Chiral Carboxylic Acid Assisted Ir(III)-Catalyzed Asymmetric C–H Activation/Desymmetrization of SulfoximinesClick to copy article linkArticle link copied!
- Rihuang PanRihuang PanDepartment of Chemistry, Zhejiang University, ZJU−NHU United R&D Center, Hangzhou 310058, ChinaMore by Rihuang Pan
- Hanxiao ShenHanxiao ShenDepartment of Chemistry, Zhejiang University, ZJU−NHU United R&D Center, Hangzhou 310058, ChinaMore by Hanxiao Shen
- Yinong WuYinong WuDepartment of Chemistry, Zhejiang University, ZJU−NHU United R&D Center, Hangzhou 310058, ChinaMore by Yinong Wu
- Yongtao Wang*Yongtao Wang*[email protected]Department of Chemistry, Zhejiang University, ZJU−NHU United R&D Center, Hangzhou 310058, ChinaMore by Yongtao Wang
- Xufeng Lin*Xufeng Lin*[email protected]Department of Chemistry, Zhejiang University, ZJU−NHU United R&D Center, Hangzhou 310058, ChinaMore by Xufeng Lin
Abstract
The development of privileged chiral carboxylic acids is crucial for enabling asymmetric C–H activation catalyzed by achiral high-valent d6 transition metals. We report the synthesis of a new chiral carboxylic acid that enables Ir(III)-catalyzed asymmetric C–H activation/desymmetrization of sulfoximines with α-bromo ketones. A diverse range of sulfur-stereogenic sulfoximines were synthesized with high yields (up to 90%) and excellent enantioselectivity (up to 97:3 er). The reaction maintained its efficiency upon scale-up, and one of the products 6aa could be readily transformed into the five-membered chiral cyclic sulfoximine derivative. Detailed mechanistic studies establish C–H bond cleavage as the rate-determining step. Furthermore, the alkylated reaction intermediate was successfully isolated and demonstrated complete retention of enantioselectivity upon conversion to the corresponding product.
This publication is licensed for personal use by The American Chemical Society.
Transition-metal-catalyzed asymmetric C–H activation provides an efficient and atom-economical strategy for introducing chirality into inert C–H bonds. (1) Among the catalysts employed in such reactions, half-sandwich high-valent d6 transition metal (Co(III), Rh(III), Ir(III), Ru(II)) complexes exhibit exceptional efficacy. (1e,2) However, the structural constraints of these complexes typically limit the metal center to only one vacant coordination site for ligand binding, which presents a significant challenge for the design of effective chiral ligands. (2b,3) Over the past decade, high-valent d6 transition-metal complexes incorporating tailored chiral cyclopentadienyl (Cpx) (4) or chiral aryl (5) ligands have emerged as highly efficient catalysts for asymmetric C–H activation. Despite their exceptional enantiocontrol in C–H functionalization, the widespread adoption of these sophisticated chiral ligands is hampered by their laborious, multistep synthesis. In recent years, the synergistic combination of achiral half-sandwich high-valent d6 metal complexes with monodentate chiral carboxylic acids (CCAs) has emerged as a powerful strategy for achieving highly enantioselective C–H activation, as pioneered by the research groups of Cramer, (4) Ackermann, (6) Matsunaga and Yoshino, (7) Shi, (8) and He (9) (Scheme 1). Traditional designs of chiral carboxylic acids (CCAs) predominantly relied on steric control, whereas recent advances have demonstrated that incorporating noncovalent interactions (NCIs) into CCA ligands also enables highly enantioselective C–H activation. However, the limited structural diversity of chiral carboxylic acids and scarce catalytic systems for high enantioselectivity make developing new scaffolds essential for advancing the field.
Scheme 1
Sulfur-stereogenic sulfoximines constitute privileged structural motifs in pharmaceuticals, agrochemicals, and bioactive molecules, rendering their asymmetric synthesis a subject of enduring interest in organic chemistry. (10) Current approaches for constructing chiral sulfoximines via C–H activation primarily involve desymmetrization and kinetic resolution strategies, with the former offering superior synthetic efficiency. Recent advances in sulfoximine desymmetrization have been realized through two principal catalytic systems: Cpx–metal complexes or achiral Cp catalysts combined with chiral carboxylic acids. Previous studies have identified diazo compounds, (8d,11) sulfur ylides, (5c,7f,8b) iodonium ylides, (12) dioxazolones, (7g,8a,e) alkynes (8g) and α,β-unsaturated ketones (8f) as competent coupling partners, enabling access to target sulfoximines with high enantioselectivity. Notably, the Yi group developed a Cpx Rh(III)-catalyzed desymmetrization of sulfoximines with α-chloro ketones, affording chiral sulfoximines in good yield and enantioselectivity. (13) Despite these achievements, the use of achiral Cp catalysts in conjunction with chiral carboxylic acids for asymmetric C–H activation of sulfoximines with α-halo ketones remains an unexplored avenue of research.
Our group has established a platform for chiral spirocyclic phosphoric acid (SPA) catalysis, focusing on new synthetic methodologies. (14) Recently, we achieved a highly enantioselective synthesis of 3-aryl-3-indolizine-substituted isoindolin-1-ones, enabling efficient access to diverse CCAs with tunable electronic and steric properties. (15) Herein, we report that these new CCAs enable highly enantioselective Ir(III)-catalyzed C–H activation and desymmetrizing cyclization of sulfoximines with α-bromo ketones, enabling the efficient construction of sulfur-stereogenic sulfoximines with a broad substrate scope, good yields, and high enantioselectivity (Scheme 2). Mechanistic studies indicate that C–H bond cleavage is rate-limiting, and a key alkylated intermediate was isolated and converted to the product with complete stereoretention.
Scheme 2
Building upon our previous findings, we first developed a series of novel chiral carboxylic acids through a modular synthetic approach (Scheme 3). The chiral spirocyclic phosphoric acid-catalyzed reaction between isoindolinone 1 and indolizine 2 initially provided the corresponding chiral ester intermediate, which upon basic hydrolysis yielded the CCA 3. Systematic variation of the aryl substituents on 3 enabled the incorporation of diverse electron-donating and electron-withdrawing groups, providing access to 12 distinct derivatives (3a-3l) with good to excellent enantioselectivity (94:6 to >99.5:0.5 er). Furthermore, the enantiopurity of 3l could be elevated to >99.5:0.5 er through improved after-treatment, or by chiral resolution in gram-scale synthesis (see Supporting Information for details).
