Deaminative Ring Contraction for the Modular Synthesis of Pyrido[n]helicenesClick to copy article linkArticle link copied!
- Zachary T. SchwartzZachary T. SchwartzDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Zachary T. Schwartz
- Chelsea D. ValitonChelsea D. ValitonDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Chelsea D. Valiton
- Myles A. LovaszMyles A. LovaszDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Myles A. Lovasz
- Farah SadatFarah SadatDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Farah Sadat
- Michelle PhanMichelle PhanDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Michelle Phan
- Ryan T. VanderLindenRyan T. VanderLindenDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Ryan T. VanderLinden
- Thomas G. RichmondThomas G. RichmondDepartment of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Thomas G. Richmond
- Andrew G. Roberts*Andrew G. Roberts*Email: [email protected]Department of Chemistry, The University of Utah, Salt Lake City, Utah 84112, United States of AmericaMore by Andrew G. Roberts
Abstract
A modular strategy involving tertiary amines as templates to build multiple carbon–carbon bonds via reductive cyclization and deaminative contraction steps enables the scalable synthesis of (di)aza[5]helicenes and (di)aza[6]helicenes. The methods permit the rapid and gram-scale assembly of key pyridyl-containing dihydroazepines that are advanced using a developed deaminative contraction to access the N-atom positional isomers of pyrido[5] and [6]helicenes. A telescoped synthesis demonstrates expedited access to (±)-1-aza[6]helicene, a resolvable helicene valued for its circularly polarized luminescence properties.
This publication is licensed for personal use by The American Chemical Society.
Helicenes are a fascinating class of polycyclic aromatics, aptly named after the chiral helicity (handedness) that results when ortho-condensed aromatic systems are extended beyond three consecutive rings. (1) Pyrido[n]helicenes, like 1 (n = 5) and 2 (n = 6), are of particular importance as their pyridyl moieties can be regioselectively functionalized and the pyridyl group can be leveraged for the scalable resolution of helical enantiomers (Figure 1A). (2,3) Due to its relative structural simplicity, room temperature configurational stability, and pH-dependent chiroptical properties, 1-aza[6]helicene 2a (3−5) has emerged as a promising chiral small molecule additive for the design of novel optical and electronic materials. (6−9) Additionally, 2a and its N-oxide derivative (not shown) are useful in asymmetric catalysis. (4,10) However, even the prototypical 1-aza[6]helicene, 2a, is not commercially available and can be challenging to prepare─requiring the multistep conversion of advanced building blocks and an intermediary resolution step (3) if a single enantiomer is desired (see 2a, (P)-isomer shown). (3−5)
Figure 1
Figure 1. Pyrido[n]helicenes: Structure, properties, and synthetic strategy. (A) Pyridyl-containing [5] and [6]helicenes. (B) Proposed modular assembly of pyridyl-containing [5] and [6]helicenes from tertiary amines via a reductive cyclization and deaminative contraction sequence.
While developed strategies have recognized the C and D rings as convergent assembly points (Figure 1A, shown in orange for 1 and 2), modular access to #-aza (e.g., # = 1-, 2-, 3-, 4-, 5-, or 6-) and #,#′-diaza variants (e.g., #,#′ = 1,14- or 1,16-) that could be useful in tuning materials and catalysis applications is underdeveloped. The general strategies for the synthesis of pyrido[n]helicenes (3−5,11−14) are limited because the incorporation of a single heteroatom (e.g., N) can complicate isomer outcome during alkene-forming events (i.e., intermediary E- and Z-alkene isomers) (4,11) and ring-forming events (i.e., two or more isomers possible). (12,13) Direct access to chiral pyrido[n]helicenes is further challenged (14,15) by the added Lewis basic site(s) that can compromise the catalytic efficiency and asymmetry of involved bond-forming steps. (5) Recently, Yang and co-workers developed a route to chiral 4-aza[4] and 4-aza[5]helicenes using an asymmetric multicomponent condensation reaction to build the pyridyl ring last; however, access to other N-atom positional isomers is limited. (15)
Motivated by the potential value of N-atom positional isomers, we envisioned the development of a process to convert simple-to-prepare prepare tertiary amines, 4 and 5, into 1-aza[5]helicenes (1a) and 1-aza[6]helicenes (2a), respectively (Figure 1B). Reductive cyclization (16,17) would regioselectively conjugate the amine-templated ring system (e.g., AB + DE (4), ABC + E (5), and ABC + EF (5)), and a subsequent deaminative contraction process would forge the C or D ring (shown in orange). The realization of these exemplars could motivate general and modular access to #-aza[5]helicenes (1) and #-aza[6]helicenes (2) from appropriate tertiary amine templates. Furthermore, the sequence has the potential to be telescoped, without the purification of intermediates, which, when coupled with a simple resolution protocol, would drastically simplify access to chiral pyridyl-containing helicenes.
In 1985, Maigrot and Mazaleyrat showed that the double alkylation of l-(−)-ephedrine 6 with (±)-7 formed separable chiral azepinium diastereomers that could be transformed into chiral 1,1′-binaphthyl derivatives (Figure 2). (18) Inspired by this, Závada and co-workers realized that the chiral auxiliary component could be removed via epoxide (8) formation (19) and, thus, advanced chiral azepinium, (S)-(+)-9, transformed into chiral [5]helicene (P)-(+)-10 via a [1,2]-Stevens rearrangement, dehydroamination cascade reaction (confer ‘Stevens intermediate’ 11). (20) This singular demonstration, where (±)-7 derives from the homocoupling of 1-bromo-2-methyl naphthalene in two steps, (18) further motivates the need for a more direct and asymmetric assembly strategy to prepare biaryl-linked dihydroazepines that can be advanced to pyrido[5] and [6]helicenes.
