Synthesis and Biological Evaluation of a Novel Dual-Targeting Small Molecule Drug Conjugate Modulating the Crosstalk between α5β1 Integrin and MDM2 in GlioblastomaClick to copy article linkArticle link copied!
- Federico ArrigoniFederico ArrigoniDepartment of Science and High Technology Università degli Studi dell’Insubria, Via Valleggio 11, Como 22100, ItalyMore by Federico Arrigoni
- Ana FerrariAna FerrariA. Ferrari, A. Muzi, and Dr. G. Roscilli Takis s.r.l., Via Castel Romano 100, 00128 Rome, ItalyMore by Ana Ferrari
- Helena PrpićHelena PrpićDepartment of Science and High Technology Università degli Studi dell’Insubria, Via Valleggio 11, Como 22100, ItalyMore by Helena Prpić
- Elena MarkeviciuteElena MarkeviciuteDepartment of Science and High Technology Università degli Studi dell’Insubria, Via Valleggio 11, Como 22100, ItalyMore by Elena Markeviciute
- Alessia MuziAlessia MuziA. Ferrari, A. Muzi, and Dr. G. Roscilli Takis s.r.l., Via Castel Romano 100, 00128 Rome, ItalyMore by Alessia Muzi
- Giuseppe Roscilli*Giuseppe Roscilli*[email protected]A. Ferrari, A. Muzi, and Dr. G. Roscilli Takis s.r.l., Via Castel Romano 100, 00128 Rome, ItalyMore by Giuseppe Roscilli
- Silvia Gazzola*Silvia Gazzola*[email protected]Department of Science and High Technology Università degli Studi dell’Insubria, Via Valleggio 11, Como 22100, ItalyMore by Silvia Gazzola
- Umberto Piarulli*Umberto Piarulli*[email protected]Department of Science and High Technology Università degli Studi dell’Insubria, Via Valleggio 11, Como 22100, ItalyMore by Umberto Piarulli
Abstract
Negative crosstalk between α5β1 integrin and the p53-MDM2 regulatory axis contributes to glioblastoma progression and therapeutic resistance. To explore the potential of dual inhibition of these two biological targets, the dual targeting small molecule drug conjugate (SMDC) (1) was designed by coupling the MDM2 inhibitor SAR405838 to a selective α5β1 integrin ligand cyclo(phg-isoDGR-k) (7) through a stable chemical linker. The resulting conjugate retained antiproliferative activity in U87-MG glioblastoma cells and induced p53 reactivation with minimal MDM2 induction. Cell cycle distribution analysis revealed a redistribution of cells from the G0/G1 phase to the G2/M phase exclusively upon treatment with conjugate 1, suggesting that a different mechanism of action is engaged. These findings support the potential of this dual-targeting approach through a dual-targeting SMDC as a promising therapeutic strategy against high-grade glioma overexpressing the α5β1 integrin receptor.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
Glioblastoma multiforme (GBM) remains one of the most challenging brain tumors due to its aggressiveness and intrinsic resistance to standard therapies. (1) Besides the alkylating agent Temozolomide (TMZ), which represents the first-line treatment, (2) the pharmacological pipeline for glioblastoma is extremely limited, with only a handful of additional agents showing modest benefits. (3) The lack of effective treatments contributes to the poor prognosis of GBM, with the average survival of approximately 5 years. (4) The difficulty in treating GBM stems from its intrinsically heterogeneous nature and its ability to develop resistance. In this context, one of the mechanisms of resistance in GBM is the alteration of O6-methylguanine DNA methyl transferase (MGMT) activity, an enzyme responsible for repairing TMZ-induced lesions by removing methyl groups from the O6 position of guanine. (5) Notably, earlier studies have demonstrated that increased intracellular levels of the tumor suppressor p53 can repress MGMT expression. (5,6) The tumor suppressor p53, also known as the “guardian of the human genome”, is involved in a variety of biological pathways, such as apoptosis, cell cycle arrest, DNA repair, and autophagy, often related to suppressing aberrant cells. (7,8) Due to many transcriptional activities, in tumors the p53 functions are often downregulated by the overexpression of a p53-negative regulator, mouse double minute protein (MDM2, also known in humans as HDM2), an E3 ligase that binds the N-terminal domain of p53, and through nuclear export, promotes its 26S proteasomal degradation. (9,10)
Notably, more than 85% of glioblastoma cases involve alterations affecting the p53 pathway. (11,12) For these reasons, the development of small molecules targeting the MDM2 protein, thus impairing the MDM2-p53 complex formation and subsequent p53 degradation, has emerged as a promising strategy to restore p53 tumor-suppressor functions in GBM expressing p53. (13) After the elucidation of the p53-MDM2 crystal structure (PDB ID: 4HFZ), (14) numerous MDM2 inhibitors have been rationally designed to mimic the interactions between p53 and the three key hydrophobic residues of MDM2 (Phe19, Trp23, and Leu26). (15) One of the early successful classes of p53-MDM2 inhibitors was cis-imidazoline small molecules Nutlin-3a (Figure 1), which causes cell cycle arrest predominantly at the G0/G1 phase (16) and induces p53-dependent apoptosis in GBM and other cancer cell lines and xenograft models. (16,17) Other classes of next-generation p53-MDM2 inhibitors include stapled peptides (ATSP-7041, ALRN-6924), (18,19) benzodiazepine-2,5-diones (e.g., BDP), (20) piperidinones (e.g., AMG232), (21) spiro-oxindoles (e.g., SAR405838, APG115, BI907828) (22,23) and β-carbolines (e.g., CPI-7c, SP141). (24,25) In 2016, Verreault and co-workers (13) reported the preclinical efficacy of the MDM2 inhibitor RG7112 in GBM, demonstrating induction of tumor cell death, reduced tumor growth, and increased survival. More recently, the use of the cis-imidazoline derivative RG7388 (Idasanutlin) (26) and the piperidone AMG232 (27) has further highlighted the potential clinical relevance of MDM2 inhibition strategies in the treatment of glioblastoma. However, despite the extensive exploration of MDM2-p53 interactions, no FDA-approved MDM2 inhibitors are currently available, mainly due to p53’s pleiotropic functions and the resulting off-target effects. (28) In addition, activation of p53 expression also leads to an increase in MDM2 levels, as a result of the p53-MDM2 negative feedback loop, in which p53 promotes the transcription of MDM2, potentially attenuating the overall antitumor efficacy of such inhibitors. (29) A decreased survival of GBM patients has also been associated with a high expression of the α5 subunit of the α5β1 integrin receptor. (30) Specifically, preclinical studies have demonstrated that this integrin contributes to key GBM hallmarks, including enhanced cell survival, migratory capacity, therapy resistance, and promotion of neo-angiogenesis. (31−33) Remarkably, a “negative” crosstalk (in terms of downregulation and expression) between α5β1 and p53 pathway was proposed by Dontenwill and co-workers (34,35) as a significant contribution to the chemoresistance in GBM. (36) Indeed, by promoting the p53 activation with Nutlin 3a and inhibiting the integrin, a significant enhancement of the cell apoptosis process was observed. In a parallel approach, Marinelli and co-workers (37) investigated the simultaneous targeting of α5β1 integrin and MDM2 protein in glioblastoma multiforme using a dual-targeting small molecule. Their results showed that the simultaneous dual inhibition enhanced cell cycle arrest and reduced proliferation of the U87MG glioblastoma cancer cell line, while also markedly decreasing cell invasiveness compared to treatment with Nutlin-3a alone.
Figure 1
Figure 1. Summary of key studies targeting α5β1 and p53 pathways in GBM. Renner et al. (35) used coadministration of α5β1 antagonists (36) and Nutlin-3a; Merlino et al. (37) developed the first dual-targeting molecule; our work introduces the first small-molecule drug conjugate 1 (SMDC) linking an α5β1 ligand to an MDM2 inhibitor.
Considering our general interest in the synthesis and biological evaluation of integrin ligand–based small-molecule drug conjugates (SMDCs) for the selective delivery of cytotoxic agents to cancer cells, (38−41) and given the therapeutic relevance of both α5β1 integrin and MDM2 as cancer targets in GBM, we report herein the synthesis and biological evaluation of a new dual targeting conjugate, combining the spiro-oxindole derivative SAR405838 (22,23) (a potent MDM2 inhibitor with subnanomolar binding affinity), and the α5β1 integrin ligand cyclo(phg-isoDGR-k), (42,43) originally identified by Kessler and co-workers as a highly potent and selective α5β1 integrin ligand, connected through a stable chemical spacer as depicted in Figure 1. The latter was selected over other candidates due to the convenient conjugation site provided by the lysine side-chain amino group, as well as its recently demonstrated ability to recognize α5β1 integrin-overexpressing cells in vivo, as validated by nano-SPECT/CT imaging using 99mTc labeling. (44,45) To design the desired dual-inhibitor conjugate featuring a stable linker, we first examined how chemical modification of the MDM2 inhibitor SAR405838 influences its biological activity in the U87-MG cancer cell line, which expresses wild-type p53. To our knowledge, no previous studies have reported functionalization of SAR405838. We therefore targeted the secondary hydroxyl group on the cyclohexyl ring for derivatization, as this site is solvent-exposed according to the cocrystal structure of the human MDM2-SAR405838 complex (PDB: 5TRF, Figure 2). (46)
Figure 2
Figure 2. Predicted binding pose of SAR405838 in the MDM2 pocket (AutoDock Vina, based on PDB: 5TRF). The solvent-accessible hydroxyl group on the cyclohexyl ring is indicated as the conjugation site.
