
Sensing Disease in Apple Trees: Potential Signature Compounds for European Canker (Neonectria ditissima) InfectionClick to copy article linkArticle link copied!
- Catherine E. SansomCatherine E. SansomThe New Zealand Institute for Plant and Food Research Limited, Department of Chemistry, University of Otago, Dunedin 9054, New ZealandMore by Catherine E. Sansom
- Elaine J. BurgessElaine J. BurgessThe New Zealand Institute for Plant and Food Research Limited, Department of Chemistry, University of Otago, Dunedin 9054, New ZealandMore by Elaine J. Burgess
- Lesley LarsenLesley LarsenThe New Zealand Institute for Plant and Food Research Limited, Department of Chemistry, University of Otago, Dunedin 9054, New ZealandMore by Lesley Larsen
- Nigel I. JoyceNigel I. JoyceThe New Zealand Institute for Plant and Food Research Limited, Canterbury Agriculture and Science Centre, Lincoln 7608, New ZealandMore by Nigel I. Joyce
- Peter JaksonsPeter JaksonsThe New Zealand Institute for Plant and Food Research Limited, Canterbury Agriculture and Science Centre, Lincoln 7608, New ZealandMore by Peter Jaksons
- Rebecca E. CampbellRebecca E. CampbellThe New Zealand Institute for Plant and Food Research Limited, Brooklyn, Motueka 7198, New ZealandMore by Rebecca E. Campbell
- Ria S. RebstockRia S. RebstockThe New Zealand Institute for Plant and Food Research Limited, Mt Albert, Auckland 1025, New ZealandMore by Ria S. Rebstock
- Ian C. HallettIan C. HallettThe New Zealand Institute for Plant and Food Research Limited, Mt Albert, Auckland 1025, New ZealandMore by Ian C. Hallett
- Jonathan Rees-GeorgeJonathan Rees-GeorgeThe New Zealand Institute for Plant and Food Research Limited, Mt Albert, Auckland 1025, New ZealandMore by Jonathan Rees-George
- Kerry R. EverettKerry R. EverettThe New Zealand Institute for Plant and Food Research Limited, Mt Albert, Auckland 1025, New ZealandMore by Kerry R. Everett
- Reiny W. A. ScheperReiny W. A. ScheperThe New Zealand Institute for Plant and Food Research Limited, Havelock North, Hawke’s Bay 4130, New ZealandMore by Reiny W. A. Scheper
- Monika WalterMonika WalterThe New Zealand Institute for Plant and Food Research Limited, 412 No 1 Road, RD 2, Te Puke 3182, New ZealandMore by Monika Walter
- Nigel B. Perry*Nigel B. Perry*Email: [email protected]The New Zealand Institute for Plant and Food Research Limited, Department of Chemistry, University of Otago, Dunedin 9054, New ZealandMore by Nigel B. Perry
Abstract
Neonectria ditissima is the fungal cause of costly European Canker (EC) in apple trees. A range of secondary metabolites were found at higher concentrations in infected twigs than in disease free twigs. Apple trees were then experimentally inoculated with N. ditissima and analyzed periodically until EC symptoms were visible at 12–13 weeks post-inoculation. Established destructive detection methods used were microscopy, which showed extensive hyphal penetration by 8 weeks post inoculation, and qPCR analyses, which confirmed the presence of N. ditissima. Headspace solid-phase microextraction GC-MS data showed significantly higher concentrations of styrene in apple twigs at six weeks after inoculation, and LC-MS data showed phloretin, triterpene acids, and 1-benzoyl β-d-glucose at raised concentrations after this time. Therefore, these compounds could be useful indicators of N. ditissima infection prior to visible canker formation, suitable for nondestructive disease detection development after further research on apple variety, pathogen specificity, and on field detection technology.
Introduction
Materials and Methods
Apple Twig Samples for Signature Compound Discovery
Figure 1
Figure 1. (A) Apple twig leaf scar, with leaf bud above (image by Tony Corbett, PFR). (B) Apple twig with visible European Canker infection. (C) Cross-section of infected apple twig leaf scar showing Neonectria ditissima symptoms (brown tissue at the top).
Apple Twig Samples for the Time Course Experiment
HS-SPME-GC-MS Analyses
LC-MS Analyses
Microscopy
qPCR Analyses
Statistical Analyses
Compound Synthesis for Identification
Compound Isolation for Identification: Triterpene Acids and Phloridzin
Preparative HPLC
Results and Discussion
Potential Volatile Signature Compounds
Figure 2
Figure 2. (A) Two-dimensional projection of principal components analysis of headspace solid-phase micro-extraction GC-MS data of apple twigs showing the variability between different treatments. Red triangles = ‘Scifresh’ inoculated, purple squares = ‘Scifresh’ control, green triangles = ‘Royal Gala’ inoculated, blue squares = ‘Royal Gala’ control from Motueka, light blue squares = ‘Royal Gala’ control from Hawke’s Bay. (B) Loading plot for PC-1, showing positive loadings corresponding to peaks for hexanol and styrene, and negative loadings for sesquiterpenes. The x-axis numbers correspond to GC retention time (min).
Figure 3
Figure 3. Structures of potential signature compounds for Neonectria ditissima infection of apple twigs.
Potential Nonvolatile Signature Compounds
Figure 4
Figure 4. LC-MS differential plots for all component mass features that changed between infected and control ‘Royal Gala’ apple twigs by reverse phase liquid chromatography with negative ion (A) and positive ion (B) MS. Plot x-axis log2fold change (negative values indicate down regulation and positive values up regulation due to infection); y-axis ion species mass features as m/z (compounds represented by one or more signal ions M, 2M, M+ adduct, etc.); marker dimension is the maximum area response for an ion species mass feature; color represents Welch’s t-stat as negative (red, down regulation) with a gradient through zero (yellow) to positive values (dark green, up regulation).
Time Course of Neonectria ditissima Infection and Production of Potential Signature Compounds
Figure 5
Figure 5. Fluorescence immunolabeling of fungal hyphae (green) using a (1,3)-β-glucan antibody in tissue sections of inoculated apple twigs. (A) Four weeks post-inoculation: hyphae are mainly visible on the outside of the apple leaf scar but some are showing penetration into the woody tissue (arrows). (B) Thirteen weeks post-inoculation: large areas of hyphal infection inside the woody apple tissue (stars).
| weeks post-inoculation | mean (mm) | range (mm) |
|---|---|---|
| 4 | 0.23 | 0–0.25 |
| 6 | 0.27 | 0–0.35 |
| 8 | 0.47 | 0–0.75 |
| 10 | 1.58 | 0–1.95 |
| 12 | 1.58 | 0.3–3 |
| 13 | 3.03 | 2.75–6 |
| distance from inoculation site (mm) | |||||
|---|---|---|---|---|---|
| weeks post-inoculation | 0–2 | 2–4 | 4–6 | 6–8 | 8–10 |
| 6 | 5 | 0 | 0 | 0 | 0 |
| 8 | 5 | 2 | 1 | 0 | 0 |
| 10 | 5 | 4 | 0 | 0 | 0 |
| 12 | 5 | 4 | 4 | 1 | 0 |
| 13 | 5 | 4 | 5 | 2 | 2 |
| 13 uninoculated control | 0 | 0 | 0 | 0 | 0 |
Figure 6
Figure 6. Styrene signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with Neonectria ditissima (at zero weeks) and uninoculated control leaf nodes of apple twigs. Peak values are rescaled to values between zero and one by dividing each peak area by the maximum observed peak area for this compound. A local polynomial spline curve was fitted through the data, with 95% error bounds giving an indication of the time point after which the control and inoculated twigs became different in their volatile compound production. The dashed red line indicates the maximum observed peak area for the uninoculated controls during the study.
