Binary Mixtures of n-Alkylbenzenes and Pentadecane: Densities, Speeds of Sound, and Viscosities within the Range of 288.15 and 333.15 K and at 0.1 MPaClick to copy article linkArticle link copied!
- Dianne J. Luning Prak*Dianne J. Luning Prak*Email: [email protected]Phone: 410-293-6339. Fax: 410-293-2218.Department of Chemistry, U.S. Naval Academy, 572 M Holloway Road, Annapolis, Maryland 21402, United StatesMore by Dianne J. Luning Prak
- Jim S. CowartJim S. CowartDepartment of Mechanical and Nuclear Engineering, U.S. Naval Academy, 590 Holloway Rd, Annapolis, Maryland 21402, United StatesMore by Jim S. Cowart
Abstract
The physical properties of mixtures of alkanes and aromatic compounds can aid in the understanding and modeling of their combustion in engines. Herein, densities, viscosities, and speeds of sound of binary mixtures of n-alkylbenzenes with pentadecane and their corresponding excess molar volumes (VmE), excess speeds of sound (cE), excess isentropic compressibilities (ΚsE), and viscosity deviations (Δη) are reported. In general, mixture densities, viscosities, and speeds of sound increased monotonically with changing mole fractions, except for the speeds of sound of toluene, ethylbenzene, propylbenzene, and butylbenzene mixtures. These properties can be used for fuel modeling purposes. VmE’s, Δη’s, cE’s, and KsE’s ranged from 0.04 to 0.56 cm·mol–1, −0.14 to 0.09 mPa·s, −14.8 to 2.5 m·s–1, and −2.2 to 18.1 TPa–1, respectively. Increasing the n-alkylbenzene size (up to decylbenzene) caused the equimolar mixture’s VmE’s, Δη’s, and KsE’s to decrease and cE’s to increase at 293.15 K. The properties of smaller n-alkylbenzene molecules were more affected by the aromatic group. In comparison with n-alkylcyclohexane/pentadecane mixtures, n-alkylbenzene/pentadecane mixture Δη trends were similar, but VmE’s, cE’s, and KsE’s trends were different, suggesting that molecular interactions of the benzyl and cyclohexyl groups affect volume and compressibility more than they affect viscosity.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
| linear alkane | n-alkyl-benzene | density temperatures (K) | viscosity temperatures (K) | speed of sound temperatures (K) |
|---|---|---|---|---|
| hexane | methyl- | 298.15 and 323.15 (15) | 313.15 (12) | |
| 313.15 (12) | ||||
| hexane | ethyl- | 298.15 and 323.15 (15) | 313.15 (12) | |
| 313.15 (12) | ||||
| hexane | propyl- | 298.15 and 323.15 (15) | ||
| hexane | butyl- | 298.15 and 323.15 (15) | ||
| heptane | methyl- | 313.15 (12) | 313.15 (12) | |
| heptane | ethyl | 313.15 (12) | 313.15 (12) | |
| heptane | nonyl- | 313.15–353.15 (13) | ||
| octane | methyl- | 293.15–313.15 (9) | ||
| 313.15 (12) | ||||
| octane | ethyl- | 293.15–313.15 (9) | 313.15 (12) | |
| 313.15 (12) | ||||
| nonane | methyl- | 313.15 (12) | 313.15 (12) | |
| nonane | ethyl- | 313.15 (12) | 313.15 (12) | |
| decane | methyl- | 293.15–313.15 (9) | 298.15 (11) | |
| 298.15 (11) | ||||
| decane | butyl- | 293.15–373.15 (6) | 293.15–373.15 (6) | 293.15–333.15 (6) |
| decane | dodecyl- | 288.15–333.15 (16) | 288.15–323.15 (16) | 288.15–333.15 (16) |
| dodecane | methyl- | 293.15–313.15 (9) | ||
| dodecane | butyl- | 293.15–373.15 (6) | 293.15–373.15 (6) | 293.15–333.15 (6) |
| dodecane | nonyl- | 288.15–333.15 (10) | 293.15–333.15 (10) | 288.15–333.15 (10) |
| dodecane | dodecyl- | 288.15–333.15 (16) | 288.15–333.15 (16) | 288.15–333.15 (16) |
| tridecane | dodecyl- | 288.15–333.15 (16) | 288.15–333.15 (16) | 288.15–333.15 (16) |
| tetradecane | methyl- | 293.15–313.15 (9) | 298.15 (11) | |
| 298.15 (11) | ||||
| tetradecane | ethyl- | 293.15–313.15 (9) | ||
| tetradecane | butyl- | 293.15–373.15 (6) | 293.15–373.15 (6) | 293.15–333.15 (6) |
| tetradecane | dodecyl- | 288.15–333.15 (16) | 288.15–333.15 (16) | 288.15–333.15 (16) |
| pentadecane | dodecyl- | 288.15–333.15 (16) | 288.15–333.15 (16) | 288.15–333.15 (16) |
| hexadecane | methyl- | 293.15–313.15 (9) | 293.15–373.15 (7,26) | 293.15–333.15 (7,26) |
| 293.15–373.15 (7,26) | ||||
| hexadecane | ethyl- | 293.15–313.15 (9) | 293.15–373.15 (7,26) | 293.15–333.15 (7,26) |
| 293.15–373.15 (7,26) | ||||
| hexadecane | propyl- | 298.15–318.15 (14) | 298.15–318.15 (14) | 298.15–318.15 (14) |
| hexadecane | butyl- | 293.15–373.15 (6) | 293.15–373.15 (6) | 293.15–333.15 (6) |
| hexadecane | hexyl- | 293.15–373.15 (8) | 293.15–373.15 (8) | 293.15–333.15 (8) |
| hexadecane | octyl- | 293.15–373.15 (8) | 293.15–373.15 (8) | 293.15–333.15 (8) |
| hexadecane | nonyl- | 293.15–333.15 (10) | 293.15–333.15 (10) | 293.15–333.15 (10) |
| hexadecane | dodecyl- | 293.15–373.15 (8) | 293.15–373.15 (8) | 293.15–333.15 (8) |
| heptadecane | butyl- | 303.15–373.15 (6) | 303.15–373.15 (6) | 303.15–333.15 (6) |
2. Experimental and/or Computational Methods
| chemical name | CAS number | molar mass (g/mol)a | source | mole fraction purityb |
|---|---|---|---|---|
| pentadecane (C15H32) | 629-62-9 | 212.41 ± 0.04 | TCI | 0.998 |
| n-decylbenzene (C16H26) | 2189-60-8 | 218.38 ± 0.03 | TCI | 0.994 |
| n-octylbenzene (C14H22) | 2189-60-8 | 190.32 ± 0.03 | TCI | 0.982 |
| n-heptylbenzene (C13H20) | 1078-71-3 | 176.30 ± 0.03 | TCI | 0.995 |
| n-hexylbenzene (C12H18) | 1077-16-3 | 162.27 ± 0.02 | TCI | 0.993 |
| n-pentylbenzene (C11H16) | 538-68-1 | 148.25 ± 0.02 | Aldrich | 0.999 |
| n-butylbenzene (C10H14) | 104-51-8 | 134.22 ± 0.02 | TCI | 0.999 |
| n-propylbenzene (C9H12) | 103-65-1 | 120.20 ± 0.02 | TCI | 0.999 |
| ethylbenzene (C8H10) | 100-41-4 | 106.17 ± 0.02 | TCI | 1.000 |
| toluene (C7H8) | 108-88-3 | 92.14 ± 0.01 | Pharmco | 0.9987 |
Calculated using values in the work by Harris. (30)
The method of analysis for all the compounds is gas–liquid chromatography, as specified in the Certificates of Analysis provided by the chemical suppliers.
3. Results and Discussion
3.1. Pure Components: Comparison of Densities, Viscosities, and Speeds of Sound with Literature Values
| T/K | meas.a ρ/kg·m–3 | literature ρ/kg·m–3 | meas.a ρ/kg·m–3 | literature ρ/kg·m–3 |
|---|---|---|---|---|
| Pentadecane | n-octylbenzene | |||
| 288.15 | 772.23 | 772.01c, 771.8b, 772.24, (16,34) 772.4 (35) | 860.6 | 859.86 (36) |
| 293.15 | 768.72 | 768.28, (35) 768.3, (35) 768.35b, 768.45, (37) 768.4, (35) 768.5, (35) 768.52c, 768.73, (34) 768.9 (35) | 857.0 | 856.24, (36) 856.27, (8) 856.30, (38) 856.5, (39) 856.70, (38) 857.2, (40) 857.60, (41) 858.20, (42) 858.3 (38) |
| 298.15 | 765.22 | 764.88, (35) 764.9b, 765, (43) 765.04c, 765.07, (35) 765.20, (34) 765.23, (35) 765.42 (35) | 853.4 | 852.61 (36) |
| 303.15 | 761.73 | 761.38, (35) 761.4, (43) 761.48b, 761.56c, 761.48, (37) 761.70, (34) 761.74 (16) | 849.7 | 848.98, (36) 849.02, (8) 851.20 ± 1.00 (38) |
| 313.15 | 754.74 | 754.6c, 754.63b, 754.52, (37) 754.75, (16) 754.80 (34) | 842.5 | 841.74, (8) 841.9 ± 0.8, (39) 842.71 (36) |
| 323.15 | 747.75 | 747.56, (37) 747.65c, 747.77, (16) 747.78b, 747.80 (34) | 835.2 | 834.44, (36) 834.47 (8) |
| 333.15 | 740.75 | 740.58, (37) 740.66, (35) 740.68c, 740.77, (16) 740.8, (35) 740.92b, 741.02 (35) | 827.9 | 827.16, (36) 827.17, (8) 827.50 ± 0.60 (38) |
| average MAPE = 0.09 | average MAPE = 0.09 | |||
| n-heptylbenzene | n-hexylbenzene | |||
| 288.15 | 860.04 | 860.1 (36) | 861.39 | 861.92b, 862.3, (33) 862.7, (36) 863.92 (38) |
| 293.15 | 856.35 | 857.5 (44) (uncertainty = 1 kg·m–3), (44) 856.4 (36) | 857.60 | 857.68, (8) 858.00, (38) 858.02, (38) 858.47b, 858.6, (33) 858.9, (36) 860.0, (45) 862.4 (39) |
| 298.15 | 852.65 | 852.7 (36) | 853.81 | 855.01b, 855.1 (36) |
| 303.15 | 848.96 | 849.0, (36) 850.1 (44) (uncertainty = 1 kg·m–3) (44) | 850.02 | 851.0, (33) 851.3, (36) 851.56b,852.40 (38) |
| 313.15 | 841.55 | 841.6, (36) 842.5 (44) (uncertainty = 1 kg·m–3) (44) | 842.43 | 843.4, (33) 844.65b, 846.8, (39) 843.7 (36) |
| 323.15 | 834.14 | 834.2, (36) 835.1 (44) (uncertainty = 1 kg·m–3) (44) | 834.81 | 834.88, (8) 835.8, (33) 836.1, (36) 837.7b |
| 333.15 | 826.69 | 826.7, (36) 827.6 (44) (uncertainty = 1 kg·m–3) (44) | 827.17 | 828.2, (33) 828.4, (36) 830.8b, 831.7 (39) |
| average MAPE = 0.05 | average MAPE = 0.2 | |||
| n-pentylbenzene | n-butylbenzene | |||
| 288.15 | 862.81 | 862.278, (46) 862.59, (38) 862.82 (36) | 864.51 | 864.35, (36) 864.73 (38) |
| 293.15 | 858.92 | 858.93, (36) 859.02, (38) 859.1, (38) 859.2, (38) 859.3, (38) 859.4, (38) 860 (38) | 860.50 | 859.50, (38) 860.15, (38) 860.25, (38) 860.34, (36) 861.26 (47) |
| 298.15 | 855.02 | 855.02, (46) 855.04 (36) | 856.49 | 856.33 (36) |
| 303.15 | 851.13 | 850.98, (38) 852.6, (38) 851.14 (36) | 852.47 | 851.3, (48) 852.16, (38) 852.23, (47) 852.32, (36) 852.43 (38) |
| 313.15 | 843.31 | 843.33 (36) | 844.42 | 844.27, (36) 844.6, (48) 844.82 (38) |
| 323.15 | 835.48 | 835.49 (36) | 836.33 | 836.18, (36) 836.34, (6) 837.00 (38) |
| 333.15 | 827.60 | 827.62 (36) | 828.20 | 828.06, (36) 828.21 (6) |
| average MAPE = 0.02 | average MAPE = 0.03 | |||
| n-propylbenzene | ethylbenzene | |||
| 288.15 | 867.01 | 866.20 (36) | 871.46 | 871.08, (46) 871.37, (38) 871.67 (36) |
| 293.15 | 862.80 | 862.00, (36) 861.83, (38) 861.6, (38) 861.9, (38) 861.96, (38) 861.3, (38) 862.02, (38) 863.13 (47) | 867.07 | 866.94, (38) 866.95, (26) 867.28, (36) 866.97, (38) 867 (38) |
| 298.15 | 858.58 | 857.78, (36) 857.77, (38) 857.68, (38) 857.74, (38) 867.78, (38) 857.96, (38) 858.1 (38) | 862.67 | 862.25, (38) 862.29, (46) 862.56, (38) 862.62, (38) 862.89 (36) |
| 303.15 | 854.36 | 853.56, (36) 853.68, (38) 854.71 (47) | 858.26 | 857.98, (38) 858.15, (26) 858.26, (38) 858.49 (36) |
| 313.15 | 845.90 | 845.10, (36) 844.9, 845.08 (38) | 849.41 | 849.29, (26) 849.47, (38) 849.63 (36) |
| 323.15 | 837.39 | 836.59 (36) | 840.50 | 839.78, (38) 840.38, (26) 840.39 (38) |
| 333.15 | 828.84 | 828.05 (49) | 831.52 | 831.18, (38) 831.4, (26) 831.69, (38) 831.8 (38) |
| average MAPE = 0.09 | average MAPE = 0.02 | |||
| toluene | decylbenzene | |||
| 288.15 | 871.37 | 871.46 (36) | ||
| 293.15 | 866.73 | 866.8138, 866.8236, 866.8338, 866.8638 | 855.49 | 855.40, 855.53 |
| 298.15 | 862.07 | 861.89, 862.13, (38) 862.16, (36) 862.17, (38) 862.19, (38) 866.22 (38) | 851.97 | 851.89 |
| 303.15 | 857.41 | 857.5, (36) 857.53, (38) 853.54, (38) 857.55 (38) | 848.45 | 848.33 |
| 333.15 | average MAPE = 0.01 | 827.33 | 827.5 | |
| average MAPE = 0.01 | ||||
“Meas.” is the measured value. The average pressure for these measurements was 0.102 MPa. Standard uncertainties are u(T) = 0.01 K and u(p) = 1 kPa; relative standard uncertainties are ur(ρ) = 0.0006 for n-decylbenzene, ur(ρ) = 0.0018 for n-octylbenzene, ur(ρ) = 0.0005 for n-heptylbenzene, ur(ρ) = 0.0007 for n-hexylbenzene, and ur(ρ) = 0.0002 for pentadecane, n-pentylbenzene, n-butylbenzene, n-propylbenzene, ethylbenzene, and toluene.
Best-fit equation from the Wilhoit compendium of density data for n-pentadecane: (35) ρ = 1059.18 – 1.54195 × T + 0.00271745 × T2 – 2.87122 × 10–6 × T3 and hexylbenzene: (38) ρ/kg·m3 = 1060.96 – 0.690758 × T.
