The Role of Li+ Ions in Polyzwitterionic Ionogels: Gelator or Mobile Charge Carrier?Click to copy article linkArticle link copied!
- Sajal ArwishSajal ArwishInstitute of Physical Chemistry, University of Münster, Corrensstr. 2830, 48149 Münster, GermanyMore by Sajal Arwish
- Mossab K. AlsaediMossab K. AlsaediTufts University, Department of Chemical & Biological Engineering, 4 Colby St., Medford, Massachusetts 02155, United StatesMore by Mossab K. Alsaedi
- Ryan P. O’HaraRyan P. O’HaraTufts University, Department of Chemical & Biological Engineering, 4 Colby St., Medford, Massachusetts 02155, United StatesMore by Ryan P. O’Hara
- Ayse AsatekinAyse AsatekinTufts University, Department of Chemical & Biological Engineering, 4 Colby St., Medford, Massachusetts 02155, United StatesMore by Ayse Asatekin
- Matthew J. PanzerMatthew J. PanzerTufts University, Department of Chemical & Biological Engineering, 4 Colby St., Medford, Massachusetts 02155, United StatesMore by Matthew J. Panzer
- Monika Schönhoff*Monika Schönhoff*Email: [email protected]Institute of Physical Chemistry, University of Münster, Corrensstr. 2830, 48149 Münster, GermanyMore by Monika Schönhoff
Abstract
Introducing zwitterionic (ZI) polymers to lithium-containing, ionic liquid-based electrolytes can change the dynamics and local environment of Li+ ions. While Li+ ions enable the formation of noncovalently cross-linked ionogels, they may also act as free charge carriers. Aiming at an understanding of these roles, we investigate Li+ ion coordination and transport in ionogels consisting of 1-butyl-1-methyl pyrrolidinium bis(trifluoromethyl sulfonamide) (BMP TFSI), LiTFSI, and poly(2-(methacryloyloxyethyl phosphorylcholine)) p(MPC), obtained via in situ free radical polymerization. Ionic conductivity as well as self-diffusion of both IL ions benefit from increasing p(MPC) content, while Li+ diffusion is reduced. Simultaneously, 7Li spin relaxation documents Li+ ion immobilization, attributed to a strong affinity of Li+ to the negatively charged phosphate group of p(MPC). Raman spectroscopy confirms decreasing TFSI-Li+ coordination with increasing p(MPC) content. Mechanical analysis reveals a drastic increase in elastic modulus, suggesting noncovalent cross-link formation. Finally, the distribution of Li+ on different sites, such as mobile and anion-coordinated, single chain-coordinated, or dual-chain cross-linking Li+ is analyzed. The results allow for an in-depth discussion of the role of Li+, partly acting as a cross-linker and partly as a mobile charge carrier, providing guidelines for optimizing the balance between ionic conductivity and mechanical strength.
This publication is licensed for personal use by The American Chemical Society.
Introduction
Scheme 1
aBridging Li+ that acts as noncovalent cross-linker between ZI units, non-bridging Li+ coordinated to a single ZI unit, and mobile Li+ in solution (bottom).
Materials and Methods
Materials
Ionogel Preparation
Dynamic Light Scattering (DLS)
Ionic Conductivity
NMR Spectroscopy and Pulsed-Field Gradient NMR (PFG NMR)
Raman Spectroscopy
Dynamic Mechanical Analysis
Results and Discussion
In Situ Polymerization and Molecular Weight
| p(MPC) content (wt %) | Stokes diameter (nm) | Polydispersity index |
|---|---|---|
| 2 | 42 ± 2 | 0.15 ± 0.04 |
| 4 | 39 ± 3 | 0.16 ± 0.02 |
| 6 | 38 ± 1 | 0.14 ± 0.01 |
| 8 | 35 ± 3 | 0.15 ± 0.04 |
| 10 | 29 ± 2 | 0.15 ± 0.03 |
| 12 | 25 ± 0.2 | 0.14 ± 0.02 |
| 14 | 21 ± 0.8 | 0.16 ± 0.02 |
Ionic Conductivity
Figure 1
Figure 1. Total ionic conductivity at room temperature (25 °C) as a function of p(MPC) wt %.
Diffusion Coefficients of the Constituents
Figure 2
Figure 2. (a) Self-diffusion coefficients of the nuclei (1H, 19F, 7Li) obtained from PFG-NMR at room temperature (25 °C) as function of p(MPC) content. (b) Activation energies of diffusion as a function of p(MPC) content measured over a temperature range of 25–65 °C. The dotted lines are merely guides to the eye.
Li+ Ion Apparent Transference Number
Figure 3
Figure 3. Li+ ion apparent transference number as function of p(MPC) content.
7Li NMR Spectra and Spin–Spin Relaxation Rates
Figure 4
Figure 4. (a) 7Li NMR spectra, (b) peak integral, (c) line width, and (d) 7Li spin–spin relaxation rate R2 determined by a stimulated echo sequence (black) and R2* determined by line width analysis (red star) for 1 M LiTFSI/BMP TFSI solution (0 wt % p(MPC)), 2 wt % p(MPC) solution, and 4–8 wt % p(MPC) free-standing ionogels.
Raman Spectroscopy
Figure 5
Figure 5. (a) Raman spectra and (b) Li+ coordination number (coordinated anions per Li+) as extracted from the fraction of coordinated anions in dependence on p(MPC) content. The green dotted line in a) marks the position of the band of coordinated TFSI– and the black arrow represents the increase in p(MPC) content from 2 wt % (green spectrum) to 14 wt % (dark blue).
Dynamic Mechanical Analysis (DMA)
Figure 6
Figure 6. (a) Elastic modulus values for ionogels containing 4 to 14 wt % p(MPC). (b) Calculated cross-link density within the ionogels, representing the number of Li+ ion-mediated noncovalent cross-links per nm3, and (c) the corresponding fraction of Li+ that is not participating in noncovalent cross-links.
