Peptide-Based ROR1-Targeting PET Ligands for Melanoma Tumor Imaging: Design and Preclinical EvaluationClick to copy article linkArticle link copied!
- Donglan HuangDonglan HuangKey Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Donglan Huang
- Xingru LongXingru LongDepartment of Nuclear Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430022, P.R. ChinaHubei Key Laboratory of Molecular Imaging, Wuhan, Hubei 430022, P.R. ChinaKey Laboratory of Biological Targeted Therapy, The Ministry of Education, Wuhan, Hubei 430022, P.R. ChinaMore by Xingru Long
- Li ZhongLi ZhongCenter of Drug Discovery, State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P.R. ChinaMore by Li Zhong
- Yajing WangYajing WangCenter of Drug Discovery, State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P.R. ChinaMore by Yajing Wang
- Xuan DiXuan DiKey Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Xuan Di
- Zihan WangZihan WangKey Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Zihan Wang
- Shuhan ZhouShuhan ZhouKey Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Shuhan Zhou
- Xiaoyu DuXiaoyu DuKey Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Xiaoyu Du
- Yang Zhang*Yang Zhang*Email: [email protected]Center of Drug Discovery, State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P.R. ChinaMore by Yang Zhang
- Hai Qian*Hai Qian*Email: [email protected], [email protected]; Phone: +86-25-86185286; Fax: +86-25-83271050.Center of Drug Discovery, State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, P.R. ChinaJiangsu Key Laboratory of Drug Discovery for Metabolic Disease, China Pharmaceutical University, Nanjing 210009, P.R. ChinaMore by Hai Qian
- Dawei Jiang*Dawei Jiang*Email: [email protected]Department of Nuclear Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430022, P.R. ChinaHubei Key Laboratory of Molecular Imaging, Wuhan, Hubei 430022, P.R. ChinaKey Laboratory of Biological Targeted Therapy, The Ministry of Education, Wuhan, Hubei 430022, P.R. ChinaMore by Dawei Jiang
- Hualong Fu*Hualong Fu*Email: [email protected]Key Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, P.R. ChinaMore by Hualong Fu
Abstract
Receptor tyrosine kinase-like orphan receptor 1 (ROR1) is overexpressed in multiple cancers while remaining largely absent in adult tissues, which makes it an attractive target for both tumor diagnosis and therapy. To enable noninvasive imaging of ROR1, four peptide-based PET ligands [68Ga]1–4 were rationally designed and evaluated for melanoma imaging. In vitro assays confirmed reasonable ROR1 binding affinity (KD = 481.0 and 44.9 nM, respectively) and specific cellular uptake of [68Ga]2 and [68Ga]3, which are functionalized with serum albumin-binding groups. Notably, microPET/CT imaging and biodistribution studies in B16F10, A375, and SK-MEL-28 tumor-bearing mice demonstrated that [68Ga]2 achieved the most favorable imaging performance, characterized by high tumor accumulation (up to 9.18% ID/g), sustained retention, and a relatively lower nonspecific background signal. These findings highlight [68Ga]2 as a promising candidate for ROR1-targeting PET imaging and underscore the potential of peptide-based ROR1 PET probes for tumor imaging and therapy guidance.
This publication is licensed for personal use by The American Chemical Society.
Introduction
Figure 1
Figure 1. Ligand design and primary evaluations. (A) Schematic diagram and chemical structure of [68Ga]1. (B) MicroPET/CT images (left panel) and biodistribution results (right panel) in B16F10 tumor-bearing mice (n = 2–4) after intravenous injection of [68Ga]1. BL, bladder; K, kidneys; T, tumor.
Scheme 1
Results and Discussion
Ligand Design
Binding Potency
Figure 2
Figure 2. BLI binding curves of the interaction of newly developed ligands with the ROR1 protein. A–D correspond to PR3-DOTA, PR3-ABCF3-DOTA, PR3-C16-DOTA, and MCP-14-DOTA, respectively.
Radiolabeling and In Vitro Stability
| Radioligand | RCY | RCPa | Stability in salineb | Stability in mouse plasmab | Log D7.4 |
|---|---|---|---|---|---|
| [68Ga]1 | 94% (n = 3) | 93% | 90% (2 h) | 81% (2.5 h) | –2.84 ± 0.28 |
| [68Ga]2 | 85% (n = 3) | 95% | 95% (2 h) | 73% (2.5 h) | –2.76 ± 0.15 |
| [68Ga]3 | 70% (n = 3) | 96% | 96% (2 h) | 88% (2 h) | –1.93 ± 0.08 |
| [68Ga]4 | 61% (n = 2) | 96% | 95% (2 h) | 7% (2 h) | –0.93 ± 0.15 |
Determined upon the purification with a C18 cartridge.
In vitro stability was measured after incubation for 2 or 2.5 h, as indicated in the parentheses.
Cellular Uptake and Self-Blocking Assays
Figure 3
Figure 3. Cell uptake assay (A) and self-blocking assay (B) of [68Ga]1–4 in melanoma cell lines B16F10, A375, and SK-MEL-28; n.d., not determined. Asterisks indicate statistical significance: *p < 0.05, **p < 0.01, and ***p < 0.001 vs control. Data were analyzed using a two-tailed unpaired Student’s t-test.
MicroPET/CT Imaging of [68Ga]1–4 in B16F10 Tumor-Bearing Mice
Figure 4
Figure 4. (A, C) MicroPET/CT images of B16F10 tumor-bearing mice following the injection of [68Ga]2 (A) and [68Ga]3 (C), respectively. Self-blocking studies were performed by coinjecting the corresponding nonradioactive competitors, PR3-ABCF3-DOTA (0.25 mg/mouse) for [68Ga]2 and PR3-C16-DOTA (0.33 mg/mouse) for [68Ga]3. (B,D) The radioactivity uptake values of [68Ga]2 (B) and [68Ga]3 (D) are expressed as SUVmax in tumor and muscle. BL, bladder (red arrow); H, heart (black arrow); T, tumor (white arrow).
Ex Vivo Biodistribution Studies of [68Ga]2 and [68Ga]3 in B16F10 Tumor-Bearing Mice
Figure 5
Figure 5. Ex vivo biodistribution of [68Ga]2 (A) and [68Ga]3 (B) in B16F10 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as the percentage of injected dose per gram of tissue (% ID/g), with the exception of the stomach and small intestine, for which uptake is presented as %ID per organ (% ID/organ).
MicroPET/CT Imaging of [68Ga]1–4 and Ex Vivo Biodistribution Studies of [68Ga]2 in A375 Tumor-Bearing Mice
Figure 6
Figure 6. MicroPET/CT imaging and ex vivo biodistribution studies in A375 tumor-bearing mice. (A, B) MicroPET/CT images of A375 tumor-bearing mice following the injection of [68Ga]1–4. Representative images acquired at selected time points postinjection. [68Ga]1 (A) and [68Ga]2 (B), which achieved high T/M ratios, were shown in three time points of 0.5, 1, and 2 h, while [68Ga]3 and [68Ga]4 (B) that had lower T/M ratios were displayed at the 2 h time point only. (C) Quantitative analysis of tumor uptake for [68Ga]1–4 expressed as SUVmax. (D) Ex vivo biodistribution of [68Ga]2 in A375 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as % ID/g, with the exception of the stomach and small intestine, for which uptake is presented as % ID/organ. BL, bladder (red arrow); H, heart (purple arrow); L, liver (yellow arrow); and T, tumor (white arrow).
MicroPET/CT Imaging of [68Ga]1–3 and Ex Vivo Biodistribution Studies of [68Ga]2 in SK-MEL-28 Tumor-Bearing Mice
Figure 7
Figure 7. MicroPET/CT imaging and ex vivo biodistribution studies in SK-MEL-28 tumor-bearing mice. (A,B) MicroPET/CT images of SK-MEL-28 tumor-bearing mice following the injection of [68Ga]1–3. Representative images acquired at selected time points postinjection. [68Ga]1 (A) and [68Ga]2 (B), which achieved high T/M ratios, were shown in three time points of 0.5, 1, and 2 h, while [68Ga]3 (B) that had a lower T/M ratio was displayed at the 2 h time point only. (C) Quantitative analysis of tumor uptake for [68Ga]1–3 expressed as SUVmax. (D) Ex vivo biodistribution of [68Ga]2 in SK-MEL-28 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as % ID/g, with the exception of the stomach and small intestine, for which uptake is presented as % ID/organ. BL, bladder (red arrow); H, heart (purple arrow); L, liver (yellow arrow); T, tumor (white arrow).
Characterization of ROR1 Expression in Melanoma Models
Figure 8
Figure 8. Histopathological and immunohistochemical (IHC) characterization of B16F10, A375, and SK-MEL-28 melanoma tumors. Representative hematoxylin and eosin (HE)-stained sections (top panels) and corresponding IHC staining (middle panels) of paraffin-embedded tumor tissues. The lower panels show the quantitative analysis of protein expression levels based on histochemistry scores (H-scores). Data are expressed as mean ± SD (n = 10 fields per section). Scale bars: 150 μm.
Conclusion
Experimental Section
General Information
Chemistry and Radiochemistry
Binding Kinetics Assays
In Vitro Stability
Partition Coefficient
Cellular Uptake and Inhibition Assays
Tumor Model Establishment
MicroPET/CT Imaging
Ex Vivo Biodistribution Studies
IHC and HE Staining
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jmedchem.6c00570.