Scheme 3
We then optimized the Ir(III)-catalyzed asymmetric C–H activation/desymmetrization of sulfoximine 4a with α-bromo ketone 5a (Table 1). Initially, both sterically hindered tert-leucine derivatives 7 and 8 were tested and gave good yields of 6aa, but monodentate acid 7 outperformed bidentate 8 (86% yield, 82.5:17.5 er, entries 1 and 2). Subsequent evaluation of Shi group’s chiral binaphthyl monocarboxylic acids 9 revealed that analogue 9b offered improved stereocontrol (87:13 er) over 9a and 9c, though still insufficient (entries 3 to 5). We then investigated our newly developed CCAs 3. Phenyl-substituted 3a achieved 80% yield with 91:9 er (entry 6). However, adding a para-methoxy group (3b) reduced enantioselectivity (81:19 er, entry 7). In contrast, para-electron-withdrawing groups consistently enhanced enantioselectivity. The para-halogenated catalysts (3c–3e) gave moderate to good yields (62–76%) with good and consistent enantioselectivity (88.5:11.5 to 91.5:8.5 er), with chloro-substituted 3d performing best (entries 8 to 10). The para-trifluoromethyl analogue 3f gave slightly lower enantioselectivity (88.5:11.5 er, entry 11). Building on the optimal para-chloro substitution, meta-chloro variants 3g and 3h showed no further improvement (entries 12 and 13). To enhance enantioselectivity via steric modulation while retaining the para-chloro group, we modified the aryl ring. Mono-meta-substituted variants (3i–3k) were ineffective (entries 14 to 16), whereas the 3,4,5-trichlorophenyl analogue 3l showed dramatically improved performance (83% yield, 95:5 er, entry 17). This improvement is attributed to restricted rotation in the doubly meta-substituted system, which creates a rigid chiral environment, unlike the flexible monosubstituted analogues. Temperature screening identified 100 °C as optimal (85% yield, 95.5:4.5 er). Higher temperatures or longer reaction times reduced both yield and enantioselectivity (entries 18 to 21).

| entry | CCA ligand | yield (%)b | erc |
|---|---|---|---|
| 1 | 7 | 86 | 17.5:82.5 |
| 2 | 8 | 73 | 77:23 |
| 3 | 9a | 50 | 70.5:29.5 |
| 4 | 9b | 41 | 87:13 |
| 5 | 9c | 44 | 75:25 |
| 6 | 3a | 80 | 91:9 |
| 7 | 3b | 82 | 81:19 |
| 8 | 3c | 76 | 89:11 |
| 9 | 3d | 72 | 91.5:8.5 |
| 10 | 3e | 62 | 88.5:11.5 |
| 11 | 3f | 68 | 88.5:11.5 |
| 12 | 3g | 77 | 89.5:10.5 |
| 13 | 3h | 69 | 85:15 |
| 14 | 3i | 86 | 90:10 |
| 15 | 3j | 68 | 89:11 |
| 16 | 3k | 65 | 87.5:12.5 |
| 17 | 3l | 83 | 95:5 |
| 18e | 3l | 85 | 95:5 |
| 19f | 3l | 85(70)d | 95.5:4.5 |
| 20g | 3l | 83 | 94:6 |
| 21f,h | 3l | 79 | 92.5:7.5 |
Reaction conditions: 4a (0.1 mmol), 5a (0.12 mmol), [Cp*IrCl2]2 (5 mol %), Ag2CO3 (0.75 equiv), CCA (15 mol %) and NHEt2 (30 mol %) in 0.5 mL HFIP at 60 °C for 24 h.
1H NMR yield using 1,3,5-trimethoxybenzene as the internal standard.
The er value was determined by chiral HPLC.
Isolated yield.
80 °C.
100 °C.
120 °C.
48 h.
With optimized conditions established, we investigated the substrate scope (Table 2A). Various para-substituted sulfoximines proved compatible. Electron-donating alkyl groups (methyl 6ba, ethyl 6ca, isopropyl 6da) afforded products in good to excellent yields (76–87%) and selectivity (94:6 to 96:4 er), though tert-butyl and trimethylsilyl groups lowered enantioselectivity. The para-methoxy substrate 6ga maintained good stereocontrol (92:8 er, 69% yield). Electron-withdrawing para-substituents also performed well: trifluoromethoxy (6ha), phenyl (6ia), and halogen-substituted derivatives (6ja–6la) gave moderate to good yields (50–87%) with good enantioselectivity (90:10 to 94:6 er). Strong electron-withdrawing groups such as trifluoromethyl (6ma) and ester (6na) were also tolerated, though acetyl-, cyano-, and nitro-substituted substrates showed significantly reduced yield and selectivity. For sterically demanding or strongly electron-deficient substrates, the less hindered catalyst 3d generally improved both efficiency and enantioselectivity. Naphthyl substrates (6sa and 6ta) achieved outstanding enantioselectivity (96:4 er). Meta-substituted phenyl groups, presenting two reactive sites, gave diverse outcomes: the meta-methyl substrate 6ua reacted selectively at the less hindered position (74% yield, 97:3 er); the meta-methoxy analogue produced two regioisomers, with the less hindered isomer 6va dominating in yield and the more congested isomer 6va′ showing superior selectivity; the meta-chloro substrate furnished both isomers in nearly equal yields and comparably high enantioselectivity. Ortho-substituted sulfoximines exhibited excellent reactivity: ortho-methyl and ortho-methoxy substrates afforded products in 90% yield (96:4 er) and 64% yield (95:5 er), respectively. The methodology was also successfully extended to the parallel kinetic resolution of asymmetric sulfoximine 4z, delivering both enantiomeric products (6za and 6za′) in comparable yields and with good enantioselectivity.

4 (0.1 mmol), 5 (0.12 mmol), [Cp*IrCl2]2 (5 mol %), Ag2CO3 (0.75 equiv), 3l (15 mol %), NHEt2 (30 mol %) in HFIP (0.5 mL) at 100 °C for 24 h in a seal tube.
3d was used instead of 3l.
We subsequently explored the substrate scope with respect to α-bromo ketones 5 using sulfoximine 4b as the model substrate (Table 2B). Difluoromethyl- and pentafluoroethyl-substituted α-bromo ketones reacted efficiently to afford products 6bb and 6bc in 71% yield (94:6 er) and 64% yield (92:8 er), respectively. Ester-substituted α-bromo ketones also performed well, delivering chiral sulfoximines with good to excellent enantioselectivity (6bd, 93:7 er; 6be, 95:5 er). The acetyl-substituted analogue also performed satisfactorily, providing product 6bf in 68% yield with 91.5:8.5 er. While cyclic α-bromo ketones remained reactive, their products showed somewhat diminished enantioselectivity (6bg, 89:11 er). However, ketone with phenyl group lacking strong electron-withdrawing properties failed to produce the target compound (6bh).
To establish the practical utility of this methodology, we performed a scale-up synthesis (1 mmol scale) under the optimized conditions (Scheme 4A). The transformation proceeded efficiently to afford chiral sulfoximine 6aa in 71% isolated yield with excellent retention of enantioselectivity (96:4 er), demonstrating the robustness and scalability of this catalytic system. Furthermore, the synthetic value of product 6aa was highlighted by its successful oxidation using m-CPBA, which cleanly furnished the five-membered cyclic sulfoximine 10 in 84% yield while completely preserving the stereochemical integrity (Scheme 4B).
