
Synthesis and Identification of 3-Oxazolines in CocoaClick to copy article linkArticle link copied!
- Heather G. SpoonerHeather G. SpoonerDepartment of Food and Nutritional Sciences, University of Reading, Whiteknights, Reading RG6 6AP, U.K.More by Heather G. Spooner
- Dimitris P. BalagiannisDimitris P. BalagiannisDepartment of Food and Nutritional Sciences, University of Reading, Whiteknights, Reading RG6 6AP, U.K.More by Dimitris P. Balagiannis
- Andreas CzepaAndreas CzepaMondele̅z International, Whiteknights Campus, Pepper Lane, Reading RG6 6LA, U.K.More by Andreas Czepa
- Barbara SuessBarbara SuessMondele̅z International, Whiteknights Campus, Pepper Lane, Reading RG6 6LA, U.K.More by Barbara Suess
- Martine TrotinMartine TrotinMondele̅z International, Whiteknights Campus, Pepper Lane, Reading RG6 6LA, U.K.More by Martine Trotin
- Paul O’NionPaul O’NionReading Scientific Services Ltd, Whiteknights Campus, Pepper Lane, Reading RG6 6LA, U.K.More by Paul O’Nion
- Jane K. Parker*Jane K. Parker*E-mail: [email protected]. Tel: + 44 118 378 7455.Department of Food and Nutritional Sciences, University of Reading, Whiteknights, Reading RG6 6AP, U.K.More by Jane K. Parker
Abstract
Adding water to chocolate is known to cause a large increase in the concentration of Strecker aldehydes, which are key aroma compounds in cocoa. 3-Oxazolines may be precursors responsible for this; however, only a low concentration of 2-isobutyl-5-methyl-3-oxazoline was previously identified in chocolate. This study investigates the possibility that other types of 3-oxazolines are the relevant precursors present in cocoa. A range of novel 3-oxazolines were synthesized and characterized by gas chromatography–mass spectrometry (GC–MS) and nuclear magnetic resonance spectroscopy. Using the synthesized compounds as references, four of these were identified by GC–MS for the first time in aroma extracts of cacao nibs, cocoa liquor, and chocolate, obtained by solvent-assisted flavor evaporation. This study may reveal a new focus for enhancing cocoa aroma and potentially other roasted food products as well.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Special Issue
Published as part of Journal of Agricultural and Food Chemistry special issue “17th International Weurman Flavour Research Symposium: From Flavour Generation to Flavour Perception, Analytics, Modelling and Health”.
Introduction
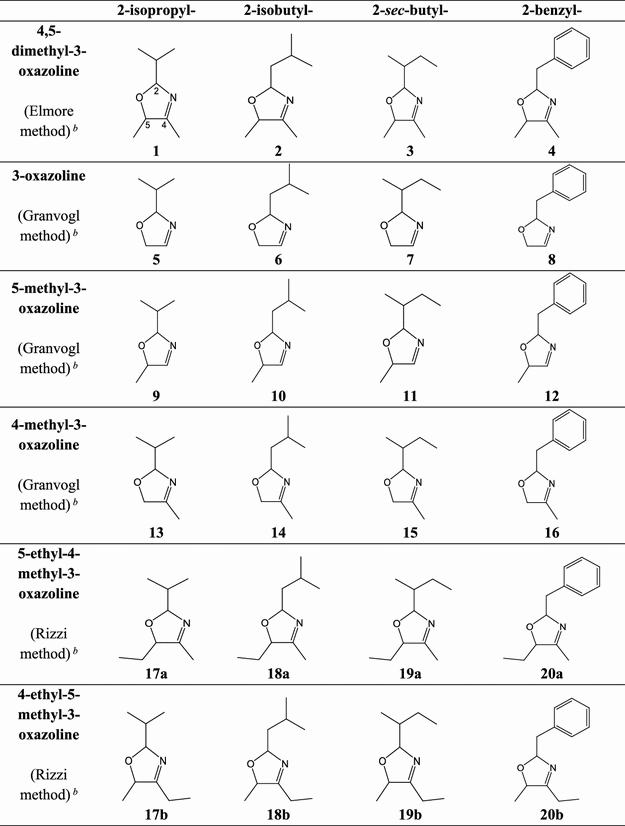
The 3-oxazolines were selected on the basis of the substituent of the carbon atom at position 2 (denoted C2, labeled in compound 1), giving rise to the Strecker aldehydes predominant in cocoa (2-methylpropanal, 3-methylbutanal, 2-methylbutanal, and phenylacetaldehyde) (22) and the C4 and C5 substituents arising from α-dicarbonyl compounds commonly found in food (see mechanism in Figure S1 ).
The preparation method of each 3-oxazoline depended on the class of oxazoline, defined by the C4 and C5 substituents. For details of each synthetic method, see Figure 1.
