Direct Observation of Hydroxyls Formed from Water and Oxygen on Ag(100)Click to copy article linkArticle link copied!
- Cole A. EastonCole A. EastonDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Cole A. Easton
- Sarah M. StrattonSarah M. StrattonDepartment of Chemical and Biomolecular Engineering, Tulane University, New Orleans, Louisiana 70115, United StatesMore by Sarah M. Stratton
- Nima RajabiNima RajabiDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Nima Rajabi
- Nishadi AmarathungaNishadi AmarathungaDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Nishadi Amarathunga
- Elizabeth E. HappelElizabeth E. HappelDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Elizabeth E. Happel
- Avery S. DanielsAvery S. DanielsDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Avery S. Daniels
- Adrian HuntAdrian HuntNational Synchrotron Light Source II, Brookhaven National Laboratory, Upton, New York 11973, United StatesMore by Adrian Hunt
- Hojoon LimHojoon LimNational Synchrotron Light Source II, Brookhaven National Laboratory, Upton, New York 11973, United StatesMore by Hojoon Lim
- Vinita LalVinita LalDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Vinita Lal
- Nipun T.S.K. DewageNipun T.S.K. DewageDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Nipun T.S.K. Dewage
- Dennis MeierDennis MeierDepartment of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by Dennis Meier
- Iradwikanari Waluyo*Iradwikanari Waluyo*Email: [email protected]National Synchrotron Light Source II, Brookhaven National Laboratory, Upton, New York 11973, United StatesMore by Iradwikanari Waluyo
- Matthew M. Montemore*Matthew M. Montemore*Email: [email protected]Department of Chemical and Biomolecular Engineering, Tulane University, New Orleans, Louisiana 70115, United StatesMore by Matthew M. Montemore
- E. Charles H. Sykes*E. Charles H. Sykes*Email: [email protected]Department of Chemistry, Tufts University, Medford, Massachusetts 02155, United StatesMore by E. Charles H. Sykes
Abstract
The interaction of oxygen with silver is a key descriptor of the catalytic reactivity of silver nanoparticles which are ubiquitous in large-scale partial oxidation reactions like ethylene epoxidation. Despite Ag(100) being proposed as the most selective facet, it is less studied than (111) and (110) surfaces. Using scanning tunneling microscopy and synchrotron X-ray photoelectron spectroscopy, we report that, in addition to the well-known O adatoms formed from O2 dissociation on Ag(100), hydroxyl groups (OH), at a binding energy of ∼ 531 eV, are also present. The O/OH ratio depends on exposure to water and surface temperature. These assignments are consistent with our density functional theory calculations, which indicate that the formation of two OH groups from an O atom and H2O molecule is exothermic. These results indicate that, in addition to O, OH is present even under ultrahigh vacuum conditions and therefore should be considered in proposed catalytic pathways.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Figure 1
Figure 1. Products resulting from O2 dissociation on Ag(100). (A) 78 K STM image of ∼ 1% ML depressions (red arrow) and ∼ 1.5% ML protrusions (blue arrow) resulting from a 1000 L dose of O2 on Ag(100) at room temperature. Imaging conditions: 10 mV, 1 nA. (B) O 1s XPS spectra of clean Ag(100), 12,000 L O2 300 K (7% O530 and 4% O531) on Ag(100), and subsequent anneal. (C) Plot of the coverage of each O 1s component from the fits in A.
Figure 2
Figure 2. 78 K STM images of the Ag(100) surface before and after exposure to O2 under different imaging conditions. (A) Depiction of oxygen dissociation and reaction with water on the Ag(100) surface and associated reaction energies and activation barriers. Dissociation barriers are shown in blue (Ediss), and adsorption energies are shown beneath in black (Eads). All energy values are relative to gas phase O2. (B) 78 K STM image of the bare Ag(100) surface before exposure to O2. Imaging conditions: 300 mV, 300 pA. (C)-(D) Images of O adatoms (depressions) and OH groups (protrusions) on Ag(100) imaged at (C) 3.00 V, 300 pA, (D) 300 mV, 300 pA. (C) and (D) are of the same area. At 3.00 V, only protrusions are visible, and at 300 mV only depressions are visible. Surface coverages were 1.0% O and 1.5% OH in (C) and (D). All images are 50 × 50 nm2, scale bar shown in C.
Figure 3
Figure 3. Binding site characterization of O adatoms and OH groups on O–Ag(100). (A) Atomic resolution 78 K STM image of an oxygen adatom on Ag(100) imaged at 10 mV, 1 nA. (B) The same area of the surface under typical resolution imaged at 100 mV, 1 nA. Both STM images are the same size, and a scale bar is shown in (A). (C) STM image of O and OH on Ag(100) imaged at 25 mV, 1 nA, and a scale bar is shown. Insets in panel C are (top) Simulated STM image of an OH group on Ag(100), (bottom) simulated STM of an O adatom on Ag(100). (D) and (E) are the calculated preferred binding sites for (D) OH and (E) O atoms. (F) STM image of O and OH with the Ag(100) 4-fold hollow lattice corrected for drift overlaid. The centers of several OH groups are marked with blue circles or arrows. The locations where the grid lines cross are the 4-fold hollow sites. Imaging conditions: 25 mV, 1 nA, and a scale bar is shown. Images were acquired at 78 K.
Figure 4
Figure 4. STM Voltage Pulse Experiments. (A) Depiction of STM tip dissociating OH into O and H using a localized voltage pulse. (B) 78 K STM image of the Ag(100) surface before voltage pulses. OH groups appear as faint protrusions and are marked by blue arrows. (C) 78 K STM image of the same area after 4 V pulses. Each OH is now a depression in the same surface location. Oxygen was dosed at 300 K, and the pulse locations are indicated by the blue arrows. (D) Depiction of the STM tip dissociating O2 into two O adatoms via a voltage pulse. (E) LT-STM image of the Ag(100) surface with oxygen dosed at 78 K. Oxygen is present as molecular oxygen and diffuses too fast at 78 K to be imaged. The pulse location is indicated by the blue arrow. (F) LT-STM image of the same area following one 8 V pulse. Oxygen adatoms have formed as a result of the pulse. No further changes occurred after pulsing on the O adatoms. Imaging conditions: 1.0 nA and 50 mV.
Figure 5
Figure 5. Reaction of O atoms with adsorbed H2O. (A) 78 K STM image of 1000 L O2 dosed at 300 K on Ag(100) imaged at 1 V. O adatoms are visible as depressions. (B) Same area imaged at 3 V where OH groups are visible as protrusions. (C) STM image of O–Ag(100) surface after a 400 K anneal scanned at 1 V. (D) Same area scanned at 3 V showing a significant reduction in the number of OH species. (E) STM image of the surface after 3 L H2O was dosed at 78 K, annealed to 400 K, and then imaged at 1 V at 78 K. (F) Same area imaged at 3.0 V showing an increase in the number of OH species. (G) Quantification of the surface coverages of O and OH for each stage of the experiment. (H) Schematic summarizing O2 dissociation, OH formation, and water desorption from Ag(100). Tunneling current was constant for all STM images at 300 pA. All images are 50 × 50 nm2.
Experimental Methods
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpclett.5c03296.
Details of experimental methods and additional STM, DFT and XPS data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
The authors would like to acknowledge Phillip Christopher for helpful discussions and Allison Zupan for help at the synchrotron. Financial support for the experimental work at Tufts University was provided by the US Department of Energy, BES, Catalysis Science program under contract No. DE-SC0021196. SMS acknowledges support from the U.S. – Israel Center for Fossil Fuels, administered by the BIRD foundation. MMM was supported by the National Science Foundation through grant CHE-2334969. This work used resources of the 23-ID-2 (IOS) beamline of the National Synchrotron Light Source II, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-SC0012704. All authors declare no competing interests.
References
This article references 69 other publications.