Best-fit equation from Bessieres et al.: (50) ρ = 1038.677 – 1.3320105 × T + 0.00203628 × T2 – 2.17 × 10–6 × T3.
| T/K | meas.a η/mPa·s | literature η/mPa·s | meas.a η/mPa·s | literature η/mPa·s |
|---|---|---|---|---|
| pentadecane | n-octylbenzene | |||
| 288.15 | 3.24 | 3.232, (51) 3.248, (51) 3.25 (16) | 2.92 | 2.87 (51) |
| 293.15 | 2.88 | 2.841, (51) 2.842, (51) 2.862, (51) 2.87, (16,34) 2.872 (51) | 2.61 | 2.57, (51) 2.575, (51) 2.58, (51) 2.606 (51) |
| 298.15 | 2.57 | 2.534, (51) 2.544, (51) 2.568, (51) 2.57 (16) | 2.33 | 2.31 (51) |
| 303.15 | 2.31 | 2.283, (51) 2.31 (16,51) | 2.10 | 2.08, (8) 2.09 (51) |
| 313.15 | 1.90 | 1.868, (51) 1.873, (51) 1.89, (16,34) 1.9, (51) 1.95 (51) | 1.75 | 1.72, (36) 1.73, (8) 1.744 (51) |
| 323.15 | 1.59 | 1.567, (51) 1.58, (16,34) 1.591 (51) | 1.48 | 1.46, (8) 1.479 (51) |
| 333.15 | 1.35 | 1.331, (51) 1.335, (51) 1.34, (16,34) 1.353, (51) 1.36 (51) | 1.27 | 1.25, (8) 1.254 (51) 1.274 (51) |
| MAPE = 0.6 | MAPE = 1 | |||
| n-heptylbenzene | n-hexylbenzene | |||
| 288.15 | 2.30 | 2.31 (51) | 1.83 | 1.833, (51) 1.846 (51) |
| 293.15 | 2.07 | 2.066, (51) 2.08, (36,51) 2.1 (44) | 1.67 | 1.65, (36) 1.655, (51) 1.67, (51) 1.68, (8) 1.70 (51) |
| 298.15 | 1.89 | 1.887 (51) | 1.52 | 1.528 (51) |
| 303.15 | 1.72 | 1.71, (36) 1.722, (51) 1.74 (44) | 1.39 | 1.38, (36) 1.40, (8) 1.403, (51) 1.409 (51) |
| 313.15 | 1.44 | 1.43, (36) 1.451, (51) 1.48 (44) | 1.18 | 1.18, (36) 1.19, (8) 1.196, (51) 1.2 (51) |
| 323.15 | 1.23 | 1.22, (36) 1.243, (51) 1.26 (44) | 1.02 | 1.02, (36) 1.03, (8) 1.035 (51) |
| 333.15 | 1.06 | 1.06, (36) 1.081, (51) 1.09 (44) | 0.89 | 0.900, (36) 0.901, (8) 0.909 (51) |
| MAPE = 0.9 | MAPE = 0.8 | |||
| n-pentylbenzene | n-butylbenzene | |||
| 288.15 | 1.45 | 1.46, (51) 1.487 (51) | 1.10 | 1.09, (51) 1.112, (51) 1.119 (51) |
| 293.15 | 1.33 | 1.32, (36) 1.334 (51) | 1.01 | 1.01, (36) 1.03, (51) 1.032, (51) 1.034, (52) 1.035, (51) 1.050 (51) |
| 298.15 | 1.22 | 1.21, (51) 1.225, (51) 1.25 (46) | 0.94 | 0.96 (51) |
| 303.15 | 1.13 | 1.12, (36) 1.126, (51) 1.132 (51) | 0.88 | 0.882, (36) 0.893 (52) 0.894, (51) 0.895, (51) 0.901, (48) 0.9035 (51) |
| 313.15 | 0.97 | 0.967, (36) 0.976 (51) | 0.78 | 0.774, (36) 0.787, (48) 0.781, (51) 0.79 (51) |
| 323.15 | 0.85 | 0.853 (36,51) | 0.70 | 0.684, (51) 0.686, (36) 0.7015 (51) |
| 333.15 | 0.75 | 0.754, (36) 0.756 (51) | MAPE = 2 | |
| MAPE = 0.7 | ||||
| n-propylbenzene | n-decylbenzene | |||
| 288.15 | 0.89 | 0.917, (51) 0.9181, (51) 0.9234 (51) | 4.38 | 4.31 (51) |
| 293.15 | 0.83 | 0.842, (36) 0.8545, (51) 0.855, (51) 0.8571, (51) 0.859 (51) | 3.84 | 3.80, (51) 3.81 (51) |
| 298.15 | 0.78 | 0.790, (51) 0.7966 (51) | 3.39 | 3.37 (51) |
| 303.15 | 0.74 | 0.722, (36) 0.7444, (51) 0.7452, (51) 0.7466 (51) | 3.02 | 3.02 (51) |
| 313.15 | MAPE = 2 | 2.44 | 2.46 (51) | |
| 323.15 | 2.03 | 2.00 (51) | ||
| MAPE = 0.9. | ||||
“Meas” is the measured value. Standard uncertainties u are u(T) = 0.01 K and u(p) = 0.001 MPa, and combined standard uncertainties uc are uc(η) = 0.02 mPa·s for all compounds except n-octylbenzene. The relative standard uncertainty is ur(η) = 0.018 for n-octylbenzene. The average pressure for these measurements was 0.102 MPa.
| T/K | meas.a η/mPa·s | literature η/mPa·s | meas.a η/mPa·s | literature η/mPa·s |
|---|---|---|---|---|
| pentadecane | n-octylbenzene | |||
| 288.15 | 1364.3 | 1364.0. (34) 1364.2 (16) | 1416.7 | 1415.9 (36) |
| 293.15 | 1345.3 | 1344.9, (34) 1345.2, (16) 1345.9 (37) | 1398.4 | 1397.3, (36) 1397.5 (8) |
| 298.15 | 1326.3 | 1325.8, (34) 1326.5 (16) | 1379.8 | 1378.7 (36) |
| 303.15 | 1307.5 | 1307.0, (34) 1307.3, (53) 1307.5, (16) 1308.1 (37) | 1361.5 | 1360.2, (36) 1361.0 (8) |
| 313.15 | 1270.8 | 1269.9, (53) 1270.1, (34) 1270.7, (16) 1270.9 (37) | 1325.5 | 1324.1, (36) 1324.9 (8) |
| 323.15 | 1234.7 | 1234.0, (34) 1234.1, (53) 1234.4, (16) 1234.5 (37) | 1290.1 | 1288.6, (36) 1289.4 (8) |
| 333.15 | 1199.2 | 1197.6, (53) 1198.3, (37) 1198.9, (34) 1199.1 (16) | 1255.1 | 1254.2, (36) 1254.3 (8) |
| MAPE = 0.03 | MAPE = 0.07 | |||
| n-heptylbenzene | n- hexylbenzene | |||
| 288.15 | 1405.8 | 1405.8 (36) | 1394.9 | 1395.0, (36) 1395.8, (33) 1395.6 (33) |
| 293.15 | 1387.1 | 1387.0 (36) | 1376.1 | 1375.9, (36) 1376.0, (33) 1376.1, (8) 1376.5 (33) |
| 298.15 | 1368.4 | 1368.2 (36) | 1357.0 | 1356.9 (36) |
| 303.15 | 1349.9 | 1349.6 (36) | 1338.3 | 1338.0, (36) 1338.1, (33) 1338.7 (8,33) |
| 313.15 | 1313.5 | 1313.0 (36) | 1301.4 | 1301.0, (36) 1301.1, (33) 1301.6, (33) 1301.7 (8) |
| 323.15 | 1277.7 | 1277.2 (36) | 1265.1 | 1264.7, (33) 1265.3 (8,33) |
| 333.15 | 1242.4 | 1242.1 (36) | 1229.2 | 1229.3, (36) 1229.4, (8,33) 1229.8 (33) |
| MAPE = 0.02 | MAPE = 0.02 | |||
| n-pentylbenzene | n-butylbenzene | |||
| 288.15 | 1385.4 | 1385.2 (36) | 1373.8 | 1373.7 (33) |
| 293.15 | 1366.0 | 1365.8 (36) | 1354.0 | 1353.2, (33) 1353.4, (54) 1353.8 (55) |
| 298.15 | 1346.7 | 1346.4, (36) 1346 (46) | 1334.3 | 1334.1 (55) |
| 303.15 | 1327.5 | 1327.1 (36) | 1314.8 | 1308, (48) 1314.3, (33,54) 1314.5 (55) |
| 313.15 | 1289.9 | 1289.4 (36) | 1276.5 | 1275.7, (54) 1276, (48) 1276.1 (33) |
| 323.15 | 1252.8 | 1252.3 (36) | 1238.7 | 1238.4 (33) |
| 333.15 | 1216.2 | 1216.2 (36) | 1201.5 | 1201.5, (33) 1201 (55) |
| MAPE = 0.03 | MAPE = 0.05 | |||
| n-propylbenzene | ethylbenzene | |||
| 288.15 | 1362.0 | 1361.3 (36,49) | 1361.3 | 1361.0 (36) |
| 293.15 | 1341.6 | 1340.7, (49) 1340.8, (36) 1341.5 (14) | 1340.2 | 1339.70, (26) 1339.9, (36) 1340 (56) |
| 298.15 | 1321.2 | 1320.6, (14) 1320.3, (36) 1321.0 (57) | 1319.1 | 1318.8 (36) |
| 303.15 | 1301.1 | 1300.0, (49) 1300.1, (36) 1300.4 (14) | 1298.2 | 1298.0, (36) 1298.05 (26) |
| 313.15 | 1261.5 | 1260.2, (36,49) 1260.9 (14) | 1257.1 | 1256, (58) 1256.89, (26) 1257, (56) 1257.2 (36) |
| 323.15 | 1222.4 | 1221.2, (36,49) 1221.514 | 1216.7 | 1216.35 (26) |
| 333.15 | 1183.9 | 1183.1 (36,49) | 1176.8 | 1176.40 (26) |
| MAPE = 0.07 | MAPE = 0.03 | |||
| toluene | ||||
| 288.15 | 1348.3 | 1348.2 (36) | ||
| 293.15 | 1326.6 | 1326.5, (36) 1326.8, (26) 1326.9 (59) | ||
| 298.15 | 1304.8 | 1304.7, (36) 1304.77 ± 0.10 (60) | ||
| 303.15 | 1283.2 | 1283, (61) 1283.20 ± 0.10 (60) | ||
| MAPE = 0.01 | ||||
“Meas.” is the measured value. The average pressure for these measurements was 0.102 MPa. Standard uncertainties are u(T) = 0.01 K and u(p) = 1 kPa, and relative standard uncertainties are ur(c) = 0.0007, except for n-octylbenzene mixtures where the relative standard uncertainties ur(c) = 0.0018.
3.2. Binary Mixtures: Densities, Speeds of Sound, and Viscosities
| n-alkyl-benzene | x1 | T = 288.15 K | T = 293.15 K | T = 298.15 K | T = 303.15 K | T = 313.15 K | T = 323.15 K | T = 333.15 K |
|---|---|---|---|---|---|---|---|---|
| 0.0000 | 772.23 | 768.72 | 765.22 | 761.73 | 754.74 | 747.75 | 740.75 | |
| toluene | 0.1017 | 775.91 | 772.38 | 768.84 | 765.30 | |||
| toluene | 0.1987 | 779.98 | 776.40 | 772.82 | 769.24 | |||
| toluene | 0.2985 | 784.81 | 781.18 | 777.55 | 773.92 | |||
| toluene | 0.3992 | 790.59 | 786.90 | 783.21 | 779.51 | |||
| toluene | 0.5014 | 797.62 | 793.85 | 790.08 | 786.30 | |||
| toluene | 0.5996 | 805.83 | 801.97 | 798.11 | 794.23 | |||
| toluene | 0.7000 | 816.26 | 812.29 | 808.30 | 804.30 | |||
| toluene | 0.8004 | 829.59 | 825.45 | 821.30 | 817.15 | |||
| toluene | 0.9010 | 847.26 | 842.92 | 838.56 | 834.19 | |||
| toluene | 1.0000 | 871.37 | 866.73 | 862.07 | 857.41 | |||
| ethyl- | 0.1000 | 776.41 | 772.88 | 769.35 | 765.82 | 758.77 | 751.70 | 744.63 |
| ethyl- | 0.2010 | 781.22 | 777.65 | 774.08 | 770.52 | 763.38 | 756.23 | 749.06 |
| ethyl- | 0.3002 | 786.66 | 783.05 | 779.44 | 775.82 | 768.59 | 761.34 | 754.06 |
| ethyl- | 0.3994 | 792.95 | 789.29 | 785.62 | 781.96 | 774.61 | 767.24 | 759.84 |
| ethyl- | 0.5011 | 800.52 | 796.80 | 793.07 | 789.34 | 781.86 | 774.34 | 766.79 |
| ethyl- | 0.6001 | 809.31 | 805.51 | 801.70 | 797.88 | 790.23 | 782.54 | 774.81 |
| ethyl- | 0.7002 | 820.05 | 816.15 | 812.25 | 808.33 | 800.48 | 792.58 | 784.63 |
| ethyl- | 0.7990 | 833.05 | 829.03 | 825.00 | 820.96 | 812.86 | 804.71 | 796.50 |
| ethyl- | 0.9003 | 849.88 | 845.70 | 841.51 | 837.31 | 828.88 | 820.40 | 811.85 |
| ethyl- | 1.0000 | 871.46 | 867.07 | 862.67 | 858.26 | 849.41 | 840.50 | 831.52 |
| propyl- | 0.1020 | 776.92 | 773.39 | 769.86 | 766.34 | 759.29 | 752.23 | 745.16 |