Scheme 4
To gain mechanistic insight, we conducted detailed experiments (Scheme 5). Treatment of sulfoximine 4b in d-HFIP under standard conditions without 5a gave low ortho-aryl H/D exchange, indicating that C–H activation is slow and likely irreversible. In the presence of 5a, product 6aa displayed distinct deuteration patterns (Scheme 5A). Substantial deuterium incorporation at the alkene position suggests rapid and reversible cyclization from the reaction intermediate to the final product. While deuterium levels on the unactivated phenyl ring matched those without 5a, the unactivated ortho-position of the activated ring exhibited enhanced exchange. This can be attributed to increased electron density from an intermediate species, which raises the reactivity of this ring and facilitates deuterium incorporation at its otherwise unreactive ortho-position. A primary kinetic isotope effect (kH/kD = 5.7) was measured, confirming C–H bond cleavage as the rate-determining step (Scheme 5B). A strong linear correlation (R2 = 0.98) between the enantiopurity of CCA 3l and product 6aa suggests that a monomeric CCA species participates in the enantio-determining step. Intermolecular competition experiments revealed a preference for electron-donating over electron-withdrawing sulfoximines, supporting an electrophilic cyclization pathway. Control experiments further elucidated the mechanism. Adding TEMPO under standard conditions did not yield radical-trapping adducts, and 6aa was still obtained with moderate yield and enantioselectivity, ruling out a radical pathway. No C–H activation occurred with the N-methyl-protected substrate 4a′, confirming the essential role of the NH group. When N-methyl-protected CCA 3l′ was used, enantioselectivity dropped significantly, indicating that the NH moiety in 3l is crucial for high stereocontrol (see Supporting Information for details). Building on these results, reactive intermediates were isolated under modified conditions (lower temperature, shorter time) as a mixture of alkylated intermediate 11 and cyclized intermediate 11′, along with minor amounts of 6aa possessing comparable er (Scheme 5C). Subjecting this mixture to standard conditions afforded 6aa in 86% yield and 95:5 er, confirming 11/11′ as genuine intermediates that undergo dehydration to form the final product. On the basis of these mechanistic investigations and prior reports, (8d,13,16) a plausible catalytic cycle with a postulated stereoinduction model involving hydrogen bonding interaction is proposed for this transformation. (see the Supporting Information for details).
Scheme 5
In summary, we have developed a new chiral carboxylic acid scaffold and successfully applied it to an iridium-catalyzed asymmetric C–H activation/desymmetrization of sulfoximines with α-bromo ketones. This method provides access to a wide range of sulfur-stereogenic sulfoximines in high yields (up to 90%) with excellent enantioselectivities (up to 97:3 er). The practicality of this transformation was demonstrated through scale-up synthesis and downstream derivatizations. Detailed mechanistic studies established C–H bond cleavage as the rate-determining step and the reaction intermediate was successfully isolated. We anticipate that this CCA design will inspire further developments in transition-metal-catalyzed asymmetric C–H functionalization. Current efforts in our laboratory are focused on expanding the application scope of this catalytic system to other challenging transformations.
Data Availability
The data underlying this study are available in the published article and it is Supporting Information.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.5c04553.
Experimental procedures, Characterization details, Mechanic studies, Crystallographic data for compound 3l (CCDC 2498768) and 6xa (CCDC 2484114), NMR and HPLC spectra. (PDF)
Deposition Numbers 2484114 and 2498768 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
Financial support from the National Natural Science Foundation of China (22071213) is gratefully acknowledged. We would also like to thank Mr Jiyong Liu for X-ray diffraction analysis at the Chemistry Instrumentation Center Zhejiang University.
References
This article references 16 other publications.
- 1(a) Newton, C. G.; Wang, S.-G.; Oliveira, C. C.; Cramer, N. Catalytic Enantioselective Transformations Involving C-H Bond Cleavage by Transition-Metal Complexes. Chem. Rev. 2017, 117, 8908– 8976, DOI: 10.1021/acs.chemrev.6b00692Google ScholarThere is no corresponding record for this reference.(b) Saint-Denis, T. G.; Zhu, R.-Y.; Chen, G.; Wu, Q.-F.; Yu, J.-Q. Enantioselective C(Sp3)-H Bond Activation by Chiral Transition Metal Catalysts. Science 2018, 359, eaao4798 DOI: 10.1126/science.aao4798Google ScholarThere is no corresponding record for this reference.(c) Loup, J.; Dhawa, U.; Pesciaioli, F.; Wencel-Delord, J.; Ackermann, L. Enantioselective C-H Activation with Earth-Abundant 3d Transition Metals. Angew. Chem., Int. Ed. 2019, 58, 12803– 12818, DOI: 10.1002/anie.201904214Google ScholarThere is no corresponding record for this reference.(d) Yoshino, T.; Satake, S.; Matsunaga, S. Diverse Approaches for Enantioselective C-H Functionalization Reactions Using Group 9 CpxMIII Catalysts. Chem. - Eur. J. 2020, 26, 7346– 7357, DOI: 10.1002/chem.201905417Google ScholarThere is no corresponding record for this reference.(e) Shi, B.-F.; Zhang, Y.-H.; Lam, J. K.; Wang, D.-H.; Yu, J.-Q. Pd(II)-Catalyzed Enantioselective C–H Olefination of Diphenylacetic Acids. J. Am. Chem. Soc. 2010, 132, 460– 461, DOI: 10.1021/ja909571zGoogle ScholarThere is no corresponding record for this reference.(f) Cheng, X.-F.; Li, Y.; Su, Y.-M.; Yin, F.; Wang, J.-Y.; Sheng, J.; Vora, H. U.; Wang, X.-S.; Yu, J.-Q. Pd(II)-Catalyzed Enantioselective C–H Activation/C–O Bond Formation: Synthesis of Chiral Benzofuranones. J. Am. Chem. Soc. 2013, 135, 1236– 1239, DOI: 10.1021/ja311259xGoogle ScholarThere is no corresponding record for this reference.
- 2(a) Wozniak, Łu.; Tan, J.-F.; Nguyen, Q.-H.; Madron du Vigne, A.; Smal, V.; Cao, Y.-X.; Cramer, N. Catalytic Enantioselective Functionalizations of C-H Bonds by Chiral Iridium Complexes. Chem. Rev. 2020, 120, 10516– 10543, DOI: 10.1021/acs.chemrev.0c00559Google ScholarThere is no corresponding record for this reference.(b) Yoshino, T.; Matsunaga, S. Chiral Carboxylic Acid Assisted Enantioselective C-H Activation with Achiral CpxMIII (M = Co, Rh, Ir) Catalysts. ACS Catal. 2021, 11, 6455– 6466, DOI: 10.1021/acscatal.1c01351Google ScholarThere is no corresponding record for this reference.(c) Liang, H.; Wang, J. Enantioselective C-H Bond Functionalization Involving Arene Ruthenium(II) Catalysis. Chem. - Eur. J. 2023, 29, e202202461 DOI: 10.1002/chem.202202461Google ScholarThere is no corresponding record for this reference.(d) Liu, C.-X.; Yin, S.-Y.; Zhao, F.; Yang, H.; Feng, Z.; Gu, Q.; You, S.-L. Rhodium-Catalyzed Asymmetric C-H Functionalization Reactions. Chem. Rev. 2023, 123, 10079– 10134, DOI: 10.1021/acs.chemrev.3c00149Google ScholarThere is no corresponding record for this reference.(e) Yao, Q.-J.; Shi, B.-F. Cobalt(III)-Catalyzed Enantioselective C-H Functionalization: Ligand Innovation and Reaction Development. Acc. Chem. Res. 2025, 58, 971– 990, DOI: 10.1021/acs.accounts.5c00013Google ScholarThere is no corresponding record for this reference.
