Role of the Electrolyte Concentration in Polysulfide Shuttle and Electrochemical Performance of Lithium–Sulfur BatteriesClick to copy article linkArticle link copied!
- Md Shahriar NahianMd Shahriar NahianSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Md Shahriar Nahian
- Arpan K. SharmaArpan K. SharmaSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Arpan K. Sharma
- Bairav S. VishnugopiBairav S. VishnugopiSchool of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Bairav S. Vishnugopi
- JiYoung SeoJiYoung SeoEnergy & Environment Directorate, Pacific Northwest National Laboratory, Richland, Washington 99354, United StatesMore by JiYoung Seo
- Lirong ZhongLirong ZhongEnergy & Environment Directorate, Pacific Northwest National Laboratory, Richland, Washington 99354, United StatesMore by Lirong Zhong
- Lili ShiLili ShiEnergy & Environment Directorate, Pacific Northwest National Laboratory, Richland, Washington 99354, United StatesMore by Lili Shi
- Partha P. Mukherjee*Partha P. Mukherjee*Email: [email protected]School of Mechanical Engineering, Purdue University, West Lafayette, Indiana 47907, United StatesMore by Partha P. Mukherjee
Abstract
Lithium–sulfur (Li–S) batteries promise high energy density, yet their operation is constrained by a complex interplay of electrolyte, electrode, and interfacial processes. The electrolyte governs dissolution, redox kinetics, and ionic transport, serving as both the enabler and the bottleneck of cell performance. In this work, we develop a microstructure-coupled mechanistic framework to examine how the electrolyte concentration, volume, and solvation strength jointly regulate charge-carrier availability, ionic mobility, and shuttle reactions. The analysis reveals distinct regimes defined by the competition among dissolution, transport, and conversion processes. The electrolyte-to-sulfur ratio and cathode porosity further modulate this balance, delineating a narrow, conductivity-optimal window where kinetics and transport remain coupled. Solvent solubility shifts this window: weak solvation impedes conversion, whereas excessive solvation accelerates shuttle losses. These insights establish a comprehensive conductivity-based design framework linking electrolyte chemistry, solvent properties, and cathode architecture for achieving high-efficiency and durable liquid Li–S batteries.
This publication is licensed for personal use by The American Chemical Society.
Figure 1
Figure 1. (a) During the Li–S discharge process, species evolution occurs within both the cathode microstructure and the electrolyte. S8(s) first dissolves into the electrolyte, followed by a multi-step electrochemical reduction at the cathode, ultimately leading to the precipitation of solid Li2S. (b) Electrolyte properties influence reaction kinetics, dissolution–precipitation behavior, and species transport in Li–S batteries. Variations in the electrolyte-to-sulfur (E/S) ratio, solvation strength, and LiTFSI concentration modify ion transport and electrochemical reaction pathways, collectively defining the electrolyte signatures governing Li–S performance. Solvation free energies are adapted from Kim et al. (37)
Figure 2
Figure 2. Simulation results illustrating the effect of the salt concentration on the Li–S cell behavior. (a) Electrochemical performance of a Li–S cell at varying salt concentrations in the electrolyte, evaluated with 80% cathode porosity and 20 vol % sulfur loading. (b) Sulfur fractions of unconverted S42– species and precipitated Li2S in the cathode at different salt concentrations. (c) Evolution of ionic conductivity during discharge for different salt concentrations, where the conductivity is normalized by the maximum ionic conductivity observed during discharge. (d) Electrolyte potential drop during discharge at different salt concentrations. (e) Evolution of PSs during discharge for different salt concentrations.
Figure 3
Figure 3. Nyquist plots of Li–S cells with electrolytes at different LiTFSI concentrations (a) before and (b) after discharge. (c) Experimental ionic conductivity measurement of electrolytes at different LiTFSI concentrations. (d) Simulated sulfur utilization maps as a function of the electrolyte-to-sulfur (E/S) ratio and salt concentration for pristine cathodes with different porosities (60–90 vol %). (e) Map of the salt concentration versus E/S ratio highlighting regions of high sulfur utilization and the “sudden death” zone. (f) Variation in the number of charge carriers and their interactions with changes in the salt concentration and E/S ratio.
Figure 4
Figure 4. (a) Reversible shuttle and (b) irreversible shuttle behavior as functions of the salt concentration and E/S ratio. (c) Key performance descriptors of a Li–S battery and their dependence on the salt concentration. (d) Shuttle influx and consumption at the anode varies with the salt concentration, correlating with transport resistance. Shuttle influx and consumption calculated for (e) 0.1 M and (f) 1.0 M salt concentrations. (g) Variation of irreversible chemical redox reactions with the initial salt concentration.
Figure 5
Figure 5. (a) Schematic representation depicting the correlation of solvation with Li+ diffusivity; stronger solvation enhances solubility, thereby improving reaction kinetics. (b) Electrochemical performance of the Li–S cell for different ksp values. Species evolution during discharge for electrolytes with (c) ksp = 5, (d) ksp = 7, and (e) ksp = 9. (f) Discharge capacity as a function of solubility (ksp). (g) Sulfur loss due to the shuttle effect as a function of ksp. (h) Qualitative Sankey diagram illustrating the interdependencies and multivariate impact of the cathode architecture, electrolyte properties, and solubility on the mechanistic response of Li–S batteries, including transport, reaction kinetics, capacity, and shuttle loss.
Experimental Methods
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.6c00391.
Computational framework to predict electrochemical performance (section S1) and experimental methods and results (section S2) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This work was supported by the U.S. Department of Energy under Award DE-EE0011166, as part of the Advanced Battery Materials Research (BMR) Program.
References
This article references 69 other publications.
- 1Barchasz, C.; Molton, F.; Duboc, C.; Leprêtre, J.-C.; Patoux, S.; Alloin, F. Lithium/Sulfur Cell Discharge Mechanism: An Original Approach for Intermediate Species Identification. Anal. Chem. 2012, 84 (9), 3973– 3980, DOI: 10.1021/ac2032244Google ScholarThere is no corresponding record for this reference.
- 2Viswanathan, V.; Epstein, A. H.; Chiang, Y.-M.; Takeuchi, E.; Bradley, M.; Langford, J.; Winter, M. The Challenges and Opportunities of Battery-Powered Flight. Nature 2022, 601 (7894), 519– 525, DOI: 10.1038/s41586-021-04139-1Google ScholarThere is no corresponding record for this reference.
- 3Yari, S.; Conde Reis, A.; Pang, Q.; Safari, M. Performance Benchmarking and Analysis of Lithium–Sulfur Batteries for next-Generation Cell Design. Nat. Commun. 2025, 16 (1), 5473, DOI: 10.1038/s41467-025-60528-4Google ScholarThere is no corresponding record for this reference.
- 4Ji, X.; Lee, K. T.; Nazar, L. F. A Highly Ordered Nanostructured Carbon–Sulphur Cathode for Lithium–Sulphur Batteries. Nat. Mater. 2009, 8 (6), 500– 506, DOI: 10.1038/nmat2460Google ScholarThere is no corresponding record for this reference.
- 5Elemental Sulfur and Sulfur-Rich Compounds II; Steudel, R., Ed.; Springer Science & Business Media: Berlin, Germany, 2003; DOI: 10.1007/b11909 .Google ScholarThere is no corresponding record for this reference.
- 6Rauh, R. D.; Shuker, F. S.; Marston, J. M.; Brummer, S. B. Formation of Lithium Polysulfides in Aprotic Media. Journal of Inorganic and Nuclear Chemistry 1977, 39 (10), 1761– 1766, DOI: 10.1016/0022-1902(77)80198-XGoogle ScholarThere is no corresponding record for this reference.
- 7Zhou, G.; Chen, H.; Cui, Y. Formulating Energy Density for Designing Practical Lithium–Sulfur Batteries. Nat. Energy 2022, 7 (4), 312– 319, DOI: 10.1038/s41560-022-01001-0Google ScholarThere is no corresponding record for this reference.
- 8Sharma, A. K.; Vishnugopi, B. S.; Alsaç, E. P.; McDowell, M. T.; Mukherjee, P. P. Passivation-Induced Species Dynamics and Microstructural Evolution in Solid-State Lithium–Sulfur Cathodes. Advanced Science 2026, e20537 DOI: 10.1002/advs.202520537Google ScholarThere is no corresponding record for this reference.