Materials and Methods
Materials
Food Samples
Chemicals
Synthetic Methods and Characterization of 3-Oxazolines
Synthesis of 4,5-Dimethyl-3-oxazolines (1–4) (See Table 1)
2-Isopropyl-4,5-dimethyl-3-oxazoline (1)
2-Isobutyl-4,5-dimethyl-3-oxazoline (2)
2-sec-Butyl-4,5-dimethyl-3-oxazoline (3)
2-Benzyl-4,5-dimethyl-3-oxazoline (4)
Synthesis of 3-Oxazolines, 5-Methyl-3-oxazolines, and 4-Methyl-3-oxazolines (5–16)
Synthesis of 4/5-Ethyl–4/5-Methyl-3-oxazolines (17–20)
Extraction and Identification of 3-Oxazolines in Cacao Nibs, Cocoa Liquor, and Chocolate
SAFE Extraction (18)
Identification by GC–MS
Two-Dimensional Gas Chromatography–Mass Spectrometry (2D-GC–MS)
Gas Chromatography–Olfactometry (GC-O)
NMR Spectroscopy
Results and Discussion
Synthesis and Characterization of 4,5-Dimethyl-3-oxazolines (1–4)
Figure 1
Figure 1. Examples of the three synthetic routes used to generate 3-oxazolines. (A) The Elmore method, used for the preparation of 1–4, adapted from 3-thiazoline synthesis, reported by Elmore and Mottram. (17) (B) The Granvogl method, used for the preparation of 5–16, adapted from the synthesis for 2-substituted-5-methyl-3-oxazolines, reported by Granvogl et al. (1) The amino alcohol reagent was varied in order to control the substituents at C4 and C5. (C) The Rizzi method, used for the preparation of 17–20, adapted from the synthesis for 2-isopropyl-4,5-dimethyl-3-oxazoline, reported by Rizzi. (16)
Figure 2
Figure 2. Stereoisomers of 2-isobutyl-4,5-dimethyl-3-oxazoline (2). (A) Two chiral centers, indicated by asterisks, gives rise to four stereoisomers, existing as two pairs of enantiomers that are diastereomers of each other, assigned as (S) or (R) by the Cahn–Ingold–Prelog rules. (B) All four stereoisomers were observed on a chiral GC–MS column (CP-Chirasil-Dex CB).
| LRI | MS fragmentation (EI)a | [M + H] exact mass (CI)b | ||||
|---|---|---|---|---|---|---|
| Compound | HP-5 | ZB-Wax | m/z (% relative intensity cf. base peak) | theoretical | observed | |
| 1 | 2-isopropyl-4,5-dimethyl-3-oxazoline | 954, 964 | 1244, 1253 | 98, 43 (44), 71 (39), 97 (26), 56 (22), 41 (16), 42 (13), 82 (9), 39 (8), 140 (tr), 141 (tr) | 142.1226 | 142.1230, 142.1231 |
| 2 | 2-isobutyl-4,5-dimethyl-3-oxazoline | 1054, 1057 | 1354, 1356 | 98, 71 (39), 43 (31), 99 (24), 42 (13), 68 (13), 41 (11), 114 (7), 39 (7), 154 (tr), 155 (tr) | 156.1383 | 156.1386, 156.1387 |
| 3 | 2-sec-butyl-4,5-dimethyl-3-oxazoline | 1057, 1067 | 1354, 1363 | 98, 71 (33), 43 (29), 97 (14), 70 (13), 42 (12), 41 (11), 55 (10), 99 (8), 154 (tr), 155 (tr) | 156.1383 | 156.1384, 156.1386 |
| 4 | 2-benzyl-4,5-dimethyl-3-oxazoline | 1416, 1442 | 2019, 2052 | 98, 71 (37), 43 (37), 91 (26), 97 (26), 65 (9), 77 (8), 103 (7), 99 (6), 187 (tr), 189 (tr) | 190.1226 | 190.1237, 190.1236 |
| 5 | 2-isopropyl-3-oxazoline | 859 | 1245 | 70, 43 (60), 42 (58), 41 (47), 71 (34), 69 (24), 39 (22), 56 (21), 98 (15), 112 (2), 113 (2) | 114.0913 | 114.0913 |
| 6 | 2-isobutyl-3-oxazoline | 958 | 1352 | 70, 42 (34), 41 (26), 69 (25), 71 (24), 43 (22), 85 (18), 54 (16), 39 (13), 126 (1), 127 (tr) | 128.1070 | 128.1070 |
| 7 | 2-sec-butyl-3-oxazoline | 968 | 1334 | 70, 71 (74), 72 (64), 43 (58), 41 (55), 42 (51), 98 (48), 29 (27), 39 (23), 126 (1), 127 (tr) | 128.1070 | 128.1071 |
| 8 | 2-benzyl-3-oxazoline | 1351 | 2026 | 91, 92 (62), 70 (41), 131 (17), 65 (16), 77 (8), 104 (8), 51 (8), 132 (8), 160 (2), 161 (5) | 162.0913 | 162.0924 |
| 9 | 2-isopropyl-5-methyl-3-oxazoline | 889, 891 | 1197, 1200 | 84, 56 (49), 57 (46), 112 (40), 83 (28), 41 (25), 70 (18), 68 (17), 43 (16), 126 (2), 127 (3) | 128.1070 | 128.1070, 128.1070 |
| 10 | 2-isobutyl-5-methyl-3-oxazoline | 982, 984 | 1306, 1316 | 84, 57 (35), 41 (22), 43 (21), 54 (20), 56 (17), 85 (13), 39 (12), 82 (11), 140 (1), 141 (tr) | 142.1226 | 142.1223, 142.1225 |
| 11 | 2-sec-butyl-5-methyl-3-oxazoline | 994, 998 | 1306, 1307 | 84, 56 (57), 57 (50), 112 (40), 70 (33), 41 (30), 85 (28), 29 (20), 68 (19), 140 (1), 141 (1) | 142.1226 | 142.1228, 142.1225 |
| 12 | 2-benzyl-5-methyl-3-oxazoline | 1370, 1380 | 1972, 1979 | 84, 91 (64), 92 (38), 57 (35), 65 (13), 131 (12), 104 (10), 77 (9), 103 (8), 174 (tr), 175 (3) | 176.1070 | 176.1073, 176.1071 |
| 13 | 2-isopropyl-4-methyl-3-oxazoline | 924 | 1241 | 84, 83 (32), 56 (19), 57 (18), 41 (10), 42 (10), 82 (8), 39 (7), 29 (7), 126 (tr), 127 (1) | 128.1070 | 128.1072 |
| 14 | 2-isobutyl-4-methyl-3-oxazoline | 1026 | 1375 | 84, 83 (17), 57 (15), 85 (15), 68 (10), 42 (10), 41 (8), 29 (8), 39 (5), 140 (tr), 141 (tr) | 142.1226 | 142.1230 |
| 15 | 2-sec-butyl-4-methyl-3-oxazoline | 1029 | 1352 | 84, 83 (32), 57 (18), 42 (10), 41 (9), 29 (8), 55 (8), 85 (7), 39 (6), 140 (tr), 141 (tr) | 142.1226 | 142.1230 |
