
Phenolic Compounds and Derivatives in Ruminant Meat and Milk: A Systematic ReviewClick to copy article linkArticle link copied!
- Muhammad Ahsin*Muhammad Ahsin*Email: [email protected]Department of Nutrition, Dietetics and Food Sciences, College of Agriculture and Applied Sciences, Utah State University, Logan, Utah 84322, United StatesMore by Muhammad Ahsin
- Sulaiman K. MatarnehSulaiman K. MatarnehDepartment of Nutrition, Dietetics and Food Sciences, College of Agriculture and Applied Sciences, Utah State University, Logan, Utah 84322, United StatesMore by Sulaiman K. Matarneh
- Kara J. ThorntonKara J. ThorntonDepartment of Animal, Dairy, and Veterinary Sciences, Utah State University, Logan, Utah 84322, United StatesMore by Kara J. Thornton
- Scott KronbergScott KronbergUSDA-Agricultural Research Service, Mandan, North Dakota 58554, United StatesMore by Scott Kronberg
- Mamoona AmirMamoona AmirDepartment of Animal Food Products Technology, Faculty of Food Science and Nutrition, Bahauddin Zakariya University, Multan, Punjab PK 60800, PakistanMore by Mamoona Amir
- Stephan van Vliet*Stephan van Vliet*Email: [email protected]Department of Nutrition, Dietetics and Food Sciences, College of Agriculture and Applied Sciences, Utah State University, Logan, Utah 84322, United StatesMore by Stephan van Vliet
Abstract
This review synthesizes evidence on phenolic concentrations and diversity in ruminant meat and milk, considering animal species, management, forage, seasonality, and analytical methods. From 39 studies, 356 distinct phenolics were identified in meat and milk, including several from medicinal and non-staple-forage plants. Goat milk showed the highest concentrations as measured by total phenolic content assays (1390 μg GAE/mL) and targeted mass spectrometry (26.79 μg/mL). Beef had the greatest diversity (164 metabolites), followed by sheep milk (110 metabolites); however, beef is also most studied. Organic/agroecological versus conventional systems, fresh versus preserved forages, and younger versus mature pastures were generally associated with a higher phenolic content. Among forages, red clover supported greater diversity than chicory, lucerne, or white clover, while maize silage yielded a higher phenolic content than ryegrass silage. Ruminants can act as biological mediators linking soils, plants, and human diets, often resulting in upcycling of phenolic-derived metabolites from plants not consumed by humans. Future research should integrate soil, plant, animal, and food sciences to fully reveal this role and its potential significance to human health.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Study Inclusion
3.2. Risk of Bias and Quality of Reporting Assessment
3.3. Extraction Solvents for Meat and Milk Phenolics
3.4. Phenolics Quantitation Method for the Ruminant Meat and Milk
3.5. Metabolic Fate of Plant Phenolics in Ruminants
Figure 1
Figure 1. Model of in-depth pathway analysis of phenolic compounds as they flow from plants to ruminant digestion to tissue (meat) and milk. The biochemical processes these compounds undergo in the rumen and small intestine, routes of absorption through enterocyte barriers, metabolism in the liver and kidney, and transfer pathways into muscle and milk. SGLT1 = sodium-glucose transport, CBG = corticosteroid-binding globulin, and LPH = lactase-phlorizin hydrolase, UGTs = UDP-glucuronosyltransferases, SULTs = sulfotransferases, and COMT = catechol O-methyltransferase. The figure was created with biorender.com.
3.6. Phenolic Concentrations in Ruminant Milk
3.6.1. Cow Milk
Figure 2
Figure 2. Total phenolic concentration (TPC) in milk and/or meat from the included studies, organized by experimental groups as reported in the original studies. Values are group means; n denotes the number of biological replicates per group. ‘ref:’ shows the reference of the study, and country names are given in International Organization for Standardization (ISO) 3166-1 alpha-2 codes.
Figure 3
Figure 3. Concentrations of measured phenolics in milk and/or meat from the included studies, organized by experimental groups as reported in the original studies. Values are group means; ‘P’ indicates the number of phenolics quantified, ‘ref:’ shows the reference of the study; ‘n’ indicates the number of biological replicates per group. The study country is denoted by International Organization for Standardization (ISO) 3166–1 alpha-2 codes.
3.6.2. Goat Milk
3.6.3. Sheep Milk
3.7. Phenolics Concentration in Ruminant Meat
3.8. Phenolic Diversity in Ruminant Milk and Meat
Figure 4
Figure 4. Phenolic diversity across animal-source foods identified by the current systematic review (n = 39 studies). Circle size is the number of phenolics reported within each class; circle color indicates the food product. Because flavonoids, phenolic acids, and other polyphenols vary widely, these classes are presented separately.
| latin name/species | common name | product | no. of citations | total phenolics | unique phenolics |
|---|---|---|---|---|---|
| Bos taurus | cattle | milk | 17 | 98 | 54 |
| meat | 4 | 164 | 132 | ||
| Capra aegagrus hircus | goat | milk | 5 | 70 | 29 |
| meat | 1 | 17 | 17 | ||
| Ovis aries | sheep | milk | 6 | 160 | 110 |
| meat | 1 | 5 | 0 | ||
| Bubalus bubalis | buffalo | milk | 1 | 28 | 0 |
| Bison | bison | meat | 1 | 12 | 0 |
Values are counts of phenolics summarized across 39 studies, reported as Total (all detections) and Unique (distinct compounds after deduplication with any other product in this review).
3.9. Medicinal-Plant Phenolics in the Ruminant Milk and Meat

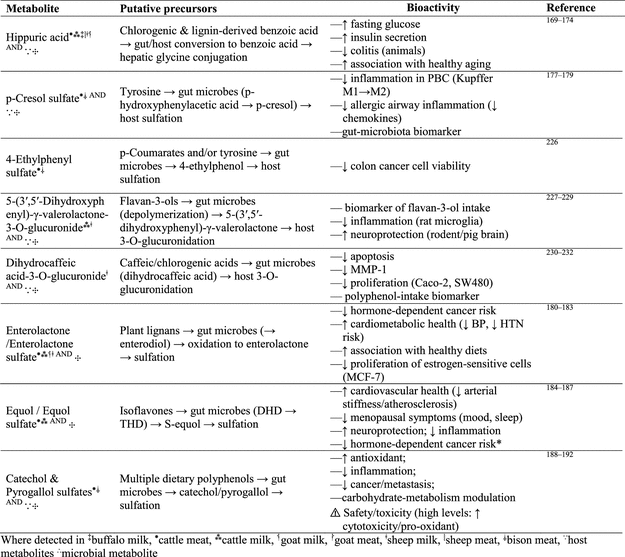
3.10. Bioactivity of Phenolic-Derived Mammalian Metabolites in Their Meat and Milk
3.11. Challenges and Limitations in Current Research
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.5c06118.
Figure S1. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram summarizing the literature search and study selection process followed to selected eligible studied to be this systematically review. Figure S2. Risk of bias and quality of reporting assessment based on SYRCLE’s risk of bias tool 1: (A) indicator of quality of reporting included study design and objectives, population and sample characteristics, intervention and comparison groups, analytical methods, data analysis and presentation, ethical considerations, funding and conflicts of interest (B) risk-of-bias analysis in selection, measurement, confounding, and selective reporting. Table S1 List of phenolic compounds identified in selected ruminants meat and milk samples from studies included in the systematic review, organized by matrix (species/product). Table S2. Study-level data extraction for 39 studies, including sample type; country of origin; experimental group (e.g., breed, fresh vs conserved forage, short- vs long-term pasture, month of year, commercial samples, milk-fat content); measured phenolics (relevant compounds extracted from metabolomics data sets); concentrations (where reported, with n per group); analytical platform; and references (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
S.V.V. acknowledges current grant support from USDA-NIFA-SARE (2020-38640-31521; 2021-38640-34714), USDA-ARS (USDA-2022-58-3064-2-007), the Greenacres Foundation, the Turner Institute of Ecoagriculture, Perdue Foods LLC, and the Bionutrient Institute for (cofunded) projects that link agricultural production systems to the nutritional/metabolite composition of animal and plant foods. S.V.V. also reports travel honoraria and speaker fees related to research presentations. S.V.V. is a nonpaid member of the Scientific Advisory Committee of the Food and Agriculture Organization of the United Nations. We would like to extend our gratitude to Drs. Korry Hintze and Taylor Oberg at Utah State University for their invaluable support and initial guidance during the writing process of this systematic review. Table of Contents (TOC) and Figure 1 were created with biorender.com.
| 3-PPA | 3-phenylpropionic acid |
| COMT | catechol-O-methyltransferase |
| CTs | condensed tannins |
| DOAJ | Directory of Open Access Journals |
| ER-β | estrogen receptor beta |
| FTIR | Fourier-transform infrared spectroscopy |
| GAE | gallic acid equivalents |
| GC | gas chromatography |
| GC-MS/MS | gas chromatography–tandem mass spectrometry |
| HPLC | high-performance liquid chromatography |
| HRAM | high-resolution accurate mass |
| HIV | human immunodeficiency virus (appears in “anti-HIV”) |
| HP-2 | (enzyme prep type) Helix pomatia β-glucuronidase/sulfatase “HP-2 |
| LC | liquid chromatography |
| LC-MS/MS | liquid chromatography–tandem mass spectrometry |
| m/z | mass-to-charge ratio |
| MS | mass spectrometry |
| MS/MS | tandem mass spectrometry |
| PDA | photodiode array (detector) |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| QqQ | triple quadrupole (mass spectrometer) |
| QTOF | quadrupole time-of-flight |
| QTOF-MS | quadrupole time-of-flight mass spectrometry |
| SGLT1 | sodium-dependent glucose transporter 1 |
| SULT(s) | sulfotransferase(s) |
| SYRCLE | Systematic Review Centre for Laboratory animal Experimentation |
| TMR | total mixed ration |
| TPC | total phenolic content |
| UGT(s) | UDP-glucuronosyltransferase(s) |
| UHT | ultrahigh-temperature (processing) |
| UK | United Kingdom |
| US/USA | United States/United States of America |
| UV–Vis | ultraviolet–visible (spectrophotometry) |
| v/v | volume/volume (solution ratio) |
References
This article references 232 other publications.
- 1Stampa, E.; Schipmann-Schwarze, C.; Hamm, U. Consumer Perceptions, Preferences, and Behavior Regarding Pasture-Raised Livestock Products: A Review. Food Qual. Prefer. 2020, 82, 103872 DOI: 10.1016/j.foodqual.2020.103872Google ScholarThere is no corresponding record for this reference.
- 2van Vliet, S.; Provenza, F. D.; Kronberg, S. L. Health-Promoting Phytonutrients Are Higher in Grass-Fed Meat and Milk. Front. Sustain. Food Syst. 2021, 4, 555426 DOI: 10.3389/fsufs.2020.555426Google ScholarThere is no corresponding record for this reference.
- 3Villalba, J. J.; Ramsey, R. D.; Athanasiadou, S. Review: Herbivory and the Power of Phytochemical Diversity on Animal Health. animal 2024, 101287 DOI: 10.1016/j.animal.2024.101287Google ScholarThere is no corresponding record for this reference.
- 4Evans, N.; Cloward, J.; Ward, R. E.; Van Wietmarschen, H. A.; Van Eekeren, N.; Kronberg, S. L.; Provenza, F. D.; Van Vliet, S. Pasture-Finishing of Cattle in Western U.S. Rangelands Improves Markers of Animal Metabolic Health and Nutritional Compounds in Beef. Sci. Rep. 2024, 14 (1), 20240, DOI: 10.1038/s41598-024-71073-3Google ScholarThere is no corresponding record for this reference.
- 5Krusinski, L.; Maciel, I. C. F.; van Vliet, S.; Ahsin, M.; Lu, G.; Rowntree, J. E.; Fenton, J. I. Measuring the Phytochemical Richness of Meat: Effects of Grass/Grain Finishing Systems and grapeseed Extract Supplementation on the Fatty Acid and Phytochemical Content of Beef. Foods 2023, 12 (19), 3547, DOI: 10.3390/foods12193547Google ScholarThere is no corresponding record for this reference.
- 6van Vliet, S.; Blair, A. D.; Hite, L. M.; Cloward, J.; Ward, R. E.; Kruse, C.; van Wietmarchsen, H. A.; van Eekeren, N.; Kronberg, S. L.; Provenza, F. D. Pasture-Finishing of Bison Improves Animal Metabolic Health and Potential Health-Promoting Compounds in Meat. J. Anim. Sci. Biotechnol. 2023, 14 (1), 49, DOI: 10.1186/s40104-023-00843-2Google ScholarThere is no corresponding record for this reference.
- 7Krusinski, L.; Castanon, C.; Hellberg, R. S.; Maciel, I. C. F.; Ahsin, M.; van Vliet, S.; Rowntree, J. E.; Fenton, J. I. Highly Targeted Metabolomics Coupled With Gene Expression Analysis by RT–qPCR Improves Beef Separation Based on Grass, Grain, or Grape Supplemented Diet. Food Front. 2025, 6 (3), 1483– 1497, DOI: 10.1002/fft2.70022Google ScholarThere is no corresponding record for this reference.
- 8Provenza, F. D.; Meuret, M.; Gregorini, P. Our Landscapes, Our Livestock, Ourselves: Restoring Broken Linkages among Plants, Herbivores, and Humans with Diets That Nourish and Satiate. Appetite 2015, 95, 500– 519, DOI: 10.1016/j.appet.2015.08.004Google ScholarThere is no corresponding record for this reference.
- 9Villalba, J. J.; Beauchemin, K. A.; Gregorini, P.; MacAdam, J. W. Pasture Chemoscapes and Their Ecological Services. Transl. Anim. Sci. 2019, 3 (2), 829– 841, DOI: 10.1093/tas/txz003Google ScholarThere is no corresponding record for this reference.
- 10Distel, R. A.; Arroquy, J. I.; Lagrange, S.; Villalba, J. J. Designing Diverse Agricultural Pastures for Improving Ruminant Production Systems. Front. Sustain. Food Syst. 2020, 4, 596869 DOI: 10.3389/fsufs.2020.596869Google ScholarThere is no corresponding record for this reference.
- 11Pereira, F. C.; Gregorini, P. Applying Spatio-Chemical Analysis to Grassland Ecosystems for the Illustration of Chemoscapes and Creation of Healthscapes. Front. Sustain. Food Syst. 2022, 6, 927568 DOI: 10.3389/fsufs.2022.927568Google ScholarThere is no corresponding record for this reference.
- 12Ahsin, M.; Poore, M. H.; Rogers, J.; Franzluebbers, A.; Young, S. N.; Kronberg, S. L.; Provenza, F. D.; Bain, J. R.; van Vliet, S. Soil and Pasture Health Underlie Improved Beef Nutrient Density Determined by Untargeted Metabolomics in Southern US Grass Finished Beef Systems. Npj Sci. Food 2025, 9 (1), 151, DOI: 10.1038/s41538-025-00471-2Google ScholarThere is no corresponding record for this reference.
- 13Kumar, A.; P, N.; Kumar, M.; Jose, A.; Tomer, V.; Oz, E.; Proestos, C.; Zeng, M.; Elobeid, T.; K, S.; Oz, F. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules 2023, 28, 887, DOI: 10.3390/molecules28020887Google ScholarThere is no corresponding record for this reference.
- 14Hou, Q.; Wang, Y.; Hu, J.; Zhang, J.; Zhang, C.; Song, W.; Wang, X.; Zheng, B.; Zhou, X. Simultaneous Determination of Phenothiazine Drugs and Their Metabolites Residues in Animal Derived Foods by High Performance Liquid Chromatography Tandem Mass Spectrometry. Food Control 2025, 167, 110799 DOI: 10.1016/j.foodcont.2024.110799Google ScholarThere is no corresponding record for this reference.
- 15Delgadillo-Puga, C.; Cuchillo-Hilario, M.; León-Ortiz, L.; Ramírez-Rodríguez, A.; Cabiddu, A.; Navarro-Ocaña, A.; Morales-Romero, A. M.; Medina-Campos, O. N.; Pedraza-Chaverri, J. Goats’ Feeding Supplementation with Acacia Farnesiana Pods and Their Relationship with Milk Composition: Fatty Acids, Polyphenols, and Antioxidant Activity. Animals 2019, 9 (8), 515, DOI: 10.3390/ani9080515Google ScholarThere is no corresponding record for this reference.
- 16Delgadillo-Puga, C.; Noriega, L. G.; Morales-Romero, A. M.; Nieto-Camacho, A.; Granados-Portillo, O.; Rodríguez-López, L. A.; Alemán, G.; Furuzawa-Carballeda, J.; Tovar, A. R.; Cisneros-Zevallos, L.; Torre-Villalvazo, I. Goat’s Milk Intake Prevents Obesity, Hepatic Steatosis and Insulin Resistance in Mice Fed A High-Fat Diet by Reducing Inflammatory Markers and Increasing Energy Expenditure and Mitochondrial Content in Skeletal Muscle. Int. J. Mol. Sci. 2020, 21 (15), 5530, DOI: 10.3390/ijms21155530Google ScholarThere is no corresponding record for this reference.
- 17Sofi, F.; Buccioni, A.; Cesari, F.; Gori, A. M.; Minieri, S.; Mannini, L.; Casini, A.; Gensini, G. F.; Abbate, R.; Antongiovanni, M. Effects of a Dairy Product (Pecorino Cheese) Naturally Rich in Cis-9, Trans-11 Conjugated Linoleic Acid on Lipid, Inflammatory and Haemorheological Variables: A Dietary Intervention Study. Nutr. Metab. Cardiovasc. Dis. 2010, 20 (2), 117– 124, DOI: 10.1016/j.numecd.2009.03.004Google ScholarThere is no corresponding record for this reference.
- 18Yang, M.; Sun, J.; Lu, Z.; Chen, G.; Guan, S.; Liu, X.; Jiang, B.; Ye, M.; Guo, D. Phytochemical Analysis of Traditional Chinese Medicine Using Liquid Chromatography Coupled with Mass Spectrometry. J. Chromatogr. A 2009, 1216 (11), 2045– 2062, DOI: 10.1016/j.chroma.2008.08.097Google ScholarThere is no corresponding record for this reference.
- 19Wang, B.; Sun, Z.; Tu, Y.; Si, B.; Liu, Y.; Yang, L.; Luo, H.; Yu, Z. Untargeted Metabolomic Investigate Milk and Ruminal Fluid of Holstein Cows Supplemented with Perilla Frutescens Leaf. Food Res. Int. 2021, 140, 110017 DOI: 10.1016/j.foodres.2020.110017Google ScholarThere is no corresponding record for this reference.
- 20Rousu, T.; Herttuainen, J.; Tolonen, A. Comparison of Triple Quadrupole, Hybrid Linear Ion Trap Triple Quadrupole, Time-of-Flight and LTQ-Orbitrap Mass Spectrometers in Drug Discovery Phase Metabolite Screening and Identification in Vitro – Amitriptyline and Verapamil as Model Compounds. Rapid Commun. Mass Spectrom. 2010, 24 (7), 939– 957, DOI: 10.1002/rcm.4465Google ScholarThere is no corresponding record for this reference.
- 21Guironnet, A.; Wiest, L.; Vulliet, E. Advantages of MS/MS/MS (MRM3) vs Classic MRM Quantification for Complex Environmental Matrices: Analysis of Beta-Lactams in WWTP Sludge. Anal. Chim. Acta 2022, 1205, 339773 DOI: 10.1016/j.aca.2022.339773Google ScholarThere is no corresponding record for this reference.
- 22Kasperkiewicz, A.; Pawliszyn, J. Multiresidue Pesticide Quantitation in Multiple Fruit Matrices via Automated Coated Blade Spray and Liquid Chromatography Coupled to Triple Quadrupole Mass Spectrometry. Food Chem. 2021, 339, 127815 DOI: 10.1016/j.foodchem.2020.127815Google ScholarThere is no corresponding record for this reference.
- 23Sleiman, H.; De Oliveira, J. M.; De Freitas, G. B. L. Isoflavones Alter Male and Female Fertility in Different Development Windows. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 140, 111448 DOI: 10.1016/j.biopha.2021.111448Google ScholarThere is no corresponding record for this reference.
- 24Li, D.; Dang, D.; Xu, S.; Tian, Y.-M.; Wu, D.; Su, Y. Soy Isoflavones Supplementation Improves Reproductive Performance and Serum Antioxidant Status of Sows and the Growth Performance of Their Offspring. J. Anim. Physiol. Anim. Nutr. 2022, 106, 1268, DOI: 10.1111/jpn.13667Google ScholarThere is no corresponding record for this reference.
- 25Min, B.; Barry, T.; Attwood, G.; McNabb, W. The Effect of Condensed Tannins on the Nutrition and Health of Ruminants Fed Fresh Temperate Forages: A Review. Anim. Feed Sci. Technol. 2003, 106, 3– 19, DOI: 10.1016/S0377-8401(03)00041-5Google ScholarThere is no corresponding record for this reference.
- 26Kelln, B.; Penner, G.; Acharya, S.; McAllister, T.; Lardner, H. Impact of Condensed Tannin-Containing Legumes on Ruminal Fermentation, Nutrition, and Performance in Ruminants: A Review. Can. J. Anim. Sci. 2021, 101, 210– 223, DOI: 10.1139/cjas-2020-0096Google ScholarThere is no corresponding record for this reference.
- 27Hossain, Md. S.; Wazed, M. A.; Asha, S.; Amin, M. R.; Shimul, I. M. Dietary Phytochemicals in Health and Disease: Mechanisms, Clinical Evidence, and Applications─A Comprehensive Review. Food Sci. Nutr. 2025, 13, e70101 DOI: 10.1002/fsn3.70101Google ScholarThere is no corresponding record for this reference.
- 28Tava, A.; Biazzi, E.; Ronga, D.; Pecetti, L.; Avato, P. Biologically Active Compounds from Forage Plants. Phytochem. Rev. 2022, 21 (2), 471– 501, DOI: 10.1007/s11101-021-09779-9Google ScholarThere is no corresponding record for this reference.
- 29Krusinski, L.; Sergin, S.; Jambunathan, V.; Rowntree, J. E.; Fenton, J. I. Attention to the Details: How Variations in U.S. Grass-Fed Cattle-Feed Supplementation and Finishing Date Influence Human Health. Front. Sustain. Food Syst. 2022, 6, 851494 DOI: 10.3389/fsufs.2022.851494Google ScholarThere is no corresponding record for this reference.
- 30Provenza, F. D.; Villalba, J. J.; Dziba, L. E.; Atwood, S. B.; Banner, R. E. Linking Herbivore Experience, Varied Diets, and Plant Biochemical Diversity. Adv. Res. Nutr. Sheep Goats Spec. Ref. Pasture Rangel. Use 2003, 49 (3), 257– 274, DOI: 10.1016/S0921-4488(03)00143-3Google ScholarThere is no corresponding record for this reference.
- 31Soder, K. J.; Gregorini, P.; Scaglia, G.; Rook, A. J. Dietary Selection by Domestic Grazing Ruminants in Temperate Pastures: Current State of Knowledge, Methodologies, and Future Direction. Rangel. Ecol. Manag. 2009, 62 (5), 389– 398, DOI: 10.2111/08-068.1Google ScholarThere is no corresponding record for this reference.
- 32Provenza, F. D. What Does It Mean to Be Locally Adapted and Who Cares Anyway?. J. Anim. Sci. 2008, 86 (suppl_14), E271– E284, DOI: 10.2527/jas.2007-0468Google ScholarThere is no corresponding record for this reference.
- 33Reynaud, A.; Fraisse, D.; Cornu, A.; Farruggia, A.; Pujos-Guillot, E.; Besle, J.-M.; Martin, B.; Lamaison, J.-L.; Paquet, D.; Doreau, M.; Graulet, B. Variation in Content and Composition of Phenolic Compounds in Permanent Pastures According to Botanical Variation. J. Agric. Food Chem. 2010, 58 (9), 5485– 5494, DOI: 10.1021/jf1000293Google ScholarThere is no corresponding record for this reference.
- 34Fleming, A.; Wescombe, P.; Gregorini, P. Review: A Vade-Mecum for Ruminant Grazing and Health. animal 2025, 101548 DOI: 10.1016/j.animal.2025.101548Google ScholarThere is no corresponding record for this reference.
- 35Beck, M. R.; Gregorini, P. How Dietary Diversity Enhances Hedonic and Eudaimonic Well-Being in Grazing Ruminants. Front. Vet. Sci. 2020, 7, 191, DOI: 10.3389/fvets.2020.00191Google ScholarThere is no corresponding record for this reference.
- 36Kearns, M.; Ponnampalam, E. N.; Jacquier, J.-C.; Grasso, S.; Boland, T. M.; Sheridan, H.; Monahan, F. J. Can Botanically-Diverse Pastures Positively Impact the Nutritional and Antioxidant Composition of Ruminant Meat? – Invited Review. Meat Sci. 2023, 197, 109055 DOI: 10.1016/j.meatsci.2022.109055Google ScholarThere is no corresponding record for this reference.
- 37Schreiner, B.; Ribeiro, G.; Lardner, H.; Penner, G. PSVIII-17 A Comparison of a Monoculture Barley Crop to a Barley-Based Complex Mixture on Forage Yield, Quality, Dry Matter Intake, Enteric Methane Emissions and Growth Performance of Pregnant Yearling Heifers. J. Anim. Sci. 2024, 102, 582, DOI: 10.1093/jas/skae234.655Google ScholarThere is no corresponding record for this reference.