- 9Kausthubharam; Vishnugopi, B. S.; Alujjage, A. S. J.; Premnath, V.; Tang, W. S.; Jeevarajan, J. A.; Mukherjee, P. P. Mechanistic Understanding of Thermal Stability and Safety in Lithium Metal Batteries. Chem. Rev. 2026, 126 (1), 404– 447, DOI: 10.1021/acs.chemrev.5c00621Google ScholarThere is no corresponding record for this reference.
- 10Barchasz, C.; Leprêtre, J.-C.; Alloin, F.; Patoux, S. New Insights into the Limiting Parameters of the Li/S Rechargeable Cell. J. Power Sources 2012, 199, 322– 330, DOI: 10.1016/j.jpowsour.2011.07.021Google ScholarThere is no corresponding record for this reference.
- 11Lai, C.; Gao, X. P.; Zhang, B.; Yan, T. Y.; Zhou, Z. Synthesis and Electrochemical Performance of Sulfur/Highly Porous Carbon Composites. J. Phys. Chem. C 2009, 113 (11), 4712– 4716, DOI: 10.1021/jp809473eGoogle ScholarThere is no corresponding record for this reference.
- 12Chen, S.-R.; Zhai, Y.-P.; Xu, G.-L.; Jiang, Y.-X.; Zhao, D.-Y.; Li, J.-T.; Huang, L.; Sun, S.-G. Ordered Mesoporous Carbon/Sulfur Nanocomposite of High Performances as Cathode for Lithium–Sulfur Battery. Electrochim. Acta 2011, 56 (26), 9549– 9555, DOI: 10.1016/j.electacta.2011.03.005Google ScholarThere is no corresponding record for this reference.
- 13Li, X.; Cao, Y.; Qi, W.; Saraf, L. V.; Xiao, J.; Nie, Z.; Mietek, J.; Zhang, J.-G.; Schwenzer, B.; Liu, J. Optimization of Mesoporous Carbon Structures for Lithium–Sulfur Battery Applications. J. Mater. Chem. 2011, 21 (41), 16603– 16610, DOI: 10.1039/c1jm12979aGoogle ScholarThere is no corresponding record for this reference.
- 14Liang, X.; Wen, Z.; Liu, Y.; Zhang, H.; Huang, L.; Jin, J. Highly Dispersed Sulfur in Ordered Mesoporous Carbon Sphere as a Composite Cathode for Rechargeable Polymer Li/S Battery. J. Power Sources 2011, 196 (7), 3655– 3658, DOI: 10.1016/j.jpowsour.2010.12.052Google ScholarThere is no corresponding record for this reference.
- 15Mistry, A. N.; Mukherjee, P. P. “Shuttle” in Polysulfide Shuttle: Friend or Foe?. J. Phys. Chem. C 2018, 122 (42), 23845– 23851, DOI: 10.1021/acs.jpcc.8b06077Google ScholarThere is no corresponding record for this reference.
- 16Mistry, A.; Mukherjee, P. P. Precipitation–Microstructure Interactions in the Li–Sulfur Battery Electrode. J. Phys. Chem. C 2017, 121 (47), 26256– 26264, DOI: 10.1021/acs.jpcc.7b09997Google ScholarThere is no corresponding record for this reference.
- 17Nahian, M. S.; Jayan, R.; Islam, M. M. Atomic-Scale Insights into Comparative Mechanisms and Kinetics of Na–S and Li–S Batteries. ACS Catal. 2022, 12 (13), 7664– 7676, DOI: 10.1021/acscatal.2c01174Google ScholarThere is no corresponding record for this reference.
- 18Rao, M.; Song, X.; Cairns, E. J. Nano-Carbon/Sulfur Composite Cathode Materials with Carbon Nanofiber as Electrical Conductor for Advanced Secondary Lithium/Sulfur Cells. J. Power Sources 2012, 205, 474– 478, DOI: 10.1016/j.jpowsour.2012.01.047Google ScholarThere is no corresponding record for this reference.
- 19Zheng, W.; Liu, Y. W.; Hu, X. G.; Zhang, C. F. Novel Nanosized Adsorbing Sulfur Composite Cathode Materials for the Advanced Secondary Lithium Batteries. Electrochim. Acta 2006, 51 (7), 1330– 1335, DOI: 10.1016/j.electacta.2005.06.021Google ScholarThere is no corresponding record for this reference.
- 20Cistjakov, W.; Hoppe, J.; Jung, J.; Röder, F.; Kim, H.-T.; Krewer, U. Insight into the Impact of Electrolyte on Passivation of Lithium–Sulfur Cathodes. Advanced Materials Interfaces 2025, 12 (5), 2400632, DOI: 10.1002/admi.202400632Google ScholarThere is no corresponding record for this reference.
- 21Liu, Z.; Mukherjee, P. P. Mesoscale Elucidation of Surface Passivation in the Li–Sulfur Battery Cathode. ACS Appl. Mater. Interfaces 2017, 9 (6), 5263– 5271, DOI: 10.1021/acsami.6b15066Google ScholarThere is no corresponding record for this reference.
- 22Nahian, M. S.; Sharma, A. K.; Vishnugopi, B. S. Precipitate-Driven Electrochemical Interactions in Lithium–Sulfur Batteries. J. Electrochem. En. Conv. Stor 2026, 23, 024704, DOI: 10.1115/1.4070879Google ScholarThere is no corresponding record for this reference.
- 23Jana, M.; Xu, R.; Cheng, X.-B.; Yeon, J. S.; Park, J. M.; Huang, J.-Q.; Zhang, Q.; Park, H. S. Rational Design of Two-Dimensional Nanomaterials for Lithium–Sulfur Batteries. Energy Environ. Sci. 2020, 13 (4), 1049– 1075, DOI: 10.1039/C9EE02049GGoogle ScholarThere is no corresponding record for this reference.
- 24Lin, H.; Yang, L.; Jiang, X.; Li, G.; Zhang, T.; Yao, Q.; Zheng, G. W.; Lee, J. Y. Electrocatalysis of Polysulfide Conversion by Sulfur-Deficient MoS2 Nanoflakes for Lithium–Sulfur Batteries. Energy Environ. Sci. 2017, 10 (6), 1476– 1486, DOI: 10.1039/C7EE01047HGoogle ScholarThere is no corresponding record for this reference.
- 25Yuan, Z.; Peng, H.-J.; Huang, J.-Q.; Liu, X.-Y.; Wang, D.-W.; Cheng, X.-B.; Zhang, Q. Hierarchical Free-Standing Carbon-Nanotube Paper Electrodes with Ultrahigh Sulfur-Loading for Lithium–Sulfur Batteries. Adv. Funct. Mater. 2014, 24 (39), 6105– 6112, DOI: 10.1002/adfm.201401501Google ScholarThere is no corresponding record for this reference.
- 26Fan, F. Y.; Carter, W. C.; Chiang, Y.-M. Mechanism and Kinetics of Li2S Precipitation in Lithium–Sulfur Batteries. Adv. Mater. 2015, 27 (35), 5203– 5209, DOI: 10.1002/adma.201501559Google ScholarThere is no corresponding record for this reference.
- 27Kim, Y.; Kim, W. I.; Park, H.; Kim, J. S.; Cho, H.; Yeon, J. S.; Kim, J.; Kim, Y.-J.; Lee, J.; Park, H. S. Multifunctional Polymeric Phthalocyanine-Coated Carbon Nanotubes for Efficient Redox Mediators of Lithium–Sulfur Batteries. Adv. Energy Mater. 2023, 13 (22), 2204353, DOI: 10.1002/aenm.202204353Google ScholarThere is no corresponding record for this reference.
- 28Kim, W. I.; Shin, J. C.; Kim, M. J.; Jang, G.; Lee, M.; Park, H. S. Ionic Structured Redox-Mediating Polymeric Sulfurs for Lithium–Sulfur Batteries. ACS Energy Lett. 2025, 10 (5), 2410– 2418, DOI: 10.1021/acsenergylett.5c00808Google ScholarThere is no corresponding record for this reference.
- 29Liang, X.; Hart, C.; Pang, Q.; Garsuch, A.; Weiss, T.; Nazar, L. F. A Highly Efficient Polysulfide Mediator for Lithium–Sulfur Batteries. Nat. Commun. 2015, 6 (1), 5682, DOI: 10.1038/ncomms6682Google ScholarThere is no corresponding record for this reference.
- 30Liu, Y.; Elias, Y.; Meng, J.; Aurbach, D.; Zou, R.; Xia, D.; Pang, Q. Electrolyte Solutions Design for Lithium–Sulfur Batteries. Joule 2021, 5 (9), 2323– 2364, DOI: 10.1016/j.joule.2021.06.009Google ScholarThere is no corresponding record for this reference.