- 1Vodyankina, O. V. Silver Catalysts for the Partial Oxidation of Alcohols. Catal. Ind. 2022, 14 (3), 314– 326, DOI: 10.1134/S2070050422030059Google ScholarThere is no corresponding record for this reference.
- 2Torbina, V. V.; Vodyankin, A. A.; Ten, S.; Mamontov, G. V.; Salaev, M. A.; Sobolev, V. I.; Vodyankina, O. V. Ag-Based Catalysts in Heterogeneous Selective Oxidation of Alcohols: A Review. Catalysts 2018, 8 (10), 447 DOI: 10.3390/catal8100447Google ScholarThere is no corresponding record for this reference.
- 3Lockemeyer, J. R.; Lohr, T. L. Ethylene Oxide Catalysis Under Commercial Conditions – A Guide for Researchers. ChemCatChem 2023, 15 (13), e202201511 DOI: 10.1002/cctc.202201511Google ScholarThere is no corresponding record for this reference.
- 4Salmi, T.; Roche, M.; Hernández Carucci, J.; Eränen, K.; Murzin, D. Ethylene Oxide – Kinetics and Mechanism. Curr. Opin. Chem. Eng. 2012, 1 (3), 321– 327, DOI: 10.1016/j.coche.2012.06.002Google ScholarThere is no corresponding record for this reference.
- 5Montemore, M. M.; Spronsen, M. A. van; Madix, R. J.; Friend, C. M. O2 Activation by Metal Surfaces: Implications for Bonding and Reactivity on Heterogeneous Catalysts. Chem. Rev. 2018, 118 (5), 2816– 2862, DOI: 10.1021/acs.chemrev.7b00217Google ScholarThere is no corresponding record for this reference.
- 6Pu, T.; Setiawan, A.; Rangarajan, S.; Wachs, I. E. Progress and Perspective on the Fundamental Understanding of Structure–Activity/Selectivity Relationships for Ag Catalyzed Ethylene Epoxidation. Catal. Today 2025, 455, 115301 DOI: 10.1016/j.cattod.2025.115301Google ScholarThere is no corresponding record for this reference.
- 7Carbonio, E. A.; Rocha, T. C. R.; Klyushin, A. Yu.; Píš, I.; Magnano, E.; Nappini, S.; Piccinin, S.; Knop-Gericke, A.; Schlögl, R.; Jones, T. E. Are Multiple Oxygen Species Selective in Ethylene Epoxidation on Silver?. Chem. Sci. 2018, 9 (4), 990– 998, DOI: 10.1039/C7SC04728BGoogle ScholarThere is no corresponding record for this reference.
- 8Liu, J.-X.; Lu, S.; Ann, S.-B.; Linic, S. Mechanisms of Ethylene Epoxidation over Silver from Machine Learning-Accelerated First-Principles Modeling and Microkinetic Simulations. ACS Catal. 2023, 13 (13), 8955– 8962, DOI: 10.1021/acscatal.3c00410Google ScholarThere is no corresponding record for this reference.
- 9Rocha, T. C. R.; Hävecker, M.; Knop-Gericke, A.; Schlögl, R. Promoters in Heterogeneous Catalysis: The Role of Cl on Ethylene Epoxidation over Ag. J. Catal. 2014, 312, 12– 16, DOI: 10.1016/j.jcat.2014.01.002Google ScholarThere is no corresponding record for this reference.
- 10Henderson, M. The Interaction of Water with Solid Surfaces: Fundamental Aspects Revisited. Surf. Sci. Rep. 2002, 46 (1–8), 1– 308, DOI: 10.1016/S0167-5729(01)00020-6Google ScholarThere is no corresponding record for this reference.
- 11Bao, X.; Muhler, M.; Pettinger, B.; Uchida, Y.; Lehmpfuhl, G.; Schlögl, R.; Ertl, G. The Effect of Water on the Formation of Strongly Bound Oxygen on Silver Surfaces. Catal. Lett. 1995, 32 (1–2), 171– 183, DOI: 10.1007/BF00806112Google ScholarThere is no corresponding record for this reference.
- 12Klaua, M.; Madey, T. E. The Adsorption of H2O on Clean and Oxygen-Dosed Silver Single Crystal Surfaces. Surf. Sci. 1984, 136 (1), L42– L50, DOI: 10.1016/0039-6028(84)90650-2Google ScholarThere is no corresponding record for this reference.
- 13Guillemot, L.; Bobrov, K. On the Formation of OH Ordered Layers by Dissociation of H2O on an Oxygen Covered Ag(110) Surface: An STM Investigation. Surf. Sci. 2007, 601 (3), 871– 875, DOI: 10.1016/j.susc.2006.11.027Google ScholarThere is no corresponding record for this reference.
- 14Chu, Y.; Liang, H.; Lin, Q.; Huang, H.; Che, P.; Han, Y. Effect of Water Vapor on the Structure and Performance of Silver Catalysts for Ethylene Oxidation. Chem. Eng. J. 2025, 518, 164442 DOI: 10.1016/j.cej.2025.164442Google ScholarThere is no corresponding record for this reference.
- 15Liu, W.; Eliyas, A.; Petrov, L. Influence of Feed Water Vapour on the Selective Oxidation of Ethylene over Silver Catalyst. Appl. Catal. 1990, 61 (1), 265– 274, DOI: 10.1016/S0166-9834(00)82149-1Google ScholarThere is no corresponding record for this reference.
- 16Bowker, M.; Barteau, M. A.; Madix, R. J. Oxygen Induced Adsorption and Reaction of H2, H2O, CO and CO2 on Single Crystal Ag(110). Surf. Sci. 1980, 92 (2–3), 528– 548, DOI: 10.1016/0039-6028(80)90221-6Google ScholarThere is no corresponding record for this reference.
- 17Campbell, C. T.; Paffett, M. T. The Interactions of O2, CO and CO2 with Ag(110). Surf. Sci. 1984, 143 (2–3), 517– 535, DOI: 10.1016/0039-6028(84)90557-0Google ScholarThere is no corresponding record for this reference.
- 18Codeço, C. F. S.; Klyushin, A. Y.; Carbonio, E. A.; Knop-Gericke, A.; Schlögl, R.; Jones, T.; Rocha, T. C. R. Insights into the Electronic Structure of Hydroxyl on Ag(110) under near Ambient Conditions. Phys. Chem. Chem. Phys. 2022, 24 (15), 8832– 8838, DOI: 10.1039/D1CP02929KGoogle ScholarThere is no corresponding record for this reference.
- 19Stuve, E. M.; Madix, R. J.; Sexton, B. A. The Adsorption and Reaction of H2O on Clean and Oxygen Covered Ag(110). Surf. Sci. 1981, 111 (1), 11– 25, DOI: 10.1016/0039-6028(81)90471-4Google ScholarThere is no corresponding record for this reference.
- 20Savio, L.; Smerieri, M.; Vattuone, L.; Gussoni, A.; Tassistro, C.; Rocca, M. STM Study of Hydroxyl Formation at OAg(100). Phys. Rev. B 2006, 74 (23), 235412 DOI: 10.1103/PhysRevB.74.235412Google ScholarThere is no corresponding record for this reference.
- 21Wachs, I. E.; Keleman, S. R. The Interaction of Ethylene with Surface Carbonate and Hydroxide Intermediates on Silver. J. Catal. 1981, 71 (1), 78– 87 DOI: 10.1016/0021-9517(81)90203-7 .Google ScholarThere is no corresponding record for this reference.
- 22Bange, K.; Madey, T. E.; Sass, J. K.; Stuve, E. M. The Adsorption of Water and Oxygen on Ag(110): A Study of the Interactions among Water Molecules, Hydroxyl Groups, and Oxygen Atoms. Surf. Sci. 1987, 183 (3), 334– 362, DOI: 10.1016/S0039-6028(87)80215-7Google ScholarThere is no corresponding record for this reference.