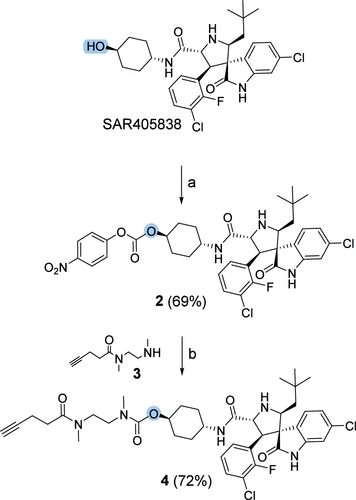
The choice of conjugation site was supported by our recent work, in which inserting a hydroxyalkyl chain at a solvent-exposed position of the β-carboline-based MDM2 inhibitor did not significantly impair the compound’s biological activity. (47) To functionalize the selected hydroxyl group, we chose an N-methyl ethylenediamine–pentynoic acid linker (4) with a terminal alkyne for click reaction with the azide moiety and an amide-based backbone to avoid a hydrolyzable ester bond in the final conjugate. As outlined in Scheme 1, the secondary hydroxyl group of SAR405838 was first activated by conversion to the corresponding p-nitrophenyl carbonate 2 (69% yield). The intermediate was coupled under basic conditions with the previously synthesized alkyne linker 3, (47) affording the desired compound 4 in 72% yield. To evaluate whether linker installation affected biological activity, we tested compound 4 on U87-MG glioblastoma cells using the CellTiter-Glo luminescent cell viability assay after 72 h of incubation.
Scheme 1
aReagents and conditions: 4-nitrophenylchloroformate, pyridine, CH2Cl2, from −50 to −20 °C, 7 h.
bDMAP, iPr2NEt, CH2Cl2, 16 h.
The antiproliferative activity of 4 (0.76 ± 1.27 μM) was fully comparable to that of SAR405838 (2.01 ± 1.34 μM, respectively), thus confirming the suitability of the selected conjugation site (Figure 3). The previously identified α5β1 ligand, cyclo(phg-isoDGR-k) (7), was synthesized by slightly modifying a reported protocol by Kessler et al. (43) The synthesis was initiated with the solid-phase peptide synthesis (SPPS) of Fmoc-k(Boc)-phg-isoD(t-Bu)GR(Pbf)–OH (5) using DIC/Oxyma as coupling agents. After cleavage from 2-CTC resin using 20% HFIP in DCM, macrolactamization was performed using HATU/Oxyma/DIPEA (43) under pseudodilution conditions (7.5 mM, peptide concentration), delivered via syringe pump, which afforded compound 6 in 72% yield. Obtained compound 6 was treated with cleavage cocktail TFA/EDT/TIS/water (95:2:2:1) to afford cyclo(phg-isoDGR-phg-k) (7) (Supp info, Synthesis 1.4. – 1.6.) which was then functionalized at the ε-amino group of lysine side chain with the azido-tetraethylene glycol spacer in 75% yield. The resulting intermediate 8 was subsequently coupled to compound 4 through the copper-catalyzed azide alkyne cycloaddition (CuAAC), affording the final conjugate 1 in 51% yield (Scheme 2).
Figure 3
Figure 3. Antiproliferative activity of SAR405838 and compound 4 on U87-MG glioblastoma cells. Cells were treated with increasing concentrations of SAR405838 or compound 4 for 72 h, and cell viability was assessed using the CellTiter-Glo Luminescent Cell Viability Assay. Data represent mean ± SD of three technical replicates from one representative experiment out of at least three independent experiments, all yielding consistent results. IC50 values were calculated by nonlinear regression using GraphPad Prism; reported standard deviations reflect the fitting error of the regression model.
Scheme 2
aReagents and conditions: Compound 8 (1.1 equiv), compound 4 (1 equiv), CuSO4 × 5H2O (0.5 equiv), sodium ascorbate (0.6 equiv), N2, DMF:H2O = 1:1, 0–25 °C, 16 h.
The cytotoxicity of conjugate 1 was evaluated in the U87-MG cell line alongside the α5β1 integrin ligand 7, SAR405838, and a 1:1 coadministration of 7 and SAR405838. Prior to these studies, the high expression of the α5β1 integrin receptor on our targeted U87-MG cancer cell line was confirmed by flow cytometry (FACS) analysis (Table S2, Figure S1 in Supp info). Interestingly, SMDC 1 displayed cytotoxicity comparable to that of the free drug SAR405838 (IC50 = 1.46 ± 0.41 μM vs 2.0 ± 1.34 μM) with no apparent loss of potency. Similar activity was observed for the coadministration of 7 and SAR405838, while integrin ligand alone (7) displayed negligible cytotoxicity (Figure 4).
Figure 4
Figure 4. Antiproliferative activity of SAR405838, cyclo(phg-isoDGR-k) (7), their coadministration, and conjugate 1 in U87-MG glioblastoma cells. Cells were treated for 72 h with increasing concentrations of the indicated compounds, and viability was assessed using the CellTiter-Glo assay. Data represent mean ± SD of three technical replicates from one representative experiment out of at least three independent replicates, all showing consistent trends. IC50 values were calculated by nonlinear regression using GraphPad Prism; reported standard deviations reflect the fitting error of the regression model.
Subsequently, we interrogated the effect of the compounds on the expression of p53 and its associated proteins p21 and MDM2 in U87-MG cells by Western blot. Treatment with conjugate 1 for 24 h caused a dose-dependent induction of p53 (0.1, 1, 3, 10 μM), along with upregulation of p21. Notably, while SAR405838 at 3 μM robustly induced MDM2 via the canonical p53-MDM2 negative feedback (as expected for MDM2 inhibitors such as Nutlin-3a), conjugate 1 markedly attenuated MDM2 induction at the same concentration (Figure 5a). At 10 μM of 1 a slight increment of the expression of MDM2 is observed, which might reasonably rise by the activation of parallel regulatory pathways that upregulate MDM2 under highly stressed conditions. (48) Nevertheless, while SAR405838 alone confirms the classical MDM2 inhibitor action (p53 stabilization + MDM2 feedback), the conjugate 1 demonstrates an ability to modulate that regulatory feedback (Figure 5a). This pattern suggests that conjugate 1 may partially disconnect p53 activation from its negative feedback control, sustaining a more robust p53/p21 response, which can reasonably be attributed to a synergistic effect arising from the simultaneous targeting of both α5β1 integrin and MDM2. (34,35) To gain deeper insight into these results, we evaluated the effects of conjugate 1 on cell cycle distribution using FACS analysis. According to literature reports, in U87-MG cells expressing wt-p53, the MDM2 inhibitors, such as Nutlin-3a, induce cell cycle arrest predominantly in the G0/G1 phase. (16) Consistent with the published data, treatment of the U87-MG cells with 10 μM of SAR405838, whose effect on this cell line has not been previously reported, resulted in a modest increase in the apoptotic sub-G1 fraction (3.91% vs 1.60% in control), which nonetheless corresponds to a measurable cytotoxic effect in the longer-term proliferation assay (Figure 5b, histogram 1). In contrast, 10 μM conjugate 1 induced a substantially higher sub-G1 peak (12.01%), indicating enhanced apoptosis (Figure 5b, histogram 4).
Figure 5
Figure 5. (a) Western blot response-dependent analysis of conjugate 1, cyclo(phg-isoDGR-k) (7), and SAR405838 on p53, p21, and MDM2 expression in U87-MG cells. Concentration of SAR405838 chosen on previously performed dose-dependent Western blot analysis. (b) Cell cycle distribution analysis in U87-MG cells treated with 10 μM of the respective compounds for 24 h. DNA content was assessed by FxCycle staining and analyzed by FACS; histograms shown represent SAR405838 (histogram 1), cyclo(phg-isoDGR-k) (7, histogram 2), 1:1 molar coadministration of 7 and SAR405838 (histogram 3), and compound 1 (histogram 4). Cell population distributions in G0/G1, S, G2/M, and sub-G1 phases are summarized in the bar graph. Full gating strategy, replicate histograms, and control conditions are available in the Supporting Information (Table S3, Figure S2).
Conjugate 1 also drove a prominent accumulation in G2/M (35.90%), whereas SAR405838 alone showed only about 7.4% in G2/M, a percentage similar to control, indicating limited engagement of that checkpoint, as expected. With SAR405838 alone, we did not observe strong accumulation in G0/G1 relative to control as might be expected for MDM2 inhibitors: this may be because the G0/G1 arrest induced by p53 activation is transient; by the 24 h measurement point, some cells may already have progressed into apoptosis (sub-G1), thereby masking an earlier G0/G1 signature. Interestingly, glioma cells such as U87MG are particularly prone to p53-driven, (16) reinforcing the fact that in our data we observed predominantly sub-G1 increase rather than robust G0/G1 arrest. Co-administration of SAR405838 and compound 7 (1:1) produced a profile close to SAR405838 indicating that most of the effect was attributed to the MDM2 inhibitor. This shift in the cell cycle profile suggests that conjugate 1, by simultaneously inhibiting the overexpressed α5β1 integrin receptor and the MDM2–p53 complex formation, may influence cell cycle regulation through mechanisms not observed with either component alone or in coadministration. These effects may involve activation of stress signaling or DNA damage response pathways, (49) resulting in a distinct cellular response compared to that of SAR405838.
In conclusion, we have developed conjugate 1, a dual-targeted small-molecule drug conjugate that links the MDM2–p53 inhibitor SAR405838 to the α5β1 integrin ligand (7) through a stable linker, with the aim of investigating a possible synergistic effect arising by the modulation of the crosstalk between α5β1 integrin and MDM2 in glioblastoma by integrin–MDM2 crosstalk. Importantly, conjugate 1 retains potent antiproliferative activity in U87-MG glioblastoma cells, showing no loss of efficacy compared to the unconjugated MDM2 inhibitor (1). Western blot data confirms reactivation of p53 and its transcriptional downstream target p21, while concurrently attenuating the compensatory upregulation of MDM2. The cell cycle analysis further reveals a distinct arrest profile relative to compound SAR405838 alone, with increased accumulation in the G2/M and sub-G1 phases. Although further work is needed to elucidate the mechanism of action of conjugate 1, as well as to assess its selectivity toward α5β1 overexpressing cancer lines, our findings support the dual-targeting approach and provide the foundation for further development of precision therapeutics against glioblastoma.
