Surface Chemistry in the Initial Stages of Titanium Nitride Atomic Layer Deposition Using Operando Ambient Pressure X-ray Photoelectron SpectroscopyClick to copy article linkArticle link copied!
- Pamburayi Mpofu*Pamburayi Mpofu*[email protected]Department of Physics, Chemistry and Biology, Linköping University, 581 83 Linköping, SwedenMore by Pamburayi Mpofu
- Peggy Bagherzadeh TabriziPeggy Bagherzadeh TabriziDepartment of Physics, Chemistry and Biology, Linköping University, 581 83 Linköping, SwedenMore by Peggy Bagherzadeh Tabrizi
- Houyem HafdiHouyem HafdiDepartment of Physics, Chemistry and Biology, Linköping University, 581 83 Linköping, SwedenMore by Houyem Hafdi
- Premrudee PromdetPremrudee PromdetDepartment of Physics, Chemistry and Biology, Linköping University, 581 83 Linköping, SwedenMore by Premrudee Promdet
- Jonas Lauridsen
- Oscar Alm
- Tommy Larsson
- Rosemary JonesRosemary JonesMAX IV Laboratory, Lund University, Box 118, 221 00 Lund, SwedenDivision of Synchrotron Radiation Research, Department of Physics, Lund University, Box 118, 221 00 Lund, SwedenMore by Rosemary Jones
- Esko Kokkonen
- Joachim SchnadtJoachim SchnadtMAX IV Laboratory, Lund University, Box 118, 221 00 Lund, SwedenDivision of Synchrotron Radiation Research, Department of Physics, Lund University, Box 118, 221 00 Lund, SwedenNanoLund, Lund University, Box 118, 221 00 Lund, SwedenMore by Joachim Schnadt
- Henrik Pedersen*Henrik Pedersen*[email protected]Department of Physics, Chemistry and Biology, Linköping University, 581 83 Linköping, SwedenMore by Henrik Pedersen
Abstract
Studies of the surface chemistry of the first few cycles of atomic layer deposition (ALD) using in situ and time-resolved operando techniques are attractive for realizing, understanding, and obtaining true mechanistic information during the deposition. However, the latter techniques are yet to be applied to ALD of metal nitrides. Here, we present a surface-chemistry investigation through a time-resolved ambient pressure X-ray photoelectron spectroscopy (APXPS) study of the initial growth of titanium nitride (TiN). The Ti 2p, O 1s, N 1s, C 1s, and Si 2p core-level spectra recorded at different stages during the deposition show that chemisorption occurs immediately on the silicon dioxide surface due to the interaction of tetrakis(dimethylamido)titanium(IV) (TDMAT) with the surface. A delay in nucleation on the TDMAT-terminated surface was observed during the NH3 pulse. The intensity of the Ti 2p and N 1s core levels began to increase after four ALD cycles, showing that the surface was coated with Ti and N atoms and no Si signals were observed with time. The results show that ligand exchange reactions take place before transamination reactions. This was verified using the periodic changes in the intensity and peak positions of the above-mentioned spectra and complemented by residual gas analysis using mass spectrometry. These results can provide insights into the ALD surface growth of not only TiN but also other metal nitrides.
This publication is licensed under
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
License Summary*
You are free to share(copy and redistribute) this article in any medium or format and to adapt(remix, transform, and build upon) the material for any purpose, even commercially within the parameters below:
Creative Commons (CC): This is a Creative Commons license.
Attribution (BY): Credit must be given to the creator.
*Disclaimer
This summary highlights only some of the key features and terms of the actual license. It is not a license and has no legal value. Carefully review the actual license before using these materials.
1. Introduction
2. Methods
2.1. APXPS
2.2. Film Deposition
2.3. Data Analysis
2.4. Residual Gas Analysis
3. Results
First ALD Half-Cycle: TDMAT
Figure 1
Figure 1. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXP spectra measured during the TDMAT half-cycle at 250 °C while f) shows the peak position evolution of the main peak regions in C 1s and Ti 2p plots as a function of time during the TDMAT pulse. The spectra were measured using a photon energy of 680 eV. The color scale shows how the intensity of the indicated core levels changes with time. For ease of visibility, the Ti 2p, N 1s, and C 1s intensities were multiplied by factors, as indicated, relative to the Si 2p intensity.
Second ALD Half-Cycle: NH3
Figure 2
Figure 2. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXPS spectra measured during the NH3 half-cycle at 250 °C while f) shows the center of gravity plot as a function of time during the first and second NH3 pulse trains to visualize the dynamic changes. The spectra were measured using a photon energy of 680 eV. The color scale shows how the intensity of the indicated core levels changes with time. For ease of visibility, the Ti 2p, N 1s, and C 1s intensities were multiplied by factors, as indicated, relative to the Si 2p intensity.
Figure 3
Figure 3. a) N 1s and b) C 1s intensity evolutions extracted at defined time points from the image plots in Figure 2c) and d), respectively, allowing a more direct comparison of the temporal evolution indicating that no ligand removal is evident in the second half-cycle through NH3-assisted ligand exchange surface reactions.
Three Additional ALD Cycles: 3 × (TDMAT + NH3)
Figure 4
Figure 4. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXP spectra measured during three additional cycles at 250 °C, while f) and g) show the peak position evolution of the main peak regions in Ti 2p and C 1s plots as a function of time during the ALD cycles. The color scale shows how the intensity of the indicated core levels changes over time. The spectra were measured using a photon energy of 680 eV. The time at which each ALD half-cycle starts is marked by horizontal dashed lines and denoted TDMAT or NH3 to symbolize when each corresponding half-cycle starts and ends. Note that this is a continuation of Figures 1 and 2, hence precursor pulsing starts at TDMAT (2). We can clearly see the five short pulses of TDMAT in each of the TDMAT pulse trains as drops in Ti 2p intensity (this is superimposed on the raster-induced intensity changes). The dotted red line on the Ti 2p map shows the shift toward lower binding energies during the three ALD cycles with time, indicating a shift from oxide toward nitride character in the films. The oscillating behavior shown on the maps has nothing to do with the ALD process itself but emanates from the continuous movement of the sample during analysis as mentioned in the Methods section. For ease of visibility, the O 1s intensity was multiplied by a factor of 2 relative to the Si 2p intensity.
Figure 5
Figure 5. APXPS core level scans of the a) O 1s, b) Ti 2p, c) N 1s, and d) C 1s lines at 250 °C from an initial substrate surface until after the three additional ALD cycles. The spectra were measured using photon energies of 680 eV for the O 1s and Ti 2p lines, while 550 and 410 eV were used in the measurement of the N 1s and C 1s spectra, respectively. To make incorporation of N and Ti more visible and easier to follow, the corresponding survey spectrum for each of the four conditions is available in the Supporting Information. For ease of identification of minor components, the relevant spectra were multiplied by corresponding factors as shown.
O 1s
Ti 2p
N 1s
C 1s
Mass Spectrometry
Figure 6
Figure 6. Time-of-flight mass spectrometric behavior of detected species during TiN ALD from TDMAT and NH3.
4. Discussion
Reaction Mechanisms
Figure 7
Figure 7. A simplified illustration of the early stages of TiN ALD showing TDMAT reacting with an OH-terminated surface, emitting the protonated ligand, dimethylamine, leaving the surface terminated in titanium dimethylamide, completing the first half of the reaction. In the second half, the introduced ammonia removes the surface bound dimethylamide as dimethylamine, resulting in an NH2-terminated surface.
5. Conclusions
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.chemmater.5c02974.
Additional details from the APXPS measurements (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
We gratefully acknowledge financial support by Seco Tools, by Vinnova, through the project “Surface chemical mechanisms during atomic layer deposition of hard nitrides” (2023-02815) and by the Stiftelsen för Strategisk Forskning (Swedish foundation for Strategic Research, SSF) through the project ‘‘Time-resolved low temperature CVD for III-nitrides’’ (SSF-RMA 15-0018). H.P. acknowledge financial support from the Swedish Government Strategic Research Area in Materials Science on Advanced Functional Materials at Linköping University (Faculty Grant SFO-Mat-LiU No. 2009-00971). P.M. is grateful to Marcus Lorentzon for supplying the MIST code. J.S. acknowledges support by Vetenskapsrådet (Swedish Research Council, VR) under project grant no. 2023-03492. We acknowledge the MAX IV Laboratory for beamtime on the SPECIES beamline under proposal 20231261. Research conducted at MAX IV, a Swedish national user facility, is supported by VR under contract 2018-07152, Vinnova (Swedish Governmental Agency for Innovation Systems) under contract 2018-04969 and Formas under contract 2019-02496.
References
This article references 95 other publications.
- 1Puurunen, R. L. Surface Chemistry of Atomic Layer Deposition: A Case Study for the Trimethylaluminum/Water Process. J. Appl. Phys. 2005, 97, 121301 DOI: 10.1063/1.1940727Google ScholarThere is no corresponding record for this reference.
- 2Ritala, M.; Leskelä, M. Atomic Layer Deposition. In Handbook of Thin Films Materials: Deposition and Processing of Thin Films, Nalwa, H. S., Ed.; Academic Press: United Kingdom, 2002; Vol. 1, pp 103– 159.Google ScholarThere is no corresponding record for this reference.
- 3Richey, N. E.; De Paula, C.; Bent, S. F. Understanding Chemical and Physical Mechanisms in Atomic Layer Deposition. J. Chem. Phys. 2020, 152, 040902 DOI: 10.1063/1.5133390Google ScholarThere is no corresponding record for this reference.
- 4Langereis, E.; Heil, S. B. S.; Van De Sanden, M. C. M.; Kessels, W. M. M. Initial Growth and Properties of Atomic Layer Deposited TiN Films Studied by in Situ Spectroscopic Ellipsometry. Phys. Status Solidi C Conf. 2005, 2, 3958– 3962, DOI: 10.1002/pssc.200562218Google ScholarThere is no corresponding record for this reference.
- 5Puurunen, R. L.; Vandervorst, W. Island Growth as a Growth Mode in Atomic Layer Deposition: A Phenomenological Model. J. Appl. Phys. 2004, 96, 7686– 7695, DOI: 10.1063/1.1810193Google ScholarThere is no corresponding record for this reference.
- 6D’Acunto, G.; Tsyshevsky, R.; Shayesteh, P.; Gallet, J. J.; Bournel, F.; Rochet, F.; Pinsard, I.; Timm, R.; Head, A. R.; Kuklja, M.; Schnadt, J. Bimolecular Reaction Mechanism in the Amido Complex-Based Atomic Layer Deposition of HfO2. Chem. Mater. 2023, 35, 529– 538, DOI: 10.1021/acs.chemmater.2c02947Google ScholarThere is no corresponding record for this reference.
- 7Knapas, K.; Ritala, M. In Situ Studies on Reaction Mechanisms in Atomic Layer Deposition. Crit. Rev. Solid State Mater. Sci. 2013, 38, 167– 202, DOI: 10.1080/10408436.2012.693460Google ScholarThere is no corresponding record for this reference.
- 8Jones, R.; Kokkonen, E.; Eads, C.; Küst, U. K.; Prumbs, J.; Knudsen, J.; Schnadt, J. Time-Resolved Ambient Pressure x-Ray Photoelectron Spectroscopy: Advancing the Operando Study of ALD Chemistry. Surf. Sci. 2025, 753, 122656 DOI: 10.1016/j.susc.2024.122656Google ScholarThere is no corresponding record for this reference.