Figure 7
Figure 7. Phloretin signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) inoculated with Neonectria ditissima (at zero weeks) and uninoculated control leaf nodes of apple twigs.
Summary and Future Work
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.4c10597.
Figure S1: Headspace solid-phase micro-extraction gas chromatography–mass spectroscopy total ion current chromatograms (from first experiment) of (top) an EC infected ‘Scifresh’ apple twig node; Figure S2: 4-ethylphenol (artifact from p-hydroxystyrene) signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S3: benzaldehyde signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S4: 1-benzoyl β-d-glucose signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S5: pomaceic acid signals (by LC-MS, C18, m/z [M + H]+) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S6: 2-oxopomolic acid signals (by LC-MS, C18, m/z [M + H]+) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S7: phloretin-cinnamic-hexoside-A signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Table S1: identification of significant compounds differentially detected in extracts of Neonectria ditissima-infected and control apple twigs (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We thank I. Stewart for NMR spectra and R. Atkinson for suggestions.
References
This article references 65 other publications.
- 1Flack, N.; Swinburne, T. Host range of Nectria galligena Bres. and the pathogenicity of some Northern Ireland isolates. Transactions of the British Mycological Society 1977, 68, 185– 192, DOI: 10.1016/S0007-1536(77)80007-7Google ScholarThere is no corresponding record for this reference.
- 2Walter, M.; Glaister, M. K.; Clarke, N. R.; Lutz, H. v.; Eld, Z.; Amponsah, N. T.; Shaw, N. F. Are shelter belts potential inoculum sources for Neonectria ditissima apple tree infections?. N. Z. Plant Prot. 2015, 68, 227– 240, DOI: 10.30843/nzpp.2015.68.5797Google ScholarThere is no corresponding record for this reference.
- 3Lohman, M. L.; Watson, A. J. Identity and host relations of Nectria species associated with diseases of hardwoods in the United States. Lloydia 1943, 6, 77– 108Google ScholarThere is no corresponding record for this reference.
- 4Castlebury, L. A.; Rossman, A. Y.; Hyten, A. S. Phylogenetic relationships of Neonectria/Cylindrocarpon on Fagus in North America. Canadian Journal of Botany 2006, 84, 1417– 1433, DOI: 10.1139/b06-105Google ScholarThere is no corresponding record for this reference.
- 5Zeller, S. M. European canker of pomaceous fruit trees. Station Bull. 1926, 222.Google ScholarThere is no corresponding record for this reference.
- 6Swinburne, T. R. European canker of apple (Nectria galligena). Rev. Plant Pathol. 1975, 54, 787– 799Google ScholarThere is no corresponding record for this reference.
- 7Beresford, R. M.; Kim, K. S. Identification of regional climatic conditions favorable for development of European Canker in apple. Phytopathology 2011, 101, 135– 146, DOI: 10.1094/PHYTO-05-10-0137Google ScholarThere is no corresponding record for this reference.
- 8Amponsah, N. T.; Beresford, M. W. R. M.; Scheper, R. W. A. Seasonal wound presence and susceptibility to Neonectria ditissima infection in New Zealand apple trees. N. Z. Plant Prot. 2015, 68, 250– 256, DOI: 10.30843/nzpp.2015.68.5799Google ScholarThere is no corresponding record for this reference.
- 9Campbell, R. E.; Roy, S.; Curnow, T.; Walter, M. Monitoring methods and spatial patterns of European canker disease in commercial orchards. New Zealand Plant Protection 2016, 69, 213– 220, DOI: 10.30843/nzpp.2016.69.5883Google ScholarThere is no corresponding record for this reference.
- 10Amponsah, N.; Scheper, R.; Fisher, B.; Walter, M.; Smits, J.; Jesson, L. The effect of wood age on infection by Neonectria ditissima through artificial wounds on different apple cultivars. New Zealand Plant Protection 2017, 70, 97– 105, DOI: 10.30843/nzpp.2017.70.34Google ScholarThere is no corresponding record for this reference.
- 11Dubin, H.; English, H. Factors affecting apple leaf scar infection by Nectria galligena conidia. Phytopathology 1974, 64, 1201– 1203, DOI: 10.1094/Phyto-64-1201Google ScholarThere is no corresponding record for this reference.
- 12Xu, X. M.; Butt, D. J.; Ridout, M. S. The effects of inoculum dose, duration of wet period, temperature and wound age on infection by Nectria galligena of pruning wounds on apple. Eur. J.Plant Pathol. 1998, 104, 511– 519, DOI: 10.1023/A:1008689406350Google ScholarThere is no corresponding record for this reference.
- 13Walter, M.; Roy, S.; Fisher, B.; Mackle, L.; Amponsah, N.; Curnow, T.; Campbell, R.; Braun, P.; Reineke, A.; Scheper, R. How many conidia are required for wound infection of apple plants by Neonectria ditissima. New Zealand Plant Protection 2016, 69, 238– 245, DOI: 10.30843/nzpp.2016.69.5886Google ScholarThere is no corresponding record for this reference.
- 14McCracken, A. R.; Berrie, A.; Barbara, D. J.; Locke, T.; Cooke, L. R.; Phelps, K.; Swinburne, T. R.; Brown, A. E.; Ellerker, B.; Langrell, S. R. H. Relative significance of nursery infections and orchard inoculum in the development and spread of apple canker (Nectria galligena) in young orchards. Plant Pathol. 2003, 52, 553– 566, DOI: 10.1046/j.1365-3059.2003.00924.xGoogle ScholarThere is no corresponding record for this reference.
- 15Scheper, R. W. A.; Vorster, L.; Turner, L.; Campbell, R. E.; Colhoun, K.; McArley, D.; Murti, R.; Hodson, A.; Beresford, R.; Stock, M.; Fisher, B. M.; Hedderley, D. I.; Walter, M. Lesion development and conidial production of Neonectria ditissima on apple trees in four New Zealand regions. N. Z. Plant Prot. 2019, 72, 123– 134, DOI: 10.30843/nzpp.2019.72.302Google ScholarThere is no corresponding record for this reference.
- 16Walter, M.; Stevenson, O. D.; Amponsah, N. T.; Scheper, R. W. A.; Rainham, D. G.; Hornblow, C. G.; Kerer, U.; Dryden, G. H.; Latter, I.; Butler, R. C. Control of Neonectria ditissima with copper based products in New Zealand. New Zealand Plant Protection 2015, 68, 241– 249, DOI: 10.30843/nzpp.2015.68.5798Google ScholarThere is no corresponding record for this reference.
- 17Walter, M.; Manktelow, D. W. L.; Le Berre, F.; Campbell, R. E.; Turner, L.; Vorster, L.; Patrick, E.; Butler, R. C.; Northcott, G. L. How much captan is required for wound protection of Neonectria ditissima conidial infection in apple?. New Zealand Plant Protection 2019, 72, 95– 102, DOI: 10.30843/nzpp.2019.72.273Google ScholarThere is no corresponding record for this reference.
- 18Weber, R. W. S.; Børve, J. Infection biology as the basis of integrated control of apple canker (Neonectria ditissima) in Northern Europe. CABI Agric. Biosci. 2021, 2, 1– 16, DOI: 10.1186/s43170-021-00024-zGoogle ScholarThere is no corresponding record for this reference.
