Self-Propulsion of a Soap at an Oil/Aqueous Interface Depending on pHClick to copy article linkArticle link copied!
- Takahito AraiTakahito AraiGraduate School of Integrated Sciences for Life, Hiroshima University, 1-3-1 Kagamiyama, Higashihiroshima, Hiroshima739-8526, JapanMore by Takahito Arai
- Masakazu KuzeMasakazu KuzeGraduate School of Integrated Sciences for Life, Hiroshima University, 1-3-1 Kagamiyama, Higashihiroshima, Hiroshima739-8526, JapanMeiji Institute for Advanced Study of Mathematical Sciences (MIMS), Meiji University, 4-21-1 Nakano, Nakano-ku, Tokyo164-8525, JapanMore by Masakazu Kuze
- Muneyuki Matsuo*Muneyuki Matsuo*Email: [email protected]Graduate School of Integrated Sciences for Life, Hiroshima University, 1-3-1 Kagamiyama, Higashihiroshima, Hiroshima739-8526, JapanGraduate School of Arts and Sciences, The University of Tokyo, 3-8-1 Komaba, Meguro, Tokyo153-8902, JapanMore by Muneyuki Matsuo
- Satoshi Nakata*Satoshi Nakata*Email: [email protected]Graduate School of Integrated Sciences for Life, Hiroshima University, 1-3-1 Kagamiyama, Higashihiroshima, Hiroshima739-8526, JapanMore by Satoshi Nakata
Abstract
A self-propelled sodium oleate (OleNa) disk was investigated at an oil/aqueous interface prepared in an annular channel to induce characteristic features of self-propulsion. When the pH of the aqueous phase was changed, three types of motion were observed, i.e., unidirectional motion at 3.0 ≤ pH ≤ 6.0, motion with inversion at 8.0 ≤ pH ≤ 9.0, and no motion at 11.0 ≤ pH ≤ 12.0. At pH = 8.0, the interfacial tension and complementary contact angle of the meniscus oscillated simultaneously. The mechanism of the three types of motion is discussed in relation to the acidity constant (pKa) between protonated and deprotonated oleic acids, their distribution ratios in the oil and aqueous phases, and the driving force of motion. The present study suggests that the mode of self-propulsion is determined by the nature of the energy-source molecule, specifically its pKa, interfacial tension, and adsorption/desorption of protonated and deprotonated oleic acid molecules at the interface.
This publication is licensed for personal use by The American Chemical Society.
Note
Made available for a limited time for personal research and study only License.
Introduction
Experimental Section
Figure 1
Figure 1. Schematic illustration of (a) the experimental apparatus for observing a self-propelled OleNa disk placed at an oil/aqueous interface, (b) the measurement of the interfacial tension, and (c) the simultaneous measurement of the interfacial tension and movie around the OleNa disk fixed at the oil/aqueous interface. In (a), θ and θ = 0 were defined to analyze the motion in the polar coordinates.
Results and Discussion
Mode Switching of an OleNa Disk at the Oil/Aqueous Interface Depending on pH
Figure 2
Figure 2. Time variations of θ for self-propulsion of an OleNa disk at pH = (a) 6.0, (b) 8.0, and (c) 12.0. θ and θ = 0 are defined in Figure 1. The movies of motion in parts (a) and (b) are provided in the Supporting Information as Movies S1 and S2, respectively.
Figure 3
Figure 3. (a) Phase diagram of self-propelled motion, (b) absolute value of angular speed, |ω|, and (c) ratio of unidirectional motion per 1 min, R, depending on pH of the aqueous phase. Error bars represent the standard deviations obtained from three or four examinations. In the boundary regions at pH 6.9 and 9.8 in (a), both unidirectional motion and motion with inversion were observed, and both motion with inversion and no motion were observed, respectively.
Measurement of Interfacial Tension as the Driving Force of Motion
Figure 4
Figure 4. Time variation of the oil/aqueous interfacial tension at pH 6.0 (red line), 8.0 (blue line), and 12.0 (green line). The downward and upward arrows denote the time when the OleNa disk is placed in contact with the interface from above (t = 10 s) and when it is moved upward away from the interface to the air phase (t = 30 s), respectively.
Figure 5
Figure 5. Difference in the interfacial tension, Δγ (= γ0 – γs), at the equilibrium state, depending on the pH of the aqueous phase, where γs and γ0 are the interfacial tension at the oil/aqueous interface with and without OleNa in the aqueous and oil phase, respectively. Error bars represent the standard deviation obtained from three examinations.
Figure 6
Figure 6. (a) Snapshots of the OleNa disk fixed on the interface at pH 8.0 at t = (i) 680, (ii) 700, and (iii) 760 s (slanted view). The movie at pH 8.0 is provided in the Supporting Information as Movie S3 (500 ≤ t ≤ 900 s). (b) Simultaneous measurement of the time variation of the sum of γs/w, γo/s, and γo/w in the vertical direction (γv) and the complementary contact angle (θc). The data were smoothed by a seven-point moving average (corresponding to ±3 frames, total ∼1.2 s) to reduce noise. The original time resolution was 0.167 s per frame. The vertical position of the disk was adjusted so that the upper surface of the disk almost coincided with the oil/aqueous interface. We confirmed the reproducibility of the oscillatory phenomena.
Estimation of the Total Concentration of Oleic Acid in the Oil and Aqueous Phases
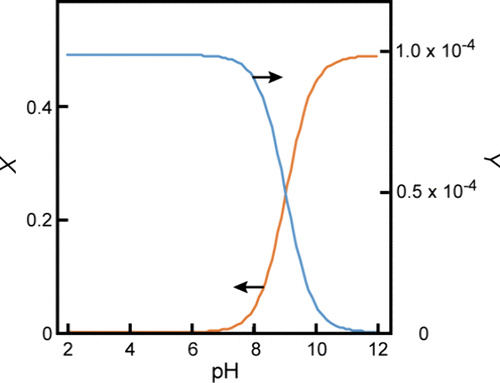
Figure 7
Figure 7. Total concentration of oleic acid in the oil (filled circles) and aqueous (empty circles) phases estimated at different pH values.
Numerical Calculation of Oleic Acid Concentrations
Figure 8
Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.langmuir.5c04037.
