Itaconic Acid–Based Compounds Fulfilling the Role of Biotechnologically Produced Alternatives to Fossil-Based Precursors for Material ApplicationsClick to copy article linkArticle link copied!
- Vojtěch Jašek*Vojtěch Jašek*Email: [email protected]Institute of Materials Chemistry, Faculty of Chemistry, Brno University of Technology, 61200 Brno, Czech RepublicMore by Vojtěch Jašek
- Silvestr FigallaSilvestr FigallaInstitute of Materials Chemistry, Faculty of Chemistry, Brno University of Technology, 61200 Brno, Czech RepublicMore by Silvestr Figalla
- Radek PřikrylRadek PřikrylInstitute of Materials Chemistry, Faculty of Chemistry, Brno University of Technology, 61200 Brno, Czech RepublicMore by Radek Přikryl
Abstract
Itaconic acid (IA) attracts significant attention in several application fields, including the materials industry, due to its solid-state form, which is associated with lower volatility and toxicity, and its exceptional potential for chemical functionalization. This debate presents numerous positive aspects of itaconate-based materials for specific applications, such as additive manufacturing, composite fabrication, coating production, adhesive synthesis, textile additives incorporation, and particular engineering for hydrogels. We evaluate many of the efficient viewpoints and advantages itaconic acid offers for the currently engineered systems that use fossil-based compounds and precursors. Unlike similar functional molecules and species, such as acrylates, methacrylates, or vinyl esters, itaconic acid’s liquid derivatives exhibit negligible volatility, thereby reducing potential health hazards and VOC emissions. Moreover, countless derivatives can be proposed and synthesized when appropriate nucleophiles are selected (e.g., alcohols, amines, and others). Due to the two vacant carboxyl acid groups (−COOH), itaconic acid can be incorporated into multicomponent polyesters, polyethers, polyamides, or other functional systems to enhance their performance, improve processability, or ensure degradability. Finally, itaconates provide a route to biocompatible and degradable/biodegradable systems that can develop into new, unique compounds suitable for specific materials.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
Figure 1
Figure 1. Graphical illustration of the itaconic acid’s molecular structure contains two carboxylic functional groups exhibiting the reactive delocalized electrons in resonance, a radically polymerizable terminal alkene functional group, and the schematic potential functional derivatives, such as amides (derived from amines and acids), esters (derived from alcohols and acids), and anhydride (derived from two acids).
| 1. | Reactive Precursors for Additive Manufacturing (3D printing); | ||||
| 2. | Curable Matrixes for Composite Fabrication; | ||||
| 3. | Entering Compounds for the Functional Polymeric Coatings; | ||||
| 4. | Molecular Substances for Adhesives; | ||||
| 5. | Initial Representatives for Textile Additives; | ||||
| 6. | Assisting Monomers for Hydrogel Fabrication. | ||||
2. Precursors for 3D Printing
Figure 2
Figure 2. Primary parameters influencing the stereolithography 3D printing. (a) Irradiation source type (the characteristics wavelength and power value), (b) the specific reactivity of the precursor (determined by Jacob’s working curve or differential scanning calorimetry (DSC)), (c) the rheological profile of the resin-forming systems (dependent on the temperature by the Arrhenius law and affected by the reactive diluent’s content) (d) the published diluting properties of the experimentally applied itaconate derivaties. (84) Reproduced from ref (84). Copyright [2017] American Chemical Society.
2.1. Itaconic Acid-Derived Polyester Resins
Figure 3
Figure 3. (a) Itaconic acid–based polyester used in combination with 4-acryloyl morpholine (ACMO) as a reactive diluent, (60) Reproduced from ref (60). Copyright [2023] American Chemical Society (b) oligomer itaconate precursor systems containing biobased reactive diluents derived from furfuryl, tetrahydrofurfuryl, solketal, and diacetone glucose reactive derivatives, (63) Reproduced from ref (63). Copyright [2025] American Chemical Society (c) Polylactone/itaconate elastomer resins processed by reversible addition–fragmentation chain transfer (RAFT) mediated 3D-printing with polypeptide surface functionalization, (67) Reproduced from ref (67). Copyright [2023] American Chemical Society (d) modified grapheme oxide (GO) nanoparticles incorporated into a itaconate-based 3D printing resin for the enhancement of the material properties while maintaining the advanced functional properties ensured by GO’s the conjugated π-electron system. (66) Reproduced from ref (66). Copyright [2025] American Chemical Society.
| polyester itaconate-based precursors | |||||
|---|---|---|---|---|---|
| application | carboxyl acids (derivative) | polyol (derivative) | renewable content | additive for 3D printing | refs |
| 3D Printed Nanocomposite | Itaconic Acid, Sebacic Acid | 1,3-Propanediol | 50 wt % | Reactive Diluent (ACMO) | (68) |
| Hydrolyzable Sustainable Resin | Itaconic Acid, 1,12-Dodecanedioic Acid | 1,3-Propanediol | ≈99 wt % | Processed Without 3D Printing | (69) |
| Vat Polymerization | Monomethyl Itaconoyl Chloride, Caprolactone | Sorbitol | 29–97 wt % | (Meth)acrylate Reactive Diluents | (70) |
| 3D Printing Resin | Itaconic Acid, Succinic Acid | 1,2-Propanediol, 1,4-Butanediol, 1,8-Octanediol | 50 wt % | Reactive Diluent (TEGDMA) | (71) |
| Extrusion-Based 3D Printing | Itaconic Acid, Succinic Anhydride | 1,8-Octanediol | Not Available | Processed Without 3D Printing | (72) |
| Phosphorescent 3D Printing Resin | Itaconic Acid, Vanillic Acid | Glycerol, 1,3-Propanediol | 48.5 wt % | Reactive Diluent (THC, BHI) | (73) |
| 3D Printing Resin | Itaconic Acid, Dimethyl Terephthalate | Ethylene Glycol, 1,3-Propanediol, 1,4-Butanediol, 1,5-Pentanediol, 1,2-Propanediol, 2-Methyl-1,3-Propanediol, 1,3-Butanediol, 3-Methyl-1,5-Pentanediol | 48–57 wt % | Reactive Diluent (Styrene) | (74) |
2.2. Other Curable Itaconate Resins
Figure 4
2.3. Itaconate-Based Reactive Diluents
Figure 5
Figure 5. Complex synthetic scheme composed of reactions described and evaluated in the investigation provided by Arnaud and the team. (59) The scheme describes three different chemical approaches leading to the symmetric esters produced via Fischer esterification, asymmetric esters combining anhydride and epoxide compounds, and symmetric esters derived from epoxy entering compounds. Reproduced from ref (59). Copyright [2021] American Chemical Society.
Figure 6
Figure 6. Illustrated polyester itaconic acid–based cured resins, modified with selected itaconate-reactive diluents, fabricated through 3D printing. (a) 3D-printed DMA specimen containing dicyclohexyl itaconate (DE-1), (b) 3D-printed DMA specimen containing bis(2-hydroxybutyl) itaconate (DHE-1). (59) Reproduced from ref (59). Copyright [2021] American Chemical Society.
The significantly lower reactivity of itaconates represents an obstacle to efficient 3D printing at a commercially attractive rate. Low-molecular-weight itaconate derivatives can solve this issue;
The lower degrees of cure of the itaconate polymers often result in decreases in several mechanical properties. Structural engineering of the precursor may lead to an efficient solution;
The rheology levels of itaconate oligomers complicate the 3D printing process. Their adjustment has to be considered.
3. Matrix for Composites
3.1. Carbon Fiber Composites
3.2. Natural Itaconate Composites
Figure 7
Figure 7. Schematic illustration of the proposed fully biobased composite from itaconate resin and cotton fabrics as the solid-phase filler along with thermo-mechanical properties measured by the DMA analysis. (a–e) SEM pictures of the obtained composite prototype, (f) the photo of the fabricated composite. (100) Reproduced from ref (100). Copyright [2018] American Chemical Society.
The polymerization reactivity of itaconates. A suitable cross-linker or reactivity can solve this issue;
The reinforcement-matrix compatibility from the chemical structure standpoint. The itaconic acid–based system must be engineered to address this potential problem;
Suitable surface modification of the natural fibers before material fabrication. The compatibility of any resin (including the itaconate) may be problematic.
4. Functional Coatings
Figure 8
Figure 8. Schematic list of potential and investigated application fields for itaconic acid–based polymeric coatings for material applications. The illustrated reaction scheme describes the modification of epoxidized soybean oil (ESO) with itaconic acid monoester, yielding itaconated epoxidized soybean oil (IESO), which represents a green alternative to the commercially used acrylated epoxidized soybean oil (AESO) for general coating applications. (101,102) Reproduced from ref (101) Copyright [2016] American Chemical Society and from ref (101) Copyright [2019] American Chemical Society.
4.1. Bio-Based Alternative Itaconate Coatings
| itaconate coating precursors | ||||
|---|---|---|---|---|
| type of the system | itaconic acid function | renewable content | suggested application | refs |
| Polyester | Cross-linker and Group Modifier | Unspecified | Encapsulation Resin | (111) |
| Epoxy Resin | Main Carbon Backbone | 40–60 wt % | Wood Protective Coating | (112) |
| Polyester | Carbon Backbone | 95–98 wt % | Bio-Based Coating Alternative | (113) |
| Polyurethane | Polyol Component | 30–45 wt % | Bio-Based Coating Alternative | (114) |
| Polyester | Cross-linker | 67–95 wt % | UV-Curable Biobased Coating | (115) |
4.2. Functional Itaconate Coatings
Figure 9
Figure 9. Synthesis of the deprotected zwitterionic itaconic acid involving copolymer (IA-BP-PZI) and the coating strategy ensuring the antimicrobial activity, protein repellency, and cell compatibility investigated by Schneider-Chaabane et al. (118) Reproduced from ref (118). Copyright [2019] American Chemical Society.
High viscosity levels of itaconate derivatives. The coating industry relies heavily on precisely defined rheological behavior;
The reactivity and degree of cure must be considered and affected by a specific itaconic acid derivative or other suitable additive.
5. Adhesive Precursors
Figure 10
Figure 10. Schematic interpretation of itaconate-based photocurable adhesive investigated by Jamaludin et al., (119) and an illustration of the forming (and potential) noncovalent molecular interactions, such as hydrogen bonding, dispersion forces, and π–π stacking. (120−122) Reproduced from ref (119) Copyright [2022] American Chemical Society, from ref (120) Copyright [2023] American Chemical Society, from ref (121) Copyright [2023] American Chemical Society, and from ref (122) Copyright [2016] American Chemical Society.
5.1. (Photo)Curable Thermoset Adhesives
Figure 11
5.2. Pressure-Sensitive Adhesives (PSA)
Figure 12
Figure 12. (a) Highly crosslinked itaconic acid derivative for PSA, (b) itaconic acid ester system enhanced with acrylic derivatives for PSA. Investigated pressure-sensitive adhesives (PSA) containing itaconic acid and its derivatives. (147) Copyright (2024), American Chemical Society. The multicomponent cross-linkable PSA synthesized from sustainable sources is illustrated in the bottom-left corner, (145) while the underwater-performing PSAs for potential wound treatment are displayed in the right half. (146) Reproduced from ref (145) copyright [2025] American Chemical Society and from ref (146) copyright [2024] American Chemical Society.
5.3. Starch-Itaconate-Based Adhesives
A sufficient linear polymerization stage must be achieved to achieve the required adhesive performance. Also, the precisely engineered itaconate structure contributes to the overall functional performance;
The optical properties of the used precursors must be taken into account, as photocuring applications often require highly transparent precursors, which can be challenging to achieve in biobased systems.
6. Functional Textile Additives
Figure 13
7. Assisting Monomer for Hydrogels
Figure 14
Figure 14. Two particular approaches for incorporating sustainable itaconic acid into the hydrogel structures. (a) The presynthesized itaconic monoesters serve as reactive molecules that can modify the structure of the primary hydrogel site. (154) Reproduced from ref (154). Copyright [2024] American Chemical Society. (b) Pure itaconic acid can be esterified onto the main polymer chain, providing the required properties. (153) Reproduced from ref (153). Copyright [2023] American Chemical Society.
The suitable substitution of another dicarboxylic acid with itaconic acid must be compatible with the hydrogel substrates (if the specific hydrogel requires it);
The viscoelastic properties of the produced hydrogels must meet the required levels. Itaconic acid may influence the reactivity and, in particular, the rheological profile of the produced hydrogel.
8. Conclusion and Future Outlook
Acknowledgments
V.J. acknowledges the financial support from the Ministry of Education, Youth and Sport of the Czech Republic (project No. FCH-S-26-9028).
References
This article references 171 other publications.
- 1Cywar, R. M.; Rorrer, N. A.; Hoyt, C. B.; Beckham, G. T.; Chen, E. Y.-X. Bio-Based Polymers With Performance-Advantaged Properties. Nat. Rev. Mater. 2022, 7 (2), 83– 103, DOI: 10.1038/s41578-021-00363-3Google ScholarThere is no corresponding record for this reference.
- 2Álvarez-Chávez, C. R.; Edwards, S.; Moure-Eraso, R.; Geiser, K. Sustainability Of Bio-Based Plastics: General Comparative Analysis And Recommendations For Improvement. J. Cleaner Prod. 2012, 23 (1), 47– 56, DOI: 10.1016/j.jclepro.2011.10.003Google ScholarThere is no corresponding record for this reference.
- 3Lee, J.-Y.; Lee, S.-E.; Lee, D.-W. Current Status And Future Prospects Of Biological Routes To Bio-Based Products Using Raw Materials, Wastes, And Residues As Renewable Resources. Crit. Rev. Environ. Sci. Technol. 2022, 52 (14), 2453– 2509, DOI: 10.1080/10643389.2021.1880259Google ScholarThere is no corresponding record for this reference.
- 4Wang, Z. Statistical Analysis Of The Structure Of Total Petroleum Consumption In China. IOP Conf. Ser.: Earth Environ. Sci. 2019, 300 (4), 042089 DOI: 10.1088/1755-1315/300/4/042089Google ScholarThere is no corresponding record for this reference.
- 5Patidar, A. K.; Jain, P.; Dhasmana, P.; Choudhury, T. Impact Of Global Events On Crude Oil Economy: A Comprehensive Review Of The Geopolitics Of Energy And Economic Polarization. GeoJournal 2024, 89 (2), 50 DOI: 10.1007/s10708-024-11054-1Google ScholarThere is no corresponding record for this reference.
- 6Soto, J. M.; Martín-Lara, M. A.; Blázquez, G.; Godoy, V.; Quesada, L.; Calero, M. Novel Pre-Treatment Of Dirty Post-Consumer Polyethylene Film For Its Mechanical Recycling. Process Saf. Environ. Prot. 2020, 139, 315– 324, DOI: 10.1016/j.psep.2020.04.044Google ScholarThere is no corresponding record for this reference.
- 7Streit, A. F. M.; de Santana, M. P.; de Oliveira Júnior, D. L.; Bassaco, M. M.; Tanabe, E. H.; Dotto, G. L.; Bertuol, D. A. Development Of A Pre-Treatment Process Of Polymeric Wastes (Hdpe, Ldpe/Lldpe, Pp) For Application In The Qualification Of Selectors Of Recyclable Materials. Environ. Dev. Sustainability 2022, 24 (5), 6349– 6371, DOI: 10.1007/s10668-021-01705-5Google ScholarThere is no corresponding record for this reference.
- 8Zhang, X.; Zu, W.; Lee, L. Y. S. Crucial Role Of Pre-Treatment In Plastic Photoreforming For Precision Upcycling. npj Mater. Sustainability 2025, 3 (1), 3 DOI: 10.1038/s44296-024-00045-5Google ScholarThere is no corresponding record for this reference.
- 9Schade, A.; Melzer, M.; Zimmermann, S.; Schwarz, T.; Stöwe, K.; Kühn, H. Plastic Waste Recycling─A Chemical Recycling Perspective. ACS Sustainable Chem. Eng. 2024, 12 (33), 12270– 12288, DOI: 10.1021/acssuschemeng.4c02551Google ScholarThere is no corresponding record for this reference.
- 10Sandoval, S. S.; Amenábar, A.; Toledo, I.; Silva, N.; Contreras, P. Advances In The Sustainable Development Of Biobased Materials Using Plant And Animal Waste As Raw Materials: A Review. Sustainability 2024, 16 (3), 1073 DOI: 10.3390/su16031073Google ScholarThere is no corresponding record for this reference.
- 11Rajput, C. V.; Sastry, N.V.; Chikhaliya, N. P. Vegetable Oils Based Precursors: Modifications And Scope For Futuristic Bio-Based Polymeric Materials. J. Polym. Res. 2023, 30 (4), 159 DOI: 10.1007/s10965-023-03534-8Google ScholarThere is no corresponding record for this reference.
- 12Wahlen, C.; Frey, H. Anionic Polymerization Of Terpene Monomers: New Options For Bio-Based Thermoplastic Elastomers. Macromolecules 2021, 54 (16), 7323– 7336, DOI: 10.1021/acs.macromol.1c00770Google ScholarThere is no corresponding record for this reference.
- 13Zhu, Y.; Romain, C.; Williams, C. K. Sustainable Polymers From Renewable Resources. Nature 2016, 540 (7633), 354– 362, DOI: 10.1038/nature21001Google ScholarThere is no corresponding record for this reference.
- 14Kharissova, O. V.; Kharisov, B. I.; González, C. M. O.; Méndez, Y. P.; López, I. Greener Synthesis Of Chemical Compounds And Materials. R. Soc. Open Sci. 2019, 6 (11), 191378 DOI: 10.1098/rsos.191378Google ScholarThere is no corresponding record for this reference.
- 15Miturska-Barańska, I.; Rudawská, A.; Sobotová, L.; Badida, M.; Olewnik-Kruszkowska, E.; Müller, M.; Hromasová, M. Analysis Of Acoustic Absorption Coefficients And Characterization Of Epoxy Adhesive Compositions Based On The Reaction Product Of Bisphenol A With Epichlorohydrin Modified With Fillers. Materials 2024, 17 (18), 4452 DOI: 10.3390/ma17184452Google ScholarThere is no corresponding record for this reference.
- 16Wang, X.; Li, Y.; Jiang, Y.; Meng, L.; Nie, Z. In-Depth Free Fatty Acids Annotation Of Edible Oil By Mcpba Epoxidation And Tandem Mass Spectrometry. Food Chem. 2022, 374, 131793 DOI: 10.1016/j.foodchem.2021.131793Google ScholarThere is no corresponding record for this reference.
- 17Samyn, P.; Bosmans, J.; Cosemans, P. Role Of Bio-Based And Fossil-Based Reactive Diluents In Epoxy Coatings With Amine And Phenalkamine Crosslinker. Polymers 2023, 15 (19), 3856 DOI: 10.3390/polym15193856Google ScholarThere is no corresponding record for this reference.
- 18Pradhan, S.; Pandey, P.; Mohanty, S.; Nayak, S. K. Insight On The Chemistry Of Epoxy And Its Curing For Coating Applications: A Detailed Investigation And Future Perspectives. Polym.-Plast. Technol. Eng. 2016, 55 (8), 862– 877, DOI: 10.1080/03602559.2015.1103269Google ScholarThere is no corresponding record for this reference.
- 19Pappa, C.; Féghali, E.; Vanbroekhoven, K.; Triantafyllidis, K. S. Recent Advances In Epoxy Resins And Composites Derived From Lignin And Related Bio-Oils. Curr. Opin. Green Sustainable Chem. 2022, 38, 100687 DOI: 10.1016/j.cogsc.2022.100687Google ScholarThere is no corresponding record for this reference.