- 19Saville, R.; Olivieri, L. Fungal diseases of fruit: apple cankers in Europe. In Integrated management of diseases and insect pests of tree fruit; Xu, X.; Fountain, M., Eds.; Burleigh Dodds: Cambridge, 2019; pp 59– 84.Google ScholarThere is no corresponding record for this reference.
- 20Scheper, R. W.A.; Stevenson, O.; Beresford, R. Wound protection from European canker (Neonectria galligena) infection of apple in winter and autumn. J. Plant Pathol. 2008, 90 (Suppl. 2), 368Google ScholarThere is no corresponding record for this reference.
- 21Harteveld, D. O. C.; de Jong, P. F.; Wenneker, M. Managing Neonectria ditissima at high risk steps in the chain from tree nursery to fruit growing in the Netherlands. In Book of Abstracts: 4th International Workshop on European Fruit Tree Canker and Resilient Orchards; OT Team Fruit-Bomen 2020.Google ScholarThere is no corresponding record for this reference.
- 22Harteveld, D. O. C.; Goedhart, P. W.; Houwers, I.; Köhl, J.; de Jong, P. F.; Wenneker, M. Detecting the asymptomatic colonization of apple branches by Neonectria ditissima, causing European canker of apple. Eur. J. Plant Pathol 2023, 166, 291– 301, DOI: 10.1007/s10658-023-02662-7Google ScholarThere is no corresponding record for this reference.
- 23Hung, W.-L.; Wang, Y. A Targeted Mass Spectrometry-Based Metabolomics Approach toward the Understanding of Host Responses to Huanglongbing Disease. J. Agric. Food Chem. 2018, 66, 10651– 10661, DOI: 10.1021/acs.jafc.8b04033Google ScholarThere is no corresponding record for this reference.
- 24Xu, Q.; Ma, L.; Zhou, R.; Wang, C.; Bai, J.; Sun, L.; Cai, J. Detection of huanglongbing infection in citrus using compositional analysis of volatile organic compounds. Plant Pathology 2024, 73, 2084– 2100, DOI: 10.1111/ppa.13964Google ScholarThere is no corresponding record for this reference.
- 25Liu, X.; Yi, M.; Mo, W.; Huang, Q.; Huang, Z.; Hu, B. Portable Mass Spectrometry Approach Combined with Machine Learning for Onsite Field Detection of Huanglongbing Disease. Anal. Chem. 2023, 95, 10769– 10776, DOI: 10.1021/acs.analchem.3c01825Google ScholarThere is no corresponding record for this reference.
- 26Brown, A. E.; Swinburne, T. R. Benzoic acid: an antifungal compound formed in Bramley’s seedling apple fruits following infection by Nectria galligena. Physiology and Plant Pathology 1971, 1, 469– 475, DOI: 10.1016/0048-4059(71)90009-9Google ScholarThere is no corresponding record for this reference.
- 27Gutierrez, M.; Theoduloz, C.; Rodriguez, J.; Lolas, M.; Schmeda-Hirschmann, G. Bioactive metabolites from the fungus Nectria galligena, the main apple canker agent in Chile. J. Agric. Food Chem. 2005, 53, 7701– 7708, DOI: 10.1021/jf051021lGoogle ScholarThere is no corresponding record for this reference.
- 28Zhao, K.; Yu, Y.; Xie, J.; Hu, C.; Huang, Y. Components of volatile metabolites of Nectria analysed by GC-MS. J. Southwest Univ. 2007, 29, 150– 153Google ScholarThere is no corresponding record for this reference.
- 29Knapp, D. R. Handbook of analytical derivatisation reactions; John Wiley & Sons: New York, 1979.Google ScholarThere is no corresponding record for this reference.
- 30Frank, D. L.; Zhang, A.; Leskey, T. C.; Bergh, J. C. Electrophysiological response of female Dogwood Borer (Lepidoptera: Sesiidae) to volatile compounds from apple trees. Journal of Entomological Science 2011, 46, 204– 215, DOI: 10.18474/0749-8004-46.3.204Google ScholarThere is no corresponding record for this reference.
- 31Suckling, D. M.; Twidle, A. M.; Gibb, A. R.; Manning, L. M.; Mitchell, V. J.; Sullivan, T. E. S.; Wee, S. L.; El-Sayed, A. M. Volatiles from apple trees infested with light brown apple moth larvae attract the parasitoid Dolichogenidia tasmanica. J. Agric. Food Chem. 2012, 60, 9562– 9556, DOI: 10.1021/jf302874gGoogle ScholarThere is no corresponding record for this reference.
- 32Casado, D.; Gemeno, C.; Avilla, J.; Riba, M. Day-night and phenological variation of apple tree volatiles and electroantennogram responses in Cydia pomonella (Lepidoptera: Tortricidae). Chemical Ecology 2006, 35, 258– 267, DOI: 10.1603/0046-225X-35.2.258Google ScholarThere is no corresponding record for this reference.
- 33Sutherland, P.; Hallett, I.; Jones, M. Probing cell wall structure and development by the use of antibodies: a personal perspective. N. Z. J. Forestry Sci. 2009, 39, 197– 205Google ScholarThere is no corresponding record for this reference.
- 34Hallett, I. C.; Boyd-Wilson, K. S. H.; Everett, K. R. Microscope methods for observation of the phylloplane flora. New Zealand Plant Protection 2010, 63, 15– 23, DOI: 10.30843/nzpp.2010.63.6608Google ScholarThere is no corresponding record for this reference.
- 35Ghasemkhani, M.; Holefors, A.; Marttila, S.; Dalman, K.; Zborowska, A.; Rur, M.; Rees-George, J.; Nybom, H.; Everett, K. R.; Scheper, R. W. A.; Garkava-Gustavsson, L. Real-time PCR for detection and quantification, and histological characterization of Neonectria ditissima in apple trees. Trees - Structure and Function 2016, 30, 1111– 1125, DOI: 10.1007/s00468-015-1350-9Google ScholarThere is no corresponding record for this reference.
- 36Kenward, M. G.; Roger, J. H. The precision of fixed effects estimates from restricted maximum likelihood. Biometrics 1997, 53, 983– 997, DOI: 10.2307/2533558Google ScholarThere is no corresponding record for this reference.
- 37Heimhuber, B.; Wray, V.; Galensa, R.; Herrmann, K. Benzoylglucoses from two Vaccinium species. Phytochemistry 1990, 29, 2726– 2727, DOI: 10.1016/0031-9422(90)85230-DGoogle ScholarThere is no corresponding record for this reference.
- 38Kemp, M. S.; Holloway, P. J.; Burden, R. S. 3β,19α-Dihydroxy-2-oxours-12-en-28-oic acid: a pentacyclic triterpene induced in the wood of Malus pumila Mill. infected with Chondrostereum purpureum and a constituent of the cuticular wax of apple fruits. J. Chem. Res. 1985, 1848– 1876Google ScholarThere is no corresponding record for this reference.
- 39Waldbauer, K.; Seiringer, G.; Nguyen, D. L.; Winkler, J.; Blaschke, M.; McKinnon, R.; Urban, E.; Ladurner, A.; Dirsch, V. M.; Zehl, M.; Kopp, B. Triterpenoic Acids from Apple Pomace Enhance the Activity of the Endothelial Nitric Oxide Synthase (eNOS). J. Agric. Food Chem. 2016, 64, 185– 194, DOI: 10.1021/acs.jafc.5b05061Google ScholarThere is no corresponding record for this reference.
