
From First-Principles to Quantum Electrodynamics: Pushing the Limits of Theory with the Hydrogen MoleculeClick to copy article linkArticle link copied!
- Krzysztof Pachucki*Krzysztof Pachucki*Email: [email protected]Faculty of Physics, University of Warsaw, Pasteura 5, 02-093 Warsaw, PolandMore by Krzysztof Pachucki
- Jacek Komasa*Jacek Komasa*Email: [email protected]Faculty of Chemistry, Adam Mickiewicz University, Uniwersytetu Poznanskiego 8, 61-614 Poznań, PolandMore by Jacek Komasa
Abstract
Modern spectroscopic techniques enable the determination of the spacing between rovibrational levels of H2 with a relative accuracy of approximately 10–11. At this extreme level of precision, subtle quantum electrodynamic (QED) effects, such as electron self-interaction and vacuum polarization, are probed. A theoretical model aiming to achieve similar accuracy must precisely describe not only these relatively small QED effects but also the more significant contributions related to electron correlation, coupling between electronic and nuclear motions, and relativistic effects. Although the hydrogen molecule exhibits most of the phenomena found in larger molecules, it is simple enough to meet the requirements mentioned above. In this article, we report on enhancements to the current capabilities of quantum mechanical calculations for the hydrogen molecule. We present a method based on exponential functions that fully captures electron correlation or, more broadly, interparticle correlation, enabling a comprehensive description of effects related to nuclear motion. Specifically, we solve the four-particle Schrödinger equation without invoking commonly used approximations such as the one-electron or the Born–Oppenheimer approximation. The only source of nonrelativistic energy error comes from the finite size of the basis set. The explicitly correlated nonadiabatic wave function used here is then employed to determine the relativistic and QED effects. As a result, the dissociation energy for the lowest rovibrational levels in the electronic ground state of H2 has been obtained with a relative accuracy of 7 × 10–10, while the frequencies of intervals between these levels have been determined with sub-MHz accuracy, corresponding to a relative accuracy of 3 × 10–9. In consequence, the discrepancies between the highest precision measurements and earlier theoretical predictions have been resolved.
This publication is licensed for personal use by The American Chemical Society.
Special Issue
Published as part of Journal of Chemical Theory and Computation special issue “100 Years of Quantum Mechanics-All About Molecules”.
1. Introduction
2. Theoretical Background
2.1. The Wave Function
2.2. Relativistic Correction, E(4)
2.3. QED Correction, E(5)
2.4. Higher-Order QED Correction, E(6)
2.5. Estimation of E(7)
2.6. Atomic Limits
3. Results
3.1. Dissociation Energy
| component | D0,0 | D1,0 | D2,0 |
|---|---|---|---|
| E(2) | 36,118.797744716(4)a | 31,957.63367420(2) | 28,031.79525303(2) |
| E(4) | –0.531217263(61) | –0.554783333(62) | –0.572886764(67) |
| Efs(4) | –0.000030900(33) | –0.000027745(30) | –0.000024797(27) |
| E(5) | –0.19491021(15) | –0.173629(10) | –0.153814(20) |
| E(6) | –0.0020577(66) | –0.0018664(60) | –0.0016868(54) |
| E(7) | 0.000101(25) | 0.000090(23) | 0.000081(20) |
| total | 36,118.069630(26) | 31,956.903458(26) | 28,031.066922(29) |
| component | D0,1 | D1,1 | D2,1 |
|---|---|---|---|
| E(2) | 36,000.31248423(1) | 31,845.06061442(1) | 27,925.00480794(2) |
| E(4) | –0.53380004(31) | –0.55714556(38) | –0.57502713(41) |
| Efs(4) | –0.000030749(33) | –0.000027603(30) | –0.000024664(27) |
| E(5) | –0.19388906(52) | –0.172669(11) | –0.152912(20) |
| E(6) | –0.0020488(66) | –0.0018581(60) | –0.0016789(54) |
| E(7) | 0.000100(25) | 0.000090(22) | 0.000080(20) |
| total | 35,999.582816(26) | 31,844.329004(25) | 27,924.275245(29) |
| component | D0,2 | D1,2 | D2,2 |
|---|---|---|---|
| E(2) | 35,764.42923080(1) | 31,620.96551340(1) | 27,712.44065580(2) |
| E(4) | –0.53890776(38) | –0.56181401(38) | –0.57925386(41) |
| Efs(4) | –0.000030451(33) | –0.000027322(29) | –0.000024399(26) |
| E(5) | –0.1918636(15) | –0.170765(12) | –0.151123(21) |
| E(6) | –0.0020312(65) | –0.0018415(59) | –0.0016633(53) |
| E(7) | 0.000099(25) | 0.000089(22) | 0.000079(20) |
| total | 35,763.696497(26) | 31,620.231155(26) | 27,711.708670(29) |
| component | D0,3 | D1,3 | D2,3 |
|---|---|---|---|
| E(2) | 35,413.28801899(1) | 31,287.41651194(2) | 27,396.10305521(2) |
| E(4) | –0.54642754(38) | –0.56868046(38) | –0.58546271(41) |
| Efs(4) | –0.000030009(32) | –0.000026906(29) | –0.000024007(26) |
| E(5) | –0.1888668(30) | –0.167948(13) | –0.148477(22) |
| E(6) | –0.0020052(64) | –0.0018169(58) | –0.0016401(53) |
| E(7) | 0.000098(24) | 0.000088(22) | 0.000078(20) |
| total | 35,412.550787(25) | 31,286.678128(26) | 27,395.367529(30) |
| component | D0,4 | D1,4 | D2,4 |
|---|---|---|---|
| E(2) | 34,950.01523845(2) | 30,847.43371054(2) | 26,978.91200753(2) |
| E(4) | –0.55619744(35) | –0.57758917(37) | –0.59350370(40) |
| Efs(4) | –0.000029431(32) | –0.000026362(28) | –0.000023495(25) |
| E(5) | –0.1849460(49) | –0.164264(15) | –0.145018(24) |
| E(6) | –0.0019711(63) | –0.0017848(57) | –0.0016098(52) |
| E(7) | 0.000096(24) | 0.000086(21) | 0.000076(19) |
| total | 34,949.272190(25) | 30,846.690132(26) | 26,978.171929(31) |
This energy differs from that in our previous studies (11,12,74) due to differences in the physical constants used.
Components of Dv,J related to subsequent terms E(n) of the α-expansion (1) are also included.
3.2. Rovibrational Transition Energy
| component | S0(0): (0, 2) → (0, 0) | S0(1): (0, 3) → (0, 1) | Q1(1): (1, 1) → (0, 1) |
|---|---|---|---|
| ν(2) | 10,623,700.7825(3) | 17,598,550.7339(4) | 124,571,317.1657(4) |
| ν(4) | 230.555(11) | 378.562(11) | 699.881(11) |
| νfs(4) | –0.01346(2) | –0.02219(3) | –0.0943(1) |
| ν(5) | –91.335(47) | –150.565(77) | –636.16(33) |
| ν(6) | –0.793(3) | –1.308(4) | –5.719(18) |
| ν(7) | 0.040(10) | 0.070(17) | 0.310(78) |
| theory | 10,623,839.229(49) | 17,598,777.469(80) | 124,571,375.38(34) |
| experiment | 10,623,839.09(39)a | 17,598,777.46(75)a | 124,571,374.73(31)b |
| difference | 0.14(39) | 0.01(75) | 0.65(46) |
| component | S1(0): (1, 2) → (0, 0) | Q2(1): (2, 1) → (0, 1) | Q2(2): (2, 2) → (0, 2) |
|---|---|---|---|
| ν(2) | 134,841,618.0297(4) | 242,091,633.7378(5) | 241,392,544.6686(5) |
| ν(4) | 917.267(11) | 1235.957(12) | 1209.546(12) |
| νfs(4) | –0.1072(2) | –0.1824(3) | –0.1814(3) |
| ν(5) | –723.86(37) | –1228.46(63) | –1221.38(63) |
| ν(6) | –6.482(21) | –11.089(36) | –11.031(35) |
| ν(7) | 0.340(85) | 0.59(15) | 0.59(15) |
| theory | 134,841,805.19(38) | 242,091,630.55(65) | 241,392,522.22(65) |
| experiment | 134,841,805.102(15)c | 242,091,630.140(9)d | 241,392,522.00(34)a |
| difference | 0.09(38) | 0.41(65) | 0.22(73) |
| component | Q2(3): (2, 3) → (0, 3) | S2(0): (2, 2) → (0, 0) | S2(1): (2, 3) → (0, 1) |
|---|---|---|---|
| ν(2) | 240,349,158.6533(5) | 252,016,245.4511(5) | 257,947,709.3872(5) |
| ν(4) | 1170.245(12) | 1440.101(12) | 1548.807(12) |
| νfs(4) | –0.1799(3) | –0.1949(3) | –0.2021(3) |
| ν(5) | –1210.85(62) | –1312.71(68) | –1361.42(70) |
| ν(6) | –10.945(35) | –11.824(38) | –12.253(39) |
| ν(7) | 0.59(15) | 0.63(16) | 0.66(17) |
| theory | 240,349,107.52(64) | 25,2016,361.45(69) | 25,7947,884.98(72) |
| experiment | 240,349,107.15(71)a | 25,2016,361.164(8)e | 25,7947,884.597(30)a |
| difference | 0.36(96) | 0.28(69) | 0.39(72) |
Fleurbaey et al., 2023. (69)
Lamperti et al., 2023. (70)
Stankiewicz et al., 2025. (73)
Diouf et al., 2024. (85)
Cozijn et al., 2023. (71)
The components ν(n) = ΔE(n) of the theoretical frequency, corresponding to subsequent terms of the α-expansion (1), are also included. CODATA 2022-recommended physical constants were used. (80)
Figure 1
Figure 1. Differences between theoretically predicted and experimental line positions shown against the ±1 MHz error band (in blue) and the theoretical error band (in green). The error bars around the dots come from the experiment.