- 23Hu, Z.-M.; Nakatsuji, H. Adsorption and Disproportionation Reaction of OH on Ag Surfaces: Dipped Adcluster Model Study. Surf. Sci. 1999, 425 (2–3), 296– 312, DOI: 10.1016/S0039-6028(99)00215-0Google ScholarThere is no corresponding record for this reference.
- 24Qin, C.; Whitten, J. L. Adsorption of O, H, OH, and H2O on Ag(100). J. Phys. Chem. B 2005, 109 (18), 8852– 8856, DOI: 10.1021/jp044067aGoogle ScholarThere is no corresponding record for this reference.
- 25Rocca, M.; Savio, L.; Vattuone, L.; Burghaus, U.; Palomba, V.; Novelli, N.; Mongeot, F. B. de; Valbusa, U.; Gunnella, R.; Comelli, G.; Baraldi, A.; Lizzit, S.; Paolucci, G. Phase Transition of Dissociatively Adsorbed Oxygen on Ag(001). Phys. Rev. B 2000, 61 (1), 213– 227, DOI: 10.1103/PhysRevB.61.213Google ScholarThere is no corresponding record for this reference.
- 26Hsieh, M.-F.; Lin, D.-S.; Gawronski, H.; Morgenstern, K. Hard Repulsive Barrier in Hot Adatom Motion during Dissociative Adsorption of Oxygen on Ag(100). J. Chem. Phys. 2009, 131 (17), 174709 DOI: 10.1063/1.3258849Google ScholarThere is no corresponding record for this reference.
- 27Schintke, S.; Messerli, S.; Morgenstern, K.; Nieminen, J.; Schneider, W.-D. Far-Ranged Transient Motion of “Hot” Oxygen Atoms upon Dissociation. J. Chem. Phys. 2001, 114 (9), 4206– 4209, DOI: 10.1063/1.1346687Google ScholarThere is no corresponding record for this reference.
- 28Sprodowski, C.; Mehlhorn, M.; Morgenstern, K. Dissociation of Oxygen on Ag(100) Induced by Inelastic Electron Tunneling. J. Phys.: Condens. Matter 2010, 22 (26), 264005 DOI: 10.1088/0953-8984/22/26/264005Google ScholarThere is no corresponding record for this reference.
- 29Messerli, S.; Schintke, S.; Morgenstern, K.; Nieminen, J.; Schneider, W.-D. Oxygen Molecules on Ag(001): Superstructure, Binding Site and Molecular Orientation. Chem. Phys. Lett. 2000, 328 (4–6), 330– 336 DOI: 10.1016/S0009-2614(00)00951-9 .Google ScholarThere is no corresponding record for this reference.
- 30Garfunkel, E. L.; Ding, X.; Dong, G.; Yang, S.; Hou, X.; Wang, X. The Coadsorption of Sodium and Oxygen on Ag(100): An XPS, UPS and HREELS Study. Surf. Sci. 1985, 164 (2–3), 511– 525 DOI: 10.1016/0039-6028(85)90763-0 .Google ScholarThere is no corresponding record for this reference.
- 31Engelhardt, H. A.; Menzel, D. Adsorption of Oxygen on Silver Single Crystal Surfaces. Surf. Sci. 1976, 57 (2), 591– 618, DOI: 10.1016/0039-6028(76)90350-2Google ScholarThere is no corresponding record for this reference.
- 32De Mongeot, F. B.; Cupolillo, A.; Valbusa, U.; Rocca, M. O2 Dissociation on Ag(001): The Role of Kink Sites. Chem. Phys. Lett. 1997, 270 (3–4), 345– 350 DOI: 10.1016/S0009-2614(97)00381-3 .Google ScholarThere is no corresponding record for this reference.
- 33Vattuone, L.; Burghaus, U.; Savio, L.; Rocca, M.; Costantini, G.; Buatier De Mongeot, F.; Boragno, C.; Rusponi, S.; Valbusa, U. Oxygen Interaction with Disordered and Nanostructured Ag(001) Surfaces. J. Chem. Phys. 2001, 115 (7), 3346– 3355, DOI: 10.1063/1.1386432Google ScholarThere is no corresponding record for this reference.
- 34Ercelik, M.; Solé, A. P.; Zhang, L.; Kot, P.; Kim, J.; Chae, J.; Spree, L. E.; Guo, H.; Heinrich, A. J.; Bae, Y.; Borodin, D. Characterization of an Unexpected μ3 Adsorption of Molecular Oxygen on Ag(100) with Low-Temperature STM. J. Phys. Chem. C 2025, 129 (2), 1110– 1119, DOI: 10.1021/acs.jpcc.4c06572Google ScholarThere is no corresponding record for this reference.
- 35Christopher, P.; Linic, S. Engineering Selectivity in Heterogeneous Catalysis: Ag Nanowires as Selective Ethylene Epoxidation Catalysts. J. Am. Chem. Soc. 2008, 130 (34), 11264– 11265, DOI: 10.1021/ja803818kGoogle ScholarThere is no corresponding record for this reference.
- 36Huš, M.; Hellman, A. Ethylene Epoxidation on Ag(100), Ag(110), and Ag(111): A Joint Ab Initio and Kinetic Monte Carlo Study and Comparison with Experiments. ACS Catal. 2019, 9 (2), 1183– 1196, DOI: 10.1021/acscatal.8b04512Google ScholarThere is no corresponding record for this reference.
- 37Chen, D.; Chen, L.; Zhao, Q.-C.; Yang, Z.-X.; Shang, C.; Liu, Z.-P. Square-Pyramidal Subsurface Oxygen [Ag4OAg] Drives Selective Ethene Epoxidation on Silver. Nat. Catal. 2024, 7 (5), 536– 545, DOI: 10.1038/s41929-024-01135-2Google ScholarThere is no corresponding record for this reference.
- 38Chen, D.; Kang, P.-L.; Liu, Z.-P. Active Site of Catalytic Ethene Epoxidation: Machine-Learning Global Pathway Sampling Rules Out the Metal Sites. ACS Catal. 2021, 11 (13), 8317– 8326, DOI: 10.1021/acscatal.1c02029Google ScholarThere is no corresponding record for this reference.
- 39Mongeot, F. B. de; Rocca, M.; Valbusa, U. Energy and Angle Dependence of the Initial Sticking Coefficient of O2 on Ag(001). Surf. Sci. 1996, 363 (1–3), 68– 72 DOI: 10.1016/0039-6028(96)00111-2 .Google ScholarThere is no corresponding record for this reference.
- 40Campbell, C. T. Atomic and Molecular Oxygen Adsorption on Ag(111). Surf. Sci. 1985, 157 (1), 43– 60, DOI: 10.1016/0039-6028(85)90634-XGoogle ScholarThere is no corresponding record for this reference.
- 41Bukhtiyarov, V. I.; Prosvirin, I. P.; Kvon, R. I. Study of Reactivity of Oxygen States Adsorbed at a Silver Surface towards C2H4 by XPS, TPD and TPR. Surf. Sci. 1994, 320 (1–2), L47– L50 DOI: 10.1016/0039-6028(94)00562-1 .Google ScholarThere is no corresponding record for this reference.
- 42Bukhtiyarov, V. I.; Kaichev, V. V.; Prosvirin, I. P. Oxygen Adsorption on Ag(111): X-Ray Photoelectron Spectroscopy (XPS), Angular Dependent x-Ray Photoelectron Spectroscopy (ADXPS) and Temperature-Programmed Desorption (TPD) Studies. J. Chem. Phys. 1999, 111 (5), 2169– 2175, DOI: 10.1063/1.479488Google ScholarThere is no corresponding record for this reference.