- 40Lang, L.-J.; Wang, M.; Lei, C.; Shen, Y.; Zhu, Q.-J.; Diao, H.-M.; Chen, H.; Shen, L.; Dong, X.; Jiang, B.; Xiao, C.-J. Phloridzin Highly Accumulated in Malus rockii Rehder and Its Structure Revision and Hypolipidemic Activity. Planta Med. 2022, 88, 1190– 1198, DOI: 10.1055/a-1716-0958Google ScholarThere is no corresponding record for this reference.
- 41Anon Chromatography products for analysis and purification; USA: Supelco, 2009/2010.Google ScholarThere is no corresponding record for this reference.
- 42Sansom, C. E.; Perry, N. B. Analytical artefacts: H2 carrier gas hydrogenation of plant volatiles during headspace solid-phase microextraction gas chromatography. Phytochemical Analysis 2022, 33, 386– 391, DOI: 10.1002/pca.3096Google ScholarThere is no corresponding record for this reference.
- 43Waghmode, B.; Masoodi, L.; Kushwaha, K.; Mir, J. I.; Sircar, D. Volatile components are non-invasive biomarkers to track shelf-life and nutritional changes in apple cv. ‘Golden Delicious’ during low-temperature postharvest storage. Journal of Food Composition and Analysis 2021, 102, 104075 DOI: 10.1016/j.jfca.2021.104075Google ScholarThere is no corresponding record for this reference.
- 44Yahyaa, M.; Rachmany, D.; Shaltiel-Harpaz, L.; Nawade, B.; Sadeh, A.; Ibdah, M.; Gerchman, Y.; Holland, D.; Ibdah, M. A Pyrus communis gene for p-hydroxystyrene biosynthesis, has a role in defense against the pear psylla Cacopsylla biden. Phytochemistry 2019, 161, 107– 116, DOI: 10.1016/j.phytochem.2019.02.010Google ScholarThere is no corresponding record for this reference.
- 45D’Abrosca, B.; Fiorentino, A.; Monaco, P.; Pacifico, S. Radical-scavenging activities of new hydroxylated ursane triterpenes from cv. Annurca apples. Chem. Biodivers 2005, 2, 953– 958, DOI: 10.1002/cbdv.200590072Google ScholarThere is no corresponding record for this reference.
- 46D’Abrosca, B.; Fiorentino, A.; Monaco, P.; Oriano, P.; Pacifico, S. Annurcoic acid: A new antioxidant ursane triterpene from fruits of cv. Annurca apple. Food Chemistry 2006, 98, 285– 290, DOI: 10.1016/j.foodchem.2005.05.072Google ScholarThere is no corresponding record for this reference.
- 47He, X. J.; Liu, R. H. Triterpenoids isolated from apple peels have potent antiproliferative activity and may be partially responsible for apple’s anticancer activity. J. Agric. Food Chem. 2007, 55, 4366– 4370, DOI: 10.1021/jf063563oGoogle ScholarThere is no corresponding record for this reference.
- 48McGhie, T. K.; Hudault, S.; Lunken, R. C. M.; Christeller, J. T. Apple Peels, from Seven Cultivars, Have Lipase-Inhibitory Activity and Contain Numerous Ursenoic Acids As Identified by LC-ESI-QTOF-HRMS. J. Agric. Food Chem. 2012, 60, 482– 491, DOI: 10.1021/jf203970jGoogle ScholarThere is no corresponding record for this reference.
- 49Andre, C. M.; Greenwood, J. M.; Walker, E. G.; Rassam, M.; Sullivan, M.; Evers, D.; Perry, N. B.; Laing, W. A. Anti-inflammatory procyanidins and triterpenes in 109 apple varieties. J. Agric. Food Chem. 2012, 60, 10546– 10554, DOI: 10.1021/jf302809kGoogle ScholarThere is no corresponding record for this reference.
- 50Sut, S.; Poloniato, G.; Malagoli, M.; Dall’Acqua, S. Fragmentation of the main triterpene acids of apple by LC-APCI-MSn. J. Mass Spectrom. 2018, 53, 882– 892, DOI: 10.1002/jms.4264Google ScholarThere is no corresponding record for this reference.
- 51Resende, M. L. V.; Flood, J.; Ramsden, J. D.; Rowan, M. G.; Beale, M. H.; Cooper, R. M. Novel phytoalexins including elemental sulfur in the resistance of cocoa (Theobroma cacao L.) to verticillium wilt (Verticillium dahliae Kleb.). Physiological and Molecular Plant Pathology 1996, 48, 347– 359, DOI: 10.1006/pmpp.1996.0028Google ScholarThere is no corresponding record for this reference.
- 52El Lahlou, H.; Hirai, N.; Tsuda, M.; Ohigashi, H. Triterpene phytoalexins from nectarine fruits. Phytochemistry 1999, 52, 623– 629, DOI: 10.1016/S0031-9422(99)00316-7Google ScholarThere is no corresponding record for this reference.
- 53Hirai, N.; Sugie, M.; Wada, M.; Lahlou, E. H.; Kamo, T.; Yoshida, R.; Tsuda, M.; Ohigashi, H. Triterpene phytoalexins from strawberry fruit. Biosci., Biotechnol., Biochem. 2000, 64, 1707– 1712, DOI: 10.1271/bbb.64.1707Google ScholarThere is no corresponding record for this reference.
- 54Lahlou, E. H.; Hirai, N.; Kamo, T.; Tsuda, M.; Ohigashi, H. Actinidic acid, a new triterpene phytoalexin from unripe kiwi fruit. Biosci., Biotechnol., Biochem. 2001, 65, 480– 483, DOI: 10.1271/bbb.65.480Google ScholarThere is no corresponding record for this reference.
- 55Golding, J. B.; McGlasson, W. B.; Leach, D. N.; Wyllie, S. G. Comparison of the phenolic profiles in the peel of scalded and non scalded ″Granny Smith″ and ″Crofton″ apples. Acta Horticulturae 1998, 464, 183– 187, DOI: 10.17660/ActaHortic.1998.464.25Google ScholarThere is no corresponding record for this reference.
- 56Stingl, C.; Knapp, H.; Winterhalter, P. 3,4-Dihydroxy-7,8-dihydro-β-ionone 3-O-β-D-glucopyranoside and other glycosidic constituents from apple leaves. Natural Product Letters 2002, 16, 87– 93, DOI: 10.1080/10575630290019976Google ScholarThere is no corresponding record for this reference.
- 57Chong, J.; Pierrel, M. A.; Atanassova, R.; Werck-Reichhart, D.; Fritig, B.; Saindrenan, P. Free and conjugated benzoic acid in tobacco plants and cell cultures. induced accumulation upon elicitation of defense responses and role as salicylic acid precursors. Plant Physiol. 2001, 125, 318– 328, DOI: 10.1104/pp.125.1.318Google ScholarThere is no corresponding record for this reference.
- 58Gosch, C.; Halbwirth, H.; Stich, K. Phloridzin: Biosynthesis, distribution and physiological relevance in plants. Phytochemistry 2010, 71, 838– 843, DOI: 10.1016/j.phytochem.2010.03.003Google ScholarThere is no corresponding record for this reference.
- 59Zhao, Q.; Li, X.; Jiao, Y.; Chen, Y.; Yan, Y.; Wang, Y.; Hamiaux, C.; Wang, Y.; Ma, F.; Atkinson, R. G.; Li, P. Identification of two key genes involved in flavonoid catabolism and their different roles in apple resistance to biotic stresses. New Phytol. 2024, 242, 1238– 1256, DOI: 10.1111/nph.19644Google ScholarThere is no corresponding record for this reference.