- 20Ahmat, Y. M.; Madadi, S.; Charbonneau, L.; Kaliaguine, S. Epoxidation Of Terpenes. Catalysts 2021, 11 (7), 847 DOI: 10.3390/catal11070847Google ScholarThere is no corresponding record for this reference.
- 21Kumar, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Recent Development Of Biobased Epoxy Resins: A Review. Polym.-Plast. Technol. Eng. 2018, 57 (3), 133– 155, DOI: 10.1080/03602559.2016.1253742Google ScholarThere is no corresponding record for this reference.
- 22Ingle, A. A.; Ansari, S. Z.; Shende, D. Z.; Wasewar, K. L.; Pandit, A. B. Progress And Prospective Of Heterogeneous Catalysts For H2O2 Production Via Anthraquinone Process. Environ. Sci. Pollut. Res. 2022, 29 (57), 86468– 86484, DOI: 10.1007/s11356-022-21354-zGoogle ScholarThere is no corresponding record for this reference.
- 23Meadows, S.; Hosur, M.; Celikbag, Y.; Jeelani, S. Comparative Analysis On The Epoxidation Of Soybean Oil Using Formic And Acetic Acids. Polym. Polym. Compos. 2018, 26 (4), 289– 298, DOI: 10.1177/096739111802600403Google ScholarThere is no corresponding record for this reference.
- 24Meng, Y.; Taddeo, F.; Aguilera, A. F.; Cai, X.-S.; Russo, V.; Tolvanen, P.; Leveneur, S. The Lord Of The Chemical Rings: Catalytic Synthesis Of Important Industrial Epoxide Compounds. Catalysts 2021, 11 (7), 765 DOI: 10.3390/catal11070765Google ScholarThere is no corresponding record for this reference.
- 25Mao, S.; Budweg, S.; Spannenberg, A.; Wen, X.; Yang, Y.; Li, Y.; Junge, K.; Beller, M. Iron-Catalyzed Epoxidation Of Linear A-Olefins With Hydrogen Peroxide. ChemCatChem 2022, 14 (4), e202101668 DOI: 10.1002/cctc.202101668Google ScholarThere is no corresponding record for this reference.
- 26Găină, C.; Ursache, O.; Gaina, V.; Serban, A.-M.; Asăndulesa, M. Novel Bio-Based Materials: From Castor Oil To Epoxy Resins For Engineering Applications. Materials 2023, 16 (16), 5649 DOI: 10.3390/ma16165649Google ScholarThere is no corresponding record for this reference.
- 27Bodhak, C.; Patel, D.; Gupta, R. K. Mechanically Robust, Self-Healable, And Reprocessable Geraniol-Based Epoxy Vitrimer By Dynamic Boronic Ester Bonds. ACS Appl. Eng. Mater. 2025, 3 (6), 1599– 1612, DOI: 10.1021/acsaenm.5c00155Google ScholarThere is no corresponding record for this reference.
- 28Zhu, Z.; Zhang, E.; Tu, Y.; Ye, M.; Chen, N. An Eco-Friendly Wood Adhesive Consisting Of Soybean Protein And Cardanol-Based Epoxy For Wood Based Composites. Polymers 2022, 14 (14), 2831 DOI: 10.3390/polym14142831Google ScholarThere is no corresponding record for this reference.
- 29Yuan, J. S.; Pavlovich, M. J.; Ragauskas, A. J.; Han, B. Biotechnology For A Sustainable Future: Biomass And Beyond. Trends Biotechnol. 2022, 40 (12), 1395– 1398, DOI: 10.1016/j.tibtech.2022.09.020Google ScholarThere is no corresponding record for this reference.
- 30Teleky, B.-E.; Vodnar, D. C. Recent Advances In Biotechnological Itaconic Acid Production, And Application For A Sustainable Approach. Polymers 2021, 13 (20), 3574 DOI: 10.3390/polym13203574Google ScholarThere is no corresponding record for this reference.
- 31Zhang, R.; Liu, H.; Ning, Y.; Yu, Y.; Deng, L.; Wang, F. Recent Advances On The Production Of Itaconic Acid Via The Fermentation And Metabolic Engineering. Fermentation 2023, 9 (1), 71 DOI: 10.3390/fermentation9010071Google ScholarThere is no corresponding record for this reference.
- 32Sollka, L.; Lienkamp, K. Progress In The Free And Controlled Radical Homo- And Co-Polymerization Of Itaconic Acid Derivatives: Toward Functional Polymers With Controlled Molar Mass Distribution And Architecture. Macromol. Rapid Commun. 2021, 42 (4), e2000546 DOI: 10.1002/marc.202000546Google ScholarThere is no corresponding record for this reference.
- 33Kumar, S.; Krishnan, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Itaconic Acid Used As A Versatile Building Block For The Synthesis Of Renewable Resource-Based Resins And Polyesters For Future Prospective: A Review. Polym. Int. 2017, 66 (10), 1349– 1363, DOI: 10.1002/pi.5399Google ScholarThere is no corresponding record for this reference.
- 34Yang, S.; He, Y.; Zhang, J. High-Performance Acrylate-Free Co-Cured Coatings Enabled By Tailoring Itaconic Acid-Modified Dual-Phase Photocurable Resins. Prog. Org. Coat. 2024, 189, 108293 DOI: 10.1016/j.porgcoat.2024.108293Google ScholarThere is no corresponding record for this reference.
- 35Mu, S.; Guo, J.; Yu, Y.; An, Q.; Zhang, S.; Wang, D.; Chen, S.; Huang, X.; Li, S. Synthesis And Thermal Properties Of Cross-Linked Poly(Acrylonitrile-Co-Itaconate)/Polyethylene Glycol As Novel Form-Stable Change Material. Energy Convers. Manage. 2016, 110, 176– 183, DOI: 10.1016/j.enconman.2015.12.004Google ScholarThere is no corresponding record for this reference.
- 36Robert, T.; Friebel, S. Itaconic Acid – A Versatile Building Block For Renewable Polyesters With Enhanced Functionality. Green Chem. 2016, 18 (10), 2922– 2934, DOI: 10.1039/C6GC00605AGoogle ScholarThere is no corresponding record for this reference.
- 37Campbell, S. B.; Wu, Q.; Yazbeck, J.; Liu, C.; Okhovatian, S.; Radisic, M. Beyond Polydimethylsiloxane: Alternative Materials For Fabrication Of Organ-On-A-Chip Devices And Microphysiological Systems. ACS Biomater. Sci. Eng. 2021, 7 (7), 2880– 2899, DOI: 10.1021/acsbiomaterials.0c00640Google ScholarThere is no corresponding record for this reference.
- 38Fathi, M.; Alami-Milani, M.; Geranmayeh, M. H.; Barar, J.; Erfan-Niya, H.; Omidi, Y. Dual Thermo-And Ph-Sensitive Injectable Hydrogels Of Chitosan/(Poly(N-Isopropylacrylamide-Co-Itaconic Acid)) For Doxorubicin Delivery In Breast Cancer. Int. J. Biol. Macromol. 2019, 128, 957– 964, DOI: 10.1016/j.ijbiomac.2019.01.122Google ScholarThere is no corresponding record for this reference.
- 39Kim, H. C.; Kwon, Y. R.; Kim, J. S.; Kim, J.-H.; Kim, D. H. Surface-Crosslinking In The Presence Of Nanoclay And Characteristics Of The Itaconic Acid-Based Superabsorbent Polymer Composites. Polym.-Plast. Technol. Mater. 2023, 62 (6), 701– 711, DOI: 10.1080/25740881.2022.2133613Google ScholarThere is no corresponding record for this reference.
- 40Bantchev, G. B.; Doll, K. M. Comparative Amine-Catalyzed Thia-Michael Reactions Of Primary And Secondary Thiols With Maleic And Itaconic Anhydrides And Esters. ChemistrySelect 2022, 7 (48), e202204138 DOI: 10.1002/slct.202204138Google ScholarThere is no corresponding record for this reference.
- 41Niang, F.; Brunou-Bouard, A.; Cruz, G.; Pantoustier, N.; Coumes, F.; Illy, N. Synthesis Of Biobased And Versatile Monomers From Itaconic Acid And Homocysteine Thiolactone And Their Applications In Step-Growth And Radical Polymerization Approaches. Polym. Chem. 2024, 15 (35), 3597– 3607, DOI: 10.1039/D4PY00565AGoogle ScholarThere is no corresponding record for this reference.
- 42Stevens, L. M.; Almada, N. T.; Kim, H. S.; Page, Z. A. Visible-Light-Fueled Polymerizations For 3D Printing. Acc. Chem. Res. 2025, 58 (2), 250– 260, DOI: 10.1021/acs.accounts.4c00680Google ScholarThere is no corresponding record for this reference.
- 43Kim, G.-T.; Go, H.-B.; Yu, J.-H.; Yang, S.-Y.; Kim, K.-M.; Choi, S.-H.; Kwon, J.-S. Cytotoxicity, Colour Stability And Dimensional Accuracy Of 3D Printing Resin With Three Different Photoinitiators. Polymers 2022, 14 (5), 979 DOI: 10.3390/polym14050979Google ScholarThere is no corresponding record for this reference.
- 44Xu, M.; Liu, M.; Wang, F.; Chi, H. Strategies To Improve Water Solubility Of Photoinitiator For Photopolymerization-Based 3D Printing In Biomedical Applications. ACS Appl. Polym. Mater. 2025, 7 (7), 4077– 4092, DOI: 10.1021/acsapm.5c00184Google ScholarThere is no corresponding record for this reference.
- 45Šimunović, L.; Brenko, L.; Marić, A. J.; Meštrović, S.; Haramina, T. Rheology Of Dental Photopolymers For Sla/Dlp/Msla 3D Printing. Polymers 2025, 17 (19), 2706 DOI: 10.3390/polym17192706Google ScholarThere is no corresponding record for this reference.
- 46Vyas, A.; Garg, V.; Ghosh, S. B.; Bandyopadhyay-Ghosh, S. Photopolymerizable Resin-Based 3D Printed Biomedical Composites: Factors Affecting Resin Viscosity. Mater. Today Proc. 2022, 62, 1435– 1439, DOI: 10.1016/j.matpr.2022.01.172Google ScholarThere is no corresponding record for this reference.
- 47Konuray, O.; Morancho, J. M.; Fernández-Francos, X.; García-Alvarez, M.; Ramis, X. Curing Kinetics Of Dually-Processed Acrylate-Epoxy 3D Printing Resins. Thermochim. Acta 2021, 701, 178963 DOI: 10.1016/j.tca.2021.178963Google ScholarThere is no corresponding record for this reference.
- 48Uzcategui, A. C.; Muralidharan, A.; Ferguson, V. L.; Bryant, S. J.; McLeod, R. R. Understanding And Improving Mechanical Properties In 3D Printed Parts Using A Dual-Cure Acrylate-Based Resin For Stereolithography. Adv. Eng. Mater. 2018, 20 (12), 1800876 DOI: 10.1002/adem.201800876Google ScholarThere is no corresponding record for this reference.
- 49Kolibaba, T. J.; Killgore, J. P.; Caplins, B. W.; Higgins, C. I.; Arp, U.; Miller, C.; Poster, D. L.; Zong, Y.; Broce, S.; Wang, T.; Talačka, V.; Andersson, J.; Davenport, A. T.; Panzer, M. A.; Tumbleston, J. R.; Gonzalez, J. M.; Huffstetler, J.; Lund, B.; Billerbeck, K.; Clay, A.; Fratarcangeli, M. R.; Qi, H. J.; Porcincula, D. H.; Bezek, L. B.; Kikuta, K.; Pearlson, M. N.; Walker, D.; Long, C. J.; Hasa, E.; Aguirre-Soto, A.; Celis-Guzman, A.; Backman, D. E.; Sridhar, R. L.; Cavicchi, K. A.; Viereckl, R. J.; Tong, E.; Hansen, C. J.; Shah, D. M.; Kinane, C.; Pena-Francesch, A.; Antonini, C.; Chaudhary, R.; Muraca, G.; Bensouda, Y.; Zhang, Y.; Zhao, X. Results Of An Interlaboratory Study On The Working Curve In Vat Photopolymerization. Addit. Manuf. 2024, 84, 104082 DOI: 10.1016/j.addma.2024.104082Google ScholarThere is no corresponding record for this reference.
- 50Li, Y.; Mao, Q.; Yin, J.; Wang, Y.; Fu, J.; Huang, Y. Theoretical Prediction And Experimental Validation Of The Digital Light Processing (Dlp) Working Curve For Photocurable Materials. Addit. Manuf. 2021, 37, 101716 DOI: 10.1016/j.addma.2020.101716Google ScholarThere is no corresponding record for this reference.
- 51Hofstetter, C. P.; Orman, S.; Baudis, S.; Stampfl, J. Combining Cure Depth And Cure Degree, A New Way To Fully Characterize Novel Photopolymers. Addit. Manuf. 2018, 24, 166– 172, DOI: 10.1016/j.addma.2018.09.025Google ScholarThere is no corresponding record for this reference.
- 52Bösche, C.; Hagenlocher, L.; Bianchi, E.; Bragato, N.; Lotti, N.; Robert, T. Biobased Uv-Curing Additive Manufacturing Materials Derived From Itaconic Acid–Based Polyester Resin And Biobased Reactive Diluents From (Meth)Acrylic Acid. ACS Sustainable Chem. Eng. 2025, 13 (44), 19405– 19415, DOI: 10.1021/acssuschemeng.5c09188Google ScholarThere is no corresponding record for this reference.
- 53Gupta, A.; Salajeghe, R.; Spangenberg, J.; Marla, D. A Computational Model For Stereolithography Apparatus (Sla) 3D Printing. Prog. Addit. Manuf. 2023, 9 (6), 1605– 1619, DOI: 10.1007/s40964-023-00525-5Google ScholarThere is no corresponding record for this reference.
- 54Yi, X.; Kuang, X.; Kong, L.; Dong, X.; Feng, Z.; Wang, D. A Simplified Chemorheological Model Of Viscosity Evolution For Solvent Containing Resol Resin In Scprtm/Scp Process. J. Appl. Polym. Sci. 2017, 134 (36), 45282 DOI: 10.1002/app.45282Google ScholarThere is no corresponding record for this reference.
- 55Kolibaba, T. J.; Caplins, B. W.; Higgins, C. I.; Mansfield, E.; Miller, S. L.; Chandler, C.; Killgore, J. P. Tailoring Properties Of 3D-Printable Polyelectrolyte Photopolymer Complexes With Reactive Diluents. ACS Appl. Polym. Mater. 2024, 6 (12), 6957– 6965, DOI: 10.1021/acsapm.4c00441Google ScholarThere is no corresponding record for this reference.
- 56Hevus, I.; Tiwari, S.; Thorat, S. S.; Gibbon, L.; La Scala, J. J.; Ulven, C. A.; Sibi, M. P.; Webster, D. C. Vanillin-Derived Veratrole Reactive Diluents In Stereolithography. ACS Appl. Polym. Materi. 2024, 6 (13), 7705– 7715, DOI: 10.1021/acsapm.4c01183Google ScholarThere is no corresponding record for this reference.
- 57Jagtap, A. R.; More, A. P. Developments In Reactive Diluents: A Review. Polym. Bull. 2022, 79 (8), 5667– 5708, DOI: 10.1007/s00289-021-03808-5Google ScholarThere is no corresponding record for this reference.
- 58Wei, M. G.; Xu, H.; Chen, L.; Li, Z.; Liu, R. Isosorbide-Based High Performance Uv-Curable Reactive Diluents. Prog. Org. Coat. 2019, 126, 162– 167, DOI: 10.1016/j.porgcoat.2018.10.028Google ScholarThere is no corresponding record for this reference.
- 59Arnaud, S. P.; Malitowski, N. M.; Casamayor, K. M.; Robert, T. Itaconic Acid-Based Reactive Diluents For Renewable And Acrylate-Free Uv-Curing Additive Manufacturing Materials. ACS Sustainable Chem. Eng. 2021, 9 (50), 17142– 17151, DOI: 10.1021/acssuschemeng.1c06713Google ScholarThere is no corresponding record for this reference.
- 60Papadopoulos, L.; Pezzana, L.; Malitowski, N. M.; Sangermano, M.; Bikiaris, D. N.; Robert, T. Uv-Curing Additive Manufacturing Of Bio-Based Thermosets: Effect Of Diluent Concentration On Printing And Material Properties Of Itaconic Acid-Based Materials. ACS Omega 2023, 8 (34), 31009– 31020, DOI: 10.1021/acsomega.3c02808Google ScholarThere is no corresponding record for this reference.
- 61Papadopoulos, L.; Malitowski, N. M.; Bikiaris, D. N.; Robert, T. Bio-Based Additive Manufacturing Materials: An In-Depth Structure-Property Relationship Study Of Uv-Curing Polyesters From Itaconic Acid. Eur. Polym. J. 2023, 186, 111872 DOI: 10.1016/j.eurpolymj.2023.111872Google ScholarThere is no corresponding record for this reference.
- 62Cazin, I.; Ocepek, M.; Kecelj, J.; Stražar, A. S.; Schlögl, S. Synthesis Of Bio-Based Polyester Resins For Vat Photopolymerization 3D Printing. Materials 2024, 17 (8), 1890 DOI: 10.3390/ma17081890Google ScholarThere is no corresponding record for this reference.
- 63Bösche, C.; Hagenlocher, L.; Bianchi, E.; Bragato, N.; Lotti, N.; Robert, T. Biobased Uv-Curing Additive Manufacturing Materials derived From Itaconic Acid–Based Polyester Resin And Biobased Reactive Diluents From (Meth)Acrylic Acid. ACS Sustainable Chem. Eng. 2025, 13 (44), 19405– 19415, DOI: 10.1021/acssuschemeng.5c09188Google ScholarThere is no corresponding record for this reference.
- 64Gao, Y.; Li, Y.; Hu, X.; Wu, W.; Wang, Z.; Wang, R.; Zhang, L. Preparation And Properties Of Novel Thermoplastic Vulcanizate Based On Bio-Based Polyester/Polylactic Acid, And Its Application In 3D Printing. Polymers 2017, 9 (12), 694 DOI: 10.3390/polym9120694Google ScholarThere is no corresponding record for this reference.
- 65Carmenini, R.; de León, A. S.; Benelli, T.; Giorgini, L.; Franchini, M. C.; Molina, S. I.; Maturi, M. One-Pot Depolymerization–Repolymerization Of Pet Waste Into Sustainable Photocurable Liquid Copolyesters For High-Performance Additive Manufacturing. Green Chem. 2025, 27 (40), 12830– 12843, DOI: 10.1039/d5gc02696bGoogle ScholarThere is no corresponding record for this reference.
- 66Maturi, M.; Maturi, S.; de León, A. S.; Migliorini, L.; de la Mata, M.; Benelli, T.; Giorgini, L.; Milani, P.; Franchini, M. C.; Molina, S. I. Enhanced Properties Of 3D-Printed Graphene Oxide Nanocomposites Through Itaconic Acid Polyester Grafting. ACS Appl. Polym. Mater. 2025, 7 (7), 4371– 4382, DOI: 10.1021/acsapm.5c00014Google ScholarThere is no corresponding record for this reference.
- 67Torres, G. B.; Xia, T.; Yu, D.; Thijssen, Q.; Van Vlierberghe, S.; Li, B.; Heise, A. Raft-Mediated 3D Printing Of Polylactones/Itaconate Elastomers With Polypeptide Surface Functionalization. ACS Polym. Au 2025, 5, 956– 966, DOI: 10.1021/acspolymersau.5c00117Google ScholarThere is no corresponding record for this reference.
