraSensors and Actuators, A: Physical (2018), 270 (), 177-194CODEN: SAAPEB; ISSN:0924-4247. (Elsevier B.V.)This paper presents an overview of the work done on graphene in recent years. It explains the prepn. techniques, the properties of graphene related to its physio-chem. structure and some key applications. Graphene, due to its outstanding elec., mech. and thermal properties, has been one of the most popular choices to develop the electrodes of a sensor. It has been used in different forms including nanoparticle and oxide forms. Along with the prepn. and properties of graphene, the categorization of the applications has been done based on the type of sensors. Comparisons between different research studies for each type have been made to highlight their performances. The challenges faced by the current graphene-based sensors along with some of the probable solns. and their future opportunities are also briefly explained in this paper.
- 14Manjunatha J. Swamy B. K. Deraman M. Mamatha G. Simultaneous voltammetric measurement of ascorbic acid and dopamine at poly (vanillin) modified carbon paste electrode: A cyclic voltammetric study Pharma Chem 2012 4 2489 2497Google Scholar14Simultaneous voltammetric measurement of ascorbic acid and dopamine at poly(vanillin) modified carbon paste electrode: a cyclic voltammetric studyManjunatha, J. G.; Swamy, B. E. Kumara; Deraman, M.; Mamatha, G. P.Pharma Chemica (2012), 4 (6), 2489-2497CODEN: PCHHAX; ISSN:0975-413X. (Scholars Research Library)Polymd. film of vanillin was prepd. on to the surface of carbon paste electrode in alk. soln. by cyclic voltammetric (CV) technique after pretreatment by H2SO4 media. The polyvanillin (VA) film-coated carbon paste electrode (CPE) exhibited excellent electrocatalytic activity toward the oxidn. of dopamine (DA) and ascorbic acid (AA) in 0.2M acetate buffer soln. at pH 7. This modified electrode exhibited a potent and persistent electron-mediating behavior followed by well-sepd. oxidn. peaks towards DA and AA. The modified electrode shows good sensitivity, selectivity and stability and was applied to the detn. of DA and AA.
- 15Manjunatha J. G. Surfactant modified carbon nanotube paste electrode for the sensitive determination of mitoxantrone anticancer drug Journal of Electrochemical Science and Engineering 2017 7 39 49Google Scholar15Surfactant modified carbon nanotube paste electrode for the sensitive determination of mitoxantrone anticancer drugManjunatha, Jamballi G.Journal of Electrochemical Science and Engineering (2017), 7 (1), 39-49CODEN: JESEGX; ISSN:1847-9286. (International Association of Physical Chemists)Surfactant modified carbon nanotube paste electrode is prepd. as electrochem. sensor with high sensitivity responding to mitoxantrone (MTX). Electrochem. oxidn. of MTX is investigated in buffered soln. by cyclic voltammetry that is found very sensi-tive method for detection of MTX. It is shown that the sodium dodecyl sulfate modified carbon nanotube paste electrode (SDSMCNTPE) gives enhanced current response for MTX compared to the bare carbon nanotube paste electrode (BCNTPE). Different parameters were tested to optimize the conditions for MTX detn. The effects of different surfactant and surfactant concn., pH, scan rate, and concn. of MTX on the oxidn. peak current values were detd. Excellent results were obtained by cyclic voltammetry using SDSMCNTPE, where two MTX oxidn. peaks appeared around 370 and 600 mV vs. Detailed anal. of the second voltammetric peak showed the linear dependence on concn. between 2×10-7 and 7×10-6 M MTX with the slope of the corelation coeff. of 0.99271. LOD and LOQ were detd. as 3.5 ×10-8 M and 11×10-8 M, resp. The SDSMCNTPE showed very good reproducibility, high stability in its voltammetric response, high electrochem. sensitivity and low detection limit for MTX.
- 16Hareesha N. Manjunatha J. A simple and low-cost poly (dl-phenylalanine) modified carbon sensor for the improved electrochemical analysis of Riboflavin Journal of Science: Advanced Materials and Devices 2020 5 502 511Google Scholar16A simple and low-cost poly (DL-phenylalanine) modified carbon sensor for the improved electrochemical analysis of RiboflavinHareesha, N.; Manjunatha, J. G.Journal of Science: Advanced Materials and Devices (2020), 5 (4), 502-511CODEN: JSAMCR; ISSN:2468-2179. (Elsevier B.V.)The current study reports on the electroanal. of Riboflavin (RF) on the surface of a poly DL-phenylalanine modified carbon paste sensor (PDLPAMCPS). The developed sensor caused a noticeable electrocatalytic action for the redox activity of RF in a phosphate buffer soln. (PBS) of pH 6.5. This anal. was evaluated using differential pulse voltammetry (DPV) and cyclic voltammetry (CV) techniques. The active surface of the functioning sensor exposes well developed anodic peak with prominent electron transfer. PDLPAMCPS shows distinctive and improved DPV and CV peaks for RF with and without the presence of org. dyes and dopamine (DA). The impact of the soln. pH, scan rate, accumulation potential and concn. on RF at PDLPAMCPS were studied. A vibrant linear growth was obsd. between the oxidn. peak current and the RF concn. in the range of 6.0μM-30.0μM, which provides a lower limit of detection (LOD) of 92 nM with excellent sensitivity. The proposed method and sensor were applied for the assessment of RF in medicine sample as an anal. application with fine recoveries and low LOD.
- 17Hareesha N. Manjunatha J. Raril C. Tigari G. Sensitive and selective electrochemical resolution of tyrosine with ascorbic acid through the development of electropolymerized alizarin sodium sulfonate modified carbon nanotube paste electrodes ChemistrySelect 2019 4 4559 4567Google Scholar17Sensitive and Selective Electrochemical Resolution of Tyrosine with Ascorbic Acid through the Development of Electropolymerized Alizarin Sodium Sulfonate Modified Carbon Nanotube Paste ElectrodesHareesha, N.; Manjunatha, J. G.; Raril, C.; Tigari, GirishChemistrySelect (2019), 4 (15), 4559-4567CODEN: CHEMUD; ISSN:2365-6549. (Wiley-VCH Verlag GmbH & Co. KGaA)An approachable and a selective electrocatalytic investigation of tyrosine (TR) with ascorbic acid (AA) in phosphate buffer soln. (PBS) was done using a sensitive Poly Alizarin sodium sulfonate modified Carbon nanotube paste electrode (PASSMCNTPE). The morphol. and elemental studies on PASSMCNTPE and bare carbon nanotube paste electrode (BCNTPE) were resolute through field emission SEM (FESEM) and energy dispersive X-ray spectroscopy (EDX). The PASSMCNTPE shows excellent sensitivity, selectivity, stability, repeatability, and reproducibility for the electrochem. characterization of TR through cyclic voltammetry (CV) and differential pulse voltammetry (DPV). The prepd. sensor provides an elevated current response towards TR as compared to the BCNTPE. A plot of the anodic peak current of TR and the concn. of TR was obtained linearly in the range 1.4×10-4 M to 2×10-4 M with the limit of detection (LOD) 7.8×10-7 M and the limit of quantification (LOQ) 26×10-7 M resp. The equipped sensor was also helpful for the quant. and qual. anal. of TR moiety in a pharmaceutical drug as a real sample.
- 18Hareesha N. Manjunatha J. G. Surfactant and polymer layered carbon composite electrochemical sensor for the analysis of estriol with ciprofloxacin Materials Research Innovations 2020 24 349 362Google Scholar18Surfactant and polymer layered carbon composite electrochemical senor for the analysis of estriol with ciprofloxacinHareesha, Nagarajappa; Manjunatha, Jamballi G.Materials Research Innovations (2020), 24 (6), 349-362CODEN: MRINFV; ISSN:1433-075X. (Taylor & Francis Ltd.)This article exposes an approachable strategy for the responsive electrochem. anal. of estriol (ER) hormone by designing a sensitive and selective sensor of sodium dodecyl sulfate and polymerised xanthacridinum modified carbon nanotube and graphite composite paste electrode (SDS-PXAMCNTGCPE) via linear sweep voltammetry (LSV), cyclic voltammetry (CV) and differential pulse voltammetry (DPV) techniques. ER exposed a fine resolved irreversibly oxidised peak in phosphate buffer soln. (PBS) of pH 7.0. CV and DPV reveal a commendable linear relation between oxidative peak current of ER and concn. of ER in the range 2 UM to 200 UM and 10 UM to 70 UM and they provide a promising lowest limit of detection (LOD) values of 2.9 × 10-7 M and 1.9 × 10-7 M. CV and DPV techniques were also applied to examine the selectivity of ER with Ciprofloxacin (CF). The adapting capability of SDS-PXAMCNTGCPE was accountable for the lower level identification of ER in sewage water as a real sample. The proposed sensor needs an easy prepn. method with low cost and it exhibits superior sensitivity, stability, repeatability, and reproducibility.
- 19Manjunatha J. A surfactant enhanced graphene paste electrode as an effective electrochemical sensor for the sensitive and simultaneous determination of catechol and resorcinol Chemical Data Collections 2020 25 100331Google Scholar19A surfactant enhanced graphene paste electrode as an effective electrochemical sensor for the sensitive and simultaneous determination of catechol and resorcinolManjunatha, J. G.Chemical Data Collections (2020), 25 (), 100331CODEN: CDCHA8; ISSN:2405-8300. (Elsevier B.V.)A highly sensitive and selective electrochem. sensor based on surfactant modified graphene paste electrode was designed for the detn. of catechol (CC) and resorcinol (RS). The developed electrodes were characterized by cyclic voltammetry (CV), differential pulse voltammetry (DPV), and Field emission SEM (FE-SEM). Under the ideal conditions, CV and DPV studies revealed that, compared with bare graphene paste electrode (BGPE), sodium dodecyl sulfate modified graphene paste electrode (SDSMGPE) demonstrated an increase in the efficiency of the electrocatalytic oxidn. of CC. Under the optimized condition the peak current for CC showed a good linear relationship with concn. in the range 2.0 x 10-6 to 1.0 x 10-5 and 1.5 x 10-5 to 1.5 x 10-4 M, and the detection limit was 106 nM (S/N = 3). The CV results show that CC and RS could be detected simultaneously using SDSMGPE with peak-to-peak sepn. of 0.728 V for CC-RS. The sensor is very promising for CC sensing attributing to its high sensitivity, fast response, excellent stability, and good reproducibility. The SDSMGPE sensor utilized for detection of CC in real sample with a satisfactory result.
- 20Manjunatha J. G. Poly (Adenine) modified graphene-based voltammetric sensor for the electrochemical determination of catechol, hydroquinone and resorcinol The Open Chemical Engineering Journal 2020 14Google ScholarThere is no corresponding record for this reference.
- 21Prinith N. S. Manjunatha J. G. Polymethionine modified carbon nanotube sensor for sensitive and selective determination of L-tryptophan Journal of Electrochemical Science and Engineering 2020 10 305 315Google Scholar21Polymethionine modified carbon nanotube sensor for sensitive and selective determination of L-tryptophanPrinith, Nambudumada S.; Manjunatha, Jamballi G.Journal of Electrochemical Science and Engineering (2020), 10 (4), 305-315CODEN: JESEGX; ISSN:1847-9286. (International Association of Physical Chemists)The electrochem. initiated catalytic oxidn. of amino acid L-tryptophan (L-TPN) in phosphate buffer soln. has been scrutinized using highly conductive polymethionine modified carbon nanotube paste sensor (PMETCNTPS) through cyclic voltammetry (CV) technique. Compared to the bare carbon nanotube paste sensor (BCNTPS), PMETCNTPS exhibited a quantifiable current signal by CV method. PMETCNTPS was found sensitive to LTPN concns. within the linear segment of detection range 1.5 - 8.0x10-5 M. By employing the calibration plot, the detection limit was detd. as 6.99x10-7 M. In addn., PMETCNTPS was successfully exploited and validated in detg. L-TPN in the pharmaceutical supplement.
- 22Hareesha N. Manjunatha J. Amrutha B. Pushpanjali P. Charithra M. Prinith Subbaiah N. Electrochemical analysis of indigo carmine in food and water samples using a poly (glutamic acid) layered multi-walled carbon nanotube paste electrode Journal of Electronic Materials 2021 50 1230 1238Google Scholar22Electrochemical Analysis of Indigo Carmine in Food and Water Samples Using a Poly(Glutamic Acid) Layered Multi-walled Carbon Nanotube Paste ElectrodeHareesha, N.; Manjunatha, J. G.; Amrutha, B. M.; Pushpanjali, P. A.; Charithra, M. M.; Prinith Subbaiah, N.Journal of Electronic Materials (2021), 50 (3), 1230-1238CODEN: JECMA5; ISSN:0361-5235. (Springer)Some colorants are hazardous to living species; hence, a powerful and fast methodol. is required for the anal. of those colorants in food and water samples. A modest electrochem. polymerised glutamic acid layered multi-walled carbon nanotube paste electrode [P(GA)LMWCNTPE] was functionalised for the sensing of indigo carmine (IC) by powerful differential pulse voltammetry (DPV) and cyclic voltammetry (CV) approaches. Within the optimized exptl. conditions, the P(GA)LMWCNTPE holds an acceptable and high rate of electro-catalytic activity towards the redox behavior of IC. The projected P(GA)LMWCNTPE shows a decent selectivity for IC in the presence of methyl orange. The modified sensor shows an acceptable linear growth between oxidative peak current and concn. in both CV and DPV methods with fine limit of detection values of 4.2μM and 0.36μM, resp. Addnl., the developed sensor was effectively applied to detect IC in food and water samples. The morphol. and surface activities of the modified and unmodified electrodes were detd. through field emission SEM, electrochem. impedance spectroscopy, and CV techniques. The P(GA)LMWCNTPE requires a simple prepn. procedure and is low-cost, with acceptable storage stability, sensitivity, and reproducibility.
- 23Monnappa B. Manjunatha J. G. Bhatt A. S. Chenthattil R. Ananda P. Electrochemical sensor for the determination of alizarin red-s at non-ionic surfactant modified carbon nanotube paste electrode Physical Chemistry Research 2019 7 523 533Google ScholarThere is no corresponding record for this reference.
- 24Raril C. Manjunatha J. G. Sensitive electrochemical analysis of resorcinol using polymer modified carbon paste electrode: a cyclic voltammetric study Anal. Bioanal. Electrochem 2018 10 488 498Google Scholar24Sensitive electrochemical analysis of resorcinol using polymer modified carbon paste electrode: a cyclic voltammetric studyRaril, Chenthattil; Manjunatha, Jamballi G.Analytical & Bioanalytical Electrochemistry (2018), 10 (4), 488-498CODEN: ABENC2; ISSN:2008-4226. (University of Tehran, Center of Excellence in Electrochemistry)Glycine was electropolymd. on a carbon paste electrode (CPE) to form the polymer film in the electrooxidn. process of the amino and carboxylic group contg. compd. by cyclic voltammetric technique and the electrochem. oxidn. of resorcinol (RC) was studied in 0.2 M phosphate buffer soln. (PBS) having pH 7.0. Modified carbon paste electrode (MCPE) shows a good electrocatalytic activity towards RC compared to bare carbon paste electrode (BCPE). The oxidn. of RC at MCPE showed a linear relation vs. concn. of RC in the range of 6×10-5 to 1×10-3 M with a detection limit 8.6×10-6 M.
- 25Charithra M. M. Manjunatha J. G. G. Raril C. Surfactant modified graphite paste electrode as an electrochemical sensor for the enhanced voltammetric detection of estriol with dopamine and uric acid Advanced Pharmaceutical Bulletin 2020 10 247Google Scholar25Surfactant modified graphite paste electrode as an electrochemical sensor for the enhanced voltammetric detection of estriol with dopamine and uric acidCharithra, Madikeri Manjunath; Manjunatha, Jamballi Gangadharappa Gowda; Raril, ChenthattilAdvanced Pharmaceutical Bulletin (2020), 10 (2), 247-253CODEN: APBDCL; ISSN:2251-7308. (Tabriz University of Medical Sciences)Purpose: Estriol (ERL) is a type of hormone among the groups of estrogen hormone that was detected through the voltammetric technique by constructing an electrochem. sensor based on the octoxynol-9 modified graphite paste electrode (OXL-9MGPE). Methods: Using the strategy of cyclic voltammetry (CV) and differential pulse voltammetry (DPV) with a bare graphite paste electrode (BGPE) immobilized with OXL-9, ERL electro-oxidn. has been assessed in 0.2 M phosphate buffer soln. (PBS) of pH 6.0. The fabricated electrode has substantial electrochem. sensing efficiency, and the ERL oxidn. at the OXL-9MGPE was the irreversible process. The surface morphol. characteristics of BGPE and OXL-9MGPE were differentiated with the help of field emission SEM (FE-SEM). Results: The impact of various factors such as scan rate, pH, reproducibility, repeatability, and stability on the electro-oxidn. of ERL was evaluated. Techniques of CV and DPV were utilized to det. ERL, dopamine (DAN), and uric acid (URA) simultaneously with the projected sensor. The peak current was varied with ERL concn. in the range from 4x10-5 to 1.2x10-4 M at OXL-9MGPE. From this, the detection limit 1.4x10-6 M and limit of quantification (LOQ) 4.7x10-6 M have been attained. Conclusion: As a result, OXL-9MGPE was successfully achieved as an electrochem. detector for the electro anal. of ERL via the CV technique.
- 26Manjunatha J. G. Electrochemical polymerised graphene paste electrode and application to catechol sensing The Open Chemical Engineering Journal 2019 13Google ScholarThere is no corresponding record for this reference.
- 27Prinith N. S. Manjunatha J. G. Raril C. Electrocatalytic analysis of dopamine, uric acid and ascorbic acid at poly (adenine) modified carbon nanotube paste electrode: a cyclic voltammetric study Anal. Bioanal., Electrochem 2019 11 742 756Google Scholar27Electrocatalytic analysis of dopamine, uric acid and ascorbic acid at poly(adenine) modified carbon nanotube paste electrode: a cyclic voltammetric studyPrinith, Nambudumada S.; Manjunatha, Jamballi G.; Raril, ChenthattilAnalytical & Bioanalytical Electrochemistry (2019), 11 (6), 742-756CODEN: ABENC2; ISSN:2008-4226. (University of Tehran, Center of Excellence in Electrochemistry)In this work, Poly (adenine) film modified carbon nanotube paste electrode (PAEMCNTPE), prepd. by electropolymn. of adenine on a carbon nanotube paste electrode, applied for the sensitive and selective anal. of dopamine (DA), Uric acid (UA) and Ascorbic acid (AA) based on cyclic voltammetric technique. The polyadenine modified carbon nanotube paste electrode has presented with an impressive electrocatalytic activity towards oxidn. of DA, UA, and AA in the presence of supporting electrolyte, 0.2 M PBS pH 6.5. The surface of fabricated sensor displayed with a permselective layer with transducing, anti- interfering and anti-fouling properties followed by solving the overlapped peaks of DA, AA and UA obtained from the conventional electrode by presenting with distinct three anodic peaks. The result of this electrochem. sensor displayed with excellent sensitivity, selectivity, reproducibility, repeatability and highly stable. The detection limit for DA was 6.7×10-7 M. Further; this fabricated electrode was carried out for the quantification of DA in the real sample.
- 28Manjunatha J. G. Swamy B. E. K. Mamatha G. P. Gilbert O. Sherigara B. S. Poly (maleic acid) modified carbon paste electrode for simultaneous detection of dopamine in the presence of uric acid: a cyclic voltammetric study ANALYTICAL & BIOANALYTICAL ELECTROCHEMISTRY 2011 3 146 159Google ScholarThere is no corresponding record for this reference.
- 29Liu C. Huang N. Xu F. Tong J. Chen Z. Gui X. Fu Y. Lao C. 3D printing technologies for flexible tactile sensors toward wearable electronics and electronic skin Polymers 2018 10 629Google Scholar293D printing technologies for flexible tactile sensors toward wearable electronics and electronic skinLiu, Changyong; Huang, Ninggui; Xu, Feng; Tong, Junda; Chen, Zhangwei; Gui, Xuchun; Fu, Yuelong; Lao, ChangshiPolymers (Basel, Switzerland) (2018), 10 (6), 629/1-629/31CODEN: POLYCK; ISSN:2073-4360. (MDPI AG)3D printing has attracted a lot of attention in recent years. Over the past three decades, various 3D printing technologies have been developed including photopolymn.-based, materials extrusion-based, sheet lamination-based, binder jetting-based, power bed fusion-based and direct energy deposition-based processes. 3D printing offers unparalleled flexibility and simplicity in the fabrication of highly complex 3D objects. Tactile sensors that emulate human tactile perceptions are used to translate mech. signals such as force, pressure, strain, shear, torsion, bend, vibration, etc. into elec. signals and play a crucial role toward the realization of wearable electronics and electronic skin. To date, many types of 3D printing technologies have been applied in the manufg. of various types of tactile sensors including piezoresistive, capacitive and piezoelec. sensors. This review attempts to summarize the current state-of-the-art 3D printing technologies and their applications in tactile sensors for wearable electronics and electronic skin. The applications are categorized into five aspects: 3D-printed molds for microstructuring substrate, electrodes and sensing element; 3D-printed flexible sensor substrate and sensor body for tactile sensors; 3D-printed sensing element; 3D-printed flexible and stretchable electrodes for tactile sensors; and fully 3D-printed tactile sensors. Latest advances in the fabrication of tactile sensors by 3D printing are reviewed and the advantages and limitations of various 3D printing technologies and printable materials are discussed. Finally, future development of 3D-printed tactile sensors is discussed.
- 30Kundu S. Hussain M. Kumar V. Kumar S. Das A. K. Direct metal laser sintering of TiN reinforced Ti6Al4V alloy based metal matrix composite: Fabrication and characterization The International Journal of Advanced Manufacturing Technology 2018 97 2635 2646Google ScholarThere is no corresponding record for this reference.
- 31Siegel A. C. Phillips S. T. Dickey M. D. Lu N. Suo Z. Whitesides G. M. Foldable printed circuit boards on paper substrates Advanced Functional Materials 2010 20 28 35Google Scholar31Foldable Printed Circuit Boards on Paper SubstratesSiegel, Adam C.; Phillips, Scott T.; Dickey, Michael D.; Lu, Nanshu; Suo, Zhigang; Whitesides, George M.Advanced Functional Materials (2010), 20 (1), 28-35CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)This paper describes several low-cost methods for fabricating flexible electronic circuits on paper. The circuits comprise (i) metallic wires (e.g., tin or zinc) that are deposited on the substrate by evapn., sputtering, or airbrushing, and (ii) discrete surface-mountable electronic components that are fastened with conductive adhesive directly to the wires. These electronic circuits-like conventional printed circuit boards-can be produced with electronic components that connect on both sides of the substrate. Unlike printed circuit boards made from fiberglass, ceramics, or polyimides, however, paper can be folded and creased (repeatedly), shaped to form three-dimensional structures, trimmed using scissors, used to wick fluids (e.g., for microfluidic applications) and disposed of by incineration. Paper-based electronic circuits are thin and lightwt.; they should be useful for applications in consumer electronics and packaging, for disposable systems for uses in the military and homeland security, for applications in medical sensing or low-cost portable diagnostics, for paper-based microelectromech. systems, and for applications involving textiles.
- 32Zittel H. Miller F. A Glassy-Carbon Electrode for Voltammetry Analytical Chemistry 1965 37 200 203Google Scholar32A glassy-carbon electrode for voltammetryZittel, H. E.; Miller, F. J.(1965), 37 (2), 200-3CODEN: ANCHAM; ISSN:0003-2700.A piece of glassy C, which is a gas-impermeable, elec.-conductive, chem.-resistant material, was cut from a small boat and sealed into an epoxy resin rod, having a central tube to permit Hg contact, and was used as indicator electrode in a system contg. a pyrolytic graphite counter electrode and a S.C.E. reference electrode. The app. was useful in voltammetric studies from + 1.2 to -0.8 v. vs. S.C.E. with acidic solns. of Ce+++, Ce4+, Cr6+, Fe++, Hg+, Fe(CN)64-, Ag+, Cu++, and UO2++, and results compared favorably with those obtained with other solid electrodes with respect to ease of fabrication and maintenance.
- 33Taylor R. Humffray A. Electrochemical studies on glassy carbon electrodes: I. Electron transfer kinetics Journal of electroanalytical chemistry and interfacial electrochemistry 1973 42 347 354Google Scholar33Electrochemical studies on glassy carbon electrodes. I. Electron transfer kineticsTaylor, R. J.; Humffray, A. A.Journal of Electroanalytical Chemistry and Interfacial Electrochemistry (1973), 42 (3), 347-54CODEN: JEIEBC; ISSN:0022-0728.Rate const. ks measurements for charge transfer processes at various electrodes show the general sequence ks(Pt) > ks (glassy C) > ks (was-impregnated graphite) > ks (C paste). Max. rates of glassy C electrodes were obsd. after treatment with chromic acid, except for the redn. of IO3- ion, where simple polishing of the electrode surface produces the highest rate. Time effects on rate consts. are also discussed.
- 34Uskoković V. A historical review of glassy carbon: Synthesis, structure, properties and applications Carbon Trends 2021 5 100116Google ScholarThere is no corresponding record for this reference.
- 35Švancara I. Vytřas K. Kalcher K. Walcarius A. Wang J. Carbon paste electrodes in facts, numbers, and notes: a review on the occasion of the 50‐years jubilee of carbon paste in electrochemistry and electroanalysis Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2009 21 7 28Google Scholar35Carbon paste electrodes in facts, numbers, and notes: a review on the occasion of the 50-years jubilee of carbon paste in electrochemistry and electroanalysisSvancara, Ivan; Vytras, Karel; Kalcher, Kurt; Walcarius, Alain; Wang, JosephElectroanalysis (2009), 21 (1), 7-28CODEN: ELANEU; ISSN:1040-0397. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. This article reviews the electrochem. and electroanal. applications of carbon paste-based electrodes, sensors, and detectors on the occasion of the half-of-century anniversary since the discovery of carbon paste. The review (with 333 refs.) has been prepd. in the form of a retrospective compilation presenting the field by means of various facts, notes, data, surveys, and summaries, including numerous rarities or curiosities that illustrate the individual achievements and milestones. Carbon paste-based electrodes are discussed in their entirety by covering all important areas of the field, starting from basic characterization of carbon paste as the electrode material, via its typical physicochem. and electrochem. properties or specific features, up to a representative documentation of their applicability in electrochem. and electroanal. measurements.
- 36P. S. Adarakatti, S. K. Kempahanumakkagari , Modified electrodes for sensing, in: Electrochemistry , 2018, pp. 58-95.Google ScholarThere is no corresponding record for this reference.