- 60Vilanova, L.; Vinas, I.; Torres, R.; Usall, J.; Buron-Moles, G.; Teixido, N. Acidification of apple and orange hosts by Penicillium digitatum and Penicillium expansum. Int. J. Food Microbiol. 2014, 178, 39– 49, DOI: 10.1016/j.ijfoodmicro.2014.02.022Google ScholarThere is no corresponding record for this reference.
- 61Meikle, P. J.; Bonig, I.; Hoogenraad, N. J.; Clarke, A. E.; Stone, B. A. The location of (1→3)-β-glucans in the walls of pollen tubes of Nicotiana alata using a (1→3)-β-glucan-specific monoclonal antibody. Planta 1991, 185, 1– 8, DOI: 10.1007/BF00194507Google ScholarThere is no corresponding record for this reference.
- 62Dryden, G.; Nelson, M.; Smith, J.; Walter, M. Postharvest foliar nitrogen applications increase Neonectria ditissima leaf scar infection in apple trees. New Zealand Plant Protection 2016, 69, 230– 237, DOI: 10.30843/nzpp.2016.69.5885Google ScholarThere is no corresponding record for this reference.
- 63Saure, M. Der Einfluß von Wuchshemmung und Wuchsförderung auf die Krebsanfälligkeit von Apfelbäumen. Mitt. Obstbauversuchsr Alt. Land. 1963, 18, 54– 58Google ScholarThere is no corresponding record for this reference.
- 64Gaucher, M.; Dugé de Bernonville, T.; Lohou, D.; Guyot, S.; Guillemette, T.; Brisset, M.-N.; Dat, J. F. Histolocalization and physico-chemical characterization of dihydrochalcones: Insight into the role of apple major flavonoids. Phytochemistry 2013, 90, 78– 89, DOI: 10.1016/j.phytochem.2013.02.009Google ScholarThere is no corresponding record for this reference.
- 65Enaud, E.; Humeau, C.; Piffaut, B.; Girardin, M. Enzymatic synthesis of new aromatic esters of phloridzin. J. Mol. Catal. B: Enzym. 2004, 27, 1– 6, DOI: 10.1016/j.molcatb.2003.08.002Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
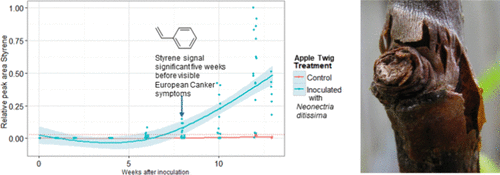
Figure 1

Figure 1. (A) Apple twig leaf scar, with leaf bud above (image by Tony Corbett, PFR). (B) Apple twig with visible European Canker infection. (C) Cross-section of infected apple twig leaf scar showing Neonectria ditissima symptoms (brown tissue at the top).
Figure 2

Figure 2. (A) Two-dimensional projection of principal components analysis of headspace solid-phase micro-extraction GC-MS data of apple twigs showing the variability between different treatments. Red triangles = ‘Scifresh’ inoculated, purple squares = ‘Scifresh’ control, green triangles = ‘Royal Gala’ inoculated, blue squares = ‘Royal Gala’ control from Motueka, light blue squares = ‘Royal Gala’ control from Hawke’s Bay. (B) Loading plot for PC-1, showing positive loadings corresponding to peaks for hexanol and styrene, and negative loadings for sesquiterpenes. The x-axis numbers correspond to GC retention time (min).
Figure 3

Figure 3. Structures of potential signature compounds for Neonectria ditissima infection of apple twigs.
Figure 4
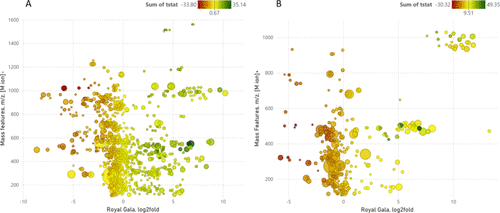
Figure 4. LC-MS differential plots for all component mass features that changed between infected and control ‘Royal Gala’ apple twigs by reverse phase liquid chromatography with negative ion (A) and positive ion (B) MS. Plot x-axis log2fold change (negative values indicate down regulation and positive values up regulation due to infection); y-axis ion species mass features as m/z (compounds represented by one or more signal ions M, 2M, M+ adduct, etc.); marker dimension is the maximum area response for an ion species mass feature; color represents Welch’s t-stat as negative (red, down regulation) with a gradient through zero (yellow) to positive values (dark green, up regulation).
Figure 5
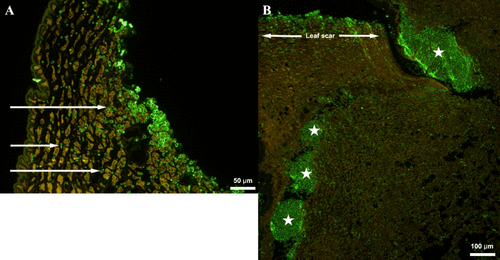
Figure 5. Fluorescence immunolabeling of fungal hyphae (green) using a (1,3)-β-glucan antibody in tissue sections of inoculated apple twigs. (A) Four weeks post-inoculation: hyphae are mainly visible on the outside of the apple leaf scar but some are showing penetration into the woody tissue (arrows). (B) Thirteen weeks post-inoculation: large areas of hyphal infection inside the woody apple tissue (stars).
Figure 6
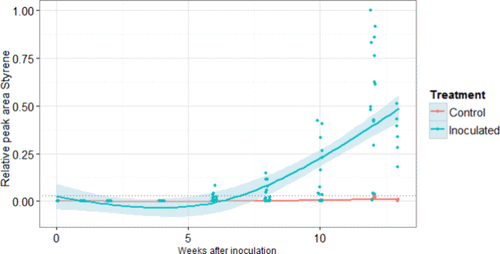
Figure 6. Styrene signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with Neonectria ditissima (at zero weeks) and uninoculated control leaf nodes of apple twigs. Peak values are rescaled to values between zero and one by dividing each peak area by the maximum observed peak area for this compound. A local polynomial spline curve was fitted through the data, with 95% error bounds giving an indication of the time point after which the control and inoculated twigs became different in their volatile compound production. The dashed red line indicates the maximum observed peak area for the uninoculated controls during the study.
Figure 7

Figure 7. Phloretin signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) inoculated with Neonectria ditissima (at zero weeks) and uninoculated control leaf nodes of apple twigs.
References
This article references 65 other publications.
- 1Flack, N.; Swinburne, T. Host range of Nectria galligena Bres. and the pathogenicity of some Northern Ireland isolates. Transactions of the British Mycological Society 1977, 68, 185– 192, DOI: 10.1016/S0007-1536(77)80007-7There is no corresponding record for this reference.
- 2Walter, M.; Glaister, M. K.; Clarke, N. R.; Lutz, H. v.; Eld, Z.; Amponsah, N. T.; Shaw, N. F. Are shelter belts potential inoculum sources for Neonectria ditissima apple tree infections?. N. Z. Plant Prot. 2015, 68, 227– 240, DOI: 10.30843/nzpp.2015.68.5797There is no corresponding record for this reference.
- 3Lohman, M. L.; Watson, A. J. Identity and host relations of Nectria species associated with diseases of hardwoods in the United States. Lloydia 1943, 6, 77– 108There is no corresponding record for this reference.
