
About the Cover:
Aqueous biphasic systems based on ethyl lactate and organic salts efficiently recover riboflavin (vitamin B2), revealing solubility-driven partitioning and offering a sustainable route for circular economy applications.
View the article.Reviews

Solubility Data of CO2 in the MDEA+PZ Solvent: A Review of Experimental Data, Setups, and Procedures
Camilla Barbieri - and
Stefania Moioli *
This publication is Open Access under the license indicated. Learn More
Chemical absorption is one of the most applied and efficient methods for CO2 removal, and it is primarily employed in the processes related to the production of energy (natural gas, power plants, etc.). This work is the first review focusing on bibliographic research related to experimental data of CO2 solubility in the MDEA+PZ aqueous solution, which is of interest for the CO2 removal from several types of gaseous streams in important industrial and environmental processes. An in-depth review of the available experimental setups and procedures for collecting points of CO2 solubility in the MDEA+PZ solvent is presented. The main methods to measure and compute the solubility of the acid gas in the solvent are analyzed, and the experimental points from all the sources are detailed and compared, considering different values of temperature, pressure, CO2 loading, and composition of the solvent, as for the content of MDEA and that of PZ.

Hydrophobic Deep Eutectic Solvents: Synthesis, Properties, Applications, and Future Directions
Aswin Falahudin - ,
Numpon Insin - , and
Mohammad Mansoob Khan *
Hydrophobic deep eutectic solvents (HDES) have emerged as a transformative frontier in green chemistry owing to their exceptional properties, which include biodegradability, thermal stability, low vapor pressure, and the ability to produce high-purity products at low temperatures. In the field of separation technologies, HDES provide unparalleled advantages as a sustainable alternative to conventional hazardous organic solvents. The versatility of HDES is harnessed across diverse applications, from food analysis to environmental remediation. Their remarkable separation performance provides tailored approaches for specific applications. Momentum is growing for green chemistry research, and HDES are increasingly present at the intersection of sustainability and innovation. The development of HDES is expected to transform the future of sustainable materials science and drive deeper insights into ecoconscious technological solutions.
Thermophysical and Thermochemical Properties
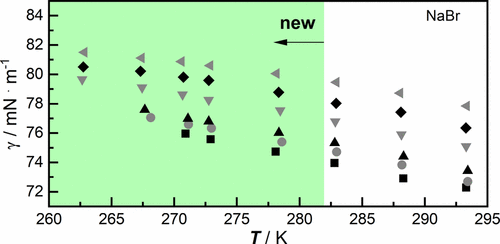
Surface Tension and Surface Thermodynamics of Aqueous Inorganic Salt Solutions in the Atmospherically Relevant Temperature Range
Alexandra Giermann - and
Manuela Reichelt *
This publication is Open Access under the license indicated. Learn More
ACS Editors' Choice® is a collection designed to feature scientific articles of broad public interest. Read the latest articles
The surface tension γ of aqueous solutions of NaCl, NaBr, NaI, LiCl, KCl, MgCl2, and Na2SO4 (0.1 mol·kg–1 to 5 mol·kg–1 in maximum) was investigated within the atmospheric relevant temperature range of 263.15 to 293.15 K. The measured densities ρ of the aqueous inorganic salt solutions between 278.15 and 293.15 K are also reported. From experimental data the excess surface tension Δγ, the concentration (as molality m) and temperature derivatives, (dγ/dm)T, p and (dγ/dT)p, m as well as thermodynamic quantities of surface formation such as surface excess entropy Δsσ, surface excess Helmholtz energy Δaσ, surface excess energy Δuσ, and surface excess enthalpy Δhσ were estimated. The surface excess entropy decreases slightly with increasing molality of the inorganic salt, which shows a weak order dependence of the water molecules on the salt concentration. The surface tension data and thermodynamic quantities complement existing data sets especially at temperatures below 283.15 K.
