Flame-Synthesized Zinc Oxide Tetrapods for Photoprotection in Sunscreen FormulationsClick to copy article linkArticle link copied!
- Ajoa J. Addae*Ajoa J. Addae*E-mail: [email protected]Department of Chemistry and Biochemistry, University of California Los Angeles, Los Angeles, California 90095, United StatesCalifornia NanoSystems Institute, University of California Los Angeles, Los Angeles, California 90095, United StatesMore by Ajoa J. Addae
- Jennifer UyangaJennifer UyangaDepartment of Chemistry and Biochemistry, University of California Los Angeles, Los Angeles, California 90095, United StatesMore by Jennifer Uyanga
- Yogendra Kumar MishraYogendra Kumar MishraSmart Materials, NanoSYD, Mads Clausen Institute, University of Southern Denmark, Alsion 2, 6400 Sønderborg, DenmarkMore by Yogendra Kumar Mishra
- Justin CaramJustin CaramDepartment of Chemistry and Biochemistry, University of California Los Angeles, Los Angeles, California 90095, United StatesMore by Justin Caram
- Paul S. Weiss*Paul S. Weiss*[email protected]Department of Chemistry and Biochemistry, University of California Los Angeles, Los Angeles, California 90095, United StatesCalifornia NanoSystems Institute, University of California Los Angeles, Los Angeles, California 90095, United StatesDepartment of Bioengineering, University of California, Los Angeles, Los Angeles, California 90095, United StatesDepartment of Materials Science and Engineering, University of California Los Angeles, Los Angeles, California 90095, United StatesMore by Paul S. Weiss
Abstract
ZnO nanoparticles are widely used in sunscreen formulations due to their strong light absorption in the ultraviolet (UV) and ease of synthesis. Here, we explore flame-synthesized tetrapod-shaped zinc oxide (ZnO) nanostructured particles (NPs) as UV-absorbing materials for sunscreen formulations. We compare their optical properties to conventional sol–gel-synthesized ZnO NPs. We then tested sunscreen formulations of each and measured and compared their optical, colloidal, and cosmetic properties. Notably, the test sunscreen containing mass-equalized tetrapodal ZnO absorbs in the UVB region and has a sun protection factor (SPF) value comparable to that of sol–gel-synthesized ZnO. Furthermore, colloidal and white cast evaluations of flame-synthesized tetrapodal ZnO demonstrated favorable properties for the storage and usage of sunscreen formulations.
This publication is licensed for personal use by The American Chemical Society.
| ZnO Type | Synthetic Summary | Properties |
|---|---|---|
| Sol–gel-synthesized ZnO (SgZnO) particles | Zinc precursor (Zn(CH3CO2)2·2H2O) vortexed in water, pH adjusted to 12 with NaOH, vacuum-dried at 50 °C for 12 h | Platelet-shaped ZnO particles, with aggregates ranging in size from 20 to 200 nm |
| Flame-synthesized ZnO tetrapods (ZOTeN) particles | Zinc precursor (Zn2+) and ethanol heated to 900 °C and vapor flame | Tetrapod-shaped ZnO particle morphologies with arm lengths spanning 10–100 μm |
| French-process ZnO (FpZnO) particles | Zinc precursor (Zn2+) vaporized at ∼1000 °C, oxidized in oxidation chamber to yield polycrystalline ZnO powder | Platelet-shaped ZnO particles ranging in size from 0.39 to 1.81 μm |
Figure 1
Figure 1. (a) Schematic of anisotropic growth of ZnO within the wurtzite host lattice structure. (b) Transmission electron microscopy image of sol–gel-synthesized ZnO (SgZnO) particles; scale bar 50 μm. (c) Transmission electron microscopy image of flame-synthesized ZnO (ZOTeN) particles; scale bar 2 μm. (d) Transmission electron microscopy image of FpZnO particles; scale bar 0.5 μm. (e) Scanning electron microscopy image of 0.1 g of SgZnO particles; scale bar 50 μm. (f) Scanning electron microscopy image of 0.1 g of FpZnO particles; scale bar 50 μm. (g) Scanning electron microscopy image of 0.1 g of ZOTeN particles; scale bar 50 μm. Absorption spectra of 0.1 g/mL of flame-synthesized ZnO, French-process-synthesized ZnO, and sol–gel-synthesized ZnO in the ultraviolet and visible regions.
Characterization of ZnO Particles
Formulation of Sunscreen Dispersions and Test Emulsions
Figure 2
Figure 2. Schematic of preparing flame-synthesized ZnO (ZOTeN) particle dispersions for incorporation into a sunscreen formulation.
Sunscreen Formulation Characterization
Figure 3
Figure 3. (a) Light microscope image of a 1:50 dilution of test formulation without ZnO in water, with scale bars of 200 μm. (b) Light microscope image of a 1:50 dilution of test formulation with sol–gel-synthesized ZnO (SgZnO) particles in water, with scale bars of 200 μm. (c) Light micrograph of a 1:50 dilution of test formulation with flame-synthesized ZnO (ZOTeN) particles in water, with scale bars of 200 μm. (d) Absorbance of ZOTeN particle sunscreen test formulation, SgZnO particle sunscreen test formulation, and FpZnO particle sunscreen test formulation.
Figure 4
Figure 4. (a) Schematic of oil-in-water emulsions containing sol–gel-synthesized ZnO and (b) flame-synthesized ZnO. (c) Viscosity measurements of flame-synthesized ZnO sunscreen and sol–gel-synthesized ZnO sunscreen over 12 weeks at 25 °C.