Calibration curve of oleic acid concentration, interfacial tension over time at pH 6.0, steady-state interfacial tension vs pH, spatiotemporal plot on the meniscus around the OleNa disk, pH dependence of deprotonated and protonated oleic acid concentrations, and adsorption behavior of deprotonated and protonated oleic acids at the interface (PDF)
Self-propulsion of the OleNa disk at pH 6.0 (real time) (AVI)
Self-propulsion of the OleNa disk at pH 8.0 (real time) (AVI)
Meniscus around the OleNa disk pH 8.0 (5× speed) (AVI)
Meniscus around the OleNa disk at pH 6.0 (5× speed) (AVI)
Meniscus around the OleNa disk at pH 12.0 (5× speed) (AVI)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This study was supported by JSPS KAKENHI (grant nos. JP20H02712, 24KJ1731, 21H00996, and 25K18011), Iketani Science and Technology Foundation (0351181-A), Cooperative Research Program of “Network Joint Research Center for Materials and Devices” (No. 20251018), Research Fund for Young Scientists; Grant-in-aid of Graduate School of Integrated Sciences for Life, the JSPS Bilateral Joint Research Project between Japan and the Polish Academy of Sciences (JPJSBP120204602), JSPS-Hungary Bilateral Joint Research Project (JPJSBP120213801), ExCELLS, National Institute of Sciences, Project Research (25K18011), JST ACT-X (JP24031207), and MEXT Leading Initiative for Excellent Young Researchers (JPMXS0320230007). We would like to thank Editage (www.editage.jp) for English language editing.
References
This article references 52 other publications.
- 1Nakata, S.; Pimienta, V.; Lagzi, I.; Kitahata, H.; Suematsu, N. J. Self-Organized Motion: Physicochemical Design Based on Nonlinear Dynamics; The Royal Society of Cambridge: 2019. DOI: 10.1039/9781788013499-FGoogle ScholarThere is no corresponding record for this reference.
- 2Soto, F.; Karshalev, E.; Zhang, F.; Esteban Fernandez de Avila, B.; Nourhani, A.; Wang, J. Smart Materials for Microrobots. Chem. Rev. 2022, 122, 3562– 3613, DOI: 10.1021/acs.chemrev.0c00999Google ScholarThere is no corresponding record for this reference.
- 3Dwivedi, P.; Pillai, D.; Mangal, R. Self-Propelled Swimming Droplets. Curr. Opin. Colloid Interface Sci. 2022, 61, 101614 DOI: 10.1016/j.cocis.2022.101614Google ScholarThere is no corresponding record for this reference.
- 4Kong, B.; Yan, M.; Xie, L.; Tang, J.; Liang, K.; Mei, Y. Recent Advances in Heterosilica-Based Micro/Nanomotors: Designs, Biomedical Applications, and Future Perspectives. Chem. Mater. 2021, 33, 3022– 3046, DOI: 10.1021/acs.chemmater.1c00192Google ScholarThere is no corresponding record for this reference.
- 5Yi, Y.; Sanchez, L.; Gao, Y.; Lee, K.; Yu, Y. Interrogating Cellular Functions with Designer Janus Particles. Chem. Mater. 2017, 29, 1448– 1460, DOI: 10.1021/acs.chemmater.6b05322Google ScholarThere is no corresponding record for this reference.
- 6Karshalev, E.; Kumar, R.; Jeerapan, I.; Castillo, R.; Campos, I.; Wang, J. Multistimuli-Responsive Camouflage Swimmers. Chem. Mater. 2018, 30, 1593– 1601, DOI: 10.1021/acs.chemmater.7b04792Google ScholarThere is no corresponding record for this reference.
- 7Banno, T. Toward Self-Propelled Microrobots: A Systems Chemistry that Induces Non-Linear Phenomena of Oil Droplets in Surfactant Solution. J. Oleo Sci. 2025, 74, 123– 130, DOI: 10.5650/jos.ess24246Google ScholarThere is no corresponding record for this reference.
- 8Yoshizumi, Y.; Suzuki, H. Self-Propelled Metal-Polymer Hybrid Micromachines with Bending and Rotational Motions. ACS Appl. Mater. Interfaces 2017, 9, 21355– 21361, DOI: 10.1021/acsami.7b03656Google ScholarThere is no corresponding record for this reference.
- 9Lin, C.-H.; Kinane, C.; Zhang, Z.; Pena-Francesch, A. Functional Chemical Motor Coatings for Modular Powering of Self-Propelled Particles. ACS Appl. Mater. Interfaces 2022, 14, 39332– 39342, DOI: 10.1021/acsami.2c08061Google ScholarThere is no corresponding record for this reference.
- 10Li, Y.; Zhang, H.; Du, J.; Min, Q.; Wu, X.; Sun, L. Coalescence-Induced Self-Propelled Particle Transport with Asymmetry Arrangement. ACS Appl. Mater. Interfaces 2024, 16, 18184– 18193, DOI: 10.1021/acsami.4c01355Google ScholarThere is no corresponding record for this reference.
- 11Yang, S.; Li, M.; Li, C.; Yan, L.; Li, Q.; Gong, Q.; Li, Y. Droplet-Driven Self-Propelled Devices Fabricated by a Femtosecond Laser. ACS Appl. Mater. Interfaces 2023, 15, 36899– 36907, DOI: 10.1021/acsami.3c04339Google ScholarThere is no corresponding record for this reference.
- 12Wang, J.; Si, J.; Li, J.; Zhang, P.; Wang, Y.; Zhang, W.; Jin, B.; Li, W.; Li, N.; Miao, S. Self-Propelled Nanojets for Fenton Catalysts Based on Halloysite with Embedded Pt and Outside-Grafted Fe3O4. ACS Appl. Mater. Interfaces 2021, 13, 49017– 49026, DOI: 10.1021/acsami.1c13974Google ScholarThere is no corresponding record for this reference.
- 13Chen, L.; Yuan, H.; Chen, S.; Zheng, C.; Wu, X.; Li, Z.; Liang, C.; Dai, P.; Wang, Q.; Ma, X.; Yan, X. Cost-Effective, High-Yield Production of Biotemplated Catalytic Tubular Micromotors as Self-Propelled Microcleaners for Water Treatment. ACS Appl. Mater. Interfaces 2021, 13, 31226– 31235, DOI: 10.1021/acsami.1c03595Google ScholarThere is no corresponding record for this reference.
- 14Fang, J.; Zhang, Y.; Xiao, L.; Jiao, Y.; Tang, X.; Cheng, H.; Cui, Z.; Li, X.; Li, G.; Cao, M.; Zhong, L. Self-Propelled and Electrobraking Synergetic Liquid Manipulator toward Microsampling and Bioanalysis. ACS Appl. Mater. Interfaces 2021, 13, 14741– 14751, DOI: 10.1021/acsami.1c01494Google ScholarThere is no corresponding record for this reference.
