About the Cover:
Azobenzene-containing cetyltrimethylammonium bromide (Azo-CTAB) surfactant molecules are originally self-assembled with their head group (red) at the interface of an oil‒water emulsion. Upon UV light illumination, they lose their surface activity and dissolve in the aqueous phase, inducing phase separation of the emulsion. Upon blue (visible) light illumination, the emulsion is reconstructed, suggesting photoswitchability. [Open AI was used to create the background.]
View the article.Reviews

Functional Amyloids as Multifunctional Platforms for Targeted Drug Delivery and Immunotherapy
Brianna McVay - ,
Dana Wolfe - , and
Ayyalusamy Ramamoorthy *
Self-assembly of molecules is increasingly utilized in the design and development of novel materials for various chemical and biomedical applications. Amyloid fibrils, traditionally associated with neurodegenerative diseases, make up a class of biomaterials formed through the self-assembly of peptides and proteins. The excellent stability and biocompatibility of cross-β-sheet nanostructured amyloid fibrils make them promising biomaterials for therapeutic delivery and immunomodulation. Recent studies have demonstrated the ability of amyloid fibrils to encapsulate small molecules, support receptor-specific targeting through surface functionalization, and enable environmentally responsive drug release. Amyloid-based scaffolds have also shown notable potential in vaccine development and the controlled delivery of checkpoint inhibitors. Current research aims to hybridize amyloid-based scaffolds with commonly used drug delivery tools, such as liposomes and hydrogels, to further enhance therapeutic efficacy while minimizing toxicity. In immunological applications, amyloid fibrils serve as both carriers and adjuvants, promoting antigen presentation and sustained immune activation. This review provides an overview of current progress in the design and application of functional amyloid nanostructures for targeted drug delivery and immunotherapy. The growing understanding of engineered amyloid-based drug delivery platforms may drive the development of precision medicine and support the design of innovative biomaterials.
Articles

Activation of Surface Lattice Oxygen by Ni-Doped CeO2 To Enhance CO-SCR Reactivity and Antioxidant Performance Testing: Density Functional Theory Study
Mingtao Yang - ,
Shuhao Li - ,
Zihang Liu - ,
Yang Bai - ,
Long Chen - , and
Boxiong Shen *
Selective catalytic reduction of nitric oxide using carbon monoxide from industrial flue gas is considered an economical and environmentally friendly method of denitrification (CO-SCR). Ni-doped CeO2 catalysts exhibited high denitrification performance, where the distribution and number of oxygen vacancies were the key factors affecting the performance. In this paper, Ni-doped CeO2 (1 1 1) surface CO reduction of NO has been thoroughly investigated and its reactivity has been evaluated by using density functional theory, electronic structure analysis, and transition state theory. The negative oxygen vacancy formation energy of the Ni-CeO2 (1 1 1) surface is manifested by the easier activation of the surface lattice oxygen. The Ni-CeO2 (1 1 1)-O1VO5V structure the reaction path that generates N2 via the ONCO intermediate has the lowest energy barrier and is the best reaction path. When oxygen is present, not only does it compete with NO for the active site, but the adsorption of O2 at other sites also changes the position of already adsorbed NO. It also destroys ONNO and ONCO intermediates, thus blocking or hindering subsequent reactions. When trace amounts of oxygen are present in the reaction system, only the trioxide vacancy CeO2 (1 1 1)-O1VO5VO7v structure via the N2O2 intermediate to generate the N2 reaction pathway can go through. However, the reactivity was reduced. In this study, the intrinsic mechanism of Ni doped CeO2 catalysts with high denitrification performance was deeply elucidated at the atomic level, and the effect of O2 on the reaction process was also revealed.
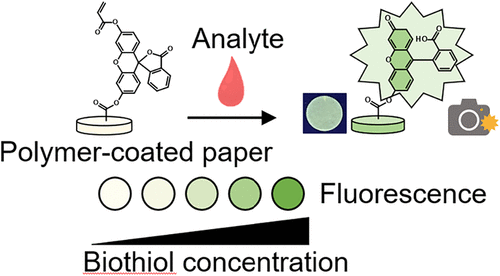
Paper-Based Analytical Device Coated with a Fluorescent Polymer for the Visual Detection of Biothiols
Hikaru Amo - ,
Naoki Sasaki - ,
Takahiro Saijo - ,
Ryoko Kawabata - ,
Kenta Morita - , and
Tatsuo Maruyama *
The reducing capacity of biological and environmental aqueous samples is a key indicator of oxidative stress and redox balance. Thiol-containing compounds (biothiols) are essential in maintaining this balance within biological systems. In this study, we developed a paper-based analytical device (PAD) for the rapid and quantitative detection of biothiols in aqueous solutions and cell homogenates. The PAD uses a cysteine-specific fluorescent probe that generates a signal proportional to the biothiol concentration. Assay conditions and reaction kinetics were optimized to expand the dynamic range and lower the detection limit for cysteine (Cys). By incorporation of cystine, PAD enabled accurate quantification of other biothiols with high reproducibility and sensitivity. We validated the PAD using standard reducing agents and complex biological samples, demonstrating a strong correlation with the conventional spectroscopic method using 5,5′-dithiobis(2-nitrobenzoic acid). This cost-effective, portable, and user-friendly PAD offers a versatile solution for on-site and real-time redox monitoring, with potential applications in biomedical research, diagnostics, and environmental monitoring.
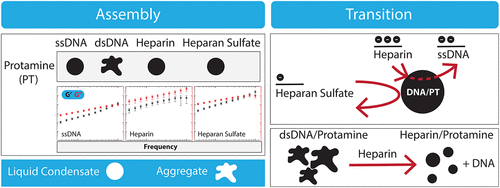
Protamine Drives Liquid and Solid Condensation of DNA and Glycosaminoglycans
Florian J. F. van der Harten - ,
Vahid Sheikhhassani - , and
Alireza Mashaghi *
This publication is Open Access under the license indicated. Learn More
Phase-separated biomolecular condensates play a crucial role in cellular organization. In the nucleus, phase separation regulates the assembly and function of nuclear proteins and the genome. Here, we demonstrate that protamine, an arginine-rich nuclear protein responsible for promoting double-stranded DNA (dsDNA) compaction in sperm, undergoes phase separation with single-stranded DNA (ssDNA) and induces dsDNA aggregation. The protamine-ssDNA condensates behave as viscoelastic liquid droplets and can be modulated by varying salt concentrations or by treatment with heparin, a glycosaminoglycan polyanion, which we show displaces ssDNA. Moreover, protamine-dsDNA aggregates dissolve upon exposure to heparin, leading to the formation of condensates with a distinct morphology. This observation provides a compelling example of an aggregate-to-condensate transition in polyelectrolyte systems. Notably, direct combination of protamine with heparin results in the formation of similar phase-separated liquid-like droplets, suggesting that heparin can compete with both ssDNA and dsDNA for protamine binding. We performed a comparative analysis using other positively charged proteins and negatively charged glycosaminoglycans to gain insights into the condensation behavior of protamine and heparin. Finally, we leveraged these findings to conduct a proof-of-concept analysis aimed at developing programmable biomolecular condensates for protamine-assisted nucleic acid delivery. Given that polyanionic glycosaminoglycans have been used for sperm capacitation and that DNA fragmentation is a biomarker for infertility in males, our findings offer new insights into the mechanisms of protamine-driven DNA compaction in sperm and its potential implications for reproductive medicine.
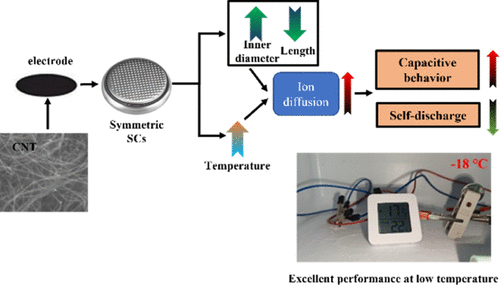
High-Performance Carbon-Nanotube-Based Supercapacitors at a Wide Temperature Range: Geometrical Effect on Diffusion of Electrolytic Ions
Yunkuo Sun - ,
Baohong Ding *- ,
Yonghua Jiao - , and
Wei Sun *
Supercapacitors (SCs) characterized by excellent charge and discharge rates, high power density, and stable cycling performance, exhibit crucial applications in various fields, while it faces significant performance degradation at low temperature. Herein, we systematically investigate the electrochemical performances of carbon nanotube (CNT)-based SCs over a temperature range of −18 to 60 °C and establish quantitative relationships between CNT geometry and temperature-dependent electrochemical kinetics in symmetric SCs. The results and analysis supported by electrochemical impedance spectroscopy (EIS) with Warburg diffusion analysis, Arrhenius modeling of ion diffusion kinetics, and multiterm self-discharge modeling reveal exceptional low-temperature resilience where CNT-based SCs retain >85% peak specific capacitance (75.76 F/g at 0.5 A/g) with 87% rate retention at 20 A/g (−18 °C), attributable to minimized diffusion barriers in CNTs with shorter length and wider channel that reduce Arrhenius activation energy by 33% (CNT-8-L: Q = 15.40 kJ/mol vs CNT-3-L: 23.07 kJ/mol). In addition, a symmetric CNT-based SC is successfully employed to power a digital thermometer. The self-discharge compensation effect enables optimized energy deliver of the CNT-based SC for over 40 min to the digital thermometer at −18 °C (approximately 4 times longer than at 60 °C) through suppression of current leakage as well as ion diffusion, although the specific capacitance is lower. The experimental findings and analyses contribute to the design and optimization of CNT geometries for low-temperature applications and deepen our understanding of the underlying energy storage mechanisms.
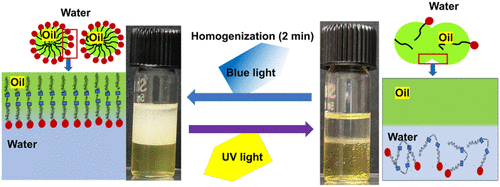
Stability, Photoinduced Destabilization, and Photoswitchability of Azo-CTAB-Based Emulsions
Guguloth Naresh - ,
Roopesh Patali - ,
Ethayaraja Mani *- , and
Sreeram K. Kalpathy *
Stimuli-induced interfacial destabilization of oil–water emulsions is an attractive approach for emulsion processing, as it typically requires less energy input than conventional bulk manipulation methods. In this work, we demonstrate the photoinduced destabilization and reconstruction of n-decane-in-water emulsions that were rendered highly stable (>45 days) with the help of an azobenzene-containing cetyltrimethylammonium bromide (Azo-CTAB) surfactant. Phase separation was induced easily in Azo-CTAB stabilized emulsions by the illumination of ultraviolet (UV) light, which activates photoisomerization of the Azo-CTAB molecule and would, in turn, affect its interfacial adsorption. A simple analysis based on the size of the dispersed phase droplets shows that phase separation occurs by coalescence of the droplets, following second-order kinetics. The coalescence rate constant is higher by about 5 orders of magnitude when demulsification is induced by UV exposure, compared to those in samples stored under ambient conditions. The degree of phase separation can be controlled by tuning various factors, such as the power intensity of UV light, the addition of an electrolyte, and the addition of a mutually soluble solute. The coalescence rate is faster by about 4–5 times in the presence of an added electrolyte (NaCl). When UV light of low intensity (∼1 mW cm–2) is used, complete demulsification can still be achieved if a small quantity of a volatile liquid such as ethanol is added to the emulsion. More interestingly, we show that the original emulsion can be reconstructed by subsequently illuminating the phase-separated mixture with visible light, thus demonstrating photoswitchability in Azo-CTAB stabilized emulsions. The reversible photoswitchability persists for multiple (5–7) cycles, without any appreciable change in the appearance and size of the dispersed droplets. These results have great significance in applications that require repeated and facile stabilization and destabilization of emulsions, such as oil recovery and wastewater treatment.