Figure 2
Figure 2. Chiral [5]helicene from a chiral azepinium. The use of a chiral amine (6) to relay stereochemical information toward chiral [5]helicene, (P)-(+)-10.
We began our studies by examining the scope of tertiary amines that would be transformed into a variety of representative (di)aza[5] (1) and (di)aza[6]helicenes (2) in two steps (Figure 1B). While the synthesis of some targeted (di)aza[5] and (di)aza[6]helicenes are reported, (3−5,11,13) the strategies for their assembly are disparate and of varied yield. The required tertiary amines (12) were prepared in moderate to high isolated yields by alkylating benzylic secondary amines with benzylic bromides (see the Supporting Information). (21,22) The ortho-bromo-bearing tertiary amines (12) are readily cyclized into biaryl-linked dihydroazepines (13) using Ni-promoted conditions (Figure 3A). (21−25) Alternatively, related tertiary amines can be prepared using reductive amination methods. (21−23) The C*-ring-forming cyclizations proceed with moderate to high efficiency to provide 13a–13i in 54–84% yields, except for 13d, obtained in 21% yield. We tentatively attribute this lower yield to the ability of 12d and its reduced intermediates to chelate the Ni catalyst nonproductively. (22,26) Using a similar sequence, the D*-ring-forming cyclizations proceed with moderate to high efficiency to provide 13j–13n in 33–84% yields.
Figure 3
Figure 3. Pyrido[n]helicenes: Structure, properties, and synthetic strategy. (A) Reductive cyclization: modular synthesis of N-methyl biaryl-linked dihydrozepines from simple to prepare tertiary amines. (B) Deaminative contraction: modular synthesis of pyridyl-containing [5]helicenes and [6]helicenes from N-methyl biaryl-linked dihydroazepines. aAg+ and CF3SO2– (trifluoromethanesulfonate) counterions have been omitted for clarity (see the Supporting Information).
With biaryl-linked dihydroazepine intermediates (13) in hand, we show that these stable intermediates can undergo mild, pyridyl-tolerant deaminative contractions to give (di)aza[5] (1) and (di)aza[6]helicenes (2) (Figure 3B). Under optimized conditions using trimethyl phosphate, (21,22,27,28) the C-ring-forming contractions proceed with moderate to high efficiency, providing 1a–1i in 50–89% yields. Optimization with 13a to form 1a found 18-crown-6 equivalency and anhydrous THF (<20 ppm of H2O) to be critical factors for the high conversion and reproducible isolation of 1a (CSD 2402615, for substrates with observable side products, reducing the reaction exposure time can improve the isolated yields; see the Supporting Information). Similarly, the D-ring-forming contractions proceed with moderate efficiency, providing 1d, 1k–1l, and 2a–2b, in 31–67% yields. Having optimized the cyclization and deaminative contraction conditions for #-aza[n]helicenes, we investigated the scalability of the developed methods and pursued the synthesis of novel C-ring-alkylated 1-aza[n]helicenes. We targeted 1-aza[6]helicene 2a to demonstrate synthesis on scale due to its configurational stability (3) and established use in optical devices. (6−9) The gram-scale synthesis of 2a proceeds smoothly (see the Supporting Information for minor procedural deviations). Starting with 4 g of 12m (7.7 mmol), its reductive cyclization produces 13m in 72% isolated yield (1.99 g, 5.5 mmol). Next, we converted 13m (1.94 g, 5.4 mmol) into more than 1.5 g of (±)-1-aza[6]helicene 2a─an 85% isolated yield (1.52 g, 4.6 mmol). We attribute the improved yields to the larger scale, which may benefit from less adventitious water.
With the successful assembly of pyridyl-containing [5] (1) and [6]helicenes (2) (Figure 3), we considered if the tertiary amine substrate could bear an α-alkyl substituent that could then be translated into a C7/C8-alkylated 1-aza[5]helicene variant (Figure 4A). We envision that C-ring-alkylated 1-aza[5]helicenes derivatives can be designed or functionalized (e.g., radical halogenation) to improve their properties, enable covalent conjugation handles, and motivate novel applications in materials science. By design, the alkyl group can be incorporated at either the C7- or C8-position of the target (confer 1o). To evaluate this, we prepared C8-α-methyl tertiary amine 12o (see the Supporting Information), which undergoes reductive cyclization to yield α-methyl biaryl-linked azepine 13o in 53% isolated yield as an inconsequential mixture of diastereomers (d.r. 1:1, determined by integration of the 1H NMR spectrum). Subjecting the diastereomers (13o) to the standard deaminative contraction conditions gives 1-aza-8-methyl[5]helicene 1o (25%) alongside vinyl-bearing naphthyl derivative, 14 (see the Supporting Information), which likely arises via a competitive Hofmann elimination pathway. (29) Efforts to optimize the desired [1,2]-Stevens, dehydroamination pathway toward 1o and with other C7/C8-alkylated variants are ongoing.
Figure 4
Figure 4. Pyrido[n]helicene modification strategies and scalable access to chiral 1-aza[6]helicene. (A) Synthesis of a C-ring alkylated 1-aza[5]helicene. Conditions: aNiBr2(PPh3)2, Et4NI, Zn, THF, 50 °C; bLiI, PO(OMe)3, and THF, 65 °C; t-BuOK, 18-crown-6, and THF, 65 °C. (B) Modular and streamlined syntheses of 1-aza[6]helicene. Conditions: aNiBr2(PPh3)2, Et4NI, Zn, THF, 50 °C; bLiI, PO(OMe)3, THF, 65 °C; t-BuOK, 18-crown-6, THF, 65 °C; cNBS, Pd(OAc)2, CH3CN; dNBS, (BzO)2, PhH; eadd 17, THF; fsee ref (3) for details; g0.5 equiv of AgOTf; htrifluoromethanesulfonate (CF3SO2–) counterion and THF solvate omitted for clarity. (C) Summarized access to [4], [5], and [6]helicenes.