- 38Muñoz, C.; Letelier, P.; Ungerfeld, E.; Morales, J.; Hube, S.; Pérez-Prieto, L. Effects of Pregrazing Herbage Mass in Late Spring on Enteric Methane Emissions, Dry Matter Intake, and Milk Production of Dairy Cows. J. Dairy Sci. 2016, 99 (10), 7945– 7955, DOI: 10.3168/jds.2016-10919Google ScholarThere is no corresponding record for this reference.
- 39Segueni, K.; Chouikh, A.; Laouini, S. E.; Bouafia, A.; Tlili, M. L.; Laib, I.; Boudebia, O.; Khelef, Y.; Abdullah, M.; Abdullah, J. A. A.; Emran, T. B. Evaluation of Dermal Wound Healing Potential: Phytochemical Characterization, Anti-Inflammatory, Antioxidant, and Antimicrobial Activities of Euphorbia Guyoniana Boiss. & Reut. Latex. Chem. Biodivers. 2024, 22, e202402284 DOI: 10.1002/cbdv.202402284Google ScholarThere is no corresponding record for this reference.
- 40Riaz, A.; Ali, S.; Summer, M.; Noor, S.; Nazakat, L.; Aqsa; Sharjeel, M. Exploring the Underlying Pharmacological, Immunomodulatory, and Anti-Inflammatory Mechanisms of Phytochemicals against Wounds: A Molecular Insight. Inflammopharmacology 2024, 32, 2695, DOI: 10.1007/s10787-024-01545-5Google ScholarThere is no corresponding record for this reference.
- 41Redan, B. W.; Buhman, K. K.; Novotny, J. A.; Ferruzzi, M. G. Altered Transport and Metabolism of Phenolic Compounds in Obesity and Diabetes: Implications for Functional Food Development and Assessment. Adv. Nutr. 2016, 7 (6), 1090– 1104, DOI: 10.3945/an.116.013029Google ScholarThere is no corresponding record for this reference.
- 42Monfalouti, H. E.; Kartah, B. E.; Monfalouti, H. E.; Kartah, B. E. Enhancing Polyphenol Bioavailability through Nanotechnology: Current Trends and Challenges; IntechOpen, 2024.Google ScholarThere is no corresponding record for this reference.
- 43Kasapidou, E.; Iliadis, I.-V.; Mitlianga, P.; Papatzimos, G.; Karatzia, M.-A.; Papadopoulos, V.; Amanatidis, M.; Tortoka, V.; Tsiftsi, E.; Aggou, A.; Basdagianni, Z. Variations in Composition, Antioxidant Profile, and Physical Traits of Goat Milk within the Semi-Intensive Production System in Mountainous Areas during the Post-Weaning to End-of-Lactation Period. Animals 2023, 13 (22), 3505, DOI: 10.3390/ani13223505Google ScholarThere is no corresponding record for this reference.
- 44Lee, K.; Kim, Y.; Lee, H.; Lee, C. Y. Cocoa Has More Phenolic Phytochemicals and a Higher Antioxidant Capacity than Teas and Red Wine. J. Agric. Food Chem. 2003, 51 (25), 7292– 7295, DOI: 10.1021/jf0344385Google ScholarThere is no corresponding record for this reference.
- 45Martín, M.; Goya, L.; Ramos, S. Protective Effects of Tea, Red Wine and Cocoa in Diabetes. Evidences from Human Studies. Food Chem. Toxicol. 2017, 109 (Pt 1), 302– 314, DOI: 10.1016/j.fct.2017.09.015Google ScholarThere is no corresponding record for this reference.
- 46Ohishi, T.; Fukutomi, R.; Shoji, Y.; Goto, S.; Isemura, M. The Beneficial Effects of Principal Polyphenols from Green Tea, Coffee, Wine, and Curry on Obesity. Molecules 2021, 26, 453, DOI: 10.3390/molecules26020453Google ScholarThere is no corresponding record for this reference.
- 47van Vliet, S.; Bain, J. R.; Muehlbauer, M. J.; Provenza, F. D.; Kronberg, S. L.; Pieper, C. F.; Huffman, K. M. A Metabolomics Comparison of Plant-Based Meat and Grass-Fed Meat Indicates Large Nutritional Differences despite Comparable Nutrition Facts Panels. Sci. Rep. 2021, 11 (1), 13828, DOI: 10.1038/s41598-021-93100-3Google ScholarThere is no corresponding record for this reference.
- 48Gregorini, P.; Provenza, F.; Kronberg, S. Is Grassfed Meat and Dairy Better for Human and Environmental Health?. Front. Nutr. 2019, 6, 26, DOI: 10.3389/fnut.2019.00026Google ScholarThere is no corresponding record for this reference.
- 49Agulló, V.; Favari, C.; Pilla, N.; Bresciani, L.; Tomás-Barberán, F. A.; Crozier, A.; Del Rio, D.; Mena, P. Using Targeted Metabolomics to Unravel Phenolic Metabolites of Plant Origin in Animal Milk. Int. J. Mol. Sci. 2024, 25 (8), 4536, DOI: 10.3390/ijms25084536Google ScholarThere is no corresponding record for this reference.
- 50Page, M. J.; McKenzie, J. E.; Bossuyt, P. M.; Boutron, I.; Hoffmann, T. C.; Mulrow, C. D.; Shamseer, L.; Tetzlaff, J. M.; Akl, E. A.; Brennan, S. E.; Chou, R.; Glanville, J.; Grimshaw, J. M.; Hróbjartsson, A.; Lalu, M. M.; Li, T.; Loder, E. W.; Mayo-Wilson, E.; McDonald, S.; McGuinness, L. A.; Stewart, L. A.; Thomas, J.; Tricco, A. C.; Welch, V. A.; Whiting, P.; Moher, D. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. PLOS Med. 2021, 18 (3), e1003583 DOI: 10.1371/journal.pmed.1003583Google ScholarThere is no corresponding record for this reference.
- 51Newman, M.; Gough, D. Systematic Reviews in Educational Research: Methodology, Perspectives and Application. Syst. Rev. Educ. Res. Methodol. Perspect. Appl. 2020, 3– 22, DOI: 10.1007/978-3-658-27602-7_1Google ScholarThere is no corresponding record for this reference.
- 52Hooijmans, C. R.; Rovers, M. M.; De Vries, R. B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M. W. SYRCLE’s Risk of Bias Tool for Animal Studies. BMC Med. Res. Methodol. 2014, 14, 43, DOI: 10.1186/1471-2288-14-43Google ScholarThere is no corresponding record for this reference.
- 53Zhang, W.; Jiang, Y.; Shang, Z.; Zhang, N.; Tao, G.; Zhang, T.; Hu, K.; Li, Y.; Shi, X.; Zhang, Y.; Yang, J.; Ma, B.; Yang, K. The Methodological Quality of Animal Studies: A Cross-Sectional Study Based on the SYRCLE’s Risk of Bias Tool. bioRxiv 2019, DOI: DOI: 10.1101/701110 .Google ScholarThere is no corresponding record for this reference.
- 54Bennato, F.; Ianni, A.; Oliva, E.; Franceschini, N.; Grotta, L.; Sergi, M.; Martino, G. Characterization of Phenolic Profile in Milk Obtained by Ewes Fed Grape Pomace: Reflection on Antioxidant and Anti-Inflammatory Status. Biomolecules 2023, 13 (7), 1026, DOI: 10.3390/biom13071026Google ScholarThere is no corresponding record for this reference.
- 55Lee, J.-E.; Jayakody, J. T.; Kim, J.-I.; Jeong, J.-W.; Choi, K.-M.; Kim, T.-S.; Seo, C.; Azimi, I.; Hyun, J.; Ryu, B. The Influence of Solvent Choice on the Extraction of Bioactive Compounds from Asteraceae: A Comparative Review. Foods 2024, 13 (19), 3151, DOI: 10.3390/foods13193151Google ScholarThere is no corresponding record for this reference.
- 56Rocchetti, G.; Becchi, P. P.; Salis, L.; Lucini, L.; Cabiddu, A. Impact of Pasture-Based Diets on the Untargeted Metabolomics Profile of Sarda Sheep Milk. Foods 2023, 12 (1), 143, DOI: 10.3390/foods12010143Google ScholarThere is no corresponding record for this reference.
- 57Hernandez, M. S.; Kim, Y. H. B.; Woerner, D. R.; Brooks, J. C.; Legako, J. F. Untargeted Metabolomics Reveals Divergent Metabolomes between Three Plant-Based Meat Alternatives and Two Lean Levels of Ground Beef. ACS Food Sci. Technol. 2025, 5 (4), 1632– 1644, DOI: 10.1021/acsfoodscitech.5c00041Google ScholarThere is no corresponding record for this reference.
- 58Mokrani, A.; Madani, K. Effect of Solvent, Time and Temperature on the Extraction of Phenolic Compounds and Antioxidant Capacity of Peach (Prunus Persica L.). Fruit. Sep. Purif. Technol. 2016, 162, 68– 76, DOI: 10.1016/j.seppur.2016.01.043Google ScholarThere is no corresponding record for this reference.
- 59Di-Grigoli, A.; Bonanno, A.; Rabie Ashkezary, M.; Laddomada, B.; Alabiso, M.; Vitale, F.; Mazza, F.; Maniaci, G.; Ruisi, P.; Di Miceli, G. Meat Production from Dairy Breed Lambs Due to Slaughter Age and Feeding Plan Based on Wheat Bran. Animals 2019, 9 (11), 892, DOI: 10.3390/ani9110892Google ScholarThere is no corresponding record for this reference.
- 60Bennato, F.; Ianni, A.; Florio, M.; Grotta, L.; Pomilio, F.; Saletti, M. A.; Martino, G. Nutritional Properties of Milk from Dairy Ewes Fed with a Diet Containing Grape Pomace. Foods 2022, 11 (13), 1878, DOI: 10.3390/foods11131878Google ScholarThere is no corresponding record for this reference.
- 61Chávez-Servín, J. L.; Andrade-Montemayor, H. M.; Velázquez Vázquez, C.; Aguilera Barreyro, A.; García-Gasca, T.; Ferríz Martínez, R. A.; Olvera Ramírez, A. M.; De La Torre-Carbot, K. Effects of Feeding System, Heat Treatment and Season on Phenolic Compounds and Antioxidant Capacity in Goat Milk, Whey and Cheese. Small Rumin. Res. 2018, 160, 54– 58, DOI: 10.1016/j.smallrumres.2018.01.011Google ScholarThere is no corresponding record for this reference.
- 62Di-Trana, A.; Bonanno, A.; Cecchini, S.; Giorgio, D.; Di Grigoli, A.; Claps, S. Effects of Sulla Forage (Sulla Coronarium L.) on the Oxidative Status and Milk Polyphenol Content in Goats. J. Dairy Sci. 2015, 98 (1), 37– 46, DOI: 10.3168/jds.2014-8414Google ScholarThere is no corresponding record for this reference.
- 63Ianni, A.; Innosa, D.; Oliva, E.; Bennato, F.; Grotta, L.; Saletti, M. A.; Pomilio, F.; Sergi, M.; Martino, G. Effect of Olive Leaves Feeding on Phenolic Composition and Lipolytic Volatile Profile in Goat Milk. J. Dairy Sci. 2021, 104 (8), 8835– 8845, DOI: 10.3168/jds.2021-20211Google ScholarThere is no corresponding record for this reference.
- 64Leparmarai, P. T.; Sinz, S.; Kunz, C.; Liesegang, A.; Ortmann, S.; Kreuzer, M.; Marquardt, S. Transfer of Total Phenols from a grapeseed-Supplemented Diet to Dairy Sheep and Goat Milk, and Effects on Performance and Milk Quality1. J. Anim. Sci. 2019, 97 (4), 1840– 1851, DOI: 10.1093/jas/skz046Google ScholarThere is no corresponding record for this reference.
- 65Leparmarai, P. T.; Kunz, C.; Mwangi, D. M.; Gluecks, I.; Kreuzer, M.; Marquardt, S. Camels and Cattle Respond Differently in Milk Phenol Excretion and Milk Fatty Acid Profile to Free Ranging Conditions in East-African Rangelands. Sci. Afr. 2021, 13, e00896 DOI: 10.1016/j.sciaf.2021.e00896Google ScholarThere is no corresponding record for this reference.
- 66Sik, B.; Buzás, H.; Kapcsándi, V.; Lakatos, E.; Daróczi, F.; Székelyhidi, R. Antioxidant and Polyphenol Content of Different Milk and Dairy Products. J. King Saud Univ. - Sci. 2023, 35 (7), 102839 DOI: 10.1016/j.jksus.2023.102839Google ScholarThere is no corresponding record for this reference.
- 67Ainsworth, E. A.; Gillespie, K. M. Estimation of Total Phenolic Content and Other Oxidation Substrates in Plant Tissues Using Folin–Ciocalteu Reagent. Nat. Protoc. 2007, 2 (4), 875– 877, DOI: 10.1038/nprot.2007.102Google ScholarThere is no corresponding record for this reference.
- 68Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L. D.; Hidalgo, F. J.; Miyashita, K.; Camp, J. van; Alasalvar, C.; Ismail, A. B.; Elmore, S.; Birch, G. G.; Charalampopoulos, D.; Astley, S. B.; Pegg, R.; Zhou, P.; Finglas, P. Antioxidant Activity, Total phenolics and Flavonoids Contents: Should We Ban in Vitro Screening Methods?. Food Chem. 2018, 264, 471– 475, DOI: 10.1016/j.foodchem.2018.04.012Google ScholarThere is no corresponding record for this reference.
- 69Chociej, P.; Foss, K.; Jabłońska, M.; Ustarbowska, M.; Sawicki, T. The Profile and Content of Polyphenolic Compounds and Antioxidant and Anti-Glycation Properties of Root Extracts of Selected Medicinal Herbs. Plant Foods Hum. Nutr. 2024, 79 (2), 468– 473, DOI: 10.1007/s11130-024-01180-zGoogle ScholarThere is no corresponding record for this reference.
- 70Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 370438, DOI: 10.3389/fnut.2018.00087Google ScholarThere is no corresponding record for this reference.
- 71Donato, P.; Cacciola, F.; Tranchida, P. Q.; Dugo, P.; Mondello, L. Mass Spectrometry Detection in Comprehensive Liquid Chromatography: Basic Concepts, Instrumental Aspects. Applications and Trends. Mass Spectrom. Rev. 2012, 31 (5), 523– 559, DOI: 10.1002/mas.20353Google ScholarThere is no corresponding record for this reference.
- 72Lamuela-Raventós, R. M.; Vallverdú-Queralt, A.; Jáuregui, O.; Martínez-Huélamo, M.; Quifer-Rada, P. Chapter 14 - Improved Characterization of Polyphenols Using Liquid Chromatography. In Polyphenols in Plants; Watson, R. R., Ed.; Academic Press: San Diego, 2014; pp. 261– 292.Google ScholarThere is no corresponding record for this reference.
- 73Andersen, C.; Nielsen, T. S.; Purup, S.; Kristensen, T.; Eriksen, J.; So̷egaard, K.; So̷rensen, J.; Fretté, X. C. Phyto-Oestrogens in Herbage and Milk from Cows Grazing White Clover, Red Clover, Lucerne or Chicory-Rich Pastures. Animal 2009, 3 (8), 1189– 1195, DOI: 10.1017/S1751731109004613Google ScholarThere is no corresponding record for this reference.
- 74Hoikkala, A.; Mustonen, E.; Saastamoinen, I.; Jokela, T.; Taponen, J.; Saloniemi, H.; Wähälä, K. High Levels of Equol in Organic Skimmed Finnish Cow Milk. Mol. Nutr. Food Res. 2007, 51 (7), 782– 786, DOI: 10.1002/mnfr.200600222Google ScholarThere is no corresponding record for this reference.
- 75Mustonen, E. A.; Tuori, M.; Saastamoinen, I.; Taponen, J.; Wähälä, K.; Saloniemi, H.; Vanhatalo, A. Equol in Milk of Dairy Cows Is Derived from Forage Legumes Such as Red Clover. Br. J. Nutr. 2009, 102 (11), 1552, DOI: 10.1017/S0007114509990857Google ScholarThere is no corresponding record for this reference.
- 76Třináctý, J.; Křížová, L.; Schulzová, V.; Hajšlová, J.; Hanuš, O. The Effect of Feeding Soybean-Derived Phytoestogens on Their Concentration in Plasma and Milk of Lactating Dairy Cows. Arch. Anim. Nutr. 2009, 63 (3), 219– 229, DOI: 10.1080/17450390902859739Google ScholarThere is no corresponding record for this reference.
- 77Alonso, A.; Marsal, S.; Julià, A. Analytical Methods in Untargeted Metabolomics: State of the Art in 2015. Front. Bioeng. Biotechnol. 2015, 3, 23, DOI: 10.3389/fbioe.2015.00023Google ScholarThere is no corresponding record for this reference.
- 78Zarrouk, E.; El Balkhi, S.; Saint-Marcoux, F. Critical Evaluation of High-Resolution and Low-Resolution Mass Spectrometry for Biomonitoring of Human Environmental Exposure to Pesticides. Environ. Technol. Innov. 2024, 36, 103834 DOI: 10.1016/j.eti.2024.103834Google ScholarThere is no corresponding record for this reference.
- 79Zheng, F.; Zhao, X.; Zeng, Z.; Wang, L.; Lv, W.; Wang, Q.; Xu, G. Development of a Plasma Pseudotargeted Metabolomics Method Based on Ultra-High-Performance Liquid Chromatography–Mass Spectrometry. Nat. Protoc. 2020, 15, 2519– 2537, DOI: 10.1038/s41596-020-0341-5Google ScholarThere is no corresponding record for this reference.
- 80Wang, W.; Sun, B.; Hu, P.; Zhou, M.; Sun, S.; Du, P.; Ru, Y.; Suvorov, A.; Li, Y.; Liu, Y.; Wang, S. Comparison of Differential Flavor Metabolites in Meat of Lubei White Goat, Jining Gray Goat and Boer Goat. Metabolites 2019, 9 (9), 176, DOI: 10.3390/metabo9090176Google ScholarThere is no corresponding record for this reference.
- 81Ottaviani, J. I.; Fong, R. Y.; Borges, G.; Schroeter, H.; Crozier, A. Use of LC-MS for the Quantitative Analysis of (Poly)Phenol Metabolites Does Not Necessarily Yield Accurate Results: Implications for Assessing Existing Data and Conducting Future Research. Free Radic. Biol. Med. 2018, 124, 97– 103, DOI: 10.1016/j.freeradbiomed.2018.05.092Google ScholarThere is no corresponding record for this reference.
- 82Bloszies, C. S.; Fiehn, O. Using Untargeted Metabolomics for Detecting Exposome Compounds. Mech. Toxicol. Metab. Disrupt. Environ. Dis. 2018, 8, 87– 92, DOI: 10.1016/j.cotox.2018.03.002Google ScholarThere is no corresponding record for this reference.
- 83Fang, X.; Liu, Y.; Xiao, J.; Ma, C.; Huang, Y. GC–MS and LC-MS/MS Metabolomics Revealed Dynamic Changes of Volatile and Non-Volatile Compounds during Withering Process of Black Tea. Food Chem. 2023, 410, 135396 DOI: 10.1016/j.foodchem.2023.135396Google ScholarThere is no corresponding record for this reference.
- 84Scano, P.; Carta, P.; Ibba, I.; Manis, C.; Caboni, P. An Untargeted Metabolomic Comparison of Milk Composition from Sheep Kept Under Different Grazing Systems. Dairy 2020, 1 (1), 30– 41, DOI: 10.3390/dairy1010004Google ScholarThere is no corresponding record for this reference.
- 85Jiao, L.; Guo, Y.; Chen, J.; Zhao, X.; Dong, D. Detecting Volatile Compounds in Food by Open-Path Fourier-Transform Infrared Spectroscopy. Food Res. Int. 2019, 119, 968– 973, DOI: 10.1016/j.foodres.2018.11.042Google ScholarThere is no corresponding record for this reference.
- 86Foroutan, A.; Guo, A. C.; Vazquez-Fresno, R.; Lipfert, M.; Zhang, L.; Zheng, J.; Badran, H.; Budinski, Z.; Mandal, R.; Ametaj, B. N.; Wishart, D. S. Chemical Composition of Commercial Cow’s Milk. J. Agric. Food Chem. 2019, 67 (17), 4897– 4914, DOI: 10.1021/acs.jafc.9b00204Google ScholarThere is no corresponding record for this reference.
- 87Chiriac, E. R.; Chiţescu, C. L.; Geană, E.-I.; Gird, C. E.; Socoteanu, R. P.; Boscencu, R. Advanced Analytical Approaches for the Analysis of Polyphenols in Plants Matrices─A Review. Separations 2021, 8 (5), 65, DOI: 10.3390/separations8050065Google ScholarThere is no corresponding record for this reference.
- 88Teng, H.; Chen, L. Polyphenols and Bioavailability: An Update. Crit. Rev. Food Sci. Nutr. 2019, 59 (13), 2040– 2051, DOI: 10.1080/10408398.2018.1437023Google ScholarThere is no corresponding record for this reference.
- 89Chesson, A.; Provan, G. J.; Russell, W. R.; Scobbie, L.; Richardson, A. J.; Stewart, C. Hydroxycinnamic Acids in the Digestive Tract of Livestock and Humans. J. Sci. Food Agric. 1999, 79 (3), 373– 378, DOI: 10.1002/(SICI)1097-0010(19990301)79:3<373::AID-JSFA257>3.3.CO;2-YGoogle ScholarThere is no corresponding record for this reference.
- 90Romero, P.; Huang, R.; Jiménez, E.; Palma-Hidalgo, J.; Ungerfeld, E.; Popova, M.; Morgavi, D.; Belanche, A.; Yáñez-Ruiz, D. Evaluating the Effect of Phenolic Compounds as Hydrogen Acceptors When Ruminal Methanogenesis Is Inhibited in Vitro - Part 2. Dairy Goats. Anim. Int. J. Anim. Biosci. 2023, 17 (5), 100789 DOI: 10.1016/j.animal.2023.100789Google ScholarThere is no corresponding record for this reference.
- 91Krumholz, L.; Bryant, M. Eubacterium Oxidoreducens Sp. Nov. Requiring H2 or Formate to Degrade Gallate, Pyrogallol, Phloroglucinol and Quercetin. Arch. Microbiol. 1986, 144, 8– 14, DOI: 10.1007/BF00454948Google ScholarThere is no corresponding record for this reference.
- 92Martin, A. K. The Origin of Urinary Aromatic Compounds Excreted by Ruminants 2. The Metabolism of Phenolic Cinnamic Acids to Benzoic Acid. Br. J. Nutr. 1982, 47 (1), 155– 164, DOI: 10.1079/BJN19820020Google ScholarThere is no corresponding record for this reference.
- 93Batterham, T.; Shutt, D.; Hart, N.; Braden, A.; Tweeddale, H. Metabolism of Intraruminally Administered [4–14C]Formononetic and [4–14C]Biochanin a in Sheep. Crop Pasture Sci. 1971, 22, 131– 138, DOI: 10.1071/AR9710131Google ScholarThere is no corresponding record for this reference.
- 94Trnková, A.; Šancová, K.; Zapletalová, M.; Kašparovská, J.; Dadáková, K.; Křížová, L.; Lochman, J.; Hadrová, S.; Ihnatová, I.; Kašparovský, T. Determination of in Vitro Isoflavone Degradation in Rumen Fluid. J. Dairy Sci. 2018, 101 (6), 5134– 5144, DOI: 10.3168/jds.2017-13610Google ScholarThere is no corresponding record for this reference.
- 95Guo, Y.; Weber, W. J.; Yao, D.; Caixeta, L.; Zimmerman, N. P.; Thompson, J.; Block, E.; Rehberger, T. G.; Crooker, B. A.; Chen, C. Forming 4-Methylcatechol as the Dominant Bioavailable Metabolite of Intraruminal Rutin Inhibits P-Cresol Production in Dairy Cows. Metabolites 2022, 12 (1), 16, DOI: 10.3390/metabo12010016Google ScholarThere is no corresponding record for this reference.
- 96Berger, L. M.; Blank, R.; Zorn, F.; Wein, S.; Metges, C. C.; Wolffram, S. Ruminal Degradation of Quercetin and Its Influence on Fermentation in Ruminants. J. Dairy Sci. 2015, 98 (8), 5688– 5698, DOI: 10.3168/jds.2015-9633Google ScholarThere is no corresponding record for this reference.
- 97Terrill, T. H.; Waghorn, G. C.; Woolley, D. J.; Mcnabb, W. C.; Barry, T. N. Assay and Digestion of 14C-Labelled Condensed Tannins in the Gastrointestinal Tract of Sheep. Br. J. Nutr. 1994, 72 (3), 467– 477, DOI: 10.1079/BJN19940048Google ScholarThere is no corresponding record for this reference.
- 98Gladine, C.; Rock, E.; Morand, C.; Bauchart, D.; Durand, D. Bioavailability and Antioxidant Capacity of Plant Extracts Rich in Polyphenols, given as a Single Acute Dose, in Sheep Made Highly Susceptible to Lipoperoxidation. Br. J. Nutr. 2007, 98 (4), 691– 701, DOI: 10.1017/S0007114507742666Google ScholarThere is no corresponding record for this reference.
- 99Gessner, D. K.; Ringseis, R.; Eder, K. Potential of Plant Polyphenols to Combat Oxidative Stress and Inflammatory Processes in Farm Animals. J. Anim. Physiol. Anim. Nutr. 2017, 101 (4), 605– 628, DOI: 10.1111/jpn.12579Google ScholarThere is no corresponding record for this reference.