- 31Guo, J.; Xu, Y.; Wang, C. Sulfur-Impregnated Disordered Carbon Nanotubes Cathode for Lithium–Sulfur Batteries. Nano Lett. 2011, 11 (10), 4288– 4294, DOI: 10.1021/nl202297pGoogle ScholarThere is no corresponding record for this reference.
- 32Tarascon, J.-M.; Armand, M. Issues and Challenges Facing Rechargeable Lithium Batteries. Nature 2001, 414 (6861), 359– 367, DOI: 10.1038/35104644Google ScholarThere is no corresponding record for this reference.
- 33Bruce, P. G.; Freunberger, S. A.; Hardwick, L. J.; Tarascon, J.-M. Li–O2 and Li–S Batteries with High Energy Storage. Nat. Mater. 2012, 11 (1), 19– 29, DOI: 10.1038/nmat3191Google ScholarThere is no corresponding record for this reference.
- 34Goodenough, J. B.; Park, K.-S. The Li-Ion Rechargeable Battery: A Perspective. J. Am. Chem. Soc. 2013, 135 (4), 1167– 1176, DOI: 10.1021/ja3091438Google ScholarThere is no corresponding record for this reference.
- 35Zhang, B.; Wu, J.; Gu, J.; Li, S.; Yan, T.; Gao, X.-P. The Fundamental Understanding of Lithium Polysulfides in Ether-Based Electrolyte for Lithium–Sulfur Batteries. ACS Energy Lett. 2021, 6 (2), 537– 546, DOI: 10.1021/acsenergylett.0c02527Google ScholarThere is no corresponding record for this reference.
- 36Mistry, A. N.; Mukherjee, P. P. Electrolyte Transport Evolution Dynamics in Lithium–Sulfur Batteries. J. Phys. Chem. C 2018, 122 (32), 18329– 18335, DOI: 10.1021/acs.jpcc.8b05442Google ScholarThere is no corresponding record for this reference.
- 37Kim, S. C.; Gao, X.; Liao, S.-L.; Su, H.; Chen, Y.; Zhang, W.; Greenburg, L. C.; Pan, J.-A.; Zheng, X.; Ye, Y.; Kim, M. S.; Sayavong, P.; Brest, A.; Qin, J.; Bao, Z.; Cui, Y. Solvation-Property Relationship of Lithium–Sulphur Battery Electrolytes. Nat. Commun. 2024, 15 (1), 1268, DOI: 10.1038/s41467-023-44527-xGoogle ScholarThere is no corresponding record for this reference.
- 38Huang, Y.; Shaibani, M.; Abedin, Md. J.; Mendoza, D. J.; Xu, Z.; Gamot, T. D.; Cooray, M. C. D.; Lin, M.; Garnier, G.; Hill, M. R.; Majumder, M. Sulfur Cathodes with Self-Organized Cellulose Nanofibers in Stable Ah-Level, > 300 Wh kg–1 Lithium–Sulfur Cells. Adv. Energy Mater. 2022, 12 (45), 2202474, DOI: 10.1002/aenm.202202474Google ScholarThere is no corresponding record for this reference.
- 39Chung, S.-H.; Chang, C.-H.; Manthiram, A. Progress on the Critical Parameters for Lithium–Sulfur Batteries to Be Practically Viable. Adv. Funct. Mater. 2018, 28 (28), 1801188, DOI: 10.1002/adfm.201801188Google ScholarThere is no corresponding record for this reference.
- 40Li, Z.; Rao, H.; Atwi, R.; Sivakumar, B. M.; Gwalani, B.; Gray, S.; Han, K. S.; Everett, T. A.; Ajantiwalay, T. A.; Murugesan, V.; Rajput, N. N.; Pol, V. G. Non-Polar Ether-Based Electrolyte Solutions for Stable High-Voltage Non-Aqueous Lithium Metal Batteries. Nat. Commun. 2023, 14 (1), 868, DOI: 10.1038/s41467-023-36647-1Google ScholarThere is no corresponding record for this reference.
- 41Cheng, Q.; Chen, Z.-X.; Li, X.-Y.; Hou, L.-P.; Bi, C.-X.; Zhang, X.-Q.; Huang, J.-Q.; Li, B.-Q. Constructing a 700 Wh kg–1-Level Rechargeable Lithium–Sulfur Pouch Cell. Journal of Energy Chemistry 2023, 76, 181– 186, DOI: 10.1016/j.jechem.2022.09.029Google ScholarThere is no corresponding record for this reference.
- 42Hagen, M.; Hanselmann, D.; Ahlbrecht, K.; Maça, R.; Gerber, D.; Tübke, J. Lithium–Sulfur Cells: The Gap between the State-of-the-Art and the Requirements for High Energy Battery Cells. Adv. Energy Mater. 2015, 5 (16), 1401986, DOI: 10.1002/aenm.201401986Google ScholarThere is no corresponding record for this reference.
- 43Jozwiuk, A.; Berkes, B. B.; Weiß, T.; Sommer, H.; Janek, J.; Brezesinski, T. The Critical Role of Lithium Nitrate in the Gas Evolution of Lithium–Sulfur Batteries. Energy Environ. Sci. 2016, 9 (8), 2603– 2608, DOI: 10.1039/C6EE00789AGoogle ScholarThere is no corresponding record for this reference.
- 44Cleaver, T.; Kovacik, P.; Marinescu, M.; Zhang, T.; Offer, G. Perspective─Commercializing Lithium Sulfur Batteries: Are We Doing the Right Research?. J. Electrochem. Soc. 2018, 165 (1), A6029, DOI: 10.1149/2.0071801jesGoogle ScholarThere is no corresponding record for this reference.
- 45Rajput, N. N.; Murugesan, V.; Shin, Y.; Han, K. S.; Lau, K. C.; Chen, J.; Liu, J.; Curtiss, L. A.; Mueller, K. T.; Persson, K. A. Elucidating the Solvation Structure and Dynamics of Lithium Polysulfides Resulting from Competitive Salt and Solvent Interactions. Chem. Mater. 2017, 29 (8), 3375– 3379, DOI: 10.1021/acs.chemmater.7b00068Google ScholarThere is no corresponding record for this reference.
- 46Peng, H.-J.; Huang, J.-Q.; Cheng, X.-B.; Zhang, Q. Review on High-Loading and High-Energy Lithium–Sulfur Batteries. Adv. Energy Mater. 2017, 7 (24), 1700260, DOI: 10.1002/aenm.201700260Google ScholarThere is no corresponding record for this reference.
- 47Jarrold, G.; Manthiram, A. Electrolyte Strategies for Practically Viable All-Solid-State Lithium–Sulfur Batteries. Commun. Mater. 2025, 6 (1), 240, DOI: 10.1038/s43246-025-00960-7Google ScholarThere is no corresponding record for this reference.
- 48Zheng, J.; Lv, D.; Gu, M.; Wang, C.; Zhang, J.-G.; Liu, J.; Xiao, J. How to Obtain Reproducible Results for Lithium Sulfur Batteries?. J. Electrochem. Soc. 2013, 160 (11), A2288, DOI: 10.1149/2.106311jesGoogle ScholarThere is no corresponding record for this reference.
- 49Nagpure, S. C.; Tanim, T. R.; Dufek, E. J.; Viswanathan, V. V.; Crawford, A. J.; Wood, S. M.; Xiao, J.; Dickerson, C. C.; Liaw, B. Impacts of Lean Electrolyte on Cycle Life for Rechargeable Li Metal Batteries. J. Power Sources 2018, 407, 53– 62, DOI: 10.1016/j.jpowsour.2018.10.060Google ScholarThere is no corresponding record for this reference.
- 50Cheng, L.; Curtiss, L. A.; Zavadil, K. R.; Gewirth, A. A.; Shao, Y.; Gallagher, K. G. Sparingly Solvating Electrolytes for High Energy Density Lithium–Sulfur Batteries. ACS Energy Lett. 2016, 1 (3), 503– 509, DOI: 10.1021/acsenergylett.6b00194Google ScholarThere is no corresponding record for this reference.
- 51Deng, D. R.; Xue, F.; Bai, C.-D.; Lei, J.; Yuan, R.; Zheng, M. S.; Dong, Q. F. Enhanced Adsorptions to Polysulfides on Graphene-Supported BN Nanosheets with Excellent Li–S Battery Performance in a Wide Temperature Range. ACS Nano 2018, 12 (11), 11120– 11129, DOI: 10.1021/acsnano.8b05534Google ScholarThere is no corresponding record for this reference.