Colorimetric Evaluation of Sunscreen Formulations
Figure 5
Figure 5. (a) Image of flame-synthesized (ZOTeN) particle-based sunscreen and sol–gel-synthesized (SgZnO) particle-based sunscreen application on hands categorized as (left) Fitzpatrick Skin Type 6 (darker tone) and image of ZOTeN particles and SgZnO particles application on hands categorized as (right) Fitzpatrick Skin Type 2 (lighter tone). (b) Lightness versus saturation of 20% w/w SgZnO particle-based sunscreen, 20% w/w ZOTeN particle-based sunscreen, 20% w/w FpZnO particle-based sunscreen, and 20% w/w ZnO commercial sunscreen on black and white backgrounds. (c) Lightness versus hue of 20% w/w sol–gel-synthesized ZnO (SgZnO) particle sunscreen, 20% w/w flame-synthesized (ZOTeN) particle sunscreen, 20% w/w FpZnO particle-based sunscreen, and 20% w/w ZnO commercial sunscreen particle sunscreen on black and white backgrounds. (d) Light microscope image of 1 g of 20% w/w SgZnO particle sunscreen with scale bars of 100 μm with UV/visible spectrum-simulated color swatch superimposed in the bottom right corner (left) and 1 g of 20% w/w ZOTeN particle sunscreen with scale bars of 100 μm with UV/Visible spectrum-simulated color swatch superimposed in the bottom right corner (right). (e) Diagram depicting contrast of sunscreen white cast on absolute black and white backgrounds.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsmaterialslett.5c01351.
X-ray diffraction data of ZnO particles and SEM and calculated values of saturation and hue. Methods of synthesis and Python processing of colors. Centrifugal aging data of sunscreens. (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
The authors thank the Challenge Initiative at UCLA and Sigma Xi IFoRE Grant-in-Aid for support. A.J.A. thanks the National Science Foundation for graduate fellowship support (Grant DGE-2034835 and DGE-2444110). Y.K.M. acknowledges NANOCHEM (national infrastructure UFM 5229-00010B, NANOCHEM, Denmark) and Fabrikant Mads Clausen Fonden Denmark for a Tetrapod Upscaling Grant. We thank the California NanoSystems Institute Core Voucher program for supporting measurements. The authors thank Antonio Macias, Jr. for assistance with X-ray diffraction, Alexandra Wright for images of her hands, Daniel Bilezikian for assistance with SEM, Elijah Cook for assistance with TEM, Bradley Kroes for assistance with ball milling, and Dr. Krzysztof Konieczny and Prof. Thomas Mason for helpful discussions.
References
This article references 48 other publications.
- 1Taylor, J. S. Unraveling the Molecular Pathway from Sunlight to Skin Cancer. Acc. Chem. Res. 1994, 27, 76– 82, DOI: 10.1021/ar00039a003Google ScholarThere is no corresponding record for this reference.
- 2Rastogi, R. P.; Richa; Kumar, A.; Tyagi, M. B.; Sinha, R. P. Molecular Mechanisms of Ultraviolet Radiation-Induced DNA Damage and Repair. J. Nucleic Acids 2010, 2010, 592980, DOI: 10.4061/2010/592980Google ScholarThere is no corresponding record for this reference.
- 3Neale, R.; Williams, G.; Green, A. Application Patterns among Participants Randomized to Daily Sunscreen Use in a Skin Cancer Prevention Trial. Arch. Dermatol. 2002, 138, 1319– 1325, DOI: 10.1001/archderm.138.10.1319Google ScholarThere is no corresponding record for this reference.
- 4Addae, A. J.; Weiss, P. S. Standardizing the White Cast Potential of Sunscreens with Metal Oxide Ultraviolet Filters. Acc. Mater. Res. 2024, 5, 392– 399, DOI: 10.1021/accountsmr.4c00004Google ScholarThere is no corresponding record for this reference.
- 5Addae, A. J.; Uyanga, J.; Chifamba, J.; Weiss, P. S. Advances in Enhancing Photoprotection of Sunscreens Using Hydrocarbon Film Formers and Carbon Nanomaterials. Adv. Drug Delivery Rev. 2025, 222, 115607, DOI: 10.1016/j.addr.2025.115607Google ScholarThere is no corresponding record for this reference.
- 6Xu, S.; Kwa, M.; Agarwal, A.; Rademaker, A.; Kundu, R. V. Sunscreen Product Performance and Other Determinants of Consumer Preferences. JAMA Dermatol. 2016, 152, 920– 927, DOI: 10.1001/jamadermatol.2016.2344Google ScholarThere is no corresponding record for this reference.
- 7Herzog, B.; Schultheiss, A.; Giesinger, J. On the Validity of Beer-Lambert Law and Its Significance for Sunscreens. Photochem. Photobiol. 2018, 94, 384– 389, DOI: 10.1111/php.12861Google ScholarThere is no corresponding record for this reference.
- 8Lowe, N. J.; Shaath, N. A.; Pathak, M. A. Sunscreens: Development: Evaluation, and Regulatory Aspects, 2nd ed.; Marcel Dekker, Inc., 1997.Google ScholarThere is no corresponding record for this reference.
- 9Khunkitti, W.; Satthanakul, P.; Waranuch, N.; Pitaksuteepong, T.; Kitikhun, P. Method for Screening Sunscreen Cream Formulations by Determination of in Vitro SPF and PA Values Using UV Transmission Spectroscopy and Texture Profile Analysis. J. Cosmet. Sci. 2014, 65, 147– 159Google ScholarThere is no corresponding record for this reference.
- 10Binks, B. P.; Brown, J.; Fletcher, P. D. I.; Johnson, A. J.; Marinopoulos, I.; Crowther, J. M.; Thompson, M. A. Evaporation of Sunscreen Films: How the UV Protection Properties Change. ACS Appl. Mater. Interfaces 2016, 8, 13270– 13281, DOI: 10.1021/acsami.6b02696Google ScholarThere is no corresponding record for this reference.
- 11Stiller, S.; Gers-Barlag, H.; Lergenmueller, M.; Pflücker, F.; Schulz, J.; Wittern, K. P.; Daniels, R. Investigation of the Stability in Emulsions Stabilized with Different Surface Modified Titanium Dioxides. Colloids Surf. Physicochem. Eng. Asp. 2004, 232, 261– 267, DOI: 10.1016/j.colsurfa.2003.11.003Google ScholarThere is no corresponding record for this reference.