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsmedchemlett.5c00669.
The PDF file includes general procedure for the chemical synthesis of the compounds, NMR spectra, HPLC purity analysis, HRMS/LC–MS spectra, and all the procedures of the performed biological assays, including data of α5β1 integrin expression levels on different cancer cell lines and FACS analysis data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We thank the European Commission (Marie Skłodowska-Curie ITN MAGICBULLET: RELOADED 861316) and Ministero dell’Università e della Ricerca (PRIN 2020 project 2020833Y75) for the fundings. The scientific support from CRIETT center of University of Insubria (instrument code: MAC01 and MAC15, which was acquired thanks to the funding by Regione Lombardia, regional law No. 9/2020, resolution No. 3776/2020) is greatly acknowledged.
| GBM | glioblastoma multiforme |
| TMZ | temozolomide |
| MGMT | O6-methylguanine DNA methyl transferase |
| MDM2 | mouse double minute protein |
| SMDC | small molecule drug conjugate |
| DMAP | dimethylaminopyridine |
| SD | standard deviation |
| DMF | dimethylformamide |
| DMSO | dimethyl sulfoxide |
References
This article references 49 other publications.
- 1Alirezaei, Z.; Amouheidari, A.; BasirianJahromi, R.; Seyyedhosseini, S.; Hamidi, A. Survival Analysis of Glioblastoma: A Scientometric Perspective. World Neurosurg. 2025, 194, 123476, DOI: 10.1016/j.wneu.2024.11.059Google ScholarThere is no corresponding record for this reference.
- 2Stupp, R.; Mason, W. P.; Van Den Bent, M. J.; Weller, M.; Fisher, B.; Taphoorn, M. J. B.; Belanger, K.; Brandes, A. A.; Marosi, C.; Bogdahn, U.; Curschmann, J.; Janzer, R. C.; Ludwin, S. K.; Gorlia, T.; Allgeier, A.; Lacombe, D.; Cairncross, J. G.; Eisenhauer, E.; Mirimanoff, R. O. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352 (10), 987– 996, DOI: 10.1056/NEJMoa043330Google ScholarThere is no corresponding record for this reference.
- 3Obrador, E.; Moreno-Murciano, P.; Oriol-Caballo, M.; López-Blanch, R.; Pineda, B.; Gutiérrez-Arroyo, J.; Loras, A.; Gonzalez-Bonet, L.; Martinez-Cadenas, C.; Estrela, J.; Marqués-Torrejón, M. Glioblastoma Therapy: Past, Present and Future. Int. J. Mol. Sci. 2024, 25 (5), 2529, DOI: 10.3390/ijms25052529Google ScholarThere is no corresponding record for this reference.
- 4Cruz Da Silva, E.; Mercier, M.-C.; Etienne-Selloum, N.; Dontenwill, M.; Choulier, L. A Systematic Review of Glioblastoma-Targeted Therapies in Phases II, III, IV Clinical Trials. Cancers 2021, 13 (8), 1795, DOI: 10.3390/cancers13081795Google ScholarThere is no corresponding record for this reference.
- 5Park, C.-K.; Kim, J. E.; Kim, J. Y.; Song, S. W.; Kim, J. W.; Choi, S. H.; Kim, T. M.; Lee, S.-H.; Kim, I. H.; Park, S.-H. The Changes in MGMT Promoter Methylation Status in Initial and Recurrent Glioblastomas. Transl. Oncol. 2012, 5 (5), 393– IN19, DOI: 10.1593/tlo.12253Google ScholarThere is no corresponding record for this reference.
- 6Bocangel, D.; Sengupta, S.; Mitra, S.; Bhakat, K. K. P53-Mediated down-Regulation of the Human DNA Repair Gene O6-Methylguanine-DNA Methyltransferase (MGMT) via Interaction with Sp1 Transcription Factor. Anticancer Res. 2009, 29 (10), 3741– 3750Google ScholarThere is no corresponding record for this reference.
- 7Kastenhuber, E. R.; Lowe, S. W. Putting P53 in Context. Cell 2017, 170 (6), 1062– 1078, DOI: 10.1016/j.cell.2017.08.028Google ScholarThere is no corresponding record for this reference.
- 8Sullivan, K. D.; Galbraith, M. D.; Andrysik, Z.; Espinosa, J. M. Mechanisms of Transcriptional Regulation by P53. Cell Death Differ. 2018, 25 (1), 133– 143, DOI: 10.1038/cdd.2017.174Google ScholarThere is no corresponding record for this reference.
- 9Crespo, I.; Vital, A. L.; Gonzalez-Tablas, M.; Patino, M. D. C.; Otero, A.; Lopes, M. C.; De Oliveira, C.; Domingues, P.; Orfao, A.; Tabernero, M. D. Molecular and Genomic Alterations in Glioblastoma Multiforme. Am. J. Pathol. 2015, 185 (7), 1820– 1833, DOI: 10.1016/j.ajpath.2015.02.023Google ScholarThere is no corresponding record for this reference.
- 10England, B.; Huang, T.; Karsy, M. Current Understanding of the Role and Targeting of Tumor Suppressor P53 in Glioblastoma Multiforme. Tumor Biol. 2013, 34 (4), 2063– 2074, DOI: 10.1007/s13277-013-0871-3Google ScholarThere is no corresponding record for this reference.
- 11Pouyan, A.; Ghorbanlo, M.; Eslami, M.; Jahanshahi, M.; Ziaei, E.; Salami, A.; Mokhtari, K.; Shahpasand, K.; Farahani, N.; Meybodi, T. E.; Entezari, M.; Taheriazam, A.; Hushmandi, K.; Hashemi, M. Glioblastoma Multiforme: Insights into Pathogenesis, Key Signaling Pathways, and Therapeutic Strategies. Mol. Cancer 2025, 24 (1), 58, DOI: 10.1186/s12943-025-02267-0Google ScholarThere is no corresponding record for this reference.
- 12The Cancer Genome Atlas Research Network. Comprehensive Genomic Characterization Defines Human Glioblastoma Genes and Core Pathways. Nature 2008, 455 (7216), 1061– 1068, DOI: 10.1038/nature07385Google ScholarThere is no corresponding record for this reference.
- 13Verreault, M.; Schmitt, C.; Goldwirt, L.; Pelton, K.; Haidar, S.; Levasseur, C.; Guehennec, J.; Knoff, D.; Labussière, M.; Marie, Y.; Ligon, A. H.; Mokhtari, K.; Hoang-Xuan, K.; Sanson, M.; Alexander, B. M.; Wen, P. Y.; Delattre, J.-Y.; Ligon, K. L.; Idbaih, A. Preclinical Efficacy of the MDM2 Inhibitor RG7112 in MDM2 -Amplified and TP53 Wild-Type Glioblastomas. Clin. Cancer Res. 2016, 22 (5), 1185– 1196, DOI: 10.1158/1078-0432.CCR-15-1015Google ScholarThere is no corresponding record for this reference.
- 14Kussie, P. H.; Gorina, S.; Marechal, V.; Elenbaas, B.; Moreau, J.; Levine, A. J.; Pavletich, N. P. Structure of the MDM2 Oncoprotein Bound to the P53 Tumor Suppressor Transactivation Domain. Science 1996, 274 (5289), 948– 953, DOI: 10.1126/science.274.5289.948Google ScholarThere is no corresponding record for this reference.
- 15Vassilev, L. T.; Vu, B. T.; Graves, B.; Carvajal, D.; Podlaski, F.; Filipovic, Z.; Kong, N.; Kammlott, U.; Lukacs, C.; Klein, C.; Fotouhi, N.; Liu, E. A. In Vivo Activation of the P53 Pathway by Small-Molecule Antagonists of MDM2. Science 2004, 303 (5659), 844– 848, DOI: 10.1126/science.1092472Google ScholarThere is no corresponding record for this reference.
- 16Villalonga-Planells, R.; Coll-Mulet, L.; Martínez-Soler, F.; Castaño, E.; Acebes, J.-J.; Giménez-Bonafé, P.; Gil, J.; Tortosa, A. Activation of P53 by Nutlin-3a Induces Apoptosis and Cellular Senescence in Human Glioblastoma Multiforme. PLoS One 2011, 6 (4), e18588 DOI: 10.1371/journal.pone.0018588Google ScholarThere is no corresponding record for this reference.
- 17Tovar, C.; Rosinski, J.; Filipovic, Z.; Higgins, B.; Kolinsky, K.; Hilton, H.; Zhao, X.; Vu, B. T.; Qing, W.; Packman, K.; Myklebost, O.; Heimbrook, D. C.; Vassilev, L. T. Small-Molecule MDM2 Antagonists Reveal Aberrant P53 Signaling in Cancer: Implications for Therapy. Proc. Natl. Acad. Sci. U. S. A. 2006, 103 (6), 1888– 1893, DOI: 10.1073/pnas.0507493103Google ScholarThere is no corresponding record for this reference.
- 18Carvajal, L. A.; Neriah, D. B.; Senecal, A.; Benard, L.; Thiruthuvanathan, V.; Yatsenko, T.; Narayanagari, S.-R.; Wheat, J. C.; Todorova, T. I.; Mitchell, K.; Kenworthy, C.; Guerlavais, V.; Annis, D. A.; Bartholdy, B.; Will, B.; Anampa, J. D.; Mantzaris, I.; Aivado, M.; Singer, R. H.; Coleman, R. A.; Verma, A.; Steidl, U. Dual Inhibition of MDMX and MDM2 as a Therapeutic Strategy in Leukemia. Sci. Transl. Med. 2018, 10 (436), eaao3003 DOI: 10.1126/scitranslmed.aao3003Google ScholarThere is no corresponding record for this reference.
