Polystyrene Microplastics and Bisphenol A Exposure Worsen Intestinal Injury in Diabetic Mice by Disrupting Gut Microbiota and MetabolitesClick to copy article linkArticle link copied!
- Ying ZhangYing ZhangKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Ying Zhang
- Qiyao NongQiyao NongKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Qiyao Nong
- Yuanyuan ZhangYuanyuan ZhangKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Yuanyuan Zhang
- Fanfei MengFanfei MengKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Fanfei Meng
- Xinyuan HaoXinyuan HaoKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Xinyuan Hao
- Yuan TianYuan TianKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaMore by Yuan Tian
- Zunjian ZhangZunjian ZhangKey Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Zunjian Zhang
- Fengguo Xu*Fengguo Xu*Email: [email protected]. Tel.: +86-25-83271021. Fax: +86-25-83271021.Key Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaState Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P. R. ChinaMore by Fengguo Xu
- Pei Zhang*Pei Zhang*Email: [email protected]. Tel.: +86-25-83271021. Fax: +86-25-83271021.Key Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education), Nanjing 210009, P. R. ChinaAffiliated Jiangning Chinese Medicine Hospital, China Pharmaceutical University, Nanjing 211100, P. R. ChinaNanjing Jiangning Hospital of Chinese Medicine, Nanjing 211100, P. R. ChinaMore by Pei Zhang
Abstract
Environmental pollutants can induce multiorgan damage, with the digestive tract particularly susceptible. Diabetic enteropathy is a significant complication of type 2 diabetes mellitus (T2D). However, the relationship between environmental pollutant exposure and T2D-associated intestinal injury has not been previously explored. In this study, T2D mice were subjected to polystyrene microplastics (PS-MPs, 100 μg/day, 3 weeks) and bisphenol A (BPA, 100 μg/kg/day, 2 weeks). Metabolomics and 16S rRNA sequencing were used to detect changes in colonic metabolites and gut microbial composition. Caco-2 cells were utilized to investigate the functions of the altered metabolites. Compared to the T2D group, mice exposed to PS-MPs and BPA exhibited shorter colon length and reduced levels of gut barrier proteins ZO-1 and Occludin. Metabolomics analysis revealed that PS-MPs primarily affected colonic long-chain fatty acids (LCFAs) and adenosine metabolism, while BPA disrupted α-ketoisovaleric acid (KIVA) and pyruvic acid (PyrA) homeostasis. Moreover, PS-MPs exposure altered the abundance of Duncaniella and Olsenella, while BPA primarily affected Phocaeicola, Olsenella, and Variovorax. In vitro experiments showed that palmitoleic acid (C16:1), γ-linolenic acid (C18:3), adenosine (Ado), and KIVA promoted the expression of ZO-1 in Caco-2 cells. Our findings provide valuable insights into the impact of environmental pollutants on intestinal injury in T2D, underscoring the importance of environmental contaminant management, particularly in susceptible populations.
This publication is licensed for personal use by The American Chemical Society.
1. Introduction
2. Experimental Procedures
2.1. Chemicals and Reagents
2.2. Cell Culture and Treatment
2.3. Animal Experiments and Dose Selection
2.3.1. Dose Selection
2.3.2. Animal Experiment
2.4. Western Blot Analysis
2.5. Metabolomics Analysis
2.6. 16S rRNA Gene Sequencing
2.7. Histopathologic Analysis
2.8. Statistical Analysis
3. Results
3.1. T2D Mice Exhibit Mild Gut Injury
Figure 1
Figure 1. T2D mice exhibit mild gut injury. (A) FBG levels and (B) colon length in T2D and control groups (n = 10). (C) Correlation analysis between FBG and colon length. (D) Correlation analysis among FBG, colon length, body weight, and organ coefficients. (E) Protein expression levels of ZO-1 and occludin in the colon (n = 6). (F) Representative images of H&E and PAS staining of colon tissues (n = 3) (100×). Significance levels are indicated as ns (not significant), **p < 0.01, ****p < 0.0001.
3.2. PS-MPs and BPA Exposures Promote Intestinal Injury in T2D Mice
Figure 2
Figure 2. PS-MPs and BPA exposures promote intestinal injury in T2D mice. (A) PS-MPs confirmation by Fourier-transform infrared spectroscopy. (B) Scanning electron microscopy of PS-MPs. (C) Zeta potentials of the PS-MPs. Colon length of (D) PS-MPs (n ≥ 7) and (E) BPA group (n = 6). (F) Representative images of H&E and PAS staining of colon tissues (n = 3) (100×, yellow arrows indicate reduced goblet cells and mucus secretion). Intestinal ZO-1 and occludin expression of (G) PS-MPs and (H) BPA group (n = 6). Significance levels are indicated as ns (not significant), *p < 0.05, **p < 0.01, and ****p < 0.0001.
3.3. PS-MPs and BPA Exposures Alter Gut Microbiota in T2D Mice
Figure 3
Figure 3. PS-MPs and BPA exposures alter gut microbiota in T2D mice (n = 3). (A–C) Alpha diversity indices of the T2D, PS-MPs, and BPA groups. (D–F) PCoA analysis of the T2D, PS-MPs, and BPA groups. Linear discriminant analysis effective size (LEfSe) identifying differentially abundant taxa at the genus level between (G) PS-MPs-C and PS-MPs, and (H) BPA-C and BPA groups. Significance determined with a linear discriminant analysis (LDA) score (log 10) > 2. **p < 0.01.
3.4. PS-MPs and BPA Exposures Induce Colonic Metabolic Dysregulations in T2D Mice
Figure 4
Figure 4. PS-MPs and BPA exposures induce gut metabolic dysregulations in T2D mice. (A) PCA analysis based on LC-MS metabolomics data. (B, E, H) Pie chart showing the categories of differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively. (C, F, I) Volcano plots illustrating differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively. (D, G, J) Pathway enrichment analysis of differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively.
3.5. Intestinal Metabolic Dysregulation Is Linked to Gut Damage in T2D Mice with PS-MPs Exposure
Figure 5
Figure 5. Intestinal metabolic dysregulation is linked to intestinal injury in T2D mice with PS-MPs exposure. Intestinal concentration of C14 (A), C15 (B), C16:1 (C), C18:3 (D), and Ado (E) in T2D (n = 10), PS-MPs (n ≥ 7), and their corresponding control. (F) Heatmap of differential metabolites in T2D (n = 10) and PS-MPs (n ≥ 7). (G) Correlation analysis between FBG, colon length, body weight, and differential metabolites in PS-MPs. Intestinal concentration of KIVA (H) and PyrA (I) in T2D (n = 10), BPA (n = 6), and their corresponding control. (J) Heatmap of differential metabolites in T2D (n = 10) and BPA (n = 6). (K) Correlation analysis between FBG, colon length, body weight, and differential metabolites in BPA.
3.6. Altered Metabolites Promote the Expression of Gut Barrier Proteins In Vitro
Figure 6
Figure 6. Altered metabolites promote the expression of gut barrier proteins (n = 3). (A) Protein expression level of ZO-1 and occludin with 20 μM (A) and 100 μM (B) C14, C15, C16:1, C18:3, and Ado treatment. Protein expression level of ZO-1 and occludin with 0.1 μM (C), 1 μM (D), and 10 μM (E) KIVA and PyrA treatment. Significance levels are indicated as *p < 0.05 and **p < 0.01.