- 12Yeon, J. Y.; Seo, J. M.; Kim, T. H.; Shim, J. G. Emulsion Stability of Low Viscosity W/O Emulsion and Application of Inorganic Sunscreen Agents. J. Korean Appl. Sci. Technol. 2018, 35, 985– 1001, DOI: 10.12925/jkocs.2018.35.3.985Google ScholarThere is no corresponding record for this reference.
- 13Chao, C.; Génot, C.; Rodriguez, C.; Magniez, H.; Lacourt, S.; Fievez, A.; Len, C.; Pezron, I.; Luart, D.; van Hecke, E. Emollients for Cosmetic Formulations: Towards Relationships between Physico-Chemical Properties and Sensory Perceptions. Colloids Surf. Physicochem. Eng. Asp. 2018, 536, 156– 164, DOI: 10.1016/j.colsurfa.2017.07.025Google ScholarThere is no corresponding record for this reference.
- 14Mishra, Y. K.; Adelung, R. ZnO Tetrapod Materials for Functional Applications. Mater. Today 2018, 21, 631– 651, DOI: 10.1016/j.mattod.2017.11.003Google ScholarThere is no corresponding record for this reference.
- 15Mishra, Y. K.; Kaps, S.; Schuchardt, A.; Paulowicz, I.; Jin, X.; Gedamu, D.; Freitag, S.; Claus, M.; Wille, S.; Kovalev, A.; Gorb, S. N.; Adelung, R. Fabrication of Macroscopically Flexible and Highly Porous 3D Semiconductor Networks from Interpenetrating Nanostructures by a Simple Flame Transport Approach. Part. Part. Syst. Charact. 2013, 30, 775– 783, DOI: 10.1002/ppsc.201300197Google ScholarThere is no corresponding record for this reference.
- 16Klingshirn, C. ZnO: Material, Physics and Applications. ChemPhysChem 2007, 8, 782– 803, DOI: 10.1002/cphc.200700002Google ScholarThere is no corresponding record for this reference.
- 17Lu, P. J.; Fang, S. W.; Cheng, W. L.; Huang, S. C.; Huang, M. C.; Cheng, H. F. Characterization of Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreen Powder by Comparing Different Measurement Methods. J. Food Drug Anal. 2018, 26, 1192– 1200, DOI: 10.1016/j.jfda.2018.01.010Google ScholarThere is no corresponding record for this reference.
- 18Saito, G. P.; Bizari, M.; Cebim, M. A.; Correa, M. A.; Jafelicci Junior, M.; Davolos, M. R. Study of the Colloidal Stability and Optical Properties of Sunscreen Creams. Eclet. Quim. J. 2019, 44, 26– 36, DOI: 10.26850/1678-4618eqj.v44.2.2019.p26-36Google ScholarThere is no corresponding record for this reference.
- 19Faure, B.; Salazar-Alvarez, G.; Ahniyaz, A.; Villaluenga, I.; Berriozabal, G.; De Miguel, Y. R.; Bergström, L. Dispersion and Surface Functionalization of Oxide Nanoparticles for Transparent Photocatalytic and UV-Protecting Coatings and Sunscreens. Sci. Technol. Adv. Mater. 2013, 14, 023001, DOI: 10.1088/1468-6996/14/2/023001Google ScholarThere is no corresponding record for this reference.
- 20Paiva, J. P.; Diniz, R. R.; Leitão, A. C.; Cabral, L. M.; Fortunato, R. S.; Santos, B. A. M. C.; de Pádula, M. Insights and Controversies on Sunscreen Safety. Crit. Rev. Toxicol. 2020, 50, 707– 723, DOI: 10.1080/10408444.2020.1826899Google ScholarThere is no corresponding record for this reference.
- 21Schilling, K.; Bradford, B.; Castelli, D.; Dufour, E.; Nash, J. F.; Pape, W.; Schulte, S.; Tooley, I.; van den Bosch, J.; Schellauf, F. Human Safety Review of “Nano” Titanium Dioxide and Zinc Oxide. Photochem. Photobiol. Sci. 2010, 9, 495– 509, DOI: 10.1039/b9pp00180hGoogle ScholarThere is no corresponding record for this reference.
- 22Aditya, A.; Chattopadhyay, S.; Gupta, N.; Alam, S.; Veedu, A. P.; Pal, M.; Singh, A.; Santhiya, D.; Ansari, K. M.; Ganguli, M. ZnO Nanoparticles Modified with an Amphipathic Peptide Show Improved Photoprotection in Skin. ACS Appl. Mater. Interfaces 2019, 11, 56– 72, DOI: 10.1021/acsami.8b08431Google ScholarThere is no corresponding record for this reference.
- 23Abadi, P. G.-S.; Shirazi, F. H.; Joshaghani, M.; Moghimi, H. R. Ag+-Promoted Zinc Oxide [Zn(O):Ag]: A Novel Structure for Safe Protection of Human Skin against UVA Radiation. Toxicol. In Vitro 2018, 50, 318– 327, DOI: 10.1016/j.tiv.2018.02.016Google ScholarThere is no corresponding record for this reference.
- 24Khanam, B. R.; Nidhi, M. K. J.; Nagaraja, H.; Manjunatha, T.; Angadi, B.; Uma Reddy, B.; Udaykumar, K. Physicochemical and Therapeutic Studies of Microwave-Synthesized ZnO and Cr-Doped ZnO Nanoparticles for Biomedical Applications. Inorg. Chem. Commun. 2024, 170, 113454, DOI: 10.1016/j.inoche.2024.113454Google ScholarThere is no corresponding record for this reference.
- 25Bouie, A.; Baki, G. Iron Oxides and Ultramarine Blue in Inorganic Sunscreens for Dark Skin. J. Cosmet. Sci. 2025, 76, 123– 139Google ScholarThere is no corresponding record for this reference.
- 26Dipak, Nipane; Thakare, S. R.; Khati, N. T. ZnO Nanoparticle by Sol-Gel and Its UV Application in Cosmetics Formulation. Int. J. Knowl. Eng. 2012, 3, 168– 169Google ScholarThere is no corresponding record for this reference.