- 68Papadopoulos, L.; Pezzana, L.; Malitowski, N. M.; Kladovasilakis, N.; Tzetzis, D.; Sangermano, M.; Bikiaris, D. N.; Robert, T. Itaconic Acid-Based 3D Printed Nanocomposites: An In-Depth Study On The Effect Of Nanoinclusions On The Physicochemical Properties And The Printability Of Formulations Based On Polyester Itaconates. Giant 2024, 18, 100275 DOI: 10.1016/j.giant.2024.100275Google ScholarThere is no corresponding record for this reference.
- 69Lok, T.-J.; Wong, J.-W.; Li, X.; Fu, Y.; Xue, Y.; Jamaludin, F. H.; Fong, M.; Edward, E. B.; Ma, C.; Chandren, S.; Goh, P.-S.; Wong, T.-W. Biobased Itaconate Polyester Thermoset With Tunable Mechanical Properties. Macromolecules 2024, 57 (5), 2317– 2328, DOI: 10.1021/acs.macromol.3c01743Google ScholarThere is no corresponding record for this reference.
- 70Spanu, C.; Locatelli, E.; Sambri, L.; Franchini, M. C.; Maturi, M. Photocurable Itaconic Acid-Functionalized Star Polycaprolactone In Biobased Formulations For Vat Photopolymerization. ACS Appl. Polym. Mater. 2024, 6 (4), 2417– 2424, DOI: 10.1021/acsapm.3c03159Google ScholarThere is no corresponding record for this reference.
- 71Bokhari, S. M. Q.; Sevening, J. N.; Catchmark, J. M.; Chmely, S. C. Composition–Property Engineering Of Bio-Derived Uv-Curable Acrylate Oligoester Resins For Tunable Mechanics In 3D Printing. RSC Appl. Polym. 2025, 3 (4), 948– 959, DOI: 10.1039/d5lp00085hGoogle ScholarThere is no corresponding record for this reference.
- 72Miętus, M.; Gołofit, T.; Gadomska-Gajadhur, A. Towards Greener Polymers: Poly(Octamethylene Itaconate-Co-Succinate) Synthesis Parameters. Polymers 2025, 17 (16), 2220 DOI: 10.3390/polym17162220Google ScholarThere is no corresponding record for this reference.
- 73Maturi, M.; Pulignani, C.; Locatelli, E.; Vetri Buratti, V.; Tortorella, S.; Sambri, L.; Franchini, M. C. Phosphorescent Bio-Based Resin For Digital Light Processing (Dlp) 3D-Printing. Green Chem. 2020, 22 (18), 6212– 6224, DOI: 10.1039/d0gc01983fGoogle ScholarThere is no corresponding record for this reference.
- 74Jao, C.-Y.; Mao, H.-I.; Shiu, J.-W.; Chen, Y.-C.; Chu, R.-J.; Chen, C.-W. Influence Of Diol Structure On The Mechanical And Thermal Properties Of Bio-Based Uv-Curable Unsaturated Polyesters For 3D Printing. ACS Appl. Polym. Mater. 2025, 7 (17), 11284– 11299, DOI: 10.1021/acsapm.5c01805Google ScholarThere is no corresponding record for this reference.
- 75Carmenini, R.; Spanu, C.; Locatelli, E.; Sambri, L.; Franchini, M. C.; Maturi, M. Isocyanate-Free Urethanediol Itaconates As Biobased Liquid Monomers In Photopolymerization-Based 3D Printing. Prog. Addit. Manuf. 2024, 9 (6), 2499– 2510, DOI: 10.1007/s40964-024-00598-wGoogle ScholarThere is no corresponding record for this reference.
- 76Vetri Buratti, V.; Sanz de Leon, A.; Maturi, M.; Sambri, L.; Molina, S. I.; Comes Franchini, M. Itaconic-Acid-Based Sustainable Poly(Ester Amide) Resin For Stereolithography. Macromolecules 2022, 55 (8), 3087– 3095, DOI: 10.1021/acs.macromol.1c02525Google ScholarThere is no corresponding record for this reference.
- 77Papadopoulos, L.; Kluge, M.; Bikiaris, D. N.; Robert, T. Straightforward Synthetic Protocol To Bio-Based Unsaturated Poly(Ester Amide)S From Itaconic Acid With Thixotropic Behavior. Polymers 2020, 12 (4), 980 DOI: 10.3390/polym12040980Google ScholarThere is no corresponding record for this reference.
- 78Ouhichi, R.; Arnaud, S. P.; Bougarech, A.; Abid, S.; Abid, M.; Robert, T. First Example Of Unsaturated Poly(Ester Amide)S Derived From Itaconic Acid And Their Application As Bio-Based Uv-Curing Polymers. Appl. Sci. 2020, 10 (6), 2163, DOI: 10.3390/app10062163Google ScholarThere is no corresponding record for this reference.
- 79Kumar, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Synthesis And Characterization Of Itaconic-Based Epoxy Resins. Polym. Adv. Technol. 2018, 29 (1), 160– 170, DOI: 10.1002/pat.4098Google ScholarThere is no corresponding record for this reference.
- 80Luo, Y.; Le Fer, G.; Dean, D.; Becker, M. L. 3D Printing Of Poly(Propylene Fumarate) Oligomers: Evaluation Of Resin Viscosity, Printing Characteristics And Mechanical Properties. Biomacromolecules 2019, 20 (4), 1699– 1708, DOI: 10.1021/acs.biomac.9b00076Google ScholarThere is no corresponding record for this reference.
- 81Lu, D.; Pen, G.; Zhang, H.; Bao, J. Engineering Non-Covalent Interactions: Hierarchical Hydrogen-Bonding Strategy For Advanced 3D Printer Resins. Polymer 2025, 326, 128323 DOI: 10.1016/j.polymer.2025.128323Google ScholarThere is no corresponding record for this reference.
- 82Montoya, J.; Medina, J.; Molina, A.; Gutierrez, J. A.; Rodriguez, B. E.; Marín, R. Impact Of Viscoelastic And Structural Properties From Starch-Mango And Starch-Arabinoxylans Hydrocolloids In 3D Food Printing. Addit. Manuf. 2021, 39, 101891 DOI: 10.1016/j.addma.2021.101891Google ScholarThere is no corresponding record for this reference.
- 83Li, Y.; Wang, M.; Wu, T.; Yang, X.; Qu, Z.; Yao, X.; He, Y.; Guo, Y.; Pu, Q.; Wang, X. Green Synthesis Of Vat Photopolymerization 3D Printing Polyimide. Small 2025, 21 (27), e2502406 DOI: 10.1002/smll.202502406Google ScholarThere is no corresponding record for this reference.
- 84Panić, V. V.; Šešlija, S. I.; Popović, I. G.; Spasojević, V.; Popović, A.; Nikolić, V.; Spasojević, P. Simple One-Pot Synthesis Of Fully Biobased Unsaturated Polyester Resins Based On Itaconic Acid. Biomacromolecules 2017, 18 (12), 3881– 3891, DOI: 10.1021/acs.biomac.7b00840Google ScholarThere is no corresponding record for this reference.
- 85Deng, K.; Zhang, C.; Fu, K. Additive Manufacturing Of Continuously Reinforced Thermally Curable Thermoset Composites With Rapid Interlayer Curing. Composites, Part B 2023, 257, 110671, DOI: 10.1016/j.compositesb.2023.110671Google ScholarThere is no corresponding record for this reference.
- 86Lee, J.; Ni, X.; Daso, F.; Xiao, X.; King, D. C.; Gómez, J. S.; Varela, T. B.; Kessler, S. S.; Wardle, B. L. Advanced Carbon Fiber Composite Out-Of-Autoclave Laminate Manufacture Via Nanostructured Out-Of-Oven Conductive Curing. Compos. Sci. Technol. 2018, 166, 150– 159, DOI: 10.1016/j.compscitech.2018.02.031Google ScholarThere is no corresponding record for this reference.
- 87Li, Y.; Cheng, L.; Zhou, J. Curing Multidirectional Carbon Fiber Reinforced Polymer Composites With Indirect Microwave Heating. Int. J. Adv. Manuf. Technol. 2018, 97 (1–4), 1137– 1147, DOI: 10.1007/s00170-018-1974-1Google ScholarThere is no corresponding record for this reference.
- 88Zanjanijam, A. R.; Wang, X.; Ramezani, M.; Holberg, S.; Johnson, P. A. Phenolic Resin/Coal Char Composites: Curing Kinetics And Thermal/Mechanical Performance. Polymer 2023, 281, 126103 DOI: 10.1016/j.polymer.2023.126103Google ScholarThere is no corresponding record for this reference.
- 89Chen, J.; Gao, X.; Zhao, K.-Q.; Wu, W. Research On Curing Kinetics Of Multiaxial Warp-Knitted Composites. Polym. Compos. 2023, 44 (9), 5934 DOI: 10.1002/pc.27538Google ScholarThere is no corresponding record for this reference.
- 90Xiong, X.; Guan, H.; Li, B.; Yang, S.; Li, W.; Ren, R.; Wang, J.; Chen, P. Cure Kinetics And Thermal Decomposition Behavior Of Novel Phenylacetylene-Capped Polyimide Resins. Polymers 2024, 16 (8), 1149, DOI: 10.3390/polym16081149Google ScholarThere is no corresponding record for this reference.
- 91Guo, Z.; Yi-liang, W.; Chiumsenti, M.; Wu, Y.; Zhen, S.; Liu, H.; Rossi, R.; Fu, X. Research On Curing Reaction Kinetics And Curing Process Of Hydroxy-Terminated Polybutadiene (Htpb) Propellants. Sci. Rep. 2025, 15 (1), 21006 DOI: 10.1038/s41598-025-07125-zGoogle ScholarThere is no corresponding record for this reference.
- 92Matykiewicz, D.; Dudziec, B. Curing And Degradation Kinetics Of Phosphorus-Modified Eugenol-Based Epoxy Resin. ACS Omega 2025, 10 (5), 4353– 4366, DOI: 10.1021/acsomega.4c06532Google ScholarThere is no corresponding record for this reference.
- 93Li, X.; Yao, H.; Lu, X.; Chen, C.; Cao, Y.; Xin, Z. Effect Of Pyrogallol On The Ring-Opening Polymerization And Curing Kinetics Of A Fully Bio-Based Benzoxazine. Thermochim. Acta 2020, 694, 178787 DOI: 10.1016/j.tca.2020.178787Google ScholarThere is no corresponding record for this reference.
- 94Marotta, A.; Faggio, N.; Brondi, C. Curing Kinetics Of Bioderived Furan-Based Epoxy Resins: Study On The Effect Of The Epoxy Monomer/Hardener Ratio. Polymers 2022, 14 (23), 5322 DOI: 10.3390/polym14235322Google ScholarThere is no corresponding record for this reference.
- 95Wang, Y.; Zhang, F.; Li, F.; Du, S.; Dai, S.; Jiang, T.; Sun, K.; Liu, X.; Ma, S. Covalent Adaptable Networks Derived From Itaconic Acid-Based Epoxy And Sulfur: Facile Synthesis And Applications In Adhesives And Carbon Fiber Composites. Compos. Commun. 2025, 59, 102575 DOI: 10.1016/j.coco.2025.102575Google ScholarThere is no corresponding record for this reference.
- 96Xiao, K.; Fang, Y.; Wang, Z.; Ni, N.; Liu, Z.; Kim, S.; An, Z.; Lyu, Z.; Xu, Y.; Yang, X. Bio-Sourced, High-Performance Carbon Fiber Reinforced Itaconic Acid-Based Epoxy Composites With High Hygrothermal Stability And Durability. Polymers 2024, 16 (12), 1649 DOI: 10.3390/polym16121649Google ScholarThere is no corresponding record for this reference.
- 97Shang, L.; Zhang, J.; Liu, L.; Xiao, L.; Li, M.; Ao, Y. Improving The Interfacial Property Of Carbon Fibre/Epoxy Resin Composites By Grafting Amine-Capped Cross-Linked Poly-Itaconic Acid. Surf. Interface Anal. 2019, 51 (2), 199– 209, DOI: 10.1002/sia.6565Google ScholarThere is no corresponding record for this reference.
- 98Kocaman, S.; Akyay, İ. Itaconic Acid Based Epoxy Resin And Application Of Olive Pomace On The Production Of Composite Materials. Ind. Crops Prod. 2024, 222, 120022 DOI: 10.1016/j.indcrop.2024.120022Google ScholarThere is no corresponding record for this reference.
- 99Ghosh, T.; Elo, T.; Parihar, V. S.; Maiti, P.; Layek, R. K. Poly (Itaconic Acid) Functionalized Lignin/Polyvinyl Acetate Composite Resin With Improved Sustainability And Wood Adhesion Strength. Ind. Crops Prod. 2022, 187, 115299 DOI: 10.1016/j.indcrop.2022.115299Google ScholarThere is no corresponding record for this reference.
- 100Dai, Z.; Yang, Z.; Chen, Z.; Zhao, Z.; Lou, Y.; Zhang, Y.; Liu, T. X.; Fu, F.; Fu, Y.; Liu, X. Fully Biobased Composites Of An Itaconic Acid Derived Unsaturated Polyester Reinforced With Cotton Fabrics. ACS Sustainable Chem. Eng. 2018, 6 (11), 15056– 15063, DOI: 10.1021/acssuschemeng.8b03539Google ScholarThere is no corresponding record for this reference.
- 101Li, P.; Ma, S.; Dai, J.; Liu, X.; Jiang, Y.; Wang, S.; Wei, J.; Chen, J.; Zhu, J. Itaconic Acid As A Green Alternative To Acrylic Acid For Producing A Soybean Oil-Based Thermoset: Synthesis And Properties. ACS Sustainable Chem. Eng. 2016, 5 (1), 1228– 1236, DOI: 10.1021/acssuschemeng.6b02654Google ScholarThere is no corresponding record for this reference.
- 102Luo, J.; Ma, B.; Peng, J.; Wu, Z.; Luo, Z.; Wang, X. Modified Chestnut-Like Structure Silicon Carbon Composite As Anode Material For Lithium-Ion Batteries. ACS Sustainable Chem. Eng. 2019, 7 (12), 10415– 10424, DOI: 10.1021/acssuschemeng.9b00616Google ScholarThere is no corresponding record for this reference.
- 103Kausar, A. Polymer Coating Technology For High Performance Applications: Fundamentals And Advances. J. Macromol. Sci., Part A 2018, 55 (5), 440– 448, DOI: 10.1080/10601325.2018.1453266Google ScholarThere is no corresponding record for this reference.
- 104Pan, Y.; Liu, Y.; Yang, S.; Zhang, C.; Ullah, Z. Recent Research Progress On The Stimuli-Responsive Smart Membrane: A Review. Nanotechnol. Rev. 2023, 12 (1), 20220538 DOI: 10.1515/ntrev-2022-0538Google ScholarThere is no corresponding record for this reference.
- 105K A, T.-Z.; A D, Z.; Yu, B. E.; I V, B.; E A, M. Foam Polymers In Multifunctional Insulating Coatings. Polymers 2021, 13 (21), 3698 DOI: 10.3390/polym13213698Google ScholarThere is no corresponding record for this reference.
- 106Chhipa, S. M.; Sharma, S.; Bagha, A. K. Recent Development In Polymer Coating To Prevent Corrosion In Metals: A Review Mater. Today Proc. 2024 DOI: 10.1016/j.matpr.2024.09.001 .Google ScholarThere is no corresponding record for this reference.
- 107Paraskar, P. M.; Prabhudesai, M.; Kulkarni, R. D. Synthesis And Characterizations Of Air-Cured Polyurethane Coatings From Vegetable Oils And Itaconic Acid. React. Funct. Polym. 2020, 156, 104734 DOI: 10.1016/j.reactfunctpolym.2020.104734Google ScholarThere is no corresponding record for this reference.
- 108Dong, X.; Ren, J.; Duan, Y.; Wu, D.; Lin, L.; Jichao, S.; Jia, R.; Xu, X.; He, X.; Dong, X.; Ren, J.; Duan, Y.; Wu, D.; Lin, L.; Jichao, S.; Jia, R.; Xu, X.; He, X. Preparation And Properties Of Green UV-Curable Itaconic Acid Cross-Linked Modified Waterborne Polyurethane Coating. J. Appl. Polym. Sci. 2021, 139 (17), 52042 DOI: 10.1002/app.52042Google ScholarThere is no corresponding record for this reference.
- 109Huang, J.; Zhang, J.; Zhu, G.; Yu, X.; Hu, Y.; Shang, Q.; Chen, J.; Hu, L.; Zhou, Y.; Liu, C. Self-Healing, High-Performance, And High-Biobased-Content Uv-Curable Coatings Derived From Rubber Seed Oil And Itaconic Acid. Prog. Org. Coat. 2021, 159, 106391 DOI: 10.1016/j.porgcoat.2021.106391Google ScholarThere is no corresponding record for this reference.
- 110Mehta, L. B.; Wadgaonkar, K. K.; Jagtap, R. N. Synthesis And Characterization Of High Bio-Based Content Unsaturated Polyester Resin For Wood Coating From Itaconic Acid: Effect Of Various Reactive Diluents As An Alternative To Styrene. J. Dispersion Sci. Technol. 2019, 40 (5), 756– 765, DOI: 10.1080/01932691.2018.1480964Google ScholarThere is no corresponding record for this reference.
- 111Yang, S.; He, Y.; Zhang, J. High-Performance Acrylate-Free Co-Cured coatings Enabled By Tailoring Itaconic Acid-Modified Dual-Phase Photocurable Resins. Prog. Org. Coat. 2024, 189, 108293 DOI: 10.1016/j.porgcoat.2024.108293Google ScholarThere is no corresponding record for this reference.
- 112Zhang, Y.; Zhuo, G.; Huang, Y.; Qin, M. J.; Liu, M.; Li, L.; Guo, C. Synthesis Of Bio-Based Epoxy Resins Derived From Itaconic Acid And Application In Rubber Wood Surface Coating. Ind. Crops Prod. 2024, 222, 119529 DOI: 10.1016/j.indcrop.2024.119529Google ScholarThere is no corresponding record for this reference.
- 113Brännström, S.; Malmström, E.; Johansson, M. Biobased Uv-Curable Coatings Based On Itaconic Acid. J. Coat. Technol. Res. 2017, 14 (4), 851– 861, DOI: 10.1007/s11998-017-9949-yGoogle ScholarThere is no corresponding record for this reference.
- 114Patil, D. M.; Phalak, G. A.; Mhaske, S. T. Design And Synthesis Of Bio-Based Uv Curable Pu Acrylate Resin From Itaconic Acid For Coating Applications. Des. Monomers Polym. 2017, 20 (1), 269– 282, DOI: 10.1080/15685551.2016.1231045Google ScholarThere is no corresponding record for this reference.
- 115Dai, J.; Liu, X.; Ma, S.; Wang, J.; Shen, X.; You, S.; Zhu, J. Soybean Oil-Based Uv-Curable Coatings Strengthened By Crosslink Agent Derived From Itaconic Acid Together With 2-Hydroxyethyl Methacrylate Phosphate. Prog. Org. Coat. 2016, 97, 210– 215, DOI: 10.1016/j.porgcoat.2016.04.014Google ScholarThere is no corresponding record for this reference.
- 116Dixit, A.; Sabnis, A.; Balgude, D.; Kale, S.; Gada, A. H.; Kudu, B.; Mehta, K.; Kasar, S.; Handa, D.; Mehta, R.; Kshirsagar, S.; Singh, A.; Dalvi, R.; Mishra, S. Synthesis And Characterization Of Citric Acid And Itaconic Acid-Based Two-Pack Polyurethane Antimicrobial Coatings. Polym. Bull. 2023, 80 (2), 2187– 2216, DOI: 10.1007/s00289-022-04638-9Google ScholarThere is no corresponding record for this reference.
