Perspective on Thermal Stability and Safety of Sodium-Ion BatteriesClick to copy article linkArticle link copied!
- KausthubharamKausthubharamSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Kausthubharam
- Bairav S. VishnugopiBairav S. VishnugopiSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Bairav S. Vishnugopi
- Abhinanda SenguptaAbhinanda SenguptaSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Abhinanda Sengupta
- Dhevathi Rajan Rajagopalan KannanDhevathi Rajan Rajagopalan KannanElectrochemical Safety Research Institute, UL Research Institutes, Houston, Texas 77204, United States
- Vinay PremnathVinay PremnathElectrochemical Safety Research Institute, UL Research Institutes, Houston, Texas 77204, United StatesMore by Vinay Premnath
- Wan Si TangWan Si TangElectrochemical Safety Research Institute, UL Research Institutes, Houston, Texas 77204, United StatesMore by Wan Si Tang
- Judith A. Jeevarajan*Judith A. Jeevarajan*[email protected],.Electrochemical Safety Research Institute, UL Research Institutes, Houston, Texas 77204, United StatesMore by Judith A. Jeevarajan
- Partha P. Mukherjee*Partha P. Mukherjee*[email protected]School of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Partha P. Mukherjee
Abstract
Sodium-ion batteries (SIBs) are gaining traction as an emerging contender for sustainable and cost-effective energy storage, due to the abundance and low cost of sodium resources. Although notable advancements have been made in improving electrochemical performance, the thermal stability of SIBs and the role of intrinsic degradation pathways are yet to be fully understood. This Perspective examines the mechanistic interactions that drive thermal instability in SIBs across material, electrode, and cell levels under operational extremes and abuse conditions. We analyze the thermo-electrochemical characteristics of key electrode and electrolyte components, including their interphases, to identify the underlying factors responsible for the distinct thermal response of SIBs compared to lithium-ion batteries (LIBs). By benchmarking current SIB prototypes against commercial LIB technologies in terms of cost–performance trade-offs, we outline critical challenges that must be addressed to enable safe and scalable deployment of SIB systems.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
Non-Commercial (NC): Only non-commercial uses of the work are permitted.
No Derivatives (ND): Derivative works may be created for non-commercial purposes, but sharing is prohibited.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Figure 1
Figure 1. (a) Historical timeline of sodium-ion battery (SIB) development, highlighting key discoveries in cathode and anode chemistries, along with the emergence of recent commercial and mass-manufacturing efforts. (b) Comparison of energy storage capacities of various cathode and anode materials. (6,23−25) (Electrochemical properties are provided in Table S1 in the Supporting Information.) (c) Comparison of key characteristics of sodium ions and lithium ions. (26−28) (d) Schematic overview of key considerations for developing practical SIBs.
Material-Level Thermal Stability
Figure 2
Figure 2. (a) Schematic illustration of typical thermal degradation pathways in sodium-ion cathodes upon heating, including layered, PBA, and polyanionic compounds. (b) Comparison of phase transitions during charging for layered sodium-ion cathodes: NaNi0.33Mn0.33Co0.33O2 (NNMCO), NaNi0.5Mn0.5O2 (NNMO), and NaCoO2 (NCO) with their lithium-ion counterparts, LiCoO2 (LCO) and LiNi0.33Mn0.33Co0.33O2 (NMC). Here, each number corresponds to different phases exhibited by the cathode, as provided in Table S2 of the Supporting Information. (37,38,56) (c) In situ XRD profile of Na0.8Ni0.33Mn0.67O2 (NNMO) during heating using X-ray beam of wavelength 0.154 nm (Reproduced with permission from ref (32). Copyright 2025 Wiley-VCH GmbH, Weinheim.) (d) Thermodynamic phase diagram of the Na-Pb binary alloy system. (Adapted from ref (57). Copyright 2024 Electrochemical Society (ECS) & IOP Science.) (e) Schematic showing the influence of hard carbon (HC) fabrication temperature on anode microstructure and sodium metal agglomeration. (f) Influence of fabrication temperature on the ion storage mechanism in HC. (Adapted from ref (52). Copyright 2020 Royal Society of Chemistry (RSC).)
Interface and Interphase-Level Mechanistic Interactions
Figure 3
Figure 3. (a) Comparison of thermal stability of the anode interphase for various salt–solvent combinations used in SIBs (additional details are provided in Table S3 of the Supporting Information). (59,73) (b) Schematic illustration showing the relationship between solvation energy and thermal stability of the anode interphase. (c) Spider chart depicting thermal stability characteristics of different Sn alloy anodes with and without FEC additives after cycling. (Adapted from ref (55). Copyright 2025 American Chemical Society (ACS).) Here μ-Sn and n-Sn refer to micro-Sn and nano-Sn, respectively. (d) SEM images of pristine and cycled n-Sn particles showing nonuniform interphase formation after cycling without additives. (Reproduced with permission from ref (66). Copyright 2022 American Chemical Society (ACS).) (e) Comparison of the effect of FEC on the CE of HC and Sn anodes. (66,72)
Figure 4
Figure 4. (a) Schematic illustration of typical thermal instability pathways of the CEI in layered oxide and PBA cathodes. (b) In situ XRD pattern of charged NFM in 1 M NaPF6 in propylene carbonate:ethyl methyl carbonate (PC:EMC) with 2 wt% FEC using an X-ray beam of wavelength 0.012 nm. (Reproduced with permission from ref (77). Copyright 2018 American Chemical Society (ACS).) (c) Comparative analysis of gas evolution normalized to volumetric energy density for NVPF and LFP cathodes. (Adapted from ref (82). Copyright 2022 American Chemical Society (ACS).) (d) Comparison of the thermal stability of different cathodes based on the relationship between the onset temperature for thermal degradation and cutoff voltage (figure numbers for the representative data sets are summarized in Table S4 of the Supporting Information). (e) Thermal stability profile of carbon-coated NVP as a function of SOC. (Adapted from ref (83). Copyright 2024 Elsevier.) (f) Schematic depiction of how increasing SOC in layered oxide cathodes influences degradation mechanisms and the severity of thermal runaway.
Cell-Level Thermal Stability and Safety
Figure 5
Figure 5. (a) Schematic illustration of the typical thermal runaway pathways in SIB cells. (b) Thermal safety characteristics in NaxTMO2 (NTM) and LFP cathodes. (Adapted from ref (87). Copyright 2024 Elsevier.) (c) Heat release comparison between LIB and SIB cells with cathode and anode contributions. (Adapted from ref (86). Copyright 2021 American Chemical Society (ACS).) (d) SEM micrographs of SIB cathode and anode after thermal abuse testing. (Reproduced with permission from ref (88). Copyright 2018 Elsevier.) (e) Microcalorimetry profile showing heat generation during a single charge/discharge cycle in a pristine NVP/HC full cell. (92) (f) Comparison of heat release during thermal abuse tests of different cathodes and anodes (more details regarding the electrode–electrolyte combination for this data set are provided in Table S5 in the Supporting Information). (g) Maximum temperatures observed during nail penetration and overcharge tests for SIBs using NVPF, NFM, and NCFMO cathodes, compared to LFP-based cells (8,89,93)
Cost Considerations and Thermal Stability Metrics for Commercialization of SIBs
Figure 6
Figure 6. (a) Comparison of energy density and cycle life (at 80% capacity retention) of various SIB prototypes from different manufacturers relative to BYD’s lithium-ion blade battery. (Adapted from refs (96and97). Copyright 2023 Springer Nature and 2023 MDPI.) (b) Breakdown of material and processing costs for commercially available LIB and SIB systems. (Adapted from refs (98and99). Copyright 2019 MDPI and 2025 Cell Press.) (c) Specific energy comparison of different cathode materials as a function of areal loading. (Adapted from ref (94). Copyright 2021 Elsevier.) (d) Effect of heating power during thermal abuse tests on the maximum temperature (T3) and time interval between onset of self-heating and thermal runaway (t2 – t1) for layered oxide-based SIBs. (Adapted from ref (95). Copyright 2025 Elsevier.) (e) Representative illustration of heat generation as a function of charging rate. (f) Schematic showing the hierarchical set of factors (i.e., at the particle, interphase, and electrode levels) affecting SIB safety.
Hierarchical Factors Affecting SIB Safety
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c02345.
Summary of electrochemical properties of sodium-ion cathodes and anodes (Table S1), Comparison of phase behavior of different sodium-ion and lithium-ion layered oxides (Table S2), Exothermic parameters of different sodium salt-solvent combinations with the salt concentration of 1 M (Table S3), Thermal stability of different sodium-ion cathodes as a function of cutoff voltages (Table S4), Typical electrode–electrolyte configurations used in thermal stability investigations of sodium-ion batteries (Table S5), Properties of commonly used solvents in sodium-ion batteries (Table S6) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Biographies
Kausthubharam
Kausthubharam is a doctoral candidate in the School of Mechanical Engineering at Purdue University. He received his M.S. in Mechanical Engineering from Seoul National University, Korea, in 2023. His research focuses on interrogating the thermo-electrochemical interactions and degradation pathways in beyond Li-ion chemistries.
Bairav S. Vishnugopi
Bairav S. Vishnugopi is a Research Assistant Professor in the School of Mechanical Engineering at Purdue University. His research focuses on understanding the electro-chemo-mechanical, transport, and thermal interactions in electrode architectures and interfaces for energy storage. He has co-authored more than 60 journal publications spanning different battery chemistries and applications.
Abhinanda Sengupta
Abhinanda Sengupta is a postdoctoral associate in the School of Mechanical Engineering at Purdue University. She received her doctorate degree from the Indian Institute of Technology Bombay, India. Her research focuses on fundamental interrogation of electrochemical and thermal instability for sodium-ion batteries using operando experiments and thermo-electrochemical analytics.
Dhevathi Rajan Rajagopalan Kannan
Dhevathi Rajan Rajagopalan Kannan is a Research Scientist at UL Research Institutes (ULRI). He has over seven years of experience in battery research. Before joining ULRI, he worked in automotive and solid-state battery companies. His current research focuses on the safety and performance of various lithium-ion and sodium-ion battery chemistries.
Vinay Premnath
Vinay Premnath is the Director of Research with focus on energy storage safety at the Electrochemical Safety Research Institute (UL Research Institutes). Prior to joining UL Research Institutes, Vinay was a Principal Engineer at Southwest Research Institute. Vinay received his MS degree from the University of Minnesota, Twin Cities.
Wan Si Tang
Wan Si Tang is the Director of Research at the Electrochemical Safety Research Institute (UL Research Institutes). She has 15+ years of experience in novel functional materials for energy storage and conversion and advanced battery manufacturing. Wan Si is the recipient of the 2025 ECD Jubilee Global Diversity Award. (https://wansitang.wordpress.com/)
Judith A. Jeevarajan
Judith A. Jeevarajan is the Vice President and Executive Director for the Electrochemical Safety Research Institute at UL Research Institutes (ULRI). With more than 29 years of experience, she specializes in battery safety for lithium-ion and sodium-ion cells and modules, recycling, thermal runaway, fire, smoke, particulate emissions, and fire suppressants.
Partha P. Mukherjee
Partha P. Mukherjee is a University Faculty Scholar and Professor of Mechanical Engineering at Purdue University, specializing in mesoscale modeling and experimental analytics of transport, chemistry, microstructure, and interface interactions in energy storage and conversion. He has published >250 journal articles on different battery chemistries and energy conversion systems. (https://engineering.purdue.edu/ETSL/)
Acknowledgments
The funding for this research was provided by UL Research Institutes through the Center for Advances in Resilient Energy Storage (CARES).
References
This article references 99 other publications.
- 1Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 Years of Lithium-Ion Batteries. Adv. Mater. 2018, 30 (33), e1800561 DOI: 10.1002/adma.201800561Google ScholarThere is no corresponding record for this reference.