- 27Al-luhaibi, A. A.; Sendi, R. K. Synthesis, Potential of Hydrogen Activity, Biological and Chemical Stability of Zinc Oxide Nanoparticle Preparation by Sol-Gel: A Review. J. Radiat. Res. Appl. Sci. 2022, 15, 238– 254, DOI: 10.1016/j.jrras.2022.07.008Google ScholarThere is no corresponding record for this reference.
- 28Hong, H. S. Current Status of Zinc Oxide Nanopowder Manufacturing Technology for UV Filters. Korean J. Mater. Res. 2025, 35, 167– 174, DOI: 10.3740/MRSK.2025.35.5.167Google ScholarThere is no corresponding record for this reference.
- 29Mishra, Y. K.; Modi, G.; Cretu, V.; Postica, V.; Lupan, O.; Reimer, T.; Paulowicz, I.; Hrkac, V.; Benecke, W.; Kienle, L.; Adelung, R. Direct Growth of Freestanding ZnO Tetrapod Networks for Multifunctional Applications in Photocatalysis, UV Photodetection, and Gas Sensing. ACS Appl. Mater. Interfaces 2015, 7, 14303– 14316, DOI: 10.1021/acsami.5b02816Google ScholarThere is no corresponding record for this reference.
- 30Modi, G. Zinc Oxide Tetrapod: A Morphology with Multifunctional Applications. Adv. Nat. Sci.: Nanosci. Nanotechnol. 2015, 6, 033002, DOI: 10.1088/2043-6262/6/3/033002Google ScholarThere is no corresponding record for this reference.
- 31Mueen, R.; Lerch, M.; Cheng, Z.; Konstantinov, K. Na-Doped ZnO UV Filters with Reduced Photocatalytic Activity for Sunscreen Applications. J. Mater. Sci. 2020, 55, 2772– 2786, DOI: 10.1007/s10853-019-04122-2Google ScholarThere is no corresponding record for this reference.
- 32Chegeni, M.; Pour, S. K.; Faraji Dizaji, B. Synthesis and Characterization of Novel Antibacterial Sol-Gel Derived TiO2/Zn2TiO4/Ag Nanocomposite as an Active Agent in Sunscreens. Ceram. Int. 2019, 45, 24413– 24418, DOI: 10.1016/j.ceramint.2019.08.163Google ScholarThere is no corresponding record for this reference.
- 33Hasnidawani, J. N.; Azlina, H. N.; Norita, H.; Bonnia, N. N.; Ratim, S.; Ali, E. S. Synthesis of ZnO Nanostructures Using Sol-Gel Method. Procedia Chem. 2016, 19, 211– 216, DOI: 10.1016/j.proche.2016.03.095Google ScholarThere is no corresponding record for this reference.
- 34Elbrolesy, A.; Elhussiny, F. A.; Abdou, Y.; Morsy, R. Facile Synthesis and Biophysical Characterization of Novel Zinc Oxide/Fe3O4 Hybrid Nanocomposite as a Potentially Active Agent in Sunscreens. Arab. J. Sci. Eng. 2024, 49, 1083– 1093, DOI: 10.1007/s13369-023-08082-3Google ScholarThere is no corresponding record for this reference.
- 35Aikens, P. Formulation of Sunscreens in the United States. In Handbook of Formulating Dermal Applications; John Wiley & Sons, Ltd, 2016; pp 589– 609.Google ScholarThere is no corresponding record for this reference.
- 36Egerton, T. A.; Tooley, I. R. UV Absorption and Scattering Properties of Inorganic-Based Sunscreens. Int. J. Cosmet. Sci. 2012, 34, 117– 122, DOI: 10.1111/j.1468-2494.2011.00689.xGoogle ScholarThere is no corresponding record for this reference.
- 37Arif, A.; Belahssen, O.; Gareh, S.; Benramache, S. The Calculation of Band Gap Energy in Zinc Oxide Films. J. Semicond. 2015, 36, 013001– 013006, DOI: 10.1088/1674-4926/36/1/013001Google ScholarThere is no corresponding record for this reference.
- 38Marín, E.; Calderón, A. Conversion of Wavelength and Energy Scales and the Analysis of Optical Emission Spectra. J. Phys. Chem. Lett. 2022, 13, 8376– 8379, DOI: 10.1021/acs.jpclett.2c02621Google ScholarThere is no corresponding record for this reference.
- 39Catalano, R.; Masion, A.; Ziarelli, F.; Slomberg, D.; Laisney, J.; Unrine, J. M.; Campos, A.; Labille, J. Optimizing the Dispersion of Nanoparticulate TiO2-Based UV Filters in a Non-Polar Medium Used in Sunscreen Formulations - The Roles of Surfactants and Particle Coatings. Colloids Surf. Physicochem. Eng. Asp. 2020, 599, 124792, DOI: 10.1016/j.colsurfa.2020.124792Google ScholarThere is no corresponding record for this reference.
- 40Labille, J.; Feng, J.; Botta, C.; Borschneck, D.; Sammut, M.; Cabie, M.; Auffan, M.; Rose, J.; Bottero, J.-Y. Aging of TiO2 Nanocomposites Used in Sunscreen. Dispersion and Fate of the Degradation Products in Aqueous Environment. Environ. Pollut. 2010, 158, 3482– 3489, DOI: 10.1016/j.envpol.2010.02.012Google ScholarThere is no corresponding record for this reference.
- 41Franco-Gil, M. E.; Graça, A.; Martins, A.; Marto, J.; Ribeiro, H. M. Emollients in Dermatological Creams: Early Evaluation for Tailoring Formulation and Therapeutic Performance. Int. J. Pharm. 2024, 653, 123825, DOI: 10.1016/j.ijpharm.2024.123825Google ScholarThere is no corresponding record for this reference.
- 42Sapcharoenkun, C.; Klamchuen, A.; Kasamechonchung, P.; Iemsam-arng, J. Role of Rheological Behavior of Sunscreens Containing Nanoparticles on Thin Film Preparation. Mater. Sci. Eng., B 2020, 259, 114608, DOI: 10.1016/j.mseb.2020.114608Google ScholarThere is no corresponding record for this reference.