3.7. Gut Microbiota Dysbiosis Is Closely Correlated to Colonic Metabolic Alterations
Figure 7
Figure 7. Correlation between gut microbiota dysbiosis and colonic metabolic alterations. (A) Correlation analysis of FBG, colon length, body weight, Duncaniella, Olsenella, and differential metabolites in the PS-MPs group. (B) Correlation between colon length and Duncaniella. (C) Correlation between C16:1 and Duncaniella. (D) Correlation between C18:3 and Duncaniella. (E) Correlation between Ado and Duncaniella. (F) Correlation between colon length and Olsenella. (G) Correlation between C16:1 and Olsenella. (H) Correlation between C18:3 and Olsenella. (I) Correlation between Ado and Olsenella. (J) Correlation analysis of FBG, colon length, body weight, Variovorax, Phocaeicola, Olsenella, and differential metabolites in BPA. Correlation analyses were performed using Pearson’s method.
4. Discussion
5. Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.chemrestox.5c00359.
Figure S1. Colonic inflammatory factors expression in T2D mice. Figure S2. Organ coefficient and FBG in T2D mice with PS-MPs/BPA exposure. Figure S3. Colonic inflammatory factors expression in T2D mice with PS-MPs/BPA exposure. Figure S4. Colonic protein expression in T2D mice with PS-MPs/BPA exposure. Figure S5. Linear discriminant analysis effect size (LEfSe) comparing the Control and T2D groups. Figure S6. Metabolomics analysis in T2D mice with PS-MPs/BPA exposure. Table S1. Metabolite retention time and MRM parameters. Table S2. Intestinal differential metabolites between groups (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
The authors would like to thank Qiang Wang, Meiyu Gao, and Qinwen Xiao from China Pharmaceutical University (Nanjing, China) for their help with data analysis, instrumental operation, and sample collection.
| T2D | type 2 diabetes |
| LCFAs | long-chain fatty acids |
| KIVA | α-ketoisovaleric acid |
| PyrA | pyruvic acid |
| C16:1 | palmitoleic acid |
| MPs | microplastics |
| BPA | bisphenol A |
| PS-MPs | polystyrene microplastics |
| ACN | acetonitrile |
| MeOH | methanol |
| C14 | myristic acid |
| C15 | pentadecanoic acid |
| C18:3 | γ-linolenic acid |
| Ado | adenosine |
| SPF | specific pathogen free |
| FBG | fast blood glucose |
| PVDF | polyvinylidene difluoride |
| HRP | horseradish peroxidase |
| ECL | enhanced chemiluminescence |
| OTUs | operational taxonomic units |
| H&E | hematoxylin and eosin |
| PAS | periodic acid-schiff stain |
| FTIR | Fourier-transform infrared spectroscopy |
| SEM | scanning electron microscopy |
| DLC | dynamic light scattering |
| PS | polystyrene |
| PDI | polydispersion index |
References
This article references 49 other publications.
- 1Carpenter, E. J.; Smith, K. L., Jr Plastics on the Sargasso sea surface. Science 1972, 175 (4027), 1240– 1241, DOI: 10.1126/science.175.4027.1240Google ScholarThere is no corresponding record for this reference.
- 2Wright, S. L.; Ulke, J.; Font, A.; Chan, K. L. A.; Kelly, F. J. Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environ. Int. 2020, 136, 105411 DOI: 10.1016/j.envint.2019.105411Google ScholarThere is no corresponding record for this reference.
- 3Golwala, H.; Zhang, X.; Iskander, S. M.; Smith, A. L. Solid waste: An overlooked source of microplastics to the environment. Sci. Total Environ. 2021, 769, 144581 DOI: 10.1016/j.scitotenv.2020.144581Google ScholarThere is no corresponding record for this reference.
- 4Thompson, R. C.; Courtene-Jones, W.; Boucher, J.; Pahl, S.; Raubenheimer, K.; Koelmans, A. A. Twenty years of microplastic pollution research-what have we learned?. Science 2024, 386 (6720), eadl2746 DOI: 10.1126/science.adl2746Google ScholarThere is no corresponding record for this reference.
- 5Hernandez, L. M.; Xu, E. G.; Larsson, H. C. E.; Tahara, R.; Maisuria, V. B.; Tufenkji, N. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 2019, 53 (21), 12300– 12310, DOI: 10.1021/acs.est.9b02540Google ScholarThere is no corresponding record for this reference.
- 6Hussain, K. A.; Romanova, S.; Okur, I.; Zhang, D.; Kuebler, J.; Huang, X.; Wang, B.; Fernandez-Ballester, L.; Lu, Y.; Schubert, M.; Li, Y. Assessing the Release of Microplastics and Nanoplastics from Plastic Containers and Reusable Food Pouches: Implications for Human Health. Environ. Sci. Technol. 2023, 57 (26), 9782– 9792, DOI: 10.1021/acs.est.3c01942Google ScholarThere is no corresponding record for this reference.
- 7Wu, P.; Fan, Y.; Zhang, X.; Wu, W.; Zhang, Z.; Wu, Y.; Wang, J.; Xu, J.; Chen, T.; Gao, B. Seasonal dynamics, tidal influences, and anthropogenic impacts on microplastic distribution in the Yangtze River estuary: A comprehensive characterization and comparative analysis. J. Hazard Mater. 2024, 476, 135167 DOI: 10.1016/j.jhazmat.2024.135167Google ScholarThere is no corresponding record for this reference.
- 8Zhang, Z.; Chen, W.; Chan, H.; Peng, J.; Zhu, P.; Li, J.; Jiang, X.; Zhang, Z.; Wang, Y.; Tan, Z.; Peng, Y.; Zhang, S.; Lin, K.; Yung, K. K. Polystyrene microplastics induce size-dependent multi-organ damage in mice: Insights into gut microbiota and fecal metabolites. J. Hazard Mater. 2024, 461, 132503 DOI: 10.1016/j.jhazmat.2023.132503Google ScholarThere is no corresponding record for this reference.
- 9Wang, Y. L.; Lee, Y. H.; Hsu, Y. H.; Chiu, I. J.; Huang, C. C.; Huang, C. C.; Chia, Z. C.; Lee, C. P.; Lin, Y. F.; Chiu, H. W. The Kidney-Related Effects of Polystyrene Microplastics on Human Kidney Proximal Tubular Epithelial Cells HK-2 and Male C57BL/6 Mice. Environ. Health Perspect. 2021, 129 (5), 57003, DOI: 10.1289/EHP7612Google ScholarThere is no corresponding record for this reference.
- 10Geens, T.; Aerts, D.; Berthot, C.; Bourguignon, J. P.; Goeyens, L.; Lecomte, P.; Maghuin-Rogister, G.; Pironnet, A. M.; Pussemier, L.; Scippo, M. L.; Van Loco, J.; Covaci, A. A review of dietary and non-dietary exposure to bisphenol-A. Food Chem. Toxicol. 2012, 50 (10), 3725– 3740, DOI: 10.1016/j.fct.2012.07.059Google ScholarThere is no corresponding record for this reference.