- 43Bukhtiyarov, V. I.; Hävecker, M.; Kaichev, V. V.; Knop-Gericke, A.; Mayer, R. W.; Schlögl, R. Atomic Oxygen Species on Silver: Photoelectron Spectroscopy and x-Ray Absorption Studies. Phys. Rev. B 2003, 67 (23), 235422 DOI: 10.1103/PhysRevB.67.235422Google ScholarThere is no corresponding record for this reference.
- 44Isegawa, K.; Ueda, K.; Hiwasa, S.; Amemiya, K.; Mase, K.; Kondoh, H. Formation of Carbonate on Ag(111) under Exposure to Ethylene and Oxygen Gases Evidenced by Near Ambient Pressure XPS and NEXAFS. Chem. Lett. 2019, 48 (2), 159– 162, DOI: 10.1246/cl.180891Google ScholarThere is no corresponding record for this reference.
- 45Campbell, C. T. An XPS Study of Molecularly Chemisorbed Oxygen on Ag(111). Surf. Sci. Lett. 1986, 173 (2–3), L641– L646 DOI: 10.1016/0167-2584(86)90808-X .Google ScholarThere is no corresponding record for this reference.
- 46Peuckert, M. On the Adsorption of Oxygen and Potassium Hydroxide on Silver. Surf. Sci. 1984, 146 (2–3), 329– 340 DOI: 10.1016/0039-6028(84)90434-5 .Google ScholarThere is no corresponding record for this reference.
- 47Lützenkirchen-Hecht, D.; Strehblow, H. The Anodic Oxidation of Silver in 1 M NaOH: Electrochemistry, Ex Situ XPS and in Situ X-ray Absorption Spectroscopy. Surf. Interface Anal. 2006, 38 (4), 686– 690, DOI: 10.1002/sia.2188Google ScholarThere is no corresponding record for this reference.
- 48Nagy, A. J.; Mestl, G.; Herein, D.; Weinberg, G.; Kitzelmann, E.; Schlögl, R. The Correlation of Subsurface Oxygen Diffusion with Variations of Silver Morphology in the Silver-Oxygen System. J. Catal. 1999, 182 (2), 417– 429, DOI: 10.1006/jcat.1998.2388Google ScholarThere is no corresponding record for this reference.
- 49Savio, L.; Vattuone, L.; Rocca, M. From Adsorption at the Surface to Incorporation into Subsurface Sites: The Role of Steps for O/Ag. Appl. Phys. A: Mater. Sci. Process. 2007, 87 (3), 399– 404, DOI: 10.1007/s00339-007-3916-xGoogle ScholarThere is no corresponding record for this reference.
- 50Bao, X.; Muhler, M.; Schedel-Niedrig, T.; Schlögl, R. Interaction of Oxygen with Silver at High Temperature and Atmospheric Pressure: A Spectroscopic and Structural Analysis of a Strongly Bound Surface Species. Phys. Rev. B 1996, 54 (3), 2249– 2262, DOI: 10.1103/PhysRevB.54.2249Google ScholarThere is no corresponding record for this reference.
- 51Costina, I.; Schmid, M.; Schiechl, H.; Gajdoš, M.; Stierle, A.; Kumaragurubaran, S.; Hafner, J.; Dosch, H.; Varga, P. Combined STM, LEED and DFT Study of Ag(100) Exposed to Oxygen near Atmospheric Pressures. Surf. Sci. 2006, 600 (3), 617– 624, DOI: 10.1016/j.susc.2005.11.020Google ScholarThere is no corresponding record for this reference.
- 52Gajdoš, M.; Eichler, A.; Hafner, J. Ab Initio Density Functional Study of O on the Ag(001) Surface. Surf. Sci. 2003, 531 (3), 272– 286, DOI: 10.1016/S0039-6028(03)00514-4Google ScholarThere is no corresponding record for this reference.
- 53Cipriani, G.; Loffreda, D.; Dal Corso, A.; De Gironcoli, S.; Baroni, S. Adsorption of Atomic Oxygen on Ag(001): A Study Based on Density-Functional Theory. Surf. Sci. 2002, 501 (3), 182– 190, DOI: 10.1016/S0039-6028(01)01959-8Google ScholarThere is no corresponding record for this reference.
- 54Wang, Y.; Jia, L.; Wang, W.; Fan, K. O/Ag(100) Surface: A Density Functional Study with Slab Model. J. Phys. Chem. B 2002, 106 (14), 3662– 3667, DOI: 10.1021/jp012552pGoogle ScholarThere is no corresponding record for this reference.
- 55Hahn, J. R.; Ho, W. Chemisorption and Dissociation of Single Oxygen Molecules on Ag(110). J. Chem. Phys. 2005, 123 (21), 214702 DOI: 10.1063/1.2131064 .Google ScholarThere is no corresponding record for this reference.
- 56Spurgeon, P. M.; Liu, D.-J.; Walen, H.; Oh, J.; Yang, H. J.; Kim, Y.; Thiel, P. A. Characteristics of Sulfur Atoms Adsorbed on Ag(100), Ag(110), and Ag(111) as Probed with Scanning Tunneling Microscopy: Experiment and Theory. Phys. Chem. Chem. Phys. 2019, 21 (20), 10540– 10551, DOI: 10.1039/C9CP01626KGoogle ScholarThere is no corresponding record for this reference.
- 57Hoflund, G. B.; Hazos, Z. F.; Salaita, G. N. Surface Characterization Study of Ag, AgO, and Ag2O Using x-Ray Photoelectron Spectroscopy and Electron Energy-Loss Spectroscopy. Phys. Rev. B 2000, 62 (16), 11126– 11133, DOI: 10.1103/PhysRevB.62.11126Google ScholarThere is no corresponding record for this reference.
- 58Savinova, E. R.; Zemlyanov, D.; Pettinger, B.; Scheybal, A.; Schlögl, R.; Doblhofer, K. On the Mechanism of Ag(111) Sub-Monolayer Oxidation: A Combined Electrochemical, in Situ SERS and Ex Situ XPS Study. Electrochim. Acta 2000, 46 (2–3), 175– 183 DOI: 10.1016/S0013-4686(00)00571-5 .Google ScholarThere is no corresponding record for this reference.
- 59Zemlyanov, D. Y.; Savinova, E.; Scheybal, A.; Doblhofer, K.; Schlögl, R. XPS Observation of OH Groups Incorporated in an Ag(111) Electrode. Surf. Sci. 1998, 418 (2), 441– 456, DOI: 10.1016/S0039-6028(98)00728-6Google ScholarThere is no corresponding record for this reference.
- 60Stipe, B. C.; Rezaei, M. A.; Ho, W.; Gao, S.; Persson, M.; Lundqvist, B. I. Single-Molecule Dissociation by Tunneling Electrons. Phys. Rev. Lett. 1997, 78 (23), 4410– 4413, DOI: 10.1103/PhysRevLett.78.4410Google ScholarThere is no corresponding record for this reference.
- 61Fang, C. S. A. Surface Structural Transition of Adsorption of Oxygen on Ag(100). Surf. Sci. 1990, 235 (1), L291– L294, DOI: 10.1016/0039-6028(90)90098-SGoogle ScholarThere is no corresponding record for this reference.
- 62Lei, X.; Mbamalu, G.; Qin, C. CO Oxidation by Molecular and Atomic Oxygen on Ag(100): A Density Functional Theory Study. J. Phys. Chem. C 2017, 121 (5), 2635– 2642, DOI: 10.1021/acs.jpcc.6b09105Google ScholarThere is no corresponding record for this reference.
- 63Waluyo, I.; Hunt, A. Ambient Pressure X-Ray Photoelectron Spectroscopy at the IOS (23-ID-2) Beamline at the National Synchrotron Light Source II. Synchrotron Radiat. News 2022, 35 (3), 31– 38, DOI: 10.1080/08940886.2022.2082180Google ScholarThere is no corresponding record for this reference.