- 15Nakata, S.; Hayashima, Y. Spontaneous Motion of a Solid Is Sensitive to the pH of an Aqueous Phase. Langmuir 1999, 15, 1872– 1875, DOI: 10.1021/la981208pGoogle ScholarThere is no corresponding record for this reference.
- 16Suematsu, N. J.; Ikura, Y.; Nagayama, M.; Kitahata, H.; Kawagishi, N.; Murakami, M.; Nakata, S. Mode-Switching of the Self-Motion of a Camphor Boat Depending on the Diffusion Distance of Camphor Molecules. J. Phys. Chem. C 2010, 114, 9876– 9882, DOI: 10.1021/jp101838hGoogle ScholarThere is no corresponding record for this reference.
- 17Kitahata, H.; Aihara, R.; Magome, N.; Yoshikawa, K. Convective and Periodic Motion Driven by a Chemical Wave. J. Chem. Phys. 2002, 116, 5666– 5672, DOI: 10.1063/1.1456023Google ScholarThere is no corresponding record for this reference.
- 18Suematsu, N. J.; Mori, Y.; Amemiya, T.; Nakata, S. Oscillation of Speed of a Self-Propelled Belousov-Zhabotinsky Droplet. J. Phys. Chem. Lett. 2016, 7, 3424– 3428, DOI: 10.1021/acs.jpclett.6b01539Google ScholarThere is no corresponding record for this reference.
- 19Nakata, S.; Arima, Y. Self-Motion of a Phenanthroline Disk on Divalent Metal Ion Aqueous Solutions Coupled with Complex Formation. Colloids Surf., A 2008, 324, 222– 227, DOI: 10.1016/j.colsurfa.2008.04.021Google ScholarThere is no corresponding record for this reference.
- 20Kuze, M.; Kubodera, Y.; Hashishita, H.; Matsuo, M.; Nishimori, H.; Nakata, S. Self-Propulsion Mode Switching of a Briggs–Rauscher Droplet. ChemSystemsChem. 2023, 5, e202200030 DOI: 10.1002/syst.202200030Google ScholarThere is no corresponding record for this reference.
- 21Ikegami, T.; Kojima, H.; Adachi, R. Spatiotemporal Characterization of Emergent Behavior of Self-Propelled Oil Droplet. arXiv 2024, arXiv2412.17381, DOI: 10.48550/arXiv.2412.17381Google ScholarThere is no corresponding record for this reference.
- 22Takabatake, F.; Yoshikawa, K.; Ichikawa, M. Communication: Mode Bifurcation of Droplet Motion under Stationary Laser Irradiation. J. Chem. Phys. 2014, 141, 051103 DOI: 10.1063/1.4892085Google ScholarThere is no corresponding record for this reference.
- 23Kitahata, H.; Koyano, Y.; Löffler, R. J. G.; Górecki, J. Complexity and Bifurcations in the Motion of a Self-Propelled Rectangle Confined in a Circular Aqueous Chamber. Phys. Chem. Chem. Phys. 2022, 24, 20326– 20335, DOI: 10.1039/D2CP02456JGoogle ScholarThere is no corresponding record for this reference.
- 24Shimokawa, M.; Arimitsu, J.; Kitahata, H.; Sakaguchi, H. Transition of the Bifurcations in the Rotation of a Camphor-Coated Elliptical Paper Disk. J. Phys. Soc. Jpn. 2024, 93, 034002 DOI: 10.7566/JPSJ.93.034002Google ScholarThere is no corresponding record for this reference.
- 25Yasugahira, Y.; Nagayama, M. On a Numerical Bifurcation Analysis of a Particle Reaction-Diffusion Model for a Motion of Two Self-Propelled Disks. J. Math. Fluid Mech. 2022, 39, 631– 652, DOI: 10.1007/s13160-021-00498-4Google ScholarThere is no corresponding record for this reference.
- 26Yu, H.; Ren, L.; Wang, Y.; Wang, H.; Zhang, M.; Pan, C.; Yuan, L.; Zhang, J.; Epstein, I. R.; Gao, Q. Chiral Locomotion Transitions of an Active Gel and Their Chemomechanical Origin. J. Am. Chem. Soc. 2025, 147, 5182– 5188, DOI: 10.1021/jacs.4c15550Google ScholarThere is no corresponding record for this reference.
- 27Xu, Y.; Sun, M.; Zhang, L.; Guo, W.; Zhang, Q. Decoupling Nonlinear Motion of the Self-Propelled Disk Sensitive to the Metal Salts. ACS Appl. Mater. Interfaces 2025, 17, 42372– 42379, DOI: 10.1021/acsami.4c22995Google ScholarThere is no corresponding record for this reference.
- 28Levis, D.; Pagonabarraga, I.; Díaz-Guilera, A. Synchronization in Dynamical Networks of Locally Coupled Self-Propelled Oscillators. Phys. Rev. X 2017, 7, 011028 DOI: 10.1103/PhysRevX.7.011028Google ScholarThere is no corresponding record for this reference.
- 29Roy, T.; Chaurasia, S. S.; Cruz, J.-M.; Pimienta, V.; Parmananda, P. Modes of Synchrony in Self-Propelled Pentanol Drops. Soft Matter 2022, 18, 1688– 1695, DOI: 10.1039/D1SM01488AGoogle ScholarThere is no corresponding record for this reference.
- 30Sato, S.; Sakuta, H.; Sadakane, K.; Yoshikawa, K. Self-Synchronous Swinging Motion of a Pair of Autonomous Droplets. ACS Omega 2019, 4, 12766– 12770, DOI: 10.1021/acsomega.9b01533Google ScholarThere is no corresponding record for this reference.
- 31Sharma, J.; Tiwari, I.; Das, D.; Parmananda, P.; Akella, V. S.; Pimienta, V. Rotational Synchronization of Camphor Ribbons. Phys. Rev. E 2019, 99, 012204 DOI: 10.1103/PhysRevE.99.012204Google ScholarThere is no corresponding record for this reference.
- 32Thutupalli, S.; Seemann, R.; Herminghaus, S. Swarming Behavior of Simple Model Squirmers. New J. Phys. 2011, 13, 073021 DOI: 10.1088/1367-2630/13/7/073021Google ScholarThere is no corresponding record for this reference.