4. Conclusion
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jctc.5c01702.
Convergence of relativistic expectation values for individual rovibrational levels of H2 (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.
Acknowledgments
This research was supported by the National Science Center (Poland) Grant No. 2021/41/B/ST4/00089. A computer grant from the Poznań Supercomputing and Networking Center was used to carry out the numerical calculations.
References
This article references 90 other publications.
- 1Heitler, W.; London, F. Wechselwirkung neutraler Atome und homeopolare Bindung nach der Quantenmechanik. Z. Phys. 1927, 44, 455, DOI: 10.1007/BF01397394Google ScholarThere is no corresponding record for this reference.
- 2James, H. M.; Coolidge, A. S. The Ground State of the Hydrogen Molecule. J. Chem. Phys. 1933, 1, 825, DOI: 10.1063/1.1749252Google ScholarThere is no corresponding record for this reference.
- 3Kolos, W.; Roothaan, C. C. J. Accurate Electronic Wave Functions for the H2 Molecule. Rev. Mod. Phys. 1960, 32, 219– 232, DOI: 10.1103/RevModPhys.32.219Google ScholarThere is no corresponding record for this reference.
- 4Kołos, W.; Wolniewicz, L. The coupling between electronic and nuclear motion and the relativistic effects in the ground state of the H2 molecule. Acta Phys. Polonica 1961, 20, 129– 140Google ScholarThere is no corresponding record for this reference.
- 5Kołos, W.; Wolniewicz, L. Accurate Adiabatic Treatment of the Ground State of the Hydrogen Molecule. J. Chem. Phys. 1964, 41, 3663– 3673, DOI: 10.1063/1.1725796Google ScholarThere is no corresponding record for this reference.
- 6Kołos, W.; Wolniewicz, L. Accurate Computation of Vibronic Energies and of Some Expectation Values for H2, D2, and T2. J. Chem. Phys. 1964, 41, 3674– 3678, DOI: 10.1063/1.1725797Google ScholarThere is no corresponding record for this reference.
- 7Pachucki, K.; Komasa, J. Nonadiabatic corrections to rovibrational levels of H2. J. Chem. Phys. 2009, 130, 164113, DOI: 10.1063/1.3114680Google ScholarThere is no corresponding record for this reference.
- 8Pachucki, K.; Komasa, J. Leading order nonadiabatic corrections to rovibrational levels of H2, D2, and T2. J. Chem. Phys. 2015, 143, 034111, DOI: 10.1063/1.4927079Google ScholarThere is no corresponding record for this reference.
- 9Kołos, W.; Wolniewicz, L. Nonadiabatic Theory for Diatomic Molecules and Its Application to the Hydrogen Molecule. Rev. Mod. Phys. 1963, 35, 473, DOI: 10.1103/RevModPhys.35.473Google ScholarThere is no corresponding record for this reference.
- 10Kinghorn, D. B.; Adamowicz, L. Improved Nonadiabatic Ground-State Energy Upper Bound for Dihydrogen. Phys. Rev. Lett. 1999, 83, 2541– 2543, DOI: 10.1103/PhysRevLett.83.2541Google ScholarThere is no corresponding record for this reference.
- 11Puchalski, M.; Komasa, J.; Czachorowski, P.; Pachucki, K. Nonadiabatic QED Correction to the Dissociation Energy of the Hydrogen Molecule. Phys. Rev. Lett. 2019, 122, 103003, DOI: 10.1103/PhysRevLett.122.103003Google ScholarThere is no corresponding record for this reference.
- 12Pachucki, K.; Komasa, J. Nonadiabatic rotational states of the hydrogen molecule. Phys. Chem. Chem. Phys. 2018, 20, 247– 255, DOI: 10.1039/C7CP06516GGoogle ScholarThere is no corresponding record for this reference.
- 13Wolniewicz, L. Relativistic energies of the ground state of the hydrogen molecule. J. Chem. Phys. 1993, 99, 1851, DOI: 10.1063/1.465303Google ScholarThere is no corresponding record for this reference.
- 14Piszczatowski, K.; Łach, G.; Przybytek, M.; Komasa, J.; Pachucki, K.; Jeziorski, B. Theoretical Determination of the Dissociation Energy of Molecular Hydrogen. J. Chem. Theory Comput. 2009, 5, 3039, DOI: 10.1021/ct900391pGoogle ScholarThere is no corresponding record for this reference.
- 15Komasa, J.; Piszczatowski, K.; Łach, G.; Przybytek, M.; Jeziorski, B.; Pachucki, K. Quantum Electrodynamics Effects in Rovibrational Spectra of Molecular Hydrogen. J. Chem. Theory Comput. 2011, 7, 3105– 3115, DOI: 10.1021/ct200438tGoogle ScholarThere is no corresponding record for this reference.
- 16Puchalski, M.; Komasa, J.; Czachorowski, P.; Pachucki, K. Complete α6 m Corrections to the Ground State of H2. Phys. Rev. Lett. 2016, 117, 263002, DOI: 10.1103/PhysRevLett.117.263002Google ScholarThere is no corresponding record for this reference.
- 17Czachorowski, P.; Puchalski, M.; Komasa, J.; Pachucki, K. Nonadiabatic relativistic correction in H2, D2, and HD. Phys. Rev. A 2018, 98, 052506, DOI: 10.1103/PhysRevA.98.052506Google ScholarThere is no corresponding record for this reference.
- 18Siłkowski, M.; Pachucki, K.; Komasa, J.; Puchalski, M. Leading-order QED effects in the ground electronic state of molecular hydrogen. Phys. Rev. A 2023, 107, 032807, DOI: 10.1103/PhysRevA.107.032807Google ScholarThere is no corresponding record for this reference.
- 19Wang, L. M.; Yan, Z.-C. Relativistic corrections to the ground state of H2 calculated without using the Born-Oppenheimer approximation. Phys. Rev. A 2018, 97, 060501, DOI: 10.1103/PhysRevA.97.060501Google ScholarThere is no corresponding record for this reference.
- 20Puchalski, M.; Moszynski, R.; Komasa, J. In Polish Quantum Chemistry from Kołos to Now. In Advances in Quantum Chemistry; Musiał, M., Grabowski, I., Eds.; Academic Press, 2023; Vol. 87, pp 1– 36.Google ScholarThere is no corresponding record for this reference.
- 21Pachucki, K.; Komasa, J. Magnetic dipole transitions in the hydrogen molecule. Phys. Rev. A 2011, 83, 032501, DOI: 10.1103/PhysRevA.83.032501Google ScholarThere is no corresponding record for this reference.
- 22Margolis, J.; Hunt, G. On the level of H2 quadrupole absorption in the Jovian atmosphere. Icarus 1973, 18, 593– 598, DOI: 10.1016/0019-1035(73)90061-4Google ScholarThere is no corresponding record for this reference.
- 23Roueff, E.; Abgrall, H.; Czachorowski, P.; Pachucki, K.; Puchalski, M.; Komasa, J. The full infrared spectrum of molecular hydrogen. Astron. Astrophys. 2019, 630, A58, DOI: 10.1051/0004-6361/201936249Google ScholarThere is no corresponding record for this reference.
- 24Tchernyshyov, K. A Detection of H2 in a High-velocity Cloud toward the Large Magellanic Cloud. Astrophys. J. 2022, 931, 78, DOI: 10.3847/1538-4357/ac68e0Google ScholarThere is no corresponding record for this reference.
- 25Jacob, A. Small molecules, big impact: a tale of hydrides past, present, and future. Astrophys. Space Sci. 2023, 368, 76, DOI: 10.1007/s10509-023-04229-8Google ScholarThere is no corresponding record for this reference.