Discussion of Li+ Sites
| r | m | b | n | fLi | |
|---|---|---|---|---|---|
| Composition wt % p(MPC) (target value) | MPC:Li+ molar ratio (actual) | mobile fraction of Li+ (7Li NMR) | Immobilized Li+ per MPC | Coord. no. TFSI– per Li+ (Raman) | noncross-linked Li+ (DMA) |
| 0 | 0 | 1 | 0 | 1.81 | 1 |
| 2 | 0.13 | 0.96 | 0.31 | 1.65 | 1 |
| 4 | 0.24 | 0.73 | 1.13 | 1.51 | 1 |
| 6 | 0.34 | 0.47 | 1.55 | 1.25 | 1 |
| 8 | 0.50 | 0.24 | 1.52 | 0.98 | 0.98 |
| 10 | 0.62 | 0 | 1.60 | 0.88 | 0.90 |
| 12 | 0.69 | 0 | 1.40 | 0.81 | 0.75 |
| 14 | 0.92 | 0 | 1.10 | 0.72 | 0.44 |
Figure 7
Figure 7. (a) Distribution of Li+ in different sites; (b) – (d) Li+ coordination scenarios at 0, 8, and 14 wt % p(MPC), showing free, immobilized, and cross-linking Li+ ions. For a legend of the symbols, see Scheme 1. Note that the MPC:Li+ molar ratio and number of coordinating anions are shown according to scale, while the total number of IL cations and anions is not to scale to enhance clarity.
Implications for Transport
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06389.
Supporting data such as 1H NMR spectra of dissolved ionogels, temperature-dependent diffusion coefficients, spin relaxation rates, deconvolution of Raman spectra and calculation of coordination number, estimation of cross-linking density from DMA data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We would like to thank Professor David Kaplan in the Department of Biomedical Engineering at Tufts University for providing us with access to the texture analyzer used to obtain dynamic mechanical analysis (DMA) results. This project is funded by the DFG-NSF (German Science Foundation-US National Science Foundation) joint program CONFINE, project no. 509154483 (NSF award 2234243). The NMR spectrometer is funded by the DFG via proposal INST 211/999-1 FUGG, project ID 452849940.
References
This article references 52 other publications.
- 1Kim, T.; Song, W.; Son, D.-Y.; Ono, L. K.; Qi, Y. Lithium-ion batteries: outlook on present, future, and hybridized technologies. J. Mater. Chem. A 2019, 7 (7), 2942– 2964, DOI: 10.1039/C8TA10513HGoogle ScholarThere is no corresponding record for this reference.
- 2Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sources 2013, 226, 272– 288, DOI: 10.1016/j.jpowsour.2012.10.060Google ScholarThere is no corresponding record for this reference.
- 3Wang, Y.; Zhong, W.-H. Development of Electrolytes towards Achieving Safe and High-Performance Energy-Storage Devices: A Review. ChemElectroChem. 2015, 2 (1), 22– 36, DOI: 10.1002/celc.201402277Google ScholarThere is no corresponding record for this reference.
- 4Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210– 224, DOI: 10.1016/j.jpowsour.2012.02.038Google ScholarThere is no corresponding record for this reference.
- 5Goodenough, J. B.; Kim, Y. Challenges for Rechargeable Li Batteries. Chem. Mater. 2010, 22 (3), 587– 603, DOI: 10.1021/cm901452zGoogle ScholarThere is no corresponding record for this reference.
- 6Balducci, A. Ionic Liquids in Lithium-Ion Batteries. Top. Curr. Chem. 2017, 375 (2), 20, DOI: 10.1007/s41061-017-0109-8Google ScholarThere is no corresponding record for this reference.
- 7Francis, C. F. J.; Kyratzis, I. L.; Best, A. S. Lithium-Ion Battery Separators for Ionic-Liquid Electrolytes: A Review. Adv. Mater. 2020, 32 (18), 1904205, DOI: 10.1002/adma.201904205Google ScholarThere is no corresponding record for this reference.
- 8Tang, X.; Lv, S. Y.; Jiang, K.; Zhou, G. H.; Liu, X. M. Recent development of ionic liquid-based electrolytes in lithium-ion batteries. J. Power Sources 2022, 542, 231792, DOI: 10.1016/j.jpowsour.2022.231792Google ScholarThere is no corresponding record for this reference.
- 9Liu, J.; Yao, Y. C.; Zhang, S. Z.; Zhang, K. Y.; Peng, C. J.; Liu, H. L. Application of Ionic Liquids in Lithium Metal Batteries. Prog. Chem. 2025, 37 (5), 788– 800Google ScholarThere is no corresponding record for this reference.
- 10Watanabe, M.; Thomas, M. L.; Zhang, S. G.; Ueno, K.; Yasuda, T.; Dokko, K. Application of Ionic Liquids to Energy Storage and Conversion Materials and Devices. Chem. Rev. 2017, 117 (10), 7190– 7239, DOI: 10.1021/acs.chemrev.6b00504Google ScholarThere is no corresponding record for this reference.
- 11Armand, M.; Endres, F.; MacFarlane, D. R.; Ohno, H.; Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 2009, 8 (8), 621– 629, DOI: 10.1038/nmat2448Google ScholarThere is no corresponding record for this reference.
- 12Lewandowski, A.; Świderska-Mocek, A. Ionic liquids as electrolytes for Li-ion batteries─An overview of electrochemical studies. J. Power Sources 2009, 194 (2), 601– 609, DOI: 10.1016/j.jpowsour.2009.06.089Google ScholarThere is no corresponding record for this reference.
- 13Piatti, E.; Guglielmero, L.; Tofani, G.; Mezzetta, A.; Guazzelli, L.; D’Andrea, F.; Roddaro, S.; Pomelli, C. S. Ionic liquids for electrochemical applications: Correlation between molecular structure and electrochemical stability window. J. Mol. Liq. 2022, 364, 120001, DOI: 10.1016/j.molliq.2022.120001Google ScholarThere is no corresponding record for this reference.
- 14Hayyan, M.; Mjalli, F. S.; Hashim, M. A.; AlNashef, I. M.; Mei, T. X. Investigating the electrochemical windows of ionic liquids. J. Ind. Eng. Chem. 2013, 19 (1), 106– 112, DOI: 10.1016/j.jiec.2012.07.011Google ScholarThere is no corresponding record for this reference.