- 33Nishiguchi, D.; Sano, M. Mesoscopic Turbulence and Local Order in Janus Particles Self-Propelling under an AC Electric Field. Phys. Rev. E 2015, 92, 052309 DOI: 10.1103/PhysRevE.92.052309Google ScholarThere is no corresponding record for this reference.
- 34Suematsu, N. J.; Nakata, S.; Awazu, A. H.; Nishimori, H. S Collective behavior of inanimate boats. Phys. Rev. E 2010, 81, 056210 DOI: 10.1103/PhysRevE.81.056210Google ScholarThere is no corresponding record for this reference.
- 35Zhang, J.; Laskar, A.; Song, J.; Shklyaev, O. E.; Mou, F.; Guan, J.; Balazs, A. C.; Sen, A. Light-Powered, Fuel-Free Oscillation, Migration, and Reversible Manipulation of Multiple Cargo Types by Micromotor Swarms. ACS Nano 2023, 17, 251– 262, DOI: 10.1021/acsnano.2c07266Google ScholarThere is no corresponding record for this reference.
- 36Liebchen, B.; Cates, M. E.; Marenduzzo, D. Pattern Formation in Chemically Interacting Active Rotors with Self-Propulsion. Soft Matter 2016, 12, 7259– 7264, DOI: 10.1039/C6SM01162DGoogle ScholarThere is no corresponding record for this reference.
- 37Nakata, S.; Hiromatsu, S. Self-Motion of Soap at an Oil-Aqueous Interface. Chem. Phys. Lett. 2005, 405, 39– 42, DOI: 10.1016/j.cplett.2005.02.010Google ScholarThere is no corresponding record for this reference.
- 38Watahiki, Y.; Nomoto, T.; Chiari, L.; Toyota, T.; Fujinami, M. Experimental Investigation of the Self-Propelled Motion of a Sodium Oleate Tablet and Boat at an Oil-Aqueous Interface. Langmuir 2018, 34, 5487– 5494, DOI: 10.1021/acs.langmuir.8b01090Google ScholarThere is no corresponding record for this reference.
- 39Okamoto, Y.; Sasaki, Y.; Nawa-Okita, E.; Yamamoto, D.; Shioi, A. Autonomous Movement System Induced by Synergy between pH Oscillation and a pH-Responsive Oil Droplet. Langmuir 2019, 35, 14266– 14271, DOI: 10.1021/acs.langmuir.9b02072Google ScholarThere is no corresponding record for this reference.
- 40Nawa, E.; Nishigaki, Y.; Yamamoto, D.; Shioi, A. Rhythmic Shape Change of a Vesicle under a pH Gradient. Soft Matter 2013, 9, 7832– 7842, DOI: 10.1039/c3sm51100fGoogle ScholarThere is no corresponding record for this reference.
- 41Nawa, E.; Yamamoto, D.; Shioi, A. Chemotactic Amoeboid-Like Shape Change of a Vesicle under a pH Gradient. Bull. Chem. Soc. Jpn. 2015, 88, 1536– 1544, DOI: 10.1246/bcsj.20150190Google ScholarThere is no corresponding record for this reference.
- 42Hanczyc, M. M.; Toyota, T.; Ikegami, T.; Packard, N.; Sugawara, T. Fatty Acid Chemistry at the Oil–Aqueous Interface: Self-Propelled Oil Droplets. J. Am. Chem. Soc. 2007, 129, 9386– 9391, DOI: 10.1021/ja0706955Google ScholarThere is no corresponding record for this reference.
- 43Takabatake, F.; Magome, N.; Ichikawa, M.; Yoshikawa, K. Spontaneous Mode-Selection in the Self-Propelled Motion of a Solid/Liquid Composite Driven by Interfacial Instability. J. Chem. Phys. 2011, 134, 114704, DOI: 10.1063/1.3567096Google ScholarThere is no corresponding record for this reference.
- 44Nagai, K. H.; Takabatake, F.; Sumino, Y.; Kitahata, H.; Ichikawa, M.; Yoshinaga, N. Rotational Motion of a Droplet Induced by Interfacial Tension. Phys. Rev. E 2013, 87, 013009 DOI: 10.1103/PhysRevE.87.013009Google ScholarThere is no corresponding record for this reference.
- 45Sur, S.; Masoud, H.; Rothstein, J. P. Translational and rotational motion of disk-shaped Marangoni surfers. Phys. Fluids 2019, 31, 102101, DOI: 10.1063/1.5119360Google ScholarThere is no corresponding record for this reference.
- 46Noguchi, M.; Yamada, M.; Sawada, H. Analysis of Different Self-Propulsion Types of Oil Droplets Based on Electrostatic Interaction Effects. RSC Adv. 2022, 12, 18354– 18362, DOI: 10.1039/D2RA02076AGoogle ScholarThere is no corresponding record for this reference.
- 47Johns, W. H.; Bates, T. R. Quantification of the Binding Tendencies of Cholestyramine II: Mechanism of Interaction with Bile Salt and Fatty Acid Salt Anions. J. Pharm. Sci. 1970, 59 (3), 329– 333, DOI: 10.1002/jps.2600590311Google ScholarThere is no corresponding record for this reference.
- 48Nagayama, M.; Nakata, S.; Doi, Y.; Hayashima, Y. A Theoretical and Experimental Study on the Unidirectional Motion of a Camphor Disk. Physica D 2004, 194, 151– 165, DOI: 10.1016/j.physd.2004.02.003Google ScholarThere is no corresponding record for this reference.
- 49Tomoaia-Cotisel, M.; Zsakó, J.; Mocanu, A.; Lupea, M.; Chifu, E. The Ionization Characteristics of Some Fatty Acids at the Air/Water Interface. J. Colloid Interface Sci. 1987, 117, 464– 476, DOI: 10.1016/0021-9797(87)90407-3Google ScholarThere is no corresponding record for this reference.
- 50Nakata, S.; Hiromatsu, S. Self-Motion of Soap at an Oil-Aqueous Interface. Chem. Phys. Lett. 2005, 405, 39– 42, DOI: 10.1016/j.cplett.2005.02.010Google ScholarThere is no corresponding record for this reference.
- 51Kitahata, H.; Kawata, K.; Sumino, Y.; Nakata, S. Oscillation of an Aqueous Surface in Contact with a Fixed Camphor Disk. Chem. Phys. Lett. 2008, 457, 254– 258, DOI: 10.1016/j.cplett.2008.03.075Google ScholarThere is no corresponding record for this reference.