- 52Zhang, G.; Peng, H.-J.; Zhao, C.-Z.; Chen, X.; Zhao, L.-D.; Li, P.; Huang, J.-Q.; Zhang, Q. The Radical Pathway Based on a Lithium-Metal-Compatible High-Dielectric Electrolyte for Lithium–Sulfur Batteries. Angew. Chem. 2018, 130 (51), 16974– 16978, DOI: 10.1002/ange.201810132Google ScholarThere is no corresponding record for this reference.
- 53Li, G.; Wang, S.; Zhang, Y.; Li, M.; Chen, Z.; Lu, J. Revisiting the Role of Polysulfides in Lithium–Sulfur Batteries. Adv. Mater. 2018, 30 (22), 1705590, DOI: 10.1002/adma.201705590Google ScholarThere is no corresponding record for this reference.
- 54Han, P.; Chung, S.-H.; Manthiram, A. Pyrrolic-Type Nitrogen-Doped Hierarchical Macro/Mesoporous Carbon as a Bifunctional Host for High-Performance Thick Cathodes for Lithium–Sulfur Batteries. Small 2019, 15 (16), 1900690, DOI: 10.1002/smll.201900690Google ScholarThere is no corresponding record for this reference.
- 55Thieme, S.; Brückner, J.; Meier, A.; Bauer, I.; Gruber, K.; Kaspar, J.; Helmer, A.; Althues, H.; Schmuck, M.; Kaskel, S. A Lithium–Sulfur Full Cell with Ultralong Cycle Life: Influence of Cathode Structure and Polysulfide Additive. Journal of Materials Chemistry A 2015, 3 (7), 3808– 3820, DOI: 10.1039/C4TA06748GGoogle ScholarThere is no corresponding record for this reference.
- 56Chen, S.; Dai, F.; Gordin, M. L.; Yu, Z.; Gao, Y.; Song, J.; Wang, D. Functional Organosulfide Electrolyte Promotes an Alternate Reaction Pathway to Achieve High Performance in Lithium–Sulfur Batteries. Angew. Chem., Int. Ed. 2016, 55 (13), 4231– 4235, DOI: 10.1002/anie.201511830Google ScholarThere is no corresponding record for this reference.
- 57Park, C.; Kanduč, M.; Chudoba, R.; Ronneburg, A.; Risse, S.; Ballauff, M.; Dzubiella, J. Molecular Simulations of Electrolyte Structure and Dynamics in Lithium–Sulfur Battery Solvents. J. Power Sources 2018, 373, 70– 78, DOI: 10.1016/j.jpowsour.2017.10.081Google ScholarThere is no corresponding record for this reference.
- 58Chen, S.; Wang, D.; Zhao, Y.; Wang, D. Superior Performance of a Lithium–Sulfur Battery Enabled by a Dimethyl Trisulfide Containing Electrolyte. Small Methods 2018, 2 (6), 1800038, DOI: 10.1002/smtd.201800038Google ScholarThere is no corresponding record for this reference.
- 59Chu, H.; Noh, H.; Kim, Y.-J.; Yuk, S.; Lee, J.-H.; Lee, J.; Kwack, H.; Kim, Y.; Yang, D.-K.; Kim, H.-T. Achieving Three-Dimensional Lithium Sulfide Growth in Lithium–Sulfur Batteries Using High-Donor-Number Anions. Nat. Commun. 2019, 10 (1), 188, DOI: 10.1038/s41467-018-07975-4Google ScholarThere is no corresponding record for this reference.
- 60Zou, Q.; Lu, Y.-C. Solvent-Dictated Lithium Sulfur Redox Reactions: An Operando UV–Vis Spectroscopic Study. J. Phys. Chem. Lett. 2016, 7 (8), 1518– 1525, DOI: 10.1021/acs.jpclett.6b00228Google ScholarThere is no corresponding record for this reference.
- 61Baek, M.; Shin, H.; Char, K.; Choi, J. W. New High Donor Electrolyte for Lithium–Sulfur Batteries. Adv. Mater. 2020, 32 (52), 2005022, DOI: 10.1002/adma.202005022Google ScholarThere is no corresponding record for this reference.
- 62Park, C.; Ronneburg, A.; Risse, S.; Ballauff, M.; Kanduč, M.; Dzubiella, J. Structural and Transport Properties of Li/S Battery Electrolytes: Role of the Polysulfide Species. J. Phys. Chem. C 2019, 123 (16), 10167– 10177, DOI: 10.1021/acs.jpcc.8b10175Google ScholarThere is no corresponding record for this reference.
- 63Lv, D.; Zheng, J.; Li, Q.; Xie, X.; Ferrara, S.; Nie, Z.; Mehdi, L. B.; Browning, N. D.; Zhang, J.-G.; Graff, G. L.; Liu, J.; Xiao, J. High Energy Density Lithium–Sulfur Batteries: Challenges of Thick Sulfur Cathodes. Adv. Energy Mater. 2015, 5 (16), 1402290, DOI: 10.1002/aenm.201402290Google ScholarThere is no corresponding record for this reference.
- 64Shi, L.; Bak, S.-M.; Shadike, Z.; Wang, C.; Niu, C.; Northrup, P.; Lee, H.; Baranovskiy, A. Y.; Anderson, C. S.; Qin, J.; Feng, S.; Ren, X.; Liu, D.; Yang, X.-Q.; Gao, F.; Lu, D.; Xiao, J.; Liu, J. Reaction Heterogeneity in Practical High-Energy Lithium–Sulfur Pouch Cells. Energy Environ. Sci. 2020, 13 (10), 3620– 3632, DOI: 10.1039/D0EE02088EGoogle ScholarThere is no corresponding record for this reference.
- 65Park, J.-W.; Ueno, K.; Tachikawa, N.; Dokko, K.; Watanabe, M. Ionic Liquid Electrolytes for Lithium–Sulfur Batteries. J. Phys. Chem. C 2013, 117 (40), 20531– 20541, DOI: 10.1021/jp408037eGoogle ScholarThere is no corresponding record for this reference.
- 66Park, J.-W.; Yamauchi, K.; Takashima, E.; Tachikawa, N.; Ueno, K.; Dokko, K.; Watanabe, M. Solvent Effect of Room Temperature Ionic Liquids on Electrochemical Reactions in Lithium–Sulfur Batteries. J. Phys. Chem. C 2013, 117 (9), 4431– 4440, DOI: 10.1021/jp400153mGoogle ScholarThere is no corresponding record for this reference.
- 67Zheng, D.; Zhang, X.; Li, C.; McKinnon, M. E.; Sadok, R. G.; Qu, D.; Yu, X.; Lee, H.-S.; Yang, X.-Q.; Qu, D. Quantitative Chromatographic Determination of Dissolved Elemental Sulfur in the Non-Aqueous Electrolyte for Lithium–Sulfur Batteries. J. Electrochem. Soc. 2015, 162 (1), A203, DOI: 10.1149/2.1011501jesGoogle ScholarThere is no corresponding record for this reference.
- 68Dokko, K.; Tachikawa, N.; Yamauchi, K.; Tsuchiya, M.; Yamazaki, A.; Takashima, E.; Park, J.-W.; Ueno, K.; Seki, S.; Serizawa, N.; Watanabe, M. Solvate Ionic Liquid Electrolyte for Li–S Batteries. J. Electrochem. Soc. 2013, 160 (8), A1304, DOI: 10.1149/2.111308jesGoogle ScholarThere is no corresponding record for this reference.