Next, we set out to show that a designed aza[6]helicene can be efficiently prepared on scale by two iterations of a cyclize–contract sequence (Figure 4B). Advancing our previously developed two-step method, where biaryl-linked dihydroazepines were isolated, we streamlined the synthesis of the key 9-methyl-benzo[h]quinoline 15 building block from tertiary amine 16, itself available on multigram scale in two steps from commercial reagents. (22) The purification of biaryl-linked dihydroazepines using column chromatography on preparative scales can be challenging due to their highly polar tertiary amine moieties that tend to streak on SiO2 and coelute with triphenylphosphine oxide. To mitigate this, the cyclize–contract events (steps 3a and 3b) can be performed in series, without the purification of intermediates, to form 15 in 67% isolated yield from 16. This overall efficiency compares favorably to that of the two-step process. Next, 15 is brominated at C10 (step 4) (30) and then again at the C9 methyl (step 5), and the resultant product reacts with secondary amine 17 (step 6) to afford tertiary amine 12m in 44% yield over three steps. (4,22) Encouragingly, the same cyclize–contract events (steps 7a and 7b) proceed to form the target 1-aza[6]helicene 2a in 31% yield on a 1.9 mmol scale. Here, the overall efficiency is near that of the two-step process (step 7a: 67% of isolated 13m; step 7b: 67% of 2a from 13m; calculated 45% over two steps). Efforts to further improve this streamlined process, where the B and D rings are modular assembly points, are ongoing. Finally, (±)-2a can be readily resolved by leveraging the differential solubility of its dibenzoyl-l-tartrate salts to access (+)-(P)-2a (shown) and (−)-(M)-2a on scale. (3) A dimeric complex (18) suitable for X-ray diffraction forms when (+)-(P)-2a (calculated 92% ee) is treated with 0.5 equiv of silver(I) trifluoromethanesulfonate in THF (Figure 4B, (+)-(P)-2a, CSD 2402616). Overall, the telescoped process is modular, provides access to both enantiomers, and is anticipated to be useful for the design of other room temperature configurationally stable #-aza[6]helicenes, like 2a.
Pyridyl-containing helicenes are interesting molecules on account of their unique chiroptical properties and the ease by which their enantiomers can be resolved, but further exploration of their applications has been thwarted by their limited and challenging syntheses. We have developed a reductive cyclization–deaminative contraction strategy that enables modular access to various pyridyl-containing helicenes (15 demonstrated examples), including access to enantioenriched 1-aza[6]helicenes (2a), showing that the methods can be quickly adapted to synthesize desired analogues, including N-atom positional isomers (1a–1l) and (di)aza[6]helicene variants, 2a and 2b, on preparative scales (Figure 4C). (31) Previous efforts show that 1-aza[4]helicene 19 and [4] and [5]carbohelicenes (e.g., 20 and 21) are also accessible, connecting general access to pyrido- and carbohelicenes. (22) Efforts to modify the prepared parent helicenes and C-ring-alkylated variant (1o) to enable further design granularity are ongoing.
Data Availability
The data underlying this study are available in the published article and its Supporting Information.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.5c04734.
Experimental procedures and characterization data (HRMS and 1H and 13C NMR spectra for the reported synthetic compounds) (PDF)
Deposition Numbers 2402615–2402616 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We are grateful for financial support of this research from the University of Utah and the Donors of the American Chemical Society Petroleum Research Fund (ACS PRF 62220-DNI1). NSF Research Experiences for Undergraduates supported the training and research contributions of F.S. (NSF REU 2150526). This research is based upon work supported by the National Science Foundation under Grant No. 2247651. We thank Prof. Ryan E. Looper, Prof. Jon D. Rainier, Prof. Matthew S. Sigman, Prof. Ming Lee Tang, and Prof. Qilei Zhu for thoughtful discussions and their review of this manuscript. We thank Paulina Jaimes, Alex Malin, and Annie Thompson for experimental assistance. The NMR spectroscopy results included in this report were recorded at the David M. Grant NMR Center, a University of Utah Core Facility. Funds for construction of the Center and the helium recovery system were obtained from the University of Utah and the National Institutes of Health awards 1C06RR017539-01A1 and 3R01GM063540-17W1, respectively. NMR spectroscopy instruments were purchased with support of the University of Utah and the National Institutes of Health award 1S10OD25241-01. The single crystal X-ray diffraction data was collected on an instrument purchased with support by the National Institutes of Health award S10OD030326.
References
This article references 31 other publications.
- 1Stara, I. G.; Crassous, J.; Stary, I. Helicenes synthesis, properties, and applications; Wiley-VCH, 2023.Google ScholarThere is no corresponding record for this reference.
- 2Chen, J.; Captain, B.; Takenaka, N. Helical Chiral 2,2′-Bipyridine N- Monoxides as Catalysts in the Enantioselective Propargylation of Aldehydes with Allenyltrichlorosilane. Org. Lett. 2011, 13 (7), 1654– 1657, DOI: 10.1021/ol200102cGoogle ScholarThere is no corresponding record for this reference.
- 3Míšek, J.; Teplý, F.; Stará, I. G.; Tichý, M.; Šaman, D.; Císařová, I.; Vojtíšek, P.; Starý, I. A Straightforward Route to Helically Chiral N-Heteroaromatic Compounds: Practical Synthesis of Racemic 1,14-Diaza[5]Helicene and Optically Pure 1- and 2-Aza[6]Helicenes. Angew. Chem., Int. Ed. 2008, 47 (17), 3188– 3191, DOI: 10.1002/anie.200705463Google ScholarThere is no corresponding record for this reference.