- 26Tennyson, J. The 2024 release of the ExoMol database: Molecular line lists for exoplanet and other hot atmospheres. J. Quant. Spectrosc. Radiat. Transfer 2024, 326, 109083, DOI: 10.1016/j.jqsrt.2024.109083Google ScholarThere is no corresponding record for this reference.
- 27Bruno, D. Uncertainty quantification of a collisional-radiative model for hydrogen plasmas. Phys. Scr. 2025, 100, 035611, DOI: 10.1088/1402-4896/adb227Google ScholarThere is no corresponding record for this reference.
- 28Zúñiga, J.; Bastida, A.; Requena, A.; Cerezo, J. Ab Initio Partition Functions and Thermodynamic Quantities for the Molecular Hydrogen Isotopologues. J. Phys. Chem. A 2021, 125, 9226– 9241, DOI: 10.1021/acs.jpca.1c06468Google ScholarThere is no corresponding record for this reference.
- 29Leachman, J. W.; Wilhelmsen, Ø.; Matveev, K. I. Cool Fuel: The Science and Engineering of Cryogenic Hydrogen; Oxford University Press, 2025.Google ScholarThere is no corresponding record for this reference.
- 30Puchalski, M.; Komasa, J.; Pachucki, K. Hyperfine Structure of the First Rotational Level in H2, D2 and HD Molecules and the Deuteron Quadrupole Moment. Phys. Rev. Lett. 2020, 125, 253001, DOI: 10.1103/PhysRevLett.125.253001Google ScholarThere is no corresponding record for this reference.
- 31Puchalski, M.; Komasa, J.; Spyszkiewicz, A.; Pachucki, K. Nuclear magnetic shielding in HD and HT. Phys. Rev. A 2022, 105, 042802, DOI: 10.1103/PhysRevA.105.042802Google ScholarThere is no corresponding record for this reference.
- 32Kassi, S.; Campargue, A. Electric quadrupole and dipole transitions of the first overtone band of HD by CRDS between 1.45 and 1.33 μm. J. Mol. Spectrosc. 2011, 267, 36, DOI: 10.1016/j.jms.2011.02.001Google ScholarThere is no corresponding record for this reference.
- 33Campargue, A.; Kassi, S.; Pachucki, K.; Komasa, J. The absorption spectrum of H2: CRDS measurements of the (2–0) band, review of the literature data and accurate ab initio line list up to 35000 cm–1. Phys. Chem. Chem. Phys. 2012, 14, 802, DOI: 10.1039/C1CP22912EGoogle ScholarThere is no corresponding record for this reference.
- 34Kassi, S.; Campargue, A.; Pachucki, K.; Komasa, J. The absorption spectrum of D2: Ultrasensitive cavity ring down spectroscopy of the (2–0) band near 1.7 μm and accurate ab initio line list up to 24 000 cm–1. J. Chem. Phys. 2012, 136, 184309, DOI: 10.1063/1.4707708Google ScholarThere is no corresponding record for this reference.
- 35Cheng, C.-F.; Sun, Y. R.; Pan, H.; Wang, J.; Liu, A.-W.; Campargue, A.; Hu, S.-M. Electric-quadrupole transition of H2 determined to 10–9 precision. Phys. Rev. A 2012, 85, 024501, DOI: 10.1103/PhysRevA.85.024501Google ScholarThere is no corresponding record for this reference.
- 36Dickenson, G. D.; Niu, M. L.; Salumbides, E. J.; Komasa, J.; Eikema, K. S. E.; Pachucki, K.; Ubachs, W. Fundamental Vibration of Molecular Hydrogen. Phys. Rev. Lett. 2013, 110, 193601, DOI: 10.1103/PhysRevLett.110.193601Google ScholarThere is no corresponding record for this reference.
- 37Niu, M.; Salumbides, E.; Dickenson, G.; Eikema, K.; Ubachs, W. Precision spectroscopy of the rovibrational splittings in H2, HD and D2. J. Mol. Spectrosc. 2014, 300, 44– 54, DOI: 10.1016/j.jms.2014.03.011Google ScholarThere is no corresponding record for this reference.
- 38Tan, Y.; Wang, J.; Cheng, C.-F.; Zhao, X.-Q.; Liu, A.-W.; Hu, S.-M. Cavity ring-down spectroscopy of the electric quadrupole transitions of in the 784–852 nm region. J. Mol. Spectrosc. 2014, 300, 60– 64, DOI: 10.1016/j.jms.2014.03.010Google ScholarThere is no corresponding record for this reference.
- 39Kassi, S.; Campargue, A. Electric quadrupole transitions and collision-induced absorption in the region of the first overtone band of H2 near 1.25μm. J. Mol. Spectrosc. 2014, 300, 55– 59, DOI: 10.1016/j.jms.2014.03.022Google ScholarThere is no corresponding record for this reference.
- 40Mondelain, D.; Kassi, S.; Sala, T.; Romanini, D.; Gatti, D.; Campargue, A. Sub-MHz accuracy measurement of the S(2) 2–0 transition frequency of D2 by Comb-Assisted Cavity Ring Down spectroscopy. J. Mol. Spectrosc. 2016, 326, 5– 8, DOI: 10.1016/j.jms.2016.02.008Google ScholarThere is no corresponding record for this reference.
- 41Wcisło, P.; Thibault, F.; Zaborowski, M.; Wójtewicz, S.; Cygan, A.; Kowzan, G.; Masłowski, P.; Komasa, J.; Puchalski, M.; Pachucki, K.; Ciuryło, R.; Lisak, D. Accurate deuterium spectroscopy for fundamental studies. J. Quant. Spectrosc. Radiat. Transfer 2018, 213, 41– 51, DOI: 10.1016/j.jqsrt.2018.04.011Google ScholarThere is no corresponding record for this reference.
- 42Trivikram, T. M.; Schlösser, M.; Ubachs, W.; Salumbides, E. J. Relativistic and QED Effects in the Fundamental Vibration of T2. Phys. Rev. Lett. 2018, 120, 163002, DOI: 10.1103/PhysRevLett.120.163002Google ScholarThere is no corresponding record for this reference.
- 43Tao, L.-G.; Liu, A.-W.; Pachucki, K.; Komasa, J.; Sun, Y. R.; Wang, J.; Hu, S.-M. Toward a Determination of the Proton-Electron Mass Ratio from the Lamb-Dip Measurement of HD. Phys. Rev. Lett. 2018, 120, 153001, DOI: 10.1103/PhysRevLett.120.153001Google ScholarThere is no corresponding record for this reference.
- 44Fasci, E.; Castrillo, A.; Dinesan, H.; Gravina, S.; Moretti, L.; Gianfrani, L. Precision spectroscopy of HD at 1.38μm. Phys. Rev. A 2018, 98, 022516, DOI: 10.1103/PhysRevA.98.022516Google ScholarThere is no corresponding record for this reference.
- 45Martinez, R. Z.; Bermejo, D.; Wcisło, P.; Thibault, F. Accurate wavenumber measurements for the S0(0), S0(1), and S0(2) pure rotational Raman lines of D2. J. Raman Spectrosc. 2019, 50, 127– 129, DOI: 10.1002/jrs.5499Google ScholarThere is no corresponding record for this reference.
- 46Hölsch, N.; Beyer, M.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W.; Jungen, C.; Merkt, F. Benchmarking Theory with an Improved Measurement of the Ionization and Dissociation Energies of H2. Phys. Rev. Lett. 2019, 122, 103002, DOI: 10.1103/PhysRevLett.122.103002Google ScholarThere is no corresponding record for this reference.
- 47Beyer, M.; Hölsch, N.; Hussels, J.; Cheng, C.-F.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W.; Jungen, C.; Merkt, F. Determination of the Interval between the Ground States of Para- and Ortho-H2. Phys. Rev. Lett. 2019, 123, 163002, DOI: 10.1103/PhysRevLett.123.163002Google ScholarThere is no corresponding record for this reference.
- 48Lai, K.-F.; Czachorowski, P.; Schlösser, M.; Puchalski, M.; Komasa, J.; Pachucki, K.; Ubachs, W.; Salumbides, E. J. Precision tests of nonadiabatic perturbation theory with measurements on the DT molecule. Phys. Rev. Res. 2019, 1, 033124, DOI: 10.1103/PhysRevResearch.1.033124Google ScholarThere is no corresponding record for this reference.
- 49Lai, K.-F.; Hermann, V.; Trivikram, M.; Diouf, M.; Schlösser, M.; Ubachs, W.; Salumbides, E. Precision measurement of the fundamental vibrational frequencies of tritium-bearing hydrogen molecules: T2, DT, HT. Phys. Chem. Chem. Phys. 2020, 22, 8973– 8987, DOI: 10.1039/D0CP00596GGoogle ScholarThere is no corresponding record for this reference.