- 69Zhang, C.; Yamazaki, A.; Murai, J.; Park, J.-W.; Mandai, T.; Ueno, K.; Dokko, K.; Watanabe, M. Chelate Effects in Glyme/Lithium Bis(Trifluoromethanesulfonyl)Amide Solvate Ionic Liquids, Part 2: Importance of Solvate-Structure Stability for Electrolytes of Lithium Batteries. J. Phys. Chem. C 2014, 118 (31), 17362– 17373, DOI: 10.1021/jp504099qGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
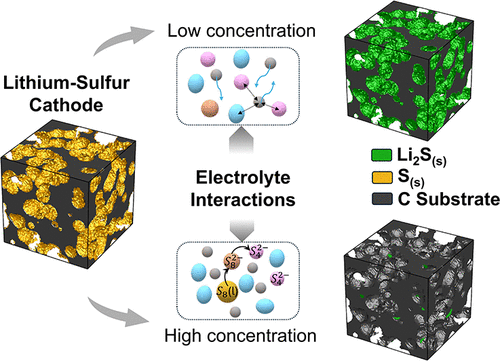
Figure 1
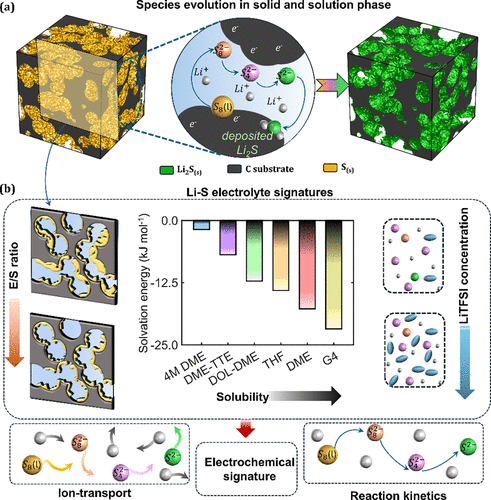
Figure 1. (a) During the Li–S discharge process, species evolution occurs within both the cathode microstructure and the electrolyte. S8(s) first dissolves into the electrolyte, followed by a multi-step electrochemical reduction at the cathode, ultimately leading to the precipitation of solid Li2S. (b) Electrolyte properties influence reaction kinetics, dissolution–precipitation behavior, and species transport in Li–S batteries. Variations in the electrolyte-to-sulfur (E/S) ratio, solvation strength, and LiTFSI concentration modify ion transport and electrochemical reaction pathways, collectively defining the electrolyte signatures governing Li–S performance. Solvation free energies are adapted from Kim et al. (37)
Figure 2
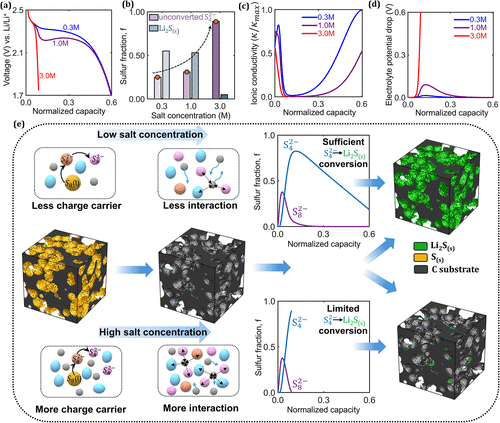
Figure 2. Simulation results illustrating the effect of the salt concentration on the Li–S cell behavior. (a) Electrochemical performance of a Li–S cell at varying salt concentrations in the electrolyte, evaluated with 80% cathode porosity and 20 vol % sulfur loading. (b) Sulfur fractions of unconverted S42– species and precipitated Li2S in the cathode at different salt concentrations. (c) Evolution of ionic conductivity during discharge for different salt concentrations, where the conductivity is normalized by the maximum ionic conductivity observed during discharge. (d) Electrolyte potential drop during discharge at different salt concentrations. (e) Evolution of PSs during discharge for different salt concentrations.
Figure 3
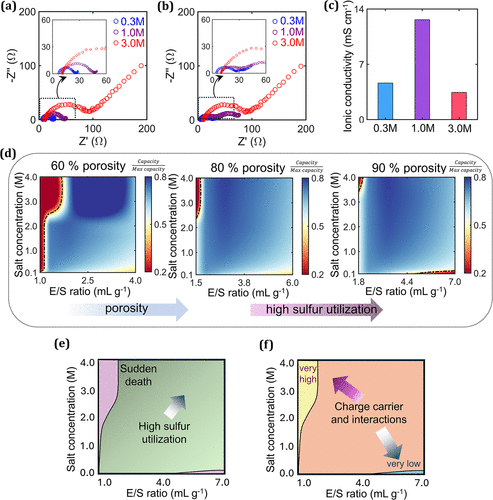
Figure 3. Nyquist plots of Li–S cells with electrolytes at different LiTFSI concentrations (a) before and (b) after discharge. (c) Experimental ionic conductivity measurement of electrolytes at different LiTFSI concentrations. (d) Simulated sulfur utilization maps as a function of the electrolyte-to-sulfur (E/S) ratio and salt concentration for pristine cathodes with different porosities (60–90 vol %). (e) Map of the salt concentration versus E/S ratio highlighting regions of high sulfur utilization and the “sudden death” zone. (f) Variation in the number of charge carriers and their interactions with changes in the salt concentration and E/S ratio.
Figure 4
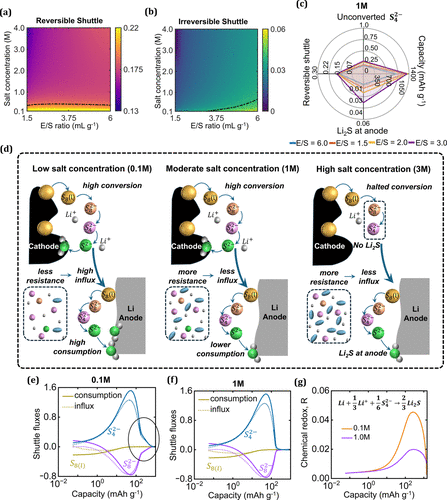
Figure 4. (a) Reversible shuttle and (b) irreversible shuttle behavior as functions of the salt concentration and E/S ratio. (c) Key performance descriptors of a Li–S battery and their dependence on the salt concentration. (d) Shuttle influx and consumption at the anode varies with the salt concentration, correlating with transport resistance. Shuttle influx and consumption calculated for (e) 0.1 M and (f) 1.0 M salt concentrations. (g) Variation of irreversible chemical redox reactions with the initial salt concentration.
Figure 5
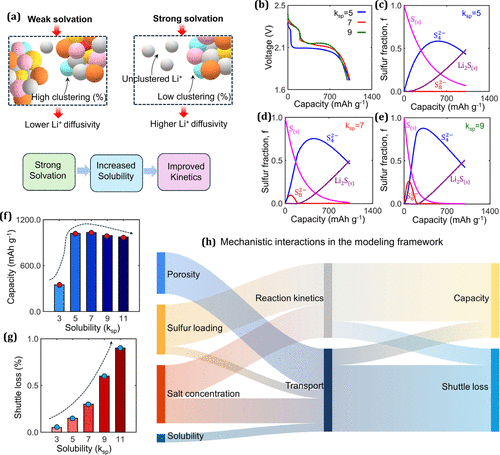
Figure 5. (a) Schematic representation depicting the correlation of solvation with Li+ diffusivity; stronger solvation enhances solubility, thereby improving reaction kinetics. (b) Electrochemical performance of the Li–S cell for different ksp values. Species evolution during discharge for electrolytes with (c) ksp = 5, (d) ksp = 7, and (e) ksp = 9. (f) Discharge capacity as a function of solubility (ksp). (g) Sulfur loss due to the shuttle effect as a function of ksp. (h) Qualitative Sankey diagram illustrating the interdependencies and multivariate impact of the cathode architecture, electrolyte properties, and solubility on the mechanistic response of Li–S batteries, including transport, reaction kinetics, capacity, and shuttle loss.
References
This article references 69 other publications.
- 1Barchasz, C.; Molton, F.; Duboc, C.; Leprêtre, J.-C.; Patoux, S.; Alloin, F. Lithium/Sulfur Cell Discharge Mechanism: An Original Approach for Intermediate Species Identification. Anal. Chem. 2012, 84 (9), 3973– 3980, DOI: 10.1021/ac2032244There is no corresponding record for this reference.
- 2Viswanathan, V.; Epstein, A. H.; Chiang, Y.-M.; Takeuchi, E.; Bradley, M.; Langford, J.; Winter, M. The Challenges and Opportunities of Battery-Powered Flight. Nature 2022, 601 (7894), 519– 525, DOI: 10.1038/s41586-021-04139-1There is no corresponding record for this reference.
- 3Yari, S.; Conde Reis, A.; Pang, Q.; Safari, M. Performance Benchmarking and Analysis of Lithium–Sulfur Batteries for next-Generation Cell Design. Nat. Commun. 2025, 16 (1), 5473, DOI: 10.1038/s41467-025-60528-4There is no corresponding record for this reference.
- 4Ji, X.; Lee, K. T.; Nazar, L. F. A Highly Ordered Nanostructured Carbon–Sulphur Cathode for Lithium–Sulphur Batteries. Nat. Mater. 2009, 8 (6), 500– 506, DOI: 10.1038/nmat2460There is no corresponding record for this reference.