- 15Le Bideau, J.; Viau, L.; Vioux, A. Ionogels, ionic liquid based hybrid materials. Chem. Soc. Rev. 2011, 40 (2), 907– 925, DOI: 10.1039/C0CS00059KGoogle ScholarThere is no corresponding record for this reference.
- 16Giffin, G. A. Ionic liquid-based electrolytes for ″beyond lithium″ battery technologies. J. Mater. Chem. A 2016, 4 (35), 13378– 13389, DOI: 10.1039/C6TA05260FGoogle ScholarThere is no corresponding record for this reference.
- 17Galiński, M.; Lewandowski, A.; Stępniak, I. Ionic liquids as electrolytes. Electrochim. Acta 2006, 51 (26), 5567– 5580, DOI: 10.1016/j.electacta.2006.03.016Google ScholarThere is no corresponding record for this reference.
- 18Hayamizu, K.; Aihara, Y.; Nakagawa, H.; Nukuda, T.; Price, W. S. Ionic conduction and ion diffusion in binary room-temperature ionic liquids composed of emim BF4 and LiBF4. J. Phys. Chem. B 2004, 108 (50), 19527– 19532, DOI: 10.1021/jp0476601Google ScholarThere is no corresponding record for this reference.
- 19Saito, Y.; Umecky, T.; Niwa, J.; Sakai, T.; Maeda, S. Existing condition and migration property of ions in lithium electrolytes with ionic liquid solvent. J. Phys. Chem. B 2007, 111 (40), 11794– 11802, DOI: 10.1021/jp072998rGoogle ScholarThere is no corresponding record for this reference.
- 20Nicotera, I.; Oliviero, C.; Henderson, W. A.; Appetecchi, G. B.; Passerini, S. NMR investigation of ionic liquid-LiX mixtures: Pyrrolidinium cations and TFSI- anions. J. Phys. Chem. B 2005, 109 (48), 22814– 22819, DOI: 10.1021/jp053799fGoogle ScholarThere is no corresponding record for this reference.
- 21Castiglione, F.; Famulari, A.; Raos, G.; Meille, S. V.; Mele, A.; Appetecchi, G. B.; Passerini, S. Pyrrolidinium-Based Ionic Liquids Doped with Lithium Salts: How Does Li+ Coordination Affect Its Diffusivity?. J. Phys. Chem. B 2014, 118 (47), 13679– 13688, DOI: 10.1021/jp509387rGoogle ScholarThere is no corresponding record for this reference.
- 22Tong, J. H.; Wu, S. L.; von Solms, N.; Liang, X. D.; Huo, F.; Zhou, Q.; He, H. Y.; Zhang, S. J. The Effect of Concentration of Lithium Salt on the Structural and Transport Properties of Ionic Liquid-Based Electrolytes. Front. Chem. 2020, 7, 945, DOI: 10.3389/fchem.2019.00945Google ScholarThere is no corresponding record for this reference.
- 23Umebayashi, Y.; Mori, S.; Fujii, K.; Tsuzuki, S.; Seki, S.; Hayamizu, K.; Ishiguro, S. Raman Spectroscopic Studies and Ab Initio Calculations on Conformational Isomerism of 1-Butyl-3-methylimidazolium Bis-(trifluoromethanesulfonyl)amide Solvated to a Lithium Ion in Ionic Liquids: Effects of the Second Solvation Sphere of the Lithium Ion. J. Phys. Chem. B 2010, 114 (19), 6513– 6521, DOI: 10.1021/jp100898hGoogle ScholarThere is no corresponding record for this reference.
- 24Méndez-Morales, T.; Carrete, J.; Bouzón-Capelo, S.; Pérez-Rodríguez, M.; Cabeza, O.; Gallego, L. J.; Varela, L. M. MD Simulations of the Formation of Stable Clusters in Mixtures of Alkaline Salts and Imidazolium-Based Ionic Liquids. J. Phys. Chem. B 2013, 117 (11), 3207– 3220, DOI: 10.1021/jp312669rGoogle ScholarThere is no corresponding record for this reference.
- 25Umebayashi, Y.; Mitsugi, T.; Fukuda, S.; Fujimori, T.; Fujii, K.; Kanzaki, R.; Takeuchi, M.; Ishiguro, S. I. Lithium ion solvation in room-temperature ionic liquids involving bis(trifluoromethanesulfonyl) imide anion studied by Raman Spectroscopy and DFT calculations. J. Phys. Chem. B 2007, 111 (45), 13028– 13032, DOI: 10.1021/jp076869mGoogle ScholarThere is no corresponding record for this reference.
- 26Pitawala, J.; Martinelli, A.; Johansson, P.; Jacobsson, P.; Matic, A. Coordination and interactions in a Li-salt doped ionic liquid. J. Non-Cryst. Solids 2015, 407, 318– 323, DOI: 10.1016/j.jnoncrysol.2014.08.043Google ScholarThere is no corresponding record for this reference.
- 27Gouverneur, M.; Schmidt, F.; Schönhoff, M. Negative effective Li transference numbers in Li salt/ionic liquid mixtures: does Li drift in the ″Wrong″ direction?. Phys. Chem. Chem. Phys. 2018, 20 (11), 7470– 7478, DOI: 10.1039/C7CP08580JGoogle ScholarThere is no corresponding record for this reference.
- 28Brinkkötter, M.; Giffin, G. A.; Moretti, A.; Jeong, S.; Passerini, S.; Schönhoff, M. Relevance of ion clusters for Li transport at elevated salt concentrations in [Pyr(12O1)][FTFSI] ionic liquid-based electrolytes. Chem. Commun. 2018, 54 (34), 4278– 4281, DOI: 10.1039/C8CC01416GGoogle ScholarThere is no corresponding record for this reference.
- 29Molinari, N.; Mailoa, J. P.; Kozinsky, B. General Trend of a Negative Li Effective Charge in Ionic Liquid Electrolytes. J. Phys. Chem. Lett. 2019, 10 (10), 2313– 2319, DOI: 10.1021/acs.jpclett.9b00798Google ScholarThere is no corresponding record for this reference.