- 50Mondelain, D.; Kassi, S.; Campargue, A. Transition frequencies in the (2–0) band of D2 with MHz accuracy. J. Quant. Spectrosc. Radiat. Transfer 2020, 253, 107020, DOI: 10.1016/j.jqsrt.2020.107020Google ScholarThere is no corresponding record for this reference.
- 51Jóźwiak, H.; Cybulski, H.; Wcisło, P. Hyperfine structure of quadrupole rovibrational transitions in tritium-bearing hydrogen isotopologues. J. Quant. Spectrosc. Radiat. Transfer 2020, 256, 107255, DOI: 10.1016/j.jqsrt.2020.107255Google ScholarThere is no corresponding record for this reference.
- 52Kassi, S.; Fleurbaey, H.; Campargue, A. First detection and absolute transition frequencies in the (3–0) band of D2. J. Chem. Phys. 2024, 160, 094306, DOI: 10.1063/5.0196903Google ScholarThere is no corresponding record for this reference.
- 53Salumbides, E. J.; Dickenson, G. D.; Ivanov, T. I.; Ubachs, W. QED Effects in Molecules: Test on Rotational Quantum States of H2. Phys. Rev. Lett. 2011, 107, 043005, DOI: 10.1103/PhysRevLett.107.043005Google ScholarThere is no corresponding record for this reference.
- 54Salumbides, E. J.; Koelemeij, J. C. J.; Komasa, J.; Pachucki, K.; Eikema, K. S. E.; Ubachs, W. Bounds on fifth forces from precision measurements on molecules. Phys. Rev. D 2013, 87, 112008, DOI: 10.1103/PhysRevD.87.112008Google ScholarThere is no corresponding record for this reference.
- 55Salumbides, E. J.; Schellekens, A. N.; Gato-Rivera, B.; Ubachs, W. Constraints on extra dimensions from precision molecular spectroscopy. New J. Phys. 2015, 17, 033015, DOI: 10.1088/1367-2630/17/3/033015Google ScholarThere is no corresponding record for this reference.
- 56Ubachs, W.; Koelemeij, J.; Eikema, K.; Salumbides, E. Physics beyond the Standard Model from hydrogen spectroscopy. J. Mol. Spectrosc. 2016, 320, 1– 12, DOI: 10.1016/j.jms.2015.12.003Google ScholarThere is no corresponding record for this reference.
- 57Drouin, B. J.; Yu, S.; Pearson, J. C.; Gupta, H. Terahertz spectroscopy for space applications: 2.5–2.7 THz spectra of HD, H2O and NH3. J. Mol. Spectrosc. 2011, 1006, 2– 12, DOI: 10.1016/j.molstruc.2011.05.062Google ScholarThere is no corresponding record for this reference.
- 58Cozijn, F. M. J.; Dupré, P.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W. Sub-Doppler Frequency Metrology in HD for Tests of Fundamental Physics. Phys. Rev. Lett. 2018, 120, 153002, DOI: 10.1103/PhysRevLett.120.153002Google ScholarThere is no corresponding record for this reference.
- 59Diouf, M. L.; Cozijn, F. M. J.; Darquié, B.; Salumbides, E. J.; Ubachs, W. Lamb-dips and Lamb-peaks in the saturation spectrum of HD. Opt. Lett. 2019, 44, 4733– 4736, DOI: 10.1364/OL.44.004733Google ScholarThere is no corresponding record for this reference.
- 60Diouf, M. L.; Cozijn, F. M. J.; Lai, K.-F.; Salumbides, E. J.; Ubachs, W. Lamb-peak spectrum of the HD (2–0) P(1) line. Phys. Rev. Res. 2020, 2, 023209, DOI: 10.1103/PhysRevResearch.2.023209Google ScholarThere is no corresponding record for this reference.
- 61Hua, T.-P.; Sun, Y. R.; Hu, S.-M. Dispersion-like lineshape observed in cavity-enhanced saturation spectroscopy of HD at 1.4 μm. Opt. Lett. 2020, 45, 4863– 4866, DOI: 10.1364/OL.401879Google ScholarThere is no corresponding record for this reference.
- 62Wójtewicz, S.; Gotti, R.; Gatti, D.; Lamperti, M.; Laporta, P.; Jóźwiak, H.; Thibault, F.; Wcisło, P.; Marangoni, M. Accurate deuterium spectroscopy and comparison with ab initio calculations. Phys. Rev. A 2020, 101, 052504, DOI: 10.1103/PhysRevA.101.052504Google ScholarThere is no corresponding record for this reference.
- 63Fast, A.; Meek, S. A. Sub-ppb Measurement of a Fundamental Band Rovibrational Transition in HD. Phys. Rev. Lett. 2020, 125, 023001, DOI: 10.1103/PhysRevLett.125.023001Google ScholarThere is no corresponding record for this reference.
- 64Castrillo, A.; Fasci, E.; Gianfrani, L. Doppler-limited precision spectroscopy of HD at 1.4 μm: An improved determination of the R(1) center frequency. Phys. Rev. A 2021, 103, 022828, DOI: 10.1103/PhysRevA.103.022828Google ScholarThere is no corresponding record for this reference.
- 65Fast, A.; Meek, S. A. Precise measurement of the D2 S1(0) vibrational transition frequency. Mol. Phys. 2022, 120, e1999520 DOI: 10.1080/00268976.2021.1999520Google ScholarThere is no corresponding record for this reference.
- 66Kassi, S.; Lauzin, C.; Chaillot, J.; Campargue, A. The (2–0) R(0) and R(1) transition frequencies of HD determined to a 10–10 relative accuracy by Doppler spectroscopy at 80 K. Phys. Chem. Chem. Phys. 2022, 24, 23164– 23172, DOI: 10.1039/D2CP02151JGoogle ScholarThere is no corresponding record for this reference.
- 67Cozijn, F. M. J.; Diouf, M. L.; Hermann, V.; Salumbides, E. J.; Schlösser, M.; Ubachs, W. Rotational level spacings in HD from vibrational saturation spectroscopy. Phys. Rev. A 2022, 105, 062823, DOI: 10.1103/PhysRevA.105.062823Google ScholarThere is no corresponding record for this reference.
- 68Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W. Saturation spectroscopy of R(0), R(2) and P(2) lines in the (2–0) band of HD. Eur. Phys. J. D 2022, 76, 220, DOI: 10.1140/epjd/s10053-022-00552-xGoogle ScholarThere is no corresponding record for this reference.
- 69Fleurbaey, H.; Koroleva, A. O.; Kassi, S.; Campargue, A. The high-accuracy spectroscopy of H2 rovibrational transitions in the (2–0) band near 1.2 μm. Phys. Chem. Chem. Phys. 2023, 25, 14749– 14756, DOI: 10.1039/D3CP01136DGoogle ScholarThere is no corresponding record for this reference.
- 70Lamperti, M.; Rutkowski, L.; Ronchetti, D.; Gatti, D.; Gotti, R.; Cerullo, G.; Thibault, F.; Jóźwiak, H.; Wójtewicz, S.; Masłowski, P.; Wcisło, P.; Polli, D.; Marangoni, M. Stimulated Raman scattering metrology of molecular hydrogen. Commun. Phys. 2023, 6, 67, DOI: 10.1038/s42005-023-01187-zGoogle ScholarThere is no corresponding record for this reference.
- 71Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W. Lamb Dip of a Quadrupole Transition in H2. Phys. Rev. Lett. 2023, 131, 073001, DOI: 10.1103/PhysRevLett.131.073001Google ScholarThere is no corresponding record for this reference.
- 72Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W.; Hermann, V.; Schlösser, M. Precision Measurement of Vibrational Quanta in Tritium Hydride. Phys. Rev. Lett. 2024, 132, 113002, DOI: 10.1103/PhysRevLett.132.113002Google ScholarThere is no corresponding record for this reference.
- 73Stankiewicz, K.; Makowski, M.; Słowinski, M.; Sołtys, K. L.; Bednarski, B.; Jóźwiak, H.; Stolarczyk, N.; Narożnik, M.; Kierski, D.; Wójtewicz, S.; Cygan, A.; Kowzan, G.; Masłowski, P.; Piwinski, M.; Lisak, D.; Wcisło, P. Cavity-enhanced spectroscopy in the deep cryogenic regime – new hydrogen technologies for quantum sensing. arXiv 2025, arXiv:2502.12703, DOI: 10.48550/arXiv.2502.12703Google ScholarThere is no corresponding record for this reference.
- 74Pachucki, K.; Komasa, J. Schrödinger equation solved for the hydrogen molecule with unprecedented accuracy. J. Chem. Phys. 2016, 144, 164306, DOI: 10.1063/1.4948309Google ScholarThere is no corresponding record for this reference.
- 75Pachucki, K.; Komasa, J. Integrals for relativistic nonadiabatic energies of H2 in an exponential basis. Phys. Rev. A 2024, 109, 032822, DOI: 10.1103/PhysRevA.109.032822Google ScholarThere is no corresponding record for this reference.