- 52Ren, L.; Wang, M.; Pan, C.; Gao, Q.; Liu, Y.; Epstein, I. R. Autonomous Reciprocating Migration of an Active Material. Proc. Natl. Acad. Sci. U.S.A. 2017, 114, 8704– 8709, DOI: 10.1073/pnas.1704094114Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
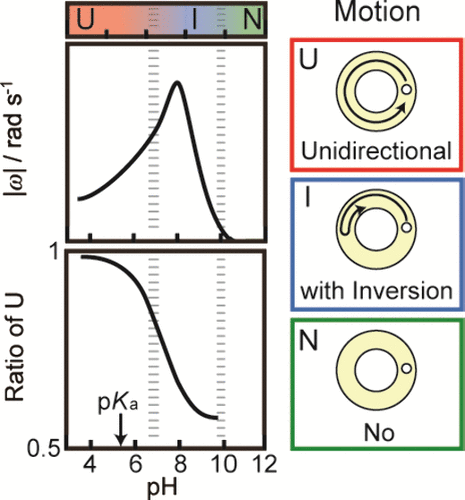
Figure 1
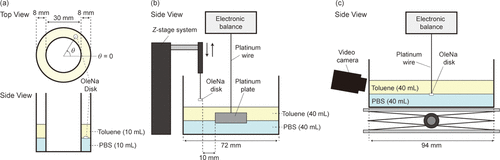
Figure 1. Schematic illustration of (a) the experimental apparatus for observing a self-propelled OleNa disk placed at an oil/aqueous interface, (b) the measurement of the interfacial tension, and (c) the simultaneous measurement of the interfacial tension and movie around the OleNa disk fixed at the oil/aqueous interface. In (a), θ and θ = 0 were defined to analyze the motion in the polar coordinates.
Figure 2
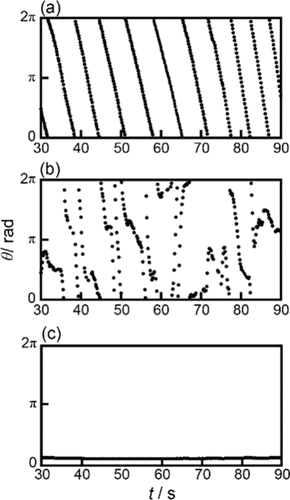
Figure 2. Time variations of θ for self-propulsion of an OleNa disk at pH = (a) 6.0, (b) 8.0, and (c) 12.0. θ and θ = 0 are defined in Figure 1. The movies of motion in parts (a) and (b) are provided in the Supporting Information as Movies S1 and S2, respectively.
Figure 3
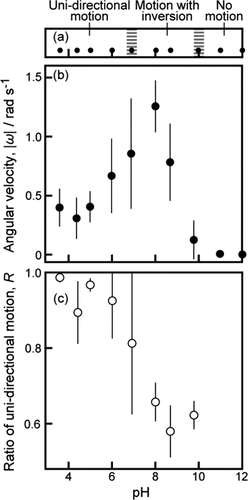
Figure 3. (a) Phase diagram of self-propelled motion, (b) absolute value of angular speed, |ω|, and (c) ratio of unidirectional motion per 1 min, R, depending on pH of the aqueous phase. Error bars represent the standard deviations obtained from three or four examinations. In the boundary regions at pH 6.9 and 9.8 in (a), both unidirectional motion and motion with inversion were observed, and both motion with inversion and no motion were observed, respectively.
Figure 4
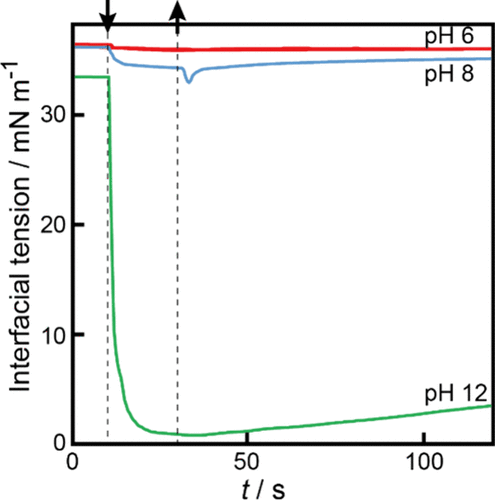
Figure 4. Time variation of the oil/aqueous interfacial tension at pH 6.0 (red line), 8.0 (blue line), and 12.0 (green line). The downward and upward arrows denote the time when the OleNa disk is placed in contact with the interface from above (t = 10 s) and when it is moved upward away from the interface to the air phase (t = 30 s), respectively.
Figure 5
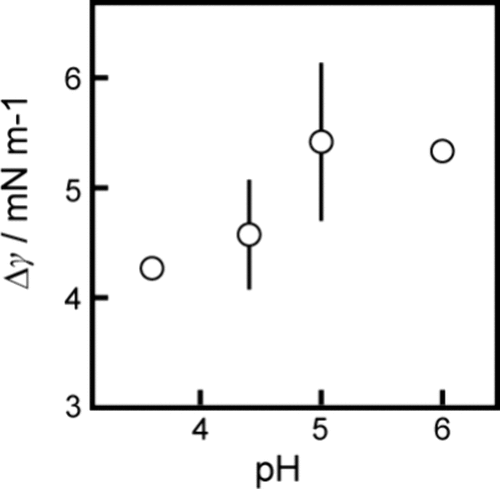
Figure 5. Difference in the interfacial tension, Δγ (= γ0 – γs), at the equilibrium state, depending on the pH of the aqueous phase, where γs and γ0 are the interfacial tension at the oil/aqueous interface with and without OleNa in the aqueous and oil phase, respectively. Error bars represent the standard deviation obtained from three examinations.
Figure 6
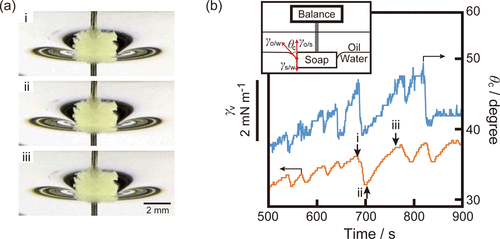
Figure 6. (a) Snapshots of the OleNa disk fixed on the interface at pH 8.0 at t = (i) 680, (ii) 700, and (iii) 760 s (slanted view). The movie at pH 8.0 is provided in the Supporting Information as Movie S3 (500 ≤ t ≤ 900 s). (b) Simultaneous measurement of the time variation of the sum of γs/w, γo/s, and γo/w in the vertical direction (γv) and the complementary contact angle (θc). The data were smoothed by a seven-point moving average (corresponding to ±3 frames, total ∼1.2 s) to reduce noise. The original time resolution was 0.167 s per frame. The vertical position of the disk was adjusted so that the upper surface of the disk almost coincided with the oil/aqueous interface. We confirmed the reproducibility of the oscillatory phenomena.