- 5Elemental Sulfur and Sulfur-Rich Compounds II; Steudel, R., Ed.; Springer Science & Business Media: Berlin, Germany, 2003; DOI: 10.1007/b11909 .There is no corresponding record for this reference.
- 6Rauh, R. D.; Shuker, F. S.; Marston, J. M.; Brummer, S. B. Formation of Lithium Polysulfides in Aprotic Media. Journal of Inorganic and Nuclear Chemistry 1977, 39 (10), 1761– 1766, DOI: 10.1016/0022-1902(77)80198-XThere is no corresponding record for this reference.
- 7Zhou, G.; Chen, H.; Cui, Y. Formulating Energy Density for Designing Practical Lithium–Sulfur Batteries. Nat. Energy 2022, 7 (4), 312– 319, DOI: 10.1038/s41560-022-01001-0There is no corresponding record for this reference.
- 8Sharma, A. K.; Vishnugopi, B. S.; Alsaç, E. P.; McDowell, M. T.; Mukherjee, P. P. Passivation-Induced Species Dynamics and Microstructural Evolution in Solid-State Lithium–Sulfur Cathodes. Advanced Science 2026, e20537 DOI: 10.1002/advs.202520537There is no corresponding record for this reference.
- 9Kausthubharam; Vishnugopi, B. S.; Alujjage, A. S. J.; Premnath, V.; Tang, W. S.; Jeevarajan, J. A.; Mukherjee, P. P. Mechanistic Understanding of Thermal Stability and Safety in Lithium Metal Batteries. Chem. Rev. 2026, 126 (1), 404– 447, DOI: 10.1021/acs.chemrev.5c00621There is no corresponding record for this reference.
- 10Barchasz, C.; Leprêtre, J.-C.; Alloin, F.; Patoux, S. New Insights into the Limiting Parameters of the Li/S Rechargeable Cell. J. Power Sources 2012, 199, 322– 330, DOI: 10.1016/j.jpowsour.2011.07.021There is no corresponding record for this reference.
- 11Lai, C.; Gao, X. P.; Zhang, B.; Yan, T. Y.; Zhou, Z. Synthesis and Electrochemical Performance of Sulfur/Highly Porous Carbon Composites. J. Phys. Chem. C 2009, 113 (11), 4712– 4716, DOI: 10.1021/jp809473eThere is no corresponding record for this reference.
- 12Chen, S.-R.; Zhai, Y.-P.; Xu, G.-L.; Jiang, Y.-X.; Zhao, D.-Y.; Li, J.-T.; Huang, L.; Sun, S.-G. Ordered Mesoporous Carbon/Sulfur Nanocomposite of High Performances as Cathode for Lithium–Sulfur Battery. Electrochim. Acta 2011, 56 (26), 9549– 9555, DOI: 10.1016/j.electacta.2011.03.005There is no corresponding record for this reference.
- 13Li, X.; Cao, Y.; Qi, W.; Saraf, L. V.; Xiao, J.; Nie, Z.; Mietek, J.; Zhang, J.-G.; Schwenzer, B.; Liu, J. Optimization of Mesoporous Carbon Structures for Lithium–Sulfur Battery Applications. J. Mater. Chem. 2011, 21 (41), 16603– 16610, DOI: 10.1039/c1jm12979aThere is no corresponding record for this reference.
- 14Liang, X.; Wen, Z.; Liu, Y.; Zhang, H.; Huang, L.; Jin, J. Highly Dispersed Sulfur in Ordered Mesoporous Carbon Sphere as a Composite Cathode for Rechargeable Polymer Li/S Battery. J. Power Sources 2011, 196 (7), 3655– 3658, DOI: 10.1016/j.jpowsour.2010.12.052There is no corresponding record for this reference.
- 15Mistry, A. N.; Mukherjee, P. P. “Shuttle” in Polysulfide Shuttle: Friend or Foe?. J. Phys. Chem. C 2018, 122 (42), 23845– 23851, DOI: 10.1021/acs.jpcc.8b06077There is no corresponding record for this reference.
- 16Mistry, A.; Mukherjee, P. P. Precipitation–Microstructure Interactions in the Li–Sulfur Battery Electrode. J. Phys. Chem. C 2017, 121 (47), 26256– 26264, DOI: 10.1021/acs.jpcc.7b09997There is no corresponding record for this reference.
- 17Nahian, M. S.; Jayan, R.; Islam, M. M. Atomic-Scale Insights into Comparative Mechanisms and Kinetics of Na–S and Li–S Batteries. ACS Catal. 2022, 12 (13), 7664– 7676, DOI: 10.1021/acscatal.2c01174There is no corresponding record for this reference.
- 18Rao, M.; Song, X.; Cairns, E. J. Nano-Carbon/Sulfur Composite Cathode Materials with Carbon Nanofiber as Electrical Conductor for Advanced Secondary Lithium/Sulfur Cells. J. Power Sources 2012, 205, 474– 478, DOI: 10.1016/j.jpowsour.2012.01.047There is no corresponding record for this reference.
- 19Zheng, W.; Liu, Y. W.; Hu, X. G.; Zhang, C. F. Novel Nanosized Adsorbing Sulfur Composite Cathode Materials for the Advanced Secondary Lithium Batteries. Electrochim. Acta 2006, 51 (7), 1330– 1335, DOI: 10.1016/j.electacta.2005.06.021There is no corresponding record for this reference.
- 20Cistjakov, W.; Hoppe, J.; Jung, J.; Röder, F.; Kim, H.-T.; Krewer, U. Insight into the Impact of Electrolyte on Passivation of Lithium–Sulfur Cathodes. Advanced Materials Interfaces 2025, 12 (5), 2400632, DOI: 10.1002/admi.202400632There is no corresponding record for this reference.
- 21Liu, Z.; Mukherjee, P. P. Mesoscale Elucidation of Surface Passivation in the Li–Sulfur Battery Cathode. ACS Appl. Mater. Interfaces 2017, 9 (6), 5263– 5271, DOI: 10.1021/acsami.6b15066There is no corresponding record for this reference.
- 22Nahian, M. S.; Sharma, A. K.; Vishnugopi, B. S. Precipitate-Driven Electrochemical Interactions in Lithium–Sulfur Batteries. J. Electrochem. En. Conv. Stor 2026, 23, 024704, DOI: 10.1115/1.4070879There is no corresponding record for this reference.
- 23Jana, M.; Xu, R.; Cheng, X.-B.; Yeon, J. S.; Park, J. M.; Huang, J.-Q.; Zhang, Q.; Park, H. S. Rational Design of Two-Dimensional Nanomaterials for Lithium–Sulfur Batteries. Energy Environ. Sci. 2020, 13 (4), 1049– 1075, DOI: 10.1039/C9EE02049GThere is no corresponding record for this reference.
- 24Lin, H.; Yang, L.; Jiang, X.; Li, G.; Zhang, T.; Yao, Q.; Zheng, G. W.; Lee, J. Y. Electrocatalysis of Polysulfide Conversion by Sulfur-Deficient MoS2 Nanoflakes for Lithium–Sulfur Batteries. Energy Environ. Sci. 2017, 10 (6), 1476– 1486, DOI: 10.1039/C7EE01047HThere is no corresponding record for this reference.
- 25Yuan, Z.; Peng, H.-J.; Huang, J.-Q.; Liu, X.-Y.; Wang, D.-W.; Cheng, X.-B.; Zhang, Q. Hierarchical Free-Standing Carbon-Nanotube Paper Electrodes with Ultrahigh Sulfur-Loading for Lithium–Sulfur Batteries. Adv. Funct. Mater. 2014, 24 (39), 6105– 6112, DOI: 10.1002/adfm.201401501There is no corresponding record for this reference.
- 26Fan, F. Y.; Carter, W. C.; Chiang, Y.-M. Mechanism and Kinetics of Li2S Precipitation in Lithium–Sulfur Batteries. Adv. Mater. 2015, 27 (35), 5203– 5209, DOI: 10.1002/adma.201501559There is no corresponding record for this reference.
- 27Kim, Y.; Kim, W. I.; Park, H.; Kim, J. S.; Cho, H.; Yeon, J. S.; Kim, J.; Kim, Y.-J.; Lee, J.; Park, H. S. Multifunctional Polymeric Phthalocyanine-Coated Carbon Nanotubes for Efficient Redox Mediators of Lithium–Sulfur Batteries. Adv. Energy Mater. 2023, 13 (22), 2204353, DOI: 10.1002/aenm.202204353There is no corresponding record for this reference.