- 37Banks C. E. Moore R. R. Davies T. J. Compton R. G. Investigation of modified basal plane pyrolytic graphite electrodes: definitive evidence for the electrocatalytic properties of the ends of carbon nanotubes Chemical Communications 2004 1804 1805Google Scholar37Investigation of modified basal plane pyrolytic graphite electrodes: definitive evidence for the electrocatalytic properties of the ends of carbon nanotubesBanks, Craig E.; Moore, Ryan R.; Davies, Trevor J.; Compton, Richard G.Chemical Communications (Cambridge, United Kingdom) (2004), (16), 1804-1805CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)The basis of the electrocatalytic nature of multi-wall C nanotubes is suggested to reside in electron transfer from the ends of nanotubes, which structurally resemble the behavior of edge plane (as opposed to basal plane) graphite, and is demonstrated via the comparison of the electrochem. oxidn. of epinephrine and the electrochem. redn. of ferricyanide at nanotube-modified electrodes using different types of graphite electrodes and with C60-modified electrodes.
- 38Banks C. E. Compton R. G. Edge plane pyrolytic graphite electrodes in electroanalysis: an overview Analytical Sciences 2005 21 1263 1268Google Scholar38Edge plane pyrolytic graphite electrodes in electroanalysis: An overviewBanks, Craig E.; Compton, Richard G.Analytical Sciences (2005), 21 (11), 1263-1268CODEN: ANSCEN; ISSN:0910-6340. (Japan Society for Analytical Chemistry)A review. The recent development, behavior and scope of edge plane pyrolytic graphite electrodes in electroanal. are overviewed. Similarities to, and advantages, over multi-walled CNT modified electrodes are noted and the wide scope of applications, ranging through gas sensing, stripping voltammetry and biosensing, illustrated.
- 39Adarakatti P. S. Foster C. W. Banks C. E. NS A. K. Malingappa P. Calixarene bulk modified screen-printed electrodes (SPCCEs) as a one-shot disposable sensor for the simultaneous detection of lead (II), copper (II) and mercury (II) ions: Application to environmental samples Sensors and Actuators A: Physical 2017 267 517 525Google ScholarThere is no corresponding record for this reference.
- 40Saidur M. Aziz A. A. Basirun W. Recent advances in DNA-based electrochemical biosensors for heavy metal ion detection: A review Biosensors and Bioelectronics 2017 90 125 139Google Scholar40Recent advances in DNA-based electrochemical biosensors for heavy metal ion detection: A reviewSaidur, M. R.; Abdul Aziz, A. R.; Basirun, W. J.Biosensors & Bioelectronics (2017), 90 (), 125-139CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)The presence of heavy metal in food chains due to the rapid industrialization poses a serious threat on the environment. Therefore, detection and monitoring of heavy metals contamination are gaining more attention nowadays. However, the current anal. methods (based on spectroscopy) for the detection of heavy metal contamination are often very expensive, tedious and can only be handled by trained personnel. DNA biosensors, which are based on electrochem. transduction, is a sensitive but inexpensive method of detection. The principles, sensitivity, selectivity and challenges of electrochem. biosensors are discussed in this review. This review also highlights the major advances of DNA-based electrochem. biosensors for the detection of heavy metal ions such as Hg2+, Ag+, Cu2+ and Pb2+.
- 41Apilux A. Dungchai W. Siangproh W. Praphairaksit N. Henry C. S. Chailapakul O. Lab-on-paper with dual electrochemical/colorimetric detection for simultaneous determination of gold and iron Analytical chemistry 2010 82 1727 1732Google Scholar41Lab-on-Paper with Dual Electrochemical/Colorimetric Detection for Simultaneous Determination of Gold and IronApilux, Amara; Dungchai, Wijitar; Siangproh, Weena; Praphairaksit, Narong; Henry, Charles S.; Chailapakul, OrawonAnalytical Chemistry (Washington, DC, United States) (2010), 82 (5), 1727-1732CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)A novel lab-on-paper device combining electrochem. and colorimetric detection for the rapid screening of Au(III) in the presence of a common interference, Fe(III), in industrial waste solns. is presented here. With dil. aqua regia (0.1M HCl + 0.05M HNO3) as the supporting electrolyte, square wave voltammetry on paper provided a well-defined redn. peak for Au(III) at ∼287 ± 12 mV vs. Ag/AgCl. Under the optimized working conditions, the calibration curve showed good linearity in the concn. range of 1-200 ppm of Au(III) with a correlation coeff. of 0.997. The limit of detection (LOD) of the proposed method is 1 ppm. Interferences from various cations were also studied. Fe(III) is the only metal that affects the electrochem. detn. of Au(III) when present above a 2.5-fold excess concn. of that of the Au(III). To overcome this limitation, a colorimetric method was used to simultaneously detect Fe(III) as a screening tool. The procedure was then successfully applied to det. Au(III) in Au-refining waste solns. The results are in agreement with those obtained from inductively coupled plasma-at. emission spectrometry (ICP-AES).
- 42Li X. Tian J. Garnier G. Shen W. Fabrication of paper-based microfluidic sensors by printing Colloids and surfaces B: Biointerfaces 2010 76 564 570Google Scholar42Fabrication of paper-based microfluidic sensors by printingLi, Xu; Tian, Junfei; Garnier, Gil; Shen, WeiColloids and Surfaces, B: Biointerfaces (2010), 76 (2), 564-570CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)A novel method for the fabrication of paper-based microfluidic diagnostic devices is reported; it consists of selectively hydrophobizing paper using cellulose reactive hydrophobization agents. The hydrophilic-hydrophobic contrast of patterns so created has excellent ability to control capillary penetration of aq. liqs. in paper channels. Incorporating this idea with digital ink jet printing techniques, a new fabrication method of paper-based microfluidic devices is established. Ink jet printing can deliver biomols. and indicator reagents with precision into the microfluidic patterns to form bio-chem. sensing zones within the device. This method thus allows the complete sensor, i.e. channel patterns and the detecting chemistries, to be fabricated only by two printing steps. This fabrication method can be scaled up and adapted to use high speed, high vol. and low cost com. printing technol. Sensors can be fabricated for specific tests, or they can be made as general devices to perform on-demand quant. anal. tasks by incorporating the required detection chemistries for the required tasks.
- 43Lu Y. Shi W. Qin J. Lin B. Fabrication and characterization of paper-based microfluidics prepared in nitrocellulose membrane by wax printing Analytical chemistry 2010 82 329 335Google Scholar43Fabrication and Characterization of Paper-Based Microfluidics Prepared in Nitrocellulose Membrane By Wax PrintingLu, Yao; Shi, Weiwei; Qin, Jianhua; Lin, BingchengAnalytical Chemistry (Washington, DC, United States) (2010), 82 (1), 329-335CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)Paper-based microfluidics is a promising technol. to develop a simple, low-cost, portable, and disposable diagnostic platform for resource-limited settings. Here the authors report the fabrication of paper-based microfluidic devices in nitrocellulose membrane by wax printing for protein immobilization related applications. The fabrication process, which can be finished within 10 min, includes mainly printing and baking steps. Wax patterning will form hydrophobic regions in the membrane, which can be used to direct the flow path or sep. reaction zones. The fabrication parameters like printing mode and baking time were optimized, and performances of the wax-patterned nitrocellulose membrane such as printing resoln., protein immobilization, and sample purifn. capabilities were also characterized in this report. The authors believe the wax-patterned nitrocellulose membrane will enhance the capabilities of paper microfluidic devices and bring new applications in this field.
- 44Lu Y. Lin B. Qin J. Patterned paper as a low-cost, flexible substrate for rapid prototyping of PDMS microdevices via “liquid molding” Analytical chemistry 2011 83 1830 1835Google Scholar44Patterned Paper as a Low-Cost, Flexible Substrate for Rapid Prototyping of PDMS Microdevices via "Liquid Molding"Lu, Yao; Lin, Bingcheng; Qin, JianhuaAnalytical Chemistry (Washington, DC, United States) (2011), 83 (5), 1830-1835CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)This report describes the use of patterned paper as a low-cost, flexible substrate for rapidly prototyping PDMS microdevices via "liq. molding". The entire fabrication process consists simply of three steps: (1) fabrication of patterned paper in NC membrane by direct wax printing (or modified wax printing that we call "transfer wax printing"); (2) formation of liq. mold on wax-patterned NC membrane; (3) PDMS molding and curing on wax-patterned NC membrane anchored with liq. micropatterns. All these procedures can be finished within only 1.5 h without the use of a photomask, photoresist, UV lamp, etc. Through the use of wax-patterned NC membrane coupled with a liq. mold as a template, different PDMS microdevices such as microwells and microchannels have been fabricated to demonstrate the usefulness of the method for PDMS microfabrication. The height of microwells and microchannels can also be tailored flexibly by adjusting the liq. filling vol. This method for prototyping PDMS microdevices has some favorable merits including simple operation procedures, fast concept-to-device time, and low cost, indicating its potential for simple PDMS microdevice fabrication and applications.
- 45Arena A. Donato N. Saitta G. Bonavita A. Rizzo G. Neri G. Flexible ethanol sensors on glossy paper substrates operating at room temperature Sensors and Actuators B: Chemical 2010 145 488 494Google Scholar45Flexible ethanol sensors on glossy paper substrates operating at room temperatureArena, A.; Donato, N.; Saitta, G.; Bonavita, A.; Rizzo, G.; Neri, G.Sensors and Actuators, B: Chemical (2010), 145 (1), 488-494CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Flexible sensors for the detection of ethanol at room temp. have been developed by depositing sensitive layers consisting of ITO nanopowder dispersed into poly-diallyldimethylammonium chloride (PDDAC), on the top of interdigitated electrodes based on conductive carbon nanotubes, previously printed onto flexible glossy paper substrates. The morphol. and the microstructure of the interdigitated electrodes together with org.-inorg. sensing layer have been investigated by means of SEM, XRD and FT-IR. The elec. behavior of the developed sensors at room temp. in response to low ethanol concns. has been discussed as object of our research. The good ethanol sensing properties exhibited at room temp. by the composite film was supposed to originate from the electrostatic interaction occurring between the pos. charged PDDAC polyelectrolyte and neg. charged ITO particles.
- 46Dungchai W. Chailapakul O. Henry C. S. A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printing Analyst 2011 136 77 82Google Scholar46A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printingDungchai, Wijitar; Chailapakul, Orawon; Henry, Charles S.Analyst (Cambridge, United Kingdom) (2011), 136 (1), 77-82CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Wax screen-printing as a low-cost, simple, and rapid method for fabricating paper-based microfluidic devices (μPADs) is reported here. Solid wax was rubbed through a screen onto paper filters. The printed wax was then melted into the paper to form hydrophobic barriers using only a hot plate. The authors first studied the relationship between the width of a hydrophobic barrier and the width of the original design line. The authors also optimized the heating temp. and time and detd. the resoln. of structures fabricated using this technique. The min. width of hydrophilic channel and hydrophobic barrier is 650 and 1300 μm, resp. Next, the authors fabrication method was compared to a photolithog. method using the reaction between bicinchoninic acid (BCA) and Cu1+ to demonstrate differences in background reactivity. Photolithog. defined channels exhibited a high background while wax printed channels showed a very low background. Finally, the utility of wax screen-printing was demonstrated for the simultaneous detn. of glucose and total iron in control human serum samples using an electrochem. method with glucose oxidase and a colorimetric method with 1,10-phenanthroline. This study demonstrates that wax screen-printing is an easy-to-use and inexpensive alternative fabrication method for μPAD, which will be esp. useful in developing countries.
- 47Dungchai W. Chailapakul O. Henry C. S. Electrochemical detection for paper-based microfluidics Analytical chemistry 2009 81 5821 5826Google Scholar47Electrochemical Detection for Paper-Based MicrofluidicsDungchai, Wijitar; Chailapakul, Orawon; Henry, Charles S.Analytical Chemistry (Washington, DC, United States) (2009), 81 (14), 5821-5826CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)The authors report the first demonstration of electrochem. detection for paper-based microfluidic devices. Photolithog. was used to make microfluidic channels on filter paper, and screen-printing technol. was used to fabricate electrodes on the paper-based microfluidic devices. Screen-printed electrodes on paper were characterized using cyclic voltammetry to demonstrate the basic electrochem. performance of the system. The utility of the authors' devices was then demonstrated with the detn. of glucose, lactate, and uric acid in biol. samples using oxidase enzyme (glucose oxidase, lactate oxidase, and uricase, resp.) reactions. Oxidase enzyme reactions produce H2O2 while decompg. their resp. substrates, and therefore a single electrode type is needed for detection of multiple species. Selectivity of the working electrode for H2O2 was improved using Prussian Blue as a redox mediator. The detn. of glucose, lactate, and uric acid in control serum samples was performed using chronoamperometry at the optimal detection potential for H2O2 (0 V vs. the on-chip Ag/AgCl ref. electrode). Levels of glucose and lactate in control serum samples measured using the paper devices were 4.9±0.6 and 1.2±0.2 mM (level I control sample), and 16.3±0.7 and 3.2±0.3 mM (level II control sample), resp., and were within error of the values measured using traditional tests. This study shows the successful integration of paper-based microfluidics and electrochem. detection as an easy-to-use, inexpensive, and portable alternative for point of care monitoring.
- 48Suresh R. R. Lakshmanakumar M. Arockia Jayalatha J. Rajan K. Sethuraman S. Krishnan U. M. Rayappan J. B. B. Fabrication of screen-printed electrodes: opportunities and challenges Journal of Materials Science 2021 56 8951 9006Google Scholar48Fabrication of screen-printed electrodes: opportunities and challengesSuresh, Raghavv Raghavender; Lakshmanakumar, Muthaiyan; Arockia Jayalatha, J. B. B.; Rajan, K. S.; Sethuraman, Swaminathan; Krishnan, Uma Maheswari; Rayappan, John Bosco BalaguruJournal of Materials Science (2021), 56 (15), 8951-9006CODEN: JMTSAS; ISSN:0022-2461. (Springer)Abstr.: In recent times, wearable electronics, real-time sensing devices and point-of-care device applications have seen a surge in demand. This demand inherently calls for a more economic and reliable method of mass prodn. One such robust technique is screen printing that offers advantages in terms of being versatile, economical and easy to use. This technique has been proclaimed to be the best amongst the other additive manufg. techniques by many research groups. This can be solely attributed to its simple equipment design, requiring a paste for printing purpose, a meshwork for housing the design and a squeegee to carry out printing through an up-and-down motion. This subsequently calls for optimizing the parameters such as ink rheol., pore size of the mesh, proper choice of mesh design and motion of the squeeze in order to fabricate electrodes for desired purpose. Due to this technique's immense potential to open up broad inroads in the domain of flexible electronics, whose concept has radically redefined the perception towards the field of electronics, a review article encompassing an elaborate description on the science and other quintessential parameters involved in this mass printing technique would prove to be extremely useful. In this purview, the review article is compartmentalised into sections encompassing discussion on the manufg. of different kinds of inks for screen printing applications, substrates used, electrode design and pre-treatment procedures. Graphical abstr.: [graphic not available: see fulltext].
- 49Clark L. C. Jr Lyons C. Electrode systems for continuous monitoring in cardiovascular surgery Annals of the New York Academy of sciences 1962 102 29 45Google Scholar49Electrode systems for continuous monitoring in cardiovascular surgeryClark, Leland C., Jr.; Lyons, ChampAnnals of the New York Academy of Sciences (1962), 102 (Art. 1), 29-45CODEN: ANYAA9; ISSN:0077-8923.App. is described for the continuous monitoring of blood chemistry in surgical patients. External electrode systems record quant. pH, O, and CO2; intravascular electrode systems record H and ascorbate indicator diln. curves. 21 references.
- 50Bachan Upadhyay L. S. Verma N. Enzyme inhibition based biosensors: A review Analytical Letters 2013 46 225 241Google ScholarThere is no corresponding record for this reference.
- 51Abdelwahab A. A. Won M. S. Shim Y. B. Direct electrochemistry of cholesterol oxidase immobilized on a conducting polymer: Application for a cholesterol biosensor Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2010 22 21 25Google ScholarThere is no corresponding record for this reference.
- 52Premanode B. Toumazou C. A novel, low power biosensor for real time monitoring of creatinine and urea in peritoneal dialysis Sensors and Actuators B: Chemical 2007 120 732 735Google Scholar52A novel, low power biosensor for real time monitoring of creatinine and urea in peritoneal dialysisPremanode, Bhusana; Toumazou, ChrisSensors and Actuators, B: Chemical (2007), 120 (2), 732-735CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Novel biosensors, based on immobilized creatininase, creatinase and urease are developed, using ISFETs with weak inversion at pH 6-8 and 37.0 °C. The ISFETs with circuitry, demonstrate a linear relationship of urea and creatinine at the range of 0-200 and 0-20 mM, resp. Preliminary results show that biosensors operating in weak inversion mode can eliminate many of the disadvantages of ISFETs operating in the strong inversion region, providing a wide dynamic range output in nanoAmp. Such characteristics fit the anal. requirements for improving real-time monitoring in peritoneal dialysis (PD). Further work covers stability of ISFET sensors biased with CMOS circuits in the weak inversion mode working in the room temp. of 15-40 °C.
- 53Zourob M. Simonian A. Wild J. Mohr S. Fan X. Abdulhalim I. Goddard N. Optical leaky waveguide biosensors for the detection of organophosphorus pesticides Analyst 2007 132 114 120Google Scholar53Optical leaky waveguide biosensors for the detection of organophosphorus pesticidesZourob, M.; Simonian, A.; Wild, J.; Mohr, S.; Fan, Xudong; Abdulhalim, I.; Goddard, N. J.Analyst (Cambridge, United Kingdom) (2007), 132 (2), 114-120CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Organophosphorus (OP) pesticides can be rapidly detected by integrating organophosphorus hydrolase with an optical leaky waveguide biosensor. This enzyme catalyzes the hydrolysis of a wide range of organophosphorus compds. causing an increase in the pH. Thus, the direct detection of OP is possible by monitoring of the pH changes assocd. with the enzyme's activity. The use of an optical, leaky waveguide clad with absorbing materials for the detection of OP pesticides by measuring changes in refractive index, absorbance and fluorescence was described. In the most effective configuration, a thick sensing layer was used to increase the amt. of immobilized enzyme and to increase the light interaction with the sensing layer, resulting in a greatly enhanced sensitivity. The platforms developed in this work were successfully used to detect paraoxon and parathion down to 4 nM concns.
- 54Zhu H. Li L. Zhou W. Shao Z. Chen X. Advances in non-enzymatic glucose sensors based on metal oxides Journal of Materials Chemistry B 2016 4 7333 7349Google Scholar54Advances in non-enzymatic glucose sensors based on metal oxidesZhu, Hua; Li, Li; Zhou, Wei; Shao, Zongping; Chen, XianjianJournal of Materials Chemistry B: Materials for Biology and Medicine (2016), 4 (46), 7333-7349CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)A review. Glucose sensors have been extensively developed because of their broad applications, esp. in diabetes diagnosis. Up to date, electrochem. enzymic glucose sensors are commonly used in daily life for glucose detection and com. successful as glucose-meters because they exhibit excellent selectivity, high reliability, and could be handled under physiol. pH conditions. However, considering some intrinsic disadvantages of enzymes, such as high fabrication cost and poor stability, non-enzymic glucose sensors have attracted increasing research interest in recent years due to their low cost, high stability, prompt response, and low detection limit. Furthermore, the development of nanotechnol. has also offered new opportunities to construct nanostructured electrodes for glucose sensing applications. With distinguished advantages, metal oxides have garnered extensive effort in the development of cost-effective sensors with high stability, sensitivity and quick response for the detn. of glucose via electrochem. oxidn. Hence, this review summarizes the advances in non-enzymic glucose sensors based on different metal oxides (such as ZnO, CuO/Cu2O, NiO, Co3O4, MnO2, etc.) and their nanocomposites. Addnl., a brief prospective is presented on metal oxides for glucose sensors.
- 55Raziq A. Tariq M. Hussian R. Mehmood M. H. Khan M. S. Hassan A. Electrochemical investigation of glucose oxidation on a glassy carbon electrode using voltammetric, amperometric, and digital simulation methods ChemistrySelect 2017 2 9711 9717Google Scholar55Electrochemical Investigation of Glucose Oxidation on a Glassy Carbon Electrode Using Voltammetric, Amperometric, and Digital Simulation MethodsRaziq, Abdul; Tariq, Muhammad; Hussian, Riaz; Mehmood, Muhammad Haris; Khan, Muhammad Saleem; Hassan, AyazChemistrySelect (2017), 2 (30), 9711-9717CODEN: CHEMUD; ISSN:2365-6549. (Wiley-VCH Verlag GmbH & Co. KGaA)In this paper, we report the use of glassy carbon electrode for electrochem. oxidn. of glucose in basic medium. The effect of glucose concn., scan rate, and point potential was studied. Heterogeneous electron transfer parameters such as std. electron transfer rate const. (k0), transfer coeff. (a), and E0 were calcd. using cyclic voltammetry (CV), semi integration of cyclic voltammetry (SICV), semi differentiation of cyclic voltammetry (SDCV), and digital simulation techniques. Amperometric expts. were performed for detn. of glucose concn. in basic media. These expts. show a broad linear concn. range, up to 11 mM with a sensitivity of 927.96μAmM-1cm-2. The lower detection limit (LOD) and Lower Quantification limit (LOQ) were found to be 50μM and 0.5 mM, resp. Effect of interfering species such as uric acid, ascorbic acid, acetaminophen, and chloride ion on the electrocatalytic behavior of glassy carbon electrode (GCE) was also studied.
- 56Pletcher D. Electrocatalysis: present and future Journal of applied electrochemistry 1984 14 403 415Google Scholar56Electrocatalysis: present and futurePletcher, DerekJournal of Applied Electrochemistry (1984), 14 (4), 403-15CODEN: JAELBJ; ISSN:0021-891X.A review with 61 refs.
- 57Zhu H. Li L. Zhou W. Shao Z. Chen X. Correction: Advances in non-enzymatic glucose sensors based on metal oxides Journal of Materials Chemistry B 2017 5 1117 1117Google Scholar57Correction: Advances in non-enzymatic glucose sensors based on metal oxides [Erratum to document cited in CA165:577563]Zhu, Hua; Li, Li; Zhou, Wei; Shao, Zongping; Chen, XiangjianJournal of Materials Chemistry B: Materials for Biology and Medicine (2017), 5 (5), 1117CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)A review. The name of Xiangjian Chen was spelled incorrectly. There is also a correction for the acknowledgments.
- 58Burke L. Premonolayer oxidation and its role in electrocatalysis Electrochimica Acta 1994 39 1841 1848Google Scholar58Premonolayer oxidation and its role in electrocatalysisBurke, L. D.Electrochimica Acta (1994), 39 (11-12), 1841-8CODEN: ELCAAV; ISSN:0013-4686.A discussion with 70 refs. is given. Hydrous oxides are considered here as an integral component of noble metal electrochem.; they are regarded as quite distinct from, and in certain areas more important than, monolayer oxide deposits. The usual polycryst. metal surface is assumed to be composed of two major components: (i) atoms of high lattice co-ordination no. (the vast majority) than oxidize to yield monolayer oxide species, and (ii) atoms of quite low lattice coordination no. (and of very low coverage) that oxidize to yield incipient hydrous oxide species. Evidence, that is now quite strong in the case of gold, supporting this view is summarized. Data are provided to support the view that in many instances electrocatalytic processes at noble metal electrode/aq. soln. interfaces involve hydrous oxides as mediators and inhibitors in oxidn. and redn. processes, resp. Premonolayer oxidn. of metals, in terms of this approach, is considered as an interesting, relevant and challenging area of research.
- 59Warsinke A. Benkert A. Scheller F. Electrochemical immunoassays Fresenius’ journal of analytical chemistry 2000 366 622 634Google Scholar59Electrochemical immunoassaysWarsinke, A.; Benkert, A.; Scheller, F. W.Fresenius' Journal of Analytical Chemistry (2000), 366 (6-7), 622-634CODEN: FJACES; ISSN:0937-0633. (Springer-Verlag)A review with 105 refs. Immunoassays (IA) use the specific antigen-antibody complexation for anal. purposes. RIAs, fluorescence immunoassays (FIA) and enzyme immunoassays (EIA) are well established in clin. diagnostics. For the development of hand-held devices which can be used for point of care measurements, electrochem. immunoassays are promising alternatives to existing immunochem. tests. Moreover, for opaque or optically dense matrixes electrochem. methods are superior. Potentiometric, capacitive and amperometric transducers have been applied for direct and indirect electrochem. immunoassays. However, due to their fast detection, broad linear range and low detection limit, amperometric transducers are preferred. Competitive and noncompetitive amperometric immunoassays have been developed with redox compds. or enzymes as labels. This review gives an overview of the most frequently applied principles in electrochem. immunoassays. The potential of an indirect competitive amperometric immunoassay for the detn. of creatinine within nanomolar range and the circumvention of the most serious problem in electrochem. immunoassays, namely regeneration, is discussed.
- 60Volpe G. Fares G. Delli Quadri F. Draisci R. Ferretti G. Marchiafava C. Moscone D. Palleschi G. A disposable immunosensor for detection of 17β-estradiol in non-extracted bovine serum Analytica chimica acta 2006 572 11 16Google Scholar60A disposable immunosensor for detection of 17β-estradiol in non-extracted bovine serumVolpe, G.; Fares, G.; delli Quadri, F.; Draisci, R.; Ferretti, G.; Marchiafava, C.; Moscone, D.; Palleschi, G.Analytica Chimica Acta (2006), 572 (1), 11-16CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)This paper reports the assembly of a disposable immunosensor based on the direct competitive ELISA, for simple and fast measurement of 17β-estradiol (17β-E2) in bovine serum, using screen-printed electrodes (SPEs) and a Palm-Sens portable electrochem. detector. The immunosensor strip was assembled immobilizing, by passive adsorption, anti-rabbit IgG onto the surface of the working SPE electrode. After the interaction between anti-rabbit IgG and rabbit anti-17β-E2 polyclonal antibodies (PAb), the competition was performed using 17β-estradiol-alk. phosphatase conjugate (17β-E2-AP) synthesized in our lab. The enzymic substrate used for signal generation was 1-naphthylphosphate and its conversion to an electroactive product (1-naphthol) was measured using differential pulse voltammetry (DPV). To develop a prototype for field measurements, the entire competitive protocol has been optimized directly in a blank non-extd. bovine serum. According to the new EU criteria established by the Commission Decision 2002/657/EC for qual. and quant. screening methods, the detection capability (CCβ), was detd. The CCβ value resulted below the action limit (40 pg mL-1) fixed for 17β-E2. Spiked and real samples were analyzed using the electrochem. immunostrips obtaining precision values (relative std. deviation, R.S.D.%) ranging from 8.6 to 17.0% and a recovery (R%) from 88.2 to 120.0%. Results obtained on real samples were confirmed by liq. chromatog. coupled online with tandem mass spectrometry (LC-MS/MS) using an atm. pressure chem. ionization (APCI) source and a heated nebulizer (HN) interface; this is the method currently used to confirm illegal hormone administration for regulatory purposes. The disposable immunosensor appears suitable as a screening tool for field anal. of bovine serum estradiol.
