Polar Opposites: Ligand-Mediated Polarity Inversion for Perovskite Quantum Dots with Sub-Nanometer Ligand ShellsClick to copy article linkArticle link copied!
- Fei HeFei HeChair for Photonics and Optoelectronics, Nano-Institute Munich, Department of Physics, Ludwig-Maximilians-Universität (LMU), Königinstraße 10, 80539 Munich, GermanyMore by Fei He
- Lena S. StickelLena S. StickelChair for Photonics and Optoelectronics, Nano-Institute Munich, Department of Physics, Ludwig-Maximilians-Universität (LMU), Königinstraße 10, 80539 Munich, GermanyMore by Lena S. Stickel
- Markus DöblingerMarkus DöblingerDepartment of Chemistry, Ludwig-Maximilians-Universität München, Butenandtstr. 5-13 (E), 81377 Munich, GermanyMore by Markus Döblinger
- Quinten A. Akkerman*Quinten A. Akkerman*[email protected]Chair for Photonics and Optoelectronics, Nano-Institute Munich, Department of Physics, Ludwig-Maximilians-Universität (LMU), Königinstraße 10, 80539 Munich, GermanyMore by Quinten A. Akkerman
Abstract
Perovskite quantum dots are promising classical and quantum light emitters but have limited chemical and colloidal stability in polar solvents due to their highly ionic lattices. In this work we demonstrate that perovskite quantum dots capped with strongly binding Gemini ligands with thin 0.7 nm hydroxyl-terminated ligands can be colloidally dispersed in polar solvents like ethanol.
This publication is licensed for personal use by The American Chemical Society.
Perovskite nanocrystals (pNCs) and quantum dots (pQDs) are exciting bright emitters with tremendous potential for efficient and affordable classical and quantum light. (1,2) Although promising, their ionic lattice makes it challenging to process them in polar solvents, leading to rapid dissolution and degradation. To solve this problem, several strategies have been developed (see Supporting Information (SI), Table SI1). For instance, encapsulating pNCs in polymeric ligand shells (like Solutol, polystyrene or polystyrene-block-poly(acrylic acid)) (3−7) results in large shells in the range of 10–100 nm, allowing the pNCs to be stable in polar solvents. However, these thick polymeric ligand shells often sacrifice colloidal stability and render the pNCs nonconductive, thus hindering the formation of dense and conductive QD films. An alternative strategy is encapsulating pNCs in inorganic shells like silica or aluminum oxide. (8−11) In most cases, these shells are often several nanometers thick, but recent encapsulation of alumina-gel-coated NCs demonstrated pQDs with thin shells of <0.5 nm, dispersible in polar solvents like butanol. (11) However, such inorganic shells require anhydrous and inert synthesis conditions, as metallo-organic precursors (like aluminum ethoxide) are moisture sensitive. This limits the practical use of such inorganic shells and opposes the promise of pQDs being cheap and scalable light emitters. However, strategies developed for conventional QDs, like molecular inorganic ligands, (12) will not work, as the ionic pQDs still require protection from the polar solvents due to their high ionic solubility. Thus, for pQDs to be dispersible in polar solvents using only ligands, we must develop new ligand chemistry tailored for pQDs.
Here, we demonstrate that pQDs can be dispersed in polar solvents like ethanol (EtOH) when capped with dicationic Gemini ligands terminated with hydroxyl groups. These ligands act as a polar membrane, with the dicationic ammonium groups binding to the pQDs, the alkyl chains providing an apolar barrier, and the hydroxyl heads providing a polar outer layer (see TOC). This strategy works for pQDs, as the hydroxyl groups have no affinity to the pQD surface, ensuring a unilateral binding of the ligands. Such ligands result in thin ligand shells of 0.7 nm and colloidally and chemically stable pQDs. Together with the pQDs’ size and composition tunability, and the range of green polar solvents to stabilize them in, it creates new processing, photocatalysis and electrochemistry opportunities for pQDs.