- 2Tarascon, J.-M. Na-ion versus Li-ion Batteries: Complementarity Rather than Competitiveness. Joule 2020, 4 (8), 1616– 1620, DOI: 10.1016/j.joule.2020.06.003Google ScholarThere is no corresponding record for this reference.
- 3Tarascon, J. M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414 (6861), 359– 367, DOI: 10.1038/35104644Google ScholarThere is no corresponding record for this reference.
- 4Hirsh, H. S.; Li, Y.; Tan, D. H. S.; Zhang, M.; Zhao, E.; Meng, Y. S. Sodium-Ion Batteries Paving the Way for Grid Energy Storage. Adv. Energy Mater. 2020, 10 (32), 2001274 DOI: 10.1002/aenm.202001274Google ScholarThere is no corresponding record for this reference.
- 5Hwang, J. Y.; Myung, S. T.; Sun, Y. K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46 (12), 3529– 3614, DOI: 10.1039/C6CS00776GGoogle ScholarThere is no corresponding record for this reference.
- 6Usiskin, R.; Lu, Y.; Popovic, J.; Law, M.; Balaya, P.; Hu, Y.-S.; Maier, J. Fundamentals, status and promise of sodium-based batteries. Nature Reviews Materials 2021, 6 (11), 1020– 1035, DOI: 10.1038/s41578-021-00324-wGoogle ScholarThere is no corresponding record for this reference.
- 7Velumani, D.; Bansal, A. Thermal Behavior of Lithium- and Sodium-Ion Batteries: A Review on Heat Generation, Battery Degradation, Thermal Runway – Perspective and Future Directions. Energy Fuels 2022, 36 (23), 14000– 14029, DOI: 10.1021/acs.energyfuels.2c02889Google ScholarThere is no corresponding record for this reference.
- 8Bates, A.; Torres-Castro, L.; Albertus, P.; Mukherjee, P. P.; Jeevarajan, J.; Johnson, N.; Bhargava, B.; Wang, Z.; Ogundipe, T.; Vishnugopi, B. S.; Karmakar, A.; Tang, W. S. Multi-scale safety evaluation of emerging battery technologies. ECS Interface 2024, 33 (3), 69, DOI: 10.1149/2.F10243IFGoogle ScholarThere is no corresponding record for this reference.
- 9Delmas, C. Sodium and Sodium-Ion Batteries: 50 Years of Research. Adv. Energy Mater. 2018, 8 (17), 1703137 DOI: 10.1002/aenm.201703137Google ScholarThere is no corresponding record for this reference.
- 10Yu, T.; Li, G.; Duan, Y.; Wu, Y.; Zhang, T.; Zhao, X.; Luo, M.; Liu, Y. The research and industrialization progress and prospects of sodium ion battery. J. Alloys Compd. 2023, 958, 170486 DOI: 10.1016/j.jallcom.2023.170486Google ScholarThere is no corresponding record for this reference.
- 11Yao, A.; Benson, S. M.; Chueh, W. C. Critically assessing sodium-ion technology roadmaps and scenarios for techno-economic competitiveness against lithium-ion batteries. Nature Energy 2025, 10 (3), 404– 416, DOI: 10.1038/s41560-024-01701-9Google ScholarThere is no corresponding record for this reference.
- 12Han, M. H.; Gonzalo, E.; Singh, G.; Rojo, T. A comprehensive review of sodium layered oxides: powerful cathodes for Na-ion batteries. Energy Environ. Sci. 2015, 8 (1), 81– 102, DOI: 10.1039/C4EE03192JGoogle ScholarThere is no corresponding record for this reference.
- 13Gao, X.; Chen, J.; Liu, H.; Yin, S.; Tian, Y.; Cao, X.; Zou, G.; Hou, H.; Wei, W.; Chen, L. Copper-substituted NaxMO2 (M = Fe, Mn) cathodes for sodium ion batteries: Enhanced cycling stability through suppression of Mn(III) formation. Chemical Engineering Journal 2021, 406, 126830 DOI: 10.1016/j.cej.2020.126830Google ScholarThere is no corresponding record for this reference.
- 14Subasinghe, L. U.; Satyanarayana Reddy, G.; Rudola, A.; Balaya, P. Analysis of Heat Generation and Impedance Characteristics of Prussian Blue Analogue Cathode-based 18650-type Sodium-ion Cells. J. Electrochem. Soc. 2020, 167 (11), 110504 DOI: 10.1149/1945-7111/ab9ee9Google ScholarThere is no corresponding record for this reference.
- 15Dugas, R.; Zhang, B.; Rozier, P.; Tarascon, J. M. Optimization of Na-Ion Battery Systems Based on Polyanionic or Layered Positive Electrodes and Carbon Anodes. J. Electrochem. Soc. 2016, 163 (6), A867– A874, DOI: 10.1149/2.0051605jesGoogle ScholarThere is no corresponding record for this reference.
- 16Lee, D. H.; Xu, J.; Meng, Y. S. An advanced cathode for Na-ion batteries with high rate and excellent structural stability. Phys. Chem. Chem. Phys. 2013, 15 (9), 3304– 3312, DOI: 10.1039/c2cp44467dGoogle ScholarThere is no corresponding record for this reference.
- 17Mukherjee, S.; Bin Mujib, S.; Soares, D.; Singh, G. Electrode Materials for High-Performance Sodium-Ion Batteries. Materials (Basel) 2019, 12 (12), 1952, DOI: 10.3390/ma12121952Google ScholarThere is no corresponding record for this reference.
- 18Fang, Y.; Yu, X.-Y.; Lou, X. W. Nanostructured Electrode Materials for Advanced Sodium-Ion Batteries. Matter 2019, 1 (1), 90– 114, DOI: 10.1016/j.matt.2019.05.007Google ScholarThere is no corresponding record for this reference.
- 19Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical Analysis on De-Solvation of Lithium, Sodium, and Magnesium Cations to Organic Electrolyte Solvents. J. Electrochem. Soc. 2013, 160 (11), A2160, DOI: 10.1149/2.074311jesGoogle ScholarThere is no corresponding record for this reference.
- 20Hu, Y.-S.; Pan, H. Solvation Structures in Electrolyte and the Interfacial Chemistry for Na-Ion Batteries. ACS Energy Letters 2022, 7 (12), 4501– 4503, DOI: 10.1021/acsenergylett.2c02529Google ScholarThere is no corresponding record for this reference.
- 21Nayak, P. K.; Yang, L.; Brehm, W.; Adelhelm, P. From Lithium-Ion to Sodium-Ion Batteries: Advantages, Challenges, and Surprises. Angew. Chem., Int. Ed. Engl. 2018, 57 (1), 102– 120, DOI: 10.1002/anie.201703772Google ScholarThere is no corresponding record for this reference.
- 22Slater, M. D.; Kim, D.; Lee, E.; Johnson, C. S. Sodium-Ion Batteries. Adv. Funct. Mater. 2013, 23 (8), 947– 958, DOI: 10.1002/adfm.201200691Google ScholarThere is no corresponding record for this reference.
- 23Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46 (12), 3529– 3614, DOI: 10.1039/C6CS00776GGoogle ScholarThere is no corresponding record for this reference.
- 24Lyu, Y.; Liu, Y.; Yu, Z.-E.; Su, N.; Liu, Y.; Li, W.; Li, Q.; Guo, B.; Liu, B. Recent advances in high energy-density cathode materials for sodium-ion batteries. Sustainable Materials and Technologies 2019, 21, e00098 DOI: 10.1016/j.susmat.2019.e00098Google ScholarThere is no corresponding record for this reference.
- 25Goikolea, E.; Palomares, V.; Wang, S.; de Larramendi, I. R.; Guo, X.; Wang, G.; Rojo, T. Na-Ion Batteries─Approaching Old and New Challenges. Adv. Energy Mater. 2020, 10 (44), 2002055 DOI: 10.1002/aenm.202002055Google ScholarThere is no corresponding record for this reference.
- 26Islam, M. S.; Fisher, C. A. J. Lithium and sodium battery cathode materials: computational insights into voltage, diffusion and nanostructural properties. Chem. Soc. Rev. 2014, 43 (1), 185– 204, DOI: 10.1039/C3CS60199DGoogle ScholarThere is no corresponding record for this reference.
- 27Kim, S. C.; Gao, X.; Liao, S.-L.; Su, H.; Chen, Y.; Zhang, W.; Greenburg, L. C.; Pan, J.-A.; Zheng, X.; Ye, Y. Solvation-property relationship of lithium-sulphur battery electrolytes. Nat. Commun. 2024, 15 (1), 1268, DOI: 10.1038/s41467-023-44527-xGoogle ScholarThere is no corresponding record for this reference.
- 28Klein, F.; Jache, B.; Bhide, A.; Adelhelm, P. Conversion reactions for sodium-ion batteries. Phys. Chem. Chem. Phys. 2013, 15 (38), 15876– 15887, DOI: 10.1039/c3cp52125gGoogle ScholarThere is no corresponding record for this reference.
- 29Chen, C.; Han, Z.; Chen, S.; Qi, S.; Lan, X.; Zhang, C.; Chen, L.; Wang, P.; Wei, W. Core-Shell Layered Oxide Cathode for High-Performance Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2020, 12 (6), 7144– 7152, DOI: 10.1021/acsami.9b19260Google ScholarThere is no corresponding record for this reference.
- 30Cui, Z.; Manthiram, A. Thermal Stability and Outgassing Behaviors of High-nickel Cathodes in Lithium-ion Batteries. Angew. Chem., Int. Ed. 2023, 62 (43), e202307243 DOI: 10.1002/anie.202307243Google ScholarThere is no corresponding record for this reference.
- 31Deng, J.; Luo, W. B.; Lu, X.; Yao, Q.; Wang, Z.; Liu, H. K.; Zhou, H.; Dou, S. X. High Energy Density Sodium-Ion Battery with Industrially Feasible and Air-Stable O3-Type Layered Oxide Cathode. Adv. Energy Mater. 2018, 8 (5), 1701610 DOI: 10.1002/aenm.201701610Google ScholarThere is no corresponding record for this reference.
- 32Li, Q.; Li, Y.; Liu, M.; Li, Y.; Zhao, H.; Ren, H.; Zhao, Y.; Zhou, Q.; Feng, X.; Shi, J. Elucidating Thermal Decomposition Kinetic Mechanism of Charged Layered Oxide Cathode for Sodium-Ion Batteries. Adv. Mater. 2025, 37 (10), e2415610 DOI: 10.1002/adma.202415610Google ScholarThere is no corresponding record for this reference.
- 33Hu, E.; Wang, X.; Yu, X.; Yang, X.-Q. Probing the Complexities of Structural Changes in Layered Oxide Cathode Materials for Li-Ion Batteries during Fast Charge–Discharge Cycling and Heating. Acc. Chem. Res. 2018, 51 (2), 290– 298, DOI: 10.1021/acs.accounts.7b00506Google ScholarThere is no corresponding record for this reference.
- 34Zuo, W.; Qiu, J.; Liu, X.; Ren, F.; Liu, H.; He, H.; Luo, C.; Li, J.; Ortiz, G. F.; Duan, H. The stability of P2-layered sodium transition metal oxides in ambient atmospheres. Nat. Commun. 2020, 11 (1), 3544, DOI: 10.1038/s41467-020-17290-6Google ScholarThere is no corresponding record for this reference.
- 35Xiao, B.; Omenya, F.; Reed, D.; Li, X. A glance of the layered transition metal oxide cathodes in sodium and lithium-ion batteries: difference and similarities. Nanotechnology 2021, 32 (42), 422501 DOI: 10.1088/1361-6528/ac12ebGoogle ScholarThere is no corresponding record for this reference.