Figure 7
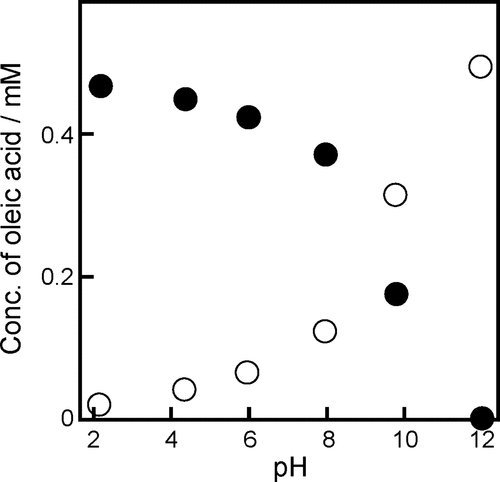
Figure 7. Total concentration of oleic acid in the oil (filled circles) and aqueous (empty circles) phases estimated at different pH values.
Figure 8
References
This article references 52 other publications.
- 1Nakata, S.; Pimienta, V.; Lagzi, I.; Kitahata, H.; Suematsu, N. J. Self-Organized Motion: Physicochemical Design Based on Nonlinear Dynamics; The Royal Society of Cambridge: 2019. DOI: 10.1039/9781788013499-FThere is no corresponding record for this reference.
- 2Soto, F.; Karshalev, E.; Zhang, F.; Esteban Fernandez de Avila, B.; Nourhani, A.; Wang, J. Smart Materials for Microrobots. Chem. Rev. 2022, 122, 3562– 3613, DOI: 10.1021/acs.chemrev.0c00999There is no corresponding record for this reference.
- 3Dwivedi, P.; Pillai, D.; Mangal, R. Self-Propelled Swimming Droplets. Curr. Opin. Colloid Interface Sci. 2022, 61, 101614 DOI: 10.1016/j.cocis.2022.101614There is no corresponding record for this reference.
- 4Kong, B.; Yan, M.; Xie, L.; Tang, J.; Liang, K.; Mei, Y. Recent Advances in Heterosilica-Based Micro/Nanomotors: Designs, Biomedical Applications, and Future Perspectives. Chem. Mater. 2021, 33, 3022– 3046, DOI: 10.1021/acs.chemmater.1c00192There is no corresponding record for this reference.
- 5Yi, Y.; Sanchez, L.; Gao, Y.; Lee, K.; Yu, Y. Interrogating Cellular Functions with Designer Janus Particles. Chem. Mater. 2017, 29, 1448– 1460, DOI: 10.1021/acs.chemmater.6b05322There is no corresponding record for this reference.
- 6Karshalev, E.; Kumar, R.; Jeerapan, I.; Castillo, R.; Campos, I.; Wang, J. Multistimuli-Responsive Camouflage Swimmers. Chem. Mater. 2018, 30, 1593– 1601, DOI: 10.1021/acs.chemmater.7b04792There is no corresponding record for this reference.
- 7Banno, T. Toward Self-Propelled Microrobots: A Systems Chemistry that Induces Non-Linear Phenomena of Oil Droplets in Surfactant Solution. J. Oleo Sci. 2025, 74, 123– 130, DOI: 10.5650/jos.ess24246There is no corresponding record for this reference.
- 8Yoshizumi, Y.; Suzuki, H. Self-Propelled Metal-Polymer Hybrid Micromachines with Bending and Rotational Motions. ACS Appl. Mater. Interfaces 2017, 9, 21355– 21361, DOI: 10.1021/acsami.7b03656There is no corresponding record for this reference.
- 9Lin, C.-H.; Kinane, C.; Zhang, Z.; Pena-Francesch, A. Functional Chemical Motor Coatings for Modular Powering of Self-Propelled Particles. ACS Appl. Mater. Interfaces 2022, 14, 39332– 39342, DOI: 10.1021/acsami.2c08061There is no corresponding record for this reference.
- 10Li, Y.; Zhang, H.; Du, J.; Min, Q.; Wu, X.; Sun, L. Coalescence-Induced Self-Propelled Particle Transport with Asymmetry Arrangement. ACS Appl. Mater. Interfaces 2024, 16, 18184– 18193, DOI: 10.1021/acsami.4c01355There is no corresponding record for this reference.
- 11Yang, S.; Li, M.; Li, C.; Yan, L.; Li, Q.; Gong, Q.; Li, Y. Droplet-Driven Self-Propelled Devices Fabricated by a Femtosecond Laser. ACS Appl. Mater. Interfaces 2023, 15, 36899– 36907, DOI: 10.1021/acsami.3c04339There is no corresponding record for this reference.
- 12Wang, J.; Si, J.; Li, J.; Zhang, P.; Wang, Y.; Zhang, W.; Jin, B.; Li, W.; Li, N.; Miao, S. Self-Propelled Nanojets for Fenton Catalysts Based on Halloysite with Embedded Pt and Outside-Grafted Fe3O4. ACS Appl. Mater. Interfaces 2021, 13, 49017– 49026, DOI: 10.1021/acsami.1c13974There is no corresponding record for this reference.
- 13Chen, L.; Yuan, H.; Chen, S.; Zheng, C.; Wu, X.; Li, Z.; Liang, C.; Dai, P.; Wang, Q.; Ma, X.; Yan, X. Cost-Effective, High-Yield Production of Biotemplated Catalytic Tubular Micromotors as Self-Propelled Microcleaners for Water Treatment. ACS Appl. Mater. Interfaces 2021, 13, 31226– 31235, DOI: 10.1021/acsami.1c03595There is no corresponding record for this reference.
- 14Fang, J.; Zhang, Y.; Xiao, L.; Jiao, Y.; Tang, X.; Cheng, H.; Cui, Z.; Li, X.; Li, G.; Cao, M.; Zhong, L. Self-Propelled and Electrobraking Synergetic Liquid Manipulator toward Microsampling and Bioanalysis. ACS Appl. Mater. Interfaces 2021, 13, 14741– 14751, DOI: 10.1021/acsami.1c01494There is no corresponding record for this reference.
- 15Nakata, S.; Hayashima, Y. Spontaneous Motion of a Solid Is Sensitive to the pH of an Aqueous Phase. Langmuir 1999, 15, 1872– 1875, DOI: 10.1021/la981208pThere is no corresponding record for this reference.