- 64Kresse, G.; Furthmüller, J. Efficiency of Ab-Initio Total Energy Calculations for Metals and Semiconductors Using a Plane-Wave Basis Set. Comput. Mater. Sci. 1996, 6 (1), 15– 50, DOI: 10.1016/0927-0256(96)00008-0Google ScholarThere is no corresponding record for this reference.
- 65Kresse, G.; Hafner, J. Ab Initio Molecular Dynamics for Liquid Metals. Phys. Rev. B 1993, 47 (1), 558– 561, DOI: 10.1103/PhysRevB.47.558Google ScholarThere is no corresponding record for this reference.
- 66Blöchl, P. E. Projector Augmented-Wave Method. Phys. Rev. B 1994, 50 (24), 17953– 17979, DOI: 10.1103/PhysRevB.50.17953Google ScholarThere is no corresponding record for this reference.
- 67Tkatchenko, A.; Scheffler, M. Accurate Molecular Van Der Waals Interactions from Ground-State Electron Density and Free-Atom Reference Data. Phys. Rev. Lett. 2009, 102 (7), 073005 DOI: 10.1103/PhysRevLett.102.073005Google ScholarThere is no corresponding record for this reference.
- 68Henkelman, G.; Jónsson, H. A Dimer Method for Finding Saddle Points on High Dimensional Potential Surfaces Using Only First Derivatives. J. Chem. Phys. 1999, 111 (15), 7010– 7022, DOI: 10.1063/1.480097Google ScholarThere is no corresponding record for this reference.
- 69Heyden, A.; Bell, A. T.; Keil, F. J. Efficient Methods for Finding Transition States in Chemical Reactions: Comparison of Improved Dimer Method and Partitioned Rational Function Optimization Method. J. Chem. Phys. 2005, 123 (22), 224101, DOI: 10.1063/1.2104507Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. Products resulting from O2 dissociation on Ag(100). (A) 78 K STM image of ∼ 1% ML depressions (red arrow) and ∼ 1.5% ML protrusions (blue arrow) resulting from a 1000 L dose of O2 on Ag(100) at room temperature. Imaging conditions: 10 mV, 1 nA. (B) O 1s XPS spectra of clean Ag(100), 12,000 L O2 300 K (7% O530 and 4% O531) on Ag(100), and subsequent anneal. (C) Plot of the coverage of each O 1s component from the fits in A.
Figure 2
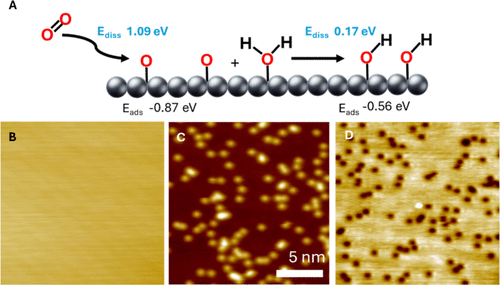
Figure 2. 78 K STM images of the Ag(100) surface before and after exposure to O2 under different imaging conditions. (A) Depiction of oxygen dissociation and reaction with water on the Ag(100) surface and associated reaction energies and activation barriers. Dissociation barriers are shown in blue (Ediss), and adsorption energies are shown beneath in black (Eads). All energy values are relative to gas phase O2. (B) 78 K STM image of the bare Ag(100) surface before exposure to O2. Imaging conditions: 300 mV, 300 pA. (C)-(D) Images of O adatoms (depressions) and OH groups (protrusions) on Ag(100) imaged at (C) 3.00 V, 300 pA, (D) 300 mV, 300 pA. (C) and (D) are of the same area. At 3.00 V, only protrusions are visible, and at 300 mV only depressions are visible. Surface coverages were 1.0% O and 1.5% OH in (C) and (D). All images are 50 × 50 nm2, scale bar shown in C.
Figure 3

Figure 3. Binding site characterization of O adatoms and OH groups on O–Ag(100). (A) Atomic resolution 78 K STM image of an oxygen adatom on Ag(100) imaged at 10 mV, 1 nA. (B) The same area of the surface under typical resolution imaged at 100 mV, 1 nA. Both STM images are the same size, and a scale bar is shown in (A). (C) STM image of O and OH on Ag(100) imaged at 25 mV, 1 nA, and a scale bar is shown. Insets in panel C are (top) Simulated STM image of an OH group on Ag(100), (bottom) simulated STM of an O adatom on Ag(100). (D) and (E) are the calculated preferred binding sites for (D) OH and (E) O atoms. (F) STM image of O and OH with the Ag(100) 4-fold hollow lattice corrected for drift overlaid. The centers of several OH groups are marked with blue circles or arrows. The locations where the grid lines cross are the 4-fold hollow sites. Imaging conditions: 25 mV, 1 nA, and a scale bar is shown. Images were acquired at 78 K.
Figure 4

Figure 4. STM Voltage Pulse Experiments. (A) Depiction of STM tip dissociating OH into O and H using a localized voltage pulse. (B) 78 K STM image of the Ag(100) surface before voltage pulses. OH groups appear as faint protrusions and are marked by blue arrows. (C) 78 K STM image of the same area after 4 V pulses. Each OH is now a depression in the same surface location. Oxygen was dosed at 300 K, and the pulse locations are indicated by the blue arrows. (D) Depiction of the STM tip dissociating O2 into two O adatoms via a voltage pulse. (E) LT-STM image of the Ag(100) surface with oxygen dosed at 78 K. Oxygen is present as molecular oxygen and diffuses too fast at 78 K to be imaged. The pulse location is indicated by the blue arrow. (F) LT-STM image of the same area following one 8 V pulse. Oxygen adatoms have formed as a result of the pulse. No further changes occurred after pulsing on the O adatoms. Imaging conditions: 1.0 nA and 50 mV.
Figure 5
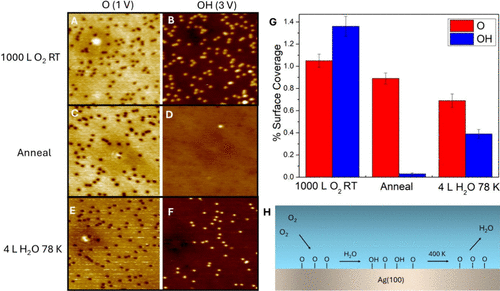
Figure 5. Reaction of O atoms with adsorbed H2O. (A) 78 K STM image of 1000 L O2 dosed at 300 K on Ag(100) imaged at 1 V. O adatoms are visible as depressions. (B) Same area imaged at 3 V where OH groups are visible as protrusions. (C) STM image of O–Ag(100) surface after a 400 K anneal scanned at 1 V. (D) Same area scanned at 3 V showing a significant reduction in the number of OH species. (E) STM image of the surface after 3 L H2O was dosed at 78 K, annealed to 400 K, and then imaged at 1 V at 78 K. (F) Same area imaged at 3.0 V showing an increase in the number of OH species. (G) Quantification of the surface coverages of O and OH for each stage of the experiment. (H) Schematic summarizing O2 dissociation, OH formation, and water desorption from Ag(100). Tunneling current was constant for all STM images at 300 pA. All images are 50 × 50 nm2.
References
This article references 69 other publications.
- 1Vodyankina, O. V. Silver Catalysts for the Partial Oxidation of Alcohols. Catal. Ind. 2022, 14 (3), 314– 326, DOI: 10.1134/S2070050422030059There is no corresponding record for this reference.
- 2Torbina, V. V.; Vodyankin, A. A.; Ten, S.; Mamontov, G. V.; Salaev, M. A.; Sobolev, V. I.; Vodyankina, O. V. Ag-Based Catalysts in Heterogeneous Selective Oxidation of Alcohols: A Review. Catalysts 2018, 8 (10), 447 DOI: 10.3390/catal8100447There is no corresponding record for this reference.