- 19Chang, Y. S.; Graves, B.; Guerlavais, V.; Tovar, C.; Packman, K.; To, K.-H.; Olson, K. A.; Kesavan, K.; Gangurde, P.; Mukherjee, A.; Baker, T.; Darlak, K.; Elkin, C.; Filipovic, Z.; Qureshi, F. Z.; Cai, H.; Berry, P.; Feyfant, E.; Shi, X. E.; Horstick, J.; Annis, D. A.; Manning, A. M.; Fotouhi, N.; Nash, H.; Vassilev, L. T.; Sawyer, T. K. Stapled Α-helical Peptide Drug Development: A Potent Dual Inhibitor of MDM2 and MDMX for P53-Dependent Cancer Therapy. Proc. Natl. Acad. Sci. U. S. A. 2013, 110 (36), E3445– E3454, DOI: 10.1073/pnas.1303002110Google ScholarThere is no corresponding record for this reference.
- 20Koblish, H. K.; Zhao, S.; Franks, C. F.; Donatelli, R. R.; Tominovich, R. M.; LaFrance, L. V.; Leonard, K. A.; Gushue, J. M.; Parks, D. J.; Calvo, R. R.; Milkiewicz, K. L.; Marugán, J. J.; Raboisson, P.; Cummings, M. D.; Grasberger, B. L.; Johnson, D. L.; Lu, T.; Molloy, C. J.; Maroney, A. C. Benzodiazepinedione Inhibitors of the Hdm2:P53 Complex Suppress Human Tumor Cell Proliferation in Vitro and Sensitize Tumors to Doxorubicin in Vivo. Mol. Cancer Ther. 2006, 5 (1), 160– 169, DOI: 10.1158/1535-7163.MCT-05-0199Google ScholarThere is no corresponding record for this reference.
- 21Rew, Y.; Sun, D. Discovery of a Small Molecule MDM2 Inhibitor (AMG 232) for Treating Cancer. J. Med. Chem. 2014, 57 (15), 6332– 6341, DOI: 10.1021/jm500627sGoogle ScholarThere is no corresponding record for this reference.
- 22Gollner, A.; Weinstabl, H.; Fuchs, J. E.; Rudolph, D.; Garavel, G.; Hofbauer, K. S.; Karolyi-Oezguer, J.; Gmaschitz, G.; Hela, W.; Kerres, N.; Grondal, E.; Werni, P.; Ramharter, J.; Broeker, J.; McConnell, D. B. Targeted Synthesis of Complex Spiro[3 H -indole-3,2′-pyrrolidin]-2(1 H)-ones by Intramolecular Cyclization of Azomethine Ylides: Highly Potent MDM2-P53 Inhibitors. ChemMedChem. 2019, 14 (1), 88– 93, DOI: 10.1002/cmdc.201800617Google ScholarThere is no corresponding record for this reference.
- 23Ding, K.; Lu, Y.; Nikolovska-Coleska, Z.; Wang, G.; Qiu, S.; Shangary, S.; Gao, W.; Qin, D.; Stuckey, J.; Krajewski, K.; Roller, P. P.; Wang, S. Structure-Based Design of Spiro-Oxindoles as Potent, Specific Small-Molecule Inhibitors of the MDM2-p53 Interaction. J. Med. Chem. 2006, 49 (12), 3432– 3435, DOI: 10.1021/jm051122aGoogle ScholarThere is no corresponding record for this reference.
- 24Wang, W.; Qin, J.-J.; Voruganti, S.; Srivenugopal, K. S.; Nag, S.; Patil, S.; Sharma, H.; Wang, M.-H.; Wang, H.; Buolamwini, J. K.; Zhang, R. The Pyrido[b]Indole MDM2 Inhibitor SP-141 Exerts Potent Therapeutic Effects in Breast Cancer Models. Nat. Commun. 2014, 5 (1), 5086, DOI: 10.1038/ncomms6086Google ScholarThere is no corresponding record for this reference.
- 25Chauhan, S. S.; Singh, A. K.; Meena, S.; Lohani, M.; Singh, A.; Arya, R. K.; Cheruvu, S. H.; Sarkar, J.; Gayen, J. R.; Datta, D.; Chauhan, P. M. S. Synthesis of Novel β-Carboline Based Chalcones with High Cytotoxic Activity against Breast Cancer Cells. Bioorg. Med. Chem. Lett. 2014, 24 (13), 2820– 2824, DOI: 10.1016/j.bmcl.2014.04.109Google ScholarThere is no corresponding record for this reference.
- 26Berberich, A.; Kessler, T.; Thomé, C. M.; Pusch, S.; Hielscher, T.; Sahm, F.; Oezen, I.; Schmitt, L.-M.; Ciprut, S.; Hucke, N.; Ruebmann, P.; Fischer, M.; Lemke, D.; Breckwoldt, M. O.; Von Deimling, A.; Bendszus, M.; Platten, M.; Wick, W. Targeting Resistance against the MDM2 Inhibitor RG7388 in Glioblastoma Cells by the MEK Inhibitor Trametinib. Clin. Cancer Res. 2019, 25 (1), 253– 265, DOI: 10.1158/1078-0432.CCR-18-1580Google ScholarThere is no corresponding record for this reference.
- 27Her, N.-G.; Oh, J.-W.; Oh, Y. J.; Han, S.; Cho, H. J.; Lee, Y.; Ryu, G. H.; Nam, D.-H. Potent Effect of the MDM2 Inhibitor AMG232 on Suppression of Glioblastoma Stem Cells. Cell Death Dis. 2018, 9 (8), 792, DOI: 10.1038/s41419-018-0825-1Google ScholarThere is no corresponding record for this reference.
- 28Punganuru, S. R.; Arutla, V.; Zhao, W.; Rajaei, M.; Deokar, H.; Zhang, R.; Buolamwini, J. K.; Srivenugopal, K. S.; Wang, W. Targeted Brain Tumor Therapy by Inhibiting the MDM2 Oncogene: In Vitro and In Vivo Antitumor Activity and Mechanism of Action. Cells 2020, 9 (7), 1592, DOI: 10.3390/cells9071592Google ScholarThere is no corresponding record for this reference.
- 29Wang, Z.; Zhang, S.; Irakoze, L. M.; Zhao, Y. Targeting P53 Activation: Recent Therapeutic Advances in Cancer and Diabetic Macular Edema. Eur. J. Med. Chem. 2025, 297, 117909, DOI: 10.1016/j.ejmech.2025.117909Google ScholarThere is no corresponding record for this reference.
- 30Etienne-Selloum, N.; Prades, J.; Bello-Roufai, D.; Boone, M.; Sevestre, H.; Trudel, S.; Caillet, P.; Coutte, A.; Desenclos, C.; Constans, J.-M.; Martin, S.; Choulier, L.; Chauffert, B.; Dontenwill, M. Expression Analysis of Α5 Integrin Subunit Reveals Its Upregulation as a Negative Prognostic Biomarker for Glioblastoma. Pharmaceuticals 2021, 14 (9), 882, DOI: 10.3390/ph14090882Google ScholarThere is no corresponding record for this reference.
- 31Schaffner, F.; Ray, A.; Dontenwill, M. Integrin Α5β1, the Fibronectin Receptor, as a Pertinent Therapeutic Target in Solid Tumors. Cancers 2013, 5 (1), 27– 47, DOI: 10.3390/cancers5010027Google ScholarThere is no corresponding record for this reference.
- 32Renner, G.; Noulet, F.; Mercier, M.-C.; Choulier, L.; Etienne-Selloum, N.; Gies, J.-P.; Lehmann, M.; Lelong-Rebel, I.; Martin, S.; Dontenwill, M. Expression/Activation of Α5β1 Integrin Is Linked to the β-Catenin Signaling Pathway to Drive Migration in Glioma Cells. Oncotarget 2016, 7 (38), 62194– 62207, DOI: 10.18632/oncotarget.11552Google ScholarThere is no corresponding record for this reference.
- 33Sani, S.; Pallaoro, N.; Messe, M.; Bernhard, C.; Etienne-Selloum, N.; Kessler, H.; Marinelli, L.; Entz-Werle, N.; Foppolo, S.; Martin, S.; Reita, D.; Dontenwill, M. Temozolomide-Acquired Resistance Is Associated with Modulation of the Integrin Repertoire in Glioblastoma, Impact of Α5β1 Integrin. Cancers 2022, 14 (2), 369, DOI: 10.3390/cancers14020369Google ScholarThere is no corresponding record for this reference.
- 34Janouskova, H.; Maglott, A.; Leger, D. Y.; Bossert, C.; Noulet, F.; Guerin, E.; Guenot, D.; Pinel, S.; Chastagner, P.; Plenat, F.; Entz-Werle, N.; Lehmann-Che, J.; Godet, J.; Martin, S.; Teisinger, J.; Dontenwill, M. Integrin Α5β1 Plays a Critical Role in Resistance to Temozolomide by Interfering with the P53 Pathway in High-Grade Glioma. Cancer Res. 2012, 72 (14), 3463– 3470, DOI: 10.1158/0008-5472.CAN-11-4199Google ScholarThere is no corresponding record for this reference.
- 35Renner, G.; Janouskova, H.; Noulet, F.; Koenig, V.; Guerin, E.; Bär, S.; Nuesch, J.; Rechenmacher, F.; Neubauer, S.; Kessler, H.; Blandin, A.-F.; Choulier, L.; Etienne-Selloum, N.; Lehmann, M.; Lelong-Rebel, I.; Martin, S.; Dontenwill, M. Integrin Α5β1 and P53 Convergent Pathways in the Control of Anti-Apoptotic Proteins PEA-15 and Survivin in High-Grade Glioma. Cell Death Differ. 2016, 23 (4), 640– 653, DOI: 10.1038/cdd.2015.131Google ScholarThere is no corresponding record for this reference.