| 16 | 2-benzyl-4-methyl-3-oxazoline | 1394 | 2001 | 84, 92 (28), 91 (26), 57 (16), 65 (7), 28 (6), 77 (6), 29 (5), 85 (5), 174 (tr), 175 (tr) | 176.1070 | 176.1073 |
| 17a | 2-isopropyl-5-ethyl-4-methyl-3-oxazoline | 1031, 1044 | 1301, 1318 | 112, 57 (39), 85 (26), 111 (19), 41 (12), 42 (12), 56 (12), 43 (11), 84 (9), 154 (tr), 155 (tr) | 156.1383 | 156.1386, 156.1387 |
| 17b | 2-isopropyl-4-ethyl-5-methyl-3-oxazoline | 1029, 1040 | 1284, 1291 | 112, 43 (53), 56 (39), 111 (18), 85 (14), 41 (14), 70 (12), 100 (10), 96 (9), 154 (tr), 155 (tr) | 156.1383 | 156.1381, 156.1384 |
| 18a | 2-isobutyl-5-ethyl-4-methyl-3-oxazoline | 1133, 1141 | 1400, 1410 | 112, 57 (28), 85 (26), 113 (21), 43 (17), 41 (16), 71 (15), 42 (15), 68 (13), 168 (tr), 169 (tr) | 170.1539 | 170.1541, 170.1543 |
| 18b | 2-isobutyl-4-ethyl-5-methyl-3-oxazoline | 1130, 1135 | 1382, 1384 | 112, 43 (58), 99 (30), 71 (17), 41 (15), 82 (14), 70 (13), 85 (12), 114 (10), 168 (tr), 169 (tr) | 170.1539 | 170.1541, 170.1541, |
| 19a | 2-sec-butyl-5-ethyl-4-methyl-3-oxazoline | 1129, 1131, 1142, 1143 | 1387, 1391, 1405 | 112, 57 (33), 85 (24), 111 (18), 41 (12), 70 (12), 42 (11), 43 (11), 84 (9), 168 (tr), 169 (tr) | 170.1539 | 170.1541, 170.1543, 170.1544, 170.1545 |
| 19b | 2-sec-butyl-4-ethyl-5-methyl-3-oxazoline | 1139 | 1371, 1373, 1378, 1382 | 112, 43 (46), 70 (29), 111 (19), 85 (14), 41 (14), 96 (13), 56 (12), 55 (11), 168 (tr), 169 (tr) | 170.1539 | 170.1543 |
| 20a | 2-benzyl-5-ethyl-4-methyl-3-oxazoline | 1497, 1532 | 2022, 2076 | 112, 57 (43), 91 (33), 85 (28), 111 (23), 43 (11), 65 (8), 77 (7), 103 (7), 202 (tr), 203 (tr) | 204.1383 | 204.1387, 204.1385 |
| 20b | 2-benzyl-4-ethyl-5-methyl-3-oxazoline | 1495, 1523 | 1999, 2034 | 112, 43 (71), 91 (27), 111 (26), 70 (15), 85 (14), 77 (9), 103 (9), 65 (8), 202 (tr), 203 (tr) | 204.1383 | 204.1385, 204.1385 |
Where multiple stereoisomers are reported, just the fragmentation pattern of the largest isomer is provided; the first number is the base peak; the molecular ion M+ is in bold type; tr = trace <0.5%.
The protonated molecular mass, observed by QToF CI mass spectrometry, matched the theoretical values (±10 ppm) predicted from the chemical formula. Depending on the number of chiral centers in the compound, more than one stereoisomer was observed, supported by very similar, if not identical, mass spectra, LRI values, and protonated exact mass. For 17–20, structural isomers were also observed, supported by pairs of mass spectra differing in the intensity of m/z43 and 57.
Figure 3
Figure 3. HMBC NMR spectrum (A) that confirms the annotated structure (B) of 2-isobutyl-4,5-dimethyl-3-oxazoline. The signals along the F1 and F2 axes (13C and 1H spectra, respectively) are assigned according to the atom numbers displayed in (B). The most important cross-peaks, showing the arrangement of atoms key to 3-oxazolines, have been highlighted in colored boxes, with the signals within pink boxes correlating to the pink arrows in (B), and likewise for blue, green, and orange. Each cross-peak indicates that the horizontally and vertically aligned carbon and hydrogen atoms (annotated in red, connected by an arrow) are separated by either two or three bonds within the molecule (H–C–C or H–C–C–C, respectively), confirming the structure in (B). To minimize cluttering of the figure, not all of the cross-peaks have been annotated. The dashed lines circling cross-peak pairs, aligned with a carbon signal but evenly spaced on either side of a hydrogen alignment, indicate that the aligned carbon and hydrogen atoms are directly connected, separated by just one bond (H–C). These signals are artifacts of an HMBC spectrum, therefore are not observed for all atoms; however, the relevant signals can be observed in the HSQC spectrum (Figure S2.9).
| compound | assessor 1 | assessor 2 | assessor 3 | assessor 4 | assessor 5 |
|---|---|---|---|---|---|
| 1aa | grassy, musty | cardboard | dark chocolate, cocoa powder | odd, vegetable, cardboard | green, drain-ish |
| 1b | strange, vegetable | cardboard | floral, chocolate | herbal, medicinal | green, drain-ish |
| 2 | musty, cocoa | xb | chocolate | green, aldehyde-ish | green, minty |
| 3a | grassy, musty | powder, makeup | green, waxy, earthy | cardboardy, musty | soil, pyrazine-like |
| 3b | green, strange | floral | pink sweets, linalool-like | herbal | x |
| 4a | x | x | x | x | x |
| 4b | x | x | x | x | x |
a and b refer to two isomers of each compound, which were separable by GC-FID; however for compound 2, the isomers could not be completely separated, and most assessors could only detect one odor.
x indicates where an odor could not be detected.