- 30Brinkkötter, M.; Mariani, A.; Jeong, S.; Passerini, S.; Schönhoff, M. Ionic Liquid in Li Salt Electrolyte: Modifying the Li+ Transport Mechanism by Coordination to an Asymmetric Anion. Adv. Energy Sustainability Res. 2021, 2 (2), 2000078, DOI: 10.1002/aesr.202000078Google ScholarThere is no corresponding record for this reference.
- 31Nürnberg, P.; Lozinskaya, E. I.; Shaplov, A. S.; Schönhoff, M. Li Coordination of a Novel Asymmetric Anion in Ionic Liquid-in-Li Salt Electrolytes. J. Phys. Chem. B 2020, 124 (5), 861– 870, DOI: 10.1021/acs.jpcb.9b11051Google ScholarThere is no corresponding record for this reference.
- 32Lorenz, M.; Schönhoff, M. Evaluating Strategies to Enhance Li Transference in Salt-in-Ionic Liquid Electrolytes: Mixed Anions, Coordinating Cations, and High Salt Concentration. J. Phys. Chem. B 2024, 128 (11), 2782– 2791, DOI: 10.1021/acs.jpcb.3c08354Google ScholarThere is no corresponding record for this reference.
- 33Rosenwinkel, M. P.; Schönhoff, M. Polymer-Induced Inversion of the Li+ Drift Direction in Ionic Liquid-Based Ternary Polymer Electrolytes. Macromol. Chem. Phys. 2022, 223 (8), 2100320, DOI: 10.1002/macp.202100320Google ScholarThere is no corresponding record for this reference.
- 34Ackermann, F.; Schönhoff, M. Chelating Additives Reversing the Lithium Migration Direction in Ionic Liquid Electrolytes. J. Phys. Chem. C 2021, 125 (1), 266– 274, DOI: 10.1021/acs.jpcc.0c09828Google ScholarThere is no corresponding record for this reference.
- 35Nürnberg, P.; Atik, J.; Borodin, O.; Winter, M.; Paillard, E.; Schönhoff, M. Superionicity in Ionic-Liquid-Based Electrolytes Induced by Positive Ion-Ion Correlations. J. Am. Chem. Soc. 2022, 144 (10), 4657– 4666, DOI: 10.1021/jacs.2c00818Google ScholarThere is no corresponding record for this reference.
- 36Li, Z. H.; Xia, Q. L.; Liu, L. L.; Lei, G. T.; Xiao, Q. Z.; Gao, D. S.; Zhou, X. D. Effect of zwitterionic salt on the electrochemical properties of a solid polymer electrolyte with high temperature stability for lithium ion batteries. Electrochim. Acta 2010, 56 (2), 804– 809, DOI: 10.1016/j.electacta.2010.09.068Google ScholarThere is no corresponding record for this reference.
- 37D’Angelo, A. J.; Panzer, M. J. Decoupling the Ionic Conductivity and Elastic Modulus of Gel Electrolytes: Fully Zwitterionic Copolymer Scaffolds in Lithium Salt/Ionic Liquid Solutions. Adv. Energy Mater. 2018, 8 (26), 1801646, DOI: 10.1002/aenm.201801646Google ScholarThere is no corresponding record for this reference.
- 38Alsaedi, M. K.; Tadesse, M. Y.; Ganesan, V.; Panzer, M. J. Zwitterionic Polymer Ionogel Electrolytes Supported by Coulombic Cross-Links: Impacts of Alkali Metal Cation Identity. J. Phys. Chem. B 2024, 128 (13), 3273– 3281, DOI: 10.1021/acs.jpcb.3c08144Google ScholarThere is no corresponding record for this reference.
- 39Taylor, M. E.; Clarkson, D.; Greenbaum, S. G.; Panzer, M. J. Examining the Impact of Polyzwitterion Chemistry on Lithium Ion Transport in Ionogel Electrolytes. ACS Appl. Polym. Mater. 2021, 3 (5), 2635– 2645, DOI: 10.1021/acsapm.1c00229Google ScholarThere is no corresponding record for this reference.
- 40Tadesse, M. Y.; Marioni, N.; Zhang, Z. D.; Ganesan, V. Influence of Cation and Anion Chemistry on the Ionic Conductivity and Transference Number of Zwitterionic Polymer-Supported Ionic Liquid Electrolytes. ACS Appl. Energy Mater. 2025, 8 (6), 3314– 3326, DOI: 10.1021/acsaem.4c02650Google ScholarThere is no corresponding record for this reference.
- 41Stejskal, E. O.; Tanner, J. E. Spin Diffusion Measurements: Spin Echoes in the Presence of a Time-Dependent Field Gradient. J. Phys. Chem. 1965, 42 (1), 288– 292, DOI: 10.1063/1.1695690Google ScholarThere is no corresponding record for this reference.
- 42Carr, H. Y.; Purcell, E. M. Effects of diffusion on free precession in nuclear magnetic resonance experiments. Phys. Rev. 1954, 94 (3), 630– 638, DOI: 10.1103/PhysRev.94.630Google ScholarThere is no corresponding record for this reference.
- 43Meiboom, S.; Gill, D. Modified spin-echo method for measuring nuclear relaxation times. Rev. Sci. Instrum. 1958, 29 (8), 688– 691, DOI: 10.1063/1.1716296Google ScholarThere is no corresponding record for this reference.
- 44Kikuchi, M.; Terayama, Y.; Ishikawa, T.; Hoshino, T.; Kobayashi, M.; Ogawa, H.; Masunaga, H.; Koike, J.; Horigome, M.; Ishihara, K. Chain dimension of polyampholytes in solution and immobilized brush states. Polym. J. 2012, 44 (1), 121– 130, DOI: 10.1038/pj.2011.116Google ScholarThere is no corresponding record for this reference.
- 45Kobayashi, M.; Terayama, Y.; Kikuchi, M.; Takahara, A. Chain dimensions and surface characterization of superhydrophilic polymer brushes with zwitterion side groups. Soft Matter 2013, 9 (21), 5138– 5148, DOI: 10.1039/c3sm27700cGoogle ScholarThere is no corresponding record for this reference.