- 36Wang, P.-F.; You, Y.; Yin, Y.-X.; Guo, Y.-G. An O3-type NaNi0.5Mn0.5O2cathode for sodium-ion batteries with improved rate performance and cycling stability. Journal of Materials Chemistry A 2016, 4 (45), 17660– 17664, DOI: 10.1039/C6TA07589DGoogle ScholarThere is no corresponding record for this reference.
- 37Sathiya, M.; Hemalatha, K.; Ramesha, K.; Tarascon, J. M.; Prakash, A. S. Synthesis, Structure, and Electrochemical Properties of the Layered Sodium Insertion Cathode Material: NaNi1/3Mn1/3Co1/3O2. Chem. Mater. 2012, 24 (10), 1846– 1853, DOI: 10.1021/cm300466bGoogle ScholarThere is no corresponding record for this reference.
- 38Lei, Y.; Li, X.; Liu, L.; Ceder, G. Synthesis and Stoichiometry of Different Layered Sodium Cobalt Oxides. Chem. Mater. 2014, 26 (18), 5288– 5296, DOI: 10.1021/cm5021788Google ScholarThere is no corresponding record for this reference.
- 39Sengupta, A.; Kumar, A.; Barik, G.; Ahuja, A.; Ghosh, J.; Lohani, H.; Kumari, P.; Bhandakkar, T. K.; Mitra, S. Lower Diffusion-Induced Stress in Nano-Crystallites of P2-Na(2/3) Ni(1/3) Mn(1/2) Ti(1/6) O(2) Novel Cathode for High Energy Na-ion Batteries. Small 2023, 19 (12), e2206248 DOI: 10.1002/smll.202206248Google ScholarThere is no corresponding record for this reference.
- 40Sengupta, A.; Kumar, A.; Bano, A.; Ahuja, A.; Lohani, H.; Akella, S. H.; Kumari, P.; Noked, M.; Major, D. T.; Mitra, S. Unleashing the impact of Nb-doped, single crystal, cobalt-free P2-type Na0.67Ni0.33Mn0.67O2 on elevating the cycle life of sodium-ion batteries. Energy Storage Materials 2024, 69, 103435 DOI: 10.1016/j.ensm.2024.103435Google ScholarThere is no corresponding record for this reference.
- 41Guo, S.; Li, Q.; Liu, P.; Chen, M.; Zhou, H. Environmentally stable interface of layered oxide cathodes for sodium-ion batteries. Nat. Commun. 2017, 8 (1), 135, DOI: 10.1038/s41467-017-00157-8Google ScholarThere is no corresponding record for this reference.
- 42You, Y.; Dolocan, A.; Li, W.; Manthiram, A. Understanding the Air-Exposure Degradation Chemistry at a Nanoscale of Layered Oxide Cathodes for Sodium-Ion Batteries. Nano Lett. 2019, 19 (1), 182– 188, DOI: 10.1021/acs.nanolett.8b03637Google ScholarThere is no corresponding record for this reference.
- 43Xiao, Y.; Xiao, J.; Zhao, H.; Li, J.; Zhang, G.; Zhang, D.; Guo, X.; Gao, H.; Wang, Y.; Chen, J. Prussian Blue Analogues for Sodium-Ion Battery Cathodes: A Review of Mechanistic Insights, Current Challenges, and Future Pathways. Small 2024, 20 (35), e2401957 DOI: 10.1002/smll.202401957Google ScholarThere is no corresponding record for this reference.
- 44Wang, W.; Gang, Y.; Peng, J.; Hu, Z.; Yan, Z.; Lai, W.; Zhu, Y.; Appadoo, D.; Ye, M.; Cao, Y. Effect of Eliminating Water in Prussian Blue Cathode for Sodium-Ion Batteries. Adv. Funct. Mater. 2022, 32 (25), 2111727 DOI: 10.1002/adfm.202111727Google ScholarThere is no corresponding record for this reference.
- 45Liu, Q.; Hu, Z.; Chen, M.; Zou, C.; Jin, H.; Wang, S.; Chou, S. L.; Liu, Y.; Dou, S. X. The Cathode Choice for Commercialization of Sodium-Ion Batteries: Layered Transition Metal Oxides versus Prussian Blue Analogs. Adv. Funct. Mater. 2020, 30 (14), 1909530 DOI: 10.1002/adfm.201909530Google ScholarThere is no corresponding record for this reference.
- 46He, M.; Mejdoubi, A. E. L.; Chartouni, D.; Morcrette, M.; Troendle, P.; Castiglioni, R. High power NVPF/HC-based sodium-ion batteries. J. Power Sources 2023, 588, 233741 DOI: 10.1016/j.jpowsour.2023.233741Google ScholarThere is no corresponding record for this reference.
- 47Yan, G.; Mariyappan, S.; Rousse, G.; Jacquet, Q.; Deschamps, M.; David, R.; Mirvaux, B.; Freeland, J. W.; Tarascon, J. M. Higher energy and safer sodium ion batteries via an electrochemically made disordered Na(3)V(2)(PO(4))(2)F(3) material. Nat. Commun. 2019, 10 (1), 585, DOI: 10.1038/s41467-019-08359-yGoogle ScholarThere is no corresponding record for this reference.
- 48Mao, Y.; Zhang, X.; Zhou, Y.; Chu, W. Microwave-assisted synthesis of porous nano-sized Na3V2(PO4)2F3@C nanospheres for sodium ion batteries with enhanced stability. Scripta Materialia 2020, 181, 92– 96, DOI: 10.1016/j.scriptamat.2020.02.023Google ScholarThere is no corresponding record for this reference.
- 49Zhang, J.; Zheng, T.; Guo, X.; Nguyen, H. Q.; Cheng, K.-w. E.; Lam, K.-H.; Rettenwander, D.; Jin, W.; Boles, S. T. Identifying Problematic Phase Transformations in Pb Foil Anodes for Sodium-Ion Batteries. J. Electrochem. Soc. 2024, 171 (9), 090516 DOI: 10.1149/1945-7111/ad76e0Google ScholarThere is no corresponding record for this reference.
- 50Park, J.; Han, J.; Gim, J.; Garcia, J.; Iddir, H.; Ahmed, S.; Xu, G.-L.; Amine, K.; Johnson, C.; Jung, Y. Evidence of Zintl Intermediate Phase and Its Impacts on Li and Na Storage Performance of Pb-Based Alloying Anodes. Chem. Mater. 2023, 35 (11), 4171– 4180, DOI: 10.1021/acs.chemmater.3c00061Google ScholarThere is no corresponding record for this reference.
- 51Mason, P. E.; Uhlig, F.; Vaněk, V.; Buttersack, T.; Bauerecker, S.; Jungwirth, P. Coulomb explosion during the early stages of the reaction of alkali metals with water. Nat. Chem. 2015, 7 (3), 250– 254, DOI: 10.1038/nchem.2161Google ScholarThere is no corresponding record for this reference.
- 52Au, H.; Alptekin, H.; Jensen, A. C. S.; Olsson, E.; O’Keefe, C. A.; Smith, T.; Crespo-Ribadeneyra, M.; Headen, T. F.; Grey, C. P.; Cai, Q. A revised mechanistic model for sodium insertion in hard carbons. Energy Environ. Sci. 2020, 13 (10), 3469– 3479, DOI: 10.1039/D0EE01363CGoogle ScholarThere is no corresponding record for this reference.
- 53Ponrouch, A.; Goñi, A. R.; Palacín, M. R. High capacity hard carbon anodes for sodium ion batteries in additive free electrolyte. Electrochem. Commun. 2013, 27, 85– 88, DOI: 10.1016/j.elecom.2012.10.038Google ScholarThere is no corresponding record for this reference.
- 54Ge, N.; Gao, W.; Gan, Y. Comparison of thermal stability of three sodium-ion battery electrolytes. J. Power Sources 2024, 623, 235377 DOI: 10.1016/j.jpowsour.2024.235377Google ScholarThere is no corresponding record for this reference.
- 55Zhou, X.; Yang, C.; Hu, W.; Han, J.; You, Y. Assessment of Thermal Safety for Organic Electrolytes: from Material to Cell Level. ACS Energy Letters 2025, 10 (7), 3143– 3150, DOI: 10.1021/acsenergylett.5c01147Google ScholarThere is no corresponding record for this reference.
- 56Wang, P.-F.; You, Y.; Yin, Y.-X.; Guo, Y.-G. An O3-type NaNi0.5Mn0.5O2 cathode for sodium-ion batteries with improved rate performance and cycling stability. Journal of Materials Chemistry A 2016, 4 (45), 17660– 17664, DOI: 10.1039/C6TA07589DGoogle ScholarThere is no corresponding record for this reference.
- 57Zhang, J.; Zheng, T.; Guo, X.; Nguyen, H. Q.; Cheng, K.-w. E.; Lam, K.-H.; Rettenwander, D.; Jin, W.; Boles, S. T. Identifying Problematic Phase Transformations in Pb Foil Anodes for Sodium-Ion Batteries. J. Electrochem. Soc. 2024, 171 (9), 090516 DOI: 10.1149/1945-7111/ad76e0Google ScholarThere is no corresponding record for this reference.
- 58Eshetu, G. G.; Grugeon, S.; Kim, H.; Jeong, S.; Wu, L.; Gachot, G.; Laruelle, S.; Armand, M.; Passerini, S. Comprehensive Insights into the Reactivity of Electrolytes Based on Sodium Ions. ChemSusChem 2016, 9 (5), 462– 471, DOI: 10.1002/cssc.201501605Google ScholarThere is no corresponding record for this reference.
- 59Samigullin, R. R.; Bobyleva, Z. V.; Zakharkin, M. V.; Zharikova, E. V.; Rozova, M. G.; Drozhzhin, O. A.; Antipov, E. V. On the Thermal Stability of Selected Electrode Materials and Electrolytes for Na-Ion Batteries. Energies 2024, 17 (16), 3970, DOI: 10.3390/en17163970Google ScholarThere is no corresponding record for this reference.
- 60Sarkar, S.; Karmakar, A.; Vishnugopi, B. S.; Jeevarajan, J. A.; Mukherjee, P. P. Electrode-electrolyte interactions dictate thermal stability of sodium-ion batteries. Chem. Commun. (Camb) 2024, 60 (88), 12868– 12871, DOI: 10.1039/D4CC03889DGoogle ScholarThere is no corresponding record for this reference.
- 61Hirsh, H. S.; Sayahpour, B.; Shen, A.; Li, W.; Lu, B.; Zhao, E.; Zhang, M.; Meng, Y. S. Role of electrolyte in stabilizing hard carbon as an anode for rechargeable sodium-ion batteries with long cycle life. Energy Storage Materials 2021, 42, 78– 87, DOI: 10.1016/j.ensm.2021.07.021Google ScholarThere is no corresponding record for this reference.
- 62Sarkar, S.; Lefler, M. J.; Vishnugopi, B. S.; Nuwayhid, R. B.; Love, C. T.; Carter, R.; Mukherjee, P. P. Fluorinated ethylene carbonate as additive to glyme electrolytes for robust sodium solid electrolyte interface. Cell Reports Physical Science 2023, 4 (4), 101356 DOI: 10.1016/j.xcrp.2023.101356Google ScholarThere is no corresponding record for this reference.
- 63Eshetu, G. G.; Elia, G. A.; Armand, M.; Forsyth, M.; Komaba, S.; Rojo, T.; Passerini, S. Electrolytes and Interphases in Sodium-Based Rechargeable Batteries: Recent Advances and Perspectives. Adv. Energy Mater. 2020, 10 (20), 2000093 DOI: 10.1002/aenm.202000093Google ScholarThere is no corresponding record for this reference.
- 64Cheng, F.; Hu, J.; Zhang, W.; Guo, B.; Yu, P.; Sun, X.; Peng, J. Reviving ether-based electrolytes for sodium-ion batteries. Energy Environ. Sci. 2025, 18, 6874– 6898, DOI: 10.1039/D5EE00725AGoogle ScholarThere is no corresponding record for this reference.