- 100Day, A.; Cañada, F.; Díaz, J.; Kroon, P.; Mclauchlan, R.; Faulds, C.; Plumb, G.; Morgan, M.; Williamson, G. Dietary Flavonoid and Isoflavone Glycosides Are Hydrolysed by the Lactase Site of Lactase Phlorizin Hydrolase. FEBS Lett. 2000, 468, 166– 170, DOI: 10.1016/S0014-5793(00)01211-4Google ScholarThere is no corresponding record for this reference.
- 101Wang, X.; Qi, Y.; Zheng, H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants 2022, 11 (6), 1212, DOI: 10.3390/antiox11061212Google ScholarThere is no corresponding record for this reference.
- 102Zeb, A. Metabolism of Phenolic Antioxidants. In Phenolic Antioxidants in Foods: Chemistry, Biochemistry and Analysis; Zeb, A., Ed.; Springer International Publishing: Cham, 2021; pp. 333– 383.Google ScholarThere is no corresponding record for this reference.
- 103Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D. J.; Preston, T.; Kroon, P. A.; Botting, N. P.; Kay, C. D. Human Metabolism and Elimination of the Anthocyanin, Cyanidin-3-Glucoside: A 13C-Tracer Study123. Am. J. Clin. Nutr. 2013, 97 (5), 995– 1003, DOI: 10.3945/ajcn.112.049247Google ScholarThere is no corresponding record for this reference.
- 104Renouf, M.; Guy, P. A.; Marmet, C.; Fraering, A.-L.; Longet, K.; Moulin, J.; Enslen, M.; Barron, D.; Dionisi, F.; Cavin, C.; Williamson, G.; Steiling, H. Measurement of Caffeic and Ferulic Acid Equivalents in Plasma after Coffee Consumption: Small Intestine and Colon Are Key Sites for Coffee Metabolism. Mol. Nutr. Food Res. 2010, 54 (6), 760– 766, DOI: 10.1002/mnfr.200900056Google ScholarThere is no corresponding record for this reference.
- 105Yang, G.; Ge, S.; Singh, R.; Basu, S.; Shatzer, K.; Zen, M.; Liu, J.; Tu, Y.; Zhang, C.; Wei, J.; Shi, J.; Zhu, L.; Liu, Z.; Wang, Y.; Gao, S.; Hu, M. Glucuronidation: Driving Factors and Their Impact on Glucuronide Disposition. Drug Metab. Rev. 2017, 49 (2), 105– 138, DOI: 10.1080/03602532.2017.1293682Google ScholarThere is no corresponding record for this reference.
- 106Zhang, H.; Tsao, R. Dietary Polyphenols, Oxidative Stress and Antioxidant and Anti-Inflammatory Effects. Food Microbiol. Funct. Foods Nutr. 2016, 8, 33– 42, DOI: 10.1016/j.cofs.2016.02.002Google ScholarThere is no corresponding record for this reference.
- 107Tak, Y.; Kaur, M.; Gautam, C.; Kumar, R.; Tilgam, J.; Natta, S. Phenolic Biosynthesis and Metabolic Pathways to Alleviate Stresses in Plants. In Plant phenolics in Abiotic Stress Management; Lone, R.; Khan, S.; Mohammed Al-Sadi, A., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 63– 87.Google ScholarThere is no corresponding record for this reference.
- 108Kuhnle, G. G. C.; Dell’Aquila, C.; Aspinall, S. M.; Runswick, S. A.; Mulligan, A. A.; Bingham, S. A. Phytoestrogen Content of Foods of Animal Origin: Dairy Products, Eggs, Meat, Fish, and Seafood. J. Agric. Food Chem. 2008, 56 (21), 10099– 10104, DOI: 10.1021/jf801344xGoogle ScholarThere is no corresponding record for this reference.
- 109Steinshamn, H.; Purup, S.; Thuen, E.; Hansen-Mo̷ller, J. Effects of Clover-Grass Silages and Concentrate Supplementation on the Content of Phytoestrogens in Dairy Cow Milk. J. Dairy Sci. 2008, 91 (7), 2715– 2725, DOI: 10.3168/jds.2007-0857Google ScholarThere is no corresponding record for this reference.
- 110Höjer, A.; Adler, S.; Purup, S.; Hansen-Mo̷ller, J.; Martinsson, K.; Steinshamn, H.; Gustavsson, A.-M. Effects of Feeding Dairy Cows Different Legume-Grass Silages on Milk Phytoestrogen Concentration. J. Dairy Sci. 2012, 95 (8), 4526– 4540, DOI: 10.3168/jds.2011-5226Google ScholarThere is no corresponding record for this reference.
- 111Caradus, J.; Voisey, C.; Cousin, G.; Kaur, R.; Woodfield, D.; Blanc, A.; Roldan, M. The Hunt for the “Holy Grail”: Condensed Tannins in the Perennial Forage Legume White Clover (Trifolium Repens L.). Grass Forage Sci. 2022, 77, 111, DOI: 10.1111/gfs.12567Google ScholarThere is no corresponding record for this reference.
- 112Seeno, E.; MacAdam, J.; Melathopoulos, A.; Filley, S.; Ates, S. Management of Perennial Forbs Sown with or without Self-regenerating Annual Clovers for Forage and Nectar Sources in a Low-input Dryland Production System. Grass Forage Sci. 2023, 78, 462, DOI: 10.1111/gfs.12640Google ScholarThere is no corresponding record for this reference.
- 113Besle, J. M.; Viala, D.; Martin, B.; Pradel, P.; Meunier, B.; Berdagué, J. L.; Fraisse, D.; Lamaison, J. L.; Coulon, J. B. Ultraviolet-Absorbing Compounds in Milk Are Related to Forage Polyphenols. J. Dairy Sci. 2010, 93 (7), 2846– 2856, DOI: 10.3168/jds.2009-2939Google ScholarThere is no corresponding record for this reference.
- 114Adler, S. A.; Purup, S.; Hansen-Mo̷ller, J.; Thuen, E.; Steinshamn, H. Phytoestrogens and Their Metabolites in Bulk-Tank Milk: Effects of Farm Management and Season. PLoS One 2015, 10 (5), e0127187 DOI: 10.1371/journal.pone.0127187Google ScholarThere is no corresponding record for this reference.
- 115Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24 (13), 2452, DOI: 10.3390/molecules24132452Google ScholarThere is no corresponding record for this reference.
- 116Er, M.; Keles, G. Buckwheat Conservation as Hay or Silage: Agronomic Evaluation, Nutritive Value, Conservation Quality, and Intake by Lactating Dairy Goats. Trop. Anim. Health Prod. 2021, 53, 215, DOI: 10.1007/s11250-021-02655-wGoogle ScholarThere is no corresponding record for this reference.
- 117Rufino-Moya, P.; Bertolín, J.; Blanco, M.; Lobón, S.; Joy, M. Fatty Acid Profile, Secondary Compounds and Antioxidant Activities in the Fresh Forage, Hay and Silage of Sainfoin (Onobrychis Viciifolia) and Sulla (Hedysarum Coronarium). J. Sci. Food Agric. 2022, 102, 4736– 4743, DOI: 10.1002/jsfa.11834Google ScholarThere is no corresponding record for this reference.
- 118Lv, J.; Jin, S.; Zhang, Y.; Zhou, Y.; Li, M.; Feng, N. Equol: A Metabolite of Gut Microbiota with Potential Antitumor Effects. Gut Pathog. 2024, 16 (1), 35, DOI: 10.1186/s13099-024-00625-9Google ScholarThere is no corresponding record for this reference.
- 119Róin, N. R.; Poulsen, N. A.; No̷rskov, N. P.; Purup, S.; Larsen, L. B. Seasonal Variation in Contents of Phytoestrogens in Danish Dairy Milk Lines of Different Farm Management Systems. Int. Dairy J. 2023, 144, 105694 DOI: 10.1016/j.idairyj.2023.105694Google ScholarThere is no corresponding record for this reference.
- 120Hilario, M. C.; Puga, C. D.; Ocaña, A. N.; Romo, F. P.-G. Antioxidant Activity, Bioactive Polyphenols in Mexican Goats’ Milk Cheeses on Summer Grazing. J. Dairy Res. 2010, 77 (1), 20– 26, DOI: 10.1017/S0022029909990161Google ScholarThere is no corresponding record for this reference.
- 121Manis, C.; Scano, P.; Nudda, A.; Carta, S.; Pulina, G.; Caboni, P. LC-QTOF/MS Untargeted Metabolomics of Sheep Milk under Cocoa Husks Enriched Diet. Dairy 2021, 2 (1), 112– 121, DOI: 10.3390/dairy2010011Google ScholarThere is no corresponding record for this reference.
- 122Zhang, F.; Wang, Y.; Liu, B.; Gong, P.; Shi, C.; Zhu, L.; Zhao, J.; Yao, W.; Liu, Q.; Luo, J. Widely Targeted Metabolomic Analysis Revealed the Diversity in Milk from Goats, Sheep, Cows, and Buffaloes and Its Association with Flavor Profiles. Foods 2024, 13 (9), 1365, DOI: 10.3390/foods13091365Google ScholarThere is no corresponding record for this reference.
- 123No̷rskov, N. P.; Givens, I.; Purup, S.; Stergiadis, S. Concentrations of Phytoestrogens in Conventional, Organic and Free-Range Retail Milk in England. Food Chem. 2019, 295, 1– 9, DOI: 10.1016/j.foodchem.2019.05.081Google ScholarThere is no corresponding record for this reference.
- 124Rocchetti, G.; Ghilardelli, F.; Bonini, P.; Lucini, L.; Masoero, F.; Gallo, A. Changes of Milk Metabolomic Profiles Resulting from a Mycotoxins-Contaminated Corn Silage Intake by Dairy Cows. Metabolites 2021, 11 (8), 475, DOI: 10.3390/metabo11080475Google ScholarThere is no corresponding record for this reference.
- 125Zhang, F.; Zhao, Y.; Wang, H.; Nan, X.; Wang, Y.; Guo, Y.; Xiong, B. Alterations in the Milk Metabolome of Dairy Cows Supplemented with Different Levels of Calcium Propionate in Early Lactation. Metabolites 2022, 12 (8), 699, DOI: 10.3390/metabo12080699Google ScholarThere is no corresponding record for this reference.
- 126Rocchetti, G.; Ghilardelli, F.; Mosconi, M.; Masoero, F.; Gallo, A. Occurrence of Polyphenols, Isoflavonoids, and Their Metabolites in Milk Samples from Different Cow Feeding Regimens. Dairy 2022, 3 (2), 314– 325, DOI: 10.3390/dairy3020024Google ScholarThere is no corresponding record for this reference.
- 127Casula, M.; Scano, P.; Manis, C.; Tolle, G.; Nudda, A.; Carta, S.; Pulina, G.; Caboni, P. UHPLC-QTOF/MS Untargeted Lipidomics and Caffeine Carry-Over in Milk of Goats under Spent Coffee Ground Enriched Diet. Appl. Sci. 2023, 13 (4), 2477, DOI: 10.3390/app13042477Google ScholarThere is no corresponding record for this reference.
- 128Landau, S. Y.; Glasser, T. A.; Zachut, M.; Klein, J. D.; Deutch-Traubman, T.; Voet, H.; Kra, G.; Davidovich-Rikanati, R. Milking Performance and Plant Specialized Metabolites in the Milk of Goats Fed Silage from Willow (Salix Acmophylla) Irrigated with Saline Water. Livest. Sci. 2023, 270, 105205 DOI: 10.1016/j.livsci.2023.105205Google ScholarThere is no corresponding record for this reference.
- 129Tahir, M.; Ali, S.; Zhang, W.; Lv, B.; Qiu, W.; Wang, J. Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases. Biomedicines 2022, 10, 905, DOI: 10.3390/biomedicines10040905Google ScholarThere is no corresponding record for this reference.
- 130Jain, B.; Jain, N.; Jain, S.; Teja, P. K.; Chauthe, S.; Jain, A. Exploring Brucine Alkaloid: A Comprehensive Review on Pharmacology, Therapeutic Applications, Toxicity, Extraction and Purification Techniques. Phytomedicine Plus 2023, 3, 100490 DOI: 10.1016/j.phyplu.2023.100490Google ScholarThere is no corresponding record for this reference.
- 131Feng, L.; Gao, L. Anti-Hyperlipidemic, Antioxidant, Anti-Inflammatory and Antidiabetic Effect of Brucine Against Streptozotocin-Induced Diabetic Nephropathy in Rats. J. Biochem. Mol. Toxicol. 2025, 39, e70098 DOI: 10.1002/jbt.70098Google ScholarThere is no corresponding record for this reference.
- 132Noman, M.; Qazi, N.; Rehman, N.; Khan, A.-U. Pharmacological Investigation of Brucine Anti-Ulcer Potential. Front. Pharmacol. 2022, 13, 886433 DOI: 10.3389/fphar.2022.886433Google ScholarThere is no corresponding record for this reference.
- 133Seshadri, V. Brucine Promotes Apoptosis in Cervical Cancer Cells (ME-180) via Suppression of Inflammation and Cell Proliferation by Regulating PI3K/AKT/mTOR Signaling Pathway. Environ. Toxicol. 2021, 36, 1841– 1847, DOI: 10.1002/tox.23304Google ScholarThere is no corresponding record for this reference.
- 134Ji, H.; Liu, K.-H.; Lee, H.; Im, S.; Shim, H.; Son, M.; Lee, H. Corydaline Inhibits Multiple Cytochrome P450 and UDP-Glucuronosyltransferase Enzyme Activities in Human Liver Microsomes. Molecules 2011, 16, 6591– 6602, DOI: 10.3390/molecules16086591Google ScholarThere is no corresponding record for this reference.
- 135Guo, Y.; Sun, Q.; Wang, S.; Zhang, M.; Lei, Y.; Wu, J.; Wang, X.; Hu, W.; Meng, H.; Li, Z.; Xu, L.; Huang, F.; Qiu, Z. Corydalis Saxicola Bunting Total Alkaloids Improve NAFLD by Suppressing de Novo Lipogenesis through the AMPK-SREBP1 Axis. J. Ethnopharmacol. 2024, 319, 117162 DOI: 10.1016/j.jep.2023.117162Google ScholarThere is no corresponding record for this reference.
- 136Zhou, K.; Xu, S. Corydaline Alleviates Parkinson’s Disease by Regulating Autophagy and GSK-3β Phosphorylation. Psychopharmacology (Berl.) 2024, 241, 1027, DOI: 10.1007/s00213-024-06536-6Google ScholarThere is no corresponding record for this reference.
- 137Lu, Q.; Wang, H.; Zhang, X.; Yuan, T.; Wang, Y.; Feng, C.-J.; Li, Z.; Sun, S. Corydaline Attenuates Osteolysis in Rheumatoid Arthritis via Mitigating Reactive Oxygen Species Production and Suppressing Calcineurin-Nfatc1 Signaling. Int. Immunopharmacol. 2024, 142 Pt B, 113158 DOI: 10.1016/j.intimp.2024.113158Google ScholarThere is no corresponding record for this reference.
- 138Yang, N.; Shao, H.; Deng, J.; Yang, Y.; Tang, Z.; Wu, G.; Liu, Y. Dictamnine Ameliorates Chronic Itch in DNFB-Induced Atopic Dermatitis Mice via Inhibiting MrgprA3. Biochem. Pharmacol. 2023, 208, 115368 DOI: 10.1016/j.bcp.2022.115368Google ScholarThere is no corresponding record for this reference.
- 139Liu, R.; Zhang, Y.; Wang, Y.; Huang, Y.; Gao, J.; Tian, X.; Tian-You; Tao, Z. Anti-inflammatory Effect of Dictamnine on Allergic Rhinitis via Suppression of the LYN Kinase-mediated Molecular Signaling Pathway during Mast Cell Activation. Phytother. Res. 2023, 37, 4236– 4250, DOI: 10.1002/ptr.7904Google ScholarThere is no corresponding record for this reference.
- 140Yu, J.; Zhang, L.; Peng, J.; Ward, R.; Hao, P.; Wang, J.; Zhang, N.; Yang, Y.; Guo, X.; Xiang, C.; An, S.; Xu, T. Dictamnine, a Novel c-Met Inhibitor, Suppresses the Proliferation of Lung Cancer Cells by Downregulating the PI3K/AKT/mTOR and MAPK Signaling Pathways. Biochem. Pharmacol. 2020, 195, 114864 DOI: 10.1016/j.bcp.2021.114864Google ScholarThere is no corresponding record for this reference.
- 141Yang, W.; Liu, P.; Chen, Y.; Lv, Q.; Wang, Z.; Huang, W.; Jiang, H.; Zheng, Y.; Jiang, Y.; Sun, L. Dictamnine Inhibits the Adhesion to and Invasion of Uropathogenic Escherichia Coli (UPEC) to Urothelial Cells. Molecules 2022, 27, 272, DOI: 10.3390/molecules27010272Google ScholarThere is no corresponding record for this reference.
- 142Yin, X.; Liu, Z.; Wang, J. Tetrahydropalmatine Ameliorates Hepatic Steatosis in Nonalcoholic Fatty Liver Disease by Switching Lipid Metabolism via AMPK-SREBP-1c-Sirt1 Signaling Axis. Phytomedicine Int. J. Phytother. Phytopharm. 2023, 119, 155005 DOI: 10.1016/j.phymed.2023.155005Google ScholarThere is no corresponding record for this reference.
- 143Zhi, L.; Yang, S.; Chen, J.; Lu, Y.; Chen, J.; Qin, Z.; Tang, X.-M. Tetrahydropalmatine Has a Therapeutic Effect in a Lipopolysaccharide-Induced Disseminated Intravascular Coagulation Model. J. Int. Med. Res. 2020, 48, 0300060519889430 DOI: 10.1177/0300060519889430Google ScholarThere is no corresponding record for this reference.
- 144Zhang, X.; Wang, Y.; Zhang, K.; Sheng, H.; Wu, Y.; Wu, H.; Wang, Y.; Guan, J.; Meng, Q.; Li, H.; Li, Z.; Fan, G. Discovery of Tetrahydropalmatine and Protopine Regulate the Expression of Dopamine Receptor D2 to Alleviate Migraine from Yuanhu Zhitong Formula. Phytomed. Int. J. Phytother. Phytopharm. 2021, 91, 153702 DOI: 10.1016/j.phymed.2021.153702Google ScholarThere is no corresponding record for this reference.
- 145Liu, J.; Dai, R.; Damiescu, R.; Efferth, T.; Lee, D. Role of Levo-Tetrahydropalmatine and Its Metabolites for Management of Chronic Pain and Opioid Use Disorders. Phytomedicine Int. J. Phytother. Phytopharm. 2021, 90, 153594 DOI: 10.1016/j.phymed.2021.153594Google ScholarThere is no corresponding record for this reference.
- 146Jahan, S.; Mahmud, M.; Khan, Z.; Alam, A.; Khalil, A.; Rauf, A.; Tareq, A. M.; Nainu, F.; Tareq, S.; Emran, T.; Khan, M.; Khan, I.; Wilairatana, P.; Mubarak, M. Health Promoting Benefits of Pongamol: An Overview. Biomed. Pharmacother. Biomedecine Pharmacother. 2021, 142, 112109 DOI: 10.1016/j.biopha.2021.112109Google ScholarThere is no corresponding record for this reference.
- 147Wu, S.; Miao, J.; Zhu, S.; Wu, X.; Shi, J.; Zhou, J.; Xing, Y.; Hu, K.; Ren, J.; Yang, H. Pongamol Prevents Neurotoxicity via the Activation of MAPKs/Nrf2 Signaling Pathway in H2O2-Induced Neuronal PC12 Cells and Prolongs the Lifespan of Caenorhabditis Elegans. Mol. Neurobiol. 2024, 61, 8219, DOI: 10.1007/s12035-024-04110-xGoogle ScholarThere is no corresponding record for this reference.
- 148Dhakal, H.; Lee, S.; Kim, E.-N.; Choi, J.; Kim, M.-J.; Kang, J.; Choi, Y.-A.; Baek, M.; Lee, B.; Lee, H.-S.; Shin, T.; Jeong, G.; Kim, S.-H. Gomisin M2 Inhibits Mast Cell-Mediated Allergic Inflammation via Attenuation of FcεRI-Mediated Lyn and Fyn Activation and Intracellular Calcium Levels. Front. Pharmacol. 2019, 10, 869, DOI: 10.3389/fphar.2019.00869Google ScholarThere is no corresponding record for this reference.
- 149Chen, M.; Kilgore, N.; Lee, K.; Chen, D.-F. Rubrisandrins A and B, Lignans and Related Anti-HIV Compounds from Schisandra Rubriflora. J. Nat. Prod. 2006, 69 (12), 1697– 1701, DOI: 10.1021/np060239eGoogle ScholarThere is no corresponding record for this reference.
- 150Park, J.; Lee, T.-K.; Kim, D.; Sim, H.; Lee, J.; Kim, J.; Ahn, J.; Lee, C. H.; Kim, Y.-M.; Won, M.; Choi, S. Neuroprotective Effects of Salicin in a Gerbil Model of Transient Forebrain Ischemia by Attenuating Oxidative Stress and Activating PI3K/Akt/GSK3β Pathway. Antioxidants 2021, 10, 629, DOI: 10.3390/antiox10040629Google ScholarThere is no corresponding record for this reference.
- 151Wu, P.-Q.; Li, Y.; Ren, Y.-H.; Zhou, J.-S.; Liu, Q.-F.; Wu, Y.; Yu, J.-H.; Zhou, B.; Yue, J.-M. Anti-Inflammatory Salicin Derivatives from the Barks of Salix Tetrasperma. J. Agric. Food Chem. 2024, DOI: 10.1021/acs.jafc.4c01061Google ScholarThere is no corresponding record for this reference.
- 152Zhai, K.; Duan, H.; Khan, G.; Xu, H.; Han, F.-K.; Cao, W.; Gao, G.-Z.; Shan, L.-L.; Wei, Z. Salicin from Alangium Chinense Ameliorates Rheumatoid Arthritis by Modulating the Nrf2-HO-1-ROS Pathways. J. Agric. Food Chem. 2018, 66 (24), 6073– 6082, DOI: 10.1021/acs.jafc.8b02241Google ScholarThere is no corresponding record for this reference.
- 153Sutrapu, S.; Pal, R.; Khurana, N.; Vancha, H.; Mohd, S.; Chinnala, K. M.; Kumar, B.; Pilli, G. Diabetes Warriors from Heart Wood: Unveiling Dalbergin and Isoliquiritigenin from Dalbergia Latifolia as Potential Antidiabetic Agents in-Vitro and in-Vivo. Cell Biochem. Biophys. 2024, 82, 1309, DOI: 10.1007/s12013-024-01285-xGoogle ScholarThere is no corresponding record for this reference.
- 154Valojerdi, F.; Goliaei, B.; Parivar, K.; Nikoofar, A. Effect of a Neoflavonoid (Dalbergin) on T47D Breast Cancer Cell Line and mRNA Levels of P53, Bcl-2, and STAT3 Genes. Iran. Red Crescent Med. J. 2019, 21, e87175 DOI: 10.5812/IRCMJ.87175Google ScholarThere is no corresponding record for this reference.
- 155Wang, C.; Gong, B.; Wu, Y.; Bai, C.; Yang, M.; Zhao, X.; Wei, J. Pharmacokinetics and Molecular Docking of the Cardioprotective Flavonoids in Dalbergia Odorifera. J. Sep. Sci. 2024, DOI: 10.1002/jssc.202300614Google ScholarThere is no corresponding record for this reference.
- 156Shen, P.; Bai, Z.; Zhou, L.; Wang, N.-N.; Ni, Z.; Sun, D.; Huang, C.-S.; Hu, Y.; Xiao, C.-R.; Zhou, W.; Zhang, B.-L.; Gao, Y. A Scd1-Mediated Metabolic Alteration Participates in Liver Responses to Low-Dose Bavachin. J. Pharm. Anal. 2023, 13, 806– 816, DOI: 10.1016/j.jpha.2023.03.010Google ScholarThere is no corresponding record for this reference.
- 157Carrillo, J. A.; He, Y.; Li, Y.; Liu, J.; Erdman, R. A.; Sonstegard, T. S.; Song, J. Integrated Metabolomic and Transcriptome Analyses Reveal Finishing Forage Affects Metabolic Pathways Related to Beef Quality and Animal Welfare. Sci. Rep. 2016, 6 (1), 25948, DOI: 10.1038/srep25948Google ScholarThere is no corresponding record for this reference.
- 158Ahsin, M.; Pasha, I.; Liaquat, M.; Amir, M. Genetic and Agronomic Zinc Biofortification Modify Processing and Nutritional Quality of Common Wheat. Cereal Chem. 2023, 100 (1), 131– 141, DOI: 10.1002/cche.10604Google ScholarThere is no corresponding record for this reference.
- 159Bodnar, R. Conditioned Flavor Preferences in Animals: Merging Pharmacology. Brain Sites and Genetic Variance. Appetite 2018, 122, 17– 25, DOI: 10.1016/j.appet.2016.12.015Google ScholarThere is no corresponding record for this reference.
- 160Jin, H.; Fishman, Z.; Ye, M.; Wang, L.; Zuker, C. Top-Down Control of Sweet and Bitter Taste in the Mammalian Brain. Cell 2021, 184, 257– 271, DOI: 10.1016/j.cell.2020.12.014Google ScholarThere is no corresponding record for this reference.