- 46Kubisa, P. Kinetics of radical polymerization in ionic liquids. Eur. Polym. J. 2020, 133, 109778, DOI: 10.1016/j.eurpolymj.2020.109778Google ScholarThere is no corresponding record for this reference.
- 47Tokuda, H.; Hayamizu, K.; Ishii, K.; Abu Bin Hasan Susan, M.; Watanabe, M. Physicochemical properties and structures of room temperature ionic liquids. 1. Variation of anionic species. J. Phys. Chem. B 2004, 108 (42), 16593– 16600, DOI: 10.1021/jp047480rGoogle ScholarThere is no corresponding record for this reference.
- 48Stolwijk, N. A.; Kösters, J.; Wiencierz, M.; Schönhoff, M. On the extraction of ion association data and transference numbers from ionic diffusivity and conductivity data in polymer electrolytes. Electrochim. Acta 2013, 102, 451– 458, DOI: 10.1016/j.electacta.2013.04.028Google ScholarThere is no corresponding record for this reference.
- 49Alía, J. M.; Edwards, H. G. M. Ion solvation and ion association in lithium trifluoromethanesulfonate solutions in three aprotic solvents. An FT-Raman spectroscopic study. Vib. Spectrosc. 2000, 24 (2), 185– 200, DOI: 10.1016/S0924-2031(00)00073-4Google ScholarThere is no corresponding record for this reference.
- 50Lassegues, J. C.; Grondin, J.; Talaga, D. Lithium solvation in bis(trifluoromethanesulfonyl)imide-based ionic liquids. Phys. Chem. Chem. Phys. 2006, 8 (48), 5629– 5632, DOI: 10.1039/B615127BGoogle ScholarThere is no corresponding record for this reference.
- 51Lassègues, J.-C.; Grondin, J.; Aupetit, C.; Johansson, P. Spectroscopic Identification of the Lithium Ion Transporting Species in LiTFSI-Doped Ionic Liquids. J. Phys. Chem. A 2009, 113 (1), 305– 314, DOI: 10.1021/jp806124wGoogle ScholarThere is no corresponding record for this reference.
- 52Wieck, K. W.; Panzer, M. J. Ionogel Electrolytes Supported by Zwitterionic Copolymers Featuring Lithium Ion-Mediated. Noncovalent Cross-Links. ACS Appl. Polym. Mater. 2023, 5 (4), 2887– 2894, DOI: 10.1021/acsapm.3c00110Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
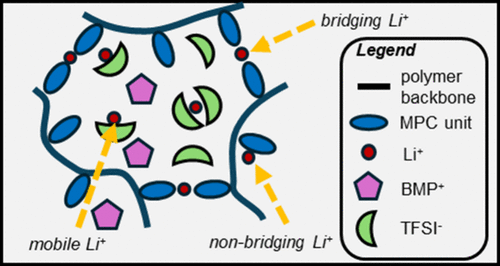
Scheme 1
 Scheme 1. Structures of the Main Electrolyte Components (Top) and Illustration of Different Li+ Species within a p(MPC)-Supported Ionogela
Scheme 1. Structures of the Main Electrolyte Components (Top) and Illustration of Different Li+ Species within a p(MPC)-Supported IonogelaaBridging Li+ that acts as noncovalent cross-linker between ZI units, non-bridging Li+ coordinated to a single ZI unit, and mobile Li+ in solution (bottom).
Figure 1

Figure 1. Total ionic conductivity at room temperature (25 °C) as a function of p(MPC) wt %.
Figure 2

Figure 2. (a) Self-diffusion coefficients of the nuclei (1H, 19F, 7Li) obtained from PFG-NMR at room temperature (25 °C) as function of p(MPC) content. (b) Activation energies of diffusion as a function of p(MPC) content measured over a temperature range of 25–65 °C. The dotted lines are merely guides to the eye.
Figure 3

Figure 3. Li+ ion apparent transference number as function of p(MPC) content.
Figure 4

Figure 4. (a) 7Li NMR spectra, (b) peak integral, (c) line width, and (d) 7Li spin–spin relaxation rate R2 determined by a stimulated echo sequence (black) and R2* determined by line width analysis (red star) for 1 M LiTFSI/BMP TFSI solution (0 wt % p(MPC)), 2 wt % p(MPC) solution, and 4–8 wt % p(MPC) free-standing ionogels.
Figure 5

Figure 5. (a) Raman spectra and (b) Li+ coordination number (coordinated anions per Li+) as extracted from the fraction of coordinated anions in dependence on p(MPC) content. The green dotted line in a) marks the position of the band of coordinated TFSI– and the black arrow represents the increase in p(MPC) content from 2 wt % (green spectrum) to 14 wt % (dark blue).
Figure 6
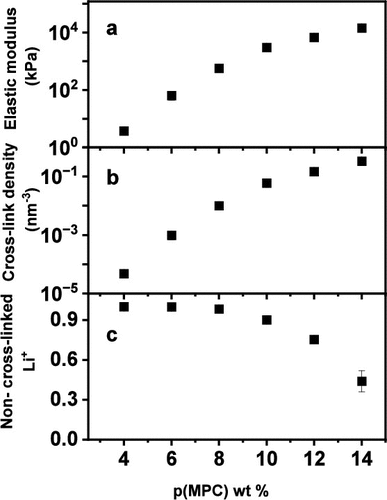
Figure 6. (a) Elastic modulus values for ionogels containing 4 to 14 wt % p(MPC). (b) Calculated cross-link density within the ionogels, representing the number of Li+ ion-mediated noncovalent cross-links per nm3, and (c) the corresponding fraction of Li+ that is not participating in noncovalent cross-links.
Figure 7

Figure 7. (a) Distribution of Li+ in different sites; (b) – (d) Li+ coordination scenarios at 0, 8, and 14 wt % p(MPC), showing free, immobilized, and cross-linking Li+ ions. For a legend of the symbols, see Scheme 1. Note that the MPC:Li+ molar ratio and number of coordinating anions are shown according to scale, while the total number of IL cations and anions is not to scale to enhance clarity.