- 43Calixto, L. S.; Maia Campos, P. M. B. G. Physical-Mechanical Characterization of Cosmetic Formulations and Correlation between Instrumental Measurements and Sensorial Properties. Int. J. Cosmet. Sci. 2017, 39, 527– 534, DOI: 10.1111/ics.12406Google ScholarThere is no corresponding record for this reference.
- 44Hedjar, L.; Toscani, M.; Gegenfurtner, K. R. Importance of Hue: The Effect of Saturation on Hue-Chroma Asymmetries. J. Opt. Soc. Am. A 2025, 42, B305– B312, DOI: 10.1364/JOSAA.544641Google ScholarThere is no corresponding record for this reference.
- 45Braun, G. J.; Fairchild, M. D.; Ebner, F. Color Gamut Mapping in a Hue-Linearized CIELAB Color Space. Color Imaging Conf. 1998, 6, 163– 168, DOI: 10.2352/CIC.1998.6.1.art00034Google ScholarThere is no corresponding record for this reference.
- 46Bao, W.; Wei, M.; Xiao, K. Investigating Unique Hues at Different Chroma Levels with a Smaller Hue Angle Step. J. Opt. Soc. Am. A 2020, 37, 671– 679, DOI: 10.1364/JOSAA.383002Google ScholarThere is no corresponding record for this reference.
- 47Wrolstad, R. E.; Smith, D. E. Color Analysis. In Food Analysis; Nielsen, S. S., Ed.; Springer US: Boston, MA, 2010; pp 573– 586.Google ScholarThere is no corresponding record for this reference.
- 48Kim, M. H.; Weyrich, T.; Kautz, J. Modeling Human Color Perception under Extended Luminance Levels. ACM Transactions on Graphics 2009, 28, 27, DOI: 10.1145/1531326.1531333Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1

Figure 1. (a) Schematic of anisotropic growth of ZnO within the wurtzite host lattice structure. (b) Transmission electron microscopy image of sol–gel-synthesized ZnO (SgZnO) particles; scale bar 50 μm. (c) Transmission electron microscopy image of flame-synthesized ZnO (ZOTeN) particles; scale bar 2 μm. (d) Transmission electron microscopy image of FpZnO particles; scale bar 0.5 μm. (e) Scanning electron microscopy image of 0.1 g of SgZnO particles; scale bar 50 μm. (f) Scanning electron microscopy image of 0.1 g of FpZnO particles; scale bar 50 μm. (g) Scanning electron microscopy image of 0.1 g of ZOTeN particles; scale bar 50 μm. Absorption spectra of 0.1 g/mL of flame-synthesized ZnO, French-process-synthesized ZnO, and sol–gel-synthesized ZnO in the ultraviolet and visible regions.
Figure 2
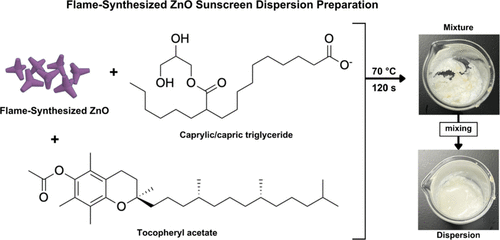
Figure 2. Schematic of preparing flame-synthesized ZnO (ZOTeN) particle dispersions for incorporation into a sunscreen formulation.
Figure 3
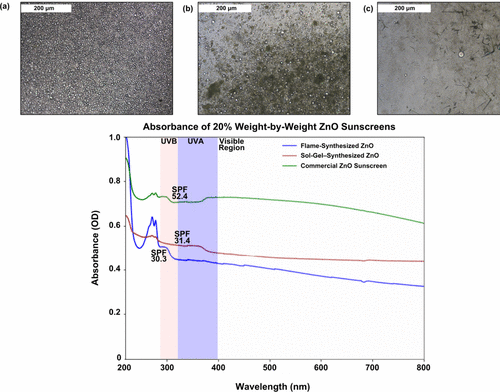
Figure 3. (a) Light microscope image of a 1:50 dilution of test formulation without ZnO in water, with scale bars of 200 μm. (b) Light microscope image of a 1:50 dilution of test formulation with sol–gel-synthesized ZnO (SgZnO) particles in water, with scale bars of 200 μm. (c) Light micrograph of a 1:50 dilution of test formulation with flame-synthesized ZnO (ZOTeN) particles in water, with scale bars of 200 μm. (d) Absorbance of ZOTeN particle sunscreen test formulation, SgZnO particle sunscreen test formulation, and FpZnO particle sunscreen test formulation.
Figure 4

Figure 4. (a) Schematic of oil-in-water emulsions containing sol–gel-synthesized ZnO and (b) flame-synthesized ZnO. (c) Viscosity measurements of flame-synthesized ZnO sunscreen and sol–gel-synthesized ZnO sunscreen over 12 weeks at 25 °C.
Figure 5
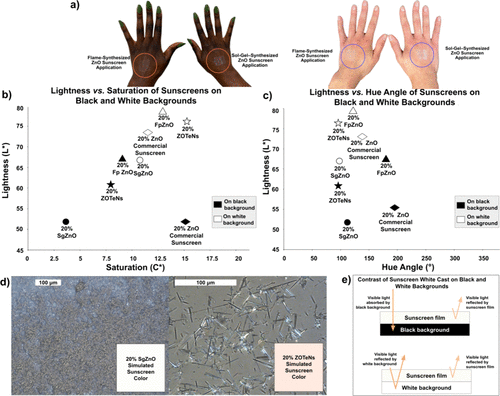
Figure 5. (a) Image of flame-synthesized (ZOTeN) particle-based sunscreen and sol–gel-synthesized (SgZnO) particle-based sunscreen application on hands categorized as (left) Fitzpatrick Skin Type 6 (darker tone) and image of ZOTeN particles and SgZnO particles application on hands categorized as (right) Fitzpatrick Skin Type 2 (lighter tone). (b) Lightness versus saturation of 20% w/w SgZnO particle-based sunscreen, 20% w/w ZOTeN particle-based sunscreen, 20% w/w FpZnO particle-based sunscreen, and 20% w/w ZnO commercial sunscreen on black and white backgrounds. (c) Lightness versus hue of 20% w/w sol–gel-synthesized ZnO (SgZnO) particle sunscreen, 20% w/w flame-synthesized (ZOTeN) particle sunscreen, 20% w/w FpZnO particle-based sunscreen, and 20% w/w ZnO commercial sunscreen particle sunscreen on black and white backgrounds. (d) Light microscope image of 1 g of 20% w/w SgZnO particle sunscreen with scale bars of 100 μm with UV/visible spectrum-simulated color swatch superimposed in the bottom right corner (left) and 1 g of 20% w/w ZOTeN particle sunscreen with scale bars of 100 μm with UV/Visible spectrum-simulated color swatch superimposed in the bottom right corner (right). (e) Diagram depicting contrast of sunscreen white cast on absolute black and white backgrounds.