- 161Provenza, F. After Ten Thousand Years of Domestication, Can Livestock Still Self-Medicate?. Planta Med. 2021, 87, 1237– 1237, DOI: 10.1055/s-0041-1736740Google ScholarThere is no corresponding record for this reference.
- 162Gradé, J.; Tabuti, J.; Van Damme, P. Four Footed Pharmacists: Indications of Self-Medicating Livestock in Karamoja. Uganda. Econ. Bot. 2009, 63, 29– 42, DOI: 10.1007/s12231-008-9058-zGoogle ScholarThere is no corresponding record for this reference.
- 163Villalba, J.; Provenza, F. Nutrient-Specific Preferences by Lambs Conditioned with Intraruminal Infusions of Starch, Casein, and Water. J. Anim. Sci. 1999, 77 (2), 378– 387, DOI: 10.2527/1999.772378xGoogle ScholarThere is no corresponding record for this reference.
- 164Iommelli, P.; Spina, A.; Vastolo, A.; Infascelli, L.; Lotito, D.; Musco, N.; Tudisco, R. Functional and Economic Role of Some Mediterranean Medicinal Plants in Dairy Ruminants’ Feeding: A Review of the Effects of Garlic, Oregano, and Rosemary. Animals 2025, 15, 657, DOI: 10.3390/ani15050657Google ScholarThere is no corresponding record for this reference.
- 165Vasta, V.; Luciano, G. The Effects of Dietary Consumption of Plants Secondary Compounds on Small Ruminants’ Products Quality. Small Rumin. Res. 2011, 101, 150– 159, DOI: 10.1016/j.smallrumres.2011.09.035Google ScholarThere is no corresponding record for this reference.
- 166Sun, R.; Jiang, X.; Reichelt, M.; Gershenzon, J.; Pandit, S.; Vassão, G. Tritrophic Metabolism of Plant Chemical Defenses and Its Effects on Herbivore and Predator Performance. eLife 2019, 8, e51029 DOI: 10.7554/eLife.51029Google ScholarThere is no corresponding record for this reference.
- 167Mithöfer, A.; Boland, W. Plant Defense against Herbivores: Chemical Aspects. Annu. Rev. Plant Biol. 2012, 63, 431– 450, DOI: 10.1146/annurev-arplant-042110-103854Google ScholarThere is no corresponding record for this reference.
- 168Beringue, A.; Queffelec, J.; Lann, C. L.; Sulmon, C. Sublethal Pesticide Exposure in Non-Target Terrestrial Ecosystems: From Known Effects on Individuals to Potential Consequences on Trophic Interactions and Network Functioning. Environ. Res. 2024, 260, 119620 DOI: 10.1016/j.envres.2024.119620Google ScholarThere is no corresponding record for this reference.
- 169Bertram, J.; Clough, T.; Sherlock, R.; Condron, L.; O’Callaghan, M.; Wells, N.; Ray, J. Hippuric Acid and Benzoic Acid Inhibition of Urine Derived N2O Emissions from Soil. Glob. Change Biol. 2009, 15, 2067, DOI: 10.1111/j.1365-2486.2008.01779.xGoogle ScholarThere is no corresponding record for this reference.
- 170Dijkstra, J.; Oenema, O.; Groenigen, J.; Spek, J.; Vuuren, A.; Bannink, A. Diet Effects on Urine Composition of Cattle and N2O Emissions. Anim. Int. J. Anim. Biosci. 2013, 7 (Suppl 2), 292– 302, DOI: 10.1017/S1751731113000578Google ScholarThere is no corresponding record for this reference.
- 171Zhang, W.; Sun, S.; Zhang, Y.; Zhang, Y.; Wang, J.; Liu, Z.; Yang, K. Benzoic Acid Supplementation Improves the Growth Performance, Nutrient Digestibility and Nitrogen Metabolism of Weaned Lambs. Front. Vet. Sci. 2024, 11, 1351394 DOI: 10.3389/fvets.2024.1351394Google ScholarThere is no corresponding record for this reference.
- 172Xu, Y.; Liu, L.; Zhu, J.; Zhu, S.; Ye, B.-Q.; Yang, J.-L.; Huang, J.-Y.; Huang, Z.-H.; You, Y.; Li, W.; He, J.; Xia, M.; Liu, Y. Alistipes Indistinctus-Derived Hippuric Acid Promotes Intestinal Urate Excretion to Alleviate Hyperuricemia. Cell Host Microbe 2024, 32, 366, DOI: 10.1016/j.chom.2024.02.001Google ScholarThere is no corresponding record for this reference.
- 173Yang, Y.; Huang, S.; Liao, Y.; Wu, X.; Zhang, C.; Wang, X.; Yang, Z. Hippuric Acid Alleviates Dextran Sulfate Sodium-Induced Colitis via Suppressing Inflammatory Activity and Modulating Gut Microbiota. Biochem. Biophys. Res. Commun. 2024, 710, 149879 DOI: 10.1016/j.bbrc.2024.149879Google ScholarThere is no corresponding record for this reference.
- 174De Mello, V.; Lankinen, M.; Lindström, J.; Puupponen-Pimiä, R.; Laaksonen, D.; Pihlajamäki, J.; Lehtonen, M.; Uusitupa, M.; Tuomilehto, J.; Kolehmainen, M.; Törrönen, R.; Hanhineva, K. Fasting Serum Hippuric Acid Is Elevated after Bilberry (Vaccinium Myrtillus) Consumption and Associates with Improvement of Fasting Glucose Levels and Insulin Secretion in Persons at High Risk of Developing Type 2 Diabetes. Mol. Nutr. Food Res. 2017, 61 (9), 1700019 DOI: 10.1002/mnfr.201700019Google ScholarThere is no corresponding record for this reference.
- 175Pallister, T.; Jackson, M. A.; Martin, T. C.; Zierer, J.; Jennings, A.; Mohney, R. P.; MacGregor, A.; Steves, C. J.; Cassidy, A.; Spector, T. D.; Menni, C. Hippurate as a Metabolomic Marker of Gut Microbiome Diversity: Modulation by Diet and Relationship to Metabolic Syndrome. Sci. Rep. 2017, 7 (1), 13670, DOI: 10.1038/s41598-017-13722-4Google ScholarThere is no corresponding record for this reference.
- 176Li, W.; Dong, H.; Niu, K.; Wang, H.-Y.; Cheng, W.; Song, H.; Ying, A.-K.; Zhai, X.; Li, K.; Yu, H.; Guo, D.-S.; Wang, Y. Analyzing Urinary Hippuric Acid as a Metabolic Health Biomarker through a Supramolecular Architecture. Talanta 2024, 278, 126480 DOI: 10.1016/j.Talanta.2024.126480Google ScholarThere is no corresponding record for this reference.
- 177Fu, H.; Xu, J.; Ai, X.; Dang, F.-T.; Tan, X.; Yu, H.-Y.; Feng, J.; Yang, W.; Haining; Tu, R.; Gupta, A.; Manandhar, L. K.; Bao, W.-M.; Tang, Y.-M. The Clostridium Metabolite P-Cresol Sulfate Relieves Inflammation of Primary Biliary Cholangitis by Regulating Kupffer Cells. Cells 2022, 11, 3782, DOI: 10.3390/cells11233782Google ScholarThere is no corresponding record for this reference.
- 178Grant, R.; Macowan, M.; Daunt, C.; Perdijk, O.; Marsland, B. Late Breaking Abstract - P-Cresol Sulfate Acts on Epithelial Cells to Reduce Allergic Airway Inflammation. Mol. Pathol. Funct. Genomics 2023, 62, PA1850, DOI: 10.1183/13993003.congress-2023.pa1850Google ScholarThere is no corresponding record for this reference.
- 179Zhou, Y.; Bi, Z.; Hamilton, M.; Zhang, L.; Su, R.; Sadowsky, M.; Roy, S.; Khoruts, A.; Chen, C. P-Cresol Sulfate Is a Sensitive Urinary Marker of Fecal Microbiota Transplantation and Antibiotics Treatments in Human Patients and Mouse Models. Int. J. Mol. Sci. 2023, 24, 14621, DOI: 10.3390/ijms241914621Google ScholarThere is no corresponding record for this reference.
- 180Mousavi, Y.; Adlercreutz, H. Enterolactone and Estradiol Inhibit Each Other’s Proliferative Effect on MCF-7 Breast Cancer Cells in Culture. J. Steroid Biochem. Mol. Biol. 1992, 41, 615– 619, DOI: 10.1016/0960-0760(92)90393-WGoogle ScholarThere is no corresponding record for this reference.
- 181Weiner, C.; Khan, S.; Leong, C.; Ranadive, S.; Campbell, S.; Howard, J.; Heffernan, K. Association of Enterolactone with Blood Pressure and Hypertension Risk in NHANES. PLoS One 2024, 19, e0302254 DOI: 10.1371/journal.pone.0302254Google ScholarThere is no corresponding record for this reference.
- 182Tuomisto, A.; No̷rskov, N.; Sirniö, P.; Väyrynen, J.; Mutt, S.; Klintrup, K.; Mäkelä, J.; Knudsen, K. B.; Mäkinen, M.; Herzig, K. Serum Enterolactone Concentrations Are Low in Colon but Not in Rectal Cancer Patients. Sci. Rep. 2019, 9, 11209, DOI: 10.1038/s41598-019-47622-6Google ScholarThere is no corresponding record for this reference.
- 183Pietinen, P.; Stumpf, K.; Männistö, S.; Kataja, V.; Uusitupa, M.; Adlercreutz, H. Serum Enterolactone and Risk of Breast Cancer: A Case-Control Study in Eastern Finland. Cancer Epidemiol. Biomark. Prev. 2001, 10 (4), 339– 344Google ScholarThere is no corresponding record for this reference.
- 184Zhang, X.; Veliky, C.; Birru, R.; Barinas-Mitchell, E.; Magnani, J.; Sekikawa, A. Potential Protective Effects of Equol (Soy Isoflavone Metabolite) on Coronary Heart Diseases─From Molecular Mechanisms to Studies in Humans. Nutrients 2021, 13, 3739, DOI: 10.3390/nu13113739Google ScholarThere is no corresponding record for this reference.
- 185Yoshikata, R.; Myint, K.; Ohta, H.; Ishigaki, Y. Effects of an Equol-Containing Supplement on Advanced Glycation End Products, Visceral Fat and Climacteric Symptoms in Postmenopausal Women: A Randomized Controlled Trial. PLoS One 2021, 16, e0257332 DOI: 10.1371/journal.pone.0257332Google ScholarThere is no corresponding record for this reference.
- 186Subedi, L.; Ji, E.; Shin, D.; Jin, J.-S.; Yeo, J.; Kim, S. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9, 207, DOI: 10.3390/nu9030207Google ScholarThere is no corresponding record for this reference.
- 187Mayo, B.; Vázquez, L.; Flórez, A. Equol: A Bacterial Metabolite from The Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients 2019, 11, 2231, DOI: 10.3390/nu11092231Google ScholarThere is no corresponding record for this reference.
- 188Puertas-Bartolomé, M.; Benito-Garzón, L.; Fung, S.; Kohn, J.; Vázquez-Lasa, B.; Román, S. Bioadhesive Functional Hydrogels: Controlled Release of Catechol Species with Antioxidant and Antiinflammatory Behavior. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 105, 110040 DOI: 10.1016/J.MSEC.2019.110040Google ScholarThere is no corresponding record for this reference.
- 189Lim, W.-C.; Kim, H.; Kim, Y.-J.; Jeon, B.; Kang, H.; Ko, H. Catechol Inhibits Epidermal Growth Factor-Induced Epithelial-to-Mesenchymal Transition and Stem Cell-like Properties in Hepatocellular Carcinoma Cells. Sci. Rep. 2020, 10, 7620, DOI: 10.1038/s41598-020-64603-2Google ScholarThere is no corresponding record for this reference.
- 190Chang, M.; Chang, H.-H.; Wang, T.-M.; Chan, C.; Lin, B.; Yeung, S.; Yeh, C.-Y.; Cheng, R.-H.; Jeng, J. Antiplatelet Effect of Catechol Is Related to Inhibition of Cyclooxygenase, Reactive Oxygen Species, ERK/P38 Signaling and Thromboxane A2 Production. PLoS One 2014, 9, e104310 DOI: 10.1371/journal.pone.0104310Google ScholarThere is no corresponding record for this reference.
- 191Jothi, R.; Sangavi, R.; Kumar, P.; Pandian, S.; Gowrishankar, S. Catechol Thwarts Virulent Dimorphism in Candida Albicans and Potentiates the Antifungal Efficacy of Azoles and Polyenes. Sci. Rep. 2021, 11, 21049, DOI: 10.1038/s41598-021-00485-2Google ScholarThere is no corresponding record for this reference.
- 192Bukowska, B.; Michałowicz, J.; Marczak, A. The Effect of Catechol on Human Peripheral Blood Mononuclear Cells (in Vitro Study). Environ. Toxicol. Pharmacol. 2015, 39 (1), 187– 193, DOI: 10.1016/j.etap.2014.11.017Google ScholarThere is no corresponding record for this reference.
- 193Mhawish, R.; Komarnytsky, S. Small Phenolic Metabolites at the Nexus of Nutrient Transport and Energy Metabolism. Molecules 2025, 30, 1026, DOI: 10.3390/molecules30051026Google ScholarThere is no corresponding record for this reference.
- 194Gaur, G.; Gänzle, M. Conversion of (Poly)Phenolic Compounds in Food Fermentations by Lactic Acid Bacteria: Novel Insights into Metabolic Pathways and Functional Metabolites. Curr. Res. Food Sci. 2023, 6, 100448 DOI: 10.1016/j.crfs.2023.100448Google ScholarThere is no corresponding record for this reference.
- 195Del Carmen Villegas-Aguilar, M.; Fernández-Ochoa, Á.; Cádiz-Gurrea, M.; Pimentel-Moral, S.; Lozano-Sánchez, J.; Arráez-Román, D.; Segura-Carretero, A. Pleiotropic Biological Effects of Dietary Phenolic Compounds and Their Metabolites on Energy Metabolism, Inflammation and Aging. Molecules 2020, 25, 596, DOI: 10.3390/molecules25030596Google ScholarThere is no corresponding record for this reference.
- 196Tonolo, F.; Folda, A.; Cesaro, L.; Scalcon, V.; Marin, O.; Ferro, S.; Bindoli, A.; Rigobello, M. Milk-Derived Bioactive Peptides Exhibit Antioxidant Activity through the Keap1-Nrf2 Signaling Pathway. J. Funct. Foods 2020, 64, 103696 DOI: 10.1016/j.jff.2019.103696Google ScholarThere is no corresponding record for this reference.
- 197Husted, A.; Trauelsen, M.; Rudenko, O.; Hjorth, S.; Schwartz, T. GPCR-Mediated Signaling of Metabolites. Cell Metab. 2017, 25 (4), 777– 796, DOI: 10.1016/j.cmet.2017.03.008Google ScholarThere is no corresponding record for this reference.
- 198Li, W.; Qiu, H.; Van Gestel, C.; Peijnenburg, W.; He, E. Trophic Transfer and Toxic Potency of Rare Earth Elements along a Terrestrial Plant-Herbivore Food Chain. Environ. Sci. Technol. 2024, 58, 5705, DOI: 10.1021/acs.est.3c09179Google ScholarThere is no corresponding record for this reference.
- 199Nfon, E.; Cousins, I.; Broman, D. Biomagnification of Organic Pollutants in Benthic and Pelagic Marine Food Chains from the Baltic Sea. Sci. Total Environ. 2008, 397 (1–3), 190– 204, DOI: 10.1016/j.scitotenv.2008.02.029Google ScholarThere is no corresponding record for this reference.
- 200Scalbert, A.; Williamson, G. Dietary Intake and Bioavailability of Polyphenols. J. Nutr. 2000, 130 (8S Suppl), 2073S– 2085, DOI: 10.1093/jn/130.8.2073SGoogle ScholarThere is no corresponding record for this reference.
- 201Huang, Q.; Braffett, B.; Simmens, S.; Young, H.; Ogden, C. Dietary Polyphenol Intake in US Adults and 10-Year Trends: 2007–2016. J. Acad. Nutr. Diet. 2020, 120, 1821, DOI: 10.1016/j.jand.2020.06.016Google ScholarThere is no corresponding record for this reference.
- 202Del Bo’, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; Zamora-Ros, R.; Liberona, N. H.; Andrés-Lacueva, C.; Riso, P. Systematic Review on Polyphenol Intake and Health Outcomes: Is There Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern?. Nutrients 2019, 11, 1355, DOI: 10.3390/nu11061355Google ScholarThere is no corresponding record for this reference.
- 203Komatsu, S.; Sakamoto, S.; Yusakul, G.; Putalun, W.; Miyamoto, T.; Tanaka, H.; Morimoto, S. A Single-Chain Variable Fragment Antibody against Anti-Leukemia Agent, Harringtonine as a Tool for Immunomodulation. Planta Med. 2016, 82, S1– S381, DOI: 10.1055/S-0036-1596806Google ScholarThere is no corresponding record for this reference.
- 204Takeda, S.; Yajima, N.; Kitazato, K.; Unemi, N. Antitumor Activities of Harringtonine and Homoharringtonine, Cephalotaxus Alkaloids Which Are Active Principles from Plant by Intraperitoneal and Oral Administration. J. Pharmacobiodyn. 1982, 5 (10), 841– 847, DOI: 10.1248/bpb1978.5.841Google ScholarThere is no corresponding record for this reference.
- 205Gupta, S.; Khajuria, V.; Wani, A.; Nalli, Y.; Bhagat, A.; Ali, A.; Ahmed, Z. Murrayanine Attenuates Lipopolysaccharide-induced Inflammation and Protects Mice from Sepsis-associated Organ Failure. Basic Clin. Pharmacol. Toxicol. 2019, 124, 351, DOI: 10.1111/bcpt.13032Google ScholarThere is no corresponding record for this reference.
- 206Mahapatra, D. K.; Chhajed, S.; Shivhare, R. Development of Murrayanine-Chalcone Hybrids: An Effort to Combine Two Privilege Scaffolds for Enhancing Hypoglycemic Activity. Int. J. Pharm. Chem. 2017, 4, 30– 34Google ScholarThere is no corresponding record for this reference.
- 207Mahapatra, D. K.; Dadure, K.; Shivhare, R. Edema Reducing Potentials of Some Emerging Schiff’s Bases of Murrayanine. MOJ. Bioorg. Org. Chem. 2018, 2, 171– 174, DOI: 10.15406/mojboc.2018.02.0076Google ScholarThere is no corresponding record for this reference.
- 208Zhou, H.; Li, H.; Cao, Y.-P.; Sang, X.; Liu, X. Murrayanine Exerts Antiproliferative Effects on Human Oral Cancer Cells through Inhibition of AKT/mTOR and Raf/MEK/ERK Signalling Pathways in Vitro and Inhibits Tumor Growth in Vivo. J. BUON Off. J. Balk. Union Oncol. 2019, 24 (6), 2423– 2428Google ScholarThere is no corresponding record for this reference.
- 209Cui, Z.; Wu, Y.; Wang, C.-Y.; Dai, X.; Nan, J.-X.; Liu, S.-H.; Lian, L.; Guo, J.; Jiang, Y.-C. Vincamine Ameliorates Hepatic Fibrosis via Inhibiting S100A4-mediated Farnesoid X Receptor Activation: Based on Liver Microenvironment and Enterohepatic Circulation Dependence. Br. J. Pharmacol. 2025, 182, 2447– 2465, DOI: 10.1111/bph.17471Google ScholarThere is no corresponding record for this reference.
- 210Du, T.; Yang, L.; Xu, X.; Shi, X.-F.; Xu, X.; Lu, J.; Lv, J.; Huang, X.; Chen, J.; Wang, H.; Ye, J.; Hu, L.; Shen, X. Vincamine as a GPR40 Agonist Improves Glucose Homeostasis in Type 2 Diabetic Mice. J. Endocrinol. 2019, 240 (2), 195– 214, DOI: 10.1530/JOE-18-0432Google ScholarThere is no corresponding record for this reference.
- 211Nandini, H.; Naik, P. Antidiabetic, Antihyperlipidemic and Antioxidant Effect of Vincamine, in Streptozotocin-induced Diabetic Rats. Eur. J. Pharmacol. 2019, 843, 233, DOI: 10.1016/j.ejphar.2018.11.034Google ScholarThere is no corresponding record for this reference.
- 212Sanz, F.; Solana-Manrique, C.; Paricio, N. Disease-Modifying Effects of Vincamine Supplementation in Drosophila and Human Cell Models of Parkinson’s Disease Based on DJ-1 Deficiency. ACS Chem. Neurosci. 2023, 14, 2294– 2301, DOI: 10.1021/acschemneuro.3c00026Google ScholarThere is no corresponding record for this reference.
- 213Renushe, A. P.; Banothu, A. K.; Bharani, K. K.; Mekala, L.; Kumar, M.; Neeradi, D.; Hanuman, D. D. V.; Gadige, A.; Khurana, A. Vincamine, an Active Constituent of Vinca Rosea Ameliorates Experimentally Induced Acute Lung Injury in Swiss Albino Mice through Modulation of Nrf-2/NF-κB Signaling Cascade. Int. Immunopharmacol. 2022, 108, 108773 DOI: 10.1016/j.intimp.2022.108773Google ScholarThere is no corresponding record for this reference.
- 214Qin, N.; Xu, G.; Wang, Y.; Zhan, X.; Gao, Y.; Wang, Z.; Fu, S.; Shi, W.; Hou, X.; Wang, C.; Li, R.-S.; Liu, Y.; Wang, J.; Zhao, H.; Xiao, X.; Bai, Z. Bavachin Enhances NLRP3 Inflammasome Activation Induced by ATP or Nigericin and Causes Idiosyncratic Hepatotoxicity. Front. Med. 2021, 15, 594– 607, DOI: 10.1007/s11684-020-0809-2Google ScholarThere is no corresponding record for this reference.
- 215Zhang, X.; Guo, Y.; Zhang, Z.; Wu, X.-Y.; Li, L.; Yang, Z.; Li, Z. Neuroprotective Effects of Bavachin against Neuroinflammation and Oxidative Stress-Induced Neuronal Damage via Activation of Sirt1/Nrf2 Pathway and Inhibition of NF-κB Pathway. J. Funct. Foods 2023, 107, 105655 DOI: 10.1016/j.jff.2023.105655Google ScholarThere is no corresponding record for this reference.
- 216Park, J.; Seo, E.; Jun, H. Bavachin Alleviates Diabetic Nephropathy in Db/Db Mice by Inhibition of Oxidative Stress and Improvement of Mitochondria Function. Biomed. Pharmacother. Biomedecine Pharmacother. 2023, 161, 114479 DOI: 10.1016/j.biopha.2023.114479Google ScholarThere is no corresponding record for this reference.
- 217Chakraborty, D.; Malik, S.; Mann, S.; Agnihotri, P.; Joshi, L.; Biswas, S. Chronic Disease Management via Modulation of Cellular Signaling by Phytoestrogen Bavachin. Mol. Biol. Rep. 2024, 51 (1), 921, DOI: 10.1007/s11033-024-09849-zGoogle ScholarThere is no corresponding record for this reference.
- 218Wang, C.; Hu, X.; Song, T.; Hu, F.; Du, L.; Yan, C.; Shen, T.; Li, N.; Yang, W.; Li, L.; Deng, N.; Jiang, X.; Wu, Y.; Ye, R. Magnolin Mitigates Skin Ageing Through the CXCL10/P38 Signalling Pathway. J. Cell. Mol. Med. 2025, 29, e70507 DOI: 10.1111/jcmm.70507Google ScholarThere is no corresponding record for this reference.
- 219Xu, K.; Gao, Y.; Yang, L.; Liu, Y.; Wang, C. Magnolin Exhibits Anti-Inflammatory Effects on Chondrocytes via the NF-κB Pathway for Attenuating Anterior Cruciate Ligament Transection-Induced Osteoarthritis. Connect. Tissue Res. 2021, 62, 475– 484, DOI: 10.1080/03008207.2020.1778679Google ScholarThere is no corresponding record for this reference.
- 220Patel, D. Therapeutic Effectiveness of Magnolin on Cancers and Other Human Complications. Pharmacol. Res. - Mod. Chin. Med. 2023, 6, 100203 DOI: 10.1016/j.prmcm.2022.100203Google ScholarThere is no corresponding record for this reference.
- 221Changan, S.; Tomar, M.; Prajapati, U.; Saurabh, V.; Hasan, M.; Sasi, M.; Maheshwari, C.; Singh, S.; Dhumal, S. S.; Radha; Thakur, M.; Punia, S.; Satankar, V.; Amarowicz, R.; Mekhemar, M. Custard Apple (Annona Squamosa L.) Leaves: Nutritional Composition, Phytochemical Profile, and Health-Promoting Biological Activities. Biomolecules 2021, 11, 614, DOI: 10.3390/biom11050614Google ScholarThere is no corresponding record for this reference.
- 222Toropova, A.; Razuvaeva, Y.; Olennikov, D. Dihydrosamidin: The Basic Khellactone Ester Derived from Phlojodicarpus Komarovii and Its Impact on Neurotrophic Factors, Energy and Antioxidant Metabolism after Rat Cerebral Ischemia-Reperfusion Injury. Nat. Prod. Res. 2024, 1– 6, DOI: 10.1080/14786419.2024.2433189Google ScholarThere is no corresponding record for this reference.