| propyl- | 0.2021 | 782.11 | 778.55 | 774.99 | 771.43 | 764.31 | 757.17 | 750.02 |
| propyl- | 0.3075 | 788.29 | 784.70 | 781.10 | 777.50 | 770.29 | 763.06 | 755.81 |
| propyl- | 0.4001 | 794.53 | 790.89 | 787.25 | 783.61 | 776.31 | 768.98 | 761.63 |
| propyl- | 0.4998 | 802.09 | 798.40 | 794.70 | 791.01 | 783.59 | 776.15 | 768.67 |
| propyl- | 0.6006 | 811.03 | 807.28 | 803.51 | 799.74 | 792.19 | 784.61 | 776.99 |
| propyl- | 0.6985 | 821.17 | 817.34 | 813.49 | 809.65 | 801.94 | 794.19 | 786.41 |
| propyl- | 0.8001 | 833.71 | 829.78 | 825.84 | 821.89 | 813.98 | 806.03 | 798.03 |
| propyl- | 0.8997 | 848.55 | 844.50 | 840.44 | 836.37 | 828.21 | 820.02 | 811.77 |
| propyl- | 1.0000 | 867.01 | 862.80 | 858.58 | 854.36 | 845.90 | 837.39 | 828.84 |
| butyl- | 0.1004 | 777.33 | 773.80 | 770.28 | 766.77 | 759.73 | 752.69 | 745.63 |
| butyl- | 0.2175 | 783.98 | 780.43 | 776.88 | 773.33 | 766.23 | 759.12 | 751.98 |
| butyl- | 0.3011 | 789.26 | 785.69 | 782.12 | 778.54 | 771.39 | 764.21 | 757.01 |
| butyl- | 0.4011 | 796.26 | 792.66 | 789.05 | 785.44 | 778.20 | 770.95 | 763.67 |
| butyl- | 0.5065 | 804.59 | 800.94 | 797.28 | 793.63 | 786.30 | 778.95 | 771.57 |
| butyl- | 0.5999 | 812.94 | 809.24 | 805.54 | 801.84 | 794.42 | 786.96 | 779.48 |
| butyl- | 0.7004 | 823.16 | 819.41 | 815.65 | 811.88 | 804.34 | 796.76 | 789.15 |
| butyl- | 0.7994 | 834.76 | 830.94 | 827.12 | 823.29 | 815.60 | 807.89 | 800.13 |
| butyl- | 0.8994 | 848.36 | 844.46 | 840.55 | 836.63 | 828.78 | 820.90 | 812.97 |
| butyl- | 1.0000 | 864.51 | 860.50 | 856.49 | 852.47 | 844.42 | 836.33 | 828.20 |
| pentyl- | 0.4999 | 805.85 | 802.23 | 798.60 | 794.98 | 787.71 | 780.42 | 773.11 |
| pentyl- | 1.000 | 862.81 | 858.92 | 855.02 | 851.13 | 843.31 | 835.48 | 827.60 |
| hexyl- | 0.000 | 772.24 | 768.73 | 765.23 | 761.74 | 754.75 | 747.77 | 740.77 |
| hexyl- | 0.1001 | 778.27 | 774.75 | 771.24 | 767.73 | 760.71 | 753.68 | 746.64 |
| hexyl- | 0.2002 | 784.79 | 781.26 | 777.73 | 774.20 | 767.14 | 760.07 | 752.98 |
| hexyl- | 0.3007 | 791.81 | 788.27 | 784.71 | 781.17 | 774.07 | 766.95 | 759.82 |
| hexyl- | 0.4011 | 799.43 | 795.86 | 792.29 | 788.72 | 781.57 | 774.40 | 767.21 |
| hexyl- | 0.5002 | 807.56 | 803.96 | 800.36 | 796.77 | 789.56 | 782.34 | 775.09 |
| hexyl- | 0.5997 | 816.42 | 812.80 | 809.17 | 805.54 | 798.28 | 791.00 | 783.68 |
| hexyl- | 0.6996 | 826.13 | 822.47 | 818.81 | 815.14 | 807.81 | 800.46 | 793.07 |
| hexyl- | 0.8003 | 836.77 | 833.07 | 829.37 | 825.67 | 818.26 | 810.83 | 803.37 |
| hexyl- | 0.8996 | 848.44 | 844.70 | 840.95 | 837.21 | 829.71 | 822.20 | 814.65 |
| hexyl- | 1.0000 | 861.39 | 857.60 | 853.81 | 850.02 | 842.43 | 834.81 | 827.17 |
| heptyl- | 0.4996 | 809.00 | 805.44 | 801.87 | 798.30 | 791.16 | 784.00 | 776.83 |
| heptyl- | 1.0000 | 860.04 | 856.35 | 852.65 | 848.96 | 841.55 | 834.14 | 826.69 |
| octyl- | 0.1013 | 779.4 | 775.9 | 772.4 | 768.9 | 761.9 | 754.9 | 747.8 |
| octyl- | 0.2002 | 786.7 | 783.2 | 779.7 | 776.2 | 769.2 | 762.1 | 755.1 |
| octyl- | 0.3589 | 799.2 | 795.7 | 792.1 | 788.6 | 781.6 | 774.5 | 767.4 |
| octyl- | 0.4554 | 807.2 | 803.7 | 800.2 | 796.6 | 789.5 | 782.4 | 775.3 |
| octyl- | 0.5010 | 811.2 | 807.6 | 804.1 | 800.5 | 793.4 | 786.3 | 779.2 |
| octyl- | 0.5999 | 820.0 | 816.4 | 812.9 | 809.3 | 802.2 | 795.1 | 787.9 |
| octyl- | 0.7000 | 829.4 | 825.8 | 822.3 | 818.7 | 811.5 | 804.4 | 797.2 |
| octyl- | 0.7998 | 839.2 | 835.7 | 832.1 | 828.5 | 821.3 | 814.1 | 806.9 |
| octyl- | 0.8990 | 849.5 | 845.9 | 842.3 | 838.7 | 831.5 | 824.3 | 817.0 |
| octyl- | 1.0000 | 860.6 | 857.0 | 853.4 | 849.7 | 842.5 | 835.2 | 827.9 |
| decyl- | 0.4987 | 813.58 | 810.07 | 806.57 | 803.06 | 796.06 | 789.05 | 782.03 |
| decyl- | 1.0000 | 859.01 | 855.49 | 851.97 | 848.45 | 841.42 | 834.38 | 827.33 |
x1 is the mole fraction of an n-alkylbenzene (1) in pentadecane (2). The average pressure for these measurements was 0.102 MPa. Standard uncertainties are u(T) = 0.01 K and u(p) = 1 kPa; relative standard uncertainties for mixtures with pentadecane are ur(ρ) = 0.0007 for n-decylbenzene, ur(ρ) = 0.0019 for n-octylbenzene mixtures, ur(ρ) = 0.0006 for n-heptylbenzene mixtures, ur(ρ) = 0.0008 for n-hexylbenzene mixtures, and ur(ρ) = 0.0003 for remaining mixtures, and combined expanded uncertainty U(x1) = 0.0001 (level of confidence = 0.95, k = 2).
| n-alkyl-benzene | x1 | T = 288.15 K | T = 293.15 K | T = 298.15 K | T = 303.15 K | T = 313.15 K | T = 323.15 K | T = 333.15 K |
|---|---|---|---|---|---|---|---|---|
| 0.0000 | 3.24 | 2.88 | 2.57 | 2.31 | ||||
| propyl- | 0.1020 | 2.88 | 2.57 | 2.31 | 2.10 | |||
| propyl- | 0.2021 | 2.56 | 2.30 | 2.09 | 1.90 | |||
| propyl- | 0.3075 | 2.25 | 2.05 | 1.86 | 1.70 | |||
| propyl- | 0.4001 | 2.02 | 1.84 | 1.67 | 1.53 | |||
| propyl- | 0.4998 | 1.78 | 1.63 | 1.49 | 1.37 | |||
| propyl- | 0.6006 | 1.56 | 1.43 | 1.31 | 1.21 | |||
| propyl- | 0.6985 | 1.36 | 1.25 | 1.15 | 1.07 | |||
| propyl- | 0.8001 | 1.18 | 1.09 | 1.01 | 0.94 | |||
| propyl- | 0.8997 | 1.02 | 0.95 | 0.89 | 0.83 | |||
| propyl- | 1.0000 | 0.89 | 0.83 | 0.78 | 0.74 | |||
| butyl- | 0.0000 | 3.24 | 2.88 | 2.57 | 2.31 | 1.90 | 1.59 | |
| butyl- | 0.1004 | 2.92 | 2.61 | 2.34 | 2.11 | 1.76 | 1.48 | |
| butyl- | 0.2175 | 2.59 | 2.32 | 2.11 | 1.91 | 1.60 | 1.36 | |
| butyl- | 0.3011 | 2.37 | 2.13 | 1.95 | 1.77 | 1.49 | 1.27 | |
| butyl- | 0.4011 | 2.12 | 1.94 | 1.76 | 1.61 | 1.37 | 1.17 | |
| butyl- | 0.5065 | 1.90 | 1.74 | 1.58 | 1.45 | 1.24 | 1.07 | |
| butyl- | 0.5999 | 1.72 | 1.57 | 1.44 | 1.33 | 1.14 | 0.99 | |
| butyl- | 0.7004 | 1.54 | 1.41 | 1.29 | 1.20 | 1.03 | 0.91 | |
| butyl- | 0.7994 | 1.38 | 1.26 | 1.17 | 1.08 | 0.94 | 0.84 | |
| butyl- | 0.8994 | 1.23 | 1.13 | 1.05 | 0.97 | 0.86 | 0.77 | |
| butyl- | 1.0000 | 1.10 | 1.01 | 0.94 | 0.88 | 0.78 | 0.70 | |
| pentyl- | 0.0000 | 3.24 | 2.88 | 2.57 | 2.31 | 1.90 | 1.59 | 1.35 |
| pentyl- | 0.4999 | 2.14 | 1.95 | 1.77 | 1.62 | 1.37 | 1.17 | 1.02 |
| pentyl- | 1.0000 | 1.45 | 1.33 | 1.22 | 1.13 | 0.97 | 0.85 | 0.75 |
| hexyl- | 0.000 | 3.24 | 2.88 | 2.57 | 2.31 | 1.90 | 1.59 | 1.35 |
| hexyl- | 0.1001 | 3.04 | 2.71 | 2.42 | 2.18 | 1.82 | 1.53 | 1.30 |
| hexyl- | 0.2002 | 2.85 | 2.55 | 2.28 | 2.08 | 1.73 | 1.46 | 1.25 |
| hexyl- | 0.3007 | 2.67 | 2.40 | 2.15 | 1.97 | 1.64 | 1.39 | 1.20 |
| hexyl- | 0.4011 | 2.51 | 2.25 | 2.05 | 1.86 | 1.56 | 1.33 | 1.15 |
| hexyl- | 0.5002 | 2.36 | 2.13 | 1.94 | 1.77 | 1.49 | 1.27 | 1.10 |
| hexyl- | 0.5997 | 2.23 | 2.03 | 1.84 | 1.68 | 1.41 | 1.21 | 1.05 |
| hexyl- | 0.6996 | 2.11 | 1.92 | 1.74 | 1.59 | 1.35 | 1.16 | 1.01 |
| hexyl- | 0.8003 | 2.02 | 1.83 | 1.66 | 1.52 | 1.29 | 1.11 | 0.97 |
| hexyl- | 0.8996 | 1.92 | 1.74 | 1.58 | 1.45 | 1.23 | 1.06 | 0.93 |
| hexyl- | 1.0000 | 1.83 | 1.67 | 1.52 | 1.39 | 1.18 | 1.02 | 0.89 |
| heptyl- | 0.000 | 3.24 | 2.88 | 2.57 | 2.31 | 1.90 | 1.59 | 1.35 |
| heptyl- | 0.4996 | 2.63 | 2.36 | 2.12 | 1.94 | 1.62 | 1.38 | 1.19 |
| heptyl- | 1.000 | 2.30 | 2.07 | 1.89 | 1.72 | 1.44 | 1.23 | 1.06 |
| octyl- | 0.0000 | 3.24 | 2.88 | 2.57 | 2.31 | 1.90 | 1.59 | 1.35 |
| octyl- | 0.1013 | 3.16 | 2.81 | 2.51 | 2.26 | 1.88 | 1.57 | 1.34 |
| octyl- | 0.2002 | 3.09 | 2.75 | 2.46 | 2.22 | 1.84 | 1.55 | 1.33 |
| octyl- | 0.3589 | 3.00 | 2.67 | 2.39 | 2.15 | 1.80 | 1.52 | 1.30 |
| octyl- | 0.4554 | 2.95 | 2.63 | 2.36 | 2.13 | 1.78 | 1.50 | 1.29 |
| octyl- | 0.5010 | 2.94 | 2.62 | 2.35 | 2.12 | 1.77 | 1.50 | 1.28 |
| octyl- | 0.5999 | 2.91 | 2.60 | 2.33 | 2.10 | 1.76 | 1.49 | 1.27 |
| octyl- | 0.7000 | 2.90 | 2.58 | 2.31 | 2.09 | 1.75 | 1.48 | 1.27 |
| octyl- | 0.7998 | 2.89 | 2.58 | 2.31 | 2.08 | 1.74 | 1.47 | 1.27 |
| octyl- | 0.8990 | 2.90 | 2.59 | 2.32 | 2.09 | 1.75 | 1.47 | 1.27 |
| octyl- | 1.0000 | 2.92 | 2.61 | 2.33 | 2.10 | 1.75 | 1.48 | 1.27 |
| decyl- | 0.4987 | 3.59 | 3.18 | 2.83 | 2.54 | 2.09 | 1.75 | |
| decyl- | 1.0000 | 4.38 | 3.84 | 3.39 | 3.02 | 2.44 | 2.03 |
x1 is the mole fraction of an n-alkylbenzene (1) in pentadecane (2). Standard uncertainties u are u(T) = 0.01 K and u(p) = 0.001 MPa, and combined standard uncertainties uc are uc(η) = 0.02 mPa·s for all mixtures except n-octylbenzene. The relative standard uncertainty is ur(η) = 0.018 for n-octylbenzene mixtures, and combined expanded uncertainty U(x1) = 0.0001 (level of confidence = 0.95, k = 2). The average pressure for these measurements was 0.102 MPa.