- 4Takenaka, N.; Sarangthem, R. S.; Captain, B. Helical Chiral Pyridine N-Oxides: A New Family of Asymmetric Catalysts. Angew. Chem., Int. Ed. 2008, 47 (50), 9708– 9710, DOI: 10.1002/anie.200803338Google ScholarThere is no corresponding record for this reference.
- 5Weimar, M.; Correa da Costa, R.; Lee, F.-H.; Fuchter, M. J. A Scalable and Expedient Route to 1-Aza[6]Helicene Derivatives and Its Subsequent Application to a Chiral-Relay Asymmetric Strategy. Org. Lett. 2013, 15 (7), 1706– 1709, DOI: 10.1021/ol400493xGoogle ScholarThere is no corresponding record for this reference.
- 6Brandt, J. R.; Pospisil, L.; Bednarova, L.; da Costa, R. C.; White, A. J. P.; Mori, T.; Teply, F.; Fuchter, M. J. Intense Redox-Driven Chiroptical Switching with a 580 mV Hysteresis Actuated through Reversible Dimerization of an Azoniahelicene. Chem. Commun. 2017, 53 (65), 9059– 9062, DOI: 10.1039/C7CC04903JGoogle ScholarThere is no corresponding record for this reference.
- 7Yang, Y.; Rice, B.; Shi, X.; Brandt, J. R.; Correa da Costa, R.; Hedley, G. J.; Smilgies, D.-M.; Frost, J. M.; Samuel, I. D. W.; Otero-de-la-Roza, A.; Johnson, E. R.; Jelfs, K. E.; Nelson, J.; Campbell, A. J.; Fuchter, M. J. Emergent Properties of an Organic Semiconductor Driven by Its Molecular Chirality. ACS Nano 2017, 11 (8), 8329– 8338, DOI: 10.1021/acsnano.7b03540Google ScholarThere is no corresponding record for this reference.
- 8Wade, J.; Hilfiker, J. N.; Brandt, J. R.; Liirò-Peluso, L.; Wan, L.; Shi, X.; Salerno, F.; Ryan, S. T. J.; Schöche, S.; Arteaga, O.; Jávorfi, T.; Siligardi, G.; Wang, C.; Amabilino, D. B.; Beton, P. H.; Campbell, A. J.; Fuchter, M. J. Natural Optical Activity as the Origin of the Large Chiroptical Properties in π-Conjugated Polymer Thin Films. Nat. Commun. 2020, 11 (1), 6137, DOI: 10.1038/s41467-020-19951-yGoogle ScholarThere is no corresponding record for this reference.
- 9Crassous, J.; Fuchter, M. J.; Freedman, D. E.; Kotov, N. A.; Moon, J.; Beard, M. C.; Feldmann, S. Materials for Chiral Light Control. Nat. Rev. Mater. 2023, 8 (6), 365– 371, DOI: 10.1038/s41578-023-00543-3Google ScholarThere is no corresponding record for this reference.
- 10Matsumoto, A.; Yonemitsu, K.; Ozaki, H.; Míšek, J.; Starý, I.; Stará, I. G.; Soai, K. Reversal of the Sense of Enantioselectivity between 1- and 2-Aza[6]Helicenes Used as Chiral Inducers of Asymmetric Autocatalysis. Org. Biomol. Chem. 2017, 15 (6), 1321– 1324, DOI: 10.1039/C6OB02745HGoogle ScholarThere is no corresponding record for this reference.
- 11Harrowven, D. C.; Guy, I. L.; Nanson, L. Efficient Phenanthrene, Helicene, and Azahelicene Syntheses. Angew. Chem., Int. Ed. 2006, 45 (14), 2242– 2245, DOI: 10.1002/anie.200504287Google ScholarThere is no corresponding record for this reference.
- 12Kawashima, T.; Matsumoto, Y.; Sato, T.; Yamada, Y. M. A.; Kono, C.; Tsurusaki, A.; Kamikawa, K. Synthesis, Structure, and Complexation of an S-Shaped Double Azahelicene with Inner-Edge Nitrogen Atoms. Chem. – Eur. J. 2020, 26 (58), 13170– 13176, DOI: 10.1002/chem.202002405Google ScholarThere is no corresponding record for this reference.
- 13Abbate, S.; Bazzini, C.; Caronna, T.; Fontana, F.; Gambarotti, C.; Gangemi, F.; Longhi, G.; Mele, A.; Sora, I. N.; Panzeri, W. Monoaza[5]Helicenes. Part 2: Synthesis, Characterisation and Theoretical Calculations. Tetrahedron 2006, 62 (1), 139– 148, DOI: 10.1016/j.tet.2005.09.132Google ScholarThere is no corresponding record for this reference.
- 14Yu, T.; Li, Z.-Q.; Li, J.; Cheng, S.; Xu, J.; Huang, J.; Zhong, Y.-W.; Luo, S.; Zhu, Q. Palladium-Catalyzed Modular Synthesis of Enantioenriched Pyridohelicenes through Double Imidoylative Cyclization. ACS Catal. 2022, 12 (20), 13034– 13041, DOI: 10.1021/acscatal.2c04461Google ScholarThere is no corresponding record for this reference.
- 15Liu, W.; Qin, T.; Xie, W.; Zhou, J.; Ye, Z.; Yang, X. Enantioselective Synthesis of Azahelicenes through Organocatalyzed Multicomponent Reactions. Angew. Chem., Int. Ed. 2023, 62 (27), e202303430 DOI: 10.1002/anie.202303430Google ScholarThere is no corresponding record for this reference.