We first synthesized CsPbBr3 QDs with apolar ligands, (13) using Gemini bromide ligands with dodecyl tails, as these are short enough for efficient perovskite LEDs, (14) and a hexyl linker (referred to as 12–6–12 Gemini, Figure 1a and SI1), which has been proven to stabilize pNCs. (15) These ligands are ideal for CsPbBr3 QDs, as the dicationic quaternary ammonium groups strongly bind to the perovskite surface while the Br– aids in trap passivation. (15) As shown in Figure 1a, this results in pQDs that are dispersible in apolar solvents (toluene). The pQDs’ monodispersity is supported by High-Angle Annular Dark-Field Scanning Transmission Electron Microscopy (HAADF-STEM, Figures 1b and SI2), giving a size of 7.1 nm. These apolar pQDs exhibit a high photoluminescence quantum yield (PLQY) of 73% . To switch the pQDs’ polarity, we synthesized 12–6–12 Gemini ligands with hydroxyl terminated tails (12–6–12-OH Gemini, Figures 1c and SI3). The 12–6–12-OH Gemini-coated pQDs, now exhibiting a polar surface, can be dispersed in polar solvents like EtOH. These polar pQDs exhibit the same absorption spectrum, PLQY (74%) and size as their apolar counterparts (Figures1c,d and SI4). The high PLQY’s are also supported by a fast, single exponential 6.0 ns PL lifetime (Figure SI5). Nuclear Magnetic Resonance spectroscopy (NMR) on dissolved pQDs showed no residual apolar ligands after synthesis (Figure SI6) and a rough ligand coverage of 1 ligand/nm2, matching with half of the reported 1.2–1.7 ligands/nm2 for monodentate ligands, see SI and Figure SI7. (16) The EtOH-dispersed pQDs exhibit remarkable long-term chemical and colloidal stability in solution, with no degradation after two months (Figure SI8). Polar QD films fully preserve the absorption and single exponential PL lifetime of 6.1 ns, and exhibit good PL stability under continuous excitation (Figure SI9). The change in the pQDs’ polarity is supported by a biphasic mixture of mineral oil and EtOH (see TOC and Figure SI10); the apolar QDs reside exclusively in the mineral oil, and the polar pQDs in the EtOH layer.
Figure 1
Figure 1. Polar and apolar Gemini caped CsPbBr3 QDs. (a) Absorption and emission spectra of apolar 12–6–12 Gemini pQDs in toluene, with (b) their respective HAADF-STEM images. (c) Absorption and emission spectra of polar 12–6–12-OH Gemini pQDs in EtOH, with (d) their respective HAADF-STEM images. (e) Comparison of core and hydrodynamic QD sizes from TEM and DLS. (f) Polar pQDs at different concentrations. (g) Photo of polar pQDs dispersed in various polar solvents (i-PrOH = isopropanol, CyOH = cyclohexanol, BuOH = butanol).
To demonstrate that the polar ligand shells are small (<1 nm), we performed dynamic light scattering (DLS, Figure 1e). We found that the hydrodynamic size of the polar pQDs in EtOH was about 8.5 nm, which, compared to the 7.1 nm from HAADF-STEM, suggests a ligand shell of about 0.7 nm. This matches the interparticle distance of 1.4 nm obtained via HAADF-STEM (Figure SI11). Although the ligand shell is small, the pQDs exhibit excellent colloidal stability, with solutions up to 10 mg/mL (Figure 1f). We also dispersed the polar pQDs in various alcohols, including isopropanol cyclopropanol and n-butanol (Figures 1g and SI12, see Supporting Note). These solvents are also nontoxic and green solvents for sustainable chemistry. (17) As QD dispersions are generally a few wt %, switching to green solvents will significantly lower their chemical footprint.