- 3Yoshino, T.; Matsunaga, S. Cp*CoIII-Catalyzed C-H Functionalization and Asymmetric Reactions Using External Chiral Sources. Synlett 2019, 30, 1384– 1400, DOI: 10.1055/s-0037-1611814Google ScholarThere is no corresponding record for this reference.
- 4Mas-Roselló, J.; Herraiz, A. G.; Audic, B.; Laverny, A.; Cramer, N. Chiral Cyclopentadienyl Ligands: Design, Syntheses, and Applications in Asymmetric Catalysis. Angew. Chem., Int. Ed. 2021, 60, 13198– 13224, DOI: 10.1002/anie.202008166Google ScholarThere is no corresponding record for this reference.
- 5(a) Liang, H.; Guo, W.; Li, J.; Jiang, J.; Wang, J. Chiral Arene Ligand as Stereocontroller for Asymmetric C-H Activation. Angew. Chem., Int. Ed. 2022, 61, e202204926 DOI: 10.1002/anie.202204926Google ScholarThere is no corresponding record for this reference.(b) Li, J.; Wang, G.; Guo, W.; Jiang, J.; Wang, J. H8-BINOL-Derived Chiral η6-Benzene Ligands: New Opportunities for the Ruthenium-Catalyzed Asymmetric C-H Activation. Angew. Chem., Int. Ed. 2024, 63, e202405782 DOI: 10.1002/anie.202405782Google ScholarThere is no corresponding record for this reference.(c) Liu, H.; Jiang, J.-J.; Wang, J. Catalytic Asymmetric C-H Activation/Cyclization of Sulfoximines with Sulfoxonium Ylides by a Chiral η6-Benzene Ruthenium(II) Catalyst. ACS Catal. 2024, 14, 17398– 17404, DOI: 10.1021/acscatal.4c04798Google ScholarThere is no corresponding record for this reference.
- 6(a) Pesciaioli, F.; Dhawa, U.; Oliveira, J. C. A.; Yin, R.; John, M.; Ackermann, L. Enantioselective Cobalt(III)-Catalyzed C-H Activation Enabled by Chiral Carboxylic Acid Cooperation. Angew. Chem., Int. Ed. 2018, 57, 15425– 15429, DOI: 10.1002/anie.201808595Google ScholarThere is no corresponding record for this reference.(b) Li, Y.; Liou, Y.; Oliveira, J. C. A.; Ackermann, L. Ruthenium(II)/Imidazolidine Carboxylic Acid-Catalyzed C-H Alkylation for Central and Axial Double Enantio-Induction. Angew. Chem., Int. Ed. 2022, 61, e202212595 DOI: 10.1002/anie.202212595Google ScholarThere is no corresponding record for this reference.(c) Zhang, Z.-J.; Li, S.-W.; Oliveira, J. C. A.; Li, Y.; Chen, X.; Zhang, S.-Q.; Xu, L.-C.; Rogge, T.; Hong, X.; Ackermann, L. Data-Driven Design of New Chiral Carboxylic Acid for Construction of Indoles with C-Central and C–N Axial Chirality via Cobalt Catalysis. Nat. Commun. 2023, 14, 3149, DOI: 10.1038/s41467-023-38872-0Google ScholarThere is no corresponding record for this reference.
- 7(a) Lin, L.; Fukagawa, S.; Sekine, D.; Tomita, E.; Yoshino, T.; Matsunaga, S. Chiral Carboxylic Acid Enabled Achiral Rhodium(III)-Catalyzed Enantioselective C-H Functionalization. Angew. Chem., Int. Ed. 2018, 57, 12048– 12052, DOI: 10.1002/anie.201807610Google ScholarThere is no corresponding record for this reference.(b) Fukagawa, S.; Kato, Y.; Tanaka, R.; Kojima, M.; Yoshino, T.; Matsunaga, S. Enantioselective C(Sp3)-H Amidation of Thioamides Catalyzed by a CobaltIII/Chiral Carboxylic Acid Hybrid System. Angew. Chem., Int. Ed. 2019, 58, 1153– 1157, DOI: 10.1002/anie.201812215Google ScholarThere is no corresponding record for this reference.(c) Sekine, D.; Ikeda, K.; Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S. Chiral 2-Aryl Ferrocene Carboxylic Acids for the Catalytic Asymmetric C(Sp3)-H Activation of Thioamides. Organometallics 2019, 38, 3921– 3926, DOI: 10.1021/acs.organomet.9b00407Google ScholarThere is no corresponding record for this reference.(d) Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S. Catalytic Enantioselective Methylene C(Sp3)-H Amidation of 8-Alkylquinolines Using a Cp*RhIII/Chiral Carboxylic Acid System. Angew. Chem., Int. Ed. 2019, 58, 18154– 18158, DOI: 10.1002/anie.201911268Google ScholarThere is no corresponding record for this reference.(e) Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S. Development of Pseudo-C2-Symmetric Chiral Binaphthyl Monocarboxylic Acids for Enantioselective C(Sp3)-H Functionalization Reactions under Rh(III) Catalysis. ACS Catal. 2021, 11, 4271– 4277, DOI: 10.1021/acscatal.1c00765Google ScholarThere is no corresponding record for this reference.(f) Yoshino, T.; Matsunaga, S.; Huang, L.-T.; Hirata, Y.; Kato, Y.; Lin, L.; Kojima, M. Ruthenium(II)/Chiral Carboxylic Acid Catalyzed Enantioselective C-H Functionalization of Sulfoximines. Synthesis 2022, 54, 4703– 4710, DOI: 10.1055/a-1588-0072Google ScholarThere is no corresponding record for this reference.(g) Hirata, Y.; Sekine, D.; Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S. Cobalt(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective Synthesis of Benzothiadiazine-1-Oxides via C-H Activation. Angew. Chem., Int. Ed. 2022, 61, e202205341 DOI: 10.1002/anie.202205341Google ScholarThere is no corresponding record for this reference.(h) Huang, L.; Kitakawa, Y.; Yamada, K.; Kamiyama, F.; Kojima, M.; Yoshino, T.; Matsunaga, S. Enantioselective Synthesis of 1,2-Benzothiazine 1-Imines via RuII/Chiral Carboxylic Acid-Catalyzed C-H Alkylation/Cyclization. Angew. Chem., Int. Ed. 2023, 62, e202305480 DOI: 10.1002/anie.202305480Google ScholarThere is no corresponding record for this reference.