- 76Pachucki, K.; Komasa, J. Nonrelativistic energy levels of HD. Phys. Chem. Chem. Phys. 2018, 20, 26297– 26302, DOI: 10.1039/C8CP05493BGoogle ScholarThere is no corresponding record for this reference.
- 77Pachucki, K.; Komasa, J. Nonrelativistic energy levels of D2. Phys. Chem. Chem. Phys. 2019, 21, 10272– 10276, DOI: 10.1039/C9CP01308CGoogle ScholarThere is no corresponding record for this reference.
- 78Pachucki, K.; Komasa, J. Nonrelativistic energy of tritium-containing hydrogen molecule isotopologues. Mol. Phys. 2022, 120, e2040627 DOI: 10.1080/00268976.2022.2040627Google ScholarThere is no corresponding record for this reference.
- 79Hylleraas, E. Neue Berechnung der Energie des Heliums im Grundzustande, sowie des tiefsten Terms von Ortho-Helium. Z. Physik 1929, 54, 347– 366, DOI: 10.1007/BF01375457Google ScholarThere is no corresponding record for this reference.
- 80Mohr, P. J.; Newell, D. B.; Taylor, B. N.; Tiesinga, E. CODATA recommended values of the fundamental physical constants: 2022. Rev. Mod. Phys. 2025, 97, 025002, DOI: 10.1103/RevModPhys.97.025002Google ScholarThere is no corresponding record for this reference.
- 81Pachucki, K.; Komasa, J. Relativistic Correction from the Four-Body Nonadiabatic Exponential Wave Function. J. Chem. Theory Comput. 2024, 20, 8644– 8651, DOI: 10.1021/acs.jctc.4c00861Google ScholarThere is no corresponding record for this reference.
- 82Code, R. F.; Ramsey, N. F. Molecular-Beam Magnetic Resonance Studies of HD and D2. Phys. Rev. A 1971, 4, 1945– 1959, DOI: 10.1103/PhysRevA.4.1945Google ScholarThere is no corresponding record for this reference.
- 83Komasa, J. Electron-nucleus Araki-Sucher QED correction to rovibrational levels of H2. arXiv 2025, arXiv:2511.07053v2.Google ScholarThere is no corresponding record for this reference.
- 84Drake, G. W. F.; Swainson, R. A. Bethe logarithms for hydrogen up to n = 20, and approximations for two-electron atoms. Phys. Rev. A 1990, 41, 1243– 1246, DOI: 10.1103/PhysRevA.41.1243Google ScholarThere is no corresponding record for this reference.
- 85Diouf, M. L.; Cozijn, F. M. J.; Ubachs, W. Hyperfine structure in a vibrational quadrupole transition of ortho-H2. Mol. Phys. 2024, 122, e2304101 DOI: 10.1080/00268976.2024.2304101Google ScholarThere is no corresponding record for this reference.
- 86H2Spectre ver. 7.4 Fortran source code. 2022, https://www.fuw.edu.pl/~krp/, https://qcg.home.amu.edu.pl/H2Spectre.html (accessed Oct 10, 2025).Google ScholarThere is no corresponding record for this reference.
- 87Parthey, C. G.; Matveev, A.; Alnis, J.; Bernhardt, B.; Beyer, A.; Holzwarth, R.; Maistrou, A.; Pohl, R.; Predehl, K.; Udem, T.; Wilken, T.; Kolachevsky, N.; Abgrall, M.; Rovera, D.; Salomon, C.; Laurent, P.; Hänsch, T. W. Improved Measurement of the Hydrogen 1S–2S Transition Frequency. Phys. Rev. Lett. 2011, 107, 203001, DOI: 10.1103/PhysRevLett.107.203001Google ScholarThere is no corresponding record for this reference.
- 88Pachucki, K.; Jentschura, U. D. Two-Loop Bethe-Logarithm Correction in Hydrogenlike Atoms. Phys. Rev. Lett. 2003, 91, 113005, DOI: 10.1103/PhysRevLett.91.113005Google ScholarThere is no corresponding record for this reference.
- 89Clausen, G.; Scheidegger, S.; Agner, J. A.; Schmutz, H.; Merkt, F. Imaging-Assisted Single-Photon Doppler-Free Laser Spectroscopy and the Ionization Energy of Metastable Triplet Helium. Phys. Rev. Lett. 2023, 131, 103001, DOI: 10.1103/PhysRevLett.131.103001Google ScholarThere is no corresponding record for this reference.
- 90Alighanbari, S.; Schenkel, M.; Korobov, V.; Schiller, S. High-accuracy laser spectroscopy of and the proton–electron mass ratio. Nature 2025, 644, 69– 75, DOI: 10.1038/s41586-025-09306-2Google ScholarThere is no corresponding record for this reference.
Cited By
This article is cited by 3 publications.
- K. Stankiewicz, M. Makowski, M. Słowiński, K. L. Sołtys, B. Bednarski, H. J. Jóźwiak, N. Stolarczyk, M. Narożnik, D. Kierski, S. Wójtewicz, A. Cygan, G. Kowzan, P. Masłowski, M. Piwiński, D. Lisak, P. Wcisło. Cavity-enhanced spectroscopy in the deep cryogenic regime for quantum sensing and metrology. Nature Physics 2026, 47 https://doi.org/10.1038/s41567-026-03204-8
- Michał Siłkowski, Paweł Czachorowski, Jacek Komasa, Mariusz Puchalski. Progress toward spectroscopic accuracy: Relativistic and QED corrections for
HeH
+
. Physical Review A 2026, 113
(3)
https://doi.org/10.1103/k4qf-thhm
- Jacek Komasa. Electron-nucleus Araki-Sucher correction in the hydrogen molecule isotopologues. Physical Review A 2026, 113
(1)
https://doi.org/10.1103/qyz4-n1tc
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.
Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts.
The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article. Find more information on the Altmetric Attention Score and how the score is calculated.
Recommended Articles
Abstract
Figure 1
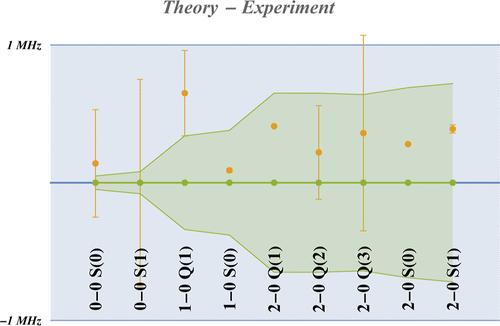
Figure 1. Differences between theoretically predicted and experimental line positions shown against the ±1 MHz error band (in blue) and the theoretical error band (in green). The error bars around the dots come from the experiment.
References
This article references 90 other publications.
- 1Heitler, W.; London, F. Wechselwirkung neutraler Atome und homeopolare Bindung nach der Quantenmechanik. Z. Phys. 1927, 44, 455, DOI: 10.1007/BF01397394There is no corresponding record for this reference.
- 2James, H. M.; Coolidge, A. S. The Ground State of the Hydrogen Molecule. J. Chem. Phys. 1933, 1, 825, DOI: 10.1063/1.1749252There is no corresponding record for this reference.
- 3Kolos, W.; Roothaan, C. C. J. Accurate Electronic Wave Functions for the H2 Molecule. Rev. Mod. Phys. 1960, 32, 219– 232, DOI: 10.1103/RevModPhys.32.219There is no corresponding record for this reference.
- 4Kołos, W.; Wolniewicz, L. The coupling between electronic and nuclear motion and the relativistic effects in the ground state of the H2 molecule. Acta Phys. Polonica 1961, 20, 129– 140There is no corresponding record for this reference.
- 5Kołos, W.; Wolniewicz, L. Accurate Adiabatic Treatment of the Ground State of the Hydrogen Molecule. J. Chem. Phys. 1964, 41, 3663– 3673, DOI: 10.1063/1.1725796There is no corresponding record for this reference.
- 6Kołos, W.; Wolniewicz, L. Accurate Computation of Vibronic Energies and of Some Expectation Values for H2, D2, and T2. J. Chem. Phys. 1964, 41, 3674– 3678, DOI: 10.1063/1.1725797There is no corresponding record for this reference.
- 7Pachucki, K.; Komasa, J. Nonadiabatic corrections to rovibrational levels of H2. J. Chem. Phys. 2009, 130, 164113, DOI: 10.1063/1.3114680There is no corresponding record for this reference.
- 8Pachucki, K.; Komasa, J. Leading order nonadiabatic corrections to rovibrational levels of H2, D2, and T2. J. Chem. Phys. 2015, 143, 034111, DOI: 10.1063/1.4927079There is no corresponding record for this reference.
- 9Kołos, W.; Wolniewicz, L. Nonadiabatic Theory for Diatomic Molecules and Its Application to the Hydrogen Molecule. Rev. Mod. Phys. 1963, 35, 473, DOI: 10.1103/RevModPhys.35.473There is no corresponding record for this reference.