References
This article references 52 other publications.
- 1Kim, T.; Song, W.; Son, D.-Y.; Ono, L. K.; Qi, Y. Lithium-ion batteries: outlook on present, future, and hybridized technologies. J. Mater. Chem. A 2019, 7 (7), 2942– 2964, DOI: 10.1039/C8TA10513HThere is no corresponding record for this reference.
- 2Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sources 2013, 226, 272– 288, DOI: 10.1016/j.jpowsour.2012.10.060There is no corresponding record for this reference.
- 3Wang, Y.; Zhong, W.-H. Development of Electrolytes towards Achieving Safe and High-Performance Energy-Storage Devices: A Review. ChemElectroChem. 2015, 2 (1), 22– 36, DOI: 10.1002/celc.201402277There is no corresponding record for this reference.
- 4Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210– 224, DOI: 10.1016/j.jpowsour.2012.02.038There is no corresponding record for this reference.
- 5Goodenough, J. B.; Kim, Y. Challenges for Rechargeable Li Batteries. Chem. Mater. 2010, 22 (3), 587– 603, DOI: 10.1021/cm901452zThere is no corresponding record for this reference.
- 6Balducci, A. Ionic Liquids in Lithium-Ion Batteries. Top. Curr. Chem. 2017, 375 (2), 20, DOI: 10.1007/s41061-017-0109-8There is no corresponding record for this reference.
- 7Francis, C. F. J.; Kyratzis, I. L.; Best, A. S. Lithium-Ion Battery Separators for Ionic-Liquid Electrolytes: A Review. Adv. Mater. 2020, 32 (18), 1904205, DOI: 10.1002/adma.201904205There is no corresponding record for this reference.
- 8Tang, X.; Lv, S. Y.; Jiang, K.; Zhou, G. H.; Liu, X. M. Recent development of ionic liquid-based electrolytes in lithium-ion batteries. J. Power Sources 2022, 542, 231792, DOI: 10.1016/j.jpowsour.2022.231792There is no corresponding record for this reference.
- 9Liu, J.; Yao, Y. C.; Zhang, S. Z.; Zhang, K. Y.; Peng, C. J.; Liu, H. L. Application of Ionic Liquids in Lithium Metal Batteries. Prog. Chem. 2025, 37 (5), 788– 800There is no corresponding record for this reference.
- 10Watanabe, M.; Thomas, M. L.; Zhang, S. G.; Ueno, K.; Yasuda, T.; Dokko, K. Application of Ionic Liquids to Energy Storage and Conversion Materials and Devices. Chem. Rev. 2017, 117 (10), 7190– 7239, DOI: 10.1021/acs.chemrev.6b00504There is no corresponding record for this reference.
- 11Armand, M.; Endres, F.; MacFarlane, D. R.; Ohno, H.; Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 2009, 8 (8), 621– 629, DOI: 10.1038/nmat2448There is no corresponding record for this reference.
- 12Lewandowski, A.; Świderska-Mocek, A. Ionic liquids as electrolytes for Li-ion batteries─An overview of electrochemical studies. J. Power Sources 2009, 194 (2), 601– 609, DOI: 10.1016/j.jpowsour.2009.06.089There is no corresponding record for this reference.
- 13Piatti, E.; Guglielmero, L.; Tofani, G.; Mezzetta, A.; Guazzelli, L.; D’Andrea, F.; Roddaro, S.; Pomelli, C. S. Ionic liquids for electrochemical applications: Correlation between molecular structure and electrochemical stability window. J. Mol. Liq. 2022, 364, 120001, DOI: 10.1016/j.molliq.2022.120001There is no corresponding record for this reference.
- 14Hayyan, M.; Mjalli, F. S.; Hashim, M. A.; AlNashef, I. M.; Mei, T. X. Investigating the electrochemical windows of ionic liquids. J. Ind. Eng. Chem. 2013, 19 (1), 106– 112, DOI: 10.1016/j.jiec.2012.07.011There is no corresponding record for this reference.
- 15Le Bideau, J.; Viau, L.; Vioux, A. Ionogels, ionic liquid based hybrid materials. Chem. Soc. Rev. 2011, 40 (2), 907– 925, DOI: 10.1039/C0CS00059KThere is no corresponding record for this reference.
- 16Giffin, G. A. Ionic liquid-based electrolytes for ″beyond lithium″ battery technologies. J. Mater. Chem. A 2016, 4 (35), 13378– 13389, DOI: 10.1039/C6TA05260FThere is no corresponding record for this reference.
- 17Galiński, M.; Lewandowski, A.; Stępniak, I. Ionic liquids as electrolytes. Electrochim. Acta 2006, 51 (26), 5567– 5580, DOI: 10.1016/j.electacta.2006.03.016There is no corresponding record for this reference.
- 18Hayamizu, K.; Aihara, Y.; Nakagawa, H.; Nukuda, T.; Price, W. S. Ionic conduction and ion diffusion in binary room-temperature ionic liquids composed of emim BF4 and LiBF4. J. Phys. Chem. B 2004, 108 (50), 19527– 19532, DOI: 10.1021/jp0476601There is no corresponding record for this reference.
- 19Saito, Y.; Umecky, T.; Niwa, J.; Sakai, T.; Maeda, S. Existing condition and migration property of ions in lithium electrolytes with ionic liquid solvent. J. Phys. Chem. B 2007, 111 (40), 11794– 11802, DOI: 10.1021/jp072998rThere is no corresponding record for this reference.
- 20Nicotera, I.; Oliviero, C.; Henderson, W. A.; Appetecchi, G. B.; Passerini, S. NMR investigation of ionic liquid-LiX mixtures: Pyrrolidinium cations and TFSI- anions. J. Phys. Chem. B 2005, 109 (48), 22814– 22819, DOI: 10.1021/jp053799fThere is no corresponding record for this reference.