- 223Jia, N.; Shen, Z.; Zhao, S.; Wang, Y.; Pei, C.; Huang, D.; Wang, X.; Wu, Y.; Shi, S.; He, Y.; Wang, Z. Eleutheroside E from Pre-Treatment of Acanthopanax Senticosus (Rupr.etMaxim.) Harms Ameliorates High-Altitude-Induced Heart Injury by Regulating NLRP3 Inflammasome-Mediated Pyroptosis via NLRP3/Caspase-1 Pathway. Int. Immunopharmacol. 2023, 121, 110423 DOI: 10.1016/j.intimp.2023.110423Google ScholarThere is no corresponding record for this reference.
- 224Song, C.; Duan, F.; Ju, T.; Qin, Y.; Zeng, D.; Shan, S.; Shi, Y.; Zhang, Y.; Lu, W. Eleutheroside E Supplementation Prevents Radiation-Induced Cognitive Impairment and Activates PKA Signaling via Gut Microbiota. Commun. Biol. 2022, 5, 680, DOI: 10.1038/s42003-022-03602-7Google ScholarThere is no corresponding record for this reference.
- 225Zhou, T.; Zhou, Y.; Ge, D.; Xie, Y.; Wang, J.; Tang, L.; Dong, Q.; Sun, P. Decoding the Mechanism of Eleutheroside E in Treating Osteoporosis via Network Pharmacological Analysis and Molecular Docking of Osteoclast-Related Genes and Gut Microbiota. Front. Endocrinol. 2023, 14, 1257298 DOI: 10.3389/fendo.2023.1257298Google ScholarThere is no corresponding record for this reference.
- 226Jaiswal, J.; Srivastav, A. K.; Kushwaha, M.; Teotia, A.; Singh, R.; Mohan, A.; Makharia, G.; Kumar, A. Gut Microbial Metabolite 4-Ethylphenylsulfate Is Selectively Deleterious and Anticancer to Colon Cancer Cells. J. Med. Chem. 2025, 68, 10425, DOI: 10.1021/acs.jmedchem.5c00609Google ScholarThere is no corresponding record for this reference.
- 227Angelino, D.; Carregosa, D.; Domenech-Coca, C.; Savi, M.; Figueira, I.; Brindani, N.; Jang, S.; Lakshman, S.; Molokin, A.; Urban, J.; Davis, C.; Brito, M.; Kim, K.; Brighenti, F.; Curti, C.; Bladé, C.; Del Bas, J.; Stilli, D.; Solano-Aguilar, G.; Santos, C.; Del Rio, D.; Mena, P. 5-(Hydroxyphenyl)-γ-Valerolactone-Sulfate, a Key Microbial Metabolite of Flavan-3-Ols, Is Able to Reach the Brain: Evidence from Different in Silico, In Vitro and In Vivo Experimental Models. Nutrients 2019, 11, 2678, DOI: 10.3390/nu11112678Google ScholarThere is no corresponding record for this reference.
- 228Marcolin, E.; Chemello, C.; Piovan, A.; Barbierato, M.; Morazzoni, P.; Ragazzi, E.; Zusso, M. A Combination of 5-(3′,4′-Dihydroxyphenyl)-γ-Valerolactone and Curcumin Synergistically Reduces Neuroinflammation in Cortical Microglia by Targeting the NLRP3 Inflammasome and the NOX2/Nrf2 Signaling Pathway. Nutrients 2025, 17, 1316, DOI: 10.3390/nu17081316Google ScholarThere is no corresponding record for this reference.
- 229Della Vedova, L.; Husain, I.; Wang, Y.-H.; Kothapalli, H.; Gado, F.; Baron, G.; Manzi, S.; Morazzoni, P.; Aldini, G.; Khan, I. Pre-ADMET Studies of 5-(3′,4′-dihydroxyphenyl)-γ-valerolactone, the Bioactive Intestinal Metabolite of Proanthocyanidins. Arch. Pharm. (Weinheim) 2025, 358, e2400575 DOI: 10.1002/ardp.202400575Google ScholarThere is no corresponding record for this reference.
- 230Oliveira, M.; Ratti, B.; Daré, R.; Silva, S.; Truiti, M.; Ueda-Nakamura, T.; Auzély-Velty, R.; Nakamura, C. Dihydrocaffeic Acid Prevents UVB-Induced Oxidative Stress Leading to the Inhibition of Apoptosis and MMP-1 Expression via P38 Signaling Pathway. Oxid. Med. Cell. Longev. 2019, 2019, 2419096 DOI: 10.1155/2019/2419096Google ScholarThere is no corresponding record for this reference.
- 231Martini, S.; Conte, A.; Tagliazucchi, D. Antiproliferative Activity and Cell Metabolism of Hydroxycinnamic Acids in Human Colon Adenocarcinoma Cell Lines. J. Agric. Food Chem. 2019, 67 (14), 3919– 3931, DOI: 10.1021/acs.jafc.9b00522Google ScholarThere is no corresponding record for this reference.
- 232Stalmach, A.; Mullen, W.; Barron, D.; Uchida, K.; Yokota, T.; Cavin, C.; Steiling, H.; Williamson, G.; Crozier, A. Metabolite Profiling of Hydroxycinnamate Derivatives in Plasma and Urine after the Ingestion of Coffee by Humans: Identification of Biomarkers of Coffee Consumption. Drug Metab. Dispos. 2009, 37, 1749– 1758, DOI: 10.1124/dmd.109.028019Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 1 publications.
- Michela Contò, Marta Castrica, Simona Rinaldi, Sebastiana Failla. Natural Bioactive Compounds as Feed Additives: Strategies for Sustainable and Functional Livestock Production. Applied Sciences 2026, 16
(5)
, 2344. https://doi.org/10.3390/app16052344
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. Model of in-depth pathway analysis of phenolic compounds as they flow from plants to ruminant digestion to tissue (meat) and milk. The biochemical processes these compounds undergo in the rumen and small intestine, routes of absorption through enterocyte barriers, metabolism in the liver and kidney, and transfer pathways into muscle and milk. SGLT1 = sodium-glucose transport, CBG = corticosteroid-binding globulin, and LPH = lactase-phlorizin hydrolase, UGTs = UDP-glucuronosyltransferases, SULTs = sulfotransferases, and COMT = catechol O-methyltransferase. The figure was created with biorender.com.
Figure 2

Figure 2. Total phenolic concentration (TPC) in milk and/or meat from the included studies, organized by experimental groups as reported in the original studies. Values are group means; n denotes the number of biological replicates per group. ‘ref:’ shows the reference of the study, and country names are given in International Organization for Standardization (ISO) 3166-1 alpha-2 codes.
Figure 3

Figure 3. Concentrations of measured phenolics in milk and/or meat from the included studies, organized by experimental groups as reported in the original studies. Values are group means; ‘P’ indicates the number of phenolics quantified, ‘ref:’ shows the reference of the study; ‘n’ indicates the number of biological replicates per group. The study country is denoted by International Organization for Standardization (ISO) 3166–1 alpha-2 codes.
Figure 4
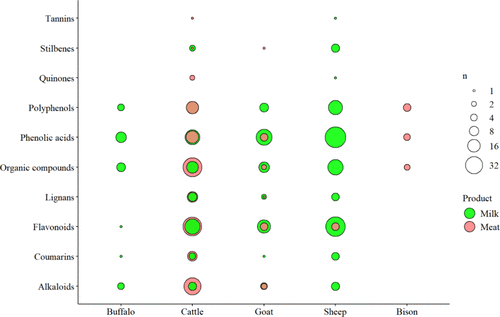
Figure 4. Phenolic diversity across animal-source foods identified by the current systematic review (n = 39 studies). Circle size is the number of phenolics reported within each class; circle color indicates the food product. Because flavonoids, phenolic acids, and other polyphenols vary widely, these classes are presented separately.
References
This article references 232 other publications.
- 1Stampa, E.; Schipmann-Schwarze, C.; Hamm, U. Consumer Perceptions, Preferences, and Behavior Regarding Pasture-Raised Livestock Products: A Review. Food Qual. Prefer. 2020, 82, 103872 DOI: 10.1016/j.foodqual.2020.103872There is no corresponding record for this reference.
- 2van Vliet, S.; Provenza, F. D.; Kronberg, S. L. Health-Promoting Phytonutrients Are Higher in Grass-Fed Meat and Milk. Front. Sustain. Food Syst. 2021, 4, 555426 DOI: 10.3389/fsufs.2020.555426There is no corresponding record for this reference.
- 3Villalba, J. J.; Ramsey, R. D.; Athanasiadou, S. Review: Herbivory and the Power of Phytochemical Diversity on Animal Health. animal 2024, 101287 DOI: 10.1016/j.animal.2024.101287There is no corresponding record for this reference.
- 4Evans, N.; Cloward, J.; Ward, R. E.; Van Wietmarschen, H. A.; Van Eekeren, N.; Kronberg, S. L.; Provenza, F. D.; Van Vliet, S. Pasture-Finishing of Cattle in Western U.S. Rangelands Improves Markers of Animal Metabolic Health and Nutritional Compounds in Beef. Sci. Rep. 2024, 14 (1), 20240, DOI: 10.1038/s41598-024-71073-3There is no corresponding record for this reference.
- 5Krusinski, L.; Maciel, I. C. F.; van Vliet, S.; Ahsin, M.; Lu, G.; Rowntree, J. E.; Fenton, J. I. Measuring the Phytochemical Richness of Meat: Effects of Grass/Grain Finishing Systems and grapeseed Extract Supplementation on the Fatty Acid and Phytochemical Content of Beef. Foods 2023, 12 (19), 3547, DOI: 10.3390/foods12193547There is no corresponding record for this reference.
- 6van Vliet, S.; Blair, A. D.; Hite, L. M.; Cloward, J.; Ward, R. E.; Kruse, C.; van Wietmarchsen, H. A.; van Eekeren, N.; Kronberg, S. L.; Provenza, F. D. Pasture-Finishing of Bison Improves Animal Metabolic Health and Potential Health-Promoting Compounds in Meat. J. Anim. Sci. Biotechnol. 2023, 14 (1), 49, DOI: 10.1186/s40104-023-00843-2There is no corresponding record for this reference.
- 7Krusinski, L.; Castanon, C.; Hellberg, R. S.; Maciel, I. C. F.; Ahsin, M.; van Vliet, S.; Rowntree, J. E.; Fenton, J. I. Highly Targeted Metabolomics Coupled With Gene Expression Analysis by RT–qPCR Improves Beef Separation Based on Grass, Grain, or Grape Supplemented Diet. Food Front. 2025, 6 (3), 1483– 1497, DOI: 10.1002/fft2.70022There is no corresponding record for this reference.
- 8Provenza, F. D.; Meuret, M.; Gregorini, P. Our Landscapes, Our Livestock, Ourselves: Restoring Broken Linkages among Plants, Herbivores, and Humans with Diets That Nourish and Satiate. Appetite 2015, 95, 500– 519, DOI: 10.1016/j.appet.2015.08.004There is no corresponding record for this reference.
- 9Villalba, J. J.; Beauchemin, K. A.; Gregorini, P.; MacAdam, J. W. Pasture Chemoscapes and Their Ecological Services. Transl. Anim. Sci. 2019, 3 (2), 829– 841, DOI: 10.1093/tas/txz003There is no corresponding record for this reference.
- 10Distel, R. A.; Arroquy, J. I.; Lagrange, S.; Villalba, J. J. Designing Diverse Agricultural Pastures for Improving Ruminant Production Systems. Front. Sustain. Food Syst. 2020, 4, 596869 DOI: 10.3389/fsufs.2020.596869There is no corresponding record for this reference.
- 11Pereira, F. C.; Gregorini, P. Applying Spatio-Chemical Analysis to Grassland Ecosystems for the Illustration of Chemoscapes and Creation of Healthscapes. Front. Sustain. Food Syst. 2022, 6, 927568 DOI: 10.3389/fsufs.2022.927568There is no corresponding record for this reference.
- 12Ahsin, M.; Poore, M. H.; Rogers, J.; Franzluebbers, A.; Young, S. N.; Kronberg, S. L.; Provenza, F. D.; Bain, J. R.; van Vliet, S. Soil and Pasture Health Underlie Improved Beef Nutrient Density Determined by Untargeted Metabolomics in Southern US Grass Finished Beef Systems. Npj Sci. Food 2025, 9 (1), 151, DOI: 10.1038/s41538-025-00471-2There is no corresponding record for this reference.
- 13Kumar, A.; P, N.; Kumar, M.; Jose, A.; Tomer, V.; Oz, E.; Proestos, C.; Zeng, M.; Elobeid, T.; K, S.; Oz, F. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules 2023, 28, 887, DOI: 10.3390/molecules28020887There is no corresponding record for this reference.
- 14Hou, Q.; Wang, Y.; Hu, J.; Zhang, J.; Zhang, C.; Song, W.; Wang, X.; Zheng, B.; Zhou, X. Simultaneous Determination of Phenothiazine Drugs and Their Metabolites Residues in Animal Derived Foods by High Performance Liquid Chromatography Tandem Mass Spectrometry. Food Control 2025, 167, 110799 DOI: 10.1016/j.foodcont.2024.110799There is no corresponding record for this reference.
- 15Delgadillo-Puga, C.; Cuchillo-Hilario, M.; León-Ortiz, L.; Ramírez-Rodríguez, A.; Cabiddu, A.; Navarro-Ocaña, A.; Morales-Romero, A. M.; Medina-Campos, O. N.; Pedraza-Chaverri, J. Goats’ Feeding Supplementation with Acacia Farnesiana Pods and Their Relationship with Milk Composition: Fatty Acids, Polyphenols, and Antioxidant Activity. Animals 2019, 9 (8), 515, DOI: 10.3390/ani9080515There is no corresponding record for this reference.
- 16Delgadillo-Puga, C.; Noriega, L. G.; Morales-Romero, A. M.; Nieto-Camacho, A.; Granados-Portillo, O.; Rodríguez-López, L. A.; Alemán, G.; Furuzawa-Carballeda, J.; Tovar, A. R.; Cisneros-Zevallos, L.; Torre-Villalvazo, I. Goat’s Milk Intake Prevents Obesity, Hepatic Steatosis and Insulin Resistance in Mice Fed A High-Fat Diet by Reducing Inflammatory Markers and Increasing Energy Expenditure and Mitochondrial Content in Skeletal Muscle. Int. J. Mol. Sci. 2020, 21 (15), 5530, DOI: 10.3390/ijms21155530There is no corresponding record for this reference.
- 17Sofi, F.; Buccioni, A.; Cesari, F.; Gori, A. M.; Minieri, S.; Mannini, L.; Casini, A.; Gensini, G. F.; Abbate, R.; Antongiovanni, M. Effects of a Dairy Product (Pecorino Cheese) Naturally Rich in Cis-9, Trans-11 Conjugated Linoleic Acid on Lipid, Inflammatory and Haemorheological Variables: A Dietary Intervention Study. Nutr. Metab. Cardiovasc. Dis. 2010, 20 (2), 117– 124, DOI: 10.1016/j.numecd.2009.03.004There is no corresponding record for this reference.
- 18Yang, M.; Sun, J.; Lu, Z.; Chen, G.; Guan, S.; Liu, X.; Jiang, B.; Ye, M.; Guo, D. Phytochemical Analysis of Traditional Chinese Medicine Using Liquid Chromatography Coupled with Mass Spectrometry. J. Chromatogr. A 2009, 1216 (11), 2045– 2062, DOI: 10.1016/j.chroma.2008.08.097There is no corresponding record for this reference.
- 19Wang, B.; Sun, Z.; Tu, Y.; Si, B.; Liu, Y.; Yang, L.; Luo, H.; Yu, Z. Untargeted Metabolomic Investigate Milk and Ruminal Fluid of Holstein Cows Supplemented with Perilla Frutescens Leaf. Food Res. Int. 2021, 140, 110017 DOI: 10.1016/j.foodres.2020.110017There is no corresponding record for this reference.
- 20Rousu, T.; Herttuainen, J.; Tolonen, A. Comparison of Triple Quadrupole, Hybrid Linear Ion Trap Triple Quadrupole, Time-of-Flight and LTQ-Orbitrap Mass Spectrometers in Drug Discovery Phase Metabolite Screening and Identification in Vitro – Amitriptyline and Verapamil as Model Compounds. Rapid Commun. Mass Spectrom. 2010, 24 (7), 939– 957, DOI: 10.1002/rcm.4465There is no corresponding record for this reference.
- 21Guironnet, A.; Wiest, L.; Vulliet, E. Advantages of MS/MS/MS (MRM3) vs Classic MRM Quantification for Complex Environmental Matrices: Analysis of Beta-Lactams in WWTP Sludge. Anal. Chim. Acta 2022, 1205, 339773 DOI: 10.1016/j.aca.2022.339773There is no corresponding record for this reference.
- 22Kasperkiewicz, A.; Pawliszyn, J. Multiresidue Pesticide Quantitation in Multiple Fruit Matrices via Automated Coated Blade Spray and Liquid Chromatography Coupled to Triple Quadrupole Mass Spectrometry. Food Chem. 2021, 339, 127815 DOI: 10.1016/j.foodchem.2020.127815There is no corresponding record for this reference.
- 23Sleiman, H.; De Oliveira, J. M.; De Freitas, G. B. L. Isoflavones Alter Male and Female Fertility in Different Development Windows. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 140, 111448 DOI: 10.1016/j.biopha.2021.111448There is no corresponding record for this reference.
- 24Li, D.; Dang, D.; Xu, S.; Tian, Y.-M.; Wu, D.; Su, Y. Soy Isoflavones Supplementation Improves Reproductive Performance and Serum Antioxidant Status of Sows and the Growth Performance of Their Offspring. J. Anim. Physiol. Anim. Nutr. 2022, 106, 1268, DOI: 10.1111/jpn.13667There is no corresponding record for this reference.
- 25Min, B.; Barry, T.; Attwood, G.; McNabb, W. The Effect of Condensed Tannins on the Nutrition and Health of Ruminants Fed Fresh Temperate Forages: A Review. Anim. Feed Sci. Technol. 2003, 106, 3– 19, DOI: 10.1016/S0377-8401(03)00041-5There is no corresponding record for this reference.
- 26Kelln, B.; Penner, G.; Acharya, S.; McAllister, T.; Lardner, H. Impact of Condensed Tannin-Containing Legumes on Ruminal Fermentation, Nutrition, and Performance in Ruminants: A Review. Can. J. Anim. Sci. 2021, 101, 210– 223, DOI: 10.1139/cjas-2020-0096There is no corresponding record for this reference.
- 27Hossain, Md. S.; Wazed, M. A.; Asha, S.; Amin, M. R.; Shimul, I. M. Dietary Phytochemicals in Health and Disease: Mechanisms, Clinical Evidence, and Applications─A Comprehensive Review. Food Sci. Nutr. 2025, 13, e70101 DOI: 10.1002/fsn3.70101There is no corresponding record for this reference.
- 28Tava, A.; Biazzi, E.; Ronga, D.; Pecetti, L.; Avato, P. Biologically Active Compounds from Forage Plants. Phytochem. Rev. 2022, 21 (2), 471– 501, DOI: 10.1007/s11101-021-09779-9There is no corresponding record for this reference.
- 29Krusinski, L.; Sergin, S.; Jambunathan, V.; Rowntree, J. E.; Fenton, J. I. Attention to the Details: How Variations in U.S. Grass-Fed Cattle-Feed Supplementation and Finishing Date Influence Human Health. Front. Sustain. Food Syst. 2022, 6, 851494 DOI: 10.3389/fsufs.2022.851494There is no corresponding record for this reference.
- 30Provenza, F. D.; Villalba, J. J.; Dziba, L. E.; Atwood, S. B.; Banner, R. E. Linking Herbivore Experience, Varied Diets, and Plant Biochemical Diversity. Adv. Res. Nutr. Sheep Goats Spec. Ref. Pasture Rangel. Use 2003, 49 (3), 257– 274, DOI: 10.1016/S0921-4488(03)00143-3There is no corresponding record for this reference.
- 31Soder, K. J.; Gregorini, P.; Scaglia, G.; Rook, A. J. Dietary Selection by Domestic Grazing Ruminants in Temperate Pastures: Current State of Knowledge, Methodologies, and Future Direction. Rangel. Ecol. Manag. 2009, 62 (5), 389– 398, DOI: 10.2111/08-068.1There is no corresponding record for this reference.
- 32Provenza, F. D. What Does It Mean to Be Locally Adapted and Who Cares Anyway?. J. Anim. Sci. 2008, 86 (suppl_14), E271– E284, DOI: 10.2527/jas.2007-0468There is no corresponding record for this reference.
- 33Reynaud, A.; Fraisse, D.; Cornu, A.; Farruggia, A.; Pujos-Guillot, E.; Besle, J.-M.; Martin, B.; Lamaison, J.-L.; Paquet, D.; Doreau, M.; Graulet, B. Variation in Content and Composition of Phenolic Compounds in Permanent Pastures According to Botanical Variation. J. Agric. Food Chem. 2010, 58 (9), 5485– 5494, DOI: 10.1021/jf1000293There is no corresponding record for this reference.
- 34Fleming, A.; Wescombe, P.; Gregorini, P. Review: A Vade-Mecum for Ruminant Grazing and Health. animal 2025, 101548 DOI: 10.1016/j.animal.2025.101548There is no corresponding record for this reference.
- 35Beck, M. R.; Gregorini, P. How Dietary Diversity Enhances Hedonic and Eudaimonic Well-Being in Grazing Ruminants. Front. Vet. Sci. 2020, 7, 191, DOI: 10.3389/fvets.2020.00191There is no corresponding record for this reference.
- 36Kearns, M.; Ponnampalam, E. N.; Jacquier, J.-C.; Grasso, S.; Boland, T. M.; Sheridan, H.; Monahan, F. J. Can Botanically-Diverse Pastures Positively Impact the Nutritional and Antioxidant Composition of Ruminant Meat? – Invited Review. Meat Sci. 2023, 197, 109055 DOI: 10.1016/j.meatsci.2022.109055There is no corresponding record for this reference.
- 37Schreiner, B.; Ribeiro, G.; Lardner, H.; Penner, G. PSVIII-17 A Comparison of a Monoculture Barley Crop to a Barley-Based Complex Mixture on Forage Yield, Quality, Dry Matter Intake, Enteric Methane Emissions and Growth Performance of Pregnant Yearling Heifers. J. Anim. Sci. 2024, 102, 582, DOI: 10.1093/jas/skae234.655There is no corresponding record for this reference.
- 38Muñoz, C.; Letelier, P.; Ungerfeld, E.; Morales, J.; Hube, S.; Pérez-Prieto, L. Effects of Pregrazing Herbage Mass in Late Spring on Enteric Methane Emissions, Dry Matter Intake, and Milk Production of Dairy Cows. J. Dairy Sci. 2016, 99 (10), 7945– 7955, DOI: 10.3168/jds.2016-10919There is no corresponding record for this reference.
- 39Segueni, K.; Chouikh, A.; Laouini, S. E.; Bouafia, A.; Tlili, M. L.; Laib, I.; Boudebia, O.; Khelef, Y.; Abdullah, M.; Abdullah, J. A. A.; Emran, T. B. Evaluation of Dermal Wound Healing Potential: Phytochemical Characterization, Anti-Inflammatory, Antioxidant, and Antimicrobial Activities of Euphorbia Guyoniana Boiss. & Reut. Latex. Chem. Biodivers. 2024, 22, e202402284 DOI: 10.1002/cbdv.202402284There is no corresponding record for this reference.
- 40Riaz, A.; Ali, S.; Summer, M.; Noor, S.; Nazakat, L.; Aqsa; Sharjeel, M. Exploring the Underlying Pharmacological, Immunomodulatory, and Anti-Inflammatory Mechanisms of Phytochemicals against Wounds: A Molecular Insight. Inflammopharmacology 2024, 32, 2695, DOI: 10.1007/s10787-024-01545-5There is no corresponding record for this reference.
- 41Redan, B. W.; Buhman, K. K.; Novotny, J. A.; Ferruzzi, M. G. Altered Transport and Metabolism of Phenolic Compounds in Obesity and Diabetes: Implications for Functional Food Development and Assessment. Adv. Nutr. 2016, 7 (6), 1090– 1104, DOI: 10.3945/an.116.013029There is no corresponding record for this reference.
- 42Monfalouti, H. E.; Kartah, B. E.; Monfalouti, H. E.; Kartah, B. E. Enhancing Polyphenol Bioavailability through Nanotechnology: Current Trends and Challenges; IntechOpen, 2024.There is no corresponding record for this reference.
- 43Kasapidou, E.; Iliadis, I.-V.; Mitlianga, P.; Papatzimos, G.; Karatzia, M.-A.; Papadopoulos, V.; Amanatidis, M.; Tortoka, V.; Tsiftsi, E.; Aggou, A.; Basdagianni, Z. Variations in Composition, Antioxidant Profile, and Physical Traits of Goat Milk within the Semi-Intensive Production System in Mountainous Areas during the Post-Weaning to End-of-Lactation Period. Animals 2023, 13 (22), 3505, DOI: 10.3390/ani13223505There is no corresponding record for this reference.
- 44Lee, K.; Kim, Y.; Lee, H.; Lee, C. Y. Cocoa Has More Phenolic Phytochemicals and a Higher Antioxidant Capacity than Teas and Red Wine. J. Agric. Food Chem. 2003, 51 (25), 7292– 7295, DOI: 10.1021/jf0344385There is no corresponding record for this reference.