- 11Lind, T.; Dunder, L.; Lejonklou, M. H.; Lind, P. M.; Melhus, H.; Lind, L. Developmental low-dose bisphenol A exposure leads to extensive transcriptome female masculinization and male feminization later in life. Commun. Med. (Lond) 2025, 5 (1), 410, DOI: 10.1038/s43856-025-01119-8Google ScholarThere is no corresponding record for this reference.
- 12Wang, R.; Liu, Z. H.; Bi, N.; Gu, X.; Huang, C.; Zhou, R.; Liu, H.; Wang, H. L. Dysfunction of the medial prefrontal cortex contributes to BPA-induced depression- and anxiety-like behavior in mice. Ecotoxicol Environ. Saf 2023, 259, 115034 DOI: 10.1016/j.ecoenv.2023.115034Google ScholarThere is no corresponding record for this reference.
- 13He, W.; Gao, Z.; Liu, S.; Tan, L.; Wu, Y.; Liu, J.; Zheng, Z.; Fan, W.; Luo, Y.; Chen, Z.; Song, S. G protein-coupled estrogen receptor activation by bisphenol-A disrupts lipid metabolism and induces ferroptosis in the liver. Environ. Pollut. 2023, 334, 122211 DOI: 10.1016/j.envpol.2023.122211Google ScholarThere is no corresponding record for this reference.
- 14Lee, I.; Park, Y. J.; Kim, M. J.; Kim, S.; Choi, S.; Park, J.; Cho, Y. H.; Hong, S.; Yoo, J.; Park, H.; Cheon, G. J.; Choi, K.; Moon, M. K. Associations of urinary concentrations of phthalate metabolites, bisphenol A, and parabens with obesity and diabetes mellitus in a Korean adult population: Korean National Environmental Health Survey (KoNEHS) 2015–2017. Environ. Int. 2021, 146, 106227 DOI: 10.1016/j.envint.2020.106227Google ScholarThere is no corresponding record for this reference.
- 15Geens, T.; Neels, H.; Covaci, A. Distribution of bisphenol-A, triclosan and n-nonylphenol in human adipose tissue, liver and brain. Chemosphere 2012, 87 (7), 796– 802, DOI: 10.1016/j.chemosphere.2012.01.002Google ScholarThere is no corresponding record for this reference.
- 16Soundararajan, A.; Prabu, P.; Mohan, V.; Gibert, Y.; Balasubramanyam, M. Novel insights of elevated systemic levels of bisphenol-A (BPA) linked to poor glycemic control, accelerated cellular senescence and insulin resistance in patients with type 2 diabetes. Mol. Cell. Biochem. 2019, 458 (1–2), 171– 183, DOI: 10.1007/s11010-019-03540-9Google ScholarThere is no corresponding record for this reference.
- 17Chiu, H. W.; Chu, C. W.; Huang, C. C.; Chia, Z. C.; Wang, Y. L.; Lee, Y. H. Polystyrene microplastics induce hepatic lipid metabolism and energy disorder by upregulating the NR4A1-AMPK signaling pathway. Environ. Pollut. 2025, 369, 125850 DOI: 10.1016/j.envpol.2025.125850Google ScholarThere is no corresponding record for this reference.
- 18Wang, W.; Guan, J.; Feng, Y.; Nie, L.; Xu, Y.; Xu, H.; Fu, F. Polystyrene microplastics induced nephrotoxicity associated with oxidative stress, inflammation, and endoplasmic reticulum stress in juvenile rats. Front. Nutr. 2022, 9, 1059660 DOI: 10.3389/fnut.2022.1059660Google ScholarThere is no corresponding record for this reference.
- 19Li, B.; Ding, Y.; Cheng, X.; Sheng, D.; Xu, Z.; Rong, Q.; Wu, Y.; Zhao, H.; Ji, X.; Zhang, Y. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 2020, 244, 125492 DOI: 10.1016/j.chemosphere.2019.125492Google ScholarThere is no corresponding record for this reference.
- 20Sun, H.; Chen, N.; Yang, X.; Xia, Y.; Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol Environ. Saf 2021, 220, 112340 DOI: 10.1016/j.ecoenv.2021.112340Google ScholarThere is no corresponding record for this reference.
- 21Jing, J.; Zhang, L.; Han, L.; Wang, J.; Zhang, W.; Liu, Z.; Gao, A. Polystyrene micro-/nanoplastics induced hematopoietic damages via the crosstalk of gut microbiota, metabolites, and cytokines. Environ. Int. 2022, 161, 107131 DOI: 10.1016/j.envint.2022.107131Google ScholarThere is no corresponding record for this reference.
- 22Zhao, Q. L.; Yang, F.; Pu, Q. Y.; Zhao, R.; Jiang, S.; Tang, Y. P. Integrative metabolomics and gut microbiota analyses reveal the protective effects of DHA-enriched phosphatidylserine on bisphenol A-induced intestinal damage. J. Funct. Foods 2024, 117, 106229 DOI: 10.1016/j.jff.2024.106229Google ScholarThere is no corresponding record for this reference.
- 23Feng, L.; Chen, S.; Zhang, L.; Qu, W.; Chen, Z. Bisphenol A increases intestinal permeability through disrupting intestinal barrier function in mice. Environ. Pollut. 2019, 254 (Pt A), 112960 DOI: 10.1016/j.envpol.2019.112960Google ScholarThere is no corresponding record for this reference.
- 24Feng, D.; Zhang, H.; Jiang, X.; Zou, J.; Li, Q.; Mai, H.; Su, D.; Ling, W.; Feng, X. Bisphenol A exposure induces gut microbiota dysbiosis and consequent activation of gut-liver axis leading to hepatic steatosis in CD-1 mice. Environ. Pollut. 2020, 265 (Pt A), 114880 DOI: 10.1016/j.envpol.2020.114880Google ScholarThere is no corresponding record for this reference.
- 25Pelaseyed, T.; Bergstrom, J. H.; Gustafsson, J. K.; Ermund, A.; Birchenough, G. M.; Schutte, A.; van der Post, S.; Svensson, F.; Rodriguez-Pineiro, A. M.; Nystrom, E. E.; Wising, C.; Johansson, M. E.; Hansson, G. C. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol Rev. 2014, 260 (1), 8– 20, DOI: 10.1111/imr.12182Google ScholarThere is no corresponding record for this reference.
- 26James, K. R.; Gomes, T.; Elmentaite, R.; Kumar, N.; Gulliver, E. L.; King, H. W.; Stares, M. D.; Bareham, B. R.; Ferdinand, J. R.; Petrova, V. N.; Polanski, K.; Forster, S. C.; Jarvis, L. B.; Suchanek, O.; Howlett, S.; James, L. K.; Jones, J. L.; Meyer, K. B.; Clatworthy, M. R.; Saeb-Parsy, K.; Lawley, T. D.; Teichmann, S. A. Distinct microbial and immune niches of the human colon. Nat. Immunol 2020, 21 (3), 343– 353, DOI: 10.1038/s41590-020-0602-zGoogle ScholarThere is no corresponding record for this reference.