| n-alkyl-benzene | x1 | T = 288.15 K | T = 293.15 K | T = 298.15 K | T = 303.15 K | T = 313.15 K | T = 323.15 K | T = 333.15 K |
|---|---|---|---|---|---|---|---|---|
| 0.0000 | 1364.3 | 1345.3 | 1326.3 | 1307.5 | 1270.8 | 1234.7 | 1199.2 | |
| toluene | 0.1017 | 1359.8 | 1340.9 | 1321.8 | 1303.0 | |||
| toluene | 0.1987 | 1355.6 | 1336.5 | 1317.5 | 1298.6 | |||
| toluene | 0.2985 | 1351.1 | 1332.0 | 1312.9 | 1293.9 | |||
| toluene | 0.3992 | 1346.5 | 1327.3 | 1308.1 | 1289.1 | |||
| toluene | 0.5014 | 1342.1 | 1322.7 | 1303.3 | 1284.1 | |||
| toluene | 0.5996 | 1338.1 | 1318.6 | 1299.0 | 1279.6 | |||
| toluene | 0.7000 | 1334.9 | 1315.2 | 1295.4 | 1275.7 | |||
| toluene | 0.8004 | 1333.9 | 1313.8 | 1293.5 | 1273.5 | |||
| toluene | 0.9010 | 1336.9 | 1316.1 | 1295.3 | 1274.6 | |||
| toluene | 1.0000 | 1348.3 | 1326.6 | 1304.8 | 1283.2 | |||
| ethyl- | 0.1000 | 1360.6 | 1341.5 | 1322.6 | 1303.8 | 1267.0 | 1230.9 | 1195.2 |
| ethyl- | 0.2010 | 1357.0 | 1338.0 | 1319.0 | 1300.2 | 1263.3 | 1227.1 | 1191.4 |
| ethyl- | 0.3002 | 1353.6 | 1334.6 | 1315.5 | 1296.7 | 1259.6 | 1223.3 | 1187.4 |
| ethyl- | 0.3994 | 1350.5 | 1331.4 | 1312.2 | 1293.3 | 1256.1 | 1219.5 | 1183.5 |
| ethyl- | 0.5011 | 1347.5 | 1328.3 | 1309.1 | 1290.2 | 1252.8 | 1215.9 | 1179.6 |
| ethyl- | 0.6001 | 1345.6 | 1326.2 | 1306.8 | 1287.6 | 1249.9 | 1212.7 | 1176.0 |
| ethyl- | 0.7002 | 1344.6 | 1325.0 | 1305.4 | 1286.1 | 1247.9 | 1210.3 | 1173.1 |
| ethyl- | 0.7990 | 1345.9 | 1325.9 | 1306.0 | 1286.2 | 1247.4 | 1209.1 | 1171.4 |
| ethyl- | 0.9003 | 1350.2 | 1329.9 | 1309.5 | 1289.4 | 1249.8 | 1210.6 | 1172.0 |
| ethyl- | 1.000 | 1361.3 | 1340.2 | 1319.1 | 1298.2 | 1257.1 | 1216.7 | 1176.8 |
| propyl- | 0.1020 | 1361.1 | 1342.1 | 1323.1 | 1304.4 | 1267.6 | 1231.5 | 1195.9 |
| propyl- | 0.2021 | 1358.3 | 1339.3 | 1320.3 | 1301.6 | 1264.7 | 1228.5 | 1192.8 |
| propyl- | 0.3075 | 1355.4 | 1336.4 | 1317.5 | 1298.7 | 1261.8 | 1225.5 | 1189.7 |
| propyl- | 0.4001 | 1353.2 | 1334.2 | 1315.2 | 1296.4 | 1259.4 | 1223.0 | 1187.1 |
| propyl- | 0.4998 | 1351.2 | 1332.1 | 1313.0 | 1294.2 | 1257.0 | 1220.4 | 1184.3 |
| propyl- | 0.6006 | 1349.9 | 1330.7 | 1311.5 | 1292.5 | 1255.1 | 1218.2 | 1181.9 |
| propyl- | 0.6985 | 1349.5 | 1330.2 | 1310.8 | 1291.7 | 1254.0 | 1216.9 | 1180.2 |
| propyl- | 0.8001 | 1350.7 | 1331.1 | 1311.5 | 1292.2 | 1254.1 | 1216.5 | 1179.5 |
| propyl- | 0.8997 | 1354.4 | 1334.4 | 1314.5 | 1294.8 | 1256.0 | 1217.9 | 1180.3 |
| propyl- | 1.0000 | 1362.0 | 1341.6 | 1321.2 | 1301.1 | 1261.5 | 1222.4 | 1183.9 |
| butyl- | 0.1004 | 1362.5 | 1343.4 | 1324.4 | 1305.7 | 1268.9 | 1232.9 | 1197.3 |
| butyl- | 0.2175 | 1360.4 | 1341.6 | 1322.5 | 1303.9 | 1267.3 | 1231.2 | 1195.6 |
| butyl- | 0.3011 | 1359.3 | 1340.5 | 1321.5 | 1302.9 | 1266.2 | 1230.0 | 1194.4 |
| butyl- | 0.4011 | 1358.5 | 1339.5 | 1320.6 | 1301.9 | 1265.1 | 1228.9 | 1193.3 |
| butyl- | 0.5065 | 1357.9 | 1339.0 | 1320.1 | 1301.4 | 1264.6 | 1228.3 | 1192.5 |
| butyl- | 0.5999 | 1358.3 | 1339.4 | 1320.3 | 1301.6 | 1264.6 | 1228.2 | 1192.2 |
| butyl- | 0.7004 | 1359.5 | 1340.5 | 1321.4 | 1302.6 | 1265.4 | 1228.8 | 1192.6 |
| butyl- | 0.7994 | 1362.2 | 1342.9 | 1323.7 | 1304.6 | 1267.1 | 1230.3 | 1193.9 |
| butyl- | 0.8994 | 1366.4 | 1347.2 | 1327.6 | 1308.5 | 1270.7 | 1233.5 | 1196.8 |
| butyl- | 1.0000 | 1373.8 | 1354.0 | 1334.3 | 1314.8 | 1276.5 | 1238.7 | 1201.5 |
| pentyl- | 0.4999 | 1364.8 | 1345.8 | 1326.9 | 1308.2 | 1271.3 | 1235.2 | 1199.6 |
| pentyl- | 1.0000 | 1385.4 | 1366.0 | 1346.7 | 1327.5 | 1289.9 | 1252.8 | 1216.2 |
| hexyl- | 0.1001 | 1364.7 | 1345.7 | 1326.8 | 1308.1 | 1271.3 | 1235.3 | 1199.8 |
| hexyl- | 0.2002 | 1365.4 | 1346.5 | 1327.6 | 1309.0 | 1272.4 | 1236.4 | 1200.9 |
| hexyl- | 0.3007 | 1366.6 | 1347.8 | 1328.9 | 1310.4 | 1273.9 | 1237.9 | 1202.5 |
| hexyl- | 0.4011 | 1368.4 | 1349.5 | 1330.7 | 1312.1 | 1275.6 | 1239.7 | 1204.3 |
| hexyl- | 0.5002 | 1370.6 | 1351.8 | 1333.0 | 1314.4 | 1277.9 | 1241.9 | 1206.5 |
| hexyl- | 0.5997 | 1373.4 | 1354.7 | 1335.8 | 1317.3 | 1280.8 | 1244.8 | 1209.4 |
| hexyl- | 0.6996 | 1377.2 | 1358.3 | 1339.5 | 1320.9 | 1284.3 | 1248.3 | 1212.8 |
| hexyl- | 0.8003 | 1381.8 | 1363.0 | 1344.1 | 1325.5 | 1288.9 | 1252.8 | 1217.2 |
| hexyl- | 0.8996 | 1387.6 | 1368.8 | 1349.8 | 1331.1 | 1294.4 | 1258.2 | 1222.5 |
| hexyl- | 1.0000 | 1394.9 | 1376.1 | 1357.0 | 1338.3 | 1301.4 | 1265.1 | 1229.2 |
| heptyl- | 0.4996 | 1376.9 | 1358.3 | 1339.5 | 1321.0 | 1284.7 | 1249.0 | 1213.7 |
| heptyl- | 1.0000 | 1405.8 | 1387.1 | 1368.4 | 1349.9 | 1313.5 | 1277.7 | 1242.4 |
| octyl- | 0.0000 | 1364.3 | 1345.3 | 1326.3 | 1307.5 | 1270.8 | 1234.7 | 1199.2 |
| octyl- | 0.1013 | 1367.2 | 1348.5 | 1329.5 | 1310.9 | 1274.3 | 1238.3 | 1202.8 |
| octyl- | 0.2002 | 1370.7 | 1351.9 | 1333.0 | 1314.4 | 1277.9 | 1242.0 | 1206.7 |
| octyl- | 0.3589 | 1376.9 | 1358.3 | 1339.5 | 1321.0 | 1284.7 | 1249.0 | 1213.7 |
| octyl- | 0.4554 | 1381.2 | 1362.8 | 1344.1 | 1325.6 | 1289.4 | 1253.6 | 1218.5 |
| octyl- | 0.5010 | 1383.7 | 1364.9 | 1346.2 | 1327.8 | 1291.6 | 1256.0 | 1220.8 |
| octyl- | 0.5999 | 1388.8 | 1370.2 | 1351.6 | 1333.2 | 1297.1 | 1261.5 | 1226.4 |
| octyl- | 0.7000 | 1394.6 | 1376.3 | 1357.7 | 1339.3 | 1303.2 | 1267.7 | 1232.7 |
| octyl- | 0.7998 | 1401.3 | 1382.7 | 1364.1 | 1345.8 | 1309.8 | 1274.3 | 1239.3 |
| octyl- | 0.8990 | 1408.6 | 1390.0 | 1371.4 | 1353.1 | 1317.1 | 1281.7 | 1246.7 |
| octyl- | 1.0000 | 1416.7 | 1398.4 | 1379.8 | 1361.5 | 1325.5 | 1290.1 | 1255.1 |
| decyl- | 0.4987 | 1394.5 | 1375.9 | 1357.4 | 1339.1 | 1303.0 | 1267.7 | 1232.9 |
| decyl- | 1.0000 | 1433.5 | 1415.2 | 1396.9 | 1378.9 | 1343.5 | 1308.7 | 1274.4 |
x1 is the mole fraction of an n-alkylbenzene (1) in pentadecane (2). The average pressure for these measurements was 0.102 MPa. Standard uncertainties are u(T) = 0.01 K and u(p) = 1 kPa; relative standard uncertainties are ur(c) = 0.0018 for n-octylbenzene and ur(c) = 0.0007 for all other mixtures, and combined expanded uncertainty U(x1) = 0.0001 (level of confidence = 0.95, k = 2).
Figure 1
Figure 1. Speed of sound, c, of binary mixtures containing pentadecane and (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, and (E) ethylbenzene and (F) toluene at various temperatures. x1’s are values of the mole fraction of n-alkylbenzene in the pentadecane. The lines on the speed-of-sound figure are polynomial fits, with the fitting parameters provided in the Supporting Information, Table S4.
Figure 2
Figure 2. Bulk modulus of n-alkylbenzene in pentadecane at 293.15 K. x1 is the mole fraction of n-alkylbenzene in pentadecane.
Figure 3
Figure 3. Kinematic viscosities, ν, containing x1 mole fraction of an n-alkylbenzene in pentadecane: (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, at various temperatures. The symbols are the data, and the lines are the fitting results using eq 5. The fitting parameters are provided in the Supporting Information, Table S5. The temperature symbols shown in graph A apply to all the figures.
3.3. Excess Molar Properties and Viscosity Deviations
Figure 4
Figure 4. Redlich–Kister expressions for excess molar volume of an n-alkylbenzene, VmE, in pentadecane: (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, (E) ethylbenzene, and (F) toluene. Fitting parameters are given in the Supporting Information, Table S5. The temperature symbols shown in graph A apply to all the figures. x1 is the mole fraction of the n-alkylbenzene in pentadecane.
Figure 5
Figure 6
Figure 7
Figure 7. (A) Excess speeds of sound, cE, and (B) excess isentropic compressibilities, KsE, of mixtures of n-alkylbenzenes x1 in pentadecane at 293.15 K. Lines are fits to the Redlich Kistler equation, with the fitting parameters given in the Supporting Information. The aromatic compound symbols shown in graph A apply to graph B.
Figure 8
Figure 8. Comparison of excess speeds of sound, cE’s, and excess isentropic compressibilities, KsE’s, of equimolar mixtures of n-alkylbenzenes (closed symbols) in pentadecane with equimolar mixtures of n-alkylcyclohexanes (open symbols) in pentadecane at 293.15 K. (16,34)
4. Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jced.5c00217.
Relative differences Δρ/ρ = [ρ(expt) – ρ(lit.)]/ρ(expt) of the experimental densities of (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; relative differences Δc/c = [c(expt) – c(lit.)]/c(expt) of the experimental speeds of sound (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; relative differences Δη/η = [η(expt) – η(lit.)]/η(expt) of the experimental viscosities (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; methods of determining errors in mole fraction, density, viscosity, and speed of sound; polynomial fits to the speed of sound–mole fraction data for n-alkylbenzene (x1) in pentadecane mixtures; McAllister equation coefficients, ν12 and ν21 (eq S1), and associated standard error σ for an n-alkylbenzene x1 in pentadecane; excess molar volumes VmE (cm3·mol–1) of binary mixtures of an n-alkylbenzene at mole fraction x1 in pentadecane; uncertainty determination for VmE, Redlich–Kister equation parameters, and standard error, s, from the fitted excess molar volume of an n-alkylbenzene in pentadecane; viscosity deviations Δη’s (mPa·s) of binary mixtures of an n-alkylbenzene in pentadecane; uncertainty determination for Δη, excess speeds of sound cE’s, and excess isentropic compressibilities KsE’s of binary mixtures of an n-alkylbenzene (mole fraction of x1) in pentadecane; uncertainty determinations for cE and KsE, Redlich–Kister equation parameters and standard error, s, from fitted excess speeds of sound cE’s of an n-alkylbenzene in pentadecane at 293.15 K; Redlich–Kister equation parameters and standard error, s, from fitted excess isentropic compressibilities KsE’s of an n-alkylbenzene in pentadecane at 293.15 K (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This work was funded in part by an Office of Naval Research Grant, grant #N0001425GI00395.
References
This article references 79 other publications.
- 1Mansfield, A. B.; Chapman, E.; Briscoe, K. Effect of market variations in gasoline composition on aspects of stochastic pre-ignition. Fuel 2016, 184, 390– 400, DOI: 10.1016/j.fuel.2016.07.010Google ScholarThere is no corresponding record for this reference.
- 2Luning Prak, D.; Fohner, D.; Neubauer, J.; Dickerson, T.; Cowart, J.; Baker, B. The impact of navy jet fuel (JP-5) and diesel fuel (F-76) on the swelling and tensile strength of additivity-manufactured and commercially-manufactured O-rings. Fuel 2023, 354, 129291, DOI: 10.1016/j.fuel.2023.129291Google ScholarThere is no corresponding record for this reference.
- 3Westbrook, C. K.; Pitz, W. J.; Herbinet, O.; Curran, H. J.; Silke, E. J. A comprehensive detailed chemical kinetic reaction mechanism for combustion of n-alkane hydrocarbons from n-octane to n-hexadecane. Combust. Flame 2009, 156, 181– 199, DOI: 10.1016/j.combustflame.2008.07.014Google ScholarThere is no corresponding record for this reference.
- 4Sabourin, S. W.; Boteler, C. I.; Hallett, W. L. H. Droplet ignition of approximately continuous liquid mixtures of n-paraffins and n-alkyl aromatics. Combust. Flame 2016, 163, 326– 336, DOI: 10.1016/j.combustflame.2015.10.008Google ScholarThere is no corresponding record for this reference.
- 5Kim, D.; Martz, J.; Violi, A. Effects of fuel physical properties on direct injection spray and ignition behavior. Fuel 2016, 180, 481– 496, DOI: 10.1016/j.fuel.2016.03.085Google ScholarThere is no corresponding record for this reference.
- 6Luning Prak, D. J.; Lee, B. G.; Cowart, J. S.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of butylbenzene + linear alkanes (n-decane, n-dodecane, n-tetradecane, n-hexadecane, or n-heptadecane) at 0.1 MPa. J. Chem. Eng. Data 2017, 62, 169– 187, DOI: 10.1021/acs.jced.6b00542Google ScholarThere is no corresponding record for this reference.
- 7Luning Prak, D. J.; Cowart, J. S.; McDaniel, A. M.; Trulove, P. C. Correction to “Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexadecane + ethylbenzene or + toluene at (293.15 to 373.15) K and 0.1 MPa”. J. Chem. Eng. Data 2016, 61, 1021– 1022, DOI: 10.1021/acs.jced.5b01076Google ScholarThere is no corresponding record for this reference.
- 8Luning Prak, D. J.; Luning Prak, P. J.; Cowart, J. S.; Trulove, P. C. Densities and viscosities at 293.15–373.15 K, speeds of sound and bulk moduli at 293.15–333.15 K, surface tensions, and flash points of binary mixtures of n-hexadecane and alkylbenzenes at 0.1 MPa. J. Chem. Eng. Data 2017, 62, 1673– 1688, DOI: 10.1021/acs.jced.7b00087Google ScholarThere is no corresponding record for this reference.
- 9Asfour, A. F. A.; Siddique, M. H.; Vavanellos, T. D. Density-composition data for eight binary systems containing toluene or ethylbenzene and C8-C16 n-alkanes at 293.15, 298.15, 308.15, and 313.15 K. J. Chem. Eng. Data 1990, 35, 192– 198, DOI: 10.1021/je00060a030Google ScholarThere is no corresponding record for this reference.
- 10Luning Prak, D. J.; Harrison, J. A.; Morrow, B. H. Thermophysical Properties of Two-Component Mixtures of n-Nonylbenzene or 1,3,5-Triisopropylbenzene with n-Hexadecane or n-Dodecane at 0.1 MPa: Experimentally Measured Densities, Viscosities, and Speeds of Sound and Molecular Packing Modeled Using Molecular Dynamics Simulations. J. Chem. Eng. Data 2021, 66, 1442– 1456, DOI: 10.1021/acs.jced.0c01043Google ScholarThere is no corresponding record for this reference.
- 11Chevalier, J. L. E.; Petrino, P. J.; Gaston-Bonhomme, Y. H. Viscosity and density of some aliphatic, cyclic, and aromatic hydrocarbons binary liquid mixtures. J. Chem. Eng. Data 1990, 35, 206– 212, DOI: 10.1021/je00060a034Google ScholarThere is no corresponding record for this reference.
- 12Calvar, N.; Gómez, E.; González, B.; Domínguez, A. ́. Experimental densities, refractive indices, and speeds of sound of 12 binary mixtures containing alkanes and aromatic compounds at T = 313.15K. J. Chem. Thermodyn. 2009, 41, 939– 944, DOI: 10.1016/j.jct.2009.03.009Google ScholarThere is no corresponding record for this reference.