- 3Lockemeyer, J. R.; Lohr, T. L. Ethylene Oxide Catalysis Under Commercial Conditions – A Guide for Researchers. ChemCatChem 2023, 15 (13), e202201511 DOI: 10.1002/cctc.202201511There is no corresponding record for this reference.
- 4Salmi, T.; Roche, M.; Hernández Carucci, J.; Eränen, K.; Murzin, D. Ethylene Oxide – Kinetics and Mechanism. Curr. Opin. Chem. Eng. 2012, 1 (3), 321– 327, DOI: 10.1016/j.coche.2012.06.002There is no corresponding record for this reference.
- 5Montemore, M. M.; Spronsen, M. A. van; Madix, R. J.; Friend, C. M. O2 Activation by Metal Surfaces: Implications for Bonding and Reactivity on Heterogeneous Catalysts. Chem. Rev. 2018, 118 (5), 2816– 2862, DOI: 10.1021/acs.chemrev.7b00217There is no corresponding record for this reference.
- 6Pu, T.; Setiawan, A.; Rangarajan, S.; Wachs, I. E. Progress and Perspective on the Fundamental Understanding of Structure–Activity/Selectivity Relationships for Ag Catalyzed Ethylene Epoxidation. Catal. Today 2025, 455, 115301 DOI: 10.1016/j.cattod.2025.115301There is no corresponding record for this reference.
- 7Carbonio, E. A.; Rocha, T. C. R.; Klyushin, A. Yu.; Píš, I.; Magnano, E.; Nappini, S.; Piccinin, S.; Knop-Gericke, A.; Schlögl, R.; Jones, T. E. Are Multiple Oxygen Species Selective in Ethylene Epoxidation on Silver?. Chem. Sci. 2018, 9 (4), 990– 998, DOI: 10.1039/C7SC04728BThere is no corresponding record for this reference.
- 8Liu, J.-X.; Lu, S.; Ann, S.-B.; Linic, S. Mechanisms of Ethylene Epoxidation over Silver from Machine Learning-Accelerated First-Principles Modeling and Microkinetic Simulations. ACS Catal. 2023, 13 (13), 8955– 8962, DOI: 10.1021/acscatal.3c00410There is no corresponding record for this reference.
- 9Rocha, T. C. R.; Hävecker, M.; Knop-Gericke, A.; Schlögl, R. Promoters in Heterogeneous Catalysis: The Role of Cl on Ethylene Epoxidation over Ag. J. Catal. 2014, 312, 12– 16, DOI: 10.1016/j.jcat.2014.01.002There is no corresponding record for this reference.
- 10Henderson, M. The Interaction of Water with Solid Surfaces: Fundamental Aspects Revisited. Surf. Sci. Rep. 2002, 46 (1–8), 1– 308, DOI: 10.1016/S0167-5729(01)00020-6There is no corresponding record for this reference.
- 11Bao, X.; Muhler, M.; Pettinger, B.; Uchida, Y.; Lehmpfuhl, G.; Schlögl, R.; Ertl, G. The Effect of Water on the Formation of Strongly Bound Oxygen on Silver Surfaces. Catal. Lett. 1995, 32 (1–2), 171– 183, DOI: 10.1007/BF00806112There is no corresponding record for this reference.
- 12Klaua, M.; Madey, T. E. The Adsorption of H2O on Clean and Oxygen-Dosed Silver Single Crystal Surfaces. Surf. Sci. 1984, 136 (1), L42– L50, DOI: 10.1016/0039-6028(84)90650-2There is no corresponding record for this reference.
- 13Guillemot, L.; Bobrov, K. On the Formation of OH Ordered Layers by Dissociation of H2O on an Oxygen Covered Ag(110) Surface: An STM Investigation. Surf. Sci. 2007, 601 (3), 871– 875, DOI: 10.1016/j.susc.2006.11.027There is no corresponding record for this reference.
- 14Chu, Y.; Liang, H.; Lin, Q.; Huang, H.; Che, P.; Han, Y. Effect of Water Vapor on the Structure and Performance of Silver Catalysts for Ethylene Oxidation. Chem. Eng. J. 2025, 518, 164442 DOI: 10.1016/j.cej.2025.164442There is no corresponding record for this reference.
- 15Liu, W.; Eliyas, A.; Petrov, L. Influence of Feed Water Vapour on the Selective Oxidation of Ethylene over Silver Catalyst. Appl. Catal. 1990, 61 (1), 265– 274, DOI: 10.1016/S0166-9834(00)82149-1There is no corresponding record for this reference.
- 16Bowker, M.; Barteau, M. A.; Madix, R. J. Oxygen Induced Adsorption and Reaction of H2, H2O, CO and CO2 on Single Crystal Ag(110). Surf. Sci. 1980, 92 (2–3), 528– 548, DOI: 10.1016/0039-6028(80)90221-6There is no corresponding record for this reference.
- 17Campbell, C. T.; Paffett, M. T. The Interactions of O2, CO and CO2 with Ag(110). Surf. Sci. 1984, 143 (2–3), 517– 535, DOI: 10.1016/0039-6028(84)90557-0There is no corresponding record for this reference.
- 18Codeço, C. F. S.; Klyushin, A. Y.; Carbonio, E. A.; Knop-Gericke, A.; Schlögl, R.; Jones, T.; Rocha, T. C. R. Insights into the Electronic Structure of Hydroxyl on Ag(110) under near Ambient Conditions. Phys. Chem. Chem. Phys. 2022, 24 (15), 8832– 8838, DOI: 10.1039/D1CP02929KThere is no corresponding record for this reference.
- 19Stuve, E. M.; Madix, R. J.; Sexton, B. A. The Adsorption and Reaction of H2O on Clean and Oxygen Covered Ag(110). Surf. Sci. 1981, 111 (1), 11– 25, DOI: 10.1016/0039-6028(81)90471-4There is no corresponding record for this reference.
- 20Savio, L.; Smerieri, M.; Vattuone, L.; Gussoni, A.; Tassistro, C.; Rocca, M. STM Study of Hydroxyl Formation at OAg(100). Phys. Rev. B 2006, 74 (23), 235412 DOI: 10.1103/PhysRevB.74.235412There is no corresponding record for this reference.
- 21Wachs, I. E.; Keleman, S. R. The Interaction of Ethylene with Surface Carbonate and Hydroxide Intermediates on Silver. J. Catal. 1981, 71 (1), 78– 87 DOI: 10.1016/0021-9517(81)90203-7 .There is no corresponding record for this reference.
- 22Bange, K.; Madey, T. E.; Sass, J. K.; Stuve, E. M. The Adsorption of Water and Oxygen on Ag(110): A Study of the Interactions among Water Molecules, Hydroxyl Groups, and Oxygen Atoms. Surf. Sci. 1987, 183 (3), 334– 362, DOI: 10.1016/S0039-6028(87)80215-7There is no corresponding record for this reference.
- 23Hu, Z.-M.; Nakatsuji, H. Adsorption and Disproportionation Reaction of OH on Ag Surfaces: Dipped Adcluster Model Study. Surf. Sci. 1999, 425 (2–3), 296– 312, DOI: 10.1016/S0039-6028(99)00215-0There is no corresponding record for this reference.
- 24Qin, C.; Whitten, J. L. Adsorption of O, H, OH, and H2O on Ag(100). J. Phys. Chem. B 2005, 109 (18), 8852– 8856, DOI: 10.1021/jp044067aThere is no corresponding record for this reference.
- 25Rocca, M.; Savio, L.; Vattuone, L.; Burghaus, U.; Palomba, V.; Novelli, N.; Mongeot, F. B. de; Valbusa, U.; Gunnella, R.; Comelli, G.; Baraldi, A.; Lizzit, S.; Paolucci, G. Phase Transition of Dissociatively Adsorbed Oxygen on Ag(001). Phys. Rev. B 2000, 61 (1), 213– 227, DOI: 10.1103/PhysRevB.61.213There is no corresponding record for this reference.