- 45Martín, M.; Goya, L.; Ramos, S. Protective Effects of Tea, Red Wine and Cocoa in Diabetes. Evidences from Human Studies. Food Chem. Toxicol. 2017, 109 (Pt 1), 302– 314, DOI: 10.1016/j.fct.2017.09.015There is no corresponding record for this reference.
- 46Ohishi, T.; Fukutomi, R.; Shoji, Y.; Goto, S.; Isemura, M. The Beneficial Effects of Principal Polyphenols from Green Tea, Coffee, Wine, and Curry on Obesity. Molecules 2021, 26, 453, DOI: 10.3390/molecules26020453There is no corresponding record for this reference.
- 47van Vliet, S.; Bain, J. R.; Muehlbauer, M. J.; Provenza, F. D.; Kronberg, S. L.; Pieper, C. F.; Huffman, K. M. A Metabolomics Comparison of Plant-Based Meat and Grass-Fed Meat Indicates Large Nutritional Differences despite Comparable Nutrition Facts Panels. Sci. Rep. 2021, 11 (1), 13828, DOI: 10.1038/s41598-021-93100-3There is no corresponding record for this reference.
- 48Gregorini, P.; Provenza, F.; Kronberg, S. Is Grassfed Meat and Dairy Better for Human and Environmental Health?. Front. Nutr. 2019, 6, 26, DOI: 10.3389/fnut.2019.00026There is no corresponding record for this reference.
- 49Agulló, V.; Favari, C.; Pilla, N.; Bresciani, L.; Tomás-Barberán, F. A.; Crozier, A.; Del Rio, D.; Mena, P. Using Targeted Metabolomics to Unravel Phenolic Metabolites of Plant Origin in Animal Milk. Int. J. Mol. Sci. 2024, 25 (8), 4536, DOI: 10.3390/ijms25084536There is no corresponding record for this reference.
- 50Page, M. J.; McKenzie, J. E.; Bossuyt, P. M.; Boutron, I.; Hoffmann, T. C.; Mulrow, C. D.; Shamseer, L.; Tetzlaff, J. M.; Akl, E. A.; Brennan, S. E.; Chou, R.; Glanville, J.; Grimshaw, J. M.; Hróbjartsson, A.; Lalu, M. M.; Li, T.; Loder, E. W.; Mayo-Wilson, E.; McDonald, S.; McGuinness, L. A.; Stewart, L. A.; Thomas, J.; Tricco, A. C.; Welch, V. A.; Whiting, P.; Moher, D. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. PLOS Med. 2021, 18 (3), e1003583 DOI: 10.1371/journal.pmed.1003583There is no corresponding record for this reference.
- 51Newman, M.; Gough, D. Systematic Reviews in Educational Research: Methodology, Perspectives and Application. Syst. Rev. Educ. Res. Methodol. Perspect. Appl. 2020, 3– 22, DOI: 10.1007/978-3-658-27602-7_1There is no corresponding record for this reference.
- 52Hooijmans, C. R.; Rovers, M. M.; De Vries, R. B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M. W. SYRCLE’s Risk of Bias Tool for Animal Studies. BMC Med. Res. Methodol. 2014, 14, 43, DOI: 10.1186/1471-2288-14-43There is no corresponding record for this reference.
- 53Zhang, W.; Jiang, Y.; Shang, Z.; Zhang, N.; Tao, G.; Zhang, T.; Hu, K.; Li, Y.; Shi, X.; Zhang, Y.; Yang, J.; Ma, B.; Yang, K. The Methodological Quality of Animal Studies: A Cross-Sectional Study Based on the SYRCLE’s Risk of Bias Tool. bioRxiv 2019, DOI: DOI: 10.1101/701110 .There is no corresponding record for this reference.
- 54Bennato, F.; Ianni, A.; Oliva, E.; Franceschini, N.; Grotta, L.; Sergi, M.; Martino, G. Characterization of Phenolic Profile in Milk Obtained by Ewes Fed Grape Pomace: Reflection on Antioxidant and Anti-Inflammatory Status. Biomolecules 2023, 13 (7), 1026, DOI: 10.3390/biom13071026There is no corresponding record for this reference.
- 55Lee, J.-E.; Jayakody, J. T.; Kim, J.-I.; Jeong, J.-W.; Choi, K.-M.; Kim, T.-S.; Seo, C.; Azimi, I.; Hyun, J.; Ryu, B. The Influence of Solvent Choice on the Extraction of Bioactive Compounds from Asteraceae: A Comparative Review. Foods 2024, 13 (19), 3151, DOI: 10.3390/foods13193151There is no corresponding record for this reference.
- 56Rocchetti, G.; Becchi, P. P.; Salis, L.; Lucini, L.; Cabiddu, A. Impact of Pasture-Based Diets on the Untargeted Metabolomics Profile of Sarda Sheep Milk. Foods 2023, 12 (1), 143, DOI: 10.3390/foods12010143There is no corresponding record for this reference.
- 57Hernandez, M. S.; Kim, Y. H. B.; Woerner, D. R.; Brooks, J. C.; Legako, J. F. Untargeted Metabolomics Reveals Divergent Metabolomes between Three Plant-Based Meat Alternatives and Two Lean Levels of Ground Beef. ACS Food Sci. Technol. 2025, 5 (4), 1632– 1644, DOI: 10.1021/acsfoodscitech.5c00041There is no corresponding record for this reference.
- 58Mokrani, A.; Madani, K. Effect of Solvent, Time and Temperature on the Extraction of Phenolic Compounds and Antioxidant Capacity of Peach (Prunus Persica L.). Fruit. Sep. Purif. Technol. 2016, 162, 68– 76, DOI: 10.1016/j.seppur.2016.01.043There is no corresponding record for this reference.
- 59Di-Grigoli, A.; Bonanno, A.; Rabie Ashkezary, M.; Laddomada, B.; Alabiso, M.; Vitale, F.; Mazza, F.; Maniaci, G.; Ruisi, P.; Di Miceli, G. Meat Production from Dairy Breed Lambs Due to Slaughter Age and Feeding Plan Based on Wheat Bran. Animals 2019, 9 (11), 892, DOI: 10.3390/ani9110892There is no corresponding record for this reference.
- 60Bennato, F.; Ianni, A.; Florio, M.; Grotta, L.; Pomilio, F.; Saletti, M. A.; Martino, G. Nutritional Properties of Milk from Dairy Ewes Fed with a Diet Containing Grape Pomace. Foods 2022, 11 (13), 1878, DOI: 10.3390/foods11131878There is no corresponding record for this reference.
- 61Chávez-Servín, J. L.; Andrade-Montemayor, H. M.; Velázquez Vázquez, C.; Aguilera Barreyro, A.; García-Gasca, T.; Ferríz Martínez, R. A.; Olvera Ramírez, A. M.; De La Torre-Carbot, K. Effects of Feeding System, Heat Treatment and Season on Phenolic Compounds and Antioxidant Capacity in Goat Milk, Whey and Cheese. Small Rumin. Res. 2018, 160, 54– 58, DOI: 10.1016/j.smallrumres.2018.01.011There is no corresponding record for this reference.
- 62Di-Trana, A.; Bonanno, A.; Cecchini, S.; Giorgio, D.; Di Grigoli, A.; Claps, S. Effects of Sulla Forage (Sulla Coronarium L.) on the Oxidative Status and Milk Polyphenol Content in Goats. J. Dairy Sci. 2015, 98 (1), 37– 46, DOI: 10.3168/jds.2014-8414There is no corresponding record for this reference.
- 63Ianni, A.; Innosa, D.; Oliva, E.; Bennato, F.; Grotta, L.; Saletti, M. A.; Pomilio, F.; Sergi, M.; Martino, G. Effect of Olive Leaves Feeding on Phenolic Composition and Lipolytic Volatile Profile in Goat Milk. J. Dairy Sci. 2021, 104 (8), 8835– 8845, DOI: 10.3168/jds.2021-20211There is no corresponding record for this reference.
- 64Leparmarai, P. T.; Sinz, S.; Kunz, C.; Liesegang, A.; Ortmann, S.; Kreuzer, M.; Marquardt, S. Transfer of Total Phenols from a grapeseed-Supplemented Diet to Dairy Sheep and Goat Milk, and Effects on Performance and Milk Quality1. J. Anim. Sci. 2019, 97 (4), 1840– 1851, DOI: 10.1093/jas/skz046There is no corresponding record for this reference.
- 65Leparmarai, P. T.; Kunz, C.; Mwangi, D. M.; Gluecks, I.; Kreuzer, M.; Marquardt, S. Camels and Cattle Respond Differently in Milk Phenol Excretion and Milk Fatty Acid Profile to Free Ranging Conditions in East-African Rangelands. Sci. Afr. 2021, 13, e00896 DOI: 10.1016/j.sciaf.2021.e00896There is no corresponding record for this reference.
- 66Sik, B.; Buzás, H.; Kapcsándi, V.; Lakatos, E.; Daróczi, F.; Székelyhidi, R. Antioxidant and Polyphenol Content of Different Milk and Dairy Products. J. King Saud Univ. - Sci. 2023, 35 (7), 102839 DOI: 10.1016/j.jksus.2023.102839There is no corresponding record for this reference.
- 67Ainsworth, E. A.; Gillespie, K. M. Estimation of Total Phenolic Content and Other Oxidation Substrates in Plant Tissues Using Folin–Ciocalteu Reagent. Nat. Protoc. 2007, 2 (4), 875– 877, DOI: 10.1038/nprot.2007.102There is no corresponding record for this reference.
- 68Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L. D.; Hidalgo, F. J.; Miyashita, K.; Camp, J. van; Alasalvar, C.; Ismail, A. B.; Elmore, S.; Birch, G. G.; Charalampopoulos, D.; Astley, S. B.; Pegg, R.; Zhou, P.; Finglas, P. Antioxidant Activity, Total phenolics and Flavonoids Contents: Should We Ban in Vitro Screening Methods?. Food Chem. 2018, 264, 471– 475, DOI: 10.1016/j.foodchem.2018.04.012There is no corresponding record for this reference.
- 69Chociej, P.; Foss, K.; Jabłońska, M.; Ustarbowska, M.; Sawicki, T. The Profile and Content of Polyphenolic Compounds and Antioxidant and Anti-Glycation Properties of Root Extracts of Selected Medicinal Herbs. Plant Foods Hum. Nutr. 2024, 79 (2), 468– 473, DOI: 10.1007/s11130-024-01180-zThere is no corresponding record for this reference.
- 70Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 370438, DOI: 10.3389/fnut.2018.00087There is no corresponding record for this reference.
- 71Donato, P.; Cacciola, F.; Tranchida, P. Q.; Dugo, P.; Mondello, L. Mass Spectrometry Detection in Comprehensive Liquid Chromatography: Basic Concepts, Instrumental Aspects. Applications and Trends. Mass Spectrom. Rev. 2012, 31 (5), 523– 559, DOI: 10.1002/mas.20353There is no corresponding record for this reference.
- 72Lamuela-Raventós, R. M.; Vallverdú-Queralt, A.; Jáuregui, O.; Martínez-Huélamo, M.; Quifer-Rada, P. Chapter 14 - Improved Characterization of Polyphenols Using Liquid Chromatography. In Polyphenols in Plants; Watson, R. R., Ed.; Academic Press: San Diego, 2014; pp. 261– 292.There is no corresponding record for this reference.
- 73Andersen, C.; Nielsen, T. S.; Purup, S.; Kristensen, T.; Eriksen, J.; So̷egaard, K.; So̷rensen, J.; Fretté, X. C. Phyto-Oestrogens in Herbage and Milk from Cows Grazing White Clover, Red Clover, Lucerne or Chicory-Rich Pastures. Animal 2009, 3 (8), 1189– 1195, DOI: 10.1017/S1751731109004613There is no corresponding record for this reference.
- 74Hoikkala, A.; Mustonen, E.; Saastamoinen, I.; Jokela, T.; Taponen, J.; Saloniemi, H.; Wähälä, K. High Levels of Equol in Organic Skimmed Finnish Cow Milk. Mol. Nutr. Food Res. 2007, 51 (7), 782– 786, DOI: 10.1002/mnfr.200600222There is no corresponding record for this reference.
- 75Mustonen, E. A.; Tuori, M.; Saastamoinen, I.; Taponen, J.; Wähälä, K.; Saloniemi, H.; Vanhatalo, A. Equol in Milk of Dairy Cows Is Derived from Forage Legumes Such as Red Clover. Br. J. Nutr. 2009, 102 (11), 1552, DOI: 10.1017/S0007114509990857There is no corresponding record for this reference.
- 76Třináctý, J.; Křížová, L.; Schulzová, V.; Hajšlová, J.; Hanuš, O. The Effect of Feeding Soybean-Derived Phytoestogens on Their Concentration in Plasma and Milk of Lactating Dairy Cows. Arch. Anim. Nutr. 2009, 63 (3), 219– 229, DOI: 10.1080/17450390902859739There is no corresponding record for this reference.
- 77Alonso, A.; Marsal, S.; Julià, A. Analytical Methods in Untargeted Metabolomics: State of the Art in 2015. Front. Bioeng. Biotechnol. 2015, 3, 23, DOI: 10.3389/fbioe.2015.00023There is no corresponding record for this reference.
- 78Zarrouk, E.; El Balkhi, S.; Saint-Marcoux, F. Critical Evaluation of High-Resolution and Low-Resolution Mass Spectrometry for Biomonitoring of Human Environmental Exposure to Pesticides. Environ. Technol. Innov. 2024, 36, 103834 DOI: 10.1016/j.eti.2024.103834There is no corresponding record for this reference.
- 79Zheng, F.; Zhao, X.; Zeng, Z.; Wang, L.; Lv, W.; Wang, Q.; Xu, G. Development of a Plasma Pseudotargeted Metabolomics Method Based on Ultra-High-Performance Liquid Chromatography–Mass Spectrometry. Nat. Protoc. 2020, 15, 2519– 2537, DOI: 10.1038/s41596-020-0341-5There is no corresponding record for this reference.
- 80Wang, W.; Sun, B.; Hu, P.; Zhou, M.; Sun, S.; Du, P.; Ru, Y.; Suvorov, A.; Li, Y.; Liu, Y.; Wang, S. Comparison of Differential Flavor Metabolites in Meat of Lubei White Goat, Jining Gray Goat and Boer Goat. Metabolites 2019, 9 (9), 176, DOI: 10.3390/metabo9090176There is no corresponding record for this reference.
- 81Ottaviani, J. I.; Fong, R. Y.; Borges, G.; Schroeter, H.; Crozier, A. Use of LC-MS for the Quantitative Analysis of (Poly)Phenol Metabolites Does Not Necessarily Yield Accurate Results: Implications for Assessing Existing Data and Conducting Future Research. Free Radic. Biol. Med. 2018, 124, 97– 103, DOI: 10.1016/j.freeradbiomed.2018.05.092There is no corresponding record for this reference.
- 82Bloszies, C. S.; Fiehn, O. Using Untargeted Metabolomics for Detecting Exposome Compounds. Mech. Toxicol. Metab. Disrupt. Environ. Dis. 2018, 8, 87– 92, DOI: 10.1016/j.cotox.2018.03.002There is no corresponding record for this reference.
- 83Fang, X.; Liu, Y.; Xiao, J.; Ma, C.; Huang, Y. GC–MS and LC-MS/MS Metabolomics Revealed Dynamic Changes of Volatile and Non-Volatile Compounds during Withering Process of Black Tea. Food Chem. 2023, 410, 135396 DOI: 10.1016/j.foodchem.2023.135396There is no corresponding record for this reference.
- 84Scano, P.; Carta, P.; Ibba, I.; Manis, C.; Caboni, P. An Untargeted Metabolomic Comparison of Milk Composition from Sheep Kept Under Different Grazing Systems. Dairy 2020, 1 (1), 30– 41, DOI: 10.3390/dairy1010004There is no corresponding record for this reference.
- 85Jiao, L.; Guo, Y.; Chen, J.; Zhao, X.; Dong, D. Detecting Volatile Compounds in Food by Open-Path Fourier-Transform Infrared Spectroscopy. Food Res. Int. 2019, 119, 968– 973, DOI: 10.1016/j.foodres.2018.11.042There is no corresponding record for this reference.
- 86Foroutan, A.; Guo, A. C.; Vazquez-Fresno, R.; Lipfert, M.; Zhang, L.; Zheng, J.; Badran, H.; Budinski, Z.; Mandal, R.; Ametaj, B. N.; Wishart, D. S. Chemical Composition of Commercial Cow’s Milk. J. Agric. Food Chem. 2019, 67 (17), 4897– 4914, DOI: 10.1021/acs.jafc.9b00204There is no corresponding record for this reference.
- 87Chiriac, E. R.; Chiţescu, C. L.; Geană, E.-I.; Gird, C. E.; Socoteanu, R. P.; Boscencu, R. Advanced Analytical Approaches for the Analysis of Polyphenols in Plants Matrices─A Review. Separations 2021, 8 (5), 65, DOI: 10.3390/separations8050065There is no corresponding record for this reference.
- 88Teng, H.; Chen, L. Polyphenols and Bioavailability: An Update. Crit. Rev. Food Sci. Nutr. 2019, 59 (13), 2040– 2051, DOI: 10.1080/10408398.2018.1437023There is no corresponding record for this reference.
- 89Chesson, A.; Provan, G. J.; Russell, W. R.; Scobbie, L.; Richardson, A. J.; Stewart, C. Hydroxycinnamic Acids in the Digestive Tract of Livestock and Humans. J. Sci. Food Agric. 1999, 79 (3), 373– 378, DOI: 10.1002/(SICI)1097-0010(19990301)79:3<373::AID-JSFA257>3.3.CO;2-YThere is no corresponding record for this reference.
- 90Romero, P.; Huang, R.; Jiménez, E.; Palma-Hidalgo, J.; Ungerfeld, E.; Popova, M.; Morgavi, D.; Belanche, A.; Yáñez-Ruiz, D. Evaluating the Effect of Phenolic Compounds as Hydrogen Acceptors When Ruminal Methanogenesis Is Inhibited in Vitro - Part 2. Dairy Goats. Anim. Int. J. Anim. Biosci. 2023, 17 (5), 100789 DOI: 10.1016/j.animal.2023.100789There is no corresponding record for this reference.
- 91Krumholz, L.; Bryant, M. Eubacterium Oxidoreducens Sp. Nov. Requiring H2 or Formate to Degrade Gallate, Pyrogallol, Phloroglucinol and Quercetin. Arch. Microbiol. 1986, 144, 8– 14, DOI: 10.1007/BF00454948There is no corresponding record for this reference.
- 92Martin, A. K. The Origin of Urinary Aromatic Compounds Excreted by Ruminants 2. The Metabolism of Phenolic Cinnamic Acids to Benzoic Acid. Br. J. Nutr. 1982, 47 (1), 155– 164, DOI: 10.1079/BJN19820020There is no corresponding record for this reference.
- 93Batterham, T.; Shutt, D.; Hart, N.; Braden, A.; Tweeddale, H. Metabolism of Intraruminally Administered [4–14C]Formononetic and [4–14C]Biochanin a in Sheep. Crop Pasture Sci. 1971, 22, 131– 138, DOI: 10.1071/AR9710131There is no corresponding record for this reference.
- 94Trnková, A.; Šancová, K.; Zapletalová, M.; Kašparovská, J.; Dadáková, K.; Křížová, L.; Lochman, J.; Hadrová, S.; Ihnatová, I.; Kašparovský, T. Determination of in Vitro Isoflavone Degradation in Rumen Fluid. J. Dairy Sci. 2018, 101 (6), 5134– 5144, DOI: 10.3168/jds.2017-13610There is no corresponding record for this reference.
- 95Guo, Y.; Weber, W. J.; Yao, D.; Caixeta, L.; Zimmerman, N. P.; Thompson, J.; Block, E.; Rehberger, T. G.; Crooker, B. A.; Chen, C. Forming 4-Methylcatechol as the Dominant Bioavailable Metabolite of Intraruminal Rutin Inhibits P-Cresol Production in Dairy Cows. Metabolites 2022, 12 (1), 16, DOI: 10.3390/metabo12010016There is no corresponding record for this reference.
- 96Berger, L. M.; Blank, R.; Zorn, F.; Wein, S.; Metges, C. C.; Wolffram, S. Ruminal Degradation of Quercetin and Its Influence on Fermentation in Ruminants. J. Dairy Sci. 2015, 98 (8), 5688– 5698, DOI: 10.3168/jds.2015-9633There is no corresponding record for this reference.
- 97Terrill, T. H.; Waghorn, G. C.; Woolley, D. J.; Mcnabb, W. C.; Barry, T. N. Assay and Digestion of 14C-Labelled Condensed Tannins in the Gastrointestinal Tract of Sheep. Br. J. Nutr. 1994, 72 (3), 467– 477, DOI: 10.1079/BJN19940048There is no corresponding record for this reference.
- 98Gladine, C.; Rock, E.; Morand, C.; Bauchart, D.; Durand, D. Bioavailability and Antioxidant Capacity of Plant Extracts Rich in Polyphenols, given as a Single Acute Dose, in Sheep Made Highly Susceptible to Lipoperoxidation. Br. J. Nutr. 2007, 98 (4), 691– 701, DOI: 10.1017/S0007114507742666There is no corresponding record for this reference.
- 99Gessner, D. K.; Ringseis, R.; Eder, K. Potential of Plant Polyphenols to Combat Oxidative Stress and Inflammatory Processes in Farm Animals. J. Anim. Physiol. Anim. Nutr. 2017, 101 (4), 605– 628, DOI: 10.1111/jpn.12579There is no corresponding record for this reference.
- 100Day, A.; Cañada, F.; Díaz, J.; Kroon, P.; Mclauchlan, R.; Faulds, C.; Plumb, G.; Morgan, M.; Williamson, G. Dietary Flavonoid and Isoflavone Glycosides Are Hydrolysed by the Lactase Site of Lactase Phlorizin Hydrolase. FEBS Lett. 2000, 468, 166– 170, DOI: 10.1016/S0014-5793(00)01211-4There is no corresponding record for this reference.
- 101Wang, X.; Qi, Y.; Zheng, H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants 2022, 11 (6), 1212, DOI: 10.3390/antiox11061212There is no corresponding record for this reference.
- 102Zeb, A. Metabolism of Phenolic Antioxidants. In Phenolic Antioxidants in Foods: Chemistry, Biochemistry and Analysis; Zeb, A., Ed.; Springer International Publishing: Cham, 2021; pp. 333– 383.There is no corresponding record for this reference.
- 103Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D. J.; Preston, T.; Kroon, P. A.; Botting, N. P.; Kay, C. D. Human Metabolism and Elimination of the Anthocyanin, Cyanidin-3-Glucoside: A 13C-Tracer Study123. Am. J. Clin. Nutr. 2013, 97 (5), 995– 1003, DOI: 10.3945/ajcn.112.049247There is no corresponding record for this reference.
- 104Renouf, M.; Guy, P. A.; Marmet, C.; Fraering, A.-L.; Longet, K.; Moulin, J.; Enslen, M.; Barron, D.; Dionisi, F.; Cavin, C.; Williamson, G.; Steiling, H. Measurement of Caffeic and Ferulic Acid Equivalents in Plasma after Coffee Consumption: Small Intestine and Colon Are Key Sites for Coffee Metabolism. Mol. Nutr. Food Res. 2010, 54 (6), 760– 766, DOI: 10.1002/mnfr.200900056There is no corresponding record for this reference.
- 105Yang, G.; Ge, S.; Singh, R.; Basu, S.; Shatzer, K.; Zen, M.; Liu, J.; Tu, Y.; Zhang, C.; Wei, J.; Shi, J.; Zhu, L.; Liu, Z.; Wang, Y.; Gao, S.; Hu, M. Glucuronidation: Driving Factors and Their Impact on Glucuronide Disposition. Drug Metab. Rev. 2017, 49 (2), 105– 138, DOI: 10.1080/03602532.2017.1293682There is no corresponding record for this reference.
- 106Zhang, H.; Tsao, R. Dietary Polyphenols, Oxidative Stress and Antioxidant and Anti-Inflammatory Effects. Food Microbiol. Funct. Foods Nutr. 2016, 8, 33– 42, DOI: 10.1016/j.cofs.2016.02.002There is no corresponding record for this reference.
- 107Tak, Y.; Kaur, M.; Gautam, C.; Kumar, R.; Tilgam, J.; Natta, S. Phenolic Biosynthesis and Metabolic Pathways to Alleviate Stresses in Plants. In Plant phenolics in Abiotic Stress Management; Lone, R.; Khan, S.; Mohammed Al-Sadi, A., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 63– 87.There is no corresponding record for this reference.
- 108Kuhnle, G. G. C.; Dell’Aquila, C.; Aspinall, S. M.; Runswick, S. A.; Mulligan, A. A.; Bingham, S. A. Phytoestrogen Content of Foods of Animal Origin: Dairy Products, Eggs, Meat, Fish, and Seafood. J. Agric. Food Chem. 2008, 56 (21), 10099– 10104, DOI: 10.1021/jf801344xThere is no corresponding record for this reference.
- 109Steinshamn, H.; Purup, S.; Thuen, E.; Hansen-Mo̷ller, J. Effects of Clover-Grass Silages and Concentrate Supplementation on the Content of Phytoestrogens in Dairy Cow Milk. J. Dairy Sci. 2008, 91 (7), 2715– 2725, DOI: 10.3168/jds.2007-0857There is no corresponding record for this reference.