Gas-Solvent-Assisted CO2 Injection for Shale Oil Recovery: Modifying Oil Properties and Enhancing Competitive Adsorption
Yuhao Lu - ,
Jian Wang *- ,
Tianhan Xu - ,
Danling Wang - ,
Hongkun Wei - , and
Yingbo Sun
Given the limited improvement in shale oil recovery from microscopic pores through pure CO2 injection, this study explores the potential of gas-solvent-assisted CO2 to enhance recovery by improving miscibility and displacement efficiency in shale oil. Systematic experiments revealed the interaction effects between gas-solvent-assisted CO2 and shale oil. Molecular dynamics simulations were utilized to examine the enhanced oil displacement performance and underlying interaction mechanisms. Furthermore, the role of gas solvents in augmenting CO2’s competitive adsorption capabilities was explored. The research results showed that the addition of gas solvents reduces the saturation pressure of shale oil, enhances the CO2 solubility, and increases the expansion coefficient while significantly decreasing the viscosity, density, and minimum miscibility pressure (MMP). In displacement models, gas-solvent-assisted CO2 systems exhibited stronger interactions with octane and SiO2, leading to markedly improved displacement efficiency compared to pure CO2 systems and enabling faster, more complete interactions between CO2 and octane. Furthermore, competitive adsorption experiments highlight the ability of gas solvents to enhance CO2 adsorption over CH4 in nanopores, increasing selectivity coefficients and optimizing adsorption profiles. These findings provide critical insights into the potential of gas-solvent-assisted CO2 injection as a viable strategy for enhancing shale oil recovery and advancing the understanding of gas–liquid interactions in unconventional reservoirs.
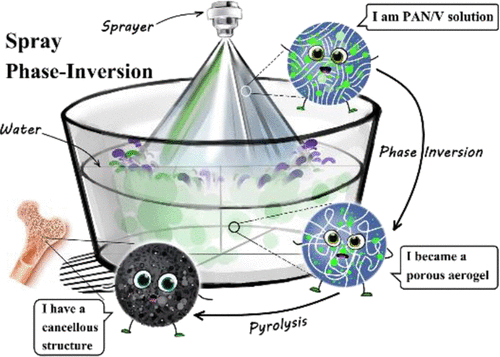
Bionic Bone Structure Anode Material of High-Capacity 3D Porous Vanadium Nitride@Carbon Nanocomposite via Spray Phase Inversion
Runze Wang - ,
Yuanyou Peng - ,
Chunjin Ai - ,
Meimei Yu - ,
Guang Liu - ,
Tianqi He - ,
Jie Zhang - ,
Junlong Chen - ,
Shengqiang Nie - , and
Fen Ran *
Vanadium nitride is expected to address the energy density challenge of supercapacitors due to its excellent performance; however, its poor cycle life and rate performance limit its application potential. Compounding vanadium nitride with carbon materials can be a good solution, but a well-designed composite structure is required to fully exhibit the electrochemical properties. In this study, porous carbon composites embedded with vanadium nitride nanoparticles are constructed by a spray phase-inversion strategy. The composites have a mesh structure, which similar to cancellous bone, and exhibit good electrochemical performance as a supercapacitor anode material: the specific capacity reaches 431.5 F g–1 at the current density of 0.5 A g–1, and capacity retentions of 78.7% after 5000 cycles in a 6 M KOH electrolyte solution. The good performance is attributed to the fact that the carbon skeleton provides an efficient channel for the transfer of electrons and ions, and the vanadium nitride nanoparticles are effective in increasing the capacity while being well confined by the carbon skeleton. The porous structural composites are versatile and may have good potential for applications in other energy conversion and energy storage systems.

Hierarchically Porous Materials via Co-Assembly of Exfoliated Layered Silicate with Mesoporous Silica Nanoparticles
Sota Hasebe - ,
Masashi Yatomi - ,
Nonna Hori - ,
Takeshi Iwakami - ,
Takamichi Matsuno *- , and
Atsushi Shimojima *
Anisotropic nanoscale building blocks offer significant potential for the construction of porous architectures with tailored structural and functional properties. In this study, we report the successful exfoliation of magadiite─a crystalline layered silicate─into two-dimensional silica nanosheets and their co-assembly with mesoporous silica nanoparticles (MSNs) to fabricate a new class of hierarchically porous silica-based materials. Structural analysis confirmed that MSNs are uniformly distributed between the silica nanosheets, retaining the in-plane crystallinity of magadiite. The resulting composite exhibited a remarkable nitrogen adsorption capacity, a high specific surface area (909 m2 g–1), and a large pore volume (1.89 cm3 g–1). A distinct feature is the emergence of new mesopores (∼11.5 nm), which are attributed to interparticle gaps formed through nanosheet–particle co-assembly. These findings demonstrate the utility of exfoliated layered silicates as versatile building blocks for the design of porous silica materials.

Self-Assembly Properties of Xylene-Derived Constitutional Isomers of Fmoc-Phenylalanine
Pamela Agredo - ,
Ritty Mohan - ,
Sydney T. Carter - , and
Bradley L. Nilsson *
This publication is Open Access under the license indicated. Learn More
Fluorenylmethoxycarbonyl-phenylalanine (Fmoc-Phe) derivatives are a privileged molecular class that readily undergoes supramolecular self-assembly into hydrogel networks. Herein, we characterize the self-assembly properties of xylene-derived constitutional isomers of Fmoc-Phe, demonstrating the impact of molecular configuration on the emergent structure and properties of supramolecular assemblies of these derivatives. The self-assembly properties of Fmoc-Phe and a cationic derivative of Fmoc-Phe that has been modified at the C terminus with diaminopropane (DAP), Fmoc-Phe-DAP, were compared to those of several corresponding xylene derivatives in which Fmoc-functionalized amine and carboxylic acid or DAP-functionalized carboxylic acid are organized around a central benzene ring, with the appended functionality oriented in ortho, meta, or para spatial arrangements. Under conditions where Fmoc-Phe and Fmoc-Phe-DAP derivatives undergo self-assembly into fibrillar supramolecular hydrogel networks, it was found that corresponding xylene derivatives assemble into distinctive nanoribbon/nanotape morphologies that fail to form supramolecular networks that elicit emergent hydrogel formation. The assemblies formed are dependent on the spatial arrangement of the xylene core structure. These studies provide insight into the significant effects of molecular arrangement on the supramolecular self-assembly properties of constitutional isomers of phenylalanine derivatives.
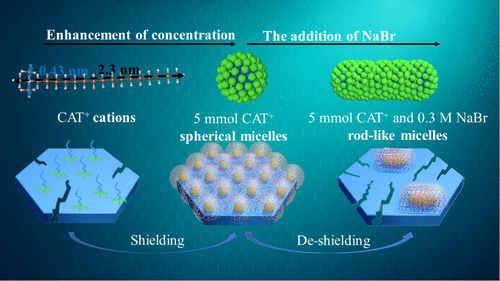
The Overlooked Roles of Long-Chain Alkyl Quaternary Ammonium Cations in the Mesopore Engineering of ZSM-5 Zeolite
Jiale Zhao - ,
Keyu Yang - ,
Ning Wei - ,
Yunxuan Ji - ,
Bingru Li - ,
Jiaxu Liu - ,
Guiqiu Wang *- ,
Peidong Li *- , and
Shengjun Huang
The introduction of a specific concentration of long-chain alkyl quaternary ammonium cations inhibits the construction of mesoporosities inside MFI zeolites through alkaline treatment. The spontaneously formed micelles bear an electrical double-layer structure, which imposes repulsion on the caustic hydroxide ions (OH–) and therefore restrains the dissolution of the zeolite in the alkaline media. The aforementioned inhibitive effect of long-chain alkyl quaternary ammonium cations can be disabled by the addition of an inorganic salt, such as NaBr, which induces a transformation in micelle morphology and the micellar electrical double-layer structure. In the case of alkyl quaternary ammonium cations with short alkyl chains (e.g., trimethyloctylammonium: C8TA+), such an inhibitive effect cannot be eliminated by the addition of an inorganic salt like NaBr. These findings update the understanding of the roles of alkyl quaternary ammonium cations and their micelle forms in tailoring the porosities inside zeolites.

Cholesterol Modulates Ionic Liquid–Lipid Membrane Interactions
Jyoti Gupta - ,
Veerendra K. Sharma *- ,
Prashant Hitaishi - ,
Akash K. Jha - ,
Jyotsna Bhatt Mitra - ,
Harish Srinivasan - ,
Sugam Kumar - ,
Ashutosh Kumar - ,
Sajal K. Ghosh - , and
Subhankur Mitra
Understanding the role of cholesterol in ionic liquid (IL)–membrane interactions is essential for advancing biomedical applications of ILs, including the development of innovative antimicrobial agents. In this study, we explore the intricate and multifaceted role of cholesterol in modulating IL–membrane interactions employing a comprehensive suite of biophysical techniques. We systematically examine how IL alkyl chain length and membrane physical state influence the impact of cholesterol on the IL–lipid membrane interaction. The incorporation of ILs is shown to increase the area per lipid in both pristine dipalmitoylphosphatidylcholine (DPPC) and DPPC/cholesterol membranes. Cholesterol modulates the impact of ILs on the lipid conformation, membrane viscoelasticity, and phase behavior. Small-angle neutron scattering and dynamic light scattering measurements reveal that cholesterol mitigates IL-induced structural perturbations in the vesicles. Interestingly, while cholesterol significantly weakens IL binding, membranes containing cholesterol exhibit a greater permeabilization. This counterintuitive behavior arises from cholesterol’s ordering effect on the lipid bilayer, which, while stabilizing the structure, increases its vulnerability to stress-induced defects. Our results underscore the complex and nonuniversal interplay between lipid composition, IL alkyl chain length, and membrane phase state. These insights provide a deeper understanding of cholesterol’s role in IL–membrane interactions, paving the way for the design of advanced applications of ILs in antimicrobial therapy and drug delivery.
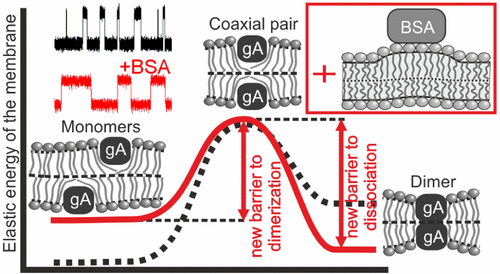
Adsorption of Bovine Serum Albumin to Lipid Membranes Increases the Number and Stability of Ion Channels of Gramicidin A
Oleg V. Kondrashov - ,
Tatyana I. Rokitskaya - ,
Zaret G. Denieva - ,
Sergey A. Akimov *- , and
Yuri N. Antonenko
Local mechanical properties of lipid membranes participate in the regulation of biological processes. Adsorption of peripheral proteins may induce lateral inhomogeneity in membranes. We studied the interaction of bovine serum albumin (BSA) with lipid membranes and its effect on the characteristics of ion channels formed by gramicidin A (gA). We proposed a qualitative mechanism for the induction of local membrane deformations by adsorbed BSA molecules. In the presence of gA in the membrane, BSA adsorption changes the characteristics of the transmembrane ion current, presumably due to gA-BSA lateral interactions mediated by membrane deformations induced by gA and BSA. With the one-sided addition of gA to the membrane, subsequent one-sided or two-sided addition of BSA significantly increased the gA-mediated ion current. We assumed that this is due to an increase in the rate of equalization of the gA concentration in two membrane monolayers by its transition through the membrane (flip-flop). In addition, it was experimentally observed that the addition of BSA to a membrane with symmetrically inserted gA increased the ion current many-fold, suggesting an increase in the average number of conducting gA dimers. An increase in the average lifetime of the gA channels was also shown. We developed a theoretical model that accounts for membrane deformations in three states of gA in the membrane: two monomers, coaxial pair, and conducting dimer. The calculated energy of membrane deformations in these states depended strongly on the presence of BSA, leading to the shift of the monomer–dimer equilibrium of gA toward dimers. The model qualitatively describes the experimentally observed effects of BSA on the characteristics of the gA channel.
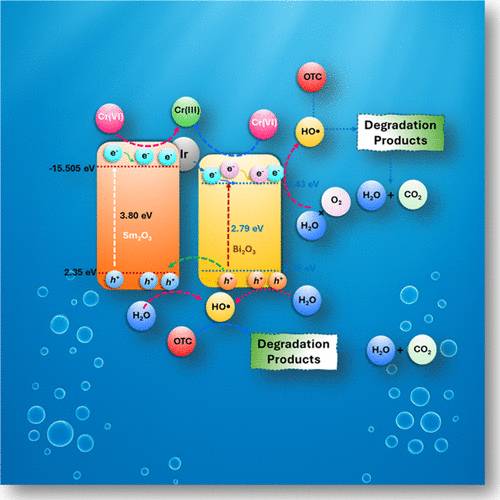
Synergistic Charge Transfer-Enhanced Photocatalytic Degradation of Oxytetracycline and Cr(VI) Reduction Using Sm2O3/Ir@Bi2O3
Sanjenuganth B - ,
Govindan Kadarkarai - ,
Mamba Gcina - ,
Maruthamani D - , and
Muthuraj Velluchamy *
In this present investigation, a Sm2O3/Ir@Bi2O3 ternary nanocomposite was fabricated using the hydrothermal method, and comprehensive physicochemical characterization was conducted to understand the morphology, crystal phase, optical behavior, and elemental composition. The prepared material was investigated for the degradation of oxytetracycline (OTC) and chromium(VI) reduction under UV–vis. irradiation. The Sm2O3/Ir@Bi2O3 composite displayed the highest photocatalytic activity toward OTC mineralization (95% in 55 min) and Cr(VI) reduction (91% in 45 min), surpassing standalone Sm2O3 and Bi2O3, due to the synergistic effect. These removal efficiencies correspond to rate constants of 0.073 min–1 and 0.058 min–1 for the degradation of the OTC and Cr(VI) reduction, respectively. These were 55 times and 40 times higher than the rates achieved using Sm2O3 as a catalyst for OTC and Cr(VI) detoxification, respectively. The influence of various reaction conditions, such as catalyst dosage, pollutant concentration, pH, and inorganic ions, was studied during both the OTC and Cr(VI) pollution mitigation. Coexisting anions greatly governed the Sm2O3/Ir@Bi2O3 photocatalytic performances. Reactive oxidative species trapping and EPR analyses revealed that the dominant reactive species follow the order HO• > O2•– > h+ > e– for OTC degradation and e– > O2•– > h+ > HO• for Cr(VI) reduction. The probable OTC degradation pathway involved both direct and indirect oxidation mechanisms, primarily decarboxylation, hydroxylation, and demethylation. This study demonstrated the successful development of a material with versatile photocatalytic properties. The high photocatalytic efficiency of the Sm2O3/Ir@Bi2O3 nanocomposite in degrading antibiotics and reducing toxic heavy metals demonstrates its potential as a practical solution for advanced wastewater treatment in pharmaceutical and industrial effluents.
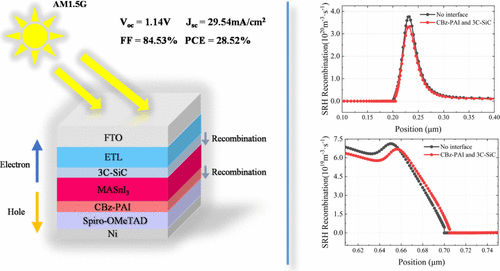
Design and Numerical Optimization of Dual Interface Layers for MASnI3 Perovskite Solar Cells Based on SCAPS-1D Simulation
Yuxing Gao - ,
Haoyu Jiang - ,
Wenjie Zhang - ,
Yongmin Ying - ,
Le Chen - ,
Limei Han - ,
Zhuoxin Dong - ,
Min Li *- ,
Chaoen Li *- , and
Jiang Wu *
Lead-based perovskite solar cells (PSCs) have achieved a power conversion efficiency (PCE) of 27%, demonstrating excellent photovoltaic performance. However, lead toxicity remains a significant barrier to large-scale commercialization. Tin-based perovskites, with lower toxicity and suitable bandgaps, are considered promising lead-free alternatives. Their implementation remains challenged by poor stability and pronounced interfacial recombination. Interface engineering, an important approach to enhance device performance, has been extensively studied in lead-based PSCs, while systematic research on tin-based PSCs remains limited. In this work, we propose a dual-interface structure combining 3C-SiC and CBz-PAI, optimized through SCAPS-1D simulations. This structure effectively suppresses carrier back-diffusion and minimizes recombination at critical interfaces. Compared to the initial structure, the dual-interface design improves the fill factor (FF) and PCE by 10.82% and 3.09%. After optimization and resistance adjustments, the device achieves an open-circuit voltage (Voc) of 1.1418 V, a short-circuit current density (Jsc) of 29.54 mA/cm2, a FF of 84.53%, and a PCE of 28.52% at 250 K. Additionally, the device maintains stable operation over a wide temperature range from 240 to 360 K. These findings provide theoretical insight into the design of high-performance lead-free PSCs and offer a pathway toward environmentally sustainable solar technologies.
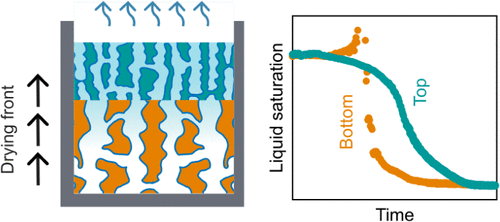
Directional Drying in Bilayer Porous Films: Funnel vs Ink-Bottle Geometries
Luisa Guadalupe Cencha *- ,
Alesio Nehuén Benitez - ,
Nicolas Budini - ,
Claudio Luis Alberto Berli - , and
Raul Urteaga
We investigate the drying dynamics of mesoporous silicon bilayers with controlled pore geometries during ethanol evaporation. Using Reflective Interferometric Fourier Transform Spectroscopy (RIFTS), we track the evolution of liquid saturation in each layer. We find that placing smaller pores atop larger ones─mimicking an ink-bottle geometry─can unexpectedly accelerate evaporation under specific thickness ratios. This behavior suggests a complex interplay of mass transport mechanisms beyond simple geometrical constraints. Additionally, we introduce a novel image-based method for spatially resolved saturation mapping over millimeter-scale areas, complementing the localized RIFTS measurements. Our results demonstrate that evaporation dynamics in porous films can be modulated through structural design, providing new experimental tools to study and tailor fluid transport in hierarchical porous materials.