- 13Kanti, M.; Lagourette, B.; Alliez, J.; Boned, C. Viscosity of binary heptane─nonylbenzene as a function of pressure and temperature: application of Flory’s theory. Fluid Phase Equilib. 1991, 65, 291– 304, DOI: 10.1016/0378-3812(91)87031-4Google ScholarThere is no corresponding record for this reference.
- 14Sirbu, F.; Dragoescu, D.; Shchamialiou, A.; Khasanshin, T. Densities, speeds of sound, refractive indices, viscosities and their related thermodynamic properties for n-hexadecane + two aromatic hydrocarbons binary mixtures at temperatures from 298.15 to 318.15 K. J. Chem. Thermodyn. 2019, 128, 383– 393, DOI: 10.1016/j.jct.2018.08.036Google ScholarThere is no corresponding record for this reference.
- 15Rice, P.; Teja, A. Densities of n-hexane-aromatic hydrocarbon mixture. J. Chem. Eng. Data 1980, 25, 346– 347, DOI: 10.1021/je60087a006Google ScholarThere is no corresponding record for this reference.
- 16Luning Prak, D. J.; Cowart, J. S. n-Dodecylbenzene Binary Mixtures with Alkanes: Densities, Speeds of Sound, and Viscosities between 288.15 and 333.15 K and at 0.1 MPa. J. Chem. Eng. Data 2025, 70, 269– 282, DOI: 10.1021/acs.jced.4c00495Google ScholarThere is no corresponding record for this reference.
- 17Li, L.; Quan, K.; Xu, J.; Liu, F.; Liu, S.; Yu, S.; Xie, C.; Zhang, B.; Ge, X. Liquid Hydrocarbon Fuels from Catalytic Cracking of Waste Cooking Oils Using Basic Mesoporous Molecular Sieves K2O/Ba-MCM-41 as Catalysts. ACS Sustain. Chem. Eng. 2013, 1, 1412– 1416, DOI: 10.1021/sc4001548Google ScholarThere is no corresponding record for this reference.
- 18Simakova, I.; Simakova, O.; Mäki-Arvela, P.; Simakov, A.; Estrada, M.; Murzin, D. Y. Deoxygenation of palmitic and stearic acid over supported Pd catalysts: Effect of metal dispersion. Appl. Catal., A 2009, 355, 100– 108, DOI: 10.1016/j.apcata.2008.12.001Google ScholarThere is no corresponding record for this reference.
- 19Luning Prak, D. J.; Cowart, J. S.; Hamilton, L. J.; Hoang, D. T.; Brown, E. K.; Trulove, P. C. Development of a surrogate mixture for algal-based hydrotreated renewable diesel. Energy Fuels 2013, 27, 954– 961, DOI: 10.1021/ef301879gGoogle ScholarThere is no corresponding record for this reference.
- 20Wu, Z.; Mao, Y.; Raza, M.; Zhu, J.; Feng, Y.; Wang, S.; Qian, Y.; Yu, L.; Lu, X. Surrogate fuels for RP-3 kerosene formulated by emulating molecular structures, functional groups, physical and chemical properties. Combust. Flame 2019, 208, 388– 401, DOI: 10.1016/j.combustflame.2019.07.024Google ScholarThere is no corresponding record for this reference.
- 21Wood, C. P.; McDonell, V. G.; Smith, R. A.; Samuelsen, G. S. Development and application of a surrogate distillate fuel. J. Propul. Power 1989, 5, 399– 405, DOI: 10.2514/3.23168Google ScholarThere is no corresponding record for this reference.
- 22Edwards, T.; Maurice, L. Q. Surrogate mixtures to represent complex aviation and rocket fuels. J. Propul. Power 2001, 17, 461– 466, DOI: 10.2514/2.5765Google ScholarThere is no corresponding record for this reference.
- 23Detail Specification Turbine Fuel Aviation, Grade JP-5, MIL-DTL-5624X; Department of Defense: Washington, DC, February 12, 2024.Google ScholarThere is no corresponding record for this reference.
- 24United States Department of Defense, Naval Distillate; Military Specification MIL-PRF-16884N. Washington, DC, April 2014.Google ScholarThere is no corresponding record for this reference.
- 25ASTM_International ASTM D7566–24d, the Standard Specification for Aviation Turbine Fuel Containing Synthesized Hydrocarbons; ASTM International: West Conshohocken, 2024.Google ScholarThere is no corresponding record for this reference.
- 26Luning Prak, D. J.; Cowart, J. S.; McDaniel, A. M.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexadecane + ethylbenzene or + toluene at (293.15 to 373.15) K and 0.1 MPa. J. Chem. Eng. Data 2014, 59, 3571– 3585, DOI: 10.1021/je500498mGoogle ScholarThere is no corresponding record for this reference.
- 27Hoekman, S. K.; Robbins, C. Review of the effects of biodiesel on NOx emissions. Fuel Process. Technol. 2012, 96, 237– 249, DOI: 10.1016/j.fuproc.2011.12.036Google ScholarThere is no corresponding record for this reference.
- 28Tat, M. E.; Van Gerpen, J. H. Measurement of Biodiesel Speed of Sound and Its Impact on Injection Timing, NREL/SR-510-31462; National Renewable Energy Laboratory: Golden, Colorado, USA, 2003.Google ScholarThere is no corresponding record for this reference.
- 29Luning Prak, D. J.; Ye, S.; McLaughlin, M.; Cowart, J. S.; Trulove, P. C. Density, Viscosity, Speed of Sound, Bulk Modulus, Surface Tension, and Flash Point of Selected Ternary Mixtures of n-Butylcyclohexane + a Linear Alkane (n-Hexadcane or n-Dodecane) + an Aromatic Compound (Toluene, n-Butylbenzene, or n-Hexylbenzene). J. Chem. Eng. Data 2017, 62, 3452– 3472, DOI: 10.1021/acs.jced.7b00466Google ScholarThere is no corresponding record for this reference.
- 30Harris, D. C. Quantitative Chemical Analysis, 9th ed.; W.H. Freeman and Company: New York, 2010.Google ScholarThere is no corresponding record for this reference.
- 31Prak, D. J. L.; Morrow, B. H.; Maskey, S.; Harrison, J. A.; Cowart, J. S.; Trulove, P. C. Densities, speeds of sound, and viscosities of binary mixtures of an n-alkylcyclohexane (n-propyl-n-pentyl-n-hexyl-n-heptyl, n-octyl-n-nonyl-n-decyl-and n-dodecyl-) with n-hexadecane. J. Chem. Eng. Data 2018, 63, 4632– 4648, DOI: 10.1021/acs.jced.8b00692Google ScholarThere is no corresponding record for this reference.
- 32Luning Prak, D.; Cowart, J.; Simms, G. Physical Properties of Binary Mixtures of n-Dodecane and Various Ten-Carbon Aromatic Compounds (2-methyl-1-phenylpropane; 2-methyl-2-phenylpropane, 2-phenylbutane, and 1,3-diethylbenzene): Densities, Viscosities, Speeds of Sound, Bulk Moduli, Surface Tensions, and Flash Points at T = (293.15 - 333.15) K and 0.1 MPa. J. Chem. Eng. Data 2020, 65, 3941– 3954, DOI: 10.1021/acs.jced.0c00280Google ScholarThere is no corresponding record for this reference.
- 33Luning Prak, D. J.; Graft, S. L.; Johnson, T. R.; Cowart, J. S.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexylbenzene (1) or n-butylbenzene (1) in 2,2,4,6,6-pentamethylheptane (2) or 2,2,4,4,6,8,8-heptamethylnonane (2) at 0.1 MPa. J. Chem. Eng. Data 2018, 63, 3503– 3519, DOI: 10.1021/acs.jced.8b00387Google ScholarThere is no corresponding record for this reference.
- 34Prak, D. J. L. n-Pentadecane and n-Alkylcyclohexane Mixtures: Viscosity Deviations and Excess Molar Volumes, Speeds of Sound, and Isentropic Compressibilities at 0.1 MPa. J. Chem. Eng. Data 2023, 68, 2198– 2211, DOI: 10.1021/acs.jced.3c00290Google ScholarThere is no corresponding record for this reference.
- 35Wilhoit, R. C.; Marsh, K. N.; Hong, X.; Gadalla, N.; Frenkel, M. Densities of Aliphatic Hydrocarbons: Alkanes. Landolt-Börnstein, New Series; Springer-Verlag: Berlin, Germany, 1996; Vol. IV/8B.Google ScholarThere is no corresponding record for this reference.
- 36Prak, D. J. L. Binary Mixtures of Benzene and Cyclohexane with n-Alkyl Functional Groups up to 12 Carbons Long: Densities, Viscosities, and Speeds of Sound within the Temperature Range (288.15–333.15) K. J. Chem. Eng. Data 2022, 67, 1378– 1396, DOI: 10.1021/acs.jced.2c00048Google ScholarThere is no corresponding record for this reference.
- 37Daridon, J. L.; Carrier, H.; Lagourette, B. Pressure dependence of the thermophysical properties of n-pentadecane and n-heptadecane. Int. J. Thermophys. 2002, 23, 697– 708, DOI: 10.1023/A:1015451020209Google ScholarThere is no corresponding record for this reference.
- 38Wilhoit, R. C.; Hong, X.; Frenkel, M.; Hall, K. R. Densities of Aromatic Hydrocarbons 2 Alkylbenzenes: Datasheet from Landolt-Börnstein─Group IV Physical Chemistry; Springer-Verlag Berlin Heidelberg, 1996; Vol. 8E.Google ScholarThere is no corresponding record for this reference.
- 39Zhou, H.; Lagourette, B.; Alliez, J.; Xans, P.; Montel, F. Extension of the application of the Simha equation of state to the calculation of the density of n-alkanes-benzene and alkylbenzenes mixtures. Fluid Phase Equilib. 1990, 59, 309– 328, DOI: 10.1016/0378-3812(90)80006-WGoogle ScholarThere is no corresponding record for this reference.
- 40Masohan, A.; Nanoti, S. M.; Sharma, K. G.; Puri, S. N.; Gupta, P.; Rawat, B. S. Liquid-liquid equilibrium studies on hydrocarbon (C12-C20)-sulfolane systems. Fluid Phase Equilib. 1990, 61, 89– 98, DOI: 10.1016/0378-3812(90)90006-9Google ScholarThere is no corresponding record for this reference.
- 41Hall, R. K.; Marsh, K. N. Landolt-Börnstein: Numerical Data Functional Relationships in Science Technology: Thermodynamic Properties of Organic Compounds Their Mixtures. In Densities of Alkylbenzenes (C2H2n–6); Springer: Berlin, Germany, 1996; Vol. 8.Google ScholarThere is no corresponding record for this reference.
- 42Schmidt, A. W. Properties of aliphatic compounds. Ber. Dtsch. Chem. Ges. B 1942, 75B, 1399– 1424, DOI: 10.1002/cber.19420751121Google ScholarThere is no corresponding record for this reference.
- 43Aminabhavi, T. M.; Patil, V. B.; Aralaguppi, M. I.; Ortego, J. D.; Hansen, K. C. Density and Refractive Index of the Binary Mixtures of Cyclohexane with Dodecane, Tridecane, Tetradecane, and Pentadecane at (298.15, 303.15, and 308.15) K. J. Chem. Eng. Data 1996, 41, 526– 528, DOI: 10.1021/je950280bGoogle ScholarThere is no corresponding record for this reference.
- 44Baylaucq, A.; Zeberg-Mikkelsen, C. K.; Dauge, P.; Boned, C. Dynamic viscosity and density of heptylbenzene and heptylcyclohexane up to 100 MPa. J. Chem. Eng. Data 2002, 47, 997– 1002, DOI: 10.1021/je025511aGoogle ScholarThere is no corresponding record for this reference.
- 45Ju, T. Y.; Shen, G.; Wood, C. E. Synthesis and properties of non-normal-alkylbenzene. II. Preparation and properties of the intermediate ketones and corresponding hydrocarbons. J. Inst. Pet 1940, 26, 514– 531Google ScholarThere is no corresponding record for this reference.
- 46Van Hecke, G. R.; Baldwin, O. W. M.; Wada, B. C. Density, Viscosity, Refractive Index, Isobaric Specific Heat Capacity, Ultrasonic Velocity, Molar Volume, Isentropic Compressibility, Isothermal Compressibility, and Heat Capacity Ratio for Binary Mixtures of the Organic Liquids Ethylbenzene, Ethylcyclohexane, Pentylbenzene, and Pentylcyclohexane at 298.15 K and 0.1 MPa. J. Chem. Eng. Data 2022, 67, 1037– 1053, DOI: 10.1021/acs.jced.1c00649Google ScholarThere is no corresponding record for this reference.
- 47Al-Kandary, J. A.; Al-Jimaz, A. S.; Abdul-Latif, A.-H. M. Viscosities, densities, and speeds of sound of binary mixtures of benzene, toluene, o-xylene, m-xylene, p-xylene, and mesitylene with anisole at (288.15, 293.15, 298.15, and 303.15) K. J. Chem. Eng. Data 2006, 51, 2074– 2082, DOI: 10.1021/je060170cGoogle ScholarThere is no corresponding record for this reference.
- 48Rathnam, M. V.; Jain, K.; Kumar, M. S. Physical properties of binary mixtures of ethyl formate with benzene, isopropyl benzene, isobutyl benzene, and butylbenzene at (303.15, 308.15, and 313.15) K. J. Chem. Eng. Data 2010, 55, 1722– 1726, DOI: 10.1021/je900689mGoogle ScholarThere is no corresponding record for this reference.
- 49Prak, D. L.; Morrow, B. H.; Cowart, J. S.; Harrison, J. A. Binary mixtures of aromatic compounds (n-Propylbenzene, 1,3,5-trimethylbenzene, and 1,2,4-trimethylbenzene) with 2,2,4,6,6-pentamethylheptane: Densities, viscosities, speeds of sound, bulk moduli, surface tensions, and flash points at 0.1 MPa. J. Chem. Eng. Data 2020, 65, 2625– 2641, DOI: 10.1021/acs.jced.0c00020Google ScholarThere is no corresponding record for this reference.
- 50Bessieres, D.; Saint-guirons, H.; Daridon, J.-L. Thermodynamic Properties of liquid n-pentadecane. Phys. Chem. Liquid 2001, 39, 301– 313, DOI: 10.1080/00319100108031664Google ScholarThere is no corresponding record for this reference.
- 51Wohlfarth, C. Viscosity of Pure Organic Liquids and Binary Liquid Mixtures; Springer Science & Business Media: Berlin, Germany, 2002; Vol. 25.Google ScholarThere is no corresponding record for this reference.
- 52Al-Kandary, J. A.; Al-Jimaz, A. S.; Abdul-Latif, A.-H. M. Densities, viscosities, and refractive indices of binary mixtures of anisole with benzene, methylbenzene, ethylbenzene, propylbenzene, and butylbenzene at (293.15 and 303.15) K. J. Chem. Eng. Data 2006, 51, 99– 103, DOI: 10.1021/je0502546Google ScholarThere is no corresponding record for this reference.
- 53Khasanshin, T. S.; Shchemelev, A. P. Sound velocity in liquid n-alkanes. High Temp. 2001, 39, 60– 67, DOI: 10.1023/A:1004170530517Google ScholarThere is no corresponding record for this reference.
- 54Resa, J.; Gonzalez, C.; Concha, R.; Iglesias †, M. Ultrasonic velocity measurements for butyl acetate+ hydrocarbon mixtures. Phys. Chem. Liq. 2004, 42, 521– 543, DOI: 10.1080/00319100410001724548Google ScholarThere is no corresponding record for this reference.