- 110Höjer, A.; Adler, S.; Purup, S.; Hansen-Mo̷ller, J.; Martinsson, K.; Steinshamn, H.; Gustavsson, A.-M. Effects of Feeding Dairy Cows Different Legume-Grass Silages on Milk Phytoestrogen Concentration. J. Dairy Sci. 2012, 95 (8), 4526– 4540, DOI: 10.3168/jds.2011-5226There is no corresponding record for this reference.
- 111Caradus, J.; Voisey, C.; Cousin, G.; Kaur, R.; Woodfield, D.; Blanc, A.; Roldan, M. The Hunt for the “Holy Grail”: Condensed Tannins in the Perennial Forage Legume White Clover (Trifolium Repens L.). Grass Forage Sci. 2022, 77, 111, DOI: 10.1111/gfs.12567There is no corresponding record for this reference.
- 112Seeno, E.; MacAdam, J.; Melathopoulos, A.; Filley, S.; Ates, S. Management of Perennial Forbs Sown with or without Self-regenerating Annual Clovers for Forage and Nectar Sources in a Low-input Dryland Production System. Grass Forage Sci. 2023, 78, 462, DOI: 10.1111/gfs.12640There is no corresponding record for this reference.
- 113Besle, J. M.; Viala, D.; Martin, B.; Pradel, P.; Meunier, B.; Berdagué, J. L.; Fraisse, D.; Lamaison, J. L.; Coulon, J. B. Ultraviolet-Absorbing Compounds in Milk Are Related to Forage Polyphenols. J. Dairy Sci. 2010, 93 (7), 2846– 2856, DOI: 10.3168/jds.2009-2939There is no corresponding record for this reference.
- 114Adler, S. A.; Purup, S.; Hansen-Mo̷ller, J.; Thuen, E.; Steinshamn, H. Phytoestrogens and Their Metabolites in Bulk-Tank Milk: Effects of Farm Management and Season. PLoS One 2015, 10 (5), e0127187 DOI: 10.1371/journal.pone.0127187There is no corresponding record for this reference.
- 115Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24 (13), 2452, DOI: 10.3390/molecules24132452There is no corresponding record for this reference.
- 116Er, M.; Keles, G. Buckwheat Conservation as Hay or Silage: Agronomic Evaluation, Nutritive Value, Conservation Quality, and Intake by Lactating Dairy Goats. Trop. Anim. Health Prod. 2021, 53, 215, DOI: 10.1007/s11250-021-02655-wThere is no corresponding record for this reference.
- 117Rufino-Moya, P.; Bertolín, J.; Blanco, M.; Lobón, S.; Joy, M. Fatty Acid Profile, Secondary Compounds and Antioxidant Activities in the Fresh Forage, Hay and Silage of Sainfoin (Onobrychis Viciifolia) and Sulla (Hedysarum Coronarium). J. Sci. Food Agric. 2022, 102, 4736– 4743, DOI: 10.1002/jsfa.11834There is no corresponding record for this reference.
- 118Lv, J.; Jin, S.; Zhang, Y.; Zhou, Y.; Li, M.; Feng, N. Equol: A Metabolite of Gut Microbiota with Potential Antitumor Effects. Gut Pathog. 2024, 16 (1), 35, DOI: 10.1186/s13099-024-00625-9There is no corresponding record for this reference.
- 119Róin, N. R.; Poulsen, N. A.; No̷rskov, N. P.; Purup, S.; Larsen, L. B. Seasonal Variation in Contents of Phytoestrogens in Danish Dairy Milk Lines of Different Farm Management Systems. Int. Dairy J. 2023, 144, 105694 DOI: 10.1016/j.idairyj.2023.105694There is no corresponding record for this reference.
- 120Hilario, M. C.; Puga, C. D.; Ocaña, A. N.; Romo, F. P.-G. Antioxidant Activity, Bioactive Polyphenols in Mexican Goats’ Milk Cheeses on Summer Grazing. J. Dairy Res. 2010, 77 (1), 20– 26, DOI: 10.1017/S0022029909990161There is no corresponding record for this reference.
- 121Manis, C.; Scano, P.; Nudda, A.; Carta, S.; Pulina, G.; Caboni, P. LC-QTOF/MS Untargeted Metabolomics of Sheep Milk under Cocoa Husks Enriched Diet. Dairy 2021, 2 (1), 112– 121, DOI: 10.3390/dairy2010011There is no corresponding record for this reference.
- 122Zhang, F.; Wang, Y.; Liu, B.; Gong, P.; Shi, C.; Zhu, L.; Zhao, J.; Yao, W.; Liu, Q.; Luo, J. Widely Targeted Metabolomic Analysis Revealed the Diversity in Milk from Goats, Sheep, Cows, and Buffaloes and Its Association with Flavor Profiles. Foods 2024, 13 (9), 1365, DOI: 10.3390/foods13091365There is no corresponding record for this reference.
- 123No̷rskov, N. P.; Givens, I.; Purup, S.; Stergiadis, S. Concentrations of Phytoestrogens in Conventional, Organic and Free-Range Retail Milk in England. Food Chem. 2019, 295, 1– 9, DOI: 10.1016/j.foodchem.2019.05.081There is no corresponding record for this reference.
- 124Rocchetti, G.; Ghilardelli, F.; Bonini, P.; Lucini, L.; Masoero, F.; Gallo, A. Changes of Milk Metabolomic Profiles Resulting from a Mycotoxins-Contaminated Corn Silage Intake by Dairy Cows. Metabolites 2021, 11 (8), 475, DOI: 10.3390/metabo11080475There is no corresponding record for this reference.
- 125Zhang, F.; Zhao, Y.; Wang, H.; Nan, X.; Wang, Y.; Guo, Y.; Xiong, B. Alterations in the Milk Metabolome of Dairy Cows Supplemented with Different Levels of Calcium Propionate in Early Lactation. Metabolites 2022, 12 (8), 699, DOI: 10.3390/metabo12080699There is no corresponding record for this reference.
- 126Rocchetti, G.; Ghilardelli, F.; Mosconi, M.; Masoero, F.; Gallo, A. Occurrence of Polyphenols, Isoflavonoids, and Their Metabolites in Milk Samples from Different Cow Feeding Regimens. Dairy 2022, 3 (2), 314– 325, DOI: 10.3390/dairy3020024There is no corresponding record for this reference.
- 127Casula, M.; Scano, P.; Manis, C.; Tolle, G.; Nudda, A.; Carta, S.; Pulina, G.; Caboni, P. UHPLC-QTOF/MS Untargeted Lipidomics and Caffeine Carry-Over in Milk of Goats under Spent Coffee Ground Enriched Diet. Appl. Sci. 2023, 13 (4), 2477, DOI: 10.3390/app13042477There is no corresponding record for this reference.
- 128Landau, S. Y.; Glasser, T. A.; Zachut, M.; Klein, J. D.; Deutch-Traubman, T.; Voet, H.; Kra, G.; Davidovich-Rikanati, R. Milking Performance and Plant Specialized Metabolites in the Milk of Goats Fed Silage from Willow (Salix Acmophylla) Irrigated with Saline Water. Livest. Sci. 2023, 270, 105205 DOI: 10.1016/j.livsci.2023.105205There is no corresponding record for this reference.
- 129Tahir, M.; Ali, S.; Zhang, W.; Lv, B.; Qiu, W.; Wang, J. Aloperine: A Potent Modulator of Crucial Biological Mechanisms in Multiple Diseases. Biomedicines 2022, 10, 905, DOI: 10.3390/biomedicines10040905There is no corresponding record for this reference.
- 130Jain, B.; Jain, N.; Jain, S.; Teja, P. K.; Chauthe, S.; Jain, A. Exploring Brucine Alkaloid: A Comprehensive Review on Pharmacology, Therapeutic Applications, Toxicity, Extraction and Purification Techniques. Phytomedicine Plus 2023, 3, 100490 DOI: 10.1016/j.phyplu.2023.100490There is no corresponding record for this reference.
- 131Feng, L.; Gao, L. Anti-Hyperlipidemic, Antioxidant, Anti-Inflammatory and Antidiabetic Effect of Brucine Against Streptozotocin-Induced Diabetic Nephropathy in Rats. J. Biochem. Mol. Toxicol. 2025, 39, e70098 DOI: 10.1002/jbt.70098There is no corresponding record for this reference.
- 132Noman, M.; Qazi, N.; Rehman, N.; Khan, A.-U. Pharmacological Investigation of Brucine Anti-Ulcer Potential. Front. Pharmacol. 2022, 13, 886433 DOI: 10.3389/fphar.2022.886433There is no corresponding record for this reference.
- 133Seshadri, V. Brucine Promotes Apoptosis in Cervical Cancer Cells (ME-180) via Suppression of Inflammation and Cell Proliferation by Regulating PI3K/AKT/mTOR Signaling Pathway. Environ. Toxicol. 2021, 36, 1841– 1847, DOI: 10.1002/tox.23304There is no corresponding record for this reference.
- 134Ji, H.; Liu, K.-H.; Lee, H.; Im, S.; Shim, H.; Son, M.; Lee, H. Corydaline Inhibits Multiple Cytochrome P450 and UDP-Glucuronosyltransferase Enzyme Activities in Human Liver Microsomes. Molecules 2011, 16, 6591– 6602, DOI: 10.3390/molecules16086591There is no corresponding record for this reference.
- 135Guo, Y.; Sun, Q.; Wang, S.; Zhang, M.; Lei, Y.; Wu, J.; Wang, X.; Hu, W.; Meng, H.; Li, Z.; Xu, L.; Huang, F.; Qiu, Z. Corydalis Saxicola Bunting Total Alkaloids Improve NAFLD by Suppressing de Novo Lipogenesis through the AMPK-SREBP1 Axis. J. Ethnopharmacol. 2024, 319, 117162 DOI: 10.1016/j.jep.2023.117162There is no corresponding record for this reference.
- 136Zhou, K.; Xu, S. Corydaline Alleviates Parkinson’s Disease by Regulating Autophagy and GSK-3β Phosphorylation. Psychopharmacology (Berl.) 2024, 241, 1027, DOI: 10.1007/s00213-024-06536-6There is no corresponding record for this reference.
- 137Lu, Q.; Wang, H.; Zhang, X.; Yuan, T.; Wang, Y.; Feng, C.-J.; Li, Z.; Sun, S. Corydaline Attenuates Osteolysis in Rheumatoid Arthritis via Mitigating Reactive Oxygen Species Production and Suppressing Calcineurin-Nfatc1 Signaling. Int. Immunopharmacol. 2024, 142 Pt B, 113158 DOI: 10.1016/j.intimp.2024.113158There is no corresponding record for this reference.
- 138Yang, N.; Shao, H.; Deng, J.; Yang, Y.; Tang, Z.; Wu, G.; Liu, Y. Dictamnine Ameliorates Chronic Itch in DNFB-Induced Atopic Dermatitis Mice via Inhibiting MrgprA3. Biochem. Pharmacol. 2023, 208, 115368 DOI: 10.1016/j.bcp.2022.115368There is no corresponding record for this reference.
- 139Liu, R.; Zhang, Y.; Wang, Y.; Huang, Y.; Gao, J.; Tian, X.; Tian-You; Tao, Z. Anti-inflammatory Effect of Dictamnine on Allergic Rhinitis via Suppression of the LYN Kinase-mediated Molecular Signaling Pathway during Mast Cell Activation. Phytother. Res. 2023, 37, 4236– 4250, DOI: 10.1002/ptr.7904There is no corresponding record for this reference.
- 140Yu, J.; Zhang, L.; Peng, J.; Ward, R.; Hao, P.; Wang, J.; Zhang, N.; Yang, Y.; Guo, X.; Xiang, C.; An, S.; Xu, T. Dictamnine, a Novel c-Met Inhibitor, Suppresses the Proliferation of Lung Cancer Cells by Downregulating the PI3K/AKT/mTOR and MAPK Signaling Pathways. Biochem. Pharmacol. 2020, 195, 114864 DOI: 10.1016/j.bcp.2021.114864There is no corresponding record for this reference.
- 141Yang, W.; Liu, P.; Chen, Y.; Lv, Q.; Wang, Z.; Huang, W.; Jiang, H.; Zheng, Y.; Jiang, Y.; Sun, L. Dictamnine Inhibits the Adhesion to and Invasion of Uropathogenic Escherichia Coli (UPEC) to Urothelial Cells. Molecules 2022, 27, 272, DOI: 10.3390/molecules27010272There is no corresponding record for this reference.
- 142Yin, X.; Liu, Z.; Wang, J. Tetrahydropalmatine Ameliorates Hepatic Steatosis in Nonalcoholic Fatty Liver Disease by Switching Lipid Metabolism via AMPK-SREBP-1c-Sirt1 Signaling Axis. Phytomedicine Int. J. Phytother. Phytopharm. 2023, 119, 155005 DOI: 10.1016/j.phymed.2023.155005There is no corresponding record for this reference.
- 143Zhi, L.; Yang, S.; Chen, J.; Lu, Y.; Chen, J.; Qin, Z.; Tang, X.-M. Tetrahydropalmatine Has a Therapeutic Effect in a Lipopolysaccharide-Induced Disseminated Intravascular Coagulation Model. J. Int. Med. Res. 2020, 48, 0300060519889430 DOI: 10.1177/0300060519889430There is no corresponding record for this reference.
- 144Zhang, X.; Wang, Y.; Zhang, K.; Sheng, H.; Wu, Y.; Wu, H.; Wang, Y.; Guan, J.; Meng, Q.; Li, H.; Li, Z.; Fan, G. Discovery of Tetrahydropalmatine and Protopine Regulate the Expression of Dopamine Receptor D2 to Alleviate Migraine from Yuanhu Zhitong Formula. Phytomed. Int. J. Phytother. Phytopharm. 2021, 91, 153702 DOI: 10.1016/j.phymed.2021.153702There is no corresponding record for this reference.
- 145Liu, J.; Dai, R.; Damiescu, R.; Efferth, T.; Lee, D. Role of Levo-Tetrahydropalmatine and Its Metabolites for Management of Chronic Pain and Opioid Use Disorders. Phytomedicine Int. J. Phytother. Phytopharm. 2021, 90, 153594 DOI: 10.1016/j.phymed.2021.153594There is no corresponding record for this reference.
- 146Jahan, S.; Mahmud, M.; Khan, Z.; Alam, A.; Khalil, A.; Rauf, A.; Tareq, A. M.; Nainu, F.; Tareq, S.; Emran, T.; Khan, M.; Khan, I.; Wilairatana, P.; Mubarak, M. Health Promoting Benefits of Pongamol: An Overview. Biomed. Pharmacother. Biomedecine Pharmacother. 2021, 142, 112109 DOI: 10.1016/j.biopha.2021.112109There is no corresponding record for this reference.
- 147Wu, S.; Miao, J.; Zhu, S.; Wu, X.; Shi, J.; Zhou, J.; Xing, Y.; Hu, K.; Ren, J.; Yang, H. Pongamol Prevents Neurotoxicity via the Activation of MAPKs/Nrf2 Signaling Pathway in H2O2-Induced Neuronal PC12 Cells and Prolongs the Lifespan of Caenorhabditis Elegans. Mol. Neurobiol. 2024, 61, 8219, DOI: 10.1007/s12035-024-04110-xThere is no corresponding record for this reference.
- 148Dhakal, H.; Lee, S.; Kim, E.-N.; Choi, J.; Kim, M.-J.; Kang, J.; Choi, Y.-A.; Baek, M.; Lee, B.; Lee, H.-S.; Shin, T.; Jeong, G.; Kim, S.-H. Gomisin M2 Inhibits Mast Cell-Mediated Allergic Inflammation via Attenuation of FcεRI-Mediated Lyn and Fyn Activation and Intracellular Calcium Levels. Front. Pharmacol. 2019, 10, 869, DOI: 10.3389/fphar.2019.00869There is no corresponding record for this reference.
- 149Chen, M.; Kilgore, N.; Lee, K.; Chen, D.-F. Rubrisandrins A and B, Lignans and Related Anti-HIV Compounds from Schisandra Rubriflora. J. Nat. Prod. 2006, 69 (12), 1697– 1701, DOI: 10.1021/np060239eThere is no corresponding record for this reference.
- 150Park, J.; Lee, T.-K.; Kim, D.; Sim, H.; Lee, J.; Kim, J.; Ahn, J.; Lee, C. H.; Kim, Y.-M.; Won, M.; Choi, S. Neuroprotective Effects of Salicin in a Gerbil Model of Transient Forebrain Ischemia by Attenuating Oxidative Stress and Activating PI3K/Akt/GSK3β Pathway. Antioxidants 2021, 10, 629, DOI: 10.3390/antiox10040629There is no corresponding record for this reference.
- 151Wu, P.-Q.; Li, Y.; Ren, Y.-H.; Zhou, J.-S.; Liu, Q.-F.; Wu, Y.; Yu, J.-H.; Zhou, B.; Yue, J.-M. Anti-Inflammatory Salicin Derivatives from the Barks of Salix Tetrasperma. J. Agric. Food Chem. 2024, DOI: 10.1021/acs.jafc.4c01061There is no corresponding record for this reference.
- 152Zhai, K.; Duan, H.; Khan, G.; Xu, H.; Han, F.-K.; Cao, W.; Gao, G.-Z.; Shan, L.-L.; Wei, Z. Salicin from Alangium Chinense Ameliorates Rheumatoid Arthritis by Modulating the Nrf2-HO-1-ROS Pathways. J. Agric. Food Chem. 2018, 66 (24), 6073– 6082, DOI: 10.1021/acs.jafc.8b02241There is no corresponding record for this reference.
- 153Sutrapu, S.; Pal, R.; Khurana, N.; Vancha, H.; Mohd, S.; Chinnala, K. M.; Kumar, B.; Pilli, G. Diabetes Warriors from Heart Wood: Unveiling Dalbergin and Isoliquiritigenin from Dalbergia Latifolia as Potential Antidiabetic Agents in-Vitro and in-Vivo. Cell Biochem. Biophys. 2024, 82, 1309, DOI: 10.1007/s12013-024-01285-xThere is no corresponding record for this reference.
- 154Valojerdi, F.; Goliaei, B.; Parivar, K.; Nikoofar, A. Effect of a Neoflavonoid (Dalbergin) on T47D Breast Cancer Cell Line and mRNA Levels of P53, Bcl-2, and STAT3 Genes. Iran. Red Crescent Med. J. 2019, 21, e87175 DOI: 10.5812/IRCMJ.87175There is no corresponding record for this reference.
- 155Wang, C.; Gong, B.; Wu, Y.; Bai, C.; Yang, M.; Zhao, X.; Wei, J. Pharmacokinetics and Molecular Docking of the Cardioprotective Flavonoids in Dalbergia Odorifera. J. Sep. Sci. 2024, DOI: 10.1002/jssc.202300614There is no corresponding record for this reference.
- 156Shen, P.; Bai, Z.; Zhou, L.; Wang, N.-N.; Ni, Z.; Sun, D.; Huang, C.-S.; Hu, Y.; Xiao, C.-R.; Zhou, W.; Zhang, B.-L.; Gao, Y. A Scd1-Mediated Metabolic Alteration Participates in Liver Responses to Low-Dose Bavachin. J. Pharm. Anal. 2023, 13, 806– 816, DOI: 10.1016/j.jpha.2023.03.010There is no corresponding record for this reference.
- 157Carrillo, J. A.; He, Y.; Li, Y.; Liu, J.; Erdman, R. A.; Sonstegard, T. S.; Song, J. Integrated Metabolomic and Transcriptome Analyses Reveal Finishing Forage Affects Metabolic Pathways Related to Beef Quality and Animal Welfare. Sci. Rep. 2016, 6 (1), 25948, DOI: 10.1038/srep25948There is no corresponding record for this reference.
- 158Ahsin, M.; Pasha, I.; Liaquat, M.; Amir, M. Genetic and Agronomic Zinc Biofortification Modify Processing and Nutritional Quality of Common Wheat. Cereal Chem. 2023, 100 (1), 131– 141, DOI: 10.1002/cche.10604There is no corresponding record for this reference.
- 159Bodnar, R. Conditioned Flavor Preferences in Animals: Merging Pharmacology. Brain Sites and Genetic Variance. Appetite 2018, 122, 17– 25, DOI: 10.1016/j.appet.2016.12.015There is no corresponding record for this reference.
- 160Jin, H.; Fishman, Z.; Ye, M.; Wang, L.; Zuker, C. Top-Down Control of Sweet and Bitter Taste in the Mammalian Brain. Cell 2021, 184, 257– 271, DOI: 10.1016/j.cell.2020.12.014There is no corresponding record for this reference.
- 161Provenza, F. After Ten Thousand Years of Domestication, Can Livestock Still Self-Medicate?. Planta Med. 2021, 87, 1237– 1237, DOI: 10.1055/s-0041-1736740There is no corresponding record for this reference.
- 162Gradé, J.; Tabuti, J.; Van Damme, P. Four Footed Pharmacists: Indications of Self-Medicating Livestock in Karamoja. Uganda. Econ. Bot. 2009, 63, 29– 42, DOI: 10.1007/s12231-008-9058-zThere is no corresponding record for this reference.
- 163Villalba, J.; Provenza, F. Nutrient-Specific Preferences by Lambs Conditioned with Intraruminal Infusions of Starch, Casein, and Water. J. Anim. Sci. 1999, 77 (2), 378– 387, DOI: 10.2527/1999.772378xThere is no corresponding record for this reference.
- 164Iommelli, P.; Spina, A.; Vastolo, A.; Infascelli, L.; Lotito, D.; Musco, N.; Tudisco, R. Functional and Economic Role of Some Mediterranean Medicinal Plants in Dairy Ruminants’ Feeding: A Review of the Effects of Garlic, Oregano, and Rosemary. Animals 2025, 15, 657, DOI: 10.3390/ani15050657There is no corresponding record for this reference.
- 165Vasta, V.; Luciano, G. The Effects of Dietary Consumption of Plants Secondary Compounds on Small Ruminants’ Products Quality. Small Rumin. Res. 2011, 101, 150– 159, DOI: 10.1016/j.smallrumres.2011.09.035There is no corresponding record for this reference.
- 166Sun, R.; Jiang, X.; Reichelt, M.; Gershenzon, J.; Pandit, S.; Vassão, G. Tritrophic Metabolism of Plant Chemical Defenses and Its Effects on Herbivore and Predator Performance. eLife 2019, 8, e51029 DOI: 10.7554/eLife.51029There is no corresponding record for this reference.
- 167Mithöfer, A.; Boland, W. Plant Defense against Herbivores: Chemical Aspects. Annu. Rev. Plant Biol. 2012, 63, 431– 450, DOI: 10.1146/annurev-arplant-042110-103854There is no corresponding record for this reference.
- 168Beringue, A.; Queffelec, J.; Lann, C. L.; Sulmon, C. Sublethal Pesticide Exposure in Non-Target Terrestrial Ecosystems: From Known Effects on Individuals to Potential Consequences on Trophic Interactions and Network Functioning. Environ. Res. 2024, 260, 119620 DOI: 10.1016/j.envres.2024.119620There is no corresponding record for this reference.
- 169Bertram, J.; Clough, T.; Sherlock, R.; Condron, L.; O’Callaghan, M.; Wells, N.; Ray, J. Hippuric Acid and Benzoic Acid Inhibition of Urine Derived N2O Emissions from Soil. Glob. Change Biol. 2009, 15, 2067, DOI: 10.1111/j.1365-2486.2008.01779.xThere is no corresponding record for this reference.
- 170Dijkstra, J.; Oenema, O.; Groenigen, J.; Spek, J.; Vuuren, A.; Bannink, A. Diet Effects on Urine Composition of Cattle and N2O Emissions. Anim. Int. J. Anim. Biosci. 2013, 7 (Suppl 2), 292– 302, DOI: 10.1017/S1751731113000578There is no corresponding record for this reference.
- 171Zhang, W.; Sun, S.; Zhang, Y.; Zhang, Y.; Wang, J.; Liu, Z.; Yang, K. Benzoic Acid Supplementation Improves the Growth Performance, Nutrient Digestibility and Nitrogen Metabolism of Weaned Lambs. Front. Vet. Sci. 2024, 11, 1351394 DOI: 10.3389/fvets.2024.1351394There is no corresponding record for this reference.
- 172Xu, Y.; Liu, L.; Zhu, J.; Zhu, S.; Ye, B.-Q.; Yang, J.-L.; Huang, J.-Y.; Huang, Z.-H.; You, Y.; Li, W.; He, J.; Xia, M.; Liu, Y. Alistipes Indistinctus-Derived Hippuric Acid Promotes Intestinal Urate Excretion to Alleviate Hyperuricemia. Cell Host Microbe 2024, 32, 366, DOI: 10.1016/j.chom.2024.02.001There is no corresponding record for this reference.
- 173Yang, Y.; Huang, S.; Liao, Y.; Wu, X.; Zhang, C.; Wang, X.; Yang, Z. Hippuric Acid Alleviates Dextran Sulfate Sodium-Induced Colitis via Suppressing Inflammatory Activity and Modulating Gut Microbiota. Biochem. Biophys. Res. Commun. 2024, 710, 149879 DOI: 10.1016/j.bbrc.2024.149879There is no corresponding record for this reference.
- 174De Mello, V.; Lankinen, M.; Lindström, J.; Puupponen-Pimiä, R.; Laaksonen, D.; Pihlajamäki, J.; Lehtonen, M.; Uusitupa, M.; Tuomilehto, J.; Kolehmainen, M.; Törrönen, R.; Hanhineva, K. Fasting Serum Hippuric Acid Is Elevated after Bilberry (Vaccinium Myrtillus) Consumption and Associates with Improvement of Fasting Glucose Levels and Insulin Secretion in Persons at High Risk of Developing Type 2 Diabetes. Mol. Nutr. Food Res. 2017, 61 (9), 1700019 DOI: 10.1002/mnfr.201700019There is no corresponding record for this reference.