- 36Ray, A.-M.; Schaffner, F.; Janouskova, H.; Noulet, F.; Rognan, D.; Lelong-Rebel, I.; Choulier, L.; Blandin, A.-F.; Lehmann, M.; Martin, S.; Kapp, T.; Neubauer, S.; Rechenmacher, F.; Kessler, H.; Dontenwill, M. Single Cell Tracking Assay Reveals an Opposite Effect of Selective Small Non-Peptidic Α5β1 or Αvβ3/Β5 Integrin Antagonists in U87MG Glioma Cells. Biochim. Biophys. Acta BBA - Gen. Subj. 2014, 1840 (9), 2978– 2987, DOI: 10.1016/j.bbagen.2014.04.024Google ScholarThere is no corresponding record for this reference.
- 37Merlino, F.; Daniele, S.; La Pietra, V.; Di Maro, S.; Di Leva, F. S.; Brancaccio, D.; Tomassi, S.; Giuntini, S.; Cerofolini, L.; Fragai, M.; Luchinat, C.; Reichart, F.; Cavallini, C.; Costa, B.; Piccarducci, R.; Taliani, S.; Da Settimo, F.; Martini, C.; Kessler, H.; Novellino, E.; Marinelli, L. Simultaneous Targeting of RGD-Integrins and Dual Murine Double Minute Proteins in Glioblastoma Multiforme. J. Med. Chem. 2018, 61 (11), 4791– 4809, DOI: 10.1021/acs.jmedchem.8b00004Google ScholarThere is no corresponding record for this reference.
- 38Feni, L.; Jütten, L.; Parente, S.; Piarulli, U.; Neundorf, I.; Diaz, D. Cell-Penetrating Peptides Containing 2,5-Diketopiperazine (DKP) Scaffolds as Shuttles for Anti-Cancer Drugs: Conformational Studies and Biological Activity. Chem. Commun. 2020, 56 (42), 5685– 5688, DOI: 10.1039/D0CC01490GGoogle ScholarThere is no corresponding record for this reference.
- 39Panzeri, S.; Arosio, D.; Gazzola, S.; Belvisi, L.; Civera, M.; Potenza, D.; Vasile, F.; Kemker, I.; Ertl, T.; Sewald, N.; Reiser, O.; Piarulli, U. Cyclic RGD and isoDGR Integrin Ligands Containing Cis-2-Amino-1-Cyclopentanecarboxylic (Cis-β-ACPC) Scaffolds. Molecules 2020, 25 (24), 5966, DOI: 10.3390/molecules25245966Google ScholarThere is no corresponding record for this reference.
- 40Zambra, M.; Ranđelović, I.; Talarico, F.; Borbély, A.; Svajda, L.; Tóvári, J.; Mező, G.; Bodero, L.; Colombo, S.; Arrigoni, F.; Fasola, E.; Gazzola, S.; Piarulli, U. Optimizing the Enzymatic Release of MMAE from isoDGR-Based Small Molecule Drug Conjugate by Incorporation of a GPLG-PABC Enzymatically Cleavable Linker. Front. Pharmacol. 2023, 14, 1215694, DOI: 10.3389/fphar.2023.1215694Google ScholarThere is no corresponding record for this reference.
- 41Bodero, L.; Parente, S.; Arrigoni, F.; Klimpel, A.; Neundorf, I.; Gazzola, S.; Piarulli, U. Synthesis and Biological Evaluation of an Iso DGR-Paclitaxel Conjugate Containing a Cell-Penetrating Peptide to Promote Cellular Uptake. Eur. J. Org. Chem. 2021, 2021 (17), 2383– 2387, DOI: 10.1002/ejoc.202100241Google ScholarThere is no corresponding record for this reference.
- 42Kapp, T. G.; Rechenmacher, F.; Neubauer, S.; Maltsev, O. V.; Cavalcanti-Adam, E. A.; Zarka, R.; Reuning, U.; Notni, J.; Wester, H.-J.; Mas-Moruno, C.; Spatz, J.; Geiger, B.; Kessler, H. A Comprehensive Evaluation of the Activity and Selectivity Profile of Ligands for RGD-Binding Integrins. Sci. Rep. 2017, 7 (1), 39805, DOI: 10.1038/srep39805Google ScholarThere is no corresponding record for this reference.
- 43Bochen, A.; Marelli, U. K.; Otto, E.; Pallarola, D.; Mas-Moruno, C.; Di Leva, F. S.; Boehm, H.; Spatz, J. P.; Novellino, E.; Kessler, H.; Marinelli, L. Biselectivity of isoDGR Peptides for Fibronectin Binding Integrin Subtypes Α5β1 and Αvβ6: Conformational Control through Flanking Amino Acids. J. Med. Chem. 2013, 56 (4), 1509– 1519, DOI: 10.1021/jm301221xGoogle ScholarThere is no corresponding record for this reference.
- 44Lau, J.; Jacobson, O.; Niu, G.; Lin, K.-S.; Bénard, F.; Chen, X. Bench to Bedside: Albumin Binders for Improved Cancer Radioligand Therapies. Bioconjugate Chem. 2019, 30 (3), 487– 502, DOI: 10.1021/acs.bioconjchem.8b00919Google ScholarThere is no corresponding record for this reference.
- 45Shi, J.; Jia, B.; Liu, Z.; Yang, Z.; Yu, Z.; Chen, K.; Chen, X.; Liu, S.; Wang, F. 99m Tc-Labeled Bombesin(7–14)NH2 with Favorable Properties for SPECT Imaging of Colon Cancer. Bioconjugate Chem. 2008, 19 (6), 1170– 1178, DOI: 10.1021/bc700471zGoogle ScholarThere is no corresponding record for this reference.
- 46Wang, S.; Sun, W.; Zhao, Y.; McEachern, D.; Meaux, I.; Barrière, C.; Stuckey, J. A.; Meagher, J. L.; Bai, L.; Liu, L.; Hoffman-Luca, C. G.; Lu, J.; Shangary, S.; Yu, S.; Bernard, D.; Aguilar, A.; Dos-Santos, O.; Besret, L.; Guerif, S.; Pannier, P.; Gorge-Bernat, D.; Debussche, L. SAR405838: An Optimized Inhibitor of MDM2-P53 Interaction That Induces Complete and Durable Tumor Regression. Cancer Res. 2014, 74 (20), 5855– 5865, DOI: 10.1158/0008-5472.CAN-14-0799Google ScholarThere is no corresponding record for this reference.
- 47Arrigoni, F.; Prpić, H.; Ferrari, A.; Zambra, M.; Roscilli, G.; Gazzola, S.; Piarulli, U. Design, Synthesis and Preliminary In-Vitro Activity of 6-Hydroxyalkyl β-Carboline Derivatives for the Development of Drug Conjugates Targeting MDM2. Eur. J. Org. Chem. 2024, 27 (47), e202400915 DOI: 10.1002/ejoc.202400915Google ScholarThere is no corresponding record for this reference.
- 48Mabry, A. R.; Singh, A.; Mulrooney, B.; Gorman, J.; Thielbar, A. R.; Wolf, E. R.; Mayo, L. D. Induction of the Mdm2 gene and protein by kinase signaling pathways is repressed by the pVHL tumor suppressor. Proc. Natl. Acad. Sci. U. S. A. 2024, 121 (31), e2400935121 DOI: 10.1073/pnas.2400935121Google ScholarThere is no corresponding record for this reference.
- 49Forte, I.; Indovina, P.; Iannuzzi, C.; Cirillo, D.; Di Marzo, D.; Barone, D.; Capone, F.; Pentimalli, F.; Giordano, A. Targeted Therapy Based on P53 Reactivation Reduces Both Glioblastoma Cell Growth and Resistance to Temozolomide. Int. J. Oncol. 2019, 54, 2189– 2199, DOI: 10.3892/ijo.2019.4788Google ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
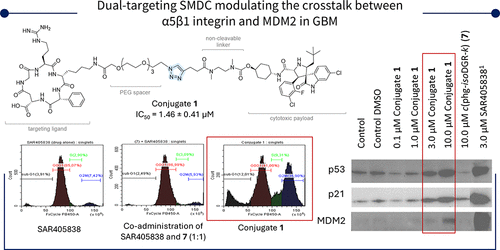
Figure 1

Figure 1. Summary of key studies targeting α5β1 and p53 pathways in GBM. Renner et al. (35) used coadministration of α5β1 antagonists (36) and Nutlin-3a; Merlino et al. (37) developed the first dual-targeting molecule; our work introduces the first small-molecule drug conjugate 1 (SMDC) linking an α5β1 ligand to an MDM2 inhibitor.
Figure 2
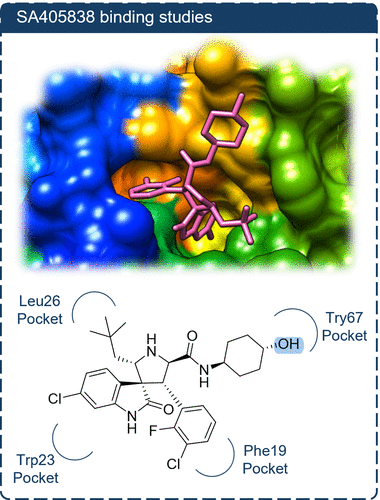
Figure 2. Predicted binding pose of SAR405838 in the MDM2 pocket (AutoDock Vina, based on PDB: 5TRF). The solvent-accessible hydroxyl group on the cyclohexyl ring is indicated as the conjugation site.
Scheme 1
 Scheme 1. Synthesis of Derivative 4a,b
Scheme 1. Synthesis of Derivative 4a,baReagents and conditions: 4-nitrophenylchloroformate, pyridine, CH2Cl2, from −50 to −20 °C, 7 h.
bDMAP, iPr2NEt, CH2Cl2, 16 h.