- 61Bagel O. Degrand C. Limoges B. Joannes M. Azek F. Brossier P. Enzyme affinity assays involving a single‐use electrochemical sensor. Applications to the enzyme immunoassay of human chorionic gonadotropin hormone and nucleic acid hybridization of human cytomegalovirus DNA Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2000 12 1447 1452Google ScholarThere is no corresponding record for this reference.
- 62Carpini G. Lucarelli F. Marrazza G. Mascini M. Oligonucleotide-modified screen-printed gold electrodes for enzyme-amplified sensing of nucleic acids Biosensors and Bioelectronics 2004 20 167 175Google Scholar62Oligonucleotide-modified screen-printed gold electrodes for enzyme-amplified sensing of nucleic acidsCarpini, Guido; Lucarelli, Fausto; Marrazza, Giovanna; Mascini, MarcoBiosensors & Bioelectronics (2004), 20 (2), 167-175CODEN: BBIOE4; ISSN:0956-5663. (Elsevier)An electrochem. genosensor for the detection of specific sequences of DNA has been developed using disposable screen-printed gold electrodes. Screen-printed gold electrodes were firstly modified with a mixed monolayer of a 25-mer thiol-tethered DNA probe and a spacer thiol, 6-mercapto-1-hexanol (MCH). The DNA probe sequence was internal to the sequence of the 35S promoter, which sequence is inserted in the genome of GMOs regulating the transgene expression. An enzyme-amplified detection scheme, based on the coupling of a streptavidin-alk. phosphatase conjugate and biotinylated target sequences was then applied. The enzyme catalyzed the hydrolysis of the electroinactive α-naphthyl phosphate to α-naphthol; this product is electroactive and has been detected by means of differential pulse voltammetry. The assay was, firstly, characterized using synthetic oligonucleotides. Relevant parameters, such as the probe concn. and the immobilization time, the use of the MCH and different enzymic conjugates, were investigated and optimized. The genosensor response was found to be linearly related to the target concn. between 0 and 25 nmol/L; the detection limit was 0.25 nmol/L. The anal. procedure was then applied for the detection of the 35S promoter sequence, which was amplified from the pBI121 plasmid by polymerase chain reaction (PCR). Hybridization conditions (i.e., hybridization buffer and hybridization time) were further optimized. The selectivity of the assay was confirmed using biotinylated non-complementary amplicons and PCR blanks. The results showed that the genosensor enabled sensitive (detection limit: 1 nmol/L) and specific detection of GMO-related sequences, thus providing a useful tool for the screening anal. of bioengineered food samples.
- 63Rao V. K. Sharma M. K. Goel A. K. Singh L. Sekhar K. Amperometric immunosensor for the detection of Vibrio cholerae O1 using disposable screen-printed electrodes Analytical sciences 2006 22 1207 1211Google Scholar63Amperometric immunosensor for the detection of Vibrio cholerae O1 using disposable screen-printed electrodesRao, Vepa K.; Sharma, Mukesh K.; Goei, Ajay K.; Singh, Lokendra; Sekhar, KrishnamurthiAnalytical Sciences (2006), 22 (9), 1207-1211CODEN: ANSCEN; ISSN:0910-6340. (Japan Society for Analytical Chemistry)A disposable amperometric immunosensor was studied for the rapid detection of Vibrio cholerae (V. cholerae), the causative agent of cholera, employing an indirect sandwich enzyme linked immunosorbent assay (ELISA) principle. Screen-printed electrodes (SPEs) were fabricated (by using com. and homemade carbon inks), electrochem. characterized and the assay conditions were optimized for capturing antibodies and antigen. Whole cell lysate (WCL) of V. cholerae was used to raise antibodies in rabbits and mice. The antibodies raised against WCL of V. cholerae were found to be specific, and no cross reactivity was obsd. with other enteric bacteria. 1-Naphthyl phosphate was used as a substrate with the amperometric detection of its enzymic hydrolysis product 1-naphthol at a potential of +400 mV vs. Ag/AgCl ref. electrode. A comparison between the amperometric detection technique and the std. ELISA was made in terms of the total assay time, the amt. of biol. materials used and the sensitivity of detection. The min. detection limit of the amperometric immunosensor for V. cholerae was found to be 105 cells/mL in 55 min, while ELISA detected 106 cells/mL in 4 h.
- 64Keay R. McNeil C. Separation-free electrochemical immunosensor for rapid determination of atrazine Biosensors and Bioelectronics 1998 13 963 970Google Scholar64Separation-free electrochemical immunosensor for rapid determination of atrazineKeay, R. W.; McNeil, C. J.Biosensors & Bioelectronics (1998), 13 (9), 963-970CODEN: BBIOE4; ISSN:0956-5663. (Elsevier Science Ltd.)A sepn.-free electrochem. immunoassay method for the detection of the pesticide atrazine is described. The method is a competitive ELISA incorporating disposable screen printed horseradish peroxidase modified electrodes as the detector element in conjunction with single-use atrazine immuno-membranes. Screen printed C electrodes were prepd. using C ink incorporating horseradish peroxidase. A monoclonal antibody for atrazine was immobilized onto Biodyne C membranes which were, in turn, placed over the electrode surface. The assay was based on competition for available binding sites between free atrazine and an atrazine-glucose oxidase conjugate prepd. inhouse. In the presence of glucose, H2O2 formed by the conjugate was reduced by enzyme-channeling via the HRP electrode. The HRP was in turn re-reduced by a direct electron transfer mechanism at a potential of +50 mV vs. Ag/AgCl. Any H2O2 formed in the bulk soln. by unbound atrazine-GOD conjugate was scavenged by excess catalase thus removing the requirement for a washing step. The performance of the method was compared with a com. immunoassay kit for atrazine.
- 65Audrey S. Beatriz P.-S. Jean-Louis M. Biosensors for pesticide detection: new trends American Journal of Analytical Chemistry 2012 2012Google ScholarThere is no corresponding record for this reference.
- 66Susmel S. Guilbault G. O’sullivan C. Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodes Biosensors and Bioelectronics 2003 18 881 889Google Scholar66Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodesSusmel, S.; Guilbault, G. G.; O'Sullivan, C. K.Biosensors & Bioelectronics (2003), 18 (7), 881-889CODEN: BBIOE4; ISSN:0956-5663. (Elsevier Science Ltd.)The demonstration of a labeless immunosensor for the detection of pathogenic bacteria using screen printed gold electrodes (SPGEs) and a potassium hexacyanoferrate(II) redox probe is reported. Gold electrodes were produced using screen printing and the gold surfaces were modified by a thiol based self assembled monolayer (SAM) to facilitate antibody immobilization. SAMs based on the use of thioctic acid (TA), mercaptopropionic acid (MPA) and mercaptoundecanoic acid (MUA) were evaluated. Following antibody immobilization via the optimum SAM, the redox behavior and diffusion co-efficient (D) of the potassium hexacyanoferrate(II) probe was monitored in the absence and presence of analyte. In the presence of analyte, a change in the apparent diffusion co-efficient of the redox probe was obsd., attributable to impedance of the diffusion of redox electrons to the electrode surface due to the formation of the antibody-bacteria immunocomplex. No change in the diffusion co-efficient was obsd. when a non-specific antibody (mouse IgG) was immobilized and antigen added. The system has been demonstrated with Listeria monocytogenes and Bacillus cereus.
- 67Simon D. Ware T. Marcotte R. Lund B. R. Smith D. W. Di Prima M. Rennaker R. L. Voit W. A comparison of polymer substrates for photolithographic processing of flexible bioelectronics Biomedical microdevices 2013 15 925 939Google Scholar67A comparison of polymer substrates for photolithographic processing of flexible bioelectronicsSimon, Dustin; Ware, Taylor; Marcotte, Ryan; Lund, Benjamin R.; Smith, Dennis W., Jr.; Prima, Matthew; Rennaker, Robert L.; Voit, WalterBiomedical Microdevices (2013), 15 (6), 925-939CODEN: BMICFC; ISSN:1387-2176. (Springer)Flexible bioelectronics encompass a new generation of sensing devices, in which controlled interactions with tissue enhance understanding of biol. processes in vivo. However, the fabrication of such thin film electronics with photolithog. processes remains a challenge for many biocompatible polymers. Recently, two shape memory polymer (SMP) systems, based on acrylate and thiol-ene/acrylate networks, were designed as substrates for softening neural interfaces with glass transitions above body temp. (37 °C) such that the materials are stiff for insertion into soft tissue and soften through low moisture absorption in physiol. conditions. These two substrates, acrylate and thiol-ene/acrylate SMPs, are compared to polyethylene naphthalate, polycarbonate, polyimide, and polydimethylsiloxane, which have been widely used in flexible electronics research and industry. These six substrates are compared via dynamic mech. anal. (DMA), thermogravimetric anal. (TGA), and swelling studies. The integrity of gold and chromium/gold thin films on SMP substrates are evaluated with optical profilometry and elec. measurements as a function of processing temp. above, below and through the glass transition temp. The effects of crosslink d., adhesion and cure stress are shown to play a crit. role in the stability of these thin film materials, and a guide for the future design of responsive polymeric materials suitable for neural interfaces is proposed. Finally, neural interfaces fabricated on thiol-ene/acrylate substrates demonstrate long-term fidelity through both in vitro impedance spectroscopy and the recording of driven local field potentials for 8 wk in the auditory cortex of lab. rats.
- 68Yin Z. Huang Y. Bu N. Wang X. Xiong Y. Inkjet printing for flexible electronics: Materials, processes and equipments Chinese Science Bulletin 2010 55 3383 3407Google ScholarThere is no corresponding record for this reference.
- 69Sele C. W. Von Werne T. Friend R. H. Sirringhaus H. Lithography‐free, self‐aligned inkjet printing with sub‐hundred‐nanometer resolution Advanced Materials 2005 17 997 1001Google Scholar69Lithography-free, self-aligned inkjet printing with sub-hundred-nanometer resolutionSele, Christoph W.; von Werne, Timothy; Friend, Richard H.; Sirringhaus, HenningAdvanced Materials (Weinheim, Germany) (2005), 17 (8), 997-1001CODEN: ADVMEW; ISSN:0935-9648. (Wiley-VCH Verlag GmbH & Co. KGaA)Direct inkjet printing of sub-100 nm nanoscale gaps, without the need for any high-resoln. lithog., is made possible by a novel self-aligned printing method (see Figure). The benefits of this method are demonstrated by fabrication of all-polymer transistor circuits, which exhibit switching speeds at least two orders of magnitude higher than those previously reported for inkjet-printed polymer devices.
- 70Moya A. Gabriel G. Villa R. del Campo F. J. Inkjet-printed electrochemical sensors Current Opinion in Electrochemistry 2017 3 29 39Google Scholar70Inkjet-printed electrochemical sensorsMoya, Ana; Gabriel, Gemma; Villa, Rosa; Javier del Campo, F.Current Opinion in Electrochemistry (2017), 3 (1), 29-39CODEN: COEUCY; ISSN:2451-9111. (Elsevier B.V.)A review. Inkjet printing is a digital and non-contact printing technique that is beginning to be used in the manuf. of electronic devices, including electrochem. sensors. This article covers the main works featuring inkjet printing for the prodn. of electrochem. sensors, and focuses esp. on the works reported since 2015. The main factor limiting the application of inkjet printing in the manuf. of advanced devices stems mainly from the strict rheol. conditions that the ink needs to meet. This is the reason why inkjet is best used in combination with other manufg. techniques. Given their high degree of maturity and accessibility, home/office printers will likely lead a future wave in the manuf. of electronic devices, particularly at the development and prototyping stages.
- 71Khan M. S. Fon D. Li X. Tian J. Forsythe J. Garnier G. Shen W. Biosurface engineering through ink jet printing Colloids and Surfaces B: Biointerfaces 2010 75 441 447Google Scholar71Biosurface engineering through ink jet printingKhan, Mohidus Samad; Fon, Deniece; Li, Xu; Tian, Junfei; Forsythe, John; Garnier, Gil; Shen, WeiColloids and Surfaces, B: Biointerfaces (2010), 75 (2), 441-447CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)The feasibility of thermal ink jet printing as a robust process for biosurface engineering was demonstrated. The strategy investigated was to reconstruct a com. printer and take advantage of its color management interface. High printing resoln. was achieved by formulating bio-inks of viscosity and surface tension similar to those of com. inks. Protein and enzyme denaturation during thermal ink jet printing was shown to be insignificant. This is because the time spent by the biomols. in the heating zone of the printer is negligible; in addn., the air and substrate of high heat capacity absorb any residual heat from the droplet. Gradients of trophic/tropic factors can serve as driving force for cell growth or migration for tissue regeneration. Concn. gradients of proteins were printed on scaffolds to show the capability of ink jet printing. The printed proteins did not desorb upon prolonged immersion in aq. solns., thus allowing printed scaffold to be used under in vitro and in vivo conditions. The authors' group portrait was ink jet printed with a protein on paper, illustrating that complex biopatterns can be printed on large area. Finally, patterns of enzymes were ink jet printed within the detection and reaction zones of a paper diagnostic.
- 72Behrent A. Griesche C. Sippel P. Baeumner A. J. Process-property correlations in laser-induced graphene electrodes for electrochemical sensing Microchimica Acta 2021 188 1 14Google ScholarThere is no corresponding record for this reference.
- 73Cuartero M. Crespo G. A. Bakker E. based thin-layer coulometric sensor for halide determination Analytical chemistry 2015 87 1981 1990Google Scholar73Paper-Based Thin-Layer Coulometric Sensor for Halide DeterminationCuartero, Maria; Crespo, Gaston A.; Bakker, EricAnalytical Chemistry (Washington, DC, United States) (2015), 87 (3), 1981-1990CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)The authors report on a paper-based anal. device (PAD) for the exhaustive, and therefore abs., detn. of halides in a range of diverse water samples and food supplements. A mixt. of chloride, bromide, and iodide ions is assessed in a wide range of concns., specifically, from 10-4.8 to 0.1M for bromide and iodide and from 10-4.5 to 0.6M for chloride, with a limit of detection of 10-5 M. As a result of a careful optimization of the electrochem. cell, a thin layer made of cellulose paper (75-μm thickness), a cation-exchange Donnan exclusion membrane (FKL), and a silver-foil working electrode were selected as optimum materials. Cyclic voltammetry (from 0 to 0.8 V) was chosen as the interrogation technique to impose the exhaustive oxidative plating and re-redn. of halides on the silver element, accompanied by outward and inward counterion fluxes. The scan rate plays an important role in the ability of the technique to resolve mixts. of ions. Moderate scan rates (10 mV s-1) provide a suitable compromise between sensitivity, limit of detection, and resoln. This paper-based microfluidic device is extremely simple in terms of manipulation, cost, and contamination risk. Paper is an excellent basis for the establishment of a confined thin aq. layer, the construction of disposable halide sensors, and portability for measuring outside the controlled lab. environment. A discussion of the relevant anal. characteristics is presented herein, followed by a demonstration of halide assessment in water samples (sea, tap, river, and mineral waters) and food supplements enriched with iodide and chloride as early examples.
- 74Lee S. H. Lee J. H. Tran V.-K. Ko E. Park C. H. Chung W. S. Seong G. H. Determination of acetaminophen using functional paper-based electrochemical devices Sensors and Actuators B: Chemical 2016 232 514 522Google Scholar74Determination of acetaminophen using functional paper-based electrochemical devicesLee, Sung Hwan; Lee, Joo Heon; Tran, Van-Khue; Ko, Euna; Park, Chan Ho; Chung, Woo Sung; Seong, Gi HunSensors and Actuators, B: Chemical (2016), 232 (), 514-522CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)In the present study, we constructed a functional paper fluidic device and evaluated its electrochem. performance by analyzing acetaminophen in the presence of ascorbic acid. The device was composed of a single-walled carbon nanotube (SWCNT) electrode and nafion-modified nitrocellulose membrane. Neg.-charged nafion was employed to build up a more neg. charge on the nitrocellulose membrane, and gold nanoparticles and polyglutamic acid (AuNP-PGA) were deposited on the SWCNT electrode to enhance the electrochem. performance of the device. The device had a vertical flow format in which the sample soln. flowed vertically through the paper. Using the nafion-modified nitrocellulose membrane and AuNP-PGA/SWCNT film electrode as a component of the paper fluidic device, we obtained a distinguishable acetaminophen oxidn. peak which was distinct from the ascorbic acid oxidn. peak. The acetaminophen oxidn. peak had a linear response with acetaminophen concn., varying from 50 μM to 300 μM (r2 = 0.992), which was broader than the std. drug dose range. The device exhibited a sensitivity of 13.3 mA/M and a detection limit of 15.0 μM. The device was stable with a relative std. deviation of 3.3% (up to 2 wk), and the reproducibility was 1.2-5.2%. Furthermore, the fabricated device accurately measured the amt. of acetaminophen in pharmaceutical samples.
- 75Gutiérrez-Capitán M. Baldi A. Fernández-Sánchez C. Electrochemical paper-based biosensor devices for rapid detection of biomarkers Sensors 2020 20 967Google Scholar75Electrochemical paper-based biosensor devices for rapid detection of biomarkersGutierrez-Capitan, Manuel; Baldi, Antonio; Fernandez-Sanchez, CesarSensors (2020), 20 (4), 967CODEN: SENSC9; ISSN:1424-8220. (MDPI AG)A review. In healthcare, new diagnostic tools that help in the diagnosis, prognosis, and monitoring of diseases rapidly and accurately are in high demand. For in-situ measurement of disease or infection biomarkers, point-of-care devices provide a dramatic speed advantage over conventional techniques, thus aiding clinicians in decision-making. During the last decade, paper-based anal. devices, combining paper substrates and electrochem. detection components, have emerged as important point-of-need diagnostic tools. This review highlights significant works on this topic over the last five years, from 2015 to 2019. The most relevant articles published in 2018 and 2019 are examd. in detail, focusing on device fabrication techniques and materials applied to the prodn. of paper fluidic and electrochem. cell architectures as well as on the final device assembly. Two main approaches were identified, that are, on one hand, those ones where the fabrication of the electrochem. cell is done on the paper substrate, where the fluidic structures are also defined, and, on the other hand, the fabrication of those ones where the electrochem. cell and liq.-driving paper component are defined on different substrates and then heterogeneously assembled. The main limitations of the current technologies are outlined and an outlook on the current technol. status and future prospects is given.
- 76Devarakonda S. Singh R. Bhardwaj J. Jang J. Cost-effective and handmade paper-based immunosensing device for electrochemical detection of influenza virus Sensors 2017 17 2597Google Scholar76Cost-effective and handmade paper-based immunosensing device for electrochemical detection of influenza virusDevarakonda, Sivaranjani; Singh, Renu; Bhardwaj, Jyoti; Jang, JaesungSensors (2017), 17 (11), 2597/1-2597/13CODEN: SENSC9; ISSN:1424-8220. (MDPI AG)Here, we present a cost-effective, handmade paper-based immunosensor for label-free electrochem. detection of influenza virus H1N1. This immunosensor was prepd. by modifying paper with a spray of hydrophobic silica nanoparticles, and using stencil-printed electrodes. We used a glass vaporizer to spray the hydrophobic silica nanoparticles onto the paper, rendering it super-hydrophobic. The super-hydrophobicity, which is essential for this paper-based biosensor, was achieved via 30-40 spray coatings, corresponding to a 0.39-0.41 mg cm-2 coating of nanoparticles on the paper and yielding a water contact angle of 150° ± 1°. Stencil-printed carbon electrodes modified with single-walled carbon nanotubes and chitosan were employed to increase the sensitivity of the sensor, and the antibodies were immobilized via glutaraldehyde crosslinking. Differential pulse voltammetry was used to assess the sensitivity of the sensors at various virus concns., ranging from 10 to 104 PFU mL-1, and the selectivity was assessed against MS2 bacteriophages and the influenza B viruses. These immunosensors showed good linear behaviors, improved detection times (30 min), and selectivity for the H1N1 virus with a limit of detection of 113 PFU mL-1, which is sufficiently sensitive for rapid on-site diagnosis. The simple and inexpensive methodologies developed in this study have great potential to be used for the development of a low-cost and disposable immunosensor for detection of pathogenic microorganisms, esp. in developing countries.
- 77Sarkar P. Tothill I. E. Setford S. J. Turner A. P. Screen-printed amperometric biosensors for the rapid measurement of L-and D-amino acids Analyst 1999 124 865 870Google Scholar77Screen-printed amperometric biosensors for the rapid measurement of L- and D-amino acidsSarkar, Priyabrata; Tothill, Ibtisam E.; Setford, Steven J.; Turner, Anthony P. F.Analyst (Cambridge, United Kingdom) (1999), 124 (6), 865-870CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Screen-printed three-electrode amperometric sensors incorporating L- and/or D-amino acid oxidase for the general purpose measurement of L- or D-amino acids are described. The working electrode incorporates rhodinized carbon, to facilitate hydrogen peroxide oxidn. at a decreased operating potential, and immobilized enzyme. The devices responded to all 20 common L-amino acids and all of the D-amino acids examd., the exceptions being L- and D-proline. Linear response profiles were obsd. for L-leucine, L-glycine and L-phenylalanine with limits of detection of 0.47, 0.15 and 0.20 mM resp. The devices were reproducible and exhibited stability over a 56 d test period. The biosensor compares favorably with a std. photometric amino acid test and was used to monitor milk aging effects. The assay is cheap, simple to perform and rapid, requiring only buffer-electrolyte and a small sample vol.
- 78Kelso E. McLean J. Cardosi M. F. Electrochemical detection of secreted alkaline phosphatase: Implications to cell based assays Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2000 12 490 494Google ScholarThere is no corresponding record for this reference.
- 79Neuhold C. G. Wang J. do Nascimento V. B. Kalcher K. Thick film voltammetric sensors for trace copper based on a cation-exchanger-modified surface Talanta 1995 42 1791 1798Google Scholar79Thick film voltammetric sensors for trace copper based on a cation-exchanger-modified surfaceNeuhold, Christian G.; Wang, Joseph; do Nascimento, Valberes B.; Kalcher, KurtTalanta (1995), 42 (11), 1791-8CODEN: TLNTA2; ISSN:0039-9140. (Elsevier)Strip-type, preconcg./voltammetric sensors, prepd. by incorporating a cation-exchange resin within screen-printed C inks, are described. Such single-use strips combine the efficient electrostatic accumulation of heavy metals using Hg-free surfaces. The uptake of Cu(II) from dil. solns. (under open circuit conditions) is followed by voltammetric measurements in a sep. blank soln. Various exptl. variables were optimized to yield low detection limits (e.g. 0.5 μg L-1 with 10 min accumulation) and good reproducibility (relative std. deviation, 2%). The applicability to assays of drinking H2O is demonstrated.
- 80Wang J. Tian B. Screen-printed stripping voltammetric/potentiometric electrodes for decentralized testing of trace lead Analytical Chemistry 1992 64 1706 1709Google Scholar80Screen-printed stripping voltammetric/potentiometric electrodes for decentralized testing of trace leadWang, Joseph; Tian, BaominAnalytical Chemistry (1992), 64 (15), 1706-9CODEN: ANCHAM; ISSN:0003-2700.The utility of screen-printed electrodes for stripping voltammetric and potentiometric measurements of trace metals have been evaluated toward their exploitation for single-use decentralized testing. The mercury-coated screen-printed carbon electrodes, on a plastic strip, perform in a manner comparable to conventional hanging mercury drop and mercury-coated glassy carbon electrodes. Well-defined peaks are thus obtained for microgram/L (ppb) concns. of heavy metals following very short preconcn. times. The morphol. of the resulting mercury film, as characterized by SEM, facilitates trace measurements in unstirred and nondeaerated solns., as desired in many decentralized applications. Reproducible measurements of ppb lead in 100-μL drops are thus obtained. A detection limit of 30 ng/L (ppt) lead is estd. following 10-min preconcn. Convenient quantitation of lead in urine and drinking water samples is accomplished. Besides having great potential for single-use decentralized clin. or environmental testing, the highly stable response of the screen-printed electrodes makes them very attractive for routine, low-cost, centralized operations.
- 81Beitollahi H. Nejad F. G. Shakeri S. GO/Fe 3 O 4@ SiO 2 core–shell nanocomposite-modified graphite screen-printed electrode for sensitive and selective electrochemical sensing of dopamine and uric acid Analytical Methods 2017 9 5541 5549Google Scholar81GO/Fe3O4@SiO2 core-shell nanocomposite-modified graphite screen-printed electrode for sensitive and selective electrochemical sensing of dopamine and uric acidBeitollahi, Hadi; Nejad, Fariba Garkani; Shakeri, ShahryarAnalytical Methods (2017), 9 (37), 5541-5549CODEN: AMNEGX; ISSN:1759-9679. (Royal Society of Chemistry)Herein, we report the construction of GO/Fe3O4@SiO2 core-shell nanocomposite-modified graphite screen-printed electrode that shows advantageous characteristics in terms of elec. cond., catalytic activity and surface area compared to bare screen-printed electrode. The designed sensor was used for the detn. of dopamine and uric acid. Cyclic voltammetry, differential pulse voltammetry and chronoamperometry methods were used as the diagnostics techniques. Using the modified electrode, dopamine and uric acid were detd. in the concn. ranges from 0.1 to 600.0μM and 0.75 to 300.0μM, resp., whereas the detection limits were found to be 8.9 × 10-8 M and 5.7 × 10-7 M, resp. Finally, real sample anal. was done successfully using the modified electrode.
- 82Zhao X. Zhang W. Chen H. Chen Y. Huang G. Disposable electrochemical ascorbic acid sensor based on molecularly imprinted poly (o-phenylenediamine)-modified dual channel screen-printed electrode for orange juice analysis Food analytical methods 2014 7 1557 1563Google ScholarThere is no corresponding record for this reference.