- 55Luning Prak, D. J.; Fries, J. M.; Gober, R. T.; Vozka, P.; Kilaz, G.; Johnson, T. R.; Graft, S. L.; Trulove, P. C.; Cowart, J. S. Densities, viscosities, speeds of sound, bulk moduli, surface tensions, and flash points of quaternary mixtures of n-dodecane (1), n-butylcyclohexane (2), n-butylbenzene (3), and 2,2,4,4,6,8,8-heptamethylnonane (4) at 0.1 MPa as potential surrogate mixtures for military jet fuel, JP-5. J. Chem. Eng. Data 2019, 64, 1725– 1745, DOI: 10.1021/acs.jced.8b01233Google ScholarThere is no corresponding record for this reference.
- 56Bhatia, S. C.; Rani, R.; Bhatia, R. Densities, speeds of sound, and isentropic compressibilities of binary mixtures of {Alkan-1-ols + 1,2-dimethylbenzene, or 1,3-dimethylbenzene, or 1,4-dimethylbenzene, or ethylbenzene} at (293.15, 303.15, and 313.15) K. J. Chem. Eng. Data 2011, 56, 1675– 1681, DOI: 10.1021/je100734tGoogle ScholarThere is no corresponding record for this reference.
- 57Fujii, S.; Tamura, K.; Murakami, S. Thermodynamic properties of (an alkylbenzene + cyclohexane) at the temperature 298.15 K. J. Chem. Thermodyn. 1995, 27, 1319– 1328, DOI: 10.1006/jcht.1995.0140Google ScholarThere is no corresponding record for this reference.
- 58Calvar, N.; Gonzalez, G.; Gomex, E.; Canosa, J. Density, speed of sound, refreactive index for binary mixtures containing cycloalkanes and aromatic compounds at T = 313.15 K. J. Chem. Eng. Data 2009, 54, 1334– 1339, DOI: 10.1021/je8009336Google ScholarThere is no corresponding record for this reference.
- 59Fortin, T. J.; Laesecke, A.; Freund, M.; Outcalt, S. Advanced calibration, adjustment, and operation of a density and sound speed analyzer. J. Chem. Thermodyn. 2013, 57, 276– 285, DOI: 10.1016/j.jct.2012.09.009Google ScholarThere is no corresponding record for this reference.
- 60Tamura, K.; Murakami, S.; Doi, S. Speeds of sound, densities, and isentropic compressiilities of {xc-C6H12 + (1-x)C6H5CH3}, {xc-C6H11CH3 + (1-x)C6H6}, and {xc-C6H11CH3 + (1-x)C6H5CH3}, from 293.15 to 303.14 K. J. Chem. Thermodyn. 1985, 17, 325– 333, DOI: 10.1016/0021-9614(85)90129-6Google ScholarThere is no corresponding record for this reference.
- 61Oswal, S. L.; Maisuria, M. M.; Gardas, R. L. Speeds of sound, isentropic compressiblities, and excess molar voluems of cycloalkanes, alkanes, and aromatic hydrocarbons at 303.15 K. II. Results for cycloalkanes + aromatic hydrocarbons. J. Mol. Liq. 2004, 109, 155– 166, DOI: 10.1016/j.molliq.2003.06.005Google ScholarThere is no corresponding record for this reference.
- 62Morrow, B. H.; Maskey, S.; Gustafson, M. Z.; Luning Prak, D. J.; Harrison, J. A. Impact of molecular structure on properties of n-hexadecane and alkylbenzene binary mixtures. J. Phys. Chem. B 2018, 122, 6595– 6603, DOI: 10.1021/acs.jpcb.8b03752Google ScholarThere is no corresponding record for this reference.
- 63McAllister, R. A. The viscosity of liquid mixtures. AIChE J. 1960, 6, 427– 431, DOI: 10.1002/aic.690060316Google ScholarThere is no corresponding record for this reference.
- 64Luning Prak, D.; Cowart, J. Undecane and n-Alkylcyclohexanes Binary Mixtures: Densities and Speeds of Sound Between 288.15 and 333.15 K and Viscosities Between 293.15 and 323.15 K at 0.1 MPa. J. Chem. Eng. Data 2023, 68, 3208– 3221, DOI: 10.1021/acs.jced.3c00506Google ScholarThere is no corresponding record for this reference.
- 65Headen, T. F.; Howard, C. A.; Skipper, N. T.; Wilkinson, M. A.; Bowron, D. T.; Soper, A. K. Structure of π–π Interactions in Aromatic Liquids. J. Am. Chem. Soc. 2010, 132, 5735– 5742, DOI: 10.1021/ja909084eGoogle ScholarThere is no corresponding record for this reference.
- 66Falkowska, M.; Bowron, D. T.; Manyar, H. G.; Hardacre, C.; Youngs, T. G. A. Neutron Scattering of Aromatic and Aliphatic Liquids. ChemPhysChem 2016, 17, 2043– 2055, DOI: 10.1002/cphc.201600149Google ScholarThere is no corresponding record for this reference.
- 67Szala-Bilnik, J.; Falkowska, M.; Bowron, D. T.; Hardacre, C.; Youngs, T. G. A. The Structure of Ethylbenzene, Styrene and Phenylacetylene Determined by Total Neutron Scattering. ChemPhysChem 2017, 18, 2541– 2548, DOI: 10.1002/cphc.201700393Google ScholarThere is no corresponding record for this reference.
- 68Maskey, S.; Morrow, B. H.; Gustafson, M. Z.; Luning Prak, D. J.; Mikulski, P. T.; Harrison, J. A. Systematic examination of the links between composition and physical properties in surrogate fuel mixtures using molecular dynamics. Fuel 2020, 261, 116247, DOI: 10.1016/j.fuel.2019.116247Google ScholarThere is no corresponding record for this reference.
- 69Douheret, G.; Davis, M. I.; Reis, J. C.; Blandamer, M. J. Isentropic compressibilities--experimental origin and the quest for their rigorous estimation in thermodynamically ideal liquid mixtures. ChemPhysChem 2001, 2, 148, DOI: 10.1002/1439-7641(20010316)2:3<148::AID-CPHC148>3.0.CO;2-JGoogle ScholarThere is no corresponding record for this reference.
- 70Richards, W. T.; Wallace, J. H. The specific heats of five organic liquids from their adiabatic temperature-pressure coefficients. J. Am. Chem. Soc. 1932, 54, 2705– 2713, DOI: 10.1021/ja01346a013Google ScholarThere is no corresponding record for this reference.
- 71Huffman, H. M.; Parks, G. S.; Daniels, A. C. Thermal data on organic compounds. VII. The heat capacities, entropies, and free energies of twelve aromatic hydrocarbons. J. Am. Chem. Soc. 1930, 52, 1547– 1558, DOI: 10.1021/ja01367a039Google ScholarThere is no corresponding record for this reference.
- 72Messerly, J. F.; Todd, S. S.; Finke, H. L. Low-temperature thermodynamic properties of n-propyl- and n-butylbenzene. J. Phys. Chem. 1965, 69, 4304– 4311, DOI: 10.1021/j100782a038Google ScholarThere is no corresponding record for this reference.
- 73Páramo, R.; Zouine, M.; Sobrón, F.; Casanova, C. Saturated heat capacities of some linear and branched alkyl-benzenes between 288 and 348 K. Int. J. Thermophys. 2003, 24, 185– 199, DOI: 10.1023/A:1022318416775Google ScholarThere is no corresponding record for this reference.
- 74Finke, H. L.; Gross, M. E.; Waddington, G.; Huffman, H. M. Low-temperature thermal data for the nine normal paraffin hydrocarbons from octane to hexadecane. J. Am. Chem. Soc. 1954, 76, 333– 341, DOI: 10.1021/ja01631a005Google ScholarThere is no corresponding record for this reference.
- 75Yaws, C. L. Caculate liquid heat capacities. Hydrocarbon Process 1991, 12, 73– 77Google ScholarThere is no corresponding record for this reference.
- 76Luning Prak, D.; Cowart, J. Binary Mixtures of n-Alkylcyclohexanes with Decane: Densities and Speeds of Sound Between 288.15 and 333.15 K and Viscosities Between 293.15 and 323.15 K at 0.1 MPa. J. Chem. Eng. Data 2024, 69, 3029– 3043, DOI: 10.1021/acs.jced.4c00311Google ScholarThere is no corresponding record for this reference.
- 77Luning Prak, D. J.; Morrow, B. H.; Cowart, J. S.; Trulove, P. C.; Harrison, J. A. Thermophysical properties of binary mixtures of n-dodecane with n-alkylcyclohexanes: Experimental measurements and molecular dynamics simulations. J. Chem. Eng. Data 2019, 64, 1550– 1568, DOI: 10.1021/acs.jced.8b01135Google ScholarThere is no corresponding record for this reference.
- 78Luning Prak, D. J. Binary Mixtures of n-Tridecane with n-Alkylcyclohexanes: Density, Viscosity, and Speed of Sound within the Temperature Range (288.15 to 333.15) K. J. Chem. Eng. Data 2023, 68, 1053– 1066, DOI: 10.1021/acs.jced.2c00772Google ScholarThere is no corresponding record for this reference.
- 79Luning Prak, D. J. Densities, Viscosities, and Speeds of Sound of n-Tetradecane and n-Alkylcyclohexane Binary Mixtures within the Temperature Range (288.15 - 333.15) K. J. Chem. Eng. Data 2023, 68, 1610– 1623, DOI: 10.1021/acs.jced.3c00182Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 3 publications.
- Abhijith Vivek, Stefanía Betancur Marquez, Arno de Klerk. Viscosity and Density of C9–C10 Aromatics at 223.15–323.15 K Relevant to Aviation Turbine Fuel. Journal of Chemical & Engineering Data 2026, 71
(3)
, 960-971. https://doi.org/10.1021/acs.jced.5c00408
- Dianne J. Luning Prak, Jim S. Cowart. Undecane and n-Alkylbenzenes: Densities, Speeds of Sound, and Viscosities within the Range (288.15 to 333.15) K and at 0.1 MPa. Journal of Chemical & Engineering Data 2026, 71
(1)
, 72-86. https://doi.org/10.1021/acs.jced.5c00580
- Dianne J. Luning Prak, Jim S. Cowart. Decane and n-Alkylbenzene Binary Mixtures: Densities and Speeds of Sound within the Range of 288.15 and 333.15 K and Viscosities within the Range of 288.15 and 323.15 K at 0.1 MPa. Journal of Chemical & Engineering Data 2025, 70
(10)
, 4051-4065. https://doi.org/10.1021/acs.jced.5c00509
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1
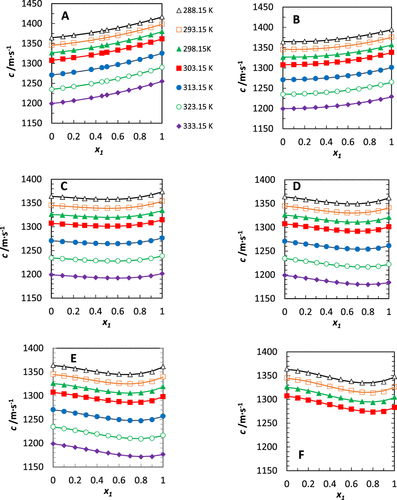
Figure 1. Speed of sound, c, of binary mixtures containing pentadecane and (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, and (E) ethylbenzene and (F) toluene at various temperatures. x1’s are values of the mole fraction of n-alkylbenzene in the pentadecane. The lines on the speed-of-sound figure are polynomial fits, with the fitting parameters provided in the Supporting Information, Table S4.
Figure 2
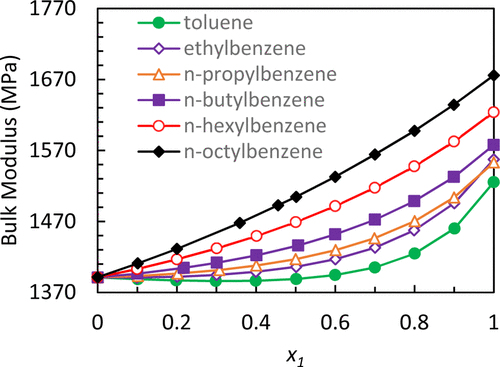
Figure 2. Bulk modulus of n-alkylbenzene in pentadecane at 293.15 K. x1 is the mole fraction of n-alkylbenzene in pentadecane.
Figure 3
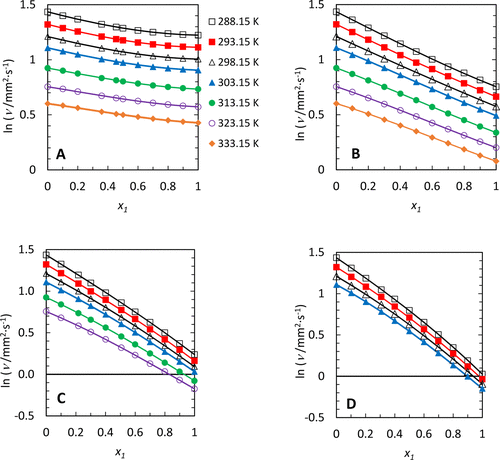
Figure 3. Kinematic viscosities, ν, containing x1 mole fraction of an n-alkylbenzene in pentadecane: (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, at various temperatures. The symbols are the data, and the lines are the fitting results using eq 5. The fitting parameters are provided in the Supporting Information, Table S5. The temperature symbols shown in graph A apply to all the figures.
Figure 4
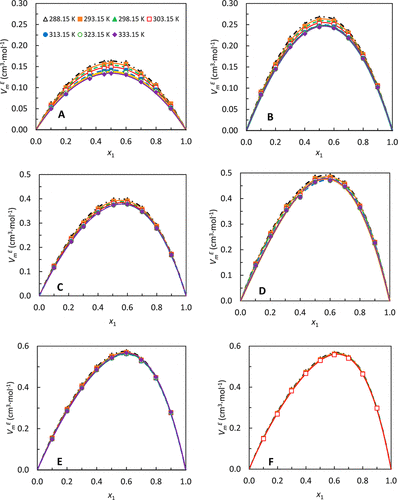
Figure 4. Redlich–Kister expressions for excess molar volume of an n-alkylbenzene, VmE, in pentadecane: (A) n-octylbenzene, (B) n-hexylbenzene, (C) n-butylbenzene, (D) n-propylbenzene, (E) ethylbenzene, and (F) toluene. Fitting parameters are given in the Supporting Information, Table S5. The temperature symbols shown in graph A apply to all the figures. x1 is the mole fraction of the n-alkylbenzene in pentadecane.
Figure 5
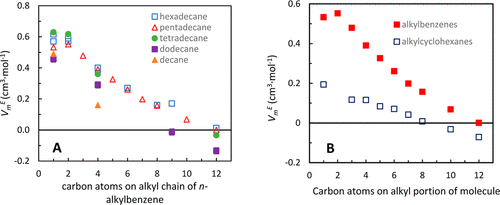
Figure 6
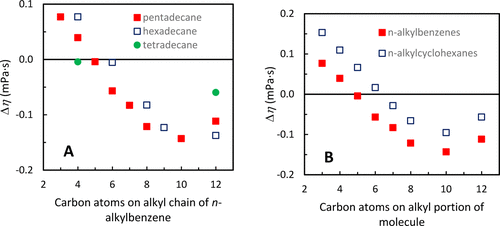
Figure 7
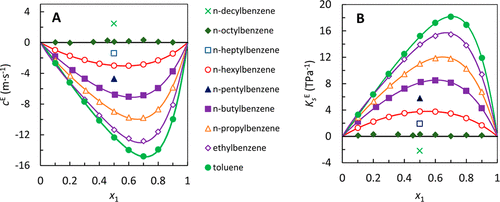
Figure 7. (A) Excess speeds of sound, cE, and (B) excess isentropic compressibilities, KsE, of mixtures of n-alkylbenzenes x1 in pentadecane at 293.15 K. Lines are fits to the Redlich Kistler equation, with the fitting parameters given in the Supporting Information. The aromatic compound symbols shown in graph A apply to graph B.