A Molecular Simulation Study for Understanding the Effects of Two Water Channel Morphologies in Nafion with Various Hydration Levels on Adsorption and Nonideal Behaviour of Methanol at Operating Conditions of Direct Methanol Fuel Cells
Sarangi Veliyil Santhamma - ,
Sahana Hanumanthappa Kenchgundi - , and
Angan Sengupta *
Fuel loss inside direct methanol fuel cells (DMFCs) having Nafion membranes with two distinct water channel morphologies (viz., lamellar water channel morphology (LWCM) and random water channel morphology (RWCM), both validated against the literature having comparable equivalent weights and membrane densities) has been quantified through a methanol adsorption (ρCH3OH) study using GCMC simulations. Subsequently, variations in nonideality of adsorbed methanol as a function of Nafion–water–methanol local mole fractions are estimated at DMFC operating conditions, which are governed by ρCH3OH inside different morphologies. For all temperatures with given hydration (λ), ρCH3OH values are significantly higher inside LWCM with a single ordered channel compared to RWCM with localized hydrophilic regions and inherent tortuosity. While ρCH3OH decreases with increasing λ inside LWCM, inside RWCM, ρCH3OH is constantly lower at λ = 3 and varies relatively for λ = 6 and 9, depending on the DMFC operating temperature. Pore volume and active site availability in hydrated Nafion result in a competitive arrangement of methanol with respect to the bound water and hydronium mixture, only at the LWCM centre and at localized hydrophilic sites of RWCM. These observations have been justified further by estimating the preferential solvation parameters. Interestingly, the molecular ordering of bound water inside all Nafion remained invariant upon methanol adsorption. However, enhanced molecular ordering of adsorbed methanol in comparison to its pure bulk phase also indicates the possibility of activity coefficients (γCH3OH) < 1 inside hydrated Nafion. Calculations of excess entropy substantiate ρCH3OH,LWCM > ρCH3OH,RWCM, at constant water compositions.
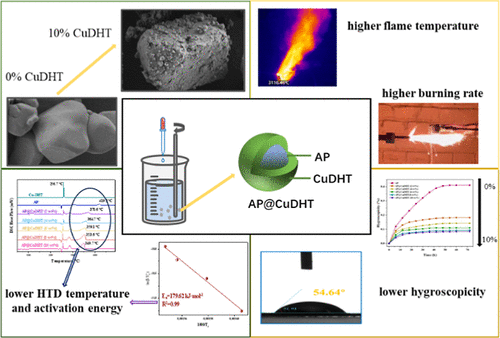
Simple Preparation of AP@CuDHT Core–Shell Structure: A “One Stone, Three Birds” Method for Comprehensively Improving the Energy, Thermal Properties, and Hygroscopicity of AP
Shilong Li - ,
Xiaodong Gou - ,
Wei Liu *- ,
Zhongliang Ma - ,
Xiaojun Wang - , and
Xiaojuan Yang
Comprehensive improvement in the thermal properties, energy characteristics, and hygroscopicity of ammonium perchlorate (AP) carries substantial significance to its utilization in solid propellants. In the present study, a novel coating agent, CuDHT, was synthesized via the self-assembly of 3,6-dihydrazino-1,2,4,5-tetrazine (DHT) and transition metal ions (Cu2+), which can comprehensively enhance the performance of AP. The morphology, composition, and thermal properties of samples prepared in this work were examined via scanning electron microscopy, Fourier transform infrared spectroscopy, powder X-ray diffraction, differential scanning calorimetry, and thermogravimetric analysis techniques. Meanwhile, the burning rate and combustion temperature of AP@CuDHT-based solid propellants were tested via infrared thermography. The findings show that AP particles have their surfaces coated with a homogeneous and compact CuDHT coating layer. Moreover, the CuDHT coating layer is capable of markedly catalyzing AP’s thermal decomposition and reducing its hygroscopicity. As the CuDHT coating content increases, the peak temperature of AP@CuDHT’s high-temperature decomposition and moisture absorption rate gradually decrease. When the CuDHT content reaches 10 wt %, the HTD peak temperature of AP@CuDHT moves forward by 69.5 °C relative to pure AP, and the moisture absorption rate is cut by 0.326 wt %. Compared with pure AP, AP@CuDHT (10%)’s impact and friction sensitivities are higher by 4% and 8%. Additionally, CuDHT can remarkably enhance the combustion performance of solid propellants. With an increase in CuDHT content, the burning rate of AP@CuDHT-based propellants and their combustion temperature gradually increase. At 10 wt % CuDHT, the burning rate and combustion temperature are enhanced by 6.1 mm·s–1 and 572.6 °C, respectively, compared to those of pure AP-based propellants. In summary, this study provides references for comprehensively improving the hygroscopicity, energy, and thermal properties of AP and, meanwhile, demonstrates a significant application outlook for promoting the combustion performance in solid propellants.
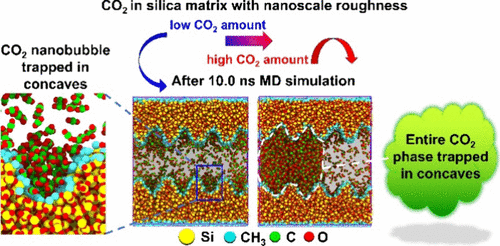
Molecular Dynamics Simulation of CO2 Molecular Behaviors in Silica Nanopores: Effect of Nanoscale Surface Roughness
Hongye Xu - ,
Yunfeng Liang - ,
Naipeng Zhao - ,
Jiangtao Pang *- , and
Fulong Ning *
Nanoscale roughness of reservoir skeleton surfaces inevitably affects the CO2 geo-sequestration, and its exact microscopic mechanism remains elusive. Here, nanosecond molecular dynamics (MD) simulations were performed to investigate this effect with silica nanopore models. We classified the surface into “nano-valleys” and “nano-peaks” by the median z-coordinate of surface atoms and further divided nanovalleys into shallow and deep types. The results demonstrate that the nanovalleys can trap CO2 molecules, resulting in lower CO2 diffusivity and higher local concentration compared to nanopeaks. Generally, the total CO2 quantity on nanovalleys and nanopeaks is increasing as surface roughness increases. A further exploration shows that the CO2 concentration of the deep valley is always higher than that of the shallow valley under the same degree of roughness and exhibits an increasing trend as surface roughness increases. Furthermore, CO2 molecules enter nanovalleys vertically and adsorb parallel to the surface, while water molecules orient randomly. In a high CO2 concentration system, CO2 nanobubbles are observed in nanovalleys. The nanobubbles are smaller but more numerous as the surface roughness increases. In a dual-phase system, the boundary between CO2 and liquid phases connects the nanopeaks of top and bottom layers, embedding the CO2 phase in concaves, which indicates the restrictive effect of nanopores on the CO2 phase. These molecular insights confirm the accumulation and retention of prestored CO2 due to nanoscale roughness on the reservoir surface.
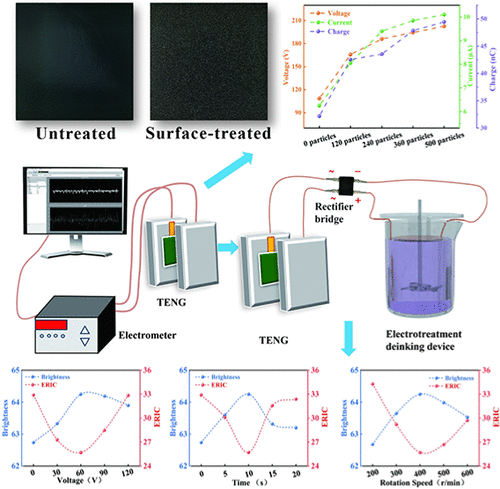
Surface Treatment of PDMS Triboelectric Composite Materials and Their Application in the De-Inking of Waste Paper
Zhuo Chen - ,
Xiaopin Sun - ,
Mengyang Wang - ,
Zhiwei Chen - ,
Wenyi Guo - ,
Xianze Li - ,
Xiaoxu Liu - , and
Lianxin Luo *
The increasing prominence of energy shortages and environmental issues has encouraged research on low-carbon, clean, and sustainable energy technologies. In the fields of sensing, motion monitoring, and energy harvesting, self-powered electronic devices are developing rapidly, posing more severe challenges for triboelectric materials. In this study, polyaniline-modified cellulose was prepared via in situ polymerization and combined with biocompatible polydimethylsiloxane to create a triboelectric material with enhanced strength and conductivity. After being treated with 500-mesh sandpaper, the material exhibited the best electrical output performance, with a voltage of 206.8 V, a current of 10.1 μA, and a charge of 50.44 nC. The triboelectric nanogenerator made from this material was integrated with the deinking of old mechanical-pulp waste paper. The deinking effects under different conditions were investigated, and the deinking principle was explored from the perspective of the ink itself.