- 65Li, C.; Xu, H.; Ni, L.; Qin, B.; Ma, Y.; Jiang, H.; Xu, G.; Zhao, J.; Cui, G. Nonaqueous Liquid Electrolytes for Sodium-Ion Batteries: Fundamentals, Progress and Perspectives. Adv. Energy Mater. 2023, 13 (40), 2301758 DOI: 10.1002/aenm.202301758Google ScholarThere is no corresponding record for this reference.
- 66Sarkar, S.; Gonzalez-Malabet, H. J.; Flannagin, M.; L’Antigua, A.; Shevchenko, P. D.; Nelson, G. J.; Mukherjee, P. P. Multiscale Electrochemomechanics Interaction and Degradation Analytics of Sn Electrodes for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2022, 14 (26), 29711– 29721, DOI: 10.1021/acsami.2c02772Google ScholarThere is no corresponding record for this reference.
- 67Teufl, T.; Pritzl, D. J.; Hartmann, L.; Solchenbach, S.; Mendez, M.; Gasteiger, H. Implications of the Thermal Stability of FEC-Based Electrolytes for Li-Ion Batteries. J. Electrochem. Soc. 2023, 170 (2), 020531 DOI: 10.1149/1945-7111/acbc52Google ScholarThere is no corresponding record for this reference.
- 68Yang, Z.; Gewirth, A. A.; Trahey, L. Investigation of fluoroethylene carbonate effects on tin-based lithium-ion battery electrodes. ACS Appl. Mater. Interfaces 2015, 7 (12), 6557– 6566, DOI: 10.1021/am508593sGoogle ScholarThere is no corresponding record for this reference.
- 69Ying, H.; Han, W. Q. Metallic Sn-Based Anode Materials: Application in High-Performance Lithium-Ion and Sodium-Ion Batteries. Adv. Sci. (Weinh) 2017, 4 (11), 1700298 DOI: 10.1002/advs.201700298Google ScholarThere is no corresponding record for this reference.
- 70Cui, K.; Hou, R.; Zhou, H.; Guo, S. Electrolyte Engineering of Hard Carbon for Sodium-Ion Batteries: From Mechanism Analysis to Design Strategies. Adv. Funct. Mater. 2025, 35 (16), 2419275 DOI: 10.1002/adfm.202419275Google ScholarThere is no corresponding record for this reference.
- 71Fondard, J.; Irisarri, E.; Courrèges, C.; Palacin, M. R.; Ponrouch, A.; Dedryvère, R. SEI Composition on Hard Carbon in Na-Ion Batteries After Long Cycling: Influence of Salts (NaPF6, NaTFSI) and Additives (FEC, DMCF). J. Electrochem. Soc. 2020, 167 (7), 070526 DOI: 10.1149/1945-7111/ab75fdGoogle ScholarThere is no corresponding record for this reference.
- 72Kim, D.-H.; Kang, B.; Lee, H. Comparative study of fluoroethylene carbonate and succinic anhydride as electrolyte additive for hard carbon anodes of Na-ion batteries. J. Power Sources 2019, 423, 137– 143, DOI: 10.1016/j.jpowsour.2019.03.047Google ScholarThere is no corresponding record for this reference.
- 73Eshetu, G. G.; Grugeon, S.; Kim, H.; Jeong, S.; Wu, L.; Gachot, G.; Laruelle, S.; Armand, M.; Passerini, S. Comprehensive Insights into the Reactivity of Electrolytes Based on Sodium Ions. ChemSusChem 2016, 9 (5), 462– 471, DOI: 10.1002/cssc.201501605Google ScholarThere is no corresponding record for this reference.
- 74Mu, L.; Feng, X.; Kou, R.; Zhang, Y.; Guo, H.; Tian, C.; Sun, C.-J.; Du, X.-W.; Nordlund, D.; Xin, H. L.; Lin, F. Deciphering the Cathode–Electrolyte Interfacial Chemistry in Sodium Layered Cathode Materials. Adv. Energy Mater. 2018, 8 (34), 1801975 DOI: 10.1002/aenm.201801975Google ScholarThere is no corresponding record for this reference.
- 75Liang, X.; Song, X.; Sun, H. H.; Kim, H.; Kim, M. C.; Sun, Y. K. High-energy and long-life O3-type layered cathode material for sodium-ion batteries. Nat. Commun. 2025, 16 (1), 3505, DOI: 10.1038/s41467-025-58637-1Google ScholarThere is no corresponding record for this reference.
- 76Han, X.; Liu, Z.; Hu, X.; Huang, Q.; Zhang, D.; Guo, H.; Yi, Q. CEI Optimization: Enable the High Capacity and Reversible Sodium-Ion Batteries for Future Massive Energy Storage. Advanced Energy and Sustainability Research 2024, 5 (1), 2300166 DOI: 10.1002/aesr.202300166Google ScholarThere is no corresponding record for this reference.
- 77Xie, Y.; Xu, G.-L.; Che, H.; Wang, H.; Yang, K.; Yang, X.; Guo, F.; Ren, Y.; Chen, Z.; Amine, K. Probing Thermal and Chemical Stability of NaxNi1/3Fe1/3Mn1/3O2 Cathode Material toward Safe Sodium-Ion Batteries. Chem. Mater. 2018, 30 (15), 4909– 4918, DOI: 10.1021/acs.chemmater.8b00047Google ScholarThere is no corresponding record for this reference.
- 78Yang, C.; Xin, S.; Mai, L.; You, Y. Materials Design for High-Safety Sodium-Ion Battery. Adv. Energy Mater. 2021, 11 (2), 2000974 DOI: 10.1002/aenm.202000974Google ScholarThere is no corresponding record for this reference.
- 79Zheng, Z.; Wang, Y.; Zhang, Y.; Li, X.; Zhang, Y.; He, Y.-S.; Che, H.; Li, L.; Ma, Z.-F. Sodium iron sulfate cathodes with ultra-long cycle-life and high safety for sodium-ion batteries. Nano Energy 2024, 128, 109907 DOI: 10.1016/j.nanoen.2024.109907Google ScholarThere is no corresponding record for this reference.
- 80Zhang, L.; Tsolakidou, C.; Mariyappan, S.; Tarascon, J.-M.; Trabesinger, S. Unraveling gas evolution in sodium batteries by online electrochemical mass spectrometry. Energy Storage Materials 2021, 42, 12– 21, DOI: 10.1016/j.ensm.2021.07.005Google ScholarThere is no corresponding record for this reference.
- 81Kwon, B.; Schraiber, A.; Jeevarajan, J. A. Evaluating Fire and Smoke Risks with Lithium-Ion Cells, Modules, and Batteries. ACS Energy Letters 2024, 9 (11), 5319– 5328, DOI: 10.1021/acsenergylett.4c02480Google ScholarThere is no corresponding record for this reference.
- 82Bordes, A.; Marlair, G.; Zantman, A.; Chesnaye, A.; Lore, P.-A. L.; Lecocq, A. Safety Evaluation of a Sodium-Ion Cell: Assessment of Vent Gas Emissions under Thermal Runaway. ACS Energy Letters 2022, 7 (10), 3386– 3391, DOI: 10.1021/acsenergylett.2c01667Google ScholarThere is no corresponding record for this reference.
- 83Mohsin, I. U.; Hofmann, A.; Ziebert, C. Exploring the reactivity of Na3V2(PO4)3/C and hard carbon electrodes in sodium-ion batteries at various charge states. Electrochim. Acta 2024, 487, 144197 DOI: 10.1016/j.electacta.2024.144197Google ScholarThere is no corresponding record for this reference.
- 84Mohsin, I. U.; Schneider, L.; Häringer, M.; Ziebert, C.; Rohde, M.; Bauer, W.; Ehrenberg, H.; Seifert, H. J. Heat generation and degradation mechanisms studied on Na3V2(PO4)3/C positive electrode material in full pouch/coin cell assembly. J. Power Sources 2022, 545, 231901 DOI: 10.1016/j.jpowsour.2022.231901Google ScholarThere is no corresponding record for this reference.
- 85Samigullin, R.; Zakharkin, M.; Drozhzhin, O.; Antipov, E. Thermal Stability of NASICON-Type Na3V2(PO4)3 and Na4VMn(PO4)3 as Cathode Materials for Sodium-ion Batteries. Energies 2023, 16 (7), 3051, DOI: 10.3390/en16073051Google ScholarThere is no corresponding record for this reference.
- 86Samigullin, R. R.; Drozhzhin, O. A.; Antipov, E. V. Comparative Study of the Thermal Stability of Electrode Materials for Li-Ion and Na-Ion Batteries. ACS Applied Energy Materials 2022, 5 (1), 14– 19, DOI: 10.1021/acsaem.1c03151Google ScholarThere is no corresponding record for this reference.
- 87Yue, Y.; Jia, Z.; Li, Y.; Wen, Y.; Lei, Q.; Duan, Q.; Sun, J.; Wang, Q. Thermal runaway hazards comparison between sodium-ion and lithium-ion batteries using accelerating rate calorimetry. Process Safety and Environmental Protection 2024, 189, 61– 70, DOI: 10.1016/j.psep.2024.06.032Google ScholarThere is no corresponding record for this reference.
- 88Robinson, J. B.; Heenan, T. M. M.; Jervis, J. R.; Tan, C.; Kendrick, E.; Brett, D. J. L.; Shearing, P. R. Multiscale tomographic analysis of the thermal failure of Na-Ion batteries. J. Power Sources 2018, 400, 360– 368, DOI: 10.1016/j.jpowsour.2018.07.098Google ScholarThere is no corresponding record for this reference.
- 89He, M.; Mejdoubi, A. E. L.; Chartouni, D.; Morcrette, M.; Troendle, P.; Castiglioni, R. High power NVPF/HC-based sodium-ion batteries. J. Power Sources 2023, 588, 233741 DOI: 10.1016/j.jpowsour.2023.233741Google ScholarThere is no corresponding record for this reference.
- 90Bertrand, M.; Johnson, N. B.; Jin, L.; Bates, A. M.; Chartrand, P.; Torres-Castro, L.; Dollé, M. Unveiling the thermite-driven lithium fire ignition in solid-state batteries. Joule 2025, 9 (5), 101953 DOI: 10.1016/j.joule.2025.101953Google ScholarThere is no corresponding record for this reference.
- 91Mao, N.; Gadkari, S.; Wang, Z.; Zhang, T.; Bai, J.; Cai, Q. A comparative analysis of lithium-ion batteries with different cathodes under overheating and nail penetration conditions. Energy 2023, 278, 128027 DOI: 10.1016/j.energy.2023.128027Google ScholarThere is no corresponding record for this reference.
- 92Mohsin, I. U.; Schneider, L.; Häringer, M.; Ziebert, C.; Rohde, M.; Bauer, W.; Ehrenberg, H.; Seifert, H. J. Heat generation and degradation mechanisms studied on Na3V2(PO4)3/C positive electrode material in full pouch/coin cell assembly. J. Power Sources 2022, 545, 231901 DOI: 10.1016/j.jpowsour.2022.231901Google ScholarThere is no corresponding record for this reference.
- 93Mao, N.; Gadkari, S.; Wang, Z.; Zhang, T.; Bai, J.; Cai, Q. A comparative analysis of lithium-ion batteries with different cathodes under overheating and nail penetration conditions. Energy 2023, 278, 128027 DOI: 10.1016/j.energy.2023.128027Google ScholarThere is no corresponding record for this reference.
- 94Hasa, I.; Mariyappan, S.; Saurel, D.; Adelhelm, P.; Koposov, A. Y.; Masquelier, C.; Croguennec, L.; Casas-Cabanas, M. Challenges of today for Na-based batteries of the future: From materials to cell metrics. J. Power Sources 2021, 482, 228872 DOI: 10.1016/j.jpowsour.2020.228872Google ScholarThere is no corresponding record for this reference.