As shown in Figure 2a and SI13, the size of the polar pQDs is tunable (see SI), with the first excitonic peak tunable from 478 to 507 nm, corresponding to pQDs of roughly 5 to 10 nm. (13) The hydrodynamic sizes (Figure 2b) of these pQDs further confirm an average 1.9 nm difference in their respective core sizes, resulting in an average ligand shell thinner than 1 nm (Figure SI14). The composition of the polar pQDs can be tuned via anion exchange using ZnCl2 and ZnI2 (Figures 2c and SI15). (13) Further engineer of the ligand shell thickness and polarity can be achieved by changing the ligand tail length as well as using alkoxy and carboxylic acid tail groups (Figure SI16). Finally, to confirm the thin ligand shell, we performed quenching experiments with a nonbinding electron scavenger (p-benzoquinone), demonstrating a nearly order of magnitude fast quenching for 12–6–12-OH capped QDs compared to methyl-16-bromohexadecanoate Gemini and lecithin capped QDs (Figure SI17).
Figure 2
Figure 2. Size and composition tuning of polar pQDs. (a) Absorption spectra of 5–10 nm polar pQDs dispersed in EtOH with (b) their respective hydrodynamic sizes from DLS. (c) Absorption spectra of anion exchange polar pQDs with the inset a photo of the PL.
To summarize, we demonstrate the synthesis of size- and composition-tunable pQDs dispersed in polar solvents by capping them with polar membrane like Gemini ligands. Although these ligand shells are thin (<1 nm), the pQDs exhibit a remarkable chemical and colloidal stability toward polar solvents. This opens opportunities for polar solvent processing pQDs optoelectronic devices using green polar solvents, orthogonal solvent processing, and apolar QD transport layers. Likewise, the polar pQDs will result in exciting new studies on photocatalysis and electrochemistry, which require pQDs to be stable in polar solvents.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c04073.
Materials and methods, additional TEM images, NMR spectra, long-term stability measurements, table with reported approaches for polar pQDs, additional data on different sized pQDs and pQDs in different solvents, PL spectra of different compositions, QDs with shorter ligands and different polar groups, quenching experiments using p-benzoquinone, data on QD films (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We acknowledge financial support by the Bavarian State Ministry of Science, Research, and Arts and the LMU Munich through the grant “Solar Technologies go Hybrid (SolTech)”. Q.A.A. acknowledges the LMUexcellent, funded by the Federal Ministry of Education and Research (BMBF) and the Free State of Bavaria under the Excellence Strategy of the Federal, as well as the Center of Nanoscience (CeNS) investment fund. F.H. thank the CSC for Ph.D. funding. We thank Prof. E. Cortes (LMU) for access to the DLS zetasizer. We also thank Prof. J. Feldmann for his support for this research, local research centers such as the CeNS for providing communicative networking structures, and the Central Analytics in the Department of Chemistry (LMU) for providing NMR measurements.
References
This article references 17 other publications.
- 1Kaplan, A. E. K. Hong–Ou–Mandel Interference in Colloidal CsPbBr3 Perovskite Nanocrystals. Nat. Photonics 2023, 17, 775– 780, DOI: 10.1038/s41566-023-01225-wGoogle ScholarThere is no corresponding record for this reference.
- 2Kim, J. S. Ultra-Bright, Efficient and Stable Perovskite Light-Emitting Diodes. Nature 2022, 611, 688– 694, DOI: 10.1038/s41586-022-05304-wGoogle ScholarThere is no corresponding record for this reference.
- 3Morad, V. Designer Phospholipid Capping Ligands for Soft Metal Halide Nanocrystals. Nature 2024, 626, 542– 548, DOI: 10.1038/s41586-023-06932-6Google ScholarThere is no corresponding record for this reference.
- 4Avugadda, S. K. Highly Emitting Perovskite Nanocrystals with 2-Year Stability in Water through an Automated Polymer Encapsulation for Bioimaging. ACS Nano 2022, 16, 13657– 13666, DOI: 10.1021/acsnano.2c01556Google ScholarThere is no corresponding record for this reference.