- 27Wang, L.; Chen, J.; Zhang, X.; Xu, M.; Zhang, X.; Zhao, W.; Cui, J. Effects of microplastics and tetracycline on intestinal injury in mice. Chemosphere 2023, 337, 139364 DOI: 10.1016/j.chemosphere.2023.139364Google ScholarThere is no corresponding record for this reference.
- 28Ahmad, E.; Lim, S.; Lamptey, R.; Webb, D. R.; Davies, M. J. Type 2 diabetes. Lancet 2022, 400 (10365), 1803– 1820, DOI: 10.1016/S0140-6736(22)01655-5Google ScholarThere is no corresponding record for this reference.
- 29Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A. A.; Ogurtsova, K.; Shaw, J. E.; Bright, D.; Williams, R.; IDF Diabetes Atlas Committee Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843 DOI: 10.1016/j.diabres.2019.107843Google ScholarThere is no corresponding record for this reference.
- 30Asgharnezhad, M.; Joukar, F.; Fathalipour, M.; Khosousi, M.; Hassanipour, S.; Pourshams, A.; Mansour-Ghanaei, R.; Mansour-Ghanaei, F. Gastrointestinal symptoms in patients with diabetes mellitus and non-diabetic: A cross-sectional study in north of Iran. Diabetes Metab Syndr 2019, 13 (3), 2236– 2240, DOI: 10.1016/j.dsx.2019.05.028Google ScholarThere is no corresponding record for this reference.
- 31Kim, J. H.; Park, H. S.; Ko, S. Y.; Hong, S. N.; Sung, I. K.; Shim, C. S.; Song, K. H.; Kim, D. L.; Kim, S. K.; Oh, J. Diabetic factors associated with gastrointestinal symptoms in patients with type 2 diabetes. World J. Gastroenterol 2010, 16 (14), 1782– 1787, DOI: 10.3748/wjg.v16.i14.1782Google ScholarThere is no corresponding record for this reference.
- 32Abdalla, M. M. I. Enteric neuropathy in diabetes: Implications for gastrointestinal function. World J. Gastroenterol 2024, 30 (22), 2852– 2865, DOI: 10.3748/wjg.v30.i22.2852Google ScholarThere is no corresponding record for this reference.
- 33Ma, J. W.; Lai, T. J.; Hu, S. Y.; Lin, T. C.; Ho, W. C.; Tsan, Y. T. Effect of ambient air pollution on the incidence of colorectal cancer among a diabetic population: a nationwide nested case-control study in Taiwan. BMJ. Open 2020, 10 (10), e036955 DOI: 10.1136/bmjopen-2020-036955Google ScholarThere is no corresponding record for this reference.
- 34Zitouni, N.; Bousserrhine, N.; Belbekhouche, S.; Missawi, O.; Alphonse, V.; Boughatass, I.; Banni, M. First report on the presence of small microplastics (≤3 μm) in tissue of the commercial fish Serranus scriba (Linnaeus. 1758) from Tunisian coasts and associated cellular alterations. Environ. Pollut. 2020, 263 (Pt A), 114576 DOI: 10.1016/j.envpol.2020.114576Google ScholarThere is no corresponding record for this reference.
- 35Senathirajah, K.; Attwood, S.; Bhagwat, G.; Carbery, M.; Wilson, S.; Palanisami, T. Estimation of the mass of microplastics ingested - A pivotal first step towards human health risk assessment. J. Hazard. Mater. 2021, 404 (Pt B), 124004 DOI: 10.1016/j.jhazmat.2020.124004Google ScholarThere is no corresponding record for this reference.
- 36Cox, K. D.; Covernton, G. A.; Davies, H. L.; Dower, J. F.; Juanes, F.; Dudas, S. E. Human Consumption of Microplastics. Environ. Sci. Technol. 2019, 53 (12), 7068– 7074, DOI: 10.1021/acs.est.9b01517Google ScholarThere is no corresponding record for this reference.
- 37Qin, S.; Gao, M.; Zhang, Q.; Xiao, Q.; Fu, J.; Tian, Y.; Jiao, Y.; Zhang, Z.; Zhang, P.; Xu, F. High-Coverage Strategy for Multi-Subcellular Metabolome Analysis Using Dansyl-Labeling-Based LC-MS/MS. Anal. Chem. 2023, 95 (26), 10034– 10043, DOI: 10.1021/acs.analchem.3c01343Google ScholarThere is no corresponding record for this reference.
- 38Baars, D. P.; Fondevila, M. F.; Meijnikman, A. S.; Nieuwdorp, M. The central role of the gut microbiota in the pathophysiology and management of type 2 diabetes. Cell Host Microbe 2024, 32 (8), 1280– 1300, DOI: 10.1016/j.chom.2024.07.017Google ScholarThere is no corresponding record for this reference.
- 39Takeuchi, T.; Kubota, T.; Nakanishi, Y.; Tsugawa, H.; Suda, W.; Kwon, A. T.; Yazaki, J.; Ikeda, K.; Nemoto, S.; Mochizuki, Y.; Kitami, T.; Yugi, K.; Mizuno, Y.; Yamamichi, N.; Yamazaki, T.; Takamoto, I.; Kubota, N.; Kadowaki, T.; Arner, E.; Carninci, P.; Ohara, O.; Arita, M.; Hattori, M.; Koyasu, S.; Ohno, H. Gut microbial carbohydrate metabolism contributes to insulin resistance. Nature 2023, 621 (7978), 389– 395, DOI: 10.1038/s41586-023-06466-xGoogle ScholarThere is no corresponding record for this reference.
- 40Herp, S.; Durai Raj, A. C.; Salvado Silva, M.; Woelfel, S.; Stecher, B. The human symbiont Mucispirillum schaedleri: causality in health and disease. Med. Microbiol Immunol 2021, 210 (4), 173– 179, DOI: 10.1007/s00430-021-00702-9Google ScholarThere is no corresponding record for this reference.
- 41Liu, J.; Zhao, B.; Wang, L.; Zhang, W.; Zan, T.; Chen, Z.; Li, Y. Occurrence, fate, and transport of N-nitrosamines and precursors in sewage treatment plants and receiving rivers in a highly urbanized basin. Environ. Pollut. 2025, 368, 125808 DOI: 10.1016/j.envpol.2025.125808Google ScholarThere is no corresponding record for this reference.
- 42Behera, S.; Das, S. Potential and prospects of Actinobacteria in the bioremediation of environmental pollutants: Cellular mechanisms and genetic regulations. Microbiol Res. 2023, 273, 127399 DOI: 10.1016/j.micres.2023.127399Google ScholarThere is no corresponding record for this reference.
- 43Bao, L.; Cui, X.; Zeng, T.; Liu, G.; Lai, W.; Zhao, H.; Gao, F.; Wu, J.; Leong, K. W.; Chen, C. Incorporation of polylactic acid microplastics into the carbon cycle as a carbon source to remodel the endogenous metabolism of the gut. Proc. Natl. Acad. Sci. U. S. A. 2025, 122 (19), e2417104122 DOI: 10.1073/pnas.2417104122Google ScholarThere is no corresponding record for this reference.
- 44Zhang, K.; Yang, J.; Chen, L.; He, J.; Qu, D.; Zhang, Z.; Liu, Y.; Li, X.; Liu, J.; Li, J.; Xie, X.; Wang, Q. Gut Microbiota Participates in Polystyrene Microplastics-Induced Hepatic Injuries by Modulating the Gut-Liver Axis. ACS Nano 2023, 17 (15), 15125– 15145, DOI: 10.1021/acsnano.3c04449Google ScholarThere is no corresponding record for this reference.