- 117Chen, B.; Wang, X.; Xu, H.; Zhang, Y.; Wang, C. Synthesis Of Itaconic Acid-Based Non-Isocyanate Polyurethane Coating With Excellent Heat Insulation And Corrosion Resistance. Arabian J. Chem. 2025, 18, 4982025 DOI: 10.25259/AJC_498_2025Google ScholarThere is no corresponding record for this reference.
- 118Schneider-Chaabane, A.; Bleicher, V.; Rau, S.; Al-Ahmad, A.; Lienkamp, K. Stimulus-Responsive Polyzwitterionic Surfaces Made From Itaconic Acid: Self-Triggered Antimicrobial Activity, Protein Repellency, And Cell Compatibility. ACS Appl. Mater. Interfaces 2020, 12 (19), 21242– 21253, DOI: 10.1021/acsami.9b17781Google ScholarThere is no corresponding record for this reference.
- 119Jamaludin, F. H.; Mohd Yusoff, N. I. S.; Chida, H.; Yang, X.; Wong, T.-W.; Zhou, H.; Li, T.; Wang, L.; Goh, P. S.; Ismail, A. F. Photo-Triggered Sustainable Adhesive Based On Itaconic Acid. ACS Sustainable Chem. Eng. 2022, 10 (19), 6389– 6401, DOI: 10.1021/acssuschemeng.2c01119Google ScholarThere is no corresponding record for this reference.
- 120Stange, P.; Verevkin, S. P.; Ludwig, R. Combined Spectroscopic, Thermodynamic, And Theoretical Approach For Detecting And Quantifying Hydrogen Bonding And Dispersion Interaction In Ionic Liquids. Acc. Chem. Res. 2023, 56 (23), 3441– 3450, DOI: 10.1021/acs.accounts.3c00530Google ScholarThere is no corresponding record for this reference.
- 121Li, W.; Wang, Y.; Ni, Z.; Li, S. Cluster-In-Molecule Local Correlation Method For Dispersion Interactions In Large Systems And Periodic Systems. Acc. Chem. Res. 2023, 56 (23), 3462– 3474, DOI: 10.1021/acs.accounts.3c00538Google ScholarThere is no corresponding record for this reference.
- 122Becker, N.; Noyes, K.; Cooper, M. M. Characterizing Students’ Mechanistic Reasoning About London Dispersion Forces. J. Chem. Educ. 2016, 93 (10), 1713– 1724, DOI: 10.1021/acs.jchemed.6b00298Google ScholarThere is no corresponding record for this reference.
- 123Law, K.-Y. Contact Angle Hysteresis On Smooth/Flat And Rough Surfaces. Interpretation, Mechanism, And Origin. Acc. Mater. Res. 2022, 3 (1), 1– 7, DOI: 10.1021/accountsmr.1c00051Google ScholarThere is no corresponding record for this reference.
- 124Bovone, G.; Dudaryeva, O. Y.; Marco-Dufort, B.; Tibbitt, M. W. Engineering Hydrogel Adhesion For Biomedical Applications Via Chemical Design Of The Junction. ACS Biomater. Sci. Eng. 2021, 7 (9), 4048– 4076, DOI: 10.1021/acsbiomaterials.0c01677Google ScholarThere is no corresponding record for this reference.
- 125Liu, M.; Li, J.; Hou, Y.; Guo, Z. Inorganic Adhesives For Robust Superwetting Surfaces. ACS Nano 2017, 11 (1), 1113– 1119, DOI: 10.1021/acsnano.6b08348Google ScholarThere is no corresponding record for this reference.
- 126Mehmood, T.; Kaynak, A.; Dai, X. J.; Kouzani, A. Z.; Magniez, K.; Rubin, D.; Hurren, C.; du Plessis, J. Study Of Oxygen Plasma Pre-Treatment Of Polyester Fabric For Improved Polypyrrole Adhesion. Mater. Chem. Phys. 2014, 143 (2), 668– 675, DOI: 10.1016/j.matchemphys.2013.09.052Google ScholarThere is no corresponding record for this reference.
- 127Lallemang, M.; Yu, L.; Cai, W.; Rischka, K.; Hartwig, A.; Haag, R.; Hugel, T.; Balzer, B. N. Multivalent Non-Covalent Interactions Lead To Strongest Polymer Adhesion. Nanoscale 2022, 14 (10), 3768– 3776, DOI: 10.1039/D1NR08338DGoogle ScholarThere is no corresponding record for this reference.
- 128Zhan, J.; Lei, Z.; Zhang, Y. Non-Covalent Interactions Of Graphene Surface: Mechanisms And Applications. Chem 2022, 8 (4), 947– 979, DOI: 10.1016/j.chempr.2021.12.015Google ScholarThere is no corresponding record for this reference.
- 129Besford, Q. A.; Van den Heuvel, W.; Christofferson, A. J. Dipolar Dispersion Forces In Water–Methanol Mixtures: Enhancement Of Water Interactions Upon Dilution Drives Self-Association. J. Phys. Chem. B 2022, 126 (33), 6231– 6239, DOI: 10.1021/acs.jpcb.2c04638Google ScholarThere is no corresponding record for this reference.
- 130Truhlar, D. G. Dispersion Forces: Neither Fluctuating Nor Dispersing. J. Chem. Educ. 2019, 96 (8), 1671– 1675, DOI: 10.1021/acs.jchemed.8b01044Google ScholarThere is no corresponding record for this reference.
- 131Echeverrigaray, F. G.; De Mello, S. S.; Leidens, L. M.; da Costa, M. E. H. M.; Alvarez, F.; Burgo, T. A. L.; Michels, A. F.; Figueroa, C. A. Towards Superlubricity In Nanostructured Surfaces: The Role Of Van Der Waals Forces. Phys. Chem. Chem. Phys. 2018, 20 (34), 21949– 21959, DOI: 10.1039/c8cp02508hGoogle ScholarThere is no corresponding record for this reference.
- 132Liang, H.-J.; Qian, W.-Y.; Liu, H.-H.; Wang, X.; Gu, Z.-Y.; Dong, F.; Deng, Y.; Tang, Y.; Zhang, J.; Zhao, J.; Wu, X.-L. Sulfite-Based Electrolyte Chemistry With Ion–Dipole Interactions And Robust Interphase Achieves Wide-Temperature Sodium-Ion Batteries. J. Am. Chem. Soc. 2025, 147 (21), 17860– 17870, DOI: 10.1021/jacs.5c01864Google ScholarThere is no corresponding record for this reference.
- 133Herschlag, D.; Pinney, M. M. Hydrogen Bonds: Simple After All?. Biochemistry 2018, 57 (24), 3338– 3352, DOI: 10.1021/acs.biochem.8b00217Google ScholarThere is no corresponding record for this reference.
- 134van der Lubbe, S. C. C.; Guerra, C. F. The Nature Of Hydrogen Bonds: A Delineation Of The Role Of Different Energy Components On Hydrogen Bond Strengths And Lengths. Chem. - Asian J. 2019, 14 (16), 2760– 2769, DOI: 10.1002/asia.201900717Google ScholarThere is no corresponding record for this reference.
- 135Schneider, H.-J. Distinction And Quantification Of Noncovalent Dispersive And Hydrophobic Effects. Molecules 2024, 29 (7), 1591 DOI: 10.3390/molecules29071591Google ScholarThere is no corresponding record for this reference.
- 136Mousavi, M.; Fini, E. H. Non-Covalent Π-Stacking Interactions Between Asphaltene And Porphyrin In Bitumen. J. Chem. Inf. Model. 2020, 60 (10), 4856– 4866, DOI: 10.1021/acs.jcim.0c00507Google ScholarThere is no corresponding record for this reference.
- 137Beber, V. C.; Wolter, N.; Schneider, B.; Koschek, K. Effect Of Aluminium Substrate Thickness On The Lap-Shear Strength Of Adhesively Bonded And Hybrid Riveted-Bonded Joints. Proc. Inst. Mech. Eng., Part E 2023, 237 (3), 607– 614, DOI: 10.1177/0954408920913434Google ScholarThere is no corresponding record for this reference.
- 138Li, J.; Hu, C.; Yang, B.; Zhao, N.; Zeng, Y. Recyclable, Self-Healing Itaconic Acid-Based Polyurethane Networks With Dynamic Boronic Ester Bonds For Recoverable Adhesion Application. Polymer 2022, 256, 125227 DOI: 10.1016/j.polymer.2022.125227Google ScholarThere is no corresponding record for this reference.
- 139Kim, H.-C.; Kwon, Y.-R.; Kim, J. S.; So, J.-H.; Kim, D. H. Dual-Cure Adhesives Using A Newly Synthesized Itaconic Acid-Based Epoxy Acrylate Oligomer. Polymers 2023, 15 (15), 3304 DOI: 10.3390/polym15153304Google ScholarThere is no corresponding record for this reference.
- 140Mapari, S.; Mestry, S.; Mhaske, S. T. Developments In Pressure-Sensitive Adhesives: A Review. Polym. Bull. 2021, 78 (7), 4075– 4108, DOI: 10.1007/s00289-020-03305-1Google ScholarThere is no corresponding record for this reference.
- 141Ju, Y. H.; Lee, H.; Han, C. J.; Lee, C.-R.; Kim, Y.; Kim, J.-W. Pressure-Sensitive Adhesive With Controllable Adhesion For Fabrication Of Ultrathin Soft Devices. ACS Appl. Mater. Interfaces 2020, 12 (36), 40794– 40801, DOI: 10.1021/acsami.0c11986Google ScholarThere is no corresponding record for this reference.
- 142Antosik, A. K.; Mozelewska, K.; Piątek-Hnat, M.; Czech, Z.; Bartkowiak, M. Silicone Pressure-Sensitive Adhesives With Increased Thermal Resistance. J. Therm. Anal. Calorim. 2022, 147 (14), 7719– 7727, DOI: 10.1007/s10973-021-11048-yGoogle ScholarThere is no corresponding record for this reference.
- 143Fang, C.; Yan, Q.; Liu, Z.; Lu, Y.; Lin, Z. The Influence Of Monobutyl Itaconate And B-Carboxyethyl Acrylate On Acrylic Latex Pressure Sensitive Adhesives. Int. J. Adhes. Adhes. 2018, 84, 387– 393, DOI: 10.1016/j.ijadhadh.2018.05.007Google ScholarThere is no corresponding record for this reference.
- 144Casas-Soto, C. R.; Conejo-Dávila, A. S.; Osuna, V.; Chávez-Flores, D.; Espinoza-Hicks, J. C.; Flores-Gallardo, S. G.; Vega-Ríos, A. Dibutyl Itaconate And Lauryl Methacrylate Copolymers By Emulsion Polymerization For Development Of Sustainable Pressure-Sensitive Adhesives. Polymers 2022, 14 (3), 632 DOI: 10.3390/polym14030632Google ScholarThere is no corresponding record for this reference.
- 145Xue, W.; Zhang, X.; Sun, C.; Wei, C.; Zhang, L.; Yang, H.; Wang, R. Biobased Itaconate Acrylic Pressure-Sensitive Adhesives Via Emulsion Polymerization: Sustainable Design For High Peel Resistance. ACS Omega 2025, 10 (37), 42494– 42506, DOI: 10.1021/acsomega.5c03586Google ScholarThere is no corresponding record for this reference.
- 146Zeng, X.; Liu, C.; Wang, X.; Cao, Y.; He, P.; Li, H.; Wang, L. Versatile Underwater Pressure Sensitive Adhesive: Uv Curing Synthesis And Substrate-Independent Adhesion. ACS Appl. Mater. Interfaces 2024, 16 (31), 41461– 41474, DOI: 10.1021/acsami.4c06163Google ScholarThere is no corresponding record for this reference.
- 147Wang, X.; Cao, Y.; Zhang, J.; Yu, H. Biodegradable Polyurethane Pressure-Sensitive Adhesives Using Biobased Polyols With Distinct Intrinsic Properties. ACS Appl. Polym. Mater. 2024, 6 (9), 5121– 5128, DOI: 10.1021/acsapm.4c00157Google ScholarThere is no corresponding record for this reference.
- 148Jin, J.; Cheng, L.; Chen, C.; Li, Z.; Hong, Y.; Li, C.; Ban, X.; Gu, Z. Synthesis, Characterization, And Application Of Starch-Based Adhesives Modified With Itaconic Acid And N-Hydroxyethyl Acrylamide. Ind. Crops Prod. 2023, 196, 116524, DOI: 10.1016/j.indcrop.2023.116524Google ScholarThere is no corresponding record for this reference.
- 149Sharif, R.; Mohsin, M.; Sardar, S.; Ramzan, N.; Raza, Z. A. Synthesis Of Nontoxic And Bio Based Oil And Water Repellent Polymers For Cotton Fabrics Using Stearic Acid, Succinic Acid, And Itaconic Acid. J. Nat. Fibers 2022, 19 (16), 12473– 12485, DOI: 10.1080/15440478.2022.2072995Google ScholarThere is no corresponding record for this reference.
- 150Boondaeng, A.; Suwanruji, P.; Vaithanomsat, P.; Apiwatanapiwat, W.; Trakunjae, C.; Janchai, P.; Apipatpapha, T.; Chanka, N.; Chollakup, R. Bio-Synthesis Of Itaconic Acid As An Anti-Crease Finish For Cellulosic Fiber Fabric. RSC Adv. 2021, 11 (42), 25943– 25950, DOI: 10.1039/D1RA05037KGoogle ScholarThere is no corresponding record for this reference.
- 151Peng, H.; Yang, C. Q.; Wang, X.; Wang, S. The Combination Of Itaconic Acid And Sodium Hypophosphite As A New Cross-Linking System For Cotton. Ind. Eng. Chem. Res. 2012, 51 (35), 11301– 11311, DOI: 10.1021/ie3005644Google ScholarThere is no corresponding record for this reference.
- 152Sakthivel, M.; Franklin, D. S.; Sudarsan, S.; Chitra, G.; Sridharan, T. B.; Guhanathan, S. Investigation On Ph/Salt-Responsive Multifunctional Itaconic Acid Based Polymeric Biocompatible, Antimicrobial And Biodegradable Hydrogels. React. Funct. Polym. 2018, 122, 9– 21, DOI: 10.1016/j.reactfunctpolym.2017.10.021Google ScholarThere is no corresponding record for this reference.
- 153Ma, J.; Li, T.; Luo, M.; Lei, B. Single-Component Self-Healing Antibacterial Anti-Inflammatory Intracellular-Antioxidative Poly(Itaconic Acid-Pluronic) Hydrogel For Rapid Repair Of Mrsa-Impaired Wound. ACS Appl. Mater. Interfaces 2023, 15 (28), 33413– 33424, DOI: 10.1021/acsami.3c05383Google ScholarThere is no corresponding record for this reference.
- 154Zhou, S.; He, J.; Liu, Q.; Chen, T.; Guan, X.; Gao, H.; Jiang, J.; Wang, J.; Peng, X.; Wu, J. Injectable Hydrogel Of Chitosan-Octyl Itaconate Conjugate Modulates Inflammatory Response. ACS Biomater. Sci. Eng. 2024, 10 (8), 4823– 4838, DOI: 10.1021/acsbiomaterials.4c00882Google ScholarThere is no corresponding record for this reference.
- 155Naderi, P.; Kabiri, K.; Jahanmardi, R.; Zohuriaan-Mehr, M. J. Preparation Of Itaconic Acid Bio-Based Cross-Linkers For Hydrogels. J. Macromol. Sci., Part A 2021, 58 (3), 165– 174, DOI: 10.1080/10601325.2020.1836492Google ScholarThere is no corresponding record for this reference.
- 156Das, A.; Saha, M.; Gupta, M. K.; Rangan, L.; Uppaluri, R.; Das, C. Comparative Efficacy Of Citric Acid/Tartaric Acid/Malic Acid Additive-Based Polyvinyl Alcohol-Starch Composite Films. J. Mater. Sci.: Mater. Eng. 2024, 19 (1), 9 DOI: 10.1186/s40712-024-00151-1Google ScholarThere is no corresponding record for this reference.
- 157Li, J.; Lei, H.; Xi, X.; Li, C.; Hou, D.; Song, J.; Du, G. A Sustainable Tannin-Citric Acid Wood Adhesive With Favorable Bonding Properties And Water Resistance. Ind. Crops Prod. 2023, 201, 116933 DOI: 10.1016/j.indcrop.2023.116933Google ScholarThere is no corresponding record for this reference.
- 158Liu, M.; Yao, W.; Shan, J.; Zheng, H.; Yang, Y.; Cao, L.; Qi, B.; Tan, H.; Sun, C.; Zhang, X.; Zhang, Y. Preparation Of High-Performance Antibacterial/Antifungal Citric Acid–Starch Adhesives Based On Physical Entanglement And Chemical Crosslinking. Int. J. Biol. Macromol. 2024, 279, 135560 DOI: 10.1016/j.ijbiomac.2024.135560Google ScholarThere is no corresponding record for this reference.
- 159Liu, X.; Allais, F.; Fadlallah, S. Itaconic Acid As A Platform Chemical For Bio-Based Polymers: From Green Polymerization Strategies To Structure-Driven Applications. Green Chem. 2026, 28 (8), 3371– 3393, DOI: 10.1039/D5GC06410DGoogle ScholarThere is no corresponding record for this reference.
- 160Satoh, K.; Lee, D.-H.; Nagai, K.; Kamigaito, M. Precision Synthesis Of Bio-Based Acrylic Thermoplastic Elastomer By Raft Polymerization Of Itaconic Acid Derivatives. Macromol. Rapid Commun. 2014, 35 (2), 161– 167, DOI: 10.1002/marc.201300638Google ScholarThere is no corresponding record for this reference.
- 161Pradeep, S. V.; Kandasubramanian, B.; Sidharth, S. A Review On Recent Trends In Bio-Based Pressure Sensitive Adhesives. J. Adhes. 2023, 99 (14), 2145– 2166, DOI: 10.1080/00218464.2023.2176761Google ScholarThere is no corresponding record for this reference.
- 162Li, B.; Torres, G. B.; Martin, B.; Taylor, N.; Barbu, E.; Christie, A.; Heise, A. Polycaprolactone–Itaconic Acid Resins For Additive Manufacturing Of Environmentally Degradable 3D And 4D Materials By Thiol-Ene Photopolymerization. Macromolecules 2025, 58 (16), 8887– 8897, DOI: 10.1021/acs.macromol.5c01310Google ScholarThere is no corresponding record for this reference.
- 163Maturi, M.; Spanu, C.; Maccaferri, E.; Locatelli, E.; Benelli, T.; Mazzocchetti, L.; Sambri, L.; Giorgini, L.; Franchini, M. C. (Meth)Acrylate-Free Three-Dimensional Printing Of Bio-Derived Photocurable Resins With Terpene- And Itaconic Acid-Derived Poly(Ester-Thioether)S. ACS Sustainable Chem. Eng. 2023, 11 (49), 17285– 17298, DOI: 10.1021/acssuschemeng.3c04576Google ScholarThere is no corresponding record for this reference.
- 164Zhang, Y.; Lin, Z.; Li, X.; Li, G. One-Step And Scalable Synthesis Of Eaa-Based Reprocessable Vitrimer With Superior Mechanical Properties. J. Polym. Environ. 2024, 32 (3), 1080– 1089, DOI: 10.1007/s10924-023-03030-6Google ScholarThere is no corresponding record for this reference.