- 83Moya A. Sowade E. del Campo F. J. Mitra K. Y. Ramon E. Villa R. Baumann R. R. Gabriel G. All-inkjet-printed dissolved oxygen sensors on flexible plastic substrates Organic Electronics 2016 39 168 176Google Scholar83All-inkjet-printed dissolved oxygen sensors on flexible plastic substratesMoya, Ana; Sowade, Enrico; del Campo, Francisco J.; Mitra, Kalyan Y.; Ramon, Eloi; Villa, Rosa; Baumann, Reinhard R.; Gabriel, GemmaOrganic Electronics (2016), 39 (), 168-176CODEN: OERLAU; ISSN:1566-1199. (Elsevier B.V.)Inkjet printing is a promising alternative manufg. method to conventional std. microfabrication techniques for the development of flexible and low-cost devices. Although the use of inkjet printing for the deposition of selected materials for the development of sensor devices has been reported many times in literature, it is still a challenge and a potential route towards commercialization to completely manuf. sensor devices with inkjet technol. In this work is demonstrated the fabrication of a functional low-cost dissolved oxygen (DO) amperometric sensor with feature sizes in the micrometer range using inkjet printing. All the required technol. steps for the fabrication of a complete electrochem. three electrodes system are discussed in detail. The working and counter electrodes have been printed using a gold nanoparticle ink, whereas a silver nanoparticle ink was used to print a pseudo-ref. electrode. Both inks are com. available and can be sintered at low temps., starting already at 120°C, which allows the use of plastic substrates. In addn., a printable SU8 ink formulation cured by UV is applied as passivation layer in the sensor device. Finally, as the performance of anal. methods strongly depends on the working electrode material, is demonstrated the electrochem. feasibility of this printed DO sensor, which shows a linear response in the range between 0 and 8 mg L-1 of DO, and affords a detection limit of 0.11 mg L-1, and a sensitivity of 0.03μA L mg-1. The use of flexible plastic substrates and biocompatible inks, and the rapid prototyping and low-cost of the fabricated sensors, makes that the proposed manufg. approach opens new opportunities in the field of biol. and medical sensor applications.
- 84Das S. R. Nian Q. Cargill A. A. Hondred J. A. Ding S. Saei M. Cheng G. J. Claussen J. C. 3D nanostructured inkjet printed graphene via UV-pulsed laser irradiation enables paper-based electronics and electrochemical devices Nanoscale 2016 8 15870 15879Google Scholar843D nanostructured inkjet printed graphene via UV-pulsed laser irradiation enables paper-based electronics and electrochemical devicesDas, Suprem R.; Nian, Qiong; Cargill, Allison A.; Hondred, John A.; Ding, Shaowei; Saei, Mojib; Cheng, Gary J.; Claussen, Jonathan C.Nanoscale (2016), 8 (35), 15870-15879CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Emerging research on printed and flexible graphene-based electronics is beginning to show tremendous promise for a wide variety of fields including wearable sensors and thin film transistors. However, post-print annealing/redn. processes that are necessary to increase the elec. cond. of the printed graphene degrade sensitive substrates (e.g., paper) and are whole substrate processes that are unable to selectively anneal/reduce only the printed graphene-leaving sensitive device components exposed to damaging heat or chems. Herein a pulsed laser process is introduced that can selectively irradiate inkjet printed reduced graphene oxide (RGO) and subsequently improve the elec. cond. (Rsheet ∼0.7 kΩ .box.-1) of printed graphene above previously published reports. Furthermore, the laser process is capable of developing 3D petal-like graphene nanostructures from 2D planar printed graphene. These visible morphol. changes display favorable electrochem. sensing characteristics-ferricyanide cyclic voltammetry with a redox peak sepn. (ΔEp) ≈ 0.7 V as well as hydrogen peroxide (H2O2) amperometry with a sensitivity of 3.32 μA mM-1 and a response time of <5 s. Thus this work paves the way for not only paper-based electronics with graphene circuits, it enables the creation of low-cost and disposable graphene-based electrochem. electrodes for myriad applications including sensors, biosensors, fuel cells, and theranostic devices.
- 85Jović M. Zhu Y. Lesch A. Bondarenko A. Cortés-Salazar F. Gumy F. Girault H. H. Inkjet-printed microtiter plates for portable electrochemical immunoassays Journal of Electroanalytical Chemistry 2017 786 69 76Google Scholar85Inkjet-printed microtiter plates for portable electrochemical immunoassaysJovic, Milica; Zhu, Yingdi; Lesch, Andreas; Bondarenko, Alexandra; Cortes-Salazar, Fernando; Gumy, Frederic; Girault, Hubert H.Journal of Electroanalytical Chemistry (2017), 786 (), 69-76CODEN: JECHES; ISSN:1873-2569. (Elsevier B.V.)Herein, the authors present the large-scale fabrication of multiplexed three-electrode sensors used in a point-of-care device platform that couples a magnetic bead-based immunoassay strategy with amperometric detection for rapid and highly sensitive anal. The multiplexed sensors consisted of eight independent electrochem. cells, each with a C nanotube (CNT) working electrode, CNT counter electrode and a Ag-Ag chloride quasi-ref. electrode. The microchips were fabricated on flexible polyethylene terephthalate (PET) sheets by sequential multilayer inkjet printing (IJP) of Ag, CNT and insulator inks that were either simultaneously or subsequently post-processed (e.g. through UV photo-polymn. or photonic curing). Finally, plastic wells were mounted on top of the inkjet-printed patterns to obtain an eight-well microtiter plate where each well had a soln. capacity of 50 μL. Due to the high precision of the IJP process, the microtiter plates showed high reproducibility among the individual electrochem. cells (1-2% of deviation). Also, the microchips can be reusable for at least up to 20 times as demonstrated herein. In a customized multichannel potentiostat with eight implemented magnets matching the positions of the working electrodes, the electrochem. readout of magnetic bead based sandwich and competitive immunoassays was successfully realized for the detection of TSH and atrazine (ATR) in aq. and urine samples, resp. The achieved limits of detection for ATR (i.e. 0.01 μg/L) and TSH (i.e. 0.5 μIU/mL) demonstrated the potential of the IJP microtiter plates for the environmental and biol. quantification of analytes in a very reliable high throughput platform. IJP has certainly reached the status of a batch prodn. tool for electroanal. sensing platforms.
- 86Pu Z. Wang R. Wu J. Yu H. Xu K. Li D. A flexible electrochemical glucose sensor with composite nanostructured surface of the working electrode Sensors and Actuators B: Chemical 2016 230 801 809Google Scholar86A flexible electrochemical glucose sensor with composite nanostructured surface of the working electrodePu, Zhihua; Wang, Ridong; Wu, Jianwei; Yu, Haixia; Xu, Kexin; Li, DachaoSensors and Actuators, B: Chemical (2016), 230 (), 801-809CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Detecting hypoglycemia remains a great challenge with regard to continuous glucose monitoring in clin. settings. This paper investigates a novel, flexible three-electrode electrochem. sensor with a composite nanostructured working electrode surface that has been modified by graphene and gold nanoparticles in order to detect low levels of glucose with high accuracy. The sensor electrodes were fabricated on a polyimide substrate using the flexible printed circuit board (PCB) method. Graphene was modified directly onto the working electrode surface via inkjet printing, an emerging method for micro-scale fabrications, to enable glucose detection at low levels. Gold nanoparticles were electrodeposited directly onto the graphene layer to enhance the sensitivity of the sensor. The exptl. results demonstrate that the proposed sensor can precisely measure glucose with a linear range of 0-40 mg/dL and a detection limit of 0.3 mg/dL (S/N = 3), thereby demonstrating potential for hypoglycemia detection. Moreover, this flexible sensor was suitable for integration within a microfluidic chip, which could be used to transdermally ext. and collect ISF, such that a wearable device could be developed for continuous glucose monitoring.
- 87Da Costa T. H. Song E. Tortorich R. P. Choi J.-W. A paper-based electrochemical sensor using inkjet-printed carbon nanotube electrodes ECS Journal of Solid State Science and Technology 2015 4 S3044Google Scholar87A Paper-Based Electrochemical Sensor Using Inkjet-Printed Carbon Nanotube Electrodesda Costa, Tallis H.; Song, Edward; Tortorich, Ryan P.; Choi, Jin-WooECS Journal of Solid State Science and Technology (2015), 4 (10), S3044-S3047CODEN: EJSSBG; ISSN:2162-8769. (Electrochemical Society)This paper presents a fully inkjet-printed electrochem. sensor on paper which consists of carbon nanotube-printed working, ref., and counter electrodes. The proposed technique aims at low-cost and disposable paper-based electrochem. sensors. First, a carbon nanotube (CNT) ink was inkjet-printed directly on paper, forming a conductive network. Addnl., a hydrophobic barrier was patterned on paper to limit the absorption of liq. to the designed area. The inkjet printing method allows for rapid patterning of electrodes on paper, resulting in a simple and effective electrochem. sensor. The sheet resistance of the CNT-printed paper was as low as 1 kΩ/square after 33 prints. A potential step voltammetry method was applied to det. the concn. of the analytes, iron ion (Fe2+) and dopamine (DA), with linear ranges of 10 μM-200 μM and 10 μM-100 μM, resp. The reported approach for a fully inkjet-printed electrochem. sensor is easy and cheap, and it has a potential for simple and rapid paper-based point-of-care diagnostics.
- 88Cinti S. Arduini F. Moscone D. Palleschi G. Gonzalez-Macia L. Killard A. J. Cholesterol biosensor based on inkjet-printed Prussian blue nanoparticle-modified screen-printed electrodes Sensors and Actuators B: Chemical 2015 221 187 190Google Scholar88Cholesterol biosensor based on inkjet-printed Prussian blue nanoparticle-modified screen-printed electrodesCinti, Stefano; Arduini, Fabiana; Moscone, Danila; Palleschi, Giuseppe; Gonzalez-Macia, Laura; Killard, Anthony J.Sensors and Actuators, B: Chemical (2015), 221 (), 187-190CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Here we describe the construction and optimization of a cholesterol biosensor based on screen-printed electrodes (SPEs) modified with inkjet-printed Prussian blue nanoparticles (PBNPs). The deposition of PBNPs using inkjet printing led to the highly facile fabrication of sensors with excellent sensitivity and reproducibility for the measurement of H2O2. Further integration of the sensor with a microfabricated low vol. (4 μL) sample cell allowed the measurement of cholesterol in serum with the addn. of cholesterol oxidase. The biosensor exhibited a sensitivity to cholesterol of 2.1 μA/mM cm2 (r2 = 0.97, n = 5) and was linear in the range of 0-15 mM.
- 89Lei Y. Alshareef A. H. Zhao W. Inal S. Laser-scribed graphene electrodes derived from lignin for biochemical sensing ACS Applied Nano Materials 2019 3 1166 1174Google ScholarThere is no corresponding record for this reference.
- 90Nayak P. Kurra N. Xia C. Alshareef H. N. Highly efficient laser scribed graphene electrodes for on‐chip electrochemical sensing applications Advanced Electronic Materials 2016 2 1600185Google ScholarThere is no corresponding record for this reference.
- 91Soares R. R. Hjort R. G. Pola C. C. Parate K. Reis E. L. Soares N. F. McLamore E. S. Claussen J. C. Gomes C. L. Laser-induced graphene electrochemical immunosensors for rapid and label-free monitoring of Salmonella enterica in chicken broth ACS sensors 2020 5 1900 1911Google Scholar91Laser-Induced Graphene Electrochemical Immunosensors for Rapid and Label-Free Monitoring of Salmonella enterica in Chicken BrothSoares, Raquel R. A.; Hjort, Robert G.; Pola, Cicero C.; Parate, Kshama; Reis, Efraim L.; Soares, Nilda F. F.; McLamore, Eric S.; Claussen, Jonathan C.; Gomes, Carmen L.ACS Sensors (2020), 5 (7), 1900-1911CODEN: ASCEFJ; ISSN:2379-3694. (American Chemical Society)Food-borne illnesses are a growing concern for the food industry and consumers, with millions of cases reported every year. Consequently, there is a crit. need to develop rapid, sensitive, and inexpensive techniques for pathogen detection in order to mitigate this problem. However, current pathogen detection strategies mainly include time-consuming lab. methods and highly trained personnel. Electrochem. in-field biosensors offer a rapid, low-cost alternative to lab. techniques, but the electrodes used in these biosensors require expensive nanomaterials to increase their sensitivity, such as noble metals (e.g., platinum, gold) or carbon nanomaterials (e.g., carbon nanotubes, or graphene). Herein, we report the fabrication of a highly sensitive and label-free laser-induced graphene (LIG) electrode that is subsequently functionalized with antibodies to electrochem. quantify the food-borne pathogen Salmonella enterica serovar Typhimurium. The LIG electrodes were produced by laser induction on the polyimide film in ambient conditions and, hence, circumvent the need for high-temp., vacuum environment, and metal seed catalysts commonly assocd. with graphene-based electrodes fabricated via chem. vapor deposition processes. After functionalization with Salmonella antibodies, the LIG biosensors were able to detect live Salmonella in chicken broth across a wide linear range (25 to 105 CFU mL-1) and with a low detection limit (13 ± 7 CFU mL-1; n = 3, mean ± std. deviation). These results were acquired with an av. response time of 22 min without the need for sample preconcn. or redox labeling techniques. Moreover, these LIG immunosensors displayed high selectivity as demonstrated by nonsignificant response to other bacteria strains. These results demonstrate how LIG-based electrodes can be used for electrochem. immunosensing in general and, more specifically, could be used as a viable option for rapid and low-cost pathogen detection in food processing facilities before contaminated foods reach the consumer.
- 92Lin X. Lu Z. Dai W. Liu B. Zhang Y. Li J. Ye J. Laser engraved nitrogen-doped graphene sensor for the simultaneous determination of Cd (II) and Pb (II) Journal of Electroanalytical Chemistry 2018 828 41 49Google Scholar92Laser engraved nitrogen-doped graphene sensor for the simultaneous determination of Cd(II) and Pb(II)Lin, Xueni; Lu, Zhiwei; Dai, Wanlin; Liu, Baichen; Zhang, Yuxin; Li, Junye; Ye, JianshanJournal of Electroanalytical Chemistry (2018), 828 (), 41-49CODEN: JECHES; ISSN:1873-2569. (Elsevier B.V.)Recently, graphene-based nanomaterials have attracted widespread attention as new electrode-modified materials for different electrochem. sensing platforms. However, graphene-based nanomaterials modified heavy metal sensors often have the disadvantages in that their detection ranges are not wide enough. To solve this problem, an easy and feasible approach derived from laser engraving technique to fabricate a high-quality 3-dimensional porous graphene-based heavy metal sensor with a quite wide detection range was established. The N-doped laser engraved graphene (N@LEG) was synthesized via introducing polyaniline (PANI) and polyvinylpyrrolidone (PVP) as N-dopant. Herein, by coupling the unique electrochem. properties and the 3-dimensional porous structure framework with large electrochem. active surface areas of LEG with the strong metal ions affinity of N atoms, N@LEG modified glassy C electrode (N@LEG/GCE) with in-situ Bi film modification was successfully used for the simultaneous detn. of Cd(II) and Pb(II) using square wave anodic stripping voltammetry (SWASV). After optimizing conditions, the proposed sensor exhibits quite wide linear ranges varying from 5 to 10 g L-1 and 10 to 380 g L-1 for Cd(II), and 0.5 to 10 μg L-1 and 10 μg L-1 to 380 μg L-1 for Pb(II), resp. The detection limits are 1.08 g L-1 (S/N = 3) for Cd(II) and 0.16 g L-1 (S/N = 3) for Pb(II), which are nearly 3 times and 60 times less than the guideline values of the drinking H2O presented by the World Health Organization (WHO), for Cd(II) and Pb(II), resp. Also, the results describe satisfactory advantages of remarkable sensitivity, selectivity, anti-interference, repeatability, reproducibility and stability. Besides, the fabricated sensor is a potential perspective to detect Cd(II) and Pb(II) in actual H2O samples.
- 93Cardoso A. R. Marques A. C. Santos L. Carvalho A. F. Costa F. M. Martins R. Sales M. G. F. Fortunato E. Molecularly-imprinted chloramphenicol sensor with laser-induced graphene electrodes Biosensors and Bioelectronics 2019 124 167 175Google Scholar93Molecularly-imprinted chloramphenicol sensor with laser-induced graphene electrodesCardoso, Ana R.; Marques, Ana C.; Santos, Lidia; Carvalho, Alexandre F.; Costa, Florinda M.; Martins, Rodrigo; Sales, M. Goreti F.; Fortunato, ElviraBiosensors & Bioelectronics (2019), 124-125 (), 167-175CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)Graphene has emerged as a novel material with enhanced elec. and structural properties that can be used for a multitude of applications from super-capacitors to biosensors. In this context, an ultra-sensitive biosensor was developed using a low-cost, simple and mask-free method based on laser-induced graphene technique for electrodes patterning. The graphene was produced on a polyimide substrate, showing a porous multi-layer structure with a resistivity of 102.4 ± 7.3 Ω/square. The biosensor was designed as a 3-electrode system. Auxiliary and working electrodes were made of graphene by laser patterning and the ref. electrode was handmade by casting a silver ink. A molecularly-imprinted polymer (MIP) was produced at the working electrode by direct electropolymn. of eriochrome black T (EBT). As proof-of-concept, the MIP film was tailored for chloramphenicol (CAP), a common contaminant in aquaculture. The resulting device was evaluated by cyclic voltammetry and electrochem. impedance spectroscopy readings against a redox std. probe. The limit of detection (LOD) was 0.62 nM and the linear response ranged from 1 nM to 10 mM. These anal. features were better than those produced by assembling the same biorecognition element on com. graphene- and carbon-based screen-printed electrodes. Overall, the simplicity and quickness of the laser-induced graphene technique, along with the better anal. features obtained with the graphene-based electrodes, shows the potential to become a com. approach for on-site sensing.
- 94S. Nasraoui, A. Al-Hamry, A. Anurag, P. R. Teixeira, S. Ameur, L. G. Paterno, M. B. Ali, O. Kanoun , Investigation of laser induced graphene electrodes modified by MWNT/AuNPs for detection of nitrite, in: 2019 16th International Multi-Conference on Systems, Signals & Devices (SSD); IEEE, 2019; pp 615- 620.Google ScholarThere is no corresponding record for this reference.
Figure 1
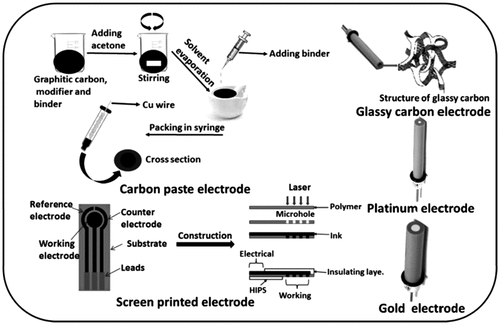 Figure 1. Various types of electrodes and their structures used for modification in sensing applications.Adapted with permission from ref. ( 36). Copyright 2019 Royal Society of Chemistry.
Figure 1. Various types of electrodes and their structures used for modification in sensing applications.Adapted with permission from ref. ( 36). Copyright 2019 Royal Society of Chemistry.Figure 2
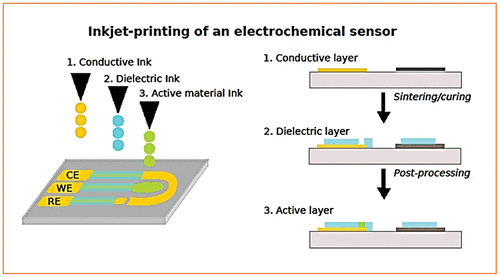 Figure 2. A schematic showing how inkjet printing is used to create electrochemical sensors. The transducers must be produced using a minimum of two inks, and frequently an additional ink is necessary to enhance sensor performance.Adapted with permission from ref. ( 70). Copyright 2017 Elsevier.
Figure 2. A schematic showing how inkjet printing is used to create electrochemical sensors. The transducers must be produced using a minimum of two inks, and frequently an additional ink is necessary to enhance sensor performance.Adapted with permission from ref. ( 70). Copyright 2017 Elsevier.Figure 3
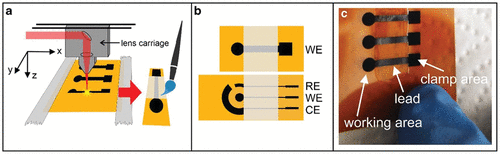 Figure 3. Electrode fabrication scheme (a), drawings of the electrode designs that were employed (WE working electrode, CE counter electrode, RE reference electrode), top=simple design, bottom=3-electrode design (b), photo of prepared LIG electrodes (c).Adapted from ref. ( 72). Copyright 2021 Springer publishers.
Figure 3. Electrode fabrication scheme (a), drawings of the electrode designs that were employed (WE working electrode, CE counter electrode, RE reference electrode), top=simple design, bottom=3-electrode design (b), photo of prepared LIG electrodes (c).Adapted from ref. ( 72). Copyright 2021 Springer publishers.References
CHAPTER SECTIONSThis chapter references 94 other publications.
- 1Killard A. J. Disposable sensors Current Opinion in Electrochemistry 2017 3 57 621Disposable sensorsKillard, Anthony J.Current Opinion in Electrochemistry (2017), 3 (1), 57-62CODEN: COEUCY; ISSN:2451-9111. (Elsevier B.V.)A review. Device disposability is a crit. feature of electrochem. sensor prodn., allowing complex measurements to be carried out in a low cost and efficient manner. Disposable sensors are increasing rapidly in their level of sophistication, supported by developments in materials and fabrication processing. This is, in turn, leading to greater levels of device integration and functionality and has the potential to significantly expand the no. of people who can use them, where they can be deployed and how they can be used. As well as becoming more complex, there is also significant emphasis in making such disposable devices more environmentally sustainable. Current themes in the development of disposable sensors are explored here.
- 2Beebe D. J. Denton D. D. Radwin R. G. Webster J. G. A silicon-based tactile sensor for finger-mounted applications IEEE Transactions on Biomedical Engineering 1998 45 151 1592A silicon-based tactile sensor for finger-mounted applicationsBeebe D J; Denton D D; Radwin R G; Webster J GIEEE transactions on bio-medical engineering (1998), 45 (2), 151-9 ISSN:0018-9294.This paper presents a silicon-based force sensor packaged in a flexible package and describes the sensors performance on human subjects. The sensing element consists of a circular silicon diaphragm (200-micron thick with a 2-mm radius) over a 10-micron sealed cavity with a solid Torlon dome providing force-to-pressure transduction to the diaphragm. Two dome heights (0.5 and 1.5 mm) were compared. The sensor with the taller dome showed improved sensitivity. Dynamic calibration and tracking experiments are performed with the sensor mounted on the dominant thumb of five human subjects. Both force and loading direction are statistically significant (P < 0.05). Subject variability accounted for 8.7% of the variance, while loading direction accounted for 1.9% of the variance. Average errors for the tracking experiment range from-2.8 to 1.0 N and are subject dependent. Three out of four subjects showed increasing negative error with increasing load.
- 3Han T. Nag A. Afsarimanesh N. Mukhopadhyay S. C. Kundu S. Xu Y. Laser-assisted printed flexible sensors: a review Sensors 2019 19 1462There is no corresponding record for this reference.
- 4Jung H.-C. Moon J.-H. Baek D.-H. Lee J.-H. Choi Y.-Y. Hong J.-S. Lee S.-H. CNT/PDMS composite flexible dry electrodesfor long-term ECG monitoring IEEE Transactions on Biomedical Engineering 2012 59 1472 14794CNT/PDMS composite flexible dry electrodes for long-term ECG monitoringJung Ha-Chul; Moon Jin-Hee; Baek Dong-Hyun; Lee Jae-Hee; Choi Yoon-Young; Hong Joung-Sook; Lee Sang-HoonIEEE transactions on bio-medical engineering (2012), 59 (5), 1472-9 ISSN:.We fabricated a carbon nanotube (CNT)/ polydimethylsiloxane (PDMS) composite-based dry ECG electrode that can be readily connected to conventional ECG devices, and showed its long-term wearable monitoring capability and robustness to motion and sweat. While the dispersion of CNTs in PDMS is challenging, we optimized the process to disperse untreated CNTs within PDMS by mechanical force only. The electrical and mechanical characteristics of the CNT/PDMS electrode were tested according to the concentration of CNTs and its thickness. The performances of ECG electrodes were evaluated by using 36 types of electrodes which were fabricated with different concentrations of CNTs, and with a differing diameter and thickness. The ECG signals were obtained by using electrodes of diverse sizes to observe the effects of motion and sweat, and the proposed electrode was shown to be robust to both factors. The CNT concentration and diameter of the electrodes were critical parameters in obtaining high-quality ECG signals. The electrode was shown to be biocompatible from the cytotoxicity test. A seven-day continuous wearability test showed that the quality of the ECG signal did not degrade over time, and skin reactions such as itching or erythema were not observed. This electrode could be used for the long-term measurement of other electrical biosignals for ubiquitous health monitoring including EMG, EEG, and ERG.
- 5El-Saftawy A. Elfalaky A. Ragheb M. Zakhary S. Electron beam induced surface modifications of PET film Radiation Physics and Chemistry 2014 102 96 1025Electron beam induced surface modifications of PET filmEl-Saftawy, A. A.; Elfalaky, A.; Ragheb, M. S.; Zakhary, S. G.Radiation Physics and Chemistry (2014), 102 (), 96-102CODEN: RPCHDM; ISSN:0969-806X. (Elsevier B. V.)The semi-cryst. poly (ethylene terephthalate) PET film was treated by low energy electrons with different fluences and the surface wettability modification is investigated. Contact angle technique is employed to prove PET surface wettability enhancement. The contact angle decreases due to alteration of surface chem., morphol. and free energy after electrons irradn. Fourier transform IR spectroscopy (FT-IR) shows the creation of polar groups on the surface which are responsible for surface wettability improvement. Atomic force microscope (AFM) observations reveal that the surface roughness changes after electrons-PET interactions. Surface free energy anal. has been obsd. as well. Low energy electrons irradn. of PET film improves and controls its wettability, adhesion and then biocompatibility.
- 6Yoo K.-P. Lim L.-T. Min N.-K. Lee M. J. Lee C. J. Park C.-W. Novel resistive-type humidity sensor based on multiwall carbon nanotube/polyimide composite films Sensors and Actuators B: Chemical 2010 145 120 1256Novel resistive-type humidity sensor based on multiwall carbon nanotube/polyimide composite filmsYoo, Kum-Pyo; Lim, Lee-Taek; Min, Nam-Ki; Lee, Myung Jin; Lee, Chul Jin; Park, Chan-WonSensors and Actuators, B: Chemical (2010), 145 (1), 120-125CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)A novel resistive-type relative humidity (RH) sensor based on plasma-treated multiwall carbon nanotube/polyimide (p-MWCNT/PI) composite films is reported. Details of the fabrication process and sensing characteristics such as linearity and sensitivity are described. The elec. resistance of the p-MWCNT/PI composite film shows typical concn. percolation behavior with increasing p-MWCNT loading. At loadings beyond the percolation threshold (0.05 wt%), this sensor exhibits very good linearity with a pos. slope over the entire operational RH range. The sensor has a humidity sensitivity of about 0.0047/% RH and a linearity correlation (R2) of 0.9999. A humidity sensing mechanism for the p-MWCNT/PI composite film is proposed on the basis of charge transfer between adsorbed water mols. and the p-MWCNTs.