Synthetic routes and procedures of peptide-DOTA conjugates (PR3-DOTA, PR3-ABCF3-DOTA, PR3-C16-DOTA, and MCP-14-DOTA), purity of the peptide-DOTA conjugates with HPLC chromatograms, radiosynthetic routes of [68Ga]1–4, HPLC profiles of radioligands, in vitro stability, ex vivo biodistribution data of [68Ga]1–3 in tumor-bearing mice, ex vivo biodistribution data of [68Ga]2 in ICR mice, tumor and muscle SUVmax from PET imaging, PET images of radioligands in PET imaging studies, time-blood radioactivity curve of [68Ga]2, estimated absorbed radiation dosimetry for [68Ga]2 in human, and MS spectra of the peptide-DOTA conjugates (PDF)
Molecular formula strings data (CSV)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This research was supported by the National Natural Science Foundation of China (No. 22306014) and the Fundamental Research Funds for the Central Universities (No. 2233300007).
| ADC | antibody-drug conjugate |
| BiTE | bispecific T-cell engager |
| BLI | biolayer interferometry |
| CAR-T | chimeric-antigen receptor-modified T |
| CLL | chronic lymphocytic leukemia |
| CRD | cysteine-rich domain |
| DAB | 3,3′-diaminobenzidine |
| DOTA | 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid |
| FDA | Food and Drug Administration |
| GEP-NET | gastroenteropancreatic neuroendocrine tumor |
| HE | hematoxylin and eosin |
| HPLC | high-performance liquid chromatography |
| H-score | histochemistry score |
| IHC | immunohistochemical |
| Kr | Kringle |
| LC-MS | liquid chromatography–mass spectrometry |
| mAb | monoclonal antibodies |
| MCL | mantle cell lymphoma |
| OSEM | ordered-subset expectation maximization |
| PBS | phosphate-buffered saline |
| PDC | peptide-drug conjugate |
| PEG | polyethylene glycol |
| PET | positron emission tomography |
| RCP | radiochemical purity |
| RCY | radiochemical yield |
| RDC | radionuclide drug conjugate |
| ROR1 | receptor tyrosine kinase-like orphan receptor 1 |
| RTK | receptor tyrosine kinase |
| SUVmax | maximum standardized uptake value |
| TBR | tumor-to-background ratio |
| T/M | tumor-to-muscle |
| TNBC | triple-negative breast cancer |
References
This article references 63 other publications.
- 1Masiakowski, P.; Carroll, R. D. A novel family of cell surface receptors with tyrosine kinase-like domain. J. Biol. Chem. 1992, 267 (36), 26181– 26190, DOI: 10.1016/S0021-9258(18)35733-8Google ScholarThere is no corresponding record for this reference.
- 2Kamizaki, K.; Endo, M.; Minami, Y.; Kobayashi, Y. Role of noncanonical Wnt ligands and Ror-family receptor tyrosine kinases in the development, regeneration, and diseases of the musculoskeletal system. Dev. Dyn. 2021, 250 (1), 27– 38, DOI: 10.1002/dvdy.151Google ScholarThere is no corresponding record for this reference.
- 3Hasan, M. K.; Yu, J.; Widhopf, G. F., II; Rassenti, L. Z.; Chen, L.; Shen, Z.; Briggs, S. P.; Neuberg, D. S.; Kipps, T. J. Wnt5a induces ROR1 to recruit DOCK2 to activate Rac1/2 in chronic lymphocytic leukemia. Blood 2018, 132 (2), 170– 178, DOI: 10.1182/blood-2017-12-819383Google ScholarThere is no corresponding record for this reference.
- 4Karvonen, H.; Perttila, R.; Niininen, W.; Hautanen, V.; Barker, H.; Murumagi, A.; Heckman, C. A.; Ungureanu, D. Wnt5a and ROR1 activate non-canonical Wnt signaling via RhoA in TCF3-PBX1 acute lymphoblastic leukemia and highlight new treatment strategies via Bcl-2 co-targeting. Oncogene 2019, 38 (17), 3288– 3300, DOI: 10.1038/s41388-018-0670-9Google ScholarThere is no corresponding record for this reference.
- 5Li, L.; Huang, W.; Ren, X.; Wang, Z.; Ding, K.; Zhao, L.; Zhang, J. Unlocking the potential: Advancements and future horizons in ROR1-targeted cancer therapies. SCI. CHINA Life Sci. 2024, 67 (12), 2603– 2616, DOI: 10.1007/s11427-024-2685-9Google ScholarThere is no corresponding record for this reference.
- 6Borcherding, N.; Kusner, D.; Liu, G.-H.; Zhang, W. ROR1, an embryonic protein with an emerging role in cancer biology. Protein Cell 2014, 5 (7), 496– 502, DOI: 10.1007/s13238-014-0059-7Google ScholarThere is no corresponding record for this reference.
- 7Balakrishnan, A.; Goodpaster, T.; Randolph-Habecker, J.; Hoffstrom, B. G.; Jalikis, F. G.; Koch, L. K.; Berger, C.; Kosasih, P. L.; Rajan, A.; Sommermeyer, D.; Porter, P. L.; Riddell, S. R. Analysis of ROR1 protein expression in human cancer and normal tissues. Clin. Cancer Res. 2017, 23 (12), 3061– 3071, DOI: 10.1158/1078-0432.CCR-16-2083Google ScholarThere is no corresponding record for this reference.
- 8Zhao, Y.; Zhang, D.; Guo, Y.; Lu, B.; Zhao, Z. J.; Xu, X.; Chen, Y. Tyrosine kinase ROR1 as a target for anti-cancer therapies. Front. Oncol. 2021, 11 (2021), 680834, DOI: 10.3389/fonc.2021.680834Google ScholarThere is no corresponding record for this reference.
- 9Raso, M. G.; Barrientos Toro, E.; Evans, K.; Rizvi, Y.; Lazcano, R.; Akcakanat, A.; Sini, P.; Trapani, F.; Madlener, E. J.; Waldmeier, L.; Lazar, A.; Meric-Bernstam, F. Heterogeneous profile of ROR1 protein expression across tumor types. Cancers 2024, 16 (10), 1874, DOI: 10.3390/cancers16101874Google ScholarThere is no corresponding record for this reference.
- 10Fernandez, N. B.; Lorenzo, D.; Picco, M. E.; Barbero, G.; Dergan-Dylon, L. S.; Marks, M. P.; Garcia-Rivello, H.; Gimenez, L.; Labovsky, V.; Grumolato, L.; Lopez-Bergami, P. ROR1 contributes to melanoma cell growth and migration by regulating N-cadherin expression via the PI3K/Akt pathway. Mol. Carcinog. 2016, 55 (11), 1772– 1785, DOI: 10.1002/mc.22426Google ScholarThere is no corresponding record for this reference.
- 11Quezada, M. J.; Lopez-Bergami, P. The signaling pathways activated by ROR1 in cancer. Cell. Signal. 2023, 104, 110588, DOI: 10.1016/j.cellsig.2023.110588Google ScholarThere is no corresponding record for this reference.
- 12Piki, E.; Dini, A.; Raivola, J.; Salokas, K.; Zhang, K.; Varjosalo, M.; Pellinen, T.; Valimaki, K.; Veskimae, K. T.; Staff, S. ROR1-STAT3 signaling contributes to ovarian cancer intra-tumor heterogeneity. Cell Death Discovery 2023, 9 (1), 222, DOI: 10.1038/s41420-023-01527-6Google ScholarThere is no corresponding record for this reference.
- 13Li, C.; Wang, S.; Xing, Z.; Lin, A.; Liang, K.; Song, J.; Hu, Q.; Yao, J.; Chen, Z.; Park, P. K.; Hawke, D. H.; Zhou, J.; Zhou, Y.; Zhang, S.; Liang, H.; Hung, M. C.; Gallick, G. E.; Han, L.; Lin, C.; Yang, L. A ROR1-HER3-lncRNA signalling axis modulates the Hippo-YAP pathway to regulate bone metastasis. Nat. Cell Biol. 2017, 19 (2), 106– 119, DOI: 10.1038/ncb3464Google ScholarThere is no corresponding record for this reference.
- 14Fukuda, T.; Chen, L.; Endo, T.; Tang, L.; Lu, D.; Castro, J. E.; Widhopf, G. F.; Rassenti, L. Z.; Cantwell, M. J.; Prussak, C. E.; Carson, D. A.; Kipps, T. J. Antisera induced by infusions of autologous Ad-CD154-leukemia B cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a. Proc. Natl. Acad. Sci. U. S. A. 2008, 105 (8), 3047– 3052, DOI: 10.1073/pnas.0712148105Google ScholarThere is no corresponding record for this reference.
- 15Gohil, S. H.; Paredes-Moscosso, S. R.; Harrasser, M.; Vezzalini, M.; Scarpa, A.; Morris, E.; Davidoff, A. M.; Sorio, C.; Nathwani, A. C.; Della Peruta, M. An ROR1 bi-specific T-cell engager provides effective targeting and cytotoxicity against a range of solid tumors. OncoImmunology 2017, 6 (7), e1326437 DOI: 10.1080/2162402X.2017.1326437Google ScholarThere is no corresponding record for this reference.
- 16Wu, D.; Gong, S.; Wu, X.; Naren, G.; Dong, L.; Lensky, S.; Wu, C. Abstract LB126: Generation of a novel ROR1 x CD3 bispecific T-cell engager for better tumor killing and minimal cytokine release. Cancer Res. 2024, 84 (7_Supplement), LB126, DOI: 10.1158/1538-7445.AM2024-LB126Google ScholarThere is no corresponding record for this reference.