- 16Zhang, S.; Zhang, D.; Liebeskind, L. S. Ambient Temperature, Ullmann-like Reductive Coupling of Aryl, Heteroaryl, and Alkenyl Halides. J. Org. Chem. 1997, 62 (8), 2312– 2313, DOI: 10.1021/jo9700078Google ScholarThere is no corresponding record for this reference.
- 17Xue, W.; Xu, H.; Liang, Z.; Qian, Q.; Gong, H. Nickel-Catalyzed Reductive Cyclization of Alkyl Dihalides. Org. Lett. 2014, 16 (19), 4984– 4987, DOI: 10.1021/ol502207zGoogle ScholarThere is no corresponding record for this reference.
- 18Maigrot, N.; Mazaleyrat, J.-P. New and Improved Synthesis of Optically Pure (R)-and (S)-2,2′-Dimethyl-1, 1′-Binaphthyl and Related Compounds. Synthesis 1985, 1985, 317– 320, DOI: 10.1055/s-1985-31196Google ScholarThere is no corresponding record for this reference.
- 19Stará, I. G.; Starý, I.; Závada, J. Optically Pure (S)- and (R)-4,5-Dihydro-3H-4-Methyldinaphth[2,1-c; 1′,2′-e]Azepines. Application to the Synthesis of New Bidentate Ligands with Axial Asymmetry. Tetrahedron Asymmetry 1992, 3 (11), 1365– 1368, DOI: 10.1016/0957-4166(92)80007-JGoogle ScholarThere is no corresponding record for this reference.
- 20Stara, I. G.; Stary, I.; Tichy, M.; Zavada, J.; Hanus, V. Stereochemical Dichotomy in the Stevens Rearrangement of Axially Twisted Dihydroazepinium and Dihydrothiepinium Salts. A Novel Enantioselective Synthesis of Pentahelicene. J. Am. Chem. Soc. 1994, 116 (12), 5084– 5088, DOI: 10.1021/ja00091a009Google ScholarThere is no corresponding record for this reference.
- 21McFadden, T. P.; Nwachukwu, C. I.; Roberts, A. G. An Amine Template Strategy to Construct Successive C–C Bonds: Synthesis of Benzo[h]quinolines by a Deaminative Ring Contraction Cascade. Org. Biomol. Chem. 2022, 20 (7), 1379– 1385, DOI: 10.1039/D1OB02245HGoogle ScholarThere is no corresponding record for this reference.
- 22Kirkeby, E. K.; Schwartz, Z. T.; Lovasz, M. A.; Roberts, A. G. Deaminative Ring Contraction for the Synthesis of Polycyclic Heteroaromatics: A Concise Total Synthesis of Toddaquinoline. Chem. Sci. 2023, 14, 10508– 10514, DOI: 10.1039/D3SC03936FGoogle ScholarThere is no corresponding record for this reference.
- 23Schwartz, Z.; Valiton, C.; Lovasz, M.; Roberts, A. G. Recent Applications of Ammonium Ylide Based [2,3]-Sigmatropic and [1,2]-Stevens Rearrangements to Transform Amines into Natural Products. Synthesis 2024, 56, 87, DOI: 10.1055/s-0042-1751446Google ScholarThere is no corresponding record for this reference.
- 24Iyoda, M.; Otsuka, H.; Sato, K.; Nisato, N.; Oda, M. Homocoupling of Aryl Halides Using Nickel(II) Complex and Zinc in the Presence of Et4NI. An Efficient Method for the Synthesis of Biaryls and Bipyridines. Bull. Chem. Soc. Jpn. 1990, 63 (1), 80– 87, DOI: 10.1246/bcsj.63.80Google ScholarThere is no corresponding record for this reference.
- 25Leleu, S.; Papamicaël, C.; Marsais, F.; Dupas, G.; Levacher, V. Preparation of Axially Chiral Quinolinium Salts Related to NAD+ Models: New Investigations of These Biomimetic Models as ‘Chiral Amide-Transferring Agents. Tetrahedron Asymmetry 2004, 15 (24), 3919– 3928, DOI: 10.1016/j.tetasy.2004.11.004Google ScholarThere is no corresponding record for this reference.
- 26Na, H.; Mirica, L. M. Deciphering the Mechanism of the Ni-Photocatalyzed C–O Cross-Coupling Reaction Using a Tridentate Pyridinophane Ligand. Nat. Commun. 2022, 13 (1), 1313, DOI: 10.1038/s41467-022-28948-8Google ScholarThere is no corresponding record for this reference.
- 27He, Z.-T.; Li, H.; Haydl, A. M.; Whiteker, G. T.; Hartwig, J. F. Trimethylphosphate as a Methylating Agent for Cross Coupling: A Slow-Release Mechanism for the Methylation of Arylboronic Esters. J. Am. Chem. Soc. 2018, 140 (49), 17197– 17202, DOI: 10.1021/jacs.8b10076Google ScholarThere is no corresponding record for this reference.
- 28Nwachukwu, C. I.; McFadden, T. P.; Roberts, A. G. Ni-Catalyzed Iterative Alkyl Transfer from Nitrogen Enabled by the In Situ Methylation of Tertiary Amines. J. Org. Chem. 2020, 85 (15), 9979– 9992, DOI: 10.1021/acs.joc.0c01274Google ScholarThere is no corresponding record for this reference.
- 29von Hofmann, A. W. Beiträge Zur Kenntniss Der Flüchtigen Organischen Basen. Justus Liebigs Ann. Chem. 1851, 78 (3), 253– 286, DOI: 10.1002/jlac.18510780302Google ScholarThere is no corresponding record for this reference.