- 7Karmakar R. S. Lu Y.-J. Fu Y. Wei K.-C. Chan S.-H. Wu M.-C. Lee J.-W. Lin T.-K. Wang J.-C. Cross-talk immunity of PEDOT: PSS pressure sensing arrays with gold nanoparticle incorporation Scientific reports 2017 7 1 107Bisphenol A Affects on the Functional Properties and Proteome of Testicular Germ Cells and Spermatogonial Stem Cells in vitro Culture ModelKarmakar, Polash Chandra; Kang, Hyun-Gu; Kim, Yong-Hee; Jung, Sang-Eun; Rahman, Md. Saidur; Lee, Hee-Seok; Kim, Young-Hyun; Pang, Myung-Geol; Ryu, Buom-YongScientific Reports (2017), 7 (1), 1-14CODEN: SRCEC3; ISSN:2045-2322. (Nature Research)The endocrine disruptor bisphenol A (BPA) is well known for its adverse effect on male fertility. Growing evidence suggests that BPA may interact with testicular germ cells and cause infertility as a result of its estrogenic activity. Objective of current in vitro study was to investigate the proliferation, survivability and stemness properties of mouse testicular germ cells exposed to BPA, and to evaluate possible expression of cellular proteome. Our results showed that germ cell viability and proliferation were not affected by low concns. (0.01, 0.1, 1, and 10μM) although significant redn. obsd. at 100μM BPA. Germ cell self-renewal and differentiation related marker proteins expression found unchanged at those concns. When BPA-exposed germ cells were transplanted into recipient testes, we obsd. fewer colonies at higher concns. (10 and 100μM). Addnl., a significant frequency of recombination failure during meiosis was obsd. in 10μM BPA-exposed germ cell transplanted recipient. Moreover, expt. on continuous BPA-exposed and 100μM BPA-recovered germ cells suggested that spermatogonial stem cells are more potential to survive in adverse environment. Finally, scrutinizing differentially expressed cellular proteins resulted from our proteomic anal., we conclude that BPA exposure might be assocd. with several health risks and infertility.
- 8Agarwal P. B. Alam B. Sharma D. S. Sharma S. Mandal S. Agarwal A. Flexible NO2 gas sensor based on single-walled carbon nanotubes on polytetrafluoroethylene substrates Flexible and Printed Electronics 2018 3 0350018Flexible NO2 gas sensor based on single-walled carbon nanotubes on polytetrafluoroethylene substratesAgarwal, Pankaj B.; Alam, Bayazeed; Sharma, Daya Shankar; Sharma, Sumit; Mandal, Soumen; Agarwal, AjayFlexible and Printed Electronics (2018), 3 (3), 035001/1-035001/8CODEN: FPELAB; ISSN:2058-8585. (IOP Publishing Ltd.)This paper presents a flexible and reliable chemiresistor-type NO2 gas sensor based on single-walled carbon nanotubes (SWNTs) on polytetrafluoroethylene (PTFE) membrane filter substrates. The sensor is realized by using a cost-effective spray coating in the prepn. of SWNTs thin film, followed by the fabrication of metal contacts using a shadow mask and polyethyleneimine (PEI) noncovalent functionalization of the SWNTs. This showed a high sensitivity to NO2 gas at room temp. in dry air; 21.58% to 167.7% for concns. of 0.75 ppm to 5 ppm, and was almost nonsensitive to ammonia. Gas sensing characterization results, obtained for various substrate bending/wrapping over different cylinders with diams. of 75 mm, 12.5 mm, and 6 mm showed that bending does not significantly affect sensitivity for NO2 concns. of 0.75 ppm to 2 ppm, while in the case of 3 ppm to 5 ppm NO2, the bent samples indicate enhanced sensitivity. This is probably because of the porous nature of PTFE substrates; these sensors were 1.5 to 2.7 times more sensitive than those fabricated over silicon substrate for 1 ppm and 5 ppm, resp. Moreover, the relative humidity of 10% and 30% significantly reduced the sensitivity of the sensors. The presented results could be useful for the future development of flexible electronics/sensors for monitoring outdoor air quality and for the detection of volatile org. compds.
- 9Rahimi R. Ochoa M. Ziaie B. Direct laser writing of porous-carbon/silver nanocomposite for flexible electronics ACS applied materials & interfaces 2016 8 16907 169139Direct Laser Writing of Porous-Carbon/Silver Nanocomposite for Flexible ElectronicsRahimi, Rahim; Ochoa, Manuel; Ziaie, BabakACS Applied Materials & Interfaces (2016), 8 (26), 16907-16913CODEN: AAMICK; ISSN:1944-8244. (American Chemical Society)In this research article, we demonstrate a facile method for the fabrication of porous-carbon/silver nanocomposites using direct laser writing on polymeric substrates. Our technique uses a combination of CO2 laser-induced carbonization and selective silver deposition on a polyimide sheet to create flexible highly conductive traces. The localized laser irradn. selectively converts the polyimide to a highly porous and conductive carbonized film with superhydrophilic wettability. The resulting pattern allows for selective trapping of aq. silver ionic ink solns. into the carbonized regions, which are converted to silver nanoparticle fillers upon an annealing step. Elemental and surface morphol. anal. via XRD and SEM reveals a uniform coating of Ag nanoparticles on the porous carbon. The Ag/C composite lowers the sheet resistance of the original laser carbonized polyimide from 50 to 0.02 Ω/.box.. The resulting patterns are flexible and electromechanically robust with less than 0.6 Ω variation in resistance after >15000 bending flexion cycles at a radius of curvature of 5 mm. Furthermore, using this technique, we demonstrate the fabrication of a wireless resonant pressure sensor capable of detecting pressures ranging from 0 to 97 kPa with an av. sensitivity of -26 kHz/kPa.
- 10Tasaltin C. Basarir F. Preparation of flexible VOC sensor based on carbon nanotubes and gold nanoparticles Sensors and Actuators B: Chemical 2014 194 173 17910Preparation of flexible VOC sensor based on carbon nanotubes and gold nanoparticlesTasaltin, Cihat; Basarir, FevzihanSensors and Actuators, B: Chemical (2014), 194 (), 173-179CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Novel flexible volatile org. compd. (VOC) sensor was prepd. by deposition of gold nanoparticle (AuNP) and amine-modified multi-walled carbon nanotubes (MWCNT-NH2) on polyimide substrate via electrospraying technique. First, interdigitated electrodes (IDE) were fabricated on polyimide (Kapton) substrate by photolithog. Then, the substrates were subjected to oxygen plasma etching, followed by electrospraying of AuNP and MWCNT-NH2 soln. on the substrate. The thin films were characterized by FE-SEM and AFM as well as elec. cond. measurement. Chem. sensing behaviors of the sensors were analyzed against polar (water, propanol and ethanol) and nonpolar (hexane, toluene, trichloroethylene and chloroform) VOCs. In addn., effect of the AuNP/MWCNT-NH2 ratio on the cond. and sensing was investigated. Bending test (100 times) resulted in negligible resistance change, demonstrating the successful prepn. of flexible sensor.
- 11Mayousse C. Celle C. Carella A. Simonato J.-P. Synthesis and purification of long copper nanowires. Application to high performance flexible transparent electrodes with and without PEDOT: PSS Nano Research 2014 7 315 32411Synthesis and purification of long copper nanowires. Application to high performance flexible transparent electrodes with and without PEDOT:PSSMayousse, Celine; Celle, Caroline; Carella, Alexandre; Simonato, Jean-PierreNano Research (2014), 7 (3), 315-324CODEN: NRAEB5; ISSN:1998-0000. (Springer GmbH)We demonstrate the hydrothermal synthesis of long copper nanowires based on a simple protocol. We show that the purifn. of the nanowires is very important and can be achieved easily by wet treatment with glacial acetic acid. Fabrication of random networks of purified copper nanowires leads to flexible transparent electrodes with excellent optoelectronic performances (e.g., 55 Ω/sq. at 94% transparency). The process is carried out at room temp. and no post-treatment is necessary. Hybrid materials with the conductive polymer PEDOT:PSS show similar properties (e.g., 46 Ω/sq. at 93% transparency), with improved mech. properties. Both electrodes were integrated in capacitive touch sensors.
- 12Jin H. Zhou J. Dong S. Feng B. Luo J. Wang D. Milne W. Yang C. Y. Deposition of c-axis orientation aluminum nitride films on flexible polymer substrates by reactive direct-current magnetron sputtering Thin Solid Films 2012 520 4863 487012Deposition of c-axis orientation aluminum nitride films on flexible polymer substrates by reactive direct-current magnetron sputteringJin, H.; Zhou, J.; Dong, S. R.; Feng, B.; Luo, J. K.; Wang, D. M.; Milne, W. I.; Yang, C. Y.Thin Solid Films (2012), 520 (15), 4863-4870CODEN: THSFAP; ISSN:0040-6090. (Elsevier B.V.)Aluminum nitride (AlN) piezoelec. thin films with c-axis crystal orientation on polymer substrates can potentially be used for development of flexible electronics and lab-on-chip systems. In this study, we investigated the effects of deposition parameters on the crystal structure of AlN thin films on polymer substrates deposited by reactive direct-current magnetron sputtering. The results show that low sputtering pressure as well as optimized N2/Ar flow ratio and sputtering power is beneficial for AlN (002) orientation and can produce a highly (002) oriented columnar structure on polymer substrates. High sputtering power and low N2/Ar flow ratio increase the deposition rate. In addn., the thickness of Al underlayer also has a strong influence on the film crystallog. The optimal deposition parameters in our expts. are: deposition pressure 0.38 Pa, N2/Ar flow ratio 2:3, sputtering power 414 W, and thickness of Al underlayer less than 100 nm.
- 13Nag A. Mitra A. Mukhopadhyay S. C. Graphene and its sensor-based applications: A review Sensors and Actuators A: Physical 2018 270 177 19413Graphene and its sensor-based applications: A reviewNag, Anindya; Mitra, Arkadeep; Mukhopadhyay, Subhas ChandraSensors and Actuators, A: Physical (2018), 270 (), 177-194CODEN: SAAPEB; ISSN:0924-4247. (Elsevier B.V.)This paper presents an overview of the work done on graphene in recent years. It explains the prepn. techniques, the properties of graphene related to its physio-chem. structure and some key applications. Graphene, due to its outstanding elec., mech. and thermal properties, has been one of the most popular choices to develop the electrodes of a sensor. It has been used in different forms including nanoparticle and oxide forms. Along with the prepn. and properties of graphene, the categorization of the applications has been done based on the type of sensors. Comparisons between different research studies for each type have been made to highlight their performances. The challenges faced by the current graphene-based sensors along with some of the probable solns. and their future opportunities are also briefly explained in this paper.
- 14Manjunatha J. Swamy B. K. Deraman M. Mamatha G. Simultaneous voltammetric measurement of ascorbic acid and dopamine at poly (vanillin) modified carbon paste electrode: A cyclic voltammetric study Pharma Chem 2012 4 2489 249714Simultaneous voltammetric measurement of ascorbic acid and dopamine at poly(vanillin) modified carbon paste electrode: a cyclic voltammetric studyManjunatha, J. G.; Swamy, B. E. Kumara; Deraman, M.; Mamatha, G. P.Pharma Chemica (2012), 4 (6), 2489-2497CODEN: PCHHAX; ISSN:0975-413X. (Scholars Research Library)Polymd. film of vanillin was prepd. on to the surface of carbon paste electrode in alk. soln. by cyclic voltammetric (CV) technique after pretreatment by H2SO4 media. The polyvanillin (VA) film-coated carbon paste electrode (CPE) exhibited excellent electrocatalytic activity toward the oxidn. of dopamine (DA) and ascorbic acid (AA) in 0.2M acetate buffer soln. at pH 7. This modified electrode exhibited a potent and persistent electron-mediating behavior followed by well-sepd. oxidn. peaks towards DA and AA. The modified electrode shows good sensitivity, selectivity and stability and was applied to the detn. of DA and AA.
- 15Manjunatha J. G. Surfactant modified carbon nanotube paste electrode for the sensitive determination of mitoxantrone anticancer drug Journal of Electrochemical Science and Engineering 2017 7 39 4915Surfactant modified carbon nanotube paste electrode for the sensitive determination of mitoxantrone anticancer drugManjunatha, Jamballi G.Journal of Electrochemical Science and Engineering (2017), 7 (1), 39-49CODEN: JESEGX; ISSN:1847-9286. (International Association of Physical Chemists)Surfactant modified carbon nanotube paste electrode is prepd. as electrochem. sensor with high sensitivity responding to mitoxantrone (MTX). Electrochem. oxidn. of MTX is investigated in buffered soln. by cyclic voltammetry that is found very sensi-tive method for detection of MTX. It is shown that the sodium dodecyl sulfate modified carbon nanotube paste electrode (SDSMCNTPE) gives enhanced current response for MTX compared to the bare carbon nanotube paste electrode (BCNTPE). Different parameters were tested to optimize the conditions for MTX detn. The effects of different surfactant and surfactant concn., pH, scan rate, and concn. of MTX on the oxidn. peak current values were detd. Excellent results were obtained by cyclic voltammetry using SDSMCNTPE, where two MTX oxidn. peaks appeared around 370 and 600 mV vs. Detailed anal. of the second voltammetric peak showed the linear dependence on concn. between 2×10-7 and 7×10-6 M MTX with the slope of the corelation coeff. of 0.99271. LOD and LOQ were detd. as 3.5 ×10-8 M and 11×10-8 M, resp. The SDSMCNTPE showed very good reproducibility, high stability in its voltammetric response, high electrochem. sensitivity and low detection limit for MTX.
- 16Hareesha N. Manjunatha J. A simple and low-cost poly (dl-phenylalanine) modified carbon sensor for the improved electrochemical analysis of Riboflavin Journal of Science: Advanced Materials and Devices 2020 5 502 51116A simple and low-cost poly (DL-phenylalanine) modified carbon sensor for the improved electrochemical analysis of RiboflavinHareesha, N.; Manjunatha, J. G.Journal of Science: Advanced Materials and Devices (2020), 5 (4), 502-511CODEN: JSAMCR; ISSN:2468-2179. (Elsevier B.V.)The current study reports on the electroanal. of Riboflavin (RF) on the surface of a poly DL-phenylalanine modified carbon paste sensor (PDLPAMCPS). The developed sensor caused a noticeable electrocatalytic action for the redox activity of RF in a phosphate buffer soln. (PBS) of pH 6.5. This anal. was evaluated using differential pulse voltammetry (DPV) and cyclic voltammetry (CV) techniques. The active surface of the functioning sensor exposes well developed anodic peak with prominent electron transfer. PDLPAMCPS shows distinctive and improved DPV and CV peaks for RF with and without the presence of org. dyes and dopamine (DA). The impact of the soln. pH, scan rate, accumulation potential and concn. on RF at PDLPAMCPS were studied. A vibrant linear growth was obsd. between the oxidn. peak current and the RF concn. in the range of 6.0μM-30.0μM, which provides a lower limit of detection (LOD) of 92 nM with excellent sensitivity. The proposed method and sensor were applied for the assessment of RF in medicine sample as an anal. application with fine recoveries and low LOD.
- 17Hareesha N. Manjunatha J. Raril C. Tigari G. Sensitive and selective electrochemical resolution of tyrosine with ascorbic acid through the development of electropolymerized alizarin sodium sulfonate modified carbon nanotube paste electrodes ChemistrySelect 2019 4 4559 456717Sensitive and Selective Electrochemical Resolution of Tyrosine with Ascorbic Acid through the Development of Electropolymerized Alizarin Sodium Sulfonate Modified Carbon Nanotube Paste ElectrodesHareesha, N.; Manjunatha, J. G.; Raril, C.; Tigari, GirishChemistrySelect (2019), 4 (15), 4559-4567CODEN: CHEMUD; ISSN:2365-6549. (Wiley-VCH Verlag GmbH & Co. KGaA)An approachable and a selective electrocatalytic investigation of tyrosine (TR) with ascorbic acid (AA) in phosphate buffer soln. (PBS) was done using a sensitive Poly Alizarin sodium sulfonate modified Carbon nanotube paste electrode (PASSMCNTPE). The morphol. and elemental studies on PASSMCNTPE and bare carbon nanotube paste electrode (BCNTPE) were resolute through field emission SEM (FESEM) and energy dispersive X-ray spectroscopy (EDX). The PASSMCNTPE shows excellent sensitivity, selectivity, stability, repeatability, and reproducibility for the electrochem. characterization of TR through cyclic voltammetry (CV) and differential pulse voltammetry (DPV). The prepd. sensor provides an elevated current response towards TR as compared to the BCNTPE. A plot of the anodic peak current of TR and the concn. of TR was obtained linearly in the range 1.4×10-4 M to 2×10-4 M with the limit of detection (LOD) 7.8×10-7 M and the limit of quantification (LOQ) 26×10-7 M resp. The equipped sensor was also helpful for the quant. and qual. anal. of TR moiety in a pharmaceutical drug as a real sample.
- 18Hareesha N. Manjunatha J. G. Surfactant and polymer layered carbon composite electrochemical sensor for the analysis of estriol with ciprofloxacin Materials Research Innovations 2020 24 349 36218Surfactant and polymer layered carbon composite electrochemical senor for the analysis of estriol with ciprofloxacinHareesha, Nagarajappa; Manjunatha, Jamballi G.Materials Research Innovations (2020), 24 (6), 349-362CODEN: MRINFV; ISSN:1433-075X. (Taylor & Francis Ltd.)This article exposes an approachable strategy for the responsive electrochem. anal. of estriol (ER) hormone by designing a sensitive and selective sensor of sodium dodecyl sulfate and polymerised xanthacridinum modified carbon nanotube and graphite composite paste electrode (SDS-PXAMCNTGCPE) via linear sweep voltammetry (LSV), cyclic voltammetry (CV) and differential pulse voltammetry (DPV) techniques. ER exposed a fine resolved irreversibly oxidised peak in phosphate buffer soln. (PBS) of pH 7.0. CV and DPV reveal a commendable linear relation between oxidative peak current of ER and concn. of ER in the range 2 UM to 200 UM and 10 UM to 70 UM and they provide a promising lowest limit of detection (LOD) values of 2.9 × 10-7 M and 1.9 × 10-7 M. CV and DPV techniques were also applied to examine the selectivity of ER with Ciprofloxacin (CF). The adapting capability of SDS-PXAMCNTGCPE was accountable for the lower level identification of ER in sewage water as a real sample. The proposed sensor needs an easy prepn. method with low cost and it exhibits superior sensitivity, stability, repeatability, and reproducibility.
- 19Manjunatha J. A surfactant enhanced graphene paste electrode as an effective electrochemical sensor for the sensitive and simultaneous determination of catechol and resorcinol Chemical Data Collections 2020 25 10033119A surfactant enhanced graphene paste electrode as an effective electrochemical sensor for the sensitive and simultaneous determination of catechol and resorcinolManjunatha, J. G.Chemical Data Collections (2020), 25 (), 100331CODEN: CDCHA8; ISSN:2405-8300. (Elsevier B.V.)A highly sensitive and selective electrochem. sensor based on surfactant modified graphene paste electrode was designed for the detn. of catechol (CC) and resorcinol (RS). The developed electrodes were characterized by cyclic voltammetry (CV), differential pulse voltammetry (DPV), and Field emission SEM (FE-SEM). Under the ideal conditions, CV and DPV studies revealed that, compared with bare graphene paste electrode (BGPE), sodium dodecyl sulfate modified graphene paste electrode (SDSMGPE) demonstrated an increase in the efficiency of the electrocatalytic oxidn. of CC. Under the optimized condition the peak current for CC showed a good linear relationship with concn. in the range 2.0 x 10-6 to 1.0 x 10-5 and 1.5 x 10-5 to 1.5 x 10-4 M, and the detection limit was 106 nM (S/N = 3). The CV results show that CC and RS could be detected simultaneously using SDSMGPE with peak-to-peak sepn. of 0.728 V for CC-RS. The sensor is very promising for CC sensing attributing to its high sensitivity, fast response, excellent stability, and good reproducibility. The SDSMGPE sensor utilized for detection of CC in real sample with a satisfactory result.
- 20Manjunatha J. G. Poly (Adenine) modified graphene-based voltammetric sensor for the electrochemical determination of catechol, hydroquinone and resorcinol The Open Chemical Engineering Journal 2020 14There is no corresponding record for this reference.
- 21Prinith N. S. Manjunatha J. G. Polymethionine modified carbon nanotube sensor for sensitive and selective determination of L-tryptophan Journal of Electrochemical Science and Engineering 2020 10 305 31521Polymethionine modified carbon nanotube sensor for sensitive and selective determination of L-tryptophanPrinith, Nambudumada S.; Manjunatha, Jamballi G.Journal of Electrochemical Science and Engineering (2020), 10 (4), 305-315CODEN: JESEGX; ISSN:1847-9286. (International Association of Physical Chemists)The electrochem. initiated catalytic oxidn. of amino acid L-tryptophan (L-TPN) in phosphate buffer soln. has been scrutinized using highly conductive polymethionine modified carbon nanotube paste sensor (PMETCNTPS) through cyclic voltammetry (CV) technique. Compared to the bare carbon nanotube paste sensor (BCNTPS), PMETCNTPS exhibited a quantifiable current signal by CV method. PMETCNTPS was found sensitive to LTPN concns. within the linear segment of detection range 1.5 - 8.0x10-5 M. By employing the calibration plot, the detection limit was detd. as 6.99x10-7 M. In addn., PMETCNTPS was successfully exploited and validated in detg. L-TPN in the pharmaceutical supplement.
- 22Hareesha N. Manjunatha J. Amrutha B. Pushpanjali P. Charithra M. Prinith Subbaiah N. Electrochemical analysis of indigo carmine in food and water samples using a poly (glutamic acid) layered multi-walled carbon nanotube paste electrode Journal of Electronic Materials 2021 50 1230 123822Electrochemical Analysis of Indigo Carmine in Food and Water Samples Using a Poly(Glutamic Acid) Layered Multi-walled Carbon Nanotube Paste ElectrodeHareesha, N.; Manjunatha, J. G.; Amrutha, B. M.; Pushpanjali, P. A.; Charithra, M. M.; Prinith Subbaiah, N.Journal of Electronic Materials (2021), 50 (3), 1230-1238CODEN: JECMA5; ISSN:0361-5235. (Springer)Some colorants are hazardous to living species; hence, a powerful and fast methodol. is required for the anal. of those colorants in food and water samples. A modest electrochem. polymerised glutamic acid layered multi-walled carbon nanotube paste electrode [P(GA)LMWCNTPE] was functionalised for the sensing of indigo carmine (IC) by powerful differential pulse voltammetry (DPV) and cyclic voltammetry (CV) approaches. Within the optimized exptl. conditions, the P(GA)LMWCNTPE holds an acceptable and high rate of electro-catalytic activity towards the redox behavior of IC. The projected P(GA)LMWCNTPE shows a decent selectivity for IC in the presence of methyl orange. The modified sensor shows an acceptable linear growth between oxidative peak current and concn. in both CV and DPV methods with fine limit of detection values of 4.2μM and 0.36μM, resp. Addnl., the developed sensor was effectively applied to detect IC in food and water samples. The morphol. and surface activities of the modified and unmodified electrodes were detd. through field emission SEM, electrochem. impedance spectroscopy, and CV techniques. The P(GA)LMWCNTPE requires a simple prepn. procedure and is low-cost, with acceptable storage stability, sensitivity, and reproducibility.
- 23Monnappa B. Manjunatha J. G. Bhatt A. S. Chenthattil R. Ananda P. Electrochemical sensor for the determination of alizarin red-s at non-ionic surfactant modified carbon nanotube paste electrode Physical Chemistry Research 2019 7 523 533There is no corresponding record for this reference.
- 24Raril C. Manjunatha J. G. Sensitive electrochemical analysis of resorcinol using polymer modified carbon paste electrode: a cyclic voltammetric study Anal. Bioanal. Electrochem 2018 10 488 49824Sensitive electrochemical analysis of resorcinol using polymer modified carbon paste electrode: a cyclic voltammetric studyRaril, Chenthattil; Manjunatha, Jamballi G.Analytical & Bioanalytical Electrochemistry (2018), 10 (4), 488-498CODEN: ABENC2; ISSN:2008-4226. (University of Tehran, Center of Excellence in Electrochemistry)Glycine was electropolymd. on a carbon paste electrode (CPE) to form the polymer film in the electrooxidn. process of the amino and carboxylic group contg. compd. by cyclic voltammetric technique and the electrochem. oxidn. of resorcinol (RC) was studied in 0.2 M phosphate buffer soln. (PBS) having pH 7.0. Modified carbon paste electrode (MCPE) shows a good electrocatalytic activity towards RC compared to bare carbon paste electrode (BCPE). The oxidn. of RC at MCPE showed a linear relation vs. concn. of RC in the range of 6×10-5 to 1×10-3 M with a detection limit 8.6×10-6 M.
- 25Charithra M. M. Manjunatha J. G. G. Raril C. Surfactant modified graphite paste electrode as an electrochemical sensor for the enhanced voltammetric detection of estriol with dopamine and uric acid Advanced Pharmaceutical Bulletin 2020 10 24725Surfactant modified graphite paste electrode as an electrochemical sensor for the enhanced voltammetric detection of estriol with dopamine and uric acidCharithra, Madikeri Manjunath; Manjunatha, Jamballi Gangadharappa Gowda; Raril, ChenthattilAdvanced Pharmaceutical Bulletin (2020), 10 (2), 247-253CODEN: APBDCL; ISSN:2251-7308. (Tabriz University of Medical Sciences)Purpose: Estriol (ERL) is a type of hormone among the groups of estrogen hormone that was detected through the voltammetric technique by constructing an electrochem. sensor based on the octoxynol-9 modified graphite paste electrode (OXL-9MGPE). Methods: Using the strategy of cyclic voltammetry (CV) and differential pulse voltammetry (DPV) with a bare graphite paste electrode (BGPE) immobilized with OXL-9, ERL electro-oxidn. has been assessed in 0.2 M phosphate buffer soln. (PBS) of pH 6.0. The fabricated electrode has substantial electrochem. sensing efficiency, and the ERL oxidn. at the OXL-9MGPE was the irreversible process. The surface morphol. characteristics of BGPE and OXL-9MGPE were differentiated with the help of field emission SEM (FE-SEM). Results: The impact of various factors such as scan rate, pH, reproducibility, repeatability, and stability on the electro-oxidn. of ERL was evaluated. Techniques of CV and DPV were utilized to det. ERL, dopamine (DAN), and uric acid (URA) simultaneously with the projected sensor. The peak current was varied with ERL concn. in the range from 4x10-5 to 1.2x10-4 M at OXL-9MGPE. From this, the detection limit 1.4x10-6 M and limit of quantification (LOQ) 4.7x10-6 M have been attained. Conclusion: As a result, OXL-9MGPE was successfully achieved as an electrochem. detector for the electro anal. of ERL via the CV technique.