- 5Ahlawat, M.; Neelakshi; Ramapanicker, R.; Govind Rao, V. Design Principle of a Water-Dispersed Photocatalytic Perovskite through Ligand Deconstruction. ACS Energy Lett. 2023, 8, 2159– 2168, DOI: 10.1021/acsenergylett.3c00532Google ScholarThere is no corresponding record for this reference.
- 6Chen, J.; Huang, X.; Xu, Z.; Chi, Y. Alcohol-Stable Perovskite Nanocrystals and Their in Situ Capsulation with Polystyrene. ACS Appl. Mater. Interfaces 2022, 14, 33703– 33711, DOI: 10.1021/acsami.2c07707Google ScholarThere is no corresponding record for this reference.
- 7Zheng, L. Water-Assisted Preparation of Ethanol-Dispersed CsPbBr3 Perovskite Nanocrystals and Emissive Gel. J. Colloid Interface Sci. 2021, 598, 166– 171, DOI: 10.1016/j.jcis.2021.04.017Google ScholarThere is no corresponding record for this reference.
- 8Rossi, C. Exploiting the Transformative Features of Metal Halides for the Synthesis of CsPbBr3@SiO2 Core–Shell Nanocrystals. Chem. Mater. 2022, 34, 405– 413, DOI: 10.1021/acs.chemmater.1c03749Google ScholarThere is no corresponding record for this reference.
- 9Loiudice, A.; Strach, M.; Saris, S.; Chernyshov, D.; Buonsanti, R. Universal Oxide Shell Growth Enables in Situ Structural Studies of Perovskite Nanocrystals During the Anion Exchange Reaction. J. Am. Chem. Soc. 2019, 141, 8254– 8263, DOI: 10.1021/jacs.9b02061Google ScholarThere is no corresponding record for this reference.
- 10Karan, N. S. Beyond Surface Facets and Morphology: Precision Silica Shelling for Enhanced Luminescence and Stability in Halide Perovskite Nanocrystals. J. Am. Chem. Soc. 2025, 147, 45043– 45056, DOI: 10.1021/jacs.5c13729Google ScholarThere is no corresponding record for this reference.
- 11Guggisberg, D. Colloidal CsPbX3 Nanocrystals with Thin Metal Oxide Gel Coatings. Chem. Mater. 2023, 35, 2827– 2834, DOI: 10.1021/acs.chemmater.2c03562Google ScholarThere is no corresponding record for this reference.
- 12Kovalenko, M. V.; Scheele, M.; Talapin, D. V. Colloidal Nanocrystals with Molecular Metal Chalcogenide Surface Ligands. Science 2009, 324, 1417– 1420, DOI: 10.1126/science.1170524Google ScholarThere is no corresponding record for this reference.
- 13Akkerman, Q. A. Spheroidal Cesium Lead Chloride-Bromide Quantum Dots and a Fast Determination of Their Size and Halide Content. Nano Lett. 2022, 22, 8168– 8173, DOI: 10.1021/acs.nanolett.2c02601Google ScholarThere is no corresponding record for this reference.
- 14Jeong, W. H. Synergistic Surface Modification for High-Efficiency Perovskite Nanocrystal Light-Emitting Diodes: Divalent Metal Ion Doping and Halide-Based Ligand Passivation. Adv. Sci. 2024, 11, 2305383, DOI: 10.1002/advs.202305383Google ScholarThere is no corresponding record for this reference.
- 15Saito, N.; Urayama, A.; Ikezawa, M.; Kondo, Y. Water-Durable Cesium Lead Halide Perovskite Nanocrystals Passivated with a Cationic Gemini Surfactant. Advanced Materials Interfaces 2022, 9, 2101836, DOI: 10.1002/admi.202101836Google ScholarThere is no corresponding record for this reference.
- 16Smock, S. R.; Williams, T. J.; Brutchey, R. L. Quantifying the Thermodynamics of Ligand Binding to CsPbBr3 Quantum Dots. Angew. Chem. Int. 2018, 57, 11711– 11715, DOI: 10.1002/anie.201806916Google ScholarThere is no corresponding record for this reference.