- 17Townsend, W.; Leong, S.; Tucker, D.; Pottinger, B.; Paneesha, S.; El-Sharkawi, D.; Eyre, T. A.; Batten, T.; Shah, M.; Cook, S. First-in-human phase I trial of a ROR1 targeting bispecific T cell engager (NVG-111) in combination with ibrutinib or as monotherapy in subjects with relapsed refractory chronic lymphocytic leukaemia (CLL) and mantle cell lymphoma (MCL). Blood 2022, 140 (Supplement 1), 4162– 4163, DOI: 10.1182/blood-2022-169658Google ScholarThere is no corresponding record for this reference.
- 18Zhang, Y.; Fan, Y.; Liu, S.; Guan, Y.; Wan, J.; Ren, Q.; Wang, J.; Zhong, L.; Hu, Z.; Shi, W.; Qian, H. Development of peptide paratope mimics derived from the anti-ROR1 antibody and long-acting peptide-drug conjugates for targeted cancer therapy. J. Med. Chem. 2024, 67 (13), 10967– 10985, DOI: 10.1021/acs.jmedchem.4c00511Google ScholarThere is no corresponding record for this reference.
- 19Hojjat-Farsangi, M.; Daneshmanesh, A. H.; Khan, A. S.; Shetye, J.; Mozaffari, F.; Kharaziha, P.; Rathje, L. S.; Kokhaei, P.; Hansson, L.; Vågberg, J.; Byström, S.; Olsson, E.; Löfberg, C.; Norström, C.; Schultz, J.; Norin, M.; Olin, T.; Österborg, A.; Mellstedt, H.; Moshfegh, A. First-in-class oral small molecule inhibitor of the tyrosine kinase ROR1 (KAN0439834) induced significant apoptosis of chronic lymphocytic leukemia cells. Leukemia 2018, 32 (10), 2291– 2295, DOI: 10.1038/s41375-018-0113-1Google ScholarThere is no corresponding record for this reference.
- 20Liu, X.; Pu, W.; He, H.; Fan, X.; Zheng, Y.; Zhou, J.-K.; Ma, R.; He, J.; Zheng, Y.; Wu, K.; Zhao, Y.; Yang, S.-Y.; Wang, C.; Wei, Y.-Q.; Wei, X.-W.; Peng, Y. Novel ROR1 inhibitor ARI-1 suppresses the development of non-small cell lung cancer. Cancer Lett. 2019, 458, 76– 85, DOI: 10.1016/j.canlet.2019.05.016Google ScholarThere is no corresponding record for this reference.
- 21Fan, Y.; Zhang, F.; Xiong, L.; Su, M.; Luo, F.; Li, M.; Li, Q.; Zhong, T.; Yuan, M.; Xu, Y.; Mu, S.; Yang, H. Design, synthesis, and biological evaluation of 6-(imidazo[1,2-a] pyridin-6-yl) quinazolin-4(3H)-one derivatives as potent anticancer agents by dual targeting Aurora kinase and ROR1. Bioorg. Chem. 2023, 135, 106484, DOI: 10.1016/j.bioorg.2023.106484Google ScholarThere is no corresponding record for this reference.
- 22Luo, D.; Qiu, X.; Zheng, Q.; Ming, Y.; Pu, W.; Ai, M.; He, J.; Peng, Y. Discovery of novel receptor tyrosine kinase-like orphan receptor 1 (ROR1) inhibitors for cancer treatment. J. Med. Chem. 2024, 67 (13), 10655– 10686, DOI: 10.1021/acs.jmedchem.4c00175Google ScholarThere is no corresponding record for this reference.
- 23Ta, T. M.; Reed, V. L.; Gupta, S. R. R.; Khan, M.; Chandra, N.; Dwyer, N.; Fultang, N.; Singh, I. K.; Peethambaran, B. Novel macromolecule CPD4 suppresses cell proliferation and metastasis of triple-negative breast cancer by targeting ROR1 protein. Int. J. Biol. Macromol. 2025, 310 (Pt 3), 143301, DOI: 10.1016/j.ijbiomac.2025.143301Google ScholarThere is no corresponding record for this reference.
- 24Li, J.; Li, L.; Hou, C.; Tian, Z.; Zhou, Y.; Zhang, J.; Ren, X.; Wang, Z.; Huang, W.; Ding, K.; Zhou, F. Discovery of the first potent ROR1 degrader for the treatment of non-small cell lung cancer. Eur. J. Med. Chem. 2025, 286, 117325, DOI: 10.1016/j.ejmech.2025.117325Google ScholarThere is no corresponding record for this reference.
- 25CUSABIO team ROR1, an Emerging Target for Tumor Immunotherapy; https://www.cusabio.com/c-21068.html?srsltid=AfmBOoosYHsnXKIFDuKMaivNn1yo_2SuMeIXNHvRBjewefiEsFcZxRWd.Google ScholarThere is no corresponding record for this reference.
- 26Bayat, A.-A.; Sadeghi, N.; Fatemi, R.; Nowroozi, M. R.; Ohadian Moghadam, S.; Borzuee, M.; Radmanesh, A.; Khodadoost, M.; Sarrafzadeh, A. R.; Zarei, O. Monoclonal antibody against ROR1 induces apoptosis in human bladder carcinoma cells. Avicenna J. Med. Biotechnol. 2020, 12 (3), 165– 171Google ScholarThere is no corresponding record for this reference.
- 27Widhopf, G. F., II; Cui, B.; Wu, C. C. N.; Yu, J.; Lao, F.; Tawatao, R.; Wu, R.; Avery, E.; Sadarangani, A.; Prussak, C. Targeting of chronic lymphocytic leukemia B cells with a humanized monoclonal antibody specific for ROR1. Blood 2013, 122 (21), 2873, DOI: 10.1182/blood.V122.21.2873.2873Google ScholarThere is no corresponding record for this reference.
- 28Lock, R. B.; Evans, K.; El-Zein, N.; Lannutti, B. J.; Jessen, K. A.; Earley, E. J.; Erickson, S. W.; Smith, M. A.; Kurmasheva, R.; Houghton, P. J. Abstract 3038: Evaluation of ROR1-targeted antibody-drug conjugates against ROR1-expressing pediatric preclinical models - a report from the pediatric preclinical testing consortium (PPTC). Canc. Res. 2021, 81 (13_Supplement), 3038, DOI: 10.1158/1538-7445.AM2021-3038Google ScholarThere is no corresponding record for this reference.
- 29Vaisitti, T.; Jessen, K.; Vo, T.-T.; Ko, M.; Arruga, F.; Vitale, N.; Braggio, E.; Di Napoli, A.; Chadburn, A.; Allan, J. N. Vls-101 is a novel therapeutic antibody-drug conjugate (ADC) targeting receptor tyrosine kinase-like orphan receptor 1 (ROR1) in Richter’s syndrome (RS). Blood 2019, 134 (Supplement_1), 2856, DOI: 10.1182/blood-2019-126827Google ScholarThere is no corresponding record for this reference.
- 30Peng, H.; Nerreter, T.; Mestermann, K.; Wachter, J.; Chang, J.; Hudecek, M.; Rader, C. ROR1-targeting switchable CAR-T cells for cancer therapy. Oncogene 2022, 41 (34), 4104– 4114, DOI: 10.1038/s41388-022-02416-5Google ScholarThere is no corresponding record for this reference.
- 31Wallstabe, L.; Göttlich, C.; Nelke, L. C.; Kühnemundt, J.; Schwarz, T.; Nerreter, T.; Einsele, H.; Walles, H.; Dandekar, G.; Nietzer, S. L. ROR1-CAR T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models. JCI Insight 2019, 4 (18), e126345 DOI: 10.1172/jci.insight.126345Google ScholarThere is no corresponding record for this reference.
- 32Choi, M. Y.; Widhopf, G. F., II; Ghia, E. M.; Kidwell, R. L.; Hasan, M. K.; Yu, J.; Rassenti, L. Z.; Chen, L.; Chen, Y.; Pittman, E. Phase I Trial: Cirmtuzumab Inhibits ROR1 Signaling and Stemness Signatures in Patients with Chronic Lymphocytic Leukemia. Cell Stem Cell 2018, 22 (6), P951– 959.E3, DOI: 10.1016/j.stem.2018.05.018Google ScholarThere is no corresponding record for this reference.
- 33Balaian, L.; Donohoe, M.; Mondala, P.; Widhopf Ii, G. F., II; Ghia, E. M.; Ball, E. D.; Kulidjian, A.; Van Der Werf, I.; Kipps, T. J.; Jamieson, C. Cirmtuzumab (Zilovertamab) eliminates ROR1 positive leukemia stem cells by blocking niche dependentROR1/WNT5a signaling. Blood 2022, 140 (Supplement 1), 3356– 3357, DOI: 10.1182/blood-2022-170128Google ScholarThere is no corresponding record for this reference.
- 34Lee, H. J.; Choi, M. Y.; Siddiqi, T.; Rhodes, J. M.; Wierda, W. G.; Isufi, I.; Tuscano, J. M.; Lamanna, N.; Subbiah, S.; Koff, J. L. Phase 1/2 study of Zilovertamab and ibrutinib in mantle cell lymphoma (MCL), chronic lymphocytic leukemia (CLL), or marginal zone lymphoma (MZL). Blood 2022, 140 (Supplement 1), 566– 568, DOI: 10.1182/blood-2022-167153Google ScholarThere is no corresponding record for this reference.
- 35Cobb, P. W.; Zhou, H.; Nahar, A.; Marinello, P. Open-label, active-control, phase 2/3 study of zilovertamab vedotin plus standard of care in patients with relapsed or refractory diffuse large B-cell lymphoma. J. Clin. Oncol. 2022, 40 (16_suppl), TPS7592, DOI: 10.1200/JCO.2022.40.16_suppl.TPS7592Google ScholarThere is no corresponding record for this reference.