- 9Jones, R.; D’Acunto, G.; Shayesteh, P.; Pinsard, I.; Rochet, F.; Bournel, F.; Gallet, J.-J.; Head, A.; Schnadt, J. Operando Study of HfO2 Atomic Layer Deposition on Partially Hydroxylated Si(111). J. Vac. Sci. Technol. A 2024, 42, 022404 DOI: 10.1116/6.0003349Google ScholarThere is no corresponding record for this reference.
- 10Kokkonen, E.; Nieminen, H.; Rehman, F.; Miikkulainen, V.; Putkonen, M.; Ritala, M.; Huotari, S.; Schnadt, J.; Urpelainen, S. Ambient Pressure X-Ray Photoelectron Spectroscopy Study on the Initial Atomic Layer Deposition Process of Platinum. J. Vac. Sci. Technol. A 2024, 42, 062406 DOI: 10.1116/6.0003871Google ScholarThere is no corresponding record for this reference.
- 11Starr, D. E.; Liu, Z.; Hävecker, M.; Knop-Gericke, A.; Bluhm, H. Investigation of Solid/Vapor Interfaces Using Ambient Pressure X-Ray Photoelectron Spectroscopy. Chem. Soc. Rev. 2013, 42, 5833– 5857, DOI: 10.1039/c3cs60057bGoogle ScholarThere is no corresponding record for this reference.
- 12Kim, H. Atomic Layer Deposition of Metal and Nitride Thin Films: Current Research Efforts and Applications for Semiconductor Device Processing. J. Vac. Sci. Technol. B Microelectron. Nanom. Struct. Process. Meas. Phenom. 2003, 21, 2231– 2261, DOI: 10.1116/1.1622676Google ScholarThere is no corresponding record for this reference.
- 13Wang, C. Y.; Chou, C. Y.; Shiue, H. F.; Chen, H. Y.; Ling, C. H.; Shyue, J. J.; Chen, M. J. Atomic Layer Annealing for Modulation of the Work Function of TiN Metal Gate for N-Type MOS Devices. Appl. Surf. Sci. 2022, 585, 152748 DOI: 10.1016/j.apsusc.2022.152748Google ScholarThere is no corresponding record for this reference.
- 14Lima, L. P. B.; Dekkers, H. F. W.; Lisoni, J. G.; Diniz, J. A.; Van Elshocht, S.; De Gendt, S. Metal Gate Work Function Tuning by Al Incorporation in TiN. J. Appl. Phys. 2014, 115, 074504 DOI: 10.1063/1.4866323Google ScholarThere is no corresponding record for this reference.
- 15Chourasia, A. R.; Chopra, D. R. X-Ray Photoelectron Study of TiN/SiO2 and TiN/Si Interfaces. Thin Solid Films 1995, 266, 298– 301, DOI: 10.1016/0040-6090(95)06651-9Google ScholarThere is no corresponding record for this reference.
- 16Jouan, P.-Y.; Lemp, G.; Peignon, C. Characterisation of TiN Coatings and of the TiN/Si Interface by X-Ray Photoelectron Spectroscopy and Auger Electron Spectroscopy. Appl. Surf. Sci. 1993, 68, 595– 603, DOI: 10.1016/0169-4332(93)90241-3Google ScholarThere is no corresponding record for this reference.
- 17Morales, M.; Cucatti, S.; Acuña, J. J. S.; Zagonel, L. F.; Antonin, O.; Hugon, M. C.; Marsot, N.; Bouchet-Fabre, B.; Minea, T.; Alvarez, F. Influence of the Structure and Composition of Titanium Nitride Substrates on Carbon Nanotubes Grown by Chemical Vapour Deposition. J. Phys. D. Appl. Phys. 2013, 46, 155308 DOI: 10.1088/0022-3727/46/15/155308Google ScholarThere is no corresponding record for this reference.
- 18Morales, M.; Merlo, R. B.; Droppa, R.; Alvarez, F. Self-Organized 2D Ni Particles Deposited on Titanium Oxynitride-Coated Si Sculpted by a Low Energy Ion Beam. J. Phys. D. Appl. Phys. 2014, 47, 195303 DOI: 10.1088/0022-3727/47/19/195303Google ScholarThere is no corresponding record for this reference.
- 19López, J. M.; Gordillo-Vázquez, F. J.; Böhme, O.; Albella, J. M. Low Grain Size TiN Thin Films Obtained by Low Energy Ion Beam Assisted Deposition. Appl. Surf. Sci. 2001, 173, 290– 295, DOI: 10.1016/S0169-4332(00)00912-0Google ScholarThere is no corresponding record for this reference.
- 20Duan, G.; Zhao, G.; Wu, L.; Lin, X.; Han, G. Structure, Electrical and Optical Properties of TiN x Films by Atmospheric Pressure Chemical Vapor Deposition. Appl. Surf. Sci. 2011, 257, 2428– 2431, DOI: 10.1016/j.apsusc.2010.11.180Google ScholarThere is no corresponding record for this reference.
- 21Pierson, H. O. A Review of the Chemical Vapor Deposition (Cvd) of the Refractory Compounds Of Titanium - A Unique Family Of Coatings. Mater. Manuf. Process. 1993, 8, 519– 534, DOI: 10.1080/10426919308934855Google ScholarThere is no corresponding record for this reference.
- 22Yokota, K.; Nakamura, K.; Kasuya, T.; Tamura, S.; Sugimoto, T.; Akamatsu, K.; Nakao, K.; Miyashita, F. Relationship between Hardness and Lattice Parameter for TiN Films Deposited on SUS 304 by an IBAD Technique. Surf. Coat. Technol. 2002, 158–159, 690– 693, DOI: 10.1016/S0257-8972(02)00245-1Google ScholarThere is no corresponding record for this reference.
- 23D’Acunto, G.; Troian, A.; Kokkonen, E.; Rehman, F.; Liu, Y. P.; Yngman, S.; Yong, Z.; McKibbin, S. R.; Gallo, T.; Lind, E.; Schnadt, J.; Timm, R. Atomic Layer Deposition of Hafnium Oxide on InAs: Insight from Time-Resolved in Situ Studies. ACS Appl. Electron. Mater. 2020, 2, 3915– 3922, DOI: 10.1021/acsaelm.0c00775Google ScholarThere is no corresponding record for this reference.
- 24D’Acunto, G.; Jones, R.; Pérez Ramírez, L.; Shayesteh, P.; Kokkonen, E.; Rehman, F.; Lim, F.; Bournel, F.; Gallet, J. J.; Timm, R.; Schnadt, J. Role of Temperature, Pressure, and Surface Oxygen Migration in the Initial Atomic Layer Deposition of HfO2 on Anatase TiO2(101). J. Phys. Chem. C 2022, 126, 12210– 12221, DOI: 10.1021/acs.jpcc.2c02683Google ScholarThere is no corresponding record for this reference.
- 25Sobell, Z. C.; George, S. M. Electron-Enhanced Atomic Layer Deposition of Titanium Nitride Films Using an Ammonia Reactive Background Gas. Chem. Mater. 2022, 34, 9624– 9633, DOI: 10.1021/acs.chemmater.2c02341Google ScholarThere is no corresponding record for this reference.
- 26Musschoot, J.; Xie, Q.; Deduytsche, D.; Van den Berghe, S.; Van Meirhaeghe, R. L.; Detavernier, C. Atomic Layer Deposition of Titanium Nitride from TDMAT Precursor. Microelectron. Eng. 2009, 86, 72– 77, DOI: 10.1016/j.mee.2008.09.036Google ScholarThere is no corresponding record for this reference.
- 27Zhu, Y.; Zhou, Z.; Zhang, X.; Xu, R.; Wang, Y.; Xu, L.; Xiao, H.; Li, X.; Li, A.; Fang, G. Surface Reaction Mechanism of Atomic Layer Deposition of Titanium Nitride Using Tetrakis(Dimethylamino)Titanium and Ammonia. Surfaces and Interfaces 2023, 36, 102579 DOI: 10.1016/j.surfin.2022.102579Google ScholarThere is no corresponding record for this reference.
- 28Cho, G.; Rhee, S.-W. Effect of the Amido Ti Precursors on the Atomic Layer Deposition of TiN with NH3. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2013, 31, 01A117 DOI: 10.1116/1.4764898Google ScholarThere is no corresponding record for this reference.
- 29Park, K.; Mun, G.; Cho, J.; Lee, S.; Min, G.; Hong, S. Properties of TiN Thin Films Deposited by ALD Using TDMAT Precursor in Low-Temperature Processes. J. Semicond. Dispersion Technol. 2024, 23, 14– 20Google ScholarThere is no corresponding record for this reference.
- 30Schnadt, J.; Knudsen, J.; Andersen, J. N.; Siegbahn, H.; Pietzsch, A.; Hennies, F.; Johansson, N.; Mårtensson, N.; Öhrwall, G.; Bahr, S.; Mähl, S.; Schaff, O. The New Ambient-Pressure X-Ray Photoelectron Spectroscopy Instrument at MAX-Lab. J. Synchrotron Radiat. 2012, 19, 701– 704, DOI: 10.1107/S0909049512032700Google ScholarThere is no corresponding record for this reference.
- 31Knudsen, J.; Andersen, J. N.; Schnadt, J. A Versatile Instrument for Ambient Pressure X-Ray Photoelectron Spectroscopy: The Lund Cell Approach. Surf. Sci. 2016, 646, 160– 169, DOI: 10.1016/j.susc.2015.10.038Google ScholarThere is no corresponding record for this reference.
- 32Kokkonen, E.; Kaipio, M.; Nieminen, H. E.; Rehman, F.; Miikkulainen, V.; Putkonen, M.; Ritala, M.; Huotari, S.; Schnadt, J.; Urpelainen, S. Ambient Pressure X-Ray Photoelectron Spectroscopy Setup for Synchrotron-Based in Situ and Operando Atomic Layer Deposition Research. Rev. Sci. Instrum. 2022, 93, 013905 DOI: 10.1063/5.0076993Google ScholarThere is no corresponding record for this reference.
- 33Kokkonen, E.; Da Silva, F. L.; Mikkela, M. H.; Johansson, N.; Huang, S. W.; Lee, J. M.; Andersson, M.; Bartalesi, A.; N. Reinecke, B.; Handrup, K.; Tarawneh, H.; Sankari, R.; Knudsen, J.; Schnadt, J.; Sathe, C.; Urpelainen, S. Upgrade of the SPECIES Beamline at the MAX IV Laboratory. J. Synchrotron Radiat. 2021, 28, 588– 601, DOI: 10.1107/S1600577521000564Google ScholarThere is no corresponding record for this reference.
- 34Tissot, H.; Gallet, J. J.; Bournel, F.; Naitabdi, A.; Pierucci, D.; Bondino, F.; Magnano, E.; Rochet, F.; Finocchi, F. Silicon Monomer Formation and Surface Patterning of Si(001)-2 × 1 Following Tetraethoxysilane Dissociative Adsorption at Room Temperature. J. Phys. Chem. C 2014, 118, 1887– 1893, DOI: 10.1021/jp407411kGoogle ScholarThere is no corresponding record for this reference.
- 35Kim, J. W.; Yeom, H. W.; Chung, Y. D.; Jeong, K.; Whang, C. N.; Lee, M. K.; Shin, H. J. Chemical Configuration of Nitrogen in Ultrathin Si Oxynitride on Si(100). Phys. Rev. B - Condens. Matter Mater. Phys. 2002, 66, 035312, DOI: 10.1103/PhysRevB.66.035312Google ScholarThere is no corresponding record for this reference.