- 8(a) Liu, L.; Song, H.; Liu, Y.-H.; Wu, L.-S.; Shi, B.-F. Achiral CpxIr(III)/Chiral Carboxylic Acid Catalyzed Enantioselective C-H Amidation of Ferrocenes under Mild Conditions. ACS Catal. 2020, 10, 7117– 7122, DOI: 10.1021/acscatal.0c02049Google ScholarThere is no corresponding record for this reference.(b) Zhou, T.; Qian, P.-F.; Li, J.-Y.; Zhou, Y.-B.; Li, H.-C.; Chen, H.-Y.; Shi, B.-F. Efficient Synthesis of Sulfur-Stereogenic Sulfoximines via Ru(II)-Catalyzed Enantioselective C–H Functionalization Enabled by Chiral Carboxylic Acid. J. Am. Chem. Soc. 2021, 143, 6810– 6816, DOI: 10.1021/jacs.1c03111Google ScholarThere is no corresponding record for this reference.(c) Liu, Y.-H.; Xie, P.-P.; Liu, L.; Fan, J.; Zhang, Z.-Z.; Hong, X.; Shi, B.-F. Cp*Co(III)-Catalyzed Enantioselective Hydroarylation of Unactivated Terminal Alkenes via C–H Activation. J. Am. Chem. Soc. 2021, 143, 19112– 19120, DOI: 10.1021/jacs.1c08562Google ScholarThere is no corresponding record for this reference.(d) Li, J.-Y.; Xie, P.-P.; Zhou, T.; Qian, P.-F.; Zhou, Y.-B.; Li, H.-C.; Hong, X.; Shi, B.-F. Ir(III)-Catalyzed Asymmetric C–H Activation/Annulation of Sulfoximines Assisted by the Hydrogen-Bonding Interaction. ACS Catal. 2022, 12, 9083– 9091, DOI: 10.1021/acscatal.2c00962Google ScholarThere is no corresponding record for this reference.(e) Zhou, Y.-B.; Zhou, T.; Qian, P.-F.; Li, J.-Y.; Shi, B.-F. Synthesis of Sulfur-Stereogenic Sulfoximines via Co(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Amidation. ACS Catal. 2022, 12, 9806– 9811, DOI: 10.1021/acscatal.2c02691Google ScholarThere is no corresponding record for this reference.(f) Qian, P.-F.; Zhou, T.; Li, J.-Y.; Zhou, Y.-B.; Shi, B.-F. Ru(II)/Chiral Carboxylic Acid-Catalyzed Asymmetric [4 + 3] Annulation of Sulfoximines with α,β-Unsaturated Ketones. ACS Catal. 2022, 12, 13876– 13883, DOI: 10.1021/acscatal.2c03531Google ScholarThere is no corresponding record for this reference.(g) Zhou, G.; Zhou, T.; Jiang, A.; Qian, P.; Li, J.; Jiang, B.; Chen, Z.; Shi, B. Electrooxidative Rhodium(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Annulation of Sulfoximines with Alkynes. Angew. Chem., Int. Ed. 2024, 63, e202319871 DOI: 10.1002/anie.202319871Google ScholarThere is no corresponding record for this reference.
- 9Liu, W.; Yang, W.; Zhu, J.; Guo, Y.; Wang, N.; Ke, J.; Yu, P.; He, C. Dual-Ligand-Enabled Ir(III)-Catalyzed Enantioselective C–H Amidation for the Synthesis of Chiral Sulfoxides. ACS Catal. 2020, 10, 7207– 7215, DOI: 10.1021/acscatal.0c02109Google ScholarThere is no corresponding record for this reference.
- 10(a) Lücking, U. Sulfoximines: A Neglected Opportunity in Medicinal Chemistry. Angew. Chem., Int. Ed. 2013, 52, 9399– 9408, DOI: 10.1002/anie.201302209Google ScholarThere is no corresponding record for this reference.(b) Mäder, P.; Kattner, L. Sulfoximines as Rising Stars in Modern Drug Discovery? Current Status and Perspective on an Emerging Functional Group in Medicinal Chemistry. J. Med. Chem. 2020, 63, 14243– 14275, DOI: 10.1021/acs.jmedchem.0c00960Google ScholarThere is no corresponding record for this reference.
- 11(a) Sun, Y.; Cramer, N. Enantioselective Synthesis of Chiral-At-Sulfur 1,2-Benzothiazines by CpxRhIII-Catalyzed C-H Functionalization of Sulfoximines. Angew. Chem., Int. Ed. 2018, 57, 15539– 15543, DOI: 10.1002/anie.201810887Google ScholarThere is no corresponding record for this reference.(b) Shen, B.; Wan, B.; Li, X. Enantiodivergent Desymmetrization in the Rhodium(III)-Catalyzed Annulation of Sulfoximines with Diazo Compounds. Angew. Chem., Int. Ed. 2018, 57, 15534– 15538, DOI: 10.1002/anie.201810472Google ScholarThere is no corresponding record for this reference.(c) Brauns, M.; Cramer, N. Efficient Kinetic Resolution of Sulfur-Stereogenic Sulfoximines by Exploiting CpXRhIII-Catalyzed C-H Functionalization. Angew. Chem., Int. Ed. 2019, 58, 8902– 8906, DOI: 10.1002/anie.201904543Google ScholarThere is no corresponding record for this reference.
- 12Xiong, Y.; Suleman, M.; Xu, S.; Chen, Z. Chiral CpxRhIII-Catalyzed Enantioselective C-H Annulation to Access Fused Tricyclic Sulfur-Stereogenic and Medium-Sized Aza-Heterocycles. Org. Chem. Front. 2025, 12, 614– 622, DOI: 10.1039/D4QO01749HGoogle ScholarThere is no corresponding record for this reference.
- 13Song, Q.; Wu, W.; Chen, W.; Gao, H.; Zhou, Z.; Zeng, Z.; Yi, W. α-Chloroketones Enabled Rh(III)-Catalyzed Enantioselective C-H [4 + 2] Annulation of Sulfoximines under Mild and Redox-Neutral Conditions. Green Synth. Catal. 2025, 6, 320– 323, DOI: 10.1016/j.gresc.2024.05.004Google ScholarThere is no corresponding record for this reference.
- 14(a) Wang, L.; Zhong, J.; Lin, X. Atroposelective Phosphoric Acid Catalyzed Three–Component Cascade Reaction: Enantioselective Synthesis of Axially Chiral N–Arylindoles. Angew. Chem., Int. Ed. 2019, 58, 15824– 15828, DOI: 10.1002/anie.201909855Google ScholarThere is no corresponding record for this reference.(b) Lin, X.; Wang, L.; Han, Z.; Chen, Z. Chiral Spirocyclic Phosphoric Acids and Their Growing Applications. Chin. J. Chem. 2021, 39, 802– 824, DOI: 10.1002/cjoc.202000446Google ScholarThere is no corresponding record for this reference.