- 4Castlebury, L. A.; Rossman, A. Y.; Hyten, A. S. Phylogenetic relationships of Neonectria/Cylindrocarpon on Fagus in North America. Canadian Journal of Botany 2006, 84, 1417– 1433, DOI: 10.1139/b06-105There is no corresponding record for this reference.
- 5Zeller, S. M. European canker of pomaceous fruit trees. Station Bull. 1926, 222.There is no corresponding record for this reference.
- 6Swinburne, T. R. European canker of apple (Nectria galligena). Rev. Plant Pathol. 1975, 54, 787– 799There is no corresponding record for this reference.
- 7Beresford, R. M.; Kim, K. S. Identification of regional climatic conditions favorable for development of European Canker in apple. Phytopathology 2011, 101, 135– 146, DOI: 10.1094/PHYTO-05-10-0137There is no corresponding record for this reference.
- 8Amponsah, N. T.; Beresford, M. W. R. M.; Scheper, R. W. A. Seasonal wound presence and susceptibility to Neonectria ditissima infection in New Zealand apple trees. N. Z. Plant Prot. 2015, 68, 250– 256, DOI: 10.30843/nzpp.2015.68.5799There is no corresponding record for this reference.
- 9Campbell, R. E.; Roy, S.; Curnow, T.; Walter, M. Monitoring methods and spatial patterns of European canker disease in commercial orchards. New Zealand Plant Protection 2016, 69, 213– 220, DOI: 10.30843/nzpp.2016.69.5883There is no corresponding record for this reference.
- 10Amponsah, N.; Scheper, R.; Fisher, B.; Walter, M.; Smits, J.; Jesson, L. The effect of wood age on infection by Neonectria ditissima through artificial wounds on different apple cultivars. New Zealand Plant Protection 2017, 70, 97– 105, DOI: 10.30843/nzpp.2017.70.34There is no corresponding record for this reference.
- 11Dubin, H.; English, H. Factors affecting apple leaf scar infection by Nectria galligena conidia. Phytopathology 1974, 64, 1201– 1203, DOI: 10.1094/Phyto-64-1201There is no corresponding record for this reference.
- 12Xu, X. M.; Butt, D. J.; Ridout, M. S. The effects of inoculum dose, duration of wet period, temperature and wound age on infection by Nectria galligena of pruning wounds on apple. Eur. J.Plant Pathol. 1998, 104, 511– 519, DOI: 10.1023/A:1008689406350There is no corresponding record for this reference.
- 13Walter, M.; Roy, S.; Fisher, B.; Mackle, L.; Amponsah, N.; Curnow, T.; Campbell, R.; Braun, P.; Reineke, A.; Scheper, R. How many conidia are required for wound infection of apple plants by Neonectria ditissima. New Zealand Plant Protection 2016, 69, 238– 245, DOI: 10.30843/nzpp.2016.69.5886There is no corresponding record for this reference.
- 14McCracken, A. R.; Berrie, A.; Barbara, D. J.; Locke, T.; Cooke, L. R.; Phelps, K.; Swinburne, T. R.; Brown, A. E.; Ellerker, B.; Langrell, S. R. H. Relative significance of nursery infections and orchard inoculum in the development and spread of apple canker (Nectria galligena) in young orchards. Plant Pathol. 2003, 52, 553– 566, DOI: 10.1046/j.1365-3059.2003.00924.xThere is no corresponding record for this reference.
- 15Scheper, R. W. A.; Vorster, L.; Turner, L.; Campbell, R. E.; Colhoun, K.; McArley, D.; Murti, R.; Hodson, A.; Beresford, R.; Stock, M.; Fisher, B. M.; Hedderley, D. I.; Walter, M. Lesion development and conidial production of Neonectria ditissima on apple trees in four New Zealand regions. N. Z. Plant Prot. 2019, 72, 123– 134, DOI: 10.30843/nzpp.2019.72.302There is no corresponding record for this reference.
- 16Walter, M.; Stevenson, O. D.; Amponsah, N. T.; Scheper, R. W. A.; Rainham, D. G.; Hornblow, C. G.; Kerer, U.; Dryden, G. H.; Latter, I.; Butler, R. C. Control of Neonectria ditissima with copper based products in New Zealand. New Zealand Plant Protection 2015, 68, 241– 249, DOI: 10.30843/nzpp.2015.68.5798There is no corresponding record for this reference.
- 17Walter, M.; Manktelow, D. W. L.; Le Berre, F.; Campbell, R. E.; Turner, L.; Vorster, L.; Patrick, E.; Butler, R. C.; Northcott, G. L. How much captan is required for wound protection of Neonectria ditissima conidial infection in apple?. New Zealand Plant Protection 2019, 72, 95– 102, DOI: 10.30843/nzpp.2019.72.273There is no corresponding record for this reference.
- 18Weber, R. W. S.; Børve, J. Infection biology as the basis of integrated control of apple canker (Neonectria ditissima) in Northern Europe. CABI Agric. Biosci. 2021, 2, 1– 16, DOI: 10.1186/s43170-021-00024-zThere is no corresponding record for this reference.
- 19Saville, R.; Olivieri, L. Fungal diseases of fruit: apple cankers in Europe. In Integrated management of diseases and insect pests of tree fruit; Xu, X.; Fountain, M., Eds.; Burleigh Dodds: Cambridge, 2019; pp 59– 84.There is no corresponding record for this reference.
- 20Scheper, R. W.A.; Stevenson, O.; Beresford, R. Wound protection from European canker (Neonectria galligena) infection of apple in winter and autumn. J. Plant Pathol. 2008, 90 (Suppl. 2), 368There is no corresponding record for this reference.
- 21Harteveld, D. O. C.; de Jong, P. F.; Wenneker, M. Managing Neonectria ditissima at high risk steps in the chain from tree nursery to fruit growing in the Netherlands. In Book of Abstracts: 4th International Workshop on European Fruit Tree Canker and Resilient Orchards; OT Team Fruit-Bomen 2020.There is no corresponding record for this reference.
- 22Harteveld, D. O. C.; Goedhart, P. W.; Houwers, I.; Köhl, J.; de Jong, P. F.; Wenneker, M. Detecting the asymptomatic colonization of apple branches by Neonectria ditissima, causing European canker of apple. Eur. J. Plant Pathol 2023, 166, 291– 301, DOI: 10.1007/s10658-023-02662-7There is no corresponding record for this reference.
- 23Hung, W.-L.; Wang, Y. A Targeted Mass Spectrometry-Based Metabolomics Approach toward the Understanding of Host Responses to Huanglongbing Disease. J. Agric. Food Chem. 2018, 66, 10651– 10661, DOI: 10.1021/acs.jafc.8b04033There is no corresponding record for this reference.
- 24Xu, Q.; Ma, L.; Zhou, R.; Wang, C.; Bai, J.; Sun, L.; Cai, J. Detection of huanglongbing infection in citrus using compositional analysis of volatile organic compounds. Plant Pathology 2024, 73, 2084– 2100, DOI: 10.1111/ppa.13964There is no corresponding record for this reference.
- 25Liu, X.; Yi, M.; Mo, W.; Huang, Q.; Huang, Z.; Hu, B. Portable Mass Spectrometry Approach Combined with Machine Learning for Onsite Field Detection of Huanglongbing Disease. Anal. Chem. 2023, 95, 10769– 10776, DOI: 10.1021/acs.analchem.3c01825There is no corresponding record for this reference.