- 36Lee, H. J.; Choi, M. Y.; Siddiqi, T.; Rhodes, J. M.; Wierda, W. G.; Isufi, I.; Tuscano, J. M.; Lamanna, N.; Subbiah, S.; Koff, J. L. Phase 1/2 study of Zilovertamab and ibrutinib in mantle cell lymphoma (MCL) or chronic lymphocytic leukemia (CLL). J. Clin. Oncol. 2022, 40 (16_suppl), 7520, DOI: 10.1200/JCO.2022.40.16_suppl.7520Google ScholarThere is no corresponding record for this reference.
- 37Jiang, V. C.; Liu, Y.; Jordan, A.; McIntosh, J.; Li, Y.; Che, Y.; Jessen, K. A.; Lannutti, B. J.; Wang, M. The antibody drug conjugate VLS-101 targeting ROR1 is effective in CAR T-resistant mantle cell lymphoma. J. Hematol. Oncol. 2021, 14 (1), 132, DOI: 10.1186/s13045-021-01143-wGoogle ScholarThere is no corresponding record for this reference.
- 38Chan, T.; Scott, S. P.; Du, M.; Bolinger, C.; Poortman, C.; Shepard, L.; Koenitzer, B.; Govekung, A.; Sailor, C.; Johnson, R. Preclinical evaluation of Prgn-3007, a non-viral, multigenic, autologous ROR1 Ultracar-T ® therapy with novel mechanism of intrinsic PD-1 blockade for treatment of hematological and solid cancers. Blood 2021, 138 (Supplement 1), 1694, DOI: 10.1182/blood-2021-149203Google ScholarThere is no corresponding record for this reference.
- 39Thurber, G. M.; Schmidt, M. M.; Wittrup, K. D. Factors determining antibody distribution in tumors. Trends Pharmacol. Sci. 2008, 29 (2), 57– 61, DOI: 10.1016/j.tips.2007.11.004Google ScholarThere is no corresponding record for this reference.
- 40Ma, J.; Wang, X.; Hu, Y.; Ma, J.; Ma, Y.; Chen, H.; Han, Z. Recent advances in augmenting the therapeutic efficacy of peptide–drug conjugates. J. Med. Chem. 2025, 68, 9037– 9056, DOI: 10.1021/acs.jmedchem.5c00007Google ScholarThere is no corresponding record for this reference.
- 41Evans, B. J.; King, A. T.; Katsifis, A.; Matesic, L.; Jamie, J. F. Methods to enhance the metabolic stability of peptide-based PET radiopharmaceuticals. Molecules 2020, 25 (10), 2314, DOI: 10.3390/molecules25102314Google ScholarThere is no corresponding record for this reference.
- 42Dean, T. T.; Jelú-Reyes, J.; Allen, A. L. C.; Moore, T. W. Peptide–drug conjugates: An emerging direction for the next generation of peptide therapeutics. J. Med. Chem. 2024, 67 (3), 1641– 1661, DOI: 10.1021/acs.jmedchem.3c01835Google ScholarThere is no corresponding record for this reference.
- 43Okarvi, S. M. Peptide-based radiopharmaceuticals: Future tools for diagnostic imaging of cancers and other diseases. Med. Res. Rev. 2004, 24 (3), 357– 397, DOI: 10.1002/med.20002Google ScholarThere is no corresponding record for this reference.
- 44Langer, M.; Beck-Sickinger, A. G. Peptides as carrier for tumor diagnosis and treatment. Curr. Med. Chem. Agents 2001, 1 (1), 71– 93, DOI: 10.2174/1568011013354877Google ScholarThere is no corresponding record for this reference.
- 45Fu, C.; Yu, L.; Miao, Y.; Liu, X.; Yu, Z.; Wei, M. Peptide–drug conjugates (PDCs): A novel trend of research and development on targeted therapy, hype or hope?. Acta Pharm. Sin. B 2023, 13 (2), 498– 516, DOI: 10.1016/j.apsb.2022.07.020Google ScholarThere is no corresponding record for this reference.
- 46Das, S.; Al-Toubah, A.-T.; El-Haddad, G.; Strosberg, J. 177Lu-DOTATATE for the treatment of gastroenteropancreatic neuroendocrine tumors. Expert Rev. Gastroenterol. Hepatol. 2019, 13 (11), 1023– 1031, DOI: 10.1080/17474124.2019.1685381Google ScholarThere is no corresponding record for this reference.
- 47Singh, S.; Halperin, D.; Myrehaug, S.; Herrmann, K.; Pavel, M.; Kunz, P. L.; Chasen, B.; Tafuto, S.; Lastoria, S.; Capdevila, J.; García-Burillo, A.; Oh, D.-Y.; Yoo, C.; Halfdanarson, T. R.; Falk, S.; Folitar, I.; Zhang, Y.; Aimone, P.; de Herder, W. W.; Ferone, D. [177Lu]Lu-DOTA-TATE plus long-acting octreotide versus high-dose long-acting octreotide for the treatment of newly diagnosed, advanced grade 2–3, well-differentiated, gastroenteropancreatic neuroendocrine tumours (NETTER-2): An open-label, randomised, phase 3 study. Lancet 2024, 403 (10446), 2807– 2817, DOI: 10.1016/S0140-6736(24)00701-3Google ScholarThere is no corresponding record for this reference.
- 48Sartor, O.; De Bono, J.; Chi, K. N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S. T.; Nordquist, L. T.; Vaishampayan, N.; El-Haddad, G. Lutetium-177–PSMA-617 for metastatic castration-resistant prostate cancer. N. Engl. J. Med. 2021, 385 (12), 1091– 1103, DOI: 10.1056/NEJMoa2107322Google ScholarThere is no corresponding record for this reference.
- 49Zhou, X.; Shi, B.; Huang, G.; Liu, J.; Wei, W. Trends in cancer imaging. Trends Cancer 2024, 10 (11), 1023– 1037, DOI: 10.1016/j.trecan.2024.08.006Google ScholarThere is no corresponding record for this reference.
- 50Hricak, H.; Mayerhoefer, M. E.; Herrmann, K.; Lewis, J. S.; Pomper, M. G.; Hess, C. P.; Riklund, K.; Scott, A. M.; Weissleder, R. Advances and challenges in precision imaging. Lancet Oncol. 2025, 26 (1), E34–E45 DOI: 10.1016/S1470-2045(24)00395-4Google ScholarThere is no corresponding record for this reference.
- 51Mou, Z.; Zhu, Y.; Zhang, L.; Ma, M.; Li, Z.; Guo, Y.; Zheng, J.; Zhao, Z.; Zhang, K.; Chen, X.; Li, Z. “AquaF” building blocks for water-compatible SN2 18F-fluorination of small-molecule radiotracers. J. Am. Chem. Soc. 2024, 146 (25), 17517– 17529, DOI: 10.1021/jacs.4c05854Google ScholarThere is no corresponding record for this reference.
- 52Zhang, K.; Feng, W.; Mou, Z.; Zhang, L.; Ma, M.; Zhao, Z.; Liu, X.; Chen, X.; Li, Z. Late-stage (radio)fluorination of alkyl phosphonates via electrophilic activation. Nat. Commun. 2024, 15 (1), 10338, DOI: 10.1038/s41467-024-54208-yGoogle ScholarThere is no corresponding record for this reference.
- 53Huang, S.; Tian, T.; Qi, M.; Li, M.; Wu, X.; Huang, R. Development of novel peptide-based 68Ga-labeled radiotracers for detecting ROR1 expression in tumors. Mol. Pharmaceutics 2025, 22 (10), 6185– 6192, DOI: 10.1021/acs.molpharmaceut.5c00917Google ScholarThere is no corresponding record for this reference.
- 54Yang, T.; Sun, J.; Huang, G.; Wang, H.; He, Z.; Lin, S.; Wang, Y.; Zhuang, R.; Zhang, X.; Liu, H.; Guo, Z. 18F-/68Ga-labeled peptide-based probes for PET imaging of ROR1 expression. J. Med. Chem. 2025, 68 (24), 26049– 26060, DOI: 10.1021/acs.jmedchem.5c02016Google ScholarThere is no corresponding record for this reference.
- 55Qi, J.; Li, X.; Peng, H.; Cook, E. M.; Dadashian, E. L.; Wiestner, A.; Park, H.; Rader, C. Potent and selective antitumor activity of a T cell-engaging bispecific antibody targeting a membrane-proximal epitope of ROR1. Proc. Natl. Acad. Sci. U. S. A. 2018, 115 (24), E5467–E5476 DOI: 10.1073/pnas.1719905115Google ScholarThere is no corresponding record for this reference.
- 56Qiao, J. X.; Witmer, M. R.; Lee, V.; Wang, T. C.; Reid, P. C.; Arioka, Y.; Farr, G.; Hill-Drzewi, M.; Schweizer, L.; Yamniuk, A.; Cheng, L.; Abramczyk, B.; Corbett, M.; Calambur, D.; Szapiel, N.; Ryseck, R.; Ponath, P.; Poss, M. A.; Carter, P. Exploration of macrocyclic peptide binders to the extracellular CRD domain of human receptor tyrosine kinase-like orphan receptor 1 (ROR1). Bioorg. Med. Chem. Lett. 2024, 98, 129589, DOI: 10.1016/j.bmcl.2023.129589Google ScholarThere is no corresponding record for this reference.
- 57Furman, J. L.; Chiu, M.; Hunter, M. J. Early engineering approaches to improve peptide developability and manufacturability. AAPS J. 2015, 17 (1), 111– 120, DOI: 10.1208/s12248-014-9681-9Google ScholarThere is no corresponding record for this reference.