- 26Manjunatha J. G. Electrochemical polymerised graphene paste electrode and application to catechol sensing The Open Chemical Engineering Journal 2019 13There is no corresponding record for this reference.
- 27Prinith N. S. Manjunatha J. G. Raril C. Electrocatalytic analysis of dopamine, uric acid and ascorbic acid at poly (adenine) modified carbon nanotube paste electrode: a cyclic voltammetric study Anal. Bioanal., Electrochem 2019 11 742 75627Electrocatalytic analysis of dopamine, uric acid and ascorbic acid at poly(adenine) modified carbon nanotube paste electrode: a cyclic voltammetric studyPrinith, Nambudumada S.; Manjunatha, Jamballi G.; Raril, ChenthattilAnalytical & Bioanalytical Electrochemistry (2019), 11 (6), 742-756CODEN: ABENC2; ISSN:2008-4226. (University of Tehran, Center of Excellence in Electrochemistry)In this work, Poly (adenine) film modified carbon nanotube paste electrode (PAEMCNTPE), prepd. by electropolymn. of adenine on a carbon nanotube paste electrode, applied for the sensitive and selective anal. of dopamine (DA), Uric acid (UA) and Ascorbic acid (AA) based on cyclic voltammetric technique. The polyadenine modified carbon nanotube paste electrode has presented with an impressive electrocatalytic activity towards oxidn. of DA, UA, and AA in the presence of supporting electrolyte, 0.2 M PBS pH 6.5. The surface of fabricated sensor displayed with a permselective layer with transducing, anti- interfering and anti-fouling properties followed by solving the overlapped peaks of DA, AA and UA obtained from the conventional electrode by presenting with distinct three anodic peaks. The result of this electrochem. sensor displayed with excellent sensitivity, selectivity, reproducibility, repeatability and highly stable. The detection limit for DA was 6.7×10-7 M. Further; this fabricated electrode was carried out for the quantification of DA in the real sample.
- 28Manjunatha J. G. Swamy B. E. K. Mamatha G. P. Gilbert O. Sherigara B. S. Poly (maleic acid) modified carbon paste electrode for simultaneous detection of dopamine in the presence of uric acid: a cyclic voltammetric study ANALYTICAL & BIOANALYTICAL ELECTROCHEMISTRY 2011 3 146 159There is no corresponding record for this reference.
- 29Liu C. Huang N. Xu F. Tong J. Chen Z. Gui X. Fu Y. Lao C. 3D printing technologies for flexible tactile sensors toward wearable electronics and electronic skin Polymers 2018 10 629293D printing technologies for flexible tactile sensors toward wearable electronics and electronic skinLiu, Changyong; Huang, Ninggui; Xu, Feng; Tong, Junda; Chen, Zhangwei; Gui, Xuchun; Fu, Yuelong; Lao, ChangshiPolymers (Basel, Switzerland) (2018), 10 (6), 629/1-629/31CODEN: POLYCK; ISSN:2073-4360. (MDPI AG)3D printing has attracted a lot of attention in recent years. Over the past three decades, various 3D printing technologies have been developed including photopolymn.-based, materials extrusion-based, sheet lamination-based, binder jetting-based, power bed fusion-based and direct energy deposition-based processes. 3D printing offers unparalleled flexibility and simplicity in the fabrication of highly complex 3D objects. Tactile sensors that emulate human tactile perceptions are used to translate mech. signals such as force, pressure, strain, shear, torsion, bend, vibration, etc. into elec. signals and play a crucial role toward the realization of wearable electronics and electronic skin. To date, many types of 3D printing technologies have been applied in the manufg. of various types of tactile sensors including piezoresistive, capacitive and piezoelec. sensors. This review attempts to summarize the current state-of-the-art 3D printing technologies and their applications in tactile sensors for wearable electronics and electronic skin. The applications are categorized into five aspects: 3D-printed molds for microstructuring substrate, electrodes and sensing element; 3D-printed flexible sensor substrate and sensor body for tactile sensors; 3D-printed sensing element; 3D-printed flexible and stretchable electrodes for tactile sensors; and fully 3D-printed tactile sensors. Latest advances in the fabrication of tactile sensors by 3D printing are reviewed and the advantages and limitations of various 3D printing technologies and printable materials are discussed. Finally, future development of 3D-printed tactile sensors is discussed.
- 30Kundu S. Hussain M. Kumar V. Kumar S. Das A. K. Direct metal laser sintering of TiN reinforced Ti6Al4V alloy based metal matrix composite: Fabrication and characterization The International Journal of Advanced Manufacturing Technology 2018 97 2635 2646There is no corresponding record for this reference.
- 31Siegel A. C. Phillips S. T. Dickey M. D. Lu N. Suo Z. Whitesides G. M. Foldable printed circuit boards on paper substrates Advanced Functional Materials 2010 20 28 3531Foldable Printed Circuit Boards on Paper SubstratesSiegel, Adam C.; Phillips, Scott T.; Dickey, Michael D.; Lu, Nanshu; Suo, Zhigang; Whitesides, George M.Advanced Functional Materials (2010), 20 (1), 28-35CODEN: AFMDC6; ISSN:1616-301X. (Wiley-VCH Verlag GmbH & Co. KGaA)This paper describes several low-cost methods for fabricating flexible electronic circuits on paper. The circuits comprise (i) metallic wires (e.g., tin or zinc) that are deposited on the substrate by evapn., sputtering, or airbrushing, and (ii) discrete surface-mountable electronic components that are fastened with conductive adhesive directly to the wires. These electronic circuits-like conventional printed circuit boards-can be produced with electronic components that connect on both sides of the substrate. Unlike printed circuit boards made from fiberglass, ceramics, or polyimides, however, paper can be folded and creased (repeatedly), shaped to form three-dimensional structures, trimmed using scissors, used to wick fluids (e.g., for microfluidic applications) and disposed of by incineration. Paper-based electronic circuits are thin and lightwt.; they should be useful for applications in consumer electronics and packaging, for disposable systems for uses in the military and homeland security, for applications in medical sensing or low-cost portable diagnostics, for paper-based microelectromech. systems, and for applications involving textiles.
- 32Zittel H. Miller F. A Glassy-Carbon Electrode for Voltammetry Analytical Chemistry 1965 37 200 20332A glassy-carbon electrode for voltammetryZittel, H. E.; Miller, F. J.(1965), 37 (2), 200-3CODEN: ANCHAM; ISSN:0003-2700.A piece of glassy C, which is a gas-impermeable, elec.-conductive, chem.-resistant material, was cut from a small boat and sealed into an epoxy resin rod, having a central tube to permit Hg contact, and was used as indicator electrode in a system contg. a pyrolytic graphite counter electrode and a S.C.E. reference electrode. The app. was useful in voltammetric studies from + 1.2 to -0.8 v. vs. S.C.E. with acidic solns. of Ce+++, Ce4+, Cr6+, Fe++, Hg+, Fe(CN)64-, Ag+, Cu++, and UO2++, and results compared favorably with those obtained with other solid electrodes with respect to ease of fabrication and maintenance.
- 33Taylor R. Humffray A. Electrochemical studies on glassy carbon electrodes: I. Electron transfer kinetics Journal of electroanalytical chemistry and interfacial electrochemistry 1973 42 347 35433Electrochemical studies on glassy carbon electrodes. I. Electron transfer kineticsTaylor, R. J.; Humffray, A. A.Journal of Electroanalytical Chemistry and Interfacial Electrochemistry (1973), 42 (3), 347-54CODEN: JEIEBC; ISSN:0022-0728.Rate const. ks measurements for charge transfer processes at various electrodes show the general sequence ks(Pt) > ks (glassy C) > ks (was-impregnated graphite) > ks (C paste). Max. rates of glassy C electrodes were obsd. after treatment with chromic acid, except for the redn. of IO3- ion, where simple polishing of the electrode surface produces the highest rate. Time effects on rate consts. are also discussed.
- 34Uskoković V. A historical review of glassy carbon: Synthesis, structure, properties and applications Carbon Trends 2021 5 100116There is no corresponding record for this reference.
- 35Švancara I. Vytřas K. Kalcher K. Walcarius A. Wang J. Carbon paste electrodes in facts, numbers, and notes: a review on the occasion of the 50‐years jubilee of carbon paste in electrochemistry and electroanalysis Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2009 21 7 2835Carbon paste electrodes in facts, numbers, and notes: a review on the occasion of the 50-years jubilee of carbon paste in electrochemistry and electroanalysisSvancara, Ivan; Vytras, Karel; Kalcher, Kurt; Walcarius, Alain; Wang, JosephElectroanalysis (2009), 21 (1), 7-28CODEN: ELANEU; ISSN:1040-0397. (Wiley-VCH Verlag GmbH & Co. KGaA)A review. This article reviews the electrochem. and electroanal. applications of carbon paste-based electrodes, sensors, and detectors on the occasion of the half-of-century anniversary since the discovery of carbon paste. The review (with 333 refs.) has been prepd. in the form of a retrospective compilation presenting the field by means of various facts, notes, data, surveys, and summaries, including numerous rarities or curiosities that illustrate the individual achievements and milestones. Carbon paste-based electrodes are discussed in their entirety by covering all important areas of the field, starting from basic characterization of carbon paste as the electrode material, via its typical physicochem. and electrochem. properties or specific features, up to a representative documentation of their applicability in electrochem. and electroanal. measurements.
- 36P. S. Adarakatti, S. K. Kempahanumakkagari , Modified electrodes for sensing, in: Electrochemistry , 2018, pp. 58-95.There is no corresponding record for this reference.
- 37Banks C. E. Moore R. R. Davies T. J. Compton R. G. Investigation of modified basal plane pyrolytic graphite electrodes: definitive evidence for the electrocatalytic properties of the ends of carbon nanotubes Chemical Communications 2004 1804 180537Investigation of modified basal plane pyrolytic graphite electrodes: definitive evidence for the electrocatalytic properties of the ends of carbon nanotubesBanks, Craig E.; Moore, Ryan R.; Davies, Trevor J.; Compton, Richard G.Chemical Communications (Cambridge, United Kingdom) (2004), (16), 1804-1805CODEN: CHCOFS; ISSN:1359-7345. (Royal Society of Chemistry)The basis of the electrocatalytic nature of multi-wall C nanotubes is suggested to reside in electron transfer from the ends of nanotubes, which structurally resemble the behavior of edge plane (as opposed to basal plane) graphite, and is demonstrated via the comparison of the electrochem. oxidn. of epinephrine and the electrochem. redn. of ferricyanide at nanotube-modified electrodes using different types of graphite electrodes and with C60-modified electrodes.
- 38Banks C. E. Compton R. G. Edge plane pyrolytic graphite electrodes in electroanalysis: an overview Analytical Sciences 2005 21 1263 126838Edge plane pyrolytic graphite electrodes in electroanalysis: An overviewBanks, Craig E.; Compton, Richard G.Analytical Sciences (2005), 21 (11), 1263-1268CODEN: ANSCEN; ISSN:0910-6340. (Japan Society for Analytical Chemistry)A review. The recent development, behavior and scope of edge plane pyrolytic graphite electrodes in electroanal. are overviewed. Similarities to, and advantages, over multi-walled CNT modified electrodes are noted and the wide scope of applications, ranging through gas sensing, stripping voltammetry and biosensing, illustrated.
- 39Adarakatti P. S. Foster C. W. Banks C. E. NS A. K. Malingappa P. Calixarene bulk modified screen-printed electrodes (SPCCEs) as a one-shot disposable sensor for the simultaneous detection of lead (II), copper (II) and mercury (II) ions: Application to environmental samples Sensors and Actuators A: Physical 2017 267 517 525There is no corresponding record for this reference.
- 40Saidur M. Aziz A. A. Basirun W. Recent advances in DNA-based electrochemical biosensors for heavy metal ion detection: A review Biosensors and Bioelectronics 2017 90 125 13940Recent advances in DNA-based electrochemical biosensors for heavy metal ion detection: A reviewSaidur, M. R.; Abdul Aziz, A. R.; Basirun, W. J.Biosensors & Bioelectronics (2017), 90 (), 125-139CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)The presence of heavy metal in food chains due to the rapid industrialization poses a serious threat on the environment. Therefore, detection and monitoring of heavy metals contamination are gaining more attention nowadays. However, the current anal. methods (based on spectroscopy) for the detection of heavy metal contamination are often very expensive, tedious and can only be handled by trained personnel. DNA biosensors, which are based on electrochem. transduction, is a sensitive but inexpensive method of detection. The principles, sensitivity, selectivity and challenges of electrochem. biosensors are discussed in this review. This review also highlights the major advances of DNA-based electrochem. biosensors for the detection of heavy metal ions such as Hg2+, Ag+, Cu2+ and Pb2+.
- 41Apilux A. Dungchai W. Siangproh W. Praphairaksit N. Henry C. S. Chailapakul O. Lab-on-paper with dual electrochemical/colorimetric detection for simultaneous determination of gold and iron Analytical chemistry 2010 82 1727 173241Lab-on-Paper with Dual Electrochemical/Colorimetric Detection for Simultaneous Determination of Gold and IronApilux, Amara; Dungchai, Wijitar; Siangproh, Weena; Praphairaksit, Narong; Henry, Charles S.; Chailapakul, OrawonAnalytical Chemistry (Washington, DC, United States) (2010), 82 (5), 1727-1732CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)A novel lab-on-paper device combining electrochem. and colorimetric detection for the rapid screening of Au(III) in the presence of a common interference, Fe(III), in industrial waste solns. is presented here. With dil. aqua regia (0.1M HCl + 0.05M HNO3) as the supporting electrolyte, square wave voltammetry on paper provided a well-defined redn. peak for Au(III) at ∼287 ± 12 mV vs. Ag/AgCl. Under the optimized working conditions, the calibration curve showed good linearity in the concn. range of 1-200 ppm of Au(III) with a correlation coeff. of 0.997. The limit of detection (LOD) of the proposed method is 1 ppm. Interferences from various cations were also studied. Fe(III) is the only metal that affects the electrochem. detn. of Au(III) when present above a 2.5-fold excess concn. of that of the Au(III). To overcome this limitation, a colorimetric method was used to simultaneously detect Fe(III) as a screening tool. The procedure was then successfully applied to det. Au(III) in Au-refining waste solns. The results are in agreement with those obtained from inductively coupled plasma-at. emission spectrometry (ICP-AES).
- 42Li X. Tian J. Garnier G. Shen W. Fabrication of paper-based microfluidic sensors by printing Colloids and surfaces B: Biointerfaces 2010 76 564 57042Fabrication of paper-based microfluidic sensors by printingLi, Xu; Tian, Junfei; Garnier, Gil; Shen, WeiColloids and Surfaces, B: Biointerfaces (2010), 76 (2), 564-570CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)A novel method for the fabrication of paper-based microfluidic diagnostic devices is reported; it consists of selectively hydrophobizing paper using cellulose reactive hydrophobization agents. The hydrophilic-hydrophobic contrast of patterns so created has excellent ability to control capillary penetration of aq. liqs. in paper channels. Incorporating this idea with digital ink jet printing techniques, a new fabrication method of paper-based microfluidic devices is established. Ink jet printing can deliver biomols. and indicator reagents with precision into the microfluidic patterns to form bio-chem. sensing zones within the device. This method thus allows the complete sensor, i.e. channel patterns and the detecting chemistries, to be fabricated only by two printing steps. This fabrication method can be scaled up and adapted to use high speed, high vol. and low cost com. printing technol. Sensors can be fabricated for specific tests, or they can be made as general devices to perform on-demand quant. anal. tasks by incorporating the required detection chemistries for the required tasks.
- 43Lu Y. Shi W. Qin J. Lin B. Fabrication and characterization of paper-based microfluidics prepared in nitrocellulose membrane by wax printing Analytical chemistry 2010 82 329 33543Fabrication and Characterization of Paper-Based Microfluidics Prepared in Nitrocellulose Membrane By Wax PrintingLu, Yao; Shi, Weiwei; Qin, Jianhua; Lin, BingchengAnalytical Chemistry (Washington, DC, United States) (2010), 82 (1), 329-335CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)Paper-based microfluidics is a promising technol. to develop a simple, low-cost, portable, and disposable diagnostic platform for resource-limited settings. Here the authors report the fabrication of paper-based microfluidic devices in nitrocellulose membrane by wax printing for protein immobilization related applications. The fabrication process, which can be finished within 10 min, includes mainly printing and baking steps. Wax patterning will form hydrophobic regions in the membrane, which can be used to direct the flow path or sep. reaction zones. The fabrication parameters like printing mode and baking time were optimized, and performances of the wax-patterned nitrocellulose membrane such as printing resoln., protein immobilization, and sample purifn. capabilities were also characterized in this report. The authors believe the wax-patterned nitrocellulose membrane will enhance the capabilities of paper microfluidic devices and bring new applications in this field.
- 44Lu Y. Lin B. Qin J. Patterned paper as a low-cost, flexible substrate for rapid prototyping of PDMS microdevices via “liquid molding” Analytical chemistry 2011 83 1830 183544Patterned Paper as a Low-Cost, Flexible Substrate for Rapid Prototyping of PDMS Microdevices via "Liquid Molding"Lu, Yao; Lin, Bingcheng; Qin, JianhuaAnalytical Chemistry (Washington, DC, United States) (2011), 83 (5), 1830-1835CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)This report describes the use of patterned paper as a low-cost, flexible substrate for rapidly prototyping PDMS microdevices via "liq. molding". The entire fabrication process consists simply of three steps: (1) fabrication of patterned paper in NC membrane by direct wax printing (or modified wax printing that we call "transfer wax printing"); (2) formation of liq. mold on wax-patterned NC membrane; (3) PDMS molding and curing on wax-patterned NC membrane anchored with liq. micropatterns. All these procedures can be finished within only 1.5 h without the use of a photomask, photoresist, UV lamp, etc. Through the use of wax-patterned NC membrane coupled with a liq. mold as a template, different PDMS microdevices such as microwells and microchannels have been fabricated to demonstrate the usefulness of the method for PDMS microfabrication. The height of microwells and microchannels can also be tailored flexibly by adjusting the liq. filling vol. This method for prototyping PDMS microdevices has some favorable merits including simple operation procedures, fast concept-to-device time, and low cost, indicating its potential for simple PDMS microdevice fabrication and applications.
- 45Arena A. Donato N. Saitta G. Bonavita A. Rizzo G. Neri G. Flexible ethanol sensors on glossy paper substrates operating at room temperature Sensors and Actuators B: Chemical 2010 145 488 49445Flexible ethanol sensors on glossy paper substrates operating at room temperatureArena, A.; Donato, N.; Saitta, G.; Bonavita, A.; Rizzo, G.; Neri, G.Sensors and Actuators, B: Chemical (2010), 145 (1), 488-494CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Flexible sensors for the detection of ethanol at room temp. have been developed by depositing sensitive layers consisting of ITO nanopowder dispersed into poly-diallyldimethylammonium chloride (PDDAC), on the top of interdigitated electrodes based on conductive carbon nanotubes, previously printed onto flexible glossy paper substrates. The morphol. and the microstructure of the interdigitated electrodes together with org.-inorg. sensing layer have been investigated by means of SEM, XRD and FT-IR. The elec. behavior of the developed sensors at room temp. in response to low ethanol concns. has been discussed as object of our research. The good ethanol sensing properties exhibited at room temp. by the composite film was supposed to originate from the electrostatic interaction occurring between the pos. charged PDDAC polyelectrolyte and neg. charged ITO particles.
- 46Dungchai W. Chailapakul O. Henry C. S. A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printing Analyst 2011 136 77 8246A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printingDungchai, Wijitar; Chailapakul, Orawon; Henry, Charles S.Analyst (Cambridge, United Kingdom) (2011), 136 (1), 77-82CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Wax screen-printing as a low-cost, simple, and rapid method for fabricating paper-based microfluidic devices (μPADs) is reported here. Solid wax was rubbed through a screen onto paper filters. The printed wax was then melted into the paper to form hydrophobic barriers using only a hot plate. The authors first studied the relationship between the width of a hydrophobic barrier and the width of the original design line. The authors also optimized the heating temp. and time and detd. the resoln. of structures fabricated using this technique. The min. width of hydrophilic channel and hydrophobic barrier is 650 and 1300 μm, resp. Next, the authors fabrication method was compared to a photolithog. method using the reaction between bicinchoninic acid (BCA) and Cu1+ to demonstrate differences in background reactivity. Photolithog. defined channels exhibited a high background while wax printed channels showed a very low background. Finally, the utility of wax screen-printing was demonstrated for the simultaneous detn. of glucose and total iron in control human serum samples using an electrochem. method with glucose oxidase and a colorimetric method with 1,10-phenanthroline. This study demonstrates that wax screen-printing is an easy-to-use and inexpensive alternative fabrication method for μPAD, which will be esp. useful in developing countries.
- 47Dungchai W. Chailapakul O. Henry C. S. Electrochemical detection for paper-based microfluidics Analytical chemistry 2009 81 5821 582647Electrochemical Detection for Paper-Based MicrofluidicsDungchai, Wijitar; Chailapakul, Orawon; Henry, Charles S.Analytical Chemistry (Washington, DC, United States) (2009), 81 (14), 5821-5826CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)The authors report the first demonstration of electrochem. detection for paper-based microfluidic devices. Photolithog. was used to make microfluidic channels on filter paper, and screen-printing technol. was used to fabricate electrodes on the paper-based microfluidic devices. Screen-printed electrodes on paper were characterized using cyclic voltammetry to demonstrate the basic electrochem. performance of the system. The utility of the authors' devices was then demonstrated with the detn. of glucose, lactate, and uric acid in biol. samples using oxidase enzyme (glucose oxidase, lactate oxidase, and uricase, resp.) reactions. Oxidase enzyme reactions produce H2O2 while decompg. their resp. substrates, and therefore a single electrode type is needed for detection of multiple species. Selectivity of the working electrode for H2O2 was improved using Prussian Blue as a redox mediator. The detn. of glucose, lactate, and uric acid in control serum samples was performed using chronoamperometry at the optimal detection potential for H2O2 (0 V vs. the on-chip Ag/AgCl ref. electrode). Levels of glucose and lactate in control serum samples measured using the paper devices were 4.9±0.6 and 1.2±0.2 mM (level I control sample), and 16.3±0.7 and 3.2±0.3 mM (level II control sample), resp., and were within error of the values measured using traditional tests. This study shows the successful integration of paper-based microfluidics and electrochem. detection as an easy-to-use, inexpensive, and portable alternative for point of care monitoring.
- 48Suresh R. R. Lakshmanakumar M. Arockia Jayalatha J. Rajan K. Sethuraman S. Krishnan U. M. Rayappan J. B. B. Fabrication of screen-printed electrodes: opportunities and challenges Journal of Materials Science 2021 56 8951 900648Fabrication of screen-printed electrodes: opportunities and challengesSuresh, Raghavv Raghavender; Lakshmanakumar, Muthaiyan; Arockia Jayalatha, J. B. B.; Rajan, K. S.; Sethuraman, Swaminathan; Krishnan, Uma Maheswari; Rayappan, John Bosco BalaguruJournal of Materials Science (2021), 56 (15), 8951-9006CODEN: JMTSAS; ISSN:0022-2461. (Springer)Abstr.: In recent times, wearable electronics, real-time sensing devices and point-of-care device applications have seen a surge in demand. This demand inherently calls for a more economic and reliable method of mass prodn. One such robust technique is screen printing that offers advantages in terms of being versatile, economical and easy to use. This technique has been proclaimed to be the best amongst the other additive manufg. techniques by many research groups. This can be solely attributed to its simple equipment design, requiring a paste for printing purpose, a meshwork for housing the design and a squeegee to carry out printing through an up-and-down motion. This subsequently calls for optimizing the parameters such as ink rheol., pore size of the mesh, proper choice of mesh design and motion of the squeeze in order to fabricate electrodes for desired purpose. Due to this technique's immense potential to open up broad inroads in the domain of flexible electronics, whose concept has radically redefined the perception towards the field of electronics, a review article encompassing an elaborate description on the science and other quintessential parameters involved in this mass printing technique would prove to be extremely useful. In this purview, the review article is compartmentalised into sections encompassing discussion on the manufg. of different kinds of inks for screen printing applications, substrates used, electrode design and pre-treatment procedures. Graphical abstr.: [graphic not available: see fulltext].
- 49Clark L. C. Jr Lyons C. Electrode systems for continuous monitoring in cardiovascular surgery Annals of the New York Academy of sciences 1962 102 29 4549Electrode systems for continuous monitoring in cardiovascular surgeryClark, Leland C., Jr.; Lyons, ChampAnnals of the New York Academy of Sciences (1962), 102 (Art. 1), 29-45CODEN: ANYAA9; ISSN:0077-8923.App. is described for the continuous monitoring of blood chemistry in surgical patients. External electrode systems record quant. pH, O, and CO2; intravascular electrode systems record H and ascorbate indicator diln. curves. 21 references.
- 50Bachan Upadhyay L. S. Verma N. Enzyme inhibition based biosensors: A review Analytical Letters 2013 46 225 241There is no corresponding record for this reference.
- 51Abdelwahab A. A. Won M. S. Shim Y. B. Direct electrochemistry of cholesterol oxidase immobilized on a conducting polymer: Application for a cholesterol biosensor Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2010 22 21 25There is no corresponding record for this reference.
- 52Premanode B. Toumazou C. A novel, low power biosensor for real time monitoring of creatinine and urea in peritoneal dialysis Sensors and Actuators B: Chemical 2007 120 732 73552A novel, low power biosensor for real time monitoring of creatinine and urea in peritoneal dialysisPremanode, Bhusana; Toumazou, ChrisSensors and Actuators, B: Chemical (2007), 120 (2), 732-735CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Novel biosensors, based on immobilized creatininase, creatinase and urease are developed, using ISFETs with weak inversion at pH 6-8 and 37.0 °C. The ISFETs with circuitry, demonstrate a linear relationship of urea and creatinine at the range of 0-200 and 0-20 mM, resp. Preliminary results show that biosensors operating in weak inversion mode can eliminate many of the disadvantages of ISFETs operating in the strong inversion region, providing a wide dynamic range output in nanoAmp. Such characteristics fit the anal. requirements for improving real-time monitoring in peritoneal dialysis (PD). Further work covers stability of ISFET sensors biased with CMOS circuits in the weak inversion mode working in the room temp. of 15-40 °C.