References
This article references 48 other publications.
- 1Taylor, J. S. Unraveling the Molecular Pathway from Sunlight to Skin Cancer. Acc. Chem. Res. 1994, 27, 76– 82, DOI: 10.1021/ar00039a003There is no corresponding record for this reference.
- 2Rastogi, R. P.; Richa; Kumar, A.; Tyagi, M. B.; Sinha, R. P. Molecular Mechanisms of Ultraviolet Radiation-Induced DNA Damage and Repair. J. Nucleic Acids 2010, 2010, 592980, DOI: 10.4061/2010/592980There is no corresponding record for this reference.
- 3Neale, R.; Williams, G.; Green, A. Application Patterns among Participants Randomized to Daily Sunscreen Use in a Skin Cancer Prevention Trial. Arch. Dermatol. 2002, 138, 1319– 1325, DOI: 10.1001/archderm.138.10.1319There is no corresponding record for this reference.
- 4Addae, A. J.; Weiss, P. S. Standardizing the White Cast Potential of Sunscreens with Metal Oxide Ultraviolet Filters. Acc. Mater. Res. 2024, 5, 392– 399, DOI: 10.1021/accountsmr.4c00004There is no corresponding record for this reference.
- 5Addae, A. J.; Uyanga, J.; Chifamba, J.; Weiss, P. S. Advances in Enhancing Photoprotection of Sunscreens Using Hydrocarbon Film Formers and Carbon Nanomaterials. Adv. Drug Delivery Rev. 2025, 222, 115607, DOI: 10.1016/j.addr.2025.115607There is no corresponding record for this reference.
- 6Xu, S.; Kwa, M.; Agarwal, A.; Rademaker, A.; Kundu, R. V. Sunscreen Product Performance and Other Determinants of Consumer Preferences. JAMA Dermatol. 2016, 152, 920– 927, DOI: 10.1001/jamadermatol.2016.2344There is no corresponding record for this reference.
- 7Herzog, B.; Schultheiss, A.; Giesinger, J. On the Validity of Beer-Lambert Law and Its Significance for Sunscreens. Photochem. Photobiol. 2018, 94, 384– 389, DOI: 10.1111/php.12861There is no corresponding record for this reference.
- 8Lowe, N. J.; Shaath, N. A.; Pathak, M. A. Sunscreens: Development: Evaluation, and Regulatory Aspects, 2nd ed.; Marcel Dekker, Inc., 1997.There is no corresponding record for this reference.
- 9Khunkitti, W.; Satthanakul, P.; Waranuch, N.; Pitaksuteepong, T.; Kitikhun, P. Method for Screening Sunscreen Cream Formulations by Determination of in Vitro SPF and PA Values Using UV Transmission Spectroscopy and Texture Profile Analysis. J. Cosmet. Sci. 2014, 65, 147– 159There is no corresponding record for this reference.
- 10Binks, B. P.; Brown, J.; Fletcher, P. D. I.; Johnson, A. J.; Marinopoulos, I.; Crowther, J. M.; Thompson, M. A. Evaporation of Sunscreen Films: How the UV Protection Properties Change. ACS Appl. Mater. Interfaces 2016, 8, 13270– 13281, DOI: 10.1021/acsami.6b02696There is no corresponding record for this reference.
- 11Stiller, S.; Gers-Barlag, H.; Lergenmueller, M.; Pflücker, F.; Schulz, J.; Wittern, K. P.; Daniels, R. Investigation of the Stability in Emulsions Stabilized with Different Surface Modified Titanium Dioxides. Colloids Surf. Physicochem. Eng. Asp. 2004, 232, 261– 267, DOI: 10.1016/j.colsurfa.2003.11.003There is no corresponding record for this reference.
- 12Yeon, J. Y.; Seo, J. M.; Kim, T. H.; Shim, J. G. Emulsion Stability of Low Viscosity W/O Emulsion and Application of Inorganic Sunscreen Agents. J. Korean Appl. Sci. Technol. 2018, 35, 985– 1001, DOI: 10.12925/jkocs.2018.35.3.985There is no corresponding record for this reference.
- 13Chao, C.; Génot, C.; Rodriguez, C.; Magniez, H.; Lacourt, S.; Fievez, A.; Len, C.; Pezron, I.; Luart, D.; van Hecke, E. Emollients for Cosmetic Formulations: Towards Relationships between Physico-Chemical Properties and Sensory Perceptions. Colloids Surf. Physicochem. Eng. Asp. 2018, 536, 156– 164, DOI: 10.1016/j.colsurfa.2017.07.025There is no corresponding record for this reference.
- 14Mishra, Y. K.; Adelung, R. ZnO Tetrapod Materials for Functional Applications. Mater. Today 2018, 21, 631– 651, DOI: 10.1016/j.mattod.2017.11.003There is no corresponding record for this reference.
- 15Mishra, Y. K.; Kaps, S.; Schuchardt, A.; Paulowicz, I.; Jin, X.; Gedamu, D.; Freitag, S.; Claus, M.; Wille, S.; Kovalev, A.; Gorb, S. N.; Adelung, R. Fabrication of Macroscopically Flexible and Highly Porous 3D Semiconductor Networks from Interpenetrating Nanostructures by a Simple Flame Transport Approach. Part. Part. Syst. Charact. 2013, 30, 775– 783, DOI: 10.1002/ppsc.201300197There is no corresponding record for this reference.