- 15Zhong, J.; Pan, R.; Lin, X. Enantioselective Synthesis of α-Tetrasubstituted (1-Indolizinyl) (Diaryl)-Methanamines Via Chiral Phosphoric Acid Catalysis. RSC Adv. 2024, 14, 1106– 1113, DOI: 10.1039/D3RA07636AGoogle ScholarThere is no corresponding record for this reference.
- 16Yu, D.; de Azambuja, F.; Glorius, F. α-MsO/TsO/Cl Ketones as Oxidized Alkyne Equivalents: Redox-Neutral Rhodium(III)-Catalyzed C-H Activation for the Synthesis of N-Heterocycles. Angew. Chem., Int. Ed. 2014, 53, 2754– 2758, DOI: 10.1002/anie.201310272Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
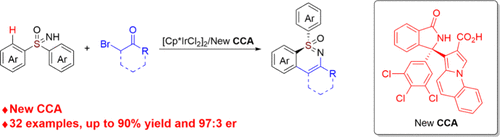
Scheme 1
 Scheme 1. Representative CCAs for Asymmetric C–H Activation
Scheme 1. Representative CCAs for Asymmetric C–H ActivationScheme 2
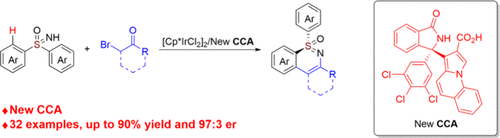 Scheme 2. New CCA Assisted Asymmetric C–H Activation (This Work)
Scheme 2. New CCA Assisted Asymmetric C–H Activation (This Work)Scheme 3
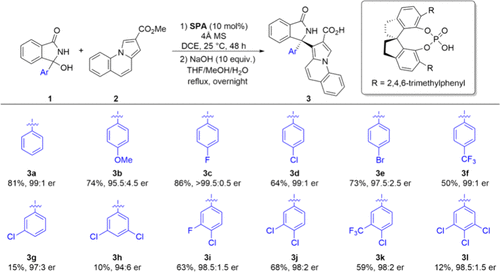 Scheme 3. Asymmetric Synthesis of CCA 3
Scheme 3. Asymmetric Synthesis of CCA 3Scheme 4
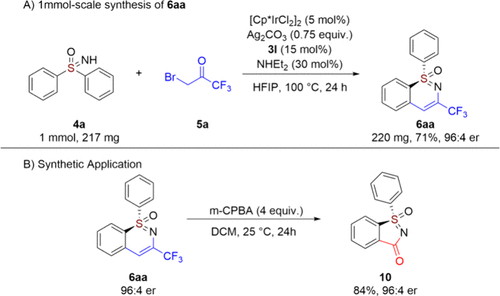 Scheme 4. Scale-up Synthesis and Synthetic Application
Scheme 4. Scale-up Synthesis and Synthetic ApplicationScheme 5
 Scheme 5. Mechanistic Studies
Scheme 5. Mechanistic StudiesReferences
This article references 16 other publications.
- 1(a) Newton, C. G.; Wang, S.-G.; Oliveira, C. C.; Cramer, N. Catalytic Enantioselective Transformations Involving C-H Bond Cleavage by Transition-Metal Complexes. Chem. Rev. 2017, 117, 8908– 8976, DOI: 10.1021/acs.chemrev.6b00692There is no corresponding record for this reference.(b) Saint-Denis, T. G.; Zhu, R.-Y.; Chen, G.; Wu, Q.-F.; Yu, J.-Q. Enantioselective C(Sp3)-H Bond Activation by Chiral Transition Metal Catalysts. Science 2018, 359, eaao4798 DOI: 10.1126/science.aao4798There is no corresponding record for this reference.(c) Loup, J.; Dhawa, U.; Pesciaioli, F.; Wencel-Delord, J.; Ackermann, L. Enantioselective C-H Activation with Earth-Abundant 3d Transition Metals. Angew. Chem., Int. Ed. 2019, 58, 12803– 12818, DOI: 10.1002/anie.201904214There is no corresponding record for this reference.(d) Yoshino, T.; Satake, S.; Matsunaga, S. Diverse Approaches for Enantioselective C-H Functionalization Reactions Using Group 9 CpxMIII Catalysts. Chem. - Eur. J. 2020, 26, 7346– 7357, DOI: 10.1002/chem.201905417There is no corresponding record for this reference.(e) Shi, B.-F.; Zhang, Y.-H.; Lam, J. K.; Wang, D.-H.; Yu, J.-Q. Pd(II)-Catalyzed Enantioselective C–H Olefination of Diphenylacetic Acids. J. Am. Chem. Soc. 2010, 132, 460– 461, DOI: 10.1021/ja909571zThere is no corresponding record for this reference.(f) Cheng, X.-F.; Li, Y.; Su, Y.-M.; Yin, F.; Wang, J.-Y.; Sheng, J.; Vora, H. U.; Wang, X.-S.; Yu, J.-Q. Pd(II)-Catalyzed Enantioselective C–H Activation/C–O Bond Formation: Synthesis of Chiral Benzofuranones. J. Am. Chem. Soc. 2013, 135, 1236– 1239, DOI: 10.1021/ja311259xThere is no corresponding record for this reference.
- 2(a) Wozniak, Łu.; Tan, J.-F.; Nguyen, Q.-H.; Madron du Vigne, A.; Smal, V.; Cao, Y.-X.; Cramer, N. Catalytic Enantioselective Functionalizations of C-H Bonds by Chiral Iridium Complexes. Chem. Rev. 2020, 120, 10516– 10543, DOI: 10.1021/acs.chemrev.0c00559There is no corresponding record for this reference.(b) Yoshino, T.; Matsunaga, S. Chiral Carboxylic Acid Assisted Enantioselective C-H Activation with Achiral CpxMIII (M = Co, Rh, Ir) Catalysts. ACS Catal. 2021, 11, 6455– 6466, DOI: 10.1021/acscatal.1c01351There is no corresponding record for this reference.(c) Liang, H.; Wang, J. Enantioselective C-H Bond Functionalization Involving Arene Ruthenium(II) Catalysis. Chem. - Eur. J. 2023, 29, e202202461 DOI: 10.1002/chem.202202461There is no corresponding record for this reference.(d) Liu, C.-X.; Yin, S.-Y.; Zhao, F.; Yang, H.; Feng, Z.; Gu, Q.; You, S.-L. Rhodium-Catalyzed Asymmetric C-H Functionalization Reactions. Chem. Rev. 2023, 123, 10079– 10134, DOI: 10.1021/acs.chemrev.3c00149There is no corresponding record for this reference.(e) Yao, Q.-J.; Shi, B.-F. Cobalt(III)-Catalyzed Enantioselective C-H Functionalization: Ligand Innovation and Reaction Development. Acc. Chem. Res. 2025, 58, 971– 990, DOI: 10.1021/acs.accounts.5c00013There is no corresponding record for this reference.