- 165Madduluri, V. R.; Bendi, A.; Chinmay; Maniam, G. P.; Roslan, R.; Ab Rahim, M. H. Recent Advances In Vitrimers: A Detailed Study On The Synthesis, Properties And Applications Of Bio-Vitrimers. J. Polym. Environ. 2025, 33 (1), 301– 322, DOI: 10.1007/s10924-024-03416-0Google ScholarThere is no corresponding record for this reference.
- 166Zhang, J.; Gong, Z.; Wu, C.; Li, T.; Tang, Y.; Wu, J.; Jiang, C.; Miao, M.; Zhang, D. Itaconic Acid-Based Hyperbranched Polymer Toughened Epoxy Resins With Rapid Stress Relaxation, Superb Solvent Resistance And Closed-Loop Recyclability. Green Chem. 2022, 24 (18), 6900– 6911, DOI: 10.1039/D2GC01541BGoogle ScholarThere is no corresponding record for this reference.
- 167Qi, Y.; Zhang, Y.; Ou, S.; Zhuo, G.; Zeng, H.; Lao, Y.; Wang, J.; Wang, Q.; Guo, C. Green And Sustainable Itaconic Acid-Based Vitrimers With Rapid Stress Relaxation, Superior Fire Safety, And Recyclability Ivia/I Synergistic Roles Of Multiple Dynamic Bonds. Green Chem. 2025, 27 (42), 13385– 13403, DOI: 10.1039/D5GC04554AGoogle ScholarThere is no corresponding record for this reference.
- 168Kong, F.; Ma, X.; Xu, X.; Cui, M.; Zhao, H.; Zhu, J.; Chen, J. Itaconic Acid-Based Sustainable Polyurethane Covalent Adaptable Networks With Robust Mechanical, Reshaping And Uv-Resistant Properties Based On Reversible Disulfide Bond. Mater. Today Chem. 2024, 35, 101881 DOI: 10.1016/j.mtchem.2023.101881Google ScholarThere is no corresponding record for this reference.
- 169Nouigues, A.; Le Gal La Salle, E.; Bailleul, J.-L. Thermo-Mechanical Characterization Of Unsaturated Polyester/Glass Fiber Composites For Recycling. Int. J. Mater. Form. 2021, 14 (1), 153– 174, DOI: 10.1007/s12289-020-01559-8Google ScholarThere is no corresponding record for this reference.
- 170Arnaud, S. P.; Andreou, E.; Köster, L. V. G. P.; Robert, T. Selective Synthesis Of Monoesters Of Itaconic Acid With Broad Substrate Scope: Biobased Alternatives To Acrylic Acid?. ACS Sustainable Chem. Eng. 2020, 8 (3), 1583– 1590, DOI: 10.1021/acssuschemeng.9b06330Google ScholarThere is no corresponding record for this reference.
- 171Shao, W.; Wang, P.; Liu, J.; Xu, H.; Cai, X.; Wu, Q.; Xia, N.; Kong, F. An Environmentally Friendly And High Wet-Bond Strength Adhesive Based On Starch, Itaconic Acid And Silicone. Industrial Crops and Products 2022, 181, 114809, DOI: 10.1016/j.indcrop.2022.114809Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
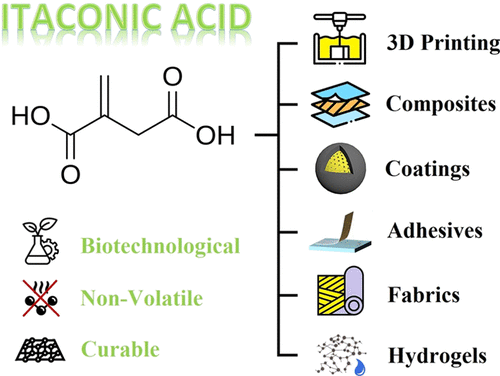
Figure 1

Figure 1. Graphical illustration of the itaconic acid’s molecular structure contains two carboxylic functional groups exhibiting the reactive delocalized electrons in resonance, a radically polymerizable terminal alkene functional group, and the schematic potential functional derivatives, such as amides (derived from amines and acids), esters (derived from alcohols and acids), and anhydride (derived from two acids).
Figure 2

Figure 2. Primary parameters influencing the stereolithography 3D printing. (a) Irradiation source type (the characteristics wavelength and power value), (b) the specific reactivity of the precursor (determined by Jacob’s working curve or differential scanning calorimetry (DSC)), (c) the rheological profile of the resin-forming systems (dependent on the temperature by the Arrhenius law and affected by the reactive diluent’s content) (d) the published diluting properties of the experimentally applied itaconate derivaties. (84) Reproduced from ref (84). Copyright [2017] American Chemical Society.
Figure 3

Figure 3. (a) Itaconic acid–based polyester used in combination with 4-acryloyl morpholine (ACMO) as a reactive diluent, (60) Reproduced from ref (60). Copyright [2023] American Chemical Society (b) oligomer itaconate precursor systems containing biobased reactive diluents derived from furfuryl, tetrahydrofurfuryl, solketal, and diacetone glucose reactive derivatives, (63) Reproduced from ref (63). Copyright [2025] American Chemical Society (c) Polylactone/itaconate elastomer resins processed by reversible addition–fragmentation chain transfer (RAFT) mediated 3D-printing with polypeptide surface functionalization, (67) Reproduced from ref (67). Copyright [2023] American Chemical Society (d) modified grapheme oxide (GO) nanoparticles incorporated into a itaconate-based 3D printing resin for the enhancement of the material properties while maintaining the advanced functional properties ensured by GO’s the conjugated π-electron system. (66) Reproduced from ref (66). Copyright [2025] American Chemical Society.
Figure 4

Figure 5

Figure 5. Complex synthetic scheme composed of reactions described and evaluated in the investigation provided by Arnaud and the team. (59) The scheme describes three different chemical approaches leading to the symmetric esters produced via Fischer esterification, asymmetric esters combining anhydride and epoxide compounds, and symmetric esters derived from epoxy entering compounds. Reproduced from ref (59). Copyright [2021] American Chemical Society.
Figure 6
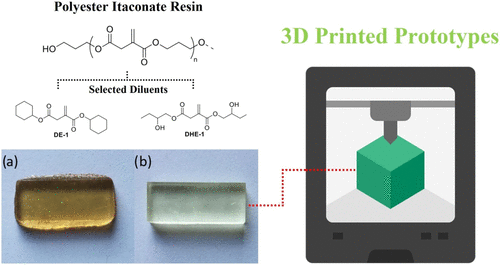
Figure 6. Illustrated polyester itaconic acid–based cured resins, modified with selected itaconate-reactive diluents, fabricated through 3D printing. (a) 3D-printed DMA specimen containing dicyclohexyl itaconate (DE-1), (b) 3D-printed DMA specimen containing bis(2-hydroxybutyl) itaconate (DHE-1). (59) Reproduced from ref (59). Copyright [2021] American Chemical Society.
Figure 7

Figure 7. Schematic illustration of the proposed fully biobased composite from itaconate resin and cotton fabrics as the solid-phase filler along with thermo-mechanical properties measured by the DMA analysis. (a–e) SEM pictures of the obtained composite prototype, (f) the photo of the fabricated composite. (100) Reproduced from ref (100). Copyright [2018] American Chemical Society.
Figure 8

Figure 8. Schematic list of potential and investigated application fields for itaconic acid–based polymeric coatings for material applications. The illustrated reaction scheme describes the modification of epoxidized soybean oil (ESO) with itaconic acid monoester, yielding itaconated epoxidized soybean oil (IESO), which represents a green alternative to the commercially used acrylated epoxidized soybean oil (AESO) for general coating applications. (101,102) Reproduced from ref (101) Copyright [2016] American Chemical Society and from ref (101) Copyright [2019] American Chemical Society.
Figure 9
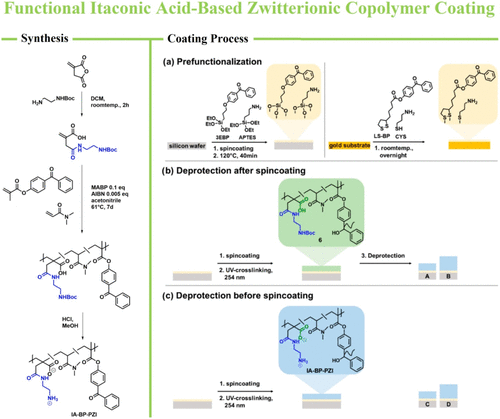
Figure 9. Synthesis of the deprotected zwitterionic itaconic acid involving copolymer (IA-BP-PZI) and the coating strategy ensuring the antimicrobial activity, protein repellency, and cell compatibility investigated by Schneider-Chaabane et al. (118) Reproduced from ref (118). Copyright [2019] American Chemical Society.
Figure 10
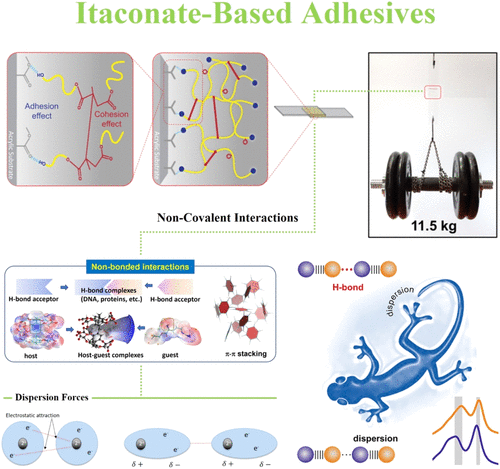
Figure 10. Schematic interpretation of itaconate-based photocurable adhesive investigated by Jamaludin et al., (119) and an illustration of the forming (and potential) noncovalent molecular interactions, such as hydrogen bonding, dispersion forces, and π–π stacking. (120−122) Reproduced from ref (119) Copyright [2022] American Chemical Society, from ref (120) Copyright [2023] American Chemical Society, from ref (121) Copyright [2023] American Chemical Society, and from ref (122) Copyright [2016] American Chemical Society.
Figure 11

Figure 12

Figure 12. (a) Highly crosslinked itaconic acid derivative for PSA, (b) itaconic acid ester system enhanced with acrylic derivatives for PSA. Investigated pressure-sensitive adhesives (PSA) containing itaconic acid and its derivatives. (147) Copyright (2024), American Chemical Society. The multicomponent cross-linkable PSA synthesized from sustainable sources is illustrated in the bottom-left corner, (145) while the underwater-performing PSAs for potential wound treatment are displayed in the right half. (146) Reproduced from ref (145) copyright [2025] American Chemical Society and from ref (146) copyright [2024] American Chemical Society.
Figure 13
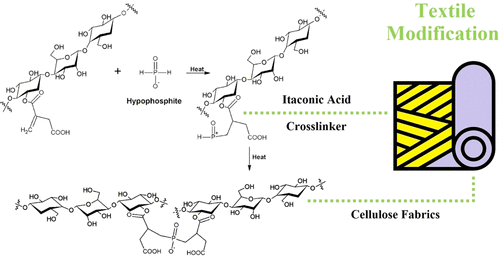
Figure 14
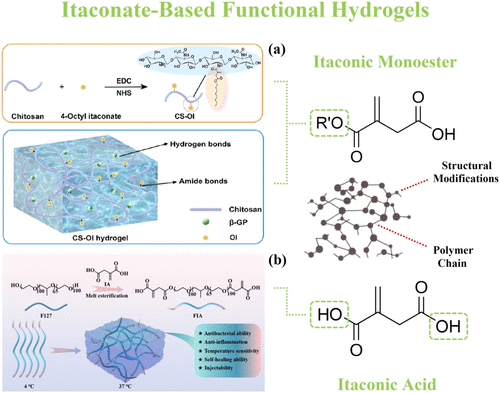
Figure 14. Two particular approaches for incorporating sustainable itaconic acid into the hydrogel structures. (a) The presynthesized itaconic monoesters serve as reactive molecules that can modify the structure of the primary hydrogel site. (154) Reproduced from ref (154). Copyright [2024] American Chemical Society. (b) Pure itaconic acid can be esterified onto the main polymer chain, providing the required properties. (153) Reproduced from ref (153). Copyright [2023] American Chemical Society.
References
This article references 171 other publications.
- 1Cywar, R. M.; Rorrer, N. A.; Hoyt, C. B.; Beckham, G. T.; Chen, E. Y.-X. Bio-Based Polymers With Performance-Advantaged Properties. Nat. Rev. Mater. 2022, 7 (2), 83– 103, DOI: 10.1038/s41578-021-00363-3There is no corresponding record for this reference.
- 2Álvarez-Chávez, C. R.; Edwards, S.; Moure-Eraso, R.; Geiser, K. Sustainability Of Bio-Based Plastics: General Comparative Analysis And Recommendations For Improvement. J. Cleaner Prod. 2012, 23 (1), 47– 56, DOI: 10.1016/j.jclepro.2011.10.003There is no corresponding record for this reference.
- 3Lee, J.-Y.; Lee, S.-E.; Lee, D.-W. Current Status And Future Prospects Of Biological Routes To Bio-Based Products Using Raw Materials, Wastes, And Residues As Renewable Resources. Crit. Rev. Environ. Sci. Technol. 2022, 52 (14), 2453– 2509, DOI: 10.1080/10643389.2021.1880259There is no corresponding record for this reference.
- 4Wang, Z. Statistical Analysis Of The Structure Of Total Petroleum Consumption In China. IOP Conf. Ser.: Earth Environ. Sci. 2019, 300 (4), 042089 DOI: 10.1088/1755-1315/300/4/042089There is no corresponding record for this reference.
- 5Patidar, A. K.; Jain, P.; Dhasmana, P.; Choudhury, T. Impact Of Global Events On Crude Oil Economy: A Comprehensive Review Of The Geopolitics Of Energy And Economic Polarization. GeoJournal 2024, 89 (2), 50 DOI: 10.1007/s10708-024-11054-1There is no corresponding record for this reference.
- 6Soto, J. M.; Martín-Lara, M. A.; Blázquez, G.; Godoy, V.; Quesada, L.; Calero, M. Novel Pre-Treatment Of Dirty Post-Consumer Polyethylene Film For Its Mechanical Recycling. Process Saf. Environ. Prot. 2020, 139, 315– 324, DOI: 10.1016/j.psep.2020.04.044There is no corresponding record for this reference.
- 7Streit, A. F. M.; de Santana, M. P.; de Oliveira Júnior, D. L.; Bassaco, M. M.; Tanabe, E. H.; Dotto, G. L.; Bertuol, D. A. Development Of A Pre-Treatment Process Of Polymeric Wastes (Hdpe, Ldpe/Lldpe, Pp) For Application In The Qualification Of Selectors Of Recyclable Materials. Environ. Dev. Sustainability 2022, 24 (5), 6349– 6371, DOI: 10.1007/s10668-021-01705-5There is no corresponding record for this reference.
- 8Zhang, X.; Zu, W.; Lee, L. Y. S. Crucial Role Of Pre-Treatment In Plastic Photoreforming For Precision Upcycling. npj Mater. Sustainability 2025, 3 (1), 3 DOI: 10.1038/s44296-024-00045-5There is no corresponding record for this reference.
- 9Schade, A.; Melzer, M.; Zimmermann, S.; Schwarz, T.; Stöwe, K.; Kühn, H. Plastic Waste Recycling─A Chemical Recycling Perspective. ACS Sustainable Chem. Eng. 2024, 12 (33), 12270– 12288, DOI: 10.1021/acssuschemeng.4c02551There is no corresponding record for this reference.
- 10Sandoval, S. S.; Amenábar, A.; Toledo, I.; Silva, N.; Contreras, P. Advances In The Sustainable Development Of Biobased Materials Using Plant And Animal Waste As Raw Materials: A Review. Sustainability 2024, 16 (3), 1073 DOI: 10.3390/su16031073There is no corresponding record for this reference.
- 11Rajput, C. V.; Sastry, N.V.; Chikhaliya, N. P. Vegetable Oils Based Precursors: Modifications And Scope For Futuristic Bio-Based Polymeric Materials. J. Polym. Res. 2023, 30 (4), 159 DOI: 10.1007/s10965-023-03534-8There is no corresponding record for this reference.
- 12Wahlen, C.; Frey, H. Anionic Polymerization Of Terpene Monomers: New Options For Bio-Based Thermoplastic Elastomers. Macromolecules 2021, 54 (16), 7323– 7336, DOI: 10.1021/acs.macromol.1c00770There is no corresponding record for this reference.
- 13Zhu, Y.; Romain, C.; Williams, C. K. Sustainable Polymers From Renewable Resources. Nature 2016, 540 (7633), 354– 362, DOI: 10.1038/nature21001There is no corresponding record for this reference.
- 14Kharissova, O. V.; Kharisov, B. I.; González, C. M. O.; Méndez, Y. P.; López, I. Greener Synthesis Of Chemical Compounds And Materials. R. Soc. Open Sci. 2019, 6 (11), 191378 DOI: 10.1098/rsos.191378There is no corresponding record for this reference.
- 15Miturska-Barańska, I.; Rudawská, A.; Sobotová, L.; Badida, M.; Olewnik-Kruszkowska, E.; Müller, M.; Hromasová, M. Analysis Of Acoustic Absorption Coefficients And Characterization Of Epoxy Adhesive Compositions Based On The Reaction Product Of Bisphenol A With Epichlorohydrin Modified With Fillers. Materials 2024, 17 (18), 4452 DOI: 10.3390/ma17184452There is no corresponding record for this reference.
- 16Wang, X.; Li, Y.; Jiang, Y.; Meng, L.; Nie, Z. In-Depth Free Fatty Acids Annotation Of Edible Oil By Mcpba Epoxidation And Tandem Mass Spectrometry. Food Chem. 2022, 374, 131793 DOI: 10.1016/j.foodchem.2021.131793There is no corresponding record for this reference.
- 17Samyn, P.; Bosmans, J.; Cosemans, P. Role Of Bio-Based And Fossil-Based Reactive Diluents In Epoxy Coatings With Amine And Phenalkamine Crosslinker. Polymers 2023, 15 (19), 3856 DOI: 10.3390/polym15193856There is no corresponding record for this reference.
- 18Pradhan, S.; Pandey, P.; Mohanty, S.; Nayak, S. K. Insight On The Chemistry Of Epoxy And Its Curing For Coating Applications: A Detailed Investigation And Future Perspectives. Polym.-Plast. Technol. Eng. 2016, 55 (8), 862– 877, DOI: 10.1080/03602559.2015.1103269There is no corresponding record for this reference.
- 19Pappa, C.; Féghali, E.; Vanbroekhoven, K.; Triantafyllidis, K. S. Recent Advances In Epoxy Resins And Composites Derived From Lignin And Related Bio-Oils. Curr. Opin. Green Sustainable Chem. 2022, 38, 100687 DOI: 10.1016/j.cogsc.2022.100687There is no corresponding record for this reference.
- 20Ahmat, Y. M.; Madadi, S.; Charbonneau, L.; Kaliaguine, S. Epoxidation Of Terpenes. Catalysts 2021, 11 (7), 847 DOI: 10.3390/catal11070847There is no corresponding record for this reference.
- 21Kumar, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Recent Development Of Biobased Epoxy Resins: A Review. Polym.-Plast. Technol. Eng. 2018, 57 (3), 133– 155, DOI: 10.1080/03602559.2016.1253742There is no corresponding record for this reference.