- 175Pallister, T.; Jackson, M. A.; Martin, T. C.; Zierer, J.; Jennings, A.; Mohney, R. P.; MacGregor, A.; Steves, C. J.; Cassidy, A.; Spector, T. D.; Menni, C. Hippurate as a Metabolomic Marker of Gut Microbiome Diversity: Modulation by Diet and Relationship to Metabolic Syndrome. Sci. Rep. 2017, 7 (1), 13670, DOI: 10.1038/s41598-017-13722-4There is no corresponding record for this reference.
- 176Li, W.; Dong, H.; Niu, K.; Wang, H.-Y.; Cheng, W.; Song, H.; Ying, A.-K.; Zhai, X.; Li, K.; Yu, H.; Guo, D.-S.; Wang, Y. Analyzing Urinary Hippuric Acid as a Metabolic Health Biomarker through a Supramolecular Architecture. Talanta 2024, 278, 126480 DOI: 10.1016/j.Talanta.2024.126480There is no corresponding record for this reference.
- 177Fu, H.; Xu, J.; Ai, X.; Dang, F.-T.; Tan, X.; Yu, H.-Y.; Feng, J.; Yang, W.; Haining; Tu, R.; Gupta, A.; Manandhar, L. K.; Bao, W.-M.; Tang, Y.-M. The Clostridium Metabolite P-Cresol Sulfate Relieves Inflammation of Primary Biliary Cholangitis by Regulating Kupffer Cells. Cells 2022, 11, 3782, DOI: 10.3390/cells11233782There is no corresponding record for this reference.
- 178Grant, R.; Macowan, M.; Daunt, C.; Perdijk, O.; Marsland, B. Late Breaking Abstract - P-Cresol Sulfate Acts on Epithelial Cells to Reduce Allergic Airway Inflammation. Mol. Pathol. Funct. Genomics 2023, 62, PA1850, DOI: 10.1183/13993003.congress-2023.pa1850There is no corresponding record for this reference.
- 179Zhou, Y.; Bi, Z.; Hamilton, M.; Zhang, L.; Su, R.; Sadowsky, M.; Roy, S.; Khoruts, A.; Chen, C. P-Cresol Sulfate Is a Sensitive Urinary Marker of Fecal Microbiota Transplantation and Antibiotics Treatments in Human Patients and Mouse Models. Int. J. Mol. Sci. 2023, 24, 14621, DOI: 10.3390/ijms241914621There is no corresponding record for this reference.
- 180Mousavi, Y.; Adlercreutz, H. Enterolactone and Estradiol Inhibit Each Other’s Proliferative Effect on MCF-7 Breast Cancer Cells in Culture. J. Steroid Biochem. Mol. Biol. 1992, 41, 615– 619, DOI: 10.1016/0960-0760(92)90393-WThere is no corresponding record for this reference.
- 181Weiner, C.; Khan, S.; Leong, C.; Ranadive, S.; Campbell, S.; Howard, J.; Heffernan, K. Association of Enterolactone with Blood Pressure and Hypertension Risk in NHANES. PLoS One 2024, 19, e0302254 DOI: 10.1371/journal.pone.0302254There is no corresponding record for this reference.
- 182Tuomisto, A.; No̷rskov, N.; Sirniö, P.; Väyrynen, J.; Mutt, S.; Klintrup, K.; Mäkelä, J.; Knudsen, K. B.; Mäkinen, M.; Herzig, K. Serum Enterolactone Concentrations Are Low in Colon but Not in Rectal Cancer Patients. Sci. Rep. 2019, 9, 11209, DOI: 10.1038/s41598-019-47622-6There is no corresponding record for this reference.
- 183Pietinen, P.; Stumpf, K.; Männistö, S.; Kataja, V.; Uusitupa, M.; Adlercreutz, H. Serum Enterolactone and Risk of Breast Cancer: A Case-Control Study in Eastern Finland. Cancer Epidemiol. Biomark. Prev. 2001, 10 (4), 339– 344There is no corresponding record for this reference.
- 184Zhang, X.; Veliky, C.; Birru, R.; Barinas-Mitchell, E.; Magnani, J.; Sekikawa, A. Potential Protective Effects of Equol (Soy Isoflavone Metabolite) on Coronary Heart Diseases─From Molecular Mechanisms to Studies in Humans. Nutrients 2021, 13, 3739, DOI: 10.3390/nu13113739There is no corresponding record for this reference.
- 185Yoshikata, R.; Myint, K.; Ohta, H.; Ishigaki, Y. Effects of an Equol-Containing Supplement on Advanced Glycation End Products, Visceral Fat and Climacteric Symptoms in Postmenopausal Women: A Randomized Controlled Trial. PLoS One 2021, 16, e0257332 DOI: 10.1371/journal.pone.0257332There is no corresponding record for this reference.
- 186Subedi, L.; Ji, E.; Shin, D.; Jin, J.-S.; Yeo, J.; Kim, S. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9, 207, DOI: 10.3390/nu9030207There is no corresponding record for this reference.
- 187Mayo, B.; Vázquez, L.; Flórez, A. Equol: A Bacterial Metabolite from The Daidzein Isoflavone and Its Presumed Beneficial Health Effects. Nutrients 2019, 11, 2231, DOI: 10.3390/nu11092231There is no corresponding record for this reference.
- 188Puertas-Bartolomé, M.; Benito-Garzón, L.; Fung, S.; Kohn, J.; Vázquez-Lasa, B.; Román, S. Bioadhesive Functional Hydrogels: Controlled Release of Catechol Species with Antioxidant and Antiinflammatory Behavior. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 105, 110040 DOI: 10.1016/J.MSEC.2019.110040There is no corresponding record for this reference.
- 189Lim, W.-C.; Kim, H.; Kim, Y.-J.; Jeon, B.; Kang, H.; Ko, H. Catechol Inhibits Epidermal Growth Factor-Induced Epithelial-to-Mesenchymal Transition and Stem Cell-like Properties in Hepatocellular Carcinoma Cells. Sci. Rep. 2020, 10, 7620, DOI: 10.1038/s41598-020-64603-2There is no corresponding record for this reference.
- 190Chang, M.; Chang, H.-H.; Wang, T.-M.; Chan, C.; Lin, B.; Yeung, S.; Yeh, C.-Y.; Cheng, R.-H.; Jeng, J. Antiplatelet Effect of Catechol Is Related to Inhibition of Cyclooxygenase, Reactive Oxygen Species, ERK/P38 Signaling and Thromboxane A2 Production. PLoS One 2014, 9, e104310 DOI: 10.1371/journal.pone.0104310There is no corresponding record for this reference.
- 191Jothi, R.; Sangavi, R.; Kumar, P.; Pandian, S.; Gowrishankar, S. Catechol Thwarts Virulent Dimorphism in Candida Albicans and Potentiates the Antifungal Efficacy of Azoles and Polyenes. Sci. Rep. 2021, 11, 21049, DOI: 10.1038/s41598-021-00485-2There is no corresponding record for this reference.
- 192Bukowska, B.; Michałowicz, J.; Marczak, A. The Effect of Catechol on Human Peripheral Blood Mononuclear Cells (in Vitro Study). Environ. Toxicol. Pharmacol. 2015, 39 (1), 187– 193, DOI: 10.1016/j.etap.2014.11.017There is no corresponding record for this reference.
- 193Mhawish, R.; Komarnytsky, S. Small Phenolic Metabolites at the Nexus of Nutrient Transport and Energy Metabolism. Molecules 2025, 30, 1026, DOI: 10.3390/molecules30051026There is no corresponding record for this reference.
- 194Gaur, G.; Gänzle, M. Conversion of (Poly)Phenolic Compounds in Food Fermentations by Lactic Acid Bacteria: Novel Insights into Metabolic Pathways and Functional Metabolites. Curr. Res. Food Sci. 2023, 6, 100448 DOI: 10.1016/j.crfs.2023.100448There is no corresponding record for this reference.
- 195Del Carmen Villegas-Aguilar, M.; Fernández-Ochoa, Á.; Cádiz-Gurrea, M.; Pimentel-Moral, S.; Lozano-Sánchez, J.; Arráez-Román, D.; Segura-Carretero, A. Pleiotropic Biological Effects of Dietary Phenolic Compounds and Their Metabolites on Energy Metabolism, Inflammation and Aging. Molecules 2020, 25, 596, DOI: 10.3390/molecules25030596There is no corresponding record for this reference.
- 196Tonolo, F.; Folda, A.; Cesaro, L.; Scalcon, V.; Marin, O.; Ferro, S.; Bindoli, A.; Rigobello, M. Milk-Derived Bioactive Peptides Exhibit Antioxidant Activity through the Keap1-Nrf2 Signaling Pathway. J. Funct. Foods 2020, 64, 103696 DOI: 10.1016/j.jff.2019.103696There is no corresponding record for this reference.
- 197Husted, A.; Trauelsen, M.; Rudenko, O.; Hjorth, S.; Schwartz, T. GPCR-Mediated Signaling of Metabolites. Cell Metab. 2017, 25 (4), 777– 796, DOI: 10.1016/j.cmet.2017.03.008There is no corresponding record for this reference.
- 198Li, W.; Qiu, H.; Van Gestel, C.; Peijnenburg, W.; He, E. Trophic Transfer and Toxic Potency of Rare Earth Elements along a Terrestrial Plant-Herbivore Food Chain. Environ. Sci. Technol. 2024, 58, 5705, DOI: 10.1021/acs.est.3c09179There is no corresponding record for this reference.
- 199Nfon, E.; Cousins, I.; Broman, D. Biomagnification of Organic Pollutants in Benthic and Pelagic Marine Food Chains from the Baltic Sea. Sci. Total Environ. 2008, 397 (1–3), 190– 204, DOI: 10.1016/j.scitotenv.2008.02.029There is no corresponding record for this reference.
- 200Scalbert, A.; Williamson, G. Dietary Intake and Bioavailability of Polyphenols. J. Nutr. 2000, 130 (8S Suppl), 2073S– 2085, DOI: 10.1093/jn/130.8.2073SThere is no corresponding record for this reference.
- 201Huang, Q.; Braffett, B.; Simmens, S.; Young, H.; Ogden, C. Dietary Polyphenol Intake in US Adults and 10-Year Trends: 2007–2016. J. Acad. Nutr. Diet. 2020, 120, 1821, DOI: 10.1016/j.jand.2020.06.016There is no corresponding record for this reference.
- 202Del Bo’, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; Zamora-Ros, R.; Liberona, N. H.; Andrés-Lacueva, C.; Riso, P. Systematic Review on Polyphenol Intake and Health Outcomes: Is There Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern?. Nutrients 2019, 11, 1355, DOI: 10.3390/nu11061355There is no corresponding record for this reference.
- 203Komatsu, S.; Sakamoto, S.; Yusakul, G.; Putalun, W.; Miyamoto, T.; Tanaka, H.; Morimoto, S. A Single-Chain Variable Fragment Antibody against Anti-Leukemia Agent, Harringtonine as a Tool for Immunomodulation. Planta Med. 2016, 82, S1– S381, DOI: 10.1055/S-0036-1596806There is no corresponding record for this reference.
- 204Takeda, S.; Yajima, N.; Kitazato, K.; Unemi, N. Antitumor Activities of Harringtonine and Homoharringtonine, Cephalotaxus Alkaloids Which Are Active Principles from Plant by Intraperitoneal and Oral Administration. J. Pharmacobiodyn. 1982, 5 (10), 841– 847, DOI: 10.1248/bpb1978.5.841There is no corresponding record for this reference.
- 205Gupta, S.; Khajuria, V.; Wani, A.; Nalli, Y.; Bhagat, A.; Ali, A.; Ahmed, Z. Murrayanine Attenuates Lipopolysaccharide-induced Inflammation and Protects Mice from Sepsis-associated Organ Failure. Basic Clin. Pharmacol. Toxicol. 2019, 124, 351, DOI: 10.1111/bcpt.13032There is no corresponding record for this reference.
- 206Mahapatra, D. K.; Chhajed, S.; Shivhare, R. Development of Murrayanine-Chalcone Hybrids: An Effort to Combine Two Privilege Scaffolds for Enhancing Hypoglycemic Activity. Int. J. Pharm. Chem. 2017, 4, 30– 34There is no corresponding record for this reference.
- 207Mahapatra, D. K.; Dadure, K.; Shivhare, R. Edema Reducing Potentials of Some Emerging Schiff’s Bases of Murrayanine. MOJ. Bioorg. Org. Chem. 2018, 2, 171– 174, DOI: 10.15406/mojboc.2018.02.0076There is no corresponding record for this reference.
- 208Zhou, H.; Li, H.; Cao, Y.-P.; Sang, X.; Liu, X. Murrayanine Exerts Antiproliferative Effects on Human Oral Cancer Cells through Inhibition of AKT/mTOR and Raf/MEK/ERK Signalling Pathways in Vitro and Inhibits Tumor Growth in Vivo. J. BUON Off. J. Balk. Union Oncol. 2019, 24 (6), 2423– 2428There is no corresponding record for this reference.
- 209Cui, Z.; Wu, Y.; Wang, C.-Y.; Dai, X.; Nan, J.-X.; Liu, S.-H.; Lian, L.; Guo, J.; Jiang, Y.-C. Vincamine Ameliorates Hepatic Fibrosis via Inhibiting S100A4-mediated Farnesoid X Receptor Activation: Based on Liver Microenvironment and Enterohepatic Circulation Dependence. Br. J. Pharmacol. 2025, 182, 2447– 2465, DOI: 10.1111/bph.17471There is no corresponding record for this reference.
- 210Du, T.; Yang, L.; Xu, X.; Shi, X.-F.; Xu, X.; Lu, J.; Lv, J.; Huang, X.; Chen, J.; Wang, H.; Ye, J.; Hu, L.; Shen, X. Vincamine as a GPR40 Agonist Improves Glucose Homeostasis in Type 2 Diabetic Mice. J. Endocrinol. 2019, 240 (2), 195– 214, DOI: 10.1530/JOE-18-0432There is no corresponding record for this reference.
- 211Nandini, H.; Naik, P. Antidiabetic, Antihyperlipidemic and Antioxidant Effect of Vincamine, in Streptozotocin-induced Diabetic Rats. Eur. J. Pharmacol. 2019, 843, 233, DOI: 10.1016/j.ejphar.2018.11.034There is no corresponding record for this reference.
- 212Sanz, F.; Solana-Manrique, C.; Paricio, N. Disease-Modifying Effects of Vincamine Supplementation in Drosophila and Human Cell Models of Parkinson’s Disease Based on DJ-1 Deficiency. ACS Chem. Neurosci. 2023, 14, 2294– 2301, DOI: 10.1021/acschemneuro.3c00026There is no corresponding record for this reference.
- 213Renushe, A. P.; Banothu, A. K.; Bharani, K. K.; Mekala, L.; Kumar, M.; Neeradi, D.; Hanuman, D. D. V.; Gadige, A.; Khurana, A. Vincamine, an Active Constituent of Vinca Rosea Ameliorates Experimentally Induced Acute Lung Injury in Swiss Albino Mice through Modulation of Nrf-2/NF-κB Signaling Cascade. Int. Immunopharmacol. 2022, 108, 108773 DOI: 10.1016/j.intimp.2022.108773There is no corresponding record for this reference.
- 214Qin, N.; Xu, G.; Wang, Y.; Zhan, X.; Gao, Y.; Wang, Z.; Fu, S.; Shi, W.; Hou, X.; Wang, C.; Li, R.-S.; Liu, Y.; Wang, J.; Zhao, H.; Xiao, X.; Bai, Z. Bavachin Enhances NLRP3 Inflammasome Activation Induced by ATP or Nigericin and Causes Idiosyncratic Hepatotoxicity. Front. Med. 2021, 15, 594– 607, DOI: 10.1007/s11684-020-0809-2There is no corresponding record for this reference.
- 215Zhang, X.; Guo, Y.; Zhang, Z.; Wu, X.-Y.; Li, L.; Yang, Z.; Li, Z. Neuroprotective Effects of Bavachin against Neuroinflammation and Oxidative Stress-Induced Neuronal Damage via Activation of Sirt1/Nrf2 Pathway and Inhibition of NF-κB Pathway. J. Funct. Foods 2023, 107, 105655 DOI: 10.1016/j.jff.2023.105655There is no corresponding record for this reference.
- 216Park, J.; Seo, E.; Jun, H. Bavachin Alleviates Diabetic Nephropathy in Db/Db Mice by Inhibition of Oxidative Stress and Improvement of Mitochondria Function. Biomed. Pharmacother. Biomedecine Pharmacother. 2023, 161, 114479 DOI: 10.1016/j.biopha.2023.114479There is no corresponding record for this reference.
- 217Chakraborty, D.; Malik, S.; Mann, S.; Agnihotri, P.; Joshi, L.; Biswas, S. Chronic Disease Management via Modulation of Cellular Signaling by Phytoestrogen Bavachin. Mol. Biol. Rep. 2024, 51 (1), 921, DOI: 10.1007/s11033-024-09849-zThere is no corresponding record for this reference.
- 218Wang, C.; Hu, X.; Song, T.; Hu, F.; Du, L.; Yan, C.; Shen, T.; Li, N.; Yang, W.; Li, L.; Deng, N.; Jiang, X.; Wu, Y.; Ye, R. Magnolin Mitigates Skin Ageing Through the CXCL10/P38 Signalling Pathway. J. Cell. Mol. Med. 2025, 29, e70507 DOI: 10.1111/jcmm.70507There is no corresponding record for this reference.
- 219Xu, K.; Gao, Y.; Yang, L.; Liu, Y.; Wang, C. Magnolin Exhibits Anti-Inflammatory Effects on Chondrocytes via the NF-κB Pathway for Attenuating Anterior Cruciate Ligament Transection-Induced Osteoarthritis. Connect. Tissue Res. 2021, 62, 475– 484, DOI: 10.1080/03008207.2020.1778679There is no corresponding record for this reference.
- 220Patel, D. Therapeutic Effectiveness of Magnolin on Cancers and Other Human Complications. Pharmacol. Res. - Mod. Chin. Med. 2023, 6, 100203 DOI: 10.1016/j.prmcm.2022.100203There is no corresponding record for this reference.
- 221Changan, S.; Tomar, M.; Prajapati, U.; Saurabh, V.; Hasan, M.; Sasi, M.; Maheshwari, C.; Singh, S.; Dhumal, S. S.; Radha; Thakur, M.; Punia, S.; Satankar, V.; Amarowicz, R.; Mekhemar, M. Custard Apple (Annona Squamosa L.) Leaves: Nutritional Composition, Phytochemical Profile, and Health-Promoting Biological Activities. Biomolecules 2021, 11, 614, DOI: 10.3390/biom11050614There is no corresponding record for this reference.
- 222Toropova, A.; Razuvaeva, Y.; Olennikov, D. Dihydrosamidin: The Basic Khellactone Ester Derived from Phlojodicarpus Komarovii and Its Impact on Neurotrophic Factors, Energy and Antioxidant Metabolism after Rat Cerebral Ischemia-Reperfusion Injury. Nat. Prod. Res. 2024, 1– 6, DOI: 10.1080/14786419.2024.2433189There is no corresponding record for this reference.
- 223Jia, N.; Shen, Z.; Zhao, S.; Wang, Y.; Pei, C.; Huang, D.; Wang, X.; Wu, Y.; Shi, S.; He, Y.; Wang, Z. Eleutheroside E from Pre-Treatment of Acanthopanax Senticosus (Rupr.etMaxim.) Harms Ameliorates High-Altitude-Induced Heart Injury by Regulating NLRP3 Inflammasome-Mediated Pyroptosis via NLRP3/Caspase-1 Pathway. Int. Immunopharmacol. 2023, 121, 110423 DOI: 10.1016/j.intimp.2023.110423There is no corresponding record for this reference.
- 224Song, C.; Duan, F.; Ju, T.; Qin, Y.; Zeng, D.; Shan, S.; Shi, Y.; Zhang, Y.; Lu, W. Eleutheroside E Supplementation Prevents Radiation-Induced Cognitive Impairment and Activates PKA Signaling via Gut Microbiota. Commun. Biol. 2022, 5, 680, DOI: 10.1038/s42003-022-03602-7There is no corresponding record for this reference.
- 225Zhou, T.; Zhou, Y.; Ge, D.; Xie, Y.; Wang, J.; Tang, L.; Dong, Q.; Sun, P. Decoding the Mechanism of Eleutheroside E in Treating Osteoporosis via Network Pharmacological Analysis and Molecular Docking of Osteoclast-Related Genes and Gut Microbiota. Front. Endocrinol. 2023, 14, 1257298 DOI: 10.3389/fendo.2023.1257298There is no corresponding record for this reference.
- 226Jaiswal, J.; Srivastav, A. K.; Kushwaha, M.; Teotia, A.; Singh, R.; Mohan, A.; Makharia, G.; Kumar, A. Gut Microbial Metabolite 4-Ethylphenylsulfate Is Selectively Deleterious and Anticancer to Colon Cancer Cells. J. Med. Chem. 2025, 68, 10425, DOI: 10.1021/acs.jmedchem.5c00609There is no corresponding record for this reference.
- 227Angelino, D.; Carregosa, D.; Domenech-Coca, C.; Savi, M.; Figueira, I.; Brindani, N.; Jang, S.; Lakshman, S.; Molokin, A.; Urban, J.; Davis, C.; Brito, M.; Kim, K.; Brighenti, F.; Curti, C.; Bladé, C.; Del Bas, J.; Stilli, D.; Solano-Aguilar, G.; Santos, C.; Del Rio, D.; Mena, P. 5-(Hydroxyphenyl)-γ-Valerolactone-Sulfate, a Key Microbial Metabolite of Flavan-3-Ols, Is Able to Reach the Brain: Evidence from Different in Silico, In Vitro and In Vivo Experimental Models. Nutrients 2019, 11, 2678, DOI: 10.3390/nu11112678There is no corresponding record for this reference.
- 228Marcolin, E.; Chemello, C.; Piovan, A.; Barbierato, M.; Morazzoni, P.; Ragazzi, E.; Zusso, M. A Combination of 5-(3′,4′-Dihydroxyphenyl)-γ-Valerolactone and Curcumin Synergistically Reduces Neuroinflammation in Cortical Microglia by Targeting the NLRP3 Inflammasome and the NOX2/Nrf2 Signaling Pathway. Nutrients 2025, 17, 1316, DOI: 10.3390/nu17081316There is no corresponding record for this reference.
- 229Della Vedova, L.; Husain, I.; Wang, Y.-H.; Kothapalli, H.; Gado, F.; Baron, G.; Manzi, S.; Morazzoni, P.; Aldini, G.; Khan, I. Pre-ADMET Studies of 5-(3′,4′-dihydroxyphenyl)-γ-valerolactone, the Bioactive Intestinal Metabolite of Proanthocyanidins. Arch. Pharm. (Weinheim) 2025, 358, e2400575 DOI: 10.1002/ardp.202400575There is no corresponding record for this reference.
- 230Oliveira, M.; Ratti, B.; Daré, R.; Silva, S.; Truiti, M.; Ueda-Nakamura, T.; Auzély-Velty, R.; Nakamura, C. Dihydrocaffeic Acid Prevents UVB-Induced Oxidative Stress Leading to the Inhibition of Apoptosis and MMP-1 Expression via P38 Signaling Pathway. Oxid. Med. Cell. Longev. 2019, 2019, 2419096 DOI: 10.1155/2019/2419096There is no corresponding record for this reference.
- 231Martini, S.; Conte, A.; Tagliazucchi, D. Antiproliferative Activity and Cell Metabolism of Hydroxycinnamic Acids in Human Colon Adenocarcinoma Cell Lines. J. Agric. Food Chem. 2019, 67 (14), 3919– 3931, DOI: 10.1021/acs.jafc.9b00522There is no corresponding record for this reference.
- 232Stalmach, A.; Mullen, W.; Barron, D.; Uchida, K.; Yokota, T.; Cavin, C.; Steiling, H.; Williamson, G.; Crozier, A. Metabolite Profiling of Hydroxycinnamate Derivatives in Plasma and Urine after the Ingestion of Coffee by Humans: Identification of Biomarkers of Coffee Consumption. Drug Metab. Dispos. 2009, 37, 1749– 1758, DOI: 10.1124/dmd.109.028019There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jafc.5c06118.
Figure S1. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram summarizing the literature search and study selection process followed to selected eligible studied to be this systematically review. Figure S2. Risk of bias and quality of reporting assessment based on SYRCLE’s risk of bias tool 1: (A) indicator of quality of reporting included study design and objectives, population and sample characteristics, intervention and comparison groups, analytical methods, data analysis and presentation, ethical considerations, funding and conflicts of interest (B) risk-of-bias analysis in selection, measurement, confounding, and selective reporting. Table S1 List of phenolic compounds identified in selected ruminants meat and milk samples from studies included in the systematic review, organized by matrix (species/product). Table S2. Study-level data extraction for 39 studies, including sample type; country of origin; experimental group (e.g., breed, fresh vs conserved forage, short- vs long-term pasture, month of year, commercial samples, milk-fat content); measured phenolics (relevant compounds extracted from metabolomics data sets); concentrations (where reported, with n per group); analytical platform; and references (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