- 30Dick, A. R.; Hull, K. L.; Sanford, M. S. A Highly Selective Catalytic Method for the Oxidative Functionalization of C–H Bonds. J. Am. Chem. Soc. 2004, 126 (8), 2300– 2301, DOI: 10.1021/ja031543mGoogle ScholarThere is no corresponding record for this reference.
- 31Schwartz, Z. T.; Valiton, C. D.; Lovasz, M. A.; Sadat, F.; Phan, M.; VanderLinden, R.; Richmond, T.; Roberts, A. G. Deaminative ring contraction for the modular synthesis of pyrido[n]helicenes. ChemRxiv 2024, DOI: 10.26434/chemrxiv-2024-qdd0nGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract

Figure 1
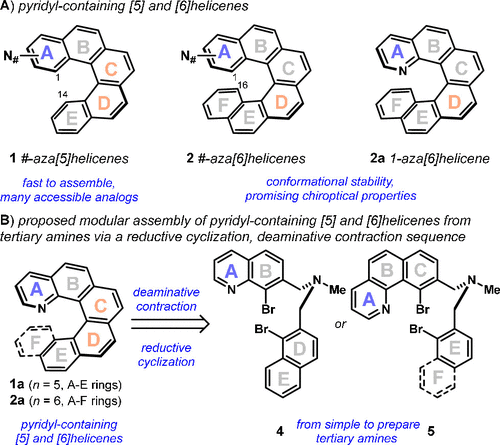
Figure 1. Pyrido[n]helicenes: Structure, properties, and synthetic strategy. (A) Pyridyl-containing [5] and [6]helicenes. (B) Proposed modular assembly of pyridyl-containing [5] and [6]helicenes from tertiary amines via a reductive cyclization and deaminative contraction sequence.
Figure 2
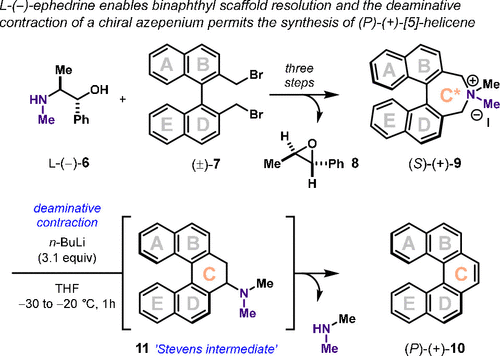
Figure 2. Chiral [5]helicene from a chiral azepinium. The use of a chiral amine (6) to relay stereochemical information toward chiral [5]helicene, (P)-(+)-10.
Figure 3

Figure 3. Pyrido[n]helicenes: Structure, properties, and synthetic strategy. (A) Reductive cyclization: modular synthesis of N-methyl biaryl-linked dihydrozepines from simple to prepare tertiary amines. (B) Deaminative contraction: modular synthesis of pyridyl-containing [5]helicenes and [6]helicenes from N-methyl biaryl-linked dihydroazepines. aAg+ and CF3SO2– (trifluoromethanesulfonate) counterions have been omitted for clarity (see the Supporting Information).
Figure 4
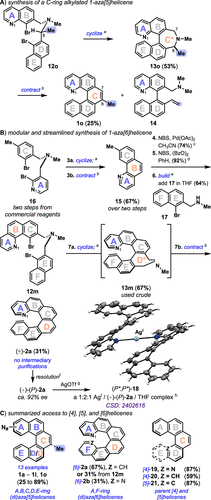
Figure 4. Pyrido[n]helicene modification strategies and scalable access to chiral 1-aza[6]helicene. (A) Synthesis of a C-ring alkylated 1-aza[5]helicene. Conditions: aNiBr2(PPh3)2, Et4NI, Zn, THF, 50 °C; bLiI, PO(OMe)3, and THF, 65 °C; t-BuOK, 18-crown-6, and THF, 65 °C. (B) Modular and streamlined syntheses of 1-aza[6]helicene. Conditions: aNiBr2(PPh3)2, Et4NI, Zn, THF, 50 °C; bLiI, PO(OMe)3, THF, 65 °C; t-BuOK, 18-crown-6, THF, 65 °C; cNBS, Pd(OAc)2, CH3CN; dNBS, (BzO)2, PhH; eadd 17, THF; fsee ref (3) for details; g0.5 equiv of AgOTf; htrifluoromethanesulfonate (CF3SO2–) counterion and THF solvate omitted for clarity. (C) Summarized access to [4], [5], and [6]helicenes.
References
This article references 31 other publications.
- 1Stara, I. G.; Crassous, J.; Stary, I. Helicenes synthesis, properties, and applications; Wiley-VCH, 2023.There is no corresponding record for this reference.
- 2Chen, J.; Captain, B.; Takenaka, N. Helical Chiral 2,2′-Bipyridine N- Monoxides as Catalysts in the Enantioselective Propargylation of Aldehydes with Allenyltrichlorosilane. Org. Lett. 2011, 13 (7), 1654– 1657, DOI: 10.1021/ol200102cThere is no corresponding record for this reference.
- 3Míšek, J.; Teplý, F.; Stará, I. G.; Tichý, M.; Šaman, D.; Císařová, I.; Vojtíšek, P.; Starý, I. A Straightforward Route to Helically Chiral N-Heteroaromatic Compounds: Practical Synthesis of Racemic 1,14-Diaza[5]Helicene and Optically Pure 1- and 2-Aza[6]Helicenes. Angew. Chem., Int. Ed. 2008, 47 (17), 3188– 3191, DOI: 10.1002/anie.200705463There is no corresponding record for this reference.
- 4Takenaka, N.; Sarangthem, R. S.; Captain, B. Helical Chiral Pyridine N-Oxides: A New Family of Asymmetric Catalysts. Angew. Chem., Int. Ed. 2008, 47 (50), 9708– 9710, DOI: 10.1002/anie.200803338There is no corresponding record for this reference.