- 28Kim, W. I.; Shin, J. C.; Kim, M. J.; Jang, G.; Lee, M.; Park, H. S. Ionic Structured Redox-Mediating Polymeric Sulfurs for Lithium–Sulfur Batteries. ACS Energy Lett. 2025, 10 (5), 2410– 2418, DOI: 10.1021/acsenergylett.5c00808There is no corresponding record for this reference.
- 29Liang, X.; Hart, C.; Pang, Q.; Garsuch, A.; Weiss, T.; Nazar, L. F. A Highly Efficient Polysulfide Mediator for Lithium–Sulfur Batteries. Nat. Commun. 2015, 6 (1), 5682, DOI: 10.1038/ncomms6682There is no corresponding record for this reference.
- 30Liu, Y.; Elias, Y.; Meng, J.; Aurbach, D.; Zou, R.; Xia, D.; Pang, Q. Electrolyte Solutions Design for Lithium–Sulfur Batteries. Joule 2021, 5 (9), 2323– 2364, DOI: 10.1016/j.joule.2021.06.009There is no corresponding record for this reference.
- 31Guo, J.; Xu, Y.; Wang, C. Sulfur-Impregnated Disordered Carbon Nanotubes Cathode for Lithium–Sulfur Batteries. Nano Lett. 2011, 11 (10), 4288– 4294, DOI: 10.1021/nl202297pThere is no corresponding record for this reference.
- 32Tarascon, J.-M.; Armand, M. Issues and Challenges Facing Rechargeable Lithium Batteries. Nature 2001, 414 (6861), 359– 367, DOI: 10.1038/35104644There is no corresponding record for this reference.
- 33Bruce, P. G.; Freunberger, S. A.; Hardwick, L. J.; Tarascon, J.-M. Li–O2 and Li–S Batteries with High Energy Storage. Nat. Mater. 2012, 11 (1), 19– 29, DOI: 10.1038/nmat3191There is no corresponding record for this reference.
- 34Goodenough, J. B.; Park, K.-S. The Li-Ion Rechargeable Battery: A Perspective. J. Am. Chem. Soc. 2013, 135 (4), 1167– 1176, DOI: 10.1021/ja3091438There is no corresponding record for this reference.
- 35Zhang, B.; Wu, J.; Gu, J.; Li, S.; Yan, T.; Gao, X.-P. The Fundamental Understanding of Lithium Polysulfides in Ether-Based Electrolyte for Lithium–Sulfur Batteries. ACS Energy Lett. 2021, 6 (2), 537– 546, DOI: 10.1021/acsenergylett.0c02527There is no corresponding record for this reference.
- 36Mistry, A. N.; Mukherjee, P. P. Electrolyte Transport Evolution Dynamics in Lithium–Sulfur Batteries. J. Phys. Chem. C 2018, 122 (32), 18329– 18335, DOI: 10.1021/acs.jpcc.8b05442There is no corresponding record for this reference.
- 37Kim, S. C.; Gao, X.; Liao, S.-L.; Su, H.; Chen, Y.; Zhang, W.; Greenburg, L. C.; Pan, J.-A.; Zheng, X.; Ye, Y.; Kim, M. S.; Sayavong, P.; Brest, A.; Qin, J.; Bao, Z.; Cui, Y. Solvation-Property Relationship of Lithium–Sulphur Battery Electrolytes. Nat. Commun. 2024, 15 (1), 1268, DOI: 10.1038/s41467-023-44527-xThere is no corresponding record for this reference.
- 38Huang, Y.; Shaibani, M.; Abedin, Md. J.; Mendoza, D. J.; Xu, Z.; Gamot, T. D.; Cooray, M. C. D.; Lin, M.; Garnier, G.; Hill, M. R.; Majumder, M. Sulfur Cathodes with Self-Organized Cellulose Nanofibers in Stable Ah-Level, > 300 Wh kg–1 Lithium–Sulfur Cells. Adv. Energy Mater. 2022, 12 (45), 2202474, DOI: 10.1002/aenm.202202474There is no corresponding record for this reference.
- 39Chung, S.-H.; Chang, C.-H.; Manthiram, A. Progress on the Critical Parameters for Lithium–Sulfur Batteries to Be Practically Viable. Adv. Funct. Mater. 2018, 28 (28), 1801188, DOI: 10.1002/adfm.201801188There is no corresponding record for this reference.
- 40Li, Z.; Rao, H.; Atwi, R.; Sivakumar, B. M.; Gwalani, B.; Gray, S.; Han, K. S.; Everett, T. A.; Ajantiwalay, T. A.; Murugesan, V.; Rajput, N. N.; Pol, V. G. Non-Polar Ether-Based Electrolyte Solutions for Stable High-Voltage Non-Aqueous Lithium Metal Batteries. Nat. Commun. 2023, 14 (1), 868, DOI: 10.1038/s41467-023-36647-1There is no corresponding record for this reference.
- 41Cheng, Q.; Chen, Z.-X.; Li, X.-Y.; Hou, L.-P.; Bi, C.-X.; Zhang, X.-Q.; Huang, J.-Q.; Li, B.-Q. Constructing a 700 Wh kg–1-Level Rechargeable Lithium–Sulfur Pouch Cell. Journal of Energy Chemistry 2023, 76, 181– 186, DOI: 10.1016/j.jechem.2022.09.029There is no corresponding record for this reference.
- 42Hagen, M.; Hanselmann, D.; Ahlbrecht, K.; Maça, R.; Gerber, D.; Tübke, J. Lithium–Sulfur Cells: The Gap between the State-of-the-Art and the Requirements for High Energy Battery Cells. Adv. Energy Mater. 2015, 5 (16), 1401986, DOI: 10.1002/aenm.201401986There is no corresponding record for this reference.
- 43Jozwiuk, A.; Berkes, B. B.; Weiß, T.; Sommer, H.; Janek, J.; Brezesinski, T. The Critical Role of Lithium Nitrate in the Gas Evolution of Lithium–Sulfur Batteries. Energy Environ. Sci. 2016, 9 (8), 2603– 2608, DOI: 10.1039/C6EE00789AThere is no corresponding record for this reference.
- 44Cleaver, T.; Kovacik, P.; Marinescu, M.; Zhang, T.; Offer, G. Perspective─Commercializing Lithium Sulfur Batteries: Are We Doing the Right Research?. J. Electrochem. Soc. 2018, 165 (1), A6029, DOI: 10.1149/2.0071801jesThere is no corresponding record for this reference.
- 45Rajput, N. N.; Murugesan, V.; Shin, Y.; Han, K. S.; Lau, K. C.; Chen, J.; Liu, J.; Curtiss, L. A.; Mueller, K. T.; Persson, K. A. Elucidating the Solvation Structure and Dynamics of Lithium Polysulfides Resulting from Competitive Salt and Solvent Interactions. Chem. Mater. 2017, 29 (8), 3375– 3379, DOI: 10.1021/acs.chemmater.7b00068There is no corresponding record for this reference.
- 46Peng, H.-J.; Huang, J.-Q.; Cheng, X.-B.; Zhang, Q. Review on High-Loading and High-Energy Lithium–Sulfur Batteries. Adv. Energy Mater. 2017, 7 (24), 1700260, DOI: 10.1002/aenm.201700260There is no corresponding record for this reference.
- 47Jarrold, G.; Manthiram, A. Electrolyte Strategies for Practically Viable All-Solid-State Lithium–Sulfur Batteries. Commun. Mater. 2025, 6 (1), 240, DOI: 10.1038/s43246-025-00960-7There is no corresponding record for this reference.
- 48Zheng, J.; Lv, D.; Gu, M.; Wang, C.; Zhang, J.-G.; Liu, J.; Xiao, J. How to Obtain Reproducible Results for Lithium Sulfur Batteries?. J. Electrochem. Soc. 2013, 160 (11), A2288, DOI: 10.1149/2.106311jesThere is no corresponding record for this reference.
- 49Nagpure, S. C.; Tanim, T. R.; Dufek, E. J.; Viswanathan, V. V.; Crawford, A. J.; Wood, S. M.; Xiao, J.; Dickerson, C. C.; Liaw, B. Impacts of Lean Electrolyte on Cycle Life for Rechargeable Li Metal Batteries. J. Power Sources 2018, 407, 53– 62, DOI: 10.1016/j.jpowsour.2018.10.060There is no corresponding record for this reference.