- 17Prat, D.; Hayler, J.; Wells, A. A Survey of Solvent Selection Guides. Green Chem. 2014, 16, 4546– 4551, DOI: 10.1039/C4GC01149JGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
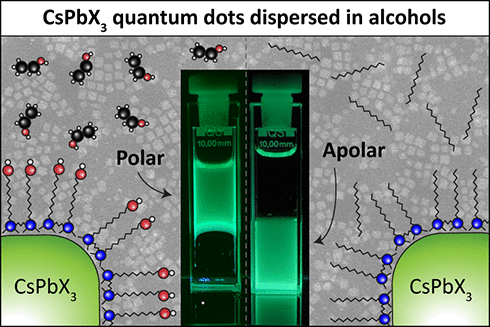
Figure 1

Figure 1. Polar and apolar Gemini caped CsPbBr3 QDs. (a) Absorption and emission spectra of apolar 12–6–12 Gemini pQDs in toluene, with (b) their respective HAADF-STEM images. (c) Absorption and emission spectra of polar 12–6–12-OH Gemini pQDs in EtOH, with (d) their respective HAADF-STEM images. (e) Comparison of core and hydrodynamic QD sizes from TEM and DLS. (f) Polar pQDs at different concentrations. (g) Photo of polar pQDs dispersed in various polar solvents (i-PrOH = isopropanol, CyOH = cyclohexanol, BuOH = butanol).
Figure 2
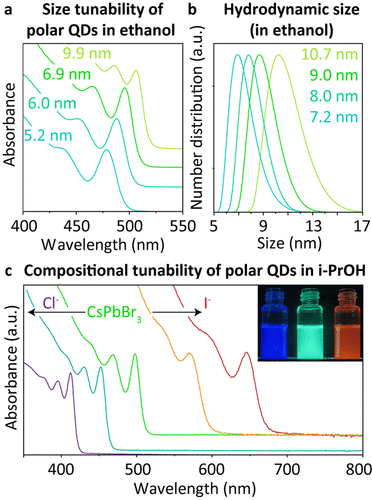
Figure 2. Size and composition tuning of polar pQDs. (a) Absorption spectra of 5–10 nm polar pQDs dispersed in EtOH with (b) their respective hydrodynamic sizes from DLS. (c) Absorption spectra of anion exchange polar pQDs with the inset a photo of the PL.
References
This article references 17 other publications.
- 1Kaplan, A. E. K. Hong–Ou–Mandel Interference in Colloidal CsPbBr3 Perovskite Nanocrystals. Nat. Photonics 2023, 17, 775– 780, DOI: 10.1038/s41566-023-01225-wThere is no corresponding record for this reference.
- 2Kim, J. S. Ultra-Bright, Efficient and Stable Perovskite Light-Emitting Diodes. Nature 2022, 611, 688– 694, DOI: 10.1038/s41586-022-05304-wThere is no corresponding record for this reference.
- 3Morad, V. Designer Phospholipid Capping Ligands for Soft Metal Halide Nanocrystals. Nature 2024, 626, 542– 548, DOI: 10.1038/s41586-023-06932-6There is no corresponding record for this reference.
- 4Avugadda, S. K. Highly Emitting Perovskite Nanocrystals with 2-Year Stability in Water through an Automated Polymer Encapsulation for Bioimaging. ACS Nano 2022, 16, 13657– 13666, DOI: 10.1021/acsnano.2c01556There is no corresponding record for this reference.
- 5Ahlawat, M.; Neelakshi; Ramapanicker, R.; Govind Rao, V. Design Principle of a Water-Dispersed Photocatalytic Perovskite through Ligand Deconstruction. ACS Energy Lett. 2023, 8, 2159– 2168, DOI: 10.1021/acsenergylett.3c00532There is no corresponding record for this reference.