- 16Klingshirn, C. ZnO: Material, Physics and Applications. ChemPhysChem 2007, 8, 782– 803, DOI: 10.1002/cphc.200700002There is no corresponding record for this reference.
- 17Lu, P. J.; Fang, S. W.; Cheng, W. L.; Huang, S. C.; Huang, M. C.; Cheng, H. F. Characterization of Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreen Powder by Comparing Different Measurement Methods. J. Food Drug Anal. 2018, 26, 1192– 1200, DOI: 10.1016/j.jfda.2018.01.010There is no corresponding record for this reference.
- 18Saito, G. P.; Bizari, M.; Cebim, M. A.; Correa, M. A.; Jafelicci Junior, M.; Davolos, M. R. Study of the Colloidal Stability and Optical Properties of Sunscreen Creams. Eclet. Quim. J. 2019, 44, 26– 36, DOI: 10.26850/1678-4618eqj.v44.2.2019.p26-36There is no corresponding record for this reference.
- 19Faure, B.; Salazar-Alvarez, G.; Ahniyaz, A.; Villaluenga, I.; Berriozabal, G.; De Miguel, Y. R.; Bergström, L. Dispersion and Surface Functionalization of Oxide Nanoparticles for Transparent Photocatalytic and UV-Protecting Coatings and Sunscreens. Sci. Technol. Adv. Mater. 2013, 14, 023001, DOI: 10.1088/1468-6996/14/2/023001There is no corresponding record for this reference.
- 20Paiva, J. P.; Diniz, R. R.; Leitão, A. C.; Cabral, L. M.; Fortunato, R. S.; Santos, B. A. M. C.; de Pádula, M. Insights and Controversies on Sunscreen Safety. Crit. Rev. Toxicol. 2020, 50, 707– 723, DOI: 10.1080/10408444.2020.1826899There is no corresponding record for this reference.
- 21Schilling, K.; Bradford, B.; Castelli, D.; Dufour, E.; Nash, J. F.; Pape, W.; Schulte, S.; Tooley, I.; van den Bosch, J.; Schellauf, F. Human Safety Review of “Nano” Titanium Dioxide and Zinc Oxide. Photochem. Photobiol. Sci. 2010, 9, 495– 509, DOI: 10.1039/b9pp00180hThere is no corresponding record for this reference.
- 22Aditya, A.; Chattopadhyay, S.; Gupta, N.; Alam, S.; Veedu, A. P.; Pal, M.; Singh, A.; Santhiya, D.; Ansari, K. M.; Ganguli, M. ZnO Nanoparticles Modified with an Amphipathic Peptide Show Improved Photoprotection in Skin. ACS Appl. Mater. Interfaces 2019, 11, 56– 72, DOI: 10.1021/acsami.8b08431There is no corresponding record for this reference.
- 23Abadi, P. G.-S.; Shirazi, F. H.; Joshaghani, M.; Moghimi, H. R. Ag+-Promoted Zinc Oxide [Zn(O):Ag]: A Novel Structure for Safe Protection of Human Skin against UVA Radiation. Toxicol. In Vitro 2018, 50, 318– 327, DOI: 10.1016/j.tiv.2018.02.016There is no corresponding record for this reference.
- 24Khanam, B. R.; Nidhi, M. K. J.; Nagaraja, H.; Manjunatha, T.; Angadi, B.; Uma Reddy, B.; Udaykumar, K. Physicochemical and Therapeutic Studies of Microwave-Synthesized ZnO and Cr-Doped ZnO Nanoparticles for Biomedical Applications. Inorg. Chem. Commun. 2024, 170, 113454, DOI: 10.1016/j.inoche.2024.113454There is no corresponding record for this reference.
- 25Bouie, A.; Baki, G. Iron Oxides and Ultramarine Blue in Inorganic Sunscreens for Dark Skin. J. Cosmet. Sci. 2025, 76, 123– 139There is no corresponding record for this reference.
- 26Dipak, Nipane; Thakare, S. R.; Khati, N. T. ZnO Nanoparticle by Sol-Gel and Its UV Application in Cosmetics Formulation. Int. J. Knowl. Eng. 2012, 3, 168– 169There is no corresponding record for this reference.
- 27Al-luhaibi, A. A.; Sendi, R. K. Synthesis, Potential of Hydrogen Activity, Biological and Chemical Stability of Zinc Oxide Nanoparticle Preparation by Sol-Gel: A Review. J. Radiat. Res. Appl. Sci. 2022, 15, 238– 254, DOI: 10.1016/j.jrras.2022.07.008There is no corresponding record for this reference.
- 28Hong, H. S. Current Status of Zinc Oxide Nanopowder Manufacturing Technology for UV Filters. Korean J. Mater. Res. 2025, 35, 167– 174, DOI: 10.3740/MRSK.2025.35.5.167There is no corresponding record for this reference.
- 29Mishra, Y. K.; Modi, G.; Cretu, V.; Postica, V.; Lupan, O.; Reimer, T.; Paulowicz, I.; Hrkac, V.; Benecke, W.; Kienle, L.; Adelung, R. Direct Growth of Freestanding ZnO Tetrapod Networks for Multifunctional Applications in Photocatalysis, UV Photodetection, and Gas Sensing. ACS Appl. Mater. Interfaces 2015, 7, 14303– 14316, DOI: 10.1021/acsami.5b02816There is no corresponding record for this reference.
- 30Modi, G. Zinc Oxide Tetrapod: A Morphology with Multifunctional Applications. Adv. Nat. Sci.: Nanosci. Nanotechnol. 2015, 6, 033002, DOI: 10.1088/2043-6262/6/3/033002There is no corresponding record for this reference.
- 31Mueen, R.; Lerch, M.; Cheng, Z.; Konstantinov, K. Na-Doped ZnO UV Filters with Reduced Photocatalytic Activity for Sunscreen Applications. J. Mater. Sci. 2020, 55, 2772– 2786, DOI: 10.1007/s10853-019-04122-2There is no corresponding record for this reference.