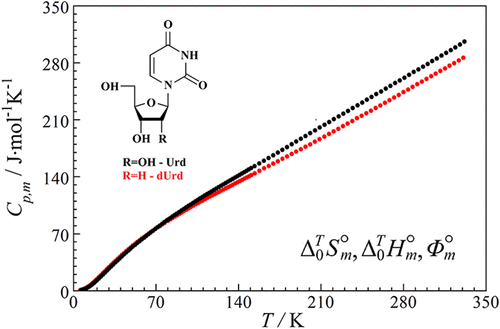
Low-Temperature Thermodynamic Properties of Nucleosides: Uridine and Deoxyuridine
Michael A. Bespyatov *
Precision data on the heat capacity in the range 6–330 K of crystalline synthetic samples of nucleosides, uridine (C9H12N2O6; CAS Number: 58–96–8; fraction purity: 0.999), and deoxyuridine (C9H12N2O5; CAS Number: 951–78–0; fraction purity: 0.999), are provided herein. This was the first time that the heat capacity of the substances was measured by vacuum adiabatic calorimetry. The heat capacity was found to have no anomalies indicating phase transitions in the tested temperature range. The heat capacity experimental data were used to calculate the entropy, enthalpy, and reduced Gibbs energy for uridine and deoxyuridine in the range 0–330 K. The obtained results are important for understanding the energetics of biochemical processes involving nucleosides, assessing their pharmacological properties and expanding the possibilities of their application.
Vapor-Liquid Equilibria and Supercritical Fluid Equilibria
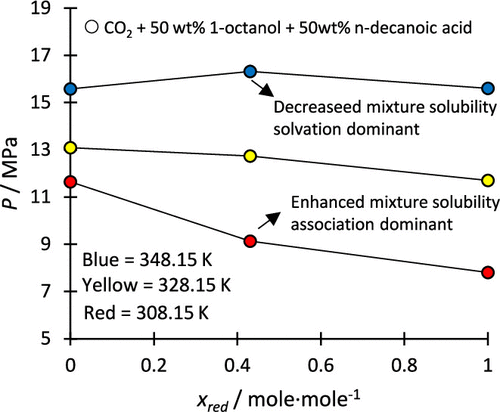
High-Pressure Bubble- and Dew-Point Data for Systems Containing CO2 + (C8/C10 1-Alcohol + C8/C10/C12n-Carboxylic Acid) Systems
Susanna H. Du Plessis - ,
Ashmika Balaram - , and
Cara E. Schwarz *
This publication is Open Access under the license indicated. Learn More
Six new high-pressure bubble-and dew-point data sets have been experimentally measured for ternary systems containing carbon dioxide (the solvent), C8 or C10 1-alcohols, and C8 or C10 or C12 n-carboxylic acids (the solutes). These data were measured at a fixed reduced solute mass fraction of 0.5000 between a total solute fraction of 0.020 to 0.640 at temperatures between 308.15 and 358.15 K. These systems displayed complex solute + solute interactions due to the capacity of the solutes to both self- and cross-associate, leading to competing interactions within the mixture. The mixtures displayed both reduced mixture solubility (mostly at higher temperatures) and enhanced mixture solubility (mostly at lower temperatures) indicating that both association and solvation interactions occur within these systems and that the interactions are temperature dependent. The CO2 + 1-octanol + n-decanoic acid and CO2 + 1-decanol + n-dodecanoic acid systems were identified as island systems, indicating that the unlike or solvation interactions are favored in these systems. The RK-Aspen model is shown to provide an adequate description of the phase behavior data including only solute + solvent binary interaction parameters, although the description is poorer at lower temperatures and in the mixture critical region.
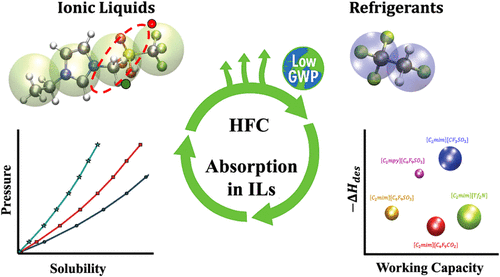
Molecular Modeling of HFC Separation in Fluorinated Ionic Liquids: A SAFT-VR Mie Study
Isaías Huenuvil-Pacheco - ,
Andrés Mejía - , and
Fèlix Llovell *
This publication is Open Access under the license indicated. Learn More
Given the critical role of hydrofluorocarbons (HFCs) in the refrigeration industry, their recovery is crucial to promoting circular economy strategies and reducing greenhouse gas emissions. In this work, the SAFT-VR Mie equation of state is applied to describe the solubility of R32, R125, and R134a in ionic liquids (ILs) with varying degrees of fluorination, which are proposed as potential absorbents for gas separation. ILs are treated as associating species, with multiple sites accounting for charge delocalization. The model provides an excellent description of density and viscosity, the latter obtained through the Helmholtz scaling theory. In addition, the effect of polarity is explicitly accounted for in the HFCs. As a result, their solubility in ILs is quantitatively reproduced using a single, temperature-independent binary parameter, ensuring a strong predictive capability. Furthermore, the working capacity and competitive selectivity of the components in commercial R410A and R407F blends are predicted, revealing significant differences in performance depending on the solvent composition and fluorination level. Additional properties, such as regeneration enthalpy and viscosity, are also evaluated to identify the most promising ILs for refrigerant recovery. Overall, this study demonstrates the capability of SAFT-VR Mie as a robust molecular-based tool for solvent screening in sustainable HFC separation technologies.