- 45Ni, Y.; Hu, L.; Yang, S.; Ni, L.; Ma, L.; Zhao, Y.; Zheng, A.; Jin, Y.; Fu, Z. Bisphenol A impairs cognitive function and 5-HT metabolism in adult male mice by modulating the microbiota-gut-brain axis. Chemosphere 2021, 282, 130952 DOI: 10.1016/j.chemosphere.2021.130952Google ScholarThere is no corresponding record for this reference.
- 46DeLuca, J. A.; Allred, K. F.; Menon, R.; Riordan, R.; Weeks, B. R.; Jayaraman, A.; Allred, C. D. Bisphenol-A alters microbiota metabolites derived from aromatic amino acids and worsens disease activity during colitis. Exp Biol. Med. (Maywood) 2018, 243 (10), 864– 875, DOI: 10.1177/1535370218782139Google ScholarThere is no corresponding record for this reference.
- 47Chen, Y.; Mai, Q.; Chen, Z.; Lin, T.; Cai, Y.; Han, J.; Wang, Y.; Zhang, M.; Tan, S.; Wu, Z.; Chen, L.; Zhang, Z.; Yang, Y.; Cui, T.; Ouyang, B.; Sun, Y.; Yang, L.; Xu, L.; Zhang, S.; Li, J.; Shen, H.; Liu, L.; Zeng, L.; Zhang, S.; Zeng, G. Dietary palmitoleic acid reprograms gut microbiota and improves biological therapy against colitis. Gut Microbes 2023, 15 (1), 2211501 DOI: 10.1080/19490976.2023.2211501Google ScholarThere is no corresponding record for this reference.
- 48Yang, J.; Liu, S.; Zhao, Q.; Li, X.; Jiang, K. Gut microbiota-related metabolite alpha-linolenic acid mitigates intestinal inflammation induced by oral infection with Toxoplasma gondii. Microbiome 2023, 11 (1), 273, DOI: 10.1186/s40168-023-01681-0Google ScholarThere is no corresponding record for this reference.
- 49Bahreyni, A.; Samani, S. S.; Khazaei, M.; Ryzhikov, M.; Avan, A.; Hassanian, S. M. Therapeutic potentials of adenosine receptors agonists and antagonists in colitis; Current status and perspectives. J. Cell Physiol 2018, 233 (4), 2733– 2740, DOI: 10.1002/jcp.26073Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 1 publications.
- Liangliang Dai, Chenjie Qiu, . Integrated Multiomics Elucidates Molecular Mechanisms of Bisphenol A in Exacerbating Crohn’s Disease. Mediators of Inflammation 2026, 2026
(1)
https://doi.org/10.1155/mi/2903373
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1

Figure 1. T2D mice exhibit mild gut injury. (A) FBG levels and (B) colon length in T2D and control groups (n = 10). (C) Correlation analysis between FBG and colon length. (D) Correlation analysis among FBG, colon length, body weight, and organ coefficients. (E) Protein expression levels of ZO-1 and occludin in the colon (n = 6). (F) Representative images of H&E and PAS staining of colon tissues (n = 3) (100×). Significance levels are indicated as ns (not significant), **p < 0.01, ****p < 0.0001.
Figure 2

Figure 2. PS-MPs and BPA exposures promote intestinal injury in T2D mice. (A) PS-MPs confirmation by Fourier-transform infrared spectroscopy. (B) Scanning electron microscopy of PS-MPs. (C) Zeta potentials of the PS-MPs. Colon length of (D) PS-MPs (n ≥ 7) and (E) BPA group (n = 6). (F) Representative images of H&E and PAS staining of colon tissues (n = 3) (100×, yellow arrows indicate reduced goblet cells and mucus secretion). Intestinal ZO-1 and occludin expression of (G) PS-MPs and (H) BPA group (n = 6). Significance levels are indicated as ns (not significant), *p < 0.05, **p < 0.01, and ****p < 0.0001.
Figure 3

Figure 3. PS-MPs and BPA exposures alter gut microbiota in T2D mice (n = 3). (A–C) Alpha diversity indices of the T2D, PS-MPs, and BPA groups. (D–F) PCoA analysis of the T2D, PS-MPs, and BPA groups. Linear discriminant analysis effective size (LEfSe) identifying differentially abundant taxa at the genus level between (G) PS-MPs-C and PS-MPs, and (H) BPA-C and BPA groups. Significance determined with a linear discriminant analysis (LDA) score (log 10) > 2. **p < 0.01.
Figure 4
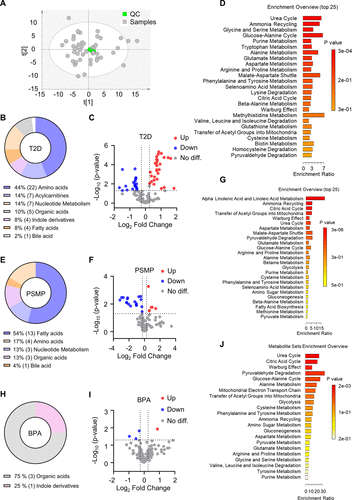
Figure 4. PS-MPs and BPA exposures induce gut metabolic dysregulations in T2D mice. (A) PCA analysis based on LC-MS metabolomics data. (B, E, H) Pie chart showing the categories of differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively. (C, F, I) Volcano plots illustrating differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively. (D, G, J) Pathway enrichment analysis of differential metabolites in the T2D (n = 10), PS-MPs (n ≥ 7), and BPA (n = 6) groups, respectively.
Figure 5

Figure 5. Intestinal metabolic dysregulation is linked to intestinal injury in T2D mice with PS-MPs exposure. Intestinal concentration of C14 (A), C15 (B), C16:1 (C), C18:3 (D), and Ado (E) in T2D (n = 10), PS-MPs (n ≥ 7), and their corresponding control. (F) Heatmap of differential metabolites in T2D (n = 10) and PS-MPs (n ≥ 7). (G) Correlation analysis between FBG, colon length, body weight, and differential metabolites in PS-MPs. Intestinal concentration of KIVA (H) and PyrA (I) in T2D (n = 10), BPA (n = 6), and their corresponding control. (J) Heatmap of differential metabolites in T2D (n = 10) and BPA (n = 6). (K) Correlation analysis between FBG, colon length, body weight, and differential metabolites in BPA.
Figure 6

Figure 6. Altered metabolites promote the expression of gut barrier proteins (n = 3). (A) Protein expression level of ZO-1 and occludin with 20 μM (A) and 100 μM (B) C14, C15, C16:1, C18:3, and Ado treatment. Protein expression level of ZO-1 and occludin with 0.1 μM (C), 1 μM (D), and 10 μM (E) KIVA and PyrA treatment. Significance levels are indicated as *p < 0.05 and **p < 0.01.