- 58Burggraef, M. J.; Oxley, A.; Zaidi, N. A.; Cutillas, P. R.; Gaffney, P. R. J.; Livingston, A. G. Exactly defined molecular weight poly(ethylene glycol) allows for facile identification of PEGylation sites on proteins. Nat. Commun. 2024, 15 (1), 9814, DOI: 10.1038/s41467-024-54076-6Google ScholarThere is no corresponding record for this reference.
- 59Veronese, F. M.; Pasut, G. PEGylation, successful approach to drug delivery. Drug Discovery Today 2005, 10 (21), 1451– 1458, DOI: 10.1016/S1359-6446(05)03575-0Google ScholarThere is no corresponding record for this reference.
- 60Ullah, A.; Shin, G.; Lim, S. I. Human serum albumin binders: A piggyback ride for long-acting therapeutics. Drug Discovery Today 2023, 28 (10), 103738, DOI: 10.1016/j.drudis.2023.103738Google ScholarThere is no corresponding record for this reference.
- 61Ji, X.; Nielsen, A. L.; Heinis, C. Cyclic peptides for drug development. Angew. Chem., Int. Ed. 2024, 63 (3), e202308251 DOI: 10.1002/anie.202308251Google ScholarThere is no corresponding record for this reference.
- 62The Human Protein Atlas 2025, https://v22.proteinatlas.org/ENSG00000185483-ROR1/cell+line#skin_cancer.Google ScholarThere is no corresponding record for this reference.
- 63Wang, Z.; Pan, B. S.; Manne, R. K.; Chen, J.; Lv, D.; Wang, M.; Tran, P.; Weldemichael, T.; Yan, W.; Zhou, H.; Martinez, G. M.; Shao, J.; Hsu, C. C.; Hromas, R.; Zhou, D.; Qin, Z.; Lin, H. K.; Li, H. Y. CD36-mediated endocytosis of proteolysis-targeting chimeras. Cell 2025, 188 (12), 3219– 3237, DOI: 10.1016/j.cell.2025.03.036Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
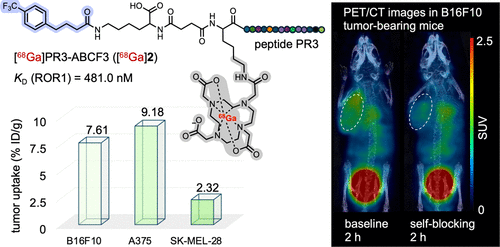
Figure 1

Figure 1. Ligand design and primary evaluations. (A) Schematic diagram and chemical structure of [68Ga]1. (B) MicroPET/CT images (left panel) and biodistribution results (right panel) in B16F10 tumor-bearing mice (n = 2–4) after intravenous injection of [68Ga]1. BL, bladder; K, kidneys; T, tumor.
Scheme 1
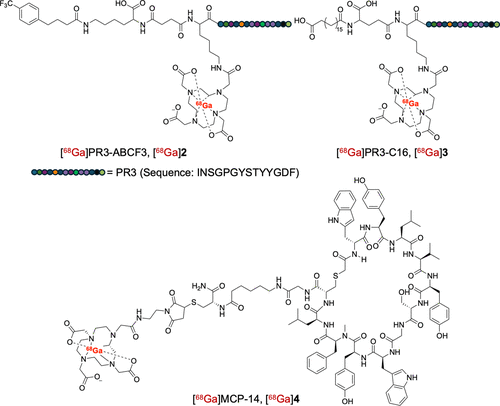 Scheme 1. Chemical Structures of [68Ga]2–4
Scheme 1. Chemical Structures of [68Ga]2–4Figure 2

Figure 2. BLI binding curves of the interaction of newly developed ligands with the ROR1 protein. A–D correspond to PR3-DOTA, PR3-ABCF3-DOTA, PR3-C16-DOTA, and MCP-14-DOTA, respectively.
Figure 3
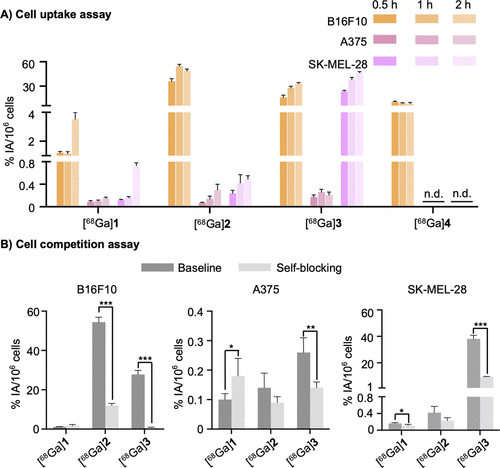
Figure 3. Cell uptake assay (A) and self-blocking assay (B) of [68Ga]1–4 in melanoma cell lines B16F10, A375, and SK-MEL-28; n.d., not determined. Asterisks indicate statistical significance: *p < 0.05, **p < 0.01, and ***p < 0.001 vs control. Data were analyzed using a two-tailed unpaired Student’s t-test.
Figure 4

Figure 4. (A, C) MicroPET/CT images of B16F10 tumor-bearing mice following the injection of [68Ga]2 (A) and [68Ga]3 (C), respectively. Self-blocking studies were performed by coinjecting the corresponding nonradioactive competitors, PR3-ABCF3-DOTA (0.25 mg/mouse) for [68Ga]2 and PR3-C16-DOTA (0.33 mg/mouse) for [68Ga]3. (B,D) The radioactivity uptake values of [68Ga]2 (B) and [68Ga]3 (D) are expressed as SUVmax in tumor and muscle. BL, bladder (red arrow); H, heart (black arrow); T, tumor (white arrow).
Figure 5
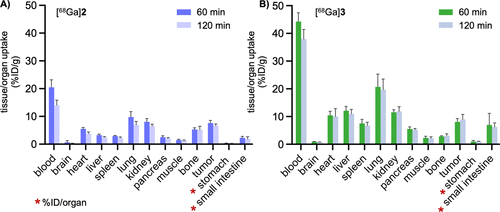
Figure 5. Ex vivo biodistribution of [68Ga]2 (A) and [68Ga]3 (B) in B16F10 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as the percentage of injected dose per gram of tissue (% ID/g), with the exception of the stomach and small intestine, for which uptake is presented as %ID per organ (% ID/organ).
Figure 6
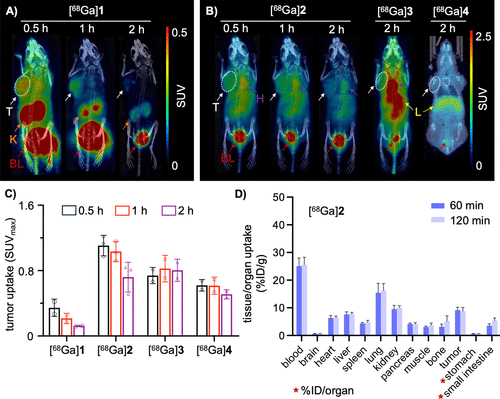
Figure 6. MicroPET/CT imaging and ex vivo biodistribution studies in A375 tumor-bearing mice. (A, B) MicroPET/CT images of A375 tumor-bearing mice following the injection of [68Ga]1–4. Representative images acquired at selected time points postinjection. [68Ga]1 (A) and [68Ga]2 (B), which achieved high T/M ratios, were shown in three time points of 0.5, 1, and 2 h, while [68Ga]3 and [68Ga]4 (B) that had lower T/M ratios were displayed at the 2 h time point only. (C) Quantitative analysis of tumor uptake for [68Ga]1–4 expressed as SUVmax. (D) Ex vivo biodistribution of [68Ga]2 in A375 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as % ID/g, with the exception of the stomach and small intestine, for which uptake is presented as % ID/organ. BL, bladder (red arrow); H, heart (purple arrow); L, liver (yellow arrow); and T, tumor (white arrow).
Figure 7
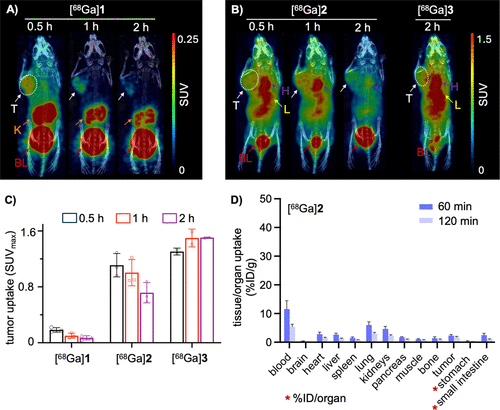
Figure 7. MicroPET/CT imaging and ex vivo biodistribution studies in SK-MEL-28 tumor-bearing mice. (A,B) MicroPET/CT images of SK-MEL-28 tumor-bearing mice following the injection of [68Ga]1–3. Representative images acquired at selected time points postinjection. [68Ga]1 (A) and [68Ga]2 (B), which achieved high T/M ratios, were shown in three time points of 0.5, 1, and 2 h, while [68Ga]3 (B) that had a lower T/M ratio was displayed at the 2 h time point only. (C) Quantitative analysis of tumor uptake for [68Ga]1–3 expressed as SUVmax. (D) Ex vivo biodistribution of [68Ga]2 in SK-MEL-28 tumor-bearing mice at 1 and 2 h postinjection. Data are expressed as % ID/g, with the exception of the stomach and small intestine, for which uptake is presented as % ID/organ. BL, bladder (red arrow); H, heart (purple arrow); L, liver (yellow arrow); T, tumor (white arrow).