- 10Kinghorn, D. B.; Adamowicz, L. Improved Nonadiabatic Ground-State Energy Upper Bound for Dihydrogen. Phys. Rev. Lett. 1999, 83, 2541– 2543, DOI: 10.1103/PhysRevLett.83.2541There is no corresponding record for this reference.
- 11Puchalski, M.; Komasa, J.; Czachorowski, P.; Pachucki, K. Nonadiabatic QED Correction to the Dissociation Energy of the Hydrogen Molecule. Phys. Rev. Lett. 2019, 122, 103003, DOI: 10.1103/PhysRevLett.122.103003There is no corresponding record for this reference.
- 12Pachucki, K.; Komasa, J. Nonadiabatic rotational states of the hydrogen molecule. Phys. Chem. Chem. Phys. 2018, 20, 247– 255, DOI: 10.1039/C7CP06516GThere is no corresponding record for this reference.
- 13Wolniewicz, L. Relativistic energies of the ground state of the hydrogen molecule. J. Chem. Phys. 1993, 99, 1851, DOI: 10.1063/1.465303There is no corresponding record for this reference.
- 14Piszczatowski, K.; Łach, G.; Przybytek, M.; Komasa, J.; Pachucki, K.; Jeziorski, B. Theoretical Determination of the Dissociation Energy of Molecular Hydrogen. J. Chem. Theory Comput. 2009, 5, 3039, DOI: 10.1021/ct900391pThere is no corresponding record for this reference.
- 15Komasa, J.; Piszczatowski, K.; Łach, G.; Przybytek, M.; Jeziorski, B.; Pachucki, K. Quantum Electrodynamics Effects in Rovibrational Spectra of Molecular Hydrogen. J. Chem. Theory Comput. 2011, 7, 3105– 3115, DOI: 10.1021/ct200438tThere is no corresponding record for this reference.
- 16Puchalski, M.; Komasa, J.; Czachorowski, P.; Pachucki, K. Complete α6 m Corrections to the Ground State of H2. Phys. Rev. Lett. 2016, 117, 263002, DOI: 10.1103/PhysRevLett.117.263002There is no corresponding record for this reference.
- 17Czachorowski, P.; Puchalski, M.; Komasa, J.; Pachucki, K. Nonadiabatic relativistic correction in H2, D2, and HD. Phys. Rev. A 2018, 98, 052506, DOI: 10.1103/PhysRevA.98.052506There is no corresponding record for this reference.
- 18Siłkowski, M.; Pachucki, K.; Komasa, J.; Puchalski, M. Leading-order QED effects in the ground electronic state of molecular hydrogen. Phys. Rev. A 2023, 107, 032807, DOI: 10.1103/PhysRevA.107.032807There is no corresponding record for this reference.
- 19Wang, L. M.; Yan, Z.-C. Relativistic corrections to the ground state of H2 calculated without using the Born-Oppenheimer approximation. Phys. Rev. A 2018, 97, 060501, DOI: 10.1103/PhysRevA.97.060501There is no corresponding record for this reference.
- 20Puchalski, M.; Moszynski, R.; Komasa, J. In Polish Quantum Chemistry from Kołos to Now. In Advances in Quantum Chemistry; Musiał, M., Grabowski, I., Eds.; Academic Press, 2023; Vol. 87, pp 1– 36.There is no corresponding record for this reference.
- 21Pachucki, K.; Komasa, J. Magnetic dipole transitions in the hydrogen molecule. Phys. Rev. A 2011, 83, 032501, DOI: 10.1103/PhysRevA.83.032501There is no corresponding record for this reference.
- 22Margolis, J.; Hunt, G. On the level of H2 quadrupole absorption in the Jovian atmosphere. Icarus 1973, 18, 593– 598, DOI: 10.1016/0019-1035(73)90061-4There is no corresponding record for this reference.
- 23Roueff, E.; Abgrall, H.; Czachorowski, P.; Pachucki, K.; Puchalski, M.; Komasa, J. The full infrared spectrum of molecular hydrogen. Astron. Astrophys. 2019, 630, A58, DOI: 10.1051/0004-6361/201936249There is no corresponding record for this reference.
- 24Tchernyshyov, K. A Detection of H2 in a High-velocity Cloud toward the Large Magellanic Cloud. Astrophys. J. 2022, 931, 78, DOI: 10.3847/1538-4357/ac68e0There is no corresponding record for this reference.
- 25Jacob, A. Small molecules, big impact: a tale of hydrides past, present, and future. Astrophys. Space Sci. 2023, 368, 76, DOI: 10.1007/s10509-023-04229-8There is no corresponding record for this reference.
- 26Tennyson, J. The 2024 release of the ExoMol database: Molecular line lists for exoplanet and other hot atmospheres. J. Quant. Spectrosc. Radiat. Transfer 2024, 326, 109083, DOI: 10.1016/j.jqsrt.2024.109083There is no corresponding record for this reference.
- 27Bruno, D. Uncertainty quantification of a collisional-radiative model for hydrogen plasmas. Phys. Scr. 2025, 100, 035611, DOI: 10.1088/1402-4896/adb227There is no corresponding record for this reference.
- 28Zúñiga, J.; Bastida, A.; Requena, A.; Cerezo, J. Ab Initio Partition Functions and Thermodynamic Quantities for the Molecular Hydrogen Isotopologues. J. Phys. Chem. A 2021, 125, 9226– 9241, DOI: 10.1021/acs.jpca.1c06468There is no corresponding record for this reference.
- 29Leachman, J. W.; Wilhelmsen, Ø.; Matveev, K. I. Cool Fuel: The Science and Engineering of Cryogenic Hydrogen; Oxford University Press, 2025.There is no corresponding record for this reference.
- 30Puchalski, M.; Komasa, J.; Pachucki, K. Hyperfine Structure of the First Rotational Level in H2, D2 and HD Molecules and the Deuteron Quadrupole Moment. Phys. Rev. Lett. 2020, 125, 253001, DOI: 10.1103/PhysRevLett.125.253001There is no corresponding record for this reference.
- 31Puchalski, M.; Komasa, J.; Spyszkiewicz, A.; Pachucki, K. Nuclear magnetic shielding in HD and HT. Phys. Rev. A 2022, 105, 042802, DOI: 10.1103/PhysRevA.105.042802There is no corresponding record for this reference.
- 32Kassi, S.; Campargue, A. Electric quadrupole and dipole transitions of the first overtone band of HD by CRDS between 1.45 and 1.33 μm. J. Mol. Spectrosc. 2011, 267, 36, DOI: 10.1016/j.jms.2011.02.001There is no corresponding record for this reference.
- 33Campargue, A.; Kassi, S.; Pachucki, K.; Komasa, J. The absorption spectrum of H2: CRDS measurements of the (2–0) band, review of the literature data and accurate ab initio line list up to 35000 cm–1. Phys. Chem. Chem. Phys. 2012, 14, 802, DOI: 10.1039/C1CP22912EThere is no corresponding record for this reference.
- 34Kassi, S.; Campargue, A.; Pachucki, K.; Komasa, J. The absorption spectrum of D2: Ultrasensitive cavity ring down spectroscopy of the (2–0) band near 1.7 μm and accurate ab initio line list up to 24 000 cm–1. J. Chem. Phys. 2012, 136, 184309, DOI: 10.1063/1.4707708There is no corresponding record for this reference.
- 35Cheng, C.-F.; Sun, Y. R.; Pan, H.; Wang, J.; Liu, A.-W.; Campargue, A.; Hu, S.-M. Electric-quadrupole transition of H2 determined to 10–9 precision. Phys. Rev. A 2012, 85, 024501, DOI: 10.1103/PhysRevA.85.024501There is no corresponding record for this reference.
- 36Dickenson, G. D.; Niu, M. L.; Salumbides, E. J.; Komasa, J.; Eikema, K. S. E.; Pachucki, K.; Ubachs, W. Fundamental Vibration of Molecular Hydrogen. Phys. Rev. Lett. 2013, 110, 193601, DOI: 10.1103/PhysRevLett.110.193601There is no corresponding record for this reference.
- 37Niu, M.; Salumbides, E.; Dickenson, G.; Eikema, K.; Ubachs, W. Precision spectroscopy of the rovibrational splittings in H2, HD and D2. J. Mol. Spectrosc. 2014, 300, 44– 54, DOI: 10.1016/j.jms.2014.03.011There is no corresponding record for this reference.
- 38Tan, Y.; Wang, J.; Cheng, C.-F.; Zhao, X.-Q.; Liu, A.-W.; Hu, S.-M. Cavity ring-down spectroscopy of the electric quadrupole transitions of in the 784–852 nm region. J. Mol. Spectrosc. 2014, 300, 60– 64, DOI: 10.1016/j.jms.2014.03.010There is no corresponding record for this reference.