Figure 7

Figure 7. Correlation between gut microbiota dysbiosis and colonic metabolic alterations. (A) Correlation analysis of FBG, colon length, body weight, Duncaniella, Olsenella, and differential metabolites in the PS-MPs group. (B) Correlation between colon length and Duncaniella. (C) Correlation between C16:1 and Duncaniella. (D) Correlation between C18:3 and Duncaniella. (E) Correlation between Ado and Duncaniella. (F) Correlation between colon length and Olsenella. (G) Correlation between C16:1 and Olsenella. (H) Correlation between C18:3 and Olsenella. (I) Correlation between Ado and Olsenella. (J) Correlation analysis of FBG, colon length, body weight, Variovorax, Phocaeicola, Olsenella, and differential metabolites in BPA. Correlation analyses were performed using Pearson’s method.
References
This article references 49 other publications.
- 1Carpenter, E. J.; Smith, K. L., Jr Plastics on the Sargasso sea surface. Science 1972, 175 (4027), 1240– 1241, DOI: 10.1126/science.175.4027.1240There is no corresponding record for this reference.
- 2Wright, S. L.; Ulke, J.; Font, A.; Chan, K. L. A.; Kelly, F. J. Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environ. Int. 2020, 136, 105411 DOI: 10.1016/j.envint.2019.105411There is no corresponding record for this reference.
- 3Golwala, H.; Zhang, X.; Iskander, S. M.; Smith, A. L. Solid waste: An overlooked source of microplastics to the environment. Sci. Total Environ. 2021, 769, 144581 DOI: 10.1016/j.scitotenv.2020.144581There is no corresponding record for this reference.
- 4Thompson, R. C.; Courtene-Jones, W.; Boucher, J.; Pahl, S.; Raubenheimer, K.; Koelmans, A. A. Twenty years of microplastic pollution research-what have we learned?. Science 2024, 386 (6720), eadl2746 DOI: 10.1126/science.adl2746There is no corresponding record for this reference.
- 5Hernandez, L. M.; Xu, E. G.; Larsson, H. C. E.; Tahara, R.; Maisuria, V. B.; Tufenkji, N. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 2019, 53 (21), 12300– 12310, DOI: 10.1021/acs.est.9b02540There is no corresponding record for this reference.
- 6Hussain, K. A.; Romanova, S.; Okur, I.; Zhang, D.; Kuebler, J.; Huang, X.; Wang, B.; Fernandez-Ballester, L.; Lu, Y.; Schubert, M.; Li, Y. Assessing the Release of Microplastics and Nanoplastics from Plastic Containers and Reusable Food Pouches: Implications for Human Health. Environ. Sci. Technol. 2023, 57 (26), 9782– 9792, DOI: 10.1021/acs.est.3c01942There is no corresponding record for this reference.
- 7Wu, P.; Fan, Y.; Zhang, X.; Wu, W.; Zhang, Z.; Wu, Y.; Wang, J.; Xu, J.; Chen, T.; Gao, B. Seasonal dynamics, tidal influences, and anthropogenic impacts on microplastic distribution in the Yangtze River estuary: A comprehensive characterization and comparative analysis. J. Hazard Mater. 2024, 476, 135167 DOI: 10.1016/j.jhazmat.2024.135167There is no corresponding record for this reference.
- 8Zhang, Z.; Chen, W.; Chan, H.; Peng, J.; Zhu, P.; Li, J.; Jiang, X.; Zhang, Z.; Wang, Y.; Tan, Z.; Peng, Y.; Zhang, S.; Lin, K.; Yung, K. K. Polystyrene microplastics induce size-dependent multi-organ damage in mice: Insights into gut microbiota and fecal metabolites. J. Hazard Mater. 2024, 461, 132503 DOI: 10.1016/j.jhazmat.2023.132503There is no corresponding record for this reference.
- 9Wang, Y. L.; Lee, Y. H.; Hsu, Y. H.; Chiu, I. J.; Huang, C. C.; Huang, C. C.; Chia, Z. C.; Lee, C. P.; Lin, Y. F.; Chiu, H. W. The Kidney-Related Effects of Polystyrene Microplastics on Human Kidney Proximal Tubular Epithelial Cells HK-2 and Male C57BL/6 Mice. Environ. Health Perspect. 2021, 129 (5), 57003, DOI: 10.1289/EHP7612There is no corresponding record for this reference.
- 10Geens, T.; Aerts, D.; Berthot, C.; Bourguignon, J. P.; Goeyens, L.; Lecomte, P.; Maghuin-Rogister, G.; Pironnet, A. M.; Pussemier, L.; Scippo, M. L.; Van Loco, J.; Covaci, A. A review of dietary and non-dietary exposure to bisphenol-A. Food Chem. Toxicol. 2012, 50 (10), 3725– 3740, DOI: 10.1016/j.fct.2012.07.059There is no corresponding record for this reference.
- 11Lind, T.; Dunder, L.; Lejonklou, M. H.; Lind, P. M.; Melhus, H.; Lind, L. Developmental low-dose bisphenol A exposure leads to extensive transcriptome female masculinization and male feminization later in life. Commun. Med. (Lond) 2025, 5 (1), 410, DOI: 10.1038/s43856-025-01119-8There is no corresponding record for this reference.
- 12Wang, R.; Liu, Z. H.; Bi, N.; Gu, X.; Huang, C.; Zhou, R.; Liu, H.; Wang, H. L. Dysfunction of the medial prefrontal cortex contributes to BPA-induced depression- and anxiety-like behavior in mice. Ecotoxicol Environ. Saf 2023, 259, 115034 DOI: 10.1016/j.ecoenv.2023.115034There is no corresponding record for this reference.
- 13He, W.; Gao, Z.; Liu, S.; Tan, L.; Wu, Y.; Liu, J.; Zheng, Z.; Fan, W.; Luo, Y.; Chen, Z.; Song, S. G protein-coupled estrogen receptor activation by bisphenol-A disrupts lipid metabolism and induces ferroptosis in the liver. Environ. Pollut. 2023, 334, 122211 DOI: 10.1016/j.envpol.2023.122211There is no corresponding record for this reference.
- 14Lee, I.; Park, Y. J.; Kim, M. J.; Kim, S.; Choi, S.; Park, J.; Cho, Y. H.; Hong, S.; Yoo, J.; Park, H.; Cheon, G. J.; Choi, K.; Moon, M. K. Associations of urinary concentrations of phthalate metabolites, bisphenol A, and parabens with obesity and diabetes mellitus in a Korean adult population: Korean National Environmental Health Survey (KoNEHS) 2015–2017. Environ. Int. 2021, 146, 106227 DOI: 10.1016/j.envint.2020.106227There is no corresponding record for this reference.
- 15Geens, T.; Neels, H.; Covaci, A. Distribution of bisphenol-A, triclosan and n-nonylphenol in human adipose tissue, liver and brain. Chemosphere 2012, 87 (7), 796– 802, DOI: 10.1016/j.chemosphere.2012.01.002There is no corresponding record for this reference.
- 16Soundararajan, A.; Prabu, P.; Mohan, V.; Gibert, Y.; Balasubramanyam, M. Novel insights of elevated systemic levels of bisphenol-A (BPA) linked to poor glycemic control, accelerated cellular senescence and insulin resistance in patients with type 2 diabetes. Mol. Cell. Biochem. 2019, 458 (1–2), 171– 183, DOI: 10.1007/s11010-019-03540-9There is no corresponding record for this reference.