- 26Hsieh, M.-F.; Lin, D.-S.; Gawronski, H.; Morgenstern, K. Hard Repulsive Barrier in Hot Adatom Motion during Dissociative Adsorption of Oxygen on Ag(100). J. Chem. Phys. 2009, 131 (17), 174709 DOI: 10.1063/1.3258849There is no corresponding record for this reference.
- 27Schintke, S.; Messerli, S.; Morgenstern, K.; Nieminen, J.; Schneider, W.-D. Far-Ranged Transient Motion of “Hot” Oxygen Atoms upon Dissociation. J. Chem. Phys. 2001, 114 (9), 4206– 4209, DOI: 10.1063/1.1346687There is no corresponding record for this reference.
- 28Sprodowski, C.; Mehlhorn, M.; Morgenstern, K. Dissociation of Oxygen on Ag(100) Induced by Inelastic Electron Tunneling. J. Phys.: Condens. Matter 2010, 22 (26), 264005 DOI: 10.1088/0953-8984/22/26/264005There is no corresponding record for this reference.
- 29Messerli, S.; Schintke, S.; Morgenstern, K.; Nieminen, J.; Schneider, W.-D. Oxygen Molecules on Ag(001): Superstructure, Binding Site and Molecular Orientation. Chem. Phys. Lett. 2000, 328 (4–6), 330– 336 DOI: 10.1016/S0009-2614(00)00951-9 .There is no corresponding record for this reference.
- 30Garfunkel, E. L.; Ding, X.; Dong, G.; Yang, S.; Hou, X.; Wang, X. The Coadsorption of Sodium and Oxygen on Ag(100): An XPS, UPS and HREELS Study. Surf. Sci. 1985, 164 (2–3), 511– 525 DOI: 10.1016/0039-6028(85)90763-0 .There is no corresponding record for this reference.
- 31Engelhardt, H. A.; Menzel, D. Adsorption of Oxygen on Silver Single Crystal Surfaces. Surf. Sci. 1976, 57 (2), 591– 618, DOI: 10.1016/0039-6028(76)90350-2There is no corresponding record for this reference.
- 32De Mongeot, F. B.; Cupolillo, A.; Valbusa, U.; Rocca, M. O2 Dissociation on Ag(001): The Role of Kink Sites. Chem. Phys. Lett. 1997, 270 (3–4), 345– 350 DOI: 10.1016/S0009-2614(97)00381-3 .There is no corresponding record for this reference.
- 33Vattuone, L.; Burghaus, U.; Savio, L.; Rocca, M.; Costantini, G.; Buatier De Mongeot, F.; Boragno, C.; Rusponi, S.; Valbusa, U. Oxygen Interaction with Disordered and Nanostructured Ag(001) Surfaces. J. Chem. Phys. 2001, 115 (7), 3346– 3355, DOI: 10.1063/1.1386432There is no corresponding record for this reference.
- 34Ercelik, M.; Solé, A. P.; Zhang, L.; Kot, P.; Kim, J.; Chae, J.; Spree, L. E.; Guo, H.; Heinrich, A. J.; Bae, Y.; Borodin, D. Characterization of an Unexpected μ3 Adsorption of Molecular Oxygen on Ag(100) with Low-Temperature STM. J. Phys. Chem. C 2025, 129 (2), 1110– 1119, DOI: 10.1021/acs.jpcc.4c06572There is no corresponding record for this reference.
- 35Christopher, P.; Linic, S. Engineering Selectivity in Heterogeneous Catalysis: Ag Nanowires as Selective Ethylene Epoxidation Catalysts. J. Am. Chem. Soc. 2008, 130 (34), 11264– 11265, DOI: 10.1021/ja803818kThere is no corresponding record for this reference.
- 36Huš, M.; Hellman, A. Ethylene Epoxidation on Ag(100), Ag(110), and Ag(111): A Joint Ab Initio and Kinetic Monte Carlo Study and Comparison with Experiments. ACS Catal. 2019, 9 (2), 1183– 1196, DOI: 10.1021/acscatal.8b04512There is no corresponding record for this reference.
- 37Chen, D.; Chen, L.; Zhao, Q.-C.; Yang, Z.-X.; Shang, C.; Liu, Z.-P. Square-Pyramidal Subsurface Oxygen [Ag4OAg] Drives Selective Ethene Epoxidation on Silver. Nat. Catal. 2024, 7 (5), 536– 545, DOI: 10.1038/s41929-024-01135-2There is no corresponding record for this reference.
- 38Chen, D.; Kang, P.-L.; Liu, Z.-P. Active Site of Catalytic Ethene Epoxidation: Machine-Learning Global Pathway Sampling Rules Out the Metal Sites. ACS Catal. 2021, 11 (13), 8317– 8326, DOI: 10.1021/acscatal.1c02029There is no corresponding record for this reference.
- 39Mongeot, F. B. de; Rocca, M.; Valbusa, U. Energy and Angle Dependence of the Initial Sticking Coefficient of O2 on Ag(001). Surf. Sci. 1996, 363 (1–3), 68– 72 DOI: 10.1016/0039-6028(96)00111-2 .There is no corresponding record for this reference.
- 40Campbell, C. T. Atomic and Molecular Oxygen Adsorption on Ag(111). Surf. Sci. 1985, 157 (1), 43– 60, DOI: 10.1016/0039-6028(85)90634-XThere is no corresponding record for this reference.
- 41Bukhtiyarov, V. I.; Prosvirin, I. P.; Kvon, R. I. Study of Reactivity of Oxygen States Adsorbed at a Silver Surface towards C2H4 by XPS, TPD and TPR. Surf. Sci. 1994, 320 (1–2), L47– L50 DOI: 10.1016/0039-6028(94)00562-1 .There is no corresponding record for this reference.
- 42Bukhtiyarov, V. I.; Kaichev, V. V.; Prosvirin, I. P. Oxygen Adsorption on Ag(111): X-Ray Photoelectron Spectroscopy (XPS), Angular Dependent x-Ray Photoelectron Spectroscopy (ADXPS) and Temperature-Programmed Desorption (TPD) Studies. J. Chem. Phys. 1999, 111 (5), 2169– 2175, DOI: 10.1063/1.479488There is no corresponding record for this reference.
- 43Bukhtiyarov, V. I.; Hävecker, M.; Kaichev, V. V.; Knop-Gericke, A.; Mayer, R. W.; Schlögl, R. Atomic Oxygen Species on Silver: Photoelectron Spectroscopy and x-Ray Absorption Studies. Phys. Rev. B 2003, 67 (23), 235422 DOI: 10.1103/PhysRevB.67.235422There is no corresponding record for this reference.
- 44Isegawa, K.; Ueda, K.; Hiwasa, S.; Amemiya, K.; Mase, K.; Kondoh, H. Formation of Carbonate on Ag(111) under Exposure to Ethylene and Oxygen Gases Evidenced by Near Ambient Pressure XPS and NEXAFS. Chem. Lett. 2019, 48 (2), 159– 162, DOI: 10.1246/cl.180891There is no corresponding record for this reference.
- 45Campbell, C. T. An XPS Study of Molecularly Chemisorbed Oxygen on Ag(111). Surf. Sci. Lett. 1986, 173 (2–3), L641– L646 DOI: 10.1016/0167-2584(86)90808-X .There is no corresponding record for this reference.
- 46Peuckert, M. On the Adsorption of Oxygen and Potassium Hydroxide on Silver. Surf. Sci. 1984, 146 (2–3), 329– 340 DOI: 10.1016/0039-6028(84)90434-5 .There is no corresponding record for this reference.