Figure 8
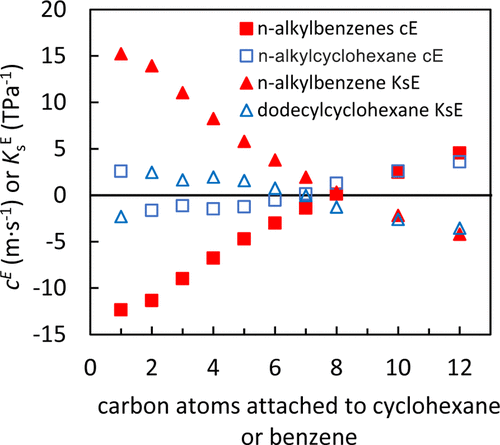
Figure 8. Comparison of excess speeds of sound, cE’s, and excess isentropic compressibilities, KsE’s, of equimolar mixtures of n-alkylbenzenes (closed symbols) in pentadecane with equimolar mixtures of n-alkylcyclohexanes (open symbols) in pentadecane at 293.15 K. (16,34)
References
This article references 79 other publications.
- 1Mansfield, A. B.; Chapman, E.; Briscoe, K. Effect of market variations in gasoline composition on aspects of stochastic pre-ignition. Fuel 2016, 184, 390– 400, DOI: 10.1016/j.fuel.2016.07.010There is no corresponding record for this reference.
- 2Luning Prak, D.; Fohner, D.; Neubauer, J.; Dickerson, T.; Cowart, J.; Baker, B. The impact of navy jet fuel (JP-5) and diesel fuel (F-76) on the swelling and tensile strength of additivity-manufactured and commercially-manufactured O-rings. Fuel 2023, 354, 129291, DOI: 10.1016/j.fuel.2023.129291There is no corresponding record for this reference.
- 3Westbrook, C. K.; Pitz, W. J.; Herbinet, O.; Curran, H. J.; Silke, E. J. A comprehensive detailed chemical kinetic reaction mechanism for combustion of n-alkane hydrocarbons from n-octane to n-hexadecane. Combust. Flame 2009, 156, 181– 199, DOI: 10.1016/j.combustflame.2008.07.014There is no corresponding record for this reference.
- 4Sabourin, S. W.; Boteler, C. I.; Hallett, W. L. H. Droplet ignition of approximately continuous liquid mixtures of n-paraffins and n-alkyl aromatics. Combust. Flame 2016, 163, 326– 336, DOI: 10.1016/j.combustflame.2015.10.008There is no corresponding record for this reference.
- 5Kim, D.; Martz, J.; Violi, A. Effects of fuel physical properties on direct injection spray and ignition behavior. Fuel 2016, 180, 481– 496, DOI: 10.1016/j.fuel.2016.03.085There is no corresponding record for this reference.
- 6Luning Prak, D. J.; Lee, B. G.; Cowart, J. S.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of butylbenzene + linear alkanes (n-decane, n-dodecane, n-tetradecane, n-hexadecane, or n-heptadecane) at 0.1 MPa. J. Chem. Eng. Data 2017, 62, 169– 187, DOI: 10.1021/acs.jced.6b00542There is no corresponding record for this reference.
- 7Luning Prak, D. J.; Cowart, J. S.; McDaniel, A. M.; Trulove, P. C. Correction to “Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexadecane + ethylbenzene or + toluene at (293.15 to 373.15) K and 0.1 MPa”. J. Chem. Eng. Data 2016, 61, 1021– 1022, DOI: 10.1021/acs.jced.5b01076There is no corresponding record for this reference.
- 8Luning Prak, D. J.; Luning Prak, P. J.; Cowart, J. S.; Trulove, P. C. Densities and viscosities at 293.15–373.15 K, speeds of sound and bulk moduli at 293.15–333.15 K, surface tensions, and flash points of binary mixtures of n-hexadecane and alkylbenzenes at 0.1 MPa. J. Chem. Eng. Data 2017, 62, 1673– 1688, DOI: 10.1021/acs.jced.7b00087There is no corresponding record for this reference.
- 9Asfour, A. F. A.; Siddique, M. H.; Vavanellos, T. D. Density-composition data for eight binary systems containing toluene or ethylbenzene and C8-C16 n-alkanes at 293.15, 298.15, 308.15, and 313.15 K. J. Chem. Eng. Data 1990, 35, 192– 198, DOI: 10.1021/je00060a030There is no corresponding record for this reference.
- 10Luning Prak, D. J.; Harrison, J. A.; Morrow, B. H. Thermophysical Properties of Two-Component Mixtures of n-Nonylbenzene or 1,3,5-Triisopropylbenzene with n-Hexadecane or n-Dodecane at 0.1 MPa: Experimentally Measured Densities, Viscosities, and Speeds of Sound and Molecular Packing Modeled Using Molecular Dynamics Simulations. J. Chem. Eng. Data 2021, 66, 1442– 1456, DOI: 10.1021/acs.jced.0c01043There is no corresponding record for this reference.
- 11Chevalier, J. L. E.; Petrino, P. J.; Gaston-Bonhomme, Y. H. Viscosity and density of some aliphatic, cyclic, and aromatic hydrocarbons binary liquid mixtures. J. Chem. Eng. Data 1990, 35, 206– 212, DOI: 10.1021/je00060a034There is no corresponding record for this reference.
- 12Calvar, N.; Gómez, E.; González, B.; Domínguez, A. ́. Experimental densities, refractive indices, and speeds of sound of 12 binary mixtures containing alkanes and aromatic compounds at T = 313.15K. J. Chem. Thermodyn. 2009, 41, 939– 944, DOI: 10.1016/j.jct.2009.03.009There is no corresponding record for this reference.
- 13Kanti, M.; Lagourette, B.; Alliez, J.; Boned, C. Viscosity of binary heptane─nonylbenzene as a function of pressure and temperature: application of Flory’s theory. Fluid Phase Equilib. 1991, 65, 291– 304, DOI: 10.1016/0378-3812(91)87031-4There is no corresponding record for this reference.
- 14Sirbu, F.; Dragoescu, D.; Shchamialiou, A.; Khasanshin, T. Densities, speeds of sound, refractive indices, viscosities and their related thermodynamic properties for n-hexadecane + two aromatic hydrocarbons binary mixtures at temperatures from 298.15 to 318.15 K. J. Chem. Thermodyn. 2019, 128, 383– 393, DOI: 10.1016/j.jct.2018.08.036There is no corresponding record for this reference.
- 15Rice, P.; Teja, A. Densities of n-hexane-aromatic hydrocarbon mixture. J. Chem. Eng. Data 1980, 25, 346– 347, DOI: 10.1021/je60087a006There is no corresponding record for this reference.
- 16Luning Prak, D. J.; Cowart, J. S. n-Dodecylbenzene Binary Mixtures with Alkanes: Densities, Speeds of Sound, and Viscosities between 288.15 and 333.15 K and at 0.1 MPa. J. Chem. Eng. Data 2025, 70, 269– 282, DOI: 10.1021/acs.jced.4c00495There is no corresponding record for this reference.
- 17Li, L.; Quan, K.; Xu, J.; Liu, F.; Liu, S.; Yu, S.; Xie, C.; Zhang, B.; Ge, X. Liquid Hydrocarbon Fuels from Catalytic Cracking of Waste Cooking Oils Using Basic Mesoporous Molecular Sieves K2O/Ba-MCM-41 as Catalysts. ACS Sustain. Chem. Eng. 2013, 1, 1412– 1416, DOI: 10.1021/sc4001548There is no corresponding record for this reference.
- 18Simakova, I.; Simakova, O.; Mäki-Arvela, P.; Simakov, A.; Estrada, M.; Murzin, D. Y. Deoxygenation of palmitic and stearic acid over supported Pd catalysts: Effect of metal dispersion. Appl. Catal., A 2009, 355, 100– 108, DOI: 10.1016/j.apcata.2008.12.001There is no corresponding record for this reference.
- 19Luning Prak, D. J.; Cowart, J. S.; Hamilton, L. J.; Hoang, D. T.; Brown, E. K.; Trulove, P. C. Development of a surrogate mixture for algal-based hydrotreated renewable diesel. Energy Fuels 2013, 27, 954– 961, DOI: 10.1021/ef301879gThere is no corresponding record for this reference.
- 20Wu, Z.; Mao, Y.; Raza, M.; Zhu, J.; Feng, Y.; Wang, S.; Qian, Y.; Yu, L.; Lu, X. Surrogate fuels for RP-3 kerosene formulated by emulating molecular structures, functional groups, physical and chemical properties. Combust. Flame 2019, 208, 388– 401, DOI: 10.1016/j.combustflame.2019.07.024There is no corresponding record for this reference.
- 21Wood, C. P.; McDonell, V. G.; Smith, R. A.; Samuelsen, G. S. Development and application of a surrogate distillate fuel. J. Propul. Power 1989, 5, 399– 405, DOI: 10.2514/3.23168There is no corresponding record for this reference.
- 22Edwards, T.; Maurice, L. Q. Surrogate mixtures to represent complex aviation and rocket fuels. J. Propul. Power 2001, 17, 461– 466, DOI: 10.2514/2.5765There is no corresponding record for this reference.
- 23Detail Specification Turbine Fuel Aviation, Grade JP-5, MIL-DTL-5624X; Department of Defense: Washington, DC, February 12, 2024.There is no corresponding record for this reference.
- 24United States Department of Defense, Naval Distillate; Military Specification MIL-PRF-16884N. Washington, DC, April 2014.There is no corresponding record for this reference.
- 25ASTM_International ASTM D7566–24d, the Standard Specification for Aviation Turbine Fuel Containing Synthesized Hydrocarbons; ASTM International: West Conshohocken, 2024.There is no corresponding record for this reference.
- 26Luning Prak, D. J.; Cowart, J. S.; McDaniel, A. M.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexadecane + ethylbenzene or + toluene at (293.15 to 373.15) K and 0.1 MPa. J. Chem. Eng. Data 2014, 59, 3571– 3585, DOI: 10.1021/je500498mThere is no corresponding record for this reference.
- 27Hoekman, S. K.; Robbins, C. Review of the effects of biodiesel on NOx emissions. Fuel Process. Technol. 2012, 96, 237– 249, DOI: 10.1016/j.fuproc.2011.12.036There is no corresponding record for this reference.
- 28Tat, M. E.; Van Gerpen, J. H. Measurement of Biodiesel Speed of Sound and Its Impact on Injection Timing, NREL/SR-510-31462; National Renewable Energy Laboratory: Golden, Colorado, USA, 2003.There is no corresponding record for this reference.
- 29Luning Prak, D. J.; Ye, S.; McLaughlin, M.; Cowart, J. S.; Trulove, P. C. Density, Viscosity, Speed of Sound, Bulk Modulus, Surface Tension, and Flash Point of Selected Ternary Mixtures of n-Butylcyclohexane + a Linear Alkane (n-Hexadcane or n-Dodecane) + an Aromatic Compound (Toluene, n-Butylbenzene, or n-Hexylbenzene). J. Chem. Eng. Data 2017, 62, 3452– 3472, DOI: 10.1021/acs.jced.7b00466There is no corresponding record for this reference.
- 30Harris, D. C. Quantitative Chemical Analysis, 9th ed.; W.H. Freeman and Company: New York, 2010.There is no corresponding record for this reference.
- 31Prak, D. J. L.; Morrow, B. H.; Maskey, S.; Harrison, J. A.; Cowart, J. S.; Trulove, P. C. Densities, speeds of sound, and viscosities of binary mixtures of an n-alkylcyclohexane (n-propyl-n-pentyl-n-hexyl-n-heptyl, n-octyl-n-nonyl-n-decyl-and n-dodecyl-) with n-hexadecane. J. Chem. Eng. Data 2018, 63, 4632– 4648, DOI: 10.1021/acs.jced.8b00692There is no corresponding record for this reference.
- 32Luning Prak, D.; Cowart, J.; Simms, G. Physical Properties of Binary Mixtures of n-Dodecane and Various Ten-Carbon Aromatic Compounds (2-methyl-1-phenylpropane; 2-methyl-2-phenylpropane, 2-phenylbutane, and 1,3-diethylbenzene): Densities, Viscosities, Speeds of Sound, Bulk Moduli, Surface Tensions, and Flash Points at T = (293.15 - 333.15) K and 0.1 MPa. J. Chem. Eng. Data 2020, 65, 3941– 3954, DOI: 10.1021/acs.jced.0c00280There is no corresponding record for this reference.
- 33Luning Prak, D. J.; Graft, S. L.; Johnson, T. R.; Cowart, J. S.; Trulove, P. C. Density, viscosity, speed of sound, bulk modulus, surface tension, and flash point of binary mixtures of n-hexylbenzene (1) or n-butylbenzene (1) in 2,2,4,6,6-pentamethylheptane (2) or 2,2,4,4,6,8,8-heptamethylnonane (2) at 0.1 MPa. J. Chem. Eng. Data 2018, 63, 3503– 3519, DOI: 10.1021/acs.jced.8b00387There is no corresponding record for this reference.
- 34Prak, D. J. L. n-Pentadecane and n-Alkylcyclohexane Mixtures: Viscosity Deviations and Excess Molar Volumes, Speeds of Sound, and Isentropic Compressibilities at 0.1 MPa. J. Chem. Eng. Data 2023, 68, 2198– 2211, DOI: 10.1021/acs.jced.3c00290There is no corresponding record for this reference.
- 35Wilhoit, R. C.; Marsh, K. N.; Hong, X.; Gadalla, N.; Frenkel, M. Densities of Aliphatic Hydrocarbons: Alkanes. Landolt-Börnstein, New Series; Springer-Verlag: Berlin, Germany, 1996; Vol. IV/8B.There is no corresponding record for this reference.
- 36Prak, D. J. L. Binary Mixtures of Benzene and Cyclohexane with n-Alkyl Functional Groups up to 12 Carbons Long: Densities, Viscosities, and Speeds of Sound within the Temperature Range (288.15–333.15) K. J. Chem. Eng. Data 2022, 67, 1378– 1396, DOI: 10.1021/acs.jced.2c00048There is no corresponding record for this reference.
- 37Daridon, J. L.; Carrier, H.; Lagourette, B. Pressure dependence of the thermophysical properties of n-pentadecane and n-heptadecane. Int. J. Thermophys. 2002, 23, 697– 708, DOI: 10.1023/A:1015451020209There is no corresponding record for this reference.
- 38Wilhoit, R. C.; Hong, X.; Frenkel, M.; Hall, K. R. Densities of Aromatic Hydrocarbons 2 Alkylbenzenes: Datasheet from Landolt-Börnstein─Group IV Physical Chemistry; Springer-Verlag Berlin Heidelberg, 1996; Vol. 8E.There is no corresponding record for this reference.
- 39Zhou, H.; Lagourette, B.; Alliez, J.; Xans, P.; Montel, F. Extension of the application of the Simha equation of state to the calculation of the density of n-alkanes-benzene and alkylbenzenes mixtures. Fluid Phase Equilib. 1990, 59, 309– 328, DOI: 10.1016/0378-3812(90)80006-WThere is no corresponding record for this reference.
- 40Masohan, A.; Nanoti, S. M.; Sharma, K. G.; Puri, S. N.; Gupta, P.; Rawat, B. S. Liquid-liquid equilibrium studies on hydrocarbon (C12-C20)-sulfolane systems. Fluid Phase Equilib. 1990, 61, 89– 98, DOI: 10.1016/0378-3812(90)90006-9There is no corresponding record for this reference.