Figure 8

Figure 8. Histopathological and immunohistochemical (IHC) characterization of B16F10, A375, and SK-MEL-28 melanoma tumors. Representative hematoxylin and eosin (HE)-stained sections (top panels) and corresponding IHC staining (middle panels) of paraffin-embedded tumor tissues. The lower panels show the quantitative analysis of protein expression levels based on histochemistry scores (H-scores). Data are expressed as mean ± SD (n = 10 fields per section). Scale bars: 150 μm.
References
This article references 63 other publications.
- 1Masiakowski, P.; Carroll, R. D. A novel family of cell surface receptors with tyrosine kinase-like domain. J. Biol. Chem. 1992, 267 (36), 26181– 26190, DOI: 10.1016/S0021-9258(18)35733-8There is no corresponding record for this reference.
- 2Kamizaki, K.; Endo, M.; Minami, Y.; Kobayashi, Y. Role of noncanonical Wnt ligands and Ror-family receptor tyrosine kinases in the development, regeneration, and diseases of the musculoskeletal system. Dev. Dyn. 2021, 250 (1), 27– 38, DOI: 10.1002/dvdy.151There is no corresponding record for this reference.
- 3Hasan, M. K.; Yu, J.; Widhopf, G. F., II; Rassenti, L. Z.; Chen, L.; Shen, Z.; Briggs, S. P.; Neuberg, D. S.; Kipps, T. J. Wnt5a induces ROR1 to recruit DOCK2 to activate Rac1/2 in chronic lymphocytic leukemia. Blood 2018, 132 (2), 170– 178, DOI: 10.1182/blood-2017-12-819383There is no corresponding record for this reference.
- 4Karvonen, H.; Perttila, R.; Niininen, W.; Hautanen, V.; Barker, H.; Murumagi, A.; Heckman, C. A.; Ungureanu, D. Wnt5a and ROR1 activate non-canonical Wnt signaling via RhoA in TCF3-PBX1 acute lymphoblastic leukemia and highlight new treatment strategies via Bcl-2 co-targeting. Oncogene 2019, 38 (17), 3288– 3300, DOI: 10.1038/s41388-018-0670-9There is no corresponding record for this reference.
- 5Li, L.; Huang, W.; Ren, X.; Wang, Z.; Ding, K.; Zhao, L.; Zhang, J. Unlocking the potential: Advancements and future horizons in ROR1-targeted cancer therapies. SCI. CHINA Life Sci. 2024, 67 (12), 2603– 2616, DOI: 10.1007/s11427-024-2685-9There is no corresponding record for this reference.
- 6Borcherding, N.; Kusner, D.; Liu, G.-H.; Zhang, W. ROR1, an embryonic protein with an emerging role in cancer biology. Protein Cell 2014, 5 (7), 496– 502, DOI: 10.1007/s13238-014-0059-7There is no corresponding record for this reference.
- 7Balakrishnan, A.; Goodpaster, T.; Randolph-Habecker, J.; Hoffstrom, B. G.; Jalikis, F. G.; Koch, L. K.; Berger, C.; Kosasih, P. L.; Rajan, A.; Sommermeyer, D.; Porter, P. L.; Riddell, S. R. Analysis of ROR1 protein expression in human cancer and normal tissues. Clin. Cancer Res. 2017, 23 (12), 3061– 3071, DOI: 10.1158/1078-0432.CCR-16-2083There is no corresponding record for this reference.
- 8Zhao, Y.; Zhang, D.; Guo, Y.; Lu, B.; Zhao, Z. J.; Xu, X.; Chen, Y. Tyrosine kinase ROR1 as a target for anti-cancer therapies. Front. Oncol. 2021, 11 (2021), 680834, DOI: 10.3389/fonc.2021.680834There is no corresponding record for this reference.
- 9Raso, M. G.; Barrientos Toro, E.; Evans, K.; Rizvi, Y.; Lazcano, R.; Akcakanat, A.; Sini, P.; Trapani, F.; Madlener, E. J.; Waldmeier, L.; Lazar, A.; Meric-Bernstam, F. Heterogeneous profile of ROR1 protein expression across tumor types. Cancers 2024, 16 (10), 1874, DOI: 10.3390/cancers16101874There is no corresponding record for this reference.
- 10Fernandez, N. B.; Lorenzo, D.; Picco, M. E.; Barbero, G.; Dergan-Dylon, L. S.; Marks, M. P.; Garcia-Rivello, H.; Gimenez, L.; Labovsky, V.; Grumolato, L.; Lopez-Bergami, P. ROR1 contributes to melanoma cell growth and migration by regulating N-cadherin expression via the PI3K/Akt pathway. Mol. Carcinog. 2016, 55 (11), 1772– 1785, DOI: 10.1002/mc.22426There is no corresponding record for this reference.
- 11Quezada, M. J.; Lopez-Bergami, P. The signaling pathways activated by ROR1 in cancer. Cell. Signal. 2023, 104, 110588, DOI: 10.1016/j.cellsig.2023.110588There is no corresponding record for this reference.
- 12Piki, E.; Dini, A.; Raivola, J.; Salokas, K.; Zhang, K.; Varjosalo, M.; Pellinen, T.; Valimaki, K.; Veskimae, K. T.; Staff, S. ROR1-STAT3 signaling contributes to ovarian cancer intra-tumor heterogeneity. Cell Death Discovery 2023, 9 (1), 222, DOI: 10.1038/s41420-023-01527-6There is no corresponding record for this reference.
- 13Li, C.; Wang, S.; Xing, Z.; Lin, A.; Liang, K.; Song, J.; Hu, Q.; Yao, J.; Chen, Z.; Park, P. K.; Hawke, D. H.; Zhou, J.; Zhou, Y.; Zhang, S.; Liang, H.; Hung, M. C.; Gallick, G. E.; Han, L.; Lin, C.; Yang, L. A ROR1-HER3-lncRNA signalling axis modulates the Hippo-YAP pathway to regulate bone metastasis. Nat. Cell Biol. 2017, 19 (2), 106– 119, DOI: 10.1038/ncb3464There is no corresponding record for this reference.
- 14Fukuda, T.; Chen, L.; Endo, T.; Tang, L.; Lu, D.; Castro, J. E.; Widhopf, G. F.; Rassenti, L. Z.; Cantwell, M. J.; Prussak, C. E.; Carson, D. A.; Kipps, T. J. Antisera induced by infusions of autologous Ad-CD154-leukemia B cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a. Proc. Natl. Acad. Sci. U. S. A. 2008, 105 (8), 3047– 3052, DOI: 10.1073/pnas.0712148105There is no corresponding record for this reference.
- 15Gohil, S. H.; Paredes-Moscosso, S. R.; Harrasser, M.; Vezzalini, M.; Scarpa, A.; Morris, E.; Davidoff, A. M.; Sorio, C.; Nathwani, A. C.; Della Peruta, M. An ROR1 bi-specific T-cell engager provides effective targeting and cytotoxicity against a range of solid tumors. OncoImmunology 2017, 6 (7), e1326437 DOI: 10.1080/2162402X.2017.1326437There is no corresponding record for this reference.
- 16Wu, D.; Gong, S.; Wu, X.; Naren, G.; Dong, L.; Lensky, S.; Wu, C. Abstract LB126: Generation of a novel ROR1 x CD3 bispecific T-cell engager for better tumor killing and minimal cytokine release. Cancer Res. 2024, 84 (7_Supplement), LB126, DOI: 10.1158/1538-7445.AM2024-LB126There is no corresponding record for this reference.
- 17Townsend, W.; Leong, S.; Tucker, D.; Pottinger, B.; Paneesha, S.; El-Sharkawi, D.; Eyre, T. A.; Batten, T.; Shah, M.; Cook, S. First-in-human phase I trial of a ROR1 targeting bispecific T cell engager (NVG-111) in combination with ibrutinib or as monotherapy in subjects with relapsed refractory chronic lymphocytic leukaemia (CLL) and mantle cell lymphoma (MCL). Blood 2022, 140 (Supplement 1), 4162– 4163, DOI: 10.1182/blood-2022-169658There is no corresponding record for this reference.
- 18Zhang, Y.; Fan, Y.; Liu, S.; Guan, Y.; Wan, J.; Ren, Q.; Wang, J.; Zhong, L.; Hu, Z.; Shi, W.; Qian, H. Development of peptide paratope mimics derived from the anti-ROR1 antibody and long-acting peptide-drug conjugates for targeted cancer therapy. J. Med. Chem. 2024, 67 (13), 10967– 10985, DOI: 10.1021/acs.jmedchem.4c00511There is no corresponding record for this reference.
- 19Hojjat-Farsangi, M.; Daneshmanesh, A. H.; Khan, A. S.; Shetye, J.; Mozaffari, F.; Kharaziha, P.; Rathje, L. S.; Kokhaei, P.; Hansson, L.; Vågberg, J.; Byström, S.; Olsson, E.; Löfberg, C.; Norström, C.; Schultz, J.; Norin, M.; Olin, T.; Österborg, A.; Mellstedt, H.; Moshfegh, A. First-in-class oral small molecule inhibitor of the tyrosine kinase ROR1 (KAN0439834) induced significant apoptosis of chronic lymphocytic leukemia cells. Leukemia 2018, 32 (10), 2291– 2295, DOI: 10.1038/s41375-018-0113-1There is no corresponding record for this reference.
- 20Liu, X.; Pu, W.; He, H.; Fan, X.; Zheng, Y.; Zhou, J.-K.; Ma, R.; He, J.; Zheng, Y.; Wu, K.; Zhao, Y.; Yang, S.-Y.; Wang, C.; Wei, Y.-Q.; Wei, X.-W.; Peng, Y. Novel ROR1 inhibitor ARI-1 suppresses the development of non-small cell lung cancer. Cancer Lett. 2019, 458, 76– 85, DOI: 10.1016/j.canlet.2019.05.016There is no corresponding record for this reference.