- 50Cheng, L.; Curtiss, L. A.; Zavadil, K. R.; Gewirth, A. A.; Shao, Y.; Gallagher, K. G. Sparingly Solvating Electrolytes for High Energy Density Lithium–Sulfur Batteries. ACS Energy Lett. 2016, 1 (3), 503– 509, DOI: 10.1021/acsenergylett.6b00194There is no corresponding record for this reference.
- 51Deng, D. R.; Xue, F.; Bai, C.-D.; Lei, J.; Yuan, R.; Zheng, M. S.; Dong, Q. F. Enhanced Adsorptions to Polysulfides on Graphene-Supported BN Nanosheets with Excellent Li–S Battery Performance in a Wide Temperature Range. ACS Nano 2018, 12 (11), 11120– 11129, DOI: 10.1021/acsnano.8b05534There is no corresponding record for this reference.
- 52Zhang, G.; Peng, H.-J.; Zhao, C.-Z.; Chen, X.; Zhao, L.-D.; Li, P.; Huang, J.-Q.; Zhang, Q. The Radical Pathway Based on a Lithium-Metal-Compatible High-Dielectric Electrolyte for Lithium–Sulfur Batteries. Angew. Chem. 2018, 130 (51), 16974– 16978, DOI: 10.1002/ange.201810132There is no corresponding record for this reference.
- 53Li, G.; Wang, S.; Zhang, Y.; Li, M.; Chen, Z.; Lu, J. Revisiting the Role of Polysulfides in Lithium–Sulfur Batteries. Adv. Mater. 2018, 30 (22), 1705590, DOI: 10.1002/adma.201705590There is no corresponding record for this reference.
- 54Han, P.; Chung, S.-H.; Manthiram, A. Pyrrolic-Type Nitrogen-Doped Hierarchical Macro/Mesoporous Carbon as a Bifunctional Host for High-Performance Thick Cathodes for Lithium–Sulfur Batteries. Small 2019, 15 (16), 1900690, DOI: 10.1002/smll.201900690There is no corresponding record for this reference.
- 55Thieme, S.; Brückner, J.; Meier, A.; Bauer, I.; Gruber, K.; Kaspar, J.; Helmer, A.; Althues, H.; Schmuck, M.; Kaskel, S. A Lithium–Sulfur Full Cell with Ultralong Cycle Life: Influence of Cathode Structure and Polysulfide Additive. Journal of Materials Chemistry A 2015, 3 (7), 3808– 3820, DOI: 10.1039/C4TA06748GThere is no corresponding record for this reference.
- 56Chen, S.; Dai, F.; Gordin, M. L.; Yu, Z.; Gao, Y.; Song, J.; Wang, D. Functional Organosulfide Electrolyte Promotes an Alternate Reaction Pathway to Achieve High Performance in Lithium–Sulfur Batteries. Angew. Chem., Int. Ed. 2016, 55 (13), 4231– 4235, DOI: 10.1002/anie.201511830There is no corresponding record for this reference.
- 57Park, C.; Kanduč, M.; Chudoba, R.; Ronneburg, A.; Risse, S.; Ballauff, M.; Dzubiella, J. Molecular Simulations of Electrolyte Structure and Dynamics in Lithium–Sulfur Battery Solvents. J. Power Sources 2018, 373, 70– 78, DOI: 10.1016/j.jpowsour.2017.10.081There is no corresponding record for this reference.
- 58Chen, S.; Wang, D.; Zhao, Y.; Wang, D. Superior Performance of a Lithium–Sulfur Battery Enabled by a Dimethyl Trisulfide Containing Electrolyte. Small Methods 2018, 2 (6), 1800038, DOI: 10.1002/smtd.201800038There is no corresponding record for this reference.
- 59Chu, H.; Noh, H.; Kim, Y.-J.; Yuk, S.; Lee, J.-H.; Lee, J.; Kwack, H.; Kim, Y.; Yang, D.-K.; Kim, H.-T. Achieving Three-Dimensional Lithium Sulfide Growth in Lithium–Sulfur Batteries Using High-Donor-Number Anions. Nat. Commun. 2019, 10 (1), 188, DOI: 10.1038/s41467-018-07975-4There is no corresponding record for this reference.
- 60Zou, Q.; Lu, Y.-C. Solvent-Dictated Lithium Sulfur Redox Reactions: An Operando UV–Vis Spectroscopic Study. J. Phys. Chem. Lett. 2016, 7 (8), 1518– 1525, DOI: 10.1021/acs.jpclett.6b00228There is no corresponding record for this reference.
- 61Baek, M.; Shin, H.; Char, K.; Choi, J. W. New High Donor Electrolyte for Lithium–Sulfur Batteries. Adv. Mater. 2020, 32 (52), 2005022, DOI: 10.1002/adma.202005022There is no corresponding record for this reference.
- 62Park, C.; Ronneburg, A.; Risse, S.; Ballauff, M.; Kanduč, M.; Dzubiella, J. Structural and Transport Properties of Li/S Battery Electrolytes: Role of the Polysulfide Species. J. Phys. Chem. C 2019, 123 (16), 10167– 10177, DOI: 10.1021/acs.jpcc.8b10175There is no corresponding record for this reference.
- 63Lv, D.; Zheng, J.; Li, Q.; Xie, X.; Ferrara, S.; Nie, Z.; Mehdi, L. B.; Browning, N. D.; Zhang, J.-G.; Graff, G. L.; Liu, J.; Xiao, J. High Energy Density Lithium–Sulfur Batteries: Challenges of Thick Sulfur Cathodes. Adv. Energy Mater. 2015, 5 (16), 1402290, DOI: 10.1002/aenm.201402290There is no corresponding record for this reference.
- 64Shi, L.; Bak, S.-M.; Shadike, Z.; Wang, C.; Niu, C.; Northrup, P.; Lee, H.; Baranovskiy, A. Y.; Anderson, C. S.; Qin, J.; Feng, S.; Ren, X.; Liu, D.; Yang, X.-Q.; Gao, F.; Lu, D.; Xiao, J.; Liu, J. Reaction Heterogeneity in Practical High-Energy Lithium–Sulfur Pouch Cells. Energy Environ. Sci. 2020, 13 (10), 3620– 3632, DOI: 10.1039/D0EE02088EThere is no corresponding record for this reference.
- 65Park, J.-W.; Ueno, K.; Tachikawa, N.; Dokko, K.; Watanabe, M. Ionic Liquid Electrolytes for Lithium–Sulfur Batteries. J. Phys. Chem. C 2013, 117 (40), 20531– 20541, DOI: 10.1021/jp408037eThere is no corresponding record for this reference.
- 66Park, J.-W.; Yamauchi, K.; Takashima, E.; Tachikawa, N.; Ueno, K.; Dokko, K.; Watanabe, M. Solvent Effect of Room Temperature Ionic Liquids on Electrochemical Reactions in Lithium–Sulfur Batteries. J. Phys. Chem. C 2013, 117 (9), 4431– 4440, DOI: 10.1021/jp400153mThere is no corresponding record for this reference.
- 67Zheng, D.; Zhang, X.; Li, C.; McKinnon, M. E.; Sadok, R. G.; Qu, D.; Yu, X.; Lee, H.-S.; Yang, X.-Q.; Qu, D. Quantitative Chromatographic Determination of Dissolved Elemental Sulfur in the Non-Aqueous Electrolyte for Lithium–Sulfur Batteries. J. Electrochem. Soc. 2015, 162 (1), A203, DOI: 10.1149/2.1011501jesThere is no corresponding record for this reference.
- 68Dokko, K.; Tachikawa, N.; Yamauchi, K.; Tsuchiya, M.; Yamazaki, A.; Takashima, E.; Park, J.-W.; Ueno, K.; Seki, S.; Serizawa, N.; Watanabe, M. Solvate Ionic Liquid Electrolyte for Li–S Batteries. J. Electrochem. Soc. 2013, 160 (8), A1304, DOI: 10.1149/2.111308jesThere is no corresponding record for this reference.
- 69Zhang, C.; Yamazaki, A.; Murai, J.; Park, J.-W.; Mandai, T.; Ueno, K.; Dokko, K.; Watanabe, M. Chelate Effects in Glyme/Lithium Bis(Trifluoromethanesulfonyl)Amide Solvate Ionic Liquids, Part 2: Importance of Solvate-Structure Stability for Electrolytes of Lithium Batteries. J. Phys. Chem. C 2014, 118 (31), 17362– 17373, DOI: 10.1021/jp504099qThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsenergylett.6c00391.
Computational framework to predict electrochemical performance (section S1) and experimental methods and results (section S2) (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