- 16Suematsu, N. J.; Ikura, Y.; Nagayama, M.; Kitahata, H.; Kawagishi, N.; Murakami, M.; Nakata, S. Mode-Switching of the Self-Motion of a Camphor Boat Depending on the Diffusion Distance of Camphor Molecules. J. Phys. Chem. C 2010, 114, 9876– 9882, DOI: 10.1021/jp101838hThere is no corresponding record for this reference.
- 17Kitahata, H.; Aihara, R.; Magome, N.; Yoshikawa, K. Convective and Periodic Motion Driven by a Chemical Wave. J. Chem. Phys. 2002, 116, 5666– 5672, DOI: 10.1063/1.1456023There is no corresponding record for this reference.
- 18Suematsu, N. J.; Mori, Y.; Amemiya, T.; Nakata, S. Oscillation of Speed of a Self-Propelled Belousov-Zhabotinsky Droplet. J. Phys. Chem. Lett. 2016, 7, 3424– 3428, DOI: 10.1021/acs.jpclett.6b01539There is no corresponding record for this reference.
- 19Nakata, S.; Arima, Y. Self-Motion of a Phenanthroline Disk on Divalent Metal Ion Aqueous Solutions Coupled with Complex Formation. Colloids Surf., A 2008, 324, 222– 227, DOI: 10.1016/j.colsurfa.2008.04.021There is no corresponding record for this reference.
- 20Kuze, M.; Kubodera, Y.; Hashishita, H.; Matsuo, M.; Nishimori, H.; Nakata, S. Self-Propulsion Mode Switching of a Briggs–Rauscher Droplet. ChemSystemsChem. 2023, 5, e202200030 DOI: 10.1002/syst.202200030There is no corresponding record for this reference.
- 21Ikegami, T.; Kojima, H.; Adachi, R. Spatiotemporal Characterization of Emergent Behavior of Self-Propelled Oil Droplet. arXiv 2024, arXiv2412.17381, DOI: 10.48550/arXiv.2412.17381There is no corresponding record for this reference.
- 22Takabatake, F.; Yoshikawa, K.; Ichikawa, M. Communication: Mode Bifurcation of Droplet Motion under Stationary Laser Irradiation. J. Chem. Phys. 2014, 141, 051103 DOI: 10.1063/1.4892085There is no corresponding record for this reference.
- 23Kitahata, H.; Koyano, Y.; Löffler, R. J. G.; Górecki, J. Complexity and Bifurcations in the Motion of a Self-Propelled Rectangle Confined in a Circular Aqueous Chamber. Phys. Chem. Chem. Phys. 2022, 24, 20326– 20335, DOI: 10.1039/D2CP02456JThere is no corresponding record for this reference.
- 24Shimokawa, M.; Arimitsu, J.; Kitahata, H.; Sakaguchi, H. Transition of the Bifurcations in the Rotation of a Camphor-Coated Elliptical Paper Disk. J. Phys. Soc. Jpn. 2024, 93, 034002 DOI: 10.7566/JPSJ.93.034002There is no corresponding record for this reference.
- 25Yasugahira, Y.; Nagayama, M. On a Numerical Bifurcation Analysis of a Particle Reaction-Diffusion Model for a Motion of Two Self-Propelled Disks. J. Math. Fluid Mech. 2022, 39, 631– 652, DOI: 10.1007/s13160-021-00498-4There is no corresponding record for this reference.
- 26Yu, H.; Ren, L.; Wang, Y.; Wang, H.; Zhang, M.; Pan, C.; Yuan, L.; Zhang, J.; Epstein, I. R.; Gao, Q. Chiral Locomotion Transitions of an Active Gel and Their Chemomechanical Origin. J. Am. Chem. Soc. 2025, 147, 5182– 5188, DOI: 10.1021/jacs.4c15550There is no corresponding record for this reference.
- 27Xu, Y.; Sun, M.; Zhang, L.; Guo, W.; Zhang, Q. Decoupling Nonlinear Motion of the Self-Propelled Disk Sensitive to the Metal Salts. ACS Appl. Mater. Interfaces 2025, 17, 42372– 42379, DOI: 10.1021/acsami.4c22995There is no corresponding record for this reference.
- 28Levis, D.; Pagonabarraga, I.; Díaz-Guilera, A. Synchronization in Dynamical Networks of Locally Coupled Self-Propelled Oscillators. Phys. Rev. X 2017, 7, 011028 DOI: 10.1103/PhysRevX.7.011028There is no corresponding record for this reference.
- 29Roy, T.; Chaurasia, S. S.; Cruz, J.-M.; Pimienta, V.; Parmananda, P. Modes of Synchrony in Self-Propelled Pentanol Drops. Soft Matter 2022, 18, 1688– 1695, DOI: 10.1039/D1SM01488AThere is no corresponding record for this reference.
- 30Sato, S.; Sakuta, H.; Sadakane, K.; Yoshikawa, K. Self-Synchronous Swinging Motion of a Pair of Autonomous Droplets. ACS Omega 2019, 4, 12766– 12770, DOI: 10.1021/acsomega.9b01533There is no corresponding record for this reference.
- 31Sharma, J.; Tiwari, I.; Das, D.; Parmananda, P.; Akella, V. S.; Pimienta, V. Rotational Synchronization of Camphor Ribbons. Phys. Rev. E 2019, 99, 012204 DOI: 10.1103/PhysRevE.99.012204There is no corresponding record for this reference.
- 32Thutupalli, S.; Seemann, R.; Herminghaus, S. Swarming Behavior of Simple Model Squirmers. New J. Phys. 2011, 13, 073021 DOI: 10.1088/1367-2630/13/7/073021There is no corresponding record for this reference.
- 33Nishiguchi, D.; Sano, M. Mesoscopic Turbulence and Local Order in Janus Particles Self-Propelling under an AC Electric Field. Phys. Rev. E 2015, 92, 052309 DOI: 10.1103/PhysRevE.92.052309There is no corresponding record for this reference.
- 34Suematsu, N. J.; Nakata, S.; Awazu, A. H.; Nishimori, H. S Collective behavior of inanimate boats. Phys. Rev. E 2010, 81, 056210 DOI: 10.1103/PhysRevE.81.056210There is no corresponding record for this reference.