- 53Zourob M. Simonian A. Wild J. Mohr S. Fan X. Abdulhalim I. Goddard N. Optical leaky waveguide biosensors for the detection of organophosphorus pesticides Analyst 2007 132 114 12053Optical leaky waveguide biosensors for the detection of organophosphorus pesticidesZourob, M.; Simonian, A.; Wild, J.; Mohr, S.; Fan, Xudong; Abdulhalim, I.; Goddard, N. J.Analyst (Cambridge, United Kingdom) (2007), 132 (2), 114-120CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Organophosphorus (OP) pesticides can be rapidly detected by integrating organophosphorus hydrolase with an optical leaky waveguide biosensor. This enzyme catalyzes the hydrolysis of a wide range of organophosphorus compds. causing an increase in the pH. Thus, the direct detection of OP is possible by monitoring of the pH changes assocd. with the enzyme's activity. The use of an optical, leaky waveguide clad with absorbing materials for the detection of OP pesticides by measuring changes in refractive index, absorbance and fluorescence was described. In the most effective configuration, a thick sensing layer was used to increase the amt. of immobilized enzyme and to increase the light interaction with the sensing layer, resulting in a greatly enhanced sensitivity. The platforms developed in this work were successfully used to detect paraoxon and parathion down to 4 nM concns.
- 54Zhu H. Li L. Zhou W. Shao Z. Chen X. Advances in non-enzymatic glucose sensors based on metal oxides Journal of Materials Chemistry B 2016 4 7333 734954Advances in non-enzymatic glucose sensors based on metal oxidesZhu, Hua; Li, Li; Zhou, Wei; Shao, Zongping; Chen, XianjianJournal of Materials Chemistry B: Materials for Biology and Medicine (2016), 4 (46), 7333-7349CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)A review. Glucose sensors have been extensively developed because of their broad applications, esp. in diabetes diagnosis. Up to date, electrochem. enzymic glucose sensors are commonly used in daily life for glucose detection and com. successful as glucose-meters because they exhibit excellent selectivity, high reliability, and could be handled under physiol. pH conditions. However, considering some intrinsic disadvantages of enzymes, such as high fabrication cost and poor stability, non-enzymic glucose sensors have attracted increasing research interest in recent years due to their low cost, high stability, prompt response, and low detection limit. Furthermore, the development of nanotechnol. has also offered new opportunities to construct nanostructured electrodes for glucose sensing applications. With distinguished advantages, metal oxides have garnered extensive effort in the development of cost-effective sensors with high stability, sensitivity and quick response for the detn. of glucose via electrochem. oxidn. Hence, this review summarizes the advances in non-enzymic glucose sensors based on different metal oxides (such as ZnO, CuO/Cu2O, NiO, Co3O4, MnO2, etc.) and their nanocomposites. Addnl., a brief prospective is presented on metal oxides for glucose sensors.
- 55Raziq A. Tariq M. Hussian R. Mehmood M. H. Khan M. S. Hassan A. Electrochemical investigation of glucose oxidation on a glassy carbon electrode using voltammetric, amperometric, and digital simulation methods ChemistrySelect 2017 2 9711 971755Electrochemical Investigation of Glucose Oxidation on a Glassy Carbon Electrode Using Voltammetric, Amperometric, and Digital Simulation MethodsRaziq, Abdul; Tariq, Muhammad; Hussian, Riaz; Mehmood, Muhammad Haris; Khan, Muhammad Saleem; Hassan, AyazChemistrySelect (2017), 2 (30), 9711-9717CODEN: CHEMUD; ISSN:2365-6549. (Wiley-VCH Verlag GmbH & Co. KGaA)In this paper, we report the use of glassy carbon electrode for electrochem. oxidn. of glucose in basic medium. The effect of glucose concn., scan rate, and point potential was studied. Heterogeneous electron transfer parameters such as std. electron transfer rate const. (k0), transfer coeff. (a), and E0 were calcd. using cyclic voltammetry (CV), semi integration of cyclic voltammetry (SICV), semi differentiation of cyclic voltammetry (SDCV), and digital simulation techniques. Amperometric expts. were performed for detn. of glucose concn. in basic media. These expts. show a broad linear concn. range, up to 11 mM with a sensitivity of 927.96μAmM-1cm-2. The lower detection limit (LOD) and Lower Quantification limit (LOQ) were found to be 50μM and 0.5 mM, resp. Effect of interfering species such as uric acid, ascorbic acid, acetaminophen, and chloride ion on the electrocatalytic behavior of glassy carbon electrode (GCE) was also studied.
- 56Pletcher D. Electrocatalysis: present and future Journal of applied electrochemistry 1984 14 403 41556Electrocatalysis: present and futurePletcher, DerekJournal of Applied Electrochemistry (1984), 14 (4), 403-15CODEN: JAELBJ; ISSN:0021-891X.A review with 61 refs.
- 57Zhu H. Li L. Zhou W. Shao Z. Chen X. Correction: Advances in non-enzymatic glucose sensors based on metal oxides Journal of Materials Chemistry B 2017 5 1117 111757Correction: Advances in non-enzymatic glucose sensors based on metal oxides [Erratum to document cited in CA165:577563]Zhu, Hua; Li, Li; Zhou, Wei; Shao, Zongping; Chen, XiangjianJournal of Materials Chemistry B: Materials for Biology and Medicine (2017), 5 (5), 1117CODEN: JMCBDV; ISSN:2050-7518. (Royal Society of Chemistry)A review. The name of Xiangjian Chen was spelled incorrectly. There is also a correction for the acknowledgments.
- 58Burke L. Premonolayer oxidation and its role in electrocatalysis Electrochimica Acta 1994 39 1841 184858Premonolayer oxidation and its role in electrocatalysisBurke, L. D.Electrochimica Acta (1994), 39 (11-12), 1841-8CODEN: ELCAAV; ISSN:0013-4686.A discussion with 70 refs. is given. Hydrous oxides are considered here as an integral component of noble metal electrochem.; they are regarded as quite distinct from, and in certain areas more important than, monolayer oxide deposits. The usual polycryst. metal surface is assumed to be composed of two major components: (i) atoms of high lattice co-ordination no. (the vast majority) than oxidize to yield monolayer oxide species, and (ii) atoms of quite low lattice coordination no. (and of very low coverage) that oxidize to yield incipient hydrous oxide species. Evidence, that is now quite strong in the case of gold, supporting this view is summarized. Data are provided to support the view that in many instances electrocatalytic processes at noble metal electrode/aq. soln. interfaces involve hydrous oxides as mediators and inhibitors in oxidn. and redn. processes, resp. Premonolayer oxidn. of metals, in terms of this approach, is considered as an interesting, relevant and challenging area of research.
- 59Warsinke A. Benkert A. Scheller F. Electrochemical immunoassays Fresenius’ journal of analytical chemistry 2000 366 622 63459Electrochemical immunoassaysWarsinke, A.; Benkert, A.; Scheller, F. W.Fresenius' Journal of Analytical Chemistry (2000), 366 (6-7), 622-634CODEN: FJACES; ISSN:0937-0633. (Springer-Verlag)A review with 105 refs. Immunoassays (IA) use the specific antigen-antibody complexation for anal. purposes. RIAs, fluorescence immunoassays (FIA) and enzyme immunoassays (EIA) are well established in clin. diagnostics. For the development of hand-held devices which can be used for point of care measurements, electrochem. immunoassays are promising alternatives to existing immunochem. tests. Moreover, for opaque or optically dense matrixes electrochem. methods are superior. Potentiometric, capacitive and amperometric transducers have been applied for direct and indirect electrochem. immunoassays. However, due to their fast detection, broad linear range and low detection limit, amperometric transducers are preferred. Competitive and noncompetitive amperometric immunoassays have been developed with redox compds. or enzymes as labels. This review gives an overview of the most frequently applied principles in electrochem. immunoassays. The potential of an indirect competitive amperometric immunoassay for the detn. of creatinine within nanomolar range and the circumvention of the most serious problem in electrochem. immunoassays, namely regeneration, is discussed.
- 60Volpe G. Fares G. Delli Quadri F. Draisci R. Ferretti G. Marchiafava C. Moscone D. Palleschi G. A disposable immunosensor for detection of 17β-estradiol in non-extracted bovine serum Analytica chimica acta 2006 572 11 1660A disposable immunosensor for detection of 17β-estradiol in non-extracted bovine serumVolpe, G.; Fares, G.; delli Quadri, F.; Draisci, R.; Ferretti, G.; Marchiafava, C.; Moscone, D.; Palleschi, G.Analytica Chimica Acta (2006), 572 (1), 11-16CODEN: ACACAM; ISSN:0003-2670. (Elsevier B.V.)This paper reports the assembly of a disposable immunosensor based on the direct competitive ELISA, for simple and fast measurement of 17β-estradiol (17β-E2) in bovine serum, using screen-printed electrodes (SPEs) and a Palm-Sens portable electrochem. detector. The immunosensor strip was assembled immobilizing, by passive adsorption, anti-rabbit IgG onto the surface of the working SPE electrode. After the interaction between anti-rabbit IgG and rabbit anti-17β-E2 polyclonal antibodies (PAb), the competition was performed using 17β-estradiol-alk. phosphatase conjugate (17β-E2-AP) synthesized in our lab. The enzymic substrate used for signal generation was 1-naphthylphosphate and its conversion to an electroactive product (1-naphthol) was measured using differential pulse voltammetry (DPV). To develop a prototype for field measurements, the entire competitive protocol has been optimized directly in a blank non-extd. bovine serum. According to the new EU criteria established by the Commission Decision 2002/657/EC for qual. and quant. screening methods, the detection capability (CCβ), was detd. The CCβ value resulted below the action limit (40 pg mL-1) fixed for 17β-E2. Spiked and real samples were analyzed using the electrochem. immunostrips obtaining precision values (relative std. deviation, R.S.D.%) ranging from 8.6 to 17.0% and a recovery (R%) from 88.2 to 120.0%. Results obtained on real samples were confirmed by liq. chromatog. coupled online with tandem mass spectrometry (LC-MS/MS) using an atm. pressure chem. ionization (APCI) source and a heated nebulizer (HN) interface; this is the method currently used to confirm illegal hormone administration for regulatory purposes. The disposable immunosensor appears suitable as a screening tool for field anal. of bovine serum estradiol.
- 61Bagel O. Degrand C. Limoges B. Joannes M. Azek F. Brossier P. Enzyme affinity assays involving a single‐use electrochemical sensor. Applications to the enzyme immunoassay of human chorionic gonadotropin hormone and nucleic acid hybridization of human cytomegalovirus DNA Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2000 12 1447 1452There is no corresponding record for this reference.
- 62Carpini G. Lucarelli F. Marrazza G. Mascini M. Oligonucleotide-modified screen-printed gold electrodes for enzyme-amplified sensing of nucleic acids Biosensors and Bioelectronics 2004 20 167 17562Oligonucleotide-modified screen-printed gold electrodes for enzyme-amplified sensing of nucleic acidsCarpini, Guido; Lucarelli, Fausto; Marrazza, Giovanna; Mascini, MarcoBiosensors & Bioelectronics (2004), 20 (2), 167-175CODEN: BBIOE4; ISSN:0956-5663. (Elsevier)An electrochem. genosensor for the detection of specific sequences of DNA has been developed using disposable screen-printed gold electrodes. Screen-printed gold electrodes were firstly modified with a mixed monolayer of a 25-mer thiol-tethered DNA probe and a spacer thiol, 6-mercapto-1-hexanol (MCH). The DNA probe sequence was internal to the sequence of the 35S promoter, which sequence is inserted in the genome of GMOs regulating the transgene expression. An enzyme-amplified detection scheme, based on the coupling of a streptavidin-alk. phosphatase conjugate and biotinylated target sequences was then applied. The enzyme catalyzed the hydrolysis of the electroinactive α-naphthyl phosphate to α-naphthol; this product is electroactive and has been detected by means of differential pulse voltammetry. The assay was, firstly, characterized using synthetic oligonucleotides. Relevant parameters, such as the probe concn. and the immobilization time, the use of the MCH and different enzymic conjugates, were investigated and optimized. The genosensor response was found to be linearly related to the target concn. between 0 and 25 nmol/L; the detection limit was 0.25 nmol/L. The anal. procedure was then applied for the detection of the 35S promoter sequence, which was amplified from the pBI121 plasmid by polymerase chain reaction (PCR). Hybridization conditions (i.e., hybridization buffer and hybridization time) were further optimized. The selectivity of the assay was confirmed using biotinylated non-complementary amplicons and PCR blanks. The results showed that the genosensor enabled sensitive (detection limit: 1 nmol/L) and specific detection of GMO-related sequences, thus providing a useful tool for the screening anal. of bioengineered food samples.
- 63Rao V. K. Sharma M. K. Goel A. K. Singh L. Sekhar K. Amperometric immunosensor for the detection of Vibrio cholerae O1 using disposable screen-printed electrodes Analytical sciences 2006 22 1207 121163Amperometric immunosensor for the detection of Vibrio cholerae O1 using disposable screen-printed electrodesRao, Vepa K.; Sharma, Mukesh K.; Goei, Ajay K.; Singh, Lokendra; Sekhar, KrishnamurthiAnalytical Sciences (2006), 22 (9), 1207-1211CODEN: ANSCEN; ISSN:0910-6340. (Japan Society for Analytical Chemistry)A disposable amperometric immunosensor was studied for the rapid detection of Vibrio cholerae (V. cholerae), the causative agent of cholera, employing an indirect sandwich enzyme linked immunosorbent assay (ELISA) principle. Screen-printed electrodes (SPEs) were fabricated (by using com. and homemade carbon inks), electrochem. characterized and the assay conditions were optimized for capturing antibodies and antigen. Whole cell lysate (WCL) of V. cholerae was used to raise antibodies in rabbits and mice. The antibodies raised against WCL of V. cholerae were found to be specific, and no cross reactivity was obsd. with other enteric bacteria. 1-Naphthyl phosphate was used as a substrate with the amperometric detection of its enzymic hydrolysis product 1-naphthol at a potential of +400 mV vs. Ag/AgCl ref. electrode. A comparison between the amperometric detection technique and the std. ELISA was made in terms of the total assay time, the amt. of biol. materials used and the sensitivity of detection. The min. detection limit of the amperometric immunosensor for V. cholerae was found to be 105 cells/mL in 55 min, while ELISA detected 106 cells/mL in 4 h.
- 64Keay R. McNeil C. Separation-free electrochemical immunosensor for rapid determination of atrazine Biosensors and Bioelectronics 1998 13 963 97064Separation-free electrochemical immunosensor for rapid determination of atrazineKeay, R. W.; McNeil, C. J.Biosensors & Bioelectronics (1998), 13 (9), 963-970CODEN: BBIOE4; ISSN:0956-5663. (Elsevier Science Ltd.)A sepn.-free electrochem. immunoassay method for the detection of the pesticide atrazine is described. The method is a competitive ELISA incorporating disposable screen printed horseradish peroxidase modified electrodes as the detector element in conjunction with single-use atrazine immuno-membranes. Screen printed C electrodes were prepd. using C ink incorporating horseradish peroxidase. A monoclonal antibody for atrazine was immobilized onto Biodyne C membranes which were, in turn, placed over the electrode surface. The assay was based on competition for available binding sites between free atrazine and an atrazine-glucose oxidase conjugate prepd. inhouse. In the presence of glucose, H2O2 formed by the conjugate was reduced by enzyme-channeling via the HRP electrode. The HRP was in turn re-reduced by a direct electron transfer mechanism at a potential of +50 mV vs. Ag/AgCl. Any H2O2 formed in the bulk soln. by unbound atrazine-GOD conjugate was scavenged by excess catalase thus removing the requirement for a washing step. The performance of the method was compared with a com. immunoassay kit for atrazine.
- 65Audrey S. Beatriz P.-S. Jean-Louis M. Biosensors for pesticide detection: new trends American Journal of Analytical Chemistry 2012 2012There is no corresponding record for this reference.
- 66Susmel S. Guilbault G. O’sullivan C. Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodes Biosensors and Bioelectronics 2003 18 881 88966Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodesSusmel, S.; Guilbault, G. G.; O'Sullivan, C. K.Biosensors & Bioelectronics (2003), 18 (7), 881-889CODEN: BBIOE4; ISSN:0956-5663. (Elsevier Science Ltd.)The demonstration of a labeless immunosensor for the detection of pathogenic bacteria using screen printed gold electrodes (SPGEs) and a potassium hexacyanoferrate(II) redox probe is reported. Gold electrodes were produced using screen printing and the gold surfaces were modified by a thiol based self assembled monolayer (SAM) to facilitate antibody immobilization. SAMs based on the use of thioctic acid (TA), mercaptopropionic acid (MPA) and mercaptoundecanoic acid (MUA) were evaluated. Following antibody immobilization via the optimum SAM, the redox behavior and diffusion co-efficient (D) of the potassium hexacyanoferrate(II) probe was monitored in the absence and presence of analyte. In the presence of analyte, a change in the apparent diffusion co-efficient of the redox probe was obsd., attributable to impedance of the diffusion of redox electrons to the electrode surface due to the formation of the antibody-bacteria immunocomplex. No change in the diffusion co-efficient was obsd. when a non-specific antibody (mouse IgG) was immobilized and antigen added. The system has been demonstrated with Listeria monocytogenes and Bacillus cereus.
- 67Simon D. Ware T. Marcotte R. Lund B. R. Smith D. W. Di Prima M. Rennaker R. L. Voit W. A comparison of polymer substrates for photolithographic processing of flexible bioelectronics Biomedical microdevices 2013 15 925 93967A comparison of polymer substrates for photolithographic processing of flexible bioelectronicsSimon, Dustin; Ware, Taylor; Marcotte, Ryan; Lund, Benjamin R.; Smith, Dennis W., Jr.; Prima, Matthew; Rennaker, Robert L.; Voit, WalterBiomedical Microdevices (2013), 15 (6), 925-939CODEN: BMICFC; ISSN:1387-2176. (Springer)Flexible bioelectronics encompass a new generation of sensing devices, in which controlled interactions with tissue enhance understanding of biol. processes in vivo. However, the fabrication of such thin film electronics with photolithog. processes remains a challenge for many biocompatible polymers. Recently, two shape memory polymer (SMP) systems, based on acrylate and thiol-ene/acrylate networks, were designed as substrates for softening neural interfaces with glass transitions above body temp. (37 °C) such that the materials are stiff for insertion into soft tissue and soften through low moisture absorption in physiol. conditions. These two substrates, acrylate and thiol-ene/acrylate SMPs, are compared to polyethylene naphthalate, polycarbonate, polyimide, and polydimethylsiloxane, which have been widely used in flexible electronics research and industry. These six substrates are compared via dynamic mech. anal. (DMA), thermogravimetric anal. (TGA), and swelling studies. The integrity of gold and chromium/gold thin films on SMP substrates are evaluated with optical profilometry and elec. measurements as a function of processing temp. above, below and through the glass transition temp. The effects of crosslink d., adhesion and cure stress are shown to play a crit. role in the stability of these thin film materials, and a guide for the future design of responsive polymeric materials suitable for neural interfaces is proposed. Finally, neural interfaces fabricated on thiol-ene/acrylate substrates demonstrate long-term fidelity through both in vitro impedance spectroscopy and the recording of driven local field potentials for 8 wk in the auditory cortex of lab. rats.
- 68Yin Z. Huang Y. Bu N. Wang X. Xiong Y. Inkjet printing for flexible electronics: Materials, processes and equipments Chinese Science Bulletin 2010 55 3383 3407There is no corresponding record for this reference.
- 69Sele C. W. Von Werne T. Friend R. H. Sirringhaus H. Lithography‐free, self‐aligned inkjet printing with sub‐hundred‐nanometer resolution Advanced Materials 2005 17 997 100169Lithography-free, self-aligned inkjet printing with sub-hundred-nanometer resolutionSele, Christoph W.; von Werne, Timothy; Friend, Richard H.; Sirringhaus, HenningAdvanced Materials (Weinheim, Germany) (2005), 17 (8), 997-1001CODEN: ADVMEW; ISSN:0935-9648. (Wiley-VCH Verlag GmbH & Co. KGaA)Direct inkjet printing of sub-100 nm nanoscale gaps, without the need for any high-resoln. lithog., is made possible by a novel self-aligned printing method (see Figure). The benefits of this method are demonstrated by fabrication of all-polymer transistor circuits, which exhibit switching speeds at least two orders of magnitude higher than those previously reported for inkjet-printed polymer devices.
- 70Moya A. Gabriel G. Villa R. del Campo F. J. Inkjet-printed electrochemical sensors Current Opinion in Electrochemistry 2017 3 29 3970Inkjet-printed electrochemical sensorsMoya, Ana; Gabriel, Gemma; Villa, Rosa; Javier del Campo, F.Current Opinion in Electrochemistry (2017), 3 (1), 29-39CODEN: COEUCY; ISSN:2451-9111. (Elsevier B.V.)A review. Inkjet printing is a digital and non-contact printing technique that is beginning to be used in the manuf. of electronic devices, including electrochem. sensors. This article covers the main works featuring inkjet printing for the prodn. of electrochem. sensors, and focuses esp. on the works reported since 2015. The main factor limiting the application of inkjet printing in the manuf. of advanced devices stems mainly from the strict rheol. conditions that the ink needs to meet. This is the reason why inkjet is best used in combination with other manufg. techniques. Given their high degree of maturity and accessibility, home/office printers will likely lead a future wave in the manuf. of electronic devices, particularly at the development and prototyping stages.
- 71Khan M. S. Fon D. Li X. Tian J. Forsythe J. Garnier G. Shen W. Biosurface engineering through ink jet printing Colloids and Surfaces B: Biointerfaces 2010 75 441 44771Biosurface engineering through ink jet printingKhan, Mohidus Samad; Fon, Deniece; Li, Xu; Tian, Junfei; Forsythe, John; Garnier, Gil; Shen, WeiColloids and Surfaces, B: Biointerfaces (2010), 75 (2), 441-447CODEN: CSBBEQ; ISSN:0927-7765. (Elsevier B.V.)The feasibility of thermal ink jet printing as a robust process for biosurface engineering was demonstrated. The strategy investigated was to reconstruct a com. printer and take advantage of its color management interface. High printing resoln. was achieved by formulating bio-inks of viscosity and surface tension similar to those of com. inks. Protein and enzyme denaturation during thermal ink jet printing was shown to be insignificant. This is because the time spent by the biomols. in the heating zone of the printer is negligible; in addn., the air and substrate of high heat capacity absorb any residual heat from the droplet. Gradients of trophic/tropic factors can serve as driving force for cell growth or migration for tissue regeneration. Concn. gradients of proteins were printed on scaffolds to show the capability of ink jet printing. The printed proteins did not desorb upon prolonged immersion in aq. solns., thus allowing printed scaffold to be used under in vitro and in vivo conditions. The authors' group portrait was ink jet printed with a protein on paper, illustrating that complex biopatterns can be printed on large area. Finally, patterns of enzymes were ink jet printed within the detection and reaction zones of a paper diagnostic.
- 72Behrent A. Griesche C. Sippel P. Baeumner A. J. Process-property correlations in laser-induced graphene electrodes for electrochemical sensing Microchimica Acta 2021 188 1 14There is no corresponding record for this reference.
- 73Cuartero M. Crespo G. A. Bakker E. based thin-layer coulometric sensor for halide determination Analytical chemistry 2015 87 1981 199073Paper-Based Thin-Layer Coulometric Sensor for Halide DeterminationCuartero, Maria; Crespo, Gaston A.; Bakker, EricAnalytical Chemistry (Washington, DC, United States) (2015), 87 (3), 1981-1990CODEN: ANCHAM; ISSN:0003-2700. (American Chemical Society)The authors report on a paper-based anal. device (PAD) for the exhaustive, and therefore abs., detn. of halides in a range of diverse water samples and food supplements. A mixt. of chloride, bromide, and iodide ions is assessed in a wide range of concns., specifically, from 10-4.8 to 0.1M for bromide and iodide and from 10-4.5 to 0.6M for chloride, with a limit of detection of 10-5 M. As a result of a careful optimization of the electrochem. cell, a thin layer made of cellulose paper (75-μm thickness), a cation-exchange Donnan exclusion membrane (FKL), and a silver-foil working electrode were selected as optimum materials. Cyclic voltammetry (from 0 to 0.8 V) was chosen as the interrogation technique to impose the exhaustive oxidative plating and re-redn. of halides on the silver element, accompanied by outward and inward counterion fluxes. The scan rate plays an important role in the ability of the technique to resolve mixts. of ions. Moderate scan rates (10 mV s-1) provide a suitable compromise between sensitivity, limit of detection, and resoln. This paper-based microfluidic device is extremely simple in terms of manipulation, cost, and contamination risk. Paper is an excellent basis for the establishment of a confined thin aq. layer, the construction of disposable halide sensors, and portability for measuring outside the controlled lab. environment. A discussion of the relevant anal. characteristics is presented herein, followed by a demonstration of halide assessment in water samples (sea, tap, river, and mineral waters) and food supplements enriched with iodide and chloride as early examples.
- 74Lee S. H. Lee J. H. Tran V.-K. Ko E. Park C. H. Chung W. S. Seong G. H. Determination of acetaminophen using functional paper-based electrochemical devices Sensors and Actuators B: Chemical 2016 232 514 52274Determination of acetaminophen using functional paper-based electrochemical devicesLee, Sung Hwan; Lee, Joo Heon; Tran, Van-Khue; Ko, Euna; Park, Chan Ho; Chung, Woo Sung; Seong, Gi HunSensors and Actuators, B: Chemical (2016), 232 (), 514-522CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)In the present study, we constructed a functional paper fluidic device and evaluated its electrochem. performance by analyzing acetaminophen in the presence of ascorbic acid. The device was composed of a single-walled carbon nanotube (SWCNT) electrode and nafion-modified nitrocellulose membrane. Neg.-charged nafion was employed to build up a more neg. charge on the nitrocellulose membrane, and gold nanoparticles and polyglutamic acid (AuNP-PGA) were deposited on the SWCNT electrode to enhance the electrochem. performance of the device. The device had a vertical flow format in which the sample soln. flowed vertically through the paper. Using the nafion-modified nitrocellulose membrane and AuNP-PGA/SWCNT film electrode as a component of the paper fluidic device, we obtained a distinguishable acetaminophen oxidn. peak which was distinct from the ascorbic acid oxidn. peak. The acetaminophen oxidn. peak had a linear response with acetaminophen concn., varying from 50 μM to 300 μM (r2 = 0.992), which was broader than the std. drug dose range. The device exhibited a sensitivity of 13.3 mA/M and a detection limit of 15.0 μM. The device was stable with a relative std. deviation of 3.3% (up to 2 wk), and the reproducibility was 1.2-5.2%. Furthermore, the fabricated device accurately measured the amt. of acetaminophen in pharmaceutical samples.