- 39Kassi, S.; Campargue, A. Electric quadrupole transitions and collision-induced absorption in the region of the first overtone band of H2 near 1.25μm. J. Mol. Spectrosc. 2014, 300, 55– 59, DOI: 10.1016/j.jms.2014.03.022There is no corresponding record for this reference.
- 40Mondelain, D.; Kassi, S.; Sala, T.; Romanini, D.; Gatti, D.; Campargue, A. Sub-MHz accuracy measurement of the S(2) 2–0 transition frequency of D2 by Comb-Assisted Cavity Ring Down spectroscopy. J. Mol. Spectrosc. 2016, 326, 5– 8, DOI: 10.1016/j.jms.2016.02.008There is no corresponding record for this reference.
- 41Wcisło, P.; Thibault, F.; Zaborowski, M.; Wójtewicz, S.; Cygan, A.; Kowzan, G.; Masłowski, P.; Komasa, J.; Puchalski, M.; Pachucki, K.; Ciuryło, R.; Lisak, D. Accurate deuterium spectroscopy for fundamental studies. J. Quant. Spectrosc. Radiat. Transfer 2018, 213, 41– 51, DOI: 10.1016/j.jqsrt.2018.04.011There is no corresponding record for this reference.
- 42Trivikram, T. M.; Schlösser, M.; Ubachs, W.; Salumbides, E. J. Relativistic and QED Effects in the Fundamental Vibration of T2. Phys. Rev. Lett. 2018, 120, 163002, DOI: 10.1103/PhysRevLett.120.163002There is no corresponding record for this reference.
- 43Tao, L.-G.; Liu, A.-W.; Pachucki, K.; Komasa, J.; Sun, Y. R.; Wang, J.; Hu, S.-M. Toward a Determination of the Proton-Electron Mass Ratio from the Lamb-Dip Measurement of HD. Phys. Rev. Lett. 2018, 120, 153001, DOI: 10.1103/PhysRevLett.120.153001There is no corresponding record for this reference.
- 44Fasci, E.; Castrillo, A.; Dinesan, H.; Gravina, S.; Moretti, L.; Gianfrani, L. Precision spectroscopy of HD at 1.38μm. Phys. Rev. A 2018, 98, 022516, DOI: 10.1103/PhysRevA.98.022516There is no corresponding record for this reference.
- 45Martinez, R. Z.; Bermejo, D.; Wcisło, P.; Thibault, F. Accurate wavenumber measurements for the S0(0), S0(1), and S0(2) pure rotational Raman lines of D2. J. Raman Spectrosc. 2019, 50, 127– 129, DOI: 10.1002/jrs.5499There is no corresponding record for this reference.
- 46Hölsch, N.; Beyer, M.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W.; Jungen, C.; Merkt, F. Benchmarking Theory with an Improved Measurement of the Ionization and Dissociation Energies of H2. Phys. Rev. Lett. 2019, 122, 103002, DOI: 10.1103/PhysRevLett.122.103002There is no corresponding record for this reference.
- 47Beyer, M.; Hölsch, N.; Hussels, J.; Cheng, C.-F.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W.; Jungen, C.; Merkt, F. Determination of the Interval between the Ground States of Para- and Ortho-H2. Phys. Rev. Lett. 2019, 123, 163002, DOI: 10.1103/PhysRevLett.123.163002There is no corresponding record for this reference.
- 48Lai, K.-F.; Czachorowski, P.; Schlösser, M.; Puchalski, M.; Komasa, J.; Pachucki, K.; Ubachs, W.; Salumbides, E. J. Precision tests of nonadiabatic perturbation theory with measurements on the DT molecule. Phys. Rev. Res. 2019, 1, 033124, DOI: 10.1103/PhysRevResearch.1.033124There is no corresponding record for this reference.
- 49Lai, K.-F.; Hermann, V.; Trivikram, M.; Diouf, M.; Schlösser, M.; Ubachs, W.; Salumbides, E. Precision measurement of the fundamental vibrational frequencies of tritium-bearing hydrogen molecules: T2, DT, HT. Phys. Chem. Chem. Phys. 2020, 22, 8973– 8987, DOI: 10.1039/D0CP00596GThere is no corresponding record for this reference.
- 50Mondelain, D.; Kassi, S.; Campargue, A. Transition frequencies in the (2–0) band of D2 with MHz accuracy. J. Quant. Spectrosc. Radiat. Transfer 2020, 253, 107020, DOI: 10.1016/j.jqsrt.2020.107020There is no corresponding record for this reference.
- 51Jóźwiak, H.; Cybulski, H.; Wcisło, P. Hyperfine structure of quadrupole rovibrational transitions in tritium-bearing hydrogen isotopologues. J. Quant. Spectrosc. Radiat. Transfer 2020, 256, 107255, DOI: 10.1016/j.jqsrt.2020.107255There is no corresponding record for this reference.
- 52Kassi, S.; Fleurbaey, H.; Campargue, A. First detection and absolute transition frequencies in the (3–0) band of D2. J. Chem. Phys. 2024, 160, 094306, DOI: 10.1063/5.0196903There is no corresponding record for this reference.
- 53Salumbides, E. J.; Dickenson, G. D.; Ivanov, T. I.; Ubachs, W. QED Effects in Molecules: Test on Rotational Quantum States of H2. Phys. Rev. Lett. 2011, 107, 043005, DOI: 10.1103/PhysRevLett.107.043005There is no corresponding record for this reference.
- 54Salumbides, E. J.; Koelemeij, J. C. J.; Komasa, J.; Pachucki, K.; Eikema, K. S. E.; Ubachs, W. Bounds on fifth forces from precision measurements on molecules. Phys. Rev. D 2013, 87, 112008, DOI: 10.1103/PhysRevD.87.112008There is no corresponding record for this reference.
- 55Salumbides, E. J.; Schellekens, A. N.; Gato-Rivera, B.; Ubachs, W. Constraints on extra dimensions from precision molecular spectroscopy. New J. Phys. 2015, 17, 033015, DOI: 10.1088/1367-2630/17/3/033015There is no corresponding record for this reference.
- 56Ubachs, W.; Koelemeij, J.; Eikema, K.; Salumbides, E. Physics beyond the Standard Model from hydrogen spectroscopy. J. Mol. Spectrosc. 2016, 320, 1– 12, DOI: 10.1016/j.jms.2015.12.003There is no corresponding record for this reference.
- 57Drouin, B. J.; Yu, S.; Pearson, J. C.; Gupta, H. Terahertz spectroscopy for space applications: 2.5–2.7 THz spectra of HD, H2O and NH3. J. Mol. Spectrosc. 2011, 1006, 2– 12, DOI: 10.1016/j.molstruc.2011.05.062There is no corresponding record for this reference.
- 58Cozijn, F. M. J.; Dupré, P.; Salumbides, E. J.; Eikema, K. S. E.; Ubachs, W. Sub-Doppler Frequency Metrology in HD for Tests of Fundamental Physics. Phys. Rev. Lett. 2018, 120, 153002, DOI: 10.1103/PhysRevLett.120.153002There is no corresponding record for this reference.
- 59Diouf, M. L.; Cozijn, F. M. J.; Darquié, B.; Salumbides, E. J.; Ubachs, W. Lamb-dips and Lamb-peaks in the saturation spectrum of HD. Opt. Lett. 2019, 44, 4733– 4736, DOI: 10.1364/OL.44.004733There is no corresponding record for this reference.
- 60Diouf, M. L.; Cozijn, F. M. J.; Lai, K.-F.; Salumbides, E. J.; Ubachs, W. Lamb-peak spectrum of the HD (2–0) P(1) line. Phys. Rev. Res. 2020, 2, 023209, DOI: 10.1103/PhysRevResearch.2.023209There is no corresponding record for this reference.
- 61Hua, T.-P.; Sun, Y. R.; Hu, S.-M. Dispersion-like lineshape observed in cavity-enhanced saturation spectroscopy of HD at 1.4 μm. Opt. Lett. 2020, 45, 4863– 4866, DOI: 10.1364/OL.401879There is no corresponding record for this reference.
- 62Wójtewicz, S.; Gotti, R.; Gatti, D.; Lamperti, M.; Laporta, P.; Jóźwiak, H.; Thibault, F.; Wcisło, P.; Marangoni, M. Accurate deuterium spectroscopy and comparison with ab initio calculations. Phys. Rev. A 2020, 101, 052504, DOI: 10.1103/PhysRevA.101.052504There is no corresponding record for this reference.
- 63Fast, A.; Meek, S. A. Sub-ppb Measurement of a Fundamental Band Rovibrational Transition in HD. Phys. Rev. Lett. 2020, 125, 023001, DOI: 10.1103/PhysRevLett.125.023001There is no corresponding record for this reference.