- 36Verdaguer, A.; Weis, C.; Oncins, G.; Ketteler, G.; Bluhm, H.; Salmero, M. Growth and Structure of Water on SiO2 Films on Si Investigated by Kelvin Probe Microscopy and in Situ X-Ray Spectroscopies. Langmuir 2007, 23, 9699– 9703, DOI: 10.1021/la700893wGoogle ScholarThere is no corresponding record for this reference.
- 37Kim, Y. K.; Hyun, S. L.; Yeom, H. W.; Ryoo, D. Y.; Huh, S. B.; Lee, J. G. Nitrogen Bonding Structure in Ultrathin Silicon Oxynitride Films on Si(100) Prepared by Plasma Nitridation. Phys. Rev. B - Condens. Matter Mater. Phys. 2004, 70, 165320 DOI: 10.1103/PhysRevB.70.165320Google ScholarThere is no corresponding record for this reference.
- 38Simonsen, M. E.; Sønderby, C.; Li, Z.; Søgaard, E. G. XPS and FT-IR Investigation of Silicate Polymers. J. Mater. Sci. 2009, 44, 2079– 2088, DOI: 10.1007/s10853-009-3270-9Google ScholarThere is no corresponding record for this reference.
- 39Namiki, A.; Tanimoto, K.; Nakamura, T.; Ohtake, N.; Suzaki, T. XPS Study on the Early Stages of Oxidation of Si(100) by Atomic Oxygen. Surf. Sci. 1989, 222, 530– 554, DOI: 10.1016/0039-6028(89)90377-4Google ScholarThere is no corresponding record for this reference.
- 40Yang, J.; Wang, E. G. Reaction of Water on Silica Surfaces. Curr. Opin. Solid State Mater. Sci. 2006, 10, 33– 39, DOI: 10.1016/j.cossms.2006.02.001Google ScholarThere is no corresponding record for this reference.
- 41Rignanese, G. M.; Charlier, J. C.; Gonze, X. First-Principles Molecular-Dynamics Investigation of the Hydration Mechanisms of the (0001) α-Quartz Surface. Phys. Chem. Chem. Phys. 2004, 6, 1920– 1925, DOI: 10.1039/B311842HGoogle ScholarThere is no corresponding record for this reference.
- 42Miyaji, F.; Iwai, M.; Kokubo, T.; Nakamura, T. Chemical Surface Treatment of Silicone for Inducing Its Bioactivity. J. Mater. Sci. Mater. Med. 1998, 9, 61– 65, DOI: 10.1023/A:1008886729050Google ScholarThere is no corresponding record for this reference.
- 43Göpel, W.; Anderson, J. A.; Frankel, D.; Jaehnig, M.; Phillips, K.; Schäfer, J. A.; Rocker, G. Surface Defects of TiO2(110): A Combined XPS, XAES AND ELS Study. Surf. Sci. 1984, 139, 333– 346, DOI: 10.1016/0039-6028(84)90054-2Google ScholarThere is no corresponding record for this reference.
- 44Saha, N. C.; Tompkins, H. G. Titanium Nitride Oxidation Chemistry: An x-Ray Photoelectron Spectroscopy Study. J. Appl. Phys. 1992, 72, 3072– 3079, DOI: 10.1063/1.351465Google ScholarThere is no corresponding record for this reference.
- 45Head, A. R.; Chaudhary, S.; Olivieri, G.; Bournel, F.; Andersen, J. N.; Rochet, F.; Gallet, J. J.; Schnadt, J. Near Ambient Pressure X-Ray Photoelectron Spectroscopy Study of the Atomic Layer Deposition of TiO2 on RuO2(110). J. Phys. Chem. C 2016, 120, 243– 251, DOI: 10.1021/acs.jpcc.5b08699Google ScholarThere is no corresponding record for this reference.
- 46Jansen, R. J. J.; van Bekkum, H. XPS of Nitrogen-Containing Functional Groups on Activated Carbon. Carbon N. Y. 1995, 33, 1021– 1027, DOI: 10.1016/0008-6223(95)00030-HGoogle ScholarThere is no corresponding record for this reference.
- 47Bruggeman, M.; Zelzer, M.; Dong, H.; Stamboulis, A. Processing and Interpretation of Core-Electron XPS Spectra of Complex Plasma-Treated Polyethylene-Based Surfaces Using a Theoretical Peak Model. Surf. Interface Anal. 2022, 54, 986– 1007, DOI: 10.1002/sia.7125Google ScholarThere is no corresponding record for this reference.
- 48Cao, X.; Hamers, R. J. Silicon Surfaces as Electron Acceptors: Dative Bonding of Amines with Si(001) and Si(111) Surfaces. J. Am. Chem. Soc. 2001, 123, 10988– 10996, DOI: 10.1021/ja0100322Google ScholarThere is no corresponding record for this reference.
- 49Peña-Juárez, M. G.; Robles-Martínez, M.; Méndez-Rodríguez, K. B.; López-Esparza, R.; Pérez, E.; Gonzalez-Calderon, J. A. Role of the Chemical Modification of Titanium Dioxide Surface on the Interaction with Silver Nanoparticles and the Capability to Enhance Antimicrobial Properties of Poly(Lactic Acid) Composites. Polym. Bull. 2021, 78, 2765– 2790, DOI: 10.1007/s00289-020-03235-yGoogle ScholarThere is no corresponding record for this reference.
- 50Ngoc Van, T. T.; Jang, D.; Jung, E.; Noh, H.; Moon, J.; Kil, D. S.; Chung, S. W.; Shong, B. Role of Cyclopentadienyl Ligands of Group 4 Precursors toward High-Temperature Atomic Layer Deposition. J. Phys. Chem. C 2022, 126, 18090– 18099, DOI: 10.1021/acs.jpcc.2c04425Google ScholarThere is no corresponding record for this reference.
- 51Li, K.; Li, S.; Li, N.; Klein, T. M.; Dixon, D. A. Tetrakis(Ethylmethylamido) Hafnium Adsorption and Reaction on Hydrogen-Terminated Si(100) Surfaces. J. Phys. Chem. C 2011, 115, 18560– 18571, DOI: 10.1021/jp111600vGoogle ScholarThere is no corresponding record for this reference.
- 52Trenczek-Zajac, A.; Radecka, M.; Zakrzewska, K.; Brudnik, A.; Kusior, E.; Bourgeois, S.; de Lucas, M. C. M.; Imhoff, L. Structural and Electrical Properties of Magnetron Sputtered Ti(ON) Thin Films: The Case of TiN Doped in Situ with Oxygen. J. Power Sources 2009, 194, 93– 103, DOI: 10.1016/j.jpowsour.2008.12.112Google ScholarThere is no corresponding record for this reference.
- 53Killampalli, A. S.; Ma, P. F.; Engstrom, J. R. The Reaction of Tetrakis (Dimethylamido) Titanium with Self-Assembled Alkyltrichlorosilane Monolayers Possessing. J. Am. Chem. Soc. 2005, 127, 6300– 6310, DOI: 10.1021/ja047922cGoogle ScholarThere is no corresponding record for this reference.
- 54Peng, Z.; Chen, G.; Zhao, Y.-P.; Zhang, X.; Song, Y.-T.; Shirkov, G.; Karamysheva, G.; Karamyshev, O.; Calabretta, L.; Caruso, A. Investigation of TiN Film on an RF Ceramic Window by Atomic Layer Deposition. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2020, 38, 052401 DOI: 10.1116/6.0000159Google ScholarThere is no corresponding record for this reference.
- 55Nguyen, T. P.; Lefrant, S. XPS Study of SiO Thin Films and SiO-Metal Interfaces. J. Phys.: Condens. Matter 1989, 1, 5197– 5204, DOI: 10.1088/0953-8984/1/31/019Google ScholarThere is no corresponding record for this reference.
- 56Desbiens, E.; Dolbec, R.; El Khakani, M. A. Reactive Pulsed Laser Deposition of High- k Silicon Dioxide and Silicon Oxynitride Thin Films for Gate-Dielectric Applications. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2002, 20, 1157– 1161, DOI: 10.1116/1.1467357Google ScholarThere is no corresponding record for this reference.
- 57Ketteler, G.; Yamamoto, S.; Bluhm, H.; Andersson, K.; Starr, D. E.; Ogletree, D. F.; Ogasawara, H.; Nilsson, A.; Salmeron, M. The Nature of Water Nucleation Sites on TiO2(110) Surfaces Revealed by Ambient Pressure X-Ray Photoelectron Spectroscopy. J. Phys. Chem. C 2007, 111, 8278– 8282, DOI: 10.1021/jp068606iGoogle ScholarThere is no corresponding record for this reference.
- 58Walle, L. E.; Borg, A.; Johansson, E. M. J.; Plogmaker, S.; Rensmo, H.; Uvdal, P.; Sandell, A. Mixed Dissociative and Molecular Water Adsorption on Anatase TiO 2(101). J. Phys. Chem. C 2011, 115, 9545– 9550, DOI: 10.1021/jp111335wGoogle ScholarThere is no corresponding record for this reference.
- 59Walle, L. E.; Borg, A.; Uvdal, P.; Sandell, A. Experimental Evidence for Mixed Dissociative and Molecular Adsorption of Water on a Rutile TiO2 (110) Surface without Oxygen Vacancies. Phys. Rev. B - Condens. Matter Mater. Phys. 2009, 80, 235436 DOI: 10.1103/PhysRevB.80.235436Google ScholarThere is no corresponding record for this reference.
- 60Esaka, F.; Furuya, K.; Shimada, H.; Imamura, M.; Matsubayashi, N.; Sato, H.; Nishijima, A.; Kawana, A.; Ichimura, H.; Kikuchi, T. Comparison of Surface Oxidation of Titanium Nitride and Chromium Nitride Films Studied by X-Ray Absorption and Photoelectron Spectroscopy. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 1997, 15, 2521– 2528, DOI: 10.1116/1.580764Google ScholarThere is no corresponding record for this reference.
- 61Jaeger, D.; Patscheider, J. Single Crystalline Oxygen-Free Titanium Nitride by XPS. Surf. Sci. Spectra 2013, 20, 1, DOI: 10.1116/11.20121107Google ScholarThere is no corresponding record for this reference.
- 62Chan, M. H.; Lu, F. H. X-Ray Photoelectron Spectroscopy Analyses of Titanium Oxynitride Films Prepared by Magnetron Sputtering Using Air/Ar Mixtures. Thin Solid Films 2009, 517, 5006– 5009, DOI: 10.1016/j.tsf.2009.03.100Google ScholarThere is no corresponding record for this reference.
- 63Robinson, K. S.; Sherwood, P. M. A. X-Ray Photoelectron Spectroscopic Studies of the Surface of Sputter Ion Plated Films. Surf. Interface Anal. 1984, 6, 261– 266, DOI: 10.1002/sia.740060603Google ScholarThere is no corresponding record for this reference.
- 64Cherono, S.; Chris-Okoro, I.; Liu, M.; Kim, R. S.; Nalawade, S.; Akande, W.; Maria-Diana, M.; Mahl, J.; Hale, C.; Yano, J.; Aravamudhan, S.; Crumlin, E.; Craciun, V.; Kumar, D. Transformation of TiN to TiNO Films via In-Situ Temperature-Dependent Oxygen Diffusion Process and Their Electrochemical Behavior. Metals (Basel). 2025, 15, 497, DOI: 10.3390/met15050497Google ScholarThere is no corresponding record for this reference.
- 65Kuznetsov, M. V.; Zhuravlev, J. F.; Zhilyaev, V. A.; Gubanov, V. A. XPS Study of the Nitrides, Oxides and Oxynitrides of Titanium. J. Electron Spectrosc. Relat. Phenom. 1992, 58, 1, DOI: 10.1016/0368-2048(92)80001-OGoogle ScholarThere is no corresponding record for this reference.