Liquid-Liquid Equilibria and Vapor-Liquid-Liquid Equilibria
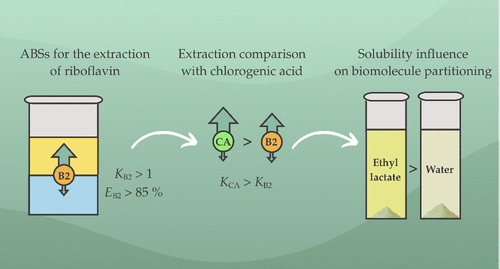
Partitioning of Riboflavin in Sustainable Aqueous Biphasic Systems and the Influence of Solubility
Gonçalo Perestrelo - ,
Pedro Velho *- , and
Eugénia A. Macedo *
This publication is Open Access under the license indicated. Learn More
Developing sustainable extraction techniques for the recovery of bioactive substances from biowaste is key toward promoting a circular economy and reducing the environmental impact in industrial processes. This work employs aqueous biphasic systems (ABSs) based on ethyl lactate (EL), water, and organic salts (Na2Tartrate, NaKTartrate, and K3Citrate) for the extraction of riboflavin (B2), a vitamin commonly found in biowaste. B2 partitioning, evaluated through the partition coefficient (K) and extraction efficiency (E), was deemed successful for all ABSs, with the Na2Tartrate-based ABS presenting the best results, yielding K = 8.23 ± 0.07 and E = (87.5 ± 0.8)% for the longest tie-line. Published data on chlorogenic acid (CA) were used for comparison, with CA showing similar partitioning trends but consistently higher performance. To assess this difference, solubility studies were conducted for both biomolecules at 283.15, 298.15, and 313.15 K. The solubility of both species in EL was higher than in water, supporting the preferential partition to the top phase. Moreover, CA presented a higher solubility in EL than B2, which is consistent with its superior partition results. Last, the van’t Hoff equation was applied to the solubility data, though the solubility of B2 in EL did not align with this model.

Reactive Extraction of Isobutyric Acid from Water Using Trioctyl Amine in Different Toxic and Nontoxic Diluents
Sourav Mukherjee - and
Basudeb Munshi *
In this work, the reactive extraction of isobutyric acid (IBA) is reported for the first time over the temperature range of 298–318 K. Trioctylamine (TOA) in eight different diluents was studied. For reducing the harmful effect of TOA on living microorganisms in the fermentation broth, nontoxic sunflower and soybean oils were used along with six other petrochemical-based diluents. For physical extraction, the distribution coefficient (KDdil) appeared reasonably high. TOA enhanced the distribution constants. In reactive extraction, the extraction efficiencies were 82–93% for decanol, 80–88% for octanol, 78–87% for hexanol, 76–86% for MIBK, 75–85% for toluene, 75–83% for petroleum ether, 73–82% for sunflower oil, and 72–81% for soybean oil. The sequence of the KDoverall was decanol > octanol > hexanol > MIBK > toluene > petroleum ether > sunflower oil > soybean oil. The obtained KDchem/KDphy > 1.0 confirmed the dominance of the chemical extraction over the physical extraction. The complexation reaction stoichiometry and equilibrium reaction constants were estimated by using the differential evolution technique. Different thermodynamic parameters like enthalpy and entropy changes were determined and analyzed. (ΔH)mass tr values showed that mixing of the aqueous and organic phases is the most difficult in sunflower oil and easiest in decanol.