- 17Chiu, H. W.; Chu, C. W.; Huang, C. C.; Chia, Z. C.; Wang, Y. L.; Lee, Y. H. Polystyrene microplastics induce hepatic lipid metabolism and energy disorder by upregulating the NR4A1-AMPK signaling pathway. Environ. Pollut. 2025, 369, 125850 DOI: 10.1016/j.envpol.2025.125850There is no corresponding record for this reference.
- 18Wang, W.; Guan, J.; Feng, Y.; Nie, L.; Xu, Y.; Xu, H.; Fu, F. Polystyrene microplastics induced nephrotoxicity associated with oxidative stress, inflammation, and endoplasmic reticulum stress in juvenile rats. Front. Nutr. 2022, 9, 1059660 DOI: 10.3389/fnut.2022.1059660There is no corresponding record for this reference.
- 19Li, B.; Ding, Y.; Cheng, X.; Sheng, D.; Xu, Z.; Rong, Q.; Wu, Y.; Zhao, H.; Ji, X.; Zhang, Y. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 2020, 244, 125492 DOI: 10.1016/j.chemosphere.2019.125492There is no corresponding record for this reference.
- 20Sun, H.; Chen, N.; Yang, X.; Xia, Y.; Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol Environ. Saf 2021, 220, 112340 DOI: 10.1016/j.ecoenv.2021.112340There is no corresponding record for this reference.
- 21Jing, J.; Zhang, L.; Han, L.; Wang, J.; Zhang, W.; Liu, Z.; Gao, A. Polystyrene micro-/nanoplastics induced hematopoietic damages via the crosstalk of gut microbiota, metabolites, and cytokines. Environ. Int. 2022, 161, 107131 DOI: 10.1016/j.envint.2022.107131There is no corresponding record for this reference.
- 22Zhao, Q. L.; Yang, F.; Pu, Q. Y.; Zhao, R.; Jiang, S.; Tang, Y. P. Integrative metabolomics and gut microbiota analyses reveal the protective effects of DHA-enriched phosphatidylserine on bisphenol A-induced intestinal damage. J. Funct. Foods 2024, 117, 106229 DOI: 10.1016/j.jff.2024.106229There is no corresponding record for this reference.
- 23Feng, L.; Chen, S.; Zhang, L.; Qu, W.; Chen, Z. Bisphenol A increases intestinal permeability through disrupting intestinal barrier function in mice. Environ. Pollut. 2019, 254 (Pt A), 112960 DOI: 10.1016/j.envpol.2019.112960There is no corresponding record for this reference.
- 24Feng, D.; Zhang, H.; Jiang, X.; Zou, J.; Li, Q.; Mai, H.; Su, D.; Ling, W.; Feng, X. Bisphenol A exposure induces gut microbiota dysbiosis and consequent activation of gut-liver axis leading to hepatic steatosis in CD-1 mice. Environ. Pollut. 2020, 265 (Pt A), 114880 DOI: 10.1016/j.envpol.2020.114880There is no corresponding record for this reference.
- 25Pelaseyed, T.; Bergstrom, J. H.; Gustafsson, J. K.; Ermund, A.; Birchenough, G. M.; Schutte, A.; van der Post, S.; Svensson, F.; Rodriguez-Pineiro, A. M.; Nystrom, E. E.; Wising, C.; Johansson, M. E.; Hansson, G. C. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol Rev. 2014, 260 (1), 8– 20, DOI: 10.1111/imr.12182There is no corresponding record for this reference.
- 26James, K. R.; Gomes, T.; Elmentaite, R.; Kumar, N.; Gulliver, E. L.; King, H. W.; Stares, M. D.; Bareham, B. R.; Ferdinand, J. R.; Petrova, V. N.; Polanski, K.; Forster, S. C.; Jarvis, L. B.; Suchanek, O.; Howlett, S.; James, L. K.; Jones, J. L.; Meyer, K. B.; Clatworthy, M. R.; Saeb-Parsy, K.; Lawley, T. D.; Teichmann, S. A. Distinct microbial and immune niches of the human colon. Nat. Immunol 2020, 21 (3), 343– 353, DOI: 10.1038/s41590-020-0602-zThere is no corresponding record for this reference.
- 27Wang, L.; Chen, J.; Zhang, X.; Xu, M.; Zhang, X.; Zhao, W.; Cui, J. Effects of microplastics and tetracycline on intestinal injury in mice. Chemosphere 2023, 337, 139364 DOI: 10.1016/j.chemosphere.2023.139364There is no corresponding record for this reference.
- 28Ahmad, E.; Lim, S.; Lamptey, R.; Webb, D. R.; Davies, M. J. Type 2 diabetes. Lancet 2022, 400 (10365), 1803– 1820, DOI: 10.1016/S0140-6736(22)01655-5There is no corresponding record for this reference.
- 29Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A. A.; Ogurtsova, K.; Shaw, J. E.; Bright, D.; Williams, R.; IDF Diabetes Atlas Committee Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843 DOI: 10.1016/j.diabres.2019.107843There is no corresponding record for this reference.
- 30Asgharnezhad, M.; Joukar, F.; Fathalipour, M.; Khosousi, M.; Hassanipour, S.; Pourshams, A.; Mansour-Ghanaei, R.; Mansour-Ghanaei, F. Gastrointestinal symptoms in patients with diabetes mellitus and non-diabetic: A cross-sectional study in north of Iran. Diabetes Metab Syndr 2019, 13 (3), 2236– 2240, DOI: 10.1016/j.dsx.2019.05.028There is no corresponding record for this reference.
- 31Kim, J. H.; Park, H. S.; Ko, S. Y.; Hong, S. N.; Sung, I. K.; Shim, C. S.; Song, K. H.; Kim, D. L.; Kim, S. K.; Oh, J. Diabetic factors associated with gastrointestinal symptoms in patients with type 2 diabetes. World J. Gastroenterol 2010, 16 (14), 1782– 1787, DOI: 10.3748/wjg.v16.i14.1782There is no corresponding record for this reference.
- 32Abdalla, M. M. I. Enteric neuropathy in diabetes: Implications for gastrointestinal function. World J. Gastroenterol 2024, 30 (22), 2852– 2865, DOI: 10.3748/wjg.v30.i22.2852There is no corresponding record for this reference.
- 33Ma, J. W.; Lai, T. J.; Hu, S. Y.; Lin, T. C.; Ho, W. C.; Tsan, Y. T. Effect of ambient air pollution on the incidence of colorectal cancer among a diabetic population: a nationwide nested case-control study in Taiwan. BMJ. Open 2020, 10 (10), e036955 DOI: 10.1136/bmjopen-2020-036955There is no corresponding record for this reference.
- 34Zitouni, N.; Bousserrhine, N.; Belbekhouche, S.; Missawi, O.; Alphonse, V.; Boughatass, I.; Banni, M. First report on the presence of small microplastics (≤3 μm) in tissue of the commercial fish Serranus scriba (Linnaeus. 1758) from Tunisian coasts and associated cellular alterations. Environ. Pollut. 2020, 263 (Pt A), 114576 DOI: 10.1016/j.envpol.2020.114576There is no corresponding record for this reference.