Figure 3
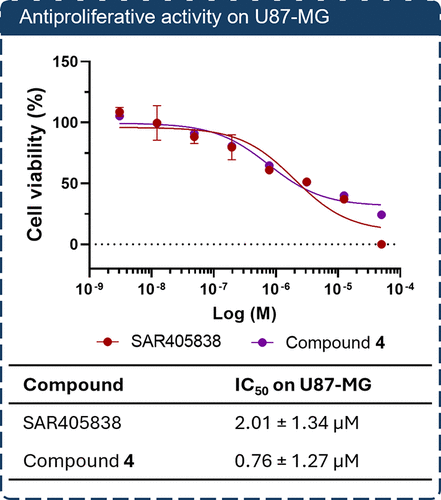
Figure 3. Antiproliferative activity of SAR405838 and compound 4 on U87-MG glioblastoma cells. Cells were treated with increasing concentrations of SAR405838 or compound 4 for 72 h, and cell viability was assessed using the CellTiter-Glo Luminescent Cell Viability Assay. Data represent mean ± SD of three technical replicates from one representative experiment out of at least three independent experiments, all yielding consistent results. IC50 values were calculated by nonlinear regression using GraphPad Prism; reported standard deviations reflect the fitting error of the regression model.
Scheme 2
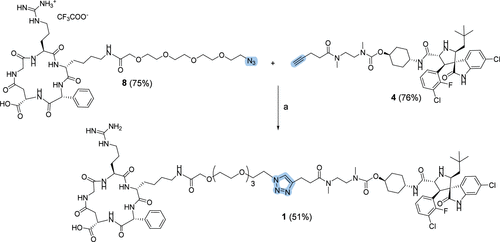 Scheme 2. CuAAC of SMDC 1a
Scheme 2. CuAAC of SMDC 1aaReagents and conditions: Compound 8 (1.1 equiv), compound 4 (1 equiv), CuSO4 × 5H2O (0.5 equiv), sodium ascorbate (0.6 equiv), N2, DMF:H2O = 1:1, 0–25 °C, 16 h.
Figure 4

Figure 4. Antiproliferative activity of SAR405838, cyclo(phg-isoDGR-k) (7), their coadministration, and conjugate 1 in U87-MG glioblastoma cells. Cells were treated for 72 h with increasing concentrations of the indicated compounds, and viability was assessed using the CellTiter-Glo assay. Data represent mean ± SD of three technical replicates from one representative experiment out of at least three independent replicates, all showing consistent trends. IC50 values were calculated by nonlinear regression using GraphPad Prism; reported standard deviations reflect the fitting error of the regression model.
Figure 5
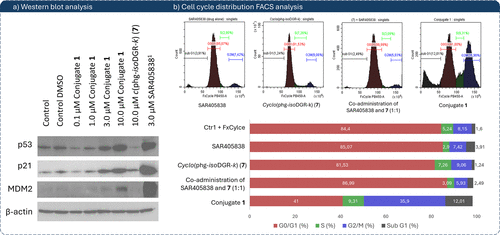
Figure 5. (a) Western blot response-dependent analysis of conjugate 1, cyclo(phg-isoDGR-k) (7), and SAR405838 on p53, p21, and MDM2 expression in U87-MG cells. Concentration of SAR405838 chosen on previously performed dose-dependent Western blot analysis. (b) Cell cycle distribution analysis in U87-MG cells treated with 10 μM of the respective compounds for 24 h. DNA content was assessed by FxCycle staining and analyzed by FACS; histograms shown represent SAR405838 (histogram 1), cyclo(phg-isoDGR-k) (7, histogram 2), 1:1 molar coadministration of 7 and SAR405838 (histogram 3), and compound 1 (histogram 4). Cell population distributions in G0/G1, S, G2/M, and sub-G1 phases are summarized in the bar graph. Full gating strategy, replicate histograms, and control conditions are available in the Supporting Information (Table S3, Figure S2).
References
This article references 49 other publications.
- 1Alirezaei, Z.; Amouheidari, A.; BasirianJahromi, R.; Seyyedhosseini, S.; Hamidi, A. Survival Analysis of Glioblastoma: A Scientometric Perspective. World Neurosurg. 2025, 194, 123476, DOI: 10.1016/j.wneu.2024.11.059There is no corresponding record for this reference.
- 2Stupp, R.; Mason, W. P.; Van Den Bent, M. J.; Weller, M.; Fisher, B.; Taphoorn, M. J. B.; Belanger, K.; Brandes, A. A.; Marosi, C.; Bogdahn, U.; Curschmann, J.; Janzer, R. C.; Ludwin, S. K.; Gorlia, T.; Allgeier, A.; Lacombe, D.; Cairncross, J. G.; Eisenhauer, E.; Mirimanoff, R. O. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352 (10), 987– 996, DOI: 10.1056/NEJMoa043330There is no corresponding record for this reference.
- 3Obrador, E.; Moreno-Murciano, P.; Oriol-Caballo, M.; López-Blanch, R.; Pineda, B.; Gutiérrez-Arroyo, J.; Loras, A.; Gonzalez-Bonet, L.; Martinez-Cadenas, C.; Estrela, J.; Marqués-Torrejón, M. Glioblastoma Therapy: Past, Present and Future. Int. J. Mol. Sci. 2024, 25 (5), 2529, DOI: 10.3390/ijms25052529There is no corresponding record for this reference.
- 4Cruz Da Silva, E.; Mercier, M.-C.; Etienne-Selloum, N.; Dontenwill, M.; Choulier, L. A Systematic Review of Glioblastoma-Targeted Therapies in Phases II, III, IV Clinical Trials. Cancers 2021, 13 (8), 1795, DOI: 10.3390/cancers13081795There is no corresponding record for this reference.
- 5Park, C.-K.; Kim, J. E.; Kim, J. Y.; Song, S. W.; Kim, J. W.; Choi, S. H.; Kim, T. M.; Lee, S.-H.; Kim, I. H.; Park, S.-H. The Changes in MGMT Promoter Methylation Status in Initial and Recurrent Glioblastomas. Transl. Oncol. 2012, 5 (5), 393– IN19, DOI: 10.1593/tlo.12253There is no corresponding record for this reference.
- 6Bocangel, D.; Sengupta, S.; Mitra, S.; Bhakat, K. K. P53-Mediated down-Regulation of the Human DNA Repair Gene O6-Methylguanine-DNA Methyltransferase (MGMT) via Interaction with Sp1 Transcription Factor. Anticancer Res. 2009, 29 (10), 3741– 3750There is no corresponding record for this reference.
- 7Kastenhuber, E. R.; Lowe, S. W. Putting P53 in Context. Cell 2017, 170 (6), 1062– 1078, DOI: 10.1016/j.cell.2017.08.028There is no corresponding record for this reference.
- 8Sullivan, K. D.; Galbraith, M. D.; Andrysik, Z.; Espinosa, J. M. Mechanisms of Transcriptional Regulation by P53. Cell Death Differ. 2018, 25 (1), 133– 143, DOI: 10.1038/cdd.2017.174There is no corresponding record for this reference.
- 9Crespo, I.; Vital, A. L.; Gonzalez-Tablas, M.; Patino, M. D. C.; Otero, A.; Lopes, M. C.; De Oliveira, C.; Domingues, P.; Orfao, A.; Tabernero, M. D. Molecular and Genomic Alterations in Glioblastoma Multiforme. Am. J. Pathol. 2015, 185 (7), 1820– 1833, DOI: 10.1016/j.ajpath.2015.02.023There is no corresponding record for this reference.
- 10England, B.; Huang, T.; Karsy, M. Current Understanding of the Role and Targeting of Tumor Suppressor P53 in Glioblastoma Multiforme. Tumor Biol. 2013, 34 (4), 2063– 2074, DOI: 10.1007/s13277-013-0871-3There is no corresponding record for this reference.
- 11Pouyan, A.; Ghorbanlo, M.; Eslami, M.; Jahanshahi, M.; Ziaei, E.; Salami, A.; Mokhtari, K.; Shahpasand, K.; Farahani, N.; Meybodi, T. E.; Entezari, M.; Taheriazam, A.; Hushmandi, K.; Hashemi, M. Glioblastoma Multiforme: Insights into Pathogenesis, Key Signaling Pathways, and Therapeutic Strategies. Mol. Cancer 2025, 24 (1), 58, DOI: 10.1186/s12943-025-02267-0There is no corresponding record for this reference.
- 12The Cancer Genome Atlas Research Network. Comprehensive Genomic Characterization Defines Human Glioblastoma Genes and Core Pathways. Nature 2008, 455 (7216), 1061– 1068, DOI: 10.1038/nature07385There is no corresponding record for this reference.
- 13Verreault, M.; Schmitt, C.; Goldwirt, L.; Pelton, K.; Haidar, S.; Levasseur, C.; Guehennec, J.; Knoff, D.; Labussière, M.; Marie, Y.; Ligon, A. H.; Mokhtari, K.; Hoang-Xuan, K.; Sanson, M.; Alexander, B. M.; Wen, P. Y.; Delattre, J.-Y.; Ligon, K. L.; Idbaih, A. Preclinical Efficacy of the MDM2 Inhibitor RG7112 in MDM2 -Amplified and TP53 Wild-Type Glioblastomas. Clin. Cancer Res. 2016, 22 (5), 1185– 1196, DOI: 10.1158/1078-0432.CCR-15-1015There is no corresponding record for this reference.
- 14Kussie, P. H.; Gorina, S.; Marechal, V.; Elenbaas, B.; Moreau, J.; Levine, A. J.; Pavletich, N. P. Structure of the MDM2 Oncoprotein Bound to the P53 Tumor Suppressor Transactivation Domain. Science 1996, 274 (5289), 948– 953, DOI: 10.1126/science.274.5289.948There is no corresponding record for this reference.