- 41Hall, R. K.; Marsh, K. N. Landolt-Börnstein: Numerical Data Functional Relationships in Science Technology: Thermodynamic Properties of Organic Compounds Their Mixtures. In Densities of Alkylbenzenes (C2H2n–6); Springer: Berlin, Germany, 1996; Vol. 8.There is no corresponding record for this reference.
- 42Schmidt, A. W. Properties of aliphatic compounds. Ber. Dtsch. Chem. Ges. B 1942, 75B, 1399– 1424, DOI: 10.1002/cber.19420751121There is no corresponding record for this reference.
- 43Aminabhavi, T. M.; Patil, V. B.; Aralaguppi, M. I.; Ortego, J. D.; Hansen, K. C. Density and Refractive Index of the Binary Mixtures of Cyclohexane with Dodecane, Tridecane, Tetradecane, and Pentadecane at (298.15, 303.15, and 308.15) K. J. Chem. Eng. Data 1996, 41, 526– 528, DOI: 10.1021/je950280bThere is no corresponding record for this reference.
- 44Baylaucq, A.; Zeberg-Mikkelsen, C. K.; Dauge, P.; Boned, C. Dynamic viscosity and density of heptylbenzene and heptylcyclohexane up to 100 MPa. J. Chem. Eng. Data 2002, 47, 997– 1002, DOI: 10.1021/je025511aThere is no corresponding record for this reference.
- 45Ju, T. Y.; Shen, G.; Wood, C. E. Synthesis and properties of non-normal-alkylbenzene. II. Preparation and properties of the intermediate ketones and corresponding hydrocarbons. J. Inst. Pet 1940, 26, 514– 531There is no corresponding record for this reference.
- 46Van Hecke, G. R.; Baldwin, O. W. M.; Wada, B. C. Density, Viscosity, Refractive Index, Isobaric Specific Heat Capacity, Ultrasonic Velocity, Molar Volume, Isentropic Compressibility, Isothermal Compressibility, and Heat Capacity Ratio for Binary Mixtures of the Organic Liquids Ethylbenzene, Ethylcyclohexane, Pentylbenzene, and Pentylcyclohexane at 298.15 K and 0.1 MPa. J. Chem. Eng. Data 2022, 67, 1037– 1053, DOI: 10.1021/acs.jced.1c00649There is no corresponding record for this reference.
- 47Al-Kandary, J. A.; Al-Jimaz, A. S.; Abdul-Latif, A.-H. M. Viscosities, densities, and speeds of sound of binary mixtures of benzene, toluene, o-xylene, m-xylene, p-xylene, and mesitylene with anisole at (288.15, 293.15, 298.15, and 303.15) K. J. Chem. Eng. Data 2006, 51, 2074– 2082, DOI: 10.1021/je060170cThere is no corresponding record for this reference.
- 48Rathnam, M. V.; Jain, K.; Kumar, M. S. Physical properties of binary mixtures of ethyl formate with benzene, isopropyl benzene, isobutyl benzene, and butylbenzene at (303.15, 308.15, and 313.15) K. J. Chem. Eng. Data 2010, 55, 1722– 1726, DOI: 10.1021/je900689mThere is no corresponding record for this reference.
- 49Prak, D. L.; Morrow, B. H.; Cowart, J. S.; Harrison, J. A. Binary mixtures of aromatic compounds (n-Propylbenzene, 1,3,5-trimethylbenzene, and 1,2,4-trimethylbenzene) with 2,2,4,6,6-pentamethylheptane: Densities, viscosities, speeds of sound, bulk moduli, surface tensions, and flash points at 0.1 MPa. J. Chem. Eng. Data 2020, 65, 2625– 2641, DOI: 10.1021/acs.jced.0c00020There is no corresponding record for this reference.
- 50Bessieres, D.; Saint-guirons, H.; Daridon, J.-L. Thermodynamic Properties of liquid n-pentadecane. Phys. Chem. Liquid 2001, 39, 301– 313, DOI: 10.1080/00319100108031664There is no corresponding record for this reference.
- 51Wohlfarth, C. Viscosity of Pure Organic Liquids and Binary Liquid Mixtures; Springer Science & Business Media: Berlin, Germany, 2002; Vol. 25.There is no corresponding record for this reference.
- 52Al-Kandary, J. A.; Al-Jimaz, A. S.; Abdul-Latif, A.-H. M. Densities, viscosities, and refractive indices of binary mixtures of anisole with benzene, methylbenzene, ethylbenzene, propylbenzene, and butylbenzene at (293.15 and 303.15) K. J. Chem. Eng. Data 2006, 51, 99– 103, DOI: 10.1021/je0502546There is no corresponding record for this reference.
- 53Khasanshin, T. S.; Shchemelev, A. P. Sound velocity in liquid n-alkanes. High Temp. 2001, 39, 60– 67, DOI: 10.1023/A:1004170530517There is no corresponding record for this reference.
- 54Resa, J.; Gonzalez, C.; Concha, R.; Iglesias †, M. Ultrasonic velocity measurements for butyl acetate+ hydrocarbon mixtures. Phys. Chem. Liq. 2004, 42, 521– 543, DOI: 10.1080/00319100410001724548There is no corresponding record for this reference.
- 55Luning Prak, D. J.; Fries, J. M.; Gober, R. T.; Vozka, P.; Kilaz, G.; Johnson, T. R.; Graft, S. L.; Trulove, P. C.; Cowart, J. S. Densities, viscosities, speeds of sound, bulk moduli, surface tensions, and flash points of quaternary mixtures of n-dodecane (1), n-butylcyclohexane (2), n-butylbenzene (3), and 2,2,4,4,6,8,8-heptamethylnonane (4) at 0.1 MPa as potential surrogate mixtures for military jet fuel, JP-5. J. Chem. Eng. Data 2019, 64, 1725– 1745, DOI: 10.1021/acs.jced.8b01233There is no corresponding record for this reference.
- 56Bhatia, S. C.; Rani, R.; Bhatia, R. Densities, speeds of sound, and isentropic compressibilities of binary mixtures of {Alkan-1-ols + 1,2-dimethylbenzene, or 1,3-dimethylbenzene, or 1,4-dimethylbenzene, or ethylbenzene} at (293.15, 303.15, and 313.15) K. J. Chem. Eng. Data 2011, 56, 1675– 1681, DOI: 10.1021/je100734tThere is no corresponding record for this reference.
- 57Fujii, S.; Tamura, K.; Murakami, S. Thermodynamic properties of (an alkylbenzene + cyclohexane) at the temperature 298.15 K. J. Chem. Thermodyn. 1995, 27, 1319– 1328, DOI: 10.1006/jcht.1995.0140There is no corresponding record for this reference.
- 58Calvar, N.; Gonzalez, G.; Gomex, E.; Canosa, J. Density, speed of sound, refreactive index for binary mixtures containing cycloalkanes and aromatic compounds at T = 313.15 K. J. Chem. Eng. Data 2009, 54, 1334– 1339, DOI: 10.1021/je8009336There is no corresponding record for this reference.
- 59Fortin, T. J.; Laesecke, A.; Freund, M.; Outcalt, S. Advanced calibration, adjustment, and operation of a density and sound speed analyzer. J. Chem. Thermodyn. 2013, 57, 276– 285, DOI: 10.1016/j.jct.2012.09.009There is no corresponding record for this reference.
- 60Tamura, K.; Murakami, S.; Doi, S. Speeds of sound, densities, and isentropic compressiilities of {xc-C6H12 + (1-x)C6H5CH3}, {xc-C6H11CH3 + (1-x)C6H6}, and {xc-C6H11CH3 + (1-x)C6H5CH3}, from 293.15 to 303.14 K. J. Chem. Thermodyn. 1985, 17, 325– 333, DOI: 10.1016/0021-9614(85)90129-6There is no corresponding record for this reference.
- 61Oswal, S. L.; Maisuria, M. M.; Gardas, R. L. Speeds of sound, isentropic compressiblities, and excess molar voluems of cycloalkanes, alkanes, and aromatic hydrocarbons at 303.15 K. II. Results for cycloalkanes + aromatic hydrocarbons. J. Mol. Liq. 2004, 109, 155– 166, DOI: 10.1016/j.molliq.2003.06.005There is no corresponding record for this reference.
- 62Morrow, B. H.; Maskey, S.; Gustafson, M. Z.; Luning Prak, D. J.; Harrison, J. A. Impact of molecular structure on properties of n-hexadecane and alkylbenzene binary mixtures. J. Phys. Chem. B 2018, 122, 6595– 6603, DOI: 10.1021/acs.jpcb.8b03752There is no corresponding record for this reference.
- 63McAllister, R. A. The viscosity of liquid mixtures. AIChE J. 1960, 6, 427– 431, DOI: 10.1002/aic.690060316There is no corresponding record for this reference.
- 64Luning Prak, D.; Cowart, J. Undecane and n-Alkylcyclohexanes Binary Mixtures: Densities and Speeds of Sound Between 288.15 and 333.15 K and Viscosities Between 293.15 and 323.15 K at 0.1 MPa. J. Chem. Eng. Data 2023, 68, 3208– 3221, DOI: 10.1021/acs.jced.3c00506There is no corresponding record for this reference.
- 65Headen, T. F.; Howard, C. A.; Skipper, N. T.; Wilkinson, M. A.; Bowron, D. T.; Soper, A. K. Structure of π–π Interactions in Aromatic Liquids. J. Am. Chem. Soc. 2010, 132, 5735– 5742, DOI: 10.1021/ja909084eThere is no corresponding record for this reference.
- 66Falkowska, M.; Bowron, D. T.; Manyar, H. G.; Hardacre, C.; Youngs, T. G. A. Neutron Scattering of Aromatic and Aliphatic Liquids. ChemPhysChem 2016, 17, 2043– 2055, DOI: 10.1002/cphc.201600149There is no corresponding record for this reference.
- 67Szala-Bilnik, J.; Falkowska, M.; Bowron, D. T.; Hardacre, C.; Youngs, T. G. A. The Structure of Ethylbenzene, Styrene and Phenylacetylene Determined by Total Neutron Scattering. ChemPhysChem 2017, 18, 2541– 2548, DOI: 10.1002/cphc.201700393There is no corresponding record for this reference.
- 68Maskey, S.; Morrow, B. H.; Gustafson, M. Z.; Luning Prak, D. J.; Mikulski, P. T.; Harrison, J. A. Systematic examination of the links between composition and physical properties in surrogate fuel mixtures using molecular dynamics. Fuel 2020, 261, 116247, DOI: 10.1016/j.fuel.2019.116247There is no corresponding record for this reference.
- 69Douheret, G.; Davis, M. I.; Reis, J. C.; Blandamer, M. J. Isentropic compressibilities--experimental origin and the quest for their rigorous estimation in thermodynamically ideal liquid mixtures. ChemPhysChem 2001, 2, 148, DOI: 10.1002/1439-7641(20010316)2:3<148::AID-CPHC148>3.0.CO;2-JThere is no corresponding record for this reference.
- 70Richards, W. T.; Wallace, J. H. The specific heats of five organic liquids from their adiabatic temperature-pressure coefficients. J. Am. Chem. Soc. 1932, 54, 2705– 2713, DOI: 10.1021/ja01346a013There is no corresponding record for this reference.
- 71Huffman, H. M.; Parks, G. S.; Daniels, A. C. Thermal data on organic compounds. VII. The heat capacities, entropies, and free energies of twelve aromatic hydrocarbons. J. Am. Chem. Soc. 1930, 52, 1547– 1558, DOI: 10.1021/ja01367a039There is no corresponding record for this reference.
- 72Messerly, J. F.; Todd, S. S.; Finke, H. L. Low-temperature thermodynamic properties of n-propyl- and n-butylbenzene. J. Phys. Chem. 1965, 69, 4304– 4311, DOI: 10.1021/j100782a038There is no corresponding record for this reference.
- 73Páramo, R.; Zouine, M.; Sobrón, F.; Casanova, C. Saturated heat capacities of some linear and branched alkyl-benzenes between 288 and 348 K. Int. J. Thermophys. 2003, 24, 185– 199, DOI: 10.1023/A:1022318416775There is no corresponding record for this reference.
- 74Finke, H. L.; Gross, M. E.; Waddington, G.; Huffman, H. M. Low-temperature thermal data for the nine normal paraffin hydrocarbons from octane to hexadecane. J. Am. Chem. Soc. 1954, 76, 333– 341, DOI: 10.1021/ja01631a005There is no corresponding record for this reference.
- 75Yaws, C. L. Caculate liquid heat capacities. Hydrocarbon Process 1991, 12, 73– 77There is no corresponding record for this reference.
- 76Luning Prak, D.; Cowart, J. Binary Mixtures of n-Alkylcyclohexanes with Decane: Densities and Speeds of Sound Between 288.15 and 333.15 K and Viscosities Between 293.15 and 323.15 K at 0.1 MPa. J. Chem. Eng. Data 2024, 69, 3029– 3043, DOI: 10.1021/acs.jced.4c00311There is no corresponding record for this reference.
- 77Luning Prak, D. J.; Morrow, B. H.; Cowart, J. S.; Trulove, P. C.; Harrison, J. A. Thermophysical properties of binary mixtures of n-dodecane with n-alkylcyclohexanes: Experimental measurements and molecular dynamics simulations. J. Chem. Eng. Data 2019, 64, 1550– 1568, DOI: 10.1021/acs.jced.8b01135There is no corresponding record for this reference.
- 78Luning Prak, D. J. Binary Mixtures of n-Tridecane with n-Alkylcyclohexanes: Density, Viscosity, and Speed of Sound within the Temperature Range (288.15 to 333.15) K. J. Chem. Eng. Data 2023, 68, 1053– 1066, DOI: 10.1021/acs.jced.2c00772There is no corresponding record for this reference.
- 79Luning Prak, D. J. Densities, Viscosities, and Speeds of Sound of n-Tetradecane and n-Alkylcyclohexane Binary Mixtures within the Temperature Range (288.15 - 333.15) K. J. Chem. Eng. Data 2023, 68, 1610– 1623, DOI: 10.1021/acs.jced.3c00182There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jced.5c00217.
Relative differences Δρ/ρ = [ρ(expt) – ρ(lit.)]/ρ(expt) of the experimental densities of (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; relative differences Δc/c = [c(expt) – c(lit.)]/c(expt) of the experimental speeds of sound (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; relative differences Δη/η = [η(expt) – η(lit.)]/η(expt) of the experimental viscosities (expt) at p = 0.1 MPa from literature values (lit.) as a function of temperature T; methods of determining errors in mole fraction, density, viscosity, and speed of sound; polynomial fits to the speed of sound–mole fraction data for n-alkylbenzene (x1) in pentadecane mixtures; McAllister equation coefficients, ν12 and ν21 (eq S1), and associated standard error σ for an n-alkylbenzene x1 in pentadecane; excess molar volumes VmE (cm3·mol–1) of binary mixtures of an n-alkylbenzene at mole fraction x1 in pentadecane; uncertainty determination for VmE, Redlich–Kister equation parameters, and standard error, s, from the fitted excess molar volume of an n-alkylbenzene in pentadecane; viscosity deviations Δη’s (mPa·s) of binary mixtures of an n-alkylbenzene in pentadecane; uncertainty determination for Δη, excess speeds of sound cE’s, and excess isentropic compressibilities KsE’s of binary mixtures of an n-alkylbenzene (mole fraction of x1) in pentadecane; uncertainty determinations for cE and KsE, Redlich–Kister equation parameters and standard error, s, from fitted excess speeds of sound cE’s of an n-alkylbenzene in pentadecane at 293.15 K; Redlich–Kister equation parameters and standard error, s, from fitted excess isentropic compressibilities KsE’s of an n-alkylbenzene in pentadecane at 293.15 K (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