Identification of 4,5-Dimethyl-3-oxazolines (1–4) in Cocoa Products
Figure 4
Figure 4. GC–MS data supporting the identification of synthesized 3-oxazolines in cocoa. Mass spectrum provided is from the larger of the two peaks, both of which have similar fragmentation patterns.
Figure 5
Figure 5. 2D-GC–MS analysis of roasted cacao nibs, extracted by SAFE. The synthesized 4,5-dimethyl-3-oxazoline standards 1–4 were each run separately and used to create a template (indicated by the green, blue, red, and orange circles, respectively) to show the expected retention times of 4,5-dimethyl-3-oxazolines. This image was created by extracting ion 98 from the chromatogram of the SAFE extract and overlaying with the template. Compounds in cocoa, shown here as blobs, were found close to or within the template regions, and possessed the same mass spectrum as the synthesized standards, indicating the presence of these 4,5-dimethyl-3-oxazolines in cocoa for the first time.

Synthesis of Other 3-Oxazolines
Figure 6
Figure 6. Methylglyoxal is an asymmetrical α-dicarbonyl compound and was proposed to react with amino acids, such as valine, in two conformations to generate 3-oxazoline regioisomers, 2-isopropyl-5-methyl-3-oxazoline and 2-isopropyl-4-methyl-3-oxazoline, in the case of valine (9 and 13, respectively). In the same way, 2,3-pentanedione can also react to form the regioisomers, 5-ethyl-4-methyl- and 4-ethyl-5-methyl-3-oxazolines.
Tentative Identification of Other 3-Oxazolines in Cocoa
Figure 7
Figure 7. (A) Mass spectrum of the peak identified as 2-isobutyl-5-ethyl-4-methyl-3-oxazoline in cacao nibs. (B) Mass spectrum of synthesized 2-isobutyl-5-ethyl-4-methyl-3-oxazoline.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.5c00898.
Figure S1: the Strecker degradation of amino acids, initiated by α-dicarbonyl compounds, to form the aroma compounds, Strecker aldehydes; Figure S2: NMR spectra (1H, 13C, 1H–1H COSY, 1H–13C HSQC, and 1H–13C HMBC) of synthesized 3-oxazolines 1–4; Figure S3: GC–MS analysis of cacao nib SAFE extract, spiked with 2-isobutyl- and 2-sec-butyl-4,5-dimethyl-3-oxazoline (2 and 3); Figure S4: GC–MS identification of 4,5-dimethyl-3-oxazoline in different cocoa products; Figure S5: EI mass spectra of additionally synthesized 3-oxazolines 5–20;Figure S6: NMR spectra (1H, 13C, 1H–1H COSY,1 H–13C HSQC, and 1H–13C HMBC) of 2-isopropyl-5-ethyl-4-methyl-3-oxazoline (17a); Figure S7: 2D-GC–MS analysis of roasted cacao nibs, extracted by SAFE (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
HGS acknowledges the Biotechnology and Biological Sciences Research Council and Mondele̅z International for funding from the Food Consortium Collaborative Training Partnership (CTP). The authors gratefully acknowledge the Technical Services staff within the Chemical Analysis Facility at the University of Reading for their technical support and assistance in performing NMR experiments for this work.
| 1D-GC–MS | 1-dimensional gas chromatography coupled to mass spectrometry |
| 2D-GC–MS | 2-dimensional gas chromatography coupled to mass spectrometry |
| AR | analytical reagent |
| COSY | correlation spectroscopy |
| GC-FID | gas chromatography coupled to flame ionization detection |
| GC–O | gas chromatography coupled to olfactometry |
| GC-QToF | gas chromatography coupled to a quadrupole time-of-flight detector |
| HMBC | heteronuclear multiple-bond correlation |
| HSQC | heteronuclear single quantum coherence |
| MCT | medium-chain triglycerides |
| NIST | National Institute of Standards and Technology |
| SAFE | solvent-assisted flavor evaporation |
References
This article references 22 other publications.
- 1Granvogl, M.; Beksan, E.; Schieberle, P. New insights into the formation of aroma-active Strecker aldehydes from 3-oxazolines as transient intermediates. J. Agric. Food Chem. 2012, 60 (25), 6312– 6322, DOI: 10.1021/jf301489jGoogle ScholarThere is no corresponding record for this reference.
- 2Buhr, K.; Pammer, C.; Schieberle, P. Influence of water on the generation of Strecker aldehydes from dry processed foods. Eur. Food Res. Technol. 2010, 230 (3), 375– 381, DOI: 10.1007/s00217-009-1169-yGoogle ScholarThere is no corresponding record for this reference.
- 3Kim, S.-Y.; Ko, J.-A.; Kang, B.-S.; Park, H.-J. Prediction of key aroma development in coffees roasted to different degrees by colorimetric sensor array. Food Chem. 2018, 240, 808– 816, DOI: 10.1016/j.foodchem.2017.07.139Google ScholarThere is no corresponding record for this reference.