- 95Li, Z.; Cheng, Z.; Yu, Y.; Wang, J.; Wang, L.; Mei, W.; Wang, Q. Thermal runaway comparison and assessment between sodium-ion and lithium-ion batteries. Process Safety and Environmental Protection 2025, 193, 842– 855, DOI: 10.1016/j.psep.2024.11.118Google ScholarThere is no corresponding record for this reference.
- 96Rudola, A.; Sayers, R.; Wright, C. J.; Barker, J. Opportunities for moderate-range electric vehicles using sustainable sodium-ion batteries. Nature Energy 2023, 8 (3), 215– 218, DOI: 10.1038/s41560-023-01215-wGoogle ScholarThere is no corresponding record for this reference.
- 97Li, M. Elevating the Practical Application of Sodium-Ion Batteries through Advanced Characterization Studies on Cathodes. Energies 2023, 16 (24), 8004, DOI: 10.3390/en16248004Google ScholarThere is no corresponding record for this reference.
- 98Peters, J. F.; Peña Cruz, A.; Weil, M. Exploring the Economic Potential of Sodium-Ion Batteries. Batteries 2019, 5 (1), 10, DOI: 10.3390/batteries5010010Google ScholarThere is no corresponding record for this reference.
- 99Munjal, M.; Prein, T.; Ramadan, M. M.; Smith, H. B.; Venugopal, V.; Rupp, J. L. M.; Abate, I. I.; Olivetti, E. A.; Huang, K. J. Process Cost Analysis of Performance Challenges and Their Mitigations in Sodium-ion Battery Cathode Materials. Joule 2025, 9 (5), 101871 DOI: 10.1016/j.joule.2025.101871Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 3 publications.
- Pooja Ranganathan, Bairav S. Vishnugopi, Avijit Karmakar, Abhinanda Sengupta, Susmita Sarkar, Dhevathi Rajan Rajagopalan Kannan, Frederick Gray, Vinay Premnath, Wan Si Tang, Judith A. Jeevarajan, Partha P. Mukherjee. Role of Anode Composition and Electrolyte Interactions on the Thermo-Electrochemical Stability of Sodium-Ion Batteries. ACS Applied Materials & Interfaces 2026, 18
(6)
, 9670-9682. https://doi.org/10.1021/acsami.5c19946
- Chenran Zhang, Kaixuan Li, Jiali Wang, Yangtian Yan, Biao Zhang, Baohua Li, Dengyun Zhai, Feiyu Kang. Nanostructure and Interface Engineering of Graphite Anodes for High‐Performance Potassium‐Ion Batteries: Mechanisms, Strategies, and Perspectives. Small 2026, 16 https://doi.org/10.1002/smll.202511225
- Xu Zhu, Shuai Sun, Mengting Liu, Peng‐Fei Wang. Low‐Cost Layered Cathodes Toward Practical Sodium‐Ion Batteries: Scientific Challenges, Resolution Strategies, and Economic Efficiency. Advanced Energy Materials 2026, 8 https://doi.org/10.1002/aenm.202506411
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. (a) Historical timeline of sodium-ion battery (SIB) development, highlighting key discoveries in cathode and anode chemistries, along with the emergence of recent commercial and mass-manufacturing efforts. (b) Comparison of energy storage capacities of various cathode and anode materials. (6,23−25) (Electrochemical properties are provided in Table S1 in the Supporting Information.) (c) Comparison of key characteristics of sodium ions and lithium ions. (26−28) (d) Schematic overview of key considerations for developing practical SIBs.
Figure 2
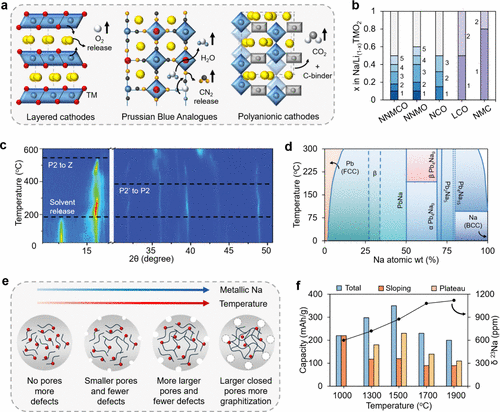
Figure 2. (a) Schematic illustration of typical thermal degradation pathways in sodium-ion cathodes upon heating, including layered, PBA, and polyanionic compounds. (b) Comparison of phase transitions during charging for layered sodium-ion cathodes: NaNi0.33Mn0.33Co0.33O2 (NNMCO), NaNi0.5Mn0.5O2 (NNMO), and NaCoO2 (NCO) with their lithium-ion counterparts, LiCoO2 (LCO) and LiNi0.33Mn0.33Co0.33O2 (NMC). Here, each number corresponds to different phases exhibited by the cathode, as provided in Table S2 of the Supporting Information. (37,38,56) (c) In situ XRD profile of Na0.8Ni0.33Mn0.67O2 (NNMO) during heating using X-ray beam of wavelength 0.154 nm (Reproduced with permission from ref (32). Copyright 2025 Wiley-VCH GmbH, Weinheim.) (d) Thermodynamic phase diagram of the Na-Pb binary alloy system. (Adapted from ref (57). Copyright 2024 Electrochemical Society (ECS) & IOP Science.) (e) Schematic showing the influence of hard carbon (HC) fabrication temperature on anode microstructure and sodium metal agglomeration. (f) Influence of fabrication temperature on the ion storage mechanism in HC. (Adapted from ref (52). Copyright 2020 Royal Society of Chemistry (RSC).)
Figure 3

Figure 3. (a) Comparison of thermal stability of the anode interphase for various salt–solvent combinations used in SIBs (additional details are provided in Table S3 of the Supporting Information). (59,73) (b) Schematic illustration showing the relationship between solvation energy and thermal stability of the anode interphase. (c) Spider chart depicting thermal stability characteristics of different Sn alloy anodes with and without FEC additives after cycling. (Adapted from ref (55). Copyright 2025 American Chemical Society (ACS).) Here μ-Sn and n-Sn refer to micro-Sn and nano-Sn, respectively. (d) SEM images of pristine and cycled n-Sn particles showing nonuniform interphase formation after cycling without additives. (Reproduced with permission from ref (66). Copyright 2022 American Chemical Society (ACS).) (e) Comparison of the effect of FEC on the CE of HC and Sn anodes. (66,72)
Figure 4

Figure 4. (a) Schematic illustration of typical thermal instability pathways of the CEI in layered oxide and PBA cathodes. (b) In situ XRD pattern of charged NFM in 1 M NaPF6 in propylene carbonate:ethyl methyl carbonate (PC:EMC) with 2 wt% FEC using an X-ray beam of wavelength 0.012 nm. (Reproduced with permission from ref (77). Copyright 2018 American Chemical Society (ACS).) (c) Comparative analysis of gas evolution normalized to volumetric energy density for NVPF and LFP cathodes. (Adapted from ref (82). Copyright 2022 American Chemical Society (ACS).) (d) Comparison of the thermal stability of different cathodes based on the relationship between the onset temperature for thermal degradation and cutoff voltage (figure numbers for the representative data sets are summarized in Table S4 of the Supporting Information). (e) Thermal stability profile of carbon-coated NVP as a function of SOC. (Adapted from ref (83). Copyright 2024 Elsevier.) (f) Schematic depiction of how increasing SOC in layered oxide cathodes influences degradation mechanisms and the severity of thermal runaway.
Figure 5
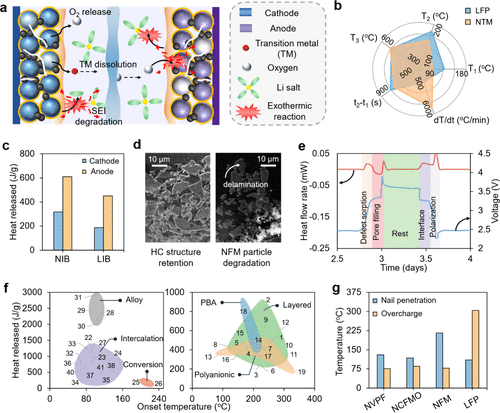
Figure 5. (a) Schematic illustration of the typical thermal runaway pathways in SIB cells. (b) Thermal safety characteristics in NaxTMO2 (NTM) and LFP cathodes. (Adapted from ref (87). Copyright 2024 Elsevier.) (c) Heat release comparison between LIB and SIB cells with cathode and anode contributions. (Adapted from ref (86). Copyright 2021 American Chemical Society (ACS).) (d) SEM micrographs of SIB cathode and anode after thermal abuse testing. (Reproduced with permission from ref (88). Copyright 2018 Elsevier.) (e) Microcalorimetry profile showing heat generation during a single charge/discharge cycle in a pristine NVP/HC full cell. (92) (f) Comparison of heat release during thermal abuse tests of different cathodes and anodes (more details regarding the electrode–electrolyte combination for this data set are provided in Table S5 in the Supporting Information). (g) Maximum temperatures observed during nail penetration and overcharge tests for SIBs using NVPF, NFM, and NCFMO cathodes, compared to LFP-based cells (8,89,93)
Figure 6
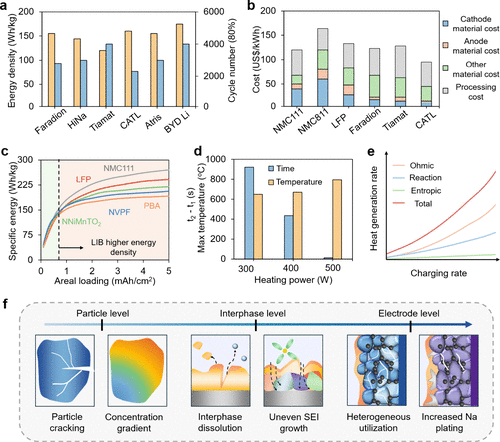
Figure 6. (a) Comparison of energy density and cycle life (at 80% capacity retention) of various SIB prototypes from different manufacturers relative to BYD’s lithium-ion blade battery. (Adapted from refs (96and97). Copyright 2023 Springer Nature and 2023 MDPI.) (b) Breakdown of material and processing costs for commercially available LIB and SIB systems. (Adapted from refs (98and99). Copyright 2019 MDPI and 2025 Cell Press.) (c) Specific energy comparison of different cathode materials as a function of areal loading. (Adapted from ref (94). Copyright 2021 Elsevier.) (d) Effect of heating power during thermal abuse tests on the maximum temperature (T3) and time interval between onset of self-heating and thermal runaway (t2 – t1) for layered oxide-based SIBs. (Adapted from ref (95). Copyright 2025 Elsevier.) (e) Representative illustration of heat generation as a function of charging rate. (f) Schematic showing the hierarchical set of factors (i.e., at the particle, interphase, and electrode levels) affecting SIB safety.
References
This article references 99 other publications.
- 1Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 Years of Lithium-Ion Batteries. Adv. Mater. 2018, 30 (33), e1800561 DOI: 10.1002/adma.201800561There is no corresponding record for this reference.
- 2Tarascon, J.-M. Na-ion versus Li-ion Batteries: Complementarity Rather than Competitiveness. Joule 2020, 4 (8), 1616– 1620, DOI: 10.1016/j.joule.2020.06.003There is no corresponding record for this reference.
- 3Tarascon, J. M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414 (6861), 359– 367, DOI: 10.1038/35104644There is no corresponding record for this reference.
- 4Hirsh, H. S.; Li, Y.; Tan, D. H. S.; Zhang, M.; Zhao, E.; Meng, Y. S. Sodium-Ion Batteries Paving the Way for Grid Energy Storage. Adv. Energy Mater. 2020, 10 (32), 2001274 DOI: 10.1002/aenm.202001274There is no corresponding record for this reference.