- 3Yoshino, T.; Matsunaga, S. Cp*CoIII-Catalyzed C-H Functionalization and Asymmetric Reactions Using External Chiral Sources. Synlett 2019, 30, 1384– 1400, DOI: 10.1055/s-0037-1611814There is no corresponding record for this reference.
- 4Mas-Roselló, J.; Herraiz, A. G.; Audic, B.; Laverny, A.; Cramer, N. Chiral Cyclopentadienyl Ligands: Design, Syntheses, and Applications in Asymmetric Catalysis. Angew. Chem., Int. Ed. 2021, 60, 13198– 13224, DOI: 10.1002/anie.202008166There is no corresponding record for this reference.
- 5(a) Liang, H.; Guo, W.; Li, J.; Jiang, J.; Wang, J. Chiral Arene Ligand as Stereocontroller for Asymmetric C-H Activation. Angew. Chem., Int. Ed. 2022, 61, e202204926 DOI: 10.1002/anie.202204926There is no corresponding record for this reference.(b) Li, J.; Wang, G.; Guo, W.; Jiang, J.; Wang, J. H8-BINOL-Derived Chiral η6-Benzene Ligands: New Opportunities for the Ruthenium-Catalyzed Asymmetric C-H Activation. Angew. Chem., Int. Ed. 2024, 63, e202405782 DOI: 10.1002/anie.202405782There is no corresponding record for this reference.(c) Liu, H.; Jiang, J.-J.; Wang, J. Catalytic Asymmetric C-H Activation/Cyclization of Sulfoximines with Sulfoxonium Ylides by a Chiral η6-Benzene Ruthenium(II) Catalyst. ACS Catal. 2024, 14, 17398– 17404, DOI: 10.1021/acscatal.4c04798There is no corresponding record for this reference.
- 6(a) Pesciaioli, F.; Dhawa, U.; Oliveira, J. C. A.; Yin, R.; John, M.; Ackermann, L. Enantioselective Cobalt(III)-Catalyzed C-H Activation Enabled by Chiral Carboxylic Acid Cooperation. Angew. Chem., Int. Ed. 2018, 57, 15425– 15429, DOI: 10.1002/anie.201808595There is no corresponding record for this reference.(b) Li, Y.; Liou, Y.; Oliveira, J. C. A.; Ackermann, L. Ruthenium(II)/Imidazolidine Carboxylic Acid-Catalyzed C-H Alkylation for Central and Axial Double Enantio-Induction. Angew. Chem., Int. Ed. 2022, 61, e202212595 DOI: 10.1002/anie.202212595There is no corresponding record for this reference.(c) Zhang, Z.-J.; Li, S.-W.; Oliveira, J. C. A.; Li, Y.; Chen, X.; Zhang, S.-Q.; Xu, L.-C.; Rogge, T.; Hong, X.; Ackermann, L. Data-Driven Design of New Chiral Carboxylic Acid for Construction of Indoles with C-Central and C–N Axial Chirality via Cobalt Catalysis. Nat. Commun. 2023, 14, 3149, DOI: 10.1038/s41467-023-38872-0There is no corresponding record for this reference.
- 7(a) Lin, L.; Fukagawa, S.; Sekine, D.; Tomita, E.; Yoshino, T.; Matsunaga, S. Chiral Carboxylic Acid Enabled Achiral Rhodium(III)-Catalyzed Enantioselective C-H Functionalization. Angew. Chem., Int. Ed. 2018, 57, 12048– 12052, DOI: 10.1002/anie.201807610There is no corresponding record for this reference.(b) Fukagawa, S.; Kato, Y.; Tanaka, R.; Kojima, M.; Yoshino, T.; Matsunaga, S. Enantioselective C(Sp3)-H Amidation of Thioamides Catalyzed by a CobaltIII/Chiral Carboxylic Acid Hybrid System. Angew. Chem., Int. Ed. 2019, 58, 1153– 1157, DOI: 10.1002/anie.201812215There is no corresponding record for this reference.(c) Sekine, D.; Ikeda, K.; Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S. Chiral 2-Aryl Ferrocene Carboxylic Acids for the Catalytic Asymmetric C(Sp3)-H Activation of Thioamides. Organometallics 2019, 38, 3921– 3926, DOI: 10.1021/acs.organomet.9b00407There is no corresponding record for this reference.(d) Fukagawa, S.; Kojima, M.; Yoshino, T.; Matsunaga, S. Catalytic Enantioselective Methylene C(Sp3)-H Amidation of 8-Alkylquinolines Using a Cp*RhIII/Chiral Carboxylic Acid System. Angew. Chem., Int. Ed. 2019, 58, 18154– 18158, DOI: 10.1002/anie.201911268There is no corresponding record for this reference.(e) Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S. Development of Pseudo-C2-Symmetric Chiral Binaphthyl Monocarboxylic Acids for Enantioselective C(Sp3)-H Functionalization Reactions under Rh(III) Catalysis. ACS Catal. 2021, 11, 4271– 4277, DOI: 10.1021/acscatal.1c00765There is no corresponding record for this reference.(f) Yoshino, T.; Matsunaga, S.; Huang, L.-T.; Hirata, Y.; Kato, Y.; Lin, L.; Kojima, M. Ruthenium(II)/Chiral Carboxylic Acid Catalyzed Enantioselective C-H Functionalization of Sulfoximines. Synthesis 2022, 54, 4703– 4710, DOI: 10.1055/a-1588-0072There is no corresponding record for this reference.(g) Hirata, Y.; Sekine, D.; Kato, Y.; Lin, L.; Kojima, M.; Yoshino, T.; Matsunaga, S. Cobalt(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective Synthesis of Benzothiadiazine-1-Oxides via C-H Activation. Angew. Chem., Int. Ed. 2022, 61, e202205341 DOI: 10.1002/anie.202205341There is no corresponding record for this reference.(h) Huang, L.; Kitakawa, Y.; Yamada, K.; Kamiyama, F.; Kojima, M.; Yoshino, T.; Matsunaga, S. Enantioselective Synthesis of 1,2-Benzothiazine 1-Imines via RuII/Chiral Carboxylic Acid-Catalyzed C-H Alkylation/Cyclization. Angew. Chem., Int. Ed. 2023, 62, e202305480 DOI: 10.1002/anie.202305480There is no corresponding record for this reference.