- 5Weimar, M.; Correa da Costa, R.; Lee, F.-H.; Fuchter, M. J. A Scalable and Expedient Route to 1-Aza[6]Helicene Derivatives and Its Subsequent Application to a Chiral-Relay Asymmetric Strategy. Org. Lett. 2013, 15 (7), 1706– 1709, DOI: 10.1021/ol400493xThere is no corresponding record for this reference.
- 6Brandt, J. R.; Pospisil, L.; Bednarova, L.; da Costa, R. C.; White, A. J. P.; Mori, T.; Teply, F.; Fuchter, M. J. Intense Redox-Driven Chiroptical Switching with a 580 mV Hysteresis Actuated through Reversible Dimerization of an Azoniahelicene. Chem. Commun. 2017, 53 (65), 9059– 9062, DOI: 10.1039/C7CC04903JThere is no corresponding record for this reference.
- 7Yang, Y.; Rice, B.; Shi, X.; Brandt, J. R.; Correa da Costa, R.; Hedley, G. J.; Smilgies, D.-M.; Frost, J. M.; Samuel, I. D. W.; Otero-de-la-Roza, A.; Johnson, E. R.; Jelfs, K. E.; Nelson, J.; Campbell, A. J.; Fuchter, M. J. Emergent Properties of an Organic Semiconductor Driven by Its Molecular Chirality. ACS Nano 2017, 11 (8), 8329– 8338, DOI: 10.1021/acsnano.7b03540There is no corresponding record for this reference.
- 8Wade, J.; Hilfiker, J. N.; Brandt, J. R.; Liirò-Peluso, L.; Wan, L.; Shi, X.; Salerno, F.; Ryan, S. T. J.; Schöche, S.; Arteaga, O.; Jávorfi, T.; Siligardi, G.; Wang, C.; Amabilino, D. B.; Beton, P. H.; Campbell, A. J.; Fuchter, M. J. Natural Optical Activity as the Origin of the Large Chiroptical Properties in π-Conjugated Polymer Thin Films. Nat. Commun. 2020, 11 (1), 6137, DOI: 10.1038/s41467-020-19951-yThere is no corresponding record for this reference.
- 9Crassous, J.; Fuchter, M. J.; Freedman, D. E.; Kotov, N. A.; Moon, J.; Beard, M. C.; Feldmann, S. Materials for Chiral Light Control. Nat. Rev. Mater. 2023, 8 (6), 365– 371, DOI: 10.1038/s41578-023-00543-3There is no corresponding record for this reference.
- 10Matsumoto, A.; Yonemitsu, K.; Ozaki, H.; Míšek, J.; Starý, I.; Stará, I. G.; Soai, K. Reversal of the Sense of Enantioselectivity between 1- and 2-Aza[6]Helicenes Used as Chiral Inducers of Asymmetric Autocatalysis. Org. Biomol. Chem. 2017, 15 (6), 1321– 1324, DOI: 10.1039/C6OB02745HThere is no corresponding record for this reference.
- 11Harrowven, D. C.; Guy, I. L.; Nanson, L. Efficient Phenanthrene, Helicene, and Azahelicene Syntheses. Angew. Chem., Int. Ed. 2006, 45 (14), 2242– 2245, DOI: 10.1002/anie.200504287There is no corresponding record for this reference.
- 12Kawashima, T.; Matsumoto, Y.; Sato, T.; Yamada, Y. M. A.; Kono, C.; Tsurusaki, A.; Kamikawa, K. Synthesis, Structure, and Complexation of an S-Shaped Double Azahelicene with Inner-Edge Nitrogen Atoms. Chem. – Eur. J. 2020, 26 (58), 13170– 13176, DOI: 10.1002/chem.202002405There is no corresponding record for this reference.
- 13Abbate, S.; Bazzini, C.; Caronna, T.; Fontana, F.; Gambarotti, C.; Gangemi, F.; Longhi, G.; Mele, A.; Sora, I. N.; Panzeri, W. Monoaza[5]Helicenes. Part 2: Synthesis, Characterisation and Theoretical Calculations. Tetrahedron 2006, 62 (1), 139– 148, DOI: 10.1016/j.tet.2005.09.132There is no corresponding record for this reference.
- 14Yu, T.; Li, Z.-Q.; Li, J.; Cheng, S.; Xu, J.; Huang, J.; Zhong, Y.-W.; Luo, S.; Zhu, Q. Palladium-Catalyzed Modular Synthesis of Enantioenriched Pyridohelicenes through Double Imidoylative Cyclization. ACS Catal. 2022, 12 (20), 13034– 13041, DOI: 10.1021/acscatal.2c04461There is no corresponding record for this reference.
- 15Liu, W.; Qin, T.; Xie, W.; Zhou, J.; Ye, Z.; Yang, X. Enantioselective Synthesis of Azahelicenes through Organocatalyzed Multicomponent Reactions. Angew. Chem., Int. Ed. 2023, 62 (27), e202303430 DOI: 10.1002/anie.202303430There is no corresponding record for this reference.
- 16Zhang, S.; Zhang, D.; Liebeskind, L. S. Ambient Temperature, Ullmann-like Reductive Coupling of Aryl, Heteroaryl, and Alkenyl Halides. J. Org. Chem. 1997, 62 (8), 2312– 2313, DOI: 10.1021/jo9700078There is no corresponding record for this reference.
- 17Xue, W.; Xu, H.; Liang, Z.; Qian, Q.; Gong, H. Nickel-Catalyzed Reductive Cyclization of Alkyl Dihalides. Org. Lett. 2014, 16 (19), 4984– 4987, DOI: 10.1021/ol502207zThere is no corresponding record for this reference.