- 5Hwang, J. Y.; Myung, S. T.; Sun, Y. K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46 (12), 3529– 3614, DOI: 10.1039/C6CS00776GThere is no corresponding record for this reference.
- 6Usiskin, R.; Lu, Y.; Popovic, J.; Law, M.; Balaya, P.; Hu, Y.-S.; Maier, J. Fundamentals, status and promise of sodium-based batteries. Nature Reviews Materials 2021, 6 (11), 1020– 1035, DOI: 10.1038/s41578-021-00324-wThere is no corresponding record for this reference.
- 7Velumani, D.; Bansal, A. Thermal Behavior of Lithium- and Sodium-Ion Batteries: A Review on Heat Generation, Battery Degradation, Thermal Runway – Perspective and Future Directions. Energy Fuels 2022, 36 (23), 14000– 14029, DOI: 10.1021/acs.energyfuels.2c02889There is no corresponding record for this reference.
- 8Bates, A.; Torres-Castro, L.; Albertus, P.; Mukherjee, P. P.; Jeevarajan, J.; Johnson, N.; Bhargava, B.; Wang, Z.; Ogundipe, T.; Vishnugopi, B. S.; Karmakar, A.; Tang, W. S. Multi-scale safety evaluation of emerging battery technologies. ECS Interface 2024, 33 (3), 69, DOI: 10.1149/2.F10243IFThere is no corresponding record for this reference.
- 9Delmas, C. Sodium and Sodium-Ion Batteries: 50 Years of Research. Adv. Energy Mater. 2018, 8 (17), 1703137 DOI: 10.1002/aenm.201703137There is no corresponding record for this reference.
- 10Yu, T.; Li, G.; Duan, Y.; Wu, Y.; Zhang, T.; Zhao, X.; Luo, M.; Liu, Y. The research and industrialization progress and prospects of sodium ion battery. J. Alloys Compd. 2023, 958, 170486 DOI: 10.1016/j.jallcom.2023.170486There is no corresponding record for this reference.
- 11Yao, A.; Benson, S. M.; Chueh, W. C. Critically assessing sodium-ion technology roadmaps and scenarios for techno-economic competitiveness against lithium-ion batteries. Nature Energy 2025, 10 (3), 404– 416, DOI: 10.1038/s41560-024-01701-9There is no corresponding record for this reference.
- 12Han, M. H.; Gonzalo, E.; Singh, G.; Rojo, T. A comprehensive review of sodium layered oxides: powerful cathodes for Na-ion batteries. Energy Environ. Sci. 2015, 8 (1), 81– 102, DOI: 10.1039/C4EE03192JThere is no corresponding record for this reference.
- 13Gao, X.; Chen, J.; Liu, H.; Yin, S.; Tian, Y.; Cao, X.; Zou, G.; Hou, H.; Wei, W.; Chen, L. Copper-substituted NaxMO2 (M = Fe, Mn) cathodes for sodium ion batteries: Enhanced cycling stability through suppression of Mn(III) formation. Chemical Engineering Journal 2021, 406, 126830 DOI: 10.1016/j.cej.2020.126830There is no corresponding record for this reference.
- 14Subasinghe, L. U.; Satyanarayana Reddy, G.; Rudola, A.; Balaya, P. Analysis of Heat Generation and Impedance Characteristics of Prussian Blue Analogue Cathode-based 18650-type Sodium-ion Cells. J. Electrochem. Soc. 2020, 167 (11), 110504 DOI: 10.1149/1945-7111/ab9ee9There is no corresponding record for this reference.
- 15Dugas, R.; Zhang, B.; Rozier, P.; Tarascon, J. M. Optimization of Na-Ion Battery Systems Based on Polyanionic or Layered Positive Electrodes and Carbon Anodes. J. Electrochem. Soc. 2016, 163 (6), A867– A874, DOI: 10.1149/2.0051605jesThere is no corresponding record for this reference.
- 16Lee, D. H.; Xu, J.; Meng, Y. S. An advanced cathode for Na-ion batteries with high rate and excellent structural stability. Phys. Chem. Chem. Phys. 2013, 15 (9), 3304– 3312, DOI: 10.1039/c2cp44467dThere is no corresponding record for this reference.
- 17Mukherjee, S.; Bin Mujib, S.; Soares, D.; Singh, G. Electrode Materials for High-Performance Sodium-Ion Batteries. Materials (Basel) 2019, 12 (12), 1952, DOI: 10.3390/ma12121952There is no corresponding record for this reference.
- 18Fang, Y.; Yu, X.-Y.; Lou, X. W. Nanostructured Electrode Materials for Advanced Sodium-Ion Batteries. Matter 2019, 1 (1), 90– 114, DOI: 10.1016/j.matt.2019.05.007There is no corresponding record for this reference.
- 19Okoshi, M.; Yamada, Y.; Yamada, A.; Nakai, H. Theoretical Analysis on De-Solvation of Lithium, Sodium, and Magnesium Cations to Organic Electrolyte Solvents. J. Electrochem. Soc. 2013, 160 (11), A2160, DOI: 10.1149/2.074311jesThere is no corresponding record for this reference.
- 20Hu, Y.-S.; Pan, H. Solvation Structures in Electrolyte and the Interfacial Chemistry for Na-Ion Batteries. ACS Energy Letters 2022, 7 (12), 4501– 4503, DOI: 10.1021/acsenergylett.2c02529There is no corresponding record for this reference.
- 21Nayak, P. K.; Yang, L.; Brehm, W.; Adelhelm, P. From Lithium-Ion to Sodium-Ion Batteries: Advantages, Challenges, and Surprises. Angew. Chem., Int. Ed. Engl. 2018, 57 (1), 102– 120, DOI: 10.1002/anie.201703772There is no corresponding record for this reference.
- 22Slater, M. D.; Kim, D.; Lee, E.; Johnson, C. S. Sodium-Ion Batteries. Adv. Funct. Mater. 2013, 23 (8), 947– 958, DOI: 10.1002/adfm.201200691There is no corresponding record for this reference.
- 23Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46 (12), 3529– 3614, DOI: 10.1039/C6CS00776GThere is no corresponding record for this reference.
- 24Lyu, Y.; Liu, Y.; Yu, Z.-E.; Su, N.; Liu, Y.; Li, W.; Li, Q.; Guo, B.; Liu, B. Recent advances in high energy-density cathode materials for sodium-ion batteries. Sustainable Materials and Technologies 2019, 21, e00098 DOI: 10.1016/j.susmat.2019.e00098There is no corresponding record for this reference.
- 25Goikolea, E.; Palomares, V.; Wang, S.; de Larramendi, I. R.; Guo, X.; Wang, G.; Rojo, T. Na-Ion Batteries─Approaching Old and New Challenges. Adv. Energy Mater. 2020, 10 (44), 2002055 DOI: 10.1002/aenm.202002055There is no corresponding record for this reference.
- 26Islam, M. S.; Fisher, C. A. J. Lithium and sodium battery cathode materials: computational insights into voltage, diffusion and nanostructural properties. Chem. Soc. Rev. 2014, 43 (1), 185– 204, DOI: 10.1039/C3CS60199DThere is no corresponding record for this reference.
- 27Kim, S. C.; Gao, X.; Liao, S.-L.; Su, H.; Chen, Y.; Zhang, W.; Greenburg, L. C.; Pan, J.-A.; Zheng, X.; Ye, Y. Solvation-property relationship of lithium-sulphur battery electrolytes. Nat. Commun. 2024, 15 (1), 1268, DOI: 10.1038/s41467-023-44527-xThere is no corresponding record for this reference.
- 28Klein, F.; Jache, B.; Bhide, A.; Adelhelm, P. Conversion reactions for sodium-ion batteries. Phys. Chem. Chem. Phys. 2013, 15 (38), 15876– 15887, DOI: 10.1039/c3cp52125gThere is no corresponding record for this reference.
- 29Chen, C.; Han, Z.; Chen, S.; Qi, S.; Lan, X.; Zhang, C.; Chen, L.; Wang, P.; Wei, W. Core-Shell Layered Oxide Cathode for High-Performance Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2020, 12 (6), 7144– 7152, DOI: 10.1021/acsami.9b19260There is no corresponding record for this reference.
- 30Cui, Z.; Manthiram, A. Thermal Stability and Outgassing Behaviors of High-nickel Cathodes in Lithium-ion Batteries. Angew. Chem., Int. Ed. 2023, 62 (43), e202307243 DOI: 10.1002/anie.202307243There is no corresponding record for this reference.
- 31Deng, J.; Luo, W. B.; Lu, X.; Yao, Q.; Wang, Z.; Liu, H. K.; Zhou, H.; Dou, S. X. High Energy Density Sodium-Ion Battery with Industrially Feasible and Air-Stable O3-Type Layered Oxide Cathode. Adv. Energy Mater. 2018, 8 (5), 1701610 DOI: 10.1002/aenm.201701610There is no corresponding record for this reference.
- 32Li, Q.; Li, Y.; Liu, M.; Li, Y.; Zhao, H.; Ren, H.; Zhao, Y.; Zhou, Q.; Feng, X.; Shi, J. Elucidating Thermal Decomposition Kinetic Mechanism of Charged Layered Oxide Cathode for Sodium-Ion Batteries. Adv. Mater. 2025, 37 (10), e2415610 DOI: 10.1002/adma.202415610There is no corresponding record for this reference.
- 33Hu, E.; Wang, X.; Yu, X.; Yang, X.-Q. Probing the Complexities of Structural Changes in Layered Oxide Cathode Materials for Li-Ion Batteries during Fast Charge–Discharge Cycling and Heating. Acc. Chem. Res. 2018, 51 (2), 290– 298, DOI: 10.1021/acs.accounts.7b00506There is no corresponding record for this reference.
- 34Zuo, W.; Qiu, J.; Liu, X.; Ren, F.; Liu, H.; He, H.; Luo, C.; Li, J.; Ortiz, G. F.; Duan, H. The stability of P2-layered sodium transition metal oxides in ambient atmospheres. Nat. Commun. 2020, 11 (1), 3544, DOI: 10.1038/s41467-020-17290-6There is no corresponding record for this reference.
- 35Xiao, B.; Omenya, F.; Reed, D.; Li, X. A glance of the layered transition metal oxide cathodes in sodium and lithium-ion batteries: difference and similarities. Nanotechnology 2021, 32 (42), 422501 DOI: 10.1088/1361-6528/ac12ebThere is no corresponding record for this reference.
- 36Wang, P.-F.; You, Y.; Yin, Y.-X.; Guo, Y.-G. An O3-type NaNi0.5Mn0.5O2cathode for sodium-ion batteries with improved rate performance and cycling stability. Journal of Materials Chemistry A 2016, 4 (45), 17660– 17664, DOI: 10.1039/C6TA07589DThere is no corresponding record for this reference.
- 37Sathiya, M.; Hemalatha, K.; Ramesha, K.; Tarascon, J. M.; Prakash, A. S. Synthesis, Structure, and Electrochemical Properties of the Layered Sodium Insertion Cathode Material: NaNi1/3Mn1/3Co1/3O2. Chem. Mater. 2012, 24 (10), 1846– 1853, DOI: 10.1021/cm300466bThere is no corresponding record for this reference.
- 38Lei, Y.; Li, X.; Liu, L.; Ceder, G. Synthesis and Stoichiometry of Different Layered Sodium Cobalt Oxides. Chem. Mater. 2014, 26 (18), 5288– 5296, DOI: 10.1021/cm5021788There is no corresponding record for this reference.