- 26Brown, A. E.; Swinburne, T. R. Benzoic acid: an antifungal compound formed in Bramley’s seedling apple fruits following infection by Nectria galligena. Physiology and Plant Pathology 1971, 1, 469– 475, DOI: 10.1016/0048-4059(71)90009-9There is no corresponding record for this reference.
- 27Gutierrez, M.; Theoduloz, C.; Rodriguez, J.; Lolas, M.; Schmeda-Hirschmann, G. Bioactive metabolites from the fungus Nectria galligena, the main apple canker agent in Chile. J. Agric. Food Chem. 2005, 53, 7701– 7708, DOI: 10.1021/jf051021lThere is no corresponding record for this reference.
- 28Zhao, K.; Yu, Y.; Xie, J.; Hu, C.; Huang, Y. Components of volatile metabolites of Nectria analysed by GC-MS. J. Southwest Univ. 2007, 29, 150– 153There is no corresponding record for this reference.
- 29Knapp, D. R. Handbook of analytical derivatisation reactions; John Wiley & Sons: New York, 1979.There is no corresponding record for this reference.
- 30Frank, D. L.; Zhang, A.; Leskey, T. C.; Bergh, J. C. Electrophysiological response of female Dogwood Borer (Lepidoptera: Sesiidae) to volatile compounds from apple trees. Journal of Entomological Science 2011, 46, 204– 215, DOI: 10.18474/0749-8004-46.3.204There is no corresponding record for this reference.
- 31Suckling, D. M.; Twidle, A. M.; Gibb, A. R.; Manning, L. M.; Mitchell, V. J.; Sullivan, T. E. S.; Wee, S. L.; El-Sayed, A. M. Volatiles from apple trees infested with light brown apple moth larvae attract the parasitoid Dolichogenidia tasmanica. J. Agric. Food Chem. 2012, 60, 9562– 9556, DOI: 10.1021/jf302874gThere is no corresponding record for this reference.
- 32Casado, D.; Gemeno, C.; Avilla, J.; Riba, M. Day-night and phenological variation of apple tree volatiles and electroantennogram responses in Cydia pomonella (Lepidoptera: Tortricidae). Chemical Ecology 2006, 35, 258– 267, DOI: 10.1603/0046-225X-35.2.258There is no corresponding record for this reference.
- 33Sutherland, P.; Hallett, I.; Jones, M. Probing cell wall structure and development by the use of antibodies: a personal perspective. N. Z. J. Forestry Sci. 2009, 39, 197– 205There is no corresponding record for this reference.
- 34Hallett, I. C.; Boyd-Wilson, K. S. H.; Everett, K. R. Microscope methods for observation of the phylloplane flora. New Zealand Plant Protection 2010, 63, 15– 23, DOI: 10.30843/nzpp.2010.63.6608There is no corresponding record for this reference.
- 35Ghasemkhani, M.; Holefors, A.; Marttila, S.; Dalman, K.; Zborowska, A.; Rur, M.; Rees-George, J.; Nybom, H.; Everett, K. R.; Scheper, R. W. A.; Garkava-Gustavsson, L. Real-time PCR for detection and quantification, and histological characterization of Neonectria ditissima in apple trees. Trees - Structure and Function 2016, 30, 1111– 1125, DOI: 10.1007/s00468-015-1350-9There is no corresponding record for this reference.
- 36Kenward, M. G.; Roger, J. H. The precision of fixed effects estimates from restricted maximum likelihood. Biometrics 1997, 53, 983– 997, DOI: 10.2307/2533558There is no corresponding record for this reference.
- 37Heimhuber, B.; Wray, V.; Galensa, R.; Herrmann, K. Benzoylglucoses from two Vaccinium species. Phytochemistry 1990, 29, 2726– 2727, DOI: 10.1016/0031-9422(90)85230-DThere is no corresponding record for this reference.
- 38Kemp, M. S.; Holloway, P. J.; Burden, R. S. 3β,19α-Dihydroxy-2-oxours-12-en-28-oic acid: a pentacyclic triterpene induced in the wood of Malus pumila Mill. infected with Chondrostereum purpureum and a constituent of the cuticular wax of apple fruits. J. Chem. Res. 1985, 1848– 1876There is no corresponding record for this reference.
- 39Waldbauer, K.; Seiringer, G.; Nguyen, D. L.; Winkler, J.; Blaschke, M.; McKinnon, R.; Urban, E.; Ladurner, A.; Dirsch, V. M.; Zehl, M.; Kopp, B. Triterpenoic Acids from Apple Pomace Enhance the Activity of the Endothelial Nitric Oxide Synthase (eNOS). J. Agric. Food Chem. 2016, 64, 185– 194, DOI: 10.1021/acs.jafc.5b05061There is no corresponding record for this reference.
- 40Lang, L.-J.; Wang, M.; Lei, C.; Shen, Y.; Zhu, Q.-J.; Diao, H.-M.; Chen, H.; Shen, L.; Dong, X.; Jiang, B.; Xiao, C.-J. Phloridzin Highly Accumulated in Malus rockii Rehder and Its Structure Revision and Hypolipidemic Activity. Planta Med. 2022, 88, 1190– 1198, DOI: 10.1055/a-1716-0958There is no corresponding record for this reference.
- 41Anon Chromatography products for analysis and purification; USA: Supelco, 2009/2010.There is no corresponding record for this reference.
- 42Sansom, C. E.; Perry, N. B. Analytical artefacts: H2 carrier gas hydrogenation of plant volatiles during headspace solid-phase microextraction gas chromatography. Phytochemical Analysis 2022, 33, 386– 391, DOI: 10.1002/pca.3096There is no corresponding record for this reference.
- 43Waghmode, B.; Masoodi, L.; Kushwaha, K.; Mir, J. I.; Sircar, D. Volatile components are non-invasive biomarkers to track shelf-life and nutritional changes in apple cv. ‘Golden Delicious’ during low-temperature postharvest storage. Journal of Food Composition and Analysis 2021, 102, 104075 DOI: 10.1016/j.jfca.2021.104075There is no corresponding record for this reference.
- 44Yahyaa, M.; Rachmany, D.; Shaltiel-Harpaz, L.; Nawade, B.; Sadeh, A.; Ibdah, M.; Gerchman, Y.; Holland, D.; Ibdah, M. A Pyrus communis gene for p-hydroxystyrene biosynthesis, has a role in defense against the pear psylla Cacopsylla biden. Phytochemistry 2019, 161, 107– 116, DOI: 10.1016/j.phytochem.2019.02.010There is no corresponding record for this reference.
- 45D’Abrosca, B.; Fiorentino, A.; Monaco, P.; Pacifico, S. Radical-scavenging activities of new hydroxylated ursane triterpenes from cv. Annurca apples. Chem. Biodivers 2005, 2, 953– 958, DOI: 10.1002/cbdv.200590072There is no corresponding record for this reference.
- 46D’Abrosca, B.; Fiorentino, A.; Monaco, P.; Oriano, P.; Pacifico, S. Annurcoic acid: A new antioxidant ursane triterpene from fruits of cv. Annurca apple. Food Chemistry 2006, 98, 285– 290, DOI: 10.1016/j.foodchem.2005.05.072There is no corresponding record for this reference.
- 47He, X. J.; Liu, R. H. Triterpenoids isolated from apple peels have potent antiproliferative activity and may be partially responsible for apple’s anticancer activity. J. Agric. Food Chem. 2007, 55, 4366– 4370, DOI: 10.1021/jf063563oThere is no corresponding record for this reference.