- 15Vassilev, L. T.; Vu, B. T.; Graves, B.; Carvajal, D.; Podlaski, F.; Filipovic, Z.; Kong, N.; Kammlott, U.; Lukacs, C.; Klein, C.; Fotouhi, N.; Liu, E. A. In Vivo Activation of the P53 Pathway by Small-Molecule Antagonists of MDM2. Science 2004, 303 (5659), 844– 848, DOI: 10.1126/science.1092472There is no corresponding record for this reference.
- 16Villalonga-Planells, R.; Coll-Mulet, L.; Martínez-Soler, F.; Castaño, E.; Acebes, J.-J.; Giménez-Bonafé, P.; Gil, J.; Tortosa, A. Activation of P53 by Nutlin-3a Induces Apoptosis and Cellular Senescence in Human Glioblastoma Multiforme. PLoS One 2011, 6 (4), e18588 DOI: 10.1371/journal.pone.0018588There is no corresponding record for this reference.
- 17Tovar, C.; Rosinski, J.; Filipovic, Z.; Higgins, B.; Kolinsky, K.; Hilton, H.; Zhao, X.; Vu, B. T.; Qing, W.; Packman, K.; Myklebost, O.; Heimbrook, D. C.; Vassilev, L. T. Small-Molecule MDM2 Antagonists Reveal Aberrant P53 Signaling in Cancer: Implications for Therapy. Proc. Natl. Acad. Sci. U. S. A. 2006, 103 (6), 1888– 1893, DOI: 10.1073/pnas.0507493103There is no corresponding record for this reference.
- 18Carvajal, L. A.; Neriah, D. B.; Senecal, A.; Benard, L.; Thiruthuvanathan, V.; Yatsenko, T.; Narayanagari, S.-R.; Wheat, J. C.; Todorova, T. I.; Mitchell, K.; Kenworthy, C.; Guerlavais, V.; Annis, D. A.; Bartholdy, B.; Will, B.; Anampa, J. D.; Mantzaris, I.; Aivado, M.; Singer, R. H.; Coleman, R. A.; Verma, A.; Steidl, U. Dual Inhibition of MDMX and MDM2 as a Therapeutic Strategy in Leukemia. Sci. Transl. Med. 2018, 10 (436), eaao3003 DOI: 10.1126/scitranslmed.aao3003There is no corresponding record for this reference.
- 19Chang, Y. S.; Graves, B.; Guerlavais, V.; Tovar, C.; Packman, K.; To, K.-H.; Olson, K. A.; Kesavan, K.; Gangurde, P.; Mukherjee, A.; Baker, T.; Darlak, K.; Elkin, C.; Filipovic, Z.; Qureshi, F. Z.; Cai, H.; Berry, P.; Feyfant, E.; Shi, X. E.; Horstick, J.; Annis, D. A.; Manning, A. M.; Fotouhi, N.; Nash, H.; Vassilev, L. T.; Sawyer, T. K. Stapled Α-helical Peptide Drug Development: A Potent Dual Inhibitor of MDM2 and MDMX for P53-Dependent Cancer Therapy. Proc. Natl. Acad. Sci. U. S. A. 2013, 110 (36), E3445– E3454, DOI: 10.1073/pnas.1303002110There is no corresponding record for this reference.
- 20Koblish, H. K.; Zhao, S.; Franks, C. F.; Donatelli, R. R.; Tominovich, R. M.; LaFrance, L. V.; Leonard, K. A.; Gushue, J. M.; Parks, D. J.; Calvo, R. R.; Milkiewicz, K. L.; Marugán, J. J.; Raboisson, P.; Cummings, M. D.; Grasberger, B. L.; Johnson, D. L.; Lu, T.; Molloy, C. J.; Maroney, A. C. Benzodiazepinedione Inhibitors of the Hdm2:P53 Complex Suppress Human Tumor Cell Proliferation in Vitro and Sensitize Tumors to Doxorubicin in Vivo. Mol. Cancer Ther. 2006, 5 (1), 160– 169, DOI: 10.1158/1535-7163.MCT-05-0199There is no corresponding record for this reference.
- 21Rew, Y.; Sun, D. Discovery of a Small Molecule MDM2 Inhibitor (AMG 232) for Treating Cancer. J. Med. Chem. 2014, 57 (15), 6332– 6341, DOI: 10.1021/jm500627sThere is no corresponding record for this reference.
- 22Gollner, A.; Weinstabl, H.; Fuchs, J. E.; Rudolph, D.; Garavel, G.; Hofbauer, K. S.; Karolyi-Oezguer, J.; Gmaschitz, G.; Hela, W.; Kerres, N.; Grondal, E.; Werni, P.; Ramharter, J.; Broeker, J.; McConnell, D. B. Targeted Synthesis of Complex Spiro[3 H -indole-3,2′-pyrrolidin]-2(1 H)-ones by Intramolecular Cyclization of Azomethine Ylides: Highly Potent MDM2-P53 Inhibitors. ChemMedChem. 2019, 14 (1), 88– 93, DOI: 10.1002/cmdc.201800617There is no corresponding record for this reference.
- 23Ding, K.; Lu, Y.; Nikolovska-Coleska, Z.; Wang, G.; Qiu, S.; Shangary, S.; Gao, W.; Qin, D.; Stuckey, J.; Krajewski, K.; Roller, P. P.; Wang, S. Structure-Based Design of Spiro-Oxindoles as Potent, Specific Small-Molecule Inhibitors of the MDM2-p53 Interaction. J. Med. Chem. 2006, 49 (12), 3432– 3435, DOI: 10.1021/jm051122aThere is no corresponding record for this reference.
- 24Wang, W.; Qin, J.-J.; Voruganti, S.; Srivenugopal, K. S.; Nag, S.; Patil, S.; Sharma, H.; Wang, M.-H.; Wang, H.; Buolamwini, J. K.; Zhang, R. The Pyrido[b]Indole MDM2 Inhibitor SP-141 Exerts Potent Therapeutic Effects in Breast Cancer Models. Nat. Commun. 2014, 5 (1), 5086, DOI: 10.1038/ncomms6086There is no corresponding record for this reference.
- 25Chauhan, S. S.; Singh, A. K.; Meena, S.; Lohani, M.; Singh, A.; Arya, R. K.; Cheruvu, S. H.; Sarkar, J.; Gayen, J. R.; Datta, D.; Chauhan, P. M. S. Synthesis of Novel β-Carboline Based Chalcones with High Cytotoxic Activity against Breast Cancer Cells. Bioorg. Med. Chem. Lett. 2014, 24 (13), 2820– 2824, DOI: 10.1016/j.bmcl.2014.04.109There is no corresponding record for this reference.
- 26Berberich, A.; Kessler, T.; Thomé, C. M.; Pusch, S.; Hielscher, T.; Sahm, F.; Oezen, I.; Schmitt, L.-M.; Ciprut, S.; Hucke, N.; Ruebmann, P.; Fischer, M.; Lemke, D.; Breckwoldt, M. O.; Von Deimling, A.; Bendszus, M.; Platten, M.; Wick, W. Targeting Resistance against the MDM2 Inhibitor RG7388 in Glioblastoma Cells by the MEK Inhibitor Trametinib. Clin. Cancer Res. 2019, 25 (1), 253– 265, DOI: 10.1158/1078-0432.CCR-18-1580There is no corresponding record for this reference.
- 27Her, N.-G.; Oh, J.-W.; Oh, Y. J.; Han, S.; Cho, H. J.; Lee, Y.; Ryu, G. H.; Nam, D.-H. Potent Effect of the MDM2 Inhibitor AMG232 on Suppression of Glioblastoma Stem Cells. Cell Death Dis. 2018, 9 (8), 792, DOI: 10.1038/s41419-018-0825-1There is no corresponding record for this reference.
- 28Punganuru, S. R.; Arutla, V.; Zhao, W.; Rajaei, M.; Deokar, H.; Zhang, R.; Buolamwini, J. K.; Srivenugopal, K. S.; Wang, W. Targeted Brain Tumor Therapy by Inhibiting the MDM2 Oncogene: In Vitro and In Vivo Antitumor Activity and Mechanism of Action. Cells 2020, 9 (7), 1592, DOI: 10.3390/cells9071592There is no corresponding record for this reference.
- 29Wang, Z.; Zhang, S.; Irakoze, L. M.; Zhao, Y. Targeting P53 Activation: Recent Therapeutic Advances in Cancer and Diabetic Macular Edema. Eur. J. Med. Chem. 2025, 297, 117909, DOI: 10.1016/j.ejmech.2025.117909There is no corresponding record for this reference.
- 30Etienne-Selloum, N.; Prades, J.; Bello-Roufai, D.; Boone, M.; Sevestre, H.; Trudel, S.; Caillet, P.; Coutte, A.; Desenclos, C.; Constans, J.-M.; Martin, S.; Choulier, L.; Chauffert, B.; Dontenwill, M. Expression Analysis of Α5 Integrin Subunit Reveals Its Upregulation as a Negative Prognostic Biomarker for Glioblastoma. Pharmaceuticals 2021, 14 (9), 882, DOI: 10.3390/ph14090882There is no corresponding record for this reference.
- 31Schaffner, F.; Ray, A.; Dontenwill, M. Integrin Α5β1, the Fibronectin Receptor, as a Pertinent Therapeutic Target in Solid Tumors. Cancers 2013, 5 (1), 27– 47, DOI: 10.3390/cancers5010027There is no corresponding record for this reference.
- 32Renner, G.; Noulet, F.; Mercier, M.-C.; Choulier, L.; Etienne-Selloum, N.; Gies, J.-P.; Lehmann, M.; Lelong-Rebel, I.; Martin, S.; Dontenwill, M. Expression/Activation of Α5β1 Integrin Is Linked to the β-Catenin Signaling Pathway to Drive Migration in Glioma Cells. Oncotarget 2016, 7 (38), 62194– 62207, DOI: 10.18632/oncotarget.11552There is no corresponding record for this reference.