Accelerating Precuring of Autoclaved Aerated Concrete (AAC) via Hybrid Alkali-Activated Pisha Sandstone: Suppression of Montmorillonite Swelling and Pore Structure Optimization
Zhihong Wu *- ,
Jin Wang - ,
Jingzhi Peng - ,
Jiawei Wang - ,
Yuexin Pan - ,
Rui Li - ,
Xiaogang Bian - , and
Yang Yang
Pisha Sandstone (PS), characterized by its high silica content (76.94% SiO2), presents a promising alternative to traditional quartz sand in construction applications due to its abundant availability and low cost. However, its montmorillonite clay minerals induce expansion during the precuring stage of autoclaved aerated concrete. When the PS content accounts for 42% of the total siliceous materials, extending the precuring time to approximately 5.1 h and hindering gas evolution within the green body, thereby severely compromising production efficiency. This study employed a NaOH and Na2SiO3 hybrid alkali activation system to modify PS. Experimental results reveal that adding 1 wt % each of NaOH and Na2SiO3 (N1S1), dynamic equilibrium between slurry thickening and gas evolution rates was achieved, yielding homogeneous pore structures and reducing precuring time by 37.3%, from 5.1 to 3.2 h. Mechanistic analysis indicates that NaOH effectively disrupts montmorillonite’s layered structure, while it synergizes with Na2SiO3 to promote cogeneration of C-(A)-S-H gels and ettringite (AFt), thereby enhancing early stage hardening.
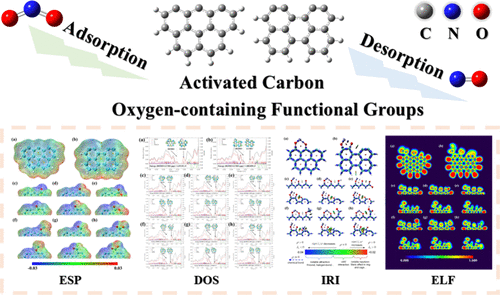
Effect of Oxygen-containing Functional Groups on the NO2 Adsorption and Reduction by Activated Carbon: A Density Functional Theory Calculation Study
Tong Hao - ,
Qian Zhou - ,
Jinyuan Jiang *- ,
Mingyao Liu - ,
Wei Tan - ,
Haoyang Song - ,
Lei He - ,
Dongni Shi - ,
Hongke Qin - ,
Yajun Li - , and
XiaoJun Pan
The activated carbon effectively removes nitrogen dioxide (NO2) gas from environmental air, and its adsorption-reduction performance is significantly influenced by surface oxygen-containing functional groups (OFGs). However, the internal mechanisms of different OFGs in the complete reaction processes remain unclear. Based on previous studies and experimental characterization results, this paper selects two typical carbon edge structure models and six different OFGs as fundamental models. Using density functional theory, wave function analysis, and thermodynamic and kinetic analyses, we comprehensively investigate the microscopic reaction pathways of the NO2 molecule on carbon edge structures modified with OFGs. The results show that most OFGs inhibit NO2 adsorption and N–O bond cleavage via van der Waals interactions, while their impact on NO desorption is negligible due to localized effects. Thermodynamic and kinetic analyses jointly validated these findings. Importantly, the results highlight that zigzag edge structures exhibit superior reactivity toward NO2 reduction, suggesting that carbon materials prepared below 400 °C with minimal OFG incorporation are more favorable. This dual-optimization strategy provides practical guidance for enhancing the NO2 conversion performance, offering a molecular-level foundation for the rational design of advanced carbon-based adsorbents or catalysts.
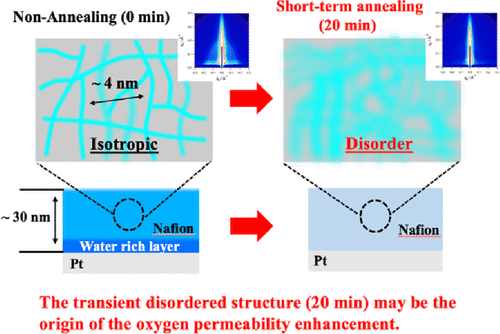
Effect of Short-Term Thermal Annealing on the Nanostructure of Nafion Thin Films on Platinum Substrates
Shigeki Uzuki *- ,
Masako Yamada - ,
Reiji Kumai - ,
Koji Michishio - ,
Kenji Yamaoka - ,
Yoshihisa Fujii - ,
Ryuji Maruyama - , and
Norifumi L. Yamada
At the cathode of a polymer electrolyte fuel cell (PEFC), protons transported from the anode react with the oxygen gas at the interface of a platinum (Pt) catalyst and an ionomer thin film, where protons pass through a water network in the thin film and the oxygen permeates through it. This implies that the conductivity and permeability of the film, which are influenced by the nanostructure of the film, can be the rate-limiting factors for the reaction. In this study, the effect of short-term thermal annealing, which imitates the industrial decal process and results in high gas permeability, on the nanostructure of a Nafion thin film coated on a Pt layer on a substrate was examined using a combination of grazing-incidence small-angle X-ray scattering (GI-SAXS), neutron reflectometry (NR), and positron annihilation spectroscopy (PAS). The structural evolution during thermal annealing was observed using GI-SAXS and NR analyses. An initial isotropic water network structure was transformed into a transient homogeneous structure after 20 min of thermal annealing, and a stable multilayered interfacial structure was induced at the interface with the Pt substrate in the out-of-plane direction after 4 h of long-term annealing. A significant increase in the free volume size of the Nafion thin film was observed using PAS analysis after 20 min of thermal annealing, which could increase the oxygen permeability a few times and one order of magnitude more than the initial state and equilibrium state after annealing, respectively. These results indicate that the transient disordered structure of the Nafion thin film may be the origin of the oxygen permeability enhancement by the decal process, in which the structure is quenched during the transformation from the initial metastable state to the final stable state during short-term thermal annealing.
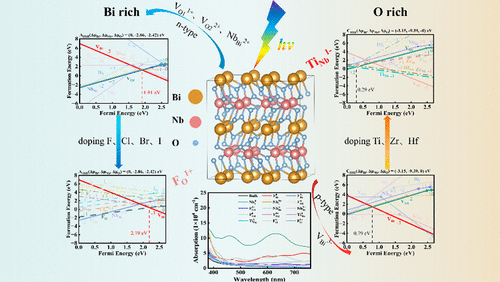
Donor and Acceptor Characteristics of Group IV and VII Doped BiNbO4: A Hybrid Density Functional Investigation
Hongchun Zheng - ,
Song Ling - ,
Bo Kong *- ,
Tixian Zeng *- ,
Shan Jiang - , and
Wentao Wang *
This work systematically investigates the intrinsic defect behavior and corresponding conductivity type of BiNbO4 under different representative thermodynamic equilibrium growth conditions using hybrid density functional theory calculations. The modulation effects of group IVB (Ti, Zr, and Hf) and group VIIA (F, Cl, Br, and I) element doping on its conductivity and electronic and optical properties are also explored. It is revealed that, under Bi-rich, relatively Nb-rich, and O-poor conditions, the easy ionization of the main native defects VO1 (two O vacancy types), NbBi, and unintentional Hi as shallow donors promotes BiNbO4 to exhibit an unintentional n-type conductivity character. Still, under O-rich, Bi-poor, and Nb-poor conditions, the ionization of the dominant defects VBi as excellent acceptors makes it present an intrinsic p-type behavior. Therefore, this affirms the experimental observation of the n-type character in BiNbO4 and predicts its p-type behavior. For extrinsic doping, the substitution of Ti on Nb (TiNb1–) significantly enhances the p-type conductivity under the presence of O-rich conditions. Ti becomes the best p-type doping candidate for BiNbO4 among the group IVB elements. In contrast, the substitution of F on O (FO1+) effectively boosts the superior n-type conductivity under the O-poor conditions, and F is the best n-type doping candidate among group VIIA elements. Furthermore, the electronic structure and optical absorption analyses indicate that the major intrinsic defects VO11+, VO12+, and VBi3– and the optimal extrinsic doping TiNb1– and FO1+ do not lead to deep-level recombination centers but instead serve as active sites for photocatalytic reactions, synergistically improving visible-light absorption and charge carrier concentration. In addition, it is found that interstitial Nbi and an O2 vacancy defects respectively induce significant visible light absorption, especially for Nbi. Thus, via the control of growth conditions and the optimization of doping elements, this study provides theoretical guidance on the intrinsic and extrinsic doping strategies for tuning the conductivity and other properties, further enhancing the photocatalytic performance of BiNbO4.
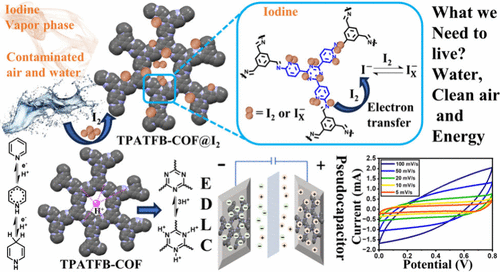
Bifunctional Covalent Organic Framework for Efficient Iodine Capture and High-Performance Energy Storage
Shubham Kumar - ,
Nakul Desai - ,
Bharatkumar Z. Dholakiya - ,
Sudhakar Narahari Yethadka *- , and
Ritambhara Jangir *
Herein, we report a nitrogen-rich triazine-based covalent organic framework (TPATFB-COF), synthesized via the condensation of 5,5′,5″-(1,3,5-triazine-2,4,6-triyl)tris(pyridin-2-amine) (TPA) and 1,3,5-triformylbenzene (TFB). The resulting TPATFB-COF, enriched with nitrogen sites, enables dual functionality in iodine capture and supercapacitor applications by facilitating strong adsorption interactions and efficient charge transport. The exceptional thermal and chemical stability of the COF is attributed to robust covalent linkages. The π-electron-rich arenes and nitrogen centers enable strong iodine adsorption, while its π-conjugated framework and redox-active sites facilitate efficient charge transport for high-performance energy storage. The exceptional iodine uptake by TPATFB-COF across various phases (5.9 g g–1: vapor, 75 °C; 2.28 g g–1: vapor, 25 °C; 1.6 g g–1: organic, 25 °C and 3.9 g g–1: aqueous, 25 °C), with excellent recyclability and retention, was observed. In energy storage, it achieves maximum specific capacitances of 455 F g–1 (GCD, 5 A g–1) and 402 F g–1 (CV, 5 mV s–1), delivering an energy density of 63.19 Wh kg–1 and a power density of 2500 W kg–1. In the symmetric configuration, it maintains 90% capacitance retention over 10,000 cycles with a high Coulombic efficiency of 95%. These results highlight TPATFB-COF as a promising material for sustainable iodine capture and high-performance energy storage. The dual functional behavior of COF offers promising avenues for addressing challenges related to nuclear waste management and the growing demand for high-performance supercapacitors.

Electrochemical Transformation of Thiol-Iodine-Based Reactions toward Multiplexed Sensing Applications for Plant-Stress Hormone and Environmental Contaminant
Mansi Gandhi - ,
Sariga - ,
Roopa Margaret Rodrigues - , and
Anitha Varghese *
Functionalized thiophenes are potential electroactive species that serve as efficient molecular electrochemical sensors. This work describes the fabrication of a 3-thiophene acetic acid (TAA)-modified screen-printed carbon electrode/multi-walled carbon nanotube (SPCE/MWCNT) platform via a facile electrochemical method in an aqueous medium. The effectual PT-Redox (product of TAA formed postpotentiostatic polarization) integration over SPCE/MWCNT was confirmed through various spectroscopic and electrochemical investigations. The SPCE/MWCNT showcased exceptional interaction with PT-Redox, creating a resilient platform for its precise binding, thereby enhancing the electrode–electrolyte electroactive region, topographic roughness, electron conductivity, host response, and comprehensive electrochemical properties. The as-prepared electrode (SPCE/MWCNT@PT-Redox) was employed for the selective detection and quantification of glutathione (GT) as well as hydrazine (HyD) in an aqueous medium. The sensor showed excellent electrocatalytic oxidation responses toward these analytes, yielding a good sensitivity of 0.32 μA mM–1, a low detection limit (DL) of 0.225 μM, a broad linear dynamic window of 0–400 μM for GT, a high sensitivity of 0.13 μA mM–1, a low DL of 0.56 μM, and a linear window of 0–350 μM for HyD, obtained via the differential pulse voltammetry (DPV) technique. This substantiates that the modification with PT-Redox significantly boosted the electrode’s interfacial activity and catalytic potential. Furthermore, the electrode exhibited robust antifouling and anti-interference traits, suggesting the composite’s enhanced stability and sensing capabilities for real-world applications. The captivating features, including excellent specificity, fast response dynamics, and simple sample preparation necessities of the proposed system, reveal a promising platform that accomplishes significant potential in futuristic sensing applications.