- 4Hofmann, T.; Münch, P.; Schieberle, P. Quantitative model studies on the formation of Aroma-Active aldehydes and acids by Strecker-type reactions. J. Agric. Food Chem. 2000, 48 (2), 434– 440, DOI: 10.1021/jf990954cGoogle ScholarThere is no corresponding record for this reference.
- 5Parker, J. K.; Elmore, J. S.; Methven, L. Flavour Development Analysis and Perception in Food and Beverages; Woodhead Publishing, 2015.Google ScholarThere is no corresponding record for this reference.
- 6Cha, J.; Debnath, T.; Lee, K.-G. Analysis of α-dicarbonyl compounds and volatiles formed in Maillard reaction model systems. Sci. Rep. 2019, 9, 5325, DOI: 10.1038/s41598-019-41824-8Google ScholarThere is no corresponding record for this reference.
- 7Rizzi, G. P. The Strecker degradation of amino acids: Newer avenues for flavor formation. Food Rev. Int. 2008, 24 (4), 416– 435, DOI: 10.1080/87559120802306058Google ScholarThere is no corresponding record for this reference.
- 8Hofmann, T.; Schieberle, P. Formation of aroma-active Strecker-aldehydes by a direct oxidative degradation of Amadori compounds. J. Agric. Food Chem. 2000, 48 (9), 4301– 4305, DOI: 10.1021/jf000076eGoogle ScholarThere is no corresponding record for this reference.
- 9Cremer, D. R.; Vollenbroeker, M.; Eichner, K. Investigation of the formation of Strecker aldehydes from the reaction of Amadori rearrangement products with α-amino acids in low moisture model systems. Eur. Food Res. Technol. 2000, 211 (6), 400– 403, DOI: 10.1007/s002170000196Google ScholarThere is no corresponding record for this reference.
- 10Hartmann, S.; Schieberle, P. On the role of Amadori rearrangement products as precursors of aroma-active Strecker aldehydes in cocoa. In Browned Flavors: Analysis, Formation, and Physiology; ACS Symposium Series; American Chemical Society, 2016, Vol. 1237, pp. 1– 13. DOI: 10.1021/bk-2016-1237.ch001 .Google ScholarThere is no corresponding record for this reference.
- 11Granvogl, M.; Bugan, S.; Schieberle, P. Formation of amines and aldehydes from parent amino acids during thermal processing of cocoa and model systems: New insights into pathways of the Strecker reaction. J. Agric. Food Chem. 2006, 54 (5), 1730– 1739, DOI: 10.1021/jf0525939Google ScholarThere is no corresponding record for this reference.
- 12Schuh, C.; Schieberle, P. Characterization of the key aroma compounds in the beverage prepared from Darjeeling black tea: Quantitative differences between tea leaves and infusion. J. Agric. Food Chem. 2006, 54 (3), 916– 924, DOI: 10.1021/jf052495nGoogle ScholarThere is no corresponding record for this reference.
- 13Rögner, N. S.; Mall, V.; Steinhaus, M. Odour-active compounds in liquid malt extracts for the baking industry. Eur. Food Res. Technol. 2021, 247 (5), 1263– 1275, DOI: 10.1007/s00217-021-03707-zGoogle ScholarThere is no corresponding record for this reference.
- 14Schieberle, P.; Pfnuer, P. Characterization of key odorants in chocolate. In Flavor Chemistry: Thirty Years of Progress, Teranishi, R.; Wick, E. L.; Hornstein, J., Eds.; Kluwer Academic/Plenum Publishers: New York, 1999; pp. 147– 153.Google ScholarThere is no corresponding record for this reference.
- 15Ullrich, L.; Neiens, S.; Hühn, T.; Steinhaus, M.; Chetschik, I. Impact of water on odor-active compounds in fermented and dried cocoa beans and chocolates made thereof. J. Agric. Food Chem. 2021, 69 (30), 8504– 8510, DOI: 10.1021/acs.jafc.1c02287Google ScholarThere is no corresponding record for this reference.
- 16Rizzi, G. P. Formation of tetramethylpyrazine and 2-isopropyl-4,5-dimethyl-3-oxazoline in the Strecker degradation of DL-valine with 2,3-butanedione. J. Org. Chem. 1969, 34 (6), 2002– 2004, DOI: 10.1021/jo01258a117Google ScholarThere is no corresponding record for this reference.
- 17Elmore, J. S.; Mottram, D. S. Investigation of the reaction between ammonium sulfide, aldehydes, and α-hydroxyketones or α-dicarbonyls to form some lipid–Maillard interaction products found in cooked beef. J. Agric. Food Chem. 1997, 45 (9), 3595– 3602, DOI: 10.1021/jf970065uGoogle ScholarThere is no corresponding record for this reference.
- 18Engel, W.; Bahr, W.; Schieberle, P. Solvent assisted flavour evaporation – a new and versatile technique for the careful and direct isolation of aroma compounds from complex food matrices. Eur. Food Res. Technol. 1999, 209, 237– 241, DOI: 10.1007/s002170050486Google ScholarThere is no corresponding record for this reference.
- 19National Institute of Standards and Technology, N. M. S. D. C. C. (Department of C. W. D. C.). 2-iso-propyl-4-ethyl-5-methyloxazole: NIST Standard Reference Database 69: NIST Chemistry WebBook, NIST, 2014. https://webbook.nist.gov/cgi/cbook.cgi?ID=C84027963&Mask=200#Mass-Spec.Google ScholarThere is no corresponding record for this reference.
- 20Maasen, K.; Scheijen, J. L. J. M.; Opperhuizen, A.; Stehouwer, C. D. A.; Van Greevenbroek, M. M.; Schalkwijk, C. G. Quantification of dicarbonyl compounds in commonly consumed foods and drinks; presentation of a food composition database for dicarbonyls. Food Chem. 2021, 339, 128063, DOI: 10.1016/j.foodchem.2020.128063Google ScholarThere is no corresponding record for this reference.