- 75Gutiérrez-Capitán M. Baldi A. Fernández-Sánchez C. Electrochemical paper-based biosensor devices for rapid detection of biomarkers Sensors 2020 20 96775Electrochemical paper-based biosensor devices for rapid detection of biomarkersGutierrez-Capitan, Manuel; Baldi, Antonio; Fernandez-Sanchez, CesarSensors (2020), 20 (4), 967CODEN: SENSC9; ISSN:1424-8220. (MDPI AG)A review. In healthcare, new diagnostic tools that help in the diagnosis, prognosis, and monitoring of diseases rapidly and accurately are in high demand. For in-situ measurement of disease or infection biomarkers, point-of-care devices provide a dramatic speed advantage over conventional techniques, thus aiding clinicians in decision-making. During the last decade, paper-based anal. devices, combining paper substrates and electrochem. detection components, have emerged as important point-of-need diagnostic tools. This review highlights significant works on this topic over the last five years, from 2015 to 2019. The most relevant articles published in 2018 and 2019 are examd. in detail, focusing on device fabrication techniques and materials applied to the prodn. of paper fluidic and electrochem. cell architectures as well as on the final device assembly. Two main approaches were identified, that are, on one hand, those ones where the fabrication of the electrochem. cell is done on the paper substrate, where the fluidic structures are also defined, and, on the other hand, the fabrication of those ones where the electrochem. cell and liq.-driving paper component are defined on different substrates and then heterogeneously assembled. The main limitations of the current technologies are outlined and an outlook on the current technol. status and future prospects is given.
- 76Devarakonda S. Singh R. Bhardwaj J. Jang J. Cost-effective and handmade paper-based immunosensing device for electrochemical detection of influenza virus Sensors 2017 17 259776Cost-effective and handmade paper-based immunosensing device for electrochemical detection of influenza virusDevarakonda, Sivaranjani; Singh, Renu; Bhardwaj, Jyoti; Jang, JaesungSensors (2017), 17 (11), 2597/1-2597/13CODEN: SENSC9; ISSN:1424-8220. (MDPI AG)Here, we present a cost-effective, handmade paper-based immunosensor for label-free electrochem. detection of influenza virus H1N1. This immunosensor was prepd. by modifying paper with a spray of hydrophobic silica nanoparticles, and using stencil-printed electrodes. We used a glass vaporizer to spray the hydrophobic silica nanoparticles onto the paper, rendering it super-hydrophobic. The super-hydrophobicity, which is essential for this paper-based biosensor, was achieved via 30-40 spray coatings, corresponding to a 0.39-0.41 mg cm-2 coating of nanoparticles on the paper and yielding a water contact angle of 150° ± 1°. Stencil-printed carbon electrodes modified with single-walled carbon nanotubes and chitosan were employed to increase the sensitivity of the sensor, and the antibodies were immobilized via glutaraldehyde crosslinking. Differential pulse voltammetry was used to assess the sensitivity of the sensors at various virus concns., ranging from 10 to 104 PFU mL-1, and the selectivity was assessed against MS2 bacteriophages and the influenza B viruses. These immunosensors showed good linear behaviors, improved detection times (30 min), and selectivity for the H1N1 virus with a limit of detection of 113 PFU mL-1, which is sufficiently sensitive for rapid on-site diagnosis. The simple and inexpensive methodologies developed in this study have great potential to be used for the development of a low-cost and disposable immunosensor for detection of pathogenic microorganisms, esp. in developing countries.
- 77Sarkar P. Tothill I. E. Setford S. J. Turner A. P. Screen-printed amperometric biosensors for the rapid measurement of L-and D-amino acids Analyst 1999 124 865 87077Screen-printed amperometric biosensors for the rapid measurement of L- and D-amino acidsSarkar, Priyabrata; Tothill, Ibtisam E.; Setford, Steven J.; Turner, Anthony P. F.Analyst (Cambridge, United Kingdom) (1999), 124 (6), 865-870CODEN: ANALAO; ISSN:0003-2654. (Royal Society of Chemistry)Screen-printed three-electrode amperometric sensors incorporating L- and/or D-amino acid oxidase for the general purpose measurement of L- or D-amino acids are described. The working electrode incorporates rhodinized carbon, to facilitate hydrogen peroxide oxidn. at a decreased operating potential, and immobilized enzyme. The devices responded to all 20 common L-amino acids and all of the D-amino acids examd., the exceptions being L- and D-proline. Linear response profiles were obsd. for L-leucine, L-glycine and L-phenylalanine with limits of detection of 0.47, 0.15 and 0.20 mM resp. The devices were reproducible and exhibited stability over a 56 d test period. The biosensor compares favorably with a std. photometric amino acid test and was used to monitor milk aging effects. The assay is cheap, simple to perform and rapid, requiring only buffer-electrolyte and a small sample vol.
- 78Kelso E. McLean J. Cardosi M. F. Electrochemical detection of secreted alkaline phosphatase: Implications to cell based assays Electroanalysis: An International Journal Devoted to Fundamental and Practical Aspects of Electroanalysis 2000 12 490 494There is no corresponding record for this reference.
- 79Neuhold C. G. Wang J. do Nascimento V. B. Kalcher K. Thick film voltammetric sensors for trace copper based on a cation-exchanger-modified surface Talanta 1995 42 1791 179879Thick film voltammetric sensors for trace copper based on a cation-exchanger-modified surfaceNeuhold, Christian G.; Wang, Joseph; do Nascimento, Valberes B.; Kalcher, KurtTalanta (1995), 42 (11), 1791-8CODEN: TLNTA2; ISSN:0039-9140. (Elsevier)Strip-type, preconcg./voltammetric sensors, prepd. by incorporating a cation-exchange resin within screen-printed C inks, are described. Such single-use strips combine the efficient electrostatic accumulation of heavy metals using Hg-free surfaces. The uptake of Cu(II) from dil. solns. (under open circuit conditions) is followed by voltammetric measurements in a sep. blank soln. Various exptl. variables were optimized to yield low detection limits (e.g. 0.5 μg L-1 with 10 min accumulation) and good reproducibility (relative std. deviation, 2%). The applicability to assays of drinking H2O is demonstrated.
- 80Wang J. Tian B. Screen-printed stripping voltammetric/potentiometric electrodes for decentralized testing of trace lead Analytical Chemistry 1992 64 1706 170980Screen-printed stripping voltammetric/potentiometric electrodes for decentralized testing of trace leadWang, Joseph; Tian, BaominAnalytical Chemistry (1992), 64 (15), 1706-9CODEN: ANCHAM; ISSN:0003-2700.The utility of screen-printed electrodes for stripping voltammetric and potentiometric measurements of trace metals have been evaluated toward their exploitation for single-use decentralized testing. The mercury-coated screen-printed carbon electrodes, on a plastic strip, perform in a manner comparable to conventional hanging mercury drop and mercury-coated glassy carbon electrodes. Well-defined peaks are thus obtained for microgram/L (ppb) concns. of heavy metals following very short preconcn. times. The morphol. of the resulting mercury film, as characterized by SEM, facilitates trace measurements in unstirred and nondeaerated solns., as desired in many decentralized applications. Reproducible measurements of ppb lead in 100-μL drops are thus obtained. A detection limit of 30 ng/L (ppt) lead is estd. following 10-min preconcn. Convenient quantitation of lead in urine and drinking water samples is accomplished. Besides having great potential for single-use decentralized clin. or environmental testing, the highly stable response of the screen-printed electrodes makes them very attractive for routine, low-cost, centralized operations.
- 81Beitollahi H. Nejad F. G. Shakeri S. GO/Fe 3 O 4@ SiO 2 core–shell nanocomposite-modified graphite screen-printed electrode for sensitive and selective electrochemical sensing of dopamine and uric acid Analytical Methods 2017 9 5541 554981GO/Fe3O4@SiO2 core-shell nanocomposite-modified graphite screen-printed electrode for sensitive and selective electrochemical sensing of dopamine and uric acidBeitollahi, Hadi; Nejad, Fariba Garkani; Shakeri, ShahryarAnalytical Methods (2017), 9 (37), 5541-5549CODEN: AMNEGX; ISSN:1759-9679. (Royal Society of Chemistry)Herein, we report the construction of GO/Fe3O4@SiO2 core-shell nanocomposite-modified graphite screen-printed electrode that shows advantageous characteristics in terms of elec. cond., catalytic activity and surface area compared to bare screen-printed electrode. The designed sensor was used for the detn. of dopamine and uric acid. Cyclic voltammetry, differential pulse voltammetry and chronoamperometry methods were used as the diagnostics techniques. Using the modified electrode, dopamine and uric acid were detd. in the concn. ranges from 0.1 to 600.0μM and 0.75 to 300.0μM, resp., whereas the detection limits were found to be 8.9 × 10-8 M and 5.7 × 10-7 M, resp. Finally, real sample anal. was done successfully using the modified electrode.
- 82Zhao X. Zhang W. Chen H. Chen Y. Huang G. Disposable electrochemical ascorbic acid sensor based on molecularly imprinted poly (o-phenylenediamine)-modified dual channel screen-printed electrode for orange juice analysis Food analytical methods 2014 7 1557 1563There is no corresponding record for this reference.
- 83Moya A. Sowade E. del Campo F. J. Mitra K. Y. Ramon E. Villa R. Baumann R. R. Gabriel G. All-inkjet-printed dissolved oxygen sensors on flexible plastic substrates Organic Electronics 2016 39 168 17683All-inkjet-printed dissolved oxygen sensors on flexible plastic substratesMoya, Ana; Sowade, Enrico; del Campo, Francisco J.; Mitra, Kalyan Y.; Ramon, Eloi; Villa, Rosa; Baumann, Reinhard R.; Gabriel, GemmaOrganic Electronics (2016), 39 (), 168-176CODEN: OERLAU; ISSN:1566-1199. (Elsevier B.V.)Inkjet printing is a promising alternative manufg. method to conventional std. microfabrication techniques for the development of flexible and low-cost devices. Although the use of inkjet printing for the deposition of selected materials for the development of sensor devices has been reported many times in literature, it is still a challenge and a potential route towards commercialization to completely manuf. sensor devices with inkjet technol. In this work is demonstrated the fabrication of a functional low-cost dissolved oxygen (DO) amperometric sensor with feature sizes in the micrometer range using inkjet printing. All the required technol. steps for the fabrication of a complete electrochem. three electrodes system are discussed in detail. The working and counter electrodes have been printed using a gold nanoparticle ink, whereas a silver nanoparticle ink was used to print a pseudo-ref. electrode. Both inks are com. available and can be sintered at low temps., starting already at 120°C, which allows the use of plastic substrates. In addn., a printable SU8 ink formulation cured by UV is applied as passivation layer in the sensor device. Finally, as the performance of anal. methods strongly depends on the working electrode material, is demonstrated the electrochem. feasibility of this printed DO sensor, which shows a linear response in the range between 0 and 8 mg L-1 of DO, and affords a detection limit of 0.11 mg L-1, and a sensitivity of 0.03μA L mg-1. The use of flexible plastic substrates and biocompatible inks, and the rapid prototyping and low-cost of the fabricated sensors, makes that the proposed manufg. approach opens new opportunities in the field of biol. and medical sensor applications.
- 84Das S. R. Nian Q. Cargill A. A. Hondred J. A. Ding S. Saei M. Cheng G. J. Claussen J. C. 3D nanostructured inkjet printed graphene via UV-pulsed laser irradiation enables paper-based electronics and electrochemical devices Nanoscale 2016 8 15870 15879843D nanostructured inkjet printed graphene via UV-pulsed laser irradiation enables paper-based electronics and electrochemical devicesDas, Suprem R.; Nian, Qiong; Cargill, Allison A.; Hondred, John A.; Ding, Shaowei; Saei, Mojib; Cheng, Gary J.; Claussen, Jonathan C.Nanoscale (2016), 8 (35), 15870-15879CODEN: NANOHL; ISSN:2040-3372. (Royal Society of Chemistry)Emerging research on printed and flexible graphene-based electronics is beginning to show tremendous promise for a wide variety of fields including wearable sensors and thin film transistors. However, post-print annealing/redn. processes that are necessary to increase the elec. cond. of the printed graphene degrade sensitive substrates (e.g., paper) and are whole substrate processes that are unable to selectively anneal/reduce only the printed graphene-leaving sensitive device components exposed to damaging heat or chems. Herein a pulsed laser process is introduced that can selectively irradiate inkjet printed reduced graphene oxide (RGO) and subsequently improve the elec. cond. (Rsheet ∼0.7 kΩ .box.-1) of printed graphene above previously published reports. Furthermore, the laser process is capable of developing 3D petal-like graphene nanostructures from 2D planar printed graphene. These visible morphol. changes display favorable electrochem. sensing characteristics-ferricyanide cyclic voltammetry with a redox peak sepn. (ΔEp) ≈ 0.7 V as well as hydrogen peroxide (H2O2) amperometry with a sensitivity of 3.32 μA mM-1 and a response time of <5 s. Thus this work paves the way for not only paper-based electronics with graphene circuits, it enables the creation of low-cost and disposable graphene-based electrochem. electrodes for myriad applications including sensors, biosensors, fuel cells, and theranostic devices.
- 85Jović M. Zhu Y. Lesch A. Bondarenko A. Cortés-Salazar F. Gumy F. Girault H. H. Inkjet-printed microtiter plates for portable electrochemical immunoassays Journal of Electroanalytical Chemistry 2017 786 69 7685Inkjet-printed microtiter plates for portable electrochemical immunoassaysJovic, Milica; Zhu, Yingdi; Lesch, Andreas; Bondarenko, Alexandra; Cortes-Salazar, Fernando; Gumy, Frederic; Girault, Hubert H.Journal of Electroanalytical Chemistry (2017), 786 (), 69-76CODEN: JECHES; ISSN:1873-2569. (Elsevier B.V.)Herein, the authors present the large-scale fabrication of multiplexed three-electrode sensors used in a point-of-care device platform that couples a magnetic bead-based immunoassay strategy with amperometric detection for rapid and highly sensitive anal. The multiplexed sensors consisted of eight independent electrochem. cells, each with a C nanotube (CNT) working electrode, CNT counter electrode and a Ag-Ag chloride quasi-ref. electrode. The microchips were fabricated on flexible polyethylene terephthalate (PET) sheets by sequential multilayer inkjet printing (IJP) of Ag, CNT and insulator inks that were either simultaneously or subsequently post-processed (e.g. through UV photo-polymn. or photonic curing). Finally, plastic wells were mounted on top of the inkjet-printed patterns to obtain an eight-well microtiter plate where each well had a soln. capacity of 50 μL. Due to the high precision of the IJP process, the microtiter plates showed high reproducibility among the individual electrochem. cells (1-2% of deviation). Also, the microchips can be reusable for at least up to 20 times as demonstrated herein. In a customized multichannel potentiostat with eight implemented magnets matching the positions of the working electrodes, the electrochem. readout of magnetic bead based sandwich and competitive immunoassays was successfully realized for the detection of TSH and atrazine (ATR) in aq. and urine samples, resp. The achieved limits of detection for ATR (i.e. 0.01 μg/L) and TSH (i.e. 0.5 μIU/mL) demonstrated the potential of the IJP microtiter plates for the environmental and biol. quantification of analytes in a very reliable high throughput platform. IJP has certainly reached the status of a batch prodn. tool for electroanal. sensing platforms.
- 86Pu Z. Wang R. Wu J. Yu H. Xu K. Li D. A flexible electrochemical glucose sensor with composite nanostructured surface of the working electrode Sensors and Actuators B: Chemical 2016 230 801 80986A flexible electrochemical glucose sensor with composite nanostructured surface of the working electrodePu, Zhihua; Wang, Ridong; Wu, Jianwei; Yu, Haixia; Xu, Kexin; Li, DachaoSensors and Actuators, B: Chemical (2016), 230 (), 801-809CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Detecting hypoglycemia remains a great challenge with regard to continuous glucose monitoring in clin. settings. This paper investigates a novel, flexible three-electrode electrochem. sensor with a composite nanostructured working electrode surface that has been modified by graphene and gold nanoparticles in order to detect low levels of glucose with high accuracy. The sensor electrodes were fabricated on a polyimide substrate using the flexible printed circuit board (PCB) method. Graphene was modified directly onto the working electrode surface via inkjet printing, an emerging method for micro-scale fabrications, to enable glucose detection at low levels. Gold nanoparticles were electrodeposited directly onto the graphene layer to enhance the sensitivity of the sensor. The exptl. results demonstrate that the proposed sensor can precisely measure glucose with a linear range of 0-40 mg/dL and a detection limit of 0.3 mg/dL (S/N = 3), thereby demonstrating potential for hypoglycemia detection. Moreover, this flexible sensor was suitable for integration within a microfluidic chip, which could be used to transdermally ext. and collect ISF, such that a wearable device could be developed for continuous glucose monitoring.
- 87Da Costa T. H. Song E. Tortorich R. P. Choi J.-W. A paper-based electrochemical sensor using inkjet-printed carbon nanotube electrodes ECS Journal of Solid State Science and Technology 2015 4 S304487A Paper-Based Electrochemical Sensor Using Inkjet-Printed Carbon Nanotube Electrodesda Costa, Tallis H.; Song, Edward; Tortorich, Ryan P.; Choi, Jin-WooECS Journal of Solid State Science and Technology (2015), 4 (10), S3044-S3047CODEN: EJSSBG; ISSN:2162-8769. (Electrochemical Society)This paper presents a fully inkjet-printed electrochem. sensor on paper which consists of carbon nanotube-printed working, ref., and counter electrodes. The proposed technique aims at low-cost and disposable paper-based electrochem. sensors. First, a carbon nanotube (CNT) ink was inkjet-printed directly on paper, forming a conductive network. Addnl., a hydrophobic barrier was patterned on paper to limit the absorption of liq. to the designed area. The inkjet printing method allows for rapid patterning of electrodes on paper, resulting in a simple and effective electrochem. sensor. The sheet resistance of the CNT-printed paper was as low as 1 kΩ/square after 33 prints. A potential step voltammetry method was applied to det. the concn. of the analytes, iron ion (Fe2+) and dopamine (DA), with linear ranges of 10 μM-200 μM and 10 μM-100 μM, resp. The reported approach for a fully inkjet-printed electrochem. sensor is easy and cheap, and it has a potential for simple and rapid paper-based point-of-care diagnostics.
- 88Cinti S. Arduini F. Moscone D. Palleschi G. Gonzalez-Macia L. Killard A. J. Cholesterol biosensor based on inkjet-printed Prussian blue nanoparticle-modified screen-printed electrodes Sensors and Actuators B: Chemical 2015 221 187 19088Cholesterol biosensor based on inkjet-printed Prussian blue nanoparticle-modified screen-printed electrodesCinti, Stefano; Arduini, Fabiana; Moscone, Danila; Palleschi, Giuseppe; Gonzalez-Macia, Laura; Killard, Anthony J.Sensors and Actuators, B: Chemical (2015), 221 (), 187-190CODEN: SABCEB; ISSN:0925-4005. (Elsevier B.V.)Here we describe the construction and optimization of a cholesterol biosensor based on screen-printed electrodes (SPEs) modified with inkjet-printed Prussian blue nanoparticles (PBNPs). The deposition of PBNPs using inkjet printing led to the highly facile fabrication of sensors with excellent sensitivity and reproducibility for the measurement of H2O2. Further integration of the sensor with a microfabricated low vol. (4 μL) sample cell allowed the measurement of cholesterol in serum with the addn. of cholesterol oxidase. The biosensor exhibited a sensitivity to cholesterol of 2.1 μA/mM cm2 (r2 = 0.97, n = 5) and was linear in the range of 0-15 mM.
- 89Lei Y. Alshareef A. H. Zhao W. Inal S. Laser-scribed graphene electrodes derived from lignin for biochemical sensing ACS Applied Nano Materials 2019 3 1166 1174There is no corresponding record for this reference.
- 90Nayak P. Kurra N. Xia C. Alshareef H. N. Highly efficient laser scribed graphene electrodes for on‐chip electrochemical sensing applications Advanced Electronic Materials 2016 2 1600185There is no corresponding record for this reference.
- 91Soares R. R. Hjort R. G. Pola C. C. Parate K. Reis E. L. Soares N. F. McLamore E. S. Claussen J. C. Gomes C. L. Laser-induced graphene electrochemical immunosensors for rapid and label-free monitoring of Salmonella enterica in chicken broth ACS sensors 2020 5 1900 191191Laser-Induced Graphene Electrochemical Immunosensors for Rapid and Label-Free Monitoring of Salmonella enterica in Chicken BrothSoares, Raquel R. A.; Hjort, Robert G.; Pola, Cicero C.; Parate, Kshama; Reis, Efraim L.; Soares, Nilda F. F.; McLamore, Eric S.; Claussen, Jonathan C.; Gomes, Carmen L.ACS Sensors (2020), 5 (7), 1900-1911CODEN: ASCEFJ; ISSN:2379-3694. (American Chemical Society)Food-borne illnesses are a growing concern for the food industry and consumers, with millions of cases reported every year. Consequently, there is a crit. need to develop rapid, sensitive, and inexpensive techniques for pathogen detection in order to mitigate this problem. However, current pathogen detection strategies mainly include time-consuming lab. methods and highly trained personnel. Electrochem. in-field biosensors offer a rapid, low-cost alternative to lab. techniques, but the electrodes used in these biosensors require expensive nanomaterials to increase their sensitivity, such as noble metals (e.g., platinum, gold) or carbon nanomaterials (e.g., carbon nanotubes, or graphene). Herein, we report the fabrication of a highly sensitive and label-free laser-induced graphene (LIG) electrode that is subsequently functionalized with antibodies to electrochem. quantify the food-borne pathogen Salmonella enterica serovar Typhimurium. The LIG electrodes were produced by laser induction on the polyimide film in ambient conditions and, hence, circumvent the need for high-temp., vacuum environment, and metal seed catalysts commonly assocd. with graphene-based electrodes fabricated via chem. vapor deposition processes. After functionalization with Salmonella antibodies, the LIG biosensors were able to detect live Salmonella in chicken broth across a wide linear range (25 to 105 CFU mL-1) and with a low detection limit (13 ± 7 CFU mL-1; n = 3, mean ± std. deviation). These results were acquired with an av. response time of 22 min without the need for sample preconcn. or redox labeling techniques. Moreover, these LIG immunosensors displayed high selectivity as demonstrated by nonsignificant response to other bacteria strains. These results demonstrate how LIG-based electrodes can be used for electrochem. immunosensing in general and, more specifically, could be used as a viable option for rapid and low-cost pathogen detection in food processing facilities before contaminated foods reach the consumer.
- 92Lin X. Lu Z. Dai W. Liu B. Zhang Y. Li J. Ye J. Laser engraved nitrogen-doped graphene sensor for the simultaneous determination of Cd (II) and Pb (II) Journal of Electroanalytical Chemistry 2018 828 41 4992Laser engraved nitrogen-doped graphene sensor for the simultaneous determination of Cd(II) and Pb(II)Lin, Xueni; Lu, Zhiwei; Dai, Wanlin; Liu, Baichen; Zhang, Yuxin; Li, Junye; Ye, JianshanJournal of Electroanalytical Chemistry (2018), 828 (), 41-49CODEN: JECHES; ISSN:1873-2569. (Elsevier B.V.)Recently, graphene-based nanomaterials have attracted widespread attention as new electrode-modified materials for different electrochem. sensing platforms. However, graphene-based nanomaterials modified heavy metal sensors often have the disadvantages in that their detection ranges are not wide enough. To solve this problem, an easy and feasible approach derived from laser engraving technique to fabricate a high-quality 3-dimensional porous graphene-based heavy metal sensor with a quite wide detection range was established. The N-doped laser engraved graphene (N@LEG) was synthesized via introducing polyaniline (PANI) and polyvinylpyrrolidone (PVP) as N-dopant. Herein, by coupling the unique electrochem. properties and the 3-dimensional porous structure framework with large electrochem. active surface areas of LEG with the strong metal ions affinity of N atoms, N@LEG modified glassy C electrode (N@LEG/GCE) with in-situ Bi film modification was successfully used for the simultaneous detn. of Cd(II) and Pb(II) using square wave anodic stripping voltammetry (SWASV). After optimizing conditions, the proposed sensor exhibits quite wide linear ranges varying from 5 to 10 g L-1 and 10 to 380 g L-1 for Cd(II), and 0.5 to 10 μg L-1 and 10 μg L-1 to 380 μg L-1 for Pb(II), resp. The detection limits are 1.08 g L-1 (S/N = 3) for Cd(II) and 0.16 g L-1 (S/N = 3) for Pb(II), which are nearly 3 times and 60 times less than the guideline values of the drinking H2O presented by the World Health Organization (WHO), for Cd(II) and Pb(II), resp. Also, the results describe satisfactory advantages of remarkable sensitivity, selectivity, anti-interference, repeatability, reproducibility and stability. Besides, the fabricated sensor is a potential perspective to detect Cd(II) and Pb(II) in actual H2O samples.
- 93Cardoso A. R. Marques A. C. Santos L. Carvalho A. F. Costa F. M. Martins R. Sales M. G. F. Fortunato E. Molecularly-imprinted chloramphenicol sensor with laser-induced graphene electrodes Biosensors and Bioelectronics 2019 124 167 17593Molecularly-imprinted chloramphenicol sensor with laser-induced graphene electrodesCardoso, Ana R.; Marques, Ana C.; Santos, Lidia; Carvalho, Alexandre F.; Costa, Florinda M.; Martins, Rodrigo; Sales, M. Goreti F.; Fortunato, ElviraBiosensors & Bioelectronics (2019), 124-125 (), 167-175CODEN: BBIOE4; ISSN:0956-5663. (Elsevier B.V.)Graphene has emerged as a novel material with enhanced elec. and structural properties that can be used for a multitude of applications from super-capacitors to biosensors. In this context, an ultra-sensitive biosensor was developed using a low-cost, simple and mask-free method based on laser-induced graphene technique for electrodes patterning. The graphene was produced on a polyimide substrate, showing a porous multi-layer structure with a resistivity of 102.4 ± 7.3 Ω/square. The biosensor was designed as a 3-electrode system. Auxiliary and working electrodes were made of graphene by laser patterning and the ref. electrode was handmade by casting a silver ink. A molecularly-imprinted polymer (MIP) was produced at the working electrode by direct electropolymn. of eriochrome black T (EBT). As proof-of-concept, the MIP film was tailored for chloramphenicol (CAP), a common contaminant in aquaculture. The resulting device was evaluated by cyclic voltammetry and electrochem. impedance spectroscopy readings against a redox std. probe. The limit of detection (LOD) was 0.62 nM and the linear response ranged from 1 nM to 10 mM. These anal. features were better than those produced by assembling the same biorecognition element on com. graphene- and carbon-based screen-printed electrodes. Overall, the simplicity and quickness of the laser-induced graphene technique, along with the better anal. features obtained with the graphene-based electrodes, shows the potential to become a com. approach for on-site sensing.
- 94S. Nasraoui, A. Al-Hamry, A. Anurag, P. R. Teixeira, S. Ameur, L. G. Paterno, M. B. Ali, O. Kanoun , Investigation of laser induced graphene electrodes modified by MWNT/AuNPs for detection of nitrite, in: 2019 16th International Multi-Conference on Systems, Signals & Devices (SSD); IEEE, 2019; pp 615- 620.There is no corresponding record for this reference.