- 48McGhie, T. K.; Hudault, S.; Lunken, R. C. M.; Christeller, J. T. Apple Peels, from Seven Cultivars, Have Lipase-Inhibitory Activity and Contain Numerous Ursenoic Acids As Identified by LC-ESI-QTOF-HRMS. J. Agric. Food Chem. 2012, 60, 482– 491, DOI: 10.1021/jf203970jThere is no corresponding record for this reference.
- 49Andre, C. M.; Greenwood, J. M.; Walker, E. G.; Rassam, M.; Sullivan, M.; Evers, D.; Perry, N. B.; Laing, W. A. Anti-inflammatory procyanidins and triterpenes in 109 apple varieties. J. Agric. Food Chem. 2012, 60, 10546– 10554, DOI: 10.1021/jf302809kThere is no corresponding record for this reference.
- 50Sut, S.; Poloniato, G.; Malagoli, M.; Dall’Acqua, S. Fragmentation of the main triterpene acids of apple by LC-APCI-MSn. J. Mass Spectrom. 2018, 53, 882– 892, DOI: 10.1002/jms.4264There is no corresponding record for this reference.
- 51Resende, M. L. V.; Flood, J.; Ramsden, J. D.; Rowan, M. G.; Beale, M. H.; Cooper, R. M. Novel phytoalexins including elemental sulfur in the resistance of cocoa (Theobroma cacao L.) to verticillium wilt (Verticillium dahliae Kleb.). Physiological and Molecular Plant Pathology 1996, 48, 347– 359, DOI: 10.1006/pmpp.1996.0028There is no corresponding record for this reference.
- 52El Lahlou, H.; Hirai, N.; Tsuda, M.; Ohigashi, H. Triterpene phytoalexins from nectarine fruits. Phytochemistry 1999, 52, 623– 629, DOI: 10.1016/S0031-9422(99)00316-7There is no corresponding record for this reference.
- 53Hirai, N.; Sugie, M.; Wada, M.; Lahlou, E. H.; Kamo, T.; Yoshida, R.; Tsuda, M.; Ohigashi, H. Triterpene phytoalexins from strawberry fruit. Biosci., Biotechnol., Biochem. 2000, 64, 1707– 1712, DOI: 10.1271/bbb.64.1707There is no corresponding record for this reference.
- 54Lahlou, E. H.; Hirai, N.; Kamo, T.; Tsuda, M.; Ohigashi, H. Actinidic acid, a new triterpene phytoalexin from unripe kiwi fruit. Biosci., Biotechnol., Biochem. 2001, 65, 480– 483, DOI: 10.1271/bbb.65.480There is no corresponding record for this reference.
- 55Golding, J. B.; McGlasson, W. B.; Leach, D. N.; Wyllie, S. G. Comparison of the phenolic profiles in the peel of scalded and non scalded ″Granny Smith″ and ″Crofton″ apples. Acta Horticulturae 1998, 464, 183– 187, DOI: 10.17660/ActaHortic.1998.464.25There is no corresponding record for this reference.
- 56Stingl, C.; Knapp, H.; Winterhalter, P. 3,4-Dihydroxy-7,8-dihydro-β-ionone 3-O-β-D-glucopyranoside and other glycosidic constituents from apple leaves. Natural Product Letters 2002, 16, 87– 93, DOI: 10.1080/10575630290019976There is no corresponding record for this reference.
- 57Chong, J.; Pierrel, M. A.; Atanassova, R.; Werck-Reichhart, D.; Fritig, B.; Saindrenan, P. Free and conjugated benzoic acid in tobacco plants and cell cultures. induced accumulation upon elicitation of defense responses and role as salicylic acid precursors. Plant Physiol. 2001, 125, 318– 328, DOI: 10.1104/pp.125.1.318There is no corresponding record for this reference.
- 58Gosch, C.; Halbwirth, H.; Stich, K. Phloridzin: Biosynthesis, distribution and physiological relevance in plants. Phytochemistry 2010, 71, 838– 843, DOI: 10.1016/j.phytochem.2010.03.003There is no corresponding record for this reference.
- 59Zhao, Q.; Li, X.; Jiao, Y.; Chen, Y.; Yan, Y.; Wang, Y.; Hamiaux, C.; Wang, Y.; Ma, F.; Atkinson, R. G.; Li, P. Identification of two key genes involved in flavonoid catabolism and their different roles in apple resistance to biotic stresses. New Phytol. 2024, 242, 1238– 1256, DOI: 10.1111/nph.19644There is no corresponding record for this reference.
- 60Vilanova, L.; Vinas, I.; Torres, R.; Usall, J.; Buron-Moles, G.; Teixido, N. Acidification of apple and orange hosts by Penicillium digitatum and Penicillium expansum. Int. J. Food Microbiol. 2014, 178, 39– 49, DOI: 10.1016/j.ijfoodmicro.2014.02.022There is no corresponding record for this reference.
- 61Meikle, P. J.; Bonig, I.; Hoogenraad, N. J.; Clarke, A. E.; Stone, B. A. The location of (1→3)-β-glucans in the walls of pollen tubes of Nicotiana alata using a (1→3)-β-glucan-specific monoclonal antibody. Planta 1991, 185, 1– 8, DOI: 10.1007/BF00194507There is no corresponding record for this reference.
- 62Dryden, G.; Nelson, M.; Smith, J.; Walter, M. Postharvest foliar nitrogen applications increase Neonectria ditissima leaf scar infection in apple trees. New Zealand Plant Protection 2016, 69, 230– 237, DOI: 10.30843/nzpp.2016.69.5885There is no corresponding record for this reference.
- 63Saure, M. Der Einfluß von Wuchshemmung und Wuchsförderung auf die Krebsanfälligkeit von Apfelbäumen. Mitt. Obstbauversuchsr Alt. Land. 1963, 18, 54– 58There is no corresponding record for this reference.
- 64Gaucher, M.; Dugé de Bernonville, T.; Lohou, D.; Guyot, S.; Guillemette, T.; Brisset, M.-N.; Dat, J. F. Histolocalization and physico-chemical characterization of dihydrochalcones: Insight into the role of apple major flavonoids. Phytochemistry 2013, 90, 78– 89, DOI: 10.1016/j.phytochem.2013.02.009There is no corresponding record for this reference.
- 65Enaud, E.; Humeau, C.; Piffaut, B.; Girardin, M. Enzymatic synthesis of new aromatic esters of phloridzin. J. Mol. Catal. B: Enzym. 2004, 27, 1– 6, DOI: 10.1016/j.molcatb.2003.08.002There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.4c10597.
Figure S1: Headspace solid-phase micro-extraction gas chromatography–mass spectroscopy total ion current chromatograms (from first experiment) of (top) an EC infected ‘Scifresh’ apple twig node; Figure S2: 4-ethylphenol (artifact from p-hydroxystyrene) signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S3: benzaldehyde signals (by HS-SPME-GC-MS) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S4: 1-benzoyl β-d-glucose signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S5: pomaceic acid signals (by LC-MS, C18, m/z [M + H]+) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S6: 2-oxopomolic acid signals (by LC-MS, C18, m/z [M + H]+) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Figure S7: phloretin-cinnamic-hexoside-A signals (by LC-MS, C18, m/z [M-H]−) from leaf nodes (ground) of apple twigs inoculated with European Canker (at time 0 weeks) and controls; Table S1: identification of significant compounds differentially detected in extracts of Neonectria ditissima-infected and control apple twigs (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