- 21Çintesun, E. E.; Tanyıldız, S. N.; Yıldırım, H.; Mızrak, Ö. F.; Yaman, M. Investigation of the α-dicarbonyl compounds in some snack foods by HPLC using precolumn derivatization with 4-nitro-1,2-phenylenediamine. Biointerface Res. Appl. Chem. 2022, 12 (2), 2242– 2250, DOI: 10.33263/BRIAC122.22422250Google ScholarThere is no corresponding record for this reference.
- 22Deuscher, Z.; Gourrat, K.; Repoux, M.; Boulanger, R.; Labouré, H.; Le Quéré, J.-L. Key aroma compounds of dark chocolates differing in organoleptic properties: A GC-O comparative study. Molecules 2020, 25 (8), 1809, DOI: 10.3390/molecules25081809Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 2 publications.
- Thien H. Nguyen, Ethan W. Darby, John P. Munafo, Jr.. Characterization of Important Odorants in Rehydrated Lobster Mushrooms. Journal of Agricultural and Food Chemistry 2025, 73
(39)
, 24898-24906. https://doi.org/10.1021/acs.jafc.5c07619
- Diner Mori-Mestanza, Jheniffer E. Valdivia-Culqui, César R. Balcázar-Zumaeta, Ilse S. Cayo-Colca, Eduardo Cassel, Efraín M. Castro-Alayo, Fiorella P. Cárdenas-Toro. Dynamic profile of metabolites and volatile compounds in post-harvest cocoa beans until chocolate production. European Food Research and Technology 2026, 252
(3)
https://doi.org/10.1007/s00217-026-05088-7
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
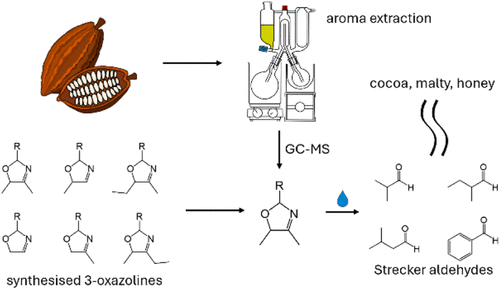
Figure 1
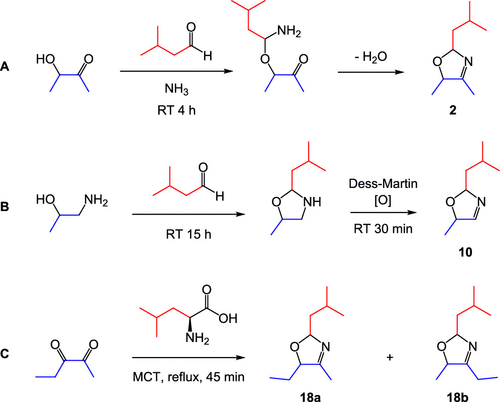
Figure 1. Examples of the three synthetic routes used to generate 3-oxazolines. (A) The Elmore method, used for the preparation of 1–4, adapted from 3-thiazoline synthesis, reported by Elmore and Mottram. (17) (B) The Granvogl method, used for the preparation of 5–16, adapted from the synthesis for 2-substituted-5-methyl-3-oxazolines, reported by Granvogl et al. (1) The amino alcohol reagent was varied in order to control the substituents at C4 and C5. (C) The Rizzi method, used for the preparation of 17–20, adapted from the synthesis for 2-isopropyl-4,5-dimethyl-3-oxazoline, reported by Rizzi. (16)
Figure 2

Figure 2. Stereoisomers of 2-isobutyl-4,5-dimethyl-3-oxazoline (2). (A) Two chiral centers, indicated by asterisks, gives rise to four stereoisomers, existing as two pairs of enantiomers that are diastereomers of each other, assigned as (S) or (R) by the Cahn–Ingold–Prelog rules. (B) All four stereoisomers were observed on a chiral GC–MS column (CP-Chirasil-Dex CB).
Figure 3
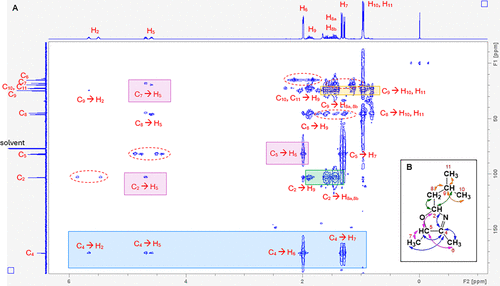
Figure 3. HMBC NMR spectrum (A) that confirms the annotated structure (B) of 2-isobutyl-4,5-dimethyl-3-oxazoline. The signals along the F1 and F2 axes (13C and 1H spectra, respectively) are assigned according to the atom numbers displayed in (B). The most important cross-peaks, showing the arrangement of atoms key to 3-oxazolines, have been highlighted in colored boxes, with the signals within pink boxes correlating to the pink arrows in (B), and likewise for blue, green, and orange. Each cross-peak indicates that the horizontally and vertically aligned carbon and hydrogen atoms (annotated in red, connected by an arrow) are separated by either two or three bonds within the molecule (H–C–C or H–C–C–C, respectively), confirming the structure in (B). To minimize cluttering of the figure, not all of the cross-peaks have been annotated. The dashed lines circling cross-peak pairs, aligned with a carbon signal but evenly spaced on either side of a hydrogen alignment, indicate that the aligned carbon and hydrogen atoms are directly connected, separated by just one bond (H–C). These signals are artifacts of an HMBC spectrum, therefore are not observed for all atoms; however, the relevant signals can be observed in the HSQC spectrum (Figure S2.9).