- 64Castrillo, A.; Fasci, E.; Gianfrani, L. Doppler-limited precision spectroscopy of HD at 1.4 μm: An improved determination of the R(1) center frequency. Phys. Rev. A 2021, 103, 022828, DOI: 10.1103/PhysRevA.103.022828There is no corresponding record for this reference.
- 65Fast, A.; Meek, S. A. Precise measurement of the D2 S1(0) vibrational transition frequency. Mol. Phys. 2022, 120, e1999520 DOI: 10.1080/00268976.2021.1999520There is no corresponding record for this reference.
- 66Kassi, S.; Lauzin, C.; Chaillot, J.; Campargue, A. The (2–0) R(0) and R(1) transition frequencies of HD determined to a 10–10 relative accuracy by Doppler spectroscopy at 80 K. Phys. Chem. Chem. Phys. 2022, 24, 23164– 23172, DOI: 10.1039/D2CP02151JThere is no corresponding record for this reference.
- 67Cozijn, F. M. J.; Diouf, M. L.; Hermann, V.; Salumbides, E. J.; Schlösser, M.; Ubachs, W. Rotational level spacings in HD from vibrational saturation spectroscopy. Phys. Rev. A 2022, 105, 062823, DOI: 10.1103/PhysRevA.105.062823There is no corresponding record for this reference.
- 68Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W. Saturation spectroscopy of R(0), R(2) and P(2) lines in the (2–0) band of HD. Eur. Phys. J. D 2022, 76, 220, DOI: 10.1140/epjd/s10053-022-00552-xThere is no corresponding record for this reference.
- 69Fleurbaey, H.; Koroleva, A. O.; Kassi, S.; Campargue, A. The high-accuracy spectroscopy of H2 rovibrational transitions in the (2–0) band near 1.2 μm. Phys. Chem. Chem. Phys. 2023, 25, 14749– 14756, DOI: 10.1039/D3CP01136DThere is no corresponding record for this reference.
- 70Lamperti, M.; Rutkowski, L.; Ronchetti, D.; Gatti, D.; Gotti, R.; Cerullo, G.; Thibault, F.; Jóźwiak, H.; Wójtewicz, S.; Masłowski, P.; Wcisło, P.; Polli, D.; Marangoni, M. Stimulated Raman scattering metrology of molecular hydrogen. Commun. Phys. 2023, 6, 67, DOI: 10.1038/s42005-023-01187-zThere is no corresponding record for this reference.
- 71Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W. Lamb Dip of a Quadrupole Transition in H2. Phys. Rev. Lett. 2023, 131, 073001, DOI: 10.1103/PhysRevLett.131.073001There is no corresponding record for this reference.
- 72Cozijn, F. M. J.; Diouf, M. L.; Ubachs, W.; Hermann, V.; Schlösser, M. Precision Measurement of Vibrational Quanta in Tritium Hydride. Phys. Rev. Lett. 2024, 132, 113002, DOI: 10.1103/PhysRevLett.132.113002There is no corresponding record for this reference.
- 73Stankiewicz, K.; Makowski, M.; Słowinski, M.; Sołtys, K. L.; Bednarski, B.; Jóźwiak, H.; Stolarczyk, N.; Narożnik, M.; Kierski, D.; Wójtewicz, S.; Cygan, A.; Kowzan, G.; Masłowski, P.; Piwinski, M.; Lisak, D.; Wcisło, P. Cavity-enhanced spectroscopy in the deep cryogenic regime – new hydrogen technologies for quantum sensing. arXiv 2025, arXiv:2502.12703, DOI: 10.48550/arXiv.2502.12703There is no corresponding record for this reference.
- 74Pachucki, K.; Komasa, J. Schrödinger equation solved for the hydrogen molecule with unprecedented accuracy. J. Chem. Phys. 2016, 144, 164306, DOI: 10.1063/1.4948309There is no corresponding record for this reference.
- 75Pachucki, K.; Komasa, J. Integrals for relativistic nonadiabatic energies of H2 in an exponential basis. Phys. Rev. A 2024, 109, 032822, DOI: 10.1103/PhysRevA.109.032822There is no corresponding record for this reference.
- 76Pachucki, K.; Komasa, J. Nonrelativistic energy levels of HD. Phys. Chem. Chem. Phys. 2018, 20, 26297– 26302, DOI: 10.1039/C8CP05493BThere is no corresponding record for this reference.
- 77Pachucki, K.; Komasa, J. Nonrelativistic energy levels of D2. Phys. Chem. Chem. Phys. 2019, 21, 10272– 10276, DOI: 10.1039/C9CP01308CThere is no corresponding record for this reference.
- 78Pachucki, K.; Komasa, J. Nonrelativistic energy of tritium-containing hydrogen molecule isotopologues. Mol. Phys. 2022, 120, e2040627 DOI: 10.1080/00268976.2022.2040627There is no corresponding record for this reference.
- 79Hylleraas, E. Neue Berechnung der Energie des Heliums im Grundzustande, sowie des tiefsten Terms von Ortho-Helium. Z. Physik 1929, 54, 347– 366, DOI: 10.1007/BF01375457There is no corresponding record for this reference.
- 80Mohr, P. J.; Newell, D. B.; Taylor, B. N.; Tiesinga, E. CODATA recommended values of the fundamental physical constants: 2022. Rev. Mod. Phys. 2025, 97, 025002, DOI: 10.1103/RevModPhys.97.025002There is no corresponding record for this reference.
- 81Pachucki, K.; Komasa, J. Relativistic Correction from the Four-Body Nonadiabatic Exponential Wave Function. J. Chem. Theory Comput. 2024, 20, 8644– 8651, DOI: 10.1021/acs.jctc.4c00861There is no corresponding record for this reference.
- 82Code, R. F.; Ramsey, N. F. Molecular-Beam Magnetic Resonance Studies of HD and D2. Phys. Rev. A 1971, 4, 1945– 1959, DOI: 10.1103/PhysRevA.4.1945There is no corresponding record for this reference.
- 83Komasa, J. Electron-nucleus Araki-Sucher QED correction to rovibrational levels of H2. arXiv 2025, arXiv:2511.07053v2.There is no corresponding record for this reference.
- 84Drake, G. W. F.; Swainson, R. A. Bethe logarithms for hydrogen up to n = 20, and approximations for two-electron atoms. Phys. Rev. A 1990, 41, 1243– 1246, DOI: 10.1103/PhysRevA.41.1243There is no corresponding record for this reference.
- 85Diouf, M. L.; Cozijn, F. M. J.; Ubachs, W. Hyperfine structure in a vibrational quadrupole transition of ortho-H2. Mol. Phys. 2024, 122, e2304101 DOI: 10.1080/00268976.2024.2304101There is no corresponding record for this reference.
- 86H2Spectre ver. 7.4 Fortran source code. 2022, https://www.fuw.edu.pl/~krp/, https://qcg.home.amu.edu.pl/H2Spectre.html (accessed Oct 10, 2025).There is no corresponding record for this reference.
- 87Parthey, C. G.; Matveev, A.; Alnis, J.; Bernhardt, B.; Beyer, A.; Holzwarth, R.; Maistrou, A.; Pohl, R.; Predehl, K.; Udem, T.; Wilken, T.; Kolachevsky, N.; Abgrall, M.; Rovera, D.; Salomon, C.; Laurent, P.; Hänsch, T. W. Improved Measurement of the Hydrogen 1S–2S Transition Frequency. Phys. Rev. Lett. 2011, 107, 203001, DOI: 10.1103/PhysRevLett.107.203001There is no corresponding record for this reference.
- 88Pachucki, K.; Jentschura, U. D. Two-Loop Bethe-Logarithm Correction in Hydrogenlike Atoms. Phys. Rev. Lett. 2003, 91, 113005, DOI: 10.1103/PhysRevLett.91.113005There is no corresponding record for this reference.
- 89Clausen, G.; Scheidegger, S.; Agner, J. A.; Schmutz, H.; Merkt, F. Imaging-Assisted Single-Photon Doppler-Free Laser Spectroscopy and the Ionization Energy of Metastable Triplet Helium. Phys. Rev. Lett. 2023, 131, 103001, DOI: 10.1103/PhysRevLett.131.103001There is no corresponding record for this reference.
- 90Alighanbari, S.; Schenkel, M.; Korobov, V.; Schiller, S. High-accuracy laser spectroscopy of and the proton–electron mass ratio. Nature 2025, 644, 69– 75, DOI: 10.1038/s41586-025-09306-2There is no corresponding record for this reference.
Supporting Information
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jctc.5c01702.
Convergence of relativistic expectation values for individual rovibrational levels of H2 (PDF)
Terms & Conditions
Most electronic Supporting Information files are available without a subscription to ACS Web Editions. Such files may be downloaded by article for research use (if there is a public use license linked to the relevant article, that license may permit other uses). Permission may be obtained from ACS for other uses through requests via the RightsLink permission system: http://pubs.acs.org/page/copyright/permissions.html.