Homochirality- and Heterochirality-Controlled H- and J-Aggregation with Terminal Residue-Directed Supramolecular Chirality in Azobenzene-Based Amphiphilic Dipeptide Self-Assembly Systems
Shunjie Cheng - ,
Hanting Wang - ,
Minghao Wang - , and
Lukang Ji *
The transfer of molecular chirality to supramolecular chirality in multichiral systems constitutes a fundamental challenge in supramolecular chemistry. Azobenzene-based dipeptide amphiphiles containing chiral glutamic acid and alanine/glycine residues were designed to investigate this phenomenon. These chiral derivatives underwent spontaneous self-assembly in aqueous media, displaying characteristic circular dichroism (CD) responses. The supramolecular chiroptical properties showed critical dependence on the stereochemical configuration of terminal glutamic acid residues, with homochiral dipeptides assembling into elongated fibrillar structures whereas heterochiral analogs produced disordered aggregates. Combined density functional theory (DFT) calculations and molecular dynamics (MD) simulations revealed the stereochemical control mechanism underlying chirality transfer and morphological evolution. This systematic investigation provides enhanced understanding of hierarchical chirality propagation in multichiral architectures and establishes stereochemical design principles for programmable chiral nanomaterials.

Stimuli-Responsive Colloidal Gate for Active Modulation of Fluid Flow in Packed Beds
Gideon Onuh - ,
Ronit Bitton - ,
Oz M. Gazit *- , and
Ofer Manor *
This publication is Open Access under the license indicated. Learn More
We present pH-responsive colloidal paste based on brushes of poly(acrylic acid) (PAA) grafted on silica microparticles (∼4 μm) that actively modulates bulk fluid flow in packed bed columns of the colloid paste. pH-dependent charge and conformational changes of the PAA brushes in aqueous 10 mM NaNO3 solutions (pH 2–10) give an 8-fold change in flow rate (0.04–0.32 mL/min) through dynamic particle rearrangement in the paste, offering potential for flow rate modulation by pH. The colloid paste is a responsive “colloidal flow gate” that actively changes its permeability based on the aqueous solution acidity. We characterize the colloidal paste permeability by measuring pH variations of the flow rate through the paste.

Multiscale Mechanistic Insights into Green Anionic Surfactants on Chemical Mechanical Polishing Performance of C-, A-, and R-Plane Sapphire: Combined Experiments and Theoretical Calculations
Yida Zou - ,
Bin Hu - ,
Xinhuan Niu *- ,
Jiakai Zhou *- ,
Ni Zhan - ,
Jianghao Liu - ,
Xinjie Li - ,
Chao He - , and
Changxin Dong
Sapphire slurry should be developed for environmental friendliness, high efficiency, and sustainability. The effects of three green anionic surfactants (dodecyl phosphate, DAP; dodecylbenzenesulfonic acid, DBSA; ammonium dodecyl sulfate, ADS) on C-, A-, and R-plane sapphire chemical mechanical polishing (CMP) were comparatively investigated. The results show that the effects of three surfactants on sapphire slurry and CMP performance are ranked in the following order: DAP < DBSA < ADS. Among them, ADS has the best effect on C-, A-, and R-plane sapphire, with the removal rate of 4.04, 2.11, and 3.20 μm/h and the surface roughness Sq of 0.215, 0.199, and 0.218 nm, respectively. X-ray photoelectron spectroscopy and quantum chemical calculations have jointly confirmed that ADS exhibits the highest reactivity. The adsorption process of surfactants in sapphire CMP was simulated using molecular dynamics (MD), with adsorption energies of −16.5, −4.4, and −4.34 eV for ADS, DBSA, and DAP, respectively, which further indicated that ADS was most likely to react with sapphire. ADS has a dual role in the slurry: it can adsorb on the sapphire surface to improve surface quality, and the NH4+ decomposed from ADS can react with Al(OH)4– to increase the removal rate of sapphire. This work provides a new atomic-scale mechanism for a sapphire CMP.

Structure–Property Relationships in MIL-101 MOFs: Influence of Organic Ligands on CO2 Adsorption
F. Alvarez-Ramírez *- and
I. García-Cruz
Metal–organic frameworks (MOFs) of the MIL-101 type have garnered significant attention as promising materials for CO2 capture due to their high surface areas, tunable porosity, and chemical versatility. Understanding how the choice of organic ligands and metal centers affects CO2 adsorption capacity and selectivity is critical for the rational design of efficient adsorbents. This study investigates the impact of functionalizing MIL-101(Cr) frameworks with a diverse set of 21 carboxylate ligands varying in size, aromaticity, and geometry, alongside an exploration of isostructural variants incorporating different metal ions (Al, Sc, Mn, Fe, Ti, V, and Cr). Using Grand Canonical Monte Carlo simulations combined with Langmuir model fitting, we quantified adsorption capacities and affinities at 298 K, revealing that longer aromatic ligands generally enhance CO2 uptake by increasing pore volume and promoting π-CO2 interactions, while compact ligands favor stronger local affinity but lower capacity. Spatial density mapping demonstrated preferential CO2 adsorption sites near tetrahedral cavities and metal nodes, influenced by ligand chemistry. Structural and textural descriptors such as accessible surface area and pore limiting diameter were correlated with adsorption performance, highlighting trade-offs between capacity and selectivity. Among metal variants, MIL-101(Al) and MIL-101(Sc) exhibited superior simulated CO2 uptake, attributable to favorable local electronic environments as evidenced by atomic charge correlations. The findings underscore the critical role of both ligand design and metal center selection in optimizing MIL-101 frameworks for CO2 capture applications, while also acknowledging potential stability challenges associated with highly porous, ligand-extended structures.

Influence Mechanism of Crystal Phase Composition on the Fixation of Zinc by Calcium Sulfate
Yaping Zhong - ,
Can Wu *- ,
Hongli Liu - ,
Guo Liu - , and
Zhang Lin
Calcium sulfate coprecipitation is a widely adopted method for removing heavy metal ions in engineering. However, the commonly used dihydrate calcium sulfate (CaSO4·2H2O, DH) phase shows limited efficiency in removing Zn2+ ions. To enhance this process, we regulated the crystal phase of calcium sulfate and explored how its composition affects the mechanism of Zn2+ immobilization. Coprecipitation experiments indicate that as the molar fraction of hemihydrate calcium sulfate (CaSO4·0.5H2O, HH) increases in the mixture, the efficiency of Zn2+ immobilization by calcium sulfate improves. When the HH molar fraction surpasses 15.6%, the immobilization capacity levels off. Mechanistic studies show that Zn2+ is mainly immobilized through structural doping within the crystal’s water channels rather than by surface adsorption. In both DH and HH, zinc ions occupy these water channels instead of replacing Ca2+ ions. The larger water channels in HH contribute to its greater capacity for zinc ion immobilization. However, high-concentration Na+ ions interfere with HH formation, preventing further increases in zinc ion immobilization capacity and causing it to reach equilibrium. This research offers theoretical insights for optimizing calcium sulfate coprecipitation in managing heavy metal pollution in water bodies.
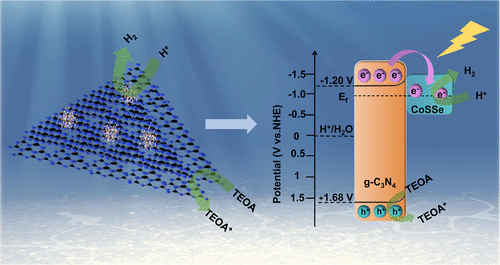
Facile Construction of CoSSe/g-C3N4 Schottky Junction for Efficient Photocatalytic H2 Evolution
Xiuxiang Zhao - ,
Zhuonan Lei - ,
Xuanqi Wang - ,
Haipeng Teng *- , and
Enzhou Liu *
In this study, the CoSSe cocatalyst was synthesized by using a sequential hydrothermal-calcination strategy, and the g-C3N4 (CN) photocatalyst was obtained through high-temperature thermal polycondensation. Subsequently, CoSSe/CN composites with varying amounts of CoSSe were fabricated using a solution-based physical drying method. The study demonstrates that the photocatalytic H2 evolution rate (rH2) of 30 wt % CoSSe/CN can reach 9675 μmol g–1 h–1, showing a boost of 93 times compared to that of bare CN. This improvement stems largely from the Schottky junction created at the interface between CoSSe and CN, which significantly enhances charge separation efficiency. In this structure, CoSSe demonstrates a charge storage capability by providing temporary storage sites for electrons (e–). Moreover, the CoSSe cocatalyst introduces more reactive sites and reduces the activation energy required for H2 production, leading to efficient and stable H2 production. This study underscores the highly prospective application value of selenosulfides in terms of the photocatalytic H2 production field.