- 39Sengupta, A.; Kumar, A.; Barik, G.; Ahuja, A.; Ghosh, J.; Lohani, H.; Kumari, P.; Bhandakkar, T. K.; Mitra, S. Lower Diffusion-Induced Stress in Nano-Crystallites of P2-Na(2/3) Ni(1/3) Mn(1/2) Ti(1/6) O(2) Novel Cathode for High Energy Na-ion Batteries. Small 2023, 19 (12), e2206248 DOI: 10.1002/smll.202206248There is no corresponding record for this reference.
- 40Sengupta, A.; Kumar, A.; Bano, A.; Ahuja, A.; Lohani, H.; Akella, S. H.; Kumari, P.; Noked, M.; Major, D. T.; Mitra, S. Unleashing the impact of Nb-doped, single crystal, cobalt-free P2-type Na0.67Ni0.33Mn0.67O2 on elevating the cycle life of sodium-ion batteries. Energy Storage Materials 2024, 69, 103435 DOI: 10.1016/j.ensm.2024.103435There is no corresponding record for this reference.
- 41Guo, S.; Li, Q.; Liu, P.; Chen, M.; Zhou, H. Environmentally stable interface of layered oxide cathodes for sodium-ion batteries. Nat. Commun. 2017, 8 (1), 135, DOI: 10.1038/s41467-017-00157-8There is no corresponding record for this reference.
- 42You, Y.; Dolocan, A.; Li, W.; Manthiram, A. Understanding the Air-Exposure Degradation Chemistry at a Nanoscale of Layered Oxide Cathodes for Sodium-Ion Batteries. Nano Lett. 2019, 19 (1), 182– 188, DOI: 10.1021/acs.nanolett.8b03637There is no corresponding record for this reference.
- 43Xiao, Y.; Xiao, J.; Zhao, H.; Li, J.; Zhang, G.; Zhang, D.; Guo, X.; Gao, H.; Wang, Y.; Chen, J. Prussian Blue Analogues for Sodium-Ion Battery Cathodes: A Review of Mechanistic Insights, Current Challenges, and Future Pathways. Small 2024, 20 (35), e2401957 DOI: 10.1002/smll.202401957There is no corresponding record for this reference.
- 44Wang, W.; Gang, Y.; Peng, J.; Hu, Z.; Yan, Z.; Lai, W.; Zhu, Y.; Appadoo, D.; Ye, M.; Cao, Y. Effect of Eliminating Water in Prussian Blue Cathode for Sodium-Ion Batteries. Adv. Funct. Mater. 2022, 32 (25), 2111727 DOI: 10.1002/adfm.202111727There is no corresponding record for this reference.
- 45Liu, Q.; Hu, Z.; Chen, M.; Zou, C.; Jin, H.; Wang, S.; Chou, S. L.; Liu, Y.; Dou, S. X. The Cathode Choice for Commercialization of Sodium-Ion Batteries: Layered Transition Metal Oxides versus Prussian Blue Analogs. Adv. Funct. Mater. 2020, 30 (14), 1909530 DOI: 10.1002/adfm.201909530There is no corresponding record for this reference.
- 46He, M.; Mejdoubi, A. E. L.; Chartouni, D.; Morcrette, M.; Troendle, P.; Castiglioni, R. High power NVPF/HC-based sodium-ion batteries. J. Power Sources 2023, 588, 233741 DOI: 10.1016/j.jpowsour.2023.233741There is no corresponding record for this reference.
- 47Yan, G.; Mariyappan, S.; Rousse, G.; Jacquet, Q.; Deschamps, M.; David, R.; Mirvaux, B.; Freeland, J. W.; Tarascon, J. M. Higher energy and safer sodium ion batteries via an electrochemically made disordered Na(3)V(2)(PO(4))(2)F(3) material. Nat. Commun. 2019, 10 (1), 585, DOI: 10.1038/s41467-019-08359-yThere is no corresponding record for this reference.
- 48Mao, Y.; Zhang, X.; Zhou, Y.; Chu, W. Microwave-assisted synthesis of porous nano-sized Na3V2(PO4)2F3@C nanospheres for sodium ion batteries with enhanced stability. Scripta Materialia 2020, 181, 92– 96, DOI: 10.1016/j.scriptamat.2020.02.023There is no corresponding record for this reference.
- 49Zhang, J.; Zheng, T.; Guo, X.; Nguyen, H. Q.; Cheng, K.-w. E.; Lam, K.-H.; Rettenwander, D.; Jin, W.; Boles, S. T. Identifying Problematic Phase Transformations in Pb Foil Anodes for Sodium-Ion Batteries. J. Electrochem. Soc. 2024, 171 (9), 090516 DOI: 10.1149/1945-7111/ad76e0There is no corresponding record for this reference.
- 50Park, J.; Han, J.; Gim, J.; Garcia, J.; Iddir, H.; Ahmed, S.; Xu, G.-L.; Amine, K.; Johnson, C.; Jung, Y. Evidence of Zintl Intermediate Phase and Its Impacts on Li and Na Storage Performance of Pb-Based Alloying Anodes. Chem. Mater. 2023, 35 (11), 4171– 4180, DOI: 10.1021/acs.chemmater.3c00061There is no corresponding record for this reference.
- 51Mason, P. E.; Uhlig, F.; Vaněk, V.; Buttersack, T.; Bauerecker, S.; Jungwirth, P. Coulomb explosion during the early stages of the reaction of alkali metals with water. Nat. Chem. 2015, 7 (3), 250– 254, DOI: 10.1038/nchem.2161There is no corresponding record for this reference.
- 52Au, H.; Alptekin, H.; Jensen, A. C. S.; Olsson, E.; O’Keefe, C. A.; Smith, T.; Crespo-Ribadeneyra, M.; Headen, T. F.; Grey, C. P.; Cai, Q. A revised mechanistic model for sodium insertion in hard carbons. Energy Environ. Sci. 2020, 13 (10), 3469– 3479, DOI: 10.1039/D0EE01363CThere is no corresponding record for this reference.
- 53Ponrouch, A.; Goñi, A. R.; Palacín, M. R. High capacity hard carbon anodes for sodium ion batteries in additive free electrolyte. Electrochem. Commun. 2013, 27, 85– 88, DOI: 10.1016/j.elecom.2012.10.038There is no corresponding record for this reference.
- 54Ge, N.; Gao, W.; Gan, Y. Comparison of thermal stability of three sodium-ion battery electrolytes. J. Power Sources 2024, 623, 235377 DOI: 10.1016/j.jpowsour.2024.235377There is no corresponding record for this reference.
- 55Zhou, X.; Yang, C.; Hu, W.; Han, J.; You, Y. Assessment of Thermal Safety for Organic Electrolytes: from Material to Cell Level. ACS Energy Letters 2025, 10 (7), 3143– 3150, DOI: 10.1021/acsenergylett.5c01147There is no corresponding record for this reference.
- 56Wang, P.-F.; You, Y.; Yin, Y.-X.; Guo, Y.-G. An O3-type NaNi0.5Mn0.5O2 cathode for sodium-ion batteries with improved rate performance and cycling stability. Journal of Materials Chemistry A 2016, 4 (45), 17660– 17664, DOI: 10.1039/C6TA07589DThere is no corresponding record for this reference.
- 57Zhang, J.; Zheng, T.; Guo, X.; Nguyen, H. Q.; Cheng, K.-w. E.; Lam, K.-H.; Rettenwander, D.; Jin, W.; Boles, S. T. Identifying Problematic Phase Transformations in Pb Foil Anodes for Sodium-Ion Batteries. J. Electrochem. Soc. 2024, 171 (9), 090516 DOI: 10.1149/1945-7111/ad76e0There is no corresponding record for this reference.
- 58Eshetu, G. G.; Grugeon, S.; Kim, H.; Jeong, S.; Wu, L.; Gachot, G.; Laruelle, S.; Armand, M.; Passerini, S. Comprehensive Insights into the Reactivity of Electrolytes Based on Sodium Ions. ChemSusChem 2016, 9 (5), 462– 471, DOI: 10.1002/cssc.201501605There is no corresponding record for this reference.
- 59Samigullin, R. R.; Bobyleva, Z. V.; Zakharkin, M. V.; Zharikova, E. V.; Rozova, M. G.; Drozhzhin, O. A.; Antipov, E. V. On the Thermal Stability of Selected Electrode Materials and Electrolytes for Na-Ion Batteries. Energies 2024, 17 (16), 3970, DOI: 10.3390/en17163970There is no corresponding record for this reference.
- 60Sarkar, S.; Karmakar, A.; Vishnugopi, B. S.; Jeevarajan, J. A.; Mukherjee, P. P. Electrode-electrolyte interactions dictate thermal stability of sodium-ion batteries. Chem. Commun. (Camb) 2024, 60 (88), 12868– 12871, DOI: 10.1039/D4CC03889DThere is no corresponding record for this reference.
- 61Hirsh, H. S.; Sayahpour, B.; Shen, A.; Li, W.; Lu, B.; Zhao, E.; Zhang, M.; Meng, Y. S. Role of electrolyte in stabilizing hard carbon as an anode for rechargeable sodium-ion batteries with long cycle life. Energy Storage Materials 2021, 42, 78– 87, DOI: 10.1016/j.ensm.2021.07.021There is no corresponding record for this reference.
- 62Sarkar, S.; Lefler, M. J.; Vishnugopi, B. S.; Nuwayhid, R. B.; Love, C. T.; Carter, R.; Mukherjee, P. P. Fluorinated ethylene carbonate as additive to glyme electrolytes for robust sodium solid electrolyte interface. Cell Reports Physical Science 2023, 4 (4), 101356 DOI: 10.1016/j.xcrp.2023.101356There is no corresponding record for this reference.
- 63Eshetu, G. G.; Elia, G. A.; Armand, M.; Forsyth, M.; Komaba, S.; Rojo, T.; Passerini, S. Electrolytes and Interphases in Sodium-Based Rechargeable Batteries: Recent Advances and Perspectives. Adv. Energy Mater. 2020, 10 (20), 2000093 DOI: 10.1002/aenm.202000093There is no corresponding record for this reference.
- 64Cheng, F.; Hu, J.; Zhang, W.; Guo, B.; Yu, P.; Sun, X.; Peng, J. Reviving ether-based electrolytes for sodium-ion batteries. Energy Environ. Sci. 2025, 18, 6874– 6898, DOI: 10.1039/D5EE00725AThere is no corresponding record for this reference.
- 65Li, C.; Xu, H.; Ni, L.; Qin, B.; Ma, Y.; Jiang, H.; Xu, G.; Zhao, J.; Cui, G. Nonaqueous Liquid Electrolytes for Sodium-Ion Batteries: Fundamentals, Progress and Perspectives. Adv. Energy Mater. 2023, 13 (40), 2301758 DOI: 10.1002/aenm.202301758There is no corresponding record for this reference.
- 66Sarkar, S.; Gonzalez-Malabet, H. J.; Flannagin, M.; L’Antigua, A.; Shevchenko, P. D.; Nelson, G. J.; Mukherjee, P. P. Multiscale Electrochemomechanics Interaction and Degradation Analytics of Sn Electrodes for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2022, 14 (26), 29711– 29721, DOI: 10.1021/acsami.2c02772There is no corresponding record for this reference.
- 67Teufl, T.; Pritzl, D. J.; Hartmann, L.; Solchenbach, S.; Mendez, M.; Gasteiger, H. Implications of the Thermal Stability of FEC-Based Electrolytes for Li-Ion Batteries. J. Electrochem. Soc. 2023, 170 (2), 020531 DOI: 10.1149/1945-7111/acbc52There is no corresponding record for this reference.
- 68Yang, Z.; Gewirth, A. A.; Trahey, L. Investigation of fluoroethylene carbonate effects on tin-based lithium-ion battery electrodes. ACS Appl. Mater. Interfaces 2015, 7 (12), 6557– 6566, DOI: 10.1021/am508593sThere is no corresponding record for this reference.