- 66Kabachkov, E.; Kurkin, E.; Vershinin, N.; Balikhin, I.; Berestenko, V.; Michtchenko, A.; Shulga, Y. Synthesis and Properties of Pt/TiN Catalyst for Low-Temperature Air Purification from Carbon Monoxide. J. Adv. Mater. Technol. 2021, 6, 131– 143, DOI: 10.17277/jamt.2021.02.pp.131-143Google ScholarThere is no corresponding record for this reference.
- 67George, S. M. Atomic Layer Deposition: An Overview. Chem. Rev. 2010, 110, 111– 131, DOI: 10.1021/cr900056bGoogle ScholarThere is no corresponding record for this reference.
- 68Corneille, J. S.; Chen, P. J.; Truong, C. M.; Oh, W. S.; Goodman, D. W. Surface Spectroscopic Studies of the Deposition of TiN Thin Films from Tetrakis-(Dimethylamido)-Titanium and Ammonia. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 1995, 13, 1116– 1120, DOI: 10.1116/1.579596Google ScholarThere is no corresponding record for this reference.
- 69Xiong, H.; Wu, L.; Liu, Y.; Gao, T.; Li, K.; Long, Y.; Zhang, R.; Zhang, L.; Qiao, Z. A.; Huo, Q.; Ge, X.; Song, S.; Zhang, H. Controllable Synthesis of Mesoporous TiO2 Polymorphs with Tunable Crystal Structure for Enhanced Photocatalytic H2 Production. Adv. Energy Mater. 2019, 9, 1901634 DOI: 10.1002/aenm.201901634Google ScholarThere is no corresponding record for this reference.
- 70Peng, F.; Cai, L.; Huang, L.; Yu, H.; Wang, H. Preparation of Nitrogen-Doped Titanium Dioxide with Visible-Light Photocatalytic Activity Using a Facile Hydrothermal Method. J. Phys. Chem. Solids 2008, 69, 1657– 1664, DOI: 10.1016/j.jpcs.2007.12.003Google ScholarThere is no corresponding record for this reference.
- 71Zhang, H.; Wang, B.; Brown, B. Atomic Layer Deposition of Titanium Oxide and Nitride on Vertically Aligned Carbon Nanotubes for Energy Dense 3D Microsupercapacitors. Appl. Surf. Sci. 2020, 521, 146349 DOI: 10.1016/j.apsusc.2020.146349Google ScholarThere is no corresponding record for this reference.
- 72Mpofu, P.; Niiranen, P.; Alm, O.; Lauridsen, J.; Larsson, T.; Pedersen, H. Atomic Layer Deposition of AlxTi1-XN via Co-Evaporation of Metal Precursors. J. Vac. Sci. Technol. A 2025, 43, 032405 DOI: 10.1116/6.0004420Google ScholarThere is no corresponding record for this reference.
- 73Jintsugawa, O.; Sakuraba, M.; Matsuura, T.; Murota, J. Thermal Nitridation of Ultrathin SiO2 on Si by NH3. Surf. Interface Anal. 2002, 34, 456– 459, DOI: 10.1002/sia.1337Google ScholarThere is no corresponding record for this reference.
- 74Graf, N.; Yegen, E.; Gross, T.; Lippitz, A.; Weigel, W.; Krakert, S.; Terfort, A.; Unger, W. E. S. XPS and NEXAFS Studies of Aliphatic and Aromatic Amine Species on Functionalized Surfaces. Surf. Sci. 2009, 603, 2849– 2860, DOI: 10.1016/j.susc.2009.07.029Google ScholarThere is no corresponding record for this reference.
- 75Dietrich, P. M.; Graf, N.; Gross, T.; Lippitz, A.; Krakert, S.; Schüpbach, B.; Terfort, A.; Unger, W. E. S. Amine Species on Self-Assembled Monolayers of ω-Aminothiolates on Gold as Identified by XPS and NEXAFS Spectroscopy. Surf. Interface Anal. 2010, 42, 1184– 1187, DOI: 10.1002/sia.3224Google ScholarThere is no corresponding record for this reference.
- 76Solís, R. R.; Gómez-Avilés, A.; Belver, C.; Rodriguez, J. J.; Bedia, J. Microwave-Assisted Synthesis of NH2-MIL-125(Ti) for the Solar Photocatalytic Degradation of Aqueous Emerging Pollutants in Batch and Continuous Tests. J. Environ. Chem. Eng. 2021, 9, 106230 DOI: 10.1016/j.jece.2021.106230Google ScholarThere is no corresponding record for this reference.
- 77Endle, J. P.; Sun, Y.; White, J. M.; Ekerdt, J. G. X-Ray Photoelectron Spectroscopy Study of TiN Films Produced with Tetrakis(Dimethylamido)Titanium and Selected N-Containing Precursors on SiO2. J. Vac. Sci. Technol. A 1998, 16, 1262– 1267, DOI: 10.1116/1.581271Google ScholarThere is no corresponding record for this reference.
- 78Quesada-Cabrera, R.; Sotelo-Vazquez, C.; Darr, J. A.; Parkin, I. P. Critical Influence of Surface Nitrogen Species on the Activity of N-Doped TiO2 Thin-Films during Photodegradation of Stearic Acid under UV Light Irradiation. Appl. Catal. B Environ. 2014, 160–161, 582– 588, DOI: 10.1016/j.apcatb.2014.06.010Google ScholarThere is no corresponding record for this reference.
- 79Asghar, M.; Yousaf, M.; Huwaimel, B.; Iqbal, T.; Ahmed, I.; Qureshi, M. T.; Abrar, M.; Shafiq, M.; Almohammedi, A.; Hameed, R. A.; AlElaimi, M.; Maryam, M.; Afsheen, S. Synthesis of Biocompatible Coating on Ni-Cr Alloy by Cathodic Cage Plasma Processing Technique as Anti-Pathogenic Bacteria for Medicinal Applications. Phys. Scr. 2023, 98, 055920 DOI: 10.1088/1402-4896/acc908Google ScholarThere is no corresponding record for this reference.
- 80Braic, L.; Vasilantonakis, N.; Mihai, A.; Villar Garcia, I. J.; Fearn, S.; Zou, B.; Alford, N. M. N.; Doiron, B.; Oulton, R. F.; Maier, S. A.; Zayats, A. V.; Petrov, P. K. Titanium Oxynitride Thin Films with Tunable Double Epsilon-Near-Zero Behavior for Nanophotonic Applications. ACS Appl. Mater. Interfaces 2017, 9, 29857– 29862, DOI: 10.1021/acsami.7b07660Google ScholarThere is no corresponding record for this reference.
- 81Li, K.; Li, S.; Li, N.; Dixon, D. A.; Klein, T. M. Tetrakis(Dimethylamido)Hafnium Adsorption and Reaction on Hydrogen Terminated Si(100) Surfaces. J. Phys. Chem. C 2010, 114, 14061– 14075, DOI: 10.1021/jp101363rGoogle ScholarThere is no corresponding record for this reference.
- 82Ruhl, G.; Rehmet, R.; Knfžvá, M.; Vepřek, S. In Situ XPS Studies of the Deposition of Thin Films from Tetrakis(Dimethylamido)Titanium Organometallic Precursor for Diffusion Barriers. MRS Online Proc. Libr. 1993, 309, 461– 466, DOI: 10.1557/PROC-309-461Google ScholarThere is no corresponding record for this reference.
- 83NIST NIST Standard Reference Database Number 20. https://dx.doi.org/10.18434/T4T88K (accessed Apr 7, 2025).Google ScholarThere is no corresponding record for this reference.
- 84Longrie, D.; Deduytsche, D.; Haemers, J.; Smet, P. F.; Driesen, K.; Detavernier, C. Thermal and Plasma-Enhanced Atomic Layer Deposition of TiN Using TDMAT and NH3 on Particles Agitated in a Rotary Reactor. ACS Appl. Mater. Interfaces 2014, 6, 7316– 7324, DOI: 10.1021/am5007222Google ScholarThere is no corresponding record for this reference.
- 85Elam, J. W.; Schuisky, M.; Ferguson, J. D.; George, S. M. Surface Chemistry and Film Growth during TiN Atomic Layer Deposition Using TDMAT and NH3. Thin Solid Films 2003, 436, 145– 156, DOI: 10.1016/S0040-6090(03)00533-9Google ScholarThere is no corresponding record for this reference.
- 86Biesinger, M. C.; Lau, L. W. M.; Gerson, A. R.; Smart, R. S. C. Resolving Surface Chemical States in XPS Analysis of First Row Transition Metals, Oxides and Hydroxides: Sc, Ti, V, Cu and Zn. Appl. Surf. Sci. 2010, 257, 887– 898, DOI: 10.1016/j.apsusc.2010.07.086Google ScholarThere is no corresponding record for this reference.
- 87Methaapanon, R.; Bent, S. F. Comparative Study of Titanium Dioxide Atomic Layer Deposition on Silicon Dioxide and Hydrogen-Terminated Silicon. J. Phys. Chem. C 2010, 114, 10498– 10504, DOI: 10.1021/jp1013303Google ScholarThere is no corresponding record for this reference.
- 88Weiller, B. H. Chemical Vapor Deposition of TiN from Tetrakis(Dimethylamido)Titanium and Ammonia: Kinetics and Mechanistic Studies of the Gas-Phase Chemistry. J. Am. Chem. Soc. 1996, 118, 4975– 4983, DOI: 10.1021/ja953468oGoogle ScholarThere is no corresponding record for this reference.
- 89Hughes, K. J.; Engstrom, J. R. Nucleation Delay in Atomic Layer Deposition on a Thin Organic Layer and the Role of Reaction Thermochemistry. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2012, 30, 01A102 DOI: 10.1116/1.3625564Google ScholarThere is no corresponding record for this reference.
- 90Mpofu, P.; Hafdi, H.; Lauridsen, J.; Alm, O.; Larsson, T.; Pedersen, H. A Mass Spectrometrical Surface Chemistry Study of Aluminum Nitride ALD from Tris-Dimethylamido Aluminum and Ammonia. Mater. Adv. 2024, 5, 9259– 9269, DOI: 10.1039/D4MA00922CGoogle ScholarThere is no corresponding record for this reference.
- 91Lee, W. J.; Yun, E. Y.; Lee, H. B. R.; Hong, S. W.; Kwon, S. H. Dataset for TiN Thin Films Prepared by Plasma-Enhanced Atomic Layer Deposition Using Tetrakis(Dimethylamino)Titanium (TDMAT) and Titanium Tetrachloride (TiCl4) Precursor. Data Br. 2020, 31, 105777 DOI: 10.1016/j.dib.2020.105777Google ScholarThere is no corresponding record for this reference.
- 92Yun, J.; Park, M.; Rhee, S. Comparison of Tetrakis(Dimethylamido)Titanium and Tetrakis(Diethylamido)Titanium as Precursors for Metallorganic Chemical Vapor Deposition of Titanium Nitride. J. Electrochem. Soc. 1999, 146, 1804– 1808, DOI: 10.1149/1.1391847Google ScholarThere is no corresponding record for this reference.
- 93Sen, K.; Banu, T.; Debnath, T.; Ghosh, D.; Das, A. K. Towards a Comprehensive Understanding of the Chemical Vapor Deposition of Titanium Nitride Using Ti(NMe2)4: A Density Functional Theory Approach. Dalt. Trans. 2014, 43, 8877– 8887, DOI: 10.1039/c4dt00690aGoogle ScholarThere is no corresponding record for this reference.