Constructing High-Efficiency Polyoxometalate-Based Antibacterial Hydrogels for Wearable Sensors
Min Ma - ,
Chuang Li - ,
Wenhui Fan - ,
Yue Su *- ,
Dongjie Guo *- ,
Mingxue Li *- , and
Yuemin Zhou
Conductive flexible hydrogel are widely used in wearable electronics owing to its desired conductivity, flexibility, adhesion, and mechanical properties similar to human tissue. Nevertheless, conductivity and bacterial infections are always critical issues for the long-term use of hydrogel wearable sensors. Herein, a multifunctional polyoxometalate-based hydrogel with both antibacterial and sensing performances are prepared by integrating polydopamine-functionalized polyoxometalates (POMs) particles into polyacrylamide matrix. To obtain rapid gelation times (to seconds), a dual autocatalytic system focused on lignin and copper ions was formed by activating ammonium persulfate to generate free radicals and initiating the free-radical polymerization of acrylamide monomers. The fabricated POM-based hydrogel exhibited high mechanical strength (135.8 kPa), conductivity (2.52 mS/cm), and antibacterial activity against Gram-positive/negative bacterial strains Escherichia coli (E. coli, 99.39%) and Staphylococcus aureus (S. aureus, 99.42%); thus, they were utilized as wearable sensors. These sensors also exhibited high stability and repeatability during 6000 s stretching/releasing cycles; therefore, it were used to monitor the human motions of finger, wrist, and elbow. Together, this strategy not only provides approaches for designing POM-based hydrogel materials but also expands the potential application of POMs in the advanced wearable strain sensors and antibacterial field.
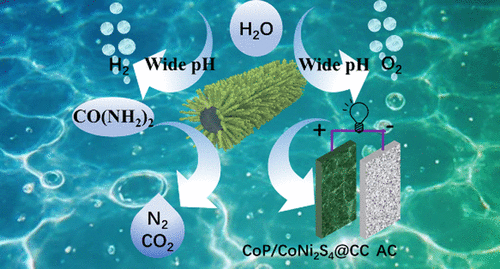
Theoretical-Interfacial Codesign Enabling a Multi-Functional Electrode for Efficient Electrocatalysis and Energy Storage
Min Zhu - ,
Xuerong Shi *- ,
Shengming Zhang - ,
Xinlei Yu - ,
Runlin Wang - ,
Peijie Wang - ,
Rujing Shang *- , and
Shusheng Xu *
The strategic design of integrated catalysts for overall water splitting, urea electrolysis, and energy storage represents an unexplored frontier with significant challenges for catalyst engineering. Inspired by theoretical predictions that CoP/CoNi2S4 composites exhibit enhanced hydrogen evolution reaction (HER) activity compared to individual components, this nanorod structure was fabricated, demonstrating exceptional HER performance across acidic, alkaline, and simulated seawater conditions. It achieved 10 mA cm–2 at overpotentials of 119 mV (acidic), 88 mV (alkaline), and 95 mV (seawater), with 100-h stability, surpassing commercial Pt/C at high current densities (200 mA cm–2) with η200 values of 232 mV (1 M KOH) and 234 mV (1 M KOH + 0.5 M NaCl). For oxygen evolution reaction (OER), it exhibited superior activity in alkaline media (η10 = 267 mV) and simulated seawater, outperforming commercial RuO2. In urea-added electrolytes, the symmetric electrolyzer required only 1.53 V to achieve 10 mA cm–2. As a hybrid supercapacitor, the assembled CoP/CoNi2S4//AC device delivered an energy density of 50.9 Wh kg–1 at 800 W kg–1 with excellent cycling stability. This TMP/TMS composite integrates electrocatalytic and energy storage functionalities, paving the way for multifunctional applications in energy conversion technologies.
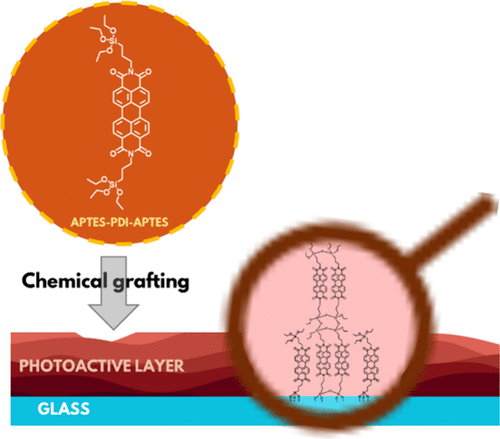
Insights into Growth of a Photoactive Layer Based on Perylene Diimide Bearing Alkoxysilane Groups
Karolina Socha - ,
Maciej Krzywiecki - ,
Patryk Mroczko - ,
Dawid Nastula - ,
Karol Erfurt - ,
Radosław Motyka *- , and
Agata Blacha-Grzechnik *
This publication is Open Access under the license indicated. Learn More
The chemical grafting of organic photosensitizers onto solid surfaces has gained increasing attention lately as an effective strategy for the formation of stable, functional layers, with promising applications in areas such as antimicrobial coatings and optoelectronic devices. In this work, a perylene diimide (PDI) derivative bearing alkoxysilane groups, APTES-PDI-APTES, was synthesized and chemically grafted onto a glass surface. The presence of two alkoxysilanes enabled competing processes to occur, i.e., condensation reactions, which led to the formation of complex multilayer structures, and covalent bonding to the glass surface. The influence of deposition process parameters, such as solvent polarity and immersion time, on the chemical structure and the morphology of the resulting layer was studied using X-ray photoelectron spectroscopy, Raman spectroscopy, and atomic force microscopy. The singlet oxygen photogeneration was tested using an indirect detection method, proving that after immobilization, APTES-PDI-APTES retains its photosensitizing properties. These findings highlight the influence of the process parameters on the development of the PDI-containing multilayer and its potential application as a heterogeneous source of singlet oxygen.
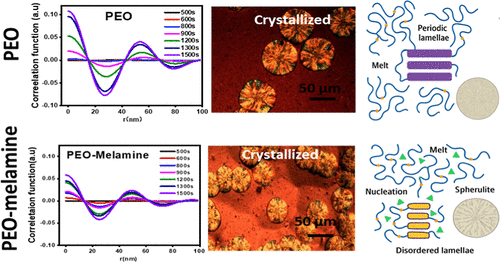
Crystallization Kinetics and Structural Perturbation in Poly(ethylene oxide)-Melamine Blends under Quiescent Isothermal Conditions
Sarmad Ali *- ,
Khurram Shehzad - ,
Mudassar Maraj - ,
Nisar Ali - ,
Xiuhong Li - ,
Nian Li *- ,
Tingyu Xu *- , and
Zhenyang Wang *
This study investigates the interfacial and crystallization behavior of poly(ethylene oxide) (PEO) and its heterogeneous blend with melamine under isothermal quiescent conditions. In situ small-angle and wide-angle X-ray scattering, complemented by optical, thermal, and rheological analyses, reveal that melamine alters the crystallization pathway of PEO. Specifically, melamine increases nucleation density and promotes the formation of denser spherulites, while simultaneously restricting chain mobility and suppressing long-range crystalline order. Interfacial interactions, most likely hydrogen bonding between melamine and the ether oxygen atoms of PEO, extend relaxation times and hinder lamellar periodicity. Despite a modest increase in crystallization temperature, the overall crystallinity decreases, highlighting the dual role of melamine as both a nucleating agent and a kinetic inhibitor. These findings provide a molecular-level framework for understanding the regulation of polymer crystallization by small organic molecules through interfacial coordination, offering insights for the design of polymer-organic hybrid systems in energy storage, coatings, and packaging applications.
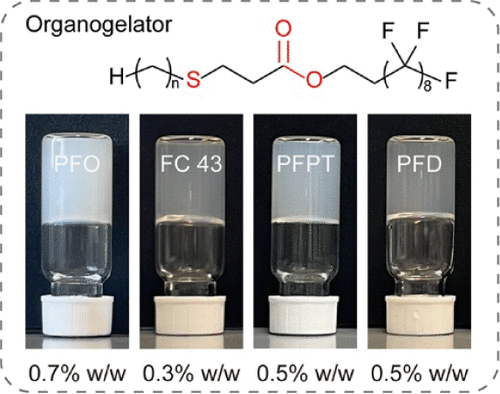
β-Thioester-Based Low-Molecular-Weight Organogelator for Highly Efficient Gelation of Perfluorocarbons
Zeying Ma - ,
Minchao Li - , and
Bing Liu *
Thermally reversible supramolecular perfluorocarbon (PFC) gels are unique soft materials and have been gaining significant interest in many applications. Developing efficient low-molecular-weight organogelators for PFCs is an attractive field, but it remains challenging. This paper reports a highly efficient β-thioester-based gelator for the gelation of PFCs. The gelators are synthesized through click chemistry, using alkyl thiols and acrylates containing perfluoroalkyl chains, and have a diblock molecular structure that incorporates thioether and ester groups. In four typical PFCs (perfluorooctane, perfluorotributylamine, perfluoropropylene trimers, and perfluorodecalin), the gel properties of β-thioester-based gelators have been studied, achieving a low critical gel concentration (CGC) of only 0.3%–0.7% w/w. Comparative studies have shown that the high gelation efficiency arises from the formation of a fine fibrous structure in which the thioether group plays a crucial role, supported by the synergistic contribution of the ester group. In addition to the functional groups, the effect of alkyl chain length is also studied, revealing that the optimal chain length is n = 12. The achieved PFC gels are thermally reversible and have a storage modulus of 103–104 Pa in the 0.5–1.5% (w/w) concentration range. As we have demonstrated, these PFC gels are promising candidate materials for constructing temperature-responsive films that can switch the transmission of visible light.

Electrocatalytic Nitrate Reduction to Ammonia Using Metal-Free Nitrogen-Doped Carbon Catalysts Derived from Setaria viridis
Yuting Qu - ,
Boran Yang - ,
Hongli Wang *- ,
Xingcheng Ma - ,
Lixin Chen - ,
Ying Gao - , and
Shicheng Sun *
ACS Editors' Choice® is a collection designed to feature scientific articles of broad public interest. Read the latest articles
Electrocatalytic reduction of nitrate to ammonia is one potential strategy to remove nitrate and produce ammonia under ambient conditions. The use of carbonaceous materials as metal-free electrocatalysts is crucial for achieving high activity and precise product selectivity. Here, we propose a facile one-step calcination strategy to produce nitrogen-doped carbon (N–C-900–3) electrocatalysts derived from Setaria viridis (SV) for highly efficient nitrate-to-ammonia reduction (NO3RR). Benefiting from the high concentration of defects and the high content of graphitic-N moiety, which play a crucial role in promoting the dissociation of water and the N–H bond formation, the resultant N–C-900–3 displays outstanding electrocatalytic performance with a maximum NH3 yield rate of 6.23 mg h–1 cm–2 and a Faradaic efficiency (FE) of ca. 97.4%.
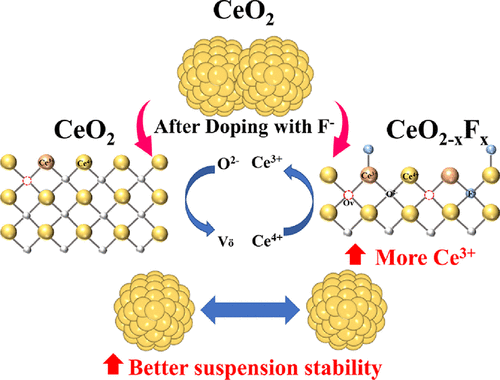
Construction of a Ce–F Bond in CeO2–xFx Nanoabrasives and Its Dispersion-Polishing Coupling Enhancement Mechanism
Ning Xu *- ,
Ziheng Gao - ,
Yu Huo - ,
Kailong Gao - ,
Yansong Wang - ,
Guosheng Zhang - ,
Yu Lin - , and
Zhuo Wang
As a commonly used abrasive in chemical mechanical polishing (CMP), CeO2 enables high-precision material removal during SiO2 polishing due to its unique chemical tooth effect. Its performance is heavily influenced by the dispersion stability of the polishing slurry and the concentration of Ce3+ in the CeO2 particles. Traditional methods that rely on dispersant additives have inherent limitations, including sensitivity to environmental factors such as pH and temperature, which can lead to dispersant failure and particle reagglomeration, thereby compromising polishing uniformity. Additionally, excessive dispersant may coat the abrasive surfaces, reducing direct contact with the workpiece and, consequently, diminishing the material removal rate (MRR) and chemical activity. To overcome these challenges, this study proposes a fluorine doping strategy that enhances both the Ce3+ concentration and dispersion stability by precisely controlling the F– doping levels in CeO2 abrasives. Experimental results show that fluorine doping significantly improves the colloidal stability, as evidenced by a reduced sedimentation rate, an increased optical absorbance, a higher zeta potential (63.1 mV), and a more uniform particle size distribution with suppressed agglomeration. These changes enhance the effective contact area between the abrasives and SiO2 substrates. Notably, at an optimal F– doping concentration of 0.05, the modified abrasives exhibited a 12.28% increase in surface oxygen vacancy density and Ce3+ concentration compared to the undoped abrasives, alongside a 31% improvement in SiO2 MRR. Furthermore, the polishing mechanism of the doped abrasives on SiO2 substrates was systematically investigated, revealing an enhanced chemical–mechanical synergy through controlled oxygen vacancy generation and optimized surface charge characteristics.
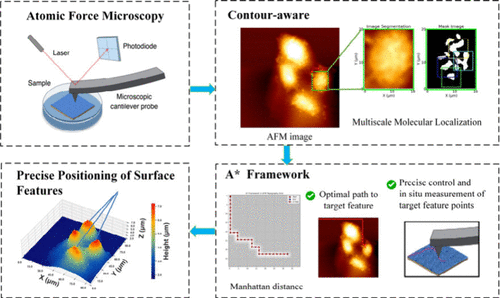
Dynamic Trajectory Planning for Atomic Force Microscopy Nanopositioning: An Enhanced A-Star Framework Addressing Displacement Errors in Nonplanar Environments
Liguo Tian - ,
Yongkun He - ,
Yang Wang - ,
Haiyue Yu - ,
Wentao Yu - ,
Baichuan Wang - ,
Lanjiao Liu - ,
Wenxiao Zhang - ,
Ying Wang - ,
Xiao Zhang - ,
Cuihua Hu - ,
Wei Ji - , and
Zuobin Wang *
The use of atomic force microscopy (AFM) for nanoscale surface characterization and mechanical property measurement has attracted considerable interest. At the level of single-molecule mechanical measurement, AFM is a powerful tool for both surface morphology analysis and mechanical assessment. However, its effectiveness is limited by dynamic displacement deviation during precise nanoscale positioning of surface target points, an essential factor in accurately determining surface mechanical properties. This study addresses this limitation by proposing an integrated enhanced A-star (A*) framework for contour-aware motion trajectory planning, ensuring nanometer-level target localization accuracy during AFM measurements on complex surface morphologies. The method employs AFM tip repositioning using prior topographic data and enables trajectory path planning on biological cell surfaces with both high and low topographical undulations. Experimental evaluations using Manhattan, Chebyshev, and Euclidean heuristic metrics in AFM grid modeling demonstrated that the Manhattan approach achieved a heuristic accuracy of 96% ± 4%, significantly outperforming Euclidean (70% ± 4%) and Chebyshev (56% ± 8%) methods (p < 0.001). In constrained environments, the Manhattan heuristic reduced target localization errors by 30% by alleviating path cost overestimation and resolved the long-standing trade-off between path smoothness (coefficient of variation, CV = 0.28) and positioning precision through adaptive cost-weighting mechanisms. The proposed approach supports precise nanoscale positioning necessary to capture ultramicroscopic topography and physical characteristics, providing a robust framework for quantitative nanomechanical characterization of heterogeneous materials.
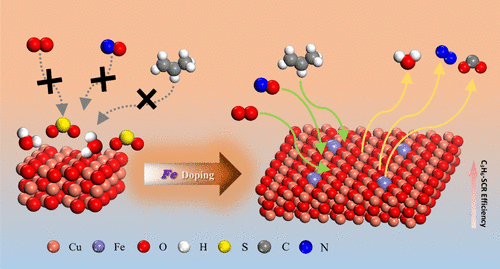
Mechanism of the Effect of Fe Doping on the CuO (111) Surface on the Reactivity and Water/Sulfur Resistance Performance of CuFe Layered Double Oxide Catalysts for Selective Catalytic Reduction of NO with C3H6: A DFT + U Study
Huqin Zheng - ,
Yaxin Su *- ,
Sameer Shahid - ,
Yuhao Wang - ,
Min Cui - ,
Mingyu Su - ,
Wenyi Deng - ,
Bingtao Zhao - , and
Jarosław Zuwała
Previous experimental research revealed the beneficial synergistic effect of Cu and Fe in CuFe-LDOs catalysts for C3H6–SCR. However, the intensive understanding of the reaction mechanism on the microscopic level remains limited. In this study, the configuration of Fe-doped CuO (111) surface was optimized, and the adsorption energy, charge distribution, projected density of states, transition state, and reaction pathway were calculated based on the density functional theory method. Results showed that doping with Fe significantly strengthened the adsorption of NO, O2, and C3H6 molecules on the catalyst surface and boosted the electronic interactions. The excellent water-sulfur resistance of Fe atoms significantly minimized the negative effects of H2O/SO2 on catalyst activity. The Cu–Fe synergistic effect reduced the energy barrier for NO oxidation from 1.08 to 0.56 eV and facilitated the formation of more stable nitrates. Meanwhile, the introduction of Fe raised the energy barrier for *H2O dehydrogenation from 0.07 to 0.46 eV, thus preserving more active sites. Furthermore, *OH adsorption on the Fe-CuO (111) surface was unstable, rendering the negative effect of H2O on the catalyst reversible. Comparing the reaction pathways of dynamic adsorption revealed that the C3H6–SCR reaction proceeded more easily when NO and O2 were saturated before C3H6 was adsorbed. The results of this study support the experimental findings and offer microscopic insights into Cu–Fe synergy in C3H6–SCR, aiding future improvements of these catalysts.
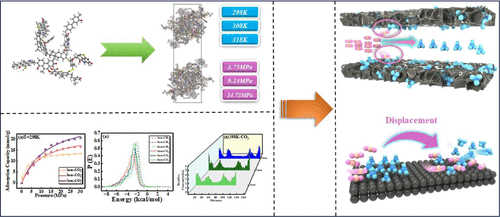
Adsorption and Diffusion Mechanism of CO2/CH4 in Bituminous Coal Pores: Molecular Dynamics Simulation of Coupling Effects of Multiple Factors on Enhanced Coalbed Methane Recovery Rate
Xiaohua Zhu *- ,
Yilin Liu - ,
Yunhai Liu *- , and
Ligao Liu
CO2-enhanced coalbed methane recovery (CO2-ECBM) improves CH4 recovery and enables geologic CO2 sequestration. Using giant canonical Monte Carlo (GCMC) and molecule dynamics (MD) simulations, this study reveals nanoscale mechanisms of CO2/CH4 adsorption–diffusion in bituminous coal under temperature–pressure coupling. The research results show that CO2 adsorption exceeds CH4 due to stronger electrostatic interactions with coal matrix. One nm pores dominate the low-pressure adsorption process, while 3/5 nm pores are conducive to high-pressure multilayer CO2 adsorption. CO2 preferentially occupies low-energy sites, with adsorption selectivity increasing with pore size/pressure. High temperatures weaken adsorption but enhance CO2 displacement efficiency in macropores. The effect of temperature and pressure coupling enhancement accelerates the diffusion of CO2/CH4, but the strong adsorption between CO2 and coal matrix hinders its migration. Due to the weak adsorption and CO2 displacement, the diffusion coefficient of CH4 is significantly higher than that of CO2. CO2 Strong adsorption, weak diffusion and CH4 weak adsorption, strong diffusion are the basis of CO2-ECBM. Strategically leveraging CO2’s adsorption advantage to displace CH4, while exploiting CH4’s rapid diffusion through macropores, optimizes methane recovery. The findings provide molecular-scale insights for designing efficient CO2-ECBM in coal seams.