- 22Ingle, A. A.; Ansari, S. Z.; Shende, D. Z.; Wasewar, K. L.; Pandit, A. B. Progress And Prospective Of Heterogeneous Catalysts For H2O2 Production Via Anthraquinone Process. Environ. Sci. Pollut. Res. 2022, 29 (57), 86468– 86484, DOI: 10.1007/s11356-022-21354-zThere is no corresponding record for this reference.
- 23Meadows, S.; Hosur, M.; Celikbag, Y.; Jeelani, S. Comparative Analysis On The Epoxidation Of Soybean Oil Using Formic And Acetic Acids. Polym. Polym. Compos. 2018, 26 (4), 289– 298, DOI: 10.1177/096739111802600403There is no corresponding record for this reference.
- 24Meng, Y.; Taddeo, F.; Aguilera, A. F.; Cai, X.-S.; Russo, V.; Tolvanen, P.; Leveneur, S. The Lord Of The Chemical Rings: Catalytic Synthesis Of Important Industrial Epoxide Compounds. Catalysts 2021, 11 (7), 765 DOI: 10.3390/catal11070765There is no corresponding record for this reference.
- 25Mao, S.; Budweg, S.; Spannenberg, A.; Wen, X.; Yang, Y.; Li, Y.; Junge, K.; Beller, M. Iron-Catalyzed Epoxidation Of Linear A-Olefins With Hydrogen Peroxide. ChemCatChem 2022, 14 (4), e202101668 DOI: 10.1002/cctc.202101668There is no corresponding record for this reference.
- 26Găină, C.; Ursache, O.; Gaina, V.; Serban, A.-M.; Asăndulesa, M. Novel Bio-Based Materials: From Castor Oil To Epoxy Resins For Engineering Applications. Materials 2023, 16 (16), 5649 DOI: 10.3390/ma16165649There is no corresponding record for this reference.
- 27Bodhak, C.; Patel, D.; Gupta, R. K. Mechanically Robust, Self-Healable, And Reprocessable Geraniol-Based Epoxy Vitrimer By Dynamic Boronic Ester Bonds. ACS Appl. Eng. Mater. 2025, 3 (6), 1599– 1612, DOI: 10.1021/acsaenm.5c00155There is no corresponding record for this reference.
- 28Zhu, Z.; Zhang, E.; Tu, Y.; Ye, M.; Chen, N. An Eco-Friendly Wood Adhesive Consisting Of Soybean Protein And Cardanol-Based Epoxy For Wood Based Composites. Polymers 2022, 14 (14), 2831 DOI: 10.3390/polym14142831There is no corresponding record for this reference.
- 29Yuan, J. S.; Pavlovich, M. J.; Ragauskas, A. J.; Han, B. Biotechnology For A Sustainable Future: Biomass And Beyond. Trends Biotechnol. 2022, 40 (12), 1395– 1398, DOI: 10.1016/j.tibtech.2022.09.020There is no corresponding record for this reference.
- 30Teleky, B.-E.; Vodnar, D. C. Recent Advances In Biotechnological Itaconic Acid Production, And Application For A Sustainable Approach. Polymers 2021, 13 (20), 3574 DOI: 10.3390/polym13203574There is no corresponding record for this reference.
- 31Zhang, R.; Liu, H.; Ning, Y.; Yu, Y.; Deng, L.; Wang, F. Recent Advances On The Production Of Itaconic Acid Via The Fermentation And Metabolic Engineering. Fermentation 2023, 9 (1), 71 DOI: 10.3390/fermentation9010071There is no corresponding record for this reference.
- 32Sollka, L.; Lienkamp, K. Progress In The Free And Controlled Radical Homo- And Co-Polymerization Of Itaconic Acid Derivatives: Toward Functional Polymers With Controlled Molar Mass Distribution And Architecture. Macromol. Rapid Commun. 2021, 42 (4), e2000546 DOI: 10.1002/marc.202000546There is no corresponding record for this reference.
- 33Kumar, S.; Krishnan, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Itaconic Acid Used As A Versatile Building Block For The Synthesis Of Renewable Resource-Based Resins And Polyesters For Future Prospective: A Review. Polym. Int. 2017, 66 (10), 1349– 1363, DOI: 10.1002/pi.5399There is no corresponding record for this reference.
- 34Yang, S.; He, Y.; Zhang, J. High-Performance Acrylate-Free Co-Cured Coatings Enabled By Tailoring Itaconic Acid-Modified Dual-Phase Photocurable Resins. Prog. Org. Coat. 2024, 189, 108293 DOI: 10.1016/j.porgcoat.2024.108293There is no corresponding record for this reference.
- 35Mu, S.; Guo, J.; Yu, Y.; An, Q.; Zhang, S.; Wang, D.; Chen, S.; Huang, X.; Li, S. Synthesis And Thermal Properties Of Cross-Linked Poly(Acrylonitrile-Co-Itaconate)/Polyethylene Glycol As Novel Form-Stable Change Material. Energy Convers. Manage. 2016, 110, 176– 183, DOI: 10.1016/j.enconman.2015.12.004There is no corresponding record for this reference.
- 36Robert, T.; Friebel, S. Itaconic Acid – A Versatile Building Block For Renewable Polyesters With Enhanced Functionality. Green Chem. 2016, 18 (10), 2922– 2934, DOI: 10.1039/C6GC00605AThere is no corresponding record for this reference.
- 37Campbell, S. B.; Wu, Q.; Yazbeck, J.; Liu, C.; Okhovatian, S.; Radisic, M. Beyond Polydimethylsiloxane: Alternative Materials For Fabrication Of Organ-On-A-Chip Devices And Microphysiological Systems. ACS Biomater. Sci. Eng. 2021, 7 (7), 2880– 2899, DOI: 10.1021/acsbiomaterials.0c00640There is no corresponding record for this reference.
- 38Fathi, M.; Alami-Milani, M.; Geranmayeh, M. H.; Barar, J.; Erfan-Niya, H.; Omidi, Y. Dual Thermo-And Ph-Sensitive Injectable Hydrogels Of Chitosan/(Poly(N-Isopropylacrylamide-Co-Itaconic Acid)) For Doxorubicin Delivery In Breast Cancer. Int. J. Biol. Macromol. 2019, 128, 957– 964, DOI: 10.1016/j.ijbiomac.2019.01.122There is no corresponding record for this reference.
- 39Kim, H. C.; Kwon, Y. R.; Kim, J. S.; Kim, J.-H.; Kim, D. H. Surface-Crosslinking In The Presence Of Nanoclay And Characteristics Of The Itaconic Acid-Based Superabsorbent Polymer Composites. Polym.-Plast. Technol. Mater. 2023, 62 (6), 701– 711, DOI: 10.1080/25740881.2022.2133613There is no corresponding record for this reference.
- 40Bantchev, G. B.; Doll, K. M. Comparative Amine-Catalyzed Thia-Michael Reactions Of Primary And Secondary Thiols With Maleic And Itaconic Anhydrides And Esters. ChemistrySelect 2022, 7 (48), e202204138 DOI: 10.1002/slct.202204138There is no corresponding record for this reference.
- 41Niang, F.; Brunou-Bouard, A.; Cruz, G.; Pantoustier, N.; Coumes, F.; Illy, N. Synthesis Of Biobased And Versatile Monomers From Itaconic Acid And Homocysteine Thiolactone And Their Applications In Step-Growth And Radical Polymerization Approaches. Polym. Chem. 2024, 15 (35), 3597– 3607, DOI: 10.1039/D4PY00565AThere is no corresponding record for this reference.
- 42Stevens, L. M.; Almada, N. T.; Kim, H. S.; Page, Z. A. Visible-Light-Fueled Polymerizations For 3D Printing. Acc. Chem. Res. 2025, 58 (2), 250– 260, DOI: 10.1021/acs.accounts.4c00680There is no corresponding record for this reference.
- 43Kim, G.-T.; Go, H.-B.; Yu, J.-H.; Yang, S.-Y.; Kim, K.-M.; Choi, S.-H.; Kwon, J.-S. Cytotoxicity, Colour Stability And Dimensional Accuracy Of 3D Printing Resin With Three Different Photoinitiators. Polymers 2022, 14 (5), 979 DOI: 10.3390/polym14050979There is no corresponding record for this reference.
- 44Xu, M.; Liu, M.; Wang, F.; Chi, H. Strategies To Improve Water Solubility Of Photoinitiator For Photopolymerization-Based 3D Printing In Biomedical Applications. ACS Appl. Polym. Mater. 2025, 7 (7), 4077– 4092, DOI: 10.1021/acsapm.5c00184There is no corresponding record for this reference.
- 45Šimunović, L.; Brenko, L.; Marić, A. J.; Meštrović, S.; Haramina, T. Rheology Of Dental Photopolymers For Sla/Dlp/Msla 3D Printing. Polymers 2025, 17 (19), 2706 DOI: 10.3390/polym17192706There is no corresponding record for this reference.
- 46Vyas, A.; Garg, V.; Ghosh, S. B.; Bandyopadhyay-Ghosh, S. Photopolymerizable Resin-Based 3D Printed Biomedical Composites: Factors Affecting Resin Viscosity. Mater. Today Proc. 2022, 62, 1435– 1439, DOI: 10.1016/j.matpr.2022.01.172There is no corresponding record for this reference.
- 47Konuray, O.; Morancho, J. M.; Fernández-Francos, X.; García-Alvarez, M.; Ramis, X. Curing Kinetics Of Dually-Processed Acrylate-Epoxy 3D Printing Resins. Thermochim. Acta 2021, 701, 178963 DOI: 10.1016/j.tca.2021.178963There is no corresponding record for this reference.
- 48Uzcategui, A. C.; Muralidharan, A.; Ferguson, V. L.; Bryant, S. J.; McLeod, R. R. Understanding And Improving Mechanical Properties In 3D Printed Parts Using A Dual-Cure Acrylate-Based Resin For Stereolithography. Adv. Eng. Mater. 2018, 20 (12), 1800876 DOI: 10.1002/adem.201800876There is no corresponding record for this reference.
- 49Kolibaba, T. J.; Killgore, J. P.; Caplins, B. W.; Higgins, C. I.; Arp, U.; Miller, C.; Poster, D. L.; Zong, Y.; Broce, S.; Wang, T.; Talačka, V.; Andersson, J.; Davenport, A. T.; Panzer, M. A.; Tumbleston, J. R.; Gonzalez, J. M.; Huffstetler, J.; Lund, B.; Billerbeck, K.; Clay, A.; Fratarcangeli, M. R.; Qi, H. J.; Porcincula, D. H.; Bezek, L. B.; Kikuta, K.; Pearlson, M. N.; Walker, D.; Long, C. J.; Hasa, E.; Aguirre-Soto, A.; Celis-Guzman, A.; Backman, D. E.; Sridhar, R. L.; Cavicchi, K. A.; Viereckl, R. J.; Tong, E.; Hansen, C. J.; Shah, D. M.; Kinane, C.; Pena-Francesch, A.; Antonini, C.; Chaudhary, R.; Muraca, G.; Bensouda, Y.; Zhang, Y.; Zhao, X. Results Of An Interlaboratory Study On The Working Curve In Vat Photopolymerization. Addit. Manuf. 2024, 84, 104082 DOI: 10.1016/j.addma.2024.104082There is no corresponding record for this reference.
- 50Li, Y.; Mao, Q.; Yin, J.; Wang, Y.; Fu, J.; Huang, Y. Theoretical Prediction And Experimental Validation Of The Digital Light Processing (Dlp) Working Curve For Photocurable Materials. Addit. Manuf. 2021, 37, 101716 DOI: 10.1016/j.addma.2020.101716There is no corresponding record for this reference.
- 51Hofstetter, C. P.; Orman, S.; Baudis, S.; Stampfl, J. Combining Cure Depth And Cure Degree, A New Way To Fully Characterize Novel Photopolymers. Addit. Manuf. 2018, 24, 166– 172, DOI: 10.1016/j.addma.2018.09.025There is no corresponding record for this reference.
- 52Bösche, C.; Hagenlocher, L.; Bianchi, E.; Bragato, N.; Lotti, N.; Robert, T. Biobased Uv-Curing Additive Manufacturing Materials Derived From Itaconic Acid–Based Polyester Resin And Biobased Reactive Diluents From (Meth)Acrylic Acid. ACS Sustainable Chem. Eng. 2025, 13 (44), 19405– 19415, DOI: 10.1021/acssuschemeng.5c09188There is no corresponding record for this reference.
- 53Gupta, A.; Salajeghe, R.; Spangenberg, J.; Marla, D. A Computational Model For Stereolithography Apparatus (Sla) 3D Printing. Prog. Addit. Manuf. 2023, 9 (6), 1605– 1619, DOI: 10.1007/s40964-023-00525-5There is no corresponding record for this reference.
- 54Yi, X.; Kuang, X.; Kong, L.; Dong, X.; Feng, Z.; Wang, D. A Simplified Chemorheological Model Of Viscosity Evolution For Solvent Containing Resol Resin In Scprtm/Scp Process. J. Appl. Polym. Sci. 2017, 134 (36), 45282 DOI: 10.1002/app.45282There is no corresponding record for this reference.
- 55Kolibaba, T. J.; Caplins, B. W.; Higgins, C. I.; Mansfield, E.; Miller, S. L.; Chandler, C.; Killgore, J. P. Tailoring Properties Of 3D-Printable Polyelectrolyte Photopolymer Complexes With Reactive Diluents. ACS Appl. Polym. Mater. 2024, 6 (12), 6957– 6965, DOI: 10.1021/acsapm.4c00441There is no corresponding record for this reference.
- 56Hevus, I.; Tiwari, S.; Thorat, S. S.; Gibbon, L.; La Scala, J. J.; Ulven, C. A.; Sibi, M. P.; Webster, D. C. Vanillin-Derived Veratrole Reactive Diluents In Stereolithography. ACS Appl. Polym. Materi. 2024, 6 (13), 7705– 7715, DOI: 10.1021/acsapm.4c01183There is no corresponding record for this reference.
- 57Jagtap, A. R.; More, A. P. Developments In Reactive Diluents: A Review. Polym. Bull. 2022, 79 (8), 5667– 5708, DOI: 10.1007/s00289-021-03808-5There is no corresponding record for this reference.
- 58Wei, M. G.; Xu, H.; Chen, L.; Li, Z.; Liu, R. Isosorbide-Based High Performance Uv-Curable Reactive Diluents. Prog. Org. Coat. 2019, 126, 162– 167, DOI: 10.1016/j.porgcoat.2018.10.028There is no corresponding record for this reference.
- 59Arnaud, S. P.; Malitowski, N. M.; Casamayor, K. M.; Robert, T. Itaconic Acid-Based Reactive Diluents For Renewable And Acrylate-Free Uv-Curing Additive Manufacturing Materials. ACS Sustainable Chem. Eng. 2021, 9 (50), 17142– 17151, DOI: 10.1021/acssuschemeng.1c06713There is no corresponding record for this reference.
- 60Papadopoulos, L.; Pezzana, L.; Malitowski, N. M.; Sangermano, M.; Bikiaris, D. N.; Robert, T. Uv-Curing Additive Manufacturing Of Bio-Based Thermosets: Effect Of Diluent Concentration On Printing And Material Properties Of Itaconic Acid-Based Materials. ACS Omega 2023, 8 (34), 31009– 31020, DOI: 10.1021/acsomega.3c02808There is no corresponding record for this reference.
- 61Papadopoulos, L.; Malitowski, N. M.; Bikiaris, D. N.; Robert, T. Bio-Based Additive Manufacturing Materials: An In-Depth Structure-Property Relationship Study Of Uv-Curing Polyesters From Itaconic Acid. Eur. Polym. J. 2023, 186, 111872 DOI: 10.1016/j.eurpolymj.2023.111872There is no corresponding record for this reference.
- 62Cazin, I.; Ocepek, M.; Kecelj, J.; Stražar, A. S.; Schlögl, S. Synthesis Of Bio-Based Polyester Resins For Vat Photopolymerization 3D Printing. Materials 2024, 17 (8), 1890 DOI: 10.3390/ma17081890There is no corresponding record for this reference.
- 63Bösche, C.; Hagenlocher, L.; Bianchi, E.; Bragato, N.; Lotti, N.; Robert, T. Biobased Uv-Curing Additive Manufacturing Materials derived From Itaconic Acid–Based Polyester Resin And Biobased Reactive Diluents From (Meth)Acrylic Acid. ACS Sustainable Chem. Eng. 2025, 13 (44), 19405– 19415, DOI: 10.1021/acssuschemeng.5c09188There is no corresponding record for this reference.
- 64Gao, Y.; Li, Y.; Hu, X.; Wu, W.; Wang, Z.; Wang, R.; Zhang, L. Preparation And Properties Of Novel Thermoplastic Vulcanizate Based On Bio-Based Polyester/Polylactic Acid, And Its Application In 3D Printing. Polymers 2017, 9 (12), 694 DOI: 10.3390/polym9120694There is no corresponding record for this reference.
- 65Carmenini, R.; de León, A. S.; Benelli, T.; Giorgini, L.; Franchini, M. C.; Molina, S. I.; Maturi, M. One-Pot Depolymerization–Repolymerization Of Pet Waste Into Sustainable Photocurable Liquid Copolyesters For High-Performance Additive Manufacturing. Green Chem. 2025, 27 (40), 12830– 12843, DOI: 10.1039/d5gc02696bThere is no corresponding record for this reference.
- 66Maturi, M.; Maturi, S.; de León, A. S.; Migliorini, L.; de la Mata, M.; Benelli, T.; Giorgini, L.; Milani, P.; Franchini, M. C.; Molina, S. I. Enhanced Properties Of 3D-Printed Graphene Oxide Nanocomposites Through Itaconic Acid Polyester Grafting. ACS Appl. Polym. Mater. 2025, 7 (7), 4371– 4382, DOI: 10.1021/acsapm.5c00014There is no corresponding record for this reference.
- 67Torres, G. B.; Xia, T.; Yu, D.; Thijssen, Q.; Van Vlierberghe, S.; Li, B.; Heise, A. Raft-Mediated 3D Printing Of Polylactones/Itaconate Elastomers With Polypeptide Surface Functionalization. ACS Polym. Au 2025, 5, 956– 966, DOI: 10.1021/acspolymersau.5c00117There is no corresponding record for this reference.
- 68Papadopoulos, L.; Pezzana, L.; Malitowski, N. M.; Kladovasilakis, N.; Tzetzis, D.; Sangermano, M.; Bikiaris, D. N.; Robert, T. Itaconic Acid-Based 3D Printed Nanocomposites: An In-Depth Study On The Effect Of Nanoinclusions On The Physicochemical Properties And The Printability Of Formulations Based On Polyester Itaconates. Giant 2024, 18, 100275 DOI: 10.1016/j.giant.2024.100275There is no corresponding record for this reference.
- 69Lok, T.-J.; Wong, J.-W.; Li, X.; Fu, Y.; Xue, Y.; Jamaludin, F. H.; Fong, M.; Edward, E. B.; Ma, C.; Chandren, S.; Goh, P.-S.; Wong, T.-W. Biobased Itaconate Polyester Thermoset With Tunable Mechanical Properties. Macromolecules 2024, 57 (5), 2317– 2328, DOI: 10.1021/acs.macromol.3c01743There is no corresponding record for this reference.
- 70Spanu, C.; Locatelli, E.; Sambri, L.; Franchini, M. C.; Maturi, M. Photocurable Itaconic Acid-Functionalized Star Polycaprolactone In Biobased Formulations For Vat Photopolymerization. ACS Appl. Polym. Mater. 2024, 6 (4), 2417– 2424, DOI: 10.1021/acsapm.3c03159There is no corresponding record for this reference.