- 21Castiglione, F.; Famulari, A.; Raos, G.; Meille, S. V.; Mele, A.; Appetecchi, G. B.; Passerini, S. Pyrrolidinium-Based Ionic Liquids Doped with Lithium Salts: How Does Li+ Coordination Affect Its Diffusivity?. J. Phys. Chem. B 2014, 118 (47), 13679– 13688, DOI: 10.1021/jp509387rThere is no corresponding record for this reference.
- 22Tong, J. H.; Wu, S. L.; von Solms, N.; Liang, X. D.; Huo, F.; Zhou, Q.; He, H. Y.; Zhang, S. J. The Effect of Concentration of Lithium Salt on the Structural and Transport Properties of Ionic Liquid-Based Electrolytes. Front. Chem. 2020, 7, 945, DOI: 10.3389/fchem.2019.00945There is no corresponding record for this reference.
- 23Umebayashi, Y.; Mori, S.; Fujii, K.; Tsuzuki, S.; Seki, S.; Hayamizu, K.; Ishiguro, S. Raman Spectroscopic Studies and Ab Initio Calculations on Conformational Isomerism of 1-Butyl-3-methylimidazolium Bis-(trifluoromethanesulfonyl)amide Solvated to a Lithium Ion in Ionic Liquids: Effects of the Second Solvation Sphere of the Lithium Ion. J. Phys. Chem. B 2010, 114 (19), 6513– 6521, DOI: 10.1021/jp100898hThere is no corresponding record for this reference.
- 24Méndez-Morales, T.; Carrete, J.; Bouzón-Capelo, S.; Pérez-Rodríguez, M.; Cabeza, O.; Gallego, L. J.; Varela, L. M. MD Simulations of the Formation of Stable Clusters in Mixtures of Alkaline Salts and Imidazolium-Based Ionic Liquids. J. Phys. Chem. B 2013, 117 (11), 3207– 3220, DOI: 10.1021/jp312669rThere is no corresponding record for this reference.
- 25Umebayashi, Y.; Mitsugi, T.; Fukuda, S.; Fujimori, T.; Fujii, K.; Kanzaki, R.; Takeuchi, M.; Ishiguro, S. I. Lithium ion solvation in room-temperature ionic liquids involving bis(trifluoromethanesulfonyl) imide anion studied by Raman Spectroscopy and DFT calculations. J. Phys. Chem. B 2007, 111 (45), 13028– 13032, DOI: 10.1021/jp076869mThere is no corresponding record for this reference.
- 26Pitawala, J.; Martinelli, A.; Johansson, P.; Jacobsson, P.; Matic, A. Coordination and interactions in a Li-salt doped ionic liquid. J. Non-Cryst. Solids 2015, 407, 318– 323, DOI: 10.1016/j.jnoncrysol.2014.08.043There is no corresponding record for this reference.
- 27Gouverneur, M.; Schmidt, F.; Schönhoff, M. Negative effective Li transference numbers in Li salt/ionic liquid mixtures: does Li drift in the ″Wrong″ direction?. Phys. Chem. Chem. Phys. 2018, 20 (11), 7470– 7478, DOI: 10.1039/C7CP08580JThere is no corresponding record for this reference.
- 28Brinkkötter, M.; Giffin, G. A.; Moretti, A.; Jeong, S.; Passerini, S.; Schönhoff, M. Relevance of ion clusters for Li transport at elevated salt concentrations in [Pyr(12O1)][FTFSI] ionic liquid-based electrolytes. Chem. Commun. 2018, 54 (34), 4278– 4281, DOI: 10.1039/C8CC01416GThere is no corresponding record for this reference.
- 29Molinari, N.; Mailoa, J. P.; Kozinsky, B. General Trend of a Negative Li Effective Charge in Ionic Liquid Electrolytes. J. Phys. Chem. Lett. 2019, 10 (10), 2313– 2319, DOI: 10.1021/acs.jpclett.9b00798There is no corresponding record for this reference.
- 30Brinkkötter, M.; Mariani, A.; Jeong, S.; Passerini, S.; Schönhoff, M. Ionic Liquid in Li Salt Electrolyte: Modifying the Li+ Transport Mechanism by Coordination to an Asymmetric Anion. Adv. Energy Sustainability Res. 2021, 2 (2), 2000078, DOI: 10.1002/aesr.202000078There is no corresponding record for this reference.
- 31Nürnberg, P.; Lozinskaya, E. I.; Shaplov, A. S.; Schönhoff, M. Li Coordination of a Novel Asymmetric Anion in Ionic Liquid-in-Li Salt Electrolytes. J. Phys. Chem. B 2020, 124 (5), 861– 870, DOI: 10.1021/acs.jpcb.9b11051There is no corresponding record for this reference.
- 32Lorenz, M.; Schönhoff, M. Evaluating Strategies to Enhance Li Transference in Salt-in-Ionic Liquid Electrolytes: Mixed Anions, Coordinating Cations, and High Salt Concentration. J. Phys. Chem. B 2024, 128 (11), 2782– 2791, DOI: 10.1021/acs.jpcb.3c08354There is no corresponding record for this reference.
- 33Rosenwinkel, M. P.; Schönhoff, M. Polymer-Induced Inversion of the Li+ Drift Direction in Ionic Liquid-Based Ternary Polymer Electrolytes. Macromol. Chem. Phys. 2022, 223 (8), 2100320, DOI: 10.1002/macp.202100320There is no corresponding record for this reference.
- 34Ackermann, F.; Schönhoff, M. Chelating Additives Reversing the Lithium Migration Direction in Ionic Liquid Electrolytes. J. Phys. Chem. C 2021, 125 (1), 266– 274, DOI: 10.1021/acs.jpcc.0c09828There is no corresponding record for this reference.
- 35Nürnberg, P.; Atik, J.; Borodin, O.; Winter, M.; Paillard, E.; Schönhoff, M. Superionicity in Ionic-Liquid-Based Electrolytes Induced by Positive Ion-Ion Correlations. J. Am. Chem. Soc. 2022, 144 (10), 4657– 4666, DOI: 10.1021/jacs.2c00818There is no corresponding record for this reference.