- 94Dubois, L. H.; Zegarski, B. R.; Girolami, G. S. Infrared Studies of the Surface and Gas Phase Reactions Leading to the Growth of Titanium Nitride Thin Films from Tetrakis(Dimethylamido)Titanium and Ammonia. J. Electrochem. Soc. 1992, 139, 3603– 3609, DOI: 10.1149/1.2087327Google ScholarThere is no corresponding record for this reference.
- 95Mpofu, P.; Hafdi, H.; Niiranen, P.; Lauridsen, J.; Alm, O.; Larsson, T.; Pedersen, H. Surface Chemistry in Atomic Layer Deposition of AlN Thin Films from Al(CH3)3 and NH3 Studied by Mass Spectrometry. J. Mater. Chem. C 2024, 12, 12818– 12824, DOI: 10.1039/D4TC01867BGoogle ScholarThere is no corresponding record for this reference.
Cited By
This article has not yet been cited by other publications.
Article Views
Altmetric
Citations
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
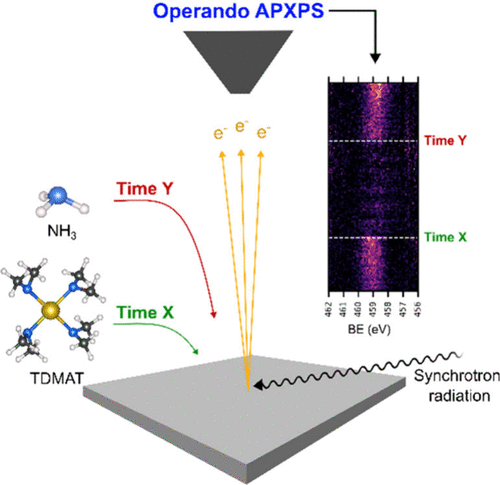
Figure 1
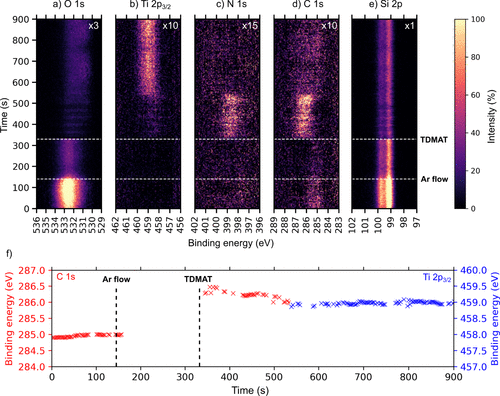
Figure 1. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXP spectra measured during the TDMAT half-cycle at 250 °C while f) shows the peak position evolution of the main peak regions in C 1s and Ti 2p plots as a function of time during the TDMAT pulse. The spectra were measured using a photon energy of 680 eV. The color scale shows how the intensity of the indicated core levels changes with time. For ease of visibility, the Ti 2p, N 1s, and C 1s intensities were multiplied by factors, as indicated, relative to the Si 2p intensity.
Figure 2
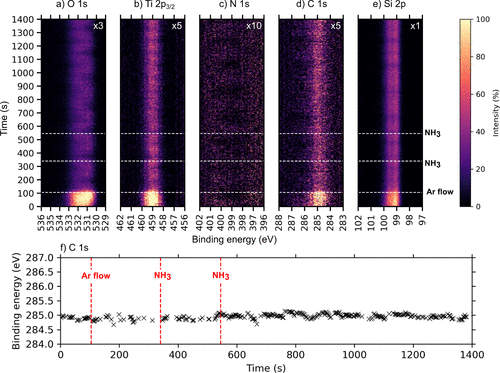
Figure 2. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXPS spectra measured during the NH3 half-cycle at 250 °C while f) shows the center of gravity plot as a function of time during the first and second NH3 pulse trains to visualize the dynamic changes. The spectra were measured using a photon energy of 680 eV. The color scale shows how the intensity of the indicated core levels changes with time. For ease of visibility, the Ti 2p, N 1s, and C 1s intensities were multiplied by factors, as indicated, relative to the Si 2p intensity.
Figure 3
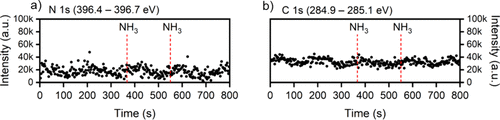
Figure 3. a) N 1s and b) C 1s intensity evolutions extracted at defined time points from the image plots in Figure 2c) and d), respectively, allowing a more direct comparison of the temporal evolution indicating that no ligand removal is evident in the second half-cycle through NH3-assisted ligand exchange surface reactions.
Figure 4
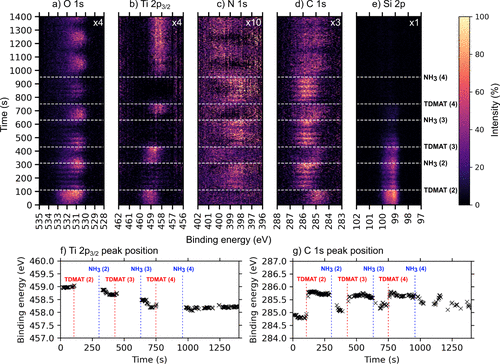
Figure 4. Time-resolved a) O 1s, b) Ti 2p, c) N 1s, d) C 1s, and e) Si 2p APXP spectra measured during three additional cycles at 250 °C, while f) and g) show the peak position evolution of the main peak regions in Ti 2p and C 1s plots as a function of time during the ALD cycles. The color scale shows how the intensity of the indicated core levels changes over time. The spectra were measured using a photon energy of 680 eV. The time at which each ALD half-cycle starts is marked by horizontal dashed lines and denoted TDMAT or NH3 to symbolize when each corresponding half-cycle starts and ends. Note that this is a continuation of Figures 1 and 2, hence precursor pulsing starts at TDMAT (2). We can clearly see the five short pulses of TDMAT in each of the TDMAT pulse trains as drops in Ti 2p intensity (this is superimposed on the raster-induced intensity changes). The dotted red line on the Ti 2p map shows the shift toward lower binding energies during the three ALD cycles with time, indicating a shift from oxide toward nitride character in the films. The oscillating behavior shown on the maps has nothing to do with the ALD process itself but emanates from the continuous movement of the sample during analysis as mentioned in the Methods section. For ease of visibility, the O 1s intensity was multiplied by a factor of 2 relative to the Si 2p intensity.
Figure 5
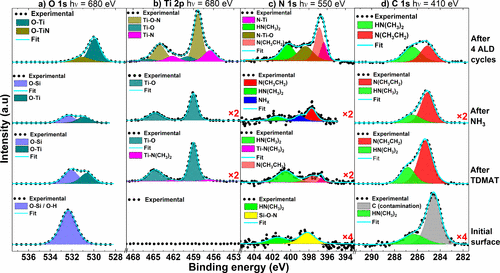
Figure 5. APXPS core level scans of the a) O 1s, b) Ti 2p, c) N 1s, and d) C 1s lines at 250 °C from an initial substrate surface until after the three additional ALD cycles. The spectra were measured using photon energies of 680 eV for the O 1s and Ti 2p lines, while 550 and 410 eV were used in the measurement of the N 1s and C 1s spectra, respectively. To make incorporation of N and Ti more visible and easier to follow, the corresponding survey spectrum for each of the four conditions is available in the Supporting Information. For ease of identification of minor components, the relevant spectra were multiplied by corresponding factors as shown.
Figure 6
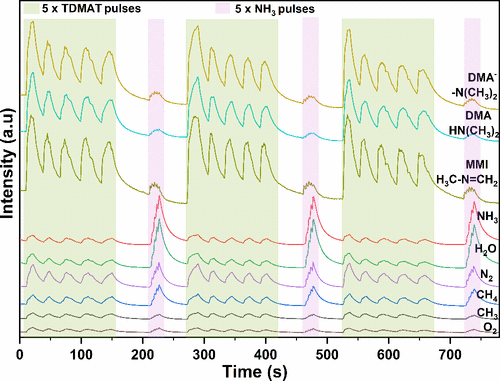
Figure 6. Time-of-flight mass spectrometric behavior of detected species during TiN ALD from TDMAT and NH3.
Figure 7
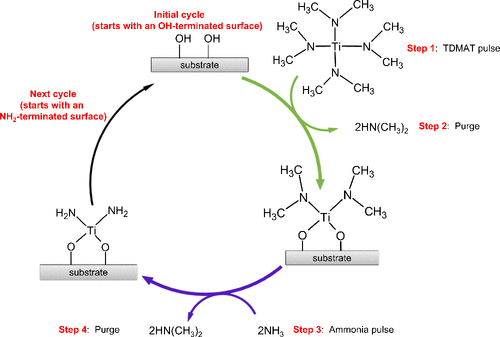
Figure 7. A simplified illustration of the early stages of TiN ALD showing TDMAT reacting with an OH-terminated surface, emitting the protonated ligand, dimethylamine, leaving the surface terminated in titanium dimethylamide, completing the first half of the reaction. In the second half, the introduced ammonia removes the surface bound dimethylamide as dimethylamine, resulting in an NH2-terminated surface.
References
This article references 95 other publications.
- 1Puurunen, R. L. Surface Chemistry of Atomic Layer Deposition: A Case Study for the Trimethylaluminum/Water Process. J. Appl. Phys. 2005, 97, 121301 DOI: 10.1063/1.1940727There is no corresponding record for this reference.
- 2Ritala, M.; Leskelä, M. Atomic Layer Deposition. In Handbook of Thin Films Materials: Deposition and Processing of Thin Films, Nalwa, H. S., Ed.; Academic Press: United Kingdom, 2002; Vol. 1, pp 103– 159.There is no corresponding record for this reference.
- 3Richey, N. E.; De Paula, C.; Bent, S. F. Understanding Chemical and Physical Mechanisms in Atomic Layer Deposition. J. Chem. Phys. 2020, 152, 040902 DOI: 10.1063/1.5133390There is no corresponding record for this reference.
- 4Langereis, E.; Heil, S. B. S.; Van De Sanden, M. C. M.; Kessels, W. M. M. Initial Growth and Properties of Atomic Layer Deposited TiN Films Studied by in Situ Spectroscopic Ellipsometry. Phys. Status Solidi C Conf. 2005, 2, 3958– 3962, DOI: 10.1002/pssc.200562218There is no corresponding record for this reference.
- 5Puurunen, R. L.; Vandervorst, W. Island Growth as a Growth Mode in Atomic Layer Deposition: A Phenomenological Model. J. Appl. Phys. 2004, 96, 7686– 7695, DOI: 10.1063/1.1810193There is no corresponding record for this reference.
- 6D’Acunto, G.; Tsyshevsky, R.; Shayesteh, P.; Gallet, J. J.; Bournel, F.; Rochet, F.; Pinsard, I.; Timm, R.; Head, A. R.; Kuklja, M.; Schnadt, J. Bimolecular Reaction Mechanism in the Amido Complex-Based Atomic Layer Deposition of HfO2. Chem. Mater. 2023, 35, 529– 538, DOI: 10.1021/acs.chemmater.2c02947There is no corresponding record for this reference.
- 7Knapas, K.; Ritala, M. In Situ Studies on Reaction Mechanisms in Atomic Layer Deposition. Crit. Rev. Solid State Mater. Sci. 2013, 38, 167– 202, DOI: 10.1080/10408436.2012.693460There is no corresponding record for this reference.