Figure 4

Figure 4. GC–MS data supporting the identification of synthesized 3-oxazolines in cocoa. Mass spectrum provided is from the larger of the two peaks, both of which have similar fragmentation patterns.
Figure 5

Figure 5. 2D-GC–MS analysis of roasted cacao nibs, extracted by SAFE. The synthesized 4,5-dimethyl-3-oxazoline standards 1–4 were each run separately and used to create a template (indicated by the green, blue, red, and orange circles, respectively) to show the expected retention times of 4,5-dimethyl-3-oxazolines. This image was created by extracting ion 98 from the chromatogram of the SAFE extract and overlaying with the template. Compounds in cocoa, shown here as blobs, were found close to or within the template regions, and possessed the same mass spectrum as the synthesized standards, indicating the presence of these 4,5-dimethyl-3-oxazolines in cocoa for the first time.
Figure 6
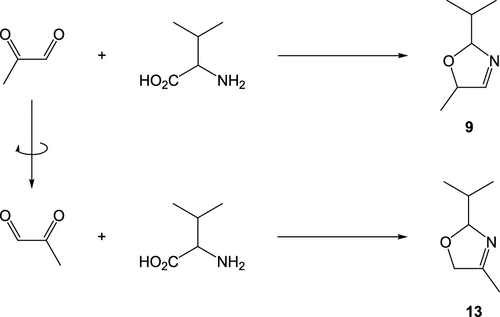
Figure 6. Methylglyoxal is an asymmetrical α-dicarbonyl compound and was proposed to react with amino acids, such as valine, in two conformations to generate 3-oxazoline regioisomers, 2-isopropyl-5-methyl-3-oxazoline and 2-isopropyl-4-methyl-3-oxazoline, in the case of valine (9 and 13, respectively). In the same way, 2,3-pentanedione can also react to form the regioisomers, 5-ethyl-4-methyl- and 4-ethyl-5-methyl-3-oxazolines.
Figure 7
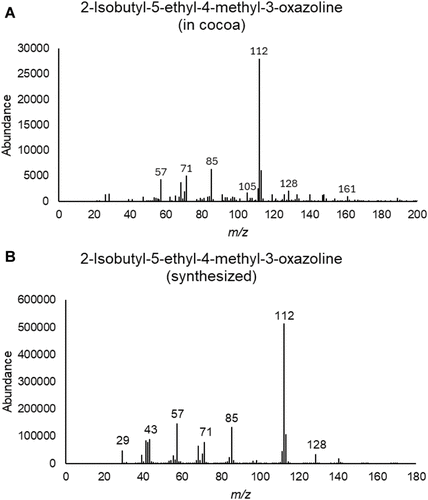
Figure 7. (A) Mass spectrum of the peak identified as 2-isobutyl-5-ethyl-4-methyl-3-oxazoline in cacao nibs. (B) Mass spectrum of synthesized 2-isobutyl-5-ethyl-4-methyl-3-oxazoline.
References
This article references 22 other publications.
- 1Granvogl, M.; Beksan, E.; Schieberle, P. New insights into the formation of aroma-active Strecker aldehydes from 3-oxazolines as transient intermediates. J. Agric. Food Chem. 2012, 60 (25), 6312– 6322, DOI: 10.1021/jf301489jThere is no corresponding record for this reference.
- 2Buhr, K.; Pammer, C.; Schieberle, P. Influence of water on the generation of Strecker aldehydes from dry processed foods. Eur. Food Res. Technol. 2010, 230 (3), 375– 381, DOI: 10.1007/s00217-009-1169-yThere is no corresponding record for this reference.
- 3Kim, S.-Y.; Ko, J.-A.; Kang, B.-S.; Park, H.-J. Prediction of key aroma development in coffees roasted to different degrees by colorimetric sensor array. Food Chem. 2018, 240, 808– 816, DOI: 10.1016/j.foodchem.2017.07.139There is no corresponding record for this reference.
- 4Hofmann, T.; Münch, P.; Schieberle, P. Quantitative model studies on the formation of Aroma-Active aldehydes and acids by Strecker-type reactions. J. Agric. Food Chem. 2000, 48 (2), 434– 440, DOI: 10.1021/jf990954cThere is no corresponding record for this reference.
- 5Parker, J. K.; Elmore, J. S.; Methven, L. Flavour Development Analysis and Perception in Food and Beverages; Woodhead Publishing, 2015.There is no corresponding record for this reference.
- 6Cha, J.; Debnath, T.; Lee, K.-G. Analysis of α-dicarbonyl compounds and volatiles formed in Maillard reaction model systems. Sci. Rep. 2019, 9, 5325, DOI: 10.1038/s41598-019-41824-8There is no corresponding record for this reference.
- 7Rizzi, G. P. The Strecker degradation of amino acids: Newer avenues for flavor formation. Food Rev. Int. 2008, 24 (4), 416– 435, DOI: 10.1080/87559120802306058There is no corresponding record for this reference.
- 8Hofmann, T.; Schieberle, P. Formation of aroma-active Strecker-aldehydes by a direct oxidative degradation of Amadori compounds. J. Agric. Food Chem. 2000, 48 (9), 4301– 4305, DOI: 10.1021/jf000076eThere is no corresponding record for this reference.
- 9Cremer, D. R.; Vollenbroeker, M.; Eichner, K. Investigation of the formation of Strecker aldehydes from the reaction of Amadori rearrangement products with α-amino acids in low moisture model systems. Eur. Food Res. Technol. 2000, 211 (6), 400– 403, DOI: 10.1007/s002170000196There is no corresponding record for this reference.