- 33Sani, S.; Pallaoro, N.; Messe, M.; Bernhard, C.; Etienne-Selloum, N.; Kessler, H.; Marinelli, L.; Entz-Werle, N.; Foppolo, S.; Martin, S.; Reita, D.; Dontenwill, M. Temozolomide-Acquired Resistance Is Associated with Modulation of the Integrin Repertoire in Glioblastoma, Impact of Α5β1 Integrin. Cancers 2022, 14 (2), 369, DOI: 10.3390/cancers14020369There is no corresponding record for this reference.
- 34Janouskova, H.; Maglott, A.; Leger, D. Y.; Bossert, C.; Noulet, F.; Guerin, E.; Guenot, D.; Pinel, S.; Chastagner, P.; Plenat, F.; Entz-Werle, N.; Lehmann-Che, J.; Godet, J.; Martin, S.; Teisinger, J.; Dontenwill, M. Integrin Α5β1 Plays a Critical Role in Resistance to Temozolomide by Interfering with the P53 Pathway in High-Grade Glioma. Cancer Res. 2012, 72 (14), 3463– 3470, DOI: 10.1158/0008-5472.CAN-11-4199There is no corresponding record for this reference.
- 35Renner, G.; Janouskova, H.; Noulet, F.; Koenig, V.; Guerin, E.; Bär, S.; Nuesch, J.; Rechenmacher, F.; Neubauer, S.; Kessler, H.; Blandin, A.-F.; Choulier, L.; Etienne-Selloum, N.; Lehmann, M.; Lelong-Rebel, I.; Martin, S.; Dontenwill, M. Integrin Α5β1 and P53 Convergent Pathways in the Control of Anti-Apoptotic Proteins PEA-15 and Survivin in High-Grade Glioma. Cell Death Differ. 2016, 23 (4), 640– 653, DOI: 10.1038/cdd.2015.131There is no corresponding record for this reference.
- 36Ray, A.-M.; Schaffner, F.; Janouskova, H.; Noulet, F.; Rognan, D.; Lelong-Rebel, I.; Choulier, L.; Blandin, A.-F.; Lehmann, M.; Martin, S.; Kapp, T.; Neubauer, S.; Rechenmacher, F.; Kessler, H.; Dontenwill, M. Single Cell Tracking Assay Reveals an Opposite Effect of Selective Small Non-Peptidic Α5β1 or Αvβ3/Β5 Integrin Antagonists in U87MG Glioma Cells. Biochim. Biophys. Acta BBA - Gen. Subj. 2014, 1840 (9), 2978– 2987, DOI: 10.1016/j.bbagen.2014.04.024There is no corresponding record for this reference.
- 37Merlino, F.; Daniele, S.; La Pietra, V.; Di Maro, S.; Di Leva, F. S.; Brancaccio, D.; Tomassi, S.; Giuntini, S.; Cerofolini, L.; Fragai, M.; Luchinat, C.; Reichart, F.; Cavallini, C.; Costa, B.; Piccarducci, R.; Taliani, S.; Da Settimo, F.; Martini, C.; Kessler, H.; Novellino, E.; Marinelli, L. Simultaneous Targeting of RGD-Integrins and Dual Murine Double Minute Proteins in Glioblastoma Multiforme. J. Med. Chem. 2018, 61 (11), 4791– 4809, DOI: 10.1021/acs.jmedchem.8b00004There is no corresponding record for this reference.
- 38Feni, L.; Jütten, L.; Parente, S.; Piarulli, U.; Neundorf, I.; Diaz, D. Cell-Penetrating Peptides Containing 2,5-Diketopiperazine (DKP) Scaffolds as Shuttles for Anti-Cancer Drugs: Conformational Studies and Biological Activity. Chem. Commun. 2020, 56 (42), 5685– 5688, DOI: 10.1039/D0CC01490GThere is no corresponding record for this reference.
- 39Panzeri, S.; Arosio, D.; Gazzola, S.; Belvisi, L.; Civera, M.; Potenza, D.; Vasile, F.; Kemker, I.; Ertl, T.; Sewald, N.; Reiser, O.; Piarulli, U. Cyclic RGD and isoDGR Integrin Ligands Containing Cis-2-Amino-1-Cyclopentanecarboxylic (Cis-β-ACPC) Scaffolds. Molecules 2020, 25 (24), 5966, DOI: 10.3390/molecules25245966There is no corresponding record for this reference.
- 40Zambra, M.; Ranđelović, I.; Talarico, F.; Borbély, A.; Svajda, L.; Tóvári, J.; Mező, G.; Bodero, L.; Colombo, S.; Arrigoni, F.; Fasola, E.; Gazzola, S.; Piarulli, U. Optimizing the Enzymatic Release of MMAE from isoDGR-Based Small Molecule Drug Conjugate by Incorporation of a GPLG-PABC Enzymatically Cleavable Linker. Front. Pharmacol. 2023, 14, 1215694, DOI: 10.3389/fphar.2023.1215694There is no corresponding record for this reference.
- 41Bodero, L.; Parente, S.; Arrigoni, F.; Klimpel, A.; Neundorf, I.; Gazzola, S.; Piarulli, U. Synthesis and Biological Evaluation of an Iso DGR-Paclitaxel Conjugate Containing a Cell-Penetrating Peptide to Promote Cellular Uptake. Eur. J. Org. Chem. 2021, 2021 (17), 2383– 2387, DOI: 10.1002/ejoc.202100241There is no corresponding record for this reference.
- 42Kapp, T. G.; Rechenmacher, F.; Neubauer, S.; Maltsev, O. V.; Cavalcanti-Adam, E. A.; Zarka, R.; Reuning, U.; Notni, J.; Wester, H.-J.; Mas-Moruno, C.; Spatz, J.; Geiger, B.; Kessler, H. A Comprehensive Evaluation of the Activity and Selectivity Profile of Ligands for RGD-Binding Integrins. Sci. Rep. 2017, 7 (1), 39805, DOI: 10.1038/srep39805There is no corresponding record for this reference.
- 43Bochen, A.; Marelli, U. K.; Otto, E.; Pallarola, D.; Mas-Moruno, C.; Di Leva, F. S.; Boehm, H.; Spatz, J. P.; Novellino, E.; Kessler, H.; Marinelli, L. Biselectivity of isoDGR Peptides for Fibronectin Binding Integrin Subtypes Α5β1 and Αvβ6: Conformational Control through Flanking Amino Acids. J. Med. Chem. 2013, 56 (4), 1509– 1519, DOI: 10.1021/jm301221xThere is no corresponding record for this reference.
- 44Lau, J.; Jacobson, O.; Niu, G.; Lin, K.-S.; Bénard, F.; Chen, X. Bench to Bedside: Albumin Binders for Improved Cancer Radioligand Therapies. Bioconjugate Chem. 2019, 30 (3), 487– 502, DOI: 10.1021/acs.bioconjchem.8b00919There is no corresponding record for this reference.
- 45Shi, J.; Jia, B.; Liu, Z.; Yang, Z.; Yu, Z.; Chen, K.; Chen, X.; Liu, S.; Wang, F. 99m Tc-Labeled Bombesin(7–14)NH2 with Favorable Properties for SPECT Imaging of Colon Cancer. Bioconjugate Chem. 2008, 19 (6), 1170– 1178, DOI: 10.1021/bc700471zThere is no corresponding record for this reference.
- 46Wang, S.; Sun, W.; Zhao, Y.; McEachern, D.; Meaux, I.; Barrière, C.; Stuckey, J. A.; Meagher, J. L.; Bai, L.; Liu, L.; Hoffman-Luca, C. G.; Lu, J.; Shangary, S.; Yu, S.; Bernard, D.; Aguilar, A.; Dos-Santos, O.; Besret, L.; Guerif, S.; Pannier, P.; Gorge-Bernat, D.; Debussche, L. SAR405838: An Optimized Inhibitor of MDM2-P53 Interaction That Induces Complete and Durable Tumor Regression. Cancer Res. 2014, 74 (20), 5855– 5865, DOI: 10.1158/0008-5472.CAN-14-0799There is no corresponding record for this reference.
- 47Arrigoni, F.; Prpić, H.; Ferrari, A.; Zambra, M.; Roscilli, G.; Gazzola, S.; Piarulli, U. Design, Synthesis and Preliminary In-Vitro Activity of 6-Hydroxyalkyl β-Carboline Derivatives for the Development of Drug Conjugates Targeting MDM2. Eur. J. Org. Chem. 2024, 27 (47), e202400915 DOI: 10.1002/ejoc.202400915There is no corresponding record for this reference.
- 48Mabry, A. R.; Singh, A.; Mulrooney, B.; Gorman, J.; Thielbar, A. R.; Wolf, E. R.; Mayo, L. D. Induction of the Mdm2 gene and protein by kinase signaling pathways is repressed by the pVHL tumor suppressor. Proc. Natl. Acad. Sci. U. S. A. 2024, 121 (31), e2400935121 DOI: 10.1073/pnas.2400935121There is no corresponding record for this reference.
- 49Forte, I.; Indovina, P.; Iannuzzi, C.; Cirillo, D.; Di Marzo, D.; Barone, D.; Capone, F.; Pentimalli, F.; Giordano, A. Targeted Therapy Based on P53 Reactivation Reduces Both Glioblastoma Cell Growth and Resistance to Temozolomide. Int. J. Oncol. 2019, 54, 2189– 2199, DOI: 10.3892/ijo.2019.4788There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsmedchemlett.5c00669.
The PDF file includes general procedure for the chemical synthesis of the compounds, NMR spectra, HPLC purity analysis, HRMS/LC–MS spectra, and all the procedures of the performed biological assays, including data of α5β1 integrin expression levels on different cancer cell lines and FACS analysis data (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