- 69Ying, H.; Han, W. Q. Metallic Sn-Based Anode Materials: Application in High-Performance Lithium-Ion and Sodium-Ion Batteries. Adv. Sci. (Weinh) 2017, 4 (11), 1700298 DOI: 10.1002/advs.201700298There is no corresponding record for this reference.
- 70Cui, K.; Hou, R.; Zhou, H.; Guo, S. Electrolyte Engineering of Hard Carbon for Sodium-Ion Batteries: From Mechanism Analysis to Design Strategies. Adv. Funct. Mater. 2025, 35 (16), 2419275 DOI: 10.1002/adfm.202419275There is no corresponding record for this reference.
- 71Fondard, J.; Irisarri, E.; Courrèges, C.; Palacin, M. R.; Ponrouch, A.; Dedryvère, R. SEI Composition on Hard Carbon in Na-Ion Batteries After Long Cycling: Influence of Salts (NaPF6, NaTFSI) and Additives (FEC, DMCF). J. Electrochem. Soc. 2020, 167 (7), 070526 DOI: 10.1149/1945-7111/ab75fdThere is no corresponding record for this reference.
- 72Kim, D.-H.; Kang, B.; Lee, H. Comparative study of fluoroethylene carbonate and succinic anhydride as electrolyte additive for hard carbon anodes of Na-ion batteries. J. Power Sources 2019, 423, 137– 143, DOI: 10.1016/j.jpowsour.2019.03.047There is no corresponding record for this reference.
- 73Eshetu, G. G.; Grugeon, S.; Kim, H.; Jeong, S.; Wu, L.; Gachot, G.; Laruelle, S.; Armand, M.; Passerini, S. Comprehensive Insights into the Reactivity of Electrolytes Based on Sodium Ions. ChemSusChem 2016, 9 (5), 462– 471, DOI: 10.1002/cssc.201501605There is no corresponding record for this reference.
- 74Mu, L.; Feng, X.; Kou, R.; Zhang, Y.; Guo, H.; Tian, C.; Sun, C.-J.; Du, X.-W.; Nordlund, D.; Xin, H. L.; Lin, F. Deciphering the Cathode–Electrolyte Interfacial Chemistry in Sodium Layered Cathode Materials. Adv. Energy Mater. 2018, 8 (34), 1801975 DOI: 10.1002/aenm.201801975There is no corresponding record for this reference.
- 75Liang, X.; Song, X.; Sun, H. H.; Kim, H.; Kim, M. C.; Sun, Y. K. High-energy and long-life O3-type layered cathode material for sodium-ion batteries. Nat. Commun. 2025, 16 (1), 3505, DOI: 10.1038/s41467-025-58637-1There is no corresponding record for this reference.
- 76Han, X.; Liu, Z.; Hu, X.; Huang, Q.; Zhang, D.; Guo, H.; Yi, Q. CEI Optimization: Enable the High Capacity and Reversible Sodium-Ion Batteries for Future Massive Energy Storage. Advanced Energy and Sustainability Research 2024, 5 (1), 2300166 DOI: 10.1002/aesr.202300166There is no corresponding record for this reference.
- 77Xie, Y.; Xu, G.-L.; Che, H.; Wang, H.; Yang, K.; Yang, X.; Guo, F.; Ren, Y.; Chen, Z.; Amine, K. Probing Thermal and Chemical Stability of NaxNi1/3Fe1/3Mn1/3O2 Cathode Material toward Safe Sodium-Ion Batteries. Chem. Mater. 2018, 30 (15), 4909– 4918, DOI: 10.1021/acs.chemmater.8b00047There is no corresponding record for this reference.
- 78Yang, C.; Xin, S.; Mai, L.; You, Y. Materials Design for High-Safety Sodium-Ion Battery. Adv. Energy Mater. 2021, 11 (2), 2000974 DOI: 10.1002/aenm.202000974There is no corresponding record for this reference.
- 79Zheng, Z.; Wang, Y.; Zhang, Y.; Li, X.; Zhang, Y.; He, Y.-S.; Che, H.; Li, L.; Ma, Z.-F. Sodium iron sulfate cathodes with ultra-long cycle-life and high safety for sodium-ion batteries. Nano Energy 2024, 128, 109907 DOI: 10.1016/j.nanoen.2024.109907There is no corresponding record for this reference.
- 80Zhang, L.; Tsolakidou, C.; Mariyappan, S.; Tarascon, J.-M.; Trabesinger, S. Unraveling gas evolution in sodium batteries by online electrochemical mass spectrometry. Energy Storage Materials 2021, 42, 12– 21, DOI: 10.1016/j.ensm.2021.07.005There is no corresponding record for this reference.
- 81Kwon, B.; Schraiber, A.; Jeevarajan, J. A. Evaluating Fire and Smoke Risks with Lithium-Ion Cells, Modules, and Batteries. ACS Energy Letters 2024, 9 (11), 5319– 5328, DOI: 10.1021/acsenergylett.4c02480There is no corresponding record for this reference.
- 82Bordes, A.; Marlair, G.; Zantman, A.; Chesnaye, A.; Lore, P.-A. L.; Lecocq, A. Safety Evaluation of a Sodium-Ion Cell: Assessment of Vent Gas Emissions under Thermal Runaway. ACS Energy Letters 2022, 7 (10), 3386– 3391, DOI: 10.1021/acsenergylett.2c01667There is no corresponding record for this reference.
- 83Mohsin, I. U.; Hofmann, A.; Ziebert, C. Exploring the reactivity of Na3V2(PO4)3/C and hard carbon electrodes in sodium-ion batteries at various charge states. Electrochim. Acta 2024, 487, 144197 DOI: 10.1016/j.electacta.2024.144197There is no corresponding record for this reference.
- 84Mohsin, I. U.; Schneider, L.; Häringer, M.; Ziebert, C.; Rohde, M.; Bauer, W.; Ehrenberg, H.; Seifert, H. J. Heat generation and degradation mechanisms studied on Na3V2(PO4)3/C positive electrode material in full pouch/coin cell assembly. J. Power Sources 2022, 545, 231901 DOI: 10.1016/j.jpowsour.2022.231901There is no corresponding record for this reference.
- 85Samigullin, R.; Zakharkin, M.; Drozhzhin, O.; Antipov, E. Thermal Stability of NASICON-Type Na3V2(PO4)3 and Na4VMn(PO4)3 as Cathode Materials for Sodium-ion Batteries. Energies 2023, 16 (7), 3051, DOI: 10.3390/en16073051There is no corresponding record for this reference.
- 86Samigullin, R. R.; Drozhzhin, O. A.; Antipov, E. V. Comparative Study of the Thermal Stability of Electrode Materials for Li-Ion and Na-Ion Batteries. ACS Applied Energy Materials 2022, 5 (1), 14– 19, DOI: 10.1021/acsaem.1c03151There is no corresponding record for this reference.
- 87Yue, Y.; Jia, Z.; Li, Y.; Wen, Y.; Lei, Q.; Duan, Q.; Sun, J.; Wang, Q. Thermal runaway hazards comparison between sodium-ion and lithium-ion batteries using accelerating rate calorimetry. Process Safety and Environmental Protection 2024, 189, 61– 70, DOI: 10.1016/j.psep.2024.06.032There is no corresponding record for this reference.
- 88Robinson, J. B.; Heenan, T. M. M.; Jervis, J. R.; Tan, C.; Kendrick, E.; Brett, D. J. L.; Shearing, P. R. Multiscale tomographic analysis of the thermal failure of Na-Ion batteries. J. Power Sources 2018, 400, 360– 368, DOI: 10.1016/j.jpowsour.2018.07.098There is no corresponding record for this reference.
- 89He, M.; Mejdoubi, A. E. L.; Chartouni, D.; Morcrette, M.; Troendle, P.; Castiglioni, R. High power NVPF/HC-based sodium-ion batteries. J. Power Sources 2023, 588, 233741 DOI: 10.1016/j.jpowsour.2023.233741There is no corresponding record for this reference.
- 90Bertrand, M.; Johnson, N. B.; Jin, L.; Bates, A. M.; Chartrand, P.; Torres-Castro, L.; Dollé, M. Unveiling the thermite-driven lithium fire ignition in solid-state batteries. Joule 2025, 9 (5), 101953 DOI: 10.1016/j.joule.2025.101953There is no corresponding record for this reference.
- 91Mao, N.; Gadkari, S.; Wang, Z.; Zhang, T.; Bai, J.; Cai, Q. A comparative analysis of lithium-ion batteries with different cathodes under overheating and nail penetration conditions. Energy 2023, 278, 128027 DOI: 10.1016/j.energy.2023.128027There is no corresponding record for this reference.
- 92Mohsin, I. U.; Schneider, L.; Häringer, M.; Ziebert, C.; Rohde, M.; Bauer, W.; Ehrenberg, H.; Seifert, H. J. Heat generation and degradation mechanisms studied on Na3V2(PO4)3/C positive electrode material in full pouch/coin cell assembly. J. Power Sources 2022, 545, 231901 DOI: 10.1016/j.jpowsour.2022.231901There is no corresponding record for this reference.
- 93Mao, N.; Gadkari, S.; Wang, Z.; Zhang, T.; Bai, J.; Cai, Q. A comparative analysis of lithium-ion batteries with different cathodes under overheating and nail penetration conditions. Energy 2023, 278, 128027 DOI: 10.1016/j.energy.2023.128027There is no corresponding record for this reference.
- 94Hasa, I.; Mariyappan, S.; Saurel, D.; Adelhelm, P.; Koposov, A. Y.; Masquelier, C.; Croguennec, L.; Casas-Cabanas, M. Challenges of today for Na-based batteries of the future: From materials to cell metrics. J. Power Sources 2021, 482, 228872 DOI: 10.1016/j.jpowsour.2020.228872There is no corresponding record for this reference.
- 95Li, Z.; Cheng, Z.; Yu, Y.; Wang, J.; Wang, L.; Mei, W.; Wang, Q. Thermal runaway comparison and assessment between sodium-ion and lithium-ion batteries. Process Safety and Environmental Protection 2025, 193, 842– 855, DOI: 10.1016/j.psep.2024.11.118There is no corresponding record for this reference.
- 96Rudola, A.; Sayers, R.; Wright, C. J.; Barker, J. Opportunities for moderate-range electric vehicles using sustainable sodium-ion batteries. Nature Energy 2023, 8 (3), 215– 218, DOI: 10.1038/s41560-023-01215-wThere is no corresponding record for this reference.
- 97Li, M. Elevating the Practical Application of Sodium-Ion Batteries through Advanced Characterization Studies on Cathodes. Energies 2023, 16 (24), 8004, DOI: 10.3390/en16248004There is no corresponding record for this reference.
- 98Peters, J. F.; Peña Cruz, A.; Weil, M. Exploring the Economic Potential of Sodium-Ion Batteries. Batteries 2019, 5 (1), 10, DOI: 10.3390/batteries5010010There is no corresponding record for this reference.
- 99Munjal, M.; Prein, T.; Ramadan, M. M.; Smith, H. B.; Venugopal, V.; Rupp, J. L. M.; Abate, I. I.; Olivetti, E. A.; Huang, K. J. Process Cost Analysis of Performance Challenges and Their Mitigations in Sodium-ion Battery Cathode Materials. Joule 2025, 9 (5), 101871 DOI: 10.1016/j.joule.2025.101871There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c02345.
Summary of electrochemical properties of sodium-ion cathodes and anodes (Table S1), Comparison of phase behavior of different sodium-ion and lithium-ion layered oxides (Table S2), Exothermic parameters of different sodium salt-solvent combinations with the salt concentration of 1 M (Table S3), Thermal stability of different sodium-ion cathodes as a function of cutoff voltages (Table S4), Typical electrode–electrolyte configurations used in thermal stability investigations of sodium-ion batteries (Table S5), Properties of commonly used solvents in sodium-ion batteries (Table S6) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