- 35Zhang, J.; Laskar, A.; Song, J.; Shklyaev, O. E.; Mou, F.; Guan, J.; Balazs, A. C.; Sen, A. Light-Powered, Fuel-Free Oscillation, Migration, and Reversible Manipulation of Multiple Cargo Types by Micromotor Swarms. ACS Nano 2023, 17, 251– 262, DOI: 10.1021/acsnano.2c07266There is no corresponding record for this reference.
- 36Liebchen, B.; Cates, M. E.; Marenduzzo, D. Pattern Formation in Chemically Interacting Active Rotors with Self-Propulsion. Soft Matter 2016, 12, 7259– 7264, DOI: 10.1039/C6SM01162DThere is no corresponding record for this reference.
- 37Nakata, S.; Hiromatsu, S. Self-Motion of Soap at an Oil-Aqueous Interface. Chem. Phys. Lett. 2005, 405, 39– 42, DOI: 10.1016/j.cplett.2005.02.010There is no corresponding record for this reference.
- 38Watahiki, Y.; Nomoto, T.; Chiari, L.; Toyota, T.; Fujinami, M. Experimental Investigation of the Self-Propelled Motion of a Sodium Oleate Tablet and Boat at an Oil-Aqueous Interface. Langmuir 2018, 34, 5487– 5494, DOI: 10.1021/acs.langmuir.8b01090There is no corresponding record for this reference.
- 39Okamoto, Y.; Sasaki, Y.; Nawa-Okita, E.; Yamamoto, D.; Shioi, A. Autonomous Movement System Induced by Synergy between pH Oscillation and a pH-Responsive Oil Droplet. Langmuir 2019, 35, 14266– 14271, DOI: 10.1021/acs.langmuir.9b02072There is no corresponding record for this reference.
- 40Nawa, E.; Nishigaki, Y.; Yamamoto, D.; Shioi, A. Rhythmic Shape Change of a Vesicle under a pH Gradient. Soft Matter 2013, 9, 7832– 7842, DOI: 10.1039/c3sm51100fThere is no corresponding record for this reference.
- 41Nawa, E.; Yamamoto, D.; Shioi, A. Chemotactic Amoeboid-Like Shape Change of a Vesicle under a pH Gradient. Bull. Chem. Soc. Jpn. 2015, 88, 1536– 1544, DOI: 10.1246/bcsj.20150190There is no corresponding record for this reference.
- 42Hanczyc, M. M.; Toyota, T.; Ikegami, T.; Packard, N.; Sugawara, T. Fatty Acid Chemistry at the Oil–Aqueous Interface: Self-Propelled Oil Droplets. J. Am. Chem. Soc. 2007, 129, 9386– 9391, DOI: 10.1021/ja0706955There is no corresponding record for this reference.
- 43Takabatake, F.; Magome, N.; Ichikawa, M.; Yoshikawa, K. Spontaneous Mode-Selection in the Self-Propelled Motion of a Solid/Liquid Composite Driven by Interfacial Instability. J. Chem. Phys. 2011, 134, 114704, DOI: 10.1063/1.3567096There is no corresponding record for this reference.
- 44Nagai, K. H.; Takabatake, F.; Sumino, Y.; Kitahata, H.; Ichikawa, M.; Yoshinaga, N. Rotational Motion of a Droplet Induced by Interfacial Tension. Phys. Rev. E 2013, 87, 013009 DOI: 10.1103/PhysRevE.87.013009There is no corresponding record for this reference.
- 45Sur, S.; Masoud, H.; Rothstein, J. P. Translational and rotational motion of disk-shaped Marangoni surfers. Phys. Fluids 2019, 31, 102101, DOI: 10.1063/1.5119360There is no corresponding record for this reference.
- 46Noguchi, M.; Yamada, M.; Sawada, H. Analysis of Different Self-Propulsion Types of Oil Droplets Based on Electrostatic Interaction Effects. RSC Adv. 2022, 12, 18354– 18362, DOI: 10.1039/D2RA02076AThere is no corresponding record for this reference.
- 47Johns, W. H.; Bates, T. R. Quantification of the Binding Tendencies of Cholestyramine II: Mechanism of Interaction with Bile Salt and Fatty Acid Salt Anions. J. Pharm. Sci. 1970, 59 (3), 329– 333, DOI: 10.1002/jps.2600590311There is no corresponding record for this reference.
- 48Nagayama, M.; Nakata, S.; Doi, Y.; Hayashima, Y. A Theoretical and Experimental Study on the Unidirectional Motion of a Camphor Disk. Physica D 2004, 194, 151– 165, DOI: 10.1016/j.physd.2004.02.003There is no corresponding record for this reference.
- 49Tomoaia-Cotisel, M.; Zsakó, J.; Mocanu, A.; Lupea, M.; Chifu, E. The Ionization Characteristics of Some Fatty Acids at the Air/Water Interface. J. Colloid Interface Sci. 1987, 117, 464– 476, DOI: 10.1016/0021-9797(87)90407-3There is no corresponding record for this reference.
- 50Nakata, S.; Hiromatsu, S. Self-Motion of Soap at an Oil-Aqueous Interface. Chem. Phys. Lett. 2005, 405, 39– 42, DOI: 10.1016/j.cplett.2005.02.010There is no corresponding record for this reference.
- 51Kitahata, H.; Kawata, K.; Sumino, Y.; Nakata, S. Oscillation of an Aqueous Surface in Contact with a Fixed Camphor Disk. Chem. Phys. Lett. 2008, 457, 254– 258, DOI: 10.1016/j.cplett.2008.03.075There is no corresponding record for this reference.
- 52Ren, L.; Wang, M.; Pan, C.; Gao, Q.; Liu, Y.; Epstein, I. R. Autonomous Reciprocating Migration of an Active Material. Proc. Natl. Acad. Sci. U.S.A. 2017, 114, 8704– 8709, DOI: 10.1073/pnas.1704094114There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.langmuir.5c04037.
Calibration curve of oleic acid concentration, interfacial tension over time at pH 6.0, steady-state interfacial tension vs pH, spatiotemporal plot on the meniscus around the OleNa disk, pH dependence of deprotonated and protonated oleic acid concentrations, and adsorption behavior of deprotonated and protonated oleic acids at the interface (PDF)
Self-propulsion of the OleNa disk at pH 6.0 (real time) (AVI)
Self-propulsion of the OleNa disk at pH 8.0 (real time) (AVI)
Meniscus around the OleNa disk pH 8.0 (5× speed) (AVI)
Meniscus around the OleNa disk at pH 6.0 (5× speed) (AVI)
Meniscus around the OleNa disk at pH 12.0 (5× speed) (AVI)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