- 21Fan, Y.; Zhang, F.; Xiong, L.; Su, M.; Luo, F.; Li, M.; Li, Q.; Zhong, T.; Yuan, M.; Xu, Y.; Mu, S.; Yang, H. Design, synthesis, and biological evaluation of 6-(imidazo[1,2-a] pyridin-6-yl) quinazolin-4(3H)-one derivatives as potent anticancer agents by dual targeting Aurora kinase and ROR1. Bioorg. Chem. 2023, 135, 106484, DOI: 10.1016/j.bioorg.2023.106484There is no corresponding record for this reference.
- 22Luo, D.; Qiu, X.; Zheng, Q.; Ming, Y.; Pu, W.; Ai, M.; He, J.; Peng, Y. Discovery of novel receptor tyrosine kinase-like orphan receptor 1 (ROR1) inhibitors for cancer treatment. J. Med. Chem. 2024, 67 (13), 10655– 10686, DOI: 10.1021/acs.jmedchem.4c00175There is no corresponding record for this reference.
- 23Ta, T. M.; Reed, V. L.; Gupta, S. R. R.; Khan, M.; Chandra, N.; Dwyer, N.; Fultang, N.; Singh, I. K.; Peethambaran, B. Novel macromolecule CPD4 suppresses cell proliferation and metastasis of triple-negative breast cancer by targeting ROR1 protein. Int. J. Biol. Macromol. 2025, 310 (Pt 3), 143301, DOI: 10.1016/j.ijbiomac.2025.143301There is no corresponding record for this reference.
- 24Li, J.; Li, L.; Hou, C.; Tian, Z.; Zhou, Y.; Zhang, J.; Ren, X.; Wang, Z.; Huang, W.; Ding, K.; Zhou, F. Discovery of the first potent ROR1 degrader for the treatment of non-small cell lung cancer. Eur. J. Med. Chem. 2025, 286, 117325, DOI: 10.1016/j.ejmech.2025.117325There is no corresponding record for this reference.
- 25CUSABIO team ROR1, an Emerging Target for Tumor Immunotherapy; https://www.cusabio.com/c-21068.html?srsltid=AfmBOoosYHsnXKIFDuKMaivNn1yo_2SuMeIXNHvRBjewefiEsFcZxRWd.There is no corresponding record for this reference.
- 26Bayat, A.-A.; Sadeghi, N.; Fatemi, R.; Nowroozi, M. R.; Ohadian Moghadam, S.; Borzuee, M.; Radmanesh, A.; Khodadoost, M.; Sarrafzadeh, A. R.; Zarei, O. Monoclonal antibody against ROR1 induces apoptosis in human bladder carcinoma cells. Avicenna J. Med. Biotechnol. 2020, 12 (3), 165– 171There is no corresponding record for this reference.
- 27Widhopf, G. F., II; Cui, B.; Wu, C. C. N.; Yu, J.; Lao, F.; Tawatao, R.; Wu, R.; Avery, E.; Sadarangani, A.; Prussak, C. Targeting of chronic lymphocytic leukemia B cells with a humanized monoclonal antibody specific for ROR1. Blood 2013, 122 (21), 2873, DOI: 10.1182/blood.V122.21.2873.2873There is no corresponding record for this reference.
- 28Lock, R. B.; Evans, K.; El-Zein, N.; Lannutti, B. J.; Jessen, K. A.; Earley, E. J.; Erickson, S. W.; Smith, M. A.; Kurmasheva, R.; Houghton, P. J. Abstract 3038: Evaluation of ROR1-targeted antibody-drug conjugates against ROR1-expressing pediatric preclinical models - a report from the pediatric preclinical testing consortium (PPTC). Canc. Res. 2021, 81 (13_Supplement), 3038, DOI: 10.1158/1538-7445.AM2021-3038There is no corresponding record for this reference.
- 29Vaisitti, T.; Jessen, K.; Vo, T.-T.; Ko, M.; Arruga, F.; Vitale, N.; Braggio, E.; Di Napoli, A.; Chadburn, A.; Allan, J. N. Vls-101 is a novel therapeutic antibody-drug conjugate (ADC) targeting receptor tyrosine kinase-like orphan receptor 1 (ROR1) in Richter’s syndrome (RS). Blood 2019, 134 (Supplement_1), 2856, DOI: 10.1182/blood-2019-126827There is no corresponding record for this reference.
- 30Peng, H.; Nerreter, T.; Mestermann, K.; Wachter, J.; Chang, J.; Hudecek, M.; Rader, C. ROR1-targeting switchable CAR-T cells for cancer therapy. Oncogene 2022, 41 (34), 4104– 4114, DOI: 10.1038/s41388-022-02416-5There is no corresponding record for this reference.
- 31Wallstabe, L.; Göttlich, C.; Nelke, L. C.; Kühnemundt, J.; Schwarz, T.; Nerreter, T.; Einsele, H.; Walles, H.; Dandekar, G.; Nietzer, S. L. ROR1-CAR T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models. JCI Insight 2019, 4 (18), e126345 DOI: 10.1172/jci.insight.126345There is no corresponding record for this reference.
- 32Choi, M. Y.; Widhopf, G. F., II; Ghia, E. M.; Kidwell, R. L.; Hasan, M. K.; Yu, J.; Rassenti, L. Z.; Chen, L.; Chen, Y.; Pittman, E. Phase I Trial: Cirmtuzumab Inhibits ROR1 Signaling and Stemness Signatures in Patients with Chronic Lymphocytic Leukemia. Cell Stem Cell 2018, 22 (6), P951– 959.E3, DOI: 10.1016/j.stem.2018.05.018There is no corresponding record for this reference.
- 33Balaian, L.; Donohoe, M.; Mondala, P.; Widhopf Ii, G. F., II; Ghia, E. M.; Ball, E. D.; Kulidjian, A.; Van Der Werf, I.; Kipps, T. J.; Jamieson, C. Cirmtuzumab (Zilovertamab) eliminates ROR1 positive leukemia stem cells by blocking niche dependentROR1/WNT5a signaling. Blood 2022, 140 (Supplement 1), 3356– 3357, DOI: 10.1182/blood-2022-170128There is no corresponding record for this reference.
- 34Lee, H. J.; Choi, M. Y.; Siddiqi, T.; Rhodes, J. M.; Wierda, W. G.; Isufi, I.; Tuscano, J. M.; Lamanna, N.; Subbiah, S.; Koff, J. L. Phase 1/2 study of Zilovertamab and ibrutinib in mantle cell lymphoma (MCL), chronic lymphocytic leukemia (CLL), or marginal zone lymphoma (MZL). Blood 2022, 140 (Supplement 1), 566– 568, DOI: 10.1182/blood-2022-167153There is no corresponding record for this reference.
- 35Cobb, P. W.; Zhou, H.; Nahar, A.; Marinello, P. Open-label, active-control, phase 2/3 study of zilovertamab vedotin plus standard of care in patients with relapsed or refractory diffuse large B-cell lymphoma. J. Clin. Oncol. 2022, 40 (16_suppl), TPS7592, DOI: 10.1200/JCO.2022.40.16_suppl.TPS7592There is no corresponding record for this reference.
- 36Lee, H. J.; Choi, M. Y.; Siddiqi, T.; Rhodes, J. M.; Wierda, W. G.; Isufi, I.; Tuscano, J. M.; Lamanna, N.; Subbiah, S.; Koff, J. L. Phase 1/2 study of Zilovertamab and ibrutinib in mantle cell lymphoma (MCL) or chronic lymphocytic leukemia (CLL). J. Clin. Oncol. 2022, 40 (16_suppl), 7520, DOI: 10.1200/JCO.2022.40.16_suppl.7520There is no corresponding record for this reference.
- 37Jiang, V. C.; Liu, Y.; Jordan, A.; McIntosh, J.; Li, Y.; Che, Y.; Jessen, K. A.; Lannutti, B. J.; Wang, M. The antibody drug conjugate VLS-101 targeting ROR1 is effective in CAR T-resistant mantle cell lymphoma. J. Hematol. Oncol. 2021, 14 (1), 132, DOI: 10.1186/s13045-021-01143-wThere is no corresponding record for this reference.
- 38Chan, T.; Scott, S. P.; Du, M.; Bolinger, C.; Poortman, C.; Shepard, L.; Koenitzer, B.; Govekung, A.; Sailor, C.; Johnson, R. Preclinical evaluation of Prgn-3007, a non-viral, multigenic, autologous ROR1 Ultracar-T ® therapy with novel mechanism of intrinsic PD-1 blockade for treatment of hematological and solid cancers. Blood 2021, 138 (Supplement 1), 1694, DOI: 10.1182/blood-2021-149203There is no corresponding record for this reference.
- 39Thurber, G. M.; Schmidt, M. M.; Wittrup, K. D. Factors determining antibody distribution in tumors. Trends Pharmacol. Sci. 2008, 29 (2), 57– 61, DOI: 10.1016/j.tips.2007.11.004There is no corresponding record for this reference.
- 40Ma, J.; Wang, X.; Hu, Y.; Ma, J.; Ma, Y.; Chen, H.; Han, Z. Recent advances in augmenting the therapeutic efficacy of peptide–drug conjugates. J. Med. Chem. 2025, 68, 9037– 9056, DOI: 10.1021/acs.jmedchem.5c00007There is no corresponding record for this reference.