Molecular Dynamics of Acid Waste Gas Replacing Oil in Hydrated Mineral Nanopores: Implications for Shale Oil Recovery and Acid Gas Storage
Lu Wang - ,
Run Zou *- ,
Yifan Yuan - ,
Yifan Zhang - ,
Liang Huang - ,
Yisheng Liu - , and
Heng Wang
Acid waste gas (H2S–CO2 mixture) has great potential in enhancing oil recovery (EOR) and achieving gas storage (GS) in water-bearing shale reservoirs. The process of oil replacement by acid gas and pure CO2 in hydrated nanopores was compared through molecular dynamics. The EOR and GS microscopic mechanisms were revealed, and the various factors that affect the acid gas-EOR and GS performances were analyzed. The results show that the water film in illite and composite nanopores promotes the migration of desorbed oil toward the middle of nanopores, while the water mass formed in kerogen nanopores retains gas and induced free oil reflux. Therefore, the acid gas-EOR performance of the former and latter are positively and negatively correlated with water content, respectively. The acid gas storage rate in illite and composite nanopores initially increases and then decreases with the increase of water content because the thickened water film increases the gas penetration resistance and gradually occupies the GS space. Only the acid gas storage stability in illite nanopores is positively correlated with water content because the water film prevents the stored gas from escaping outward and compressing the gas adsorption layer. Acid gas outperforms pure CO2 in both EOR and GS performances in illite and composite nanopores because H2S enhances the competitive adsorption and inhibits the formation of water columns on walls. The acid gas-EOR and GS performances in hydrated nanopores are positively correlated with the H2S proportion and buried depth. Although an increase in the nanopore diameter improves the oil recovery, it weakens the GS performance of acid gas. This study provides a theoretical basis for the implementation and optimization of acid gas injection in hydrated shale oil reservoirs.

Temperature-Dependent Dynamic Friction of a Single Human Corneocyte at Nanoscale Contact: An Analytical Approach and Direct Measurement Using Lateral Force Microscopy for Haptic Design Insight
Perawat Boonpuek *- ,
Thanathip Boonmatoon - , and
Jonathan R. Felts
This publication is Open Access under the license indicated. Learn More
Dynamic friction between human skin and a haptic surface is known to increase with the substrate temperature. When the substrate temperature reaches 60 °C, prolonged sliding contact for just 1 min can cause damage to the outer skin surface. However, the effect of the temperature on dynamic friction at the nanoscale interface between human skin cells and a nanosized probe remains inadequately quantified. This paper presents direct measurements of the dynamic friction of human skin corneocytes as a function of the AFM nanoprobe temperature, using atomic force microscopy (AFM). The lateral force microscopy (LFM) technique with a heated tip was employed to quantify the dynamic friction force of the corneocytes under a constant normal load of 10 nN and standard relative humidity (40% RH). The temperature of the heated tip was calibrated before each LFM experiment. The friction force, derived from the LFM signal data during each temperature scan, was compared to dynamic friction calculations based on a proposed analytical model. The results show that dynamic friction increases exponentially with sliding velocity as the temperature of the heated AFM tip rises, but the rate of increase slows at very high sliding velocities. These findings suggest that, in the nanoscopic contact regime at low haptic substrate temperatures (<35 °C) and normal humidity (40% RH), friction could significantly increase with increasing sliding velocities. In contrast, at higher temperatures (>35 °C) with faster sliding velocities, the finger may experience lubrication from skin hydration.
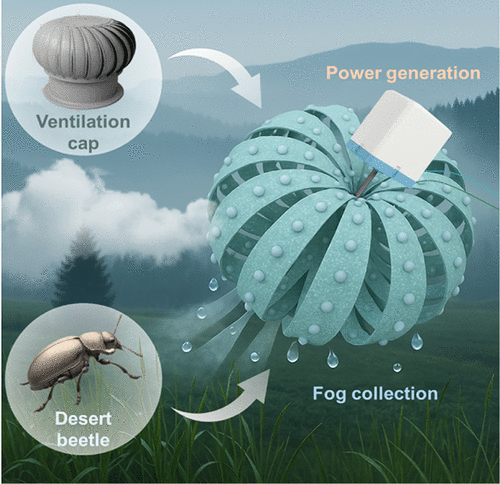
Biomimetic Ventilation Cap Structure Enables Passive Water Collection and Stable Power Generation
Junhao Liu - ,
Congji Zhang - ,
Tianze Zhang - ,
Guopeng Chen - ,
Yanshu Zhong - ,
Fengxiang Chen *- ,
Shangzhen Xie *- , and
Zhiguang Guo *
This study aimed to address the global challenges of freshwater scarcity and energy shortage by developing a bioinspired and multifunctional fog collector. Drawing inspiration from the architecture of unpowered ventilation caps and the surface structure of desert beetles, the researchers engineered a three-dimensional device featuring a wettability gradient. This advanced structure featured a 360° array of blades composed of superhydrophobic surfaces and superhydrophilic regions, enabling omnidirectional fog collection while simultaneously utilizing rotational motion to generate electricity. Experimental evaluations demonstrated that the device achieved a fog collection efficiency of 7620 mg cm–2 h–1 under conditions of 2.6 m/s wind speed and 100% relative humidity. Additionally, the system generated a stable electrical output of 0.6 V and 5 mA at a wind speed of 7 mph. The fabrication process involved laser scanning and chemical modification to establish superhydrophobic and superhydrophilic regions, thereby creating a wettability gradient. This configuration, in conjunction with centrifugal forces, facilitated the effective capture and directed transport of water droplets. The device also exhibited robust durability across repeated experimental trials, underscoring its suitability for prolonged use. This work presented a novel design paradigm for fog collection systems and offered promising applications in mitigating both water and energy scarcity.
Monolayer InISe: A Promising Thermoelectric Material with an Ultralow Phonon Thermal Conductivity and a High Power Factor
Qing-Yi Feng - ,
Zhe Wang - ,
Xia Xiang - ,
Sean Li - ,
Xiao-Tao Zu - ,
Bo Li - ,
Cai-Zheng Wang *- ,
Hong-Xiang Deng *- , and
Si-Zhao Huang *
This work reports the novel thermoelectric (TE) material InISe, which has a FeOCl-type monolayer structure. The crystal and dynamic stabilities, electronic and phonon structures, and transport properties of InISe are systematically investigated by density functional theory and Boltzmann transport theory. The study of phonon spectra and free energy fluctuation shows that InISe has excellent mechanical and thermal stability, which are important in practical applications. The calculated electronic structure shows that near the Fermi level, the conduction band of InISe exhibits two energy valleys with very close energy values. This valley degeneracy phenomenon enhances the Seebeck coefficient without obviously reducing the electrical conductivity and is beneficial for the power factor. We found that the power factor of n-type InISe can reach 27.56 mW·m–1·K–2. Remarkably, the study of crystal transport properties shows that InISe has an ultralow phonon thermal conductivity, and the value is only 0.6 W/mK at 300 K, which is valuable for thermoelectric conversion. We found that the ultralow phonon thermal conductivity of InISe is attributed to the short phonon lifetime (<102 ps), which is the result of its FeOCl-type crystal structure. Our results show that n-type monolayer InISe is a promising candidate as a TE material.