- 8Jones, R.; Kokkonen, E.; Eads, C.; Küst, U. K.; Prumbs, J.; Knudsen, J.; Schnadt, J. Time-Resolved Ambient Pressure x-Ray Photoelectron Spectroscopy: Advancing the Operando Study of ALD Chemistry. Surf. Sci. 2025, 753, 122656 DOI: 10.1016/j.susc.2024.122656There is no corresponding record for this reference.
- 9Jones, R.; D’Acunto, G.; Shayesteh, P.; Pinsard, I.; Rochet, F.; Bournel, F.; Gallet, J.-J.; Head, A.; Schnadt, J. Operando Study of HfO2 Atomic Layer Deposition on Partially Hydroxylated Si(111). J. Vac. Sci. Technol. A 2024, 42, 022404 DOI: 10.1116/6.0003349There is no corresponding record for this reference.
- 10Kokkonen, E.; Nieminen, H.; Rehman, F.; Miikkulainen, V.; Putkonen, M.; Ritala, M.; Huotari, S.; Schnadt, J.; Urpelainen, S. Ambient Pressure X-Ray Photoelectron Spectroscopy Study on the Initial Atomic Layer Deposition Process of Platinum. J. Vac. Sci. Technol. A 2024, 42, 062406 DOI: 10.1116/6.0003871There is no corresponding record for this reference.
- 11Starr, D. E.; Liu, Z.; Hävecker, M.; Knop-Gericke, A.; Bluhm, H. Investigation of Solid/Vapor Interfaces Using Ambient Pressure X-Ray Photoelectron Spectroscopy. Chem. Soc. Rev. 2013, 42, 5833– 5857, DOI: 10.1039/c3cs60057bThere is no corresponding record for this reference.
- 12Kim, H. Atomic Layer Deposition of Metal and Nitride Thin Films: Current Research Efforts and Applications for Semiconductor Device Processing. J. Vac. Sci. Technol. B Microelectron. Nanom. Struct. Process. Meas. Phenom. 2003, 21, 2231– 2261, DOI: 10.1116/1.1622676There is no corresponding record for this reference.
- 13Wang, C. Y.; Chou, C. Y.; Shiue, H. F.; Chen, H. Y.; Ling, C. H.; Shyue, J. J.; Chen, M. J. Atomic Layer Annealing for Modulation of the Work Function of TiN Metal Gate for N-Type MOS Devices. Appl. Surf. Sci. 2022, 585, 152748 DOI: 10.1016/j.apsusc.2022.152748There is no corresponding record for this reference.
- 14Lima, L. P. B.; Dekkers, H. F. W.; Lisoni, J. G.; Diniz, J. A.; Van Elshocht, S.; De Gendt, S. Metal Gate Work Function Tuning by Al Incorporation in TiN. J. Appl. Phys. 2014, 115, 074504 DOI: 10.1063/1.4866323There is no corresponding record for this reference.
- 15Chourasia, A. R.; Chopra, D. R. X-Ray Photoelectron Study of TiN/SiO2 and TiN/Si Interfaces. Thin Solid Films 1995, 266, 298– 301, DOI: 10.1016/0040-6090(95)06651-9There is no corresponding record for this reference.
- 16Jouan, P.-Y.; Lemp, G.; Peignon, C. Characterisation of TiN Coatings and of the TiN/Si Interface by X-Ray Photoelectron Spectroscopy and Auger Electron Spectroscopy. Appl. Surf. Sci. 1993, 68, 595– 603, DOI: 10.1016/0169-4332(93)90241-3There is no corresponding record for this reference.
- 17Morales, M.; Cucatti, S.; Acuña, J. J. S.; Zagonel, L. F.; Antonin, O.; Hugon, M. C.; Marsot, N.; Bouchet-Fabre, B.; Minea, T.; Alvarez, F. Influence of the Structure and Composition of Titanium Nitride Substrates on Carbon Nanotubes Grown by Chemical Vapour Deposition. J. Phys. D. Appl. Phys. 2013, 46, 155308 DOI: 10.1088/0022-3727/46/15/155308There is no corresponding record for this reference.
- 18Morales, M.; Merlo, R. B.; Droppa, R.; Alvarez, F. Self-Organized 2D Ni Particles Deposited on Titanium Oxynitride-Coated Si Sculpted by a Low Energy Ion Beam. J. Phys. D. Appl. Phys. 2014, 47, 195303 DOI: 10.1088/0022-3727/47/19/195303There is no corresponding record for this reference.
- 19López, J. M.; Gordillo-Vázquez, F. J.; Böhme, O.; Albella, J. M. Low Grain Size TiN Thin Films Obtained by Low Energy Ion Beam Assisted Deposition. Appl. Surf. Sci. 2001, 173, 290– 295, DOI: 10.1016/S0169-4332(00)00912-0There is no corresponding record for this reference.
- 20Duan, G.; Zhao, G.; Wu, L.; Lin, X.; Han, G. Structure, Electrical and Optical Properties of TiN x Films by Atmospheric Pressure Chemical Vapor Deposition. Appl. Surf. Sci. 2011, 257, 2428– 2431, DOI: 10.1016/j.apsusc.2010.11.180There is no corresponding record for this reference.
- 21Pierson, H. O. A Review of the Chemical Vapor Deposition (Cvd) of the Refractory Compounds Of Titanium - A Unique Family Of Coatings. Mater. Manuf. Process. 1993, 8, 519– 534, DOI: 10.1080/10426919308934855There is no corresponding record for this reference.
- 22Yokota, K.; Nakamura, K.; Kasuya, T.; Tamura, S.; Sugimoto, T.; Akamatsu, K.; Nakao, K.; Miyashita, F. Relationship between Hardness and Lattice Parameter for TiN Films Deposited on SUS 304 by an IBAD Technique. Surf. Coat. Technol. 2002, 158–159, 690– 693, DOI: 10.1016/S0257-8972(02)00245-1There is no corresponding record for this reference.
- 23D’Acunto, G.; Troian, A.; Kokkonen, E.; Rehman, F.; Liu, Y. P.; Yngman, S.; Yong, Z.; McKibbin, S. R.; Gallo, T.; Lind, E.; Schnadt, J.; Timm, R. Atomic Layer Deposition of Hafnium Oxide on InAs: Insight from Time-Resolved in Situ Studies. ACS Appl. Electron. Mater. 2020, 2, 3915– 3922, DOI: 10.1021/acsaelm.0c00775There is no corresponding record for this reference.
- 24D’Acunto, G.; Jones, R.; Pérez Ramírez, L.; Shayesteh, P.; Kokkonen, E.; Rehman, F.; Lim, F.; Bournel, F.; Gallet, J. J.; Timm, R.; Schnadt, J. Role of Temperature, Pressure, and Surface Oxygen Migration in the Initial Atomic Layer Deposition of HfO2 on Anatase TiO2(101). J. Phys. Chem. C 2022, 126, 12210– 12221, DOI: 10.1021/acs.jpcc.2c02683There is no corresponding record for this reference.
- 25Sobell, Z. C.; George, S. M. Electron-Enhanced Atomic Layer Deposition of Titanium Nitride Films Using an Ammonia Reactive Background Gas. Chem. Mater. 2022, 34, 9624– 9633, DOI: 10.1021/acs.chemmater.2c02341There is no corresponding record for this reference.
- 26Musschoot, J.; Xie, Q.; Deduytsche, D.; Van den Berghe, S.; Van Meirhaeghe, R. L.; Detavernier, C. Atomic Layer Deposition of Titanium Nitride from TDMAT Precursor. Microelectron. Eng. 2009, 86, 72– 77, DOI: 10.1016/j.mee.2008.09.036There is no corresponding record for this reference.
- 27Zhu, Y.; Zhou, Z.; Zhang, X.; Xu, R.; Wang, Y.; Xu, L.; Xiao, H.; Li, X.; Li, A.; Fang, G. Surface Reaction Mechanism of Atomic Layer Deposition of Titanium Nitride Using Tetrakis(Dimethylamino)Titanium and Ammonia. Surfaces and Interfaces 2023, 36, 102579 DOI: 10.1016/j.surfin.2022.102579There is no corresponding record for this reference.
- 28Cho, G.; Rhee, S.-W. Effect of the Amido Ti Precursors on the Atomic Layer Deposition of TiN with NH3. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2013, 31, 01A117 DOI: 10.1116/1.4764898There is no corresponding record for this reference.
- 29Park, K.; Mun, G.; Cho, J.; Lee, S.; Min, G.; Hong, S. Properties of TiN Thin Films Deposited by ALD Using TDMAT Precursor in Low-Temperature Processes. J. Semicond. Dispersion Technol. 2024, 23, 14– 20There is no corresponding record for this reference.
- 30Schnadt, J.; Knudsen, J.; Andersen, J. N.; Siegbahn, H.; Pietzsch, A.; Hennies, F.; Johansson, N.; Mårtensson, N.; Öhrwall, G.; Bahr, S.; Mähl, S.; Schaff, O. The New Ambient-Pressure X-Ray Photoelectron Spectroscopy Instrument at MAX-Lab. J. Synchrotron Radiat. 2012, 19, 701– 704, DOI: 10.1107/S0909049512032700There is no corresponding record for this reference.
- 31Knudsen, J.; Andersen, J. N.; Schnadt, J. A Versatile Instrument for Ambient Pressure X-Ray Photoelectron Spectroscopy: The Lund Cell Approach. Surf. Sci. 2016, 646, 160– 169, DOI: 10.1016/j.susc.2015.10.038There is no corresponding record for this reference.
- 32Kokkonen, E.; Kaipio, M.; Nieminen, H. E.; Rehman, F.; Miikkulainen, V.; Putkonen, M.; Ritala, M.; Huotari, S.; Schnadt, J.; Urpelainen, S. Ambient Pressure X-Ray Photoelectron Spectroscopy Setup for Synchrotron-Based in Situ and Operando Atomic Layer Deposition Research. Rev. Sci. Instrum. 2022, 93, 013905 DOI: 10.1063/5.0076993There is no corresponding record for this reference.
- 33Kokkonen, E.; Da Silva, F. L.; Mikkela, M. H.; Johansson, N.; Huang, S. W.; Lee, J. M.; Andersson, M.; Bartalesi, A.; N. Reinecke, B.; Handrup, K.; Tarawneh, H.; Sankari, R.; Knudsen, J.; Schnadt, J.; Sathe, C.; Urpelainen, S. Upgrade of the SPECIES Beamline at the MAX IV Laboratory. J. Synchrotron Radiat. 2021, 28, 588– 601, DOI: 10.1107/S1600577521000564There is no corresponding record for this reference.
- 34Tissot, H.; Gallet, J. J.; Bournel, F.; Naitabdi, A.; Pierucci, D.; Bondino, F.; Magnano, E.; Rochet, F.; Finocchi, F. Silicon Monomer Formation and Surface Patterning of Si(001)-2 × 1 Following Tetraethoxysilane Dissociative Adsorption at Room Temperature. J. Phys. Chem. C 2014, 118, 1887– 1893, DOI: 10.1021/jp407411kThere is no corresponding record for this reference.
- 35Kim, J. W.; Yeom, H. W.; Chung, Y. D.; Jeong, K.; Whang, C. N.; Lee, M. K.; Shin, H. J. Chemical Configuration of Nitrogen in Ultrathin Si Oxynitride on Si(100). Phys. Rev. B - Condens. Matter Mater. Phys. 2002, 66, 035312, DOI: 10.1103/PhysRevB.66.035312There is no corresponding record for this reference.