- 36Li, Z. H.; Xia, Q. L.; Liu, L. L.; Lei, G. T.; Xiao, Q. Z.; Gao, D. S.; Zhou, X. D. Effect of zwitterionic salt on the electrochemical properties of a solid polymer electrolyte with high temperature stability for lithium ion batteries. Electrochim. Acta 2010, 56 (2), 804– 809, DOI: 10.1016/j.electacta.2010.09.068There is no corresponding record for this reference.
- 37D’Angelo, A. J.; Panzer, M. J. Decoupling the Ionic Conductivity and Elastic Modulus of Gel Electrolytes: Fully Zwitterionic Copolymer Scaffolds in Lithium Salt/Ionic Liquid Solutions. Adv. Energy Mater. 2018, 8 (26), 1801646, DOI: 10.1002/aenm.201801646There is no corresponding record for this reference.
- 38Alsaedi, M. K.; Tadesse, M. Y.; Ganesan, V.; Panzer, M. J. Zwitterionic Polymer Ionogel Electrolytes Supported by Coulombic Cross-Links: Impacts of Alkali Metal Cation Identity. J. Phys. Chem. B 2024, 128 (13), 3273– 3281, DOI: 10.1021/acs.jpcb.3c08144There is no corresponding record for this reference.
- 39Taylor, M. E.; Clarkson, D.; Greenbaum, S. G.; Panzer, M. J. Examining the Impact of Polyzwitterion Chemistry on Lithium Ion Transport in Ionogel Electrolytes. ACS Appl. Polym. Mater. 2021, 3 (5), 2635– 2645, DOI: 10.1021/acsapm.1c00229There is no corresponding record for this reference.
- 40Tadesse, M. Y.; Marioni, N.; Zhang, Z. D.; Ganesan, V. Influence of Cation and Anion Chemistry on the Ionic Conductivity and Transference Number of Zwitterionic Polymer-Supported Ionic Liquid Electrolytes. ACS Appl. Energy Mater. 2025, 8 (6), 3314– 3326, DOI: 10.1021/acsaem.4c02650There is no corresponding record for this reference.
- 41Stejskal, E. O.; Tanner, J. E. Spin Diffusion Measurements: Spin Echoes in the Presence of a Time-Dependent Field Gradient. J. Phys. Chem. 1965, 42 (1), 288– 292, DOI: 10.1063/1.1695690There is no corresponding record for this reference.
- 42Carr, H. Y.; Purcell, E. M. Effects of diffusion on free precession in nuclear magnetic resonance experiments. Phys. Rev. 1954, 94 (3), 630– 638, DOI: 10.1103/PhysRev.94.630There is no corresponding record for this reference.
- 43Meiboom, S.; Gill, D. Modified spin-echo method for measuring nuclear relaxation times. Rev. Sci. Instrum. 1958, 29 (8), 688– 691, DOI: 10.1063/1.1716296There is no corresponding record for this reference.
- 44Kikuchi, M.; Terayama, Y.; Ishikawa, T.; Hoshino, T.; Kobayashi, M.; Ogawa, H.; Masunaga, H.; Koike, J.; Horigome, M.; Ishihara, K. Chain dimension of polyampholytes in solution and immobilized brush states. Polym. J. 2012, 44 (1), 121– 130, DOI: 10.1038/pj.2011.116There is no corresponding record for this reference.
- 45Kobayashi, M.; Terayama, Y.; Kikuchi, M.; Takahara, A. Chain dimensions and surface characterization of superhydrophilic polymer brushes with zwitterion side groups. Soft Matter 2013, 9 (21), 5138– 5148, DOI: 10.1039/c3sm27700cThere is no corresponding record for this reference.
- 46Kubisa, P. Kinetics of radical polymerization in ionic liquids. Eur. Polym. J. 2020, 133, 109778, DOI: 10.1016/j.eurpolymj.2020.109778There is no corresponding record for this reference.
- 47Tokuda, H.; Hayamizu, K.; Ishii, K.; Abu Bin Hasan Susan, M.; Watanabe, M. Physicochemical properties and structures of room temperature ionic liquids. 1. Variation of anionic species. J. Phys. Chem. B 2004, 108 (42), 16593– 16600, DOI: 10.1021/jp047480rThere is no corresponding record for this reference.
- 48Stolwijk, N. A.; Kösters, J.; Wiencierz, M.; Schönhoff, M. On the extraction of ion association data and transference numbers from ionic diffusivity and conductivity data in polymer electrolytes. Electrochim. Acta 2013, 102, 451– 458, DOI: 10.1016/j.electacta.2013.04.028There is no corresponding record for this reference.
- 49Alía, J. M.; Edwards, H. G. M. Ion solvation and ion association in lithium trifluoromethanesulfonate solutions in three aprotic solvents. An FT-Raman spectroscopic study. Vib. Spectrosc. 2000, 24 (2), 185– 200, DOI: 10.1016/S0924-2031(00)00073-4There is no corresponding record for this reference.
- 50Lassegues, J. C.; Grondin, J.; Talaga, D. Lithium solvation in bis(trifluoromethanesulfonyl)imide-based ionic liquids. Phys. Chem. Chem. Phys. 2006, 8 (48), 5629– 5632, DOI: 10.1039/B615127BThere is no corresponding record for this reference.
- 51Lassègues, J.-C.; Grondin, J.; Aupetit, C.; Johansson, P. Spectroscopic Identification of the Lithium Ion Transporting Species in LiTFSI-Doped Ionic Liquids. J. Phys. Chem. A 2009, 113 (1), 305– 314, DOI: 10.1021/jp806124wThere is no corresponding record for this reference.
- 52Wieck, K. W.; Panzer, M. J. Ionogel Electrolytes Supported by Zwitterionic Copolymers Featuring Lithium Ion-Mediated. Noncovalent Cross-Links. ACS Appl. Polym. Mater. 2023, 5 (4), 2887– 2894, DOI: 10.1021/acsapm.3c00110There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpcb.5c06389.
Supporting data such as 1H NMR spectra of dissolved ionogels, temperature-dependent diffusion coefficients, spin relaxation rates, deconvolution of Raman spectra and calculation of coordination number, estimation of cross-linking density from DMA data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