Walnut-Like CoS2/Carbon Freestanding Cathode: Enabling Dynamic Regulation of Polysulfide Diffusion–Conversion in Lithium–Sulfur Batteries
Ling Chen - ,
Peng Xu *- ,
Yiyang Chen - ,
Dechao Chen - , and
Jiao-Jing Shao *
Lithium–sulfur (Li–S) batteries are considered a promising next-generation energy storage system owing to their exceptionally high theoretical energy density. However, their practical deployment is hindered by the severe polysulfide shuttle effect arising from the dissolution and migration of lithium polysulfides. Herein, we report the rational design of a hierarchical freestanding cathode architecture consisting of carbon nanotube-embedded porous carbon nanofibers (PCNF/T) decorated with in situ grown CoS2 nanocrystals (PCNF/T@CoS2). This integrated structure is fabricated via electrospinning, high-temperature carbonization, and a subsequent hydrothermal process. Uniquely, the flexible, binder-free cathode directly replaces traditional aluminum foil current collectors. The 3D conductive framework not only accommodates volume fluctuations during cycling but also enables efficient charge transport. Meanwhile, CoS2 nanocrystals synergistically suppress polysulfide migration via a combination of strong chemical anchoring and physical confinement, as confirmed by density functional theory (DFT) calculations. Benefiting from this multiscale structural and chemical synergy, the optimized cathode exhibits a high initial capacity of 955.5 mAh g–1 at 1 C with an ultralow capacity fading rate of 0.07% per cycle over 310 cycles. Even at a high sulfur loading of 6.7 mg cm–2, the battery can still retain a capacity of 521 mAh g–1 after 100 cycles. This work offers a viable strategy toward the development of high-loading, long-life Li–S battery cathodes.
Induced Circular and Linear Dichroism of Achiral Cationic Dye Molecules Adsorbed on TEMPO-Oxidized Cellulose Nanofibers in Aqueous Suspensions
Hitoshi Watarai *
Chiroptical properties of cellulose nanofibers (CNFs), especially optical chirality-inducing abilities, are crucial subjects to be explored for the development of CNF as a new class of optical materials. This study investigated experimentally the induced circular dichroism (CD) of the positively charged achiral dye molecules thioflavin T (ThT), toluidine blue O (TBO), and methylene blue (MB), adsorbed on 2,2,6,6-tetrametylpiperidine (TEMPO)-oxidized CNFs in aqueous suspensions. The achiral dyes exhibited CD spectra of the sigmoidal positive exciton-coupling shape, suggesting the effect of the right-handed helicity of the fiber. The observed CD intensities were proportional to the concentrations of the bound dye molecules. The TBO–CNF systems could be used for a reproducible titration method of the total amount of carboxylate ions on the surface of CNF, which gave a value of 2.0 ± 0.3 mmol/g. The Langmuir isotherm was applicable to the adsorption equilibrium between CNFs and the dye molecules, though a stacking effect was suggested in ThT systems, and a dimerization effect was found in MB systems. Furthermore, it was discovered that sweeping the dye-adsorbed 0.2% CNF suspension by a Cu wire in a 1 mm optical cell induced the positive and negative linear dichroism (LD) spectra of the dye molecules, depending on the vertical and horizontal sweeping directions, respectively. The hydrodynamic orientation of CNFs by the “wire-sweeping” was confirmed by the measurement of the linear birefringence (LB) of CNFs. A comparison between the angular dependences of the LD of the dye and the LB of the CNF in an identical sample suggested that the averaged angle between the electron transition moment of the adsorbed dye molecule and the long axis of the fiber was between 45° and 135°.
Effect of Oxidation on the Floatability and Surface Properties of Galena
Ziyuan Liu - ,
Shujuan Dai *- ,
Pengcheng Li - ,
Lixin Dong - ,
Fangyuan Yang - , and
Jingchun Luan
A galena sample from Hunan, China, was selected as the research object. The pH of the pulp and the dosage of the collector were optimized for the flotation of galena, utilizing butyl xanthate and butyl ammonium dibuthyl dithiophosphate as the collectors. The alteration in the floatability of galena was also examined post-oxidation in different solutions. The influence mechanism of oxidation on the surface properties of galena was studied using a range of analytical techniques, including contact angle measurement, Fourier transform infrared spectroscopy (FT-IR) analysis, ζ potential detection, X-ray photoelectron spectroscopy (XPS) analysis, and scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDS) analysis. The galena recovery is found to decrease in proportion to both an increase in oxidation time and an increase in oxidant concentration. Oxidation exerts a greater detrimental impact on recovery when butyl xanthate is used compared to ammonium dibutyl dithiophosphate. Contact angle and FT-IR analyses and ζ potential indicate that intensified oxidation suppresses the natural floatability of galena and that chemical adsorption and a weak physical adsorption interaction occur between galena and both collectors. XPS analysis demonstrates that oxidation increases the content of high-valence sulfur species and generates elemental sulfur during the oxidation process. SEM-EDS analysis indicates that oxidation diminishes the adsorption of both collectors on the galena surface. These findings provide theoretical insights into the effects of oxidation on galena floatability and surface characteristics, thereby offering support for the flotation separation technology of oxidized sulfide minerals.

Bioinspired Mossene Membrane Integrating Sulfhydryl-Modified MOFs for Efficient Solar-Driven Seawater Desalination
Zhixi Wu - ,
Zhitao He - ,
Wanxiu Zhao - ,
Md Hobaib - ,
Yi Zhou - ,
Zhao Ding - ,
Hongxiang Chen - , and
Yang Zhou *
A fundamental challenge in the design of solar evaporators is balancing the conflicting requirements of salt rejection and high evaporation efficiency. Consequently, bioinspired membrane design has emerged as a promising strategy for enhancing solar-driven interfacial water evaporation and desalination. Here, we report a bioinspired asymmetric photothermal membrane that is denoted as Mossene. Its design is inspired by the ecological water-regulation strategies of niche bryophytes. This Mossene membrane integrates dual-layer functionality: a hydrophilic substrate for water transport and retention and a hydrophobic top layer for light absorption and floatation. The lower hydrophilic layer is fabricated by electrospinning polyamide-6 blended with a sulfhydrylated UiO-66 metal–organic framework. This structure enables rapid water uptake, storage, and sustained molecular transport, emulating the function of water-storage cells in bryophyte leaves. The upper hydrophobic layer, composed of multiwalled carbon nanotubes and polyvinylidene fluoride, replicates the surface hydrophobicity and chlorophyll-mimetic light-harvesting characteristics of moss leaves. This design ensures efficient solar absorption and thermal confinement. Under 1 kW m–2 irradiation, the dry surface of Mossene heats from 16 to 109.9 °C within 6 min. When floated on 3.5 wt % NaCl solution, the membrane reaches 78.5 °C in 5 min, demonstrating excellent photothermal conversion. The optimized Mossene membrane (MCM-6.5–0.75) achieves an outstanding water evaporation rate of 1.55 kg m–2 h–1 and an energy conversion efficiency of 97.5%. This study introduces Mossene as a biomimetic photothermal membrane that integrates hierarchical structure, selective wettability, and efficient energy utilization and underscores the potential of next-generation solar evaporators for practical implementation in sustainable water purification.

Biointerface Strategies for COVID-19 Spike Protein Detection: A Surface Engineering Approach
Mehak - ,
Chinmaya Panda - ,
Rushikesh Fopase - ,
Hiroki Kurita - ,
Fumio Narita - , and
Lalit M. Pandey *
Rapid diagnosis or health monitoring biosensors have been developed with the advent of technology. Silicon and metal oxides are used as the base material for these biosensors as a point-of-care unit. However, surface modification is needed to introduce functional groups for anchoring the bioreceptor. This study aims to explore the differences in the spike protein binding efficiency directly on the bare amine self-assembled monolayers (SAMs) and further cross-linked with the N-ethyl-N′-(3-(dimethylamino)propyl)carbodiimide (EDC)/N-hydroxysuccinimide (NHS) and glutaraldehyde (GA). The specificity of binding was improvised by attaching a primary antibody to the modified surfaces. The functional groups, morphology, and wettability of the engineered surface were characterized using various analytical techniques. As depicted by fluorescence imaging, the spike protein was explicitly bound to the designed surfaces, while albumin was a negative control. The surface roughness after the attachment of spike protein varied as 13.64 nm (amine) > 3.81 nm (glutaraldehyde) > 1.91 nm (EDC-NHS). The EDC-NHS modified surface showed a higher and uniform surface coverage with the lowest roughness among all of the surfaces. The maximum N/C ratio, calculated from XPS data, was 0.16 for the EDC-NHS surface, i.e., twice that of the bare amine surface. The wettability of the EDC-NHS surface after protein binding was also found unaltered compared to the other two chemistries employed. The EDC-NHS surface resulted in a contact angle (CA) of ∼57°, which is close to that of the native spike protein (CA = 58°). While CA significantly reduced to 49° and 39° in the case of amine and glutaraldehyde surfaces, respectively. Most importantly, the EDC-NHS surface retained the native-like structure of the spike protein, which is crucial for the accurate sensing of infections and other related biomedical applications.

Simultaneous, Non-Contact Measurement of Liquid and Interfacial Thermal Properties via a Differential Square-Pulsed Source Method
Tao Chen - and
Puqing Jiang *
Accurate characterization of heat transport across solid–liquid interfaces is essential for thermal management in micro- and nanoscale systems. Yet existing techniques often require prior knowledge of liquid properties, which complicates the simultaneous resolution of interfacial and bulk behaviors, and lose sensitivity once interfacial conductance exceeds 100 MW m–2 K–1. Here we present a differential square-pulsed source (DSPS) method that provides simultaneous, noncontact measurement of liquid thermal conductivity, volumetric heat capacity, and solid–liquid interfacial conductance without any predefined liquid parameters. Dual-frequency excitation combined with in situ substrate referencing enables property extraction from multilayer structures, and numerical simulations show a typical uncertainty of about 8% in interfacial conductance, confirming robustness. The protocol is validated for a wide spectrum of liquids, including oils, lubricants, aqueous electrolytes, and pure water, with excellent agreement with literature values for bulk properties. Analysis of the data set clarifies how vibrational-spectrum mismatch, ionic layering, and related interfacial phenomena govern heat transfer, and demonstrates that oleophilic hexadecyl silane modification of aluminum increases interfacial conductance by a factor of 16. The results reveal that conductance can be strongly tuned through surface wettability and chemical functionalization, offering direct guidelines for interface engineering. Because the approach is readily extendable to soft materials such as thermal-interface gels, it promises broad applicability in emerging interface-dominated thermal technologies.
The Conserved Fusion Peptide Sequence of SARS Coronaviruses Kinetically Promotes the Fusion Pore Opening
Priyanka Mahapatra - ,
Smruti Mishra - , and
Hirak Chakraborty *
Enveloped viruses enter the cell through the fusion of the viral envelope with the host membrane. These viruses either fuse at the plasma membrane or the endosome after entering the cell through endocytosis. Severe acute respiratory syndrome coronaviruses (SARS-CoVs) fuse at the plasma membrane by harnessing the capability of the spike (S) protein to fuse membranes. The binding of the S1 subunit to angiotensin-converting enzyme 2 (ACE2), the cell surface receptor, leads to the proteolytic cleavage at the S2′ site, which exposes the N-terminal fusion peptide (FP) and induces fusion. In this work, we have evaluated the effect of the conserved fusion peptide sequence of SARS-CoVs (FP) in polyethylene glycol (PEG)-induced fusion of small unilamellar vesicles (SUVs) in different lipid compositions. The analysis of kinetic data of fusion observables such as lipid mixing (LM), content mixing (CM), and content leakage (CL) in a one-intermediate fusion model provides a detailed insight into the mechanistic role of FP in membrane fusion. We have further shown that the peptide binding does not alter the hydrodynamic diameters, ζ and dipole potentials of membranes with varying lipid composition. To understand the plausible role of FP in inducing membrane fusion, we have examined the effect of peptide binding on the depth-dependent membrane ordering and polarity using steady-state and time-resolved fluorescence spectroscopy. Evaluation of kinetic data reveals that the FP promotes the rate constant of pore formation without affecting its extent, making the fusion reaction kinetically favorable. Overall, our work provides a mechanistic understanding of the role of the conserved fusion peptide sequence of SARS-CoVs in membrane fusion.

Unveiling a Rheological Response and Fractal Dimension with Micellization in a Zwitterionic-Anionic Mixed Surfactant System
Mayursing Girase - ,
Panchami Patel - ,
Arup Kumar Ghosh - ,
Prachi Thareja - ,
Jitendra Mata - ,
Ketan Kuperkar *- , and
Pratap Bahadur
This study uniquely reveals the hierarchical self-assembly and complex micellar transitions in a selective zwitterionic-anionic mix surfactant system in aqueous solution and the presence of varied salts. A combination of tensiometry, rheology, and small-angle neutron scattering (SANS) experiments was used to investigate these nanoscale transitions in a selective mixed micellar system. The critical micelle concentration (CMC) for single and mixed systems was determined at ambient temperature, and the interaction parameter (β), depicting favorable synergism at a specific mole fraction, was calculated using Rubingh’s regular solution theory (RST). Again, various thermodynamic parameters, such as Maeda’s free energy of micellization (ΔGMaeda), the free energy of micellization (ΔGM), and the free energy of adsorption (ΔGads), were calculated. Density functional theory (DFT) calculations were carried out employing the Gaussian 09W software package and visualized using Gauss View 6.0, which yielded varied quantum chemical descriptors that supported the marked influence of the anionic surfactant in favoring the micellization of the zwitterionic surfactant in the examined system. Also, the obtained reduced density gradient (RDG) and noncovalent interactions (NCI) enabled the understanding of the synergistic interactions involved therein, which is affirmed by Two-Dimensional Nuclear Overhauser Enhancement Spectroscopy (2D-NOESY) that explored the self-assembly mechanism in the examined micellar solution. The rheological parameters, such as the modulus of elasticity (G′) and modulus of viscosity (G″), described varied solution behavior covering fluidic, viscous, and gel formation, thus attributing viscoelasticity, particularly in the presence of salts. The SANS approach inferred micellar growth with various morphology transitions ranging from spheres, ellipsoids, and cylinders. Interestingly, it also displayed a fractal dimension (Dm) in the examined system, not previously reported for this class of surfactant mixtures, making this study very novel, giving an account of the specific binding of anions and cations from the added electrolyte.
Additions and Corrections
Correction to “Directly and Continuously Hydration of Higher Olefins to Higher Alcohols over Amphiphilic Hollow HZSM-5 Zeolite Catalyst”
Zhikuan Lin - ,
Haijun Guo - ,
Yifan Ma - ,
Hairong Zhang - ,
Lian Xiong *- ,
Jian Li - ,
Fen Peng - ,
Mengkun Wang - ,
Yuxuan Liu - , and
Xinde Chen *
This publication is free to access through this site. Learn More

Correction to “Fe(II) Induced Porphyrin Nanoaggregates Assembled in the Liquid–Liquid Interface with Dual Enzyme-like Activity for Colorimetric Determination of Methimazole”
Zhiwei Wang - ,
Yuqi Jia - ,
Xiajuan Wang - ,
Yanhong Liu - , and
Qingyun Liu *
This publication is free to access through this site. Learn More
Mastheads
Issue Editorial Masthead
This publication is free to access through this site. Learn More
Issue Publication Information
This publication is free to access through this site. Learn More