- 36Verdaguer, A.; Weis, C.; Oncins, G.; Ketteler, G.; Bluhm, H.; Salmero, M. Growth and Structure of Water on SiO2 Films on Si Investigated by Kelvin Probe Microscopy and in Situ X-Ray Spectroscopies. Langmuir 2007, 23, 9699– 9703, DOI: 10.1021/la700893wThere is no corresponding record for this reference.
- 37Kim, Y. K.; Hyun, S. L.; Yeom, H. W.; Ryoo, D. Y.; Huh, S. B.; Lee, J. G. Nitrogen Bonding Structure in Ultrathin Silicon Oxynitride Films on Si(100) Prepared by Plasma Nitridation. Phys. Rev. B - Condens. Matter Mater. Phys. 2004, 70, 165320 DOI: 10.1103/PhysRevB.70.165320There is no corresponding record for this reference.
- 38Simonsen, M. E.; Sønderby, C.; Li, Z.; Søgaard, E. G. XPS and FT-IR Investigation of Silicate Polymers. J. Mater. Sci. 2009, 44, 2079– 2088, DOI: 10.1007/s10853-009-3270-9There is no corresponding record for this reference.
- 39Namiki, A.; Tanimoto, K.; Nakamura, T.; Ohtake, N.; Suzaki, T. XPS Study on the Early Stages of Oxidation of Si(100) by Atomic Oxygen. Surf. Sci. 1989, 222, 530– 554, DOI: 10.1016/0039-6028(89)90377-4There is no corresponding record for this reference.
- 40Yang, J.; Wang, E. G. Reaction of Water on Silica Surfaces. Curr. Opin. Solid State Mater. Sci. 2006, 10, 33– 39, DOI: 10.1016/j.cossms.2006.02.001There is no corresponding record for this reference.
- 41Rignanese, G. M.; Charlier, J. C.; Gonze, X. First-Principles Molecular-Dynamics Investigation of the Hydration Mechanisms of the (0001) α-Quartz Surface. Phys. Chem. Chem. Phys. 2004, 6, 1920– 1925, DOI: 10.1039/B311842HThere is no corresponding record for this reference.
- 42Miyaji, F.; Iwai, M.; Kokubo, T.; Nakamura, T. Chemical Surface Treatment of Silicone for Inducing Its Bioactivity. J. Mater. Sci. Mater. Med. 1998, 9, 61– 65, DOI: 10.1023/A:1008886729050There is no corresponding record for this reference.
- 43Göpel, W.; Anderson, J. A.; Frankel, D.; Jaehnig, M.; Phillips, K.; Schäfer, J. A.; Rocker, G. Surface Defects of TiO2(110): A Combined XPS, XAES AND ELS Study. Surf. Sci. 1984, 139, 333– 346, DOI: 10.1016/0039-6028(84)90054-2There is no corresponding record for this reference.
- 44Saha, N. C.; Tompkins, H. G. Titanium Nitride Oxidation Chemistry: An x-Ray Photoelectron Spectroscopy Study. J. Appl. Phys. 1992, 72, 3072– 3079, DOI: 10.1063/1.351465There is no corresponding record for this reference.
- 45Head, A. R.; Chaudhary, S.; Olivieri, G.; Bournel, F.; Andersen, J. N.; Rochet, F.; Gallet, J. J.; Schnadt, J. Near Ambient Pressure X-Ray Photoelectron Spectroscopy Study of the Atomic Layer Deposition of TiO2 on RuO2(110). J. Phys. Chem. C 2016, 120, 243– 251, DOI: 10.1021/acs.jpcc.5b08699There is no corresponding record for this reference.
- 46Jansen, R. J. J.; van Bekkum, H. XPS of Nitrogen-Containing Functional Groups on Activated Carbon. Carbon N. Y. 1995, 33, 1021– 1027, DOI: 10.1016/0008-6223(95)00030-HThere is no corresponding record for this reference.
- 47Bruggeman, M.; Zelzer, M.; Dong, H.; Stamboulis, A. Processing and Interpretation of Core-Electron XPS Spectra of Complex Plasma-Treated Polyethylene-Based Surfaces Using a Theoretical Peak Model. Surf. Interface Anal. 2022, 54, 986– 1007, DOI: 10.1002/sia.7125There is no corresponding record for this reference.
- 48Cao, X.; Hamers, R. J. Silicon Surfaces as Electron Acceptors: Dative Bonding of Amines with Si(001) and Si(111) Surfaces. J. Am. Chem. Soc. 2001, 123, 10988– 10996, DOI: 10.1021/ja0100322There is no corresponding record for this reference.
- 49Peña-Juárez, M. G.; Robles-Martínez, M.; Méndez-Rodríguez, K. B.; López-Esparza, R.; Pérez, E.; Gonzalez-Calderon, J. A. Role of the Chemical Modification of Titanium Dioxide Surface on the Interaction with Silver Nanoparticles and the Capability to Enhance Antimicrobial Properties of Poly(Lactic Acid) Composites. Polym. Bull. 2021, 78, 2765– 2790, DOI: 10.1007/s00289-020-03235-yThere is no corresponding record for this reference.
- 50Ngoc Van, T. T.; Jang, D.; Jung, E.; Noh, H.; Moon, J.; Kil, D. S.; Chung, S. W.; Shong, B. Role of Cyclopentadienyl Ligands of Group 4 Precursors toward High-Temperature Atomic Layer Deposition. J. Phys. Chem. C 2022, 126, 18090– 18099, DOI: 10.1021/acs.jpcc.2c04425There is no corresponding record for this reference.
- 51Li, K.; Li, S.; Li, N.; Klein, T. M.; Dixon, D. A. Tetrakis(Ethylmethylamido) Hafnium Adsorption and Reaction on Hydrogen-Terminated Si(100) Surfaces. J. Phys. Chem. C 2011, 115, 18560– 18571, DOI: 10.1021/jp111600vThere is no corresponding record for this reference.
- 52Trenczek-Zajac, A.; Radecka, M.; Zakrzewska, K.; Brudnik, A.; Kusior, E.; Bourgeois, S.; de Lucas, M. C. M.; Imhoff, L. Structural and Electrical Properties of Magnetron Sputtered Ti(ON) Thin Films: The Case of TiN Doped in Situ with Oxygen. J. Power Sources 2009, 194, 93– 103, DOI: 10.1016/j.jpowsour.2008.12.112There is no corresponding record for this reference.
- 53Killampalli, A. S.; Ma, P. F.; Engstrom, J. R. The Reaction of Tetrakis (Dimethylamido) Titanium with Self-Assembled Alkyltrichlorosilane Monolayers Possessing. J. Am. Chem. Soc. 2005, 127, 6300– 6310, DOI: 10.1021/ja047922cThere is no corresponding record for this reference.
- 54Peng, Z.; Chen, G.; Zhao, Y.-P.; Zhang, X.; Song, Y.-T.; Shirkov, G.; Karamysheva, G.; Karamyshev, O.; Calabretta, L.; Caruso, A. Investigation of TiN Film on an RF Ceramic Window by Atomic Layer Deposition. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2020, 38, 052401 DOI: 10.1116/6.0000159There is no corresponding record for this reference.
- 55Nguyen, T. P.; Lefrant, S. XPS Study of SiO Thin Films and SiO-Metal Interfaces. J. Phys.: Condens. Matter 1989, 1, 5197– 5204, DOI: 10.1088/0953-8984/1/31/019There is no corresponding record for this reference.
- 56Desbiens, E.; Dolbec, R.; El Khakani, M. A. Reactive Pulsed Laser Deposition of High- k Silicon Dioxide and Silicon Oxynitride Thin Films for Gate-Dielectric Applications. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2002, 20, 1157– 1161, DOI: 10.1116/1.1467357There is no corresponding record for this reference.
- 57Ketteler, G.; Yamamoto, S.; Bluhm, H.; Andersson, K.; Starr, D. E.; Ogletree, D. F.; Ogasawara, H.; Nilsson, A.; Salmeron, M. The Nature of Water Nucleation Sites on TiO2(110) Surfaces Revealed by Ambient Pressure X-Ray Photoelectron Spectroscopy. J. Phys. Chem. C 2007, 111, 8278– 8282, DOI: 10.1021/jp068606iThere is no corresponding record for this reference.
- 58Walle, L. E.; Borg, A.; Johansson, E. M. J.; Plogmaker, S.; Rensmo, H.; Uvdal, P.; Sandell, A. Mixed Dissociative and Molecular Water Adsorption on Anatase TiO 2(101). J. Phys. Chem. C 2011, 115, 9545– 9550, DOI: 10.1021/jp111335wThere is no corresponding record for this reference.
- 59Walle, L. E.; Borg, A.; Uvdal, P.; Sandell, A. Experimental Evidence for Mixed Dissociative and Molecular Adsorption of Water on a Rutile TiO2 (110) Surface without Oxygen Vacancies. Phys. Rev. B - Condens. Matter Mater. Phys. 2009, 80, 235436 DOI: 10.1103/PhysRevB.80.235436There is no corresponding record for this reference.
- 60Esaka, F.; Furuya, K.; Shimada, H.; Imamura, M.; Matsubayashi, N.; Sato, H.; Nishijima, A.; Kawana, A.; Ichimura, H.; Kikuchi, T. Comparison of Surface Oxidation of Titanium Nitride and Chromium Nitride Films Studied by X-Ray Absorption and Photoelectron Spectroscopy. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 1997, 15, 2521– 2528, DOI: 10.1116/1.580764There is no corresponding record for this reference.
- 61Jaeger, D.; Patscheider, J. Single Crystalline Oxygen-Free Titanium Nitride by XPS. Surf. Sci. Spectra 2013, 20, 1, DOI: 10.1116/11.20121107There is no corresponding record for this reference.
- 62Chan, M. H.; Lu, F. H. X-Ray Photoelectron Spectroscopy Analyses of Titanium Oxynitride Films Prepared by Magnetron Sputtering Using Air/Ar Mixtures. Thin Solid Films 2009, 517, 5006– 5009, DOI: 10.1016/j.tsf.2009.03.100There is no corresponding record for this reference.
- 63Robinson, K. S.; Sherwood, P. M. A. X-Ray Photoelectron Spectroscopic Studies of the Surface of Sputter Ion Plated Films. Surf. Interface Anal. 1984, 6, 261– 266, DOI: 10.1002/sia.740060603There is no corresponding record for this reference.
- 64Cherono, S.; Chris-Okoro, I.; Liu, M.; Kim, R. S.; Nalawade, S.; Akande, W.; Maria-Diana, M.; Mahl, J.; Hale, C.; Yano, J.; Aravamudhan, S.; Crumlin, E.; Craciun, V.; Kumar, D. Transformation of TiN to TiNO Films via In-Situ Temperature-Dependent Oxygen Diffusion Process and Their Electrochemical Behavior. Metals (Basel). 2025, 15, 497, DOI: 10.3390/met15050497There is no corresponding record for this reference.
- 65Kuznetsov, M. V.; Zhuravlev, J. F.; Zhilyaev, V. A.; Gubanov, V. A. XPS Study of the Nitrides, Oxides and Oxynitrides of Titanium. J. Electron Spectrosc. Relat. Phenom. 1992, 58, 1, DOI: 10.1016/0368-2048(92)80001-OThere is no corresponding record for this reference.
- 66Kabachkov, E.; Kurkin, E.; Vershinin, N.; Balikhin, I.; Berestenko, V.; Michtchenko, A.; Shulga, Y. Synthesis and Properties of Pt/TiN Catalyst for Low-Temperature Air Purification from Carbon Monoxide. J. Adv. Mater. Technol. 2021, 6, 131– 143, DOI: 10.17277/jamt.2021.02.pp.131-143There is no corresponding record for this reference.