- 32Chegeni, M.; Pour, S. K.; Faraji Dizaji, B. Synthesis and Characterization of Novel Antibacterial Sol-Gel Derived TiO2/Zn2TiO4/Ag Nanocomposite as an Active Agent in Sunscreens. Ceram. Int. 2019, 45, 24413– 24418, DOI: 10.1016/j.ceramint.2019.08.163There is no corresponding record for this reference.
- 33Hasnidawani, J. N.; Azlina, H. N.; Norita, H.; Bonnia, N. N.; Ratim, S.; Ali, E. S. Synthesis of ZnO Nanostructures Using Sol-Gel Method. Procedia Chem. 2016, 19, 211– 216, DOI: 10.1016/j.proche.2016.03.095There is no corresponding record for this reference.
- 34Elbrolesy, A.; Elhussiny, F. A.; Abdou, Y.; Morsy, R. Facile Synthesis and Biophysical Characterization of Novel Zinc Oxide/Fe3O4 Hybrid Nanocomposite as a Potentially Active Agent in Sunscreens. Arab. J. Sci. Eng. 2024, 49, 1083– 1093, DOI: 10.1007/s13369-023-08082-3There is no corresponding record for this reference.
- 35Aikens, P. Formulation of Sunscreens in the United States. In Handbook of Formulating Dermal Applications; John Wiley & Sons, Ltd, 2016; pp 589– 609.There is no corresponding record for this reference.
- 36Egerton, T. A.; Tooley, I. R. UV Absorption and Scattering Properties of Inorganic-Based Sunscreens. Int. J. Cosmet. Sci. 2012, 34, 117– 122, DOI: 10.1111/j.1468-2494.2011.00689.xThere is no corresponding record for this reference.
- 37Arif, A.; Belahssen, O.; Gareh, S.; Benramache, S. The Calculation of Band Gap Energy in Zinc Oxide Films. J. Semicond. 2015, 36, 013001– 013006, DOI: 10.1088/1674-4926/36/1/013001There is no corresponding record for this reference.
- 38Marín, E.; Calderón, A. Conversion of Wavelength and Energy Scales and the Analysis of Optical Emission Spectra. J. Phys. Chem. Lett. 2022, 13, 8376– 8379, DOI: 10.1021/acs.jpclett.2c02621There is no corresponding record for this reference.
- 39Catalano, R.; Masion, A.; Ziarelli, F.; Slomberg, D.; Laisney, J.; Unrine, J. M.; Campos, A.; Labille, J. Optimizing the Dispersion of Nanoparticulate TiO2-Based UV Filters in a Non-Polar Medium Used in Sunscreen Formulations - The Roles of Surfactants and Particle Coatings. Colloids Surf. Physicochem. Eng. Asp. 2020, 599, 124792, DOI: 10.1016/j.colsurfa.2020.124792There is no corresponding record for this reference.
- 40Labille, J.; Feng, J.; Botta, C.; Borschneck, D.; Sammut, M.; Cabie, M.; Auffan, M.; Rose, J.; Bottero, J.-Y. Aging of TiO2 Nanocomposites Used in Sunscreen. Dispersion and Fate of the Degradation Products in Aqueous Environment. Environ. Pollut. 2010, 158, 3482– 3489, DOI: 10.1016/j.envpol.2010.02.012There is no corresponding record for this reference.
- 41Franco-Gil, M. E.; Graça, A.; Martins, A.; Marto, J.; Ribeiro, H. M. Emollients in Dermatological Creams: Early Evaluation for Tailoring Formulation and Therapeutic Performance. Int. J. Pharm. 2024, 653, 123825, DOI: 10.1016/j.ijpharm.2024.123825There is no corresponding record for this reference.
- 42Sapcharoenkun, C.; Klamchuen, A.; Kasamechonchung, P.; Iemsam-arng, J. Role of Rheological Behavior of Sunscreens Containing Nanoparticles on Thin Film Preparation. Mater. Sci. Eng., B 2020, 259, 114608, DOI: 10.1016/j.mseb.2020.114608There is no corresponding record for this reference.
- 43Calixto, L. S.; Maia Campos, P. M. B. G. Physical-Mechanical Characterization of Cosmetic Formulations and Correlation between Instrumental Measurements and Sensorial Properties. Int. J. Cosmet. Sci. 2017, 39, 527– 534, DOI: 10.1111/ics.12406There is no corresponding record for this reference.
- 44Hedjar, L.; Toscani, M.; Gegenfurtner, K. R. Importance of Hue: The Effect of Saturation on Hue-Chroma Asymmetries. J. Opt. Soc. Am. A 2025, 42, B305– B312, DOI: 10.1364/JOSAA.544641There is no corresponding record for this reference.
- 45Braun, G. J.; Fairchild, M. D.; Ebner, F. Color Gamut Mapping in a Hue-Linearized CIELAB Color Space. Color Imaging Conf. 1998, 6, 163– 168, DOI: 10.2352/CIC.1998.6.1.art00034There is no corresponding record for this reference.
- 46Bao, W.; Wei, M.; Xiao, K. Investigating Unique Hues at Different Chroma Levels with a Smaller Hue Angle Step. J. Opt. Soc. Am. A 2020, 37, 671– 679, DOI: 10.1364/JOSAA.383002There is no corresponding record for this reference.
- 47Wrolstad, R. E.; Smith, D. E. Color Analysis. In Food Analysis; Nielsen, S. S., Ed.; Springer US: Boston, MA, 2010; pp 573– 586.There is no corresponding record for this reference.
- 48Kim, M. H.; Weyrich, T.; Kautz, J. Modeling Human Color Perception under Extended Luminance Levels. ACM Transactions on Graphics 2009, 28, 27, DOI: 10.1145/1531326.1531333There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsmaterialslett.5c01351.
X-ray diffraction data of ZnO particles and SEM and calculated values of saturation and hue. Methods of synthesis and Python processing of colors. Centrifugal aging data of sunscreens. (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