- 8(a) Liu, L.; Song, H.; Liu, Y.-H.; Wu, L.-S.; Shi, B.-F. Achiral CpxIr(III)/Chiral Carboxylic Acid Catalyzed Enantioselective C-H Amidation of Ferrocenes under Mild Conditions. ACS Catal. 2020, 10, 7117– 7122, DOI: 10.1021/acscatal.0c02049There is no corresponding record for this reference.(b) Zhou, T.; Qian, P.-F.; Li, J.-Y.; Zhou, Y.-B.; Li, H.-C.; Chen, H.-Y.; Shi, B.-F. Efficient Synthesis of Sulfur-Stereogenic Sulfoximines via Ru(II)-Catalyzed Enantioselective C–H Functionalization Enabled by Chiral Carboxylic Acid. J. Am. Chem. Soc. 2021, 143, 6810– 6816, DOI: 10.1021/jacs.1c03111There is no corresponding record for this reference.(c) Liu, Y.-H.; Xie, P.-P.; Liu, L.; Fan, J.; Zhang, Z.-Z.; Hong, X.; Shi, B.-F. Cp*Co(III)-Catalyzed Enantioselective Hydroarylation of Unactivated Terminal Alkenes via C–H Activation. J. Am. Chem. Soc. 2021, 143, 19112– 19120, DOI: 10.1021/jacs.1c08562There is no corresponding record for this reference.(d) Li, J.-Y.; Xie, P.-P.; Zhou, T.; Qian, P.-F.; Zhou, Y.-B.; Li, H.-C.; Hong, X.; Shi, B.-F. Ir(III)-Catalyzed Asymmetric C–H Activation/Annulation of Sulfoximines Assisted by the Hydrogen-Bonding Interaction. ACS Catal. 2022, 12, 9083– 9091, DOI: 10.1021/acscatal.2c00962There is no corresponding record for this reference.(e) Zhou, Y.-B.; Zhou, T.; Qian, P.-F.; Li, J.-Y.; Shi, B.-F. Synthesis of Sulfur-Stereogenic Sulfoximines via Co(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Amidation. ACS Catal. 2022, 12, 9806– 9811, DOI: 10.1021/acscatal.2c02691There is no corresponding record for this reference.(f) Qian, P.-F.; Zhou, T.; Li, J.-Y.; Zhou, Y.-B.; Shi, B.-F. Ru(II)/Chiral Carboxylic Acid-Catalyzed Asymmetric [4 + 3] Annulation of Sulfoximines with α,β-Unsaturated Ketones. ACS Catal. 2022, 12, 13876– 13883, DOI: 10.1021/acscatal.2c03531There is no corresponding record for this reference.(g) Zhou, G.; Zhou, T.; Jiang, A.; Qian, P.; Li, J.; Jiang, B.; Chen, Z.; Shi, B. Electrooxidative Rhodium(III)/Chiral Carboxylic Acid-Catalyzed Enantioselective C–H Annulation of Sulfoximines with Alkynes. Angew. Chem., Int. Ed. 2024, 63, e202319871 DOI: 10.1002/anie.202319871There is no corresponding record for this reference.
- 9Liu, W.; Yang, W.; Zhu, J.; Guo, Y.; Wang, N.; Ke, J.; Yu, P.; He, C. Dual-Ligand-Enabled Ir(III)-Catalyzed Enantioselective C–H Amidation for the Synthesis of Chiral Sulfoxides. ACS Catal. 2020, 10, 7207– 7215, DOI: 10.1021/acscatal.0c02109There is no corresponding record for this reference.
- 10(a) Lücking, U. Sulfoximines: A Neglected Opportunity in Medicinal Chemistry. Angew. Chem., Int. Ed. 2013, 52, 9399– 9408, DOI: 10.1002/anie.201302209There is no corresponding record for this reference.(b) Mäder, P.; Kattner, L. Sulfoximines as Rising Stars in Modern Drug Discovery? Current Status and Perspective on an Emerging Functional Group in Medicinal Chemistry. J. Med. Chem. 2020, 63, 14243– 14275, DOI: 10.1021/acs.jmedchem.0c00960There is no corresponding record for this reference.
- 11(a) Sun, Y.; Cramer, N. Enantioselective Synthesis of Chiral-At-Sulfur 1,2-Benzothiazines by CpxRhIII-Catalyzed C-H Functionalization of Sulfoximines. Angew. Chem., Int. Ed. 2018, 57, 15539– 15543, DOI: 10.1002/anie.201810887There is no corresponding record for this reference.(b) Shen, B.; Wan, B.; Li, X. Enantiodivergent Desymmetrization in the Rhodium(III)-Catalyzed Annulation of Sulfoximines with Diazo Compounds. Angew. Chem., Int. Ed. 2018, 57, 15534– 15538, DOI: 10.1002/anie.201810472There is no corresponding record for this reference.(c) Brauns, M.; Cramer, N. Efficient Kinetic Resolution of Sulfur-Stereogenic Sulfoximines by Exploiting CpXRhIII-Catalyzed C-H Functionalization. Angew. Chem., Int. Ed. 2019, 58, 8902– 8906, DOI: 10.1002/anie.201904543There is no corresponding record for this reference.
- 12Xiong, Y.; Suleman, M.; Xu, S.; Chen, Z. Chiral CpxRhIII-Catalyzed Enantioselective C-H Annulation to Access Fused Tricyclic Sulfur-Stereogenic and Medium-Sized Aza-Heterocycles. Org. Chem. Front. 2025, 12, 614– 622, DOI: 10.1039/D4QO01749HThere is no corresponding record for this reference.
- 13Song, Q.; Wu, W.; Chen, W.; Gao, H.; Zhou, Z.; Zeng, Z.; Yi, W. α-Chloroketones Enabled Rh(III)-Catalyzed Enantioselective C-H [4 + 2] Annulation of Sulfoximines under Mild and Redox-Neutral Conditions. Green Synth. Catal. 2025, 6, 320– 323, DOI: 10.1016/j.gresc.2024.05.004There is no corresponding record for this reference.
- 14(a) Wang, L.; Zhong, J.; Lin, X. Atroposelective Phosphoric Acid Catalyzed Three–Component Cascade Reaction: Enantioselective Synthesis of Axially Chiral N–Arylindoles. Angew. Chem., Int. Ed. 2019, 58, 15824– 15828, DOI: 10.1002/anie.201909855There is no corresponding record for this reference.(b) Lin, X.; Wang, L.; Han, Z.; Chen, Z. Chiral Spirocyclic Phosphoric Acids and Their Growing Applications. Chin. J. Chem. 2021, 39, 802– 824, DOI: 10.1002/cjoc.202000446There is no corresponding record for this reference.
- 15Zhong, J.; Pan, R.; Lin, X. Enantioselective Synthesis of α-Tetrasubstituted (1-Indolizinyl) (Diaryl)-Methanamines Via Chiral Phosphoric Acid Catalysis. RSC Adv. 2024, 14, 1106– 1113, DOI: 10.1039/D3RA07636AThere is no corresponding record for this reference.
- 16Yu, D.; de Azambuja, F.; Glorius, F. α-MsO/TsO/Cl Ketones as Oxidized Alkyne Equivalents: Redox-Neutral Rhodium(III)-Catalyzed C-H Activation for the Synthesis of N-Heterocycles. Angew. Chem., Int. Ed. 2014, 53, 2754– 2758, DOI: 10.1002/anie.201310272There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.5c04553.
Experimental procedures, Characterization details, Mechanic studies, Crystallographic data for compound 3l (CCDC 2498768) and 6xa (CCDC 2484114), NMR and HPLC spectra. (PDF)
Deposition Numbers 2484114 and 2498768 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