- 67George, S. M. Atomic Layer Deposition: An Overview. Chem. Rev. 2010, 110, 111– 131, DOI: 10.1021/cr900056bThere is no corresponding record for this reference.
- 68Corneille, J. S.; Chen, P. J.; Truong, C. M.; Oh, W. S.; Goodman, D. W. Surface Spectroscopic Studies of the Deposition of TiN Thin Films from Tetrakis-(Dimethylamido)-Titanium and Ammonia. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 1995, 13, 1116– 1120, DOI: 10.1116/1.579596There is no corresponding record for this reference.
- 69Xiong, H.; Wu, L.; Liu, Y.; Gao, T.; Li, K.; Long, Y.; Zhang, R.; Zhang, L.; Qiao, Z. A.; Huo, Q.; Ge, X.; Song, S.; Zhang, H. Controllable Synthesis of Mesoporous TiO2 Polymorphs with Tunable Crystal Structure for Enhanced Photocatalytic H2 Production. Adv. Energy Mater. 2019, 9, 1901634 DOI: 10.1002/aenm.201901634There is no corresponding record for this reference.
- 70Peng, F.; Cai, L.; Huang, L.; Yu, H.; Wang, H. Preparation of Nitrogen-Doped Titanium Dioxide with Visible-Light Photocatalytic Activity Using a Facile Hydrothermal Method. J. Phys. Chem. Solids 2008, 69, 1657– 1664, DOI: 10.1016/j.jpcs.2007.12.003There is no corresponding record for this reference.
- 71Zhang, H.; Wang, B.; Brown, B. Atomic Layer Deposition of Titanium Oxide and Nitride on Vertically Aligned Carbon Nanotubes for Energy Dense 3D Microsupercapacitors. Appl. Surf. Sci. 2020, 521, 146349 DOI: 10.1016/j.apsusc.2020.146349There is no corresponding record for this reference.
- 72Mpofu, P.; Niiranen, P.; Alm, O.; Lauridsen, J.; Larsson, T.; Pedersen, H. Atomic Layer Deposition of AlxTi1-XN via Co-Evaporation of Metal Precursors. J. Vac. Sci. Technol. A 2025, 43, 032405 DOI: 10.1116/6.0004420There is no corresponding record for this reference.
- 73Jintsugawa, O.; Sakuraba, M.; Matsuura, T.; Murota, J. Thermal Nitridation of Ultrathin SiO2 on Si by NH3. Surf. Interface Anal. 2002, 34, 456– 459, DOI: 10.1002/sia.1337There is no corresponding record for this reference.
- 74Graf, N.; Yegen, E.; Gross, T.; Lippitz, A.; Weigel, W.; Krakert, S.; Terfort, A.; Unger, W. E. S. XPS and NEXAFS Studies of Aliphatic and Aromatic Amine Species on Functionalized Surfaces. Surf. Sci. 2009, 603, 2849– 2860, DOI: 10.1016/j.susc.2009.07.029There is no corresponding record for this reference.
- 75Dietrich, P. M.; Graf, N.; Gross, T.; Lippitz, A.; Krakert, S.; Schüpbach, B.; Terfort, A.; Unger, W. E. S. Amine Species on Self-Assembled Monolayers of ω-Aminothiolates on Gold as Identified by XPS and NEXAFS Spectroscopy. Surf. Interface Anal. 2010, 42, 1184– 1187, DOI: 10.1002/sia.3224There is no corresponding record for this reference.
- 76Solís, R. R.; Gómez-Avilés, A.; Belver, C.; Rodriguez, J. J.; Bedia, J. Microwave-Assisted Synthesis of NH2-MIL-125(Ti) for the Solar Photocatalytic Degradation of Aqueous Emerging Pollutants in Batch and Continuous Tests. J. Environ. Chem. Eng. 2021, 9, 106230 DOI: 10.1016/j.jece.2021.106230There is no corresponding record for this reference.
- 77Endle, J. P.; Sun, Y.; White, J. M.; Ekerdt, J. G. X-Ray Photoelectron Spectroscopy Study of TiN Films Produced with Tetrakis(Dimethylamido)Titanium and Selected N-Containing Precursors on SiO2. J. Vac. Sci. Technol. A 1998, 16, 1262– 1267, DOI: 10.1116/1.581271There is no corresponding record for this reference.
- 78Quesada-Cabrera, R.; Sotelo-Vazquez, C.; Darr, J. A.; Parkin, I. P. Critical Influence of Surface Nitrogen Species on the Activity of N-Doped TiO2 Thin-Films during Photodegradation of Stearic Acid under UV Light Irradiation. Appl. Catal. B Environ. 2014, 160–161, 582– 588, DOI: 10.1016/j.apcatb.2014.06.010There is no corresponding record for this reference.
- 79Asghar, M.; Yousaf, M.; Huwaimel, B.; Iqbal, T.; Ahmed, I.; Qureshi, M. T.; Abrar, M.; Shafiq, M.; Almohammedi, A.; Hameed, R. A.; AlElaimi, M.; Maryam, M.; Afsheen, S. Synthesis of Biocompatible Coating on Ni-Cr Alloy by Cathodic Cage Plasma Processing Technique as Anti-Pathogenic Bacteria for Medicinal Applications. Phys. Scr. 2023, 98, 055920 DOI: 10.1088/1402-4896/acc908There is no corresponding record for this reference.
- 80Braic, L.; Vasilantonakis, N.; Mihai, A.; Villar Garcia, I. J.; Fearn, S.; Zou, B.; Alford, N. M. N.; Doiron, B.; Oulton, R. F.; Maier, S. A.; Zayats, A. V.; Petrov, P. K. Titanium Oxynitride Thin Films with Tunable Double Epsilon-Near-Zero Behavior for Nanophotonic Applications. ACS Appl. Mater. Interfaces 2017, 9, 29857– 29862, DOI: 10.1021/acsami.7b07660There is no corresponding record for this reference.
- 81Li, K.; Li, S.; Li, N.; Dixon, D. A.; Klein, T. M. Tetrakis(Dimethylamido)Hafnium Adsorption and Reaction on Hydrogen Terminated Si(100) Surfaces. J. Phys. Chem. C 2010, 114, 14061– 14075, DOI: 10.1021/jp101363rThere is no corresponding record for this reference.
- 82Ruhl, G.; Rehmet, R.; Knfžvá, M.; Vepřek, S. In Situ XPS Studies of the Deposition of Thin Films from Tetrakis(Dimethylamido)Titanium Organometallic Precursor for Diffusion Barriers. MRS Online Proc. Libr. 1993, 309, 461– 466, DOI: 10.1557/PROC-309-461There is no corresponding record for this reference.
- 83NIST NIST Standard Reference Database Number 20. https://dx.doi.org/10.18434/T4T88K (accessed Apr 7, 2025).There is no corresponding record for this reference.
- 84Longrie, D.; Deduytsche, D.; Haemers, J.; Smet, P. F.; Driesen, K.; Detavernier, C. Thermal and Plasma-Enhanced Atomic Layer Deposition of TiN Using TDMAT and NH3 on Particles Agitated in a Rotary Reactor. ACS Appl. Mater. Interfaces 2014, 6, 7316– 7324, DOI: 10.1021/am5007222There is no corresponding record for this reference.
- 85Elam, J. W.; Schuisky, M.; Ferguson, J. D.; George, S. M. Surface Chemistry and Film Growth during TiN Atomic Layer Deposition Using TDMAT and NH3. Thin Solid Films 2003, 436, 145– 156, DOI: 10.1016/S0040-6090(03)00533-9There is no corresponding record for this reference.
- 86Biesinger, M. C.; Lau, L. W. M.; Gerson, A. R.; Smart, R. S. C. Resolving Surface Chemical States in XPS Analysis of First Row Transition Metals, Oxides and Hydroxides: Sc, Ti, V, Cu and Zn. Appl. Surf. Sci. 2010, 257, 887– 898, DOI: 10.1016/j.apsusc.2010.07.086There is no corresponding record for this reference.
- 87Methaapanon, R.; Bent, S. F. Comparative Study of Titanium Dioxide Atomic Layer Deposition on Silicon Dioxide and Hydrogen-Terminated Silicon. J. Phys. Chem. C 2010, 114, 10498– 10504, DOI: 10.1021/jp1013303There is no corresponding record for this reference.
- 88Weiller, B. H. Chemical Vapor Deposition of TiN from Tetrakis(Dimethylamido)Titanium and Ammonia: Kinetics and Mechanistic Studies of the Gas-Phase Chemistry. J. Am. Chem. Soc. 1996, 118, 4975– 4983, DOI: 10.1021/ja953468oThere is no corresponding record for this reference.
- 89Hughes, K. J.; Engstrom, J. R. Nucleation Delay in Atomic Layer Deposition on a Thin Organic Layer and the Role of Reaction Thermochemistry. J. Vac. Sci. Technol. A Vacuum, Surfaces, Film. 2012, 30, 01A102 DOI: 10.1116/1.3625564There is no corresponding record for this reference.
- 90Mpofu, P.; Hafdi, H.; Lauridsen, J.; Alm, O.; Larsson, T.; Pedersen, H. A Mass Spectrometrical Surface Chemistry Study of Aluminum Nitride ALD from Tris-Dimethylamido Aluminum and Ammonia. Mater. Adv. 2024, 5, 9259– 9269, DOI: 10.1039/D4MA00922CThere is no corresponding record for this reference.
- 91Lee, W. J.; Yun, E. Y.; Lee, H. B. R.; Hong, S. W.; Kwon, S. H. Dataset for TiN Thin Films Prepared by Plasma-Enhanced Atomic Layer Deposition Using Tetrakis(Dimethylamino)Titanium (TDMAT) and Titanium Tetrachloride (TiCl4) Precursor. Data Br. 2020, 31, 105777 DOI: 10.1016/j.dib.2020.105777There is no corresponding record for this reference.
- 92Yun, J.; Park, M.; Rhee, S. Comparison of Tetrakis(Dimethylamido)Titanium and Tetrakis(Diethylamido)Titanium as Precursors for Metallorganic Chemical Vapor Deposition of Titanium Nitride. J. Electrochem. Soc. 1999, 146, 1804– 1808, DOI: 10.1149/1.1391847There is no corresponding record for this reference.
- 93Sen, K.; Banu, T.; Debnath, T.; Ghosh, D.; Das, A. K. Towards a Comprehensive Understanding of the Chemical Vapor Deposition of Titanium Nitride Using Ti(NMe2)4: A Density Functional Theory Approach. Dalt. Trans. 2014, 43, 8877– 8887, DOI: 10.1039/c4dt00690aThere is no corresponding record for this reference.
- 94Dubois, L. H.; Zegarski, B. R.; Girolami, G. S. Infrared Studies of the Surface and Gas Phase Reactions Leading to the Growth of Titanium Nitride Thin Films from Tetrakis(Dimethylamido)Titanium and Ammonia. J. Electrochem. Soc. 1992, 139, 3603– 3609, DOI: 10.1149/1.2087327There is no corresponding record for this reference.
- 95Mpofu, P.; Hafdi, H.; Niiranen, P.; Lauridsen, J.; Alm, O.; Larsson, T.; Pedersen, H. Surface Chemistry in Atomic Layer Deposition of AlN Thin Films from Al(CH3)3 and NH3 Studied by Mass Spectrometry. J. Mater. Chem. C 2024, 12, 12818– 12824, DOI: 10.1039/D4TC01867BThere is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.chemmater.5c02974.
Additional details from the APXPS measurements (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.