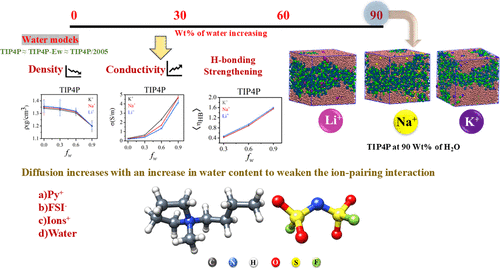
Understanding Transport and Structural Properties in Py–FSI–Alkali Metal Ion Mixtures: Role of Ion Identity and TIP4P-Based Water Potential
Puvvada Roja Satvika - ,
Preeti Nanda Sahu - ,
Suman Das *- , and
Anik Sen *
This study investigates the dynamical and structural behavior of ionic liquid–alkali metal ion mixtures in aqueous solutions with varying water content using three different water models: TIP4P, TIP4P-Ew, and TIP4P/2005. We analyze several key properties, including system density, ion diffusion, hydrogen bonding (H-bonds), ionic conductivity, and radial distribution functions (RDFs), to understand how water content affects ion-pair interactions and microscopic structuring. Density decreases by ∼10% as the water mole fraction increases to 0.9. Hydrogen bond analysis reveals a nearly 3-fold increase in the number of water–water H-bonds, emphasizing the role of water in forming structured networks. Ion diffusion increases, ranging from 10 to 50 times with higher water content, particularly for larger ions like K+. The results show that as the water content increases, ion-pair interactions (such as Py+–FSI– and Ion+–FSI–) weaken, particularly at higher water concentrations (0.9 fw). While the three water models show consistent trends in density, dynamics, and structural properties, quantitative differences exist, highlighting the sensitivity of results to model choice. These findings have implications for the understanding of electrolyte solutions, solvent engineering, and the modeling of biological and chemical systems in solution.
Solid-Solid Equilibria and Solid-Fluid Equilibria
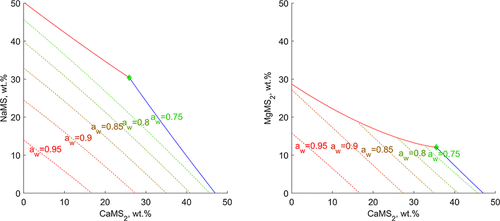
Phase Equilibria in Water–Calcium Methanesulfonate–Sodium (Magnesium) Methanesulfonate Systems: Experiment and Modeling
Ekaterina V. Belova *- ,
Daniil E. Krasnoiarov - ,
Ivan V. Mikheev - , and
Irina A. Uspenskaya
Novel solubility (SLE) and vapor–liquid phase equilibria data were obtained for ternary aqua systems containing calcium methanesulfonate and sodium (or magnesium) methanesulfonate. The isothermal solubility method was used to obtain an isothermal cross-section at 298.15 K for the Ca(CH3SO3)2–NaCH3SO3–H2O and the Ca(CH3SO3)2–Mg(CH3SO3)2–H2O systems. As expected, there are neither solid solutions nor double salts in both systems. The Mg(CH3SO3)2·12H2O hydrate is the only solid phase containing magnesium in the Ca(CH3SO3)2–Mg(CH3SO3)2–H2O system at 298.15 K. Water activity was determined at 298.15–323.15 K to expand the temperature range for NaCH3SO3–H2O by two methods (static vapor pressure measurement and hygrometer). Water vapor pressure was measured in the 288.15–308.15 K range in both ternary systems to calculate water activity. The Zdanovskii rule is applicable to both ternary systems in the composition range under investigation. The Pitzer–Simonson–Clegg model is used for liquid-phase modeling. Binary parameters for NaCH3SO3–H2O were evaluated for the first time, as well as the solubility constant for NaCH3SO3, in the 250–323 K temperature range. Ternary parameters of the PSC model were evaluated for the first time, too, needed for a correct solubility prediction. The water activity was predicted with a higher accuracy without ternary parameters.
Additions and Corrections
Correction to “Binary Mixtures of n-Alkylbenzenes and Pentadecane: Densities, Speeds of Sound, and Viscosities within the Range of 288.15 and 333.15 K and at 0.1 MPa”
Dianne J. Luning Prak *- and
Jim S. Cowart
This publication is Open Access under the license indicated. Learn More
Mastheads
Issue Editorial Masthead
This publication is free to access through this site. Learn More
Issue Publication Information
This publication is free to access through this site. Learn More