- 47Lützenkirchen-Hecht, D.; Strehblow, H. The Anodic Oxidation of Silver in 1 M NaOH: Electrochemistry, Ex Situ XPS and in Situ X-ray Absorption Spectroscopy. Surf. Interface Anal. 2006, 38 (4), 686– 690, DOI: 10.1002/sia.2188There is no corresponding record for this reference.
- 48Nagy, A. J.; Mestl, G.; Herein, D.; Weinberg, G.; Kitzelmann, E.; Schlögl, R. The Correlation of Subsurface Oxygen Diffusion with Variations of Silver Morphology in the Silver-Oxygen System. J. Catal. 1999, 182 (2), 417– 429, DOI: 10.1006/jcat.1998.2388There is no corresponding record for this reference.
- 49Savio, L.; Vattuone, L.; Rocca, M. From Adsorption at the Surface to Incorporation into Subsurface Sites: The Role of Steps for O/Ag. Appl. Phys. A: Mater. Sci. Process. 2007, 87 (3), 399– 404, DOI: 10.1007/s00339-007-3916-xThere is no corresponding record for this reference.
- 50Bao, X.; Muhler, M.; Schedel-Niedrig, T.; Schlögl, R. Interaction of Oxygen with Silver at High Temperature and Atmospheric Pressure: A Spectroscopic and Structural Analysis of a Strongly Bound Surface Species. Phys. Rev. B 1996, 54 (3), 2249– 2262, DOI: 10.1103/PhysRevB.54.2249There is no corresponding record for this reference.
- 51Costina, I.; Schmid, M.; Schiechl, H.; Gajdoš, M.; Stierle, A.; Kumaragurubaran, S.; Hafner, J.; Dosch, H.; Varga, P. Combined STM, LEED and DFT Study of Ag(100) Exposed to Oxygen near Atmospheric Pressures. Surf. Sci. 2006, 600 (3), 617– 624, DOI: 10.1016/j.susc.2005.11.020There is no corresponding record for this reference.
- 52Gajdoš, M.; Eichler, A.; Hafner, J. Ab Initio Density Functional Study of O on the Ag(001) Surface. Surf. Sci. 2003, 531 (3), 272– 286, DOI: 10.1016/S0039-6028(03)00514-4There is no corresponding record for this reference.
- 53Cipriani, G.; Loffreda, D.; Dal Corso, A.; De Gironcoli, S.; Baroni, S. Adsorption of Atomic Oxygen on Ag(001): A Study Based on Density-Functional Theory. Surf. Sci. 2002, 501 (3), 182– 190, DOI: 10.1016/S0039-6028(01)01959-8There is no corresponding record for this reference.
- 54Wang, Y.; Jia, L.; Wang, W.; Fan, K. O/Ag(100) Surface: A Density Functional Study with Slab Model. J. Phys. Chem. B 2002, 106 (14), 3662– 3667, DOI: 10.1021/jp012552pThere is no corresponding record for this reference.
- 55Hahn, J. R.; Ho, W. Chemisorption and Dissociation of Single Oxygen Molecules on Ag(110). J. Chem. Phys. 2005, 123 (21), 214702 DOI: 10.1063/1.2131064 .There is no corresponding record for this reference.
- 56Spurgeon, P. M.; Liu, D.-J.; Walen, H.; Oh, J.; Yang, H. J.; Kim, Y.; Thiel, P. A. Characteristics of Sulfur Atoms Adsorbed on Ag(100), Ag(110), and Ag(111) as Probed with Scanning Tunneling Microscopy: Experiment and Theory. Phys. Chem. Chem. Phys. 2019, 21 (20), 10540– 10551, DOI: 10.1039/C9CP01626KThere is no corresponding record for this reference.
- 57Hoflund, G. B.; Hazos, Z. F.; Salaita, G. N. Surface Characterization Study of Ag, AgO, and Ag2O Using x-Ray Photoelectron Spectroscopy and Electron Energy-Loss Spectroscopy. Phys. Rev. B 2000, 62 (16), 11126– 11133, DOI: 10.1103/PhysRevB.62.11126There is no corresponding record for this reference.
- 58Savinova, E. R.; Zemlyanov, D.; Pettinger, B.; Scheybal, A.; Schlögl, R.; Doblhofer, K. On the Mechanism of Ag(111) Sub-Monolayer Oxidation: A Combined Electrochemical, in Situ SERS and Ex Situ XPS Study. Electrochim. Acta 2000, 46 (2–3), 175– 183 DOI: 10.1016/S0013-4686(00)00571-5 .There is no corresponding record for this reference.
- 59Zemlyanov, D. Y.; Savinova, E.; Scheybal, A.; Doblhofer, K.; Schlögl, R. XPS Observation of OH Groups Incorporated in an Ag(111) Electrode. Surf. Sci. 1998, 418 (2), 441– 456, DOI: 10.1016/S0039-6028(98)00728-6There is no corresponding record for this reference.
- 60Stipe, B. C.; Rezaei, M. A.; Ho, W.; Gao, S.; Persson, M.; Lundqvist, B. I. Single-Molecule Dissociation by Tunneling Electrons. Phys. Rev. Lett. 1997, 78 (23), 4410– 4413, DOI: 10.1103/PhysRevLett.78.4410There is no corresponding record for this reference.
- 61Fang, C. S. A. Surface Structural Transition of Adsorption of Oxygen on Ag(100). Surf. Sci. 1990, 235 (1), L291– L294, DOI: 10.1016/0039-6028(90)90098-SThere is no corresponding record for this reference.
- 62Lei, X.; Mbamalu, G.; Qin, C. CO Oxidation by Molecular and Atomic Oxygen on Ag(100): A Density Functional Theory Study. J. Phys. Chem. C 2017, 121 (5), 2635– 2642, DOI: 10.1021/acs.jpcc.6b09105There is no corresponding record for this reference.
- 63Waluyo, I.; Hunt, A. Ambient Pressure X-Ray Photoelectron Spectroscopy at the IOS (23-ID-2) Beamline at the National Synchrotron Light Source II. Synchrotron Radiat. News 2022, 35 (3), 31– 38, DOI: 10.1080/08940886.2022.2082180There is no corresponding record for this reference.
- 64Kresse, G.; Furthmüller, J. Efficiency of Ab-Initio Total Energy Calculations for Metals and Semiconductors Using a Plane-Wave Basis Set. Comput. Mater. Sci. 1996, 6 (1), 15– 50, DOI: 10.1016/0927-0256(96)00008-0There is no corresponding record for this reference.
- 65Kresse, G.; Hafner, J. Ab Initio Molecular Dynamics for Liquid Metals. Phys. Rev. B 1993, 47 (1), 558– 561, DOI: 10.1103/PhysRevB.47.558There is no corresponding record for this reference.
- 66Blöchl, P. E. Projector Augmented-Wave Method. Phys. Rev. B 1994, 50 (24), 17953– 17979, DOI: 10.1103/PhysRevB.50.17953There is no corresponding record for this reference.
- 67Tkatchenko, A.; Scheffler, M. Accurate Molecular Van Der Waals Interactions from Ground-State Electron Density and Free-Atom Reference Data. Phys. Rev. Lett. 2009, 102 (7), 073005 DOI: 10.1103/PhysRevLett.102.073005There is no corresponding record for this reference.
- 68Henkelman, G.; Jónsson, H. A Dimer Method for Finding Saddle Points on High Dimensional Potential Surfaces Using Only First Derivatives. J. Chem. Phys. 1999, 111 (15), 7010– 7022, DOI: 10.1063/1.480097There is no corresponding record for this reference.
- 69Heyden, A.; Bell, A. T.; Keil, F. J. Efficient Methods for Finding Transition States in Chemical Reactions: Comparison of Improved Dimer Method and Partitioned Rational Function Optimization Method. J. Chem. Phys. 2005, 123 (22), 224101, DOI: 10.1063/1.2104507There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jpclett.5c03296.
Details of experimental methods and additional STM, DFT and XPS data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