- 18Maigrot, N.; Mazaleyrat, J.-P. New and Improved Synthesis of Optically Pure (R)-and (S)-2,2′-Dimethyl-1, 1′-Binaphthyl and Related Compounds. Synthesis 1985, 1985, 317– 320, DOI: 10.1055/s-1985-31196There is no corresponding record for this reference.
- 19Stará, I. G.; Starý, I.; Závada, J. Optically Pure (S)- and (R)-4,5-Dihydro-3H-4-Methyldinaphth[2,1-c; 1′,2′-e]Azepines. Application to the Synthesis of New Bidentate Ligands with Axial Asymmetry. Tetrahedron Asymmetry 1992, 3 (11), 1365– 1368, DOI: 10.1016/0957-4166(92)80007-JThere is no corresponding record for this reference.
- 20Stara, I. G.; Stary, I.; Tichy, M.; Zavada, J.; Hanus, V. Stereochemical Dichotomy in the Stevens Rearrangement of Axially Twisted Dihydroazepinium and Dihydrothiepinium Salts. A Novel Enantioselective Synthesis of Pentahelicene. J. Am. Chem. Soc. 1994, 116 (12), 5084– 5088, DOI: 10.1021/ja00091a009There is no corresponding record for this reference.
- 21McFadden, T. P.; Nwachukwu, C. I.; Roberts, A. G. An Amine Template Strategy to Construct Successive C–C Bonds: Synthesis of Benzo[h]quinolines by a Deaminative Ring Contraction Cascade. Org. Biomol. Chem. 2022, 20 (7), 1379– 1385, DOI: 10.1039/D1OB02245HThere is no corresponding record for this reference.
- 22Kirkeby, E. K.; Schwartz, Z. T.; Lovasz, M. A.; Roberts, A. G. Deaminative Ring Contraction for the Synthesis of Polycyclic Heteroaromatics: A Concise Total Synthesis of Toddaquinoline. Chem. Sci. 2023, 14, 10508– 10514, DOI: 10.1039/D3SC03936FThere is no corresponding record for this reference.
- 23Schwartz, Z.; Valiton, C.; Lovasz, M.; Roberts, A. G. Recent Applications of Ammonium Ylide Based [2,3]-Sigmatropic and [1,2]-Stevens Rearrangements to Transform Amines into Natural Products. Synthesis 2024, 56, 87, DOI: 10.1055/s-0042-1751446There is no corresponding record for this reference.
- 24Iyoda, M.; Otsuka, H.; Sato, K.; Nisato, N.; Oda, M. Homocoupling of Aryl Halides Using Nickel(II) Complex and Zinc in the Presence of Et4NI. An Efficient Method for the Synthesis of Biaryls and Bipyridines. Bull. Chem. Soc. Jpn. 1990, 63 (1), 80– 87, DOI: 10.1246/bcsj.63.80There is no corresponding record for this reference.
- 25Leleu, S.; Papamicaël, C.; Marsais, F.; Dupas, G.; Levacher, V. Preparation of Axially Chiral Quinolinium Salts Related to NAD+ Models: New Investigations of These Biomimetic Models as ‘Chiral Amide-Transferring Agents. Tetrahedron Asymmetry 2004, 15 (24), 3919– 3928, DOI: 10.1016/j.tetasy.2004.11.004There is no corresponding record for this reference.
- 26Na, H.; Mirica, L. M. Deciphering the Mechanism of the Ni-Photocatalyzed C–O Cross-Coupling Reaction Using a Tridentate Pyridinophane Ligand. Nat. Commun. 2022, 13 (1), 1313, DOI: 10.1038/s41467-022-28948-8There is no corresponding record for this reference.
- 27He, Z.-T.; Li, H.; Haydl, A. M.; Whiteker, G. T.; Hartwig, J. F. Trimethylphosphate as a Methylating Agent for Cross Coupling: A Slow-Release Mechanism for the Methylation of Arylboronic Esters. J. Am. Chem. Soc. 2018, 140 (49), 17197– 17202, DOI: 10.1021/jacs.8b10076There is no corresponding record for this reference.
- 28Nwachukwu, C. I.; McFadden, T. P.; Roberts, A. G. Ni-Catalyzed Iterative Alkyl Transfer from Nitrogen Enabled by the In Situ Methylation of Tertiary Amines. J. Org. Chem. 2020, 85 (15), 9979– 9992, DOI: 10.1021/acs.joc.0c01274There is no corresponding record for this reference.
- 29von Hofmann, A. W. Beiträge Zur Kenntniss Der Flüchtigen Organischen Basen. Justus Liebigs Ann. Chem. 1851, 78 (3), 253– 286, DOI: 10.1002/jlac.18510780302There is no corresponding record for this reference.
- 30Dick, A. R.; Hull, K. L.; Sanford, M. S. A Highly Selective Catalytic Method for the Oxidative Functionalization of C–H Bonds. J. Am. Chem. Soc. 2004, 126 (8), 2300– 2301, DOI: 10.1021/ja031543mThere is no corresponding record for this reference.
- 31Schwartz, Z. T.; Valiton, C. D.; Lovasz, M. A.; Sadat, F.; Phan, M.; VanderLinden, R.; Richmond, T.; Roberts, A. G. Deaminative ring contraction for the modular synthesis of pyrido[n]helicenes. ChemRxiv 2024, DOI: 10.26434/chemrxiv-2024-qdd0nThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.orglett.5c04734.
Experimental procedures and characterization data (HRMS and 1H and 13C NMR spectra for the reported synthetic compounds) (PDF)
Deposition Numbers 2402615–2402616 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via the joint Cambridge Crystallographic Data Centre (CCDC) and Fachinformationszentrum Karlsruhe Access Structures service.
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