- 71Bokhari, S. M. Q.; Sevening, J. N.; Catchmark, J. M.; Chmely, S. C. Composition–Property Engineering Of Bio-Derived Uv-Curable Acrylate Oligoester Resins For Tunable Mechanics In 3D Printing. RSC Appl. Polym. 2025, 3 (4), 948– 959, DOI: 10.1039/d5lp00085hThere is no corresponding record for this reference.
- 72Miętus, M.; Gołofit, T.; Gadomska-Gajadhur, A. Towards Greener Polymers: Poly(Octamethylene Itaconate-Co-Succinate) Synthesis Parameters. Polymers 2025, 17 (16), 2220 DOI: 10.3390/polym17162220There is no corresponding record for this reference.
- 73Maturi, M.; Pulignani, C.; Locatelli, E.; Vetri Buratti, V.; Tortorella, S.; Sambri, L.; Franchini, M. C. Phosphorescent Bio-Based Resin For Digital Light Processing (Dlp) 3D-Printing. Green Chem. 2020, 22 (18), 6212– 6224, DOI: 10.1039/d0gc01983fThere is no corresponding record for this reference.
- 74Jao, C.-Y.; Mao, H.-I.; Shiu, J.-W.; Chen, Y.-C.; Chu, R.-J.; Chen, C.-W. Influence Of Diol Structure On The Mechanical And Thermal Properties Of Bio-Based Uv-Curable Unsaturated Polyesters For 3D Printing. ACS Appl. Polym. Mater. 2025, 7 (17), 11284– 11299, DOI: 10.1021/acsapm.5c01805There is no corresponding record for this reference.
- 75Carmenini, R.; Spanu, C.; Locatelli, E.; Sambri, L.; Franchini, M. C.; Maturi, M. Isocyanate-Free Urethanediol Itaconates As Biobased Liquid Monomers In Photopolymerization-Based 3D Printing. Prog. Addit. Manuf. 2024, 9 (6), 2499– 2510, DOI: 10.1007/s40964-024-00598-wThere is no corresponding record for this reference.
- 76Vetri Buratti, V.; Sanz de Leon, A.; Maturi, M.; Sambri, L.; Molina, S. I.; Comes Franchini, M. Itaconic-Acid-Based Sustainable Poly(Ester Amide) Resin For Stereolithography. Macromolecules 2022, 55 (8), 3087– 3095, DOI: 10.1021/acs.macromol.1c02525There is no corresponding record for this reference.
- 77Papadopoulos, L.; Kluge, M.; Bikiaris, D. N.; Robert, T. Straightforward Synthetic Protocol To Bio-Based Unsaturated Poly(Ester Amide)S From Itaconic Acid With Thixotropic Behavior. Polymers 2020, 12 (4), 980 DOI: 10.3390/polym12040980There is no corresponding record for this reference.
- 78Ouhichi, R.; Arnaud, S. P.; Bougarech, A.; Abid, S.; Abid, M.; Robert, T. First Example Of Unsaturated Poly(Ester Amide)S Derived From Itaconic Acid And Their Application As Bio-Based Uv-Curing Polymers. Appl. Sci. 2020, 10 (6), 2163, DOI: 10.3390/app10062163There is no corresponding record for this reference.
- 79Kumar, S.; Samal, S. K.; Mohanty, S.; Nayak, S. K. Synthesis And Characterization Of Itaconic-Based Epoxy Resins. Polym. Adv. Technol. 2018, 29 (1), 160– 170, DOI: 10.1002/pat.4098There is no corresponding record for this reference.
- 80Luo, Y.; Le Fer, G.; Dean, D.; Becker, M. L. 3D Printing Of Poly(Propylene Fumarate) Oligomers: Evaluation Of Resin Viscosity, Printing Characteristics And Mechanical Properties. Biomacromolecules 2019, 20 (4), 1699– 1708, DOI: 10.1021/acs.biomac.9b00076There is no corresponding record for this reference.
- 81Lu, D.; Pen, G.; Zhang, H.; Bao, J. Engineering Non-Covalent Interactions: Hierarchical Hydrogen-Bonding Strategy For Advanced 3D Printer Resins. Polymer 2025, 326, 128323 DOI: 10.1016/j.polymer.2025.128323There is no corresponding record for this reference.
- 82Montoya, J.; Medina, J.; Molina, A.; Gutierrez, J. A.; Rodriguez, B. E.; Marín, R. Impact Of Viscoelastic And Structural Properties From Starch-Mango And Starch-Arabinoxylans Hydrocolloids In 3D Food Printing. Addit. Manuf. 2021, 39, 101891 DOI: 10.1016/j.addma.2021.101891There is no corresponding record for this reference.
- 83Li, Y.; Wang, M.; Wu, T.; Yang, X.; Qu, Z.; Yao, X.; He, Y.; Guo, Y.; Pu, Q.; Wang, X. Green Synthesis Of Vat Photopolymerization 3D Printing Polyimide. Small 2025, 21 (27), e2502406 DOI: 10.1002/smll.202502406There is no corresponding record for this reference.
- 84Panić, V. V.; Šešlija, S. I.; Popović, I. G.; Spasojević, V.; Popović, A.; Nikolić, V.; Spasojević, P. Simple One-Pot Synthesis Of Fully Biobased Unsaturated Polyester Resins Based On Itaconic Acid. Biomacromolecules 2017, 18 (12), 3881– 3891, DOI: 10.1021/acs.biomac.7b00840There is no corresponding record for this reference.
- 85Deng, K.; Zhang, C.; Fu, K. Additive Manufacturing Of Continuously Reinforced Thermally Curable Thermoset Composites With Rapid Interlayer Curing. Composites, Part B 2023, 257, 110671, DOI: 10.1016/j.compositesb.2023.110671There is no corresponding record for this reference.
- 86Lee, J.; Ni, X.; Daso, F.; Xiao, X.; King, D. C.; Gómez, J. S.; Varela, T. B.; Kessler, S. S.; Wardle, B. L. Advanced Carbon Fiber Composite Out-Of-Autoclave Laminate Manufacture Via Nanostructured Out-Of-Oven Conductive Curing. Compos. Sci. Technol. 2018, 166, 150– 159, DOI: 10.1016/j.compscitech.2018.02.031There is no corresponding record for this reference.
- 87Li, Y.; Cheng, L.; Zhou, J. Curing Multidirectional Carbon Fiber Reinforced Polymer Composites With Indirect Microwave Heating. Int. J. Adv. Manuf. Technol. 2018, 97 (1–4), 1137– 1147, DOI: 10.1007/s00170-018-1974-1There is no corresponding record for this reference.
- 88Zanjanijam, A. R.; Wang, X.; Ramezani, M.; Holberg, S.; Johnson, P. A. Phenolic Resin/Coal Char Composites: Curing Kinetics And Thermal/Mechanical Performance. Polymer 2023, 281, 126103 DOI: 10.1016/j.polymer.2023.126103There is no corresponding record for this reference.
- 89Chen, J.; Gao, X.; Zhao, K.-Q.; Wu, W. Research On Curing Kinetics Of Multiaxial Warp-Knitted Composites. Polym. Compos. 2023, 44 (9), 5934 DOI: 10.1002/pc.27538There is no corresponding record for this reference.
- 90Xiong, X.; Guan, H.; Li, B.; Yang, S.; Li, W.; Ren, R.; Wang, J.; Chen, P. Cure Kinetics And Thermal Decomposition Behavior Of Novel Phenylacetylene-Capped Polyimide Resins. Polymers 2024, 16 (8), 1149, DOI: 10.3390/polym16081149There is no corresponding record for this reference.
- 91Guo, Z.; Yi-liang, W.; Chiumsenti, M.; Wu, Y.; Zhen, S.; Liu, H.; Rossi, R.; Fu, X. Research On Curing Reaction Kinetics And Curing Process Of Hydroxy-Terminated Polybutadiene (Htpb) Propellants. Sci. Rep. 2025, 15 (1), 21006 DOI: 10.1038/s41598-025-07125-zThere is no corresponding record for this reference.
- 92Matykiewicz, D.; Dudziec, B. Curing And Degradation Kinetics Of Phosphorus-Modified Eugenol-Based Epoxy Resin. ACS Omega 2025, 10 (5), 4353– 4366, DOI: 10.1021/acsomega.4c06532There is no corresponding record for this reference.
- 93Li, X.; Yao, H.; Lu, X.; Chen, C.; Cao, Y.; Xin, Z. Effect Of Pyrogallol On The Ring-Opening Polymerization And Curing Kinetics Of A Fully Bio-Based Benzoxazine. Thermochim. Acta 2020, 694, 178787 DOI: 10.1016/j.tca.2020.178787There is no corresponding record for this reference.
- 94Marotta, A.; Faggio, N.; Brondi, C. Curing Kinetics Of Bioderived Furan-Based Epoxy Resins: Study On The Effect Of The Epoxy Monomer/Hardener Ratio. Polymers 2022, 14 (23), 5322 DOI: 10.3390/polym14235322There is no corresponding record for this reference.
- 95Wang, Y.; Zhang, F.; Li, F.; Du, S.; Dai, S.; Jiang, T.; Sun, K.; Liu, X.; Ma, S. Covalent Adaptable Networks Derived From Itaconic Acid-Based Epoxy And Sulfur: Facile Synthesis And Applications In Adhesives And Carbon Fiber Composites. Compos. Commun. 2025, 59, 102575 DOI: 10.1016/j.coco.2025.102575There is no corresponding record for this reference.
- 96Xiao, K.; Fang, Y.; Wang, Z.; Ni, N.; Liu, Z.; Kim, S.; An, Z.; Lyu, Z.; Xu, Y.; Yang, X. Bio-Sourced, High-Performance Carbon Fiber Reinforced Itaconic Acid-Based Epoxy Composites With High Hygrothermal Stability And Durability. Polymers 2024, 16 (12), 1649 DOI: 10.3390/polym16121649There is no corresponding record for this reference.
- 97Shang, L.; Zhang, J.; Liu, L.; Xiao, L.; Li, M.; Ao, Y. Improving The Interfacial Property Of Carbon Fibre/Epoxy Resin Composites By Grafting Amine-Capped Cross-Linked Poly-Itaconic Acid. Surf. Interface Anal. 2019, 51 (2), 199– 209, DOI: 10.1002/sia.6565There is no corresponding record for this reference.
- 98Kocaman, S.; Akyay, İ. Itaconic Acid Based Epoxy Resin And Application Of Olive Pomace On The Production Of Composite Materials. Ind. Crops Prod. 2024, 222, 120022 DOI: 10.1016/j.indcrop.2024.120022There is no corresponding record for this reference.
- 99Ghosh, T.; Elo, T.; Parihar, V. S.; Maiti, P.; Layek, R. K. Poly (Itaconic Acid) Functionalized Lignin/Polyvinyl Acetate Composite Resin With Improved Sustainability And Wood Adhesion Strength. Ind. Crops Prod. 2022, 187, 115299 DOI: 10.1016/j.indcrop.2022.115299There is no corresponding record for this reference.
- 100Dai, Z.; Yang, Z.; Chen, Z.; Zhao, Z.; Lou, Y.; Zhang, Y.; Liu, T. X.; Fu, F.; Fu, Y.; Liu, X. Fully Biobased Composites Of An Itaconic Acid Derived Unsaturated Polyester Reinforced With Cotton Fabrics. ACS Sustainable Chem. Eng. 2018, 6 (11), 15056– 15063, DOI: 10.1021/acssuschemeng.8b03539There is no corresponding record for this reference.
- 101Li, P.; Ma, S.; Dai, J.; Liu, X.; Jiang, Y.; Wang, S.; Wei, J.; Chen, J.; Zhu, J. Itaconic Acid As A Green Alternative To Acrylic Acid For Producing A Soybean Oil-Based Thermoset: Synthesis And Properties. ACS Sustainable Chem. Eng. 2016, 5 (1), 1228– 1236, DOI: 10.1021/acssuschemeng.6b02654There is no corresponding record for this reference.
- 102Luo, J.; Ma, B.; Peng, J.; Wu, Z.; Luo, Z.; Wang, X. Modified Chestnut-Like Structure Silicon Carbon Composite As Anode Material For Lithium-Ion Batteries. ACS Sustainable Chem. Eng. 2019, 7 (12), 10415– 10424, DOI: 10.1021/acssuschemeng.9b00616There is no corresponding record for this reference.
- 103Kausar, A. Polymer Coating Technology For High Performance Applications: Fundamentals And Advances. J. Macromol. Sci., Part A 2018, 55 (5), 440– 448, DOI: 10.1080/10601325.2018.1453266There is no corresponding record for this reference.
- 104Pan, Y.; Liu, Y.; Yang, S.; Zhang, C.; Ullah, Z. Recent Research Progress On The Stimuli-Responsive Smart Membrane: A Review. Nanotechnol. Rev. 2023, 12 (1), 20220538 DOI: 10.1515/ntrev-2022-0538There is no corresponding record for this reference.
- 105K A, T.-Z.; A D, Z.; Yu, B. E.; I V, B.; E A, M. Foam Polymers In Multifunctional Insulating Coatings. Polymers 2021, 13 (21), 3698 DOI: 10.3390/polym13213698There is no corresponding record for this reference.
- 106Chhipa, S. M.; Sharma, S.; Bagha, A. K. Recent Development In Polymer Coating To Prevent Corrosion In Metals: A Review Mater. Today Proc. 2024 DOI: 10.1016/j.matpr.2024.09.001 .There is no corresponding record for this reference.
- 107Paraskar, P. M.; Prabhudesai, M.; Kulkarni, R. D. Synthesis And Characterizations Of Air-Cured Polyurethane Coatings From Vegetable Oils And Itaconic Acid. React. Funct. Polym. 2020, 156, 104734 DOI: 10.1016/j.reactfunctpolym.2020.104734There is no corresponding record for this reference.
- 108Dong, X.; Ren, J.; Duan, Y.; Wu, D.; Lin, L.; Jichao, S.; Jia, R.; Xu, X.; He, X.; Dong, X.; Ren, J.; Duan, Y.; Wu, D.; Lin, L.; Jichao, S.; Jia, R.; Xu, X.; He, X. Preparation And Properties Of Green UV-Curable Itaconic Acid Cross-Linked Modified Waterborne Polyurethane Coating. J. Appl. Polym. Sci. 2021, 139 (17), 52042 DOI: 10.1002/app.52042There is no corresponding record for this reference.
- 109Huang, J.; Zhang, J.; Zhu, G.; Yu, X.; Hu, Y.; Shang, Q.; Chen, J.; Hu, L.; Zhou, Y.; Liu, C. Self-Healing, High-Performance, And High-Biobased-Content Uv-Curable Coatings Derived From Rubber Seed Oil And Itaconic Acid. Prog. Org. Coat. 2021, 159, 106391 DOI: 10.1016/j.porgcoat.2021.106391There is no corresponding record for this reference.
- 110Mehta, L. B.; Wadgaonkar, K. K.; Jagtap, R. N. Synthesis And Characterization Of High Bio-Based Content Unsaturated Polyester Resin For Wood Coating From Itaconic Acid: Effect Of Various Reactive Diluents As An Alternative To Styrene. J. Dispersion Sci. Technol. 2019, 40 (5), 756– 765, DOI: 10.1080/01932691.2018.1480964There is no corresponding record for this reference.
- 111Yang, S.; He, Y.; Zhang, J. High-Performance Acrylate-Free Co-Cured coatings Enabled By Tailoring Itaconic Acid-Modified Dual-Phase Photocurable Resins. Prog. Org. Coat. 2024, 189, 108293 DOI: 10.1016/j.porgcoat.2024.108293There is no corresponding record for this reference.
- 112Zhang, Y.; Zhuo, G.; Huang, Y.; Qin, M. J.; Liu, M.; Li, L.; Guo, C. Synthesis Of Bio-Based Epoxy Resins Derived From Itaconic Acid And Application In Rubber Wood Surface Coating. Ind. Crops Prod. 2024, 222, 119529 DOI: 10.1016/j.indcrop.2024.119529There is no corresponding record for this reference.
- 113Brännström, S.; Malmström, E.; Johansson, M. Biobased Uv-Curable Coatings Based On Itaconic Acid. J. Coat. Technol. Res. 2017, 14 (4), 851– 861, DOI: 10.1007/s11998-017-9949-yThere is no corresponding record for this reference.
- 114Patil, D. M.; Phalak, G. A.; Mhaske, S. T. Design And Synthesis Of Bio-Based Uv Curable Pu Acrylate Resin From Itaconic Acid For Coating Applications. Des. Monomers Polym. 2017, 20 (1), 269– 282, DOI: 10.1080/15685551.2016.1231045There is no corresponding record for this reference.
- 115Dai, J.; Liu, X.; Ma, S.; Wang, J.; Shen, X.; You, S.; Zhu, J. Soybean Oil-Based Uv-Curable Coatings Strengthened By Crosslink Agent Derived From Itaconic Acid Together With 2-Hydroxyethyl Methacrylate Phosphate. Prog. Org. Coat. 2016, 97, 210– 215, DOI: 10.1016/j.porgcoat.2016.04.014There is no corresponding record for this reference.
- 116Dixit, A.; Sabnis, A.; Balgude, D.; Kale, S.; Gada, A. H.; Kudu, B.; Mehta, K.; Kasar, S.; Handa, D.; Mehta, R.; Kshirsagar, S.; Singh, A.; Dalvi, R.; Mishra, S. Synthesis And Characterization Of Citric Acid And Itaconic Acid-Based Two-Pack Polyurethane Antimicrobial Coatings. Polym. Bull. 2023, 80 (2), 2187– 2216, DOI: 10.1007/s00289-022-04638-9There is no corresponding record for this reference.
- 117Chen, B.; Wang, X.; Xu, H.; Zhang, Y.; Wang, C. Synthesis Of Itaconic Acid-Based Non-Isocyanate Polyurethane Coating With Excellent Heat Insulation And Corrosion Resistance. Arabian J. Chem. 2025, 18, 4982025 DOI: 10.25259/AJC_498_2025There is no corresponding record for this reference.
- 118Schneider-Chaabane, A.; Bleicher, V.; Rau, S.; Al-Ahmad, A.; Lienkamp, K. Stimulus-Responsive Polyzwitterionic Surfaces Made From Itaconic Acid: Self-Triggered Antimicrobial Activity, Protein Repellency, And Cell Compatibility. ACS Appl. Mater. Interfaces 2020, 12 (19), 21242– 21253, DOI: 10.1021/acsami.9b17781There is no corresponding record for this reference.
- 119Jamaludin, F. H.; Mohd Yusoff, N. I. S.; Chida, H.; Yang, X.; Wong, T.-W.; Zhou, H.; Li, T.; Wang, L.; Goh, P. S.; Ismail, A. F. Photo-Triggered Sustainable Adhesive Based On Itaconic Acid. ACS Sustainable Chem. Eng. 2022, 10 (19), 6389– 6401, DOI: 10.1021/acssuschemeng.2c01119There is no corresponding record for this reference.
- 120Stange, P.; Verevkin, S. P.; Ludwig, R. Combined Spectroscopic, Thermodynamic, And Theoretical Approach For Detecting And Quantifying Hydrogen Bonding And Dispersion Interaction In Ionic Liquids. Acc. Chem. Res. 2023, 56 (23), 3441– 3450, DOI: 10.1021/acs.accounts.3c00530There is no corresponding record for this reference.
- 121Li, W.; Wang, Y.; Ni, Z.; Li, S. Cluster-In-Molecule Local Correlation Method For Dispersion Interactions In Large Systems And Periodic Systems. Acc. Chem. Res. 2023, 56 (23), 3462– 3474, DOI: 10.1021/acs.accounts.3c00538There is no corresponding record for this reference.