- 6Chen, J.; Huang, X.; Xu, Z.; Chi, Y. Alcohol-Stable Perovskite Nanocrystals and Their in Situ Capsulation with Polystyrene. ACS Appl. Mater. Interfaces 2022, 14, 33703– 33711, DOI: 10.1021/acsami.2c07707There is no corresponding record for this reference.
- 7Zheng, L. Water-Assisted Preparation of Ethanol-Dispersed CsPbBr3 Perovskite Nanocrystals and Emissive Gel. J. Colloid Interface Sci. 2021, 598, 166– 171, DOI: 10.1016/j.jcis.2021.04.017There is no corresponding record for this reference.
- 8Rossi, C. Exploiting the Transformative Features of Metal Halides for the Synthesis of CsPbBr3@SiO2 Core–Shell Nanocrystals. Chem. Mater. 2022, 34, 405– 413, DOI: 10.1021/acs.chemmater.1c03749There is no corresponding record for this reference.
- 9Loiudice, A.; Strach, M.; Saris, S.; Chernyshov, D.; Buonsanti, R. Universal Oxide Shell Growth Enables in Situ Structural Studies of Perovskite Nanocrystals During the Anion Exchange Reaction. J. Am. Chem. Soc. 2019, 141, 8254– 8263, DOI: 10.1021/jacs.9b02061There is no corresponding record for this reference.
- 10Karan, N. S. Beyond Surface Facets and Morphology: Precision Silica Shelling for Enhanced Luminescence and Stability in Halide Perovskite Nanocrystals. J. Am. Chem. Soc. 2025, 147, 45043– 45056, DOI: 10.1021/jacs.5c13729There is no corresponding record for this reference.
- 11Guggisberg, D. Colloidal CsPbX3 Nanocrystals with Thin Metal Oxide Gel Coatings. Chem. Mater. 2023, 35, 2827– 2834, DOI: 10.1021/acs.chemmater.2c03562There is no corresponding record for this reference.
- 12Kovalenko, M. V.; Scheele, M.; Talapin, D. V. Colloidal Nanocrystals with Molecular Metal Chalcogenide Surface Ligands. Science 2009, 324, 1417– 1420, DOI: 10.1126/science.1170524There is no corresponding record for this reference.
- 13Akkerman, Q. A. Spheroidal Cesium Lead Chloride-Bromide Quantum Dots and a Fast Determination of Their Size and Halide Content. Nano Lett. 2022, 22, 8168– 8173, DOI: 10.1021/acs.nanolett.2c02601There is no corresponding record for this reference.
- 14Jeong, W. H. Synergistic Surface Modification for High-Efficiency Perovskite Nanocrystal Light-Emitting Diodes: Divalent Metal Ion Doping and Halide-Based Ligand Passivation. Adv. Sci. 2024, 11, 2305383, DOI: 10.1002/advs.202305383There is no corresponding record for this reference.
- 15Saito, N.; Urayama, A.; Ikezawa, M.; Kondo, Y. Water-Durable Cesium Lead Halide Perovskite Nanocrystals Passivated with a Cationic Gemini Surfactant. Advanced Materials Interfaces 2022, 9, 2101836, DOI: 10.1002/admi.202101836There is no corresponding record for this reference.
- 16Smock, S. R.; Williams, T. J.; Brutchey, R. L. Quantifying the Thermodynamics of Ligand Binding to CsPbBr3 Quantum Dots. Angew. Chem. Int. 2018, 57, 11711– 11715, DOI: 10.1002/anie.201806916There is no corresponding record for this reference.
- 17Prat, D.; Hayler, J.; Wells, A. A Survey of Solvent Selection Guides. Green Chem. 2014, 16, 4546– 4551, DOI: 10.1039/C4GC01149JThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.5c04073.
Materials and methods, additional TEM images, NMR spectra, long-term stability measurements, table with reported approaches for polar pQDs, additional data on different sized pQDs and pQDs in different solvents, PL spectra of different compositions, QDs with shorter ligands and different polar groups, quenching experiments using p-benzoquinone, data on QD films (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