- 10Hartmann, S.; Schieberle, P. On the role of Amadori rearrangement products as precursors of aroma-active Strecker aldehydes in cocoa. In Browned Flavors: Analysis, Formation, and Physiology; ACS Symposium Series; American Chemical Society, 2016, Vol. 1237, pp. 1– 13. DOI: 10.1021/bk-2016-1237.ch001 .There is no corresponding record for this reference.
- 11Granvogl, M.; Bugan, S.; Schieberle, P. Formation of amines and aldehydes from parent amino acids during thermal processing of cocoa and model systems: New insights into pathways of the Strecker reaction. J. Agric. Food Chem. 2006, 54 (5), 1730– 1739, DOI: 10.1021/jf0525939There is no corresponding record for this reference.
- 12Schuh, C.; Schieberle, P. Characterization of the key aroma compounds in the beverage prepared from Darjeeling black tea: Quantitative differences between tea leaves and infusion. J. Agric. Food Chem. 2006, 54 (3), 916– 924, DOI: 10.1021/jf052495nThere is no corresponding record for this reference.
- 13Rögner, N. S.; Mall, V.; Steinhaus, M. Odour-active compounds in liquid malt extracts for the baking industry. Eur. Food Res. Technol. 2021, 247 (5), 1263– 1275, DOI: 10.1007/s00217-021-03707-zThere is no corresponding record for this reference.
- 14Schieberle, P.; Pfnuer, P. Characterization of key odorants in chocolate. In Flavor Chemistry: Thirty Years of Progress, Teranishi, R.; Wick, E. L.; Hornstein, J., Eds.; Kluwer Academic/Plenum Publishers: New York, 1999; pp. 147– 153.There is no corresponding record for this reference.
- 15Ullrich, L.; Neiens, S.; Hühn, T.; Steinhaus, M.; Chetschik, I. Impact of water on odor-active compounds in fermented and dried cocoa beans and chocolates made thereof. J. Agric. Food Chem. 2021, 69 (30), 8504– 8510, DOI: 10.1021/acs.jafc.1c02287There is no corresponding record for this reference.
- 16Rizzi, G. P. Formation of tetramethylpyrazine and 2-isopropyl-4,5-dimethyl-3-oxazoline in the Strecker degradation of DL-valine with 2,3-butanedione. J. Org. Chem. 1969, 34 (6), 2002– 2004, DOI: 10.1021/jo01258a117There is no corresponding record for this reference.
- 17Elmore, J. S.; Mottram, D. S. Investigation of the reaction between ammonium sulfide, aldehydes, and α-hydroxyketones or α-dicarbonyls to form some lipid–Maillard interaction products found in cooked beef. J. Agric. Food Chem. 1997, 45 (9), 3595– 3602, DOI: 10.1021/jf970065uThere is no corresponding record for this reference.
- 18Engel, W.; Bahr, W.; Schieberle, P. Solvent assisted flavour evaporation – a new and versatile technique for the careful and direct isolation of aroma compounds from complex food matrices. Eur. Food Res. Technol. 1999, 209, 237– 241, DOI: 10.1007/s002170050486There is no corresponding record for this reference.
- 19National Institute of Standards and Technology, N. M. S. D. C. C. (Department of C. W. D. C.). 2-iso-propyl-4-ethyl-5-methyloxazole: NIST Standard Reference Database 69: NIST Chemistry WebBook, NIST, 2014. https://webbook.nist.gov/cgi/cbook.cgi?ID=C84027963&Mask=200#Mass-Spec.There is no corresponding record for this reference.
- 20Maasen, K.; Scheijen, J. L. J. M.; Opperhuizen, A.; Stehouwer, C. D. A.; Van Greevenbroek, M. M.; Schalkwijk, C. G. Quantification of dicarbonyl compounds in commonly consumed foods and drinks; presentation of a food composition database for dicarbonyls. Food Chem. 2021, 339, 128063, DOI: 10.1016/j.foodchem.2020.128063There is no corresponding record for this reference.
- 21Çintesun, E. E.; Tanyıldız, S. N.; Yıldırım, H.; Mızrak, Ö. F.; Yaman, M. Investigation of the α-dicarbonyl compounds in some snack foods by HPLC using precolumn derivatization with 4-nitro-1,2-phenylenediamine. Biointerface Res. Appl. Chem. 2022, 12 (2), 2242– 2250, DOI: 10.33263/BRIAC122.22422250There is no corresponding record for this reference.
- 22Deuscher, Z.; Gourrat, K.; Repoux, M.; Boulanger, R.; Labouré, H.; Le Quéré, J.-L. Key aroma compounds of dark chocolates differing in organoleptic properties: A GC-O comparative study. Molecules 2020, 25 (8), 1809, DOI: 10.3390/molecules25081809There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.5c00898.
Figure S1: the Strecker degradation of amino acids, initiated by α-dicarbonyl compounds, to form the aroma compounds, Strecker aldehydes; Figure S2: NMR spectra (1H, 13C, 1H–1H COSY, 1H–13C HSQC, and 1H–13C HMBC) of synthesized 3-oxazolines 1–4; Figure S3: GC–MS analysis of cacao nib SAFE extract, spiked with 2-isobutyl- and 2-sec-butyl-4,5-dimethyl-3-oxazoline (2 and 3); Figure S4: GC–MS identification of 4,5-dimethyl-3-oxazoline in different cocoa products; Figure S5: EI mass spectra of additionally synthesized 3-oxazolines 5–20;Figure S6: NMR spectra (1H, 13C, 1H–1H COSY,1 H–13C HSQC, and 1H–13C HMBC) of 2-isopropyl-5-ethyl-4-methyl-3-oxazoline (17a); Figure S7: 2D-GC–MS analysis of roasted cacao nibs, extracted by SAFE (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