- 35Senathirajah, K.; Attwood, S.; Bhagwat, G.; Carbery, M.; Wilson, S.; Palanisami, T. Estimation of the mass of microplastics ingested - A pivotal first step towards human health risk assessment. J. Hazard. Mater. 2021, 404 (Pt B), 124004 DOI: 10.1016/j.jhazmat.2020.124004There is no corresponding record for this reference.
- 36Cox, K. D.; Covernton, G. A.; Davies, H. L.; Dower, J. F.; Juanes, F.; Dudas, S. E. Human Consumption of Microplastics. Environ. Sci. Technol. 2019, 53 (12), 7068– 7074, DOI: 10.1021/acs.est.9b01517There is no corresponding record for this reference.
- 37Qin, S.; Gao, M.; Zhang, Q.; Xiao, Q.; Fu, J.; Tian, Y.; Jiao, Y.; Zhang, Z.; Zhang, P.; Xu, F. High-Coverage Strategy for Multi-Subcellular Metabolome Analysis Using Dansyl-Labeling-Based LC-MS/MS. Anal. Chem. 2023, 95 (26), 10034– 10043, DOI: 10.1021/acs.analchem.3c01343There is no corresponding record for this reference.
- 38Baars, D. P.; Fondevila, M. F.; Meijnikman, A. S.; Nieuwdorp, M. The central role of the gut microbiota in the pathophysiology and management of type 2 diabetes. Cell Host Microbe 2024, 32 (8), 1280– 1300, DOI: 10.1016/j.chom.2024.07.017There is no corresponding record for this reference.
- 39Takeuchi, T.; Kubota, T.; Nakanishi, Y.; Tsugawa, H.; Suda, W.; Kwon, A. T.; Yazaki, J.; Ikeda, K.; Nemoto, S.; Mochizuki, Y.; Kitami, T.; Yugi, K.; Mizuno, Y.; Yamamichi, N.; Yamazaki, T.; Takamoto, I.; Kubota, N.; Kadowaki, T.; Arner, E.; Carninci, P.; Ohara, O.; Arita, M.; Hattori, M.; Koyasu, S.; Ohno, H. Gut microbial carbohydrate metabolism contributes to insulin resistance. Nature 2023, 621 (7978), 389– 395, DOI: 10.1038/s41586-023-06466-xThere is no corresponding record for this reference.
- 40Herp, S.; Durai Raj, A. C.; Salvado Silva, M.; Woelfel, S.; Stecher, B. The human symbiont Mucispirillum schaedleri: causality in health and disease. Med. Microbiol Immunol 2021, 210 (4), 173– 179, DOI: 10.1007/s00430-021-00702-9There is no corresponding record for this reference.
- 41Liu, J.; Zhao, B.; Wang, L.; Zhang, W.; Zan, T.; Chen, Z.; Li, Y. Occurrence, fate, and transport of N-nitrosamines and precursors in sewage treatment plants and receiving rivers in a highly urbanized basin. Environ. Pollut. 2025, 368, 125808 DOI: 10.1016/j.envpol.2025.125808There is no corresponding record for this reference.
- 42Behera, S.; Das, S. Potential and prospects of Actinobacteria in the bioremediation of environmental pollutants: Cellular mechanisms and genetic regulations. Microbiol Res. 2023, 273, 127399 DOI: 10.1016/j.micres.2023.127399There is no corresponding record for this reference.
- 43Bao, L.; Cui, X.; Zeng, T.; Liu, G.; Lai, W.; Zhao, H.; Gao, F.; Wu, J.; Leong, K. W.; Chen, C. Incorporation of polylactic acid microplastics into the carbon cycle as a carbon source to remodel the endogenous metabolism of the gut. Proc. Natl. Acad. Sci. U. S. A. 2025, 122 (19), e2417104122 DOI: 10.1073/pnas.2417104122There is no corresponding record for this reference.
- 44Zhang, K.; Yang, J.; Chen, L.; He, J.; Qu, D.; Zhang, Z.; Liu, Y.; Li, X.; Liu, J.; Li, J.; Xie, X.; Wang, Q. Gut Microbiota Participates in Polystyrene Microplastics-Induced Hepatic Injuries by Modulating the Gut-Liver Axis. ACS Nano 2023, 17 (15), 15125– 15145, DOI: 10.1021/acsnano.3c04449There is no corresponding record for this reference.
- 45Ni, Y.; Hu, L.; Yang, S.; Ni, L.; Ma, L.; Zhao, Y.; Zheng, A.; Jin, Y.; Fu, Z. Bisphenol A impairs cognitive function and 5-HT metabolism in adult male mice by modulating the microbiota-gut-brain axis. Chemosphere 2021, 282, 130952 DOI: 10.1016/j.chemosphere.2021.130952There is no corresponding record for this reference.
- 46DeLuca, J. A.; Allred, K. F.; Menon, R.; Riordan, R.; Weeks, B. R.; Jayaraman, A.; Allred, C. D. Bisphenol-A alters microbiota metabolites derived from aromatic amino acids and worsens disease activity during colitis. Exp Biol. Med. (Maywood) 2018, 243 (10), 864– 875, DOI: 10.1177/1535370218782139There is no corresponding record for this reference.
- 47Chen, Y.; Mai, Q.; Chen, Z.; Lin, T.; Cai, Y.; Han, J.; Wang, Y.; Zhang, M.; Tan, S.; Wu, Z.; Chen, L.; Zhang, Z.; Yang, Y.; Cui, T.; Ouyang, B.; Sun, Y.; Yang, L.; Xu, L.; Zhang, S.; Li, J.; Shen, H.; Liu, L.; Zeng, L.; Zhang, S.; Zeng, G. Dietary palmitoleic acid reprograms gut microbiota and improves biological therapy against colitis. Gut Microbes 2023, 15 (1), 2211501 DOI: 10.1080/19490976.2023.2211501There is no corresponding record for this reference.
- 48Yang, J.; Liu, S.; Zhao, Q.; Li, X.; Jiang, K. Gut microbiota-related metabolite alpha-linolenic acid mitigates intestinal inflammation induced by oral infection with Toxoplasma gondii. Microbiome 2023, 11 (1), 273, DOI: 10.1186/s40168-023-01681-0There is no corresponding record for this reference.
- 49Bahreyni, A.; Samani, S. S.; Khazaei, M.; Ryzhikov, M.; Avan, A.; Hassanian, S. M. Therapeutic potentials of adenosine receptors agonists and antagonists in colitis; Current status and perspectives. J. Cell Physiol 2018, 233 (4), 2733– 2740, DOI: 10.1002/jcp.26073There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.chemrestox.5c00359.
Figure S1. Colonic inflammatory factors expression in T2D mice. Figure S2. Organ coefficient and FBG in T2D mice with PS-MPs/BPA exposure. Figure S3. Colonic inflammatory factors expression in T2D mice with PS-MPs/BPA exposure. Figure S4. Colonic protein expression in T2D mice with PS-MPs/BPA exposure. Figure S5. Linear discriminant analysis effect size (LEfSe) comparing the Control and T2D groups. Figure S6. Metabolomics analysis in T2D mice with PS-MPs/BPA exposure. Table S1. Metabolite retention time and MRM parameters. Table S2. Intestinal differential metabolites between groups (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