- 41Evans, B. J.; King, A. T.; Katsifis, A.; Matesic, L.; Jamie, J. F. Methods to enhance the metabolic stability of peptide-based PET radiopharmaceuticals. Molecules 2020, 25 (10), 2314, DOI: 10.3390/molecules25102314There is no corresponding record for this reference.
- 42Dean, T. T.; Jelú-Reyes, J.; Allen, A. L. C.; Moore, T. W. Peptide–drug conjugates: An emerging direction for the next generation of peptide therapeutics. J. Med. Chem. 2024, 67 (3), 1641– 1661, DOI: 10.1021/acs.jmedchem.3c01835There is no corresponding record for this reference.
- 43Okarvi, S. M. Peptide-based radiopharmaceuticals: Future tools for diagnostic imaging of cancers and other diseases. Med. Res. Rev. 2004, 24 (3), 357– 397, DOI: 10.1002/med.20002There is no corresponding record for this reference.
- 44Langer, M.; Beck-Sickinger, A. G. Peptides as carrier for tumor diagnosis and treatment. Curr. Med. Chem. Agents 2001, 1 (1), 71– 93, DOI: 10.2174/1568011013354877There is no corresponding record for this reference.
- 45Fu, C.; Yu, L.; Miao, Y.; Liu, X.; Yu, Z.; Wei, M. Peptide–drug conjugates (PDCs): A novel trend of research and development on targeted therapy, hype or hope?. Acta Pharm. Sin. B 2023, 13 (2), 498– 516, DOI: 10.1016/j.apsb.2022.07.020There is no corresponding record for this reference.
- 46Das, S.; Al-Toubah, A.-T.; El-Haddad, G.; Strosberg, J. 177Lu-DOTATATE for the treatment of gastroenteropancreatic neuroendocrine tumors. Expert Rev. Gastroenterol. Hepatol. 2019, 13 (11), 1023– 1031, DOI: 10.1080/17474124.2019.1685381There is no corresponding record for this reference.
- 47Singh, S.; Halperin, D.; Myrehaug, S.; Herrmann, K.; Pavel, M.; Kunz, P. L.; Chasen, B.; Tafuto, S.; Lastoria, S.; Capdevila, J.; García-Burillo, A.; Oh, D.-Y.; Yoo, C.; Halfdanarson, T. R.; Falk, S.; Folitar, I.; Zhang, Y.; Aimone, P.; de Herder, W. W.; Ferone, D. [177Lu]Lu-DOTA-TATE plus long-acting octreotide versus high-dose long-acting octreotide for the treatment of newly diagnosed, advanced grade 2–3, well-differentiated, gastroenteropancreatic neuroendocrine tumours (NETTER-2): An open-label, randomised, phase 3 study. Lancet 2024, 403 (10446), 2807– 2817, DOI: 10.1016/S0140-6736(24)00701-3There is no corresponding record for this reference.
- 48Sartor, O.; De Bono, J.; Chi, K. N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S. T.; Nordquist, L. T.; Vaishampayan, N.; El-Haddad, G. Lutetium-177–PSMA-617 for metastatic castration-resistant prostate cancer. N. Engl. J. Med. 2021, 385 (12), 1091– 1103, DOI: 10.1056/NEJMoa2107322There is no corresponding record for this reference.
- 49Zhou, X.; Shi, B.; Huang, G.; Liu, J.; Wei, W. Trends in cancer imaging. Trends Cancer 2024, 10 (11), 1023– 1037, DOI: 10.1016/j.trecan.2024.08.006There is no corresponding record for this reference.
- 50Hricak, H.; Mayerhoefer, M. E.; Herrmann, K.; Lewis, J. S.; Pomper, M. G.; Hess, C. P.; Riklund, K.; Scott, A. M.; Weissleder, R. Advances and challenges in precision imaging. Lancet Oncol. 2025, 26 (1), E34–E45 DOI: 10.1016/S1470-2045(24)00395-4There is no corresponding record for this reference.
- 51Mou, Z.; Zhu, Y.; Zhang, L.; Ma, M.; Li, Z.; Guo, Y.; Zheng, J.; Zhao, Z.; Zhang, K.; Chen, X.; Li, Z. “AquaF” building blocks for water-compatible SN2 18F-fluorination of small-molecule radiotracers. J. Am. Chem. Soc. 2024, 146 (25), 17517– 17529, DOI: 10.1021/jacs.4c05854There is no corresponding record for this reference.
- 52Zhang, K.; Feng, W.; Mou, Z.; Zhang, L.; Ma, M.; Zhao, Z.; Liu, X.; Chen, X.; Li, Z. Late-stage (radio)fluorination of alkyl phosphonates via electrophilic activation. Nat. Commun. 2024, 15 (1), 10338, DOI: 10.1038/s41467-024-54208-yThere is no corresponding record for this reference.
- 53Huang, S.; Tian, T.; Qi, M.; Li, M.; Wu, X.; Huang, R. Development of novel peptide-based 68Ga-labeled radiotracers for detecting ROR1 expression in tumors. Mol. Pharmaceutics 2025, 22 (10), 6185– 6192, DOI: 10.1021/acs.molpharmaceut.5c00917There is no corresponding record for this reference.
- 54Yang, T.; Sun, J.; Huang, G.; Wang, H.; He, Z.; Lin, S.; Wang, Y.; Zhuang, R.; Zhang, X.; Liu, H.; Guo, Z. 18F-/68Ga-labeled peptide-based probes for PET imaging of ROR1 expression. J. Med. Chem. 2025, 68 (24), 26049– 26060, DOI: 10.1021/acs.jmedchem.5c02016There is no corresponding record for this reference.
- 55Qi, J.; Li, X.; Peng, H.; Cook, E. M.; Dadashian, E. L.; Wiestner, A.; Park, H.; Rader, C. Potent and selective antitumor activity of a T cell-engaging bispecific antibody targeting a membrane-proximal epitope of ROR1. Proc. Natl. Acad. Sci. U. S. A. 2018, 115 (24), E5467–E5476 DOI: 10.1073/pnas.1719905115There is no corresponding record for this reference.
- 56Qiao, J. X.; Witmer, M. R.; Lee, V.; Wang, T. C.; Reid, P. C.; Arioka, Y.; Farr, G.; Hill-Drzewi, M.; Schweizer, L.; Yamniuk, A.; Cheng, L.; Abramczyk, B.; Corbett, M.; Calambur, D.; Szapiel, N.; Ryseck, R.; Ponath, P.; Poss, M. A.; Carter, P. Exploration of macrocyclic peptide binders to the extracellular CRD domain of human receptor tyrosine kinase-like orphan receptor 1 (ROR1). Bioorg. Med. Chem. Lett. 2024, 98, 129589, DOI: 10.1016/j.bmcl.2023.129589There is no corresponding record for this reference.
- 57Furman, J. L.; Chiu, M.; Hunter, M. J. Early engineering approaches to improve peptide developability and manufacturability. AAPS J. 2015, 17 (1), 111– 120, DOI: 10.1208/s12248-014-9681-9There is no corresponding record for this reference.
- 58Burggraef, M. J.; Oxley, A.; Zaidi, N. A.; Cutillas, P. R.; Gaffney, P. R. J.; Livingston, A. G. Exactly defined molecular weight poly(ethylene glycol) allows for facile identification of PEGylation sites on proteins. Nat. Commun. 2024, 15 (1), 9814, DOI: 10.1038/s41467-024-54076-6There is no corresponding record for this reference.
- 59Veronese, F. M.; Pasut, G. PEGylation, successful approach to drug delivery. Drug Discovery Today 2005, 10 (21), 1451– 1458, DOI: 10.1016/S1359-6446(05)03575-0There is no corresponding record for this reference.
- 60Ullah, A.; Shin, G.; Lim, S. I. Human serum albumin binders: A piggyback ride for long-acting therapeutics. Drug Discovery Today 2023, 28 (10), 103738, DOI: 10.1016/j.drudis.2023.103738There is no corresponding record for this reference.
- 61Ji, X.; Nielsen, A. L.; Heinis, C. Cyclic peptides for drug development. Angew. Chem., Int. Ed. 2024, 63 (3), e202308251 DOI: 10.1002/anie.202308251There is no corresponding record for this reference.
- 62The Human Protein Atlas 2025, https://v22.proteinatlas.org/ENSG00000185483-ROR1/cell+line#skin_cancer.There is no corresponding record for this reference.
- 63Wang, Z.; Pan, B. S.; Manne, R. K.; Chen, J.; Lv, D.; Wang, M.; Tran, P.; Weldemichael, T.; Yan, W.; Zhou, H.; Martinez, G. M.; Shao, J.; Hsu, C. C.; Hromas, R.; Zhou, D.; Qin, Z.; Lin, H. K.; Li, H. Y. CD36-mediated endocytosis of proteolysis-targeting chimeras. Cell 2025, 188 (12), 3219– 3237, DOI: 10.1016/j.cell.2025.03.036There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jmedchem.6c00570.
Synthetic routes and procedures of peptide-DOTA conjugates (PR3-DOTA, PR3-ABCF3-DOTA, PR3-C16-DOTA, and MCP-14-DOTA), purity of the peptide-DOTA conjugates with HPLC chromatograms, radiosynthetic routes of [68Ga]1–4, HPLC profiles of radioligands, in vitro stability, ex vivo biodistribution data of [68Ga]1–3 in tumor-bearing mice, ex vivo biodistribution data of [68Ga]2 in ICR mice, tumor and muscle SUVmax from PET imaging, PET images of radioligands in PET imaging studies, time-blood radioactivity curve of [68Ga]2, estimated absorbed radiation dosimetry for [68Ga]2 in human, and MS spectra of the peptide-DOTA conjugates (PDF)
Molecular formula strings data (CSV)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