- 122Becker, N.; Noyes, K.; Cooper, M. M. Characterizing Students’ Mechanistic Reasoning About London Dispersion Forces. J. Chem. Educ. 2016, 93 (10), 1713– 1724, DOI: 10.1021/acs.jchemed.6b00298There is no corresponding record for this reference.
- 123Law, K.-Y. Contact Angle Hysteresis On Smooth/Flat And Rough Surfaces. Interpretation, Mechanism, And Origin. Acc. Mater. Res. 2022, 3 (1), 1– 7, DOI: 10.1021/accountsmr.1c00051There is no corresponding record for this reference.
- 124Bovone, G.; Dudaryeva, O. Y.; Marco-Dufort, B.; Tibbitt, M. W. Engineering Hydrogel Adhesion For Biomedical Applications Via Chemical Design Of The Junction. ACS Biomater. Sci. Eng. 2021, 7 (9), 4048– 4076, DOI: 10.1021/acsbiomaterials.0c01677There is no corresponding record for this reference.
- 125Liu, M.; Li, J.; Hou, Y.; Guo, Z. Inorganic Adhesives For Robust Superwetting Surfaces. ACS Nano 2017, 11 (1), 1113– 1119, DOI: 10.1021/acsnano.6b08348There is no corresponding record for this reference.
- 126Mehmood, T.; Kaynak, A.; Dai, X. J.; Kouzani, A. Z.; Magniez, K.; Rubin, D.; Hurren, C.; du Plessis, J. Study Of Oxygen Plasma Pre-Treatment Of Polyester Fabric For Improved Polypyrrole Adhesion. Mater. Chem. Phys. 2014, 143 (2), 668– 675, DOI: 10.1016/j.matchemphys.2013.09.052There is no corresponding record for this reference.
- 127Lallemang, M.; Yu, L.; Cai, W.; Rischka, K.; Hartwig, A.; Haag, R.; Hugel, T.; Balzer, B. N. Multivalent Non-Covalent Interactions Lead To Strongest Polymer Adhesion. Nanoscale 2022, 14 (10), 3768– 3776, DOI: 10.1039/D1NR08338DThere is no corresponding record for this reference.
- 128Zhan, J.; Lei, Z.; Zhang, Y. Non-Covalent Interactions Of Graphene Surface: Mechanisms And Applications. Chem 2022, 8 (4), 947– 979, DOI: 10.1016/j.chempr.2021.12.015There is no corresponding record for this reference.
- 129Besford, Q. A.; Van den Heuvel, W.; Christofferson, A. J. Dipolar Dispersion Forces In Water–Methanol Mixtures: Enhancement Of Water Interactions Upon Dilution Drives Self-Association. J. Phys. Chem. B 2022, 126 (33), 6231– 6239, DOI: 10.1021/acs.jpcb.2c04638There is no corresponding record for this reference.
- 130Truhlar, D. G. Dispersion Forces: Neither Fluctuating Nor Dispersing. J. Chem. Educ. 2019, 96 (8), 1671– 1675, DOI: 10.1021/acs.jchemed.8b01044There is no corresponding record for this reference.
- 131Echeverrigaray, F. G.; De Mello, S. S.; Leidens, L. M.; da Costa, M. E. H. M.; Alvarez, F.; Burgo, T. A. L.; Michels, A. F.; Figueroa, C. A. Towards Superlubricity In Nanostructured Surfaces: The Role Of Van Der Waals Forces. Phys. Chem. Chem. Phys. 2018, 20 (34), 21949– 21959, DOI: 10.1039/c8cp02508hThere is no corresponding record for this reference.
- 132Liang, H.-J.; Qian, W.-Y.; Liu, H.-H.; Wang, X.; Gu, Z.-Y.; Dong, F.; Deng, Y.; Tang, Y.; Zhang, J.; Zhao, J.; Wu, X.-L. Sulfite-Based Electrolyte Chemistry With Ion–Dipole Interactions And Robust Interphase Achieves Wide-Temperature Sodium-Ion Batteries. J. Am. Chem. Soc. 2025, 147 (21), 17860– 17870, DOI: 10.1021/jacs.5c01864There is no corresponding record for this reference.
- 133Herschlag, D.; Pinney, M. M. Hydrogen Bonds: Simple After All?. Biochemistry 2018, 57 (24), 3338– 3352, DOI: 10.1021/acs.biochem.8b00217There is no corresponding record for this reference.
- 134van der Lubbe, S. C. C.; Guerra, C. F. The Nature Of Hydrogen Bonds: A Delineation Of The Role Of Different Energy Components On Hydrogen Bond Strengths And Lengths. Chem. - Asian J. 2019, 14 (16), 2760– 2769, DOI: 10.1002/asia.201900717There is no corresponding record for this reference.
- 135Schneider, H.-J. Distinction And Quantification Of Noncovalent Dispersive And Hydrophobic Effects. Molecules 2024, 29 (7), 1591 DOI: 10.3390/molecules29071591There is no corresponding record for this reference.
- 136Mousavi, M.; Fini, E. H. Non-Covalent Π-Stacking Interactions Between Asphaltene And Porphyrin In Bitumen. J. Chem. Inf. Model. 2020, 60 (10), 4856– 4866, DOI: 10.1021/acs.jcim.0c00507There is no corresponding record for this reference.
- 137Beber, V. C.; Wolter, N.; Schneider, B.; Koschek, K. Effect Of Aluminium Substrate Thickness On The Lap-Shear Strength Of Adhesively Bonded And Hybrid Riveted-Bonded Joints. Proc. Inst. Mech. Eng., Part E 2023, 237 (3), 607– 614, DOI: 10.1177/0954408920913434There is no corresponding record for this reference.
- 138Li, J.; Hu, C.; Yang, B.; Zhao, N.; Zeng, Y. Recyclable, Self-Healing Itaconic Acid-Based Polyurethane Networks With Dynamic Boronic Ester Bonds For Recoverable Adhesion Application. Polymer 2022, 256, 125227 DOI: 10.1016/j.polymer.2022.125227There is no corresponding record for this reference.
- 139Kim, H.-C.; Kwon, Y.-R.; Kim, J. S.; So, J.-H.; Kim, D. H. Dual-Cure Adhesives Using A Newly Synthesized Itaconic Acid-Based Epoxy Acrylate Oligomer. Polymers 2023, 15 (15), 3304 DOI: 10.3390/polym15153304There is no corresponding record for this reference.
- 140Mapari, S.; Mestry, S.; Mhaske, S. T. Developments In Pressure-Sensitive Adhesives: A Review. Polym. Bull. 2021, 78 (7), 4075– 4108, DOI: 10.1007/s00289-020-03305-1There is no corresponding record for this reference.
- 141Ju, Y. H.; Lee, H.; Han, C. J.; Lee, C.-R.; Kim, Y.; Kim, J.-W. Pressure-Sensitive Adhesive With Controllable Adhesion For Fabrication Of Ultrathin Soft Devices. ACS Appl. Mater. Interfaces 2020, 12 (36), 40794– 40801, DOI: 10.1021/acsami.0c11986There is no corresponding record for this reference.
- 142Antosik, A. K.; Mozelewska, K.; Piątek-Hnat, M.; Czech, Z.; Bartkowiak, M. Silicone Pressure-Sensitive Adhesives With Increased Thermal Resistance. J. Therm. Anal. Calorim. 2022, 147 (14), 7719– 7727, DOI: 10.1007/s10973-021-11048-yThere is no corresponding record for this reference.
- 143Fang, C.; Yan, Q.; Liu, Z.; Lu, Y.; Lin, Z. The Influence Of Monobutyl Itaconate And B-Carboxyethyl Acrylate On Acrylic Latex Pressure Sensitive Adhesives. Int. J. Adhes. Adhes. 2018, 84, 387– 393, DOI: 10.1016/j.ijadhadh.2018.05.007There is no corresponding record for this reference.
- 144Casas-Soto, C. R.; Conejo-Dávila, A. S.; Osuna, V.; Chávez-Flores, D.; Espinoza-Hicks, J. C.; Flores-Gallardo, S. G.; Vega-Ríos, A. Dibutyl Itaconate And Lauryl Methacrylate Copolymers By Emulsion Polymerization For Development Of Sustainable Pressure-Sensitive Adhesives. Polymers 2022, 14 (3), 632 DOI: 10.3390/polym14030632There is no corresponding record for this reference.
- 145Xue, W.; Zhang, X.; Sun, C.; Wei, C.; Zhang, L.; Yang, H.; Wang, R. Biobased Itaconate Acrylic Pressure-Sensitive Adhesives Via Emulsion Polymerization: Sustainable Design For High Peel Resistance. ACS Omega 2025, 10 (37), 42494– 42506, DOI: 10.1021/acsomega.5c03586There is no corresponding record for this reference.
- 146Zeng, X.; Liu, C.; Wang, X.; Cao, Y.; He, P.; Li, H.; Wang, L. Versatile Underwater Pressure Sensitive Adhesive: Uv Curing Synthesis And Substrate-Independent Adhesion. ACS Appl. Mater. Interfaces 2024, 16 (31), 41461– 41474, DOI: 10.1021/acsami.4c06163There is no corresponding record for this reference.
- 147Wang, X.; Cao, Y.; Zhang, J.; Yu, H. Biodegradable Polyurethane Pressure-Sensitive Adhesives Using Biobased Polyols With Distinct Intrinsic Properties. ACS Appl. Polym. Mater. 2024, 6 (9), 5121– 5128, DOI: 10.1021/acsapm.4c00157There is no corresponding record for this reference.
- 148Jin, J.; Cheng, L.; Chen, C.; Li, Z.; Hong, Y.; Li, C.; Ban, X.; Gu, Z. Synthesis, Characterization, And Application Of Starch-Based Adhesives Modified With Itaconic Acid And N-Hydroxyethyl Acrylamide. Ind. Crops Prod. 2023, 196, 116524, DOI: 10.1016/j.indcrop.2023.116524There is no corresponding record for this reference.
- 149Sharif, R.; Mohsin, M.; Sardar, S.; Ramzan, N.; Raza, Z. A. Synthesis Of Nontoxic And Bio Based Oil And Water Repellent Polymers For Cotton Fabrics Using Stearic Acid, Succinic Acid, And Itaconic Acid. J. Nat. Fibers 2022, 19 (16), 12473– 12485, DOI: 10.1080/15440478.2022.2072995There is no corresponding record for this reference.
- 150Boondaeng, A.; Suwanruji, P.; Vaithanomsat, P.; Apiwatanapiwat, W.; Trakunjae, C.; Janchai, P.; Apipatpapha, T.; Chanka, N.; Chollakup, R. Bio-Synthesis Of Itaconic Acid As An Anti-Crease Finish For Cellulosic Fiber Fabric. RSC Adv. 2021, 11 (42), 25943– 25950, DOI: 10.1039/D1RA05037KThere is no corresponding record for this reference.
- 151Peng, H.; Yang, C. Q.; Wang, X.; Wang, S. The Combination Of Itaconic Acid And Sodium Hypophosphite As A New Cross-Linking System For Cotton. Ind. Eng. Chem. Res. 2012, 51 (35), 11301– 11311, DOI: 10.1021/ie3005644There is no corresponding record for this reference.
- 152Sakthivel, M.; Franklin, D. S.; Sudarsan, S.; Chitra, G.; Sridharan, T. B.; Guhanathan, S. Investigation On Ph/Salt-Responsive Multifunctional Itaconic Acid Based Polymeric Biocompatible, Antimicrobial And Biodegradable Hydrogels. React. Funct. Polym. 2018, 122, 9– 21, DOI: 10.1016/j.reactfunctpolym.2017.10.021There is no corresponding record for this reference.
- 153Ma, J.; Li, T.; Luo, M.; Lei, B. Single-Component Self-Healing Antibacterial Anti-Inflammatory Intracellular-Antioxidative Poly(Itaconic Acid-Pluronic) Hydrogel For Rapid Repair Of Mrsa-Impaired Wound. ACS Appl. Mater. Interfaces 2023, 15 (28), 33413– 33424, DOI: 10.1021/acsami.3c05383There is no corresponding record for this reference.
- 154Zhou, S.; He, J.; Liu, Q.; Chen, T.; Guan, X.; Gao, H.; Jiang, J.; Wang, J.; Peng, X.; Wu, J. Injectable Hydrogel Of Chitosan-Octyl Itaconate Conjugate Modulates Inflammatory Response. ACS Biomater. Sci. Eng. 2024, 10 (8), 4823– 4838, DOI: 10.1021/acsbiomaterials.4c00882There is no corresponding record for this reference.
- 155Naderi, P.; Kabiri, K.; Jahanmardi, R.; Zohuriaan-Mehr, M. J. Preparation Of Itaconic Acid Bio-Based Cross-Linkers For Hydrogels. J. Macromol. Sci., Part A 2021, 58 (3), 165– 174, DOI: 10.1080/10601325.2020.1836492There is no corresponding record for this reference.
- 156Das, A.; Saha, M.; Gupta, M. K.; Rangan, L.; Uppaluri, R.; Das, C. Comparative Efficacy Of Citric Acid/Tartaric Acid/Malic Acid Additive-Based Polyvinyl Alcohol-Starch Composite Films. J. Mater. Sci.: Mater. Eng. 2024, 19 (1), 9 DOI: 10.1186/s40712-024-00151-1There is no corresponding record for this reference.
- 157Li, J.; Lei, H.; Xi, X.; Li, C.; Hou, D.; Song, J.; Du, G. A Sustainable Tannin-Citric Acid Wood Adhesive With Favorable Bonding Properties And Water Resistance. Ind. Crops Prod. 2023, 201, 116933 DOI: 10.1016/j.indcrop.2023.116933There is no corresponding record for this reference.
- 158Liu, M.; Yao, W.; Shan, J.; Zheng, H.; Yang, Y.; Cao, L.; Qi, B.; Tan, H.; Sun, C.; Zhang, X.; Zhang, Y. Preparation Of High-Performance Antibacterial/Antifungal Citric Acid–Starch Adhesives Based On Physical Entanglement And Chemical Crosslinking. Int. J. Biol. Macromol. 2024, 279, 135560 DOI: 10.1016/j.ijbiomac.2024.135560There is no corresponding record for this reference.
- 159Liu, X.; Allais, F.; Fadlallah, S. Itaconic Acid As A Platform Chemical For Bio-Based Polymers: From Green Polymerization Strategies To Structure-Driven Applications. Green Chem. 2026, 28 (8), 3371– 3393, DOI: 10.1039/D5GC06410DThere is no corresponding record for this reference.
- 160Satoh, K.; Lee, D.-H.; Nagai, K.; Kamigaito, M. Precision Synthesis Of Bio-Based Acrylic Thermoplastic Elastomer By Raft Polymerization Of Itaconic Acid Derivatives. Macromol. Rapid Commun. 2014, 35 (2), 161– 167, DOI: 10.1002/marc.201300638There is no corresponding record for this reference.
- 161Pradeep, S. V.; Kandasubramanian, B.; Sidharth, S. A Review On Recent Trends In Bio-Based Pressure Sensitive Adhesives. J. Adhes. 2023, 99 (14), 2145– 2166, DOI: 10.1080/00218464.2023.2176761There is no corresponding record for this reference.
- 162Li, B.; Torres, G. B.; Martin, B.; Taylor, N.; Barbu, E.; Christie, A.; Heise, A. Polycaprolactone–Itaconic Acid Resins For Additive Manufacturing Of Environmentally Degradable 3D And 4D Materials By Thiol-Ene Photopolymerization. Macromolecules 2025, 58 (16), 8887– 8897, DOI: 10.1021/acs.macromol.5c01310There is no corresponding record for this reference.
- 163Maturi, M.; Spanu, C.; Maccaferri, E.; Locatelli, E.; Benelli, T.; Mazzocchetti, L.; Sambri, L.; Giorgini, L.; Franchini, M. C. (Meth)Acrylate-Free Three-Dimensional Printing Of Bio-Derived Photocurable Resins With Terpene- And Itaconic Acid-Derived Poly(Ester-Thioether)S. ACS Sustainable Chem. Eng. 2023, 11 (49), 17285– 17298, DOI: 10.1021/acssuschemeng.3c04576There is no corresponding record for this reference.
- 164Zhang, Y.; Lin, Z.; Li, X.; Li, G. One-Step And Scalable Synthesis Of Eaa-Based Reprocessable Vitrimer With Superior Mechanical Properties. J. Polym. Environ. 2024, 32 (3), 1080– 1089, DOI: 10.1007/s10924-023-03030-6There is no corresponding record for this reference.
- 165Madduluri, V. R.; Bendi, A.; Chinmay; Maniam, G. P.; Roslan, R.; Ab Rahim, M. H. Recent Advances In Vitrimers: A Detailed Study On The Synthesis, Properties And Applications Of Bio-Vitrimers. J. Polym. Environ. 2025, 33 (1), 301– 322, DOI: 10.1007/s10924-024-03416-0There is no corresponding record for this reference.
- 166Zhang, J.; Gong, Z.; Wu, C.; Li, T.; Tang, Y.; Wu, J.; Jiang, C.; Miao, M.; Zhang, D. Itaconic Acid-Based Hyperbranched Polymer Toughened Epoxy Resins With Rapid Stress Relaxation, Superb Solvent Resistance And Closed-Loop Recyclability. Green Chem. 2022, 24 (18), 6900– 6911, DOI: 10.1039/D2GC01541BThere is no corresponding record for this reference.
- 167Qi, Y.; Zhang, Y.; Ou, S.; Zhuo, G.; Zeng, H.; Lao, Y.; Wang, J.; Wang, Q.; Guo, C. Green And Sustainable Itaconic Acid-Based Vitrimers With Rapid Stress Relaxation, Superior Fire Safety, And Recyclability Ivia/I Synergistic Roles Of Multiple Dynamic Bonds. Green Chem. 2025, 27 (42), 13385– 13403, DOI: 10.1039/D5GC04554AThere is no corresponding record for this reference.
- 168Kong, F.; Ma, X.; Xu, X.; Cui, M.; Zhao, H.; Zhu, J.; Chen, J. Itaconic Acid-Based Sustainable Polyurethane Covalent Adaptable Networks With Robust Mechanical, Reshaping And Uv-Resistant Properties Based On Reversible Disulfide Bond. Mater. Today Chem. 2024, 35, 101881 DOI: 10.1016/j.mtchem.2023.101881There is no corresponding record for this reference.
- 169Nouigues, A.; Le Gal La Salle, E.; Bailleul, J.-L. Thermo-Mechanical Characterization Of Unsaturated Polyester/Glass Fiber Composites For Recycling. Int. J. Mater. Form. 2021, 14 (1), 153– 174, DOI: 10.1007/s12289-020-01559-8There is no corresponding record for this reference.
- 170Arnaud, S. P.; Andreou, E.; Köster, L. V. G. P.; Robert, T. Selective Synthesis Of Monoesters Of Itaconic Acid With Broad Substrate Scope: Biobased Alternatives To Acrylic Acid?. ACS Sustainable Chem. Eng. 2020, 8 (3), 1583– 1590, DOI: 10.1021/acssuschemeng.9b06330There is no corresponding record for this reference.
- 171Shao, W.; Wang, P.; Liu, J.; Xu, H.; Cai, X.; Wu, Q.; Xia, N.; Kong, F. An Environmentally Friendly And High Wet-Bond Strength Adhesive Based On Starch, Itaconic Acid And Silicone. Industrial Crops and Products 2022, 181, 114809, DOI: 10.1016/j.indcrop.2022.114809There is no corresponding record for this reference.



